User login
New AHA/ASA stroke guidelines stress rtPA, telemedicine
Expanded use of clot-busting therapy is strongly endorsed for patients with acute ischemic stroke, while mechanical thrombectomy devices garner only lukewarm support in updated acute ischemic stroke guidelines from the American Heart Association and American Stroke Association.
The "door-to-needle time" for intravenous administration of clot-busting recombinant tissue plasminogen activator (rtPA) should be within 60 minutes from hospital arrival, according to a new class I, evidence level A recommendation.
Clinicians are advised to consider a noncontrast brain CT or MRI and a series of blood tests in all patients with suspected ischemic stroke before administering rtPA, but ultimately, the guidelines (Stroke 2013 Jan. 31 [doi:10.1161/STR.0b013e318284056a]) state that "The only laboratory result required in all patients before fibrinolytics therapy is initiated is a glucose determination; use of finger-stick measurement devices is acceptable."
The treatment window for rtPA therapy is also extended from 3 hours to 4.5 hours after stroke onset – as recommended in the AHA/ASA 2009 update on the extended time window for administration of fibrinolytic agents (Stroke 2009;40:2945-8).
"It’s clear that time is brain," lead guideline author Dr. Edward Jauch, director of emergency medicine at the Medical University of South Carolina in Charleston, said in an interview. "We are making a much greater emphasis that patients should be evaluated as quickly as possible and get treated as quickly as possible to give them the maximum opportunity for benefit."
Dr. Jauch acknowledges that the recommendations could reignite the long-standing controversy over the use of rtPA in stroke patients, particularly in light of the Food and Drug Administration’s recent decision not to expand approval of rtPA to include treatment up to 4.5 hours, as the European Medicines Agency has done.
"The FDA makes decisions largely based on American data, and we make guidelines based on all available data," he said, noting that safety data were also obtained from Genentech, maker of the rtPA activase (Alteplase).
Two European trials – the third International Stroke Trial (Lancet 2012;379:2352-63) and a British meta-analysis (Lancet 2012;379:2364-72) – reported last year that rtPA therapy within 6 hours of symptom onset increased the proportion of people who were alive and independent on follow-up.
Dr. Patrick Lyden, chair of neurology and director of the stroke program at Cedars-Sinai Medical Center in Los Angeles, said reducing the battery of blood tests prior to rtPA administration is particularly important and pointed out that when the FDA first approved rtPA to treat stroke on the basis of a National Institutes of Health study, it "took the research protocol and turned it into a package insert.
"It’s taken the intervening 16 years for people to do studies and realize that you don’t need to do all the things in the package insert," he said. "So the American Heart Association, for the first time, is endorsing a much more practical, a much more optimal use of tPA for stroke."
The arrival of new classes of anticoagulants has prompted the AHA/ASA to add a new recommendation that the use of intravenous or intra-arterial rtPA in patients taking direct thrombin inhibitors like dabigatran (Pradaxa) or direct factor Xa inhibitors like rivaroxaban (Xarelto) "may be harmful" and is not recommended unless specialized testing is normal, or the patient has been off the drug for more than 2 days.
"I think that’s overreaching; I don’t think the data support that," said Dr. Lyden, who was not a member of the guidelines writing committee. He added that his team has had "no safety issues whatsoever" when administering the anticoagulant argatroban in patients on rtPA.
Dr. Jauch counters that data are lacking to support the safety of rtPA in patients on the new anticoagulants. Common blood tests – such as the international normalized ratio used for warfarin – do not register the anticoagulant effects of these drugs and reversal strategies are not yet known.
"As a community, we have a ways to go to figure out the optimal way to manage stroke in patients who come in on these drugs," he said.
When mechanical thrombectomy is pursued, stent retrievers are generally preferred to coil retrievers. The guidelines acknowledge that the Merci embolus retrieval system, Penumbra System, Solitaire FR, and TREVO thrombectomy devices "can be useful" in achieving recanalization alone or in combination with fibrinolytics in carefully selected patients, but that "their ability to improve patient outcomes has not been established" and continued study in randomized trials is warranted.
While these devices can restore blood flow very quickly, part of the problem in evaluating them is that the time from when the patient develops their stroke to when they get to the catheterization lab continues to increase, Dr. Jauch said.
"One of the challenges we have is, yes, we have a great device and if you happen to have your stroke on the cath table, you’re in great luck," he said. "But if you transfer multiple times or there’s a delay in getting the patient evaluated sufficiently, then it diminishes the chance of getting a good outcome."
Telemedicine comes of age
If feasible, patients should be transported to the closest available certified primary care stroke center or comprehensive stroke center, which in some cases may involve air transport or hospital bypass.
An estimated 40% of Americans, however, live in remote or rural areas without direct access to a comprehensive stroke center. For these patients, the updated guidelines emphasize the use of telemedicine to extend expert stroke care and optimize the use of intravenous rtPA, said guideline coauthor Dr. Bart M. Demaerschalk, professor of neurology at Mayo Clinic in Phoenix, which serves as a hub for 12 hospitals across Arizona with limited or no neurologic support.
"Even if air transport is available, the patients generally arrive when the respective treatment window is already closed," he said. "So telemedicine often means the difference between no treatment whatsoever, which is the usual case, and treatment."
The guidelines recommend teleradiology systems approved by the FDA or "an equivalent organization" for sites without in-house imaging expertise for prompt review of brain CT and MRI scans in patients with suspected acute stroke. When it’s not physically possible for a stroke team physician to be at the bedside, telemedicine should also be established so that more hospitals can potentially meet the criteria to become acute stroke-ready hospitals and primary stroke hospitals.
Telemedicine may also be cost effective, according to a recent study coauthored by Dr. Demaerschalk. It reports that a telestroke network model with one hub and seven spoke hospitals would result in 45 more patients getting intravenous thrombolysis and 20 more getting endovascular stroke therapies per year compared with usual care, and was associated with an estimated annual cost savings of $358,435 or $109,080 for each spoke hospital (Circ. Cardiovasc. Qual. Outcomes 2013;6:18-26).
Fellow guideline coauthor Dr. David Z. Wang, who has established one of the largest teleneurology networks in the country with 27 spoke hospitals in the OSF St. Francis Medical Center’s stroke network in Peoria, Ill., said the emphasis on telemedicine recognizes that there is a shortage of neurologists and radiologists, and that stroke care isn’t always as simple as giving a shot of rtPA. When they first started the teleneurology network in 1997, emergency physicians did not feel comfortable diagnosing stroke or giving rtPA. Over the years, however, things began to change as these hospitals, some as small as 20 beds, gained more experience with teleneurology, including feedback on the final diagnosis.
"Our hospitals in these smaller, rural areas, they all feel very comfortable now making a diagnosis," he said. "They make a phone call to us and jointly make a decision, offer treatment expediently and then ship them over to our center. The mentality has changed ... we’re providing better care."
Many guidelines committee members had financial ties with drug manufacturers and device makers.
Expanded use of clot-busting therapy is strongly endorsed for patients with acute ischemic stroke, while mechanical thrombectomy devices garner only lukewarm support in updated acute ischemic stroke guidelines from the American Heart Association and American Stroke Association.
The "door-to-needle time" for intravenous administration of clot-busting recombinant tissue plasminogen activator (rtPA) should be within 60 minutes from hospital arrival, according to a new class I, evidence level A recommendation.
Clinicians are advised to consider a noncontrast brain CT or MRI and a series of blood tests in all patients with suspected ischemic stroke before administering rtPA, but ultimately, the guidelines (Stroke 2013 Jan. 31 [doi:10.1161/STR.0b013e318284056a]) state that "The only laboratory result required in all patients before fibrinolytics therapy is initiated is a glucose determination; use of finger-stick measurement devices is acceptable."
The treatment window for rtPA therapy is also extended from 3 hours to 4.5 hours after stroke onset – as recommended in the AHA/ASA 2009 update on the extended time window for administration of fibrinolytic agents (Stroke 2009;40:2945-8).
"It’s clear that time is brain," lead guideline author Dr. Edward Jauch, director of emergency medicine at the Medical University of South Carolina in Charleston, said in an interview. "We are making a much greater emphasis that patients should be evaluated as quickly as possible and get treated as quickly as possible to give them the maximum opportunity for benefit."
Dr. Jauch acknowledges that the recommendations could reignite the long-standing controversy over the use of rtPA in stroke patients, particularly in light of the Food and Drug Administration’s recent decision not to expand approval of rtPA to include treatment up to 4.5 hours, as the European Medicines Agency has done.
"The FDA makes decisions largely based on American data, and we make guidelines based on all available data," he said, noting that safety data were also obtained from Genentech, maker of the rtPA activase (Alteplase).
Two European trials – the third International Stroke Trial (Lancet 2012;379:2352-63) and a British meta-analysis (Lancet 2012;379:2364-72) – reported last year that rtPA therapy within 6 hours of symptom onset increased the proportion of people who were alive and independent on follow-up.
Dr. Patrick Lyden, chair of neurology and director of the stroke program at Cedars-Sinai Medical Center in Los Angeles, said reducing the battery of blood tests prior to rtPA administration is particularly important and pointed out that when the FDA first approved rtPA to treat stroke on the basis of a National Institutes of Health study, it "took the research protocol and turned it into a package insert.
"It’s taken the intervening 16 years for people to do studies and realize that you don’t need to do all the things in the package insert," he said. "So the American Heart Association, for the first time, is endorsing a much more practical, a much more optimal use of tPA for stroke."
The arrival of new classes of anticoagulants has prompted the AHA/ASA to add a new recommendation that the use of intravenous or intra-arterial rtPA in patients taking direct thrombin inhibitors like dabigatran (Pradaxa) or direct factor Xa inhibitors like rivaroxaban (Xarelto) "may be harmful" and is not recommended unless specialized testing is normal, or the patient has been off the drug for more than 2 days.
"I think that’s overreaching; I don’t think the data support that," said Dr. Lyden, who was not a member of the guidelines writing committee. He added that his team has had "no safety issues whatsoever" when administering the anticoagulant argatroban in patients on rtPA.
Dr. Jauch counters that data are lacking to support the safety of rtPA in patients on the new anticoagulants. Common blood tests – such as the international normalized ratio used for warfarin – do not register the anticoagulant effects of these drugs and reversal strategies are not yet known.
"As a community, we have a ways to go to figure out the optimal way to manage stroke in patients who come in on these drugs," he said.
When mechanical thrombectomy is pursued, stent retrievers are generally preferred to coil retrievers. The guidelines acknowledge that the Merci embolus retrieval system, Penumbra System, Solitaire FR, and TREVO thrombectomy devices "can be useful" in achieving recanalization alone or in combination with fibrinolytics in carefully selected patients, but that "their ability to improve patient outcomes has not been established" and continued study in randomized trials is warranted.
While these devices can restore blood flow very quickly, part of the problem in evaluating them is that the time from when the patient develops their stroke to when they get to the catheterization lab continues to increase, Dr. Jauch said.
"One of the challenges we have is, yes, we have a great device and if you happen to have your stroke on the cath table, you’re in great luck," he said. "But if you transfer multiple times or there’s a delay in getting the patient evaluated sufficiently, then it diminishes the chance of getting a good outcome."
Telemedicine comes of age
If feasible, patients should be transported to the closest available certified primary care stroke center or comprehensive stroke center, which in some cases may involve air transport or hospital bypass.
An estimated 40% of Americans, however, live in remote or rural areas without direct access to a comprehensive stroke center. For these patients, the updated guidelines emphasize the use of telemedicine to extend expert stroke care and optimize the use of intravenous rtPA, said guideline coauthor Dr. Bart M. Demaerschalk, professor of neurology at Mayo Clinic in Phoenix, which serves as a hub for 12 hospitals across Arizona with limited or no neurologic support.
"Even if air transport is available, the patients generally arrive when the respective treatment window is already closed," he said. "So telemedicine often means the difference between no treatment whatsoever, which is the usual case, and treatment."
The guidelines recommend teleradiology systems approved by the FDA or "an equivalent organization" for sites without in-house imaging expertise for prompt review of brain CT and MRI scans in patients with suspected acute stroke. When it’s not physically possible for a stroke team physician to be at the bedside, telemedicine should also be established so that more hospitals can potentially meet the criteria to become acute stroke-ready hospitals and primary stroke hospitals.
Telemedicine may also be cost effective, according to a recent study coauthored by Dr. Demaerschalk. It reports that a telestroke network model with one hub and seven spoke hospitals would result in 45 more patients getting intravenous thrombolysis and 20 more getting endovascular stroke therapies per year compared with usual care, and was associated with an estimated annual cost savings of $358,435 or $109,080 for each spoke hospital (Circ. Cardiovasc. Qual. Outcomes 2013;6:18-26).
Fellow guideline coauthor Dr. David Z. Wang, who has established one of the largest teleneurology networks in the country with 27 spoke hospitals in the OSF St. Francis Medical Center’s stroke network in Peoria, Ill., said the emphasis on telemedicine recognizes that there is a shortage of neurologists and radiologists, and that stroke care isn’t always as simple as giving a shot of rtPA. When they first started the teleneurology network in 1997, emergency physicians did not feel comfortable diagnosing stroke or giving rtPA. Over the years, however, things began to change as these hospitals, some as small as 20 beds, gained more experience with teleneurology, including feedback on the final diagnosis.
"Our hospitals in these smaller, rural areas, they all feel very comfortable now making a diagnosis," he said. "They make a phone call to us and jointly make a decision, offer treatment expediently and then ship them over to our center. The mentality has changed ... we’re providing better care."
Many guidelines committee members had financial ties with drug manufacturers and device makers.
Expanded use of clot-busting therapy is strongly endorsed for patients with acute ischemic stroke, while mechanical thrombectomy devices garner only lukewarm support in updated acute ischemic stroke guidelines from the American Heart Association and American Stroke Association.
The "door-to-needle time" for intravenous administration of clot-busting recombinant tissue plasminogen activator (rtPA) should be within 60 minutes from hospital arrival, according to a new class I, evidence level A recommendation.
Clinicians are advised to consider a noncontrast brain CT or MRI and a series of blood tests in all patients with suspected ischemic stroke before administering rtPA, but ultimately, the guidelines (Stroke 2013 Jan. 31 [doi:10.1161/STR.0b013e318284056a]) state that "The only laboratory result required in all patients before fibrinolytics therapy is initiated is a glucose determination; use of finger-stick measurement devices is acceptable."
The treatment window for rtPA therapy is also extended from 3 hours to 4.5 hours after stroke onset – as recommended in the AHA/ASA 2009 update on the extended time window for administration of fibrinolytic agents (Stroke 2009;40:2945-8).
"It’s clear that time is brain," lead guideline author Dr. Edward Jauch, director of emergency medicine at the Medical University of South Carolina in Charleston, said in an interview. "We are making a much greater emphasis that patients should be evaluated as quickly as possible and get treated as quickly as possible to give them the maximum opportunity for benefit."
Dr. Jauch acknowledges that the recommendations could reignite the long-standing controversy over the use of rtPA in stroke patients, particularly in light of the Food and Drug Administration’s recent decision not to expand approval of rtPA to include treatment up to 4.5 hours, as the European Medicines Agency has done.
"The FDA makes decisions largely based on American data, and we make guidelines based on all available data," he said, noting that safety data were also obtained from Genentech, maker of the rtPA activase (Alteplase).
Two European trials – the third International Stroke Trial (Lancet 2012;379:2352-63) and a British meta-analysis (Lancet 2012;379:2364-72) – reported last year that rtPA therapy within 6 hours of symptom onset increased the proportion of people who were alive and independent on follow-up.
Dr. Patrick Lyden, chair of neurology and director of the stroke program at Cedars-Sinai Medical Center in Los Angeles, said reducing the battery of blood tests prior to rtPA administration is particularly important and pointed out that when the FDA first approved rtPA to treat stroke on the basis of a National Institutes of Health study, it "took the research protocol and turned it into a package insert.
"It’s taken the intervening 16 years for people to do studies and realize that you don’t need to do all the things in the package insert," he said. "So the American Heart Association, for the first time, is endorsing a much more practical, a much more optimal use of tPA for stroke."
The arrival of new classes of anticoagulants has prompted the AHA/ASA to add a new recommendation that the use of intravenous or intra-arterial rtPA in patients taking direct thrombin inhibitors like dabigatran (Pradaxa) or direct factor Xa inhibitors like rivaroxaban (Xarelto) "may be harmful" and is not recommended unless specialized testing is normal, or the patient has been off the drug for more than 2 days.
"I think that’s overreaching; I don’t think the data support that," said Dr. Lyden, who was not a member of the guidelines writing committee. He added that his team has had "no safety issues whatsoever" when administering the anticoagulant argatroban in patients on rtPA.
Dr. Jauch counters that data are lacking to support the safety of rtPA in patients on the new anticoagulants. Common blood tests – such as the international normalized ratio used for warfarin – do not register the anticoagulant effects of these drugs and reversal strategies are not yet known.
"As a community, we have a ways to go to figure out the optimal way to manage stroke in patients who come in on these drugs," he said.
When mechanical thrombectomy is pursued, stent retrievers are generally preferred to coil retrievers. The guidelines acknowledge that the Merci embolus retrieval system, Penumbra System, Solitaire FR, and TREVO thrombectomy devices "can be useful" in achieving recanalization alone or in combination with fibrinolytics in carefully selected patients, but that "their ability to improve patient outcomes has not been established" and continued study in randomized trials is warranted.
While these devices can restore blood flow very quickly, part of the problem in evaluating them is that the time from when the patient develops their stroke to when they get to the catheterization lab continues to increase, Dr. Jauch said.
"One of the challenges we have is, yes, we have a great device and if you happen to have your stroke on the cath table, you’re in great luck," he said. "But if you transfer multiple times or there’s a delay in getting the patient evaluated sufficiently, then it diminishes the chance of getting a good outcome."
Telemedicine comes of age
If feasible, patients should be transported to the closest available certified primary care stroke center or comprehensive stroke center, which in some cases may involve air transport or hospital bypass.
An estimated 40% of Americans, however, live in remote or rural areas without direct access to a comprehensive stroke center. For these patients, the updated guidelines emphasize the use of telemedicine to extend expert stroke care and optimize the use of intravenous rtPA, said guideline coauthor Dr. Bart M. Demaerschalk, professor of neurology at Mayo Clinic in Phoenix, which serves as a hub for 12 hospitals across Arizona with limited or no neurologic support.
"Even if air transport is available, the patients generally arrive when the respective treatment window is already closed," he said. "So telemedicine often means the difference between no treatment whatsoever, which is the usual case, and treatment."
The guidelines recommend teleradiology systems approved by the FDA or "an equivalent organization" for sites without in-house imaging expertise for prompt review of brain CT and MRI scans in patients with suspected acute stroke. When it’s not physically possible for a stroke team physician to be at the bedside, telemedicine should also be established so that more hospitals can potentially meet the criteria to become acute stroke-ready hospitals and primary stroke hospitals.
Telemedicine may also be cost effective, according to a recent study coauthored by Dr. Demaerschalk. It reports that a telestroke network model with one hub and seven spoke hospitals would result in 45 more patients getting intravenous thrombolysis and 20 more getting endovascular stroke therapies per year compared with usual care, and was associated with an estimated annual cost savings of $358,435 or $109,080 for each spoke hospital (Circ. Cardiovasc. Qual. Outcomes 2013;6:18-26).
Fellow guideline coauthor Dr. David Z. Wang, who has established one of the largest teleneurology networks in the country with 27 spoke hospitals in the OSF St. Francis Medical Center’s stroke network in Peoria, Ill., said the emphasis on telemedicine recognizes that there is a shortage of neurologists and radiologists, and that stroke care isn’t always as simple as giving a shot of rtPA. When they first started the teleneurology network in 1997, emergency physicians did not feel comfortable diagnosing stroke or giving rtPA. Over the years, however, things began to change as these hospitals, some as small as 20 beds, gained more experience with teleneurology, including feedback on the final diagnosis.
"Our hospitals in these smaller, rural areas, they all feel very comfortable now making a diagnosis," he said. "They make a phone call to us and jointly make a decision, offer treatment expediently and then ship them over to our center. The mentality has changed ... we’re providing better care."
Many guidelines committee members had financial ties with drug manufacturers and device makers.
FROM STROKE
Full-face motorcycle helmets blunt head trauma
Wearing a full-face helmet during a motorcycle crash dramatically reduced the risk of craniofacial fractures and traumatic brain injury, a prospective, observational study has shown.
Among 151 motorcycle crash victims, those wearing full-face helmets were significantly less likely than those wearing other helmet types to have a facial fracture (7.2% vs. 26.8%; P = .0221) or skull fracture (1.2% vs. 10.6%; P = .0015).
This represents a relative risk reduction of 73% and 89%, respectively.
The incidence of traumatic brain injury was cut almost in half, from 24% to 13%, with a full-face helmet, but the difference did not reach statistical significance (relative risk reduction, 45%; P = .091), Dr. Brian L. Brewer reported at the annual meeting of the Eastern Association for the Surgery of Trauma (EAST).
"Similar studies need to be performed to document the beneficial effect of full-face helmets in decreasing the incidence of skull fractures and facial fractures and, possibly, traumatic brain injury and mortality," said Dr. Brewer, whose research earned the 2013 Cox-Templeton Injury Prevention Paper Award from the EAST Foundation.
Currently, about 20 states require that all motorcyclists wear a helmet. Two states have no such law on the books, and several other states require only that certain individuals wear helmets while riding a motorcycle.
"Surprisingly, legislation in this arena is relatively anemic, given the significant financial implications for health care cost," he said, noting that 4,000 lives and an estimated $32.4 million could be saved each year with a universal helmet law.
On the opposite side of the yellow line, motorcyclists argue that helmets aren’t "cool," limit their field of vision, and increase the risk of cervical spine fractures. Others such as Michigan Gov. Rick Snyder, who last year repealed a 35-year-old law requiring helmets for all Michigan motorcycle drivers, consider it a matter of individual liberty.
The U.S. Department of Transportation has developed 218 minimal performance standards for DOT-certified helmets, but they pertain to weight, thickness, chin straps, and limitations on harmful decorations – not helmet design, explained Dr. Brewer of Sinai Hospital in Baltimore.
To explore whether full-face helmets could offer a greater degree of protection from craniofacial trauma than modular, half, and bucket helmets, the researchers prospectively enrolled 135 men and 16 women with an average age of 38.4 years (range, 19-74 years) who were involved in a motorcycle crash from 2011 to 2012.
The average Glasgow Coma Scale score was 14 in the full-face group and 13 in the other helmet group. Injury Severity Scores were 11.87 and 14.64, respectively. "Other helmet" types were 39 half-helmets and 28 modular helmets.
In all, there were cervical spine fractures in 8.5% of patients wearing full-face helmets, compared with 11.9% of those wearing other helmet types (relative risk reduction, 20%; P = .78), Dr. Brewer said. Mortality was also similar, at 4.8% and 7.9% (RRR, 36%; P = 0.36).
He acknowledged that data from this and other studies may do little to convince unwilling motorcyclists to don a helmet, particularly older riders – who surprisingly were found to be less likely to wear the safer, full-face helmets.
"We will use this and further studies to prove to the Department of Transportation that there should be a minimum standard in helmets," Dr. Brewer said in an interview. Then, "once we can affect the legislature, then we can go out to the public and say, ‘Listen, this is the law, and this is what you have to do.’ "
Dr. Brewer and his coauthors reported no conflicts of interest.
Wearing a full-face helmet during a motorcycle crash dramatically reduced the risk of craniofacial fractures and traumatic brain injury, a prospective, observational study has shown.
Among 151 motorcycle crash victims, those wearing full-face helmets were significantly less likely than those wearing other helmet types to have a facial fracture (7.2% vs. 26.8%; P = .0221) or skull fracture (1.2% vs. 10.6%; P = .0015).
This represents a relative risk reduction of 73% and 89%, respectively.
The incidence of traumatic brain injury was cut almost in half, from 24% to 13%, with a full-face helmet, but the difference did not reach statistical significance (relative risk reduction, 45%; P = .091), Dr. Brian L. Brewer reported at the annual meeting of the Eastern Association for the Surgery of Trauma (EAST).
"Similar studies need to be performed to document the beneficial effect of full-face helmets in decreasing the incidence of skull fractures and facial fractures and, possibly, traumatic brain injury and mortality," said Dr. Brewer, whose research earned the 2013 Cox-Templeton Injury Prevention Paper Award from the EAST Foundation.
Currently, about 20 states require that all motorcyclists wear a helmet. Two states have no such law on the books, and several other states require only that certain individuals wear helmets while riding a motorcycle.
"Surprisingly, legislation in this arena is relatively anemic, given the significant financial implications for health care cost," he said, noting that 4,000 lives and an estimated $32.4 million could be saved each year with a universal helmet law.
On the opposite side of the yellow line, motorcyclists argue that helmets aren’t "cool," limit their field of vision, and increase the risk of cervical spine fractures. Others such as Michigan Gov. Rick Snyder, who last year repealed a 35-year-old law requiring helmets for all Michigan motorcycle drivers, consider it a matter of individual liberty.
The U.S. Department of Transportation has developed 218 minimal performance standards for DOT-certified helmets, but they pertain to weight, thickness, chin straps, and limitations on harmful decorations – not helmet design, explained Dr. Brewer of Sinai Hospital in Baltimore.
To explore whether full-face helmets could offer a greater degree of protection from craniofacial trauma than modular, half, and bucket helmets, the researchers prospectively enrolled 135 men and 16 women with an average age of 38.4 years (range, 19-74 years) who were involved in a motorcycle crash from 2011 to 2012.
The average Glasgow Coma Scale score was 14 in the full-face group and 13 in the other helmet group. Injury Severity Scores were 11.87 and 14.64, respectively. "Other helmet" types were 39 half-helmets and 28 modular helmets.
In all, there were cervical spine fractures in 8.5% of patients wearing full-face helmets, compared with 11.9% of those wearing other helmet types (relative risk reduction, 20%; P = .78), Dr. Brewer said. Mortality was also similar, at 4.8% and 7.9% (RRR, 36%; P = 0.36).
He acknowledged that data from this and other studies may do little to convince unwilling motorcyclists to don a helmet, particularly older riders – who surprisingly were found to be less likely to wear the safer, full-face helmets.
"We will use this and further studies to prove to the Department of Transportation that there should be a minimum standard in helmets," Dr. Brewer said in an interview. Then, "once we can affect the legislature, then we can go out to the public and say, ‘Listen, this is the law, and this is what you have to do.’ "
Dr. Brewer and his coauthors reported no conflicts of interest.
Wearing a full-face helmet during a motorcycle crash dramatically reduced the risk of craniofacial fractures and traumatic brain injury, a prospective, observational study has shown.
Among 151 motorcycle crash victims, those wearing full-face helmets were significantly less likely than those wearing other helmet types to have a facial fracture (7.2% vs. 26.8%; P = .0221) or skull fracture (1.2% vs. 10.6%; P = .0015).
This represents a relative risk reduction of 73% and 89%, respectively.
The incidence of traumatic brain injury was cut almost in half, from 24% to 13%, with a full-face helmet, but the difference did not reach statistical significance (relative risk reduction, 45%; P = .091), Dr. Brian L. Brewer reported at the annual meeting of the Eastern Association for the Surgery of Trauma (EAST).
"Similar studies need to be performed to document the beneficial effect of full-face helmets in decreasing the incidence of skull fractures and facial fractures and, possibly, traumatic brain injury and mortality," said Dr. Brewer, whose research earned the 2013 Cox-Templeton Injury Prevention Paper Award from the EAST Foundation.
Currently, about 20 states require that all motorcyclists wear a helmet. Two states have no such law on the books, and several other states require only that certain individuals wear helmets while riding a motorcycle.
"Surprisingly, legislation in this arena is relatively anemic, given the significant financial implications for health care cost," he said, noting that 4,000 lives and an estimated $32.4 million could be saved each year with a universal helmet law.
On the opposite side of the yellow line, motorcyclists argue that helmets aren’t "cool," limit their field of vision, and increase the risk of cervical spine fractures. Others such as Michigan Gov. Rick Snyder, who last year repealed a 35-year-old law requiring helmets for all Michigan motorcycle drivers, consider it a matter of individual liberty.
The U.S. Department of Transportation has developed 218 minimal performance standards for DOT-certified helmets, but they pertain to weight, thickness, chin straps, and limitations on harmful decorations – not helmet design, explained Dr. Brewer of Sinai Hospital in Baltimore.
To explore whether full-face helmets could offer a greater degree of protection from craniofacial trauma than modular, half, and bucket helmets, the researchers prospectively enrolled 135 men and 16 women with an average age of 38.4 years (range, 19-74 years) who were involved in a motorcycle crash from 2011 to 2012.
The average Glasgow Coma Scale score was 14 in the full-face group and 13 in the other helmet group. Injury Severity Scores were 11.87 and 14.64, respectively. "Other helmet" types were 39 half-helmets and 28 modular helmets.
In all, there were cervical spine fractures in 8.5% of patients wearing full-face helmets, compared with 11.9% of those wearing other helmet types (relative risk reduction, 20%; P = .78), Dr. Brewer said. Mortality was also similar, at 4.8% and 7.9% (RRR, 36%; P = 0.36).
He acknowledged that data from this and other studies may do little to convince unwilling motorcyclists to don a helmet, particularly older riders – who surprisingly were found to be less likely to wear the safer, full-face helmets.
"We will use this and further studies to prove to the Department of Transportation that there should be a minimum standard in helmets," Dr. Brewer said in an interview. Then, "once we can affect the legislature, then we can go out to the public and say, ‘Listen, this is the law, and this is what you have to do.’ "
Dr. Brewer and his coauthors reported no conflicts of interest.
AT THE ANNUAL MEETING OF THE EASTERN ASSOCIATION FOR THE SURGERY OF TRAUMA
Major Finding: Patients wearing full-face helmets were significantly less likely than those wearing other helmet types to have a facial fracture (7.2% vs. 26.8%; P = .022) or skull fracture (1.2% vs. 10.6%; P = .0015).
Data Source: Prospective, observational study of 151 motorcycle crash victims.
Disclosures: Dr. Brewer and his coauthors reported no conflicts of interest.
ACS trauma verification ups survival at level II centers
SCOTTSDALE, AZ. – Level II trauma centers appeared to benefit most from attaining American College of Surgeons trauma verification, a retrospective analysis of a large national sample suggests.
Attaining ACS trauma consultation/verification was an independent predictor of patient survival in level II trauma centers (adjusted odds ratio, 1.26; P less than .01), but not in level I trauma centers (OR, 1.0; P = .84), which already performed well as a group.
Level II centers with state designation only were 3.5 times more likely to be poor performers based on survival outcomes than were level II centers that had attained ACS verification, Dr. Joshua Brown reported at the annual meeting of the Eastern Association for the Surgery of Trauma.
"Promoting ACS verification among level II trauma centers may represent an opportunity for improved outcomes in select trauma centers," he said.
A rigorous process that demands significant financial and personnel resources, ACS verification has been shown in single-center reports to improve outcomes. Yet less than 25% of level I and II trauma centers pursue ACS verification if already designated by their state as a trauma center, said Dr. Brown, a general surgery resident at the University of Pittsburgh Medical Center.
To compare mortality at ACS verified and state designated centers in a national sample, the investigators identified 900,274 patients, at least 16 years old, in the National Trauma Data Bank 2007-2008. Their mean age was 42 years, mean Injury Severity Score was 9, and 76% were transported from the trauma scene. The patients were admitted to 189 level I (65% ACS verified) and 185 level II trauma centers (67% ACS verified).
A mortality prediction model was constructed based on published Trauma Quality Improvement Program (TQIP) methodology, and multiple imputation used for missing data. The model had an area under the curve of 0.92, indicating excellent discrimination to predict death, Dr. Brown said.
Level I ACS centers had a significantly lower median observed-to-expected (O/E) mortality ratio than did level I state centers, (0.95 vs. 1.02; P less than .01).
No difference was observed in median O/E mortality ratio between level II ACS and state centers (0.94 vs. 0.87; P = .30), he said.
As noted earlier, however, level II state centers had more than three times the poor performers – high O/E ratio outliers, defined by an O/E and 90% confidence interval greater than 1.0 – than level II ACS centers (13% vs. 4%; OR 3.59; P = .03).
The proportion of good performers – low O/E outliers, defined by an O/E and C.I. less than 1.0 – was similar at state vs. ACS level II centers (21% vs. 23%;P = .57).
At level I trauma centers, no significant differences were observed between ACS-verified and state-verified centers in the proportion of high outliers (15% vs. 24%; P = .16) or low outliers (28% vs. 16%; P = .1), Dr. Brown reported.
Dr. Keith Clancy of York (Penn.) Hospital, who was invited to discuss the study, said, "It was a well-written, well-designed paper on a very important topic that will provide some pause for states that may inappropriately believe that simply by incorporating ACS standards into their state trauma standards they can automatically achieve the same outcomes as ACS verification."
Dr. Brown and Dr. Clancy reported no financial disclosures.
SCOTTSDALE, AZ. – Level II trauma centers appeared to benefit most from attaining American College of Surgeons trauma verification, a retrospective analysis of a large national sample suggests.
Attaining ACS trauma consultation/verification was an independent predictor of patient survival in level II trauma centers (adjusted odds ratio, 1.26; P less than .01), but not in level I trauma centers (OR, 1.0; P = .84), which already performed well as a group.
Level II centers with state designation only were 3.5 times more likely to be poor performers based on survival outcomes than were level II centers that had attained ACS verification, Dr. Joshua Brown reported at the annual meeting of the Eastern Association for the Surgery of Trauma.
"Promoting ACS verification among level II trauma centers may represent an opportunity for improved outcomes in select trauma centers," he said.
A rigorous process that demands significant financial and personnel resources, ACS verification has been shown in single-center reports to improve outcomes. Yet less than 25% of level I and II trauma centers pursue ACS verification if already designated by their state as a trauma center, said Dr. Brown, a general surgery resident at the University of Pittsburgh Medical Center.
To compare mortality at ACS verified and state designated centers in a national sample, the investigators identified 900,274 patients, at least 16 years old, in the National Trauma Data Bank 2007-2008. Their mean age was 42 years, mean Injury Severity Score was 9, and 76% were transported from the trauma scene. The patients were admitted to 189 level I (65% ACS verified) and 185 level II trauma centers (67% ACS verified).
A mortality prediction model was constructed based on published Trauma Quality Improvement Program (TQIP) methodology, and multiple imputation used for missing data. The model had an area under the curve of 0.92, indicating excellent discrimination to predict death, Dr. Brown said.
Level I ACS centers had a significantly lower median observed-to-expected (O/E) mortality ratio than did level I state centers, (0.95 vs. 1.02; P less than .01).
No difference was observed in median O/E mortality ratio between level II ACS and state centers (0.94 vs. 0.87; P = .30), he said.
As noted earlier, however, level II state centers had more than three times the poor performers – high O/E ratio outliers, defined by an O/E and 90% confidence interval greater than 1.0 – than level II ACS centers (13% vs. 4%; OR 3.59; P = .03).
The proportion of good performers – low O/E outliers, defined by an O/E and C.I. less than 1.0 – was similar at state vs. ACS level II centers (21% vs. 23%;P = .57).
At level I trauma centers, no significant differences were observed between ACS-verified and state-verified centers in the proportion of high outliers (15% vs. 24%; P = .16) or low outliers (28% vs. 16%; P = .1), Dr. Brown reported.
Dr. Keith Clancy of York (Penn.) Hospital, who was invited to discuss the study, said, "It was a well-written, well-designed paper on a very important topic that will provide some pause for states that may inappropriately believe that simply by incorporating ACS standards into their state trauma standards they can automatically achieve the same outcomes as ACS verification."
Dr. Brown and Dr. Clancy reported no financial disclosures.
SCOTTSDALE, AZ. – Level II trauma centers appeared to benefit most from attaining American College of Surgeons trauma verification, a retrospective analysis of a large national sample suggests.
Attaining ACS trauma consultation/verification was an independent predictor of patient survival in level II trauma centers (adjusted odds ratio, 1.26; P less than .01), but not in level I trauma centers (OR, 1.0; P = .84), which already performed well as a group.
Level II centers with state designation only were 3.5 times more likely to be poor performers based on survival outcomes than were level II centers that had attained ACS verification, Dr. Joshua Brown reported at the annual meeting of the Eastern Association for the Surgery of Trauma.
"Promoting ACS verification among level II trauma centers may represent an opportunity for improved outcomes in select trauma centers," he said.
A rigorous process that demands significant financial and personnel resources, ACS verification has been shown in single-center reports to improve outcomes. Yet less than 25% of level I and II trauma centers pursue ACS verification if already designated by their state as a trauma center, said Dr. Brown, a general surgery resident at the University of Pittsburgh Medical Center.
To compare mortality at ACS verified and state designated centers in a national sample, the investigators identified 900,274 patients, at least 16 years old, in the National Trauma Data Bank 2007-2008. Their mean age was 42 years, mean Injury Severity Score was 9, and 76% were transported from the trauma scene. The patients were admitted to 189 level I (65% ACS verified) and 185 level II trauma centers (67% ACS verified).
A mortality prediction model was constructed based on published Trauma Quality Improvement Program (TQIP) methodology, and multiple imputation used for missing data. The model had an area under the curve of 0.92, indicating excellent discrimination to predict death, Dr. Brown said.
Level I ACS centers had a significantly lower median observed-to-expected (O/E) mortality ratio than did level I state centers, (0.95 vs. 1.02; P less than .01).
No difference was observed in median O/E mortality ratio between level II ACS and state centers (0.94 vs. 0.87; P = .30), he said.
As noted earlier, however, level II state centers had more than three times the poor performers – high O/E ratio outliers, defined by an O/E and 90% confidence interval greater than 1.0 – than level II ACS centers (13% vs. 4%; OR 3.59; P = .03).
The proportion of good performers – low O/E outliers, defined by an O/E and C.I. less than 1.0 – was similar at state vs. ACS level II centers (21% vs. 23%;P = .57).
At level I trauma centers, no significant differences were observed between ACS-verified and state-verified centers in the proportion of high outliers (15% vs. 24%; P = .16) or low outliers (28% vs. 16%; P = .1), Dr. Brown reported.
Dr. Keith Clancy of York (Penn.) Hospital, who was invited to discuss the study, said, "It was a well-written, well-designed paper on a very important topic that will provide some pause for states that may inappropriately believe that simply by incorporating ACS standards into their state trauma standards they can automatically achieve the same outcomes as ACS verification."
Dr. Brown and Dr. Clancy reported no financial disclosures.
AT THE ANNUAL MEETING OF THE EASTERN ASSOCIATION FOR THE SURGERY OF TRAUMA
Major Finding: ACS trauma verification was a significant independent predictor of survival at level II trauma centers (adjusted OR, 1.26; P less than .01), but not at level I centers (OR, 1.0; P = .84).
Data Source: Retrospective analysis of 900,274 patients in the National Trauma Data Bank.
Disclosures: Dr. Brown and Dr. Clancy reported no disclosures.
VIDEO: Full-face motorcycle helmets and head injuries
Can full-face motorcycle helmets reduce riders' risk of craniofacial fractures and traumatic brain injury? Dr. Brian L. Brewer's new study provides some surprising answers.
Can full-face motorcycle helmets reduce riders' risk of craniofacial fractures and traumatic brain injury? Dr. Brian L. Brewer's new study provides some surprising answers.
Can full-face motorcycle helmets reduce riders' risk of craniofacial fractures and traumatic brain injury? Dr. Brian L. Brewer's new study provides some surprising answers.
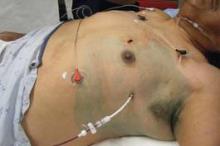
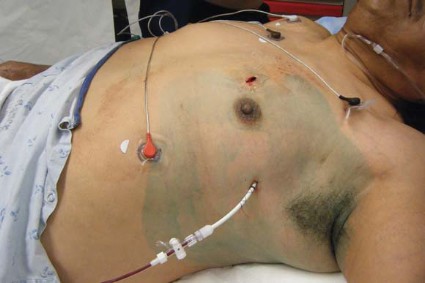
Pigtail catheters less painful than chest tubes for traumatic pneumothorax
In patients with traumatic pneumothorax, smaller really is better, a study has shown.
The prospective, randomized study found that 14-French pigtail catheters function as well as do traditional, large-bore 28-French chest tubes, and result in significantly less tube-site pain.
The average numerical rating tube-site pain score was 3.2 with the pigtail catheter vs. 7.7 with the chest tube on the day of insertion, 1.9 vs. 6.2 on day 1 (both P values less than .001), and 2.1 vs. 5.5 on day 2 (P = .04).
Chest wall pain was similar between the two groups at all three time points (6.1 vs. 6.0; 5.5 vs. 5.9; and 4.2 vs. 5.9), as was intravenous pain medication use on day 1 (mean 10.3 U vs. 15.4 U) and day 2 (5.0 U vs. 8.6 U). One unit equaled 1 mg morphine, 25 mcg fentanyl, or 0.1 mg hydromorphone HCl (Dilaudid).
It stands to reason that the pigtail catheters are less painful because they’re soft, have a coiled tip, and are inserted with a less-invasive percutaneous technique, whereas traditional, large-bore chest tubes are straight and stiff, and are typically placed via a cutdown technique, Dr. Narong Kulvatunyou said at the annual meeting of the Eastern Association for the Surgery of Trauma.
A recent prospective observational study found no difference in tube-site pain with large (36- to 40-French) versus small (28- to 32-French) chest tubes inserted for chest trauma (J. Trauma Acute Care Surg. 2012;72:422-7), but the analysis still compared two large bores and both used the cutdown technique, he observed.
Based on their early experience in trauma patients, Dr. Kulvatunyou and his colleagues at the University of Arizona, Tucson, reported that 11% of pigtail catheters failed versus 4% of chest tubes (P = .06) (J. Trauma 2011;71:1104-7).
The 2-year review did not assess tube-site pain, prompting the current prospective study involving 40 patients with traumatic pneumothorax evenly randomized to a 14F pigtail catheter placed at the bedside with a modified Seldinger technique or a 28F chest tube placed via a cutdown technique. The tubes were left on suction for 24 hours.
Patients were excluded if they required emergency pigtail or chest tube placement or were unable to respond to the nurse-led pain assessment.
Demographics in the pigtail and chest tube groups were similar in average Injury Severity Score (14.5 vs. 12.2), abbreviated chest injury score (3 vs. 3), blunt trauma injury (85% vs. 80%), rib fractures (both 1.5), and pulmonary contusion (both 25%). Patients’ mean age was 46 years; 80% were male.
Contrary to expectations, tube-site pain was similar whether the pigtail catheter was placed anteriorly between the second and third rib (n = 9) or laterally (n = 11) between the fourth and fifth rib, Dr. Kulvatunyou said.
Failure rate, defined as unresolved or recurrent pneumothorax requiring a second tube, was 5% in the pigtail group and 10% in the chest tube group (P = .55). Secondary endpoints included insertion-related complications (10% for both groups), median number of tube days (2 for both), and median hospital stay (4 days for both).
Invited discussant Dr. David King, a trauma and acute care surgeon at Massachusetts General Hospital, Boston, questioned whether patients were receiving oral pain medications, as this could impact the results, and how the team mitigated a potential Hawthorne effect and observer bias.
"I know you said that the nurses who were getting the pain scores were blinded, but it’s pretty difficult to blind someone to the difference between a garden hose and a straw coming out of their chest wall," he said.
Dr. Kulvatunyou acknowledged that 10 out of 10 patients would prefer a smaller tube if asked, but that patients were asked only whether they would be willing to receive a "different tube that works pretty well," with no mention of tube size. As for the nurses, he said the tubes were typically under dressings and that some nurses may not have noticed the difference – a remark that was not well received based on comments after the session.
Data on oral medications were not collected because they were rarely used in the first 2 days and it was difficult to compare the oral medications, but Dr. Kulvatunyou said the point was well taken.
He noted that pigtail catheters could have a role in other traumatic indications such as hemothorax, hemopneumothorax, and emergency situations, but that additional studies are needed to show that it has equal efficacy to those chest tubes.
"I’m sure everyone puts central lines in pretty quickly, so just imagine if you could do pigtails just as quick as central lines," he said. "It would change everything."
Dr. Kulvatunyou, his coauthors, and Dr. King reported no relevant financial disclosures.
In patients with traumatic pneumothorax, smaller really is better, a study has shown.
The prospective, randomized study found that 14-French pigtail catheters function as well as do traditional, large-bore 28-French chest tubes, and result in significantly less tube-site pain.
The average numerical rating tube-site pain score was 3.2 with the pigtail catheter vs. 7.7 with the chest tube on the day of insertion, 1.9 vs. 6.2 on day 1 (both P values less than .001), and 2.1 vs. 5.5 on day 2 (P = .04).
Chest wall pain was similar between the two groups at all three time points (6.1 vs. 6.0; 5.5 vs. 5.9; and 4.2 vs. 5.9), as was intravenous pain medication use on day 1 (mean 10.3 U vs. 15.4 U) and day 2 (5.0 U vs. 8.6 U). One unit equaled 1 mg morphine, 25 mcg fentanyl, or 0.1 mg hydromorphone HCl (Dilaudid).
It stands to reason that the pigtail catheters are less painful because they’re soft, have a coiled tip, and are inserted with a less-invasive percutaneous technique, whereas traditional, large-bore chest tubes are straight and stiff, and are typically placed via a cutdown technique, Dr. Narong Kulvatunyou said at the annual meeting of the Eastern Association for the Surgery of Trauma.
A recent prospective observational study found no difference in tube-site pain with large (36- to 40-French) versus small (28- to 32-French) chest tubes inserted for chest trauma (J. Trauma Acute Care Surg. 2012;72:422-7), but the analysis still compared two large bores and both used the cutdown technique, he observed.
Based on their early experience in trauma patients, Dr. Kulvatunyou and his colleagues at the University of Arizona, Tucson, reported that 11% of pigtail catheters failed versus 4% of chest tubes (P = .06) (J. Trauma 2011;71:1104-7).
The 2-year review did not assess tube-site pain, prompting the current prospective study involving 40 patients with traumatic pneumothorax evenly randomized to a 14F pigtail catheter placed at the bedside with a modified Seldinger technique or a 28F chest tube placed via a cutdown technique. The tubes were left on suction for 24 hours.
Patients were excluded if they required emergency pigtail or chest tube placement or were unable to respond to the nurse-led pain assessment.
Demographics in the pigtail and chest tube groups were similar in average Injury Severity Score (14.5 vs. 12.2), abbreviated chest injury score (3 vs. 3), blunt trauma injury (85% vs. 80%), rib fractures (both 1.5), and pulmonary contusion (both 25%). Patients’ mean age was 46 years; 80% were male.
Contrary to expectations, tube-site pain was similar whether the pigtail catheter was placed anteriorly between the second and third rib (n = 9) or laterally (n = 11) between the fourth and fifth rib, Dr. Kulvatunyou said.
Failure rate, defined as unresolved or recurrent pneumothorax requiring a second tube, was 5% in the pigtail group and 10% in the chest tube group (P = .55). Secondary endpoints included insertion-related complications (10% for both groups), median number of tube days (2 for both), and median hospital stay (4 days for both).
Invited discussant Dr. David King, a trauma and acute care surgeon at Massachusetts General Hospital, Boston, questioned whether patients were receiving oral pain medications, as this could impact the results, and how the team mitigated a potential Hawthorne effect and observer bias.
"I know you said that the nurses who were getting the pain scores were blinded, but it’s pretty difficult to blind someone to the difference between a garden hose and a straw coming out of their chest wall," he said.
Dr. Kulvatunyou acknowledged that 10 out of 10 patients would prefer a smaller tube if asked, but that patients were asked only whether they would be willing to receive a "different tube that works pretty well," with no mention of tube size. As for the nurses, he said the tubes were typically under dressings and that some nurses may not have noticed the difference – a remark that was not well received based on comments after the session.
Data on oral medications were not collected because they were rarely used in the first 2 days and it was difficult to compare the oral medications, but Dr. Kulvatunyou said the point was well taken.
He noted that pigtail catheters could have a role in other traumatic indications such as hemothorax, hemopneumothorax, and emergency situations, but that additional studies are needed to show that it has equal efficacy to those chest tubes.
"I’m sure everyone puts central lines in pretty quickly, so just imagine if you could do pigtails just as quick as central lines," he said. "It would change everything."
Dr. Kulvatunyou, his coauthors, and Dr. King reported no relevant financial disclosures.
In patients with traumatic pneumothorax, smaller really is better, a study has shown.
The prospective, randomized study found that 14-French pigtail catheters function as well as do traditional, large-bore 28-French chest tubes, and result in significantly less tube-site pain.
The average numerical rating tube-site pain score was 3.2 with the pigtail catheter vs. 7.7 with the chest tube on the day of insertion, 1.9 vs. 6.2 on day 1 (both P values less than .001), and 2.1 vs. 5.5 on day 2 (P = .04).
Chest wall pain was similar between the two groups at all three time points (6.1 vs. 6.0; 5.5 vs. 5.9; and 4.2 vs. 5.9), as was intravenous pain medication use on day 1 (mean 10.3 U vs. 15.4 U) and day 2 (5.0 U vs. 8.6 U). One unit equaled 1 mg morphine, 25 mcg fentanyl, or 0.1 mg hydromorphone HCl (Dilaudid).
It stands to reason that the pigtail catheters are less painful because they’re soft, have a coiled tip, and are inserted with a less-invasive percutaneous technique, whereas traditional, large-bore chest tubes are straight and stiff, and are typically placed via a cutdown technique, Dr. Narong Kulvatunyou said at the annual meeting of the Eastern Association for the Surgery of Trauma.
A recent prospective observational study found no difference in tube-site pain with large (36- to 40-French) versus small (28- to 32-French) chest tubes inserted for chest trauma (J. Trauma Acute Care Surg. 2012;72:422-7), but the analysis still compared two large bores and both used the cutdown technique, he observed.
Based on their early experience in trauma patients, Dr. Kulvatunyou and his colleagues at the University of Arizona, Tucson, reported that 11% of pigtail catheters failed versus 4% of chest tubes (P = .06) (J. Trauma 2011;71:1104-7).
The 2-year review did not assess tube-site pain, prompting the current prospective study involving 40 patients with traumatic pneumothorax evenly randomized to a 14F pigtail catheter placed at the bedside with a modified Seldinger technique or a 28F chest tube placed via a cutdown technique. The tubes were left on suction for 24 hours.
Patients were excluded if they required emergency pigtail or chest tube placement or were unable to respond to the nurse-led pain assessment.
Demographics in the pigtail and chest tube groups were similar in average Injury Severity Score (14.5 vs. 12.2), abbreviated chest injury score (3 vs. 3), blunt trauma injury (85% vs. 80%), rib fractures (both 1.5), and pulmonary contusion (both 25%). Patients’ mean age was 46 years; 80% were male.
Contrary to expectations, tube-site pain was similar whether the pigtail catheter was placed anteriorly between the second and third rib (n = 9) or laterally (n = 11) between the fourth and fifth rib, Dr. Kulvatunyou said.
Failure rate, defined as unresolved or recurrent pneumothorax requiring a second tube, was 5% in the pigtail group and 10% in the chest tube group (P = .55). Secondary endpoints included insertion-related complications (10% for both groups), median number of tube days (2 for both), and median hospital stay (4 days for both).
Invited discussant Dr. David King, a trauma and acute care surgeon at Massachusetts General Hospital, Boston, questioned whether patients were receiving oral pain medications, as this could impact the results, and how the team mitigated a potential Hawthorne effect and observer bias.
"I know you said that the nurses who were getting the pain scores were blinded, but it’s pretty difficult to blind someone to the difference between a garden hose and a straw coming out of their chest wall," he said.
Dr. Kulvatunyou acknowledged that 10 out of 10 patients would prefer a smaller tube if asked, but that patients were asked only whether they would be willing to receive a "different tube that works pretty well," with no mention of tube size. As for the nurses, he said the tubes were typically under dressings and that some nurses may not have noticed the difference – a remark that was not well received based on comments after the session.
Data on oral medications were not collected because they were rarely used in the first 2 days and it was difficult to compare the oral medications, but Dr. Kulvatunyou said the point was well taken.
He noted that pigtail catheters could have a role in other traumatic indications such as hemothorax, hemopneumothorax, and emergency situations, but that additional studies are needed to show that it has equal efficacy to those chest tubes.
"I’m sure everyone puts central lines in pretty quickly, so just imagine if you could do pigtails just as quick as central lines," he said. "It would change everything."
Dr. Kulvatunyou, his coauthors, and Dr. King reported no relevant financial disclosures.
AT THE ANNUAL MEETING OF THE EASTERN ASSOCIATION FOR THE SURGERY OF TRAUMA
Major Finding: The average tube-site pain score was 3.2 with the pigtail catheter vs. 7.7 with the chest tube on the day of insertion, 1.9 vs. 6.2 on day 1 (both P values less than .001), and 2.1 vs. 5.5 on day 2 (P = .04).
Data Source: A prospective, randomized trial of 40 patients with traumatic pneumothorax.
Disclosures: Dr. Kulvatunyou, his coauthors, and Dr. King reported no relevant financial disclosures.
Experimental CARS therapy breaks through relapsed CLL
ATLANTA – Responses to chimeric antigen receptor–modified T-cell therapy have been sustained beyond 2 years in a small cohort of patients with advanced chronic lymphocytic leukemia and no other curable options.
Three patients remain in complete remission at 7, 27, and 28 months, with no evidence of disease in their blood or bone marrow and complete resolution of all abnormal adenopathy.
Partial responses have been reported in four patients, including two with no evidence of CLL in their blood and bone marrow and more than a 50% reduction in bulky nodes at 2 and 3 months’ follow-up. The two other partial responders were treated earlier with an older vector lot that may not have been as potent, and had responses lasting approximately 4 months, Dr. David Porter said at the annual meeting of the American Society of Hematology.
Two of the nine evaluable patients had no response, for an overall response rate of 78%. Another patient was treated too recently to evaluate.
The findings represent a potential paradigm shift in the treatment of blood cancers, particularly for patients with advanced disease or with high-risk features. These patients are incurable except by allogeneic bone marrow or stem cell transplants, which are associated with extensive morbidity and mortality, he said.
In previous trials of chimeric antigen receptor (CAR) T cells, responses were modest and in vivo proliferation of the modified cells was not sustained. Investigators at the Hospital of the University of Pennsylvania, Philadelphia, then developed a second-generation CAR that targeted the B-cell antigen CD19, but was also linked to the CD3-zeta and CD137 (4-1BB) signaling domains. Preclinical observations showed that CD137 promotes the persistence of antigen-specific and antigen-nonspecific CAR T cells.
A pilot study reported a complete response within 3 weeks of treatment with the genetically modified T cells in a patient with advanced chronic lymphoid leukemia, who remains in remission today (N. Engl. J. Med. 2011;365:725-33). The therapy also resulted in the recovery of a 6-year-old with acute lymphoblastic leukemia as recently reported in the New York Times.
"So it’s now been in a definitive medical journal," quipped Dr. Porter.
On a more serious note, he acknowledged that in addition to its curative potential, "there is significant toxicity associated with this therapy." Five responders experienced grade 3-4 hepatotoxicity, with one patient on transient dialysis on several occasions for renal failure related to tumor lysis syndrome and hypotension.
Persistent B-cell aplasia with hypogammaglobulinemia developed in all patients achieving a complete remission, although treatment with intravenous immunoglobulin has been successful in preventing excessive or frequent infections.
In addition, all responding patients developed a rather profound cytokine release syndrome at the time of T-cell expansion, manifested by very high fevers, nausea, hypotension, and even capillary leak and hypoxia, said Dr. Porter, director of the blood and marrow transplantation program at the hospital.
Research revealed high levels of interleukin (IL)-6 (6-400x), interferon-gamma (89-1000), and IL-2R (5-25), but no significant increase in tumor necrosis factor–alpha or IL-2. Steroids reversed the syndrome in one patient. Given the cytokine profile, however, the switch was made to anticytokine therapy with tocilizumab (Actemra).
Cytokine release syndrome "can be treated effectively with anticytokine therapy, but what we don’t know is whether earlier treatment for the cytokine release syndrome is going to abrogate the T-cell activity and response," Dr. Porter said.
The 10 adult patients he reported had CD19-positive CLL that had been treated with at least two prior therapies (median 5; range 2-10) and progressed within 2 years of the last treatment. Three patients had the high-risk deletion 17p. Their median age was 66 years.
The patients received lympho-depleting chemotherapy 4-7 days before reinfusion with their genetically modified T cells using a lentivirus vector. The median cell dose was 7.5 x 108 (range 1.7-50 x 108), the infused dose of modified T cells was 1.4 x 108 per kilogram, and the median follow-up was 8 months. There was no significant infusion-related toxicity, Dr. Porter said.
Responders experienced a massive expansion of T cells – 1,000- to 10,000-fold in vivo. This translated into a 2-log expansion in patients with a partial response and more than a 3-log expansion in complete responders. The expansion was associated with tumor lysis syndrome in complete responders.
The cells are at very high numbers early on after therapy, representing almost 15% of all the CD3-positive cells at 12 months and 1.2% at 18 months. The two long-term patients have levels of about 1% at 2 years, he said.
Deep whole transcriptome sequencing revealed no evidence of minimal residual disease in patients with a complete response.
The term "serial killer cells" has been coined because there has been an effector-to-target ratio of 1 in 1,000 to 1 in 93,000, meaning that the infused T cells, or their progeny, can kill up to 93,000 tumor cells, Dr. Porter said.
"When we do the math, we estimate that’s between 2.9 and 7.5 pounds of tumor in these patients that we’ve treated," he added.
During a discussion of the results, Dr. Porter said that in patients with cytokine release syndrome, anti-IL-6 therapy is typically given at the first sign of hemodynamic instability and that improvement is almost instantaneous. To address the B-cell dysplasia, he said, there is a lot of interest in developing either more specific targets or technology such as a suicide vector where the CARS are self-limited to allow for normal B-cell development.
In August 2012, the University of Pennsylvania and Novartis announced an exclusive global research and licensing agreement to study and commercialize CAR therapies for other cancers at a Center for Advanced Cellular Therapies to be built on the university campus.
The research was supported in part by grants from the National Institutes of Health, the Leukemia and Lymphoma Society, and the Alliance for Cancer Gene Therapy. Dr. Porter reported patents and royalties from Novartis, honoraria from Celgene, spouse employment with Genentech, and research funding from Pfizer. His coauthors reported commercial relationships with TxCell and Novartis.
ATLANTA – Responses to chimeric antigen receptor–modified T-cell therapy have been sustained beyond 2 years in a small cohort of patients with advanced chronic lymphocytic leukemia and no other curable options.
Three patients remain in complete remission at 7, 27, and 28 months, with no evidence of disease in their blood or bone marrow and complete resolution of all abnormal adenopathy.
Partial responses have been reported in four patients, including two with no evidence of CLL in their blood and bone marrow and more than a 50% reduction in bulky nodes at 2 and 3 months’ follow-up. The two other partial responders were treated earlier with an older vector lot that may not have been as potent, and had responses lasting approximately 4 months, Dr. David Porter said at the annual meeting of the American Society of Hematology.
Two of the nine evaluable patients had no response, for an overall response rate of 78%. Another patient was treated too recently to evaluate.
The findings represent a potential paradigm shift in the treatment of blood cancers, particularly for patients with advanced disease or with high-risk features. These patients are incurable except by allogeneic bone marrow or stem cell transplants, which are associated with extensive morbidity and mortality, he said.
In previous trials of chimeric antigen receptor (CAR) T cells, responses were modest and in vivo proliferation of the modified cells was not sustained. Investigators at the Hospital of the University of Pennsylvania, Philadelphia, then developed a second-generation CAR that targeted the B-cell antigen CD19, but was also linked to the CD3-zeta and CD137 (4-1BB) signaling domains. Preclinical observations showed that CD137 promotes the persistence of antigen-specific and antigen-nonspecific CAR T cells.
A pilot study reported a complete response within 3 weeks of treatment with the genetically modified T cells in a patient with advanced chronic lymphoid leukemia, who remains in remission today (N. Engl. J. Med. 2011;365:725-33). The therapy also resulted in the recovery of a 6-year-old with acute lymphoblastic leukemia as recently reported in the New York Times.
"So it’s now been in a definitive medical journal," quipped Dr. Porter.
On a more serious note, he acknowledged that in addition to its curative potential, "there is significant toxicity associated with this therapy." Five responders experienced grade 3-4 hepatotoxicity, with one patient on transient dialysis on several occasions for renal failure related to tumor lysis syndrome and hypotension.
Persistent B-cell aplasia with hypogammaglobulinemia developed in all patients achieving a complete remission, although treatment with intravenous immunoglobulin has been successful in preventing excessive or frequent infections.
In addition, all responding patients developed a rather profound cytokine release syndrome at the time of T-cell expansion, manifested by very high fevers, nausea, hypotension, and even capillary leak and hypoxia, said Dr. Porter, director of the blood and marrow transplantation program at the hospital.
Research revealed high levels of interleukin (IL)-6 (6-400x), interferon-gamma (89-1000), and IL-2R (5-25), but no significant increase in tumor necrosis factor–alpha or IL-2. Steroids reversed the syndrome in one patient. Given the cytokine profile, however, the switch was made to anticytokine therapy with tocilizumab (Actemra).
Cytokine release syndrome "can be treated effectively with anticytokine therapy, but what we don’t know is whether earlier treatment for the cytokine release syndrome is going to abrogate the T-cell activity and response," Dr. Porter said.
The 10 adult patients he reported had CD19-positive CLL that had been treated with at least two prior therapies (median 5; range 2-10) and progressed within 2 years of the last treatment. Three patients had the high-risk deletion 17p. Their median age was 66 years.
The patients received lympho-depleting chemotherapy 4-7 days before reinfusion with their genetically modified T cells using a lentivirus vector. The median cell dose was 7.5 x 108 (range 1.7-50 x 108), the infused dose of modified T cells was 1.4 x 108 per kilogram, and the median follow-up was 8 months. There was no significant infusion-related toxicity, Dr. Porter said.
Responders experienced a massive expansion of T cells – 1,000- to 10,000-fold in vivo. This translated into a 2-log expansion in patients with a partial response and more than a 3-log expansion in complete responders. The expansion was associated with tumor lysis syndrome in complete responders.
The cells are at very high numbers early on after therapy, representing almost 15% of all the CD3-positive cells at 12 months and 1.2% at 18 months. The two long-term patients have levels of about 1% at 2 years, he said.
Deep whole transcriptome sequencing revealed no evidence of minimal residual disease in patients with a complete response.
The term "serial killer cells" has been coined because there has been an effector-to-target ratio of 1 in 1,000 to 1 in 93,000, meaning that the infused T cells, or their progeny, can kill up to 93,000 tumor cells, Dr. Porter said.
"When we do the math, we estimate that’s between 2.9 and 7.5 pounds of tumor in these patients that we’ve treated," he added.
During a discussion of the results, Dr. Porter said that in patients with cytokine release syndrome, anti-IL-6 therapy is typically given at the first sign of hemodynamic instability and that improvement is almost instantaneous. To address the B-cell dysplasia, he said, there is a lot of interest in developing either more specific targets or technology such as a suicide vector where the CARS are self-limited to allow for normal B-cell development.
In August 2012, the University of Pennsylvania and Novartis announced an exclusive global research and licensing agreement to study and commercialize CAR therapies for other cancers at a Center for Advanced Cellular Therapies to be built on the university campus.
The research was supported in part by grants from the National Institutes of Health, the Leukemia and Lymphoma Society, and the Alliance for Cancer Gene Therapy. Dr. Porter reported patents and royalties from Novartis, honoraria from Celgene, spouse employment with Genentech, and research funding from Pfizer. His coauthors reported commercial relationships with TxCell and Novartis.
ATLANTA – Responses to chimeric antigen receptor–modified T-cell therapy have been sustained beyond 2 years in a small cohort of patients with advanced chronic lymphocytic leukemia and no other curable options.
Three patients remain in complete remission at 7, 27, and 28 months, with no evidence of disease in their blood or bone marrow and complete resolution of all abnormal adenopathy.
Partial responses have been reported in four patients, including two with no evidence of CLL in their blood and bone marrow and more than a 50% reduction in bulky nodes at 2 and 3 months’ follow-up. The two other partial responders were treated earlier with an older vector lot that may not have been as potent, and had responses lasting approximately 4 months, Dr. David Porter said at the annual meeting of the American Society of Hematology.
Two of the nine evaluable patients had no response, for an overall response rate of 78%. Another patient was treated too recently to evaluate.
The findings represent a potential paradigm shift in the treatment of blood cancers, particularly for patients with advanced disease or with high-risk features. These patients are incurable except by allogeneic bone marrow or stem cell transplants, which are associated with extensive morbidity and mortality, he said.
In previous trials of chimeric antigen receptor (CAR) T cells, responses were modest and in vivo proliferation of the modified cells was not sustained. Investigators at the Hospital of the University of Pennsylvania, Philadelphia, then developed a second-generation CAR that targeted the B-cell antigen CD19, but was also linked to the CD3-zeta and CD137 (4-1BB) signaling domains. Preclinical observations showed that CD137 promotes the persistence of antigen-specific and antigen-nonspecific CAR T cells.
A pilot study reported a complete response within 3 weeks of treatment with the genetically modified T cells in a patient with advanced chronic lymphoid leukemia, who remains in remission today (N. Engl. J. Med. 2011;365:725-33). The therapy also resulted in the recovery of a 6-year-old with acute lymphoblastic leukemia as recently reported in the New York Times.
"So it’s now been in a definitive medical journal," quipped Dr. Porter.
On a more serious note, he acknowledged that in addition to its curative potential, "there is significant toxicity associated with this therapy." Five responders experienced grade 3-4 hepatotoxicity, with one patient on transient dialysis on several occasions for renal failure related to tumor lysis syndrome and hypotension.
Persistent B-cell aplasia with hypogammaglobulinemia developed in all patients achieving a complete remission, although treatment with intravenous immunoglobulin has been successful in preventing excessive or frequent infections.
In addition, all responding patients developed a rather profound cytokine release syndrome at the time of T-cell expansion, manifested by very high fevers, nausea, hypotension, and even capillary leak and hypoxia, said Dr. Porter, director of the blood and marrow transplantation program at the hospital.
Research revealed high levels of interleukin (IL)-6 (6-400x), interferon-gamma (89-1000), and IL-2R (5-25), but no significant increase in tumor necrosis factor–alpha or IL-2. Steroids reversed the syndrome in one patient. Given the cytokine profile, however, the switch was made to anticytokine therapy with tocilizumab (Actemra).
Cytokine release syndrome "can be treated effectively with anticytokine therapy, but what we don’t know is whether earlier treatment for the cytokine release syndrome is going to abrogate the T-cell activity and response," Dr. Porter said.
The 10 adult patients he reported had CD19-positive CLL that had been treated with at least two prior therapies (median 5; range 2-10) and progressed within 2 years of the last treatment. Three patients had the high-risk deletion 17p. Their median age was 66 years.
The patients received lympho-depleting chemotherapy 4-7 days before reinfusion with their genetically modified T cells using a lentivirus vector. The median cell dose was 7.5 x 108 (range 1.7-50 x 108), the infused dose of modified T cells was 1.4 x 108 per kilogram, and the median follow-up was 8 months. There was no significant infusion-related toxicity, Dr. Porter said.
Responders experienced a massive expansion of T cells – 1,000- to 10,000-fold in vivo. This translated into a 2-log expansion in patients with a partial response and more than a 3-log expansion in complete responders. The expansion was associated with tumor lysis syndrome in complete responders.
The cells are at very high numbers early on after therapy, representing almost 15% of all the CD3-positive cells at 12 months and 1.2% at 18 months. The two long-term patients have levels of about 1% at 2 years, he said.
Deep whole transcriptome sequencing revealed no evidence of minimal residual disease in patients with a complete response.
The term "serial killer cells" has been coined because there has been an effector-to-target ratio of 1 in 1,000 to 1 in 93,000, meaning that the infused T cells, or their progeny, can kill up to 93,000 tumor cells, Dr. Porter said.
"When we do the math, we estimate that’s between 2.9 and 7.5 pounds of tumor in these patients that we’ve treated," he added.
During a discussion of the results, Dr. Porter said that in patients with cytokine release syndrome, anti-IL-6 therapy is typically given at the first sign of hemodynamic instability and that improvement is almost instantaneous. To address the B-cell dysplasia, he said, there is a lot of interest in developing either more specific targets or technology such as a suicide vector where the CARS are self-limited to allow for normal B-cell development.
In August 2012, the University of Pennsylvania and Novartis announced an exclusive global research and licensing agreement to study and commercialize CAR therapies for other cancers at a Center for Advanced Cellular Therapies to be built on the university campus.
The research was supported in part by grants from the National Institutes of Health, the Leukemia and Lymphoma Society, and the Alliance for Cancer Gene Therapy. Dr. Porter reported patents and royalties from Novartis, honoraria from Celgene, spouse employment with Genentech, and research funding from Pfizer. His coauthors reported commercial relationships with TxCell and Novartis.
AT THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF HEMATOLOGY
Major Finding: The overall response rate was 78%, including three complete responses and four partial responses.
Data Source: Study in 10 patients with advanced CLL.
Disclosures: The research was supported in part by grants from the National Institutes of Health, the Leukemia and Lymphoma Society, and the Alliance for Cancer Gene Therapy. Dr. Porter reported patents and royalties from Novartis, honoraria from Celgene, spouse employment with Genentech, and research funding from Pfizer. His coauthors reported commercial relationships with TxCell and Novartis.
Double-unit cord blood transplants don't boost survival
ATLANTA – Survival was not enhanced among children with blood cancers after transplantation with two units of umbilical cord blood versus an adequately dosed single-unit transplant.
Among 224 children in a phase III study, overall survival at 1 year was 71% with a single-unit umbilical cord blood (UCB) transplant and 65% with a double-unit UCB transplant (P = .13).
All other outcomes were similar, except for a lower incidence of platelet recovery and higher incidence of grade III-IV acute graft-versus-host disease after double UCB transplants, Dr. John E. Wagner reported on behalf of the Blood and Marrow Transplant Clinical Trials Network at the annual meeting of the American Society of Hematology.
And for patients who don’t have an adequate single cord unit based on cell dose, "a double cord blood unit was certainly as good as, or an acceptable alternative, to the single," he said. "So it allowed us to extend the transplant experience."
One of the major shortcomings of UCB transplantation is that units containing a minimum of 2.5 x 107 total nucleated cells/kg of patient body weight are frequently unavailable. The optimal number of transplanted umbilical stem cells remains unclear, but cell dose is an important factor influencing engraftment, or the ability of cells to take root, and survival.
Pilot studies suggested that infusion of two partially HLA-matched UCB units is safe, leading investigators to hypothesize that higher cell doses achieved with double UCB transplant would translate into improved survival, explained Dr. Wagner, scientific director of clinical research for the adult and pediatric blood and bone marrow transplant program at the University of Minnesota Medical Center in Minneapolis.
Between December 2006 and February 2012, investigators at 32 transplant centers enrolled 224 patients, aged 1-21 years, with acute lymphoblastic leukemia (ALL), acute myeloid leukemia, natural killer cell leukemia, chronic myeloid leukemia, or myelodysplastic syndrome. All had at least two available UCB units – each at least 4 of 6 HLA-matched to the patient and 3 of 6 matched between units.
Patients were randomized to receive a conventional single UCB (n = 113) or double UCB (n = 111) transplant. In all, 2.7% of the single UCB group and 1% of the double UCB group crossed over to the opposite arm. Only 1.8% of patients in both groups were not transplanted.
The groups were well matched for age, gender, race, performance status, and disease status. A little more than half of all patients had ALL.
After a median follow-up of 25 months, 92% of patients were in remission, with 60% of these in a second or subsequent remission.
Neutrophil counts had recovered at day 42 in 86% of the double UCB group and in 89% of the single UCB group (P = .08). Platelet recovery at 6 months was significantly lower after double UCB transplant (66% vs. 76%; P = .04), Dr. Wagner said.
Rates were nearly identical in the single and double UCB groups for grade II-IV acute graft-versus-host-disease at 100 days (56% vs. 57%; P = .88) and for chronic GVHD at 1 year (28% vs. 31%; P = .60), although a subpopulation in the double UCB group was significantly more likely to experience grade III-IV acute GVHD (23% vs. 14%; P = .04).
More importantly, there was a very low risk of transplant-related relapse after double or single UCB transplant (14% vs. 12%; P = .37), he observed.
Disease-free survival at 1 year was also similar in the double and single UCB groups at 64% vs. 68% (P = .22).
Dr. Wagner said the average total nucleated cell doses of 3.9 x 107 in the single UCB group and 7.2 x 107 in the double UCB group were not adequate for detecting differences in overall survival. "It probably will take a much greater dose escalation to be able to see that [difference]," he said.
Still, he noted that the overall survival and engraftment rates seen in both arms are superior to what was expected based on historical data sets, including the Cord Blood Transplantation Study (COBLT). It reported cumulative incidences for neutrophil recovery and platelet engraftment of 80% and 50%, and an overall survival of 67.4% in children with hematologic malignancies, despite a median cell dose of 5.1 x 107 (Blood 2008;112:4318-27).
One possible explanation, he suggested, is a new conditioning regimen that includes 1,320 cGy of total body irradiation and cyclophosphamide 120 mg/kg, but replaces the equine antithymocyte globulin used in the COBLT study with fludarabine (Fludara) 75 mg/m2. All patients also received cyclosporine and mycophenolate mofetil for GVHD prophylaxis.
"What this showed us is that it’s not just receiving a double perhaps that has some benefit for some patients, but also the conditioning regimen that we had changed simultaneously at the University of Minnesota," Dr. Wagner said.
He also highlighted similar findings from a very recent study he coauthored that reported comparable adjusted risks of neutrophil recovery, transplant-related mortality, and overall mortality after double UCB and adequately dosed single UCB transplants in 409 adults with acute leukemia (Blood 2012 Dec. 9 [Epub ahead of print]).
The study was supported by the National Heart, Lung, and Blood Institute; the National Cancer Institute; and the Children’s Oncology Group. Dr. Wagner and his coauthors reported no relevant conflicts of interest.
ATLANTA – Survival was not enhanced among children with blood cancers after transplantation with two units of umbilical cord blood versus an adequately dosed single-unit transplant.
Among 224 children in a phase III study, overall survival at 1 year was 71% with a single-unit umbilical cord blood (UCB) transplant and 65% with a double-unit UCB transplant (P = .13).
All other outcomes were similar, except for a lower incidence of platelet recovery and higher incidence of grade III-IV acute graft-versus-host disease after double UCB transplants, Dr. John E. Wagner reported on behalf of the Blood and Marrow Transplant Clinical Trials Network at the annual meeting of the American Society of Hematology.
And for patients who don’t have an adequate single cord unit based on cell dose, "a double cord blood unit was certainly as good as, or an acceptable alternative, to the single," he said. "So it allowed us to extend the transplant experience."
One of the major shortcomings of UCB transplantation is that units containing a minimum of 2.5 x 107 total nucleated cells/kg of patient body weight are frequently unavailable. The optimal number of transplanted umbilical stem cells remains unclear, but cell dose is an important factor influencing engraftment, or the ability of cells to take root, and survival.
Pilot studies suggested that infusion of two partially HLA-matched UCB units is safe, leading investigators to hypothesize that higher cell doses achieved with double UCB transplant would translate into improved survival, explained Dr. Wagner, scientific director of clinical research for the adult and pediatric blood and bone marrow transplant program at the University of Minnesota Medical Center in Minneapolis.
Between December 2006 and February 2012, investigators at 32 transplant centers enrolled 224 patients, aged 1-21 years, with acute lymphoblastic leukemia (ALL), acute myeloid leukemia, natural killer cell leukemia, chronic myeloid leukemia, or myelodysplastic syndrome. All had at least two available UCB units – each at least 4 of 6 HLA-matched to the patient and 3 of 6 matched between units.
Patients were randomized to receive a conventional single UCB (n = 113) or double UCB (n = 111) transplant. In all, 2.7% of the single UCB group and 1% of the double UCB group crossed over to the opposite arm. Only 1.8% of patients in both groups were not transplanted.
The groups were well matched for age, gender, race, performance status, and disease status. A little more than half of all patients had ALL.
After a median follow-up of 25 months, 92% of patients were in remission, with 60% of these in a second or subsequent remission.
Neutrophil counts had recovered at day 42 in 86% of the double UCB group and in 89% of the single UCB group (P = .08). Platelet recovery at 6 months was significantly lower after double UCB transplant (66% vs. 76%; P = .04), Dr. Wagner said.
Rates were nearly identical in the single and double UCB groups for grade II-IV acute graft-versus-host-disease at 100 days (56% vs. 57%; P = .88) and for chronic GVHD at 1 year (28% vs. 31%; P = .60), although a subpopulation in the double UCB group was significantly more likely to experience grade III-IV acute GVHD (23% vs. 14%; P = .04).
More importantly, there was a very low risk of transplant-related relapse after double or single UCB transplant (14% vs. 12%; P = .37), he observed.
Disease-free survival at 1 year was also similar in the double and single UCB groups at 64% vs. 68% (P = .22).
Dr. Wagner said the average total nucleated cell doses of 3.9 x 107 in the single UCB group and 7.2 x 107 in the double UCB group were not adequate for detecting differences in overall survival. "It probably will take a much greater dose escalation to be able to see that [difference]," he said.
Still, he noted that the overall survival and engraftment rates seen in both arms are superior to what was expected based on historical data sets, including the Cord Blood Transplantation Study (COBLT). It reported cumulative incidences for neutrophil recovery and platelet engraftment of 80% and 50%, and an overall survival of 67.4% in children with hematologic malignancies, despite a median cell dose of 5.1 x 107 (Blood 2008;112:4318-27).
One possible explanation, he suggested, is a new conditioning regimen that includes 1,320 cGy of total body irradiation and cyclophosphamide 120 mg/kg, but replaces the equine antithymocyte globulin used in the COBLT study with fludarabine (Fludara) 75 mg/m2. All patients also received cyclosporine and mycophenolate mofetil for GVHD prophylaxis.
"What this showed us is that it’s not just receiving a double perhaps that has some benefit for some patients, but also the conditioning regimen that we had changed simultaneously at the University of Minnesota," Dr. Wagner said.
He also highlighted similar findings from a very recent study he coauthored that reported comparable adjusted risks of neutrophil recovery, transplant-related mortality, and overall mortality after double UCB and adequately dosed single UCB transplants in 409 adults with acute leukemia (Blood 2012 Dec. 9 [Epub ahead of print]).
The study was supported by the National Heart, Lung, and Blood Institute; the National Cancer Institute; and the Children’s Oncology Group. Dr. Wagner and his coauthors reported no relevant conflicts of interest.
ATLANTA – Survival was not enhanced among children with blood cancers after transplantation with two units of umbilical cord blood versus an adequately dosed single-unit transplant.
Among 224 children in a phase III study, overall survival at 1 year was 71% with a single-unit umbilical cord blood (UCB) transplant and 65% with a double-unit UCB transplant (P = .13).
All other outcomes were similar, except for a lower incidence of platelet recovery and higher incidence of grade III-IV acute graft-versus-host disease after double UCB transplants, Dr. John E. Wagner reported on behalf of the Blood and Marrow Transplant Clinical Trials Network at the annual meeting of the American Society of Hematology.
And for patients who don’t have an adequate single cord unit based on cell dose, "a double cord blood unit was certainly as good as, or an acceptable alternative, to the single," he said. "So it allowed us to extend the transplant experience."
One of the major shortcomings of UCB transplantation is that units containing a minimum of 2.5 x 107 total nucleated cells/kg of patient body weight are frequently unavailable. The optimal number of transplanted umbilical stem cells remains unclear, but cell dose is an important factor influencing engraftment, or the ability of cells to take root, and survival.
Pilot studies suggested that infusion of two partially HLA-matched UCB units is safe, leading investigators to hypothesize that higher cell doses achieved with double UCB transplant would translate into improved survival, explained Dr. Wagner, scientific director of clinical research for the adult and pediatric blood and bone marrow transplant program at the University of Minnesota Medical Center in Minneapolis.
Between December 2006 and February 2012, investigators at 32 transplant centers enrolled 224 patients, aged 1-21 years, with acute lymphoblastic leukemia (ALL), acute myeloid leukemia, natural killer cell leukemia, chronic myeloid leukemia, or myelodysplastic syndrome. All had at least two available UCB units – each at least 4 of 6 HLA-matched to the patient and 3 of 6 matched between units.
Patients were randomized to receive a conventional single UCB (n = 113) or double UCB (n = 111) transplant. In all, 2.7% of the single UCB group and 1% of the double UCB group crossed over to the opposite arm. Only 1.8% of patients in both groups were not transplanted.
The groups were well matched for age, gender, race, performance status, and disease status. A little more than half of all patients had ALL.
After a median follow-up of 25 months, 92% of patients were in remission, with 60% of these in a second or subsequent remission.
Neutrophil counts had recovered at day 42 in 86% of the double UCB group and in 89% of the single UCB group (P = .08). Platelet recovery at 6 months was significantly lower after double UCB transplant (66% vs. 76%; P = .04), Dr. Wagner said.
Rates were nearly identical in the single and double UCB groups for grade II-IV acute graft-versus-host-disease at 100 days (56% vs. 57%; P = .88) and for chronic GVHD at 1 year (28% vs. 31%; P = .60), although a subpopulation in the double UCB group was significantly more likely to experience grade III-IV acute GVHD (23% vs. 14%; P = .04).
More importantly, there was a very low risk of transplant-related relapse after double or single UCB transplant (14% vs. 12%; P = .37), he observed.
Disease-free survival at 1 year was also similar in the double and single UCB groups at 64% vs. 68% (P = .22).
Dr. Wagner said the average total nucleated cell doses of 3.9 x 107 in the single UCB group and 7.2 x 107 in the double UCB group were not adequate for detecting differences in overall survival. "It probably will take a much greater dose escalation to be able to see that [difference]," he said.
Still, he noted that the overall survival and engraftment rates seen in both arms are superior to what was expected based on historical data sets, including the Cord Blood Transplantation Study (COBLT). It reported cumulative incidences for neutrophil recovery and platelet engraftment of 80% and 50%, and an overall survival of 67.4% in children with hematologic malignancies, despite a median cell dose of 5.1 x 107 (Blood 2008;112:4318-27).
One possible explanation, he suggested, is a new conditioning regimen that includes 1,320 cGy of total body irradiation and cyclophosphamide 120 mg/kg, but replaces the equine antithymocyte globulin used in the COBLT study with fludarabine (Fludara) 75 mg/m2. All patients also received cyclosporine and mycophenolate mofetil for GVHD prophylaxis.
"What this showed us is that it’s not just receiving a double perhaps that has some benefit for some patients, but also the conditioning regimen that we had changed simultaneously at the University of Minnesota," Dr. Wagner said.
He also highlighted similar findings from a very recent study he coauthored that reported comparable adjusted risks of neutrophil recovery, transplant-related mortality, and overall mortality after double UCB and adequately dosed single UCB transplants in 409 adults with acute leukemia (Blood 2012 Dec. 9 [Epub ahead of print]).
The study was supported by the National Heart, Lung, and Blood Institute; the National Cancer Institute; and the Children’s Oncology Group. Dr. Wagner and his coauthors reported no relevant conflicts of interest.
AT THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF HEMATOLOGY
Major Finding: Overall survival at 1 year after umbilical cord blood transplant was 71% with a single-unit transplant and 65% with a double-unit transplant (P = .13).
Data Source: Prospective, randomized phase III trial in 224 children with hematologic malignancies.
Disclosures: The study was supported by the National Heart, Lung, and Blood Institute; the National Cancer Institute; and the Children’s Oncology Group. Dr. Wagner and his coauthors reported no relevant conflicts of interest.
Targeted CT follow-up for incidental lung nodules?
CHICAGO – Limiting CT follow-up of incidental pulmonary nodules to a small area around the suspicious nodule may help resolve a common clinical dilemma and an Achilles heel of lung cancer screening, researchers suggest.
It could also reduce the radiation dose by more than 80%.
"Why should we radiate the entire thorax on follow-up?" Dr. Gregory D. Pearson asked rhetorically at the annual meeting of the Radiological Society of North America.
In the pivotal National Lung Cancer Screening Trial, annual low-dose CT reduced lung cancer deaths by 20% and all-cause mortality by 6.7% among heavy smokers, compared with traditional x-rays. Approximately 40% of the CT group, however, had findings of small, indeterminate pulmonary nodules considered suspicious for lung cancer on at least one scan, with 1.4% experiencing a complication as a result of additional testing (N. Engl. J. Med. 2011;365:395-409).
Several societies are working to devise the best protocol for further workup of detected nodules, but none have come up with the solution. The Fleischner Society, an international society for thoracic radiology, previously proposed serial follow-up imaging at intervals of less than 1 year for solid nodules smaller than 8 mm detected incidentally at nonscreening CT (Radiology 2005;237:395-400). For all semisolid nodules and ground-glass nodules at least 5 mm in size, researchers recommended a 3-month follow-up (Radiology 2005;237:395-400).
Follow-up CT scans, however, add costs and contribute to a patient’s cumulative radiation burden, said Dr. Pearson, a thoracic radiologist at New York–Presbyterian Hospital/Columbia University in New York City.
In an effort to develop a more practical solution, Dr. Pearson and his colleagues analyzed the variability in nodule location among 50 patients with subcentimeter nodules identified on CT screening for lung cancer and emphysema, and then devised a protocol for targeted CT follow-up that was validated in 50 additional patients.
Two experts and one novice reader, a medical student, independently measured the distances of the nodules from two anatomical landmarks, the lung apex and carina, on baseline and follow-up scans by cross-referencing the axial images with the CT scanogram on a PACS (picture archiving and communication system) workstation.
The interobserver variability was quite low, with mean differences of just 2-3 mm when the two experts were paired together or individually paired with the novice reader, Dr. Pearson said. Results were significantly better measuring from the apex than from the carina for two of the three reader pairings (P = .005 and .03), although the absolute difference was just 1 mm.
To allow for a greater margin of error, measurements in the validation phase of the study were only from the ipsilateral lung apex and covered a 60-mm range. At baseline, the expert identified the nodule and placed a cut line on the scan to measure the distance from the apex.
Medical students then measured the distance of the nodule from the apex on the follow-up scan, and reviewed a region of about 6 cm to determine whether the nodule would have been included in its entirety if the range of the CT scan had been narrowed to just 60 mm.
"The results here are pretty simple; 100% of the nodules in follow-up were scanned in their entirety," Dr. Pearson said.
The average craniocaudal coverage on follow-up CT was 363 mm.
By using a targeted 60-mm range, the average craniocaudal coverage and radiation dose would be reduced by 83%, although the actual dose reduction will depend on the location of the nodules, he added.
Still, the technique needs to be validated prospectively in larger groups, and would require buy-in from radiologists, medical societies, and Medicare and private insurers, Dr. Pearson said.
Session comoderator Dr. H. Page McAdams, division chief of cardiac and thoracic imaging at Duke University in Durham, N.C., said in an interview that the concept is intriguing, but raises a number of potential societal, reimbursement, and medical-legal issues.
Roughly 75% of the 100 consecutive patients in the study had a single nodule, Dr. Pearson told this publication.
Dr. McAdams reported grants from several imaging companies.
CHICAGO – Limiting CT follow-up of incidental pulmonary nodules to a small area around the suspicious nodule may help resolve a common clinical dilemma and an Achilles heel of lung cancer screening, researchers suggest.
It could also reduce the radiation dose by more than 80%.
"Why should we radiate the entire thorax on follow-up?" Dr. Gregory D. Pearson asked rhetorically at the annual meeting of the Radiological Society of North America.
In the pivotal National Lung Cancer Screening Trial, annual low-dose CT reduced lung cancer deaths by 20% and all-cause mortality by 6.7% among heavy smokers, compared with traditional x-rays. Approximately 40% of the CT group, however, had findings of small, indeterminate pulmonary nodules considered suspicious for lung cancer on at least one scan, with 1.4% experiencing a complication as a result of additional testing (N. Engl. J. Med. 2011;365:395-409).
Several societies are working to devise the best protocol for further workup of detected nodules, but none have come up with the solution. The Fleischner Society, an international society for thoracic radiology, previously proposed serial follow-up imaging at intervals of less than 1 year for solid nodules smaller than 8 mm detected incidentally at nonscreening CT (Radiology 2005;237:395-400). For all semisolid nodules and ground-glass nodules at least 5 mm in size, researchers recommended a 3-month follow-up (Radiology 2005;237:395-400).
Follow-up CT scans, however, add costs and contribute to a patient’s cumulative radiation burden, said Dr. Pearson, a thoracic radiologist at New York–Presbyterian Hospital/Columbia University in New York City.
In an effort to develop a more practical solution, Dr. Pearson and his colleagues analyzed the variability in nodule location among 50 patients with subcentimeter nodules identified on CT screening for lung cancer and emphysema, and then devised a protocol for targeted CT follow-up that was validated in 50 additional patients.
Two experts and one novice reader, a medical student, independently measured the distances of the nodules from two anatomical landmarks, the lung apex and carina, on baseline and follow-up scans by cross-referencing the axial images with the CT scanogram on a PACS (picture archiving and communication system) workstation.
The interobserver variability was quite low, with mean differences of just 2-3 mm when the two experts were paired together or individually paired with the novice reader, Dr. Pearson said. Results were significantly better measuring from the apex than from the carina for two of the three reader pairings (P = .005 and .03), although the absolute difference was just 1 mm.
To allow for a greater margin of error, measurements in the validation phase of the study were only from the ipsilateral lung apex and covered a 60-mm range. At baseline, the expert identified the nodule and placed a cut line on the scan to measure the distance from the apex.
Medical students then measured the distance of the nodule from the apex on the follow-up scan, and reviewed a region of about 6 cm to determine whether the nodule would have been included in its entirety if the range of the CT scan had been narrowed to just 60 mm.
"The results here are pretty simple; 100% of the nodules in follow-up were scanned in their entirety," Dr. Pearson said.
The average craniocaudal coverage on follow-up CT was 363 mm.
By using a targeted 60-mm range, the average craniocaudal coverage and radiation dose would be reduced by 83%, although the actual dose reduction will depend on the location of the nodules, he added.
Still, the technique needs to be validated prospectively in larger groups, and would require buy-in from radiologists, medical societies, and Medicare and private insurers, Dr. Pearson said.
Session comoderator Dr. H. Page McAdams, division chief of cardiac and thoracic imaging at Duke University in Durham, N.C., said in an interview that the concept is intriguing, but raises a number of potential societal, reimbursement, and medical-legal issues.
Roughly 75% of the 100 consecutive patients in the study had a single nodule, Dr. Pearson told this publication.
Dr. McAdams reported grants from several imaging companies.
CHICAGO – Limiting CT follow-up of incidental pulmonary nodules to a small area around the suspicious nodule may help resolve a common clinical dilemma and an Achilles heel of lung cancer screening, researchers suggest.
It could also reduce the radiation dose by more than 80%.
"Why should we radiate the entire thorax on follow-up?" Dr. Gregory D. Pearson asked rhetorically at the annual meeting of the Radiological Society of North America.
In the pivotal National Lung Cancer Screening Trial, annual low-dose CT reduced lung cancer deaths by 20% and all-cause mortality by 6.7% among heavy smokers, compared with traditional x-rays. Approximately 40% of the CT group, however, had findings of small, indeterminate pulmonary nodules considered suspicious for lung cancer on at least one scan, with 1.4% experiencing a complication as a result of additional testing (N. Engl. J. Med. 2011;365:395-409).
Several societies are working to devise the best protocol for further workup of detected nodules, but none have come up with the solution. The Fleischner Society, an international society for thoracic radiology, previously proposed serial follow-up imaging at intervals of less than 1 year for solid nodules smaller than 8 mm detected incidentally at nonscreening CT (Radiology 2005;237:395-400). For all semisolid nodules and ground-glass nodules at least 5 mm in size, researchers recommended a 3-month follow-up (Radiology 2005;237:395-400).
Follow-up CT scans, however, add costs and contribute to a patient’s cumulative radiation burden, said Dr. Pearson, a thoracic radiologist at New York–Presbyterian Hospital/Columbia University in New York City.
In an effort to develop a more practical solution, Dr. Pearson and his colleagues analyzed the variability in nodule location among 50 patients with subcentimeter nodules identified on CT screening for lung cancer and emphysema, and then devised a protocol for targeted CT follow-up that was validated in 50 additional patients.
Two experts and one novice reader, a medical student, independently measured the distances of the nodules from two anatomical landmarks, the lung apex and carina, on baseline and follow-up scans by cross-referencing the axial images with the CT scanogram on a PACS (picture archiving and communication system) workstation.
The interobserver variability was quite low, with mean differences of just 2-3 mm when the two experts were paired together or individually paired with the novice reader, Dr. Pearson said. Results were significantly better measuring from the apex than from the carina for two of the three reader pairings (P = .005 and .03), although the absolute difference was just 1 mm.
To allow for a greater margin of error, measurements in the validation phase of the study were only from the ipsilateral lung apex and covered a 60-mm range. At baseline, the expert identified the nodule and placed a cut line on the scan to measure the distance from the apex.
Medical students then measured the distance of the nodule from the apex on the follow-up scan, and reviewed a region of about 6 cm to determine whether the nodule would have been included in its entirety if the range of the CT scan had been narrowed to just 60 mm.
"The results here are pretty simple; 100% of the nodules in follow-up were scanned in their entirety," Dr. Pearson said.
The average craniocaudal coverage on follow-up CT was 363 mm.
By using a targeted 60-mm range, the average craniocaudal coverage and radiation dose would be reduced by 83%, although the actual dose reduction will depend on the location of the nodules, he added.
Still, the technique needs to be validated prospectively in larger groups, and would require buy-in from radiologists, medical societies, and Medicare and private insurers, Dr. Pearson said.
Session comoderator Dr. H. Page McAdams, division chief of cardiac and thoracic imaging at Duke University in Durham, N.C., said in an interview that the concept is intriguing, but raises a number of potential societal, reimbursement, and medical-legal issues.
Roughly 75% of the 100 consecutive patients in the study had a single nodule, Dr. Pearson told this publication.
Dr. McAdams reported grants from several imaging companies.
Major Finding: By using a targeted 60-mm range during CT follow-up of suspicious nodules, the average craniocaudal coverage and radiation dose would be reduced by 83%.
Data Source: Two-phase validation study in 100 consecutive patients with incidental subcentimeter lung nodules.
Disclosures: Dr. McAdams reported a research grant from General Electric, serving as a consultant for American College of Radiology Image Metrix, and working as an author for Reed Elsevier and UpToDate Inc.
Crizotinib changes practice for ALK-positive NSCLC
VIENNA – Long-awaited data from the phase III PROFILE 1007 confirm that crizotinib provides superior progression-free survival and responses, compared with second-line chemo-therapy in advanced anaplastic lymphoma kinase–positive non–small cell lung cancer.
Median progression-free survival more than doubled from 3.0 months with single-agent chemotherapy to 7.7 months with crizotinib, according to an independent radiologic review (P value less than .0001; hazard ratio, 0.49). Crizotinib (Xalkori) remained superior regardless of whether chemotherapy contained docetaxel (Taxotere) (7.7 vs. 2.6 months; P less than .0001) or pemetrexed (Alimta) (7.7 vs. 4.2; P = .0004), an agent previously shown to be effective against ALK-positive NSCLC.
The overall response rate was 65.3% for crizotinib and 19.5% for chemotherapy in the intent to treat population of 347 patients (overall response rate ratio 3.4; P less than .0001).
Crizotinib was also associated with significantly greater improvement in lung cancer symptoms and quality of life, Dr. Alice Shaw said at the European Society for Medical Oncology Congress. "Taken together, these results establish crizotinib as the standard of care for patients with advanced, previously treated ALK-positive non–small cell lung cancer."
ALK rearrangements are present in about 5% of lung cancers, typically in younger, never smokers.
Overall survival in the study was 22.8 months for chemotherapy and 20.3 months for crizotinib (P = .5394; HR, 1.02). The interim survival analysis was immature with just 40% of expected deaths reported and likely confounded by the high number (87%) of chemotherapy patients who crossed over to crizotinib after progression, she noted. After adjusting for crossover, the hazard ratio suggests a survival advantage with crizotinib (HR, 0.83).
Discussant Jean-Charles Soria of Institut Gustave Roussy, Villejuif, France, agreed and said the survival times in either arm were impressive, observing that just 2 years ago survival in second-line ALK-positive NSCLC was just 9 months.
Crizotinib, an oral, first in class ALK inhibitor, was given accelerated approval in 2011 in the United States to treat advanced ALK-positive NSCLC but is not approved in Europe, where regulatory agencies have required data from the randomized trial.
Dr. Soria said that worldwide use of crizotinib will require that several financial and practical issues surrounding implementation of molecular testing in daily practice be addressed including the optimal technique, type of sample, and tissue availability.
Testing for epidermal growth factor receptor, another molecular alteration that directs targeted therapy in lung cancer, "should not compete with ALK," he said, adding that multiplexing test strategies "are key."
Investigators at 105 sites across 21 countries randomized 173 patients to crizotinib 250 mg twice-daily in a 21-day cycle and 174 patients to chemotherapy containing pemetrexed 500 mg/m2 or docetaxel 75 mg/m2 given intravenously on day 1 of a 21-day cycle.
Treatment duration varied significantly, with patients receiving a median of 11 cycles of crizotinib vs. 4 cycles of chemotherapy. This may have influenced the higher number of all-cause deaths among crizotinib patients (25 deaths vs. 7 deaths), said Dr. Shaw, a thoracic oncologist at Massachusetts General Hospital Cancer Center in Boston.
Crizotinib patients were more likely than chemotherapy patients to experience visual disturbances (any grade 60% vs. 9%), as well as diarrhea, nausea, elevated transaminases (16% grade 3/4 ), edema, upper respiratory infection, dysgeusia, and dizziness. In contrast, fatigue, alopecia, dyspnea, and rash were more common with chemotherapy.
Despite the fact that patients on crizotinib experienced more nausea and vomiting, antiemetic use was significantly higher in the chemotherapy arm (67% vs. 20%). Based on the EORTC Quality of Life Questionnaire (QLQ C-30) and QLQ-LC 13, crizotinib patients had greater improvement from baseline in cough, dyspnea, fatigue, alopecia, insomnia, and pain as well as global quality of life (both P less than .0001).
Dr. Soria reported financial relationshisps with several firms including Pfizer, the study sponsor.
VIENNA – Long-awaited data from the phase III PROFILE 1007 confirm that crizotinib provides superior progression-free survival and responses, compared with second-line chemo-therapy in advanced anaplastic lymphoma kinase–positive non–small cell lung cancer.
Median progression-free survival more than doubled from 3.0 months with single-agent chemotherapy to 7.7 months with crizotinib, according to an independent radiologic review (P value less than .0001; hazard ratio, 0.49). Crizotinib (Xalkori) remained superior regardless of whether chemotherapy contained docetaxel (Taxotere) (7.7 vs. 2.6 months; P less than .0001) or pemetrexed (Alimta) (7.7 vs. 4.2; P = .0004), an agent previously shown to be effective against ALK-positive NSCLC.
The overall response rate was 65.3% for crizotinib and 19.5% for chemotherapy in the intent to treat population of 347 patients (overall response rate ratio 3.4; P less than .0001).
Crizotinib was also associated with significantly greater improvement in lung cancer symptoms and quality of life, Dr. Alice Shaw said at the European Society for Medical Oncology Congress. "Taken together, these results establish crizotinib as the standard of care for patients with advanced, previously treated ALK-positive non–small cell lung cancer."
ALK rearrangements are present in about 5% of lung cancers, typically in younger, never smokers.
Overall survival in the study was 22.8 months for chemotherapy and 20.3 months for crizotinib (P = .5394; HR, 1.02). The interim survival analysis was immature with just 40% of expected deaths reported and likely confounded by the high number (87%) of chemotherapy patients who crossed over to crizotinib after progression, she noted. After adjusting for crossover, the hazard ratio suggests a survival advantage with crizotinib (HR, 0.83).
Discussant Jean-Charles Soria of Institut Gustave Roussy, Villejuif, France, agreed and said the survival times in either arm were impressive, observing that just 2 years ago survival in second-line ALK-positive NSCLC was just 9 months.
Crizotinib, an oral, first in class ALK inhibitor, was given accelerated approval in 2011 in the United States to treat advanced ALK-positive NSCLC but is not approved in Europe, where regulatory agencies have required data from the randomized trial.
Dr. Soria said that worldwide use of crizotinib will require that several financial and practical issues surrounding implementation of molecular testing in daily practice be addressed including the optimal technique, type of sample, and tissue availability.
Testing for epidermal growth factor receptor, another molecular alteration that directs targeted therapy in lung cancer, "should not compete with ALK," he said, adding that multiplexing test strategies "are key."
Investigators at 105 sites across 21 countries randomized 173 patients to crizotinib 250 mg twice-daily in a 21-day cycle and 174 patients to chemotherapy containing pemetrexed 500 mg/m2 or docetaxel 75 mg/m2 given intravenously on day 1 of a 21-day cycle.
Treatment duration varied significantly, with patients receiving a median of 11 cycles of crizotinib vs. 4 cycles of chemotherapy. This may have influenced the higher number of all-cause deaths among crizotinib patients (25 deaths vs. 7 deaths), said Dr. Shaw, a thoracic oncologist at Massachusetts General Hospital Cancer Center in Boston.
Crizotinib patients were more likely than chemotherapy patients to experience visual disturbances (any grade 60% vs. 9%), as well as diarrhea, nausea, elevated transaminases (16% grade 3/4 ), edema, upper respiratory infection, dysgeusia, and dizziness. In contrast, fatigue, alopecia, dyspnea, and rash were more common with chemotherapy.
Despite the fact that patients on crizotinib experienced more nausea and vomiting, antiemetic use was significantly higher in the chemotherapy arm (67% vs. 20%). Based on the EORTC Quality of Life Questionnaire (QLQ C-30) and QLQ-LC 13, crizotinib patients had greater improvement from baseline in cough, dyspnea, fatigue, alopecia, insomnia, and pain as well as global quality of life (both P less than .0001).
Dr. Soria reported financial relationshisps with several firms including Pfizer, the study sponsor.
VIENNA – Long-awaited data from the phase III PROFILE 1007 confirm that crizotinib provides superior progression-free survival and responses, compared with second-line chemo-therapy in advanced anaplastic lymphoma kinase–positive non–small cell lung cancer.
Median progression-free survival more than doubled from 3.0 months with single-agent chemotherapy to 7.7 months with crizotinib, according to an independent radiologic review (P value less than .0001; hazard ratio, 0.49). Crizotinib (Xalkori) remained superior regardless of whether chemotherapy contained docetaxel (Taxotere) (7.7 vs. 2.6 months; P less than .0001) or pemetrexed (Alimta) (7.7 vs. 4.2; P = .0004), an agent previously shown to be effective against ALK-positive NSCLC.
The overall response rate was 65.3% for crizotinib and 19.5% for chemotherapy in the intent to treat population of 347 patients (overall response rate ratio 3.4; P less than .0001).
Crizotinib was also associated with significantly greater improvement in lung cancer symptoms and quality of life, Dr. Alice Shaw said at the European Society for Medical Oncology Congress. "Taken together, these results establish crizotinib as the standard of care for patients with advanced, previously treated ALK-positive non–small cell lung cancer."
ALK rearrangements are present in about 5% of lung cancers, typically in younger, never smokers.
Overall survival in the study was 22.8 months for chemotherapy and 20.3 months for crizotinib (P = .5394; HR, 1.02). The interim survival analysis was immature with just 40% of expected deaths reported and likely confounded by the high number (87%) of chemotherapy patients who crossed over to crizotinib after progression, she noted. After adjusting for crossover, the hazard ratio suggests a survival advantage with crizotinib (HR, 0.83).
Discussant Jean-Charles Soria of Institut Gustave Roussy, Villejuif, France, agreed and said the survival times in either arm were impressive, observing that just 2 years ago survival in second-line ALK-positive NSCLC was just 9 months.
Crizotinib, an oral, first in class ALK inhibitor, was given accelerated approval in 2011 in the United States to treat advanced ALK-positive NSCLC but is not approved in Europe, where regulatory agencies have required data from the randomized trial.
Dr. Soria said that worldwide use of crizotinib will require that several financial and practical issues surrounding implementation of molecular testing in daily practice be addressed including the optimal technique, type of sample, and tissue availability.
Testing for epidermal growth factor receptor, another molecular alteration that directs targeted therapy in lung cancer, "should not compete with ALK," he said, adding that multiplexing test strategies "are key."
Investigators at 105 sites across 21 countries randomized 173 patients to crizotinib 250 mg twice-daily in a 21-day cycle and 174 patients to chemotherapy containing pemetrexed 500 mg/m2 or docetaxel 75 mg/m2 given intravenously on day 1 of a 21-day cycle.
Treatment duration varied significantly, with patients receiving a median of 11 cycles of crizotinib vs. 4 cycles of chemotherapy. This may have influenced the higher number of all-cause deaths among crizotinib patients (25 deaths vs. 7 deaths), said Dr. Shaw, a thoracic oncologist at Massachusetts General Hospital Cancer Center in Boston.
Crizotinib patients were more likely than chemotherapy patients to experience visual disturbances (any grade 60% vs. 9%), as well as diarrhea, nausea, elevated transaminases (16% grade 3/4 ), edema, upper respiratory infection, dysgeusia, and dizziness. In contrast, fatigue, alopecia, dyspnea, and rash were more common with chemotherapy.
Despite the fact that patients on crizotinib experienced more nausea and vomiting, antiemetic use was significantly higher in the chemotherapy arm (67% vs. 20%). Based on the EORTC Quality of Life Questionnaire (QLQ C-30) and QLQ-LC 13, crizotinib patients had greater improvement from baseline in cough, dyspnea, fatigue, alopecia, insomnia, and pain as well as global quality of life (both P less than .0001).
Dr. Soria reported financial relationshisps with several firms including Pfizer, the study sponsor.
Major Finding: Median progression-free survival was 3.0 months with chemotherapy and 7.7 months with crizotinib (P less than .0001; hazard ratio 0.49).
Data Source: Results came from a phase III study involving 318 patients with advanced ALK-positive non–small cell lung cancer.
Disclosures: Dr. Shaw reported an advisory relationship with Pfizer, Ariad, Chugai, Novartis, and Daiichi-Sankyo and research funding from AstraZeneca and Novartis.
MLN9708 regimen effective with reduced neuropathy risk in multiple myeloma
ATLANTA – Of 65 patients with newly diagnosed multiple myeloma, nearly all had a partial or better response to an all-oral regimen containing the experimental proteasome inhibitor MLN9708.
In the phase I/II study, the overall response rate was 92%. A complete response occurred in 23% on MLN9708 plus lenalidomide (Revlimid) and dexamethasone (Decadron), and 55% had at least a very good partial response.
Importantly, neuropathy was minimal with the regimen. Grade 1 neuropathy occurred in 13 of the 65 patients (20%); 6 (9%) had grade 2 and 2 (3%) had grade 3 neuropathy, Dr. Shaji K. Kumar said at the annual meeting of the American Society of Hematology.
In contrast, the combination of lenalidomide, bortezomib (Velcade), and dexamethasone produced a partial response or better in 100% of newly diagnosed patients with multiple myeloma in a phase II study, but 80% of them experienced neuropathy, including grade 2 in 27% and grade 3 in 32% (Blood 2010;116:679-86).
“When the MLN9708 was developed, it was clear that it has reduced neurotoxicity compared with what we have seen historically with Velcade, and secondly, it is available orally, so you can just take a pill a day rather than having to go into a physician’s office and get an injection,” Dr. Kumar, professor of medicine at Mayo Clinic in Rochester, Minn., said in an interview.
Based on the promising results, weekly and twice-weekly MLN9708 is being evaluated as a single agent and in combinations for the treatment of relapsed and newly diagnosed multiple myeloma, including as part of a phase III trial of MLN9708 plus lenalidomide and dexamethasone versus lenalidomide plus dexamethasone. The trial is currently enrolling adults with relapsed and/or refractory disease (NCT01564537). “MLN9708 is perfectly placed to be able to be combined in regimens that are highly efficacious, but at the same time are better tolerated,” he added.
In the current study, 65 patients received MLN9708 on days 1, 8, and 15; lenalidomide 25 mg on days 1-21; and dexamethasone 40 mg on days 1, 8, and 15 for up to 12 cycles of 28 days, and were maintained on MLN9708 on days 1, 8, and 15 until disease progression or toxicity. The maximum tolerated dose of MLN9708 was identified in 15 patients in phase I as 2.97 mg/m2. A fixed dose of 4 mg weekly, equivalent to 2.23 mg/m2, was given to another 50 patients in phase II.
Patients also received mandatory thromboprophylaxis with aspirin or low-molecular-weight heparin. Patients received a median of 6 cycles of MLN9708 in phase I (range, 1-19) and 7 (range, 1-19) in phase II. Their median age was 66 years, and 25% had unfavorable cytogenetics. The median time to first response was one cycle, and the median duration of response was not reached. Responses increased with the number of cycles and deepened over time, Dr. Kumar said.
Complete responses were reported in 19% of 47 patients receiving 4 cycles, very good partial responses in 30%, and partial responses in 45% (overall response rate, 94%), improving after 8 cycles in 19 patients to response rates of 32%, 26%, and 37%, respectively (overall response rate, 95%). Of three evaluable patients who completed 12 cycles, two achieved a complete response and one had a very good partial response.
“So we are fairly confident we will see an increase in response over time,” he said, noting that similar responses were seen in patients with favorable and unfavorable cytogenetics. At the time of the analysis, 27 of the 65 patients remained on treatment, 20 had proceeded to stem cell transplantation, 3 patients progressed, and 1 patient died after cycle 4, possibly related to treatment or alternatively to their underlying disease state, Dr. Kumar said.
The estimated 1-year progression-free survival probability was 93%. Seven additional patients discontinued therapy because of adverse events, including four due to treatment-related AEs. Serious AEs were seen in about 20% of patients, with grade 3 neutropenia and rash being the most common. The most common side effects of any grade were rash (68%), fatigue (48%), nausea (42%), vomiting (32%), and diarrhea (38%). Side effects were manageable with dose modifications and supportive care, Dr. Kumar said.
MLN9708 plus lenalidomide and dexamethasone did not appear to have an adverse impact on stem cell mobilization, with a median of 11.3 x 106/L CD34-positive cells harvested (range, 5-28 x 106/L). Dr. Kumar reported research funding from study sponsor Millennium Pharmaceuticals. His coauthors reported commercial relationships including employment with Millennium.
ATLANTA – Of 65 patients with newly diagnosed multiple myeloma, nearly all had a partial or better response to an all-oral regimen containing the experimental proteasome inhibitor MLN9708.
In the phase I/II study, the overall response rate was 92%. A complete response occurred in 23% on MLN9708 plus lenalidomide (Revlimid) and dexamethasone (Decadron), and 55% had at least a very good partial response.
Importantly, neuropathy was minimal with the regimen. Grade 1 neuropathy occurred in 13 of the 65 patients (20%); 6 (9%) had grade 2 and 2 (3%) had grade 3 neuropathy, Dr. Shaji K. Kumar said at the annual meeting of the American Society of Hematology.
In contrast, the combination of lenalidomide, bortezomib (Velcade), and dexamethasone produced a partial response or better in 100% of newly diagnosed patients with multiple myeloma in a phase II study, but 80% of them experienced neuropathy, including grade 2 in 27% and grade 3 in 32% (Blood 2010;116:679-86).
“When the MLN9708 was developed, it was clear that it has reduced neurotoxicity compared with what we have seen historically with Velcade, and secondly, it is available orally, so you can just take a pill a day rather than having to go into a physician’s office and get an injection,” Dr. Kumar, professor of medicine at Mayo Clinic in Rochester, Minn., said in an interview.
Based on the promising results, weekly and twice-weekly MLN9708 is being evaluated as a single agent and in combinations for the treatment of relapsed and newly diagnosed multiple myeloma, including as part of a phase III trial of MLN9708 plus lenalidomide and dexamethasone versus lenalidomide plus dexamethasone. The trial is currently enrolling adults with relapsed and/or refractory disease (NCT01564537). “MLN9708 is perfectly placed to be able to be combined in regimens that are highly efficacious, but at the same time are better tolerated,” he added.
In the current study, 65 patients received MLN9708 on days 1, 8, and 15; lenalidomide 25 mg on days 1-21; and dexamethasone 40 mg on days 1, 8, and 15 for up to 12 cycles of 28 days, and were maintained on MLN9708 on days 1, 8, and 15 until disease progression or toxicity. The maximum tolerated dose of MLN9708 was identified in 15 patients in phase I as 2.97 mg/m2. A fixed dose of 4 mg weekly, equivalent to 2.23 mg/m2, was given to another 50 patients in phase II.
Patients also received mandatory thromboprophylaxis with aspirin or low-molecular-weight heparin. Patients received a median of 6 cycles of MLN9708 in phase I (range, 1-19) and 7 (range, 1-19) in phase II. Their median age was 66 years, and 25% had unfavorable cytogenetics. The median time to first response was one cycle, and the median duration of response was not reached. Responses increased with the number of cycles and deepened over time, Dr. Kumar said.
Complete responses were reported in 19% of 47 patients receiving 4 cycles, very good partial responses in 30%, and partial responses in 45% (overall response rate, 94%), improving after 8 cycles in 19 patients to response rates of 32%, 26%, and 37%, respectively (overall response rate, 95%). Of three evaluable patients who completed 12 cycles, two achieved a complete response and one had a very good partial response.
“So we are fairly confident we will see an increase in response over time,” he said, noting that similar responses were seen in patients with favorable and unfavorable cytogenetics. At the time of the analysis, 27 of the 65 patients remained on treatment, 20 had proceeded to stem cell transplantation, 3 patients progressed, and 1 patient died after cycle 4, possibly related to treatment or alternatively to their underlying disease state, Dr. Kumar said.
The estimated 1-year progression-free survival probability was 93%. Seven additional patients discontinued therapy because of adverse events, including four due to treatment-related AEs. Serious AEs were seen in about 20% of patients, with grade 3 neutropenia and rash being the most common. The most common side effects of any grade were rash (68%), fatigue (48%), nausea (42%), vomiting (32%), and diarrhea (38%). Side effects were manageable with dose modifications and supportive care, Dr. Kumar said.
MLN9708 plus lenalidomide and dexamethasone did not appear to have an adverse impact on stem cell mobilization, with a median of 11.3 x 106/L CD34-positive cells harvested (range, 5-28 x 106/L). Dr. Kumar reported research funding from study sponsor Millennium Pharmaceuticals. His coauthors reported commercial relationships including employment with Millennium.
ATLANTA – Of 65 patients with newly diagnosed multiple myeloma, nearly all had a partial or better response to an all-oral regimen containing the experimental proteasome inhibitor MLN9708.
In the phase I/II study, the overall response rate was 92%. A complete response occurred in 23% on MLN9708 plus lenalidomide (Revlimid) and dexamethasone (Decadron), and 55% had at least a very good partial response.
Importantly, neuropathy was minimal with the regimen. Grade 1 neuropathy occurred in 13 of the 65 patients (20%); 6 (9%) had grade 2 and 2 (3%) had grade 3 neuropathy, Dr. Shaji K. Kumar said at the annual meeting of the American Society of Hematology.
In contrast, the combination of lenalidomide, bortezomib (Velcade), and dexamethasone produced a partial response or better in 100% of newly diagnosed patients with multiple myeloma in a phase II study, but 80% of them experienced neuropathy, including grade 2 in 27% and grade 3 in 32% (Blood 2010;116:679-86).
“When the MLN9708 was developed, it was clear that it has reduced neurotoxicity compared with what we have seen historically with Velcade, and secondly, it is available orally, so you can just take a pill a day rather than having to go into a physician’s office and get an injection,” Dr. Kumar, professor of medicine at Mayo Clinic in Rochester, Minn., said in an interview.
Based on the promising results, weekly and twice-weekly MLN9708 is being evaluated as a single agent and in combinations for the treatment of relapsed and newly diagnosed multiple myeloma, including as part of a phase III trial of MLN9708 plus lenalidomide and dexamethasone versus lenalidomide plus dexamethasone. The trial is currently enrolling adults with relapsed and/or refractory disease (NCT01564537). “MLN9708 is perfectly placed to be able to be combined in regimens that are highly efficacious, but at the same time are better tolerated,” he added.
In the current study, 65 patients received MLN9708 on days 1, 8, and 15; lenalidomide 25 mg on days 1-21; and dexamethasone 40 mg on days 1, 8, and 15 for up to 12 cycles of 28 days, and were maintained on MLN9708 on days 1, 8, and 15 until disease progression or toxicity. The maximum tolerated dose of MLN9708 was identified in 15 patients in phase I as 2.97 mg/m2. A fixed dose of 4 mg weekly, equivalent to 2.23 mg/m2, was given to another 50 patients in phase II.
Patients also received mandatory thromboprophylaxis with aspirin or low-molecular-weight heparin. Patients received a median of 6 cycles of MLN9708 in phase I (range, 1-19) and 7 (range, 1-19) in phase II. Their median age was 66 years, and 25% had unfavorable cytogenetics. The median time to first response was one cycle, and the median duration of response was not reached. Responses increased with the number of cycles and deepened over time, Dr. Kumar said.
Complete responses were reported in 19% of 47 patients receiving 4 cycles, very good partial responses in 30%, and partial responses in 45% (overall response rate, 94%), improving after 8 cycles in 19 patients to response rates of 32%, 26%, and 37%, respectively (overall response rate, 95%). Of three evaluable patients who completed 12 cycles, two achieved a complete response and one had a very good partial response.
“So we are fairly confident we will see an increase in response over time,” he said, noting that similar responses were seen in patients with favorable and unfavorable cytogenetics. At the time of the analysis, 27 of the 65 patients remained on treatment, 20 had proceeded to stem cell transplantation, 3 patients progressed, and 1 patient died after cycle 4, possibly related to treatment or alternatively to their underlying disease state, Dr. Kumar said.
The estimated 1-year progression-free survival probability was 93%. Seven additional patients discontinued therapy because of adverse events, including four due to treatment-related AEs. Serious AEs were seen in about 20% of patients, with grade 3 neutropenia and rash being the most common. The most common side effects of any grade were rash (68%), fatigue (48%), nausea (42%), vomiting (32%), and diarrhea (38%). Side effects were manageable with dose modifications and supportive care, Dr. Kumar said.
MLN9708 plus lenalidomide and dexamethasone did not appear to have an adverse impact on stem cell mobilization, with a median of 11.3 x 106/L CD34-positive cells harvested (range, 5-28 x 106/L). Dr. Kumar reported research funding from study sponsor Millennium Pharmaceuticals. His coauthors reported commercial relationships including employment with Millennium.
Major Finding: The overall response rate was 92% including complete responses in 23% and at least a very good partial response in 55%.
Data Source: Phase I/II study in 65 patients with newly diagnosed multiple myeloma.
Disclosures: Dr. Kumar reported research funding from study sponsor Millennium Pharmaceuticals. His coauthors reported commercial relationships, including employment, with Millennium.