User login
Microwave Device Offers Long-Term Treatment for Hyperhidrosis
GRAPEVINE, TEX. – A novel microwave device significantly reduced underarm sweating in two studies involving a total of 151 patients with axillary hyperhidrosis.
The condition affects millions of people, yet current treatments are limited by either duration of effect or efficacy. The microwave device has the potential for a longer-term and possibly even permanent effect via eradication of eccrine sweat glands, Dr. Suzanne L. Kilmer and Dr. Mark Lupin reported in separate presentations at the annual meeting of the American Society for Laser Medicine and Surgery.
Miramar Labs' miraDry system focuses microwave energy to selectively heat the interface between the skin and underlying fat, where the sweat glands reside. The system comprises a console, handpiece, and disposable tip. The in-office procedure takes approximately 40 minutes. The system was cleared for licensure in the United States by the Food and Drug Administration in January 2011, and will be marketed later this year, according to a company spokesperson.
Dr. Kilmer, of the Laser and Skin Surgery Center of Northern California, Sacramento, presented data on a second-generation version of the device that was FDA approved but never marketed. In a multicenter, sham-controlled study, 120 adults with hyperhidrosis were randomized to miraDry (81) or sham treatment (39) for one to three sessions. The patients had to have a Hyperhidrosis Disease Severity Scale (HDSS) score of 3 or 4 ("my underarm sweating is barely tolerable and frequently/always interferes with my daily activities") at baseline, and gravimetric readings greater than 50 mg at each axilla (in 5 minutes).
Follow-up was 6 months for the sham group and 12 months for the treatment group. Responders were defined as subjects reporting a reduction to an HDSS score of 1 ("My underarm sweating is never noticeable and never interferes with daily activities") or 2 ("My underarm sweating is tolerable but sometimes interferes with daily activities"). The patients had a mean age of 33 years, 58% were female, and 84% were white.
Efficacy for the treatment group was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant, Dr. Kilmer reported.
Treatment-related adverse events were generally mild, and all but one resolved. The most common adverse events in the treatment group were transient patches of altered sensation in the treatment limb, occurring in 8 patients (10%), and axillary pain requiring prescription medication in 5 patients (6%). Most subjects experienced transient post-treatment local sequelae in the axilla such as edema, tenderness, and bruising.
Follow-up of the treated subjects showed stable efficacy through 12 months, she said.
The second study, involving 31 patients, investigated the third-generation, optimized version of the device. As in the earlier study, all patients had to have primary axillary hyperhidrosis with an HDSS score of 3 or 4. The patients also were required to have a gravimetric sweat assessment of at least 50 mg in each axilla (in 5 minutes). None of the patients had surgery for axillary hyperhidrosis or botulinum toxin injections in the axillae in the prior 12 months, said Dr. Lupin of Cosmedica Laser Centre, Victoria, B.C.
The patients had a mean age of 33 years (range 18-65 years), and three-quarters were female. They had a mean BMI of 24.8 kg/m2. Of 28 patients seen at 30 days, all but one (96%) had HDSS scores of 1 or 2. Of 25 seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy). Gravimetric assessments 3 months after treatment showed that 94% of patients had at least a 50% reduction in axillary sweat compared with baseline, with an average sweat reduction of 82%.
Nineteen of the 31 initially enrolled patients (61%) experienced at least one treatment-related adverse event, of which most (88%) were rated as mild. The most common were numbness or tingling in an area of the treated limb (12 patients, 39%), and edema in the chest or treatment limb lasting about a week (9 patients, 29%). Most of the patients also experienced acute post-treatment transient effects in the treatment area such as localized edema, tenderness, or erythema. Follow-up of safety is ongoing, Dr. Lupin noted.
"The study is continuing out to 12 months, and at 6 months the preliminary data so far are showing sustained positive improvements in all measures of quality of life, reduction of sweat, and impact on daily living," Dr. Lupin said in an interview. Many patients also reported reduction of axillary hair, and a few patients noted improvement in odor, he said.
Overall, patient satisfaction, as measured by the Dermatology Life Quality Index (DLQI), was 96% at 3 months.
When Dr. Lupin was asked whether miraDry had any disadvantages compared with botulinum toxin type A, he said that there are several short-term advantages to botulinum toxin type A: It is a quicker and easier procedure (about 5-10 minutes) with sweat reduction occurring in just a few days, versus 1 hour for the miraDry procedure, which requires anesthesia and can take a week or longer to produce results. Moreover, botulinum toxin type A is a single treatment and its benefit lasts about 6-8 months, whereas miraDry takes 1-3 sessions for a benefit of at least a year and possibly longer.
Indeed, Dr. Kilmer said in an interview, duration of benefit is an advantage of miraDry over just about every current hyperhidrosis treatment other than sympathectomy, a procedure that is rarely done. So far, the longest miraDry has been studied is 12 months, so "we can’t say for sure, but it didn’t drop off much during that time, so we expect it will last much longer," she said.
Dr. Mathew Avram, director of the Massachusetts General Hospital Dermatology Laser and Cosmetic Center, Boston, said, "This is innovative and interesting technology to address an issue that is very problematic for patients. We need to learn a little more about the duration of these benefits and what if any side effects may be created with repeated treatments over time."
The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
GRAPEVINE, TEX. – A novel microwave device significantly reduced underarm sweating in two studies involving a total of 151 patients with axillary hyperhidrosis.
The condition affects millions of people, yet current treatments are limited by either duration of effect or efficacy. The microwave device has the potential for a longer-term and possibly even permanent effect via eradication of eccrine sweat glands, Dr. Suzanne L. Kilmer and Dr. Mark Lupin reported in separate presentations at the annual meeting of the American Society for Laser Medicine and Surgery.
Miramar Labs' miraDry system focuses microwave energy to selectively heat the interface between the skin and underlying fat, where the sweat glands reside. The system comprises a console, handpiece, and disposable tip. The in-office procedure takes approximately 40 minutes. The system was cleared for licensure in the United States by the Food and Drug Administration in January 2011, and will be marketed later this year, according to a company spokesperson.
Dr. Kilmer, of the Laser and Skin Surgery Center of Northern California, Sacramento, presented data on a second-generation version of the device that was FDA approved but never marketed. In a multicenter, sham-controlled study, 120 adults with hyperhidrosis were randomized to miraDry (81) or sham treatment (39) for one to three sessions. The patients had to have a Hyperhidrosis Disease Severity Scale (HDSS) score of 3 or 4 ("my underarm sweating is barely tolerable and frequently/always interferes with my daily activities") at baseline, and gravimetric readings greater than 50 mg at each axilla (in 5 minutes).
Follow-up was 6 months for the sham group and 12 months for the treatment group. Responders were defined as subjects reporting a reduction to an HDSS score of 1 ("My underarm sweating is never noticeable and never interferes with daily activities") or 2 ("My underarm sweating is tolerable but sometimes interferes with daily activities"). The patients had a mean age of 33 years, 58% were female, and 84% were white.
Efficacy for the treatment group was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant, Dr. Kilmer reported.
Treatment-related adverse events were generally mild, and all but one resolved. The most common adverse events in the treatment group were transient patches of altered sensation in the treatment limb, occurring in 8 patients (10%), and axillary pain requiring prescription medication in 5 patients (6%). Most subjects experienced transient post-treatment local sequelae in the axilla such as edema, tenderness, and bruising.
Follow-up of the treated subjects showed stable efficacy through 12 months, she said.
The second study, involving 31 patients, investigated the third-generation, optimized version of the device. As in the earlier study, all patients had to have primary axillary hyperhidrosis with an HDSS score of 3 or 4. The patients also were required to have a gravimetric sweat assessment of at least 50 mg in each axilla (in 5 minutes). None of the patients had surgery for axillary hyperhidrosis or botulinum toxin injections in the axillae in the prior 12 months, said Dr. Lupin of Cosmedica Laser Centre, Victoria, B.C.
The patients had a mean age of 33 years (range 18-65 years), and three-quarters were female. They had a mean BMI of 24.8 kg/m2. Of 28 patients seen at 30 days, all but one (96%) had HDSS scores of 1 or 2. Of 25 seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy). Gravimetric assessments 3 months after treatment showed that 94% of patients had at least a 50% reduction in axillary sweat compared with baseline, with an average sweat reduction of 82%.
Nineteen of the 31 initially enrolled patients (61%) experienced at least one treatment-related adverse event, of which most (88%) were rated as mild. The most common were numbness or tingling in an area of the treated limb (12 patients, 39%), and edema in the chest or treatment limb lasting about a week (9 patients, 29%). Most of the patients also experienced acute post-treatment transient effects in the treatment area such as localized edema, tenderness, or erythema. Follow-up of safety is ongoing, Dr. Lupin noted.
"The study is continuing out to 12 months, and at 6 months the preliminary data so far are showing sustained positive improvements in all measures of quality of life, reduction of sweat, and impact on daily living," Dr. Lupin said in an interview. Many patients also reported reduction of axillary hair, and a few patients noted improvement in odor, he said.
Overall, patient satisfaction, as measured by the Dermatology Life Quality Index (DLQI), was 96% at 3 months.
When Dr. Lupin was asked whether miraDry had any disadvantages compared with botulinum toxin type A, he said that there are several short-term advantages to botulinum toxin type A: It is a quicker and easier procedure (about 5-10 minutes) with sweat reduction occurring in just a few days, versus 1 hour for the miraDry procedure, which requires anesthesia and can take a week or longer to produce results. Moreover, botulinum toxin type A is a single treatment and its benefit lasts about 6-8 months, whereas miraDry takes 1-3 sessions for a benefit of at least a year and possibly longer.
Indeed, Dr. Kilmer said in an interview, duration of benefit is an advantage of miraDry over just about every current hyperhidrosis treatment other than sympathectomy, a procedure that is rarely done. So far, the longest miraDry has been studied is 12 months, so "we can’t say for sure, but it didn’t drop off much during that time, so we expect it will last much longer," she said.
Dr. Mathew Avram, director of the Massachusetts General Hospital Dermatology Laser and Cosmetic Center, Boston, said, "This is innovative and interesting technology to address an issue that is very problematic for patients. We need to learn a little more about the duration of these benefits and what if any side effects may be created with repeated treatments over time."
The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
GRAPEVINE, TEX. – A novel microwave device significantly reduced underarm sweating in two studies involving a total of 151 patients with axillary hyperhidrosis.
The condition affects millions of people, yet current treatments are limited by either duration of effect or efficacy. The microwave device has the potential for a longer-term and possibly even permanent effect via eradication of eccrine sweat glands, Dr. Suzanne L. Kilmer and Dr. Mark Lupin reported in separate presentations at the annual meeting of the American Society for Laser Medicine and Surgery.
Miramar Labs' miraDry system focuses microwave energy to selectively heat the interface between the skin and underlying fat, where the sweat glands reside. The system comprises a console, handpiece, and disposable tip. The in-office procedure takes approximately 40 minutes. The system was cleared for licensure in the United States by the Food and Drug Administration in January 2011, and will be marketed later this year, according to a company spokesperson.
Dr. Kilmer, of the Laser and Skin Surgery Center of Northern California, Sacramento, presented data on a second-generation version of the device that was FDA approved but never marketed. In a multicenter, sham-controlled study, 120 adults with hyperhidrosis were randomized to miraDry (81) or sham treatment (39) for one to three sessions. The patients had to have a Hyperhidrosis Disease Severity Scale (HDSS) score of 3 or 4 ("my underarm sweating is barely tolerable and frequently/always interferes with my daily activities") at baseline, and gravimetric readings greater than 50 mg at each axilla (in 5 minutes).
Follow-up was 6 months for the sham group and 12 months for the treatment group. Responders were defined as subjects reporting a reduction to an HDSS score of 1 ("My underarm sweating is never noticeable and never interferes with daily activities") or 2 ("My underarm sweating is tolerable but sometimes interferes with daily activities"). The patients had a mean age of 33 years, 58% were female, and 84% were white.
Efficacy for the treatment group was 89% at 30 days, 74% at 3 months, 67% at 6 months, and 69% at the 9- and 12-month visits. In the sham group, efficacy was 54% at 30 days and 44% for the 3- and 6-month visits. At all time points, the differences between the treatment and sham groups were significant, Dr. Kilmer reported.
Treatment-related adverse events were generally mild, and all but one resolved. The most common adverse events in the treatment group were transient patches of altered sensation in the treatment limb, occurring in 8 patients (10%), and axillary pain requiring prescription medication in 5 patients (6%). Most subjects experienced transient post-treatment local sequelae in the axilla such as edema, tenderness, and bruising.
Follow-up of the treated subjects showed stable efficacy through 12 months, she said.
The second study, involving 31 patients, investigated the third-generation, optimized version of the device. As in the earlier study, all patients had to have primary axillary hyperhidrosis with an HDSS score of 3 or 4. The patients also were required to have a gravimetric sweat assessment of at least 50 mg in each axilla (in 5 minutes). None of the patients had surgery for axillary hyperhidrosis or botulinum toxin injections in the axillae in the prior 12 months, said Dr. Lupin of Cosmedica Laser Centre, Victoria, B.C.
The patients had a mean age of 33 years (range 18-65 years), and three-quarters were female. They had a mean BMI of 24.8 kg/m2. Of 28 patients seen at 30 days, all but one (96%) had HDSS scores of 1 or 2. Of 25 seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy). Gravimetric assessments 3 months after treatment showed that 94% of patients had at least a 50% reduction in axillary sweat compared with baseline, with an average sweat reduction of 82%.
Nineteen of the 31 initially enrolled patients (61%) experienced at least one treatment-related adverse event, of which most (88%) were rated as mild. The most common were numbness or tingling in an area of the treated limb (12 patients, 39%), and edema in the chest or treatment limb lasting about a week (9 patients, 29%). Most of the patients also experienced acute post-treatment transient effects in the treatment area such as localized edema, tenderness, or erythema. Follow-up of safety is ongoing, Dr. Lupin noted.
"The study is continuing out to 12 months, and at 6 months the preliminary data so far are showing sustained positive improvements in all measures of quality of life, reduction of sweat, and impact on daily living," Dr. Lupin said in an interview. Many patients also reported reduction of axillary hair, and a few patients noted improvement in odor, he said.
Overall, patient satisfaction, as measured by the Dermatology Life Quality Index (DLQI), was 96% at 3 months.
When Dr. Lupin was asked whether miraDry had any disadvantages compared with botulinum toxin type A, he said that there are several short-term advantages to botulinum toxin type A: It is a quicker and easier procedure (about 5-10 minutes) with sweat reduction occurring in just a few days, versus 1 hour for the miraDry procedure, which requires anesthesia and can take a week or longer to produce results. Moreover, botulinum toxin type A is a single treatment and its benefit lasts about 6-8 months, whereas miraDry takes 1-3 sessions for a benefit of at least a year and possibly longer.
Indeed, Dr. Kilmer said in an interview, duration of benefit is an advantage of miraDry over just about every current hyperhidrosis treatment other than sympathectomy, a procedure that is rarely done. So far, the longest miraDry has been studied is 12 months, so "we can’t say for sure, but it didn’t drop off much during that time, so we expect it will last much longer," she said.
Dr. Mathew Avram, director of the Massachusetts General Hospital Dermatology Laser and Cosmetic Center, Boston, said, "This is innovative and interesting technology to address an issue that is very problematic for patients. We need to learn a little more about the duration of these benefits and what if any side effects may be created with repeated treatments over time."
The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR LASER MEDICINE AND SURGERY
Major Finding: First study: Efficacy of treatment in 81 patients as determined by HDSS scores was 89% at 30 days and 67% at 6 months. Second study: Of 25 patients seen at 3 months, all had HDSS scores of 1 or 2 (100% efficacy).
Data Source: Two studies of 120 and 31 patients, respectively, with axillary hyperhidrosis
Disclosures: The studies were sponsored by Miramar Labs. Dr. Lupin disclosed that he received a research grant for the study and travel expenses from Miramar, and also received honoraria and travel expenses from Allergan. Dr. Kilmer disclosed that she received research support from Miramar. Dr. Avram is a stockholder in Zeltiq.
Investigational Nd:YAG Laser Plus 3-D Optical Fiber Targets Cellulite
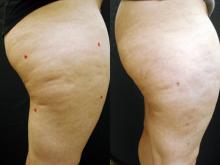
GRAPEVINE, TEX. – An investigational sidelight 3-D optical fiber and 1440-nm Nd:YAG laser produced significant improvement of cellulite with one treatment at 6 months in a study of 15 healthy women.
Cynosure Inc.'s Cellulaze Cellulite Laser Workstation's 1440 wavelength is well absorbed by adipose tissue and water. The side-firing SideLight 3-D optical fiber thermally subcises subcutaneous septa, deplanes fat cells, and heats dermal tissue to promote skin thickening and tightening, said Dr. Bruce E. Katz of the department of dermatology at Mount Sinai School of Medicine, New York.
The 15 women (aged 20-55 years) all had cellulite on their lateral or posterior thighs or buttocks, body mass indexes less than 30 kg/m2, and skin types I-V. Following local anesthesia, two 1.5-mm incisions were made, and the probe was inserted. Subcutaneous temperature was kept at lower than 47° C and surface temperature at lower than 40° C. The Nd:YAG laser delivered 1,000 J per 5- x 5-cm square.
Digital photographic evaluation by two independent observers at 6 months found that 68% of the women had excellent improvement in cellulite; Vectra 3-D imaging demonstrated significant improvement in 65% of them. By physician evaluation, 76% had good or excellent results (69% by patient evaluation). Vectra analysis of skin contour demonstrated an average 47% reduction in depth and 32% reduction in height of skin bulges, Dr. Katz reported at the annual meeting of the American Society for Laser Medicine and Surgery.
Histologically, there was an increase in coarser collagen and elastic fibers in the dermis, noted Dr. Katz, who is also director of the cosmetic surgery and laser clinic at Mount Sinai Medical Center.
Three patients experienced mild ecchymoses, and four had edema lasting less than a week. There were no other adverse events.
"This may be a game changer for the treatment of cellulite," Dr. Katz said.
Cynosure received CE marking certification for Cellulaze in the European Union in February. The U.S. Food and Drug Administration approved an Investigational Device Exemption (IDE) for the device, and a clinical IDE study is currently underway. Regulatory action on a 510(k) submission, filed in late 2010, is currently expected in the first half of 2011, according to a company statement.
Dr. Katz disclosed that he is a Cynosure stockholder.
GRAPEVINE, TEX. – An investigational sidelight 3-D optical fiber and 1440-nm Nd:YAG laser produced significant improvement of cellulite with one treatment at 6 months in a study of 15 healthy women.
Cynosure Inc.'s Cellulaze Cellulite Laser Workstation's 1440 wavelength is well absorbed by adipose tissue and water. The side-firing SideLight 3-D optical fiber thermally subcises subcutaneous septa, deplanes fat cells, and heats dermal tissue to promote skin thickening and tightening, said Dr. Bruce E. Katz of the department of dermatology at Mount Sinai School of Medicine, New York.
The 15 women (aged 20-55 years) all had cellulite on their lateral or posterior thighs or buttocks, body mass indexes less than 30 kg/m2, and skin types I-V. Following local anesthesia, two 1.5-mm incisions were made, and the probe was inserted. Subcutaneous temperature was kept at lower than 47° C and surface temperature at lower than 40° C. The Nd:YAG laser delivered 1,000 J per 5- x 5-cm square.
Digital photographic evaluation by two independent observers at 6 months found that 68% of the women had excellent improvement in cellulite; Vectra 3-D imaging demonstrated significant improvement in 65% of them. By physician evaluation, 76% had good or excellent results (69% by patient evaluation). Vectra analysis of skin contour demonstrated an average 47% reduction in depth and 32% reduction in height of skin bulges, Dr. Katz reported at the annual meeting of the American Society for Laser Medicine and Surgery.
Histologically, there was an increase in coarser collagen and elastic fibers in the dermis, noted Dr. Katz, who is also director of the cosmetic surgery and laser clinic at Mount Sinai Medical Center.
Three patients experienced mild ecchymoses, and four had edema lasting less than a week. There were no other adverse events.
"This may be a game changer for the treatment of cellulite," Dr. Katz said.
Cynosure received CE marking certification for Cellulaze in the European Union in February. The U.S. Food and Drug Administration approved an Investigational Device Exemption (IDE) for the device, and a clinical IDE study is currently underway. Regulatory action on a 510(k) submission, filed in late 2010, is currently expected in the first half of 2011, according to a company statement.
Dr. Katz disclosed that he is a Cynosure stockholder.
GRAPEVINE, TEX. – An investigational sidelight 3-D optical fiber and 1440-nm Nd:YAG laser produced significant improvement of cellulite with one treatment at 6 months in a study of 15 healthy women.
Cynosure Inc.'s Cellulaze Cellulite Laser Workstation's 1440 wavelength is well absorbed by adipose tissue and water. The side-firing SideLight 3-D optical fiber thermally subcises subcutaneous septa, deplanes fat cells, and heats dermal tissue to promote skin thickening and tightening, said Dr. Bruce E. Katz of the department of dermatology at Mount Sinai School of Medicine, New York.
The 15 women (aged 20-55 years) all had cellulite on their lateral or posterior thighs or buttocks, body mass indexes less than 30 kg/m2, and skin types I-V. Following local anesthesia, two 1.5-mm incisions were made, and the probe was inserted. Subcutaneous temperature was kept at lower than 47° C and surface temperature at lower than 40° C. The Nd:YAG laser delivered 1,000 J per 5- x 5-cm square.
Digital photographic evaluation by two independent observers at 6 months found that 68% of the women had excellent improvement in cellulite; Vectra 3-D imaging demonstrated significant improvement in 65% of them. By physician evaluation, 76% had good or excellent results (69% by patient evaluation). Vectra analysis of skin contour demonstrated an average 47% reduction in depth and 32% reduction in height of skin bulges, Dr. Katz reported at the annual meeting of the American Society for Laser Medicine and Surgery.
Histologically, there was an increase in coarser collagen and elastic fibers in the dermis, noted Dr. Katz, who is also director of the cosmetic surgery and laser clinic at Mount Sinai Medical Center.
Three patients experienced mild ecchymoses, and four had edema lasting less than a week. There were no other adverse events.
"This may be a game changer for the treatment of cellulite," Dr. Katz said.
Cynosure received CE marking certification for Cellulaze in the European Union in February. The U.S. Food and Drug Administration approved an Investigational Device Exemption (IDE) for the device, and a clinical IDE study is currently underway. Regulatory action on a 510(k) submission, filed in late 2010, is currently expected in the first half of 2011, according to a company statement.
Dr. Katz disclosed that he is a Cynosure stockholder.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR LASER MEDICINE AND SURGERY
Major Finding: Digital photographic evaluation by two independent observers at 6 months found that 68% had excellent improvement in cellulite, and Vectra 3-D imaging demonstrated significant improvement in 65%. By physician evaluation, 76% had good or excellent results (69% by patient evaluation).
Data Source: A 6-month study of 15 women aged 20-55 years with cellulite on their lateral or posterior thighs or buttocks.
Disclosures: The study was funded by Cynosure, of which Dr. Katz is a stockholder.
Study of Human Parechovirus in U.S. Finds 7% Prevalence
Human parechovirus was found in 7.4% of nearly 800 cerebrospinal fluid samples taken from children seen at a Midwestern U.S. pediatric hospital over a 3-year period, Rangaraj Selvarangan, Ph.D., and his associates reported in the March issue of the Pediatric Infectious Disease Journal.
Sepsislike syndrome was the most common clinical presentation in that group, along with irritability, fever, and nonspecific rash. However, the combination of abdominal distension and an erythematous rash was a distinctive feature of human parechovirus (HPeV) among eight infants with sepsislike illness described in a separate case series report published in the same issue of the journal (PIDJ 2011;30:260-2).
Human parechovirus (HPeV) is a newly classified genus ("Parechovirus") that was previously classified as enterovirus within the family Picornaviridae. Detectable by specific (i.e., not enteroviral) real-time reverse-transcription polymerase chain reaction (RT-PCR) assay, HPeV has been associated with sepsislike illness and meningitis, particularly in young infants. Until now, epidemiologic data on HPeV illness have come from Europe, South America, and Asia, but not the United States, said Dr. Selvarangan and his associates at Children’s Mercy Hospital, Kansas City, Mo. (PIDJ 2011;30:238-42).
Of 780 enterovirus RNA-negative cerebral spinal fluid (CSF) samples taken during 2006-2008, RT-PCR detected HPeV in 2% of 218 samples from 2006, 17% of 320 from 2007, and none of 242 from 2008. Most samples were taken between June and October. The prevalence of enterovirus in CSF from children in the Kansas City area during the same time frame ranged from 20% to 33%, the investigators noted.
Most of those positive for HPeV were male (71%), with a mean age of 6.6 weeks (range 1 week to 7 months). Two-thirds (66%) presented with sepsislike syndrome, and 19% with suspected meningitis. The most common symptoms were irritability (98%), fever (95%), and maculopapular rash (60%). Mottling of the extremities was noted in 18%, and hypothermia in 9%. Other signs included vomiting (19%), nonpurulent conjunctivitis (9%), neurologic symptoms (7%), abdominal distension (5%), and apnea (2%).
In contrast, among the eight infant cases reported in a separate article, five had abdominal distension – including one case so severe it was mistaken for a surgical condition – while six had an erythematous rash and four had both. Definite neurologic symptoms, including seizures, hypotonia, and apnea, were also present in four infants, Dr. Harish Bangalore of Evelina Children’s Hospital, London, reported.
In the larger Kansas City study, CSF pleocytosis was noted in only 7 of the 58 patients (12%), and abnormal CSF glucose and protein also were uncommon. Of the 54 patients with complete blood count data, the mean peripheral white blood cell count was 7,000/mm3, with leukopenia in 19 of 55 (35%). Of 30 patients with viral cultures, all were negative and just 1 bacterial culture was positive. All but one patient was hospitalized, with a mean stay of 3.6 days (range 0-13 days). Nearly all patients (57/58) were treated with IV antibacterials.
A limitation of the study was that it only included cases in which meningitis or sepsis was suspected, so that other clinical presentations of HPeV – such as upper respiratory infection – would not have been detected, Dr. Selvarangan and associates noted.
All of the eight case-reported infants (mean age 25 days) had lymphopenia at admission, combined in five infants with a failure to mount a C-reactive protein (CRP) response of more than 5 mg/L during their illness. CSF was the source of the HPeV-positive sample in six of the eight, stool/rectal swabs in four, and blood in one, Dr. Bangalore and associates said.
The "striking" clinical picture of prominent abdominal distension and a widespread erythematous rash in a young infant with sepsislike illness, along with fever, irritability, lymphopenia, and relative thrombocytopenia without CRP elevation, may assist in early diagnosis and avoid both confusion with surgical conditions and unnecessary broad-spectrum antibiotic use. A specific RT-PCR assay is required for HPeV detection from blood, CSF, throat, or rectal swabs, rather than the standard enterovirus detection methods, Dr. Bangalore and associates noted.
The Kansas City investigators found a triad of fever, irritability, and rash as the most common presentation of HPeV in their study, with infections occurring in late summer or autumn with "strikingly variable annual prevalence." Dr. Selvarangan and his associates suggested "that HPeV CNS infection should be considered with sepsislike illness of infants even in the absence of CSF pleocytosis. Addition of HPeV RT-PCR assay to the enterovirus RT-PCR assay on pediatric CSF specimens (particularly infants less than 6 months old) could reduce hospital stay, antibiotic usage, and hospitalization costs."
The Kansas City study was supported by research residual funds from Dr. Selvarangan. Dr. Selvarangan said he had no other relevant financial disclosures. Dr. Bangalore said he had no relevant financial disclosures.
Human parechovirus was found in 7.4% of nearly 800 cerebrospinal fluid samples taken from children seen at a Midwestern U.S. pediatric hospital over a 3-year period, Rangaraj Selvarangan, Ph.D., and his associates reported in the March issue of the Pediatric Infectious Disease Journal.
Sepsislike syndrome was the most common clinical presentation in that group, along with irritability, fever, and nonspecific rash. However, the combination of abdominal distension and an erythematous rash was a distinctive feature of human parechovirus (HPeV) among eight infants with sepsislike illness described in a separate case series report published in the same issue of the journal (PIDJ 2011;30:260-2).
Human parechovirus (HPeV) is a newly classified genus ("Parechovirus") that was previously classified as enterovirus within the family Picornaviridae. Detectable by specific (i.e., not enteroviral) real-time reverse-transcription polymerase chain reaction (RT-PCR) assay, HPeV has been associated with sepsislike illness and meningitis, particularly in young infants. Until now, epidemiologic data on HPeV illness have come from Europe, South America, and Asia, but not the United States, said Dr. Selvarangan and his associates at Children’s Mercy Hospital, Kansas City, Mo. (PIDJ 2011;30:238-42).
Of 780 enterovirus RNA-negative cerebral spinal fluid (CSF) samples taken during 2006-2008, RT-PCR detected HPeV in 2% of 218 samples from 2006, 17% of 320 from 2007, and none of 242 from 2008. Most samples were taken between June and October. The prevalence of enterovirus in CSF from children in the Kansas City area during the same time frame ranged from 20% to 33%, the investigators noted.
Most of those positive for HPeV were male (71%), with a mean age of 6.6 weeks (range 1 week to 7 months). Two-thirds (66%) presented with sepsislike syndrome, and 19% with suspected meningitis. The most common symptoms were irritability (98%), fever (95%), and maculopapular rash (60%). Mottling of the extremities was noted in 18%, and hypothermia in 9%. Other signs included vomiting (19%), nonpurulent conjunctivitis (9%), neurologic symptoms (7%), abdominal distension (5%), and apnea (2%).
In contrast, among the eight infant cases reported in a separate article, five had abdominal distension – including one case so severe it was mistaken for a surgical condition – while six had an erythematous rash and four had both. Definite neurologic symptoms, including seizures, hypotonia, and apnea, were also present in four infants, Dr. Harish Bangalore of Evelina Children’s Hospital, London, reported.
In the larger Kansas City study, CSF pleocytosis was noted in only 7 of the 58 patients (12%), and abnormal CSF glucose and protein also were uncommon. Of the 54 patients with complete blood count data, the mean peripheral white blood cell count was 7,000/mm3, with leukopenia in 19 of 55 (35%). Of 30 patients with viral cultures, all were negative and just 1 bacterial culture was positive. All but one patient was hospitalized, with a mean stay of 3.6 days (range 0-13 days). Nearly all patients (57/58) were treated with IV antibacterials.
A limitation of the study was that it only included cases in which meningitis or sepsis was suspected, so that other clinical presentations of HPeV – such as upper respiratory infection – would not have been detected, Dr. Selvarangan and associates noted.
All of the eight case-reported infants (mean age 25 days) had lymphopenia at admission, combined in five infants with a failure to mount a C-reactive protein (CRP) response of more than 5 mg/L during their illness. CSF was the source of the HPeV-positive sample in six of the eight, stool/rectal swabs in four, and blood in one, Dr. Bangalore and associates said.
The "striking" clinical picture of prominent abdominal distension and a widespread erythematous rash in a young infant with sepsislike illness, along with fever, irritability, lymphopenia, and relative thrombocytopenia without CRP elevation, may assist in early diagnosis and avoid both confusion with surgical conditions and unnecessary broad-spectrum antibiotic use. A specific RT-PCR assay is required for HPeV detection from blood, CSF, throat, or rectal swabs, rather than the standard enterovirus detection methods, Dr. Bangalore and associates noted.
The Kansas City investigators found a triad of fever, irritability, and rash as the most common presentation of HPeV in their study, with infections occurring in late summer or autumn with "strikingly variable annual prevalence." Dr. Selvarangan and his associates suggested "that HPeV CNS infection should be considered with sepsislike illness of infants even in the absence of CSF pleocytosis. Addition of HPeV RT-PCR assay to the enterovirus RT-PCR assay on pediatric CSF specimens (particularly infants less than 6 months old) could reduce hospital stay, antibiotic usage, and hospitalization costs."
The Kansas City study was supported by research residual funds from Dr. Selvarangan. Dr. Selvarangan said he had no other relevant financial disclosures. Dr. Bangalore said he had no relevant financial disclosures.
Human parechovirus was found in 7.4% of nearly 800 cerebrospinal fluid samples taken from children seen at a Midwestern U.S. pediatric hospital over a 3-year period, Rangaraj Selvarangan, Ph.D., and his associates reported in the March issue of the Pediatric Infectious Disease Journal.
Sepsislike syndrome was the most common clinical presentation in that group, along with irritability, fever, and nonspecific rash. However, the combination of abdominal distension and an erythematous rash was a distinctive feature of human parechovirus (HPeV) among eight infants with sepsislike illness described in a separate case series report published in the same issue of the journal (PIDJ 2011;30:260-2).
Human parechovirus (HPeV) is a newly classified genus ("Parechovirus") that was previously classified as enterovirus within the family Picornaviridae. Detectable by specific (i.e., not enteroviral) real-time reverse-transcription polymerase chain reaction (RT-PCR) assay, HPeV has been associated with sepsislike illness and meningitis, particularly in young infants. Until now, epidemiologic data on HPeV illness have come from Europe, South America, and Asia, but not the United States, said Dr. Selvarangan and his associates at Children’s Mercy Hospital, Kansas City, Mo. (PIDJ 2011;30:238-42).
Of 780 enterovirus RNA-negative cerebral spinal fluid (CSF) samples taken during 2006-2008, RT-PCR detected HPeV in 2% of 218 samples from 2006, 17% of 320 from 2007, and none of 242 from 2008. Most samples were taken between June and October. The prevalence of enterovirus in CSF from children in the Kansas City area during the same time frame ranged from 20% to 33%, the investigators noted.
Most of those positive for HPeV were male (71%), with a mean age of 6.6 weeks (range 1 week to 7 months). Two-thirds (66%) presented with sepsislike syndrome, and 19% with suspected meningitis. The most common symptoms were irritability (98%), fever (95%), and maculopapular rash (60%). Mottling of the extremities was noted in 18%, and hypothermia in 9%. Other signs included vomiting (19%), nonpurulent conjunctivitis (9%), neurologic symptoms (7%), abdominal distension (5%), and apnea (2%).
In contrast, among the eight infant cases reported in a separate article, five had abdominal distension – including one case so severe it was mistaken for a surgical condition – while six had an erythematous rash and four had both. Definite neurologic symptoms, including seizures, hypotonia, and apnea, were also present in four infants, Dr. Harish Bangalore of Evelina Children’s Hospital, London, reported.
In the larger Kansas City study, CSF pleocytosis was noted in only 7 of the 58 patients (12%), and abnormal CSF glucose and protein also were uncommon. Of the 54 patients with complete blood count data, the mean peripheral white blood cell count was 7,000/mm3, with leukopenia in 19 of 55 (35%). Of 30 patients with viral cultures, all were negative and just 1 bacterial culture was positive. All but one patient was hospitalized, with a mean stay of 3.6 days (range 0-13 days). Nearly all patients (57/58) were treated with IV antibacterials.
A limitation of the study was that it only included cases in which meningitis or sepsis was suspected, so that other clinical presentations of HPeV – such as upper respiratory infection – would not have been detected, Dr. Selvarangan and associates noted.
All of the eight case-reported infants (mean age 25 days) had lymphopenia at admission, combined in five infants with a failure to mount a C-reactive protein (CRP) response of more than 5 mg/L during their illness. CSF was the source of the HPeV-positive sample in six of the eight, stool/rectal swabs in four, and blood in one, Dr. Bangalore and associates said.
The "striking" clinical picture of prominent abdominal distension and a widespread erythematous rash in a young infant with sepsislike illness, along with fever, irritability, lymphopenia, and relative thrombocytopenia without CRP elevation, may assist in early diagnosis and avoid both confusion with surgical conditions and unnecessary broad-spectrum antibiotic use. A specific RT-PCR assay is required for HPeV detection from blood, CSF, throat, or rectal swabs, rather than the standard enterovirus detection methods, Dr. Bangalore and associates noted.
The Kansas City investigators found a triad of fever, irritability, and rash as the most common presentation of HPeV in their study, with infections occurring in late summer or autumn with "strikingly variable annual prevalence." Dr. Selvarangan and his associates suggested "that HPeV CNS infection should be considered with sepsislike illness of infants even in the absence of CSF pleocytosis. Addition of HPeV RT-PCR assay to the enterovirus RT-PCR assay on pediatric CSF specimens (particularly infants less than 6 months old) could reduce hospital stay, antibiotic usage, and hospitalization costs."
The Kansas City study was supported by research residual funds from Dr. Selvarangan. Dr. Selvarangan said he had no other relevant financial disclosures. Dr. Bangalore said he had no relevant financial disclosures.
FROM THE PEDIATRIC INFECTIOUS DISEASE JOURNAL
Study of Human Parechovirus in U.S. Finds 7% Prevalence
Human parechovirus was found in 7.4% of nearly 800 cerebrospinal fluid samples taken from children seen at a Midwestern U.S. pediatric hospital over a 3-year period, Rangaraj Selvarangan, Ph.D., and his associates reported in the March issue of the Pediatric Infectious Disease Journal.
Sepsislike syndrome was the most common clinical presentation in that group, along with irritability, fever, and nonspecific rash. However, the combination of abdominal distension and an erythematous rash was a distinctive feature of human parechovirus (HPeV) among eight infants with sepsislike illness described in a separate case series report published in the same issue of the journal (PIDJ 2011;30:260-2).
Human parechovirus (HPeV) is a newly classified genus ("Parechovirus") that was previously classified as enterovirus within the family Picornaviridae. Detectable by specific (i.e., not enteroviral) real-time reverse-transcription polymerase chain reaction (RT-PCR) assay, HPeV has been associated with sepsislike illness and meningitis, particularly in young infants. Until now, epidemiologic data on HPeV illness have come from Europe, South America, and Asia, but not the United States, said Dr. Selvarangan and his associates at Children’s Mercy Hospital, Kansas City, Mo. (PIDJ 2011;30:238-42).
Of 780 enterovirus RNA-negative cerebral spinal fluid (CSF) samples taken during 2006-2008, RT-PCR detected HPeV in 2% of 218 samples from 2006, 17% of 320 from 2007, and none of 242 from 2008. Most samples were taken between June and October. The prevalence of enterovirus in CSF from children in the Kansas City area during the same time frame ranged from 20% to 33%, the investigators noted.
Most of those positive for HPeV were male (71%), with a mean age of 6.6 weeks (range 1 week to 7 months). Two-thirds (66%) presented with sepsislike syndrome, and 19% with suspected meningitis. The most common symptoms were irritability (98%), fever (95%), and maculopapular rash (60%). Mottling of the extremities was noted in 18%, and hypothermia in 9%. Other signs included vomiting (19%), nonpurulent conjunctivitis (9%), neurologic symptoms (7%), abdominal distension (5%), and apnea (2%).
In contrast, among the eight infant cases reported in a separate article, five had abdominal distension – including one case so severe it was mistaken for a surgical condition – while six had an erythematous rash and four had both. Definite neurologic symptoms, including seizures, hypotonia, and apnea, were also present in four infants, Dr. Harish Bangalore of Evelina Children’s Hospital, London, reported.
In the larger Kansas City study, CSF pleocytosis was noted in only 7 of the 58 patients (12%), and abnormal CSF glucose and protein also were uncommon. Of the 54 patients with complete blood count data, the mean peripheral white blood cell count was 7,000/mm3, with leukopenia in 19 of 55 (35%). Of 30 patients with viral cultures, all were negative and just 1 bacterial culture was positive. All but one patient was hospitalized, with a mean stay of 3.6 days (range 0-13 days). Nearly all patients (57/58) were treated with IV antibacterials.
A limitation of the study was that it only included cases in which meningitis or sepsis was suspected, so that other clinical presentations of HPeV – such as upper respiratory infection – would not have been detected, Dr. Selvarangan and associates noted.
All of the eight case-reported infants (mean age 25 days) had lymphopenia at admission, combined in five infants with a failure to mount a C-reactive protein (CRP) response of more than 5 mg/L during their illness. CSF was the source of the HPeV-positive sample in six of the eight, stool/rectal swabs in four, and blood in one, Dr. Bangalore and associates said.
The "striking" clinical picture of prominent abdominal distension and a widespread erythematous rash in a young infant with sepsislike illness, along with fever, irritability, lymphopenia, and relative thrombocytopenia without CRP elevation, may assist in early diagnosis and avoid both confusion with surgical conditions and unnecessary broad-spectrum antibiotic use. A specific RT-PCR assay is required for HPeV detection from blood, CSF, throat, or rectal swabs, rather than the standard enterovirus detection methods, Dr. Bangalore and associates noted.
The Kansas City investigators found a triad of fever, irritability, and rash as the most common presentation of HPeV in their study, with infections occurring in late summer or autumn with "strikingly variable annual prevalence." Dr. Selvarangan and his associates suggested "that HPeV CNS infection should be considered with sepsislike illness of infants even in the absence of CSF pleocytosis. Addition of HPeV RT-PCR assay to the enterovirus RT-PCR assay on pediatric CSF specimens (particularly infants less than 6 months old) could reduce hospital stay, antibiotic usage, and hospitalization costs."
The Kansas City study was supported by research residual funds from Dr. Selvarangan. Dr. Selvarangan said he had no other relevant financial disclosures. Dr. Bangalore said he had no relevant financial disclosures.
Human parechovirus was found in 7.4% of nearly 800 cerebrospinal fluid samples taken from children seen at a Midwestern U.S. pediatric hospital over a 3-year period, Rangaraj Selvarangan, Ph.D., and his associates reported in the March issue of the Pediatric Infectious Disease Journal.
Sepsislike syndrome was the most common clinical presentation in that group, along with irritability, fever, and nonspecific rash. However, the combination of abdominal distension and an erythematous rash was a distinctive feature of human parechovirus (HPeV) among eight infants with sepsislike illness described in a separate case series report published in the same issue of the journal (PIDJ 2011;30:260-2).
Human parechovirus (HPeV) is a newly classified genus ("Parechovirus") that was previously classified as enterovirus within the family Picornaviridae. Detectable by specific (i.e., not enteroviral) real-time reverse-transcription polymerase chain reaction (RT-PCR) assay, HPeV has been associated with sepsislike illness and meningitis, particularly in young infants. Until now, epidemiologic data on HPeV illness have come from Europe, South America, and Asia, but not the United States, said Dr. Selvarangan and his associates at Children’s Mercy Hospital, Kansas City, Mo. (PIDJ 2011;30:238-42).
Of 780 enterovirus RNA-negative cerebral spinal fluid (CSF) samples taken during 2006-2008, RT-PCR detected HPeV in 2% of 218 samples from 2006, 17% of 320 from 2007, and none of 242 from 2008. Most samples were taken between June and October. The prevalence of enterovirus in CSF from children in the Kansas City area during the same time frame ranged from 20% to 33%, the investigators noted.
Most of those positive for HPeV were male (71%), with a mean age of 6.6 weeks (range 1 week to 7 months). Two-thirds (66%) presented with sepsislike syndrome, and 19% with suspected meningitis. The most common symptoms were irritability (98%), fever (95%), and maculopapular rash (60%). Mottling of the extremities was noted in 18%, and hypothermia in 9%. Other signs included vomiting (19%), nonpurulent conjunctivitis (9%), neurologic symptoms (7%), abdominal distension (5%), and apnea (2%).
In contrast, among the eight infant cases reported in a separate article, five had abdominal distension – including one case so severe it was mistaken for a surgical condition – while six had an erythematous rash and four had both. Definite neurologic symptoms, including seizures, hypotonia, and apnea, were also present in four infants, Dr. Harish Bangalore of Evelina Children’s Hospital, London, reported.
In the larger Kansas City study, CSF pleocytosis was noted in only 7 of the 58 patients (12%), and abnormal CSF glucose and protein also were uncommon. Of the 54 patients with complete blood count data, the mean peripheral white blood cell count was 7,000/mm3, with leukopenia in 19 of 55 (35%). Of 30 patients with viral cultures, all were negative and just 1 bacterial culture was positive. All but one patient was hospitalized, with a mean stay of 3.6 days (range 0-13 days). Nearly all patients (57/58) were treated with IV antibacterials.
A limitation of the study was that it only included cases in which meningitis or sepsis was suspected, so that other clinical presentations of HPeV – such as upper respiratory infection – would not have been detected, Dr. Selvarangan and associates noted.
All of the eight case-reported infants (mean age 25 days) had lymphopenia at admission, combined in five infants with a failure to mount a C-reactive protein (CRP) response of more than 5 mg/L during their illness. CSF was the source of the HPeV-positive sample in six of the eight, stool/rectal swabs in four, and blood in one, Dr. Bangalore and associates said.
The "striking" clinical picture of prominent abdominal distension and a widespread erythematous rash in a young infant with sepsislike illness, along with fever, irritability, lymphopenia, and relative thrombocytopenia without CRP elevation, may assist in early diagnosis and avoid both confusion with surgical conditions and unnecessary broad-spectrum antibiotic use. A specific RT-PCR assay is required for HPeV detection from blood, CSF, throat, or rectal swabs, rather than the standard enterovirus detection methods, Dr. Bangalore and associates noted.
The Kansas City investigators found a triad of fever, irritability, and rash as the most common presentation of HPeV in their study, with infections occurring in late summer or autumn with "strikingly variable annual prevalence." Dr. Selvarangan and his associates suggested "that HPeV CNS infection should be considered with sepsislike illness of infants even in the absence of CSF pleocytosis. Addition of HPeV RT-PCR assay to the enterovirus RT-PCR assay on pediatric CSF specimens (particularly infants less than 6 months old) could reduce hospital stay, antibiotic usage, and hospitalization costs."
The Kansas City study was supported by research residual funds from Dr. Selvarangan. Dr. Selvarangan said he had no other relevant financial disclosures. Dr. Bangalore said he had no relevant financial disclosures.
Human parechovirus was found in 7.4% of nearly 800 cerebrospinal fluid samples taken from children seen at a Midwestern U.S. pediatric hospital over a 3-year period, Rangaraj Selvarangan, Ph.D., and his associates reported in the March issue of the Pediatric Infectious Disease Journal.
Sepsislike syndrome was the most common clinical presentation in that group, along with irritability, fever, and nonspecific rash. However, the combination of abdominal distension and an erythematous rash was a distinctive feature of human parechovirus (HPeV) among eight infants with sepsislike illness described in a separate case series report published in the same issue of the journal (PIDJ 2011;30:260-2).
Human parechovirus (HPeV) is a newly classified genus ("Parechovirus") that was previously classified as enterovirus within the family Picornaviridae. Detectable by specific (i.e., not enteroviral) real-time reverse-transcription polymerase chain reaction (RT-PCR) assay, HPeV has been associated with sepsislike illness and meningitis, particularly in young infants. Until now, epidemiologic data on HPeV illness have come from Europe, South America, and Asia, but not the United States, said Dr. Selvarangan and his associates at Children’s Mercy Hospital, Kansas City, Mo. (PIDJ 2011;30:238-42).
Of 780 enterovirus RNA-negative cerebral spinal fluid (CSF) samples taken during 2006-2008, RT-PCR detected HPeV in 2% of 218 samples from 2006, 17% of 320 from 2007, and none of 242 from 2008. Most samples were taken between June and October. The prevalence of enterovirus in CSF from children in the Kansas City area during the same time frame ranged from 20% to 33%, the investigators noted.
Most of those positive for HPeV were male (71%), with a mean age of 6.6 weeks (range 1 week to 7 months). Two-thirds (66%) presented with sepsislike syndrome, and 19% with suspected meningitis. The most common symptoms were irritability (98%), fever (95%), and maculopapular rash (60%). Mottling of the extremities was noted in 18%, and hypothermia in 9%. Other signs included vomiting (19%), nonpurulent conjunctivitis (9%), neurologic symptoms (7%), abdominal distension (5%), and apnea (2%).
In contrast, among the eight infant cases reported in a separate article, five had abdominal distension – including one case so severe it was mistaken for a surgical condition – while six had an erythematous rash and four had both. Definite neurologic symptoms, including seizures, hypotonia, and apnea, were also present in four infants, Dr. Harish Bangalore of Evelina Children’s Hospital, London, reported.
In the larger Kansas City study, CSF pleocytosis was noted in only 7 of the 58 patients (12%), and abnormal CSF glucose and protein also were uncommon. Of the 54 patients with complete blood count data, the mean peripheral white blood cell count was 7,000/mm3, with leukopenia in 19 of 55 (35%). Of 30 patients with viral cultures, all were negative and just 1 bacterial culture was positive. All but one patient was hospitalized, with a mean stay of 3.6 days (range 0-13 days). Nearly all patients (57/58) were treated with IV antibacterials.
A limitation of the study was that it only included cases in which meningitis or sepsis was suspected, so that other clinical presentations of HPeV – such as upper respiratory infection – would not have been detected, Dr. Selvarangan and associates noted.
All of the eight case-reported infants (mean age 25 days) had lymphopenia at admission, combined in five infants with a failure to mount a C-reactive protein (CRP) response of more than 5 mg/L during their illness. CSF was the source of the HPeV-positive sample in six of the eight, stool/rectal swabs in four, and blood in one, Dr. Bangalore and associates said.
The "striking" clinical picture of prominent abdominal distension and a widespread erythematous rash in a young infant with sepsislike illness, along with fever, irritability, lymphopenia, and relative thrombocytopenia without CRP elevation, may assist in early diagnosis and avoid both confusion with surgical conditions and unnecessary broad-spectrum antibiotic use. A specific RT-PCR assay is required for HPeV detection from blood, CSF, throat, or rectal swabs, rather than the standard enterovirus detection methods, Dr. Bangalore and associates noted.
The Kansas City investigators found a triad of fever, irritability, and rash as the most common presentation of HPeV in their study, with infections occurring in late summer or autumn with "strikingly variable annual prevalence." Dr. Selvarangan and his associates suggested "that HPeV CNS infection should be considered with sepsislike illness of infants even in the absence of CSF pleocytosis. Addition of HPeV RT-PCR assay to the enterovirus RT-PCR assay on pediatric CSF specimens (particularly infants less than 6 months old) could reduce hospital stay, antibiotic usage, and hospitalization costs."
The Kansas City study was supported by research residual funds from Dr. Selvarangan. Dr. Selvarangan said he had no other relevant financial disclosures. Dr. Bangalore said he had no relevant financial disclosures.
FROM THE PEDIATRIC INFECTIOUS DISEASE JOURNAL
Major Finding: Of 780 enterovirus RNA–negative CSF samples taken during 2006-2008, RT-PCR detected HPeV in 2% of 218 samples from 2006, 17% of 320 from 2007, and in none of 242 from 2008. A separate case report found abdominal distension in 6 of 8 infants with HPeV.
Data Source: Retrospective RT-PCR analysis of frozen CSF samples from a 3-year period in the larger study, and a case report of eight infants.
Disclosures: The Kansas City study was supported by research residual funds from Dr. Selvarangan. Dr. Selvarangan said he had no other relevant financial disclosures. Dr. Bangalore said he had no relevant financial disclosures.
Near-Elimination of Tobacco Use Deemed a Priority by U.N. Body
Near-elimination of tobacco consumption worldwide by 2040 was proposed as the No. 1 priority for this fall’s United Nations High-Level Meeting on Non-Communicable Disease in a joint statement on April 6 from two stakeholder coalitions.
Dietary salt reduction, improved diet and physical activity, reduction in hazardous alcohol intake, and universal access to essential drugs and technologies were also listed as top global NCD priorities by the Lancet NCD Action Group, an informal collaboration of academics, practitioners, and civil society organizations, and the NCD Alliance, comprising four international nongovernmental organizations (Union for International Cancer Control, International Union Against Tuberculosis and Lung Disease, International Diabetes Federation, and World Heart Federation).
The UN High-Level Meeting (UN HLM) on NCDs in September 2011 is expected to focus the world’s attention on NCDs in the same way that a similar meeting did for HIV/AIDS in 2001, which concluded that dealing with the disease was central to the world development agenda. The rising global epidemic of NCDs is now responsible for two-thirds of all deaths worldwide and has become a major barrier to development, according to Dr. Robert Beaglehole of the University of Aukland, New Zealand, and 43 coauthors representing the two umbrella groups (Lancet 2011 [doi:10.1016/S0140-6736(11)60393-0])
"The UN HLM is a turning point in the way we approach global health issues, and it will place NCDs on the development agenda. The global community has to take this opportunity and sustain the momentum to achieve the goal of avoiding premature NCD deaths and disability, thus improving global health in the years to come," the authors wrote.
The five priorities were chosen because there is good evidence for each regarding their substantial impact on health and cost effectiveness, the low cost of implementation, and financial feasibility for scale-up. Specifically, the authors proposed goals of reducing worldwide tobacco consumption to less than 5% by the year 2040 and salt consumption to less than 5g (2,000 mg sodium) per person per day by 2025. "We propose a goal to achieve a world essentially free from tobacco by 2040," they said.
Tobacco reduction would be accomplished via full implementation of the 2003 World Health Organization Framework Convention on Tobacco Control, which calls for reducing demand for tobacco via methods such as raising tobacco taxes, legislation of health warnings, and smoking prohibitions. Salt reduction would be accomplished via mass-media campaigns and voluntary reformulation of food products by industry.
Using those methods, the yearly cost to implement tobacco control and salt reduction would be about 20 cents per person per year in countries such as India and China, with the total package of priority interventions priced at about $9 billion per year, the authors said.
Keys to progress include leadership at the highest levels of government, a focus on prevention, treatment, international cooperation, monitoring, reporting and accountability, they said.
"An ideal outcome of the UN HLM will be a sustained commitment to a set of feasible actions and interventions for which specific and timed targets and indicators can be developed, and progress can be readily measured."
The NCD Alliance has also issued a proposed outcomes document for the UN High-Level meeting.
Dr. Beaglehole declared that he has no disclosures. Two of the authors declared financial relationships with pharmaceutical companies, and two others received grants from charities including the Wellcome Trust. One of the authors, Richard Horton, is editor of the Lancet. The others declared that they have no disclosures.
Near-elimination of tobacco consumption worldwide by 2040 was proposed as the No. 1 priority for this fall’s United Nations High-Level Meeting on Non-Communicable Disease in a joint statement on April 6 from two stakeholder coalitions.
Dietary salt reduction, improved diet and physical activity, reduction in hazardous alcohol intake, and universal access to essential drugs and technologies were also listed as top global NCD priorities by the Lancet NCD Action Group, an informal collaboration of academics, practitioners, and civil society organizations, and the NCD Alliance, comprising four international nongovernmental organizations (Union for International Cancer Control, International Union Against Tuberculosis and Lung Disease, International Diabetes Federation, and World Heart Federation).
The UN High-Level Meeting (UN HLM) on NCDs in September 2011 is expected to focus the world’s attention on NCDs in the same way that a similar meeting did for HIV/AIDS in 2001, which concluded that dealing with the disease was central to the world development agenda. The rising global epidemic of NCDs is now responsible for two-thirds of all deaths worldwide and has become a major barrier to development, according to Dr. Robert Beaglehole of the University of Aukland, New Zealand, and 43 coauthors representing the two umbrella groups (Lancet 2011 [doi:10.1016/S0140-6736(11)60393-0])
"The UN HLM is a turning point in the way we approach global health issues, and it will place NCDs on the development agenda. The global community has to take this opportunity and sustain the momentum to achieve the goal of avoiding premature NCD deaths and disability, thus improving global health in the years to come," the authors wrote.
The five priorities were chosen because there is good evidence for each regarding their substantial impact on health and cost effectiveness, the low cost of implementation, and financial feasibility for scale-up. Specifically, the authors proposed goals of reducing worldwide tobacco consumption to less than 5% by the year 2040 and salt consumption to less than 5g (2,000 mg sodium) per person per day by 2025. "We propose a goal to achieve a world essentially free from tobacco by 2040," they said.
Tobacco reduction would be accomplished via full implementation of the 2003 World Health Organization Framework Convention on Tobacco Control, which calls for reducing demand for tobacco via methods such as raising tobacco taxes, legislation of health warnings, and smoking prohibitions. Salt reduction would be accomplished via mass-media campaigns and voluntary reformulation of food products by industry.
Using those methods, the yearly cost to implement tobacco control and salt reduction would be about 20 cents per person per year in countries such as India and China, with the total package of priority interventions priced at about $9 billion per year, the authors said.
Keys to progress include leadership at the highest levels of government, a focus on prevention, treatment, international cooperation, monitoring, reporting and accountability, they said.
"An ideal outcome of the UN HLM will be a sustained commitment to a set of feasible actions and interventions for which specific and timed targets and indicators can be developed, and progress can be readily measured."
The NCD Alliance has also issued a proposed outcomes document for the UN High-Level meeting.
Dr. Beaglehole declared that he has no disclosures. Two of the authors declared financial relationships with pharmaceutical companies, and two others received grants from charities including the Wellcome Trust. One of the authors, Richard Horton, is editor of the Lancet. The others declared that they have no disclosures.
Near-elimination of tobacco consumption worldwide by 2040 was proposed as the No. 1 priority for this fall’s United Nations High-Level Meeting on Non-Communicable Disease in a joint statement on April 6 from two stakeholder coalitions.
Dietary salt reduction, improved diet and physical activity, reduction in hazardous alcohol intake, and universal access to essential drugs and technologies were also listed as top global NCD priorities by the Lancet NCD Action Group, an informal collaboration of academics, practitioners, and civil society organizations, and the NCD Alliance, comprising four international nongovernmental organizations (Union for International Cancer Control, International Union Against Tuberculosis and Lung Disease, International Diabetes Federation, and World Heart Federation).
The UN High-Level Meeting (UN HLM) on NCDs in September 2011 is expected to focus the world’s attention on NCDs in the same way that a similar meeting did for HIV/AIDS in 2001, which concluded that dealing with the disease was central to the world development agenda. The rising global epidemic of NCDs is now responsible for two-thirds of all deaths worldwide and has become a major barrier to development, according to Dr. Robert Beaglehole of the University of Aukland, New Zealand, and 43 coauthors representing the two umbrella groups (Lancet 2011 [doi:10.1016/S0140-6736(11)60393-0])
"The UN HLM is a turning point in the way we approach global health issues, and it will place NCDs on the development agenda. The global community has to take this opportunity and sustain the momentum to achieve the goal of avoiding premature NCD deaths and disability, thus improving global health in the years to come," the authors wrote.
The five priorities were chosen because there is good evidence for each regarding their substantial impact on health and cost effectiveness, the low cost of implementation, and financial feasibility for scale-up. Specifically, the authors proposed goals of reducing worldwide tobacco consumption to less than 5% by the year 2040 and salt consumption to less than 5g (2,000 mg sodium) per person per day by 2025. "We propose a goal to achieve a world essentially free from tobacco by 2040," they said.
Tobacco reduction would be accomplished via full implementation of the 2003 World Health Organization Framework Convention on Tobacco Control, which calls for reducing demand for tobacco via methods such as raising tobacco taxes, legislation of health warnings, and smoking prohibitions. Salt reduction would be accomplished via mass-media campaigns and voluntary reformulation of food products by industry.
Using those methods, the yearly cost to implement tobacco control and salt reduction would be about 20 cents per person per year in countries such as India and China, with the total package of priority interventions priced at about $9 billion per year, the authors said.
Keys to progress include leadership at the highest levels of government, a focus on prevention, treatment, international cooperation, monitoring, reporting and accountability, they said.
"An ideal outcome of the UN HLM will be a sustained commitment to a set of feasible actions and interventions for which specific and timed targets and indicators can be developed, and progress can be readily measured."
The NCD Alliance has also issued a proposed outcomes document for the UN High-Level meeting.
Dr. Beaglehole declared that he has no disclosures. Two of the authors declared financial relationships with pharmaceutical companies, and two others received grants from charities including the Wellcome Trust. One of the authors, Richard Horton, is editor of the Lancet. The others declared that they have no disclosures.
FROM THE LANCET
Near-Elimination of Tobacco Use Deemed a Priority by U.N. Body
Near-elimination of tobacco consumption worldwide by 2040 was proposed as the No. 1 priority for this fall’s United Nations High-Level Meeting on Non-Communicable Disease in a joint statement on April 6 from two stakeholder coalitions.
Dietary salt reduction, improved diet and physical activity, reduction in hazardous alcohol intake, and universal access to essential drugs and technologies were also listed as top global NCD priorities by the Lancet NCD Action Group, an informal collaboration of academics, practitioners, and civil society organizations, and the NCD Alliance, comprising four international nongovernmental organizations (Union for International Cancer Control, International Union Against Tuberculosis and Lung Disease, International Diabetes Federation, and World Heart Federation).
The UN High-Level Meeting (UN HLM) on NCDs in September 2011 is expected to focus the world’s attention on NCDs in the same way that a similar meeting did for HIV/AIDS in 2001, which concluded that dealing with the disease was central to the world development agenda. The rising global epidemic of NCDs is now responsible for two-thirds of all deaths worldwide and has become a major barrier to development, according to Dr. Robert Beaglehole of the University of Aukland, New Zealand, and 43 coauthors representing the two umbrella groups (Lancet 2011 [doi:10.1016/S0140-6736(11)60393-0])
"The UN HLM is a turning point in the way we approach global health issues, and it will place NCDs on the development agenda. The global community has to take this opportunity and sustain the momentum to achieve the goal of avoiding premature NCD deaths and disability, thus improving global health in the years to come," the authors wrote.
The five priorities were chosen because there is good evidence for each regarding their substantial impact on health and cost effectiveness, the low cost of implementation, and financial feasibility for scale-up. Specifically, the authors proposed goals of reducing worldwide tobacco consumption to less than 5% by the year 2040 and salt consumption to less than 5g (2,000 mg sodium) per person per day by 2025. "We propose a goal to achieve a world essentially free from tobacco by 2040," they said.
Tobacco reduction would be accomplished via full implementation of the 2003 World Health Organization Framework Convention on Tobacco Control, which calls for reducing demand for tobacco via methods such as raising tobacco taxes, legislation of health warnings, and smoking prohibitions. Salt reduction would be accomplished via mass-media campaigns and voluntary reformulation of food products by industry.
Using those methods, the yearly cost to implement tobacco control and salt reduction would be about 20 cents per person per year in countries such as India and China, with the total package of priority interventions priced at about $9 billion per year, the authors said.
Keys to progress include leadership at the highest levels of government, a focus on prevention, treatment, international cooperation, monitoring, reporting and accountability, they said.
"An ideal outcome of the UN HLM will be a sustained commitment to a set of feasible actions and interventions for which specific and timed targets and indicators can be developed, and progress can be readily measured."
The NCD Alliance has also issued a proposed outcomes document for the UN High-Level meeting.
Dr. Beaglehole declared that he has no disclosures. Two of the authors declared financial relationships with pharmaceutical companies, and two others received grants from charities including the Wellcome Trust. One of the authors, Richard Horton, is editor of the Lancet. The others declared that they have no disclosures.
Near-elimination of tobacco consumption worldwide by 2040 was proposed as the No. 1 priority for this fall’s United Nations High-Level Meeting on Non-Communicable Disease in a joint statement on April 6 from two stakeholder coalitions.
Dietary salt reduction, improved diet and physical activity, reduction in hazardous alcohol intake, and universal access to essential drugs and technologies were also listed as top global NCD priorities by the Lancet NCD Action Group, an informal collaboration of academics, practitioners, and civil society organizations, and the NCD Alliance, comprising four international nongovernmental organizations (Union for International Cancer Control, International Union Against Tuberculosis and Lung Disease, International Diabetes Federation, and World Heart Federation).
The UN High-Level Meeting (UN HLM) on NCDs in September 2011 is expected to focus the world’s attention on NCDs in the same way that a similar meeting did for HIV/AIDS in 2001, which concluded that dealing with the disease was central to the world development agenda. The rising global epidemic of NCDs is now responsible for two-thirds of all deaths worldwide and has become a major barrier to development, according to Dr. Robert Beaglehole of the University of Aukland, New Zealand, and 43 coauthors representing the two umbrella groups (Lancet 2011 [doi:10.1016/S0140-6736(11)60393-0])
"The UN HLM is a turning point in the way we approach global health issues, and it will place NCDs on the development agenda. The global community has to take this opportunity and sustain the momentum to achieve the goal of avoiding premature NCD deaths and disability, thus improving global health in the years to come," the authors wrote.
The five priorities were chosen because there is good evidence for each regarding their substantial impact on health and cost effectiveness, the low cost of implementation, and financial feasibility for scale-up. Specifically, the authors proposed goals of reducing worldwide tobacco consumption to less than 5% by the year 2040 and salt consumption to less than 5g (2,000 mg sodium) per person per day by 2025. "We propose a goal to achieve a world essentially free from tobacco by 2040," they said.
Tobacco reduction would be accomplished via full implementation of the 2003 World Health Organization Framework Convention on Tobacco Control, which calls for reducing demand for tobacco via methods such as raising tobacco taxes, legislation of health warnings, and smoking prohibitions. Salt reduction would be accomplished via mass-media campaigns and voluntary reformulation of food products by industry.
Using those methods, the yearly cost to implement tobacco control and salt reduction would be about 20 cents per person per year in countries such as India and China, with the total package of priority interventions priced at about $9 billion per year, the authors said.
Keys to progress include leadership at the highest levels of government, a focus on prevention, treatment, international cooperation, monitoring, reporting and accountability, they said.
"An ideal outcome of the UN HLM will be a sustained commitment to a set of feasible actions and interventions for which specific and timed targets and indicators can be developed, and progress can be readily measured."
The NCD Alliance has also issued a proposed outcomes document for the UN High-Level meeting.
Dr. Beaglehole declared that he has no disclosures. Two of the authors declared financial relationships with pharmaceutical companies, and two others received grants from charities including the Wellcome Trust. One of the authors, Richard Horton, is editor of the Lancet. The others declared that they have no disclosures.
Near-elimination of tobacco consumption worldwide by 2040 was proposed as the No. 1 priority for this fall’s United Nations High-Level Meeting on Non-Communicable Disease in a joint statement on April 6 from two stakeholder coalitions.
Dietary salt reduction, improved diet and physical activity, reduction in hazardous alcohol intake, and universal access to essential drugs and technologies were also listed as top global NCD priorities by the Lancet NCD Action Group, an informal collaboration of academics, practitioners, and civil society organizations, and the NCD Alliance, comprising four international nongovernmental organizations (Union for International Cancer Control, International Union Against Tuberculosis and Lung Disease, International Diabetes Federation, and World Heart Federation).
The UN High-Level Meeting (UN HLM) on NCDs in September 2011 is expected to focus the world’s attention on NCDs in the same way that a similar meeting did for HIV/AIDS in 2001, which concluded that dealing with the disease was central to the world development agenda. The rising global epidemic of NCDs is now responsible for two-thirds of all deaths worldwide and has become a major barrier to development, according to Dr. Robert Beaglehole of the University of Aukland, New Zealand, and 43 coauthors representing the two umbrella groups (Lancet 2011 [doi:10.1016/S0140-6736(11)60393-0])
"The UN HLM is a turning point in the way we approach global health issues, and it will place NCDs on the development agenda. The global community has to take this opportunity and sustain the momentum to achieve the goal of avoiding premature NCD deaths and disability, thus improving global health in the years to come," the authors wrote.
The five priorities were chosen because there is good evidence for each regarding their substantial impact on health and cost effectiveness, the low cost of implementation, and financial feasibility for scale-up. Specifically, the authors proposed goals of reducing worldwide tobacco consumption to less than 5% by the year 2040 and salt consumption to less than 5g (2,000 mg sodium) per person per day by 2025. "We propose a goal to achieve a world essentially free from tobacco by 2040," they said.
Tobacco reduction would be accomplished via full implementation of the 2003 World Health Organization Framework Convention on Tobacco Control, which calls for reducing demand for tobacco via methods such as raising tobacco taxes, legislation of health warnings, and smoking prohibitions. Salt reduction would be accomplished via mass-media campaigns and voluntary reformulation of food products by industry.
Using those methods, the yearly cost to implement tobacco control and salt reduction would be about 20 cents per person per year in countries such as India and China, with the total package of priority interventions priced at about $9 billion per year, the authors said.
Keys to progress include leadership at the highest levels of government, a focus on prevention, treatment, international cooperation, monitoring, reporting and accountability, they said.
"An ideal outcome of the UN HLM will be a sustained commitment to a set of feasible actions and interventions for which specific and timed targets and indicators can be developed, and progress can be readily measured."
The NCD Alliance has also issued a proposed outcomes document for the UN High-Level meeting.
Dr. Beaglehole declared that he has no disclosures. Two of the authors declared financial relationships with pharmaceutical companies, and two others received grants from charities including the Wellcome Trust. One of the authors, Richard Horton, is editor of the Lancet. The others declared that they have no disclosures.
FROM THE LANCET
New AACE Guidelines Emphasize Comprehensive Diabetes Management
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
FROM ENDOCRINE PRACTICE
New AACE Guidelines Emphasize Comprehensive Diabetes Management
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
FROM ENDOCRINE PRACTICE
New AACE Guidelines Emphasize Comprehensive Diabetes Management
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
New clinical practice guidelines from the American Association of Clinical Endocrinologists address the development of a diabetes comprehensive care plan.
Published April 5, the new clinical practice guideline (CPG) is intended to complement and extend existing guidelines available in the literature, including previous CPGs from AACE. The new document advocates a comprehensive approach to routine diabetes management, based on evidence that, although glycemia control variables such as hemoglobin A1c and glucose excursions have an impact on cardiovascular disease risk, other factors – including obesity, blood pressure, dyslipidemia, and hypercoagulation – also play important roles (Endocr. Pract. 2011;17 [suppl2]).
"Our goals for these guidelines are to provide the health care professional with tools to develop a comprehensive care plan for the prevention and management of diabetes and its complications, addressing not just hyperglycemia, but all associated cardiovascular risk factors," writing committee cochair and AACE president-elect Yehuda Handelsman said in an interview.
The document advocates an individualized approach to treatment, with personalized goals based on duration of diabetes, comorbidities, longevity, and the ability to provide treatment safely. In addition to type 2 diabetes, the CPG also addresses the care of individuals with prediabetes, type 1 diabetes, children/adolescents and pregnant women with diabetes, and hospitalized patients with hyperglycemia.
Controversies in diabetes care, such as new diagnostic criteria of diabetes, medication, and surgical treatment are also included, as are new technologies such as insulin pumps and continuous glucose monitoring. Less familiar areas such as sleep and breathing disturbances and depression are also addressed.
The document is divided into an executive summary containing the recommendations in a question-and-answer format, and an appendix providing the respective supporting evidence for each recommendation.
According to Dr. Handelsman, medical director and principal investigator of the Metabolic Institute of America, Tarzana, Calif., "These state-of-the-art guidelines provide the most up to date evidence-based answers to real-life questions to enable the health care provider to deliver the most relevant, individualized treatment plan for patients with diabetes."
Dr. Handelsman reported that he has received speakers bureau honoraria from AstraZeneca, Bristol-Myers Squibb/AstraZeneca, Daiichi Sankyo, GlaxoSmithKline, Merck & Co., and Novartis AG; consultant honoraria from BMS/AstraZeneca, Daiichi Sankyo, Gilead, Genentech, GSK, Merck, XOMA, Tethys Bioscience, and Tolerx; and research grant support from Daiichi Sankyo, GSK, Novartis, Novo Nordisk A/S, Takeda Pharmaceuticals, Sanofi-Aventis U.S., XOMA, and Tolerx. The CPG coauthors reported similar relationships.
FROM ENDOCRINE PRACTICE
Novel Device Uses Cold Therapy to Reduce Forehead Wrinkles
GRAPEVINE, TEX. – An investigational cryoprobe device reduced dynamic forehead wrinkles immediately after application in a pilot study of 36 patients that was presented at the annual meeting of the American Society for Laser Medicine and Surgery.
The self-contained, hand-held device, made by MyoScience Inc., uses a 27-gauge needle to deliver cooling to the temporal branch of the facial nerve that controls the frontalis muscle, rather than directly into the muscle as is done with botulinum toxins. The duration of effect is similar to that of botulinum toxins, about 3-4 months.
"Basically, it's a green alternative. There's no chemical, no toxin, it's only cold therapy," principal investigator Dr. Francis Palmer said in an interview.
Results from the use of a first-generation version of the device were reported by Dr. Vic Narurkar, who directs the Bay Area Laser Institute in San Francisco. The 36 patients were aged 35-70 years, all with forehead lines during animation. Some were at risk for brow ptosis.
After lidocaine administration, the probe was inserted for 30-45 seconds, and the subsequent reduction in muscle contractility was monitored to see if additional cryoprobe insertions were needed. All patients had immediate dynamic line reduction after two to four cryoprobe insertions, Dr. Narurkar reported.
The most common adverse events were headaches and small focal areas of minor epidermal cold injury, attributed to limitations in the prototype design. No patient experienced ptosis. "Cold has the unique natural property of selectively affecting the axon, yet preserves the endoneurial sheath. ... [It] has no impact on collagen-based structures," Dr. Narurkar explained.
According to Dr. Palmer, both the absence of ptosis risk and the immediacy of the response represent significant advantages over botulinum toxins, for which peak of action is typically 4-7 days following injection.
However, "I don't see this as a replacement for toxin, but as a complement. Physicians now have something to give patients who don't want a toxin, or for use in combination in areas where you can’t use a toxin. Botox is fabulous. It doesn't need to be replaced," said Dr. Palmer, director of the Beverly Hills (Calif.) International Center for Aesthetic Surgery.
Moreover, he noted, at this point, the injection of neurotoxin is easier and somewhat less invasive, although that could change with further revision of the cryoprobe's design. "We think it offers additional things toxin therapy doesn't offer, but it’s in a very early stage. It will be refined further before it hits the market."
MyoScience plans to seek a CE marking for the cryoprobe later this year before approaching the U.S. Food and Drug Administration, he said.
Both Dr. Palmer and Dr. Narurkar are paid consultants for MyoScience, which funded the study.
GRAPEVINE, TEX. – An investigational cryoprobe device reduced dynamic forehead wrinkles immediately after application in a pilot study of 36 patients that was presented at the annual meeting of the American Society for Laser Medicine and Surgery.
The self-contained, hand-held device, made by MyoScience Inc., uses a 27-gauge needle to deliver cooling to the temporal branch of the facial nerve that controls the frontalis muscle, rather than directly into the muscle as is done with botulinum toxins. The duration of effect is similar to that of botulinum toxins, about 3-4 months.
"Basically, it's a green alternative. There's no chemical, no toxin, it's only cold therapy," principal investigator Dr. Francis Palmer said in an interview.
Results from the use of a first-generation version of the device were reported by Dr. Vic Narurkar, who directs the Bay Area Laser Institute in San Francisco. The 36 patients were aged 35-70 years, all with forehead lines during animation. Some were at risk for brow ptosis.
After lidocaine administration, the probe was inserted for 30-45 seconds, and the subsequent reduction in muscle contractility was monitored to see if additional cryoprobe insertions were needed. All patients had immediate dynamic line reduction after two to four cryoprobe insertions, Dr. Narurkar reported.
The most common adverse events were headaches and small focal areas of minor epidermal cold injury, attributed to limitations in the prototype design. No patient experienced ptosis. "Cold has the unique natural property of selectively affecting the axon, yet preserves the endoneurial sheath. ... [It] has no impact on collagen-based structures," Dr. Narurkar explained.
According to Dr. Palmer, both the absence of ptosis risk and the immediacy of the response represent significant advantages over botulinum toxins, for which peak of action is typically 4-7 days following injection.
However, "I don't see this as a replacement for toxin, but as a complement. Physicians now have something to give patients who don't want a toxin, or for use in combination in areas where you can’t use a toxin. Botox is fabulous. It doesn't need to be replaced," said Dr. Palmer, director of the Beverly Hills (Calif.) International Center for Aesthetic Surgery.
Moreover, he noted, at this point, the injection of neurotoxin is easier and somewhat less invasive, although that could change with further revision of the cryoprobe's design. "We think it offers additional things toxin therapy doesn't offer, but it’s in a very early stage. It will be refined further before it hits the market."
MyoScience plans to seek a CE marking for the cryoprobe later this year before approaching the U.S. Food and Drug Administration, he said.
Both Dr. Palmer and Dr. Narurkar are paid consultants for MyoScience, which funded the study.
GRAPEVINE, TEX. – An investigational cryoprobe device reduced dynamic forehead wrinkles immediately after application in a pilot study of 36 patients that was presented at the annual meeting of the American Society for Laser Medicine and Surgery.
The self-contained, hand-held device, made by MyoScience Inc., uses a 27-gauge needle to deliver cooling to the temporal branch of the facial nerve that controls the frontalis muscle, rather than directly into the muscle as is done with botulinum toxins. The duration of effect is similar to that of botulinum toxins, about 3-4 months.
"Basically, it's a green alternative. There's no chemical, no toxin, it's only cold therapy," principal investigator Dr. Francis Palmer said in an interview.
Results from the use of a first-generation version of the device were reported by Dr. Vic Narurkar, who directs the Bay Area Laser Institute in San Francisco. The 36 patients were aged 35-70 years, all with forehead lines during animation. Some were at risk for brow ptosis.
After lidocaine administration, the probe was inserted for 30-45 seconds, and the subsequent reduction in muscle contractility was monitored to see if additional cryoprobe insertions were needed. All patients had immediate dynamic line reduction after two to four cryoprobe insertions, Dr. Narurkar reported.
The most common adverse events were headaches and small focal areas of minor epidermal cold injury, attributed to limitations in the prototype design. No patient experienced ptosis. "Cold has the unique natural property of selectively affecting the axon, yet preserves the endoneurial sheath. ... [It] has no impact on collagen-based structures," Dr. Narurkar explained.
According to Dr. Palmer, both the absence of ptosis risk and the immediacy of the response represent significant advantages over botulinum toxins, for which peak of action is typically 4-7 days following injection.
However, "I don't see this as a replacement for toxin, but as a complement. Physicians now have something to give patients who don't want a toxin, or for use in combination in areas where you can’t use a toxin. Botox is fabulous. It doesn't need to be replaced," said Dr. Palmer, director of the Beverly Hills (Calif.) International Center for Aesthetic Surgery.
Moreover, he noted, at this point, the injection of neurotoxin is easier and somewhat less invasive, although that could change with further revision of the cryoprobe's design. "We think it offers additional things toxin therapy doesn't offer, but it’s in a very early stage. It will be refined further before it hits the market."
MyoScience plans to seek a CE marking for the cryoprobe later this year before approaching the U.S. Food and Drug Administration, he said.
Both Dr. Palmer and Dr. Narurkar are paid consultants for MyoScience, which funded the study.
FROM THE ANNUAL MEETING OF THE AMERICAN SOCIETY FOR LASER MEDICINE AND SURGERY
Major Finding: All patients experienced immediate reduction in wrinkles with two to four cryoprobe insertions.
Data Source: Pilot study of 36 adults (aged 35-70 years) with dynamic forehead wrinkles.
Disclosures: Both Dr. Palmer and Dr. Narurkar are paid consultants for MyoScience, which funded the study.