User login
Top Avoidable Tests in Emergency Medicine
Imaging studies take four of the five slots in a newly unveiled list of unnecessary tests and procedures commonly performed in the emergency department, with CT scans in low-risk trauma cases earning particular censure.
But it’s how the emergency medicine list’s creators developed it that may prove most valuable in future efforts to reduce unnecessary tests throughout medicine.
Several specialty societies have developed Top-5 lists of avoidable tests or procedures to comply with the American Board of Internal Medicine’s Choosing Wisely campaign. The lists are "a new idea to engage clinicians in resource stewardship and to address rising health care costs," said Dr. Jeremiah D. Schuur of the department of emergency medicine, Brigham and Women’s Hospital, Boston, and his associates (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed/2013.12688]).
To create the emergency medicine list, Dr. Jeremiah D. Schuur and his associates took the traditional Choosing Wisely approach, then added a consensus-based twist.
They convened a technical expert panel that included the chiefs of the two academic hospital emergency departments (EDs) and four community hospital EDs in their health care system, which covers more than 320,000 ED visits annually in eastern Massachusetts. The panel also included an emergency physician with expertise in affordability, another with expertise in diagnostic imaging, and a third with expertise in hospital admissions and transfers, as well as a chief resident in emergency medicine.
The panel devised a preliminary list of 64 "low-value clinical decisions that were under the control of emergency clinicians and were thought to have a potential for cost savings."
Then came the twist: A total of 283 frontline emergency clinicians from the health system’s six emergency departments then were invited to complete a Web-based survey tool measuring their opinions of the potential benefit or harm to patients if clinicians discontinued the top 17 potentially avoidable items on the list.
The panel reviewed results from the 174 clinicians who completed the survey, and it then distilled them into a list of the Top-5 unnecessary tests and procedures:
• Do not order CT of the cervical spine for trauma patients who do not meet the National Emergency X-ray Utilization Study (NEXUS) low-risk criteria or the Canadian C-Spine Rule.
• Do not order CT to diagnose pulmonary embolism without first risk stratifying for pulmonary embolism (pretest probability and D-dimer tests if low probability).
• Do not order MRI of the lumbar spine for patients with low-back pain without high-risk features.
• Do not order CT of the head for patients with mild traumatic head injury who do not meet New Orleans Criteria or Canadian CT Head Rule.
• Do not order coagulation studies for patients without hemorrhage or suspected coagulopathy (for example, with anticoagulation therapy or clinical coagulopathy).
Every item on this list "received similar ratings by different groups of ED clinicians, including physicians and midlevel practitioners, clinicians in academic and community-hospital EDs, and practitioners with experience ranging from less than 3 years to more than 10 years," the authors explained. That suggests that emergency health care clinicians in other locations, as well as members of other specialty societies, also can achieve such consensus, Dr. Schuur and his associates noted.
Emergency medicine "is under immense pressure" to improve the value of services, they added.
"Some emergency physicians may be hesitant to embrace stewardship efforts, such as Choosing Wisely, for fear of losing autonomy and the medicolegal risk," the investigators noted. "However, if emergency physicians, who best understand the clinical evidence and unique needs of our patients, do not define measures of overuse for our specialty, others will."
The American College of Emergency Physicians has wrestled with the question of complying with the ABIM’s Choosing Wisely campaign and developing a Top-5 list, beginning with "a passionate floor debate" at the 2012 national ACEP Council meeting and including a dramatic reversal of the initial decision not to join the effort, said Dr. Deborah Grady, Dr. Rita F. Redberg, and Dr. William K. Mallon.
At least 50 specialty societies have now developed their Top-5 lists – but most haven’t disclosed their methods, and some clearly developed their lists without much input from frontline practitioners and without clear criteria. So, emergency medicine’s contribution to the Choosing Wisely campaign "is as much about the methodology ... as it is about the final recommendations," they noted.
"We hope the article by [Dr. Schuur and his colleagues] will stimulate other professional societies to adopt clear, transparent methods for developing and revising Top-5 lists with substantial input from practicing clinicians," the three physicians said.
Dr. Grady and Dr. Redberg are in the department of medicine at the University of California, San Francisco; Dr. Grady is also at the San Francisco Veterans Affairs Medical Center. Dr. Mallon is in the department of clinical emergency medicine at the University of Southern California, Los Angeles. They reported no potential conflicts of interest. These remarks were taken from their editorial accompanying Dr. Schuur’s report (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed.2013.8272]).
The American College of Emergency Physicians has wrestled with the question of complying with the ABIM’s Choosing Wisely campaign and developing a Top-5 list, beginning with "a passionate floor debate" at the 2012 national ACEP Council meeting and including a dramatic reversal of the initial decision not to join the effort, said Dr. Deborah Grady, Dr. Rita F. Redberg, and Dr. William K. Mallon.
At least 50 specialty societies have now developed their Top-5 lists – but most haven’t disclosed their methods, and some clearly developed their lists without much input from frontline practitioners and without clear criteria. So, emergency medicine’s contribution to the Choosing Wisely campaign "is as much about the methodology ... as it is about the final recommendations," they noted.
"We hope the article by [Dr. Schuur and his colleagues] will stimulate other professional societies to adopt clear, transparent methods for developing and revising Top-5 lists with substantial input from practicing clinicians," the three physicians said.
Dr. Grady and Dr. Redberg are in the department of medicine at the University of California, San Francisco; Dr. Grady is also at the San Francisco Veterans Affairs Medical Center. Dr. Mallon is in the department of clinical emergency medicine at the University of Southern California, Los Angeles. They reported no potential conflicts of interest. These remarks were taken from their editorial accompanying Dr. Schuur’s report (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed.2013.8272]).
The American College of Emergency Physicians has wrestled with the question of complying with the ABIM’s Choosing Wisely campaign and developing a Top-5 list, beginning with "a passionate floor debate" at the 2012 national ACEP Council meeting and including a dramatic reversal of the initial decision not to join the effort, said Dr. Deborah Grady, Dr. Rita F. Redberg, and Dr. William K. Mallon.
At least 50 specialty societies have now developed their Top-5 lists – but most haven’t disclosed their methods, and some clearly developed their lists without much input from frontline practitioners and without clear criteria. So, emergency medicine’s contribution to the Choosing Wisely campaign "is as much about the methodology ... as it is about the final recommendations," they noted.
"We hope the article by [Dr. Schuur and his colleagues] will stimulate other professional societies to adopt clear, transparent methods for developing and revising Top-5 lists with substantial input from practicing clinicians," the three physicians said.
Dr. Grady and Dr. Redberg are in the department of medicine at the University of California, San Francisco; Dr. Grady is also at the San Francisco Veterans Affairs Medical Center. Dr. Mallon is in the department of clinical emergency medicine at the University of Southern California, Los Angeles. They reported no potential conflicts of interest. These remarks were taken from their editorial accompanying Dr. Schuur’s report (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed.2013.8272]).
Imaging studies take four of the five slots in a newly unveiled list of unnecessary tests and procedures commonly performed in the emergency department, with CT scans in low-risk trauma cases earning particular censure.
But it’s how the emergency medicine list’s creators developed it that may prove most valuable in future efforts to reduce unnecessary tests throughout medicine.
Several specialty societies have developed Top-5 lists of avoidable tests or procedures to comply with the American Board of Internal Medicine’s Choosing Wisely campaign. The lists are "a new idea to engage clinicians in resource stewardship and to address rising health care costs," said Dr. Jeremiah D. Schuur of the department of emergency medicine, Brigham and Women’s Hospital, Boston, and his associates (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed/2013.12688]).
To create the emergency medicine list, Dr. Jeremiah D. Schuur and his associates took the traditional Choosing Wisely approach, then added a consensus-based twist.
They convened a technical expert panel that included the chiefs of the two academic hospital emergency departments (EDs) and four community hospital EDs in their health care system, which covers more than 320,000 ED visits annually in eastern Massachusetts. The panel also included an emergency physician with expertise in affordability, another with expertise in diagnostic imaging, and a third with expertise in hospital admissions and transfers, as well as a chief resident in emergency medicine.
The panel devised a preliminary list of 64 "low-value clinical decisions that were under the control of emergency clinicians and were thought to have a potential for cost savings."
Then came the twist: A total of 283 frontline emergency clinicians from the health system’s six emergency departments then were invited to complete a Web-based survey tool measuring their opinions of the potential benefit or harm to patients if clinicians discontinued the top 17 potentially avoidable items on the list.
The panel reviewed results from the 174 clinicians who completed the survey, and it then distilled them into a list of the Top-5 unnecessary tests and procedures:
• Do not order CT of the cervical spine for trauma patients who do not meet the National Emergency X-ray Utilization Study (NEXUS) low-risk criteria or the Canadian C-Spine Rule.
• Do not order CT to diagnose pulmonary embolism without first risk stratifying for pulmonary embolism (pretest probability and D-dimer tests if low probability).
• Do not order MRI of the lumbar spine for patients with low-back pain without high-risk features.
• Do not order CT of the head for patients with mild traumatic head injury who do not meet New Orleans Criteria or Canadian CT Head Rule.
• Do not order coagulation studies for patients without hemorrhage or suspected coagulopathy (for example, with anticoagulation therapy or clinical coagulopathy).
Every item on this list "received similar ratings by different groups of ED clinicians, including physicians and midlevel practitioners, clinicians in academic and community-hospital EDs, and practitioners with experience ranging from less than 3 years to more than 10 years," the authors explained. That suggests that emergency health care clinicians in other locations, as well as members of other specialty societies, also can achieve such consensus, Dr. Schuur and his associates noted.
Emergency medicine "is under immense pressure" to improve the value of services, they added.
"Some emergency physicians may be hesitant to embrace stewardship efforts, such as Choosing Wisely, for fear of losing autonomy and the medicolegal risk," the investigators noted. "However, if emergency physicians, who best understand the clinical evidence and unique needs of our patients, do not define measures of overuse for our specialty, others will."
Imaging studies take four of the five slots in a newly unveiled list of unnecessary tests and procedures commonly performed in the emergency department, with CT scans in low-risk trauma cases earning particular censure.
But it’s how the emergency medicine list’s creators developed it that may prove most valuable in future efforts to reduce unnecessary tests throughout medicine.
Several specialty societies have developed Top-5 lists of avoidable tests or procedures to comply with the American Board of Internal Medicine’s Choosing Wisely campaign. The lists are "a new idea to engage clinicians in resource stewardship and to address rising health care costs," said Dr. Jeremiah D. Schuur of the department of emergency medicine, Brigham and Women’s Hospital, Boston, and his associates (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed/2013.12688]).
To create the emergency medicine list, Dr. Jeremiah D. Schuur and his associates took the traditional Choosing Wisely approach, then added a consensus-based twist.
They convened a technical expert panel that included the chiefs of the two academic hospital emergency departments (EDs) and four community hospital EDs in their health care system, which covers more than 320,000 ED visits annually in eastern Massachusetts. The panel also included an emergency physician with expertise in affordability, another with expertise in diagnostic imaging, and a third with expertise in hospital admissions and transfers, as well as a chief resident in emergency medicine.
The panel devised a preliminary list of 64 "low-value clinical decisions that were under the control of emergency clinicians and were thought to have a potential for cost savings."
Then came the twist: A total of 283 frontline emergency clinicians from the health system’s six emergency departments then were invited to complete a Web-based survey tool measuring their opinions of the potential benefit or harm to patients if clinicians discontinued the top 17 potentially avoidable items on the list.
The panel reviewed results from the 174 clinicians who completed the survey, and it then distilled them into a list of the Top-5 unnecessary tests and procedures:
• Do not order CT of the cervical spine for trauma patients who do not meet the National Emergency X-ray Utilization Study (NEXUS) low-risk criteria or the Canadian C-Spine Rule.
• Do not order CT to diagnose pulmonary embolism without first risk stratifying for pulmonary embolism (pretest probability and D-dimer tests if low probability).
• Do not order MRI of the lumbar spine for patients with low-back pain without high-risk features.
• Do not order CT of the head for patients with mild traumatic head injury who do not meet New Orleans Criteria or Canadian CT Head Rule.
• Do not order coagulation studies for patients without hemorrhage or suspected coagulopathy (for example, with anticoagulation therapy or clinical coagulopathy).
Every item on this list "received similar ratings by different groups of ED clinicians, including physicians and midlevel practitioners, clinicians in academic and community-hospital EDs, and practitioners with experience ranging from less than 3 years to more than 10 years," the authors explained. That suggests that emergency health care clinicians in other locations, as well as members of other specialty societies, also can achieve such consensus, Dr. Schuur and his associates noted.
Emergency medicine "is under immense pressure" to improve the value of services, they added.
"Some emergency physicians may be hesitant to embrace stewardship efforts, such as Choosing Wisely, for fear of losing autonomy and the medicolegal risk," the investigators noted. "However, if emergency physicians, who best understand the clinical evidence and unique needs of our patients, do not define measures of overuse for our specialty, others will."
FROM JAMA INTERNAL MEDICINE
Imaging no-nos top list of avoidable tests in emergency medicine
But it’s how the emergency medicine list’s creators developed it that may prove most valuable in future efforts to reduce unnecessary tests throughout medicine.
Several specialty societies have developed Top-5 lists of avoidable tests or procedures to comply with the American Board of Internal Medicine’s Choosing Wisely campaign. The lists are "a new idea to engage clinicians in resource stewardship and to address rising health care costs," said Dr. Jeremiah D. Schuur of the department of emergency medicine, Brigham and Women’s Hospital, Boston, and his associates (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed/2013.12688]).
To create the emergency medicine list, Dr. Jeremiah D. Schuur and his associates took the traditional Choosing Wisely approach, then added a consensus-based twist.
They convened a technical expert panel that included the chiefs of the two academic hospital emergency departments (EDs) and four community hospital EDs in their health care system, which covers more than 320,000 ED visits annually in eastern Massachusetts. The panel also included an emergency physician with expertise in affordability, another with expertise in diagnostic imaging, and a third with expertise in hospital admissions and transfers, as well as a chief resident in emergency medicine.
The panel devised a preliminary list of 64 "low-value clinical decisions that were under the control of emergency clinicians and were thought to have a potential for cost savings."
Then came the twist: A total of 283 frontline emergency clinicians from the health system’s six emergency departments then were invited to complete a Web-based survey tool measuring their opinions of the potential benefit or harm to patients if clinicians discontinued the top 17 potentially avoidable items on the list.
The panel reviewed results from the 174 clinicians who completed the survey, and it then distilled them into a list of the Top-5 unnecessary tests and procedures:
• Do not order CT of the cervical spine for trauma patients who do not meet the National Emergency X-ray Utilization Study (NEXUS) low-risk criteria or the Canadian C-Spine Rule.
• Do not order CT to diagnose pulmonary embolism without first risk stratifying for pulmonary embolism (pretest probability and D-dimer tests if low probability).
• Do not order MRI of the lumbar spine for patients with low-back pain without high-risk features.
• Do not order CT of the head for patients with mild traumatic head injury who do not meet New Orleans Criteria or Canadian CT Head Rule.
• Do not order coagulation studies for patients without hemorrhage or suspected coagulopathy (for example, with anticoagulation therapy or clinical coagulopathy).
Every item on this list "received similar ratings by different groups of ED clinicians, including physicians and midlevel practitioners, clinicians in academic and community-hospital EDs, and practitioners with experience ranging from less than 3 years to more than 10 years," the authors explained. That suggests that emergency health care clinicians in other locations, as well as members of other specialty societies, also can achieve such consensus, Dr. Schuur and his associates noted.
Emergency medicine "is under immense pressure" to improve the value of services, they added.
"Some emergency physicians may be hesitant to embrace stewardship efforts, such as Choosing Wisely, for fear of losing autonomy and the medicolegal risk," the investigators noted. "However, if emergency physicians, who best understand the clinical evidence and unique needs of our patients, do not define measures of overuse for our specialty, others will."
The American College of Emergency Physicians has wrestled with the question of complying with the ABIM’s Choosing Wisely campaign and developing a Top-5 list, beginning with "a passionate floor debate" at the 2012 national ACEP Council meeting and including a dramatic reversal of the initial decision not to join the effort, said Dr. Deborah Grady, Dr. Rita F. Redberg, and Dr. William K. Mallon.
At least 50 specialty societies have now developed their Top-5 lists – but most haven’t disclosed their methods, and some clearly developed their lists without much input from frontline practitioners and without clear criteria. So, emergency medicine’s contribution to the Choosing Wisely campaign "is as much about the methodology ... as it is about the final recommendations," they noted.
"We hope the article by [Dr. Schuur and his colleagues] will stimulate other professional societies to adopt clear, transparent methods for developing and revising Top-5 lists with substantial input from practicing clinicians," the three physicians said.
Dr. Grady and Dr. Redberg are in the department of medicine at the University of California, San Francisco; Dr. Grady is also at the San Francisco Veterans Affairs Medical Center. Dr. Mallon is in the department of clinical emergency medicine at the University of Southern California, Los Angeles. They reported no potential conflicts of interest. These remarks were taken from their editorial accompanying Dr. Schuur’s report (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed.2013.8272]).
The American College of Emergency Physicians has wrestled with the question of complying with the ABIM’s Choosing Wisely campaign and developing a Top-5 list, beginning with "a passionate floor debate" at the 2012 national ACEP Council meeting and including a dramatic reversal of the initial decision not to join the effort, said Dr. Deborah Grady, Dr. Rita F. Redberg, and Dr. William K. Mallon.
At least 50 specialty societies have now developed their Top-5 lists – but most haven’t disclosed their methods, and some clearly developed their lists without much input from frontline practitioners and without clear criteria. So, emergency medicine’s contribution to the Choosing Wisely campaign "is as much about the methodology ... as it is about the final recommendations," they noted.
"We hope the article by [Dr. Schuur and his colleagues] will stimulate other professional societies to adopt clear, transparent methods for developing and revising Top-5 lists with substantial input from practicing clinicians," the three physicians said.
Dr. Grady and Dr. Redberg are in the department of medicine at the University of California, San Francisco; Dr. Grady is also at the San Francisco Veterans Affairs Medical Center. Dr. Mallon is in the department of clinical emergency medicine at the University of Southern California, Los Angeles. They reported no potential conflicts of interest. These remarks were taken from their editorial accompanying Dr. Schuur’s report (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed.2013.8272]).
The American College of Emergency Physicians has wrestled with the question of complying with the ABIM’s Choosing Wisely campaign and developing a Top-5 list, beginning with "a passionate floor debate" at the 2012 national ACEP Council meeting and including a dramatic reversal of the initial decision not to join the effort, said Dr. Deborah Grady, Dr. Rita F. Redberg, and Dr. William K. Mallon.
At least 50 specialty societies have now developed their Top-5 lists – but most haven’t disclosed their methods, and some clearly developed their lists without much input from frontline practitioners and without clear criteria. So, emergency medicine’s contribution to the Choosing Wisely campaign "is as much about the methodology ... as it is about the final recommendations," they noted.
"We hope the article by [Dr. Schuur and his colleagues] will stimulate other professional societies to adopt clear, transparent methods for developing and revising Top-5 lists with substantial input from practicing clinicians," the three physicians said.
Dr. Grady and Dr. Redberg are in the department of medicine at the University of California, San Francisco; Dr. Grady is also at the San Francisco Veterans Affairs Medical Center. Dr. Mallon is in the department of clinical emergency medicine at the University of Southern California, Los Angeles. They reported no potential conflicts of interest. These remarks were taken from their editorial accompanying Dr. Schuur’s report (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed.2013.8272]).
But it’s how the emergency medicine list’s creators developed it that may prove most valuable in future efforts to reduce unnecessary tests throughout medicine.
Several specialty societies have developed Top-5 lists of avoidable tests or procedures to comply with the American Board of Internal Medicine’s Choosing Wisely campaign. The lists are "a new idea to engage clinicians in resource stewardship and to address rising health care costs," said Dr. Jeremiah D. Schuur of the department of emergency medicine, Brigham and Women’s Hospital, Boston, and his associates (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed/2013.12688]).
To create the emergency medicine list, Dr. Jeremiah D. Schuur and his associates took the traditional Choosing Wisely approach, then added a consensus-based twist.
They convened a technical expert panel that included the chiefs of the two academic hospital emergency departments (EDs) and four community hospital EDs in their health care system, which covers more than 320,000 ED visits annually in eastern Massachusetts. The panel also included an emergency physician with expertise in affordability, another with expertise in diagnostic imaging, and a third with expertise in hospital admissions and transfers, as well as a chief resident in emergency medicine.
The panel devised a preliminary list of 64 "low-value clinical decisions that were under the control of emergency clinicians and were thought to have a potential for cost savings."
Then came the twist: A total of 283 frontline emergency clinicians from the health system’s six emergency departments then were invited to complete a Web-based survey tool measuring their opinions of the potential benefit or harm to patients if clinicians discontinued the top 17 potentially avoidable items on the list.
The panel reviewed results from the 174 clinicians who completed the survey, and it then distilled them into a list of the Top-5 unnecessary tests and procedures:
• Do not order CT of the cervical spine for trauma patients who do not meet the National Emergency X-ray Utilization Study (NEXUS) low-risk criteria or the Canadian C-Spine Rule.
• Do not order CT to diagnose pulmonary embolism without first risk stratifying for pulmonary embolism (pretest probability and D-dimer tests if low probability).
• Do not order MRI of the lumbar spine for patients with low-back pain without high-risk features.
• Do not order CT of the head for patients with mild traumatic head injury who do not meet New Orleans Criteria or Canadian CT Head Rule.
• Do not order coagulation studies for patients without hemorrhage or suspected coagulopathy (for example, with anticoagulation therapy or clinical coagulopathy).
Every item on this list "received similar ratings by different groups of ED clinicians, including physicians and midlevel practitioners, clinicians in academic and community-hospital EDs, and practitioners with experience ranging from less than 3 years to more than 10 years," the authors explained. That suggests that emergency health care clinicians in other locations, as well as members of other specialty societies, also can achieve such consensus, Dr. Schuur and his associates noted.
Emergency medicine "is under immense pressure" to improve the value of services, they added.
"Some emergency physicians may be hesitant to embrace stewardship efforts, such as Choosing Wisely, for fear of losing autonomy and the medicolegal risk," the investigators noted. "However, if emergency physicians, who best understand the clinical evidence and unique needs of our patients, do not define measures of overuse for our specialty, others will."
But it’s how the emergency medicine list’s creators developed it that may prove most valuable in future efforts to reduce unnecessary tests throughout medicine.
Several specialty societies have developed Top-5 lists of avoidable tests or procedures to comply with the American Board of Internal Medicine’s Choosing Wisely campaign. The lists are "a new idea to engage clinicians in resource stewardship and to address rising health care costs," said Dr. Jeremiah D. Schuur of the department of emergency medicine, Brigham and Women’s Hospital, Boston, and his associates (JAMA Intern. Med. 2014 [doi:10.1001/jamainternmed/2013.12688]).
To create the emergency medicine list, Dr. Jeremiah D. Schuur and his associates took the traditional Choosing Wisely approach, then added a consensus-based twist.
They convened a technical expert panel that included the chiefs of the two academic hospital emergency departments (EDs) and four community hospital EDs in their health care system, which covers more than 320,000 ED visits annually in eastern Massachusetts. The panel also included an emergency physician with expertise in affordability, another with expertise in diagnostic imaging, and a third with expertise in hospital admissions and transfers, as well as a chief resident in emergency medicine.
The panel devised a preliminary list of 64 "low-value clinical decisions that were under the control of emergency clinicians and were thought to have a potential for cost savings."
Then came the twist: A total of 283 frontline emergency clinicians from the health system’s six emergency departments then were invited to complete a Web-based survey tool measuring their opinions of the potential benefit or harm to patients if clinicians discontinued the top 17 potentially avoidable items on the list.
The panel reviewed results from the 174 clinicians who completed the survey, and it then distilled them into a list of the Top-5 unnecessary tests and procedures:
• Do not order CT of the cervical spine for trauma patients who do not meet the National Emergency X-ray Utilization Study (NEXUS) low-risk criteria or the Canadian C-Spine Rule.
• Do not order CT to diagnose pulmonary embolism without first risk stratifying for pulmonary embolism (pretest probability and D-dimer tests if low probability).
• Do not order MRI of the lumbar spine for patients with low-back pain without high-risk features.
• Do not order CT of the head for patients with mild traumatic head injury who do not meet New Orleans Criteria or Canadian CT Head Rule.
• Do not order coagulation studies for patients without hemorrhage or suspected coagulopathy (for example, with anticoagulation therapy or clinical coagulopathy).
Every item on this list "received similar ratings by different groups of ED clinicians, including physicians and midlevel practitioners, clinicians in academic and community-hospital EDs, and practitioners with experience ranging from less than 3 years to more than 10 years," the authors explained. That suggests that emergency health care clinicians in other locations, as well as members of other specialty societies, also can achieve such consensus, Dr. Schuur and his associates noted.
Emergency medicine "is under immense pressure" to improve the value of services, they added.
"Some emergency physicians may be hesitant to embrace stewardship efforts, such as Choosing Wisely, for fear of losing autonomy and the medicolegal risk," the investigators noted. "However, if emergency physicians, who best understand the clinical evidence and unique needs of our patients, do not define measures of overuse for our specialty, others will."
FROM JAMA INTERNAL MEDICINE
Varenicline plus CBT keeps tobacco at bay
Patients with serious mental illness who quit smoking with a standard 12-week course of varenicline and cognitive-behavioral therapy are three times more likely to maintain that abstinence if they take a maintenance dose of the drug than if they discontinue it,according to a report publishedonline Jan. 7 in JAMA.
In what the researchers described as the first randomized controlled clinical trial of maintenance pharmacotherapy aimed at preventing smoking relapse in people with serious mental illness, the prevalence of smoking abstinence after 1 year was 60% (24 of 40 study participants) in patients assigned to maintenance varenicline, compared with 19% (9 of 47 participants) in those assigned to placebo.
"Such maintenance treatment may reduce the high prevalence of tobacco dependence and reduce the heavy burden of smoking-related morbidity and mortality in those with serious mental illness," Dr. A. Eden Evins of Massachusetts General Hospital and Harvard Medical School, Boston, and her associates wrote in JAMA.
The open-label study, released in advance of the 50th anniversary of the Surgeon General’s Report on Smoking and Health, involved 203 adults with schizophrenia spectrum disorder (185 patients) or bipolar disorder (18 patients) who reported smoking 10 or more cigarettes per day and whose expired carbon monoxide levels were higher than 9 ppm at baseline. All were outpatients at 10 community health centers in Massachusetts, Michigan, New Hampshire, Indiana, Alabama, or Minnesota, and all were taking stable doses of antipsychotic or mood- stabilizing medication.
A total of 87 of these participants successfully completed a 12-week smoking cessation program combining up to 1.0 mg of varenicline twice daily plus weekly 1-hour group cognitive-behavioral therapy (CBT) sessions. They were randomly assigned to continue for another 40 weeks with either CBT plus 1.0 mg of varenicline twice daily (40 patients) or CBT plus matching placebo (47 patients).
The CBT, which focused on relapse-prevention skills, was tapered from weekly to monthly sessions, for a total of 15 sessions during the 40 weeks.
At 52 weeks from baseline, smoking cessation treatment was stopped, and the 59 patients remaining in the study were followed through week 64 for biochemically verified smoking abstinence and safety outcomes.
At week 52, 24 of 40 participants taking maintenance varenicline (60%) were still abstaining from smoking, compared with only 9 of the 47 patients (19%) taking placebo, for an odds ratio of 6.2. At week 64, 18 participants (45%) in the varenicline group were still abstaining from smoking, compared with only 6 (13%) in the placebo group, for an odds ratio of 5.1, Dr. Evins and her associates reported (JAMA 2014 Jan. 7 [doi: 10.1001/jama.2013.285113]).
During treatment and follow-up, the two study groups showed no differences in the severity of their psychiatric symptoms, nicotine withdrawal symptoms, or self-reported overall health. No serious adverse events were attributed to the study medication.
However, because of the small sample size and the high dropout rate of the study, "it is not possible to accurately estimate the risk of serious adverse effects or to make claims regarding safety," investigators noted.
This study was funded by the National Institute on Drug Abuse andby Pfizer. Pfizer also supplied the study medication and provided other support. Dr. Evins and her associates reported ties to Pfizer.
Dr. Vera DePalo, FCCP, comments: Tobacco use is a risk factor for the development of several pathologies. Cessation is particularly difficult in patients with serious mental illness.In this study, the authors have been able to demonstrate that maintenance therapy with varenicline along with cognitive-behavioral therapy was able to extend the smoking-free period in behavioral health patients. This study holds promise for success in smoking cessation. While no serious adverse events were attributed to the medication, the authors point out, with the small sample size and the study drop out rate, more investigation is needed to accurately make claims about safety.
Patients with serious mental illness who quit smoking with a standard 12-week course of varenicline and cognitive-behavioral therapy are three times more likely to maintain that abstinence if they take a maintenance dose of the drug than if they discontinue it,according to a report publishedonline Jan. 7 in JAMA.
In what the researchers described as the first randomized controlled clinical trial of maintenance pharmacotherapy aimed at preventing smoking relapse in people with serious mental illness, the prevalence of smoking abstinence after 1 year was 60% (24 of 40 study participants) in patients assigned to maintenance varenicline, compared with 19% (9 of 47 participants) in those assigned to placebo.
"Such maintenance treatment may reduce the high prevalence of tobacco dependence and reduce the heavy burden of smoking-related morbidity and mortality in those with serious mental illness," Dr. A. Eden Evins of Massachusetts General Hospital and Harvard Medical School, Boston, and her associates wrote in JAMA.
The open-label study, released in advance of the 50th anniversary of the Surgeon General’s Report on Smoking and Health, involved 203 adults with schizophrenia spectrum disorder (185 patients) or bipolar disorder (18 patients) who reported smoking 10 or more cigarettes per day and whose expired carbon monoxide levels were higher than 9 ppm at baseline. All were outpatients at 10 community health centers in Massachusetts, Michigan, New Hampshire, Indiana, Alabama, or Minnesota, and all were taking stable doses of antipsychotic or mood- stabilizing medication.
A total of 87 of these participants successfully completed a 12-week smoking cessation program combining up to 1.0 mg of varenicline twice daily plus weekly 1-hour group cognitive-behavioral therapy (CBT) sessions. They were randomly assigned to continue for another 40 weeks with either CBT plus 1.0 mg of varenicline twice daily (40 patients) or CBT plus matching placebo (47 patients).
The CBT, which focused on relapse-prevention skills, was tapered from weekly to monthly sessions, for a total of 15 sessions during the 40 weeks.
At 52 weeks from baseline, smoking cessation treatment was stopped, and the 59 patients remaining in the study were followed through week 64 for biochemically verified smoking abstinence and safety outcomes.
At week 52, 24 of 40 participants taking maintenance varenicline (60%) were still abstaining from smoking, compared with only 9 of the 47 patients (19%) taking placebo, for an odds ratio of 6.2. At week 64, 18 participants (45%) in the varenicline group were still abstaining from smoking, compared with only 6 (13%) in the placebo group, for an odds ratio of 5.1, Dr. Evins and her associates reported (JAMA 2014 Jan. 7 [doi: 10.1001/jama.2013.285113]).
During treatment and follow-up, the two study groups showed no differences in the severity of their psychiatric symptoms, nicotine withdrawal symptoms, or self-reported overall health. No serious adverse events were attributed to the study medication.
However, because of the small sample size and the high dropout rate of the study, "it is not possible to accurately estimate the risk of serious adverse effects or to make claims regarding safety," investigators noted.
This study was funded by the National Institute on Drug Abuse andby Pfizer. Pfizer also supplied the study medication and provided other support. Dr. Evins and her associates reported ties to Pfizer.
Dr. Vera DePalo, FCCP, comments: Tobacco use is a risk factor for the development of several pathologies. Cessation is particularly difficult in patients with serious mental illness.In this study, the authors have been able to demonstrate that maintenance therapy with varenicline along with cognitive-behavioral therapy was able to extend the smoking-free period in behavioral health patients. This study holds promise for success in smoking cessation. While no serious adverse events were attributed to the medication, the authors point out, with the small sample size and the study drop out rate, more investigation is needed to accurately make claims about safety.
Patients with serious mental illness who quit smoking with a standard 12-week course of varenicline and cognitive-behavioral therapy are three times more likely to maintain that abstinence if they take a maintenance dose of the drug than if they discontinue it,according to a report publishedonline Jan. 7 in JAMA.
In what the researchers described as the first randomized controlled clinical trial of maintenance pharmacotherapy aimed at preventing smoking relapse in people with serious mental illness, the prevalence of smoking abstinence after 1 year was 60% (24 of 40 study participants) in patients assigned to maintenance varenicline, compared with 19% (9 of 47 participants) in those assigned to placebo.
"Such maintenance treatment may reduce the high prevalence of tobacco dependence and reduce the heavy burden of smoking-related morbidity and mortality in those with serious mental illness," Dr. A. Eden Evins of Massachusetts General Hospital and Harvard Medical School, Boston, and her associates wrote in JAMA.
The open-label study, released in advance of the 50th anniversary of the Surgeon General’s Report on Smoking and Health, involved 203 adults with schizophrenia spectrum disorder (185 patients) or bipolar disorder (18 patients) who reported smoking 10 or more cigarettes per day and whose expired carbon monoxide levels were higher than 9 ppm at baseline. All were outpatients at 10 community health centers in Massachusetts, Michigan, New Hampshire, Indiana, Alabama, or Minnesota, and all were taking stable doses of antipsychotic or mood- stabilizing medication.
A total of 87 of these participants successfully completed a 12-week smoking cessation program combining up to 1.0 mg of varenicline twice daily plus weekly 1-hour group cognitive-behavioral therapy (CBT) sessions. They were randomly assigned to continue for another 40 weeks with either CBT plus 1.0 mg of varenicline twice daily (40 patients) or CBT plus matching placebo (47 patients).
The CBT, which focused on relapse-prevention skills, was tapered from weekly to monthly sessions, for a total of 15 sessions during the 40 weeks.
At 52 weeks from baseline, smoking cessation treatment was stopped, and the 59 patients remaining in the study were followed through week 64 for biochemically verified smoking abstinence and safety outcomes.
At week 52, 24 of 40 participants taking maintenance varenicline (60%) were still abstaining from smoking, compared with only 9 of the 47 patients (19%) taking placebo, for an odds ratio of 6.2. At week 64, 18 participants (45%) in the varenicline group were still abstaining from smoking, compared with only 6 (13%) in the placebo group, for an odds ratio of 5.1, Dr. Evins and her associates reported (JAMA 2014 Jan. 7 [doi: 10.1001/jama.2013.285113]).
During treatment and follow-up, the two study groups showed no differences in the severity of their psychiatric symptoms, nicotine withdrawal symptoms, or self-reported overall health. No serious adverse events were attributed to the study medication.
However, because of the small sample size and the high dropout rate of the study, "it is not possible to accurately estimate the risk of serious adverse effects or to make claims regarding safety," investigators noted.
This study was funded by the National Institute on Drug Abuse andby Pfizer. Pfizer also supplied the study medication and provided other support. Dr. Evins and her associates reported ties to Pfizer.
Dr. Vera DePalo, FCCP, comments: Tobacco use is a risk factor for the development of several pathologies. Cessation is particularly difficult in patients with serious mental illness.In this study, the authors have been able to demonstrate that maintenance therapy with varenicline along with cognitive-behavioral therapy was able to extend the smoking-free period in behavioral health patients. This study holds promise for success in smoking cessation. While no serious adverse events were attributed to the medication, the authors point out, with the small sample size and the study drop out rate, more investigation is needed to accurately make claims about safety.
Major finding: At week 52, 24 of 40 participants taking maintenance varenicline (60%) were still abstaining from smoking, compared with only 9 of the 47 patients (19%) taking placebo; and at week 64, 18 participants (45%) in the varenicline group were still abstaining from smoking, compared with only 6 (13%) in the placebo group.
Data source: A randomized controlled clinical trial involving 87 patients who had schizophrenia spectrum or bipolar disorder and quit smoking after a 12-week program of CBT plus varenicline pharmacotherapy, who received either maintenance varenicline or placebo for an additional 40 weeks.
Disclosures: This study was funded by the National Institute on Drug Abuse and by Pfizer. Pfizer also supplied the study medication and provided other support. Dr. Evins reported ties to EnVivo Pharmaceuticals, GlaxoSmithKline, and Pfizer, and her associates reported ties to Publicis Healthcare Communications Group, Janssen, Otsuka, AssurEx, Eli Lilly, and Pfizer.
CPAP alternative? Implantable device cut OSA severity
An implantable device that stimulates the upper airway nerves and muscles produced long-term, clinically meaningful reductions in the severity of obstructive sleep apnea in an industry-sponsored study reported in the New England Journal of Medicine.
One year after implantation, patients showed a 68% reduction in scores on the Apnea-Hypopnea Index (AHI) and a 70% reduction in scores on the Oxygen Desaturation Index (ODI), as well as subjective improvements in daytime sleepiness and quality of life. The magnitude of these benefits was similar to that reported for continuous positive airway pressure (CPAP) therapy and superior to that reported for uvulopalatopharyngoplasty, said Dr. Patrick J. Strollo Jr., FCCP, of the department of otolaryngology, University of Pittsburgh Medical Center and his associates in the STAR (Stimulation Therapy for Apnea Reduction) trial group.
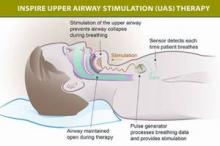
Upper airway stimulation using implanted electrodes to stimulate the hypoglossal nerve on one side of the neck "has been developed as a possible treatment option" for moderate to severe obstructive sleep apnea "and has shown promise in feasibility trials," the investigators noted.
For their study, designed in collaboration with the sponsor (Inspire Medical Systems) and the Food and Drug Administration, 126 patients who couldn’t tolerate CPAP therapy underwent surgical implantation of the device and were followed for 1 year. Otolaryngologists at 22 academic and private medical centers performed the surgery, which took a median of 140 minutes (range, 65-360 minutes).
Most (83%) of the participants were men. The mean age was 55 years (range, 31-80 years), and the mean body mass index was 28.4 kg/m2 (range, 18.4-32.5). Twenty-two of these patients (17%) had undergone uvulopalatopharyngoplasty, which had not corrected their obstructive sleep apnea.
The device includes a neurostimulator implanted in the intercostal muscles in the right mid-infraclavicular region, with one lead threaded upward inside the patient’s neck that is attached to three stimulation electrodes. The electrodes can be placed in a variety of configurations on the ipsilateral hypoglossal nerve, which, when stimulated, pushes the tongue forward and prevents the upper-airway muscles from collapsing and causing inspiratory flow obstruction.
The device also has a second, sensing lead tunneled between the internal and external intercostal muscles on the ipsilateral side to detect ventilatory effort during sleep, so that the stimulation of the hypoglossal nerve can be synchronized with the patient’s breathing.
The primary outcome of the study was the change in severity of obstructive sleep apnea, as measured by scores on the AHI and the ODI, at 12 months. The median AHI score decreased 68%, from 29.3 events per hour to 9.0 events per hour. The median ODI score dropped 70%, from 25.4 events per hour to 7.4 events <[lb]>per hour, the investigators said (N. Engl. J. Med. 2014 [doi:10.1056/<[lb]>NEJMoa1308659]).
Two-thirds of the participants showed a reduction of at least 50% in AHI score, and three-quarters showed a reduction of at least 25% in ODI score. And the median percentage of sleep time spent with poor oxygen saturation (less than 90%) declined from 5.4% to 0.9%.
In addition, patients’ scores on the Functional Outcomes of Sleep Questionnaire, which measures disease-specific quality of life, showed clinically meaningful improvement. And scores on the Epworth Sleepiness Scale normalized.
In the final, "challenge," phase of this study, the first 46 consecutive patients who had responded to this treatment at 1 year were randomly assigned to either continue it (turn the devices on at night) or to discontinue it (turn the devices off at night) for 1 more week. This challenge demonstrated that the improvements in OSA were in fact from the use of the hypoglossal-stimulation device, as sleep apnea relapsed in the patients who discontinued treatment.
There were no serious procedural complications, no rehospitalizations, and no infections. Two patients developed serious device-related adverse events, for an overall rate of less than 2%. In both cases, the device caused discomfort that was resolved by a second surgery to reposition it. Another 33 serious adverse events were considered to be unrelated to the implantation procedure or the device .
Nonserious adverse events – including sore throat from intubation during the procedure, pain at the incision sites, and muscle soreness – occurred in 88% of the study subjects. Nonserious events related to the device included discomfort during electrostimulation, reported by 40% of patients, and tongue soreness, reported by 21%. These resolved as the patients became acclimated to the device or after the device was reprogrammed to adjust the stimulation.
Twenty-three patients experienced temporary tongue weakness after the surgery, which resolved in all of them. Nine patients began using a tooth guard to resolve tongue soreness or abrasion.
"This approach may not be appropriate for persons with excessive airway collapsibility," Dr. Strollo and his associates cautioned. They screened potential study participants using endoscopy during drug-induced sleep, to identify functional upper-airway collapse originating in the retrolingual region, which would be the most amenable to neurostimulation at the base of the tongue.
The Food and Drug Administration’s Anesthesiology and Respiratory Therapy Devices Panel of the Medical Devices Advisory Committee will discuss, make recommendations, and vote on information related to the premarket approval application on Feb. 20.
Hypoglossal nerve stimulation appears effective
The findings by Strollo et al. give clinicians the rationale to consider hypoglossal nerve stimulation for selected patients who have trouble with CPAP therapy, said Dr. Atul Malhotra, FCCP.
Given the pathophysiology of OSA, a substantial proportion of patients would probably benefit from this treatment, even though symptoms were only reduced rather than completely eradicated. "Although the elimination of apnea would clearly be desirable, the observed reductions are probably similar to the benefits observed with CPAP, particularly when one considers the variability of adherence to CPAP therapy," he said.
Dr. Malhotra is in the division of pulmonary and critical care medicine at the University of California, San Diego. He reported previous ties to Philips Respironics, Apnex, and Apnicure. These remarks were taken from his editorial accompanying Dr. Strollo’s report (N. Engl. J. Med. 2014 [doi:10.1056/NEJMe1314084]).
An implantable device that stimulates the upper airway nerves and muscles produced long-term, clinically meaningful reductions in the severity of obstructive sleep apnea in an industry-sponsored study reported in the New England Journal of Medicine.
One year after implantation, patients showed a 68% reduction in scores on the Apnea-Hypopnea Index (AHI) and a 70% reduction in scores on the Oxygen Desaturation Index (ODI), as well as subjective improvements in daytime sleepiness and quality of life. The magnitude of these benefits was similar to that reported for continuous positive airway pressure (CPAP) therapy and superior to that reported for uvulopalatopharyngoplasty, said Dr. Patrick J. Strollo Jr., FCCP, of the department of otolaryngology, University of Pittsburgh Medical Center and his associates in the STAR (Stimulation Therapy for Apnea Reduction) trial group.
Upper airway stimulation using implanted electrodes to stimulate the hypoglossal nerve on one side of the neck "has been developed as a possible treatment option" for moderate to severe obstructive sleep apnea "and has shown promise in feasibility trials," the investigators noted.
For their study, designed in collaboration with the sponsor (Inspire Medical Systems) and the Food and Drug Administration, 126 patients who couldn’t tolerate CPAP therapy underwent surgical implantation of the device and were followed for 1 year. Otolaryngologists at 22 academic and private medical centers performed the surgery, which took a median of 140 minutes (range, 65-360 minutes).
Most (83%) of the participants were men. The mean age was 55 years (range, 31-80 years), and the mean body mass index was 28.4 kg/m2 (range, 18.4-32.5). Twenty-two of these patients (17%) had undergone uvulopalatopharyngoplasty, which had not corrected their obstructive sleep apnea.
The device includes a neurostimulator implanted in the intercostal muscles in the right mid-infraclavicular region, with one lead threaded upward inside the patient’s neck that is attached to three stimulation electrodes. The electrodes can be placed in a variety of configurations on the ipsilateral hypoglossal nerve, which, when stimulated, pushes the tongue forward and prevents the upper-airway muscles from collapsing and causing inspiratory flow obstruction.
The device also has a second, sensing lead tunneled between the internal and external intercostal muscles on the ipsilateral side to detect ventilatory effort during sleep, so that the stimulation of the hypoglossal nerve can be synchronized with the patient’s breathing.
The primary outcome of the study was the change in severity of obstructive sleep apnea, as measured by scores on the AHI and the ODI, at 12 months. The median AHI score decreased 68%, from 29.3 events per hour to 9.0 events per hour. The median ODI score dropped 70%, from 25.4 events per hour to 7.4 events <[lb]>per hour, the investigators said (N. Engl. J. Med. 2014 [doi:10.1056/<[lb]>NEJMoa1308659]).
Two-thirds of the participants showed a reduction of at least 50% in AHI score, and three-quarters showed a reduction of at least 25% in ODI score. And the median percentage of sleep time spent with poor oxygen saturation (less than 90%) declined from 5.4% to 0.9%.
In addition, patients’ scores on the Functional Outcomes of Sleep Questionnaire, which measures disease-specific quality of life, showed clinically meaningful improvement. And scores on the Epworth Sleepiness Scale normalized.
In the final, "challenge," phase of this study, the first 46 consecutive patients who had responded to this treatment at 1 year were randomly assigned to either continue it (turn the devices on at night) or to discontinue it (turn the devices off at night) for 1 more week. This challenge demonstrated that the improvements in OSA were in fact from the use of the hypoglossal-stimulation device, as sleep apnea relapsed in the patients who discontinued treatment.
There were no serious procedural complications, no rehospitalizations, and no infections. Two patients developed serious device-related adverse events, for an overall rate of less than 2%. In both cases, the device caused discomfort that was resolved by a second surgery to reposition it. Another 33 serious adverse events were considered to be unrelated to the implantation procedure or the device .
Nonserious adverse events – including sore throat from intubation during the procedure, pain at the incision sites, and muscle soreness – occurred in 88% of the study subjects. Nonserious events related to the device included discomfort during electrostimulation, reported by 40% of patients, and tongue soreness, reported by 21%. These resolved as the patients became acclimated to the device or after the device was reprogrammed to adjust the stimulation.
Twenty-three patients experienced temporary tongue weakness after the surgery, which resolved in all of them. Nine patients began using a tooth guard to resolve tongue soreness or abrasion.
"This approach may not be appropriate for persons with excessive airway collapsibility," Dr. Strollo and his associates cautioned. They screened potential study participants using endoscopy during drug-induced sleep, to identify functional upper-airway collapse originating in the retrolingual region, which would be the most amenable to neurostimulation at the base of the tongue.
The Food and Drug Administration’s Anesthesiology and Respiratory Therapy Devices Panel of the Medical Devices Advisory Committee will discuss, make recommendations, and vote on information related to the premarket approval application on Feb. 20.
Hypoglossal nerve stimulation appears effective
The findings by Strollo et al. give clinicians the rationale to consider hypoglossal nerve stimulation for selected patients who have trouble with CPAP therapy, said Dr. Atul Malhotra, FCCP.
Given the pathophysiology of OSA, a substantial proportion of patients would probably benefit from this treatment, even though symptoms were only reduced rather than completely eradicated. "Although the elimination of apnea would clearly be desirable, the observed reductions are probably similar to the benefits observed with CPAP, particularly when one considers the variability of adherence to CPAP therapy," he said.
Dr. Malhotra is in the division of pulmonary and critical care medicine at the University of California, San Diego. He reported previous ties to Philips Respironics, Apnex, and Apnicure. These remarks were taken from his editorial accompanying Dr. Strollo’s report (N. Engl. J. Med. 2014 [doi:10.1056/NEJMe1314084]).
An implantable device that stimulates the upper airway nerves and muscles produced long-term, clinically meaningful reductions in the severity of obstructive sleep apnea in an industry-sponsored study reported in the New England Journal of Medicine.
One year after implantation, patients showed a 68% reduction in scores on the Apnea-Hypopnea Index (AHI) and a 70% reduction in scores on the Oxygen Desaturation Index (ODI), as well as subjective improvements in daytime sleepiness and quality of life. The magnitude of these benefits was similar to that reported for continuous positive airway pressure (CPAP) therapy and superior to that reported for uvulopalatopharyngoplasty, said Dr. Patrick J. Strollo Jr., FCCP, of the department of otolaryngology, University of Pittsburgh Medical Center and his associates in the STAR (Stimulation Therapy for Apnea Reduction) trial group.
Upper airway stimulation using implanted electrodes to stimulate the hypoglossal nerve on one side of the neck "has been developed as a possible treatment option" for moderate to severe obstructive sleep apnea "and has shown promise in feasibility trials," the investigators noted.
For their study, designed in collaboration with the sponsor (Inspire Medical Systems) and the Food and Drug Administration, 126 patients who couldn’t tolerate CPAP therapy underwent surgical implantation of the device and were followed for 1 year. Otolaryngologists at 22 academic and private medical centers performed the surgery, which took a median of 140 minutes (range, 65-360 minutes).
Most (83%) of the participants were men. The mean age was 55 years (range, 31-80 years), and the mean body mass index was 28.4 kg/m2 (range, 18.4-32.5). Twenty-two of these patients (17%) had undergone uvulopalatopharyngoplasty, which had not corrected their obstructive sleep apnea.
The device includes a neurostimulator implanted in the intercostal muscles in the right mid-infraclavicular region, with one lead threaded upward inside the patient’s neck that is attached to three stimulation electrodes. The electrodes can be placed in a variety of configurations on the ipsilateral hypoglossal nerve, which, when stimulated, pushes the tongue forward and prevents the upper-airway muscles from collapsing and causing inspiratory flow obstruction.
The device also has a second, sensing lead tunneled between the internal and external intercostal muscles on the ipsilateral side to detect ventilatory effort during sleep, so that the stimulation of the hypoglossal nerve can be synchronized with the patient’s breathing.
The primary outcome of the study was the change in severity of obstructive sleep apnea, as measured by scores on the AHI and the ODI, at 12 months. The median AHI score decreased 68%, from 29.3 events per hour to 9.0 events per hour. The median ODI score dropped 70%, from 25.4 events per hour to 7.4 events <[lb]>per hour, the investigators said (N. Engl. J. Med. 2014 [doi:10.1056/<[lb]>NEJMoa1308659]).
Two-thirds of the participants showed a reduction of at least 50% in AHI score, and three-quarters showed a reduction of at least 25% in ODI score. And the median percentage of sleep time spent with poor oxygen saturation (less than 90%) declined from 5.4% to 0.9%.
In addition, patients’ scores on the Functional Outcomes of Sleep Questionnaire, which measures disease-specific quality of life, showed clinically meaningful improvement. And scores on the Epworth Sleepiness Scale normalized.
In the final, "challenge," phase of this study, the first 46 consecutive patients who had responded to this treatment at 1 year were randomly assigned to either continue it (turn the devices on at night) or to discontinue it (turn the devices off at night) for 1 more week. This challenge demonstrated that the improvements in OSA were in fact from the use of the hypoglossal-stimulation device, as sleep apnea relapsed in the patients who discontinued treatment.
There were no serious procedural complications, no rehospitalizations, and no infections. Two patients developed serious device-related adverse events, for an overall rate of less than 2%. In both cases, the device caused discomfort that was resolved by a second surgery to reposition it. Another 33 serious adverse events were considered to be unrelated to the implantation procedure or the device .
Nonserious adverse events – including sore throat from intubation during the procedure, pain at the incision sites, and muscle soreness – occurred in 88% of the study subjects. Nonserious events related to the device included discomfort during electrostimulation, reported by 40% of patients, and tongue soreness, reported by 21%. These resolved as the patients became acclimated to the device or after the device was reprogrammed to adjust the stimulation.
Twenty-three patients experienced temporary tongue weakness after the surgery, which resolved in all of them. Nine patients began using a tooth guard to resolve tongue soreness or abrasion.
"This approach may not be appropriate for persons with excessive airway collapsibility," Dr. Strollo and his associates cautioned. They screened potential study participants using endoscopy during drug-induced sleep, to identify functional upper-airway collapse originating in the retrolingual region, which would be the most amenable to neurostimulation at the base of the tongue.
The Food and Drug Administration’s Anesthesiology and Respiratory Therapy Devices Panel of the Medical Devices Advisory Committee will discuss, make recommendations, and vote on information related to the premarket approval application on Feb. 20.
Hypoglossal nerve stimulation appears effective
The findings by Strollo et al. give clinicians the rationale to consider hypoglossal nerve stimulation for selected patients who have trouble with CPAP therapy, said Dr. Atul Malhotra, FCCP.
Given the pathophysiology of OSA, a substantial proportion of patients would probably benefit from this treatment, even though symptoms were only reduced rather than completely eradicated. "Although the elimination of apnea would clearly be desirable, the observed reductions are probably similar to the benefits observed with CPAP, particularly when one considers the variability of adherence to CPAP therapy," he said.
Dr. Malhotra is in the division of pulmonary and critical care medicine at the University of California, San Diego. He reported previous ties to Philips Respironics, Apnex, and Apnicure. These remarks were taken from his editorial accompanying Dr. Strollo’s report (N. Engl. J. Med. 2014 [doi:10.1056/NEJMe1314084]).
Major finding: The median AHI score decreased 68%, from 29.3 events per hour to 9.0 events per hour, and the median ODI score declined 70%, from 25.4 events per hour to 7.4 events per hour.
Data source: A prospective multicenter cohort study involving 126 patients with moderate to severe obstructive sleep apnea who underwent implantation of an upper-airway neurostimulation device and were followed for 1 year.
Disclosures: This study was supported by Inspire Medical Systems. Dr. Strollo reported receiving funding from Inspire Medical Systems, and his associates reported ties to Inspire and numerous other industry sources.
CPAP treats resistant hypertension in OSA patients
For patients who have resistant hypertension and obstructive sleep apnea, 3 months of treatment with continuous positive airway pressure significantly reduces mean and diastolic blood pressure and improves the nocturnal blood pressure pattern, according to a report published in JAMA.
These improvements are dose related, with mean blood pressure decreasing 1.3 mm Hg, systolic blood pressure decreasing 1.9 mm Hg, and diastolic blood pressure decreasing 1.0 mm Hg for every additional hour of CPAP use, said Dr. Miguel-Angel Martinez-Garcia of the respiratory department at Hospital Universitario y Politecnico La Fe, Valencia (Spain), and his associates.
"Obstructive sleep apnea is highly prevalent in patients with resistant hypertension, regardless of other confounding variables such as the presence of obesity, thus suggesting this subgroup of hypertensive patients is a potential worthwhile population for CPAP treatment," they said.
"International guidelines have pointed out that even minimal reductions in blood pressure levels (to the order of 2-3 mm Hg of systolic pressure) could have a clinically significant effect by greatly reducing subsequent cardiovascular mortality (between 6% and 8% for stroke and 4% and 5% for coronary heart disease)," Dr. Martinez-Garcia and his colleagues noted.
Previous studies have shown that CPAP produces clinically significant decreases in blood pressure levels, but all have had "significant methodological limitations such as small cohorts or lack of randomization." So Dr. Martinez-Garcia and his associates performed a large randomized multicenter clinical trial to assess the issue.
They identified 194 adults treated at 24 teaching hospitals across Spain who had resistant hypertension unrelated to known causes such as primary aldosteronism, renal artery stenosis, or renal insufficiency. Resistant hypertension was confirmed via 24-hour ambulatory blood pressure monitoring. The study subjects also had obstructive sleep apnea, which was confirmed by standard sleep studies.
These subjects were randomly assigned to receive CPAP (98 patients) or no intervention (96 patients) while continuing their usual regimens of antihypertensive treatment. Approximately 69% of the subjects were men; mean age was 56 years, mean body mass index was 34.1, mean number of antihypertensive drugs taken was 3.8, and mean apnea-hypopnea index was 40.4 events per hour.
In the intention-to-treat analysis, after 3 months, the CPAP group achieved significantly greater decreases in 24-hour mean blood pressure and 24-hour mean diastolic blood pressure, and showed greater improvements during the night than during daytime. It also converted to more favorable nocturnal "dipper" and "riser" patterns in blood pressure, indicating decreased cardiovascular risk.
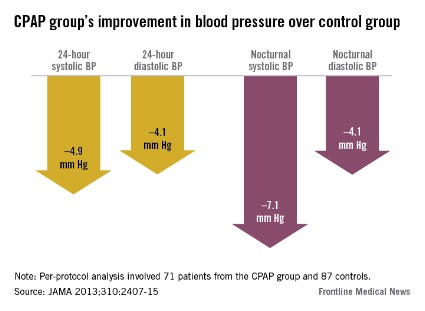
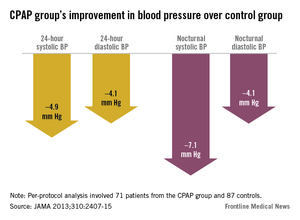
In the per-protocol analysis involving the 71 CPAP patients and 87 controls who adhered to the study protocol, these improvements were even more pronounced: The CPAP group showed a significant 4.4–mm HG decrease in 24-hour mean blood pressure, a 4.9–mm Hg decrease in systolic blood pressure, and a 4.1–mm Hg decrease in diastolic blood pressure (JAMA 2013 Dec. 10 [doi:10.1001/jama.2012.281250]).
At night, the findings were even better, at 7.1 mm Hg, with a 7.1–mm Hg decrease in systolic blood pressure and 4.1–mm Hg decrease in diastolic blood pressure, respectively. And again, the CPAP patients were more likely to convert to more favorable nocturnal "dipper" and "riser" patters in blood pressure.
There also was a positive linear correlation between the number of hours of CPAP use per night and the decrease in 24-hour mean blood pressure and diastolic blood pressure.
"Our results confirm that there is a clinically and statistically significant reduction in both 24-hour mean and diastolic blood pressure levels, especially during the night and in those patients with acceptable CPAP adherence," Dr. Martinez-Garcia and his associates said. Further research is warranted to assess whether these benefits translate into better health outcomes in the long term, they added.
This study was supported by Philips Respironics, Sociedad Espanola de Neumologia, Instituto de Salud Carlos III, and Sociedad Valencia de Neumologia. No other conflicts of interest were reported.
Dr. Paul A. Selecky, FCCP, comments: This report adds weight to the 2008 American Heart Association Scientific Statement on resistant hypertension, which stated that untreated sleep apnea is a secondary cause of resistant hypertension (Circulation 2008;117:e510-26). The present report takes the next step of showing that treatment with CPAP improves the hypertension.
For patients who have resistant hypertension and obstructive sleep apnea, 3 months of treatment with continuous positive airway pressure significantly reduces mean and diastolic blood pressure and improves the nocturnal blood pressure pattern, according to a report published in JAMA.
These improvements are dose related, with mean blood pressure decreasing 1.3 mm Hg, systolic blood pressure decreasing 1.9 mm Hg, and diastolic blood pressure decreasing 1.0 mm Hg for every additional hour of CPAP use, said Dr. Miguel-Angel Martinez-Garcia of the respiratory department at Hospital Universitario y Politecnico La Fe, Valencia (Spain), and his associates.

"Obstructive sleep apnea is highly prevalent in patients with resistant hypertension, regardless of other confounding variables such as the presence of obesity, thus suggesting this subgroup of hypertensive patients is a potential worthwhile population for CPAP treatment," they said.
"International guidelines have pointed out that even minimal reductions in blood pressure levels (to the order of 2-3 mm Hg of systolic pressure) could have a clinically significant effect by greatly reducing subsequent cardiovascular mortality (between 6% and 8% for stroke and 4% and 5% for coronary heart disease)," Dr. Martinez-Garcia and his colleagues noted.
Previous studies have shown that CPAP produces clinically significant decreases in blood pressure levels, but all have had "significant methodological limitations such as small cohorts or lack of randomization." So Dr. Martinez-Garcia and his associates performed a large randomized multicenter clinical trial to assess the issue.
They identified 194 adults treated at 24 teaching hospitals across Spain who had resistant hypertension unrelated to known causes such as primary aldosteronism, renal artery stenosis, or renal insufficiency. Resistant hypertension was confirmed via 24-hour ambulatory blood pressure monitoring. The study subjects also had obstructive sleep apnea, which was confirmed by standard sleep studies.
These subjects were randomly assigned to receive CPAP (98 patients) or no intervention (96 patients) while continuing their usual regimens of antihypertensive treatment. Approximately 69% of the subjects were men; mean age was 56 years, mean body mass index was 34.1, mean number of antihypertensive drugs taken was 3.8, and mean apnea-hypopnea index was 40.4 events per hour.
In the intention-to-treat analysis, after 3 months, the CPAP group achieved significantly greater decreases in 24-hour mean blood pressure and 24-hour mean diastolic blood pressure, and showed greater improvements during the night than during daytime. It also converted to more favorable nocturnal "dipper" and "riser" patterns in blood pressure, indicating decreased cardiovascular risk.
In the per-protocol analysis involving the 71 CPAP patients and 87 controls who adhered to the study protocol, these improvements were even more pronounced: The CPAP group showed a significant 4.4–mm HG decrease in 24-hour mean blood pressure, a 4.9–mm Hg decrease in systolic blood pressure, and a 4.1–mm Hg decrease in diastolic blood pressure (JAMA 2013 Dec. 10 [doi:10.1001/jama.2012.281250]).
At night, the findings were even better, at 7.1 mm Hg, with a 7.1–mm Hg decrease in systolic blood pressure and 4.1–mm Hg decrease in diastolic blood pressure, respectively. And again, the CPAP patients were more likely to convert to more favorable nocturnal "dipper" and "riser" patters in blood pressure.
There also was a positive linear correlation between the number of hours of CPAP use per night and the decrease in 24-hour mean blood pressure and diastolic blood pressure.
"Our results confirm that there is a clinically and statistically significant reduction in both 24-hour mean and diastolic blood pressure levels, especially during the night and in those patients with acceptable CPAP adherence," Dr. Martinez-Garcia and his associates said. Further research is warranted to assess whether these benefits translate into better health outcomes in the long term, they added.
This study was supported by Philips Respironics, Sociedad Espanola de Neumologia, Instituto de Salud Carlos III, and Sociedad Valencia de Neumologia. No other conflicts of interest were reported.
Dr. Paul A. Selecky, FCCP, comments: This report adds weight to the 2008 American Heart Association Scientific Statement on resistant hypertension, which stated that untreated sleep apnea is a secondary cause of resistant hypertension (Circulation 2008;117:e510-26). The present report takes the next step of showing that treatment with CPAP improves the hypertension.
For patients who have resistant hypertension and obstructive sleep apnea, 3 months of treatment with continuous positive airway pressure significantly reduces mean and diastolic blood pressure and improves the nocturnal blood pressure pattern, according to a report published in JAMA.
These improvements are dose related, with mean blood pressure decreasing 1.3 mm Hg, systolic blood pressure decreasing 1.9 mm Hg, and diastolic blood pressure decreasing 1.0 mm Hg for every additional hour of CPAP use, said Dr. Miguel-Angel Martinez-Garcia of the respiratory department at Hospital Universitario y Politecnico La Fe, Valencia (Spain), and his associates.

"Obstructive sleep apnea is highly prevalent in patients with resistant hypertension, regardless of other confounding variables such as the presence of obesity, thus suggesting this subgroup of hypertensive patients is a potential worthwhile population for CPAP treatment," they said.
"International guidelines have pointed out that even minimal reductions in blood pressure levels (to the order of 2-3 mm Hg of systolic pressure) could have a clinically significant effect by greatly reducing subsequent cardiovascular mortality (between 6% and 8% for stroke and 4% and 5% for coronary heart disease)," Dr. Martinez-Garcia and his colleagues noted.
Previous studies have shown that CPAP produces clinically significant decreases in blood pressure levels, but all have had "significant methodological limitations such as small cohorts or lack of randomization." So Dr. Martinez-Garcia and his associates performed a large randomized multicenter clinical trial to assess the issue.
They identified 194 adults treated at 24 teaching hospitals across Spain who had resistant hypertension unrelated to known causes such as primary aldosteronism, renal artery stenosis, or renal insufficiency. Resistant hypertension was confirmed via 24-hour ambulatory blood pressure monitoring. The study subjects also had obstructive sleep apnea, which was confirmed by standard sleep studies.
These subjects were randomly assigned to receive CPAP (98 patients) or no intervention (96 patients) while continuing their usual regimens of antihypertensive treatment. Approximately 69% of the subjects were men; mean age was 56 years, mean body mass index was 34.1, mean number of antihypertensive drugs taken was 3.8, and mean apnea-hypopnea index was 40.4 events per hour.
In the intention-to-treat analysis, after 3 months, the CPAP group achieved significantly greater decreases in 24-hour mean blood pressure and 24-hour mean diastolic blood pressure, and showed greater improvements during the night than during daytime. It also converted to more favorable nocturnal "dipper" and "riser" patterns in blood pressure, indicating decreased cardiovascular risk.
In the per-protocol analysis involving the 71 CPAP patients and 87 controls who adhered to the study protocol, these improvements were even more pronounced: The CPAP group showed a significant 4.4–mm HG decrease in 24-hour mean blood pressure, a 4.9–mm Hg decrease in systolic blood pressure, and a 4.1–mm Hg decrease in diastolic blood pressure (JAMA 2013 Dec. 10 [doi:10.1001/jama.2012.281250]).
At night, the findings were even better, at 7.1 mm Hg, with a 7.1–mm Hg decrease in systolic blood pressure and 4.1–mm Hg decrease in diastolic blood pressure, respectively. And again, the CPAP patients were more likely to convert to more favorable nocturnal "dipper" and "riser" patters in blood pressure.
There also was a positive linear correlation between the number of hours of CPAP use per night and the decrease in 24-hour mean blood pressure and diastolic blood pressure.
"Our results confirm that there is a clinically and statistically significant reduction in both 24-hour mean and diastolic blood pressure levels, especially during the night and in those patients with acceptable CPAP adherence," Dr. Martinez-Garcia and his associates said. Further research is warranted to assess whether these benefits translate into better health outcomes in the long term, they added.
This study was supported by Philips Respironics, Sociedad Espanola de Neumologia, Instituto de Salud Carlos III, and Sociedad Valencia de Neumologia. No other conflicts of interest were reported.
Dr. Paul A. Selecky, FCCP, comments: This report adds weight to the 2008 American Heart Association Scientific Statement on resistant hypertension, which stated that untreated sleep apnea is a secondary cause of resistant hypertension (Circulation 2008;117:e510-26). The present report takes the next step of showing that treatment with CPAP improves the hypertension.
Major finding: In the per-protocol analysis involving the 71 CPAP patients and 87 controls who adhered to the study protocol, the CPAP group showed a significant 4.4–mm HG decrease in 24-hour mean blood pressure, a 4.9–mm Hg decrease in systolic blood pressure, and a 4.1–mm Hg decrease in diastolic blood pressure.
Data source: An open-label multicenter randomized clinical trial involving 194 patients with concomitant resistant hypertension and obstructive sleep apnea who received either CPAP or no intervention for 3 months.
Disclosures: This study was supported by Philips Respironics, Sociedad Espanola de Neumologia, Instituto de Salud Carlos III, and Sociedad Valencia de Neumologia. No conflicts of interest were reported.
CT lung screen plagued by 18% overdiagnosis
An estimated 18% of the early lung cancers detected by low-dose CT screening in the National Lung Screening Trial were likely indolent and probably represent overdiagnosis, according to a report published in JAMA Internal Medicine.
The NLST found "an encouraging" 20% relative reduction in lung cancer-specific mortality among high-risk patients who were screened using low-dose CT, compared with chest radiography.
"These findings were met with enthusiasm, but before a widespread public health screening program is implemented, risks of screening also need to be considered," said Dr. Edward F. Patz Jr. of the department of radiology, Duke University Medical Center, Durham, N.C., and his associates in the NLST.
The chief risk in this case is overdiagnosis: identifying an early-stage lesion in an asymptomatic patient that would not progress or affect that patient’s long-term health.
It is likely that some of the tumors detected on low-dose CT were just such indolent cancers, and that those patients unnecessarily underwent invasive diagnostic procedures, surgical resection, and multiple follow-up studies.
To estimate how many of the detected cancers in the NLST were indolent and thus overdiagnosed, Dr. Patz and his colleagues used statistical probability methods to analyze extended follow-up data from the study.
The NLST involved 53,452 men and women aged 55-74 years who were enrolled during 2002-2004 and who had at least a 30-pack-year history of cigarette smoking.
The patients were randomly assigned to undergo lung cancer screening using either three annual low-dose CT exams or three annual single-view chest radiographs.
Mean follow-up was approximately 6 years. "At the end of the entire trial, there were 1,089 total lung cancer cases in the low-dose CT arm (649 detected by low-dose CT screening) and 969 cases in the [radiology] arm, for an excess of 120 cases. This gives [an] excess cancer rate of 18.5%," the investigators said (JAMA Intern. Med. 2013 Dec. 9 [doi: 10.1001/jamainternmed.2013.12738]).
"The data from this study suggest that ... 18% of persons in the low-dose CT arm with screen-detected lung cancer and 22% of those in the low-dose CT arm with screen-detected NSCLC [non-small cell lung cancer] may be cases of overdiagnosis," Dr. Patz and his associates said.
"In other words, if these individuals had not entered the NLST, they would not have received a lung cancer diagnosis or treatment, at least for the next 5 years."
In the future, the study authors noted, "once there are better biomarkers and imaging techniques to predict which individuals with a diagnosis of lung cancer will have more or less aggressive disease, treatment options can be optimized, and a mass screening program can become more valuable."
The National Institutes of Health supported the NSLT. No financial conflicts of interest were reported.
An estimated 18% of the early lung cancers detected by low-dose CT screening in the National Lung Screening Trial were likely indolent and probably represent overdiagnosis, according to a report published in JAMA Internal Medicine.
The NLST found "an encouraging" 20% relative reduction in lung cancer-specific mortality among high-risk patients who were screened using low-dose CT, compared with chest radiography.
"These findings were met with enthusiasm, but before a widespread public health screening program is implemented, risks of screening also need to be considered," said Dr. Edward F. Patz Jr. of the department of radiology, Duke University Medical Center, Durham, N.C., and his associates in the NLST.
The chief risk in this case is overdiagnosis: identifying an early-stage lesion in an asymptomatic patient that would not progress or affect that patient’s long-term health.
It is likely that some of the tumors detected on low-dose CT were just such indolent cancers, and that those patients unnecessarily underwent invasive diagnostic procedures, surgical resection, and multiple follow-up studies.
To estimate how many of the detected cancers in the NLST were indolent and thus overdiagnosed, Dr. Patz and his colleagues used statistical probability methods to analyze extended follow-up data from the study.
The NLST involved 53,452 men and women aged 55-74 years who were enrolled during 2002-2004 and who had at least a 30-pack-year history of cigarette smoking.
The patients were randomly assigned to undergo lung cancer screening using either three annual low-dose CT exams or three annual single-view chest radiographs.
Mean follow-up was approximately 6 years. "At the end of the entire trial, there were 1,089 total lung cancer cases in the low-dose CT arm (649 detected by low-dose CT screening) and 969 cases in the [radiology] arm, for an excess of 120 cases. This gives [an] excess cancer rate of 18.5%," the investigators said (JAMA Intern. Med. 2013 Dec. 9 [doi: 10.1001/jamainternmed.2013.12738]).
"The data from this study suggest that ... 18% of persons in the low-dose CT arm with screen-detected lung cancer and 22% of those in the low-dose CT arm with screen-detected NSCLC [non-small cell lung cancer] may be cases of overdiagnosis," Dr. Patz and his associates said.
"In other words, if these individuals had not entered the NLST, they would not have received a lung cancer diagnosis or treatment, at least for the next 5 years."
In the future, the study authors noted, "once there are better biomarkers and imaging techniques to predict which individuals with a diagnosis of lung cancer will have more or less aggressive disease, treatment options can be optimized, and a mass screening program can become more valuable."
The National Institutes of Health supported the NSLT. No financial conflicts of interest were reported.
An estimated 18% of the early lung cancers detected by low-dose CT screening in the National Lung Screening Trial were likely indolent and probably represent overdiagnosis, according to a report published in JAMA Internal Medicine.
The NLST found "an encouraging" 20% relative reduction in lung cancer-specific mortality among high-risk patients who were screened using low-dose CT, compared with chest radiography.
"These findings were met with enthusiasm, but before a widespread public health screening program is implemented, risks of screening also need to be considered," said Dr. Edward F. Patz Jr. of the department of radiology, Duke University Medical Center, Durham, N.C., and his associates in the NLST.
The chief risk in this case is overdiagnosis: identifying an early-stage lesion in an asymptomatic patient that would not progress or affect that patient’s long-term health.
It is likely that some of the tumors detected on low-dose CT were just such indolent cancers, and that those patients unnecessarily underwent invasive diagnostic procedures, surgical resection, and multiple follow-up studies.
To estimate how many of the detected cancers in the NLST were indolent and thus overdiagnosed, Dr. Patz and his colleagues used statistical probability methods to analyze extended follow-up data from the study.
The NLST involved 53,452 men and women aged 55-74 years who were enrolled during 2002-2004 and who had at least a 30-pack-year history of cigarette smoking.
The patients were randomly assigned to undergo lung cancer screening using either three annual low-dose CT exams or three annual single-view chest radiographs.
Mean follow-up was approximately 6 years. "At the end of the entire trial, there were 1,089 total lung cancer cases in the low-dose CT arm (649 detected by low-dose CT screening) and 969 cases in the [radiology] arm, for an excess of 120 cases. This gives [an] excess cancer rate of 18.5%," the investigators said (JAMA Intern. Med. 2013 Dec. 9 [doi: 10.1001/jamainternmed.2013.12738]).
"The data from this study suggest that ... 18% of persons in the low-dose CT arm with screen-detected lung cancer and 22% of those in the low-dose CT arm with screen-detected NSCLC [non-small cell lung cancer] may be cases of overdiagnosis," Dr. Patz and his associates said.
"In other words, if these individuals had not entered the NLST, they would not have received a lung cancer diagnosis or treatment, at least for the next 5 years."
In the future, the study authors noted, "once there are better biomarkers and imaging techniques to predict which individuals with a diagnosis of lung cancer will have more or less aggressive disease, treatment options can be optimized, and a mass screening program can become more valuable."
The National Institutes of Health supported the NSLT. No financial conflicts of interest were reported.
Pregabalin improves restless leg syndrome with less symptom augmentation
Oral pregabalin significantly improved symptoms of moderate to severe restless leg syndrome, compared with both placebo and pramipexole, in an industry-sponsored, international randomized trial.
Just as important, pregabalin was associated with significantly less iatrogenic worsening, or augmentation, of symptoms than was pramipexole, Richard P. Allen, Ph.D., of the department of neurology at Johns Hopkins University, Baltimore, and his associates reported Feb. 12 in the New England Journal of Medicine.
Pregabalin also was associated with lower rates of nausea, vomiting, and headache than pramipexole, but patients taking pregabalin had higher rates of suicidal ideation, dizziness, somnolence, and weight gain – "factors that may limit its long-term use," the investigators said.
Dopaminergic drugs such as pramipexole are known to be associated with augmentation in which symptoms intensify and may involve more parts of the body and start earlier in the day than before treatment, so researchers have been searching for an alternative medication. Pregabalin is a nondopaminergic agent with analgesic and anticonvulsant activity, and was recently reported to be effective against restless leg syndrome.
Dr. Allen and his colleagues assessed both agents in a study involving 719 adults with moderate to severe primary restless leg syndrome who were treated and followed at 102 medical centers in the United States and Europe. The patients did not undergo an objective assessment of sleep.
In the double-blind trial sponsored by Pfizer, the manufacturer of pregabalin, these patients were randomly assigned to receive 0.25 mg pramipexole, 0.5 mg pramipexole, 300 mg pregabalin, or matching placebo capsules every day for 12 weeks. At that time, all patients receiving placebo were randomly reassigned to one of the three active treatments for the remainder (40 weeks) of the 1-year study. The mean age of the patients ranged from 54 to 57 years in the groups, and 32%-45% in each group were men.
Periodically, the study participants reviewed with clinicians their daily symptom logs and completed the International RLS (IRLS) Study Group Rating Scale, which measures subjective symptom severity on a 0-40 scale, with higher scores indicating worse symptoms. They also reported on limb pain, sleep quality and quantity, and quality of life.
Clinicians also objectively assessed patients’ symptoms using the Clinical Global Impression of Improvement (CGI-I) scale, and they assessed possible augmentation using their clinical judgment, scores on the Augmentation Severity Rating Scale, and scores on the Structured Interview for the Diagnosis of Augmentation instrument.
At 12 weeks, patients who received pregabalin showed significantly greater improvement in IRLS scores than did those who received placebo, improving from 22.3 to 10.9 for pregabalin-treated patients and from 22.4 to 15.5 for placebo-treated patients. Pregabalin-treated patients also were more likely to have "very much improved" or "much improved" ratings on the CGI-I, compared with placebo (71.4% vs 46.8%).
Patients who received pregabalin also reported greater improvement in several sleep parameters, including waking after sleep onset, quality of sleep, number of nightly awakenings, and total sleep time, compared with those who received placebo. These measures also were significantly better for patients treated with 0.5 mg pramipexole when compared with placebo-treated patients, but not for those taking 0.25 mg pramipexole.
In a noninferiority assessment, pregabalin outperformed both doses of pramipexole on reduction in IRLS score at both 12 weeks and 52 weeks.
The finding that pregabalin is effective for RLS even though it has no direct effect on dopaminergic systems calls into question the rationale for dopaminergic therapies. Dopaminergic treatments have been predicated on the assumption that RLS results primarily from dopamine abnormalities, Dr. Allen and his associates noted (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056/NEJMoa1303646]).
Pregabalin was associated with significantly less augmentation than the 0.5-mg dose of pramipexole, but not the 0.25-mg dose. "Among patients receiving active treatment over the entire 52-week study period, augmentation occurred in 3 of 176 patients receiving pregabalin (1.7%), 11 of 167 receiving 0.25 mg of pramipexole (6.6%), and 16 of 178 receiving 0.5 mg of pramipexole (9.0%)," the investigators wrote.
Augmentation occurred more often among patients on higher-dose pramipexole than in those on the lower dose. Longer exposure to the medication also raised augmentation rates. "For participants who received active therapy for the full 52 weeks of the study (not 40 weeks), augmentation rarely occurred until the second half of the study," they wrote.
The rate of discontinuing the study because of adverse events was higher for the patients receiving pregabalin (27.5%) than for those receiving pramipexole at either the lower dose (18.5%) or higher dose (23.9%).
A total of 50 serious adverse events occurred in 37 patients: 16 events in the pregabalin group, 22 events in the lower-dose pramipexole group, and 12 events in the higher-dose pramipexole group. This included 11 cases of suicidal ideation: 6 in the pregabalin group, 3 in the lower-dose pramipexole group, and 2 in the higher-dose pramipexole group.
Dr. Allen reported ties to Pfizer, UCB Pharma, Impax Pharmaceuticals, Luitpold Pharma, Xenoport, GlaxoSmithKline, and Pharmacosmos. His associates reported ties to Pfizer and numerous industry sources. Four authors are employees of Pfizer.
This carefully conducted study is one of a few head-to-head studies of two classes of medications that have been reported for the treatment of restless leg syndrome. It presents compelling evidence for the efficacy of a nondopaminergic drug in the treatment of RLS and thereby implicates a role for nondopaminergic pathways in the disease, said Dr. Sudhansu Chokroverty.
Although augmentation occurred significantly more often with pramipexole, patients who took pregabalin still had a rate of 1.7%, which "raises the question of whether augmentation is related to medication, is intrinsic to RLS, or is related to individual patient characteristics," he noted.
Dr. Chokroverty is with the department of neurology at the New Jersey Neuroscience Institute, JFK Medical Center, Edison N.J., and at Seton Hall University in South Orange. He reported no relevant financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Allen’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056/NEJMe1313155]).
This carefully conducted study is one of a few head-to-head studies of two classes of medications that have been reported for the treatment of restless leg syndrome. It presents compelling evidence for the efficacy of a nondopaminergic drug in the treatment of RLS and thereby implicates a role for nondopaminergic pathways in the disease, said Dr. Sudhansu Chokroverty.
Although augmentation occurred significantly more often with pramipexole, patients who took pregabalin still had a rate of 1.7%, which "raises the question of whether augmentation is related to medication, is intrinsic to RLS, or is related to individual patient characteristics," he noted.
Dr. Chokroverty is with the department of neurology at the New Jersey Neuroscience Institute, JFK Medical Center, Edison N.J., and at Seton Hall University in South Orange. He reported no relevant financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Allen’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056/NEJMe1313155]).
This carefully conducted study is one of a few head-to-head studies of two classes of medications that have been reported for the treatment of restless leg syndrome. It presents compelling evidence for the efficacy of a nondopaminergic drug in the treatment of RLS and thereby implicates a role for nondopaminergic pathways in the disease, said Dr. Sudhansu Chokroverty.
Although augmentation occurred significantly more often with pramipexole, patients who took pregabalin still had a rate of 1.7%, which "raises the question of whether augmentation is related to medication, is intrinsic to RLS, or is related to individual patient characteristics," he noted.
Dr. Chokroverty is with the department of neurology at the New Jersey Neuroscience Institute, JFK Medical Center, Edison N.J., and at Seton Hall University in South Orange. He reported no relevant financial conflicts of interest. These remarks were taken from his editorial accompanying Dr. Allen’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056/NEJMe1313155]).
Oral pregabalin significantly improved symptoms of moderate to severe restless leg syndrome, compared with both placebo and pramipexole, in an industry-sponsored, international randomized trial.
Just as important, pregabalin was associated with significantly less iatrogenic worsening, or augmentation, of symptoms than was pramipexole, Richard P. Allen, Ph.D., of the department of neurology at Johns Hopkins University, Baltimore, and his associates reported Feb. 12 in the New England Journal of Medicine.
Pregabalin also was associated with lower rates of nausea, vomiting, and headache than pramipexole, but patients taking pregabalin had higher rates of suicidal ideation, dizziness, somnolence, and weight gain – "factors that may limit its long-term use," the investigators said.
Dopaminergic drugs such as pramipexole are known to be associated with augmentation in which symptoms intensify and may involve more parts of the body and start earlier in the day than before treatment, so researchers have been searching for an alternative medication. Pregabalin is a nondopaminergic agent with analgesic and anticonvulsant activity, and was recently reported to be effective against restless leg syndrome.
Dr. Allen and his colleagues assessed both agents in a study involving 719 adults with moderate to severe primary restless leg syndrome who were treated and followed at 102 medical centers in the United States and Europe. The patients did not undergo an objective assessment of sleep.
In the double-blind trial sponsored by Pfizer, the manufacturer of pregabalin, these patients were randomly assigned to receive 0.25 mg pramipexole, 0.5 mg pramipexole, 300 mg pregabalin, or matching placebo capsules every day for 12 weeks. At that time, all patients receiving placebo were randomly reassigned to one of the three active treatments for the remainder (40 weeks) of the 1-year study. The mean age of the patients ranged from 54 to 57 years in the groups, and 32%-45% in each group were men.
Periodically, the study participants reviewed with clinicians their daily symptom logs and completed the International RLS (IRLS) Study Group Rating Scale, which measures subjective symptom severity on a 0-40 scale, with higher scores indicating worse symptoms. They also reported on limb pain, sleep quality and quantity, and quality of life.
Clinicians also objectively assessed patients’ symptoms using the Clinical Global Impression of Improvement (CGI-I) scale, and they assessed possible augmentation using their clinical judgment, scores on the Augmentation Severity Rating Scale, and scores on the Structured Interview for the Diagnosis of Augmentation instrument.
At 12 weeks, patients who received pregabalin showed significantly greater improvement in IRLS scores than did those who received placebo, improving from 22.3 to 10.9 for pregabalin-treated patients and from 22.4 to 15.5 for placebo-treated patients. Pregabalin-treated patients also were more likely to have "very much improved" or "much improved" ratings on the CGI-I, compared with placebo (71.4% vs 46.8%).
Patients who received pregabalin also reported greater improvement in several sleep parameters, including waking after sleep onset, quality of sleep, number of nightly awakenings, and total sleep time, compared with those who received placebo. These measures also were significantly better for patients treated with 0.5 mg pramipexole when compared with placebo-treated patients, but not for those taking 0.25 mg pramipexole.
In a noninferiority assessment, pregabalin outperformed both doses of pramipexole on reduction in IRLS score at both 12 weeks and 52 weeks.
The finding that pregabalin is effective for RLS even though it has no direct effect on dopaminergic systems calls into question the rationale for dopaminergic therapies. Dopaminergic treatments have been predicated on the assumption that RLS results primarily from dopamine abnormalities, Dr. Allen and his associates noted (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056/NEJMoa1303646]).
Pregabalin was associated with significantly less augmentation than the 0.5-mg dose of pramipexole, but not the 0.25-mg dose. "Among patients receiving active treatment over the entire 52-week study period, augmentation occurred in 3 of 176 patients receiving pregabalin (1.7%), 11 of 167 receiving 0.25 mg of pramipexole (6.6%), and 16 of 178 receiving 0.5 mg of pramipexole (9.0%)," the investigators wrote.
Augmentation occurred more often among patients on higher-dose pramipexole than in those on the lower dose. Longer exposure to the medication also raised augmentation rates. "For participants who received active therapy for the full 52 weeks of the study (not 40 weeks), augmentation rarely occurred until the second half of the study," they wrote.
The rate of discontinuing the study because of adverse events was higher for the patients receiving pregabalin (27.5%) than for those receiving pramipexole at either the lower dose (18.5%) or higher dose (23.9%).
A total of 50 serious adverse events occurred in 37 patients: 16 events in the pregabalin group, 22 events in the lower-dose pramipexole group, and 12 events in the higher-dose pramipexole group. This included 11 cases of suicidal ideation: 6 in the pregabalin group, 3 in the lower-dose pramipexole group, and 2 in the higher-dose pramipexole group.
Dr. Allen reported ties to Pfizer, UCB Pharma, Impax Pharmaceuticals, Luitpold Pharma, Xenoport, GlaxoSmithKline, and Pharmacosmos. His associates reported ties to Pfizer and numerous industry sources. Four authors are employees of Pfizer.
Oral pregabalin significantly improved symptoms of moderate to severe restless leg syndrome, compared with both placebo and pramipexole, in an industry-sponsored, international randomized trial.
Just as important, pregabalin was associated with significantly less iatrogenic worsening, or augmentation, of symptoms than was pramipexole, Richard P. Allen, Ph.D., of the department of neurology at Johns Hopkins University, Baltimore, and his associates reported Feb. 12 in the New England Journal of Medicine.
Pregabalin also was associated with lower rates of nausea, vomiting, and headache than pramipexole, but patients taking pregabalin had higher rates of suicidal ideation, dizziness, somnolence, and weight gain – "factors that may limit its long-term use," the investigators said.
Dopaminergic drugs such as pramipexole are known to be associated with augmentation in which symptoms intensify and may involve more parts of the body and start earlier in the day than before treatment, so researchers have been searching for an alternative medication. Pregabalin is a nondopaminergic agent with analgesic and anticonvulsant activity, and was recently reported to be effective against restless leg syndrome.
Dr. Allen and his colleagues assessed both agents in a study involving 719 adults with moderate to severe primary restless leg syndrome who were treated and followed at 102 medical centers in the United States and Europe. The patients did not undergo an objective assessment of sleep.
In the double-blind trial sponsored by Pfizer, the manufacturer of pregabalin, these patients were randomly assigned to receive 0.25 mg pramipexole, 0.5 mg pramipexole, 300 mg pregabalin, or matching placebo capsules every day for 12 weeks. At that time, all patients receiving placebo were randomly reassigned to one of the three active treatments for the remainder (40 weeks) of the 1-year study. The mean age of the patients ranged from 54 to 57 years in the groups, and 32%-45% in each group were men.
Periodically, the study participants reviewed with clinicians their daily symptom logs and completed the International RLS (IRLS) Study Group Rating Scale, which measures subjective symptom severity on a 0-40 scale, with higher scores indicating worse symptoms. They also reported on limb pain, sleep quality and quantity, and quality of life.
Clinicians also objectively assessed patients’ symptoms using the Clinical Global Impression of Improvement (CGI-I) scale, and they assessed possible augmentation using their clinical judgment, scores on the Augmentation Severity Rating Scale, and scores on the Structured Interview for the Diagnosis of Augmentation instrument.
At 12 weeks, patients who received pregabalin showed significantly greater improvement in IRLS scores than did those who received placebo, improving from 22.3 to 10.9 for pregabalin-treated patients and from 22.4 to 15.5 for placebo-treated patients. Pregabalin-treated patients also were more likely to have "very much improved" or "much improved" ratings on the CGI-I, compared with placebo (71.4% vs 46.8%).
Patients who received pregabalin also reported greater improvement in several sleep parameters, including waking after sleep onset, quality of sleep, number of nightly awakenings, and total sleep time, compared with those who received placebo. These measures also were significantly better for patients treated with 0.5 mg pramipexole when compared with placebo-treated patients, but not for those taking 0.25 mg pramipexole.
In a noninferiority assessment, pregabalin outperformed both doses of pramipexole on reduction in IRLS score at both 12 weeks and 52 weeks.
The finding that pregabalin is effective for RLS even though it has no direct effect on dopaminergic systems calls into question the rationale for dopaminergic therapies. Dopaminergic treatments have been predicated on the assumption that RLS results primarily from dopamine abnormalities, Dr. Allen and his associates noted (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056/NEJMoa1303646]).
Pregabalin was associated with significantly less augmentation than the 0.5-mg dose of pramipexole, but not the 0.25-mg dose. "Among patients receiving active treatment over the entire 52-week study period, augmentation occurred in 3 of 176 patients receiving pregabalin (1.7%), 11 of 167 receiving 0.25 mg of pramipexole (6.6%), and 16 of 178 receiving 0.5 mg of pramipexole (9.0%)," the investigators wrote.
Augmentation occurred more often among patients on higher-dose pramipexole than in those on the lower dose. Longer exposure to the medication also raised augmentation rates. "For participants who received active therapy for the full 52 weeks of the study (not 40 weeks), augmentation rarely occurred until the second half of the study," they wrote.
The rate of discontinuing the study because of adverse events was higher for the patients receiving pregabalin (27.5%) than for those receiving pramipexole at either the lower dose (18.5%) or higher dose (23.9%).
A total of 50 serious adverse events occurred in 37 patients: 16 events in the pregabalin group, 22 events in the lower-dose pramipexole group, and 12 events in the higher-dose pramipexole group. This included 11 cases of suicidal ideation: 6 in the pregabalin group, 3 in the lower-dose pramipexole group, and 2 in the higher-dose pramipexole group.
Dr. Allen reported ties to Pfizer, UCB Pharma, Impax Pharmaceuticals, Luitpold Pharma, Xenoport, GlaxoSmithKline, and Pharmacosmos. His associates reported ties to Pfizer and numerous industry sources. Four authors are employees of Pfizer.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: During the entire 52-week study period, augmentation occurred in 3 of 176 patients receiving pregabalin (1.7%), 11 of 167 receiving 0.25 mg of pramipexole (6.6%), and 16 of 178 receiving 0.5 mg of pramipexole (9.0%).
Data source: A 1-year, international, randomized, double-blind clinical trial comparing pregabalin, pramipexole, and placebo in 719 adults with moderate to severe restless leg syndrome.
Disclosures: This study was supported by Pfizer, maker of pregabalin. Dr. Allen reported ties to Pfizer, UCB Pharma, Impax Pharmaceuticals, Luitpold Pharma, Xenoport, GlaxoSmithKline, and Pharmacosmos. His associates reported ties to Pfizer and numerous industry sources. Four authors are employees of Pfizer.
Final results validate sentinel-node biopsy for melanoma
Sentinel-node biopsy clearly benefits patients with intermediate-thickness or thick primary melanomas who are found to have nodal involvement and undergo immediate lymphadenectomy, tripling their disease-free survival and doubling their melanoma-specific survival and distant disease-free survival, according to a report published online Feb. 12 in the New England Journal of Medicine.
These long-term findings, the final results of 20 years of investigation in MSLT-I (Multicenter Selective Lymphadenectomy Trial), "clearly validate the use of sentinel-node biopsy" in this patient population. Among patients found to have no nodal involvement, "the procedure provides accurate and important staging information," while among those found to have nodal involvement, it enhances regional disease control and substantially improves survival, said Dr. Donald L. Morton of the John Wayne Cancer Institute at Saint John’s Health Center, Santa Monica, Calif., and his associates.
Dr. Morton passed away shortly before publication of this final MSLT-I report.
The MSLT-I, which began in 1994, was intended to determine whether sentinel-node biopsy – a new procedure at the time that was developed by Dr. Morton – could accurately identify clinically occult metastases. More important, since only about 20% of such patients are found to have metastases, the MSLT-I was to determine whether it was worthwhile to subject all patients to the procedure. In other words, did sentinel-node biopsy with immediate lymphadenectomy of involved nodes yield better outcomes than did watchful waiting with delayed lymphadenectomy, performed only when nodal recurrence became evident during observation?
This final report involved 1,560 patients with localized cutaneous primary melanomas: 1,270 with intermediate-thickness (1.20-3.50 mm) lesions and 290 with thick (greater than 3.50 mm) lesions. A total of 943 patients were randomly assigned to undergo sentinel-node biopsy plus immediate lymphadenectomy if metastases were detected in sentinel nodes, and 617 were randomly assigned to close observation with delayed lymphadenectomy if nodal metastases developed during observation.
All the study participants were monitored periodically throughout follow-up using clinical examination, blood testing for biomarkers of melanoma, chest radiography, PET scanning, CT scanning, and/or nodal ultrasonography.
The primary end point, melanoma-specific survival across the entire study cohort, was not significantly different between patients who underwent sentinel-node biopsy (81.4%) and those who had observation only (78.3%). This is not surprising as only the 20% of patients who actually had nodal metastases could potentially derive a survival benefit from the procedure.
Latent-subgroup statistical methods were used to assess only this 20% of patients with nodal metastases. In this, the most relevant, data analysis, disease-free survival was increased by a factor of 3.2, distant disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for patients who underwent sentinel-node biopsy compared with those who had observation only, Dr. Morton and his associates wrote (N. Engl. J. Med. 2014 Feb 12 [doi:10.1056/NEJMoa1310460]).
Among patients with intermediate-thickness melanomas, the estimated treatment effect on disease-free survival was 1.17 (P less than .001), and the estimated effect on distant disease-free survival was 0.73 (P = .04). For melanoma-specific survival, the estimated treatment effect was 0.68 (P = .05).
"Our long-term results confirm that sentinel-node biopsy correctly determines the pathologic status of the nodal basin in 96% of cases and is the most powerful prognostic indicator," they noted.
The data also indicate that "essentially all metastases detected by sentinel-node biopsy eventually would have become clinically evident if [they had] not [been] removed," the investigators added.
The MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
The final data from this landmark trial "corroborate the profound prognostic significance of micrometastasis identified by sentinel-node biopsy," said Dr. Charles M. Balch and Dr. Jeffrey E. Gershenwald.
The finding that early lymphadenectomy improved melanoma-specific survival by 44% implies that delaying lymphadenectomy until there was clinical evidence of metastasis allowed an opportunity for further dissemination of regional disease. And the fact that the primary end point (improved melanoma-specific survival across the entire cohort of patients with intermediate thickness melanomas) of MSLT-I was not achieved "doesn’t detract from the clinical importance of regional lymph-node staging," they said.
Dr. Balch is at the University of Texas Southwestern Medical Center, Dallas; he reported ties to Merck and Amgen. Dr. Gershenwald is in the department of surgical oncology at the melanoma and skin center at the University of Texas M.D. Anderson Cancer Center, Houston.; he reported ties to Navidea, GlaxoSmithKline, UpToDate, and Mercator Therapeutics. These remarks were taken from their editorial accompanying Dr. Morton’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056e1313690]).
The final data from this landmark trial "corroborate the profound prognostic significance of micrometastasis identified by sentinel-node biopsy," said Dr. Charles M. Balch and Dr. Jeffrey E. Gershenwald.
The finding that early lymphadenectomy improved melanoma-specific survival by 44% implies that delaying lymphadenectomy until there was clinical evidence of metastasis allowed an opportunity for further dissemination of regional disease. And the fact that the primary end point (improved melanoma-specific survival across the entire cohort of patients with intermediate thickness melanomas) of MSLT-I was not achieved "doesn’t detract from the clinical importance of regional lymph-node staging," they said.
Dr. Balch is at the University of Texas Southwestern Medical Center, Dallas; he reported ties to Merck and Amgen. Dr. Gershenwald is in the department of surgical oncology at the melanoma and skin center at the University of Texas M.D. Anderson Cancer Center, Houston.; he reported ties to Navidea, GlaxoSmithKline, UpToDate, and Mercator Therapeutics. These remarks were taken from their editorial accompanying Dr. Morton’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056e1313690]).
The final data from this landmark trial "corroborate the profound prognostic significance of micrometastasis identified by sentinel-node biopsy," said Dr. Charles M. Balch and Dr. Jeffrey E. Gershenwald.
The finding that early lymphadenectomy improved melanoma-specific survival by 44% implies that delaying lymphadenectomy until there was clinical evidence of metastasis allowed an opportunity for further dissemination of regional disease. And the fact that the primary end point (improved melanoma-specific survival across the entire cohort of patients with intermediate thickness melanomas) of MSLT-I was not achieved "doesn’t detract from the clinical importance of regional lymph-node staging," they said.
Dr. Balch is at the University of Texas Southwestern Medical Center, Dallas; he reported ties to Merck and Amgen. Dr. Gershenwald is in the department of surgical oncology at the melanoma and skin center at the University of Texas M.D. Anderson Cancer Center, Houston.; he reported ties to Navidea, GlaxoSmithKline, UpToDate, and Mercator Therapeutics. These remarks were taken from their editorial accompanying Dr. Morton’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056e1313690]).
Sentinel-node biopsy clearly benefits patients with intermediate-thickness or thick primary melanomas who are found to have nodal involvement and undergo immediate lymphadenectomy, tripling their disease-free survival and doubling their melanoma-specific survival and distant disease-free survival, according to a report published online Feb. 12 in the New England Journal of Medicine.
These long-term findings, the final results of 20 years of investigation in MSLT-I (Multicenter Selective Lymphadenectomy Trial), "clearly validate the use of sentinel-node biopsy" in this patient population. Among patients found to have no nodal involvement, "the procedure provides accurate and important staging information," while among those found to have nodal involvement, it enhances regional disease control and substantially improves survival, said Dr. Donald L. Morton of the John Wayne Cancer Institute at Saint John’s Health Center, Santa Monica, Calif., and his associates.
Dr. Morton passed away shortly before publication of this final MSLT-I report.
The MSLT-I, which began in 1994, was intended to determine whether sentinel-node biopsy – a new procedure at the time that was developed by Dr. Morton – could accurately identify clinically occult metastases. More important, since only about 20% of such patients are found to have metastases, the MSLT-I was to determine whether it was worthwhile to subject all patients to the procedure. In other words, did sentinel-node biopsy with immediate lymphadenectomy of involved nodes yield better outcomes than did watchful waiting with delayed lymphadenectomy, performed only when nodal recurrence became evident during observation?
This final report involved 1,560 patients with localized cutaneous primary melanomas: 1,270 with intermediate-thickness (1.20-3.50 mm) lesions and 290 with thick (greater than 3.50 mm) lesions. A total of 943 patients were randomly assigned to undergo sentinel-node biopsy plus immediate lymphadenectomy if metastases were detected in sentinel nodes, and 617 were randomly assigned to close observation with delayed lymphadenectomy if nodal metastases developed during observation.
All the study participants were monitored periodically throughout follow-up using clinical examination, blood testing for biomarkers of melanoma, chest radiography, PET scanning, CT scanning, and/or nodal ultrasonography.
The primary end point, melanoma-specific survival across the entire study cohort, was not significantly different between patients who underwent sentinel-node biopsy (81.4%) and those who had observation only (78.3%). This is not surprising as only the 20% of patients who actually had nodal metastases could potentially derive a survival benefit from the procedure.
Latent-subgroup statistical methods were used to assess only this 20% of patients with nodal metastases. In this, the most relevant, data analysis, disease-free survival was increased by a factor of 3.2, distant disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for patients who underwent sentinel-node biopsy compared with those who had observation only, Dr. Morton and his associates wrote (N. Engl. J. Med. 2014 Feb 12 [doi:10.1056/NEJMoa1310460]).
Among patients with intermediate-thickness melanomas, the estimated treatment effect on disease-free survival was 1.17 (P less than .001), and the estimated effect on distant disease-free survival was 0.73 (P = .04). For melanoma-specific survival, the estimated treatment effect was 0.68 (P = .05).
"Our long-term results confirm that sentinel-node biopsy correctly determines the pathologic status of the nodal basin in 96% of cases and is the most powerful prognostic indicator," they noted.
The data also indicate that "essentially all metastases detected by sentinel-node biopsy eventually would have become clinically evident if [they had] not [been] removed," the investigators added.
The MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
Sentinel-node biopsy clearly benefits patients with intermediate-thickness or thick primary melanomas who are found to have nodal involvement and undergo immediate lymphadenectomy, tripling their disease-free survival and doubling their melanoma-specific survival and distant disease-free survival, according to a report published online Feb. 12 in the New England Journal of Medicine.
These long-term findings, the final results of 20 years of investigation in MSLT-I (Multicenter Selective Lymphadenectomy Trial), "clearly validate the use of sentinel-node biopsy" in this patient population. Among patients found to have no nodal involvement, "the procedure provides accurate and important staging information," while among those found to have nodal involvement, it enhances regional disease control and substantially improves survival, said Dr. Donald L. Morton of the John Wayne Cancer Institute at Saint John’s Health Center, Santa Monica, Calif., and his associates.
Dr. Morton passed away shortly before publication of this final MSLT-I report.
The MSLT-I, which began in 1994, was intended to determine whether sentinel-node biopsy – a new procedure at the time that was developed by Dr. Morton – could accurately identify clinically occult metastases. More important, since only about 20% of such patients are found to have metastases, the MSLT-I was to determine whether it was worthwhile to subject all patients to the procedure. In other words, did sentinel-node biopsy with immediate lymphadenectomy of involved nodes yield better outcomes than did watchful waiting with delayed lymphadenectomy, performed only when nodal recurrence became evident during observation?
This final report involved 1,560 patients with localized cutaneous primary melanomas: 1,270 with intermediate-thickness (1.20-3.50 mm) lesions and 290 with thick (greater than 3.50 mm) lesions. A total of 943 patients were randomly assigned to undergo sentinel-node biopsy plus immediate lymphadenectomy if metastases were detected in sentinel nodes, and 617 were randomly assigned to close observation with delayed lymphadenectomy if nodal metastases developed during observation.
All the study participants were monitored periodically throughout follow-up using clinical examination, blood testing for biomarkers of melanoma, chest radiography, PET scanning, CT scanning, and/or nodal ultrasonography.
The primary end point, melanoma-specific survival across the entire study cohort, was not significantly different between patients who underwent sentinel-node biopsy (81.4%) and those who had observation only (78.3%). This is not surprising as only the 20% of patients who actually had nodal metastases could potentially derive a survival benefit from the procedure.
Latent-subgroup statistical methods were used to assess only this 20% of patients with nodal metastases. In this, the most relevant, data analysis, disease-free survival was increased by a factor of 3.2, distant disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for patients who underwent sentinel-node biopsy compared with those who had observation only, Dr. Morton and his associates wrote (N. Engl. J. Med. 2014 Feb 12 [doi:10.1056/NEJMoa1310460]).
Among patients with intermediate-thickness melanomas, the estimated treatment effect on disease-free survival was 1.17 (P less than .001), and the estimated effect on distant disease-free survival was 0.73 (P = .04). For melanoma-specific survival, the estimated treatment effect was 0.68 (P = .05).
"Our long-term results confirm that sentinel-node biopsy correctly determines the pathologic status of the nodal basin in 96% of cases and is the most powerful prognostic indicator," they noted.
The data also indicate that "essentially all metastases detected by sentinel-node biopsy eventually would have become clinically evident if [they had] not [been] removed," the investigators added.
The MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: Among patients with nodal metastases, disease-free survival was increased by a factor of 3.2, distant-disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for those who underwent sentinel-node biopsy, compared with those who had observation only.
Data source: The 20-year international randomized MSLT-I study involving 1,560 patients with intermediate-thickness or thick localized cutaneous melanomas comparing SNB with nodal observation.
Disclosures: MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
Removing opposite breast cuts 20-year mortality 48% in BRCA mutation carriers
Among early-stage breast cancer patients who carry BRCA mutations, prophylactic mastectomy of the contralateral breast appears to reduce the risk of dying from breast cancer within 20 years by 48%, according to a report published online Feb. 11 in the British Medical Journal.
This significant mortality benefit is most pronounced during the second decade after the initial breast cancer diagnosis, said Dr. Kelly Metcalfe of the University of Toronto, and her associates.
"We conclude that it is reasonable to propose bilateral mastectomy as the initial treatment option for women with early-stage breast cancer who are carriers of a BRCA1 or BRCA2 mutation. For those who have been treated in the past with unilateral mastectomy or breast-conserving surgery, the possibility of a second surgery should be discussed," they said.
Until now, the data regarding long-term survival after prophylactic mastectomy have been sparse, and no study has examined mortality specifically related to mastectomy of the contralateral breast. "Traditionally, breast cancer trialists and clinical epidemiologists focus their attention on the 10-year period after diagnosis, because this is when the majority of cancer-related deaths occur. However, a mortality benefit from preventing a second primary breast cancer is unlikely to be apparent within this narrow interval, given that second primary cancers accumulate slowly and for an extended period," Dr. Metcalfe and her colleagues said.
They reviewed the medical records and pathology reports of 390 women who had been diagnosed as having stage I or II breast cancer in 1975-2009 when they were aged 65 or younger, were known or likely carriers of a deleterious mutation in the BRCA1 or BRCA2 gene, and were treated with unilateral or bilateral mastectomy. These study participants were identified from the histories of 290 different families in which one member had received genetic counseling at any of 12 specialty clinics.
A total of 44 of the women were initially treated with bilateral mastectomy. The remaining 346 initially underwent unilateral mastectomy, but 137 of them went on to have mastectomy of the contralateral breast at a later time. Thus, in the final analysis 181 women had prophylactic mastectomy of the contralateral breast and 209 did not.
The mean follow-up was 13 years (range, 0.1-20.0 years).
Overall, 79 women (20% of the entire study population) died from breast cancer during follow-up: 18 of the 181 women who had prophylactic mastectomy of the contralateral breast (9.9%), compared with 61 of 209 who did not have prophylactic mastectomy (29.1%).
At 20 years, the survival rate for women who underwent prophylactic mastectomy was 88%, compared with a 66% survival rate among those who did not have prophylactic mastectomy of the contralateral breast.
In a multivariable analysis that controlled for age at diagnosis, tumor size, nodal status, treatment, and other prognostic factors, 20-year breast-cancer-specific mortality was 48% lower for those who underwent contralateral mastectomy than for those who did not (hazard ratio 0.52, 95% confidence interval 0.29 to 0.93; P = .03), the investigators wrote (Br. Med. J. 2014 Feb. 11 [doi:10.1136/bmj.g226]).
The reduction in mortality was much greater during the second decade of follow-up (80% reduction) than during the first decade (35% reduction). "On average, the time from first breast cancer to ... cancer of the contralateral breast is 5.7 years. Therefore, a delay in the observed benefit of contralateral mastectomy is to be expected – that is, the reduction in mortality results from a reduction in deaths from cancer of the contralateral breast," Dr. Metcalfe and her associates said.
They noted that it is critical that these findings be confirmed in other study populations, especially in view of their relatively small study population.
This study was funded by the Canadian Breast Cancer Foundation. Dr. Metcalfe is supported by the Canadian Institutes of Health Research and the Ontario Women’s Health Council. No financial conflicts of interest were reported.
The main limitation of the study by Metcalfe and colleagues is the relatively small number of endpoints (breast cancer deaths), "which provide unstable estimates and make statistical inferences more challenging," wrote Dr. Karin B. Michels, Sc.D., Ph.D. As the authors noted, larger studies of this issue are needed, she said.
Breasts are not statistics; they are essential parts of women’s identity, sexuality, and self-perception, and the decision to undergo a bilateral mastectomy is highly individual. "A woman needs to weigh up alternative options, including regular close monitoring and the use of tamoxifen or raloxifene, while considering the opportunities but also possible complications of reconstructive surgery. No statistics and no statistician can make this decision for her," Dr. Michels said.
Dr. Michels is at the Obstetrics and Gynecology Epidemiology Center at Brigham and Women’s Hospital and at Harvard Medical School, both in Boston. She reported no financial conflicts of interest. These remarks were taken from her editorial accompanying Dr. Metcalfe’s report (Br. Med. J. 2014 Feb. 11 [doi:10.1136/bmj.g1379]).
The main limitation of the study by Metcalfe and colleagues is the relatively small number of endpoints (breast cancer deaths), "which provide unstable estimates and make statistical inferences more challenging," wrote Dr. Karin B. Michels, Sc.D., Ph.D. As the authors noted, larger studies of this issue are needed, she said.
Breasts are not statistics; they are essential parts of women’s identity, sexuality, and self-perception, and the decision to undergo a bilateral mastectomy is highly individual. "A woman needs to weigh up alternative options, including regular close monitoring and the use of tamoxifen or raloxifene, while considering the opportunities but also possible complications of reconstructive surgery. No statistics and no statistician can make this decision for her," Dr. Michels said.
Dr. Michels is at the Obstetrics and Gynecology Epidemiology Center at Brigham and Women’s Hospital and at Harvard Medical School, both in Boston. She reported no financial conflicts of interest. These remarks were taken from her editorial accompanying Dr. Metcalfe’s report (Br. Med. J. 2014 Feb. 11 [doi:10.1136/bmj.g1379]).
The main limitation of the study by Metcalfe and colleagues is the relatively small number of endpoints (breast cancer deaths), "which provide unstable estimates and make statistical inferences more challenging," wrote Dr. Karin B. Michels, Sc.D., Ph.D. As the authors noted, larger studies of this issue are needed, she said.
Breasts are not statistics; they are essential parts of women’s identity, sexuality, and self-perception, and the decision to undergo a bilateral mastectomy is highly individual. "A woman needs to weigh up alternative options, including regular close monitoring and the use of tamoxifen or raloxifene, while considering the opportunities but also possible complications of reconstructive surgery. No statistics and no statistician can make this decision for her," Dr. Michels said.
Dr. Michels is at the Obstetrics and Gynecology Epidemiology Center at Brigham and Women’s Hospital and at Harvard Medical School, both in Boston. She reported no financial conflicts of interest. These remarks were taken from her editorial accompanying Dr. Metcalfe’s report (Br. Med. J. 2014 Feb. 11 [doi:10.1136/bmj.g1379]).
Among early-stage breast cancer patients who carry BRCA mutations, prophylactic mastectomy of the contralateral breast appears to reduce the risk of dying from breast cancer within 20 years by 48%, according to a report published online Feb. 11 in the British Medical Journal.
This significant mortality benefit is most pronounced during the second decade after the initial breast cancer diagnosis, said Dr. Kelly Metcalfe of the University of Toronto, and her associates.
"We conclude that it is reasonable to propose bilateral mastectomy as the initial treatment option for women with early-stage breast cancer who are carriers of a BRCA1 or BRCA2 mutation. For those who have been treated in the past with unilateral mastectomy or breast-conserving surgery, the possibility of a second surgery should be discussed," they said.
Until now, the data regarding long-term survival after prophylactic mastectomy have been sparse, and no study has examined mortality specifically related to mastectomy of the contralateral breast. "Traditionally, breast cancer trialists and clinical epidemiologists focus their attention on the 10-year period after diagnosis, because this is when the majority of cancer-related deaths occur. However, a mortality benefit from preventing a second primary breast cancer is unlikely to be apparent within this narrow interval, given that second primary cancers accumulate slowly and for an extended period," Dr. Metcalfe and her colleagues said.
They reviewed the medical records and pathology reports of 390 women who had been diagnosed as having stage I or II breast cancer in 1975-2009 when they were aged 65 or younger, were known or likely carriers of a deleterious mutation in the BRCA1 or BRCA2 gene, and were treated with unilateral or bilateral mastectomy. These study participants were identified from the histories of 290 different families in which one member had received genetic counseling at any of 12 specialty clinics.
A total of 44 of the women were initially treated with bilateral mastectomy. The remaining 346 initially underwent unilateral mastectomy, but 137 of them went on to have mastectomy of the contralateral breast at a later time. Thus, in the final analysis 181 women had prophylactic mastectomy of the contralateral breast and 209 did not.
The mean follow-up was 13 years (range, 0.1-20.0 years).
Overall, 79 women (20% of the entire study population) died from breast cancer during follow-up: 18 of the 181 women who had prophylactic mastectomy of the contralateral breast (9.9%), compared with 61 of 209 who did not have prophylactic mastectomy (29.1%).
At 20 years, the survival rate for women who underwent prophylactic mastectomy was 88%, compared with a 66% survival rate among those who did not have prophylactic mastectomy of the contralateral breast.
In a multivariable analysis that controlled for age at diagnosis, tumor size, nodal status, treatment, and other prognostic factors, 20-year breast-cancer-specific mortality was 48% lower for those who underwent contralateral mastectomy than for those who did not (hazard ratio 0.52, 95% confidence interval 0.29 to 0.93; P = .03), the investigators wrote (Br. Med. J. 2014 Feb. 11 [doi:10.1136/bmj.g226]).
The reduction in mortality was much greater during the second decade of follow-up (80% reduction) than during the first decade (35% reduction). "On average, the time from first breast cancer to ... cancer of the contralateral breast is 5.7 years. Therefore, a delay in the observed benefit of contralateral mastectomy is to be expected – that is, the reduction in mortality results from a reduction in deaths from cancer of the contralateral breast," Dr. Metcalfe and her associates said.
They noted that it is critical that these findings be confirmed in other study populations, especially in view of their relatively small study population.
This study was funded by the Canadian Breast Cancer Foundation. Dr. Metcalfe is supported by the Canadian Institutes of Health Research and the Ontario Women’s Health Council. No financial conflicts of interest were reported.
Among early-stage breast cancer patients who carry BRCA mutations, prophylactic mastectomy of the contralateral breast appears to reduce the risk of dying from breast cancer within 20 years by 48%, according to a report published online Feb. 11 in the British Medical Journal.
This significant mortality benefit is most pronounced during the second decade after the initial breast cancer diagnosis, said Dr. Kelly Metcalfe of the University of Toronto, and her associates.
"We conclude that it is reasonable to propose bilateral mastectomy as the initial treatment option for women with early-stage breast cancer who are carriers of a BRCA1 or BRCA2 mutation. For those who have been treated in the past with unilateral mastectomy or breast-conserving surgery, the possibility of a second surgery should be discussed," they said.
Until now, the data regarding long-term survival after prophylactic mastectomy have been sparse, and no study has examined mortality specifically related to mastectomy of the contralateral breast. "Traditionally, breast cancer trialists and clinical epidemiologists focus their attention on the 10-year period after diagnosis, because this is when the majority of cancer-related deaths occur. However, a mortality benefit from preventing a second primary breast cancer is unlikely to be apparent within this narrow interval, given that second primary cancers accumulate slowly and for an extended period," Dr. Metcalfe and her colleagues said.
They reviewed the medical records and pathology reports of 390 women who had been diagnosed as having stage I or II breast cancer in 1975-2009 when they were aged 65 or younger, were known or likely carriers of a deleterious mutation in the BRCA1 or BRCA2 gene, and were treated with unilateral or bilateral mastectomy. These study participants were identified from the histories of 290 different families in which one member had received genetic counseling at any of 12 specialty clinics.
A total of 44 of the women were initially treated with bilateral mastectomy. The remaining 346 initially underwent unilateral mastectomy, but 137 of them went on to have mastectomy of the contralateral breast at a later time. Thus, in the final analysis 181 women had prophylactic mastectomy of the contralateral breast and 209 did not.
The mean follow-up was 13 years (range, 0.1-20.0 years).
Overall, 79 women (20% of the entire study population) died from breast cancer during follow-up: 18 of the 181 women who had prophylactic mastectomy of the contralateral breast (9.9%), compared with 61 of 209 who did not have prophylactic mastectomy (29.1%).
At 20 years, the survival rate for women who underwent prophylactic mastectomy was 88%, compared with a 66% survival rate among those who did not have prophylactic mastectomy of the contralateral breast.
In a multivariable analysis that controlled for age at diagnosis, tumor size, nodal status, treatment, and other prognostic factors, 20-year breast-cancer-specific mortality was 48% lower for those who underwent contralateral mastectomy than for those who did not (hazard ratio 0.52, 95% confidence interval 0.29 to 0.93; P = .03), the investigators wrote (Br. Med. J. 2014 Feb. 11 [doi:10.1136/bmj.g226]).
The reduction in mortality was much greater during the second decade of follow-up (80% reduction) than during the first decade (35% reduction). "On average, the time from first breast cancer to ... cancer of the contralateral breast is 5.7 years. Therefore, a delay in the observed benefit of contralateral mastectomy is to be expected – that is, the reduction in mortality results from a reduction in deaths from cancer of the contralateral breast," Dr. Metcalfe and her associates said.
They noted that it is critical that these findings be confirmed in other study populations, especially in view of their relatively small study population.
This study was funded by the Canadian Breast Cancer Foundation. Dr. Metcalfe is supported by the Canadian Institutes of Health Research and the Ontario Women’s Health Council. No financial conflicts of interest were reported.
FROM THE BRITISH MEDICAL JOURNAL
Major finding: Twenty-year breast-cancer-specific mortality was 48% lower for women with BRCA mutations who underwent prophylactic contralateral mastectomy than for those who did not (hazard ratio 0.52, 95% confidence interval 0.29 to 0.93; P = .03).
Data source: A retrospective analysis of breast-cancer-specific mortality in 390 women with BRCA1 or BRCA2 mutations who underwent mastectomy for stage I or II breast cancer, then either underwent or did not undergo prophylactic mastectomy of the contralateral breast, and were followed for up to 20 years.
Disclosures: This study was funded by the Canadian Breast Cancer Foundation. Dr. Metcalfe is supported by the Canadian Institutes of Health Research and the Ontario Women’s Health Council. No financial conflicts of interest were reported.
Obesity at age 20 associated with increased risk of multiple sclerosis
Multiple sclerosis shows a "striking" association with obesity at age 20 years that strongly interacts with genetic susceptibility, according to an analysis of data from two case-control studies that examined environmental and genetic risk factors for MS.
This relationship between adolescent obesity and MS is of the same magnitude as the separate associations between MS and carriage of the high-risk HLA-DRB1*15 allele, absence of the protective HLA-A*02 allele, and smoking, said Dr. Anna Karin Hedström of the Institute of Environmental Medicine, Karolinska Institutet, Stockholm, and her associates.
"The biological explanations for these interactions are far from clear, but the data open [the way] for mechanistically oriented studies," they said.
Three previous studies have suggested that obesity in early life may be linked to increased risk of developing MS later. Dr. Hedström and her colleagues examined this association using data from a Swedish population-based, case-control study and from a separate American case-control study.
In the Swedish study, 1,510 adults with incident MS who were treated at 40 clinics across the country during a 7-year period and 2,017 control subjects completed detailed questionnaires concerning environmental exposures and other factors. The controls were matched for age, sex, and area of residence, and all the participants gave blood samples for HLA typing.
The American study involved 937 white adults with prevalent MS who were members of a single large health maintenance organization covering northern California and 609 white control subjects matched for age, sex, and area of residence. All the participants completed computer-assisted telephone interviews regarding environmental exposures and lifestyle factors.
All the subjects in both studies reported what their heights and weights had been at age 20 years, from which the investigators calculated body mass index (BMI).
In both studies, participants whose BMI at age 20 years was 27 kg/m2 or greater showed an increased risk of developing MS later in life, compared with those whose BMI was 18.5-21 kg/m2. The odds ratios (ORs) were 2.2 for subjects in the Swedish study and 1.8 for those in the American study, Dr. Hedström and her associates said (Neurology 2014 [doi:10.1212/WNL.0000000000000203]).
Similarly, participants with a slightly lower but still above-normal BMI of 25-27 kg/m2 showed a modestly increased risk of developing MS later in life: The ORs were 1.4 in the Swedish study and 1.3 in the American study.
These ORs were unchanged when a sensitivity analysis was performed, including only the study subjects who had been genotyped.
Participants who carried the high-risk HLA-DRB1*15 gene, did not carry the protective HLA-A*02 gene, and had a BMI of 27 kg/ m2 or greater at age 20 years had an OR of 16.2 for developing later MS, compared with those who had none of those risk factors. In contrast, subjects who had the same HLA profile but had not been obese at age 20 years had an OR of only 5.1.
The investigators proposed that the low-grade chronic inflammation associated with obesity, together with obesity’s adverse effects on autoimmunity, may raise the risk of HLA-related activation of T cells that attack the CNS.
Both the Swedish and the American study were limited in that they were retrospective and relied on participants’ self-reports. In addition, Dr. Hedström and her associates modified the usual definition of obesity for the purposes of their study. The typical standard for obesity is a BMI of greater than 30 kg/m2, not greater than 27 kg/m2. However, the number of subjects at this level of BMI was too small in the Swedish cohort to allow accurate analysis, so the researchers combined the top two categories of BMI into one designation of "obese."
This study was supported by several private nonprofit foundations, the Swedish Research Council for Health, Working Life and Welfare, and the U.S. National Institute of Neurological Disorders and Stroke. Dr. Hedström and five of the other seven authors reported no financial conflicts of interest. One coauthor reported ties to numerous industry sources and one reported receiving research support from Swedish government agencies.
This study and other research linking obesity
early in life with the later development of MS are observational and cannot
establish causality, so we don’t yet know whether decreasing obesity with diet
and exercise will lead to a decrease in the incidence of MS.
Nevertheless, these findings are concerning. “It is
time to begin developing a targeted approach to prevent MS by improving common
health behaviors, including body weight and smoking,” Dr. Marrie and Dr. Beck
said.
People who are at high genetic risk can be readily
identified by focusing HLA genotyping on the first-degree relatives of known MS
patients, they added.
Dr. Marrie is in the departments of internal medicine and
community health sciences at the University of Manitoba, Winnipeg. She has
received funding from Sanofi-Aventis and Canadian governmental agencies and
nonprofit organizations, including the MS Society of Canada and the MS
Scientific Foundation. Dr. Beck is in the department of biostatistics and
computational biology at the University of Rochester (N.Y.) Medical Center. Dr.
Beck has received support from a variety of healthcare companies as well the
U.S. Food and Drug Administration and the U.S. National Institutes of Health.
These remarks were taken from their editorial accompanying Dr. Hedström’s report (Neurology 2014 [doi:10.1212/WNL.0000000000000195]).
This study and other research linking obesity
early in life with the later development of MS are observational and cannot
establish causality, so we don’t yet know whether decreasing obesity with diet
and exercise will lead to a decrease in the incidence of MS.
Nevertheless, these findings are concerning. “It is
time to begin developing a targeted approach to prevent MS by improving common
health behaviors, including body weight and smoking,” Dr. Marrie and Dr. Beck
said.
People who are at high genetic risk can be readily
identified by focusing HLA genotyping on the first-degree relatives of known MS
patients, they added.
Dr. Marrie is in the departments of internal medicine and
community health sciences at the University of Manitoba, Winnipeg. She has
received funding from Sanofi-Aventis and Canadian governmental agencies and
nonprofit organizations, including the MS Society of Canada and the MS
Scientific Foundation. Dr. Beck is in the department of biostatistics and
computational biology at the University of Rochester (N.Y.) Medical Center. Dr.
Beck has received support from a variety of healthcare companies as well the
U.S. Food and Drug Administration and the U.S. National Institutes of Health.
These remarks were taken from their editorial accompanying Dr. Hedström’s report (Neurology 2014 [doi:10.1212/WNL.0000000000000195]).
This study and other research linking obesity
early in life with the later development of MS are observational and cannot
establish causality, so we don’t yet know whether decreasing obesity with diet
and exercise will lead to a decrease in the incidence of MS.
Nevertheless, these findings are concerning. “It is
time to begin developing a targeted approach to prevent MS by improving common
health behaviors, including body weight and smoking,” Dr. Marrie and Dr. Beck
said.
People who are at high genetic risk can be readily
identified by focusing HLA genotyping on the first-degree relatives of known MS
patients, they added.
Dr. Marrie is in the departments of internal medicine and
community health sciences at the University of Manitoba, Winnipeg. She has
received funding from Sanofi-Aventis and Canadian governmental agencies and
nonprofit organizations, including the MS Society of Canada and the MS
Scientific Foundation. Dr. Beck is in the department of biostatistics and
computational biology at the University of Rochester (N.Y.) Medical Center. Dr.
Beck has received support from a variety of healthcare companies as well the
U.S. Food and Drug Administration and the U.S. National Institutes of Health.
These remarks were taken from their editorial accompanying Dr. Hedström’s report (Neurology 2014 [doi:10.1212/WNL.0000000000000195]).
Multiple sclerosis shows a "striking" association with obesity at age 20 years that strongly interacts with genetic susceptibility, according to an analysis of data from two case-control studies that examined environmental and genetic risk factors for MS.
This relationship between adolescent obesity and MS is of the same magnitude as the separate associations between MS and carriage of the high-risk HLA-DRB1*15 allele, absence of the protective HLA-A*02 allele, and smoking, said Dr. Anna Karin Hedström of the Institute of Environmental Medicine, Karolinska Institutet, Stockholm, and her associates.
"The biological explanations for these interactions are far from clear, but the data open [the way] for mechanistically oriented studies," they said.
Three previous studies have suggested that obesity in early life may be linked to increased risk of developing MS later. Dr. Hedström and her colleagues examined this association using data from a Swedish population-based, case-control study and from a separate American case-control study.
In the Swedish study, 1,510 adults with incident MS who were treated at 40 clinics across the country during a 7-year period and 2,017 control subjects completed detailed questionnaires concerning environmental exposures and other factors. The controls were matched for age, sex, and area of residence, and all the participants gave blood samples for HLA typing.
The American study involved 937 white adults with prevalent MS who were members of a single large health maintenance organization covering northern California and 609 white control subjects matched for age, sex, and area of residence. All the participants completed computer-assisted telephone interviews regarding environmental exposures and lifestyle factors.
All the subjects in both studies reported what their heights and weights had been at age 20 years, from which the investigators calculated body mass index (BMI).
In both studies, participants whose BMI at age 20 years was 27 kg/m2 or greater showed an increased risk of developing MS later in life, compared with those whose BMI was 18.5-21 kg/m2. The odds ratios (ORs) were 2.2 for subjects in the Swedish study and 1.8 for those in the American study, Dr. Hedström and her associates said (Neurology 2014 [doi:10.1212/WNL.0000000000000203]).
Similarly, participants with a slightly lower but still above-normal BMI of 25-27 kg/m2 showed a modestly increased risk of developing MS later in life: The ORs were 1.4 in the Swedish study and 1.3 in the American study.
These ORs were unchanged when a sensitivity analysis was performed, including only the study subjects who had been genotyped.
Participants who carried the high-risk HLA-DRB1*15 gene, did not carry the protective HLA-A*02 gene, and had a BMI of 27 kg/ m2 or greater at age 20 years had an OR of 16.2 for developing later MS, compared with those who had none of those risk factors. In contrast, subjects who had the same HLA profile but had not been obese at age 20 years had an OR of only 5.1.
The investigators proposed that the low-grade chronic inflammation associated with obesity, together with obesity’s adverse effects on autoimmunity, may raise the risk of HLA-related activation of T cells that attack the CNS.
Both the Swedish and the American study were limited in that they were retrospective and relied on participants’ self-reports. In addition, Dr. Hedström and her associates modified the usual definition of obesity for the purposes of their study. The typical standard for obesity is a BMI of greater than 30 kg/m2, not greater than 27 kg/m2. However, the number of subjects at this level of BMI was too small in the Swedish cohort to allow accurate analysis, so the researchers combined the top two categories of BMI into one designation of "obese."
This study was supported by several private nonprofit foundations, the Swedish Research Council for Health, Working Life and Welfare, and the U.S. National Institute of Neurological Disorders and Stroke. Dr. Hedström and five of the other seven authors reported no financial conflicts of interest. One coauthor reported ties to numerous industry sources and one reported receiving research support from Swedish government agencies.
Multiple sclerosis shows a "striking" association with obesity at age 20 years that strongly interacts with genetic susceptibility, according to an analysis of data from two case-control studies that examined environmental and genetic risk factors for MS.
This relationship between adolescent obesity and MS is of the same magnitude as the separate associations between MS and carriage of the high-risk HLA-DRB1*15 allele, absence of the protective HLA-A*02 allele, and smoking, said Dr. Anna Karin Hedström of the Institute of Environmental Medicine, Karolinska Institutet, Stockholm, and her associates.
"The biological explanations for these interactions are far from clear, but the data open [the way] for mechanistically oriented studies," they said.
Three previous studies have suggested that obesity in early life may be linked to increased risk of developing MS later. Dr. Hedström and her colleagues examined this association using data from a Swedish population-based, case-control study and from a separate American case-control study.
In the Swedish study, 1,510 adults with incident MS who were treated at 40 clinics across the country during a 7-year period and 2,017 control subjects completed detailed questionnaires concerning environmental exposures and other factors. The controls were matched for age, sex, and area of residence, and all the participants gave blood samples for HLA typing.
The American study involved 937 white adults with prevalent MS who were members of a single large health maintenance organization covering northern California and 609 white control subjects matched for age, sex, and area of residence. All the participants completed computer-assisted telephone interviews regarding environmental exposures and lifestyle factors.
All the subjects in both studies reported what their heights and weights had been at age 20 years, from which the investigators calculated body mass index (BMI).
In both studies, participants whose BMI at age 20 years was 27 kg/m2 or greater showed an increased risk of developing MS later in life, compared with those whose BMI was 18.5-21 kg/m2. The odds ratios (ORs) were 2.2 for subjects in the Swedish study and 1.8 for those in the American study, Dr. Hedström and her associates said (Neurology 2014 [doi:10.1212/WNL.0000000000000203]).
Similarly, participants with a slightly lower but still above-normal BMI of 25-27 kg/m2 showed a modestly increased risk of developing MS later in life: The ORs were 1.4 in the Swedish study and 1.3 in the American study.
These ORs were unchanged when a sensitivity analysis was performed, including only the study subjects who had been genotyped.
Participants who carried the high-risk HLA-DRB1*15 gene, did not carry the protective HLA-A*02 gene, and had a BMI of 27 kg/ m2 or greater at age 20 years had an OR of 16.2 for developing later MS, compared with those who had none of those risk factors. In contrast, subjects who had the same HLA profile but had not been obese at age 20 years had an OR of only 5.1.
The investigators proposed that the low-grade chronic inflammation associated with obesity, together with obesity’s adverse effects on autoimmunity, may raise the risk of HLA-related activation of T cells that attack the CNS.
Both the Swedish and the American study were limited in that they were retrospective and relied on participants’ self-reports. In addition, Dr. Hedström and her associates modified the usual definition of obesity for the purposes of their study. The typical standard for obesity is a BMI of greater than 30 kg/m2, not greater than 27 kg/m2. However, the number of subjects at this level of BMI was too small in the Swedish cohort to allow accurate analysis, so the researchers combined the top two categories of BMI into one designation of "obese."
This study was supported by several private nonprofit foundations, the Swedish Research Council for Health, Working Life and Welfare, and the U.S. National Institute of Neurological Disorders and Stroke. Dr. Hedström and five of the other seven authors reported no financial conflicts of interest. One coauthor reported ties to numerous industry sources and one reported receiving research support from Swedish government agencies.
FROM NEUROLOGY
Major finding: Participants who carried the high-risk HLA-DRB1*15 gene, did not carry the protective HLA-A*02 gene, and had a BMI of 27 kg/ m2 or greater at age 20 years had an OR of 16.2 for developing later MS, compared with those who had none of those risk factors.
Data source: A secondary analysis of data from two case-control studies in Sweden (1,798 cases and 3,907 controls) and the United States (1,087 cases and 687 controls), comparing BMI at age 20 years between people who later developed MS and controls subjects.
Disclosures: This study was supported by the Swedish Research Council for Health, Working Life and Welfare, the Knut and Alice Wallenberg Foundation, the AFA Foundation, the Swedish Brain Foundation, the Swedish Association for Persons With Neurological Disabilities, and the U.S. National Institute of Neurological Disorders and Stroke. Dr. Hedström and five of the other seven authors reported no financial conflicts of interest. One coauthor reported ties to numerous industry sources and one reported receiving research support form Swedish government agencies.