User login
Cutis is a peer-reviewed clinical journal for the dermatologist, allergist, and general practitioner published monthly since 1965. Concise clinical articles present the practical side of dermatology, helping physicians to improve patient care. Cutis is referenced in Index Medicus/MEDLINE and is written and edited by industry leaders.
ass lick
assault rifle
balls
ballsac
black jack
bleach
Boko Haram
bondage
causas
cheap
child abuse
cocaine
compulsive behaviors
cost of miracles
cunt
Daech
display network stats
drug paraphernalia
explosion
fart
fda and death
fda AND warn
fda AND warning
fda AND warns
feom
fuck
gambling
gfc
gun
human trafficking
humira AND expensive
illegal
ISIL
ISIS
Islamic caliphate
Islamic state
madvocate
masturbation
mixed martial arts
MMA
molestation
national rifle association
NRA
nsfw
nuccitelli
pedophile
pedophilia
poker
porn
porn
pornography
psychedelic drug
recreational drug
sex slave rings
shit
slot machine
snort
substance abuse
terrorism
terrorist
texarkana
Texas hold 'em
UFC
section[contains(@class, 'nav-hidden')]
section[contains(@class, 'nav-hidden active')
A peer-reviewed, indexed journal for dermatologists with original research, image quizzes, cases and reviews, and columns.
Assessing Inpatient Dermatology Availability in Virginia
Assessing Inpatient Dermatology Availability in Virginia
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
Assessing Inpatient Dermatology Availability in Virginia
Assessing Inpatient Dermatology Availability in Virginia
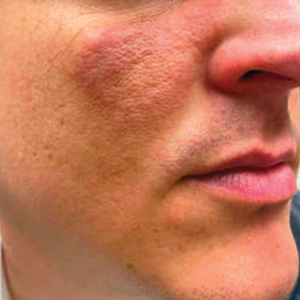
Progressive Erythematous Facial Rash
Progressive Erythematous Facial Rash
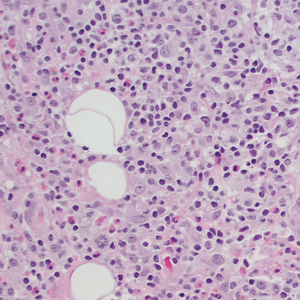
THE DIAGNOSIS: Follicular Mucinosis
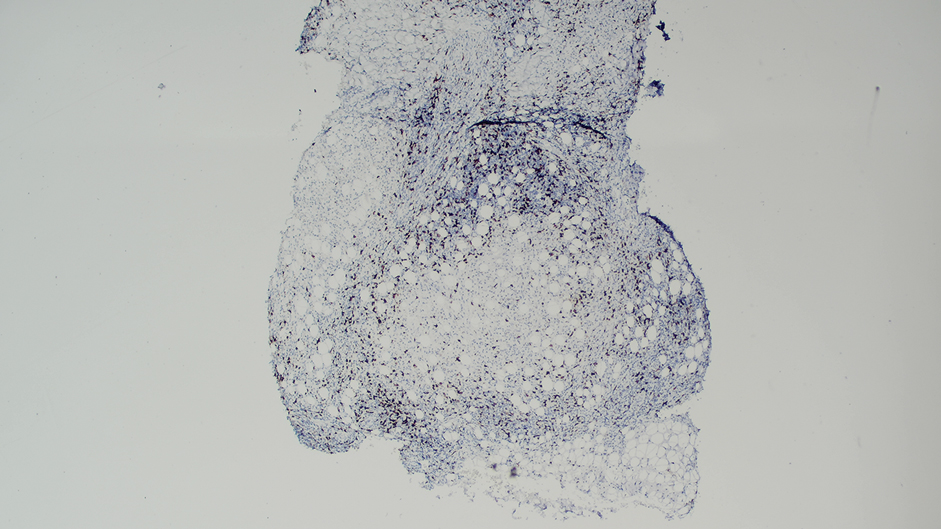
Histologic examination of the hematoxylin and eosin–stained sections of the biopsy revealed an overall moderately dense, perivascular, and perifollicular lymphocytic infiltrate with follicular intraepidermal mucin (Figure). Immunohistochemical staining showed that the lymphocytic infiltrate was predominantly CD4+ over CD8+, with moderate loss of CD7 and absence of CD20 expression. Positive T-cell receptor (TCR) gene rearrangements were detected for both TCRγ and TCRΒ. The clinical features along with the histopathologic findings suggested a diagnosis of follicular mucinosis (FM) with concern in the differential for folliculotropic mycosis fungoides.
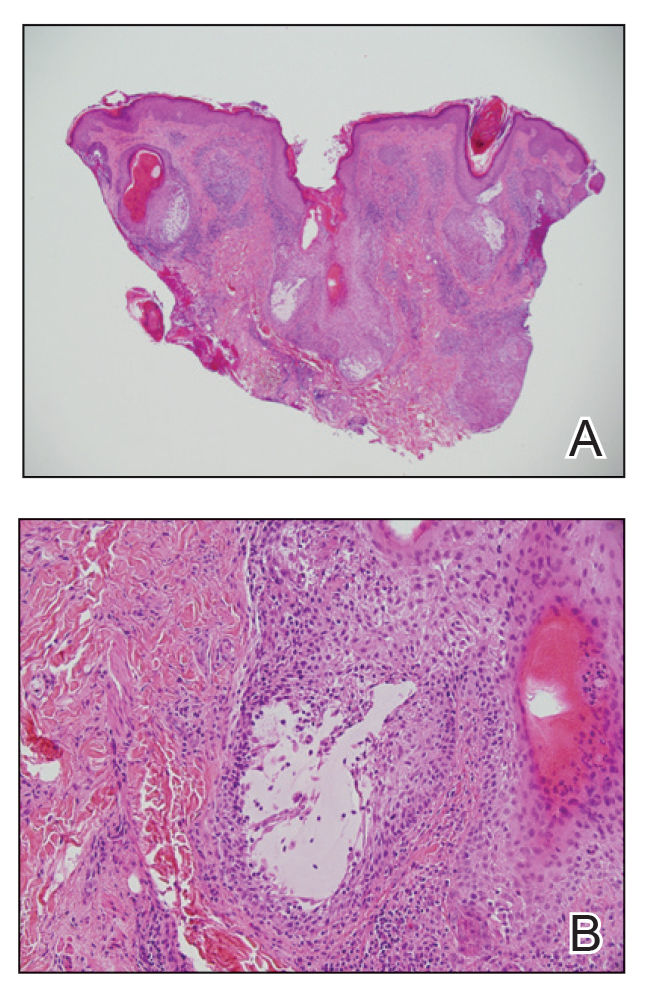
Follicular mucinosis, also known as alopecia mucinosa, is an uncommon inflammatory disorder characterized by follicular degeneration due to the accumulation of mucin within the pilosebaceous unit.1 This condition manifests clinically as indurated plaques and/or follicular papules most often on the face, neck, and scalp.2 It is further categorized as primary vs secondary FM. Primary idiopathic FM, which can further be subdivided into acute or chronic, tends to follow a more benign course, whereas secondary FM usually is associated with underlying inflammatory or neoplastic conditions, most commonly mycosis fungoides, a cutaneous T-cell lymphoma.1,2 In cases of secondary FM, treatment of the underlying cause often leads to resolution of symptoms. Regular follow-up is warranted in either classification.1,3
The initial differential diagnosis for this patient included contact dermatitis associated with mask use, with possible underlying seborrheic dermatitis or rosacea; however, the rash persisted and worsened after treatment with topical triamcinolone and ketoconazole. After the diagnosis of FM was made, the patient was started on topical betamethasone and tacrolimus with good response.
A referral to hematology/oncology revealed that the patient had primary FM and possible stage 1A folliculotropic mycosis fungoides with limited skin involvement (<10% body surface area). On physical examination, no palpable cervical or axillary lymphadenopathy were noted. Flow cytometry for lymphoma was negative with no lymphoid or blast population detected. Laboratory workup and positron emission tomography/computed tomography were unremarkable. The patient had rapid improvement with a more potent topical steroid but also was given tacrolimus ointment 0.1% for residual findings. His disease remained stable without progression at 1-year follow-up.
Contact dermatitis typically manifests as an eczematous eruption that appears on an anatomic location that was exposed to or came into contact with allergens or irritants.4 Contact dermatitis was less likely in our patient due to the lack of acute or subacute spongiosis and lymphocyte exocytosis. Rosacea is a chronic inflammatory dermatosis that presents as recurrent episodes of flushing or transient erythema, persistent erythema, phyphymatous changes, papules, pustules, and telangiectasia5; however, rosacea was less likely in our patient due to the histopathologic and immunohistochemical findings that were suggestive of FM on punch biopsy. Cutaneous lupus generally is associated with photosensitivity and manifests as erythema over the malar eminences and bridge of the nose with sparing of the nasolabial folds.6 Seborrheic dermatitis manifests as erythematous macules or patches with scale and associated pruritis on the scalp, eyebrows, eyelids, and nasolabial folds.7 This condition was less likely in our patient due to the persistence and worsening of the facial erythematous dermatitis despite the use of ketoconazole cream as well as no evidence of spongiosis, shoulder parakeratosis, vascular changes, or presence of microorganisms such as Malassezia species.
Due to the relatively rare nature of this condition as well as a wide variety of other more common etiologies for an erythematous dermatitis of the cheeks, the diagnosis of FM may be delayed or missed entirely. Physicians must have a high index of suspicion to diagnose properly and biopsy if necessary. This photoquiz serves as an important reminder to physicians to keep uncommon diseases on their differential, especially when the patient’s symptoms do not respond to treatment.
- Khalil J, Kurban M, Abbas O. Follicular mucinosis: a review. Int J Dermatol. 2021;60:159-165.
- Akinsanya AO, Tschen JA. Follicular mucinosis: a case report. Cureus. 2019;11:E4746.
- Miyagaki T. Diagnosis of early mycosis fungoides. Diagnostics (Basel). 2021;1:1721.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192.
- van Zuuren EJ, Arents BWM, van der Linden MMD, et al. Rosacea: new concepts in classification and treatment. Am J Clin Dermatol. 2021;22:457-465.
- Rothfield N, Sontheimer RD, Bernstein M. Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol. 2006;24:348-362.
- Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169.
THE DIAGNOSIS: Follicular Mucinosis
Histologic examination of the hematoxylin and eosin–stained sections of the biopsy revealed an overall moderately dense, perivascular, and perifollicular lymphocytic infiltrate with follicular intraepidermal mucin (Figure). Immunohistochemical staining showed that the lymphocytic infiltrate was predominantly CD4+ over CD8+, with moderate loss of CD7 and absence of CD20 expression. Positive T-cell receptor (TCR) gene rearrangements were detected for both TCRγ and TCRΒ. The clinical features along with the histopathologic findings suggested a diagnosis of follicular mucinosis (FM) with concern in the differential for folliculotropic mycosis fungoides.

Follicular mucinosis, also known as alopecia mucinosa, is an uncommon inflammatory disorder characterized by follicular degeneration due to the accumulation of mucin within the pilosebaceous unit.1 This condition manifests clinically as indurated plaques and/or follicular papules most often on the face, neck, and scalp.2 It is further categorized as primary vs secondary FM. Primary idiopathic FM, which can further be subdivided into acute or chronic, tends to follow a more benign course, whereas secondary FM usually is associated with underlying inflammatory or neoplastic conditions, most commonly mycosis fungoides, a cutaneous T-cell lymphoma.1,2 In cases of secondary FM, treatment of the underlying cause often leads to resolution of symptoms. Regular follow-up is warranted in either classification.1,3
The initial differential diagnosis for this patient included contact dermatitis associated with mask use, with possible underlying seborrheic dermatitis or rosacea; however, the rash persisted and worsened after treatment with topical triamcinolone and ketoconazole. After the diagnosis of FM was made, the patient was started on topical betamethasone and tacrolimus with good response.
A referral to hematology/oncology revealed that the patient had primary FM and possible stage 1A folliculotropic mycosis fungoides with limited skin involvement (<10% body surface area). On physical examination, no palpable cervical or axillary lymphadenopathy were noted. Flow cytometry for lymphoma was negative with no lymphoid or blast population detected. Laboratory workup and positron emission tomography/computed tomography were unremarkable. The patient had rapid improvement with a more potent topical steroid but also was given tacrolimus ointment 0.1% for residual findings. His disease remained stable without progression at 1-year follow-up.
Contact dermatitis typically manifests as an eczematous eruption that appears on an anatomic location that was exposed to or came into contact with allergens or irritants.4 Contact dermatitis was less likely in our patient due to the lack of acute or subacute spongiosis and lymphocyte exocytosis. Rosacea is a chronic inflammatory dermatosis that presents as recurrent episodes of flushing or transient erythema, persistent erythema, phyphymatous changes, papules, pustules, and telangiectasia5; however, rosacea was less likely in our patient due to the histopathologic and immunohistochemical findings that were suggestive of FM on punch biopsy. Cutaneous lupus generally is associated with photosensitivity and manifests as erythema over the malar eminences and bridge of the nose with sparing of the nasolabial folds.6 Seborrheic dermatitis manifests as erythematous macules or patches with scale and associated pruritis on the scalp, eyebrows, eyelids, and nasolabial folds.7 This condition was less likely in our patient due to the persistence and worsening of the facial erythematous dermatitis despite the use of ketoconazole cream as well as no evidence of spongiosis, shoulder parakeratosis, vascular changes, or presence of microorganisms such as Malassezia species.
Due to the relatively rare nature of this condition as well as a wide variety of other more common etiologies for an erythematous dermatitis of the cheeks, the diagnosis of FM may be delayed or missed entirely. Physicians must have a high index of suspicion to diagnose properly and biopsy if necessary. This photoquiz serves as an important reminder to physicians to keep uncommon diseases on their differential, especially when the patient’s symptoms do not respond to treatment.
THE DIAGNOSIS: Follicular Mucinosis
Histologic examination of the hematoxylin and eosin–stained sections of the biopsy revealed an overall moderately dense, perivascular, and perifollicular lymphocytic infiltrate with follicular intraepidermal mucin (Figure). Immunohistochemical staining showed that the lymphocytic infiltrate was predominantly CD4+ over CD8+, with moderate loss of CD7 and absence of CD20 expression. Positive T-cell receptor (TCR) gene rearrangements were detected for both TCRγ and TCRΒ. The clinical features along with the histopathologic findings suggested a diagnosis of follicular mucinosis (FM) with concern in the differential for folliculotropic mycosis fungoides.

Follicular mucinosis, also known as alopecia mucinosa, is an uncommon inflammatory disorder characterized by follicular degeneration due to the accumulation of mucin within the pilosebaceous unit.1 This condition manifests clinically as indurated plaques and/or follicular papules most often on the face, neck, and scalp.2 It is further categorized as primary vs secondary FM. Primary idiopathic FM, which can further be subdivided into acute or chronic, tends to follow a more benign course, whereas secondary FM usually is associated with underlying inflammatory or neoplastic conditions, most commonly mycosis fungoides, a cutaneous T-cell lymphoma.1,2 In cases of secondary FM, treatment of the underlying cause often leads to resolution of symptoms. Regular follow-up is warranted in either classification.1,3
The initial differential diagnosis for this patient included contact dermatitis associated with mask use, with possible underlying seborrheic dermatitis or rosacea; however, the rash persisted and worsened after treatment with topical triamcinolone and ketoconazole. After the diagnosis of FM was made, the patient was started on topical betamethasone and tacrolimus with good response.
A referral to hematology/oncology revealed that the patient had primary FM and possible stage 1A folliculotropic mycosis fungoides with limited skin involvement (<10% body surface area). On physical examination, no palpable cervical or axillary lymphadenopathy were noted. Flow cytometry for lymphoma was negative with no lymphoid or blast population detected. Laboratory workup and positron emission tomography/computed tomography were unremarkable. The patient had rapid improvement with a more potent topical steroid but also was given tacrolimus ointment 0.1% for residual findings. His disease remained stable without progression at 1-year follow-up.
Contact dermatitis typically manifests as an eczematous eruption that appears on an anatomic location that was exposed to or came into contact with allergens or irritants.4 Contact dermatitis was less likely in our patient due to the lack of acute or subacute spongiosis and lymphocyte exocytosis. Rosacea is a chronic inflammatory dermatosis that presents as recurrent episodes of flushing or transient erythema, persistent erythema, phyphymatous changes, papules, pustules, and telangiectasia5; however, rosacea was less likely in our patient due to the histopathologic and immunohistochemical findings that were suggestive of FM on punch biopsy. Cutaneous lupus generally is associated with photosensitivity and manifests as erythema over the malar eminences and bridge of the nose with sparing of the nasolabial folds.6 Seborrheic dermatitis manifests as erythematous macules or patches with scale and associated pruritis on the scalp, eyebrows, eyelids, and nasolabial folds.7 This condition was less likely in our patient due to the persistence and worsening of the facial erythematous dermatitis despite the use of ketoconazole cream as well as no evidence of spongiosis, shoulder parakeratosis, vascular changes, or presence of microorganisms such as Malassezia species.
Due to the relatively rare nature of this condition as well as a wide variety of other more common etiologies for an erythematous dermatitis of the cheeks, the diagnosis of FM may be delayed or missed entirely. Physicians must have a high index of suspicion to diagnose properly and biopsy if necessary. This photoquiz serves as an important reminder to physicians to keep uncommon diseases on their differential, especially when the patient’s symptoms do not respond to treatment.
- Khalil J, Kurban M, Abbas O. Follicular mucinosis: a review. Int J Dermatol. 2021;60:159-165.
- Akinsanya AO, Tschen JA. Follicular mucinosis: a case report. Cureus. 2019;11:E4746.
- Miyagaki T. Diagnosis of early mycosis fungoides. Diagnostics (Basel). 2021;1:1721.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192.
- van Zuuren EJ, Arents BWM, van der Linden MMD, et al. Rosacea: new concepts in classification and treatment. Am J Clin Dermatol. 2021;22:457-465.
- Rothfield N, Sontheimer RD, Bernstein M. Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol. 2006;24:348-362.
- Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169.
- Khalil J, Kurban M, Abbas O. Follicular mucinosis: a review. Int J Dermatol. 2021;60:159-165.
- Akinsanya AO, Tschen JA. Follicular mucinosis: a case report. Cureus. 2019;11:E4746.
- Miyagaki T. Diagnosis of early mycosis fungoides. Diagnostics (Basel). 2021;1:1721.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192.
- van Zuuren EJ, Arents BWM, van der Linden MMD, et al. Rosacea: new concepts in classification and treatment. Am J Clin Dermatol. 2021;22:457-465.
- Rothfield N, Sontheimer RD, Bernstein M. Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol. 2006;24:348-362.
- Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169.
Progressive Erythematous Facial Rash
Progressive Erythematous Facial Rash
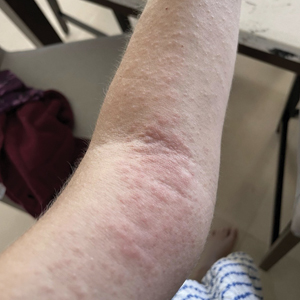
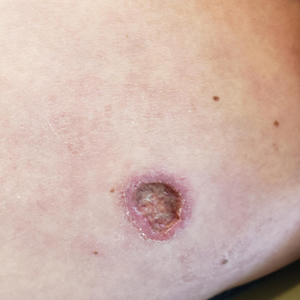
A 32-year-old man presented to the dermatology clinic for evaluation of a progressive erythematous facial rash of 4 years’ duration. The patient reported some worsening with increased face mask wear during the COVID-19 pandemic. On occasion, fluid could be expressed when the area on the right cheek was compressed. Physical examination revealed a well-demarcated erythematous plaque on the right cheek. The patient also reported intermittent mild involvement of the nose and left cheek. He initially was treated with triamcinolone and ketoconazole cream for several months, but the rash persisted. Given the chronicity and worsening of the eruption, a punch biopsy from the right cheek with immunohistochemical staining was obtained.
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
To the Editor:
A 22-year-old man presented to our clinic with a history of longstanding widespread recalcitrant atopic dermatitis (AD) since early childhood. He had been treated by an outside physician with topical steroids and nonsteroidal medications without notable improvement as well as with dupilumab, which was discontinued due to the development of severe head and neck dermatitis. Given the severity of his AD on presentation, we initiated treatment with upadacitinib 15 mg/d, which resulted in partial improvement. The dose was increased to 30 mg/d at 3 months with further clinical improvement.
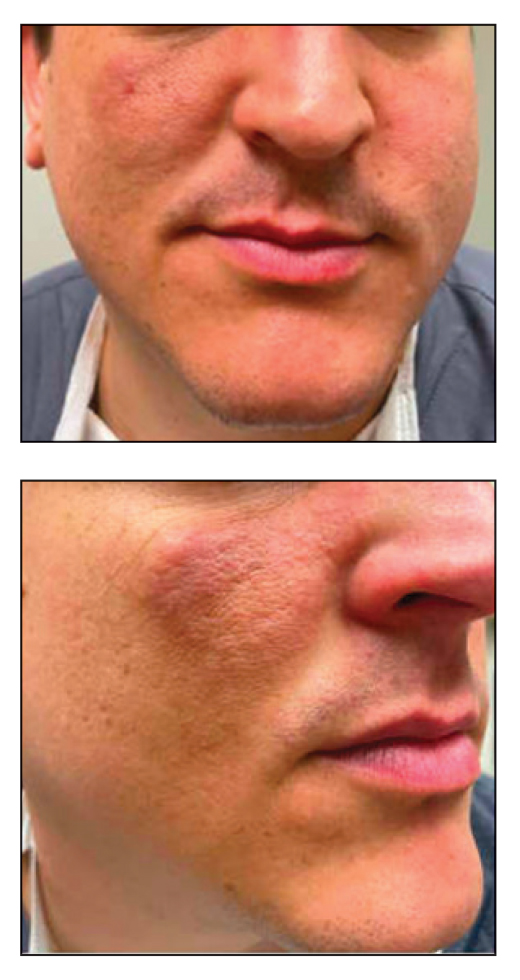
Ten months after the patient was started on upadacitinib, he presented for a follow-up evaluation and reported a new nontender nodule on the scalp. A punch biopsy revealed a dense dermal and subcutaneous lymphoid infiltrate (Figure 1) composed of many large atypical CD2+/CD5+/CD45+ T cells with partial loss of CD3 expression (Figure 2). The atypical cells demonstrated diffuse CD30+ expression (Figure 3) and a CD4:CD8 ratio of greater than 50:1 (Figures 4 and 5). He was diagnosed with anaplastic large cell lymphoma (ALCL), and the upadacitinib was discontinued. No additional therapies directed toward ALCL were initiated.
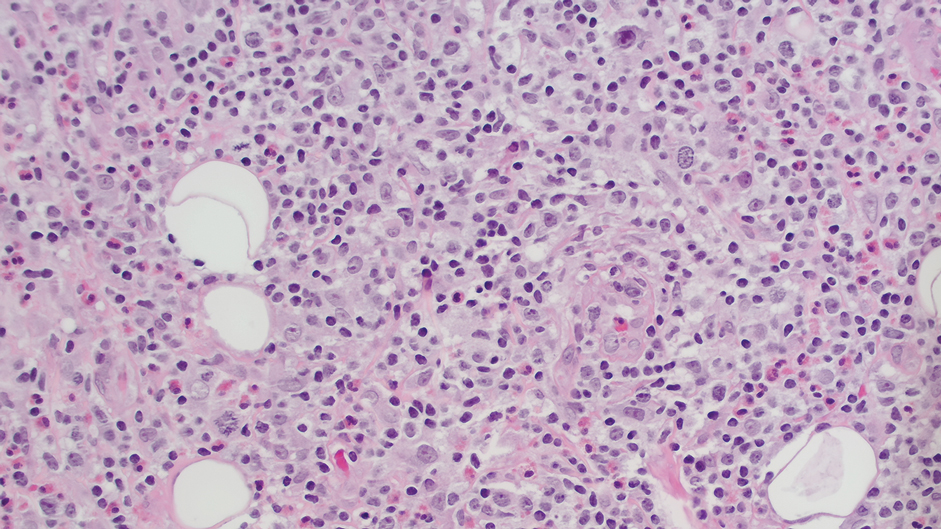
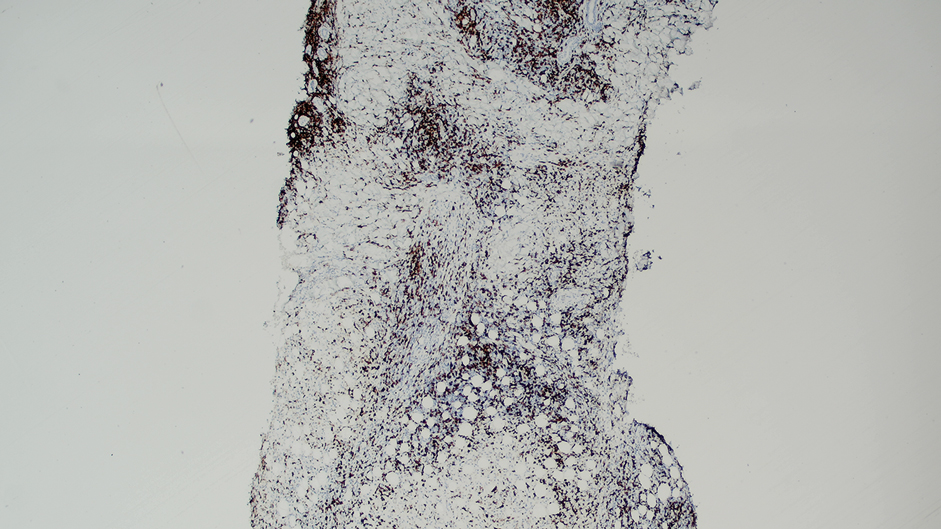
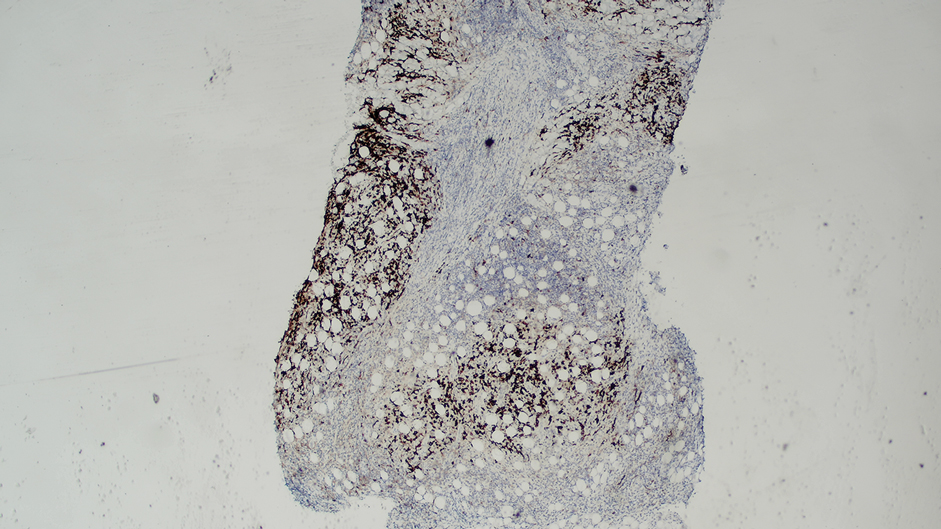
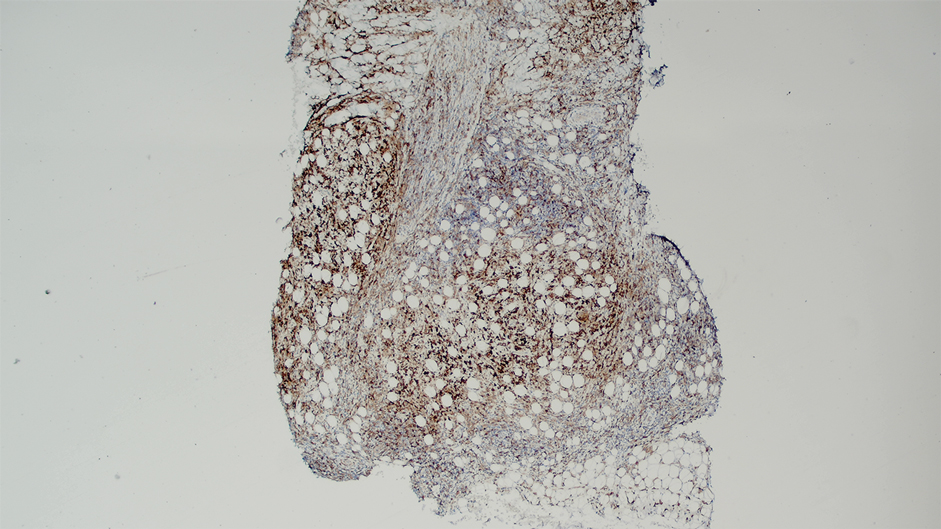
Over the next 2 weeks, the patient developed additional nodules on the postauricular skin and trunk that demonstrated similar histopathology and immunophenotype to the original scalp nodule. T-cell receptor gene rearrangement studies demonstrated shared clonal peaks in these subsequent nodules. A concurrent biopsy of an eczematous plaque on the back showed spongiotic dermatitis without evidence of cutaneous T-cell lymphoma; gene rearrangement studies from this site were negative. A positron emission tomography–computed tomography scan showed mildly hypermetabolic cervical, axillary, and inguinal lymph nodes, which were favored to be reactive. Narrow-band UVB phototherapy was initiated for management of the AD, and no additional nodules developed over the subsequent months.
Janus kinase (JAK) inhibitors are immunomodulatory small molecules that interfere with JAK–signal transducer and activator of transcription signaling involving 1 or more isoforms (eg, JAK1, JAK2, JAK3, tyrosine kinase 2) and have been used to treat various inflammatory conditions, including rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and AD.1 Upadacitinib is an oral selective JAK1 inhibitor approved by the US Food and Drug Administration for treatment of moderate to severe AD in adults and children aged 12 years and older.2 A search of PubMed using the terms upadacitinib or Rinvoq and anaplastic large cell lymphoma did not identify any cases of cutaneous ALCL arising after treatment with upadacitinib. However, a case of lymphomatoid papulosis after initiation of upadacitinib for the treatment of rheumatoid arthritis in a 74-year-old Japanese woman has been described,3 and the JAK/signal transducer and activator of transcription pathway has been implicated in the development of other CD30+ lymphoproliferative disorders.4,5
An association between JAK inhibitors and aggressive B-cell lymphomas has been described. In an observational study of 626 patients with myeloproliferative neoplasia by Porpaczy et al,6 4 of 69 (5.8%) patients treated with JAK inhibitors developed an aggressive B-cell lymphoma, whereas only 2 of 557 (0.36%) patients who did not receive JAK-inhibitor therapy developed an aggressive B-cell lymphoma. In contrast, a retrospective analysis of 2583 patients with myeloproliferative neoplasia by Pemmaraju et al7 found no significant increase in lymphoma rates in the JAK inhibitor–treated population as compared with the non-JAK inhibitor–treated group; 9 (0.56%) cases of lymphoma occurred in 1617 patients with myelofibrosis, of which 6 had exposure to JAK inhibitor therapy and 3 had no exposure to JAK inhibitor therapy (P=.082) and 5 (0.52%) cases of lymphoma occurred in 966 patients with essential thrombocythemia or polycythemia vera, none of whom had exposure to JAK inhibitor therapy.Finally, some evidence suggests the use of JAK inhibitors may be associated with an elevated risk of malignancies overall. The ORAL Surveillance study found the incidence of all cancers, excluding nonmelanoma skin cancer (NMSC), in patients treated with tofacitinib to be 4.2% (122/2911) compared with 2.9% (42/1451) in patients treated with tumor necrosis factor α inhibitors; it should be noted that the patients in this study were restricted to adults aged 50 years and older who were undergoing treatment for rheumatoid arthritis.8 In a safety profile study for upadacitinib, a higher rate of malignancies, excluding NMSC, was found in patients with AD treated with upadacitinib 30 mg/d than in patients treated with 15 mg/d; however, the overall rates of malignancies, excluding NMSC, in patients treated with upadacitinib were comparable to the standard incidence rates of malignancies in the general population derived from Surveillance, Epidemiology, and End Results data.9
In summary, we present a case of cutaneous ALCL arising after treatment with upadacitinib for AD. While some literature suggests AD may independently predispose patients to the development of CD30+ lymphoproliferative disorders, the onset of our patient’s cutaneous ALCL 10 months after initiation of upadacitinib is suggestive of an association between his lymphoproliferative disorder and JAK inhibition. Further studies are needed to better characterize the risk of lymphoproliferative disorders and other malignancies in patients treated with JAK inhibitors.
- Strangfeld A, Hierse F, Rau R, et al. Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German biologics register RABBIT. Arthritis Res Ther. 2010;12:R5. doi:10.1186/ar2904
- Rinvoq. Highlights of prescribing information. Abbvie Inc; 2024. Accessed January 31, 2026. https://www.rxabbvie.com/pdf/rinvoq_pi.pdf
- Iinuma S, Hayashi K, Noguchi A, et al. Lymphomatoid papulosis during upadacitinib treatment for rheumatoid arthritis. Eur J Dermatol. 2022;32:142-143. doi:10.1684/ejd.2022.4238
- Quesada AE, Zhang Y, Ptashkin R, et al. Next generation sequencing of breast implant-associated anaplastic large cell lymphomas reveals a novel STAT3-JAK2 fusion among other activating genetic alterations within the JAK-STAT pathway. Breast J. 2021;27:314-321. doi:10.1111/tbj.14205
- Maurus K, Appenzeller S, Roth S, et al. Recurrent oncogenic JAK and STAT alterations in cutaneous CD30-positive lymphoproliferative disorders. J Invest Dermatol. 2020;140:2023-2031.e1. doi:10.1016/j.jid.2020.02.019
- Porpaczy E, Tripolt S, Hoelbl-Kovacic A, et al. Aggressive B-cell lymphomas in patients with myelofibrosis receiving JAK1/2 inhibitor therapy. Blood. 2018;132:694-706. doi:10.1182/blood-2017-10-810739
- Pemmaraju N, Kantarjian H, Nastoupil L, et al. Characteristics of patients with myeloproliferative neoplasms with lymphoma, with or without JAK inhibitor therapy. Blood. 2019;133:2348-2351. doi:10.1182/blood-2019-01-897637
- Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386:316-326. doi:10.1056/NEJMoa2109927
- Burmester GR, Cohen SB, Winthrop KL, et al. Safety profile of upadacitinib over 15 000 patient-years across rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and atopic dermatitis. RMD Open. 2023;9:E002735. doi:10.1136/rmdopen-2022-002735
To the Editor:
A 22-year-old man presented to our clinic with a history of longstanding widespread recalcitrant atopic dermatitis (AD) since early childhood. He had been treated by an outside physician with topical steroids and nonsteroidal medications without notable improvement as well as with dupilumab, which was discontinued due to the development of severe head and neck dermatitis. Given the severity of his AD on presentation, we initiated treatment with upadacitinib 15 mg/d, which resulted in partial improvement. The dose was increased to 30 mg/d at 3 months with further clinical improvement.
Ten months after the patient was started on upadacitinib, he presented for a follow-up evaluation and reported a new nontender nodule on the scalp. A punch biopsy revealed a dense dermal and subcutaneous lymphoid infiltrate (Figure 1) composed of many large atypical CD2+/CD5+/CD45+ T cells with partial loss of CD3 expression (Figure 2). The atypical cells demonstrated diffuse CD30+ expression (Figure 3) and a CD4:CD8 ratio of greater than 50:1 (Figures 4 and 5). He was diagnosed with anaplastic large cell lymphoma (ALCL), and the upadacitinib was discontinued. No additional therapies directed toward ALCL were initiated.





Over the next 2 weeks, the patient developed additional nodules on the postauricular skin and trunk that demonstrated similar histopathology and immunophenotype to the original scalp nodule. T-cell receptor gene rearrangement studies demonstrated shared clonal peaks in these subsequent nodules. A concurrent biopsy of an eczematous plaque on the back showed spongiotic dermatitis without evidence of cutaneous T-cell lymphoma; gene rearrangement studies from this site were negative. A positron emission tomography–computed tomography scan showed mildly hypermetabolic cervical, axillary, and inguinal lymph nodes, which were favored to be reactive. Narrow-band UVB phototherapy was initiated for management of the AD, and no additional nodules developed over the subsequent months.
Janus kinase (JAK) inhibitors are immunomodulatory small molecules that interfere with JAK–signal transducer and activator of transcription signaling involving 1 or more isoforms (eg, JAK1, JAK2, JAK3, tyrosine kinase 2) and have been used to treat various inflammatory conditions, including rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and AD.1 Upadacitinib is an oral selective JAK1 inhibitor approved by the US Food and Drug Administration for treatment of moderate to severe AD in adults and children aged 12 years and older.2 A search of PubMed using the terms upadacitinib or Rinvoq and anaplastic large cell lymphoma did not identify any cases of cutaneous ALCL arising after treatment with upadacitinib. However, a case of lymphomatoid papulosis after initiation of upadacitinib for the treatment of rheumatoid arthritis in a 74-year-old Japanese woman has been described,3 and the JAK/signal transducer and activator of transcription pathway has been implicated in the development of other CD30+ lymphoproliferative disorders.4,5
An association between JAK inhibitors and aggressive B-cell lymphomas has been described. In an observational study of 626 patients with myeloproliferative neoplasia by Porpaczy et al,6 4 of 69 (5.8%) patients treated with JAK inhibitors developed an aggressive B-cell lymphoma, whereas only 2 of 557 (0.36%) patients who did not receive JAK-inhibitor therapy developed an aggressive B-cell lymphoma. In contrast, a retrospective analysis of 2583 patients with myeloproliferative neoplasia by Pemmaraju et al7 found no significant increase in lymphoma rates in the JAK inhibitor–treated population as compared with the non-JAK inhibitor–treated group; 9 (0.56%) cases of lymphoma occurred in 1617 patients with myelofibrosis, of which 6 had exposure to JAK inhibitor therapy and 3 had no exposure to JAK inhibitor therapy (P=.082) and 5 (0.52%) cases of lymphoma occurred in 966 patients with essential thrombocythemia or polycythemia vera, none of whom had exposure to JAK inhibitor therapy.Finally, some evidence suggests the use of JAK inhibitors may be associated with an elevated risk of malignancies overall. The ORAL Surveillance study found the incidence of all cancers, excluding nonmelanoma skin cancer (NMSC), in patients treated with tofacitinib to be 4.2% (122/2911) compared with 2.9% (42/1451) in patients treated with tumor necrosis factor α inhibitors; it should be noted that the patients in this study were restricted to adults aged 50 years and older who were undergoing treatment for rheumatoid arthritis.8 In a safety profile study for upadacitinib, a higher rate of malignancies, excluding NMSC, was found in patients with AD treated with upadacitinib 30 mg/d than in patients treated with 15 mg/d; however, the overall rates of malignancies, excluding NMSC, in patients treated with upadacitinib were comparable to the standard incidence rates of malignancies in the general population derived from Surveillance, Epidemiology, and End Results data.9
In summary, we present a case of cutaneous ALCL arising after treatment with upadacitinib for AD. While some literature suggests AD may independently predispose patients to the development of CD30+ lymphoproliferative disorders, the onset of our patient’s cutaneous ALCL 10 months after initiation of upadacitinib is suggestive of an association between his lymphoproliferative disorder and JAK inhibition. Further studies are needed to better characterize the risk of lymphoproliferative disorders and other malignancies in patients treated with JAK inhibitors.
To the Editor:
A 22-year-old man presented to our clinic with a history of longstanding widespread recalcitrant atopic dermatitis (AD) since early childhood. He had been treated by an outside physician with topical steroids and nonsteroidal medications without notable improvement as well as with dupilumab, which was discontinued due to the development of severe head and neck dermatitis. Given the severity of his AD on presentation, we initiated treatment with upadacitinib 15 mg/d, which resulted in partial improvement. The dose was increased to 30 mg/d at 3 months with further clinical improvement.
Ten months after the patient was started on upadacitinib, he presented for a follow-up evaluation and reported a new nontender nodule on the scalp. A punch biopsy revealed a dense dermal and subcutaneous lymphoid infiltrate (Figure 1) composed of many large atypical CD2+/CD5+/CD45+ T cells with partial loss of CD3 expression (Figure 2). The atypical cells demonstrated diffuse CD30+ expression (Figure 3) and a CD4:CD8 ratio of greater than 50:1 (Figures 4 and 5). He was diagnosed with anaplastic large cell lymphoma (ALCL), and the upadacitinib was discontinued. No additional therapies directed toward ALCL were initiated.





Over the next 2 weeks, the patient developed additional nodules on the postauricular skin and trunk that demonstrated similar histopathology and immunophenotype to the original scalp nodule. T-cell receptor gene rearrangement studies demonstrated shared clonal peaks in these subsequent nodules. A concurrent biopsy of an eczematous plaque on the back showed spongiotic dermatitis without evidence of cutaneous T-cell lymphoma; gene rearrangement studies from this site were negative. A positron emission tomography–computed tomography scan showed mildly hypermetabolic cervical, axillary, and inguinal lymph nodes, which were favored to be reactive. Narrow-band UVB phototherapy was initiated for management of the AD, and no additional nodules developed over the subsequent months.
Janus kinase (JAK) inhibitors are immunomodulatory small molecules that interfere with JAK–signal transducer and activator of transcription signaling involving 1 or more isoforms (eg, JAK1, JAK2, JAK3, tyrosine kinase 2) and have been used to treat various inflammatory conditions, including rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and AD.1 Upadacitinib is an oral selective JAK1 inhibitor approved by the US Food and Drug Administration for treatment of moderate to severe AD in adults and children aged 12 years and older.2 A search of PubMed using the terms upadacitinib or Rinvoq and anaplastic large cell lymphoma did not identify any cases of cutaneous ALCL arising after treatment with upadacitinib. However, a case of lymphomatoid papulosis after initiation of upadacitinib for the treatment of rheumatoid arthritis in a 74-year-old Japanese woman has been described,3 and the JAK/signal transducer and activator of transcription pathway has been implicated in the development of other CD30+ lymphoproliferative disorders.4,5
An association between JAK inhibitors and aggressive B-cell lymphomas has been described. In an observational study of 626 patients with myeloproliferative neoplasia by Porpaczy et al,6 4 of 69 (5.8%) patients treated with JAK inhibitors developed an aggressive B-cell lymphoma, whereas only 2 of 557 (0.36%) patients who did not receive JAK-inhibitor therapy developed an aggressive B-cell lymphoma. In contrast, a retrospective analysis of 2583 patients with myeloproliferative neoplasia by Pemmaraju et al7 found no significant increase in lymphoma rates in the JAK inhibitor–treated population as compared with the non-JAK inhibitor–treated group; 9 (0.56%) cases of lymphoma occurred in 1617 patients with myelofibrosis, of which 6 had exposure to JAK inhibitor therapy and 3 had no exposure to JAK inhibitor therapy (P=.082) and 5 (0.52%) cases of lymphoma occurred in 966 patients with essential thrombocythemia or polycythemia vera, none of whom had exposure to JAK inhibitor therapy.Finally, some evidence suggests the use of JAK inhibitors may be associated with an elevated risk of malignancies overall. The ORAL Surveillance study found the incidence of all cancers, excluding nonmelanoma skin cancer (NMSC), in patients treated with tofacitinib to be 4.2% (122/2911) compared with 2.9% (42/1451) in patients treated with tumor necrosis factor α inhibitors; it should be noted that the patients in this study were restricted to adults aged 50 years and older who were undergoing treatment for rheumatoid arthritis.8 In a safety profile study for upadacitinib, a higher rate of malignancies, excluding NMSC, was found in patients with AD treated with upadacitinib 30 mg/d than in patients treated with 15 mg/d; however, the overall rates of malignancies, excluding NMSC, in patients treated with upadacitinib were comparable to the standard incidence rates of malignancies in the general population derived from Surveillance, Epidemiology, and End Results data.9
In summary, we present a case of cutaneous ALCL arising after treatment with upadacitinib for AD. While some literature suggests AD may independently predispose patients to the development of CD30+ lymphoproliferative disorders, the onset of our patient’s cutaneous ALCL 10 months after initiation of upadacitinib is suggestive of an association between his lymphoproliferative disorder and JAK inhibition. Further studies are needed to better characterize the risk of lymphoproliferative disorders and other malignancies in patients treated with JAK inhibitors.
- Strangfeld A, Hierse F, Rau R, et al. Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German biologics register RABBIT. Arthritis Res Ther. 2010;12:R5. doi:10.1186/ar2904
- Rinvoq. Highlights of prescribing information. Abbvie Inc; 2024. Accessed January 31, 2026. https://www.rxabbvie.com/pdf/rinvoq_pi.pdf
- Iinuma S, Hayashi K, Noguchi A, et al. Lymphomatoid papulosis during upadacitinib treatment for rheumatoid arthritis. Eur J Dermatol. 2022;32:142-143. doi:10.1684/ejd.2022.4238
- Quesada AE, Zhang Y, Ptashkin R, et al. Next generation sequencing of breast implant-associated anaplastic large cell lymphomas reveals a novel STAT3-JAK2 fusion among other activating genetic alterations within the JAK-STAT pathway. Breast J. 2021;27:314-321. doi:10.1111/tbj.14205
- Maurus K, Appenzeller S, Roth S, et al. Recurrent oncogenic JAK and STAT alterations in cutaneous CD30-positive lymphoproliferative disorders. J Invest Dermatol. 2020;140:2023-2031.e1. doi:10.1016/j.jid.2020.02.019
- Porpaczy E, Tripolt S, Hoelbl-Kovacic A, et al. Aggressive B-cell lymphomas in patients with myelofibrosis receiving JAK1/2 inhibitor therapy. Blood. 2018;132:694-706. doi:10.1182/blood-2017-10-810739
- Pemmaraju N, Kantarjian H, Nastoupil L, et al. Characteristics of patients with myeloproliferative neoplasms with lymphoma, with or without JAK inhibitor therapy. Blood. 2019;133:2348-2351. doi:10.1182/blood-2019-01-897637
- Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386:316-326. doi:10.1056/NEJMoa2109927
- Burmester GR, Cohen SB, Winthrop KL, et al. Safety profile of upadacitinib over 15 000 patient-years across rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and atopic dermatitis. RMD Open. 2023;9:E002735. doi:10.1136/rmdopen-2022-002735
- Strangfeld A, Hierse F, Rau R, et al. Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German biologics register RABBIT. Arthritis Res Ther. 2010;12:R5. doi:10.1186/ar2904
- Rinvoq. Highlights of prescribing information. Abbvie Inc; 2024. Accessed January 31, 2026. https://www.rxabbvie.com/pdf/rinvoq_pi.pdf
- Iinuma S, Hayashi K, Noguchi A, et al. Lymphomatoid papulosis during upadacitinib treatment for rheumatoid arthritis. Eur J Dermatol. 2022;32:142-143. doi:10.1684/ejd.2022.4238
- Quesada AE, Zhang Y, Ptashkin R, et al. Next generation sequencing of breast implant-associated anaplastic large cell lymphomas reveals a novel STAT3-JAK2 fusion among other activating genetic alterations within the JAK-STAT pathway. Breast J. 2021;27:314-321. doi:10.1111/tbj.14205
- Maurus K, Appenzeller S, Roth S, et al. Recurrent oncogenic JAK and STAT alterations in cutaneous CD30-positive lymphoproliferative disorders. J Invest Dermatol. 2020;140:2023-2031.e1. doi:10.1016/j.jid.2020.02.019
- Porpaczy E, Tripolt S, Hoelbl-Kovacic A, et al. Aggressive B-cell lymphomas in patients with myelofibrosis receiving JAK1/2 inhibitor therapy. Blood. 2018;132:694-706. doi:10.1182/blood-2017-10-810739
- Pemmaraju N, Kantarjian H, Nastoupil L, et al. Characteristics of patients with myeloproliferative neoplasms with lymphoma, with or without JAK inhibitor therapy. Blood. 2019;133:2348-2351. doi:10.1182/blood-2019-01-897637
- Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386:316-326. doi:10.1056/NEJMoa2109927
- Burmester GR, Cohen SB, Winthrop KL, et al. Safety profile of upadacitinib over 15 000 patient-years across rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and atopic dermatitis. RMD Open. 2023;9:E002735. doi:10.1136/rmdopen-2022-002735
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
Practice Points
- Janus kinase inhibitors are immunomodulators used for the treatment of various inflammatory conditions, including atopic dermatitis.
- Treatment with Janus kinase inhibitors may be associated with the development of CD3012+ lymphoproliferative disorders such as cutaneous anaplastic large cell lymphoma.
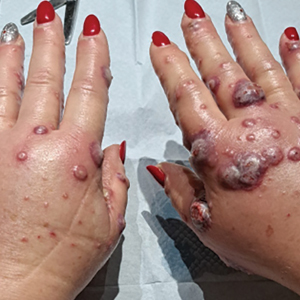
Acute Pustular Eruption on the Hands
Acute Pustular Eruption on the Hands
THE DIAGNOSIS: Neutrophilic Dermatosis of the Dorsal Hands
Histopathology showed a unilocular pustule with a dense neutrophilic infiltrate of the superficial dermis. Minimal vascular alterations also were observed. These findings were consistent with a diagnosis of neutrophilic dermatosis of the dorsal hands (NDDH). Our patient was treated successfully with systemic corticosteroids (1 mg/kg/d) with rapid improvement after 10 days of treatment.
Neutrophilic dermatosis of the dorsal hands is an evolving disease concept that was first described as pustular vasculitis by Strutton et al1 in 1995. Galaria et al2 subsequently identified NDDH as a clinical entity associating tender erythematous plaques, pustules, bullae, and/or ulcers on the dorsal hands with histologic features of Sweet syndrome (SS). After reviewing 9 cases of NDDH—all of which demonstrated clinical, laboratory, and histologic characteristics of SS—Walling et al3 concluded that NDDH was best understood as a distributional variant of SS.
Our patient presented with vascular alterations described as a reactive response to the neutrophilic infiltration. The presence of vasculitis in SS and NDDH biopsies is considered as an occasional epiphenomenon and should not rule out the diagnosis of NDDH.3 A literature review of 123 cases of NDDH revealed the presence of vasculitis in 36 (29.5%) patients.4 With regard to other clinical findings, it has been suggested that an increased white blood cell count and elevated C-reactive protein level, as was seen in our patient, may be observed in NDDH, albeit less frequently than in classical SS.4
While palmar involvement of NDDH is considered rare, the recent review of 123 cases of NDDH identified palmar lesions in 5 patients (4.1%).4 Earlier reviews had identified 12 historical cases.5 Palmar manifestations of NDDH have been shown to be associated with erythematous nonulcerated lesions (as opposed to the classical ulcerative or pustular plaques) and a lower association with hematologic malignancies.5
In our patient’s case, dyshidrosis was excluded due to the presence of painful ulcerative plaques rather than pruritic, deep-seated vesicles. Pustular psoriasis typically manifests with sterile pustules on the palms and soles; however, the rapid onset of ulcerative, necrotic plaques and substantial edema are more specific to NDDH. Poststreptococcal pustulosis generally follows a streptococcal infection and lacks the violaceous undermined borders seen in NDDH. Reactive arthritis manifests with hyperkeratotic plaques and is associated with the clinical triad of urethritis, conjunctivitis, and arthritis, which were absent in our patient.
The histologic differential diagnosis of NDDH includes infection, pyoderma gangrenosum, bowel-associated dermatosis-arthritis syndrome, rheumatoid neutrophilic dermatitis, and erythema elevatum diutinum3,4; however, these conditions typically manifest with distinct clinical features that allow for differentiation, despite histologic similarities. The wide histologic spectrum of neutrophilic dermatosis may contribute to variable clinical manifestations and an evolving disease concept, as the classification of NDDH has changed from a primary vasculitis to a variant of SS. However, this evolution does not affect the appropriate management, as they all have shown good response to corticosteroid treatment.4,6
- Strutton G, Weedon D, Robertson I. Pustular vasculitis of the hands. J Am Acad Dermatol. 1995;32(2 pt 1):192-198.
- Galaria NA, Junkins-Hopkins JM, Kligman D, et al. Neutrophilic dermatosis of the dorsal hands: pustular vasculitis revisited. J Am Acad Dermatol. 2000;43(5 pt 1):870-874.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63
- Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 cases. J Am Acad Dermatol. 2023;88:1338-1344.
- Arandes-Marcocci J, Altemir-Vidal A, Iglesias-Plaza A, et al. Neutrophilic dermatosis of the hands with palmar involvement: does it have clinical implication? Int J Dermatol. 2020;59:736-738.
- Del Pozo J, Sacristán F, Martínez W, et al. Neutrophilic dermatosis of the hands: presentation of eight cases and review of the literature. J Dermatol. 2007;34:243-247.
THE DIAGNOSIS: Neutrophilic Dermatosis of the Dorsal Hands
Histopathology showed a unilocular pustule with a dense neutrophilic infiltrate of the superficial dermis. Minimal vascular alterations also were observed. These findings were consistent with a diagnosis of neutrophilic dermatosis of the dorsal hands (NDDH). Our patient was treated successfully with systemic corticosteroids (1 mg/kg/d) with rapid improvement after 10 days of treatment.
Neutrophilic dermatosis of the dorsal hands is an evolving disease concept that was first described as pustular vasculitis by Strutton et al1 in 1995. Galaria et al2 subsequently identified NDDH as a clinical entity associating tender erythematous plaques, pustules, bullae, and/or ulcers on the dorsal hands with histologic features of Sweet syndrome (SS). After reviewing 9 cases of NDDH—all of which demonstrated clinical, laboratory, and histologic characteristics of SS—Walling et al3 concluded that NDDH was best understood as a distributional variant of SS.
Our patient presented with vascular alterations described as a reactive response to the neutrophilic infiltration. The presence of vasculitis in SS and NDDH biopsies is considered as an occasional epiphenomenon and should not rule out the diagnosis of NDDH.3 A literature review of 123 cases of NDDH revealed the presence of vasculitis in 36 (29.5%) patients.4 With regard to other clinical findings, it has been suggested that an increased white blood cell count and elevated C-reactive protein level, as was seen in our patient, may be observed in NDDH, albeit less frequently than in classical SS.4
While palmar involvement of NDDH is considered rare, the recent review of 123 cases of NDDH identified palmar lesions in 5 patients (4.1%).4 Earlier reviews had identified 12 historical cases.5 Palmar manifestations of NDDH have been shown to be associated with erythematous nonulcerated lesions (as opposed to the classical ulcerative or pustular plaques) and a lower association with hematologic malignancies.5
In our patient’s case, dyshidrosis was excluded due to the presence of painful ulcerative plaques rather than pruritic, deep-seated vesicles. Pustular psoriasis typically manifests with sterile pustules on the palms and soles; however, the rapid onset of ulcerative, necrotic plaques and substantial edema are more specific to NDDH. Poststreptococcal pustulosis generally follows a streptococcal infection and lacks the violaceous undermined borders seen in NDDH. Reactive arthritis manifests with hyperkeratotic plaques and is associated with the clinical triad of urethritis, conjunctivitis, and arthritis, which were absent in our patient.
The histologic differential diagnosis of NDDH includes infection, pyoderma gangrenosum, bowel-associated dermatosis-arthritis syndrome, rheumatoid neutrophilic dermatitis, and erythema elevatum diutinum3,4; however, these conditions typically manifest with distinct clinical features that allow for differentiation, despite histologic similarities. The wide histologic spectrum of neutrophilic dermatosis may contribute to variable clinical manifestations and an evolving disease concept, as the classification of NDDH has changed from a primary vasculitis to a variant of SS. However, this evolution does not affect the appropriate management, as they all have shown good response to corticosteroid treatment.4,6
THE DIAGNOSIS: Neutrophilic Dermatosis of the Dorsal Hands
Histopathology showed a unilocular pustule with a dense neutrophilic infiltrate of the superficial dermis. Minimal vascular alterations also were observed. These findings were consistent with a diagnosis of neutrophilic dermatosis of the dorsal hands (NDDH). Our patient was treated successfully with systemic corticosteroids (1 mg/kg/d) with rapid improvement after 10 days of treatment.
Neutrophilic dermatosis of the dorsal hands is an evolving disease concept that was first described as pustular vasculitis by Strutton et al1 in 1995. Galaria et al2 subsequently identified NDDH as a clinical entity associating tender erythematous plaques, pustules, bullae, and/or ulcers on the dorsal hands with histologic features of Sweet syndrome (SS). After reviewing 9 cases of NDDH—all of which demonstrated clinical, laboratory, and histologic characteristics of SS—Walling et al3 concluded that NDDH was best understood as a distributional variant of SS.
Our patient presented with vascular alterations described as a reactive response to the neutrophilic infiltration. The presence of vasculitis in SS and NDDH biopsies is considered as an occasional epiphenomenon and should not rule out the diagnosis of NDDH.3 A literature review of 123 cases of NDDH revealed the presence of vasculitis in 36 (29.5%) patients.4 With regard to other clinical findings, it has been suggested that an increased white blood cell count and elevated C-reactive protein level, as was seen in our patient, may be observed in NDDH, albeit less frequently than in classical SS.4
While palmar involvement of NDDH is considered rare, the recent review of 123 cases of NDDH identified palmar lesions in 5 patients (4.1%).4 Earlier reviews had identified 12 historical cases.5 Palmar manifestations of NDDH have been shown to be associated with erythematous nonulcerated lesions (as opposed to the classical ulcerative or pustular plaques) and a lower association with hematologic malignancies.5
In our patient’s case, dyshidrosis was excluded due to the presence of painful ulcerative plaques rather than pruritic, deep-seated vesicles. Pustular psoriasis typically manifests with sterile pustules on the palms and soles; however, the rapid onset of ulcerative, necrotic plaques and substantial edema are more specific to NDDH. Poststreptococcal pustulosis generally follows a streptococcal infection and lacks the violaceous undermined borders seen in NDDH. Reactive arthritis manifests with hyperkeratotic plaques and is associated with the clinical triad of urethritis, conjunctivitis, and arthritis, which were absent in our patient.
The histologic differential diagnosis of NDDH includes infection, pyoderma gangrenosum, bowel-associated dermatosis-arthritis syndrome, rheumatoid neutrophilic dermatitis, and erythema elevatum diutinum3,4; however, these conditions typically manifest with distinct clinical features that allow for differentiation, despite histologic similarities. The wide histologic spectrum of neutrophilic dermatosis may contribute to variable clinical manifestations and an evolving disease concept, as the classification of NDDH has changed from a primary vasculitis to a variant of SS. However, this evolution does not affect the appropriate management, as they all have shown good response to corticosteroid treatment.4,6
- Strutton G, Weedon D, Robertson I. Pustular vasculitis of the hands. J Am Acad Dermatol. 1995;32(2 pt 1):192-198.
- Galaria NA, Junkins-Hopkins JM, Kligman D, et al. Neutrophilic dermatosis of the dorsal hands: pustular vasculitis revisited. J Am Acad Dermatol. 2000;43(5 pt 1):870-874.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63
- Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 cases. J Am Acad Dermatol. 2023;88:1338-1344.
- Arandes-Marcocci J, Altemir-Vidal A, Iglesias-Plaza A, et al. Neutrophilic dermatosis of the hands with palmar involvement: does it have clinical implication? Int J Dermatol. 2020;59:736-738.
- Del Pozo J, Sacristán F, Martínez W, et al. Neutrophilic dermatosis of the hands: presentation of eight cases and review of the literature. J Dermatol. 2007;34:243-247.
- Strutton G, Weedon D, Robertson I. Pustular vasculitis of the hands. J Am Acad Dermatol. 1995;32(2 pt 1):192-198.
- Galaria NA, Junkins-Hopkins JM, Kligman D, et al. Neutrophilic dermatosis of the dorsal hands: pustular vasculitis revisited. J Am Acad Dermatol. 2000;43(5 pt 1):870-874.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63
- Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 cases. J Am Acad Dermatol. 2023;88:1338-1344.
- Arandes-Marcocci J, Altemir-Vidal A, Iglesias-Plaza A, et al. Neutrophilic dermatosis of the hands with palmar involvement: does it have clinical implication? Int J Dermatol. 2020;59:736-738.
- Del Pozo J, Sacristán F, Martínez W, et al. Neutrophilic dermatosis of the hands: presentation of eight cases and review of the literature. J Dermatol. 2007;34:243-247.
Acute Pustular Eruption on the Hands
Acute Pustular Eruption on the Hands
A 56-year-old woman was referred to the dermatology department for a painful acral pustular eruption of 6 days’ duration. Her medical history was otherwise unremarkable. Physical examination revealed multiple pustules on the hands with large blisters on an erythematous base and painful surface ulceration (top). Papulonodular infiltrated lesions also were observed on the dorsal aspect of the hands (bottom). There were no additional systemic symptoms. Routine laboratory tests showed hyperleukocytosis at 17.9×103/mm3 (reference range, 4-10×103/mm3) with neutrophils at 12.3×103/mm3 (1.8-7.5×103/mm3) and elevated C-reactive protein at 67 mg/L (<5 mg/L). Screening for hematologic neoplasms, solid tumors, and inflammatory bowel disease was negative. An incisional biopsy was performed on a pustule on the palm of the left hand.
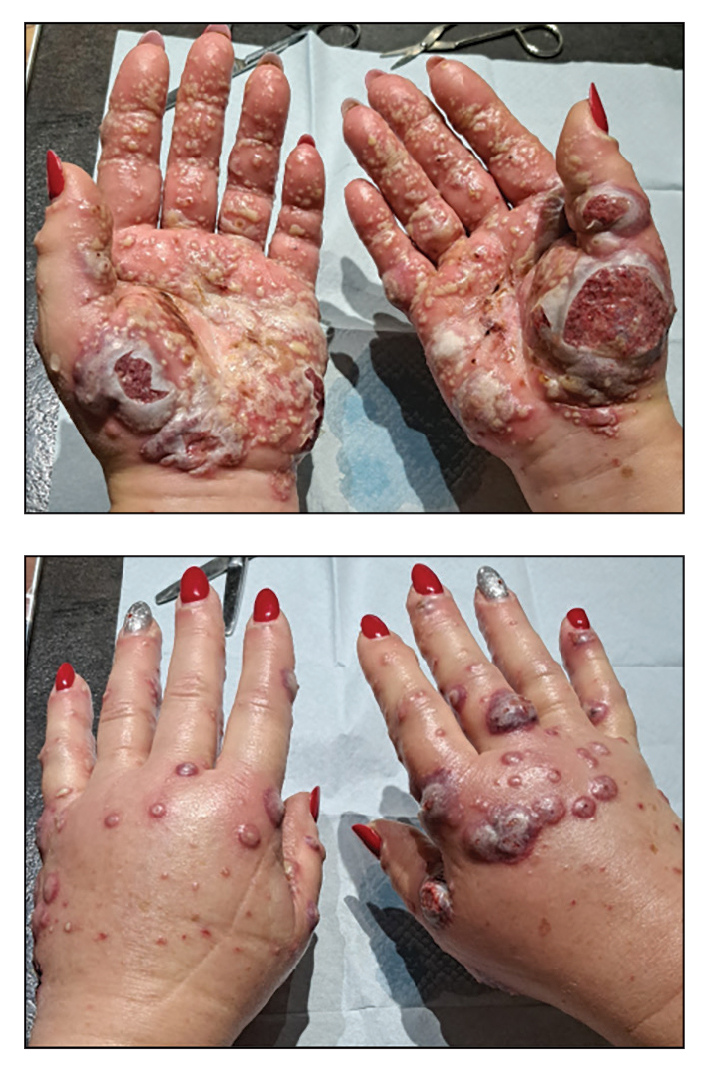
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.
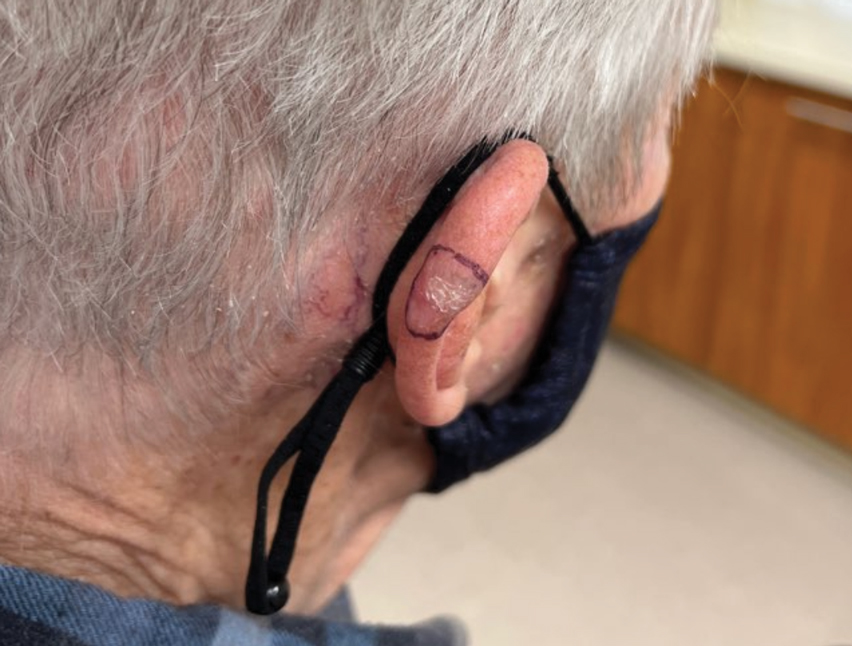
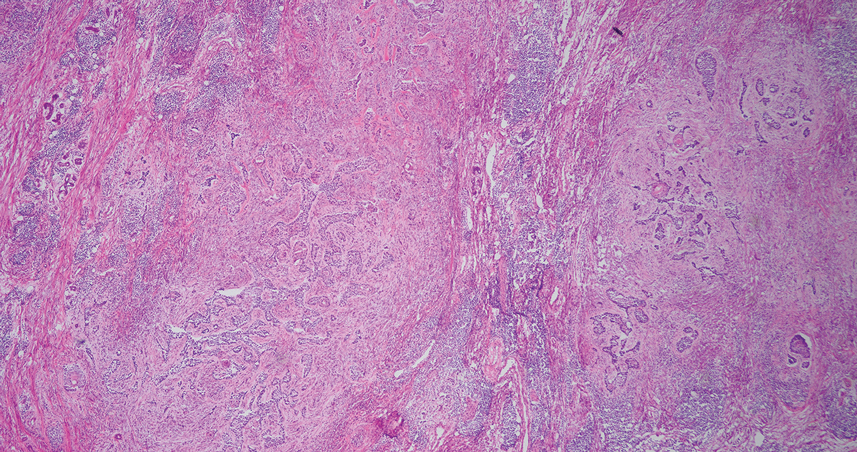
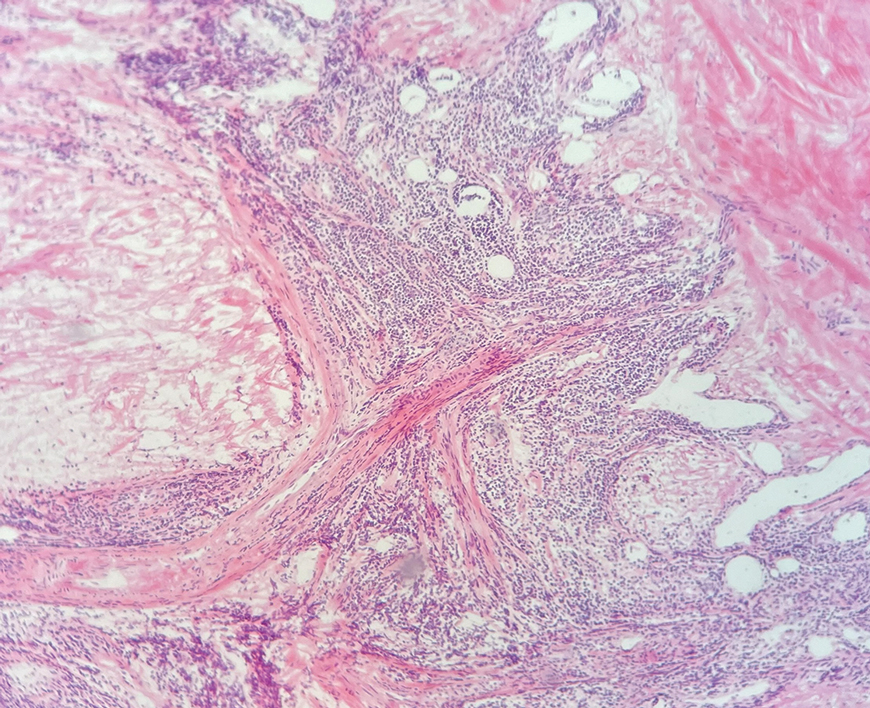
Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.



Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.



Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Practice Points
- Collision tumors of cutaneous B-cell lymphoma and basal cell carcinoma occurring within the same lesion are uncommon findings during Mohs surgery.
- Sheets of atypical monomorphic lymphocytes on deeper Mohs sections should prompt the surgeon to consider further evaluation, including sending tissue for permanent sections.
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Photosensitivity refers to clinical manifestations arising from exposure to sunlight. Photodermatoses encompass a group of skin diseases caused by varying degrees of radiation exposure, including UV radiation and visible light. Photodermatoses can be categorized into 5 main types: primary, exogenous, photoexacerbated, metabolic, and genetic.1 The clinical features of photodermatoses vary depending on the underlying cause but often include pruritic flares, wheals, or dermatitis on sun-exposed areas of the skin.2 While photodermatoses typically are not life threatening, they can greatly impact patients’ quality of life. It is crucial to emphasize the importance of photoprotection and sunlight avoidance to patients as preventive measures against the manifestations of these skin diseases. Furthermore, we present a case of photocontact dermatitis (PCD) and discuss common causative agents, diagnostic mimickers, and treatment options.
Case Report
A 51-year-old woman with no relevant medical history presented to the dermatology clinic with a rash on the neck and under the eyes of 6 days’ duration. The rash was intermittently pruritic but otherwise asymptomatic. The patient reported that she had spent extensive time on the golf course the day of the rash onset and noted that a similar rash had occurred one other time 2 to 3 months prior, also following a prolonged period on the golf course. She had been using over-the-counter fexofenadine 180 mg and over-the-counter lidocaine spray for symptom relief.
Upon physical examination, erythematous patches were appreciated in a photodistributed pattern on the arms, legs, neck, face, and chest—areas that were not covered by clothing (Figures 1-3). Due to the distribution and morphology of the erythematous patches along with clinical course of onset following exposure to various environmental agents including pesticides, herbicides, oak, and pollen, a diagnosis of PCD was made. The patient was prescribed hydrocortisone cream 2.5%, fluticasone propionate cream 0.05%, and methylprednisolone in addition to the antihistamine. Improvement was noted after 3 days with complete resolution of the skin manifestations. She was counseled on wearing clothing with a universal protection factor rating of 50+ when on the golf course and when sun exposure is expected for an extended period of time.
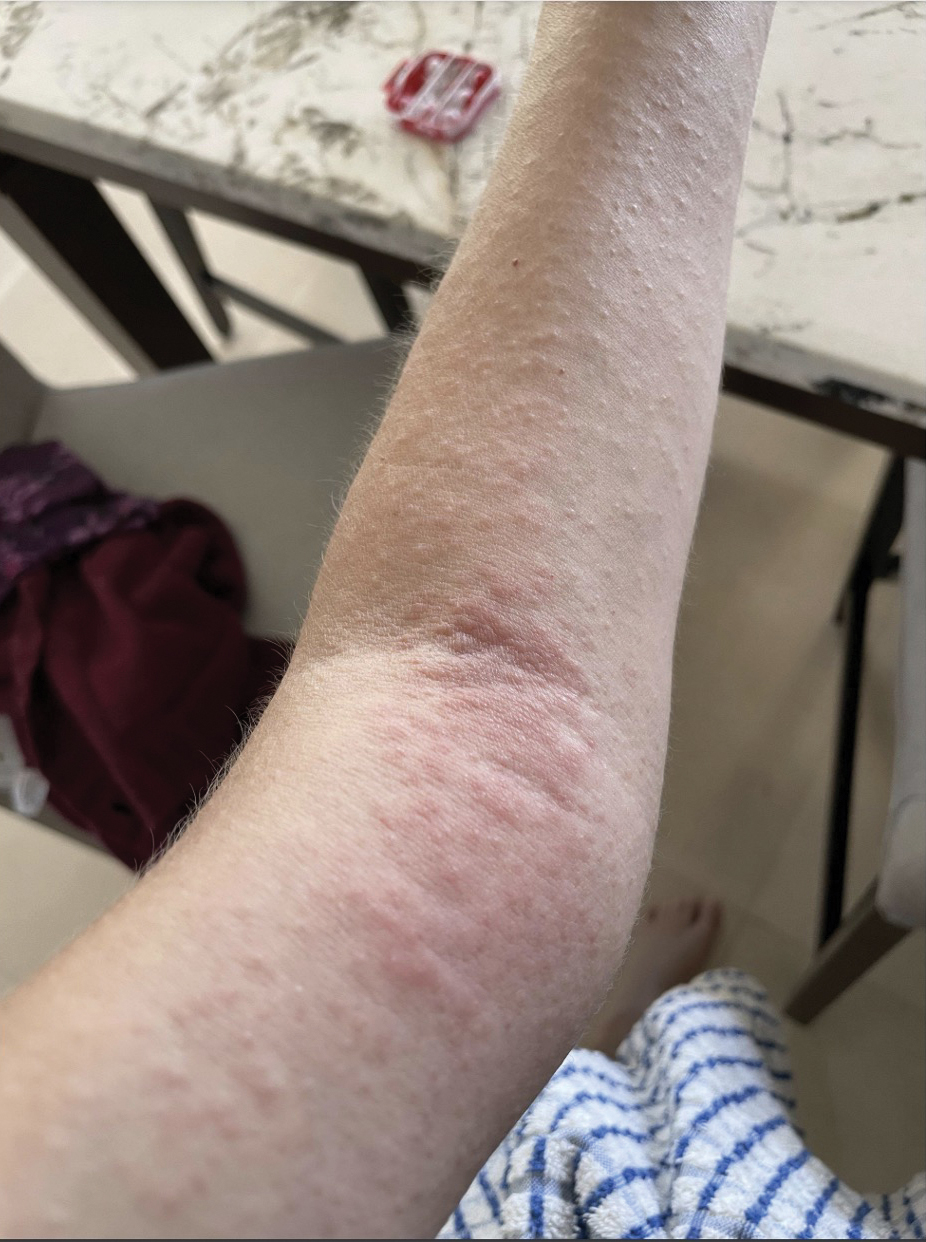
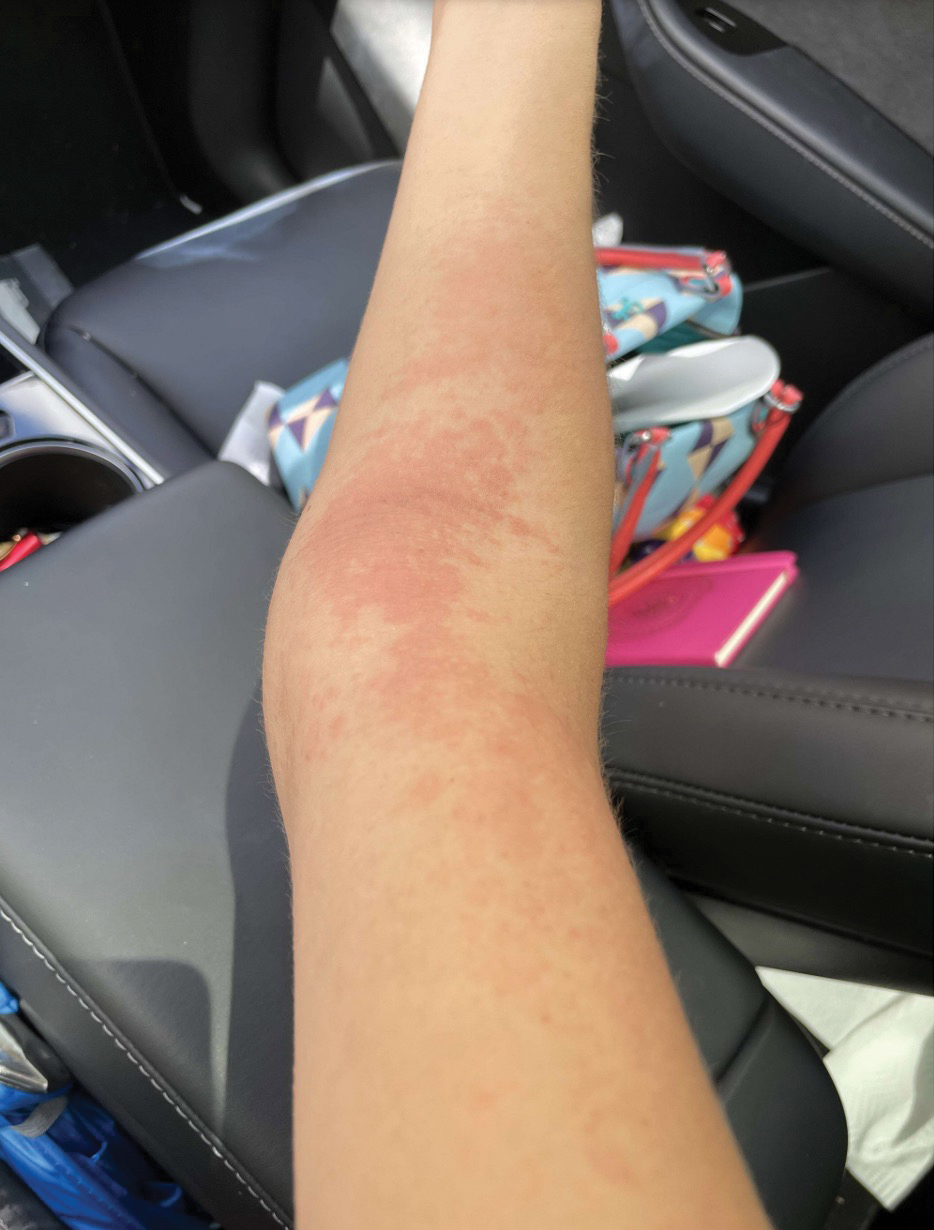
Causative Agents
Photodermatoses are caused by antigenic substances that lead to photosensitization acquired by either contact or oral ingestion with subsequent sensitization to UV radiation. Halogenated salicylanilide, fenticlor, hexachlorophene, bithionol and, in rare cases, sunscreens, have been reported as triggers.3 In a study performed in 2010, sunscreens, antimicrobial agents, medications, fragrances, plants/plant derivatives, and pesticides were the most commonly reported offending agents listed from highest to lowest frequency. Of the antimicrobial agents, fenticlor, a topical antimicrobial and antifungal that is now mostly used in veterinary medicine, was the most common culprit, causing 60% of cases.4,5
Clinical Manifestations
Clinical manifestations of photodermatoses vary depending upon the specific type of reaction. Examples of primary photodermatoses include polymorphous light eruption (PMLE) and solar urticaria. The cardinal symptoms of PMLE consist of severely pruritic skin lesions that can have macular, papular, papulovesicular, urticarial, multiformelike, and plaquelike variants that develop hours to days after sun exposure.3 Conversely, solar urticaria commonly develops more abruptly, with indurated plaques and wheals appearing on the arms and neck within 30 minutes of sun exposure. The lesions typically resolve within 24 hours.1
Examples of the exogenous subtype include drug-induced photosensitivity, PCD, and pseudoporphyria, with the common clinical presentation of eruption following contact with the causative agent. Drug-induced photosensitivity primarily manifests as a severe sunburnlike rash commonly caused by systemic drugs such as tetracyclines. Photocontact dermatitis is limited to sun-exposed areas of the skin and is caused by a reactive irritant such as chemicals or topical creams. Pseudoporphyria, usually caused by nonsteroidal anti-inflammatory drugs, can manifest with skin fragility and subepidermal blisters.6
Photoexacerbated photodermatoses encompass a variety of conditions ranging from hyperpigmentation disorders such as melasma to autoimmune conditions such as systemic lupus erythematosus (SLE) and dermatomyositis (DM). Common clinical features of these diseases include photodistributed erythema, often involving the cheeks, upper back, and anterior neck. Photo-exposed areas of the dorsal hands also are commonplace for both SLE and DM. Clinical manifestations of PCD are limited to sun-exposed areas of the body, specifically those that come into contact with photoallergic triggers.3 Manifestations of PCD can include pruritic eczematous eruptions resembling those of contact dermatitis 1 to 2 days after sun exposure.1
Photocontact dermatitis represents a specific sensitization via contact or oral ingestion acquired prior to sunlight exposure. It can be broken down into 2 distinct subtypes: photoallergic and photoirritant dermatitis, dependent on whether an allergic or irritant reaction is invoked.2 Plants are known to be a common trigger of photoirritant reactions, while extrinsic triggers include psoralens and medications such as tetracycline antibiotics or sulfonamides. Photoallergic reactions commonly can be caused by topical application of sunscreen or medications, namely nonsteroidal anti-inflammatory drugs.2 Clinical manifestations that may point to photoirritant dermatitis include a photodistributed eruption and classic morphology showing erythema and edema with bullae present in severe cases. These can be contrasted with the clinical manifestations of photoallergic reactions, which usually do not correlate to sun-exposed areas and consist of a monomorphous distribution pattern similar to that of eczema. Although there are distinguishing features of both subtypes of PCD, the overlapping clinical features can mimic those of solar urticaria, PMLE, cutaneous lupus erythematosus, and more systemic conditions such as SLE and DM.7
Systemic lupus erythematosus is associated with a broad range of cutaneous manifestations.8 Exposure to UV radiation is a common trigger for lupus and has the propensity to cause a malar (butterfly) rash that covers the cheeks and nasal bridge but classically spares the nasolabial folds. The rash may display confluent reddish-purple discoloration with papules and/or edema and typically is present at diagnosis in 40% to 52% of patients with SLE.8 Discoid lupus erythematosus, one of the most common cutaneous forms of lupus, manifests with various-sized coin-shaped plaques with adherent follicular hyperkeratosis and plugging. These lesions usually develop on the face, scalp, and ears but also may appear in non–sun-exposed areas.8 Dermatomyositis can manifest with photodistributed erythema affecting classic areas such as the upper back (shawl sign), anterior neck and upper chest (V-sign), and a malar rash similar to that seen in lupus, though DM classically does not spare the nasolabial folds.8,9
Because SLE and DM manifest with photodistributed rashes, it can be difficult to distinguish them from the classic symptoms of photoirritant dermatitis.9 Thus, it is imperative that providers have a high clinical index of suspicion when dealing with patients of similar presentations, as the treatment regimens vastly differ. Approaching the patient with a thorough medical history review, review of systems, biopsy (including immunofluorescence), and appropriate laboratory workup may aid in excluding more complex differential diagnoses such as SLE and DM.
Metabolic and genetic photodermatoses are more rare but can include conditions such as porphyria cutanea tarda and xeroderma pigmentosum, both of which demonstrate fragile skin, slow wound healing, and bullae on photo-exposed skin.1 Although the manifestations can be similar in these systemic conditions, they are caused by very different mechanisms. Porphyria cutanea tarda is caused by deficiencies in enzymes involved in the heme synthesis pathway, whereas xeroderma pigmentosum is caused by an alteration in DNA repair mechanisms.7
Prevalence and the Need for Standardized Testing
Most practicing dermatologists see cases of PCD due to its multiple causative agents; however, little is known about its overall prevalence. The incidence of PCD is fairly low in the general population, but this may be due to its clinical diagnosis, which excludes diagnostic testing such as phototesting and photopatch testing.10 While the incidence of photoallergic contact dermatitis also is fairly unknown, the inception of testing modalities has allowed statistics to be drawn. Research conducted in the United States has disclosed that the incidence of photoallergic contact dermatitis in individuals with a history of a prior photosensitivity eruption is approximately 10% to 20%.10 The development of guidelines and a registry for photopatch testing would aid in a greater understanding of the incidence of PCD and overall consistency of diagnosis.7 Regardless of this lack of consensus, these conditions can be properly managed and prevented if recognized clinically, while newer testing modalities would allow for confirmation of the diagnosis. It is important that any patient presenting with a history of photosensitivity be seen as a candidate for photopatch testing, especially today, as the general population is increasingly exposed to new chemicals entering the market and new social trends.7,10
Diagnosis and Treatment
It is important to consider a detailed history, including the timing, location, duration, family history, and seasonal variation of suspected photodermatoses. A thorough skin examination that takes note of the specific areas affected, morphology, and involvement of the rash or lesions can be helpful.1 Further diagnostic testing such as phototesting and photopatch testing can be employed and is especially important when distinguishing photoallergy from phototoxicity.11 Phototesting involves exposing the patient’s skin to different doses of UVA, UVB, and visible light, followed by an immediate clinical reading of the results and then a delayed reading conducted after 24 hours.1 Photopatch testing involves the application of 2 sets of identical photoallergens to prepped skin (typically cleansed with isopropyl alcohol), with one being irradiated with UVA after 24 hours and one serving as the control. A clinical assessment is conducted at 24 hours and repeated 7 days later.1 In photodermatoses, a visible reaction can be appreciated on the treatment arm while the control arm remains clear. When both sides reveal a visible reaction, this is more indicative of a light-independent allergic contact dermatitis.1
Photodermatoses occur only if there has been a specific sensitization, and therefore it is important to work with the patient to discover any new products that have been introduced into their regimen. Though many photosensitizers in personal care products (eg, antiseptics in soap and topical creams) have been discontinued, certain allergenic ingredients may remain.12 It also is important to note that sensitization to a substance that previously was not a known allergen for a particular patient can occur later in life. Avoiding further sun exposure can rapidly improve the dermatitis, and it is possible for spontaneous remission without further intervention; however, as photoallergic reactions can cause severely pruritic skin lesions, the mainstay of symptomatic treatment consists of topical corticosteroids. Oral and topical antihistamines may help alleviate the pruritus but should not be heavily relied on as this can lead to medication resistance and diminishing efficacy.3 Use of short-term oral steroids also may be considered for rapid improvement of symptoms when the patient is in moderate distress and there are no contraindications. By identifying a temporal association between the introduction of new products and the emergence of dermatitis, it may be possible to identify the causative agent. The patient should promptly discontinue the suspected agent and remain under close observation by the clinician for any further eruptions, especially following additional sun exposure.
Prevention Strategies
In the case of PCD, prevention is key. As PCD indicates a photoallergy, it is important to inform patients that the allergy will persist for a lifetime, much like in contact dermatitis; therefore, the causative agent should be avoided indefinitely.3 Patients with PCD should make intentional efforts to read ingredient lists when purchasing new personal care products to ensure they do not contain the specific causative allergen if one has been identified. Further steps should be taken to ensure proper photoprotection, including use of dense clothing and sunscreen with UVA and UVB filters (broad spectrum).3 It has also been suggested that utilizing sunscreen with ectoin, an amino acid–derived molecule, may result in increased protection against UVA-induced photodermatoses.13
Final Thoughts
Photodermatoses are a group of skin diseases caused by exposure to UV radiation. Photocontact dermatitis/photoallergy is a form of allergic contact dermatitis that results from exposure to an allergen, whether topical, oral, or environmental. The allergen is activated by exposure to UV radiation to sensitize the allergic response, resulting in a rash characterized by confluent erythematous patches or plaques, papular vesicles, and rarely blisters.3 Photocontact dermatitis, although rare, is an important differential diagnosis to consider when the presenting rash is restricted to sun-exposed areas of the skin such as the arms, legs, neck, and face. Diagnosis remains a challenge; however, new testing modalities such as photopatch testing may open the door for further confirmation and aid in proper diagnosis leading to earlier treatment times for patients. It is recommended that the clinician and patient work together to identify the possible causative agent to prevent further eruptions.
- Santoro FA, Lim HW. Update on photodermatoses. Semin Cutan Med Surg. 2011;30:229-238.
- Gimenez-Arnau A, Maurer M, De La Cuadra J, et al. Immediate contact skin reactions, an update of contact urticaria, contact urticaria syndrome and protein contact dermatitis—“a never ending story.” Eur J Dermatol. 2010;20:555-562.
- Lehmann P, Schwarz T. Photodermatoses: diagnosis and treatment. Dtsch Arztebl Int. 2011;108:135-141.
- Victor FC, Cohen DE, Soter NA. A 20-year analysis of previous and emerging allergens that elicit photoallergic contact dermatitis. J Am Acad Dermatol. 2010;62:605-610.
- Fenticlor (Code 65671). National Cancer Institute EVS Explore. Accessed October 28, 2025. https://ncithesaurus.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCIThesaurus&ns=ncit&code=C65671
- Elmets CA. Photosensitivity disorders (photodermatoses): clinical manifestations, diagnosis, and treatment. UptoDate. Updated February 23, 2023. Accessed October 28, 2025. https://www.uptodate.com/contents/photosensitivity-disorders-photodermatoses-clinical-manifestations-diagnosis-and-treatment
- Snyder M, Turrentine JE, Cruz PD Jr. Photocontact dermatitis and its clinical mimics: an overview for the allergist. Clin Rev Allergy Immunol. 2019;56:32-40.
- Cooper EE, Pisano CE, Shapiro SC. Cutaneous manifestations of “lupus”: systemic lupus erythematosus and beyond. Int J Rheumatol. 2021;2021:6610509.
- Christopher-Stine L, Amato AA, Vleugels RA. Diagnosis and differential diagnosis of dermatomyositis and polymyositis in adults. UptoDate. Updated March 3, 2025. Accessed October 28, 2025. https://www.uptodate.com/contents/diagnosis-and-differential-diagnosis-of-dermatomyositis-and-polymyositis-in-adults?search=Diagnosis%20and%20differential%20diagnosis%20of%20dermatomyositis%20and%20polymyositis%20in%20adults&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Deleo VA. Photocontact dermatitis. Dermatol Ther. 2004;17:279-288.
- Gonçalo M. Photopatch testing. In: Johansen J, Frosch P, Lepoittevin JP, eds. Contact Dermatitis. Springer; 2011:519-531.
- Enta T. Dermacase. Contact photodermatitis. Can Fam Physician. 1995;41:577,586-587.
- Duteil L, Queille-Roussel C, Aladren S, et al. Prevention of polymophic light eruption afforded by a very high broad-spectrum protection sunscreen containing ectoin. Dermatol Ther (Heidelb). 2022;12:1603-1613.
Photosensitivity refers to clinical manifestations arising from exposure to sunlight. Photodermatoses encompass a group of skin diseases caused by varying degrees of radiation exposure, including UV radiation and visible light. Photodermatoses can be categorized into 5 main types: primary, exogenous, photoexacerbated, metabolic, and genetic.1 The clinical features of photodermatoses vary depending on the underlying cause but often include pruritic flares, wheals, or dermatitis on sun-exposed areas of the skin.2 While photodermatoses typically are not life threatening, they can greatly impact patients’ quality of life. It is crucial to emphasize the importance of photoprotection and sunlight avoidance to patients as preventive measures against the manifestations of these skin diseases. Furthermore, we present a case of photocontact dermatitis (PCD) and discuss common causative agents, diagnostic mimickers, and treatment options.
Case Report
A 51-year-old woman with no relevant medical history presented to the dermatology clinic with a rash on the neck and under the eyes of 6 days’ duration. The rash was intermittently pruritic but otherwise asymptomatic. The patient reported that she had spent extensive time on the golf course the day of the rash onset and noted that a similar rash had occurred one other time 2 to 3 months prior, also following a prolonged period on the golf course. She had been using over-the-counter fexofenadine 180 mg and over-the-counter lidocaine spray for symptom relief.
Upon physical examination, erythematous patches were appreciated in a photodistributed pattern on the arms, legs, neck, face, and chest—areas that were not covered by clothing (Figures 1-3). Due to the distribution and morphology of the erythematous patches along with clinical course of onset following exposure to various environmental agents including pesticides, herbicides, oak, and pollen, a diagnosis of PCD was made. The patient was prescribed hydrocortisone cream 2.5%, fluticasone propionate cream 0.05%, and methylprednisolone in addition to the antihistamine. Improvement was noted after 3 days with complete resolution of the skin manifestations. She was counseled on wearing clothing with a universal protection factor rating of 50+ when on the golf course and when sun exposure is expected for an extended period of time.



Causative Agents
Photodermatoses are caused by antigenic substances that lead to photosensitization acquired by either contact or oral ingestion with subsequent sensitization to UV radiation. Halogenated salicylanilide, fenticlor, hexachlorophene, bithionol and, in rare cases, sunscreens, have been reported as triggers.3 In a study performed in 2010, sunscreens, antimicrobial agents, medications, fragrances, plants/plant derivatives, and pesticides were the most commonly reported offending agents listed from highest to lowest frequency. Of the antimicrobial agents, fenticlor, a topical antimicrobial and antifungal that is now mostly used in veterinary medicine, was the most common culprit, causing 60% of cases.4,5
Clinical Manifestations
Clinical manifestations of photodermatoses vary depending upon the specific type of reaction. Examples of primary photodermatoses include polymorphous light eruption (PMLE) and solar urticaria. The cardinal symptoms of PMLE consist of severely pruritic skin lesions that can have macular, papular, papulovesicular, urticarial, multiformelike, and plaquelike variants that develop hours to days after sun exposure.3 Conversely, solar urticaria commonly develops more abruptly, with indurated plaques and wheals appearing on the arms and neck within 30 minutes of sun exposure. The lesions typically resolve within 24 hours.1
Examples of the exogenous subtype include drug-induced photosensitivity, PCD, and pseudoporphyria, with the common clinical presentation of eruption following contact with the causative agent. Drug-induced photosensitivity primarily manifests as a severe sunburnlike rash commonly caused by systemic drugs such as tetracyclines. Photocontact dermatitis is limited to sun-exposed areas of the skin and is caused by a reactive irritant such as chemicals or topical creams. Pseudoporphyria, usually caused by nonsteroidal anti-inflammatory drugs, can manifest with skin fragility and subepidermal blisters.6
Photoexacerbated photodermatoses encompass a variety of conditions ranging from hyperpigmentation disorders such as melasma to autoimmune conditions such as systemic lupus erythematosus (SLE) and dermatomyositis (DM). Common clinical features of these diseases include photodistributed erythema, often involving the cheeks, upper back, and anterior neck. Photo-exposed areas of the dorsal hands also are commonplace for both SLE and DM. Clinical manifestations of PCD are limited to sun-exposed areas of the body, specifically those that come into contact with photoallergic triggers.3 Manifestations of PCD can include pruritic eczematous eruptions resembling those of contact dermatitis 1 to 2 days after sun exposure.1
Photocontact dermatitis represents a specific sensitization via contact or oral ingestion acquired prior to sunlight exposure. It can be broken down into 2 distinct subtypes: photoallergic and photoirritant dermatitis, dependent on whether an allergic or irritant reaction is invoked.2 Plants are known to be a common trigger of photoirritant reactions, while extrinsic triggers include psoralens and medications such as tetracycline antibiotics or sulfonamides. Photoallergic reactions commonly can be caused by topical application of sunscreen or medications, namely nonsteroidal anti-inflammatory drugs.2 Clinical manifestations that may point to photoirritant dermatitis include a photodistributed eruption and classic morphology showing erythema and edema with bullae present in severe cases. These can be contrasted with the clinical manifestations of photoallergic reactions, which usually do not correlate to sun-exposed areas and consist of a monomorphous distribution pattern similar to that of eczema. Although there are distinguishing features of both subtypes of PCD, the overlapping clinical features can mimic those of solar urticaria, PMLE, cutaneous lupus erythematosus, and more systemic conditions such as SLE and DM.7
Systemic lupus erythematosus is associated with a broad range of cutaneous manifestations.8 Exposure to UV radiation is a common trigger for lupus and has the propensity to cause a malar (butterfly) rash that covers the cheeks and nasal bridge but classically spares the nasolabial folds. The rash may display confluent reddish-purple discoloration with papules and/or edema and typically is present at diagnosis in 40% to 52% of patients with SLE.8 Discoid lupus erythematosus, one of the most common cutaneous forms of lupus, manifests with various-sized coin-shaped plaques with adherent follicular hyperkeratosis and plugging. These lesions usually develop on the face, scalp, and ears but also may appear in non–sun-exposed areas.8 Dermatomyositis can manifest with photodistributed erythema affecting classic areas such as the upper back (shawl sign), anterior neck and upper chest (V-sign), and a malar rash similar to that seen in lupus, though DM classically does not spare the nasolabial folds.8,9
Because SLE and DM manifest with photodistributed rashes, it can be difficult to distinguish them from the classic symptoms of photoirritant dermatitis.9 Thus, it is imperative that providers have a high clinical index of suspicion when dealing with patients of similar presentations, as the treatment regimens vastly differ. Approaching the patient with a thorough medical history review, review of systems, biopsy (including immunofluorescence), and appropriate laboratory workup may aid in excluding more complex differential diagnoses such as SLE and DM.
Metabolic and genetic photodermatoses are more rare but can include conditions such as porphyria cutanea tarda and xeroderma pigmentosum, both of which demonstrate fragile skin, slow wound healing, and bullae on photo-exposed skin.1 Although the manifestations can be similar in these systemic conditions, they are caused by very different mechanisms. Porphyria cutanea tarda is caused by deficiencies in enzymes involved in the heme synthesis pathway, whereas xeroderma pigmentosum is caused by an alteration in DNA repair mechanisms.7
Prevalence and the Need for Standardized Testing
Most practicing dermatologists see cases of PCD due to its multiple causative agents; however, little is known about its overall prevalence. The incidence of PCD is fairly low in the general population, but this may be due to its clinical diagnosis, which excludes diagnostic testing such as phototesting and photopatch testing.10 While the incidence of photoallergic contact dermatitis also is fairly unknown, the inception of testing modalities has allowed statistics to be drawn. Research conducted in the United States has disclosed that the incidence of photoallergic contact dermatitis in individuals with a history of a prior photosensitivity eruption is approximately 10% to 20%.10 The development of guidelines and a registry for photopatch testing would aid in a greater understanding of the incidence of PCD and overall consistency of diagnosis.7 Regardless of this lack of consensus, these conditions can be properly managed and prevented if recognized clinically, while newer testing modalities would allow for confirmation of the diagnosis. It is important that any patient presenting with a history of photosensitivity be seen as a candidate for photopatch testing, especially today, as the general population is increasingly exposed to new chemicals entering the market and new social trends.7,10
Diagnosis and Treatment
It is important to consider a detailed history, including the timing, location, duration, family history, and seasonal variation of suspected photodermatoses. A thorough skin examination that takes note of the specific areas affected, morphology, and involvement of the rash or lesions can be helpful.1 Further diagnostic testing such as phototesting and photopatch testing can be employed and is especially important when distinguishing photoallergy from phototoxicity.11 Phototesting involves exposing the patient’s skin to different doses of UVA, UVB, and visible light, followed by an immediate clinical reading of the results and then a delayed reading conducted after 24 hours.1 Photopatch testing involves the application of 2 sets of identical photoallergens to prepped skin (typically cleansed with isopropyl alcohol), with one being irradiated with UVA after 24 hours and one serving as the control. A clinical assessment is conducted at 24 hours and repeated 7 days later.1 In photodermatoses, a visible reaction can be appreciated on the treatment arm while the control arm remains clear. When both sides reveal a visible reaction, this is more indicative of a light-independent allergic contact dermatitis.1
Photodermatoses occur only if there has been a specific sensitization, and therefore it is important to work with the patient to discover any new products that have been introduced into their regimen. Though many photosensitizers in personal care products (eg, antiseptics in soap and topical creams) have been discontinued, certain allergenic ingredients may remain.12 It also is important to note that sensitization to a substance that previously was not a known allergen for a particular patient can occur later in life. Avoiding further sun exposure can rapidly improve the dermatitis, and it is possible for spontaneous remission without further intervention; however, as photoallergic reactions can cause severely pruritic skin lesions, the mainstay of symptomatic treatment consists of topical corticosteroids. Oral and topical antihistamines may help alleviate the pruritus but should not be heavily relied on as this can lead to medication resistance and diminishing efficacy.3 Use of short-term oral steroids also may be considered for rapid improvement of symptoms when the patient is in moderate distress and there are no contraindications. By identifying a temporal association between the introduction of new products and the emergence of dermatitis, it may be possible to identify the causative agent. The patient should promptly discontinue the suspected agent and remain under close observation by the clinician for any further eruptions, especially following additional sun exposure.
Prevention Strategies
In the case of PCD, prevention is key. As PCD indicates a photoallergy, it is important to inform patients that the allergy will persist for a lifetime, much like in contact dermatitis; therefore, the causative agent should be avoided indefinitely.3 Patients with PCD should make intentional efforts to read ingredient lists when purchasing new personal care products to ensure they do not contain the specific causative allergen if one has been identified. Further steps should be taken to ensure proper photoprotection, including use of dense clothing and sunscreen with UVA and UVB filters (broad spectrum).3 It has also been suggested that utilizing sunscreen with ectoin, an amino acid–derived molecule, may result in increased protection against UVA-induced photodermatoses.13
Final Thoughts
Photodermatoses are a group of skin diseases caused by exposure to UV radiation. Photocontact dermatitis/photoallergy is a form of allergic contact dermatitis that results from exposure to an allergen, whether topical, oral, or environmental. The allergen is activated by exposure to UV radiation to sensitize the allergic response, resulting in a rash characterized by confluent erythematous patches or plaques, papular vesicles, and rarely blisters.3 Photocontact dermatitis, although rare, is an important differential diagnosis to consider when the presenting rash is restricted to sun-exposed areas of the skin such as the arms, legs, neck, and face. Diagnosis remains a challenge; however, new testing modalities such as photopatch testing may open the door for further confirmation and aid in proper diagnosis leading to earlier treatment times for patients. It is recommended that the clinician and patient work together to identify the possible causative agent to prevent further eruptions.
Photosensitivity refers to clinical manifestations arising from exposure to sunlight. Photodermatoses encompass a group of skin diseases caused by varying degrees of radiation exposure, including UV radiation and visible light. Photodermatoses can be categorized into 5 main types: primary, exogenous, photoexacerbated, metabolic, and genetic.1 The clinical features of photodermatoses vary depending on the underlying cause but often include pruritic flares, wheals, or dermatitis on sun-exposed areas of the skin.2 While photodermatoses typically are not life threatening, they can greatly impact patients’ quality of life. It is crucial to emphasize the importance of photoprotection and sunlight avoidance to patients as preventive measures against the manifestations of these skin diseases. Furthermore, we present a case of photocontact dermatitis (PCD) and discuss common causative agents, diagnostic mimickers, and treatment options.
Case Report
A 51-year-old woman with no relevant medical history presented to the dermatology clinic with a rash on the neck and under the eyes of 6 days’ duration. The rash was intermittently pruritic but otherwise asymptomatic. The patient reported that she had spent extensive time on the golf course the day of the rash onset and noted that a similar rash had occurred one other time 2 to 3 months prior, also following a prolonged period on the golf course. She had been using over-the-counter fexofenadine 180 mg and over-the-counter lidocaine spray for symptom relief.
Upon physical examination, erythematous patches were appreciated in a photodistributed pattern on the arms, legs, neck, face, and chest—areas that were not covered by clothing (Figures 1-3). Due to the distribution and morphology of the erythematous patches along with clinical course of onset following exposure to various environmental agents including pesticides, herbicides, oak, and pollen, a diagnosis of PCD was made. The patient was prescribed hydrocortisone cream 2.5%, fluticasone propionate cream 0.05%, and methylprednisolone in addition to the antihistamine. Improvement was noted after 3 days with complete resolution of the skin manifestations. She was counseled on wearing clothing with a universal protection factor rating of 50+ when on the golf course and when sun exposure is expected for an extended period of time.



Causative Agents
Photodermatoses are caused by antigenic substances that lead to photosensitization acquired by either contact or oral ingestion with subsequent sensitization to UV radiation. Halogenated salicylanilide, fenticlor, hexachlorophene, bithionol and, in rare cases, sunscreens, have been reported as triggers.3 In a study performed in 2010, sunscreens, antimicrobial agents, medications, fragrances, plants/plant derivatives, and pesticides were the most commonly reported offending agents listed from highest to lowest frequency. Of the antimicrobial agents, fenticlor, a topical antimicrobial and antifungal that is now mostly used in veterinary medicine, was the most common culprit, causing 60% of cases.4,5
Clinical Manifestations
Clinical manifestations of photodermatoses vary depending upon the specific type of reaction. Examples of primary photodermatoses include polymorphous light eruption (PMLE) and solar urticaria. The cardinal symptoms of PMLE consist of severely pruritic skin lesions that can have macular, papular, papulovesicular, urticarial, multiformelike, and plaquelike variants that develop hours to days after sun exposure.3 Conversely, solar urticaria commonly develops more abruptly, with indurated plaques and wheals appearing on the arms and neck within 30 minutes of sun exposure. The lesions typically resolve within 24 hours.1
Examples of the exogenous subtype include drug-induced photosensitivity, PCD, and pseudoporphyria, with the common clinical presentation of eruption following contact with the causative agent. Drug-induced photosensitivity primarily manifests as a severe sunburnlike rash commonly caused by systemic drugs such as tetracyclines. Photocontact dermatitis is limited to sun-exposed areas of the skin and is caused by a reactive irritant such as chemicals or topical creams. Pseudoporphyria, usually caused by nonsteroidal anti-inflammatory drugs, can manifest with skin fragility and subepidermal blisters.6
Photoexacerbated photodermatoses encompass a variety of conditions ranging from hyperpigmentation disorders such as melasma to autoimmune conditions such as systemic lupus erythematosus (SLE) and dermatomyositis (DM). Common clinical features of these diseases include photodistributed erythema, often involving the cheeks, upper back, and anterior neck. Photo-exposed areas of the dorsal hands also are commonplace for both SLE and DM. Clinical manifestations of PCD are limited to sun-exposed areas of the body, specifically those that come into contact with photoallergic triggers.3 Manifestations of PCD can include pruritic eczematous eruptions resembling those of contact dermatitis 1 to 2 days after sun exposure.1
Photocontact dermatitis represents a specific sensitization via contact or oral ingestion acquired prior to sunlight exposure. It can be broken down into 2 distinct subtypes: photoallergic and photoirritant dermatitis, dependent on whether an allergic or irritant reaction is invoked.2 Plants are known to be a common trigger of photoirritant reactions, while extrinsic triggers include psoralens and medications such as tetracycline antibiotics or sulfonamides. Photoallergic reactions commonly can be caused by topical application of sunscreen or medications, namely nonsteroidal anti-inflammatory drugs.2 Clinical manifestations that may point to photoirritant dermatitis include a photodistributed eruption and classic morphology showing erythema and edema with bullae present in severe cases. These can be contrasted with the clinical manifestations of photoallergic reactions, which usually do not correlate to sun-exposed areas and consist of a monomorphous distribution pattern similar to that of eczema. Although there are distinguishing features of both subtypes of PCD, the overlapping clinical features can mimic those of solar urticaria, PMLE, cutaneous lupus erythematosus, and more systemic conditions such as SLE and DM.7
Systemic lupus erythematosus is associated with a broad range of cutaneous manifestations.8 Exposure to UV radiation is a common trigger for lupus and has the propensity to cause a malar (butterfly) rash that covers the cheeks and nasal bridge but classically spares the nasolabial folds. The rash may display confluent reddish-purple discoloration with papules and/or edema and typically is present at diagnosis in 40% to 52% of patients with SLE.8 Discoid lupus erythematosus, one of the most common cutaneous forms of lupus, manifests with various-sized coin-shaped plaques with adherent follicular hyperkeratosis and plugging. These lesions usually develop on the face, scalp, and ears but also may appear in non–sun-exposed areas.8 Dermatomyositis can manifest with photodistributed erythema affecting classic areas such as the upper back (shawl sign), anterior neck and upper chest (V-sign), and a malar rash similar to that seen in lupus, though DM classically does not spare the nasolabial folds.8,9
Because SLE and DM manifest with photodistributed rashes, it can be difficult to distinguish them from the classic symptoms of photoirritant dermatitis.9 Thus, it is imperative that providers have a high clinical index of suspicion when dealing with patients of similar presentations, as the treatment regimens vastly differ. Approaching the patient with a thorough medical history review, review of systems, biopsy (including immunofluorescence), and appropriate laboratory workup may aid in excluding more complex differential diagnoses such as SLE and DM.
Metabolic and genetic photodermatoses are more rare but can include conditions such as porphyria cutanea tarda and xeroderma pigmentosum, both of which demonstrate fragile skin, slow wound healing, and bullae on photo-exposed skin.1 Although the manifestations can be similar in these systemic conditions, they are caused by very different mechanisms. Porphyria cutanea tarda is caused by deficiencies in enzymes involved in the heme synthesis pathway, whereas xeroderma pigmentosum is caused by an alteration in DNA repair mechanisms.7
Prevalence and the Need for Standardized Testing
Most practicing dermatologists see cases of PCD due to its multiple causative agents; however, little is known about its overall prevalence. The incidence of PCD is fairly low in the general population, but this may be due to its clinical diagnosis, which excludes diagnostic testing such as phototesting and photopatch testing.10 While the incidence of photoallergic contact dermatitis also is fairly unknown, the inception of testing modalities has allowed statistics to be drawn. Research conducted in the United States has disclosed that the incidence of photoallergic contact dermatitis in individuals with a history of a prior photosensitivity eruption is approximately 10% to 20%.10 The development of guidelines and a registry for photopatch testing would aid in a greater understanding of the incidence of PCD and overall consistency of diagnosis.7 Regardless of this lack of consensus, these conditions can be properly managed and prevented if recognized clinically, while newer testing modalities would allow for confirmation of the diagnosis. It is important that any patient presenting with a history of photosensitivity be seen as a candidate for photopatch testing, especially today, as the general population is increasingly exposed to new chemicals entering the market and new social trends.7,10
Diagnosis and Treatment
It is important to consider a detailed history, including the timing, location, duration, family history, and seasonal variation of suspected photodermatoses. A thorough skin examination that takes note of the specific areas affected, morphology, and involvement of the rash or lesions can be helpful.1 Further diagnostic testing such as phototesting and photopatch testing can be employed and is especially important when distinguishing photoallergy from phototoxicity.11 Phototesting involves exposing the patient’s skin to different doses of UVA, UVB, and visible light, followed by an immediate clinical reading of the results and then a delayed reading conducted after 24 hours.1 Photopatch testing involves the application of 2 sets of identical photoallergens to prepped skin (typically cleansed with isopropyl alcohol), with one being irradiated with UVA after 24 hours and one serving as the control. A clinical assessment is conducted at 24 hours and repeated 7 days later.1 In photodermatoses, a visible reaction can be appreciated on the treatment arm while the control arm remains clear. When both sides reveal a visible reaction, this is more indicative of a light-independent allergic contact dermatitis.1
Photodermatoses occur only if there has been a specific sensitization, and therefore it is important to work with the patient to discover any new products that have been introduced into their regimen. Though many photosensitizers in personal care products (eg, antiseptics in soap and topical creams) have been discontinued, certain allergenic ingredients may remain.12 It also is important to note that sensitization to a substance that previously was not a known allergen for a particular patient can occur later in life. Avoiding further sun exposure can rapidly improve the dermatitis, and it is possible for spontaneous remission without further intervention; however, as photoallergic reactions can cause severely pruritic skin lesions, the mainstay of symptomatic treatment consists of topical corticosteroids. Oral and topical antihistamines may help alleviate the pruritus but should not be heavily relied on as this can lead to medication resistance and diminishing efficacy.3 Use of short-term oral steroids also may be considered for rapid improvement of symptoms when the patient is in moderate distress and there are no contraindications. By identifying a temporal association between the introduction of new products and the emergence of dermatitis, it may be possible to identify the causative agent. The patient should promptly discontinue the suspected agent and remain under close observation by the clinician for any further eruptions, especially following additional sun exposure.
Prevention Strategies
In the case of PCD, prevention is key. As PCD indicates a photoallergy, it is important to inform patients that the allergy will persist for a lifetime, much like in contact dermatitis; therefore, the causative agent should be avoided indefinitely.3 Patients with PCD should make intentional efforts to read ingredient lists when purchasing new personal care products to ensure they do not contain the specific causative allergen if one has been identified. Further steps should be taken to ensure proper photoprotection, including use of dense clothing and sunscreen with UVA and UVB filters (broad spectrum).3 It has also been suggested that utilizing sunscreen with ectoin, an amino acid–derived molecule, may result in increased protection against UVA-induced photodermatoses.13
Final Thoughts
Photodermatoses are a group of skin diseases caused by exposure to UV radiation. Photocontact dermatitis/photoallergy is a form of allergic contact dermatitis that results from exposure to an allergen, whether topical, oral, or environmental. The allergen is activated by exposure to UV radiation to sensitize the allergic response, resulting in a rash characterized by confluent erythematous patches or plaques, papular vesicles, and rarely blisters.3 Photocontact dermatitis, although rare, is an important differential diagnosis to consider when the presenting rash is restricted to sun-exposed areas of the skin such as the arms, legs, neck, and face. Diagnosis remains a challenge; however, new testing modalities such as photopatch testing may open the door for further confirmation and aid in proper diagnosis leading to earlier treatment times for patients. It is recommended that the clinician and patient work together to identify the possible causative agent to prevent further eruptions.
- Santoro FA, Lim HW. Update on photodermatoses. Semin Cutan Med Surg. 2011;30:229-238.
- Gimenez-Arnau A, Maurer M, De La Cuadra J, et al. Immediate contact skin reactions, an update of contact urticaria, contact urticaria syndrome and protein contact dermatitis—“a never ending story.” Eur J Dermatol. 2010;20:555-562.
- Lehmann P, Schwarz T. Photodermatoses: diagnosis and treatment. Dtsch Arztebl Int. 2011;108:135-141.
- Victor FC, Cohen DE, Soter NA. A 20-year analysis of previous and emerging allergens that elicit photoallergic contact dermatitis. J Am Acad Dermatol. 2010;62:605-610.
- Fenticlor (Code 65671). National Cancer Institute EVS Explore. Accessed October 28, 2025. https://ncithesaurus.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCIThesaurus&ns=ncit&code=C65671
- Elmets CA. Photosensitivity disorders (photodermatoses): clinical manifestations, diagnosis, and treatment. UptoDate. Updated February 23, 2023. Accessed October 28, 2025. https://www.uptodate.com/contents/photosensitivity-disorders-photodermatoses-clinical-manifestations-diagnosis-and-treatment
- Snyder M, Turrentine JE, Cruz PD Jr. Photocontact dermatitis and its clinical mimics: an overview for the allergist. Clin Rev Allergy Immunol. 2019;56:32-40.
- Cooper EE, Pisano CE, Shapiro SC. Cutaneous manifestations of “lupus”: systemic lupus erythematosus and beyond. Int J Rheumatol. 2021;2021:6610509.
- Christopher-Stine L, Amato AA, Vleugels RA. Diagnosis and differential diagnosis of dermatomyositis and polymyositis in adults. UptoDate. Updated March 3, 2025. Accessed October 28, 2025. https://www.uptodate.com/contents/diagnosis-and-differential-diagnosis-of-dermatomyositis-and-polymyositis-in-adults?search=Diagnosis%20and%20differential%20diagnosis%20of%20dermatomyositis%20and%20polymyositis%20in%20adults&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Deleo VA. Photocontact dermatitis. Dermatol Ther. 2004;17:279-288.
- Gonçalo M. Photopatch testing. In: Johansen J, Frosch P, Lepoittevin JP, eds. Contact Dermatitis. Springer; 2011:519-531.
- Enta T. Dermacase. Contact photodermatitis. Can Fam Physician. 1995;41:577,586-587.
- Duteil L, Queille-Roussel C, Aladren S, et al. Prevention of polymophic light eruption afforded by a very high broad-spectrum protection sunscreen containing ectoin. Dermatol Ther (Heidelb). 2022;12:1603-1613.
- Santoro FA, Lim HW. Update on photodermatoses. Semin Cutan Med Surg. 2011;30:229-238.
- Gimenez-Arnau A, Maurer M, De La Cuadra J, et al. Immediate contact skin reactions, an update of contact urticaria, contact urticaria syndrome and protein contact dermatitis—“a never ending story.” Eur J Dermatol. 2010;20:555-562.
- Lehmann P, Schwarz T. Photodermatoses: diagnosis and treatment. Dtsch Arztebl Int. 2011;108:135-141.
- Victor FC, Cohen DE, Soter NA. A 20-year analysis of previous and emerging allergens that elicit photoallergic contact dermatitis. J Am Acad Dermatol. 2010;62:605-610.
- Fenticlor (Code 65671). National Cancer Institute EVS Explore. Accessed October 28, 2025. https://ncithesaurus.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCIThesaurus&ns=ncit&code=C65671
- Elmets CA. Photosensitivity disorders (photodermatoses): clinical manifestations, diagnosis, and treatment. UptoDate. Updated February 23, 2023. Accessed October 28, 2025. https://www.uptodate.com/contents/photosensitivity-disorders-photodermatoses-clinical-manifestations-diagnosis-and-treatment
- Snyder M, Turrentine JE, Cruz PD Jr. Photocontact dermatitis and its clinical mimics: an overview for the allergist. Clin Rev Allergy Immunol. 2019;56:32-40.
- Cooper EE, Pisano CE, Shapiro SC. Cutaneous manifestations of “lupus”: systemic lupus erythematosus and beyond. Int J Rheumatol. 2021;2021:6610509.
- Christopher-Stine L, Amato AA, Vleugels RA. Diagnosis and differential diagnosis of dermatomyositis and polymyositis in adults. UptoDate. Updated March 3, 2025. Accessed October 28, 2025. https://www.uptodate.com/contents/diagnosis-and-differential-diagnosis-of-dermatomyositis-and-polymyositis-in-adults?search=Diagnosis%20and%20differential%20diagnosis%20of%20dermatomyositis%20and%20polymyositis%20in%20adults&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Deleo VA. Photocontact dermatitis. Dermatol Ther. 2004;17:279-288.
- Gonçalo M. Photopatch testing. In: Johansen J, Frosch P, Lepoittevin JP, eds. Contact Dermatitis. Springer; 2011:519-531.
- Enta T. Dermacase. Contact photodermatitis. Can Fam Physician. 1995;41:577,586-587.
- Duteil L, Queille-Roussel C, Aladren S, et al. Prevention of polymophic light eruption afforded by a very high broad-spectrum protection sunscreen containing ectoin. Dermatol Ther (Heidelb). 2022;12:1603-1613.
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Practice Points
- It is important to consider photodermatoses in patients presenting with a rash that is restricted to light-exposed areas of the skin, such as the arms, legs, neck, and face.
- The mainstay of treatment consists of topical corticosteroids. Oral antihistamines should not be heavily relied on, but short-term oral steroids may be considered for rapid improvement if symptoms are severe.
- It is important to note that, much like in contact dermatitis, the underlying photoallergy causing photocontact dermatitis will persist for a lifetime.
Spreading Ulcerations and Lymphadenopathy in a Traveler Returning from Costa Rica
Spreading Ulcerations and Lymphadenopathy in a Traveler Returning from Costa Rica
THE DIAGNOSIS: Cutaneous Leishmaniasis
The biopsy results revealed amastigotes at the periphery of parasitized histiocytes, consistent with a diagnosis of cutaneous leishmaniasis. Polymerase chain reaction analysis revealed Leishmania guyanensis species complex, which includes both L guyanensis and Leishmania panamensis. In this case of disseminated cutaneous leishmaniasis (Figure 1), our patient received a prolonged course of systemic therapy with oral miltefosine 50 mg 3 times daily. At the most recent follow-up appointment, she showed ongoing resolution of ulcerations, subcutaneous plaques, and lymphadenopathy on the trunk and face, but development of subcutaneous nodules continued on the arms and legs. At the next follow-up, physical examination revealed that the lesions slowly started to fade.
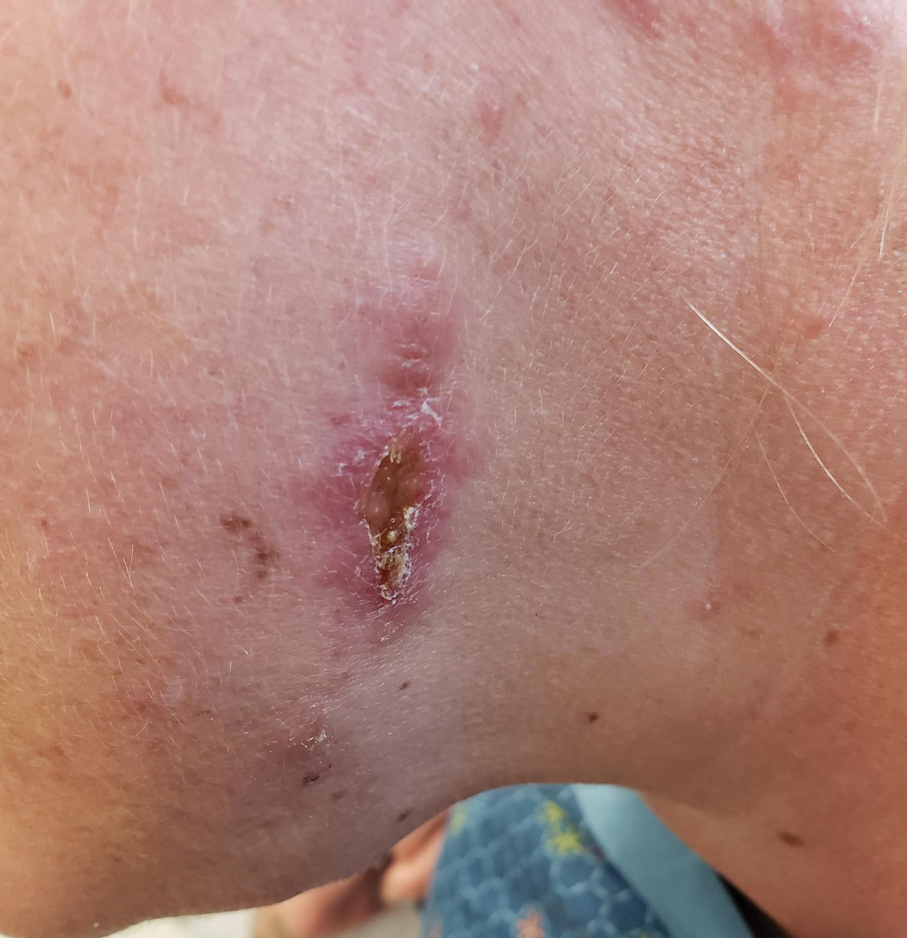
Leishmania species are parasites transmitted by bites of female sand flies, which belong to the genera Phlebotomus (Old World, Eastern Hemisphere) and Lutzomyia (New World, Western Hemisphere) genera.1 Leishmania species have a complex life cycle, propagating within human macrophages, ultimately leading to cutaneous, mucocutaneous, and visceral disease manifestations.2 Cutaneous leishmaniasis manifests classically as scattered, painless, slow-healing ulcers.3 A biopsy taken from the edge of a cutaneous ulcer for hematoxylin and eosin processing is recommended for initial diagnosis, and subsequent polymerase chain reaction of the sample is required for speciation, which guides therapeutic options.4,5 Classic hematoxylin and eosin and Giemsa stain findings include amastigotes lining the edges of parasitized histiocytes (Figure 2).
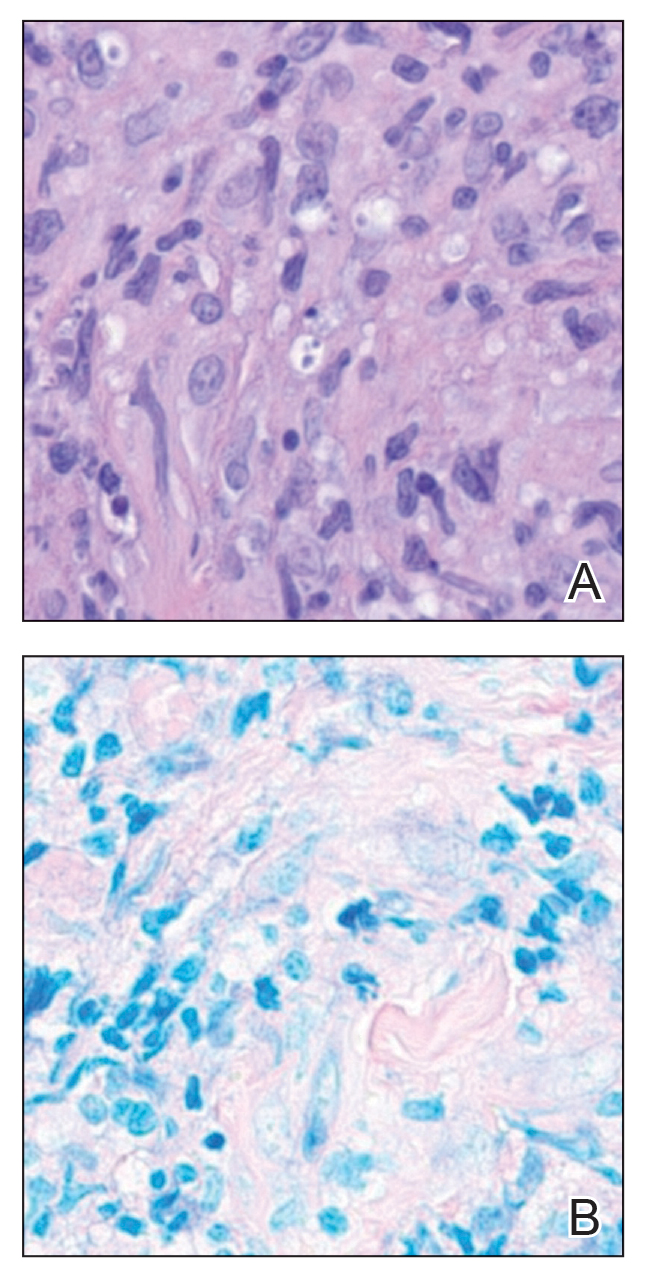
Systemic treatment options include sodium stibogluconate, amphotericin B, pentamidine, paromomycin, miltefosine, and azole antifungals.2,5 Geography often plays a critical role in selecting treatment options due to resistance rates of individual Leishmania species; for example, paromomycin compounds are more effective for cutaneous disease caused by Leishmania major than Leishmania tropica. Miltefosine is not effective for treating Leishmania braziliensis which can be acquired outside Guatemala, and higher doses of amphotericin B are recommended for visceral disease from East Africa.2,5 In patients with cutaneous leishmaniasis caused by L guyanensis, miltefosine remains a first-line option due to its oral formulation and long half-life within organisms, though there is a risk for teratogenicity.2 Amphotericin B remains the most effective treatment for visceral leishmaniasis and can be used off label to treat mucocutaneous disease or when cutaneous disease is refractory to other treatment options.3
Given the potential of L guyanensis to progress to mucocutaneous disease, monitoring for mucosal involvement should be performed at regular intervals for 6 months to 1 year.2 Treatment may be considered efficacious if no new skin lesions occur after 4 to 6 weeks of therapy; existing skin lesions should be re-epithelializing and reduced by 50% in size, with most cutaneous disease adequately controlled after 3 months of therapy.2
- Olivier M, Minguez-Menendez A, Fernandez-Prada C. Leishmania viannia guyanensis. Trends Parasitol. 2019;35:1018-1019. doi:10.1016 /j.pt.2019.06.008
- Singh R, Kashif M, Srivastava P, et al. Recent advances in chemotherapeutics for leishmaniasis: importance of the cellular biochemistry of the parasite and its molecular interaction with the host. Pathogens. 2023;12:706. doi:10.3390/pathogens12050706
- Aronson N, Herwaldt BL, Libman M, et al. Diagnosis and treatment of leishmaniasis: clinical practice guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH). Clin Infect Dis. 2016;63: 1539-1557. doi:10.1093/cid/ciw742
- Specimen Collection Guide for Laboratory Diagnosis of Leishmaniasis. Centers for Disease Control and Prevention. Accessed October 14, 2025. https://www.cdc.gov/dpdx/diagnosticprocedures /other/leish.html
- Aronson NE, Joya CA. Cutaneous leishmaniasis: updates in diagnosis and management. Infect Dis Clin North Am. 2019;33:101-117. doi:10.1016/j.idc.2018.10.004
THE DIAGNOSIS: Cutaneous Leishmaniasis
The biopsy results revealed amastigotes at the periphery of parasitized histiocytes, consistent with a diagnosis of cutaneous leishmaniasis. Polymerase chain reaction analysis revealed Leishmania guyanensis species complex, which includes both L guyanensis and Leishmania panamensis. In this case of disseminated cutaneous leishmaniasis (Figure 1), our patient received a prolonged course of systemic therapy with oral miltefosine 50 mg 3 times daily. At the most recent follow-up appointment, she showed ongoing resolution of ulcerations, subcutaneous plaques, and lymphadenopathy on the trunk and face, but development of subcutaneous nodules continued on the arms and legs. At the next follow-up, physical examination revealed that the lesions slowly started to fade.

Leishmania species are parasites transmitted by bites of female sand flies, which belong to the genera Phlebotomus (Old World, Eastern Hemisphere) and Lutzomyia (New World, Western Hemisphere) genera.1 Leishmania species have a complex life cycle, propagating within human macrophages, ultimately leading to cutaneous, mucocutaneous, and visceral disease manifestations.2 Cutaneous leishmaniasis manifests classically as scattered, painless, slow-healing ulcers.3 A biopsy taken from the edge of a cutaneous ulcer for hematoxylin and eosin processing is recommended for initial diagnosis, and subsequent polymerase chain reaction of the sample is required for speciation, which guides therapeutic options.4,5 Classic hematoxylin and eosin and Giemsa stain findings include amastigotes lining the edges of parasitized histiocytes (Figure 2).

Systemic treatment options include sodium stibogluconate, amphotericin B, pentamidine, paromomycin, miltefosine, and azole antifungals.2,5 Geography often plays a critical role in selecting treatment options due to resistance rates of individual Leishmania species; for example, paromomycin compounds are more effective for cutaneous disease caused by Leishmania major than Leishmania tropica. Miltefosine is not effective for treating Leishmania braziliensis which can be acquired outside Guatemala, and higher doses of amphotericin B are recommended for visceral disease from East Africa.2,5 In patients with cutaneous leishmaniasis caused by L guyanensis, miltefosine remains a first-line option due to its oral formulation and long half-life within organisms, though there is a risk for teratogenicity.2 Amphotericin B remains the most effective treatment for visceral leishmaniasis and can be used off label to treat mucocutaneous disease or when cutaneous disease is refractory to other treatment options.3
Given the potential of L guyanensis to progress to mucocutaneous disease, monitoring for mucosal involvement should be performed at regular intervals for 6 months to 1 year.2 Treatment may be considered efficacious if no new skin lesions occur after 4 to 6 weeks of therapy; existing skin lesions should be re-epithelializing and reduced by 50% in size, with most cutaneous disease adequately controlled after 3 months of therapy.2
THE DIAGNOSIS: Cutaneous Leishmaniasis
The biopsy results revealed amastigotes at the periphery of parasitized histiocytes, consistent with a diagnosis of cutaneous leishmaniasis. Polymerase chain reaction analysis revealed Leishmania guyanensis species complex, which includes both L guyanensis and Leishmania panamensis. In this case of disseminated cutaneous leishmaniasis (Figure 1), our patient received a prolonged course of systemic therapy with oral miltefosine 50 mg 3 times daily. At the most recent follow-up appointment, she showed ongoing resolution of ulcerations, subcutaneous plaques, and lymphadenopathy on the trunk and face, but development of subcutaneous nodules continued on the arms and legs. At the next follow-up, physical examination revealed that the lesions slowly started to fade.

Leishmania species are parasites transmitted by bites of female sand flies, which belong to the genera Phlebotomus (Old World, Eastern Hemisphere) and Lutzomyia (New World, Western Hemisphere) genera.1 Leishmania species have a complex life cycle, propagating within human macrophages, ultimately leading to cutaneous, mucocutaneous, and visceral disease manifestations.2 Cutaneous leishmaniasis manifests classically as scattered, painless, slow-healing ulcers.3 A biopsy taken from the edge of a cutaneous ulcer for hematoxylin and eosin processing is recommended for initial diagnosis, and subsequent polymerase chain reaction of the sample is required for speciation, which guides therapeutic options.4,5 Classic hematoxylin and eosin and Giemsa stain findings include amastigotes lining the edges of parasitized histiocytes (Figure 2).

Systemic treatment options include sodium stibogluconate, amphotericin B, pentamidine, paromomycin, miltefosine, and azole antifungals.2,5 Geography often plays a critical role in selecting treatment options due to resistance rates of individual Leishmania species; for example, paromomycin compounds are more effective for cutaneous disease caused by Leishmania major than Leishmania tropica. Miltefosine is not effective for treating Leishmania braziliensis which can be acquired outside Guatemala, and higher doses of amphotericin B are recommended for visceral disease from East Africa.2,5 In patients with cutaneous leishmaniasis caused by L guyanensis, miltefosine remains a first-line option due to its oral formulation and long half-life within organisms, though there is a risk for teratogenicity.2 Amphotericin B remains the most effective treatment for visceral leishmaniasis and can be used off label to treat mucocutaneous disease or when cutaneous disease is refractory to other treatment options.3
Given the potential of L guyanensis to progress to mucocutaneous disease, monitoring for mucosal involvement should be performed at regular intervals for 6 months to 1 year.2 Treatment may be considered efficacious if no new skin lesions occur after 4 to 6 weeks of therapy; existing skin lesions should be re-epithelializing and reduced by 50% in size, with most cutaneous disease adequately controlled after 3 months of therapy.2
- Olivier M, Minguez-Menendez A, Fernandez-Prada C. Leishmania viannia guyanensis. Trends Parasitol. 2019;35:1018-1019. doi:10.1016 /j.pt.2019.06.008
- Singh R, Kashif M, Srivastava P, et al. Recent advances in chemotherapeutics for leishmaniasis: importance of the cellular biochemistry of the parasite and its molecular interaction with the host. Pathogens. 2023;12:706. doi:10.3390/pathogens12050706
- Aronson N, Herwaldt BL, Libman M, et al. Diagnosis and treatment of leishmaniasis: clinical practice guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH). Clin Infect Dis. 2016;63: 1539-1557. doi:10.1093/cid/ciw742
- Specimen Collection Guide for Laboratory Diagnosis of Leishmaniasis. Centers for Disease Control and Prevention. Accessed October 14, 2025. https://www.cdc.gov/dpdx/diagnosticprocedures /other/leish.html
- Aronson NE, Joya CA. Cutaneous leishmaniasis: updates in diagnosis and management. Infect Dis Clin North Am. 2019;33:101-117. doi:10.1016/j.idc.2018.10.004
- Olivier M, Minguez-Menendez A, Fernandez-Prada C. Leishmania viannia guyanensis. Trends Parasitol. 2019;35:1018-1019. doi:10.1016 /j.pt.2019.06.008
- Singh R, Kashif M, Srivastava P, et al. Recent advances in chemotherapeutics for leishmaniasis: importance of the cellular biochemistry of the parasite and its molecular interaction with the host. Pathogens. 2023;12:706. doi:10.3390/pathogens12050706
- Aronson N, Herwaldt BL, Libman M, et al. Diagnosis and treatment of leishmaniasis: clinical practice guidelines by the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH). Clin Infect Dis. 2016;63: 1539-1557. doi:10.1093/cid/ciw742
- Specimen Collection Guide for Laboratory Diagnosis of Leishmaniasis. Centers for Disease Control and Prevention. Accessed October 14, 2025. https://www.cdc.gov/dpdx/diagnosticprocedures /other/leish.html
- Aronson NE, Joya CA. Cutaneous leishmaniasis: updates in diagnosis and management. Infect Dis Clin North Am. 2019;33:101-117. doi:10.1016/j.idc.2018.10.004
Spreading Ulcerations and Lymphadenopathy in a Traveler Returning from Costa Rica
Spreading Ulcerations and Lymphadenopathy in a Traveler Returning from Costa Rica
A 43-year-old woman presented to the dermatology clinic with widespread scaly plaques and ulcerations of 2 months’ duration. Her medical history was otherwise unremarkable. The patient reported that the eruption began after returning from a vacation to Costa Rica, during which she spent time on the beach and white-water rafting. She noted that she had been exposed to numerous insects during her trip, and that her roommate, who had accompanied her, had similar exposure history and lesions. The plaques were refractory to multiple oral antibiotics previously prescribed by primary care. Physical examination revealed submental lymphadenopathy and painless ulcerations with indurated borders without purulent drainage alongside scattered scaly papules and plaques on the face, neck, arms, and legs. A biopsy was taken from an ulceration edge on the left thigh.
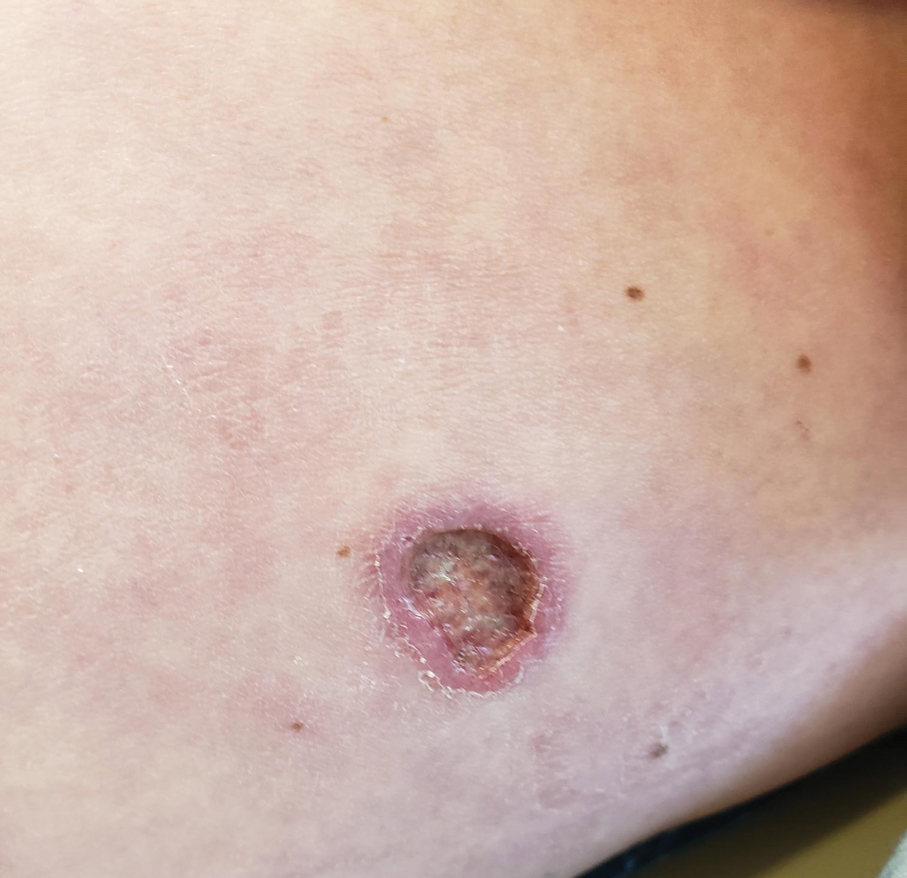
Crusted Lesion at the Implantation Site of a Pacemaker
Crusted Lesion at the Implantation Site of a Pacemaker
THE DIAGNOSIS: Pacemaker Extrusion
The lesion crust was easily scraped away to reveal extrusion of the permanent pacemaker (PPM) through the skin with a visible overlying gelatinous biofilm (Figure). The patient subsequently completed a 2-week course of clindamycin 300 mg 3 times daily followed by generator and lead removal, with reimplantation of the PPM into the right chest, as is the standard of care in the treatment of pacemaker extrusion.1
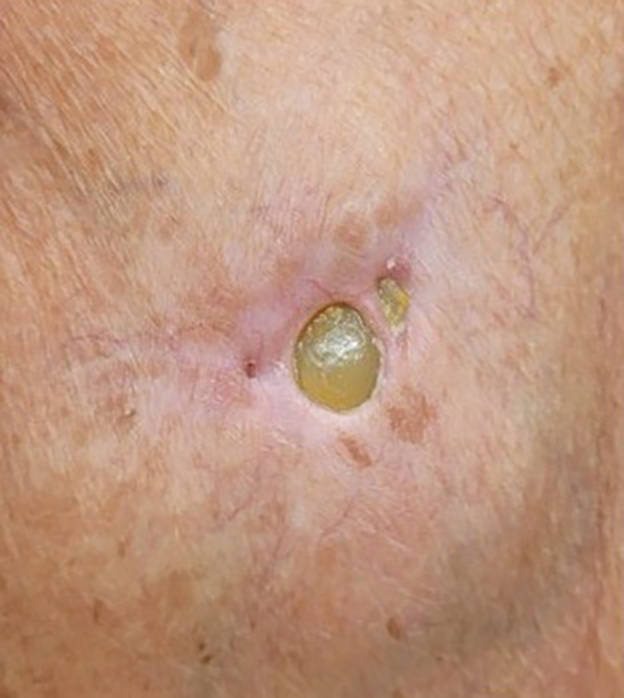
Ours is the first known reported case of pacemaker extrusion referred to dermatology with a primary concern for cutaneous malignancy. Pacemaker extrusion through the skin is not common, but it is the most common complication of PPM implantation, followed by infection.1 Pacemaker extrusion results from pressure necrosis and occurs when the PPM emerges through erythematous skin.1,2 Pacemaker extrusions generally are diagnosed by cardiology; however, it is important for dermatologists to recognize this phenomenon and differentiate it from other cutaneous pathologies, as the morphology of skin changes related to pacemaker extrusion through the skin can mimic cutaneous malignancy or other primary skin disease, especially if the outer layer of a biofilm that forms around the PPM hardens to form a crust. Our case emphasizes the importance of removing crusts when evaluating lesions.3
- Harcombe AA, Newell SA, Ludman PF, et al. Late complications following permanent pacemaker implantation or elective unit replacement. Heart. 1998;80:240-244. doi:10.1136/hrt.80.3.240
- Sanderson A, Hahn B. Pacemaker extrusion. Ann Emerg Med. 2013;62:648. doi:10.1016/j.annemergmed.2013.04.022
- Andrade AC, Hayashida MZ, Enokihara MMSES, et al. Dermoscopy of crusted lesion: diagnostic challenge and choice of technique for the analysis. An Bras Dermatol. 2021;96:387-388. doi:10.1016/j.abd.2020.06.016
THE DIAGNOSIS: Pacemaker Extrusion
The lesion crust was easily scraped away to reveal extrusion of the permanent pacemaker (PPM) through the skin with a visible overlying gelatinous biofilm (Figure). The patient subsequently completed a 2-week course of clindamycin 300 mg 3 times daily followed by generator and lead removal, with reimplantation of the PPM into the right chest, as is the standard of care in the treatment of pacemaker extrusion.1

Ours is the first known reported case of pacemaker extrusion referred to dermatology with a primary concern for cutaneous malignancy. Pacemaker extrusion through the skin is not common, but it is the most common complication of PPM implantation, followed by infection.1 Pacemaker extrusion results from pressure necrosis and occurs when the PPM emerges through erythematous skin.1,2 Pacemaker extrusions generally are diagnosed by cardiology; however, it is important for dermatologists to recognize this phenomenon and differentiate it from other cutaneous pathologies, as the morphology of skin changes related to pacemaker extrusion through the skin can mimic cutaneous malignancy or other primary skin disease, especially if the outer layer of a biofilm that forms around the PPM hardens to form a crust. Our case emphasizes the importance of removing crusts when evaluating lesions.3
THE DIAGNOSIS: Pacemaker Extrusion
The lesion crust was easily scraped away to reveal extrusion of the permanent pacemaker (PPM) through the skin with a visible overlying gelatinous biofilm (Figure). The patient subsequently completed a 2-week course of clindamycin 300 mg 3 times daily followed by generator and lead removal, with reimplantation of the PPM into the right chest, as is the standard of care in the treatment of pacemaker extrusion.1

Ours is the first known reported case of pacemaker extrusion referred to dermatology with a primary concern for cutaneous malignancy. Pacemaker extrusion through the skin is not common, but it is the most common complication of PPM implantation, followed by infection.1 Pacemaker extrusion results from pressure necrosis and occurs when the PPM emerges through erythematous skin.1,2 Pacemaker extrusions generally are diagnosed by cardiology; however, it is important for dermatologists to recognize this phenomenon and differentiate it from other cutaneous pathologies, as the morphology of skin changes related to pacemaker extrusion through the skin can mimic cutaneous malignancy or other primary skin disease, especially if the outer layer of a biofilm that forms around the PPM hardens to form a crust. Our case emphasizes the importance of removing crusts when evaluating lesions.3
- Harcombe AA, Newell SA, Ludman PF, et al. Late complications following permanent pacemaker implantation or elective unit replacement. Heart. 1998;80:240-244. doi:10.1136/hrt.80.3.240
- Sanderson A, Hahn B. Pacemaker extrusion. Ann Emerg Med. 2013;62:648. doi:10.1016/j.annemergmed.2013.04.022
- Andrade AC, Hayashida MZ, Enokihara MMSES, et al. Dermoscopy of crusted lesion: diagnostic challenge and choice of technique for the analysis. An Bras Dermatol. 2021;96:387-388. doi:10.1016/j.abd.2020.06.016
- Harcombe AA, Newell SA, Ludman PF, et al. Late complications following permanent pacemaker implantation or elective unit replacement. Heart. 1998;80:240-244. doi:10.1136/hrt.80.3.240
- Sanderson A, Hahn B. Pacemaker extrusion. Ann Emerg Med. 2013;62:648. doi:10.1016/j.annemergmed.2013.04.022
- Andrade AC, Hayashida MZ, Enokihara MMSES, et al. Dermoscopy of crusted lesion: diagnostic challenge and choice of technique for the analysis. An Bras Dermatol. 2021;96:387-388. doi:10.1016/j.abd.2020.06.016
Crusted Lesion at the Implantation Site of a Pacemaker
Crusted Lesion at the Implantation Site of a Pacemaker
A 78-year-old woman was referred to dermatology from the cardiology clinic with concerns of a nonhealing, scablike lesion on the left chest over the implantation site of a dual-chamber permanent pacemaker (PPM). Eight months prior, the patient underwent successful PPM implantation for symptomatic bradycardia and second-degree atrioventricular block. Her cardiologists subsequently noticed an oozing crusting scab at the site of implantation and eventually referred her to dermatology with concerns for squamous cell carcinoma. Physical examination at the current presentation revealed an exophytic serous crust overlying the PPM implantation site on the left chest.
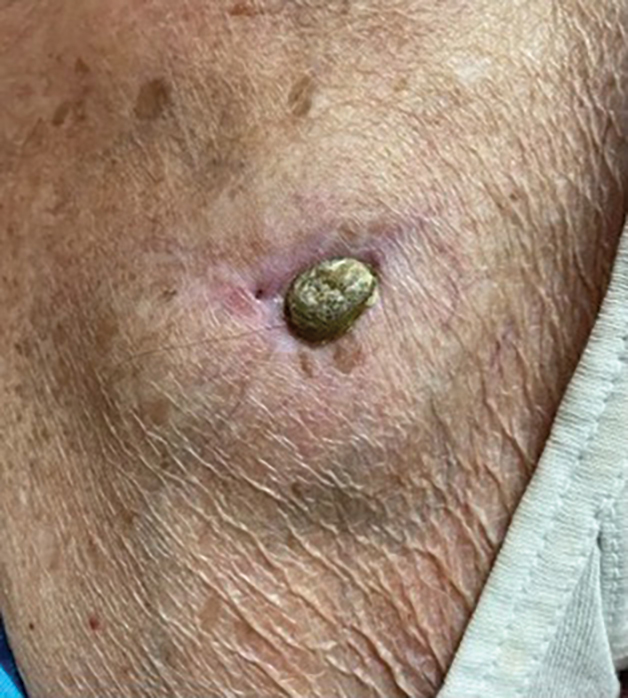
Intralesional Methotrexate: A Cost-Effective, High-Efficacy Alternative to Surgery for Cutaneous Squamous Cell Carcinoma
Intralesional Methotrexate: A Cost-Effective, High-Efficacy Alternative to Surgery for Cutaneous Squamous Cell Carcinoma
Squamous cell carcinoma (SCC) is the malignant proliferation of keratinocytes in the epidermis of the skin. Most SCCs are caused by UV light exposure, with sex and increased age acting as the primary known risk factors: SCCs are nearly twice as prevalent in men vs women, and the average age of presentation is the middle of the seventh decade of life.1 In the United States, there are an estimated 1.8 million new SCC cases annually.2 Although not usually life threatening, if left untreated, SCC can metastasize, thereby reducing the 10-year survival rate from above 90% with treatment to 16%.3-6
Most invasive SCC lesions are treated surgically, but intralesional methotrexate (IL-MTX) has emerged as an alternative treatment for cutaneous SCC. It offers the potential for lower-cost, efficacious outpatient treatment.7-12 Methotrexate competitively inhibits the enzyme dihydrofolate reductase, which converts dihydrofolate into tetrahydrofolate.13 In doing so, MTX indirectly prevents the synthesis of thymine, a nucleotide required for DNA synthesis. Thus, MTX can halt DNA synthesis and consequently, cell division. Intralesional MTX has been shown to successfully treat keratoacanthomas, lymphomas, and various inflammatory dermatologic conditions.8-12
Surgical options include standard excision, Mohs micrographic surgery, or electrodesiccation and curettage. Surgical treatment has high (92% to 99%) cure rates and typically requires only 1 or 2 appointments.14,15 Although costs can vary, one 2012 study using Medicare fee schedules found that total costs (including primary procedure, biopsy, follow-up appointments through 2 months, and other associated costs) for cutaneous SCC were $475 for electrodesiccation and curettage, $1302.92 for excision, and $2093.14 for Mohs micrographic surgery.16 For some patients, surgery is not an ideal option due to the tumor location, poor wound healing, anticoagulation, and cost. In these patients, photodynamic therapy, topical therapy with 5-fluorouracil or imiquimod, radiation, and cryotherapy are options listed in the American Academy of Dermatology guidelines.15 Compared with surgery, radiation is more demanding on the patient, often requiring multiple visits a week and including common undesirable adverse effects such as radiation dermatitis and prolonged wounds on the lower legs.17 Radiation also can be costly, with one study reporting costs between $2559 and $3431 for SCC of the forearm.18 Furthermore, in young patients, radiotherapy can increase the risk for developing nonmelanoma skin cancer later in life.16
Intralesional MTX is a localized treatment option that avoids the high costs of surgery, the side effects of radiotherapy, prolonged healing, and the systemic effects of chemotherapy. Treatment with IL-MTX can vary depending on the number of treatments necessary but usually only costs a few hundred dollars, rarely costing more than $1000.7 Although IL-MTX is less expensive, it typically requires several follow-up visits, whereas surgical removal may only require 1 visit.
Prior research has noted the efficacy of IL-MTX as a neoadjuvant therapy, with one study finding that IL-MTX can reduce the size of SCC lesions by an average of 0.52 cm2 prior to surgery.19 Several case studies also have documented the effectiveness of IL-MTX as a treatment for SCC.20-22 However, larger studies involving multiple patients to evaluate the efficacy of IL-MTX as a sole treatment for SCC are lacking. Gualdi et al23 looked at the outcomes (complete resolution, partial response, or no response) for SCC treated with IL-MTX and found that 62% (13/21) of patients experienced improvement, with 48% (10/21) experiencing at least 50% improvement. Although these results are promising, further research is needed.
Our study sought to examine IL-MTX efficacy as well as evaluate the dosage and number of appointments/sessions needed to achieve resolution of the lesions.
Methods
We conducted a retrospective chart review of patients who received only IL-MTX for clinically evident or biopsy-proven SCC at US Dermatology Partners clinics in Phoenix, Arizona, from January 1, 2022, to June 30, 2023. Patients aged 18 to 89 years were included, and they had not received other treatment for their SCC lesions such as radiation or systemic chemotherapy. Each patient received at least 1 dose of IL-MTX, beginning with a concentration of 12.5 mg/mL and with all subsequent doses at a concentration of 25 mg/mL (low dose vs high dose). Lesion resolution was categorized as no gross clinical tumor on follow-up. Patients received additional doses of IL-MTX based on the clinical appearance of their lesion(s).
Patient-level descriptive statistics are reported as mean (SD) or median (interquartile range [IQR]) for continuous variables as well as frequency and percentage for categorical variables. To account for the correlation of multiple lesions within individual patients, marginal Cox proportional hazard models were used. Time as well as cumulative dose to lesion resolution were evaluated and presented via the cumulative hazard function, while differences in resolution were estimated using separate Cox models for age, sex, and initial dose.
Results
In total, 107 different lesions from 21 patients were included in the analysis. The median number of lesions was 4 per patient (range, 1-15; IQR, 2-7), with a mean (SD) age of 80 (6) years. Patients were primarily female (81% [17/21]). From the data provided, the majority of lesions (83% [89/107]) resolved with IL-MTX. Of the 18 unresolved lesions, 5 (5%) were referred for a different procedure, and the remaining 13 (12%) were censored (lost to follow-up). Figure 1 provides the cumulative incidence function for lesion resolution. Approximately 50% of patient lesions resolved by the second appointment. Similarly, Figure 2 provides the cumulative dose function for lesion resolution; the median cumulative total dose for resolution was 5 mg (IQR, 2.5–12.5). Finally, concerning the ratio for case resolution, no difference in hazard ratio (HR) was observed for age (female vs male, HR: 1.01; 95% CI: 0.96-1.06), biological sex (HR, 1.01; 95% CI, 0.63-1.63), or initial dose (high vs low, HR: 1.13; 95% CI: 0.77-1.65).
Comment
Results of this study demonstrate the efficacy of IL-MTX for the treatment of cutaneous SCC. More than 80% of the lesions resolved by IL-MTX alone. This treatment approach is more cost-effective with fewer adverse effects when compared to other options. In our study, treatment with IL-MTX also proved to be reasonable in terms of the number of appointments and total dose required, with more than 50% of lesions resolving within 2 appointments and a median cumulative total dose of 5 mg. Intralesional MTX appears to be similarly efficacious in men and women, and the concentration of the initial dose (12.5 mg/mL vs 25 mg/mL) does not change the treatment outcome.
Although these data are encouraging for the use of IL-MTX in the treatment of SCC, future work should consider the relationships between lesion characteristics (such as size and location) and case resolution with IL-MTX as well as recurrence rates with lesions treated by IL-MTX compared to other treatment options.
Conclusion
This study demonstrated the efficacy of IL-MTX as a treatment for SCC that is cost-effective, avoids bothersome side effects, and can be accomplished in relatively few appointments. However, more data are needed to characterize the lesion type best suited to this treatment.
- Rogers HW, Weinstock MA, Feldman SR, et al. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population, 2012. JAMA Dermatol. 2015;151:1081-1086.
- The Skin Cancer Foundation. Skin cancer facts & statistics: what you need to know. Updated January 2026. Accessed January 20, 2026. https://www.skincancer.org/skin-cancer-information/skin-cancer-facts
- Rees JR, Zens MS, Celaya MO, et al. Survival after squamous cell and basal cell carcinoma of the skin: a retrospective cohort analysis. Int J Cancer. 2015;137:878-884.
- Weinberg A, Ogle C, Shin E. Metastatic cutaneous squamous cell carcinoma: an update. Dermatol Surg. 2007;33:885-899.
- Varra V, Woody NM, Reddy C, et al. Suboptimal outcomes in cutaneous squamous cell cancer of the head and neck with nodal metastases. Anticancer Res. 2018;38:5825-5830. doi:10.21873/anticanres.12923
- Epstein E, Epstein NN, Bragg K, et al. Metastases from squamous cell carcinomas of the skin. Arch Dermatol. 1968;97:245-251.
- Chitwood K, Etzkorn J, Cohen G. Topical and intralesional treatment of nonmelanoma skin cancer: efficacy and cost comparisons. Dermatol Surg. 2013;39:1306-1316
- Scalvenzi M, Patrì A, Costa C, et al. Intralesional methotrexate for the treatment of keratoacanthoma: the Neapolitan experience. Dermatol Ther. 2019;9:369-372.
- Patel NP, Cervino AL. Treatment of keratoacanthoma: is intralesional methotrexate an option? Can J Plast Surg. 2011;19:E15-E18.
- Smith C, Srivastava D, Nijhawan RI. Intralesional methotrexate for keratoacanthomas: a retrospective cohort study. JAAD Int. 2020;83:904-905.
- Blume JE, Stoll HL, Cheney RT. Treatment of primary cutaneous CD30+ anaplastic large cell lymphoma with intralesional methotrexate. J Am Acad Dermatol. 2006;54(5 Suppl):S229-S230.
- Nedelcu RI, Balaban M, Turcu G, et al. Efficacy of methotrexate as anti‑inflammatory and anti‑proliferative drug in dermatology: three case reports. Exp Ther Med. 2019;18:905-910.
- Lester RS. Methotrexate. Clin Dermatol. 1989;7:128-135.
- Roenigk RK, Roenigk HH. Current surgical management of skin cancer in dermatology. J Dermatol Surg Oncol. 1990;16:136-151.
- Alam M, Armstrong A, Baum C, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
- Wilson LS, Pregenzer M, Basu R, et al. Fee comparisons of treatments for nonmelanoma skin cancer in a private practice academic setting. Dermatol Surg. 2012;38:570-584.
- DeConti RC. Chemotherapy of squamous cell carcinoma of the skin. Semin Oncol. 2012;39:145-149.
- Rogers HW, Coldiron BM. A relative value unit–based cost comparison of treatment modalities for nonmelanoma skin cancer: effect of the loss of the Mohs multiple surgery reduction exemption. J Am Acad Dermatol. 2009;61:96-103.
- Salido-Vallejo R, Cuevas-Asencio I, Garnacho-Sucedo G, et al. Neoadjuvant intralesional methotrexate in cutaneous squamous cell carcinoma: a comparative cohort study. J Eur Acad Dermatol Venereol. 2016;30:1120-1124.
- Salido-Vallejo R, Garnacho-Saucedo G, Sánchez-Arca M, et al. Neoadjuvant intralesional methotrexate before surgical treatment of invasive squamous cell carcinoma of the lower lip. Dermatol Surg. 2012;38:1849-1850.
- Vega-González LG, Morales-Pérez MI, Molina-Pérez T, et al. Successful treatment of squamous cell carcinoma with intralesional methotrexate. JAAD Case Rep. 2022;24:68-70.
- Moye MS, Clark AH, Legler AA, et al. Intralesional methotrexate for treatment of invasive squamous cell carcinomas in a patient taking vemurafenib for treatment of metastatic melanoma. J Clin Oncol. 2016;34:E134-E136.
- Gualdi G, Caravello S, Frasci F, et al. Intralesional methotrexate for the treatment of advanced keratinocytic tumors: a multi-center retrospective study. Dermatol Ther (Heidelb). 2020;10:769-777.
Squamous cell carcinoma (SCC) is the malignant proliferation of keratinocytes in the epidermis of the skin. Most SCCs are caused by UV light exposure, with sex and increased age acting as the primary known risk factors: SCCs are nearly twice as prevalent in men vs women, and the average age of presentation is the middle of the seventh decade of life.1 In the United States, there are an estimated 1.8 million new SCC cases annually.2 Although not usually life threatening, if left untreated, SCC can metastasize, thereby reducing the 10-year survival rate from above 90% with treatment to 16%.3-6
Most invasive SCC lesions are treated surgically, but intralesional methotrexate (IL-MTX) has emerged as an alternative treatment for cutaneous SCC. It offers the potential for lower-cost, efficacious outpatient treatment.7-12 Methotrexate competitively inhibits the enzyme dihydrofolate reductase, which converts dihydrofolate into tetrahydrofolate.13 In doing so, MTX indirectly prevents the synthesis of thymine, a nucleotide required for DNA synthesis. Thus, MTX can halt DNA synthesis and consequently, cell division. Intralesional MTX has been shown to successfully treat keratoacanthomas, lymphomas, and various inflammatory dermatologic conditions.8-12
Surgical options include standard excision, Mohs micrographic surgery, or electrodesiccation and curettage. Surgical treatment has high (92% to 99%) cure rates and typically requires only 1 or 2 appointments.14,15 Although costs can vary, one 2012 study using Medicare fee schedules found that total costs (including primary procedure, biopsy, follow-up appointments through 2 months, and other associated costs) for cutaneous SCC were $475 for electrodesiccation and curettage, $1302.92 for excision, and $2093.14 for Mohs micrographic surgery.16 For some patients, surgery is not an ideal option due to the tumor location, poor wound healing, anticoagulation, and cost. In these patients, photodynamic therapy, topical therapy with 5-fluorouracil or imiquimod, radiation, and cryotherapy are options listed in the American Academy of Dermatology guidelines.15 Compared with surgery, radiation is more demanding on the patient, often requiring multiple visits a week and including common undesirable adverse effects such as radiation dermatitis and prolonged wounds on the lower legs.17 Radiation also can be costly, with one study reporting costs between $2559 and $3431 for SCC of the forearm.18 Furthermore, in young patients, radiotherapy can increase the risk for developing nonmelanoma skin cancer later in life.16
Intralesional MTX is a localized treatment option that avoids the high costs of surgery, the side effects of radiotherapy, prolonged healing, and the systemic effects of chemotherapy. Treatment with IL-MTX can vary depending on the number of treatments necessary but usually only costs a few hundred dollars, rarely costing more than $1000.7 Although IL-MTX is less expensive, it typically requires several follow-up visits, whereas surgical removal may only require 1 visit.
Prior research has noted the efficacy of IL-MTX as a neoadjuvant therapy, with one study finding that IL-MTX can reduce the size of SCC lesions by an average of 0.52 cm2 prior to surgery.19 Several case studies also have documented the effectiveness of IL-MTX as a treatment for SCC.20-22 However, larger studies involving multiple patients to evaluate the efficacy of IL-MTX as a sole treatment for SCC are lacking. Gualdi et al23 looked at the outcomes (complete resolution, partial response, or no response) for SCC treated with IL-MTX and found that 62% (13/21) of patients experienced improvement, with 48% (10/21) experiencing at least 50% improvement. Although these results are promising, further research is needed.
Our study sought to examine IL-MTX efficacy as well as evaluate the dosage and number of appointments/sessions needed to achieve resolution of the lesions.
Methods
We conducted a retrospective chart review of patients who received only IL-MTX for clinically evident or biopsy-proven SCC at US Dermatology Partners clinics in Phoenix, Arizona, from January 1, 2022, to June 30, 2023. Patients aged 18 to 89 years were included, and they had not received other treatment for their SCC lesions such as radiation or systemic chemotherapy. Each patient received at least 1 dose of IL-MTX, beginning with a concentration of 12.5 mg/mL and with all subsequent doses at a concentration of 25 mg/mL (low dose vs high dose). Lesion resolution was categorized as no gross clinical tumor on follow-up. Patients received additional doses of IL-MTX based on the clinical appearance of their lesion(s).
Patient-level descriptive statistics are reported as mean (SD) or median (interquartile range [IQR]) for continuous variables as well as frequency and percentage for categorical variables. To account for the correlation of multiple lesions within individual patients, marginal Cox proportional hazard models were used. Time as well as cumulative dose to lesion resolution were evaluated and presented via the cumulative hazard function, while differences in resolution were estimated using separate Cox models for age, sex, and initial dose.
Results
In total, 107 different lesions from 21 patients were included in the analysis. The median number of lesions was 4 per patient (range, 1-15; IQR, 2-7), with a mean (SD) age of 80 (6) years. Patients were primarily female (81% [17/21]). From the data provided, the majority of lesions (83% [89/107]) resolved with IL-MTX. Of the 18 unresolved lesions, 5 (5%) were referred for a different procedure, and the remaining 13 (12%) were censored (lost to follow-up). Figure 1 provides the cumulative incidence function for lesion resolution. Approximately 50% of patient lesions resolved by the second appointment. Similarly, Figure 2 provides the cumulative dose function for lesion resolution; the median cumulative total dose for resolution was 5 mg (IQR, 2.5–12.5). Finally, concerning the ratio for case resolution, no difference in hazard ratio (HR) was observed for age (female vs male, HR: 1.01; 95% CI: 0.96-1.06), biological sex (HR, 1.01; 95% CI, 0.63-1.63), or initial dose (high vs low, HR: 1.13; 95% CI: 0.77-1.65).


Comment
Results of this study demonstrate the efficacy of IL-MTX for the treatment of cutaneous SCC. More than 80% of the lesions resolved by IL-MTX alone. This treatment approach is more cost-effective with fewer adverse effects when compared to other options. In our study, treatment with IL-MTX also proved to be reasonable in terms of the number of appointments and total dose required, with more than 50% of lesions resolving within 2 appointments and a median cumulative total dose of 5 mg. Intralesional MTX appears to be similarly efficacious in men and women, and the concentration of the initial dose (12.5 mg/mL vs 25 mg/mL) does not change the treatment outcome.
Although these data are encouraging for the use of IL-MTX in the treatment of SCC, future work should consider the relationships between lesion characteristics (such as size and location) and case resolution with IL-MTX as well as recurrence rates with lesions treated by IL-MTX compared to other treatment options.
Conclusion
This study demonstrated the efficacy of IL-MTX as a treatment for SCC that is cost-effective, avoids bothersome side effects, and can be accomplished in relatively few appointments. However, more data are needed to characterize the lesion type best suited to this treatment.
Squamous cell carcinoma (SCC) is the malignant proliferation of keratinocytes in the epidermis of the skin. Most SCCs are caused by UV light exposure, with sex and increased age acting as the primary known risk factors: SCCs are nearly twice as prevalent in men vs women, and the average age of presentation is the middle of the seventh decade of life.1 In the United States, there are an estimated 1.8 million new SCC cases annually.2 Although not usually life threatening, if left untreated, SCC can metastasize, thereby reducing the 10-year survival rate from above 90% with treatment to 16%.3-6
Most invasive SCC lesions are treated surgically, but intralesional methotrexate (IL-MTX) has emerged as an alternative treatment for cutaneous SCC. It offers the potential for lower-cost, efficacious outpatient treatment.7-12 Methotrexate competitively inhibits the enzyme dihydrofolate reductase, which converts dihydrofolate into tetrahydrofolate.13 In doing so, MTX indirectly prevents the synthesis of thymine, a nucleotide required for DNA synthesis. Thus, MTX can halt DNA synthesis and consequently, cell division. Intralesional MTX has been shown to successfully treat keratoacanthomas, lymphomas, and various inflammatory dermatologic conditions.8-12
Surgical options include standard excision, Mohs micrographic surgery, or electrodesiccation and curettage. Surgical treatment has high (92% to 99%) cure rates and typically requires only 1 or 2 appointments.14,15 Although costs can vary, one 2012 study using Medicare fee schedules found that total costs (including primary procedure, biopsy, follow-up appointments through 2 months, and other associated costs) for cutaneous SCC were $475 for electrodesiccation and curettage, $1302.92 for excision, and $2093.14 for Mohs micrographic surgery.16 For some patients, surgery is not an ideal option due to the tumor location, poor wound healing, anticoagulation, and cost. In these patients, photodynamic therapy, topical therapy with 5-fluorouracil or imiquimod, radiation, and cryotherapy are options listed in the American Academy of Dermatology guidelines.15 Compared with surgery, radiation is more demanding on the patient, often requiring multiple visits a week and including common undesirable adverse effects such as radiation dermatitis and prolonged wounds on the lower legs.17 Radiation also can be costly, with one study reporting costs between $2559 and $3431 for SCC of the forearm.18 Furthermore, in young patients, radiotherapy can increase the risk for developing nonmelanoma skin cancer later in life.16
Intralesional MTX is a localized treatment option that avoids the high costs of surgery, the side effects of radiotherapy, prolonged healing, and the systemic effects of chemotherapy. Treatment with IL-MTX can vary depending on the number of treatments necessary but usually only costs a few hundred dollars, rarely costing more than $1000.7 Although IL-MTX is less expensive, it typically requires several follow-up visits, whereas surgical removal may only require 1 visit.
Prior research has noted the efficacy of IL-MTX as a neoadjuvant therapy, with one study finding that IL-MTX can reduce the size of SCC lesions by an average of 0.52 cm2 prior to surgery.19 Several case studies also have documented the effectiveness of IL-MTX as a treatment for SCC.20-22 However, larger studies involving multiple patients to evaluate the efficacy of IL-MTX as a sole treatment for SCC are lacking. Gualdi et al23 looked at the outcomes (complete resolution, partial response, or no response) for SCC treated with IL-MTX and found that 62% (13/21) of patients experienced improvement, with 48% (10/21) experiencing at least 50% improvement. Although these results are promising, further research is needed.
Our study sought to examine IL-MTX efficacy as well as evaluate the dosage and number of appointments/sessions needed to achieve resolution of the lesions.
Methods
We conducted a retrospective chart review of patients who received only IL-MTX for clinically evident or biopsy-proven SCC at US Dermatology Partners clinics in Phoenix, Arizona, from January 1, 2022, to June 30, 2023. Patients aged 18 to 89 years were included, and they had not received other treatment for their SCC lesions such as radiation or systemic chemotherapy. Each patient received at least 1 dose of IL-MTX, beginning with a concentration of 12.5 mg/mL and with all subsequent doses at a concentration of 25 mg/mL (low dose vs high dose). Lesion resolution was categorized as no gross clinical tumor on follow-up. Patients received additional doses of IL-MTX based on the clinical appearance of their lesion(s).
Patient-level descriptive statistics are reported as mean (SD) or median (interquartile range [IQR]) for continuous variables as well as frequency and percentage for categorical variables. To account for the correlation of multiple lesions within individual patients, marginal Cox proportional hazard models were used. Time as well as cumulative dose to lesion resolution were evaluated and presented via the cumulative hazard function, while differences in resolution were estimated using separate Cox models for age, sex, and initial dose.
Results
In total, 107 different lesions from 21 patients were included in the analysis. The median number of lesions was 4 per patient (range, 1-15; IQR, 2-7), with a mean (SD) age of 80 (6) years. Patients were primarily female (81% [17/21]). From the data provided, the majority of lesions (83% [89/107]) resolved with IL-MTX. Of the 18 unresolved lesions, 5 (5%) were referred for a different procedure, and the remaining 13 (12%) were censored (lost to follow-up). Figure 1 provides the cumulative incidence function for lesion resolution. Approximately 50% of patient lesions resolved by the second appointment. Similarly, Figure 2 provides the cumulative dose function for lesion resolution; the median cumulative total dose for resolution was 5 mg (IQR, 2.5–12.5). Finally, concerning the ratio for case resolution, no difference in hazard ratio (HR) was observed for age (female vs male, HR: 1.01; 95% CI: 0.96-1.06), biological sex (HR, 1.01; 95% CI, 0.63-1.63), or initial dose (high vs low, HR: 1.13; 95% CI: 0.77-1.65).


Comment
Results of this study demonstrate the efficacy of IL-MTX for the treatment of cutaneous SCC. More than 80% of the lesions resolved by IL-MTX alone. This treatment approach is more cost-effective with fewer adverse effects when compared to other options. In our study, treatment with IL-MTX also proved to be reasonable in terms of the number of appointments and total dose required, with more than 50% of lesions resolving within 2 appointments and a median cumulative total dose of 5 mg. Intralesional MTX appears to be similarly efficacious in men and women, and the concentration of the initial dose (12.5 mg/mL vs 25 mg/mL) does not change the treatment outcome.
Although these data are encouraging for the use of IL-MTX in the treatment of SCC, future work should consider the relationships between lesion characteristics (such as size and location) and case resolution with IL-MTX as well as recurrence rates with lesions treated by IL-MTX compared to other treatment options.
Conclusion
This study demonstrated the efficacy of IL-MTX as a treatment for SCC that is cost-effective, avoids bothersome side effects, and can be accomplished in relatively few appointments. However, more data are needed to characterize the lesion type best suited to this treatment.
- Rogers HW, Weinstock MA, Feldman SR, et al. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population, 2012. JAMA Dermatol. 2015;151:1081-1086.
- The Skin Cancer Foundation. Skin cancer facts & statistics: what you need to know. Updated January 2026. Accessed January 20, 2026. https://www.skincancer.org/skin-cancer-information/skin-cancer-facts
- Rees JR, Zens MS, Celaya MO, et al. Survival after squamous cell and basal cell carcinoma of the skin: a retrospective cohort analysis. Int J Cancer. 2015;137:878-884.
- Weinberg A, Ogle C, Shin E. Metastatic cutaneous squamous cell carcinoma: an update. Dermatol Surg. 2007;33:885-899.
- Varra V, Woody NM, Reddy C, et al. Suboptimal outcomes in cutaneous squamous cell cancer of the head and neck with nodal metastases. Anticancer Res. 2018;38:5825-5830. doi:10.21873/anticanres.12923
- Epstein E, Epstein NN, Bragg K, et al. Metastases from squamous cell carcinomas of the skin. Arch Dermatol. 1968;97:245-251.
- Chitwood K, Etzkorn J, Cohen G. Topical and intralesional treatment of nonmelanoma skin cancer: efficacy and cost comparisons. Dermatol Surg. 2013;39:1306-1316
- Scalvenzi M, Patrì A, Costa C, et al. Intralesional methotrexate for the treatment of keratoacanthoma: the Neapolitan experience. Dermatol Ther. 2019;9:369-372.
- Patel NP, Cervino AL. Treatment of keratoacanthoma: is intralesional methotrexate an option? Can J Plast Surg. 2011;19:E15-E18.
- Smith C, Srivastava D, Nijhawan RI. Intralesional methotrexate for keratoacanthomas: a retrospective cohort study. JAAD Int. 2020;83:904-905.
- Blume JE, Stoll HL, Cheney RT. Treatment of primary cutaneous CD30+ anaplastic large cell lymphoma with intralesional methotrexate. J Am Acad Dermatol. 2006;54(5 Suppl):S229-S230.
- Nedelcu RI, Balaban M, Turcu G, et al. Efficacy of methotrexate as anti‑inflammatory and anti‑proliferative drug in dermatology: three case reports. Exp Ther Med. 2019;18:905-910.
- Lester RS. Methotrexate. Clin Dermatol. 1989;7:128-135.
- Roenigk RK, Roenigk HH. Current surgical management of skin cancer in dermatology. J Dermatol Surg Oncol. 1990;16:136-151.
- Alam M, Armstrong A, Baum C, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
- Wilson LS, Pregenzer M, Basu R, et al. Fee comparisons of treatments for nonmelanoma skin cancer in a private practice academic setting. Dermatol Surg. 2012;38:570-584.
- DeConti RC. Chemotherapy of squamous cell carcinoma of the skin. Semin Oncol. 2012;39:145-149.
- Rogers HW, Coldiron BM. A relative value unit–based cost comparison of treatment modalities for nonmelanoma skin cancer: effect of the loss of the Mohs multiple surgery reduction exemption. J Am Acad Dermatol. 2009;61:96-103.
- Salido-Vallejo R, Cuevas-Asencio I, Garnacho-Sucedo G, et al. Neoadjuvant intralesional methotrexate in cutaneous squamous cell carcinoma: a comparative cohort study. J Eur Acad Dermatol Venereol. 2016;30:1120-1124.
- Salido-Vallejo R, Garnacho-Saucedo G, Sánchez-Arca M, et al. Neoadjuvant intralesional methotrexate before surgical treatment of invasive squamous cell carcinoma of the lower lip. Dermatol Surg. 2012;38:1849-1850.
- Vega-González LG, Morales-Pérez MI, Molina-Pérez T, et al. Successful treatment of squamous cell carcinoma with intralesional methotrexate. JAAD Case Rep. 2022;24:68-70.
- Moye MS, Clark AH, Legler AA, et al. Intralesional methotrexate for treatment of invasive squamous cell carcinomas in a patient taking vemurafenib for treatment of metastatic melanoma. J Clin Oncol. 2016;34:E134-E136.
- Gualdi G, Caravello S, Frasci F, et al. Intralesional methotrexate for the treatment of advanced keratinocytic tumors: a multi-center retrospective study. Dermatol Ther (Heidelb). 2020;10:769-777.
- Rogers HW, Weinstock MA, Feldman SR, et al. Incidence estimate of nonmelanoma skin cancer (keratinocyte carcinomas) in the US population, 2012. JAMA Dermatol. 2015;151:1081-1086.
- The Skin Cancer Foundation. Skin cancer facts & statistics: what you need to know. Updated January 2026. Accessed January 20, 2026. https://www.skincancer.org/skin-cancer-information/skin-cancer-facts
- Rees JR, Zens MS, Celaya MO, et al. Survival after squamous cell and basal cell carcinoma of the skin: a retrospective cohort analysis. Int J Cancer. 2015;137:878-884.
- Weinberg A, Ogle C, Shin E. Metastatic cutaneous squamous cell carcinoma: an update. Dermatol Surg. 2007;33:885-899.
- Varra V, Woody NM, Reddy C, et al. Suboptimal outcomes in cutaneous squamous cell cancer of the head and neck with nodal metastases. Anticancer Res. 2018;38:5825-5830. doi:10.21873/anticanres.12923
- Epstein E, Epstein NN, Bragg K, et al. Metastases from squamous cell carcinomas of the skin. Arch Dermatol. 1968;97:245-251.
- Chitwood K, Etzkorn J, Cohen G. Topical and intralesional treatment of nonmelanoma skin cancer: efficacy and cost comparisons. Dermatol Surg. 2013;39:1306-1316
- Scalvenzi M, Patrì A, Costa C, et al. Intralesional methotrexate for the treatment of keratoacanthoma: the Neapolitan experience. Dermatol Ther. 2019;9:369-372.
- Patel NP, Cervino AL. Treatment of keratoacanthoma: is intralesional methotrexate an option? Can J Plast Surg. 2011;19:E15-E18.
- Smith C, Srivastava D, Nijhawan RI. Intralesional methotrexate for keratoacanthomas: a retrospective cohort study. JAAD Int. 2020;83:904-905.
- Blume JE, Stoll HL, Cheney RT. Treatment of primary cutaneous CD30+ anaplastic large cell lymphoma with intralesional methotrexate. J Am Acad Dermatol. 2006;54(5 Suppl):S229-S230.
- Nedelcu RI, Balaban M, Turcu G, et al. Efficacy of methotrexate as anti‑inflammatory and anti‑proliferative drug in dermatology: three case reports. Exp Ther Med. 2019;18:905-910.
- Lester RS. Methotrexate. Clin Dermatol. 1989;7:128-135.
- Roenigk RK, Roenigk HH. Current surgical management of skin cancer in dermatology. J Dermatol Surg Oncol. 1990;16:136-151.
- Alam M, Armstrong A, Baum C, et al. Guidelines of care for the management of cutaneous squamous cell carcinoma. J Am Acad Dermatol. 2018;78:560-578.
- Wilson LS, Pregenzer M, Basu R, et al. Fee comparisons of treatments for nonmelanoma skin cancer in a private practice academic setting. Dermatol Surg. 2012;38:570-584.
- DeConti RC. Chemotherapy of squamous cell carcinoma of the skin. Semin Oncol. 2012;39:145-149.
- Rogers HW, Coldiron BM. A relative value unit–based cost comparison of treatment modalities for nonmelanoma skin cancer: effect of the loss of the Mohs multiple surgery reduction exemption. J Am Acad Dermatol. 2009;61:96-103.
- Salido-Vallejo R, Cuevas-Asencio I, Garnacho-Sucedo G, et al. Neoadjuvant intralesional methotrexate in cutaneous squamous cell carcinoma: a comparative cohort study. J Eur Acad Dermatol Venereol. 2016;30:1120-1124.
- Salido-Vallejo R, Garnacho-Saucedo G, Sánchez-Arca M, et al. Neoadjuvant intralesional methotrexate before surgical treatment of invasive squamous cell carcinoma of the lower lip. Dermatol Surg. 2012;38:1849-1850.
- Vega-González LG, Morales-Pérez MI, Molina-Pérez T, et al. Successful treatment of squamous cell carcinoma with intralesional methotrexate. JAAD Case Rep. 2022;24:68-70.
- Moye MS, Clark AH, Legler AA, et al. Intralesional methotrexate for treatment of invasive squamous cell carcinomas in a patient taking vemurafenib for treatment of metastatic melanoma. J Clin Oncol. 2016;34:E134-E136.
- Gualdi G, Caravello S, Frasci F, et al. Intralesional methotrexate for the treatment of advanced keratinocytic tumors: a multi-center retrospective study. Dermatol Ther (Heidelb). 2020;10:769-777.
Intralesional Methotrexate: A Cost-Effective, High-Efficacy Alternative to Surgery for Cutaneous Squamous Cell Carcinoma
Intralesional Methotrexate: A Cost-Effective, High-Efficacy Alternative to Surgery for Cutaneous Squamous Cell Carcinoma
PRACTICE POINTS
- Intralesional methotrexate (IL-MTX) is an efficacious treatment option for cutaneous squamous cell carcinoma lesions in patients who are not good candidates for surgical excision.
- The starting concentration of the initial IL-MTX dose did not substantially impact outcomes; however, a 25 mg/mL concentration is standard for subsequent treatments to maintain efficacy.