User login
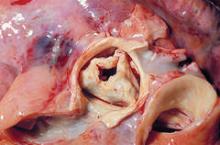
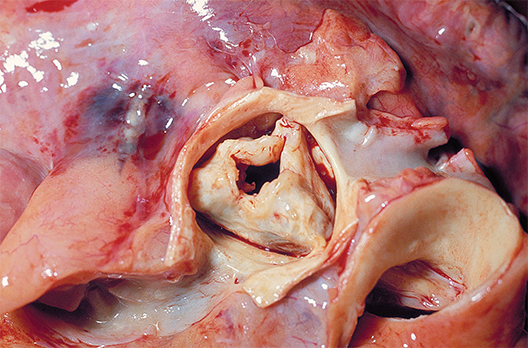
DECAAF: Assess extent of fibrosis before AF ablation
For patients scheduled to undergo AF catheter ablation, estimating the extent of atrial fibrosis using delayed enhancement MRI can help distinguish those likely to respond from patients likely to have recurrent arrhythmia, according to a report published online Feb. 4 in JAMA.
In the prospective, observational DECAAF (Delayed-Enhancement MRI Determinant of Successful Radiofrequency Ablation of Atrial Fibrillation) study, 260 such patients (mean age, 59 years) underwent quantification of left atrial fibrosis via delayed enhancement MRI with gadolinium at 15 medical centers in six countries, before undergoing AF ablation. These centers had varying levels of experience with cardiac imaging and used different ablation procedures, said Dr. Nassir F. Marrouche, director of the comprehensive arrhythmia and research management center, University of Utah, Salt Lake City, and his associates.
Four categories of fibrosis were used: involvement of less than 10% of the atrial wall (stage 1, 49 patients), 10%-19% of the atrial wall (stage 2, 107 patients), 20%-29% of the atrial wall (stage 3, 80 patients), and 30% or more of the atrial wall (stage 4, 24 patients). The estimated percentage of atrial fibrosis strongly correlated with arrhythmia recurrence at 1 year, even after the data were adjusted to account for variables such as patient age and sex; the presence of hypertension, heart failure, mitral valve disease, or diabetes; and the type of AF (paroxysmal vs persistent).
The hazard ratio for recurrent arrhythmia was 1.06 for every 1% increase in the extent of atrial fibrosis (JAMA 2014 Feb. 4 [doi:10.1001/jama.2014.3]).
This is the first multicenter study to demonstrate the feasibility and potential clinical value of quantifying the degree of AF fibrosis using delayed enhancement MRI before performing ablation, offering a noninvasive, effective method for determining which patients are likely to benefit and which should avoid the procedure, Dr. Marrouche and his associates said.
The JAMA report expands on results presented at the annual meeting of the Heart Rhythm Society last year.
The study was funded by the Comprehensive Arrhythmia and Research Management Center at the University of Utah and the George S. and Dolores Dore Eccles Foundation. Dr. Marrouche reported owning stock and being named in two patents licensed to Marrek; his associates reported numerous ties to industry sources.
For patients scheduled to undergo AF catheter ablation, estimating the extent of atrial fibrosis using delayed enhancement MRI can help distinguish those likely to respond from patients likely to have recurrent arrhythmia, according to a report published online Feb. 4 in JAMA.
In the prospective, observational DECAAF (Delayed-Enhancement MRI Determinant of Successful Radiofrequency Ablation of Atrial Fibrillation) study, 260 such patients (mean age, 59 years) underwent quantification of left atrial fibrosis via delayed enhancement MRI with gadolinium at 15 medical centers in six countries, before undergoing AF ablation. These centers had varying levels of experience with cardiac imaging and used different ablation procedures, said Dr. Nassir F. Marrouche, director of the comprehensive arrhythmia and research management center, University of Utah, Salt Lake City, and his associates.
Four categories of fibrosis were used: involvement of less than 10% of the atrial wall (stage 1, 49 patients), 10%-19% of the atrial wall (stage 2, 107 patients), 20%-29% of the atrial wall (stage 3, 80 patients), and 30% or more of the atrial wall (stage 4, 24 patients). The estimated percentage of atrial fibrosis strongly correlated with arrhythmia recurrence at 1 year, even after the data were adjusted to account for variables such as patient age and sex; the presence of hypertension, heart failure, mitral valve disease, or diabetes; and the type of AF (paroxysmal vs persistent).
The hazard ratio for recurrent arrhythmia was 1.06 for every 1% increase in the extent of atrial fibrosis (JAMA 2014 Feb. 4 [doi:10.1001/jama.2014.3]).
This is the first multicenter study to demonstrate the feasibility and potential clinical value of quantifying the degree of AF fibrosis using delayed enhancement MRI before performing ablation, offering a noninvasive, effective method for determining which patients are likely to benefit and which should avoid the procedure, Dr. Marrouche and his associates said.
The JAMA report expands on results presented at the annual meeting of the Heart Rhythm Society last year.
The study was funded by the Comprehensive Arrhythmia and Research Management Center at the University of Utah and the George S. and Dolores Dore Eccles Foundation. Dr. Marrouche reported owning stock and being named in two patents licensed to Marrek; his associates reported numerous ties to industry sources.
For patients scheduled to undergo AF catheter ablation, estimating the extent of atrial fibrosis using delayed enhancement MRI can help distinguish those likely to respond from patients likely to have recurrent arrhythmia, according to a report published online Feb. 4 in JAMA.
In the prospective, observational DECAAF (Delayed-Enhancement MRI Determinant of Successful Radiofrequency Ablation of Atrial Fibrillation) study, 260 such patients (mean age, 59 years) underwent quantification of left atrial fibrosis via delayed enhancement MRI with gadolinium at 15 medical centers in six countries, before undergoing AF ablation. These centers had varying levels of experience with cardiac imaging and used different ablation procedures, said Dr. Nassir F. Marrouche, director of the comprehensive arrhythmia and research management center, University of Utah, Salt Lake City, and his associates.
Four categories of fibrosis were used: involvement of less than 10% of the atrial wall (stage 1, 49 patients), 10%-19% of the atrial wall (stage 2, 107 patients), 20%-29% of the atrial wall (stage 3, 80 patients), and 30% or more of the atrial wall (stage 4, 24 patients). The estimated percentage of atrial fibrosis strongly correlated with arrhythmia recurrence at 1 year, even after the data were adjusted to account for variables such as patient age and sex; the presence of hypertension, heart failure, mitral valve disease, or diabetes; and the type of AF (paroxysmal vs persistent).
The hazard ratio for recurrent arrhythmia was 1.06 for every 1% increase in the extent of atrial fibrosis (JAMA 2014 Feb. 4 [doi:10.1001/jama.2014.3]).
This is the first multicenter study to demonstrate the feasibility and potential clinical value of quantifying the degree of AF fibrosis using delayed enhancement MRI before performing ablation, offering a noninvasive, effective method for determining which patients are likely to benefit and which should avoid the procedure, Dr. Marrouche and his associates said.
The JAMA report expands on results presented at the annual meeting of the Heart Rhythm Society last year.
The study was funded by the Comprehensive Arrhythmia and Research Management Center at the University of Utah and the George S. and Dolores Dore Eccles Foundation. Dr. Marrouche reported owning stock and being named in two patents licensed to Marrek; his associates reported numerous ties to industry sources.
Chinese herbal remedy found noninferior to methotrexate in RA patients
A traditional Chinese herbal remedy used to treat joint pain, fever, edema, and local inflammation was found noninferior to methotrexate for controlling active rheumatoid arthritis in an open-label, randomized clinical trial involving 207 patients.
Tripterygium wilfordii Hook F (TwHF) is a relatively inexpensive remedy (about $10 per month) often used alone or in combination with methotrexate for rheumatoid arthritis (RA) in China, but it has not been assessed in a rigorous controlled study until now.
In this trial, patients at nine general hospitals in China were randomly assigned in a nonblinded fashion to receive 6 months of oral TwHF pills three times per day (69 patients), oral methotrexate once per week (69 patients), or TwHF plus methotrexate (69 patients), in addition to their stable doses of NSAIDs and oral corticosteroids, said Qian-wen Lv of the department of rheumatology and clinical immunology, Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, and associates.
The primary efficacy measure was the proportion of patients who achieved an ACR50, or an American College of Rheumatology response of at least 50% improvement in tender and swollen joints and an improvement of at least 50% in three or more of the following: evaluator’s assessment of global health status, patient’s assessment of global health status, patient’s assessment of pain on a visual analog scale, patient’s assessment of function using the Health Assessment Questionnaire, and ESR or CRP level. This rate was 55.1% in the TwHF group, which was noninferior to the 46.4% rate in the methotrexate group. The combination of TwHF plus methotrexate was the most effective, with an ACR50 response rate of 76.8%.
A similar pattern was seen in secondary efficacy measures such as the remission rate, the ACR20 response, the ACR70 response, "good" responses on the Clinical Disease Activity Index, "good" responses on EULAR criteria, and the rate of Low Disease Activity. This pattern also persisted in a per-protocol analysis that excluded the 33 patients who withdrew from the study: The ACR50 response was 65.3% for TwHF alone, 59.2% for methotrexate alone, and 80.4% for combined TwHF plus methotrexate among patients who completed the study, the investigators said (Ann. Rheum. Dis. 2014 April 14 [doi:10.1136/annrheumdis.2013.204807]).
The main difference among the treatment groups in laboratory analyses was that TwHF monotherapy induced a much more rapid reduction in ESR than did methotrexate monotherapy. The numbers of adverse effects and severe adverse effects were comparable between TwHF and methotrexate, except that TwHF was associated with a slightly higher rate of irregular menses. This antifertility effect of the herbal remedy is well known in China, and study participants were warned about it before enrollment. It is usually reversible after treatment is discontinued, the investigators said.
This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators declared having no financial conflicts of interest.
A traditional Chinese herbal remedy used to treat joint pain, fever, edema, and local inflammation was found noninferior to methotrexate for controlling active rheumatoid arthritis in an open-label, randomized clinical trial involving 207 patients.
Tripterygium wilfordii Hook F (TwHF) is a relatively inexpensive remedy (about $10 per month) often used alone or in combination with methotrexate for rheumatoid arthritis (RA) in China, but it has not been assessed in a rigorous controlled study until now.
In this trial, patients at nine general hospitals in China were randomly assigned in a nonblinded fashion to receive 6 months of oral TwHF pills three times per day (69 patients), oral methotrexate once per week (69 patients), or TwHF plus methotrexate (69 patients), in addition to their stable doses of NSAIDs and oral corticosteroids, said Qian-wen Lv of the department of rheumatology and clinical immunology, Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, and associates.
The primary efficacy measure was the proportion of patients who achieved an ACR50, or an American College of Rheumatology response of at least 50% improvement in tender and swollen joints and an improvement of at least 50% in three or more of the following: evaluator’s assessment of global health status, patient’s assessment of global health status, patient’s assessment of pain on a visual analog scale, patient’s assessment of function using the Health Assessment Questionnaire, and ESR or CRP level. This rate was 55.1% in the TwHF group, which was noninferior to the 46.4% rate in the methotrexate group. The combination of TwHF plus methotrexate was the most effective, with an ACR50 response rate of 76.8%.
A similar pattern was seen in secondary efficacy measures such as the remission rate, the ACR20 response, the ACR70 response, "good" responses on the Clinical Disease Activity Index, "good" responses on EULAR criteria, and the rate of Low Disease Activity. This pattern also persisted in a per-protocol analysis that excluded the 33 patients who withdrew from the study: The ACR50 response was 65.3% for TwHF alone, 59.2% for methotrexate alone, and 80.4% for combined TwHF plus methotrexate among patients who completed the study, the investigators said (Ann. Rheum. Dis. 2014 April 14 [doi:10.1136/annrheumdis.2013.204807]).
The main difference among the treatment groups in laboratory analyses was that TwHF monotherapy induced a much more rapid reduction in ESR than did methotrexate monotherapy. The numbers of adverse effects and severe adverse effects were comparable between TwHF and methotrexate, except that TwHF was associated with a slightly higher rate of irregular menses. This antifertility effect of the herbal remedy is well known in China, and study participants were warned about it before enrollment. It is usually reversible after treatment is discontinued, the investigators said.
This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators declared having no financial conflicts of interest.
A traditional Chinese herbal remedy used to treat joint pain, fever, edema, and local inflammation was found noninferior to methotrexate for controlling active rheumatoid arthritis in an open-label, randomized clinical trial involving 207 patients.
Tripterygium wilfordii Hook F (TwHF) is a relatively inexpensive remedy (about $10 per month) often used alone or in combination with methotrexate for rheumatoid arthritis (RA) in China, but it has not been assessed in a rigorous controlled study until now.
In this trial, patients at nine general hospitals in China were randomly assigned in a nonblinded fashion to receive 6 months of oral TwHF pills three times per day (69 patients), oral methotrexate once per week (69 patients), or TwHF plus methotrexate (69 patients), in addition to their stable doses of NSAIDs and oral corticosteroids, said Qian-wen Lv of the department of rheumatology and clinical immunology, Peking Union Medical College and the Chinese Academy of Medical Sciences, Beijing, and associates.
The primary efficacy measure was the proportion of patients who achieved an ACR50, or an American College of Rheumatology response of at least 50% improvement in tender and swollen joints and an improvement of at least 50% in three or more of the following: evaluator’s assessment of global health status, patient’s assessment of global health status, patient’s assessment of pain on a visual analog scale, patient’s assessment of function using the Health Assessment Questionnaire, and ESR or CRP level. This rate was 55.1% in the TwHF group, which was noninferior to the 46.4% rate in the methotrexate group. The combination of TwHF plus methotrexate was the most effective, with an ACR50 response rate of 76.8%.
A similar pattern was seen in secondary efficacy measures such as the remission rate, the ACR20 response, the ACR70 response, "good" responses on the Clinical Disease Activity Index, "good" responses on EULAR criteria, and the rate of Low Disease Activity. This pattern also persisted in a per-protocol analysis that excluded the 33 patients who withdrew from the study: The ACR50 response was 65.3% for TwHF alone, 59.2% for methotrexate alone, and 80.4% for combined TwHF plus methotrexate among patients who completed the study, the investigators said (Ann. Rheum. Dis. 2014 April 14 [doi:10.1136/annrheumdis.2013.204807]).
The main difference among the treatment groups in laboratory analyses was that TwHF monotherapy induced a much more rapid reduction in ESR than did methotrexate monotherapy. The numbers of adverse effects and severe adverse effects were comparable between TwHF and methotrexate, except that TwHF was associated with a slightly higher rate of irregular menses. This antifertility effect of the herbal remedy is well known in China, and study participants were warned about it before enrollment. It is usually reversible after treatment is discontinued, the investigators said.
This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators declared having no financial conflicts of interest.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major Finding: The primary efficacy measure, the proportion of patients who achieved an ACR50 response, was 55.1% with TwHF alone, 46.4% with methotrexate alone, and 76.8% with combined TwHF plus methotrexate.
Data Source: A multicenter, open-label, randomized clinical trial involving 207 patients with active RA who were treated for 6 months with TwHF alone, methotrexate alone, or TwHF plus methotrexate.
Disclosures: This study was supported by the National Natural Science Foundation of China, the Beijing Municipal Natural Science Foundation, the Research Special Fund for Public Welfare Industry of Health, the Capital Health Research and Development of Special Fund, and the National Laboratory Special Fund. The investigators reported no financial conflicts of interest.
Intervention manages cardiac patients with depression, anxiety
A low-intensity intervention aimed at managing cardiac patients who have concomitant depression or anxiety improved mental-health–related quality of life when compared with usual care, according to a report published online April 14 in JAMA Internal Medicine.
The MOSAIC (Management of Sadness and Anxiety in Cardiology) clinical trial involved 183 patients hospitalized at a single urban academic medical center for acute coronary syndrome, heart failure, or arrhythmia during a 2-year period and who were found to have coexisting depression, generalized anxiety disorder, and/or panic disorder. The mean age of the participants was 60.5 years, 90% were white, 61% were employed, 53% were women (JAMA Intern. Med. 2014 April 14 [doi:10.1001/jamainternmend.2014.739]).
They were randomly assigned to receive either usual care or a telephone-based intervention in which a part-time social work care manager coordinated care among psychiatrists, inpatient medical providers, and outpatient medical providers; provided cognitive-behavioral therapy specific to the patient’s condition; and monitored patient symptoms for the 6-month duration of the intervention, said Dr. Jeff C. Huffman, director of cardiac psychiatry research program at Massachusetts General Hospital, and who is in the department of psychiatry at Harvard Medical School, Boston, and his associates.
Patients in the intervention group showed a significantly greater improvement than did those who received usual care in the primary outcome measure: mental-health-related quality of life (QOL), as measured by the Medical Outcomes Study Short Form-12 Mental Component Score. They also were remarkably more likely to receive treatment deemed "adequate" for their psychiatric disorders (75% vs 7%), and to show significantly greater improvement in Patient Health Questionnaire-9 scores; in overall function, as measured on the Duke Activity Status Index; and in health care–related QOL, as measured by EuroQol 5-Domain Instrument scores.
No significant differences were found between the two study groups in cardiac readmission rates at 6 months, but the mean time to readmission was significantly longer for the intervention group (92.4 days) than for the usual care group (62.5 days), Dr. Huffman and his associates said.
The investigators noted that their study involved typical patients seen for cardiac care – including many with serious medical issues and some who declined psychiatric treatment – and so should reflect results that would be found in real-world settings. In addition, using a social worker instead of a nurse as care manager and using telephone rather than in-person contacts substantially saved on costs.
However, Dr. Huffman and his colleagues cited several limitations. For example, the study was conducted in an academic medical center with mostly white patients. Also, those who delivered the intervention had experience with that population and with collaborative care programs.
Still, they expressed optimism about the intervention’s potential. "This intervention seems to have substantial promise as an adjunct or alternative to standard [collaborative care] paradigms," they wrote. "We found that a single care manager was able to coordinate care of three psychiatric conditions in patients with a wide range of cardiac diagnoses living within and outside the metropolitan area of the hospital."
This work was supported in part by the American Heart Association. No relevant financial conflicts of interest were reported.
A low-intensity intervention aimed at managing cardiac patients who have concomitant depression or anxiety improved mental-health–related quality of life when compared with usual care, according to a report published online April 14 in JAMA Internal Medicine.
The MOSAIC (Management of Sadness and Anxiety in Cardiology) clinical trial involved 183 patients hospitalized at a single urban academic medical center for acute coronary syndrome, heart failure, or arrhythmia during a 2-year period and who were found to have coexisting depression, generalized anxiety disorder, and/or panic disorder. The mean age of the participants was 60.5 years, 90% were white, 61% were employed, 53% were women (JAMA Intern. Med. 2014 April 14 [doi:10.1001/jamainternmend.2014.739]).
They were randomly assigned to receive either usual care or a telephone-based intervention in which a part-time social work care manager coordinated care among psychiatrists, inpatient medical providers, and outpatient medical providers; provided cognitive-behavioral therapy specific to the patient’s condition; and monitored patient symptoms for the 6-month duration of the intervention, said Dr. Jeff C. Huffman, director of cardiac psychiatry research program at Massachusetts General Hospital, and who is in the department of psychiatry at Harvard Medical School, Boston, and his associates.
Patients in the intervention group showed a significantly greater improvement than did those who received usual care in the primary outcome measure: mental-health-related quality of life (QOL), as measured by the Medical Outcomes Study Short Form-12 Mental Component Score. They also were remarkably more likely to receive treatment deemed "adequate" for their psychiatric disorders (75% vs 7%), and to show significantly greater improvement in Patient Health Questionnaire-9 scores; in overall function, as measured on the Duke Activity Status Index; and in health care–related QOL, as measured by EuroQol 5-Domain Instrument scores.
No significant differences were found between the two study groups in cardiac readmission rates at 6 months, but the mean time to readmission was significantly longer for the intervention group (92.4 days) than for the usual care group (62.5 days), Dr. Huffman and his associates said.
The investigators noted that their study involved typical patients seen for cardiac care – including many with serious medical issues and some who declined psychiatric treatment – and so should reflect results that would be found in real-world settings. In addition, using a social worker instead of a nurse as care manager and using telephone rather than in-person contacts substantially saved on costs.
However, Dr. Huffman and his colleagues cited several limitations. For example, the study was conducted in an academic medical center with mostly white patients. Also, those who delivered the intervention had experience with that population and with collaborative care programs.
Still, they expressed optimism about the intervention’s potential. "This intervention seems to have substantial promise as an adjunct or alternative to standard [collaborative care] paradigms," they wrote. "We found that a single care manager was able to coordinate care of three psychiatric conditions in patients with a wide range of cardiac diagnoses living within and outside the metropolitan area of the hospital."
This work was supported in part by the American Heart Association. No relevant financial conflicts of interest were reported.
A low-intensity intervention aimed at managing cardiac patients who have concomitant depression or anxiety improved mental-health–related quality of life when compared with usual care, according to a report published online April 14 in JAMA Internal Medicine.
The MOSAIC (Management of Sadness and Anxiety in Cardiology) clinical trial involved 183 patients hospitalized at a single urban academic medical center for acute coronary syndrome, heart failure, or arrhythmia during a 2-year period and who were found to have coexisting depression, generalized anxiety disorder, and/or panic disorder. The mean age of the participants was 60.5 years, 90% were white, 61% were employed, 53% were women (JAMA Intern. Med. 2014 April 14 [doi:10.1001/jamainternmend.2014.739]).
They were randomly assigned to receive either usual care or a telephone-based intervention in which a part-time social work care manager coordinated care among psychiatrists, inpatient medical providers, and outpatient medical providers; provided cognitive-behavioral therapy specific to the patient’s condition; and monitored patient symptoms for the 6-month duration of the intervention, said Dr. Jeff C. Huffman, director of cardiac psychiatry research program at Massachusetts General Hospital, and who is in the department of psychiatry at Harvard Medical School, Boston, and his associates.
Patients in the intervention group showed a significantly greater improvement than did those who received usual care in the primary outcome measure: mental-health-related quality of life (QOL), as measured by the Medical Outcomes Study Short Form-12 Mental Component Score. They also were remarkably more likely to receive treatment deemed "adequate" for their psychiatric disorders (75% vs 7%), and to show significantly greater improvement in Patient Health Questionnaire-9 scores; in overall function, as measured on the Duke Activity Status Index; and in health care–related QOL, as measured by EuroQol 5-Domain Instrument scores.
No significant differences were found between the two study groups in cardiac readmission rates at 6 months, but the mean time to readmission was significantly longer for the intervention group (92.4 days) than for the usual care group (62.5 days), Dr. Huffman and his associates said.
The investigators noted that their study involved typical patients seen for cardiac care – including many with serious medical issues and some who declined psychiatric treatment – and so should reflect results that would be found in real-world settings. In addition, using a social worker instead of a nurse as care manager and using telephone rather than in-person contacts substantially saved on costs.
However, Dr. Huffman and his colleagues cited several limitations. For example, the study was conducted in an academic medical center with mostly white patients. Also, those who delivered the intervention had experience with that population and with collaborative care programs.
Still, they expressed optimism about the intervention’s potential. "This intervention seems to have substantial promise as an adjunct or alternative to standard [collaborative care] paradigms," they wrote. "We found that a single care manager was able to coordinate care of three psychiatric conditions in patients with a wide range of cardiac diagnoses living within and outside the metropolitan area of the hospital."
This work was supported in part by the American Heart Association. No relevant financial conflicts of interest were reported.
FROM JAMA INTERNAL MEDICINE
Major finding: Patients in the intervention group showed a significantly greater improvement than did those who received usual care in the primary outcome measure, mental-health–related QOL, as well as being remarkably more likely to receive treatment deemed "adequate" for their psychiatric disorder (75% vs. 7%) and showing significantly greater improvement in overall function and health-related QOL.
Data source: A randomized clinical trial comparing psychiatric outcomes after 6 months of usual care vs. a collaborative care intervention, involving 183 cardiac patients who had concomitant depression or anxiety.
Disclosures: This work was supported in part by the American Heart Association. No relevant financial conflicts of interest were reported.
New valve guideline promotes early surgery
The updated practice guideline for managing adults with valvular heart disease has a new, "modular" format to facilitate clinicians' access to "concise, relevant bytes of information at the point of care, when clinical knowledge is needed most," according to reports published online simultaneously March 3 in Circulation and the Journal of the American College of Cardiology.
The guideline, compiled by a committee of cardiologists, interventionalists, surgeons, and anesthesiologists under the aegis of the American Heart Association and the American College of Cardiology, was last updated in 2008.
"Some recommendations from the earlier valvular heart disease guideline have been updated as warranted by new evidence or a better understanding of earlier evidence, whereas others that were inaccurate, irrelevant, or overlapping were deleted or modified," said writing committee cochairs Dr. Rick A. Nishimura of the division of cardiovascular diseases, Mayo Clinic, Rochester, Minn.; and Dr. Catherine M. Otto, director of the University of Washington Medical Center's Heart Valve Clinic, Seattle.
The narrative text of the guideline is limited, and instead it uses decision pathway diagrams and numerous summary tables of current evidence and recommendations. These include links to relevant references. It is hoped that clinicians can more easily use the new guideline as a quick reference. This format also will enable individual sections to be updated or amended as new evidence comes to light. The PDF of the guideline is available for free.
"This novel approach to evidence-based guideline development will revolutionize the clinical impact of guideline recommendations, ensuring they are always current and allowing seamless integration with electronic medical record systems," Dr. Otto said in a press statement accompanying the reports.
The guideline now includes gradations of disease severity, to help clinicians determine the optimal timing of intervention. Whether or not intervention is indicated depends on five factors: the presence or absence of symptoms, the severity of valvular heart disease, the response of the left and/or right ventricle to the volume or pressure overload caused by the valvular disease, the effect on the pulmonary or systemic circulation, and any change in heart rhythm.
Disease severity ranges from stage A, "at risk," which denotes patients who have risk factors for developing valvular heart disease; through stage B, "progressive," which indicates patients who are asymptomatic but have mildly to moderately severe disease; through stage C, "asymptomatic severe," which includes patients with severe yet still asymptomatic valvular disease in which the left or right ventricle remains compensated or in which the left or right ventricle has decompensated; to stage D, "symptomatic severe," which indicates patients whose severe valvular disease has produced symptoms.
"In patients with stenotic lesions, there is an additional category of 'very severe' stenosis based on studies of the natural history showing that prognosis becomes poorer as the severity of stenosis increases," the guideline states.
Information is provided for assessing the various disease states associated with the aortic, mitral, and tricuspid valves, and addresses the issues of valve repair, replacement, and the use of prosthetic valves.
Compared with the previous guideline, the new one suggests surgical intervention at an earlier stage for certain patients. "Due to more knowledge regarding the natural history of untreated patients with severe valvular heart disease and better outcomes from surgery, we've lowered the threshold for operation to include more patients with asymptomatic severe disease. Now, select patients with severe asymptomatic aortic stenosis and severe asymptomatic mitral regurgitation can be considered for intervention, depending on certain other factors such as operative mortality and … the ability to achieve a durable valve repair," Dr. Nishimura said in the press statement.
The new guideline also proposes a new approach to risk assessment, to be applied to all patients for whom intervention is being considered. Previous risk scoring systems were "useful but limited"; the new approach takes into consideration "procedure-specific impediments, major organ system compromise, comorbidities, patient frailty, and the Society of Thoracic Surgeons predicted risk of mortality model."
For the first time, the guideline discusses transcatheter aortic valve replacement and other catheter-based treatments, new technologies that have improved patient care but also have complicated risk assessment. Separate recommendations are now offered regarding the choice and the timing of these interventions.
In addition to the AHA and the ACC, this guideline was developed in collaboration with the American Association for Thoracic Surgery, American Society for Echocardiography, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons.
The complete 2014 Guideline for the Management of Patients With Valvular Heart Disease is available from the American College of Cardiology and the American Heart Association.
Dr. Nishimura and Dr. Otto reported no financial conflicts of interest; their associates on the ACC/AHA Task Force's writing committee reported ties to Edwards Scientific, Medtronic, and St. Jude Medical.
The updated practice guideline for managing adults with valvular heart disease has a new, "modular" format to facilitate clinicians' access to "concise, relevant bytes of information at the point of care, when clinical knowledge is needed most," according to reports published online simultaneously March 3 in Circulation and the Journal of the American College of Cardiology.
The guideline, compiled by a committee of cardiologists, interventionalists, surgeons, and anesthesiologists under the aegis of the American Heart Association and the American College of Cardiology, was last updated in 2008.
"Some recommendations from the earlier valvular heart disease guideline have been updated as warranted by new evidence or a better understanding of earlier evidence, whereas others that were inaccurate, irrelevant, or overlapping were deleted or modified," said writing committee cochairs Dr. Rick A. Nishimura of the division of cardiovascular diseases, Mayo Clinic, Rochester, Minn.; and Dr. Catherine M. Otto, director of the University of Washington Medical Center's Heart Valve Clinic, Seattle.
The narrative text of the guideline is limited, and instead it uses decision pathway diagrams and numerous summary tables of current evidence and recommendations. These include links to relevant references. It is hoped that clinicians can more easily use the new guideline as a quick reference. This format also will enable individual sections to be updated or amended as new evidence comes to light. The PDF of the guideline is available for free.
"This novel approach to evidence-based guideline development will revolutionize the clinical impact of guideline recommendations, ensuring they are always current and allowing seamless integration with electronic medical record systems," Dr. Otto said in a press statement accompanying the reports.
The guideline now includes gradations of disease severity, to help clinicians determine the optimal timing of intervention. Whether or not intervention is indicated depends on five factors: the presence or absence of symptoms, the severity of valvular heart disease, the response of the left and/or right ventricle to the volume or pressure overload caused by the valvular disease, the effect on the pulmonary or systemic circulation, and any change in heart rhythm.
Disease severity ranges from stage A, "at risk," which denotes patients who have risk factors for developing valvular heart disease; through stage B, "progressive," which indicates patients who are asymptomatic but have mildly to moderately severe disease; through stage C, "asymptomatic severe," which includes patients with severe yet still asymptomatic valvular disease in which the left or right ventricle remains compensated or in which the left or right ventricle has decompensated; to stage D, "symptomatic severe," which indicates patients whose severe valvular disease has produced symptoms.
"In patients with stenotic lesions, there is an additional category of 'very severe' stenosis based on studies of the natural history showing that prognosis becomes poorer as the severity of stenosis increases," the guideline states.
Information is provided for assessing the various disease states associated with the aortic, mitral, and tricuspid valves, and addresses the issues of valve repair, replacement, and the use of prosthetic valves.
Compared with the previous guideline, the new one suggests surgical intervention at an earlier stage for certain patients. "Due to more knowledge regarding the natural history of untreated patients with severe valvular heart disease and better outcomes from surgery, we've lowered the threshold for operation to include more patients with asymptomatic severe disease. Now, select patients with severe asymptomatic aortic stenosis and severe asymptomatic mitral regurgitation can be considered for intervention, depending on certain other factors such as operative mortality and … the ability to achieve a durable valve repair," Dr. Nishimura said in the press statement.
The new guideline also proposes a new approach to risk assessment, to be applied to all patients for whom intervention is being considered. Previous risk scoring systems were "useful but limited"; the new approach takes into consideration "procedure-specific impediments, major organ system compromise, comorbidities, patient frailty, and the Society of Thoracic Surgeons predicted risk of mortality model."
For the first time, the guideline discusses transcatheter aortic valve replacement and other catheter-based treatments, new technologies that have improved patient care but also have complicated risk assessment. Separate recommendations are now offered regarding the choice and the timing of these interventions.
In addition to the AHA and the ACC, this guideline was developed in collaboration with the American Association for Thoracic Surgery, American Society for Echocardiography, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons.
The complete 2014 Guideline for the Management of Patients With Valvular Heart Disease is available from the American College of Cardiology and the American Heart Association.
Dr. Nishimura and Dr. Otto reported no financial conflicts of interest; their associates on the ACC/AHA Task Force's writing committee reported ties to Edwards Scientific, Medtronic, and St. Jude Medical.
The updated practice guideline for managing adults with valvular heart disease has a new, "modular" format to facilitate clinicians' access to "concise, relevant bytes of information at the point of care, when clinical knowledge is needed most," according to reports published online simultaneously March 3 in Circulation and the Journal of the American College of Cardiology.
The guideline, compiled by a committee of cardiologists, interventionalists, surgeons, and anesthesiologists under the aegis of the American Heart Association and the American College of Cardiology, was last updated in 2008.
"Some recommendations from the earlier valvular heart disease guideline have been updated as warranted by new evidence or a better understanding of earlier evidence, whereas others that were inaccurate, irrelevant, or overlapping were deleted or modified," said writing committee cochairs Dr. Rick A. Nishimura of the division of cardiovascular diseases, Mayo Clinic, Rochester, Minn.; and Dr. Catherine M. Otto, director of the University of Washington Medical Center's Heart Valve Clinic, Seattle.
The narrative text of the guideline is limited, and instead it uses decision pathway diagrams and numerous summary tables of current evidence and recommendations. These include links to relevant references. It is hoped that clinicians can more easily use the new guideline as a quick reference. This format also will enable individual sections to be updated or amended as new evidence comes to light. The PDF of the guideline is available for free.
"This novel approach to evidence-based guideline development will revolutionize the clinical impact of guideline recommendations, ensuring they are always current and allowing seamless integration with electronic medical record systems," Dr. Otto said in a press statement accompanying the reports.
The guideline now includes gradations of disease severity, to help clinicians determine the optimal timing of intervention. Whether or not intervention is indicated depends on five factors: the presence or absence of symptoms, the severity of valvular heart disease, the response of the left and/or right ventricle to the volume or pressure overload caused by the valvular disease, the effect on the pulmonary or systemic circulation, and any change in heart rhythm.
Disease severity ranges from stage A, "at risk," which denotes patients who have risk factors for developing valvular heart disease; through stage B, "progressive," which indicates patients who are asymptomatic but have mildly to moderately severe disease; through stage C, "asymptomatic severe," which includes patients with severe yet still asymptomatic valvular disease in which the left or right ventricle remains compensated or in which the left or right ventricle has decompensated; to stage D, "symptomatic severe," which indicates patients whose severe valvular disease has produced symptoms.
"In patients with stenotic lesions, there is an additional category of 'very severe' stenosis based on studies of the natural history showing that prognosis becomes poorer as the severity of stenosis increases," the guideline states.
Information is provided for assessing the various disease states associated with the aortic, mitral, and tricuspid valves, and addresses the issues of valve repair, replacement, and the use of prosthetic valves.
Compared with the previous guideline, the new one suggests surgical intervention at an earlier stage for certain patients. "Due to more knowledge regarding the natural history of untreated patients with severe valvular heart disease and better outcomes from surgery, we've lowered the threshold for operation to include more patients with asymptomatic severe disease. Now, select patients with severe asymptomatic aortic stenosis and severe asymptomatic mitral regurgitation can be considered for intervention, depending on certain other factors such as operative mortality and … the ability to achieve a durable valve repair," Dr. Nishimura said in the press statement.
The new guideline also proposes a new approach to risk assessment, to be applied to all patients for whom intervention is being considered. Previous risk scoring systems were "useful but limited"; the new approach takes into consideration "procedure-specific impediments, major organ system compromise, comorbidities, patient frailty, and the Society of Thoracic Surgeons predicted risk of mortality model."
For the first time, the guideline discusses transcatheter aortic valve replacement and other catheter-based treatments, new technologies that have improved patient care but also have complicated risk assessment. Separate recommendations are now offered regarding the choice and the timing of these interventions.
In addition to the AHA and the ACC, this guideline was developed in collaboration with the American Association for Thoracic Surgery, American Society for Echocardiography, Society for Cardiovascular Angiography and Interventions, Society of Cardiovascular Anesthesiologists, and Society of Thoracic Surgeons.
The complete 2014 Guideline for the Management of Patients With Valvular Heart Disease is available from the American College of Cardiology and the American Heart Association.
Dr. Nishimura and Dr. Otto reported no financial conflicts of interest; their associates on the ACC/AHA Task Force's writing committee reported ties to Edwards Scientific, Medtronic, and St. Jude Medical.
Walking doesn’t hurt, may help in CKD
Neither acute bouts of moderate walking nor regular moderate walking are unsafe in patients who have predialysis chronic kidney disease, and both types of exercise may actually reduce systemic inflammation and improve immune function in these patients, according to a report published in the Journal of the American Society of Nephrology.
CKD is associated with "a complex state of immune dysfunction in which signs of immune depression and immune activation paradoxically coexist." Moderate exercise may therefore enhance certain aspects of immune function while simultaneously exerting detrimental effects on other aspects, said Dr. João Viana of the school of sport, exercise, and health sciences, Loughborough (England) University, and his associates.
The investigators studied blood biomarkers of immune function and inflammation in 15 adults with predialysis CKD who participated in a single bout of "somewhat hard" walking on a motorized treadmill for 30 minutes. This induced "a systemic anti-inflammatory environment," as evidenced by a marked increase in plasma IL-10 levels. It also improved neutrophil responsiveness to an in vitro bacterial challenge during the hour following the exercise. It had no effect on T-lymphocyte or monocyte activation, the researchers said (J. Am. Soc. Nephrol. 2014; April 3; doi:10.1681/ASN.2013070702]).
In a separate analysis, Dr. Viana and his colleagues assessed the same blood biomarkers in 24 participants in a previous study, who had been assigned to either 6 months of regular home-based walking (13 predialysis CKD patients) or to their usual sedentary activity level (11 control subjects). The "exercisers" walked at a "somewhat hard" rate for 30 minutes per day, 5 times per week.
This regular walking also exerted systemic anti-inflammatory effects, as evidenced by a reduction in the IL-6 to IL-10 ratio and downregulation of T-lymphocyte and monocyte activation. It had no effect on circulating numbers of immune cells or on neutrophil degranulation response. Neither acute nor regular walking affected body weight, renal function, proteinuria, or blood pressure.
These findings suggest that walking "is safe from an immune and inflammatory perspective, and exerts anti-inflammatory effects at both systemic and cellular levels" in this patient population. Regular walking "has the potential to be an effective anti-inflammatory therapy in predialysis CKD patients and may in this way reduce the high risk of cardiovascular disease in these very vulnerable patients," the investigators noted.
The authors reported no financial disclosures.
Neither acute bouts of moderate walking nor regular moderate walking are unsafe in patients who have predialysis chronic kidney disease, and both types of exercise may actually reduce systemic inflammation and improve immune function in these patients, according to a report published in the Journal of the American Society of Nephrology.
CKD is associated with "a complex state of immune dysfunction in which signs of immune depression and immune activation paradoxically coexist." Moderate exercise may therefore enhance certain aspects of immune function while simultaneously exerting detrimental effects on other aspects, said Dr. João Viana of the school of sport, exercise, and health sciences, Loughborough (England) University, and his associates.
The investigators studied blood biomarkers of immune function and inflammation in 15 adults with predialysis CKD who participated in a single bout of "somewhat hard" walking on a motorized treadmill for 30 minutes. This induced "a systemic anti-inflammatory environment," as evidenced by a marked increase in plasma IL-10 levels. It also improved neutrophil responsiveness to an in vitro bacterial challenge during the hour following the exercise. It had no effect on T-lymphocyte or monocyte activation, the researchers said (J. Am. Soc. Nephrol. 2014; April 3; doi:10.1681/ASN.2013070702]).
In a separate analysis, Dr. Viana and his colleagues assessed the same blood biomarkers in 24 participants in a previous study, who had been assigned to either 6 months of regular home-based walking (13 predialysis CKD patients) or to their usual sedentary activity level (11 control subjects). The "exercisers" walked at a "somewhat hard" rate for 30 minutes per day, 5 times per week.
This regular walking also exerted systemic anti-inflammatory effects, as evidenced by a reduction in the IL-6 to IL-10 ratio and downregulation of T-lymphocyte and monocyte activation. It had no effect on circulating numbers of immune cells or on neutrophil degranulation response. Neither acute nor regular walking affected body weight, renal function, proteinuria, or blood pressure.
These findings suggest that walking "is safe from an immune and inflammatory perspective, and exerts anti-inflammatory effects at both systemic and cellular levels" in this patient population. Regular walking "has the potential to be an effective anti-inflammatory therapy in predialysis CKD patients and may in this way reduce the high risk of cardiovascular disease in these very vulnerable patients," the investigators noted.
The authors reported no financial disclosures.
Neither acute bouts of moderate walking nor regular moderate walking are unsafe in patients who have predialysis chronic kidney disease, and both types of exercise may actually reduce systemic inflammation and improve immune function in these patients, according to a report published in the Journal of the American Society of Nephrology.
CKD is associated with "a complex state of immune dysfunction in which signs of immune depression and immune activation paradoxically coexist." Moderate exercise may therefore enhance certain aspects of immune function while simultaneously exerting detrimental effects on other aspects, said Dr. João Viana of the school of sport, exercise, and health sciences, Loughborough (England) University, and his associates.
The investigators studied blood biomarkers of immune function and inflammation in 15 adults with predialysis CKD who participated in a single bout of "somewhat hard" walking on a motorized treadmill for 30 minutes. This induced "a systemic anti-inflammatory environment," as evidenced by a marked increase in plasma IL-10 levels. It also improved neutrophil responsiveness to an in vitro bacterial challenge during the hour following the exercise. It had no effect on T-lymphocyte or monocyte activation, the researchers said (J. Am. Soc. Nephrol. 2014; April 3; doi:10.1681/ASN.2013070702]).
In a separate analysis, Dr. Viana and his colleagues assessed the same blood biomarkers in 24 participants in a previous study, who had been assigned to either 6 months of regular home-based walking (13 predialysis CKD patients) or to their usual sedentary activity level (11 control subjects). The "exercisers" walked at a "somewhat hard" rate for 30 minutes per day, 5 times per week.
This regular walking also exerted systemic anti-inflammatory effects, as evidenced by a reduction in the IL-6 to IL-10 ratio and downregulation of T-lymphocyte and monocyte activation. It had no effect on circulating numbers of immune cells or on neutrophil degranulation response. Neither acute nor regular walking affected body weight, renal function, proteinuria, or blood pressure.
These findings suggest that walking "is safe from an immune and inflammatory perspective, and exerts anti-inflammatory effects at both systemic and cellular levels" in this patient population. Regular walking "has the potential to be an effective anti-inflammatory therapy in predialysis CKD patients and may in this way reduce the high risk of cardiovascular disease in these very vulnerable patients," the investigators noted.
The authors reported no financial disclosures.
FROM THE JOURNAL OF THE AMERICAN SOCIETY OF NEPHROLOGY
Major finding: Both a single, "acute" bout of walking and a regular walking program improved several blood markers of systemic inflammation and several markers of immune function in patients with CKD.
Data source: An observational cohort study of 15 adults with predialysis CKD who participated in a single bout of walking exercise, and an analysis of blood biomarkers in 24 participants in a previous study of a 6-month program of regular walking exercise.
Disclosures: The authors reported no financial disclosures.
Diagnosing TOPCAT’s failure in HFpEF
Results of TOPCAT, one of the most talked about heart failure trials in recent years, have been published. There were no obvious surprises: Spironolactone is no more beneficial than placebo for patients who have heart failure with preserved ejection fraction.
Mineralocorticoid-receptor antagonists like spironolactone are known to reduce overall mortality and hospitalizations for HF in patients who have reduced ejection fraction, but their effect on patients with preserved ejection fraction "has not been rigorously tested" until now.
The TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist) study was a phase III, double-blind trial involving 3,445 patients treated at 233 sites in the United States, Canada, Brazil, Argentina, Russia, and Georgia. Patients were randomly assigned to receive either spironolactone (1,722 participants) or placebo (1,723 participants) and followed for a mean of 3.3 years, said Dr. Bertram Pitt of the University of Michigan, Ann Arbor, and his associates in a report published online April 9 in the New England Journal of Medicine.
The primary outcome measure – a composite of death from cardiovascular causes, aborted cardiac arrest, or hospitalization for HF – occurred in 18.6% of patients given spironolactone and 20.4% of those given placebo. This corresponds to incidences of 5.9 events per 100 person-years with active treatment and 6.6 events per 100 person-years with placebo. Both differences were nonsignificant.
There also were no significant differences between the two study groups in time to death from any cause, time to hospitalization for any reason, cause of death, frequency of hospitalization for any reason, and rates of myocardial infarction or stroke, the investigators said (N. Engl. J. Med. 2014 April 9 [doi: 10.1056/NEJMoa1313731]).
In contrast, the 17% reduction in the rate of hospitalization for HFpEF in the spironolactone group relative to controls was significant (P = .04). Moreover, the spironolactone-treated patients had a collective 394 HFpEF hospitalizations, markedly fewer than the 475 in controls. This translated to hospitalization for HFpEF occurring at a rate of 3.8 per 100 person-years in patients randomized to spironolactone, compared with 4.6 per 100 person-years in placebo-treated controls.
The mystifyingly neutral results of this large international trial, presented in November at the annual meeting of the American Heart Association, coincided with a rapidly increasing incidence and awareness of heart failure with preserved ejection fraction, or HFpEF.
Speculation has focused on the differences in treatment response in two areas: geographical location and enrollment criteria.
Geographical differences in event rates have led some to conclude that patients in Eastern Europe just weren’t sick enough to be enrolled. Among patients in the placebo group, the primary endpoint of death or heart failure hospitalization during follow-up occurred at a rate of 12.6 events per 100 patient-years among the 881 patients treated in the four Western Hemisphere countries, and at a rate of 2.3 events per 100 patient-years among the 842 patients treated in Russia or Georgia, a greater than fivefold difference between the two subgroups.
"As a whole, the patients in Russia and Georgia had a lower event rate and were certainly less severely ill than the other patients, but they supposedly still had heart failure," Dr. Scott D. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, Boston, said in an earlier interview. "Defining this disorder is difficult, and when a patient has signs and symptoms of heart failure and preserved ejection fraction, you may not be certain that heart failure is causing the symptoms. That’s why many people think that we should use another criterion" to define HFpEF in trials, such as elevated serum level of some form of natriuretic peptide, said Dr. Solomon, a TOPCAT coinvestigator.
By design, patients could enter TOPCAT either because of a recent heart failure hospitalization, which is how 72% of patients got in, or by having a threshold level of natriuretic peptide, the way the remaining 28% entered the study. Within the subgroup enrolled by natriuretic peptide level, spironolactone treatment had a statistically significant effect in reducing the primary endpoint, while in the other 72% the drug produced no discernable benefit over placebo.
The neutral results of TOPCAT represent more than another failed treatment trial; they underscore the heterogeneity of patients diagnosed with HFpEF.
TOPCAT was supported by the National Heart, Lung, and Blood Institute. Dr. Pitt reported ties to AuraSense Therapeutics, Relypsa, and other companies. His associates reported ties to numerous industry sources. Dr. Solomon has financial ties to Novartis and more than 10 other drug and device companies.
An exploratory post hoc analysis of the data showed that spironolactone was somewhat beneficial at preventing HF hospitalizations among patients in the Americas but not those in Russia and Georgia, said Dr. John J.V. McMurray and Dr. Christopher O’Connor.
This may reflect a true, though slight, treatment effect, or it may simply reflect differences between these geographically distinct regions. Perhaps the clinical characteristics of the patient populations in each area are quite different, or perhaps there are differences in standards of care or in "methodologic expertise in the conduct of clinical trials." Or, as the authors noted, there may be regional heterogeneity in coexisting conditions, in the indications for hospitalization, or in the ability to make the somewhat challenging diagnosis of HF with preserved ejection fraction.
John J. V. McMurray, M.D., is at the British Heart Foundation Cardiovascular Research Centre at the University of Glasgow (Scotland). Christopher O’Connor, M.D., is at Duke University Medical Center, Durham, N.C. Dr. McMurray reported ties to Novartis and Pfizer, and Dr. O’Connor reported ties to Roche, GE Healthcare, and other companies. These remarks were taken from their editorial accompanying Dr. Pitt’s report (N. Engl. J. Med. 2014 April 9 [doi: 10.1056/NEJMe1401231]).
An exploratory post hoc analysis of the data showed that spironolactone was somewhat beneficial at preventing HF hospitalizations among patients in the Americas but not those in Russia and Georgia, said Dr. John J.V. McMurray and Dr. Christopher O’Connor.
This may reflect a true, though slight, treatment effect, or it may simply reflect differences between these geographically distinct regions. Perhaps the clinical characteristics of the patient populations in each area are quite different, or perhaps there are differences in standards of care or in "methodologic expertise in the conduct of clinical trials." Or, as the authors noted, there may be regional heterogeneity in coexisting conditions, in the indications for hospitalization, or in the ability to make the somewhat challenging diagnosis of HF with preserved ejection fraction.
John J. V. McMurray, M.D., is at the British Heart Foundation Cardiovascular Research Centre at the University of Glasgow (Scotland). Christopher O’Connor, M.D., is at Duke University Medical Center, Durham, N.C. Dr. McMurray reported ties to Novartis and Pfizer, and Dr. O’Connor reported ties to Roche, GE Healthcare, and other companies. These remarks were taken from their editorial accompanying Dr. Pitt’s report (N. Engl. J. Med. 2014 April 9 [doi: 10.1056/NEJMe1401231]).
An exploratory post hoc analysis of the data showed that spironolactone was somewhat beneficial at preventing HF hospitalizations among patients in the Americas but not those in Russia and Georgia, said Dr. John J.V. McMurray and Dr. Christopher O’Connor.
This may reflect a true, though slight, treatment effect, or it may simply reflect differences between these geographically distinct regions. Perhaps the clinical characteristics of the patient populations in each area are quite different, or perhaps there are differences in standards of care or in "methodologic expertise in the conduct of clinical trials." Or, as the authors noted, there may be regional heterogeneity in coexisting conditions, in the indications for hospitalization, or in the ability to make the somewhat challenging diagnosis of HF with preserved ejection fraction.
John J. V. McMurray, M.D., is at the British Heart Foundation Cardiovascular Research Centre at the University of Glasgow (Scotland). Christopher O’Connor, M.D., is at Duke University Medical Center, Durham, N.C. Dr. McMurray reported ties to Novartis and Pfizer, and Dr. O’Connor reported ties to Roche, GE Healthcare, and other companies. These remarks were taken from their editorial accompanying Dr. Pitt’s report (N. Engl. J. Med. 2014 April 9 [doi: 10.1056/NEJMe1401231]).
Results of TOPCAT, one of the most talked about heart failure trials in recent years, have been published. There were no obvious surprises: Spironolactone is no more beneficial than placebo for patients who have heart failure with preserved ejection fraction.
Mineralocorticoid-receptor antagonists like spironolactone are known to reduce overall mortality and hospitalizations for HF in patients who have reduced ejection fraction, but their effect on patients with preserved ejection fraction "has not been rigorously tested" until now.
The TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist) study was a phase III, double-blind trial involving 3,445 patients treated at 233 sites in the United States, Canada, Brazil, Argentina, Russia, and Georgia. Patients were randomly assigned to receive either spironolactone (1,722 participants) or placebo (1,723 participants) and followed for a mean of 3.3 years, said Dr. Bertram Pitt of the University of Michigan, Ann Arbor, and his associates in a report published online April 9 in the New England Journal of Medicine.
The primary outcome measure – a composite of death from cardiovascular causes, aborted cardiac arrest, or hospitalization for HF – occurred in 18.6% of patients given spironolactone and 20.4% of those given placebo. This corresponds to incidences of 5.9 events per 100 person-years with active treatment and 6.6 events per 100 person-years with placebo. Both differences were nonsignificant.
There also were no significant differences between the two study groups in time to death from any cause, time to hospitalization for any reason, cause of death, frequency of hospitalization for any reason, and rates of myocardial infarction or stroke, the investigators said (N. Engl. J. Med. 2014 April 9 [doi: 10.1056/NEJMoa1313731]).
In contrast, the 17% reduction in the rate of hospitalization for HFpEF in the spironolactone group relative to controls was significant (P = .04). Moreover, the spironolactone-treated patients had a collective 394 HFpEF hospitalizations, markedly fewer than the 475 in controls. This translated to hospitalization for HFpEF occurring at a rate of 3.8 per 100 person-years in patients randomized to spironolactone, compared with 4.6 per 100 person-years in placebo-treated controls.
The mystifyingly neutral results of this large international trial, presented in November at the annual meeting of the American Heart Association, coincided with a rapidly increasing incidence and awareness of heart failure with preserved ejection fraction, or HFpEF.
Speculation has focused on the differences in treatment response in two areas: geographical location and enrollment criteria.
Geographical differences in event rates have led some to conclude that patients in Eastern Europe just weren’t sick enough to be enrolled. Among patients in the placebo group, the primary endpoint of death or heart failure hospitalization during follow-up occurred at a rate of 12.6 events per 100 patient-years among the 881 patients treated in the four Western Hemisphere countries, and at a rate of 2.3 events per 100 patient-years among the 842 patients treated in Russia or Georgia, a greater than fivefold difference between the two subgroups.
"As a whole, the patients in Russia and Georgia had a lower event rate and were certainly less severely ill than the other patients, but they supposedly still had heart failure," Dr. Scott D. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, Boston, said in an earlier interview. "Defining this disorder is difficult, and when a patient has signs and symptoms of heart failure and preserved ejection fraction, you may not be certain that heart failure is causing the symptoms. That’s why many people think that we should use another criterion" to define HFpEF in trials, such as elevated serum level of some form of natriuretic peptide, said Dr. Solomon, a TOPCAT coinvestigator.
By design, patients could enter TOPCAT either because of a recent heart failure hospitalization, which is how 72% of patients got in, or by having a threshold level of natriuretic peptide, the way the remaining 28% entered the study. Within the subgroup enrolled by natriuretic peptide level, spironolactone treatment had a statistically significant effect in reducing the primary endpoint, while in the other 72% the drug produced no discernable benefit over placebo.
The neutral results of TOPCAT represent more than another failed treatment trial; they underscore the heterogeneity of patients diagnosed with HFpEF.
TOPCAT was supported by the National Heart, Lung, and Blood Institute. Dr. Pitt reported ties to AuraSense Therapeutics, Relypsa, and other companies. His associates reported ties to numerous industry sources. Dr. Solomon has financial ties to Novartis and more than 10 other drug and device companies.
Results of TOPCAT, one of the most talked about heart failure trials in recent years, have been published. There were no obvious surprises: Spironolactone is no more beneficial than placebo for patients who have heart failure with preserved ejection fraction.
Mineralocorticoid-receptor antagonists like spironolactone are known to reduce overall mortality and hospitalizations for HF in patients who have reduced ejection fraction, but their effect on patients with preserved ejection fraction "has not been rigorously tested" until now.
The TOPCAT (Treatment of Preserved Cardiac Function Heart Failure with an Aldosterone Antagonist) study was a phase III, double-blind trial involving 3,445 patients treated at 233 sites in the United States, Canada, Brazil, Argentina, Russia, and Georgia. Patients were randomly assigned to receive either spironolactone (1,722 participants) or placebo (1,723 participants) and followed for a mean of 3.3 years, said Dr. Bertram Pitt of the University of Michigan, Ann Arbor, and his associates in a report published online April 9 in the New England Journal of Medicine.
The primary outcome measure – a composite of death from cardiovascular causes, aborted cardiac arrest, or hospitalization for HF – occurred in 18.6% of patients given spironolactone and 20.4% of those given placebo. This corresponds to incidences of 5.9 events per 100 person-years with active treatment and 6.6 events per 100 person-years with placebo. Both differences were nonsignificant.
There also were no significant differences between the two study groups in time to death from any cause, time to hospitalization for any reason, cause of death, frequency of hospitalization for any reason, and rates of myocardial infarction or stroke, the investigators said (N. Engl. J. Med. 2014 April 9 [doi: 10.1056/NEJMoa1313731]).
In contrast, the 17% reduction in the rate of hospitalization for HFpEF in the spironolactone group relative to controls was significant (P = .04). Moreover, the spironolactone-treated patients had a collective 394 HFpEF hospitalizations, markedly fewer than the 475 in controls. This translated to hospitalization for HFpEF occurring at a rate of 3.8 per 100 person-years in patients randomized to spironolactone, compared with 4.6 per 100 person-years in placebo-treated controls.
The mystifyingly neutral results of this large international trial, presented in November at the annual meeting of the American Heart Association, coincided with a rapidly increasing incidence and awareness of heart failure with preserved ejection fraction, or HFpEF.
Speculation has focused on the differences in treatment response in two areas: geographical location and enrollment criteria.
Geographical differences in event rates have led some to conclude that patients in Eastern Europe just weren’t sick enough to be enrolled. Among patients in the placebo group, the primary endpoint of death or heart failure hospitalization during follow-up occurred at a rate of 12.6 events per 100 patient-years among the 881 patients treated in the four Western Hemisphere countries, and at a rate of 2.3 events per 100 patient-years among the 842 patients treated in Russia or Georgia, a greater than fivefold difference between the two subgroups.
"As a whole, the patients in Russia and Georgia had a lower event rate and were certainly less severely ill than the other patients, but they supposedly still had heart failure," Dr. Scott D. Solomon, professor of medicine at Harvard Medical School and director of noninvasive cardiology at Brigham and Women’s Hospital, Boston, said in an earlier interview. "Defining this disorder is difficult, and when a patient has signs and symptoms of heart failure and preserved ejection fraction, you may not be certain that heart failure is causing the symptoms. That’s why many people think that we should use another criterion" to define HFpEF in trials, such as elevated serum level of some form of natriuretic peptide, said Dr. Solomon, a TOPCAT coinvestigator.
By design, patients could enter TOPCAT either because of a recent heart failure hospitalization, which is how 72% of patients got in, or by having a threshold level of natriuretic peptide, the way the remaining 28% entered the study. Within the subgroup enrolled by natriuretic peptide level, spironolactone treatment had a statistically significant effect in reducing the primary endpoint, while in the other 72% the drug produced no discernable benefit over placebo.
The neutral results of TOPCAT represent more than another failed treatment trial; they underscore the heterogeneity of patients diagnosed with HFpEF.
TOPCAT was supported by the National Heart, Lung, and Blood Institute. Dr. Pitt reported ties to AuraSense Therapeutics, Relypsa, and other companies. His associates reported ties to numerous industry sources. Dr. Solomon has financial ties to Novartis and more than 10 other drug and device companies.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: The primary outcome measure – a composite of death from cardiovascular causes, aborted cardiac arrest, or hospitalization for HF – occurred in 18.6% of patients given spironolactone and 20.4% of those given placebo, a nonsignificant difference.
Data source: TOPCAT, an international randomized, double-blind, phase III trial in 3,445 HFpEF patients treated with either spironolactone or placebo and followed for a mean of 3.3 years.
Disclosures: TOPCAT was supported by the National Heart, Lung, and Blood Institute. Dr. Pitt reported ties to AuraSense Therapeutics, Relypsa, and other companies. His associates reported ties to numerous industry sources. Dr. Solomon has financial ties to Novartis and more than 10 other drug and device companies.
Tenecteplase has risks but cuts hemodynamic decompensation in pulmonary embolism
Fibrinolytic therapy with tenecteplase prevents hemodynamic decompensation in patients with intermediate-risk pulmonary embolism but raises the risk of major hemorrhage and stroke, so its use is likely to remain controversial in this patient population, according to a report published online April 9 in the New England Journal of Medicine.
Fibrinolysis is warranted for high-risk pulmonary embolism characterized by hemodynamic instability, but its risks may outweigh its benefits when normotensive patients have acute right ventricular dysfunction and myocardial injury but no overt hemodynamic compromise. The international PEITHO (Pulmonary Embolism Thrombolysis) trial was performed to examine the safety and efficacy of a single-bolus injection of tenecteplase, in addition to standard anticoagulation therapy, in such patients, who were deemed to be at intermediate risk of an adverse outcome, said Dr. Guy Meyer of Université Paris Descartes and his associates.
PEITHO involved 1,006 adults (median age, 70 years) treated at 76 sites in 13 countries; 506 were randomly assigned to receive a tenecteplase infusion plus unfractionated heparin and 500 to receive a matching placebo infusion plus unfractionated heparin, and all were followed for 30 days. The primary efficacy outcome – a composite of death from any cause or hemodynamic decompensation (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than was the 5.6% rate in the placebo group.
Hemodynamic decompensation occurred in only 1.6% of the tenecteplase group, compared with 5.0% of the placebo group, the investigators wrote (N. Engl. J. Med. 2014 April 9;370:1402-11 [doi:101056/NEJMoa1302097]).
However, major bleeding developed in 11.5% of the tenecteplase group within 7 days, compared with only 2.4% of the placebo group. And the fibrinolytic treatment was associated with a 2.0% risk of hemorrhagic stroke and a 6.3% rate of major extracranial hemorrhage. "Therefore, great caution is warranted when considering fibrinolytic therapy for hemodynamically stable patients with PE," Dr. Meyer and his associates said.
PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
Dr. Frank Podbielski, FCCP, comments: The primary efficacy outcome of PEITHO was all-cause mortality or hemodynamic collapse. Lead investigators Dr. Konstantidnides and Dr. Meyer found a significant difference in these endpoints of 2.6% in the full full-dose tenecteplase group vs. 5.6% in the placebo group.
Hemodynamic collapse, however, did not automatically translate into certain mortality. All-cause mortality between the groups differed by four patients, with over one-half of the patients in the tenecteplase group dying of stroke or hemorrhage. Thrombolytics remain a powerful tool for treatment of pulmonary embolism.
The potential for catastrophic complications should, however, temper enthusiasm for their use except in the direst of situations.
Dr. Frank Podbielski, FCCP, comments: The primary efficacy outcome of PEITHO was all-cause mortality or hemodynamic collapse. Lead investigators Dr. Konstantidnides and Dr. Meyer found a significant difference in these endpoints of 2.6% in the full full-dose tenecteplase group vs. 5.6% in the placebo group.
Hemodynamic collapse, however, did not automatically translate into certain mortality. All-cause mortality between the groups differed by four patients, with over one-half of the patients in the tenecteplase group dying of stroke or hemorrhage. Thrombolytics remain a powerful tool for treatment of pulmonary embolism.
The potential for catastrophic complications should, however, temper enthusiasm for their use except in the direst of situations.
Dr. Frank Podbielski, FCCP, comments: The primary efficacy outcome of PEITHO was all-cause mortality or hemodynamic collapse. Lead investigators Dr. Konstantidnides and Dr. Meyer found a significant difference in these endpoints of 2.6% in the full full-dose tenecteplase group vs. 5.6% in the placebo group.
Hemodynamic collapse, however, did not automatically translate into certain mortality. All-cause mortality between the groups differed by four patients, with over one-half of the patients in the tenecteplase group dying of stroke or hemorrhage. Thrombolytics remain a powerful tool for treatment of pulmonary embolism.
The potential for catastrophic complications should, however, temper enthusiasm for their use except in the direst of situations.
Fibrinolytic therapy with tenecteplase prevents hemodynamic decompensation in patients with intermediate-risk pulmonary embolism but raises the risk of major hemorrhage and stroke, so its use is likely to remain controversial in this patient population, according to a report published online April 9 in the New England Journal of Medicine.
Fibrinolysis is warranted for high-risk pulmonary embolism characterized by hemodynamic instability, but its risks may outweigh its benefits when normotensive patients have acute right ventricular dysfunction and myocardial injury but no overt hemodynamic compromise. The international PEITHO (Pulmonary Embolism Thrombolysis) trial was performed to examine the safety and efficacy of a single-bolus injection of tenecteplase, in addition to standard anticoagulation therapy, in such patients, who were deemed to be at intermediate risk of an adverse outcome, said Dr. Guy Meyer of Université Paris Descartes and his associates.
PEITHO involved 1,006 adults (median age, 70 years) treated at 76 sites in 13 countries; 506 were randomly assigned to receive a tenecteplase infusion plus unfractionated heparin and 500 to receive a matching placebo infusion plus unfractionated heparin, and all were followed for 30 days. The primary efficacy outcome – a composite of death from any cause or hemodynamic decompensation (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than was the 5.6% rate in the placebo group.
Hemodynamic decompensation occurred in only 1.6% of the tenecteplase group, compared with 5.0% of the placebo group, the investigators wrote (N. Engl. J. Med. 2014 April 9;370:1402-11 [doi:101056/NEJMoa1302097]).
However, major bleeding developed in 11.5% of the tenecteplase group within 7 days, compared with only 2.4% of the placebo group. And the fibrinolytic treatment was associated with a 2.0% risk of hemorrhagic stroke and a 6.3% rate of major extracranial hemorrhage. "Therefore, great caution is warranted when considering fibrinolytic therapy for hemodynamically stable patients with PE," Dr. Meyer and his associates said.
PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
Fibrinolytic therapy with tenecteplase prevents hemodynamic decompensation in patients with intermediate-risk pulmonary embolism but raises the risk of major hemorrhage and stroke, so its use is likely to remain controversial in this patient population, according to a report published online April 9 in the New England Journal of Medicine.
Fibrinolysis is warranted for high-risk pulmonary embolism characterized by hemodynamic instability, but its risks may outweigh its benefits when normotensive patients have acute right ventricular dysfunction and myocardial injury but no overt hemodynamic compromise. The international PEITHO (Pulmonary Embolism Thrombolysis) trial was performed to examine the safety and efficacy of a single-bolus injection of tenecteplase, in addition to standard anticoagulation therapy, in such patients, who were deemed to be at intermediate risk of an adverse outcome, said Dr. Guy Meyer of Université Paris Descartes and his associates.
PEITHO involved 1,006 adults (median age, 70 years) treated at 76 sites in 13 countries; 506 were randomly assigned to receive a tenecteplase infusion plus unfractionated heparin and 500 to receive a matching placebo infusion plus unfractionated heparin, and all were followed for 30 days. The primary efficacy outcome – a composite of death from any cause or hemodynamic decompensation (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than was the 5.6% rate in the placebo group.
Hemodynamic decompensation occurred in only 1.6% of the tenecteplase group, compared with 5.0% of the placebo group, the investigators wrote (N. Engl. J. Med. 2014 April 9;370:1402-11 [doi:101056/NEJMoa1302097]).
However, major bleeding developed in 11.5% of the tenecteplase group within 7 days, compared with only 2.4% of the placebo group. And the fibrinolytic treatment was associated with a 2.0% risk of hemorrhagic stroke and a 6.3% rate of major extracranial hemorrhage. "Therefore, great caution is warranted when considering fibrinolytic therapy for hemodynamically stable patients with PE," Dr. Meyer and his associates said.
PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: The primary efficacy outcome – a composite of death from any cause or hemodynamic (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than the 5.6% rate was in the placebo group.
Data source: An international randomized controlled trial involving 1,006 patients with intermediate-risk PE who received either tenecteplase (506 subjects) or a matching placebo infusion (500 subjects) and were followed for 30 days for death, hemodynamic decompensation, bleeding, stroke, recurrent PE, and serious adverse events.
Disclosures: PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
Tenecteplase has risks but cuts hemodynamic decompensation in pulmonary embolism
Fibrinolytic therapy with tenecteplase prevents hemodynamic decompensation in patients with intermediate-risk pulmonary embolism but raises the risk of major hemorrhage and stroke, so its use is likely to remain controversial in this patient population, according to a report published online April 9 in the New England Journal of Medicine.
Fibrinolysis is warranted for high-risk pulmonary embolism characterized by hemodynamic instability, but its risks may outweigh its benefits when normotensive patients have acute right ventricular dysfunction and myocardial injury but no overt hemodynamic compromise. The international PEITHO (Pulmonary Embolism Thrombolysis) trial was performed to examine the safety and efficacy of a single-bolus injection of tenecteplase, in addition to standard anticoagulation therapy, in such patients, who were deemed to be at intermediate risk of an adverse outcome, said Dr. Guy Meyer of Université Paris Descartes and his associates.
PEITHO involved 1,006 adults (median age, 70 years) treated at 76 sites in 13 countries; 506 were randomly assigned to receive a tenecteplase infusion plus unfractionated heparin and 500 to receive a matching placebo infusion plus unfractionated heparin, and all were followed for 30 days. The primary efficacy outcome – a composite of death from any cause or hemodynamic decompensation (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than was the 5.6% rate in the placebo group.
Hemodynamic decompensation occurred in only 1.6% of the tenecteplase group, compared with 5.0% of the placebo group, the investigators wrote (N. Engl. J. Med. 2014 April 9;370:1402-11 [doi:101056/NEJMoa1302097]).
However, major bleeding developed in 11.5% of the tenecteplase group within 7 days, compared with only 2.4% of the placebo group. And the fibrinolytic treatment was associated with a 2.0% risk of hemorrhagic stroke and a 6.3% rate of major extracranial hemorrhage. "Therefore, great caution is warranted when considering fibrinolytic therapy for hemodynamically stable patients with PE," Dr. Meyer and his associates said.
PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
For the practicing clinician, the results of the PEITHO trial "provide valuable insight but no definitive answer" because they confirm the risks as well as the benefits of fibrinolysis for intermediate-risk PE, said Dr. C. Gregory Elliott.
The findings show that it is relatively safe to withhold fibrinolysis unless hemodynamic decompensation occurs. Perhaps a strategy of initiating anticoagulation but reserving fibrinolysis for cases of hemodynamic decompensation would minimize overall risks for this patient population, he said.
Dr. Elliott is in the department of medicine at the Intermountain Medical Center in Murray (Utah) and at the University of Utah, Salt Lake City. He reported ties to Bayer, Bristol-Myers Squibb, Janssen, and Boehringer Ingelheim. These remarks were taken from his editorial accompanying Dr. Meyer’s report (N. Engl. J. Med. 2014 April 9;370:1457-8 [doi:10.1056/NEJMe1401025]).
For the practicing clinician, the results of the PEITHO trial "provide valuable insight but no definitive answer" because they confirm the risks as well as the benefits of fibrinolysis for intermediate-risk PE, said Dr. C. Gregory Elliott.
The findings show that it is relatively safe to withhold fibrinolysis unless hemodynamic decompensation occurs. Perhaps a strategy of initiating anticoagulation but reserving fibrinolysis for cases of hemodynamic decompensation would minimize overall risks for this patient population, he said.
Dr. Elliott is in the department of medicine at the Intermountain Medical Center in Murray (Utah) and at the University of Utah, Salt Lake City. He reported ties to Bayer, Bristol-Myers Squibb, Janssen, and Boehringer Ingelheim. These remarks were taken from his editorial accompanying Dr. Meyer’s report (N. Engl. J. Med. 2014 April 9;370:1457-8 [doi:10.1056/NEJMe1401025]).
For the practicing clinician, the results of the PEITHO trial "provide valuable insight but no definitive answer" because they confirm the risks as well as the benefits of fibrinolysis for intermediate-risk PE, said Dr. C. Gregory Elliott.
The findings show that it is relatively safe to withhold fibrinolysis unless hemodynamic decompensation occurs. Perhaps a strategy of initiating anticoagulation but reserving fibrinolysis for cases of hemodynamic decompensation would minimize overall risks for this patient population, he said.
Dr. Elliott is in the department of medicine at the Intermountain Medical Center in Murray (Utah) and at the University of Utah, Salt Lake City. He reported ties to Bayer, Bristol-Myers Squibb, Janssen, and Boehringer Ingelheim. These remarks were taken from his editorial accompanying Dr. Meyer’s report (N. Engl. J. Med. 2014 April 9;370:1457-8 [doi:10.1056/NEJMe1401025]).
Fibrinolytic therapy with tenecteplase prevents hemodynamic decompensation in patients with intermediate-risk pulmonary embolism but raises the risk of major hemorrhage and stroke, so its use is likely to remain controversial in this patient population, according to a report published online April 9 in the New England Journal of Medicine.
Fibrinolysis is warranted for high-risk pulmonary embolism characterized by hemodynamic instability, but its risks may outweigh its benefits when normotensive patients have acute right ventricular dysfunction and myocardial injury but no overt hemodynamic compromise. The international PEITHO (Pulmonary Embolism Thrombolysis) trial was performed to examine the safety and efficacy of a single-bolus injection of tenecteplase, in addition to standard anticoagulation therapy, in such patients, who were deemed to be at intermediate risk of an adverse outcome, said Dr. Guy Meyer of Université Paris Descartes and his associates.
PEITHO involved 1,006 adults (median age, 70 years) treated at 76 sites in 13 countries; 506 were randomly assigned to receive a tenecteplase infusion plus unfractionated heparin and 500 to receive a matching placebo infusion plus unfractionated heparin, and all were followed for 30 days. The primary efficacy outcome – a composite of death from any cause or hemodynamic decompensation (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than was the 5.6% rate in the placebo group.
Hemodynamic decompensation occurred in only 1.6% of the tenecteplase group, compared with 5.0% of the placebo group, the investigators wrote (N. Engl. J. Med. 2014 April 9;370:1402-11 [doi:101056/NEJMoa1302097]).
However, major bleeding developed in 11.5% of the tenecteplase group within 7 days, compared with only 2.4% of the placebo group. And the fibrinolytic treatment was associated with a 2.0% risk of hemorrhagic stroke and a 6.3% rate of major extracranial hemorrhage. "Therefore, great caution is warranted when considering fibrinolytic therapy for hemodynamically stable patients with PE," Dr. Meyer and his associates said.
PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
Fibrinolytic therapy with tenecteplase prevents hemodynamic decompensation in patients with intermediate-risk pulmonary embolism but raises the risk of major hemorrhage and stroke, so its use is likely to remain controversial in this patient population, according to a report published online April 9 in the New England Journal of Medicine.
Fibrinolysis is warranted for high-risk pulmonary embolism characterized by hemodynamic instability, but its risks may outweigh its benefits when normotensive patients have acute right ventricular dysfunction and myocardial injury but no overt hemodynamic compromise. The international PEITHO (Pulmonary Embolism Thrombolysis) trial was performed to examine the safety and efficacy of a single-bolus injection of tenecteplase, in addition to standard anticoagulation therapy, in such patients, who were deemed to be at intermediate risk of an adverse outcome, said Dr. Guy Meyer of Université Paris Descartes and his associates.
PEITHO involved 1,006 adults (median age, 70 years) treated at 76 sites in 13 countries; 506 were randomly assigned to receive a tenecteplase infusion plus unfractionated heparin and 500 to receive a matching placebo infusion plus unfractionated heparin, and all were followed for 30 days. The primary efficacy outcome – a composite of death from any cause or hemodynamic decompensation (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than was the 5.6% rate in the placebo group.
Hemodynamic decompensation occurred in only 1.6% of the tenecteplase group, compared with 5.0% of the placebo group, the investigators wrote (N. Engl. J. Med. 2014 April 9;370:1402-11 [doi:101056/NEJMoa1302097]).
However, major bleeding developed in 11.5% of the tenecteplase group within 7 days, compared with only 2.4% of the placebo group. And the fibrinolytic treatment was associated with a 2.0% risk of hemorrhagic stroke and a 6.3% rate of major extracranial hemorrhage. "Therefore, great caution is warranted when considering fibrinolytic therapy for hemodynamically stable patients with PE," Dr. Meyer and his associates said.
PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: The primary efficacy outcome – a composite of death from any cause or hemodynamic (including hemodynamic collapse) within 7 days – occurred in only 2.6% of the tenecteplase group, which was significantly lower than the 5.6% rate was in the placebo group.
Data source: An international randomized controlled trial involving 1,006 patients with intermediate-risk PE who received either tenecteplase (506 subjects) or a matching placebo infusion (500 subjects) and were followed for 30 days for death, hemodynamic decompensation, bleeding, stroke, recurrent PE, and serious adverse events.
Disclosures: PEITHO was supported by the Programme Hospitalier de Recherche Clinique in France, the Federal Ministry of Education and Research in Germany, and Boehringer Ingelheim. Dr. Meyer reported ties to Boehringer Ingelheim, Leo Pharma, Bayer Healthcare, and Sanofi-Aventis, and his associates reported ties to numerous industry sources.
Conventional chemo bests tyrosine kinase inhibitors in wild-type lung cancers
Conventional chemotherapy prolonged progression-free survival and induced a higher tumor response rate, compared with tyrosine kinase inhibitors, in a meta-analysis involving 1,605 patients with advanced wild-type non–small cell lung cancer, according to a report published online April 8 in JAMA.
First-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors such as erlotinib and gefitinib are first-line treatments for non–small cell lung cancers that are known to harbor EGFR-activating mutations, but their benefit is less pronounced against wild-type NSCLCs (no mutation detected in the EGFR gene). Several small studies assessing the efficacy of these agents in wild-type NSCLCs have produced inconclusive results, mostly because of the small number of participating patients, said Dr. June-Koo Lee of the department of internal medicine, Seoul (Republic of Korea) National University Hospital, and associates.
The investigators therefore pooled data from a meta-analysis of 11 randomized, controlled, open-label trials, to obtain sufficient data to compare EGFR tyrosine kinase inhibitors (811 patients) against conventional chemotherapeutic agents such as cisplatin, carboplatin, docetaxel, and pemetrexed (794 patients) in wild-type NSCLCs.
Progression-free survival was significantly longer with conventional chemotherapy than with EGFR tyrosine kinase inhibitors (6.4 vs 1.9 months; hazard ratio, 1.41; 95% confidence interval, 1.10-1.81). Conventional chemotherapy also induced a significantly higher objective response rate than did tyrosine kinase inhibitors (16.8% vs 7.2%; relative risk of nonresponse from tyrosine kinase inhibitors, 1.11; 95% CI, 1.02-1.21).
Overall survival was not significantly different between the two study groups, but that "can be explained by the large crossover rates of the included trials," Dr. Lee and associates reported (JAMA 2014;311:1430-7).
The findings "suggest that current guidelines recommending EGFR tyrosine kinase inhibitors as a standard treatment in this setting ... may need to be reevaluated," they wrote.
However, it is important to note that a treatment’s toxicity profile is crucial when choosing among different options, and EGFR tyrosine kinase inhibitors are known to have a better toxicity profile than standard chemotherapeutic agents. They should therefore be considered for certain patients, such as those who have a poor performance status, the investigators said.
This study was supported in part by the National Research Foundation of Korea. Dr. Lee reported no financial conflicts of interest; two coauthors reported ties to Pfizer, Lilly, and other companies.
Conventional chemotherapy prolonged progression-free survival and induced a higher tumor response rate, compared with tyrosine kinase inhibitors, in a meta-analysis involving 1,605 patients with advanced wild-type non–small cell lung cancer, according to a report published online April 8 in JAMA.
First-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors such as erlotinib and gefitinib are first-line treatments for non–small cell lung cancers that are known to harbor EGFR-activating mutations, but their benefit is less pronounced against wild-type NSCLCs (no mutation detected in the EGFR gene). Several small studies assessing the efficacy of these agents in wild-type NSCLCs have produced inconclusive results, mostly because of the small number of participating patients, said Dr. June-Koo Lee of the department of internal medicine, Seoul (Republic of Korea) National University Hospital, and associates.
The investigators therefore pooled data from a meta-analysis of 11 randomized, controlled, open-label trials, to obtain sufficient data to compare EGFR tyrosine kinase inhibitors (811 patients) against conventional chemotherapeutic agents such as cisplatin, carboplatin, docetaxel, and pemetrexed (794 patients) in wild-type NSCLCs.
Progression-free survival was significantly longer with conventional chemotherapy than with EGFR tyrosine kinase inhibitors (6.4 vs 1.9 months; hazard ratio, 1.41; 95% confidence interval, 1.10-1.81). Conventional chemotherapy also induced a significantly higher objective response rate than did tyrosine kinase inhibitors (16.8% vs 7.2%; relative risk of nonresponse from tyrosine kinase inhibitors, 1.11; 95% CI, 1.02-1.21).
Overall survival was not significantly different between the two study groups, but that "can be explained by the large crossover rates of the included trials," Dr. Lee and associates reported (JAMA 2014;311:1430-7).
The findings "suggest that current guidelines recommending EGFR tyrosine kinase inhibitors as a standard treatment in this setting ... may need to be reevaluated," they wrote.
However, it is important to note that a treatment’s toxicity profile is crucial when choosing among different options, and EGFR tyrosine kinase inhibitors are known to have a better toxicity profile than standard chemotherapeutic agents. They should therefore be considered for certain patients, such as those who have a poor performance status, the investigators said.
This study was supported in part by the National Research Foundation of Korea. Dr. Lee reported no financial conflicts of interest; two coauthors reported ties to Pfizer, Lilly, and other companies.
Conventional chemotherapy prolonged progression-free survival and induced a higher tumor response rate, compared with tyrosine kinase inhibitors, in a meta-analysis involving 1,605 patients with advanced wild-type non–small cell lung cancer, according to a report published online April 8 in JAMA.
First-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors such as erlotinib and gefitinib are first-line treatments for non–small cell lung cancers that are known to harbor EGFR-activating mutations, but their benefit is less pronounced against wild-type NSCLCs (no mutation detected in the EGFR gene). Several small studies assessing the efficacy of these agents in wild-type NSCLCs have produced inconclusive results, mostly because of the small number of participating patients, said Dr. June-Koo Lee of the department of internal medicine, Seoul (Republic of Korea) National University Hospital, and associates.
The investigators therefore pooled data from a meta-analysis of 11 randomized, controlled, open-label trials, to obtain sufficient data to compare EGFR tyrosine kinase inhibitors (811 patients) against conventional chemotherapeutic agents such as cisplatin, carboplatin, docetaxel, and pemetrexed (794 patients) in wild-type NSCLCs.
Progression-free survival was significantly longer with conventional chemotherapy than with EGFR tyrosine kinase inhibitors (6.4 vs 1.9 months; hazard ratio, 1.41; 95% confidence interval, 1.10-1.81). Conventional chemotherapy also induced a significantly higher objective response rate than did tyrosine kinase inhibitors (16.8% vs 7.2%; relative risk of nonresponse from tyrosine kinase inhibitors, 1.11; 95% CI, 1.02-1.21).
Overall survival was not significantly different between the two study groups, but that "can be explained by the large crossover rates of the included trials," Dr. Lee and associates reported (JAMA 2014;311:1430-7).
The findings "suggest that current guidelines recommending EGFR tyrosine kinase inhibitors as a standard treatment in this setting ... may need to be reevaluated," they wrote.
However, it is important to note that a treatment’s toxicity profile is crucial when choosing among different options, and EGFR tyrosine kinase inhibitors are known to have a better toxicity profile than standard chemotherapeutic agents. They should therefore be considered for certain patients, such as those who have a poor performance status, the investigators said.
This study was supported in part by the National Research Foundation of Korea. Dr. Lee reported no financial conflicts of interest; two coauthors reported ties to Pfizer, Lilly, and other companies.
FROM JAMA
Key clinical point:
Major finding: Progression-free survival was significantly longer with conventional chemotherapy than with EGFR tyrosine kinase inhibitors (6.4 vs. 1.9 months), and the objective response rate was significantly higher (16.8% vs. 7.2%).
Data source: A review of the literature and meta-analysis of 11 prospective, open-label, randomized controlled trials involving 1,605 patients with wild-type NSCLC who were treated with either first-generation EGFR tyrosine kinase inhibitors (erlotinib or gefitinib) or conventional chemotherapy (cisplatin, carboplatin, docetaxel, or pemetrexed).
Disclosures: This study was supported in part by the National Research Foundation of Korea. Dr. Lee reported no financial conflicts of interest; two coauthors reported ties to Pfizer, Lilly, and other companies.
Three key subdimensions of mania identified, validated
Mania, the defining feature of bipolar disorder, is not the monolithic entity it is portrayed to be in the DSM-5, but instead has at least three distinct "subdimensions" that distinguish it from other mental disorders, according to a report in the Journal of Affective Disorders.
Researchers who found the DSM-5 portrayal of mania to be "unidimensional" and "misleading" sought to establish a more nuanced characterization by using detailed clinical interviews of 422 psychiatric outpatients to identify the key features unique to mania. These patients, predominantly women with a mean age of 42 years, were currently receiving mental health treatment; 17% had a diagnosis of bipolar disorder and 6% were in a manic episode at the time of the interview, said Camilo J. Ruggero, Ph.D., of the department of psychology, University of North Texas, Denton, and his associates.
Using the Interview for Mood and Anxiety Symptoms; the Structured Clinical Interview for DSM-IV Axis I Disorders; the Inventory of Depression and Anxiety Symptoms, Expanded Version; the Global Assessment of Functioning; and the Sheehan Disability Scale, the investigators identified three subdimensions that consistently characterized mania and distinguished it from other disorders: euphoric activation, which reflected increased energy; hyperactive cognition, which reflected flight of ideas, pressured speech, and distractibility; and reckless overconfidence, which reflected extreme overconfidence and resulting poor judgment and recklessness. The latter subdimension is similar to grandiosity but not necessarily delusional. A fourth subdimension, irritability, also was characteristic of mania but was not as unique to it as were the other three, since it can also occur in depression and other disorders (J. Affect. Disord. 2014;161:8-15).
After creating an instrument to reflect these four subdimensions of mania, Dr. Ruggero and his associates validated their findings in clinical interviews with a separate study population: 306 student volunteers who had a history of mental health treatment, 31% of whom were currently in treatment. All four subdimensions were highly prevalent in patients who had mania but not in those with other disorders.
Identifying these key subdimensions could elucidate the etiology of bipolar disorder. And, instead of focusing on "the higher-order syndrome" of mania, highlighting these subdimensions might help resolve discrepancies in results from pathophysiologic studies, since different facets of mania might reflect different pathophysiological processes, the researchers noted.
"Moreover, assessing distinct types of symptoms has important clinical implications; not all subdimensions affect functioning equally." For example, in these two patient populations, euphoric activation had little impact on participants’ daily functioning, compared with other symptoms, the researchers added.
Dr. Ruggero and his associates cited several limitations. Neither psychotic nor depressive symptoms were included in their analyses. Also, a few of the patients were in a current manic episode at the time of the study. Despite those limitations, their study "provides important new findings about mania. Future work can use these dimensions ... to better discern the pathophysiology of bipolar disorder," they wrote.
This work was supported in part by the Feldstein Medical Foundation. Dr. Ruggero and his associates reported no conflicts of interest.
Mania, the defining feature of bipolar disorder, is not the monolithic entity it is portrayed to be in the DSM-5, but instead has at least three distinct "subdimensions" that distinguish it from other mental disorders, according to a report in the Journal of Affective Disorders.
Researchers who found the DSM-5 portrayal of mania to be "unidimensional" and "misleading" sought to establish a more nuanced characterization by using detailed clinical interviews of 422 psychiatric outpatients to identify the key features unique to mania. These patients, predominantly women with a mean age of 42 years, were currently receiving mental health treatment; 17% had a diagnosis of bipolar disorder and 6% were in a manic episode at the time of the interview, said Camilo J. Ruggero, Ph.D., of the department of psychology, University of North Texas, Denton, and his associates.
Using the Interview for Mood and Anxiety Symptoms; the Structured Clinical Interview for DSM-IV Axis I Disorders; the Inventory of Depression and Anxiety Symptoms, Expanded Version; the Global Assessment of Functioning; and the Sheehan Disability Scale, the investigators identified three subdimensions that consistently characterized mania and distinguished it from other disorders: euphoric activation, which reflected increased energy; hyperactive cognition, which reflected flight of ideas, pressured speech, and distractibility; and reckless overconfidence, which reflected extreme overconfidence and resulting poor judgment and recklessness. The latter subdimension is similar to grandiosity but not necessarily delusional. A fourth subdimension, irritability, also was characteristic of mania but was not as unique to it as were the other three, since it can also occur in depression and other disorders (J. Affect. Disord. 2014;161:8-15).
After creating an instrument to reflect these four subdimensions of mania, Dr. Ruggero and his associates validated their findings in clinical interviews with a separate study population: 306 student volunteers who had a history of mental health treatment, 31% of whom were currently in treatment. All four subdimensions were highly prevalent in patients who had mania but not in those with other disorders.
Identifying these key subdimensions could elucidate the etiology of bipolar disorder. And, instead of focusing on "the higher-order syndrome" of mania, highlighting these subdimensions might help resolve discrepancies in results from pathophysiologic studies, since different facets of mania might reflect different pathophysiological processes, the researchers noted.
"Moreover, assessing distinct types of symptoms has important clinical implications; not all subdimensions affect functioning equally." For example, in these two patient populations, euphoric activation had little impact on participants’ daily functioning, compared with other symptoms, the researchers added.
Dr. Ruggero and his associates cited several limitations. Neither psychotic nor depressive symptoms were included in their analyses. Also, a few of the patients were in a current manic episode at the time of the study. Despite those limitations, their study "provides important new findings about mania. Future work can use these dimensions ... to better discern the pathophysiology of bipolar disorder," they wrote.
This work was supported in part by the Feldstein Medical Foundation. Dr. Ruggero and his associates reported no conflicts of interest.
Mania, the defining feature of bipolar disorder, is not the monolithic entity it is portrayed to be in the DSM-5, but instead has at least three distinct "subdimensions" that distinguish it from other mental disorders, according to a report in the Journal of Affective Disorders.
Researchers who found the DSM-5 portrayal of mania to be "unidimensional" and "misleading" sought to establish a more nuanced characterization by using detailed clinical interviews of 422 psychiatric outpatients to identify the key features unique to mania. These patients, predominantly women with a mean age of 42 years, were currently receiving mental health treatment; 17% had a diagnosis of bipolar disorder and 6% were in a manic episode at the time of the interview, said Camilo J. Ruggero, Ph.D., of the department of psychology, University of North Texas, Denton, and his associates.
Using the Interview for Mood and Anxiety Symptoms; the Structured Clinical Interview for DSM-IV Axis I Disorders; the Inventory of Depression and Anxiety Symptoms, Expanded Version; the Global Assessment of Functioning; and the Sheehan Disability Scale, the investigators identified three subdimensions that consistently characterized mania and distinguished it from other disorders: euphoric activation, which reflected increased energy; hyperactive cognition, which reflected flight of ideas, pressured speech, and distractibility; and reckless overconfidence, which reflected extreme overconfidence and resulting poor judgment and recklessness. The latter subdimension is similar to grandiosity but not necessarily delusional. A fourth subdimension, irritability, also was characteristic of mania but was not as unique to it as were the other three, since it can also occur in depression and other disorders (J. Affect. Disord. 2014;161:8-15).
After creating an instrument to reflect these four subdimensions of mania, Dr. Ruggero and his associates validated their findings in clinical interviews with a separate study population: 306 student volunteers who had a history of mental health treatment, 31% of whom were currently in treatment. All four subdimensions were highly prevalent in patients who had mania but not in those with other disorders.
Identifying these key subdimensions could elucidate the etiology of bipolar disorder. And, instead of focusing on "the higher-order syndrome" of mania, highlighting these subdimensions might help resolve discrepancies in results from pathophysiologic studies, since different facets of mania might reflect different pathophysiological processes, the researchers noted.
"Moreover, assessing distinct types of symptoms has important clinical implications; not all subdimensions affect functioning equally." For example, in these two patient populations, euphoric activation had little impact on participants’ daily functioning, compared with other symptoms, the researchers added.
Dr. Ruggero and his associates cited several limitations. Neither psychotic nor depressive symptoms were included in their analyses. Also, a few of the patients were in a current manic episode at the time of the study. Despite those limitations, their study "provides important new findings about mania. Future work can use these dimensions ... to better discern the pathophysiology of bipolar disorder," they wrote.
This work was supported in part by the Feldstein Medical Foundation. Dr. Ruggero and his associates reported no conflicts of interest.
FROM THE JOURNAL OF AFFECTIVE DISORDERS
Major finding: Three subdimensions of mania consistently characterize the disorder and distinguish it from other mental disorders: euphoric activation, which reflects increased energy; hyperactive cognition, which reflects flight of ideas, pressured speech, and distractibility; and reckless overconfidence, which reflects extreme overconfidence and the resulting poor judgment and recklessness. The latter is similar to grandiosity but is not necessarily delusional.
Data source: Clinical interviews with 422 psychiatric outpatients to identify subdimensions that are specific to mania, and separate interviews with 306 students with a history of mental health treatment to validate that four subdimensions characterize mania.
Disclosures: This work was supported in part by the Feldstein Medical Foundation. Dr. Ruggero and his associates reported no conflicts of interest.