User login
Topical NSAIDs Safe in Elderly Osteoarthritis Patients
LONG BEACH, CALIF. — A higher rate of adverse events in older patients with knee osteoarthritis treated topically for 12 weeks with an NSAID, compared with placebo, was caused mainly by application-site reactions but included one serious cardiovascular event that might have been related to the drug treatment, a post hoc analysis of data on 538 patients found.
The investigators analyzed data on people aged 65 years and older with symptomatic knee osteoarthritis (433 had comorbid hypertension, diabetes, or cardiovascular disease). Their source was three larger randomized, double-blind trials—two unpublished—that had looked at broader populations. Patients applied 4 g/day of either diclofenac sodium 1% gel (Voltaren) or the drug's vehicle to one painful knee.
One 80-year-old woman with hypertension and diabetes, among the 274 patients on diclofenac sodium 1% gel, developed deep vein thrombosis and pulmonary embolism that possibly was related to treatment, Dr. H. Richard Barthel and his associates reported in a poster presentation.
Overall, 56% of patients on diclofenac gel developed adverse events, compared with 44% of 264 patients treated with placebo gel, added Dr. Barthel, a rheumatologist in Santa Barbara, Calif.
NSAIDs are known to increase risk for cardiovascular or renal problems in a dose-related fashion, especially in older patients and people with hypertension, diabetes, or cardiovascular disease. Topical formulations may reduce this risk by reducing systemic exposure to NSAIDs compared with oral formulations. The ad hoc analysis compared the gel only to placebo, not to oral therapy, and found higher rates of adverse events for the drug vs. placebo.
Application-site reactions occurred in 8.8% on diclofenac gel and 1.1% on placebo. Serious adverse events occurred in 2.6% on diclofenac and 1.1% on placebo. Adverse cardiovascular events were seen in 2.6% on diclofenac and 1.1% on placebo. Adverse renal events were seen in 1.1% on diclofenac and 0.4% on placebo.
Among more common adverse events, 11% of subjects on diclofenac and 10% on placebo reported headache, 8% on diclofenac and 7% on placebo reported arthralgia, and 8% on diclofenac and 6% on placebo reported back pain.
The analysis included 307 patients with hypertension, 84 with diabetes, and 42 with cardiovascular disease. In the hypertension subgroup, adverse events were seen in 54% of 159 people randomized to diclofenac gel, compared with 45% of 148 people using placebo. In the diabetes subgroup, adverse events occurred in 19 (51%) of 37 patients treated with diclofenac and in 21 (48%) of 47 treated with placebo. In the subgroup with cardiovascular disease, adverse events occurred in 15 (56%) of 27 on diclofenac and in 2 (13%) of 15 on placebo, though none developed an adverse cardiovascular event.
Dr. Barthel conducted the study under a research contract for Novartis, which makes Voltaren. His associates in the study were employees of Novartis or of Endo Pharmaceuticals, which markets the drug.
The therapy of osteoarthritis remains insufficient in many patients.
It is particularly problematic in the elderly where there are often
concomitant diseases that limit our options for several of the oral
medications, particularly NSAIDs and potent analgesics. The recent Food
and Drug Administration approval of diclofenac has changed the
therapeutic paradigm. Diclofenac gel 1% has been approved for
osteoarthritis of the knee, hand, and other superficial joints, and
Pennsaid has been approved for osteoarthritis of the knee.
In this posthoc pooled analysis of 538 patients over 65 years of age
treated for 3 months with the diclofenac gel 1% for osteoarthritis of
the knee, we see an increase in irritation at the site of application,
but a minimal increase in adverse events involving blood pressure, renal
function, hepatic dysfunction, and gastrointestinal ulcer disease.
Pharmacokinetic studies have shown that systemic absorption of the
topical diclofenac is 40 times less than oral diclofenac. This improved
safety allows us to provide therapy to patients otherwise unable to
receive anti-inflammatory drugs.
It will be no surprise if the guidelines for therapy of
osteoarthritis from the United States will soon approximate those from
Europe, where topical NSAIDs are part of the therapeutic algorithm for
osteoarthritis.
ROY D. ALTMAN, M.D., is professor of medicine in the division of
rheumatology and immunology at the University of California, Los
Angeles. He has been a consultant to Novartis, Eli Lilly, Ferring
Pharmaceuticals, and Rottapharm/Madaus.
The therapy of osteoarthritis remains insufficient in many patients.
It is particularly problematic in the elderly where there are often
concomitant diseases that limit our options for several of the oral
medications, particularly NSAIDs and potent analgesics. The recent Food
and Drug Administration approval of diclofenac has changed the
therapeutic paradigm. Diclofenac gel 1% has been approved for
osteoarthritis of the knee, hand, and other superficial joints, and
Pennsaid has been approved for osteoarthritis of the knee.
In this posthoc pooled analysis of 538 patients over 65 years of age
treated for 3 months with the diclofenac gel 1% for osteoarthritis of
the knee, we see an increase in irritation at the site of application,
but a minimal increase in adverse events involving blood pressure, renal
function, hepatic dysfunction, and gastrointestinal ulcer disease.
Pharmacokinetic studies have shown that systemic absorption of the
topical diclofenac is 40 times less than oral diclofenac. This improved
safety allows us to provide therapy to patients otherwise unable to
receive anti-inflammatory drugs.
It will be no surprise if the guidelines for therapy of
osteoarthritis from the United States will soon approximate those from
Europe, where topical NSAIDs are part of the therapeutic algorithm for
osteoarthritis.
ROY D. ALTMAN, M.D., is professor of medicine in the division of
rheumatology and immunology at the University of California, Los
Angeles. He has been a consultant to Novartis, Eli Lilly, Ferring
Pharmaceuticals, and Rottapharm/Madaus.
The therapy of osteoarthritis remains insufficient in many patients.
It is particularly problematic in the elderly where there are often
concomitant diseases that limit our options for several of the oral
medications, particularly NSAIDs and potent analgesics. The recent Food
and Drug Administration approval of diclofenac has changed the
therapeutic paradigm. Diclofenac gel 1% has been approved for
osteoarthritis of the knee, hand, and other superficial joints, and
Pennsaid has been approved for osteoarthritis of the knee.
In this posthoc pooled analysis of 538 patients over 65 years of age
treated for 3 months with the diclofenac gel 1% for osteoarthritis of
the knee, we see an increase in irritation at the site of application,
but a minimal increase in adverse events involving blood pressure, renal
function, hepatic dysfunction, and gastrointestinal ulcer disease.
Pharmacokinetic studies have shown that systemic absorption of the
topical diclofenac is 40 times less than oral diclofenac. This improved
safety allows us to provide therapy to patients otherwise unable to
receive anti-inflammatory drugs.
It will be no surprise if the guidelines for therapy of
osteoarthritis from the United States will soon approximate those from
Europe, where topical NSAIDs are part of the therapeutic algorithm for
osteoarthritis.
ROY D. ALTMAN, M.D., is professor of medicine in the division of
rheumatology and immunology at the University of California, Los
Angeles. He has been a consultant to Novartis, Eli Lilly, Ferring
Pharmaceuticals, and Rottapharm/Madaus.
LONG BEACH, CALIF. — A higher rate of adverse events in older patients with knee osteoarthritis treated topically for 12 weeks with an NSAID, compared with placebo, was caused mainly by application-site reactions but included one serious cardiovascular event that might have been related to the drug treatment, a post hoc analysis of data on 538 patients found.
The investigators analyzed data on people aged 65 years and older with symptomatic knee osteoarthritis (433 had comorbid hypertension, diabetes, or cardiovascular disease). Their source was three larger randomized, double-blind trials—two unpublished—that had looked at broader populations. Patients applied 4 g/day of either diclofenac sodium 1% gel (Voltaren) or the drug's vehicle to one painful knee.
One 80-year-old woman with hypertension and diabetes, among the 274 patients on diclofenac sodium 1% gel, developed deep vein thrombosis and pulmonary embolism that possibly was related to treatment, Dr. H. Richard Barthel and his associates reported in a poster presentation.
Overall, 56% of patients on diclofenac gel developed adverse events, compared with 44% of 264 patients treated with placebo gel, added Dr. Barthel, a rheumatologist in Santa Barbara, Calif.
NSAIDs are known to increase risk for cardiovascular or renal problems in a dose-related fashion, especially in older patients and people with hypertension, diabetes, or cardiovascular disease. Topical formulations may reduce this risk by reducing systemic exposure to NSAIDs compared with oral formulations. The ad hoc analysis compared the gel only to placebo, not to oral therapy, and found higher rates of adverse events for the drug vs. placebo.
Application-site reactions occurred in 8.8% on diclofenac gel and 1.1% on placebo. Serious adverse events occurred in 2.6% on diclofenac and 1.1% on placebo. Adverse cardiovascular events were seen in 2.6% on diclofenac and 1.1% on placebo. Adverse renal events were seen in 1.1% on diclofenac and 0.4% on placebo.
Among more common adverse events, 11% of subjects on diclofenac and 10% on placebo reported headache, 8% on diclofenac and 7% on placebo reported arthralgia, and 8% on diclofenac and 6% on placebo reported back pain.
The analysis included 307 patients with hypertension, 84 with diabetes, and 42 with cardiovascular disease. In the hypertension subgroup, adverse events were seen in 54% of 159 people randomized to diclofenac gel, compared with 45% of 148 people using placebo. In the diabetes subgroup, adverse events occurred in 19 (51%) of 37 patients treated with diclofenac and in 21 (48%) of 47 treated with placebo. In the subgroup with cardiovascular disease, adverse events occurred in 15 (56%) of 27 on diclofenac and in 2 (13%) of 15 on placebo, though none developed an adverse cardiovascular event.
Dr. Barthel conducted the study under a research contract for Novartis, which makes Voltaren. His associates in the study were employees of Novartis or of Endo Pharmaceuticals, which markets the drug.
LONG BEACH, CALIF. — A higher rate of adverse events in older patients with knee osteoarthritis treated topically for 12 weeks with an NSAID, compared with placebo, was caused mainly by application-site reactions but included one serious cardiovascular event that might have been related to the drug treatment, a post hoc analysis of data on 538 patients found.
The investigators analyzed data on people aged 65 years and older with symptomatic knee osteoarthritis (433 had comorbid hypertension, diabetes, or cardiovascular disease). Their source was three larger randomized, double-blind trials—two unpublished—that had looked at broader populations. Patients applied 4 g/day of either diclofenac sodium 1% gel (Voltaren) or the drug's vehicle to one painful knee.
One 80-year-old woman with hypertension and diabetes, among the 274 patients on diclofenac sodium 1% gel, developed deep vein thrombosis and pulmonary embolism that possibly was related to treatment, Dr. H. Richard Barthel and his associates reported in a poster presentation.
Overall, 56% of patients on diclofenac gel developed adverse events, compared with 44% of 264 patients treated with placebo gel, added Dr. Barthel, a rheumatologist in Santa Barbara, Calif.
NSAIDs are known to increase risk for cardiovascular or renal problems in a dose-related fashion, especially in older patients and people with hypertension, diabetes, or cardiovascular disease. Topical formulations may reduce this risk by reducing systemic exposure to NSAIDs compared with oral formulations. The ad hoc analysis compared the gel only to placebo, not to oral therapy, and found higher rates of adverse events for the drug vs. placebo.
Application-site reactions occurred in 8.8% on diclofenac gel and 1.1% on placebo. Serious adverse events occurred in 2.6% on diclofenac and 1.1% on placebo. Adverse cardiovascular events were seen in 2.6% on diclofenac and 1.1% on placebo. Adverse renal events were seen in 1.1% on diclofenac and 0.4% on placebo.
Among more common adverse events, 11% of subjects on diclofenac and 10% on placebo reported headache, 8% on diclofenac and 7% on placebo reported arthralgia, and 8% on diclofenac and 6% on placebo reported back pain.
The analysis included 307 patients with hypertension, 84 with diabetes, and 42 with cardiovascular disease. In the hypertension subgroup, adverse events were seen in 54% of 159 people randomized to diclofenac gel, compared with 45% of 148 people using placebo. In the diabetes subgroup, adverse events occurred in 19 (51%) of 37 patients treated with diclofenac and in 21 (48%) of 47 treated with placebo. In the subgroup with cardiovascular disease, adverse events occurred in 15 (56%) of 27 on diclofenac and in 2 (13%) of 15 on placebo, though none developed an adverse cardiovascular event.
Dr. Barthel conducted the study under a research contract for Novartis, which makes Voltaren. His associates in the study were employees of Novartis or of Endo Pharmaceuticals, which markets the drug.
Labetalol Use Appears Safe During Pregnancy
SAN FRANCISCO — Results of nonstress tests in 112 pregnant women being treated for chronic hypertension from January 2003 to September 2007 did not differ significantly in patients on labetalol, compared with those on methyldopa, results of a retrospective study found.
“Attending physicians should feel comfortable using labetalol or methyldopa for pregnant patients with hypertension. Those medications have no effect on the baby,” Dr. Ramata Niang said in an interview at her prize-winning poster presentation at the meeting.
Nonstress tests were reactive in 84% of 76 patients on labetalol and in 81% of 36 patients on methyldopa, a difference that was not statistically significant, reported Dr. Niang, an ob.gyn. at the University of Illinois at Chicago.
The investigators used the average of each patient's nonstress test results to categorize results as reactive or nonreactive.
The study started with charts on 188 women treated for hypertension during pregnancy and excluded women with multiple-gestation pregnancies, other antihypertensive treatment, or incomplete prenatal testing charts, to focus on the remaining 112 patients.
Among secondary outcomes, there were no significant differences between the two treatment groups in maternal age (29 years for women on labetalol and 31 years for those on methyldopa), gestational age at delivery (37 and 38 weeks), birth weight (2,823 g and 3,048 g), or the rate of preeclampsia (less than 1% in both groups).
SAN FRANCISCO — Results of nonstress tests in 112 pregnant women being treated for chronic hypertension from January 2003 to September 2007 did not differ significantly in patients on labetalol, compared with those on methyldopa, results of a retrospective study found.
“Attending physicians should feel comfortable using labetalol or methyldopa for pregnant patients with hypertension. Those medications have no effect on the baby,” Dr. Ramata Niang said in an interview at her prize-winning poster presentation at the meeting.
Nonstress tests were reactive in 84% of 76 patients on labetalol and in 81% of 36 patients on methyldopa, a difference that was not statistically significant, reported Dr. Niang, an ob.gyn. at the University of Illinois at Chicago.
The investigators used the average of each patient's nonstress test results to categorize results as reactive or nonreactive.
The study started with charts on 188 women treated for hypertension during pregnancy and excluded women with multiple-gestation pregnancies, other antihypertensive treatment, or incomplete prenatal testing charts, to focus on the remaining 112 patients.
Among secondary outcomes, there were no significant differences between the two treatment groups in maternal age (29 years for women on labetalol and 31 years for those on methyldopa), gestational age at delivery (37 and 38 weeks), birth weight (2,823 g and 3,048 g), or the rate of preeclampsia (less than 1% in both groups).
SAN FRANCISCO — Results of nonstress tests in 112 pregnant women being treated for chronic hypertension from January 2003 to September 2007 did not differ significantly in patients on labetalol, compared with those on methyldopa, results of a retrospective study found.
“Attending physicians should feel comfortable using labetalol or methyldopa for pregnant patients with hypertension. Those medications have no effect on the baby,” Dr. Ramata Niang said in an interview at her prize-winning poster presentation at the meeting.
Nonstress tests were reactive in 84% of 76 patients on labetalol and in 81% of 36 patients on methyldopa, a difference that was not statistically significant, reported Dr. Niang, an ob.gyn. at the University of Illinois at Chicago.
The investigators used the average of each patient's nonstress test results to categorize results as reactive or nonreactive.
The study started with charts on 188 women treated for hypertension during pregnancy and excluded women with multiple-gestation pregnancies, other antihypertensive treatment, or incomplete prenatal testing charts, to focus on the remaining 112 patients.
Among secondary outcomes, there were no significant differences between the two treatment groups in maternal age (29 years for women on labetalol and 31 years for those on methyldopa), gestational age at delivery (37 and 38 weeks), birth weight (2,823 g and 3,048 g), or the rate of preeclampsia (less than 1% in both groups).
Long-Acting Implants Are the Frontier for Contraception
SAN FRANCISCO — Fifty years after the introduction of oral contraceptives, physicians are looking to long-acting removable contraceptives to provide the next leap forward in preventing unintended pregnancy.
Three themes emerged in interviews with multiple physicians who gave separate presentations on contraception in sessions at the annual meeting of the American College of Obstetricians and Gynecologists.
First, nothing in the future of contraception is likely to match the revolutionary impact of oral contraception. Second, the great advances in contraception over the last half-decade still are accompanied by an unacceptably high rate of unintended pregnancies. And third, when asked to imagine what contraception might look like 50 years from now, most physicians looked to greater use of long-acting removable contraception (LARC) such as intrauterine devices (IUDs) or implants.
“The Pill is still one of the most important inventions ever, in terms of advancing not just women's health care but also women's rights and the ability for women to be in the workforce and to contribute in more ways that they want as members of society,” said Dr. Sarah Prager of the University of Washington, Seattle.
Long-acting removable contraception is the wave of the future, she said, because once the devices are inserted, they don't require patient participation in contraception, which has been one of the major stumbling blocks in contraceptive failures.
The next 50 years probably will see improvements in long-acting removable contraceptives, Dr. Prager added. “Our longest-acting LARC method only lasts for 10–12 years,” she noted.
Dr. Joseph Anthony Ogburn of the University of New Mexico, Albuquerque, shared the same vision for the future. “We did better than we did in the past, but we have great room for improvement,” he said. Approximately half of U.S. pregnancies are unintended, giving the United States the worst unintended pregnancy rate among developed countries.
Only about 1%–2% of U.S. women on contraception use an IUD, and less than 1% have a contraceptive implant. “If we could increase those numbers significantly, I think we could have a dramatic impact on the unintended pregnancy rate,” he said. Advances in the next 50 years probably will entail tweaking existing long-acting removable contraceptives to make them more acceptable.
But “I don't see anything as revolutionary as the Pill coming along any time in my lifetime,” he added.
Dr. Pouru Bhiwandi was more enthusiastic. “It's a very, very exciting time for all of contraception,” said Dr. Bhiwandi, an ob.gyn. in Raleigh, N.C., and an international consultant in women's health. “We have so many choices today, which we've never had before.”
Since the first oral contraceptive was approved in 1960, women's options grew with approval of more than 40 birth control pills and the development of other forms of hormonal contraception in transdermal patches, the vaginal ring, implants, and intrauterine devices (IUDs).
Modifications in dosing since the first oral contraceptives, which contained nearly four times the amount of estrogen and nearly 10 times the amount of progestin as today's formulations, have made the Pill safer and more acceptable, as have the development of newer estrogens and progesterones, she added. Newer regimens mean women no longer have to bleed while on hormonal contraception.
On the horizon are “exciting possibilities” for new products in barrier contraception that are both spermicidal and microbicidal to prevent sexually transmitted infections, plus “a whole range of new IUDs, a 1-year vaginal ring with a new progestin, and other products, Dr. Bhiwandi said.
The U.S. should emulate Europe, where longer-acting methods of birth control, including IUDs and contraceptives are much more prevalent, said Dr. Andrew Kaunitz, professor of ob.gyn. at the University of Florida, Jacksonville.
Although U.S. clinical trials report a failure rate of 1%–2% with oral contraceptive use, in “typical practice” it's much higher, Dr. Kaunitz said—around nine women per 100 couples annually have unintended pregnancies on the Pill. “That's too high,” he said.
Dr. Bliss Kaneshiro of the University of Hawaii, Honolulu, noted that long-acting contraceptive devices are expensive and not always covered by insurance. “One of the big challenges, I think, is cost,” she said. “Our challenge for the next 50 years is improving access to those good contraceptive methods.”
Dr. David Plourd of the Naval Medical Center, San Diego, said he hopes to see more options in long-acting removable contraceptives in the next 50 years, such as “a flexible IUD, not the semi-rigid T-shaped ones we have currently.”
The development of contraception methods targeted to men should play more of a significant role, he added.
Dr. Kaunitz disclosed financial relationships with Teva Pharmaceuticals, Bayer, Ortho (Johnson & Johnson), Merck, Procter & Gamble, Becton Dickinson, Sanofi-Aventis, and Medical Diagnostic Laboratories. Dr. Ogburn has been a consultant for Organon/Schering-Plough. Dr. Bhiwandi disclosed financial relationships with Teva Pharmaceuticals, Warner Chilcott, Boehringer Ingelheim, and Watson Pharmaceuticals. Dr. Plourd has been a speaker for Merck, Novartis, Sanofi-Aventis, Graceway, and Warner Chilcott.
Dr. Prager and Dr. Kaneshiro said they have no conflicts of interest.
SAN FRANCISCO — Fifty years after the introduction of oral contraceptives, physicians are looking to long-acting removable contraceptives to provide the next leap forward in preventing unintended pregnancy.
Three themes emerged in interviews with multiple physicians who gave separate presentations on contraception in sessions at the annual meeting of the American College of Obstetricians and Gynecologists.
First, nothing in the future of contraception is likely to match the revolutionary impact of oral contraception. Second, the great advances in contraception over the last half-decade still are accompanied by an unacceptably high rate of unintended pregnancies. And third, when asked to imagine what contraception might look like 50 years from now, most physicians looked to greater use of long-acting removable contraception (LARC) such as intrauterine devices (IUDs) or implants.
“The Pill is still one of the most important inventions ever, in terms of advancing not just women's health care but also women's rights and the ability for women to be in the workforce and to contribute in more ways that they want as members of society,” said Dr. Sarah Prager of the University of Washington, Seattle.
Long-acting removable contraception is the wave of the future, she said, because once the devices are inserted, they don't require patient participation in contraception, which has been one of the major stumbling blocks in contraceptive failures.
The next 50 years probably will see improvements in long-acting removable contraceptives, Dr. Prager added. “Our longest-acting LARC method only lasts for 10–12 years,” she noted.
Dr. Joseph Anthony Ogburn of the University of New Mexico, Albuquerque, shared the same vision for the future. “We did better than we did in the past, but we have great room for improvement,” he said. Approximately half of U.S. pregnancies are unintended, giving the United States the worst unintended pregnancy rate among developed countries.
Only about 1%–2% of U.S. women on contraception use an IUD, and less than 1% have a contraceptive implant. “If we could increase those numbers significantly, I think we could have a dramatic impact on the unintended pregnancy rate,” he said. Advances in the next 50 years probably will entail tweaking existing long-acting removable contraceptives to make them more acceptable.
But “I don't see anything as revolutionary as the Pill coming along any time in my lifetime,” he added.
Dr. Pouru Bhiwandi was more enthusiastic. “It's a very, very exciting time for all of contraception,” said Dr. Bhiwandi, an ob.gyn. in Raleigh, N.C., and an international consultant in women's health. “We have so many choices today, which we've never had before.”
Since the first oral contraceptive was approved in 1960, women's options grew with approval of more than 40 birth control pills and the development of other forms of hormonal contraception in transdermal patches, the vaginal ring, implants, and intrauterine devices (IUDs).
Modifications in dosing since the first oral contraceptives, which contained nearly four times the amount of estrogen and nearly 10 times the amount of progestin as today's formulations, have made the Pill safer and more acceptable, as have the development of newer estrogens and progesterones, she added. Newer regimens mean women no longer have to bleed while on hormonal contraception.
On the horizon are “exciting possibilities” for new products in barrier contraception that are both spermicidal and microbicidal to prevent sexually transmitted infections, plus “a whole range of new IUDs, a 1-year vaginal ring with a new progestin, and other products, Dr. Bhiwandi said.
The U.S. should emulate Europe, where longer-acting methods of birth control, including IUDs and contraceptives are much more prevalent, said Dr. Andrew Kaunitz, professor of ob.gyn. at the University of Florida, Jacksonville.
Although U.S. clinical trials report a failure rate of 1%–2% with oral contraceptive use, in “typical practice” it's much higher, Dr. Kaunitz said—around nine women per 100 couples annually have unintended pregnancies on the Pill. “That's too high,” he said.
Dr. Bliss Kaneshiro of the University of Hawaii, Honolulu, noted that long-acting contraceptive devices are expensive and not always covered by insurance. “One of the big challenges, I think, is cost,” she said. “Our challenge for the next 50 years is improving access to those good contraceptive methods.”
Dr. David Plourd of the Naval Medical Center, San Diego, said he hopes to see more options in long-acting removable contraceptives in the next 50 years, such as “a flexible IUD, not the semi-rigid T-shaped ones we have currently.”
The development of contraception methods targeted to men should play more of a significant role, he added.
Dr. Kaunitz disclosed financial relationships with Teva Pharmaceuticals, Bayer, Ortho (Johnson & Johnson), Merck, Procter & Gamble, Becton Dickinson, Sanofi-Aventis, and Medical Diagnostic Laboratories. Dr. Ogburn has been a consultant for Organon/Schering-Plough. Dr. Bhiwandi disclosed financial relationships with Teva Pharmaceuticals, Warner Chilcott, Boehringer Ingelheim, and Watson Pharmaceuticals. Dr. Plourd has been a speaker for Merck, Novartis, Sanofi-Aventis, Graceway, and Warner Chilcott.
Dr. Prager and Dr. Kaneshiro said they have no conflicts of interest.
SAN FRANCISCO — Fifty years after the introduction of oral contraceptives, physicians are looking to long-acting removable contraceptives to provide the next leap forward in preventing unintended pregnancy.
Three themes emerged in interviews with multiple physicians who gave separate presentations on contraception in sessions at the annual meeting of the American College of Obstetricians and Gynecologists.
First, nothing in the future of contraception is likely to match the revolutionary impact of oral contraception. Second, the great advances in contraception over the last half-decade still are accompanied by an unacceptably high rate of unintended pregnancies. And third, when asked to imagine what contraception might look like 50 years from now, most physicians looked to greater use of long-acting removable contraception (LARC) such as intrauterine devices (IUDs) or implants.
“The Pill is still one of the most important inventions ever, in terms of advancing not just women's health care but also women's rights and the ability for women to be in the workforce and to contribute in more ways that they want as members of society,” said Dr. Sarah Prager of the University of Washington, Seattle.
Long-acting removable contraception is the wave of the future, she said, because once the devices are inserted, they don't require patient participation in contraception, which has been one of the major stumbling blocks in contraceptive failures.
The next 50 years probably will see improvements in long-acting removable contraceptives, Dr. Prager added. “Our longest-acting LARC method only lasts for 10–12 years,” she noted.
Dr. Joseph Anthony Ogburn of the University of New Mexico, Albuquerque, shared the same vision for the future. “We did better than we did in the past, but we have great room for improvement,” he said. Approximately half of U.S. pregnancies are unintended, giving the United States the worst unintended pregnancy rate among developed countries.
Only about 1%–2% of U.S. women on contraception use an IUD, and less than 1% have a contraceptive implant. “If we could increase those numbers significantly, I think we could have a dramatic impact on the unintended pregnancy rate,” he said. Advances in the next 50 years probably will entail tweaking existing long-acting removable contraceptives to make them more acceptable.
But “I don't see anything as revolutionary as the Pill coming along any time in my lifetime,” he added.
Dr. Pouru Bhiwandi was more enthusiastic. “It's a very, very exciting time for all of contraception,” said Dr. Bhiwandi, an ob.gyn. in Raleigh, N.C., and an international consultant in women's health. “We have so many choices today, which we've never had before.”
Since the first oral contraceptive was approved in 1960, women's options grew with approval of more than 40 birth control pills and the development of other forms of hormonal contraception in transdermal patches, the vaginal ring, implants, and intrauterine devices (IUDs).
Modifications in dosing since the first oral contraceptives, which contained nearly four times the amount of estrogen and nearly 10 times the amount of progestin as today's formulations, have made the Pill safer and more acceptable, as have the development of newer estrogens and progesterones, she added. Newer regimens mean women no longer have to bleed while on hormonal contraception.
On the horizon are “exciting possibilities” for new products in barrier contraception that are both spermicidal and microbicidal to prevent sexually transmitted infections, plus “a whole range of new IUDs, a 1-year vaginal ring with a new progestin, and other products, Dr. Bhiwandi said.
The U.S. should emulate Europe, where longer-acting methods of birth control, including IUDs and contraceptives are much more prevalent, said Dr. Andrew Kaunitz, professor of ob.gyn. at the University of Florida, Jacksonville.
Although U.S. clinical trials report a failure rate of 1%–2% with oral contraceptive use, in “typical practice” it's much higher, Dr. Kaunitz said—around nine women per 100 couples annually have unintended pregnancies on the Pill. “That's too high,” he said.
Dr. Bliss Kaneshiro of the University of Hawaii, Honolulu, noted that long-acting contraceptive devices are expensive and not always covered by insurance. “One of the big challenges, I think, is cost,” she said. “Our challenge for the next 50 years is improving access to those good contraceptive methods.”
Dr. David Plourd of the Naval Medical Center, San Diego, said he hopes to see more options in long-acting removable contraceptives in the next 50 years, such as “a flexible IUD, not the semi-rigid T-shaped ones we have currently.”
The development of contraception methods targeted to men should play more of a significant role, he added.
Dr. Kaunitz disclosed financial relationships with Teva Pharmaceuticals, Bayer, Ortho (Johnson & Johnson), Merck, Procter & Gamble, Becton Dickinson, Sanofi-Aventis, and Medical Diagnostic Laboratories. Dr. Ogburn has been a consultant for Organon/Schering-Plough. Dr. Bhiwandi disclosed financial relationships with Teva Pharmaceuticals, Warner Chilcott, Boehringer Ingelheim, and Watson Pharmaceuticals. Dr. Plourd has been a speaker for Merck, Novartis, Sanofi-Aventis, Graceway, and Warner Chilcott.
Dr. Prager and Dr. Kaneshiro said they have no conflicts of interest.
ABD Retreat Seeks Consensus on Procedural Dermatology Exam
MONTEREY, Calif. — An American Board of Dermatology retreat on June 12 will attempt to reach peace between conflicting factions within the specialty on the issue of a certifying exam in procedural dermatology.
The gathering in Chicago will bring together leaders of various dermatologic associations who favor or oppose an idea first presented in 2008 by the American Board of Dermatology (ABD) to create a subspecialty certifying exam in procedural dermatology.
The proposal drew fire from the American Society for Mohs Surgery, the American Academy of Dermatology, and others who feared that eventually only fellowship-trained physicians would be eligible to take the exam and that insurers would refuse payments for procedures by noncertified physicians, thus restricting access to patient care.
The controversy prompted the ABD to slow consideration of the exam, and to schedule the upcoming retreat to obtain input from dermatologic organizations, Dr. Lee Portnoff, president of the American Society for Mohs Surgery (ASMS), said in an interview at the Society's annual meeting. The ASMS, which does not require fellowship training for membership, opposes the exam proposal. The American College of Mohs Surgery (ACMS), which requires members to have completed a 1- to 2-year fellowship, favors the proposal.
Under the ABD's original proposal, dermatologists who had completed a procedural dermatology fellowship would have the option of sitting for the certification exam and - for a limited time - physicians who had not completed a fellowship but who had significant surgical experience would be "grandfathered" in.
"I don't know if the current proposal differs significantly enough from last year's proposal that any of the dermatology organizations would support it, and I don't know how seriously the ABD would incorporate any comments at the retreat," Dr. Portnoff said.
"Our [ASMS's] position is that sufficient dermatologic surgery training should be included in all dermatology residencies so that certification by the ABD would assume competence in dermatologic surgery," he added.
"One of the large questions is, who will be deemed qualified to sit for the exam? That very question may be the dividing point, and is part of the main controversy," Dr. Matthew M. Goodman said at the meeting.
"We feel, in this Society [ASMS], that if you're appropriately trained - which can occur in residency or post residency - you can be competent to do Mohs surgery in a quality way for the best care of your patients without having done a fellowship or passed a certifying exam," added Dr. Goodman, the founding president of the Society and a Mohs surgeon in private practice in Santa Ana, Calif. "There's nothing against fellowships or against a certifying exam per se. We just would rather not see those as divisive forces."
Dr. Leonard M. Dzubow, president of the ACMS and a former director of the ABD, noted in an interview that procedural dermatology includes not just Mohs surgery but the entire spectrum of dermatologic surgery, so subspecialty certification in the field should be of interest to all dermatologists with advanced experience.
The ACMS's mission includes fostering excellence in treatment and in patient education about treatment options and different subspecialties. "The concept of certification will include several pathways for qualification, which will make it available to all physicians with sufficient experience and knowledge," said Dr. Dzubow, professor of dermatology at the University of Pennsylvania, Philadelphia.
Dr. Randall K. Roenigk, a past president of the ABD, said in an interview that the impetus for the exam was to give residents and fellows who complete training programs accredited by the Accreditation Council for Graduate Medical Education the option of being board certified.
"Just like all 24 core specialties and 117 subspecialties associated with the American Board of Medical Specialties, the mission of the ABD is to assure the public that physicians who hold themselves out as specialists are competent in a defined body of knowledge," said Dr. Roenigk, chairman of the department of dermatology at the Mayo Clinic in Rochester, Minn.
Dr. Elaine C. Siegfried, current president of the ABD and professor of pediatrics and dermatology at St. Louis University, said the ABD would have no comment on the topic until after the retreat.
If the ABD creates the certifying exam, procedural dermatology would join dermatopathology, pediatric dermatology, and clinical and laboratory dermatologic immunology as board-certified subspecialties.
MONTEREY, Calif. — An American Board of Dermatology retreat on June 12 will attempt to reach peace between conflicting factions within the specialty on the issue of a certifying exam in procedural dermatology.
The gathering in Chicago will bring together leaders of various dermatologic associations who favor or oppose an idea first presented in 2008 by the American Board of Dermatology (ABD) to create a subspecialty certifying exam in procedural dermatology.
The proposal drew fire from the American Society for Mohs Surgery, the American Academy of Dermatology, and others who feared that eventually only fellowship-trained physicians would be eligible to take the exam and that insurers would refuse payments for procedures by noncertified physicians, thus restricting access to patient care.
The controversy prompted the ABD to slow consideration of the exam, and to schedule the upcoming retreat to obtain input from dermatologic organizations, Dr. Lee Portnoff, president of the American Society for Mohs Surgery (ASMS), said in an interview at the Society's annual meeting. The ASMS, which does not require fellowship training for membership, opposes the exam proposal. The American College of Mohs Surgery (ACMS), which requires members to have completed a 1- to 2-year fellowship, favors the proposal.
Under the ABD's original proposal, dermatologists who had completed a procedural dermatology fellowship would have the option of sitting for the certification exam and - for a limited time - physicians who had not completed a fellowship but who had significant surgical experience would be "grandfathered" in.
"I don't know if the current proposal differs significantly enough from last year's proposal that any of the dermatology organizations would support it, and I don't know how seriously the ABD would incorporate any comments at the retreat," Dr. Portnoff said.
"Our [ASMS's] position is that sufficient dermatologic surgery training should be included in all dermatology residencies so that certification by the ABD would assume competence in dermatologic surgery," he added.
"One of the large questions is, who will be deemed qualified to sit for the exam? That very question may be the dividing point, and is part of the main controversy," Dr. Matthew M. Goodman said at the meeting.
"We feel, in this Society [ASMS], that if you're appropriately trained - which can occur in residency or post residency - you can be competent to do Mohs surgery in a quality way for the best care of your patients without having done a fellowship or passed a certifying exam," added Dr. Goodman, the founding president of the Society and a Mohs surgeon in private practice in Santa Ana, Calif. "There's nothing against fellowships or against a certifying exam per se. We just would rather not see those as divisive forces."
Dr. Leonard M. Dzubow, president of the ACMS and a former director of the ABD, noted in an interview that procedural dermatology includes not just Mohs surgery but the entire spectrum of dermatologic surgery, so subspecialty certification in the field should be of interest to all dermatologists with advanced experience.
The ACMS's mission includes fostering excellence in treatment and in patient education about treatment options and different subspecialties. "The concept of certification will include several pathways for qualification, which will make it available to all physicians with sufficient experience and knowledge," said Dr. Dzubow, professor of dermatology at the University of Pennsylvania, Philadelphia.
Dr. Randall K. Roenigk, a past president of the ABD, said in an interview that the impetus for the exam was to give residents and fellows who complete training programs accredited by the Accreditation Council for Graduate Medical Education the option of being board certified.
"Just like all 24 core specialties and 117 subspecialties associated with the American Board of Medical Specialties, the mission of the ABD is to assure the public that physicians who hold themselves out as specialists are competent in a defined body of knowledge," said Dr. Roenigk, chairman of the department of dermatology at the Mayo Clinic in Rochester, Minn.
Dr. Elaine C. Siegfried, current president of the ABD and professor of pediatrics and dermatology at St. Louis University, said the ABD would have no comment on the topic until after the retreat.
If the ABD creates the certifying exam, procedural dermatology would join dermatopathology, pediatric dermatology, and clinical and laboratory dermatologic immunology as board-certified subspecialties.
MONTEREY, Calif. — An American Board of Dermatology retreat on June 12 will attempt to reach peace between conflicting factions within the specialty on the issue of a certifying exam in procedural dermatology.
The gathering in Chicago will bring together leaders of various dermatologic associations who favor or oppose an idea first presented in 2008 by the American Board of Dermatology (ABD) to create a subspecialty certifying exam in procedural dermatology.
The proposal drew fire from the American Society for Mohs Surgery, the American Academy of Dermatology, and others who feared that eventually only fellowship-trained physicians would be eligible to take the exam and that insurers would refuse payments for procedures by noncertified physicians, thus restricting access to patient care.
The controversy prompted the ABD to slow consideration of the exam, and to schedule the upcoming retreat to obtain input from dermatologic organizations, Dr. Lee Portnoff, president of the American Society for Mohs Surgery (ASMS), said in an interview at the Society's annual meeting. The ASMS, which does not require fellowship training for membership, opposes the exam proposal. The American College of Mohs Surgery (ACMS), which requires members to have completed a 1- to 2-year fellowship, favors the proposal.
Under the ABD's original proposal, dermatologists who had completed a procedural dermatology fellowship would have the option of sitting for the certification exam and - for a limited time - physicians who had not completed a fellowship but who had significant surgical experience would be "grandfathered" in.
"I don't know if the current proposal differs significantly enough from last year's proposal that any of the dermatology organizations would support it, and I don't know how seriously the ABD would incorporate any comments at the retreat," Dr. Portnoff said.
"Our [ASMS's] position is that sufficient dermatologic surgery training should be included in all dermatology residencies so that certification by the ABD would assume competence in dermatologic surgery," he added.
"One of the large questions is, who will be deemed qualified to sit for the exam? That very question may be the dividing point, and is part of the main controversy," Dr. Matthew M. Goodman said at the meeting.
"We feel, in this Society [ASMS], that if you're appropriately trained - which can occur in residency or post residency - you can be competent to do Mohs surgery in a quality way for the best care of your patients without having done a fellowship or passed a certifying exam," added Dr. Goodman, the founding president of the Society and a Mohs surgeon in private practice in Santa Ana, Calif. "There's nothing against fellowships or against a certifying exam per se. We just would rather not see those as divisive forces."
Dr. Leonard M. Dzubow, president of the ACMS and a former director of the ABD, noted in an interview that procedural dermatology includes not just Mohs surgery but the entire spectrum of dermatologic surgery, so subspecialty certification in the field should be of interest to all dermatologists with advanced experience.
The ACMS's mission includes fostering excellence in treatment and in patient education about treatment options and different subspecialties. "The concept of certification will include several pathways for qualification, which will make it available to all physicians with sufficient experience and knowledge," said Dr. Dzubow, professor of dermatology at the University of Pennsylvania, Philadelphia.
Dr. Randall K. Roenigk, a past president of the ABD, said in an interview that the impetus for the exam was to give residents and fellows who complete training programs accredited by the Accreditation Council for Graduate Medical Education the option of being board certified.
"Just like all 24 core specialties and 117 subspecialties associated with the American Board of Medical Specialties, the mission of the ABD is to assure the public that physicians who hold themselves out as specialists are competent in a defined body of knowledge," said Dr. Roenigk, chairman of the department of dermatology at the Mayo Clinic in Rochester, Minn.
Dr. Elaine C. Siegfried, current president of the ABD and professor of pediatrics and dermatology at St. Louis University, said the ABD would have no comment on the topic until after the retreat.
If the ABD creates the certifying exam, procedural dermatology would join dermatopathology, pediatric dermatology, and clinical and laboratory dermatologic immunology as board-certified subspecialties.
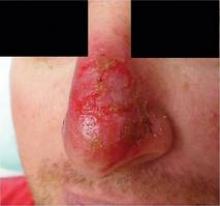
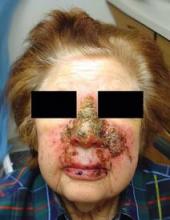
Imiquimod Before Mohs Is No Help for Nodular, Nasal BCC
MONTEREY, Calif. — Using imiquimod on nodular basal cell carcinomas on the nose before Mohs surgery failed to simplify the surgery or reduce costs and significantly increased local adverse reactions, in a randomized, controlled study of 28 patients.
"Doctors across the country are using imiquimod 'off label' to treat BCCs on the nose, and they should know these findings before doing that," said Dr. David F. Butler.
The study was inspired by a patient who refused surgery or radiation for her nodular, nasal BCC and was treated with good results using imiquimod, he said at the annual meeting of the American Society for Mohs Surgery. Perhaps, he reasoned, adjunctive imiquimod might reduce similar tumors before treatment with Mohs surgery.
"I was surprised that imiquimod did not reduce the number of stages, reduce the cost of Mohs surgery, or reduce the cost of repair," said Dr. Butler, chair of dermatology at the Scott and White Clinic and professor of internal medicine at Texas A&M University, both in Temple, Tex.
Of the 31 patients who enrolled in the study, 3 dropped out, 2 because of local adverse events and 1 because of other illness. Among the 28 patients who completed the study, 10 of 12 (83%) in the imiquimod arm and 4 of 16 (25%) in the control arm developed local adverse events after 3 weeks of treatment, including redness, blisters, erosions, and crusting. After 6 weeks, the same number in the imiquimod group and two patients in the control group (12%) had local adverse reactions. The differences between the groups were statistically significant.
The frequency of local adverse events limits the usefulness of imiquimod as adjunctive therapy in these cases, he said.
Only one stage of Mohs surgery was needed for 11 patients in each group. Two stages were needed for one patient in the imiquimod arm (8%) and five patients in the control arm (31%). Surgical defect sizes averaged 88 mm2 in the imiquimod arm and 100 mm2 in the control arm (Dermatol. Surg. 2009;35:24-9).
A larger study might show a difference, he acknowledged. The current study did not include the cost of imiquimod, so even if a larger study finds that adjunctive imiquimod therapy reduces the number of Mohs stages needed, the medication cost might negate any cost savings in the surgery.
Patients applied 5% imiquimod cream or vehicle to the tumor five nights a week for 6 weeks and covered it with a bandage supplied by investigators. One month after stopping the imiquimod, they underwent Mohs surgery.
Dr. Butler said in an interview that he was surprised by the "inordinately high" proportion of patients with tumor remaining at the time of Mohs surgery after imiquimod treatment.
Only 5 of 12 patients (42%) in the treatment group had complete clearance of the tumor after imiquimod therapy, with tumor absent (presumably destroyed) in the first stage block. "That's a relatively low number when you compare it to the 80% cure rate that you get when using imiquimod to treat superficial BCCs on the trunk or extremities," he said. "My concern is that nodular BCCs on the nose may be a different problem."
Imiquimod was approved in 2004 to treat superficial BCC on the trunk and extremities, with histologic cure rates of 79%-82% at those sites, previous studies have shown. Clearance rates have been lower, however, for nodular BCCs, with reports ranging from 65% to 76%, he noted. In addition, the most common site for BCCs is not the trunk or extremities but the nose, accounting for 25%-30%.
Dr. Butler advised against using imiquimod as a stand-alone therapy for nodular BCCs on the nose, but said it may be a reasonable option for patients who cannot or will not undergo other treatments.
"We do recommend, however, that if you're going to use imiquimod as a single therapy for nodular BCCs on the nose, that once you finish the treatment, that you go back and do a little biopsy of the area to document that the cancer is gone," he added.
Graceway Pharmaceuticals and 3M funded the study. Dr. Butler reported that he had no conflicts of interest.
MONTEREY, Calif. — Using imiquimod on nodular basal cell carcinomas on the nose before Mohs surgery failed to simplify the surgery or reduce costs and significantly increased local adverse reactions, in a randomized, controlled study of 28 patients.
"Doctors across the country are using imiquimod 'off label' to treat BCCs on the nose, and they should know these findings before doing that," said Dr. David F. Butler.
The study was inspired by a patient who refused surgery or radiation for her nodular, nasal BCC and was treated with good results using imiquimod, he said at the annual meeting of the American Society for Mohs Surgery. Perhaps, he reasoned, adjunctive imiquimod might reduce similar tumors before treatment with Mohs surgery.
"I was surprised that imiquimod did not reduce the number of stages, reduce the cost of Mohs surgery, or reduce the cost of repair," said Dr. Butler, chair of dermatology at the Scott and White Clinic and professor of internal medicine at Texas A&M University, both in Temple, Tex.
Of the 31 patients who enrolled in the study, 3 dropped out, 2 because of local adverse events and 1 because of other illness. Among the 28 patients who completed the study, 10 of 12 (83%) in the imiquimod arm and 4 of 16 (25%) in the control arm developed local adverse events after 3 weeks of treatment, including redness, blisters, erosions, and crusting. After 6 weeks, the same number in the imiquimod group and two patients in the control group (12%) had local adverse reactions. The differences between the groups were statistically significant.
The frequency of local adverse events limits the usefulness of imiquimod as adjunctive therapy in these cases, he said.
Only one stage of Mohs surgery was needed for 11 patients in each group. Two stages were needed for one patient in the imiquimod arm (8%) and five patients in the control arm (31%). Surgical defect sizes averaged 88 mm2 in the imiquimod arm and 100 mm2 in the control arm (Dermatol. Surg. 2009;35:24-9).
A larger study might show a difference, he acknowledged. The current study did not include the cost of imiquimod, so even if a larger study finds that adjunctive imiquimod therapy reduces the number of Mohs stages needed, the medication cost might negate any cost savings in the surgery.
Patients applied 5% imiquimod cream or vehicle to the tumor five nights a week for 6 weeks and covered it with a bandage supplied by investigators. One month after stopping the imiquimod, they underwent Mohs surgery.
Dr. Butler said in an interview that he was surprised by the "inordinately high" proportion of patients with tumor remaining at the time of Mohs surgery after imiquimod treatment.
Only 5 of 12 patients (42%) in the treatment group had complete clearance of the tumor after imiquimod therapy, with tumor absent (presumably destroyed) in the first stage block. "That's a relatively low number when you compare it to the 80% cure rate that you get when using imiquimod to treat superficial BCCs on the trunk or extremities," he said. "My concern is that nodular BCCs on the nose may be a different problem."
Imiquimod was approved in 2004 to treat superficial BCC on the trunk and extremities, with histologic cure rates of 79%-82% at those sites, previous studies have shown. Clearance rates have been lower, however, for nodular BCCs, with reports ranging from 65% to 76%, he noted. In addition, the most common site for BCCs is not the trunk or extremities but the nose, accounting for 25%-30%.
Dr. Butler advised against using imiquimod as a stand-alone therapy for nodular BCCs on the nose, but said it may be a reasonable option for patients who cannot or will not undergo other treatments.
"We do recommend, however, that if you're going to use imiquimod as a single therapy for nodular BCCs on the nose, that once you finish the treatment, that you go back and do a little biopsy of the area to document that the cancer is gone," he added.
Graceway Pharmaceuticals and 3M funded the study. Dr. Butler reported that he had no conflicts of interest.
MONTEREY, Calif. — Using imiquimod on nodular basal cell carcinomas on the nose before Mohs surgery failed to simplify the surgery or reduce costs and significantly increased local adverse reactions, in a randomized, controlled study of 28 patients.
"Doctors across the country are using imiquimod 'off label' to treat BCCs on the nose, and they should know these findings before doing that," said Dr. David F. Butler.
The study was inspired by a patient who refused surgery or radiation for her nodular, nasal BCC and was treated with good results using imiquimod, he said at the annual meeting of the American Society for Mohs Surgery. Perhaps, he reasoned, adjunctive imiquimod might reduce similar tumors before treatment with Mohs surgery.
"I was surprised that imiquimod did not reduce the number of stages, reduce the cost of Mohs surgery, or reduce the cost of repair," said Dr. Butler, chair of dermatology at the Scott and White Clinic and professor of internal medicine at Texas A&M University, both in Temple, Tex.
Of the 31 patients who enrolled in the study, 3 dropped out, 2 because of local adverse events and 1 because of other illness. Among the 28 patients who completed the study, 10 of 12 (83%) in the imiquimod arm and 4 of 16 (25%) in the control arm developed local adverse events after 3 weeks of treatment, including redness, blisters, erosions, and crusting. After 6 weeks, the same number in the imiquimod group and two patients in the control group (12%) had local adverse reactions. The differences between the groups were statistically significant.
The frequency of local adverse events limits the usefulness of imiquimod as adjunctive therapy in these cases, he said.
Only one stage of Mohs surgery was needed for 11 patients in each group. Two stages were needed for one patient in the imiquimod arm (8%) and five patients in the control arm (31%). Surgical defect sizes averaged 88 mm2 in the imiquimod arm and 100 mm2 in the control arm (Dermatol. Surg. 2009;35:24-9).
A larger study might show a difference, he acknowledged. The current study did not include the cost of imiquimod, so even if a larger study finds that adjunctive imiquimod therapy reduces the number of Mohs stages needed, the medication cost might negate any cost savings in the surgery.
Patients applied 5% imiquimod cream or vehicle to the tumor five nights a week for 6 weeks and covered it with a bandage supplied by investigators. One month after stopping the imiquimod, they underwent Mohs surgery.
Dr. Butler said in an interview that he was surprised by the "inordinately high" proportion of patients with tumor remaining at the time of Mohs surgery after imiquimod treatment.
Only 5 of 12 patients (42%) in the treatment group had complete clearance of the tumor after imiquimod therapy, with tumor absent (presumably destroyed) in the first stage block. "That's a relatively low number when you compare it to the 80% cure rate that you get when using imiquimod to treat superficial BCCs on the trunk or extremities," he said. "My concern is that nodular BCCs on the nose may be a different problem."
Imiquimod was approved in 2004 to treat superficial BCC on the trunk and extremities, with histologic cure rates of 79%-82% at those sites, previous studies have shown. Clearance rates have been lower, however, for nodular BCCs, with reports ranging from 65% to 76%, he noted. In addition, the most common site for BCCs is not the trunk or extremities but the nose, accounting for 25%-30%.
Dr. Butler advised against using imiquimod as a stand-alone therapy for nodular BCCs on the nose, but said it may be a reasonable option for patients who cannot or will not undergo other treatments.
"We do recommend, however, that if you're going to use imiquimod as a single therapy for nodular BCCs on the nose, that once you finish the treatment, that you go back and do a little biopsy of the area to document that the cancer is gone," he added.
Graceway Pharmaceuticals and 3M funded the study. Dr. Butler reported that he had no conflicts of interest.
Monitoring Helps Addicted Anesthesiologists on Naltrexone
SAN FRANCISCO – A monitoring program for physicians with substance use disorders greatly boosted the chances of anesthesiologists staying clean and returning to work after the program mandated 2 years of naltrexone therapy in addition to usual treatment, preliminary data suggest.
The study tracked 18 anesthesiologists and 4 anesthesiology residents with opiate abuse or dependence who entered the Florida Medical Association's Professionals Resource Network, the state-sanctioned “physician health program” that monitors impaired physicians. Half the cohort entered monitoring just before (and half after) a 2005 rule change requiring 2 years of naltrexone therapy in addition to usual program requirements.
In the group without naltrexone, 8 of the 11 physicians relapsed on opiates. Of the three who did not relapse, one left anesthesiology to become a consultant, one switched practice to pain medicine, and one successfully returned to anesthesiology.
In the group of 11 on naltrexone, only 1 anesthesiologist relapsed on opiates and another relapsed on nitrous oxide; 9 of 11 successfully returned to practice, Lisa J. Merlo, Ph.D., and her associates reported. The results with naltrexone might be more impressive, considering that 5 of the 11 physicians in the naltrexone group had a history of relapse on opiates or other drugs prior to starting naltrexone, added Dr. Merlo of the University of Florida, Gainesville.
These preliminary data suggest that adding naltrexone increased the chances of avoiding relapse ninefold and improved the chance of returning to work 11-fold.
Naltrexone is an opioid receptor antagonist used mainly in the management of alcohol or opioid dependence. If further research supports the small study's findings, naltrexone pharmacotherapy might be a useful addition to comprehensive treatment and monitoring contracts, but its potential advantages should be weighed against potential side effects when considering using naltrexone in specialists other than anesthesiologists, said Dr. Merlo, who received the society's 2010 Young Investigator Award for her study.
Naltrexone was taken orally as 50 mg 5 days per week or on 3 days per week in doses of 100 mg, 100 mg, and 150 mg, or by monthly injections. Ingestion was witnessed, with random urine testing to confirm the presence of naltrexone in urine.
Its use was added to the program's usual requirements for 2–5 years of monitoring for substance abuse or dependence, random urine drug screens, and attendance in monitoring groups and recovery programs.
The investigators said they have no pertinent conflicts of interest. The National Institute on Drug Abuse and the Florida Medical Association funded the study.
SAN FRANCISCO – A monitoring program for physicians with substance use disorders greatly boosted the chances of anesthesiologists staying clean and returning to work after the program mandated 2 years of naltrexone therapy in addition to usual treatment, preliminary data suggest.
The study tracked 18 anesthesiologists and 4 anesthesiology residents with opiate abuse or dependence who entered the Florida Medical Association's Professionals Resource Network, the state-sanctioned “physician health program” that monitors impaired physicians. Half the cohort entered monitoring just before (and half after) a 2005 rule change requiring 2 years of naltrexone therapy in addition to usual program requirements.
In the group without naltrexone, 8 of the 11 physicians relapsed on opiates. Of the three who did not relapse, one left anesthesiology to become a consultant, one switched practice to pain medicine, and one successfully returned to anesthesiology.
In the group of 11 on naltrexone, only 1 anesthesiologist relapsed on opiates and another relapsed on nitrous oxide; 9 of 11 successfully returned to practice, Lisa J. Merlo, Ph.D., and her associates reported. The results with naltrexone might be more impressive, considering that 5 of the 11 physicians in the naltrexone group had a history of relapse on opiates or other drugs prior to starting naltrexone, added Dr. Merlo of the University of Florida, Gainesville.
These preliminary data suggest that adding naltrexone increased the chances of avoiding relapse ninefold and improved the chance of returning to work 11-fold.
Naltrexone is an opioid receptor antagonist used mainly in the management of alcohol or opioid dependence. If further research supports the small study's findings, naltrexone pharmacotherapy might be a useful addition to comprehensive treatment and monitoring contracts, but its potential advantages should be weighed against potential side effects when considering using naltrexone in specialists other than anesthesiologists, said Dr. Merlo, who received the society's 2010 Young Investigator Award for her study.
Naltrexone was taken orally as 50 mg 5 days per week or on 3 days per week in doses of 100 mg, 100 mg, and 150 mg, or by monthly injections. Ingestion was witnessed, with random urine testing to confirm the presence of naltrexone in urine.
Its use was added to the program's usual requirements for 2–5 years of monitoring for substance abuse or dependence, random urine drug screens, and attendance in monitoring groups and recovery programs.
The investigators said they have no pertinent conflicts of interest. The National Institute on Drug Abuse and the Florida Medical Association funded the study.
SAN FRANCISCO – A monitoring program for physicians with substance use disorders greatly boosted the chances of anesthesiologists staying clean and returning to work after the program mandated 2 years of naltrexone therapy in addition to usual treatment, preliminary data suggest.
The study tracked 18 anesthesiologists and 4 anesthesiology residents with opiate abuse or dependence who entered the Florida Medical Association's Professionals Resource Network, the state-sanctioned “physician health program” that monitors impaired physicians. Half the cohort entered monitoring just before (and half after) a 2005 rule change requiring 2 years of naltrexone therapy in addition to usual program requirements.
In the group without naltrexone, 8 of the 11 physicians relapsed on opiates. Of the three who did not relapse, one left anesthesiology to become a consultant, one switched practice to pain medicine, and one successfully returned to anesthesiology.
In the group of 11 on naltrexone, only 1 anesthesiologist relapsed on opiates and another relapsed on nitrous oxide; 9 of 11 successfully returned to practice, Lisa J. Merlo, Ph.D., and her associates reported. The results with naltrexone might be more impressive, considering that 5 of the 11 physicians in the naltrexone group had a history of relapse on opiates or other drugs prior to starting naltrexone, added Dr. Merlo of the University of Florida, Gainesville.
These preliminary data suggest that adding naltrexone increased the chances of avoiding relapse ninefold and improved the chance of returning to work 11-fold.
Naltrexone is an opioid receptor antagonist used mainly in the management of alcohol or opioid dependence. If further research supports the small study's findings, naltrexone pharmacotherapy might be a useful addition to comprehensive treatment and monitoring contracts, but its potential advantages should be weighed against potential side effects when considering using naltrexone in specialists other than anesthesiologists, said Dr. Merlo, who received the society's 2010 Young Investigator Award for her study.
Naltrexone was taken orally as 50 mg 5 days per week or on 3 days per week in doses of 100 mg, 100 mg, and 150 mg, or by monthly injections. Ingestion was witnessed, with random urine testing to confirm the presence of naltrexone in urine.
Its use was added to the program's usual requirements for 2–5 years of monitoring for substance abuse or dependence, random urine drug screens, and attendance in monitoring groups and recovery programs.
The investigators said they have no pertinent conflicts of interest. The National Institute on Drug Abuse and the Florida Medical Association funded the study.
Tailor Therapy for GLBT Substance Abusers
SAN FRANCISCO – To help sexual minorities with addiction, physicians need to consider factors ranging from details in their waiting rooms to the attitudes of their patients and themselves.
Not all treatment providers are comfortable with patients' sexual diversity (see box). Physicians who are uncomfortable treating GLBT patients and other sexual minorities either can obtain education and experience to desensitize themselves, overcome prejudices and become more accepting, or they can refer these patients to other providers who might better meet the patients' needs, Dr. Penelope P. Ziegler suggested.
Those who are comfortable treating sexual minorities still have extra work to do. They can ensure their offices or treatment programs let patients know it's “okay” to be a sexual minority by including art work or literature relevant to sexual minority cultures and by designing intake forms so that same-sex relationships or diverse sexual identities can be reported.
When treating sexual minorities, it's essential to know the developmental stages of “coming out,” said Dr. Ziegler, medical director of Virginia Health Practitioners Intervention Program, Richmond.
Patients can start addiction treatment at any of the six stages of self-discovery of sexual orientation. The stages start with an individual's confusion about sexual identity to comparison (accepting the possibility of being gay, lesbian, and so on), followed by tolerance of a sexual identity, then acceptance, pride, and finally synthesis of the identity into the patient's life.
Similarly, there's a continuum of sensitivity to sexual minorities in addiction treatment programs, she added. Some are actively antigay, especially some faith-based programs, whereas others are traditional heterosexist programs. Then there are the GLBT-naive programs that assume everyone there is heterosexual, which sends a message that no one should be queer. Better programs are tolerant, and then sensitive, and finally affirming of GLBT patients, Dr. Ziegler said.
“Everyone in the addiction field needs to have some training to overcome heterosexism, shock, and revulsion toward sexual minorities,” she believes. It's important to reinforce boundaries for practitioners and patients so that value judgments aren't verbalized and attempts to direct behavioral changes are based on patient need, not the practitioner's values.
Residential treatment programs should examine any heterosexist rules dealing with roommates and address any homophobia being acted out in the community, she suggested. In group therapy, the treatment leader should know whether a sexual minority patient is “out” to the other members, and know how to interrupt heterosexist or homophobic behavior.
In individual therapy, besides addressing the patient's stage of self-awareness of sexual identity as it relates to addiction recovery, the provider should consider specific risks or triggers for relapse that might be related to the patient's sexual identity. Social settings such as clubs and bars or sexual practices that commonly involve drug use increase the risk of relapse.
In 12-step recovery groups, facilitators need to put extra thought into whether a sexual minority individual may do better with a sponsor of the same or opposite sex, gay or straight, and whether some gay 12-step meetings may or may not be better than “regular” meetings for some.
Support groups or treatment programs designed specifically for sexual minorities have advantages and disadvantages, and aren't available everywhere, but mainstream programs can take steps to enhance the treatment experience for GLBT patients, Dr. Ziegler said.
Try adding a “special interest” group with regular group therapy sessions, she suggested. Develop contacts in the GLBT community to find people in addiction recovery to act as volunteer sponsors. Provide in-service training to all staff so that all can help GLBT patients. “It is not cool to have a designated queer on your staff” to be the only one doing this work, commented Dr. Ziegler, who said she has no pertinent conflicts of interest.
All staff should be trained to help GLBT patients. 'It is not cool to have a designated queer on your staff' to do this work.
Source DR. ZIEGLER
An Acronym for Sexual Minorities
The ingredients of the sexual minorities “alphabet soup”–GLBTQQAAi2S–include the following:
Gay: Males attracted to males.
Lesbian: Females attracted to females.
Bisexual: People attracted to both sexes.
Transgender: People whose gender identity does not match their birth sex.
Queer: An all-encompassing term for orientations other that heterosexual.
Questioning: Individuals who are exploring sexual orientation or identity.
Asexual: Not sexual.
Allies: Important to sexual minorities.
intersex: People born with ambiguous genitalia. (The intersex community chose the lower-case “i.”)
2-Spirit people: Native American or indigenous Canadian term for gays and lesbians.
SAN FRANCISCO – To help sexual minorities with addiction, physicians need to consider factors ranging from details in their waiting rooms to the attitudes of their patients and themselves.
Not all treatment providers are comfortable with patients' sexual diversity (see box). Physicians who are uncomfortable treating GLBT patients and other sexual minorities either can obtain education and experience to desensitize themselves, overcome prejudices and become more accepting, or they can refer these patients to other providers who might better meet the patients' needs, Dr. Penelope P. Ziegler suggested.
Those who are comfortable treating sexual minorities still have extra work to do. They can ensure their offices or treatment programs let patients know it's “okay” to be a sexual minority by including art work or literature relevant to sexual minority cultures and by designing intake forms so that same-sex relationships or diverse sexual identities can be reported.
When treating sexual minorities, it's essential to know the developmental stages of “coming out,” said Dr. Ziegler, medical director of Virginia Health Practitioners Intervention Program, Richmond.
Patients can start addiction treatment at any of the six stages of self-discovery of sexual orientation. The stages start with an individual's confusion about sexual identity to comparison (accepting the possibility of being gay, lesbian, and so on), followed by tolerance of a sexual identity, then acceptance, pride, and finally synthesis of the identity into the patient's life.
Similarly, there's a continuum of sensitivity to sexual minorities in addiction treatment programs, she added. Some are actively antigay, especially some faith-based programs, whereas others are traditional heterosexist programs. Then there are the GLBT-naive programs that assume everyone there is heterosexual, which sends a message that no one should be queer. Better programs are tolerant, and then sensitive, and finally affirming of GLBT patients, Dr. Ziegler said.
“Everyone in the addiction field needs to have some training to overcome heterosexism, shock, and revulsion toward sexual minorities,” she believes. It's important to reinforce boundaries for practitioners and patients so that value judgments aren't verbalized and attempts to direct behavioral changes are based on patient need, not the practitioner's values.
Residential treatment programs should examine any heterosexist rules dealing with roommates and address any homophobia being acted out in the community, she suggested. In group therapy, the treatment leader should know whether a sexual minority patient is “out” to the other members, and know how to interrupt heterosexist or homophobic behavior.
In individual therapy, besides addressing the patient's stage of self-awareness of sexual identity as it relates to addiction recovery, the provider should consider specific risks or triggers for relapse that might be related to the patient's sexual identity. Social settings such as clubs and bars or sexual practices that commonly involve drug use increase the risk of relapse.
In 12-step recovery groups, facilitators need to put extra thought into whether a sexual minority individual may do better with a sponsor of the same or opposite sex, gay or straight, and whether some gay 12-step meetings may or may not be better than “regular” meetings for some.
Support groups or treatment programs designed specifically for sexual minorities have advantages and disadvantages, and aren't available everywhere, but mainstream programs can take steps to enhance the treatment experience for GLBT patients, Dr. Ziegler said.
Try adding a “special interest” group with regular group therapy sessions, she suggested. Develop contacts in the GLBT community to find people in addiction recovery to act as volunteer sponsors. Provide in-service training to all staff so that all can help GLBT patients. “It is not cool to have a designated queer on your staff” to be the only one doing this work, commented Dr. Ziegler, who said she has no pertinent conflicts of interest.
All staff should be trained to help GLBT patients. 'It is not cool to have a designated queer on your staff' to do this work.
Source DR. ZIEGLER
An Acronym for Sexual Minorities
The ingredients of the sexual minorities “alphabet soup”–GLBTQQAAi2S–include the following:
Gay: Males attracted to males.
Lesbian: Females attracted to females.
Bisexual: People attracted to both sexes.
Transgender: People whose gender identity does not match their birth sex.
Queer: An all-encompassing term for orientations other that heterosexual.
Questioning: Individuals who are exploring sexual orientation or identity.
Asexual: Not sexual.
Allies: Important to sexual minorities.
intersex: People born with ambiguous genitalia. (The intersex community chose the lower-case “i.”)
2-Spirit people: Native American or indigenous Canadian term for gays and lesbians.
SAN FRANCISCO – To help sexual minorities with addiction, physicians need to consider factors ranging from details in their waiting rooms to the attitudes of their patients and themselves.
Not all treatment providers are comfortable with patients' sexual diversity (see box). Physicians who are uncomfortable treating GLBT patients and other sexual minorities either can obtain education and experience to desensitize themselves, overcome prejudices and become more accepting, or they can refer these patients to other providers who might better meet the patients' needs, Dr. Penelope P. Ziegler suggested.
Those who are comfortable treating sexual minorities still have extra work to do. They can ensure their offices or treatment programs let patients know it's “okay” to be a sexual minority by including art work or literature relevant to sexual minority cultures and by designing intake forms so that same-sex relationships or diverse sexual identities can be reported.
When treating sexual minorities, it's essential to know the developmental stages of “coming out,” said Dr. Ziegler, medical director of Virginia Health Practitioners Intervention Program, Richmond.
Patients can start addiction treatment at any of the six stages of self-discovery of sexual orientation. The stages start with an individual's confusion about sexual identity to comparison (accepting the possibility of being gay, lesbian, and so on), followed by tolerance of a sexual identity, then acceptance, pride, and finally synthesis of the identity into the patient's life.
Similarly, there's a continuum of sensitivity to sexual minorities in addiction treatment programs, she added. Some are actively antigay, especially some faith-based programs, whereas others are traditional heterosexist programs. Then there are the GLBT-naive programs that assume everyone there is heterosexual, which sends a message that no one should be queer. Better programs are tolerant, and then sensitive, and finally affirming of GLBT patients, Dr. Ziegler said.
“Everyone in the addiction field needs to have some training to overcome heterosexism, shock, and revulsion toward sexual minorities,” she believes. It's important to reinforce boundaries for practitioners and patients so that value judgments aren't verbalized and attempts to direct behavioral changes are based on patient need, not the practitioner's values.
Residential treatment programs should examine any heterosexist rules dealing with roommates and address any homophobia being acted out in the community, she suggested. In group therapy, the treatment leader should know whether a sexual minority patient is “out” to the other members, and know how to interrupt heterosexist or homophobic behavior.
In individual therapy, besides addressing the patient's stage of self-awareness of sexual identity as it relates to addiction recovery, the provider should consider specific risks or triggers for relapse that might be related to the patient's sexual identity. Social settings such as clubs and bars or sexual practices that commonly involve drug use increase the risk of relapse.
In 12-step recovery groups, facilitators need to put extra thought into whether a sexual minority individual may do better with a sponsor of the same or opposite sex, gay or straight, and whether some gay 12-step meetings may or may not be better than “regular” meetings for some.
Support groups or treatment programs designed specifically for sexual minorities have advantages and disadvantages, and aren't available everywhere, but mainstream programs can take steps to enhance the treatment experience for GLBT patients, Dr. Ziegler said.
Try adding a “special interest” group with regular group therapy sessions, she suggested. Develop contacts in the GLBT community to find people in addiction recovery to act as volunteer sponsors. Provide in-service training to all staff so that all can help GLBT patients. “It is not cool to have a designated queer on your staff” to be the only one doing this work, commented Dr. Ziegler, who said she has no pertinent conflicts of interest.
All staff should be trained to help GLBT patients. 'It is not cool to have a designated queer on your staff' to do this work.
Source DR. ZIEGLER
An Acronym for Sexual Minorities
The ingredients of the sexual minorities “alphabet soup”–GLBTQQAAi2S–include the following:
Gay: Males attracted to males.
Lesbian: Females attracted to females.
Bisexual: People attracted to both sexes.
Transgender: People whose gender identity does not match their birth sex.
Queer: An all-encompassing term for orientations other that heterosexual.
Questioning: Individuals who are exploring sexual orientation or identity.
Asexual: Not sexual.
Allies: Important to sexual minorities.
intersex: People born with ambiguous genitalia. (The intersex community chose the lower-case “i.”)
2-Spirit people: Native American or indigenous Canadian term for gays and lesbians.
Zero Tolerance Is Effective for Physician Addicts : Monitoring, testing, and consequences for relapse could be extended to abusers in other settings.
SAN FRANCISCO – Zero tolerance for substance abuse and random testing have been the keys to successful rehabilitation of addicted doctors in physicians' health programs–and might help nonphysician populations of addicts.
Some of the program elements that have worked well for addicted physicians are beginning to be applied with surprisingly good results in other settings, especially the criminal justice system, Dr. Robert L. DuPont said. “If it's good enough for physicians, why not for everyone else?” he asked.
Intensive monitoring with frequent, random drug and alcohol testing backed by swift and certain consequences for a single relapse appear to keep a large majority of addicts substance free during monitoring periods of up to 5–7 years in recent studies.
That kind of zero-tolerance intensive monitoring differs markedly from competing theories of “harm reduction” and “compassion” for addicts that often amount to “enabling” the addiction, DuPont said.
“If the environment is tolerant of the drug use, then the drug use is more prevalent and it persists. If the environment is intolerant, the use stops, whether it's in physicians or convicted felons,” said Dr. DuPont, who was the first director of the National Institute on Drug Abuse 37 years ago and now is head of the Institute for Behavior and Health, a drug policy nonprofit in Rockville, Md. “If you expect and tolerate relapse, you'll see more of it.”
He and his associates studied 904 consecutive admissions to 16 state physicians' health programs and found that 72% of the physicians were still licensed and practicing with no sign of substance abuse in 5–7 years of monitoring. Twelve percent had their license suspended or dropped, 6% were licensed but not practicing, 4% had retired or left practice, 4% had died, and the outcomes of the rest were unknown (J. Subst. Abuse Treat. 2009;36:159–71).
“It's striking how many of them go through successfully,” he said.
The physicians' health programs start with a careful initial evaluation followed by referral to intensive, high-quality treatment, which usually lasts for 1–3 months and is mostly in residential facilities. After treatment, the physicians undergo frequent, random drug and alcohol testing for 5 years or longer. Each work day, the physician must call a phone number to find out whether he or she will be tested that day.
The programs are closely tied to community support, mainly the 12-step groups of Alcoholics Anonymous and Narcotics Anonymous.
Participants who leave the program or have a single positive test for drugs or alcohol are removed from practice and sent to more intensive treatment. In addition, they risk losing their medical license after a repeat relapse.
“It's interesting that it's not a treatment program. They don't provide the treatment. It's a care management program,” and the treatment is only a few months out of years of monitoring, Dr. DuPont said.
A separate program applied similar intensive monitoring and zero-tolerance elements but without treatment and in nonphysicians. South Dakota's “24/7 Sobriety Project” required people convicted of driving while intoxicated or driving under the influence of alcohol to undergo 4 months of frequent testing. Any positive result or a missed test resulted in an immediate short-term stay in jail, usually for a few days. Testing initially required participants to come to a sheriff's office at 7 a.m. and 7 p.m. for alcohol breath tests, but the program later offered the alternatives of wearing an alcohol-monitoring ankle bracelet, frequent urinalysis, or wearing drug patches that collect sweat samples for drug testing.
From 2005 to 2009, 67% of 11,956 participants who underwent twice-daily breath tests never failed a test, and 17% failed only once, according to an unpublished analysis by Dr. DuPont and his associates. Among 1,383 participants who wore ankle bracelets, 75% had no violations. The program did 415 tests of patches from 45 participants, and 94% of tests were passes. The 1,261 participants who took 17,730 urine tests passed 98% of the time.
Dr. DuPont said the “quite remarkable” results probably were tied to intensive monitoring plus swift and certain consequences that were serious but not severe. He acknowledged, however, that monitoring was short term and that the program did not cover drugs of abuse.
Another model is being tried in Hawaii's Opportunity Probation with Enforcement (HOPE) program, which enrolls people on probation for the most serious drug problems or crimes (such as murder or rape) regardless of substance abuse.
In that program, participants undergo random drug testing for up to 6 years and are offered a treatment option. Noncompliance will result in immediate, short-term jail stays, usually within 72 hours of the offense, and possible mandatory inpatient or residential treatment.
The techniques used in these three program models in South Dakota, Hawaii, and physicians' health programs might be applicable to other settings where there is leverage to impose swift, meaningful consequences, such as the workplace, employee assistance programs, insurance or health care organizations, or families, Dr. DuPont suggested.
More research is needed, especially on the durability of outcomes after monitoring stops, he said.
Dr. DuPont said he has no pertinent conflicts of interest.
'If the environment is tolerant of drug use, then the drug use is more prevalent and persists.'
Source DR. DUPONT
SAN FRANCISCO – Zero tolerance for substance abuse and random testing have been the keys to successful rehabilitation of addicted doctors in physicians' health programs–and might help nonphysician populations of addicts.
Some of the program elements that have worked well for addicted physicians are beginning to be applied with surprisingly good results in other settings, especially the criminal justice system, Dr. Robert L. DuPont said. “If it's good enough for physicians, why not for everyone else?” he asked.
Intensive monitoring with frequent, random drug and alcohol testing backed by swift and certain consequences for a single relapse appear to keep a large majority of addicts substance free during monitoring periods of up to 5–7 years in recent studies.
That kind of zero-tolerance intensive monitoring differs markedly from competing theories of “harm reduction” and “compassion” for addicts that often amount to “enabling” the addiction, DuPont said.
“If the environment is tolerant of the drug use, then the drug use is more prevalent and it persists. If the environment is intolerant, the use stops, whether it's in physicians or convicted felons,” said Dr. DuPont, who was the first director of the National Institute on Drug Abuse 37 years ago and now is head of the Institute for Behavior and Health, a drug policy nonprofit in Rockville, Md. “If you expect and tolerate relapse, you'll see more of it.”
He and his associates studied 904 consecutive admissions to 16 state physicians' health programs and found that 72% of the physicians were still licensed and practicing with no sign of substance abuse in 5–7 years of monitoring. Twelve percent had their license suspended or dropped, 6% were licensed but not practicing, 4% had retired or left practice, 4% had died, and the outcomes of the rest were unknown (J. Subst. Abuse Treat. 2009;36:159–71).
“It's striking how many of them go through successfully,” he said.
The physicians' health programs start with a careful initial evaluation followed by referral to intensive, high-quality treatment, which usually lasts for 1–3 months and is mostly in residential facilities. After treatment, the physicians undergo frequent, random drug and alcohol testing for 5 years or longer. Each work day, the physician must call a phone number to find out whether he or she will be tested that day.
The programs are closely tied to community support, mainly the 12-step groups of Alcoholics Anonymous and Narcotics Anonymous.
Participants who leave the program or have a single positive test for drugs or alcohol are removed from practice and sent to more intensive treatment. In addition, they risk losing their medical license after a repeat relapse.
“It's interesting that it's not a treatment program. They don't provide the treatment. It's a care management program,” and the treatment is only a few months out of years of monitoring, Dr. DuPont said.
A separate program applied similar intensive monitoring and zero-tolerance elements but without treatment and in nonphysicians. South Dakota's “24/7 Sobriety Project” required people convicted of driving while intoxicated or driving under the influence of alcohol to undergo 4 months of frequent testing. Any positive result or a missed test resulted in an immediate short-term stay in jail, usually for a few days. Testing initially required participants to come to a sheriff's office at 7 a.m. and 7 p.m. for alcohol breath tests, but the program later offered the alternatives of wearing an alcohol-monitoring ankle bracelet, frequent urinalysis, or wearing drug patches that collect sweat samples for drug testing.
From 2005 to 2009, 67% of 11,956 participants who underwent twice-daily breath tests never failed a test, and 17% failed only once, according to an unpublished analysis by Dr. DuPont and his associates. Among 1,383 participants who wore ankle bracelets, 75% had no violations. The program did 415 tests of patches from 45 participants, and 94% of tests were passes. The 1,261 participants who took 17,730 urine tests passed 98% of the time.
Dr. DuPont said the “quite remarkable” results probably were tied to intensive monitoring plus swift and certain consequences that were serious but not severe. He acknowledged, however, that monitoring was short term and that the program did not cover drugs of abuse.
Another model is being tried in Hawaii's Opportunity Probation with Enforcement (HOPE) program, which enrolls people on probation for the most serious drug problems or crimes (such as murder or rape) regardless of substance abuse.
In that program, participants undergo random drug testing for up to 6 years and are offered a treatment option. Noncompliance will result in immediate, short-term jail stays, usually within 72 hours of the offense, and possible mandatory inpatient or residential treatment.
The techniques used in these three program models in South Dakota, Hawaii, and physicians' health programs might be applicable to other settings where there is leverage to impose swift, meaningful consequences, such as the workplace, employee assistance programs, insurance or health care organizations, or families, Dr. DuPont suggested.
More research is needed, especially on the durability of outcomes after monitoring stops, he said.
Dr. DuPont said he has no pertinent conflicts of interest.
'If the environment is tolerant of drug use, then the drug use is more prevalent and persists.'
Source DR. DUPONT
SAN FRANCISCO – Zero tolerance for substance abuse and random testing have been the keys to successful rehabilitation of addicted doctors in physicians' health programs–and might help nonphysician populations of addicts.
Some of the program elements that have worked well for addicted physicians are beginning to be applied with surprisingly good results in other settings, especially the criminal justice system, Dr. Robert L. DuPont said. “If it's good enough for physicians, why not for everyone else?” he asked.
Intensive monitoring with frequent, random drug and alcohol testing backed by swift and certain consequences for a single relapse appear to keep a large majority of addicts substance free during monitoring periods of up to 5–7 years in recent studies.
That kind of zero-tolerance intensive monitoring differs markedly from competing theories of “harm reduction” and “compassion” for addicts that often amount to “enabling” the addiction, DuPont said.
“If the environment is tolerant of the drug use, then the drug use is more prevalent and it persists. If the environment is intolerant, the use stops, whether it's in physicians or convicted felons,” said Dr. DuPont, who was the first director of the National Institute on Drug Abuse 37 years ago and now is head of the Institute for Behavior and Health, a drug policy nonprofit in Rockville, Md. “If you expect and tolerate relapse, you'll see more of it.”
He and his associates studied 904 consecutive admissions to 16 state physicians' health programs and found that 72% of the physicians were still licensed and practicing with no sign of substance abuse in 5–7 years of monitoring. Twelve percent had their license suspended or dropped, 6% were licensed but not practicing, 4% had retired or left practice, 4% had died, and the outcomes of the rest were unknown (J. Subst. Abuse Treat. 2009;36:159–71).
“It's striking how many of them go through successfully,” he said.
The physicians' health programs start with a careful initial evaluation followed by referral to intensive, high-quality treatment, which usually lasts for 1–3 months and is mostly in residential facilities. After treatment, the physicians undergo frequent, random drug and alcohol testing for 5 years or longer. Each work day, the physician must call a phone number to find out whether he or she will be tested that day.
The programs are closely tied to community support, mainly the 12-step groups of Alcoholics Anonymous and Narcotics Anonymous.
Participants who leave the program or have a single positive test for drugs or alcohol are removed from practice and sent to more intensive treatment. In addition, they risk losing their medical license after a repeat relapse.
“It's interesting that it's not a treatment program. They don't provide the treatment. It's a care management program,” and the treatment is only a few months out of years of monitoring, Dr. DuPont said.
A separate program applied similar intensive monitoring and zero-tolerance elements but without treatment and in nonphysicians. South Dakota's “24/7 Sobriety Project” required people convicted of driving while intoxicated or driving under the influence of alcohol to undergo 4 months of frequent testing. Any positive result or a missed test resulted in an immediate short-term stay in jail, usually for a few days. Testing initially required participants to come to a sheriff's office at 7 a.m. and 7 p.m. for alcohol breath tests, but the program later offered the alternatives of wearing an alcohol-monitoring ankle bracelet, frequent urinalysis, or wearing drug patches that collect sweat samples for drug testing.
From 2005 to 2009, 67% of 11,956 participants who underwent twice-daily breath tests never failed a test, and 17% failed only once, according to an unpublished analysis by Dr. DuPont and his associates. Among 1,383 participants who wore ankle bracelets, 75% had no violations. The program did 415 tests of patches from 45 participants, and 94% of tests were passes. The 1,261 participants who took 17,730 urine tests passed 98% of the time.
Dr. DuPont said the “quite remarkable” results probably were tied to intensive monitoring plus swift and certain consequences that were serious but not severe. He acknowledged, however, that monitoring was short term and that the program did not cover drugs of abuse.
Another model is being tried in Hawaii's Opportunity Probation with Enforcement (HOPE) program, which enrolls people on probation for the most serious drug problems or crimes (such as murder or rape) regardless of substance abuse.
In that program, participants undergo random drug testing for up to 6 years and are offered a treatment option. Noncompliance will result in immediate, short-term jail stays, usually within 72 hours of the offense, and possible mandatory inpatient or residential treatment.
The techniques used in these three program models in South Dakota, Hawaii, and physicians' health programs might be applicable to other settings where there is leverage to impose swift, meaningful consequences, such as the workplace, employee assistance programs, insurance or health care organizations, or families, Dr. DuPont suggested.
More research is needed, especially on the durability of outcomes after monitoring stops, he said.
Dr. DuPont said he has no pertinent conflicts of interest.
'If the environment is tolerant of drug use, then the drug use is more prevalent and persists.'
Source DR. DUPONT
Guidelines Target Long-Term Care Transitions : Timely communication of adequate clinical data is instrumental to safe transitions.
LONG BEACH, CALIF. — New clinical practice guidelines for the first time provide principles and tools for safely transitioning long-term care patients from one care setting to another.
The 99-page online document for physicians and other health care professionals is not a “how to” guide but a distillation of key steps in better care transitions, Dr. James E. Lett II said at the meeting.
He said that, when he tries to talk to long-term care professionals about good transitions of care, the common response is, “We already do that.”
“I hate this phrase, and I hear it so many times,” said Dr. Lett of the California Department of Corrections and Rehabilitation, Sacramento. “Usually, what they have is a series of preprinted forms with no accountability for who fills them out or how they're completed.”
Transitions of care, as currently practiced, result in nearly one-fifth of Medicare beneficiaries discharged from hospitals being rehospitalized within 30 days. Studies suggest that 90% of these readmissions are unplanned and 13% are potentially avoidable, which the Centers for Medicare and Medicaid Services has estimated costs an extra $12 billion per year, Dr. Lett said. He chaired the interdisciplinary working group that created the new clinical practice guidelines “Transitions of Care in the Long-Term Care Continuum” for the American Medical Directors Association, with participants from the American Medical Association, the American Geriatrics Society, and other organizations.
One foundation of the guidelines is that a care transition should be a patient-centered activity. “You don't shoehorn a patient into your transition process. You build the transition process around the patient,” Dr. Lett said. Information moves with the patient. The patient and his or her family participate in decisions. The needs of the patient predominate, and advance directives should be available at each site of care.
Another central concept is that medication reconciliation must occur with every transition at both the sending and receiving sites of care.
Good transitions of care are “the ultimate interdisciplinary team activity,” with every member of the care team involved, accountable, and responsive, Dr. Lett said. Caregivers from the sending site must maintain responsibility for a patient, or at least be available, until caregivers at the receiving site can assume management of the person's care.
“No longer can we expect in this complex, fractionated world that the receiving site will have all they need and the patient will do well,” Dr. Lett said.
Timely communication of adequate clinical data is instrumental to safe transitions, he added. The new guidelines include a universal transfer form that can be modified by individual institutions. Tables and appendices cover the essential elements in medication reconciliation, provide sample policies, suggest a pretransition checklist and information that emergency medical services transport may request, and review myths and facts about HIPAA as it applies to transitions of care.
Dr. Lett reviewed the basic steps for implementing a care-transition program:
▸ Clinicians should be ready for surveillance and monitoring of a status change in both planned and unplanned transitions.
▸ Interdisciplinary team members must communicate with each other and with the patient and/or family to determine the most appropriate care transition. An advance directive also should be discussed.
▸ In a planned transition, the sending facility communicates with the receiving facility, which receives patient information prior to the patient's arrival. Lists and sample forms in the guidelines can help you plan.
▸ Patients can have an acute change of condition and need an unplanned transfer to an emergency department at any time. Keep the necessary forms available at all hours.
▸ When a patient is being transferred to another care site by emergency medical services, make sure the handoff is documented. Dr. Lett described one case of a patient with dementia and stroke who was “lost” after the emergency medical services transport team said it delivered the patient to a room and left the transfer paperwork on a dresser.
▸ When a patient's condition improves to the point that a planned transfer to his or her community home is appropriate, clinicians in the sending facility should address pending tests or other imminent needs of the patient.
▸ When a patient is nearing the end of life, a planned transition to solely comfort care is appropriate. Make sure any needed equipment is available and don't forget medication reconciliation.
▸ After a patient is physically handed over to the receiving setting of care in a planned transition, “you need to convey recommendations on next steps” and do medication reconciliation.
▸ If a patient is discharged to his or her community home in a planned transition, make sure you address the availability of transportation, the affordability of medications in the new setting, and medication reconciliation.
▸ In planned and unplanned transitions, both the sending and receiving entities must verify that the patient has been handed over and that essential patient information has been received. “You must expect that the other side will verify this with you, and if they don't, you push it.”
▸ In both planned and unplanned transitions, the sending facility follows up to confirm and document that the patient has successfully transitioned to the new setting of care.
▸ Monitor a facility's performance in managing care transitions. “You can't improve what you can't measure,” Dr. Lett said. “Being able to say, 'We've been monitoring, and we didn't receive a history and physical 53% of the time' is a different discussion than saying, 'You never sent us a history and physical.'”
An appendix in the guidelines lists quality indicators and performance measures for care transitions.
Disclosures: Dr. Lett disclosed having no relevant conflicts of interest. Sanofi-Aventis helped fund distribution of the guidelines, which are available at www.amda.com/tools/clinical/TOCCPG/index.html
'No longer can we expect in this complex, fractionated world that the receiving site will have all they need.'
Source DR. LETT
LONG BEACH, CALIF. — New clinical practice guidelines for the first time provide principles and tools for safely transitioning long-term care patients from one care setting to another.
The 99-page online document for physicians and other health care professionals is not a “how to” guide but a distillation of key steps in better care transitions, Dr. James E. Lett II said at the meeting.
He said that, when he tries to talk to long-term care professionals about good transitions of care, the common response is, “We already do that.”
“I hate this phrase, and I hear it so many times,” said Dr. Lett of the California Department of Corrections and Rehabilitation, Sacramento. “Usually, what they have is a series of preprinted forms with no accountability for who fills them out or how they're completed.”
Transitions of care, as currently practiced, result in nearly one-fifth of Medicare beneficiaries discharged from hospitals being rehospitalized within 30 days. Studies suggest that 90% of these readmissions are unplanned and 13% are potentially avoidable, which the Centers for Medicare and Medicaid Services has estimated costs an extra $12 billion per year, Dr. Lett said. He chaired the interdisciplinary working group that created the new clinical practice guidelines “Transitions of Care in the Long-Term Care Continuum” for the American Medical Directors Association, with participants from the American Medical Association, the American Geriatrics Society, and other organizations.
One foundation of the guidelines is that a care transition should be a patient-centered activity. “You don't shoehorn a patient into your transition process. You build the transition process around the patient,” Dr. Lett said. Information moves with the patient. The patient and his or her family participate in decisions. The needs of the patient predominate, and advance directives should be available at each site of care.
Another central concept is that medication reconciliation must occur with every transition at both the sending and receiving sites of care.
Good transitions of care are “the ultimate interdisciplinary team activity,” with every member of the care team involved, accountable, and responsive, Dr. Lett said. Caregivers from the sending site must maintain responsibility for a patient, or at least be available, until caregivers at the receiving site can assume management of the person's care.
“No longer can we expect in this complex, fractionated world that the receiving site will have all they need and the patient will do well,” Dr. Lett said.
Timely communication of adequate clinical data is instrumental to safe transitions, he added. The new guidelines include a universal transfer form that can be modified by individual institutions. Tables and appendices cover the essential elements in medication reconciliation, provide sample policies, suggest a pretransition checklist and information that emergency medical services transport may request, and review myths and facts about HIPAA as it applies to transitions of care.
Dr. Lett reviewed the basic steps for implementing a care-transition program:
▸ Clinicians should be ready for surveillance and monitoring of a status change in both planned and unplanned transitions.
▸ Interdisciplinary team members must communicate with each other and with the patient and/or family to determine the most appropriate care transition. An advance directive also should be discussed.
▸ In a planned transition, the sending facility communicates with the receiving facility, which receives patient information prior to the patient's arrival. Lists and sample forms in the guidelines can help you plan.
▸ Patients can have an acute change of condition and need an unplanned transfer to an emergency department at any time. Keep the necessary forms available at all hours.
▸ When a patient is being transferred to another care site by emergency medical services, make sure the handoff is documented. Dr. Lett described one case of a patient with dementia and stroke who was “lost” after the emergency medical services transport team said it delivered the patient to a room and left the transfer paperwork on a dresser.
▸ When a patient's condition improves to the point that a planned transfer to his or her community home is appropriate, clinicians in the sending facility should address pending tests or other imminent needs of the patient.
▸ When a patient is nearing the end of life, a planned transition to solely comfort care is appropriate. Make sure any needed equipment is available and don't forget medication reconciliation.
▸ After a patient is physically handed over to the receiving setting of care in a planned transition, “you need to convey recommendations on next steps” and do medication reconciliation.
▸ If a patient is discharged to his or her community home in a planned transition, make sure you address the availability of transportation, the affordability of medications in the new setting, and medication reconciliation.
▸ In planned and unplanned transitions, both the sending and receiving entities must verify that the patient has been handed over and that essential patient information has been received. “You must expect that the other side will verify this with you, and if they don't, you push it.”
▸ In both planned and unplanned transitions, the sending facility follows up to confirm and document that the patient has successfully transitioned to the new setting of care.
▸ Monitor a facility's performance in managing care transitions. “You can't improve what you can't measure,” Dr. Lett said. “Being able to say, 'We've been monitoring, and we didn't receive a history and physical 53% of the time' is a different discussion than saying, 'You never sent us a history and physical.'”
An appendix in the guidelines lists quality indicators and performance measures for care transitions.
Disclosures: Dr. Lett disclosed having no relevant conflicts of interest. Sanofi-Aventis helped fund distribution of the guidelines, which are available at www.amda.com/tools/clinical/TOCCPG/index.html
'No longer can we expect in this complex, fractionated world that the receiving site will have all they need.'
Source DR. LETT
LONG BEACH, CALIF. — New clinical practice guidelines for the first time provide principles and tools for safely transitioning long-term care patients from one care setting to another.
The 99-page online document for physicians and other health care professionals is not a “how to” guide but a distillation of key steps in better care transitions, Dr. James E. Lett II said at the meeting.
He said that, when he tries to talk to long-term care professionals about good transitions of care, the common response is, “We already do that.”
“I hate this phrase, and I hear it so many times,” said Dr. Lett of the California Department of Corrections and Rehabilitation, Sacramento. “Usually, what they have is a series of preprinted forms with no accountability for who fills them out or how they're completed.”
Transitions of care, as currently practiced, result in nearly one-fifth of Medicare beneficiaries discharged from hospitals being rehospitalized within 30 days. Studies suggest that 90% of these readmissions are unplanned and 13% are potentially avoidable, which the Centers for Medicare and Medicaid Services has estimated costs an extra $12 billion per year, Dr. Lett said. He chaired the interdisciplinary working group that created the new clinical practice guidelines “Transitions of Care in the Long-Term Care Continuum” for the American Medical Directors Association, with participants from the American Medical Association, the American Geriatrics Society, and other organizations.
One foundation of the guidelines is that a care transition should be a patient-centered activity. “You don't shoehorn a patient into your transition process. You build the transition process around the patient,” Dr. Lett said. Information moves with the patient. The patient and his or her family participate in decisions. The needs of the patient predominate, and advance directives should be available at each site of care.
Another central concept is that medication reconciliation must occur with every transition at both the sending and receiving sites of care.
Good transitions of care are “the ultimate interdisciplinary team activity,” with every member of the care team involved, accountable, and responsive, Dr. Lett said. Caregivers from the sending site must maintain responsibility for a patient, or at least be available, until caregivers at the receiving site can assume management of the person's care.
“No longer can we expect in this complex, fractionated world that the receiving site will have all they need and the patient will do well,” Dr. Lett said.
Timely communication of adequate clinical data is instrumental to safe transitions, he added. The new guidelines include a universal transfer form that can be modified by individual institutions. Tables and appendices cover the essential elements in medication reconciliation, provide sample policies, suggest a pretransition checklist and information that emergency medical services transport may request, and review myths and facts about HIPAA as it applies to transitions of care.
Dr. Lett reviewed the basic steps for implementing a care-transition program:
▸ Clinicians should be ready for surveillance and monitoring of a status change in both planned and unplanned transitions.
▸ Interdisciplinary team members must communicate with each other and with the patient and/or family to determine the most appropriate care transition. An advance directive also should be discussed.
▸ In a planned transition, the sending facility communicates with the receiving facility, which receives patient information prior to the patient's arrival. Lists and sample forms in the guidelines can help you plan.
▸ Patients can have an acute change of condition and need an unplanned transfer to an emergency department at any time. Keep the necessary forms available at all hours.
▸ When a patient is being transferred to another care site by emergency medical services, make sure the handoff is documented. Dr. Lett described one case of a patient with dementia and stroke who was “lost” after the emergency medical services transport team said it delivered the patient to a room and left the transfer paperwork on a dresser.
▸ When a patient's condition improves to the point that a planned transfer to his or her community home is appropriate, clinicians in the sending facility should address pending tests or other imminent needs of the patient.
▸ When a patient is nearing the end of life, a planned transition to solely comfort care is appropriate. Make sure any needed equipment is available and don't forget medication reconciliation.
▸ After a patient is physically handed over to the receiving setting of care in a planned transition, “you need to convey recommendations on next steps” and do medication reconciliation.
▸ If a patient is discharged to his or her community home in a planned transition, make sure you address the availability of transportation, the affordability of medications in the new setting, and medication reconciliation.
▸ In planned and unplanned transitions, both the sending and receiving entities must verify that the patient has been handed over and that essential patient information has been received. “You must expect that the other side will verify this with you, and if they don't, you push it.”
▸ In both planned and unplanned transitions, the sending facility follows up to confirm and document that the patient has successfully transitioned to the new setting of care.
▸ Monitor a facility's performance in managing care transitions. “You can't improve what you can't measure,” Dr. Lett said. “Being able to say, 'We've been monitoring, and we didn't receive a history and physical 53% of the time' is a different discussion than saying, 'You never sent us a history and physical.'”
An appendix in the guidelines lists quality indicators and performance measures for care transitions.
Disclosures: Dr. Lett disclosed having no relevant conflicts of interest. Sanofi-Aventis helped fund distribution of the guidelines, which are available at www.amda.com/tools/clinical/TOCCPG/index.html
'No longer can we expect in this complex, fractionated world that the receiving site will have all they need.'
Source DR. LETT
No. of Steps Vary to Reach Activity Goals
Public health recommendations to get 150 minutes per week of moderate-intensity physical activity could be met by walking 100 steps per minute in that time, but the number of steps needed varies depending on the height of the individual, preliminary data in a small study suggest.
Investigators measured the height, body mass index, and leg length of 20 healthy adults, then measured oxygen uptake by connecting them to a portable spirometer during five 6-minute walks at different speeds. One researcher set the pace in front of the subject using a distance-measuring wheel and cycle computer, and another followed behind to measure steps using a hand-operated counter.
They defined moderate-intensity activity as expending three metabolic equivalents (METs), with each MET defined as oxygen uptake of 3.5 mL/kg per minute. Using the subjects' mean values for body mass index and leg length, they estimated that 100 steps per minute would be needed to expend 3 METs, reported Michael W. Beets, Ph.D., of the University of South Carolina, Columbia, and his associates.
Individuals with longer legs, however, needed fewer the steps for moderate-intensity activity. The correlation between height and leg length suggests that to expend 3 METs, a 5-foot-tall person would need 111 steps per minute, someone who is 5 feet 6 inches tall would need 103 steps per minute, a 6-foot-tall person would need 94 steps per minute, and someone who is 6 feet 6 inches tall would need 85 steps per minute, the investigators said (J. Sci. Med. Sport 2010 [doi:10.1016/j.jsams.2009.11.002]).
The researchers extrapolated that, in order to get 30 minutes of moderate-intensity physical activity, a 5-foot person would need to take 3,330 steps, while a 6-foot person would need only 2,820 steps—a 510-step difference. To meet the public health goal of 150 minutes per week of moderate-intensity activity would require 16,650 steps within that time period for someone 5 feet tall, compared with 14,100 steps for someone 6 feet tall—a difference of 2,550 steps.
The small size of the study limits the ability to generalize the results, but some of the findings are supported by previous data suggesting that 100 steps per minute may be a useful marker on average for meeting moderate-intensity activity goals (Am. J. Prev. Med. 2009;36:410-5).
Disclosures: The investigators stated that they did not have any conflicts of interest.
Public health recommendations to get 150 minutes per week of moderate-intensity physical activity could be met by walking 100 steps per minute in that time, but the number of steps needed varies depending on the height of the individual, preliminary data in a small study suggest.
Investigators measured the height, body mass index, and leg length of 20 healthy adults, then measured oxygen uptake by connecting them to a portable spirometer during five 6-minute walks at different speeds. One researcher set the pace in front of the subject using a distance-measuring wheel and cycle computer, and another followed behind to measure steps using a hand-operated counter.
They defined moderate-intensity activity as expending three metabolic equivalents (METs), with each MET defined as oxygen uptake of 3.5 mL/kg per minute. Using the subjects' mean values for body mass index and leg length, they estimated that 100 steps per minute would be needed to expend 3 METs, reported Michael W. Beets, Ph.D., of the University of South Carolina, Columbia, and his associates.
Individuals with longer legs, however, needed fewer the steps for moderate-intensity activity. The correlation between height and leg length suggests that to expend 3 METs, a 5-foot-tall person would need 111 steps per minute, someone who is 5 feet 6 inches tall would need 103 steps per minute, a 6-foot-tall person would need 94 steps per minute, and someone who is 6 feet 6 inches tall would need 85 steps per minute, the investigators said (J. Sci. Med. Sport 2010 [doi:10.1016/j.jsams.2009.11.002]).
The researchers extrapolated that, in order to get 30 minutes of moderate-intensity physical activity, a 5-foot person would need to take 3,330 steps, while a 6-foot person would need only 2,820 steps—a 510-step difference. To meet the public health goal of 150 minutes per week of moderate-intensity activity would require 16,650 steps within that time period for someone 5 feet tall, compared with 14,100 steps for someone 6 feet tall—a difference of 2,550 steps.
The small size of the study limits the ability to generalize the results, but some of the findings are supported by previous data suggesting that 100 steps per minute may be a useful marker on average for meeting moderate-intensity activity goals (Am. J. Prev. Med. 2009;36:410-5).
Disclosures: The investigators stated that they did not have any conflicts of interest.
Public health recommendations to get 150 minutes per week of moderate-intensity physical activity could be met by walking 100 steps per minute in that time, but the number of steps needed varies depending on the height of the individual, preliminary data in a small study suggest.
Investigators measured the height, body mass index, and leg length of 20 healthy adults, then measured oxygen uptake by connecting them to a portable spirometer during five 6-minute walks at different speeds. One researcher set the pace in front of the subject using a distance-measuring wheel and cycle computer, and another followed behind to measure steps using a hand-operated counter.
They defined moderate-intensity activity as expending three metabolic equivalents (METs), with each MET defined as oxygen uptake of 3.5 mL/kg per minute. Using the subjects' mean values for body mass index and leg length, they estimated that 100 steps per minute would be needed to expend 3 METs, reported Michael W. Beets, Ph.D., of the University of South Carolina, Columbia, and his associates.
Individuals with longer legs, however, needed fewer the steps for moderate-intensity activity. The correlation between height and leg length suggests that to expend 3 METs, a 5-foot-tall person would need 111 steps per minute, someone who is 5 feet 6 inches tall would need 103 steps per minute, a 6-foot-tall person would need 94 steps per minute, and someone who is 6 feet 6 inches tall would need 85 steps per minute, the investigators said (J. Sci. Med. Sport 2010 [doi:10.1016/j.jsams.2009.11.002]).
The researchers extrapolated that, in order to get 30 minutes of moderate-intensity physical activity, a 5-foot person would need to take 3,330 steps, while a 6-foot person would need only 2,820 steps—a 510-step difference. To meet the public health goal of 150 minutes per week of moderate-intensity activity would require 16,650 steps within that time period for someone 5 feet tall, compared with 14,100 steps for someone 6 feet tall—a difference of 2,550 steps.
The small size of the study limits the ability to generalize the results, but some of the findings are supported by previous data suggesting that 100 steps per minute may be a useful marker on average for meeting moderate-intensity activity goals (Am. J. Prev. Med. 2009;36:410-5).
Disclosures: The investigators stated that they did not have any conflicts of interest.