User login
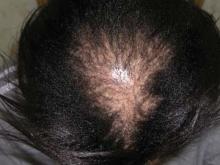
Scarring Alopecia in Black Women Still Not Understood
Despite being the most common pattern of scarring hair loss among African American women, central centrifugal cicatricial alopecia is still poorly understood, according to a report published online April 11 in Archives of Dermatology.
The etiology of and risk factors for the disorder have not been established. And although African American women commonly present with the condition, the exact prevalence remains unknown, said Dr. Angela Kyei and her associates at the Cleveland Clinic Institute of Dermatology and Plastic Surgery.
Central centrifugal cicatricial alopecia (CCCA) – previously known as hot comb alopecia or follicular degeneration syndrome – is a scarring hair loss centered on the vertex of the scalp and spreading peripherally, which is described almost exclusively in African American women. In the late 1960s it was linked to the use of hot combs, but since then almost every method of hair grooming in this population has been associated, albeit weakly, with the disorder. Few studies have examined other possible etiologies, such as immunologic, dermatologic, or genetic factors.
"Given the lack of epidemiologic data, the main goal of [our] study was to elucidate environmental as well as medical risk factors that may be associated with CCCA," the researchers wrote.
They administered questionnaires to 326 African American women attending two churches and a health fair in the Cleveland area. Sixteen of the women ended up being excluded from the analysis.
The questionnaires asked detailed information about family history of male- and female-pattern hair loss; personal medical history, including bacterial and fungal skin infections, autoimmune conditions, and hormonal dysregulation; and hair grooming methods used. The study subjects also underwent a scalp examination that included the grading of hair loss.
A total of 86 of the 310 women (28%) were judged to have central hair loss. Of these, 52 women – 17% of the total study population – had CCCA.
It appeared that hair grooming practices that cause traction, such as weaves and braids, may contribute to the development of CCCA, as women with the most severe central hair loss used these styles more frequently than did those without hair loss. "This has some clinical bearing because traction can clinically produce folliculitis of the scalp, which can cause scarring if this inflammation is prolonged," noted Dr. Kyei and her colleagues (Arch. Dermatol. 2011 April 11 [doi:10.1001/archdermatol.2011.66]).
After paying hundreds of dollars for such hair styling, women often maintain weaves and braids for weeks to months, so any reactive inflammation usually is prolonged, the investigators added.
However, "the relationship between chemical relaxer use and the development of CCCA continues to be murky," they noted. These products clearly weaken the hair shaft, which can cause breakage, but that does not necessarily lead to CCCA. "The fact that most African Americans use chemical relaxers in combination with braiding and other hair grooming practices makes it even more difficult to tease out a relationship," Dr. Kyei and her associates added.
It is difficult to find subjects who do not use chemical relaxers for comparison, since most African Americans begin the practice in childhood. In this study, 91% of the women reported using chemical relaxers, and began doing so at an average age of 10 years.
Nevertheless, "we feel that it is not unreasonable to assume that the scalp may absorb some of the caustic chemicals found in relaxers, which in time leads to damage of the scalp in the form of scarring," they wrote.
Although the prevalence of bacterial skin infections in these study subjects was only 11%, there was a significant elevation among women with CCCA, compared with women who did not have CCCA. The rate of acne also was elevated in affected subjects, but not to a significant degree. Thus, the researchers said that their study "demonstrates that inflammation in the form of bacterial infection and acne may also be contributing to the development of CCCA, a finding consistent with the histopathologic characteristics of this disease," which show a lymphocytic perifollicular infiltrate in its early stages.
In contrast, there was no such association with fungal infections of the scalp, ringworm, or vaginal yeast infections, which was "surprising given how prone this population is to fungal infection," they added.
"One of the most surprising findings ... was the overrepresentation of type 2 [diabetes mellitus] in those with CCCA," the authors noted. Again, the prevalence of type 2 diabetes was low, at 8%, but it was significantly elevated in women with CCCA, compared with those without CCCA.
"These are important data that need further study because CCCA may be a marker of metabolic dysfunction and, when present, can prompt clinicians to do further testing for diabetes mellitus," Dr. Kyei and her colleagues wrote.
Only 9% of the study subjects had thyroid abnormalities, and three-fourths of them had no or minimal hair loss.
A history of male-pattern baldness in the maternal grandfather was found to be a risk factor for CCCA, which suggests that genetics may play a role.
Hormonal dysregulation did not appear to be associated with development of CCCA. Neither were scarring disorders, since only 6% of the subjects reported a history of keloids, and the rate was no higher in women with CCCA than in those without CCCA.
Similarly, there were relatively high prevalences of seborrheic dermatitis (24%), eczema (13%), and contact dermatitis from chemical relaxers (9%), as would be expected in an African American population. However, these conditions were unrelated to CCCA.
This study was supported in part by the North American Hair Research Society and Procter & Gamble. No financial conflicts of interest were reported.
Despite being the most common pattern of scarring hair loss among African American women, central centrifugal cicatricial alopecia is still poorly understood, according to a report published online April 11 in Archives of Dermatology.
The etiology of and risk factors for the disorder have not been established. And although African American women commonly present with the condition, the exact prevalence remains unknown, said Dr. Angela Kyei and her associates at the Cleveland Clinic Institute of Dermatology and Plastic Surgery.
Central centrifugal cicatricial alopecia (CCCA) – previously known as hot comb alopecia or follicular degeneration syndrome – is a scarring hair loss centered on the vertex of the scalp and spreading peripherally, which is described almost exclusively in African American women. In the late 1960s it was linked to the use of hot combs, but since then almost every method of hair grooming in this population has been associated, albeit weakly, with the disorder. Few studies have examined other possible etiologies, such as immunologic, dermatologic, or genetic factors.
"Given the lack of epidemiologic data, the main goal of [our] study was to elucidate environmental as well as medical risk factors that may be associated with CCCA," the researchers wrote.
They administered questionnaires to 326 African American women attending two churches and a health fair in the Cleveland area. Sixteen of the women ended up being excluded from the analysis.
The questionnaires asked detailed information about family history of male- and female-pattern hair loss; personal medical history, including bacterial and fungal skin infections, autoimmune conditions, and hormonal dysregulation; and hair grooming methods used. The study subjects also underwent a scalp examination that included the grading of hair loss.
A total of 86 of the 310 women (28%) were judged to have central hair loss. Of these, 52 women – 17% of the total study population – had CCCA.
It appeared that hair grooming practices that cause traction, such as weaves and braids, may contribute to the development of CCCA, as women with the most severe central hair loss used these styles more frequently than did those without hair loss. "This has some clinical bearing because traction can clinically produce folliculitis of the scalp, which can cause scarring if this inflammation is prolonged," noted Dr. Kyei and her colleagues (Arch. Dermatol. 2011 April 11 [doi:10.1001/archdermatol.2011.66]).
After paying hundreds of dollars for such hair styling, women often maintain weaves and braids for weeks to months, so any reactive inflammation usually is prolonged, the investigators added.
However, "the relationship between chemical relaxer use and the development of CCCA continues to be murky," they noted. These products clearly weaken the hair shaft, which can cause breakage, but that does not necessarily lead to CCCA. "The fact that most African Americans use chemical relaxers in combination with braiding and other hair grooming practices makes it even more difficult to tease out a relationship," Dr. Kyei and her associates added.
It is difficult to find subjects who do not use chemical relaxers for comparison, since most African Americans begin the practice in childhood. In this study, 91% of the women reported using chemical relaxers, and began doing so at an average age of 10 years.
Nevertheless, "we feel that it is not unreasonable to assume that the scalp may absorb some of the caustic chemicals found in relaxers, which in time leads to damage of the scalp in the form of scarring," they wrote.
Although the prevalence of bacterial skin infections in these study subjects was only 11%, there was a significant elevation among women with CCCA, compared with women who did not have CCCA. The rate of acne also was elevated in affected subjects, but not to a significant degree. Thus, the researchers said that their study "demonstrates that inflammation in the form of bacterial infection and acne may also be contributing to the development of CCCA, a finding consistent with the histopathologic characteristics of this disease," which show a lymphocytic perifollicular infiltrate in its early stages.
In contrast, there was no such association with fungal infections of the scalp, ringworm, or vaginal yeast infections, which was "surprising given how prone this population is to fungal infection," they added.
"One of the most surprising findings ... was the overrepresentation of type 2 [diabetes mellitus] in those with CCCA," the authors noted. Again, the prevalence of type 2 diabetes was low, at 8%, but it was significantly elevated in women with CCCA, compared with those without CCCA.
"These are important data that need further study because CCCA may be a marker of metabolic dysfunction and, when present, can prompt clinicians to do further testing for diabetes mellitus," Dr. Kyei and her colleagues wrote.
Only 9% of the study subjects had thyroid abnormalities, and three-fourths of them had no or minimal hair loss.
A history of male-pattern baldness in the maternal grandfather was found to be a risk factor for CCCA, which suggests that genetics may play a role.
Hormonal dysregulation did not appear to be associated with development of CCCA. Neither were scarring disorders, since only 6% of the subjects reported a history of keloids, and the rate was no higher in women with CCCA than in those without CCCA.
Similarly, there were relatively high prevalences of seborrheic dermatitis (24%), eczema (13%), and contact dermatitis from chemical relaxers (9%), as would be expected in an African American population. However, these conditions were unrelated to CCCA.
This study was supported in part by the North American Hair Research Society and Procter & Gamble. No financial conflicts of interest were reported.
Despite being the most common pattern of scarring hair loss among African American women, central centrifugal cicatricial alopecia is still poorly understood, according to a report published online April 11 in Archives of Dermatology.
The etiology of and risk factors for the disorder have not been established. And although African American women commonly present with the condition, the exact prevalence remains unknown, said Dr. Angela Kyei and her associates at the Cleveland Clinic Institute of Dermatology and Plastic Surgery.
Central centrifugal cicatricial alopecia (CCCA) – previously known as hot comb alopecia or follicular degeneration syndrome – is a scarring hair loss centered on the vertex of the scalp and spreading peripherally, which is described almost exclusively in African American women. In the late 1960s it was linked to the use of hot combs, but since then almost every method of hair grooming in this population has been associated, albeit weakly, with the disorder. Few studies have examined other possible etiologies, such as immunologic, dermatologic, or genetic factors.
"Given the lack of epidemiologic data, the main goal of [our] study was to elucidate environmental as well as medical risk factors that may be associated with CCCA," the researchers wrote.
They administered questionnaires to 326 African American women attending two churches and a health fair in the Cleveland area. Sixteen of the women ended up being excluded from the analysis.
The questionnaires asked detailed information about family history of male- and female-pattern hair loss; personal medical history, including bacterial and fungal skin infections, autoimmune conditions, and hormonal dysregulation; and hair grooming methods used. The study subjects also underwent a scalp examination that included the grading of hair loss.
A total of 86 of the 310 women (28%) were judged to have central hair loss. Of these, 52 women – 17% of the total study population – had CCCA.
It appeared that hair grooming practices that cause traction, such as weaves and braids, may contribute to the development of CCCA, as women with the most severe central hair loss used these styles more frequently than did those without hair loss. "This has some clinical bearing because traction can clinically produce folliculitis of the scalp, which can cause scarring if this inflammation is prolonged," noted Dr. Kyei and her colleagues (Arch. Dermatol. 2011 April 11 [doi:10.1001/archdermatol.2011.66]).
After paying hundreds of dollars for such hair styling, women often maintain weaves and braids for weeks to months, so any reactive inflammation usually is prolonged, the investigators added.
However, "the relationship between chemical relaxer use and the development of CCCA continues to be murky," they noted. These products clearly weaken the hair shaft, which can cause breakage, but that does not necessarily lead to CCCA. "The fact that most African Americans use chemical relaxers in combination with braiding and other hair grooming practices makes it even more difficult to tease out a relationship," Dr. Kyei and her associates added.
It is difficult to find subjects who do not use chemical relaxers for comparison, since most African Americans begin the practice in childhood. In this study, 91% of the women reported using chemical relaxers, and began doing so at an average age of 10 years.
Nevertheless, "we feel that it is not unreasonable to assume that the scalp may absorb some of the caustic chemicals found in relaxers, which in time leads to damage of the scalp in the form of scarring," they wrote.
Although the prevalence of bacterial skin infections in these study subjects was only 11%, there was a significant elevation among women with CCCA, compared with women who did not have CCCA. The rate of acne also was elevated in affected subjects, but not to a significant degree. Thus, the researchers said that their study "demonstrates that inflammation in the form of bacterial infection and acne may also be contributing to the development of CCCA, a finding consistent with the histopathologic characteristics of this disease," which show a lymphocytic perifollicular infiltrate in its early stages.
In contrast, there was no such association with fungal infections of the scalp, ringworm, or vaginal yeast infections, which was "surprising given how prone this population is to fungal infection," they added.
"One of the most surprising findings ... was the overrepresentation of type 2 [diabetes mellitus] in those with CCCA," the authors noted. Again, the prevalence of type 2 diabetes was low, at 8%, but it was significantly elevated in women with CCCA, compared with those without CCCA.
"These are important data that need further study because CCCA may be a marker of metabolic dysfunction and, when present, can prompt clinicians to do further testing for diabetes mellitus," Dr. Kyei and her colleagues wrote.
Only 9% of the study subjects had thyroid abnormalities, and three-fourths of them had no or minimal hair loss.
A history of male-pattern baldness in the maternal grandfather was found to be a risk factor for CCCA, which suggests that genetics may play a role.
Hormonal dysregulation did not appear to be associated with development of CCCA. Neither were scarring disorders, since only 6% of the subjects reported a history of keloids, and the rate was no higher in women with CCCA than in those without CCCA.
Similarly, there were relatively high prevalences of seborrheic dermatitis (24%), eczema (13%), and contact dermatitis from chemical relaxers (9%), as would be expected in an African American population. However, these conditions were unrelated to CCCA.
This study was supported in part by the North American Hair Research Society and Procter & Gamble. No financial conflicts of interest were reported.
FROM ARCHIVES OF DERMATOLOGY
Major Finding: A total of 86 of the 310 women (28%) were judged to have central hair loss. Of these, 52 women – 17% of the total study population – had CCCA.
Data Source: A community-based cross-sectional survey of 326 African American women.
Disclosures: This study was supported in part by the North American Hair Research Society and Procter & Gamble. No financial conflicts of interest were reported.
Meta-Analysis Questions Memantine Effectiveness for Mild Alzheimer’s
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
Body text goes here
Doctor’s Bio
Body text goes here
Doctor’s Bio
Body text goes here
Doctor’s Bio
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
FROM ARCHIVES OF NEUROLOGY
Major Finding: Memantine showed no efficacy on any measures of cognition, global change, functional activities, or behavior in patients with mild Alzheimer’s disease, and very little efficacy in those with moderate AD.
Data Source: A meta-analysis of three double-blind, randomized clinical trials assessing memantine in 1,128 patients with AD, including 431 with mild AD.
Disclosures: This study was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
Meta-Analysis Questions Memantine Effectiveness for Mild Alzheimer’s
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
Body text goes here
Doctor’s Bio
Body text goes here
Doctor’s Bio
Body text goes here
Doctor’s Bio
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
FROM ARCHIVES OF NEUROLOGY
Major Finding: Memantine showed no efficacy on any measures of cognition, global change, functional activities, or behavior in patients with mild Alzheimer’s disease, and very little efficacy in those with moderate AD.
Data Source: A meta-analysis of three double-blind, randomized clinical trials assessing memantine in 1,128 patients with AD, including 431 with mild AD.
Disclosures: This study was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
Meta-Analysis Questions Memantine Effectiveness for Mild Alzheimer’s
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
Body text goes here
Doctor’s Bio
Body text goes here
Doctor’s Bio
Body text goes here
Doctor’s Bio
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
There is no evidence that memantine is effective for mild Alzheimer’s disease in the very studies that two manufacturers used to win approval of the drug for that indication, according to a report published online April 11 in the Archives of Neurology.
In addition, the effect sizes of memantine’s "benefit" for moderate Alzheimer’s disease (AD) are so small in the same studies that the drug’s usefulness is questionable in that patient group as well, wrote Dr. Lon S. Schneider of the University of Southern California, Los Angeles, and his associates.
Memantine is indicated for moderate to severe AD in the United States and Europe, but often is prescribed "off label" for mild AD or even mild cognitive impairment. Research indicates that up to 63% of patients with mild AD receive the drug, and one study showed that "nearly 40% of U.S. neurologists surveyed reported prescribing memantine at least sometimes to patients with mild cognitive impairment," Dr. Schneider and his colleagues noted.
An American manufacturer recently applied to the Food and Drug Administration and a European manufacturer applied to the European Medicines Agency, to expand approval of memantine to mild AD. In support of their applications, both companies submitted meta-analyses of the same three clinical trials assessing the drug in combined cohorts with mild to moderate AD.
Neither company supplied data separating mild from moderate AD. However, by subtracting data in one meta-analysis from that in the other, Dr. Schneider and his associates were able to directly assess the effects of memantine in mild AD and in moderate AD separately, in their own meta-analysis of these same three clinical trials.
"All three trials included random allocation to treatment, patients and investigators blinded to treatments, and outcome assessments performed while blinded to treatment assignment," they noted.
A total of 1,128 patients completed the studies, including 431 (38%) with mild AD (Arch. Neurol. 2011 April 11 [doi:10.1001/archneurol.2011.69]).
"There was no evidence for the efficacy of memantine in the subset of patients with mild AD on any of the outcomes in any trial or when statistically combined," the investigators wrote.
Memantine exerted "no significant effects" whether patients with mild AD were assessed using the Alzheimer Disease Assessment Scale–Cognitive Subscale (ADAS-cog), the Clinician’s Interview–Based Impression of Change Plus Caregiver’s Input (CIBIC-plus), the Alzheimer Disease Cooperative Study–Activities of Daily Living (ADCS-ADL) scale, or the Neuropsychiatric Inventory (NPI). These instruments assess cognition, global change, functional activities, and behavior, respectively.
Moreover, once data on moderate AD was separated from that on mild AD, there also was no significant difference between memantine and placebo for moderate AD on any of these measures in any of the individual trials, except for a significant effect on the CIBIC-plus in one of the three trials. When the trials were combined, the difference between active drug and placebo was significant on two of the instruments (the ADAS-cog and the CIBIC-plus), albeit with very small effect sizes.
To put these effects sizes into perspective, they were about half of those found in many trials of cholinesterase inhibitors, Dr. Schneider and his colleagues wrote.
"Contrary to the conclusions of the U.S. meta-analysis, we found no effects for memantine on any of the four outcomes in mild AD and no significant effects for memantine on activities of daily living or behavior in the subset of patients with moderate AD," Dr. Schneider and his colleagues wrote.
"The authors of the U.S. meta-analysis incorrectly claimed that memantine ‘demonstrated consistent benefits compared with placebo for cognitive measures, global clinical impression, and functional outcomes across the overall spectrum of AD severity,’ and they further elaborated that their results ‘support the hypothesis that drugs indicated for AD are beneficial for all patients with clinically diagnosed disease, and that arbitrary divisions into severity groups might not be necessary for treatment or drug development in this field.’
"Our results ... do not support these assertions, and they provide insight into why the FDA did not approve the manufacturer’s application to market the drug for mild AD," the investigators wrote.
The investigators added that this lack of evidence for memantine’s efficacy "could come to light only because the European meta-analysis was a subset of the U.S. meta-analysis. If the European meta-analysis had not excluded patients with higher MMSE scores, then it would not have been possible to distinguish the particular lack of evidence for the efficacy of memantine in the mild AD group, which comprised nearly 40% of the patients overall," they wrote.
The findings of the authors’ meta-analysis also support the argument that "results of randomized clinical trials of marketed drugs should be made available soon after trials are completed. Timely release combined with the willingness to openly examine subsets of patients and individual patient outcomes would have allowed earlier recognition of some limitations in memantine’s broader efficacy."
In the United States, that in turn could have spared the half of patients with mild AD and the "substantial portion" of those with mild cognitive impairment from receiving memantine when there is no evidence that the drug is helpful to them and some evidence that it is not helpful, the researchers said.
The meta-analysis was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
FROM ARCHIVES OF NEUROLOGY
Major Finding: Memantine showed no efficacy on any measures of cognition, global change, functional activities, or behavior in patients with mild Alzheimer’s disease, and very little efficacy in those with moderate AD.
Data Source: A meta-analysis of three double-blind, randomized clinical trials assessing memantine in 1,128 patients with AD, including 431 with mild AD.
Disclosures: This study was supported by the U.K.’s Medical Research Council, the California Alzheimer Disease Center program, and the University of Southern California Alzheimer Disease Research Center. Dr. Schneider reported being an editor of the Cochrane Collaboration Dementia and Cognitive Improvement Group, which oversees the systematic review of drugs for cognitive impairment and dementia; he also reported having ties to numerous drug companies, including some that market memantine.
Recurrent Symptoms Do Not Necessarily Reflect Treatment Failure in Crohn's
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Recurrent Symptoms Do Not Necessarily Reflect Treatment Failure in Crohn's
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Recurrent Symptoms Do Not Necessarily Reflect Treatment Failure in Crohn's
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
When patients with Crohn’s disease experience recurrent or persistent symptoms despite taking anti–tumor necrosis factor therapy, clinicians should not automatically assume that the treatment has failed, Dr. David H. Bruining and Dr. William J. Sandborn said in a commentary published in the May issue of Clinical Gastroenterology and Hepatology.
Instead, it is crucial to systematically reassess the patient before making any changes in the treatment regimen, said Dr. Bruining of the Mayo Clinic in Rochester, Minn., and Dr. Sandborn of the University of California, San Diego.
First, make sure the symptoms actually result from Crohn’s disease rather than some other cause, because several studies have shown that symptoms correlate poorly with objective measures of inflammatory disease in the lumen.
Clinical symptoms as measured by the CDAI (Crohn’s Disease Activity Index) have been shown not to correlate with colonoscopic evidence of mucosal ulceration and inflammation as measured by the CDEIS (Crohn’s Disease Endoscopic Index of Severity) or the SES-CD (Simple Endoscopic Score for Crohn’s Disease). Symptoms also do not correlate with measures of C-reactive protein, fecal calprotectin, and fecal lactoferrin. In addition, the latter two measurements cannot detect the presence of structural damage or disease complications, both of which can cause symptoms that overlap with those of active Crohn’s disease.
"CT or MR enterography, and in some selected cases, capsule endoscopy, should be performed prior to altering treatment," with the caveat that capsule endoscopy could potentially precipitate small-bowel obstruction requiring surgery, they wrote (Clin Gastroenterol Hepatol 2011 [doi:10.1016/j.cgh.2011.01.019]).
"Further experience with use of the patency capsule prior to capsule endoscopy may obviate this caveat."
CT or MR assessments are important because they can differentiate the many other comorbid conditions that can produce symptoms that overlap with those of Crohn’s disease. Fibrosis with formation of stricture, as well as transmural penetration of ulcers with the formation of fistulas and abscesses, are two possibilities. Surgery to address ileal involvement can lead to bile acid–induced diarrhea, steatorrhea, and bacterial overgrowth in the small bowel.
Enteric infections (either bacterial or viral) can simulate Crohn’s exacerbations. Clostridium difficile infections have increased dramatically and have become a major concern in patients with Crohn’s disease. Cytomegalovirus (CMV) is less common but can also mislead clinicians into thinking that anti-TNF therapy has failed.
"Both C. difficile and CMV infections should be considered in all symptomatic patients on anti-TNF therapy," Dr. Bruining and Dr. Sandborn noted.
Concomitant irritable bowel syndrome also can present as an exacerbation of Crohn’s symptoms. One study using the Rome II diagnostic criteria for IBS found that nearly 8% of patients had IBS rather than a flare-up of Crohn’s, and another study using the Manning criteria for diagnosing IBS found a rate of 9%.
Major depression also can account for some symptoms – including fatigue – that might be attributed to Crohn’s. One study of 24 patients with Crohn’s disease identified major depression in all 24 of them.
Notably, patients with concurrent depression showed lower response rates to the anti-TNF agent infliximab, compared with patients who were not depressed.
In general, "in those patients with symptoms in whom active disease is not demonstrated or the degree of inflammation is out of proportion to the symptoms, [an alternative] etiology should be sought and treated," the authors wrote.
If the problem turns out to be actual failure of anti-TNF therapy, determine if it is only a partial failure that merely requires some adjustment. Perhaps the current agent "can be manipulated or maximized to improve efficacy" before it is assumed that all anti-TNF agents will be ineffective, they said.
Patients who have mild intolerance, for example, can be managed with conservative support measures. Others who may be intolerant of one specific drug rather than the entire class of anti-TNF agents can be switched to a different anti-TNF drug. One study showed that among 95 patients who were intolerant of infliximab, 22% remitted and more than half responded within 4 weeks to a switch to adalimumab.
In another study involving 198 patients who were intolerant of infliximab because of hypersensitivity reactions, a switch to certolizumab led to a 63% response rate at 6 weeks.
Finally, infliximab is unique in that there is a commercial laboratory test – a measurement of HACA (human antichimeric antibody) levels – to determine whether therapeutic concentrations of the drug have been attained. In one study of 153 patients who were showing loss of response, HACA testing demonstrated that only one-third of the group had achieved therapeutic concentrations of the drug in the circulation. Simple dose escalation improved the rate of clinical response to 86%.
Dr. Bruining reported receiving support in the past from Centocor Ortho Biotech. Dr. Sandborn is a consultant for and has received research grants from Centocor Ortho Biotech, Abbott Laboratories, UCB Pharma, Schering Plough, and Given Imaging.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Young, Healthy Patients Who Get ARDS May Not Recover Completely
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Adults who survive acute respiratory distress syndrome may never recover completely even though their pulmonary function returns to normal, because physical and neuropsychological impairment persists.
Data Source: A longitudinal cohort study of 109 adult survivors of ARDS followed for 5 years.
Disclosures: This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
Young, Healthy Patients Who Get ARDS May Not Recover Completely
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Young, Healthy Patients Who Get ARDS May Not Recover Completely
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Ironically, near-complete recovery of lung function can be expected in the average ARDS survivor, while physical weakness presents the largest burden, said Dr. Jesse B. Hall and Dr. John P. Kress.
The fact that in this study, lung recovery was rapid and sustained "is both remarkable and encouraging" news for patients and their caregivers. Now, strategies to prevent or minimize ICU-acquired weakness are needed, such as early mobilization.
Most of these survivors were gradually able to return to some type of work despite their physical limitations, which "suggests that adaptation to a new handicap was an important part of the recovery process," they noted.
Jesse B. Hall, M.D., and John P. Kress, M.D., are in the section of pulmonary and critical care medicine at the University of Chicago. Dr. Hall reported ties to Hospira and serving as a consultant/witness in legal cases involving product liability, patents, and malpractice. These remarks were taken from their editorial accompanying Dr. Herridge’s report (N. Engl. J. Med. 2011;364:1358-9).
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
Adults who are relatively young and healthy when they develop acute respiratory distress syndrome may never recover completely, according to a report in the April 7 issue of the New England Journal of Medicine.
In a longitudinal study of 109 survivors of ARDS, patients showed persistent limitations on physical activity, a spectrum of physical and neuropsychological impairments, and reduced quality of life 5 years after discharge from an ICU, even though their pulmonary function had returned to near-normal levels, said Dr. Margaret S. Herridge of Sunnybrook Health Sciences Centre, University of Toronto, and her associates.
To date, the highest-quality prospective studies of long-term outcomes in adult ARDS survivors have been limited to only 2 years of follow-up. Dr. Herridge and her colleagues followed a cohort of 109 patients for 1 year, and 83 of these patients for an additional 4 years, to document longer-term outcomes.
The patients had been treated at four academic medical-surgical ICUs in Toronto in 1998-2001. Their median age was 44 years. More than 80% had been working full time and had zero or only one coexisting condition at the time of hospitalization. Most developed ARDS after contracting pneumonia or sepsis.
None of the study subjects had demonstrable weakness at 5 years, and all attained normal or near-normal results on volumetric and spirometric pulmonary testing. Among the 25 who underwent chest CT imaging at some point during follow-up, the most common finding was minor, nondependent pulmonary fibrotic changes consistent with ventilator-induced lung injury. Only the few patients with bronchiectasis, pulmonary fibrosis, or both on CT examination showed clinically important respiratory symptoms such as cough, sputum production, and exertional dyspnea.
Nevertheless, all the patients said they had residual physical weakness of varying degrees, and all said that their capacity for physical activity was reduced after ARDS. These subjective reports were corroborated by an impaired performance on the 6-minute walk test; the median distance achieved was only 76% of that in an age- and sex-matched control population in the literature.
The mean score on the physical component of the SF-36 also remained approximately one standard deviation below that for an age- and sex-matched control population, indicating increased weakness and reduced ability to engage in physical activity.
The study subjects also had a variety of new or persistent impairments related to physical and neuropsychological disorders with an onset related to the ARDS. These included tracheal stenosis requiring surgical resection and dilation (two patients), disability from heterotopic ossification of the knees and elbows (four patients), frozen shoulder (two patients), vocal cord dysfunction and voice changes (one patient), reactive airway disease (four patients), and dental implants after tooth damage acquired in the ICU (one patient).
One patient was disabled from a bilateral forefoot amputation that was required to treat vasopressor-related necrosis, and two others had sensorineural hearing loss and tinnitus attributed to ototoxic ICU medications.
In addition, 10 patients had cosmesis concerns, which caused social isolation or sexual dysfunction, that were related to medical procedures such as laparotomy; tracheostomy; insertion of chest tubes, central lines, or arterial lines; and burns, striae, and facial scars from prolonged mask ventilation.
More than half of the study subjects reported at least one episode of physician-diagnosed depression, anxiety, or both. One patient had an acute psychotic episode due to ICU-related posttraumatic stress disorder, two had severe agitated depression and agoraphobia, and several had other "substantial" mental health challenges.
Moreover, 27% of the subjects reported new-onset mental health problems such as anxiety, depression, or PTSD in their family members. Job loss, disputes over disability and insurance claims, and social isolation also were commonplace. "Patients often required a gradual transition to work, a modified work schedule, or job retraining," Dr. Herridge and her colleagues wrote (N. Engl. J. Med. 2011;364:1293-304).
Overall, the study results suggest that survivors of ARDS "may have an irreversible decrease in function" and that the process of ICU treatment may entail longer-term outcomes than have been appreciated thus far.
Also, "the finding that relatively young, previously working persons with modest coexisting disorders do not return to baseline levels of health care utilization after an episode of critical illness may have important public health ramifications," they noted.
This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Adults who survive acute respiratory distress syndrome may never recover completely even though their pulmonary function returns to normal, because physical and neuropsychological impairment persists.
Data Source: A longitudinal cohort study of 109 adult survivors of ARDS followed for 5 years.
Disclosures: This study was supported by the Canadian Intensive Care Foundation, the Physician’s Services Incorporated Foundation, and the Ontario Thoracic Society. No conflicts of interest were reported.