User login
BMN 673 monotherapy makes splash in BRCA-mutated ovarian, breast cancer
AMSTERDAM – The experimental PARP inhibitor BMN 673 had impressive single-agent activity in pretreated breast and ovarian cancer patients carrying BRCA 1 and 2 mutations, based on results from a small, ongoing phase I/II trial.
The objective response rate by RECIST (Response Evaluation Criteria in Solid Tumors) criteria was 44% for both the breast (8/18) and ovarian (11/25) cancer patients.
Responses were durable in both groups at a median of about 8 months, leading to a median progression-free survival of 32.3 weeks in ovarian cancer patients and 33.1 weeks in breast cancer patients, Dr. Ramesh Ramanathan reported in a late-breaking abstract at the multidisciplinary European cancer congresses.
"There’s very little to discuss here. This is a flat-out, very successful phase 1 trial," said Dr. Peter Dubsky, a surgeon with the Medical University of Vienna, who was invited to discuss the results.
He observed that toxicity with BMN 673, described in the abstract as the most potent and specific inhibitor of poly-ADP ribose polymerase (PARP) in clinical development, was lower than would be expected from a PARP inhibitor. In addition, "responses were seen starting at 100 mcg, but the maximum tolerated dose is 10 times that," Dr. Dubsky said.
The maximum tolerated dose of 1 mg/day is being used in a newly launched, open-label, 429-patient phase III trial, said Dr. Ramanathan, medical director of the Scottsdale (Ariz.) Healthcare Research Institute. The multicenter trial is evaluating BMN 673 against the physician’s choice of capecitabine (Xeloda), eribulin (Halaven), gemcitabine (Gemzar), or vinorelbine (Navelbine) in germline BRCA-mutation patients with locally advanced or metastatic breast cancer treated with no more than two prior chemotherapy regimens for metastatic disease.
The dose-escalation portion of the phase I study involved 80 patients dosed at 25-1,100 mcg/day. The study was then expanded and included 18 patients with germline BRCA-mutant breast cancer and 28 patients with germline BRCA-mutant ovarian cancer. Patients were dosed once daily at 100 mcg and above.
The heavily pretreated expansion cohort had received a median of three prior chemotherapy regimens (range, 0-8). BRCA 1 mutations were more common in ovarian cancer patients (20/28), and BRCA 2 mutations were more frequent in the breast group (11/18). Their median age was 58 years and 42 years, respectively.
Among 25 ovarian cancer patients evaluable by RECIST, 1 patient had a complete response, 10 had partial responses, and 4 had stable disease lasting 24 weeks or more, Dr. Ramanathan reported.
Responses were seen in 17 of 22 platinum-sensitive patients. "Platinum sensitivity may be important in identifying patients sensitive to BMN 673," he said.
Cancer antigen 125 levels evaluable in 27 ovarian cancer patients dropped by more than 50% in 19 (70%) and returned to the normal range in 9.
All 18 breast cancer patients were evaluable by RECIST, with a complete response in 1, partial responses in 7, and stable disease in 5. The clinical benefit response rate (complete response, partial response, or stable disease for at least 24 weeks) was 72% (13/18).
At the 1-mg/day phase III dose, however, the RECIST response rate reached 50% (7/14), and the clinical benefit response rate, 86% (12/14), Dr. Ramanathan said.
He noted that BMN 673 was generally well tolerated, with the most common drug-related toxicities being myelosuppression, fatigue, nausea, and alopecia occurring in less than 30% of patients. In the phase I portion of the trial, thrombocytopenia was dose limiting, occurring as a grade 3 event in 14 patients (17.5%) and a grade 4 event in 1 patient (1.3%). Grade 3 anemia was seen in 12 patients (15%).
BioMarin Pharmaceuticals funded the trial. Dr. Ramanathan reported clinical research and travel support for the presentation. His coauthors reported similar relationships as well as employment with BioMarin.
AMSTERDAM – The experimental PARP inhibitor BMN 673 had impressive single-agent activity in pretreated breast and ovarian cancer patients carrying BRCA 1 and 2 mutations, based on results from a small, ongoing phase I/II trial.
The objective response rate by RECIST (Response Evaluation Criteria in Solid Tumors) criteria was 44% for both the breast (8/18) and ovarian (11/25) cancer patients.
Responses were durable in both groups at a median of about 8 months, leading to a median progression-free survival of 32.3 weeks in ovarian cancer patients and 33.1 weeks in breast cancer patients, Dr. Ramesh Ramanathan reported in a late-breaking abstract at the multidisciplinary European cancer congresses.
"There’s very little to discuss here. This is a flat-out, very successful phase 1 trial," said Dr. Peter Dubsky, a surgeon with the Medical University of Vienna, who was invited to discuss the results.
He observed that toxicity with BMN 673, described in the abstract as the most potent and specific inhibitor of poly-ADP ribose polymerase (PARP) in clinical development, was lower than would be expected from a PARP inhibitor. In addition, "responses were seen starting at 100 mcg, but the maximum tolerated dose is 10 times that," Dr. Dubsky said.
The maximum tolerated dose of 1 mg/day is being used in a newly launched, open-label, 429-patient phase III trial, said Dr. Ramanathan, medical director of the Scottsdale (Ariz.) Healthcare Research Institute. The multicenter trial is evaluating BMN 673 against the physician’s choice of capecitabine (Xeloda), eribulin (Halaven), gemcitabine (Gemzar), or vinorelbine (Navelbine) in germline BRCA-mutation patients with locally advanced or metastatic breast cancer treated with no more than two prior chemotherapy regimens for metastatic disease.
The dose-escalation portion of the phase I study involved 80 patients dosed at 25-1,100 mcg/day. The study was then expanded and included 18 patients with germline BRCA-mutant breast cancer and 28 patients with germline BRCA-mutant ovarian cancer. Patients were dosed once daily at 100 mcg and above.
The heavily pretreated expansion cohort had received a median of three prior chemotherapy regimens (range, 0-8). BRCA 1 mutations were more common in ovarian cancer patients (20/28), and BRCA 2 mutations were more frequent in the breast group (11/18). Their median age was 58 years and 42 years, respectively.
Among 25 ovarian cancer patients evaluable by RECIST, 1 patient had a complete response, 10 had partial responses, and 4 had stable disease lasting 24 weeks or more, Dr. Ramanathan reported.
Responses were seen in 17 of 22 platinum-sensitive patients. "Platinum sensitivity may be important in identifying patients sensitive to BMN 673," he said.
Cancer antigen 125 levels evaluable in 27 ovarian cancer patients dropped by more than 50% in 19 (70%) and returned to the normal range in 9.
All 18 breast cancer patients were evaluable by RECIST, with a complete response in 1, partial responses in 7, and stable disease in 5. The clinical benefit response rate (complete response, partial response, or stable disease for at least 24 weeks) was 72% (13/18).
At the 1-mg/day phase III dose, however, the RECIST response rate reached 50% (7/14), and the clinical benefit response rate, 86% (12/14), Dr. Ramanathan said.
He noted that BMN 673 was generally well tolerated, with the most common drug-related toxicities being myelosuppression, fatigue, nausea, and alopecia occurring in less than 30% of patients. In the phase I portion of the trial, thrombocytopenia was dose limiting, occurring as a grade 3 event in 14 patients (17.5%) and a grade 4 event in 1 patient (1.3%). Grade 3 anemia was seen in 12 patients (15%).
BioMarin Pharmaceuticals funded the trial. Dr. Ramanathan reported clinical research and travel support for the presentation. His coauthors reported similar relationships as well as employment with BioMarin.
AMSTERDAM – The experimental PARP inhibitor BMN 673 had impressive single-agent activity in pretreated breast and ovarian cancer patients carrying BRCA 1 and 2 mutations, based on results from a small, ongoing phase I/II trial.
The objective response rate by RECIST (Response Evaluation Criteria in Solid Tumors) criteria was 44% for both the breast (8/18) and ovarian (11/25) cancer patients.
Responses were durable in both groups at a median of about 8 months, leading to a median progression-free survival of 32.3 weeks in ovarian cancer patients and 33.1 weeks in breast cancer patients, Dr. Ramesh Ramanathan reported in a late-breaking abstract at the multidisciplinary European cancer congresses.
"There’s very little to discuss here. This is a flat-out, very successful phase 1 trial," said Dr. Peter Dubsky, a surgeon with the Medical University of Vienna, who was invited to discuss the results.
He observed that toxicity with BMN 673, described in the abstract as the most potent and specific inhibitor of poly-ADP ribose polymerase (PARP) in clinical development, was lower than would be expected from a PARP inhibitor. In addition, "responses were seen starting at 100 mcg, but the maximum tolerated dose is 10 times that," Dr. Dubsky said.
The maximum tolerated dose of 1 mg/day is being used in a newly launched, open-label, 429-patient phase III trial, said Dr. Ramanathan, medical director of the Scottsdale (Ariz.) Healthcare Research Institute. The multicenter trial is evaluating BMN 673 against the physician’s choice of capecitabine (Xeloda), eribulin (Halaven), gemcitabine (Gemzar), or vinorelbine (Navelbine) in germline BRCA-mutation patients with locally advanced or metastatic breast cancer treated with no more than two prior chemotherapy regimens for metastatic disease.
The dose-escalation portion of the phase I study involved 80 patients dosed at 25-1,100 mcg/day. The study was then expanded and included 18 patients with germline BRCA-mutant breast cancer and 28 patients with germline BRCA-mutant ovarian cancer. Patients were dosed once daily at 100 mcg and above.
The heavily pretreated expansion cohort had received a median of three prior chemotherapy regimens (range, 0-8). BRCA 1 mutations were more common in ovarian cancer patients (20/28), and BRCA 2 mutations were more frequent in the breast group (11/18). Their median age was 58 years and 42 years, respectively.
Among 25 ovarian cancer patients evaluable by RECIST, 1 patient had a complete response, 10 had partial responses, and 4 had stable disease lasting 24 weeks or more, Dr. Ramanathan reported.
Responses were seen in 17 of 22 platinum-sensitive patients. "Platinum sensitivity may be important in identifying patients sensitive to BMN 673," he said.
Cancer antigen 125 levels evaluable in 27 ovarian cancer patients dropped by more than 50% in 19 (70%) and returned to the normal range in 9.
All 18 breast cancer patients were evaluable by RECIST, with a complete response in 1, partial responses in 7, and stable disease in 5. The clinical benefit response rate (complete response, partial response, or stable disease for at least 24 weeks) was 72% (13/18).
At the 1-mg/day phase III dose, however, the RECIST response rate reached 50% (7/14), and the clinical benefit response rate, 86% (12/14), Dr. Ramanathan said.
He noted that BMN 673 was generally well tolerated, with the most common drug-related toxicities being myelosuppression, fatigue, nausea, and alopecia occurring in less than 30% of patients. In the phase I portion of the trial, thrombocytopenia was dose limiting, occurring as a grade 3 event in 14 patients (17.5%) and a grade 4 event in 1 patient (1.3%). Grade 3 anemia was seen in 12 patients (15%).
BioMarin Pharmaceuticals funded the trial. Dr. Ramanathan reported clinical research and travel support for the presentation. His coauthors reported similar relationships as well as employment with BioMarin.
AT THE EUROPEAN CANCER CONFERENCE 2013
Major finding: The RECIST response rate was 44% for breast and ovarian cancer patients.
Data source: Ongoing phase I/II study in ovarian and breast cancer patients with BRCA 1 and 2 mutations.
Disclosures: BioMarin Pharmaceuticals funded the trial. Dr. Ramanathan reported clinical research and travel support for the presentation. His coauthors reported similar relationships as well as employment with BioMarin.
Breast Cancer Chemoprevention Recommendations: We’re in This Together
The U.S. Preventive Services Task Force has issued updated recommendations on the use of chemoprevention for breast cancer that emphasize the need for informed, shared decision-making.
"It’s a complicated decision, and there are a lot of moving parts, if you will," USPSTF member Dr. Mark Ebell said in an interview.
New research since the original 2002 guidance, including updates from the pivotal STAR trial (Cancer Prev. Res. 2010;3:696-706) and a recent meta-analysis of risk-reducing medications (Ann. Intern. Med. 2013;158:604-14), have helped clarify the balance of benefits and harms by a woman’s age, breast cancer risk, race, and whether she’s had a hysterectomy.
The 2013 guidelines, published in Annals of Internal Medicine, plug those data into a series of tables that present the net benefit as stratified by those risk factors. For example, Table 1 is for white, non-Hispanic women who have a uterus.
"So, you’d go there, look up [the patient’s] 5-year projected risk, look up [the patient’s] age, and it provides a general color-coded assessment of whether there are more risks or harms," Dr. Ebell said. "It’s a tough decision, and two women with similar risk factors may make different decisions, and that’s perfectly okay. Everyone’s different; everyone assesses risk differently and has different values. We just wanted to make sure they have the best possible information when they make their decision."
Both the original and updated versions carry the same overall recommendation: Women who are at an average or low risk should not use chemoprevention, while women at high risk should consider therapy if they don’t have any contraindications.
The 2013 recommendations are trying, however, to be a bit more explicit about which medications have been approved by the Food and Drug Administration (FDA) for breast cancer chemoprevention, said Dr. Ebell, a family physician with the College of Public Health, University of Georgia, Athens.
The USPSTF recommends the use of the selective estrogen receptor modulators tamoxifen and raloxifene (Evista) to reduce the incidence of invasive breast cancer in asymptomatic women aged 35 years and older who are without a prior diagnosis of breast cancer, ductal carcinoma in situ, or lobular carcinoma in situ and who are at increased risk of breast cancer. The daily doses are 20 mg and 60 mg, respectively, for 5 years.
Tamoxifen is FDA approved for this use in women aged 35 years and older, while raloxifene is approved for this indication in postmenopausal women.
In a draft version of the guidance released this April, the target audience for the recommendation was asymptomatic women aged 40 years and older, but the age limit was lowered to 35 years in the final version because the Breast Cancer Prevention Trial enrolled women over age 35, Dr. Ebell said.
Notably absent in the new guidelines is an endorsement of the aromatase inhibitor exemestane (Aromasin). Updated chemoprevention guidelines issued earlier this year by the American Society of Clinical Oncology recommend that exemestane be discussed as an alternative to reduce the risk of invasive, estrogen receptor–positive breast cancer in postmenopausal women (J. Clin. Onc. 2013;31:2942-62).
The task force only considers FDA-approved medications for the indication of breast cancer chemoprevention, and exemestane is only approved for breast cancer treatment, Dr. Ebell explained.
The aromatase inhibitor anastrozole (Arimidex) is not recommended by either ASCO or the USPSTF, with data awaited from the ongoing phase III, placebo-controlled British IBIS II (Anastrozole in Preventing Breast Cancer in Postmenopausal Women at Increased Risk of Breast Cancer) study.
Dr. Joanna Cain, professor of obstetrics/gynecology at the University of Massachusetts, Worcester, said the task force made the decision regarding exemestane on the basis of the present evidence.
"UPSTF has a level of standards that exemestane does not yet meet, while ASCO is willing to move with less evidence," she said in an interview. "They are not ruling it out but saying by their criteria, it does not yet meet this. Having said all that, if a patient is intolerant of tamoxifen and raloxifene and needs chemopreventive therapy, we would consider using exemestane as an alternative."
Dr. Cain noted that she welcomes the 2013 guidance, particularly the appropriate caution about individual patient risks for these medications such as age, venous thrombosis, and endometrial cancer, which also includes the risk conferred by obesity and genetics such as Lynch syndrome.
"In particular, the clarity around benefit, and therefore, use only for those at increased risk, is helpful," she said.
The use of risk-reducing medications is quite low, but that’s because only about 5% of women are really appropriate candidates and only a minority of these actually take the medication, Dr. Ebell said.
Just 12% of high-risk women opted to take tamoxifen to reduce their risk for breast cancer in a national survey highlighted by the task force, with 77% of women declining primarily because of concerns about serious adverse events and small therapeutic benefit (Arch. Intern. Med. 2006;166:2260-5).
Further, only 27% of the 350 primary care physicians surveyed had prescribed tamoxifen for breast cancer prevention at least once in the prior 12 months.
"We do need to engage the primary care community more broadly, not just ob.gyns., in this informed decision-making and make sure they are comfortable and confident when they have a patient with questions about chemoprevention," Dr. Ebell said.
Dr. Ebell reported having no financial disclosures.
The U.S. Preventive Services Task Force has issued updated recommendations on the use of chemoprevention for breast cancer that emphasize the need for informed, shared decision-making.
"It’s a complicated decision, and there are a lot of moving parts, if you will," USPSTF member Dr. Mark Ebell said in an interview.
New research since the original 2002 guidance, including updates from the pivotal STAR trial (Cancer Prev. Res. 2010;3:696-706) and a recent meta-analysis of risk-reducing medications (Ann. Intern. Med. 2013;158:604-14), have helped clarify the balance of benefits and harms by a woman’s age, breast cancer risk, race, and whether she’s had a hysterectomy.
The 2013 guidelines, published in Annals of Internal Medicine, plug those data into a series of tables that present the net benefit as stratified by those risk factors. For example, Table 1 is for white, non-Hispanic women who have a uterus.
"So, you’d go there, look up [the patient’s] 5-year projected risk, look up [the patient’s] age, and it provides a general color-coded assessment of whether there are more risks or harms," Dr. Ebell said. "It’s a tough decision, and two women with similar risk factors may make different decisions, and that’s perfectly okay. Everyone’s different; everyone assesses risk differently and has different values. We just wanted to make sure they have the best possible information when they make their decision."
Both the original and updated versions carry the same overall recommendation: Women who are at an average or low risk should not use chemoprevention, while women at high risk should consider therapy if they don’t have any contraindications.
The 2013 recommendations are trying, however, to be a bit more explicit about which medications have been approved by the Food and Drug Administration (FDA) for breast cancer chemoprevention, said Dr. Ebell, a family physician with the College of Public Health, University of Georgia, Athens.
The USPSTF recommends the use of the selective estrogen receptor modulators tamoxifen and raloxifene (Evista) to reduce the incidence of invasive breast cancer in asymptomatic women aged 35 years and older who are without a prior diagnosis of breast cancer, ductal carcinoma in situ, or lobular carcinoma in situ and who are at increased risk of breast cancer. The daily doses are 20 mg and 60 mg, respectively, for 5 years.
Tamoxifen is FDA approved for this use in women aged 35 years and older, while raloxifene is approved for this indication in postmenopausal women.
In a draft version of the guidance released this April, the target audience for the recommendation was asymptomatic women aged 40 years and older, but the age limit was lowered to 35 years in the final version because the Breast Cancer Prevention Trial enrolled women over age 35, Dr. Ebell said.
Notably absent in the new guidelines is an endorsement of the aromatase inhibitor exemestane (Aromasin). Updated chemoprevention guidelines issued earlier this year by the American Society of Clinical Oncology recommend that exemestane be discussed as an alternative to reduce the risk of invasive, estrogen receptor–positive breast cancer in postmenopausal women (J. Clin. Onc. 2013;31:2942-62).
The task force only considers FDA-approved medications for the indication of breast cancer chemoprevention, and exemestane is only approved for breast cancer treatment, Dr. Ebell explained.
The aromatase inhibitor anastrozole (Arimidex) is not recommended by either ASCO or the USPSTF, with data awaited from the ongoing phase III, placebo-controlled British IBIS II (Anastrozole in Preventing Breast Cancer in Postmenopausal Women at Increased Risk of Breast Cancer) study.
Dr. Joanna Cain, professor of obstetrics/gynecology at the University of Massachusetts, Worcester, said the task force made the decision regarding exemestane on the basis of the present evidence.
"UPSTF has a level of standards that exemestane does not yet meet, while ASCO is willing to move with less evidence," she said in an interview. "They are not ruling it out but saying by their criteria, it does not yet meet this. Having said all that, if a patient is intolerant of tamoxifen and raloxifene and needs chemopreventive therapy, we would consider using exemestane as an alternative."
Dr. Cain noted that she welcomes the 2013 guidance, particularly the appropriate caution about individual patient risks for these medications such as age, venous thrombosis, and endometrial cancer, which also includes the risk conferred by obesity and genetics such as Lynch syndrome.
"In particular, the clarity around benefit, and therefore, use only for those at increased risk, is helpful," she said.
The use of risk-reducing medications is quite low, but that’s because only about 5% of women are really appropriate candidates and only a minority of these actually take the medication, Dr. Ebell said.
Just 12% of high-risk women opted to take tamoxifen to reduce their risk for breast cancer in a national survey highlighted by the task force, with 77% of women declining primarily because of concerns about serious adverse events and small therapeutic benefit (Arch. Intern. Med. 2006;166:2260-5).
Further, only 27% of the 350 primary care physicians surveyed had prescribed tamoxifen for breast cancer prevention at least once in the prior 12 months.
"We do need to engage the primary care community more broadly, not just ob.gyns., in this informed decision-making and make sure they are comfortable and confident when they have a patient with questions about chemoprevention," Dr. Ebell said.
Dr. Ebell reported having no financial disclosures.
The U.S. Preventive Services Task Force has issued updated recommendations on the use of chemoprevention for breast cancer that emphasize the need for informed, shared decision-making.
"It’s a complicated decision, and there are a lot of moving parts, if you will," USPSTF member Dr. Mark Ebell said in an interview.
New research since the original 2002 guidance, including updates from the pivotal STAR trial (Cancer Prev. Res. 2010;3:696-706) and a recent meta-analysis of risk-reducing medications (Ann. Intern. Med. 2013;158:604-14), have helped clarify the balance of benefits and harms by a woman’s age, breast cancer risk, race, and whether she’s had a hysterectomy.
The 2013 guidelines, published in Annals of Internal Medicine, plug those data into a series of tables that present the net benefit as stratified by those risk factors. For example, Table 1 is for white, non-Hispanic women who have a uterus.
"So, you’d go there, look up [the patient’s] 5-year projected risk, look up [the patient’s] age, and it provides a general color-coded assessment of whether there are more risks or harms," Dr. Ebell said. "It’s a tough decision, and two women with similar risk factors may make different decisions, and that’s perfectly okay. Everyone’s different; everyone assesses risk differently and has different values. We just wanted to make sure they have the best possible information when they make their decision."
Both the original and updated versions carry the same overall recommendation: Women who are at an average or low risk should not use chemoprevention, while women at high risk should consider therapy if they don’t have any contraindications.
The 2013 recommendations are trying, however, to be a bit more explicit about which medications have been approved by the Food and Drug Administration (FDA) for breast cancer chemoprevention, said Dr. Ebell, a family physician with the College of Public Health, University of Georgia, Athens.
The USPSTF recommends the use of the selective estrogen receptor modulators tamoxifen and raloxifene (Evista) to reduce the incidence of invasive breast cancer in asymptomatic women aged 35 years and older who are without a prior diagnosis of breast cancer, ductal carcinoma in situ, or lobular carcinoma in situ and who are at increased risk of breast cancer. The daily doses are 20 mg and 60 mg, respectively, for 5 years.
Tamoxifen is FDA approved for this use in women aged 35 years and older, while raloxifene is approved for this indication in postmenopausal women.
In a draft version of the guidance released this April, the target audience for the recommendation was asymptomatic women aged 40 years and older, but the age limit was lowered to 35 years in the final version because the Breast Cancer Prevention Trial enrolled women over age 35, Dr. Ebell said.
Notably absent in the new guidelines is an endorsement of the aromatase inhibitor exemestane (Aromasin). Updated chemoprevention guidelines issued earlier this year by the American Society of Clinical Oncology recommend that exemestane be discussed as an alternative to reduce the risk of invasive, estrogen receptor–positive breast cancer in postmenopausal women (J. Clin. Onc. 2013;31:2942-62).
The task force only considers FDA-approved medications for the indication of breast cancer chemoprevention, and exemestane is only approved for breast cancer treatment, Dr. Ebell explained.
The aromatase inhibitor anastrozole (Arimidex) is not recommended by either ASCO or the USPSTF, with data awaited from the ongoing phase III, placebo-controlled British IBIS II (Anastrozole in Preventing Breast Cancer in Postmenopausal Women at Increased Risk of Breast Cancer) study.
Dr. Joanna Cain, professor of obstetrics/gynecology at the University of Massachusetts, Worcester, said the task force made the decision regarding exemestane on the basis of the present evidence.
"UPSTF has a level of standards that exemestane does not yet meet, while ASCO is willing to move with less evidence," she said in an interview. "They are not ruling it out but saying by their criteria, it does not yet meet this. Having said all that, if a patient is intolerant of tamoxifen and raloxifene and needs chemopreventive therapy, we would consider using exemestane as an alternative."
Dr. Cain noted that she welcomes the 2013 guidance, particularly the appropriate caution about individual patient risks for these medications such as age, venous thrombosis, and endometrial cancer, which also includes the risk conferred by obesity and genetics such as Lynch syndrome.
"In particular, the clarity around benefit, and therefore, use only for those at increased risk, is helpful," she said.
The use of risk-reducing medications is quite low, but that’s because only about 5% of women are really appropriate candidates and only a minority of these actually take the medication, Dr. Ebell said.
Just 12% of high-risk women opted to take tamoxifen to reduce their risk for breast cancer in a national survey highlighted by the task force, with 77% of women declining primarily because of concerns about serious adverse events and small therapeutic benefit (Arch. Intern. Med. 2006;166:2260-5).
Further, only 27% of the 350 primary care physicians surveyed had prescribed tamoxifen for breast cancer prevention at least once in the prior 12 months.
"We do need to engage the primary care community more broadly, not just ob.gyns., in this informed decision-making and make sure they are comfortable and confident when they have a patient with questions about chemoprevention," Dr. Ebell said.
Dr. Ebell reported having no financial disclosures.
FROM ANNALS OF INTERNAL MEDICINE
USPSTF breast cancer chemoprevention recommendations: We’re in this together
The U.S. Preventive Services Task Force has issued updated recommendations on the use of chemoprevention for breast cancer that emphasize the need for informed, shared decision-making.
"It’s a complicated decision, and there are a lot of moving parts, if you will," USPSTF member Dr. Mark Ebell said in an interview.
New research since the original 2002 guidance, including updates from the pivotal STAR trial (Cancer Prev. Res. 2010;3:696-706) and a recent meta-analysis of risk-reducing medications (Ann. Intern. Med. 2013;158:604-14), have helped clarify the balance of benefits and harms by a woman’s age, breast cancer risk, race, and whether she’s had a hysterectomy.
The 2013 guidelines, published in Annals of Internal Medicine, plug those data into a series of tables that present the net benefit as stratified by those risk factors. For example, Table 1 is for white, non-Hispanic women who have a uterus.
"So, you’d go there, look up [the patient’s] 5-year projected risk, look up [the patient’s] age, and it provides a general color-coded assessment of whether there are more risks or harms," Dr. Ebell said. "It’s a tough decision, and two women with similar risk factors may make different decisions, and that’s perfectly okay. Everyone’s different; everyone assesses risk differently and has different values. We just wanted to make sure they have the best possible information when they make their decision."
Both the original and updated versions carry the same overall recommendation: Women who are at an average or low risk should not use chemoprevention, while women at high risk should consider therapy if they don’t have any contraindications.
The 2013 recommendations are trying, however, to be a bit more explicit about which medications have been approved by the Food and Drug Administration (FDA) for breast cancer chemoprevention, said Dr. Ebell, a family physician with the College of Public Health, University of Georgia, Athens.
The USPSTF recommends the use of the selective estrogen receptor modulators tamoxifen and raloxifene (Evista) to reduce the incidence of invasive breast cancer in asymptomatic women aged 35 years and older who are without a prior diagnosis of breast cancer, ductal carcinoma in situ, or lobular carcinoma in situ and who are at increased risk of breast cancer. The daily doses are 20 mg and 60 mg, respectively, for 5 years.
Tamoxifen is FDA approved for this use in women aged 35 years and older, while raloxifene is approved for this indication in postmenopausal women.
In a draft version of the guidance released this April, the target audience for the recommendation was asymptomatic women aged 40 years and older, but the age limit was lowered to 35 years in the final version because the Breast Cancer Prevention Trial enrolled women over age 35, Dr. Ebell said.
Notably absent in the new guidelines is an endorsement of the aromatase inhibitor exemestane (Aromasin). Updated chemoprevention guidelines issued earlier this year by the American Society of Clinical Oncology recommend that exemestane be discussed as an alternative to reduce the risk of invasive, estrogen receptor–positive breast cancer in postmenopausal women (J. Clin. Onc. 2013;31:2942-62).
The task force only considers FDA-approved medications for the indication of breast cancer chemoprevention, and exemestane is only approved for breast cancer treatment, Dr. Ebell explained.
The aromatase inhibitor anastrozole (Arimidex) is not recommended by either ASCO or the USPSTF, with data awaited from the ongoing phase III, placebo-controlled British IBIS II (Anastrozole in Preventing Breast Cancer in Postmenopausal Women at Increased Risk of Breast Cancer) study.
Dr. Joanna Cain, professor of obstetrics/gynecology at the University of Massachusetts, Worcester, said the task force made the decision regarding exemestane on the basis of the present evidence.
"UPSTF has a level of standards that exemestane does not yet meet, while ASCO is willing to move with less evidence," she said in an interview. "They are not ruling it out but saying by their criteria, it does not yet meet this. Having said all that, if a patient is intolerant of tamoxifen and raloxifene and needs chemopreventive therapy, we would consider using exemestane as an alternative."
Dr. Cain noted that she welcomes the 2013 guidance, particularly the appropriate caution about individual patient risks for these medications such as age, venous thrombosis, and endometrial cancer, which also includes the risk conferred by obesity and genetics such as Lynch syndrome.
"In particular, the clarity around benefit, and therefore, use only for those at increased risk, is helpful," she said.
The use of risk-reducing medications is quite low, but that’s because only about 5% of women are really appropriate candidates and only a minority of these actually take the medication, Dr. Ebell said.
Just 12% of high-risk women opted to take tamoxifen to reduce their risk for breast cancer in a national survey highlighted by the task force, with 77% of women declining primarily because of concerns about serious adverse events and small therapeutic benefit (Arch. Intern. Med. 2006;166:2260-5).
Further, only 27% of the 350 primary care physicians surveyed had prescribed tamoxifen for breast cancer prevention at least once in the prior 12 months.
"We do need to engage the primary care community more broadly, not just ob.gyns., in this informed decision-making and make sure they are comfortable and confident when they have a patient with questions about chemoprevention," Dr. Ebell said.
Dr. Ebell reported having no financial disclosures.
The U.S. Preventive Services Task Force has issued updated recommendations on the use of chemoprevention for breast cancer that emphasize the need for informed, shared decision-making.
"It’s a complicated decision, and there are a lot of moving parts, if you will," USPSTF member Dr. Mark Ebell said in an interview.
New research since the original 2002 guidance, including updates from the pivotal STAR trial (Cancer Prev. Res. 2010;3:696-706) and a recent meta-analysis of risk-reducing medications (Ann. Intern. Med. 2013;158:604-14), have helped clarify the balance of benefits and harms by a woman’s age, breast cancer risk, race, and whether she’s had a hysterectomy.
The 2013 guidelines, published in Annals of Internal Medicine, plug those data into a series of tables that present the net benefit as stratified by those risk factors. For example, Table 1 is for white, non-Hispanic women who have a uterus.
"So, you’d go there, look up [the patient’s] 5-year projected risk, look up [the patient’s] age, and it provides a general color-coded assessment of whether there are more risks or harms," Dr. Ebell said. "It’s a tough decision, and two women with similar risk factors may make different decisions, and that’s perfectly okay. Everyone’s different; everyone assesses risk differently and has different values. We just wanted to make sure they have the best possible information when they make their decision."
Both the original and updated versions carry the same overall recommendation: Women who are at an average or low risk should not use chemoprevention, while women at high risk should consider therapy if they don’t have any contraindications.
The 2013 recommendations are trying, however, to be a bit more explicit about which medications have been approved by the Food and Drug Administration (FDA) for breast cancer chemoprevention, said Dr. Ebell, a family physician with the College of Public Health, University of Georgia, Athens.
The USPSTF recommends the use of the selective estrogen receptor modulators tamoxifen and raloxifene (Evista) to reduce the incidence of invasive breast cancer in asymptomatic women aged 35 years and older who are without a prior diagnosis of breast cancer, ductal carcinoma in situ, or lobular carcinoma in situ and who are at increased risk of breast cancer. The daily doses are 20 mg and 60 mg, respectively, for 5 years.
Tamoxifen is FDA approved for this use in women aged 35 years and older, while raloxifene is approved for this indication in postmenopausal women.
In a draft version of the guidance released this April, the target audience for the recommendation was asymptomatic women aged 40 years and older, but the age limit was lowered to 35 years in the final version because the Breast Cancer Prevention Trial enrolled women over age 35, Dr. Ebell said.
Notably absent in the new guidelines is an endorsement of the aromatase inhibitor exemestane (Aromasin). Updated chemoprevention guidelines issued earlier this year by the American Society of Clinical Oncology recommend that exemestane be discussed as an alternative to reduce the risk of invasive, estrogen receptor–positive breast cancer in postmenopausal women (J. Clin. Onc. 2013;31:2942-62).
The task force only considers FDA-approved medications for the indication of breast cancer chemoprevention, and exemestane is only approved for breast cancer treatment, Dr. Ebell explained.
The aromatase inhibitor anastrozole (Arimidex) is not recommended by either ASCO or the USPSTF, with data awaited from the ongoing phase III, placebo-controlled British IBIS II (Anastrozole in Preventing Breast Cancer in Postmenopausal Women at Increased Risk of Breast Cancer) study.
Dr. Joanna Cain, professor of obstetrics/gynecology at the University of Massachusetts, Worcester, said the task force made the decision regarding exemestane on the basis of the present evidence.
"UPSTF has a level of standards that exemestane does not yet meet, while ASCO is willing to move with less evidence," she said in an interview. "They are not ruling it out but saying by their criteria, it does not yet meet this. Having said all that, if a patient is intolerant of tamoxifen and raloxifene and needs chemopreventive therapy, we would consider using exemestane as an alternative."
Dr. Cain noted that she welcomes the 2013 guidance, particularly the appropriate caution about individual patient risks for these medications such as age, venous thrombosis, and endometrial cancer, which also includes the risk conferred by obesity and genetics such as Lynch syndrome.
"In particular, the clarity around benefit, and therefore, use only for those at increased risk, is helpful," she said.
The use of risk-reducing medications is quite low, but that’s because only about 5% of women are really appropriate candidates and only a minority of these actually take the medication, Dr. Ebell said.
Just 12% of high-risk women opted to take tamoxifen to reduce their risk for breast cancer in a national survey highlighted by the task force, with 77% of women declining primarily because of concerns about serious adverse events and small therapeutic benefit (Arch. Intern. Med. 2006;166:2260-5).
Further, only 27% of the 350 primary care physicians surveyed had prescribed tamoxifen for breast cancer prevention at least once in the prior 12 months.
"We do need to engage the primary care community more broadly, not just ob.gyns., in this informed decision-making and make sure they are comfortable and confident when they have a patient with questions about chemoprevention," Dr. Ebell said.
Dr. Ebell reported having no financial disclosures.
The U.S. Preventive Services Task Force has issued updated recommendations on the use of chemoprevention for breast cancer that emphasize the need for informed, shared decision-making.
"It’s a complicated decision, and there are a lot of moving parts, if you will," USPSTF member Dr. Mark Ebell said in an interview.
New research since the original 2002 guidance, including updates from the pivotal STAR trial (Cancer Prev. Res. 2010;3:696-706) and a recent meta-analysis of risk-reducing medications (Ann. Intern. Med. 2013;158:604-14), have helped clarify the balance of benefits and harms by a woman’s age, breast cancer risk, race, and whether she’s had a hysterectomy.
The 2013 guidelines, published in Annals of Internal Medicine, plug those data into a series of tables that present the net benefit as stratified by those risk factors. For example, Table 1 is for white, non-Hispanic women who have a uterus.
"So, you’d go there, look up [the patient’s] 5-year projected risk, look up [the patient’s] age, and it provides a general color-coded assessment of whether there are more risks or harms," Dr. Ebell said. "It’s a tough decision, and two women with similar risk factors may make different decisions, and that’s perfectly okay. Everyone’s different; everyone assesses risk differently and has different values. We just wanted to make sure they have the best possible information when they make their decision."
Both the original and updated versions carry the same overall recommendation: Women who are at an average or low risk should not use chemoprevention, while women at high risk should consider therapy if they don’t have any contraindications.
The 2013 recommendations are trying, however, to be a bit more explicit about which medications have been approved by the Food and Drug Administration (FDA) for breast cancer chemoprevention, said Dr. Ebell, a family physician with the College of Public Health, University of Georgia, Athens.
The USPSTF recommends the use of the selective estrogen receptor modulators tamoxifen and raloxifene (Evista) to reduce the incidence of invasive breast cancer in asymptomatic women aged 35 years and older who are without a prior diagnosis of breast cancer, ductal carcinoma in situ, or lobular carcinoma in situ and who are at increased risk of breast cancer. The daily doses are 20 mg and 60 mg, respectively, for 5 years.
Tamoxifen is FDA approved for this use in women aged 35 years and older, while raloxifene is approved for this indication in postmenopausal women.
In a draft version of the guidance released this April, the target audience for the recommendation was asymptomatic women aged 40 years and older, but the age limit was lowered to 35 years in the final version because the Breast Cancer Prevention Trial enrolled women over age 35, Dr. Ebell said.
Notably absent in the new guidelines is an endorsement of the aromatase inhibitor exemestane (Aromasin). Updated chemoprevention guidelines issued earlier this year by the American Society of Clinical Oncology recommend that exemestane be discussed as an alternative to reduce the risk of invasive, estrogen receptor–positive breast cancer in postmenopausal women (J. Clin. Onc. 2013;31:2942-62).
The task force only considers FDA-approved medications for the indication of breast cancer chemoprevention, and exemestane is only approved for breast cancer treatment, Dr. Ebell explained.
The aromatase inhibitor anastrozole (Arimidex) is not recommended by either ASCO or the USPSTF, with data awaited from the ongoing phase III, placebo-controlled British IBIS II (Anastrozole in Preventing Breast Cancer in Postmenopausal Women at Increased Risk of Breast Cancer) study.
Dr. Joanna Cain, professor of obstetrics/gynecology at the University of Massachusetts, Worcester, said the task force made the decision regarding exemestane on the basis of the present evidence.
"UPSTF has a level of standards that exemestane does not yet meet, while ASCO is willing to move with less evidence," she said in an interview. "They are not ruling it out but saying by their criteria, it does not yet meet this. Having said all that, if a patient is intolerant of tamoxifen and raloxifene and needs chemopreventive therapy, we would consider using exemestane as an alternative."
Dr. Cain noted that she welcomes the 2013 guidance, particularly the appropriate caution about individual patient risks for these medications such as age, venous thrombosis, and endometrial cancer, which also includes the risk conferred by obesity and genetics such as Lynch syndrome.
"In particular, the clarity around benefit, and therefore, use only for those at increased risk, is helpful," she said.
The use of risk-reducing medications is quite low, but that’s because only about 5% of women are really appropriate candidates and only a minority of these actually take the medication, Dr. Ebell said.
Just 12% of high-risk women opted to take tamoxifen to reduce their risk for breast cancer in a national survey highlighted by the task force, with 77% of women declining primarily because of concerns about serious adverse events and small therapeutic benefit (Arch. Intern. Med. 2006;166:2260-5).
Further, only 27% of the 350 primary care physicians surveyed had prescribed tamoxifen for breast cancer prevention at least once in the prior 12 months.
"We do need to engage the primary care community more broadly, not just ob.gyns., in this informed decision-making and make sure they are comfortable and confident when they have a patient with questions about chemoprevention," Dr. Ebell said.
Dr. Ebell reported having no financial disclosures.
FROM ANNALS OF INTERNAL MEDICINE
Major finding: Asymptomatic women without a prior diagnosis of breast cancer, ductal carcinoma in situ, or lobular carcinoma in situ who are at increased risk of breast cancer should consider taking tamoxifen 20 mg daily and raloxifene 60 mg daily for 5 years to reduce the risk of breast cancer.
Data source: USPSTF chemoprevention recommendations for women.
Disclosures: Dr. Ebell reported having no financial disclosures.
Pediatric osteomyelitis fraught with clinical challenges
Molecular and imaging tests have increased the identification of pediatric osteomyelitis, but there are still plenty of challenges and conundrums in the management of these cases.
Part of the problem is that not all children fit the diagnosis of acute uncomplicated osteomyelitis, and fall into a "data-free zone," suggests Dr. Russell McCulloh, an infectious disease specialist with Children’s Mercy Hospital in Kansas City, Mo. Acute hematogenous osteomyelitis (AHOM) accounts for 1% of pediatric hospitalizations, although most studies don’t address complicated osteomyelitis, culture-negative cases, neonates, surgical interventions, cases with hardware in place, and colonization outside of the long bone, the most usual site for osteomyelitis.
"It’s easy, I think, to look at the abstracts and inadvertently extrapolate the data to patients we just don’t have the data on," he said during a pediatric osteomyelitis roundtable at Pediatric Hospital Medicine 2013 in New Orleans.
In the absence of a consensus on how to manage complicated or culture-negative patients, a surprising number of groups are turning to medium-course therapy. Data have shown that if a peripherally inserted central catheter (PICC) line is in place for 2 weeks or less, there are far fewer complications than if it’s in place for 4-6 weeks. This has led some groups to take the approach of giving intravenous antibiotics for 2 weeks and then transitioning to oral antibiotics if the patient is doing well, observed Dr. Samir S. Shah, director of hospital medicine at Cincinnati Children’s Hospital Medical Center.
"Again, there’s no great data on whether that’s a good strategy or not, but some folks feel that it mitigates some of the challenges with doing a more prolonged course of IV therapy, with their nervousness of transitioning to oral therapy in the process," he said.
The data are fairly clear, however, that oral antibiotics are effective in treating acute, uncomplicated osteomyelitis, and that early transition to oral antibiotics demonstrates similar success to that of long-term IV therapy, said copresenter Dr. Joanna Thomson, a fellow in hospital medicine at Cincinnati Children’s.
One of the best analyses on this issue is a systematic review of 12 studies showing similar 6-month cure rates among children, aged 3 months to 16 years, with AHOM given IV antibiotics for less than 7 days or for 1 week or longer (95.2% vs. 98.8%) (BMC Infect. Dis. 2002;2:16).
Six-month treatment failure rates also were similar between prolonged IV antibiotics and early transition to oral therapy in one of the largest studies of pediatric osteomyelitis, a retrospective cohort study of about 2,000 children carefully screened to exclude all but the most straightforward cases (Pediatrics 2009;123:636-42), she said.
An audience member questioned whether orthopedic surgical involvement tips the diagnostic scales from simple to complicated osteomyelitis, to which Dr. Shah replied that complicated cases typically involve additional problems such as thrombosis, pulmonary septic emboli, metastatic dissemination of infection, or prolonged bacteremia. Another attendee also suggested that cancer should be on clinicians’ radar in patients failing to respond, noting that she had two recent cases including a 5-year-old with neuroblastoma and extensive metastases.
The Pediatric Infectious Diseases Society and the Infectious Diseases Society of America (IDSA) have partnered to create new pediatric guidelines for the diagnosis and management of AHOM. At present, they are considering a possible recommendation that, if approved, would call for early transition to oral antibiotics, said Dr. Shah, a member of the guideline committee.
Clindamycin is a good choice for empiric coverage of Staphylococcus aureus for acute osteomyelitis in stable patients, if the local susceptibility patterns allow, said copresenter Dr. Amanda C. Schondelmeyer, also a hospital medicine fellow at Cincinnati Children’s. Vancomycin should be considered for empiric coverage of S. aureus in patients with bacteremia or abnormal vital signs, while beta-lactam antibiotics can be added for coverage of Kingella kingae in patients under age 3 with septic arthritis.
Current IDSA guidelines recommend IV vancomycin for children with acute hematogenous methicillin-resistant S. aureus (MRSA) osteomyelitis or septic arthritis. If the patient is stable without ongoing bacteremia or intravascular infection, clindamycin can be used as empiric therapy, if the clindamycin resistance rate is low, for example, less than 10%.
A quick show of hands revealed that while most attendees knew the clindamycin susceptibility pattern for MRSA at their institution, only a handful knew if it exceeded 10%.
An informal survey of the overflow crowd also indicated no clear consensus on what factors are most important in transitioning patients to oral antibiotics. A normalized C-reactive protein (CRP), afebrile state, and at least 48 hours of IV antibiotics were suggested, but some attendees commented that it was rare for CRP values to normalize within 2-3 days of initiating therapy and expressed uncertainty about the optimal CRP threshold to use.
A recent single-center, retrospective study reported using a CRP lower than 2.0-3.0 mg/L and evidence of clinical response for oral step-down therapy among 194 children with culture-positive, acute bacterial osteomyelitis or acute bacterial arthritis. This combination strategy, in place at Rady Children’s Hospital in San Diego for more than a decade, yielded only one microbiologic failure and a long-term success rate with early transition to oral antibiotics that was similar to outcomes for long-term IV courses (Pediatrics 2012;130:e821-8).
A smaller prospective study in 44 children with AHOM reported that CRP values peaked 2 days after admission and normalized within a week of initiating therapy and transitioning to oral antibiotics within 4 days (Pediatrics 1994;93:59-62).
Regardless of the antibiotic plan, follow-up after discharge is key, said copresenter Dr. Lauren G. Solan, a hospital medicine fellow at Cincinnati Children’s. Hospitalists should ensure that the primary care physician is comfortable with the discharge plan and consider follow-up with an infectious disease specialist, particularly in children sent home on IV therapy. Catheter-associated complications requiring attention occur in about 30% of children with PICC lines at home, with malfunction or displacement reported in nearly one-fourth of those receiving more than 2 weeks of IV therapy, she observed. Notably, just five audience members acknowledged requesting an orthopedic follow-up, even in cases involving surgical procedures.
The presenters reported having no financial disclosures.
Molecular and imaging tests have increased the identification of pediatric osteomyelitis, but there are still plenty of challenges and conundrums in the management of these cases.
Part of the problem is that not all children fit the diagnosis of acute uncomplicated osteomyelitis, and fall into a "data-free zone," suggests Dr. Russell McCulloh, an infectious disease specialist with Children’s Mercy Hospital in Kansas City, Mo. Acute hematogenous osteomyelitis (AHOM) accounts for 1% of pediatric hospitalizations, although most studies don’t address complicated osteomyelitis, culture-negative cases, neonates, surgical interventions, cases with hardware in place, and colonization outside of the long bone, the most usual site for osteomyelitis.
"It’s easy, I think, to look at the abstracts and inadvertently extrapolate the data to patients we just don’t have the data on," he said during a pediatric osteomyelitis roundtable at Pediatric Hospital Medicine 2013 in New Orleans.
In the absence of a consensus on how to manage complicated or culture-negative patients, a surprising number of groups are turning to medium-course therapy. Data have shown that if a peripherally inserted central catheter (PICC) line is in place for 2 weeks or less, there are far fewer complications than if it’s in place for 4-6 weeks. This has led some groups to take the approach of giving intravenous antibiotics for 2 weeks and then transitioning to oral antibiotics if the patient is doing well, observed Dr. Samir S. Shah, director of hospital medicine at Cincinnati Children’s Hospital Medical Center.
"Again, there’s no great data on whether that’s a good strategy or not, but some folks feel that it mitigates some of the challenges with doing a more prolonged course of IV therapy, with their nervousness of transitioning to oral therapy in the process," he said.
The data are fairly clear, however, that oral antibiotics are effective in treating acute, uncomplicated osteomyelitis, and that early transition to oral antibiotics demonstrates similar success to that of long-term IV therapy, said copresenter Dr. Joanna Thomson, a fellow in hospital medicine at Cincinnati Children’s.
One of the best analyses on this issue is a systematic review of 12 studies showing similar 6-month cure rates among children, aged 3 months to 16 years, with AHOM given IV antibiotics for less than 7 days or for 1 week or longer (95.2% vs. 98.8%) (BMC Infect. Dis. 2002;2:16).
Six-month treatment failure rates also were similar between prolonged IV antibiotics and early transition to oral therapy in one of the largest studies of pediatric osteomyelitis, a retrospective cohort study of about 2,000 children carefully screened to exclude all but the most straightforward cases (Pediatrics 2009;123:636-42), she said.
An audience member questioned whether orthopedic surgical involvement tips the diagnostic scales from simple to complicated osteomyelitis, to which Dr. Shah replied that complicated cases typically involve additional problems such as thrombosis, pulmonary septic emboli, metastatic dissemination of infection, or prolonged bacteremia. Another attendee also suggested that cancer should be on clinicians’ radar in patients failing to respond, noting that she had two recent cases including a 5-year-old with neuroblastoma and extensive metastases.
The Pediatric Infectious Diseases Society and the Infectious Diseases Society of America (IDSA) have partnered to create new pediatric guidelines for the diagnosis and management of AHOM. At present, they are considering a possible recommendation that, if approved, would call for early transition to oral antibiotics, said Dr. Shah, a member of the guideline committee.
Clindamycin is a good choice for empiric coverage of Staphylococcus aureus for acute osteomyelitis in stable patients, if the local susceptibility patterns allow, said copresenter Dr. Amanda C. Schondelmeyer, also a hospital medicine fellow at Cincinnati Children’s. Vancomycin should be considered for empiric coverage of S. aureus in patients with bacteremia or abnormal vital signs, while beta-lactam antibiotics can be added for coverage of Kingella kingae in patients under age 3 with septic arthritis.
Current IDSA guidelines recommend IV vancomycin for children with acute hematogenous methicillin-resistant S. aureus (MRSA) osteomyelitis or septic arthritis. If the patient is stable without ongoing bacteremia or intravascular infection, clindamycin can be used as empiric therapy, if the clindamycin resistance rate is low, for example, less than 10%.
A quick show of hands revealed that while most attendees knew the clindamycin susceptibility pattern for MRSA at their institution, only a handful knew if it exceeded 10%.
An informal survey of the overflow crowd also indicated no clear consensus on what factors are most important in transitioning patients to oral antibiotics. A normalized C-reactive protein (CRP), afebrile state, and at least 48 hours of IV antibiotics were suggested, but some attendees commented that it was rare for CRP values to normalize within 2-3 days of initiating therapy and expressed uncertainty about the optimal CRP threshold to use.
A recent single-center, retrospective study reported using a CRP lower than 2.0-3.0 mg/L and evidence of clinical response for oral step-down therapy among 194 children with culture-positive, acute bacterial osteomyelitis or acute bacterial arthritis. This combination strategy, in place at Rady Children’s Hospital in San Diego for more than a decade, yielded only one microbiologic failure and a long-term success rate with early transition to oral antibiotics that was similar to outcomes for long-term IV courses (Pediatrics 2012;130:e821-8).
A smaller prospective study in 44 children with AHOM reported that CRP values peaked 2 days after admission and normalized within a week of initiating therapy and transitioning to oral antibiotics within 4 days (Pediatrics 1994;93:59-62).
Regardless of the antibiotic plan, follow-up after discharge is key, said copresenter Dr. Lauren G. Solan, a hospital medicine fellow at Cincinnati Children’s. Hospitalists should ensure that the primary care physician is comfortable with the discharge plan and consider follow-up with an infectious disease specialist, particularly in children sent home on IV therapy. Catheter-associated complications requiring attention occur in about 30% of children with PICC lines at home, with malfunction or displacement reported in nearly one-fourth of those receiving more than 2 weeks of IV therapy, she observed. Notably, just five audience members acknowledged requesting an orthopedic follow-up, even in cases involving surgical procedures.
The presenters reported having no financial disclosures.
Molecular and imaging tests have increased the identification of pediatric osteomyelitis, but there are still plenty of challenges and conundrums in the management of these cases.
Part of the problem is that not all children fit the diagnosis of acute uncomplicated osteomyelitis, and fall into a "data-free zone," suggests Dr. Russell McCulloh, an infectious disease specialist with Children’s Mercy Hospital in Kansas City, Mo. Acute hematogenous osteomyelitis (AHOM) accounts for 1% of pediatric hospitalizations, although most studies don’t address complicated osteomyelitis, culture-negative cases, neonates, surgical interventions, cases with hardware in place, and colonization outside of the long bone, the most usual site for osteomyelitis.
"It’s easy, I think, to look at the abstracts and inadvertently extrapolate the data to patients we just don’t have the data on," he said during a pediatric osteomyelitis roundtable at Pediatric Hospital Medicine 2013 in New Orleans.
In the absence of a consensus on how to manage complicated or culture-negative patients, a surprising number of groups are turning to medium-course therapy. Data have shown that if a peripherally inserted central catheter (PICC) line is in place for 2 weeks or less, there are far fewer complications than if it’s in place for 4-6 weeks. This has led some groups to take the approach of giving intravenous antibiotics for 2 weeks and then transitioning to oral antibiotics if the patient is doing well, observed Dr. Samir S. Shah, director of hospital medicine at Cincinnati Children’s Hospital Medical Center.
"Again, there’s no great data on whether that’s a good strategy or not, but some folks feel that it mitigates some of the challenges with doing a more prolonged course of IV therapy, with their nervousness of transitioning to oral therapy in the process," he said.
The data are fairly clear, however, that oral antibiotics are effective in treating acute, uncomplicated osteomyelitis, and that early transition to oral antibiotics demonstrates similar success to that of long-term IV therapy, said copresenter Dr. Joanna Thomson, a fellow in hospital medicine at Cincinnati Children’s.
One of the best analyses on this issue is a systematic review of 12 studies showing similar 6-month cure rates among children, aged 3 months to 16 years, with AHOM given IV antibiotics for less than 7 days or for 1 week or longer (95.2% vs. 98.8%) (BMC Infect. Dis. 2002;2:16).
Six-month treatment failure rates also were similar between prolonged IV antibiotics and early transition to oral therapy in one of the largest studies of pediatric osteomyelitis, a retrospective cohort study of about 2,000 children carefully screened to exclude all but the most straightforward cases (Pediatrics 2009;123:636-42), she said.
An audience member questioned whether orthopedic surgical involvement tips the diagnostic scales from simple to complicated osteomyelitis, to which Dr. Shah replied that complicated cases typically involve additional problems such as thrombosis, pulmonary septic emboli, metastatic dissemination of infection, or prolonged bacteremia. Another attendee also suggested that cancer should be on clinicians’ radar in patients failing to respond, noting that she had two recent cases including a 5-year-old with neuroblastoma and extensive metastases.
The Pediatric Infectious Diseases Society and the Infectious Diseases Society of America (IDSA) have partnered to create new pediatric guidelines for the diagnosis and management of AHOM. At present, they are considering a possible recommendation that, if approved, would call for early transition to oral antibiotics, said Dr. Shah, a member of the guideline committee.
Clindamycin is a good choice for empiric coverage of Staphylococcus aureus for acute osteomyelitis in stable patients, if the local susceptibility patterns allow, said copresenter Dr. Amanda C. Schondelmeyer, also a hospital medicine fellow at Cincinnati Children’s. Vancomycin should be considered for empiric coverage of S. aureus in patients with bacteremia or abnormal vital signs, while beta-lactam antibiotics can be added for coverage of Kingella kingae in patients under age 3 with septic arthritis.
Current IDSA guidelines recommend IV vancomycin for children with acute hematogenous methicillin-resistant S. aureus (MRSA) osteomyelitis or septic arthritis. If the patient is stable without ongoing bacteremia or intravascular infection, clindamycin can be used as empiric therapy, if the clindamycin resistance rate is low, for example, less than 10%.
A quick show of hands revealed that while most attendees knew the clindamycin susceptibility pattern for MRSA at their institution, only a handful knew if it exceeded 10%.
An informal survey of the overflow crowd also indicated no clear consensus on what factors are most important in transitioning patients to oral antibiotics. A normalized C-reactive protein (CRP), afebrile state, and at least 48 hours of IV antibiotics were suggested, but some attendees commented that it was rare for CRP values to normalize within 2-3 days of initiating therapy and expressed uncertainty about the optimal CRP threshold to use.
A recent single-center, retrospective study reported using a CRP lower than 2.0-3.0 mg/L and evidence of clinical response for oral step-down therapy among 194 children with culture-positive, acute bacterial osteomyelitis or acute bacterial arthritis. This combination strategy, in place at Rady Children’s Hospital in San Diego for more than a decade, yielded only one microbiologic failure and a long-term success rate with early transition to oral antibiotics that was similar to outcomes for long-term IV courses (Pediatrics 2012;130:e821-8).
A smaller prospective study in 44 children with AHOM reported that CRP values peaked 2 days after admission and normalized within a week of initiating therapy and transitioning to oral antibiotics within 4 days (Pediatrics 1994;93:59-62).
Regardless of the antibiotic plan, follow-up after discharge is key, said copresenter Dr. Lauren G. Solan, a hospital medicine fellow at Cincinnati Children’s. Hospitalists should ensure that the primary care physician is comfortable with the discharge plan and consider follow-up with an infectious disease specialist, particularly in children sent home on IV therapy. Catheter-associated complications requiring attention occur in about 30% of children with PICC lines at home, with malfunction or displacement reported in nearly one-fourth of those receiving more than 2 weeks of IV therapy, she observed. Notably, just five audience members acknowledged requesting an orthopedic follow-up, even in cases involving surgical procedures.
The presenters reported having no financial disclosures.
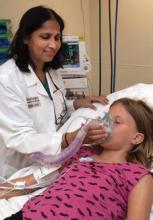
Hypertonic saline a washout in bronchiolitis
NEW ORLEANS – Hypertonic saline alone does not affect length of stay among infants hospitalized for bronchiolitis, according to preliminary results of an ongoing randomized, prospective U.S. trial.
Among 127 evaluable patients, the median length of stay was 2.2 days with hypertonic saline and 2.0 days with normal saline (P = .65).
Infants in one still-blinded group were significantly younger (3.3 vs. 4.4 months), but hospital stay did not differ between groups, even when adjusted for age.
"Preliminary results suggest little utility for the routine use of hypertonic saline in treating infants hospitalized with bronchiolitis," Dr. Alyssa Silver said at Pediatric Hospital Medicine 2013.
However, she added, the results do show that 3% hypertonic saline without adjunctive bronchodilators is safe for infants, including those with a history of prior wheeze, even in a population with a high endemic prevalence of asthma.
A theoretical risk of bronchospasm has been identified in patients with cystic fibrosis at much higher concentrations and quantities.
This is the first prospective, randomized, double-blind, controlled clinical trial of nebulized hypertonic saline conducted in the United States.
Several studies have been conducted outside the United States, but adjuvant bronchodilators were used in all but one, patients with a history of wheeze were excluded, and they had long lengths of stay that make the data difficult to generalize to the United States, said Dr. Silver, a pediatric hospitalist with the Children’s Hospital at Montefiore in New York.
Dr. Shawn Ralston, cochair of the American Academy of Pediatrics subcommittee on diagnosis and management of bronchiolitis, which is revising the 2006 AAP clinical practice guidelines for bronchiolitis,said in an interview that a clearer picture of the utility of hypertonic saline is beginning to emerge. All of the studies where hypertonic saline is used in the ED or short-stay units show no utility, and the only inpatient studies that show benefit seem to be in settings where there is longer length of stay. Further, Montefiore has a very short length of stay, which is typical of U.S. hospitals.
"I think what we’re learning about hypertonic saline as the studies come in is that any treatment effect, if there does prove to be one, will be seen over several days," said Dr. Ralston, chief of the pediatrics section, Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
"From a guidelines standpoint, it is very important for us to keep up with the emerging data, and we are unlikely to be able to recommend the use of hypertonic saline given what has been published or presented within the last year. That said, I would actually welcome studies of outpatient use of hypertonic saline, as I think that is where the real utility may lie, based on my review of the literature in preparation for the guideline."
The current study included 156 infants, less than 12 months of age, admitted with a diagnosis of bronchiolitis from November 2011 through January 2013 and randomized by the pharmacy to 4 mL of hypertonic saline or 4 mL of normal saline every 4 hours until discharge.
The readmission rate was identical at 6.3% in both study groups (P = 1.0), Dr. Silver reported. Also similar were clinically important adverse events, including readmission (four events in each group) and worsening respiratory status (six events in each group; both P = 1.0).
A subgroup analysis showed no difference in length of stay among patients treated with hypertonic saline vs. normal saline who were positive for respiratory syncytial virus (2.5 vs. 2.0 days; P = .35), had a history of previous wheeze (1.8 vs. 2.0 days; P = .58), or had a history of prematurity (3.5 vs. 2.5 days; P = .11).
The study was limited by the single-center design and the inability to randomize all patients within 12 hours of admission. Despite the lack of funding, however, the study captured 80% of bronchiolitis admissions among infants during the study period, Dr. Silver noted.
"These findings cast significant doubt on the utility of nebulized 3% hypertonic saline for the routine treatment of infants hospitalized with bronchiolitis," she said in an interview. "Unfortunately, the mainstay of routine inpatient treatment remains supportive care."
Future directions include enrolling patients directly from the emergency department with a minimum severity score for study entry, use of higher concentrations of sodium chloride, and possibly an outpatient study.
Dr. Silver reported having no relevant financial disclosures.
NEW ORLEANS – Hypertonic saline alone does not affect length of stay among infants hospitalized for bronchiolitis, according to preliminary results of an ongoing randomized, prospective U.S. trial.
Among 127 evaluable patients, the median length of stay was 2.2 days with hypertonic saline and 2.0 days with normal saline (P = .65).
Infants in one still-blinded group were significantly younger (3.3 vs. 4.4 months), but hospital stay did not differ between groups, even when adjusted for age.
"Preliminary results suggest little utility for the routine use of hypertonic saline in treating infants hospitalized with bronchiolitis," Dr. Alyssa Silver said at Pediatric Hospital Medicine 2013.
However, she added, the results do show that 3% hypertonic saline without adjunctive bronchodilators is safe for infants, including those with a history of prior wheeze, even in a population with a high endemic prevalence of asthma.
A theoretical risk of bronchospasm has been identified in patients with cystic fibrosis at much higher concentrations and quantities.
This is the first prospective, randomized, double-blind, controlled clinical trial of nebulized hypertonic saline conducted in the United States.
Several studies have been conducted outside the United States, but adjuvant bronchodilators were used in all but one, patients with a history of wheeze were excluded, and they had long lengths of stay that make the data difficult to generalize to the United States, said Dr. Silver, a pediatric hospitalist with the Children’s Hospital at Montefiore in New York.
Dr. Shawn Ralston, cochair of the American Academy of Pediatrics subcommittee on diagnosis and management of bronchiolitis, which is revising the 2006 AAP clinical practice guidelines for bronchiolitis,said in an interview that a clearer picture of the utility of hypertonic saline is beginning to emerge. All of the studies where hypertonic saline is used in the ED or short-stay units show no utility, and the only inpatient studies that show benefit seem to be in settings where there is longer length of stay. Further, Montefiore has a very short length of stay, which is typical of U.S. hospitals.
"I think what we’re learning about hypertonic saline as the studies come in is that any treatment effect, if there does prove to be one, will be seen over several days," said Dr. Ralston, chief of the pediatrics section, Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
"From a guidelines standpoint, it is very important for us to keep up with the emerging data, and we are unlikely to be able to recommend the use of hypertonic saline given what has been published or presented within the last year. That said, I would actually welcome studies of outpatient use of hypertonic saline, as I think that is where the real utility may lie, based on my review of the literature in preparation for the guideline."
The current study included 156 infants, less than 12 months of age, admitted with a diagnosis of bronchiolitis from November 2011 through January 2013 and randomized by the pharmacy to 4 mL of hypertonic saline or 4 mL of normal saline every 4 hours until discharge.
The readmission rate was identical at 6.3% in both study groups (P = 1.0), Dr. Silver reported. Also similar were clinically important adverse events, including readmission (four events in each group) and worsening respiratory status (six events in each group; both P = 1.0).
A subgroup analysis showed no difference in length of stay among patients treated with hypertonic saline vs. normal saline who were positive for respiratory syncytial virus (2.5 vs. 2.0 days; P = .35), had a history of previous wheeze (1.8 vs. 2.0 days; P = .58), or had a history of prematurity (3.5 vs. 2.5 days; P = .11).
The study was limited by the single-center design and the inability to randomize all patients within 12 hours of admission. Despite the lack of funding, however, the study captured 80% of bronchiolitis admissions among infants during the study period, Dr. Silver noted.
"These findings cast significant doubt on the utility of nebulized 3% hypertonic saline for the routine treatment of infants hospitalized with bronchiolitis," she said in an interview. "Unfortunately, the mainstay of routine inpatient treatment remains supportive care."
Future directions include enrolling patients directly from the emergency department with a minimum severity score for study entry, use of higher concentrations of sodium chloride, and possibly an outpatient study.
Dr. Silver reported having no relevant financial disclosures.
NEW ORLEANS – Hypertonic saline alone does not affect length of stay among infants hospitalized for bronchiolitis, according to preliminary results of an ongoing randomized, prospective U.S. trial.
Among 127 evaluable patients, the median length of stay was 2.2 days with hypertonic saline and 2.0 days with normal saline (P = .65).
Infants in one still-blinded group were significantly younger (3.3 vs. 4.4 months), but hospital stay did not differ between groups, even when adjusted for age.
"Preliminary results suggest little utility for the routine use of hypertonic saline in treating infants hospitalized with bronchiolitis," Dr. Alyssa Silver said at Pediatric Hospital Medicine 2013.
However, she added, the results do show that 3% hypertonic saline without adjunctive bronchodilators is safe for infants, including those with a history of prior wheeze, even in a population with a high endemic prevalence of asthma.
A theoretical risk of bronchospasm has been identified in patients with cystic fibrosis at much higher concentrations and quantities.
This is the first prospective, randomized, double-blind, controlled clinical trial of nebulized hypertonic saline conducted in the United States.
Several studies have been conducted outside the United States, but adjuvant bronchodilators were used in all but one, patients with a history of wheeze were excluded, and they had long lengths of stay that make the data difficult to generalize to the United States, said Dr. Silver, a pediatric hospitalist with the Children’s Hospital at Montefiore in New York.
Dr. Shawn Ralston, cochair of the American Academy of Pediatrics subcommittee on diagnosis and management of bronchiolitis, which is revising the 2006 AAP clinical practice guidelines for bronchiolitis,said in an interview that a clearer picture of the utility of hypertonic saline is beginning to emerge. All of the studies where hypertonic saline is used in the ED or short-stay units show no utility, and the only inpatient studies that show benefit seem to be in settings where there is longer length of stay. Further, Montefiore has a very short length of stay, which is typical of U.S. hospitals.
"I think what we’re learning about hypertonic saline as the studies come in is that any treatment effect, if there does prove to be one, will be seen over several days," said Dr. Ralston, chief of the pediatrics section, Dartmouth-Hitchcock Medical Center, Lebanon, N.H.
"From a guidelines standpoint, it is very important for us to keep up with the emerging data, and we are unlikely to be able to recommend the use of hypertonic saline given what has been published or presented within the last year. That said, I would actually welcome studies of outpatient use of hypertonic saline, as I think that is where the real utility may lie, based on my review of the literature in preparation for the guideline."
The current study included 156 infants, less than 12 months of age, admitted with a diagnosis of bronchiolitis from November 2011 through January 2013 and randomized by the pharmacy to 4 mL of hypertonic saline or 4 mL of normal saline every 4 hours until discharge.
The readmission rate was identical at 6.3% in both study groups (P = 1.0), Dr. Silver reported. Also similar were clinically important adverse events, including readmission (four events in each group) and worsening respiratory status (six events in each group; both P = 1.0).
A subgroup analysis showed no difference in length of stay among patients treated with hypertonic saline vs. normal saline who were positive for respiratory syncytial virus (2.5 vs. 2.0 days; P = .35), had a history of previous wheeze (1.8 vs. 2.0 days; P = .58), or had a history of prematurity (3.5 vs. 2.5 days; P = .11).
The study was limited by the single-center design and the inability to randomize all patients within 12 hours of admission. Despite the lack of funding, however, the study captured 80% of bronchiolitis admissions among infants during the study period, Dr. Silver noted.
"These findings cast significant doubt on the utility of nebulized 3% hypertonic saline for the routine treatment of infants hospitalized with bronchiolitis," she said in an interview. "Unfortunately, the mainstay of routine inpatient treatment remains supportive care."
Future directions include enrolling patients directly from the emergency department with a minimum severity score for study entry, use of higher concentrations of sodium chloride, and possibly an outpatient study.
Dr. Silver reported having no relevant financial disclosures.
AT PEDIATRIC HOSPITAL MEDICINE 2013
Major finding: Median length of stay was 2.2 days with hypertonic saline and 2.0 days with normal saline (P = .65).
Data source: A prospective, double-blind randomized controlled trial in 127 infants with bronchiolitis.
Disclosures: Dr. Silver reported having no relevant financial disclosures.
Pearls of wisdom for pediatric nitrous oxide sedation
Washington University School of Medicine in St. Louis has led the charge for pediatric hospitalist sedation, offering a panel of experts who shared their insights on nitrous oxide sedation in a wide-ranging, hands-on session at Pediatric Hospital Medicine 2013. Some highlights of that session were:
• Administer 50%-70% nitrous oxide (N2O) based on the need for minimal, moderate, or deep sedation. Consider N2O alone for procedures such as joint injections or voiding cystourethrograms.
• Augment N2O with 0.15-0.3 mg/kg of oxycodone (maximum dose of 10 mg) to achieve deeper sedation and greater analgesia for more painful procedures such as abscess incision and drainage, burn debridement, and postoperative dressing change. To maximize the analgesic effect of the opioid, wait for its effect to peak at 60 minutes before administering N2O.
• The peak effect of N2O is usually seen within 2-3 minutes, so plan to perform the most painful portion of the procedure at least 2 minutes after the start of 70% N2O. Titrate to 50% after about 5 minutes of 70% N2O, with a total duration of N2O not to exceed 15 minutes.
• Be alert for signs of emesis during sedation such as pallor, sweating, and elevation in heart rate, and position the patients appropriately so they do not aspirate, if there is emesis.
• The hospitalists’ only duty is to perform the sedation, but monitoring should be performed by qualified personnel according to American Academy of Pediatrics, American Society of Anesthesiologists, and local institutional guidelines. At St. Louis Children’s Hospital, a trained sedation nurse is present during and after sedation. Rescue equipment including continuous positive airway pressure (CPAP) bag, and suction and airway supplies should be readily available, if needed.
• Administer 100% oxygen for 2-3 minutes after turning off the N2O to prevent diffusion hypoxia and improve scavenging of exhaled N2O and, thus, reduce the environmental N2O exposure to sedation providers, nursing staff, and family members.
• Ensure that scavenging systems and proper airflow parameters are in place for rooms that use N2O to reduce N2O exposure. Use passive air monitoring badges to measure exposure and avoid exposing pregnant health care workers and family members by using good delivery techniques.
• Contraindications to N2O sedation include, but are not limited to patients with any trapped air in a body cavity, such as those with bowel obstruction, recent tympanoplasty, or recent intraocular surgery; patients with elevated intracranial pressure; and those with a deficiency in vitamin B12 or the enzyme 5, 10 methylenetetrahydrofolate reductase. Caution should be used in sickle cell patients, although gastroesophageal reflux disease is not a contraindication.
It has been noted that there have been reports of infertility, blood dyscrasias, and neurologic deficits with chronic exposure to nitrous oxide exposure. However, N2O has been used safely in dental offices and OR suites for several decades.
The Washington University team included Dr. Mythili Srinivasan, Dr. Jon Chiles, Dr. Mike Turmelle, and Dr. Yasmeen Daud.
Washington University School of Medicine in St. Louis has led the charge for pediatric hospitalist sedation, offering a panel of experts who shared their insights on nitrous oxide sedation in a wide-ranging, hands-on session at Pediatric Hospital Medicine 2013. Some highlights of that session were:
• Administer 50%-70% nitrous oxide (N2O) based on the need for minimal, moderate, or deep sedation. Consider N2O alone for procedures such as joint injections or voiding cystourethrograms.
• Augment N2O with 0.15-0.3 mg/kg of oxycodone (maximum dose of 10 mg) to achieve deeper sedation and greater analgesia for more painful procedures such as abscess incision and drainage, burn debridement, and postoperative dressing change. To maximize the analgesic effect of the opioid, wait for its effect to peak at 60 minutes before administering N2O.
• The peak effect of N2O is usually seen within 2-3 minutes, so plan to perform the most painful portion of the procedure at least 2 minutes after the start of 70% N2O. Titrate to 50% after about 5 minutes of 70% N2O, with a total duration of N2O not to exceed 15 minutes.
• Be alert for signs of emesis during sedation such as pallor, sweating, and elevation in heart rate, and position the patients appropriately so they do not aspirate, if there is emesis.
• The hospitalists’ only duty is to perform the sedation, but monitoring should be performed by qualified personnel according to American Academy of Pediatrics, American Society of Anesthesiologists, and local institutional guidelines. At St. Louis Children’s Hospital, a trained sedation nurse is present during and after sedation. Rescue equipment including continuous positive airway pressure (CPAP) bag, and suction and airway supplies should be readily available, if needed.
• Administer 100% oxygen for 2-3 minutes after turning off the N2O to prevent diffusion hypoxia and improve scavenging of exhaled N2O and, thus, reduce the environmental N2O exposure to sedation providers, nursing staff, and family members.
• Ensure that scavenging systems and proper airflow parameters are in place for rooms that use N2O to reduce N2O exposure. Use passive air monitoring badges to measure exposure and avoid exposing pregnant health care workers and family members by using good delivery techniques.
• Contraindications to N2O sedation include, but are not limited to patients with any trapped air in a body cavity, such as those with bowel obstruction, recent tympanoplasty, or recent intraocular surgery; patients with elevated intracranial pressure; and those with a deficiency in vitamin B12 or the enzyme 5, 10 methylenetetrahydrofolate reductase. Caution should be used in sickle cell patients, although gastroesophageal reflux disease is not a contraindication.
It has been noted that there have been reports of infertility, blood dyscrasias, and neurologic deficits with chronic exposure to nitrous oxide exposure. However, N2O has been used safely in dental offices and OR suites for several decades.
The Washington University team included Dr. Mythili Srinivasan, Dr. Jon Chiles, Dr. Mike Turmelle, and Dr. Yasmeen Daud.
Washington University School of Medicine in St. Louis has led the charge for pediatric hospitalist sedation, offering a panel of experts who shared their insights on nitrous oxide sedation in a wide-ranging, hands-on session at Pediatric Hospital Medicine 2013. Some highlights of that session were:
• Administer 50%-70% nitrous oxide (N2O) based on the need for minimal, moderate, or deep sedation. Consider N2O alone for procedures such as joint injections or voiding cystourethrograms.
• Augment N2O with 0.15-0.3 mg/kg of oxycodone (maximum dose of 10 mg) to achieve deeper sedation and greater analgesia for more painful procedures such as abscess incision and drainage, burn debridement, and postoperative dressing change. To maximize the analgesic effect of the opioid, wait for its effect to peak at 60 minutes before administering N2O.
• The peak effect of N2O is usually seen within 2-3 minutes, so plan to perform the most painful portion of the procedure at least 2 minutes after the start of 70% N2O. Titrate to 50% after about 5 minutes of 70% N2O, with a total duration of N2O not to exceed 15 minutes.
• Be alert for signs of emesis during sedation such as pallor, sweating, and elevation in heart rate, and position the patients appropriately so they do not aspirate, if there is emesis.
• The hospitalists’ only duty is to perform the sedation, but monitoring should be performed by qualified personnel according to American Academy of Pediatrics, American Society of Anesthesiologists, and local institutional guidelines. At St. Louis Children’s Hospital, a trained sedation nurse is present during and after sedation. Rescue equipment including continuous positive airway pressure (CPAP) bag, and suction and airway supplies should be readily available, if needed.
• Administer 100% oxygen for 2-3 minutes after turning off the N2O to prevent diffusion hypoxia and improve scavenging of exhaled N2O and, thus, reduce the environmental N2O exposure to sedation providers, nursing staff, and family members.
• Ensure that scavenging systems and proper airflow parameters are in place for rooms that use N2O to reduce N2O exposure. Use passive air monitoring badges to measure exposure and avoid exposing pregnant health care workers and family members by using good delivery techniques.
• Contraindications to N2O sedation include, but are not limited to patients with any trapped air in a body cavity, such as those with bowel obstruction, recent tympanoplasty, or recent intraocular surgery; patients with elevated intracranial pressure; and those with a deficiency in vitamin B12 or the enzyme 5, 10 methylenetetrahydrofolate reductase. Caution should be used in sickle cell patients, although gastroesophageal reflux disease is not a contraindication.
It has been noted that there have been reports of infertility, blood dyscrasias, and neurologic deficits with chronic exposure to nitrous oxide exposure. However, N2O has been used safely in dental offices and OR suites for several decades.
The Washington University team included Dr. Mythili Srinivasan, Dr. Jon Chiles, Dr. Mike Turmelle, and Dr. Yasmeen Daud.
Pediatric hospital medicine certification moving forward
NEW ORLEANS – The process for creating a new pediatric hospital medicine subspecialty continues to move forward, but concerns are being raised that a PHM fellowship may not meet the needs of community hospitalists and could cut off the pipeline of potential talent.
A recent medical school graduate put a very real and poignant face to those concerns during a panel session at the Pediatric Hospital Medicine 2013 meeting, where experts spent the better part of an hour clarifying and updating the subspecialty process. With $500,000 in medical school debts at more than 6% interest for himself and his physician spouse, the fellowship is not an option.
"I see (the fellowship) as a potential risk for our subspecialty to join the other subspecialties as being one that is in shortage in rural areas and unattractive to a large portion of our workforce," he said.
Ultimately, the goal of the PHM fellowship track is to provide the best patient care for hospitalized children and to move the field forward, panel members stressed. In April 2013, national PHM leaders convened to review multiple sources of data and debate options, before overwhelmingly agreeing that the practice of pediatric hospital medicine requires postresidency training (16 for, 1 against, 1 abstain) and that a 2-year accredited fellowship would be the best option (16 for, 2 abstain).
"I see (the fellowship) as a potential risk for our subspecialty to join the other subspecialties as being one that is in shortage in rural areas and unattractive to a large portion of our workforce..."
An audience member said the new fellowship is a good idea and will be particularly beneficial to pediatric hospitalists in the community setting both for patient trust and peer recognition. She went on to say, however, that the skill set for community pediatric hospitalists is unique in terms of the breadth of knowledge and services they provide and that a strong working relationship will be needed with hospitalists in the university setting, where the fellowship programs will be based, to ensure that community hospitalists get the skill sets they need.
Panelist Dr. Rebecca Blankenburg of Stanford (Calif.) University, said the issue has been a "huge area of conversation" for the group and "what we realized, certainly in fellowship, people have different training goals and training needs. But in all fellowships, we should have very strong community partnerships and so the university programs are really exploring that."
Dr. Blankenburg said the group would welcome input on community hospitalists’ skill sets and that a subgroup is also looking at how those skills should look in residency as well as in fellowship training.
Having worked in both the community and university setting, panelist Dr. Mary Ottolini, vice chair of medical education, Children’s National Medical Center, Washington, D.C., said PHM certification will offer enrichment to both groups.
"The more we can have our training programs bridge these two types of hospital medicine settings, it is just going to strengthen pediatric hospital medicine as a whole," she said.
Panelist Daniel Rauch, associate director of pediatrics at Elmhurst Hospital Center, Queens, N.Y., said there was strong feeling that a 3-year fellowship would be more appropriate for hospitalists to be prepared for work in the community setting. Ultimately, though, they had to balance this with workforce issues and people’s willingness to endure more training after residency.
A 2-year program is not the current standard for fellowships at the American Board of Pediatrics, so the ABP may not accept the group’s petition to create the new subspecialty. Even if it does, it will take awhile. The process for petitioning the ABP for subspecialty status typically takes 6 years, and it could be another 1-2 years once a certifying examination has been implemented before the Accreditation Council for Graduate Medical Education begins the fellowship program accreditation process.
Pediatric hospitalists who are currently in practice or in a currently nonaccredited fellowship will be "grandfathered" in and will be able to sit for the exam the first three test dates, if they meet experiential standards, according to the panel, led by Dr. Jeffrey Simmons, director of pediatric hospital medicine fellowship, Cincinnati Children’s Hospital and Medical Center.
The test will be given every other year, so there will be 4 years from the first test for non–fellowship-trained individuals to be eligible to take the exam. This process allows everyone currently practicing PHM and, most future pediatric hospitalists for at least the next 7-8 years, the ability to sit for the exam, according to the panel.
An audience member expressed concern about meeting the standards for certification, and asked whether the group had considered offering 6 months intensive training in specific areas such as pediatric sedation or pediatric lines for early career hospitalists like himself, whose nearest academic center is 150 miles away and who can’t take 2 years off the job for a fellowship.
Audience member Dr. Gail McGuinness, ABP executive vice president, said that experts in hospital medicine joined the ABP in setting the minimal criteria to sit for the boards.
"The goal with early certification is really to be inclusive and not prevent people from gaining certification, recognizing the training in the future is going to be somewhat different from the training, or lack of training, that those of you who are practicing the subspecialty now have," she said.
That said, Dr. McGuiness observed that "a new certificate has to say something to the public that everyone who holds that certificate has had the same experiences and same standardized training" and suggested that individuals who feel they have deficits in procedural areas need to address those deficits.
Once the fellowship accreditation process goes online, then only ACGME-approved fellowships will be able to have their graduates sit for the exam. Hospitalists who take the PHM subspecialty board, however, do not need to maintain board certification in general pediatrics once PHM certification is attained.
The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. The panel reported having no financial disclosures.
NEW ORLEANS – The process for creating a new pediatric hospital medicine subspecialty continues to move forward, but concerns are being raised that a PHM fellowship may not meet the needs of community hospitalists and could cut off the pipeline of potential talent.
A recent medical school graduate put a very real and poignant face to those concerns during a panel session at the Pediatric Hospital Medicine 2013 meeting, where experts spent the better part of an hour clarifying and updating the subspecialty process. With $500,000 in medical school debts at more than 6% interest for himself and his physician spouse, the fellowship is not an option.
"I see (the fellowship) as a potential risk for our subspecialty to join the other subspecialties as being one that is in shortage in rural areas and unattractive to a large portion of our workforce," he said.
Ultimately, the goal of the PHM fellowship track is to provide the best patient care for hospitalized children and to move the field forward, panel members stressed. In April 2013, national PHM leaders convened to review multiple sources of data and debate options, before overwhelmingly agreeing that the practice of pediatric hospital medicine requires postresidency training (16 for, 1 against, 1 abstain) and that a 2-year accredited fellowship would be the best option (16 for, 2 abstain).
"I see (the fellowship) as a potential risk for our subspecialty to join the other subspecialties as being one that is in shortage in rural areas and unattractive to a large portion of our workforce..."
An audience member said the new fellowship is a good idea and will be particularly beneficial to pediatric hospitalists in the community setting both for patient trust and peer recognition. She went on to say, however, that the skill set for community pediatric hospitalists is unique in terms of the breadth of knowledge and services they provide and that a strong working relationship will be needed with hospitalists in the university setting, where the fellowship programs will be based, to ensure that community hospitalists get the skill sets they need.
Panelist Dr. Rebecca Blankenburg of Stanford (Calif.) University, said the issue has been a "huge area of conversation" for the group and "what we realized, certainly in fellowship, people have different training goals and training needs. But in all fellowships, we should have very strong community partnerships and so the university programs are really exploring that."
Dr. Blankenburg said the group would welcome input on community hospitalists’ skill sets and that a subgroup is also looking at how those skills should look in residency as well as in fellowship training.
Having worked in both the community and university setting, panelist Dr. Mary Ottolini, vice chair of medical education, Children’s National Medical Center, Washington, D.C., said PHM certification will offer enrichment to both groups.
"The more we can have our training programs bridge these two types of hospital medicine settings, it is just going to strengthen pediatric hospital medicine as a whole," she said.
Panelist Daniel Rauch, associate director of pediatrics at Elmhurst Hospital Center, Queens, N.Y., said there was strong feeling that a 3-year fellowship would be more appropriate for hospitalists to be prepared for work in the community setting. Ultimately, though, they had to balance this with workforce issues and people’s willingness to endure more training after residency.
A 2-year program is not the current standard for fellowships at the American Board of Pediatrics, so the ABP may not accept the group’s petition to create the new subspecialty. Even if it does, it will take awhile. The process for petitioning the ABP for subspecialty status typically takes 6 years, and it could be another 1-2 years once a certifying examination has been implemented before the Accreditation Council for Graduate Medical Education begins the fellowship program accreditation process.
Pediatric hospitalists who are currently in practice or in a currently nonaccredited fellowship will be "grandfathered" in and will be able to sit for the exam the first three test dates, if they meet experiential standards, according to the panel, led by Dr. Jeffrey Simmons, director of pediatric hospital medicine fellowship, Cincinnati Children’s Hospital and Medical Center.
The test will be given every other year, so there will be 4 years from the first test for non–fellowship-trained individuals to be eligible to take the exam. This process allows everyone currently practicing PHM and, most future pediatric hospitalists for at least the next 7-8 years, the ability to sit for the exam, according to the panel.
An audience member expressed concern about meeting the standards for certification, and asked whether the group had considered offering 6 months intensive training in specific areas such as pediatric sedation or pediatric lines for early career hospitalists like himself, whose nearest academic center is 150 miles away and who can’t take 2 years off the job for a fellowship.
Audience member Dr. Gail McGuinness, ABP executive vice president, said that experts in hospital medicine joined the ABP in setting the minimal criteria to sit for the boards.
"The goal with early certification is really to be inclusive and not prevent people from gaining certification, recognizing the training in the future is going to be somewhat different from the training, or lack of training, that those of you who are practicing the subspecialty now have," she said.
That said, Dr. McGuiness observed that "a new certificate has to say something to the public that everyone who holds that certificate has had the same experiences and same standardized training" and suggested that individuals who feel they have deficits in procedural areas need to address those deficits.
Once the fellowship accreditation process goes online, then only ACGME-approved fellowships will be able to have their graduates sit for the exam. Hospitalists who take the PHM subspecialty board, however, do not need to maintain board certification in general pediatrics once PHM certification is attained.
The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. The panel reported having no financial disclosures.
NEW ORLEANS – The process for creating a new pediatric hospital medicine subspecialty continues to move forward, but concerns are being raised that a PHM fellowship may not meet the needs of community hospitalists and could cut off the pipeline of potential talent.
A recent medical school graduate put a very real and poignant face to those concerns during a panel session at the Pediatric Hospital Medicine 2013 meeting, where experts spent the better part of an hour clarifying and updating the subspecialty process. With $500,000 in medical school debts at more than 6% interest for himself and his physician spouse, the fellowship is not an option.
"I see (the fellowship) as a potential risk for our subspecialty to join the other subspecialties as being one that is in shortage in rural areas and unattractive to a large portion of our workforce," he said.
Ultimately, the goal of the PHM fellowship track is to provide the best patient care for hospitalized children and to move the field forward, panel members stressed. In April 2013, national PHM leaders convened to review multiple sources of data and debate options, before overwhelmingly agreeing that the practice of pediatric hospital medicine requires postresidency training (16 for, 1 against, 1 abstain) and that a 2-year accredited fellowship would be the best option (16 for, 2 abstain).
"I see (the fellowship) as a potential risk for our subspecialty to join the other subspecialties as being one that is in shortage in rural areas and unattractive to a large portion of our workforce..."
An audience member said the new fellowship is a good idea and will be particularly beneficial to pediatric hospitalists in the community setting both for patient trust and peer recognition. She went on to say, however, that the skill set for community pediatric hospitalists is unique in terms of the breadth of knowledge and services they provide and that a strong working relationship will be needed with hospitalists in the university setting, where the fellowship programs will be based, to ensure that community hospitalists get the skill sets they need.
Panelist Dr. Rebecca Blankenburg of Stanford (Calif.) University, said the issue has been a "huge area of conversation" for the group and "what we realized, certainly in fellowship, people have different training goals and training needs. But in all fellowships, we should have very strong community partnerships and so the university programs are really exploring that."
Dr. Blankenburg said the group would welcome input on community hospitalists’ skill sets and that a subgroup is also looking at how those skills should look in residency as well as in fellowship training.
Having worked in both the community and university setting, panelist Dr. Mary Ottolini, vice chair of medical education, Children’s National Medical Center, Washington, D.C., said PHM certification will offer enrichment to both groups.
"The more we can have our training programs bridge these two types of hospital medicine settings, it is just going to strengthen pediatric hospital medicine as a whole," she said.
Panelist Daniel Rauch, associate director of pediatrics at Elmhurst Hospital Center, Queens, N.Y., said there was strong feeling that a 3-year fellowship would be more appropriate for hospitalists to be prepared for work in the community setting. Ultimately, though, they had to balance this with workforce issues and people’s willingness to endure more training after residency.
A 2-year program is not the current standard for fellowships at the American Board of Pediatrics, so the ABP may not accept the group’s petition to create the new subspecialty. Even if it does, it will take awhile. The process for petitioning the ABP for subspecialty status typically takes 6 years, and it could be another 1-2 years once a certifying examination has been implemented before the Accreditation Council for Graduate Medical Education begins the fellowship program accreditation process.
Pediatric hospitalists who are currently in practice or in a currently nonaccredited fellowship will be "grandfathered" in and will be able to sit for the exam the first three test dates, if they meet experiential standards, according to the panel, led by Dr. Jeffrey Simmons, director of pediatric hospital medicine fellowship, Cincinnati Children’s Hospital and Medical Center.
The test will be given every other year, so there will be 4 years from the first test for non–fellowship-trained individuals to be eligible to take the exam. This process allows everyone currently practicing PHM and, most future pediatric hospitalists for at least the next 7-8 years, the ability to sit for the exam, according to the panel.
An audience member expressed concern about meeting the standards for certification, and asked whether the group had considered offering 6 months intensive training in specific areas such as pediatric sedation or pediatric lines for early career hospitalists like himself, whose nearest academic center is 150 miles away and who can’t take 2 years off the job for a fellowship.
Audience member Dr. Gail McGuinness, ABP executive vice president, said that experts in hospital medicine joined the ABP in setting the minimal criteria to sit for the boards.
"The goal with early certification is really to be inclusive and not prevent people from gaining certification, recognizing the training in the future is going to be somewhat different from the training, or lack of training, that those of you who are practicing the subspecialty now have," she said.
That said, Dr. McGuiness observed that "a new certificate has to say something to the public that everyone who holds that certificate has had the same experiences and same standardized training" and suggested that individuals who feel they have deficits in procedural areas need to address those deficits.
Once the fellowship accreditation process goes online, then only ACGME-approved fellowships will be able to have their graduates sit for the exam. Hospitalists who take the PHM subspecialty board, however, do not need to maintain board certification in general pediatrics once PHM certification is attained.
The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. The panel reported having no financial disclosures.
AT PEDIATRIC HOSPITAL MEDICINE 2013
Hospitalist comanagement cuts LOS for pediatric neurosurgery patients
NEW ORLEANS – Hospitalist comanagement of pediatric neurosurgical cases significantly shortens hospital length of stay without increasing readmissions or complications, according to a retrospective analysis of 526 patients.
The mean LOS fell from 8.6 days to 5.6 days and median LOS from 4 days to 3 days after the transition to hospitalist comanagement. Both differences were significant.
Comanagement also had a great impact on the variability of LOS, as demonstrated by a reduction in the standard deviation from 20.0 to 7.2, observed Dr. Catherine Sullivan, with Connecticut Children’s Medical Center in Hartford.
"It looked like our greatest effect was on the variability of the outliers," she said at Pediatric Hospital Medicine 2013.
Ten patients stayed for more than 50 days before hospitalist comanagement was implemented, whereas none did in the time after.
One extreme outlier stayed for about 400 days prior to comanagement, and likely explains the large discrepancy between the mean and median LOS, Dr. Sullivan explained.
The chart review involved 347 neurosurgical patients cared for in the 2 years prior to hospitalist comanagement and 179 patients cared for in the year after the launch of the program in March 2011. Children with routine ventriculoperitoneal shunt revision or those hospitalized for brief neurologic observations were excluded.
Two staff neurosurgeons provided education to hospitalists and pediatric residents prior to implementation.
Hospitalists provided care in the areas of nutrition, pulmonary clearance, pain management, rehabilitation, and discharge planning. Areas where hospitalists could write orders without running them by the neurosurgeon were hashed out in a departmental meeting at the attending level. A monthly hospitalist meeting was also dedicated to the project.
"We did really focus on that communication piece," Dr. Sullivan said. "I think that’s probably a challenge between many medical and surgical teams. It was a change in culture, and it has improved relationships between the two divisions."
About 80% of pediatric neurosurgical cases have been hospitalist-comanaged since the project began. The LOS reduction was maintained across neurosurgeons.
Despite the shorter stay, no differences have been observed in rates of complications or readmissions, Dr. Sullivan said.
The study sparked a lively debate, with audience members lauding the relevancy of the study, but also questioning whether the involvement of pediatric residents or other factors might have impacted LOS.
Dr. Sullivan acknowledged this was hard to tease out since the pediatric residents also began to follow these patients, and had done so "only peripherally" in the past.
"So whether the pediatric residents impacted the length of stay as much as, or more than, the hospitalists, is really not clear," she said.
Other attendees suggested performing subgroup analyses or a case-mix adjustment to strengthen the results, or applying a winsoring technique to address the statistical outliers.
"Because the data were so striking, one of the first things we did was to have our people go back and make sure we weren’t having an increase in readmissions, and, in fact, we weren’t," Dr. Sullivan remarked at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
An informal poll was performed, however, with only favorable responses received from both neurosurgeons regarding the areas of communication, management, and perceived provider and family satisfaction, she said.
The hospital is looking at expanding the project to include other surgical subspecialties such as orthopedics, urology, ENT, craniofacial, and ophthalmology, particularly for cases with high co-medical morbidities such as spinal fusion or Pierre Robin malformation sequences, Dr. Sullivan said.
Cost analyses are also ongoing, and the hospital is debating whether to have hospitalists meet with patients upon admission rather than after surgery. However, "it creates a staffing challenge of who is going to cover that visit," Dr. Sullivan said
Session comoderator and pediatric hospitalist Dr. Jack Percelay, with E.L.M.O. Pediatrics in New York, encouraged Dr. Sullivan and her colleagues to publish the data, observing that "it really struck a chord."
Dr. Sullivan and her coauthors reported having no financial disclosures.
NEW ORLEANS – Hospitalist comanagement of pediatric neurosurgical cases significantly shortens hospital length of stay without increasing readmissions or complications, according to a retrospective analysis of 526 patients.
The mean LOS fell from 8.6 days to 5.6 days and median LOS from 4 days to 3 days after the transition to hospitalist comanagement. Both differences were significant.
Comanagement also had a great impact on the variability of LOS, as demonstrated by a reduction in the standard deviation from 20.0 to 7.2, observed Dr. Catherine Sullivan, with Connecticut Children’s Medical Center in Hartford.
"It looked like our greatest effect was on the variability of the outliers," she said at Pediatric Hospital Medicine 2013.
Ten patients stayed for more than 50 days before hospitalist comanagement was implemented, whereas none did in the time after.
One extreme outlier stayed for about 400 days prior to comanagement, and likely explains the large discrepancy between the mean and median LOS, Dr. Sullivan explained.
The chart review involved 347 neurosurgical patients cared for in the 2 years prior to hospitalist comanagement and 179 patients cared for in the year after the launch of the program in March 2011. Children with routine ventriculoperitoneal shunt revision or those hospitalized for brief neurologic observations were excluded.
Two staff neurosurgeons provided education to hospitalists and pediatric residents prior to implementation.
Hospitalists provided care in the areas of nutrition, pulmonary clearance, pain management, rehabilitation, and discharge planning. Areas where hospitalists could write orders without running them by the neurosurgeon were hashed out in a departmental meeting at the attending level. A monthly hospitalist meeting was also dedicated to the project.
"We did really focus on that communication piece," Dr. Sullivan said. "I think that’s probably a challenge between many medical and surgical teams. It was a change in culture, and it has improved relationships between the two divisions."
About 80% of pediatric neurosurgical cases have been hospitalist-comanaged since the project began. The LOS reduction was maintained across neurosurgeons.
Despite the shorter stay, no differences have been observed in rates of complications or readmissions, Dr. Sullivan said.
The study sparked a lively debate, with audience members lauding the relevancy of the study, but also questioning whether the involvement of pediatric residents or other factors might have impacted LOS.
Dr. Sullivan acknowledged this was hard to tease out since the pediatric residents also began to follow these patients, and had done so "only peripherally" in the past.
"So whether the pediatric residents impacted the length of stay as much as, or more than, the hospitalists, is really not clear," she said.
Other attendees suggested performing subgroup analyses or a case-mix adjustment to strengthen the results, or applying a winsoring technique to address the statistical outliers.
"Because the data were so striking, one of the first things we did was to have our people go back and make sure we weren’t having an increase in readmissions, and, in fact, we weren’t," Dr. Sullivan remarked at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
An informal poll was performed, however, with only favorable responses received from both neurosurgeons regarding the areas of communication, management, and perceived provider and family satisfaction, she said.
The hospital is looking at expanding the project to include other surgical subspecialties such as orthopedics, urology, ENT, craniofacial, and ophthalmology, particularly for cases with high co-medical morbidities such as spinal fusion or Pierre Robin malformation sequences, Dr. Sullivan said.
Cost analyses are also ongoing, and the hospital is debating whether to have hospitalists meet with patients upon admission rather than after surgery. However, "it creates a staffing challenge of who is going to cover that visit," Dr. Sullivan said
Session comoderator and pediatric hospitalist Dr. Jack Percelay, with E.L.M.O. Pediatrics in New York, encouraged Dr. Sullivan and her colleagues to publish the data, observing that "it really struck a chord."
Dr. Sullivan and her coauthors reported having no financial disclosures.
NEW ORLEANS – Hospitalist comanagement of pediatric neurosurgical cases significantly shortens hospital length of stay without increasing readmissions or complications, according to a retrospective analysis of 526 patients.
The mean LOS fell from 8.6 days to 5.6 days and median LOS from 4 days to 3 days after the transition to hospitalist comanagement. Both differences were significant.
Comanagement also had a great impact on the variability of LOS, as demonstrated by a reduction in the standard deviation from 20.0 to 7.2, observed Dr. Catherine Sullivan, with Connecticut Children’s Medical Center in Hartford.
"It looked like our greatest effect was on the variability of the outliers," she said at Pediatric Hospital Medicine 2013.
Ten patients stayed for more than 50 days before hospitalist comanagement was implemented, whereas none did in the time after.
One extreme outlier stayed for about 400 days prior to comanagement, and likely explains the large discrepancy between the mean and median LOS, Dr. Sullivan explained.
The chart review involved 347 neurosurgical patients cared for in the 2 years prior to hospitalist comanagement and 179 patients cared for in the year after the launch of the program in March 2011. Children with routine ventriculoperitoneal shunt revision or those hospitalized for brief neurologic observations were excluded.
Two staff neurosurgeons provided education to hospitalists and pediatric residents prior to implementation.
Hospitalists provided care in the areas of nutrition, pulmonary clearance, pain management, rehabilitation, and discharge planning. Areas where hospitalists could write orders without running them by the neurosurgeon were hashed out in a departmental meeting at the attending level. A monthly hospitalist meeting was also dedicated to the project.
"We did really focus on that communication piece," Dr. Sullivan said. "I think that’s probably a challenge between many medical and surgical teams. It was a change in culture, and it has improved relationships between the two divisions."
About 80% of pediatric neurosurgical cases have been hospitalist-comanaged since the project began. The LOS reduction was maintained across neurosurgeons.
Despite the shorter stay, no differences have been observed in rates of complications or readmissions, Dr. Sullivan said.
The study sparked a lively debate, with audience members lauding the relevancy of the study, but also questioning whether the involvement of pediatric residents or other factors might have impacted LOS.
Dr. Sullivan acknowledged this was hard to tease out since the pediatric residents also began to follow these patients, and had done so "only peripherally" in the past.
"So whether the pediatric residents impacted the length of stay as much as, or more than, the hospitalists, is really not clear," she said.
Other attendees suggested performing subgroup analyses or a case-mix adjustment to strengthen the results, or applying a winsoring technique to address the statistical outliers.
"Because the data were so striking, one of the first things we did was to have our people go back and make sure we weren’t having an increase in readmissions, and, in fact, we weren’t," Dr. Sullivan remarked at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
An informal poll was performed, however, with only favorable responses received from both neurosurgeons regarding the areas of communication, management, and perceived provider and family satisfaction, she said.
The hospital is looking at expanding the project to include other surgical subspecialties such as orthopedics, urology, ENT, craniofacial, and ophthalmology, particularly for cases with high co-medical morbidities such as spinal fusion or Pierre Robin malformation sequences, Dr. Sullivan said.
Cost analyses are also ongoing, and the hospital is debating whether to have hospitalists meet with patients upon admission rather than after surgery. However, "it creates a staffing challenge of who is going to cover that visit," Dr. Sullivan said
Session comoderator and pediatric hospitalist Dr. Jack Percelay, with E.L.M.O. Pediatrics in New York, encouraged Dr. Sullivan and her colleagues to publish the data, observing that "it really struck a chord."
Dr. Sullivan and her coauthors reported having no financial disclosures.
AT PEDIATRIC HOSPITAL MEDICINE 2013
Major finding: After the transition to hospitalist comanagement, the mean length of stay fell significantly, from 8.6 days to 5.6 days, and the median LOS fell from 4 days to 3 days.
Data source: Chart review of 526 pediatric neurosurgery cases.
Disclosures: Dr. Sullivan and her coauthors reported having no financial disclosures.
Trio of studies tackles trends affecting pediatric residents
NEW ORLEANS – Inpatient staffing in the wake of tighter residency work hours, avoiding premature diagnostic closure, and hospitalist perceptions of resident effects on quality of care and costs were tackled in a trio of oral abstracts presented at Pediatric Hospital Medicine 2013.
Work-rules effects
A Web-based survey of 152 pediatric residency programs in the United States revealed marked variation in how programs have responded after the Accreditation Council for Graduate Medical Education mandated new residency work hour restrictions in July 2011.
The most common changes were the addition of a resident night-float system and an increase in the number of attending physicians present at night, primarily in the form of pediatric hospitalists and pediatric intensivists, said Dr. Jennifer Oshimura, with Indiana University, Riley Hospital for Children, Indianapolis.
The number of programs using a night-float system increased from 43% to 71% after the new residency work hours, while the number of programs utilizing resident admission caps increased only slightly, from 12% to 15%.
In all, 23% of programs increased night in-house attending coverage following the work-hour restrictions.
Of these, 63% of programs increased their night-time attending coverage by adding pediatric hospitalists, 37% added pediatric intensivists, and 26% added neonatologists, Dr. Oshimura said.
Overall, the number of programs with pediatric hospitalist attendings in-house 24/7 rose from 16% to 20%, with 41% of programs planning to add this kind of coverage within the next 5 years.
Finally, only 12% of residency programs had no in-house attending coverage at night of any kind. This compares with 22% of programs in a previous study conducted by the group in 2010, before the work hour restrictions were in place, she noted.
The anonymous survey was sent to 198 pediatric residency training programs in the United States. The response rate was 77% (152/198), representing 7,828 pediatric residents or 79% of all pediatric residents in the United States.
The programs varied "drastically" in terms of size (average, 51.5 residents; range, 6-168) and average daily patient census per ward intern during the daytime (7.4 patients; range, 2-30), Dr. Oshimura remarked.
Premature closure
Results of a randomized trial show that simulated patient encounters can be used to help teach residents to avoid premature closure, or the tendency to accept a diagnosis before it is fully verified. The phenomenon prevents consideration of alternative diagnoses and is a common source of error in the care of inpatients.
"Premature closure can be summarized with the axiom: ‘When the diagnosis is made, the thinking stops,’ " Dr. Lauren Nassetta said at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
Dr. Nassetta and her colleagues at the University of Alabama at Birmingham randomly assigned 61 residents on inpatient pediatrics rotations to receive a text page with a brief description of a patient that contained either an incorrect diagnosis (stem A) or a symptom (stem B). The remainder of the case was carefully scripted and presented identically. Each of the 36 teams received 2 cases, and the cases were paired such that residents who received stem A for case 1 received stem B for case 2, and vice versa.
The cases involved common pediatric presentations for unusual reasons, such as a 14-month-old who presents with wheezing, secondary to congestive heart failure. For this case, the incorrect diagnosis was given as asthma.
The residents had 10 minutes to evaluate (history, physical exam, laboratory, and radiology) and treat simulated patients. High-fidelity mannequins were used to provide responses to the residents, while investigators played the role of nurse and family member.
Residents obtained significantly more elements in the evaluation when they received the symptom stem versus the incorrect diagnosis stem (63% vs. 55%; P less than .05), Dr. Nassetta said.
Residents given the symptom stem made the correct diagnosis 86% of the time, compared with only 69% of the time when given the incorrect diagnosis. The difference did not reach statistical significance at the group level (P = .16), but was significant on an individual participant level.
Still, the finding resulted in a relative risk of 2.2, "meaning that having the incorrect diagnosis made it twice as likely to miss the correct diagnosis," she said.
When data from individual participants were used in a multivariable logistic regression model, the adjusted odds ratio was 14.9 for making a correct diagnosis with the symptom stem vs. the incorrect diagnosis. The model adjusted for a variety of variables, including prior simulation time, case difficulty, level of training, program type, and confidence in independent care of patients.
Other predictors of making a correct diagnosis were obtaining a patient history (adjusted OR, 1.35) and obtaining an exam, labs, and radiology (aOR, 1.47).
"It’s obvious that residents lack insight into the causes of premature closure, but this does set us up for some opportunities for education," Dr. Nassetta said. "We can use simulated cases to demonstrate diagnostic error, discuss premature closure, and strategies for avoiding it."
Strategies she employs with residents include stressing the importance of the history and physical exam, having residents ask what doesn’t fit the diagnosis they have in mind, encouraging them to come up with one alternative diagnosis for every patient they see, and having residents describe rather than diagnose the patient at the time of handoff.
Are residents perceived as pricey?
Most pediatric hospitalists surveyed think residents increase the quality of care, but do so at increased cost to the hospital, according to a 15-item, anonymous, electronic survey of 127 pediatric hospitalists at 113 institutions.
Overall, 60% of respondents thought residents increase the quality of care, 23% said they have no impact, and 17% thought they actually decrease quality, said Dr. David P. Johnson, of Monroe Carell Jr. Children’s Hospital at Vanderbilt, Nashville, Tenn.
Subgroup analyses showed that 71% of hospitalists who always work with residents thought residents improve the quality of care, whereas only 43% of those who sometimes do, and 53% who never do felt the same (P less than .05).
Hospitalists affiliated with academic hospitals were also significantly more likely to say residents improve quality than were those at community hospitals (65% vs. 38%), he said.
In all, 72% of respondents said residents increase hospital costs, while 23% said they had no impact and 6% said they decrease costs.
In subgroup analyses, however, the perception that residents increase costs was similar no matter how much respondents worked with residents (71%, 71%, 73%; always, sometimes, never) or what their affiliation was (69% academic, 83% community), Dr. Johnson said.
One of the most intriguing findings from the survey was that 78% of hospitalists deny having any formal training in resource utilization, yet 91% think they provide cost-effective care, and 81% feel they are qualified to teach it, Dr. Johnson said.
"A gap exists between what we think we know and the training that we received," he said. "I don’t think that’s necessarily a bad thing, but it’s something we need to be cognizant of, both as we are taking care of our patients and teaching our residents."
Dr. Johnson hypothesized that informal training, reading, and being at the right place at the right time might explain some of the hospitalists’ confidence in the absence of formal training, but that experience may also matter.
Hospitalists with more than 10 years of experience were significantly more likely to say they had the knowledge to provide cost-effective care, compared with those with 6-10 years of experience or 5 years or less (100% vs. 91% vs. 85%; P less than .05). A similar, but nonsignificant, trend was present when respondents were asked whether they were qualified to teach cost-effective care (93% vs. 77% vs. 75%).
At baseline, 31% of respondents had been in practice for more than 10 years, 28% for 6-10 years, and 41% for 0-5 years. The majority had an academic affiliation (81%); 55% always worked with residents, 33% sometimes, and 12% never.
Dr. Oshimura, Dr. Nassetta, and Dr. Johnson reported having no financial disclosures.
NEW ORLEANS – Inpatient staffing in the wake of tighter residency work hours, avoiding premature diagnostic closure, and hospitalist perceptions of resident effects on quality of care and costs were tackled in a trio of oral abstracts presented at Pediatric Hospital Medicine 2013.
Work-rules effects
A Web-based survey of 152 pediatric residency programs in the United States revealed marked variation in how programs have responded after the Accreditation Council for Graduate Medical Education mandated new residency work hour restrictions in July 2011.
The most common changes were the addition of a resident night-float system and an increase in the number of attending physicians present at night, primarily in the form of pediatric hospitalists and pediatric intensivists, said Dr. Jennifer Oshimura, with Indiana University, Riley Hospital for Children, Indianapolis.
The number of programs using a night-float system increased from 43% to 71% after the new residency work hours, while the number of programs utilizing resident admission caps increased only slightly, from 12% to 15%.
In all, 23% of programs increased night in-house attending coverage following the work-hour restrictions.
Of these, 63% of programs increased their night-time attending coverage by adding pediatric hospitalists, 37% added pediatric intensivists, and 26% added neonatologists, Dr. Oshimura said.
Overall, the number of programs with pediatric hospitalist attendings in-house 24/7 rose from 16% to 20%, with 41% of programs planning to add this kind of coverage within the next 5 years.
Finally, only 12% of residency programs had no in-house attending coverage at night of any kind. This compares with 22% of programs in a previous study conducted by the group in 2010, before the work hour restrictions were in place, she noted.
The anonymous survey was sent to 198 pediatric residency training programs in the United States. The response rate was 77% (152/198), representing 7,828 pediatric residents or 79% of all pediatric residents in the United States.
The programs varied "drastically" in terms of size (average, 51.5 residents; range, 6-168) and average daily patient census per ward intern during the daytime (7.4 patients; range, 2-30), Dr. Oshimura remarked.
Premature closure
Results of a randomized trial show that simulated patient encounters can be used to help teach residents to avoid premature closure, or the tendency to accept a diagnosis before it is fully verified. The phenomenon prevents consideration of alternative diagnoses and is a common source of error in the care of inpatients.
"Premature closure can be summarized with the axiom: ‘When the diagnosis is made, the thinking stops,’ " Dr. Lauren Nassetta said at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
Dr. Nassetta and her colleagues at the University of Alabama at Birmingham randomly assigned 61 residents on inpatient pediatrics rotations to receive a text page with a brief description of a patient that contained either an incorrect diagnosis (stem A) or a symptom (stem B). The remainder of the case was carefully scripted and presented identically. Each of the 36 teams received 2 cases, and the cases were paired such that residents who received stem A for case 1 received stem B for case 2, and vice versa.
The cases involved common pediatric presentations for unusual reasons, such as a 14-month-old who presents with wheezing, secondary to congestive heart failure. For this case, the incorrect diagnosis was given as asthma.
The residents had 10 minutes to evaluate (history, physical exam, laboratory, and radiology) and treat simulated patients. High-fidelity mannequins were used to provide responses to the residents, while investigators played the role of nurse and family member.
Residents obtained significantly more elements in the evaluation when they received the symptom stem versus the incorrect diagnosis stem (63% vs. 55%; P less than .05), Dr. Nassetta said.
Residents given the symptom stem made the correct diagnosis 86% of the time, compared with only 69% of the time when given the incorrect diagnosis. The difference did not reach statistical significance at the group level (P = .16), but was significant on an individual participant level.
Still, the finding resulted in a relative risk of 2.2, "meaning that having the incorrect diagnosis made it twice as likely to miss the correct diagnosis," she said.
When data from individual participants were used in a multivariable logistic regression model, the adjusted odds ratio was 14.9 for making a correct diagnosis with the symptom stem vs. the incorrect diagnosis. The model adjusted for a variety of variables, including prior simulation time, case difficulty, level of training, program type, and confidence in independent care of patients.
Other predictors of making a correct diagnosis were obtaining a patient history (adjusted OR, 1.35) and obtaining an exam, labs, and radiology (aOR, 1.47).
"It’s obvious that residents lack insight into the causes of premature closure, but this does set us up for some opportunities for education," Dr. Nassetta said. "We can use simulated cases to demonstrate diagnostic error, discuss premature closure, and strategies for avoiding it."
Strategies she employs with residents include stressing the importance of the history and physical exam, having residents ask what doesn’t fit the diagnosis they have in mind, encouraging them to come up with one alternative diagnosis for every patient they see, and having residents describe rather than diagnose the patient at the time of handoff.
Are residents perceived as pricey?
Most pediatric hospitalists surveyed think residents increase the quality of care, but do so at increased cost to the hospital, according to a 15-item, anonymous, electronic survey of 127 pediatric hospitalists at 113 institutions.
Overall, 60% of respondents thought residents increase the quality of care, 23% said they have no impact, and 17% thought they actually decrease quality, said Dr. David P. Johnson, of Monroe Carell Jr. Children’s Hospital at Vanderbilt, Nashville, Tenn.
Subgroup analyses showed that 71% of hospitalists who always work with residents thought residents improve the quality of care, whereas only 43% of those who sometimes do, and 53% who never do felt the same (P less than .05).
Hospitalists affiliated with academic hospitals were also significantly more likely to say residents improve quality than were those at community hospitals (65% vs. 38%), he said.
In all, 72% of respondents said residents increase hospital costs, while 23% said they had no impact and 6% said they decrease costs.
In subgroup analyses, however, the perception that residents increase costs was similar no matter how much respondents worked with residents (71%, 71%, 73%; always, sometimes, never) or what their affiliation was (69% academic, 83% community), Dr. Johnson said.
One of the most intriguing findings from the survey was that 78% of hospitalists deny having any formal training in resource utilization, yet 91% think they provide cost-effective care, and 81% feel they are qualified to teach it, Dr. Johnson said.
"A gap exists between what we think we know and the training that we received," he said. "I don’t think that’s necessarily a bad thing, but it’s something we need to be cognizant of, both as we are taking care of our patients and teaching our residents."
Dr. Johnson hypothesized that informal training, reading, and being at the right place at the right time might explain some of the hospitalists’ confidence in the absence of formal training, but that experience may also matter.
Hospitalists with more than 10 years of experience were significantly more likely to say they had the knowledge to provide cost-effective care, compared with those with 6-10 years of experience or 5 years or less (100% vs. 91% vs. 85%; P less than .05). A similar, but nonsignificant, trend was present when respondents were asked whether they were qualified to teach cost-effective care (93% vs. 77% vs. 75%).
At baseline, 31% of respondents had been in practice for more than 10 years, 28% for 6-10 years, and 41% for 0-5 years. The majority had an academic affiliation (81%); 55% always worked with residents, 33% sometimes, and 12% never.
Dr. Oshimura, Dr. Nassetta, and Dr. Johnson reported having no financial disclosures.
NEW ORLEANS – Inpatient staffing in the wake of tighter residency work hours, avoiding premature diagnostic closure, and hospitalist perceptions of resident effects on quality of care and costs were tackled in a trio of oral abstracts presented at Pediatric Hospital Medicine 2013.
Work-rules effects
A Web-based survey of 152 pediatric residency programs in the United States revealed marked variation in how programs have responded after the Accreditation Council for Graduate Medical Education mandated new residency work hour restrictions in July 2011.
The most common changes were the addition of a resident night-float system and an increase in the number of attending physicians present at night, primarily in the form of pediatric hospitalists and pediatric intensivists, said Dr. Jennifer Oshimura, with Indiana University, Riley Hospital for Children, Indianapolis.
The number of programs using a night-float system increased from 43% to 71% after the new residency work hours, while the number of programs utilizing resident admission caps increased only slightly, from 12% to 15%.
In all, 23% of programs increased night in-house attending coverage following the work-hour restrictions.
Of these, 63% of programs increased their night-time attending coverage by adding pediatric hospitalists, 37% added pediatric intensivists, and 26% added neonatologists, Dr. Oshimura said.
Overall, the number of programs with pediatric hospitalist attendings in-house 24/7 rose from 16% to 20%, with 41% of programs planning to add this kind of coverage within the next 5 years.
Finally, only 12% of residency programs had no in-house attending coverage at night of any kind. This compares with 22% of programs in a previous study conducted by the group in 2010, before the work hour restrictions were in place, she noted.
The anonymous survey was sent to 198 pediatric residency training programs in the United States. The response rate was 77% (152/198), representing 7,828 pediatric residents or 79% of all pediatric residents in the United States.
The programs varied "drastically" in terms of size (average, 51.5 residents; range, 6-168) and average daily patient census per ward intern during the daytime (7.4 patients; range, 2-30), Dr. Oshimura remarked.
Premature closure
Results of a randomized trial show that simulated patient encounters can be used to help teach residents to avoid premature closure, or the tendency to accept a diagnosis before it is fully verified. The phenomenon prevents consideration of alternative diagnoses and is a common source of error in the care of inpatients.
"Premature closure can be summarized with the axiom: ‘When the diagnosis is made, the thinking stops,’ " Dr. Lauren Nassetta said at the meeting, sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association.
Dr. Nassetta and her colleagues at the University of Alabama at Birmingham randomly assigned 61 residents on inpatient pediatrics rotations to receive a text page with a brief description of a patient that contained either an incorrect diagnosis (stem A) or a symptom (stem B). The remainder of the case was carefully scripted and presented identically. Each of the 36 teams received 2 cases, and the cases were paired such that residents who received stem A for case 1 received stem B for case 2, and vice versa.
The cases involved common pediatric presentations for unusual reasons, such as a 14-month-old who presents with wheezing, secondary to congestive heart failure. For this case, the incorrect diagnosis was given as asthma.
The residents had 10 minutes to evaluate (history, physical exam, laboratory, and radiology) and treat simulated patients. High-fidelity mannequins were used to provide responses to the residents, while investigators played the role of nurse and family member.
Residents obtained significantly more elements in the evaluation when they received the symptom stem versus the incorrect diagnosis stem (63% vs. 55%; P less than .05), Dr. Nassetta said.
Residents given the symptom stem made the correct diagnosis 86% of the time, compared with only 69% of the time when given the incorrect diagnosis. The difference did not reach statistical significance at the group level (P = .16), but was significant on an individual participant level.
Still, the finding resulted in a relative risk of 2.2, "meaning that having the incorrect diagnosis made it twice as likely to miss the correct diagnosis," she said.
When data from individual participants were used in a multivariable logistic regression model, the adjusted odds ratio was 14.9 for making a correct diagnosis with the symptom stem vs. the incorrect diagnosis. The model adjusted for a variety of variables, including prior simulation time, case difficulty, level of training, program type, and confidence in independent care of patients.
Other predictors of making a correct diagnosis were obtaining a patient history (adjusted OR, 1.35) and obtaining an exam, labs, and radiology (aOR, 1.47).
"It’s obvious that residents lack insight into the causes of premature closure, but this does set us up for some opportunities for education," Dr. Nassetta said. "We can use simulated cases to demonstrate diagnostic error, discuss premature closure, and strategies for avoiding it."
Strategies she employs with residents include stressing the importance of the history and physical exam, having residents ask what doesn’t fit the diagnosis they have in mind, encouraging them to come up with one alternative diagnosis for every patient they see, and having residents describe rather than diagnose the patient at the time of handoff.
Are residents perceived as pricey?
Most pediatric hospitalists surveyed think residents increase the quality of care, but do so at increased cost to the hospital, according to a 15-item, anonymous, electronic survey of 127 pediatric hospitalists at 113 institutions.
Overall, 60% of respondents thought residents increase the quality of care, 23% said they have no impact, and 17% thought they actually decrease quality, said Dr. David P. Johnson, of Monroe Carell Jr. Children’s Hospital at Vanderbilt, Nashville, Tenn.
Subgroup analyses showed that 71% of hospitalists who always work with residents thought residents improve the quality of care, whereas only 43% of those who sometimes do, and 53% who never do felt the same (P less than .05).
Hospitalists affiliated with academic hospitals were also significantly more likely to say residents improve quality than were those at community hospitals (65% vs. 38%), he said.
In all, 72% of respondents said residents increase hospital costs, while 23% said they had no impact and 6% said they decrease costs.
In subgroup analyses, however, the perception that residents increase costs was similar no matter how much respondents worked with residents (71%, 71%, 73%; always, sometimes, never) or what their affiliation was (69% academic, 83% community), Dr. Johnson said.
One of the most intriguing findings from the survey was that 78% of hospitalists deny having any formal training in resource utilization, yet 91% think they provide cost-effective care, and 81% feel they are qualified to teach it, Dr. Johnson said.
"A gap exists between what we think we know and the training that we received," he said. "I don’t think that’s necessarily a bad thing, but it’s something we need to be cognizant of, both as we are taking care of our patients and teaching our residents."
Dr. Johnson hypothesized that informal training, reading, and being at the right place at the right time might explain some of the hospitalists’ confidence in the absence of formal training, but that experience may also matter.
Hospitalists with more than 10 years of experience were significantly more likely to say they had the knowledge to provide cost-effective care, compared with those with 6-10 years of experience or 5 years or less (100% vs. 91% vs. 85%; P less than .05). A similar, but nonsignificant, trend was present when respondents were asked whether they were qualified to teach cost-effective care (93% vs. 77% vs. 75%).
At baseline, 31% of respondents had been in practice for more than 10 years, 28% for 6-10 years, and 41% for 0-5 years. The majority had an academic affiliation (81%); 55% always worked with residents, 33% sometimes, and 12% never.
Dr. Oshimura, Dr. Nassetta, and Dr. Johnson reported having no financial disclosures.
AT PEDIATRIC HOSPITAL MEDICINE 2013
Study points to 24-hour window to rule out bacteremia
NEW ORLEANS – Monitoring blood cultures for more than 24 hours may be unnecessary to rule out bacteremia in febrile, nontoxic infants, a new study suggests.
Among 393 blood cultures drawn at 17 institutions, 9.7% turned positive after 24 hours, compared with 3.8% after 36 hours and 1.8% after 48 hours, Dr. Eric Biondi said at the Pediatric Hospital Medicine 2013 meeting.
"Our data demonstrate that about 90% of blood cultures from febrile infants are going to turn positive by 24 hours when they are positive, and more than 95% are going to turn positive by 36 hours," said Dr. Biondi of the University of Rochester (N.Y.) Medical Center.
Using the Pediatric Research in Inpatient Settings (PRIS) Network, the investigators performed a retrospective chart review of 393 positive blood cultures drawn between January 2009 and January 2013 from infants less than 91 days old admitted for fever and treated as pathogenic. Their average age was 39 days and 51% were male.
Samples drawn from infants who had central lines, a surgical history, or were admitted to the ICU in less than 5 hours after the culture were excluded.
Overall Escherichia coli was the most common pathogen (41%), although there was significant variability of species between sites when compared with the reference site in Minnesota (P = .04), Dr. Biondi said. E. coli was the most common at 12 of the 17 sites, while group B streptococcus was the most common at 5.
The median time to positivity did not vary dramatically between species or sites, reaching a median of 13 hours for E. coli (range 12-15 hours) and 11 hours for group B strep (range 9-12 hours).
Based on the generally accepted premise that 2% of febrile infants will have bacteremia, the number needed to treat would be 515 infants for one blood culture to turn positive in more than 24 hours, Dr. Biondi said. That number increases to 1,315 infants after 36 hours and to 6,250 infants after 48 hours.
"You have to think about this in context," he remarked. "Even though about 10% of samples will turn positive after 24 hours, we’re talking about a group that includes only 2% of infants in the first place."
Although ruling out sepsis in febrile infants is one of the most common tasks hospitalists do, the inpatient observational period varies widely from 24 to as much as 72 hours. Dr. Biondi called for a prospective study to develop a national approach, but said that the current data are strong enough to guide clinicians now. The samples were all analyzed with an automated detection system and the PRIS Network is geographically diverse.
"If you’ve got a well-appearing kid staring at you and you’re just waiting for your blood culture, I’m hoping we can use these data to say, ‘We’re probably safe to send this one home,’ "he said.
Dr. Shawn Ralston, session comoderator and chief of the pediatrics section at Children’s Hospital at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said that she trained in the era of the 72-hour rule-out and described the self-funded, multicenter study as a tremendous undertaking that provides clinically important data hospitalists can use in the management of febrile infants.
"This contributes to patient-centered decision making with the number-needed-to-treat analysis, and I think it’s awesome," she said.
Dr. Biondi said in an interview that he and his colleagues are in the early stages of designing a prospective feasibility study at three sites that would provide more detailed information on positive, as well as negative, cultures and provide pilot data that could be used for a national study with the PRIS Network.
"We hope, in the end, that these data will lead to a cultural shift in the way we ‘rule out sepsis,’ which will allow us to treat kids with fewer doses of antibiotics and get them home with their families, where they belong," he said.
The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. Dr. Biondi and his coauthors reported having no financial disclosures.
NEW ORLEANS – Monitoring blood cultures for more than 24 hours may be unnecessary to rule out bacteremia in febrile, nontoxic infants, a new study suggests.
Among 393 blood cultures drawn at 17 institutions, 9.7% turned positive after 24 hours, compared with 3.8% after 36 hours and 1.8% after 48 hours, Dr. Eric Biondi said at the Pediatric Hospital Medicine 2013 meeting.
"Our data demonstrate that about 90% of blood cultures from febrile infants are going to turn positive by 24 hours when they are positive, and more than 95% are going to turn positive by 36 hours," said Dr. Biondi of the University of Rochester (N.Y.) Medical Center.
Using the Pediatric Research in Inpatient Settings (PRIS) Network, the investigators performed a retrospective chart review of 393 positive blood cultures drawn between January 2009 and January 2013 from infants less than 91 days old admitted for fever and treated as pathogenic. Their average age was 39 days and 51% were male.
Samples drawn from infants who had central lines, a surgical history, or were admitted to the ICU in less than 5 hours after the culture were excluded.
Overall Escherichia coli was the most common pathogen (41%), although there was significant variability of species between sites when compared with the reference site in Minnesota (P = .04), Dr. Biondi said. E. coli was the most common at 12 of the 17 sites, while group B streptococcus was the most common at 5.
The median time to positivity did not vary dramatically between species or sites, reaching a median of 13 hours for E. coli (range 12-15 hours) and 11 hours for group B strep (range 9-12 hours).
Based on the generally accepted premise that 2% of febrile infants will have bacteremia, the number needed to treat would be 515 infants for one blood culture to turn positive in more than 24 hours, Dr. Biondi said. That number increases to 1,315 infants after 36 hours and to 6,250 infants after 48 hours.
"You have to think about this in context," he remarked. "Even though about 10% of samples will turn positive after 24 hours, we’re talking about a group that includes only 2% of infants in the first place."
Although ruling out sepsis in febrile infants is one of the most common tasks hospitalists do, the inpatient observational period varies widely from 24 to as much as 72 hours. Dr. Biondi called for a prospective study to develop a national approach, but said that the current data are strong enough to guide clinicians now. The samples were all analyzed with an automated detection system and the PRIS Network is geographically diverse.
"If you’ve got a well-appearing kid staring at you and you’re just waiting for your blood culture, I’m hoping we can use these data to say, ‘We’re probably safe to send this one home,’ "he said.
Dr. Shawn Ralston, session comoderator and chief of the pediatrics section at Children’s Hospital at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said that she trained in the era of the 72-hour rule-out and described the self-funded, multicenter study as a tremendous undertaking that provides clinically important data hospitalists can use in the management of febrile infants.
"This contributes to patient-centered decision making with the number-needed-to-treat analysis, and I think it’s awesome," she said.
Dr. Biondi said in an interview that he and his colleagues are in the early stages of designing a prospective feasibility study at three sites that would provide more detailed information on positive, as well as negative, cultures and provide pilot data that could be used for a national study with the PRIS Network.
"We hope, in the end, that these data will lead to a cultural shift in the way we ‘rule out sepsis,’ which will allow us to treat kids with fewer doses of antibiotics and get them home with their families, where they belong," he said.
The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. Dr. Biondi and his coauthors reported having no financial disclosures.
NEW ORLEANS – Monitoring blood cultures for more than 24 hours may be unnecessary to rule out bacteremia in febrile, nontoxic infants, a new study suggests.
Among 393 blood cultures drawn at 17 institutions, 9.7% turned positive after 24 hours, compared with 3.8% after 36 hours and 1.8% after 48 hours, Dr. Eric Biondi said at the Pediatric Hospital Medicine 2013 meeting.
"Our data demonstrate that about 90% of blood cultures from febrile infants are going to turn positive by 24 hours when they are positive, and more than 95% are going to turn positive by 36 hours," said Dr. Biondi of the University of Rochester (N.Y.) Medical Center.
Using the Pediatric Research in Inpatient Settings (PRIS) Network, the investigators performed a retrospective chart review of 393 positive blood cultures drawn between January 2009 and January 2013 from infants less than 91 days old admitted for fever and treated as pathogenic. Their average age was 39 days and 51% were male.
Samples drawn from infants who had central lines, a surgical history, or were admitted to the ICU in less than 5 hours after the culture were excluded.
Overall Escherichia coli was the most common pathogen (41%), although there was significant variability of species between sites when compared with the reference site in Minnesota (P = .04), Dr. Biondi said. E. coli was the most common at 12 of the 17 sites, while group B streptococcus was the most common at 5.
The median time to positivity did not vary dramatically between species or sites, reaching a median of 13 hours for E. coli (range 12-15 hours) and 11 hours for group B strep (range 9-12 hours).
Based on the generally accepted premise that 2% of febrile infants will have bacteremia, the number needed to treat would be 515 infants for one blood culture to turn positive in more than 24 hours, Dr. Biondi said. That number increases to 1,315 infants after 36 hours and to 6,250 infants after 48 hours.
"You have to think about this in context," he remarked. "Even though about 10% of samples will turn positive after 24 hours, we’re talking about a group that includes only 2% of infants in the first place."
Although ruling out sepsis in febrile infants is one of the most common tasks hospitalists do, the inpatient observational period varies widely from 24 to as much as 72 hours. Dr. Biondi called for a prospective study to develop a national approach, but said that the current data are strong enough to guide clinicians now. The samples were all analyzed with an automated detection system and the PRIS Network is geographically diverse.
"If you’ve got a well-appearing kid staring at you and you’re just waiting for your blood culture, I’m hoping we can use these data to say, ‘We’re probably safe to send this one home,’ "he said.
Dr. Shawn Ralston, session comoderator and chief of the pediatrics section at Children’s Hospital at Dartmouth-Hitchcock Medical Center, Lebanon, N.H., said that she trained in the era of the 72-hour rule-out and described the self-funded, multicenter study as a tremendous undertaking that provides clinically important data hospitalists can use in the management of febrile infants.
"This contributes to patient-centered decision making with the number-needed-to-treat analysis, and I think it’s awesome," she said.
Dr. Biondi said in an interview that he and his colleagues are in the early stages of designing a prospective feasibility study at three sites that would provide more detailed information on positive, as well as negative, cultures and provide pilot data that could be used for a national study with the PRIS Network.
"We hope, in the end, that these data will lead to a cultural shift in the way we ‘rule out sepsis,’ which will allow us to treat kids with fewer doses of antibiotics and get them home with their families, where they belong," he said.
The meeting was sponsored by the Society of Hospital Medicine, the American Academy of Pediatrics, and the Academic Pediatric Association. Dr. Biondi and his coauthors reported having no financial disclosures.
AT PEDIATRIC HOSPITAL MEDICINE 2013
Major finding: The number needed to treat for one blood culture to turn positive after 24 hours was 515 infants admitted for fever.
Data source: Multicenter, retrospective cross-sectional study of 393 positive blood cultures.
Disclosures: Dr. Biondi and his coauthors reported having no financial disclosures.