User login
Cardiovascular MRI: Superior to SPECT for Assessing Stable Angina
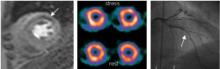
Multiparametric cardiovascular MRI was found superior to single-photon emission computed tomography for evaluating stable angina in "the largest prospective, real-world assessment" of the newer technology done to date, published online Dec. 23 in the Lancet.
Both imaging techniques were compared against x-ray coronary angiography, the reference standard, in 752 men and women with suspected angina pectoris that required assessment to identify significant coronary heart disease.
The study findings "support the wider adoption of [multiparametric cardiovascular magnetic resonance imaging] for the diagnosis and management of stable coronary heart disease patients, in view of the growing concern of the cancer risk associated with medical-source ionizing radiation," said John P. Greenwood, Ph.D., of the Multidisciplinary Cardiovascular Research Centre and Leeds (U.K.) Institute of Genetics, Health, and Therapeutics, and his associates.
Multiparametric cardiovascular MRI allows the assessment of several possible pathologies in a single examination, including ventricular dysfunction, impaired myocardial perfusion, and abnormalities in the coronary artery anatomy. Dr. Greenwood and his colleagues performed the Clinical Evaluation of Magnetic Resonance Imaging in Coronary Heart Disease (CE-MARC) study "to establish the diagnostic accuracy [of the technique] in a large real-world population."
The study subjects were evaluated using cardiovascular MRI, single-photon emission computed tomography (SPECT), and x-ray angiography. The overall prevalence of significant coronary heart disease on x-ray angiography was 39%, which is typical in an outpatient population.
The sensitivity of cardiovascular MRI at detecting coronary heart disease (CHD) was 86.5%, which was significantly greater than the 66.5% sensitivity for SPECT. The specificity of cardiovascular MRI was 83.4%, compared with 82.6% for SPECT, a nonsignificant difference.
The positive predictive value of cardiovascular MRI was 77.2%, compared with 71.4% with SPECT, a nonsignificant difference. And the negative predictive value of cardiovascular MRI was 90.5%, which was significantly greater than the 79.1% negative predictive value for SPECT.
Thus, cardiovascular MRI was as accurate as SPECT in specificity and positive predictive value, but was superior in sensitivity and negative predictive value. "These findings support the wider adoption of cardiovascular MRI for coronary heart disease diagnosis and its inclusion in evidence-based clinical management guidelines," the investigators said (Lancet 2011 Dec. 23 [doi:10.1016/S0140-6736(11)61335-4]).
"Although we have not specifically sought to address why cardiovascular MRI has superior accuracy to SPECT, it might relate partly to its higher spatial resolution (2.2-3.2 mm, vs. approximately 10 mm in-plane for SPECT)," they added.
This study was limited in that the subjects were predominantly white and of northern European ancestry; results might be different in populations of other ethnicities. In addition, CE-MARC was conducted at a single center with a high volume of coronary imaging procedures, the researchers wrote. "Extrapolation to low-volume centers should be made with caution," they said.
This study was supported by the British Heart Foundation. No conflicts of interest were reported.
It is still uncertain whether cardiovascular MRI will replace SPECT or other established tests such as stress echocardiography, said Dr. Robert O. Bonow.
The technique’s superior diagnostic accuracy must be balanced against its limited availability and high cost. Moreover, there is no evidence yet that use of cardiovascular MRI measurably improves patient outcomes.
"Comparative effectiveness trials, such as those underway comparing standard stress imaging methods with noninvasive CT coronary angiography, must show whether cardiovascular MRI provides an incremental increase in value compared with SPECT and echocardiography in clinical practice," he said.
Dr. Bonow is at the Center for Cardiovascular Innovation at Northwestern University Feinberg School of Medicine, Chicago. He reported no financial conflicts of interest. These remarks were taken from his commentary accompanying the report of Dr. Greenwood and colleagues (Lancet 2011 [doi:10.1016/S0140-6736(11)61671-1]).
It is still uncertain whether cardiovascular MRI will replace SPECT or other established tests such as stress echocardiography, said Dr. Robert O. Bonow.
The technique’s superior diagnostic accuracy must be balanced against its limited availability and high cost. Moreover, there is no evidence yet that use of cardiovascular MRI measurably improves patient outcomes.
"Comparative effectiveness trials, such as those underway comparing standard stress imaging methods with noninvasive CT coronary angiography, must show whether cardiovascular MRI provides an incremental increase in value compared with SPECT and echocardiography in clinical practice," he said.
Dr. Bonow is at the Center for Cardiovascular Innovation at Northwestern University Feinberg School of Medicine, Chicago. He reported no financial conflicts of interest. These remarks were taken from his commentary accompanying the report of Dr. Greenwood and colleagues (Lancet 2011 [doi:10.1016/S0140-6736(11)61671-1]).
It is still uncertain whether cardiovascular MRI will replace SPECT or other established tests such as stress echocardiography, said Dr. Robert O. Bonow.
The technique’s superior diagnostic accuracy must be balanced against its limited availability and high cost. Moreover, there is no evidence yet that use of cardiovascular MRI measurably improves patient outcomes.
"Comparative effectiveness trials, such as those underway comparing standard stress imaging methods with noninvasive CT coronary angiography, must show whether cardiovascular MRI provides an incremental increase in value compared with SPECT and echocardiography in clinical practice," he said.
Dr. Bonow is at the Center for Cardiovascular Innovation at Northwestern University Feinberg School of Medicine, Chicago. He reported no financial conflicts of interest. These remarks were taken from his commentary accompanying the report of Dr. Greenwood and colleagues (Lancet 2011 [doi:10.1016/S0140-6736(11)61671-1]).
Multiparametric cardiovascular MRI was found superior to single-photon emission computed tomography for evaluating stable angina in "the largest prospective, real-world assessment" of the newer technology done to date, published online Dec. 23 in the Lancet.
Both imaging techniques were compared against x-ray coronary angiography, the reference standard, in 752 men and women with suspected angina pectoris that required assessment to identify significant coronary heart disease.
The study findings "support the wider adoption of [multiparametric cardiovascular magnetic resonance imaging] for the diagnosis and management of stable coronary heart disease patients, in view of the growing concern of the cancer risk associated with medical-source ionizing radiation," said John P. Greenwood, Ph.D., of the Multidisciplinary Cardiovascular Research Centre and Leeds (U.K.) Institute of Genetics, Health, and Therapeutics, and his associates.
Multiparametric cardiovascular MRI allows the assessment of several possible pathologies in a single examination, including ventricular dysfunction, impaired myocardial perfusion, and abnormalities in the coronary artery anatomy. Dr. Greenwood and his colleagues performed the Clinical Evaluation of Magnetic Resonance Imaging in Coronary Heart Disease (CE-MARC) study "to establish the diagnostic accuracy [of the technique] in a large real-world population."
The study subjects were evaluated using cardiovascular MRI, single-photon emission computed tomography (SPECT), and x-ray angiography. The overall prevalence of significant coronary heart disease on x-ray angiography was 39%, which is typical in an outpatient population.
The sensitivity of cardiovascular MRI at detecting coronary heart disease (CHD) was 86.5%, which was significantly greater than the 66.5% sensitivity for SPECT. The specificity of cardiovascular MRI was 83.4%, compared with 82.6% for SPECT, a nonsignificant difference.
The positive predictive value of cardiovascular MRI was 77.2%, compared with 71.4% with SPECT, a nonsignificant difference. And the negative predictive value of cardiovascular MRI was 90.5%, which was significantly greater than the 79.1% negative predictive value for SPECT.
Thus, cardiovascular MRI was as accurate as SPECT in specificity and positive predictive value, but was superior in sensitivity and negative predictive value. "These findings support the wider adoption of cardiovascular MRI for coronary heart disease diagnosis and its inclusion in evidence-based clinical management guidelines," the investigators said (Lancet 2011 Dec. 23 [doi:10.1016/S0140-6736(11)61335-4]).
"Although we have not specifically sought to address why cardiovascular MRI has superior accuracy to SPECT, it might relate partly to its higher spatial resolution (2.2-3.2 mm, vs. approximately 10 mm in-plane for SPECT)," they added.
This study was limited in that the subjects were predominantly white and of northern European ancestry; results might be different in populations of other ethnicities. In addition, CE-MARC was conducted at a single center with a high volume of coronary imaging procedures, the researchers wrote. "Extrapolation to low-volume centers should be made with caution," they said.
This study was supported by the British Heart Foundation. No conflicts of interest were reported.
Multiparametric cardiovascular MRI was found superior to single-photon emission computed tomography for evaluating stable angina in "the largest prospective, real-world assessment" of the newer technology done to date, published online Dec. 23 in the Lancet.
Both imaging techniques were compared against x-ray coronary angiography, the reference standard, in 752 men and women with suspected angina pectoris that required assessment to identify significant coronary heart disease.
The study findings "support the wider adoption of [multiparametric cardiovascular magnetic resonance imaging] for the diagnosis and management of stable coronary heart disease patients, in view of the growing concern of the cancer risk associated with medical-source ionizing radiation," said John P. Greenwood, Ph.D., of the Multidisciplinary Cardiovascular Research Centre and Leeds (U.K.) Institute of Genetics, Health, and Therapeutics, and his associates.
Multiparametric cardiovascular MRI allows the assessment of several possible pathologies in a single examination, including ventricular dysfunction, impaired myocardial perfusion, and abnormalities in the coronary artery anatomy. Dr. Greenwood and his colleagues performed the Clinical Evaluation of Magnetic Resonance Imaging in Coronary Heart Disease (CE-MARC) study "to establish the diagnostic accuracy [of the technique] in a large real-world population."
The study subjects were evaluated using cardiovascular MRI, single-photon emission computed tomography (SPECT), and x-ray angiography. The overall prevalence of significant coronary heart disease on x-ray angiography was 39%, which is typical in an outpatient population.
The sensitivity of cardiovascular MRI at detecting coronary heart disease (CHD) was 86.5%, which was significantly greater than the 66.5% sensitivity for SPECT. The specificity of cardiovascular MRI was 83.4%, compared with 82.6% for SPECT, a nonsignificant difference.
The positive predictive value of cardiovascular MRI was 77.2%, compared with 71.4% with SPECT, a nonsignificant difference. And the negative predictive value of cardiovascular MRI was 90.5%, which was significantly greater than the 79.1% negative predictive value for SPECT.
Thus, cardiovascular MRI was as accurate as SPECT in specificity and positive predictive value, but was superior in sensitivity and negative predictive value. "These findings support the wider adoption of cardiovascular MRI for coronary heart disease diagnosis and its inclusion in evidence-based clinical management guidelines," the investigators said (Lancet 2011 Dec. 23 [doi:10.1016/S0140-6736(11)61335-4]).
"Although we have not specifically sought to address why cardiovascular MRI has superior accuracy to SPECT, it might relate partly to its higher spatial resolution (2.2-3.2 mm, vs. approximately 10 mm in-plane for SPECT)," they added.
This study was limited in that the subjects were predominantly white and of northern European ancestry; results might be different in populations of other ethnicities. In addition, CE-MARC was conducted at a single center with a high volume of coronary imaging procedures, the researchers wrote. "Extrapolation to low-volume centers should be made with caution," they said.
This study was supported by the British Heart Foundation. No conflicts of interest were reported.
FROM THE LANCET
Major Finding: Cardiovascular MRI had superior sensitivity (86.5% vs 66.5%) and negative predictive value (90.5% vs 79.1%), compared with SPECT, and had comparable specificity (83.4% vs 82.6%) and positive predictive value (77.2% vs 71.4%).
Data Source: Prospective trial comparing cardiovascular MRI and SPECT against x-ray coronary angiography in assessing stable angina in 752 patients.
Disclosures: This study was supported by the British Heart Foundation. No conflicts of interest were reported.
Aflibercept Prevents Ascites Recurrence in Advanced Ovarian Cancer
The monoclonal antibody aflibercept, a vascular endothelial growth factor blocker, prevented the recurrence of malignant ascites in a phase II clinical trial involving 55 patients with advanced ovarian cancer, according to a report published online Dec. 21 in Lancet Oncology.
Compared with placebo, aflibercept extended the time to repeat paracentesis, increased the duration of paracentesis-free survival, and reduced the frequency of paracentesis during 60 days of follow-up. It also improved symptoms of abdominal bloating, discomfort, and pain, and allowed patients to move more comfortably, said Dr. Walter H. Gotlieb of McGill University and Jewish General Hospital, Montreal, and his associates.
"Anti-VEGF [vascular endothelial growth factor] treatments such as aflibercept appear to reduce the formation of malignant ascites and could dramatically improve the quality of life for patients with this debilitating complication. But clinicians must exercise caution in their use of aflibercept because of the significantly increased risk of fatal bowel perforation," Dr. Gotlieb noted in a press statement accompanying the report.
There were three fatal gastrointestinal perforations among study subjects who received aflibercept and one among those who received placebo. This complication has been reported previously in a pilot study of the drug, as well as in studies of the related monoclonal antibody bevacizumab, the investigators noted.
In this double-blind trial, women with advanced ovarian epithelial cancer that was resistant to a median of four types of chemotherapy were enrolled at 23 sites in Belgium, Canada, Hungary, India, Israel, the United Kingdom, and the United States. Most had serous cancer and poorly differentiated tumors. The study subjects had experienced recurrent malignant ascites and required up to four paracenteses per month to relieve their symptoms.
The subjects were randomly assigned to receive IV aflibercept (29 women) or placebo (26 women) every 2 weeks for at least 60 days, but for no longer than 6 months. They were allowed to cross over to open-label treatment at 6 months.
The primary end point was the time to repeat paracentesis. This interval was significantly longer with aflibercept (55 days) than with placebo (23 days).
Paracentesis-free survival also was significantly longer with aflibercept (42 days) than with placebo (18 days). Notably, two women who received the active drug did not require any repeat paracentesis during 6 months of treatment.
The median frequency of paracentesis procedures within 60 days of beginning treatment was 2 (range, 0-9 procedures) with aflibercept, which was significantly less than the median frequency of 4 procedures (range, 0-17 procedures) that were required with placebo.
In addition, the women who received the active drug reported greater improvement on a questionnaire that assessed abdominal pain, abdominal bloating, abdominal discomfort, and restricted movement, the investigators said (Lancet Oncol. 2011 [doi:10.1016/S1470-2045(11)70338-2]).
There were no significant differences between aflibercept and placebo in overall survival or progression-free survival.
All the women who received aflibercept and all but two of those who received placebo reported adverse effects that commonly occur with advanced intraperitoneal disease, chiefly nausea, vomiting, diarrhea, fatigue, asthenia, peripheral edema, dyspnea, and cough. Potentially serious adverse effects possibly related to VEGF blockade were more common in the aflibercept group, and included venous thromboembolism, hypertension, proteinuria, and bowel perforation.
There was "an imbalance" between the aflibercept and placebo groups in the number of deaths related to study treatment (four vs. zero) and in the number of fatal gastrointestinal perforations (three vs. one).
"Anti-VEGF treatments such as aflibercept appear to reduce the formation of malignant ascites."
All three intestinal perforations in the aflibercept group occurred early in the course of treatment (during the first or second treatment cycle), and two occurred in the context of disease progression. One of these patients had experienced two recent episodes of intestinal obstruction and another had a deep tumor mass infiltrating the sigmoid.
Six deaths among women receiving aflibercept were unrelated to cancer progression: one due to suspected pulmonary embolus possibly related to the study drug, and one each due to dyspnea, intestinal perforation, pneumonia, aspiration pneumonia, and an unknown cause. In comparison, there were two deaths in the placebo group, which were due to sepsis and aspiration.
Four patients who received aflibercept developed grade 3 or 4 liver abnormalities, as did one patient who received placebo.
The study results "clearly show the effectiveness of VEGF blockade in the reduction of ascites formation." However, the number of bowel perforations "point[s] to an unfavorable benefit-risk balance for this therapeutic anti-VEGF intervention in heavily pretreated patients with ovarian cancer," Dr. Gotlieb and his associates said.
"A patient population not as advanced in their disease or without a history of bowel obstruction or with alternative regimens might have a different benefit-risk balance," they added.
This study was sponsored by Sanofi Oncology. Dr. Gotlieb reported ties to Sanofi-Aventis, and his associates reported ties to numerous industry sources.
This study clearly demonstrates aflibercept’s efficacy at preventing recurrent ascites, but symptom relief must be weighed against potentially life-threatening adverse events such as bowel perforation, said Dr. Gerhild Becker and Dr. Hubert E. Blum.
It is possible that careful patient selection could decrease the risk of such perforations. However, "before a general recommendation of aflibercept for the treatment of malignant ascites can be made, further studies ... are needed to compare the effectiveness of the different therapeutic strategies in daily clinical practice," they said.
Dr. Becker is in the department of palliative care and Dr. Blum is in internal medicine at the University Hospital Freiburg (Germany). They reported no financial conflicts of interest. These remarks were taken from their editorial comment accompanying Dr. Gotlieb’s report (Lancet Oncol. 2011 [doi:10.1016/S1470-2045(11)70394-1]).
This study clearly demonstrates aflibercept’s efficacy at preventing recurrent ascites, but symptom relief must be weighed against potentially life-threatening adverse events such as bowel perforation, said Dr. Gerhild Becker and Dr. Hubert E. Blum.
It is possible that careful patient selection could decrease the risk of such perforations. However, "before a general recommendation of aflibercept for the treatment of malignant ascites can be made, further studies ... are needed to compare the effectiveness of the different therapeutic strategies in daily clinical practice," they said.
Dr. Becker is in the department of palliative care and Dr. Blum is in internal medicine at the University Hospital Freiburg (Germany). They reported no financial conflicts of interest. These remarks were taken from their editorial comment accompanying Dr. Gotlieb’s report (Lancet Oncol. 2011 [doi:10.1016/S1470-2045(11)70394-1]).
This study clearly demonstrates aflibercept’s efficacy at preventing recurrent ascites, but symptom relief must be weighed against potentially life-threatening adverse events such as bowel perforation, said Dr. Gerhild Becker and Dr. Hubert E. Blum.
It is possible that careful patient selection could decrease the risk of such perforations. However, "before a general recommendation of aflibercept for the treatment of malignant ascites can be made, further studies ... are needed to compare the effectiveness of the different therapeutic strategies in daily clinical practice," they said.
Dr. Becker is in the department of palliative care and Dr. Blum is in internal medicine at the University Hospital Freiburg (Germany). They reported no financial conflicts of interest. These remarks were taken from their editorial comment accompanying Dr. Gotlieb’s report (Lancet Oncol. 2011 [doi:10.1016/S1470-2045(11)70394-1]).
The monoclonal antibody aflibercept, a vascular endothelial growth factor blocker, prevented the recurrence of malignant ascites in a phase II clinical trial involving 55 patients with advanced ovarian cancer, according to a report published online Dec. 21 in Lancet Oncology.
Compared with placebo, aflibercept extended the time to repeat paracentesis, increased the duration of paracentesis-free survival, and reduced the frequency of paracentesis during 60 days of follow-up. It also improved symptoms of abdominal bloating, discomfort, and pain, and allowed patients to move more comfortably, said Dr. Walter H. Gotlieb of McGill University and Jewish General Hospital, Montreal, and his associates.
"Anti-VEGF [vascular endothelial growth factor] treatments such as aflibercept appear to reduce the formation of malignant ascites and could dramatically improve the quality of life for patients with this debilitating complication. But clinicians must exercise caution in their use of aflibercept because of the significantly increased risk of fatal bowel perforation," Dr. Gotlieb noted in a press statement accompanying the report.
There were three fatal gastrointestinal perforations among study subjects who received aflibercept and one among those who received placebo. This complication has been reported previously in a pilot study of the drug, as well as in studies of the related monoclonal antibody bevacizumab, the investigators noted.
In this double-blind trial, women with advanced ovarian epithelial cancer that was resistant to a median of four types of chemotherapy were enrolled at 23 sites in Belgium, Canada, Hungary, India, Israel, the United Kingdom, and the United States. Most had serous cancer and poorly differentiated tumors. The study subjects had experienced recurrent malignant ascites and required up to four paracenteses per month to relieve their symptoms.
The subjects were randomly assigned to receive IV aflibercept (29 women) or placebo (26 women) every 2 weeks for at least 60 days, but for no longer than 6 months. They were allowed to cross over to open-label treatment at 6 months.
The primary end point was the time to repeat paracentesis. This interval was significantly longer with aflibercept (55 days) than with placebo (23 days).
Paracentesis-free survival also was significantly longer with aflibercept (42 days) than with placebo (18 days). Notably, two women who received the active drug did not require any repeat paracentesis during 6 months of treatment.
The median frequency of paracentesis procedures within 60 days of beginning treatment was 2 (range, 0-9 procedures) with aflibercept, which was significantly less than the median frequency of 4 procedures (range, 0-17 procedures) that were required with placebo.
In addition, the women who received the active drug reported greater improvement on a questionnaire that assessed abdominal pain, abdominal bloating, abdominal discomfort, and restricted movement, the investigators said (Lancet Oncol. 2011 [doi:10.1016/S1470-2045(11)70338-2]).
There were no significant differences between aflibercept and placebo in overall survival or progression-free survival.
All the women who received aflibercept and all but two of those who received placebo reported adverse effects that commonly occur with advanced intraperitoneal disease, chiefly nausea, vomiting, diarrhea, fatigue, asthenia, peripheral edema, dyspnea, and cough. Potentially serious adverse effects possibly related to VEGF blockade were more common in the aflibercept group, and included venous thromboembolism, hypertension, proteinuria, and bowel perforation.
There was "an imbalance" between the aflibercept and placebo groups in the number of deaths related to study treatment (four vs. zero) and in the number of fatal gastrointestinal perforations (three vs. one).
"Anti-VEGF treatments such as aflibercept appear to reduce the formation of malignant ascites."
All three intestinal perforations in the aflibercept group occurred early in the course of treatment (during the first or second treatment cycle), and two occurred in the context of disease progression. One of these patients had experienced two recent episodes of intestinal obstruction and another had a deep tumor mass infiltrating the sigmoid.
Six deaths among women receiving aflibercept were unrelated to cancer progression: one due to suspected pulmonary embolus possibly related to the study drug, and one each due to dyspnea, intestinal perforation, pneumonia, aspiration pneumonia, and an unknown cause. In comparison, there were two deaths in the placebo group, which were due to sepsis and aspiration.
Four patients who received aflibercept developed grade 3 or 4 liver abnormalities, as did one patient who received placebo.
The study results "clearly show the effectiveness of VEGF blockade in the reduction of ascites formation." However, the number of bowel perforations "point[s] to an unfavorable benefit-risk balance for this therapeutic anti-VEGF intervention in heavily pretreated patients with ovarian cancer," Dr. Gotlieb and his associates said.
"A patient population not as advanced in their disease or without a history of bowel obstruction or with alternative regimens might have a different benefit-risk balance," they added.
This study was sponsored by Sanofi Oncology. Dr. Gotlieb reported ties to Sanofi-Aventis, and his associates reported ties to numerous industry sources.
The monoclonal antibody aflibercept, a vascular endothelial growth factor blocker, prevented the recurrence of malignant ascites in a phase II clinical trial involving 55 patients with advanced ovarian cancer, according to a report published online Dec. 21 in Lancet Oncology.
Compared with placebo, aflibercept extended the time to repeat paracentesis, increased the duration of paracentesis-free survival, and reduced the frequency of paracentesis during 60 days of follow-up. It also improved symptoms of abdominal bloating, discomfort, and pain, and allowed patients to move more comfortably, said Dr. Walter H. Gotlieb of McGill University and Jewish General Hospital, Montreal, and his associates.
"Anti-VEGF [vascular endothelial growth factor] treatments such as aflibercept appear to reduce the formation of malignant ascites and could dramatically improve the quality of life for patients with this debilitating complication. But clinicians must exercise caution in their use of aflibercept because of the significantly increased risk of fatal bowel perforation," Dr. Gotlieb noted in a press statement accompanying the report.
There were three fatal gastrointestinal perforations among study subjects who received aflibercept and one among those who received placebo. This complication has been reported previously in a pilot study of the drug, as well as in studies of the related monoclonal antibody bevacizumab, the investigators noted.
In this double-blind trial, women with advanced ovarian epithelial cancer that was resistant to a median of four types of chemotherapy were enrolled at 23 sites in Belgium, Canada, Hungary, India, Israel, the United Kingdom, and the United States. Most had serous cancer and poorly differentiated tumors. The study subjects had experienced recurrent malignant ascites and required up to four paracenteses per month to relieve their symptoms.
The subjects were randomly assigned to receive IV aflibercept (29 women) or placebo (26 women) every 2 weeks for at least 60 days, but for no longer than 6 months. They were allowed to cross over to open-label treatment at 6 months.
The primary end point was the time to repeat paracentesis. This interval was significantly longer with aflibercept (55 days) than with placebo (23 days).
Paracentesis-free survival also was significantly longer with aflibercept (42 days) than with placebo (18 days). Notably, two women who received the active drug did not require any repeat paracentesis during 6 months of treatment.
The median frequency of paracentesis procedures within 60 days of beginning treatment was 2 (range, 0-9 procedures) with aflibercept, which was significantly less than the median frequency of 4 procedures (range, 0-17 procedures) that were required with placebo.
In addition, the women who received the active drug reported greater improvement on a questionnaire that assessed abdominal pain, abdominal bloating, abdominal discomfort, and restricted movement, the investigators said (Lancet Oncol. 2011 [doi:10.1016/S1470-2045(11)70338-2]).
There were no significant differences between aflibercept and placebo in overall survival or progression-free survival.
All the women who received aflibercept and all but two of those who received placebo reported adverse effects that commonly occur with advanced intraperitoneal disease, chiefly nausea, vomiting, diarrhea, fatigue, asthenia, peripheral edema, dyspnea, and cough. Potentially serious adverse effects possibly related to VEGF blockade were more common in the aflibercept group, and included venous thromboembolism, hypertension, proteinuria, and bowel perforation.
There was "an imbalance" between the aflibercept and placebo groups in the number of deaths related to study treatment (four vs. zero) and in the number of fatal gastrointestinal perforations (three vs. one).
"Anti-VEGF treatments such as aflibercept appear to reduce the formation of malignant ascites."
All three intestinal perforations in the aflibercept group occurred early in the course of treatment (during the first or second treatment cycle), and two occurred in the context of disease progression. One of these patients had experienced two recent episodes of intestinal obstruction and another had a deep tumor mass infiltrating the sigmoid.
Six deaths among women receiving aflibercept were unrelated to cancer progression: one due to suspected pulmonary embolus possibly related to the study drug, and one each due to dyspnea, intestinal perforation, pneumonia, aspiration pneumonia, and an unknown cause. In comparison, there were two deaths in the placebo group, which were due to sepsis and aspiration.
Four patients who received aflibercept developed grade 3 or 4 liver abnormalities, as did one patient who received placebo.
The study results "clearly show the effectiveness of VEGF blockade in the reduction of ascites formation." However, the number of bowel perforations "point[s] to an unfavorable benefit-risk balance for this therapeutic anti-VEGF intervention in heavily pretreated patients with ovarian cancer," Dr. Gotlieb and his associates said.
"A patient population not as advanced in their disease or without a history of bowel obstruction or with alternative regimens might have a different benefit-risk balance," they added.
This study was sponsored by Sanofi Oncology. Dr. Gotlieb reported ties to Sanofi-Aventis, and his associates reported ties to numerous industry sources.
FROM LANCET ONCOLOGY
Major Finding: Aflibercept extended the time until ascites recurrence required paracentesis to 55 days, compared with 23 days for placebo.
Data Source: A multicenter double-blind randomized phase II clinical trial involving 55 women with advanced, chemoresistant ovarian cancer and recurrent malignant ascites.
Disclosures: This study was sponsored by Sanofi Oncology. Dr. Gotlieb reported ties to Sanofi-Aventis, and his associates reported ties to numerous industry sources.
Immunosuppression Phase in Sepsis May Hold Treatment Option
During the natural course of sepsis, patients enter an immunosuppressed state after the initial intense inflammatory response well known to clinicians as a "cytokine storm," according to a report in the Dec. 21 issue of JAMA.
Most therapies for sepsis target this initial hyperinflammatory state and are focused on blocking inflammation and immune activation. They may be successful if used early in the course of sepsis, but harmful if used during the later, underrecognized immunosuppressed phase, said Jonathan S. Boomer, Ph.D., of the department of medicine, Washington University, St. Louis, and his associates.
This latter phase of sepsis only came to light once clinical management improved enough to allow patients to survive the early hyperinflammatory phase. Then clinicians began noting that patients who survived early sepsis often developed nosocomial infections with organisms that typically do not affect immunocompetent hosts. These patients also frequently experienced reactivation of latent viruses, Dr. Boomer and his colleagues said.
These observations lead some to hypothesize that hyperinflammation gives way to significant immunosuppression in such patients. The hypothesis has been controversial. Dr. Boomer and his associates explored the issue through rapid postmortem examination of cells from the spleens and lungs of affected patients – "a lymphoid organ and a peripheral organ that [are] frequent site[s]of nosocomial infection."
They assessed the organs of 40 patients who died while being treated for sepsis in surgical or medical ICUs, and in control samples from 29 patients who had critical illnesses that did not involve sepsis. The control tissue came from organ donors, trauma patients who required emergency splenectomy, and non–tumor-involved lung tissue from lung cancer patients who underwent lobectomies.
The causes of sepsis included ventilator-associated pneumonia, peritonitis, necrotizing fasciitis, retroperitoneal abscess, infected intravascular catheters, UTI, intrapelvic abscess, and osteomyelitis.
Compared with cells from control spleens, splenocytes from sepsis patients showed profound impairment of cytokine production when stimulated in vitro. At 5 hours after collection, the secretion of cytokines from splenocytes of sepsis patients was less than 10% of that secreted by control splenocytes, the investigators said (JAMA 2011;306:2594-605).
Splenocytes from most sepsis patients showed some recovery of cytokine production at 22 hours, but they still secreted only one-third the number of cytokines produced by control splenocytes. This result was consistent for all the cytokines tested and for all subgroups of patients, regardless of the duration of sepsis, patient age, whether or not corticosteroids had been received, and patient nutritional status at the time of death.
The researchers identified many separate mechanisms by which immune responses were inhibited in spleen cells. They found a decrease in stimulatory molecules such as CD28 on T cells, a deficiency in antigen-presenting cells such as macrophages and dendritic cells, increased expression of inhibitory ligands that suppress immune function, and an excess of inhibitory cells such as myeloid-derived suppressor cells and regulatory T cells.
Lung tissue similarly showed significant immunosuppression in sepsis patients, compared with control patients. In particular, lung alveolar epithelial cells and endothelial cells showed an excess of inhibitory receptors and ligands.
The study findings suggest that it may be possible to identify sepsis patients who enter this phase of immunocompromise and treat them with immune-enhancing therapies such as interleukin, the researchers said.
The study was limited by its small sample size and the heterogeneous nature of both the sepsis and control patients, the authors said. They further emphasized that malnutrition should be recognized for its possible effects on host immunity and that for a number of reasons, their study "must be viewed cautiously." The research "serves as a bridge between preclinical and early clinical findings," they wrote.
This study was supported by the National Institutes of Health. One of Dr. Boomer’s associates reported receiving grants from Pfizer, Bristol-Meyers Squibb, and Aurigine.
Dr. Boomer and colleagues have demonstrated a broad array of cellular changes associated with the loss of immune competence in patients with sepsis, noted Dr. Peter A. Ward.
The next step would be to determine whether these derangements can be reversed by treatments to enhance immune competence, such as interleukins. The study findings underscore "the desperate need for a better understanding of sepsis and the urgent need for new therapeutic strategies," he said.
Dr. Ward is in the department of pathology at the University of Michigan, Ann Arbor. His work is supported in part by the National Institutes of Health. He reported no financial conflicts of interest. These remarks were adapted from his editorial accompanying Dr. Boomer’s report (JAMA 2011;306:2618-9).
Dr. Boomer and colleagues have demonstrated a broad array of cellular changes associated with the loss of immune competence in patients with sepsis, noted Dr. Peter A. Ward.
The next step would be to determine whether these derangements can be reversed by treatments to enhance immune competence, such as interleukins. The study findings underscore "the desperate need for a better understanding of sepsis and the urgent need for new therapeutic strategies," he said.
Dr. Ward is in the department of pathology at the University of Michigan, Ann Arbor. His work is supported in part by the National Institutes of Health. He reported no financial conflicts of interest. These remarks were adapted from his editorial accompanying Dr. Boomer’s report (JAMA 2011;306:2618-9).
Dr. Boomer and colleagues have demonstrated a broad array of cellular changes associated with the loss of immune competence in patients with sepsis, noted Dr. Peter A. Ward.
The next step would be to determine whether these derangements can be reversed by treatments to enhance immune competence, such as interleukins. The study findings underscore "the desperate need for a better understanding of sepsis and the urgent need for new therapeutic strategies," he said.
Dr. Ward is in the department of pathology at the University of Michigan, Ann Arbor. His work is supported in part by the National Institutes of Health. He reported no financial conflicts of interest. These remarks were adapted from his editorial accompanying Dr. Boomer’s report (JAMA 2011;306:2618-9).
During the natural course of sepsis, patients enter an immunosuppressed state after the initial intense inflammatory response well known to clinicians as a "cytokine storm," according to a report in the Dec. 21 issue of JAMA.
Most therapies for sepsis target this initial hyperinflammatory state and are focused on blocking inflammation and immune activation. They may be successful if used early in the course of sepsis, but harmful if used during the later, underrecognized immunosuppressed phase, said Jonathan S. Boomer, Ph.D., of the department of medicine, Washington University, St. Louis, and his associates.
This latter phase of sepsis only came to light once clinical management improved enough to allow patients to survive the early hyperinflammatory phase. Then clinicians began noting that patients who survived early sepsis often developed nosocomial infections with organisms that typically do not affect immunocompetent hosts. These patients also frequently experienced reactivation of latent viruses, Dr. Boomer and his colleagues said.
These observations lead some to hypothesize that hyperinflammation gives way to significant immunosuppression in such patients. The hypothesis has been controversial. Dr. Boomer and his associates explored the issue through rapid postmortem examination of cells from the spleens and lungs of affected patients – "a lymphoid organ and a peripheral organ that [are] frequent site[s]of nosocomial infection."
They assessed the organs of 40 patients who died while being treated for sepsis in surgical or medical ICUs, and in control samples from 29 patients who had critical illnesses that did not involve sepsis. The control tissue came from organ donors, trauma patients who required emergency splenectomy, and non–tumor-involved lung tissue from lung cancer patients who underwent lobectomies.
The causes of sepsis included ventilator-associated pneumonia, peritonitis, necrotizing fasciitis, retroperitoneal abscess, infected intravascular catheters, UTI, intrapelvic abscess, and osteomyelitis.
Compared with cells from control spleens, splenocytes from sepsis patients showed profound impairment of cytokine production when stimulated in vitro. At 5 hours after collection, the secretion of cytokines from splenocytes of sepsis patients was less than 10% of that secreted by control splenocytes, the investigators said (JAMA 2011;306:2594-605).
Splenocytes from most sepsis patients showed some recovery of cytokine production at 22 hours, but they still secreted only one-third the number of cytokines produced by control splenocytes. This result was consistent for all the cytokines tested and for all subgroups of patients, regardless of the duration of sepsis, patient age, whether or not corticosteroids had been received, and patient nutritional status at the time of death.
The researchers identified many separate mechanisms by which immune responses were inhibited in spleen cells. They found a decrease in stimulatory molecules such as CD28 on T cells, a deficiency in antigen-presenting cells such as macrophages and dendritic cells, increased expression of inhibitory ligands that suppress immune function, and an excess of inhibitory cells such as myeloid-derived suppressor cells and regulatory T cells.
Lung tissue similarly showed significant immunosuppression in sepsis patients, compared with control patients. In particular, lung alveolar epithelial cells and endothelial cells showed an excess of inhibitory receptors and ligands.
The study findings suggest that it may be possible to identify sepsis patients who enter this phase of immunocompromise and treat them with immune-enhancing therapies such as interleukin, the researchers said.
The study was limited by its small sample size and the heterogeneous nature of both the sepsis and control patients, the authors said. They further emphasized that malnutrition should be recognized for its possible effects on host immunity and that for a number of reasons, their study "must be viewed cautiously." The research "serves as a bridge between preclinical and early clinical findings," they wrote.
This study was supported by the National Institutes of Health. One of Dr. Boomer’s associates reported receiving grants from Pfizer, Bristol-Meyers Squibb, and Aurigine.
During the natural course of sepsis, patients enter an immunosuppressed state after the initial intense inflammatory response well known to clinicians as a "cytokine storm," according to a report in the Dec. 21 issue of JAMA.
Most therapies for sepsis target this initial hyperinflammatory state and are focused on blocking inflammation and immune activation. They may be successful if used early in the course of sepsis, but harmful if used during the later, underrecognized immunosuppressed phase, said Jonathan S. Boomer, Ph.D., of the department of medicine, Washington University, St. Louis, and his associates.
This latter phase of sepsis only came to light once clinical management improved enough to allow patients to survive the early hyperinflammatory phase. Then clinicians began noting that patients who survived early sepsis often developed nosocomial infections with organisms that typically do not affect immunocompetent hosts. These patients also frequently experienced reactivation of latent viruses, Dr. Boomer and his colleagues said.
These observations lead some to hypothesize that hyperinflammation gives way to significant immunosuppression in such patients. The hypothesis has been controversial. Dr. Boomer and his associates explored the issue through rapid postmortem examination of cells from the spleens and lungs of affected patients – "a lymphoid organ and a peripheral organ that [are] frequent site[s]of nosocomial infection."
They assessed the organs of 40 patients who died while being treated for sepsis in surgical or medical ICUs, and in control samples from 29 patients who had critical illnesses that did not involve sepsis. The control tissue came from organ donors, trauma patients who required emergency splenectomy, and non–tumor-involved lung tissue from lung cancer patients who underwent lobectomies.
The causes of sepsis included ventilator-associated pneumonia, peritonitis, necrotizing fasciitis, retroperitoneal abscess, infected intravascular catheters, UTI, intrapelvic abscess, and osteomyelitis.
Compared with cells from control spleens, splenocytes from sepsis patients showed profound impairment of cytokine production when stimulated in vitro. At 5 hours after collection, the secretion of cytokines from splenocytes of sepsis patients was less than 10% of that secreted by control splenocytes, the investigators said (JAMA 2011;306:2594-605).
Splenocytes from most sepsis patients showed some recovery of cytokine production at 22 hours, but they still secreted only one-third the number of cytokines produced by control splenocytes. This result was consistent for all the cytokines tested and for all subgroups of patients, regardless of the duration of sepsis, patient age, whether or not corticosteroids had been received, and patient nutritional status at the time of death.
The researchers identified many separate mechanisms by which immune responses were inhibited in spleen cells. They found a decrease in stimulatory molecules such as CD28 on T cells, a deficiency in antigen-presenting cells such as macrophages and dendritic cells, increased expression of inhibitory ligands that suppress immune function, and an excess of inhibitory cells such as myeloid-derived suppressor cells and regulatory T cells.
Lung tissue similarly showed significant immunosuppression in sepsis patients, compared with control patients. In particular, lung alveolar epithelial cells and endothelial cells showed an excess of inhibitory receptors and ligands.
The study findings suggest that it may be possible to identify sepsis patients who enter this phase of immunocompromise and treat them with immune-enhancing therapies such as interleukin, the researchers said.
The study was limited by its small sample size and the heterogeneous nature of both the sepsis and control patients, the authors said. They further emphasized that malnutrition should be recognized for its possible effects on host immunity and that for a number of reasons, their study "must be viewed cautiously." The research "serves as a bridge between preclinical and early clinical findings," they wrote.
This study was supported by the National Institutes of Health. One of Dr. Boomer’s associates reported receiving grants from Pfizer, Bristol-Meyers Squibb, and Aurigine.
FROM JAMA
Major Finding: Compared with spleen and lung cells from control samples, those from sepsis patients showed profound cytokine production impairment, indicating immunosuppression. It may be possible to identify sepsis patients who survive into this later, immunocompromised phase and treat them with immune-enhancing therapies.
Data Source: Postmortem examination of immune function in spleen and lung tissue from 40 patients who died of sepsis, and from 29 controls who had critical but nonseptic illness.
Disclosures: This study was supported by the National Institutes of Health. One of Dr. Boomer’s associates reported receiving grants from Pfizer, Bristol-Meyers Squibb, and Aurigine.
Prior Hypertension Treatment Raises Long-Term Life Expectancy
Older patients with isolated systolic hypertension who were treated with chlorthalidone as part of a clinical trial in the late 1980s showed a significant gain in life expectancy 22 years later, according to a report in the Dec. 21 issue of JAMA.
"The gain in life expectancy free from cardiovascular death corresponds with approximately 1 day gained for each month of treatment," said Dr. John B. Kostis of the Cardiovascular Institute at the Robert Wood Johnson Medical School, New Brunswick, N.J., and his associates.
Getting this message out to patients and clinicians "may result in increased patient adherence to drug therapy, and decrease the degree of therapeutic inertia by health care providers," the investigators noted.
Although antihypertensive drug therapy is known to decrease cardiovascular events, there have been no long-term data on a possible gain in life expectancy. To assess such long-term outcomes, Dr. Kostis and his colleagues obtained mortality data on subjects who had participated in SHEP (Systolic Hypertension in the Elderly Program), a randomized, placebo-controlled trial that began in 1984 and concluded in 1990.
"The gain in life expectancy free from cardiovascular death corresponds with approximately 1 day gained for each month of [chlorthalidone] treatment."
In the trial, men and women aged 60 years and older (mean age, 72 years) at baseline who had isolated systolic hypertension were treated for a mean of 4.5 years with either stepped-care chlorthalidone (2,365 subjects) or placebo (2,371 subjects). At the end of the study, active drug was found to prevent one out of two admissions for heart failure, one out of three fatal or nonfatal strokes, and one out of four coronary heart disease events.
However, the effects on all-cause mortality and cardiovascular mortality were not significant at that time.
At the conclusion of the SHEP study, all the subjects were advised to continue taking active therapy.
Dr. Kostis and his associates assessed mortality in these study subjects by matching their personal identifiers to the National Death Index through the end of 2006. At that time, about 60% of both the active therapy group and the placebo group had died.
Life expectancy gain was greater for the study subjects who had received chlorthalidone than for those who had received placebo. The gain was 158 days for cardiovascular death and 105 days for all-cause mortality.
These gains were even greater in the subgroup of patients who achieved their target systolic blood pressure level while taking chlorthalidone.
For the study population as a whole, each month of antihypertensive therapy was associated with a 1-day extension of life expectancy free from cardiovascular death, the researchers said (JAMA 2011;306:2588-93).
It is important to note that newer antihypertensive agents that have been developed since the SHEP trial concluded "may be equally or more effective in decreasing cardiovascular events or may have a better adverse event profile," they noted.
This study was supported in part by the National Heart, Lung, and Blood Institute; the National Institute on Aging; and the Robert Wood Johnson Foundation. One of Dr. Kostis’s associates reported ties to Amgen and Merck; no other financial conflicts of interest were reported.
Older patients with isolated systolic hypertension who were treated with chlorthalidone as part of a clinical trial in the late 1980s showed a significant gain in life expectancy 22 years later, according to a report in the Dec. 21 issue of JAMA.
"The gain in life expectancy free from cardiovascular death corresponds with approximately 1 day gained for each month of treatment," said Dr. John B. Kostis of the Cardiovascular Institute at the Robert Wood Johnson Medical School, New Brunswick, N.J., and his associates.
Getting this message out to patients and clinicians "may result in increased patient adherence to drug therapy, and decrease the degree of therapeutic inertia by health care providers," the investigators noted.
Although antihypertensive drug therapy is known to decrease cardiovascular events, there have been no long-term data on a possible gain in life expectancy. To assess such long-term outcomes, Dr. Kostis and his colleagues obtained mortality data on subjects who had participated in SHEP (Systolic Hypertension in the Elderly Program), a randomized, placebo-controlled trial that began in 1984 and concluded in 1990.
"The gain in life expectancy free from cardiovascular death corresponds with approximately 1 day gained for each month of [chlorthalidone] treatment."
In the trial, men and women aged 60 years and older (mean age, 72 years) at baseline who had isolated systolic hypertension were treated for a mean of 4.5 years with either stepped-care chlorthalidone (2,365 subjects) or placebo (2,371 subjects). At the end of the study, active drug was found to prevent one out of two admissions for heart failure, one out of three fatal or nonfatal strokes, and one out of four coronary heart disease events.
However, the effects on all-cause mortality and cardiovascular mortality were not significant at that time.
At the conclusion of the SHEP study, all the subjects were advised to continue taking active therapy.
Dr. Kostis and his associates assessed mortality in these study subjects by matching their personal identifiers to the National Death Index through the end of 2006. At that time, about 60% of both the active therapy group and the placebo group had died.
Life expectancy gain was greater for the study subjects who had received chlorthalidone than for those who had received placebo. The gain was 158 days for cardiovascular death and 105 days for all-cause mortality.
These gains were even greater in the subgroup of patients who achieved their target systolic blood pressure level while taking chlorthalidone.
For the study population as a whole, each month of antihypertensive therapy was associated with a 1-day extension of life expectancy free from cardiovascular death, the researchers said (JAMA 2011;306:2588-93).
It is important to note that newer antihypertensive agents that have been developed since the SHEP trial concluded "may be equally or more effective in decreasing cardiovascular events or may have a better adverse event profile," they noted.
This study was supported in part by the National Heart, Lung, and Blood Institute; the National Institute on Aging; and the Robert Wood Johnson Foundation. One of Dr. Kostis’s associates reported ties to Amgen and Merck; no other financial conflicts of interest were reported.
Older patients with isolated systolic hypertension who were treated with chlorthalidone as part of a clinical trial in the late 1980s showed a significant gain in life expectancy 22 years later, according to a report in the Dec. 21 issue of JAMA.
"The gain in life expectancy free from cardiovascular death corresponds with approximately 1 day gained for each month of treatment," said Dr. John B. Kostis of the Cardiovascular Institute at the Robert Wood Johnson Medical School, New Brunswick, N.J., and his associates.
Getting this message out to patients and clinicians "may result in increased patient adherence to drug therapy, and decrease the degree of therapeutic inertia by health care providers," the investigators noted.
Although antihypertensive drug therapy is known to decrease cardiovascular events, there have been no long-term data on a possible gain in life expectancy. To assess such long-term outcomes, Dr. Kostis and his colleagues obtained mortality data on subjects who had participated in SHEP (Systolic Hypertension in the Elderly Program), a randomized, placebo-controlled trial that began in 1984 and concluded in 1990.
"The gain in life expectancy free from cardiovascular death corresponds with approximately 1 day gained for each month of [chlorthalidone] treatment."
In the trial, men and women aged 60 years and older (mean age, 72 years) at baseline who had isolated systolic hypertension were treated for a mean of 4.5 years with either stepped-care chlorthalidone (2,365 subjects) or placebo (2,371 subjects). At the end of the study, active drug was found to prevent one out of two admissions for heart failure, one out of three fatal or nonfatal strokes, and one out of four coronary heart disease events.
However, the effects on all-cause mortality and cardiovascular mortality were not significant at that time.
At the conclusion of the SHEP study, all the subjects were advised to continue taking active therapy.
Dr. Kostis and his associates assessed mortality in these study subjects by matching their personal identifiers to the National Death Index through the end of 2006. At that time, about 60% of both the active therapy group and the placebo group had died.
Life expectancy gain was greater for the study subjects who had received chlorthalidone than for those who had received placebo. The gain was 158 days for cardiovascular death and 105 days for all-cause mortality.
These gains were even greater in the subgroup of patients who achieved their target systolic blood pressure level while taking chlorthalidone.
For the study population as a whole, each month of antihypertensive therapy was associated with a 1-day extension of life expectancy free from cardiovascular death, the researchers said (JAMA 2011;306:2588-93).
It is important to note that newer antihypertensive agents that have been developed since the SHEP trial concluded "may be equally or more effective in decreasing cardiovascular events or may have a better adverse event profile," they noted.
This study was supported in part by the National Heart, Lung, and Blood Institute; the National Institute on Aging; and the Robert Wood Johnson Foundation. One of Dr. Kostis’s associates reported ties to Amgen and Merck; no other financial conflicts of interest were reported.
FROM JAMA
Major Finding: Older patients who received chlorthalidone for isolated systolic hypertension had an extended life expectancy, compared with those who received placebo, with a gain of 158 days free of cardiovascular death and 105 days free of all-cause mortality.
Data Source: A 22-year follow-up of mortality among 4,736 subjects who participated in SHEP (Systolic Hypertension in the Elderly Program), a randomized, controlled trial of chlorthalidone therapy for isolated systolic hypertension in the late 1980s.
Disclosures: This study was supported in part by the National Heart, Lung, and Blood Institute; the National Institute on Aging; and the Robert Wood Johnson Foundation. One of Dr. Kostis’s associates reported ties to Amgen and Merck; no other financial conflicts of interest were reported.
CPAP Improves Metabolic Syndrome in Apnea Patients
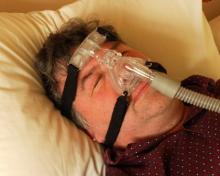
Continuous positive airway pressure therapy improved several components of the metabolic syndrome along with obstructive sleep apnea in patients who had both disorders, according to a report in the Dec. 15 issue of the New England Journal of Medicine.
In most cases, only one component of the metabolic syndrome improved significantly after CPAP, but that improvement was significant enough to "reverse" the syndrome, said Dr. Surendra K. Sharma of All India Institute of Medical Sciences, New Delhi, and his associates.
No particular component stood out as being the most responsive to CPAP; statistically significant improvements were seen in systolic BP, diastolic BP, total cholesterol, non-HDL cholesterol, LDL cholesterol, triglycerides, glycated hemoglobin, weight, and visceral and subcutaneous fat. "These results suggest a significant clinical benefit that will lead to a reduction in cardiovascular risk," they noted.
To examine the effect of CPAP on components of the metabolic syndrome, the researchers recruited 86 patients aged 30-65 years from the sleep laboratory at the institute who had obstructive sleep apnea that was moderate or worse in severity. All the subjects reported excessive daytime somnolence.
A total of 75 study subjects (87%) had the metabolic syndrome, and the remainder had some of the components of the metabolic syndrome.
These patients were randomly assigned to undergo either CPAP or sham CPAP for 3 months, followed by a washout period of 1 month. They then crossed over to receive the other intervention for 3 months.
The sham CPAP was not discernible to the study subjects or the investigators.
The metabolic syndrome resolved in 14 (20%) of the study subjects after CPAP. This was due to decreased blood pressure in five; decreased fasting blood glucose in two; decreased triglycerides in two; increased HDL cholesterol in three; improved triglycerides plus HDL cholesterol in one; and improved triglycerides, HDL cholesterol, and fasting blood glucose in one, Dr. Sharma and his colleagues said. Symptoms of the syndrome developed in three patients who did not have metabolic syndrome at the start of the study.
Overall, CPAP was associated with a mean decrease in systolic BP of 3.9 mm Hg, a mean decrease in diastolic BP of 2.5 mm Hg, a mean decrease in total cholesterol of 13.3 mg/dL, and a mean decrease in triglycerides of 18.7 mg/dL.
CT scans revealed a significant decrease in both visceral and subcutaneous fat, which was accompanied by a decrease in BMI, with CPAP therapy. "These findings could be secondary to a decrease in daytime somnolence and a consequent increase in physical activity after CPAP use at night."
In addition, "we speculate that CPAP has a favorable effect on leptin levels, which have been shown to be elevated in patients with obstructive sleep apnea and to normalize with CPAP therapy," the investigators said (N. Engl. J. Med. 2011;365:2277-86).
In a subgroup analysis involving only the 51 subjects who were most compliant with CPAP, with a mean use of at least 5 hours every night, the improvements in components of the metabolic syndrome were even greater. In particular, systolic BP decreased by 5.6 mm Hg and diastolic BP decreased by 3.3 mm Hg.
This subgroup of patients also showed significant improvement in carotid intima-media thickness, "suggesting a potential role for CPAP therapy in reversing endothelial damage due to obstructive sleep apnea and the metabolic syndrome," Dr. Sharma and his associates said.
Two patients could not tolerate CPAP and one could not tolerate sham CPAP within the first month of treatment, and they withdrew from the study. "Other adverse events reported included skin irritation (in 51% of all patients), nasal bridge discomfort (in 44%), nasal congestion (in 28%), headache (in 26%), and mask leaks (in 30%)."
This study was funded by Pfizer. All investigators reported having no financial conflicts of interest. The investigators received technical support from ResMed Corp. in designing a sham CPAP machine.
Continuous positive airway pressure therapy improved several components of the metabolic syndrome along with obstructive sleep apnea in patients who had both disorders, according to a report in the Dec. 15 issue of the New England Journal of Medicine.
In most cases, only one component of the metabolic syndrome improved significantly after CPAP, but that improvement was significant enough to "reverse" the syndrome, said Dr. Surendra K. Sharma of All India Institute of Medical Sciences, New Delhi, and his associates.
No particular component stood out as being the most responsive to CPAP; statistically significant improvements were seen in systolic BP, diastolic BP, total cholesterol, non-HDL cholesterol, LDL cholesterol, triglycerides, glycated hemoglobin, weight, and visceral and subcutaneous fat. "These results suggest a significant clinical benefit that will lead to a reduction in cardiovascular risk," they noted.
To examine the effect of CPAP on components of the metabolic syndrome, the researchers recruited 86 patients aged 30-65 years from the sleep laboratory at the institute who had obstructive sleep apnea that was moderate or worse in severity. All the subjects reported excessive daytime somnolence.
A total of 75 study subjects (87%) had the metabolic syndrome, and the remainder had some of the components of the metabolic syndrome.
These patients were randomly assigned to undergo either CPAP or sham CPAP for 3 months, followed by a washout period of 1 month. They then crossed over to receive the other intervention for 3 months.
The sham CPAP was not discernible to the study subjects or the investigators.
The metabolic syndrome resolved in 14 (20%) of the study subjects after CPAP. This was due to decreased blood pressure in five; decreased fasting blood glucose in two; decreased triglycerides in two; increased HDL cholesterol in three; improved triglycerides plus HDL cholesterol in one; and improved triglycerides, HDL cholesterol, and fasting blood glucose in one, Dr. Sharma and his colleagues said. Symptoms of the syndrome developed in three patients who did not have metabolic syndrome at the start of the study.
Overall, CPAP was associated with a mean decrease in systolic BP of 3.9 mm Hg, a mean decrease in diastolic BP of 2.5 mm Hg, a mean decrease in total cholesterol of 13.3 mg/dL, and a mean decrease in triglycerides of 18.7 mg/dL.
CT scans revealed a significant decrease in both visceral and subcutaneous fat, which was accompanied by a decrease in BMI, with CPAP therapy. "These findings could be secondary to a decrease in daytime somnolence and a consequent increase in physical activity after CPAP use at night."
In addition, "we speculate that CPAP has a favorable effect on leptin levels, which have been shown to be elevated in patients with obstructive sleep apnea and to normalize with CPAP therapy," the investigators said (N. Engl. J. Med. 2011;365:2277-86).
In a subgroup analysis involving only the 51 subjects who were most compliant with CPAP, with a mean use of at least 5 hours every night, the improvements in components of the metabolic syndrome were even greater. In particular, systolic BP decreased by 5.6 mm Hg and diastolic BP decreased by 3.3 mm Hg.
This subgroup of patients also showed significant improvement in carotid intima-media thickness, "suggesting a potential role for CPAP therapy in reversing endothelial damage due to obstructive sleep apnea and the metabolic syndrome," Dr. Sharma and his associates said.
Two patients could not tolerate CPAP and one could not tolerate sham CPAP within the first month of treatment, and they withdrew from the study. "Other adverse events reported included skin irritation (in 51% of all patients), nasal bridge discomfort (in 44%), nasal congestion (in 28%), headache (in 26%), and mask leaks (in 30%)."
This study was funded by Pfizer. All investigators reported having no financial conflicts of interest. The investigators received technical support from ResMed Corp. in designing a sham CPAP machine.
Continuous positive airway pressure therapy improved several components of the metabolic syndrome along with obstructive sleep apnea in patients who had both disorders, according to a report in the Dec. 15 issue of the New England Journal of Medicine.
In most cases, only one component of the metabolic syndrome improved significantly after CPAP, but that improvement was significant enough to "reverse" the syndrome, said Dr. Surendra K. Sharma of All India Institute of Medical Sciences, New Delhi, and his associates.
No particular component stood out as being the most responsive to CPAP; statistically significant improvements were seen in systolic BP, diastolic BP, total cholesterol, non-HDL cholesterol, LDL cholesterol, triglycerides, glycated hemoglobin, weight, and visceral and subcutaneous fat. "These results suggest a significant clinical benefit that will lead to a reduction in cardiovascular risk," they noted.
To examine the effect of CPAP on components of the metabolic syndrome, the researchers recruited 86 patients aged 30-65 years from the sleep laboratory at the institute who had obstructive sleep apnea that was moderate or worse in severity. All the subjects reported excessive daytime somnolence.
A total of 75 study subjects (87%) had the metabolic syndrome, and the remainder had some of the components of the metabolic syndrome.
These patients were randomly assigned to undergo either CPAP or sham CPAP for 3 months, followed by a washout period of 1 month. They then crossed over to receive the other intervention for 3 months.
The sham CPAP was not discernible to the study subjects or the investigators.
The metabolic syndrome resolved in 14 (20%) of the study subjects after CPAP. This was due to decreased blood pressure in five; decreased fasting blood glucose in two; decreased triglycerides in two; increased HDL cholesterol in three; improved triglycerides plus HDL cholesterol in one; and improved triglycerides, HDL cholesterol, and fasting blood glucose in one, Dr. Sharma and his colleagues said. Symptoms of the syndrome developed in three patients who did not have metabolic syndrome at the start of the study.
Overall, CPAP was associated with a mean decrease in systolic BP of 3.9 mm Hg, a mean decrease in diastolic BP of 2.5 mm Hg, a mean decrease in total cholesterol of 13.3 mg/dL, and a mean decrease in triglycerides of 18.7 mg/dL.
CT scans revealed a significant decrease in both visceral and subcutaneous fat, which was accompanied by a decrease in BMI, with CPAP therapy. "These findings could be secondary to a decrease in daytime somnolence and a consequent increase in physical activity after CPAP use at night."
In addition, "we speculate that CPAP has a favorable effect on leptin levels, which have been shown to be elevated in patients with obstructive sleep apnea and to normalize with CPAP therapy," the investigators said (N. Engl. J. Med. 2011;365:2277-86).
In a subgroup analysis involving only the 51 subjects who were most compliant with CPAP, with a mean use of at least 5 hours every night, the improvements in components of the metabolic syndrome were even greater. In particular, systolic BP decreased by 5.6 mm Hg and diastolic BP decreased by 3.3 mm Hg.
This subgroup of patients also showed significant improvement in carotid intima-media thickness, "suggesting a potential role for CPAP therapy in reversing endothelial damage due to obstructive sleep apnea and the metabolic syndrome," Dr. Sharma and his associates said.
Two patients could not tolerate CPAP and one could not tolerate sham CPAP within the first month of treatment, and they withdrew from the study. "Other adverse events reported included skin irritation (in 51% of all patients), nasal bridge discomfort (in 44%), nasal congestion (in 28%), headache (in 26%), and mask leaks (in 30%)."
This study was funded by Pfizer. All investigators reported having no financial conflicts of interest. The investigators received technical support from ResMed Corp. in designing a sham CPAP machine.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The metabolic syndrome resolved in 11 of 86 patients after CPAP therapy, compared with 1 of those same patients after sham therapy. The treatment also significantly improved systolic and diastolic BP; total, LDL, and non-HDL cholesterol; triglycerides; glycated hemoglobin; weight; and visceral and subcutaneous fat.
Data Source: A double-blind, randomized trial involving 86 patients with moderate to severe obstructive sleep apnea and components of the metabolic syndrome who received 3 months of real and 3 months of sham CPAP therapy.
Disclosures: This study was funded by Pfizer. All investigators reported having no financial conflicts of interest. The investigators received technical support from ResMed Corp. in designing a sham CPAP machine.
Readmission Rates Tied to Regions' Propensity to Hospitalize
Regional rates of hospital readmission within 30 days are strongly correlated with the overall tendency to hospitalize patients for any reason in that region, according to a report in the Dec. 15 issue of the New England Journal of Medicine.
High rates of 30-day hospital readmission are seen as an indication that patients received poor transitional care at discharge and immediately thereafter. But rather than reflecting inadequate care, high readmission rates may simply reflect the customary tendency to hospitalize patients in any given geographic area, said Dr. Arnold M. Epstein of the department of health policy and management at the Harvard School of Public Health, Boston, and his associates.
The investigators examined the factors that correlated with readmission rates using information from a Medicare database on patients aged 65 years and older who were discharged with a diagnosis of heart failure or pneumonia during a recent 6-month period. The subjects were seen at 306 "hospital referral regions" (HRRs) across the country.
Data on hospitals’ discharge practices were obtained from a separate survey of different patients who were asked about their experiences at discharge from hospitals in the same HRRs. In addition, the probability that each study subject would require readmission was estimated based on more than 32 clinical and demographic factors listed in their medical records.
The study included 234,477 discharges with heart failure from 4,432 hospitals and 237,025 discharges with pneumonia from 4,497 hospitals in the 306 HRRs.
Readmission rates varied considerably among the hospitals studied. The single factor that explained most of this variation was not case mix, quality of discharge planning, patient traits, or hospital characteristics; rather, it was the overall rate of hospitalizations in the region, the investigators said (N. Engl. J. Med. 2011;365:2287-95).
"Although most interventions designed to reduce readmissions thus far have focused on better disease management and the coordination of care, our results underscore the importance of policy efforts directed at reducing the general incentives to use hospital services," they noted.
Regional patterns of care rather than patient traits or hospital characteristics may be the key to reducing readmissions. "We estimate that each year there are approximately 115,568 readmissions within 30 days after discharge among patients initially hospitalized for [heart failure] and 84,854 readmissions among those initially hospitalized for pneumonia.
"We found that if all-cause admission rates for the HRRs in the upper quintiles of hospital utilization were reduced to the rate in the lowest quintile, the readmission rate for [heart failure] would be reduced from 24.6% to 21.2%, eliminating approximately 16,166 (14.0%) of the readmissions for Medicare beneficiaries. For pneumonia, the readmission rate would be reduced from 17.9% to 15.5%, eliminating 11,434 (13.5%) of the readmissions," Dr. Epstein and his associates said.
This study was supported by the Commonwealth Fund. One of Dr. Epstein’s associates reported receiving a grant from the Robert Wood Johnson Foundation. No financial conflicts of interest were reported.
Regional rates of hospital readmission within 30 days are strongly correlated with the overall tendency to hospitalize patients for any reason in that region, according to a report in the Dec. 15 issue of the New England Journal of Medicine.
High rates of 30-day hospital readmission are seen as an indication that patients received poor transitional care at discharge and immediately thereafter. But rather than reflecting inadequate care, high readmission rates may simply reflect the customary tendency to hospitalize patients in any given geographic area, said Dr. Arnold M. Epstein of the department of health policy and management at the Harvard School of Public Health, Boston, and his associates.
The investigators examined the factors that correlated with readmission rates using information from a Medicare database on patients aged 65 years and older who were discharged with a diagnosis of heart failure or pneumonia during a recent 6-month period. The subjects were seen at 306 "hospital referral regions" (HRRs) across the country.
Data on hospitals’ discharge practices were obtained from a separate survey of different patients who were asked about their experiences at discharge from hospitals in the same HRRs. In addition, the probability that each study subject would require readmission was estimated based on more than 32 clinical and demographic factors listed in their medical records.
The study included 234,477 discharges with heart failure from 4,432 hospitals and 237,025 discharges with pneumonia from 4,497 hospitals in the 306 HRRs.
Readmission rates varied considerably among the hospitals studied. The single factor that explained most of this variation was not case mix, quality of discharge planning, patient traits, or hospital characteristics; rather, it was the overall rate of hospitalizations in the region, the investigators said (N. Engl. J. Med. 2011;365:2287-95).
"Although most interventions designed to reduce readmissions thus far have focused on better disease management and the coordination of care, our results underscore the importance of policy efforts directed at reducing the general incentives to use hospital services," they noted.
Regional patterns of care rather than patient traits or hospital characteristics may be the key to reducing readmissions. "We estimate that each year there are approximately 115,568 readmissions within 30 days after discharge among patients initially hospitalized for [heart failure] and 84,854 readmissions among those initially hospitalized for pneumonia.
"We found that if all-cause admission rates for the HRRs in the upper quintiles of hospital utilization were reduced to the rate in the lowest quintile, the readmission rate for [heart failure] would be reduced from 24.6% to 21.2%, eliminating approximately 16,166 (14.0%) of the readmissions for Medicare beneficiaries. For pneumonia, the readmission rate would be reduced from 17.9% to 15.5%, eliminating 11,434 (13.5%) of the readmissions," Dr. Epstein and his associates said.
This study was supported by the Commonwealth Fund. One of Dr. Epstein’s associates reported receiving a grant from the Robert Wood Johnson Foundation. No financial conflicts of interest were reported.
Regional rates of hospital readmission within 30 days are strongly correlated with the overall tendency to hospitalize patients for any reason in that region, according to a report in the Dec. 15 issue of the New England Journal of Medicine.
High rates of 30-day hospital readmission are seen as an indication that patients received poor transitional care at discharge and immediately thereafter. But rather than reflecting inadequate care, high readmission rates may simply reflect the customary tendency to hospitalize patients in any given geographic area, said Dr. Arnold M. Epstein of the department of health policy and management at the Harvard School of Public Health, Boston, and his associates.
The investigators examined the factors that correlated with readmission rates using information from a Medicare database on patients aged 65 years and older who were discharged with a diagnosis of heart failure or pneumonia during a recent 6-month period. The subjects were seen at 306 "hospital referral regions" (HRRs) across the country.
Data on hospitals’ discharge practices were obtained from a separate survey of different patients who were asked about their experiences at discharge from hospitals in the same HRRs. In addition, the probability that each study subject would require readmission was estimated based on more than 32 clinical and demographic factors listed in their medical records.
The study included 234,477 discharges with heart failure from 4,432 hospitals and 237,025 discharges with pneumonia from 4,497 hospitals in the 306 HRRs.
Readmission rates varied considerably among the hospitals studied. The single factor that explained most of this variation was not case mix, quality of discharge planning, patient traits, or hospital characteristics; rather, it was the overall rate of hospitalizations in the region, the investigators said (N. Engl. J. Med. 2011;365:2287-95).
"Although most interventions designed to reduce readmissions thus far have focused on better disease management and the coordination of care, our results underscore the importance of policy efforts directed at reducing the general incentives to use hospital services," they noted.
Regional patterns of care rather than patient traits or hospital characteristics may be the key to reducing readmissions. "We estimate that each year there are approximately 115,568 readmissions within 30 days after discharge among patients initially hospitalized for [heart failure] and 84,854 readmissions among those initially hospitalized for pneumonia.
"We found that if all-cause admission rates for the HRRs in the upper quintiles of hospital utilization were reduced to the rate in the lowest quintile, the readmission rate for [heart failure] would be reduced from 24.6% to 21.2%, eliminating approximately 16,166 (14.0%) of the readmissions for Medicare beneficiaries. For pneumonia, the readmission rate would be reduced from 17.9% to 15.5%, eliminating 11,434 (13.5%) of the readmissions," Dr. Epstein and his associates said.
This study was supported by the Commonwealth Fund. One of Dr. Epstein’s associates reported receiving a grant from the Robert Wood Johnson Foundation. No financial conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: The overall rate of hospitalization in a given geographic region, rather than patient or hospital factors, is the strongest predictor of readmission within 30 days.
Data Source: An analysis of national Medicare and other data regarding readmissions among 234,477 cases of CHF discharged from 4,432 hospitals and 237,025 cases of pneumonia discharged from 4,497 hospitals during a 6-month period.
Disclosures: This study was supported by the Commonwealth Fund. One of Dr. Epstein’s associates reported receiving a grant from the Robert Wood Johnson Foundation. No financial conflicts of interest were reported.
Despite Guidelines, Elderly Are Getting Cancer Tests
Most Americans aged 75 years and older continue to undergo routine screening for breast, cervical, colorectal, or prostate cancer even though recommendations advise against it, according to a report in the Dec. 12/26 issue of Archives of Internal Medicine.
Approximately half of these older adults report that their physicians recommended the cancer screening.
"These persistently elevated rates of screening raise the question of whether the decision to be screened is being made without fully knowing or discussing the risks and benefits," said Keith M. Bellizzi, Ph.D., of the department of human development and family studies, University of Connecticut, Storrs, and his associates.
According to current U.S. Preventive Services Task Force guidelines, routine screening for breast, colorectal, and prostate cancer are not advised once patients reach age 75 years, and routine screening for cervical cancer is not advised once patients reach age 65 years. "There is general agreement that screening decisions should be individualized" in this age group, based on each patient's functional status, comorbidities, life expectancy, and personal preferences.
Dr. Bellizzi and his colleagues examined real-world cancer screening practices using data from the National Health Interview Survey, an annual canvass of 30,000 households across the country that is used to monitor trends in illness. They combined data from the 2005 and 2008 surveys, resulting in a sample of 49,575 adults. A total of 1,697 of these subjects were aged 75-79 years and another 2,376 were 80 years and older.
Sixty-two percent of women aged 75-79 years and 50% of those aged 80 years and older reported that they had recently undergone mammography. Those proportions are nearly as high as the 74% rate seen in the target population for mammographic screening, women aged 50-74 years, the investigators said (Arch. Intern. Med. 2011;171:2031-7).
Similarly, 53% of women aged 75-79 years and 38% of those aged 80 years and older reported having been screened for cervical cancer.
The rate of colorectal cancer screening was actually highest (57%) in men and women aged 75-79 years, and lower (48%) in the younger age groups specified by the guidelines as the target population. The rate of colorectal cancer screening was almost as high among subjects aged 80 years and older (47%) as it was in the target population.
Similarly, the rate of PSA screening for prostate cancer was highest in the 75- to 79-year-old group (57%), followed by the oldest group (42%), and was lower in the target population of men aged 50-74 years (40%).
Patients also were asked whether their physicians had recommended cancer screening. Such a recommendation was the strongest predictor of obtaining the screening.
As many as 62% of women aged 75-79 years said that their physicians had advised them to get mammograms; 48% of that population reported that they were advised to be screened for cervical cancer. As many as 65% of older men and women said their physicians had recommended colonoscopy, including 54% of those aged 80 years and older. And as many as 62% of older men said their physicians had advised them to get PSA testing.
"This finding reinforces the critical role for health care providers to make informed screening decisions for older adults," Dr. Bellizzi and his associates said.
This study was supported by the National Cancer Institute. No financial conflicts of interest were reported.
Dr. Walter’s work is supported by the National Cancer Institute and the San Francisco Veterans Affairs Medical Center. She reported no financial conflicts of interest.
It can be argued that at least some of the screening rates in this study were not too high. For example, 56% of U.S. women aged 75-79 years are expected to live 10 years or more, so cancer screening rates of about 60% might not be unreasonable in this age group, said Dr. Louise C. Walter.
Age alone may not be as important as clinical factors, such as functional status, in judging the appropriateness of cancer screening. Such screening may be appropriate in a healthy 80-year-old man but inappropriate in a frail 80-year-old man with serious comorbidities, she noted.
"While arguments persist about what is the ‘right’ rate of cancer screening in older persons, it seems clear that the rate of informed decision-making should approach 100%," Dr. Walter added.
Dr. Walter is in the division of geriatrics at the University of California, San Francisco. Her work is supported by the National Cancer Institute and the San Francisco VA Medical Center. She reported no financial conflicts of interest. These remarks were taken from her invited commentary accompanying Dr. Bellizzi’s report (Arch. Intern. Med. 2011;171:2037-8).
It can be argued that at least some of the screening rates in this study were not too high. For example, 56% of U.S. women aged 75-79 years are expected to live 10 years or more, so cancer screening rates of about 60% might not be unreasonable in this age group, said Dr. Louise C. Walter.
Age alone may not be as important as clinical factors, such as functional status, in judging the appropriateness of cancer screening. Such screening may be appropriate in a healthy 80-year-old man but inappropriate in a frail 80-year-old man with serious comorbidities, she noted.
"While arguments persist about what is the ‘right’ rate of cancer screening in older persons, it seems clear that the rate of informed decision-making should approach 100%," Dr. Walter added.
Dr. Walter is in the division of geriatrics at the University of California, San Francisco. Her work is supported by the National Cancer Institute and the San Francisco VA Medical Center. She reported no financial conflicts of interest. These remarks were taken from her invited commentary accompanying Dr. Bellizzi’s report (Arch. Intern. Med. 2011;171:2037-8).
It can be argued that at least some of the screening rates in this study were not too high. For example, 56% of U.S. women aged 75-79 years are expected to live 10 years or more, so cancer screening rates of about 60% might not be unreasonable in this age group, said Dr. Louise C. Walter.
Age alone may not be as important as clinical factors, such as functional status, in judging the appropriateness of cancer screening. Such screening may be appropriate in a healthy 80-year-old man but inappropriate in a frail 80-year-old man with serious comorbidities, she noted.
"While arguments persist about what is the ‘right’ rate of cancer screening in older persons, it seems clear that the rate of informed decision-making should approach 100%," Dr. Walter added.
Dr. Walter is in the division of geriatrics at the University of California, San Francisco. Her work is supported by the National Cancer Institute and the San Francisco VA Medical Center. She reported no financial conflicts of interest. These remarks were taken from her invited commentary accompanying Dr. Bellizzi’s report (Arch. Intern. Med. 2011;171:2037-8).
Most Americans aged 75 years and older continue to undergo routine screening for breast, cervical, colorectal, or prostate cancer even though recommendations advise against it, according to a report in the Dec. 12/26 issue of Archives of Internal Medicine.
Approximately half of these older adults report that their physicians recommended the cancer screening.
"These persistently elevated rates of screening raise the question of whether the decision to be screened is being made without fully knowing or discussing the risks and benefits," said Keith M. Bellizzi, Ph.D., of the department of human development and family studies, University of Connecticut, Storrs, and his associates.
According to current U.S. Preventive Services Task Force guidelines, routine screening for breast, colorectal, and prostate cancer are not advised once patients reach age 75 years, and routine screening for cervical cancer is not advised once patients reach age 65 years. "There is general agreement that screening decisions should be individualized" in this age group, based on each patient's functional status, comorbidities, life expectancy, and personal preferences.
Dr. Bellizzi and his colleagues examined real-world cancer screening practices using data from the National Health Interview Survey, an annual canvass of 30,000 households across the country that is used to monitor trends in illness. They combined data from the 2005 and 2008 surveys, resulting in a sample of 49,575 adults. A total of 1,697 of these subjects were aged 75-79 years and another 2,376 were 80 years and older.
Sixty-two percent of women aged 75-79 years and 50% of those aged 80 years and older reported that they had recently undergone mammography. Those proportions are nearly as high as the 74% rate seen in the target population for mammographic screening, women aged 50-74 years, the investigators said (Arch. Intern. Med. 2011;171:2031-7).
Similarly, 53% of women aged 75-79 years and 38% of those aged 80 years and older reported having been screened for cervical cancer.
The rate of colorectal cancer screening was actually highest (57%) in men and women aged 75-79 years, and lower (48%) in the younger age groups specified by the guidelines as the target population. The rate of colorectal cancer screening was almost as high among subjects aged 80 years and older (47%) as it was in the target population.
Similarly, the rate of PSA screening for prostate cancer was highest in the 75- to 79-year-old group (57%), followed by the oldest group (42%), and was lower in the target population of men aged 50-74 years (40%).
Patients also were asked whether their physicians had recommended cancer screening. Such a recommendation was the strongest predictor of obtaining the screening.
As many as 62% of women aged 75-79 years said that their physicians had advised them to get mammograms; 48% of that population reported that they were advised to be screened for cervical cancer. As many as 65% of older men and women said their physicians had recommended colonoscopy, including 54% of those aged 80 years and older. And as many as 62% of older men said their physicians had advised them to get PSA testing.
"This finding reinforces the critical role for health care providers to make informed screening decisions for older adults," Dr. Bellizzi and his associates said.
This study was supported by the National Cancer Institute. No financial conflicts of interest were reported.
Dr. Walter’s work is supported by the National Cancer Institute and the San Francisco Veterans Affairs Medical Center. She reported no financial conflicts of interest.
Most Americans aged 75 years and older continue to undergo routine screening for breast, cervical, colorectal, or prostate cancer even though recommendations advise against it, according to a report in the Dec. 12/26 issue of Archives of Internal Medicine.
Approximately half of these older adults report that their physicians recommended the cancer screening.
"These persistently elevated rates of screening raise the question of whether the decision to be screened is being made without fully knowing or discussing the risks and benefits," said Keith M. Bellizzi, Ph.D., of the department of human development and family studies, University of Connecticut, Storrs, and his associates.
According to current U.S. Preventive Services Task Force guidelines, routine screening for breast, colorectal, and prostate cancer are not advised once patients reach age 75 years, and routine screening for cervical cancer is not advised once patients reach age 65 years. "There is general agreement that screening decisions should be individualized" in this age group, based on each patient's functional status, comorbidities, life expectancy, and personal preferences.
Dr. Bellizzi and his colleagues examined real-world cancer screening practices using data from the National Health Interview Survey, an annual canvass of 30,000 households across the country that is used to monitor trends in illness. They combined data from the 2005 and 2008 surveys, resulting in a sample of 49,575 adults. A total of 1,697 of these subjects were aged 75-79 years and another 2,376 were 80 years and older.
Sixty-two percent of women aged 75-79 years and 50% of those aged 80 years and older reported that they had recently undergone mammography. Those proportions are nearly as high as the 74% rate seen in the target population for mammographic screening, women aged 50-74 years, the investigators said (Arch. Intern. Med. 2011;171:2031-7).
Similarly, 53% of women aged 75-79 years and 38% of those aged 80 years and older reported having been screened for cervical cancer.
The rate of colorectal cancer screening was actually highest (57%) in men and women aged 75-79 years, and lower (48%) in the younger age groups specified by the guidelines as the target population. The rate of colorectal cancer screening was almost as high among subjects aged 80 years and older (47%) as it was in the target population.
Similarly, the rate of PSA screening for prostate cancer was highest in the 75- to 79-year-old group (57%), followed by the oldest group (42%), and was lower in the target population of men aged 50-74 years (40%).
Patients also were asked whether their physicians had recommended cancer screening. Such a recommendation was the strongest predictor of obtaining the screening.
As many as 62% of women aged 75-79 years said that their physicians had advised them to get mammograms; 48% of that population reported that they were advised to be screened for cervical cancer. As many as 65% of older men and women said their physicians had recommended colonoscopy, including 54% of those aged 80 years and older. And as many as 62% of older men said their physicians had advised them to get PSA testing.
"This finding reinforces the critical role for health care providers to make informed screening decisions for older adults," Dr. Bellizzi and his associates said.
This study was supported by the National Cancer Institute. No financial conflicts of interest were reported.
Dr. Walter’s work is supported by the National Cancer Institute and the San Francisco Veterans Affairs Medical Center. She reported no financial conflicts of interest.
FROM ARCHIVES OF INTERNAL MEDICINE
Major Finding: Among study subjects aged 75-79 years, 62% were screened for breast cancer, 53% for cervical cancer, 57% for colorectal cancer, and 56% for prostate cancer; the corresponding rates for subjects aged 80 years and older were 50%, 38%, 47%, and 42%.
Data Source: The 2005 and 2008 National Health Interview Surveys.
Disclosures: This study was supported by the National Cancer Institute. No financial conflicts of interest were reported.
ADHD Medications Found Not to Raise Risk of Serious CVD Events
Medications for attention-deficit/hyperactivity disorder do not increase the risk of serious cardiovascular events among young and middle-aged adults, according to the results of a retrospective, population-based cohort study published online Dec. 12 in JAMA.
Stimulants such as methylphenidate, amphetamines, and pemoline, as well as the selective norepinephrine reuptake inhibitor atomoxetine, are known to raise heart rate and to increase systolic blood pressure by approximately 2-5 mm Hg and diastolic blood pressure by 1-3 mm Hg. "Although these effects would be expected to slightly increase risk for myocardial infarction, sudden cardiac death, and stroke, clinical trials have not been large enough to assess risk of these events," said Laurel A. Habel, Ph.D., of Kaiser Permanente Northern California, Oakland, and her associates.
Moreover, cardiovascular safety data from pharmacoepidemiologic studies have been "limited and inconsistent" to date, they said.
Dr. Habel and her colleagues examined the relationship between attention-deficit/hyperactivity disorder (ADHD) medications and cardiovascular risk in a large, geographically and sociodemographically diverse cohort of 443,198 adults aged 25-64 years. A total of 150,359 subjects used ADHD medications, including methylphenidate (45%), amphetamines (44%), atomoxetine, (8%), and pemoline (3%).
Medication use was assessed using pharmacy data on filled prescriptions. In addition, the medical records for all potential cases of MI, sudden cardiac death, and stroke were reviewed to verify those events.
There were 1,357 MIs, 296 sudden cardiac deaths, and 575 strokes in 806,182 person-years of follow-up.
The rates of all of these events combined were not significantly different between current users of ADHD medications and nonusers, with a multivariate adjusted rate ratio of 0.83. There also were no significant differences in these rates between current users and subjects who had used ADHD medications in the past but had stopped taking them (rate ratio, 1.03), the investigators said (JAMA 2011 Dec. 12 [doi:10.1001/jama.2011.1830]).
These results remained consistent when each medication was assessed individually, as well as when ischemic and hemorrhagic strokes were examined separately.
To control for potential differences in cardiovascular disease risk between exposed and nonexposed study subjects, the investigators assigned each subject a summary cardiovascular risk score at baseline. After the data were adjusted for these scores, the results were essentially the same: no increase in the risk of CVD events among ADHD medication users vs. nonusers, regardless of the subjects’ baseline CVD risk.
The data also were analyzed according to whether subjects were new to using ADHD medications or were long-term users, and again there was no difference in CVD risk. Rate ratios also remained consistent in all subgroup analyses, including an assessment of CVD risk by subject age.
Information on some important risk factors and on medication dosage was unavailable, so this study, despite its large size, could not completely rule out a "modestly" elevated risk with ADHD medications, the researchers noted.
This study was funded in part by the Agency for Healthcare Research and Quality, the Food and Drug Administration, and the National Institute on Aging. Dr. Habel reported ties to Merck, Takeda, and Sanofi-Aventis, and her associates reported ties to Optuminsight, Merck, GlaxoSmithKline, Novartis, and Abbott. Dr. Shaw reported receiving travel grants from Jannsen-Cilag, maker of Concerta (methylphenidate).
The methodology was rigorous in this study by Dr. Habel and her colleagues, by far the largest, most comprehensive study of the cardiovascular safety of ADHD medications done to date, said Dr. Philip Shaw.
One elegant feature of the study was the authors’ presentation of several worst-case scenarios in which it was assumed that the upper range of the risk estimates applied. "Even with these assumptions, the increase in absolute risk difference is small" between users and nonusers of ADHD medications, Dr. Shaw noted.
"Such absolute risk estimates allow clinicians to form their own opinion of the clinical importance of the worst-case scenario," he said. In addition, "no link was found between increasing risk and increasing duration of medication use, and the upper range of use was as high as 13.5 years," he said.
Therefore, this study is of value to clinicians when counseling patients on the risks and benefits of ADHD medications. "Now there is solid evidence – perhaps even some heartening news – that physicians can use to address concerns about cardiovascular risk," he concluded.
Dr. Shaw is at the National Human Genome Research Institute, Bethesda,
Md. He reported receiving travel grants from Jannsen-Cilag, maker of Concerta (methylphenidate). These remarks were taken from his editorial accompanying Dr. Habel’s report (JAMA 2011 Dec. 12 [doi:10.1001/jama.2011.1866]).
The methodology was rigorous in this study by Dr. Habel and her colleagues, by far the largest, most comprehensive study of the cardiovascular safety of ADHD medications done to date, said Dr. Philip Shaw.
One elegant feature of the study was the authors’ presentation of several worst-case scenarios in which it was assumed that the upper range of the risk estimates applied. "Even with these assumptions, the increase in absolute risk difference is small" between users and nonusers of ADHD medications, Dr. Shaw noted.
"Such absolute risk estimates allow clinicians to form their own opinion of the clinical importance of the worst-case scenario," he said. In addition, "no link was found between increasing risk and increasing duration of medication use, and the upper range of use was as high as 13.5 years," he said.
Therefore, this study is of value to clinicians when counseling patients on the risks and benefits of ADHD medications. "Now there is solid evidence – perhaps even some heartening news – that physicians can use to address concerns about cardiovascular risk," he concluded.
Dr. Shaw is at the National Human Genome Research Institute, Bethesda,
Md. He reported receiving travel grants from Jannsen-Cilag, maker of Concerta (methylphenidate). These remarks were taken from his editorial accompanying Dr. Habel’s report (JAMA 2011 Dec. 12 [doi:10.1001/jama.2011.1866]).
The methodology was rigorous in this study by Dr. Habel and her colleagues, by far the largest, most comprehensive study of the cardiovascular safety of ADHD medications done to date, said Dr. Philip Shaw.
One elegant feature of the study was the authors’ presentation of several worst-case scenarios in which it was assumed that the upper range of the risk estimates applied. "Even with these assumptions, the increase in absolute risk difference is small" between users and nonusers of ADHD medications, Dr. Shaw noted.
"Such absolute risk estimates allow clinicians to form their own opinion of the clinical importance of the worst-case scenario," he said. In addition, "no link was found between increasing risk and increasing duration of medication use, and the upper range of use was as high as 13.5 years," he said.
Therefore, this study is of value to clinicians when counseling patients on the risks and benefits of ADHD medications. "Now there is solid evidence – perhaps even some heartening news – that physicians can use to address concerns about cardiovascular risk," he concluded.
Dr. Shaw is at the National Human Genome Research Institute, Bethesda,
Md. He reported receiving travel grants from Jannsen-Cilag, maker of Concerta (methylphenidate). These remarks were taken from his editorial accompanying Dr. Habel’s report (JAMA 2011 Dec. 12 [doi:10.1001/jama.2011.1866]).
Medications for attention-deficit/hyperactivity disorder do not increase the risk of serious cardiovascular events among young and middle-aged adults, according to the results of a retrospective, population-based cohort study published online Dec. 12 in JAMA.
Stimulants such as methylphenidate, amphetamines, and pemoline, as well as the selective norepinephrine reuptake inhibitor atomoxetine, are known to raise heart rate and to increase systolic blood pressure by approximately 2-5 mm Hg and diastolic blood pressure by 1-3 mm Hg. "Although these effects would be expected to slightly increase risk for myocardial infarction, sudden cardiac death, and stroke, clinical trials have not been large enough to assess risk of these events," said Laurel A. Habel, Ph.D., of Kaiser Permanente Northern California, Oakland, and her associates.
Moreover, cardiovascular safety data from pharmacoepidemiologic studies have been "limited and inconsistent" to date, they said.
Dr. Habel and her colleagues examined the relationship between attention-deficit/hyperactivity disorder (ADHD) medications and cardiovascular risk in a large, geographically and sociodemographically diverse cohort of 443,198 adults aged 25-64 years. A total of 150,359 subjects used ADHD medications, including methylphenidate (45%), amphetamines (44%), atomoxetine, (8%), and pemoline (3%).
Medication use was assessed using pharmacy data on filled prescriptions. In addition, the medical records for all potential cases of MI, sudden cardiac death, and stroke were reviewed to verify those events.
There were 1,357 MIs, 296 sudden cardiac deaths, and 575 strokes in 806,182 person-years of follow-up.
The rates of all of these events combined were not significantly different between current users of ADHD medications and nonusers, with a multivariate adjusted rate ratio of 0.83. There also were no significant differences in these rates between current users and subjects who had used ADHD medications in the past but had stopped taking them (rate ratio, 1.03), the investigators said (JAMA 2011 Dec. 12 [doi:10.1001/jama.2011.1830]).
These results remained consistent when each medication was assessed individually, as well as when ischemic and hemorrhagic strokes were examined separately.
To control for potential differences in cardiovascular disease risk between exposed and nonexposed study subjects, the investigators assigned each subject a summary cardiovascular risk score at baseline. After the data were adjusted for these scores, the results were essentially the same: no increase in the risk of CVD events among ADHD medication users vs. nonusers, regardless of the subjects’ baseline CVD risk.
The data also were analyzed according to whether subjects were new to using ADHD medications or were long-term users, and again there was no difference in CVD risk. Rate ratios also remained consistent in all subgroup analyses, including an assessment of CVD risk by subject age.
Information on some important risk factors and on medication dosage was unavailable, so this study, despite its large size, could not completely rule out a "modestly" elevated risk with ADHD medications, the researchers noted.
This study was funded in part by the Agency for Healthcare Research and Quality, the Food and Drug Administration, and the National Institute on Aging. Dr. Habel reported ties to Merck, Takeda, and Sanofi-Aventis, and her associates reported ties to Optuminsight, Merck, GlaxoSmithKline, Novartis, and Abbott. Dr. Shaw reported receiving travel grants from Jannsen-Cilag, maker of Concerta (methylphenidate).
Medications for attention-deficit/hyperactivity disorder do not increase the risk of serious cardiovascular events among young and middle-aged adults, according to the results of a retrospective, population-based cohort study published online Dec. 12 in JAMA.
Stimulants such as methylphenidate, amphetamines, and pemoline, as well as the selective norepinephrine reuptake inhibitor atomoxetine, are known to raise heart rate and to increase systolic blood pressure by approximately 2-5 mm Hg and diastolic blood pressure by 1-3 mm Hg. "Although these effects would be expected to slightly increase risk for myocardial infarction, sudden cardiac death, and stroke, clinical trials have not been large enough to assess risk of these events," said Laurel A. Habel, Ph.D., of Kaiser Permanente Northern California, Oakland, and her associates.
Moreover, cardiovascular safety data from pharmacoepidemiologic studies have been "limited and inconsistent" to date, they said.
Dr. Habel and her colleagues examined the relationship between attention-deficit/hyperactivity disorder (ADHD) medications and cardiovascular risk in a large, geographically and sociodemographically diverse cohort of 443,198 adults aged 25-64 years. A total of 150,359 subjects used ADHD medications, including methylphenidate (45%), amphetamines (44%), atomoxetine, (8%), and pemoline (3%).
Medication use was assessed using pharmacy data on filled prescriptions. In addition, the medical records for all potential cases of MI, sudden cardiac death, and stroke were reviewed to verify those events.
There were 1,357 MIs, 296 sudden cardiac deaths, and 575 strokes in 806,182 person-years of follow-up.
The rates of all of these events combined were not significantly different between current users of ADHD medications and nonusers, with a multivariate adjusted rate ratio of 0.83. There also were no significant differences in these rates between current users and subjects who had used ADHD medications in the past but had stopped taking them (rate ratio, 1.03), the investigators said (JAMA 2011 Dec. 12 [doi:10.1001/jama.2011.1830]).
These results remained consistent when each medication was assessed individually, as well as when ischemic and hemorrhagic strokes were examined separately.
To control for potential differences in cardiovascular disease risk between exposed and nonexposed study subjects, the investigators assigned each subject a summary cardiovascular risk score at baseline. After the data were adjusted for these scores, the results were essentially the same: no increase in the risk of CVD events among ADHD medication users vs. nonusers, regardless of the subjects’ baseline CVD risk.
The data also were analyzed according to whether subjects were new to using ADHD medications or were long-term users, and again there was no difference in CVD risk. Rate ratios also remained consistent in all subgroup analyses, including an assessment of CVD risk by subject age.
Information on some important risk factors and on medication dosage was unavailable, so this study, despite its large size, could not completely rule out a "modestly" elevated risk with ADHD medications, the researchers noted.
This study was funded in part by the Agency for Healthcare Research and Quality, the Food and Drug Administration, and the National Institute on Aging. Dr. Habel reported ties to Merck, Takeda, and Sanofi-Aventis, and her associates reported ties to Optuminsight, Merck, GlaxoSmithKline, Novartis, and Abbott. Dr. Shaw reported receiving travel grants from Jannsen-Cilag, maker of Concerta (methylphenidate).
FROM JAMA
Major Finding: Rates of myocardial infarction, sudden cardiac death, and stroke were not significantly different between adults who used ADHD medications and those who did not.
Data Source: A retrospective population-based cohort study involving 443,198 subjects aged 25-64 years, of whom 150,359 were using methylphenidate, amphetamines, pemoline, or atomoxetine for ADHD.
Disclosures: This study was funded in part by the Agency for Healthcare Research and Quality, the Food and Drug Administration, and the National Institute on Aging. Dr. Habel reported ties to Merck, Takeda, and Sanofi-Aventis, and her associates reported ties to Optuminsight, Merck, GlaxoSmithKline, Novartis, and Abbott.
Cortical Demyelination, Inflammation Found in Early MS
Cortical demyelination is common early in the course of multiple sclerosis and is inflammatory in nature, according to an analysis of brain biopsy samples containing cortical tissue.
"These findings do not support a primary (noninflammatory) neurodegenerative process during early-stage multiple sclerosis," Dr. Claudia F. Lucchinetti of the Mayo Clinic, Rochester, Minn., and her associates wrote in the Dec. 8 issue of the New England Journal of Medicine.
Most previous studies of cortical lesions have focused on autopsy findings in patients with longstanding multiple sclerosis and "have suggested that neurodegeneration proceeds independently of parenchymal inflammation," the investigators noted.
They chose instead to study the prevalence and histopathologic features of cortical demyelination in brain biopsy samples from 563 patients who underwent the procedure to rule out other possible causes of their neurological symptoms, such as brain tumors. The cortical matter was obtained "in passing," in samples that were targeting white-matter lesions. The samples were obtained within a median of 27 days from the onset of symptoms.
A total of 138 patients’ samples contained a sufficient amount of cortex for analysis. Of these 138 patients, 77 had clinical follow-up for a median of 3.5 years. MS was eventually diagnosed in 58 (75%), and a clinically isolated syndrome was diagnosed in the remaining 19 (25%).
In all, 53 of the 138 samples (38%) showed cortical demyelination, Dr. Lucchinetti and her colleagues reported (N. Engl. J. Med. 2011;365:2188-97).
The lesions were highly inflammatory and had a high prevalence of CD3-positive and CD8-positive T-cell infiltrates as well as myelin-laden macrophages.
In addition, among patients who had sufficient meningeal tissue for analysis, meningeal inflammation was topographically adjacent to the cortical demyelination.
The researchers also found concurrent subpial and leukocortical lesions within individual tissue sections, "suggesting that superficial demyelinating disease may contribute to the generation of deeper lesions by means of cytokine diffusion."
In addition, "our findings of microglial activation, neuritic injury, pyknotic neurons, and reduced oligodendrocyte density ... are consonant with the findings in patients with progressive MS, underscoring the potential of cortical demyelination to cause irreversible injury, although inflammation may resolve rapidly."
They speculated that the mechanism of MS progression might involve "myelin-laden macrophages leaving the cortex, entering the cerebrospinal fluid, and gaining access to deep cervical lymph nodes to promote epitope spreading."
This study was supported by the National Multiple Sclerosis Society and the National Institutes of Health. Dr. Lucchinetti’s associates reported ties to numerous companies that market and develop drugs for MS, as well as receiving research funding or travel awards from research institutions or patient advocacy organizations.
The "provocative" findings of Dr. Lucchinetti and her colleagues "provide definitive evidence that inflammatory disease of the gray matter commences early in the pathogenesis of some cases of multiple sclerosis," wrote Dr. Peter A. Calabresi.
Before now, macrophages laden with myelin – the hallmark of an active plaque – have not been seen in cortical tissue, and gray-matter lesions have been "routinely underestimated" because conventional MRI doesn’t pick up MS plaques in the cortical and deep gray structures, he said.
This study suggests that cortical neuronal loss "is directly associated with inflammatory demyelination, and therefore early therapeutic efforts to suppress inflammation may be neuroprotective in both gray-matter and white-matter compartments," Dr. Calabresi added.
Dr. Calabresi is in the department of neurology at Johns Hopkins Hospital, Baltimore. He reported ties to numerous companies that market and develop drugs for MS. These remarks were adapted from his editorial comment accompanying Dr. Lucchinetti’s report (N. Engl. J. Med. 2011;365:2231-3).
The "provocative" findings of Dr. Lucchinetti and her colleagues "provide definitive evidence that inflammatory disease of the gray matter commences early in the pathogenesis of some cases of multiple sclerosis," wrote Dr. Peter A. Calabresi.
Before now, macrophages laden with myelin – the hallmark of an active plaque – have not been seen in cortical tissue, and gray-matter lesions have been "routinely underestimated" because conventional MRI doesn’t pick up MS plaques in the cortical and deep gray structures, he said.
This study suggests that cortical neuronal loss "is directly associated with inflammatory demyelination, and therefore early therapeutic efforts to suppress inflammation may be neuroprotective in both gray-matter and white-matter compartments," Dr. Calabresi added.
Dr. Calabresi is in the department of neurology at Johns Hopkins Hospital, Baltimore. He reported ties to numerous companies that market and develop drugs for MS. These remarks were adapted from his editorial comment accompanying Dr. Lucchinetti’s report (N. Engl. J. Med. 2011;365:2231-3).
The "provocative" findings of Dr. Lucchinetti and her colleagues "provide definitive evidence that inflammatory disease of the gray matter commences early in the pathogenesis of some cases of multiple sclerosis," wrote Dr. Peter A. Calabresi.
Before now, macrophages laden with myelin – the hallmark of an active plaque – have not been seen in cortical tissue, and gray-matter lesions have been "routinely underestimated" because conventional MRI doesn’t pick up MS plaques in the cortical and deep gray structures, he said.
This study suggests that cortical neuronal loss "is directly associated with inflammatory demyelination, and therefore early therapeutic efforts to suppress inflammation may be neuroprotective in both gray-matter and white-matter compartments," Dr. Calabresi added.
Dr. Calabresi is in the department of neurology at Johns Hopkins Hospital, Baltimore. He reported ties to numerous companies that market and develop drugs for MS. These remarks were adapted from his editorial comment accompanying Dr. Lucchinetti’s report (N. Engl. J. Med. 2011;365:2231-3).
Cortical demyelination is common early in the course of multiple sclerosis and is inflammatory in nature, according to an analysis of brain biopsy samples containing cortical tissue.
"These findings do not support a primary (noninflammatory) neurodegenerative process during early-stage multiple sclerosis," Dr. Claudia F. Lucchinetti of the Mayo Clinic, Rochester, Minn., and her associates wrote in the Dec. 8 issue of the New England Journal of Medicine.
Most previous studies of cortical lesions have focused on autopsy findings in patients with longstanding multiple sclerosis and "have suggested that neurodegeneration proceeds independently of parenchymal inflammation," the investigators noted.
They chose instead to study the prevalence and histopathologic features of cortical demyelination in brain biopsy samples from 563 patients who underwent the procedure to rule out other possible causes of their neurological symptoms, such as brain tumors. The cortical matter was obtained "in passing," in samples that were targeting white-matter lesions. The samples were obtained within a median of 27 days from the onset of symptoms.
A total of 138 patients’ samples contained a sufficient amount of cortex for analysis. Of these 138 patients, 77 had clinical follow-up for a median of 3.5 years. MS was eventually diagnosed in 58 (75%), and a clinically isolated syndrome was diagnosed in the remaining 19 (25%).
In all, 53 of the 138 samples (38%) showed cortical demyelination, Dr. Lucchinetti and her colleagues reported (N. Engl. J. Med. 2011;365:2188-97).
The lesions were highly inflammatory and had a high prevalence of CD3-positive and CD8-positive T-cell infiltrates as well as myelin-laden macrophages.
In addition, among patients who had sufficient meningeal tissue for analysis, meningeal inflammation was topographically adjacent to the cortical demyelination.
The researchers also found concurrent subpial and leukocortical lesions within individual tissue sections, "suggesting that superficial demyelinating disease may contribute to the generation of deeper lesions by means of cytokine diffusion."
In addition, "our findings of microglial activation, neuritic injury, pyknotic neurons, and reduced oligodendrocyte density ... are consonant with the findings in patients with progressive MS, underscoring the potential of cortical demyelination to cause irreversible injury, although inflammation may resolve rapidly."
They speculated that the mechanism of MS progression might involve "myelin-laden macrophages leaving the cortex, entering the cerebrospinal fluid, and gaining access to deep cervical lymph nodes to promote epitope spreading."
This study was supported by the National Multiple Sclerosis Society and the National Institutes of Health. Dr. Lucchinetti’s associates reported ties to numerous companies that market and develop drugs for MS, as well as receiving research funding or travel awards from research institutions or patient advocacy organizations.
Cortical demyelination is common early in the course of multiple sclerosis and is inflammatory in nature, according to an analysis of brain biopsy samples containing cortical tissue.
"These findings do not support a primary (noninflammatory) neurodegenerative process during early-stage multiple sclerosis," Dr. Claudia F. Lucchinetti of the Mayo Clinic, Rochester, Minn., and her associates wrote in the Dec. 8 issue of the New England Journal of Medicine.
Most previous studies of cortical lesions have focused on autopsy findings in patients with longstanding multiple sclerosis and "have suggested that neurodegeneration proceeds independently of parenchymal inflammation," the investigators noted.
They chose instead to study the prevalence and histopathologic features of cortical demyelination in brain biopsy samples from 563 patients who underwent the procedure to rule out other possible causes of their neurological symptoms, such as brain tumors. The cortical matter was obtained "in passing," in samples that were targeting white-matter lesions. The samples were obtained within a median of 27 days from the onset of symptoms.
A total of 138 patients’ samples contained a sufficient amount of cortex for analysis. Of these 138 patients, 77 had clinical follow-up for a median of 3.5 years. MS was eventually diagnosed in 58 (75%), and a clinically isolated syndrome was diagnosed in the remaining 19 (25%).
In all, 53 of the 138 samples (38%) showed cortical demyelination, Dr. Lucchinetti and her colleagues reported (N. Engl. J. Med. 2011;365:2188-97).
The lesions were highly inflammatory and had a high prevalence of CD3-positive and CD8-positive T-cell infiltrates as well as myelin-laden macrophages.
In addition, among patients who had sufficient meningeal tissue for analysis, meningeal inflammation was topographically adjacent to the cortical demyelination.
The researchers also found concurrent subpial and leukocortical lesions within individual tissue sections, "suggesting that superficial demyelinating disease may contribute to the generation of deeper lesions by means of cytokine diffusion."
In addition, "our findings of microglial activation, neuritic injury, pyknotic neurons, and reduced oligodendrocyte density ... are consonant with the findings in patients with progressive MS, underscoring the potential of cortical demyelination to cause irreversible injury, although inflammation may resolve rapidly."
They speculated that the mechanism of MS progression might involve "myelin-laden macrophages leaving the cortex, entering the cerebrospinal fluid, and gaining access to deep cervical lymph nodes to promote epitope spreading."
This study was supported by the National Multiple Sclerosis Society and the National Institutes of Health. Dr. Lucchinetti’s associates reported ties to numerous companies that market and develop drugs for MS, as well as receiving research funding or travel awards from research institutions or patient advocacy organizations.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: A total of 53 (38%) of 138 biopsy samples of cortical tissue showed demyelination, with a high prevalence of inflammation.
Data Source: An analysis of brain biopsy samples containing cortical tissue from 138 patients early in the course of MS, often before the disease was diagnosed.
Disclosures: This study was supported by the National Multiple Sclerosis Society and the National Institutes of Health. Dr. Lucchinetti’s associates reported ties to numerous companies that market and develop drugs for MS, as well as receiving research funding or travel awards from research institutions or patient advocacy organizations.
Investigational Norovirus Vaccine Trims Norwalk Infection Risk
An investigational norovirus vaccine was protective against Norwalk virus infection and its associated gastroenteritis in a proof-of-concept study reported in the Dec. 8 issue of the New England Journal of Medicine.
The two-dose, intranasal vaccine, which contains norovirus viruslike particles (VLP) plus the adjuvants chitosan and monophosphoryl lipid A, reduced the relative rate of infection by 26% and the relative rate of gastroenteritis by 47% compared with placebo.
The findings demonstrate that "it may be possible to use a vaccination strategy to prevent norovirus disease," which currently causes an estimated 21 million cases of gastroenteritis each year in the United States and 218,000 deaths among children alone, said Dr. Robert L. Atmar of Baylor College of Medicine, Houston, and his associates.
The double-blind clinical trial involved 98 healthy adults aged 18-50 years (mean age, 32 years) who were followed at four clinical sites. In the first phase of the study, subjects were randomly assigned to receive the study vaccine or placebo in two intranasal doses administered 3 weeks apart. Serum samples were collected another 3 weeks after the second dose was given, to assess immunogenicity.
Subjects who had not dropped out of the study at that time were eligible for the second phase, in which they underwent challenge by oral administration of Norwalk virus in an inpatient facility where they could be assessed for gastroenteritis and treated, if necessary.
A total of 89 subjects received both doses of placebo or vaccine, and 84 participated in the Norwalk virus challenge.
Norwalk infection, documented by assessment of stool samples, developed in 61% who received active vaccine and 82% who received placebo, for a relative reduction of 26%. Norwalk-associated gastroenteritis developed in 37% who received active vaccine and 69% who received placebo, for a relative reduction of 47%, the investigators said (N. Engl. J. Med. 2011;365:2178-87).
Moreover, the severity of gastroenteritis was significantly decreased among subjects who received active vaccine. The onset of illness also was delayed by several hours, although the overall duration was not significantly shorter with the active vaccine.
In all, 70% of the subjects who received active vaccine showed Norwalk virus–specific serum IgA antibody responses. "The frequency and magnitude of serum antibody responses after vaccination were lower than those induced by infection. Immunity after natural infection is short-lived [less than 2 years], and the duration of protection after vaccination remains to be determined," they noted.
"No vaccine-related severe adverse events occurred during the study, and no new medically significant conditions occurred," Dr. Atmar and his colleagues said.
The intranasal delivery system used in this study "malfunctioned several times, adversely affecting vaccine delivery. Alternative intranasal delivery systems and parenteral delivery of vaccine antigens are being evaluated to determine the optimal route of delivery," they added.
Future studies are needed to determine vaccine immunogenicity and protection in other patient populations, notably children and the elderly. "Also, it is possible that norovirus vaccines, like rotavirus vaccines, will provide greater protection against severe disease than against milder illness," they said.
This study was sponsored by LigoCyte Pharmaceuticals, maker of the vaccine, and the National Institutes of Health. Dr. Atmar reported being a consultant to GlaxoSmithKline, Novartis, and Haymarket Media; his associates reported ties to Novartis, Denka Pharmaceuticals, Global Vaccines, Immucell, and the EMMES Corporation.
An investigational norovirus vaccine was protective against Norwalk virus infection and its associated gastroenteritis in a proof-of-concept study reported in the Dec. 8 issue of the New England Journal of Medicine.
The two-dose, intranasal vaccine, which contains norovirus viruslike particles (VLP) plus the adjuvants chitosan and monophosphoryl lipid A, reduced the relative rate of infection by 26% and the relative rate of gastroenteritis by 47% compared with placebo.
The findings demonstrate that "it may be possible to use a vaccination strategy to prevent norovirus disease," which currently causes an estimated 21 million cases of gastroenteritis each year in the United States and 218,000 deaths among children alone, said Dr. Robert L. Atmar of Baylor College of Medicine, Houston, and his associates.
The double-blind clinical trial involved 98 healthy adults aged 18-50 years (mean age, 32 years) who were followed at four clinical sites. In the first phase of the study, subjects were randomly assigned to receive the study vaccine or placebo in two intranasal doses administered 3 weeks apart. Serum samples were collected another 3 weeks after the second dose was given, to assess immunogenicity.
Subjects who had not dropped out of the study at that time were eligible for the second phase, in which they underwent challenge by oral administration of Norwalk virus in an inpatient facility where they could be assessed for gastroenteritis and treated, if necessary.
A total of 89 subjects received both doses of placebo or vaccine, and 84 participated in the Norwalk virus challenge.
Norwalk infection, documented by assessment of stool samples, developed in 61% who received active vaccine and 82% who received placebo, for a relative reduction of 26%. Norwalk-associated gastroenteritis developed in 37% who received active vaccine and 69% who received placebo, for a relative reduction of 47%, the investigators said (N. Engl. J. Med. 2011;365:2178-87).
Moreover, the severity of gastroenteritis was significantly decreased among subjects who received active vaccine. The onset of illness also was delayed by several hours, although the overall duration was not significantly shorter with the active vaccine.
In all, 70% of the subjects who received active vaccine showed Norwalk virus–specific serum IgA antibody responses. "The frequency and magnitude of serum antibody responses after vaccination were lower than those induced by infection. Immunity after natural infection is short-lived [less than 2 years], and the duration of protection after vaccination remains to be determined," they noted.
"No vaccine-related severe adverse events occurred during the study, and no new medically significant conditions occurred," Dr. Atmar and his colleagues said.
The intranasal delivery system used in this study "malfunctioned several times, adversely affecting vaccine delivery. Alternative intranasal delivery systems and parenteral delivery of vaccine antigens are being evaluated to determine the optimal route of delivery," they added.
Future studies are needed to determine vaccine immunogenicity and protection in other patient populations, notably children and the elderly. "Also, it is possible that norovirus vaccines, like rotavirus vaccines, will provide greater protection against severe disease than against milder illness," they said.
This study was sponsored by LigoCyte Pharmaceuticals, maker of the vaccine, and the National Institutes of Health. Dr. Atmar reported being a consultant to GlaxoSmithKline, Novartis, and Haymarket Media; his associates reported ties to Novartis, Denka Pharmaceuticals, Global Vaccines, Immucell, and the EMMES Corporation.
An investigational norovirus vaccine was protective against Norwalk virus infection and its associated gastroenteritis in a proof-of-concept study reported in the Dec. 8 issue of the New England Journal of Medicine.
The two-dose, intranasal vaccine, which contains norovirus viruslike particles (VLP) plus the adjuvants chitosan and monophosphoryl lipid A, reduced the relative rate of infection by 26% and the relative rate of gastroenteritis by 47% compared with placebo.
The findings demonstrate that "it may be possible to use a vaccination strategy to prevent norovirus disease," which currently causes an estimated 21 million cases of gastroenteritis each year in the United States and 218,000 deaths among children alone, said Dr. Robert L. Atmar of Baylor College of Medicine, Houston, and his associates.
The double-blind clinical trial involved 98 healthy adults aged 18-50 years (mean age, 32 years) who were followed at four clinical sites. In the first phase of the study, subjects were randomly assigned to receive the study vaccine or placebo in two intranasal doses administered 3 weeks apart. Serum samples were collected another 3 weeks after the second dose was given, to assess immunogenicity.
Subjects who had not dropped out of the study at that time were eligible for the second phase, in which they underwent challenge by oral administration of Norwalk virus in an inpatient facility where they could be assessed for gastroenteritis and treated, if necessary.
A total of 89 subjects received both doses of placebo or vaccine, and 84 participated in the Norwalk virus challenge.
Norwalk infection, documented by assessment of stool samples, developed in 61% who received active vaccine and 82% who received placebo, for a relative reduction of 26%. Norwalk-associated gastroenteritis developed in 37% who received active vaccine and 69% who received placebo, for a relative reduction of 47%, the investigators said (N. Engl. J. Med. 2011;365:2178-87).
Moreover, the severity of gastroenteritis was significantly decreased among subjects who received active vaccine. The onset of illness also was delayed by several hours, although the overall duration was not significantly shorter with the active vaccine.
In all, 70% of the subjects who received active vaccine showed Norwalk virus–specific serum IgA antibody responses. "The frequency and magnitude of serum antibody responses after vaccination were lower than those induced by infection. Immunity after natural infection is short-lived [less than 2 years], and the duration of protection after vaccination remains to be determined," they noted.
"No vaccine-related severe adverse events occurred during the study, and no new medically significant conditions occurred," Dr. Atmar and his colleagues said.
The intranasal delivery system used in this study "malfunctioned several times, adversely affecting vaccine delivery. Alternative intranasal delivery systems and parenteral delivery of vaccine antigens are being evaluated to determine the optimal route of delivery," they added.
Future studies are needed to determine vaccine immunogenicity and protection in other patient populations, notably children and the elderly. "Also, it is possible that norovirus vaccines, like rotavirus vaccines, will provide greater protection against severe disease than against milder illness," they said.
This study was sponsored by LigoCyte Pharmaceuticals, maker of the vaccine, and the National Institutes of Health. Dr. Atmar reported being a consultant to GlaxoSmithKline, Novartis, and Haymarket Media; his associates reported ties to Novartis, Denka Pharmaceuticals, Global Vaccines, Immucell, and the EMMES Corporation.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: A total of 61% of subjects who received active vaccine and 82% who received placebo developed Norwalk infection, for a relative reduction of 26%; 37% who received active vaccine and 69% who received placebo developed Norwalk-associated gastroenteritis, for a relative reduction of 47%.
Data Source: A multicenter, randomized, double-blind proof-of-concept study to assess the safety, efficacy, and immunogenicity of an investigational norovirus vaccine in 98 healthy men and women.
Disclosures: This study was sponsored by LigoCyte Pharmaceuticals, maker of the vaccine, and the National Institutes of Health. Dr. Atmar reported being a consultant to GlaxoSmithKline, Novartis, and Haymarket Media; his associates reported ties to Novartis, Denka Pharmaceuticals, Global Vaccines, Immucell, and the EMMES Corporation.