User login
Link Between PPIs and C. difficile Inconsistent
CHICAGO – The effect of proton pump inhibitors on Clostridium difficile infection was neither strong nor consistent in a longitudinal hospital cohort of 61,834 patients.
Previously conducted case-control studies reported that PPIs were associated with a two- to threefold increase in the risk of C. difficile infection (CDI), but these latest findings suggest that the impact of PPIs may be overstated, Dr. Kyoung Sup Hong said at the annual Digestive Disease Week.
In his study, CDI incidence per 1,000 person-years was 3.5 in patients receiving PPIs for less than 3 months (group 1), 7.4 in those on PPIs for 3 months, but less than 1 year, (group 2), and 4.5 in those on PPIs for at least 1 year (group 3).
In logistic regression analysis that adjusted for PPI exposure, age, comorbid conditions, and antibiotic use in the previous 8 weeks, the association between PPI duration and CDI was significant, increasing from an odds ratio of 1 in group 1 (reference) to 2.59 in group 2 (P value less than .01), and to 2.17 in group 3 (P less than .01), Dr. Hong said.
Notably, the use of antibiotics in the previous 8 weeks significantly increased the risk of C. difficile infection to a whopping odds ratio of 31.73 (P less than .01).
In a Cox proportional hazard model that further adjusted for the interval between PPI start to CDI attack or last follow-up, however, the association between PPI use and CDI was significant only for group 2 (hazard ratio 1.94, P less than .001) and not group 3 (HR. 1.22, P = .25), said Dr. Hong of Seoul (South Korea) National University.
"We conclude that PPIs’ impact on CDI is neither strong nor consistent; therefore, PPIs seem to be an important confounder rather than a cause of CDI," he said.
A recent study among hospital discharges (Arch. Intern. Med. 2010;170:784-90) reported a dose-response effect with acid suppression and the risk of CDI, but Dr. Hong noted that it provided no information about medications before admission. In that study, the risk of CDI on or after the third hospital day increased from 0.3% in nonusers to 0.6% in those receiving histamine2 receptor antagonist therapy, to 0.9% in those receiving daily PPI therapy, and to 1.4% in those receiving more-frequent PPI therapy.
Dr. Hong and his colleagues aimed to evaluate the effect of PPI treatment duration on CDI development among all adults, older than age 20 years, who visited the Seoul National University Hospital and took a prescription PPI from January 2005 to December 2009.
Among the 61,834 patients, there were 534 CDI cases, of which 5 were identified with endoscopy only, and the remaining 529 by a positive C. difficile toxin assay.
CDI was reported in 319 of the 50,534 (.63%) patients in group 1; 176 of the 9,122 (1.94%) patients in group 2; and 38 of the 2,178 (1.74%) patients in group 3. The average follow-up time from the first PPI prescription until the last visit was 22 months, 32 months, and 47 months, respectively.
Patients infected with C. difficile were significantly older (61 years) than those without CDI at (56 years) (P less than .01). Age remained a significant risk factor for CDI in both multivariate logistic regression (OR 1.016, P less than .001) and Cox proportional (HR 1.018, P less than .001) analyses.
Dr. Hong acknowledged that the study was limited by use of a hospital cohort, and thus the data could not be translated directly to the community.
Dr. Hong and his colleagues reported no conflicts of interest.
CHICAGO – The effect of proton pump inhibitors on Clostridium difficile infection was neither strong nor consistent in a longitudinal hospital cohort of 61,834 patients.
Previously conducted case-control studies reported that PPIs were associated with a two- to threefold increase in the risk of C. difficile infection (CDI), but these latest findings suggest that the impact of PPIs may be overstated, Dr. Kyoung Sup Hong said at the annual Digestive Disease Week.
In his study, CDI incidence per 1,000 person-years was 3.5 in patients receiving PPIs for less than 3 months (group 1), 7.4 in those on PPIs for 3 months, but less than 1 year, (group 2), and 4.5 in those on PPIs for at least 1 year (group 3).
In logistic regression analysis that adjusted for PPI exposure, age, comorbid conditions, and antibiotic use in the previous 8 weeks, the association between PPI duration and CDI was significant, increasing from an odds ratio of 1 in group 1 (reference) to 2.59 in group 2 (P value less than .01), and to 2.17 in group 3 (P less than .01), Dr. Hong said.
Notably, the use of antibiotics in the previous 8 weeks significantly increased the risk of C. difficile infection to a whopping odds ratio of 31.73 (P less than .01).
In a Cox proportional hazard model that further adjusted for the interval between PPI start to CDI attack or last follow-up, however, the association between PPI use and CDI was significant only for group 2 (hazard ratio 1.94, P less than .001) and not group 3 (HR. 1.22, P = .25), said Dr. Hong of Seoul (South Korea) National University.
"We conclude that PPIs’ impact on CDI is neither strong nor consistent; therefore, PPIs seem to be an important confounder rather than a cause of CDI," he said.
A recent study among hospital discharges (Arch. Intern. Med. 2010;170:784-90) reported a dose-response effect with acid suppression and the risk of CDI, but Dr. Hong noted that it provided no information about medications before admission. In that study, the risk of CDI on or after the third hospital day increased from 0.3% in nonusers to 0.6% in those receiving histamine2 receptor antagonist therapy, to 0.9% in those receiving daily PPI therapy, and to 1.4% in those receiving more-frequent PPI therapy.
Dr. Hong and his colleagues aimed to evaluate the effect of PPI treatment duration on CDI development among all adults, older than age 20 years, who visited the Seoul National University Hospital and took a prescription PPI from January 2005 to December 2009.
Among the 61,834 patients, there were 534 CDI cases, of which 5 were identified with endoscopy only, and the remaining 529 by a positive C. difficile toxin assay.
CDI was reported in 319 of the 50,534 (.63%) patients in group 1; 176 of the 9,122 (1.94%) patients in group 2; and 38 of the 2,178 (1.74%) patients in group 3. The average follow-up time from the first PPI prescription until the last visit was 22 months, 32 months, and 47 months, respectively.
Patients infected with C. difficile were significantly older (61 years) than those without CDI at (56 years) (P less than .01). Age remained a significant risk factor for CDI in both multivariate logistic regression (OR 1.016, P less than .001) and Cox proportional (HR 1.018, P less than .001) analyses.
Dr. Hong acknowledged that the study was limited by use of a hospital cohort, and thus the data could not be translated directly to the community.
Dr. Hong and his colleagues reported no conflicts of interest.
CHICAGO – The effect of proton pump inhibitors on Clostridium difficile infection was neither strong nor consistent in a longitudinal hospital cohort of 61,834 patients.
Previously conducted case-control studies reported that PPIs were associated with a two- to threefold increase in the risk of C. difficile infection (CDI), but these latest findings suggest that the impact of PPIs may be overstated, Dr. Kyoung Sup Hong said at the annual Digestive Disease Week.
In his study, CDI incidence per 1,000 person-years was 3.5 in patients receiving PPIs for less than 3 months (group 1), 7.4 in those on PPIs for 3 months, but less than 1 year, (group 2), and 4.5 in those on PPIs for at least 1 year (group 3).
In logistic regression analysis that adjusted for PPI exposure, age, comorbid conditions, and antibiotic use in the previous 8 weeks, the association between PPI duration and CDI was significant, increasing from an odds ratio of 1 in group 1 (reference) to 2.59 in group 2 (P value less than .01), and to 2.17 in group 3 (P less than .01), Dr. Hong said.
Notably, the use of antibiotics in the previous 8 weeks significantly increased the risk of C. difficile infection to a whopping odds ratio of 31.73 (P less than .01).
In a Cox proportional hazard model that further adjusted for the interval between PPI start to CDI attack or last follow-up, however, the association between PPI use and CDI was significant only for group 2 (hazard ratio 1.94, P less than .001) and not group 3 (HR. 1.22, P = .25), said Dr. Hong of Seoul (South Korea) National University.
"We conclude that PPIs’ impact on CDI is neither strong nor consistent; therefore, PPIs seem to be an important confounder rather than a cause of CDI," he said.
A recent study among hospital discharges (Arch. Intern. Med. 2010;170:784-90) reported a dose-response effect with acid suppression and the risk of CDI, but Dr. Hong noted that it provided no information about medications before admission. In that study, the risk of CDI on or after the third hospital day increased from 0.3% in nonusers to 0.6% in those receiving histamine2 receptor antagonist therapy, to 0.9% in those receiving daily PPI therapy, and to 1.4% in those receiving more-frequent PPI therapy.
Dr. Hong and his colleagues aimed to evaluate the effect of PPI treatment duration on CDI development among all adults, older than age 20 years, who visited the Seoul National University Hospital and took a prescription PPI from January 2005 to December 2009.
Among the 61,834 patients, there were 534 CDI cases, of which 5 were identified with endoscopy only, and the remaining 529 by a positive C. difficile toxin assay.
CDI was reported in 319 of the 50,534 (.63%) patients in group 1; 176 of the 9,122 (1.94%) patients in group 2; and 38 of the 2,178 (1.74%) patients in group 3. The average follow-up time from the first PPI prescription until the last visit was 22 months, 32 months, and 47 months, respectively.
Patients infected with C. difficile were significantly older (61 years) than those without CDI at (56 years) (P less than .01). Age remained a significant risk factor for CDI in both multivariate logistic regression (OR 1.016, P less than .001) and Cox proportional (HR 1.018, P less than .001) analyses.
Dr. Hong acknowledged that the study was limited by use of a hospital cohort, and thus the data could not be translated directly to the community.
Dr. Hong and his colleagues reported no conflicts of interest.
FROM THE ANNUAL DIGESTIVE DISEASE WEEK
Major Finding: In an adjusted Cox proportional hazard model, the association between proton pump inhibitor use and C. difficile infection was significant for patients on PPIs for 3 months, but less than 1 year, (hazard ratio, 1.94), but not for those on PPIs for at least 1 year (HR, 1.22).
Data Source: Longitudinal hospital cohort study of 61,834 patients treated at Seoul (South Korea) National University Hospital.
Disclosures: Dr. Hong and his colleagues reported no conflicts of interest.
Childhood Cancer Survivors at Higher Risk for Melanoma Risk
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The cumulative incidence of a first subsequent melanoma was 0.52% at 35 years from initial cancer.
Data Source: Retrospective analysis of 14,358 5-year childhood cancer survivors.
Disclosures: The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
Childhood Cancer Survivors at Higher Risk for Melanoma
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease, and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development was 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms' tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease, and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development was 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms' tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease, and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development was 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms' tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The cumulative incidence of a first subsequent melanoma was 0.52% at 35 years from initial cancer.
Data Source: Retrospective analysis of 14,358 5-year childhood cancer survivors.
Disclosures: The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
Childhood Cancer Survivors at Higher Risk for Melanoma Risk
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The cumulative incidence of a first subsequent melanoma was 0.52% at 35 years from initial cancer.
Data Source: Retrospective analysis of 14,358 5-year childhood cancer survivors.
Disclosures: The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
Childhood Cancer Survivors at Higher Risk for Melanoma Risk
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
CHICAGO – Survivors of childhood cancer have a nearly threefold increased risk of melanoma, compared with the general population, according to an analysis of the Childhood Cancer Survivor Study.
Among 14,358 5-year survivors, the cumulative incidence of a subsequent melanoma was 0.52% at 35 years from their initial cancer. The standard incidence ratio for subsequent melanoma was 2.83 and excess absolute risk 0.11/1,000 person-years.
"Although the cumulative incidence is low, this is potentially a significant problem," lead author Dr. Alberto Pappo said at the annual meeting of the American Society of Clinical Oncology. "Our findings justify increased awareness and the need for improved adherence to recommended surveillance guidelines in childhood cancer survivors."
Dr. Pappo said the analysis was sparked by a report last year from the Childhood Cancer Survivor Study showing that 5-year survivors had an increased cumulative incidence of subsequent neoplasms (20.5%) as well as second malignant neoplasms (8%) (J. Natl. Cancer Inst. 2010 Jul 21;102:1083-95).
The 14,358 patients in the current analysis were diagnosed with childhood cancer between 1970 and 1986, and compared with participants in the Surveillance, Epidemiology, and End Results (SEER) database. Median follow-up was 24 years.
In all, 53 survivors developed 59 melanomas, said Dr. Pappo, director of the solid tumor division at St. Jude Children’s Research Hospital, Memphis. Among them, two patients had ocular melanoma, nine had in situ disease and two had other secondary malignancies before the diagnosis of melanoma. Five patients developed two or more melanomas, he pointed out.
The median age at the time of melanoma diagnosis was 32 years, and median time for tumor development 20.7 years. At the time of the analysis, 43 of the 53 patients (81%) were still alive. Four patients died of melanoma.
Among the 53 melanoma patients, primary diagnoses included acute lymphoblastic leukemia (15), lymphoma (14), bone/soft tissue (15), brain (5), Wilms’ tumor (3), and neuroblastoma (1).
The cumulative incidence of melanoma ranged from 0.29% for a primary diagnosis of brain cancer to 0.87% for survivors of soft tissue and bone sarcoma. The incidence was 0.43% for childhood leukemia and 0.55% for lymphoma.
Survivors who developed melanoma were significantly more likely to be more than 10 years old at the time of their initial cancer diagnosis (P less than .001) and to have a family history of cancer (P = .01). However, univariate analysis by age at diagnosis, sex, family history of cancer, treatment era, race, and alkylating score identified no significant risk factors, he said.
Attendees asked whether there is a cut point at which clinicians should pay particular attention to melanoma in this population. Dr. Pappo replied, "I am not sure if annual skin examinations are recommended for all childhood cancer survivors or just for those who receive radiotherapy, but perhaps for specific subsets, such as those with ALL or soft tissue sarcomas, that may be something worth implementing."
Dr. Pappo pointed out that the researchers are seeking more detailed tumor location and radiotherapy exposure data and that the study lacked data on factors known to contribute to melanoma such as sun exposure, number of nevi, and modifying genes.
Invited discussant Dr. Louise C. Strong, a professor of genetics at the University of Texas M.D. Anderson Cancer Center, Houston, called for further studies to elucidate the risk factors and genetics responsible for the increased incidence of melanoma observed in the study. She went on to say that studies in other populations at higher-risk for melanoma, such as transplant and immunosuppressed patients, have shown that prognosis for early stage I and II melanoma is the same as the general population but may actually be worse for late-stage disease.
"To me this is mostly a good news message because as long as we can educate our patients and can develop prevention and intervention programs, then it should work," she said.
The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The cumulative incidence of a first subsequent melanoma was 0.52% at 35 years from initial cancer.
Data Source: Retrospective analysis of 14,358 5-year childhood cancer survivors.
Disclosures: The Childhood Cancer Survivor Study is funded by the National Cancer Institute. Dr. Pappo, his coauthors, and Dr. Strong reported no relevant conflicts of interest.
In-Hospital Mortality Risk Increases With DIDO Time for STEMI Patients
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
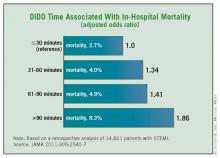
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
FROM JAMA
Major Finding: The recommended door-in to door-out time of 30 minutes or less was observed in only 11% of transferred STEMI patients requiring revascularization.
Data Source: Retrospective analysis of 14,821 patients with STEMI transferred to 298 STEMI receiving hospitals for primary PCI.
Disclosures: The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sankyo Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
In-Hospital Mortality Risk Increases With DIDO Time for STEMI Patients
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
FROM JAMA
In-Hospital Mortality Risk Increases With DIDO Time for STEMI Patients
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
Only 11% of patients with ST-elevation myocardial infarction who presented at a hospital without acute percutaneous coronary intervention capability got in and out of the referral hospital within the recommended benchmark of 30 minutes or less, in a retrospective analysis of 14,821 patients.
Moreover, STEMI patients who had a door-in to door-out (DIDO) time of more than 30 minutes had significantly higher in-hospital mortality of 5.9% compared with 2.7% for patients with a DIDO time of 30 minutes or less.
This mortality risk remained significant even after adjustment for differences in baseline patient characteristics and presenting features (adjusted odds ratio 1.56), Dr. Tracy Y. Wang and her associates reportedin the June 22/29 issue of JAMA (2011;305:2540-7).
"DIDO time is a useful performance measure attributable to STEMI referral hospitals that can be used to assess and iteratively improve effectiveness of regional STEMI networks and may further emerge as a quality benchmark to ascertain performance and accountability," the authors wrote.
They go on to suggest that "further attention and improvement of this performance measure will translate into substantial improvement in the timeliness of primary PCI and clinical outcomes for transferred STEMI patients."
Hospitals typically focus on shortening overall door-to-balloon (DTB) times as a way to improve the outcomes of STEMI patients, but little has been known about the impact of DIDO times as a component of the interhospital transfer process. DIDO times are increasingly being advocated as a new quality of care metric for transferred STEMI patients, with a national benchmark of 30 minutes or less recommended by the 2008 American College of Cardiology/American Heart Association performance measures for acute myocardial infarction (J. Am. Coll. Cardiol. 2008;52:2046-99).
Dr. Wang and her associates identified 14,821 STEMI patients transferred to 298 STEMI receiving hospitals for primary PCI in the ACTION (Acute Coronary Treatment and Intervention Outcomes Network) Registry–Get With the Guidelines during January 2007–March 2010. The median DIDO time was 68 minutes (interquartile range 43-120 minutes).
Only 11% (1,627) of patients had a DIDO time of 30 minutes or less, while 56% had a DIDO time greater than 60 minutes, and more than one-third (35%) had a DIDO time greater than 90 minutes, reported Dr. Wang of Duke Clinical Research Institute, Durham, N.C.
Patients with a DIDO time of more than 30 minutes were significantly more likely than were those with shorter DIDO times to be older; to be female; to have comorbidities such as hypertension, diabetes, and prior heart failure or stroke; and to present during off hours. A left bundle branch block or signs of posterior MI on the presenting ECG were also more common in those with prolonged DIDO times.
Notably, only a minority of patients with a prolonged DIDO time had contraindications to fibrinolytic therapy, which is the preferred reperfusion strategy for STEMI when access to timely primary PCI is not a viable option, Dr. Wang pointed out.
The percentage of patients achieving the guideline-recommended overall DTB time of 90 minutes or less was significantly higher for patients with a DIDO time of 30 minutes or less, compared with patients with a DIDO time greater than 30 minutes (60% vs. 13%), she reported. Median DTB times were significantly shorter at 85 minutes vs. 127 minutes, respectively.
The observed in-hospital mortality rate was 5.5% during the study period. The median length of hospitalization was 3 days among all patients.
Using patients with a DIDO time of 30 minutes or less as the reference, risk adjusted mortality increased as DIDO times lengthened from a range of 31-60 minutes (OR 1.34) to 61-90 minutes (OR 1.41) and beyond 90 minutes (OR 1.86).
"Our results underscore the importance of optimizing regional and statewide networks for STEMI systems of care," Dr. Wang wrote.
Most American hospitals lack round-the-clock PCI capacity, although a substantial proportion of contemporary STEMI patients require interhospital transfer for primary PCI. An analysis reported earlier this year by the same group revealed that between 2005 and 2007, fewer than 10% of transferred patients with STEMI met the metric of overall DTB time of less than 90 minutes (Am. Heart J. 2011;161:76-83, e1).
Finally, the current analysis is not without a sliver of good news. The researchers observed that overall DTB time has further improved for transferred patients with STEMI, with about 1 in 5 patients treated within a DTB time of less than 90 minutes. The proportion of patients with a DIDO time of 30 minutes or less also showed improvement over time, with median DIDO times falling from 90 minutes in January 2007 to 58 minutes in March 2010.
The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sanyko Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
FROM JAMA
Major Finding: The recommended door-in to door-out time of 30 minutes or less was observed in only 11% of transferred STEMI patients requiring revascularization.
Data Source: Retrospective analysis of 14,821 patients with STEMI transferred to 298 STEMI receiving hospitals for primary PCI.
Disclosures: The American College of Cardiology Foundation’s National Cardiovascular Data Registry sponsored the study. The ACTION Registry-GWTG is supported in part by the Bristol-Myers Squibb partnership. Dr. Wang reports receiving research grants from the BMS/Sanofi partnership, Schering Plough/Merck, The Medicines Company, Heartscape, Canyon Pharmaceuticals and Eli Lilly/Daiichi Sankyo Alliance, as well as consulting fees/honoraria from Medco and Astra Zeneca. Four of her coauthors report financial relationships with several drug firms.
Iniparib Loses Blockbuster Image in Triple-Negative Breast Cancer
CHICAGO – After having reported striking phase II data that sent both clinicians and patients with triple-negative breast cancer clamoring for iniparib, researchers are puzzling over a puny survival advantage in the phase III trial, and are questioning how the novel drug actually works.
Iniparib (BSI-201) was thought to belong to the class of targeted therapeutics known as PARP (poly [ADP (adenosine diphosphate)–ribose] polymerase) inhibitors, which have shown promise as single agents in BRCA1- and BRCA2-associated breast and ovarian tumors.
Hopes for a targeted therapy in triple-negative breast cancer were raised when iniparib plus gemcitabine and carboplatin posted significant gains over chemotherapy alone in progression-free (hazard ratio, 0.59) and overall survival (HR, 0.57) without significant added toxicity in the metastatic setting in the 123-patient, phase II study (N. Engl. J. Med. 2011;364:205-14).
Adding iniparib to the same chemotherapy doublet in the much larger phase III trial provided an insignificant survival advantage, however, and failed to meet the coprimary end points of median progression-free survival (4.1 months vs. 5.1 months; HR, 0.79; P = .027) and overall survival (11.1 months vs. 11.8 months; HR, 0.88; P = 0.28).
Exploratory analyses suggested a potential efficacy benefit among second- and third-line patients, who accounted for 43% of the study population, lead author Dr. Joyce O’Shaughnessy said at the annual meeting of the American Society of Clinical Oncology.
Among these patients, iniparib plus chemotherapy increased progression-free survival from 2.9 months with chemotherapy alone to 4.2 months (HR, 0.67), and overall survival from 8.1 months to 10.8 months (HR, 0.65). There was no difference in first-line patients in either progression-free (4.6 months vs. 5.6 months; HR, 0.88) or overall survival (12.6 months vs. 12.4 months; HR, 1.1).
Multivariate analyses that adjusted for several prespecified baselines factors and replaced "time since diagnosis of metastatic disease" with "disease-free interval from primary breast cancer surgery to onset of metastatic disease" showed a significant improvement in overall survival (HR. 0.78; P = .05) in the overall population and in second-and third-line patients (HR, 0.71; P = .05), said Dr. O’Shaughnessy of the Baylor Charles A. Sammons Cancer Center at Dallas.
The difference between the two iniparib trials could not be explained by disease-free interval, although this was not assessed in the phase II trial, she said.
It’s possible that the phase III trial was enriched with patients who had BRCA1/2 genetic mutations, as the phase II results caused what was described by one attendee as "hysteria" among BRCA1/2 carriers to get on the trial. Testing for the genetic mutation was voluntary and was completed by only a minority of patients.
Part of the problem could lie in the heterogeneous nature of triple-negative breast cancer. TNBC is made up of a diverse group of molecular subtypes, which makes it hard to classify and thus target with biologic agents. Indeed, ongoing tumor subtyping identified several molecular subtypes in the phase III trial, including luminal A, luminal B, and basal-like subtypes, Dr. O’Shaughnessy said.
Invited discussant Dr. Lisa Carey of the cancer center at the University of North Carolina at Chapel Hill said that TNBC has been used as a surrogate for molecular subtypes of biological interest, and that the triple-negative phenotype alone is unlikely to be an adequate selection strategy for PARP inhibition.
"Whenever we use triple-negative eligibility to get at a subtype that’s defined biologically, we will always be misclassifying some of these tumors," she said.
TNBC is mostly made up of the basal-like subtype, which is also present in about 80% of BRCA1-associated breast cancer. The shared phenotypic pattern between sporadic basal-like breast cancer and BRCA1-associated cancer – sometimes referred to as "BRCAness" – has therapeutic implications because one of its hallmarks is sensitivity to DNA damage, Dr. Carey explained. PARP inhibitors work by inhibiting DNA-damage repair in BRCA-associated tumors.
Although iniparib has long been included in lists of PARP inhibitors, there is some question about its mechanism of action, she said. Iniparib has substantially lower (1,000-fold) PARP inhibitory activity than do other agents in this class; it does not have additive toxicity as observed in multiple PARP trials; and recent preclinical studies show that although it induces gamma-H2AX foci (a pharmacodynamic readout of DNA damage), it does not appear to do so via PARP 1 and 2 inhibition.
"The role (and frankly the mechanism) of iniparib is still a work in progress, but you can also say that clinical activity based on this study is certainly far less than was predicted," Dr. Carey said, adding that a better understanding of the mechanism is needed for rational determination of a target population.
The phase III, open-label trial randomly assigned 519 patients who had received at least two prior cytotoxic regimens for metastatic triple-negative breast cancer to 21-day cycles of gemcitabine (1,000 mg/m2 IV) and carboplatin (area under the curve 2 IV), both on days 1 and 8, or the same chemotherapy doublet plus iniparib (5.6 mg/kg IV) on days 1, 4, 8 and 11.
The overall response rates by independent radiology review were 30% among the 258 chemotherapy patients and 34% among the 261 iniparib patients, Dr. O’Shaughnessy said. The clinical benefit rates were 36% and 41%.
The toxicity of iniparib plus gemcitabine and carboplatin was comparable with that of chemotherapy alone. One death from upper GI hemorrhage was attributed to iniparib therapy. Biomarker analyses are underway to evaluate patient populations that may benefit from iniparib, she said.
Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough and Wyeth.
CHICAGO – After having reported striking phase II data that sent both clinicians and patients with triple-negative breast cancer clamoring for iniparib, researchers are puzzling over a puny survival advantage in the phase III trial, and are questioning how the novel drug actually works.
Iniparib (BSI-201) was thought to belong to the class of targeted therapeutics known as PARP (poly [ADP (adenosine diphosphate)–ribose] polymerase) inhibitors, which have shown promise as single agents in BRCA1- and BRCA2-associated breast and ovarian tumors.
Hopes for a targeted therapy in triple-negative breast cancer were raised when iniparib plus gemcitabine and carboplatin posted significant gains over chemotherapy alone in progression-free (hazard ratio, 0.59) and overall survival (HR, 0.57) without significant added toxicity in the metastatic setting in the 123-patient, phase II study (N. Engl. J. Med. 2011;364:205-14).
Adding iniparib to the same chemotherapy doublet in the much larger phase III trial provided an insignificant survival advantage, however, and failed to meet the coprimary end points of median progression-free survival (4.1 months vs. 5.1 months; HR, 0.79; P = .027) and overall survival (11.1 months vs. 11.8 months; HR, 0.88; P = 0.28).
Exploratory analyses suggested a potential efficacy benefit among second- and third-line patients, who accounted for 43% of the study population, lead author Dr. Joyce O’Shaughnessy said at the annual meeting of the American Society of Clinical Oncology.
Among these patients, iniparib plus chemotherapy increased progression-free survival from 2.9 months with chemotherapy alone to 4.2 months (HR, 0.67), and overall survival from 8.1 months to 10.8 months (HR, 0.65). There was no difference in first-line patients in either progression-free (4.6 months vs. 5.6 months; HR, 0.88) or overall survival (12.6 months vs. 12.4 months; HR, 1.1).
Multivariate analyses that adjusted for several prespecified baselines factors and replaced "time since diagnosis of metastatic disease" with "disease-free interval from primary breast cancer surgery to onset of metastatic disease" showed a significant improvement in overall survival (HR. 0.78; P = .05) in the overall population and in second-and third-line patients (HR, 0.71; P = .05), said Dr. O’Shaughnessy of the Baylor Charles A. Sammons Cancer Center at Dallas.
The difference between the two iniparib trials could not be explained by disease-free interval, although this was not assessed in the phase II trial, she said.
It’s possible that the phase III trial was enriched with patients who had BRCA1/2 genetic mutations, as the phase II results caused what was described by one attendee as "hysteria" among BRCA1/2 carriers to get on the trial. Testing for the genetic mutation was voluntary and was completed by only a minority of patients.
Part of the problem could lie in the heterogeneous nature of triple-negative breast cancer. TNBC is made up of a diverse group of molecular subtypes, which makes it hard to classify and thus target with biologic agents. Indeed, ongoing tumor subtyping identified several molecular subtypes in the phase III trial, including luminal A, luminal B, and basal-like subtypes, Dr. O’Shaughnessy said.
Invited discussant Dr. Lisa Carey of the cancer center at the University of North Carolina at Chapel Hill said that TNBC has been used as a surrogate for molecular subtypes of biological interest, and that the triple-negative phenotype alone is unlikely to be an adequate selection strategy for PARP inhibition.
"Whenever we use triple-negative eligibility to get at a subtype that’s defined biologically, we will always be misclassifying some of these tumors," she said.
TNBC is mostly made up of the basal-like subtype, which is also present in about 80% of BRCA1-associated breast cancer. The shared phenotypic pattern between sporadic basal-like breast cancer and BRCA1-associated cancer – sometimes referred to as "BRCAness" – has therapeutic implications because one of its hallmarks is sensitivity to DNA damage, Dr. Carey explained. PARP inhibitors work by inhibiting DNA-damage repair in BRCA-associated tumors.
Although iniparib has long been included in lists of PARP inhibitors, there is some question about its mechanism of action, she said. Iniparib has substantially lower (1,000-fold) PARP inhibitory activity than do other agents in this class; it does not have additive toxicity as observed in multiple PARP trials; and recent preclinical studies show that although it induces gamma-H2AX foci (a pharmacodynamic readout of DNA damage), it does not appear to do so via PARP 1 and 2 inhibition.
"The role (and frankly the mechanism) of iniparib is still a work in progress, but you can also say that clinical activity based on this study is certainly far less than was predicted," Dr. Carey said, adding that a better understanding of the mechanism is needed for rational determination of a target population.
The phase III, open-label trial randomly assigned 519 patients who had received at least two prior cytotoxic regimens for metastatic triple-negative breast cancer to 21-day cycles of gemcitabine (1,000 mg/m2 IV) and carboplatin (area under the curve 2 IV), both on days 1 and 8, or the same chemotherapy doublet plus iniparib (5.6 mg/kg IV) on days 1, 4, 8 and 11.
The overall response rates by independent radiology review were 30% among the 258 chemotherapy patients and 34% among the 261 iniparib patients, Dr. O’Shaughnessy said. The clinical benefit rates were 36% and 41%.
The toxicity of iniparib plus gemcitabine and carboplatin was comparable with that of chemotherapy alone. One death from upper GI hemorrhage was attributed to iniparib therapy. Biomarker analyses are underway to evaluate patient populations that may benefit from iniparib, she said.
Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough and Wyeth.
CHICAGO – After having reported striking phase II data that sent both clinicians and patients with triple-negative breast cancer clamoring for iniparib, researchers are puzzling over a puny survival advantage in the phase III trial, and are questioning how the novel drug actually works.
Iniparib (BSI-201) was thought to belong to the class of targeted therapeutics known as PARP (poly [ADP (adenosine diphosphate)–ribose] polymerase) inhibitors, which have shown promise as single agents in BRCA1- and BRCA2-associated breast and ovarian tumors.
Hopes for a targeted therapy in triple-negative breast cancer were raised when iniparib plus gemcitabine and carboplatin posted significant gains over chemotherapy alone in progression-free (hazard ratio, 0.59) and overall survival (HR, 0.57) without significant added toxicity in the metastatic setting in the 123-patient, phase II study (N. Engl. J. Med. 2011;364:205-14).
Adding iniparib to the same chemotherapy doublet in the much larger phase III trial provided an insignificant survival advantage, however, and failed to meet the coprimary end points of median progression-free survival (4.1 months vs. 5.1 months; HR, 0.79; P = .027) and overall survival (11.1 months vs. 11.8 months; HR, 0.88; P = 0.28).
Exploratory analyses suggested a potential efficacy benefit among second- and third-line patients, who accounted for 43% of the study population, lead author Dr. Joyce O’Shaughnessy said at the annual meeting of the American Society of Clinical Oncology.
Among these patients, iniparib plus chemotherapy increased progression-free survival from 2.9 months with chemotherapy alone to 4.2 months (HR, 0.67), and overall survival from 8.1 months to 10.8 months (HR, 0.65). There was no difference in first-line patients in either progression-free (4.6 months vs. 5.6 months; HR, 0.88) or overall survival (12.6 months vs. 12.4 months; HR, 1.1).
Multivariate analyses that adjusted for several prespecified baselines factors and replaced "time since diagnosis of metastatic disease" with "disease-free interval from primary breast cancer surgery to onset of metastatic disease" showed a significant improvement in overall survival (HR. 0.78; P = .05) in the overall population and in second-and third-line patients (HR, 0.71; P = .05), said Dr. O’Shaughnessy of the Baylor Charles A. Sammons Cancer Center at Dallas.
The difference between the two iniparib trials could not be explained by disease-free interval, although this was not assessed in the phase II trial, she said.
It’s possible that the phase III trial was enriched with patients who had BRCA1/2 genetic mutations, as the phase II results caused what was described by one attendee as "hysteria" among BRCA1/2 carriers to get on the trial. Testing for the genetic mutation was voluntary and was completed by only a minority of patients.
Part of the problem could lie in the heterogeneous nature of triple-negative breast cancer. TNBC is made up of a diverse group of molecular subtypes, which makes it hard to classify and thus target with biologic agents. Indeed, ongoing tumor subtyping identified several molecular subtypes in the phase III trial, including luminal A, luminal B, and basal-like subtypes, Dr. O’Shaughnessy said.
Invited discussant Dr. Lisa Carey of the cancer center at the University of North Carolina at Chapel Hill said that TNBC has been used as a surrogate for molecular subtypes of biological interest, and that the triple-negative phenotype alone is unlikely to be an adequate selection strategy for PARP inhibition.
"Whenever we use triple-negative eligibility to get at a subtype that’s defined biologically, we will always be misclassifying some of these tumors," she said.
TNBC is mostly made up of the basal-like subtype, which is also present in about 80% of BRCA1-associated breast cancer. The shared phenotypic pattern between sporadic basal-like breast cancer and BRCA1-associated cancer – sometimes referred to as "BRCAness" – has therapeutic implications because one of its hallmarks is sensitivity to DNA damage, Dr. Carey explained. PARP inhibitors work by inhibiting DNA-damage repair in BRCA-associated tumors.
Although iniparib has long been included in lists of PARP inhibitors, there is some question about its mechanism of action, she said. Iniparib has substantially lower (1,000-fold) PARP inhibitory activity than do other agents in this class; it does not have additive toxicity as observed in multiple PARP trials; and recent preclinical studies show that although it induces gamma-H2AX foci (a pharmacodynamic readout of DNA damage), it does not appear to do so via PARP 1 and 2 inhibition.
"The role (and frankly the mechanism) of iniparib is still a work in progress, but you can also say that clinical activity based on this study is certainly far less than was predicted," Dr. Carey said, adding that a better understanding of the mechanism is needed for rational determination of a target population.
The phase III, open-label trial randomly assigned 519 patients who had received at least two prior cytotoxic regimens for metastatic triple-negative breast cancer to 21-day cycles of gemcitabine (1,000 mg/m2 IV) and carboplatin (area under the curve 2 IV), both on days 1 and 8, or the same chemotherapy doublet plus iniparib (5.6 mg/kg IV) on days 1, 4, 8 and 11.
The overall response rates by independent radiology review were 30% among the 258 chemotherapy patients and 34% among the 261 iniparib patients, Dr. O’Shaughnessy said. The clinical benefit rates were 36% and 41%.
The toxicity of iniparib plus gemcitabine and carboplatin was comparable with that of chemotherapy alone. One death from upper GI hemorrhage was attributed to iniparib therapy. Biomarker analyses are underway to evaluate patient populations that may benefit from iniparib, she said.
Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough and Wyeth.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The addition of iniparib to chemotherapy resulted in insignificant improvements in median progression-free survival (4.1 months vs. 5.1 months; P = .027) and overall survival (11.1 months vs. 11.8 months; P = 0.28).
Data Source: A phase III, prospective, randomized trial in 519 patients with metastatic triple-negative breast cancer.
Disclosures: Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough, and Wyeth.
Iniparib Loses Blockbuster Image in Triple-Negative Breast Cancer
CHICAGO – After having reported striking phase II data that sent both clinicians and patients with triple-negative breast cancer clamoring for iniparib, researchers are puzzling over a puny survival advantage in the phase III trial, and are questioning how the novel drug actually works.
Iniparib (BSI-201) was thought to belong to the class of targeted therapeutics known as PARP (poly [ADP (adenosine diphosphate)–ribose] polymerase) inhibitors, which have shown promise as single agents in BRCA1- and BRCA2-associated breast and ovarian tumors.
Hopes for a targeted therapy in triple-negative breast cancer were raised when iniparib plus gemcitabine and carboplatin posted significant gains over chemotherapy alone in progression-free (hazard ratio, 0.59) and overall survival (HR, 0.57) without significant added toxicity in the metastatic setting in the 123-patient, phase II study (N. Engl. J. Med. 2011;364:205-14).
Adding iniparib to the same chemotherapy doublet in the much larger phase III trial provided an insignificant survival advantage, however, and failed to meet the coprimary end points of median progression-free survival (4.1 months vs. 5.1 months; HR, 0.79; P = .027) and overall survival (11.1 months vs. 11.8 months; HR, 0.88; P = 0.28).
Exploratory analyses suggested a potential efficacy benefit among second- and third-line patients, who accounted for 43% of the study population, lead author Dr. Joyce O’Shaughnessy said at the annual meeting of the American Society of Clinical Oncology.
Among these patients, iniparib plus chemotherapy increased progression-free survival from 2.9 months with chemotherapy alone to 4.2 months (HR, 0.67), and overall survival from 8.1 months to 10.8 months (HR, 0.65). There was no difference in first-line patients in either progression-free (4.6 months vs. 5.6 months; HR, 0.88) or overall survival (12.6 months vs. 12.4 months; HR, 1.1).
Multivariate analyses that adjusted for several prespecified baselines factors and replaced "time since diagnosis of metastatic disease" with "disease-free interval from primary breast cancer surgery to onset of metastatic disease" showed a significant improvement in overall survival (HR. 0.78; P = .05) in the overall population and in second-and third-line patients (HR, 0.71; P = .05), said Dr. O’Shaughnessy of the Baylor Charles A. Sammons Cancer Center at Dallas.
The difference between the two iniparib trials could not be explained by disease-free interval, although this was not assessed in the phase II trial, she said.
It’s possible that the phase III trial was enriched with patients who had BRCA1/2 genetic mutations, as the phase II results caused what was described by one attendee as "hysteria" among BRCA1/2 carriers to get on the trial. Testing for the genetic mutation was voluntary and was completed by only a minority of patients.
Part of the problem could lie in the heterogeneous nature of triple-negative breast cancer. TNBC is made up of a diverse group of molecular subtypes, which makes it hard to classify and thus target with biologic agents. Indeed, ongoing tumor subtyping identified several molecular subtypes in the phase III trial, including luminal A, luminal B, and basal-like subtypes, Dr. O’Shaughnessy said.
Invited discussant Dr. Lisa Carey of the cancer center at the University of North Carolina at Chapel Hill said that TNBC has been used as a surrogate for molecular subtypes of biological interest, and that the triple-negative phenotype alone is unlikely to be an adequate selection strategy for PARP inhibition.
"Whenever we use triple-negative eligibility to get at a subtype that’s defined biologically, we will always be misclassifying some of these tumors," she said.
TNBC is mostly made up of the basal-like subtype, which is also present in about 80% of BRCA1-associated breast cancer. The shared phenotypic pattern between sporadic basal-like breast cancer and BRCA1-associated cancer – sometimes referred to as "BRCAness" – has therapeutic implications because one of its hallmarks is sensitivity to DNA damage, Dr. Carey explained. PARP inhibitors work by inhibiting DNA-damage repair in BRCA-associated tumors.
Although iniparib has long been included in lists of PARP inhibitors, there is some question about its mechanism of action, she said. Iniparib has substantially lower (1,000-fold) PARP inhibitory activity than do other agents in this class; it does not have additive toxicity as observed in multiple PARP trials; and recent preclinical studies show that although it induces gamma-H2AX foci (a pharmacodynamic readout of DNA damage), it does not appear to do so via PARP 1 and 2 inhibition.
"The role (and frankly the mechanism) of iniparib is still a work in progress, but you can also say that clinical activity based on this study is certainly far less than was predicted," Dr. Carey said, adding that a better understanding of the mechanism is needed for rational determination of a target population.
The phase III, open-label trial randomly assigned 519 patients who had received at least two prior cytotoxic regimens for metastatic triple-negative breast cancer to 21-day cycles of gemcitabine (1,000 mg/m2 IV) and carboplatin (area under the curve 2 IV), both on days 1 and 8, or the same chemotherapy doublet plus iniparib (5.6 mg/kg IV) on days 1, 4, 8 and 11.
The overall response rates by independent radiology review were 30% among the 258 chemotherapy patients and 34% among the 261 iniparib patients, Dr. O’Shaughnessy said. The clinical benefit rates were 36% and 41%.
The toxicity of iniparib plus gemcitabine and carboplatin was comparable with that of chemotherapy alone. One death from upper GI hemorrhage was attributed to iniparib therapy. Biomarker analyses are underway to evaluate patient populations that may benefit from iniparib, she said.
Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough and Wyeth.
CHICAGO – After having reported striking phase II data that sent both clinicians and patients with triple-negative breast cancer clamoring for iniparib, researchers are puzzling over a puny survival advantage in the phase III trial, and are questioning how the novel drug actually works.
Iniparib (BSI-201) was thought to belong to the class of targeted therapeutics known as PARP (poly [ADP (adenosine diphosphate)–ribose] polymerase) inhibitors, which have shown promise as single agents in BRCA1- and BRCA2-associated breast and ovarian tumors.
Hopes for a targeted therapy in triple-negative breast cancer were raised when iniparib plus gemcitabine and carboplatin posted significant gains over chemotherapy alone in progression-free (hazard ratio, 0.59) and overall survival (HR, 0.57) without significant added toxicity in the metastatic setting in the 123-patient, phase II study (N. Engl. J. Med. 2011;364:205-14).
Adding iniparib to the same chemotherapy doublet in the much larger phase III trial provided an insignificant survival advantage, however, and failed to meet the coprimary end points of median progression-free survival (4.1 months vs. 5.1 months; HR, 0.79; P = .027) and overall survival (11.1 months vs. 11.8 months; HR, 0.88; P = 0.28).
Exploratory analyses suggested a potential efficacy benefit among second- and third-line patients, who accounted for 43% of the study population, lead author Dr. Joyce O’Shaughnessy said at the annual meeting of the American Society of Clinical Oncology.
Among these patients, iniparib plus chemotherapy increased progression-free survival from 2.9 months with chemotherapy alone to 4.2 months (HR, 0.67), and overall survival from 8.1 months to 10.8 months (HR, 0.65). There was no difference in first-line patients in either progression-free (4.6 months vs. 5.6 months; HR, 0.88) or overall survival (12.6 months vs. 12.4 months; HR, 1.1).
Multivariate analyses that adjusted for several prespecified baselines factors and replaced "time since diagnosis of metastatic disease" with "disease-free interval from primary breast cancer surgery to onset of metastatic disease" showed a significant improvement in overall survival (HR. 0.78; P = .05) in the overall population and in second-and third-line patients (HR, 0.71; P = .05), said Dr. O’Shaughnessy of the Baylor Charles A. Sammons Cancer Center at Dallas.
The difference between the two iniparib trials could not be explained by disease-free interval, although this was not assessed in the phase II trial, she said.
It’s possible that the phase III trial was enriched with patients who had BRCA1/2 genetic mutations, as the phase II results caused what was described by one attendee as "hysteria" among BRCA1/2 carriers to get on the trial. Testing for the genetic mutation was voluntary and was completed by only a minority of patients.
Part of the problem could lie in the heterogeneous nature of triple-negative breast cancer. TNBC is made up of a diverse group of molecular subtypes, which makes it hard to classify and thus target with biologic agents. Indeed, ongoing tumor subtyping identified several molecular subtypes in the phase III trial, including luminal A, luminal B, and basal-like subtypes, Dr. O’Shaughnessy said.
Invited discussant Dr. Lisa Carey of the cancer center at the University of North Carolina at Chapel Hill said that TNBC has been used as a surrogate for molecular subtypes of biological interest, and that the triple-negative phenotype alone is unlikely to be an adequate selection strategy for PARP inhibition.
"Whenever we use triple-negative eligibility to get at a subtype that’s defined biologically, we will always be misclassifying some of these tumors," she said.
TNBC is mostly made up of the basal-like subtype, which is also present in about 80% of BRCA1-associated breast cancer. The shared phenotypic pattern between sporadic basal-like breast cancer and BRCA1-associated cancer – sometimes referred to as "BRCAness" – has therapeutic implications because one of its hallmarks is sensitivity to DNA damage, Dr. Carey explained. PARP inhibitors work by inhibiting DNA-damage repair in BRCA-associated tumors.
Although iniparib has long been included in lists of PARP inhibitors, there is some question about its mechanism of action, she said. Iniparib has substantially lower (1,000-fold) PARP inhibitory activity than do other agents in this class; it does not have additive toxicity as observed in multiple PARP trials; and recent preclinical studies show that although it induces gamma-H2AX foci (a pharmacodynamic readout of DNA damage), it does not appear to do so via PARP 1 and 2 inhibition.
"The role (and frankly the mechanism) of iniparib is still a work in progress, but you can also say that clinical activity based on this study is certainly far less than was predicted," Dr. Carey said, adding that a better understanding of the mechanism is needed for rational determination of a target population.
The phase III, open-label trial randomly assigned 519 patients who had received at least two prior cytotoxic regimens for metastatic triple-negative breast cancer to 21-day cycles of gemcitabine (1,000 mg/m2 IV) and carboplatin (area under the curve 2 IV), both on days 1 and 8, or the same chemotherapy doublet plus iniparib (5.6 mg/kg IV) on days 1, 4, 8 and 11.
The overall response rates by independent radiology review were 30% among the 258 chemotherapy patients and 34% among the 261 iniparib patients, Dr. O’Shaughnessy said. The clinical benefit rates were 36% and 41%.
The toxicity of iniparib plus gemcitabine and carboplatin was comparable with that of chemotherapy alone. One death from upper GI hemorrhage was attributed to iniparib therapy. Biomarker analyses are underway to evaluate patient populations that may benefit from iniparib, she said.
Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough and Wyeth.
CHICAGO – After having reported striking phase II data that sent both clinicians and patients with triple-negative breast cancer clamoring for iniparib, researchers are puzzling over a puny survival advantage in the phase III trial, and are questioning how the novel drug actually works.
Iniparib (BSI-201) was thought to belong to the class of targeted therapeutics known as PARP (poly [ADP (adenosine diphosphate)–ribose] polymerase) inhibitors, which have shown promise as single agents in BRCA1- and BRCA2-associated breast and ovarian tumors.
Hopes for a targeted therapy in triple-negative breast cancer were raised when iniparib plus gemcitabine and carboplatin posted significant gains over chemotherapy alone in progression-free (hazard ratio, 0.59) and overall survival (HR, 0.57) without significant added toxicity in the metastatic setting in the 123-patient, phase II study (N. Engl. J. Med. 2011;364:205-14).
Adding iniparib to the same chemotherapy doublet in the much larger phase III trial provided an insignificant survival advantage, however, and failed to meet the coprimary end points of median progression-free survival (4.1 months vs. 5.1 months; HR, 0.79; P = .027) and overall survival (11.1 months vs. 11.8 months; HR, 0.88; P = 0.28).
Exploratory analyses suggested a potential efficacy benefit among second- and third-line patients, who accounted for 43% of the study population, lead author Dr. Joyce O’Shaughnessy said at the annual meeting of the American Society of Clinical Oncology.
Among these patients, iniparib plus chemotherapy increased progression-free survival from 2.9 months with chemotherapy alone to 4.2 months (HR, 0.67), and overall survival from 8.1 months to 10.8 months (HR, 0.65). There was no difference in first-line patients in either progression-free (4.6 months vs. 5.6 months; HR, 0.88) or overall survival (12.6 months vs. 12.4 months; HR, 1.1).
Multivariate analyses that adjusted for several prespecified baselines factors and replaced "time since diagnosis of metastatic disease" with "disease-free interval from primary breast cancer surgery to onset of metastatic disease" showed a significant improvement in overall survival (HR. 0.78; P = .05) in the overall population and in second-and third-line patients (HR, 0.71; P = .05), said Dr. O’Shaughnessy of the Baylor Charles A. Sammons Cancer Center at Dallas.
The difference between the two iniparib trials could not be explained by disease-free interval, although this was not assessed in the phase II trial, she said.
It’s possible that the phase III trial was enriched with patients who had BRCA1/2 genetic mutations, as the phase II results caused what was described by one attendee as "hysteria" among BRCA1/2 carriers to get on the trial. Testing for the genetic mutation was voluntary and was completed by only a minority of patients.
Part of the problem could lie in the heterogeneous nature of triple-negative breast cancer. TNBC is made up of a diverse group of molecular subtypes, which makes it hard to classify and thus target with biologic agents. Indeed, ongoing tumor subtyping identified several molecular subtypes in the phase III trial, including luminal A, luminal B, and basal-like subtypes, Dr. O’Shaughnessy said.
Invited discussant Dr. Lisa Carey of the cancer center at the University of North Carolina at Chapel Hill said that TNBC has been used as a surrogate for molecular subtypes of biological interest, and that the triple-negative phenotype alone is unlikely to be an adequate selection strategy for PARP inhibition.
"Whenever we use triple-negative eligibility to get at a subtype that’s defined biologically, we will always be misclassifying some of these tumors," she said.
TNBC is mostly made up of the basal-like subtype, which is also present in about 80% of BRCA1-associated breast cancer. The shared phenotypic pattern between sporadic basal-like breast cancer and BRCA1-associated cancer – sometimes referred to as "BRCAness" – has therapeutic implications because one of its hallmarks is sensitivity to DNA damage, Dr. Carey explained. PARP inhibitors work by inhibiting DNA-damage repair in BRCA-associated tumors.
Although iniparib has long been included in lists of PARP inhibitors, there is some question about its mechanism of action, she said. Iniparib has substantially lower (1,000-fold) PARP inhibitory activity than do other agents in this class; it does not have additive toxicity as observed in multiple PARP trials; and recent preclinical studies show that although it induces gamma-H2AX foci (a pharmacodynamic readout of DNA damage), it does not appear to do so via PARP 1 and 2 inhibition.
"The role (and frankly the mechanism) of iniparib is still a work in progress, but you can also say that clinical activity based on this study is certainly far less than was predicted," Dr. Carey said, adding that a better understanding of the mechanism is needed for rational determination of a target population.
The phase III, open-label trial randomly assigned 519 patients who had received at least two prior cytotoxic regimens for metastatic triple-negative breast cancer to 21-day cycles of gemcitabine (1,000 mg/m2 IV) and carboplatin (area under the curve 2 IV), both on days 1 and 8, or the same chemotherapy doublet plus iniparib (5.6 mg/kg IV) on days 1, 4, 8 and 11.
The overall response rates by independent radiology review were 30% among the 258 chemotherapy patients and 34% among the 261 iniparib patients, Dr. O’Shaughnessy said. The clinical benefit rates were 36% and 41%.
The toxicity of iniparib plus gemcitabine and carboplatin was comparable with that of chemotherapy alone. One death from upper GI hemorrhage was attributed to iniparib therapy. Biomarker analyses are underway to evaluate patient populations that may benefit from iniparib, she said.
Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough and Wyeth.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY OF CLINICAL ONCOLOGY
Major Finding: The addition of iniparib to chemotherapy resulted in insignificant improvements in median progression-free survival (4.1 months vs. 5.1 months; P = .027) and overall survival (11.1 months vs. 11.8 months; P = 0.28).
Data Source: A phase III, prospective, randomized trial in 519 patients with metastatic triple-negative breast cancer.
Disclosures: Sanofi-Aventis sponsored the trial in collaboration with BiPar Sciences. Dr. O’Shaughnessy disclosed honoraria from Sanofi-Aventis, which provided editorial support for the trial. A coauthor reported consulting/advising for Sanofi-Aventis. Dr. Carey disclosed consulting or advising for Sanofi-Aventis, Genentech, Novartis, GlaxoSmithKline, Pfizer, Schering-Plough, and Wyeth.