User login
Peter Chin, MD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: The Maker Movement and Hospital Medicine
The Maker Movement, the 21st century's upgrade of do-it-yourself that includes 3D printers and "the Internet of Things," is showing up in hospitals in interesting ways. Clinical teams confronted with a nagging issue on the wards can work together to design and prototype physical-product solutions. Two Beth Israel Deaconess hospitalists talk about the Maker Movement, and how they've turned it into a team-based, near real-time collaborative process for addressing quality improvement challenges.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The Maker Movement, the 21st century's upgrade of do-it-yourself that includes 3D printers and "the Internet of Things," is showing up in hospitals in interesting ways. Clinical teams confronted with a nagging issue on the wards can work together to design and prototype physical-product solutions. Two Beth Israel Deaconess hospitalists talk about the Maker Movement, and how they've turned it into a team-based, near real-time collaborative process for addressing quality improvement challenges.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The Maker Movement, the 21st century's upgrade of do-it-yourself that includes 3D printers and "the Internet of Things," is showing up in hospitals in interesting ways. Clinical teams confronted with a nagging issue on the wards can work together to design and prototype physical-product solutions. Two Beth Israel Deaconess hospitalists talk about the Maker Movement, and how they've turned it into a team-based, near real-time collaborative process for addressing quality improvement challenges.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: A case study in managing opioid addiction after a back injury
Has your patient’s opioid use escalated to opioid addiction? How can you tell, and what interventions can you take to help this patient in the context of a 15-minute appointment?
In this edition of Mental Health Consult, our panel discusses their recommendations for workup and next steps for managing a 45-year-old man who has no history of psychiatric disorders and appears to now be dependent on the opioid painkillers that he initially received after a back injury.
Join our panel of experts from George Washington University, Washington, including Daniel Lieberman, MD, professor of psychiatry and behavioral sciences clinical director; April Barbour, MD, MPH, director of the division of general internal medicine; and Lorenzo Norris, MD, medical director of psychiatric and behavioral services, as they discuss how to treat pain medication–related addiction, including when to refer to pain specialists and how various practice models drive treatment decisions and reimbursement.
On Twitter @whitneymcknight
Has your patient’s opioid use escalated to opioid addiction? How can you tell, and what interventions can you take to help this patient in the context of a 15-minute appointment?
In this edition of Mental Health Consult, our panel discusses their recommendations for workup and next steps for managing a 45-year-old man who has no history of psychiatric disorders and appears to now be dependent on the opioid painkillers that he initially received after a back injury.
Join our panel of experts from George Washington University, Washington, including Daniel Lieberman, MD, professor of psychiatry and behavioral sciences clinical director; April Barbour, MD, MPH, director of the division of general internal medicine; and Lorenzo Norris, MD, medical director of psychiatric and behavioral services, as they discuss how to treat pain medication–related addiction, including when to refer to pain specialists and how various practice models drive treatment decisions and reimbursement.
On Twitter @whitneymcknight
Has your patient’s opioid use escalated to opioid addiction? How can you tell, and what interventions can you take to help this patient in the context of a 15-minute appointment?
In this edition of Mental Health Consult, our panel discusses their recommendations for workup and next steps for managing a 45-year-old man who has no history of psychiatric disorders and appears to now be dependent on the opioid painkillers that he initially received after a back injury.
Join our panel of experts from George Washington University, Washington, including Daniel Lieberman, MD, professor of psychiatry and behavioral sciences clinical director; April Barbour, MD, MPH, director of the division of general internal medicine; and Lorenzo Norris, MD, medical director of psychiatric and behavioral services, as they discuss how to treat pain medication–related addiction, including when to refer to pain specialists and how various practice models drive treatment decisions and reimbursement.
On Twitter @whitneymcknight
VIDEO: Expert roundtable explores 50 years of contraception
WASHINGTON – What has the increased access to contraception over the last 50 years meant for American women?
We asked Ob.Gyn. News editorial advisory board member Dr. Eve Espey, professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque, along with three experts in family planning, to explore how expanded contraception options have affected public health, what barriers still remain, and what new products are in the pipeline.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Perhaps the biggest impact for women has been the ability to participate in the workforce, and that includes women entering medical school.
“Over the last 50 years, we’ve seen a big increase in the number of women who are professionals, who are physicians,” Dr. Espey said during the roundtable. “And during that same period of time, we’ve seen the growth of more focus on family planning.”
“I think that really would not be possible without the ability to control our fertility,” said Dr. Sarah W. Prager, associate professor of obstetrics and gynecology and director of the Ryan Family Planning Program at the University of Washington, Seattle.
And the widespread availability of contraception has translated into a decrease in maternal mortality as a result of fewer unintended pregnancies. “The implications for public health – for women and children in this country – is huge,” Dr. Prager said.
LARCs
One of the big shifts in contraceptive trends has been the slow but increasing uptake of long-acting reversible contraceptives (LARCs), such as IUDs and implants. After a drop-off in the 1970s following safety problems with the Dalkon Shield, there has been a resurgence in interest.
In the last decade, the rate of LARC use has grown from about 2% to 10%, corresponding to a slight drop in rates of unintended pregnancy, said Dr. Nikki B. Zite, professor and residency program director in the department of obstetrics and gynecology at the University of Tennessee, Knoxville.
The Contraceptive CHOICE Project, which enrolled more than 9,000 women who were provided with the no-cost reversible contraceptive method of their choice, found that about three-quarters of the women chose an IUD or an implant, which was associated with significant reductions in unintended and teen pregnancies.
“What we saw was that when we removed barriers to contraception in general, that uptake of IUDs and implants really went up,” said Dr. Tessa Madden, director of the division of family planning and associate professor in the department of obstetrics and gynecology at Washington University, St. Louis.
There is no “best” contraceptive method, Dr. Madden said. “Contraception really needs to be tailored to the individual woman [ensuring] that her values and preferences about contraception are taken into consideration during counseling, to help her choose the method that’s going to be the best fit for her.”
Resources
There are resources available to aid in tailoring contraception methods to the needs of patients.
U.S. Medical Eligibility Criteria for Contraceptive Use, guidance that is available through the Centers for Disease Control and Prevention, allows physicians to tailor the method to a patient’s comorbid medical conditions, Dr. Zite said. Physicians can search by contraceptive method or patient characteristic to determine the risk for a given patient, rated on a scale of 1-4 (where 1 or 2 means generally safe, 3 means that the risks may outweigh the benefits, and 4 means that the risks clearly outweigh the benefits).
“It’s a really easy starting-off point to use with patients and physicians when trying to decide what contraceptive method is safe for their patient,” Dr. Zite said.
Common medical comorbidities, including obesity, diabetes, thyroid disease, and hypertension, are all addressed in the medical eligibility criteria.
Another resource is the U.S. Selected Practice Recommendations for Contraceptive Use, which can help in deciding when it is appropriate to start a contraceptive method, what exams and tests are needed before initiation, what follow-up is needed, and how to handle problems such as missed pills or potential side effects.
Barriers
Over the years, many of the barriers to contraceptive access have been reduced. Some forms of emergency contraception are now available over the counter to women of all ages; more states are considering laws allowing women to access up to a year’s supply of hormonal contraceptives at one time; and a few states have passed laws allowing pharmacists to prescribe hormonal birth control directly. In addition, the Affordable Care Act’s mandate for insurers to cover approved methods of contraception without cost sharing has eliminated some cost barriers.
But other systems barriers still remain, such as making women return for multiple visits for the insertion of an IUD or implant, or limiting LARC use only to women who have already had a child. “There’s not a reason to avoid use of IUDs in women that have not had babies but there are still providers out there who will not insert an IUD, so we need to still do a better job to increase access even more,” Dr. Zite said.
Future trends
What new contraceptive options are in the pipeline? Dr. Prager predicted more development in the area of longer-acting injectables and implants, potentially even biodegradable implants. Also likely is the development of nonsurgical sterilization methods for women that eliminate some of the risk and cost barriers. And male contraceptive methods are in the works, both hormonal and nonhormonal, Dr. Prager said.
There’s an increasing interested in nonhormonal longer-acting methods, beyond just the copper IUD, Dr. Madden said, and there are new products on the horizon in that area. Researchers are exploring new methods to protect against HIV and other sexually transmitted infections, while offering contraception, Dr. Zite said.
Dr. Espey reported having no relevant financial disclosures. Dr. Prager is an unpaid trainer for Nexplanon (Merck). Dr. Zite is an unpaid trainer for Nexplanon and serves on an international IUD advisory board for Bayer. Dr. Madden serves on a scientific advisory board for Bayer and on a data safety monitoring board for phase IV safety studies of Bayer contraceptive products.
Throughout 2016, Ob.Gyn. News is celebrating its 50th anniversary with exclusive articles looking at the evolution of the specialty, including the history of infertility treatment, changes in gynecologic surgery, and the transformation of the well-woman visit. Look for these articles and more special features in the pages of Ob.Gyn. News and online at obgynnews.com.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
WASHINGTON – What has the increased access to contraception over the last 50 years meant for American women?
We asked Ob.Gyn. News editorial advisory board member Dr. Eve Espey, professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque, along with three experts in family planning, to explore how expanded contraception options have affected public health, what barriers still remain, and what new products are in the pipeline.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Perhaps the biggest impact for women has been the ability to participate in the workforce, and that includes women entering medical school.
“Over the last 50 years, we’ve seen a big increase in the number of women who are professionals, who are physicians,” Dr. Espey said during the roundtable. “And during that same period of time, we’ve seen the growth of more focus on family planning.”
“I think that really would not be possible without the ability to control our fertility,” said Dr. Sarah W. Prager, associate professor of obstetrics and gynecology and director of the Ryan Family Planning Program at the University of Washington, Seattle.
And the widespread availability of contraception has translated into a decrease in maternal mortality as a result of fewer unintended pregnancies. “The implications for public health – for women and children in this country – is huge,” Dr. Prager said.
LARCs
One of the big shifts in contraceptive trends has been the slow but increasing uptake of long-acting reversible contraceptives (LARCs), such as IUDs and implants. After a drop-off in the 1970s following safety problems with the Dalkon Shield, there has been a resurgence in interest.
In the last decade, the rate of LARC use has grown from about 2% to 10%, corresponding to a slight drop in rates of unintended pregnancy, said Dr. Nikki B. Zite, professor and residency program director in the department of obstetrics and gynecology at the University of Tennessee, Knoxville.
The Contraceptive CHOICE Project, which enrolled more than 9,000 women who were provided with the no-cost reversible contraceptive method of their choice, found that about three-quarters of the women chose an IUD or an implant, which was associated with significant reductions in unintended and teen pregnancies.
“What we saw was that when we removed barriers to contraception in general, that uptake of IUDs and implants really went up,” said Dr. Tessa Madden, director of the division of family planning and associate professor in the department of obstetrics and gynecology at Washington University, St. Louis.
There is no “best” contraceptive method, Dr. Madden said. “Contraception really needs to be tailored to the individual woman [ensuring] that her values and preferences about contraception are taken into consideration during counseling, to help her choose the method that’s going to be the best fit for her.”
Resources
There are resources available to aid in tailoring contraception methods to the needs of patients.
U.S. Medical Eligibility Criteria for Contraceptive Use, guidance that is available through the Centers for Disease Control and Prevention, allows physicians to tailor the method to a patient’s comorbid medical conditions, Dr. Zite said. Physicians can search by contraceptive method or patient characteristic to determine the risk for a given patient, rated on a scale of 1-4 (where 1 or 2 means generally safe, 3 means that the risks may outweigh the benefits, and 4 means that the risks clearly outweigh the benefits).
“It’s a really easy starting-off point to use with patients and physicians when trying to decide what contraceptive method is safe for their patient,” Dr. Zite said.
Common medical comorbidities, including obesity, diabetes, thyroid disease, and hypertension, are all addressed in the medical eligibility criteria.
Another resource is the U.S. Selected Practice Recommendations for Contraceptive Use, which can help in deciding when it is appropriate to start a contraceptive method, what exams and tests are needed before initiation, what follow-up is needed, and how to handle problems such as missed pills or potential side effects.
Barriers
Over the years, many of the barriers to contraceptive access have been reduced. Some forms of emergency contraception are now available over the counter to women of all ages; more states are considering laws allowing women to access up to a year’s supply of hormonal contraceptives at one time; and a few states have passed laws allowing pharmacists to prescribe hormonal birth control directly. In addition, the Affordable Care Act’s mandate for insurers to cover approved methods of contraception without cost sharing has eliminated some cost barriers.
But other systems barriers still remain, such as making women return for multiple visits for the insertion of an IUD or implant, or limiting LARC use only to women who have already had a child. “There’s not a reason to avoid use of IUDs in women that have not had babies but there are still providers out there who will not insert an IUD, so we need to still do a better job to increase access even more,” Dr. Zite said.
Future trends
What new contraceptive options are in the pipeline? Dr. Prager predicted more development in the area of longer-acting injectables and implants, potentially even biodegradable implants. Also likely is the development of nonsurgical sterilization methods for women that eliminate some of the risk and cost barriers. And male contraceptive methods are in the works, both hormonal and nonhormonal, Dr. Prager said.
There’s an increasing interested in nonhormonal longer-acting methods, beyond just the copper IUD, Dr. Madden said, and there are new products on the horizon in that area. Researchers are exploring new methods to protect against HIV and other sexually transmitted infections, while offering contraception, Dr. Zite said.
Dr. Espey reported having no relevant financial disclosures. Dr. Prager is an unpaid trainer for Nexplanon (Merck). Dr. Zite is an unpaid trainer for Nexplanon and serves on an international IUD advisory board for Bayer. Dr. Madden serves on a scientific advisory board for Bayer and on a data safety monitoring board for phase IV safety studies of Bayer contraceptive products.
Throughout 2016, Ob.Gyn. News is celebrating its 50th anniversary with exclusive articles looking at the evolution of the specialty, including the history of infertility treatment, changes in gynecologic surgery, and the transformation of the well-woman visit. Look for these articles and more special features in the pages of Ob.Gyn. News and online at obgynnews.com.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
WASHINGTON – What has the increased access to contraception over the last 50 years meant for American women?
We asked Ob.Gyn. News editorial advisory board member Dr. Eve Espey, professor and chair of the department of obstetrics and gynecology at the University of New Mexico, Albuquerque, along with three experts in family planning, to explore how expanded contraception options have affected public health, what barriers still remain, and what new products are in the pipeline.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Perhaps the biggest impact for women has been the ability to participate in the workforce, and that includes women entering medical school.
“Over the last 50 years, we’ve seen a big increase in the number of women who are professionals, who are physicians,” Dr. Espey said during the roundtable. “And during that same period of time, we’ve seen the growth of more focus on family planning.”
“I think that really would not be possible without the ability to control our fertility,” said Dr. Sarah W. Prager, associate professor of obstetrics and gynecology and director of the Ryan Family Planning Program at the University of Washington, Seattle.
And the widespread availability of contraception has translated into a decrease in maternal mortality as a result of fewer unintended pregnancies. “The implications for public health – for women and children in this country – is huge,” Dr. Prager said.
LARCs
One of the big shifts in contraceptive trends has been the slow but increasing uptake of long-acting reversible contraceptives (LARCs), such as IUDs and implants. After a drop-off in the 1970s following safety problems with the Dalkon Shield, there has been a resurgence in interest.
In the last decade, the rate of LARC use has grown from about 2% to 10%, corresponding to a slight drop in rates of unintended pregnancy, said Dr. Nikki B. Zite, professor and residency program director in the department of obstetrics and gynecology at the University of Tennessee, Knoxville.
The Contraceptive CHOICE Project, which enrolled more than 9,000 women who were provided with the no-cost reversible contraceptive method of their choice, found that about three-quarters of the women chose an IUD or an implant, which was associated with significant reductions in unintended and teen pregnancies.
“What we saw was that when we removed barriers to contraception in general, that uptake of IUDs and implants really went up,” said Dr. Tessa Madden, director of the division of family planning and associate professor in the department of obstetrics and gynecology at Washington University, St. Louis.
There is no “best” contraceptive method, Dr. Madden said. “Contraception really needs to be tailored to the individual woman [ensuring] that her values and preferences about contraception are taken into consideration during counseling, to help her choose the method that’s going to be the best fit for her.”
Resources
There are resources available to aid in tailoring contraception methods to the needs of patients.
U.S. Medical Eligibility Criteria for Contraceptive Use, guidance that is available through the Centers for Disease Control and Prevention, allows physicians to tailor the method to a patient’s comorbid medical conditions, Dr. Zite said. Physicians can search by contraceptive method or patient characteristic to determine the risk for a given patient, rated on a scale of 1-4 (where 1 or 2 means generally safe, 3 means that the risks may outweigh the benefits, and 4 means that the risks clearly outweigh the benefits).
“It’s a really easy starting-off point to use with patients and physicians when trying to decide what contraceptive method is safe for their patient,” Dr. Zite said.
Common medical comorbidities, including obesity, diabetes, thyroid disease, and hypertension, are all addressed in the medical eligibility criteria.
Another resource is the U.S. Selected Practice Recommendations for Contraceptive Use, which can help in deciding when it is appropriate to start a contraceptive method, what exams and tests are needed before initiation, what follow-up is needed, and how to handle problems such as missed pills or potential side effects.
Barriers
Over the years, many of the barriers to contraceptive access have been reduced. Some forms of emergency contraception are now available over the counter to women of all ages; more states are considering laws allowing women to access up to a year’s supply of hormonal contraceptives at one time; and a few states have passed laws allowing pharmacists to prescribe hormonal birth control directly. In addition, the Affordable Care Act’s mandate for insurers to cover approved methods of contraception without cost sharing has eliminated some cost barriers.
But other systems barriers still remain, such as making women return for multiple visits for the insertion of an IUD or implant, or limiting LARC use only to women who have already had a child. “There’s not a reason to avoid use of IUDs in women that have not had babies but there are still providers out there who will not insert an IUD, so we need to still do a better job to increase access even more,” Dr. Zite said.
Future trends
What new contraceptive options are in the pipeline? Dr. Prager predicted more development in the area of longer-acting injectables and implants, potentially even biodegradable implants. Also likely is the development of nonsurgical sterilization methods for women that eliminate some of the risk and cost barriers. And male contraceptive methods are in the works, both hormonal and nonhormonal, Dr. Prager said.
There’s an increasing interested in nonhormonal longer-acting methods, beyond just the copper IUD, Dr. Madden said, and there are new products on the horizon in that area. Researchers are exploring new methods to protect against HIV and other sexually transmitted infections, while offering contraception, Dr. Zite said.
Dr. Espey reported having no relevant financial disclosures. Dr. Prager is an unpaid trainer for Nexplanon (Merck). Dr. Zite is an unpaid trainer for Nexplanon and serves on an international IUD advisory board for Bayer. Dr. Madden serves on a scientific advisory board for Bayer and on a data safety monitoring board for phase IV safety studies of Bayer contraceptive products.
Throughout 2016, Ob.Gyn. News is celebrating its 50th anniversary with exclusive articles looking at the evolution of the specialty, including the history of infertility treatment, changes in gynecologic surgery, and the transformation of the well-woman visit. Look for these articles and more special features in the pages of Ob.Gyn. News and online at obgynnews.com.
mschneider@frontlinemedcom.com
On Twitter @maryellenny
Hidradenitis Suppurativa Video Roundtable
This 4-part video series is moderated by Jeffrey M. Weinberg, MD, and features discussion among dermatologists on this chronic inflammatory condition that has a significant impact on the quality of life of patients. Moderated by Jeffrey M. Weinberg, MD, this series covers pathogenesis, comorbidities, diagnosis, treatment, and patient education.
This video roundtable was produced by the Custom Programs division of Frontline Medical Communications. The editorial staff of Dermatology News was not involved in developing the video roundtable.
Participants include:
- Robert G. Micheletti, MD
- George Han, MD, PhD
- Mary Ruth Buchness, MD
Disclosure: The faculty received modest honoraria from Frontline Medical Communications for their time participating in this roundtable, and maintained complete editorial control over all content presented.
Dr. Weinberg discloses that he has received honoraria from AbbVie Inc.
Dr. Buchness discloses that she is on the speakers’ bureau for AbbVie Inc.
Dr. Micheletti and Dr. Han have nothing to disclose.
This 4-part video series is moderated by Jeffrey M. Weinberg, MD, and features discussion among dermatologists on this chronic inflammatory condition that has a significant impact on the quality of life of patients. Moderated by Jeffrey M. Weinberg, MD, this series covers pathogenesis, comorbidities, diagnosis, treatment, and patient education.
This video roundtable was produced by the Custom Programs division of Frontline Medical Communications. The editorial staff of Dermatology News was not involved in developing the video roundtable.
Participants include:
- Robert G. Micheletti, MD
- George Han, MD, PhD
- Mary Ruth Buchness, MD
Disclosure: The faculty received modest honoraria from Frontline Medical Communications for their time participating in this roundtable, and maintained complete editorial control over all content presented.
Dr. Weinberg discloses that he has received honoraria from AbbVie Inc.
Dr. Buchness discloses that she is on the speakers’ bureau for AbbVie Inc.
Dr. Micheletti and Dr. Han have nothing to disclose.
This 4-part video series is moderated by Jeffrey M. Weinberg, MD, and features discussion among dermatologists on this chronic inflammatory condition that has a significant impact on the quality of life of patients. Moderated by Jeffrey M. Weinberg, MD, this series covers pathogenesis, comorbidities, diagnosis, treatment, and patient education.
This video roundtable was produced by the Custom Programs division of Frontline Medical Communications. The editorial staff of Dermatology News was not involved in developing the video roundtable.
Participants include:
- Robert G. Micheletti, MD
- George Han, MD, PhD
- Mary Ruth Buchness, MD
Disclosure: The faculty received modest honoraria from Frontline Medical Communications for their time participating in this roundtable, and maintained complete editorial control over all content presented.
Dr. Weinberg discloses that he has received honoraria from AbbVie Inc.
Dr. Buchness discloses that she is on the speakers’ bureau for AbbVie Inc.
Dr. Micheletti and Dr. Han have nothing to disclose.
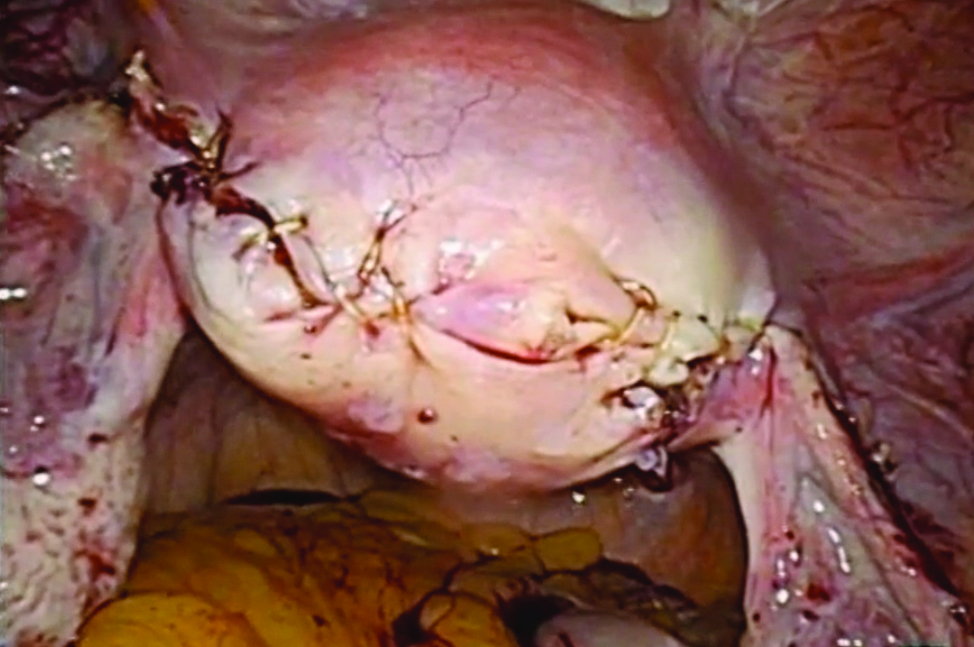
Laparoscopic salpingectomy and cornual resection repurposed
I am pleased to introduce this month’s video, from the Division of Minimally Invasive Gynecologic Surgery (MIGS) at Penn State. Dr. Michelle Pacis addresses an increasingly important topic to MIGS surgeons: removal of previously hysteroscopically placed tubal occlusion devices. These devices offer a permanent alterative to laparoscopic sterilization. If women require their removal, however, whether desired or because of complications, gynecologic surgeons must be familiar with the steps involved.
The following video was produced in order to demonstrate a reproducible technique for laparoscopic removal of a device for women requesting this procedure. Key objectives of the video include:
- review of the techniques available to remove hysteroscopic tubal occlusion devices, as well as the contraindications and advantages/disadvantages of these approaches.
- discuss recommended imaging and materials required for the technique described and tips for their use.
- demonstrate salpingectomy and repair technique.
I hope that you find this month’s video helpful to your surgical practice.

Share your thoughts on this video! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com
I am pleased to introduce this month’s video, from the Division of Minimally Invasive Gynecologic Surgery (MIGS) at Penn State. Dr. Michelle Pacis addresses an increasingly important topic to MIGS surgeons: removal of previously hysteroscopically placed tubal occlusion devices. These devices offer a permanent alterative to laparoscopic sterilization. If women require their removal, however, whether desired or because of complications, gynecologic surgeons must be familiar with the steps involved.
The following video was produced in order to demonstrate a reproducible technique for laparoscopic removal of a device for women requesting this procedure. Key objectives of the video include:
- review of the techniques available to remove hysteroscopic tubal occlusion devices, as well as the contraindications and advantages/disadvantages of these approaches.
- discuss recommended imaging and materials required for the technique described and tips for their use.
- demonstrate salpingectomy and repair technique.
I hope that you find this month’s video helpful to your surgical practice.

Share your thoughts on this video! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com
I am pleased to introduce this month’s video, from the Division of Minimally Invasive Gynecologic Surgery (MIGS) at Penn State. Dr. Michelle Pacis addresses an increasingly important topic to MIGS surgeons: removal of previously hysteroscopically placed tubal occlusion devices. These devices offer a permanent alterative to laparoscopic sterilization. If women require their removal, however, whether desired or because of complications, gynecologic surgeons must be familiar with the steps involved.
The following video was produced in order to demonstrate a reproducible technique for laparoscopic removal of a device for women requesting this procedure. Key objectives of the video include:
- review of the techniques available to remove hysteroscopic tubal occlusion devices, as well as the contraindications and advantages/disadvantages of these approaches.
- discuss recommended imaging and materials required for the technique described and tips for their use.
- demonstrate salpingectomy and repair technique.
I hope that you find this month’s video helpful to your surgical practice.

Share your thoughts on this video! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com
VIDEO: Nearly half of Medicaid patients denied antivirals for HCV
State Medicaid programs denied nearly half of requests to cover direct-acting antiviral drugs for patients with chronic hepatitis C virus (HCV) infection, according to a prospective study of beneficiaries in Delaware, Maryland, New Jersey, and Pennsylvania reported in the July issue of Clinical Gastroenterology and Hepatology.
In contrast, only 5% of Medicare patients and 10% of privately insured patients were denied coverage, said Dr. Vincent Lo Re III of the University of Pennsylvania, Philadelphia, together with his associates. “Notably, nearly one-quarter of Medicaid recipients with cirrhosis experienced treatment denial. Medicaid patients from these states also experienced a longer time to prescription fill than those with Medicare or commercial insurance,” the researchers said.
Their study included 2,321 HCV patients prescribed a direct-acting antiviral regimen between November 1, 2014 and April 30, 2015. All prescriptions were submitted to a specialty pharmacy that serves HCV patients in Maryland, Pennsylvania, Delaware, and New Jersey, the investigators noted. They focused on “absolute denial,” meaning that the prescription was never filled because the insurer denied coverage, regardless of appeals. “Data are lacking on the incidence of absolute denial of direct-acting antiviral prescriptions and factors associated with this outcome,” the investigators said. “These data are important because absolute denial of HCV treatment by insurers might have adverse outcomes on patients and could harm patient-provider relationships” (Clin Gastroenterol Hepatol. 2016 Apr 5. doi: 10.1016/j.cgh.2016.03.040).
Source: American Gastroenterological Association
A total of 1,023 study patients were privately insured, 795 were enrolled in Medicare, and 503 were Medicaid patients, according to the researchers. In all, 377 patients (16%) received absolute denials, most often because of “insufficient information to assess medical need” (36% of denials) and “lack of medical necessity” (35%). Medicaid patients faced an absolute denial rate of 46% – significantly higher than rates for private insurers (10.5%) or Medicare (5%; P less than .001 for both comparisons). After adjusting for potential confounders, Medicaid patients were more than four times as likely to be denied coverage for direct-acting antivirals, compared with privately insured patients (relative risk, 4.1; 95% confidence interval, 3.4-5.1). Delaware’s Medicaid program had the highest rate of absolute denial (57%), followed by Pennsylvania (48%), Maryland (47%), and New Jersey (37%).
Medicaid programs even refused to cover direct-acting antiviral prescriptions for 25% of patients who had cirrhosis, compared with absolute denial rates of only 1% for Medicare and 3% of private insurers (P less than .001 for both comparisons), according to the researchers. The implications of these denials “remain unknown,” but lack of treatment increases the risk of end-stage liver disease, hepatocellular carcinoma, extrahepatic disease, HCV transmission, and “anxiety and stress about HCV disease progression, [which can] provoke distrust among patients of the health care system and their providers,” they added. “Clinicians then are challenged to explain the denial, and important opportunities for patient engagement, education, and cure could be irrevocably lost.”
Medicaid programs also initially denied about 25% of prescriptions before eventually approving them, despite the fact that “patients had complete prior authorization requests that should have contained the materials needed to justify approval,” said the investigators. Furthermore, denial letters usually did not specify the information that was needed, “making it difficult [for clinicians] to appeal the decision.” In contrast, only about 8% of privately insured patients and 13% of Medicare enrollees were initially denied coverage.
Prescriptions as a whole were more likely to be filled in the last 3 months of the study than earlier, perhaps because insurers are starting to relax their reimbursement criteria, said the investigators. Indeed, in October 2015, the American Association for the Study of Liver Diseases and the Infectious Disease Society of America stopped triaging groups of patients for direct-acting antiviral therapy, they noted.
The research was supported by the Penn Center for AIDS Research, which is funded by the National Institutes of Health. Dr. Lo Re received investigator-initiated research support from AstraZeneca. Five coinvestigators reported relationships with a number of pharmaceutical companies. Four coinvestigators reported employment by the study site, Burman’s Specialty Pharmacy. The other four coauthors had no disclosures.
State Medicaid programs denied nearly half of requests to cover direct-acting antiviral drugs for patients with chronic hepatitis C virus (HCV) infection, according to a prospective study of beneficiaries in Delaware, Maryland, New Jersey, and Pennsylvania reported in the July issue of Clinical Gastroenterology and Hepatology.
In contrast, only 5% of Medicare patients and 10% of privately insured patients were denied coverage, said Dr. Vincent Lo Re III of the University of Pennsylvania, Philadelphia, together with his associates. “Notably, nearly one-quarter of Medicaid recipients with cirrhosis experienced treatment denial. Medicaid patients from these states also experienced a longer time to prescription fill than those with Medicare or commercial insurance,” the researchers said.
Their study included 2,321 HCV patients prescribed a direct-acting antiviral regimen between November 1, 2014 and April 30, 2015. All prescriptions were submitted to a specialty pharmacy that serves HCV patients in Maryland, Pennsylvania, Delaware, and New Jersey, the investigators noted. They focused on “absolute denial,” meaning that the prescription was never filled because the insurer denied coverage, regardless of appeals. “Data are lacking on the incidence of absolute denial of direct-acting antiviral prescriptions and factors associated with this outcome,” the investigators said. “These data are important because absolute denial of HCV treatment by insurers might have adverse outcomes on patients and could harm patient-provider relationships” (Clin Gastroenterol Hepatol. 2016 Apr 5. doi: 10.1016/j.cgh.2016.03.040).
Source: American Gastroenterological Association
A total of 1,023 study patients were privately insured, 795 were enrolled in Medicare, and 503 were Medicaid patients, according to the researchers. In all, 377 patients (16%) received absolute denials, most often because of “insufficient information to assess medical need” (36% of denials) and “lack of medical necessity” (35%). Medicaid patients faced an absolute denial rate of 46% – significantly higher than rates for private insurers (10.5%) or Medicare (5%; P less than .001 for both comparisons). After adjusting for potential confounders, Medicaid patients were more than four times as likely to be denied coverage for direct-acting antivirals, compared with privately insured patients (relative risk, 4.1; 95% confidence interval, 3.4-5.1). Delaware’s Medicaid program had the highest rate of absolute denial (57%), followed by Pennsylvania (48%), Maryland (47%), and New Jersey (37%).
Medicaid programs even refused to cover direct-acting antiviral prescriptions for 25% of patients who had cirrhosis, compared with absolute denial rates of only 1% for Medicare and 3% of private insurers (P less than .001 for both comparisons), according to the researchers. The implications of these denials “remain unknown,” but lack of treatment increases the risk of end-stage liver disease, hepatocellular carcinoma, extrahepatic disease, HCV transmission, and “anxiety and stress about HCV disease progression, [which can] provoke distrust among patients of the health care system and their providers,” they added. “Clinicians then are challenged to explain the denial, and important opportunities for patient engagement, education, and cure could be irrevocably lost.”
Medicaid programs also initially denied about 25% of prescriptions before eventually approving them, despite the fact that “patients had complete prior authorization requests that should have contained the materials needed to justify approval,” said the investigators. Furthermore, denial letters usually did not specify the information that was needed, “making it difficult [for clinicians] to appeal the decision.” In contrast, only about 8% of privately insured patients and 13% of Medicare enrollees were initially denied coverage.
Prescriptions as a whole were more likely to be filled in the last 3 months of the study than earlier, perhaps because insurers are starting to relax their reimbursement criteria, said the investigators. Indeed, in October 2015, the American Association for the Study of Liver Diseases and the Infectious Disease Society of America stopped triaging groups of patients for direct-acting antiviral therapy, they noted.
The research was supported by the Penn Center for AIDS Research, which is funded by the National Institutes of Health. Dr. Lo Re received investigator-initiated research support from AstraZeneca. Five coinvestigators reported relationships with a number of pharmaceutical companies. Four coinvestigators reported employment by the study site, Burman’s Specialty Pharmacy. The other four coauthors had no disclosures.
State Medicaid programs denied nearly half of requests to cover direct-acting antiviral drugs for patients with chronic hepatitis C virus (HCV) infection, according to a prospective study of beneficiaries in Delaware, Maryland, New Jersey, and Pennsylvania reported in the July issue of Clinical Gastroenterology and Hepatology.
In contrast, only 5% of Medicare patients and 10% of privately insured patients were denied coverage, said Dr. Vincent Lo Re III of the University of Pennsylvania, Philadelphia, together with his associates. “Notably, nearly one-quarter of Medicaid recipients with cirrhosis experienced treatment denial. Medicaid patients from these states also experienced a longer time to prescription fill than those with Medicare or commercial insurance,” the researchers said.
Their study included 2,321 HCV patients prescribed a direct-acting antiviral regimen between November 1, 2014 and April 30, 2015. All prescriptions were submitted to a specialty pharmacy that serves HCV patients in Maryland, Pennsylvania, Delaware, and New Jersey, the investigators noted. They focused on “absolute denial,” meaning that the prescription was never filled because the insurer denied coverage, regardless of appeals. “Data are lacking on the incidence of absolute denial of direct-acting antiviral prescriptions and factors associated with this outcome,” the investigators said. “These data are important because absolute denial of HCV treatment by insurers might have adverse outcomes on patients and could harm patient-provider relationships” (Clin Gastroenterol Hepatol. 2016 Apr 5. doi: 10.1016/j.cgh.2016.03.040).
Source: American Gastroenterological Association
A total of 1,023 study patients were privately insured, 795 were enrolled in Medicare, and 503 were Medicaid patients, according to the researchers. In all, 377 patients (16%) received absolute denials, most often because of “insufficient information to assess medical need” (36% of denials) and “lack of medical necessity” (35%). Medicaid patients faced an absolute denial rate of 46% – significantly higher than rates for private insurers (10.5%) or Medicare (5%; P less than .001 for both comparisons). After adjusting for potential confounders, Medicaid patients were more than four times as likely to be denied coverage for direct-acting antivirals, compared with privately insured patients (relative risk, 4.1; 95% confidence interval, 3.4-5.1). Delaware’s Medicaid program had the highest rate of absolute denial (57%), followed by Pennsylvania (48%), Maryland (47%), and New Jersey (37%).
Medicaid programs even refused to cover direct-acting antiviral prescriptions for 25% of patients who had cirrhosis, compared with absolute denial rates of only 1% for Medicare and 3% of private insurers (P less than .001 for both comparisons), according to the researchers. The implications of these denials “remain unknown,” but lack of treatment increases the risk of end-stage liver disease, hepatocellular carcinoma, extrahepatic disease, HCV transmission, and “anxiety and stress about HCV disease progression, [which can] provoke distrust among patients of the health care system and their providers,” they added. “Clinicians then are challenged to explain the denial, and important opportunities for patient engagement, education, and cure could be irrevocably lost.”
Medicaid programs also initially denied about 25% of prescriptions before eventually approving them, despite the fact that “patients had complete prior authorization requests that should have contained the materials needed to justify approval,” said the investigators. Furthermore, denial letters usually did not specify the information that was needed, “making it difficult [for clinicians] to appeal the decision.” In contrast, only about 8% of privately insured patients and 13% of Medicare enrollees were initially denied coverage.
Prescriptions as a whole were more likely to be filled in the last 3 months of the study than earlier, perhaps because insurers are starting to relax their reimbursement criteria, said the investigators. Indeed, in October 2015, the American Association for the Study of Liver Diseases and the Infectious Disease Society of America stopped triaging groups of patients for direct-acting antiviral therapy, they noted.
The research was supported by the Penn Center for AIDS Research, which is funded by the National Institutes of Health. Dr. Lo Re received investigator-initiated research support from AstraZeneca. Five coinvestigators reported relationships with a number of pharmaceutical companies. Four coinvestigators reported employment by the study site, Burman’s Specialty Pharmacy. The other four coauthors had no disclosures.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Key clinical point: Nearly half of Medicaid patients with chronic hepatitis C infections were denied coverage for direct-acting antivirals in a prospective study.
Major finding: In all, 46% of Medicaid patients received absolute denials (46%), compared with 5% of Medicare patients and 10% of privately insured patients (P less than .001 for both comparisons).
Data source: A prospective cohort study of 2,321 patients who submitted prescriptions for direct-acting antivirals to a specialty pharmacy serving the HCV populations of Delaware, Maryland, New Jersey, and Pennsylvania.
Disclosures: The research was supported by the Penn Center for AIDS Research, which is funded by the National Institutes of Health. Dr. Lo Re received investigator-initiated research support from AstraZeneca. Five coinvestigators reported relationships with a number of pharmaceutical companies. Four coinvestigators reported employment by the study site, Burman’s Specialty Pharmacy. The other four coauthors had no disclosures.
Rediscovering clozapine
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
Sylvia Lucas, MD, PhD
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel