User login
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
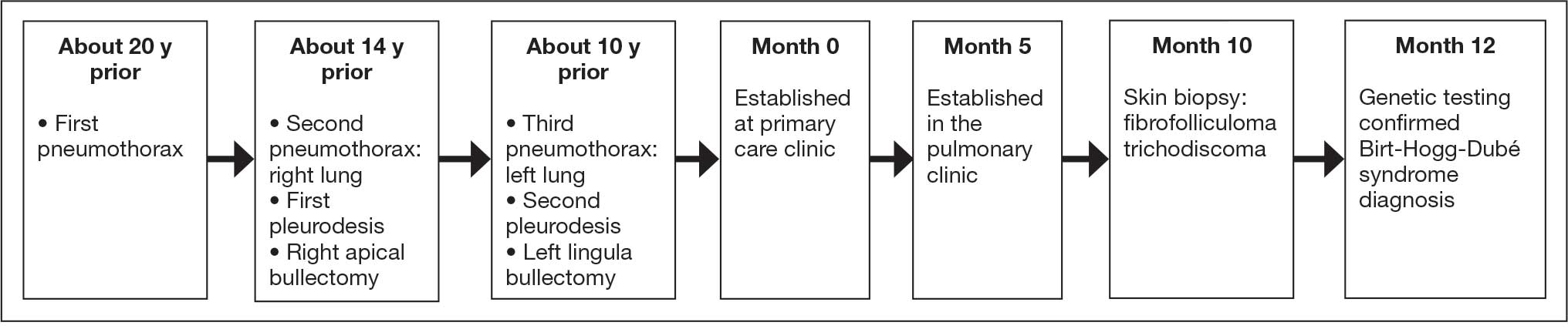
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.
On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
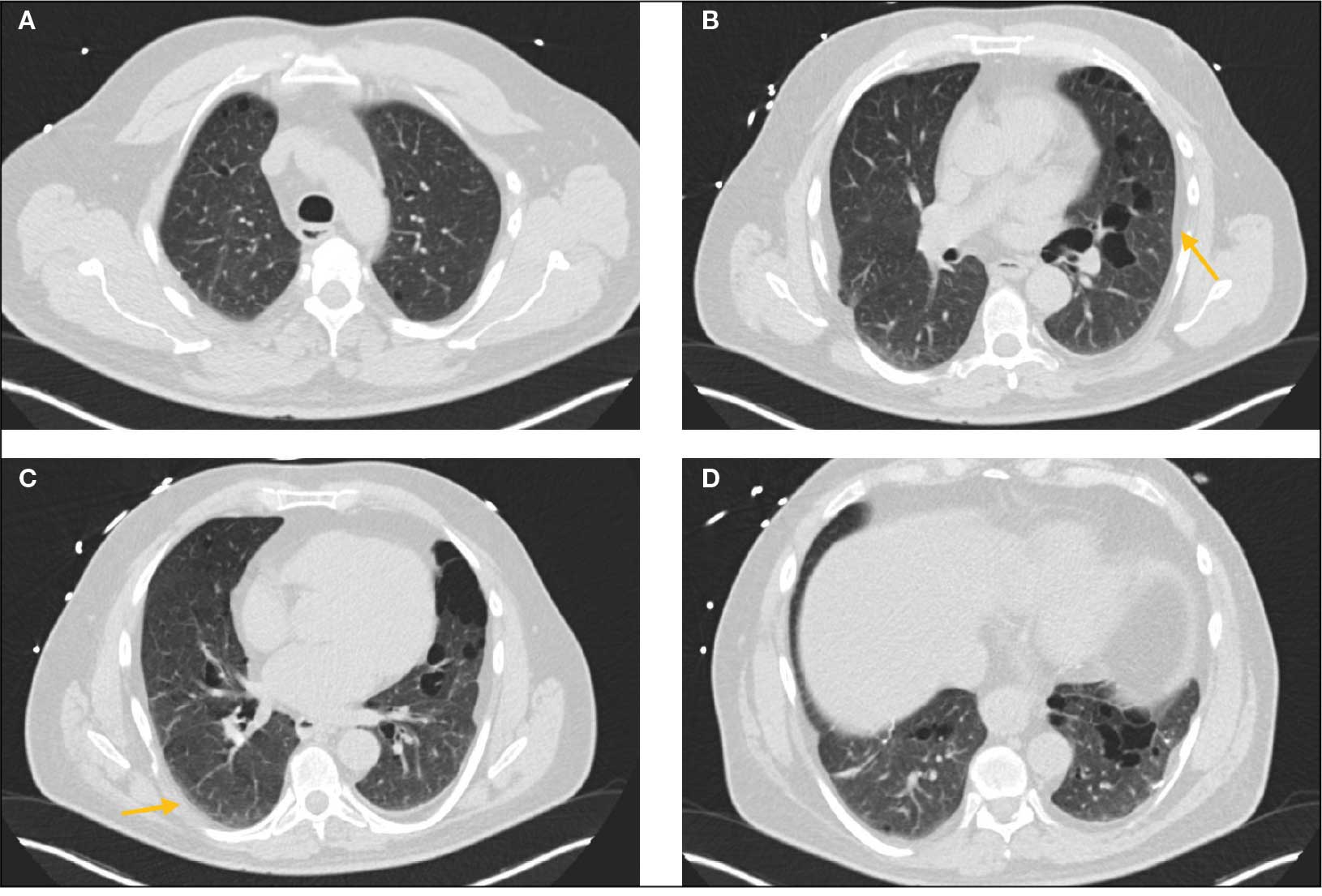
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).
Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
Birt-Hogg-Dubé syndrome (BHD) is an autosomal dominant disease that arises from loss-of-function mutations in the FLCN gene. FLCN encodes folliculin, which is presumed to function as a tumor suppressor, though its precise role is incompletely understood.1,2 BHD is characterized by multiple pulmonary cysts leading to recurrent spontaneous pneumothoraces, cutaneous lesions—specifically fibrofolliculomas—and an increased risk of renal malignancies. Diagnosing BHD is challenging due to the variable presentation of the disease. Some patients may only have cystic lung diseases, while others may not have characteristic skin lesions.3-5 It is important to maintain awareness of BHD, especially when the diagnosis dictates the need for genetic counseling.
Case Presentation
A male veteran in his 60s, who was a lifelong nonsmoker with a history of extensive bullous emphysema and recurrent pneumothoraces, presented to the Veterans Affairs Greater Los Angeles Healthcare System pulmonary clinic while transferring care from a separate institution.
According to the patient, the first pneumothorax episode occurred about 20 years before presentation, followed by a recurrence a few years later after he was diagnosed with emphysema. He underwent pleurodesis of the right lung during his service abroad. Another episode nearly a decade after the first pneumothorax necessitated pleurodesis of the left lung (Figure 1). The patient's family history revealed pulmonary cysts in 1 immediate family member but no history of renal tumors. Notably, his mother passed away at a young age due to tuberculosis.

On physical examination, numerous skin tags and acrochordons on the face were observed, which had been stable for > 30 years. Despite a slow decline in exercise capacity following pleurodesis, the patient could still walk multiple miles daily and climb 3 flights of stairs before needing to rest. Pulmonary function testing (PFT) showed a forced expiratory volume in 1 second (FEV1)/forced vital capacity ratio of 0.84 with reduced FEV1, total lung capacity (TLC), and diffusion capacity for carbon monoxide (DLCO), indicating a mild restrictive ventilatory defect and reduced diffusing capacity.
Laboratory results revealed a normal α-1 antitrypsin level: 133 mg/dL (reference, 83-199 mg/dL), with a Pi*MS phenotype and undetectable antinuclear antibodies. The most recent chest computed tomography (CT) in 2019, displayed paraseptal and centrilobular emphysema, scattered blebs, and scarring consistent with prior pleurodesis procedures (Figure 2).

Genetic testing for the FLCN gene revealed heterozygous pathogenic mutation: c.1285del and p.His429Thrfs*39, which confirmed the diagnosis of BHD. A shave biopsy of a postauricular papular lesion confirmed a histologic pattern of fibrofolliculoma/trichodiscoma.
Follow-up and Outcomes
After confirmation of the BHD diagnosis, the patient was referred to genetic counseling and scheduled for annual magnetic resonance imaging (MRI) of the abdomen and pelvis to screen for renal malignancies. As the patient was able to establish care with a new long-term primary care practitioner in the outpatient setting, he continues regular follow-up visits in the pulmonary clinic with stable respiratory symptoms and no recurrent pneumothoraces thus far.
Discussion
Differential Diagnoses of Cystic Pulmonary Lesions
BHD is an important differential diagnosis to consider in the presentation of diffuse cystic lung diseases. Still, 2 other crucial considerations are pulmonary Langerhans cell histiocytosis (PLCH) and lymphangioleiomyomatosis (LAM), which occur at slightly higher frequencies than BHD.6
One of the first steps in radiographically evaluating cystic lung diseases is to characterize the cysts. The Fleischner Society defines true cysts as a “round parenchymal lucency or low-attenuating area with a well-defined interface with normal lung.”7 Mimics of cystic lesions may include cavitary lung lesions, thick-walled spaces within another area of mass, nodule, or consolidation. Another mimic is a pneumatocele, a pseudocyst that lacks epithelial lining and may be secondary to bacterial pneumonia, pneumocystis infections, trauma, or prior mechanical ventilation.8After characterizing true cysts, different patterns of cystic lesions can also be associated with specific diseases. Cysts in PLCH are commonly more uniform and round, whereas the cysts in LAM may be more irregularly shaped. 9 Cysts in BHD may be larger and predominantly located in basal and paramediastinal areas.4LAM is associated with tuberous sclerosis, which can also present with skin lesions (angiofibromas) and renal tumors (angiomyolipomas), thus creating a very similar picture to BHD. Therefore, tissue biopsies of skin lesions are essential as histopathology can identify characteristic fibrofolliculomas specific to BHD. While genetic testing would also strongly support the diagnosis of BHD, it is essential to note that negative genetic testing does not rule out BHD.4Lastly, lymphoid interstitial pneumonia (LIP) is another important consideration in the differential diagnosis of cystic lung diseases. LIP presents with not only perivascular cysts and centrilobular nodules but also diffuse ground-glass attenuation.10 In contrast to BHD, LIP is associated with autoimmune diseases such as Sjögren syndrome and infectious diseases such as HIV; thus, it may be differentiated from BHD by the presence of underlying disease processes and may warrant serologic testing for potential rheumatologic disorders.
Characteristics and Diagnostic Criteria
Cystic lung disease is the most common presentation of BHD. It presents in > 80% of cases and confers a 50-fold increase in the risk of spontaneous pneumothorax compared with the general population.4,11 Recurrent pneumothoraces are observed in about 25% to 30% of patients with BHD, typically occurring between the third and fifth decades of life and at significantly decreased rates after 50 years of age.12 A spontaneous pneumothorax might serve as the initial and perhaps the sole clinical presentation for some patients with BHD, but others may present with other respiratory symptoms such as cough and exertional dyspnea. PFT results may be normal or reveal a mild restrictive ventilatory defect and reduced DLCO, as reported in a few cases.6 The management of pulmonary complications primarily revolves around reducing the risk of pneumothoraces, which includes precautions such as avoiding positive pressure ventilation and air travel. Early pleurodesis with the first occurrence of a spontaneous pneumothorax is considered in some cases.13
The distinctive dermatologic features associated with BHD include multiple white papules primarily found on the nose and face. Pathologically, these manifestations have a range of histologic distinctions, from fibrofolliculomas to benign hamartomas of the hair follicles and trichodiscomas.5 The diagnostic criteria outlined by Menko et al note that confirmation of BHD requires the presence of either ≥ 5 pathologically confirmed fibrofolliculomas or trichodiscomas, a documented pathogenic FLCN gene mutation, or the fulfillment of 2 minor criteria. These minor criteria include the presence of multiple lung cysts, early-onset renal cancer, or a first-degree relative with BHD.5
Recurrent Pneumothoraces Management
After the first episode of spontaneous pneumothorax, early pleurodesis is indicated as the risk of recurrence can be as high as 75%.4,14 Specific pleurodesis modalities have shown promising results, such as total pleural covering with cellulose mesh. In a small retrospective review, cellulose mesh demonstrated a significant reduction in the recurrence rate of pneumothorax at 7.5 years for patients with BHD compared with partial covering.15 Apart from preventing further pneumothorax episodes in the affected lung, it is also important to highlight patient education and monitoring after initial pleurodesis, as the contralateral lung is also at risk. As demonstrated in this case, the patient had received pleurodesis of his right lung but experienced another pneumothorax of his contralateral lung a few years later.
Lastly, the patient was advised to avoid air travel altogether; however, current data may suggest that air travel may not be an absolute contraindication for patients with BHD. Although the literature on this topic is limited, a retrospective study by Johannesma et al involving 158 patients with BHD surveyed on pneumothorax occurrence after air travel indicated a calculated risk of 0.63% per flight. Notably, only 3 of 13 patients with BHD and recurrent pneumothoraces after travel had undergone pleurodesis in the past.16 Therefore, counseling patients on the potential risks of air travel and allowing essential flights while diligently monitoring for symptoms during and after travel may be a reasonable, patient-centered approach in contrast to a complete restriction on air travel.
Timing to Diagnosis
Diagnosing BHD is challenging and often delayed. In a 2022 study by Steinlein et al, the average delay in BHD diagnoses in their cohort was 9.3 years, with 4 patients also diagnosed with renal malignancy during the study period.17 The difficulty in diagnosis can be attributed to the heterogeneous presentation among affected family members, some of whom may exclusively exhibit pulmonary cystic lesions without dermatologic findings.
A lack of longitudinal care for this patient may have contributed to the diagnostic delay. The patient had pneumothorax events across separate care settings and locations, and due to employment-related relocations, he often re-established care at various health care systems. This case highlights the importance of continuity of care, especially in BHD, where monitoring for renal tumors is also essential to long-term management.17,18
Renal Tumor Monitoring
Finally, once BHD is diagnosed, one of the most important considerations is to begin routine monitoring for renal malignancies. Current recommendations advise starting lifelong renal cancer screening, even as early as age 20 years, with annual MRIs, as renal ultrasound may not be sufficiently sensitive to detect smaller lesions.19 The screening interval can be extended to every 2 years for patients without a family history of renal tumors or suspicious renal lesions. If tumors are found, then nephron-sparing surgery is recommended, given the potential for the development of chronic renal insufficiency in patients with BHD.20
Conclusions
BHD is a rare and complex syndrome in which early recognition and diagnosis play a pivotal role in preventing potentially severe complications such as renal malignancies. Suspicion of a genetic disorder, such as BHD, LAM, or PLCH, should arise in patients who experience spontaneous pneumothorax, especially in the presence of multiple cystic lesions or a family history of pneumothoraces. Early consideration of pleurodesis after the first spontaneous pneumothorax is advisable. The complex presentation of BHD contributes to the difficulty of diagnosis and may delay recognition, which can be exacerbated by variable continuity of care.
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
- Schmidt LS, Linehan WM. Molecular genetics and clinical features of Birt-Hogg-Dubé-Syndrome. Nat Rev Urol. 2015;12:558-569. doi:10.1038/nrurol.2015.206
- Lim DHK, Rehal PK, Nahorski MS, et al. A new locus-specific database (LSDB) for mutations in the folliculin (FLCN) gene. Hum Mutat. 2010;31:E1043-1051. doi:10.1002/humu.21130
- Aivaz O, Berkman S, Middelton L, et al. Comedonal and cystic fibrofolliculomas in Birt-Hogg-Dube syndrome. JAMA Dermatology. 2015;151:770-774. doi:10.1001/jamadermatol.2015.0215
- Daccord C, Good JM, Morren MA, et al. Birt–Hogg–Dubé syndrome. Eur Respir Rev. 2020;29:200042. doi:10.1183/16000617.0042-2020
- Menko FH, van Steensel MA, Giraud S, et al. Birt-Hogg-Dubé syndrome: diagnosis and management. The Lancet Oncology. 2009;10:1199-1206. doi:10.1016/S1470-2045(09)70188-3
- Daccord C, Cottin V, Prévot G, et al. Lung function in Birt-Hogg-Dubé syndrome: a retrospective analysis of 96 patients. Orphanet J Rare Dis. 2020;15:120. doi:10.1186/s13023-020-01402-y
- Hansell DM, Bankier AA, MacMahon H, et al. Fleischner Society: glossary of terms for thoracic imaging. Radiology. 2008;246:697-722. doi:10.1148/radiol.2462070712
- Jamil A, Kasi A. Pneumatocele. In: StatPearls. StatPearls Publishing; 2024. Accessed March 2, 2026. http://www.ncbi.nlm.nih.gov/books/NBK556146/
- Bhardwaj H, Bhardwaj B. Differentiating pulmonary lymphangioleiomyomatosis from pulmonary langerhans cell histiocytosis and Birt-Hogg-Dube syndrome. Lung India. 2013;30:372-373. doi:10.4103/0970-2113.120611
- Swigris JJ, Berry GJ, Raffin TA, et al. Lymphoid interstitial pneumonia: a narrative review. Chest. 2002;122:2150-2164. doi:10.1378/chest.122.6.2150
- Zbar B, Alvord WG, Glenn G, et al. Risk of renal and colonic neoplasms and spontaneous pneumothorax in the Birt-Hogg-Dubé syndrome. Cancer Epidemiol Biomarkers Prev. 2002;11:393-400.
- Sattler EC, Steinlein OK. Delayed diagnosis of Birt-Hogg-Dubé syndrome due to marked intrafamilial clinical variability: a case report. BMC Med Genet. 2018;19:45. doi:10.1186/s12881-018-0558-0
- Gupta N, Seyama K, McCormack FX. Pulmonary manifestations of Birt-Hogg-Dubé syndrome. Fam Cancer. 2013;12:387-396. doi:10.1007/s10689-013-9660-9
- Gupta N, Kopras EJ, Henske EP, et al. Spontaneous pneumothoraces in patients with Birt–Hogg–Dubé syndrome. Ann Am Thorac Soc. 2017;14:706-713. doi:10.1513/AnnalsATS.201611-886OC
- Mizobuchi T, Kurihara M, Ebana H, et al. A total pleural covering of absorbable cellulose mesh prevents pneumothorax recurrence in patients with Birt-Hogg-Dubé syndrome. Orphanet J Rare Dis. 2018;13:78. doi:10.1186/s13023-018-0790-x
- Johannesma PC, van de Beek I, van der Wel JWT, et al. Risk of spontaneous pneumothorax due to air travel and diving in patients with Birt–Hogg–Dubé syndrome. Springerplus. 2016;5:1506. doi:10.1186/s40064-016-3009-4
- Steinlein OK, Reithmair M, Syunyaeva Z, et al. Delayed diagnosis of Birt-Hogg-Dubé syndrome might be aggravated by gender bias. eClinicalMedicine. 2022;51:101572. doi:10.1016/j.eclinm.2022.101572
- Pereira Gray DJ, Sidaway-Lee K, White E, et al. Continuity of care with doctors—a matter of life and death? A systematic review of continuity of care and mortality. BMJ Open. 2018;8:e021161. doi:10.1136/bmjopen-2017-021161
- Sattler EC, Steinlein OK. GeneReviews Birt-Hogg-Dubé syndrome. January 30, 2020. Accessed March 2, 2026. https://www.ncbi.nlm.nih.gov/books/NBK1522/table
- Stamatakis L, Metwalli AR, Middelton LA, et al. Diagnosis and management of BHD-associated kidney cancer. Fam Cancer. 2013;12:397-402. doi:10.1007/s10689-013-9657-4
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider
A Case of Birt-Hogg-Dubé Syndrome: A Rare but Essential Diagnosis to Consider