User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
TOLAC: Induction poses no maternal risk vs. expectant management
SAN DIEGO – Maternal morbidity was not consistently increased in a study of women with one prior cesarean delivery who were attempting a trial of labor and who were induced between 37 and 40 weeks, according to a secondary analysis of data from the National Institutes of Health Consortium on Safe Labor.
But the risk of admission to the neonatal intensive care unit did increase among women induced at 37 weeks, Dr. Justin Lappen reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Further, induction at 37, 38, and 39 weeks of gestation increased the risk of a failed trial of labor after cesarean section (TOLAC) in the 1,626 women in the retrospective multicenter study who were induced, compared with 4,407 who were managed expectantly (odds ratios, 1.53, 1.74, and 2.16, respectively).
The induction group was compared at gestational weeks 37-40 with all undelivered women in the expectant management group. After adjustment for prior spontaneous vaginal delivery, body mass index, hypertensive disorders, maternal age, week of gestation, birth weight, and neonatal gender, induction was not associated with any consistent increase in the risk of maternal morbidity (odds ratios at weeks 37-40 were 0.49, 0.64, 1.87, and 1.06, respectively).
The researchers defined maternal morbidity based on a composite of outcomes including hysterectomy, transfusion, intensive care unit transfer, venous thromboembolism, and death.
The increased risk of the composite maternal morbidity at 39 weeks was primarily the result of a significantly increased risk of transfusion among women with failed TOLAC, said Dr. Lappen of Case Western Reserve University, Cleveland.
However, among those who were induced at 37 weeks, the risk of neonatal ICU admission was increased, compared with women being expectantly managed who delivered at or beyond 37 weeks (OR, 2.51).
No increased risk was seen in those induced beyond 37 weeks, and no increased risk in neonatal morbidity – defined as a composite of 5-minute Apgar scores of less than 5, arterial cord pH less than 7.0, asphyxia, hypoxic ischemic encephalopathy, and death – was seen with induction at any week of gestation analyzed, Dr. Lappen said.
“Our findings should not change the current obstetric practice, which supports an individualized approach to the use of induction in appropriate candidates attempting TOLAC, Dr. Lappen said.
The researchers also performed a subset analysis in a low-risk cohort derived by excluding all women with chronic conditions who were ineligible for expectant management and by limiting the induction group to include only nonmedically indicated inductions of labor.
The association between induction and composite maternal morbidity was unchanged in this subset analysis, with greater risk seen at 39 weeks – again in association with increased risk of transfusion among those with a failed TOLAC attempt.
However, the association between induction and NICU admission at 37 weeks was no longer present in the low-risk cohort, and the risk was cut in half at 39 weeks in this cohort (OR, 0.52). As in the entire cohort, no association between induction and neonatal morbidity was present.
“Consistent with the primary cohort, nonmedically indicated induction was associated with an increased risk of failed TOLAC,” Dr. Lappen said. This finding was statistically significant at 38 and 39 weeks (ORs, 2.67 and 2.34, respectively), he said.
No association was seen between induction and uterine rupture in either the entire cohort or the low-risk cohort, he said.
Study subjects were women with a live, singleton gestation of at least 37 weeks identified from the Consortium on Safe Labor, a cohort of more than 228,600 deliveries at 12 centers between 2002 and 2008, which was designed to characterize current obstetric practice and outcomes.
Dr. Lappen reported having no financial disclosures.
SAN DIEGO – Maternal morbidity was not consistently increased in a study of women with one prior cesarean delivery who were attempting a trial of labor and who were induced between 37 and 40 weeks, according to a secondary analysis of data from the National Institutes of Health Consortium on Safe Labor.
But the risk of admission to the neonatal intensive care unit did increase among women induced at 37 weeks, Dr. Justin Lappen reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Further, induction at 37, 38, and 39 weeks of gestation increased the risk of a failed trial of labor after cesarean section (TOLAC) in the 1,626 women in the retrospective multicenter study who were induced, compared with 4,407 who were managed expectantly (odds ratios, 1.53, 1.74, and 2.16, respectively).
The induction group was compared at gestational weeks 37-40 with all undelivered women in the expectant management group. After adjustment for prior spontaneous vaginal delivery, body mass index, hypertensive disorders, maternal age, week of gestation, birth weight, and neonatal gender, induction was not associated with any consistent increase in the risk of maternal morbidity (odds ratios at weeks 37-40 were 0.49, 0.64, 1.87, and 1.06, respectively).
The researchers defined maternal morbidity based on a composite of outcomes including hysterectomy, transfusion, intensive care unit transfer, venous thromboembolism, and death.
The increased risk of the composite maternal morbidity at 39 weeks was primarily the result of a significantly increased risk of transfusion among women with failed TOLAC, said Dr. Lappen of Case Western Reserve University, Cleveland.
However, among those who were induced at 37 weeks, the risk of neonatal ICU admission was increased, compared with women being expectantly managed who delivered at or beyond 37 weeks (OR, 2.51).
No increased risk was seen in those induced beyond 37 weeks, and no increased risk in neonatal morbidity – defined as a composite of 5-minute Apgar scores of less than 5, arterial cord pH less than 7.0, asphyxia, hypoxic ischemic encephalopathy, and death – was seen with induction at any week of gestation analyzed, Dr. Lappen said.
“Our findings should not change the current obstetric practice, which supports an individualized approach to the use of induction in appropriate candidates attempting TOLAC, Dr. Lappen said.
The researchers also performed a subset analysis in a low-risk cohort derived by excluding all women with chronic conditions who were ineligible for expectant management and by limiting the induction group to include only nonmedically indicated inductions of labor.
The association between induction and composite maternal morbidity was unchanged in this subset analysis, with greater risk seen at 39 weeks – again in association with increased risk of transfusion among those with a failed TOLAC attempt.
However, the association between induction and NICU admission at 37 weeks was no longer present in the low-risk cohort, and the risk was cut in half at 39 weeks in this cohort (OR, 0.52). As in the entire cohort, no association between induction and neonatal morbidity was present.
“Consistent with the primary cohort, nonmedically indicated induction was associated with an increased risk of failed TOLAC,” Dr. Lappen said. This finding was statistically significant at 38 and 39 weeks (ORs, 2.67 and 2.34, respectively), he said.
No association was seen between induction and uterine rupture in either the entire cohort or the low-risk cohort, he said.
Study subjects were women with a live, singleton gestation of at least 37 weeks identified from the Consortium on Safe Labor, a cohort of more than 228,600 deliveries at 12 centers between 2002 and 2008, which was designed to characterize current obstetric practice and outcomes.
Dr. Lappen reported having no financial disclosures.
SAN DIEGO – Maternal morbidity was not consistently increased in a study of women with one prior cesarean delivery who were attempting a trial of labor and who were induced between 37 and 40 weeks, according to a secondary analysis of data from the National Institutes of Health Consortium on Safe Labor.
But the risk of admission to the neonatal intensive care unit did increase among women induced at 37 weeks, Dr. Justin Lappen reported at the annual Pregnancy Meeting sponsored by the Society for Maternal-Fetal Medicine.
Further, induction at 37, 38, and 39 weeks of gestation increased the risk of a failed trial of labor after cesarean section (TOLAC) in the 1,626 women in the retrospective multicenter study who were induced, compared with 4,407 who were managed expectantly (odds ratios, 1.53, 1.74, and 2.16, respectively).
The induction group was compared at gestational weeks 37-40 with all undelivered women in the expectant management group. After adjustment for prior spontaneous vaginal delivery, body mass index, hypertensive disorders, maternal age, week of gestation, birth weight, and neonatal gender, induction was not associated with any consistent increase in the risk of maternal morbidity (odds ratios at weeks 37-40 were 0.49, 0.64, 1.87, and 1.06, respectively).
The researchers defined maternal morbidity based on a composite of outcomes including hysterectomy, transfusion, intensive care unit transfer, venous thromboembolism, and death.
The increased risk of the composite maternal morbidity at 39 weeks was primarily the result of a significantly increased risk of transfusion among women with failed TOLAC, said Dr. Lappen of Case Western Reserve University, Cleveland.
However, among those who were induced at 37 weeks, the risk of neonatal ICU admission was increased, compared with women being expectantly managed who delivered at or beyond 37 weeks (OR, 2.51).
No increased risk was seen in those induced beyond 37 weeks, and no increased risk in neonatal morbidity – defined as a composite of 5-minute Apgar scores of less than 5, arterial cord pH less than 7.0, asphyxia, hypoxic ischemic encephalopathy, and death – was seen with induction at any week of gestation analyzed, Dr. Lappen said.
“Our findings should not change the current obstetric practice, which supports an individualized approach to the use of induction in appropriate candidates attempting TOLAC, Dr. Lappen said.
The researchers also performed a subset analysis in a low-risk cohort derived by excluding all women with chronic conditions who were ineligible for expectant management and by limiting the induction group to include only nonmedically indicated inductions of labor.
The association between induction and composite maternal morbidity was unchanged in this subset analysis, with greater risk seen at 39 weeks – again in association with increased risk of transfusion among those with a failed TOLAC attempt.
However, the association between induction and NICU admission at 37 weeks was no longer present in the low-risk cohort, and the risk was cut in half at 39 weeks in this cohort (OR, 0.52). As in the entire cohort, no association between induction and neonatal morbidity was present.
“Consistent with the primary cohort, nonmedically indicated induction was associated with an increased risk of failed TOLAC,” Dr. Lappen said. This finding was statistically significant at 38 and 39 weeks (ORs, 2.67 and 2.34, respectively), he said.
No association was seen between induction and uterine rupture in either the entire cohort or the low-risk cohort, he said.
Study subjects were women with a live, singleton gestation of at least 37 weeks identified from the Consortium on Safe Labor, a cohort of more than 228,600 deliveries at 12 centers between 2002 and 2008, which was designed to characterize current obstetric practice and outcomes.
Dr. Lappen reported having no financial disclosures.
AT THE PREGNANCY MEETING
Key clinical point: An individualized approach is warranted for induction in appropriate candidates attempting TOLAC.
Major finding: Induction was not consistently associated with composite maternal morbidity (odds ratios at weeks 37-40 were 0.49, 0.64, 1.87, and 1.06, respectively).
Data source: A secondary analysis of data from a retrospective multicenter study of 6,033 women.
Disclosures: Dr. Lappen reported having no financial disclosures.
New Psoriasis Drugs Offer Treatment Advantages
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Continue for information on secukinumab >>
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Continue for information on secukinumab >>
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Continue for information on secukinumab >>
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
EXPERT ANALYSIS FROM THE ANNUAL SOUTH BEACH SYMPOSIUM
New psoriasis drugs offer treatment advantages
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
MIAMI BEACH – Two of the newest treatments available for psoriasis – apremilast and secukinumab – are true “game-changers,” according to Dr. David M. Pariser.
Apremilast (Otezla), a recently approved oral phosphodiesterase 4 inhibitor, will be a particularly attractive treatment option for many dermatologists and patients, he said at the South Beach Symposium.
Apremilast has a very limited effect on the immune system, and it’s an oral therapy and thus requires no needles. It is very safe – with “strikingly few” serious adverse events – and no laboratory monitoring is required, he explained.
Efficacy results with apremilast are modest. In the phase III ESTEEM trial, for example, 33% of patients achieved at least 75% improvement (PASI-75), compared with 5% of patients who received placebo, said Dr. Pariser of Eastern Virginia Medical School, Norfolk, and an investigator for the trial.
Further, the drug can be used for almost any patient and type of psoriasis; it is an option for those who want systemic therapy, but who don’t want to go on a biologic or methotrexate, and its use is not precluded by a history of cancer or infections, as is the case with biologics, he added.
Apremilast also will be attractive for dermatologists who do not currently prescribe systemic therapy for psoriasis, or who don’t use aggressive systemic therapy for psoriasis, he said.
“If a prescriber feels safety is more important than efficacy, this might be a good choice,” he said.
Secukinumab (Cosentyx), on the other hand, is a “big gun,” Dr. Pariser said of the biologic, which was approved in January 2015 for the treatment of adults with moderate to severe plaque psoriasis.
“It’s the biggest gun we’ve got now … and it really has a safety profile similar to existing biologics so far,” he said.
The fully human monoclonal antibody inhibits interleukin-17A and is administered by subcutaneous injection. Its safety and efficacy were demonstrated in numerous of studies involving about 4,500 patients. Treatment was associated with significant improvement, compared with placebo, said Dr. Pariser, who also was an investigator on secukinumab trials.
Of note, while the PASI-75 findings for secukinumab are “a nice number but not dramatically higher than things we have had in the past,” the PASI-90 and PASI-100 scores are remarkable, he said.
At the highest dose studied (300 mg given at weeks 1, 2, 3, 4, and 8, and monthly thereafter), PASI-90 was achieved by 59% of patients in one study, and PASI-100 was achieved in 28%.
“That’s significant. We haven’t had that before, and that is really, really, really nice,” he said.
Further, the primary efficacy endpoint of the study was outcome at 12 weeks, but patients continued to improve at least until 16 weeks.
PASI-100 – no psoriasis whatsoever – was 40% at 16 weeks, he said.
In another phase III trial, secukinumab was again shown to be superior to placebo, but it also compared favorably with etanercept, he noted.
Safety was reasonable in the secukinumab trials. No deaths occurred, but there were more serious adverse events and discontinuations in the active treatment group. Nasopharyngitis was the most common serious adverse events, and it occurred in all groups. Upper respiratory tract infections appeared to be more common in the secukinumab patients, he noted.
An important consideration with secukinumab, however, is the need for continuous treatment, he said.
“The bottom line, really, is that patients should stay on it. They will lose effectiveness if they go off of it or take it on an as-needed basis,” he said.
Dr. Pariser is a consultant and/or researcher for Amgen, AbbVie, Celgene, Eli Lilly, Janssen Pharmaceuticals, Merck, and Pfizer.
EXPERT ANALYSIS FROM THE ANNUAL SOUTH BEACH SYMPOSIUM
Clear guidance, clearer skin for acne patients
MIAMI BEACH – The value of specific instructions for general skin care in acne patients is often underestimated, but clear guidance can improve outcomes, according to Dr. James Q. Del Rosso.
“Skin care is very important. It sounds trivial, but it is extremely important to tell patients exactly what they should be using as far as skin care,” he said at the South Beach Symposium.
Using the wrong products can reduce the efficacy of acne treatment, he said. Patients may use products that damage the stratum corneum and cause excess water loss, thereby increasing the likelihood of irritation. This sets their skin up for additional irritation or sensitization from prescribed topical medications, he explained.
“If you controlled the skin care, you wouldn’t have that problem at all,” said Dr. Del Rosso of Touro University in Henderson, Nev.
Additionally, patients may be engaging in procedures at home, such as microdermabrasion, that can increase transepidermal water loss and promote inflammation as part of the skin’s self-repair mechanism, he noted.
Such inflammation can increase skin sensitivity to medications.
“So controlling the skin care has a lot to do with what the therapeutic outcome will be,” he said.
Dr. Del Russo and colleagues found that a specific regimen consisting of a foam moisturizer and an SPF 30 moisturizer formulated with a lower dose of sunscreen (DermaControl) to reduce the potential for irritation was safe, effective, and well liked by patients when used in conjunction with daily application of a topical gel containing adapalene 1% and benzoyl peroxide 2.5% (Epiduo). Their findings appeared earlier this year (J. Clin. Aesthet. Dermatol. 2015; 8:22-30).
Further, a review of the types of patients included in the original pivotal trials for Epiduo and those in Dr. Del Rosso’s study suggests that use of the DermaControl skin care regimen lessened skin irritation caused by the prescription treatment. The same types of patients were included in both studies, and the same treatment regimen was used, he said.
Additional studies comparing various treatments used with and without specific skin care regimens are needed, and are likely in the future, he said.
Dr. Del Rosso has served as a consultant, researcher, and/or speaker for multiple companies including Allergan, Anacor Pharmaceuticals, Aqua Pharmaceuticals, Bayer HealthCare Pharmaceuticals, Celgene, Dermira, Ferndale Pharmaceuticals, Galderma, Genentech, Innocutis, LEO Pharmaceuticals, Promius Pharma, PuraCap Pharmaceutical, Quinnova Pharmaceuticals, Ranbaxy Pharmaceuticals, Sebacia, Suneva Medical, Taro Pharmaceutical Industries, Unilever, and Valeant. His spouse is employed by Novartis.
MIAMI BEACH – The value of specific instructions for general skin care in acne patients is often underestimated, but clear guidance can improve outcomes, according to Dr. James Q. Del Rosso.
“Skin care is very important. It sounds trivial, but it is extremely important to tell patients exactly what they should be using as far as skin care,” he said at the South Beach Symposium.
Using the wrong products can reduce the efficacy of acne treatment, he said. Patients may use products that damage the stratum corneum and cause excess water loss, thereby increasing the likelihood of irritation. This sets their skin up for additional irritation or sensitization from prescribed topical medications, he explained.
“If you controlled the skin care, you wouldn’t have that problem at all,” said Dr. Del Rosso of Touro University in Henderson, Nev.
Additionally, patients may be engaging in procedures at home, such as microdermabrasion, that can increase transepidermal water loss and promote inflammation as part of the skin’s self-repair mechanism, he noted.
Such inflammation can increase skin sensitivity to medications.
“So controlling the skin care has a lot to do with what the therapeutic outcome will be,” he said.
Dr. Del Russo and colleagues found that a specific regimen consisting of a foam moisturizer and an SPF 30 moisturizer formulated with a lower dose of sunscreen (DermaControl) to reduce the potential for irritation was safe, effective, and well liked by patients when used in conjunction with daily application of a topical gel containing adapalene 1% and benzoyl peroxide 2.5% (Epiduo). Their findings appeared earlier this year (J. Clin. Aesthet. Dermatol. 2015; 8:22-30).
Further, a review of the types of patients included in the original pivotal trials for Epiduo and those in Dr. Del Rosso’s study suggests that use of the DermaControl skin care regimen lessened skin irritation caused by the prescription treatment. The same types of patients were included in both studies, and the same treatment regimen was used, he said.
Additional studies comparing various treatments used with and without specific skin care regimens are needed, and are likely in the future, he said.
Dr. Del Rosso has served as a consultant, researcher, and/or speaker for multiple companies including Allergan, Anacor Pharmaceuticals, Aqua Pharmaceuticals, Bayer HealthCare Pharmaceuticals, Celgene, Dermira, Ferndale Pharmaceuticals, Galderma, Genentech, Innocutis, LEO Pharmaceuticals, Promius Pharma, PuraCap Pharmaceutical, Quinnova Pharmaceuticals, Ranbaxy Pharmaceuticals, Sebacia, Suneva Medical, Taro Pharmaceutical Industries, Unilever, and Valeant. His spouse is employed by Novartis.
MIAMI BEACH – The value of specific instructions for general skin care in acne patients is often underestimated, but clear guidance can improve outcomes, according to Dr. James Q. Del Rosso.
“Skin care is very important. It sounds trivial, but it is extremely important to tell patients exactly what they should be using as far as skin care,” he said at the South Beach Symposium.
Using the wrong products can reduce the efficacy of acne treatment, he said. Patients may use products that damage the stratum corneum and cause excess water loss, thereby increasing the likelihood of irritation. This sets their skin up for additional irritation or sensitization from prescribed topical medications, he explained.
“If you controlled the skin care, you wouldn’t have that problem at all,” said Dr. Del Rosso of Touro University in Henderson, Nev.
Additionally, patients may be engaging in procedures at home, such as microdermabrasion, that can increase transepidermal water loss and promote inflammation as part of the skin’s self-repair mechanism, he noted.
Such inflammation can increase skin sensitivity to medications.
“So controlling the skin care has a lot to do with what the therapeutic outcome will be,” he said.
Dr. Del Russo and colleagues found that a specific regimen consisting of a foam moisturizer and an SPF 30 moisturizer formulated with a lower dose of sunscreen (DermaControl) to reduce the potential for irritation was safe, effective, and well liked by patients when used in conjunction with daily application of a topical gel containing adapalene 1% and benzoyl peroxide 2.5% (Epiduo). Their findings appeared earlier this year (J. Clin. Aesthet. Dermatol. 2015; 8:22-30).
Further, a review of the types of patients included in the original pivotal trials for Epiduo and those in Dr. Del Rosso’s study suggests that use of the DermaControl skin care regimen lessened skin irritation caused by the prescription treatment. The same types of patients were included in both studies, and the same treatment regimen was used, he said.
Additional studies comparing various treatments used with and without specific skin care regimens are needed, and are likely in the future, he said.
Dr. Del Rosso has served as a consultant, researcher, and/or speaker for multiple companies including Allergan, Anacor Pharmaceuticals, Aqua Pharmaceuticals, Bayer HealthCare Pharmaceuticals, Celgene, Dermira, Ferndale Pharmaceuticals, Galderma, Genentech, Innocutis, LEO Pharmaceuticals, Promius Pharma, PuraCap Pharmaceutical, Quinnova Pharmaceuticals, Ranbaxy Pharmaceuticals, Sebacia, Suneva Medical, Taro Pharmaceutical Industries, Unilever, and Valeant. His spouse is employed by Novartis.
AT THE SOUTH BEACH SYMPOSIUM
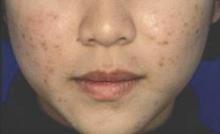
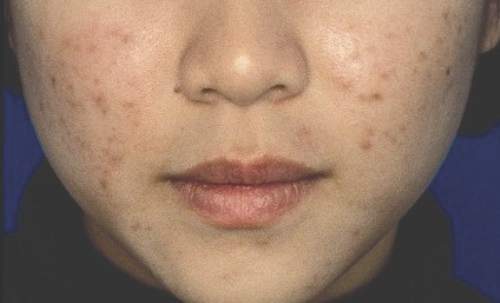
Adapalene/Benzoyl Peroxide Gel Improves Acne in Adult Women
MIAMI BEACH – A fixed-dose adapalene/benzoyl peroxide gel is a suitable treatment for acne in adult women, according to a meta-analysis of data from three randomized, vehicle-controlled multicenter studies.
The median percent change in total lesions at 12 weeks in 130 women who were treated with the 0.1% adapalene/2.5% benzoyl peroxide gel (A-BPO) was –71%, compared with –45% in 124 treated with vehicle gel, Dr. Linda Stein Gold of Henry Ford Medical Center, Detroit, reported in a poster at the annual South Beach Symposium.
Similarly, the median percent change in inflammatory lesions was –73% vs. –41%, and the median percent change in noninflammatory lesions was –70% vs. –45% in the groups, respectively.
Response to the A-BPO treatment was rapid, with significant improvements seen at 1 week, compared with vehicle. Treatment was also well tolerated; adverse events occurring in at least 3% of those in the A-BPO treatment group were dry skin, contact dermatitis, and application site irritation, but all were mild, Dr. Gold noted.
Study participants were women aged 25 years and older. Data increasingly demonstrate that the prevalence of acne in women in this age group is increasing, with up to 54% of women affected, she said.
“Furthermore, recent data show that the mandibular form of acne traditionally associated with adult women is less prevalent than previously thought and that most acne in adult women more closely resembles adolescent acne,” she wrote.
Treatment with benzoyl peroxide in combination with a retinoid has been recommended as a first-line treatment for women with acne, and the fixed-dose A-BPO combination used in this study has been shown to be safe in patients as young as age 9 years.
The findings of this meta-analysis suggest that it is also effective and safe – with quick onset of action – in adult women, she concluded.
This study was funded by Galderma Research and Development and Galderma Laboratories.
MIAMI BEACH – A fixed-dose adapalene/benzoyl peroxide gel is a suitable treatment for acne in adult women, according to a meta-analysis of data from three randomized, vehicle-controlled multicenter studies.
The median percent change in total lesions at 12 weeks in 130 women who were treated with the 0.1% adapalene/2.5% benzoyl peroxide gel (A-BPO) was –71%, compared with –45% in 124 treated with vehicle gel, Dr. Linda Stein Gold of Henry Ford Medical Center, Detroit, reported in a poster at the annual South Beach Symposium.
Similarly, the median percent change in inflammatory lesions was –73% vs. –41%, and the median percent change in noninflammatory lesions was –70% vs. –45% in the groups, respectively.
Response to the A-BPO treatment was rapid, with significant improvements seen at 1 week, compared with vehicle. Treatment was also well tolerated; adverse events occurring in at least 3% of those in the A-BPO treatment group were dry skin, contact dermatitis, and application site irritation, but all were mild, Dr. Gold noted.
Study participants were women aged 25 years and older. Data increasingly demonstrate that the prevalence of acne in women in this age group is increasing, with up to 54% of women affected, she said.
“Furthermore, recent data show that the mandibular form of acne traditionally associated with adult women is less prevalent than previously thought and that most acne in adult women more closely resembles adolescent acne,” she wrote.
Treatment with benzoyl peroxide in combination with a retinoid has been recommended as a first-line treatment for women with acne, and the fixed-dose A-BPO combination used in this study has been shown to be safe in patients as young as age 9 years.
The findings of this meta-analysis suggest that it is also effective and safe – with quick onset of action – in adult women, she concluded.
This study was funded by Galderma Research and Development and Galderma Laboratories.
MIAMI BEACH – A fixed-dose adapalene/benzoyl peroxide gel is a suitable treatment for acne in adult women, according to a meta-analysis of data from three randomized, vehicle-controlled multicenter studies.
The median percent change in total lesions at 12 weeks in 130 women who were treated with the 0.1% adapalene/2.5% benzoyl peroxide gel (A-BPO) was –71%, compared with –45% in 124 treated with vehicle gel, Dr. Linda Stein Gold of Henry Ford Medical Center, Detroit, reported in a poster at the annual South Beach Symposium.
Similarly, the median percent change in inflammatory lesions was –73% vs. –41%, and the median percent change in noninflammatory lesions was –70% vs. –45% in the groups, respectively.
Response to the A-BPO treatment was rapid, with significant improvements seen at 1 week, compared with vehicle. Treatment was also well tolerated; adverse events occurring in at least 3% of those in the A-BPO treatment group were dry skin, contact dermatitis, and application site irritation, but all were mild, Dr. Gold noted.
Study participants were women aged 25 years and older. Data increasingly demonstrate that the prevalence of acne in women in this age group is increasing, with up to 54% of women affected, she said.
“Furthermore, recent data show that the mandibular form of acne traditionally associated with adult women is less prevalent than previously thought and that most acne in adult women more closely resembles adolescent acne,” she wrote.
Treatment with benzoyl peroxide in combination with a retinoid has been recommended as a first-line treatment for women with acne, and the fixed-dose A-BPO combination used in this study has been shown to be safe in patients as young as age 9 years.
The findings of this meta-analysis suggest that it is also effective and safe – with quick onset of action – in adult women, she concluded.
This study was funded by Galderma Research and Development and Galderma Laboratories.
Adapalene/benzoyl peroxide gel improves acne in adult women
MIAMI BEACH – A fixed-dose adapalene/benzoyl peroxide gel is a suitable treatment for acne in adult women, according to a meta-analysis of data from three randomized, vehicle-controlled multicenter studies.
The median percent change in total lesions at 12 weeks in 130 women who were treated with the 0.1% adapalene/2.5% benzoyl peroxide gel (A-BPO) was –71%, compared with –45% in 124 treated with vehicle gel, Dr. Linda Stein Gold of Henry Ford Medical Center, Detroit, reported in a poster at the annual South Beach Symposium.
Similarly, the median percent change in inflammatory lesions was –73% vs. –41%, and the median percent change in noninflammatory lesions was –70% vs. –45% in the groups, respectively.
Response to the A-BPO treatment was rapid, with significant improvements seen at 1 week, compared with vehicle. Treatment was also well tolerated; adverse events occurring in at least 3% of those in the A-BPO treatment group were dry skin, contact dermatitis, and application site irritation, but all were mild, Dr. Gold noted.
Study participants were women aged 25 years and older. Data increasingly demonstrate that the prevalence of acne in women in this age group is increasing, with up to 54% of women affected, she said.
“Furthermore, recent data show that the mandibular form of acne traditionally associated with adult women is less prevalent than previously thought and that most acne in adult women more closely resembles adolescent acne,” she wrote.
Treatment with benzoyl peroxide in combination with a retinoid has been recommended as a first-line treatment for women with acne, and the fixed-dose A-BPO combination used in this study has been shown to be safe in patients as young as age 9 years.
The findings of this meta-analysis suggest that it is also effective and safe – with quick onset of action – in adult women, she concluded.
This study was funded by Galderma Research and Development and Galderma Laboratories.
MIAMI BEACH – A fixed-dose adapalene/benzoyl peroxide gel is a suitable treatment for acne in adult women, according to a meta-analysis of data from three randomized, vehicle-controlled multicenter studies.
The median percent change in total lesions at 12 weeks in 130 women who were treated with the 0.1% adapalene/2.5% benzoyl peroxide gel (A-BPO) was –71%, compared with –45% in 124 treated with vehicle gel, Dr. Linda Stein Gold of Henry Ford Medical Center, Detroit, reported in a poster at the annual South Beach Symposium.
Similarly, the median percent change in inflammatory lesions was –73% vs. –41%, and the median percent change in noninflammatory lesions was –70% vs. –45% in the groups, respectively.
Response to the A-BPO treatment was rapid, with significant improvements seen at 1 week, compared with vehicle. Treatment was also well tolerated; adverse events occurring in at least 3% of those in the A-BPO treatment group were dry skin, contact dermatitis, and application site irritation, but all were mild, Dr. Gold noted.
Study participants were women aged 25 years and older. Data increasingly demonstrate that the prevalence of acne in women in this age group is increasing, with up to 54% of women affected, she said.
“Furthermore, recent data show that the mandibular form of acne traditionally associated with adult women is less prevalent than previously thought and that most acne in adult women more closely resembles adolescent acne,” she wrote.
Treatment with benzoyl peroxide in combination with a retinoid has been recommended as a first-line treatment for women with acne, and the fixed-dose A-BPO combination used in this study has been shown to be safe in patients as young as age 9 years.
The findings of this meta-analysis suggest that it is also effective and safe – with quick onset of action – in adult women, she concluded.
This study was funded by Galderma Research and Development and Galderma Laboratories.
MIAMI BEACH – A fixed-dose adapalene/benzoyl peroxide gel is a suitable treatment for acne in adult women, according to a meta-analysis of data from three randomized, vehicle-controlled multicenter studies.
The median percent change in total lesions at 12 weeks in 130 women who were treated with the 0.1% adapalene/2.5% benzoyl peroxide gel (A-BPO) was –71%, compared with –45% in 124 treated with vehicle gel, Dr. Linda Stein Gold of Henry Ford Medical Center, Detroit, reported in a poster at the annual South Beach Symposium.
Similarly, the median percent change in inflammatory lesions was –73% vs. –41%, and the median percent change in noninflammatory lesions was –70% vs. –45% in the groups, respectively.
Response to the A-BPO treatment was rapid, with significant improvements seen at 1 week, compared with vehicle. Treatment was also well tolerated; adverse events occurring in at least 3% of those in the A-BPO treatment group were dry skin, contact dermatitis, and application site irritation, but all were mild, Dr. Gold noted.
Study participants were women aged 25 years and older. Data increasingly demonstrate that the prevalence of acne in women in this age group is increasing, with up to 54% of women affected, she said.
“Furthermore, recent data show that the mandibular form of acne traditionally associated with adult women is less prevalent than previously thought and that most acne in adult women more closely resembles adolescent acne,” she wrote.
Treatment with benzoyl peroxide in combination with a retinoid has been recommended as a first-line treatment for women with acne, and the fixed-dose A-BPO combination used in this study has been shown to be safe in patients as young as age 9 years.
The findings of this meta-analysis suggest that it is also effective and safe – with quick onset of action – in adult women, she concluded.
This study was funded by Galderma Research and Development and Galderma Laboratories.
Key clinical point: Adapalene/benzoyl peroxide gel is safe and effective for acne in adult women.
Major finding: Mean percent change in total lesions at 12 weeks with adapalene/benzoyl peroxide gel vs. vehicle: –71% vs. –45%.
Data source: A meta-analysis of three studies involving 254 women.
Disclosures: This study was funded by Galderma Research and Development and Galderma Laboratories.
Data Suggest Link Between Tonsillectomy, Psoriasis Improvement
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
Data suggest link between tonsillectomy, psoriasis improvement
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
MIAMI BEACH – If all else fails in treating psoriasis, a tonsillectomy may do the trick – at least in patients whose psoriasis is associated with recurrent tonsillitis, according to findings from a systematic review of available data.
“Maybe taking the tonsils out removes resident bacteria like strep whose antigens tend to promote psoriasis,” Dr. Theodore Rosen, professor of dermatology at Baylor College of Medicine, Houston, suggested during a presentation at the South Beach Symposium.
The relationship between streptococcal pharyngitis and guttate psoriasis is well known, he explained during the talk on the latest findings in clinical dermatology.
Indeed, the authors of the systematic review noted that streptococcal infection is associated with psoriasis onset in some patients, but said it was unknown whether tonsillectomy decreases psoriasis symptoms in those patients.
Dr. Tara D. Rachakonda of Salt Lake City and her colleagues included studies dating back to 1960, and identified 20 relevant studies – including 5 controlled studies – involving 545 patients with psoriasis who were evaluated for or underwent tonsillectomy. The overall improvement rate across the studies was about 70% in 410 patients who underwent tonsillectomy, and in one of the studies the response rate was 86%.
In some cases, the response was sustained, but some patients experienced relapse, they reported (JAAD 2015;72:261-75).
The authors concluded that tonsillectomy may be a potential treatment option in patients with recalcitrant psoriasis associated with episodes of tonsillitis, but noted that additional study with long-term follow-up is needed to examine both the extent and persistence of benefit of tonsillectomy in psoriasis patients.
“The evidence is not sufficient to recommend tonsillectomy in all of your psoriasis patients,” Dr. Rosen said, but he noted that it may be something worth considering in patients who still have their tonsils and who are not responding to psoriasis treatments.
Dr. Rosen reported having no relevant financial disclosures.
Ask Teens With Acne About Whey Protein Use
MIAMI BEACH – Acne on the face and trunk in adolescents could be associated with the use of whey protein supplements, according to Dr. Jonette E. Keri.
Teenage boys, especially, may use whey protein supplements in an effort to increase muscle mass or gain weight, Dr. Keri of the University of Miami said at the South Beach Symposium.
She cited a 2012 case series of five male teenage athletes aged 14-18 years who were using whey protein shakes or reconstituted powder. The teens presented with moderate to severe acne that responded poorly to standard treatment, but four of the five experienced clearing of the acne upon discontinuation of the whey protein supplement.
All five had been treated unsuccessfully with traditional therapies, including oral antibiotics, topical retinoids, and benzoyl peroxide. One teen stopped using the whey protein immediately after being counseled to do so, and he experienced improvement. Two teens didn’t stop using whey protein immediately but experienced improvement in their acne when they did discontinue it. Another teen experienced clearing after discontinuing the whey protein during a second course of isotretinoin therapy, and the fifth was lost to follow-up (Cutis 2012;90:70-2).
One of the teens who cleared after discontinuing the whey protein experienced a flare of the acne when he reinitiated supplementation, Dr. Keri said.
Three of the boys were trying to build muscle for football and two were trying to gain weight, which underscores the importance of asking teens about supplement use, she noted, adding that, importantly, it’s not just boys who are affected.
Continue for another study >>
In fact, a Brazilian study of 30 patients showed a very strong association between the onset or exacerbation of acne and initiation of whey protein use – and the finding was more pronounced among women than men, and among those without a personal or family history of acne (An. Bras. Dermatol. 2013;88:907-12).
In that study, subjects were examined on three occasions and followed for 60 days.
“They went the gym, wanted to work out a little bit and get bigger, took some whey, and broke out in acne,” Dr. Keri said.
Although acne breakouts won’t happen to everyone who uses whey protein, it’s something worth considering in teens, particularly if they fail to respond to standard therapies, she said.
Although the reason for the association hasn’t yet been “teased out,” whey protein is derived from cow’s milk, and it appears to be related to activation of the insulin cascade, explained Dr. Keri.
Dr. Nanette B. Silverberg of the Mount Sinai Health System, New York, author of the case series involving the five male athletes, noted that milk is known to be associated with acne and suggested that whey protein may be the fraction of dairy products that promote acne formation.
Similarly, based on findings from a study of five adult male bodybuilders who developed moderate to severe facial or truncal acne after consuming whey protein, Dr. Thierry Simonart of Brussels noted that the observations are “in line with biochemical and epidemiological data supporting the effects of milk and dairy products as enhancers of insulin/insulin-like growth factor 1 signaling and acne aggravation” (Dermatology 2012;225:256-8).
Dr. Kerri reported having no relevant disclosures.
MIAMI BEACH – Acne on the face and trunk in adolescents could be associated with the use of whey protein supplements, according to Dr. Jonette E. Keri.
Teenage boys, especially, may use whey protein supplements in an effort to increase muscle mass or gain weight, Dr. Keri of the University of Miami said at the South Beach Symposium.
She cited a 2012 case series of five male teenage athletes aged 14-18 years who were using whey protein shakes or reconstituted powder. The teens presented with moderate to severe acne that responded poorly to standard treatment, but four of the five experienced clearing of the acne upon discontinuation of the whey protein supplement.
All five had been treated unsuccessfully with traditional therapies, including oral antibiotics, topical retinoids, and benzoyl peroxide. One teen stopped using the whey protein immediately after being counseled to do so, and he experienced improvement. Two teens didn’t stop using whey protein immediately but experienced improvement in their acne when they did discontinue it. Another teen experienced clearing after discontinuing the whey protein during a second course of isotretinoin therapy, and the fifth was lost to follow-up (Cutis 2012;90:70-2).
One of the teens who cleared after discontinuing the whey protein experienced a flare of the acne when he reinitiated supplementation, Dr. Keri said.
Three of the boys were trying to build muscle for football and two were trying to gain weight, which underscores the importance of asking teens about supplement use, she noted, adding that, importantly, it’s not just boys who are affected.
Continue for another study >>
In fact, a Brazilian study of 30 patients showed a very strong association between the onset or exacerbation of acne and initiation of whey protein use – and the finding was more pronounced among women than men, and among those without a personal or family history of acne (An. Bras. Dermatol. 2013;88:907-12).
In that study, subjects were examined on three occasions and followed for 60 days.
“They went the gym, wanted to work out a little bit and get bigger, took some whey, and broke out in acne,” Dr. Keri said.
Although acne breakouts won’t happen to everyone who uses whey protein, it’s something worth considering in teens, particularly if they fail to respond to standard therapies, she said.
Although the reason for the association hasn’t yet been “teased out,” whey protein is derived from cow’s milk, and it appears to be related to activation of the insulin cascade, explained Dr. Keri.
Dr. Nanette B. Silverberg of the Mount Sinai Health System, New York, author of the case series involving the five male athletes, noted that milk is known to be associated with acne and suggested that whey protein may be the fraction of dairy products that promote acne formation.
Similarly, based on findings from a study of five adult male bodybuilders who developed moderate to severe facial or truncal acne after consuming whey protein, Dr. Thierry Simonart of Brussels noted that the observations are “in line with biochemical and epidemiological data supporting the effects of milk and dairy products as enhancers of insulin/insulin-like growth factor 1 signaling and acne aggravation” (Dermatology 2012;225:256-8).
Dr. Kerri reported having no relevant disclosures.
MIAMI BEACH – Acne on the face and trunk in adolescents could be associated with the use of whey protein supplements, according to Dr. Jonette E. Keri.
Teenage boys, especially, may use whey protein supplements in an effort to increase muscle mass or gain weight, Dr. Keri of the University of Miami said at the South Beach Symposium.
She cited a 2012 case series of five male teenage athletes aged 14-18 years who were using whey protein shakes or reconstituted powder. The teens presented with moderate to severe acne that responded poorly to standard treatment, but four of the five experienced clearing of the acne upon discontinuation of the whey protein supplement.
All five had been treated unsuccessfully with traditional therapies, including oral antibiotics, topical retinoids, and benzoyl peroxide. One teen stopped using the whey protein immediately after being counseled to do so, and he experienced improvement. Two teens didn’t stop using whey protein immediately but experienced improvement in their acne when they did discontinue it. Another teen experienced clearing after discontinuing the whey protein during a second course of isotretinoin therapy, and the fifth was lost to follow-up (Cutis 2012;90:70-2).
One of the teens who cleared after discontinuing the whey protein experienced a flare of the acne when he reinitiated supplementation, Dr. Keri said.
Three of the boys were trying to build muscle for football and two were trying to gain weight, which underscores the importance of asking teens about supplement use, she noted, adding that, importantly, it’s not just boys who are affected.
Continue for another study >>
In fact, a Brazilian study of 30 patients showed a very strong association between the onset or exacerbation of acne and initiation of whey protein use – and the finding was more pronounced among women than men, and among those without a personal or family history of acne (An. Bras. Dermatol. 2013;88:907-12).
In that study, subjects were examined on three occasions and followed for 60 days.
“They went the gym, wanted to work out a little bit and get bigger, took some whey, and broke out in acne,” Dr. Keri said.
Although acne breakouts won’t happen to everyone who uses whey protein, it’s something worth considering in teens, particularly if they fail to respond to standard therapies, she said.
Although the reason for the association hasn’t yet been “teased out,” whey protein is derived from cow’s milk, and it appears to be related to activation of the insulin cascade, explained Dr. Keri.
Dr. Nanette B. Silverberg of the Mount Sinai Health System, New York, author of the case series involving the five male athletes, noted that milk is known to be associated with acne and suggested that whey protein may be the fraction of dairy products that promote acne formation.
Similarly, based on findings from a study of five adult male bodybuilders who developed moderate to severe facial or truncal acne after consuming whey protein, Dr. Thierry Simonart of Brussels noted that the observations are “in line with biochemical and epidemiological data supporting the effects of milk and dairy products as enhancers of insulin/insulin-like growth factor 1 signaling and acne aggravation” (Dermatology 2012;225:256-8).
Dr. Kerri reported having no relevant disclosures.
EXPERT ANALYSIS FROM THE SOUTH BEACH SYMPOSIUM
Ask teens with acne about whey protein use
MIAMI BEACH – Acne on the face and trunk in adolescents could be associated with the use of whey protein supplements, according to Dr. Jonette E. Keri.
Teenage boys, especially, may use whey protein supplements in an effort to increase muscle mass or gain weight, Dr. Keri of the University of Miami said at the South Beach Symposium.
She cited a 2012 case series of five male teenage athletes aged 14-18 years who were using whey protein shakes or reconstituted powder. The teens presented with moderate to severe acne that responded poorly to standard treatment, but four of the five experienced clearing of the acne upon discontinuation of the whey protein supplement.
All five had been treated unsuccessfully with traditional therapies, including oral antibiotics, topical retinoids, and benzoyl peroxide. One teen stopped using the whey protein immediately after being counseled to do so, and he experienced improvement. Two teens didn’t stop using whey protein immediately but experienced improvement in their acne when they did discontinue it. Another teen experienced clearing after discontinuing the whey protein during a second course of isotretinoin therapy, and the fifth was lost to follow-up (Cutis 2012;90:70-2).
One of the teens who cleared after discontinuing the whey protein experienced a flare of the acne when he reinitiated supplementation, Dr. Keri said.
Three of the boys were trying to build muscle for football and two were trying to gain weight, which underscores the importance of asking teens about supplement use, she noted, adding that, importantly, it’s not just boys who are affected.
In fact, a Brazilian study of 30 patients showed a very strong association between the onset or exacerbation of acne and initiation of whey protein use – and the finding was more pronounced among women than men, and among those without a personal or family history of acne (An. Bras. Dermatol. 2013;88:907-12).
In that study, subjects were examined on three occasions and followed for 60 days.
“They went the gym, wanted to work out a little bit and get bigger, took some whey, and broke out in acne,” Dr. Keri said.
Although acne breakouts won’t happen to everyone who uses whey protein, it’s something worth considering in teens, particularly if they fail to respond to standard therapies, she said.
Although the reason for the association hasn’t yet been “teased out,” whey protein is derived from cow’s milk, and it appears to be related to activation of the insulin cascade, explained Dr. Keri.
Dr. Nanette B. Silverberg of the Mount Sinai Health System, New York, author of the case series involving the five male athletes, noted that milk is known to be associated with acne and suggested that whey protein may be the fraction of dairy products that promote acne formation.
Similarly, based on findings from a study of five adult male bodybuilders who developed moderate to severe facial or truncal acne after consuming whey protein, Dr. Thierry Simonart of Brussels noted that the observations are “in line with biochemical and epidemiological data supporting the effects of milk and dairy products as enhancers of insulin/insulin-like growth factor 1 signaling and acne aggravation” (Dermatology 2012;225:256-8).
Dr. Kerri reported having no relevant disclosures.
MIAMI BEACH – Acne on the face and trunk in adolescents could be associated with the use of whey protein supplements, according to Dr. Jonette E. Keri.
Teenage boys, especially, may use whey protein supplements in an effort to increase muscle mass or gain weight, Dr. Keri of the University of Miami said at the South Beach Symposium.
She cited a 2012 case series of five male teenage athletes aged 14-18 years who were using whey protein shakes or reconstituted powder. The teens presented with moderate to severe acne that responded poorly to standard treatment, but four of the five experienced clearing of the acne upon discontinuation of the whey protein supplement.
All five had been treated unsuccessfully with traditional therapies, including oral antibiotics, topical retinoids, and benzoyl peroxide. One teen stopped using the whey protein immediately after being counseled to do so, and he experienced improvement. Two teens didn’t stop using whey protein immediately but experienced improvement in their acne when they did discontinue it. Another teen experienced clearing after discontinuing the whey protein during a second course of isotretinoin therapy, and the fifth was lost to follow-up (Cutis 2012;90:70-2).
One of the teens who cleared after discontinuing the whey protein experienced a flare of the acne when he reinitiated supplementation, Dr. Keri said.
Three of the boys were trying to build muscle for football and two were trying to gain weight, which underscores the importance of asking teens about supplement use, she noted, adding that, importantly, it’s not just boys who are affected.
In fact, a Brazilian study of 30 patients showed a very strong association between the onset or exacerbation of acne and initiation of whey protein use – and the finding was more pronounced among women than men, and among those without a personal or family history of acne (An. Bras. Dermatol. 2013;88:907-12).
In that study, subjects were examined on three occasions and followed for 60 days.
“They went the gym, wanted to work out a little bit and get bigger, took some whey, and broke out in acne,” Dr. Keri said.
Although acne breakouts won’t happen to everyone who uses whey protein, it’s something worth considering in teens, particularly if they fail to respond to standard therapies, she said.
Although the reason for the association hasn’t yet been “teased out,” whey protein is derived from cow’s milk, and it appears to be related to activation of the insulin cascade, explained Dr. Keri.
Dr. Nanette B. Silverberg of the Mount Sinai Health System, New York, author of the case series involving the five male athletes, noted that milk is known to be associated with acne and suggested that whey protein may be the fraction of dairy products that promote acne formation.
Similarly, based on findings from a study of five adult male bodybuilders who developed moderate to severe facial or truncal acne after consuming whey protein, Dr. Thierry Simonart of Brussels noted that the observations are “in line with biochemical and epidemiological data supporting the effects of milk and dairy products as enhancers of insulin/insulin-like growth factor 1 signaling and acne aggravation” (Dermatology 2012;225:256-8).
Dr. Kerri reported having no relevant disclosures.
MIAMI BEACH – Acne on the face and trunk in adolescents could be associated with the use of whey protein supplements, according to Dr. Jonette E. Keri.
Teenage boys, especially, may use whey protein supplements in an effort to increase muscle mass or gain weight, Dr. Keri of the University of Miami said at the South Beach Symposium.
She cited a 2012 case series of five male teenage athletes aged 14-18 years who were using whey protein shakes or reconstituted powder. The teens presented with moderate to severe acne that responded poorly to standard treatment, but four of the five experienced clearing of the acne upon discontinuation of the whey protein supplement.
All five had been treated unsuccessfully with traditional therapies, including oral antibiotics, topical retinoids, and benzoyl peroxide. One teen stopped using the whey protein immediately after being counseled to do so, and he experienced improvement. Two teens didn’t stop using whey protein immediately but experienced improvement in their acne when they did discontinue it. Another teen experienced clearing after discontinuing the whey protein during a second course of isotretinoin therapy, and the fifth was lost to follow-up (Cutis 2012;90:70-2).
One of the teens who cleared after discontinuing the whey protein experienced a flare of the acne when he reinitiated supplementation, Dr. Keri said.
Three of the boys were trying to build muscle for football and two were trying to gain weight, which underscores the importance of asking teens about supplement use, she noted, adding that, importantly, it’s not just boys who are affected.
In fact, a Brazilian study of 30 patients showed a very strong association between the onset or exacerbation of acne and initiation of whey protein use – and the finding was more pronounced among women than men, and among those without a personal or family history of acne (An. Bras. Dermatol. 2013;88:907-12).
In that study, subjects were examined on three occasions and followed for 60 days.
“They went the gym, wanted to work out a little bit and get bigger, took some whey, and broke out in acne,” Dr. Keri said.
Although acne breakouts won’t happen to everyone who uses whey protein, it’s something worth considering in teens, particularly if they fail to respond to standard therapies, she said.
Although the reason for the association hasn’t yet been “teased out,” whey protein is derived from cow’s milk, and it appears to be related to activation of the insulin cascade, explained Dr. Keri.
Dr. Nanette B. Silverberg of the Mount Sinai Health System, New York, author of the case series involving the five male athletes, noted that milk is known to be associated with acne and suggested that whey protein may be the fraction of dairy products that promote acne formation.
Similarly, based on findings from a study of five adult male bodybuilders who developed moderate to severe facial or truncal acne after consuming whey protein, Dr. Thierry Simonart of Brussels noted that the observations are “in line with biochemical and epidemiological data supporting the effects of milk and dairy products as enhancers of insulin/insulin-like growth factor 1 signaling and acne aggravation” (Dermatology 2012;225:256-8).
Dr. Kerri reported having no relevant disclosures.
EXPERT ANALYSIS FROM THE SOUTH BEACH SYMPOSIUM