User login
Blog: NIAMS Celebrates 25 Years of Progress
It has been 25 years since the establishment of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and great strides have been made in diagnosis, treatment, and management of numerous conditions, "but you ain't seen nothing yet," said Dr. Francis Collins, director of the National Institutes of Health.
Opportunities for medical research have never been as great as they are today, said Dr. Collins, who gave the welcome address for NIAMS' 25th anniversary at the NIH campus in Bethesda, Md.
Although prominent researchers in the field agreed that research has come a long way in the past 25 years, they stressed that there is still a long way to go. Currently, the molecular basis for 4,000 diseases is known, said Dr. Collins. "But we have effective treatment for only 200."
In broad strokes, the day-long event touched on the past, present, and future of major diseases of bones, joints, muscles, and skin through panels and discussion involving prominent researchers, physicians, and patient advocates.
"These diseases are chronic, crippling, and common," said Dr. Stephen Katz, director of NIAMS, in his opening address. "They affect every family in the United States."
Among the attendees were many researchers and clinicians who said they felt loyalty and appreciation for receiving funding from NIAMS at some point in their career. For some, the progress in the past 2 decades was quite tangible.
"Public investment in osteoporosis research has really changed how we take care of the patients," said Dr. Sundeep Khosla, president of the American Society for Bone and Mineral Research. Dr. Khosla, professor at the Mayo Medical School, Rochester, Minn., recalled a time more than 2 decades ago when calcium, vitamin D, and estrogen were the only options he could offer to patients with osteoporosis.
A few years later, bisphosphonates became available, then came anabolic drugs, and now more drugs are in the pipeline. Patient diagnosis also has advanced, he said. Although he agreed that the field still has a long way to go, he was optimistic about more progress. "Who knows what will happen in the next 25 years," he said.
There was talk of individualized therapy, balancing research and treatment, and a closer collaboration among scientists, all in the spirit of bringing better diagnosis and treatment to patients.
"We're in a different world from when all we had was aspirin," said Dr. Daniel Kastner, a scientific director at the National Human Genome Research Institute. "But what we really want is a cure. And we're not there yet."
--Naseem S. Miller (@NaseemSMiller)
It has been 25 years since the establishment of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and great strides have been made in diagnosis, treatment, and management of numerous conditions, "but you ain't seen nothing yet," said Dr. Francis Collins, director of the National Institutes of Health.
Opportunities for medical research have never been as great as they are today, said Dr. Collins, who gave the welcome address for NIAMS' 25th anniversary at the NIH campus in Bethesda, Md.
Although prominent researchers in the field agreed that research has come a long way in the past 25 years, they stressed that there is still a long way to go. Currently, the molecular basis for 4,000 diseases is known, said Dr. Collins. "But we have effective treatment for only 200."
In broad strokes, the day-long event touched on the past, present, and future of major diseases of bones, joints, muscles, and skin through panels and discussion involving prominent researchers, physicians, and patient advocates.
"These diseases are chronic, crippling, and common," said Dr. Stephen Katz, director of NIAMS, in his opening address. "They affect every family in the United States."
Among the attendees were many researchers and clinicians who said they felt loyalty and appreciation for receiving funding from NIAMS at some point in their career. For some, the progress in the past 2 decades was quite tangible.
"Public investment in osteoporosis research has really changed how we take care of the patients," said Dr. Sundeep Khosla, president of the American Society for Bone and Mineral Research. Dr. Khosla, professor at the Mayo Medical School, Rochester, Minn., recalled a time more than 2 decades ago when calcium, vitamin D, and estrogen were the only options he could offer to patients with osteoporosis.
A few years later, bisphosphonates became available, then came anabolic drugs, and now more drugs are in the pipeline. Patient diagnosis also has advanced, he said. Although he agreed that the field still has a long way to go, he was optimistic about more progress. "Who knows what will happen in the next 25 years," he said.
There was talk of individualized therapy, balancing research and treatment, and a closer collaboration among scientists, all in the spirit of bringing better diagnosis and treatment to patients.
"We're in a different world from when all we had was aspirin," said Dr. Daniel Kastner, a scientific director at the National Human Genome Research Institute. "But what we really want is a cure. And we're not there yet."
--Naseem S. Miller (@NaseemSMiller)
It has been 25 years since the establishment of the National Institute of Arthritis and Musculoskeletal and Skin Diseases, and great strides have been made in diagnosis, treatment, and management of numerous conditions, "but you ain't seen nothing yet," said Dr. Francis Collins, director of the National Institutes of Health.
Opportunities for medical research have never been as great as they are today, said Dr. Collins, who gave the welcome address for NIAMS' 25th anniversary at the NIH campus in Bethesda, Md.
Although prominent researchers in the field agreed that research has come a long way in the past 25 years, they stressed that there is still a long way to go. Currently, the molecular basis for 4,000 diseases is known, said Dr. Collins. "But we have effective treatment for only 200."
In broad strokes, the day-long event touched on the past, present, and future of major diseases of bones, joints, muscles, and skin through panels and discussion involving prominent researchers, physicians, and patient advocates.
"These diseases are chronic, crippling, and common," said Dr. Stephen Katz, director of NIAMS, in his opening address. "They affect every family in the United States."
Among the attendees were many researchers and clinicians who said they felt loyalty and appreciation for receiving funding from NIAMS at some point in their career. For some, the progress in the past 2 decades was quite tangible.
"Public investment in osteoporosis research has really changed how we take care of the patients," said Dr. Sundeep Khosla, president of the American Society for Bone and Mineral Research. Dr. Khosla, professor at the Mayo Medical School, Rochester, Minn., recalled a time more than 2 decades ago when calcium, vitamin D, and estrogen were the only options he could offer to patients with osteoporosis.
A few years later, bisphosphonates became available, then came anabolic drugs, and now more drugs are in the pipeline. Patient diagnosis also has advanced, he said. Although he agreed that the field still has a long way to go, he was optimistic about more progress. "Who knows what will happen in the next 25 years," he said.
There was talk of individualized therapy, balancing research and treatment, and a closer collaboration among scientists, all in the spirit of bringing better diagnosis and treatment to patients.
"We're in a different world from when all we had was aspirin," said Dr. Daniel Kastner, a scientific director at the National Human Genome Research Institute. "But what we really want is a cure. And we're not there yet."
--Naseem S. Miller (@NaseemSMiller)
An Open-Label, Multicenter Study of the Efficacy and Safety of an AM/PM Treatment Regimen With Clobetasol Propionate Spray 0.05% and Calcitriol Ointment 3 μg/g in the Management of Plaque Psoriasis
Topical Combo Shows Early Promise in Moderate Psoriasis
SEOUL, SOUTH KOREA – The combination of calcipotriene and nicotinamide may be an attractive corticosteroid-sparing option for topical psoriasis therapy, according to results from a pilot trial.
"Both agents are somewhat effective for psoriasis; the combination of the two is markedly superior," Dr. Mark Lebwohl said in summarizing the findings of a pilot bilateral comparative study presented at the World Congress of Dermatology.
The investigational topical combination therapy is being developed by Dermipsor, an Israeli pharmaceutical company.
Dr. Lebwohl was a coinvestigator in the multicenter, double-blind, 12-week pilot study involving 168 patients with symmetric moderate psoriasis (defined as not more than 15% body surface area involvement).
The patients were randomized to two of seven treatments: calcipotriene 0.005% monotherapy, nicotinamide 1.4% alone, placebo, or calcipotriene 0.005% plus nicotinamide in a concentration of either 0.05%, 0.1%, 0.7%, or 1.4%. Patients treated the lesions on one side of the body with one of their two assigned treatments, and lesions on the opposite side with the other.
The primary end point was efficacy (defined as a clear to almost-clear outcome at week 12), which was achieved in 19% of patients who were treated with placebo, 25% of those on nicotinamide 1.4% alone, 31.5% of those treated with calcipotriene alone, and 50% of patients in the calcipotriene plus nicotinamide 1.4% group, reported Dr. Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York.
The 50% clear or almost-clear rate with calcipotriene plus nicotinamide 1.4% is similar to the 48% rate of absent to very mild disease that was previously reported with a calcipotriol/betamethasone combination in a 52-week study (Dermatology 2006;213:319-26).
Thus, the calcipotriene/nicotinamide combination is an agent that will be investigated further in larger, lengthier studies as a possible steroid-sparing agent in comparative trials vs. calcipotriene-steroid combination therapy, Dr. Lebwohl said.
Topical combination therapy continues to be an active area in psoriasis research, he observed.
"It's really a no-brainer: The combination of two ingredients is going to be superior to either one alone. It's a very simple concept: Two is better than one," he said.
Dr. Lebwohl disclosed that he is a consultant to Dermipsor and has advisory board relationships with other pharmaceutical companies that are developing dermatologic drugs.
SEOUL, SOUTH KOREA – The combination of calcipotriene and nicotinamide may be an attractive corticosteroid-sparing option for topical psoriasis therapy, according to results from a pilot trial.
"Both agents are somewhat effective for psoriasis; the combination of the two is markedly superior," Dr. Mark Lebwohl said in summarizing the findings of a pilot bilateral comparative study presented at the World Congress of Dermatology.
The investigational topical combination therapy is being developed by Dermipsor, an Israeli pharmaceutical company.
Dr. Lebwohl was a coinvestigator in the multicenter, double-blind, 12-week pilot study involving 168 patients with symmetric moderate psoriasis (defined as not more than 15% body surface area involvement).
The patients were randomized to two of seven treatments: calcipotriene 0.005% monotherapy, nicotinamide 1.4% alone, placebo, or calcipotriene 0.005% plus nicotinamide in a concentration of either 0.05%, 0.1%, 0.7%, or 1.4%. Patients treated the lesions on one side of the body with one of their two assigned treatments, and lesions on the opposite side with the other.
The primary end point was efficacy (defined as a clear to almost-clear outcome at week 12), which was achieved in 19% of patients who were treated with placebo, 25% of those on nicotinamide 1.4% alone, 31.5% of those treated with calcipotriene alone, and 50% of patients in the calcipotriene plus nicotinamide 1.4% group, reported Dr. Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York.
The 50% clear or almost-clear rate with calcipotriene plus nicotinamide 1.4% is similar to the 48% rate of absent to very mild disease that was previously reported with a calcipotriol/betamethasone combination in a 52-week study (Dermatology 2006;213:319-26).
Thus, the calcipotriene/nicotinamide combination is an agent that will be investigated further in larger, lengthier studies as a possible steroid-sparing agent in comparative trials vs. calcipotriene-steroid combination therapy, Dr. Lebwohl said.
Topical combination therapy continues to be an active area in psoriasis research, he observed.
"It's really a no-brainer: The combination of two ingredients is going to be superior to either one alone. It's a very simple concept: Two is better than one," he said.
Dr. Lebwohl disclosed that he is a consultant to Dermipsor and has advisory board relationships with other pharmaceutical companies that are developing dermatologic drugs.
SEOUL, SOUTH KOREA – The combination of calcipotriene and nicotinamide may be an attractive corticosteroid-sparing option for topical psoriasis therapy, according to results from a pilot trial.
"Both agents are somewhat effective for psoriasis; the combination of the two is markedly superior," Dr. Mark Lebwohl said in summarizing the findings of a pilot bilateral comparative study presented at the World Congress of Dermatology.
The investigational topical combination therapy is being developed by Dermipsor, an Israeli pharmaceutical company.
Dr. Lebwohl was a coinvestigator in the multicenter, double-blind, 12-week pilot study involving 168 patients with symmetric moderate psoriasis (defined as not more than 15% body surface area involvement).
The patients were randomized to two of seven treatments: calcipotriene 0.005% monotherapy, nicotinamide 1.4% alone, placebo, or calcipotriene 0.005% plus nicotinamide in a concentration of either 0.05%, 0.1%, 0.7%, or 1.4%. Patients treated the lesions on one side of the body with one of their two assigned treatments, and lesions on the opposite side with the other.
The primary end point was efficacy (defined as a clear to almost-clear outcome at week 12), which was achieved in 19% of patients who were treated with placebo, 25% of those on nicotinamide 1.4% alone, 31.5% of those treated with calcipotriene alone, and 50% of patients in the calcipotriene plus nicotinamide 1.4% group, reported Dr. Lebwohl, professor and chairman of the department of dermatology at Mount Sinai School of Medicine, New York.
The 50% clear or almost-clear rate with calcipotriene plus nicotinamide 1.4% is similar to the 48% rate of absent to very mild disease that was previously reported with a calcipotriol/betamethasone combination in a 52-week study (Dermatology 2006;213:319-26).
Thus, the calcipotriene/nicotinamide combination is an agent that will be investigated further in larger, lengthier studies as a possible steroid-sparing agent in comparative trials vs. calcipotriene-steroid combination therapy, Dr. Lebwohl said.
Topical combination therapy continues to be an active area in psoriasis research, he observed.
"It's really a no-brainer: The combination of two ingredients is going to be superior to either one alone. It's a very simple concept: Two is better than one," he said.
Dr. Lebwohl disclosed that he is a consultant to Dermipsor and has advisory board relationships with other pharmaceutical companies that are developing dermatologic drugs.
FROM THE WORLD CONGRESS OF DERMATOLOGY
Major Finding: In a study of 168 patients, 50% of patients with moderate psoriasis who used a combination of calcipotriene 0.005% plus nicotinamide 1.4% achieved a clear- to-almost-clear outcome at week 12, compared with 19% of patients using placebo.
Data Source: A double-blind, bilateral, comparative 12-week study of 168 patients.
Disclosures: Dr. Lebwohl disclosed that he is a consultant to Dermipsor and has advisory board relationships with other pharmaceutical companies that are developing dermatologic drugs.
DMARDs for Psoriasis May Cut Diabetes Risk
Some disease-modifying antirheumatic drugs that are taken for rheumatoid arthritis or psoriasis appear to reduce the rate of incident diabetes, according to a new report in JAMA.
In a retrospective cohort study of patients who had RA or psoriasis, the use of a tumor necrosis factor (TNF) inhibitor or hydroxychloroquine to treat the systemic inflammatory disorder was associated with a reduced risk of developing diabetes, compared with the use of methotrexate or nonbiologic DMARDs, said Dr. Daniel H. Solomon of the divisions of pharmacoepidemiology and rheumatology at Brigham and Women’s Hospital, Boston, and his associates.
"Considering these results, in light of prior findings regarding improved insulin and glucose metabolism and reduced diabetes risk with hydroxychloroquine and TNF inhibitors, there is evidence suggesting a possible role for DMARDs and immunosuppression in diabetes prevention," they noted.
The investigators assessed the relationship between DMARDs and the risk of new-onset diabetes because previous studies have demonstrated that inflammatory conditions such as RA and psoriasis predispose patients to insulin resistance, and that some of these anti-inflammatory medications appear to improve insulin resistance and prevent the onset of diabetes.
They analyzed information from the databases of a Canadian health care system and a commercial U.S. health plan to identify 13,905 adults with RA or psoriasis who had filled at least one prescription for a DMARD and could be followed for approximately 6 months.
The DMARDs were divided into four mutually exclusive groups: TNF inhibitors such as adalimumab, etanercept, or infliximab; methotrexate; hydroxychloroquine; and other nonbiologic DMARDs such as sulfasalazine, leflunomide, cyclosporine, azathioprine, cyclophosphamide, mycophenolate mofetil, 6-thioguanine, acitretin, d-penicillamine, and the following gold preparations: auranofin, myochrysine, or solganol.
A total of 267 study subjects developed incident diabetes. The incidence was highest among patients who were taking nonbiologic DMARDs.
Patients taking a TNF inhibitor or hydroxychloroquine showed a reduced risk of diabetes, compared with patients taking any other agents. After accounting for the effects of potentially confounding factors such as patient age, sex, and several clinical variables, investigators found that the hazard ratios for diabetes were 0.62 for TNF inhibitors and 0.54 for hydroxychloroquine, compared with the nonbiologic DMARDs, Dr. Solomon and his colleagues said (JAMA 2011;305:2525-31).
"These findings held up across a variety of sensitivity analyses," they added.
"Taken in the context of prior research, [our] study supports the potential role for systemic immunosuppression in prevention and control of diabetes. If future studies show this convincingly, systemic immunosuppression in such situations would be predicated on a favorable risk-benefit profile."
For example, the benefit of immunosuppression may outweigh the risk in a patient with a systemic rheumatic disease for which a DMARD is already indicated. But immunosuppression may not outweigh the risk in a patient who already has diabetes and is prone to infection.
The investigators emphasized that this was an observational epidemiologic study without randomized-treatment assignment, so causation cannot be inferred. "It is possible that patients receiving a TNF inhibitor or hydroxychloroquine were different from the reference group of other nonbiologic DMARD users in ways that went unmeasured, such as body mass index, exercise participation, family history, or disease severity," they noted.
They added that the findings warrant confirmation in a randomized, controlled trial to test "the ability of these agents to prevent diabetes among participants with systemic inflammatory disorders."
This study was supported by Amgen. Dr. Solomon reported ties to Abbott, Amgen, Bristol-Myers Squibb, and Pfizer, and his associates reported ties to numerous industry sources.
"Prospective trials are needed to confirm [these] observational data and clarify which patients may benefit from these possible pleiotropic effects of specific anti-inflammatories," said Dr. Tim Bongartz and Dr. Yogish C. Kudva.
If TNF inhibitors or hydroxychloroquine prove to address two complex disease processes at once, "it will be crucial to investigate how much of their potential antidiabetic effects would add to good disease control, the durability of these effects, and the timing of treatment."
Even if treatment of chronic inflammatory disease can reduce the risk of diabetes, "clinicians still will have to learn how to use specific anti-inflammatory agents to achieve optimal outcomes for both conditions," they said.
Dr. Bongartz is in the division of rheumatology and Dr. Kudva is in the division of endocrinology, diabetes, metabolism, and nutrition at the Mayo Clinic in Rochester, Minn. Dr. Bongartz reported ties to Wyeth and Abbott. Dr. Kudva reported no conflicts of interest. These remarks were taken from their editorial accompanying Dr. Solomon’s report (JAMA 2011;305:2573-4).
"Prospective trials are needed to confirm [these] observational data and clarify which patients may benefit from these possible pleiotropic effects of specific anti-inflammatories," said Dr. Tim Bongartz and Dr. Yogish C. Kudva.
If TNF inhibitors or hydroxychloroquine prove to address two complex disease processes at once, "it will be crucial to investigate how much of their potential antidiabetic effects would add to good disease control, the durability of these effects, and the timing of treatment."
Even if treatment of chronic inflammatory disease can reduce the risk of diabetes, "clinicians still will have to learn how to use specific anti-inflammatory agents to achieve optimal outcomes for both conditions," they said.
Dr. Bongartz is in the division of rheumatology and Dr. Kudva is in the division of endocrinology, diabetes, metabolism, and nutrition at the Mayo Clinic in Rochester, Minn. Dr. Bongartz reported ties to Wyeth and Abbott. Dr. Kudva reported no conflicts of interest. These remarks were taken from their editorial accompanying Dr. Solomon’s report (JAMA 2011;305:2573-4).
"Prospective trials are needed to confirm [these] observational data and clarify which patients may benefit from these possible pleiotropic effects of specific anti-inflammatories," said Dr. Tim Bongartz and Dr. Yogish C. Kudva.
If TNF inhibitors or hydroxychloroquine prove to address two complex disease processes at once, "it will be crucial to investigate how much of their potential antidiabetic effects would add to good disease control, the durability of these effects, and the timing of treatment."
Even if treatment of chronic inflammatory disease can reduce the risk of diabetes, "clinicians still will have to learn how to use specific anti-inflammatory agents to achieve optimal outcomes for both conditions," they said.
Dr. Bongartz is in the division of rheumatology and Dr. Kudva is in the division of endocrinology, diabetes, metabolism, and nutrition at the Mayo Clinic in Rochester, Minn. Dr. Bongartz reported ties to Wyeth and Abbott. Dr. Kudva reported no conflicts of interest. These remarks were taken from their editorial accompanying Dr. Solomon’s report (JAMA 2011;305:2573-4).
Some disease-modifying antirheumatic drugs that are taken for rheumatoid arthritis or psoriasis appear to reduce the rate of incident diabetes, according to a new report in JAMA.
In a retrospective cohort study of patients who had RA or psoriasis, the use of a tumor necrosis factor (TNF) inhibitor or hydroxychloroquine to treat the systemic inflammatory disorder was associated with a reduced risk of developing diabetes, compared with the use of methotrexate or nonbiologic DMARDs, said Dr. Daniel H. Solomon of the divisions of pharmacoepidemiology and rheumatology at Brigham and Women’s Hospital, Boston, and his associates.
"Considering these results, in light of prior findings regarding improved insulin and glucose metabolism and reduced diabetes risk with hydroxychloroquine and TNF inhibitors, there is evidence suggesting a possible role for DMARDs and immunosuppression in diabetes prevention," they noted.
The investigators assessed the relationship between DMARDs and the risk of new-onset diabetes because previous studies have demonstrated that inflammatory conditions such as RA and psoriasis predispose patients to insulin resistance, and that some of these anti-inflammatory medications appear to improve insulin resistance and prevent the onset of diabetes.
They analyzed information from the databases of a Canadian health care system and a commercial U.S. health plan to identify 13,905 adults with RA or psoriasis who had filled at least one prescription for a DMARD and could be followed for approximately 6 months.
The DMARDs were divided into four mutually exclusive groups: TNF inhibitors such as adalimumab, etanercept, or infliximab; methotrexate; hydroxychloroquine; and other nonbiologic DMARDs such as sulfasalazine, leflunomide, cyclosporine, azathioprine, cyclophosphamide, mycophenolate mofetil, 6-thioguanine, acitretin, d-penicillamine, and the following gold preparations: auranofin, myochrysine, or solganol.
A total of 267 study subjects developed incident diabetes. The incidence was highest among patients who were taking nonbiologic DMARDs.
Patients taking a TNF inhibitor or hydroxychloroquine showed a reduced risk of diabetes, compared with patients taking any other agents. After accounting for the effects of potentially confounding factors such as patient age, sex, and several clinical variables, investigators found that the hazard ratios for diabetes were 0.62 for TNF inhibitors and 0.54 for hydroxychloroquine, compared with the nonbiologic DMARDs, Dr. Solomon and his colleagues said (JAMA 2011;305:2525-31).
"These findings held up across a variety of sensitivity analyses," they added.
"Taken in the context of prior research, [our] study supports the potential role for systemic immunosuppression in prevention and control of diabetes. If future studies show this convincingly, systemic immunosuppression in such situations would be predicated on a favorable risk-benefit profile."
For example, the benefit of immunosuppression may outweigh the risk in a patient with a systemic rheumatic disease for which a DMARD is already indicated. But immunosuppression may not outweigh the risk in a patient who already has diabetes and is prone to infection.
The investigators emphasized that this was an observational epidemiologic study without randomized-treatment assignment, so causation cannot be inferred. "It is possible that patients receiving a TNF inhibitor or hydroxychloroquine were different from the reference group of other nonbiologic DMARD users in ways that went unmeasured, such as body mass index, exercise participation, family history, or disease severity," they noted.
They added that the findings warrant confirmation in a randomized, controlled trial to test "the ability of these agents to prevent diabetes among participants with systemic inflammatory disorders."
This study was supported by Amgen. Dr. Solomon reported ties to Abbott, Amgen, Bristol-Myers Squibb, and Pfizer, and his associates reported ties to numerous industry sources.
Some disease-modifying antirheumatic drugs that are taken for rheumatoid arthritis or psoriasis appear to reduce the rate of incident diabetes, according to a new report in JAMA.
In a retrospective cohort study of patients who had RA or psoriasis, the use of a tumor necrosis factor (TNF) inhibitor or hydroxychloroquine to treat the systemic inflammatory disorder was associated with a reduced risk of developing diabetes, compared with the use of methotrexate or nonbiologic DMARDs, said Dr. Daniel H. Solomon of the divisions of pharmacoepidemiology and rheumatology at Brigham and Women’s Hospital, Boston, and his associates.
"Considering these results, in light of prior findings regarding improved insulin and glucose metabolism and reduced diabetes risk with hydroxychloroquine and TNF inhibitors, there is evidence suggesting a possible role for DMARDs and immunosuppression in diabetes prevention," they noted.
The investigators assessed the relationship between DMARDs and the risk of new-onset diabetes because previous studies have demonstrated that inflammatory conditions such as RA and psoriasis predispose patients to insulin resistance, and that some of these anti-inflammatory medications appear to improve insulin resistance and prevent the onset of diabetes.
They analyzed information from the databases of a Canadian health care system and a commercial U.S. health plan to identify 13,905 adults with RA or psoriasis who had filled at least one prescription for a DMARD and could be followed for approximately 6 months.
The DMARDs were divided into four mutually exclusive groups: TNF inhibitors such as adalimumab, etanercept, or infliximab; methotrexate; hydroxychloroquine; and other nonbiologic DMARDs such as sulfasalazine, leflunomide, cyclosporine, azathioprine, cyclophosphamide, mycophenolate mofetil, 6-thioguanine, acitretin, d-penicillamine, and the following gold preparations: auranofin, myochrysine, or solganol.
A total of 267 study subjects developed incident diabetes. The incidence was highest among patients who were taking nonbiologic DMARDs.
Patients taking a TNF inhibitor or hydroxychloroquine showed a reduced risk of diabetes, compared with patients taking any other agents. After accounting for the effects of potentially confounding factors such as patient age, sex, and several clinical variables, investigators found that the hazard ratios for diabetes were 0.62 for TNF inhibitors and 0.54 for hydroxychloroquine, compared with the nonbiologic DMARDs, Dr. Solomon and his colleagues said (JAMA 2011;305:2525-31).
"These findings held up across a variety of sensitivity analyses," they added.
"Taken in the context of prior research, [our] study supports the potential role for systemic immunosuppression in prevention and control of diabetes. If future studies show this convincingly, systemic immunosuppression in such situations would be predicated on a favorable risk-benefit profile."
For example, the benefit of immunosuppression may outweigh the risk in a patient with a systemic rheumatic disease for which a DMARD is already indicated. But immunosuppression may not outweigh the risk in a patient who already has diabetes and is prone to infection.
The investigators emphasized that this was an observational epidemiologic study without randomized-treatment assignment, so causation cannot be inferred. "It is possible that patients receiving a TNF inhibitor or hydroxychloroquine were different from the reference group of other nonbiologic DMARD users in ways that went unmeasured, such as body mass index, exercise participation, family history, or disease severity," they noted.
They added that the findings warrant confirmation in a randomized, controlled trial to test "the ability of these agents to prevent diabetes among participants with systemic inflammatory disorders."
This study was supported by Amgen. Dr. Solomon reported ties to Abbott, Amgen, Bristol-Myers Squibb, and Pfizer, and his associates reported ties to numerous industry sources.
FROM JAMA
Major Finding: The hazard ratios for diabetes were 0.62 for patients taking TNF inhibitors and 0.54 for patients taking hydroxychloroquine, compared with patients taking nonbiologic DMARDS to treat their rheumatoid arthritis or psoriasis.
Data Source: A retrospective observational study involving 13,905 adults who had either RA or psoriasis, received a DMARD, and were followed for approximately 6 months for the development of diabetes. Participants were enrolled in one of two health care systems.
Disclosures: This study was supported by Amgen. Dr. Solomon reported ties to Abbott, Amgen, Bristol-Myers Squibb, and Pfizer, and his associates reported ties to numerous industry sources.
Biologics Don't Increase Herpes Zoster Risk
SEOUL, SOUTH KOREA – Treatment of psoriasis patients with biologic agents isn't associated with increased risk of herpes zoster, according to a large population-based Israeli study.
In contrast, the use of methotrexate, cyclosporine, acitretin, and systemic corticosteroids is associated with an increased risk of herpes zoster in psoriasis patients, Dr. Arnon D. Cohen said at the World Congress of Dermatology.
He reported on 22,330 psoriasis patients followed over 10 years at Clalit Health Services, Israel’s largest HMO. This was a real-world patient population, 53% of whom were female, in contrast to the highly selected populations in randomized clinical trials, which are typically 65%-69% male.
During nearly 220,000 person-years of follow-up there were 1,321 cases of herpes zoster. The baseline rate during periods when patients weren’t on any form of systemic therapy was 4.6 cases/1,000 patient-years. In contrast, the rate during cyclosporine therapy was more than 10-fold higher at 48.4 cases/1,000 patient-years.
Eighteen percent of psoriasis patients were on systemic corticosteroids at some point during the study period; that medication was associated with a herpes zoster rate of 25.7/1,000 patient-years of exposure.
Other therapies that conferred significantly increased risk were methotrexate at 17 cases/1,000 patient-years and acitretin (Soriatane) at 5.4 cases/1,000 patient-years, reported Dr. Cohen, director of the department of quality measures at Clalit and a dermatologist at Ben-Gurion University of the Negev, Beer-Sheva, Israel.
In contrast, no cases of herpes zoster occurred in psoriasis patients on Amevive, Raptiva, or, Humira. Nor was the incidence increased beyond baseline in patients on PUVA, UVB, or Enbrel.
In a , systemic corticosteroid therapy was associated with a 2.4-fold increased risk of developing herpes zoster. Being female was associated with a significant 16% increase in risk. Remicade therapy was associated with a 1.8-fold increase in risk, a trend that did not achieve statistical significance (P =.09).
Dr. Cohen and his coinvestigators conducted this study because, even though the biologic agents are recognized as conferring increased risk of tuberculosis and serious bacterial infections, very little is known about their association, if any, with viral infections.
Dr. Cohen declared having no relevant financial interests.
SEOUL, SOUTH KOREA – Treatment of psoriasis patients with biologic agents isn't associated with increased risk of herpes zoster, according to a large population-based Israeli study.
In contrast, the use of methotrexate, cyclosporine, acitretin, and systemic corticosteroids is associated with an increased risk of herpes zoster in psoriasis patients, Dr. Arnon D. Cohen said at the World Congress of Dermatology.
He reported on 22,330 psoriasis patients followed over 10 years at Clalit Health Services, Israel’s largest HMO. This was a real-world patient population, 53% of whom were female, in contrast to the highly selected populations in randomized clinical trials, which are typically 65%-69% male.
During nearly 220,000 person-years of follow-up there were 1,321 cases of herpes zoster. The baseline rate during periods when patients weren’t on any form of systemic therapy was 4.6 cases/1,000 patient-years. In contrast, the rate during cyclosporine therapy was more than 10-fold higher at 48.4 cases/1,000 patient-years.
Eighteen percent of psoriasis patients were on systemic corticosteroids at some point during the study period; that medication was associated with a herpes zoster rate of 25.7/1,000 patient-years of exposure.
Other therapies that conferred significantly increased risk were methotrexate at 17 cases/1,000 patient-years and acitretin (Soriatane) at 5.4 cases/1,000 patient-years, reported Dr. Cohen, director of the department of quality measures at Clalit and a dermatologist at Ben-Gurion University of the Negev, Beer-Sheva, Israel.
In contrast, no cases of herpes zoster occurred in psoriasis patients on Amevive, Raptiva, or, Humira. Nor was the incidence increased beyond baseline in patients on PUVA, UVB, or Enbrel.
In a , systemic corticosteroid therapy was associated with a 2.4-fold increased risk of developing herpes zoster. Being female was associated with a significant 16% increase in risk. Remicade therapy was associated with a 1.8-fold increase in risk, a trend that did not achieve statistical significance (P =.09).
Dr. Cohen and his coinvestigators conducted this study because, even though the biologic agents are recognized as conferring increased risk of tuberculosis and serious bacterial infections, very little is known about their association, if any, with viral infections.
Dr. Cohen declared having no relevant financial interests.
SEOUL, SOUTH KOREA – Treatment of psoriasis patients with biologic agents isn't associated with increased risk of herpes zoster, according to a large population-based Israeli study.
In contrast, the use of methotrexate, cyclosporine, acitretin, and systemic corticosteroids is associated with an increased risk of herpes zoster in psoriasis patients, Dr. Arnon D. Cohen said at the World Congress of Dermatology.
He reported on 22,330 psoriasis patients followed over 10 years at Clalit Health Services, Israel’s largest HMO. This was a real-world patient population, 53% of whom were female, in contrast to the highly selected populations in randomized clinical trials, which are typically 65%-69% male.
During nearly 220,000 person-years of follow-up there were 1,321 cases of herpes zoster. The baseline rate during periods when patients weren’t on any form of systemic therapy was 4.6 cases/1,000 patient-years. In contrast, the rate during cyclosporine therapy was more than 10-fold higher at 48.4 cases/1,000 patient-years.
Eighteen percent of psoriasis patients were on systemic corticosteroids at some point during the study period; that medication was associated with a herpes zoster rate of 25.7/1,000 patient-years of exposure.
Other therapies that conferred significantly increased risk were methotrexate at 17 cases/1,000 patient-years and acitretin (Soriatane) at 5.4 cases/1,000 patient-years, reported Dr. Cohen, director of the department of quality measures at Clalit and a dermatologist at Ben-Gurion University of the Negev, Beer-Sheva, Israel.
In contrast, no cases of herpes zoster occurred in psoriasis patients on Amevive, Raptiva, or, Humira. Nor was the incidence increased beyond baseline in patients on PUVA, UVB, or Enbrel.
In a , systemic corticosteroid therapy was associated with a 2.4-fold increased risk of developing herpes zoster. Being female was associated with a significant 16% increase in risk. Remicade therapy was associated with a 1.8-fold increase in risk, a trend that did not achieve statistical significance (P =.09).
Dr. Cohen and his coinvestigators conducted this study because, even though the biologic agents are recognized as conferring increased risk of tuberculosis and serious bacterial infections, very little is known about their association, if any, with viral infections.
Dr. Cohen declared having no relevant financial interests.
FROM THE WORLD CONGRESS OF DERMATOLOGY
Major Finding: No cases of herpes zoster occurred in psoriasis patients on alefacept (Amevive), efalizumab (Raptiva) or adalimumab (Humira).
Data Source: A population-based study of 22,330 psoriasis patients followed over 10 years at Clalit Health Services, Israel’s largest HMO.
Disclosures: Dr. Cohen reported having no financial conflicts.
308-nm Excimer Laser Effective for Treating Palmoplantar Psoriasis
GRAPEVINE, TEX. – The 308-nm excimer laser significantly reduced palmoplantar psoriasis severity in a study of 30 patients.
Multiple studies have demonstrated the efficacy of the excimer laser in the treatment of plaque psoriasis, but few have investigated its use for palmoplantar psoriasis. "Palmoplantar psoriasis is difficult to treat and often recalcitrant to traditional therapies such as corticosteroids. Since the excimer laser can selectively treat psoriatic plaque with higher fluences than is tolerated with traditional phototherapy, it could be a therapeutic modality for the thicker skin on the palms and soles," said Dr. David Goldberg, director of dermatologic laser research at Mount Sinai School of Medicine, New York.
The 30 patients were aged 18-75 years, with mild to severe psoriasis on their hands and/or feet. All discontinued other treatments 4 weeks prior to starting the study. By study design, patients could receive up to 16 biweekly laser treatments over the course of 3 months with an excimer laser (PHAROS EX-308, RA Medical Systems). Fluences ranged from 400-600 mJ/cm2 depending on disease severity. Short pulses were delivered with a flexible handpiece, with a maximal output of 15 mJ/pulse (J. Cosmet. Laser Ther. 2011;13:47-9).
Each treatment was tailored to the individual patient’s response from the previous session. If there was no response or minimal erythema, the dose was increased by 30%. If there was moderate erythema, the dose was increased by 20%, and if significant erythema, the dose was increased by 10% until the patient could not tolerate further increases. For severe reactions or blistering, the dose was decreased by 20%. The number of sessions ranged from 7 to 14, with a mean of 11.
All of the subjects had some improvement by week 5, as measured by the Psoriasis Area and Severity Index (PASI). At the end of the treatments, all showed 50%-100% reductions in scaling, erythema, and flattened plaques. No patient had a relapse detected at the 3-month follow-up, but two-thirds of the patients had relapses by 6 months. There was no evidence of persistent pigmentary changes or scarring. Periodic retreatments will be required, Dr. Goldberg said.
"Although no treatment can be expected to ‘cure’ palmoplantar psoriasis, our data do support the use of the excimer laser to treat patients with hand and foot psoriasis. The excimer laser should be strongly considered for patients with palmoplantar psoriasis unresponsive to other treatments," he concluded.
The excimer laser study was provided on loan by the manufacturer during the course of the study. Dr. Goldberg stated that he had no other disclosures.
GRAPEVINE, TEX. – The 308-nm excimer laser significantly reduced palmoplantar psoriasis severity in a study of 30 patients.
Multiple studies have demonstrated the efficacy of the excimer laser in the treatment of plaque psoriasis, but few have investigated its use for palmoplantar psoriasis. "Palmoplantar psoriasis is difficult to treat and often recalcitrant to traditional therapies such as corticosteroids. Since the excimer laser can selectively treat psoriatic plaque with higher fluences than is tolerated with traditional phototherapy, it could be a therapeutic modality for the thicker skin on the palms and soles," said Dr. David Goldberg, director of dermatologic laser research at Mount Sinai School of Medicine, New York.
The 30 patients were aged 18-75 years, with mild to severe psoriasis on their hands and/or feet. All discontinued other treatments 4 weeks prior to starting the study. By study design, patients could receive up to 16 biweekly laser treatments over the course of 3 months with an excimer laser (PHAROS EX-308, RA Medical Systems). Fluences ranged from 400-600 mJ/cm2 depending on disease severity. Short pulses were delivered with a flexible handpiece, with a maximal output of 15 mJ/pulse (J. Cosmet. Laser Ther. 2011;13:47-9).
Each treatment was tailored to the individual patient’s response from the previous session. If there was no response or minimal erythema, the dose was increased by 30%. If there was moderate erythema, the dose was increased by 20%, and if significant erythema, the dose was increased by 10% until the patient could not tolerate further increases. For severe reactions or blistering, the dose was decreased by 20%. The number of sessions ranged from 7 to 14, with a mean of 11.
All of the subjects had some improvement by week 5, as measured by the Psoriasis Area and Severity Index (PASI). At the end of the treatments, all showed 50%-100% reductions in scaling, erythema, and flattened plaques. No patient had a relapse detected at the 3-month follow-up, but two-thirds of the patients had relapses by 6 months. There was no evidence of persistent pigmentary changes or scarring. Periodic retreatments will be required, Dr. Goldberg said.
"Although no treatment can be expected to ‘cure’ palmoplantar psoriasis, our data do support the use of the excimer laser to treat patients with hand and foot psoriasis. The excimer laser should be strongly considered for patients with palmoplantar psoriasis unresponsive to other treatments," he concluded.
The excimer laser study was provided on loan by the manufacturer during the course of the study. Dr. Goldberg stated that he had no other disclosures.
GRAPEVINE, TEX. – The 308-nm excimer laser significantly reduced palmoplantar psoriasis severity in a study of 30 patients.
Multiple studies have demonstrated the efficacy of the excimer laser in the treatment of plaque psoriasis, but few have investigated its use for palmoplantar psoriasis. "Palmoplantar psoriasis is difficult to treat and often recalcitrant to traditional therapies such as corticosteroids. Since the excimer laser can selectively treat psoriatic plaque with higher fluences than is tolerated with traditional phototherapy, it could be a therapeutic modality for the thicker skin on the palms and soles," said Dr. David Goldberg, director of dermatologic laser research at Mount Sinai School of Medicine, New York.
The 30 patients were aged 18-75 years, with mild to severe psoriasis on their hands and/or feet. All discontinued other treatments 4 weeks prior to starting the study. By study design, patients could receive up to 16 biweekly laser treatments over the course of 3 months with an excimer laser (PHAROS EX-308, RA Medical Systems). Fluences ranged from 400-600 mJ/cm2 depending on disease severity. Short pulses were delivered with a flexible handpiece, with a maximal output of 15 mJ/pulse (J. Cosmet. Laser Ther. 2011;13:47-9).
Each treatment was tailored to the individual patient’s response from the previous session. If there was no response or minimal erythema, the dose was increased by 30%. If there was moderate erythema, the dose was increased by 20%, and if significant erythema, the dose was increased by 10% until the patient could not tolerate further increases. For severe reactions or blistering, the dose was decreased by 20%. The number of sessions ranged from 7 to 14, with a mean of 11.
All of the subjects had some improvement by week 5, as measured by the Psoriasis Area and Severity Index (PASI). At the end of the treatments, all showed 50%-100% reductions in scaling, erythema, and flattened plaques. No patient had a relapse detected at the 3-month follow-up, but two-thirds of the patients had relapses by 6 months. There was no evidence of persistent pigmentary changes or scarring. Periodic retreatments will be required, Dr. Goldberg said.
"Although no treatment can be expected to ‘cure’ palmoplantar psoriasis, our data do support the use of the excimer laser to treat patients with hand and foot psoriasis. The excimer laser should be strongly considered for patients with palmoplantar psoriasis unresponsive to other treatments," he concluded.
The excimer laser study was provided on loan by the manufacturer during the course of the study. Dr. Goldberg stated that he had no other disclosures.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR LASER MEDICINE AND SURGERY
Major Finding: At the end of the treatments, all patients showed 50%-100% reductions in scaling, erythema, and flattened plaques
Data Source: The 30 patients were aged 18-75 years, with mild to severe psoriasis on their hands and/or feet.
Disclosures: The excimer laser study was provided on-loan by the manufacturer during the course of the study. Dr. Goldberg stated that he had no other disclosures.
Rituximab Deemed Useful for Certain Lupus Subgroups
DESTIN, FLA. – Rituximab has a place in the treatment of certain subsets of patients with lupus, but generally the biologic agent has proved disappointing for this disease, according to Dr. R. John Looney.
The chimeric monoclonal antibody against the protein CD20 has worked very well in certain autoimmune diseases, and it is approved for a number of indications including refractory rheumatoid arthritis and antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis.
Rituximab is commonly used in relapsing and remitting multiple sclerosis, and early trial data suggest that the agent may have promise in the management of type 1 diabetes, said Dr. Looney, a professor of medicine at the University of Rochester (N.Y.).
In the "amazingly negative" EXPLORER (A Study to Evaluate the Efficacy and Safety of Rituximab in Patients With Severe Systemic Lupus Erythematosus) trial, however, overall clinical response rates did not differ significantly in rituximab- and placebo-treated nonrenal lupus patients (Arthritis Rheum. 2010;62:222-33).
And in the LUNAR (A Study to Evaluate the Efficacy and Safety of Rituximab in Subjects With ISN/RPS Class III or IV Lupus Nephritis) trial, patients with lupus nephritis who were treated with rituximab had a modest improvement in proteinuria, compared with those who received placebo, but only a small, non–statistically significant improvement in overall renal outcomes of about 11%, compared with those who received placebo, according to data reported at conferences, including the annual conference of the American College of Rheumatology in 2009, Dr. Looney said.
"So rituximab has been a remarkably effective drug in certain types of autoimmune disease, but it has been a relative failure in lupus," he added.
An exception is in patients with antibodies associated with neuromyelitis optica, he said.
Neuromyelitis optica (NMO), which can also present as a primary disease, is remarkably responsive to rituximab, and there is a subset of lupus patients who have the NMO IgG/aquaporin-4 antibody that is associated with the disease, he said.
"If you’re looking at a patient with lupus who develops optic neuritis or if they develop transverse myelitis, you have to think about whether they are in a subgroup with these NMO antibodies," he said, noting that these patients will tend to have longitudinally extensive transverse myelitis that extends over three vertebral bodies, and they will tend to be positive for aquaporin-4.
"In this subgroup, we have moved to using rituximab early in the course of disease if [these patients] have been very refractory to other treatments," he said.
Dr. Looney said he also continues to use rituximab in severe refractory systemic lupus erythematosus, particularly in patients with central nervous system disease or lupus nephritis, as well as in patients with refractory idiopathic thrombocytopenic purpura (ITP).
Although rituximab is not approved for ITP, it is widely used as a secondary therapy for this condition, he said.
Dr. Looney disclosed that he has been an advisor for Genentech.
DESTIN, FLA. – Rituximab has a place in the treatment of certain subsets of patients with lupus, but generally the biologic agent has proved disappointing for this disease, according to Dr. R. John Looney.
The chimeric monoclonal antibody against the protein CD20 has worked very well in certain autoimmune diseases, and it is approved for a number of indications including refractory rheumatoid arthritis and antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis.
Rituximab is commonly used in relapsing and remitting multiple sclerosis, and early trial data suggest that the agent may have promise in the management of type 1 diabetes, said Dr. Looney, a professor of medicine at the University of Rochester (N.Y.).
In the "amazingly negative" EXPLORER (A Study to Evaluate the Efficacy and Safety of Rituximab in Patients With Severe Systemic Lupus Erythematosus) trial, however, overall clinical response rates did not differ significantly in rituximab- and placebo-treated nonrenal lupus patients (Arthritis Rheum. 2010;62:222-33).
And in the LUNAR (A Study to Evaluate the Efficacy and Safety of Rituximab in Subjects With ISN/RPS Class III or IV Lupus Nephritis) trial, patients with lupus nephritis who were treated with rituximab had a modest improvement in proteinuria, compared with those who received placebo, but only a small, non–statistically significant improvement in overall renal outcomes of about 11%, compared with those who received placebo, according to data reported at conferences, including the annual conference of the American College of Rheumatology in 2009, Dr. Looney said.
"So rituximab has been a remarkably effective drug in certain types of autoimmune disease, but it has been a relative failure in lupus," he added.
An exception is in patients with antibodies associated with neuromyelitis optica, he said.
Neuromyelitis optica (NMO), which can also present as a primary disease, is remarkably responsive to rituximab, and there is a subset of lupus patients who have the NMO IgG/aquaporin-4 antibody that is associated with the disease, he said.
"If you’re looking at a patient with lupus who develops optic neuritis or if they develop transverse myelitis, you have to think about whether they are in a subgroup with these NMO antibodies," he said, noting that these patients will tend to have longitudinally extensive transverse myelitis that extends over three vertebral bodies, and they will tend to be positive for aquaporin-4.
"In this subgroup, we have moved to using rituximab early in the course of disease if [these patients] have been very refractory to other treatments," he said.
Dr. Looney said he also continues to use rituximab in severe refractory systemic lupus erythematosus, particularly in patients with central nervous system disease or lupus nephritis, as well as in patients with refractory idiopathic thrombocytopenic purpura (ITP).
Although rituximab is not approved for ITP, it is widely used as a secondary therapy for this condition, he said.
Dr. Looney disclosed that he has been an advisor for Genentech.
DESTIN, FLA. – Rituximab has a place in the treatment of certain subsets of patients with lupus, but generally the biologic agent has proved disappointing for this disease, according to Dr. R. John Looney.
The chimeric monoclonal antibody against the protein CD20 has worked very well in certain autoimmune diseases, and it is approved for a number of indications including refractory rheumatoid arthritis and antineutrophil cytoplasmic antibody (ANCA)–associated vasculitis.
Rituximab is commonly used in relapsing and remitting multiple sclerosis, and early trial data suggest that the agent may have promise in the management of type 1 diabetes, said Dr. Looney, a professor of medicine at the University of Rochester (N.Y.).
In the "amazingly negative" EXPLORER (A Study to Evaluate the Efficacy and Safety of Rituximab in Patients With Severe Systemic Lupus Erythematosus) trial, however, overall clinical response rates did not differ significantly in rituximab- and placebo-treated nonrenal lupus patients (Arthritis Rheum. 2010;62:222-33).
And in the LUNAR (A Study to Evaluate the Efficacy and Safety of Rituximab in Subjects With ISN/RPS Class III or IV Lupus Nephritis) trial, patients with lupus nephritis who were treated with rituximab had a modest improvement in proteinuria, compared with those who received placebo, but only a small, non–statistically significant improvement in overall renal outcomes of about 11%, compared with those who received placebo, according to data reported at conferences, including the annual conference of the American College of Rheumatology in 2009, Dr. Looney said.
"So rituximab has been a remarkably effective drug in certain types of autoimmune disease, but it has been a relative failure in lupus," he added.
An exception is in patients with antibodies associated with neuromyelitis optica, he said.
Neuromyelitis optica (NMO), which can also present as a primary disease, is remarkably responsive to rituximab, and there is a subset of lupus patients who have the NMO IgG/aquaporin-4 antibody that is associated with the disease, he said.
"If you’re looking at a patient with lupus who develops optic neuritis or if they develop transverse myelitis, you have to think about whether they are in a subgroup with these NMO antibodies," he said, noting that these patients will tend to have longitudinally extensive transverse myelitis that extends over three vertebral bodies, and they will tend to be positive for aquaporin-4.
"In this subgroup, we have moved to using rituximab early in the course of disease if [these patients] have been very refractory to other treatments," he said.
Dr. Looney said he also continues to use rituximab in severe refractory systemic lupus erythematosus, particularly in patients with central nervous system disease or lupus nephritis, as well as in patients with refractory idiopathic thrombocytopenic purpura (ITP).
Although rituximab is not approved for ITP, it is widely used as a secondary therapy for this condition, he said.
Dr. Looney disclosed that he has been an advisor for Genentech.
EXPERT ANALYSIS FROM THE CONGRESS OF CLINICAL RHEUMATOLOGY
Bosentan May Reduce Skin Fibrosis in Scleroderma
DESTIN, FLA. – Bosentan, which has been shown to prevent digital ulcers in systemic sclerosis, also may have beneficial effects on skin fibrosis.
In a small, prospective, open-label study, treatment with the dual endothelin receptor antagonist was associated with a significant reduction in skin thickening as measured by the modified Rodnan skin score (mRSS), Dr. Annegret Kuhn said at the Congress of Clinical Rheumatology.
The mean change from baseline in the mRSS in the 10 systemic sclerosis patients in the study was 6.4 points on the 0-51 point scale, said Dr. Kuhn of Westfälische Wilhelms-Universität Münster (Germany).
Significant responses were seen in patients with both diffuse and limited disease, and there also was significant healing of digital ulcers. No differences were seen from baseline and week 24, however, on 20-MHz ultrasound, fist-closure evaluation, U.K. systemic sclerosis functional score, or the modified sclerosis health assessment questionnaire (SHAQ) and its visual analogue scale (Rheumatology [Oxford] 2010;49:1336-45).
Patients were treated with 62.5 mg of bosentan twice daily for 4 weeks, then with 125 mg twice daily for 20 weeks. This regimen is similar to the dosing used in the RAPIDS-1 and -2 (Randomized Placebo-Controlled Study on Prevention of Ischemic Digital Ulcers in Systemic Scleroderma–1 and –2) trials, which demonstrated the drug's efficacy for the prevention of digital ulcers. Bosentan is currently approved for use in the United States for pulmonary artery hypertension, and – based on the RAPIDS-1 trial (Arthritis Rheum. 2004;50:3985-93) – it was also approved in Europe in 2007 for prevention of digital ulcers.
The RAPIDS-2 trial that was published this year (Ann. Rheum. Dis. 2011;70:32-8), included treatment for at least 24 weeks, and confirmed the preventive effects seen in RAPIDS-1. Bosentan was not found in either trial to be associated with improved healing in existing ulcers, however.
"Iloprost remains the best treatment for digital ulcers in systemic scleroderma," Dr. Kuhn said,
But bosentan is recommended for prevention, and it may also improve fibrosis, she noted.
Guidelines that were developed this year by a group of German experts on scleroderma call for first-line treatment with iloprost for the healing of existing ulcers, with repeated infusions for long-term care. They also call for the use of bosentan for prevention, and note that current experimental treatments include sildenafil and atorvastatin, she said.
Treatment of underlying disease (such as Raynaud's phenomenon, vasculopathy, and the like) is also important, as are general preventive measures including nicotine abstinence, cold protection, physical treatments, lymphatic draining, elimination of sympathomimetics, and vasoconstrictors.
Topical treatments can include antiseptic agents, becaplermin gel, hydrocolloid membrane, topical lidocaine, and vitamin E gel, according to the guidelines.
Dr. Kuhn had no disclosures to report.
DESTIN, FLA. – Bosentan, which has been shown to prevent digital ulcers in systemic sclerosis, also may have beneficial effects on skin fibrosis.
In a small, prospective, open-label study, treatment with the dual endothelin receptor antagonist was associated with a significant reduction in skin thickening as measured by the modified Rodnan skin score (mRSS), Dr. Annegret Kuhn said at the Congress of Clinical Rheumatology.
The mean change from baseline in the mRSS in the 10 systemic sclerosis patients in the study was 6.4 points on the 0-51 point scale, said Dr. Kuhn of Westfälische Wilhelms-Universität Münster (Germany).
Significant responses were seen in patients with both diffuse and limited disease, and there also was significant healing of digital ulcers. No differences were seen from baseline and week 24, however, on 20-MHz ultrasound, fist-closure evaluation, U.K. systemic sclerosis functional score, or the modified sclerosis health assessment questionnaire (SHAQ) and its visual analogue scale (Rheumatology [Oxford] 2010;49:1336-45).
Patients were treated with 62.5 mg of bosentan twice daily for 4 weeks, then with 125 mg twice daily for 20 weeks. This regimen is similar to the dosing used in the RAPIDS-1 and -2 (Randomized Placebo-Controlled Study on Prevention of Ischemic Digital Ulcers in Systemic Scleroderma–1 and –2) trials, which demonstrated the drug's efficacy for the prevention of digital ulcers. Bosentan is currently approved for use in the United States for pulmonary artery hypertension, and – based on the RAPIDS-1 trial (Arthritis Rheum. 2004;50:3985-93) – it was also approved in Europe in 2007 for prevention of digital ulcers.
The RAPIDS-2 trial that was published this year (Ann. Rheum. Dis. 2011;70:32-8), included treatment for at least 24 weeks, and confirmed the preventive effects seen in RAPIDS-1. Bosentan was not found in either trial to be associated with improved healing in existing ulcers, however.
"Iloprost remains the best treatment for digital ulcers in systemic scleroderma," Dr. Kuhn said,
But bosentan is recommended for prevention, and it may also improve fibrosis, she noted.
Guidelines that were developed this year by a group of German experts on scleroderma call for first-line treatment with iloprost for the healing of existing ulcers, with repeated infusions for long-term care. They also call for the use of bosentan for prevention, and note that current experimental treatments include sildenafil and atorvastatin, she said.
Treatment of underlying disease (such as Raynaud's phenomenon, vasculopathy, and the like) is also important, as are general preventive measures including nicotine abstinence, cold protection, physical treatments, lymphatic draining, elimination of sympathomimetics, and vasoconstrictors.
Topical treatments can include antiseptic agents, becaplermin gel, hydrocolloid membrane, topical lidocaine, and vitamin E gel, according to the guidelines.
Dr. Kuhn had no disclosures to report.
DESTIN, FLA. – Bosentan, which has been shown to prevent digital ulcers in systemic sclerosis, also may have beneficial effects on skin fibrosis.
In a small, prospective, open-label study, treatment with the dual endothelin receptor antagonist was associated with a significant reduction in skin thickening as measured by the modified Rodnan skin score (mRSS), Dr. Annegret Kuhn said at the Congress of Clinical Rheumatology.
The mean change from baseline in the mRSS in the 10 systemic sclerosis patients in the study was 6.4 points on the 0-51 point scale, said Dr. Kuhn of Westfälische Wilhelms-Universität Münster (Germany).
Significant responses were seen in patients with both diffuse and limited disease, and there also was significant healing of digital ulcers. No differences were seen from baseline and week 24, however, on 20-MHz ultrasound, fist-closure evaluation, U.K. systemic sclerosis functional score, or the modified sclerosis health assessment questionnaire (SHAQ) and its visual analogue scale (Rheumatology [Oxford] 2010;49:1336-45).
Patients were treated with 62.5 mg of bosentan twice daily for 4 weeks, then with 125 mg twice daily for 20 weeks. This regimen is similar to the dosing used in the RAPIDS-1 and -2 (Randomized Placebo-Controlled Study on Prevention of Ischemic Digital Ulcers in Systemic Scleroderma–1 and –2) trials, which demonstrated the drug's efficacy for the prevention of digital ulcers. Bosentan is currently approved for use in the United States for pulmonary artery hypertension, and – based on the RAPIDS-1 trial (Arthritis Rheum. 2004;50:3985-93) – it was also approved in Europe in 2007 for prevention of digital ulcers.
The RAPIDS-2 trial that was published this year (Ann. Rheum. Dis. 2011;70:32-8), included treatment for at least 24 weeks, and confirmed the preventive effects seen in RAPIDS-1. Bosentan was not found in either trial to be associated with improved healing in existing ulcers, however.
"Iloprost remains the best treatment for digital ulcers in systemic scleroderma," Dr. Kuhn said,
But bosentan is recommended for prevention, and it may also improve fibrosis, she noted.
Guidelines that were developed this year by a group of German experts on scleroderma call for first-line treatment with iloprost for the healing of existing ulcers, with repeated infusions for long-term care. They also call for the use of bosentan for prevention, and note that current experimental treatments include sildenafil and atorvastatin, she said.
Treatment of underlying disease (such as Raynaud's phenomenon, vasculopathy, and the like) is also important, as are general preventive measures including nicotine abstinence, cold protection, physical treatments, lymphatic draining, elimination of sympathomimetics, and vasoconstrictors.
Topical treatments can include antiseptic agents, becaplermin gel, hydrocolloid membrane, topical lidocaine, and vitamin E gel, according to the guidelines.
Dr. Kuhn had no disclosures to report.
EXPERT ANALYSIS FROM THE CONGRESS OF CLINICAL RHEUMATOLOGY
Patient Subgroup Response to Belimumab Remains Unclear
DESTIN, FLA. – For now, the lupus patients most likely to respond to recently approved belimumab may be those with moderate to severe disease without renal or central nervous system involvement who have failed to respond to standard therapy, according to Dr. R. John Looney.
More specific details on which patients are most likely to benefit will require further experience with the drug, he said at the Congress of Clinical Rheumatology.
Belimumab (Benlysta), which was approved in March, is the first new drug for systemic lupus erythematosus (SLE) in 50 years. It has a very good safety profile, although the effect size, while statistically significant, is relatively small, with only an average of about a 12 percentage point difference in the response rates between treatment and placebo in trials of the fully human monoclonal antibody that inhibits B-lymphocyte stimulator, said Dr. Looney, professor of medicine at the University of Rochester (N.Y.).
Still, that reflects improvement of about 25%-30%, he noted.
The standard dosage is 10 mg/kg, given intravenously every 2 weeks for three times, then every 4 weeks, in addition to standard care.
Keep in mind that the onset is somewhat slow, so this "may not be a drug you would use in a flaring patient," Dr. Looney said.
Also, it has not been studied in severe renal or central nervous system disease, nor in combination with cyclophosphamide, he noted.
Furthermore, no particular subgroup has yet been identified in whom the drug has particular benefits, so it is a bit of a challenge to determine who should be put on this drug.
"I would consider it in patients with moderate to severe, nonrenal, nonneurologic disease who have failed standard therapy," Dr. Looney said, explaining that he would consider those to be patients with continued disease activity or patients in whom steroids can’t be tapered or stopped despite treatment.
"So I look at this as a drug that might help me get my patients who are on hydroxychloroquine and let's say CellCept [mycophenolate] or Imuran [azathioprine], down to a manageable dose of steroids," he added.
Determining how long to treat, however, is a challenge.
"We have no good way to look at an individual patient and say they are better and they are better because they are on belimumab," Dr. Looney said, noting that based on belimumab trial data, three out of four patients who were responders at 1 year would have been responders on placebo, so further study is needed to help guide treatment duration.
"I would also really like to see predictors of response," he said, noting that such predictors have thus far not been reported.
Further analysis of existing data may be useful for identifying subgroups who will respond best to belimumab. Additional studies in renal and CNS disease would also be useful.
"It may be that you add this drug to current therapies [in renal and CNS patients] and they do that much better," Dr. Looney said, noting that it would also be useful to look at belimumab treatment in conjunction with cyclophosphamide and rituximab as well "to see what happens when B cells are targeted in two different ways."
Dr. Looney disclosed that he has been an adviser for Genentech.
DESTIN, FLA. – For now, the lupus patients most likely to respond to recently approved belimumab may be those with moderate to severe disease without renal or central nervous system involvement who have failed to respond to standard therapy, according to Dr. R. John Looney.
More specific details on which patients are most likely to benefit will require further experience with the drug, he said at the Congress of Clinical Rheumatology.
Belimumab (Benlysta), which was approved in March, is the first new drug for systemic lupus erythematosus (SLE) in 50 years. It has a very good safety profile, although the effect size, while statistically significant, is relatively small, with only an average of about a 12 percentage point difference in the response rates between treatment and placebo in trials of the fully human monoclonal antibody that inhibits B-lymphocyte stimulator, said Dr. Looney, professor of medicine at the University of Rochester (N.Y.).
Still, that reflects improvement of about 25%-30%, he noted.
The standard dosage is 10 mg/kg, given intravenously every 2 weeks for three times, then every 4 weeks, in addition to standard care.
Keep in mind that the onset is somewhat slow, so this "may not be a drug you would use in a flaring patient," Dr. Looney said.
Also, it has not been studied in severe renal or central nervous system disease, nor in combination with cyclophosphamide, he noted.
Furthermore, no particular subgroup has yet been identified in whom the drug has particular benefits, so it is a bit of a challenge to determine who should be put on this drug.
"I would consider it in patients with moderate to severe, nonrenal, nonneurologic disease who have failed standard therapy," Dr. Looney said, explaining that he would consider those to be patients with continued disease activity or patients in whom steroids can’t be tapered or stopped despite treatment.
"So I look at this as a drug that might help me get my patients who are on hydroxychloroquine and let's say CellCept [mycophenolate] or Imuran [azathioprine], down to a manageable dose of steroids," he added.
Determining how long to treat, however, is a challenge.
"We have no good way to look at an individual patient and say they are better and they are better because they are on belimumab," Dr. Looney said, noting that based on belimumab trial data, three out of four patients who were responders at 1 year would have been responders on placebo, so further study is needed to help guide treatment duration.
"I would also really like to see predictors of response," he said, noting that such predictors have thus far not been reported.
Further analysis of existing data may be useful for identifying subgroups who will respond best to belimumab. Additional studies in renal and CNS disease would also be useful.
"It may be that you add this drug to current therapies [in renal and CNS patients] and they do that much better," Dr. Looney said, noting that it would also be useful to look at belimumab treatment in conjunction with cyclophosphamide and rituximab as well "to see what happens when B cells are targeted in two different ways."
Dr. Looney disclosed that he has been an adviser for Genentech.
DESTIN, FLA. – For now, the lupus patients most likely to respond to recently approved belimumab may be those with moderate to severe disease without renal or central nervous system involvement who have failed to respond to standard therapy, according to Dr. R. John Looney.
More specific details on which patients are most likely to benefit will require further experience with the drug, he said at the Congress of Clinical Rheumatology.
Belimumab (Benlysta), which was approved in March, is the first new drug for systemic lupus erythematosus (SLE) in 50 years. It has a very good safety profile, although the effect size, while statistically significant, is relatively small, with only an average of about a 12 percentage point difference in the response rates between treatment and placebo in trials of the fully human monoclonal antibody that inhibits B-lymphocyte stimulator, said Dr. Looney, professor of medicine at the University of Rochester (N.Y.).
Still, that reflects improvement of about 25%-30%, he noted.
The standard dosage is 10 mg/kg, given intravenously every 2 weeks for three times, then every 4 weeks, in addition to standard care.
Keep in mind that the onset is somewhat slow, so this "may not be a drug you would use in a flaring patient," Dr. Looney said.
Also, it has not been studied in severe renal or central nervous system disease, nor in combination with cyclophosphamide, he noted.
Furthermore, no particular subgroup has yet been identified in whom the drug has particular benefits, so it is a bit of a challenge to determine who should be put on this drug.
"I would consider it in patients with moderate to severe, nonrenal, nonneurologic disease who have failed standard therapy," Dr. Looney said, explaining that he would consider those to be patients with continued disease activity or patients in whom steroids can’t be tapered or stopped despite treatment.
"So I look at this as a drug that might help me get my patients who are on hydroxychloroquine and let's say CellCept [mycophenolate] or Imuran [azathioprine], down to a manageable dose of steroids," he added.
Determining how long to treat, however, is a challenge.
"We have no good way to look at an individual patient and say they are better and they are better because they are on belimumab," Dr. Looney said, noting that based on belimumab trial data, three out of four patients who were responders at 1 year would have been responders on placebo, so further study is needed to help guide treatment duration.
"I would also really like to see predictors of response," he said, noting that such predictors have thus far not been reported.
Further analysis of existing data may be useful for identifying subgroups who will respond best to belimumab. Additional studies in renal and CNS disease would also be useful.
"It may be that you add this drug to current therapies [in renal and CNS patients] and they do that much better," Dr. Looney said, noting that it would also be useful to look at belimumab treatment in conjunction with cyclophosphamide and rituximab as well "to see what happens when B cells are targeted in two different ways."
Dr. Looney disclosed that he has been an adviser for Genentech.
EXPERT ANALYSIS FROM THE CONGRESS OF CLINICAL RHEUMATOLOGY
Clobex Followed by Vectical Wallops Psoriasis
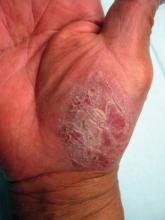
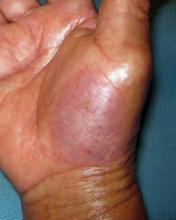
Sequential topical therapy with clobetasol propionate spray followed by calcitriol ointment has joined the ranks of therapies that can boast documented safety and efficacy for moderate to severe plaque psoriasis.
The treatment strategy here is to wallop the disease initially with the super-high-potency corticosteroid to quickly quell an emerging flare, then turn to the safer, slower-acting metabolically active topical vitamin D to reduce the frequency of recurrent flare-ups.
This strategy proved effective and was well tolerated in a recent open-label multicenter study involving 170 patients. They received clobetasol propionate 0.05% spray (Clobex) twice daily for 4 weeks, then calcitriol 3 mcg/g (Vectical) twice daily for the next 8 weeks.
Eighty percent of patients were rated clear, almost clear, or mild at week 2, as were 93% at week 4, 92% at week 8, and 74% at week 12. Treatment success, defined as at least a one grade improvement in a five-point overall disease severity score at week 12, was accomplished in 84% of subjects. Body surface area involvement decreased significantly from 7.1% at baseline to 3.9% at week 12.
Side effects consisted mainly of mild telangiectasias and stinging and burning. Less than 1% of subjects had signs of skin atrophy.
This sequential regimen is attractive because patients only have to remember to take one drug at a time, and also because topical vitamin D and its analogs are at present the only available agents targeting the abnormal keratinocyte differentiation that's a core feature of psoriasis, Dr. James Q. Del Rosso noted at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation.
"Abnormal differentiation of keratinocytes is the one place where we have only one therapy. You really have to decide if that's important enough to you that you think it needs to be built into your treatment of psoriasis pretty much all of the time. You have to decide that for yourself," said Dr. Del Rosso of the University of Nevada, Las Vegas.
Dr. Del Rosso is a consultant to a number of pharmaceutical companies, including Galderma Laboratories, which funded the recently published open-label study (J. Drugs Dermatol. 2011;10:158-64).
SDEF and this publication are owned by Elsevier.
Sequential topical therapy with clobetasol propionate spray followed by calcitriol ointment has joined the ranks of therapies that can boast documented safety and efficacy for moderate to severe plaque psoriasis.
The treatment strategy here is to wallop the disease initially with the super-high-potency corticosteroid to quickly quell an emerging flare, then turn to the safer, slower-acting metabolically active topical vitamin D to reduce the frequency of recurrent flare-ups.
This strategy proved effective and was well tolerated in a recent open-label multicenter study involving 170 patients. They received clobetasol propionate 0.05% spray (Clobex) twice daily for 4 weeks, then calcitriol 3 mcg/g (Vectical) twice daily for the next 8 weeks.
Eighty percent of patients were rated clear, almost clear, or mild at week 2, as were 93% at week 4, 92% at week 8, and 74% at week 12. Treatment success, defined as at least a one grade improvement in a five-point overall disease severity score at week 12, was accomplished in 84% of subjects. Body surface area involvement decreased significantly from 7.1% at baseline to 3.9% at week 12.
Side effects consisted mainly of mild telangiectasias and stinging and burning. Less than 1% of subjects had signs of skin atrophy.
This sequential regimen is attractive because patients only have to remember to take one drug at a time, and also because topical vitamin D and its analogs are at present the only available agents targeting the abnormal keratinocyte differentiation that's a core feature of psoriasis, Dr. James Q. Del Rosso noted at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation.
"Abnormal differentiation of keratinocytes is the one place where we have only one therapy. You really have to decide if that's important enough to you that you think it needs to be built into your treatment of psoriasis pretty much all of the time. You have to decide that for yourself," said Dr. Del Rosso of the University of Nevada, Las Vegas.
Dr. Del Rosso is a consultant to a number of pharmaceutical companies, including Galderma Laboratories, which funded the recently published open-label study (J. Drugs Dermatol. 2011;10:158-64).
SDEF and this publication are owned by Elsevier.
Sequential topical therapy with clobetasol propionate spray followed by calcitriol ointment has joined the ranks of therapies that can boast documented safety and efficacy for moderate to severe plaque psoriasis.
The treatment strategy here is to wallop the disease initially with the super-high-potency corticosteroid to quickly quell an emerging flare, then turn to the safer, slower-acting metabolically active topical vitamin D to reduce the frequency of recurrent flare-ups.
This strategy proved effective and was well tolerated in a recent open-label multicenter study involving 170 patients. They received clobetasol propionate 0.05% spray (Clobex) twice daily for 4 weeks, then calcitriol 3 mcg/g (Vectical) twice daily for the next 8 weeks.
Eighty percent of patients were rated clear, almost clear, or mild at week 2, as were 93% at week 4, 92% at week 8, and 74% at week 12. Treatment success, defined as at least a one grade improvement in a five-point overall disease severity score at week 12, was accomplished in 84% of subjects. Body surface area involvement decreased significantly from 7.1% at baseline to 3.9% at week 12.
Side effects consisted mainly of mild telangiectasias and stinging and burning. Less than 1% of subjects had signs of skin atrophy.
This sequential regimen is attractive because patients only have to remember to take one drug at a time, and also because topical vitamin D and its analogs are at present the only available agents targeting the abnormal keratinocyte differentiation that's a core feature of psoriasis, Dr. James Q. Del Rosso noted at the annual Hawaii Dermatology Seminar sponsored by Skin Disease Education Foundation.
"Abnormal differentiation of keratinocytes is the one place where we have only one therapy. You really have to decide if that's important enough to you that you think it needs to be built into your treatment of psoriasis pretty much all of the time. You have to decide that for yourself," said Dr. Del Rosso of the University of Nevada, Las Vegas.
Dr. Del Rosso is a consultant to a number of pharmaceutical companies, including Galderma Laboratories, which funded the recently published open-label study (J. Drugs Dermatol. 2011;10:158-64).
SDEF and this publication are owned by Elsevier.
FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Major Finding: Eighty percent of patients were rated clear, almost clear, or mild at week 2, as were 93% at week 4, 92% at week 8, and 74% at week 12.
Data Source: An open-label multicenter study involving 170 patients.
Disclosures: Dr. Del Rosso is a consultant to a number of pharmaceutical companies, including Galderma Laboratories, which funded the study.