User login
Babies who have neonatal herpes with CNS involvement show improved neurodevelopmental outcomes at age 1 year when they are given 6 months of oral acyclovir to suppress the virus, according to a report in the Oct. 6 New England Journal of Medicine.
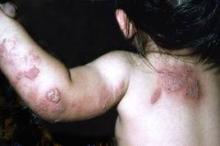
In addition, the treatment prevents cutaneous recurrences in babies who have neonatal herpes simplex virus (HSV) involving the skin, eye, or mouth, said Dr. David W. Kimberlin of the University of Alabama at Birmingham and his associates.
These results have been "implied" but not definitively established in previous small, uncontrolled case series. But the current report marks the first time that two parallel, phase III, placebo-controlled clinical trials provided strong evidence to support the use of suppressive acyclovir after neonatal HSV.
The disease is so rare that these two trials, conducted by the National Institute of Allergy and Infectious Diseases’ Collaborative Antiviral Study Group (CASG), required 11 years and 19 participating medical centers to enroll a sufficient number of patients.
In the first trial, CASG 103, 45 infants who had CNS HSV (37 subjects) or disseminated HSV with CNS involvement (8 subjects) were randomly assigned to receive 6 months of oral acyclovir or matching placebo after first undergoing a standard 21-day course of parenteral acyclovir. According to the study protocol, babies who developed a second recurrence of cutaneous lesions during the study period were removed from random assignment and given open-label acyclovir suppression.
Thirty-nine subjects (87%) either completed 6 months of treatment or had two skin recurrences and were placed on open-label treatment. The other six infants were lost to follow-up, withdrew consent, or were dropped from the study for nonadherence; one infant in the placebo group died.
Twenty-eight of the original 45 infants (62%) underwent assessment using the Bayley Scales of Infant Development at age 1 year. Those who had received suppressive acyclovir showed a significantly higher mean mental score (88.2) than did babies who had received placebo (68.1).
Moreover, 69% of the babies in the acyclovir group were classified as having normal neurologic outcomes, compared with only 33% of those in the placebo group. In the acyclovir group, 6% had mild neurologic impairment, 6% had moderate impairment, and 19% had severe impairment; the corresponding proportions in the placebo group were 8%, 25%, and 33%.
In addition, within the group randomly assigned to receive acyclovir, 15 infants received the full 6-month course of therapy, 6 received only part of the 6-month course, and 7 did not receive any active drug. Bayley mental development scores rose incrementally with increasing time on acyclovir, so that babies who took the full 6 months had a mean score of 85, those who took less than the full 6 months had a mean score of 80, and those who took no acyclovir had a mean score of 73, the investigators said (N. Engl. J. Med. 2011;365:1284-92). Bayley motor development scores did not differ significantly between the acyclovir and placebo groups.
The finding that acyclovir suppression of HSV improves neurodevelopment "should be tempered by the fact that ... assessments were not performed in 38% of subjects in the CASG 103 study. This substantial attrition renders the primary protocol end point less interpretable," they noted.
"Ongoing neurologic injury occurs in infants who survive neonatal HSV disease and ... it can be decreased by longer-term antiviral suppression."
Nevertheless, this study "provides the first controlled data that suggest that ongoing neurologic injury occurs in infants who survive neonatal HSV disease and that it can be decreased by longer-term antiviral suppression," Dr. Kimberlin and his colleagues said.
In the second trial, CASG 104, 29 babies with neonatal skin, eye, or mouth HSV were randomly assigned to receive 6 months of oral acyclovir or matching placebo after first undergoing a standard 14-day course of parenteral acyclovir. Twenty-six (90%) either completed 6 months of therapy or experienced two cutaneous recurrences and switched to open-label acyclovir. The remaining three were lost to follow-up or were dropped from the study because of nonadherence.
None of the babies in this trial developed CNS involvement during cutaneous recurrences. There were no significant differences in Bayley mental development scores between babies who received acyclovir and those who received placebo.
However, as expected, the active treatment did prevent cutaneous recurrences, compared with placebo. The positive socioeconomic effect of decreased recurrences should not be underestimated, the investigators noted.
Turning to adverse effects of active therapy, the researchers found no differences between acyclovir and placebo groups, and there were no adverse events that led to discontinuation of the study drug.
Previous studies have suggested that acyclovir may be associated with neutropenia. There were no differences in absolute neutrophil counts between acyclovir and placebo groups in either CASG 103 or CASG 104, although there was a nonsignificant trend toward neutropenia with acyclovir. Low-normal absolute neutrophil counts (500 cells/mcL or less) developed in 25% of the subjects receiving acyclovir in CASG 103 and in 20% of those receiving acyclovir in CASG 104, compared with only 5% and 7%, respectively, of babies receiving placebo.
However, "it is possible that there is indeed an association that our studies were underpowered to detect; thus, we believe that neutropenia should continue to be considered as a possible toxic effect of longer-term oral acyclovir therapy," Dr. Kimberlin and his associates said.
These studies were supported by the National Institute of Allergy and Infectious Diseases. Oral acyclovir and matching placebo were provided by GlaxoSmithKline, Alpharma USPD, and Pharm Ops. Dr. Kimberlin’s associates reported ties to numerous drug companies.
Babies who have neonatal herpes with CNS involvement show improved neurodevelopmental outcomes at age 1 year when they are given 6 months of oral acyclovir to suppress the virus, according to a report in the Oct. 6 New England Journal of Medicine.
In addition, the treatment prevents cutaneous recurrences in babies who have neonatal herpes simplex virus (HSV) involving the skin, eye, or mouth, said Dr. David W. Kimberlin of the University of Alabama at Birmingham and his associates.
These results have been "implied" but not definitively established in previous small, uncontrolled case series. But the current report marks the first time that two parallel, phase III, placebo-controlled clinical trials provided strong evidence to support the use of suppressive acyclovir after neonatal HSV.
The disease is so rare that these two trials, conducted by the National Institute of Allergy and Infectious Diseases’ Collaborative Antiviral Study Group (CASG), required 11 years and 19 participating medical centers to enroll a sufficient number of patients.
In the first trial, CASG 103, 45 infants who had CNS HSV (37 subjects) or disseminated HSV with CNS involvement (8 subjects) were randomly assigned to receive 6 months of oral acyclovir or matching placebo after first undergoing a standard 21-day course of parenteral acyclovir. According to the study protocol, babies who developed a second recurrence of cutaneous lesions during the study period were removed from random assignment and given open-label acyclovir suppression.
Thirty-nine subjects (87%) either completed 6 months of treatment or had two skin recurrences and were placed on open-label treatment. The other six infants were lost to follow-up, withdrew consent, or were dropped from the study for nonadherence; one infant in the placebo group died.
Twenty-eight of the original 45 infants (62%) underwent assessment using the Bayley Scales of Infant Development at age 1 year. Those who had received suppressive acyclovir showed a significantly higher mean mental score (88.2) than did babies who had received placebo (68.1).
Moreover, 69% of the babies in the acyclovir group were classified as having normal neurologic outcomes, compared with only 33% of those in the placebo group. In the acyclovir group, 6% had mild neurologic impairment, 6% had moderate impairment, and 19% had severe impairment; the corresponding proportions in the placebo group were 8%, 25%, and 33%.
In addition, within the group randomly assigned to receive acyclovir, 15 infants received the full 6-month course of therapy, 6 received only part of the 6-month course, and 7 did not receive any active drug. Bayley mental development scores rose incrementally with increasing time on acyclovir, so that babies who took the full 6 months had a mean score of 85, those who took less than the full 6 months had a mean score of 80, and those who took no acyclovir had a mean score of 73, the investigators said (N. Engl. J. Med. 2011;365:1284-92). Bayley motor development scores did not differ significantly between the acyclovir and placebo groups.
The finding that acyclovir suppression of HSV improves neurodevelopment "should be tempered by the fact that ... assessments were not performed in 38% of subjects in the CASG 103 study. This substantial attrition renders the primary protocol end point less interpretable," they noted.
"Ongoing neurologic injury occurs in infants who survive neonatal HSV disease and ... it can be decreased by longer-term antiviral suppression."
Nevertheless, this study "provides the first controlled data that suggest that ongoing neurologic injury occurs in infants who survive neonatal HSV disease and that it can be decreased by longer-term antiviral suppression," Dr. Kimberlin and his colleagues said.
In the second trial, CASG 104, 29 babies with neonatal skin, eye, or mouth HSV were randomly assigned to receive 6 months of oral acyclovir or matching placebo after first undergoing a standard 14-day course of parenteral acyclovir. Twenty-six (90%) either completed 6 months of therapy or experienced two cutaneous recurrences and switched to open-label acyclovir. The remaining three were lost to follow-up or were dropped from the study because of nonadherence.
None of the babies in this trial developed CNS involvement during cutaneous recurrences. There were no significant differences in Bayley mental development scores between babies who received acyclovir and those who received placebo.
However, as expected, the active treatment did prevent cutaneous recurrences, compared with placebo. The positive socioeconomic effect of decreased recurrences should not be underestimated, the investigators noted.
Turning to adverse effects of active therapy, the researchers found no differences between acyclovir and placebo groups, and there were no adverse events that led to discontinuation of the study drug.
Previous studies have suggested that acyclovir may be associated with neutropenia. There were no differences in absolute neutrophil counts between acyclovir and placebo groups in either CASG 103 or CASG 104, although there was a nonsignificant trend toward neutropenia with acyclovir. Low-normal absolute neutrophil counts (500 cells/mcL or less) developed in 25% of the subjects receiving acyclovir in CASG 103 and in 20% of those receiving acyclovir in CASG 104, compared with only 5% and 7%, respectively, of babies receiving placebo.
However, "it is possible that there is indeed an association that our studies were underpowered to detect; thus, we believe that neutropenia should continue to be considered as a possible toxic effect of longer-term oral acyclovir therapy," Dr. Kimberlin and his associates said.
These studies were supported by the National Institute of Allergy and Infectious Diseases. Oral acyclovir and matching placebo were provided by GlaxoSmithKline, Alpharma USPD, and Pharm Ops. Dr. Kimberlin’s associates reported ties to numerous drug companies.
Babies who have neonatal herpes with CNS involvement show improved neurodevelopmental outcomes at age 1 year when they are given 6 months of oral acyclovir to suppress the virus, according to a report in the Oct. 6 New England Journal of Medicine.
In addition, the treatment prevents cutaneous recurrences in babies who have neonatal herpes simplex virus (HSV) involving the skin, eye, or mouth, said Dr. David W. Kimberlin of the University of Alabama at Birmingham and his associates.
These results have been "implied" but not definitively established in previous small, uncontrolled case series. But the current report marks the first time that two parallel, phase III, placebo-controlled clinical trials provided strong evidence to support the use of suppressive acyclovir after neonatal HSV.
The disease is so rare that these two trials, conducted by the National Institute of Allergy and Infectious Diseases’ Collaborative Antiviral Study Group (CASG), required 11 years and 19 participating medical centers to enroll a sufficient number of patients.
In the first trial, CASG 103, 45 infants who had CNS HSV (37 subjects) or disseminated HSV with CNS involvement (8 subjects) were randomly assigned to receive 6 months of oral acyclovir or matching placebo after first undergoing a standard 21-day course of parenteral acyclovir. According to the study protocol, babies who developed a second recurrence of cutaneous lesions during the study period were removed from random assignment and given open-label acyclovir suppression.
Thirty-nine subjects (87%) either completed 6 months of treatment or had two skin recurrences and were placed on open-label treatment. The other six infants were lost to follow-up, withdrew consent, or were dropped from the study for nonadherence; one infant in the placebo group died.
Twenty-eight of the original 45 infants (62%) underwent assessment using the Bayley Scales of Infant Development at age 1 year. Those who had received suppressive acyclovir showed a significantly higher mean mental score (88.2) than did babies who had received placebo (68.1).
Moreover, 69% of the babies in the acyclovir group were classified as having normal neurologic outcomes, compared with only 33% of those in the placebo group. In the acyclovir group, 6% had mild neurologic impairment, 6% had moderate impairment, and 19% had severe impairment; the corresponding proportions in the placebo group were 8%, 25%, and 33%.
In addition, within the group randomly assigned to receive acyclovir, 15 infants received the full 6-month course of therapy, 6 received only part of the 6-month course, and 7 did not receive any active drug. Bayley mental development scores rose incrementally with increasing time on acyclovir, so that babies who took the full 6 months had a mean score of 85, those who took less than the full 6 months had a mean score of 80, and those who took no acyclovir had a mean score of 73, the investigators said (N. Engl. J. Med. 2011;365:1284-92). Bayley motor development scores did not differ significantly between the acyclovir and placebo groups.
The finding that acyclovir suppression of HSV improves neurodevelopment "should be tempered by the fact that ... assessments were not performed in 38% of subjects in the CASG 103 study. This substantial attrition renders the primary protocol end point less interpretable," they noted.
"Ongoing neurologic injury occurs in infants who survive neonatal HSV disease and ... it can be decreased by longer-term antiviral suppression."
Nevertheless, this study "provides the first controlled data that suggest that ongoing neurologic injury occurs in infants who survive neonatal HSV disease and that it can be decreased by longer-term antiviral suppression," Dr. Kimberlin and his colleagues said.
In the second trial, CASG 104, 29 babies with neonatal skin, eye, or mouth HSV were randomly assigned to receive 6 months of oral acyclovir or matching placebo after first undergoing a standard 14-day course of parenteral acyclovir. Twenty-six (90%) either completed 6 months of therapy or experienced two cutaneous recurrences and switched to open-label acyclovir. The remaining three were lost to follow-up or were dropped from the study because of nonadherence.
None of the babies in this trial developed CNS involvement during cutaneous recurrences. There were no significant differences in Bayley mental development scores between babies who received acyclovir and those who received placebo.
However, as expected, the active treatment did prevent cutaneous recurrences, compared with placebo. The positive socioeconomic effect of decreased recurrences should not be underestimated, the investigators noted.
Turning to adverse effects of active therapy, the researchers found no differences between acyclovir and placebo groups, and there were no adverse events that led to discontinuation of the study drug.
Previous studies have suggested that acyclovir may be associated with neutropenia. There were no differences in absolute neutrophil counts between acyclovir and placebo groups in either CASG 103 or CASG 104, although there was a nonsignificant trend toward neutropenia with acyclovir. Low-normal absolute neutrophil counts (500 cells/mcL or less) developed in 25% of the subjects receiving acyclovir in CASG 103 and in 20% of those receiving acyclovir in CASG 104, compared with only 5% and 7%, respectively, of babies receiving placebo.
However, "it is possible that there is indeed an association that our studies were underpowered to detect; thus, we believe that neutropenia should continue to be considered as a possible toxic effect of longer-term oral acyclovir therapy," Dr. Kimberlin and his associates said.
These studies were supported by the National Institute of Allergy and Infectious Diseases. Oral acyclovir and matching placebo were provided by GlaxoSmithKline, Alpharma USPD, and Pharm Ops. Dr. Kimberlin’s associates reported ties to numerous drug companies.
FROM NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Babies with neonatal herpes who had received 6 months of oral acyclovir to suppress CNS HSV had a significantly higher mean score (88.2) on the Bayley mental development assessment at age 1 year than did those given placebo (68.1).
Data Source: Two parallel, multicenter, phase III, randomized, double-blind clinical trials including 45 patients with neonatal HSV involving the CNS and 29 with HSV involving the skin, eye, or mouth who were followed for 1 year.
Disclosures: These studies were supported by the National Institute of Allergy and Infectious Diseases. Oral acyclovir and matching placebo were provided by GlaxoSmithKline, Alpharma USPD, and Pharm Ops. Dr. Kimberlin’s associates reported ties to numerous drug companies.