User login
Bringing you the latest news, research and reviews, exclusive interviews, podcasts, quizzes, and more.
div[contains(@class, 'header__large-screen')]
div[contains(@class, 'read-next-article')]
div[contains(@class, 'nav-primary')]
nav[contains(@class, 'nav-primary')]
section[contains(@class, 'footer-nav-section-wrapper')]
footer[@id='footer']
div[contains(@class, 'main-prefix')]
section[contains(@class, 'nav-hidden')]
div[contains(@class, 'ce-card-content')]
nav[contains(@class, 'nav-ce-stack')]
The Future of Psoriasis Care
The Future of Psoriasis Care
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
Psoriasis research and treatment have come a long way in the past 2 to 3 decades. With the advent of biologic therapy, increasingly more targeted therapies, and a better pathophysiological understanding, our treatment paradigms and ability to treat psoriatic disease have shown great improvement; however, despite these advances, there remain several areas in need of further development to continue to improve our care of patients with psoriasis, including comorbidities, access to care, technology, and clinical care.
Treatment Implications of Comorbidities
It has become increasingly clear that psoriasis carries with it numerous medical and psychiatric comorbidities; however, our ability to utilize these factors in treatment decision-making is still nascent. Clinically, multiple studies have demonstrated a connection between cardiovascular disease (CVD) and psoriasis, often with a direct relationship between CVD and psoriasis severity.1-3 The cytokines involved (interleukin [IL]–17) and cell types (primarily neutrophils) are the same in psoriatic disease and evolving atherosclerotic plaques.4,5 In contrast, other analyses do not support a relationship between CVD and psoriasis, and there has been no direct and definitive demonstration that giving patients a specific psoriasis treatment could help reduce cardiovascular risk. Perhaps this is due to the sample sizes and time needed to demonstrate such a connection, as we are dealing with fairly rare events overall. Strategies to identify patients at risk for cardiovascular events, such as starting from a cohort with existing CVD and investigating treatment effects in that population, may yield worthwhile dividends. Perhaps one day we will be able to offer treatments that not only help clear psoriasis but also modulate cardiovascular health.
Our understanding of the psychiatric effects of psoriasis is even less developed. The strongest links have been demonstrated between psoriasis and depression, anxiety, and suicidal ideation.6 Some of these connections have been recognized for more than 3 decades: one study from 1993 showed that almost 10% of patients with psoriasis wished to be dead and 5.6% reported active suicidal ideation at the time of the study.7 Why is it, then, that we still do not have a good understanding of the interrelationship between psoriasis, mental health, and therapeutics? There likely is a connection between these components, as it is now well accepted that cytokines (eg, interferons) can have a considerable impact on depression and that treatment with biologics for psoriasis tends to improve depressive symptoms.8 This is an area in which we need better awareness and understanding as well as some guidance on how to approach this topic with our patients—particularly how mental health may play into therapeutic decisions for psoriasis, such as earlier escalation to rapid-acting systemic therapy in patients with psychiatric comorbidities.
Access to Psoriasis Care
With so many effective treatments for psoriasis, one of the most frustrating challenges we face is that many patients with psoriasis still experience notable barriers to care. While access in urban areas generally is reasonable, in rural areas, 75% of patients have no psoriasis-treating providers in their ZIP code and have to seek psoriasis-related care outside the 3-digit ZIP code prefix.9 Unfortunately, in most cases, even after traveling and waiting for an appointment patients will not be offered the full spectrum of available psoriasis treatments. Dermatologists already are much harder to find in rural areas, but the proportion of rural counties without a dermatologist who prescribes biologics approaches 90%.10 Functionally, this places a huge burden on our patients, who frustratingly see commercials for highly effective psoriasis treatments on television but are not able to access them. What good is having medicines that can help more than two-thirds of patients achieve 100% clearance11 when patients cannot access them?
Technology and Treatment Optimization
As our society becomes ever more technologically advanced, medicine seems to be caught in a bit of a quagmire, with our practices often using outdated technology in the name of HIPAA compliance and communicating via fax on important matters such as medication coverage. Nevertheless, dermatologists are beginning to increase integration of artificial intelligence (AI) and advanced technologies to make patient care more efficient and effective via education/awareness, image analysis, remote management, and telemedicine.12 Recently, the National Psoriasis Foundation published guidance for the use of telemedicine, suggesting that it could be used for expanded access and expedited care in appropriate settings.13 However, some caution should be used when interpreting data in this sphere. While AI technology has been purported to outpace dermatologists’ diagnosis of psoriasis in some cases, the conditions tested (ie, the training set and evaluation image bank) and special tools used (such as dermoscopy, which is not routinely used in clinical practice for psoriasis diagnosis) may make the results inapplicable to general care.14
Perhaps more promising is the use of digital aids to help with long-term care, treatment reminders, and comorbidity evaluation/screening. Similarly, telemedicine can be utilized to provide skilled psoriasis care to patients in rural areas who otherwise might not have access. One such program demonstrated that asynchronous e-consults were able to achieve Psoriasis Area and Severity Index (PASI) and body surface area outcomes similar to in-person dermatologist care.15 Using AI and technology also could assist with drug development and guide treatment. For example, a psoriatic arthritis (PsA) risk model developed in a Danish cohort suggested that early treatment with an IL-17 inhibitor in high-risk patients could reduce PsA incidence by 64%.16
Personalized Clinical Care
Even as we become accustomed to higher PASI 90, PASI 100, and mean PASI improvement numbers with our newer biologics, drug development in psoriasis has not stopped. Pipeline medications include an oral peptide-based IL-23 inhibitor17 and targeted tyrosine kinase 2 inhibitors.18,19 What is perhaps most interesting is to envision a future in which we could select treatments based on either patient phenotype (eg, involvement of hands and feet could suggest a certain single or class of medicine) or genotype.20 This has clear impacts on patient care, as dedicated trials of psoriasis medications tend to result in lower achievement of outcome measure thresholds than subanalyses of clinical trials; for example, in a dedicated trial of risankizumab for nonpustular palmoplantar psoriasis, achievement of a palmoplantar Investigator Global Assessment score of clear or almost clear was demonstrated in 33.3% of treated patients vs 16.1% of those receiving placebo at week 16 (P=.006).21 A subanalysis from the pivotal UltiMMA trials showed that more than 70% of risankizumab-treated patients achieved complete clearance (palmoplantar PASI score of 0) by week 16.22 Indeed, there is some evidence to suggest that the pathophysiology of plaque psoriasis, nonpustular palmoplantar psoriasis, and palmoplantar pustular psoriasis are different, with more interferon-γ signaling involved in nonpustular palmoplantar psoriasis23—which may explain why some limited case reports have suggested the use of Janus kinase inhibitors for recalcitrant cases of palmoplantar plaque psoriasis.24
Even with such high rates of skin clearance, the treatment landscape in PsA lags behind. There is a need for higher-efficacy treatments in PsA. On a positive note, it may be reflective of how advanced our treatment conversations about psoriasis have become that rather than analyzing gross PASI improvements between one drug and another, we now are able to address nuanced differences between various presentations of psoriasis to help us select the right tool from our treatment toolbox.
Final Thoughts
We are lucky to practice dermatology in a time when there has been so much development, with many good treatment options for patients with psoriasis. What we had thought of as the ultimate goal in the past—to get the skin relatively clear—is now a realistic outcome for most patients. This allows us to focus on other important considerations, such as assessing and addressing comorbidities, improving access to care, implementing technology to improve psoriasis care, and refining our understanding of how different manifestations of psoriasis should alter our approach to treating patients. And though we have come a long way in recent years, there still is much to be done to lift up the psoriasis community as a whole. It’s reassuring to know that many are still working toward this goal.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
- Cui P, Li D, Shi L, et al. Cardiovascular comorbidities among patients with psoriasis: a national register-based study in China. Sci Rep. 2024;14:19683.
- Tinggaard AB, Hjuler KF, Andersen IT, et al. Prevalence and severity of coronary artery disease linked to prognosis in psoriasis and psoriatic arthritis patients: a multi-centre cohort study. J Intern Med. 2021;290:693-703.
- Yang YW, Keller JJ, Lin HC. Medical comorbidity associated with psoriasis in adults: a population-based study. Br J Dermatol. 2011;165:1037-1043.
- Silvestre-Roig C, Braster Q, Ortega-Gomez A, et al. Neutrophils as regulators of cardiovascular inflammation. Nat Rev Cardiol. 2020;17:327-340.
- von Stebut E, Boehncke WH, Ghoreschi K, et al. IL-17A in psoriasis and beyond: cardiovascular and metabolic implications. Front Immunol. 2019;10:3096.
- Kurd SK, Troxel AB, Crits-Christoph P, et al. The risk of depression, anxiety, and suicidality in patients with psoriasis: a population-based cohort study. Arch Dermatol. 2010;146:891-895.
- Gupta MA, Schork NJ, Gupta AK, et al. Suicidal ideation in psoriasis. Int J Dermatol. 1993;32:188-190.
- Mrowietz U, Sumbul M, Gerdes S. Depression, a major comorbidity of psoriatic disease, is caused by metabolic inflammation. J Eur Acad Dermatol Venereol. 2023;37:1731-1738.
- Seigel L, Shoaib S, Maughn K, et al. Health disparities in psoriasis: geographic barriers to access in the United States. J Dermatolog Treat. 2024;35:2365820.
- Sun QW, Feng H, Cohen JM. Geographic trends in psoriasis injectable biologic prescriptions by dermatologists among Medicare beneficiaries. J Am Acad Dermatol. 2024;91:1214-1216.
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152.
- Wongvibulsin S, Lee I. Artificial intelligence and dermatology. JAMA Dermatol. 2025;161:344.
- Pelet Del Toro N, Yahia R, Feldman SR, et al. National Psoriasis Foundation Telemedicine Task Force guidance for management of psoriatic disease via telemedicine. JAAD Int. 2023;12:32-36.
- Yang Y, Wang J, Xie F, et al. A convolutional neural network trained with dermoscopic images of psoriasis performed on par with 230 dermatologists. Comput Biol Med. 2021;139:104924.
- Armstrong AW, Chambers CJ, Maverakis E, et al. Effectiveness of online vs in-person care for adults with psoriasis: a randomized clinical trial. JAMA Netw Open. 2018;1:E183062.
- Gong Z, Cheng Y, Wei X, et al. Digital technologies in psoriasis management: from precision diagnosis to therapeutic innovation and holistic care. Front Digit Health. 2025;8:1656585.
- Bissonnette R, Soung J, Hebert AA, et al. Oral icotrokinra for plaque psoriasis in adults and adolescents. N Engl J Med. 2025;393:1784-1795.
- Armstrong AW, Gooderham M, Lynde C, et al. Tyrosine kinase 2 inhibition with zasocitinib (TAK-279) in psoriasis: a randomized clinical trial. JAMA Dermatol. 2024;160:1066-1074.
- Blauvelt A, Arenberger P, Sauder MB, et al. Highly selective, allosteric inhibition of TYK2 with oral ESK-001 in patients with moderate-to-severe plaque psoriasis: results from STRIDE, a 12-week, randomized, double-blinded, placebo-controlled, dose-ranging phase 2 study. J Am Acad Dermatol. 2026;94:57-65.
- Tchack M KN, Sandeep R, Muller E, et al. Gene expression or mutational profile for psoriasis in guiding treatment: a systematic review. JAAD Reviews. 2025;5:100-102.
- Lebwohl M, Bukhalo M, Stein Gold L, et al. A randomized phase 3b study evaluating the safety and efficacy of risankizumab in adult patients with moderate-to-severe plaque psoriasis with non-pustular palmoplantar involvement. J Am Acad Dermatol. 2024;91:1150-1157.
- Strober B, Bachelez H, Crowley J, et al. Efficacy of long-term risankizumab treatment for moderate-to-severe plaque psoriasis: subgroup analyses by baseline characteristics and psoriatic disease manifestations through 256 weeks (LIMMitless trial). J Eur Acad Dermatol Venereol. 2024;38:864-872.
- Wang CQ, Haxhinasto S, Garcet S, et al. Comparison of the inflammatory circuits in psoriasis vulgaris, non‒pustular palmoplantar psoriasis, and palmoplantar pustular psoriasis. J Invest Dermatol. 2023;143:87-97.
- Choi B, Li HO, Glassman SJ. Palmoplantar plaque psoriasis responsive to upadacitinib: a report of two cases. SAGE Open Med Case Rep. 2025;13:2050313X251317763.
The Future of Psoriasis Care
The Future of Psoriasis Care
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Combination Guselkumab and Secukinumab for Plaque Psoriasis
To the Editor:
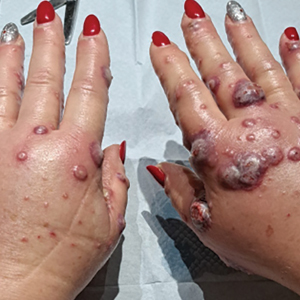
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.
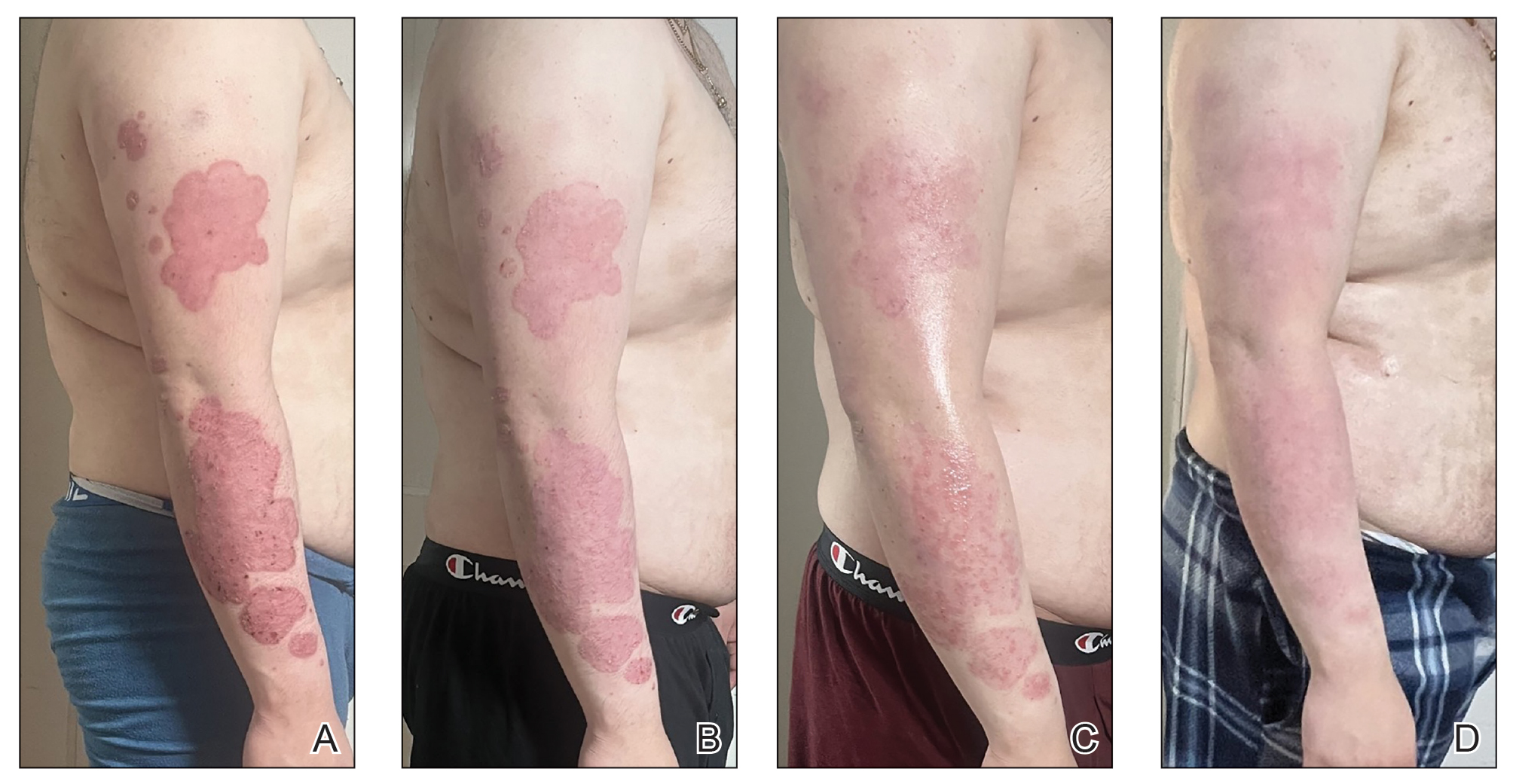
Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.
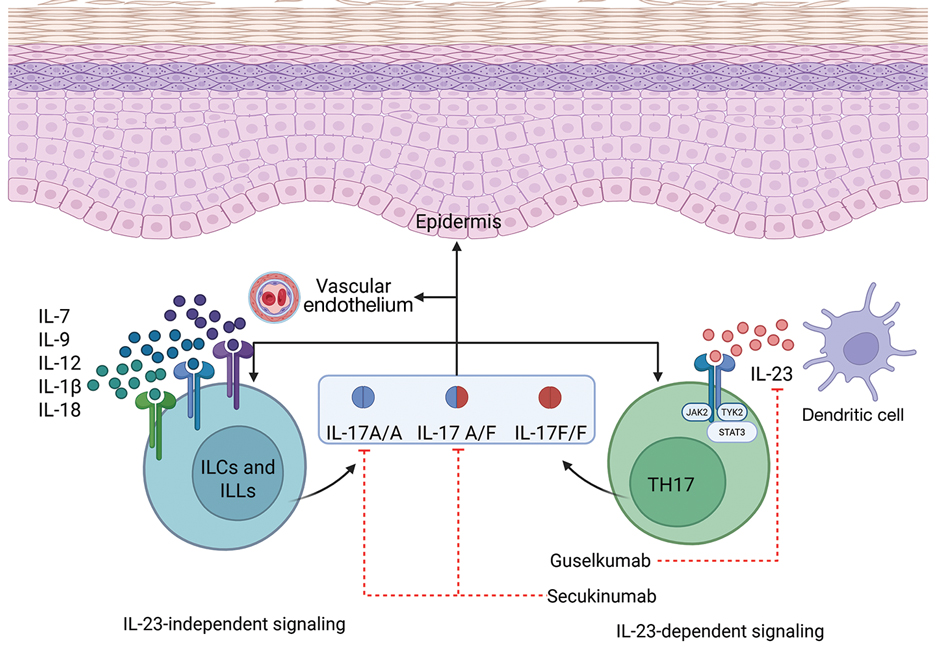
The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
To the Editor:
A 23-year-old man with plaque psoriasis experienced a flare after a 3-month interruption in his secukinumab therapy. The patient’s psoriasis previously was well controlled with secukinumab 300 mg every 4 weeks. Due to social impairment from the current flare, he received a one-time 100-mg dose of sample guselkumab. Secukinumab therapy was resumed 3 weeks later at 300 mg every 4 weeks. Improvement was gradual; 3 weeks after receiving guselkumab, plaques remained on the bilateral dorsal forearms and the lower abdomen. By 2 months posttreatment, there was decreased erythema and reduced scaling with the continued secukinumab therapy (eFigure 1). The patient reported complete clearance of the plaques 4 months after receiving guselkumab but experienced a brief recurrence at month 5. By month 6, his psoriasis returned to its prior baseline of mild disease. In this 6-month period after resuming treatment, the patient reported subjective improvement in itch and quality of life. He reported no infection or other adverse effects from the additional therapy.

Psoriasis is driven by the interleukin (IL)-23–tyrosine kinase (TYK) 2–IL-17 axis.1 Guselkumab, an IL-23 inhibitor, and secukinumab, an IL-17A inhibitor, target distinct yet interconnected pathways in the pathogenesis of psoriasis.1 A key upstream regulator of the IL-17 pathway, IL-23 drives the expansion and survival of T helper 17 cells, which in turn produce IL-17A and IL-17F—cytokines involved in psoriatic inflammation.1 While monotherapy with either IL-23 or IL-17A inhibition generally is safe and highly effective, it does not always provide adequate disease control. By simultaneously inhibiting IL-23 and IL-17A, a combined approach may offer enhanced disease control compared to monotherapy.
Combined therapy with a single 100-mg dose of guselkumab and secukinumab 300 mg every 4 weeks may be effective for short-term control in patients who experience psoriasis flares. Although guselkumab has a half-life of approximately 15 to 18 days, its effects may extend beyond 4 to 5 half-lives, as improvement was seen in our case for 57 to 180 days. Although we did not test for the development of antidrug antibodies to secukinumab, there is evidence that the incidence of such development is exceedingly low and not correlated with clinical efficacy.2 We cannot exclude the possibility that the single dose of guselkumab may have been responsible for the entirety of our patient’s improvement, as up to 46% of patients treated with 12 months of guselkumab continued to experience remission at 6 months after discontinuation.3 Population data suggest a longer duration of activity or disease-modifying potential of guselkumab; however, affected patients had a longer duration of initial treatment before cessation.3
While data on the safety of dual cytokine inhibition in psoriasis are limited, patients in clinical trials for psoriatic arthritis often are treated with a biologic and methotrexate and/or oral corticosteroids; the safety of biologics even with such a combination is excellent.4 Combination biologic therapy with ustekinumab and etanercept in refractory psoriasis caused few adverse events.4,5 Combination tumor necrosis factor α and IL-12/23 inhibition is comparatively more immunosuppressive than combination IL-23 and IL-17 inhibition. Additionally, there may be redundancies in the immune system to the IL-23–TYK-2–IL-17 axis.1 While inhibition of both IL-17A and IL-17F increases cutaneous candidiasis risk, inhibiting IL-23 or IL-17A alone is associated with less risk for candidal infection.6,7 This suggests that redundancy via IL-17F derived from non-IL-23–dependent pathways may offer protection during dual inhibition (eFigure 2). Our patient experienced no adverse effects with a single 100-mg dose of guselkumab, suggesting that at least short-term combination therapy may be well tolerated.

The safety profile seen in our case supports forgoing a washout period when switching between biologics, which can delay access to much-needed therapy and induce flares. Our patient had no adverse events, which provides anecdotal evidence supporting initiation of new biologics without delay. It is possible that the risks of overlapping two low-risk biologics may be lower than combining one biologic with a more immunosuppressive oral immunomodulator.
Combining standard-therapy secukinumab with a one-time dose of guselkumab may provide a synergistic effect by targeting different cytokines involved in the pathogenesis of psoriasis. Using 2 monoclonal antibodies with complementary targets may offer a strategy for rapid control of severe psoriasis that is refractory to single biologics.
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315. doi:10.1016/S0140-6736(20)32549-6
- Tsakok T, Rispens T, Spuls P, et al. Immunogenicity of biologic therapies in psoriasis: myths, facts and a suggested approach. J Eur Acad Dermatol Venereol. 2021;35:329-337. doi:10.1111/jdv.16980
- Zhdanava M, Fitzgerald T, Pilon D, et al. Comparative analysis of persistence and remission with guselkumab versus secukinumab and ixekizumab in the United States. J Dermatolog Treat. 2024;35:1. doi:10.1080/09546634.2024.2349658
- Kumthekar A, Ashrafi M, Deodhar A. Difficult to treat psoriatic arthritis—how should we manage? Clin Rheumatol. 2023;42:2251-2265. doi:10.1007/S10067-023-06605-9
- Thibodeaux Q, Ly K, Reddy V, et al. Dual biologic therapy for recalcitrant psoriasis and psoriatic arthritis. JAAD Case Rep. 2019;5:928-930. doi:10.1016/j.jdcr.2019.08.015
- Lebwohl MG, Merola JF, Rowland K, et al. Safety of guselkumab treatment for up to 5 years in patients with moderate-to-severe psoriasis: pooled analyses across seven clinical trials with more than 8600 patient-years of exposure. Br J Dermatol. 2023;189:42-52. doi:10.1093/BJD/LJAD115
- Reich K, Warren RB, Lebwohl M, et al. Bimekizumab versus secukinumab in plaque psoriasis. N Engl J Med. 2021;385:142-152. doi:10.1056/NEJMOA2102383
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Combination Guselkumab and Secukinumab for Plaque Psoriasis
Practice Points
- Combination therapy with 2 complementary biologics for psoriasis may have synergistic effects.
- Washout periods likely are unnecessary when switching between guselkumab and secukinumab.
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
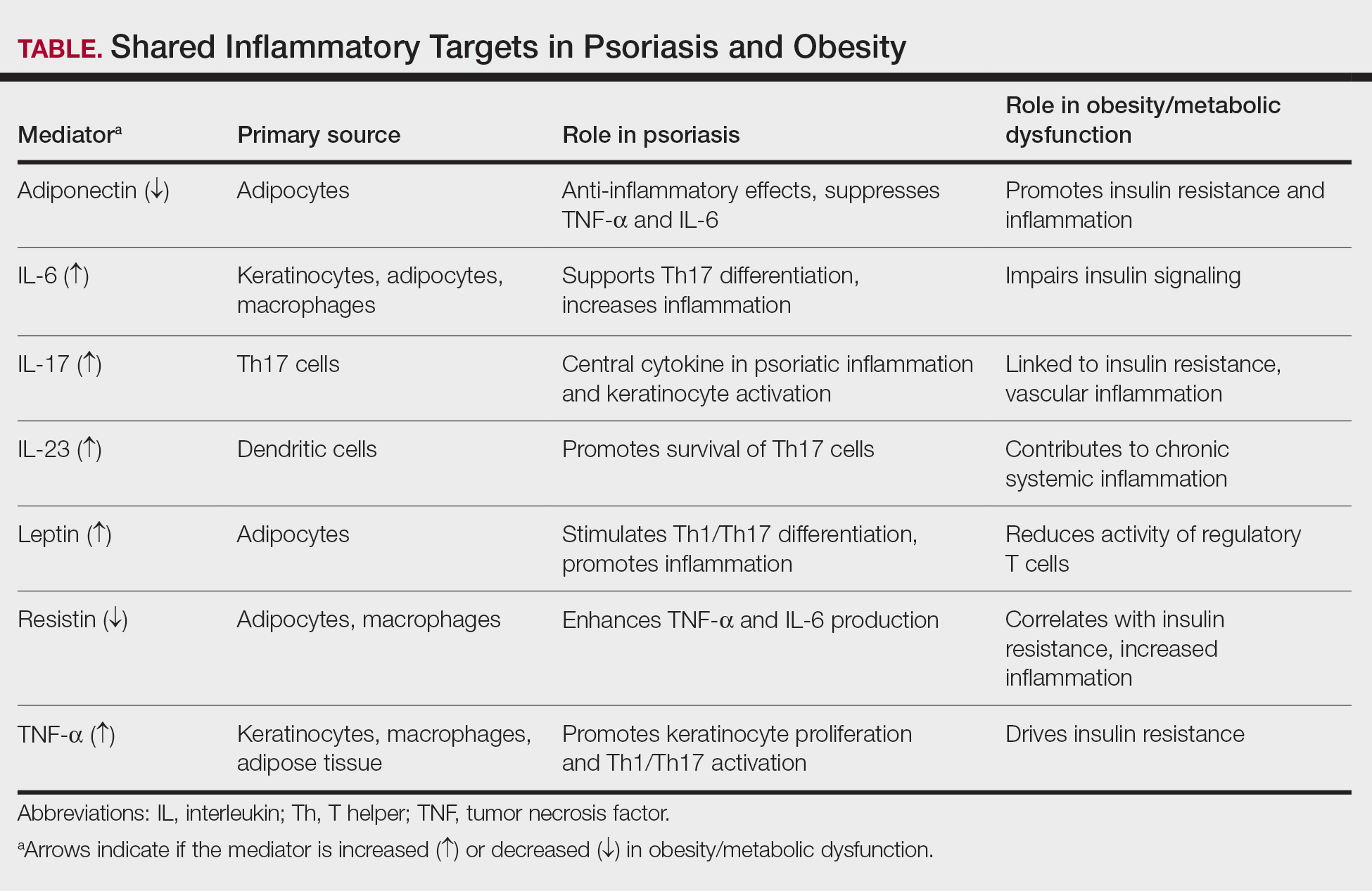
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).
Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
Psoriasis, a chronic immune-mediated skin disease, is increasingly recognized for its systemic inflammation and associated cardiometabolic risk. The global rise of obesity has revealed a critical link between these 2 conditions. Current evidence confirms that obesity is an independent risk factor that can trigger psoriasis onset, drive more severe disease, and substantially impair treatment efficacy.1,2 This review synthesizes the latest evidence on the shared pathophysiology, clinical consequences, and integrated management strategies for patients with both psoriasis and obesity.
Pathophysiologic Links Between Psoriasis and Obesity
Emerging evidence highlights a bidirectional relationship between psoriasis and obesity rooted in overlapping inflammatory pathways. Both conditions are characterized by chronic inflammation mediated by cytokines that sustain systemic immune activation and metabolic dysfunction. This interplay creates a reciprocal process in which psoriatic inflammation promotes metabolic disturbances while obesity amplifies systemic inflammation and disease severity.3
Psoriasis may contribute to obesity through cytokine-driven metabolic alterations in insulin signaling and adipocyte function. The psoriatic immune response is dominated by T helper (Th) 1, Th17, and Th22 cell activation, leading to elevated levels of interferon-γ, tumor necrosis factor (TNF) α, interleukin (IL) 6, IL-17, and IL-22 from keratinocytes.4 These cytokines contribute not only to cutaneous inflammation but also to insulin resistance and adipocyte dysfunction.5 Tumor necrosis factor α and IL-6 interfere with insulin signaling via activation of stress kinases (eg, IκB kinase and c-Jun N-terminal kinase), implicating these molecules in insulin resistance and weight gain.6 Moreover, IL-17, a central cytokine in psoriasis, has been implicated in vascular inflammation, insulin resistance, and type 2 diabetes, suggesting a mechanistic link between psoriatic inflammation and metabolic disease.5 Additionally, chronic systemic inflammation in psoriasis suppresses adiponectin, a protective adipokine that enhances insulin sensitivity and exerts anti-inflammatory effects by inhibiting TNF-α and IL-6 production while promoting IL-10 synthesis. Reduced adiponectin levels have consistently been observed in patients with psoriasis and concomitant obesity or metabolic syndrome.3 The resultant imbalance between proinflammatory and anti-inflammatory mediators creates a metabolic environment conducive to obesity.3
Conversely, obesity itself may intensify both the incidence and severity of psoriasis through shared inflammatory pathways. Leptin, whose expression rises proportionally with adipocyte mass, acts as a proinflammatory mediator linking obesity to psoriasis exacerbation. By promoting Th1 and Th17 cell differentiation and suppressing regulatory T-cell activity, leptin increases IL-17A, IL-6, and TNF-α production.7 These cytokines stimulate keratinocyte proliferation and perpetuate cutaneous inflammation, thereby intensifying disease activity. Similarly, resistin, another adipokine that is elevated in obesity, stimulates monocytes and macrophages to secrete TNF-α and IL-6, creating a proinflammatory state in the body that drives the relationship between excessive fat storage (adiposity) and the development and severity of psoriasis.8 In contrast, reduced adiponectin levels in obesity remove a key anti-inflammatory regulator that normally inhibits TNF-α and IL-6 synthesis and promotes IL-10 production. This deficiency provokes unrestrained cytokine activation within both adipose and cutaneous tissue, exacerbating psoriatic immune dysregulation.9
Free fatty acids derived from abundant adipocytes in obesity further activate nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB)–signaling and induce oxidative stress, augmenting systemic inflammation. Adipose tissue macrophages additionally secrete IL-1Β, IL-6, and TNF-α, which promote keratinocyte proliferation and amplify the psoriatic inflammatory cascade. Importantly, anti–TNF-α therapy has been shown to improve metabolic parameters, reducing low-density lipoprotein and total cholesterol levels and enhancing insulin sensitivity, highlighting the reciprocal benefits of dampening the inflammatory signaling from TNF-α.5
Together, these findings highlight that psoriasis and obesity are interconnected inflammatory disorders driven by overlapping cytokine networks, most notably TNF-α, IL-6, IL-17, and IL-23, and by imbalances in adipokines such as leptin, resistin, and adiponectin (Table).

Clinical Implications of Obesity-Related Comorbidities
Higher body mass index (BMI) has been associated with an increased and earlier incidence of psoriasis. When treated as continuous variables, both Psoriasis Area and Severity Index (PASI) and Dermatology Life Quality Index scores were positively correlated with increases in BMI.10,11 This close association suggests BMI may function as a practical indicator of disease severity and prognosis.7,8,11
Psoriatic arthritis (PsA) is a major comorbidity of psoriasis that impacts disease severity and quality of life. Obesity is associated with an increased risk for PsA after accounting for traditional risk factors. Psoriatic arthritis can cause chronic mobility issues and complicate a patient’s ability to stay active. Early rheumatologist involvement may be warranted to prevent PsA development in patients with obesity and psoriasis.11,12
Patients with psoriasis have an increased baseline risk for metabolic syndrome, including obesity, type 2 diabetes, hypertension, and dyslipidemia11,13,14; therefore, the presence of obesity warrants regular metabolic evaluation. Unmanaged metabolic syndrome contributes to the increased prevalence of myocardial infarction, stroke, and heart failure seen with psoriasis.13,14 Obesity also may cause obstructive sleep apnea, which can exacerbate hypertension and heart disease due to chronic hypoxia.11 The interplay of these metabolic factors puts patients with psoriasis and obesity at heightened cardiovascular risk.13,14
Both psoriasis and obesity present psychosocial challenges such as elevated rates of depression, anxiety, and body-image concerns, all of which become more pronounced when these conditions coexist. These psychological stressors may hinder a patient’s motivation for lifestyle changes or treatment adherence.13
Dermatology visits serve as an important opportunity to monitor obesity-related comorbidities.13,15 Dermatologists are uniquely positioned to initiate metabolic screening while collaborating with primary care physicians for ongoing cardiometabolic management. Metabolic and cardiovascular baselines should be measured when care is established and should be followed longitudinally—these include BMI, waist circumference measurements, blood pressure, lipid panels, fasting glucose or A1C levels, and liver enzymes.10,14,15 Regular screening for depression, suicidality, and disordered eating also is encouraged.13 Clinicians should follow established guidelines to identify and manage metabolic, cardiovascular, and psychological comorbidities.13
Impact of Obesity on Psoriasis Treatment
Obesity is a critical factor in clinical decision-making, as it consistently is associated with diminished response to numerous systemic psoriasis therapies. This reduced efficacy has been observed with conventional oral agents such as methotrexate and cyclosporine and is particularly well documented in the context of biologic therapies.15,16 Several meta-analyses and large real-world studies have shown that higher BMI is associated with a suboptimal treatment response, with patients in higher BMI categories achieving lower rates of PASI 75 and PASI 90 than their nonobese counterparts receiving the same regimen.15,17
This efficacy gap often is rooted in pharmacokinetic challenges. For many biologics administered via subcutaneous injection, increased BMI can lead to altered drug distribution, such as sequestration in adipose tissue. Altered distribution combined with potentially increased drug clearance can result in lower overall serum drug concentrations and subsequent underdosing for a patient’s inflammatory burden.15 This reality highlights the important distinction between fixed and weight-based dosing strategies. Therapies dosed by weight, such as infliximab, have demonstrated more consistent efficacy in populations with obesity, as the dose is escalated to match patient size.18 Despite the weight-dependent effect, recent real-world studies have suggested that fixed dosing of some IL-17A inhibitors (eg, ixekizumab) remains highly effective across BMI categories, while others (eg, secukinumab) show diminished efficacy in obese patients.16,19 Furthermore, some real-world studies have reported an inverse relationship between elevated BMI and efficacy of IL-23 inhibitors, particularly guselkumab, while other studies reported no association.20,21 These mixed observations support a nuanced interpretation of BMI’s role in treatment modification; consideration should be given to the specific medication and the dosing strategy over biologic class alone.
Whether obesity independently drives psoriasis severity or mainly diminishes treatment efficacy through pharmacokinetic effects remains debated. Observational studies show a dose-dependent relationship between BMI and disease severity even in untreated patients, supporting a proinflammatory role for obesity.10 Concurrently, higher BMI may predict lower responses to some fixed-dose biologics, likely due to altered distribution and sequestration in adipose tissue.15,20 The precise contributions are challenging to delineate; both likely converge to heighten disease severity and reduce treatment response.
Beyond efficacy, obesity compounds the safety considerations of systemic treatments. Psoriasis and obesity are both strong independent risk factors for metabolic dysfunction–associated steatotic liver disease. This prevalent comorbidity creates a substantial clinical dilemma, as a first-line, cost-effective agent such as methotrexate carries a known risk for hepatotoxicity, which is amplified in patients with pre-existing liver steatosis.13 Consequently, the presence of obesity and metabolic dysfunction–associated steatotic liver disease often limits the use of methotrexate, forcing a change in therapy. Thorough baseline comorbidity screening in all patients with psoriasis and obesity is necessary to select a therapy that balances efficacy with safety.13,14
Management Strategies
Weight management plays a pivotal role in improving psoriasis outcomes. Even modest weight loss of 5% to 10% has been shown in randomized and observational studies to substantially reduce disease severity, reflected by lower PASI and Dermatology Life Quality Index scores and enhance treatment responsiveness.22,23 Dietary approaches emphasizing hypocaloric, Mediterranean, or anti-inflammatory patterns have demonstrated additional improvements in disease activity, likely through attenuation of systemic inflammation and metabolic dysregulation.21 Bariatric surgery provides more sustained benefits, with multiple studies reporting long-term remission or reduced psoriasis severity following substantial postoperative weight loss.23,24
Pharmacologic weight-loss therapies, particularly glucagonlike peptide-1 receptor agonists, have emerged as potential adjuncts in psoriasis management. Although data remain limited, these agents may reduce systemic inflammation, improve insulin sensitivity, and indirectly enhance biologic response.25
Conclusion
Psoriasis and obesity are interconnected chronic inflammatory conditions that share overlapping cytokine pathways and mutually exacerbate the clinical course. Systemic inflammation driven by cytokines such as TNF-α, IL-6, and IL-17 not only promotes psoriatic skin disease but also contributes to metabolic dysfunction and cardiovascular risk. In turn, excess adiposity amplifies inflammatory signaling and diminishes therapeutic response, creating a self-perpetuating cycle of disease.
Dermatologists should identify obesity-related risks early; counsel patients on lifestyle changes; initiate metabolic screening; and coordinate care across primary care, nutrition, and rheumatology. Regular screening for metabolic syndrome, cardiovascular comorbidities, and psychosocial distress should be integrated into psoriasis management. Future research should focus on personalized treatment strategies that integrate management of inflammatory skin disease with underlying metabolic health, such as optimizing biologic dosing and identifying novel targets that disrupt the pathophysiologic loop. By recognizing and addressing the shared inflammatory mechanisms of psoriasis and obesity, clinicians can improve both dermatologic and systemic outcomes for affected patients.
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
- Barrea L, Muscogiuri G, Annunziata G, et al. Update on obesity in psoriasis patients: narrative review and practical insights. Clin Cosmet Investig Dermatol. 2023;16:3089-3104.
- Owczarczyk-Saczonek A, Gornowicz-Porowska J, Zegarska B. Psoriasis comorbidities: obesity, diet, and metabolic syndrome. Int J Mol Sci. 2024;25:1832.
- Vata D, Tarcau BM, Popescu IA, et al. Update on obesity in psoriasis patients. Life (Basel). 2023;13:1947.
- Piaserico S, Orlando G, Messina F. Psoriasis and cardiometabolic diseases: shared genetic and molecular pathways. Int J Mol Sci. 2022;23:9063.
- Hao Y, Zhu YJ, Zou S, et al. Metabolic syndrome and psoriasis: mechanisms and future directions. Front Immunol. 2021;12:711060.
- Kern L, Mittenbühler MJ, Vesting AJ, et al. Obesity-induced TNF-α and IL-6 signaling: the missing link between obesity and inflammation-driven liver and colorectal cancers. Cancers (Basel). 2019;11:24.
- Hwang J, Yoo JA, Yoon H, et al. Role of leptin in the association between obesity and psoriasis. Biomol Ther (Seoul). 2021;29:11-21.
- Smith B, Devjani S, Collier MR, et al. Association between psoriasis and obesity among US adults in the 2009-2014 National Health and Nutrition Examination Survey. Cutis. 2023;112:49-51. doi:10.12788/cutis.0807
- Ellulu MS, Patimah I, Khaza’ai H. Obesity and inflammation: the linking mechanism and the complications. Arch Med Sci. 2017;13:851-863.
- Wang H, Hou S, Kang X, et al. BMI matters: understanding the link between weight and severe psoriasis. Sci Rep. 2025;15:11158.
- Norden A, Rekhtman S, Strunk A, et al. Risk of psoriasis according to body mass index: a retrospective cohort analysis. J Am Acad Dermatol. 2022;86:1020-1026.
- Di Caprio R, Nigro E, Di Brizzi EV, et al. Exploring the link between psoriasis and adipose tissue: one amplifies the other. Int J Mol Sci. 2024;25:13435.
- Elmets CA, Leonardi CL, Davis DMR, et al. Joint AAD-NPF guidelines of care for the management and treatment of psoriasis with awareness and attention to comorbidities. J Am Acad Dermatol. 2019;80:1073-1113.
- Secchiero P, Rimondi E, Marcuzzi A, et al. Metabolic syndrome and psoriasis: pivotal roles of chronic inflammation and gut microbiota. Int J Mol Sci. 2024;25:8098.
- Burshtein J, Armstrong A, Chow M, et al. Association between obesity and efficacy of psoriasis therapies: an expert consensus panel. J Am Acad Dermatol. 2025;92:807-815. doi:10.1016/j.jaad.2024.12.016
- Pirro F, Caldarola G, Chiricozzi A, et al. Impact of body mass index on the efficacy of biological therapies in patients with psoriasis: a real-world study. Clin Drug Investig. 2021;41:917-925.
- Hjort G, Schwarz CW, Skov L, et al. Clinical characteristics associated with response to biologics in the treatment of psoriasis: a meta-analysis. JAMA Dermatol. 2024;160:830-837.
- Naldi L, Chimenti S, Girolomoni G, et al. Efficacy and safety of infliximab in obese and non-obese patients with plaque-type psoriasis: subanalysis of the EXPRESS II trial. Br J Dermatol. 2008;159:761-766.
- Puig L, Thom H, Mollon P, et al. Effect of body weight on the efficacy of biologics in moderate-to-severe plaque psoriasis: a systematic review and meta-analysis. J Eur Acad Dermatol Venereol. 2020;34:237-245.
- Dai M, Jiang Y, Wang Y, et al. Differential clinical factors influencing the effectiveness of distinct biologic agents in psoriasis: insights from a prospective cohort study in China. Inflamm Res. 2026;75:25. doi:10.1007/s00011-025-02179-1
- Ricceri F, Chiricozzi A, Peris K, et al. Successful use of anti–IL-23 molecules in overweight-to-obese psoriatic patients: a multicentric retrospective study. Dermatol Ther. 2022;35:E15793. doi:10.1111/dth.15793
- Jensen P, Zachariae C, Christensen R, et al. Effect of weight loss on the severity of psoriasis: a randomized clinical study. Br J Dermatol. 2013;168:319-327.
- Hossler EW, Wood GC, Still CD, et al. Psoriasis improvement following bariatric surgery is durable: 5-year follow-up in the Geisinger bariatric surgery cohort. Obes Surg. 2020;30:3350-3356.
- Romero-Talamás H, Daigle CR, Aminian A, et al. Psoriasis improvement after bariatric surgery. Surg Obes Relat Dis. 2014;10:1155-1159.
- Buonanno S, Gaggiano C, Terribili R, et al. Potential role of GLP-1 receptor agonists in the management of psoriatic disease: a scoping review. Inflamm Res. 2025;74:167. doi:10.1007/s00011-025-02140-2
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Psoriasis and Obesity: A Clinical Review of the Bidirectional Link and Management Implications
Practice Points
- Obesity is an independent risk factor for psoriasis onset and severity. Both conditions share overlapping inflammatory pathways that create a self-perpetuating cycle of metabolic and cutaneous dysfunction.
- Dermatologists play a key role in early detection of comorbidities, and patients with psoriasis and obesity should undergo regular screening for metabolic syndrome, liver disease, and psoriatic arthritis.
- Weight loss is a critical therapeutic intervention that may improve Psoriasis Area and Severity Index scores and restore therapeutic responsiveness.
Black Dots on the Scalp of a Child
Black Dots on the Scalp of a Child
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).
Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).


Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
THE DIAGNOSIS: Terra Firma-Forme Dermatosis
During clinical examination, a 70% alcohol swab was utilized to gently rub several of the lesions, which were successfully removed. This confirmed a diagnosis of terra firma-forme dermatosis (TFFD)(also known as Duncan’s dirty dermatosis). The patient’s mother was counseled about the diagnosis and was instructed on how to use alcohol pads to remove the remaining lesions. Three days later, after several treatment sessions at home, the mother reported complete resolution of the lesions with no residual pigmentary changes, ulceration, or scarring (Figures 1 and 2).


Terra firma-forme dermatosis was first described in 1987 in a 12-year-old girl with hyperpigmented plaques on the neck that cleared when rubbing alcohol was applied before biopsy.1,2 The term terra firma is Latin for “firm land” (or essentially “dirt”) in reference to what often is described as a characteristically “dirty” clinical appearance.2 Terra firmaforme dermatosis can manifest anywhere on the body but shows a predilection for the neck, arms and legs, axillae, inguinal region, and umbilicus.3 Lesions typically are described as asymptomatic, smooth, well-circumscribed, reticular papules or patches that are brown or black. Terra firma-forme dermatosis also may demonstrate secondary features such as hyperkeratotic, scaly, velvety, or verrucous plaques and nodules.3
The etiology of this condition is theorized to be a result of abnormal or delayed keratinization and prolonged keratinocyte adhesion.3,4 There are limited epidemiologic data, but TFFD has shown a predominance in children younger than 18 years (average age of onset, 10 years) with no known predilection for sex or race and no recognized pattern of inheritance.3-5
Histopathology typically demonstrates epidermal atrophy, hyperkeratosis, and often a component of trapping and compaction of melanin, sebum, microorganisms, and environmental debris.5
Management of TFFD is straightforward and generally consists of rubbing with 70% isopropyl alcohol to remove the lesions. For more adherent lesions or for extensive involvement, other keratolytics such as salicylic acid or alpha-hydroxy acids may be used.5 For TFFD manifesting in infants and young children, widespread involvement, or lesions involving the face or genitals, a urea-based keratolytic with or without a topical anti-inflammatory is suggested.5 Other treatment options include other alpha-hydroxy acids, topical retinoids, and nonpolar solvents such as acetone or CO2 laser for recalcitrant cases.4,5 Fortunately, most TFFD lesions respond well to conservative therapies, with recurrence reported only in 6.3% (5/79) of patients in one study.3
Dermatosis neglecta is clinically similar to TFFD and often is considered on the same spectrum of disease6; however, this entity is associated with decreased bathing or limited hygiene, which could be related to child or elder abuse/neglect or comorbid psychiatric disorders. These conditions can be distinguished by attempting to remove the lesions using soap and water; lesions of dermatosis neglecta will clear, whereas those of TFFD will not.
Metastatic melanoma in pediatric patients has a polymorphous appearance and may or may not be pigmented. Lesions often may be associated with lymphadenopathy of the draining lymph node basins, and nodules and lesions may be firm on palpation.7 Linear configurations of metastatic melanoma may represent a satellite or in-transit metastasis. Fortunately, melanoma is extraordinarily rare in children, with an estimated incidence of 2.1 per million for individuals younger than 20 years.8
Acanthosis nigricans is characterized by velvety plaques most commonly affecting the posterior neck, axillae, and flexor extremities. These lesions commonly are associated with obesity and insulin resistance but occasionally can be associated with underlying malignancy. In the latter association, acanthosis nigricans lesions tend to manifest more abruptly, often are pruritic, and can involve the mucous membranes. Fortunately, acanthosis nigricans related to malignancy in the pediatric population is rare.9
Epidermal nevi may exhibit clinical similarities to TFFD, particularly in lesions with brown/black pigment or with a reticulated or verrucous appearance; however, epidermal nevi often are congenital or manifest within the first few years of life. They commonly are distributed over the lines of Blaschko and have a linear appearance; they also enlarge and thicken as the patient ages.10
Black-dot tinea capitis, a classic manifestation of endothrix infection, manifests as alopecia with broken hairs and is most commonly caused by Tinea tonsurans.11 The black dots refer to the appearance of the infected hair shafts, which have been weakened and broken off at the follicular ostia. As such, lesions typically are monomorphic and may be interspersed with uninvolved hair shafts. There often is associated scale and a lack of inflammation.11,12
Additional differential diagnoses to consider include seborrheic keratoses and confluent and reticulated papillomatosis. Further workup (eg, potassium hydroxide preparation of skin scrapings or skin biopsy) may help elucidate the diagnosis.5 A simple and cost-effective initial diagnostic tool involves wiping suspicious lesions with a 70% isopropyl alcohol pad to confirm this diagnosis.
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
- Duncan WC. Terra firma-forme dermatosis. Arch Dermatol. 1987;123:567. doi:10.1001/archderm.1987.01660290031009
- Greywal T, Cohen PR. Terra firma-forme dermatosis: a report of ten individuals with Duncan’s dirty dermatosis and literature review. Dermatol Pract Concept. 2015:29-33. doi:10.5826/dpc.0503a08
- Aslan NÇ, Güler S, Demirci K, et al. Features of terra firma-forme dermatosis. Ann Fam Med. 2018;16:52-54. doi:10.1370/afm.2175
- Sechi A, Patrizi A, Savoia F, et al. Terra firma-forme dermatosis. Clin Dermatol. 2021;39:202-205. doi:10.1016/j.clindermatol.2020.10.019
- Mohta A, Sarkar R, Narayan RV, et al. Terra firma-forme dermatosis—more than just dirty. Indian Dermatol Online J. 2024;15:99-104. doi:10.4103/idoj.idoj_424_23
- Erkek E, Çetin E, Sahin S, et al. Terra firma-forme dermatosis. Indian J Dermatol Venereol Leprol. 2012;78:358. doi:10.4103 /0378-6323.95455
- McMullan P, Grant-Kels JM. Childhood and adolescent melanoma: an update. Clin Dermatol. 2025;43:16-23. doi:10.1016 /j.clindermatol.2025.01.010
- NCCR*Explorer: An interactive website for NCCR cancer statistics. National Cancer Institute website. Accessed January 10, 2025. https://nccrexplorer.ccdi.cancer.gov/data-products.html
- Sinha S, Schwartz RA. Juvenile acanthosis nigricans. J Am Acad Dermatol. 2007;57:502-508. doi:10.1016/j.jaad.2006.08.016
- Waldman AR, Garzon MC, Morel KD. Epidermal nevi: what is new. Dermatol Clin. 2022;40:61-71. doi:10.1016/j.det.2021.09.006
- Wang X. Black dot tinea capitis. N Engl J Med. 2024; 391:E7. doi:10.1056/NEJMicm2401964
- Gupta AK, Summerbell RC. Tinea capitis. Med Mycol. 2000; 38:255-287. doi:10.1080/mmy.38.4.255.287
Black Dots on the Scalp of a Child
Black Dots on the Scalp of a Child
A 4-year-old boy was referred to the dermatology clinic by his pediatrician for evaluation of persistent black spots on the scalp of 1 month’s duration. The patient was otherwise healthy, and his mother stated that the lesions had appeared gradually, were not tender or pruritic, and did not wash off with shampoo and scrubbing. The patient had no history of any systemic illness, recent travel, genetic disorders, or genodermatoses. Physical examination revealed multiple well-circumscribed, 1- to 2-mm black papules and macules with confluence scattered over the vertex scalp. No erythema, scale, or induration was noted.
Assessing Inpatient Dermatology Availability in Virginia
Assessing Inpatient Dermatology Availability in Virginia
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
To the Editor:
It is known that dermatologist evaluation of skin conditions in hospitalized patients confers enhanced diagnostic accuracy, timely and appropriate treatment, and an overall reduction in readmissions compared to assessments by nondermatology hospitalists.1 Dermatology consultations have been shown to alter diagnoses in up to 50% of cases and lead to changes in management in nearly 75% of cases, even for prevalent dermatologic conditions such as drug rashes, cellulitis, and stasis dermatitis.1,2 Previous studies have observed a multiday reduction in length of hospital stay, a 10-fold reduction in readmission rate, and lower 30-day mortality, all leading to a reduction in patient morbidity and costs to both the patient and the health care system.3,4 Despite these benefits, there has been a decrease in the number of dermatologists providing inpatient services and a reduction in medical centers offering dermatology consultations over the past several years.5 To better appreciate current trends of declining dermatology inpatient and consultative services within our region, we evaluated the availability of dermatology care at hospitals across Virginia.
A simple telephone survey was conducted across community hospitals in Virginia wherein medical staff administrators were asked to provide details regarding their dermatology staffing. The following figures were collected: number of dermatologists on staff, number of dermatologists with consulting privileges, number of affiliated dermatologists, and number of advanced-practice dermatology providers. Follow-up calls were carried out to elaborate on how dermatologists (when available) were integrated into inpatient care workflow and made accessible to hospitalists and emergency medicine departments. Academic centers, military hospitals, and specialty hospitals were excluded from the survey.
To better appreciate the relationships between hospital and population characteristics and the availability of dermatology care, publicly available data were collected on hospital bed counts and regional population density for each facility.6-9 Spearman rank correlation analyses were conducted in Microsoft Excel to evaluate the association between the number of dermatologists on staff, number of consulting dermatologists, staffed inpatient beds, and population size.
Sixty-four hospitals—more than 70% of the 90 eligible community hospitals—responded to the survey between May and August 2024 and were included in the study. On-staff dermatologists were present at 8 (12.5%) of the hospitals surveyed; of these, 4 (50.0%) hospitals had between 1 and 5 dermatologists, 3 (37.5%) had between 6 and 10 dermatologists, and 1 (12.5%) had between 11 and 15 dermatologists. An additional 4 (6.3%) hospitals provided consultative dermatology services from outside dermatology clinics. Urban hospitals accounted for 9 of 12 (75%) hospitals offering in-house dermatology services, either through on-staff physicians or consultations with clinic-based providers.
Based on Spearman rank correlation analysis, there was a positive correlation between the number of dermatologists on staff and the number of staffed hospital beds (r=0.61; P <.001). Similarly, there was a positive correlation between the number of dermatologists on staff and the population density of the affiliated region (r=0.58; P <.001). Finally, there was a positive correlation between the number of dermatologists on staff and the number of available consulting dermatologists (r=0.89; P <.001).
At facilities with only consultative dermatology services accessible, there often was no formal dermatology team or department present. Rather, the hospitals relied on a loosely affiliated network of dermatology providers or navigated inpatient dermatology needs almost exclusively via internal medicine hospitalists or emergency medicine physicians. When available, dermatology support from dermatology physicians often was provided through teledermatology platforms. Although teledermatology has a large role in increasing access to care within underserved areas, its reliance on images and second-hand case descriptions can limit the provider’s ability to perform a comprehensive examination and assessment. Moreover, it was noted that few hospital representatives could offer clarity on how dermatologists were integrated into the inpatient setting. It remained unclear whether dermatologists were practically accessible to the inpatient care teams in a structured manner.
The uneven distribution and limited availability of dermatology inpatient care in Virginia reflect national trends and underscore ongoing access issues for patients. Without intentional intervention, these trends are expected to continue, contributing to a glaring gap in hospital services as well as to patient morbidity and mortality. The correlation data obtained in our study further qualify these disparities. The positive correlations between dermatologist availability and hospital size and population density suggest that larger, more urban facilities are more likely to offer inpatient dermatology care, whether through staffing or consultation. This relationship is not unexpected, given the greater financial resources and specialist networks available to facilities with large patient volumes. This suggests that dermatology care is shaped by institutional capacity and geographic leverage rather than clinical need, reinforcing existing disparities.
Importantly, it should be noted that the data may overestimate the true availability of dermatologists to these patients. As revealed via follow-up survey calls, respondent facilities that provided dermatology via consultative services often did not have a defined structure for integrating this care into the inpatient workflow. In some instances, dermatologists were technically affiliated with the hospital but had varying levels of practical interaction with the hospital providers and their patients. Administrative staff's differing awareness regarding dermatology interaction with the hospital facility may reveal systemic underutilization and opportunities to improve coordination to achieve the greatest benefit from dermatology services. These observations are further informed by the scope of our study, which focused specifically on community hospitals. The exclusion of academic and military institutions—and the tendency of these to exist in more densely populated areas—may have limited how broadly our findings reflect nationwide dermatology access by omitting more established dermatology departments and specialty care. As a result, regional variations in predominant facility type should be considered when interpreting the implications of these results beyond Virginia’s community hospital system.
In response to access limitations and differences in availability, facilities are turning to integrated teledermatology as a valuable tool to expand the reach of specialist care, particularly in rural or resource-limited settings. This modality acts as an important step toward improving equity in care by beginning to bridge geographic gaps; however, along with these logistical advantages, teledermatology also confers diagnostic limitations and clinical trade-offs that should be thoughtfully considered. Our findings highlight the need to expand access in a way that integrates technological advances with in-person care to build a sustainable and effective path forward without compromising the quality of care patients receive. We present these outcomes to emphasize the importance of increasing dermatology involvement in the care of hospitalized patients, which is a promising strategy to improve patient outcomes and reduce existing disparities in Virginia and nationwide.
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
- Hu L, Haynes H, Ferrazza D, et al. Impact of specialist consultations on inpatient admissions for dermatology-specific and related DRGs. J Gen Intern Med. 2013;28:1477-1482. doi:10.1007s11606-013-2440-2
- Madigan LM, Fox LP. Where are we now with inpatient consultative dermatology?: Assessing the value and evolution of this subspecialty over the past decade. J Am Acad Dermatol. 2019;80:1804-1808. doi:10.1016/j.jaad.2019.01.031
- Milani-Nejad N, Zhang M, Kaffenberger BH. Association of dermatology consultations with patient care outcomes in hospitalized patients with inflammatory skin diseases. JAMA Dermatol. 2017;153:523-528. doi:10.1001/jamadermatol.2016.6130
- Puri P, Pollock BD, Yousif M, et al. Association of society of dermatology hospitalist institutions with improved outcomes in Medicare beneficiaries hospitalized for skin disease. J Am Acad Dermatol. 2023;88:1372-1375. doi:10.1016/j.jaad.2023.01.021
- Hydol-Smith JA, Gallardo MA, Korman A, et al. The United States dermatology inpatient workforce between 2013 and 2019: a Medicare analysis reveals contraction of the workforce and vast access deserts—a cross-sectional analysis. Arch Dermatol Res. 2024;316:103. doi:10.1007/s00403-024-02845-0
- QuickFacts: Virginia. 2024. Census Bureau QuickFacts. https://www.census.gov/quickfacts/fact/table/VA/PST045224
- American Hospital Directory. Individual hospital statistics for Virginia. Updated May 7, 2023. Accessed November 12, 2025. https://www.ahd.com/states/hospital_VA.html
- Virginia Office of Data Governance and Analytics. Definitive healthcare: USA hospital beds (CSV). Virginia Open Data Portal. Accessed November 12, 2025. https://data.virginia.gov/dataset/definitive-healthcare-usa-hospital-beds/resource/c39226d7-1b28-4ce0-8f35-3a0ff974eba5
- Virginia Health Information. Virginia hospitals. Updated February 26, 2021. Accessed November 12, 2025. https://www.vhi.org/Hospitals/vahospitals.asp
Assessing Inpatient Dermatology Availability in Virginia
Assessing Inpatient Dermatology Availability in Virginia
Progressive Erythematous Facial Rash
Progressive Erythematous Facial Rash
THE DIAGNOSIS: Follicular Mucinosis
Histologic examination of the hematoxylin and eosin–stained sections of the biopsy revealed an overall moderately dense, perivascular, and perifollicular lymphocytic infiltrate with follicular intraepidermal mucin (Figure). Immunohistochemical staining showed that the lymphocytic infiltrate was predominantly CD4+ over CD8+, with moderate loss of CD7 and absence of CD20 expression. Positive T-cell receptor (TCR) gene rearrangements were detected for both TCRγ and TCRΒ. The clinical features along with the histopathologic findings suggested a diagnosis of follicular mucinosis (FM) with concern in the differential for folliculotropic mycosis fungoides.
Follicular mucinosis, also known as alopecia mucinosa, is an uncommon inflammatory disorder characterized by follicular degeneration due to the accumulation of mucin within the pilosebaceous unit.1 This condition manifests clinically as indurated plaques and/or follicular papules most often on the face, neck, and scalp.2 It is further categorized as primary vs secondary FM. Primary idiopathic FM, which can further be subdivided into acute or chronic, tends to follow a more benign course, whereas secondary FM usually is associated with underlying inflammatory or neoplastic conditions, most commonly mycosis fungoides, a cutaneous T-cell lymphoma.1,2 In cases of secondary FM, treatment of the underlying cause often leads to resolution of symptoms. Regular follow-up is warranted in either classification.1,3
The initial differential diagnosis for this patient included contact dermatitis associated with mask use, with possible underlying seborrheic dermatitis or rosacea; however, the rash persisted and worsened after treatment with topical triamcinolone and ketoconazole. After the diagnosis of FM was made, the patient was started on topical betamethasone and tacrolimus with good response.
A referral to hematology/oncology revealed that the patient had primary FM and possible stage 1A folliculotropic mycosis fungoides with limited skin involvement (<10% body surface area). On physical examination, no palpable cervical or axillary lymphadenopathy were noted. Flow cytometry for lymphoma was negative with no lymphoid or blast population detected. Laboratory workup and positron emission tomography/computed tomography were unremarkable. The patient had rapid improvement with a more potent topical steroid but also was given tacrolimus ointment 0.1% for residual findings. His disease remained stable without progression at 1-year follow-up.
Contact dermatitis typically manifests as an eczematous eruption that appears on an anatomic location that was exposed to or came into contact with allergens or irritants.4 Contact dermatitis was less likely in our patient due to the lack of acute or subacute spongiosis and lymphocyte exocytosis. Rosacea is a chronic inflammatory dermatosis that presents as recurrent episodes of flushing or transient erythema, persistent erythema, phyphymatous changes, papules, pustules, and telangiectasia5; however, rosacea was less likely in our patient due to the histopathologic and immunohistochemical findings that were suggestive of FM on punch biopsy. Cutaneous lupus generally is associated with photosensitivity and manifests as erythema over the malar eminences and bridge of the nose with sparing of the nasolabial folds.6 Seborrheic dermatitis manifests as erythematous macules or patches with scale and associated pruritis on the scalp, eyebrows, eyelids, and nasolabial folds.7 This condition was less likely in our patient due to the persistence and worsening of the facial erythematous dermatitis despite the use of ketoconazole cream as well as no evidence of spongiosis, shoulder parakeratosis, vascular changes, or presence of microorganisms such as Malassezia species.
Due to the relatively rare nature of this condition as well as a wide variety of other more common etiologies for an erythematous dermatitis of the cheeks, the diagnosis of FM may be delayed or missed entirely. Physicians must have a high index of suspicion to diagnose properly and biopsy if necessary. This photoquiz serves as an important reminder to physicians to keep uncommon diseases on their differential, especially when the patient’s symptoms do not respond to treatment.
- Khalil J, Kurban M, Abbas O. Follicular mucinosis: a review. Int J Dermatol. 2021;60:159-165.
- Akinsanya AO, Tschen JA. Follicular mucinosis: a case report. Cureus. 2019;11:E4746.
- Miyagaki T. Diagnosis of early mycosis fungoides. Diagnostics (Basel). 2021;1:1721.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192.
- van Zuuren EJ, Arents BWM, van der Linden MMD, et al. Rosacea: new concepts in classification and treatment. Am J Clin Dermatol. 2021;22:457-465.
- Rothfield N, Sontheimer RD, Bernstein M. Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol. 2006;24:348-362.
- Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169.
THE DIAGNOSIS: Follicular Mucinosis
Histologic examination of the hematoxylin and eosin–stained sections of the biopsy revealed an overall moderately dense, perivascular, and perifollicular lymphocytic infiltrate with follicular intraepidermal mucin (Figure). Immunohistochemical staining showed that the lymphocytic infiltrate was predominantly CD4+ over CD8+, with moderate loss of CD7 and absence of CD20 expression. Positive T-cell receptor (TCR) gene rearrangements were detected for both TCRγ and TCRΒ. The clinical features along with the histopathologic findings suggested a diagnosis of follicular mucinosis (FM) with concern in the differential for folliculotropic mycosis fungoides.

Follicular mucinosis, also known as alopecia mucinosa, is an uncommon inflammatory disorder characterized by follicular degeneration due to the accumulation of mucin within the pilosebaceous unit.1 This condition manifests clinically as indurated plaques and/or follicular papules most often on the face, neck, and scalp.2 It is further categorized as primary vs secondary FM. Primary idiopathic FM, which can further be subdivided into acute or chronic, tends to follow a more benign course, whereas secondary FM usually is associated with underlying inflammatory or neoplastic conditions, most commonly mycosis fungoides, a cutaneous T-cell lymphoma.1,2 In cases of secondary FM, treatment of the underlying cause often leads to resolution of symptoms. Regular follow-up is warranted in either classification.1,3
The initial differential diagnosis for this patient included contact dermatitis associated with mask use, with possible underlying seborrheic dermatitis or rosacea; however, the rash persisted and worsened after treatment with topical triamcinolone and ketoconazole. After the diagnosis of FM was made, the patient was started on topical betamethasone and tacrolimus with good response.
A referral to hematology/oncology revealed that the patient had primary FM and possible stage 1A folliculotropic mycosis fungoides with limited skin involvement (<10% body surface area). On physical examination, no palpable cervical or axillary lymphadenopathy were noted. Flow cytometry for lymphoma was negative with no lymphoid or blast population detected. Laboratory workup and positron emission tomography/computed tomography were unremarkable. The patient had rapid improvement with a more potent topical steroid but also was given tacrolimus ointment 0.1% for residual findings. His disease remained stable without progression at 1-year follow-up.
Contact dermatitis typically manifests as an eczematous eruption that appears on an anatomic location that was exposed to or came into contact with allergens or irritants.4 Contact dermatitis was less likely in our patient due to the lack of acute or subacute spongiosis and lymphocyte exocytosis. Rosacea is a chronic inflammatory dermatosis that presents as recurrent episodes of flushing or transient erythema, persistent erythema, phyphymatous changes, papules, pustules, and telangiectasia5; however, rosacea was less likely in our patient due to the histopathologic and immunohistochemical findings that were suggestive of FM on punch biopsy. Cutaneous lupus generally is associated with photosensitivity and manifests as erythema over the malar eminences and bridge of the nose with sparing of the nasolabial folds.6 Seborrheic dermatitis manifests as erythematous macules or patches with scale and associated pruritis on the scalp, eyebrows, eyelids, and nasolabial folds.7 This condition was less likely in our patient due to the persistence and worsening of the facial erythematous dermatitis despite the use of ketoconazole cream as well as no evidence of spongiosis, shoulder parakeratosis, vascular changes, or presence of microorganisms such as Malassezia species.
Due to the relatively rare nature of this condition as well as a wide variety of other more common etiologies for an erythematous dermatitis of the cheeks, the diagnosis of FM may be delayed or missed entirely. Physicians must have a high index of suspicion to diagnose properly and biopsy if necessary. This photoquiz serves as an important reminder to physicians to keep uncommon diseases on their differential, especially when the patient’s symptoms do not respond to treatment.
THE DIAGNOSIS: Follicular Mucinosis
Histologic examination of the hematoxylin and eosin–stained sections of the biopsy revealed an overall moderately dense, perivascular, and perifollicular lymphocytic infiltrate with follicular intraepidermal mucin (Figure). Immunohistochemical staining showed that the lymphocytic infiltrate was predominantly CD4+ over CD8+, with moderate loss of CD7 and absence of CD20 expression. Positive T-cell receptor (TCR) gene rearrangements were detected for both TCRγ and TCRΒ. The clinical features along with the histopathologic findings suggested a diagnosis of follicular mucinosis (FM) with concern in the differential for folliculotropic mycosis fungoides.

Follicular mucinosis, also known as alopecia mucinosa, is an uncommon inflammatory disorder characterized by follicular degeneration due to the accumulation of mucin within the pilosebaceous unit.1 This condition manifests clinically as indurated plaques and/or follicular papules most often on the face, neck, and scalp.2 It is further categorized as primary vs secondary FM. Primary idiopathic FM, which can further be subdivided into acute or chronic, tends to follow a more benign course, whereas secondary FM usually is associated with underlying inflammatory or neoplastic conditions, most commonly mycosis fungoides, a cutaneous T-cell lymphoma.1,2 In cases of secondary FM, treatment of the underlying cause often leads to resolution of symptoms. Regular follow-up is warranted in either classification.1,3
The initial differential diagnosis for this patient included contact dermatitis associated with mask use, with possible underlying seborrheic dermatitis or rosacea; however, the rash persisted and worsened after treatment with topical triamcinolone and ketoconazole. After the diagnosis of FM was made, the patient was started on topical betamethasone and tacrolimus with good response.
A referral to hematology/oncology revealed that the patient had primary FM and possible stage 1A folliculotropic mycosis fungoides with limited skin involvement (<10% body surface area). On physical examination, no palpable cervical or axillary lymphadenopathy were noted. Flow cytometry for lymphoma was negative with no lymphoid or blast population detected. Laboratory workup and positron emission tomography/computed tomography were unremarkable. The patient had rapid improvement with a more potent topical steroid but also was given tacrolimus ointment 0.1% for residual findings. His disease remained stable without progression at 1-year follow-up.
Contact dermatitis typically manifests as an eczematous eruption that appears on an anatomic location that was exposed to or came into contact with allergens or irritants.4 Contact dermatitis was less likely in our patient due to the lack of acute or subacute spongiosis and lymphocyte exocytosis. Rosacea is a chronic inflammatory dermatosis that presents as recurrent episodes of flushing or transient erythema, persistent erythema, phyphymatous changes, papules, pustules, and telangiectasia5; however, rosacea was less likely in our patient due to the histopathologic and immunohistochemical findings that were suggestive of FM on punch biopsy. Cutaneous lupus generally is associated with photosensitivity and manifests as erythema over the malar eminences and bridge of the nose with sparing of the nasolabial folds.6 Seborrheic dermatitis manifests as erythematous macules or patches with scale and associated pruritis on the scalp, eyebrows, eyelids, and nasolabial folds.7 This condition was less likely in our patient due to the persistence and worsening of the facial erythematous dermatitis despite the use of ketoconazole cream as well as no evidence of spongiosis, shoulder parakeratosis, vascular changes, or presence of microorganisms such as Malassezia species.
Due to the relatively rare nature of this condition as well as a wide variety of other more common etiologies for an erythematous dermatitis of the cheeks, the diagnosis of FM may be delayed or missed entirely. Physicians must have a high index of suspicion to diagnose properly and biopsy if necessary. This photoquiz serves as an important reminder to physicians to keep uncommon diseases on their differential, especially when the patient’s symptoms do not respond to treatment.
- Khalil J, Kurban M, Abbas O. Follicular mucinosis: a review. Int J Dermatol. 2021;60:159-165.
- Akinsanya AO, Tschen JA. Follicular mucinosis: a case report. Cureus. 2019;11:E4746.
- Miyagaki T. Diagnosis of early mycosis fungoides. Diagnostics (Basel). 2021;1:1721.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192.
- van Zuuren EJ, Arents BWM, van der Linden MMD, et al. Rosacea: new concepts in classification and treatment. Am J Clin Dermatol. 2021;22:457-465.
- Rothfield N, Sontheimer RD, Bernstein M. Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol. 2006;24:348-362.
- Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169.
- Khalil J, Kurban M, Abbas O. Follicular mucinosis: a review. Int J Dermatol. 2021;60:159-165.
- Akinsanya AO, Tschen JA. Follicular mucinosis: a case report. Cureus. 2019;11:E4746.
- Miyagaki T. Diagnosis of early mycosis fungoides. Diagnostics (Basel). 2021;1:1721.
- Elmas ÖF, Akdeniz N, Atasoy M, et al. Contact dermatitis: a great imitator. Clin Dermatol. 2020;38:176-192.
- van Zuuren EJ, Arents BWM, van der Linden MMD, et al. Rosacea: new concepts in classification and treatment. Am J Clin Dermatol. 2021;22:457-465.
- Rothfield N, Sontheimer RD, Bernstein M. Lupus erythematosus: systemic and cutaneous manifestations. Clin Dermatol. 2006;24:348-362.
- Borda LJ, Perper M, Keri JE. Treatment of seborrheic dermatitis: a comprehensive review. J Dermatolog Treat. 2019;30:158-169.
Progressive Erythematous Facial Rash
Progressive Erythematous Facial Rash
A 32-year-old man presented to the dermatology clinic for evaluation of a progressive erythematous facial rash of 4 years’ duration. The patient reported some worsening with increased face mask wear during the COVID-19 pandemic. On occasion, fluid could be expressed when the area on the right cheek was compressed. Physical examination revealed a well-demarcated erythematous plaque on the right cheek. The patient also reported intermittent mild involvement of the nose and left cheek. He initially was treated with triamcinolone and ketoconazole cream for several months, but the rash persisted. Given the chronicity and worsening of the eruption, a punch biopsy from the right cheek with immunohistochemical staining was obtained.
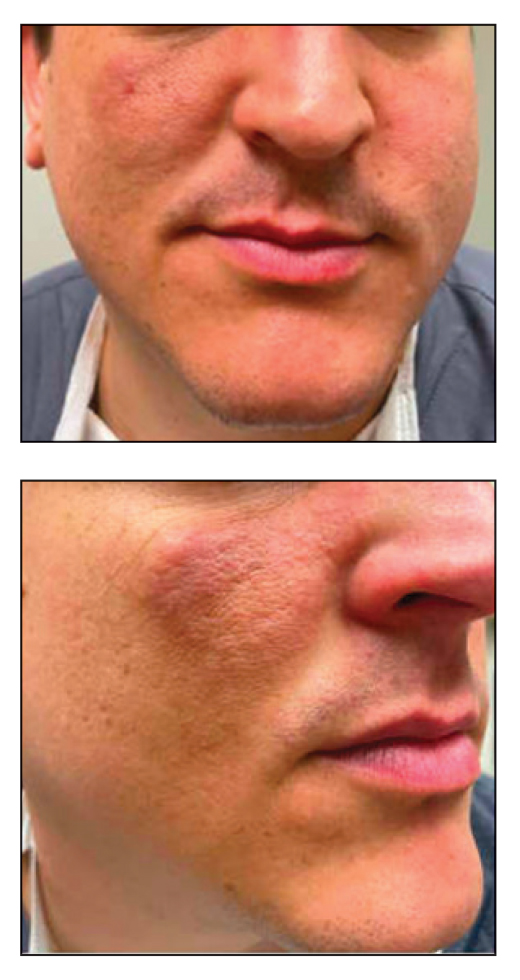
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
To the Editor:
A 22-year-old man presented to our clinic with a history of longstanding widespread recalcitrant atopic dermatitis (AD) since early childhood. He had been treated by an outside physician with topical steroids and nonsteroidal medications without notable improvement as well as with dupilumab, which was discontinued due to the development of severe head and neck dermatitis. Given the severity of his AD on presentation, we initiated treatment with upadacitinib 15 mg/d, which resulted in partial improvement. The dose was increased to 30 mg/d at 3 months with further clinical improvement.
Ten months after the patient was started on upadacitinib, he presented for a follow-up evaluation and reported a new nontender nodule on the scalp. A punch biopsy revealed a dense dermal and subcutaneous lymphoid infiltrate (Figure 1) composed of many large atypical CD2+/CD5+/CD45+ T cells with partial loss of CD3 expression (Figure 2). The atypical cells demonstrated diffuse CD30+ expression (Figure 3) and a CD4:CD8 ratio of greater than 50:1 (Figures 4 and 5). He was diagnosed with anaplastic large cell lymphoma (ALCL), and the upadacitinib was discontinued. No additional therapies directed toward ALCL were initiated.
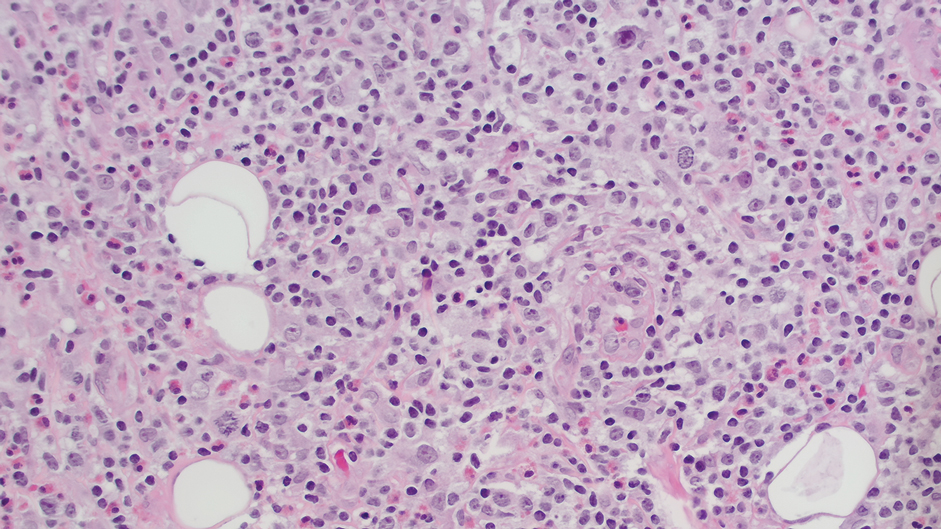
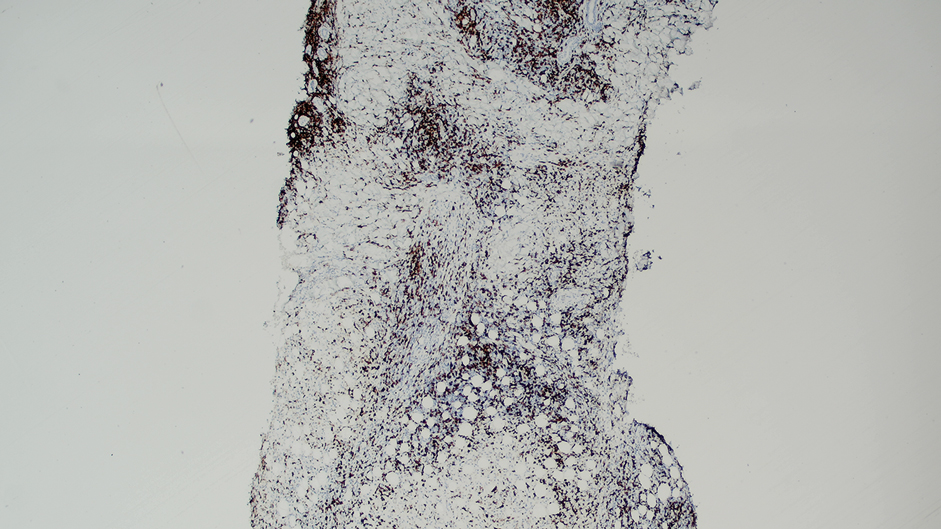
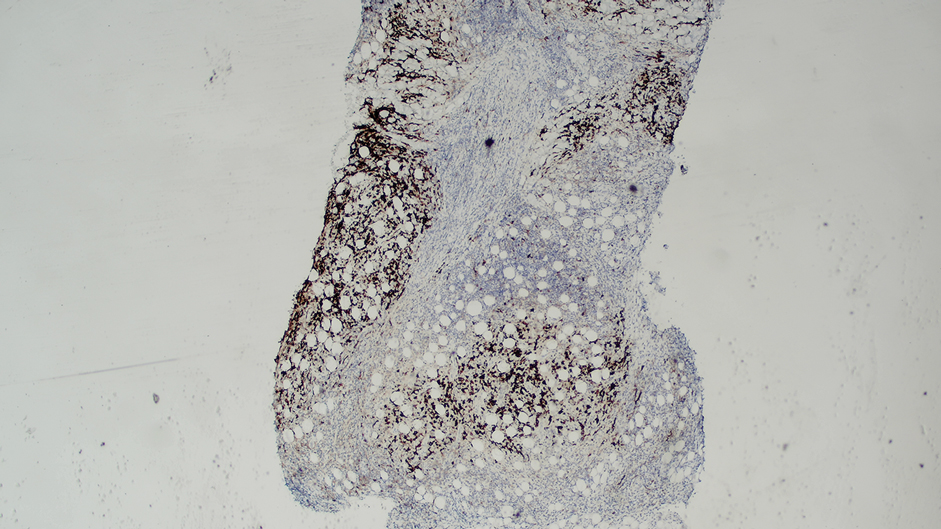
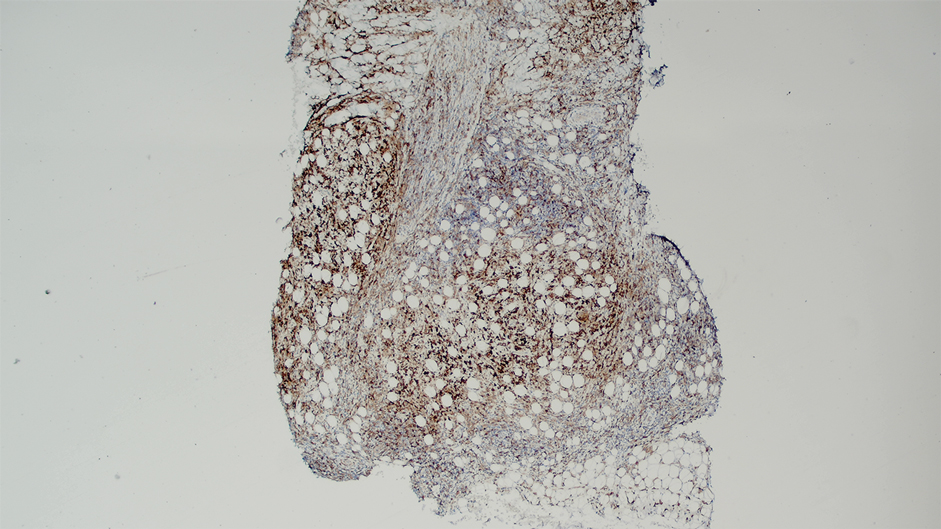
Over the next 2 weeks, the patient developed additional nodules on the postauricular skin and trunk that demonstrated similar histopathology and immunophenotype to the original scalp nodule. T-cell receptor gene rearrangement studies demonstrated shared clonal peaks in these subsequent nodules. A concurrent biopsy of an eczematous plaque on the back showed spongiotic dermatitis without evidence of cutaneous T-cell lymphoma; gene rearrangement studies from this site were negative. A positron emission tomography–computed tomography scan showed mildly hypermetabolic cervical, axillary, and inguinal lymph nodes, which were favored to be reactive. Narrow-band UVB phototherapy was initiated for management of the AD, and no additional nodules developed over the subsequent months.
Janus kinase (JAK) inhibitors are immunomodulatory small molecules that interfere with JAK–signal transducer and activator of transcription signaling involving 1 or more isoforms (eg, JAK1, JAK2, JAK3, tyrosine kinase 2) and have been used to treat various inflammatory conditions, including rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and AD.1 Upadacitinib is an oral selective JAK1 inhibitor approved by the US Food and Drug Administration for treatment of moderate to severe AD in adults and children aged 12 years and older.2 A search of PubMed using the terms upadacitinib or Rinvoq and anaplastic large cell lymphoma did not identify any cases of cutaneous ALCL arising after treatment with upadacitinib. However, a case of lymphomatoid papulosis after initiation of upadacitinib for the treatment of rheumatoid arthritis in a 74-year-old Japanese woman has been described,3 and the JAK/signal transducer and activator of transcription pathway has been implicated in the development of other CD30+ lymphoproliferative disorders.4,5
An association between JAK inhibitors and aggressive B-cell lymphomas has been described. In an observational study of 626 patients with myeloproliferative neoplasia by Porpaczy et al,6 4 of 69 (5.8%) patients treated with JAK inhibitors developed an aggressive B-cell lymphoma, whereas only 2 of 557 (0.36%) patients who did not receive JAK-inhibitor therapy developed an aggressive B-cell lymphoma. In contrast, a retrospective analysis of 2583 patients with myeloproliferative neoplasia by Pemmaraju et al7 found no significant increase in lymphoma rates in the JAK inhibitor–treated population as compared with the non-JAK inhibitor–treated group; 9 (0.56%) cases of lymphoma occurred in 1617 patients with myelofibrosis, of which 6 had exposure to JAK inhibitor therapy and 3 had no exposure to JAK inhibitor therapy (P=.082) and 5 (0.52%) cases of lymphoma occurred in 966 patients with essential thrombocythemia or polycythemia vera, none of whom had exposure to JAK inhibitor therapy.Finally, some evidence suggests the use of JAK inhibitors may be associated with an elevated risk of malignancies overall. The ORAL Surveillance study found the incidence of all cancers, excluding nonmelanoma skin cancer (NMSC), in patients treated with tofacitinib to be 4.2% (122/2911) compared with 2.9% (42/1451) in patients treated with tumor necrosis factor α inhibitors; it should be noted that the patients in this study were restricted to adults aged 50 years and older who were undergoing treatment for rheumatoid arthritis.8 In a safety profile study for upadacitinib, a higher rate of malignancies, excluding NMSC, was found in patients with AD treated with upadacitinib 30 mg/d than in patients treated with 15 mg/d; however, the overall rates of malignancies, excluding NMSC, in patients treated with upadacitinib were comparable to the standard incidence rates of malignancies in the general population derived from Surveillance, Epidemiology, and End Results data.9
In summary, we present a case of cutaneous ALCL arising after treatment with upadacitinib for AD. While some literature suggests AD may independently predispose patients to the development of CD30+ lymphoproliferative disorders, the onset of our patient’s cutaneous ALCL 10 months after initiation of upadacitinib is suggestive of an association between his lymphoproliferative disorder and JAK inhibition. Further studies are needed to better characterize the risk of lymphoproliferative disorders and other malignancies in patients treated with JAK inhibitors.
- Strangfeld A, Hierse F, Rau R, et al. Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German biologics register RABBIT. Arthritis Res Ther. 2010;12:R5. doi:10.1186/ar2904
- Rinvoq. Highlights of prescribing information. Abbvie Inc; 2024. Accessed January 31, 2026. https://www.rxabbvie.com/pdf/rinvoq_pi.pdf
- Iinuma S, Hayashi K, Noguchi A, et al. Lymphomatoid papulosis during upadacitinib treatment for rheumatoid arthritis. Eur J Dermatol. 2022;32:142-143. doi:10.1684/ejd.2022.4238
- Quesada AE, Zhang Y, Ptashkin R, et al. Next generation sequencing of breast implant-associated anaplastic large cell lymphomas reveals a novel STAT3-JAK2 fusion among other activating genetic alterations within the JAK-STAT pathway. Breast J. 2021;27:314-321. doi:10.1111/tbj.14205
- Maurus K, Appenzeller S, Roth S, et al. Recurrent oncogenic JAK and STAT alterations in cutaneous CD30-positive lymphoproliferative disorders. J Invest Dermatol. 2020;140:2023-2031.e1. doi:10.1016/j.jid.2020.02.019
- Porpaczy E, Tripolt S, Hoelbl-Kovacic A, et al. Aggressive B-cell lymphomas in patients with myelofibrosis receiving JAK1/2 inhibitor therapy. Blood. 2018;132:694-706. doi:10.1182/blood-2017-10-810739
- Pemmaraju N, Kantarjian H, Nastoupil L, et al. Characteristics of patients with myeloproliferative neoplasms with lymphoma, with or without JAK inhibitor therapy. Blood. 2019;133:2348-2351. doi:10.1182/blood-2019-01-897637
- Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386:316-326. doi:10.1056/NEJMoa2109927
- Burmester GR, Cohen SB, Winthrop KL, et al. Safety profile of upadacitinib over 15 000 patient-years across rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and atopic dermatitis. RMD Open. 2023;9:E002735. doi:10.1136/rmdopen-2022-002735
To the Editor:
A 22-year-old man presented to our clinic with a history of longstanding widespread recalcitrant atopic dermatitis (AD) since early childhood. He had been treated by an outside physician with topical steroids and nonsteroidal medications without notable improvement as well as with dupilumab, which was discontinued due to the development of severe head and neck dermatitis. Given the severity of his AD on presentation, we initiated treatment with upadacitinib 15 mg/d, which resulted in partial improvement. The dose was increased to 30 mg/d at 3 months with further clinical improvement.
Ten months after the patient was started on upadacitinib, he presented for a follow-up evaluation and reported a new nontender nodule on the scalp. A punch biopsy revealed a dense dermal and subcutaneous lymphoid infiltrate (Figure 1) composed of many large atypical CD2+/CD5+/CD45+ T cells with partial loss of CD3 expression (Figure 2). The atypical cells demonstrated diffuse CD30+ expression (Figure 3) and a CD4:CD8 ratio of greater than 50:1 (Figures 4 and 5). He was diagnosed with anaplastic large cell lymphoma (ALCL), and the upadacitinib was discontinued. No additional therapies directed toward ALCL were initiated.





Over the next 2 weeks, the patient developed additional nodules on the postauricular skin and trunk that demonstrated similar histopathology and immunophenotype to the original scalp nodule. T-cell receptor gene rearrangement studies demonstrated shared clonal peaks in these subsequent nodules. A concurrent biopsy of an eczematous plaque on the back showed spongiotic dermatitis without evidence of cutaneous T-cell lymphoma; gene rearrangement studies from this site were negative. A positron emission tomography–computed tomography scan showed mildly hypermetabolic cervical, axillary, and inguinal lymph nodes, which were favored to be reactive. Narrow-band UVB phototherapy was initiated for management of the AD, and no additional nodules developed over the subsequent months.
Janus kinase (JAK) inhibitors are immunomodulatory small molecules that interfere with JAK–signal transducer and activator of transcription signaling involving 1 or more isoforms (eg, JAK1, JAK2, JAK3, tyrosine kinase 2) and have been used to treat various inflammatory conditions, including rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and AD.1 Upadacitinib is an oral selective JAK1 inhibitor approved by the US Food and Drug Administration for treatment of moderate to severe AD in adults and children aged 12 years and older.2 A search of PubMed using the terms upadacitinib or Rinvoq and anaplastic large cell lymphoma did not identify any cases of cutaneous ALCL arising after treatment with upadacitinib. However, a case of lymphomatoid papulosis after initiation of upadacitinib for the treatment of rheumatoid arthritis in a 74-year-old Japanese woman has been described,3 and the JAK/signal transducer and activator of transcription pathway has been implicated in the development of other CD30+ lymphoproliferative disorders.4,5
An association between JAK inhibitors and aggressive B-cell lymphomas has been described. In an observational study of 626 patients with myeloproliferative neoplasia by Porpaczy et al,6 4 of 69 (5.8%) patients treated with JAK inhibitors developed an aggressive B-cell lymphoma, whereas only 2 of 557 (0.36%) patients who did not receive JAK-inhibitor therapy developed an aggressive B-cell lymphoma. In contrast, a retrospective analysis of 2583 patients with myeloproliferative neoplasia by Pemmaraju et al7 found no significant increase in lymphoma rates in the JAK inhibitor–treated population as compared with the non-JAK inhibitor–treated group; 9 (0.56%) cases of lymphoma occurred in 1617 patients with myelofibrosis, of which 6 had exposure to JAK inhibitor therapy and 3 had no exposure to JAK inhibitor therapy (P=.082) and 5 (0.52%) cases of lymphoma occurred in 966 patients with essential thrombocythemia or polycythemia vera, none of whom had exposure to JAK inhibitor therapy.Finally, some evidence suggests the use of JAK inhibitors may be associated with an elevated risk of malignancies overall. The ORAL Surveillance study found the incidence of all cancers, excluding nonmelanoma skin cancer (NMSC), in patients treated with tofacitinib to be 4.2% (122/2911) compared with 2.9% (42/1451) in patients treated with tumor necrosis factor α inhibitors; it should be noted that the patients in this study were restricted to adults aged 50 years and older who were undergoing treatment for rheumatoid arthritis.8 In a safety profile study for upadacitinib, a higher rate of malignancies, excluding NMSC, was found in patients with AD treated with upadacitinib 30 mg/d than in patients treated with 15 mg/d; however, the overall rates of malignancies, excluding NMSC, in patients treated with upadacitinib were comparable to the standard incidence rates of malignancies in the general population derived from Surveillance, Epidemiology, and End Results data.9
In summary, we present a case of cutaneous ALCL arising after treatment with upadacitinib for AD. While some literature suggests AD may independently predispose patients to the development of CD30+ lymphoproliferative disorders, the onset of our patient’s cutaneous ALCL 10 months after initiation of upadacitinib is suggestive of an association between his lymphoproliferative disorder and JAK inhibition. Further studies are needed to better characterize the risk of lymphoproliferative disorders and other malignancies in patients treated with JAK inhibitors.
To the Editor:
A 22-year-old man presented to our clinic with a history of longstanding widespread recalcitrant atopic dermatitis (AD) since early childhood. He had been treated by an outside physician with topical steroids and nonsteroidal medications without notable improvement as well as with dupilumab, which was discontinued due to the development of severe head and neck dermatitis. Given the severity of his AD on presentation, we initiated treatment with upadacitinib 15 mg/d, which resulted in partial improvement. The dose was increased to 30 mg/d at 3 months with further clinical improvement.
Ten months after the patient was started on upadacitinib, he presented for a follow-up evaluation and reported a new nontender nodule on the scalp. A punch biopsy revealed a dense dermal and subcutaneous lymphoid infiltrate (Figure 1) composed of many large atypical CD2+/CD5+/CD45+ T cells with partial loss of CD3 expression (Figure 2). The atypical cells demonstrated diffuse CD30+ expression (Figure 3) and a CD4:CD8 ratio of greater than 50:1 (Figures 4 and 5). He was diagnosed with anaplastic large cell lymphoma (ALCL), and the upadacitinib was discontinued. No additional therapies directed toward ALCL were initiated.





Over the next 2 weeks, the patient developed additional nodules on the postauricular skin and trunk that demonstrated similar histopathology and immunophenotype to the original scalp nodule. T-cell receptor gene rearrangement studies demonstrated shared clonal peaks in these subsequent nodules. A concurrent biopsy of an eczematous plaque on the back showed spongiotic dermatitis without evidence of cutaneous T-cell lymphoma; gene rearrangement studies from this site were negative. A positron emission tomography–computed tomography scan showed mildly hypermetabolic cervical, axillary, and inguinal lymph nodes, which were favored to be reactive. Narrow-band UVB phototherapy was initiated for management of the AD, and no additional nodules developed over the subsequent months.
Janus kinase (JAK) inhibitors are immunomodulatory small molecules that interfere with JAK–signal transducer and activator of transcription signaling involving 1 or more isoforms (eg, JAK1, JAK2, JAK3, tyrosine kinase 2) and have been used to treat various inflammatory conditions, including rheumatoid arthritis, psoriatic arthritis, psoriasis, axial spondyloarthritis, inflammatory bowel disease, and AD.1 Upadacitinib is an oral selective JAK1 inhibitor approved by the US Food and Drug Administration for treatment of moderate to severe AD in adults and children aged 12 years and older.2 A search of PubMed using the terms upadacitinib or Rinvoq and anaplastic large cell lymphoma did not identify any cases of cutaneous ALCL arising after treatment with upadacitinib. However, a case of lymphomatoid papulosis after initiation of upadacitinib for the treatment of rheumatoid arthritis in a 74-year-old Japanese woman has been described,3 and the JAK/signal transducer and activator of transcription pathway has been implicated in the development of other CD30+ lymphoproliferative disorders.4,5
An association between JAK inhibitors and aggressive B-cell lymphomas has been described. In an observational study of 626 patients with myeloproliferative neoplasia by Porpaczy et al,6 4 of 69 (5.8%) patients treated with JAK inhibitors developed an aggressive B-cell lymphoma, whereas only 2 of 557 (0.36%) patients who did not receive JAK-inhibitor therapy developed an aggressive B-cell lymphoma. In contrast, a retrospective analysis of 2583 patients with myeloproliferative neoplasia by Pemmaraju et al7 found no significant increase in lymphoma rates in the JAK inhibitor–treated population as compared with the non-JAK inhibitor–treated group; 9 (0.56%) cases of lymphoma occurred in 1617 patients with myelofibrosis, of which 6 had exposure to JAK inhibitor therapy and 3 had no exposure to JAK inhibitor therapy (P=.082) and 5 (0.52%) cases of lymphoma occurred in 966 patients with essential thrombocythemia or polycythemia vera, none of whom had exposure to JAK inhibitor therapy.Finally, some evidence suggests the use of JAK inhibitors may be associated with an elevated risk of malignancies overall. The ORAL Surveillance study found the incidence of all cancers, excluding nonmelanoma skin cancer (NMSC), in patients treated with tofacitinib to be 4.2% (122/2911) compared with 2.9% (42/1451) in patients treated with tumor necrosis factor α inhibitors; it should be noted that the patients in this study were restricted to adults aged 50 years and older who were undergoing treatment for rheumatoid arthritis.8 In a safety profile study for upadacitinib, a higher rate of malignancies, excluding NMSC, was found in patients with AD treated with upadacitinib 30 mg/d than in patients treated with 15 mg/d; however, the overall rates of malignancies, excluding NMSC, in patients treated with upadacitinib were comparable to the standard incidence rates of malignancies in the general population derived from Surveillance, Epidemiology, and End Results data.9
In summary, we present a case of cutaneous ALCL arising after treatment with upadacitinib for AD. While some literature suggests AD may independently predispose patients to the development of CD30+ lymphoproliferative disorders, the onset of our patient’s cutaneous ALCL 10 months after initiation of upadacitinib is suggestive of an association between his lymphoproliferative disorder and JAK inhibition. Further studies are needed to better characterize the risk of lymphoproliferative disorders and other malignancies in patients treated with JAK inhibitors.
- Strangfeld A, Hierse F, Rau R, et al. Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German biologics register RABBIT. Arthritis Res Ther. 2010;12:R5. doi:10.1186/ar2904
- Rinvoq. Highlights of prescribing information. Abbvie Inc; 2024. Accessed January 31, 2026. https://www.rxabbvie.com/pdf/rinvoq_pi.pdf
- Iinuma S, Hayashi K, Noguchi A, et al. Lymphomatoid papulosis during upadacitinib treatment for rheumatoid arthritis. Eur J Dermatol. 2022;32:142-143. doi:10.1684/ejd.2022.4238
- Quesada AE, Zhang Y, Ptashkin R, et al. Next generation sequencing of breast implant-associated anaplastic large cell lymphomas reveals a novel STAT3-JAK2 fusion among other activating genetic alterations within the JAK-STAT pathway. Breast J. 2021;27:314-321. doi:10.1111/tbj.14205
- Maurus K, Appenzeller S, Roth S, et al. Recurrent oncogenic JAK and STAT alterations in cutaneous CD30-positive lymphoproliferative disorders. J Invest Dermatol. 2020;140:2023-2031.e1. doi:10.1016/j.jid.2020.02.019
- Porpaczy E, Tripolt S, Hoelbl-Kovacic A, et al. Aggressive B-cell lymphomas in patients with myelofibrosis receiving JAK1/2 inhibitor therapy. Blood. 2018;132:694-706. doi:10.1182/blood-2017-10-810739
- Pemmaraju N, Kantarjian H, Nastoupil L, et al. Characteristics of patients with myeloproliferative neoplasms with lymphoma, with or without JAK inhibitor therapy. Blood. 2019;133:2348-2351. doi:10.1182/blood-2019-01-897637
- Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386:316-326. doi:10.1056/NEJMoa2109927
- Burmester GR, Cohen SB, Winthrop KL, et al. Safety profile of upadacitinib over 15 000 patient-years across rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and atopic dermatitis. RMD Open. 2023;9:E002735. doi:10.1136/rmdopen-2022-002735
- Strangfeld A, Hierse F, Rau R, et al. Risk of incident or recurrent malignancies among patients with rheumatoid arthritis exposed to biologic therapy in the German biologics register RABBIT. Arthritis Res Ther. 2010;12:R5. doi:10.1186/ar2904
- Rinvoq. Highlights of prescribing information. Abbvie Inc; 2024. Accessed January 31, 2026. https://www.rxabbvie.com/pdf/rinvoq_pi.pdf
- Iinuma S, Hayashi K, Noguchi A, et al. Lymphomatoid papulosis during upadacitinib treatment for rheumatoid arthritis. Eur J Dermatol. 2022;32:142-143. doi:10.1684/ejd.2022.4238
- Quesada AE, Zhang Y, Ptashkin R, et al. Next generation sequencing of breast implant-associated anaplastic large cell lymphomas reveals a novel STAT3-JAK2 fusion among other activating genetic alterations within the JAK-STAT pathway. Breast J. 2021;27:314-321. doi:10.1111/tbj.14205
- Maurus K, Appenzeller S, Roth S, et al. Recurrent oncogenic JAK and STAT alterations in cutaneous CD30-positive lymphoproliferative disorders. J Invest Dermatol. 2020;140:2023-2031.e1. doi:10.1016/j.jid.2020.02.019
- Porpaczy E, Tripolt S, Hoelbl-Kovacic A, et al. Aggressive B-cell lymphomas in patients with myelofibrosis receiving JAK1/2 inhibitor therapy. Blood. 2018;132:694-706. doi:10.1182/blood-2017-10-810739
- Pemmaraju N, Kantarjian H, Nastoupil L, et al. Characteristics of patients with myeloproliferative neoplasms with lymphoma, with or without JAK inhibitor therapy. Blood. 2019;133:2348-2351. doi:10.1182/blood-2019-01-897637
- Ytterberg SR, Bhatt DL, Mikuls TR, et al. Cardiovascular and cancer risk with tofacitinib in rheumatoid arthritis. N Engl J Med. 2022;386:316-326. doi:10.1056/NEJMoa2109927
- Burmester GR, Cohen SB, Winthrop KL, et al. Safety profile of upadacitinib over 15 000 patient-years across rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis and atopic dermatitis. RMD Open. 2023;9:E002735. doi:10.1136/rmdopen-2022-002735
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
Development of Primary Cutaneous Anaplastic Large Cell Lymphoma Following Treatment With Upadacitinib for Atopic Dermatitis
Practice Points
- Janus kinase inhibitors are immunomodulators used for the treatment of various inflammatory conditions, including atopic dermatitis.
- Treatment with Janus kinase inhibitors may be associated with the development of CD3012+ lymphoproliferative disorders such as cutaneous anaplastic large cell lymphoma.
Acute Pustular Eruption on the Hands
Acute Pustular Eruption on the Hands
THE DIAGNOSIS: Neutrophilic Dermatosis of the Dorsal Hands
Histopathology showed a unilocular pustule with a dense neutrophilic infiltrate of the superficial dermis. Minimal vascular alterations also were observed. These findings were consistent with a diagnosis of neutrophilic dermatosis of the dorsal hands (NDDH). Our patient was treated successfully with systemic corticosteroids (1 mg/kg/d) with rapid improvement after 10 days of treatment.
Neutrophilic dermatosis of the dorsal hands is an evolving disease concept that was first described as pustular vasculitis by Strutton et al1 in 1995. Galaria et al2 subsequently identified NDDH as a clinical entity associating tender erythematous plaques, pustules, bullae, and/or ulcers on the dorsal hands with histologic features of Sweet syndrome (SS). After reviewing 9 cases of NDDH—all of which demonstrated clinical, laboratory, and histologic characteristics of SS—Walling et al3 concluded that NDDH was best understood as a distributional variant of SS.
Our patient presented with vascular alterations described as a reactive response to the neutrophilic infiltration. The presence of vasculitis in SS and NDDH biopsies is considered as an occasional epiphenomenon and should not rule out the diagnosis of NDDH.3 A literature review of 123 cases of NDDH revealed the presence of vasculitis in 36 (29.5%) patients.4 With regard to other clinical findings, it has been suggested that an increased white blood cell count and elevated C-reactive protein level, as was seen in our patient, may be observed in NDDH, albeit less frequently than in classical SS.4
While palmar involvement of NDDH is considered rare, the recent review of 123 cases of NDDH identified palmar lesions in 5 patients (4.1%).4 Earlier reviews had identified 12 historical cases.5 Palmar manifestations of NDDH have been shown to be associated with erythematous nonulcerated lesions (as opposed to the classical ulcerative or pustular plaques) and a lower association with hematologic malignancies.5
In our patient’s case, dyshidrosis was excluded due to the presence of painful ulcerative plaques rather than pruritic, deep-seated vesicles. Pustular psoriasis typically manifests with sterile pustules on the palms and soles; however, the rapid onset of ulcerative, necrotic plaques and substantial edema are more specific to NDDH. Poststreptococcal pustulosis generally follows a streptococcal infection and lacks the violaceous undermined borders seen in NDDH. Reactive arthritis manifests with hyperkeratotic plaques and is associated with the clinical triad of urethritis, conjunctivitis, and arthritis, which were absent in our patient.
The histologic differential diagnosis of NDDH includes infection, pyoderma gangrenosum, bowel-associated dermatosis-arthritis syndrome, rheumatoid neutrophilic dermatitis, and erythema elevatum diutinum3,4; however, these conditions typically manifest with distinct clinical features that allow for differentiation, despite histologic similarities. The wide histologic spectrum of neutrophilic dermatosis may contribute to variable clinical manifestations and an evolving disease concept, as the classification of NDDH has changed from a primary vasculitis to a variant of SS. However, this evolution does not affect the appropriate management, as they all have shown good response to corticosteroid treatment.4,6
- Strutton G, Weedon D, Robertson I. Pustular vasculitis of the hands. J Am Acad Dermatol. 1995;32(2 pt 1):192-198.
- Galaria NA, Junkins-Hopkins JM, Kligman D, et al. Neutrophilic dermatosis of the dorsal hands: pustular vasculitis revisited. J Am Acad Dermatol. 2000;43(5 pt 1):870-874.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63
- Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 cases. J Am Acad Dermatol. 2023;88:1338-1344.
- Arandes-Marcocci J, Altemir-Vidal A, Iglesias-Plaza A, et al. Neutrophilic dermatosis of the hands with palmar involvement: does it have clinical implication? Int J Dermatol. 2020;59:736-738.
- Del Pozo J, Sacristán F, Martínez W, et al. Neutrophilic dermatosis of the hands: presentation of eight cases and review of the literature. J Dermatol. 2007;34:243-247.
THE DIAGNOSIS: Neutrophilic Dermatosis of the Dorsal Hands
Histopathology showed a unilocular pustule with a dense neutrophilic infiltrate of the superficial dermis. Minimal vascular alterations also were observed. These findings were consistent with a diagnosis of neutrophilic dermatosis of the dorsal hands (NDDH). Our patient was treated successfully with systemic corticosteroids (1 mg/kg/d) with rapid improvement after 10 days of treatment.
Neutrophilic dermatosis of the dorsal hands is an evolving disease concept that was first described as pustular vasculitis by Strutton et al1 in 1995. Galaria et al2 subsequently identified NDDH as a clinical entity associating tender erythematous plaques, pustules, bullae, and/or ulcers on the dorsal hands with histologic features of Sweet syndrome (SS). After reviewing 9 cases of NDDH—all of which demonstrated clinical, laboratory, and histologic characteristics of SS—Walling et al3 concluded that NDDH was best understood as a distributional variant of SS.
Our patient presented with vascular alterations described as a reactive response to the neutrophilic infiltration. The presence of vasculitis in SS and NDDH biopsies is considered as an occasional epiphenomenon and should not rule out the diagnosis of NDDH.3 A literature review of 123 cases of NDDH revealed the presence of vasculitis in 36 (29.5%) patients.4 With regard to other clinical findings, it has been suggested that an increased white blood cell count and elevated C-reactive protein level, as was seen in our patient, may be observed in NDDH, albeit less frequently than in classical SS.4
While palmar involvement of NDDH is considered rare, the recent review of 123 cases of NDDH identified palmar lesions in 5 patients (4.1%).4 Earlier reviews had identified 12 historical cases.5 Palmar manifestations of NDDH have been shown to be associated with erythematous nonulcerated lesions (as opposed to the classical ulcerative or pustular plaques) and a lower association with hematologic malignancies.5
In our patient’s case, dyshidrosis was excluded due to the presence of painful ulcerative plaques rather than pruritic, deep-seated vesicles. Pustular psoriasis typically manifests with sterile pustules on the palms and soles; however, the rapid onset of ulcerative, necrotic plaques and substantial edema are more specific to NDDH. Poststreptococcal pustulosis generally follows a streptococcal infection and lacks the violaceous undermined borders seen in NDDH. Reactive arthritis manifests with hyperkeratotic plaques and is associated with the clinical triad of urethritis, conjunctivitis, and arthritis, which were absent in our patient.
The histologic differential diagnosis of NDDH includes infection, pyoderma gangrenosum, bowel-associated dermatosis-arthritis syndrome, rheumatoid neutrophilic dermatitis, and erythema elevatum diutinum3,4; however, these conditions typically manifest with distinct clinical features that allow for differentiation, despite histologic similarities. The wide histologic spectrum of neutrophilic dermatosis may contribute to variable clinical manifestations and an evolving disease concept, as the classification of NDDH has changed from a primary vasculitis to a variant of SS. However, this evolution does not affect the appropriate management, as they all have shown good response to corticosteroid treatment.4,6
THE DIAGNOSIS: Neutrophilic Dermatosis of the Dorsal Hands
Histopathology showed a unilocular pustule with a dense neutrophilic infiltrate of the superficial dermis. Minimal vascular alterations also were observed. These findings were consistent with a diagnosis of neutrophilic dermatosis of the dorsal hands (NDDH). Our patient was treated successfully with systemic corticosteroids (1 mg/kg/d) with rapid improvement after 10 days of treatment.
Neutrophilic dermatosis of the dorsal hands is an evolving disease concept that was first described as pustular vasculitis by Strutton et al1 in 1995. Galaria et al2 subsequently identified NDDH as a clinical entity associating tender erythematous plaques, pustules, bullae, and/or ulcers on the dorsal hands with histologic features of Sweet syndrome (SS). After reviewing 9 cases of NDDH—all of which demonstrated clinical, laboratory, and histologic characteristics of SS—Walling et al3 concluded that NDDH was best understood as a distributional variant of SS.
Our patient presented with vascular alterations described as a reactive response to the neutrophilic infiltration. The presence of vasculitis in SS and NDDH biopsies is considered as an occasional epiphenomenon and should not rule out the diagnosis of NDDH.3 A literature review of 123 cases of NDDH revealed the presence of vasculitis in 36 (29.5%) patients.4 With regard to other clinical findings, it has been suggested that an increased white blood cell count and elevated C-reactive protein level, as was seen in our patient, may be observed in NDDH, albeit less frequently than in classical SS.4
While palmar involvement of NDDH is considered rare, the recent review of 123 cases of NDDH identified palmar lesions in 5 patients (4.1%).4 Earlier reviews had identified 12 historical cases.5 Palmar manifestations of NDDH have been shown to be associated with erythematous nonulcerated lesions (as opposed to the classical ulcerative or pustular plaques) and a lower association with hematologic malignancies.5
In our patient’s case, dyshidrosis was excluded due to the presence of painful ulcerative plaques rather than pruritic, deep-seated vesicles. Pustular psoriasis typically manifests with sterile pustules on the palms and soles; however, the rapid onset of ulcerative, necrotic plaques and substantial edema are more specific to NDDH. Poststreptococcal pustulosis generally follows a streptococcal infection and lacks the violaceous undermined borders seen in NDDH. Reactive arthritis manifests with hyperkeratotic plaques and is associated with the clinical triad of urethritis, conjunctivitis, and arthritis, which were absent in our patient.
The histologic differential diagnosis of NDDH includes infection, pyoderma gangrenosum, bowel-associated dermatosis-arthritis syndrome, rheumatoid neutrophilic dermatitis, and erythema elevatum diutinum3,4; however, these conditions typically manifest with distinct clinical features that allow for differentiation, despite histologic similarities. The wide histologic spectrum of neutrophilic dermatosis may contribute to variable clinical manifestations and an evolving disease concept, as the classification of NDDH has changed from a primary vasculitis to a variant of SS. However, this evolution does not affect the appropriate management, as they all have shown good response to corticosteroid treatment.4,6
- Strutton G, Weedon D, Robertson I. Pustular vasculitis of the hands. J Am Acad Dermatol. 1995;32(2 pt 1):192-198.
- Galaria NA, Junkins-Hopkins JM, Kligman D, et al. Neutrophilic dermatosis of the dorsal hands: pustular vasculitis revisited. J Am Acad Dermatol. 2000;43(5 pt 1):870-874.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63
- Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 cases. J Am Acad Dermatol. 2023;88:1338-1344.
- Arandes-Marcocci J, Altemir-Vidal A, Iglesias-Plaza A, et al. Neutrophilic dermatosis of the hands with palmar involvement: does it have clinical implication? Int J Dermatol. 2020;59:736-738.
- Del Pozo J, Sacristán F, Martínez W, et al. Neutrophilic dermatosis of the hands: presentation of eight cases and review of the literature. J Dermatol. 2007;34:243-247.
- Strutton G, Weedon D, Robertson I. Pustular vasculitis of the hands. J Am Acad Dermatol. 1995;32(2 pt 1):192-198.
- Galaria NA, Junkins-Hopkins JM, Kligman D, et al. Neutrophilic dermatosis of the dorsal hands: pustular vasculitis revisited. J Am Acad Dermatol. 2000;43(5 pt 1):870-874.
- Walling HW, Snipes CJ, Gerami P, et al. The relationship between neutrophilic dermatosis of the dorsal hands and sweet syndrome: report of 9 cases and comparison to atypical pyoderma gangrenosum. Arch Dermatol. 2006;142:57-63
- Micallef D, Bonnici M, Pisani D, et al. Neutrophilic dermatosis of the dorsal hands: a review of 123 cases. J Am Acad Dermatol. 2023;88:1338-1344.
- Arandes-Marcocci J, Altemir-Vidal A, Iglesias-Plaza A, et al. Neutrophilic dermatosis of the hands with palmar involvement: does it have clinical implication? Int J Dermatol. 2020;59:736-738.
- Del Pozo J, Sacristán F, Martínez W, et al. Neutrophilic dermatosis of the hands: presentation of eight cases and review of the literature. J Dermatol. 2007;34:243-247.
Acute Pustular Eruption on the Hands
Acute Pustular Eruption on the Hands
A 56-year-old woman was referred to the dermatology department for a painful acral pustular eruption of 6 days’ duration. Her medical history was otherwise unremarkable. Physical examination revealed multiple pustules on the hands with large blisters on an erythematous base and painful surface ulceration (top). Papulonodular infiltrated lesions also were observed on the dorsal aspect of the hands (bottom). There were no additional systemic symptoms. Routine laboratory tests showed hyperleukocytosis at 17.9×103/mm3 (reference range, 4-10×103/mm3) with neutrophils at 12.3×103/mm3 (1.8-7.5×103/mm3) and elevated C-reactive protein at 67 mg/L (<5 mg/L). Screening for hematologic neoplasms, solid tumors, and inflammatory bowel disease was negative. An incisional biopsy was performed on a pustule on the palm of the left hand.
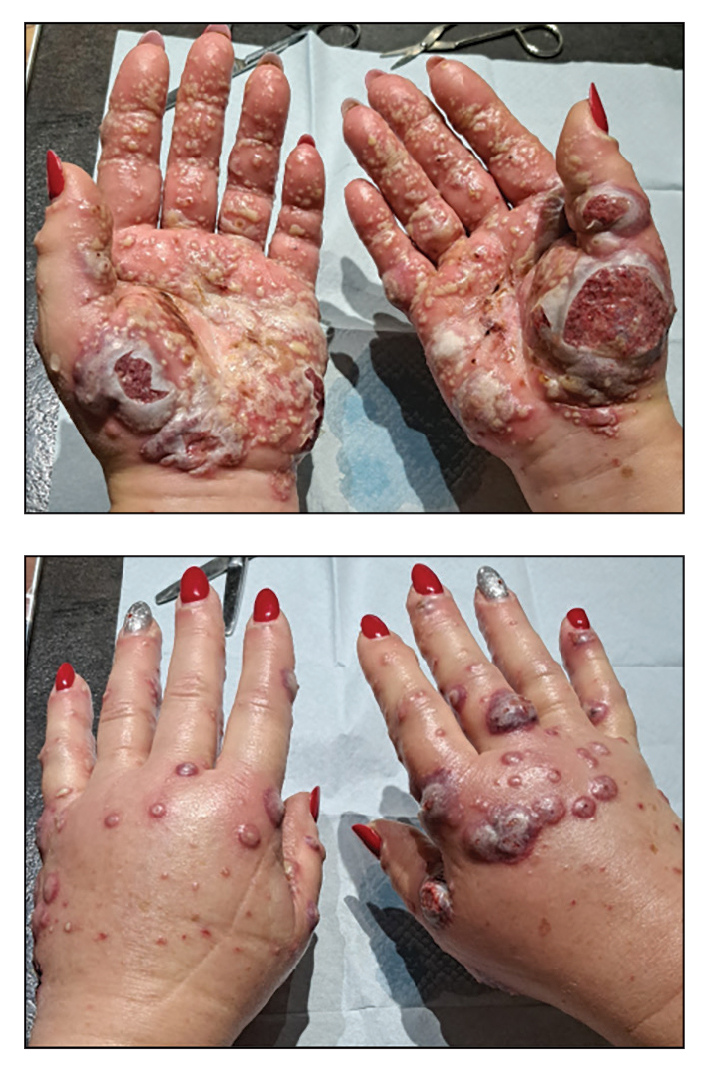
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.
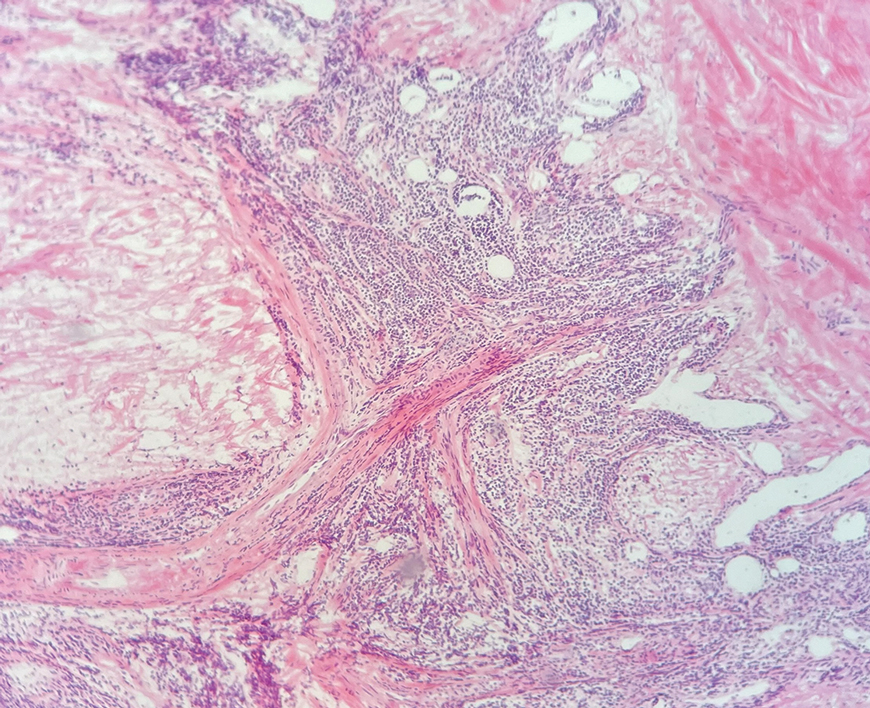
Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.



Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
To the Editor:
Primary cutaneous B-cell lymphomas (pcBCLs) can clinically mimic basal cell carcinomas (BCCs); however, histopathologic examination typically demonstrates features of lymphoma without evidence of an epithelial tumor. We present the case of a patient who demonstrated histologic features of both pcBCL and BCC in the same lesion, which was discovered during Mohs micrographic surgery.
An 84-year-old man presented for Mohs surgery for a biopsy-proven nodular and infiltrative BCC on the right superior helix of the ear of 1 year’s duration. Physical examination of the ear revealed a 1.0×1.3–cm ulcerated indurated plaque with rolled borders and a central hyperkeratotic crust (Figure 1). Frozen sections from the first Mohs stage demonstrated residual superficial, infiltrative, and basosquamous BCC (Figure 2). In addition, there was a brisk inflammatory infiltrate throughout the deep margins. The second stage showed no residual BCC, but there still was a brisk atypical lymphocytic infiltrate, with some areas showing lymphocytes in a linear cordlike distribution (Figure 3). Permanent sections demonstrated infiltration of small to medium lymphoid cells. Immunohistochemistry stains were positive for CD20 and BCL2 and negative for CD5, CD10, BCL6, and CD43; a low Ki-67 proliferation fraction also was observed. B-cell clonality studies and polymerase chain reaction demonstrated rearrangements of the IgH and IgK genes, consistent with primary cutaneous marginal zone lymphoma (pcMZL). Positron emission tomography showed no spread of malignancy; therefore, medical oncology recommended observation and close monitoring.



Primary cutaneous B-cell lymphoma accounts for approximately 25% of all cutaneous lymphomas.1 Three main cutaneous subtypes exist: pcMZL; primary cutaneous follicular center lymphoma; and primary cutaneous diffuse large B-cell lymphoma, leg type. The second most common type of cutaneous lymphoma, pcMZL, accounts for 25% of cases of pcBCL.1 Primary cutaneous follicular center lymphoma makes up 60% of cutaneous lymphomas, and the remainder are primary cutaneous diffuse large B-cell lymphoma, leg type. All share a notable male predominance and onset most commonly in the sixth through eighth decades of life, although they also can occur in younger patients.1
Histologically, pcMZL has 2 distinct subtypes: one resembling mucosal-associated lymphoid tissue lymphomas and a more clinically aggressive subtype with heavy chain class switching, although intermediate forms also exist. Both are characterized by diffuse and/or nodular infiltrates in the subcutis and dermis with sparing of the epidermis. Often, these infiltrates are more prominent in the deeper sections examined, and occasionally they may be accompanied by germinal center follicles. Immunohistochemical stains are key in determining the pcBCL subtype. Primary cutaneous marginal zone lymphoma will most commonly show a BCL2+, BCL6–, CD20+, and CD10– immunophenotype, as in our case. If a majority of cells have undergone plasmacytoid differentiation, loss of CD20 can occur, but retention of other B-cell markers, such as CD79a and CD19, will be seen. Proliferation fraction via Ki-67 commonly is low, reflecting the indolence of this subtype of lymphoma.1
Monoclonal rearrangement of immunoglobulins also can occur, with IgH rearrangements detected in 60% to 80% of cases of pcMZL. Translocations are not a reliable method of diagnosis for pcMZL but can be present in a variable manner, with t(14;18), t(3;14), and t(11;18) reported in a subset of cases.2 Leukemic infiltrates encountered on frozen sections should prompt the Mohs surgeon to consider the possibility of a concomitant leukemia or lymphoma. In one study, 36% (20/55) of patients with chronic lymphocytic leukemia (CLL) were found to have predominantly leukemic B-cell infiltrates on frozen sections.3 Numerous reports also exist of asymptomatic patients being diagnosed with CLL due to leukemic infiltrates identified during Mohs surgery.4,5 Patients with systemic hematologic malignancies, including CLL and non-Hodgkin lymphoma, also are known to be at an increased risk for skin cancers, including keratinocyte cancers, melanoma, and Merkel cell carcinoma. This can be attributed partially to immunosuppression, a well-known risk factor for development of cutaneous malignancies.5 Padgett et al5 speculated that local immune suppression due to underlying pcBCL and reaction of lymphocytes to tumor antigens could have played a role in the development of BCC at this site. If a leukemic infiltrate is demonstrated, the surgeon should consider sending tissue for permanent section and immunostaining. This can be helpful to determine if it is a reactive or neoplastic process and aid in characterizing the leukemic infiltrate if it is suspected to be neoplastic in nature.
There are numerous reports of pcBCL imitating the cutaneous findings of BCC clinically, but this is quite uncommon on histopathology. As in our case, findings of sheets of dense, monomorphic lymphocytes; inability to clear inflammation on deeper Mohs sections; presence of primordial follicles; and atypical cytology, including predominance of blastic forms, plasmacytoid cells, or cleaved lymphocytes, should give the clinician pause to consider further evaluation through permanent sections as well as genetic and immunoglobulin studies by a dermatopathologist. This case highlights the importance of further evaluation when an atypical finding is encountered during Mohs surgery.
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
- Goyal A, LeBlanc RE, Carter JB. Cutaneous B-cell lymphoma. Hematol Oncol Clin North Am. 2019;33:149-161. doi:10.1016/j.hoc.2018.08.006
- Vitiello P, Sica A, Ronchi A, et al. Primary cutaneous B-cell lymphomas: an update. Front Oncol. 2020;10:651. doi:10.3389/fonc.2020.00651
- Mehrany K, Byrd DR, Roenigk RK, et al. Lymphocytic infiltrates and subclinical epithelial tumor extension in patients with chronic leukemia and solid-organ transplantation. Dermatol Surg. 2003;29:129-134. doi:10.1046/j.1524-4725.2003.29034.x
- Walters M, Chang C, Castillo JR. Diagnosis of chronic lymphocytic leukemia during Mohs micrographic surgery. JAAD Case Rep. 2023;33:1-3. doi:10.1016/j.jdcr.2022.12.012
- Padgett JK, Parlette HL, English JC. A diagnosis of chronic lymphocytic leukemia prompted by cutaneous lymphocytic infiltrates present in mohs micrographic surgery frozen sections. Dermatol Surg. 2003;29:769-771. doi:10.1046/j.1524-4725.2003.29194.x
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Primary Cutaneous Marginal Zone B-Cell Lymphoma Discovered During Mohs Surgery for Basal Cell Carcinoma
Practice Points
- Collision tumors of cutaneous B-cell lymphoma and basal cell carcinoma occurring within the same lesion are uncommon findings during Mohs surgery.
- Sheets of atypical monomorphic lymphocytes on deeper Mohs sections should prompt the surgeon to consider further evaluation, including sending tissue for permanent sections.
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Photosensitivity refers to clinical manifestations arising from exposure to sunlight. Photodermatoses encompass a group of skin diseases caused by varying degrees of radiation exposure, including UV radiation and visible light. Photodermatoses can be categorized into 5 main types: primary, exogenous, photoexacerbated, metabolic, and genetic.1 The clinical features of photodermatoses vary depending on the underlying cause but often include pruritic flares, wheals, or dermatitis on sun-exposed areas of the skin.2 While photodermatoses typically are not life threatening, they can greatly impact patients’ quality of life. It is crucial to emphasize the importance of photoprotection and sunlight avoidance to patients as preventive measures against the manifestations of these skin diseases. Furthermore, we present a case of photocontact dermatitis (PCD) and discuss common causative agents, diagnostic mimickers, and treatment options.
Case Report
A 51-year-old woman with no relevant medical history presented to the dermatology clinic with a rash on the neck and under the eyes of 6 days’ duration. The rash was intermittently pruritic but otherwise asymptomatic. The patient reported that she had spent extensive time on the golf course the day of the rash onset and noted that a similar rash had occurred one other time 2 to 3 months prior, also following a prolonged period on the golf course. She had been using over-the-counter fexofenadine 180 mg and over-the-counter lidocaine spray for symptom relief.
Upon physical examination, erythematous patches were appreciated in a photodistributed pattern on the arms, legs, neck, face, and chest—areas that were not covered by clothing (Figures 1-3). Due to the distribution and morphology of the erythematous patches along with clinical course of onset following exposure to various environmental agents including pesticides, herbicides, oak, and pollen, a diagnosis of PCD was made. The patient was prescribed hydrocortisone cream 2.5%, fluticasone propionate cream 0.05%, and methylprednisolone in addition to the antihistamine. Improvement was noted after 3 days with complete resolution of the skin manifestations. She was counseled on wearing clothing with a universal protection factor rating of 50+ when on the golf course and when sun exposure is expected for an extended period of time.
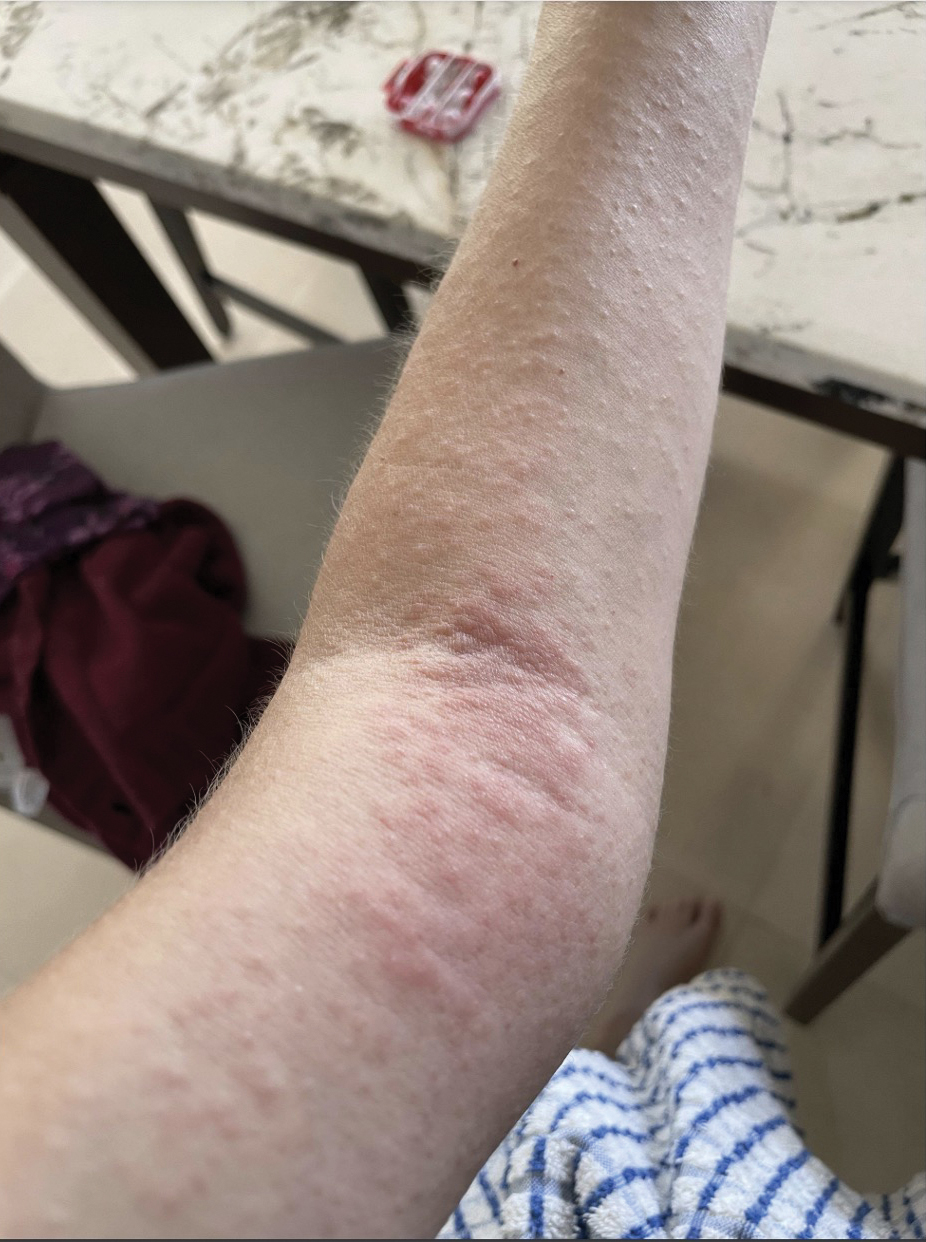
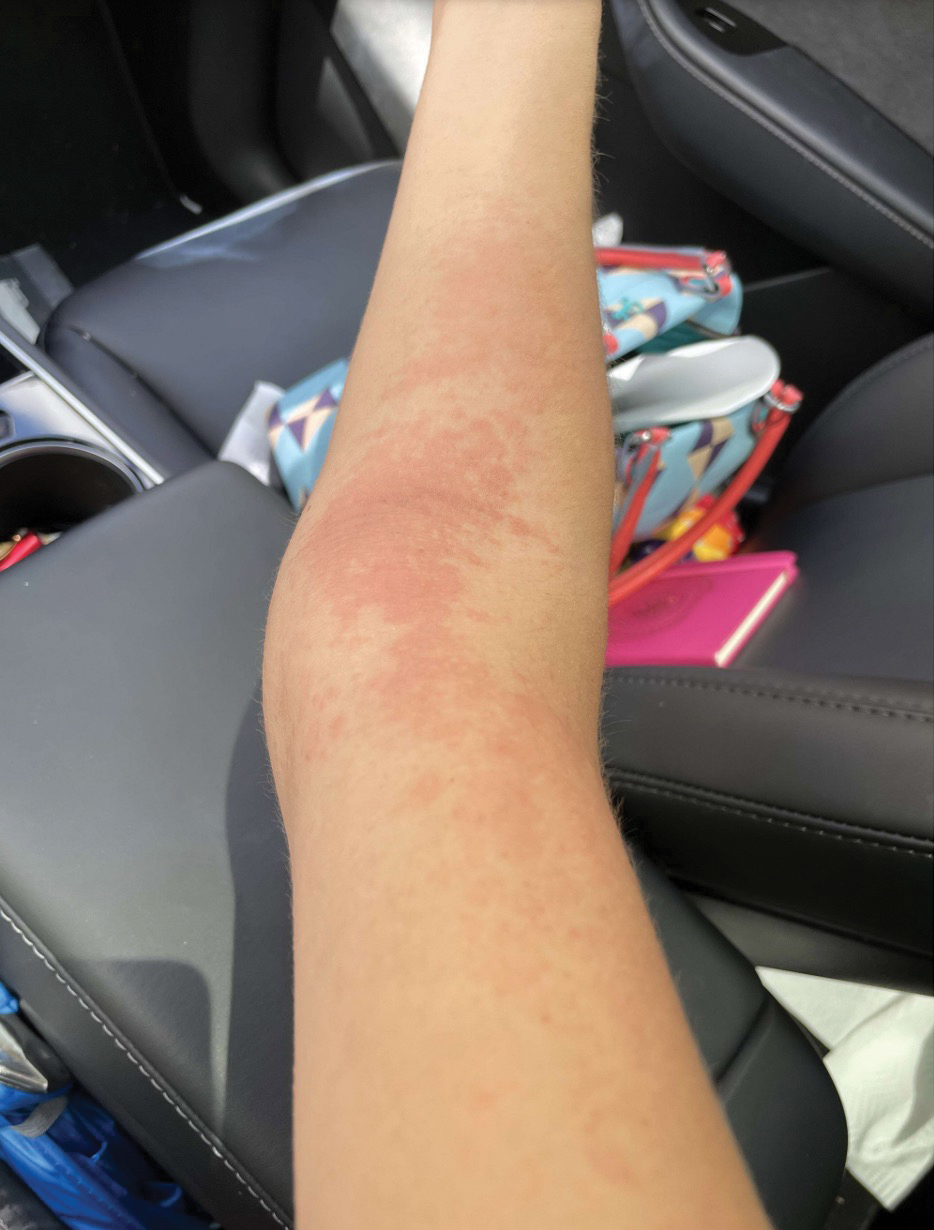
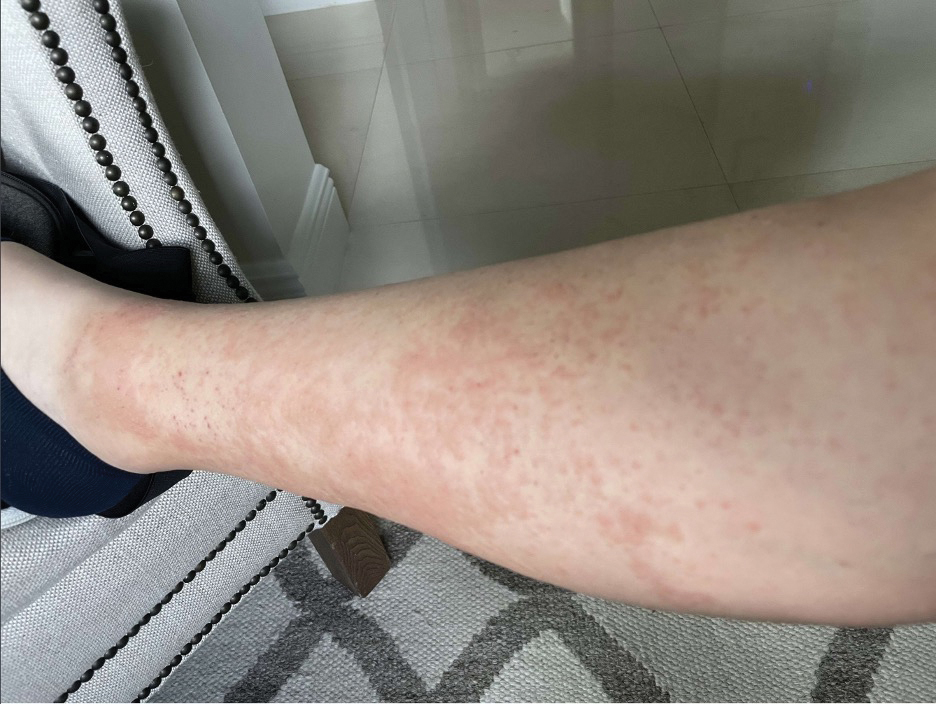
Causative Agents
Photodermatoses are caused by antigenic substances that lead to photosensitization acquired by either contact or oral ingestion with subsequent sensitization to UV radiation. Halogenated salicylanilide, fenticlor, hexachlorophene, bithionol and, in rare cases, sunscreens, have been reported as triggers.3 In a study performed in 2010, sunscreens, antimicrobial agents, medications, fragrances, plants/plant derivatives, and pesticides were the most commonly reported offending agents listed from highest to lowest frequency. Of the antimicrobial agents, fenticlor, a topical antimicrobial and antifungal that is now mostly used in veterinary medicine, was the most common culprit, causing 60% of cases.4,5
Clinical Manifestations
Clinical manifestations of photodermatoses vary depending upon the specific type of reaction. Examples of primary photodermatoses include polymorphous light eruption (PMLE) and solar urticaria. The cardinal symptoms of PMLE consist of severely pruritic skin lesions that can have macular, papular, papulovesicular, urticarial, multiformelike, and plaquelike variants that develop hours to days after sun exposure.3 Conversely, solar urticaria commonly develops more abruptly, with indurated plaques and wheals appearing on the arms and neck within 30 minutes of sun exposure. The lesions typically resolve within 24 hours.1
Examples of the exogenous subtype include drug-induced photosensitivity, PCD, and pseudoporphyria, with the common clinical presentation of eruption following contact with the causative agent. Drug-induced photosensitivity primarily manifests as a severe sunburnlike rash commonly caused by systemic drugs such as tetracyclines. Photocontact dermatitis is limited to sun-exposed areas of the skin and is caused by a reactive irritant such as chemicals or topical creams. Pseudoporphyria, usually caused by nonsteroidal anti-inflammatory drugs, can manifest with skin fragility and subepidermal blisters.6
Photoexacerbated photodermatoses encompass a variety of conditions ranging from hyperpigmentation disorders such as melasma to autoimmune conditions such as systemic lupus erythematosus (SLE) and dermatomyositis (DM). Common clinical features of these diseases include photodistributed erythema, often involving the cheeks, upper back, and anterior neck. Photo-exposed areas of the dorsal hands also are commonplace for both SLE and DM. Clinical manifestations of PCD are limited to sun-exposed areas of the body, specifically those that come into contact with photoallergic triggers.3 Manifestations of PCD can include pruritic eczematous eruptions resembling those of contact dermatitis 1 to 2 days after sun exposure.1
Photocontact dermatitis represents a specific sensitization via contact or oral ingestion acquired prior to sunlight exposure. It can be broken down into 2 distinct subtypes: photoallergic and photoirritant dermatitis, dependent on whether an allergic or irritant reaction is invoked.2 Plants are known to be a common trigger of photoirritant reactions, while extrinsic triggers include psoralens and medications such as tetracycline antibiotics or sulfonamides. Photoallergic reactions commonly can be caused by topical application of sunscreen or medications, namely nonsteroidal anti-inflammatory drugs.2 Clinical manifestations that may point to photoirritant dermatitis include a photodistributed eruption and classic morphology showing erythema and edema with bullae present in severe cases. These can be contrasted with the clinical manifestations of photoallergic reactions, which usually do not correlate to sun-exposed areas and consist of a monomorphous distribution pattern similar to that of eczema. Although there are distinguishing features of both subtypes of PCD, the overlapping clinical features can mimic those of solar urticaria, PMLE, cutaneous lupus erythematosus, and more systemic conditions such as SLE and DM.7
Systemic lupus erythematosus is associated with a broad range of cutaneous manifestations.8 Exposure to UV radiation is a common trigger for lupus and has the propensity to cause a malar (butterfly) rash that covers the cheeks and nasal bridge but classically spares the nasolabial folds. The rash may display confluent reddish-purple discoloration with papules and/or edema and typically is present at diagnosis in 40% to 52% of patients with SLE.8 Discoid lupus erythematosus, one of the most common cutaneous forms of lupus, manifests with various-sized coin-shaped plaques with adherent follicular hyperkeratosis and plugging. These lesions usually develop on the face, scalp, and ears but also may appear in non–sun-exposed areas.8 Dermatomyositis can manifest with photodistributed erythema affecting classic areas such as the upper back (shawl sign), anterior neck and upper chest (V-sign), and a malar rash similar to that seen in lupus, though DM classically does not spare the nasolabial folds.8,9
Because SLE and DM manifest with photodistributed rashes, it can be difficult to distinguish them from the classic symptoms of photoirritant dermatitis.9 Thus, it is imperative that providers have a high clinical index of suspicion when dealing with patients of similar presentations, as the treatment regimens vastly differ. Approaching the patient with a thorough medical history review, review of systems, biopsy (including immunofluorescence), and appropriate laboratory workup may aid in excluding more complex differential diagnoses such as SLE and DM.
Metabolic and genetic photodermatoses are more rare but can include conditions such as porphyria cutanea tarda and xeroderma pigmentosum, both of which demonstrate fragile skin, slow wound healing, and bullae on photo-exposed skin.1 Although the manifestations can be similar in these systemic conditions, they are caused by very different mechanisms. Porphyria cutanea tarda is caused by deficiencies in enzymes involved in the heme synthesis pathway, whereas xeroderma pigmentosum is caused by an alteration in DNA repair mechanisms.7
Prevalence and the Need for Standardized Testing
Most practicing dermatologists see cases of PCD due to its multiple causative agents; however, little is known about its overall prevalence. The incidence of PCD is fairly low in the general population, but this may be due to its clinical diagnosis, which excludes diagnostic testing such as phototesting and photopatch testing.10 While the incidence of photoallergic contact dermatitis also is fairly unknown, the inception of testing modalities has allowed statistics to be drawn. Research conducted in the United States has disclosed that the incidence of photoallergic contact dermatitis in individuals with a history of a prior photosensitivity eruption is approximately 10% to 20%.10 The development of guidelines and a registry for photopatch testing would aid in a greater understanding of the incidence of PCD and overall consistency of diagnosis.7 Regardless of this lack of consensus, these conditions can be properly managed and prevented if recognized clinically, while newer testing modalities would allow for confirmation of the diagnosis. It is important that any patient presenting with a history of photosensitivity be seen as a candidate for photopatch testing, especially today, as the general population is increasingly exposed to new chemicals entering the market and new social trends.7,10
Diagnosis and Treatment
It is important to consider a detailed history, including the timing, location, duration, family history, and seasonal variation of suspected photodermatoses. A thorough skin examination that takes note of the specific areas affected, morphology, and involvement of the rash or lesions can be helpful.1 Further diagnostic testing such as phototesting and photopatch testing can be employed and is especially important when distinguishing photoallergy from phototoxicity.11 Phototesting involves exposing the patient’s skin to different doses of UVA, UVB, and visible light, followed by an immediate clinical reading of the results and then a delayed reading conducted after 24 hours.1 Photopatch testing involves the application of 2 sets of identical photoallergens to prepped skin (typically cleansed with isopropyl alcohol), with one being irradiated with UVA after 24 hours and one serving as the control. A clinical assessment is conducted at 24 hours and repeated 7 days later.1 In photodermatoses, a visible reaction can be appreciated on the treatment arm while the control arm remains clear. When both sides reveal a visible reaction, this is more indicative of a light-independent allergic contact dermatitis.1
Photodermatoses occur only if there has been a specific sensitization, and therefore it is important to work with the patient to discover any new products that have been introduced into their regimen. Though many photosensitizers in personal care products (eg, antiseptics in soap and topical creams) have been discontinued, certain allergenic ingredients may remain.12 It also is important to note that sensitization to a substance that previously was not a known allergen for a particular patient can occur later in life. Avoiding further sun exposure can rapidly improve the dermatitis, and it is possible for spontaneous remission without further intervention; however, as photoallergic reactions can cause severely pruritic skin lesions, the mainstay of symptomatic treatment consists of topical corticosteroids. Oral and topical antihistamines may help alleviate the pruritus but should not be heavily relied on as this can lead to medication resistance and diminishing efficacy.3 Use of short-term oral steroids also may be considered for rapid improvement of symptoms when the patient is in moderate distress and there are no contraindications. By identifying a temporal association between the introduction of new products and the emergence of dermatitis, it may be possible to identify the causative agent. The patient should promptly discontinue the suspected agent and remain under close observation by the clinician for any further eruptions, especially following additional sun exposure.
Prevention Strategies
In the case of PCD, prevention is key. As PCD indicates a photoallergy, it is important to inform patients that the allergy will persist for a lifetime, much like in contact dermatitis; therefore, the causative agent should be avoided indefinitely.3 Patients with PCD should make intentional efforts to read ingredient lists when purchasing new personal care products to ensure they do not contain the specific causative allergen if one has been identified. Further steps should be taken to ensure proper photoprotection, including use of dense clothing and sunscreen with UVA and UVB filters (broad spectrum).3 It has also been suggested that utilizing sunscreen with ectoin, an amino acid–derived molecule, may result in increased protection against UVA-induced photodermatoses.13
Final Thoughts
Photodermatoses are a group of skin diseases caused by exposure to UV radiation. Photocontact dermatitis/photoallergy is a form of allergic contact dermatitis that results from exposure to an allergen, whether topical, oral, or environmental. The allergen is activated by exposure to UV radiation to sensitize the allergic response, resulting in a rash characterized by confluent erythematous patches or plaques, papular vesicles, and rarely blisters.3 Photocontact dermatitis, although rare, is an important differential diagnosis to consider when the presenting rash is restricted to sun-exposed areas of the skin such as the arms, legs, neck, and face. Diagnosis remains a challenge; however, new testing modalities such as photopatch testing may open the door for further confirmation and aid in proper diagnosis leading to earlier treatment times for patients. It is recommended that the clinician and patient work together to identify the possible causative agent to prevent further eruptions.
- Santoro FA, Lim HW. Update on photodermatoses. Semin Cutan Med Surg. 2011;30:229-238.
- Gimenez-Arnau A, Maurer M, De La Cuadra J, et al. Immediate contact skin reactions, an update of contact urticaria, contact urticaria syndrome and protein contact dermatitis—“a never ending story.” Eur J Dermatol. 2010;20:555-562.
- Lehmann P, Schwarz T. Photodermatoses: diagnosis and treatment. Dtsch Arztebl Int. 2011;108:135-141.
- Victor FC, Cohen DE, Soter NA. A 20-year analysis of previous and emerging allergens that elicit photoallergic contact dermatitis. J Am Acad Dermatol. 2010;62:605-610.
- Fenticlor (Code 65671). National Cancer Institute EVS Explore. Accessed October 28, 2025. https://ncithesaurus.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCIThesaurus&ns=ncit&code=C65671
- Elmets CA. Photosensitivity disorders (photodermatoses): clinical manifestations, diagnosis, and treatment. UptoDate. Updated February 23, 2023. Accessed October 28, 2025. https://www.uptodate.com/contents/photosensitivity-disorders-photodermatoses-clinical-manifestations-diagnosis-and-treatment
- Snyder M, Turrentine JE, Cruz PD Jr. Photocontact dermatitis and its clinical mimics: an overview for the allergist. Clin Rev Allergy Immunol. 2019;56:32-40.
- Cooper EE, Pisano CE, Shapiro SC. Cutaneous manifestations of “lupus”: systemic lupus erythematosus and beyond. Int J Rheumatol. 2021;2021:6610509.
- Christopher-Stine L, Amato AA, Vleugels RA. Diagnosis and differential diagnosis of dermatomyositis and polymyositis in adults. UptoDate. Updated March 3, 2025. Accessed October 28, 2025. https://www.uptodate.com/contents/diagnosis-and-differential-diagnosis-of-dermatomyositis-and-polymyositis-in-adults?search=Diagnosis%20and%20differential%20diagnosis%20of%20dermatomyositis%20and%20polymyositis%20in%20adults&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Deleo VA. Photocontact dermatitis. Dermatol Ther. 2004;17:279-288.
- Gonçalo M. Photopatch testing. In: Johansen J, Frosch P, Lepoittevin JP, eds. Contact Dermatitis. Springer; 2011:519-531.
- Enta T. Dermacase. Contact photodermatitis. Can Fam Physician. 1995;41:577,586-587.
- Duteil L, Queille-Roussel C, Aladren S, et al. Prevention of polymophic light eruption afforded by a very high broad-spectrum protection sunscreen containing ectoin. Dermatol Ther (Heidelb). 2022;12:1603-1613.
Photosensitivity refers to clinical manifestations arising from exposure to sunlight. Photodermatoses encompass a group of skin diseases caused by varying degrees of radiation exposure, including UV radiation and visible light. Photodermatoses can be categorized into 5 main types: primary, exogenous, photoexacerbated, metabolic, and genetic.1 The clinical features of photodermatoses vary depending on the underlying cause but often include pruritic flares, wheals, or dermatitis on sun-exposed areas of the skin.2 While photodermatoses typically are not life threatening, they can greatly impact patients’ quality of life. It is crucial to emphasize the importance of photoprotection and sunlight avoidance to patients as preventive measures against the manifestations of these skin diseases. Furthermore, we present a case of photocontact dermatitis (PCD) and discuss common causative agents, diagnostic mimickers, and treatment options.
Case Report
A 51-year-old woman with no relevant medical history presented to the dermatology clinic with a rash on the neck and under the eyes of 6 days’ duration. The rash was intermittently pruritic but otherwise asymptomatic. The patient reported that she had spent extensive time on the golf course the day of the rash onset and noted that a similar rash had occurred one other time 2 to 3 months prior, also following a prolonged period on the golf course. She had been using over-the-counter fexofenadine 180 mg and over-the-counter lidocaine spray for symptom relief.
Upon physical examination, erythematous patches were appreciated in a photodistributed pattern on the arms, legs, neck, face, and chest—areas that were not covered by clothing (Figures 1-3). Due to the distribution and morphology of the erythematous patches along with clinical course of onset following exposure to various environmental agents including pesticides, herbicides, oak, and pollen, a diagnosis of PCD was made. The patient was prescribed hydrocortisone cream 2.5%, fluticasone propionate cream 0.05%, and methylprednisolone in addition to the antihistamine. Improvement was noted after 3 days with complete resolution of the skin manifestations. She was counseled on wearing clothing with a universal protection factor rating of 50+ when on the golf course and when sun exposure is expected for an extended period of time.



Causative Agents
Photodermatoses are caused by antigenic substances that lead to photosensitization acquired by either contact or oral ingestion with subsequent sensitization to UV radiation. Halogenated salicylanilide, fenticlor, hexachlorophene, bithionol and, in rare cases, sunscreens, have been reported as triggers.3 In a study performed in 2010, sunscreens, antimicrobial agents, medications, fragrances, plants/plant derivatives, and pesticides were the most commonly reported offending agents listed from highest to lowest frequency. Of the antimicrobial agents, fenticlor, a topical antimicrobial and antifungal that is now mostly used in veterinary medicine, was the most common culprit, causing 60% of cases.4,5
Clinical Manifestations
Clinical manifestations of photodermatoses vary depending upon the specific type of reaction. Examples of primary photodermatoses include polymorphous light eruption (PMLE) and solar urticaria. The cardinal symptoms of PMLE consist of severely pruritic skin lesions that can have macular, papular, papulovesicular, urticarial, multiformelike, and plaquelike variants that develop hours to days after sun exposure.3 Conversely, solar urticaria commonly develops more abruptly, with indurated plaques and wheals appearing on the arms and neck within 30 minutes of sun exposure. The lesions typically resolve within 24 hours.1
Examples of the exogenous subtype include drug-induced photosensitivity, PCD, and pseudoporphyria, with the common clinical presentation of eruption following contact with the causative agent. Drug-induced photosensitivity primarily manifests as a severe sunburnlike rash commonly caused by systemic drugs such as tetracyclines. Photocontact dermatitis is limited to sun-exposed areas of the skin and is caused by a reactive irritant such as chemicals or topical creams. Pseudoporphyria, usually caused by nonsteroidal anti-inflammatory drugs, can manifest with skin fragility and subepidermal blisters.6
Photoexacerbated photodermatoses encompass a variety of conditions ranging from hyperpigmentation disorders such as melasma to autoimmune conditions such as systemic lupus erythematosus (SLE) and dermatomyositis (DM). Common clinical features of these diseases include photodistributed erythema, often involving the cheeks, upper back, and anterior neck. Photo-exposed areas of the dorsal hands also are commonplace for both SLE and DM. Clinical manifestations of PCD are limited to sun-exposed areas of the body, specifically those that come into contact with photoallergic triggers.3 Manifestations of PCD can include pruritic eczematous eruptions resembling those of contact dermatitis 1 to 2 days after sun exposure.1
Photocontact dermatitis represents a specific sensitization via contact or oral ingestion acquired prior to sunlight exposure. It can be broken down into 2 distinct subtypes: photoallergic and photoirritant dermatitis, dependent on whether an allergic or irritant reaction is invoked.2 Plants are known to be a common trigger of photoirritant reactions, while extrinsic triggers include psoralens and medications such as tetracycline antibiotics or sulfonamides. Photoallergic reactions commonly can be caused by topical application of sunscreen or medications, namely nonsteroidal anti-inflammatory drugs.2 Clinical manifestations that may point to photoirritant dermatitis include a photodistributed eruption and classic morphology showing erythema and edema with bullae present in severe cases. These can be contrasted with the clinical manifestations of photoallergic reactions, which usually do not correlate to sun-exposed areas and consist of a monomorphous distribution pattern similar to that of eczema. Although there are distinguishing features of both subtypes of PCD, the overlapping clinical features can mimic those of solar urticaria, PMLE, cutaneous lupus erythematosus, and more systemic conditions such as SLE and DM.7
Systemic lupus erythematosus is associated with a broad range of cutaneous manifestations.8 Exposure to UV radiation is a common trigger for lupus and has the propensity to cause a malar (butterfly) rash that covers the cheeks and nasal bridge but classically spares the nasolabial folds. The rash may display confluent reddish-purple discoloration with papules and/or edema and typically is present at diagnosis in 40% to 52% of patients with SLE.8 Discoid lupus erythematosus, one of the most common cutaneous forms of lupus, manifests with various-sized coin-shaped plaques with adherent follicular hyperkeratosis and plugging. These lesions usually develop on the face, scalp, and ears but also may appear in non–sun-exposed areas.8 Dermatomyositis can manifest with photodistributed erythema affecting classic areas such as the upper back (shawl sign), anterior neck and upper chest (V-sign), and a malar rash similar to that seen in lupus, though DM classically does not spare the nasolabial folds.8,9
Because SLE and DM manifest with photodistributed rashes, it can be difficult to distinguish them from the classic symptoms of photoirritant dermatitis.9 Thus, it is imperative that providers have a high clinical index of suspicion when dealing with patients of similar presentations, as the treatment regimens vastly differ. Approaching the patient with a thorough medical history review, review of systems, biopsy (including immunofluorescence), and appropriate laboratory workup may aid in excluding more complex differential diagnoses such as SLE and DM.
Metabolic and genetic photodermatoses are more rare but can include conditions such as porphyria cutanea tarda and xeroderma pigmentosum, both of which demonstrate fragile skin, slow wound healing, and bullae on photo-exposed skin.1 Although the manifestations can be similar in these systemic conditions, they are caused by very different mechanisms. Porphyria cutanea tarda is caused by deficiencies in enzymes involved in the heme synthesis pathway, whereas xeroderma pigmentosum is caused by an alteration in DNA repair mechanisms.7
Prevalence and the Need for Standardized Testing
Most practicing dermatologists see cases of PCD due to its multiple causative agents; however, little is known about its overall prevalence. The incidence of PCD is fairly low in the general population, but this may be due to its clinical diagnosis, which excludes diagnostic testing such as phototesting and photopatch testing.10 While the incidence of photoallergic contact dermatitis also is fairly unknown, the inception of testing modalities has allowed statistics to be drawn. Research conducted in the United States has disclosed that the incidence of photoallergic contact dermatitis in individuals with a history of a prior photosensitivity eruption is approximately 10% to 20%.10 The development of guidelines and a registry for photopatch testing would aid in a greater understanding of the incidence of PCD and overall consistency of diagnosis.7 Regardless of this lack of consensus, these conditions can be properly managed and prevented if recognized clinically, while newer testing modalities would allow for confirmation of the diagnosis. It is important that any patient presenting with a history of photosensitivity be seen as a candidate for photopatch testing, especially today, as the general population is increasingly exposed to new chemicals entering the market and new social trends.7,10
Diagnosis and Treatment
It is important to consider a detailed history, including the timing, location, duration, family history, and seasonal variation of suspected photodermatoses. A thorough skin examination that takes note of the specific areas affected, morphology, and involvement of the rash or lesions can be helpful.1 Further diagnostic testing such as phototesting and photopatch testing can be employed and is especially important when distinguishing photoallergy from phototoxicity.11 Phototesting involves exposing the patient’s skin to different doses of UVA, UVB, and visible light, followed by an immediate clinical reading of the results and then a delayed reading conducted after 24 hours.1 Photopatch testing involves the application of 2 sets of identical photoallergens to prepped skin (typically cleansed with isopropyl alcohol), with one being irradiated with UVA after 24 hours and one serving as the control. A clinical assessment is conducted at 24 hours and repeated 7 days later.1 In photodermatoses, a visible reaction can be appreciated on the treatment arm while the control arm remains clear. When both sides reveal a visible reaction, this is more indicative of a light-independent allergic contact dermatitis.1
Photodermatoses occur only if there has been a specific sensitization, and therefore it is important to work with the patient to discover any new products that have been introduced into their regimen. Though many photosensitizers in personal care products (eg, antiseptics in soap and topical creams) have been discontinued, certain allergenic ingredients may remain.12 It also is important to note that sensitization to a substance that previously was not a known allergen for a particular patient can occur later in life. Avoiding further sun exposure can rapidly improve the dermatitis, and it is possible for spontaneous remission without further intervention; however, as photoallergic reactions can cause severely pruritic skin lesions, the mainstay of symptomatic treatment consists of topical corticosteroids. Oral and topical antihistamines may help alleviate the pruritus but should not be heavily relied on as this can lead to medication resistance and diminishing efficacy.3 Use of short-term oral steroids also may be considered for rapid improvement of symptoms when the patient is in moderate distress and there are no contraindications. By identifying a temporal association between the introduction of new products and the emergence of dermatitis, it may be possible to identify the causative agent. The patient should promptly discontinue the suspected agent and remain under close observation by the clinician for any further eruptions, especially following additional sun exposure.
Prevention Strategies
In the case of PCD, prevention is key. As PCD indicates a photoallergy, it is important to inform patients that the allergy will persist for a lifetime, much like in contact dermatitis; therefore, the causative agent should be avoided indefinitely.3 Patients with PCD should make intentional efforts to read ingredient lists when purchasing new personal care products to ensure they do not contain the specific causative allergen if one has been identified. Further steps should be taken to ensure proper photoprotection, including use of dense clothing and sunscreen with UVA and UVB filters (broad spectrum).3 It has also been suggested that utilizing sunscreen with ectoin, an amino acid–derived molecule, may result in increased protection against UVA-induced photodermatoses.13
Final Thoughts
Photodermatoses are a group of skin diseases caused by exposure to UV radiation. Photocontact dermatitis/photoallergy is a form of allergic contact dermatitis that results from exposure to an allergen, whether topical, oral, or environmental. The allergen is activated by exposure to UV radiation to sensitize the allergic response, resulting in a rash characterized by confluent erythematous patches or plaques, papular vesicles, and rarely blisters.3 Photocontact dermatitis, although rare, is an important differential diagnosis to consider when the presenting rash is restricted to sun-exposed areas of the skin such as the arms, legs, neck, and face. Diagnosis remains a challenge; however, new testing modalities such as photopatch testing may open the door for further confirmation and aid in proper diagnosis leading to earlier treatment times for patients. It is recommended that the clinician and patient work together to identify the possible causative agent to prevent further eruptions.
Photosensitivity refers to clinical manifestations arising from exposure to sunlight. Photodermatoses encompass a group of skin diseases caused by varying degrees of radiation exposure, including UV radiation and visible light. Photodermatoses can be categorized into 5 main types: primary, exogenous, photoexacerbated, metabolic, and genetic.1 The clinical features of photodermatoses vary depending on the underlying cause but often include pruritic flares, wheals, or dermatitis on sun-exposed areas of the skin.2 While photodermatoses typically are not life threatening, they can greatly impact patients’ quality of life. It is crucial to emphasize the importance of photoprotection and sunlight avoidance to patients as preventive measures against the manifestations of these skin diseases. Furthermore, we present a case of photocontact dermatitis (PCD) and discuss common causative agents, diagnostic mimickers, and treatment options.
Case Report
A 51-year-old woman with no relevant medical history presented to the dermatology clinic with a rash on the neck and under the eyes of 6 days’ duration. The rash was intermittently pruritic but otherwise asymptomatic. The patient reported that she had spent extensive time on the golf course the day of the rash onset and noted that a similar rash had occurred one other time 2 to 3 months prior, also following a prolonged period on the golf course. She had been using over-the-counter fexofenadine 180 mg and over-the-counter lidocaine spray for symptom relief.
Upon physical examination, erythematous patches were appreciated in a photodistributed pattern on the arms, legs, neck, face, and chest—areas that were not covered by clothing (Figures 1-3). Due to the distribution and morphology of the erythematous patches along with clinical course of onset following exposure to various environmental agents including pesticides, herbicides, oak, and pollen, a diagnosis of PCD was made. The patient was prescribed hydrocortisone cream 2.5%, fluticasone propionate cream 0.05%, and methylprednisolone in addition to the antihistamine. Improvement was noted after 3 days with complete resolution of the skin manifestations. She was counseled on wearing clothing with a universal protection factor rating of 50+ when on the golf course and when sun exposure is expected for an extended period of time.



Causative Agents
Photodermatoses are caused by antigenic substances that lead to photosensitization acquired by either contact or oral ingestion with subsequent sensitization to UV radiation. Halogenated salicylanilide, fenticlor, hexachlorophene, bithionol and, in rare cases, sunscreens, have been reported as triggers.3 In a study performed in 2010, sunscreens, antimicrobial agents, medications, fragrances, plants/plant derivatives, and pesticides were the most commonly reported offending agents listed from highest to lowest frequency. Of the antimicrobial agents, fenticlor, a topical antimicrobial and antifungal that is now mostly used in veterinary medicine, was the most common culprit, causing 60% of cases.4,5
Clinical Manifestations
Clinical manifestations of photodermatoses vary depending upon the specific type of reaction. Examples of primary photodermatoses include polymorphous light eruption (PMLE) and solar urticaria. The cardinal symptoms of PMLE consist of severely pruritic skin lesions that can have macular, papular, papulovesicular, urticarial, multiformelike, and plaquelike variants that develop hours to days after sun exposure.3 Conversely, solar urticaria commonly develops more abruptly, with indurated plaques and wheals appearing on the arms and neck within 30 minutes of sun exposure. The lesions typically resolve within 24 hours.1
Examples of the exogenous subtype include drug-induced photosensitivity, PCD, and pseudoporphyria, with the common clinical presentation of eruption following contact with the causative agent. Drug-induced photosensitivity primarily manifests as a severe sunburnlike rash commonly caused by systemic drugs such as tetracyclines. Photocontact dermatitis is limited to sun-exposed areas of the skin and is caused by a reactive irritant such as chemicals or topical creams. Pseudoporphyria, usually caused by nonsteroidal anti-inflammatory drugs, can manifest with skin fragility and subepidermal blisters.6
Photoexacerbated photodermatoses encompass a variety of conditions ranging from hyperpigmentation disorders such as melasma to autoimmune conditions such as systemic lupus erythematosus (SLE) and dermatomyositis (DM). Common clinical features of these diseases include photodistributed erythema, often involving the cheeks, upper back, and anterior neck. Photo-exposed areas of the dorsal hands also are commonplace for both SLE and DM. Clinical manifestations of PCD are limited to sun-exposed areas of the body, specifically those that come into contact with photoallergic triggers.3 Manifestations of PCD can include pruritic eczematous eruptions resembling those of contact dermatitis 1 to 2 days after sun exposure.1
Photocontact dermatitis represents a specific sensitization via contact or oral ingestion acquired prior to sunlight exposure. It can be broken down into 2 distinct subtypes: photoallergic and photoirritant dermatitis, dependent on whether an allergic or irritant reaction is invoked.2 Plants are known to be a common trigger of photoirritant reactions, while extrinsic triggers include psoralens and medications such as tetracycline antibiotics or sulfonamides. Photoallergic reactions commonly can be caused by topical application of sunscreen or medications, namely nonsteroidal anti-inflammatory drugs.2 Clinical manifestations that may point to photoirritant dermatitis include a photodistributed eruption and classic morphology showing erythema and edema with bullae present in severe cases. These can be contrasted with the clinical manifestations of photoallergic reactions, which usually do not correlate to sun-exposed areas and consist of a monomorphous distribution pattern similar to that of eczema. Although there are distinguishing features of both subtypes of PCD, the overlapping clinical features can mimic those of solar urticaria, PMLE, cutaneous lupus erythematosus, and more systemic conditions such as SLE and DM.7
Systemic lupus erythematosus is associated with a broad range of cutaneous manifestations.8 Exposure to UV radiation is a common trigger for lupus and has the propensity to cause a malar (butterfly) rash that covers the cheeks and nasal bridge but classically spares the nasolabial folds. The rash may display confluent reddish-purple discoloration with papules and/or edema and typically is present at diagnosis in 40% to 52% of patients with SLE.8 Discoid lupus erythematosus, one of the most common cutaneous forms of lupus, manifests with various-sized coin-shaped plaques with adherent follicular hyperkeratosis and plugging. These lesions usually develop on the face, scalp, and ears but also may appear in non–sun-exposed areas.8 Dermatomyositis can manifest with photodistributed erythema affecting classic areas such as the upper back (shawl sign), anterior neck and upper chest (V-sign), and a malar rash similar to that seen in lupus, though DM classically does not spare the nasolabial folds.8,9
Because SLE and DM manifest with photodistributed rashes, it can be difficult to distinguish them from the classic symptoms of photoirritant dermatitis.9 Thus, it is imperative that providers have a high clinical index of suspicion when dealing with patients of similar presentations, as the treatment regimens vastly differ. Approaching the patient with a thorough medical history review, review of systems, biopsy (including immunofluorescence), and appropriate laboratory workup may aid in excluding more complex differential diagnoses such as SLE and DM.
Metabolic and genetic photodermatoses are more rare but can include conditions such as porphyria cutanea tarda and xeroderma pigmentosum, both of which demonstrate fragile skin, slow wound healing, and bullae on photo-exposed skin.1 Although the manifestations can be similar in these systemic conditions, they are caused by very different mechanisms. Porphyria cutanea tarda is caused by deficiencies in enzymes involved in the heme synthesis pathway, whereas xeroderma pigmentosum is caused by an alteration in DNA repair mechanisms.7
Prevalence and the Need for Standardized Testing
Most practicing dermatologists see cases of PCD due to its multiple causative agents; however, little is known about its overall prevalence. The incidence of PCD is fairly low in the general population, but this may be due to its clinical diagnosis, which excludes diagnostic testing such as phototesting and photopatch testing.10 While the incidence of photoallergic contact dermatitis also is fairly unknown, the inception of testing modalities has allowed statistics to be drawn. Research conducted in the United States has disclosed that the incidence of photoallergic contact dermatitis in individuals with a history of a prior photosensitivity eruption is approximately 10% to 20%.10 The development of guidelines and a registry for photopatch testing would aid in a greater understanding of the incidence of PCD and overall consistency of diagnosis.7 Regardless of this lack of consensus, these conditions can be properly managed and prevented if recognized clinically, while newer testing modalities would allow for confirmation of the diagnosis. It is important that any patient presenting with a history of photosensitivity be seen as a candidate for photopatch testing, especially today, as the general population is increasingly exposed to new chemicals entering the market and new social trends.7,10
Diagnosis and Treatment
It is important to consider a detailed history, including the timing, location, duration, family history, and seasonal variation of suspected photodermatoses. A thorough skin examination that takes note of the specific areas affected, morphology, and involvement of the rash or lesions can be helpful.1 Further diagnostic testing such as phototesting and photopatch testing can be employed and is especially important when distinguishing photoallergy from phototoxicity.11 Phototesting involves exposing the patient’s skin to different doses of UVA, UVB, and visible light, followed by an immediate clinical reading of the results and then a delayed reading conducted after 24 hours.1 Photopatch testing involves the application of 2 sets of identical photoallergens to prepped skin (typically cleansed with isopropyl alcohol), with one being irradiated with UVA after 24 hours and one serving as the control. A clinical assessment is conducted at 24 hours and repeated 7 days later.1 In photodermatoses, a visible reaction can be appreciated on the treatment arm while the control arm remains clear. When both sides reveal a visible reaction, this is more indicative of a light-independent allergic contact dermatitis.1
Photodermatoses occur only if there has been a specific sensitization, and therefore it is important to work with the patient to discover any new products that have been introduced into their regimen. Though many photosensitizers in personal care products (eg, antiseptics in soap and topical creams) have been discontinued, certain allergenic ingredients may remain.12 It also is important to note that sensitization to a substance that previously was not a known allergen for a particular patient can occur later in life. Avoiding further sun exposure can rapidly improve the dermatitis, and it is possible for spontaneous remission without further intervention; however, as photoallergic reactions can cause severely pruritic skin lesions, the mainstay of symptomatic treatment consists of topical corticosteroids. Oral and topical antihistamines may help alleviate the pruritus but should not be heavily relied on as this can lead to medication resistance and diminishing efficacy.3 Use of short-term oral steroids also may be considered for rapid improvement of symptoms when the patient is in moderate distress and there are no contraindications. By identifying a temporal association between the introduction of new products and the emergence of dermatitis, it may be possible to identify the causative agent. The patient should promptly discontinue the suspected agent and remain under close observation by the clinician for any further eruptions, especially following additional sun exposure.
Prevention Strategies
In the case of PCD, prevention is key. As PCD indicates a photoallergy, it is important to inform patients that the allergy will persist for a lifetime, much like in contact dermatitis; therefore, the causative agent should be avoided indefinitely.3 Patients with PCD should make intentional efforts to read ingredient lists when purchasing new personal care products to ensure they do not contain the specific causative allergen if one has been identified. Further steps should be taken to ensure proper photoprotection, including use of dense clothing and sunscreen with UVA and UVB filters (broad spectrum).3 It has also been suggested that utilizing sunscreen with ectoin, an amino acid–derived molecule, may result in increased protection against UVA-induced photodermatoses.13
Final Thoughts
Photodermatoses are a group of skin diseases caused by exposure to UV radiation. Photocontact dermatitis/photoallergy is a form of allergic contact dermatitis that results from exposure to an allergen, whether topical, oral, or environmental. The allergen is activated by exposure to UV radiation to sensitize the allergic response, resulting in a rash characterized by confluent erythematous patches or plaques, papular vesicles, and rarely blisters.3 Photocontact dermatitis, although rare, is an important differential diagnosis to consider when the presenting rash is restricted to sun-exposed areas of the skin such as the arms, legs, neck, and face. Diagnosis remains a challenge; however, new testing modalities such as photopatch testing may open the door for further confirmation and aid in proper diagnosis leading to earlier treatment times for patients. It is recommended that the clinician and patient work together to identify the possible causative agent to prevent further eruptions.
- Santoro FA, Lim HW. Update on photodermatoses. Semin Cutan Med Surg. 2011;30:229-238.
- Gimenez-Arnau A, Maurer M, De La Cuadra J, et al. Immediate contact skin reactions, an update of contact urticaria, contact urticaria syndrome and protein contact dermatitis—“a never ending story.” Eur J Dermatol. 2010;20:555-562.
- Lehmann P, Schwarz T. Photodermatoses: diagnosis and treatment. Dtsch Arztebl Int. 2011;108:135-141.
- Victor FC, Cohen DE, Soter NA. A 20-year analysis of previous and emerging allergens that elicit photoallergic contact dermatitis. J Am Acad Dermatol. 2010;62:605-610.
- Fenticlor (Code 65671). National Cancer Institute EVS Explore. Accessed October 28, 2025. https://ncithesaurus.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCIThesaurus&ns=ncit&code=C65671
- Elmets CA. Photosensitivity disorders (photodermatoses): clinical manifestations, diagnosis, and treatment. UptoDate. Updated February 23, 2023. Accessed October 28, 2025. https://www.uptodate.com/contents/photosensitivity-disorders-photodermatoses-clinical-manifestations-diagnosis-and-treatment
- Snyder M, Turrentine JE, Cruz PD Jr. Photocontact dermatitis and its clinical mimics: an overview for the allergist. Clin Rev Allergy Immunol. 2019;56:32-40.
- Cooper EE, Pisano CE, Shapiro SC. Cutaneous manifestations of “lupus”: systemic lupus erythematosus and beyond. Int J Rheumatol. 2021;2021:6610509.
- Christopher-Stine L, Amato AA, Vleugels RA. Diagnosis and differential diagnosis of dermatomyositis and polymyositis in adults. UptoDate. Updated March 3, 2025. Accessed October 28, 2025. https://www.uptodate.com/contents/diagnosis-and-differential-diagnosis-of-dermatomyositis-and-polymyositis-in-adults?search=Diagnosis%20and%20differential%20diagnosis%20of%20dermatomyositis%20and%20polymyositis%20in%20adults&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Deleo VA. Photocontact dermatitis. Dermatol Ther. 2004;17:279-288.
- Gonçalo M. Photopatch testing. In: Johansen J, Frosch P, Lepoittevin JP, eds. Contact Dermatitis. Springer; 2011:519-531.
- Enta T. Dermacase. Contact photodermatitis. Can Fam Physician. 1995;41:577,586-587.
- Duteil L, Queille-Roussel C, Aladren S, et al. Prevention of polymophic light eruption afforded by a very high broad-spectrum protection sunscreen containing ectoin. Dermatol Ther (Heidelb). 2022;12:1603-1613.
- Santoro FA, Lim HW. Update on photodermatoses. Semin Cutan Med Surg. 2011;30:229-238.
- Gimenez-Arnau A, Maurer M, De La Cuadra J, et al. Immediate contact skin reactions, an update of contact urticaria, contact urticaria syndrome and protein contact dermatitis—“a never ending story.” Eur J Dermatol. 2010;20:555-562.
- Lehmann P, Schwarz T. Photodermatoses: diagnosis and treatment. Dtsch Arztebl Int. 2011;108:135-141.
- Victor FC, Cohen DE, Soter NA. A 20-year analysis of previous and emerging allergens that elicit photoallergic contact dermatitis. J Am Acad Dermatol. 2010;62:605-610.
- Fenticlor (Code 65671). National Cancer Institute EVS Explore. Accessed October 28, 2025. https://ncithesaurus.nci.nih.gov/ncitbrowser/ConceptReport.jsp?dictionary=NCIThesaurus&ns=ncit&code=C65671
- Elmets CA. Photosensitivity disorders (photodermatoses): clinical manifestations, diagnosis, and treatment. UptoDate. Updated February 23, 2023. Accessed October 28, 2025. https://www.uptodate.com/contents/photosensitivity-disorders-photodermatoses-clinical-manifestations-diagnosis-and-treatment
- Snyder M, Turrentine JE, Cruz PD Jr. Photocontact dermatitis and its clinical mimics: an overview for the allergist. Clin Rev Allergy Immunol. 2019;56:32-40.
- Cooper EE, Pisano CE, Shapiro SC. Cutaneous manifestations of “lupus”: systemic lupus erythematosus and beyond. Int J Rheumatol. 2021;2021:6610509.
- Christopher-Stine L, Amato AA, Vleugels RA. Diagnosis and differential diagnosis of dermatomyositis and polymyositis in adults. UptoDate. Updated March 3, 2025. Accessed October 28, 2025. https://www.uptodate.com/contents/diagnosis-and-differential-diagnosis-of-dermatomyositis-and-polymyositis-in-adults?search=Diagnosis%20and%20differential%20diagnosis%20of%20dermatomyositis%20and%20polymyositis%20in%20adults&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1
- Deleo VA. Photocontact dermatitis. Dermatol Ther. 2004;17:279-288.
- Gonçalo M. Photopatch testing. In: Johansen J, Frosch P, Lepoittevin JP, eds. Contact Dermatitis. Springer; 2011:519-531.
- Enta T. Dermacase. Contact photodermatitis. Can Fam Physician. 1995;41:577,586-587.
- Duteil L, Queille-Roussel C, Aladren S, et al. Prevention of polymophic light eruption afforded by a very high broad-spectrum protection sunscreen containing ectoin. Dermatol Ther (Heidelb). 2022;12:1603-1613.
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Photodermatoses: Exploring Clinical Presentations, Causative Factors, Differential Diagnoses, and Treatment Strategies
Practice Points
- It is important to consider photodermatoses in patients presenting with a rash that is restricted to light-exposed areas of the skin, such as the arms, legs, neck, and face.
- The mainstay of treatment consists of topical corticosteroids. Oral antihistamines should not be heavily relied on, but short-term oral steroids may be considered for rapid improvement if symptoms are severe.
- It is important to note that, much like in contact dermatitis, the underlying photoallergy causing photocontact dermatitis will persist for a lifetime.