User login
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).
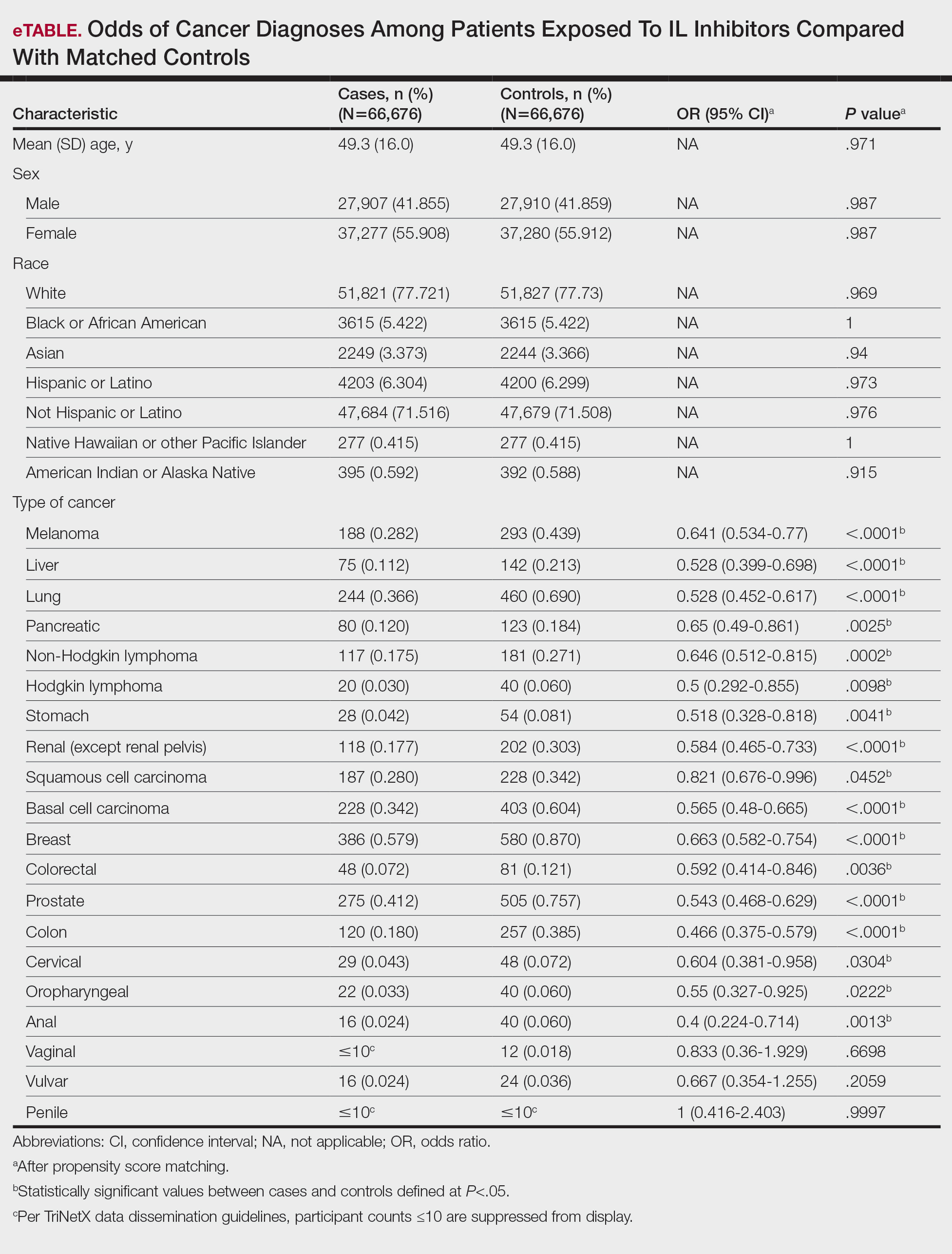
Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).

Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
To the Editor:
Psoriasis is a chronic immune-mediated inflammatory skin disease that affects approximately 2% to 3% of the global population and an estimated 7.5 million adults in the United States.1 The condition is characterized by recurrent episodes of erythematous scaly plaques driven by dysregulated immune responses, particularly involving the interleukin (IL) 23/T-helper (Th) 17 axis.2 Although cutaneous symptoms are the most visible manifestation, psoriasis is a systemic disorder with broad multisystem involvement. Comorbidities include psoriatic arthritis, metabolic syndrome, cardiovascular disease, inflammatory bowel disease, depression, and anxiety.1 These conditions contribute to a heightened risk for premature mortality, increased health care utilization, and an estimated direct cost burden exceeding $11 billion annually in the United States alone.3 Patients with moderate to severe disease frequently require systemic therapy, and long-term disease control is essential to prevent cumulative inflammatory damage and reduce associated morbidity.4
Globally, psoriasis prevalence and disease severity vary by geography, ethnicity, and environmental factors, with higher rates in Northern Europe and North America and lower reported prevalence in East Asia and sub-Saharan Africa.5 In lower-resource settings, access to advanced therapies is limited, and patients often are treated with less effective or more toxic systemic agents, such as methotrexate or cyclosporine.5 These disparities not only affect quality of life but also may influence comorbidity and malignancy patterns, underscoring the importance of studying biologic safety in diverse real-world populations.
Over the past decade, the therapeutic landscape for psoriasis has been transformed by biologic agents targeting specific immune pathways.6 Interleukin 17 inhibitors (eg, secukinumab, ixekizumab, brodalumab, bimekizumab) act by neutralizing IL-17A, IL-17F, or the IL-17 receptor, thereby reducing keratinocyte activation, neutrophil recruitment, and downstream cytokine production.6 Interleukin 23 inhibitors (eg, guselkumab, risankizumab, tildrakizumab) block the p19 subunit of IL-23, halting the expansion and maintenance of pathogenic Th17 cells.6 Ustekinumab, an IL-12/23 inhibitor, targets the shared p40 subunit of IL-12 and IL-23, attenuating both Th1 and Th17 signaling.6 These agents achieve rapid, durable skin clearance in a large proportion of patients, improve psoriatic arthritis symptoms, and generally are well tolerated, even with long-term use.6
Although efficacy is well established, the immunomodulatory nature of IL inhibitors raises theoretical concerns about malignancy risk. Immune surveillance plays a critical role in detecting and eliminating emerging tumor cells.7 Data from other systemic immunosuppressants, such as cyclosporine, show increased risks for certain cancers8; however, the IL-17 and IL-23 pathways have dual roles in cancer biology.7 In some tumor contexts, these cytokines promote carcinogenesis through angiogenesis, epithelial proliferation, and suppression of antitumor immunity; therefore, inhibiting these pathways could theoretically reduce cancer risk.7 The uncertainty around this risk-benefit balance has made malignancy a central consideration for dermatologists, particularly when initiating therapy in patients with a history of cancer or other risk factors.
The perception of malignancy risk can influence patient willingness to start biologics as well as physician prescribing patterns.9 Some clinicians opt for alternative therapies in individuals with a personal or family history of cancer despite limited direct evidence of harm from IL inhibitors. Conversely, a reassuring malignancy safety profile may support broader adoption of these therapies, especially in patients requiring lifelong disease control.9 Shared decision-making in this context requires robust, real-world evidence that accounts for both common and rare malignancy outcomes.
Randomized controlled trials of IL inhibitors have not demonstrated a consistent malignancy signal, but these studies often are underpowered for rare outcomes and limited by short follow-up durations, typically less than 1 year. They also frequently exclude high-risk populations, limiting generalizability.10 Observational studies using real-world data can address these gaps by including more diverse patient populations, longer observation windows, and larger sample sizes capable of detecting differences in uncommon outcomes.
The TriNetX Analytics Network (http://www.trinetx.com) offers a unique platform for large-scale, real-world pharmacoepidemiologic research. This federated database aggregates deidentified electronic health record data from more than 100 million patients across the United States and internationally, including at academic medical centers, integrated delivery networks, and community hospitals.4 Data contributors refresh their datasets regularly, ensuring near-contemporary representation of prescribing trends and clinical outcomes. Standardized terminology mapping, consistent International Classification of Diseases, Tenth Revision, Clinical Modification (ICD-10-CM) coding, and centralized data-quality checks enhance the reliability of analyses. Governance protocols and compliance with the Health Insurance Portability and Accountability Act deidentification standards further ensure ethical use of the data. The breadth and depth of the TriNetX network make it possible to evaluate not only common malignancies but also rare cancer types that smaller studies cannot assess with sufficient statistical power.
We performed a retrospective matched-cohort study, querying data from January 1, 2014, through December 31, 2024, using TriNetX to examine whether IL inhibitor exposure is associated with differences in incident malignancy risk among adults with psoriasis. Patients aged 18 years or older with a psoriasis diagnosis (ICD-10-CM code L40.x) and documented exposure to an IL-17, IL-23, or IL-12/23 inhibitor were eligible. Patients with a prior malignancy diagnosis were excluded to reduce prevalence bias. To ensure that malignancies were incident, we included only those diagnosed at least 1 day after initiation of an IL inhibitor.
The comparison cohort consisted of psoriasis patients without IL inhibitor exposure during their observation period. We used 1:1 propensity score matching based on age, sex, race, and ethnicity, applying a caliper of 0.1 to balance baseline characteristics and minimize demographic confounding. The index date for unexposed patients was randomly assigned within their observation period to align follow-up timing with exposed patients. Outcomes were identified by ICD-10-CM codes grouped by skin, hematologic, and solid-organ malignancies. Odds ratios (ORs) with 95% confidence intervals (CIs) were calculated, with statistical significance set at P<.05. Odds ratios were selected over hazard ratios due to variability in precise follow-up time capture and the primary analytic goal of comparing proportional odds within matched follow-up windows.
Propensity score matching was employed because it is a well-established technique in pharmacoepidemiology to mimic some of the balance achieved in randomized trials. By equating treatment and control groups on measured confounders, matching helps isolate the treatment effect, particularly important in nonrandomized datasets in which prescribing decisions may be influenced by baseline characteristics. Grouping cancers into clinically relevant categories allowed us to assess patterns of association, as some cancer types (eg, melanoma, lymphomas) may have pathophysiologic links to inflammatory pathways targeted by IL inhibitors.
The final cohort included 133,352 patients, with 66,676 in each group. The mean (SD) age was 49.3 (16.0) years, and demographic variables were well balanced after matching. The mean follow-up was approximately 3.8 years. Interleukin 17 inhibitors were the most frequently prescribed, followed by IL-23 inhibitors and ustekinumab. Baseline comorbidities such as cardiovascular disease, diabetes, and obesity were comparable between groups, reducing the likelihood of confounding from these factors.
Interleukin inhibitor exposure was associated with significantly reduced odds of several malignancies (eTable). Among skin cancers, melanoma risk was reduced by 36% (OR, 0.641; 95% CI, 0.534-0.77; P<.0001), basal cell carcinoma by 43% (OR, 0.565; 95% CI, 0.48-0.665; P<.0001), and squamous cell carcinoma by 18% (OR, 0.821; 95% CI, 0.676-0.996; P=.0452). Hematologic malignancies showed similar reductions, with non-Hodgkin lymphoma odds reduced by 35% (OR, 0.646; 95% CI, 0.512-0.815; P=.0002) and Hodgkin lymphoma by 50% (OR, 0.5; 95% CI, 0.292-0.855; P=.0098).

Protective associations also were observed for several solid tumors: lung (OR, 0.528; 95% CI, 0.452-0.617; P<.0001), liver (OR, 0.528; 95% CI, 0.399-0.698; P<.0001), pancreatic (OR, 0.65; 95% CI, 0.49-0.861; P=.0025), breast (OR, 0.663; 95% CI, 0.582-0.754; P<.0001), prostate (OR, 0.543; 95% CI, 0.468-0.629; P<.0001), colorectal (OR, 0.592; 95% CI, 0.414-0.846; P=.0036), colon (OR, 0.466; 95% CI, 0.375-0.579; P<.0001), and oropharyngeal (OR, 0.55; 95% CI, 0.327-0.925; P=.0222) cancers. Cervical cancer (OR, 0.604; 95% CI, 0.381-0.958; P=.0304) and anal cancer (OR, 0.4; 95% CI, 0.224-0.714; P=.0013) also showed significant reductions. Vaginal, vulvar, and penile cancers demonstrated no significant differences, likely due to their low incidence and limited statistical power.
The biological plausibility of these findings is supported by preclinical studies implicating IL-17 and IL-23 in tumor-promoting inflammation.11 These cytokines can recruit myeloid-derived suppressor cells, promote angiogenesis, and facilitate tumor-immune evasion. Inhibition may shift the immune microenvironment toward enhanced tumor surveillance, reduce protumorigenic cytokine signaling, and normalize regulatory T-cell function.11 These mechanisms could explain observed reductions in melanoma, lymphomas, and certain solid tumors.
Our results are consistent with several large registry studies showing no increased cancer incidence in IL inhibitor users and extend prior findings by demonstrating significant reductions in multiple cancer types.12 The melanoma reduction contrasts with the findings in earlier biologic safety studies, possibly due to our larger sample size, broader geographic representation, and inclusion of multiple IL inhibitor classes.13 Similar reductions have not been consistently observed with tumor necrosis factor α inhibitors, which have different immunologic targets and a more complex malignancy safety history.14
Limitations of our study include the retrospective design, potential misclassification of cancer diagnoses, and lack of data on unmeasured confounders such as sun exposure, smoking, alcohol use, and family cancer history. Surveillance bias is possible, though it would likely bias toward higher, not lower, cancer detection in biologic users. Our mean follow-up period of 3.8 years may not be sufficient for cancers with long latency periods.
If replicated, our findings could have meaningful public health implications. Reassurance regarding malignancy safety may increase patient acceptance and physician confidence in prescribing IL inhibitors, particularly for patients requiring long-term therapy. From a payer perspective, the potential for reduced cancer incidence could translate into substantial cost savings over time, offsetting the high up-front cost of biologics. Additionally, these results may be relevant to other IL inhibitor indications, including psoriatic arthritis, ankylosing spondylitis, and inflammatory bowel disease, in which similar pathophysiologic mechanisms may be at play.
In conclusion, this large matched-cohort study found that IL inhibitor therapy in psoriasis was associated with significantly reduced odds of multiple malignancies, including melanoma, lymphomas, and several solid tumors. These findings contribute to the growing body of real-world evidence supporting the long-term safety of IL inhibitors and underscore the need for continued pharmacovigilance and mechanistic research.
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
- Armstrong AW, Mehta MD, Schupp CW, et al. Psoriasis prevalence in adults in the United States. JAMA Dermatol. 2021;157:940-946. doi:10.1001/jamadermatol.2021.2007
- Deng Z, Wang S, Wu C, et al. IL-17 inhibitor-associated inflammatory bowel disease: a study based on literature and database analysis. Front Pharmacol. 2023;14:1124628. doi:10.3389/fphar.2023.1124628
- Al Sawah S, Foster SA, Goldblum OM, et al. Healthcare costs in psoriasis and psoriasis sub-groups over time following psoriasis diagnosis. J Med Econ. 2017;20:982-990. doi:10.1080/13696998.2017.1345749
- Korman NJ. Management of psoriasis as a systemic disease: what is the evidence? Br J Dermatol. 2020;182:840-848. doi:10.1111/bjd.18245
- Damiani G, Bragazzi NL, Karimkhani Aksut C, et al. The global, regional, and national burden of psoriasis: results and insights from the Global Burden of Disease 2019 Study. Front Med (Lausanne). 2021;8:743180. doi:10.3389/fmed.2021.743180
- Metko D, Torres T, Vender R. Viewpoint about biologic agents for psoriasis: are they immunosuppressants or immunomodulators? J Int Med Res. 2023;51:3000605231175547. doi:10.1177/03000605231175547
- Tsai YC, Tsai TF. Anti-interleukin and interleukin therapies for psoriasis: current evidence and clinical usefulness. Ther Adv Musculoskelet Dis. 2017;9:277-294. doi:10.1177/1759720X17735756
- Durnian JM, Stewart RM, Tatham R, et al. Cyclosporin-A associated malignancy. Clin Ophthalmol. 2007;1:421-430.
- DeWitt EM, Lin L, Glick HA, et al. Pattern and predictors of the initiation of biologic agents for the treatment of rheumatoid arthritis in the United States: an analysis using a large observational data bank. Clin Ther. 2009;31:1871-1858. doi:10.1016/j.clinthera.2009.08.020
- Vangilbergen M, Stockman A, Van De Velde A, et al. The role of interleukin-17 and interleukin-23 inhibitors in the development, progression, and recurrence of cancer: a systematic review. JAAD Int. 2024;17:71-79. doi:10.1016/j.jdin.2024.06.006
- Navarro-Compán V, Puig L, Vidal S, et al. The paradigm of IL-23-independent production of IL-17F and IL-17A and their role in chronic inflammatory diseases. Front Immunol. 2023;14:1191782. doi:10.3389/fimmu.2023.1191782
- Bencardino S, Bernardi F, Allocca M, et al. Advanced therapies for inflammatory bowel disease and risk of skin cancer: what’s new? Cancers (Basel). 2025;17:1710. doi:10.3390/cancers17101710
- Esse S, Mason KJ, Green AC, et al. Melanoma risk in patients treated with biologic therapy for common inflammatory diseases: a systematic review and meta-analysis. JAMA Dermatol. 2020;156:787-794. doi:10.1001/jamadermatol.2020.1300
- Solomon DH, Mercer E, Kavanaugh A. Observational studies on the risk of cancer associated with tumor necrosis factor inhibitors in rheumatoid arthritis: a review of their methodologies and results. Arthritis Rheum. 2012;64:21-32. doi:10.1002/art.30653
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Malignancy Risk Among Psoriasis Patients Treated With Interleukin Inhibitors: A Retrospective Matched-Cohort Study
Practice Points
- Interleukin (IL) inhibitor therapy for psoriasis was associated with reduced odds of multiple malignancies in a large matched-cohort analysis.
- Potential mechanisms for reduced cancer risk include inhibition of tumor-promoting inflammation and restoration of antitumor immune surveillance, although further mechanistic and longitudinal studies are needed.
- These findings provide real-world evidence supporting the long-term malignancy safety of IL inhibitors, which may reassure clinicians and patients considering these agents for chronic disease management.
Testosterone Pellet–Induced Generalized Drug Eruption
To the Editor:
Testosterone-replacement therapy (TRT) is indicated for hypogonadism. The benefits of TRT are well documented, with multiple options available for delivery. Testosterone pellet implantation (TPI) is an effective treatment option for hypogonadism with minimal adverse reactions. Availability of TRT is increasing, as facilities are offering off-label applications. Although TPI generally is well tolerated, cutaneous reactions have been documented. We present a patient with drug-induced dermatitis following TPI.
A 51-year-old man with hypogonadism presented with an extremely pruritic rash that began on the left buttock 3 days after receiving his fourth TPI. The patient had received subcutaneous insertions of 8 testosterone pellets (75 mg per pellet every 6 months) to the left buttock. He denied any history of a similar rash. His medical history was remarkable for hyperlipidemia, which was controlled with niacin and omega-3 fatty acids (fish oil). Other medications included glucosamine. Before presenting to our clinic, he was given a 40-mg intramuscular injection of triamcinolone acetonide and trimethoprim-sulfamethoxazole twice daily for 7 days, a methylprednisolone dose pack, and triamcinolone ointment 0.1% twice daily by his primary care physician, all without improvement of the rash.
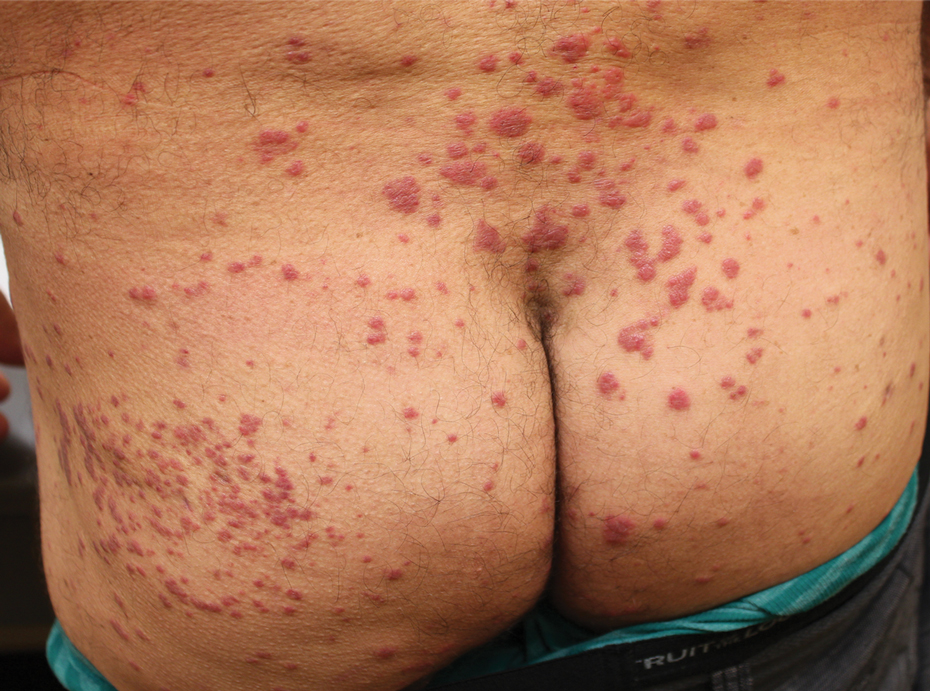
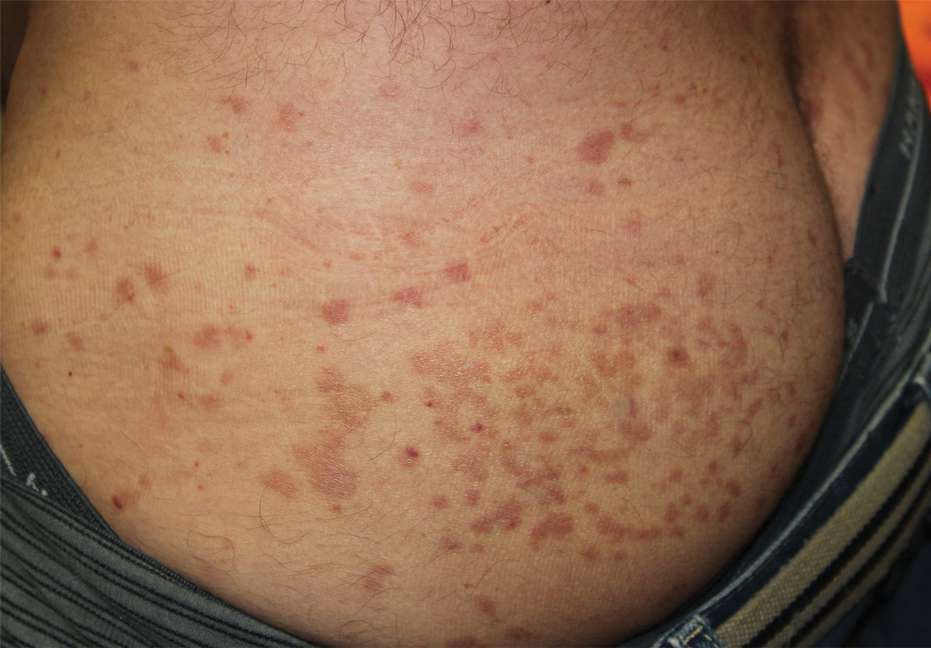
Physical examination revealed multiple well-circumscribed, coalescing clusters of darkly erythematous papules and dermal plaques of varying size on the buttocks with extension to the lower back, abdomen, and thighs (Figure 1). The differential diagnosis included lichenoid eruption, pseudolymphoma, sarcoidosis, and granuloma annulare.
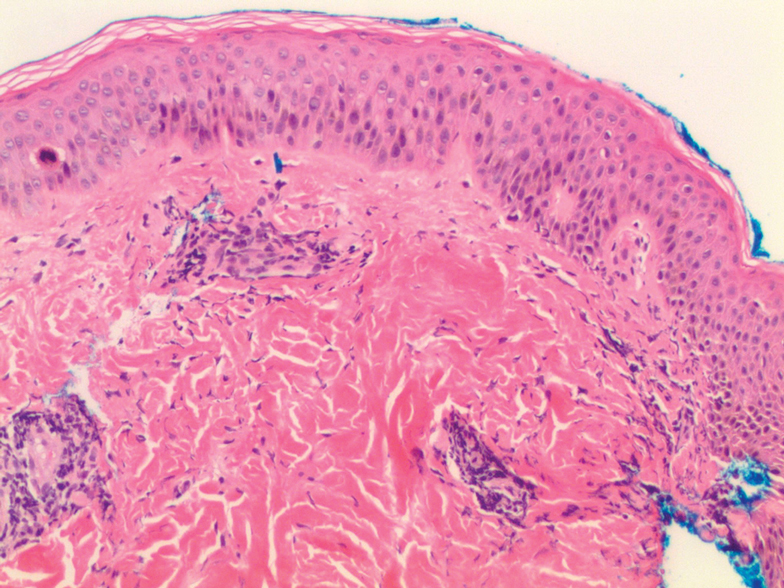
Histologic examination of a punch biopsy revealed an epidermis with a normal stratum corneum and subtle cell-poor vacuolar interface dermatitis with rare necrotic keratinocytes. There was a mild perivascular lymphocytic infiltrate with slight edema within the dermis without notable eosinophils or findings indicative of a vasculitic process (Figure 2).
Oral prednisone 60 mg daily and betamethasone ointment 0.05% applied twice daily were started, with notable improvement of the rash in 1 week (Figure 3). Given the temporal relationship of the TPI, histologic findings suggestive of drug eruption, and resolution of symptoms shortly after treatment, a diagnosis of testosterone pellet–induced generalized dermatitis was established.
Testosterone-replacement therapy is the principal treatment of male pathologic hypoandrogenism, but off-label prescription frequently occurs for age-related hypogonadism and hypoactive sexual desire disorder.1 Testosterone-replacement therapy also can enhance sexual desire and function and improve mood in premenopausal and postmenopausal women with testosterone deficiency.2 Delivery options include topicals, intramuscular injections, oral formulations, transdermal patches and gels, and subcutaneous placement of testosterone pellets (TPI).Cutaneous reactions to TPI are rare. Hirsutism, male-pattern hair loss, and acne are possible cutaneous adverse reactions.3 In addition, a localized erythematous pruritic eruption at the implantation site and an immunologic foreign-body reaction to testosterone pellets have been reported.4
In one case report, a man developed recurrent ill-defined, erythematous, scaly plaques and patches over the buttocks and thighs, consistent with testosterone-induced eczematous dermatitis, subsequent to his second TPI. The patient presented with the eruption within 4 weeks after the most recent implantation, similar to our case, but differed temporally in initial presentation, presenting after the second implantation.5 Our case differed in morphologic presentation (dermal plaques as opposed to eczematous change) and refractoriness to triamcinolone injection.
Testosterone-replacement therapy is becoming more widely available. Lack of regulation of proper marketing by such facilities as medical spas that offer TPI for off-label applications has led to a rampant increase in TRT prescribing, possibly foreshadowing an increase in adverse cutaneous reactions to TRT.6
Our case of histologically consistent testosterone pellet–induced dermatitis highlights a rare cutaneous adverse reaction that can occur subsequent to TPI and illustrates the efficacy of high-dose oral steroids as a treatment option. With increased use of TRT, physicians should be cognizant of the potential adverse cutaneous effects related to this treatment and counsel patients appropriately prior to initiating treatment.
Acknowledgment
We thank the patient for granting permission to publish this case.
- Clayton AH, Kingsberg SA, Goldstein I. Evaluation and management of hypoactive sexual desire disorder. Sex Med. 2018;6:59-74.
- Glaser R, Dimitrakakis C. Testosterone therapy in women: myths and misconceptions. Maturitas. 2013;74:230-234.
- Testopel (testosterone pellet) [package insert]. Endo Pharmaceuticals, Inc; 2016. Accessed December 16, 2020. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=a1741a0b-3d4c-42dc-880d-a06e96cce9ef&type=display
- Cavender RK, Fairall M. Subcutaneous testosterone pellet implant (Testopel) therapy for men with testosterone deficiency syndrome: a single-site retrospective safety analysis. J Sex Med. 2009;6:3177-3192.
- Heldt Manica LA, Cohen PR. Testosterone pellet associated dermatitis: report and review of Testopel-related cutaneous adverse effects. Cureus. 2017;9:e1560.
- Mintzes B. The marketing of testosterone treatments for age-related low testosterone or ‘Low T’. Curr Opin Endocrinol Diabetes Obes. 2018;25:224-230.
To the Editor:
Testosterone-replacement therapy (TRT) is indicated for hypogonadism. The benefits of TRT are well documented, with multiple options available for delivery. Testosterone pellet implantation (TPI) is an effective treatment option for hypogonadism with minimal adverse reactions. Availability of TRT is increasing, as facilities are offering off-label applications. Although TPI generally is well tolerated, cutaneous reactions have been documented. We present a patient with drug-induced dermatitis following TPI.
A 51-year-old man with hypogonadism presented with an extremely pruritic rash that began on the left buttock 3 days after receiving his fourth TPI. The patient had received subcutaneous insertions of 8 testosterone pellets (75 mg per pellet every 6 months) to the left buttock. He denied any history of a similar rash. His medical history was remarkable for hyperlipidemia, which was controlled with niacin and omega-3 fatty acids (fish oil). Other medications included glucosamine. Before presenting to our clinic, he was given a 40-mg intramuscular injection of triamcinolone acetonide and trimethoprim-sulfamethoxazole twice daily for 7 days, a methylprednisolone dose pack, and triamcinolone ointment 0.1% twice daily by his primary care physician, all without improvement of the rash.
Physical examination revealed multiple well-circumscribed, coalescing clusters of darkly erythematous papules and dermal plaques of varying size on the buttocks with extension to the lower back, abdomen, and thighs (Figure 1). The differential diagnosis included lichenoid eruption, pseudolymphoma, sarcoidosis, and granuloma annulare.

Histologic examination of a punch biopsy revealed an epidermis with a normal stratum corneum and subtle cell-poor vacuolar interface dermatitis with rare necrotic keratinocytes. There was a mild perivascular lymphocytic infiltrate with slight edema within the dermis without notable eosinophils or findings indicative of a vasculitic process (Figure 2).

Oral prednisone 60 mg daily and betamethasone ointment 0.05% applied twice daily were started, with notable improvement of the rash in 1 week (Figure 3). Given the temporal relationship of the TPI, histologic findings suggestive of drug eruption, and resolution of symptoms shortly after treatment, a diagnosis of testosterone pellet–induced generalized dermatitis was established.

Testosterone-replacement therapy is the principal treatment of male pathologic hypoandrogenism, but off-label prescription frequently occurs for age-related hypogonadism and hypoactive sexual desire disorder.1 Testosterone-replacement therapy also can enhance sexual desire and function and improve mood in premenopausal and postmenopausal women with testosterone deficiency.2 Delivery options include topicals, intramuscular injections, oral formulations, transdermal patches and gels, and subcutaneous placement of testosterone pellets (TPI).Cutaneous reactions to TPI are rare. Hirsutism, male-pattern hair loss, and acne are possible cutaneous adverse reactions.3 In addition, a localized erythematous pruritic eruption at the implantation site and an immunologic foreign-body reaction to testosterone pellets have been reported.4
In one case report, a man developed recurrent ill-defined, erythematous, scaly plaques and patches over the buttocks and thighs, consistent with testosterone-induced eczematous dermatitis, subsequent to his second TPI. The patient presented with the eruption within 4 weeks after the most recent implantation, similar to our case, but differed temporally in initial presentation, presenting after the second implantation.5 Our case differed in morphologic presentation (dermal plaques as opposed to eczematous change) and refractoriness to triamcinolone injection.
Testosterone-replacement therapy is becoming more widely available. Lack of regulation of proper marketing by such facilities as medical spas that offer TPI for off-label applications has led to a rampant increase in TRT prescribing, possibly foreshadowing an increase in adverse cutaneous reactions to TRT.6
Our case of histologically consistent testosterone pellet–induced dermatitis highlights a rare cutaneous adverse reaction that can occur subsequent to TPI and illustrates the efficacy of high-dose oral steroids as a treatment option. With increased use of TRT, physicians should be cognizant of the potential adverse cutaneous effects related to this treatment and counsel patients appropriately prior to initiating treatment.
Acknowledgment
We thank the patient for granting permission to publish this case.
To the Editor:
Testosterone-replacement therapy (TRT) is indicated for hypogonadism. The benefits of TRT are well documented, with multiple options available for delivery. Testosterone pellet implantation (TPI) is an effective treatment option for hypogonadism with minimal adverse reactions. Availability of TRT is increasing, as facilities are offering off-label applications. Although TPI generally is well tolerated, cutaneous reactions have been documented. We present a patient with drug-induced dermatitis following TPI.
A 51-year-old man with hypogonadism presented with an extremely pruritic rash that began on the left buttock 3 days after receiving his fourth TPI. The patient had received subcutaneous insertions of 8 testosterone pellets (75 mg per pellet every 6 months) to the left buttock. He denied any history of a similar rash. His medical history was remarkable for hyperlipidemia, which was controlled with niacin and omega-3 fatty acids (fish oil). Other medications included glucosamine. Before presenting to our clinic, he was given a 40-mg intramuscular injection of triamcinolone acetonide and trimethoprim-sulfamethoxazole twice daily for 7 days, a methylprednisolone dose pack, and triamcinolone ointment 0.1% twice daily by his primary care physician, all without improvement of the rash.
Physical examination revealed multiple well-circumscribed, coalescing clusters of darkly erythematous papules and dermal plaques of varying size on the buttocks with extension to the lower back, abdomen, and thighs (Figure 1). The differential diagnosis included lichenoid eruption, pseudolymphoma, sarcoidosis, and granuloma annulare.

Histologic examination of a punch biopsy revealed an epidermis with a normal stratum corneum and subtle cell-poor vacuolar interface dermatitis with rare necrotic keratinocytes. There was a mild perivascular lymphocytic infiltrate with slight edema within the dermis without notable eosinophils or findings indicative of a vasculitic process (Figure 2).

Oral prednisone 60 mg daily and betamethasone ointment 0.05% applied twice daily were started, with notable improvement of the rash in 1 week (Figure 3). Given the temporal relationship of the TPI, histologic findings suggestive of drug eruption, and resolution of symptoms shortly after treatment, a diagnosis of testosterone pellet–induced generalized dermatitis was established.

Testosterone-replacement therapy is the principal treatment of male pathologic hypoandrogenism, but off-label prescription frequently occurs for age-related hypogonadism and hypoactive sexual desire disorder.1 Testosterone-replacement therapy also can enhance sexual desire and function and improve mood in premenopausal and postmenopausal women with testosterone deficiency.2 Delivery options include topicals, intramuscular injections, oral formulations, transdermal patches and gels, and subcutaneous placement of testosterone pellets (TPI).Cutaneous reactions to TPI are rare. Hirsutism, male-pattern hair loss, and acne are possible cutaneous adverse reactions.3 In addition, a localized erythematous pruritic eruption at the implantation site and an immunologic foreign-body reaction to testosterone pellets have been reported.4
In one case report, a man developed recurrent ill-defined, erythematous, scaly plaques and patches over the buttocks and thighs, consistent with testosterone-induced eczematous dermatitis, subsequent to his second TPI. The patient presented with the eruption within 4 weeks after the most recent implantation, similar to our case, but differed temporally in initial presentation, presenting after the second implantation.5 Our case differed in morphologic presentation (dermal plaques as opposed to eczematous change) and refractoriness to triamcinolone injection.
Testosterone-replacement therapy is becoming more widely available. Lack of regulation of proper marketing by such facilities as medical spas that offer TPI for off-label applications has led to a rampant increase in TRT prescribing, possibly foreshadowing an increase in adverse cutaneous reactions to TRT.6
Our case of histologically consistent testosterone pellet–induced dermatitis highlights a rare cutaneous adverse reaction that can occur subsequent to TPI and illustrates the efficacy of high-dose oral steroids as a treatment option. With increased use of TRT, physicians should be cognizant of the potential adverse cutaneous effects related to this treatment and counsel patients appropriately prior to initiating treatment.
Acknowledgment
We thank the patient for granting permission to publish this case.
- Clayton AH, Kingsberg SA, Goldstein I. Evaluation and management of hypoactive sexual desire disorder. Sex Med. 2018;6:59-74.
- Glaser R, Dimitrakakis C. Testosterone therapy in women: myths and misconceptions. Maturitas. 2013;74:230-234.
- Testopel (testosterone pellet) [package insert]. Endo Pharmaceuticals, Inc; 2016. Accessed December 16, 2020. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=a1741a0b-3d4c-42dc-880d-a06e96cce9ef&type=display
- Cavender RK, Fairall M. Subcutaneous testosterone pellet implant (Testopel) therapy for men with testosterone deficiency syndrome: a single-site retrospective safety analysis. J Sex Med. 2009;6:3177-3192.
- Heldt Manica LA, Cohen PR. Testosterone pellet associated dermatitis: report and review of Testopel-related cutaneous adverse effects. Cureus. 2017;9:e1560.
- Mintzes B. The marketing of testosterone treatments for age-related low testosterone or ‘Low T’. Curr Opin Endocrinol Diabetes Obes. 2018;25:224-230.
- Clayton AH, Kingsberg SA, Goldstein I. Evaluation and management of hypoactive sexual desire disorder. Sex Med. 2018;6:59-74.
- Glaser R, Dimitrakakis C. Testosterone therapy in women: myths and misconceptions. Maturitas. 2013;74:230-234.
- Testopel (testosterone pellet) [package insert]. Endo Pharmaceuticals, Inc; 2016. Accessed December 16, 2020. https://dailymed.nlm.nih.gov/dailymed/fda/fdaDrugXsl.cfm?setid=a1741a0b-3d4c-42dc-880d-a06e96cce9ef&type=display
- Cavender RK, Fairall M. Subcutaneous testosterone pellet implant (Testopel) therapy for men with testosterone deficiency syndrome: a single-site retrospective safety analysis. J Sex Med. 2009;6:3177-3192.
- Heldt Manica LA, Cohen PR. Testosterone pellet associated dermatitis: report and review of Testopel-related cutaneous adverse effects. Cureus. 2017;9:e1560.
- Mintzes B. The marketing of testosterone treatments for age-related low testosterone or ‘Low T’. Curr Opin Endocrinol Diabetes Obes. 2018;25:224-230.
Practice Points
- Dermatologists should be aware that testosterone pellet implantation can cause dermatitis overlying the implantation site, which can generalize and differ in morphologic presentation.
- For patients presenting with a suspected case of testosterone pellet–induced dermatitis, a high-dose oral corticosteroid can be deployed as an effective therapy.