User login
Black Dots on the Scalp of a Child
Black Dots on the Scalp of a Child
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
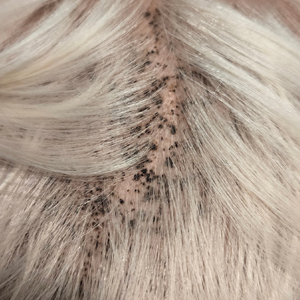
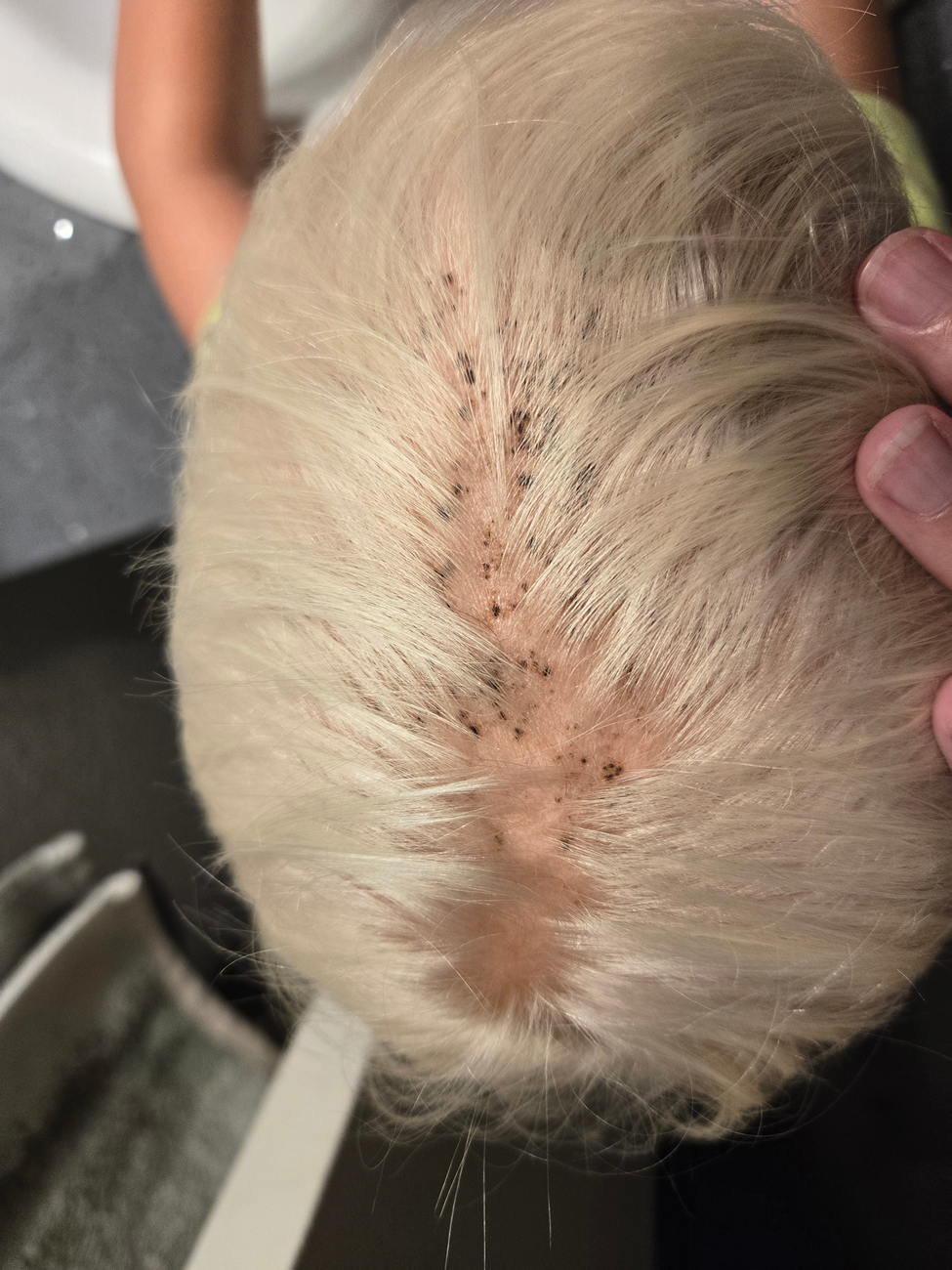
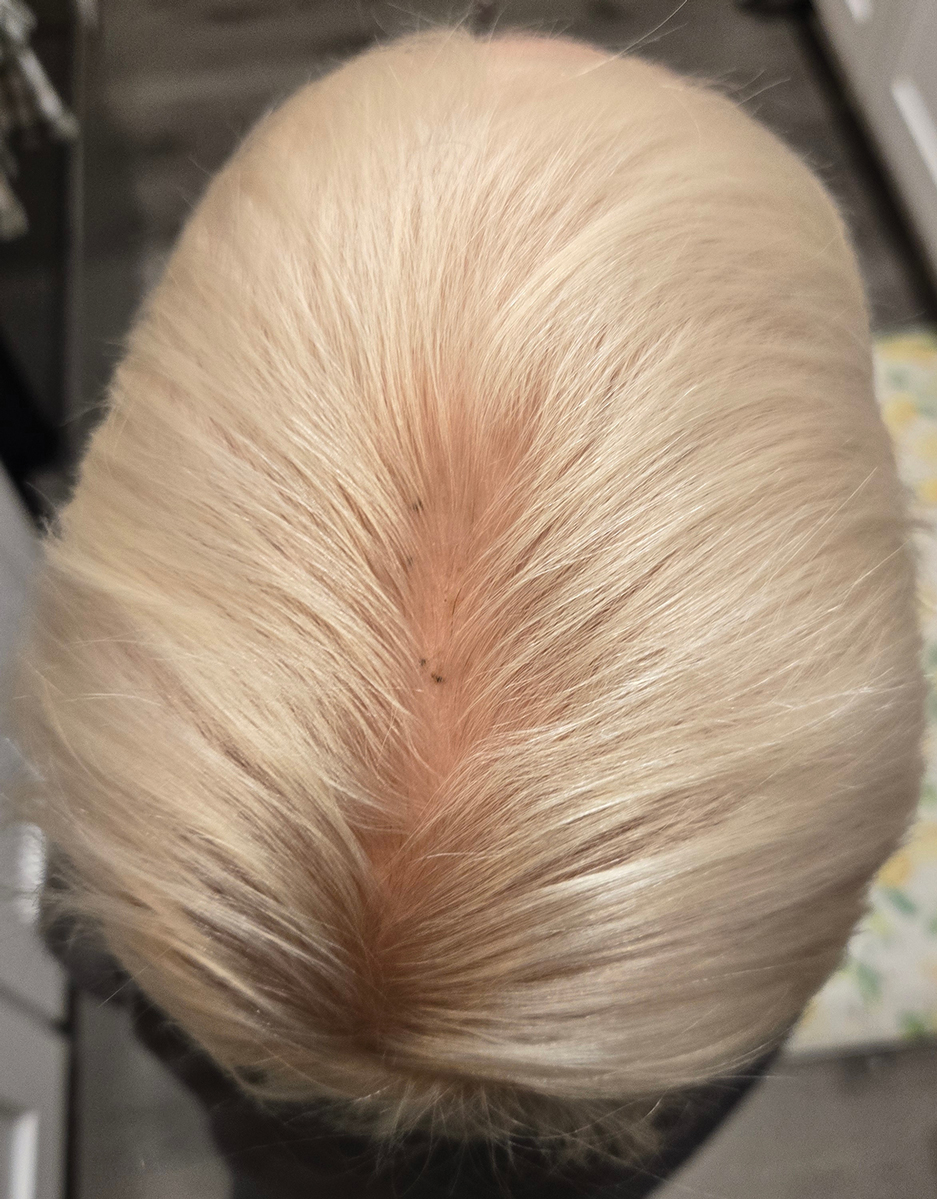
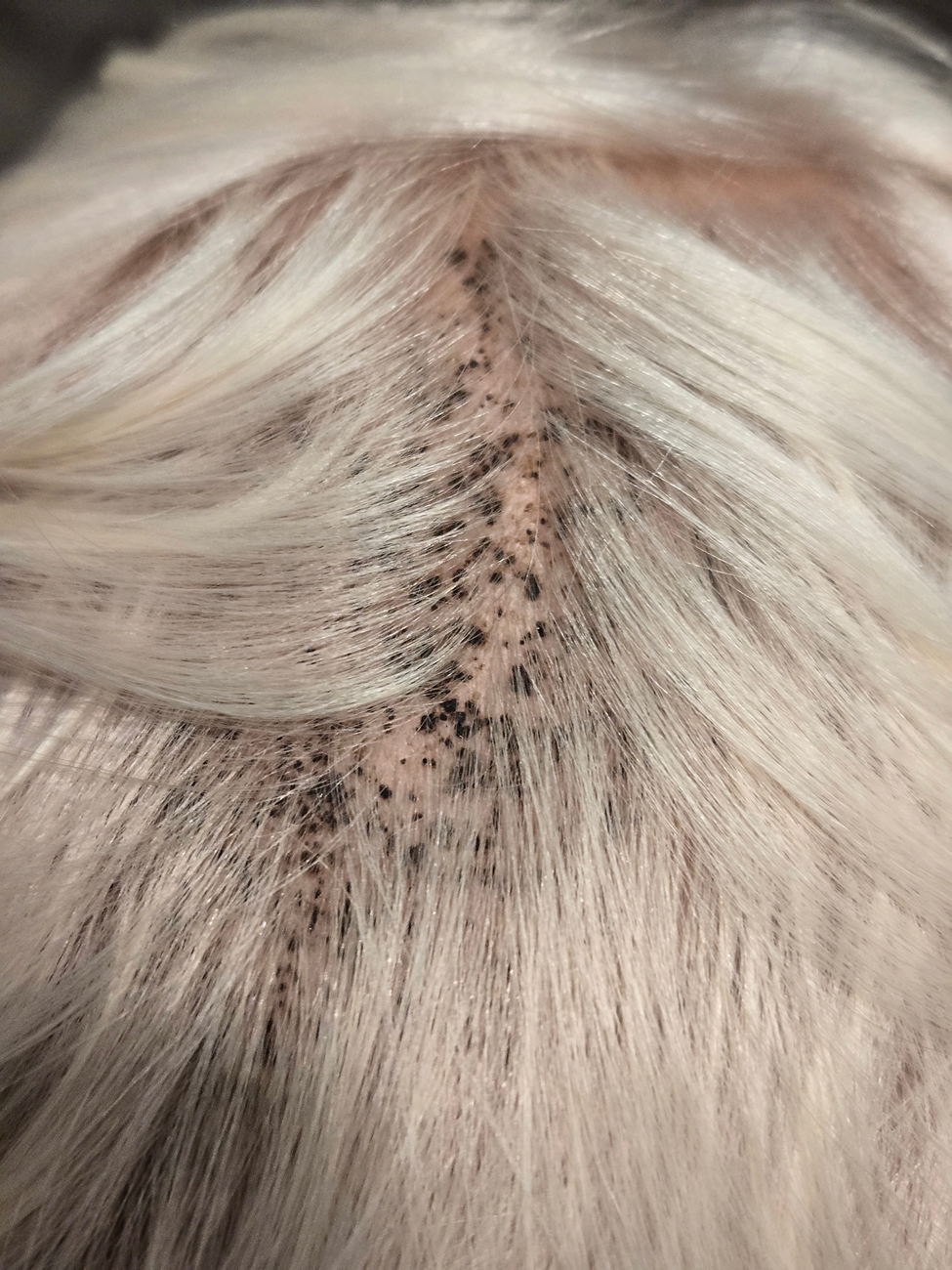
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).
Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).


Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).


Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
Black Dots on the Scalp of a Child
Black Dots on the Scalp of a Child
A 4-year-old boy was referred to the dermatology clinic by his pediatrician for evaluation of persistent black spots on the scalp of 1 month’s duration. The patient was otherwise healthy, and his mother stated that the lesions had appeared gradually, were not tender or pruritic, and did not wash off with shampoo and scrubbing. The patient had no history of any systemic illness, recent travel, genetic disorders, or genodermatoses. Physical examination revealed multiple well-circumscribed, 1- to 2-mm black papules and macules with confluence scattered over the vertex scalp. No erythema, scale, or induration was noted.
Over-the-counter Topical Musculoskeletal Pain Relievers Used With a Heat Source: A Dangerous Combination
To the Editor:
The combination of menthol and methyl salicylate found in a variety of over-the-counter (OTC) creams in conjunction with a heat source such as a heating pad used for musculoskeletal symptoms can be a dire combination due to increased systemic absorption with associated toxicity and localized effects ranging from contact dermatitis or irritation to burn or necrosis.1-6 We present a case of localized burn due a combination of topical methyl salicylate and heating pad use. We also discuss 2 commonly encountered side effects in the literature—localized burns and systemic toxicity associated with percutaneous absorption—and provide specific considerations related to the geriatric and pediatric populations.
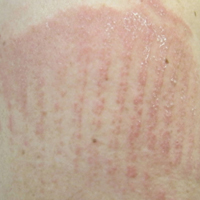
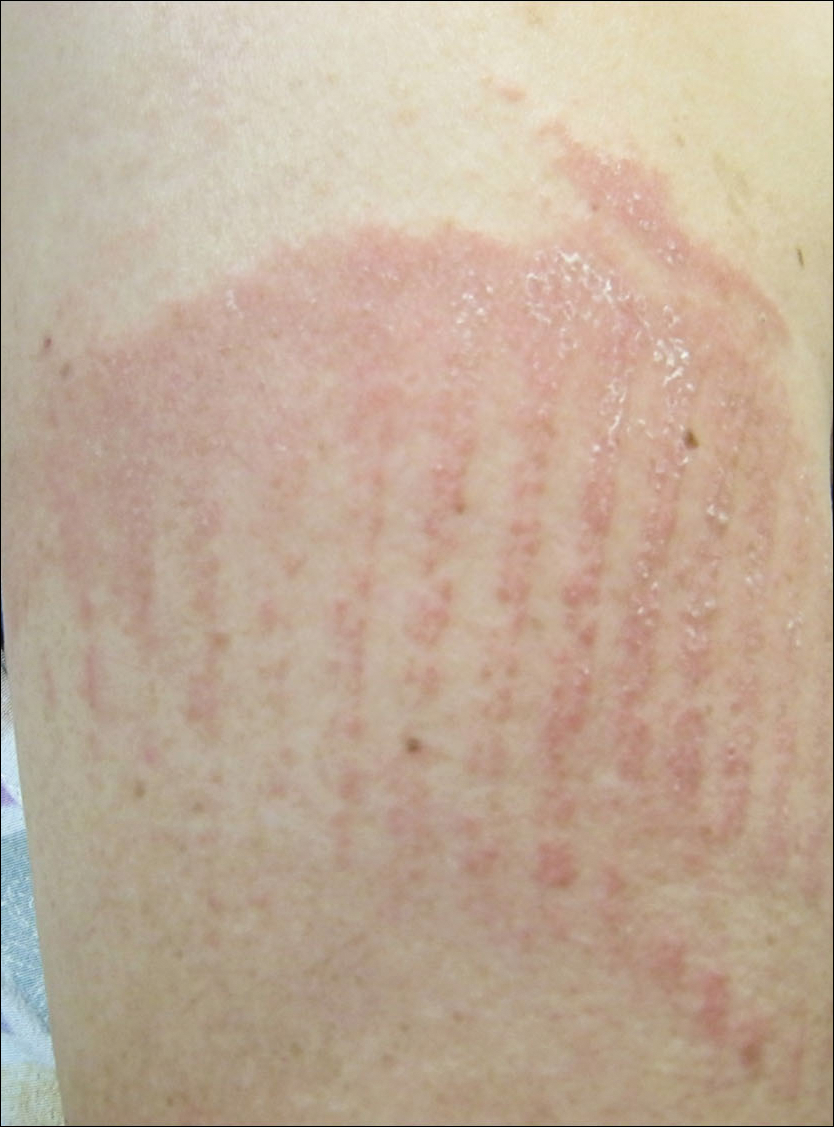
A 62-year-old woman with a history of eczematous dermatitis and osteoarthritis with pain of the left shoulder presented to the dermatology clinic with painful skin-related changes on the left arm of 1 week’s duration. She was prescribed acetaminophen and ibuprofen. However, she self-medicated the left shoulder pain with 2 OTC products containing topical menthol and/or methyl salicylate in combination with a heating pad and likely fell asleep with this combination therapy applied. She noticed the burn the next morning. On examination, the left arm exhibited a geometric, irregularly shaped, erythematous, scaly plaque with a sharp transverse linear demarcation proximally and numerous erythematous linear scaly plaques oriented in an axial orientation with less-defined borders distally (Figure). The patient was diagnosed with burn secondary to combination of topical methyl salicylate and heating pad use. The patient was advised to discontinue the topical medication and to use caution with the heating pad in the future. She was prescribed pramoxine-hydrocortisone lotion to be applied to the affected area twice daily up to 5 days weekly until resolution. Subsequent evaluations revealed progressive improvement with only mild postinflammatory hyperpigmentation noted at 6 months after the burn.
The US Food and Drug Administration (FDA) released statements in 2012 regarding concern for burns related to use of OTC musculoskeletal pain relievers, with 43 cases of burns reported due to methyl salicylate and menthol from 2004 to 2010. Most of the second- and third-degree burns occurred following topical applications of products containing either menthol monotherapy or a combination of methyl salicylate and menthol.1,2 In 2006, the FDA had already ordered 5 firms to stop compounding topical pain relief formulations containing these ingredients, with concerns that it puts patients at increased risk because the compounded formulations had not received FDA approval.3 Despite package warnings, patients may not be aware of the concerning side effects and risks associated with use of OTC creams, especially in combination with occlusion or heating pad use. Our case highlights the importance of ongoing patient education and physician counseling when encountering patients with arthritis or musculoskeletal pain who may often try various OTC self-treatments for pain relief.7
In 2012, the FDA reports stated that the cases of mild to serious burns were associated with methyl salicylate and menthol usage, in some cases 24 hours after first usage. Typically, these effects occur when concentrations are more than either 3% menthol alone or a combination of more than 3% menthol and more than 10% methyl salicylate.1,2 In our case, the patient had been using 2 different OTC products that may have contained as much as 11% menthol and/or 30% methyl salicylate. Electronic resources are available that disclose safety instructions including not to occlude the site, not to use on wounds, and not to be used in conjunction with a heating pad.8,9 Skin breakdown and vasodilation are more likely to occur in a setting of heat and occlusion, which allows for more absorption and localized side effects.4,10 Localized reactions may range from contact dermatitis4 to muscle necrosis.5
The most noteworthy case of localized destruction described a 62-year-old man who had applied topical methyl salicylate and menthol to the forearms, calves, and thighs, then intermittently used a heating pad for 15 to 20 minutes (total duration).5 He subsequently developed erythema and numerous 7.62- to 10.16-cm bullae, which was thought to be consistent with contact dermatitis. Three days later, he was found to have full-thickness cutaneous, fascial, and muscle necrosis in a linear pattern. He was hospitalized for approximately 1 year and treated with extensive debridement and a skin graft. His serum creatinine level increased from 0.7 mg per 100 mL to 2.7 mg per 100 mL (reference range, 0.6–1.2 mg/dL) with evidence of toxic nephrosis and persistent interstitial nephritis, demonstrating the severity of localized destruction that may result when combining these products with direct heat and potential subsequent systemic consequences of this combination.5
The systemic absorption of OTC formulations also has been studied. Morra et al10 studied 12 volunteers (6 women, 6 men) who applied either 5 g of methyl salicylate ointment 12.5% twice daily for 4 days to an area on the thigh (approximately equal to 567 mg salicylate) or trolamine cream 10% twice for 1 day. The participants underwent a break for 7 days and then switched to the alternate treatment. They found that 0.31 to 0.91 mg/L methyl salicylate was detected in the serum 1 hour after applying the ointment consisting of methyl salicylate, and 2 to 6 mg/L methyl salicylate was detected on day 4. Therapeutic serum salicylate levels are 150 to 300 mg/L. They found that approximately 22% of the methyl salicylate also was found in urine samples on day 4. Although these figures may appear small, this study was prompted when a 62-year-old man presented to the emergency department with symptoms of salicylate toxicity and a serum concentration of 518 mg/L from twice-daily use of an OTC formulation containing methyl salicylate over the course of multiple weeks.10 Additionally, those who have aspirin hypersensitivity should be cautious when using such products due to the risk for reported angioedema.4
Providers must exercise extreme caution while caring for geriatric patients, especially if patients are taking warfarin. The combined effects of warfarin and methyl salicylate have previously caused cutaneous purpura, gastrointestinal bleeding, and elevated international normalized ratio values.4,10 Older individuals also have increased skin fragility, allowing microtraumatic insult to easily develop. This fragility, along with an overall decreased intactness of the skin barrier, may lead to increased skin absorption. Furthermore, the addition of applying any heat source places the geriatric patient at greater risk for adverse events.10
In considering the limits of age, the pediatric population also has been studied regarding salicylate toxicity. Most commonly, oral ingestion has caused fatalities, as oil of wintergreen has been cited as extremely dangerous for children if swallowed; doses as small as a teaspoon (5 mL: 7000 mg salicylate) have resulted in fatalities.4,6 Although the consumption of a large amount of a cream- or ointment-based product is unlikely due to the consistency of the medication,6 the thought does merit consideration in the inquisitive toddler age group. For a 15-kg toddler, 150 mg/kg of aspirin or 2250 mg of aspirin, is considered the toxic level, which upon conversion to methyl salicylate levels using a 1.4 factor equates to 1607 mg of methyl salicylate to reach toxicity.6 If using a product with methyl salicylate 30% composition, 1 g of the product contains 300 mg of methyl salicylate; therefore if the toddler consumed approximately 5.3 g of the product (1607 mg methyl salicylate [toxic level] divided by 300 mg methyl salicylate per 1 g of product), he/she would reach toxic levels.6,11 To put this into perspective, a 2-oz tube contains 57 g (approximately 10 times the toxic dose) of the product.8 Thus, although there is less concern overall for consumption of cream- or ointment-based methyl salicylate, there still is potential for harm if a small child were to ingest such a product containing higher percentages of methyl salicylate.6
There also have been reports of pediatric toxicity related to percutaneous absorption, even leading to pediatric fatality.4,6 In particular, there was a case of a young boy hospitalized with ichthyosis who received escalating doses of percutaneous salicylate, which resulted in toxicity; when therapy was discontinued, he experienced full recovery.12 In 2007, a 17-year-old adolescent girl died from methyl salicylate toxicity after numerous applications of salicylate-containing products in conjunction with medicated pads.7
Although the FDA has drawn attention and encouraged caution with use of OTC topical musculoskeletal pain relievers, the importance of ensuring patients are fully aware of potential burns, permanent skin or muscle damage, and even death if used inappropriately cannot be overstated. The FDA consumer health information website has 2 patient-directed handouts2,3 that may be useful to post in patient waiting areas to increase overall understanding of the risks associated with OTC products containing methyl salicylate and menthol ingredients. Fortunately, our patient suffered only mild postinflammatory hyperpigmentation without substantial sustained consequences.
- US Food and Drug Administration. FDA Drug Safety Communication: rare cases of serious burns with the use of over-the-counter topical muscle and joint pain relievers. http://www.fda.gov/Drugs/DrugSafety/ucm318858.htm. Published September 13, 2012. Updated February 11, 2016. Accessed October 31, 2017.
- US Food and Drug Administration. Topical pain relievers may cause burns. http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm318674.htm. Published September 13, 2012. Updated November 5, 2015. Accessed October 31, 2017.
- US Food and Drug Administration. Use caution with over-the-counter creams, ointments. http://www.fda.gov/forconsumers/consumerupdates/ucm049367.htm. Updated October 17, 2017. Accessed October 31, 2017.
- Chan TY. Potential dangers from topical preparations containing methyl salicylate. Hum Exp Toxicol. 1996;15:747-750.
- Heng MC. Local necrosis and interstitial nephritis due to topical methyl salicylate and menthol. Cutis. 1987;39:442-444.
- Davis JE. Are one or two dangerous? methyl salicylate exposure in toddlers. J Emerg Med. 2007;32:63-69.
- Associated Press. Sports cream warnings urged after teen’s death: track star’s overdose points to risks of popular muscle salve. NBC News. http://www.nbcnews.com/id/19208195. Updated June 13, 2007. Accessed October 31, 2017.
- Ultra Strength Bengay Cream. Bengay website. http://www.bengay.com/bengay-ultra-strength-cream. Accessed November 1, 2017.
- Tiger Balm Arthritis Rub. Tiger Balm website. http://www.tigerbalm.com/us/pages/tb_product?product_id=6. Accessed November 1, 2017.
- Morra P, Bartle WR, Walker SE, et al. Serum concentrations of salicylic acid following topically applied salicylate derivatives. Ann Pharmacother. 1996;9:935-940.
- US National Library of Medicine. Bengay Ultra Strength non greasy pain relieving- camphor (synthetic), menthol, and methyl salicylate cream. Daily Med website. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=5aa265f8-ab45-47b2-b5ab-d4df54daed01. Updated November 3, 2016. Accessed November 1, 2017.
- Aspinall JB, Goel KM. Salicylate poisoning in dermatological therapy. Br Med J. 1978;2:1373.
To the Editor:
The combination of menthol and methyl salicylate found in a variety of over-the-counter (OTC) creams in conjunction with a heat source such as a heating pad used for musculoskeletal symptoms can be a dire combination due to increased systemic absorption with associated toxicity and localized effects ranging from contact dermatitis or irritation to burn or necrosis.1-6 We present a case of localized burn due a combination of topical methyl salicylate and heating pad use. We also discuss 2 commonly encountered side effects in the literature—localized burns and systemic toxicity associated with percutaneous absorption—and provide specific considerations related to the geriatric and pediatric populations.
A 62-year-old woman with a history of eczematous dermatitis and osteoarthritis with pain of the left shoulder presented to the dermatology clinic with painful skin-related changes on the left arm of 1 week’s duration. She was prescribed acetaminophen and ibuprofen. However, she self-medicated the left shoulder pain with 2 OTC products containing topical menthol and/or methyl salicylate in combination with a heating pad and likely fell asleep with this combination therapy applied. She noticed the burn the next morning. On examination, the left arm exhibited a geometric, irregularly shaped, erythematous, scaly plaque with a sharp transverse linear demarcation proximally and numerous erythematous linear scaly plaques oriented in an axial orientation with less-defined borders distally (Figure). The patient was diagnosed with burn secondary to combination of topical methyl salicylate and heating pad use. The patient was advised to discontinue the topical medication and to use caution with the heating pad in the future. She was prescribed pramoxine-hydrocortisone lotion to be applied to the affected area twice daily up to 5 days weekly until resolution. Subsequent evaluations revealed progressive improvement with only mild postinflammatory hyperpigmentation noted at 6 months after the burn.

The US Food and Drug Administration (FDA) released statements in 2012 regarding concern for burns related to use of OTC musculoskeletal pain relievers, with 43 cases of burns reported due to methyl salicylate and menthol from 2004 to 2010. Most of the second- and third-degree burns occurred following topical applications of products containing either menthol monotherapy or a combination of methyl salicylate and menthol.1,2 In 2006, the FDA had already ordered 5 firms to stop compounding topical pain relief formulations containing these ingredients, with concerns that it puts patients at increased risk because the compounded formulations had not received FDA approval.3 Despite package warnings, patients may not be aware of the concerning side effects and risks associated with use of OTC creams, especially in combination with occlusion or heating pad use. Our case highlights the importance of ongoing patient education and physician counseling when encountering patients with arthritis or musculoskeletal pain who may often try various OTC self-treatments for pain relief.7
In 2012, the FDA reports stated that the cases of mild to serious burns were associated with methyl salicylate and menthol usage, in some cases 24 hours after first usage. Typically, these effects occur when concentrations are more than either 3% menthol alone or a combination of more than 3% menthol and more than 10% methyl salicylate.1,2 In our case, the patient had been using 2 different OTC products that may have contained as much as 11% menthol and/or 30% methyl salicylate. Electronic resources are available that disclose safety instructions including not to occlude the site, not to use on wounds, and not to be used in conjunction with a heating pad.8,9 Skin breakdown and vasodilation are more likely to occur in a setting of heat and occlusion, which allows for more absorption and localized side effects.4,10 Localized reactions may range from contact dermatitis4 to muscle necrosis.5
The most noteworthy case of localized destruction described a 62-year-old man who had applied topical methyl salicylate and menthol to the forearms, calves, and thighs, then intermittently used a heating pad for 15 to 20 minutes (total duration).5 He subsequently developed erythema and numerous 7.62- to 10.16-cm bullae, which was thought to be consistent with contact dermatitis. Three days later, he was found to have full-thickness cutaneous, fascial, and muscle necrosis in a linear pattern. He was hospitalized for approximately 1 year and treated with extensive debridement and a skin graft. His serum creatinine level increased from 0.7 mg per 100 mL to 2.7 mg per 100 mL (reference range, 0.6–1.2 mg/dL) with evidence of toxic nephrosis and persistent interstitial nephritis, demonstrating the severity of localized destruction that may result when combining these products with direct heat and potential subsequent systemic consequences of this combination.5
The systemic absorption of OTC formulations also has been studied. Morra et al10 studied 12 volunteers (6 women, 6 men) who applied either 5 g of methyl salicylate ointment 12.5% twice daily for 4 days to an area on the thigh (approximately equal to 567 mg salicylate) or trolamine cream 10% twice for 1 day. The participants underwent a break for 7 days and then switched to the alternate treatment. They found that 0.31 to 0.91 mg/L methyl salicylate was detected in the serum 1 hour after applying the ointment consisting of methyl salicylate, and 2 to 6 mg/L methyl salicylate was detected on day 4. Therapeutic serum salicylate levels are 150 to 300 mg/L. They found that approximately 22% of the methyl salicylate also was found in urine samples on day 4. Although these figures may appear small, this study was prompted when a 62-year-old man presented to the emergency department with symptoms of salicylate toxicity and a serum concentration of 518 mg/L from twice-daily use of an OTC formulation containing methyl salicylate over the course of multiple weeks.10 Additionally, those who have aspirin hypersensitivity should be cautious when using such products due to the risk for reported angioedema.4
Providers must exercise extreme caution while caring for geriatric patients, especially if patients are taking warfarin. The combined effects of warfarin and methyl salicylate have previously caused cutaneous purpura, gastrointestinal bleeding, and elevated international normalized ratio values.4,10 Older individuals also have increased skin fragility, allowing microtraumatic insult to easily develop. This fragility, along with an overall decreased intactness of the skin barrier, may lead to increased skin absorption. Furthermore, the addition of applying any heat source places the geriatric patient at greater risk for adverse events.10
In considering the limits of age, the pediatric population also has been studied regarding salicylate toxicity. Most commonly, oral ingestion has caused fatalities, as oil of wintergreen has been cited as extremely dangerous for children if swallowed; doses as small as a teaspoon (5 mL: 7000 mg salicylate) have resulted in fatalities.4,6 Although the consumption of a large amount of a cream- or ointment-based product is unlikely due to the consistency of the medication,6 the thought does merit consideration in the inquisitive toddler age group. For a 15-kg toddler, 150 mg/kg of aspirin or 2250 mg of aspirin, is considered the toxic level, which upon conversion to methyl salicylate levels using a 1.4 factor equates to 1607 mg of methyl salicylate to reach toxicity.6 If using a product with methyl salicylate 30% composition, 1 g of the product contains 300 mg of methyl salicylate; therefore if the toddler consumed approximately 5.3 g of the product (1607 mg methyl salicylate [toxic level] divided by 300 mg methyl salicylate per 1 g of product), he/she would reach toxic levels.6,11 To put this into perspective, a 2-oz tube contains 57 g (approximately 10 times the toxic dose) of the product.8 Thus, although there is less concern overall for consumption of cream- or ointment-based methyl salicylate, there still is potential for harm if a small child were to ingest such a product containing higher percentages of methyl salicylate.6
There also have been reports of pediatric toxicity related to percutaneous absorption, even leading to pediatric fatality.4,6 In particular, there was a case of a young boy hospitalized with ichthyosis who received escalating doses of percutaneous salicylate, which resulted in toxicity; when therapy was discontinued, he experienced full recovery.12 In 2007, a 17-year-old adolescent girl died from methyl salicylate toxicity after numerous applications of salicylate-containing products in conjunction with medicated pads.7
Although the FDA has drawn attention and encouraged caution with use of OTC topical musculoskeletal pain relievers, the importance of ensuring patients are fully aware of potential burns, permanent skin or muscle damage, and even death if used inappropriately cannot be overstated. The FDA consumer health information website has 2 patient-directed handouts2,3 that may be useful to post in patient waiting areas to increase overall understanding of the risks associated with OTC products containing methyl salicylate and menthol ingredients. Fortunately, our patient suffered only mild postinflammatory hyperpigmentation without substantial sustained consequences.
To the Editor:
The combination of menthol and methyl salicylate found in a variety of over-the-counter (OTC) creams in conjunction with a heat source such as a heating pad used for musculoskeletal symptoms can be a dire combination due to increased systemic absorption with associated toxicity and localized effects ranging from contact dermatitis or irritation to burn or necrosis.1-6 We present a case of localized burn due a combination of topical methyl salicylate and heating pad use. We also discuss 2 commonly encountered side effects in the literature—localized burns and systemic toxicity associated with percutaneous absorption—and provide specific considerations related to the geriatric and pediatric populations.
A 62-year-old woman with a history of eczematous dermatitis and osteoarthritis with pain of the left shoulder presented to the dermatology clinic with painful skin-related changes on the left arm of 1 week’s duration. She was prescribed acetaminophen and ibuprofen. However, she self-medicated the left shoulder pain with 2 OTC products containing topical menthol and/or methyl salicylate in combination with a heating pad and likely fell asleep with this combination therapy applied. She noticed the burn the next morning. On examination, the left arm exhibited a geometric, irregularly shaped, erythematous, scaly plaque with a sharp transverse linear demarcation proximally and numerous erythematous linear scaly plaques oriented in an axial orientation with less-defined borders distally (Figure). The patient was diagnosed with burn secondary to combination of topical methyl salicylate and heating pad use. The patient was advised to discontinue the topical medication and to use caution with the heating pad in the future. She was prescribed pramoxine-hydrocortisone lotion to be applied to the affected area twice daily up to 5 days weekly until resolution. Subsequent evaluations revealed progressive improvement with only mild postinflammatory hyperpigmentation noted at 6 months after the burn.

The US Food and Drug Administration (FDA) released statements in 2012 regarding concern for burns related to use of OTC musculoskeletal pain relievers, with 43 cases of burns reported due to methyl salicylate and menthol from 2004 to 2010. Most of the second- and third-degree burns occurred following topical applications of products containing either menthol monotherapy or a combination of methyl salicylate and menthol.1,2 In 2006, the FDA had already ordered 5 firms to stop compounding topical pain relief formulations containing these ingredients, with concerns that it puts patients at increased risk because the compounded formulations had not received FDA approval.3 Despite package warnings, patients may not be aware of the concerning side effects and risks associated with use of OTC creams, especially in combination with occlusion or heating pad use. Our case highlights the importance of ongoing patient education and physician counseling when encountering patients with arthritis or musculoskeletal pain who may often try various OTC self-treatments for pain relief.7
In 2012, the FDA reports stated that the cases of mild to serious burns were associated with methyl salicylate and menthol usage, in some cases 24 hours after first usage. Typically, these effects occur when concentrations are more than either 3% menthol alone or a combination of more than 3% menthol and more than 10% methyl salicylate.1,2 In our case, the patient had been using 2 different OTC products that may have contained as much as 11% menthol and/or 30% methyl salicylate. Electronic resources are available that disclose safety instructions including not to occlude the site, not to use on wounds, and not to be used in conjunction with a heating pad.8,9 Skin breakdown and vasodilation are more likely to occur in a setting of heat and occlusion, which allows for more absorption and localized side effects.4,10 Localized reactions may range from contact dermatitis4 to muscle necrosis.5
The most noteworthy case of localized destruction described a 62-year-old man who had applied topical methyl salicylate and menthol to the forearms, calves, and thighs, then intermittently used a heating pad for 15 to 20 minutes (total duration).5 He subsequently developed erythema and numerous 7.62- to 10.16-cm bullae, which was thought to be consistent with contact dermatitis. Three days later, he was found to have full-thickness cutaneous, fascial, and muscle necrosis in a linear pattern. He was hospitalized for approximately 1 year and treated with extensive debridement and a skin graft. His serum creatinine level increased from 0.7 mg per 100 mL to 2.7 mg per 100 mL (reference range, 0.6–1.2 mg/dL) with evidence of toxic nephrosis and persistent interstitial nephritis, demonstrating the severity of localized destruction that may result when combining these products with direct heat and potential subsequent systemic consequences of this combination.5
The systemic absorption of OTC formulations also has been studied. Morra et al10 studied 12 volunteers (6 women, 6 men) who applied either 5 g of methyl salicylate ointment 12.5% twice daily for 4 days to an area on the thigh (approximately equal to 567 mg salicylate) or trolamine cream 10% twice for 1 day. The participants underwent a break for 7 days and then switched to the alternate treatment. They found that 0.31 to 0.91 mg/L methyl salicylate was detected in the serum 1 hour after applying the ointment consisting of methyl salicylate, and 2 to 6 mg/L methyl salicylate was detected on day 4. Therapeutic serum salicylate levels are 150 to 300 mg/L. They found that approximately 22% of the methyl salicylate also was found in urine samples on day 4. Although these figures may appear small, this study was prompted when a 62-year-old man presented to the emergency department with symptoms of salicylate toxicity and a serum concentration of 518 mg/L from twice-daily use of an OTC formulation containing methyl salicylate over the course of multiple weeks.10 Additionally, those who have aspirin hypersensitivity should be cautious when using such products due to the risk for reported angioedema.4
Providers must exercise extreme caution while caring for geriatric patients, especially if patients are taking warfarin. The combined effects of warfarin and methyl salicylate have previously caused cutaneous purpura, gastrointestinal bleeding, and elevated international normalized ratio values.4,10 Older individuals also have increased skin fragility, allowing microtraumatic insult to easily develop. This fragility, along with an overall decreased intactness of the skin barrier, may lead to increased skin absorption. Furthermore, the addition of applying any heat source places the geriatric patient at greater risk for adverse events.10
In considering the limits of age, the pediatric population also has been studied regarding salicylate toxicity. Most commonly, oral ingestion has caused fatalities, as oil of wintergreen has been cited as extremely dangerous for children if swallowed; doses as small as a teaspoon (5 mL: 7000 mg salicylate) have resulted in fatalities.4,6 Although the consumption of a large amount of a cream- or ointment-based product is unlikely due to the consistency of the medication,6 the thought does merit consideration in the inquisitive toddler age group. For a 15-kg toddler, 150 mg/kg of aspirin or 2250 mg of aspirin, is considered the toxic level, which upon conversion to methyl salicylate levels using a 1.4 factor equates to 1607 mg of methyl salicylate to reach toxicity.6 If using a product with methyl salicylate 30% composition, 1 g of the product contains 300 mg of methyl salicylate; therefore if the toddler consumed approximately 5.3 g of the product (1607 mg methyl salicylate [toxic level] divided by 300 mg methyl salicylate per 1 g of product), he/she would reach toxic levels.6,11 To put this into perspective, a 2-oz tube contains 57 g (approximately 10 times the toxic dose) of the product.8 Thus, although there is less concern overall for consumption of cream- or ointment-based methyl salicylate, there still is potential for harm if a small child were to ingest such a product containing higher percentages of methyl salicylate.6
There also have been reports of pediatric toxicity related to percutaneous absorption, even leading to pediatric fatality.4,6 In particular, there was a case of a young boy hospitalized with ichthyosis who received escalating doses of percutaneous salicylate, which resulted in toxicity; when therapy was discontinued, he experienced full recovery.12 In 2007, a 17-year-old adolescent girl died from methyl salicylate toxicity after numerous applications of salicylate-containing products in conjunction with medicated pads.7
Although the FDA has drawn attention and encouraged caution with use of OTC topical musculoskeletal pain relievers, the importance of ensuring patients are fully aware of potential burns, permanent skin or muscle damage, and even death if used inappropriately cannot be overstated. The FDA consumer health information website has 2 patient-directed handouts2,3 that may be useful to post in patient waiting areas to increase overall understanding of the risks associated with OTC products containing methyl salicylate and menthol ingredients. Fortunately, our patient suffered only mild postinflammatory hyperpigmentation without substantial sustained consequences.
- US Food and Drug Administration. FDA Drug Safety Communication: rare cases of serious burns with the use of over-the-counter topical muscle and joint pain relievers. http://www.fda.gov/Drugs/DrugSafety/ucm318858.htm. Published September 13, 2012. Updated February 11, 2016. Accessed October 31, 2017.
- US Food and Drug Administration. Topical pain relievers may cause burns. http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm318674.htm. Published September 13, 2012. Updated November 5, 2015. Accessed October 31, 2017.
- US Food and Drug Administration. Use caution with over-the-counter creams, ointments. http://www.fda.gov/forconsumers/consumerupdates/ucm049367.htm. Updated October 17, 2017. Accessed October 31, 2017.
- Chan TY. Potential dangers from topical preparations containing methyl salicylate. Hum Exp Toxicol. 1996;15:747-750.
- Heng MC. Local necrosis and interstitial nephritis due to topical methyl salicylate and menthol. Cutis. 1987;39:442-444.
- Davis JE. Are one or two dangerous? methyl salicylate exposure in toddlers. J Emerg Med. 2007;32:63-69.
- Associated Press. Sports cream warnings urged after teen’s death: track star’s overdose points to risks of popular muscle salve. NBC News. http://www.nbcnews.com/id/19208195. Updated June 13, 2007. Accessed October 31, 2017.
- Ultra Strength Bengay Cream. Bengay website. http://www.bengay.com/bengay-ultra-strength-cream. Accessed November 1, 2017.
- Tiger Balm Arthritis Rub. Tiger Balm website. http://www.tigerbalm.com/us/pages/tb_product?product_id=6. Accessed November 1, 2017.
- Morra P, Bartle WR, Walker SE, et al. Serum concentrations of salicylic acid following topically applied salicylate derivatives. Ann Pharmacother. 1996;9:935-940.
- US National Library of Medicine. Bengay Ultra Strength non greasy pain relieving- camphor (synthetic), menthol, and methyl salicylate cream. Daily Med website. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=5aa265f8-ab45-47b2-b5ab-d4df54daed01. Updated November 3, 2016. Accessed November 1, 2017.
- Aspinall JB, Goel KM. Salicylate poisoning in dermatological therapy. Br Med J. 1978;2:1373.
- US Food and Drug Administration. FDA Drug Safety Communication: rare cases of serious burns with the use of over-the-counter topical muscle and joint pain relievers. http://www.fda.gov/Drugs/DrugSafety/ucm318858.htm. Published September 13, 2012. Updated February 11, 2016. Accessed October 31, 2017.
- US Food and Drug Administration. Topical pain relievers may cause burns. http://www.fda.gov/ForConsumers/ConsumerUpdates/ucm318674.htm. Published September 13, 2012. Updated November 5, 2015. Accessed October 31, 2017.
- US Food and Drug Administration. Use caution with over-the-counter creams, ointments. http://www.fda.gov/forconsumers/consumerupdates/ucm049367.htm. Updated October 17, 2017. Accessed October 31, 2017.
- Chan TY. Potential dangers from topical preparations containing methyl salicylate. Hum Exp Toxicol. 1996;15:747-750.
- Heng MC. Local necrosis and interstitial nephritis due to topical methyl salicylate and menthol. Cutis. 1987;39:442-444.
- Davis JE. Are one or two dangerous? methyl salicylate exposure in toddlers. J Emerg Med. 2007;32:63-69.
- Associated Press. Sports cream warnings urged after teen’s death: track star’s overdose points to risks of popular muscle salve. NBC News. http://www.nbcnews.com/id/19208195. Updated June 13, 2007. Accessed October 31, 2017.
- Ultra Strength Bengay Cream. Bengay website. http://www.bengay.com/bengay-ultra-strength-cream. Accessed November 1, 2017.
- Tiger Balm Arthritis Rub. Tiger Balm website. http://www.tigerbalm.com/us/pages/tb_product?product_id=6. Accessed November 1, 2017.
- Morra P, Bartle WR, Walker SE, et al. Serum concentrations of salicylic acid following topically applied salicylate derivatives. Ann Pharmacother. 1996;9:935-940.
- US National Library of Medicine. Bengay Ultra Strength non greasy pain relieving- camphor (synthetic), menthol, and methyl salicylate cream. Daily Med website. http://dailymed.nlm.nih.gov/dailymed/lookup.cfm?setid=5aa265f8-ab45-47b2-b5ab-d4df54daed01. Updated November 3, 2016. Accessed November 1, 2017.
- Aspinall JB, Goel KM. Salicylate poisoning in dermatological therapy. Br Med J. 1978;2:1373.
Practice Points
- Recognize the potential complication of burn from use of over-the-counter (OTC) musculoskeletal relievers in combination with a heat source.
- Screen for OTC product use as well as device application when evaluating an atypically patterned cutaneous eruption.
- Recognize potential toxicity associated with both topical application and accidental ingestion in the pediatric population.
- Physicians should become familiar with resources available, including patient handouts that describe risks associated with use of OTC musculoskeletal relievers containing methyl salicylate and menthol ingredients.