User login
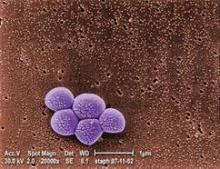
Screen Open-Airway Surgery Patients for MRSA
Children who are to have open airway surgery should first be screened for methicillin-resistant Staphylococcus aureus colonization because the prevalence is particularly high in this patient group and treatment drastically reduces postoperative infections, graft loss, and wound dehiscence, researchers reported.
In a retrospective cohort study at a single tertiary pediatric medical center, the prevalence of MRSA colonization was 32.5% during a 2-year period among 175 children who underwent 197 open airway operations, a rate considerably higher than has been reported in patients undergoing other types of surgery, said Dr. Melissa McCarty Statham of the department of otolaryngology–head and neck surgery, Emory University, Atlanta, and her associates. The findings were reported in the February issue of the Archives of Otolaryngology and Head & Neck Surgery.
Because these MRSA-colonized patients were identified and treated appropriately, they did not develop any postoperative MRSA infections, graft losses, or cases of surgical site dehiscence, the investigators noted.
Dr. McCarty Statham and her colleagues studied this issue because, "in our experience, MRSA infection in open airway procedures can be a devastating complication." Such procedures include laryngotracheal reconstruction and grafting, correction of laryngotracheoesophageal clefts, repair of tracheoesophageal fistulas, and laryngotracheal separations.
These patients are at high risk for MRSA colonization because most are preterm; have been tracheotomized; and have serious comorbidities such as pulmonary, gastrointestinal, and cardiac disease. "We consider these factors to be proxies for frequent hospitalization and exposure to antibiotics," the researchers said.
They assessed the 175 patients who underwent such surgery (at a median age of 4 years) at the Cincinnati Children’s Hospital Medical Center in the 2 years after a program of MRSA screening and treatment had been instituted there. Their purpose was to document the prevalence of MRSA colonization in this vulnerable patient population and to assess the effect of the program. Preoperatively, all patients were cultured for MRSA at the nares, perianal area, axila, gastrostomy tube (if present), and tracheotomy tube aspirate (if present).
Colonized patients were given double-strength trimethoprim-sulfamethoxazole empirically for 72 hours before surgery, with clindamycin serving as an alternative in patients who were allergic to sulfa drugs or were carrying organisms resistant to TMP-SMX. Patients with positive nasal cultures also received intranasal mupirocin twice daily.
Perioperatively, colonized patients received either intravenous vancomycin or clindamycin. Postoperatively, they received the same antibiotic regimen for 14 days as they had been given before surgery.
No MRSA-associated infections developed in patients treated according to this protocol, Dr. McCarty Statham and her associates said (Arch. Otolaryngol. Head Neck Surg. 2012;138:153-7).
Postoperative rates of any infection were comparable between the patients colonized with MRSA and those not colonized. There were 10 infections in the MRSA-positive patients (a rate of 15.9%) and 23 infections in the noncolonized patients (a rate of 17.4%).
All 10 infections in the MRSA-colonized patients were caused by nosocomial non-MRSA organisms, as were 19 of the 23 infections in the noncolonized patients.
Three patients who had been MRSA-negative at screening nevertheless developed postoperative MRSA infections after surgery, suggesting that their MRSA was acquired during this hospitalization, the investigators said.
Overall, there were two failures of laryngotracheal reconstruction cartilage grafts and one case of surgical site dehiscence, but none of them occurred in MRSA-positive patients. One graft failure was attributed to impaired wound healing as a result of corticosteroid use; the other to beta-hemolytic Streptococcus infection. The dehiscence was caused by Haemophilus influenzae infection at the surgical site.
This finding suggests that "there is an inherent risk of graft loss and dehiscence in all patients who undergo airway surgery. Infections other than MRSA may be causative factors," Dr. McCarty Statham and her associates noted.
"In view of our results, we advise instituting MRSA screening and treatment protocols in patients undergoing airway surgery," they added.
One of Dr. McCarty Statham’s associates reported being an adviser or consultant to Acclarent, Gyrus/Olympus, Boston Medical Products, Hood Laboratories, Bryan Medical, and Karl Storz.
Children who are to have open airway surgery should first be screened for methicillin-resistant Staphylococcus aureus colonization because the prevalence is particularly high in this patient group and treatment drastically reduces postoperative infections, graft loss, and wound dehiscence, researchers reported.
In a retrospective cohort study at a single tertiary pediatric medical center, the prevalence of MRSA colonization was 32.5% during a 2-year period among 175 children who underwent 197 open airway operations, a rate considerably higher than has been reported in patients undergoing other types of surgery, said Dr. Melissa McCarty Statham of the department of otolaryngology–head and neck surgery, Emory University, Atlanta, and her associates. The findings were reported in the February issue of the Archives of Otolaryngology and Head & Neck Surgery.
Because these MRSA-colonized patients were identified and treated appropriately, they did not develop any postoperative MRSA infections, graft losses, or cases of surgical site dehiscence, the investigators noted.
Dr. McCarty Statham and her colleagues studied this issue because, "in our experience, MRSA infection in open airway procedures can be a devastating complication." Such procedures include laryngotracheal reconstruction and grafting, correction of laryngotracheoesophageal clefts, repair of tracheoesophageal fistulas, and laryngotracheal separations.
These patients are at high risk for MRSA colonization because most are preterm; have been tracheotomized; and have serious comorbidities such as pulmonary, gastrointestinal, and cardiac disease. "We consider these factors to be proxies for frequent hospitalization and exposure to antibiotics," the researchers said.
They assessed the 175 patients who underwent such surgery (at a median age of 4 years) at the Cincinnati Children’s Hospital Medical Center in the 2 years after a program of MRSA screening and treatment had been instituted there. Their purpose was to document the prevalence of MRSA colonization in this vulnerable patient population and to assess the effect of the program. Preoperatively, all patients were cultured for MRSA at the nares, perianal area, axila, gastrostomy tube (if present), and tracheotomy tube aspirate (if present).
Colonized patients were given double-strength trimethoprim-sulfamethoxazole empirically for 72 hours before surgery, with clindamycin serving as an alternative in patients who were allergic to sulfa drugs or were carrying organisms resistant to TMP-SMX. Patients with positive nasal cultures also received intranasal mupirocin twice daily.
Perioperatively, colonized patients received either intravenous vancomycin or clindamycin. Postoperatively, they received the same antibiotic regimen for 14 days as they had been given before surgery.
No MRSA-associated infections developed in patients treated according to this protocol, Dr. McCarty Statham and her associates said (Arch. Otolaryngol. Head Neck Surg. 2012;138:153-7).
Postoperative rates of any infection were comparable between the patients colonized with MRSA and those not colonized. There were 10 infections in the MRSA-positive patients (a rate of 15.9%) and 23 infections in the noncolonized patients (a rate of 17.4%).
All 10 infections in the MRSA-colonized patients were caused by nosocomial non-MRSA organisms, as were 19 of the 23 infections in the noncolonized patients.
Three patients who had been MRSA-negative at screening nevertheless developed postoperative MRSA infections after surgery, suggesting that their MRSA was acquired during this hospitalization, the investigators said.
Overall, there were two failures of laryngotracheal reconstruction cartilage grafts and one case of surgical site dehiscence, but none of them occurred in MRSA-positive patients. One graft failure was attributed to impaired wound healing as a result of corticosteroid use; the other to beta-hemolytic Streptococcus infection. The dehiscence was caused by Haemophilus influenzae infection at the surgical site.
This finding suggests that "there is an inherent risk of graft loss and dehiscence in all patients who undergo airway surgery. Infections other than MRSA may be causative factors," Dr. McCarty Statham and her associates noted.
"In view of our results, we advise instituting MRSA screening and treatment protocols in patients undergoing airway surgery," they added.
One of Dr. McCarty Statham’s associates reported being an adviser or consultant to Acclarent, Gyrus/Olympus, Boston Medical Products, Hood Laboratories, Bryan Medical, and Karl Storz.
Children who are to have open airway surgery should first be screened for methicillin-resistant Staphylococcus aureus colonization because the prevalence is particularly high in this patient group and treatment drastically reduces postoperative infections, graft loss, and wound dehiscence, researchers reported.
In a retrospective cohort study at a single tertiary pediatric medical center, the prevalence of MRSA colonization was 32.5% during a 2-year period among 175 children who underwent 197 open airway operations, a rate considerably higher than has been reported in patients undergoing other types of surgery, said Dr. Melissa McCarty Statham of the department of otolaryngology–head and neck surgery, Emory University, Atlanta, and her associates. The findings were reported in the February issue of the Archives of Otolaryngology and Head & Neck Surgery.
Because these MRSA-colonized patients were identified and treated appropriately, they did not develop any postoperative MRSA infections, graft losses, or cases of surgical site dehiscence, the investigators noted.
Dr. McCarty Statham and her colleagues studied this issue because, "in our experience, MRSA infection in open airway procedures can be a devastating complication." Such procedures include laryngotracheal reconstruction and grafting, correction of laryngotracheoesophageal clefts, repair of tracheoesophageal fistulas, and laryngotracheal separations.
These patients are at high risk for MRSA colonization because most are preterm; have been tracheotomized; and have serious comorbidities such as pulmonary, gastrointestinal, and cardiac disease. "We consider these factors to be proxies for frequent hospitalization and exposure to antibiotics," the researchers said.
They assessed the 175 patients who underwent such surgery (at a median age of 4 years) at the Cincinnati Children’s Hospital Medical Center in the 2 years after a program of MRSA screening and treatment had been instituted there. Their purpose was to document the prevalence of MRSA colonization in this vulnerable patient population and to assess the effect of the program. Preoperatively, all patients were cultured for MRSA at the nares, perianal area, axila, gastrostomy tube (if present), and tracheotomy tube aspirate (if present).
Colonized patients were given double-strength trimethoprim-sulfamethoxazole empirically for 72 hours before surgery, with clindamycin serving as an alternative in patients who were allergic to sulfa drugs or were carrying organisms resistant to TMP-SMX. Patients with positive nasal cultures also received intranasal mupirocin twice daily.
Perioperatively, colonized patients received either intravenous vancomycin or clindamycin. Postoperatively, they received the same antibiotic regimen for 14 days as they had been given before surgery.
No MRSA-associated infections developed in patients treated according to this protocol, Dr. McCarty Statham and her associates said (Arch. Otolaryngol. Head Neck Surg. 2012;138:153-7).
Postoperative rates of any infection were comparable between the patients colonized with MRSA and those not colonized. There were 10 infections in the MRSA-positive patients (a rate of 15.9%) and 23 infections in the noncolonized patients (a rate of 17.4%).
All 10 infections in the MRSA-colonized patients were caused by nosocomial non-MRSA organisms, as were 19 of the 23 infections in the noncolonized patients.
Three patients who had been MRSA-negative at screening nevertheless developed postoperative MRSA infections after surgery, suggesting that their MRSA was acquired during this hospitalization, the investigators said.
Overall, there were two failures of laryngotracheal reconstruction cartilage grafts and one case of surgical site dehiscence, but none of them occurred in MRSA-positive patients. One graft failure was attributed to impaired wound healing as a result of corticosteroid use; the other to beta-hemolytic Streptococcus infection. The dehiscence was caused by Haemophilus influenzae infection at the surgical site.
This finding suggests that "there is an inherent risk of graft loss and dehiscence in all patients who undergo airway surgery. Infections other than MRSA may be causative factors," Dr. McCarty Statham and her associates noted.
"In view of our results, we advise instituting MRSA screening and treatment protocols in patients undergoing airway surgery," they added.
One of Dr. McCarty Statham’s associates reported being an adviser or consultant to Acclarent, Gyrus/Olympus, Boston Medical Products, Hood Laboratories, Bryan Medical, and Karl Storz.
FROM ARCHIVES OF OTOLARYNGOLOGY AND HEAD & NECK SURGERY
Major Finding: The overall rate of MRSA colonization was 33%, but no MRSA infections developed in the screened and treated carriers; rates of any postoperative infection were comparable between colonized (15.9%) and noncolonized (17.4%) patients.
Data Source: This was a retrospective cohort study of 175 children who underwent 197 open airway surgeries at a single pediatric medical center in a 2-year period.
Disclosures: One of Dr. McCarty Statham’s associates reported ties to Acclarent, Gyrus/Olympus, Boston Medical Products, Hood Laboratories, Bryan Medical, and Karl Storz.
Prophylactic Semuloparin Cuts Rate of VTE in Cancer Patients
Semuloparin, an ultralow-molecular-weight heparin, reduced the incidence of venous thromboembolic events when given prophylactically to patients with a variety of metastatic or locally advanced solid tumors, according to a report in the Feb. 16 issue of the New England Journal of Medicine.
Semuloparin prevented both deep vein thrombosis and pulmonary embolism (PE) without raising the risk of major bleeding in patients undergoing chemotherapy for lung, pancreatic, stomach, colon, rectal, bladder, or ovarian cancer. It did not improve overall survival, however, said Dr. Giancarlo Agnelli of the department of internal medicine at the University of Perugia (Italy) and his associates.
The investigators assessed the safety and efficacy of semuloparin because this patient population has a high incidence of venous thromboembolism (VTE). They conducted the SAVE-ONCO trial (NCT00694382), which was sponsored by semuloparin’s manufacturer, Sanofi-Aventis, at 395 medical centers in 47 countries.
A total of 3,212 patients were randomly assigned to receive daily subcutaneous injections of either semuloparin (1,608 patients) or placebo (1,604 patients), beginning on the first day of chemotherapy and continuing until chemotherapy was stopped or changed to a different regimen. The concomitant use of antiplatelet agents and nonsteroidal anti-inflammatory drugs (NSAIDs) was permitted at the treating physician’s discretion.
The mean duration of treatment was 3.5 months. The reasons for discontinuing treatment were similar between patients receiving semuloparin and those receiving placebo – chiefly adverse events (15.9% and 17.5%, respectively) and the patient’s request to discontinue treatment (18.1% and 17.4%, respectively).
The primary efficacy outcome – a composite end point of any symptomatic deep vein thrombosis in the lower or upper limbs, any nonfatal pulmonary embolism, or death related to VTE – occurred in 1.2% of the semuloparin group, a significantly lower rate than the 3.4% rate in the placebo group.
Semuloparin significantly reduced the incidence of deep vein thrombosis, fatal PE, and nonfatal PE. This benefit was seen across all groups of patients categorized by cancer stage, cancer site, and geographic region, Dr. Agnelli and his colleagues reported (N. Engl. J. Med. 2012;366:601-9).
The treatment benefit also was consistent across numerous baseline risks for VTE, such as personal or family history of VTE; use of a central venous catheter; obesity; age of 75 years or more; presence of chronic respiratory or heart failure; presence of venous insufficiency or varicose veins; and use of hormone therapy.
However, semuloparin did not improve overall survival. Mortality was 43.4% in the semuloparin group and 44.5% in the placebo group.
The overall rate of major and clinically relevant nonmajor bleeding was 2.8% with semuloparin and 2.0% with placebo, a nonsignificant difference. Two fatal bleeding events occurred with semuloparin and four with placebo. "Five episodes of nonfatal bleeding in a critical area or organ were noted, all in the semuloparin group," the researchers said.
Serious adverse events developed in 26.3% of patients taking semuloparin and 25.5% of those taking placebo, a nonsignificant difference. In particular, there were no cases of heparin-induced thrombocytopenia, and the rates of thrombocytopenia were comparable between the semuloparin and placebo groups (7.1% and 7.6%, respectively).
"Current guidelines recommend antithrombotic prophylaxis for patients with cancer who are admitted to the hospital for medical illness (administered for the duration of the hospital stay) and for patients who have undergone surgery for cancer (extended for up to 5 weeks) but not for routine use in ambulatory patients receiving chemotherapy. This study suggests a benefit from antithrombotic prophylaxis with semuloparin in patients receiving chemotherapy for cancer," Dr. Agnelli and his associates said.
This study was supported by Sanofi, manufacturer of semuloparin. Dr. Agnelli and his associates reported ties to numerous industry sources.
The findings by Agnelli et al. indicate that many cancer patients "can probably avert hospitalization for deep vein thrombosis and pulmonary embolism and might live longer, if they accept an increased risk of bleeding and its subsequent treatment," said Dr. Elie A. Aki and Dr. Holger J. Schünemann.
According to the SAVE-ONCO data, if 1,000 cancer patients used a prophylactic dose of ultralow-molecular-weight heparin, "over a period of 12 months death would be averted in approximately 30 patients, venous thromboembolism would be averted in 20, and 1 would have a major bleeding episode," they noted.
Elie A. Akl, M.D., Ph.D., is in the department of medicine, State University of New York at Buffalo, and the departments of clinical epidemiology and biostatistics at McMaster University, Hamilton, Ont. Holger J. Schünemann, M.D., Ph.D., is in the departments of medicine and clinical epidemiology and biostatistics at McMaster University. They reported no relevant financial conflicts of interest. These remarks were taken from their editorial accompanying the report of Dr. Agnelli and his colleagues (N. Engl. J. Med. 2012;366:661-2).
The findings by Agnelli et al. indicate that many cancer patients "can probably avert hospitalization for deep vein thrombosis and pulmonary embolism and might live longer, if they accept an increased risk of bleeding and its subsequent treatment," said Dr. Elie A. Aki and Dr. Holger J. Schünemann.
According to the SAVE-ONCO data, if 1,000 cancer patients used a prophylactic dose of ultralow-molecular-weight heparin, "over a period of 12 months death would be averted in approximately 30 patients, venous thromboembolism would be averted in 20, and 1 would have a major bleeding episode," they noted.
Elie A. Akl, M.D., Ph.D., is in the department of medicine, State University of New York at Buffalo, and the departments of clinical epidemiology and biostatistics at McMaster University, Hamilton, Ont. Holger J. Schünemann, M.D., Ph.D., is in the departments of medicine and clinical epidemiology and biostatistics at McMaster University. They reported no relevant financial conflicts of interest. These remarks were taken from their editorial accompanying the report of Dr. Agnelli and his colleagues (N. Engl. J. Med. 2012;366:661-2).
The findings by Agnelli et al. indicate that many cancer patients "can probably avert hospitalization for deep vein thrombosis and pulmonary embolism and might live longer, if they accept an increased risk of bleeding and its subsequent treatment," said Dr. Elie A. Aki and Dr. Holger J. Schünemann.
According to the SAVE-ONCO data, if 1,000 cancer patients used a prophylactic dose of ultralow-molecular-weight heparin, "over a period of 12 months death would be averted in approximately 30 patients, venous thromboembolism would be averted in 20, and 1 would have a major bleeding episode," they noted.
Elie A. Akl, M.D., Ph.D., is in the department of medicine, State University of New York at Buffalo, and the departments of clinical epidemiology and biostatistics at McMaster University, Hamilton, Ont. Holger J. Schünemann, M.D., Ph.D., is in the departments of medicine and clinical epidemiology and biostatistics at McMaster University. They reported no relevant financial conflicts of interest. These remarks were taken from their editorial accompanying the report of Dr. Agnelli and his colleagues (N. Engl. J. Med. 2012;366:661-2).
Semuloparin, an ultralow-molecular-weight heparin, reduced the incidence of venous thromboembolic events when given prophylactically to patients with a variety of metastatic or locally advanced solid tumors, according to a report in the Feb. 16 issue of the New England Journal of Medicine.
Semuloparin prevented both deep vein thrombosis and pulmonary embolism (PE) without raising the risk of major bleeding in patients undergoing chemotherapy for lung, pancreatic, stomach, colon, rectal, bladder, or ovarian cancer. It did not improve overall survival, however, said Dr. Giancarlo Agnelli of the department of internal medicine at the University of Perugia (Italy) and his associates.
The investigators assessed the safety and efficacy of semuloparin because this patient population has a high incidence of venous thromboembolism (VTE). They conducted the SAVE-ONCO trial (NCT00694382), which was sponsored by semuloparin’s manufacturer, Sanofi-Aventis, at 395 medical centers in 47 countries.
A total of 3,212 patients were randomly assigned to receive daily subcutaneous injections of either semuloparin (1,608 patients) or placebo (1,604 patients), beginning on the first day of chemotherapy and continuing until chemotherapy was stopped or changed to a different regimen. The concomitant use of antiplatelet agents and nonsteroidal anti-inflammatory drugs (NSAIDs) was permitted at the treating physician’s discretion.
The mean duration of treatment was 3.5 months. The reasons for discontinuing treatment were similar between patients receiving semuloparin and those receiving placebo – chiefly adverse events (15.9% and 17.5%, respectively) and the patient’s request to discontinue treatment (18.1% and 17.4%, respectively).
The primary efficacy outcome – a composite end point of any symptomatic deep vein thrombosis in the lower or upper limbs, any nonfatal pulmonary embolism, or death related to VTE – occurred in 1.2% of the semuloparin group, a significantly lower rate than the 3.4% rate in the placebo group.
Semuloparin significantly reduced the incidence of deep vein thrombosis, fatal PE, and nonfatal PE. This benefit was seen across all groups of patients categorized by cancer stage, cancer site, and geographic region, Dr. Agnelli and his colleagues reported (N. Engl. J. Med. 2012;366:601-9).
The treatment benefit also was consistent across numerous baseline risks for VTE, such as personal or family history of VTE; use of a central venous catheter; obesity; age of 75 years or more; presence of chronic respiratory or heart failure; presence of venous insufficiency or varicose veins; and use of hormone therapy.
However, semuloparin did not improve overall survival. Mortality was 43.4% in the semuloparin group and 44.5% in the placebo group.
The overall rate of major and clinically relevant nonmajor bleeding was 2.8% with semuloparin and 2.0% with placebo, a nonsignificant difference. Two fatal bleeding events occurred with semuloparin and four with placebo. "Five episodes of nonfatal bleeding in a critical area or organ were noted, all in the semuloparin group," the researchers said.
Serious adverse events developed in 26.3% of patients taking semuloparin and 25.5% of those taking placebo, a nonsignificant difference. In particular, there were no cases of heparin-induced thrombocytopenia, and the rates of thrombocytopenia were comparable between the semuloparin and placebo groups (7.1% and 7.6%, respectively).
"Current guidelines recommend antithrombotic prophylaxis for patients with cancer who are admitted to the hospital for medical illness (administered for the duration of the hospital stay) and for patients who have undergone surgery for cancer (extended for up to 5 weeks) but not for routine use in ambulatory patients receiving chemotherapy. This study suggests a benefit from antithrombotic prophylaxis with semuloparin in patients receiving chemotherapy for cancer," Dr. Agnelli and his associates said.
This study was supported by Sanofi, manufacturer of semuloparin. Dr. Agnelli and his associates reported ties to numerous industry sources.
Semuloparin, an ultralow-molecular-weight heparin, reduced the incidence of venous thromboembolic events when given prophylactically to patients with a variety of metastatic or locally advanced solid tumors, according to a report in the Feb. 16 issue of the New England Journal of Medicine.
Semuloparin prevented both deep vein thrombosis and pulmonary embolism (PE) without raising the risk of major bleeding in patients undergoing chemotherapy for lung, pancreatic, stomach, colon, rectal, bladder, or ovarian cancer. It did not improve overall survival, however, said Dr. Giancarlo Agnelli of the department of internal medicine at the University of Perugia (Italy) and his associates.
The investigators assessed the safety and efficacy of semuloparin because this patient population has a high incidence of venous thromboembolism (VTE). They conducted the SAVE-ONCO trial (NCT00694382), which was sponsored by semuloparin’s manufacturer, Sanofi-Aventis, at 395 medical centers in 47 countries.
A total of 3,212 patients were randomly assigned to receive daily subcutaneous injections of either semuloparin (1,608 patients) or placebo (1,604 patients), beginning on the first day of chemotherapy and continuing until chemotherapy was stopped or changed to a different regimen. The concomitant use of antiplatelet agents and nonsteroidal anti-inflammatory drugs (NSAIDs) was permitted at the treating physician’s discretion.
The mean duration of treatment was 3.5 months. The reasons for discontinuing treatment were similar between patients receiving semuloparin and those receiving placebo – chiefly adverse events (15.9% and 17.5%, respectively) and the patient’s request to discontinue treatment (18.1% and 17.4%, respectively).
The primary efficacy outcome – a composite end point of any symptomatic deep vein thrombosis in the lower or upper limbs, any nonfatal pulmonary embolism, or death related to VTE – occurred in 1.2% of the semuloparin group, a significantly lower rate than the 3.4% rate in the placebo group.
Semuloparin significantly reduced the incidence of deep vein thrombosis, fatal PE, and nonfatal PE. This benefit was seen across all groups of patients categorized by cancer stage, cancer site, and geographic region, Dr. Agnelli and his colleagues reported (N. Engl. J. Med. 2012;366:601-9).
The treatment benefit also was consistent across numerous baseline risks for VTE, such as personal or family history of VTE; use of a central venous catheter; obesity; age of 75 years or more; presence of chronic respiratory or heart failure; presence of venous insufficiency or varicose veins; and use of hormone therapy.
However, semuloparin did not improve overall survival. Mortality was 43.4% in the semuloparin group and 44.5% in the placebo group.
The overall rate of major and clinically relevant nonmajor bleeding was 2.8% with semuloparin and 2.0% with placebo, a nonsignificant difference. Two fatal bleeding events occurred with semuloparin and four with placebo. "Five episodes of nonfatal bleeding in a critical area or organ were noted, all in the semuloparin group," the researchers said.
Serious adverse events developed in 26.3% of patients taking semuloparin and 25.5% of those taking placebo, a nonsignificant difference. In particular, there were no cases of heparin-induced thrombocytopenia, and the rates of thrombocytopenia were comparable between the semuloparin and placebo groups (7.1% and 7.6%, respectively).
"Current guidelines recommend antithrombotic prophylaxis for patients with cancer who are admitted to the hospital for medical illness (administered for the duration of the hospital stay) and for patients who have undergone surgery for cancer (extended for up to 5 weeks) but not for routine use in ambulatory patients receiving chemotherapy. This study suggests a benefit from antithrombotic prophylaxis with semuloparin in patients receiving chemotherapy for cancer," Dr. Agnelli and his associates said.
This study was supported by Sanofi, manufacturer of semuloparin. Dr. Agnelli and his associates reported ties to numerous industry sources.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: VTE occurred in 1.2% of cancer patients taking semuloparin, which was significantly lower than the 3.4% rate in the placebo group.
Data Source: SAVE-ONCO was a double-blind, international clinical trial comparing 3 months of daily subcutaneous injections of semuloparin against placebo in 3,212 patients who were undergoing a broad range of chemotherapy regimens for a variety of tumors.
Disclosures: This study was supported by Sanofi, manufacturer of semuloparin. Dr. Agnelli and his associates reported ties to numerous industry sources.
Post-MI Stem Cell Therapy Improves Cardiac Function
Autologous bone marrow–derived intracardiac stem cell therapy after revascularized acute MI "seems to be safe and improves heart function moderately but significantly," according to a review and meta-analysis of all randomized controlled trials of the treatment done through 2011, which was published online Feb. 14 by the Cochrane Collaboration.
This form of intracardiac stem cell therapy improves global left ventricular function and reduces infarct (scar) size – changes that persist in the long term. Nevertheless, because morbidity and mortality are so low in this patient population, there is no evidence yet that this experimental treatment has any significant effect on these outcomes, said Dr. David M. Clifford of the stem cell research lab at the University of Oxford (England), and his associates.
It is hoped that ongoing research will provide the data to answer the crucial question of whether bone-marrow stem-cell therapy actually improves morbidity, mortality, and quality of life, they noted.
Investigators for the Cochrane Collaboration previously published a review of this topic in 2008, and research since then has progressed to the extent that an updated Cochrane Review of safety and efficacy was called for. The previous review evaluated 13 studies, almost all of which had only short-term follow-up. A total of 20 more studies are now available, with follow-ups as long as 5 years.
Dr. Clifford and his colleagues reviewed the literature and performed a meta-analysis of these 33 randomized clinical trials involving 1,765 subjects. These studies were conducted in 17 countries in 2004-2011, and 13 of them are still ongoing.
All the study subjects underwent either PCI, thrombolysis, or both as the primary treatment for MI. Most of the trials compared adjuvant stem cell therapy against no further intervention, but some compared it against an active intervention with placebo infusions.
Pooled results demonstrated that this form of stem-cell therapy significantly improves left ventricular function, reducing left ventricular end-systolic and end-diastolic volumes as well as infarct size and cardiac wall motion. These benefits persisted throughout long-term follow-up.
Moreover, the improvements correlated with the amount of stem cells infused, so that an increasing dose yielded increasing benefit.
"The results of all trials together suggest a small reduction in the incidence of mortality and morbidity, in favor of [stem-cell therapy], but this finding is not statistically significant," the investigators said (Cochrane Database Syst. Rev. 2012 [doi:10.1002/14651858.CD006536.pub3]).
There was no increase in adverse events related to stem cell therapy, except for a few events related to G-CSF stimulation used in two small studies. Overall, "there is no evidence that the treatment is harmful," Dr. Clifford and his associates said.
In summary, intracardiac bone marrow–derived stem cell therapy may yield benefits beyond those obtained with conventional treatments such as PCI. The researchers acknowledged that this conclusion "is perhaps optimistic." But some of the trials did report improved survival, and the 13 ongoing RCTs will address that question and will be included in the next Cochrane Review update, they noted.
The Cochrane Collaboration is a nonprofit international network of health experts. Dr. Clifford and his associates reported no financial conflicts of interest.
Autologous bone marrow–derived intracardiac stem cell therapy after revascularized acute MI "seems to be safe and improves heart function moderately but significantly," according to a review and meta-analysis of all randomized controlled trials of the treatment done through 2011, which was published online Feb. 14 by the Cochrane Collaboration.
This form of intracardiac stem cell therapy improves global left ventricular function and reduces infarct (scar) size – changes that persist in the long term. Nevertheless, because morbidity and mortality are so low in this patient population, there is no evidence yet that this experimental treatment has any significant effect on these outcomes, said Dr. David M. Clifford of the stem cell research lab at the University of Oxford (England), and his associates.
It is hoped that ongoing research will provide the data to answer the crucial question of whether bone-marrow stem-cell therapy actually improves morbidity, mortality, and quality of life, they noted.
Investigators for the Cochrane Collaboration previously published a review of this topic in 2008, and research since then has progressed to the extent that an updated Cochrane Review of safety and efficacy was called for. The previous review evaluated 13 studies, almost all of which had only short-term follow-up. A total of 20 more studies are now available, with follow-ups as long as 5 years.
Dr. Clifford and his colleagues reviewed the literature and performed a meta-analysis of these 33 randomized clinical trials involving 1,765 subjects. These studies were conducted in 17 countries in 2004-2011, and 13 of them are still ongoing.
All the study subjects underwent either PCI, thrombolysis, or both as the primary treatment for MI. Most of the trials compared adjuvant stem cell therapy against no further intervention, but some compared it against an active intervention with placebo infusions.
Pooled results demonstrated that this form of stem-cell therapy significantly improves left ventricular function, reducing left ventricular end-systolic and end-diastolic volumes as well as infarct size and cardiac wall motion. These benefits persisted throughout long-term follow-up.
Moreover, the improvements correlated with the amount of stem cells infused, so that an increasing dose yielded increasing benefit.
"The results of all trials together suggest a small reduction in the incidence of mortality and morbidity, in favor of [stem-cell therapy], but this finding is not statistically significant," the investigators said (Cochrane Database Syst. Rev. 2012 [doi:10.1002/14651858.CD006536.pub3]).
There was no increase in adverse events related to stem cell therapy, except for a few events related to G-CSF stimulation used in two small studies. Overall, "there is no evidence that the treatment is harmful," Dr. Clifford and his associates said.
In summary, intracardiac bone marrow–derived stem cell therapy may yield benefits beyond those obtained with conventional treatments such as PCI. The researchers acknowledged that this conclusion "is perhaps optimistic." But some of the trials did report improved survival, and the 13 ongoing RCTs will address that question and will be included in the next Cochrane Review update, they noted.
The Cochrane Collaboration is a nonprofit international network of health experts. Dr. Clifford and his associates reported no financial conflicts of interest.
Autologous bone marrow–derived intracardiac stem cell therapy after revascularized acute MI "seems to be safe and improves heart function moderately but significantly," according to a review and meta-analysis of all randomized controlled trials of the treatment done through 2011, which was published online Feb. 14 by the Cochrane Collaboration.
This form of intracardiac stem cell therapy improves global left ventricular function and reduces infarct (scar) size – changes that persist in the long term. Nevertheless, because morbidity and mortality are so low in this patient population, there is no evidence yet that this experimental treatment has any significant effect on these outcomes, said Dr. David M. Clifford of the stem cell research lab at the University of Oxford (England), and his associates.
It is hoped that ongoing research will provide the data to answer the crucial question of whether bone-marrow stem-cell therapy actually improves morbidity, mortality, and quality of life, they noted.
Investigators for the Cochrane Collaboration previously published a review of this topic in 2008, and research since then has progressed to the extent that an updated Cochrane Review of safety and efficacy was called for. The previous review evaluated 13 studies, almost all of which had only short-term follow-up. A total of 20 more studies are now available, with follow-ups as long as 5 years.
Dr. Clifford and his colleagues reviewed the literature and performed a meta-analysis of these 33 randomized clinical trials involving 1,765 subjects. These studies were conducted in 17 countries in 2004-2011, and 13 of them are still ongoing.
All the study subjects underwent either PCI, thrombolysis, or both as the primary treatment for MI. Most of the trials compared adjuvant stem cell therapy against no further intervention, but some compared it against an active intervention with placebo infusions.
Pooled results demonstrated that this form of stem-cell therapy significantly improves left ventricular function, reducing left ventricular end-systolic and end-diastolic volumes as well as infarct size and cardiac wall motion. These benefits persisted throughout long-term follow-up.
Moreover, the improvements correlated with the amount of stem cells infused, so that an increasing dose yielded increasing benefit.
"The results of all trials together suggest a small reduction in the incidence of mortality and morbidity, in favor of [stem-cell therapy], but this finding is not statistically significant," the investigators said (Cochrane Database Syst. Rev. 2012 [doi:10.1002/14651858.CD006536.pub3]).
There was no increase in adverse events related to stem cell therapy, except for a few events related to G-CSF stimulation used in two small studies. Overall, "there is no evidence that the treatment is harmful," Dr. Clifford and his associates said.
In summary, intracardiac bone marrow–derived stem cell therapy may yield benefits beyond those obtained with conventional treatments such as PCI. The researchers acknowledged that this conclusion "is perhaps optimistic." But some of the trials did report improved survival, and the 13 ongoing RCTs will address that question and will be included in the next Cochrane Review update, they noted.
The Cochrane Collaboration is a nonprofit international network of health experts. Dr. Clifford and his associates reported no financial conflicts of interest.
FROM A COCHRANE REVIEW
Major Finding: Autologous bone marrow-derived intracardiac stem-cell therapy after acute MI significantly improves left ventricular ejection fraction and reduces infarct size.
Data Source: Researchers conducted a meta-analysis of 33 randomized clinical trials done in 2004-2011 evaluating this form of stem-cell therapy in 1,765 patients.
Disclosures: The Cochrane Collaboration is a nonprofit international network of health experts. Dr. Clifford and his associates reported no financial conflicts.
Amoxicillin No Better Than Placebo for Rhinosinusitis
A 10-day course of amoxicillin "offers little clinical benefit for most patients with clinically diagnosed uncomplicated acute rhinosinusitis," the results of a placebo-controlled trial published Feb. 15 in JAMA have shown.
"Our findings support recommendations to avoid routine antibiotic treatment for patients with uncomplicated acute rhinosinusitis," according to Dr. Jane M. Garbutt and her associates at Washington University, St. Louis.
Their study involved 166 adults with a history of rhinosinusitis symptoms that had not improved, or that had worsened, for 7-28 days. Participants were randomly assigned to receive either 10 days of daily amoxicillin (85 patients) or a matching placebo (81 patients). Those who received amoxicillin reported no significant difference in symptoms at 3 days or at 10 days, compared with patients who received placebo. Some patients in the amoxicillin group were more improved at 7 days, but the difference "was too small to represent any clinically important change," the researchers wrote.
Dr. Garbutt said that this was the first trial of antibiotic therapy for rhinosinusitis that looked at disease-specific quality of life as the primary outcome. The investigators used the modified Sino-Nasal Outcome Test–16 (SNOT-16) questionnaire, which assesses both the severity and the frequency of 16 sinus-related symptoms that have bothered respondents during the past few days.
The patients were representative of those "for whom antibiotics might be prescribed" in routine practice. They were adults reporting maxillary pain or tenderness in the face or teeth and purulent nasal secretions who presented to any of 10 primary care practices in St. Louis.
In addition to amoxicillin or placebo, all patients also used as-needed acetaminophen, guaifenesin, dextromethorphan, pseudoephedrine, and nasal saline spray.
The primary end point was the effect of treatment on disease-specific quality of life at day 3, as measured by responses to the SNOT-16. "We expected any benefit of antibiotic treatment to be evident 48-72 hours after the treatment was begun," the researchers explained.
Secondary end points included the patient’s retrospective assessment of change in sinus symptoms and functional status, recurrence or relapse, and satisfaction with and adverse effects of treatment. These outcomes were assessed by telephone interview at days 3, 7, 10, and 28.
A total of 37% of the amoxicillin group and 34% of the placebo group reported symptom improvement at day 3, a nonsignificant difference. The results were similar between study groups at day 10, with 78% of the amoxicillin group and 80% of the placebo group reporting symptom improvement, the investigators said (JAMA 2012;307:685-92).
The two groups differed only at the assessment on day 7. At that time, significantly more of the amoxicillin group (74%) than of the placebo group (56%) reported symptom improvement, but the difference in scores was deemed clinically insignificant. No factor was found to predict response to amoxicillin at day 7.
When the analysis was restricted only to the 143 patients who completed their 10-day course of study treatment, the results were the same. Amoxicillin also conferred no added benefit for any subgroup of patients such as smokers, those with the longest duration of symptoms, those with a history of sinus infections, or those with asthma or allergic rhinitis.
In addition, secondary outcomes, including number of days missed from work, number of days unable to perform usual activities, rates of relapse and recurrence at 28 days, the use of additional health care services, and patient satisfaction with treatment, also were the same between patients who received amoxicillin and those who received placebo.
There were no serious adverse events, and the rates of any adverse event were the same between the two groups.
These results add to the existing "considerable body of evidence from clinical trials conducted in the primary care setting that antibiotics provide little if any benefit for patients with clinically diagnosed acute rhinosinusitis," Dr. Garbutt and her associates said.
However, "it is important to note that patients with symptoms indicative of serious complications were excluded from this trial and likely need a different management strategy," they added.
This study was supported by the National Institute of Allergy and Infectious Diseases. One of Dr. Garbutt’s associates reported receiving royalties for the Sino-Nasal Outcome Test and reported ties to Apex Medical.
A 10-day course of amoxicillin "offers little clinical benefit for most patients with clinically diagnosed uncomplicated acute rhinosinusitis," the results of a placebo-controlled trial published Feb. 15 in JAMA have shown.
"Our findings support recommendations to avoid routine antibiotic treatment for patients with uncomplicated acute rhinosinusitis," according to Dr. Jane M. Garbutt and her associates at Washington University, St. Louis.
Their study involved 166 adults with a history of rhinosinusitis symptoms that had not improved, or that had worsened, for 7-28 days. Participants were randomly assigned to receive either 10 days of daily amoxicillin (85 patients) or a matching placebo (81 patients). Those who received amoxicillin reported no significant difference in symptoms at 3 days or at 10 days, compared with patients who received placebo. Some patients in the amoxicillin group were more improved at 7 days, but the difference "was too small to represent any clinically important change," the researchers wrote.
Dr. Garbutt said that this was the first trial of antibiotic therapy for rhinosinusitis that looked at disease-specific quality of life as the primary outcome. The investigators used the modified Sino-Nasal Outcome Test–16 (SNOT-16) questionnaire, which assesses both the severity and the frequency of 16 sinus-related symptoms that have bothered respondents during the past few days.
The patients were representative of those "for whom antibiotics might be prescribed" in routine practice. They were adults reporting maxillary pain or tenderness in the face or teeth and purulent nasal secretions who presented to any of 10 primary care practices in St. Louis.
In addition to amoxicillin or placebo, all patients also used as-needed acetaminophen, guaifenesin, dextromethorphan, pseudoephedrine, and nasal saline spray.
The primary end point was the effect of treatment on disease-specific quality of life at day 3, as measured by responses to the SNOT-16. "We expected any benefit of antibiotic treatment to be evident 48-72 hours after the treatment was begun," the researchers explained.
Secondary end points included the patient’s retrospective assessment of change in sinus symptoms and functional status, recurrence or relapse, and satisfaction with and adverse effects of treatment. These outcomes were assessed by telephone interview at days 3, 7, 10, and 28.
A total of 37% of the amoxicillin group and 34% of the placebo group reported symptom improvement at day 3, a nonsignificant difference. The results were similar between study groups at day 10, with 78% of the amoxicillin group and 80% of the placebo group reporting symptom improvement, the investigators said (JAMA 2012;307:685-92).
The two groups differed only at the assessment on day 7. At that time, significantly more of the amoxicillin group (74%) than of the placebo group (56%) reported symptom improvement, but the difference in scores was deemed clinically insignificant. No factor was found to predict response to amoxicillin at day 7.
When the analysis was restricted only to the 143 patients who completed their 10-day course of study treatment, the results were the same. Amoxicillin also conferred no added benefit for any subgroup of patients such as smokers, those with the longest duration of symptoms, those with a history of sinus infections, or those with asthma or allergic rhinitis.
In addition, secondary outcomes, including number of days missed from work, number of days unable to perform usual activities, rates of relapse and recurrence at 28 days, the use of additional health care services, and patient satisfaction with treatment, also were the same between patients who received amoxicillin and those who received placebo.
There were no serious adverse events, and the rates of any adverse event were the same between the two groups.
These results add to the existing "considerable body of evidence from clinical trials conducted in the primary care setting that antibiotics provide little if any benefit for patients with clinically diagnosed acute rhinosinusitis," Dr. Garbutt and her associates said.
However, "it is important to note that patients with symptoms indicative of serious complications were excluded from this trial and likely need a different management strategy," they added.
This study was supported by the National Institute of Allergy and Infectious Diseases. One of Dr. Garbutt’s associates reported receiving royalties for the Sino-Nasal Outcome Test and reported ties to Apex Medical.
A 10-day course of amoxicillin "offers little clinical benefit for most patients with clinically diagnosed uncomplicated acute rhinosinusitis," the results of a placebo-controlled trial published Feb. 15 in JAMA have shown.
"Our findings support recommendations to avoid routine antibiotic treatment for patients with uncomplicated acute rhinosinusitis," according to Dr. Jane M. Garbutt and her associates at Washington University, St. Louis.
Their study involved 166 adults with a history of rhinosinusitis symptoms that had not improved, or that had worsened, for 7-28 days. Participants were randomly assigned to receive either 10 days of daily amoxicillin (85 patients) or a matching placebo (81 patients). Those who received amoxicillin reported no significant difference in symptoms at 3 days or at 10 days, compared with patients who received placebo. Some patients in the amoxicillin group were more improved at 7 days, but the difference "was too small to represent any clinically important change," the researchers wrote.
Dr. Garbutt said that this was the first trial of antibiotic therapy for rhinosinusitis that looked at disease-specific quality of life as the primary outcome. The investigators used the modified Sino-Nasal Outcome Test–16 (SNOT-16) questionnaire, which assesses both the severity and the frequency of 16 sinus-related symptoms that have bothered respondents during the past few days.
The patients were representative of those "for whom antibiotics might be prescribed" in routine practice. They were adults reporting maxillary pain or tenderness in the face or teeth and purulent nasal secretions who presented to any of 10 primary care practices in St. Louis.
In addition to amoxicillin or placebo, all patients also used as-needed acetaminophen, guaifenesin, dextromethorphan, pseudoephedrine, and nasal saline spray.
The primary end point was the effect of treatment on disease-specific quality of life at day 3, as measured by responses to the SNOT-16. "We expected any benefit of antibiotic treatment to be evident 48-72 hours after the treatment was begun," the researchers explained.
Secondary end points included the patient’s retrospective assessment of change in sinus symptoms and functional status, recurrence or relapse, and satisfaction with and adverse effects of treatment. These outcomes were assessed by telephone interview at days 3, 7, 10, and 28.
A total of 37% of the amoxicillin group and 34% of the placebo group reported symptom improvement at day 3, a nonsignificant difference. The results were similar between study groups at day 10, with 78% of the amoxicillin group and 80% of the placebo group reporting symptom improvement, the investigators said (JAMA 2012;307:685-92).
The two groups differed only at the assessment on day 7. At that time, significantly more of the amoxicillin group (74%) than of the placebo group (56%) reported symptom improvement, but the difference in scores was deemed clinically insignificant. No factor was found to predict response to amoxicillin at day 7.
When the analysis was restricted only to the 143 patients who completed their 10-day course of study treatment, the results were the same. Amoxicillin also conferred no added benefit for any subgroup of patients such as smokers, those with the longest duration of symptoms, those with a history of sinus infections, or those with asthma or allergic rhinitis.
In addition, secondary outcomes, including number of days missed from work, number of days unable to perform usual activities, rates of relapse and recurrence at 28 days, the use of additional health care services, and patient satisfaction with treatment, also were the same between patients who received amoxicillin and those who received placebo.
There were no serious adverse events, and the rates of any adverse event were the same between the two groups.
These results add to the existing "considerable body of evidence from clinical trials conducted in the primary care setting that antibiotics provide little if any benefit for patients with clinically diagnosed acute rhinosinusitis," Dr. Garbutt and her associates said.
However, "it is important to note that patients with symptoms indicative of serious complications were excluded from this trial and likely need a different management strategy," they added.
This study was supported by the National Institute of Allergy and Infectious Diseases. One of Dr. Garbutt’s associates reported receiving royalties for the Sino-Nasal Outcome Test and reported ties to Apex Medical.
FROM JAMA
Major Finding: Symptom improvement at 3 days was reported in 37% of the amoxicillin group and 34% of the placebo group. The difference was not significant.
Data Source: A randomized placebo-controlled trial was conducted involving 166 adults with uncomplicated acute rhinosinusitis whose symptoms were assessed every few days.
Disclosures: This study was supported by the National Institute of Allergy and Infectious Diseases. One of Dr. Garbutt’s associates reported receiving royalties for the Sino-Nasal Outcome Test and reported ties to Apex Medical.
Stem Cell Therapy Prompts 'Unprecedented' Myocardial Regeneration After MI
The intracoronary infusion of autologous cardiosphere-derived stem cells soon after MI was found to be safe and feasible in a phase I proof-of-concept study, but – more importantly – the treatment reduced scar tissue and increased viable myocardial tissue, according to a report published online Feb. 14 in Lancet.
This is the first study to demonstrate a "striking" reduction in damaged myocardium and a concomitant "unprecedented" increase in viable myocardium following MI, which indicates myocardial regeneration. The findings show that "therapeutic regeneration might indeed be possible in cardiac tissue" when stem cell therapy is used, said Dr. Raj R. Makkar, director of interventional cardiology at Cedars-Sinai Heart Institute, Los Angeles, and his associates.
"Despite more than a decade of clinical trials of cardiac regenerative therapy, this ambitious goal [has] remained elusive" – until now, they noted.
In the CADUCEUS (Cardiosphere-Derived Autologous Stem Cells to Reverse Ventricular Dysfunction) study, Dr. Makkar and his colleagues at Cedars-Sinai and Johns Hopkins Hospital, Baltimore, tested their stem cell therapy in 31 patients who had a recent (within 30 days) left-sided acute MI, had undergone successful PCI (percutaneous coronary intervention) with stent placement, and had residual mild to moderate presymptomatic left ventricular dysfunction (ejection fraction, 25%-45%).
The study subjects were randomly assigned in a 2:1 ratio to receive either stem cell therapy or routine conventional treatment (the control group). The stem cells were harvested from each patient via percutaneous biopsy of the right ventricular myocardium, and were cultured to yield the large quantities necessary for treatment. They were then infused in three boluses given over 15 minutes in a saline solution containing heparin and nitroglycerin.
The infusion was delivered through an over-the-wire angioplasty catheter, with the balloon inflated at the stented site of the previous blockage in the infarct-related artery. All stem cell infusions were completed within 3 months of the MI.
All the study subjects were scheduled for follow-ups at 2 weeks and at 1, 2, 3, 6, and 12 months following stem cell infusion. At the time of publication, all the study subjects had reached their 6-month assessments and all but four had reached their 12-month assessments.
The primary safety end points of this phase I study were infusion-related death (defined as death due to ventricular tachycardia, ventricular fibrillation, sudden unexpected death, or MI) and, at 6-month follow-up, major adverse cardiac events or the formation of cardiac tumors. No complications occurred within 24 hours of either the biopsy sampling or the stem cell infusion. There were no cases of ventricular fibrillation or sustained ventricular tachycardia. There also were no deaths, no cases of major adverse cardiac events, and no cardiac tumors at either 6-month or 12-month follow-up.
However, one non–Q-wave MI occurred in a single patient 7 months after stem cell infusion, and was deemed "possibly" related to treatment. Adverse events that were considered unrelated to treatment included one MI, two cases of angina, one coronary revascularization procedure, and one insertion of an implantable defibrillator in the stem cell group, as well as one case of atypical chest pain in the control group, after the 6-month mark.
Three patients who had been assigned to the study intervention withdrew or were disqualified from participation. There also were three technical failures. In addition, cardiac MRIs were judged to be technically "uninterpretable" in two patients and were excluded from the analysis.
At 6 months, contrast-enhanced MRI showed that scars were smaller in circumference and thickness in patients who received stem cell infusions, but not in the control subjects. Similarly, the amount of viable myocardium increased in the stem cell patients but not in the control subjects, the investigators said (Lancet 2012 Feb. 14 [doi:10.1016/S0140-6736(12)60195-0]).
When the results were pooled for each group, patients who received stem cell infusions showed an 8% absolute decrease in scar size at 6 months and a 12% absolute decrease at 12 months. Scar size was unchanged in the control group.
Scar mass decreased by 8 g (28%) at 6 months and 13 g (42%) at 12 months in the stem cell patients, but remained unchanged in control subjects. The reductions in scar mass correlated well with increases in viable myocardium at both time points in the stem cell group.
Moreover, "viable mass increased on average about 60% more than scar [mass] shrunk, leading to partial restoration of lost left ventricular mass in patients treated with [stem cells]," Dr. Makkar and his associates said.
Clinically, patients in the stem cell group showed a mean increase in 6-minute walking distance of 11 meters at 6 months and 33 meters at 12 months. In contrast, the control subjects showed a mean increase of 13 meters at 6 months, but a mean decrease of 10 meters at 12 months.
However, scores on the Minnesota Living with Heart Failure Questionnaire decreased to the same degree in the two study groups. And both groups also showed nonsignificant changes in left-ventricular ejection fraction at 6 months.
Both regional contractility and systolic wall thickening improved in the stem cell group but worsened in the control group.
Previous studies have reported small reductions in scar size after infusions of bone marrow mononuclear cells, mesenchymal cells, and another heart-derived cell product, purified c-KIT–positive cells. However, animal studies demonstrate that the cardiosphere-derived cells used in this study outperform all of them "in terms of paracrine potency, antiapoptotic properties, tissue engraftment, and regenerative efficacy," the CADUCEUS researchers said.
"This discovery challenges the conventional wisdom that, once established, cardiac scarring is permanent and that, once lost, healthy heart muscle cannot be restored," they concluded.
CADUCEUS was supported by the National Heart, Lung, and Blood Institute and Cedars-Sinai Heart Stem Cell Center. Dr. Makkar’s associates reported ties to Capricor, a biotech company that develops and commercializes cardiac stem cell treatments.
From the report by Dr. Makkar and colleagues, "we can conclude that cardiosphere-derived cell harvesting and intracoronary delivery are feasible in patients with recent MI, but investigations involving more patients, longer follow-up, and a true placebo arm will be needed to confirm the safety and efficacy" of the treatment, said Dr. Chung-Wah Siu and Dr. Hug-Fat Tse.
The MRI imaging provided striking evidence of cardiac regeneration, with a reduction of 28% in average scar mass at 6 months and 42% at 12 months, they noted.
Dr. Siu and Dr. Tse are in the division of cardiology and the research center of the heart, brain, hormone, and healthy aging at the University of Hong Kong. They declared that they had no financial conflicts of interest (Lancet 2012 Feb. 14 [doi: 10.1016/S0140-6736(12)60236-0]).
From the report by Dr. Makkar and colleagues, "we can conclude that cardiosphere-derived cell harvesting and intracoronary delivery are feasible in patients with recent MI, but investigations involving more patients, longer follow-up, and a true placebo arm will be needed to confirm the safety and efficacy" of the treatment, said Dr. Chung-Wah Siu and Dr. Hug-Fat Tse.
The MRI imaging provided striking evidence of cardiac regeneration, with a reduction of 28% in average scar mass at 6 months and 42% at 12 months, they noted.
Dr. Siu and Dr. Tse are in the division of cardiology and the research center of the heart, brain, hormone, and healthy aging at the University of Hong Kong. They declared that they had no financial conflicts of interest (Lancet 2012 Feb. 14 [doi: 10.1016/S0140-6736(12)60236-0]).
From the report by Dr. Makkar and colleagues, "we can conclude that cardiosphere-derived cell harvesting and intracoronary delivery are feasible in patients with recent MI, but investigations involving more patients, longer follow-up, and a true placebo arm will be needed to confirm the safety and efficacy" of the treatment, said Dr. Chung-Wah Siu and Dr. Hug-Fat Tse.
The MRI imaging provided striking evidence of cardiac regeneration, with a reduction of 28% in average scar mass at 6 months and 42% at 12 months, they noted.
Dr. Siu and Dr. Tse are in the division of cardiology and the research center of the heart, brain, hormone, and healthy aging at the University of Hong Kong. They declared that they had no financial conflicts of interest (Lancet 2012 Feb. 14 [doi: 10.1016/S0140-6736(12)60236-0]).
The intracoronary infusion of autologous cardiosphere-derived stem cells soon after MI was found to be safe and feasible in a phase I proof-of-concept study, but – more importantly – the treatment reduced scar tissue and increased viable myocardial tissue, according to a report published online Feb. 14 in Lancet.
This is the first study to demonstrate a "striking" reduction in damaged myocardium and a concomitant "unprecedented" increase in viable myocardium following MI, which indicates myocardial regeneration. The findings show that "therapeutic regeneration might indeed be possible in cardiac tissue" when stem cell therapy is used, said Dr. Raj R. Makkar, director of interventional cardiology at Cedars-Sinai Heart Institute, Los Angeles, and his associates.
"Despite more than a decade of clinical trials of cardiac regenerative therapy, this ambitious goal [has] remained elusive" – until now, they noted.
In the CADUCEUS (Cardiosphere-Derived Autologous Stem Cells to Reverse Ventricular Dysfunction) study, Dr. Makkar and his colleagues at Cedars-Sinai and Johns Hopkins Hospital, Baltimore, tested their stem cell therapy in 31 patients who had a recent (within 30 days) left-sided acute MI, had undergone successful PCI (percutaneous coronary intervention) with stent placement, and had residual mild to moderate presymptomatic left ventricular dysfunction (ejection fraction, 25%-45%).
The study subjects were randomly assigned in a 2:1 ratio to receive either stem cell therapy or routine conventional treatment (the control group). The stem cells were harvested from each patient via percutaneous biopsy of the right ventricular myocardium, and were cultured to yield the large quantities necessary for treatment. They were then infused in three boluses given over 15 minutes in a saline solution containing heparin and nitroglycerin.
The infusion was delivered through an over-the-wire angioplasty catheter, with the balloon inflated at the stented site of the previous blockage in the infarct-related artery. All stem cell infusions were completed within 3 months of the MI.
All the study subjects were scheduled for follow-ups at 2 weeks and at 1, 2, 3, 6, and 12 months following stem cell infusion. At the time of publication, all the study subjects had reached their 6-month assessments and all but four had reached their 12-month assessments.
The primary safety end points of this phase I study were infusion-related death (defined as death due to ventricular tachycardia, ventricular fibrillation, sudden unexpected death, or MI) and, at 6-month follow-up, major adverse cardiac events or the formation of cardiac tumors. No complications occurred within 24 hours of either the biopsy sampling or the stem cell infusion. There were no cases of ventricular fibrillation or sustained ventricular tachycardia. There also were no deaths, no cases of major adverse cardiac events, and no cardiac tumors at either 6-month or 12-month follow-up.
However, one non–Q-wave MI occurred in a single patient 7 months after stem cell infusion, and was deemed "possibly" related to treatment. Adverse events that were considered unrelated to treatment included one MI, two cases of angina, one coronary revascularization procedure, and one insertion of an implantable defibrillator in the stem cell group, as well as one case of atypical chest pain in the control group, after the 6-month mark.
Three patients who had been assigned to the study intervention withdrew or were disqualified from participation. There also were three technical failures. In addition, cardiac MRIs were judged to be technically "uninterpretable" in two patients and were excluded from the analysis.
At 6 months, contrast-enhanced MRI showed that scars were smaller in circumference and thickness in patients who received stem cell infusions, but not in the control subjects. Similarly, the amount of viable myocardium increased in the stem cell patients but not in the control subjects, the investigators said (Lancet 2012 Feb. 14 [doi:10.1016/S0140-6736(12)60195-0]).
When the results were pooled for each group, patients who received stem cell infusions showed an 8% absolute decrease in scar size at 6 months and a 12% absolute decrease at 12 months. Scar size was unchanged in the control group.
Scar mass decreased by 8 g (28%) at 6 months and 13 g (42%) at 12 months in the stem cell patients, but remained unchanged in control subjects. The reductions in scar mass correlated well with increases in viable myocardium at both time points in the stem cell group.
Moreover, "viable mass increased on average about 60% more than scar [mass] shrunk, leading to partial restoration of lost left ventricular mass in patients treated with [stem cells]," Dr. Makkar and his associates said.
Clinically, patients in the stem cell group showed a mean increase in 6-minute walking distance of 11 meters at 6 months and 33 meters at 12 months. In contrast, the control subjects showed a mean increase of 13 meters at 6 months, but a mean decrease of 10 meters at 12 months.
However, scores on the Minnesota Living with Heart Failure Questionnaire decreased to the same degree in the two study groups. And both groups also showed nonsignificant changes in left-ventricular ejection fraction at 6 months.
Both regional contractility and systolic wall thickening improved in the stem cell group but worsened in the control group.
Previous studies have reported small reductions in scar size after infusions of bone marrow mononuclear cells, mesenchymal cells, and another heart-derived cell product, purified c-KIT–positive cells. However, animal studies demonstrate that the cardiosphere-derived cells used in this study outperform all of them "in terms of paracrine potency, antiapoptotic properties, tissue engraftment, and regenerative efficacy," the CADUCEUS researchers said.
"This discovery challenges the conventional wisdom that, once established, cardiac scarring is permanent and that, once lost, healthy heart muscle cannot be restored," they concluded.
CADUCEUS was supported by the National Heart, Lung, and Blood Institute and Cedars-Sinai Heart Stem Cell Center. Dr. Makkar’s associates reported ties to Capricor, a biotech company that develops and commercializes cardiac stem cell treatments.
The intracoronary infusion of autologous cardiosphere-derived stem cells soon after MI was found to be safe and feasible in a phase I proof-of-concept study, but – more importantly – the treatment reduced scar tissue and increased viable myocardial tissue, according to a report published online Feb. 14 in Lancet.
This is the first study to demonstrate a "striking" reduction in damaged myocardium and a concomitant "unprecedented" increase in viable myocardium following MI, which indicates myocardial regeneration. The findings show that "therapeutic regeneration might indeed be possible in cardiac tissue" when stem cell therapy is used, said Dr. Raj R. Makkar, director of interventional cardiology at Cedars-Sinai Heart Institute, Los Angeles, and his associates.
"Despite more than a decade of clinical trials of cardiac regenerative therapy, this ambitious goal [has] remained elusive" – until now, they noted.
In the CADUCEUS (Cardiosphere-Derived Autologous Stem Cells to Reverse Ventricular Dysfunction) study, Dr. Makkar and his colleagues at Cedars-Sinai and Johns Hopkins Hospital, Baltimore, tested their stem cell therapy in 31 patients who had a recent (within 30 days) left-sided acute MI, had undergone successful PCI (percutaneous coronary intervention) with stent placement, and had residual mild to moderate presymptomatic left ventricular dysfunction (ejection fraction, 25%-45%).
The study subjects were randomly assigned in a 2:1 ratio to receive either stem cell therapy or routine conventional treatment (the control group). The stem cells were harvested from each patient via percutaneous biopsy of the right ventricular myocardium, and were cultured to yield the large quantities necessary for treatment. They were then infused in three boluses given over 15 minutes in a saline solution containing heparin and nitroglycerin.
The infusion was delivered through an over-the-wire angioplasty catheter, with the balloon inflated at the stented site of the previous blockage in the infarct-related artery. All stem cell infusions were completed within 3 months of the MI.
All the study subjects were scheduled for follow-ups at 2 weeks and at 1, 2, 3, 6, and 12 months following stem cell infusion. At the time of publication, all the study subjects had reached their 6-month assessments and all but four had reached their 12-month assessments.
The primary safety end points of this phase I study were infusion-related death (defined as death due to ventricular tachycardia, ventricular fibrillation, sudden unexpected death, or MI) and, at 6-month follow-up, major adverse cardiac events or the formation of cardiac tumors. No complications occurred within 24 hours of either the biopsy sampling or the stem cell infusion. There were no cases of ventricular fibrillation or sustained ventricular tachycardia. There also were no deaths, no cases of major adverse cardiac events, and no cardiac tumors at either 6-month or 12-month follow-up.
However, one non–Q-wave MI occurred in a single patient 7 months after stem cell infusion, and was deemed "possibly" related to treatment. Adverse events that were considered unrelated to treatment included one MI, two cases of angina, one coronary revascularization procedure, and one insertion of an implantable defibrillator in the stem cell group, as well as one case of atypical chest pain in the control group, after the 6-month mark.
Three patients who had been assigned to the study intervention withdrew or were disqualified from participation. There also were three technical failures. In addition, cardiac MRIs were judged to be technically "uninterpretable" in two patients and were excluded from the analysis.
At 6 months, contrast-enhanced MRI showed that scars were smaller in circumference and thickness in patients who received stem cell infusions, but not in the control subjects. Similarly, the amount of viable myocardium increased in the stem cell patients but not in the control subjects, the investigators said (Lancet 2012 Feb. 14 [doi:10.1016/S0140-6736(12)60195-0]).
When the results were pooled for each group, patients who received stem cell infusions showed an 8% absolute decrease in scar size at 6 months and a 12% absolute decrease at 12 months. Scar size was unchanged in the control group.
Scar mass decreased by 8 g (28%) at 6 months and 13 g (42%) at 12 months in the stem cell patients, but remained unchanged in control subjects. The reductions in scar mass correlated well with increases in viable myocardium at both time points in the stem cell group.
Moreover, "viable mass increased on average about 60% more than scar [mass] shrunk, leading to partial restoration of lost left ventricular mass in patients treated with [stem cells]," Dr. Makkar and his associates said.
Clinically, patients in the stem cell group showed a mean increase in 6-minute walking distance of 11 meters at 6 months and 33 meters at 12 months. In contrast, the control subjects showed a mean increase of 13 meters at 6 months, but a mean decrease of 10 meters at 12 months.
However, scores on the Minnesota Living with Heart Failure Questionnaire decreased to the same degree in the two study groups. And both groups also showed nonsignificant changes in left-ventricular ejection fraction at 6 months.
Both regional contractility and systolic wall thickening improved in the stem cell group but worsened in the control group.
Previous studies have reported small reductions in scar size after infusions of bone marrow mononuclear cells, mesenchymal cells, and another heart-derived cell product, purified c-KIT–positive cells. However, animal studies demonstrate that the cardiosphere-derived cells used in this study outperform all of them "in terms of paracrine potency, antiapoptotic properties, tissue engraftment, and regenerative efficacy," the CADUCEUS researchers said.
"This discovery challenges the conventional wisdom that, once established, cardiac scarring is permanent and that, once lost, healthy heart muscle cannot be restored," they concluded.
CADUCEUS was supported by the National Heart, Lung, and Blood Institute and Cedars-Sinai Heart Stem Cell Center. Dr. Makkar’s associates reported ties to Capricor, a biotech company that develops and commercializes cardiac stem cell treatments.
FROM THE LANCET
Major Finding: Patients who received stem cell infusions showed an 8% absolute decrease in myocardial scar size at 6 months and a 12% absolute decrease at 12 months. Scar size was unchanged in the control group.
Data Source: Data were taken from a phase I, proof-of-concept trial in which 17 MI patients who underwent stem cell therapy and 8 who served as controls were assessed at intervals for 1 year using contrast-enhanced cardiac MRI.
Disclosures: This study was supported by the NHLBI and Cedars-Sinai Heart Stem Cell Center. Dr. Makkar’s associates reported ties to Capricor, a biotech company that develops and commercializes cardiac stem cell treatments.
JIA Cancer Rates Not Linked to Medication
Patients with juvenile idiopathic arthritis have an increased rate of malignancy, compared with unaffected children, according to a report published online Feb. 13 in Arthritis & Rheumatism.
The increased cancer incidence, which ranged from 1.4 to 4.5 times higher than that in comparison groups of children with other chronic diseases, is not significantly associated with JIA treatments, including tumor necrosis factor–inhibiting agents, said Dr. Timothy Beukelman of the division of pediatric rheumatology, University of Alabama at Birmingham, and his associates.
The Food and Drug Administration issued a black box warning on TNF inhibitors in 2009, cautioning that there was an increased risk of malignancy (particularly lymphoma) in pediatric patients who used the drugs, based on reports to the Adverse Event Reporting System. But critics argued that the warning was premature because of the limited data on the background rate of malignancy in this patient population.
"Chronic autoimmune inflammatory conditions such as JIA may be associated with an increased risk of malignancy irrespective of specific therapeutic agents. For example, an increased risk of lymphoma has been observed among adults with rheumatoid arthritis, particularly among those with a high burden of inflammatory activity," De. Beukelman and his colleagues noted.
Moreover, most JIA patients who are treated with TNF inhibitors also receive other drugs, including methotrexate, which may themselves contribute to a higher cancer risk.
The investigators assessed the background rate of malignancy in JIA by using the Medicaid Analytic eXtract (MAX) administrative database, hoping that such a large source would contain sufficient numbers to allow study of two rare disorders: JIA and childhood cancer. The MAX database includes the medical and pharmacy records of all low-income children who receive government medical assistance.
The study assessed 7,812 JIA patients who were followed for approximately 2 years, for a total follow-up time of 12, 614 person-years.
For an "internal" comparison, the researchers also assessed the background cancer rate among children in the database who had two other chronic conditions: asthma (652,234 subjects) and attention-deficit/hyperactivity disorder (321,821 subjects). For an "external" comparison, they obtained population-based estimates of cancer rates from the SEER (Surveillance Epidemiology and End Results) database.
The JIA patients’ medication exposures were divided into three categories by drug class: methotrexate or leflunomide; TNF inhibitors (etanercept, infliximab, or adalimumab); and other immunomodulatory agents (abatacept, alefacept, anakinra, azathioprine, cyclophosphamide, cyclosporine, efalizumab, 6-mercaptopurine, mycophenolate mofetil, rituximab, or tacrolimus).
A total of 3,423 JIA patients (44%) had taken methotrexate or leflunomide; 1,484 (19%) had taken TNF inhibitors; 398 (5%) had taken other immunomodulatory agents; and 2,507 (32%) had not taken any of these drugs.
Only 10 cancers were identified in the JIA patients. Six of them (three brain malignancies, one leukemia, one soft tissue cancer, and one gastrointestinal cancer) developed in the children who had not been exposed to any of the drugs. Three malignancies (two leukemias and one soft tissue cancer) developed in children who had taken methotrexate but not TNF inhibitors. And one malignancy (uterine cancer) developed in a child who had taken TNF inhibitors.
Thus, there was no association between TNF inhibitors and cancer, and there were no cases of lymphoma in any of the children who had JIA, the investigators said (Arthritis Rheum. 2012 Feb. 13 [doi:10.1002/art.34348]).
Compared with the study subjects who had asthma or ADHD, the JIA group had an overall cancer rate that was 1.4-4.5 times higher. The findings were similar when the analysis was restricted only to hematologic cancers.
"The SEER external comparator standardized malignancy rates were significantly lower than the ADHD and asthma internal comparator rates," the researchers noted. This means that study subjects with JIA had even higher rates of cancer, regardless of the treatments they received, than would be expected in the general population based on SEER data.
There are several reasons why JIA might be associated with an increased risk of cancer. Rheumatoid arthritis is known to raise the risk of malignancy, particularly lymphoma. And arthritis medications suppress the immune system, although "we also found an increased rate of malignancies among children not treated with systemic immunosuppression."
It is also possible that JIA patients are more carefully screened for cancer because of the extensive contact they have with the health care system. And finally, some malignancies, especially acute leukemias, may initially be misdiagnosed as JIA, Dr. Beukelman and his colleagues said.
This study was supported by the Agency for Healthcare Research and Quality, the FDA, and the National Institutes of Health. No financial conflicts of interest were reported by the researchers.
The findings of Dr. Beukelman and colleagues, together with flaws in the data that lead to the FDA’s black box warning, "suggest that this drastic step may have been premature," said Dr. Karen B. Onel and Dr. Kenan Onel.
The study findings "are at once concerning and reassuring for physicians, parents, and patients." It is reassuring that there appears to be no additional increase in cancer risk associated with frequently used medications, but it is concerning that JIA itself raises the risk of malignancy, "even for children whose disease severity does not mandate treatment with disease-modifying agents or biologic therapies."
Dr. Karen B. Onel is in the department of pediatrics, and Dr. Kenan Onel is on the committee on cancer biology, both at the University of Chicago. They reported no financial conflicts of interest. These remarks were adapted from the editorial accompanying Dr. Beukelman’s report (Arthritis Rheum. 2012 Feb. 13 [doi:10.102/art.34349]).
The findings of Dr. Beukelman and colleagues, together with flaws in the data that lead to the FDA’s black box warning, "suggest that this drastic step may have been premature," said Dr. Karen B. Onel and Dr. Kenan Onel.
The study findings "are at once concerning and reassuring for physicians, parents, and patients." It is reassuring that there appears to be no additional increase in cancer risk associated with frequently used medications, but it is concerning that JIA itself raises the risk of malignancy, "even for children whose disease severity does not mandate treatment with disease-modifying agents or biologic therapies."
Dr. Karen B. Onel is in the department of pediatrics, and Dr. Kenan Onel is on the committee on cancer biology, both at the University of Chicago. They reported no financial conflicts of interest. These remarks were adapted from the editorial accompanying Dr. Beukelman’s report (Arthritis Rheum. 2012 Feb. 13 [doi:10.102/art.34349]).
The findings of Dr. Beukelman and colleagues, together with flaws in the data that lead to the FDA’s black box warning, "suggest that this drastic step may have been premature," said Dr. Karen B. Onel and Dr. Kenan Onel.
The study findings "are at once concerning and reassuring for physicians, parents, and patients." It is reassuring that there appears to be no additional increase in cancer risk associated with frequently used medications, but it is concerning that JIA itself raises the risk of malignancy, "even for children whose disease severity does not mandate treatment with disease-modifying agents or biologic therapies."
Dr. Karen B. Onel is in the department of pediatrics, and Dr. Kenan Onel is on the committee on cancer biology, both at the University of Chicago. They reported no financial conflicts of interest. These remarks were adapted from the editorial accompanying Dr. Beukelman’s report (Arthritis Rheum. 2012 Feb. 13 [doi:10.102/art.34349]).
Patients with juvenile idiopathic arthritis have an increased rate of malignancy, compared with unaffected children, according to a report published online Feb. 13 in Arthritis & Rheumatism.
The increased cancer incidence, which ranged from 1.4 to 4.5 times higher than that in comparison groups of children with other chronic diseases, is not significantly associated with JIA treatments, including tumor necrosis factor–inhibiting agents, said Dr. Timothy Beukelman of the division of pediatric rheumatology, University of Alabama at Birmingham, and his associates.
The Food and Drug Administration issued a black box warning on TNF inhibitors in 2009, cautioning that there was an increased risk of malignancy (particularly lymphoma) in pediatric patients who used the drugs, based on reports to the Adverse Event Reporting System. But critics argued that the warning was premature because of the limited data on the background rate of malignancy in this patient population.
"Chronic autoimmune inflammatory conditions such as JIA may be associated with an increased risk of malignancy irrespective of specific therapeutic agents. For example, an increased risk of lymphoma has been observed among adults with rheumatoid arthritis, particularly among those with a high burden of inflammatory activity," De. Beukelman and his colleagues noted.
Moreover, most JIA patients who are treated with TNF inhibitors also receive other drugs, including methotrexate, which may themselves contribute to a higher cancer risk.
The investigators assessed the background rate of malignancy in JIA by using the Medicaid Analytic eXtract (MAX) administrative database, hoping that such a large source would contain sufficient numbers to allow study of two rare disorders: JIA and childhood cancer. The MAX database includes the medical and pharmacy records of all low-income children who receive government medical assistance.
The study assessed 7,812 JIA patients who were followed for approximately 2 years, for a total follow-up time of 12, 614 person-years.
For an "internal" comparison, the researchers also assessed the background cancer rate among children in the database who had two other chronic conditions: asthma (652,234 subjects) and attention-deficit/hyperactivity disorder (321,821 subjects). For an "external" comparison, they obtained population-based estimates of cancer rates from the SEER (Surveillance Epidemiology and End Results) database.
The JIA patients’ medication exposures were divided into three categories by drug class: methotrexate or leflunomide; TNF inhibitors (etanercept, infliximab, or adalimumab); and other immunomodulatory agents (abatacept, alefacept, anakinra, azathioprine, cyclophosphamide, cyclosporine, efalizumab, 6-mercaptopurine, mycophenolate mofetil, rituximab, or tacrolimus).
A total of 3,423 JIA patients (44%) had taken methotrexate or leflunomide; 1,484 (19%) had taken TNF inhibitors; 398 (5%) had taken other immunomodulatory agents; and 2,507 (32%) had not taken any of these drugs.
Only 10 cancers were identified in the JIA patients. Six of them (three brain malignancies, one leukemia, one soft tissue cancer, and one gastrointestinal cancer) developed in the children who had not been exposed to any of the drugs. Three malignancies (two leukemias and one soft tissue cancer) developed in children who had taken methotrexate but not TNF inhibitors. And one malignancy (uterine cancer) developed in a child who had taken TNF inhibitors.
Thus, there was no association between TNF inhibitors and cancer, and there were no cases of lymphoma in any of the children who had JIA, the investigators said (Arthritis Rheum. 2012 Feb. 13 [doi:10.1002/art.34348]).
Compared with the study subjects who had asthma or ADHD, the JIA group had an overall cancer rate that was 1.4-4.5 times higher. The findings were similar when the analysis was restricted only to hematologic cancers.
"The SEER external comparator standardized malignancy rates were significantly lower than the ADHD and asthma internal comparator rates," the researchers noted. This means that study subjects with JIA had even higher rates of cancer, regardless of the treatments they received, than would be expected in the general population based on SEER data.
There are several reasons why JIA might be associated with an increased risk of cancer. Rheumatoid arthritis is known to raise the risk of malignancy, particularly lymphoma. And arthritis medications suppress the immune system, although "we also found an increased rate of malignancies among children not treated with systemic immunosuppression."
It is also possible that JIA patients are more carefully screened for cancer because of the extensive contact they have with the health care system. And finally, some malignancies, especially acute leukemias, may initially be misdiagnosed as JIA, Dr. Beukelman and his colleagues said.
This study was supported by the Agency for Healthcare Research and Quality, the FDA, and the National Institutes of Health. No financial conflicts of interest were reported by the researchers.
Patients with juvenile idiopathic arthritis have an increased rate of malignancy, compared with unaffected children, according to a report published online Feb. 13 in Arthritis & Rheumatism.
The increased cancer incidence, which ranged from 1.4 to 4.5 times higher than that in comparison groups of children with other chronic diseases, is not significantly associated with JIA treatments, including tumor necrosis factor–inhibiting agents, said Dr. Timothy Beukelman of the division of pediatric rheumatology, University of Alabama at Birmingham, and his associates.
The Food and Drug Administration issued a black box warning on TNF inhibitors in 2009, cautioning that there was an increased risk of malignancy (particularly lymphoma) in pediatric patients who used the drugs, based on reports to the Adverse Event Reporting System. But critics argued that the warning was premature because of the limited data on the background rate of malignancy in this patient population.
"Chronic autoimmune inflammatory conditions such as JIA may be associated with an increased risk of malignancy irrespective of specific therapeutic agents. For example, an increased risk of lymphoma has been observed among adults with rheumatoid arthritis, particularly among those with a high burden of inflammatory activity," De. Beukelman and his colleagues noted.
Moreover, most JIA patients who are treated with TNF inhibitors also receive other drugs, including methotrexate, which may themselves contribute to a higher cancer risk.
The investigators assessed the background rate of malignancy in JIA by using the Medicaid Analytic eXtract (MAX) administrative database, hoping that such a large source would contain sufficient numbers to allow study of two rare disorders: JIA and childhood cancer. The MAX database includes the medical and pharmacy records of all low-income children who receive government medical assistance.
The study assessed 7,812 JIA patients who were followed for approximately 2 years, for a total follow-up time of 12, 614 person-years.
For an "internal" comparison, the researchers also assessed the background cancer rate among children in the database who had two other chronic conditions: asthma (652,234 subjects) and attention-deficit/hyperactivity disorder (321,821 subjects). For an "external" comparison, they obtained population-based estimates of cancer rates from the SEER (Surveillance Epidemiology and End Results) database.
The JIA patients’ medication exposures were divided into three categories by drug class: methotrexate or leflunomide; TNF inhibitors (etanercept, infliximab, or adalimumab); and other immunomodulatory agents (abatacept, alefacept, anakinra, azathioprine, cyclophosphamide, cyclosporine, efalizumab, 6-mercaptopurine, mycophenolate mofetil, rituximab, or tacrolimus).
A total of 3,423 JIA patients (44%) had taken methotrexate or leflunomide; 1,484 (19%) had taken TNF inhibitors; 398 (5%) had taken other immunomodulatory agents; and 2,507 (32%) had not taken any of these drugs.
Only 10 cancers were identified in the JIA patients. Six of them (three brain malignancies, one leukemia, one soft tissue cancer, and one gastrointestinal cancer) developed in the children who had not been exposed to any of the drugs. Three malignancies (two leukemias and one soft tissue cancer) developed in children who had taken methotrexate but not TNF inhibitors. And one malignancy (uterine cancer) developed in a child who had taken TNF inhibitors.
Thus, there was no association between TNF inhibitors and cancer, and there were no cases of lymphoma in any of the children who had JIA, the investigators said (Arthritis Rheum. 2012 Feb. 13 [doi:10.1002/art.34348]).
Compared with the study subjects who had asthma or ADHD, the JIA group had an overall cancer rate that was 1.4-4.5 times higher. The findings were similar when the analysis was restricted only to hematologic cancers.
"The SEER external comparator standardized malignancy rates were significantly lower than the ADHD and asthma internal comparator rates," the researchers noted. This means that study subjects with JIA had even higher rates of cancer, regardless of the treatments they received, than would be expected in the general population based on SEER data.
There are several reasons why JIA might be associated with an increased risk of cancer. Rheumatoid arthritis is known to raise the risk of malignancy, particularly lymphoma. And arthritis medications suppress the immune system, although "we also found an increased rate of malignancies among children not treated with systemic immunosuppression."
It is also possible that JIA patients are more carefully screened for cancer because of the extensive contact they have with the health care system. And finally, some malignancies, especially acute leukemias, may initially be misdiagnosed as JIA, Dr. Beukelman and his colleagues said.
This study was supported by the Agency for Healthcare Research and Quality, the FDA, and the National Institutes of Health. No financial conflicts of interest were reported by the researchers.
FROM ARTHRITIS & RHEUMATISM
Major Finding: The cancer rate was 1.4 to 4.5 times higher among children with JIA than among those with other chronic diseases, regardless of the types of medications the JIA patients received.
Data Source: Data were taken from an analysis of cancer incidence among 7,812 JIA patients, 652,234 pediatric asthma patients, and 321,821 pediatric ADHD patients in a Medicaid database.
Disclosures: This study was supported by the AHRQ, the FDA, and the NIH. No financial conflicts of interest were reported.
Brand Names Drove Differences in Part D Spending
Most of the wide geographic variation in Medicare spending for prescription drugs is due to the cost of the drugs selected rather than to the volume of drugs used in each region, according to a report in the Feb. 9 issue of the New England Journal of Medicine.
The choice of brand-name drugs rather than generics accounts for approximately 76% of this variation. If the geographic areas with the highest drug spending had used generics as often as those with the lowest drug spending in 2008, Medicare and its beneficiaries would have saved $4.5 billion that year, said Julie M. Donohue, Ph.D., of the University of Pittsburgh and her associates.
A considerable variation in prescription-drug spending was reported in 2010, but the reasons for that variation were not identified. Some speculated that costs were higher in regions with the most patients or the most seriously ill patients than regions with fewer patients or less seriously ill patients.
Dr. Donohue and her colleagues analyzed data on 4,666,866 Part D beneficiaries in 2008. They assessed spending for three classes of drugs commonly used by the elderly for which there are no over-the-counter substitutes and which together account for nearly one-fourth of all drug spending annually: ACE inhibitors and ARBs, statins, and newer antidepressants (SSRIs and SNRIs).
After adjustment for demographic, socioeconomic, and health-status differences among the 306 geographic regions studied, per capita prescription-drug spending varied by 25% from the area with the lowest drug spending ($2,413 per patient) to that with the highest ($3,008 per patient).
Overall, most (76%) of the regional variation was ascribed to the cost per prescription filled, and the remaining 24% was ascribed to differences in volume. However this pattern differed among the three drug categories studied, the investigators said (N. Engl. J. Med. 2012;366:530-8).
For ACE inhibitors and ARBs, 90% of the variation in costs was due to the use of brand-name ARBs, which had no generic equivalents during the study period. For statins, 56% of the variation was due to the use of brand-name rather than generic drugs.
But for antidepressants, only about 36% of the variation was due to the use of brand-name rather than generic drugs. Most of the variation instead was due to wide differences in the volume of SSRIs and SNRIs used in various areas of the country. The highest-use areas filled nearly 30% more prescriptions per capita for these antidepressants than did the lowest-use areas.
Some areas of the country consistently used a high percentage of brand-name drugs, while others consistently used a high percentage of generics. For example, in Miami, patients almost always were prescribed brand-name ACE inhibitors, ARBs, statins, SSRIs, and SNRIs, while patients in Rochester, Minn., rarely were prescribed brand-name drugs in any of these categories.
If the regions in the highest quintile of prescription-drug spending had used the same number of generics as those in the lowest quintile of spending, "we estimate that overall Part D drug spending would have been 10% lower in 2008." This translates into a nationwide savings of $4.5 billion just for Medicare and its beneficiaries, Dr. Donohue and her associates noted.
This study was supported by the National Institute on Aging, the Agency for Healthcare Research and Quality, the National Institute of Nursing Research, the National Institute of Mental Health, the Robert Wood Johnson Foundation, and the Veterans Affairs Health Services Research and Development Service. No relevant financial conflicts of interest were reported.
Most of the wide geographic variation in Medicare spending for prescription drugs is due to the cost of the drugs selected rather than to the volume of drugs used in each region, according to a report in the Feb. 9 issue of the New England Journal of Medicine.
The choice of brand-name drugs rather than generics accounts for approximately 76% of this variation. If the geographic areas with the highest drug spending had used generics as often as those with the lowest drug spending in 2008, Medicare and its beneficiaries would have saved $4.5 billion that year, said Julie M. Donohue, Ph.D., of the University of Pittsburgh and her associates.
A considerable variation in prescription-drug spending was reported in 2010, but the reasons for that variation were not identified. Some speculated that costs were higher in regions with the most patients or the most seriously ill patients than regions with fewer patients or less seriously ill patients.
Dr. Donohue and her colleagues analyzed data on 4,666,866 Part D beneficiaries in 2008. They assessed spending for three classes of drugs commonly used by the elderly for which there are no over-the-counter substitutes and which together account for nearly one-fourth of all drug spending annually: ACE inhibitors and ARBs, statins, and newer antidepressants (SSRIs and SNRIs).
After adjustment for demographic, socioeconomic, and health-status differences among the 306 geographic regions studied, per capita prescription-drug spending varied by 25% from the area with the lowest drug spending ($2,413 per patient) to that with the highest ($3,008 per patient).
Overall, most (76%) of the regional variation was ascribed to the cost per prescription filled, and the remaining 24% was ascribed to differences in volume. However this pattern differed among the three drug categories studied, the investigators said (N. Engl. J. Med. 2012;366:530-8).
For ACE inhibitors and ARBs, 90% of the variation in costs was due to the use of brand-name ARBs, which had no generic equivalents during the study period. For statins, 56% of the variation was due to the use of brand-name rather than generic drugs.
But for antidepressants, only about 36% of the variation was due to the use of brand-name rather than generic drugs. Most of the variation instead was due to wide differences in the volume of SSRIs and SNRIs used in various areas of the country. The highest-use areas filled nearly 30% more prescriptions per capita for these antidepressants than did the lowest-use areas.
Some areas of the country consistently used a high percentage of brand-name drugs, while others consistently used a high percentage of generics. For example, in Miami, patients almost always were prescribed brand-name ACE inhibitors, ARBs, statins, SSRIs, and SNRIs, while patients in Rochester, Minn., rarely were prescribed brand-name drugs in any of these categories.
If the regions in the highest quintile of prescription-drug spending had used the same number of generics as those in the lowest quintile of spending, "we estimate that overall Part D drug spending would have been 10% lower in 2008." This translates into a nationwide savings of $4.5 billion just for Medicare and its beneficiaries, Dr. Donohue and her associates noted.
This study was supported by the National Institute on Aging, the Agency for Healthcare Research and Quality, the National Institute of Nursing Research, the National Institute of Mental Health, the Robert Wood Johnson Foundation, and the Veterans Affairs Health Services Research and Development Service. No relevant financial conflicts of interest were reported.
Most of the wide geographic variation in Medicare spending for prescription drugs is due to the cost of the drugs selected rather than to the volume of drugs used in each region, according to a report in the Feb. 9 issue of the New England Journal of Medicine.
The choice of brand-name drugs rather than generics accounts for approximately 76% of this variation. If the geographic areas with the highest drug spending had used generics as often as those with the lowest drug spending in 2008, Medicare and its beneficiaries would have saved $4.5 billion that year, said Julie M. Donohue, Ph.D., of the University of Pittsburgh and her associates.
A considerable variation in prescription-drug spending was reported in 2010, but the reasons for that variation were not identified. Some speculated that costs were higher in regions with the most patients or the most seriously ill patients than regions with fewer patients or less seriously ill patients.
Dr. Donohue and her colleagues analyzed data on 4,666,866 Part D beneficiaries in 2008. They assessed spending for three classes of drugs commonly used by the elderly for which there are no over-the-counter substitutes and which together account for nearly one-fourth of all drug spending annually: ACE inhibitors and ARBs, statins, and newer antidepressants (SSRIs and SNRIs).
After adjustment for demographic, socioeconomic, and health-status differences among the 306 geographic regions studied, per capita prescription-drug spending varied by 25% from the area with the lowest drug spending ($2,413 per patient) to that with the highest ($3,008 per patient).
Overall, most (76%) of the regional variation was ascribed to the cost per prescription filled, and the remaining 24% was ascribed to differences in volume. However this pattern differed among the three drug categories studied, the investigators said (N. Engl. J. Med. 2012;366:530-8).
For ACE inhibitors and ARBs, 90% of the variation in costs was due to the use of brand-name ARBs, which had no generic equivalents during the study period. For statins, 56% of the variation was due to the use of brand-name rather than generic drugs.
But for antidepressants, only about 36% of the variation was due to the use of brand-name rather than generic drugs. Most of the variation instead was due to wide differences in the volume of SSRIs and SNRIs used in various areas of the country. The highest-use areas filled nearly 30% more prescriptions per capita for these antidepressants than did the lowest-use areas.
Some areas of the country consistently used a high percentage of brand-name drugs, while others consistently used a high percentage of generics. For example, in Miami, patients almost always were prescribed brand-name ACE inhibitors, ARBs, statins, SSRIs, and SNRIs, while patients in Rochester, Minn., rarely were prescribed brand-name drugs in any of these categories.
If the regions in the highest quintile of prescription-drug spending had used the same number of generics as those in the lowest quintile of spending, "we estimate that overall Part D drug spending would have been 10% lower in 2008." This translates into a nationwide savings of $4.5 billion just for Medicare and its beneficiaries, Dr. Donohue and her associates noted.
This study was supported by the National Institute on Aging, the Agency for Healthcare Research and Quality, the National Institute of Nursing Research, the National Institute of Mental Health, the Robert Wood Johnson Foundation, and the Veterans Affairs Health Services Research and Development Service. No relevant financial conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Seventy-six percent of the regional variation in Medicare Part D prescription-drug spending was due to the cost of the drugs, which in turn was due to the use of brand-name rather than generic drugs.
Data Source: Analysis of 2008 data on prescription-drug use among 4.6 million Part D beneficiaries.
Disclosures: This study was supported by the National Institutes of Health, the Agency for Healthcare Research and Quality, the Robert Wood Johnson Foundation, and the Department of Veterans Affairs. No relevant financial conflicts of interest were reported.
Tai Chi Improved Postural Stability in Parkinson's Disease
Tai chi improved postural stability and other functional outcomes in patients with mild to moderate Parkinson’s disease to a significantly greater extent than did resistance training over the course of a 6-month period in a randomized, controlled trial.
The number of falls among patients who performed tai chi also significantly declined, making the clinical trial the first to demonstrate the efficacy of an exercise intervention in decreasing falls among Parkinson’s patients, Fuzhong Li, Ph.D., of the Oregon Research Institute, Eugene, and his associates reported in the Feb. 9 issue of the New England Journal of Medicine.
Tai chi is a balance-based exercise that has been demonstrated to improve strength and physical function in the elderly, as well as to improve the axial symptoms of Parkinson’s disease (PD) in two pilot studies.
Dr. Li and his colleagues enrolled 195 participants, aged 40-85 years, in four Oregon cities for 6 months of twice-weekly 1-hour tai chi classes, resistance-training classes, or low-impact stretching classes. They had Hoehn and Yahr scale scores of 2-4 out of a possible 5, indicating mild to moderate PD severity.
Sixty-five study subjects were randomly assigned to the tai chi program, which was deliberately designed to tax balance and gait. It stressed "weight shifting and ankle sway to effectively move the person’s center of gravity toward the limits of stability, alternating between a narrow stance and a wide stance to continually change the base of support, increasing support-leg standing time and trailing-leg swing time, engaging rotational trunk movements with upright posture, and performing heel-to-toe (forward) and toe-to-heel (backward) stepping movements to strengthen dorsiflexion and plantar flexion."
Another 65 subjects were assigned to resistance training, in which gradually increasing ankle weights and weighted vests were worn while the patients performed forward and side steps, squats, forward and side lunges, and heel and toe raises. These exercises focused on strengthening the muscles that are important for posture, balance, and gait.
The final 65 subjects were assigned to a variety of seated and standing stretches involving gentle trunk rotation and joint extension and flexion in the neck, upper back, shoulders, arms, quadriceps, hamstrings, calves, and hips. This was considered a control condition and was intended to provide similar social interaction as the other interventions without the training benefits.
The primary outcome measure was improvement in two indicators of postural stability, maximum excursion and directional control, which were measured using computerized dynamic posturography at baseline, 3 months, 6 months (the conclusion of the intervention), and 3 months after the end of the intervention. "Maximum excursion is an assessment of the limits of self-initiated movements as patients shift or lean their center of gravity, without falling, toward the theoretical limit (100%) in each of eight target directions. Directional control, a measure of movement accuracy, is calculated by comparing the amount of movement toward the target with the amount of extraneous movement," the investigators explained.
After the 6-month intervention, the tai chi group performed significantly better in both of those measures than did the other two groups. Between baseline and 6 months, the tai chi group improved by an average of about 10 percentage points on maximum excursion and 8 percentage points on directional control, while the strength-training group improved by 4 percentage points on maximum excursion but did not improve significantly on directional control. The stretching group showed no improvement in either measure, the investigators said (N. Engl. J. Med. 2012;366:511-9).
The tai chi group also performed significantly better than did the other two study groups in the secondary outcomes of gait (stride length and walking velocity); strength of knee extensors and flexors; reach (the maximal distance the patient could reach forward beyond arm’s length while maintaining a fixed standing position); and mobility (the time taken to rise from a chair, walk 3 meters, return, and sit down).
These results suggest that tai chi "reduced dyskinesia by increasing the ability of the participants to adopt effective sway strategies (at the ankle or hip), engage in controlled movements with improved balance control near the limit of stability, or both," according to the investigators. In practical terms, patients could more effectively perform the tasks of daily living, such as reaching forward to take items from a cabinet, rising from a chair, and walking.
Perhaps as important, the rate of falls during the study period was lower in the tai chi group (0.22/subject-month) than in the resistance training group (0.51) and in the stretching group (0.62). There were a total of 62 falls among 19 participants in the tai chi group, 133 among 31 participants in the resistance group, and 186 among 26 participants in the stretching group.
At a follow-up visit 3 months after the intervention was over, the patients in the tai chi group maintained these gains in stability and mobility, and continued to have fewer falls than did those in the other two groups.
"No serious adverse events were observed during tai chi training, indicating the safety and usefulness of this intervention for persons with Parkinson’s disease," Dr. Li and his associates wrote.
This study was supported by the National Institute of Neurological Disorders and Stroke. The investigators had no financial conflicts of interest to report.
Tai chi improved postural stability and other functional outcomes in patients with mild to moderate Parkinson’s disease to a significantly greater extent than did resistance training over the course of a 6-month period in a randomized, controlled trial.
The number of falls among patients who performed tai chi also significantly declined, making the clinical trial the first to demonstrate the efficacy of an exercise intervention in decreasing falls among Parkinson’s patients, Fuzhong Li, Ph.D., of the Oregon Research Institute, Eugene, and his associates reported in the Feb. 9 issue of the New England Journal of Medicine.
Tai chi is a balance-based exercise that has been demonstrated to improve strength and physical function in the elderly, as well as to improve the axial symptoms of Parkinson’s disease (PD) in two pilot studies.
Dr. Li and his colleagues enrolled 195 participants, aged 40-85 years, in four Oregon cities for 6 months of twice-weekly 1-hour tai chi classes, resistance-training classes, or low-impact stretching classes. They had Hoehn and Yahr scale scores of 2-4 out of a possible 5, indicating mild to moderate PD severity.
Sixty-five study subjects were randomly assigned to the tai chi program, which was deliberately designed to tax balance and gait. It stressed "weight shifting and ankle sway to effectively move the person’s center of gravity toward the limits of stability, alternating between a narrow stance and a wide stance to continually change the base of support, increasing support-leg standing time and trailing-leg swing time, engaging rotational trunk movements with upright posture, and performing heel-to-toe (forward) and toe-to-heel (backward) stepping movements to strengthen dorsiflexion and plantar flexion."
Another 65 subjects were assigned to resistance training, in which gradually increasing ankle weights and weighted vests were worn while the patients performed forward and side steps, squats, forward and side lunges, and heel and toe raises. These exercises focused on strengthening the muscles that are important for posture, balance, and gait.
The final 65 subjects were assigned to a variety of seated and standing stretches involving gentle trunk rotation and joint extension and flexion in the neck, upper back, shoulders, arms, quadriceps, hamstrings, calves, and hips. This was considered a control condition and was intended to provide similar social interaction as the other interventions without the training benefits.
The primary outcome measure was improvement in two indicators of postural stability, maximum excursion and directional control, which were measured using computerized dynamic posturography at baseline, 3 months, 6 months (the conclusion of the intervention), and 3 months after the end of the intervention. "Maximum excursion is an assessment of the limits of self-initiated movements as patients shift or lean their center of gravity, without falling, toward the theoretical limit (100%) in each of eight target directions. Directional control, a measure of movement accuracy, is calculated by comparing the amount of movement toward the target with the amount of extraneous movement," the investigators explained.
After the 6-month intervention, the tai chi group performed significantly better in both of those measures than did the other two groups. Between baseline and 6 months, the tai chi group improved by an average of about 10 percentage points on maximum excursion and 8 percentage points on directional control, while the strength-training group improved by 4 percentage points on maximum excursion but did not improve significantly on directional control. The stretching group showed no improvement in either measure, the investigators said (N. Engl. J. Med. 2012;366:511-9).
The tai chi group also performed significantly better than did the other two study groups in the secondary outcomes of gait (stride length and walking velocity); strength of knee extensors and flexors; reach (the maximal distance the patient could reach forward beyond arm’s length while maintaining a fixed standing position); and mobility (the time taken to rise from a chair, walk 3 meters, return, and sit down).
These results suggest that tai chi "reduced dyskinesia by increasing the ability of the participants to adopt effective sway strategies (at the ankle or hip), engage in controlled movements with improved balance control near the limit of stability, or both," according to the investigators. In practical terms, patients could more effectively perform the tasks of daily living, such as reaching forward to take items from a cabinet, rising from a chair, and walking.
Perhaps as important, the rate of falls during the study period was lower in the tai chi group (0.22/subject-month) than in the resistance training group (0.51) and in the stretching group (0.62). There were a total of 62 falls among 19 participants in the tai chi group, 133 among 31 participants in the resistance group, and 186 among 26 participants in the stretching group.
At a follow-up visit 3 months after the intervention was over, the patients in the tai chi group maintained these gains in stability and mobility, and continued to have fewer falls than did those in the other two groups.
"No serious adverse events were observed during tai chi training, indicating the safety and usefulness of this intervention for persons with Parkinson’s disease," Dr. Li and his associates wrote.
This study was supported by the National Institute of Neurological Disorders and Stroke. The investigators had no financial conflicts of interest to report.
Tai chi improved postural stability and other functional outcomes in patients with mild to moderate Parkinson’s disease to a significantly greater extent than did resistance training over the course of a 6-month period in a randomized, controlled trial.
The number of falls among patients who performed tai chi also significantly declined, making the clinical trial the first to demonstrate the efficacy of an exercise intervention in decreasing falls among Parkinson’s patients, Fuzhong Li, Ph.D., of the Oregon Research Institute, Eugene, and his associates reported in the Feb. 9 issue of the New England Journal of Medicine.
Tai chi is a balance-based exercise that has been demonstrated to improve strength and physical function in the elderly, as well as to improve the axial symptoms of Parkinson’s disease (PD) in two pilot studies.
Dr. Li and his colleagues enrolled 195 participants, aged 40-85 years, in four Oregon cities for 6 months of twice-weekly 1-hour tai chi classes, resistance-training classes, or low-impact stretching classes. They had Hoehn and Yahr scale scores of 2-4 out of a possible 5, indicating mild to moderate PD severity.
Sixty-five study subjects were randomly assigned to the tai chi program, which was deliberately designed to tax balance and gait. It stressed "weight shifting and ankle sway to effectively move the person’s center of gravity toward the limits of stability, alternating between a narrow stance and a wide stance to continually change the base of support, increasing support-leg standing time and trailing-leg swing time, engaging rotational trunk movements with upright posture, and performing heel-to-toe (forward) and toe-to-heel (backward) stepping movements to strengthen dorsiflexion and plantar flexion."
Another 65 subjects were assigned to resistance training, in which gradually increasing ankle weights and weighted vests were worn while the patients performed forward and side steps, squats, forward and side lunges, and heel and toe raises. These exercises focused on strengthening the muscles that are important for posture, balance, and gait.
The final 65 subjects were assigned to a variety of seated and standing stretches involving gentle trunk rotation and joint extension and flexion in the neck, upper back, shoulders, arms, quadriceps, hamstrings, calves, and hips. This was considered a control condition and was intended to provide similar social interaction as the other interventions without the training benefits.
The primary outcome measure was improvement in two indicators of postural stability, maximum excursion and directional control, which were measured using computerized dynamic posturography at baseline, 3 months, 6 months (the conclusion of the intervention), and 3 months after the end of the intervention. "Maximum excursion is an assessment of the limits of self-initiated movements as patients shift or lean their center of gravity, without falling, toward the theoretical limit (100%) in each of eight target directions. Directional control, a measure of movement accuracy, is calculated by comparing the amount of movement toward the target with the amount of extraneous movement," the investigators explained.
After the 6-month intervention, the tai chi group performed significantly better in both of those measures than did the other two groups. Between baseline and 6 months, the tai chi group improved by an average of about 10 percentage points on maximum excursion and 8 percentage points on directional control, while the strength-training group improved by 4 percentage points on maximum excursion but did not improve significantly on directional control. The stretching group showed no improvement in either measure, the investigators said (N. Engl. J. Med. 2012;366:511-9).
The tai chi group also performed significantly better than did the other two study groups in the secondary outcomes of gait (stride length and walking velocity); strength of knee extensors and flexors; reach (the maximal distance the patient could reach forward beyond arm’s length while maintaining a fixed standing position); and mobility (the time taken to rise from a chair, walk 3 meters, return, and sit down).
These results suggest that tai chi "reduced dyskinesia by increasing the ability of the participants to adopt effective sway strategies (at the ankle or hip), engage in controlled movements with improved balance control near the limit of stability, or both," according to the investigators. In practical terms, patients could more effectively perform the tasks of daily living, such as reaching forward to take items from a cabinet, rising from a chair, and walking.
Perhaps as important, the rate of falls during the study period was lower in the tai chi group (0.22/subject-month) than in the resistance training group (0.51) and in the stretching group (0.62). There were a total of 62 falls among 19 participants in the tai chi group, 133 among 31 participants in the resistance group, and 186 among 26 participants in the stretching group.
At a follow-up visit 3 months after the intervention was over, the patients in the tai chi group maintained these gains in stability and mobility, and continued to have fewer falls than did those in the other two groups.
"No serious adverse events were observed during tai chi training, indicating the safety and usefulness of this intervention for persons with Parkinson’s disease," Dr. Li and his associates wrote.
This study was supported by the National Institute of Neurological Disorders and Stroke. The investigators had no financial conflicts of interest to report.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major Finding: Patients who practiced tai chi improved two measures of postural stability significantly more than did those who performed resistance training – by about 6 percentage points for maximum excursion and by about 10 percentage points for directional control.
Data Source: A randomized controlled trial involving 195 patients with mildly to moderately severe PD who participated in one of three activity programs for 6 months.
Disclosures: This study was supported by the National Institute of Neurological Disorders and Stroke. The investigators had no financial conflicts of interest to report.
Rotavirus: No Increased Intussusception Risk in Infants Given RV5
After nearly 800,000 doses of the pentavalent rotavirus vaccine have been given in the United States, infants who received them showed no increase in the risk of developing intussusception during the week or month following immunization, according to a report in the Feb. 8 issue of JAMA.
Moreover, no rise in the overall rate of intussusception occurred after introduction of the RV5 (RotaTeq, Merck) vaccine in 2006, compared with the rate in prevaccine years.
However, severe rotavirus disease has shown large declines, with an estimated decrease of 55,000 rotavirus hospitalizations in 1 year alone. "Thus, the known benefits of rotavirus vaccination in the U.S. outweigh any potential low-level risk for intussusception that might exist," said Irene M. Shui, Sc.D., of the department of epidemiology, Harvard School of Public Health, Boston, and her associates.
Two recent reports from overseas have raised concerns about intussusception risk associated with the rotavirus vaccine. An Australian study found a nearly fivefold rise in risk during the week after the first dose of RV5. And another study found a fivefold increase in risk during that week in Mexico but no such increase in Brazil. Previous studies have identified the 30-day period following vaccination as another "window of risk."
"The known benefits of rotavirus vaccination in the U.S. outweigh any potential low-level risk for intussusception that might exist."
In what they described as "the largest prospective study to date to assess the association of RV5 and intussusception," Dr. Shui and her colleagues assessed postlicensure safety of the immunization using data from the Vaccine Safety Datalink population. The VSD is a collaborative project between the Centers for Disease Control and Prevention (CDC) and several managed care organizations containing detailed medical data on member-patients.
For this study, rates of intussusception were analyzed among infants aged 4-34 weeks who received the vaccine in 2006-2010, an interval in which 786,725 doses of RV5 were given. These were compared with rates of intussusception among infants who received other recommended vaccines but not RV5 during the same period (389,026 office visits) and with an estimate of the expected number of intussusception visits based on background rates in 2001-2005.
A total of 56 cases of intussusception were identified among the vaccinated infants – 30 after receiving RV5 and 26 after receiving other immunizations, a difference that was not significant. Cases of intussusception showed no increase during either the 7-day "risk window" after vaccination or the 30-day "risk window."
These results remained robust in several sensitivity analyses of the data, the researchers said (JAMA 2012;307:598-604).
In the historical comparison, 589 cases of intussusception were identified, for an overall incidence of 47 per 100,000 person-years. "No clear trend in intussusception rates was noted after the implementation of rotavirus vaccination in 2006," said Dr. Shui, who is also in the department of population medicine at Harvard University, and her associates.
Again, cases of intussusception showed no increase during either the 7-day "risk window" or the 30-day "risk window" after vaccination.
The study findings "are especially important given that rotavirus vaccine coverage in the U.S. has steadily increased since its introduction and averaged 72% in June 2009 among 5-month-olds selected from 8 different sentinel sites across the country," the investigators noted.
It remains unclear why this U.S. experience and the experience reported from Brazil differ so markedly from those reported in Australia and Mexico. "Because intussusception is a rare event, we cannot rule out a chance finding of risk in Australia and Mexico as well as the possibility of not detecting a low-level risk in the United States and Brazil," they said.
It’s also possible that some environmental or genetic factor that differs among these populations could be a contributor, such as differences in infant diet, breast-feeding practices, or maternal antibodies.
This study was supported in part by the Vaccine Safety Datalink contract with America’s Health Insurance Plan (AHIP), funded by the CDC. Dr. Shui reported ties to AHIP, and her associates reported ties to MedImmune, Merck, GlaxoSmithKline, Pfizer, Sanofi Pasteur, and Novartis.
After nearly 800,000 doses of the pentavalent rotavirus vaccine have been given in the United States, infants who received them showed no increase in the risk of developing intussusception during the week or month following immunization, according to a report in the Feb. 8 issue of JAMA.
Moreover, no rise in the overall rate of intussusception occurred after introduction of the RV5 (RotaTeq, Merck) vaccine in 2006, compared with the rate in prevaccine years.
However, severe rotavirus disease has shown large declines, with an estimated decrease of 55,000 rotavirus hospitalizations in 1 year alone. "Thus, the known benefits of rotavirus vaccination in the U.S. outweigh any potential low-level risk for intussusception that might exist," said Irene M. Shui, Sc.D., of the department of epidemiology, Harvard School of Public Health, Boston, and her associates.
Two recent reports from overseas have raised concerns about intussusception risk associated with the rotavirus vaccine. An Australian study found a nearly fivefold rise in risk during the week after the first dose of RV5. And another study found a fivefold increase in risk during that week in Mexico but no such increase in Brazil. Previous studies have identified the 30-day period following vaccination as another "window of risk."
"The known benefits of rotavirus vaccination in the U.S. outweigh any potential low-level risk for intussusception that might exist."
In what they described as "the largest prospective study to date to assess the association of RV5 and intussusception," Dr. Shui and her colleagues assessed postlicensure safety of the immunization using data from the Vaccine Safety Datalink population. The VSD is a collaborative project between the Centers for Disease Control and Prevention (CDC) and several managed care organizations containing detailed medical data on member-patients.
For this study, rates of intussusception were analyzed among infants aged 4-34 weeks who received the vaccine in 2006-2010, an interval in which 786,725 doses of RV5 were given. These were compared with rates of intussusception among infants who received other recommended vaccines but not RV5 during the same period (389,026 office visits) and with an estimate of the expected number of intussusception visits based on background rates in 2001-2005.
A total of 56 cases of intussusception were identified among the vaccinated infants – 30 after receiving RV5 and 26 after receiving other immunizations, a difference that was not significant. Cases of intussusception showed no increase during either the 7-day "risk window" after vaccination or the 30-day "risk window."
These results remained robust in several sensitivity analyses of the data, the researchers said (JAMA 2012;307:598-604).
In the historical comparison, 589 cases of intussusception were identified, for an overall incidence of 47 per 100,000 person-years. "No clear trend in intussusception rates was noted after the implementation of rotavirus vaccination in 2006," said Dr. Shui, who is also in the department of population medicine at Harvard University, and her associates.
Again, cases of intussusception showed no increase during either the 7-day "risk window" or the 30-day "risk window" after vaccination.
The study findings "are especially important given that rotavirus vaccine coverage in the U.S. has steadily increased since its introduction and averaged 72% in June 2009 among 5-month-olds selected from 8 different sentinel sites across the country," the investigators noted.
It remains unclear why this U.S. experience and the experience reported from Brazil differ so markedly from those reported in Australia and Mexico. "Because intussusception is a rare event, we cannot rule out a chance finding of risk in Australia and Mexico as well as the possibility of not detecting a low-level risk in the United States and Brazil," they said.
It’s also possible that some environmental or genetic factor that differs among these populations could be a contributor, such as differences in infant diet, breast-feeding practices, or maternal antibodies.
This study was supported in part by the Vaccine Safety Datalink contract with America’s Health Insurance Plan (AHIP), funded by the CDC. Dr. Shui reported ties to AHIP, and her associates reported ties to MedImmune, Merck, GlaxoSmithKline, Pfizer, Sanofi Pasteur, and Novartis.
After nearly 800,000 doses of the pentavalent rotavirus vaccine have been given in the United States, infants who received them showed no increase in the risk of developing intussusception during the week or month following immunization, according to a report in the Feb. 8 issue of JAMA.
Moreover, no rise in the overall rate of intussusception occurred after introduction of the RV5 (RotaTeq, Merck) vaccine in 2006, compared with the rate in prevaccine years.
However, severe rotavirus disease has shown large declines, with an estimated decrease of 55,000 rotavirus hospitalizations in 1 year alone. "Thus, the known benefits of rotavirus vaccination in the U.S. outweigh any potential low-level risk for intussusception that might exist," said Irene M. Shui, Sc.D., of the department of epidemiology, Harvard School of Public Health, Boston, and her associates.
Two recent reports from overseas have raised concerns about intussusception risk associated with the rotavirus vaccine. An Australian study found a nearly fivefold rise in risk during the week after the first dose of RV5. And another study found a fivefold increase in risk during that week in Mexico but no such increase in Brazil. Previous studies have identified the 30-day period following vaccination as another "window of risk."
"The known benefits of rotavirus vaccination in the U.S. outweigh any potential low-level risk for intussusception that might exist."
In what they described as "the largest prospective study to date to assess the association of RV5 and intussusception," Dr. Shui and her colleagues assessed postlicensure safety of the immunization using data from the Vaccine Safety Datalink population. The VSD is a collaborative project between the Centers for Disease Control and Prevention (CDC) and several managed care organizations containing detailed medical data on member-patients.
For this study, rates of intussusception were analyzed among infants aged 4-34 weeks who received the vaccine in 2006-2010, an interval in which 786,725 doses of RV5 were given. These were compared with rates of intussusception among infants who received other recommended vaccines but not RV5 during the same period (389,026 office visits) and with an estimate of the expected number of intussusception visits based on background rates in 2001-2005.
A total of 56 cases of intussusception were identified among the vaccinated infants – 30 after receiving RV5 and 26 after receiving other immunizations, a difference that was not significant. Cases of intussusception showed no increase during either the 7-day "risk window" after vaccination or the 30-day "risk window."
These results remained robust in several sensitivity analyses of the data, the researchers said (JAMA 2012;307:598-604).
In the historical comparison, 589 cases of intussusception were identified, for an overall incidence of 47 per 100,000 person-years. "No clear trend in intussusception rates was noted after the implementation of rotavirus vaccination in 2006," said Dr. Shui, who is also in the department of population medicine at Harvard University, and her associates.
Again, cases of intussusception showed no increase during either the 7-day "risk window" or the 30-day "risk window" after vaccination.
The study findings "are especially important given that rotavirus vaccine coverage in the U.S. has steadily increased since its introduction and averaged 72% in June 2009 among 5-month-olds selected from 8 different sentinel sites across the country," the investigators noted.
It remains unclear why this U.S. experience and the experience reported from Brazil differ so markedly from those reported in Australia and Mexico. "Because intussusception is a rare event, we cannot rule out a chance finding of risk in Australia and Mexico as well as the possibility of not detecting a low-level risk in the United States and Brazil," they said.
It’s also possible that some environmental or genetic factor that differs among these populations could be a contributor, such as differences in infant diet, breast-feeding practices, or maternal antibodies.
This study was supported in part by the Vaccine Safety Datalink contract with America’s Health Insurance Plan (AHIP), funded by the CDC. Dr. Shui reported ties to AHIP, and her associates reported ties to MedImmune, Merck, GlaxoSmithKline, Pfizer, Sanofi Pasteur, and Novartis.
FROM JAMA
Major Finding: There was no significant difference in the rate of intussusception between U.S. infants who received the RV5 vaccine in 2006-2010 (30 cases) and those who did not (26 cases), nor has there been any increase in cases in the years since the RV5 vaccine was introduced.
Data Source: A prospective cohort study comparing rates of intussusception among infants who received the RV5 vaccine in 2006-2010, infants who received other vaccines but not the RV5, and the estimated rate that would be expected during the study period based on historical data.
Disclosures: This study was supported in part by the Vaccine Safety Datalink contract with America’s Health Insurance Plan (AHIP), funded by the CDC. Dr. Shui reported ties to AHIP, and her associates reported ties to MedImmune, Merck, GlaxoSmithKline, Pfizer, Sanofi Pasteur, and Novartis.
Breast Cancer Mortality Rises with Age in Older Women
Among postmenopausal women who have hormone receptor–positive breast cancer, increasing age correlates with a rise in disease-specific mortality, independently of tumor, treatment, and patient characteristics, according to a report in the Feb. 9 issue of JAMA.
This increased cancer-specific mortality is particularly noteworthy because it occurs against a background in which other, competing causes of mortality are also increasing as these women age, said Dr. Willemien van de Water of the departments of surgical oncology and gerontology, Leiden (the Netherlands) University Medical Center, and associates.
To assess the relationship between aging and breast-cancer-specific mortality, the investigators analyzed data from the TEAM (Tamoxifen Exemestane Adjuvant Multinational) study, a phase III, randomized, open-label trial that had no upper age limit and included subjects up to 96 years old. The TEAM study involved 9,766 women who had estrogen receptor–positive tumors, progesterone receptor–positive tumors, or both, and were treated in Belgium, the Netherlands, the United Kingdom, Ireland, the United States, Japan, Greece, Germany, and France.
Because the 5-year TEAM trial found no significant differences in outcomes between the study groups and comparable mortality from other causes, it proved ideal for this post hoc analysis of disease-specific mortality, Dr. van de Water and colleagues said.
They divided the study subjects into three age groups: younger than 65 years (5,349 women; 55% of the entire cohort), 65-74 years (3,060 women; 31% of the cohort), and 75 years and older (1,357 women; 14% of the cohort).
The cumulative incidence of death from breast cancer rose from 5.7% in the youngest group to 6.3% in the intermediate group and 8.3% in the oldest group, the investigators said (JAMA 2012;307:590-7).
"Since tumor and treatment characteristics may be associated with disease-specific mortality, multivariable analyses were performed in an attempt to adjust for unequal distributions among age categories. ... Again, disease-specific mortality increased with age," with hazard ratios of 1.25 for patients aged 65-74 years and 1.62 for those aged 75 and older, compared with women younger than 65.
"To test the robustness of the age cut points, additional analyses were performed with age as a continuous variable, which confirmed an increased risk of breast cancer death per 10-year increase in age," they said.
Because there was a tendency for older patients to have larger tumors at diagnosis than did younger patients, additional analyses were performed to adjust for any residual confounding that might be related to tumor size. The results were unchanged.
Finally, the data were adjusted to account for mortality from competing causes such as infection, trauma, dementia, and cardiovascular disease. Again, the results were not affected by study subjects’ comorbidities.
This study was not designed to assess the reasons why breast-cancer mortality rises with increasing patient age, but the researchers proposed four possible underlying mechanisms.
First, older patients may be undertreated. Several other studies have shown that older age at diagnosis correlates with greater deviation from treatment guidelines for surgery, radiotherapy, chemotherapy, and endocrine therapy, as well as with more frequent cessation of medication.
Next, older patients may be more vulnerable to treatment-related toxicities. Third, tumor biology might be more aggressive in older patients. And fourth, polypharmacy, which is more common in older patients, might cause adverse interactions with anticancer therapies, rendering them less effective, Dr. van de Water and associates said.
The study findings "underline the need for age-specific breast cancer studies in order to improve breast cancer outcome in patients of all ages," they said.
The TEAM trial was supported by Pfizer. Dr. van de Water's associates reported ties to numerous industry sources.
TEAM study, Tamoxifen Exemestane Adjuvant Multinational, estrogen receptor–positive tumors, progesterone receptor–positive tumors, breast cancer,
Among postmenopausal women who have hormone receptor–positive breast cancer, increasing age correlates with a rise in disease-specific mortality, independently of tumor, treatment, and patient characteristics, according to a report in the Feb. 9 issue of JAMA.
This increased cancer-specific mortality is particularly noteworthy because it occurs against a background in which other, competing causes of mortality are also increasing as these women age, said Dr. Willemien van de Water of the departments of surgical oncology and gerontology, Leiden (the Netherlands) University Medical Center, and associates.
To assess the relationship between aging and breast-cancer-specific mortality, the investigators analyzed data from the TEAM (Tamoxifen Exemestane Adjuvant Multinational) study, a phase III, randomized, open-label trial that had no upper age limit and included subjects up to 96 years old. The TEAM study involved 9,766 women who had estrogen receptor–positive tumors, progesterone receptor–positive tumors, or both, and were treated in Belgium, the Netherlands, the United Kingdom, Ireland, the United States, Japan, Greece, Germany, and France.
Because the 5-year TEAM trial found no significant differences in outcomes between the study groups and comparable mortality from other causes, it proved ideal for this post hoc analysis of disease-specific mortality, Dr. van de Water and colleagues said.
They divided the study subjects into three age groups: younger than 65 years (5,349 women; 55% of the entire cohort), 65-74 years (3,060 women; 31% of the cohort), and 75 years and older (1,357 women; 14% of the cohort).
The cumulative incidence of death from breast cancer rose from 5.7% in the youngest group to 6.3% in the intermediate group and 8.3% in the oldest group, the investigators said (JAMA 2012;307:590-7).
"Since tumor and treatment characteristics may be associated with disease-specific mortality, multivariable analyses were performed in an attempt to adjust for unequal distributions among age categories. ... Again, disease-specific mortality increased with age," with hazard ratios of 1.25 for patients aged 65-74 years and 1.62 for those aged 75 and older, compared with women younger than 65.
"To test the robustness of the age cut points, additional analyses were performed with age as a continuous variable, which confirmed an increased risk of breast cancer death per 10-year increase in age," they said.
Because there was a tendency for older patients to have larger tumors at diagnosis than did younger patients, additional analyses were performed to adjust for any residual confounding that might be related to tumor size. The results were unchanged.
Finally, the data were adjusted to account for mortality from competing causes such as infection, trauma, dementia, and cardiovascular disease. Again, the results were not affected by study subjects’ comorbidities.
This study was not designed to assess the reasons why breast-cancer mortality rises with increasing patient age, but the researchers proposed four possible underlying mechanisms.
First, older patients may be undertreated. Several other studies have shown that older age at diagnosis correlates with greater deviation from treatment guidelines for surgery, radiotherapy, chemotherapy, and endocrine therapy, as well as with more frequent cessation of medication.
Next, older patients may be more vulnerable to treatment-related toxicities. Third, tumor biology might be more aggressive in older patients. And fourth, polypharmacy, which is more common in older patients, might cause adverse interactions with anticancer therapies, rendering them less effective, Dr. van de Water and associates said.
The study findings "underline the need for age-specific breast cancer studies in order to improve breast cancer outcome in patients of all ages," they said.
The TEAM trial was supported by Pfizer. Dr. van de Water's associates reported ties to numerous industry sources.
Among postmenopausal women who have hormone receptor–positive breast cancer, increasing age correlates with a rise in disease-specific mortality, independently of tumor, treatment, and patient characteristics, according to a report in the Feb. 9 issue of JAMA.
This increased cancer-specific mortality is particularly noteworthy because it occurs against a background in which other, competing causes of mortality are also increasing as these women age, said Dr. Willemien van de Water of the departments of surgical oncology and gerontology, Leiden (the Netherlands) University Medical Center, and associates.
To assess the relationship between aging and breast-cancer-specific mortality, the investigators analyzed data from the TEAM (Tamoxifen Exemestane Adjuvant Multinational) study, a phase III, randomized, open-label trial that had no upper age limit and included subjects up to 96 years old. The TEAM study involved 9,766 women who had estrogen receptor–positive tumors, progesterone receptor–positive tumors, or both, and were treated in Belgium, the Netherlands, the United Kingdom, Ireland, the United States, Japan, Greece, Germany, and France.
Because the 5-year TEAM trial found no significant differences in outcomes between the study groups and comparable mortality from other causes, it proved ideal for this post hoc analysis of disease-specific mortality, Dr. van de Water and colleagues said.
They divided the study subjects into three age groups: younger than 65 years (5,349 women; 55% of the entire cohort), 65-74 years (3,060 women; 31% of the cohort), and 75 years and older (1,357 women; 14% of the cohort).
The cumulative incidence of death from breast cancer rose from 5.7% in the youngest group to 6.3% in the intermediate group and 8.3% in the oldest group, the investigators said (JAMA 2012;307:590-7).
"Since tumor and treatment characteristics may be associated with disease-specific mortality, multivariable analyses were performed in an attempt to adjust for unequal distributions among age categories. ... Again, disease-specific mortality increased with age," with hazard ratios of 1.25 for patients aged 65-74 years and 1.62 for those aged 75 and older, compared with women younger than 65.
"To test the robustness of the age cut points, additional analyses were performed with age as a continuous variable, which confirmed an increased risk of breast cancer death per 10-year increase in age," they said.
Because there was a tendency for older patients to have larger tumors at diagnosis than did younger patients, additional analyses were performed to adjust for any residual confounding that might be related to tumor size. The results were unchanged.
Finally, the data were adjusted to account for mortality from competing causes such as infection, trauma, dementia, and cardiovascular disease. Again, the results were not affected by study subjects’ comorbidities.
This study was not designed to assess the reasons why breast-cancer mortality rises with increasing patient age, but the researchers proposed four possible underlying mechanisms.
First, older patients may be undertreated. Several other studies have shown that older age at diagnosis correlates with greater deviation from treatment guidelines for surgery, radiotherapy, chemotherapy, and endocrine therapy, as well as with more frequent cessation of medication.
Next, older patients may be more vulnerable to treatment-related toxicities. Third, tumor biology might be more aggressive in older patients. And fourth, polypharmacy, which is more common in older patients, might cause adverse interactions with anticancer therapies, rendering them less effective, Dr. van de Water and associates said.
The study findings "underline the need for age-specific breast cancer studies in order to improve breast cancer outcome in patients of all ages," they said.
The TEAM trial was supported by Pfizer. Dr. van de Water's associates reported ties to numerous industry sources.
TEAM study, Tamoxifen Exemestane Adjuvant Multinational, estrogen receptor–positive tumors, progesterone receptor–positive tumors, breast cancer,
TEAM study, Tamoxifen Exemestane Adjuvant Multinational, estrogen receptor–positive tumors, progesterone receptor–positive tumors, breast cancer,
FROM JAMA
Major Finding: The rate of death from breast cancer rose from 5.7% in women younger than 65, to 6.3% in women aged 65-74, and to 8.3% in women aged 75 and older.
Data Source: This was a post hoc analysis of data from the TEAM trial, an international phase III study involving 9,766 women aged 35-96 years who had HR-positive breast cancer and were followed for 5 years.
Disclosures: The TEAM trial was supported by Pfizer. Dr. van de Water's associates reported ties to numerous industry sources.