User login
Perception of Aortic Valve Surgery Risk May Be Exaggerated
CHICAGO – Despite a steady rise in the number of patients presenting with aortic stenosis, many patients are not being referred for surgery because of the perception that the surgical risk is too high, according to Dr. Leonard Girardi.
The challenge for surgeons is to get the message out to their cardiology colleagues and to an increasingly sophisticated patient population what the real risks of aortic valve replacement are, Dr. Girardi said at Heart Valve Summit 2011.
Asymptomatic severe aortic stenosis has generally been considered a benign prognosis, and thus the American College of Cardiology/American Heart Association guidelines have not recommended aortic valve replacement (AVR) for patients with isolated asymptomatic severe stenosis in the absence of concomitant cardiac or aortic surgery. Data from three large trials, however, show that the probability of patients with severe aortic stenosis remaining symptom- or event-free at 2 years is 21%-67%.
Moreover, in a study of 740 patients with severe aortic stenosis, mean survival more than doubled from 3.9 years without AVR to 8.2 years with surgery among asymptomatic patients, and from 2.9 years to 6.5 years among symptomatic patients (Annals Thor. Surg. 2006;82:2116-22). While 10% of patients in the cohort opted not to have surgery, a full 50% were not even referred for surgery because their cardiologist deemed them too high risk, said Dr. Girardi, the O. Wayne Isom Professor of Cardiothoracic Surgery at Weill Cornell Medical College in New York.
Given that most centers have an operating mortality of 3% overall and 1% or less among low-risk patients, there is ever-increasing evidence that surgery is the right thing to do, he said.
Accurate risk assessment for AVR is problematic, however, because all of the risk stratification algorithms are flawed and overestimate mortality risk. The Society of Thoracic Surgeons (STS) PROM algorithm is the most accurate, but it has no way to report ejection fraction (EF) – a variable that would clearly stratify a patient with an EF of 20% from one with an EF of 60%, he said.
The logistic and additive EuroSCORES do not take into account cerebral vascular accident or presence of a previous stroke. Instead, they use the vague classification of "neurological dysfunction," which simply means neurologic activity affecting daily function.
Finally, the Ambler risk score, which was recently developed for valve surgery, takes into account only 13 variables, Dr. Girardi said at the meeting, cosponsored by the American Association for Thoracic Surgery and the American College of Cardiology Foundation.
Researchers at Cornell used the four different algorithms to calculate the risk of mortality among 638 patients requiring AVR, in whom actual mortality was 3.76%. The STS PROM score was the closest at 4.26%, while estimated mortality came in at a whopping 13.21% using the logistic EuroSCORE, 7.46% with the additive EuroSCORE, and 6.99% with the Ambler score, Dr. Girardi said. The ability of these algorithms to estimate mortality risk in high-risk patients – even those in the upper 10th percentile of risk – proved even worse, he added.
So what’s a surgeon to do?
The answer may be homegrown. "You have to look at your local and regional databases to see what surgeons are doing, because I think they’re certainly doing a lot better job than these scoring systems are suggesting they are," Dr. Girardi said.
New York State has one of the nation’s oldest databases – all New York cardiac surgery centers are required to participate in it, and the State Department of Health routinely audits it. The 40,000-patient database puts the mortality risk for AVR among all comers at 2.6%, increasing to 4.5% when AVR is combined with coronary artery bypass grafting (CABG). The outcomes are similar at 2.7% and 4.2%, respectively, based on data from the latest STS database, Dr. Girardi said.
Mortality risk estimates must also be coupled with complication rates, particularly given the increased risk of stroke with transcatheter aortic valve implantation. At Cornell, the risk of AVR and AVR/CABG was 1.1% in 2009 and 0.4% in 2010. During the same period, the stroke rate for these patients was 0.7%, acute renal failure 0.5%, and respiratory failure rate 5%, but that includes intubation for more than 72 hours, he pointed out.
Data should also be analyzed for the increasingly important population of octogenarians. Over the past 5 years, there was a 30% increase in the overall number of AVR patients in New York State but a 70% increase in the number of octogenarians undergoing AVR with or without CABG, Dr. Girardi observed. For 2009 and 2010, the mortality rate at Cornell for octogenarians – of whom 25% were older than 90 years – was 1.1% among all comers, with a stroke rate of 1% and a respiratory failure rate of 7%, largely because of increased patient frailty.
"High-risk cohorts clearly need even more analysis to figure out what we can do with them," he said. "But I think we’re going to have to get better at this as we compare our therapies with less invasive therapies coming along."
Dr. Girardi reported no relevant conflicts of interest.
CHICAGO – Despite a steady rise in the number of patients presenting with aortic stenosis, many patients are not being referred for surgery because of the perception that the surgical risk is too high, according to Dr. Leonard Girardi.
The challenge for surgeons is to get the message out to their cardiology colleagues and to an increasingly sophisticated patient population what the real risks of aortic valve replacement are, Dr. Girardi said at Heart Valve Summit 2011.
Asymptomatic severe aortic stenosis has generally been considered a benign prognosis, and thus the American College of Cardiology/American Heart Association guidelines have not recommended aortic valve replacement (AVR) for patients with isolated asymptomatic severe stenosis in the absence of concomitant cardiac or aortic surgery. Data from three large trials, however, show that the probability of patients with severe aortic stenosis remaining symptom- or event-free at 2 years is 21%-67%.
Moreover, in a study of 740 patients with severe aortic stenosis, mean survival more than doubled from 3.9 years without AVR to 8.2 years with surgery among asymptomatic patients, and from 2.9 years to 6.5 years among symptomatic patients (Annals Thor. Surg. 2006;82:2116-22). While 10% of patients in the cohort opted not to have surgery, a full 50% were not even referred for surgery because their cardiologist deemed them too high risk, said Dr. Girardi, the O. Wayne Isom Professor of Cardiothoracic Surgery at Weill Cornell Medical College in New York.
Given that most centers have an operating mortality of 3% overall and 1% or less among low-risk patients, there is ever-increasing evidence that surgery is the right thing to do, he said.
Accurate risk assessment for AVR is problematic, however, because all of the risk stratification algorithms are flawed and overestimate mortality risk. The Society of Thoracic Surgeons (STS) PROM algorithm is the most accurate, but it has no way to report ejection fraction (EF) – a variable that would clearly stratify a patient with an EF of 20% from one with an EF of 60%, he said.
The logistic and additive EuroSCORES do not take into account cerebral vascular accident or presence of a previous stroke. Instead, they use the vague classification of "neurological dysfunction," which simply means neurologic activity affecting daily function.
Finally, the Ambler risk score, which was recently developed for valve surgery, takes into account only 13 variables, Dr. Girardi said at the meeting, cosponsored by the American Association for Thoracic Surgery and the American College of Cardiology Foundation.
Researchers at Cornell used the four different algorithms to calculate the risk of mortality among 638 patients requiring AVR, in whom actual mortality was 3.76%. The STS PROM score was the closest at 4.26%, while estimated mortality came in at a whopping 13.21% using the logistic EuroSCORE, 7.46% with the additive EuroSCORE, and 6.99% with the Ambler score, Dr. Girardi said. The ability of these algorithms to estimate mortality risk in high-risk patients – even those in the upper 10th percentile of risk – proved even worse, he added.
So what’s a surgeon to do?
The answer may be homegrown. "You have to look at your local and regional databases to see what surgeons are doing, because I think they’re certainly doing a lot better job than these scoring systems are suggesting they are," Dr. Girardi said.
New York State has one of the nation’s oldest databases – all New York cardiac surgery centers are required to participate in it, and the State Department of Health routinely audits it. The 40,000-patient database puts the mortality risk for AVR among all comers at 2.6%, increasing to 4.5% when AVR is combined with coronary artery bypass grafting (CABG). The outcomes are similar at 2.7% and 4.2%, respectively, based on data from the latest STS database, Dr. Girardi said.
Mortality risk estimates must also be coupled with complication rates, particularly given the increased risk of stroke with transcatheter aortic valve implantation. At Cornell, the risk of AVR and AVR/CABG was 1.1% in 2009 and 0.4% in 2010. During the same period, the stroke rate for these patients was 0.7%, acute renal failure 0.5%, and respiratory failure rate 5%, but that includes intubation for more than 72 hours, he pointed out.
Data should also be analyzed for the increasingly important population of octogenarians. Over the past 5 years, there was a 30% increase in the overall number of AVR patients in New York State but a 70% increase in the number of octogenarians undergoing AVR with or without CABG, Dr. Girardi observed. For 2009 and 2010, the mortality rate at Cornell for octogenarians – of whom 25% were older than 90 years – was 1.1% among all comers, with a stroke rate of 1% and a respiratory failure rate of 7%, largely because of increased patient frailty.
"High-risk cohorts clearly need even more analysis to figure out what we can do with them," he said. "But I think we’re going to have to get better at this as we compare our therapies with less invasive therapies coming along."
Dr. Girardi reported no relevant conflicts of interest.
CHICAGO – Despite a steady rise in the number of patients presenting with aortic stenosis, many patients are not being referred for surgery because of the perception that the surgical risk is too high, according to Dr. Leonard Girardi.
The challenge for surgeons is to get the message out to their cardiology colleagues and to an increasingly sophisticated patient population what the real risks of aortic valve replacement are, Dr. Girardi said at Heart Valve Summit 2011.
Asymptomatic severe aortic stenosis has generally been considered a benign prognosis, and thus the American College of Cardiology/American Heart Association guidelines have not recommended aortic valve replacement (AVR) for patients with isolated asymptomatic severe stenosis in the absence of concomitant cardiac or aortic surgery. Data from three large trials, however, show that the probability of patients with severe aortic stenosis remaining symptom- or event-free at 2 years is 21%-67%.
Moreover, in a study of 740 patients with severe aortic stenosis, mean survival more than doubled from 3.9 years without AVR to 8.2 years with surgery among asymptomatic patients, and from 2.9 years to 6.5 years among symptomatic patients (Annals Thor. Surg. 2006;82:2116-22). While 10% of patients in the cohort opted not to have surgery, a full 50% were not even referred for surgery because their cardiologist deemed them too high risk, said Dr. Girardi, the O. Wayne Isom Professor of Cardiothoracic Surgery at Weill Cornell Medical College in New York.
Given that most centers have an operating mortality of 3% overall and 1% or less among low-risk patients, there is ever-increasing evidence that surgery is the right thing to do, he said.
Accurate risk assessment for AVR is problematic, however, because all of the risk stratification algorithms are flawed and overestimate mortality risk. The Society of Thoracic Surgeons (STS) PROM algorithm is the most accurate, but it has no way to report ejection fraction (EF) – a variable that would clearly stratify a patient with an EF of 20% from one with an EF of 60%, he said.
The logistic and additive EuroSCORES do not take into account cerebral vascular accident or presence of a previous stroke. Instead, they use the vague classification of "neurological dysfunction," which simply means neurologic activity affecting daily function.
Finally, the Ambler risk score, which was recently developed for valve surgery, takes into account only 13 variables, Dr. Girardi said at the meeting, cosponsored by the American Association for Thoracic Surgery and the American College of Cardiology Foundation.
Researchers at Cornell used the four different algorithms to calculate the risk of mortality among 638 patients requiring AVR, in whom actual mortality was 3.76%. The STS PROM score was the closest at 4.26%, while estimated mortality came in at a whopping 13.21% using the logistic EuroSCORE, 7.46% with the additive EuroSCORE, and 6.99% with the Ambler score, Dr. Girardi said. The ability of these algorithms to estimate mortality risk in high-risk patients – even those in the upper 10th percentile of risk – proved even worse, he added.
So what’s a surgeon to do?
The answer may be homegrown. "You have to look at your local and regional databases to see what surgeons are doing, because I think they’re certainly doing a lot better job than these scoring systems are suggesting they are," Dr. Girardi said.
New York State has one of the nation’s oldest databases – all New York cardiac surgery centers are required to participate in it, and the State Department of Health routinely audits it. The 40,000-patient database puts the mortality risk for AVR among all comers at 2.6%, increasing to 4.5% when AVR is combined with coronary artery bypass grafting (CABG). The outcomes are similar at 2.7% and 4.2%, respectively, based on data from the latest STS database, Dr. Girardi said.
Mortality risk estimates must also be coupled with complication rates, particularly given the increased risk of stroke with transcatheter aortic valve implantation. At Cornell, the risk of AVR and AVR/CABG was 1.1% in 2009 and 0.4% in 2010. During the same period, the stroke rate for these patients was 0.7%, acute renal failure 0.5%, and respiratory failure rate 5%, but that includes intubation for more than 72 hours, he pointed out.
Data should also be analyzed for the increasingly important population of octogenarians. Over the past 5 years, there was a 30% increase in the overall number of AVR patients in New York State but a 70% increase in the number of octogenarians undergoing AVR with or without CABG, Dr. Girardi observed. For 2009 and 2010, the mortality rate at Cornell for octogenarians – of whom 25% were older than 90 years – was 1.1% among all comers, with a stroke rate of 1% and a respiratory failure rate of 7%, largely because of increased patient frailty.
"High-risk cohorts clearly need even more analysis to figure out what we can do with them," he said. "But I think we’re going to have to get better at this as we compare our therapies with less invasive therapies coming along."
Dr. Girardi reported no relevant conflicts of interest.
EXPERT ANALYSIS FROM HEART VALVE SUMMIT 2011
Risk Assessment Systems Found Flawed, Imprecise
CHICAGO – Risk scoring systems are increasingly being used to assign an indication for therapy and to guide reimbursement, but they are rarely sufficiently calibrated and discriminatory to direct individual patient decisions, according to Dr. Anelechi Anyanwu.
"Risk models are designed as tools to define risks of a population and not risks in the individual patient," Dr. Anyanwu said at the Heart Valve Summit 2011.
Dr. Anyanwu, a cardiothoracic surgeon at Mount Sinai Medical Center in New York City, ran through a checklist of what’s missing in today’s risk scoring systems, starting with the absence of all relevant risk factors. Models can adjust only for known risk factors or things that can be measured easily and objectively, but don’t adjust for unproven, unknown, or rare risk factors. "The reality is that most of the variance in outcome is not explained," he said.
Even such known and probable risk factors as poor lung reserve, unclampable aorta, cirrhosis, frailty, radiation, and socioeconomic status are not included in the scoring systems, but almost certainly influence outcome. A study of 230 patients undergoing cardiac surgery after thoracic radiation found worse in-hospital mortality, respiratory complications, and 4-year survival among those receiving extensive radiation vs. variable or tangential radiation – even though the extensively irradiated group was the youngest at 51 years vs. 64 years and 72 years, respectively (J. Thorac. Cardiovasc. Surg. 2007;133:404-13).
Differential Effect of Risk
Risk systems also don’t allow for the fact that risk factors can vary, or their relevance can disappear over time like the once-worrisome variable of 90% stenosis of the left main artery during primary isolated coronary artery bypass grafting (CABG), Dr. Anyanwu said.
The effect of the same risk fact can also differ in various patient subsets. For example, the EuroSCORE identifies a creatinine level of more than 200 mmol/L as a risk factor, but assumes similar risk for the subsets of creatinine clearance 30 mL/min, creatinine clearance 50 mL/min, and dialysis patients.
"Risk systems are imprecise and need further development to allow for more robust prediction."
Risk systems also assume equal weighting in all geographic regions, he said. The EuroSCORE Study Group reported that the predictive value of its scoring system among the six largest national samples in its database varied from "excellent" in Finland (area under the ROC curve 0.87) to "good" in Spain (AUC 0.74) (Eur. J. Cardiothorac. Surg. 2000;18:27-30).
Certainty
Scoring systems can never be 100% specific, Dr. Anyanwu reminded the audience. Quite often the probability range is broad, with imprecision greater if subjective or poorly defined predictor variables have been included in the model.
A high severity score cannot indicate absolute poor risk, and low severity scores cannot guarantee against poor outcome. "At the end of the day, outcome is always uncertain," he said.
Gaming the System
Patient selection is a powerful risk modification tool that cannot be captured by risk models. The more selective a center or surgeon is, the less the effect a specific risk factor will have, Dr. Anyanwu said. A low ejection fraction of less than 30% denotes poor prognosis in aortic valve replacement (AVR), and is accounted for in all risk systems. But not all low ejection fractions are the same, and surgeons can game the system by cherry-picking the better ones, he said.
He cited a study among 81 consecutive patients with symptomatic, calcified low-flow/low-gradient aortic stenosis that reported an operative mortality of 67% in the subgroup of patients with a mean pressure gradient of 20 mm Hg or less, compared with 16% in patients with an mean pressure gradient of more than 20 mm Hg. Moreover, if a surgeon opted not to perform CABG at the time of the AVR, operative mortality was just 10%, compared with 53% for patients in whom CABG was performed at the time of AVR (J. Am. Coll. Cardiol. 2009;53:1865-73).
Other Considerations
Numerous systems show varied outcome by different geographic regions, centers, and surgeons, and by surgical volume. In one study, the risk-adjusted odds of death among patients undergoing first-time elective surgery for mitral regurgitation was 0.71 at centers with an annual volume of 36-70 mitral operations, 0.74 at centers performing 71-140 mitral operations, and just 0.48 at the highest volume centers doing more than 140 mitral operations per year (Circulation 2007;115:881-7).
Despite such variations, risk scores will predict the same outcome for patient groups that clearly have different outcomes in practice, Dr. Anyanwu said at the meeting, sponsored by the American Association for Thoracic Surgery, American College of Cardiology Foundation, and The Society of Thoracic Surgeons.
He suggested that a good risk system has to be recalibrated frequently, and that further study is needed to allow for incorporation of nonpatient factors in risk prediction.
"Risk systems are imprecise and need further development to allow for more robust prediction," he concluded.
Dr. Anyanwu reported no relevant conflicts of interest.
CHICAGO – Risk scoring systems are increasingly being used to assign an indication for therapy and to guide reimbursement, but they are rarely sufficiently calibrated and discriminatory to direct individual patient decisions, according to Dr. Anelechi Anyanwu.
"Risk models are designed as tools to define risks of a population and not risks in the individual patient," Dr. Anyanwu said at the Heart Valve Summit 2011.
Dr. Anyanwu, a cardiothoracic surgeon at Mount Sinai Medical Center in New York City, ran through a checklist of what’s missing in today’s risk scoring systems, starting with the absence of all relevant risk factors. Models can adjust only for known risk factors or things that can be measured easily and objectively, but don’t adjust for unproven, unknown, or rare risk factors. "The reality is that most of the variance in outcome is not explained," he said.
Even such known and probable risk factors as poor lung reserve, unclampable aorta, cirrhosis, frailty, radiation, and socioeconomic status are not included in the scoring systems, but almost certainly influence outcome. A study of 230 patients undergoing cardiac surgery after thoracic radiation found worse in-hospital mortality, respiratory complications, and 4-year survival among those receiving extensive radiation vs. variable or tangential radiation – even though the extensively irradiated group was the youngest at 51 years vs. 64 years and 72 years, respectively (J. Thorac. Cardiovasc. Surg. 2007;133:404-13).
Differential Effect of Risk
Risk systems also don’t allow for the fact that risk factors can vary, or their relevance can disappear over time like the once-worrisome variable of 90% stenosis of the left main artery during primary isolated coronary artery bypass grafting (CABG), Dr. Anyanwu said.
The effect of the same risk fact can also differ in various patient subsets. For example, the EuroSCORE identifies a creatinine level of more than 200 mmol/L as a risk factor, but assumes similar risk for the subsets of creatinine clearance 30 mL/min, creatinine clearance 50 mL/min, and dialysis patients.
"Risk systems are imprecise and need further development to allow for more robust prediction."
Risk systems also assume equal weighting in all geographic regions, he said. The EuroSCORE Study Group reported that the predictive value of its scoring system among the six largest national samples in its database varied from "excellent" in Finland (area under the ROC curve 0.87) to "good" in Spain (AUC 0.74) (Eur. J. Cardiothorac. Surg. 2000;18:27-30).
Certainty
Scoring systems can never be 100% specific, Dr. Anyanwu reminded the audience. Quite often the probability range is broad, with imprecision greater if subjective or poorly defined predictor variables have been included in the model.
A high severity score cannot indicate absolute poor risk, and low severity scores cannot guarantee against poor outcome. "At the end of the day, outcome is always uncertain," he said.
Gaming the System
Patient selection is a powerful risk modification tool that cannot be captured by risk models. The more selective a center or surgeon is, the less the effect a specific risk factor will have, Dr. Anyanwu said. A low ejection fraction of less than 30% denotes poor prognosis in aortic valve replacement (AVR), and is accounted for in all risk systems. But not all low ejection fractions are the same, and surgeons can game the system by cherry-picking the better ones, he said.
He cited a study among 81 consecutive patients with symptomatic, calcified low-flow/low-gradient aortic stenosis that reported an operative mortality of 67% in the subgroup of patients with a mean pressure gradient of 20 mm Hg or less, compared with 16% in patients with an mean pressure gradient of more than 20 mm Hg. Moreover, if a surgeon opted not to perform CABG at the time of the AVR, operative mortality was just 10%, compared with 53% for patients in whom CABG was performed at the time of AVR (J. Am. Coll. Cardiol. 2009;53:1865-73).
Other Considerations
Numerous systems show varied outcome by different geographic regions, centers, and surgeons, and by surgical volume. In one study, the risk-adjusted odds of death among patients undergoing first-time elective surgery for mitral regurgitation was 0.71 at centers with an annual volume of 36-70 mitral operations, 0.74 at centers performing 71-140 mitral operations, and just 0.48 at the highest volume centers doing more than 140 mitral operations per year (Circulation 2007;115:881-7).
Despite such variations, risk scores will predict the same outcome for patient groups that clearly have different outcomes in practice, Dr. Anyanwu said at the meeting, sponsored by the American Association for Thoracic Surgery, American College of Cardiology Foundation, and The Society of Thoracic Surgeons.
He suggested that a good risk system has to be recalibrated frequently, and that further study is needed to allow for incorporation of nonpatient factors in risk prediction.
"Risk systems are imprecise and need further development to allow for more robust prediction," he concluded.
Dr. Anyanwu reported no relevant conflicts of interest.
CHICAGO – Risk scoring systems are increasingly being used to assign an indication for therapy and to guide reimbursement, but they are rarely sufficiently calibrated and discriminatory to direct individual patient decisions, according to Dr. Anelechi Anyanwu.
"Risk models are designed as tools to define risks of a population and not risks in the individual patient," Dr. Anyanwu said at the Heart Valve Summit 2011.
Dr. Anyanwu, a cardiothoracic surgeon at Mount Sinai Medical Center in New York City, ran through a checklist of what’s missing in today’s risk scoring systems, starting with the absence of all relevant risk factors. Models can adjust only for known risk factors or things that can be measured easily and objectively, but don’t adjust for unproven, unknown, or rare risk factors. "The reality is that most of the variance in outcome is not explained," he said.
Even such known and probable risk factors as poor lung reserve, unclampable aorta, cirrhosis, frailty, radiation, and socioeconomic status are not included in the scoring systems, but almost certainly influence outcome. A study of 230 patients undergoing cardiac surgery after thoracic radiation found worse in-hospital mortality, respiratory complications, and 4-year survival among those receiving extensive radiation vs. variable or tangential radiation – even though the extensively irradiated group was the youngest at 51 years vs. 64 years and 72 years, respectively (J. Thorac. Cardiovasc. Surg. 2007;133:404-13).
Differential Effect of Risk
Risk systems also don’t allow for the fact that risk factors can vary, or their relevance can disappear over time like the once-worrisome variable of 90% stenosis of the left main artery during primary isolated coronary artery bypass grafting (CABG), Dr. Anyanwu said.
The effect of the same risk fact can also differ in various patient subsets. For example, the EuroSCORE identifies a creatinine level of more than 200 mmol/L as a risk factor, but assumes similar risk for the subsets of creatinine clearance 30 mL/min, creatinine clearance 50 mL/min, and dialysis patients.
"Risk systems are imprecise and need further development to allow for more robust prediction."
Risk systems also assume equal weighting in all geographic regions, he said. The EuroSCORE Study Group reported that the predictive value of its scoring system among the six largest national samples in its database varied from "excellent" in Finland (area under the ROC curve 0.87) to "good" in Spain (AUC 0.74) (Eur. J. Cardiothorac. Surg. 2000;18:27-30).
Certainty
Scoring systems can never be 100% specific, Dr. Anyanwu reminded the audience. Quite often the probability range is broad, with imprecision greater if subjective or poorly defined predictor variables have been included in the model.
A high severity score cannot indicate absolute poor risk, and low severity scores cannot guarantee against poor outcome. "At the end of the day, outcome is always uncertain," he said.
Gaming the System
Patient selection is a powerful risk modification tool that cannot be captured by risk models. The more selective a center or surgeon is, the less the effect a specific risk factor will have, Dr. Anyanwu said. A low ejection fraction of less than 30% denotes poor prognosis in aortic valve replacement (AVR), and is accounted for in all risk systems. But not all low ejection fractions are the same, and surgeons can game the system by cherry-picking the better ones, he said.
He cited a study among 81 consecutive patients with symptomatic, calcified low-flow/low-gradient aortic stenosis that reported an operative mortality of 67% in the subgroup of patients with a mean pressure gradient of 20 mm Hg or less, compared with 16% in patients with an mean pressure gradient of more than 20 mm Hg. Moreover, if a surgeon opted not to perform CABG at the time of the AVR, operative mortality was just 10%, compared with 53% for patients in whom CABG was performed at the time of AVR (J. Am. Coll. Cardiol. 2009;53:1865-73).
Other Considerations
Numerous systems show varied outcome by different geographic regions, centers, and surgeons, and by surgical volume. In one study, the risk-adjusted odds of death among patients undergoing first-time elective surgery for mitral regurgitation was 0.71 at centers with an annual volume of 36-70 mitral operations, 0.74 at centers performing 71-140 mitral operations, and just 0.48 at the highest volume centers doing more than 140 mitral operations per year (Circulation 2007;115:881-7).
Despite such variations, risk scores will predict the same outcome for patient groups that clearly have different outcomes in practice, Dr. Anyanwu said at the meeting, sponsored by the American Association for Thoracic Surgery, American College of Cardiology Foundation, and The Society of Thoracic Surgeons.
He suggested that a good risk system has to be recalibrated frequently, and that further study is needed to allow for incorporation of nonpatient factors in risk prediction.
"Risk systems are imprecise and need further development to allow for more robust prediction," he concluded.
Dr. Anyanwu reported no relevant conflicts of interest.
EXPERT ANALYSIS FROM THE HEART VALVE SUMMIT 2011
CLEVER Trial: Supervised Exercise, Stenting Beats Medical Therapy for PAD
ORLANDO – In patients with aortoiliac peripheral artery disease, adding supervised exercise to optimal medical therapy offers better treadmill walking performance than does aortoiliac stenting.
On the other hand, aortoiliac stenting plus optimal medical care results in better quality of life than does supervised exercise, according to the 6-month results of the CLEVER (Claudication vs. Endoluminal Revascularization) trial.
The CLEVER trial produced discordant results at 6 months, but also a clear signal that the combination interventions are superior to optimal medical therapy alone.
"This idea that you can go home and walk isn’t, and hasn’t been, effective," coauthor Dr. Alan Hirsch said at the annual scientific sessions of the American Heart Association.
CLEVER is the first prospective, head-to-head, comparative trial of three guideline-recommended interventions: supervised exercise, stenting, and optimal medical therapy. It is also the first trial to be conducted exclusively in patients with aortoiliac peripheral artery disease, a group generally considered ideal for stent revascularization, he said.
CLEVER randomized 111 patients to either optimal medical care, (OMC) including cilostazol (Pletal) 100 mg twice daily, plus advice about home exercise and diet with monthly coordinator contact; OMC plus supervised exercise for 1 hour thrice weekly for 26 weeks; or OMC plus aortoiliac stenting. All patients had moderate to severe claudication.
The patients averaged 64 years of age. Their baseline ankle-brachial index was 0.68, peak walking time was about 5 minutes, and claudication-onset time was 1.5 minutes.
The primary end point of change in peak walking time from baseline to 6 months was just 1.2 minutes in the OMC group, compared with 5.8 minutes in the supervised exercise group and a clinically significant 3.7 minutes in the stenting group, said Dr. Hirsch, director of the vascular medicine program at the University of Minnesota in Minneapolis. The changes were significantly different between supervised exercise and OMC (4.6 minutes), stenting and OMC (2.5 minutes), and supervised exercise and stenting (2.1 minutes).
The change in claudication-onset time from baseline to 6 months was again low with OMC at just 0.7 minutes, compared with 3 minutes with the supervised exercise and 3.6 minutes with stenting. The changes were significantly different between supervised exercise and OMC (2.2 minutes) and stenting, compared with OMC (2.9 minutes), but not between supervised exercise and stenting (0.7 minutes), again suggesting the similarity of the two interventions, he said.
Self-reported quality of life scores on the Walking Impairment Questionnaire and Peripheral Artery Questionnaire improved significantly with supervised exercise plus stenting, compared with OMC for most domains including walking distance, walking speed, stair climbing, physical limitation, social limitation, and summary score. The improvements were significantly greater with stenting than with supervised exercise for three of the four domains that were evaluated on each questionnaire.
Dr. Hirsch said the quality of life finding raises questions that CLEVER was not designed to answer, and he hoped the trial would inspire future research. Ongoing 18-month follow-up is expected to provide greater insight into the relative durability of the treatments, as well as the health economic impact.
Dr. Conte added that reimbursement for stenting is significant and not linked to outcomes, and providers and industry are therefore incentivized to provide invasive treatments.
He called for greater investment in clinical trials in PAD, more trials in intermittent claudication to clarify the best outcome measures that correlate with patient improvement in everyday life, increased pressure on the Centers for Medicare and Medicaid Services to provide reimbursement for supervised exercise programs, and more effective pharmacotherapies in intermittent claudication.
The study, which was led by Dr. Timothy Murphy, medical director of the vascular research center at Brown University in Providence, R.I., was simultaneously published online (Circulation 2011 Nov.16 [doi:10.1161/circulationaha.111.075770]).
The National Heart, Lung, and Blood Institute funded the trial, with additional support from Cordis/Johnson & Johnson, eV3, Boston Scientific, and Otsuka America. Dr. Hirsch reported grant support from Abbott Vascular, Cytokinetics, and Viromed, as well as having consulted for AstraZeneca, Merck, Novartis, and Pozen. Dr. Conte reported that he had no relevant disclosures.
The discordance between the treadmill and quality of life outcomes requires further definition, and may limit the trial’s potential impact on clinical practice, according to Dr. Michael Conte.
"We await longer term data on durability, but most importantly, this trial is unlikely to influence practice unless policies change," he said. "We actually have a lot of evidence that supervised exercise works, but we don’t have reimbursement.
"So until we get reimbursement, we will not be able to offer this to our patients, and the Medicare beneficiaries are the largest group with PAD."
Reimbursement for stenting is significant and not linked to outcomes, and providers and industry are therefore incentivized to provide invasive treatments, he said.
Greater investment in clinical trials in PAD, more trials in intermittent claudication to clarify the best outcome measures that correlate with patient improvement in everyday life, increased pressure on the Centers for Medicare and Medicaid Services to provide reimbursement for supervised exercise programs, and more effective pharmacotherapies in intermittent claudication are needed, he added.
Dr. Conte is codirector of the heart and vascular Center at the University of California, San Francisco. He was the invited discussant for the CLEVER presentation. He reported that he had no relevant disclosures.
The discordance between the treadmill and quality of life outcomes requires further definition, and may limit the trial’s potential impact on clinical practice, according to Dr. Michael Conte.
"We await longer term data on durability, but most importantly, this trial is unlikely to influence practice unless policies change," he said. "We actually have a lot of evidence that supervised exercise works, but we don’t have reimbursement.
"So until we get reimbursement, we will not be able to offer this to our patients, and the Medicare beneficiaries are the largest group with PAD."
Reimbursement for stenting is significant and not linked to outcomes, and providers and industry are therefore incentivized to provide invasive treatments, he said.
Greater investment in clinical trials in PAD, more trials in intermittent claudication to clarify the best outcome measures that correlate with patient improvement in everyday life, increased pressure on the Centers for Medicare and Medicaid Services to provide reimbursement for supervised exercise programs, and more effective pharmacotherapies in intermittent claudication are needed, he added.
Dr. Conte is codirector of the heart and vascular Center at the University of California, San Francisco. He was the invited discussant for the CLEVER presentation. He reported that he had no relevant disclosures.
The discordance between the treadmill and quality of life outcomes requires further definition, and may limit the trial’s potential impact on clinical practice, according to Dr. Michael Conte.
"We await longer term data on durability, but most importantly, this trial is unlikely to influence practice unless policies change," he said. "We actually have a lot of evidence that supervised exercise works, but we don’t have reimbursement.
"So until we get reimbursement, we will not be able to offer this to our patients, and the Medicare beneficiaries are the largest group with PAD."
Reimbursement for stenting is significant and not linked to outcomes, and providers and industry are therefore incentivized to provide invasive treatments, he said.
Greater investment in clinical trials in PAD, more trials in intermittent claudication to clarify the best outcome measures that correlate with patient improvement in everyday life, increased pressure on the Centers for Medicare and Medicaid Services to provide reimbursement for supervised exercise programs, and more effective pharmacotherapies in intermittent claudication are needed, he added.
Dr. Conte is codirector of the heart and vascular Center at the University of California, San Francisco. He was the invited discussant for the CLEVER presentation. He reported that he had no relevant disclosures.
ORLANDO – In patients with aortoiliac peripheral artery disease, adding supervised exercise to optimal medical therapy offers better treadmill walking performance than does aortoiliac stenting.
On the other hand, aortoiliac stenting plus optimal medical care results in better quality of life than does supervised exercise, according to the 6-month results of the CLEVER (Claudication vs. Endoluminal Revascularization) trial.
The CLEVER trial produced discordant results at 6 months, but also a clear signal that the combination interventions are superior to optimal medical therapy alone.
"This idea that you can go home and walk isn’t, and hasn’t been, effective," coauthor Dr. Alan Hirsch said at the annual scientific sessions of the American Heart Association.
CLEVER is the first prospective, head-to-head, comparative trial of three guideline-recommended interventions: supervised exercise, stenting, and optimal medical therapy. It is also the first trial to be conducted exclusively in patients with aortoiliac peripheral artery disease, a group generally considered ideal for stent revascularization, he said.
CLEVER randomized 111 patients to either optimal medical care, (OMC) including cilostazol (Pletal) 100 mg twice daily, plus advice about home exercise and diet with monthly coordinator contact; OMC plus supervised exercise for 1 hour thrice weekly for 26 weeks; or OMC plus aortoiliac stenting. All patients had moderate to severe claudication.
The patients averaged 64 years of age. Their baseline ankle-brachial index was 0.68, peak walking time was about 5 minutes, and claudication-onset time was 1.5 minutes.
The primary end point of change in peak walking time from baseline to 6 months was just 1.2 minutes in the OMC group, compared with 5.8 minutes in the supervised exercise group and a clinically significant 3.7 minutes in the stenting group, said Dr. Hirsch, director of the vascular medicine program at the University of Minnesota in Minneapolis. The changes were significantly different between supervised exercise and OMC (4.6 minutes), stenting and OMC (2.5 minutes), and supervised exercise and stenting (2.1 minutes).
The change in claudication-onset time from baseline to 6 months was again low with OMC at just 0.7 minutes, compared with 3 minutes with the supervised exercise and 3.6 minutes with stenting. The changes were significantly different between supervised exercise and OMC (2.2 minutes) and stenting, compared with OMC (2.9 minutes), but not between supervised exercise and stenting (0.7 minutes), again suggesting the similarity of the two interventions, he said.
Self-reported quality of life scores on the Walking Impairment Questionnaire and Peripheral Artery Questionnaire improved significantly with supervised exercise plus stenting, compared with OMC for most domains including walking distance, walking speed, stair climbing, physical limitation, social limitation, and summary score. The improvements were significantly greater with stenting than with supervised exercise for three of the four domains that were evaluated on each questionnaire.
Dr. Hirsch said the quality of life finding raises questions that CLEVER was not designed to answer, and he hoped the trial would inspire future research. Ongoing 18-month follow-up is expected to provide greater insight into the relative durability of the treatments, as well as the health economic impact.
Dr. Conte added that reimbursement for stenting is significant and not linked to outcomes, and providers and industry are therefore incentivized to provide invasive treatments.
He called for greater investment in clinical trials in PAD, more trials in intermittent claudication to clarify the best outcome measures that correlate with patient improvement in everyday life, increased pressure on the Centers for Medicare and Medicaid Services to provide reimbursement for supervised exercise programs, and more effective pharmacotherapies in intermittent claudication.
The study, which was led by Dr. Timothy Murphy, medical director of the vascular research center at Brown University in Providence, R.I., was simultaneously published online (Circulation 2011 Nov.16 [doi:10.1161/circulationaha.111.075770]).
The National Heart, Lung, and Blood Institute funded the trial, with additional support from Cordis/Johnson & Johnson, eV3, Boston Scientific, and Otsuka America. Dr. Hirsch reported grant support from Abbott Vascular, Cytokinetics, and Viromed, as well as having consulted for AstraZeneca, Merck, Novartis, and Pozen. Dr. Conte reported that he had no relevant disclosures.
ORLANDO – In patients with aortoiliac peripheral artery disease, adding supervised exercise to optimal medical therapy offers better treadmill walking performance than does aortoiliac stenting.
On the other hand, aortoiliac stenting plus optimal medical care results in better quality of life than does supervised exercise, according to the 6-month results of the CLEVER (Claudication vs. Endoluminal Revascularization) trial.
The CLEVER trial produced discordant results at 6 months, but also a clear signal that the combination interventions are superior to optimal medical therapy alone.
"This idea that you can go home and walk isn’t, and hasn’t been, effective," coauthor Dr. Alan Hirsch said at the annual scientific sessions of the American Heart Association.
CLEVER is the first prospective, head-to-head, comparative trial of three guideline-recommended interventions: supervised exercise, stenting, and optimal medical therapy. It is also the first trial to be conducted exclusively in patients with aortoiliac peripheral artery disease, a group generally considered ideal for stent revascularization, he said.
CLEVER randomized 111 patients to either optimal medical care, (OMC) including cilostazol (Pletal) 100 mg twice daily, plus advice about home exercise and diet with monthly coordinator contact; OMC plus supervised exercise for 1 hour thrice weekly for 26 weeks; or OMC plus aortoiliac stenting. All patients had moderate to severe claudication.
The patients averaged 64 years of age. Their baseline ankle-brachial index was 0.68, peak walking time was about 5 minutes, and claudication-onset time was 1.5 minutes.
The primary end point of change in peak walking time from baseline to 6 months was just 1.2 minutes in the OMC group, compared with 5.8 minutes in the supervised exercise group and a clinically significant 3.7 minutes in the stenting group, said Dr. Hirsch, director of the vascular medicine program at the University of Minnesota in Minneapolis. The changes were significantly different between supervised exercise and OMC (4.6 minutes), stenting and OMC (2.5 minutes), and supervised exercise and stenting (2.1 minutes).
The change in claudication-onset time from baseline to 6 months was again low with OMC at just 0.7 minutes, compared with 3 minutes with the supervised exercise and 3.6 minutes with stenting. The changes were significantly different between supervised exercise and OMC (2.2 minutes) and stenting, compared with OMC (2.9 minutes), but not between supervised exercise and stenting (0.7 minutes), again suggesting the similarity of the two interventions, he said.
Self-reported quality of life scores on the Walking Impairment Questionnaire and Peripheral Artery Questionnaire improved significantly with supervised exercise plus stenting, compared with OMC for most domains including walking distance, walking speed, stair climbing, physical limitation, social limitation, and summary score. The improvements were significantly greater with stenting than with supervised exercise for three of the four domains that were evaluated on each questionnaire.
Dr. Hirsch said the quality of life finding raises questions that CLEVER was not designed to answer, and he hoped the trial would inspire future research. Ongoing 18-month follow-up is expected to provide greater insight into the relative durability of the treatments, as well as the health economic impact.
Dr. Conte added that reimbursement for stenting is significant and not linked to outcomes, and providers and industry are therefore incentivized to provide invasive treatments.
He called for greater investment in clinical trials in PAD, more trials in intermittent claudication to clarify the best outcome measures that correlate with patient improvement in everyday life, increased pressure on the Centers for Medicare and Medicaid Services to provide reimbursement for supervised exercise programs, and more effective pharmacotherapies in intermittent claudication.
The study, which was led by Dr. Timothy Murphy, medical director of the vascular research center at Brown University in Providence, R.I., was simultaneously published online (Circulation 2011 Nov.16 [doi:10.1161/circulationaha.111.075770]).
The National Heart, Lung, and Blood Institute funded the trial, with additional support from Cordis/Johnson & Johnson, eV3, Boston Scientific, and Otsuka America. Dr. Hirsch reported grant support from Abbott Vascular, Cytokinetics, and Viromed, as well as having consulted for AstraZeneca, Merck, Novartis, and Pozen. Dr. Conte reported that he had no relevant disclosures.
FROM THE ANNUAL SCIENTIFIC SESSIONS OF THE AMERICAN HEART ASSOCIATION
Major Finding: The 6-month change in peak walking time was 1.2 minutes with optimal medical care alone, compared with 5.8 minutes with OMC plus supervised exercise and 3.7 minutes with OMC plus stenting.
Data Source: A prospective comparative intervention trial in 111 patients with aortoiliac PAD.
Disclosures: The NHLBI funded the trial, with additional support from Cordis/Johnson & Johnson, eV3, Boston Scientific, and Otsuka America. Dr. Hirsch reported grant support from Abbott Vascular, Cytokinetics, and Viromed, as well as having consulted for AstraZeneca, Merck, Novartis, and Pozen.
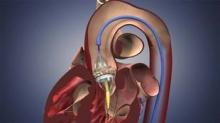
Clinical Use of TAVI Poses Potential Quandaries
CHICAGO – Approval of the first transcatheter aortic valve will transform heart valve repair in the United States and prompt a range of inevitable clinical quandaries, experts suggest.
The Edwards SAPIEN transcatheter valve was approved Nov. 2 for inoperable patients with severe tricuspid aortic stenosis, but already transcatheter aortic valve implantation (TAVI) is being eyed for congenital bicuspid aortic valves and as an alternative strategy for patients with degenerating bioprostheses, Dr. Howard Herrmann said at the 2011 Heart Valve Summit.
The feasibility of TAVI in symptomatic, severe bicuspid aortic valve was recently demonstrated in 11 patients, aged 52-90 years, with one case of malpositioning and late migration at 63 days, but no more aortic insufficiency than in tri-leaflet aortic valves (J. Am. Coll. Cardiol. Intv. 2010;3:1122-25).
"At the moment, young patients are not the ideal candidates for PARTNER or TAVI valves, but by the same token, we do see bicuspid valves in the 80-plus group and I think there are still some unknowns about how to use this device in that setting," said Dr. Herrmann, director of the interventional cardiology program and cardiac catheterization laboratories at the University of Pennsylvania Medical Center in Philadelphia.
Unknowns include the risk of malposition, long-term outcome, aortic insufficiency and the effect of calcification, and anatomy such as bulky leaflets and dilated root.
Successes with "valve-in-valve" implantations have been mostly transapical to date, Dr. Herrmann said at the meeting, cosponsored by the American Association for Thoracic Surgery and American College of Cardiology Foundation. Use of the transapical approach resulted in a success rate of 96% among 24 high-risk patients with failed bioprostheses (Circulation 2010;121;1848-57). The stroke rate was 4% and there were no postprocedure embolizations.
"This has clearly already altered the discussion we have with patients," Dr. Herrmann said. "I see patients in my office all the time who are 50 to 55 [years old] who want a bioprosthetic valve because they don’t want to be on Coumadin, but expect us to figure out how to put a transcatheter valve in them in 15 years.
"There are certainly unknowns, but it’s out there in the community and something we have to think about, so that we can give intelligent answers to these patients."
The overall TAVI landscape is expected to change even more in light of the European approval in recent weeks of two second-generation transapical TAVI devices – JenaValve Technology’s JenaValve both available in three sizes covering aortic annulus diameters from 21 mm to 27 mm.
In November, Medtronic also announced that its CoreValve system, currently limited to investigational use in the United States, has gained approval in Europe as the first transcatheter valve to be delivered using direct aortic access.
Dr. David H. Adams, coprincipal investigator of the CoreValve-Medtronic U.S. Pivotal trial and professor and chair of cardiothoracic surgery at Mount Sinai Medical Center in New York City, said alternative arterial access is being evaluated in both the ongoing CoreValve Extreme Risk and High Risk studies, which has implications because patients treated with apical access in the PARTNER (Placement of AoRTic TraNcathetER Valve) trial had more adverse events.
The use of smaller devices like Medtronic’s 18-French CoreValve prosthesis also has implications for clinical practice because the smaller system can access small femoral arteries and more diseased femoral vessels, he said. Among 126 patients with calcific aortic stenosis, including 54 patients with moderate surgical risk, the overall success rate with the 18-F CoreValve device was 83% and 30-day all-cause mortality 15% (J. Am. Coll. Cardiol. 2011;57:1650-7). All-cause mortality at 2 years was significantly improved among moderate-risk patients, but this was attributed to an increased risk of noncardiac mortality among high-risk patients.
The approval of the Edwards SAPIEN and CoreValve in Europe in 2007 has led to what some describe as a "runaway train" in Europe, where patients and physicians are voting with their feet. TAVI procedures made up just 1.2% of valve procedures in 2007, but are expected to exceed 30% in the first half of 2011 in some countries, Dr. Herrmann said. Symetis, which is launching a 150-patient pivotal trial of its new device in the U.S., estimates that the TAVI market will exceed $2 billion by 2014.
This explosive growth in Europe has been driven by reimbursement, and has resulted in a drift in TAVI usage among lower-risk patients. The mean logistic EuroScore now in some series has fallen below 15% or comparable to a Society of Thoracic Surgery (STS) score of 5%, Dr. Herrmann said.
Dr. Adams cautioned that it will be important to duplicate the rigorous risk assessment process of clinical trials at individual centers and urged clinical estimation of surgical mortality to take into account all incremental risks associated with a patient, as is being done in the CoreValve pivotal trial through the use of an incremental scoring system. For example, a 70-year-old patient with New York Heart Association Class III disease, a history of peripheral vascular disease, pulmonary hypertension, Child’s Class A liver dysfunction and one prior open heart surgery may have a STS mortality risk score of around 8%, but the total incremental mortality risk reaches close to 20% after accounting for the pulmonary hypertension and liver dysfunction, neither of which is part of the STS scoring system at this time.
When audience members asked about the learning curve for TAVI, Dr. Herrmann said it takes a minimum of 10 to 30 cases to even know what you’re doing, and that this number differs as to whether patients are treated with a transfemoral or transapical approach. He noted that Edwards Lifesciences and the U.S. Food and Drug Administration are setting up an intensive training program with simulations, an expert review of cases, and a proctoring system. Only four sites will be designated to train American physicians in the procedure.
Still, the challenge for Edwards and other companies that will follow will be enormous in terms of launching this technology outside the clinical trial setting, Dr. Adams said.
"You cannot overestimate the amount of company support you’re going to need to do these things safely," he said. "This is not a new widget you can pick up and get comfortable with in one or two cases like a new repair ring or new stent ... It’s a whole new process."
Dr. Herrmann reported financial relationships with Abbott, Edwards Lifesciences, Medtronic, St. Jude Medical, and Micro Interventional Devices. Dr. Adams reported royalties as an inventor for Edwards and Medtronic and serving as an uncompensated coprincipal investigator for the Medtronic CoreValve U.S. trial.
CHICAGO – Approval of the first transcatheter aortic valve will transform heart valve repair in the United States and prompt a range of inevitable clinical quandaries, experts suggest.
The Edwards SAPIEN transcatheter valve was approved Nov. 2 for inoperable patients with severe tricuspid aortic stenosis, but already transcatheter aortic valve implantation (TAVI) is being eyed for congenital bicuspid aortic valves and as an alternative strategy for patients with degenerating bioprostheses, Dr. Howard Herrmann said at the 2011 Heart Valve Summit.
The feasibility of TAVI in symptomatic, severe bicuspid aortic valve was recently demonstrated in 11 patients, aged 52-90 years, with one case of malpositioning and late migration at 63 days, but no more aortic insufficiency than in tri-leaflet aortic valves (J. Am. Coll. Cardiol. Intv. 2010;3:1122-25).
"At the moment, young patients are not the ideal candidates for PARTNER or TAVI valves, but by the same token, we do see bicuspid valves in the 80-plus group and I think there are still some unknowns about how to use this device in that setting," said Dr. Herrmann, director of the interventional cardiology program and cardiac catheterization laboratories at the University of Pennsylvania Medical Center in Philadelphia.
Unknowns include the risk of malposition, long-term outcome, aortic insufficiency and the effect of calcification, and anatomy such as bulky leaflets and dilated root.
Successes with "valve-in-valve" implantations have been mostly transapical to date, Dr. Herrmann said at the meeting, cosponsored by the American Association for Thoracic Surgery and American College of Cardiology Foundation. Use of the transapical approach resulted in a success rate of 96% among 24 high-risk patients with failed bioprostheses (Circulation 2010;121;1848-57). The stroke rate was 4% and there were no postprocedure embolizations.
"This has clearly already altered the discussion we have with patients," Dr. Herrmann said. "I see patients in my office all the time who are 50 to 55 [years old] who want a bioprosthetic valve because they don’t want to be on Coumadin, but expect us to figure out how to put a transcatheter valve in them in 15 years.
"There are certainly unknowns, but it’s out there in the community and something we have to think about, so that we can give intelligent answers to these patients."
The overall TAVI landscape is expected to change even more in light of the European approval in recent weeks of two second-generation transapical TAVI devices – JenaValve Technology’s JenaValve both available in three sizes covering aortic annulus diameters from 21 mm to 27 mm.
In November, Medtronic also announced that its CoreValve system, currently limited to investigational use in the United States, has gained approval in Europe as the first transcatheter valve to be delivered using direct aortic access.
Dr. David H. Adams, coprincipal investigator of the CoreValve-Medtronic U.S. Pivotal trial and professor and chair of cardiothoracic surgery at Mount Sinai Medical Center in New York City, said alternative arterial access is being evaluated in both the ongoing CoreValve Extreme Risk and High Risk studies, which has implications because patients treated with apical access in the PARTNER (Placement of AoRTic TraNcathetER Valve) trial had more adverse events.
The use of smaller devices like Medtronic’s 18-French CoreValve prosthesis also has implications for clinical practice because the smaller system can access small femoral arteries and more diseased femoral vessels, he said. Among 126 patients with calcific aortic stenosis, including 54 patients with moderate surgical risk, the overall success rate with the 18-F CoreValve device was 83% and 30-day all-cause mortality 15% (J. Am. Coll. Cardiol. 2011;57:1650-7). All-cause mortality at 2 years was significantly improved among moderate-risk patients, but this was attributed to an increased risk of noncardiac mortality among high-risk patients.
The approval of the Edwards SAPIEN and CoreValve in Europe in 2007 has led to what some describe as a "runaway train" in Europe, where patients and physicians are voting with their feet. TAVI procedures made up just 1.2% of valve procedures in 2007, but are expected to exceed 30% in the first half of 2011 in some countries, Dr. Herrmann said. Symetis, which is launching a 150-patient pivotal trial of its new device in the U.S., estimates that the TAVI market will exceed $2 billion by 2014.
This explosive growth in Europe has been driven by reimbursement, and has resulted in a drift in TAVI usage among lower-risk patients. The mean logistic EuroScore now in some series has fallen below 15% or comparable to a Society of Thoracic Surgery (STS) score of 5%, Dr. Herrmann said.
Dr. Adams cautioned that it will be important to duplicate the rigorous risk assessment process of clinical trials at individual centers and urged clinical estimation of surgical mortality to take into account all incremental risks associated with a patient, as is being done in the CoreValve pivotal trial through the use of an incremental scoring system. For example, a 70-year-old patient with New York Heart Association Class III disease, a history of peripheral vascular disease, pulmonary hypertension, Child’s Class A liver dysfunction and one prior open heart surgery may have a STS mortality risk score of around 8%, but the total incremental mortality risk reaches close to 20% after accounting for the pulmonary hypertension and liver dysfunction, neither of which is part of the STS scoring system at this time.
When audience members asked about the learning curve for TAVI, Dr. Herrmann said it takes a minimum of 10 to 30 cases to even know what you’re doing, and that this number differs as to whether patients are treated with a transfemoral or transapical approach. He noted that Edwards Lifesciences and the U.S. Food and Drug Administration are setting up an intensive training program with simulations, an expert review of cases, and a proctoring system. Only four sites will be designated to train American physicians in the procedure.
Still, the challenge for Edwards and other companies that will follow will be enormous in terms of launching this technology outside the clinical trial setting, Dr. Adams said.
"You cannot overestimate the amount of company support you’re going to need to do these things safely," he said. "This is not a new widget you can pick up and get comfortable with in one or two cases like a new repair ring or new stent ... It’s a whole new process."
Dr. Herrmann reported financial relationships with Abbott, Edwards Lifesciences, Medtronic, St. Jude Medical, and Micro Interventional Devices. Dr. Adams reported royalties as an inventor for Edwards and Medtronic and serving as an uncompensated coprincipal investigator for the Medtronic CoreValve U.S. trial.
CHICAGO – Approval of the first transcatheter aortic valve will transform heart valve repair in the United States and prompt a range of inevitable clinical quandaries, experts suggest.
The Edwards SAPIEN transcatheter valve was approved Nov. 2 for inoperable patients with severe tricuspid aortic stenosis, but already transcatheter aortic valve implantation (TAVI) is being eyed for congenital bicuspid aortic valves and as an alternative strategy for patients with degenerating bioprostheses, Dr. Howard Herrmann said at the 2011 Heart Valve Summit.
The feasibility of TAVI in symptomatic, severe bicuspid aortic valve was recently demonstrated in 11 patients, aged 52-90 years, with one case of malpositioning and late migration at 63 days, but no more aortic insufficiency than in tri-leaflet aortic valves (J. Am. Coll. Cardiol. Intv. 2010;3:1122-25).
"At the moment, young patients are not the ideal candidates for PARTNER or TAVI valves, but by the same token, we do see bicuspid valves in the 80-plus group and I think there are still some unknowns about how to use this device in that setting," said Dr. Herrmann, director of the interventional cardiology program and cardiac catheterization laboratories at the University of Pennsylvania Medical Center in Philadelphia.
Unknowns include the risk of malposition, long-term outcome, aortic insufficiency and the effect of calcification, and anatomy such as bulky leaflets and dilated root.
Successes with "valve-in-valve" implantations have been mostly transapical to date, Dr. Herrmann said at the meeting, cosponsored by the American Association for Thoracic Surgery and American College of Cardiology Foundation. Use of the transapical approach resulted in a success rate of 96% among 24 high-risk patients with failed bioprostheses (Circulation 2010;121;1848-57). The stroke rate was 4% and there were no postprocedure embolizations.
"This has clearly already altered the discussion we have with patients," Dr. Herrmann said. "I see patients in my office all the time who are 50 to 55 [years old] who want a bioprosthetic valve because they don’t want to be on Coumadin, but expect us to figure out how to put a transcatheter valve in them in 15 years.
"There are certainly unknowns, but it’s out there in the community and something we have to think about, so that we can give intelligent answers to these patients."
The overall TAVI landscape is expected to change even more in light of the European approval in recent weeks of two second-generation transapical TAVI devices – JenaValve Technology’s JenaValve both available in three sizes covering aortic annulus diameters from 21 mm to 27 mm.
In November, Medtronic also announced that its CoreValve system, currently limited to investigational use in the United States, has gained approval in Europe as the first transcatheter valve to be delivered using direct aortic access.
Dr. David H. Adams, coprincipal investigator of the CoreValve-Medtronic U.S. Pivotal trial and professor and chair of cardiothoracic surgery at Mount Sinai Medical Center in New York City, said alternative arterial access is being evaluated in both the ongoing CoreValve Extreme Risk and High Risk studies, which has implications because patients treated with apical access in the PARTNER (Placement of AoRTic TraNcathetER Valve) trial had more adverse events.
The use of smaller devices like Medtronic’s 18-French CoreValve prosthesis also has implications for clinical practice because the smaller system can access small femoral arteries and more diseased femoral vessels, he said. Among 126 patients with calcific aortic stenosis, including 54 patients with moderate surgical risk, the overall success rate with the 18-F CoreValve device was 83% and 30-day all-cause mortality 15% (J. Am. Coll. Cardiol. 2011;57:1650-7). All-cause mortality at 2 years was significantly improved among moderate-risk patients, but this was attributed to an increased risk of noncardiac mortality among high-risk patients.
The approval of the Edwards SAPIEN and CoreValve in Europe in 2007 has led to what some describe as a "runaway train" in Europe, where patients and physicians are voting with their feet. TAVI procedures made up just 1.2% of valve procedures in 2007, but are expected to exceed 30% in the first half of 2011 in some countries, Dr. Herrmann said. Symetis, which is launching a 150-patient pivotal trial of its new device in the U.S., estimates that the TAVI market will exceed $2 billion by 2014.
This explosive growth in Europe has been driven by reimbursement, and has resulted in a drift in TAVI usage among lower-risk patients. The mean logistic EuroScore now in some series has fallen below 15% or comparable to a Society of Thoracic Surgery (STS) score of 5%, Dr. Herrmann said.
Dr. Adams cautioned that it will be important to duplicate the rigorous risk assessment process of clinical trials at individual centers and urged clinical estimation of surgical mortality to take into account all incremental risks associated with a patient, as is being done in the CoreValve pivotal trial through the use of an incremental scoring system. For example, a 70-year-old patient with New York Heart Association Class III disease, a history of peripheral vascular disease, pulmonary hypertension, Child’s Class A liver dysfunction and one prior open heart surgery may have a STS mortality risk score of around 8%, but the total incremental mortality risk reaches close to 20% after accounting for the pulmonary hypertension and liver dysfunction, neither of which is part of the STS scoring system at this time.
When audience members asked about the learning curve for TAVI, Dr. Herrmann said it takes a minimum of 10 to 30 cases to even know what you’re doing, and that this number differs as to whether patients are treated with a transfemoral or transapical approach. He noted that Edwards Lifesciences and the U.S. Food and Drug Administration are setting up an intensive training program with simulations, an expert review of cases, and a proctoring system. Only four sites will be designated to train American physicians in the procedure.
Still, the challenge for Edwards and other companies that will follow will be enormous in terms of launching this technology outside the clinical trial setting, Dr. Adams said.
"You cannot overestimate the amount of company support you’re going to need to do these things safely," he said. "This is not a new widget you can pick up and get comfortable with in one or two cases like a new repair ring or new stent ... It’s a whole new process."
Dr. Herrmann reported financial relationships with Abbott, Edwards Lifesciences, Medtronic, St. Jude Medical, and Micro Interventional Devices. Dr. Adams reported royalties as an inventor for Edwards and Medtronic and serving as an uncompensated coprincipal investigator for the Medtronic CoreValve U.S. trial.
EXPERT ANALYSIS FROM THE 2011 HEART VALVE SUMMIT
TBI: Not a Single Disease, but Many
MADISON, WIS. – The one-size-fits-all approach to traumatic brain injury may be facing the same demise observed in the management and treatment of cancer and its protean forms.
"One of the arguments for why traumatic brain injury has been so difficult to get anything to work in a clinical trial is that we’ve been really naive in terms of trying to consider it one disease," said Dr. Patrick Kochanek, director of the Safar Center for Resuscitation Research at the University of Pittsburgh. "People have argued that we are treating traumatic brain injury as if we were treating cancer as a single disease. Maybe we need to do classifications."
To punctuate the point, Dr. Kochanek highlighted the now-classic computed tomography slide by the renowned neurosurgeon Dr. Geoffrey Manley, vice chair of neurological surgery at University of California, San Francisco, that illustrates six unique paths to a Glasgow Coma Scale score of four in traumatic brain injury (TBI) – epidural hematoma, contusion/hematoma, diffuse axonal injury, subdural hematoma, subarachnoid/intraventricular hemorrhages, and diffuse swelling.
The need to target distinct TBI subgroups was further illustrated by results from the recent NABISH II (National Acute Brain Injury Study: Hypothermia II), which failed to confirm the overall benefit of early hypothermia as a neuroprotectant among 232 patients with severe TBI. Subgroup analyses, however, revealed a significantly improved outcome in patients with hematomas treated with hypothermia, while those with diffuse brain injuries appeared to do worse (Lancet Neurol. 2011;10:131-9).
Pediatric considerations only further magnify the challenge of translating neuroprotective therapies for TBI to clinical practice, Dr. Kochanek said at the annual meeting of the Midwest Society for Pediatric Research. Diffuse cerebral swelling, the most common cause of brain death following severe TBI in both adults and children, is 3.5 times more common in children than in adults and typically more common in younger age groups than in older children.
Hypothermia has been shown to reduce intracranial pressure after pediatric TBI and to be beneficial in pediatric cardiac arrest and hypoxic-ischemic encephalopathy (HIE). Still, a lack of quality data demonstrating a consistent benefit has contributed to the lack of a recommendation of prophylactic hypothermia in pediatric severe head trauma management. Hopes for additional, high-quality data were dashed after the $11.5 million-dollar, multicenter Cool Kids trial evaluating 48 hours of hypothermia within 6 hours of injury recently was stopped for futility.
It’s possible that 6 hours is too late for a therapeutic window or that patients are rewarmed too early during peak edema or too fast, thereby exacerbating hypotension, or that the brain responds differently to hypothermia following TBI versus HIE or cardiac arrest, Dr. Kochanek suggested.
A biomarker trial led by colleague Dr. Rachel Berger used serial serum neuron-specific enolase measurements to demonstrate that neuronal death is frequently delayed in HIE, but predominantly acute in children with TBI (Dev. Neurosci. 2006;28:327-35). This may explain why hypothermia and other therapies have not been effective other than to reduce intracranial pressure after TBI, he said.
"The take-home message is that if you are targeting neuroprotection in TBI, you better get the therapy on board fast," he added.
Better monitoring and management of fever after brain injury also is needed. Notably, the odds of death or disability increased 3.6- to fourfold for every single degree Celsius increase in a study involving 99 infants with HIE (Pediatrics 2008;122:491-9).
"We do a poor job of dealing with this issue," Dr. Kochanek said. "Just go through the ICU, particularly a pediatric ICU, and take a look at the temperature recordings of patients with head injury or cardiac arrest, and you’ll be pretty much disappointed."
The lack of surface cooling devices specifically designed for children or endovascular cooling catheters for pediatric patients prompted colleagues, led by Dr. Ericka Fink, to use iced intravenous saline 20 mL/kg infused over 15 minutes to treat fever among 18 children, aged 1 week to 17 years, with TBI, intracranial hemorrhage, or cardiac arrest. The treatment resulted in a significant reduction in core temperature after infusion for fever and a trend toward lower temperatures for induction of hypothermia (Pediatr. Crit. Care Med. 2010 [doi: 10.1097/PCC.0b013e3181fe27c7]).
"People have argued that we are treating traumatic brain injury as if we were treating cancer as a single disease."
What is uncertain is how long the brain remains vulnerable to fever after an injury, Dr. Kochanek said. "It’s one thing to be comatose and aggressively cool to control fever, and it’s quite another to do this 5 days out when you’re awake and your stress response to cooling and need for sedation become a real problem," he observed.
Much of the emphasis in treating pediatric TBI patients has been on reducing secondary insult through the use of intracranial pressure (ICP) monitoring and osmolar therapy, but this may not be enough. A retrospective analysis from the University of Pittsburgh team reported a 50% incidence of unfavorable outcome among 22 infants with severe TBI, despite nearly 900 hourly ICP readings in a PICU with rigorous ICP control and an excellent 8.6% severe TBI mortality rate (Dev. Neurosci. 2010;32:413-9).
"In many cases, we may need to identify other therapeutic targets," he said, adding that axonal injury may offer a temporally friendly therapeutic target in both TBI and abusive head trauma.
Finally, Dr. Kochanek suggested that abusive head trauma should be established as a separate entity from accidental TBI. He pointed out that the biomarker study, cited above, also found that patients with abusive head trauma demonstrate considerable delayed neuronal death, mirroring HIE more than TBI.
"Abusive head trauma is a separate entity and deserves its own therapeutic targets," he said.
Dr. Kochanek reported funding from the U.S. Army, the National Institute of Child Health and Human Development, the National Institute of Neurological Disorders and Stroke, the Defense Advanced Research Projects Agency, the Laerdal Foundation for Acute Medicine, and the American Heart Association.
MADISON, WIS. – The one-size-fits-all approach to traumatic brain injury may be facing the same demise observed in the management and treatment of cancer and its protean forms.
"One of the arguments for why traumatic brain injury has been so difficult to get anything to work in a clinical trial is that we’ve been really naive in terms of trying to consider it one disease," said Dr. Patrick Kochanek, director of the Safar Center for Resuscitation Research at the University of Pittsburgh. "People have argued that we are treating traumatic brain injury as if we were treating cancer as a single disease. Maybe we need to do classifications."
To punctuate the point, Dr. Kochanek highlighted the now-classic computed tomography slide by the renowned neurosurgeon Dr. Geoffrey Manley, vice chair of neurological surgery at University of California, San Francisco, that illustrates six unique paths to a Glasgow Coma Scale score of four in traumatic brain injury (TBI) – epidural hematoma, contusion/hematoma, diffuse axonal injury, subdural hematoma, subarachnoid/intraventricular hemorrhages, and diffuse swelling.
The need to target distinct TBI subgroups was further illustrated by results from the recent NABISH II (National Acute Brain Injury Study: Hypothermia II), which failed to confirm the overall benefit of early hypothermia as a neuroprotectant among 232 patients with severe TBI. Subgroup analyses, however, revealed a significantly improved outcome in patients with hematomas treated with hypothermia, while those with diffuse brain injuries appeared to do worse (Lancet Neurol. 2011;10:131-9).
Pediatric considerations only further magnify the challenge of translating neuroprotective therapies for TBI to clinical practice, Dr. Kochanek said at the annual meeting of the Midwest Society for Pediatric Research. Diffuse cerebral swelling, the most common cause of brain death following severe TBI in both adults and children, is 3.5 times more common in children than in adults and typically more common in younger age groups than in older children.
Hypothermia has been shown to reduce intracranial pressure after pediatric TBI and to be beneficial in pediatric cardiac arrest and hypoxic-ischemic encephalopathy (HIE). Still, a lack of quality data demonstrating a consistent benefit has contributed to the lack of a recommendation of prophylactic hypothermia in pediatric severe head trauma management. Hopes for additional, high-quality data were dashed after the $11.5 million-dollar, multicenter Cool Kids trial evaluating 48 hours of hypothermia within 6 hours of injury recently was stopped for futility.
It’s possible that 6 hours is too late for a therapeutic window or that patients are rewarmed too early during peak edema or too fast, thereby exacerbating hypotension, or that the brain responds differently to hypothermia following TBI versus HIE or cardiac arrest, Dr. Kochanek suggested.
A biomarker trial led by colleague Dr. Rachel Berger used serial serum neuron-specific enolase measurements to demonstrate that neuronal death is frequently delayed in HIE, but predominantly acute in children with TBI (Dev. Neurosci. 2006;28:327-35). This may explain why hypothermia and other therapies have not been effective other than to reduce intracranial pressure after TBI, he said.
"The take-home message is that if you are targeting neuroprotection in TBI, you better get the therapy on board fast," he added.
Better monitoring and management of fever after brain injury also is needed. Notably, the odds of death or disability increased 3.6- to fourfold for every single degree Celsius increase in a study involving 99 infants with HIE (Pediatrics 2008;122:491-9).
"We do a poor job of dealing with this issue," Dr. Kochanek said. "Just go through the ICU, particularly a pediatric ICU, and take a look at the temperature recordings of patients with head injury or cardiac arrest, and you’ll be pretty much disappointed."
The lack of surface cooling devices specifically designed for children or endovascular cooling catheters for pediatric patients prompted colleagues, led by Dr. Ericka Fink, to use iced intravenous saline 20 mL/kg infused over 15 minutes to treat fever among 18 children, aged 1 week to 17 years, with TBI, intracranial hemorrhage, or cardiac arrest. The treatment resulted in a significant reduction in core temperature after infusion for fever and a trend toward lower temperatures for induction of hypothermia (Pediatr. Crit. Care Med. 2010 [doi: 10.1097/PCC.0b013e3181fe27c7]).
"People have argued that we are treating traumatic brain injury as if we were treating cancer as a single disease."
What is uncertain is how long the brain remains vulnerable to fever after an injury, Dr. Kochanek said. "It’s one thing to be comatose and aggressively cool to control fever, and it’s quite another to do this 5 days out when you’re awake and your stress response to cooling and need for sedation become a real problem," he observed.
Much of the emphasis in treating pediatric TBI patients has been on reducing secondary insult through the use of intracranial pressure (ICP) monitoring and osmolar therapy, but this may not be enough. A retrospective analysis from the University of Pittsburgh team reported a 50% incidence of unfavorable outcome among 22 infants with severe TBI, despite nearly 900 hourly ICP readings in a PICU with rigorous ICP control and an excellent 8.6% severe TBI mortality rate (Dev. Neurosci. 2010;32:413-9).
"In many cases, we may need to identify other therapeutic targets," he said, adding that axonal injury may offer a temporally friendly therapeutic target in both TBI and abusive head trauma.
Finally, Dr. Kochanek suggested that abusive head trauma should be established as a separate entity from accidental TBI. He pointed out that the biomarker study, cited above, also found that patients with abusive head trauma demonstrate considerable delayed neuronal death, mirroring HIE more than TBI.
"Abusive head trauma is a separate entity and deserves its own therapeutic targets," he said.
Dr. Kochanek reported funding from the U.S. Army, the National Institute of Child Health and Human Development, the National Institute of Neurological Disorders and Stroke, the Defense Advanced Research Projects Agency, the Laerdal Foundation for Acute Medicine, and the American Heart Association.
MADISON, WIS. – The one-size-fits-all approach to traumatic brain injury may be facing the same demise observed in the management and treatment of cancer and its protean forms.
"One of the arguments for why traumatic brain injury has been so difficult to get anything to work in a clinical trial is that we’ve been really naive in terms of trying to consider it one disease," said Dr. Patrick Kochanek, director of the Safar Center for Resuscitation Research at the University of Pittsburgh. "People have argued that we are treating traumatic brain injury as if we were treating cancer as a single disease. Maybe we need to do classifications."
To punctuate the point, Dr. Kochanek highlighted the now-classic computed tomography slide by the renowned neurosurgeon Dr. Geoffrey Manley, vice chair of neurological surgery at University of California, San Francisco, that illustrates six unique paths to a Glasgow Coma Scale score of four in traumatic brain injury (TBI) – epidural hematoma, contusion/hematoma, diffuse axonal injury, subdural hematoma, subarachnoid/intraventricular hemorrhages, and diffuse swelling.
The need to target distinct TBI subgroups was further illustrated by results from the recent NABISH II (National Acute Brain Injury Study: Hypothermia II), which failed to confirm the overall benefit of early hypothermia as a neuroprotectant among 232 patients with severe TBI. Subgroup analyses, however, revealed a significantly improved outcome in patients with hematomas treated with hypothermia, while those with diffuse brain injuries appeared to do worse (Lancet Neurol. 2011;10:131-9).
Pediatric considerations only further magnify the challenge of translating neuroprotective therapies for TBI to clinical practice, Dr. Kochanek said at the annual meeting of the Midwest Society for Pediatric Research. Diffuse cerebral swelling, the most common cause of brain death following severe TBI in both adults and children, is 3.5 times more common in children than in adults and typically more common in younger age groups than in older children.
Hypothermia has been shown to reduce intracranial pressure after pediatric TBI and to be beneficial in pediatric cardiac arrest and hypoxic-ischemic encephalopathy (HIE). Still, a lack of quality data demonstrating a consistent benefit has contributed to the lack of a recommendation of prophylactic hypothermia in pediatric severe head trauma management. Hopes for additional, high-quality data were dashed after the $11.5 million-dollar, multicenter Cool Kids trial evaluating 48 hours of hypothermia within 6 hours of injury recently was stopped for futility.
It’s possible that 6 hours is too late for a therapeutic window or that patients are rewarmed too early during peak edema or too fast, thereby exacerbating hypotension, or that the brain responds differently to hypothermia following TBI versus HIE or cardiac arrest, Dr. Kochanek suggested.
A biomarker trial led by colleague Dr. Rachel Berger used serial serum neuron-specific enolase measurements to demonstrate that neuronal death is frequently delayed in HIE, but predominantly acute in children with TBI (Dev. Neurosci. 2006;28:327-35). This may explain why hypothermia and other therapies have not been effective other than to reduce intracranial pressure after TBI, he said.
"The take-home message is that if you are targeting neuroprotection in TBI, you better get the therapy on board fast," he added.
Better monitoring and management of fever after brain injury also is needed. Notably, the odds of death or disability increased 3.6- to fourfold for every single degree Celsius increase in a study involving 99 infants with HIE (Pediatrics 2008;122:491-9).
"We do a poor job of dealing with this issue," Dr. Kochanek said. "Just go through the ICU, particularly a pediatric ICU, and take a look at the temperature recordings of patients with head injury or cardiac arrest, and you’ll be pretty much disappointed."
The lack of surface cooling devices specifically designed for children or endovascular cooling catheters for pediatric patients prompted colleagues, led by Dr. Ericka Fink, to use iced intravenous saline 20 mL/kg infused over 15 minutes to treat fever among 18 children, aged 1 week to 17 years, with TBI, intracranial hemorrhage, or cardiac arrest. The treatment resulted in a significant reduction in core temperature after infusion for fever and a trend toward lower temperatures for induction of hypothermia (Pediatr. Crit. Care Med. 2010 [doi: 10.1097/PCC.0b013e3181fe27c7]).
"People have argued that we are treating traumatic brain injury as if we were treating cancer as a single disease."
What is uncertain is how long the brain remains vulnerable to fever after an injury, Dr. Kochanek said. "It’s one thing to be comatose and aggressively cool to control fever, and it’s quite another to do this 5 days out when you’re awake and your stress response to cooling and need for sedation become a real problem," he observed.
Much of the emphasis in treating pediatric TBI patients has been on reducing secondary insult through the use of intracranial pressure (ICP) monitoring and osmolar therapy, but this may not be enough. A retrospective analysis from the University of Pittsburgh team reported a 50% incidence of unfavorable outcome among 22 infants with severe TBI, despite nearly 900 hourly ICP readings in a PICU with rigorous ICP control and an excellent 8.6% severe TBI mortality rate (Dev. Neurosci. 2010;32:413-9).
"In many cases, we may need to identify other therapeutic targets," he said, adding that axonal injury may offer a temporally friendly therapeutic target in both TBI and abusive head trauma.
Finally, Dr. Kochanek suggested that abusive head trauma should be established as a separate entity from accidental TBI. He pointed out that the biomarker study, cited above, also found that patients with abusive head trauma demonstrate considerable delayed neuronal death, mirroring HIE more than TBI.
"Abusive head trauma is a separate entity and deserves its own therapeutic targets," he said.
Dr. Kochanek reported funding from the U.S. Army, the National Institute of Child Health and Human Development, the National Institute of Neurological Disorders and Stroke, the Defense Advanced Research Projects Agency, the Laerdal Foundation for Acute Medicine, and the American Heart Association.
EXPERT ANALYSIS FROM THE ANNUAL MEETING OF THE MIDWEST SOCIETY FOR PEDIATRIC RESEARCH
OSHA Denial Roils Resident Work-Hour Reformists
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
OSHA Denial Roils Resident Work-Hour Reformists
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
OSHA Denial Roils Resident Work-Hour Reformists
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
Reaction has been mixed to the Occupational Safety and Health Administration’s recent decision to deny a second petition from the Public Citizen Health Research Group and other groups to have OSHA, rather than the Accreditation Council for Graduate Medical Education, regulate resident/fellow work hours.
The American Medical Association, which had worked to keep ACGME at the helm, applauded OSHA’s decision in a recent statement.
“The ACGME is the appropriate body to regulate and monitor resident duty hours, as it is optimally suited to oversee resident and fellow physician duty hours on behalf of both the profession and the public,” AMA president Dr. Peter Carmel said. “We are pleased that OSHA agrees.”
In denying the petition, OSHA officials wrote that resident duty hour standards are "best addressed within the context of resident training and education," and that new duty hour standards and enforcement mechanisms that took effect in July 2011 “provide an opportunity for ACGME to take meaningful steps to protect the health of resident physicians within the context of their overall residency experience.”
OSHA officials also noted that federal whistleblowers provisions protect residents and interns who voice concerns related to extended work hours.
Public Citizen fired back in a letter to OSHA that the Obama administration was rehashing “the same discredited Bush-era arguments of nine years ago when our first petition was rejected on almost identical grounds.”
The group went on to say that “OSHA has, once again, opted out of its legal obligation to protect residents from excessive work hours, deferring instead to a largely unaccountable private entity, the ACGME.”
Currently, when a resident reports work-hour violations, they risk retaliation from colleagues and put their training programs at risk for probation and even loss of accreditation, Sonia Lazreg, health justice fellow with the American Medical Student Association, said in an interview.
After the 2003 work rules were implemented, more than 80% of residents reported that their programs were in violation when they could report anonymously to an external body, she said. During the same period, ACGME resident survey reporting suggested that only 3% of programs were in violation.
“Only when we have external enforcement, beyond the ACGME, will we see true implementation of duty hours,” she added.
The AMSA co-petitioned OSHA based on what Lazreg described as overwhelming evidence that current schedules cause an increase in mood disorders, motor vehicle accidents, pregnancy complications and needle-stick injuries among residents. As the federal body tasked with ensuring employee safety and health, she said OSHA has a responsibility to intervene when evidence so strongly points to worker harm.
“OSHA’s denial translates into continued employee risk, injury and death,” Lazreg said.
With so much on the line, it’s unlikely that either side will give ground on this contentious issue any time soon. Notably, OSHA said it had received 15 letters in support of OSHA regulating resident hours and 26 letters in opposition.
Where do you fit it? Let us know.
By Patrice Wendling
Many Neonatal PICC Lines Migrate 24 Hours After Insertion
CHICAGO – A full 45% of all peripherally inserted central catheter lines placed in the basilic or cephalic veins migrated 24 hours after insertion in a retrospective analysis of 100 consecutive lines placed in a neonatal intensive care unit.
Of the 76 peripherally inserted central catheter (PICC) lines placed in the basilic vein, 35.5% migrated inferiorly and medially (mean 1.48 cm), 14.5% migrated laterally (mean 1.74 cm), and 50% did not change position.
Of the 19 PICC lines placed in the cephalic vein, 21% migrated inferiorly and medially (mean 1.36 cm), 15.7% migrated laterally (mean 1.75 cm), and 63.3% did not change position.
None of the five PICC lines placed in the saphenous veins migrated, lead author Dr. Ansel Tjin-A-Tam reported at the annual meeting of the Midwest Society for Pediatric Research.
"We’re not sure why they migrated," he said.
In light of the findings and the potential for catheter migration to result in neonatal death due to cardiac tamponade, he recommended that all neonates have x-rays repeated at 24 hours post-insertion and PICC lines adjusted accordingly.
The chart review involved 100 consecutively placed PICC lines in the NICU at Mount Sinai Hospital in Chicago from January 2010 to March 31. All PICC lines were placed by two certified NICU nurses. X-rays were obtained immediately after insertion and 24 hours post-insertion, and reviewed by a single board-certified pediatric radiologist. The position of the arm was adducted and internally rotated at the shoulder and extended at the elbow on all the x-rays, explained Dr. Tjin-A-Tam, a third-year resident at the hospital.
A recent literature review of studies evaluating the best method to confirm PICC placement in neonates, identified a paucity of information on the subject and the importance of arm position when performing radiographs because movement of the arm can cause migration of the catheter (Neonatal Netw. 2010;29:23-35). The authors suggest supplementing supine chest radiographs with contrast or ultrasound when the line tip position is unclear, and that supine and lateral abdominal radiographs are useful to ensure placement in the inferior vena cava when placing a PICC in the saphenous vein of a neonate.
Dr. Tjin-A-Tam said all of the patients in the study were supine at the time of the radiograph, and agreed that ultrasound could be useful during placement, particularly given concerns for radiation exposure in neonates.
In the current analysis, the neonates’ average birth weight was 1,266 grams (range 420-3,795 g), and the mean dwell time of the PICC lines was 30 days (range 1-45 days).
A recently published review found that 164 (14%) of 1,148 PICC insertions in a NICU had to be replaced due to blockage or migration, and that those exchanged by using the old PICC as a guide wire were associated with a significantly higher risk of central line-associated blood stream infections, compared with those inserted into a new site (9.8% vs. 1%). This association remained significant even after adjusting for confounders (Am. J. Perinatol. 2011;28:419-24).
Dr. Tjin-A-Tam said he was unaware of any infections during the study period.
"If the radiologist shows that it has migrated, they don’t replace the whole PICC line, they just adjust it," he said. "If there is an infection, obviously you have to take the line out."
Dr. Tjin-A-Tam and his coauthors reported no relevant financial disclosures.
saphenous veins, Dr. Ansel Tjin-A-Tam, the Midwest Society for Pediatric Research, NICU, pediatric radiologist,
CHICAGO – A full 45% of all peripherally inserted central catheter lines placed in the basilic or cephalic veins migrated 24 hours after insertion in a retrospective analysis of 100 consecutive lines placed in a neonatal intensive care unit.
Of the 76 peripherally inserted central catheter (PICC) lines placed in the basilic vein, 35.5% migrated inferiorly and medially (mean 1.48 cm), 14.5% migrated laterally (mean 1.74 cm), and 50% did not change position.
Of the 19 PICC lines placed in the cephalic vein, 21% migrated inferiorly and medially (mean 1.36 cm), 15.7% migrated laterally (mean 1.75 cm), and 63.3% did not change position.
None of the five PICC lines placed in the saphenous veins migrated, lead author Dr. Ansel Tjin-A-Tam reported at the annual meeting of the Midwest Society for Pediatric Research.
"We’re not sure why they migrated," he said.
In light of the findings and the potential for catheter migration to result in neonatal death due to cardiac tamponade, he recommended that all neonates have x-rays repeated at 24 hours post-insertion and PICC lines adjusted accordingly.
The chart review involved 100 consecutively placed PICC lines in the NICU at Mount Sinai Hospital in Chicago from January 2010 to March 31. All PICC lines were placed by two certified NICU nurses. X-rays were obtained immediately after insertion and 24 hours post-insertion, and reviewed by a single board-certified pediatric radiologist. The position of the arm was adducted and internally rotated at the shoulder and extended at the elbow on all the x-rays, explained Dr. Tjin-A-Tam, a third-year resident at the hospital.
A recent literature review of studies evaluating the best method to confirm PICC placement in neonates, identified a paucity of information on the subject and the importance of arm position when performing radiographs because movement of the arm can cause migration of the catheter (Neonatal Netw. 2010;29:23-35). The authors suggest supplementing supine chest radiographs with contrast or ultrasound when the line tip position is unclear, and that supine and lateral abdominal radiographs are useful to ensure placement in the inferior vena cava when placing a PICC in the saphenous vein of a neonate.
Dr. Tjin-A-Tam said all of the patients in the study were supine at the time of the radiograph, and agreed that ultrasound could be useful during placement, particularly given concerns for radiation exposure in neonates.
In the current analysis, the neonates’ average birth weight was 1,266 grams (range 420-3,795 g), and the mean dwell time of the PICC lines was 30 days (range 1-45 days).
A recently published review found that 164 (14%) of 1,148 PICC insertions in a NICU had to be replaced due to blockage or migration, and that those exchanged by using the old PICC as a guide wire were associated with a significantly higher risk of central line-associated blood stream infections, compared with those inserted into a new site (9.8% vs. 1%). This association remained significant even after adjusting for confounders (Am. J. Perinatol. 2011;28:419-24).
Dr. Tjin-A-Tam said he was unaware of any infections during the study period.
"If the radiologist shows that it has migrated, they don’t replace the whole PICC line, they just adjust it," he said. "If there is an infection, obviously you have to take the line out."
Dr. Tjin-A-Tam and his coauthors reported no relevant financial disclosures.
CHICAGO – A full 45% of all peripherally inserted central catheter lines placed in the basilic or cephalic veins migrated 24 hours after insertion in a retrospective analysis of 100 consecutive lines placed in a neonatal intensive care unit.
Of the 76 peripherally inserted central catheter (PICC) lines placed in the basilic vein, 35.5% migrated inferiorly and medially (mean 1.48 cm), 14.5% migrated laterally (mean 1.74 cm), and 50% did not change position.
Of the 19 PICC lines placed in the cephalic vein, 21% migrated inferiorly and medially (mean 1.36 cm), 15.7% migrated laterally (mean 1.75 cm), and 63.3% did not change position.
None of the five PICC lines placed in the saphenous veins migrated, lead author Dr. Ansel Tjin-A-Tam reported at the annual meeting of the Midwest Society for Pediatric Research.
"We’re not sure why they migrated," he said.
In light of the findings and the potential for catheter migration to result in neonatal death due to cardiac tamponade, he recommended that all neonates have x-rays repeated at 24 hours post-insertion and PICC lines adjusted accordingly.
The chart review involved 100 consecutively placed PICC lines in the NICU at Mount Sinai Hospital in Chicago from January 2010 to March 31. All PICC lines were placed by two certified NICU nurses. X-rays were obtained immediately after insertion and 24 hours post-insertion, and reviewed by a single board-certified pediatric radiologist. The position of the arm was adducted and internally rotated at the shoulder and extended at the elbow on all the x-rays, explained Dr. Tjin-A-Tam, a third-year resident at the hospital.
A recent literature review of studies evaluating the best method to confirm PICC placement in neonates, identified a paucity of information on the subject and the importance of arm position when performing radiographs because movement of the arm can cause migration of the catheter (Neonatal Netw. 2010;29:23-35). The authors suggest supplementing supine chest radiographs with contrast or ultrasound when the line tip position is unclear, and that supine and lateral abdominal radiographs are useful to ensure placement in the inferior vena cava when placing a PICC in the saphenous vein of a neonate.
Dr. Tjin-A-Tam said all of the patients in the study were supine at the time of the radiograph, and agreed that ultrasound could be useful during placement, particularly given concerns for radiation exposure in neonates.
In the current analysis, the neonates’ average birth weight was 1,266 grams (range 420-3,795 g), and the mean dwell time of the PICC lines was 30 days (range 1-45 days).
A recently published review found that 164 (14%) of 1,148 PICC insertions in a NICU had to be replaced due to blockage or migration, and that those exchanged by using the old PICC as a guide wire were associated with a significantly higher risk of central line-associated blood stream infections, compared with those inserted into a new site (9.8% vs. 1%). This association remained significant even after adjusting for confounders (Am. J. Perinatol. 2011;28:419-24).
Dr. Tjin-A-Tam said he was unaware of any infections during the study period.
"If the radiologist shows that it has migrated, they don’t replace the whole PICC line, they just adjust it," he said. "If there is an infection, obviously you have to take the line out."
Dr. Tjin-A-Tam and his coauthors reported no relevant financial disclosures.
saphenous veins, Dr. Ansel Tjin-A-Tam, the Midwest Society for Pediatric Research, NICU, pediatric radiologist,
saphenous veins, Dr. Ansel Tjin-A-Tam, the Midwest Society for Pediatric Research, NICU, pediatric radiologist,
FROM THE ANNUAL MEETING OF THE MIDWEST SOCIETY FOR PEDIATRIC RESEARCH
Major Finding: A full 45% of all PICC lines placed in the basilic or cephalic veins of neonates migrated 24 hours after insertion.
Data Source: Retrospective analysis of 100 consecutive PICC lines placed in an NICU.
Disclosures: Dr. Tjin-A-Tam and his co-authors reported no relevant financial disclosures.
Genotyping Guides Clopidogrel Dosing in CVD
ORLANDO – Genotyping can provide more targeted use of the commonly prescribed drug clopidogrel among patients with cardiovascular disease, according to data from the ELEVATE-TIMI 56 trial.
"We’re at the point that clopidogrel may not be a one-size-fits-all drug for our patients," Dr. Jessica Mega said at the annual scientific sessions of the American Heart Association.
She reported on the prospective ELEVATE-TIMI 56 trial involving 333 patients with stable cardiovascular disease who underwent genotyping for alleles of CYP2C19, which is primarily responsible for producing active metabolite from clopidogrel (Plavix). Research has shown that patients who carry nonfunctional alleles of CYP2C19 such as *2 have reduced plasma active metabolite levels, an increased risk of ex vivo residual platelet activity, and a greater risk of cardiac events such as stent thrombosis.
Based on the genotyping results, 86 carriers (80 heterozygotes and 6 homozygotes) of the CYP2C19*2 allele were randomized to daily clopidogrel doses of 75 mg, 150 mg, 225 mg, and 300 mg for four 14-day treatment periods, while 247 CYP2C19*2 noncarriers were assigned to daily clopidogrel 75 mg and 150 mg for two 14-day periods each.
Platelet testing at the end of each treatment period revealed that both CYP2C19*2 heterozygotes and homozygotes had significantly higher platelet reactivity than did noncarriers, said Dr. Mega, a cardiologist at Brigham and Women’s Hospital, Boston.
The mean vasodilator-stimulated phosphoprotein phosphorylation (VASP) platelet reactivity index (PRI) was 70% for carriers vs. 58% for noncarriers, while mean P2Y12 reaction units (PRUs) were 226 vs. 164, respectively.
Among CYP2C19*2 heterozygotes, tripling the daily maintenance dose of clopidogrel to 225 mg was needed to reduce platelet reactivity to the levels achieved with a standard 75-mg dose in noncarriers, she said. Specifically, the percentage of CYP2C19*2 heterozygotes who did not respond to standard dosing was reduced from 52% to 26% with a 150-mg dose, and to 10% with at least 225 mg of clopidogrel.
Among CYP2C19*2 homozygotes, who made up 2% of the study population, even 300 mg, or four times the standard dose, of clopidogrel did not result in optimal degrees of platelet inhibition.
Dr. Mega said alternative agents with higher degrees of platelet inhibition are available, but that clopidogrel will become generic next year and continues to be used in a significant number of patients.
"It’s good news for patients that we’re able to optimize their [platelet] inhibition with clopidogrel," she said. "I think we need to get creative in how we use clopidogrel, and this study is one step in the right direction."
Invited discussant Lawrence Lesko, Ph.D., director of the center for pharmacometrics and systems pharmacology at the University of Florida in Lake Nona, pointed out that clopidogrel carries a "black box" warning indicating that poor metabolizers are at heightened risk for cardiovascular events, and that ELEVATE-TIMI 56 fills in some of the gaps in the labeling of what to do with these patients as well as intermediate metabolizers.
"The results of this study point us in the direction of higher doses for the *1 and *2 genotypes to normalize them to the active metabolites of 75 mg," he said. "The most important open question in my mind after the study is, What do we do next to integrate genotyping more routinely into clinical practice for patients about to go on clopidogrel or already on clopidogrel?"
Dr. Mega responded that point of care tests are available that can be run by individuals in the catheter laboratory and deliver genotype results in 1 hour.
"So, I don’t think technology is going to be the rate-limiting step," she said.
In the study, VASP PRI was determined from blood samples sent to the Center for Platelet Research Studies in Boston. Platelet function testing was also conducted at each site using the point of care Verify Now P2Y12 test (Accumetrics).
ELEVATE-TIMI 56 included patients taking 75 mg clopidogrel for a myocardial infarction and/or undergoing percutaneous coronary intervention at least 4 weeks and not more than 6 months prior to study entry. Compliance during the study was 97.3% at the 75-mg dose, 98.1% at the 150-mg dose, 98.6% at the 225-mg dose, and 98.3% at the 300-mg dose. There were no deaths, cerebral events, or Thrombolysis in Myocardial Infarction (TIMI) major or minor bleeding events, Dr. Mega said.
The study was simultaneously published online (JAMA 2011 Nov. 16;306[doi:10.1001/jama.2011.1703]).
Bristol-Myers Squibb and Sanofi-Aventis sponsored the trial. Accumetrics and Nanosphere provided research supplies. Dr. Mega reported receiving grants from the National Institutes of Health. Dr. Lesko said he had no relevant financial disclosures.
ORLANDO – Genotyping can provide more targeted use of the commonly prescribed drug clopidogrel among patients with cardiovascular disease, according to data from the ELEVATE-TIMI 56 trial.
"We’re at the point that clopidogrel may not be a one-size-fits-all drug for our patients," Dr. Jessica Mega said at the annual scientific sessions of the American Heart Association.
She reported on the prospective ELEVATE-TIMI 56 trial involving 333 patients with stable cardiovascular disease who underwent genotyping for alleles of CYP2C19, which is primarily responsible for producing active metabolite from clopidogrel (Plavix). Research has shown that patients who carry nonfunctional alleles of CYP2C19 such as *2 have reduced plasma active metabolite levels, an increased risk of ex vivo residual platelet activity, and a greater risk of cardiac events such as stent thrombosis.
Based on the genotyping results, 86 carriers (80 heterozygotes and 6 homozygotes) of the CYP2C19*2 allele were randomized to daily clopidogrel doses of 75 mg, 150 mg, 225 mg, and 300 mg for four 14-day treatment periods, while 247 CYP2C19*2 noncarriers were assigned to daily clopidogrel 75 mg and 150 mg for two 14-day periods each.
Platelet testing at the end of each treatment period revealed that both CYP2C19*2 heterozygotes and homozygotes had significantly higher platelet reactivity than did noncarriers, said Dr. Mega, a cardiologist at Brigham and Women’s Hospital, Boston.
The mean vasodilator-stimulated phosphoprotein phosphorylation (VASP) platelet reactivity index (PRI) was 70% for carriers vs. 58% for noncarriers, while mean P2Y12 reaction units (PRUs) were 226 vs. 164, respectively.
Among CYP2C19*2 heterozygotes, tripling the daily maintenance dose of clopidogrel to 225 mg was needed to reduce platelet reactivity to the levels achieved with a standard 75-mg dose in noncarriers, she said. Specifically, the percentage of CYP2C19*2 heterozygotes who did not respond to standard dosing was reduced from 52% to 26% with a 150-mg dose, and to 10% with at least 225 mg of clopidogrel.
Among CYP2C19*2 homozygotes, who made up 2% of the study population, even 300 mg, or four times the standard dose, of clopidogrel did not result in optimal degrees of platelet inhibition.
Dr. Mega said alternative agents with higher degrees of platelet inhibition are available, but that clopidogrel will become generic next year and continues to be used in a significant number of patients.
"It’s good news for patients that we’re able to optimize their [platelet] inhibition with clopidogrel," she said. "I think we need to get creative in how we use clopidogrel, and this study is one step in the right direction."
Invited discussant Lawrence Lesko, Ph.D., director of the center for pharmacometrics and systems pharmacology at the University of Florida in Lake Nona, pointed out that clopidogrel carries a "black box" warning indicating that poor metabolizers are at heightened risk for cardiovascular events, and that ELEVATE-TIMI 56 fills in some of the gaps in the labeling of what to do with these patients as well as intermediate metabolizers.
"The results of this study point us in the direction of higher doses for the *1 and *2 genotypes to normalize them to the active metabolites of 75 mg," he said. "The most important open question in my mind after the study is, What do we do next to integrate genotyping more routinely into clinical practice for patients about to go on clopidogrel or already on clopidogrel?"
Dr. Mega responded that point of care tests are available that can be run by individuals in the catheter laboratory and deliver genotype results in 1 hour.
"So, I don’t think technology is going to be the rate-limiting step," she said.
In the study, VASP PRI was determined from blood samples sent to the Center for Platelet Research Studies in Boston. Platelet function testing was also conducted at each site using the point of care Verify Now P2Y12 test (Accumetrics).
ELEVATE-TIMI 56 included patients taking 75 mg clopidogrel for a myocardial infarction and/or undergoing percutaneous coronary intervention at least 4 weeks and not more than 6 months prior to study entry. Compliance during the study was 97.3% at the 75-mg dose, 98.1% at the 150-mg dose, 98.6% at the 225-mg dose, and 98.3% at the 300-mg dose. There were no deaths, cerebral events, or Thrombolysis in Myocardial Infarction (TIMI) major or minor bleeding events, Dr. Mega said.
The study was simultaneously published online (JAMA 2011 Nov. 16;306[doi:10.1001/jama.2011.1703]).
Bristol-Myers Squibb and Sanofi-Aventis sponsored the trial. Accumetrics and Nanosphere provided research supplies. Dr. Mega reported receiving grants from the National Institutes of Health. Dr. Lesko said he had no relevant financial disclosures.
ORLANDO – Genotyping can provide more targeted use of the commonly prescribed drug clopidogrel among patients with cardiovascular disease, according to data from the ELEVATE-TIMI 56 trial.
"We’re at the point that clopidogrel may not be a one-size-fits-all drug for our patients," Dr. Jessica Mega said at the annual scientific sessions of the American Heart Association.
She reported on the prospective ELEVATE-TIMI 56 trial involving 333 patients with stable cardiovascular disease who underwent genotyping for alleles of CYP2C19, which is primarily responsible for producing active metabolite from clopidogrel (Plavix). Research has shown that patients who carry nonfunctional alleles of CYP2C19 such as *2 have reduced plasma active metabolite levels, an increased risk of ex vivo residual platelet activity, and a greater risk of cardiac events such as stent thrombosis.
Based on the genotyping results, 86 carriers (80 heterozygotes and 6 homozygotes) of the CYP2C19*2 allele were randomized to daily clopidogrel doses of 75 mg, 150 mg, 225 mg, and 300 mg for four 14-day treatment periods, while 247 CYP2C19*2 noncarriers were assigned to daily clopidogrel 75 mg and 150 mg for two 14-day periods each.
Platelet testing at the end of each treatment period revealed that both CYP2C19*2 heterozygotes and homozygotes had significantly higher platelet reactivity than did noncarriers, said Dr. Mega, a cardiologist at Brigham and Women’s Hospital, Boston.
The mean vasodilator-stimulated phosphoprotein phosphorylation (VASP) platelet reactivity index (PRI) was 70% for carriers vs. 58% for noncarriers, while mean P2Y12 reaction units (PRUs) were 226 vs. 164, respectively.
Among CYP2C19*2 heterozygotes, tripling the daily maintenance dose of clopidogrel to 225 mg was needed to reduce platelet reactivity to the levels achieved with a standard 75-mg dose in noncarriers, she said. Specifically, the percentage of CYP2C19*2 heterozygotes who did not respond to standard dosing was reduced from 52% to 26% with a 150-mg dose, and to 10% with at least 225 mg of clopidogrel.
Among CYP2C19*2 homozygotes, who made up 2% of the study population, even 300 mg, or four times the standard dose, of clopidogrel did not result in optimal degrees of platelet inhibition.
Dr. Mega said alternative agents with higher degrees of platelet inhibition are available, but that clopidogrel will become generic next year and continues to be used in a significant number of patients.
"It’s good news for patients that we’re able to optimize their [platelet] inhibition with clopidogrel," she said. "I think we need to get creative in how we use clopidogrel, and this study is one step in the right direction."
Invited discussant Lawrence Lesko, Ph.D., director of the center for pharmacometrics and systems pharmacology at the University of Florida in Lake Nona, pointed out that clopidogrel carries a "black box" warning indicating that poor metabolizers are at heightened risk for cardiovascular events, and that ELEVATE-TIMI 56 fills in some of the gaps in the labeling of what to do with these patients as well as intermediate metabolizers.
"The results of this study point us in the direction of higher doses for the *1 and *2 genotypes to normalize them to the active metabolites of 75 mg," he said. "The most important open question in my mind after the study is, What do we do next to integrate genotyping more routinely into clinical practice for patients about to go on clopidogrel or already on clopidogrel?"
Dr. Mega responded that point of care tests are available that can be run by individuals in the catheter laboratory and deliver genotype results in 1 hour.
"So, I don’t think technology is going to be the rate-limiting step," she said.
In the study, VASP PRI was determined from blood samples sent to the Center for Platelet Research Studies in Boston. Platelet function testing was also conducted at each site using the point of care Verify Now P2Y12 test (Accumetrics).
ELEVATE-TIMI 56 included patients taking 75 mg clopidogrel for a myocardial infarction and/or undergoing percutaneous coronary intervention at least 4 weeks and not more than 6 months prior to study entry. Compliance during the study was 97.3% at the 75-mg dose, 98.1% at the 150-mg dose, 98.6% at the 225-mg dose, and 98.3% at the 300-mg dose. There were no deaths, cerebral events, or Thrombolysis in Myocardial Infarction (TIMI) major or minor bleeding events, Dr. Mega said.
The study was simultaneously published online (JAMA 2011 Nov. 16;306[doi:10.1001/jama.2011.1703]).
Bristol-Myers Squibb and Sanofi-Aventis sponsored the trial. Accumetrics and Nanosphere provided research supplies. Dr. Mega reported receiving grants from the National Institutes of Health. Dr. Lesko said he had no relevant financial disclosures.
FROM THE ANNUAL SCIENTIFIC SESSIONS OF THE AMERICAN HEART ASSOCIATION
Major Finding: The percentage of CYP2C19*2 heterozygotes with no response in platelet activity to standard clopidogrel dosing was reduced from 52% to 26% with a 150-mg dose, and to 10% with at least 225 mg of clopidogrel.
Data Source: ELEVATE-TIMI 56, a prospective, randomized double-blind trial in 333 patients with stable cardiovascular disease.
Disclosures: Bristol-Myers Squibb and Sanofi-Aventis sponsored the trial. Accumetrics and Nanosphere provided research supplies. Dr. Mega reported receiving grants from the National Institutes of Health. Dr. Lesko said he had no relevant financial disclosures.