User login
Flu vaccine linked to lower cardiovascular risk
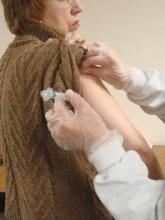
Use of the flu vaccine was consistently associated with a lower risk of adverse cardiovascular events in a meta-analysis of the worldwide medical literature, according to a report published online Oct. 23 in JAMA.
The risk reduction was greatest among people at highest cardiovascular risk, said Dr. Jacob A. Udell of Women’s College Hospital, University of Toronto, and his associates.
"Our findings provide some support for current guideline recommendations for influenza vaccination of patients with acute coronary syndromes," they noted.
In particular, the finding that a simple, once-yearly injection may prevent scores of cardiovascular deaths, hospitalizations, MIs, strokes, and cases of heart failure, urgent coronary revascularization, and unstable angina also is of "considerable clinical and health policy importance, given the profound underuse of vaccination among the general public and the potential impact this preventive strategy may have on high-risk patients," the investigators added.
Several observational studies have suggested an association between acute respiratory infection and increased risk of cardiac and cerebrovascular events, and some have suggested that such infection may trigger rupturing of atherosclerotic plaque, fluid-overload heart failure, myocarditis, or arrhythmia. But whether or not influenza immunization could actually prevent such events remains controversial.
Dr. Udell and his colleagues performed a systematic review of the literature from the inception of database entry (1946) to the present, identifying 71 potentially relevant studies to review. They then performed a meta-analysis of the 12 randomized clinical trials in which influenza vaccination was compared against either placebo or standard care, and in which cardiovascular outcomes during the year following vaccination were reported.
Five of the 12 randomized clinical trials were considered to be of high quality and the remainder were of low or uncertain quality. The meta-analysis included 6,469 participants (mean age, 67 years) who had varying degrees of cardiovascular risk.
"Despite differences in trial designs, risk of bias, sample size, cardiovascular risk of participants, circulating influenza activity, vaccination strategy, duration of follow-up, and number of observed events, our meta-analysis demonstrated a consistent association between influenza vaccination and a lower risk of CV events," the investigators said.
The overall rate of the primary end point, a composite of all major adverse cardiovascular events, was 2.9% among recipients of the influenza vaccine (95 of 3,238 patients). This was significantly lower than the 4.7% rate (151 of 3,231 patients) among control subjects.
The number needed to treat to prevent a single major adverse CV event was 58, Dr. Udell and his associates said (JAMA 2013 Oct. 23 [doi:10.1001/jama.2013.279206]).
In a subgroup analysis involving patients known to have coronary artery disease, influenza vaccination was even more protective. For example, the rate of major adverse CV events was 10.25% among vaccinated patients with a history of recent acute coronary syndrome, compared with a rate of 23.1% among control subjects. The number needed to treat to prevent a single CV event in this subset of patients was only 8.
Less than one-third of the general population in North America and less than half of those at high cardiovascular risk currently receive the annual flu vaccine. Therefore, the flu vaccine "could address a sizable component of residual CV risk not addressed by current therapy, and provide yearlong coverage through a simple inoculation," the researchers said.
"Future research with an adequately powered multicenter trial to confirm the efficacy of this low-cost, annual, safe, easily administered, and well-tolerated therapy to reduce CV risk beyond current therapies is warranted."
The Canadian Institutes for Health Research and the Canadian Foundation for Women’s Health supported the study. Dr. Udell reported no financial conflicts of interest; his associates reported numerous ties to industry sources.
Use of the flu vaccine was consistently associated with a lower risk of adverse cardiovascular events in a meta-analysis of the worldwide medical literature, according to a report published online Oct. 23 in JAMA.
The risk reduction was greatest among people at highest cardiovascular risk, said Dr. Jacob A. Udell of Women’s College Hospital, University of Toronto, and his associates.
"Our findings provide some support for current guideline recommendations for influenza vaccination of patients with acute coronary syndromes," they noted.
In particular, the finding that a simple, once-yearly injection may prevent scores of cardiovascular deaths, hospitalizations, MIs, strokes, and cases of heart failure, urgent coronary revascularization, and unstable angina also is of "considerable clinical and health policy importance, given the profound underuse of vaccination among the general public and the potential impact this preventive strategy may have on high-risk patients," the investigators added.
Several observational studies have suggested an association between acute respiratory infection and increased risk of cardiac and cerebrovascular events, and some have suggested that such infection may trigger rupturing of atherosclerotic plaque, fluid-overload heart failure, myocarditis, or arrhythmia. But whether or not influenza immunization could actually prevent such events remains controversial.
Dr. Udell and his colleagues performed a systematic review of the literature from the inception of database entry (1946) to the present, identifying 71 potentially relevant studies to review. They then performed a meta-analysis of the 12 randomized clinical trials in which influenza vaccination was compared against either placebo or standard care, and in which cardiovascular outcomes during the year following vaccination were reported.
Five of the 12 randomized clinical trials were considered to be of high quality and the remainder were of low or uncertain quality. The meta-analysis included 6,469 participants (mean age, 67 years) who had varying degrees of cardiovascular risk.
"Despite differences in trial designs, risk of bias, sample size, cardiovascular risk of participants, circulating influenza activity, vaccination strategy, duration of follow-up, and number of observed events, our meta-analysis demonstrated a consistent association between influenza vaccination and a lower risk of CV events," the investigators said.
The overall rate of the primary end point, a composite of all major adverse cardiovascular events, was 2.9% among recipients of the influenza vaccine (95 of 3,238 patients). This was significantly lower than the 4.7% rate (151 of 3,231 patients) among control subjects.
The number needed to treat to prevent a single major adverse CV event was 58, Dr. Udell and his associates said (JAMA 2013 Oct. 23 [doi:10.1001/jama.2013.279206]).
In a subgroup analysis involving patients known to have coronary artery disease, influenza vaccination was even more protective. For example, the rate of major adverse CV events was 10.25% among vaccinated patients with a history of recent acute coronary syndrome, compared with a rate of 23.1% among control subjects. The number needed to treat to prevent a single CV event in this subset of patients was only 8.
Less than one-third of the general population in North America and less than half of those at high cardiovascular risk currently receive the annual flu vaccine. Therefore, the flu vaccine "could address a sizable component of residual CV risk not addressed by current therapy, and provide yearlong coverage through a simple inoculation," the researchers said.
"Future research with an adequately powered multicenter trial to confirm the efficacy of this low-cost, annual, safe, easily administered, and well-tolerated therapy to reduce CV risk beyond current therapies is warranted."
The Canadian Institutes for Health Research and the Canadian Foundation for Women’s Health supported the study. Dr. Udell reported no financial conflicts of interest; his associates reported numerous ties to industry sources.
Use of the flu vaccine was consistently associated with a lower risk of adverse cardiovascular events in a meta-analysis of the worldwide medical literature, according to a report published online Oct. 23 in JAMA.
The risk reduction was greatest among people at highest cardiovascular risk, said Dr. Jacob A. Udell of Women’s College Hospital, University of Toronto, and his associates.
"Our findings provide some support for current guideline recommendations for influenza vaccination of patients with acute coronary syndromes," they noted.
In particular, the finding that a simple, once-yearly injection may prevent scores of cardiovascular deaths, hospitalizations, MIs, strokes, and cases of heart failure, urgent coronary revascularization, and unstable angina also is of "considerable clinical and health policy importance, given the profound underuse of vaccination among the general public and the potential impact this preventive strategy may have on high-risk patients," the investigators added.
Several observational studies have suggested an association between acute respiratory infection and increased risk of cardiac and cerebrovascular events, and some have suggested that such infection may trigger rupturing of atherosclerotic plaque, fluid-overload heart failure, myocarditis, or arrhythmia. But whether or not influenza immunization could actually prevent such events remains controversial.
Dr. Udell and his colleagues performed a systematic review of the literature from the inception of database entry (1946) to the present, identifying 71 potentially relevant studies to review. They then performed a meta-analysis of the 12 randomized clinical trials in which influenza vaccination was compared against either placebo or standard care, and in which cardiovascular outcomes during the year following vaccination were reported.
Five of the 12 randomized clinical trials were considered to be of high quality and the remainder were of low or uncertain quality. The meta-analysis included 6,469 participants (mean age, 67 years) who had varying degrees of cardiovascular risk.
"Despite differences in trial designs, risk of bias, sample size, cardiovascular risk of participants, circulating influenza activity, vaccination strategy, duration of follow-up, and number of observed events, our meta-analysis demonstrated a consistent association between influenza vaccination and a lower risk of CV events," the investigators said.
The overall rate of the primary end point, a composite of all major adverse cardiovascular events, was 2.9% among recipients of the influenza vaccine (95 of 3,238 patients). This was significantly lower than the 4.7% rate (151 of 3,231 patients) among control subjects.
The number needed to treat to prevent a single major adverse CV event was 58, Dr. Udell and his associates said (JAMA 2013 Oct. 23 [doi:10.1001/jama.2013.279206]).
In a subgroup analysis involving patients known to have coronary artery disease, influenza vaccination was even more protective. For example, the rate of major adverse CV events was 10.25% among vaccinated patients with a history of recent acute coronary syndrome, compared with a rate of 23.1% among control subjects. The number needed to treat to prevent a single CV event in this subset of patients was only 8.
Less than one-third of the general population in North America and less than half of those at high cardiovascular risk currently receive the annual flu vaccine. Therefore, the flu vaccine "could address a sizable component of residual CV risk not addressed by current therapy, and provide yearlong coverage through a simple inoculation," the researchers said.
"Future research with an adequately powered multicenter trial to confirm the efficacy of this low-cost, annual, safe, easily administered, and well-tolerated therapy to reduce CV risk beyond current therapies is warranted."
The Canadian Institutes for Health Research and the Canadian Foundation for Women’s Health supported the study. Dr. Udell reported no financial conflicts of interest; his associates reported numerous ties to industry sources.
FROM JAMA
Major Finding: The rate of major adverse cardiovascular events during the year following vaccination was 2.9, significantly lower than the 4.7% rate among unvaccinated controls.
Data Source: A systematic review and meta-analysis of 12 randomized clinical trials comparing CV outcomes between 3,238 recipients of the influenza vaccine and 3,231 controls.
Disclosures: The Canadian Institutes for Health Research and the Canadian Foundation for Women’s Health supported the study. Dr. Udell reported no financial conflicts of interest; his associates reported numerous ties to industry sources.
Sixty-four percent of autistic children given psychotropic meds
Sixty-four percent of children with autism spectrum disorders took at least one, and 35% took multiple, psychotropic medications in a national sample of more than 33,000 children with autism spectrum disorders, according to a report published online Oct. 21 in Pediatrics.
Approximately 15% of the children given multiple psychotropic agents took three or more classes of the drugs. Polypharmacy was not uncommon even in the very youngest children, occurring in 33% of those aged 2-10 years and in 10% of babies aged 1 year old or younger, said Donna Spencer, Ph.D., who works for a company that provides analytics, technology, and consulting services in Eden Prairie, Minn., and her associates.
These findings are alarming given the scant evidence that these drugs are safe or effective during childhood, "when developing brains and bodies may be particularly vulnerable to environmental or biological influences," the investigators noted.
"Our study underscores an immediate need to develop standards of care around the prescription of psychotropic medications based on the best available evidence and a coordinated, multidisciplinary approach to improving the health and quality of life of children with [autism spectrum disorders] and their families. It [highlights] the need for rigorous trials of psychotropic medications for children with [autism spectrum disorders] and other psychiatric disorders to assess the value of these medications when weighed against their potential for harm," Dr. Spencer and her colleagues said.
Rates of psychotropic drug use are reportedly increasing among children in general and among those with autism specifically, but to date studies have not yielded accurate estimates of use because they’ve been flawed by small sample sizes, reliance on parent reports, and assessment of very short time periods. Current estimates of the proportion of autistic children who receive the drugs vary dramatically, from 27% to 83% in different studies.
To get a more accurate estimate, the researchers performed a retrospective observational study using a claims database for a large U.S. commercial health plan. They identified 33,565 children across the country who were aged 0-20 years, had autism spectrum disorders, and were enrolled in the health plan for a mean of 44 months during the 8-year study period.
A total of 21,334 (64%) of the patients were given at least one prescription for psychotropic drugs, and 11,598 (35%) were given multiple prescriptions for more than one class of psychotropic drugs. In the latter group, the average number of "episodes" of polypharmacy was 5.6, and the average number of days of polypharmacy was 525.
Approximately 20% of the children given polypharmacy filled prescriptions for two classes of drugs, 10% for three classes, and 5% for four or more classes. Common class combinations were antidepressants and [attention-deficit hyperactivity disorder] medications (38% of subjects); antipsychotics and ADHD medications (28%); antipsychotics and antidepressants (20%); and antipsychotic, antidepressant, and ADHD medications (18%), the investigators said (Pediatrics 2013 Oct. 21 [doi:10.1542/peds.2012-3774]). Forty-five percent or about 15,000 of the patients with autism spectrum disorders had visited a psychiatrist, and 85% of those patients had "any psychotropic use," the investigators said.
The factor most strongly associated with both the use of any psychotropic drugs and the use of multiple psychotropic drugs was comorbidity, particularly comorbid seizures, ADHD, bipolar disorder, anxiety, or depression. More white children and more children aged 11 years or under received psychotropic agents as well as multiple psychotropic agents, Dr. Spencer and her associates reported.
The investigators cited several limitations. For example, administrative claims data are not able to capture whether the patient actually took the medication or whether they did so as directed. In addition Dr. Spencer and her colleagues did not examine dose or appropriateness of dose for particular children because "dosing often depends on a child’s weight." This omission might have led to an overestimation of psychotropic use and polypharmacy in cases in which medications were tapered but still prescribed.
Nevertheless, Dr. Spencer and her associates said the study shows that primary care providers must elicit medication histories carefully and monitor patients’ symptoms to make sure that the medications are effective.
This study was supported by the National Institute of Mental Health. No relevant financial conflicts were reported.
Sixty-four percent of children with autism spectrum disorders took at least one, and 35% took multiple, psychotropic medications in a national sample of more than 33,000 children with autism spectrum disorders, according to a report published online Oct. 21 in Pediatrics.
Approximately 15% of the children given multiple psychotropic agents took three or more classes of the drugs. Polypharmacy was not uncommon even in the very youngest children, occurring in 33% of those aged 2-10 years and in 10% of babies aged 1 year old or younger, said Donna Spencer, Ph.D., who works for a company that provides analytics, technology, and consulting services in Eden Prairie, Minn., and her associates.
These findings are alarming given the scant evidence that these drugs are safe or effective during childhood, "when developing brains and bodies may be particularly vulnerable to environmental or biological influences," the investigators noted.
"Our study underscores an immediate need to develop standards of care around the prescription of psychotropic medications based on the best available evidence and a coordinated, multidisciplinary approach to improving the health and quality of life of children with [autism spectrum disorders] and their families. It [highlights] the need for rigorous trials of psychotropic medications for children with [autism spectrum disorders] and other psychiatric disorders to assess the value of these medications when weighed against their potential for harm," Dr. Spencer and her colleagues said.
Rates of psychotropic drug use are reportedly increasing among children in general and among those with autism specifically, but to date studies have not yielded accurate estimates of use because they’ve been flawed by small sample sizes, reliance on parent reports, and assessment of very short time periods. Current estimates of the proportion of autistic children who receive the drugs vary dramatically, from 27% to 83% in different studies.
To get a more accurate estimate, the researchers performed a retrospective observational study using a claims database for a large U.S. commercial health plan. They identified 33,565 children across the country who were aged 0-20 years, had autism spectrum disorders, and were enrolled in the health plan for a mean of 44 months during the 8-year study period.
A total of 21,334 (64%) of the patients were given at least one prescription for psychotropic drugs, and 11,598 (35%) were given multiple prescriptions for more than one class of psychotropic drugs. In the latter group, the average number of "episodes" of polypharmacy was 5.6, and the average number of days of polypharmacy was 525.
Approximately 20% of the children given polypharmacy filled prescriptions for two classes of drugs, 10% for three classes, and 5% for four or more classes. Common class combinations were antidepressants and [attention-deficit hyperactivity disorder] medications (38% of subjects); antipsychotics and ADHD medications (28%); antipsychotics and antidepressants (20%); and antipsychotic, antidepressant, and ADHD medications (18%), the investigators said (Pediatrics 2013 Oct. 21 [doi:10.1542/peds.2012-3774]). Forty-five percent or about 15,000 of the patients with autism spectrum disorders had visited a psychiatrist, and 85% of those patients had "any psychotropic use," the investigators said.
The factor most strongly associated with both the use of any psychotropic drugs and the use of multiple psychotropic drugs was comorbidity, particularly comorbid seizures, ADHD, bipolar disorder, anxiety, or depression. More white children and more children aged 11 years or under received psychotropic agents as well as multiple psychotropic agents, Dr. Spencer and her associates reported.
The investigators cited several limitations. For example, administrative claims data are not able to capture whether the patient actually took the medication or whether they did so as directed. In addition Dr. Spencer and her colleagues did not examine dose or appropriateness of dose for particular children because "dosing often depends on a child’s weight." This omission might have led to an overestimation of psychotropic use and polypharmacy in cases in which medications were tapered but still prescribed.
Nevertheless, Dr. Spencer and her associates said the study shows that primary care providers must elicit medication histories carefully and monitor patients’ symptoms to make sure that the medications are effective.
This study was supported by the National Institute of Mental Health. No relevant financial conflicts were reported.
Sixty-four percent of children with autism spectrum disorders took at least one, and 35% took multiple, psychotropic medications in a national sample of more than 33,000 children with autism spectrum disorders, according to a report published online Oct. 21 in Pediatrics.
Approximately 15% of the children given multiple psychotropic agents took three or more classes of the drugs. Polypharmacy was not uncommon even in the very youngest children, occurring in 33% of those aged 2-10 years and in 10% of babies aged 1 year old or younger, said Donna Spencer, Ph.D., who works for a company that provides analytics, technology, and consulting services in Eden Prairie, Minn., and her associates.
These findings are alarming given the scant evidence that these drugs are safe or effective during childhood, "when developing brains and bodies may be particularly vulnerable to environmental or biological influences," the investigators noted.
"Our study underscores an immediate need to develop standards of care around the prescription of psychotropic medications based on the best available evidence and a coordinated, multidisciplinary approach to improving the health and quality of life of children with [autism spectrum disorders] and their families. It [highlights] the need for rigorous trials of psychotropic medications for children with [autism spectrum disorders] and other psychiatric disorders to assess the value of these medications when weighed against their potential for harm," Dr. Spencer and her colleagues said.
Rates of psychotropic drug use are reportedly increasing among children in general and among those with autism specifically, but to date studies have not yielded accurate estimates of use because they’ve been flawed by small sample sizes, reliance on parent reports, and assessment of very short time periods. Current estimates of the proportion of autistic children who receive the drugs vary dramatically, from 27% to 83% in different studies.
To get a more accurate estimate, the researchers performed a retrospective observational study using a claims database for a large U.S. commercial health plan. They identified 33,565 children across the country who were aged 0-20 years, had autism spectrum disorders, and were enrolled in the health plan for a mean of 44 months during the 8-year study period.
A total of 21,334 (64%) of the patients were given at least one prescription for psychotropic drugs, and 11,598 (35%) were given multiple prescriptions for more than one class of psychotropic drugs. In the latter group, the average number of "episodes" of polypharmacy was 5.6, and the average number of days of polypharmacy was 525.
Approximately 20% of the children given polypharmacy filled prescriptions for two classes of drugs, 10% for three classes, and 5% for four or more classes. Common class combinations were antidepressants and [attention-deficit hyperactivity disorder] medications (38% of subjects); antipsychotics and ADHD medications (28%); antipsychotics and antidepressants (20%); and antipsychotic, antidepressant, and ADHD medications (18%), the investigators said (Pediatrics 2013 Oct. 21 [doi:10.1542/peds.2012-3774]). Forty-five percent or about 15,000 of the patients with autism spectrum disorders had visited a psychiatrist, and 85% of those patients had "any psychotropic use," the investigators said.
The factor most strongly associated with both the use of any psychotropic drugs and the use of multiple psychotropic drugs was comorbidity, particularly comorbid seizures, ADHD, bipolar disorder, anxiety, or depression. More white children and more children aged 11 years or under received psychotropic agents as well as multiple psychotropic agents, Dr. Spencer and her associates reported.
The investigators cited several limitations. For example, administrative claims data are not able to capture whether the patient actually took the medication or whether they did so as directed. In addition Dr. Spencer and her colleagues did not examine dose or appropriateness of dose for particular children because "dosing often depends on a child’s weight." This omission might have led to an overestimation of psychotropic use and polypharmacy in cases in which medications were tapered but still prescribed.
Nevertheless, Dr. Spencer and her associates said the study shows that primary care providers must elicit medication histories carefully and monitor patients’ symptoms to make sure that the medications are effective.
This study was supported by the National Institute of Mental Health. No relevant financial conflicts were reported.
FROM PEDIATRICS
Major finding: In a national sample, 21,334 autistic children (64%) were given prescriptions for psychotropic medications, and 11,598 (35%) were given prescriptions for multiple classes of psychotropic medications.
Data source: An 8-year retrospective observational cohort study of prescription practices among 33,565 children across the country who had autism spectrum disorders.
Disclosures: This study was supported by the National Institute of Mental Health. No relevant financial conflicts were reported.
Spanking linked to later behavioral, cognitive problems
Young children who are spanked by their mothers or fathers show more externalizing behavior and less cognitive capacity several years later, compared with their peers who are not spanked, according to a report published online Oct. 21 in Pediatrics.
This secondary analysis of data from a longitudinal cohort of 1,933 families across the United States could not establish whether spanking exerts a direct effect on cognitive and emotional development "through stress, trauma, or other physiologic or neural processes, or whether spanking is simply an indirect proxy for other unmeasured parenting practices that negatively affect cognitive development." However, all the data in this rigorously controlled analysis point toward a direct effect "that cannot be simply explained away as spanking families being also less likely to speak to or engage their child in ways important for cognitive development," said Michael J. MacKenzie, Ph.D., of Columbia University School of Social Work, New York, and his associates.
These study findings are of particular concern because a full 57% of these children were spanked by their mothers and 40% by their fathers at 3 years of age, and those rates declined only slightly at 5 years of age, the investigators said.
At age 5 years, 52% of the children were spanked by their mothers and 33% by their fathers. Most mothers estimated that they spanked their children less than twice a week, but 5.5% reported spanking two or more times per week. Most fathers said they spanked their children less than twice a week, but 3% reported doing so two or more times per week (Pediatrics 2013 Oct. 21 [doi:10.1542/peds.2013-1227]).
Compared with no spanking, both high-frequency and low-frequency spanking by mothers at ages 3 years and 5 years were strongly associated with significantly higher levels of externalizing behaviors at age 9 years. This association remained robust after the data were adjusted to account for numerous childhood characteristics such as gender, birth weight, birth order within the family, and temperament as a baby, and maternal traits such as age at the child’s birth, marital status, racial/ethnic background, education level, household income-to-needs ratio, life stress, history of depression or anxiety, impulsivity, and cognitive capacity.
In addition, paternal spanking at age 5 years correlated with reduced receptive verbal capacity in the child at age 9 years, an indication of cognitive ability measured via the Peabody Picture Vocabulary Test.
"Future work should focus on providing families a clearer picture of the outcomes associated with spanking and more information about what discipline practices may have the desired effect on improving functioning, so that they can move beyond punishment practices to the incorporation of positive parenting behaviors with the potential to encourage healthy child trajectories," the investigators noted.
The United States "stands out as one of the few high-income countries that have not followed Sweden’s lead in banning spanking," even though experts, including the American Academy of Pediatrics, warn against the practice and recommend other methods of discipline. To date, several studies have reported a link between being spanked and exhibiting adverse behavioral outcomes, but most have not used longitudinal samples, have not accounted for measures of family stress and socioeconomic variables, have examined maternal spanking only, and have not focused on cognitive or developmental outcomes.
Dr. MacKenzie and his colleagues addressed these issues in their study using a longitudinal data set that followed a large and diverse sample of children from birth through age 9 years. They assessed 1,933 children and their families who participated in the Fragile Families and Child Well-Being (FFCW) study, which included a representative sample of children born during 1988-2000 in 20 medium- to large-size cities.
This subset of FFCW participants had more resources and appeared more stable than did the entire cohort, but still represented "a fairly disadvantaged urban sample," the researchers said.
This study was funded by the National Institutes of Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Science Foundation, and the Department of Health and Human Services. No financial conflicts of interest were reported.
Why do parents spank a child? Were they themselves spanked growing up? Is the child’s behavior so difficult to control or is the parent’s range of options too limited? Is spanking a reflection of a child rearing that reflects less reliance on words, expectations, and relationship to manage behavior? Is spanking a secondary result of stress on the family from medical or psychiatric illness, financial pressures, or insufficient social supports (child care or extended family)? Is infrequent spanking the same as more frequent spanking? How do we assess the severity of a spanking or the context of the before and after communication?
No study can address all of the many variables that are part of the parental history, nature of the child, various circumstances, psychosocial stressors, and quality of communication. However, this study does add a level of detail and longer term outcomes. It includes a large sample size and a well validated instrument to assess aggressive symptoms several years after early childhood spanking. Additionally, it assessed the impact of demographic and other psychosocial factors that could add stress to the family.

|
|
The key point is noteworthy and relevant to pediatric primary care practice. More than 50% of both 3- and 5-year-old children were spanked, 12% of 3-year-olds at a frequency of twice a week. Spanking increases the likelihood that a child will have more aggressive behaviors at age 9 years as well as potentially limiting the child’s cognitive development. More frequent spanking and spanking above age 3 years is more predictive, mother’s spanking is more predictive than father’s, and the study has a number of variables that further refine these findings. This study adds considerable credibility to the growing literature indicating that spanking encourages more aggressive behaviors in later childhood and may have a number of additional undesirable outcomes.
Michael S. Jellinek, M.D., is professor of psychiatry and of pediatrics at Harvard Medical School, Boston. He is chief clinical officer at Partners HealthCare, also in Boston. He said he had no disclosures relevant to this topic. Dr. Jellinek was asked to comment on this article.
Why do parents spank a child? Were they themselves spanked growing up? Is the child’s behavior so difficult to control or is the parent’s range of options too limited? Is spanking a reflection of a child rearing that reflects less reliance on words, expectations, and relationship to manage behavior? Is spanking a secondary result of stress on the family from medical or psychiatric illness, financial pressures, or insufficient social supports (child care or extended family)? Is infrequent spanking the same as more frequent spanking? How do we assess the severity of a spanking or the context of the before and after communication?
No study can address all of the many variables that are part of the parental history, nature of the child, various circumstances, psychosocial stressors, and quality of communication. However, this study does add a level of detail and longer term outcomes. It includes a large sample size and a well validated instrument to assess aggressive symptoms several years after early childhood spanking. Additionally, it assessed the impact of demographic and other psychosocial factors that could add stress to the family.

|
|
The key point is noteworthy and relevant to pediatric primary care practice. More than 50% of both 3- and 5-year-old children were spanked, 12% of 3-year-olds at a frequency of twice a week. Spanking increases the likelihood that a child will have more aggressive behaviors at age 9 years as well as potentially limiting the child’s cognitive development. More frequent spanking and spanking above age 3 years is more predictive, mother’s spanking is more predictive than father’s, and the study has a number of variables that further refine these findings. This study adds considerable credibility to the growing literature indicating that spanking encourages more aggressive behaviors in later childhood and may have a number of additional undesirable outcomes.
Michael S. Jellinek, M.D., is professor of psychiatry and of pediatrics at Harvard Medical School, Boston. He is chief clinical officer at Partners HealthCare, also in Boston. He said he had no disclosures relevant to this topic. Dr. Jellinek was asked to comment on this article.
Why do parents spank a child? Were they themselves spanked growing up? Is the child’s behavior so difficult to control or is the parent’s range of options too limited? Is spanking a reflection of a child rearing that reflects less reliance on words, expectations, and relationship to manage behavior? Is spanking a secondary result of stress on the family from medical or psychiatric illness, financial pressures, or insufficient social supports (child care or extended family)? Is infrequent spanking the same as more frequent spanking? How do we assess the severity of a spanking or the context of the before and after communication?
No study can address all of the many variables that are part of the parental history, nature of the child, various circumstances, psychosocial stressors, and quality of communication. However, this study does add a level of detail and longer term outcomes. It includes a large sample size and a well validated instrument to assess aggressive symptoms several years after early childhood spanking. Additionally, it assessed the impact of demographic and other psychosocial factors that could add stress to the family.

|
|
The key point is noteworthy and relevant to pediatric primary care practice. More than 50% of both 3- and 5-year-old children were spanked, 12% of 3-year-olds at a frequency of twice a week. Spanking increases the likelihood that a child will have more aggressive behaviors at age 9 years as well as potentially limiting the child’s cognitive development. More frequent spanking and spanking above age 3 years is more predictive, mother’s spanking is more predictive than father’s, and the study has a number of variables that further refine these findings. This study adds considerable credibility to the growing literature indicating that spanking encourages more aggressive behaviors in later childhood and may have a number of additional undesirable outcomes.
Michael S. Jellinek, M.D., is professor of psychiatry and of pediatrics at Harvard Medical School, Boston. He is chief clinical officer at Partners HealthCare, also in Boston. He said he had no disclosures relevant to this topic. Dr. Jellinek was asked to comment on this article.
Young children who are spanked by their mothers or fathers show more externalizing behavior and less cognitive capacity several years later, compared with their peers who are not spanked, according to a report published online Oct. 21 in Pediatrics.
This secondary analysis of data from a longitudinal cohort of 1,933 families across the United States could not establish whether spanking exerts a direct effect on cognitive and emotional development "through stress, trauma, or other physiologic or neural processes, or whether spanking is simply an indirect proxy for other unmeasured parenting practices that negatively affect cognitive development." However, all the data in this rigorously controlled analysis point toward a direct effect "that cannot be simply explained away as spanking families being also less likely to speak to or engage their child in ways important for cognitive development," said Michael J. MacKenzie, Ph.D., of Columbia University School of Social Work, New York, and his associates.
These study findings are of particular concern because a full 57% of these children were spanked by their mothers and 40% by their fathers at 3 years of age, and those rates declined only slightly at 5 years of age, the investigators said.
At age 5 years, 52% of the children were spanked by their mothers and 33% by their fathers. Most mothers estimated that they spanked their children less than twice a week, but 5.5% reported spanking two or more times per week. Most fathers said they spanked their children less than twice a week, but 3% reported doing so two or more times per week (Pediatrics 2013 Oct. 21 [doi:10.1542/peds.2013-1227]).
Compared with no spanking, both high-frequency and low-frequency spanking by mothers at ages 3 years and 5 years were strongly associated with significantly higher levels of externalizing behaviors at age 9 years. This association remained robust after the data were adjusted to account for numerous childhood characteristics such as gender, birth weight, birth order within the family, and temperament as a baby, and maternal traits such as age at the child’s birth, marital status, racial/ethnic background, education level, household income-to-needs ratio, life stress, history of depression or anxiety, impulsivity, and cognitive capacity.
In addition, paternal spanking at age 5 years correlated with reduced receptive verbal capacity in the child at age 9 years, an indication of cognitive ability measured via the Peabody Picture Vocabulary Test.
"Future work should focus on providing families a clearer picture of the outcomes associated with spanking and more information about what discipline practices may have the desired effect on improving functioning, so that they can move beyond punishment practices to the incorporation of positive parenting behaviors with the potential to encourage healthy child trajectories," the investigators noted.
The United States "stands out as one of the few high-income countries that have not followed Sweden’s lead in banning spanking," even though experts, including the American Academy of Pediatrics, warn against the practice and recommend other methods of discipline. To date, several studies have reported a link between being spanked and exhibiting adverse behavioral outcomes, but most have not used longitudinal samples, have not accounted for measures of family stress and socioeconomic variables, have examined maternal spanking only, and have not focused on cognitive or developmental outcomes.
Dr. MacKenzie and his colleagues addressed these issues in their study using a longitudinal data set that followed a large and diverse sample of children from birth through age 9 years. They assessed 1,933 children and their families who participated in the Fragile Families and Child Well-Being (FFCW) study, which included a representative sample of children born during 1988-2000 in 20 medium- to large-size cities.
This subset of FFCW participants had more resources and appeared more stable than did the entire cohort, but still represented "a fairly disadvantaged urban sample," the researchers said.
This study was funded by the National Institutes of Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Science Foundation, and the Department of Health and Human Services. No financial conflicts of interest were reported.
Young children who are spanked by their mothers or fathers show more externalizing behavior and less cognitive capacity several years later, compared with their peers who are not spanked, according to a report published online Oct. 21 in Pediatrics.
This secondary analysis of data from a longitudinal cohort of 1,933 families across the United States could not establish whether spanking exerts a direct effect on cognitive and emotional development "through stress, trauma, or other physiologic or neural processes, or whether spanking is simply an indirect proxy for other unmeasured parenting practices that negatively affect cognitive development." However, all the data in this rigorously controlled analysis point toward a direct effect "that cannot be simply explained away as spanking families being also less likely to speak to or engage their child in ways important for cognitive development," said Michael J. MacKenzie, Ph.D., of Columbia University School of Social Work, New York, and his associates.
These study findings are of particular concern because a full 57% of these children were spanked by their mothers and 40% by their fathers at 3 years of age, and those rates declined only slightly at 5 years of age, the investigators said.
At age 5 years, 52% of the children were spanked by their mothers and 33% by their fathers. Most mothers estimated that they spanked their children less than twice a week, but 5.5% reported spanking two or more times per week. Most fathers said they spanked their children less than twice a week, but 3% reported doing so two or more times per week (Pediatrics 2013 Oct. 21 [doi:10.1542/peds.2013-1227]).
Compared with no spanking, both high-frequency and low-frequency spanking by mothers at ages 3 years and 5 years were strongly associated with significantly higher levels of externalizing behaviors at age 9 years. This association remained robust after the data were adjusted to account for numerous childhood characteristics such as gender, birth weight, birth order within the family, and temperament as a baby, and maternal traits such as age at the child’s birth, marital status, racial/ethnic background, education level, household income-to-needs ratio, life stress, history of depression or anxiety, impulsivity, and cognitive capacity.
In addition, paternal spanking at age 5 years correlated with reduced receptive verbal capacity in the child at age 9 years, an indication of cognitive ability measured via the Peabody Picture Vocabulary Test.
"Future work should focus on providing families a clearer picture of the outcomes associated with spanking and more information about what discipline practices may have the desired effect on improving functioning, so that they can move beyond punishment practices to the incorporation of positive parenting behaviors with the potential to encourage healthy child trajectories," the investigators noted.
The United States "stands out as one of the few high-income countries that have not followed Sweden’s lead in banning spanking," even though experts, including the American Academy of Pediatrics, warn against the practice and recommend other methods of discipline. To date, several studies have reported a link between being spanked and exhibiting adverse behavioral outcomes, but most have not used longitudinal samples, have not accounted for measures of family stress and socioeconomic variables, have examined maternal spanking only, and have not focused on cognitive or developmental outcomes.
Dr. MacKenzie and his colleagues addressed these issues in their study using a longitudinal data set that followed a large and diverse sample of children from birth through age 9 years. They assessed 1,933 children and their families who participated in the Fragile Families and Child Well-Being (FFCW) study, which included a representative sample of children born during 1988-2000 in 20 medium- to large-size cities.
This subset of FFCW participants had more resources and appeared more stable than did the entire cohort, but still represented "a fairly disadvantaged urban sample," the researchers said.
This study was funded by the National Institutes of Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Science Foundation, and the Department of Health and Human Services. No financial conflicts of interest were reported.
FROM PEDIATRICS
Major Finding: Fifty-seven percent of children were spanked by their mothers and 40% by their fathers at age 3 years, and 52% were spanked by mothers and 33% by fathers at age 5 years. Compared with no spanking, both high-frequency and low-frequency spanking by mothers at ages 3 years and 5 years were strongly associated with significantly higher levels of externalizing behaviors at age 9 years.
Data Source: A longitudinal birth cohort of 1,933 children and families residing in 20 medium- to large-size cities and followed for 9 years.
Disclosures: This study was funded by the National Institutes of Health, the Eunice Kennedy Shriver National Institute of Child Health and Human Development, the National Science Foundation, and the Department of Health and Human Services. No financial conflicts of interest were reported.
Hospitalist/hepatologist comanagement best for chronic liver disease
Patients with chronic liver disease who are hospitalized for spontaneous bacterial peritonitis were more likely to be treated according to evidence-based guidelines if their care was comanaged by a hospitalist and a hepatologist, rather than the traditional approach, according to a report in the Journal of Clinical Gastroenterology.
In a study comparing outcomes before the implementation of a hospitalist/hepatologist comanagement program with those afterward at one medical center, the comanaged care improved adherence to a variety of recommendations, such as performing diagnostic paracentesis within 24 hours, administering albumin appropriately, and providing ongoing peritonitis prophylaxis at discharge, said Dr. Archita P. Desai of the section of gastroenterology, hepatology, and nutrition, University of Chicago Medical Center, and her associates.
"No study to date has evaluated the quality of comanaged hospitalist care for complex medical subspecialty patients," they noted.
Dr. Desai and her colleagues performed a chart review of 56 adults with chronic liver disease (CLD) who were admitted either during the 2 years before the comanagement program was implemented (January 2004 through June 2006) or the 4 years afterward (July 2006 through December 2010). A total of 26 patients received conventional care during the earlier period, and 30 received joint care under the new comanagement program during the latter.
The two study groups were similar regarding patient age, sex, race, etiology of cirrhosis, serum bilirubin levels, serum INR (international normalized ratio), end-stage liver disease scores, and percentage with bacteremia and/or variceal hemorrhage.
With the comanagement program, patients were admitted to a hepatology unit by a hospitalist team comprising an academic hospitalist and several nurse practitioners and physician assistants. The hepatology team included a hepatologist, a gastroenterology fellow, and internal medicine residents and medical students rotating through the unit.
Both teams evaluated patients separately but met for daily "sit-down" rounds in the early afternoon to discuss the cases.
Patients in the new program were significantly more likely to undergo paracentesis within 24 hours (100% vs. 79%), to receive albumin appropriately while hospitalized (97% vs. 65%), and to receive at discharge medication to prevent further peritonitis (91% vs. 32%).
Patients under comanagement also were less likely to receive fresh-frozen plasma, which is contraindicated, and more likely to have antibiotic therapy initiated within 6 hours of presentation (J. Clin. Gastroenterol. 2013 [doi:10.1097/MCG.0b013e3182a87f70]).
The two groups were similar regarding other processes of care such as the choice of appropriate antibiotics, the use of DVT prophylaxis while confined to bed, and the offer of pneumococcal vaccination at discharge.
There was a statistical trend, which did not reach significance, for comanaged patients to have a lower rate of transfer to an ICU (27% vs 17%). Similarly, there was a nonsignificant trend toward a lower inpatient mortality with comanaged 13%) than with conventional (27%) care.
Mortality at 30 days after discharge also was lower after comanagement than conventional care (0% vs 5.3%). However, this trend also didn’t reach statistical significance because 30-day mortality data were missing for a substantial number of patients in both study groups.
"We speculate that the close interactions between the primary inpatient team and the hepatology team during formal daily rounds facilitated structured communication between the hospitalist and the hepatology teams, and likely played a significant role in the intervention," Dr. Desai and her associates said.
In addition, comanagement allowed the hepatology team to be more involved with detailed discharge planning specifically related to liver disease, "which in turn improves transition to outpatient care, a time of vulnerability for these complex patients," they said.
Both the length of stay and the total cost of hospitalization tended to be higher under the comanagement program. However, "overall care may be more cost-effective after considering variables such as improved access to specialty-specific resources in the short term and the prospect of increasing transplantation rates earlier in the course of disease, which in the long term leads to the prevention of costly complications," the investigators noted.
This study was supported by the University of Chicago’s Liver Research Fund. No financial conflicts of interest were reported.
Patients with chronic liver disease who are hospitalized for spontaneous bacterial peritonitis were more likely to be treated according to evidence-based guidelines if their care was comanaged by a hospitalist and a hepatologist, rather than the traditional approach, according to a report in the Journal of Clinical Gastroenterology.
In a study comparing outcomes before the implementation of a hospitalist/hepatologist comanagement program with those afterward at one medical center, the comanaged care improved adherence to a variety of recommendations, such as performing diagnostic paracentesis within 24 hours, administering albumin appropriately, and providing ongoing peritonitis prophylaxis at discharge, said Dr. Archita P. Desai of the section of gastroenterology, hepatology, and nutrition, University of Chicago Medical Center, and her associates.
"No study to date has evaluated the quality of comanaged hospitalist care for complex medical subspecialty patients," they noted.
Dr. Desai and her colleagues performed a chart review of 56 adults with chronic liver disease (CLD) who were admitted either during the 2 years before the comanagement program was implemented (January 2004 through June 2006) or the 4 years afterward (July 2006 through December 2010). A total of 26 patients received conventional care during the earlier period, and 30 received joint care under the new comanagement program during the latter.
The two study groups were similar regarding patient age, sex, race, etiology of cirrhosis, serum bilirubin levels, serum INR (international normalized ratio), end-stage liver disease scores, and percentage with bacteremia and/or variceal hemorrhage.
With the comanagement program, patients were admitted to a hepatology unit by a hospitalist team comprising an academic hospitalist and several nurse practitioners and physician assistants. The hepatology team included a hepatologist, a gastroenterology fellow, and internal medicine residents and medical students rotating through the unit.
Both teams evaluated patients separately but met for daily "sit-down" rounds in the early afternoon to discuss the cases.
Patients in the new program were significantly more likely to undergo paracentesis within 24 hours (100% vs. 79%), to receive albumin appropriately while hospitalized (97% vs. 65%), and to receive at discharge medication to prevent further peritonitis (91% vs. 32%).
Patients under comanagement also were less likely to receive fresh-frozen plasma, which is contraindicated, and more likely to have antibiotic therapy initiated within 6 hours of presentation (J. Clin. Gastroenterol. 2013 [doi:10.1097/MCG.0b013e3182a87f70]).
The two groups were similar regarding other processes of care such as the choice of appropriate antibiotics, the use of DVT prophylaxis while confined to bed, and the offer of pneumococcal vaccination at discharge.
There was a statistical trend, which did not reach significance, for comanaged patients to have a lower rate of transfer to an ICU (27% vs 17%). Similarly, there was a nonsignificant trend toward a lower inpatient mortality with comanaged 13%) than with conventional (27%) care.
Mortality at 30 days after discharge also was lower after comanagement than conventional care (0% vs 5.3%). However, this trend also didn’t reach statistical significance because 30-day mortality data were missing for a substantial number of patients in both study groups.
"We speculate that the close interactions between the primary inpatient team and the hepatology team during formal daily rounds facilitated structured communication between the hospitalist and the hepatology teams, and likely played a significant role in the intervention," Dr. Desai and her associates said.
In addition, comanagement allowed the hepatology team to be more involved with detailed discharge planning specifically related to liver disease, "which in turn improves transition to outpatient care, a time of vulnerability for these complex patients," they said.
Both the length of stay and the total cost of hospitalization tended to be higher under the comanagement program. However, "overall care may be more cost-effective after considering variables such as improved access to specialty-specific resources in the short term and the prospect of increasing transplantation rates earlier in the course of disease, which in the long term leads to the prevention of costly complications," the investigators noted.
This study was supported by the University of Chicago’s Liver Research Fund. No financial conflicts of interest were reported.
Patients with chronic liver disease who are hospitalized for spontaneous bacterial peritonitis were more likely to be treated according to evidence-based guidelines if their care was comanaged by a hospitalist and a hepatologist, rather than the traditional approach, according to a report in the Journal of Clinical Gastroenterology.
In a study comparing outcomes before the implementation of a hospitalist/hepatologist comanagement program with those afterward at one medical center, the comanaged care improved adherence to a variety of recommendations, such as performing diagnostic paracentesis within 24 hours, administering albumin appropriately, and providing ongoing peritonitis prophylaxis at discharge, said Dr. Archita P. Desai of the section of gastroenterology, hepatology, and nutrition, University of Chicago Medical Center, and her associates.
"No study to date has evaluated the quality of comanaged hospitalist care for complex medical subspecialty patients," they noted.
Dr. Desai and her colleagues performed a chart review of 56 adults with chronic liver disease (CLD) who were admitted either during the 2 years before the comanagement program was implemented (January 2004 through June 2006) or the 4 years afterward (July 2006 through December 2010). A total of 26 patients received conventional care during the earlier period, and 30 received joint care under the new comanagement program during the latter.
The two study groups were similar regarding patient age, sex, race, etiology of cirrhosis, serum bilirubin levels, serum INR (international normalized ratio), end-stage liver disease scores, and percentage with bacteremia and/or variceal hemorrhage.
With the comanagement program, patients were admitted to a hepatology unit by a hospitalist team comprising an academic hospitalist and several nurse practitioners and physician assistants. The hepatology team included a hepatologist, a gastroenterology fellow, and internal medicine residents and medical students rotating through the unit.
Both teams evaluated patients separately but met for daily "sit-down" rounds in the early afternoon to discuss the cases.
Patients in the new program were significantly more likely to undergo paracentesis within 24 hours (100% vs. 79%), to receive albumin appropriately while hospitalized (97% vs. 65%), and to receive at discharge medication to prevent further peritonitis (91% vs. 32%).
Patients under comanagement also were less likely to receive fresh-frozen plasma, which is contraindicated, and more likely to have antibiotic therapy initiated within 6 hours of presentation (J. Clin. Gastroenterol. 2013 [doi:10.1097/MCG.0b013e3182a87f70]).
The two groups were similar regarding other processes of care such as the choice of appropriate antibiotics, the use of DVT prophylaxis while confined to bed, and the offer of pneumococcal vaccination at discharge.
There was a statistical trend, which did not reach significance, for comanaged patients to have a lower rate of transfer to an ICU (27% vs 17%). Similarly, there was a nonsignificant trend toward a lower inpatient mortality with comanaged 13%) than with conventional (27%) care.
Mortality at 30 days after discharge also was lower after comanagement than conventional care (0% vs 5.3%). However, this trend also didn’t reach statistical significance because 30-day mortality data were missing for a substantial number of patients in both study groups.
"We speculate that the close interactions between the primary inpatient team and the hepatology team during formal daily rounds facilitated structured communication between the hospitalist and the hepatology teams, and likely played a significant role in the intervention," Dr. Desai and her associates said.
In addition, comanagement allowed the hepatology team to be more involved with detailed discharge planning specifically related to liver disease, "which in turn improves transition to outpatient care, a time of vulnerability for these complex patients," they said.
Both the length of stay and the total cost of hospitalization tended to be higher under the comanagement program. However, "overall care may be more cost-effective after considering variables such as improved access to specialty-specific resources in the short term and the prospect of increasing transplantation rates earlier in the course of disease, which in the long term leads to the prevention of costly complications," the investigators noted.
This study was supported by the University of Chicago’s Liver Research Fund. No financial conflicts of interest were reported.
FROM THE JOURNAL OF CLINICAL GASTROENTEROLOGY
Major finding: Patients cared for in the hospitalist/hepatologist comanagement program were significantly more likely to undergo paracentesis within 24 hours (100% vs 79%), to receive albumin appropriately while hospitalized (97% vs. 65%), and to receive medication at discharge to prevent further peritonitis (91% vs 32%), compared with those who were not.
Data source: A retrospective chart review involving 26 inpatients with chronic liver disease who were treated before implementation of a joint hospitalist/hepatologist management program and 30 who were treated afterward.
Disclosures: This study was supported by the University of Chicago’s Liver Research Fund. No financial conflicts of interest were reported.
Adjuvant fish oil may help recent-onset RA
Fish oil supplements raised the rate of remission of rheumatoid arthritis when added to background therapy with disease-modifying antirheumatic drugs in a double-blind, randomized controlled trial of patients with recent-onset disease who previously had not taken DMARDs.
Adjuvant fish oil also reduced the use of nonsteroidal anti-inflammatory drugs (NSAIDs) in the trial, said Dr. Susanna M. Proudman of the rheumatology unit, Royal Adelaide (Australia) Hospital, and her associates (Ann. Rheum. Dis. 2013 Sept. 30 [doi:10.1136/annrheumdis-2013-204145]) .
"It can be presumed that fish oil added to this ... treatment regimen will at least delay progression to biological therapy, and the effects may even exceed this minimal expectation," they wrote.
As a side benefit, fish oil also may exert important cardiovascular effects in rheumatoid arthritis (RA) patients, who are at increased cardiovascular risk and have almost twice the risk of sudden cardiac death as the general population, Dr. Proudman and her colleagues noted.
The investigators assessed consecutive patients presenting to the early arthritis clinic at the hospital, all of whom were started on standard, treat-to-target triple-DMARD therapy (methotrexate plus folic acid, sulfasalazine, and hydroxychloroquine) and had RA for less than 1 year. The combination treatment was adjusted individually according to each patient’s treatment response and tolerance of adverse effects.
A total of 139 patients were assigned in a double-blind fashion to receive 10 mL/day of fish oil concentrate that provided 5.5 g/day of the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) or 10 mL/day of a 2:1 ratio of sunola oil and capelin oil that provided 400 mg/day EPA and DHA. The control group’s mixture contained only enough fish oil to impart a camouflaging "fishy" odor.
The participants were discouraged from using NSAIDs or corticosteroids during the study.
Patients were assessed every 3 weeks for 3 months, then every 6 weeks thereafter for the 1-year duration of the trial.
The primary outcome measure was the failure of triple-DMARD therapy to control disease activity, as evidenced by progression to leflunomide treatment or biologic agents. The investigators noted that the level of disease activity at which drug dosing was escalated was equivalent to a 28-joint Disease Activity Score of 3.2. This rate was only 10.5% (9 of 86 patients) in the fish oil group, compared with 32.1% (17 of 53 patients) in the control group.
Only one study subject, a member of the control group, progressed from leflunomide to biologic agents.
In addition to a lower rate of triple-DMARD therapy failure, subjects in the active-treatment group were significantly more likely to reach RA remission, as measured by American College of Rheumatology criteria, than were those in the control group (hazard ratio, 2.09). They also were nearly twice as likely to discontinue the use of NSAIDS within 12 weeks of baseline, the investigators said.
However, there were no significant differences between the two study groups in scores on a measure of activities of daily living, the rate of using oral or parenteral steroids, the cumulative dose of steroids, or the mean dose of methotrexate.
The proportion of patients who reported an adverse event or a serious adverse event was similar between the two study groups, and there was no pattern of events suggesting any linkage with the fish oil supplements, Dr. Proudman and her associates said.
This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
Fish oil supplements raised the rate of remission of rheumatoid arthritis when added to background therapy with disease-modifying antirheumatic drugs in a double-blind, randomized controlled trial of patients with recent-onset disease who previously had not taken DMARDs.
Adjuvant fish oil also reduced the use of nonsteroidal anti-inflammatory drugs (NSAIDs) in the trial, said Dr. Susanna M. Proudman of the rheumatology unit, Royal Adelaide (Australia) Hospital, and her associates (Ann. Rheum. Dis. 2013 Sept. 30 [doi:10.1136/annrheumdis-2013-204145]) .
"It can be presumed that fish oil added to this ... treatment regimen will at least delay progression to biological therapy, and the effects may even exceed this minimal expectation," they wrote.
As a side benefit, fish oil also may exert important cardiovascular effects in rheumatoid arthritis (RA) patients, who are at increased cardiovascular risk and have almost twice the risk of sudden cardiac death as the general population, Dr. Proudman and her colleagues noted.
The investigators assessed consecutive patients presenting to the early arthritis clinic at the hospital, all of whom were started on standard, treat-to-target triple-DMARD therapy (methotrexate plus folic acid, sulfasalazine, and hydroxychloroquine) and had RA for less than 1 year. The combination treatment was adjusted individually according to each patient’s treatment response and tolerance of adverse effects.
A total of 139 patients were assigned in a double-blind fashion to receive 10 mL/day of fish oil concentrate that provided 5.5 g/day of the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) or 10 mL/day of a 2:1 ratio of sunola oil and capelin oil that provided 400 mg/day EPA and DHA. The control group’s mixture contained only enough fish oil to impart a camouflaging "fishy" odor.
The participants were discouraged from using NSAIDs or corticosteroids during the study.
Patients were assessed every 3 weeks for 3 months, then every 6 weeks thereafter for the 1-year duration of the trial.
The primary outcome measure was the failure of triple-DMARD therapy to control disease activity, as evidenced by progression to leflunomide treatment or biologic agents. The investigators noted that the level of disease activity at which drug dosing was escalated was equivalent to a 28-joint Disease Activity Score of 3.2. This rate was only 10.5% (9 of 86 patients) in the fish oil group, compared with 32.1% (17 of 53 patients) in the control group.
Only one study subject, a member of the control group, progressed from leflunomide to biologic agents.
In addition to a lower rate of triple-DMARD therapy failure, subjects in the active-treatment group were significantly more likely to reach RA remission, as measured by American College of Rheumatology criteria, than were those in the control group (hazard ratio, 2.09). They also were nearly twice as likely to discontinue the use of NSAIDS within 12 weeks of baseline, the investigators said.
However, there were no significant differences between the two study groups in scores on a measure of activities of daily living, the rate of using oral or parenteral steroids, the cumulative dose of steroids, or the mean dose of methotrexate.
The proportion of patients who reported an adverse event or a serious adverse event was similar between the two study groups, and there was no pattern of events suggesting any linkage with the fish oil supplements, Dr. Proudman and her associates said.
This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
Fish oil supplements raised the rate of remission of rheumatoid arthritis when added to background therapy with disease-modifying antirheumatic drugs in a double-blind, randomized controlled trial of patients with recent-onset disease who previously had not taken DMARDs.
Adjuvant fish oil also reduced the use of nonsteroidal anti-inflammatory drugs (NSAIDs) in the trial, said Dr. Susanna M. Proudman of the rheumatology unit, Royal Adelaide (Australia) Hospital, and her associates (Ann. Rheum. Dis. 2013 Sept. 30 [doi:10.1136/annrheumdis-2013-204145]) .
"It can be presumed that fish oil added to this ... treatment regimen will at least delay progression to biological therapy, and the effects may even exceed this minimal expectation," they wrote.
As a side benefit, fish oil also may exert important cardiovascular effects in rheumatoid arthritis (RA) patients, who are at increased cardiovascular risk and have almost twice the risk of sudden cardiac death as the general population, Dr. Proudman and her colleagues noted.
The investigators assessed consecutive patients presenting to the early arthritis clinic at the hospital, all of whom were started on standard, treat-to-target triple-DMARD therapy (methotrexate plus folic acid, sulfasalazine, and hydroxychloroquine) and had RA for less than 1 year. The combination treatment was adjusted individually according to each patient’s treatment response and tolerance of adverse effects.
A total of 139 patients were assigned in a double-blind fashion to receive 10 mL/day of fish oil concentrate that provided 5.5 g/day of the omega-3 fatty acids eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) or 10 mL/day of a 2:1 ratio of sunola oil and capelin oil that provided 400 mg/day EPA and DHA. The control group’s mixture contained only enough fish oil to impart a camouflaging "fishy" odor.
The participants were discouraged from using NSAIDs or corticosteroids during the study.
Patients were assessed every 3 weeks for 3 months, then every 6 weeks thereafter for the 1-year duration of the trial.
The primary outcome measure was the failure of triple-DMARD therapy to control disease activity, as evidenced by progression to leflunomide treatment or biologic agents. The investigators noted that the level of disease activity at which drug dosing was escalated was equivalent to a 28-joint Disease Activity Score of 3.2. This rate was only 10.5% (9 of 86 patients) in the fish oil group, compared with 32.1% (17 of 53 patients) in the control group.
Only one study subject, a member of the control group, progressed from leflunomide to biologic agents.
In addition to a lower rate of triple-DMARD therapy failure, subjects in the active-treatment group were significantly more likely to reach RA remission, as measured by American College of Rheumatology criteria, than were those in the control group (hazard ratio, 2.09). They also were nearly twice as likely to discontinue the use of NSAIDS within 12 weeks of baseline, the investigators said.
However, there were no significant differences between the two study groups in scores on a measure of activities of daily living, the rate of using oral or parenteral steroids, the cumulative dose of steroids, or the mean dose of methotrexate.
The proportion of patients who reported an adverse event or a serious adverse event was similar between the two study groups, and there was no pattern of events suggesting any linkage with the fish oil supplements, Dr. Proudman and her associates said.
This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
FROM ANNALS OF THE RHEUMATIC DISEASES
Major Finding: The primary outcome measure, failure of triple-DMARD therapy, occurred in only 10.5% of study subjects receiving fish oil supplements, compared with 32.1% of control subjects.
Data Source: A double-blind, randomized controlled trial involving 139 consecutive patients with recently diagnosed RA who received standard triple-DMARD treatment plus either fish oil (86 patients) or a placebo (53 patients) and were followed for 1 year.
Disclosures: This study was supported by the National Health Medical Research Council of Australia, the Royal Adelaide Hospital Research Committee, and Melrose Health. No relevant financial disclosures were reported.
Use lasers? Lawsuits soar when staff does the procedure
Litigation concerning cutaneous laser surgery has surged in recent years, and the proportion involving nonphysician operators soared from 36% in 1999 to 78% in 2012, according to a report published online Oct. 16 in JAMA Dermatology.
For the most commonly performed laser procedure – hair removal – the proportion of lawsuits involving nonphysician operators is even higher. A "remarkable" 91% of the cases filed in the most recent year for which complete data are available were brought against nonphysician operators, said Dr. H. Ray Jalian of the division of dermatology, University of California, Los Angeles, and his associates.
"The overall trend in increased litigation for laser surgery is in part explained by greater numbers of nonphysician operators performing these procedures, in particular those practicing without direct supervision in ‘medical spas,’ " the investigators said.
Dr. Jalian and his colleagues searched a single legal database and identified 175 cases concerning injury arising from cutaneous laser surgery since 1999. Nonphysician operators included registered nurses, nurse practitioners, medical assistants, electrologists, technicians, aestheticians, and others.
Overall, nonphysician operators were sued in 43% of all cases. But this total is somewhat misleading, because that percentage has risen markedly over time. It represented 78% of all cases in 2011 and both of the two cases filed in early 2012.
Hair removal was the most frequent laser procedure involved in litigation. A total of 76% of the cases brought for hair removal–associated injury in 2004-2012 involved nonphysician operators. Again, this number is markedly higher when the data are categorized by time period: 86% of all lawsuits in 2008-2012 and 91% of all lawsuits in 2010-2012 involved nonphysicians performing laser hair removal.
When the data were analyzed by the location where laser procedures took place, the majority (64%) of cases involving nonphysician operators occurred at facilities such as spas and salons. Again, in more recent years (2008-2012), "nonphysician-operator procedures performed in ‘medical spas’ represented 77% of lawsuits," Dr. Jalian and his associates said (JAMA Dermatology 2013 Oct. 16 [doi:10.1001/jamadermatol.2013. 7117]).
Clinicians also should be aware that almost all the malpractice cases arising from nonphysician operators’ negligence extend beyond the operator to his or her employer, be that a physician, a medical facility, or a spa.
By far the most common allegation against physicians was failure to supervise a nonphysician operator (27 cases), followed by failure to train or hire appropriate staff (23 cases).
The investigators noted that "the actual data likely understate the true incidence of nonphysician operator laser complications" since they were culled from the large legal database that excluded complaints that were handled outside the judicial system.
Laws regulating the use of cutaneous lasers vary widely by state, from edicts specifying that only physicians may operate lasers for hair removal (Maine) to no regulations at all on laser use (Nevada). Even when clinicians supervise so-called physician extenders, rules concerning the degree of supervision, protocol requirements, and employee training are ambiguous.
"Because laws and regulations are constantly evolving, it is imperative for [those] who use physician extenders to be up to date," the researchers said.
Dr. Jalian reported no relevant financial conflicts of interest. One of his associates reported ties to Zeltiq Aesthetics, Cytrellis Biosystems, Unilever, and Allergan.
Litigation concerning cutaneous laser surgery has surged in recent years, and the proportion involving nonphysician operators soared from 36% in 1999 to 78% in 2012, according to a report published online Oct. 16 in JAMA Dermatology.
For the most commonly performed laser procedure – hair removal – the proportion of lawsuits involving nonphysician operators is even higher. A "remarkable" 91% of the cases filed in the most recent year for which complete data are available were brought against nonphysician operators, said Dr. H. Ray Jalian of the division of dermatology, University of California, Los Angeles, and his associates.
"The overall trend in increased litigation for laser surgery is in part explained by greater numbers of nonphysician operators performing these procedures, in particular those practicing without direct supervision in ‘medical spas,’ " the investigators said.
Dr. Jalian and his colleagues searched a single legal database and identified 175 cases concerning injury arising from cutaneous laser surgery since 1999. Nonphysician operators included registered nurses, nurse practitioners, medical assistants, electrologists, technicians, aestheticians, and others.
Overall, nonphysician operators were sued in 43% of all cases. But this total is somewhat misleading, because that percentage has risen markedly over time. It represented 78% of all cases in 2011 and both of the two cases filed in early 2012.
Hair removal was the most frequent laser procedure involved in litigation. A total of 76% of the cases brought for hair removal–associated injury in 2004-2012 involved nonphysician operators. Again, this number is markedly higher when the data are categorized by time period: 86% of all lawsuits in 2008-2012 and 91% of all lawsuits in 2010-2012 involved nonphysicians performing laser hair removal.
When the data were analyzed by the location where laser procedures took place, the majority (64%) of cases involving nonphysician operators occurred at facilities such as spas and salons. Again, in more recent years (2008-2012), "nonphysician-operator procedures performed in ‘medical spas’ represented 77% of lawsuits," Dr. Jalian and his associates said (JAMA Dermatology 2013 Oct. 16 [doi:10.1001/jamadermatol.2013. 7117]).
Clinicians also should be aware that almost all the malpractice cases arising from nonphysician operators’ negligence extend beyond the operator to his or her employer, be that a physician, a medical facility, or a spa.
By far the most common allegation against physicians was failure to supervise a nonphysician operator (27 cases), followed by failure to train or hire appropriate staff (23 cases).
The investigators noted that "the actual data likely understate the true incidence of nonphysician operator laser complications" since they were culled from the large legal database that excluded complaints that were handled outside the judicial system.
Laws regulating the use of cutaneous lasers vary widely by state, from edicts specifying that only physicians may operate lasers for hair removal (Maine) to no regulations at all on laser use (Nevada). Even when clinicians supervise so-called physician extenders, rules concerning the degree of supervision, protocol requirements, and employee training are ambiguous.
"Because laws and regulations are constantly evolving, it is imperative for [those] who use physician extenders to be up to date," the researchers said.
Dr. Jalian reported no relevant financial conflicts of interest. One of his associates reported ties to Zeltiq Aesthetics, Cytrellis Biosystems, Unilever, and Allergan.
Litigation concerning cutaneous laser surgery has surged in recent years, and the proportion involving nonphysician operators soared from 36% in 1999 to 78% in 2012, according to a report published online Oct. 16 in JAMA Dermatology.
For the most commonly performed laser procedure – hair removal – the proportion of lawsuits involving nonphysician operators is even higher. A "remarkable" 91% of the cases filed in the most recent year for which complete data are available were brought against nonphysician operators, said Dr. H. Ray Jalian of the division of dermatology, University of California, Los Angeles, and his associates.
"The overall trend in increased litigation for laser surgery is in part explained by greater numbers of nonphysician operators performing these procedures, in particular those practicing without direct supervision in ‘medical spas,’ " the investigators said.
Dr. Jalian and his colleagues searched a single legal database and identified 175 cases concerning injury arising from cutaneous laser surgery since 1999. Nonphysician operators included registered nurses, nurse practitioners, medical assistants, electrologists, technicians, aestheticians, and others.
Overall, nonphysician operators were sued in 43% of all cases. But this total is somewhat misleading, because that percentage has risen markedly over time. It represented 78% of all cases in 2011 and both of the two cases filed in early 2012.
Hair removal was the most frequent laser procedure involved in litigation. A total of 76% of the cases brought for hair removal–associated injury in 2004-2012 involved nonphysician operators. Again, this number is markedly higher when the data are categorized by time period: 86% of all lawsuits in 2008-2012 and 91% of all lawsuits in 2010-2012 involved nonphysicians performing laser hair removal.
When the data were analyzed by the location where laser procedures took place, the majority (64%) of cases involving nonphysician operators occurred at facilities such as spas and salons. Again, in more recent years (2008-2012), "nonphysician-operator procedures performed in ‘medical spas’ represented 77% of lawsuits," Dr. Jalian and his associates said (JAMA Dermatology 2013 Oct. 16 [doi:10.1001/jamadermatol.2013. 7117]).
Clinicians also should be aware that almost all the malpractice cases arising from nonphysician operators’ negligence extend beyond the operator to his or her employer, be that a physician, a medical facility, or a spa.
By far the most common allegation against physicians was failure to supervise a nonphysician operator (27 cases), followed by failure to train or hire appropriate staff (23 cases).
The investigators noted that "the actual data likely understate the true incidence of nonphysician operator laser complications" since they were culled from the large legal database that excluded complaints that were handled outside the judicial system.
Laws regulating the use of cutaneous lasers vary widely by state, from edicts specifying that only physicians may operate lasers for hair removal (Maine) to no regulations at all on laser use (Nevada). Even when clinicians supervise so-called physician extenders, rules concerning the degree of supervision, protocol requirements, and employee training are ambiguous.
"Because laws and regulations are constantly evolving, it is imperative for [those] who use physician extenders to be up to date," the researchers said.
Dr. Jalian reported no relevant financial conflicts of interest. One of his associates reported ties to Zeltiq Aesthetics, Cytrellis Biosystems, Unilever, and Allergan.
FROM JAMA DERMATOLOGY
Major finding: The proportion of lawsuits concerning cutaneous laser procedures that have been brought against nonphysician operators rose from 36% in 1999 to 78% in 2012.
Data source: A retrospective review of 175 legal claims concerning cutaneous laser surgery that were brought throughout the United States in 1999-2012.
Disclosures: Dr. Jalian reported no relevant financial conflicts of interest. One of his associates reported ties to Zeltiq Aesthetics, Cytrellis Biosystems, Unilever, and Allergan.
Bracing effective for idiopathic scoliosis in adolescence
Use of a rigid back brace was significantly more effective than observation alone in preventing the progression of idiopathic scoliosis among adolescents who were at high risk of progressing to the point at which surgery would be warranted, according to a report in the Oct. 17 issue of the New England Journal of Medicine.
Moreover, this benefit increased as the average daily hours of wearing the back brace increased, indicating a significant dose-response effect, said Dr. Stuart L. Weinstein of the department of orthopedics and rehabilitation, University of Iowa, Iowa City, and his associates.
Although rigid bracing (thoracolumbosacral orthosis) is common in adolescent idiopathic scoliosis, there are little data supporting this practice. Until now, most studies of the issue have been observational and have yielded inconsistent results; only one prospective study has compared patients who wore back braces with patients who did not, the investigators said.
"Our findings have direct clinical applicability because they are derived from assessment of a group of patients for whom bracing would have been recommended in a typical orthopedic practice but in the absence of rigorous supporting data," they noted (N. Engl. J. Med. 2013;369:1512-21).
Dr. Weinstein and his colleagues performed the Bracing in Adolescent Idiopathic Scoliosis Trial (BRAIST) to compare bracing against observation alone in preventing the progression of scoliosis to 50 degrees or more, a common indication for surgery. They assessed 383 treatment-naive patients aged 10-15 years who attended 25 medical centers across the United States and Canada in 2007-2011.
In all the study subjects, the Cobb angle for the largest curve was 20-40 degrees at baseline.
Enrollment in the randomized trial proved difficult because so many families were unwilling to assign a patient to one group or the other, so one portion of the trial involving 155 patients was randomized (the randomized cohort), while another portion involving 228 patients was allowed to choose group assignment (the preference cohort).
Patients in the bracing groups were instructed to wear their rigid thoracolumbosacral braces for a minimum of 18 hours a day until they reached skeletal maturity. Adherence was measured by the use of a temperature sensor imbedded in the device, which indicated by the presence or absence of body heat how long the brace was worn.
The study could not be blinded because both patients and physicians were aware of treatment assignment; however, all radiographic evaluations and the final determination of patient outcomes were done by a musculoskeletal radiologist and a research associate who were blinded to treatment assignment.
The trial was terminated early when an interim analysis demonstrated a clear benefit in favor of bracing, with an odds ratio of 2.03.
The primary outcome was progression of the curvature to 50 degrees or more, which signaled treatment failure, or attainment of skeletal maturity without progression to 50 degrees or more of curvature, which signaled treatment success.
A total of 242 patients were included in the primary analysis: 51 patients assigned to back bracing, 65 assigned to observation, 88 who chose back bracing, and 38 who chose observation. After an average follow-up of 21-23 months, the rate of treatment success was 72% with bracing and 48% with observation alone in this analysis, Dr. Weinstein and his associates said.
In the intention-to-treat analysis, the rate of treatment success was 75% in patients randomly assigned to bracing, compared with 42% in those randomly assigned to observation only. This represents a relative risk reduction of 56% with bracing.
The number needed to treat to prevent a single case of curve progression warranting surgery was 3.
Duration of brace wear positively correlated with treatment success. Patients in the lowest quartile of brace wear (mean of 0-6 hours of wear daily) had a treatment success rate of 41%, very similar to that in the observation groups. In contrast, patients who wore their braces for an average of at least 13 hours per day had a treatment success rate of 90%-93%.
Scores on measures of general quality of life were not significantly different between the bracing and observation groups at baseline or at the final follow-up assessment.
The number of adverse events also was no different between the two study groups, and the same percentage of patients in both groups reported the most common adverse event (back pain). Eight percent of patients who wore a brace reported skin problems related to the device. The only serious adverse event occurred in a patient who wore a brace, who sought hospitalization for anxiety and depression.
BRAIST was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Children’s Miracle Network, Canadian Institutes of Health Research, Shriners Hospitals for Children, the University of Rochester, and Children’s Mercy Hospitals and Clinics. Dr. James G. Wright reported other support as the R.B. Salter Chair in Surgical Research during the conduct of the study. No financial conflicts of interest were reported.
The investigators found that brace treatment was effective, and the longer that brace was worn each day, the better the outcome.
An equally important finding of this study was that so many growing children with adolescent idiopathic scoliosis "seem to do just fine with no treatment at all," said Dr. Eugene J. Carragee and Dr. Ronald A. Lehman Jr.
Nearly half (48%) of the untreated patients in this study had a successful outcome, as did 41% of the "treated" patients who actually spent little time wearing their braces.
Thus, "the bracing indications described are probably too broad, resulting in what may be unnecessary treatment for many patients," Dr. Carragee and Dr. Lehman said. Although there do not appear to be physiological side effects to bracing in adolescent idiopathic scoliosis, "it carries financial, emotional, and social burdens that need to be considered."
"The challenge in the field going forward is to identify children who are most likely to benefit from bracing and those who are unlikely to benefit," they concluded.
Dr. Eugene J. Carragee is with Stanford (Calif.) University. Dr. Ronald A. Lehman Jr. is with Walter Reed National Military Medical Center in Bethesda, Md. Dr. Lehman reported receiving grants from DePuy Spine, Centinel Spine, the Defense Medical Research and Development Program, and Defense Advanced Research Projects Agency apart from this work; no other potential financial conflicts of interest were reported. These remarks were taken from an editorial accompanying Dr. Weinstein’s report (N. Engl. J. Med. 2013;369:1558-60).
The investigators found that brace treatment was effective, and the longer that brace was worn each day, the better the outcome.
An equally important finding of this study was that so many growing children with adolescent idiopathic scoliosis "seem to do just fine with no treatment at all," said Dr. Eugene J. Carragee and Dr. Ronald A. Lehman Jr.
Nearly half (48%) of the untreated patients in this study had a successful outcome, as did 41% of the "treated" patients who actually spent little time wearing their braces.
Thus, "the bracing indications described are probably too broad, resulting in what may be unnecessary treatment for many patients," Dr. Carragee and Dr. Lehman said. Although there do not appear to be physiological side effects to bracing in adolescent idiopathic scoliosis, "it carries financial, emotional, and social burdens that need to be considered."
"The challenge in the field going forward is to identify children who are most likely to benefit from bracing and those who are unlikely to benefit," they concluded.
Dr. Eugene J. Carragee is with Stanford (Calif.) University. Dr. Ronald A. Lehman Jr. is with Walter Reed National Military Medical Center in Bethesda, Md. Dr. Lehman reported receiving grants from DePuy Spine, Centinel Spine, the Defense Medical Research and Development Program, and Defense Advanced Research Projects Agency apart from this work; no other potential financial conflicts of interest were reported. These remarks were taken from an editorial accompanying Dr. Weinstein’s report (N. Engl. J. Med. 2013;369:1558-60).
The investigators found that brace treatment was effective, and the longer that brace was worn each day, the better the outcome.
An equally important finding of this study was that so many growing children with adolescent idiopathic scoliosis "seem to do just fine with no treatment at all," said Dr. Eugene J. Carragee and Dr. Ronald A. Lehman Jr.
Nearly half (48%) of the untreated patients in this study had a successful outcome, as did 41% of the "treated" patients who actually spent little time wearing their braces.
Thus, "the bracing indications described are probably too broad, resulting in what may be unnecessary treatment for many patients," Dr. Carragee and Dr. Lehman said. Although there do not appear to be physiological side effects to bracing in adolescent idiopathic scoliosis, "it carries financial, emotional, and social burdens that need to be considered."
"The challenge in the field going forward is to identify children who are most likely to benefit from bracing and those who are unlikely to benefit," they concluded.
Dr. Eugene J. Carragee is with Stanford (Calif.) University. Dr. Ronald A. Lehman Jr. is with Walter Reed National Military Medical Center in Bethesda, Md. Dr. Lehman reported receiving grants from DePuy Spine, Centinel Spine, the Defense Medical Research and Development Program, and Defense Advanced Research Projects Agency apart from this work; no other potential financial conflicts of interest were reported. These remarks were taken from an editorial accompanying Dr. Weinstein’s report (N. Engl. J. Med. 2013;369:1558-60).
Use of a rigid back brace was significantly more effective than observation alone in preventing the progression of idiopathic scoliosis among adolescents who were at high risk of progressing to the point at which surgery would be warranted, according to a report in the Oct. 17 issue of the New England Journal of Medicine.
Moreover, this benefit increased as the average daily hours of wearing the back brace increased, indicating a significant dose-response effect, said Dr. Stuart L. Weinstein of the department of orthopedics and rehabilitation, University of Iowa, Iowa City, and his associates.
Although rigid bracing (thoracolumbosacral orthosis) is common in adolescent idiopathic scoliosis, there are little data supporting this practice. Until now, most studies of the issue have been observational and have yielded inconsistent results; only one prospective study has compared patients who wore back braces with patients who did not, the investigators said.
"Our findings have direct clinical applicability because they are derived from assessment of a group of patients for whom bracing would have been recommended in a typical orthopedic practice but in the absence of rigorous supporting data," they noted (N. Engl. J. Med. 2013;369:1512-21).
Dr. Weinstein and his colleagues performed the Bracing in Adolescent Idiopathic Scoliosis Trial (BRAIST) to compare bracing against observation alone in preventing the progression of scoliosis to 50 degrees or more, a common indication for surgery. They assessed 383 treatment-naive patients aged 10-15 years who attended 25 medical centers across the United States and Canada in 2007-2011.
In all the study subjects, the Cobb angle for the largest curve was 20-40 degrees at baseline.
Enrollment in the randomized trial proved difficult because so many families were unwilling to assign a patient to one group or the other, so one portion of the trial involving 155 patients was randomized (the randomized cohort), while another portion involving 228 patients was allowed to choose group assignment (the preference cohort).
Patients in the bracing groups were instructed to wear their rigid thoracolumbosacral braces for a minimum of 18 hours a day until they reached skeletal maturity. Adherence was measured by the use of a temperature sensor imbedded in the device, which indicated by the presence or absence of body heat how long the brace was worn.
The study could not be blinded because both patients and physicians were aware of treatment assignment; however, all radiographic evaluations and the final determination of patient outcomes were done by a musculoskeletal radiologist and a research associate who were blinded to treatment assignment.
The trial was terminated early when an interim analysis demonstrated a clear benefit in favor of bracing, with an odds ratio of 2.03.
The primary outcome was progression of the curvature to 50 degrees or more, which signaled treatment failure, or attainment of skeletal maturity without progression to 50 degrees or more of curvature, which signaled treatment success.
A total of 242 patients were included in the primary analysis: 51 patients assigned to back bracing, 65 assigned to observation, 88 who chose back bracing, and 38 who chose observation. After an average follow-up of 21-23 months, the rate of treatment success was 72% with bracing and 48% with observation alone in this analysis, Dr. Weinstein and his associates said.
In the intention-to-treat analysis, the rate of treatment success was 75% in patients randomly assigned to bracing, compared with 42% in those randomly assigned to observation only. This represents a relative risk reduction of 56% with bracing.
The number needed to treat to prevent a single case of curve progression warranting surgery was 3.
Duration of brace wear positively correlated with treatment success. Patients in the lowest quartile of brace wear (mean of 0-6 hours of wear daily) had a treatment success rate of 41%, very similar to that in the observation groups. In contrast, patients who wore their braces for an average of at least 13 hours per day had a treatment success rate of 90%-93%.
Scores on measures of general quality of life were not significantly different between the bracing and observation groups at baseline or at the final follow-up assessment.
The number of adverse events also was no different between the two study groups, and the same percentage of patients in both groups reported the most common adverse event (back pain). Eight percent of patients who wore a brace reported skin problems related to the device. The only serious adverse event occurred in a patient who wore a brace, who sought hospitalization for anxiety and depression.
BRAIST was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Children’s Miracle Network, Canadian Institutes of Health Research, Shriners Hospitals for Children, the University of Rochester, and Children’s Mercy Hospitals and Clinics. Dr. James G. Wright reported other support as the R.B. Salter Chair in Surgical Research during the conduct of the study. No financial conflicts of interest were reported.
Use of a rigid back brace was significantly more effective than observation alone in preventing the progression of idiopathic scoliosis among adolescents who were at high risk of progressing to the point at which surgery would be warranted, according to a report in the Oct. 17 issue of the New England Journal of Medicine.
Moreover, this benefit increased as the average daily hours of wearing the back brace increased, indicating a significant dose-response effect, said Dr. Stuart L. Weinstein of the department of orthopedics and rehabilitation, University of Iowa, Iowa City, and his associates.
Although rigid bracing (thoracolumbosacral orthosis) is common in adolescent idiopathic scoliosis, there are little data supporting this practice. Until now, most studies of the issue have been observational and have yielded inconsistent results; only one prospective study has compared patients who wore back braces with patients who did not, the investigators said.
"Our findings have direct clinical applicability because they are derived from assessment of a group of patients for whom bracing would have been recommended in a typical orthopedic practice but in the absence of rigorous supporting data," they noted (N. Engl. J. Med. 2013;369:1512-21).
Dr. Weinstein and his colleagues performed the Bracing in Adolescent Idiopathic Scoliosis Trial (BRAIST) to compare bracing against observation alone in preventing the progression of scoliosis to 50 degrees or more, a common indication for surgery. They assessed 383 treatment-naive patients aged 10-15 years who attended 25 medical centers across the United States and Canada in 2007-2011.
In all the study subjects, the Cobb angle for the largest curve was 20-40 degrees at baseline.
Enrollment in the randomized trial proved difficult because so many families were unwilling to assign a patient to one group or the other, so one portion of the trial involving 155 patients was randomized (the randomized cohort), while another portion involving 228 patients was allowed to choose group assignment (the preference cohort).
Patients in the bracing groups were instructed to wear their rigid thoracolumbosacral braces for a minimum of 18 hours a day until they reached skeletal maturity. Adherence was measured by the use of a temperature sensor imbedded in the device, which indicated by the presence or absence of body heat how long the brace was worn.
The study could not be blinded because both patients and physicians were aware of treatment assignment; however, all radiographic evaluations and the final determination of patient outcomes were done by a musculoskeletal radiologist and a research associate who were blinded to treatment assignment.
The trial was terminated early when an interim analysis demonstrated a clear benefit in favor of bracing, with an odds ratio of 2.03.
The primary outcome was progression of the curvature to 50 degrees or more, which signaled treatment failure, or attainment of skeletal maturity without progression to 50 degrees or more of curvature, which signaled treatment success.
A total of 242 patients were included in the primary analysis: 51 patients assigned to back bracing, 65 assigned to observation, 88 who chose back bracing, and 38 who chose observation. After an average follow-up of 21-23 months, the rate of treatment success was 72% with bracing and 48% with observation alone in this analysis, Dr. Weinstein and his associates said.
In the intention-to-treat analysis, the rate of treatment success was 75% in patients randomly assigned to bracing, compared with 42% in those randomly assigned to observation only. This represents a relative risk reduction of 56% with bracing.
The number needed to treat to prevent a single case of curve progression warranting surgery was 3.
Duration of brace wear positively correlated with treatment success. Patients in the lowest quartile of brace wear (mean of 0-6 hours of wear daily) had a treatment success rate of 41%, very similar to that in the observation groups. In contrast, patients who wore their braces for an average of at least 13 hours per day had a treatment success rate of 90%-93%.
Scores on measures of general quality of life were not significantly different between the bracing and observation groups at baseline or at the final follow-up assessment.
The number of adverse events also was no different between the two study groups, and the same percentage of patients in both groups reported the most common adverse event (back pain). Eight percent of patients who wore a brace reported skin problems related to the device. The only serious adverse event occurred in a patient who wore a brace, who sought hospitalization for anxiety and depression.
BRAIST was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Children’s Miracle Network, Canadian Institutes of Health Research, Shriners Hospitals for Children, the University of Rochester, and Children’s Mercy Hospitals and Clinics. Dr. James G. Wright reported other support as the R.B. Salter Chair in Surgical Research during the conduct of the study. No financial conflicts of interest were reported.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: In the intention-to-treat analysis, the rate of treatment success was 75% with bracing vs. 42% with observation alone, and the number needed to treat to prevent one case of scoliosis from progressing to surgery was 3.
Data source: A multicenter cohort study of the effectiveness of rigid thoracolumbosacral orthosis in preventing progression of idiopathic scoliosis in 383 adolescents followed for approximately 2 years.
Disclosures: BRAIST was supported by the National Institute of Arthritis and Musculoskeletal and Skin Diseases, Children’s Miracle Network, Canadian Institutes of Health Research, Shriners Hospitals for Children, the University of Rochester, and Children’s Mercy Hospitals and Clinics. Dr. James G. Wright reported other support as the R.B. Salter Chair in Surgical Research during the conduct of the study. No financial conflicts of interest were reported.
Safety of metoclopramide in pregnancy affirmed
The use of metoclopramide during pregnancy was not associated with any increased risk of major congenital malformations, spontaneous abortion, stillbirth, preterm birth, low birth weight, or fetal growth restriction in a study assessing 10 times as many exposed pregnancies as all previous cohort studies of this issue combined.
This registry-based cohort study assessed 1,222,503 pregnancies throughout Denmark from 1997 through 2011, including 45,002 (3.7%) in which the mother took metoclopramide, presumably to treat nausea or vomiting. It examined 20 different categories of congenital malformation, said Dr. Björn Pasternak and his associates of the Statens Serum Institut, Copenhagen.
"The number of included pregnancies in our study permitted analyses with precise estimation of risk for most outcomes and allowed analyses of serious adverse outcomes that are rare," the authors noted in a report online Oct. 15 in JAMA.
The findings confirm and significantly extend those of previously published cohort studies of metoclopramide taken during pregnancy, which included only 4,261 exposed pregnancies in total.
Dr. Pasternak and his colleagues first assessed major congenital malformations in 28,486 liveborn infants exposed to metoclopramide during the first trimester and 113,698 infants who were not exposed and who were matched for age, year of birth, and propensity score. The rate of any major malformation was 25.3 per 1,000 among the exposed infants and 26.6 per 1,000 among the unexposed infants, a nonsignificant difference.
They then analyzed 20 individual categories of congenital malformation, such as neural tube defects, hydronephrosis, ventricular or atrial septal defects, hypospadias, clubfoot, or cleft lip. There were no significant associations between metoclopramide and any type of malformation.
The researchers also assessed 10,171 cases of spontaneous abortion that occurred among 37,946 metoclopramide-exposed pregnancies and 151,661 matched unexposed pregnancies. The rate of spontaneous abortion was slightly lower in exposed than in unexposed pregnancies.
This finding was "not unexpected" because the drug is given to treat nausea and vomiting, conditions that are known to correlate with lower rates of spontaneous abortion in the general population. "Indeed, the strength of the association between nausea and vomiting and spontaneous abortion in previous studies was similar in magnitude to the association between metoclopramide exposure and spontaneous abortion observed in our study," Dr. Pasternak and his associates reported (JAMA 2013;310:1601-11).
They also found that the rate of stillbirth among 40,306 metoclopramide-exposed pregnancies (3.5 per 1,000) was not significantly different from that among 161,098 matched unexposed pregnancies (3.9 per 1,000). So the drug did not raise the risk of stillbirth.
Further analyses showed that metoclopramide exposure was not associated with any of the secondary outcomes of preterm birth, low birth weight, or small-for-gestational-age infants.
The investigators also performed several sensitivity analyses, all of which yielded results similar to those of the primary analyses. For example, the rates of all adverse outcomes were not significantly different between women who filled only one prescription for the drug and those who filled multiple prescriptions, indicating that there was no dose-response effect.
They also assessed a possible exposure effect among pregnancies that were terminated because of major fetal malformations. Again, there was no evidence of an increased risk of malformations overall or of particular types of malformations after exposure to metoclopramide.
These findings "may help inform clinical decisions when treatment with metoclopramide is considered in pregnancy," Dr. Pasternak and his associates said.
This study was supported by the Danish Medical Research Council. No financial conflicts of interest were reported.
The use of metoclopramide during pregnancy was not associated with any increased risk of major congenital malformations, spontaneous abortion, stillbirth, preterm birth, low birth weight, or fetal growth restriction in a study assessing 10 times as many exposed pregnancies as all previous cohort studies of this issue combined.
This registry-based cohort study assessed 1,222,503 pregnancies throughout Denmark from 1997 through 2011, including 45,002 (3.7%) in which the mother took metoclopramide, presumably to treat nausea or vomiting. It examined 20 different categories of congenital malformation, said Dr. Björn Pasternak and his associates of the Statens Serum Institut, Copenhagen.
"The number of included pregnancies in our study permitted analyses with precise estimation of risk for most outcomes and allowed analyses of serious adverse outcomes that are rare," the authors noted in a report online Oct. 15 in JAMA.
The findings confirm and significantly extend those of previously published cohort studies of metoclopramide taken during pregnancy, which included only 4,261 exposed pregnancies in total.
Dr. Pasternak and his colleagues first assessed major congenital malformations in 28,486 liveborn infants exposed to metoclopramide during the first trimester and 113,698 infants who were not exposed and who were matched for age, year of birth, and propensity score. The rate of any major malformation was 25.3 per 1,000 among the exposed infants and 26.6 per 1,000 among the unexposed infants, a nonsignificant difference.
They then analyzed 20 individual categories of congenital malformation, such as neural tube defects, hydronephrosis, ventricular or atrial septal defects, hypospadias, clubfoot, or cleft lip. There were no significant associations between metoclopramide and any type of malformation.
The researchers also assessed 10,171 cases of spontaneous abortion that occurred among 37,946 metoclopramide-exposed pregnancies and 151,661 matched unexposed pregnancies. The rate of spontaneous abortion was slightly lower in exposed than in unexposed pregnancies.
This finding was "not unexpected" because the drug is given to treat nausea and vomiting, conditions that are known to correlate with lower rates of spontaneous abortion in the general population. "Indeed, the strength of the association between nausea and vomiting and spontaneous abortion in previous studies was similar in magnitude to the association between metoclopramide exposure and spontaneous abortion observed in our study," Dr. Pasternak and his associates reported (JAMA 2013;310:1601-11).
They also found that the rate of stillbirth among 40,306 metoclopramide-exposed pregnancies (3.5 per 1,000) was not significantly different from that among 161,098 matched unexposed pregnancies (3.9 per 1,000). So the drug did not raise the risk of stillbirth.
Further analyses showed that metoclopramide exposure was not associated with any of the secondary outcomes of preterm birth, low birth weight, or small-for-gestational-age infants.
The investigators also performed several sensitivity analyses, all of which yielded results similar to those of the primary analyses. For example, the rates of all adverse outcomes were not significantly different between women who filled only one prescription for the drug and those who filled multiple prescriptions, indicating that there was no dose-response effect.
They also assessed a possible exposure effect among pregnancies that were terminated because of major fetal malformations. Again, there was no evidence of an increased risk of malformations overall or of particular types of malformations after exposure to metoclopramide.
These findings "may help inform clinical decisions when treatment with metoclopramide is considered in pregnancy," Dr. Pasternak and his associates said.
This study was supported by the Danish Medical Research Council. No financial conflicts of interest were reported.
The use of metoclopramide during pregnancy was not associated with any increased risk of major congenital malformations, spontaneous abortion, stillbirth, preterm birth, low birth weight, or fetal growth restriction in a study assessing 10 times as many exposed pregnancies as all previous cohort studies of this issue combined.
This registry-based cohort study assessed 1,222,503 pregnancies throughout Denmark from 1997 through 2011, including 45,002 (3.7%) in which the mother took metoclopramide, presumably to treat nausea or vomiting. It examined 20 different categories of congenital malformation, said Dr. Björn Pasternak and his associates of the Statens Serum Institut, Copenhagen.
"The number of included pregnancies in our study permitted analyses with precise estimation of risk for most outcomes and allowed analyses of serious adverse outcomes that are rare," the authors noted in a report online Oct. 15 in JAMA.
The findings confirm and significantly extend those of previously published cohort studies of metoclopramide taken during pregnancy, which included only 4,261 exposed pregnancies in total.
Dr. Pasternak and his colleagues first assessed major congenital malformations in 28,486 liveborn infants exposed to metoclopramide during the first trimester and 113,698 infants who were not exposed and who were matched for age, year of birth, and propensity score. The rate of any major malformation was 25.3 per 1,000 among the exposed infants and 26.6 per 1,000 among the unexposed infants, a nonsignificant difference.
They then analyzed 20 individual categories of congenital malformation, such as neural tube defects, hydronephrosis, ventricular or atrial septal defects, hypospadias, clubfoot, or cleft lip. There were no significant associations between metoclopramide and any type of malformation.
The researchers also assessed 10,171 cases of spontaneous abortion that occurred among 37,946 metoclopramide-exposed pregnancies and 151,661 matched unexposed pregnancies. The rate of spontaneous abortion was slightly lower in exposed than in unexposed pregnancies.
This finding was "not unexpected" because the drug is given to treat nausea and vomiting, conditions that are known to correlate with lower rates of spontaneous abortion in the general population. "Indeed, the strength of the association between nausea and vomiting and spontaneous abortion in previous studies was similar in magnitude to the association between metoclopramide exposure and spontaneous abortion observed in our study," Dr. Pasternak and his associates reported (JAMA 2013;310:1601-11).
They also found that the rate of stillbirth among 40,306 metoclopramide-exposed pregnancies (3.5 per 1,000) was not significantly different from that among 161,098 matched unexposed pregnancies (3.9 per 1,000). So the drug did not raise the risk of stillbirth.
Further analyses showed that metoclopramide exposure was not associated with any of the secondary outcomes of preterm birth, low birth weight, or small-for-gestational-age infants.
The investigators also performed several sensitivity analyses, all of which yielded results similar to those of the primary analyses. For example, the rates of all adverse outcomes were not significantly different between women who filled only one prescription for the drug and those who filled multiple prescriptions, indicating that there was no dose-response effect.
They also assessed a possible exposure effect among pregnancies that were terminated because of major fetal malformations. Again, there was no evidence of an increased risk of malformations overall or of particular types of malformations after exposure to metoclopramide.
These findings "may help inform clinical decisions when treatment with metoclopramide is considered in pregnancy," Dr. Pasternak and his associates said.
This study was supported by the Danish Medical Research Council. No financial conflicts of interest were reported.
FROM JAMA
Major finding: The rate of any major congenital malformation was 25.3 per 1,000 among infants exposed to metoclopramide during the first trimester and 26.6 per 1,000 among unexposed infants, a nonsignificant difference.
Data source: A nationwide cohort study of 1,222,503 pregnancies throughout Denmark in 1997-2011, in which 45,002 mothers took metoclopramide and the remainder did not.
Disclosures: This study was supported by the Danish Medical Research Council. No financial conflicts of interest were reported.
CABG edges PCI in quality-of-life measures for diabetes patients
For diabetic patients who have multivessel coronary artery disease, bypass surgery provides slightly better quality of life and cardiovascular-related health status than does stenting for roughly 2 years, according to a substudy of the FREEDOM trial reported online Oct. 15 in JAMA.
Beyond 2 years, there are no significant differences between the two approaches regarding health status and quality of life in this patient group, said Dr. Mouin S. Abdallah of St. Luke’s Mid America Heart Institute, Kansas City, Mo., and his associates.
Both revascularization strategies yield substantial and sustained improvements for patients who have concomitant multivessel coronary artery disease (CAD) and diabetes, but coronary artery bypass graft surgery generally is preferred because it has a small but significant edge in reducing morbidity and mortality, is less expensive, and produces markedly more durable results. However, the risk of stroke is higher with CABG, and it requires a longer recovery period because it is more invasive, "which may be particularly relevant to patients who are more concerned about quality rather than duration of life," the investigators noted.
"For such patients, our study provides reassurance that there are not major differences in long-term health status and quality of life between the two treatment strategies," they said.
Dr. Abdallah and his colleagues performed a prospective substudy of quality-of-life issues alongside the FREEDOM (Future Revascularization Evaluation in Patients With Diabetes Mellitus: Optimal Management of Multivessel Disease) clinical trial. In FREEDOM, 1,900 patients from 18 countries were randomly assigned to undergo either CABG or percutaneous coronary intervention with drug-eluting stents during 2005-2010.
For their substudy, Dr. Abdallah and his associates assessed 935 participants who were assigned to CABG and 945 assigned to PCI. The mean patient age was 63 years, and 72% were men. Median follow-up was 44-47 months.
Patients in both study groups reported substantial and long-lasting improvements in cardiovascular-specific health status, as measured by the Seattle Angina Questionnaire and the Rose Dyspnea Scale. Scores on these instruments improved markedly within 1 month of both procedures and remained high throughout follow-up.
Patients in the PCI group showed more rapid improvement following the procedure, "but these benefits were transient and largely restricted to the first month of follow-up," the researchers said.
"Between 6 months and 2 years, health status was slightly better with CABG across a range of cardiac-specific domains including angina relief, physical function, and overall quality of life. Beyond 2 years, there were no consistent differences in any health status or quality-of-life domains between the CABG and PCI strategies," they reported (JAMA 2013;310:1581-90).
For example, the proportion of angina-free patients was slightly but significantly greater with CABG than with PCI at 6 months (83.7% vs. 78.1%) and at 12 months (83.5% vs. 79.5%), but was not significantly different thereafter.
Similarly, measures of physical limitations imposed by CAD were "modestly" higher with CABG than with PCI for 3 years after the procedure, but there were no significant between-group differences after year 3.
And dyspnea improved faster after PCI than after CABG, but by 6 months this difference had disappeared. By 1 year, the proportion of patients who reported moderate dyspnea was only 9% in both groups, and that proportion stayed fairly steady at 10%-12% in both groups for the remainder of follow-up.
The findings were similar in a sensitivity analysis and in a further analysis restricted only to patients who had reported daily or weekly angina at baseline.
In the FREEDOM trial, CABG showed a clear benefit over PCI for the composite endpoint of death, myocardial infarction, or stroke in patients with concomitant multivessel CAD and diabetes. CABG also afforded slightly better angina relief, especially in patients who had the most severe angina at baseline.
Moreover, patients in the PCI group were more likely to require continuing antianginal medication and twice as likely to undergo repeat revascularization procedures than those in the CABG group.
However, the study findings demonstrate that PCI is clearly beneficial for patients who want to avoid the acute risks of CABG surgery, and is an excellent alternative for those who want a less invasive treatment, Dr. Abdallah and his associates said.
This study was supported by the National Heart, Lung, and Blood Institute. Cordis and Boston Scientific provided the drug-eluting stents, Eli Lilly provided abciximab and research funds, and Sanofi-Aventis and Bristol-Myers Squibb provided clopidogrel. Dr. Abdallah reported no relevant financial conflicts of interest; his associates reported numerous ties to industry sources.
For diabetic patients who have multivessel coronary artery disease, bypass surgery provides slightly better quality of life and cardiovascular-related health status than does stenting for roughly 2 years, according to a substudy of the FREEDOM trial reported online Oct. 15 in JAMA.
Beyond 2 years, there are no significant differences between the two approaches regarding health status and quality of life in this patient group, said Dr. Mouin S. Abdallah of St. Luke’s Mid America Heart Institute, Kansas City, Mo., and his associates.
Both revascularization strategies yield substantial and sustained improvements for patients who have concomitant multivessel coronary artery disease (CAD) and diabetes, but coronary artery bypass graft surgery generally is preferred because it has a small but significant edge in reducing morbidity and mortality, is less expensive, and produces markedly more durable results. However, the risk of stroke is higher with CABG, and it requires a longer recovery period because it is more invasive, "which may be particularly relevant to patients who are more concerned about quality rather than duration of life," the investigators noted.
"For such patients, our study provides reassurance that there are not major differences in long-term health status and quality of life between the two treatment strategies," they said.
Dr. Abdallah and his colleagues performed a prospective substudy of quality-of-life issues alongside the FREEDOM (Future Revascularization Evaluation in Patients With Diabetes Mellitus: Optimal Management of Multivessel Disease) clinical trial. In FREEDOM, 1,900 patients from 18 countries were randomly assigned to undergo either CABG or percutaneous coronary intervention with drug-eluting stents during 2005-2010.
For their substudy, Dr. Abdallah and his associates assessed 935 participants who were assigned to CABG and 945 assigned to PCI. The mean patient age was 63 years, and 72% were men. Median follow-up was 44-47 months.
Patients in both study groups reported substantial and long-lasting improvements in cardiovascular-specific health status, as measured by the Seattle Angina Questionnaire and the Rose Dyspnea Scale. Scores on these instruments improved markedly within 1 month of both procedures and remained high throughout follow-up.
Patients in the PCI group showed more rapid improvement following the procedure, "but these benefits were transient and largely restricted to the first month of follow-up," the researchers said.
"Between 6 months and 2 years, health status was slightly better with CABG across a range of cardiac-specific domains including angina relief, physical function, and overall quality of life. Beyond 2 years, there were no consistent differences in any health status or quality-of-life domains between the CABG and PCI strategies," they reported (JAMA 2013;310:1581-90).
For example, the proportion of angina-free patients was slightly but significantly greater with CABG than with PCI at 6 months (83.7% vs. 78.1%) and at 12 months (83.5% vs. 79.5%), but was not significantly different thereafter.
Similarly, measures of physical limitations imposed by CAD were "modestly" higher with CABG than with PCI for 3 years after the procedure, but there were no significant between-group differences after year 3.
And dyspnea improved faster after PCI than after CABG, but by 6 months this difference had disappeared. By 1 year, the proportion of patients who reported moderate dyspnea was only 9% in both groups, and that proportion stayed fairly steady at 10%-12% in both groups for the remainder of follow-up.
The findings were similar in a sensitivity analysis and in a further analysis restricted only to patients who had reported daily or weekly angina at baseline.
In the FREEDOM trial, CABG showed a clear benefit over PCI for the composite endpoint of death, myocardial infarction, or stroke in patients with concomitant multivessel CAD and diabetes. CABG also afforded slightly better angina relief, especially in patients who had the most severe angina at baseline.
Moreover, patients in the PCI group were more likely to require continuing antianginal medication and twice as likely to undergo repeat revascularization procedures than those in the CABG group.
However, the study findings demonstrate that PCI is clearly beneficial for patients who want to avoid the acute risks of CABG surgery, and is an excellent alternative for those who want a less invasive treatment, Dr. Abdallah and his associates said.
This study was supported by the National Heart, Lung, and Blood Institute. Cordis and Boston Scientific provided the drug-eluting stents, Eli Lilly provided abciximab and research funds, and Sanofi-Aventis and Bristol-Myers Squibb provided clopidogrel. Dr. Abdallah reported no relevant financial conflicts of interest; his associates reported numerous ties to industry sources.
For diabetic patients who have multivessel coronary artery disease, bypass surgery provides slightly better quality of life and cardiovascular-related health status than does stenting for roughly 2 years, according to a substudy of the FREEDOM trial reported online Oct. 15 in JAMA.
Beyond 2 years, there are no significant differences between the two approaches regarding health status and quality of life in this patient group, said Dr. Mouin S. Abdallah of St. Luke’s Mid America Heart Institute, Kansas City, Mo., and his associates.
Both revascularization strategies yield substantial and sustained improvements for patients who have concomitant multivessel coronary artery disease (CAD) and diabetes, but coronary artery bypass graft surgery generally is preferred because it has a small but significant edge in reducing morbidity and mortality, is less expensive, and produces markedly more durable results. However, the risk of stroke is higher with CABG, and it requires a longer recovery period because it is more invasive, "which may be particularly relevant to patients who are more concerned about quality rather than duration of life," the investigators noted.
"For such patients, our study provides reassurance that there are not major differences in long-term health status and quality of life between the two treatment strategies," they said.
Dr. Abdallah and his colleagues performed a prospective substudy of quality-of-life issues alongside the FREEDOM (Future Revascularization Evaluation in Patients With Diabetes Mellitus: Optimal Management of Multivessel Disease) clinical trial. In FREEDOM, 1,900 patients from 18 countries were randomly assigned to undergo either CABG or percutaneous coronary intervention with drug-eluting stents during 2005-2010.
For their substudy, Dr. Abdallah and his associates assessed 935 participants who were assigned to CABG and 945 assigned to PCI. The mean patient age was 63 years, and 72% were men. Median follow-up was 44-47 months.
Patients in both study groups reported substantial and long-lasting improvements in cardiovascular-specific health status, as measured by the Seattle Angina Questionnaire and the Rose Dyspnea Scale. Scores on these instruments improved markedly within 1 month of both procedures and remained high throughout follow-up.
Patients in the PCI group showed more rapid improvement following the procedure, "but these benefits were transient and largely restricted to the first month of follow-up," the researchers said.
"Between 6 months and 2 years, health status was slightly better with CABG across a range of cardiac-specific domains including angina relief, physical function, and overall quality of life. Beyond 2 years, there were no consistent differences in any health status or quality-of-life domains between the CABG and PCI strategies," they reported (JAMA 2013;310:1581-90).
For example, the proportion of angina-free patients was slightly but significantly greater with CABG than with PCI at 6 months (83.7% vs. 78.1%) and at 12 months (83.5% vs. 79.5%), but was not significantly different thereafter.
Similarly, measures of physical limitations imposed by CAD were "modestly" higher with CABG than with PCI for 3 years after the procedure, but there were no significant between-group differences after year 3.
And dyspnea improved faster after PCI than after CABG, but by 6 months this difference had disappeared. By 1 year, the proportion of patients who reported moderate dyspnea was only 9% in both groups, and that proportion stayed fairly steady at 10%-12% in both groups for the remainder of follow-up.
The findings were similar in a sensitivity analysis and in a further analysis restricted only to patients who had reported daily or weekly angina at baseline.
In the FREEDOM trial, CABG showed a clear benefit over PCI for the composite endpoint of death, myocardial infarction, or stroke in patients with concomitant multivessel CAD and diabetes. CABG also afforded slightly better angina relief, especially in patients who had the most severe angina at baseline.
Moreover, patients in the PCI group were more likely to require continuing antianginal medication and twice as likely to undergo repeat revascularization procedures than those in the CABG group.
However, the study findings demonstrate that PCI is clearly beneficial for patients who want to avoid the acute risks of CABG surgery, and is an excellent alternative for those who want a less invasive treatment, Dr. Abdallah and his associates said.
This study was supported by the National Heart, Lung, and Blood Institute. Cordis and Boston Scientific provided the drug-eluting stents, Eli Lilly provided abciximab and research funds, and Sanofi-Aventis and Bristol-Myers Squibb provided clopidogrel. Dr. Abdallah reported no relevant financial conflicts of interest; his associates reported numerous ties to industry sources.
FROM JAMA
Major finding: The proportion of angina-free patients was slightly but significantly greater with CABG than with PCI at 6 months (83.7% vs. 78.1%) and 12 months (83.5% vs. 79.5%), but was not significantly different thereafter.
Data source: A prospective substudy of the international FREEDOM clinical trial comparing quality of life and CAD-related health status between 935 patients assigned to CABG and 945 assigned to PCI who were followed for a median of 4 years.
Disclosures: This study was supported by the National Heart, Lung, and Blood Institute. Cordis and Boston Scientific provided the drug-eluting stents, Eli Lilly provided abciximab and research funds, and Sanofi-Aventis and Bristol-Myers Squibb provided clopidogrel. Dr. Abdallah reported no relevant financial conflicts of interest; his associates reported numerous ties to industry sources.
Measles Vaccination = Lower Seizure Risk
Measles-containing vaccines are less likely to be associated with seizures when given at 12-15 months of age as recommended than when delayed until 16-23 months of age, according to results from a study published online Oct. 14 in JAMA Pediatrics.
In the retrospective cohort study examining the effect of age at immunization on 840,348 U.S. children given the MMR or MMRV vaccines during the second year of life in 2001-2011, "we found that the magnitude of increased risk of seizures following immunization with measles-containing vaccines during the second year of life depends on age.
"While measles-containing vaccines administered at 12-15 months of age are associated with an increased risk of seizures 7-10 days following immunization, their delayed administration at 16-23 months of age may result in an even greater increased risk of that adverse event following immunization," said Dr. Ali Rowhani-Rahbar of the department of epidemiology at the University of Washington, Seattle, and his associates.
The reason for this age-related difference in seizure risk is not yet known. It may simply be attributable to the fact that the background risk of seizures is higher in the older age group. Or a better-functioning immune system at the older age may allow children to mount a more rigorous immunologic response to the vaccine, raising the risk of febrile seizures.
It also is possible that vaccination is delayed among children thought to be susceptible to seizures for medical reasons, and that these children are more likely to develop seizures when they finally are vaccinated at the later age.
Regardless of the reasons for this age association, the increase in risk of seizures was very small for all age groups.
"Our findings are of direct relevance to the recommended childhood immunization schedule; they support the timely immunization of children with the first dose of measles-containing vaccine in accordance with current recommendations," the researchers noted.
Dr. Rowhani-Rahbar and his colleagues used data from the Centers for Disease Control and Prevention’s Vaccine Safety Datalink to examine this issue. The incidence of seizures was analyzed for each child during the 7- to 10-day "exposure period" following immunization and compared with the "control period" of the 42 days after that interval.
Then the rates of seizures were compared between children who were vaccinated at 12-15 months of age and those who were vaccinated at 16-23 months of age.
Overall, there were 1,810 seizure events during the 42 days following immunization, including 519 events (28.7%) that occurred during the 7- to 10-day exposure period.
The incidence of seizures during the control intervals was consistent with background rates reported in the general population. It also peaked at age 16-18 months, which is consistent with the peak that occurs in the general population.
The overall incidence of seizures was significantly higher during the exposure periods than the control intervals at all ages.
The most important finding was that the relative risk of seizures during the exposure interval was significantly greater among children vaccinated at 16-23 months of age (RR, 6.5) than among those vaccinated at 12-15 months of age (RR, 3.4) , the investigators reported (JAMA Pediatr. 2013 Oct. 14 [doi: 10.1001/jamapediatrics.2013.2745]).
The attributable risk was 9.5 excess cases of seizure per 10,000 doses given at 16-23 months of age, compared with 4.0 excess cases of seizure per 10,000 doses given at 12-15 months of age. These results remained robust in a sensitivity analysis of the data.
"These findings collectively suggest that the administration of measles-containing vaccines at the age of highest vulnerability to febrile seizures may result in an even greater increased risk of those adverse events following immunization," Dr. Rowhani-Rahbar and his associates said.
This study was supported by the CDC-sponsored Vaccine Safety Database through a subcontract with America’s Health Insurance Plans. Dr. Rowhani-Rahbar reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Merck, and other companies.
The findings of this study confirm the well-established safety of the current recommended vaccination schedule, "which is based on many years of ... safety and effectiveness data."
Many parents delay immunization, thinking that this will minimize the risk of an adverse event such as seizures. But this study clearly shows that "if vaccine safety is a concern, the currently recommended schedule is the best choice for preventing disease and minimizing adverse events," they said.
Dr. Feemster and Dr. Offit are both in the department of pediatrics at the University of Pennsylvania, Philadelphia, and in the division of infectious diseases and the Vaccine Education Center at Children’s Hospital of Philadelphia. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Rowhani-Rahbar’s report (JAMA Pediatrics 2013 [doi: 10.1001/jamapediatrics.2013.3071]).
The findings of this study confirm the well-established safety of the current recommended vaccination schedule, "which is based on many years of ... safety and effectiveness data."
Many parents delay immunization, thinking that this will minimize the risk of an adverse event such as seizures. But this study clearly shows that "if vaccine safety is a concern, the currently recommended schedule is the best choice for preventing disease and minimizing adverse events," they said.
Dr. Feemster and Dr. Offit are both in the department of pediatrics at the University of Pennsylvania, Philadelphia, and in the division of infectious diseases and the Vaccine Education Center at Children’s Hospital of Philadelphia. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Rowhani-Rahbar’s report (JAMA Pediatrics 2013 [doi: 10.1001/jamapediatrics.2013.3071]).
The findings of this study confirm the well-established safety of the current recommended vaccination schedule, "which is based on many years of ... safety and effectiveness data."
Many parents delay immunization, thinking that this will minimize the risk of an adverse event such as seizures. But this study clearly shows that "if vaccine safety is a concern, the currently recommended schedule is the best choice for preventing disease and minimizing adverse events," they said.
Dr. Feemster and Dr. Offit are both in the department of pediatrics at the University of Pennsylvania, Philadelphia, and in the division of infectious diseases and the Vaccine Education Center at Children’s Hospital of Philadelphia. They reported no financial conflicts of interest. These remarks were taken from their editorial accompanying Dr. Rowhani-Rahbar’s report (JAMA Pediatrics 2013 [doi: 10.1001/jamapediatrics.2013.3071]).
Measles-containing vaccines are less likely to be associated with seizures when given at 12-15 months of age as recommended than when delayed until 16-23 months of age, according to results from a study published online Oct. 14 in JAMA Pediatrics.
In the retrospective cohort study examining the effect of age at immunization on 840,348 U.S. children given the MMR or MMRV vaccines during the second year of life in 2001-2011, "we found that the magnitude of increased risk of seizures following immunization with measles-containing vaccines during the second year of life depends on age.
"While measles-containing vaccines administered at 12-15 months of age are associated with an increased risk of seizures 7-10 days following immunization, their delayed administration at 16-23 months of age may result in an even greater increased risk of that adverse event following immunization," said Dr. Ali Rowhani-Rahbar of the department of epidemiology at the University of Washington, Seattle, and his associates.
The reason for this age-related difference in seizure risk is not yet known. It may simply be attributable to the fact that the background risk of seizures is higher in the older age group. Or a better-functioning immune system at the older age may allow children to mount a more rigorous immunologic response to the vaccine, raising the risk of febrile seizures.
It also is possible that vaccination is delayed among children thought to be susceptible to seizures for medical reasons, and that these children are more likely to develop seizures when they finally are vaccinated at the later age.
Regardless of the reasons for this age association, the increase in risk of seizures was very small for all age groups.
"Our findings are of direct relevance to the recommended childhood immunization schedule; they support the timely immunization of children with the first dose of measles-containing vaccine in accordance with current recommendations," the researchers noted.
Dr. Rowhani-Rahbar and his colleagues used data from the Centers for Disease Control and Prevention’s Vaccine Safety Datalink to examine this issue. The incidence of seizures was analyzed for each child during the 7- to 10-day "exposure period" following immunization and compared with the "control period" of the 42 days after that interval.
Then the rates of seizures were compared between children who were vaccinated at 12-15 months of age and those who were vaccinated at 16-23 months of age.
Overall, there were 1,810 seizure events during the 42 days following immunization, including 519 events (28.7%) that occurred during the 7- to 10-day exposure period.
The incidence of seizures during the control intervals was consistent with background rates reported in the general population. It also peaked at age 16-18 months, which is consistent with the peak that occurs in the general population.
The overall incidence of seizures was significantly higher during the exposure periods than the control intervals at all ages.
The most important finding was that the relative risk of seizures during the exposure interval was significantly greater among children vaccinated at 16-23 months of age (RR, 6.5) than among those vaccinated at 12-15 months of age (RR, 3.4) , the investigators reported (JAMA Pediatr. 2013 Oct. 14 [doi: 10.1001/jamapediatrics.2013.2745]).
The attributable risk was 9.5 excess cases of seizure per 10,000 doses given at 16-23 months of age, compared with 4.0 excess cases of seizure per 10,000 doses given at 12-15 months of age. These results remained robust in a sensitivity analysis of the data.
"These findings collectively suggest that the administration of measles-containing vaccines at the age of highest vulnerability to febrile seizures may result in an even greater increased risk of those adverse events following immunization," Dr. Rowhani-Rahbar and his associates said.
This study was supported by the CDC-sponsored Vaccine Safety Database through a subcontract with America’s Health Insurance Plans. Dr. Rowhani-Rahbar reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Merck, and other companies.
Measles-containing vaccines are less likely to be associated with seizures when given at 12-15 months of age as recommended than when delayed until 16-23 months of age, according to results from a study published online Oct. 14 in JAMA Pediatrics.
In the retrospective cohort study examining the effect of age at immunization on 840,348 U.S. children given the MMR or MMRV vaccines during the second year of life in 2001-2011, "we found that the magnitude of increased risk of seizures following immunization with measles-containing vaccines during the second year of life depends on age.
"While measles-containing vaccines administered at 12-15 months of age are associated with an increased risk of seizures 7-10 days following immunization, their delayed administration at 16-23 months of age may result in an even greater increased risk of that adverse event following immunization," said Dr. Ali Rowhani-Rahbar of the department of epidemiology at the University of Washington, Seattle, and his associates.
The reason for this age-related difference in seizure risk is not yet known. It may simply be attributable to the fact that the background risk of seizures is higher in the older age group. Or a better-functioning immune system at the older age may allow children to mount a more rigorous immunologic response to the vaccine, raising the risk of febrile seizures.
It also is possible that vaccination is delayed among children thought to be susceptible to seizures for medical reasons, and that these children are more likely to develop seizures when they finally are vaccinated at the later age.
Regardless of the reasons for this age association, the increase in risk of seizures was very small for all age groups.
"Our findings are of direct relevance to the recommended childhood immunization schedule; they support the timely immunization of children with the first dose of measles-containing vaccine in accordance with current recommendations," the researchers noted.
Dr. Rowhani-Rahbar and his colleagues used data from the Centers for Disease Control and Prevention’s Vaccine Safety Datalink to examine this issue. The incidence of seizures was analyzed for each child during the 7- to 10-day "exposure period" following immunization and compared with the "control period" of the 42 days after that interval.
Then the rates of seizures were compared between children who were vaccinated at 12-15 months of age and those who were vaccinated at 16-23 months of age.
Overall, there were 1,810 seizure events during the 42 days following immunization, including 519 events (28.7%) that occurred during the 7- to 10-day exposure period.
The incidence of seizures during the control intervals was consistent with background rates reported in the general population. It also peaked at age 16-18 months, which is consistent with the peak that occurs in the general population.
The overall incidence of seizures was significantly higher during the exposure periods than the control intervals at all ages.
The most important finding was that the relative risk of seizures during the exposure interval was significantly greater among children vaccinated at 16-23 months of age (RR, 6.5) than among those vaccinated at 12-15 months of age (RR, 3.4) , the investigators reported (JAMA Pediatr. 2013 Oct. 14 [doi: 10.1001/jamapediatrics.2013.2745]).
The attributable risk was 9.5 excess cases of seizure per 10,000 doses given at 16-23 months of age, compared with 4.0 excess cases of seizure per 10,000 doses given at 12-15 months of age. These results remained robust in a sensitivity analysis of the data.
"These findings collectively suggest that the administration of measles-containing vaccines at the age of highest vulnerability to febrile seizures may result in an even greater increased risk of those adverse events following immunization," Dr. Rowhani-Rahbar and his associates said.
This study was supported by the CDC-sponsored Vaccine Safety Database through a subcontract with America’s Health Insurance Plans. Dr. Rowhani-Rahbar reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Merck, and other companies.
FROM JAMA PEDIATRICS