User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
Estimating PTSD Prevalence Requires Precise Exposure Details
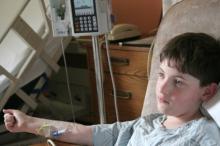
The intensity and type of exposure to trauma should be carefully evaluated when estimating the magnitude of the posttraumatic stress disorder burden in the aftermath of a terrorist attack, findings from a study of 379 surviving employees from eight companies affected by the September 11, 2001 attack on the World Trade Center suggest.
The findings call into question prior estimates of PTSD prevalence after 9/11. Several studies provided such estimates for broadly affected populations following the 9/11 terrorist attacks (people throughout New York City, for example), but none sufficiently addressed the details of the exposure in order to link cases of PTSD to the terrorist attacks, the investigators noted.
More than a third of the subjects in the study who experienced direct exposure to danger during the tragic events of 9/11 met the Diagnostic and Statistical Manual, Fourth Edition, Text Revision (DSM-IV-TR) criteria for posttraumatic stress disorder at any time following the disaster. Furthermore, 20% of those who were exposed to danger through witnessed experiences only, and 35% who were exposed through a close associate’s direct exposure only also met the criteria for PTSD at any time after the events, Dr. Carol S. North of the University of Texas Southwestern Medical Center, Dallas, and her colleagues reported in the Sept. 7 Disaster Medicine and Public Health Preparedness.
Outside of these exposed groups, "few other possible sources of exposure were evident among the few individuals who were symptomatic, most of whom had preexisting psychiatric illness," the investigators wrote (Disaster Med. Public Health Prep. 2011 [doi: 10.1001/dmp.2011.50]).
The assumption that the New York City population was exposed and was therefore at risk for PTSD "has important implications for both prevalence estimates and service provision; it has not, however, been tested with respect to the DSM-IV-TR criteria for PTSD," they wrote. "Although the 9/11 terrorist attacks constituted an undeniable traumatic event, this is not sufficient for the diagnosis of PTSD; a qualifying exposure to the traumatic event is also necessary for consideration of this diagnosis."
That is, symptom scales applied without assessment of trauma exposure may count symptoms unrelated to trauma exposure and cannot accurately estimate PTSD incidence, they explained.
The problem is magnified in low-exposure – and thus low PTSD risk – populations such as geographically broad 9/11–affected populations. The result is a gross overestimate of PTSD prevalence, which could result in inaccurate diagnosis, unnecessary interventions, and missed opportunities for appropriate treatment.
Indeed, the findings indicate that it is unlikely that widespread PTSD-qualifying trauma exposure occurred in populations geographically distant from the attack sites. The findings have implications for disaster mental health planning and response. "Effective disaster mental health planning and response depend on accurate information about the numbers of people who will need distinct types of services," they wrote.
For the current study, investigators interviewed 102 individuals with direct exposure to danger following the attacks, including 65 who were in the World Trade Center towers at the time of the attack, and 277 with various other types of exposure. Subjects were interviewed about 3 years after the attack using the Diagnostic Interview Schedule/Disaster Supplement, and reassessment was performed at 6 years following the attack.
"This study uniquely combined assessment of PTSD according to full DMS-IV-TR criteria with detailed data about geographical proximity and specific experiences of the 9/11 attacks for determination of qualifying trauma exposure," they noted.
While the data can’t be presumed to represent an accurate estimate of PTSD prevalence (because the sample cannot be assumed to be representative of New York City or affected populations), the "major value of these data are, rather, in the opportunity to study the occurrence of PTSD in relation to objective 9/11 trauma exposures," they wrote.
The findings underscore the need for differentiating distress from PTSD and other psychiatric disorders to help prevent "inappropriate pathologizing of normal responses."
"This study has clarified the value of fully considering exposures to terrorist attacks when estimating PTSD prevalence, and it has demonstrated these considerations to be especially pertinent in populations outside close geographic proximity to the incident."
They also noted that the importance of examining trauma exposures with precision is relevant for other trauma-exposed populations as well, such as military combat veterans.
This study was funded by the National Institute of Mental Health, the National Memorial Institute for the Prevention of Terrorism, and the Office of State and Local Government Coordination and Preparedness of the U.S. Department of Homeland Security.
The intensity and type of exposure to trauma should be carefully evaluated when estimating the magnitude of the posttraumatic stress disorder burden in the aftermath of a terrorist attack, findings from a study of 379 surviving employees from eight companies affected by the September 11, 2001 attack on the World Trade Center suggest.
The findings call into question prior estimates of PTSD prevalence after 9/11. Several studies provided such estimates for broadly affected populations following the 9/11 terrorist attacks (people throughout New York City, for example), but none sufficiently addressed the details of the exposure in order to link cases of PTSD to the terrorist attacks, the investigators noted.
More than a third of the subjects in the study who experienced direct exposure to danger during the tragic events of 9/11 met the Diagnostic and Statistical Manual, Fourth Edition, Text Revision (DSM-IV-TR) criteria for posttraumatic stress disorder at any time following the disaster. Furthermore, 20% of those who were exposed to danger through witnessed experiences only, and 35% who were exposed through a close associate’s direct exposure only also met the criteria for PTSD at any time after the events, Dr. Carol S. North of the University of Texas Southwestern Medical Center, Dallas, and her colleagues reported in the Sept. 7 Disaster Medicine and Public Health Preparedness.
Outside of these exposed groups, "few other possible sources of exposure were evident among the few individuals who were symptomatic, most of whom had preexisting psychiatric illness," the investigators wrote (Disaster Med. Public Health Prep. 2011 [doi: 10.1001/dmp.2011.50]).
The assumption that the New York City population was exposed and was therefore at risk for PTSD "has important implications for both prevalence estimates and service provision; it has not, however, been tested with respect to the DSM-IV-TR criteria for PTSD," they wrote. "Although the 9/11 terrorist attacks constituted an undeniable traumatic event, this is not sufficient for the diagnosis of PTSD; a qualifying exposure to the traumatic event is also necessary for consideration of this diagnosis."
That is, symptom scales applied without assessment of trauma exposure may count symptoms unrelated to trauma exposure and cannot accurately estimate PTSD incidence, they explained.
The problem is magnified in low-exposure – and thus low PTSD risk – populations such as geographically broad 9/11–affected populations. The result is a gross overestimate of PTSD prevalence, which could result in inaccurate diagnosis, unnecessary interventions, and missed opportunities for appropriate treatment.
Indeed, the findings indicate that it is unlikely that widespread PTSD-qualifying trauma exposure occurred in populations geographically distant from the attack sites. The findings have implications for disaster mental health planning and response. "Effective disaster mental health planning and response depend on accurate information about the numbers of people who will need distinct types of services," they wrote.
For the current study, investigators interviewed 102 individuals with direct exposure to danger following the attacks, including 65 who were in the World Trade Center towers at the time of the attack, and 277 with various other types of exposure. Subjects were interviewed about 3 years after the attack using the Diagnostic Interview Schedule/Disaster Supplement, and reassessment was performed at 6 years following the attack.
"This study uniquely combined assessment of PTSD according to full DMS-IV-TR criteria with detailed data about geographical proximity and specific experiences of the 9/11 attacks for determination of qualifying trauma exposure," they noted.
While the data can’t be presumed to represent an accurate estimate of PTSD prevalence (because the sample cannot be assumed to be representative of New York City or affected populations), the "major value of these data are, rather, in the opportunity to study the occurrence of PTSD in relation to objective 9/11 trauma exposures," they wrote.
The findings underscore the need for differentiating distress from PTSD and other psychiatric disorders to help prevent "inappropriate pathologizing of normal responses."
"This study has clarified the value of fully considering exposures to terrorist attacks when estimating PTSD prevalence, and it has demonstrated these considerations to be especially pertinent in populations outside close geographic proximity to the incident."
They also noted that the importance of examining trauma exposures with precision is relevant for other trauma-exposed populations as well, such as military combat veterans.
This study was funded by the National Institute of Mental Health, the National Memorial Institute for the Prevention of Terrorism, and the Office of State and Local Government Coordination and Preparedness of the U.S. Department of Homeland Security.
The intensity and type of exposure to trauma should be carefully evaluated when estimating the magnitude of the posttraumatic stress disorder burden in the aftermath of a terrorist attack, findings from a study of 379 surviving employees from eight companies affected by the September 11, 2001 attack on the World Trade Center suggest.
The findings call into question prior estimates of PTSD prevalence after 9/11. Several studies provided such estimates for broadly affected populations following the 9/11 terrorist attacks (people throughout New York City, for example), but none sufficiently addressed the details of the exposure in order to link cases of PTSD to the terrorist attacks, the investigators noted.
More than a third of the subjects in the study who experienced direct exposure to danger during the tragic events of 9/11 met the Diagnostic and Statistical Manual, Fourth Edition, Text Revision (DSM-IV-TR) criteria for posttraumatic stress disorder at any time following the disaster. Furthermore, 20% of those who were exposed to danger through witnessed experiences only, and 35% who were exposed through a close associate’s direct exposure only also met the criteria for PTSD at any time after the events, Dr. Carol S. North of the University of Texas Southwestern Medical Center, Dallas, and her colleagues reported in the Sept. 7 Disaster Medicine and Public Health Preparedness.
Outside of these exposed groups, "few other possible sources of exposure were evident among the few individuals who were symptomatic, most of whom had preexisting psychiatric illness," the investigators wrote (Disaster Med. Public Health Prep. 2011 [doi: 10.1001/dmp.2011.50]).
The assumption that the New York City population was exposed and was therefore at risk for PTSD "has important implications for both prevalence estimates and service provision; it has not, however, been tested with respect to the DSM-IV-TR criteria for PTSD," they wrote. "Although the 9/11 terrorist attacks constituted an undeniable traumatic event, this is not sufficient for the diagnosis of PTSD; a qualifying exposure to the traumatic event is also necessary for consideration of this diagnosis."
That is, symptom scales applied without assessment of trauma exposure may count symptoms unrelated to trauma exposure and cannot accurately estimate PTSD incidence, they explained.
The problem is magnified in low-exposure – and thus low PTSD risk – populations such as geographically broad 9/11–affected populations. The result is a gross overestimate of PTSD prevalence, which could result in inaccurate diagnosis, unnecessary interventions, and missed opportunities for appropriate treatment.
Indeed, the findings indicate that it is unlikely that widespread PTSD-qualifying trauma exposure occurred in populations geographically distant from the attack sites. The findings have implications for disaster mental health planning and response. "Effective disaster mental health planning and response depend on accurate information about the numbers of people who will need distinct types of services," they wrote.
For the current study, investigators interviewed 102 individuals with direct exposure to danger following the attacks, including 65 who were in the World Trade Center towers at the time of the attack, and 277 with various other types of exposure. Subjects were interviewed about 3 years after the attack using the Diagnostic Interview Schedule/Disaster Supplement, and reassessment was performed at 6 years following the attack.
"This study uniquely combined assessment of PTSD according to full DMS-IV-TR criteria with detailed data about geographical proximity and specific experiences of the 9/11 attacks for determination of qualifying trauma exposure," they noted.
While the data can’t be presumed to represent an accurate estimate of PTSD prevalence (because the sample cannot be assumed to be representative of New York City or affected populations), the "major value of these data are, rather, in the opportunity to study the occurrence of PTSD in relation to objective 9/11 trauma exposures," they wrote.
The findings underscore the need for differentiating distress from PTSD and other psychiatric disorders to help prevent "inappropriate pathologizing of normal responses."
"This study has clarified the value of fully considering exposures to terrorist attacks when estimating PTSD prevalence, and it has demonstrated these considerations to be especially pertinent in populations outside close geographic proximity to the incident."
They also noted that the importance of examining trauma exposures with precision is relevant for other trauma-exposed populations as well, such as military combat veterans.
This study was funded by the National Institute of Mental Health, the National Memorial Institute for the Prevention of Terrorism, and the Office of State and Local Government Coordination and Preparedness of the U.S. Department of Homeland Security.
FROM DISASTER MEDICINE AND PUBLIC HEALTH PREPAREDNESS
Major Finding: More than a third of the subjects in the study who experienced direct exposure to danger during the tragic events of 9/11 met criteria for PTSD. Furthermore, 20% of those who were exposed to danger only through witnessed experiences, and 35% who were exposed only through a close associate’s direct exposure also met the criteria for PTSD. Outside of these exposed groups, few other possible sources of exposure were evident among the few individuals who were symptomatic, most of whom had preexisting psychiatric illness.
Data Source: Interviews with 379 surviving employees from eight companies affected by the 9/11 attack on the World Trade Center.
Disclosures: This study was funded by the National Institute of Mental Health, the National Memorial Institute for the Prevention of Terrorism, and the Office of State and local Government Coordination and Preparedness of the U.S. Department of Homeland Security.
Sleep Deprivation Predicts PTSD in Redeployed Soldiers
Short sleep duration is common among redeployed soldiers – particularly those who experience combat – and is associated with numerous impairments, including posttraumatic stress disorder, according to findings from a cross-sectional study of more than 2,700 members of a brigade combat team.
Mean sleep duration was 5.8 hours among the soldiers, who were surveyed 90-180 days after completion of a 6-15 month deployment to Iraq as part of Operation Iraqi Freedom. The majority (72%) of the 2,738 subjects reported short sleep duration – with 43% reporting less than 7 hours nightly (short sleep duration), and 29% reporting less than 6 hours nightly (very short sleep duration), David D. Luxton, Ph.D., and his colleagues reported.
Symptoms of insufficient sleep were reported by 16% of the subjects, and were much more common among those with short sleep duration (odds ratio, 2.9) and those with very short sleep duration (OR, 9.8), compared with those with normal sleep duration, said Dr. Luxton, of the National Center for Telehealth & Technology, Defense Centers of Excellence for Psychological Health and Traumatic Brain Injury in Tacoma, Wash.
Short sleep duration was significantly more common among the 77% of subjects who reported experiencing combat (OR, 0.44), particularly among those who were wounded or injured during combat operations (Sleep 2011;34:1189-95).
The prevalence of medical comorbidities and high-risk behaviors varied based on sleep duration; those with very short sleep duration were at greater risk for all conditions, compared with those with either short or normal sleep duration, the investigators noted.
Furthermore, symptoms of insufficient sleep independently predicted PTSD, depression, mild traumatic brain injury, panic syndrome, and suicide risk, they said.
Among those who met screening criteria for PTSD, 37% reported symptoms of insufficient sleep, and in fact, the strongest predictor of PTSD was sleep duration of less than 6 hours nightly (adjusted OR, 4.7).
Depression also was strongly associated with sleep duration and symptoms of insufficient sleep (adjusted OR, 7.9 and 2.5, respectively), and while mild traumatic brain injury was most strongly associated with combat exposures (adjusted OR, 16.7), it was also associated with very short sleep duration (adjusted OR, 1.8) and symptoms of insufficient sleep (adjusted OR, 1.8 and 2.0, respectively).
Panic syndrome was associated with very short sleep duration and symptoms of insufficient sleep (adjusted OR, 3.9 and 3.0, respectively). Also, very short sleep duration – but not short sleep duration, predicted obesity, and tobacco and alcohol abuse (adjusted OR, 3.3), and both very short sleep duration and symptoms of insufficient sleep predicted suicidal ideation or having attempted suicide (adjusted OR, 3.8 and 2.4, respectively).
Short sleep duration also has been shown in prior studies to be linked with many deleterious outcomes. Few studies, however, have detailed sleep disturbances associated with war or traumatic events or among deployed soldiers, and none have assessed postdeployment sleep duration and comorbid illnesses in a redeployed combat arms unit such as this one, the investigators noted.
Data for the study were collected in 2007 as part of the Health Risk Assessment II project initiated by the Department of Defense, which incorporated many previously validated mental health screening instruments. Subjects were a mean age of 25.5 years, 55% had completed a second, third, or fourth deployment in support of the global war on terror when they were enrolled in the study, and the mean length of their most recent deployment was 398 days.
Furthermore, 23% reported being hurt during deployment, and 67% said they were still affected by their injuries at the time of the survey. Just over half (51%) had one or more medical comorbidities.
The findings provide evidence of the importance of addressing sleep disturbances as a separate disorder upon redeployment, the investigators said, noting that short sleep duration, excessive sleepiness, and fatigue can lead to work-related accidents, and that insufficient sleep also is associated with – and can exacerbate – psychiatric symptoms.
While limited by the self-reporting of symptoms by the subjects and by the cross-sectional nature of this study, the findings suggest that "efforts to reestablish good sleep practices and aggressive evaluation of soldiers with persistent short sleep duration following deployment may aid in the prevention and management of a variety of health risks and potentially minimize long-term health care utilization," and they point to a need for additional educational, behavioral, and clinical services for service members who have a very high prevalence of short sleep duration after deployment, the investigators concluded.
The authors of this study said they had no conflicts to report.
Short sleep duration is common among redeployed soldiers – particularly those who experience combat – and is associated with numerous impairments, including posttraumatic stress disorder, according to findings from a cross-sectional study of more than 2,700 members of a brigade combat team.
Mean sleep duration was 5.8 hours among the soldiers, who were surveyed 90-180 days after completion of a 6-15 month deployment to Iraq as part of Operation Iraqi Freedom. The majority (72%) of the 2,738 subjects reported short sleep duration – with 43% reporting less than 7 hours nightly (short sleep duration), and 29% reporting less than 6 hours nightly (very short sleep duration), David D. Luxton, Ph.D., and his colleagues reported.
Symptoms of insufficient sleep were reported by 16% of the subjects, and were much more common among those with short sleep duration (odds ratio, 2.9) and those with very short sleep duration (OR, 9.8), compared with those with normal sleep duration, said Dr. Luxton, of the National Center for Telehealth & Technology, Defense Centers of Excellence for Psychological Health and Traumatic Brain Injury in Tacoma, Wash.
Short sleep duration was significantly more common among the 77% of subjects who reported experiencing combat (OR, 0.44), particularly among those who were wounded or injured during combat operations (Sleep 2011;34:1189-95).
The prevalence of medical comorbidities and high-risk behaviors varied based on sleep duration; those with very short sleep duration were at greater risk for all conditions, compared with those with either short or normal sleep duration, the investigators noted.
Furthermore, symptoms of insufficient sleep independently predicted PTSD, depression, mild traumatic brain injury, panic syndrome, and suicide risk, they said.
Among those who met screening criteria for PTSD, 37% reported symptoms of insufficient sleep, and in fact, the strongest predictor of PTSD was sleep duration of less than 6 hours nightly (adjusted OR, 4.7).
Depression also was strongly associated with sleep duration and symptoms of insufficient sleep (adjusted OR, 7.9 and 2.5, respectively), and while mild traumatic brain injury was most strongly associated with combat exposures (adjusted OR, 16.7), it was also associated with very short sleep duration (adjusted OR, 1.8) and symptoms of insufficient sleep (adjusted OR, 1.8 and 2.0, respectively).
Panic syndrome was associated with very short sleep duration and symptoms of insufficient sleep (adjusted OR, 3.9 and 3.0, respectively). Also, very short sleep duration – but not short sleep duration, predicted obesity, and tobacco and alcohol abuse (adjusted OR, 3.3), and both very short sleep duration and symptoms of insufficient sleep predicted suicidal ideation or having attempted suicide (adjusted OR, 3.8 and 2.4, respectively).
Short sleep duration also has been shown in prior studies to be linked with many deleterious outcomes. Few studies, however, have detailed sleep disturbances associated with war or traumatic events or among deployed soldiers, and none have assessed postdeployment sleep duration and comorbid illnesses in a redeployed combat arms unit such as this one, the investigators noted.
Data for the study were collected in 2007 as part of the Health Risk Assessment II project initiated by the Department of Defense, which incorporated many previously validated mental health screening instruments. Subjects were a mean age of 25.5 years, 55% had completed a second, third, or fourth deployment in support of the global war on terror when they were enrolled in the study, and the mean length of their most recent deployment was 398 days.
Furthermore, 23% reported being hurt during deployment, and 67% said they were still affected by their injuries at the time of the survey. Just over half (51%) had one or more medical comorbidities.
The findings provide evidence of the importance of addressing sleep disturbances as a separate disorder upon redeployment, the investigators said, noting that short sleep duration, excessive sleepiness, and fatigue can lead to work-related accidents, and that insufficient sleep also is associated with – and can exacerbate – psychiatric symptoms.
While limited by the self-reporting of symptoms by the subjects and by the cross-sectional nature of this study, the findings suggest that "efforts to reestablish good sleep practices and aggressive evaluation of soldiers with persistent short sleep duration following deployment may aid in the prevention and management of a variety of health risks and potentially minimize long-term health care utilization," and they point to a need for additional educational, behavioral, and clinical services for service members who have a very high prevalence of short sleep duration after deployment, the investigators concluded.
The authors of this study said they had no conflicts to report.
Short sleep duration is common among redeployed soldiers – particularly those who experience combat – and is associated with numerous impairments, including posttraumatic stress disorder, according to findings from a cross-sectional study of more than 2,700 members of a brigade combat team.
Mean sleep duration was 5.8 hours among the soldiers, who were surveyed 90-180 days after completion of a 6-15 month deployment to Iraq as part of Operation Iraqi Freedom. The majority (72%) of the 2,738 subjects reported short sleep duration – with 43% reporting less than 7 hours nightly (short sleep duration), and 29% reporting less than 6 hours nightly (very short sleep duration), David D. Luxton, Ph.D., and his colleagues reported.
Symptoms of insufficient sleep were reported by 16% of the subjects, and were much more common among those with short sleep duration (odds ratio, 2.9) and those with very short sleep duration (OR, 9.8), compared with those with normal sleep duration, said Dr. Luxton, of the National Center for Telehealth & Technology, Defense Centers of Excellence for Psychological Health and Traumatic Brain Injury in Tacoma, Wash.
Short sleep duration was significantly more common among the 77% of subjects who reported experiencing combat (OR, 0.44), particularly among those who were wounded or injured during combat operations (Sleep 2011;34:1189-95).
The prevalence of medical comorbidities and high-risk behaviors varied based on sleep duration; those with very short sleep duration were at greater risk for all conditions, compared with those with either short or normal sleep duration, the investigators noted.
Furthermore, symptoms of insufficient sleep independently predicted PTSD, depression, mild traumatic brain injury, panic syndrome, and suicide risk, they said.
Among those who met screening criteria for PTSD, 37% reported symptoms of insufficient sleep, and in fact, the strongest predictor of PTSD was sleep duration of less than 6 hours nightly (adjusted OR, 4.7).
Depression also was strongly associated with sleep duration and symptoms of insufficient sleep (adjusted OR, 7.9 and 2.5, respectively), and while mild traumatic brain injury was most strongly associated with combat exposures (adjusted OR, 16.7), it was also associated with very short sleep duration (adjusted OR, 1.8) and symptoms of insufficient sleep (adjusted OR, 1.8 and 2.0, respectively).
Panic syndrome was associated with very short sleep duration and symptoms of insufficient sleep (adjusted OR, 3.9 and 3.0, respectively). Also, very short sleep duration – but not short sleep duration, predicted obesity, and tobacco and alcohol abuse (adjusted OR, 3.3), and both very short sleep duration and symptoms of insufficient sleep predicted suicidal ideation or having attempted suicide (adjusted OR, 3.8 and 2.4, respectively).
Short sleep duration also has been shown in prior studies to be linked with many deleterious outcomes. Few studies, however, have detailed sleep disturbances associated with war or traumatic events or among deployed soldiers, and none have assessed postdeployment sleep duration and comorbid illnesses in a redeployed combat arms unit such as this one, the investigators noted.
Data for the study were collected in 2007 as part of the Health Risk Assessment II project initiated by the Department of Defense, which incorporated many previously validated mental health screening instruments. Subjects were a mean age of 25.5 years, 55% had completed a second, third, or fourth deployment in support of the global war on terror when they were enrolled in the study, and the mean length of their most recent deployment was 398 days.
Furthermore, 23% reported being hurt during deployment, and 67% said they were still affected by their injuries at the time of the survey. Just over half (51%) had one or more medical comorbidities.
The findings provide evidence of the importance of addressing sleep disturbances as a separate disorder upon redeployment, the investigators said, noting that short sleep duration, excessive sleepiness, and fatigue can lead to work-related accidents, and that insufficient sleep also is associated with – and can exacerbate – psychiatric symptoms.
While limited by the self-reporting of symptoms by the subjects and by the cross-sectional nature of this study, the findings suggest that "efforts to reestablish good sleep practices and aggressive evaluation of soldiers with persistent short sleep duration following deployment may aid in the prevention and management of a variety of health risks and potentially minimize long-term health care utilization," and they point to a need for additional educational, behavioral, and clinical services for service members who have a very high prevalence of short sleep duration after deployment, the investigators concluded.
The authors of this study said they had no conflicts to report.
FROM SLEEP
Major Finding: The majority (72%) of the 2,738 subjects reported short sleep duration – with 43% reporting less than 7 hours nightly (short sleep duration), and 29% reporting less than 6 hours nightly (very short sleep duration) ... the strongest predictor of PTSD was sleep duration less than 6 hours nightly (adjusted odds ratio, 4.7).
Data Source: A cross-sectional study of sleep patterns and comorbid medical conditions among members of a brigade combat team upon return to Fort Lewis, Wash., from Iraq.
Disclosures: The authors said they have no conflicts to report.
Unusual Swine Flu Cases Have H1N1 Links
Two swine-origin influenza A (H3N2) virus strains that triggered febrile respiratory illness in two children this summer contain genetic material from the 2009 influenza A (H1N1) virus – a genetic reassortment that hasn’t been seen before, according to the Centers for Disease Control and Prevention.
The report coincides with the Food and Drug Administration’s approval of a new in vitro diagnostic kit for seasonal influenza and novel influenza A viruses with pandemic potential. The kit will be distributed at no cost to qualified international public health laboratories.
Both of the H3N2 cases were reported in August. There were no epidemiologic links between the cases, and no other human infections with the virus have been detected, although investigations are ongoing, according to the CDC’s Sept. 2 report (MMWR 2011;60:1-4).
The first case involved a boy younger than 5 years, who has since recovered. The Indiana State Department of Health Laboratories reported the case Aug. 17.
The child, who had received flu vaccine in September 2010, presented in July 2011 with fever, cough, shortness of breath, diarrhea, and sore throat. A respiratory specimen taken at a local emergency department tested positive for influenza A (H3). Further testing of the specimen, including testing by the CDC, confirmed swine-origin influenza A (H3N2).
The child had no direct exposure to swine, but a caretaker of the child reported direct contact with asymptomatic swine in the weeks before onset of the boy’s illness.
On Aug. 24, the Pennsylvania Department of Health reported the second case, which involved a girl younger than 5 years who also has completely recovered. That child also had received influenza vaccine in September 2010, and presented in August 2011 with acute onset of fever, nonproductive cough, and lethargy.
A nasopharyngeal swab at a local emergency department was positive for influenza A. Additional testing, including genome sequencing by the CDC, confirmed the virus as swine-origin influenza A (H3N2).
The girl had recently visited an agricultural fair, where she had direct exposure to swine and other animals.
The two viruses are similar but not identical, the CDC reported.
"Seven of the eight gene segments, including the hemagglutinin (HA) and neuraminidase (NA) genes, are similar to those of swine H3N2 influenza viruses circulating among U.S. pigs since 1998 and previously identified in the eight other sporadic cases of human infections with swine-origin influenza A (H3N2) viruses in the United States since 2009," the report’s authors noted.
There’s a notable difference from those eight earlier cases, however: The two new viruses have a matrix gene from the 2009 influenza A (H1N1) virus, which has replaced the matrix gene found in the previous eight swine-origin infections in humans.
Although reassortment between swine influenza and 2009 influenza A (H1N1) has been reported in pigs, that particular genetic combination of swine influenza virus segments is unique, and has not been previously reported in swine or humans.
Researchers have since identified two additional influenza A (H3N2) isolates from swine containing the M gene from the 2009 influenza A (H1N1) virus. Those isolates are undergoing genome sequencing to characterize their genetic composition.
Although nothing is known about the new viral strains’ ability to transmit efficiently in humans or swine, or between swine and humans, they have been found to be resistant to amantadine and rimantadine. They are susceptible, however, to the neuraminidase inhibitor drugs oseltamivir and zanamivir.
The CDC offered clinicians the following diagnostic and treatment recommendations:
– If influenza virus infection is suspected in an individual with recent exposure to swine, a nasopharyngeal swab should be obtained for timely diagnosis at a state public health laboratory.
– Empiric neuraminidase inhibitor antiviral treatment should be considered to quickly limit potential human transmission.
New Test Could Speed Diagnosis
The newly approved diagnostic test – the CDC Human Influenza Virus Real-Time RT-PCR Diagnostic Panel – utilizes a three-module design that incorporates and streamlines previous versions of the CDC’s two separate FDA-cleared diagnostic test kits, according to a CDC statement.
The first module identifies and distinguishes influenza A and B viruses. The second module classifies influenza A viruses by subtype, such as H1N1, H3N2, or 2009 H1N1. The third module detects highly pathogenic avian influenza A (H5N1) viral infection in human respiratory tract specimens.
CDC officials are hopeful that the test, which uses specimens from a patient’s upper or lower respiratory tract, will improve the ability to make quick diagnoses.
"As the spread of the H1N1 pandemic slowed last year, we conducted an end-to-end review of our nation’s medical countermeasure enterprise, which showed a clear need for better diagnostic tests," Dr. Nicole Lurie, assistant secretary for preparedness and response in the Department of Health and Human Services, said in a statement.
"In helping public health officials quickly identify seasonal flu as well as the flu viruses that could become pandemic, this kit can make a real difference in protecting health and saving lives in the United States and around the world," she added.
An H3N2 virus is one of three vaccine viruses selected by the FDA for inclusion in the 2011-2012 U.S. seasonal flu vaccine, based on recommendations from the World Health Organization. The other two in the trivalent vaccine are an influenza A (H1N1) virus and an influenza B virus.
Two swine-origin influenza A (H3N2) virus strains that triggered febrile respiratory illness in two children this summer contain genetic material from the 2009 influenza A (H1N1) virus – a genetic reassortment that hasn’t been seen before, according to the Centers for Disease Control and Prevention.
The report coincides with the Food and Drug Administration’s approval of a new in vitro diagnostic kit for seasonal influenza and novel influenza A viruses with pandemic potential. The kit will be distributed at no cost to qualified international public health laboratories.
Both of the H3N2 cases were reported in August. There were no epidemiologic links between the cases, and no other human infections with the virus have been detected, although investigations are ongoing, according to the CDC’s Sept. 2 report (MMWR 2011;60:1-4).
The first case involved a boy younger than 5 years, who has since recovered. The Indiana State Department of Health Laboratories reported the case Aug. 17.
The child, who had received flu vaccine in September 2010, presented in July 2011 with fever, cough, shortness of breath, diarrhea, and sore throat. A respiratory specimen taken at a local emergency department tested positive for influenza A (H3). Further testing of the specimen, including testing by the CDC, confirmed swine-origin influenza A (H3N2).
The child had no direct exposure to swine, but a caretaker of the child reported direct contact with asymptomatic swine in the weeks before onset of the boy’s illness.
On Aug. 24, the Pennsylvania Department of Health reported the second case, which involved a girl younger than 5 years who also has completely recovered. That child also had received influenza vaccine in September 2010, and presented in August 2011 with acute onset of fever, nonproductive cough, and lethargy.
A nasopharyngeal swab at a local emergency department was positive for influenza A. Additional testing, including genome sequencing by the CDC, confirmed the virus as swine-origin influenza A (H3N2).
The girl had recently visited an agricultural fair, where she had direct exposure to swine and other animals.
The two viruses are similar but not identical, the CDC reported.
"Seven of the eight gene segments, including the hemagglutinin (HA) and neuraminidase (NA) genes, are similar to those of swine H3N2 influenza viruses circulating among U.S. pigs since 1998 and previously identified in the eight other sporadic cases of human infections with swine-origin influenza A (H3N2) viruses in the United States since 2009," the report’s authors noted.
There’s a notable difference from those eight earlier cases, however: The two new viruses have a matrix gene from the 2009 influenza A (H1N1) virus, which has replaced the matrix gene found in the previous eight swine-origin infections in humans.
Although reassortment between swine influenza and 2009 influenza A (H1N1) has been reported in pigs, that particular genetic combination of swine influenza virus segments is unique, and has not been previously reported in swine or humans.
Researchers have since identified two additional influenza A (H3N2) isolates from swine containing the M gene from the 2009 influenza A (H1N1) virus. Those isolates are undergoing genome sequencing to characterize their genetic composition.
Although nothing is known about the new viral strains’ ability to transmit efficiently in humans or swine, or between swine and humans, they have been found to be resistant to amantadine and rimantadine. They are susceptible, however, to the neuraminidase inhibitor drugs oseltamivir and zanamivir.
The CDC offered clinicians the following diagnostic and treatment recommendations:
– If influenza virus infection is suspected in an individual with recent exposure to swine, a nasopharyngeal swab should be obtained for timely diagnosis at a state public health laboratory.
– Empiric neuraminidase inhibitor antiviral treatment should be considered to quickly limit potential human transmission.
New Test Could Speed Diagnosis
The newly approved diagnostic test – the CDC Human Influenza Virus Real-Time RT-PCR Diagnostic Panel – utilizes a three-module design that incorporates and streamlines previous versions of the CDC’s two separate FDA-cleared diagnostic test kits, according to a CDC statement.
The first module identifies and distinguishes influenza A and B viruses. The second module classifies influenza A viruses by subtype, such as H1N1, H3N2, or 2009 H1N1. The third module detects highly pathogenic avian influenza A (H5N1) viral infection in human respiratory tract specimens.
CDC officials are hopeful that the test, which uses specimens from a patient’s upper or lower respiratory tract, will improve the ability to make quick diagnoses.
"As the spread of the H1N1 pandemic slowed last year, we conducted an end-to-end review of our nation’s medical countermeasure enterprise, which showed a clear need for better diagnostic tests," Dr. Nicole Lurie, assistant secretary for preparedness and response in the Department of Health and Human Services, said in a statement.
"In helping public health officials quickly identify seasonal flu as well as the flu viruses that could become pandemic, this kit can make a real difference in protecting health and saving lives in the United States and around the world," she added.
An H3N2 virus is one of three vaccine viruses selected by the FDA for inclusion in the 2011-2012 U.S. seasonal flu vaccine, based on recommendations from the World Health Organization. The other two in the trivalent vaccine are an influenza A (H1N1) virus and an influenza B virus.
Two swine-origin influenza A (H3N2) virus strains that triggered febrile respiratory illness in two children this summer contain genetic material from the 2009 influenza A (H1N1) virus – a genetic reassortment that hasn’t been seen before, according to the Centers for Disease Control and Prevention.
The report coincides with the Food and Drug Administration’s approval of a new in vitro diagnostic kit for seasonal influenza and novel influenza A viruses with pandemic potential. The kit will be distributed at no cost to qualified international public health laboratories.
Both of the H3N2 cases were reported in August. There were no epidemiologic links between the cases, and no other human infections with the virus have been detected, although investigations are ongoing, according to the CDC’s Sept. 2 report (MMWR 2011;60:1-4).
The first case involved a boy younger than 5 years, who has since recovered. The Indiana State Department of Health Laboratories reported the case Aug. 17.
The child, who had received flu vaccine in September 2010, presented in July 2011 with fever, cough, shortness of breath, diarrhea, and sore throat. A respiratory specimen taken at a local emergency department tested positive for influenza A (H3). Further testing of the specimen, including testing by the CDC, confirmed swine-origin influenza A (H3N2).
The child had no direct exposure to swine, but a caretaker of the child reported direct contact with asymptomatic swine in the weeks before onset of the boy’s illness.
On Aug. 24, the Pennsylvania Department of Health reported the second case, which involved a girl younger than 5 years who also has completely recovered. That child also had received influenza vaccine in September 2010, and presented in August 2011 with acute onset of fever, nonproductive cough, and lethargy.
A nasopharyngeal swab at a local emergency department was positive for influenza A. Additional testing, including genome sequencing by the CDC, confirmed the virus as swine-origin influenza A (H3N2).
The girl had recently visited an agricultural fair, where she had direct exposure to swine and other animals.
The two viruses are similar but not identical, the CDC reported.
"Seven of the eight gene segments, including the hemagglutinin (HA) and neuraminidase (NA) genes, are similar to those of swine H3N2 influenza viruses circulating among U.S. pigs since 1998 and previously identified in the eight other sporadic cases of human infections with swine-origin influenza A (H3N2) viruses in the United States since 2009," the report’s authors noted.
There’s a notable difference from those eight earlier cases, however: The two new viruses have a matrix gene from the 2009 influenza A (H1N1) virus, which has replaced the matrix gene found in the previous eight swine-origin infections in humans.
Although reassortment between swine influenza and 2009 influenza A (H1N1) has been reported in pigs, that particular genetic combination of swine influenza virus segments is unique, and has not been previously reported in swine or humans.
Researchers have since identified two additional influenza A (H3N2) isolates from swine containing the M gene from the 2009 influenza A (H1N1) virus. Those isolates are undergoing genome sequencing to characterize their genetic composition.
Although nothing is known about the new viral strains’ ability to transmit efficiently in humans or swine, or between swine and humans, they have been found to be resistant to amantadine and rimantadine. They are susceptible, however, to the neuraminidase inhibitor drugs oseltamivir and zanamivir.
The CDC offered clinicians the following diagnostic and treatment recommendations:
– If influenza virus infection is suspected in an individual with recent exposure to swine, a nasopharyngeal swab should be obtained for timely diagnosis at a state public health laboratory.
– Empiric neuraminidase inhibitor antiviral treatment should be considered to quickly limit potential human transmission.
New Test Could Speed Diagnosis
The newly approved diagnostic test – the CDC Human Influenza Virus Real-Time RT-PCR Diagnostic Panel – utilizes a three-module design that incorporates and streamlines previous versions of the CDC’s two separate FDA-cleared diagnostic test kits, according to a CDC statement.
The first module identifies and distinguishes influenza A and B viruses. The second module classifies influenza A viruses by subtype, such as H1N1, H3N2, or 2009 H1N1. The third module detects highly pathogenic avian influenza A (H5N1) viral infection in human respiratory tract specimens.
CDC officials are hopeful that the test, which uses specimens from a patient’s upper or lower respiratory tract, will improve the ability to make quick diagnoses.
"As the spread of the H1N1 pandemic slowed last year, we conducted an end-to-end review of our nation’s medical countermeasure enterprise, which showed a clear need for better diagnostic tests," Dr. Nicole Lurie, assistant secretary for preparedness and response in the Department of Health and Human Services, said in a statement.
"In helping public health officials quickly identify seasonal flu as well as the flu viruses that could become pandemic, this kit can make a real difference in protecting health and saving lives in the United States and around the world," she added.
An H3N2 virus is one of three vaccine viruses selected by the FDA for inclusion in the 2011-2012 U.S. seasonal flu vaccine, based on recommendations from the World Health Organization. The other two in the trivalent vaccine are an influenza A (H1N1) virus and an influenza B virus.
FROM MORBIDITY AND MORTALITY WEEKLY REPORT
Substantial Polypharmacy Common Among Pediatric Inpatients
Polypharmacy exposure is exceedingly common in hospitalized pediatric patients, particularly those with rare conditions, according to findings from a large retrospective cohort study involving nearly 600,000 children.
On their first day of hospitalization, children’s hospital patients younger than 1 year, for example, received a median of four distinct medications, and general hospital patients received a median of three. Among those at the 90th percentile of daily exposure to distinct medications, the median numbers were 11 and 8 for children’s hospital and general hospital patients, respectively, Dr. Chris Feudtner and his colleagues of the Children’s Hospital of Philadelphia reported online Sept. 5 in Archives of Pediatrics and Adolescent Medicine (2011 [doi:10.1001/archpediatrics.2011.161]).
Among those older than 1 year, the median number of medications received on day 1 of hospitalization was 5 for both children’s and general hospital patients, and among those at the 90th percentile of daily exposure to distinct medications, the median numbers were 13 and 12 for children’s and general hospital patients, respectively.
The median cumulative number of distinct generic drug and therapeutic agent exposures for each successive day of hospitalization for those under age 1 year increased from 4 on the first hospital day to 25 by day 30 (for those who remained hospitalized), and for those in the 90th percentile of median cumulative exposure the number increased from 11 on the first hospital day to 51 by hospital day 30. For those over age 1 year, the median cumulative exposure increased from 5 to 42 by day 30, and for those in the 90th percentile of median cumulative exposure, the number increased from 13 to 66 by day 30.
Although the types of exposures varied by age and hospital type, the most common exposures were to acetaminophen, albuterol, antibiotics, fentanyl, heparin, ibuprofen, morphine, ondansetron, propofol, and ranitidine. Higher rates of exposures were shown to be significantly more likely to occur in patients with rare conditions, regardless of hospital type, and the differences in the number of medications used by the hospitals were found to reflect the underlying differences in their patient populations, the investigators noted.
The findings are based on data from the Pediatric Health Information System and the Perspective Data Warehouse, including 587,427 patients hospitalized during 2006 at any of 411 general hospitals and 52 children’s hospitals across the United States. These hospitalizations represent about one-fifth of all pediatric hospitalizations in the United States.
A total of 365,868 patients experienced 491,451 hospitalizations in the children’s hospitals, and a total of 221,559 experienced 260,740 hospitalizations in the general hospitals. Patients were under age 18 years; healthy newborns were excluded.
Despite limitations of the study – such as lack of certainty about the accuracy of data and about whether patients actually received the prescribed medications, as well as lack of information on the indication for which the medications were prescribed – the findings offer at least three important means of improving pediatric inpatient medication efficacy, effectiveness, and safety, the investigators said. For example, the ranking of medications based on the prevalence of exposures can guide prioritization of further research.
"Second, the level of polypharmacy exposure found in this study raises patient safety concerns, given the relationship between polypharmacy and adverse drug events that has been documented for adults in both hospital and nursing home settings," the authors noted. They added that "the common exposure to polypharmacy among hospitalized children motivates investigation of combinations of drugs and therapeutic agents to which patients are exposed and studies to detect potentially adverse drug-drug interactions, especially for patients in intensive care unit settings and patients with complex chronic conditions, who are at higher risk of polypharmacy."
Also, the databases used for the study "offer the potential to conduct rigorous comparative effectiveness and safety studies," they said.
The study authors had no disclosures to report.
Since Food and Drug Administration approval generally only requires the testing of agents in isolation, uncertainty about the effects of polypharmacy is the norm. Given the paucity of data on efficacy and safety in the pediatric population, this uncertainly is magnified when it comes to the care of children. Nonetheless, as this study illustrates, polypharmacy is common in hospitalized infants and children, Dr. Nancy E. Morden and Dr. David Goodman noted.
"We share the authors’ unease with the level of polypharmacy in pediatric inpatient settings, but our biggest concern is the lack of information that would tell us if these prescribing patterns are problematic for patients," they wrote.
Data are needed to help clinicians find the appropriate balance between alleviating symptoms and avoiding medication overuse; prior research, for example, suggests that pain is too often undertreated, while antimicrobials and respiratory agents are too often overused, they noted.
Underscoring this point is the extent of variations in Diagnosis Related Group (DRG)-specific drug exposures seen across hospitals in this study, which suggests a lack of consensus among clinicians about medication prescribing for this population.
"Studying the variation in further detail will help to identify concerning patterns of prescribing and guide further research and clinical care improvement," they said, noting the findings could improve understanding of the health impact of pediatric polypharmacy, improve outcomes, and decrease costs.
Research should also address polypharmacy effects in outpatient and chronic care settings, they said.
"Clinicians, policy makers, researchers, and patient advocates should collaborate on efforts aimed at promoting the development of evidence to inform pediatric pharmacotherapy. Moving toward an understanding of the balance of risks and benefits will require better data, a sustained research program, and commitment to translating new knowledge into better practice," they wrote.
Dr. Morden and Dr. Goodman are with the Dartmouth Institute for Health Policy and Clinical Practice at Dartmouth Medical School, Lebanon, N.H. They made their comments in an accompanying editorial (Arch. Pediatr. Adolesc. Med. 2011 [doi:10.1001/archpediatrics.2011.162]). Dr. Goodman made numerous financial disclosures relating to speaking fees, consulting fees, and research funding. Dr. Morden made no financial disclosures.
Since Food and Drug Administration approval generally only requires the testing of agents in isolation, uncertainty about the effects of polypharmacy is the norm. Given the paucity of data on efficacy and safety in the pediatric population, this uncertainly is magnified when it comes to the care of children. Nonetheless, as this study illustrates, polypharmacy is common in hospitalized infants and children, Dr. Nancy E. Morden and Dr. David Goodman noted.
"We share the authors’ unease with the level of polypharmacy in pediatric inpatient settings, but our biggest concern is the lack of information that would tell us if these prescribing patterns are problematic for patients," they wrote.
Data are needed to help clinicians find the appropriate balance between alleviating symptoms and avoiding medication overuse; prior research, for example, suggests that pain is too often undertreated, while antimicrobials and respiratory agents are too often overused, they noted.
Underscoring this point is the extent of variations in Diagnosis Related Group (DRG)-specific drug exposures seen across hospitals in this study, which suggests a lack of consensus among clinicians about medication prescribing for this population.
"Studying the variation in further detail will help to identify concerning patterns of prescribing and guide further research and clinical care improvement," they said, noting the findings could improve understanding of the health impact of pediatric polypharmacy, improve outcomes, and decrease costs.
Research should also address polypharmacy effects in outpatient and chronic care settings, they said.
"Clinicians, policy makers, researchers, and patient advocates should collaborate on efforts aimed at promoting the development of evidence to inform pediatric pharmacotherapy. Moving toward an understanding of the balance of risks and benefits will require better data, a sustained research program, and commitment to translating new knowledge into better practice," they wrote.
Dr. Morden and Dr. Goodman are with the Dartmouth Institute for Health Policy and Clinical Practice at Dartmouth Medical School, Lebanon, N.H. They made their comments in an accompanying editorial (Arch. Pediatr. Adolesc. Med. 2011 [doi:10.1001/archpediatrics.2011.162]). Dr. Goodman made numerous financial disclosures relating to speaking fees, consulting fees, and research funding. Dr. Morden made no financial disclosures.
Since Food and Drug Administration approval generally only requires the testing of agents in isolation, uncertainty about the effects of polypharmacy is the norm. Given the paucity of data on efficacy and safety in the pediatric population, this uncertainly is magnified when it comes to the care of children. Nonetheless, as this study illustrates, polypharmacy is common in hospitalized infants and children, Dr. Nancy E. Morden and Dr. David Goodman noted.
"We share the authors’ unease with the level of polypharmacy in pediatric inpatient settings, but our biggest concern is the lack of information that would tell us if these prescribing patterns are problematic for patients," they wrote.
Data are needed to help clinicians find the appropriate balance between alleviating symptoms and avoiding medication overuse; prior research, for example, suggests that pain is too often undertreated, while antimicrobials and respiratory agents are too often overused, they noted.
Underscoring this point is the extent of variations in Diagnosis Related Group (DRG)-specific drug exposures seen across hospitals in this study, which suggests a lack of consensus among clinicians about medication prescribing for this population.
"Studying the variation in further detail will help to identify concerning patterns of prescribing and guide further research and clinical care improvement," they said, noting the findings could improve understanding of the health impact of pediatric polypharmacy, improve outcomes, and decrease costs.
Research should also address polypharmacy effects in outpatient and chronic care settings, they said.
"Clinicians, policy makers, researchers, and patient advocates should collaborate on efforts aimed at promoting the development of evidence to inform pediatric pharmacotherapy. Moving toward an understanding of the balance of risks and benefits will require better data, a sustained research program, and commitment to translating new knowledge into better practice," they wrote.
Dr. Morden and Dr. Goodman are with the Dartmouth Institute for Health Policy and Clinical Practice at Dartmouth Medical School, Lebanon, N.H. They made their comments in an accompanying editorial (Arch. Pediatr. Adolesc. Med. 2011 [doi:10.1001/archpediatrics.2011.162]). Dr. Goodman made numerous financial disclosures relating to speaking fees, consulting fees, and research funding. Dr. Morden made no financial disclosures.
Polypharmacy exposure is exceedingly common in hospitalized pediatric patients, particularly those with rare conditions, according to findings from a large retrospective cohort study involving nearly 600,000 children.
On their first day of hospitalization, children’s hospital patients younger than 1 year, for example, received a median of four distinct medications, and general hospital patients received a median of three. Among those at the 90th percentile of daily exposure to distinct medications, the median numbers were 11 and 8 for children’s hospital and general hospital patients, respectively, Dr. Chris Feudtner and his colleagues of the Children’s Hospital of Philadelphia reported online Sept. 5 in Archives of Pediatrics and Adolescent Medicine (2011 [doi:10.1001/archpediatrics.2011.161]).
Among those older than 1 year, the median number of medications received on day 1 of hospitalization was 5 for both children’s and general hospital patients, and among those at the 90th percentile of daily exposure to distinct medications, the median numbers were 13 and 12 for children’s and general hospital patients, respectively.
The median cumulative number of distinct generic drug and therapeutic agent exposures for each successive day of hospitalization for those under age 1 year increased from 4 on the first hospital day to 25 by day 30 (for those who remained hospitalized), and for those in the 90th percentile of median cumulative exposure the number increased from 11 on the first hospital day to 51 by hospital day 30. For those over age 1 year, the median cumulative exposure increased from 5 to 42 by day 30, and for those in the 90th percentile of median cumulative exposure, the number increased from 13 to 66 by day 30.
Although the types of exposures varied by age and hospital type, the most common exposures were to acetaminophen, albuterol, antibiotics, fentanyl, heparin, ibuprofen, morphine, ondansetron, propofol, and ranitidine. Higher rates of exposures were shown to be significantly more likely to occur in patients with rare conditions, regardless of hospital type, and the differences in the number of medications used by the hospitals were found to reflect the underlying differences in their patient populations, the investigators noted.
The findings are based on data from the Pediatric Health Information System and the Perspective Data Warehouse, including 587,427 patients hospitalized during 2006 at any of 411 general hospitals and 52 children’s hospitals across the United States. These hospitalizations represent about one-fifth of all pediatric hospitalizations in the United States.
A total of 365,868 patients experienced 491,451 hospitalizations in the children’s hospitals, and a total of 221,559 experienced 260,740 hospitalizations in the general hospitals. Patients were under age 18 years; healthy newborns were excluded.
Despite limitations of the study – such as lack of certainty about the accuracy of data and about whether patients actually received the prescribed medications, as well as lack of information on the indication for which the medications were prescribed – the findings offer at least three important means of improving pediatric inpatient medication efficacy, effectiveness, and safety, the investigators said. For example, the ranking of medications based on the prevalence of exposures can guide prioritization of further research.
"Second, the level of polypharmacy exposure found in this study raises patient safety concerns, given the relationship between polypharmacy and adverse drug events that has been documented for adults in both hospital and nursing home settings," the authors noted. They added that "the common exposure to polypharmacy among hospitalized children motivates investigation of combinations of drugs and therapeutic agents to which patients are exposed and studies to detect potentially adverse drug-drug interactions, especially for patients in intensive care unit settings and patients with complex chronic conditions, who are at higher risk of polypharmacy."
Also, the databases used for the study "offer the potential to conduct rigorous comparative effectiveness and safety studies," they said.
The study authors had no disclosures to report.
Polypharmacy exposure is exceedingly common in hospitalized pediatric patients, particularly those with rare conditions, according to findings from a large retrospective cohort study involving nearly 600,000 children.
On their first day of hospitalization, children’s hospital patients younger than 1 year, for example, received a median of four distinct medications, and general hospital patients received a median of three. Among those at the 90th percentile of daily exposure to distinct medications, the median numbers were 11 and 8 for children’s hospital and general hospital patients, respectively, Dr. Chris Feudtner and his colleagues of the Children’s Hospital of Philadelphia reported online Sept. 5 in Archives of Pediatrics and Adolescent Medicine (2011 [doi:10.1001/archpediatrics.2011.161]).
Among those older than 1 year, the median number of medications received on day 1 of hospitalization was 5 for both children’s and general hospital patients, and among those at the 90th percentile of daily exposure to distinct medications, the median numbers were 13 and 12 for children’s and general hospital patients, respectively.
The median cumulative number of distinct generic drug and therapeutic agent exposures for each successive day of hospitalization for those under age 1 year increased from 4 on the first hospital day to 25 by day 30 (for those who remained hospitalized), and for those in the 90th percentile of median cumulative exposure the number increased from 11 on the first hospital day to 51 by hospital day 30. For those over age 1 year, the median cumulative exposure increased from 5 to 42 by day 30, and for those in the 90th percentile of median cumulative exposure, the number increased from 13 to 66 by day 30.
Although the types of exposures varied by age and hospital type, the most common exposures were to acetaminophen, albuterol, antibiotics, fentanyl, heparin, ibuprofen, morphine, ondansetron, propofol, and ranitidine. Higher rates of exposures were shown to be significantly more likely to occur in patients with rare conditions, regardless of hospital type, and the differences in the number of medications used by the hospitals were found to reflect the underlying differences in their patient populations, the investigators noted.
The findings are based on data from the Pediatric Health Information System and the Perspective Data Warehouse, including 587,427 patients hospitalized during 2006 at any of 411 general hospitals and 52 children’s hospitals across the United States. These hospitalizations represent about one-fifth of all pediatric hospitalizations in the United States.
A total of 365,868 patients experienced 491,451 hospitalizations in the children’s hospitals, and a total of 221,559 experienced 260,740 hospitalizations in the general hospitals. Patients were under age 18 years; healthy newborns were excluded.
Despite limitations of the study – such as lack of certainty about the accuracy of data and about whether patients actually received the prescribed medications, as well as lack of information on the indication for which the medications were prescribed – the findings offer at least three important means of improving pediatric inpatient medication efficacy, effectiveness, and safety, the investigators said. For example, the ranking of medications based on the prevalence of exposures can guide prioritization of further research.
"Second, the level of polypharmacy exposure found in this study raises patient safety concerns, given the relationship between polypharmacy and adverse drug events that has been documented for adults in both hospital and nursing home settings," the authors noted. They added that "the common exposure to polypharmacy among hospitalized children motivates investigation of combinations of drugs and therapeutic agents to which patients are exposed and studies to detect potentially adverse drug-drug interactions, especially for patients in intensive care unit settings and patients with complex chronic conditions, who are at higher risk of polypharmacy."
Also, the databases used for the study "offer the potential to conduct rigorous comparative effectiveness and safety studies," they said.
The study authors had no disclosures to report.
FROM ARCHIVES OF PEDIATRICS AND ADOLESCENT MEDICINE
Major Finding: The median cumulative number of distinct generic drug and therapeutic agent exposures for each successive day of hospitalization for patients under age 1 year increased from 4 on the first hospital day to 25 by day 30 (for those who remained hospitalized), and for patients in the 90th percentile of median cumulative exposure, the number increased from 11 on the first hospital day to 51 by hospital day 30. For those over age 1 year, the median cumulative exposure increased from 5 to 42 by day 30, and for those in the 90th percentile of median cumulative exposure, the number increased from 13 to 66 by day 30.
Data Source: A large retrospective cohort study involving nearly 600,000 children.
Disclosures: The study authors had no disclosures to report.
Cerebrovascular Pathologies May Underlie Parkinsonian Signs in Elderly
Parkinsonian signs such as motor slowing, gait disturbance, and tremor in old age may be caused by underlying cerebrovascular pathologies that cannot be detected by conventional brain imaging, according to findings from a clinical-autopsy study involving 418 cases.
"Together these data suggest that a substantial portion of older people have brain tissue damage and small vessel disease that are unlikely to be detected before death and suggest that cerebrovascular disease may be an even larger public health challenge than currently estimated," Dr. Aron S. Buchman and his colleagues wrote in a report published Sept. 1 in Stroke: Journal of the American Heart Association.
Macroscopic infarcts were found in 150 (36%) of the 418 brains that were autopsied for the study, and 110 of those also had evidence of pathologies that can’t be detected by conventional imaging, including microinfarcts, arteriolosclerosis, or both.
Furthermore, those pathologies were detected in almost 30% of cases without macroscopic infarcts, according to Dr. Buchman, associate professor of neurological sciences at Rush University Medical Center, Chicago, and his coauthors.
Of the 268 patients without macroinfarcts, 33 (7.9%) had microinfarcts, 62 (14.8%) had arteriolosclerosis, and 24 (5.7%) had both microinfarcts and arteriolosclerosis.
All three cerebrovascular pathologies were related to global parkinsonian scores in unadjusted analyses, and in linear regression analyses, significant associations were found between higher global parkinsonian scores before death and macroscopic infarcts, particularly for multiple (three or more) macroscopic infarcts. This was true for both cortical and subcortical macroscopic infarcts (Stroke 2011 Sept. 1 [doi:10.1161/STROKEAHA.111.623462]).
On further analysis, multiple cortical infarcts, or a single subcortical macroscopic infarct, were related to a higher global parkinsonian score, the investigators said.
"Microinfarcts showed a trend for an association with global parkinsonian [score], but the effect was attenuated and no longer significant when controlling for macroscopic infarcts," they wrote.
After the researchers controlled for macroscopic infarcts, an association between multiple microscopic infarcts and higher global parkinsonism was found, particularly for three or more microscopic infarcts.
Cortical microinfarcts, but not subcortical microinfarcts, also were associated with a higher global parkinsonian score. The association was particularly pronounced for those with multiple cortical microinfarcts.
An association between global parkinsonian score and arteriolosclerosis was no longer statistically significant after accounting for infarcts, the investigators noted.
However, analyses of the relationships between individual parkinsonian signs and each of the three pathologies (arteriolosclerosis, microinfarcts, and macroinfarcts – particularly subcortical microinfarcts and macroinfarcts) was found to be independently and significantly associated with gait disturbance, they found.
This association was present for both single and multiple subcortical macroscopic infarcts, and for multiple subcortical microinfarcts.
The brain autopsies used for this study were from deceased participants from the Religious Order Study, a longitudinal clinical-pathological study of aging in community-dwelling nuns and priests who had no known dementia at enrollment, and who were evaluated for parkinsonian signs with the 26-item modified version of the motor portion of the United Parkinson’s Disease Rating Scale prior to death. The study involved more women (61%) than men, and participants had a mean global parkinsonian score of 18.6 and a mean age of 88.5 years at death.
The findings are important because mild parkinsonian signs are common in community-dwelling older adults, affecting up to 50% of those aged 85 and older without known neurological disease, and are the source of significant morbidity and mortality in many of those affected, the investigators said.
They also noted that the findings could have important translational implications: They raise the question of whether improved public health strategies to increase prevention and more aggressively treat vascular risk factors and disease before death could decrease the burden of mild age-related parkinsonian signs; they suggest that the contribution of cerebrovascular disease to loss of motor function in older age is likely underestimated; and they suggest – given that small vessel disease was shown to have a separate relationship with parkinsonian gait – that there are either structural or functional brain tissue changes other than infarcts that might also be contributing to parkinsonian signs.
"Further clinical-pathological-imaging studies will be needed to delineate antemortem imaging markers as well as the pathophysiology of both microinfarcts and small vessel disease in the development of motor impairment in old age," they wrote.
The study was funded by National Institutes of Health grants and the Illinois Department of Public Health. The authors had no disclosures.
Macroscopic infarcts, arteriolosclerosis,
Parkinsonian signs such as motor slowing, gait disturbance, and tremor in old age may be caused by underlying cerebrovascular pathologies that cannot be detected by conventional brain imaging, according to findings from a clinical-autopsy study involving 418 cases.
"Together these data suggest that a substantial portion of older people have brain tissue damage and small vessel disease that are unlikely to be detected before death and suggest that cerebrovascular disease may be an even larger public health challenge than currently estimated," Dr. Aron S. Buchman and his colleagues wrote in a report published Sept. 1 in Stroke: Journal of the American Heart Association.
Macroscopic infarcts were found in 150 (36%) of the 418 brains that were autopsied for the study, and 110 of those also had evidence of pathologies that can’t be detected by conventional imaging, including microinfarcts, arteriolosclerosis, or both.
Furthermore, those pathologies were detected in almost 30% of cases without macroscopic infarcts, according to Dr. Buchman, associate professor of neurological sciences at Rush University Medical Center, Chicago, and his coauthors.
Of the 268 patients without macroinfarcts, 33 (7.9%) had microinfarcts, 62 (14.8%) had arteriolosclerosis, and 24 (5.7%) had both microinfarcts and arteriolosclerosis.
All three cerebrovascular pathologies were related to global parkinsonian scores in unadjusted analyses, and in linear regression analyses, significant associations were found between higher global parkinsonian scores before death and macroscopic infarcts, particularly for multiple (three or more) macroscopic infarcts. This was true for both cortical and subcortical macroscopic infarcts (Stroke 2011 Sept. 1 [doi:10.1161/STROKEAHA.111.623462]).
On further analysis, multiple cortical infarcts, or a single subcortical macroscopic infarct, were related to a higher global parkinsonian score, the investigators said.
"Microinfarcts showed a trend for an association with global parkinsonian [score], but the effect was attenuated and no longer significant when controlling for macroscopic infarcts," they wrote.
After the researchers controlled for macroscopic infarcts, an association between multiple microscopic infarcts and higher global parkinsonism was found, particularly for three or more microscopic infarcts.
Cortical microinfarcts, but not subcortical microinfarcts, also were associated with a higher global parkinsonian score. The association was particularly pronounced for those with multiple cortical microinfarcts.
An association between global parkinsonian score and arteriolosclerosis was no longer statistically significant after accounting for infarcts, the investigators noted.
However, analyses of the relationships between individual parkinsonian signs and each of the three pathologies (arteriolosclerosis, microinfarcts, and macroinfarcts – particularly subcortical microinfarcts and macroinfarcts) was found to be independently and significantly associated with gait disturbance, they found.
This association was present for both single and multiple subcortical macroscopic infarcts, and for multiple subcortical microinfarcts.
The brain autopsies used for this study were from deceased participants from the Religious Order Study, a longitudinal clinical-pathological study of aging in community-dwelling nuns and priests who had no known dementia at enrollment, and who were evaluated for parkinsonian signs with the 26-item modified version of the motor portion of the United Parkinson’s Disease Rating Scale prior to death. The study involved more women (61%) than men, and participants had a mean global parkinsonian score of 18.6 and a mean age of 88.5 years at death.
The findings are important because mild parkinsonian signs are common in community-dwelling older adults, affecting up to 50% of those aged 85 and older without known neurological disease, and are the source of significant morbidity and mortality in many of those affected, the investigators said.
They also noted that the findings could have important translational implications: They raise the question of whether improved public health strategies to increase prevention and more aggressively treat vascular risk factors and disease before death could decrease the burden of mild age-related parkinsonian signs; they suggest that the contribution of cerebrovascular disease to loss of motor function in older age is likely underestimated; and they suggest – given that small vessel disease was shown to have a separate relationship with parkinsonian gait – that there are either structural or functional brain tissue changes other than infarcts that might also be contributing to parkinsonian signs.
"Further clinical-pathological-imaging studies will be needed to delineate antemortem imaging markers as well as the pathophysiology of both microinfarcts and small vessel disease in the development of motor impairment in old age," they wrote.
The study was funded by National Institutes of Health grants and the Illinois Department of Public Health. The authors had no disclosures.
Parkinsonian signs such as motor slowing, gait disturbance, and tremor in old age may be caused by underlying cerebrovascular pathologies that cannot be detected by conventional brain imaging, according to findings from a clinical-autopsy study involving 418 cases.
"Together these data suggest that a substantial portion of older people have brain tissue damage and small vessel disease that are unlikely to be detected before death and suggest that cerebrovascular disease may be an even larger public health challenge than currently estimated," Dr. Aron S. Buchman and his colleagues wrote in a report published Sept. 1 in Stroke: Journal of the American Heart Association.
Macroscopic infarcts were found in 150 (36%) of the 418 brains that were autopsied for the study, and 110 of those also had evidence of pathologies that can’t be detected by conventional imaging, including microinfarcts, arteriolosclerosis, or both.
Furthermore, those pathologies were detected in almost 30% of cases without macroscopic infarcts, according to Dr. Buchman, associate professor of neurological sciences at Rush University Medical Center, Chicago, and his coauthors.
Of the 268 patients without macroinfarcts, 33 (7.9%) had microinfarcts, 62 (14.8%) had arteriolosclerosis, and 24 (5.7%) had both microinfarcts and arteriolosclerosis.
All three cerebrovascular pathologies were related to global parkinsonian scores in unadjusted analyses, and in linear regression analyses, significant associations were found between higher global parkinsonian scores before death and macroscopic infarcts, particularly for multiple (three or more) macroscopic infarcts. This was true for both cortical and subcortical macroscopic infarcts (Stroke 2011 Sept. 1 [doi:10.1161/STROKEAHA.111.623462]).
On further analysis, multiple cortical infarcts, or a single subcortical macroscopic infarct, were related to a higher global parkinsonian score, the investigators said.
"Microinfarcts showed a trend for an association with global parkinsonian [score], but the effect was attenuated and no longer significant when controlling for macroscopic infarcts," they wrote.
After the researchers controlled for macroscopic infarcts, an association between multiple microscopic infarcts and higher global parkinsonism was found, particularly for three or more microscopic infarcts.
Cortical microinfarcts, but not subcortical microinfarcts, also were associated with a higher global parkinsonian score. The association was particularly pronounced for those with multiple cortical microinfarcts.
An association between global parkinsonian score and arteriolosclerosis was no longer statistically significant after accounting for infarcts, the investigators noted.
However, analyses of the relationships between individual parkinsonian signs and each of the three pathologies (arteriolosclerosis, microinfarcts, and macroinfarcts – particularly subcortical microinfarcts and macroinfarcts) was found to be independently and significantly associated with gait disturbance, they found.
This association was present for both single and multiple subcortical macroscopic infarcts, and for multiple subcortical microinfarcts.
The brain autopsies used for this study were from deceased participants from the Religious Order Study, a longitudinal clinical-pathological study of aging in community-dwelling nuns and priests who had no known dementia at enrollment, and who were evaluated for parkinsonian signs with the 26-item modified version of the motor portion of the United Parkinson’s Disease Rating Scale prior to death. The study involved more women (61%) than men, and participants had a mean global parkinsonian score of 18.6 and a mean age of 88.5 years at death.
The findings are important because mild parkinsonian signs are common in community-dwelling older adults, affecting up to 50% of those aged 85 and older without known neurological disease, and are the source of significant morbidity and mortality in many of those affected, the investigators said.
They also noted that the findings could have important translational implications: They raise the question of whether improved public health strategies to increase prevention and more aggressively treat vascular risk factors and disease before death could decrease the burden of mild age-related parkinsonian signs; they suggest that the contribution of cerebrovascular disease to loss of motor function in older age is likely underestimated; and they suggest – given that small vessel disease was shown to have a separate relationship with parkinsonian gait – that there are either structural or functional brain tissue changes other than infarcts that might also be contributing to parkinsonian signs.
"Further clinical-pathological-imaging studies will be needed to delineate antemortem imaging markers as well as the pathophysiology of both microinfarcts and small vessel disease in the development of motor impairment in old age," they wrote.
The study was funded by National Institutes of Health grants and the Illinois Department of Public Health. The authors had no disclosures.
Macroscopic infarcts, arteriolosclerosis,
Macroscopic infarcts, arteriolosclerosis,
FROM STROKE: JOURNAL OF THE AMERICAN HEART ASSOCIATION
Major Finding: Macroscopic infarcts were found in 150 (36%) of the brains that were autopsied for the study, and 110 also had evidence of pathologies that can’t be detected by conventional imaging, including microinfarcts, arteriosclerosis, or both.
Data Source: Clinical-autopsy data from 418 participants in the Religious Order Study.
Disclosures: This study was funded by NIH grants and the Illinois Department of Public Health. The authors had no disclosures.
HPV-16, -18 Testing Outperforms Cytology : Liquid-based cytology added little to cobas HPV testing for detecting CIN 3 or worse.
Major Finding: The cobas HPV test was significantly more sensitive for detecting CIN 3 or worse than was liquid-based cytology at a threshold of ASCUS or worse (92% vs. 53.3%). Adding cytology to HPV testing increased the sensitivity for detection of CIN 3 or worse by less than 5% (from 92% to 96.7%), but increased the number of screen positives by more than 35%.
Data Source: A subanalysis of the ATHENA study population to compare the screening performance of the cobas HPV test versus liquid-based cytology for cervical cancer screening.
Disclosures: The study was funded by Roche Molecular Systems. Dr. Castle said he has a nondisclosure agreement to work with Roche on the analysis of their clinical trial but receives no financial compensation. Other authors on the study disclosed that they are employed by Roche Molecular Systems and/or have stock or stock options in the company, or that they have received consulting fees, honoraria, and/or other compensation from Roche, BD Diagnostics, Qiagen, Gen-Probe, Ventana, and/or Merck.
Cobas human papillomavirus testing with individual human papillomavirus types 16 and 18 detection could serve as a more sensitive and more efficient approach to cervical cancer screening than traditional liquid-based cytology methods alone, according to a subanalysis of data from the ATHENA study.
The findings of the current analysis have implications for the development of strategies – including triage to colposcopy – for managing HPV-positive women, according to Philip E. Castle, Ph.D., of the American Society for Clinical Pathology Institute, Washington, and his colleagues.
The investigators analyzed data from 41,955 women aged 25 years and older who were part of the ATHENA (Addressing the Need for Advanced HPV Diagnostics) study, which was designed to assess the performance of HPV testing and HPV-16 and HPV-18 genotyping, compared with liquid-based cytology for cervical cancer screening. Of 40,901 women who had valid cobas HPV and liquid-based cytology test results available, 10% (4,275) tested HPV positive and 6% (2,617) had abnormal cytology; of these, 1.1 % (431) were diagnosed with cervical intraepithelial neoplasia grade 2 (CIN 2) or worse, and 0.7% (274) were diagnosed with CIN 3 or worse.
An analysis of the results of colposcopy, which was performed in 2,609 women found to have atypical squamous cells of undetermined significance (ASCUS) or worse cytology, 5,726 women with a positive HPV test by either of the first-generation HPV DNA assays used in the study (Amplicor HPV test and Linear Array HPG genotyping), and 1,041 women who were HPV negative and who had negative for intraepithelial or malignant (NILM) cervical cytology, showed that the cobas HPV test was significantly more sensitive for detecting CIN 3 or worse than was liquid-based cytology at a threshold of ASCUS or worse (92% vs. 53.3%), according to the investigators (Lancet Oncol. 2011; 12:880-90).
The addition of cytology to HPV testing increased the sensitivity for detection of CIN 3 or worse by less than 5% (from 92% to 96.7%), but increased the number of screen positives by more than 35%, Dr. Castle and his associates said.
When used as a triage test for identifying high-grade CIN (grade 3 or higher), the detection of HPV-16, HPV-18, or both alone was statistically equivalent to the detection of ASCUS or worse alone in terms of both sensitivity (59.5% and 52.8%, respectively) and positive predictive value (15.5% and 14.1%), they noted.
Sensitivity was further increased – and the positive predictive value (PPV) decreased – by use of HPV-16, HPV-18, or both as an additional or complementary triage strategy to ASCUS or worse, they said.
The authors added that “notably, testing positive for HPV-16, HPV-18, or both had a sensitivity of 53.8% … and [a] PPV of 10.2% … for CIN 3 or worse in women aged 25 years or older who were HPV positive and had NILM cytology.”
In addition, the use of a threshold of low-grade squamous intraepithelial lesion (LSIL) or worse with HPV-16, HPV-18, or both was more sensitive than detection of ASCUS or worse alone with similar PPV, and detection of high-grade squamous intraepithelial lesion (HSIL) or worse with HPV-16, HPV-18, or both had a higher sensitivity and PPV than ASCUS or worse alone.
The study findings suggest that that the use of HPV testing as the primary screening test to rule out cervical disease, along with a specific test such as liquid-based cytology to help determine which women should be sent for immediate colposcopy, is a rational approach.
The findings also support the premise that co-testing has little benefit over HPV testing alone, the investigators noted.
“However, until clinicians become comfortable with the use of HPV as a first-line test, they might initially favor co-testing, and so co-testing could have an underlying merit that is difficult to quantify,” Dr. Castle and his associates wrote.
In addition, “the decision to switch from co-testing to HPV testing alone, and the intervals between screenings, will ultimately depend on clinicians' perceptions of acceptable risks,” they said.
“Nevertheless, on the basis of our findings, we suggest that detection of HPV-16, HPV-18, or both combined with a raised threshold of abnormal cervical cytology (LSIL or worse) might be preferable to the existing recommendations for management of HPV-positive women,” according to the investigators.
They noted that testing and genotyping for HPV-16, HPV-18, or both – with or without liquid-based cytology – can provide potentially cost-effective and safe cervical cancer screening.
“Because the HPV-16 and HPV-18 readouts for the cobas HPV test are provided concurrently with the pooled detection of other carcinogenic HPV genotypes,” the use of this test to triage HPV-positive women for colposcopy could prove much more efficient than cytology, they added.
Also, a strategy of applying cytology reflexively to those who are HPV positive without HPV-16 or HPV-18 genotype, with referral to colposcopy only if they have LSIL or HSIL, or worse, would increase the sensitivity for detection of CIN 3 or worse in HPV-positive women to a level above that provided by HPV-16 and HPV-18, or both, without sacrificing good PPV, they said. The comparative performance and cost-effectiveness of various strategies will need to be assessed in future studies to identify best practices, they noted.
View on The News
Standalone HPV Testing as Primary Cervical Cancer Screening?
A cervical cancer screening strategy that allows immediate identification of all women with lesions needing treatment would be preferable to the current approach of rescreening HPV-positive women with normal cytology at 1 year, with colposcopy performed if infection is still present or if cytology has become abnormal.
“Unfortunately, all combinations of genotyping and cytology in Castle and colleagues' study had less than 80% sensitivity, leading the investigators to recommend test repetitions after 1 year,” Dr. Guglielmo Ronco and his colleagues wrote in an accompanying editorial (Lancet Oncol. 2011;12:831-2).
Still, the increased sensitivity provided by the combined triage tests would allow some CIN 3 or worse lesions to be detected earlier, they noted.
Furthermore, strategies using other biomarkers to triage HPV-positive women are currently being assessed; the cross-sectional sensitivity of immunochemistry for p16INK4a overexpression for CIN 3 or worse, for example, is 91%, which suggests that short-term retesting could be avoided in those who test negative for p16INK4a. Dr. Ronco and his colleagues warned, however, that since HPV-positive women are at increased risk for developing new lesions, premature reallocation to screening intervals as long as those recommended for HPV-negative women “might not be advisable.”
“Additional longitudinal data are needed to define the safest time interval before retesting in women with HPV infection who were negative for p16INK4a or any other triage test,” they wrote.
They also noted that the findings of this study, though designed for developed countries, can provide useful information about triage strategies for “countries where high-quality cytology has been difficult to implement and combinations of HPV tests might eventually offer a more sustainable option.”
DR. RONCO is with the Centre of Cancer Prevention in Turin, Italy. He and his coauthors said they had no relevant financial disclosures.
Major Finding: The cobas HPV test was significantly more sensitive for detecting CIN 3 or worse than was liquid-based cytology at a threshold of ASCUS or worse (92% vs. 53.3%). Adding cytology to HPV testing increased the sensitivity for detection of CIN 3 or worse by less than 5% (from 92% to 96.7%), but increased the number of screen positives by more than 35%.
Data Source: A subanalysis of the ATHENA study population to compare the screening performance of the cobas HPV test versus liquid-based cytology for cervical cancer screening.
Disclosures: The study was funded by Roche Molecular Systems. Dr. Castle said he has a nondisclosure agreement to work with Roche on the analysis of their clinical trial but receives no financial compensation. Other authors on the study disclosed that they are employed by Roche Molecular Systems and/or have stock or stock options in the company, or that they have received consulting fees, honoraria, and/or other compensation from Roche, BD Diagnostics, Qiagen, Gen-Probe, Ventana, and/or Merck.
Cobas human papillomavirus testing with individual human papillomavirus types 16 and 18 detection could serve as a more sensitive and more efficient approach to cervical cancer screening than traditional liquid-based cytology methods alone, according to a subanalysis of data from the ATHENA study.
The findings of the current analysis have implications for the development of strategies – including triage to colposcopy – for managing HPV-positive women, according to Philip E. Castle, Ph.D., of the American Society for Clinical Pathology Institute, Washington, and his colleagues.
The investigators analyzed data from 41,955 women aged 25 years and older who were part of the ATHENA (Addressing the Need for Advanced HPV Diagnostics) study, which was designed to assess the performance of HPV testing and HPV-16 and HPV-18 genotyping, compared with liquid-based cytology for cervical cancer screening. Of 40,901 women who had valid cobas HPV and liquid-based cytology test results available, 10% (4,275) tested HPV positive and 6% (2,617) had abnormal cytology; of these, 1.1 % (431) were diagnosed with cervical intraepithelial neoplasia grade 2 (CIN 2) or worse, and 0.7% (274) were diagnosed with CIN 3 or worse.
An analysis of the results of colposcopy, which was performed in 2,609 women found to have atypical squamous cells of undetermined significance (ASCUS) or worse cytology, 5,726 women with a positive HPV test by either of the first-generation HPV DNA assays used in the study (Amplicor HPV test and Linear Array HPG genotyping), and 1,041 women who were HPV negative and who had negative for intraepithelial or malignant (NILM) cervical cytology, showed that the cobas HPV test was significantly more sensitive for detecting CIN 3 or worse than was liquid-based cytology at a threshold of ASCUS or worse (92% vs. 53.3%), according to the investigators (Lancet Oncol. 2011; 12:880-90).
The addition of cytology to HPV testing increased the sensitivity for detection of CIN 3 or worse by less than 5% (from 92% to 96.7%), but increased the number of screen positives by more than 35%, Dr. Castle and his associates said.
When used as a triage test for identifying high-grade CIN (grade 3 or higher), the detection of HPV-16, HPV-18, or both alone was statistically equivalent to the detection of ASCUS or worse alone in terms of both sensitivity (59.5% and 52.8%, respectively) and positive predictive value (15.5% and 14.1%), they noted.
Sensitivity was further increased – and the positive predictive value (PPV) decreased – by use of HPV-16, HPV-18, or both as an additional or complementary triage strategy to ASCUS or worse, they said.
The authors added that “notably, testing positive for HPV-16, HPV-18, or both had a sensitivity of 53.8% … and [a] PPV of 10.2% … for CIN 3 or worse in women aged 25 years or older who were HPV positive and had NILM cytology.”
In addition, the use of a threshold of low-grade squamous intraepithelial lesion (LSIL) or worse with HPV-16, HPV-18, or both was more sensitive than detection of ASCUS or worse alone with similar PPV, and detection of high-grade squamous intraepithelial lesion (HSIL) or worse with HPV-16, HPV-18, or both had a higher sensitivity and PPV than ASCUS or worse alone.
The study findings suggest that that the use of HPV testing as the primary screening test to rule out cervical disease, along with a specific test such as liquid-based cytology to help determine which women should be sent for immediate colposcopy, is a rational approach.
The findings also support the premise that co-testing has little benefit over HPV testing alone, the investigators noted.
“However, until clinicians become comfortable with the use of HPV as a first-line test, they might initially favor co-testing, and so co-testing could have an underlying merit that is difficult to quantify,” Dr. Castle and his associates wrote.
In addition, “the decision to switch from co-testing to HPV testing alone, and the intervals between screenings, will ultimately depend on clinicians' perceptions of acceptable risks,” they said.
“Nevertheless, on the basis of our findings, we suggest that detection of HPV-16, HPV-18, or both combined with a raised threshold of abnormal cervical cytology (LSIL or worse) might be preferable to the existing recommendations for management of HPV-positive women,” according to the investigators.
They noted that testing and genotyping for HPV-16, HPV-18, or both – with or without liquid-based cytology – can provide potentially cost-effective and safe cervical cancer screening.
“Because the HPV-16 and HPV-18 readouts for the cobas HPV test are provided concurrently with the pooled detection of other carcinogenic HPV genotypes,” the use of this test to triage HPV-positive women for colposcopy could prove much more efficient than cytology, they added.
Also, a strategy of applying cytology reflexively to those who are HPV positive without HPV-16 or HPV-18 genotype, with referral to colposcopy only if they have LSIL or HSIL, or worse, would increase the sensitivity for detection of CIN 3 or worse in HPV-positive women to a level above that provided by HPV-16 and HPV-18, or both, without sacrificing good PPV, they said. The comparative performance and cost-effectiveness of various strategies will need to be assessed in future studies to identify best practices, they noted.
View on The News
Standalone HPV Testing as Primary Cervical Cancer Screening?
A cervical cancer screening strategy that allows immediate identification of all women with lesions needing treatment would be preferable to the current approach of rescreening HPV-positive women with normal cytology at 1 year, with colposcopy performed if infection is still present or if cytology has become abnormal.
“Unfortunately, all combinations of genotyping and cytology in Castle and colleagues' study had less than 80% sensitivity, leading the investigators to recommend test repetitions after 1 year,” Dr. Guglielmo Ronco and his colleagues wrote in an accompanying editorial (Lancet Oncol. 2011;12:831-2).
Still, the increased sensitivity provided by the combined triage tests would allow some CIN 3 or worse lesions to be detected earlier, they noted.
Furthermore, strategies using other biomarkers to triage HPV-positive women are currently being assessed; the cross-sectional sensitivity of immunochemistry for p16INK4a overexpression for CIN 3 or worse, for example, is 91%, which suggests that short-term retesting could be avoided in those who test negative for p16INK4a. Dr. Ronco and his colleagues warned, however, that since HPV-positive women are at increased risk for developing new lesions, premature reallocation to screening intervals as long as those recommended for HPV-negative women “might not be advisable.”
“Additional longitudinal data are needed to define the safest time interval before retesting in women with HPV infection who were negative for p16INK4a or any other triage test,” they wrote.
They also noted that the findings of this study, though designed for developed countries, can provide useful information about triage strategies for “countries where high-quality cytology has been difficult to implement and combinations of HPV tests might eventually offer a more sustainable option.”
DR. RONCO is with the Centre of Cancer Prevention in Turin, Italy. He and his coauthors said they had no relevant financial disclosures.
Major Finding: The cobas HPV test was significantly more sensitive for detecting CIN 3 or worse than was liquid-based cytology at a threshold of ASCUS or worse (92% vs. 53.3%). Adding cytology to HPV testing increased the sensitivity for detection of CIN 3 or worse by less than 5% (from 92% to 96.7%), but increased the number of screen positives by more than 35%.
Data Source: A subanalysis of the ATHENA study population to compare the screening performance of the cobas HPV test versus liquid-based cytology for cervical cancer screening.
Disclosures: The study was funded by Roche Molecular Systems. Dr. Castle said he has a nondisclosure agreement to work with Roche on the analysis of their clinical trial but receives no financial compensation. Other authors on the study disclosed that they are employed by Roche Molecular Systems and/or have stock or stock options in the company, or that they have received consulting fees, honoraria, and/or other compensation from Roche, BD Diagnostics, Qiagen, Gen-Probe, Ventana, and/or Merck.
Cobas human papillomavirus testing with individual human papillomavirus types 16 and 18 detection could serve as a more sensitive and more efficient approach to cervical cancer screening than traditional liquid-based cytology methods alone, according to a subanalysis of data from the ATHENA study.
The findings of the current analysis have implications for the development of strategies – including triage to colposcopy – for managing HPV-positive women, according to Philip E. Castle, Ph.D., of the American Society for Clinical Pathology Institute, Washington, and his colleagues.
The investigators analyzed data from 41,955 women aged 25 years and older who were part of the ATHENA (Addressing the Need for Advanced HPV Diagnostics) study, which was designed to assess the performance of HPV testing and HPV-16 and HPV-18 genotyping, compared with liquid-based cytology for cervical cancer screening. Of 40,901 women who had valid cobas HPV and liquid-based cytology test results available, 10% (4,275) tested HPV positive and 6% (2,617) had abnormal cytology; of these, 1.1 % (431) were diagnosed with cervical intraepithelial neoplasia grade 2 (CIN 2) or worse, and 0.7% (274) were diagnosed with CIN 3 or worse.
An analysis of the results of colposcopy, which was performed in 2,609 women found to have atypical squamous cells of undetermined significance (ASCUS) or worse cytology, 5,726 women with a positive HPV test by either of the first-generation HPV DNA assays used in the study (Amplicor HPV test and Linear Array HPG genotyping), and 1,041 women who were HPV negative and who had negative for intraepithelial or malignant (NILM) cervical cytology, showed that the cobas HPV test was significantly more sensitive for detecting CIN 3 or worse than was liquid-based cytology at a threshold of ASCUS or worse (92% vs. 53.3%), according to the investigators (Lancet Oncol. 2011; 12:880-90).
The addition of cytology to HPV testing increased the sensitivity for detection of CIN 3 or worse by less than 5% (from 92% to 96.7%), but increased the number of screen positives by more than 35%, Dr. Castle and his associates said.
When used as a triage test for identifying high-grade CIN (grade 3 or higher), the detection of HPV-16, HPV-18, or both alone was statistically equivalent to the detection of ASCUS or worse alone in terms of both sensitivity (59.5% and 52.8%, respectively) and positive predictive value (15.5% and 14.1%), they noted.
Sensitivity was further increased – and the positive predictive value (PPV) decreased – by use of HPV-16, HPV-18, or both as an additional or complementary triage strategy to ASCUS or worse, they said.
The authors added that “notably, testing positive for HPV-16, HPV-18, or both had a sensitivity of 53.8% … and [a] PPV of 10.2% … for CIN 3 or worse in women aged 25 years or older who were HPV positive and had NILM cytology.”
In addition, the use of a threshold of low-grade squamous intraepithelial lesion (LSIL) or worse with HPV-16, HPV-18, or both was more sensitive than detection of ASCUS or worse alone with similar PPV, and detection of high-grade squamous intraepithelial lesion (HSIL) or worse with HPV-16, HPV-18, or both had a higher sensitivity and PPV than ASCUS or worse alone.
The study findings suggest that that the use of HPV testing as the primary screening test to rule out cervical disease, along with a specific test such as liquid-based cytology to help determine which women should be sent for immediate colposcopy, is a rational approach.
The findings also support the premise that co-testing has little benefit over HPV testing alone, the investigators noted.
“However, until clinicians become comfortable with the use of HPV as a first-line test, they might initially favor co-testing, and so co-testing could have an underlying merit that is difficult to quantify,” Dr. Castle and his associates wrote.
In addition, “the decision to switch from co-testing to HPV testing alone, and the intervals between screenings, will ultimately depend on clinicians' perceptions of acceptable risks,” they said.
“Nevertheless, on the basis of our findings, we suggest that detection of HPV-16, HPV-18, or both combined with a raised threshold of abnormal cervical cytology (LSIL or worse) might be preferable to the existing recommendations for management of HPV-positive women,” according to the investigators.
They noted that testing and genotyping for HPV-16, HPV-18, or both – with or without liquid-based cytology – can provide potentially cost-effective and safe cervical cancer screening.
“Because the HPV-16 and HPV-18 readouts for the cobas HPV test are provided concurrently with the pooled detection of other carcinogenic HPV genotypes,” the use of this test to triage HPV-positive women for colposcopy could prove much more efficient than cytology, they added.
Also, a strategy of applying cytology reflexively to those who are HPV positive without HPV-16 or HPV-18 genotype, with referral to colposcopy only if they have LSIL or HSIL, or worse, would increase the sensitivity for detection of CIN 3 or worse in HPV-positive women to a level above that provided by HPV-16 and HPV-18, or both, without sacrificing good PPV, they said. The comparative performance and cost-effectiveness of various strategies will need to be assessed in future studies to identify best practices, they noted.
View on The News
Standalone HPV Testing as Primary Cervical Cancer Screening?
A cervical cancer screening strategy that allows immediate identification of all women with lesions needing treatment would be preferable to the current approach of rescreening HPV-positive women with normal cytology at 1 year, with colposcopy performed if infection is still present or if cytology has become abnormal.
“Unfortunately, all combinations of genotyping and cytology in Castle and colleagues' study had less than 80% sensitivity, leading the investigators to recommend test repetitions after 1 year,” Dr. Guglielmo Ronco and his colleagues wrote in an accompanying editorial (Lancet Oncol. 2011;12:831-2).
Still, the increased sensitivity provided by the combined triage tests would allow some CIN 3 or worse lesions to be detected earlier, they noted.
Furthermore, strategies using other biomarkers to triage HPV-positive women are currently being assessed; the cross-sectional sensitivity of immunochemistry for p16INK4a overexpression for CIN 3 or worse, for example, is 91%, which suggests that short-term retesting could be avoided in those who test negative for p16INK4a. Dr. Ronco and his colleagues warned, however, that since HPV-positive women are at increased risk for developing new lesions, premature reallocation to screening intervals as long as those recommended for HPV-negative women “might not be advisable.”
“Additional longitudinal data are needed to define the safest time interval before retesting in women with HPV infection who were negative for p16INK4a or any other triage test,” they wrote.
They also noted that the findings of this study, though designed for developed countries, can provide useful information about triage strategies for “countries where high-quality cytology has been difficult to implement and combinations of HPV tests might eventually offer a more sustainable option.”
DR. RONCO is with the Centre of Cancer Prevention in Turin, Italy. He and his coauthors said they had no relevant financial disclosures.
From Lancet Oncology
BRCA2 Gene Mutation Tied to Better Survival in Ovarian Ca
Major Finding: The 5-year survival was 36% in those with no BRCA mutation, 46% in those with the BRCA1 mutation, and 61% in those with the BRCA2 mutation, after adjustment for stage, grade, histology, and age at diagnosis.
Data Source: A large, multicenter study investigating the impact of germline BRCA1 and BRCA2 mutations in 3,531 women with invasive epithelial ovarian cancer.
Disclosures: Ms. Bolton said she had no relevant financial disclosures.
ORLANDO – Ovarian cancer patients with BRCA1 or BRCA2 gene mutations have better survival than do those with neither mutation, and those with the BRCA2 mutation have better survival than do those with the BRCA1 mutation, according to the findings of a large, multicenter study.
The findings confirm the results of several prior smaller studies showing a survival advantage in mutation carriers vs. nonmutation carriers, and they provide the first direct evidence that BRCA1 and BRCA2 mutations have differing effects on survival, Kelly L. Bolton reported.
She and her colleagues studied 3,531 women with invasive epithelial ovarian cancer who were enrolled in one of 24 studies in the United States, Europe, Israel, and Asia, and for whom survival data were available. Included were 1,178 women with BRCA1, 367 with BRCA2, and 1,986 who were BRCA-negative.
The 5-year survival was 36% in those with no mutation, 46% of those with the BRCA1 mutation, and 61% of those with the BRCA2 mutation, after adjustment for stage, grade, histology, and age at diagnosis, said Ms. Bolton, a predoctoral fellow at the National Cancer Institute.
The difference in survival between the BRCA1 mutation carriers and those with no mutation was modest (hazard ratio, 0.84), although not statistically significant. However, the difference between the BRCA2 mutation carriers and both the noncarriers and the BRCA1 mutation carriers (after adjustment for age at diagnosis) did reach statistical significance (HR, 0.57 and 0.69, respectively;. Even after the exclusion of all but high-grade, advanced-stage serous cases, the survival differences persisted, Ms. Bolton reported.
A possible explanation for the differences, based on in vitro work and some retrospective trials, may lie in patients' responses to chemotherapy; those with the BRCA2 mutation may have an improved response, but unidentified biological differences among BRCA1 carriers, BRCA2 carriers, and noncarriers could also be driving the association, she said.
The BRCA1 and BRCA2 mutation carriers in this study did not differ in regard to tumor stage, grade, or histology. Compared with noncarriers, however, BRCA1 carriers were younger and BRCA2 carriers were older at diagnosis.
Furthermore, compared with noncarriers, BRCA1 and BRCA2 carriers were more likely to present with advanced-stage disease, high-grade disease and serous disease (
“The findings don't have any immediate impact on clinical practice, but they do have important implications [for both] clinical prediction and also trial design, particularly for clinical trials,” Ms. Bolton said, noting that although germline mutations in the BRCA1 and BRCA2 genes are rare in the general population, they are present in 10%-15% of those with ovarian cancer.
Major Finding: The 5-year survival was 36% in those with no BRCA mutation, 46% in those with the BRCA1 mutation, and 61% in those with the BRCA2 mutation, after adjustment for stage, grade, histology, and age at diagnosis.
Data Source: A large, multicenter study investigating the impact of germline BRCA1 and BRCA2 mutations in 3,531 women with invasive epithelial ovarian cancer.
Disclosures: Ms. Bolton said she had no relevant financial disclosures.
ORLANDO – Ovarian cancer patients with BRCA1 or BRCA2 gene mutations have better survival than do those with neither mutation, and those with the BRCA2 mutation have better survival than do those with the BRCA1 mutation, according to the findings of a large, multicenter study.
The findings confirm the results of several prior smaller studies showing a survival advantage in mutation carriers vs. nonmutation carriers, and they provide the first direct evidence that BRCA1 and BRCA2 mutations have differing effects on survival, Kelly L. Bolton reported.
She and her colleagues studied 3,531 women with invasive epithelial ovarian cancer who were enrolled in one of 24 studies in the United States, Europe, Israel, and Asia, and for whom survival data were available. Included were 1,178 women with BRCA1, 367 with BRCA2, and 1,986 who were BRCA-negative.
The 5-year survival was 36% in those with no mutation, 46% of those with the BRCA1 mutation, and 61% of those with the BRCA2 mutation, after adjustment for stage, grade, histology, and age at diagnosis, said Ms. Bolton, a predoctoral fellow at the National Cancer Institute.
The difference in survival between the BRCA1 mutation carriers and those with no mutation was modest (hazard ratio, 0.84), although not statistically significant. However, the difference between the BRCA2 mutation carriers and both the noncarriers and the BRCA1 mutation carriers (after adjustment for age at diagnosis) did reach statistical significance (HR, 0.57 and 0.69, respectively;. Even after the exclusion of all but high-grade, advanced-stage serous cases, the survival differences persisted, Ms. Bolton reported.
A possible explanation for the differences, based on in vitro work and some retrospective trials, may lie in patients' responses to chemotherapy; those with the BRCA2 mutation may have an improved response, but unidentified biological differences among BRCA1 carriers, BRCA2 carriers, and noncarriers could also be driving the association, she said.
The BRCA1 and BRCA2 mutation carriers in this study did not differ in regard to tumor stage, grade, or histology. Compared with noncarriers, however, BRCA1 carriers were younger and BRCA2 carriers were older at diagnosis.
Furthermore, compared with noncarriers, BRCA1 and BRCA2 carriers were more likely to present with advanced-stage disease, high-grade disease and serous disease (
“The findings don't have any immediate impact on clinical practice, but they do have important implications [for both] clinical prediction and also trial design, particularly for clinical trials,” Ms. Bolton said, noting that although germline mutations in the BRCA1 and BRCA2 genes are rare in the general population, they are present in 10%-15% of those with ovarian cancer.
Major Finding: The 5-year survival was 36% in those with no BRCA mutation, 46% in those with the BRCA1 mutation, and 61% in those with the BRCA2 mutation, after adjustment for stage, grade, histology, and age at diagnosis.
Data Source: A large, multicenter study investigating the impact of germline BRCA1 and BRCA2 mutations in 3,531 women with invasive epithelial ovarian cancer.
Disclosures: Ms. Bolton said she had no relevant financial disclosures.
ORLANDO – Ovarian cancer patients with BRCA1 or BRCA2 gene mutations have better survival than do those with neither mutation, and those with the BRCA2 mutation have better survival than do those with the BRCA1 mutation, according to the findings of a large, multicenter study.
The findings confirm the results of several prior smaller studies showing a survival advantage in mutation carriers vs. nonmutation carriers, and they provide the first direct evidence that BRCA1 and BRCA2 mutations have differing effects on survival, Kelly L. Bolton reported.
She and her colleagues studied 3,531 women with invasive epithelial ovarian cancer who were enrolled in one of 24 studies in the United States, Europe, Israel, and Asia, and for whom survival data were available. Included were 1,178 women with BRCA1, 367 with BRCA2, and 1,986 who were BRCA-negative.
The 5-year survival was 36% in those with no mutation, 46% of those with the BRCA1 mutation, and 61% of those with the BRCA2 mutation, after adjustment for stage, grade, histology, and age at diagnosis, said Ms. Bolton, a predoctoral fellow at the National Cancer Institute.
The difference in survival between the BRCA1 mutation carriers and those with no mutation was modest (hazard ratio, 0.84), although not statistically significant. However, the difference between the BRCA2 mutation carriers and both the noncarriers and the BRCA1 mutation carriers (after adjustment for age at diagnosis) did reach statistical significance (HR, 0.57 and 0.69, respectively;. Even after the exclusion of all but high-grade, advanced-stage serous cases, the survival differences persisted, Ms. Bolton reported.
A possible explanation for the differences, based on in vitro work and some retrospective trials, may lie in patients' responses to chemotherapy; those with the BRCA2 mutation may have an improved response, but unidentified biological differences among BRCA1 carriers, BRCA2 carriers, and noncarriers could also be driving the association, she said.
The BRCA1 and BRCA2 mutation carriers in this study did not differ in regard to tumor stage, grade, or histology. Compared with noncarriers, however, BRCA1 carriers were younger and BRCA2 carriers were older at diagnosis.
Furthermore, compared with noncarriers, BRCA1 and BRCA2 carriers were more likely to present with advanced-stage disease, high-grade disease and serous disease (
“The findings don't have any immediate impact on clinical practice, but they do have important implications [for both] clinical prediction and also trial design, particularly for clinical trials,” Ms. Bolton said, noting that although germline mutations in the BRCA1 and BRCA2 genes are rare in the general population, they are present in 10%-15% of those with ovarian cancer.
From the Annual Meeting of the American Association for Cancer Research
Manage Cardiovascular Risk in Vulnerable RA Patients
The jury is still out on just how cardiovascular risk should be screened and managed in rheumatoid arthritis patients, but it is clear that the risk is increased and must be addressed.
Patients with RA are known to have a lower probability of survival than do controls, and a major cause of excess death is from cardiovascular disease. In one study, silent MI occurred more often in RA patients, and sudden death was more likely in these patients (Arthritis Rheum. 2005;52:402-11). In another study, survival among patients with acute cardiac syndrome was substantially reduced in RA vs. non-RA patients (Ann. Rheum. Dis. 2006;65:348-53).
Some experts say RA is now equivalent to diabetes in terms of the extent to which it confers cardiovascular risk, according to Dr. Joan Bathon, director of the division of rheumatology at Columbia University, New York.
The European League Against Rheumatism has proposed multiplying conventional cardiovascular risk models by 1.5 when risk is assessed in RA patients, said Dr. Bathon (Ann. Rheum. Dis. 2010;69:325-31).
This approach is not well validated, and may not be widely used, she said. But the proposal illustrates the importance of focusing on cardiovascular risk in RA patients. It suggests that considering RA as a risk factor equivalent to diabetes – at least for making decisions about LDL cholesterol goals – is a reasonable strategy, she said.
Consider yearly cardiovascular risk screening, she said. The benefits of imaging and biomarkers are unclear, and no guidelines are in place. As a management strategy, aspirin therapy might be useful, but should be considered in the context of the patient's other medications. Statins are a potential management tool, but questions remain about whether all RA patients should be treated regardless of LDL cholesterol level, Dr. Bathon said.
Definite treatment strategies for RA patients include weight management for overweight patients, to help reduce inflammation, as well as exercise for all RA patients, because good quality muscle building will help restore insulin sensitivity and reduce fat depots that are the most inflammatory. Tight blood pressure control and tight RA control are imperative, Dr. Bathon said.
She noted that conventional risk factors do not fully explain the excess risk in RA patients, and that inflammation probably plays a role.
Dr. Bathon said she had no disclosures.
The jury is still out on just how cardiovascular risk should be screened and managed in rheumatoid arthritis patients, but it is clear that the risk is increased and must be addressed.
Patients with RA are known to have a lower probability of survival than do controls, and a major cause of excess death is from cardiovascular disease. In one study, silent MI occurred more often in RA patients, and sudden death was more likely in these patients (Arthritis Rheum. 2005;52:402-11). In another study, survival among patients with acute cardiac syndrome was substantially reduced in RA vs. non-RA patients (Ann. Rheum. Dis. 2006;65:348-53).
Some experts say RA is now equivalent to diabetes in terms of the extent to which it confers cardiovascular risk, according to Dr. Joan Bathon, director of the division of rheumatology at Columbia University, New York.
The European League Against Rheumatism has proposed multiplying conventional cardiovascular risk models by 1.5 when risk is assessed in RA patients, said Dr. Bathon (Ann. Rheum. Dis. 2010;69:325-31).
This approach is not well validated, and may not be widely used, she said. But the proposal illustrates the importance of focusing on cardiovascular risk in RA patients. It suggests that considering RA as a risk factor equivalent to diabetes – at least for making decisions about LDL cholesterol goals – is a reasonable strategy, she said.
Consider yearly cardiovascular risk screening, she said. The benefits of imaging and biomarkers are unclear, and no guidelines are in place. As a management strategy, aspirin therapy might be useful, but should be considered in the context of the patient's other medications. Statins are a potential management tool, but questions remain about whether all RA patients should be treated regardless of LDL cholesterol level, Dr. Bathon said.
Definite treatment strategies for RA patients include weight management for overweight patients, to help reduce inflammation, as well as exercise for all RA patients, because good quality muscle building will help restore insulin sensitivity and reduce fat depots that are the most inflammatory. Tight blood pressure control and tight RA control are imperative, Dr. Bathon said.
She noted that conventional risk factors do not fully explain the excess risk in RA patients, and that inflammation probably plays a role.
Dr. Bathon said she had no disclosures.
The jury is still out on just how cardiovascular risk should be screened and managed in rheumatoid arthritis patients, but it is clear that the risk is increased and must be addressed.
Patients with RA are known to have a lower probability of survival than do controls, and a major cause of excess death is from cardiovascular disease. In one study, silent MI occurred more often in RA patients, and sudden death was more likely in these patients (Arthritis Rheum. 2005;52:402-11). In another study, survival among patients with acute cardiac syndrome was substantially reduced in RA vs. non-RA patients (Ann. Rheum. Dis. 2006;65:348-53).
Some experts say RA is now equivalent to diabetes in terms of the extent to which it confers cardiovascular risk, according to Dr. Joan Bathon, director of the division of rheumatology at Columbia University, New York.
The European League Against Rheumatism has proposed multiplying conventional cardiovascular risk models by 1.5 when risk is assessed in RA patients, said Dr. Bathon (Ann. Rheum. Dis. 2010;69:325-31).
This approach is not well validated, and may not be widely used, she said. But the proposal illustrates the importance of focusing on cardiovascular risk in RA patients. It suggests that considering RA as a risk factor equivalent to diabetes – at least for making decisions about LDL cholesterol goals – is a reasonable strategy, she said.
Consider yearly cardiovascular risk screening, she said. The benefits of imaging and biomarkers are unclear, and no guidelines are in place. As a management strategy, aspirin therapy might be useful, but should be considered in the context of the patient's other medications. Statins are a potential management tool, but questions remain about whether all RA patients should be treated regardless of LDL cholesterol level, Dr. Bathon said.
Definite treatment strategies for RA patients include weight management for overweight patients, to help reduce inflammation, as well as exercise for all RA patients, because good quality muscle building will help restore insulin sensitivity and reduce fat depots that are the most inflammatory. Tight blood pressure control and tight RA control are imperative, Dr. Bathon said.
She noted that conventional risk factors do not fully explain the excess risk in RA patients, and that inflammation probably plays a role.
Dr. Bathon said she had no disclosures.
Biomarkers Offer Clues For Earlier Alzheimer's Diagnosis
PARIS – Two studies highlighted at the Alzheimer’s Association International Conference 2011 underscored the increasing focus on improving and promoting early diagnosis of Alzheimer’s disease using biomarkers.
One study focused on a potential blood-based predictor of neocortical amyloid burden – a known predictor of progression to Alzheimer’s disease – and suggests there is hope for the development of a low-cost and noninvasive screening tool.
The other study delved deeper into the value of cerebrospinal fluid biomarkers, which are already known to be associated with mild cognitive impairment leading to Alzheimer’s disease. In it, researchers analyzed just how early those markers – including amyloid beta 42, total tau, and phosphorylated tau (p-tau) – become useful as predictors of disease onset, which have particular implications for patient selection in ongoing and future clinical trials.
In the blood biomarker study, a panel of nine known blood-based markers of Alzheimer’s disease was found to have high sensitivity and specificity for identifying key levels of amyloid in the brain, suggesting that an effective and economical screening test for early identification of individuals at risk for the disease is within reach, Samantha Burnham, Ph.D., reported during a press briefing at the conference.
Currently, neocortical amyloid burden is assessable only by expensive positron emission tomography, which requires radiotracers such as Pittsburgh Compound B (PiB) that are not widely available, and by invasive cerebrospinal fluid (CSF) evaluation for markers that have been shown to correlate with neocortical amyloid burden, said Dr. Burnham of the Commonwealth Scientific and Industrial Research Organization (CSIRO), Perth, Australia.
She and her colleagues used 273 samples from patients with baseline blood and PiB-PET measurements from the Australian Imaging, Biomarkers, and Lifestyle (AIBL) study to build a model based on the nine blood-based markers (including amyloid beta 42, apolipoprotein E gene status, and cortisol levels) to estimate the amount of deposited amyloid in the brain. The panel of markers had 83% sensitivity and 85% specificity for identifying the level of amyloid deemed to indicate the risk of Alzheimer’s, she said.
In 817 nonimaged AIBL study participants, the researchers used the panel to predict if the percentage of patients with a high neocortical amyloid burden matched prior findings in the literature in regard to clinical diagnosis. Overall, 34% of healthy controls, 87% of individuals with mild cognitive impairment, and 100% of Alzheimer’s disease patients had a high burden, Dr. Burnham said.
Additionally, the investigators sought to validate the panel of markers in 74 subjects from the Alzheimer’s Disease Neuroimaging Initiative. In this validation study, measurements for two of the markers from the panel were unavailable, and the sensitivity and specificity were slightly lower, but still impressive at 76% for both. Furthermore, when the samples from the AIBL study were reassessed without the two missing markers, the sensitivity and specificity dropped to 74%, a level comparable to that seen in the validation study, suggesting that the accuracy in the validation study might have been even greater if all nine markers were available, she noted.
Because neocortical amyloid burden predicts progression to Alzheimer’s disease, and because the model used in this study accurately predicts neocortical amyloid burden, it appears likely that the findings will lead to the development of a useful, affordable screening test for the early identification of individuals at risk for developing Alzheimer’s disease, she concluded.
"This is a good start toward having a blood-based diagnosis," she said.
The development of such a diagnostic test is a long sought after goal among Alzheimer’s researchers and clinicians, which is why Dr. Burnham’s data were among the findings highlighted at the conference, according to Dr. Reisa Sperling of the department of neurology at Harvard Medical School, Boston, and a member of the panel of investigators at the press briefing.
"We all want a simpler test – we all want a blood test, an eye test, a skin test – something where it would be less expensive and less invasive," she said, commenting on the value of the findings.
The role of amyloid load is established, and using PiB-PET imaging as a marker of amyloid as the new gold standard with which to evaluate the usefulness of biomarkers, as was done in this study, is "really a wonderful way to use these biomarkers to look for simpler tests such as a blood test," she said.
In the study of cerebrospinal fluid biomarkers, Dr. Henrik Zetterberg presented data indicating that baseline levels of amyloid beta 42 and tau (both total and phosphorylated) are highly predictive of Alzheimer’s disease at up to 10 years prior to the onset of dementia.
"These are by far the best-studied [biomarkers], and show the greatest differences with regard to Alzheimer’s disease, and also they have the most direct link to the neuropathology of Alzheimer’s disease," said Dr. Zetterberg, professor and group leader of the research team on neurochemical pathogenesis and diagnostics at the University of Gothenburg (Sweden).
Although more biomarkers will most likely be identified in the future, these CSF proteins have shown the most consistent results across studies thus far, and they have high sensitivity and specificity for Alzheimer’s disease.
"The basic question we were trying to address is actually, ‘When do these biomarkers turn positive? When can we detect Alzheimer’s disease by measuring these proteins?’ " he said.
In his study of 137 patients with mild cognitive impairment, the levels of amyloid beta 42 were already fully decreased to levels that indicate a high risk of developing Alzheimer’s dementia in 5-10 years.
During nearly 10 years of follow-up, 54% of subjects developed Alzheimer’s disease and 16% developed other forms of dementia. Baseline amyloid beta 42 levels in those who developed Alzheimer’s disease were significantly reduced, compared with nonconverters, and total and p-tau levels were elevated, Dr. Zetterberg said.
Furthermore, the amyloid beta 42 levels were equally reduced at baseline in early and later converters (those who converted within 0-5 years, and those who converted between 5 and 10 years, respectively), while CSF levels of total and p-tau were significantly higher in the early converters, compared with the later converters.
A ratio of baseline levels of amyloid beta 42 to p-tau predicted development of Alzheimer’s disease within the study period with a sensitivity of 88% and a specificity of 90%, he said.
"So the conclusion might be that Alzheimer’s disease can actually be accurately diagnosed 5-10 years before onset of dementia," he said.
The findings support the hypothesis that altered amyloid beta metabolism precedes tau-related pathology and neuronal degeneration. These CSF biomarkers may be useful for selecting patients for early intervention in clinical trials, as well as for monitoring treatment effects, Dr. Zetterberg concluded.
Dr. Burnham and Dr. Zetterberg said they had no relevant financial disclosures.
PARIS – Two studies highlighted at the Alzheimer’s Association International Conference 2011 underscored the increasing focus on improving and promoting early diagnosis of Alzheimer’s disease using biomarkers.
One study focused on a potential blood-based predictor of neocortical amyloid burden – a known predictor of progression to Alzheimer’s disease – and suggests there is hope for the development of a low-cost and noninvasive screening tool.
The other study delved deeper into the value of cerebrospinal fluid biomarkers, which are already known to be associated with mild cognitive impairment leading to Alzheimer’s disease. In it, researchers analyzed just how early those markers – including amyloid beta 42, total tau, and phosphorylated tau (p-tau) – become useful as predictors of disease onset, which have particular implications for patient selection in ongoing and future clinical trials.
In the blood biomarker study, a panel of nine known blood-based markers of Alzheimer’s disease was found to have high sensitivity and specificity for identifying key levels of amyloid in the brain, suggesting that an effective and economical screening test for early identification of individuals at risk for the disease is within reach, Samantha Burnham, Ph.D., reported during a press briefing at the conference.
Currently, neocortical amyloid burden is assessable only by expensive positron emission tomography, which requires radiotracers such as Pittsburgh Compound B (PiB) that are not widely available, and by invasive cerebrospinal fluid (CSF) evaluation for markers that have been shown to correlate with neocortical amyloid burden, said Dr. Burnham of the Commonwealth Scientific and Industrial Research Organization (CSIRO), Perth, Australia.
She and her colleagues used 273 samples from patients with baseline blood and PiB-PET measurements from the Australian Imaging, Biomarkers, and Lifestyle (AIBL) study to build a model based on the nine blood-based markers (including amyloid beta 42, apolipoprotein E gene status, and cortisol levels) to estimate the amount of deposited amyloid in the brain. The panel of markers had 83% sensitivity and 85% specificity for identifying the level of amyloid deemed to indicate the risk of Alzheimer’s, she said.
In 817 nonimaged AIBL study participants, the researchers used the panel to predict if the percentage of patients with a high neocortical amyloid burden matched prior findings in the literature in regard to clinical diagnosis. Overall, 34% of healthy controls, 87% of individuals with mild cognitive impairment, and 100% of Alzheimer’s disease patients had a high burden, Dr. Burnham said.
Additionally, the investigators sought to validate the panel of markers in 74 subjects from the Alzheimer’s Disease Neuroimaging Initiative. In this validation study, measurements for two of the markers from the panel were unavailable, and the sensitivity and specificity were slightly lower, but still impressive at 76% for both. Furthermore, when the samples from the AIBL study were reassessed without the two missing markers, the sensitivity and specificity dropped to 74%, a level comparable to that seen in the validation study, suggesting that the accuracy in the validation study might have been even greater if all nine markers were available, she noted.
Because neocortical amyloid burden predicts progression to Alzheimer’s disease, and because the model used in this study accurately predicts neocortical amyloid burden, it appears likely that the findings will lead to the development of a useful, affordable screening test for the early identification of individuals at risk for developing Alzheimer’s disease, she concluded.
"This is a good start toward having a blood-based diagnosis," she said.
The development of such a diagnostic test is a long sought after goal among Alzheimer’s researchers and clinicians, which is why Dr. Burnham’s data were among the findings highlighted at the conference, according to Dr. Reisa Sperling of the department of neurology at Harvard Medical School, Boston, and a member of the panel of investigators at the press briefing.
"We all want a simpler test – we all want a blood test, an eye test, a skin test – something where it would be less expensive and less invasive," she said, commenting on the value of the findings.
The role of amyloid load is established, and using PiB-PET imaging as a marker of amyloid as the new gold standard with which to evaluate the usefulness of biomarkers, as was done in this study, is "really a wonderful way to use these biomarkers to look for simpler tests such as a blood test," she said.
In the study of cerebrospinal fluid biomarkers, Dr. Henrik Zetterberg presented data indicating that baseline levels of amyloid beta 42 and tau (both total and phosphorylated) are highly predictive of Alzheimer’s disease at up to 10 years prior to the onset of dementia.
"These are by far the best-studied [biomarkers], and show the greatest differences with regard to Alzheimer’s disease, and also they have the most direct link to the neuropathology of Alzheimer’s disease," said Dr. Zetterberg, professor and group leader of the research team on neurochemical pathogenesis and diagnostics at the University of Gothenburg (Sweden).
Although more biomarkers will most likely be identified in the future, these CSF proteins have shown the most consistent results across studies thus far, and they have high sensitivity and specificity for Alzheimer’s disease.
"The basic question we were trying to address is actually, ‘When do these biomarkers turn positive? When can we detect Alzheimer’s disease by measuring these proteins?’ " he said.
In his study of 137 patients with mild cognitive impairment, the levels of amyloid beta 42 were already fully decreased to levels that indicate a high risk of developing Alzheimer’s dementia in 5-10 years.
During nearly 10 years of follow-up, 54% of subjects developed Alzheimer’s disease and 16% developed other forms of dementia. Baseline amyloid beta 42 levels in those who developed Alzheimer’s disease were significantly reduced, compared with nonconverters, and total and p-tau levels were elevated, Dr. Zetterberg said.
Furthermore, the amyloid beta 42 levels were equally reduced at baseline in early and later converters (those who converted within 0-5 years, and those who converted between 5 and 10 years, respectively), while CSF levels of total and p-tau were significantly higher in the early converters, compared with the later converters.
A ratio of baseline levels of amyloid beta 42 to p-tau predicted development of Alzheimer’s disease within the study period with a sensitivity of 88% and a specificity of 90%, he said.
"So the conclusion might be that Alzheimer’s disease can actually be accurately diagnosed 5-10 years before onset of dementia," he said.
The findings support the hypothesis that altered amyloid beta metabolism precedes tau-related pathology and neuronal degeneration. These CSF biomarkers may be useful for selecting patients for early intervention in clinical trials, as well as for monitoring treatment effects, Dr. Zetterberg concluded.
Dr. Burnham and Dr. Zetterberg said they had no relevant financial disclosures.
PARIS – Two studies highlighted at the Alzheimer’s Association International Conference 2011 underscored the increasing focus on improving and promoting early diagnosis of Alzheimer’s disease using biomarkers.
One study focused on a potential blood-based predictor of neocortical amyloid burden – a known predictor of progression to Alzheimer’s disease – and suggests there is hope for the development of a low-cost and noninvasive screening tool.
The other study delved deeper into the value of cerebrospinal fluid biomarkers, which are already known to be associated with mild cognitive impairment leading to Alzheimer’s disease. In it, researchers analyzed just how early those markers – including amyloid beta 42, total tau, and phosphorylated tau (p-tau) – become useful as predictors of disease onset, which have particular implications for patient selection in ongoing and future clinical trials.
In the blood biomarker study, a panel of nine known blood-based markers of Alzheimer’s disease was found to have high sensitivity and specificity for identifying key levels of amyloid in the brain, suggesting that an effective and economical screening test for early identification of individuals at risk for the disease is within reach, Samantha Burnham, Ph.D., reported during a press briefing at the conference.
Currently, neocortical amyloid burden is assessable only by expensive positron emission tomography, which requires radiotracers such as Pittsburgh Compound B (PiB) that are not widely available, and by invasive cerebrospinal fluid (CSF) evaluation for markers that have been shown to correlate with neocortical amyloid burden, said Dr. Burnham of the Commonwealth Scientific and Industrial Research Organization (CSIRO), Perth, Australia.
She and her colleagues used 273 samples from patients with baseline blood and PiB-PET measurements from the Australian Imaging, Biomarkers, and Lifestyle (AIBL) study to build a model based on the nine blood-based markers (including amyloid beta 42, apolipoprotein E gene status, and cortisol levels) to estimate the amount of deposited amyloid in the brain. The panel of markers had 83% sensitivity and 85% specificity for identifying the level of amyloid deemed to indicate the risk of Alzheimer’s, she said.
In 817 nonimaged AIBL study participants, the researchers used the panel to predict if the percentage of patients with a high neocortical amyloid burden matched prior findings in the literature in regard to clinical diagnosis. Overall, 34% of healthy controls, 87% of individuals with mild cognitive impairment, and 100% of Alzheimer’s disease patients had a high burden, Dr. Burnham said.
Additionally, the investigators sought to validate the panel of markers in 74 subjects from the Alzheimer’s Disease Neuroimaging Initiative. In this validation study, measurements for two of the markers from the panel were unavailable, and the sensitivity and specificity were slightly lower, but still impressive at 76% for both. Furthermore, when the samples from the AIBL study were reassessed without the two missing markers, the sensitivity and specificity dropped to 74%, a level comparable to that seen in the validation study, suggesting that the accuracy in the validation study might have been even greater if all nine markers were available, she noted.
Because neocortical amyloid burden predicts progression to Alzheimer’s disease, and because the model used in this study accurately predicts neocortical amyloid burden, it appears likely that the findings will lead to the development of a useful, affordable screening test for the early identification of individuals at risk for developing Alzheimer’s disease, she concluded.
"This is a good start toward having a blood-based diagnosis," she said.
The development of such a diagnostic test is a long sought after goal among Alzheimer’s researchers and clinicians, which is why Dr. Burnham’s data were among the findings highlighted at the conference, according to Dr. Reisa Sperling of the department of neurology at Harvard Medical School, Boston, and a member of the panel of investigators at the press briefing.
"We all want a simpler test – we all want a blood test, an eye test, a skin test – something where it would be less expensive and less invasive," she said, commenting on the value of the findings.
The role of amyloid load is established, and using PiB-PET imaging as a marker of amyloid as the new gold standard with which to evaluate the usefulness of biomarkers, as was done in this study, is "really a wonderful way to use these biomarkers to look for simpler tests such as a blood test," she said.
In the study of cerebrospinal fluid biomarkers, Dr. Henrik Zetterberg presented data indicating that baseline levels of amyloid beta 42 and tau (both total and phosphorylated) are highly predictive of Alzheimer’s disease at up to 10 years prior to the onset of dementia.
"These are by far the best-studied [biomarkers], and show the greatest differences with regard to Alzheimer’s disease, and also they have the most direct link to the neuropathology of Alzheimer’s disease," said Dr. Zetterberg, professor and group leader of the research team on neurochemical pathogenesis and diagnostics at the University of Gothenburg (Sweden).
Although more biomarkers will most likely be identified in the future, these CSF proteins have shown the most consistent results across studies thus far, and they have high sensitivity and specificity for Alzheimer’s disease.
"The basic question we were trying to address is actually, ‘When do these biomarkers turn positive? When can we detect Alzheimer’s disease by measuring these proteins?’ " he said.
In his study of 137 patients with mild cognitive impairment, the levels of amyloid beta 42 were already fully decreased to levels that indicate a high risk of developing Alzheimer’s dementia in 5-10 years.
During nearly 10 years of follow-up, 54% of subjects developed Alzheimer’s disease and 16% developed other forms of dementia. Baseline amyloid beta 42 levels in those who developed Alzheimer’s disease were significantly reduced, compared with nonconverters, and total and p-tau levels were elevated, Dr. Zetterberg said.
Furthermore, the amyloid beta 42 levels were equally reduced at baseline in early and later converters (those who converted within 0-5 years, and those who converted between 5 and 10 years, respectively), while CSF levels of total and p-tau were significantly higher in the early converters, compared with the later converters.
A ratio of baseline levels of amyloid beta 42 to p-tau predicted development of Alzheimer’s disease within the study period with a sensitivity of 88% and a specificity of 90%, he said.
"So the conclusion might be that Alzheimer’s disease can actually be accurately diagnosed 5-10 years before onset of dementia," he said.
The findings support the hypothesis that altered amyloid beta metabolism precedes tau-related pathology and neuronal degeneration. These CSF biomarkers may be useful for selecting patients for early intervention in clinical trials, as well as for monitoring treatment effects, Dr. Zetterberg concluded.
Dr. Burnham and Dr. Zetterberg said they had no relevant financial disclosures.
FROM THE ALZHEIMER'S ASSOCIATION INTERNATIONAL CONFERENCE 2011
AAP Updates UTI Guidelines for Febrile Infants
Antimicrobial prophylaxis to prevent febrile recurrent urinary tract infection in infants aged 2 months to 2 years without vesicoureteral reflux or with grade I to IV VUR is not supported by currently available data, and therefore voiding cystourethrography to detect VUR should no longer be used routinely after a first UTI in this population, according to a newly updated American Academy of Pediatrics practice guideline.
The update – the first since 1999 to the guideline regarding the diagnosis and management of the initial UTI in febrile infants and young children – is published in the Aug. 29 issue of Pediatrics (2011;128:595-610 [doi:10.1542/peds.2011-1330]).
The previous guideline included strong encouragement of imaging studies despite insufficient evidence supporting a routine voiding cystourethrogram (VCUG), but the "position of the current subcommittee reflects the new evidence demonstrating antimicrobial prophylaxis not to be effective as presumed previously. Moreover, prompt diagnosis and effective treatment of a febrile UTI recurrence may be of greater importance regardless of whether VUR is present or the child is receiving antimicrobial prophylaxis," according to the AAP Subcommittee on Urinary Tract Infection, which authored the updated guideline.
The update, which is intended for use in various clinical settings including offices, hospitals, and emergency departments for the diagnosis and management of the approximately 5% of children in this age group who are affected by UTIs, is based in large part on new data regarding imaging and diagnostic criteria in this population.
It also includes changes as to which children should undergo urine testing, how the sample should be obtained, and how treatment and follow-up should be conducted in those found to have a UTI, according to lead author Dr. Kenneth B. Roberts of Greensboro, N.C., and his subcommittee colleagues.
The "long-awaited update" provides an "exceptionally evidence-based guideline that differs in important ways from the 1999 guidelines and sets a high standard for transparency and scholarship," Dr. Thomas B. Newman of the University of California, San Francisco, wrote in an accompanying editorial (Pediatrics 2011;128:572-75 [doi:10.1542/peds.2011-1818]).
Dr. Newman also said that the guideline and a technical report containing the data on which the recommendations are based represent "a significant advance."
The guideline includes seven recommendations or action statements:
• Action Statement 1. It calls for obtaining a urine specimen for both culture and urinalysis prior to administration of an antimicrobial agent in infants with no apparent source for their fever, and in whom the clinician decides antimicrobial therapy should be administered due to ill appearance or another pressing reason. Since they assert that the diagnosis of UTI cannot be reliably established through culture of urine collected in a bag, the subcommittee says the specimen should be obtained through catheterization or suprapubic aspirate (SPA). The evidence quality for this "strong recommendation" was level A, meaning it is based on well-designed randomized controlled trials or diagnostic studies of relevant populations.
Although catheterization is invasive, the subcommittee determined that the risks of this approach are outweighed by the potential benefit of diagnosing a UTI and treating it appropriately before renal scarring occurs, and, conversely, by avoiding overtreatment and unnecessary and expensive imaging.
• Action Statements 2a and 2b. These also represent "strong recommendations" based on level A evidence, but address those febrile infants who the clinician does not feel require immediate antimicrobial therapy. In these infants, the clinician should assess whether the likelihood of UTI is low or high. Those with a low likelihood of UTI (such as those with another obvious infection, which reduces the likelihood of UTI) can undergo clinical follow-up monitoring without testing; those not in a low-risk group (such as girls and uncircumcised boys, who have a higher risk for UTI) can be managed either by obtaining a urine specimen via catheterization or SPA for culture and urinalysis, or by the most convenient means for urinalysis. If the urinalysis results suggest a UTI, a specimen should then be obtained by catheterization or SPA for culture.
A negative urinalysis, however, suggests it is reasonable to monitor the clinical course without initiating antimicrobial therapy.
• Action Statement 3. This also addresses UTI diagnosis, and states that clinicians should require both urinalysis results that suggest infection (pyuria and/or bacteriuria) and the presence of at least 50,000 colony-forming units per milliliter of a uropathogen cultured from a specimen obtained via catheterization or SPA. Unlike the first two action statements, this one is based on observational studies, and thus is considered level C evidence and represents a "recommendation" rather than a "strong recommendation."
"These criteria reduce the likelihood of overdiagnosis of UTI in infants with asymptomatic bacteriuria or contaminated specimens," the subcommittee wrote.
• Action Statement 4a. This recommendation states that a clinician initiating treatment should base the choice of route of administration on practical considerations; oral and parenteral treatment are equally efficacious, so the choice depends on local antimicrobial sensitivity patterns and sensitivity testing of the isolated uropathogen. This "strong recommendation" is based on level A evidence.
• Action Statement 4b. This "recommendation," based on level B evidence, states that treatment duration should be 7-14 days.
Most children can be treated orally, and although no data comparing 7-, 10-, and 14-day regimens were available, evidence does exist showing that courses of 1-3 days are inferior. Therefore, the minimal duration of treatment should be 7 days, the subcommittee said.
• Action Statement 5. This recommendation addresses the need for renal and bladder ultrasonography (RBUS) in all febrile infants with UTIs. Level C evidence resulted in this "recommendation."
"The purpose of RBUS is to detect anatomic abnormalities that require further evaluation, such as additional imaging or urologic consultation. RBUS also provides an evaluation of the renal parenchyma and an assessment of renal size that can be used to monitor renal growth" they wrote, adding that RBUS is recommended during the first 2 days of treatment unless substantial clinical improvement is demonstrated before that. In those cases, imaging should not occur early during acute infection, as the findings may be misleading.
• Action Statement 6a. This recommendation notes that VCUG should not be used routinely after a first febrile UTI, but should be reserved for use if RBUS reveals hydronephrosis, scarring, or other findings suggestive of high-grade VUR or obstructive uropathy. It may also be used in "other atypical or complex clinical circumstances." Level B evidence supports this "recommendation."
• Action Statement 6b states that further evaluation should be conducted if there is a recurrence of febrile UTI. This statement is supported by level X evidence, indicating that the "recommendation" is based on "exceptional situations where validating studies cannot be performed and there is a clear preponderance of benefit or harm."
Specifically, this statement reflects the fact that studies in recent years have brought into question the value of prophylaxis for preventing recurrent UTI, as well as the rationale for performing VCUG routinely after an initial febrile UTI.
The previous guideline included strong encouragement of imaging studies despite insufficient evidence supporting routine VCUG, but the "position of the current subcommittee reflects the new evidence demonstrating antimicrobial prophylaxis not to be effective as presumed previously. Moreover, prompt diagnosis and effective treatment of a febrile UTI recurrence may be of greater importance regardless of whether VUR is present or the child is receiving antimicrobial prophylaxis," they wrote.
• Action Statement 7. This recommendation states that after confirmation of a UTI, the clinician should instruct parents or guardians to seek prompt medical evaluations – ideally within 48 hours – if febrile illness recurs.
This is to ensure that recent infections are detected and treated promptly, they noted. Level C evidence supports this "recommendation."
"Early treatment limits renal damage better than late treatment, and the risk of renal scarring increases as the number of recurrences increases. For these reasons, all infants who have sustained a febrile UTI should have a urine specimen obtained at the onset of subsequent febrile illnesses so that UTI can be diagnosed and treated promptly," they wrote.
This updated guideline focuses on diagnosis and management of an initial UTI in children aged 2-24 months with no obvious neurologic or anatomic abnormalities known to be associated with recurrent UTI or renal damage. Fever for the purpose of the guideline was defined as a temperature of at least 100.4° F.
In developing the update, the subcommittee performed literature searches, including surveillance of Medline-listed literature over the past 10 years, and – in light of the increasing evidence questioning the value of antimicrobial prophylaxis to prevent recurrent febrile UTI in children with VUR – they also performed a systematic review of literature on the effectiveness of prophylactic antimicrobial therapy to prevent recurrent febrile UTI in this population. Using data from six randomized controlled trials for which raw data on infants aged 2-24 months were available, they also performed a meta-analysis addressing the effectiveness of prophylactic antimicrobial therapy. The findings are published in the companion technical report (Pediatrics 2011;128:e749–e770 [doi:10.1542/peds.2011-1332).
The authors of the guideline stressed that their recommendations do not represent standard of care or indicate an exclusive course of treatment, but rather are intended to assist clinicians in decision-making.
In his accompanying editorial, Dr. Newman praised many aspects of the updated guideline, but also highlighted some instances where he believes "alternative recommendations are supported by available evidence."
For example, while he agrees that the recommendation on selective urine testing based on the prior probability of UTI is "an important improvement," and he lauded the subcommittee’s job summarizing the factors that determine that probability, he questioned the assertion that the threshold probability for urine testing is below 3%, and proposed that until this is proven, the decision to test should be individualized.
Additionally, while he said he is "glad the new guideline continues to offer the option of obtaining urine for urinalyses noninvasively," he also said he is not convinced that the bag urine can never be used for culture. "If the urinalysis is used to select urine for culture, the prior probability may sometimes be in a range where the bag culture will be useful," he explained.
Despite these and other concerns addressed by Dr. Newman, he saluted the authors and noted that the updated guideline should prove helpful to clinicians and families dealing with this common problem.
Members of the subcommittee that developed this guideline included individuals with expertise in the fields of academic general pediatrics, epidemiology and informatics, pediatric infectious disease, pediatric nephrology, pediatric practice, pediatric radiology, and pediatric urology. The AAP funded the development of the guidelines; none of the subcommittee members had financial conflicts of interest. Dr. Newman also had no financial conflicts to report.
Antimicrobial prophylaxis to prevent febrile recurrent urinary tract infection in infants aged 2 months to 2 years without vesicoureteral reflux or with grade I to IV VUR is not supported by currently available data, and therefore voiding cystourethrography to detect VUR should no longer be used routinely after a first UTI in this population, according to a newly updated American Academy of Pediatrics practice guideline.
The update – the first since 1999 to the guideline regarding the diagnosis and management of the initial UTI in febrile infants and young children – is published in the Aug. 29 issue of Pediatrics (2011;128:595-610 [doi:10.1542/peds.2011-1330]).
The previous guideline included strong encouragement of imaging studies despite insufficient evidence supporting a routine voiding cystourethrogram (VCUG), but the "position of the current subcommittee reflects the new evidence demonstrating antimicrobial prophylaxis not to be effective as presumed previously. Moreover, prompt diagnosis and effective treatment of a febrile UTI recurrence may be of greater importance regardless of whether VUR is present or the child is receiving antimicrobial prophylaxis," according to the AAP Subcommittee on Urinary Tract Infection, which authored the updated guideline.
The update, which is intended for use in various clinical settings including offices, hospitals, and emergency departments for the diagnosis and management of the approximately 5% of children in this age group who are affected by UTIs, is based in large part on new data regarding imaging and diagnostic criteria in this population.
It also includes changes as to which children should undergo urine testing, how the sample should be obtained, and how treatment and follow-up should be conducted in those found to have a UTI, according to lead author Dr. Kenneth B. Roberts of Greensboro, N.C., and his subcommittee colleagues.
The "long-awaited update" provides an "exceptionally evidence-based guideline that differs in important ways from the 1999 guidelines and sets a high standard for transparency and scholarship," Dr. Thomas B. Newman of the University of California, San Francisco, wrote in an accompanying editorial (Pediatrics 2011;128:572-75 [doi:10.1542/peds.2011-1818]).
Dr. Newman also said that the guideline and a technical report containing the data on which the recommendations are based represent "a significant advance."
The guideline includes seven recommendations or action statements:
• Action Statement 1. It calls for obtaining a urine specimen for both culture and urinalysis prior to administration of an antimicrobial agent in infants with no apparent source for their fever, and in whom the clinician decides antimicrobial therapy should be administered due to ill appearance or another pressing reason. Since they assert that the diagnosis of UTI cannot be reliably established through culture of urine collected in a bag, the subcommittee says the specimen should be obtained through catheterization or suprapubic aspirate (SPA). The evidence quality for this "strong recommendation" was level A, meaning it is based on well-designed randomized controlled trials or diagnostic studies of relevant populations.
Although catheterization is invasive, the subcommittee determined that the risks of this approach are outweighed by the potential benefit of diagnosing a UTI and treating it appropriately before renal scarring occurs, and, conversely, by avoiding overtreatment and unnecessary and expensive imaging.
• Action Statements 2a and 2b. These also represent "strong recommendations" based on level A evidence, but address those febrile infants who the clinician does not feel require immediate antimicrobial therapy. In these infants, the clinician should assess whether the likelihood of UTI is low or high. Those with a low likelihood of UTI (such as those with another obvious infection, which reduces the likelihood of UTI) can undergo clinical follow-up monitoring without testing; those not in a low-risk group (such as girls and uncircumcised boys, who have a higher risk for UTI) can be managed either by obtaining a urine specimen via catheterization or SPA for culture and urinalysis, or by the most convenient means for urinalysis. If the urinalysis results suggest a UTI, a specimen should then be obtained by catheterization or SPA for culture.
A negative urinalysis, however, suggests it is reasonable to monitor the clinical course without initiating antimicrobial therapy.
• Action Statement 3. This also addresses UTI diagnosis, and states that clinicians should require both urinalysis results that suggest infection (pyuria and/or bacteriuria) and the presence of at least 50,000 colony-forming units per milliliter of a uropathogen cultured from a specimen obtained via catheterization or SPA. Unlike the first two action statements, this one is based on observational studies, and thus is considered level C evidence and represents a "recommendation" rather than a "strong recommendation."
"These criteria reduce the likelihood of overdiagnosis of UTI in infants with asymptomatic bacteriuria or contaminated specimens," the subcommittee wrote.
• Action Statement 4a. This recommendation states that a clinician initiating treatment should base the choice of route of administration on practical considerations; oral and parenteral treatment are equally efficacious, so the choice depends on local antimicrobial sensitivity patterns and sensitivity testing of the isolated uropathogen. This "strong recommendation" is based on level A evidence.
• Action Statement 4b. This "recommendation," based on level B evidence, states that treatment duration should be 7-14 days.
Most children can be treated orally, and although no data comparing 7-, 10-, and 14-day regimens were available, evidence does exist showing that courses of 1-3 days are inferior. Therefore, the minimal duration of treatment should be 7 days, the subcommittee said.
• Action Statement 5. This recommendation addresses the need for renal and bladder ultrasonography (RBUS) in all febrile infants with UTIs. Level C evidence resulted in this "recommendation."
"The purpose of RBUS is to detect anatomic abnormalities that require further evaluation, such as additional imaging or urologic consultation. RBUS also provides an evaluation of the renal parenchyma and an assessment of renal size that can be used to monitor renal growth" they wrote, adding that RBUS is recommended during the first 2 days of treatment unless substantial clinical improvement is demonstrated before that. In those cases, imaging should not occur early during acute infection, as the findings may be misleading.
• Action Statement 6a. This recommendation notes that VCUG should not be used routinely after a first febrile UTI, but should be reserved for use if RBUS reveals hydronephrosis, scarring, or other findings suggestive of high-grade VUR or obstructive uropathy. It may also be used in "other atypical or complex clinical circumstances." Level B evidence supports this "recommendation."
• Action Statement 6b states that further evaluation should be conducted if there is a recurrence of febrile UTI. This statement is supported by level X evidence, indicating that the "recommendation" is based on "exceptional situations where validating studies cannot be performed and there is a clear preponderance of benefit or harm."
Specifically, this statement reflects the fact that studies in recent years have brought into question the value of prophylaxis for preventing recurrent UTI, as well as the rationale for performing VCUG routinely after an initial febrile UTI.
The previous guideline included strong encouragement of imaging studies despite insufficient evidence supporting routine VCUG, but the "position of the current subcommittee reflects the new evidence demonstrating antimicrobial prophylaxis not to be effective as presumed previously. Moreover, prompt diagnosis and effective treatment of a febrile UTI recurrence may be of greater importance regardless of whether VUR is present or the child is receiving antimicrobial prophylaxis," they wrote.
• Action Statement 7. This recommendation states that after confirmation of a UTI, the clinician should instruct parents or guardians to seek prompt medical evaluations – ideally within 48 hours – if febrile illness recurs.
This is to ensure that recent infections are detected and treated promptly, they noted. Level C evidence supports this "recommendation."
"Early treatment limits renal damage better than late treatment, and the risk of renal scarring increases as the number of recurrences increases. For these reasons, all infants who have sustained a febrile UTI should have a urine specimen obtained at the onset of subsequent febrile illnesses so that UTI can be diagnosed and treated promptly," they wrote.
This updated guideline focuses on diagnosis and management of an initial UTI in children aged 2-24 months with no obvious neurologic or anatomic abnormalities known to be associated with recurrent UTI or renal damage. Fever for the purpose of the guideline was defined as a temperature of at least 100.4° F.
In developing the update, the subcommittee performed literature searches, including surveillance of Medline-listed literature over the past 10 years, and – in light of the increasing evidence questioning the value of antimicrobial prophylaxis to prevent recurrent febrile UTI in children with VUR – they also performed a systematic review of literature on the effectiveness of prophylactic antimicrobial therapy to prevent recurrent febrile UTI in this population. Using data from six randomized controlled trials for which raw data on infants aged 2-24 months were available, they also performed a meta-analysis addressing the effectiveness of prophylactic antimicrobial therapy. The findings are published in the companion technical report (Pediatrics 2011;128:e749–e770 [doi:10.1542/peds.2011-1332).
The authors of the guideline stressed that their recommendations do not represent standard of care or indicate an exclusive course of treatment, but rather are intended to assist clinicians in decision-making.
In his accompanying editorial, Dr. Newman praised many aspects of the updated guideline, but also highlighted some instances where he believes "alternative recommendations are supported by available evidence."
For example, while he agrees that the recommendation on selective urine testing based on the prior probability of UTI is "an important improvement," and he lauded the subcommittee’s job summarizing the factors that determine that probability, he questioned the assertion that the threshold probability for urine testing is below 3%, and proposed that until this is proven, the decision to test should be individualized.
Additionally, while he said he is "glad the new guideline continues to offer the option of obtaining urine for urinalyses noninvasively," he also said he is not convinced that the bag urine can never be used for culture. "If the urinalysis is used to select urine for culture, the prior probability may sometimes be in a range where the bag culture will be useful," he explained.
Despite these and other concerns addressed by Dr. Newman, he saluted the authors and noted that the updated guideline should prove helpful to clinicians and families dealing with this common problem.
Members of the subcommittee that developed this guideline included individuals with expertise in the fields of academic general pediatrics, epidemiology and informatics, pediatric infectious disease, pediatric nephrology, pediatric practice, pediatric radiology, and pediatric urology. The AAP funded the development of the guidelines; none of the subcommittee members had financial conflicts of interest. Dr. Newman also had no financial conflicts to report.
Antimicrobial prophylaxis to prevent febrile recurrent urinary tract infection in infants aged 2 months to 2 years without vesicoureteral reflux or with grade I to IV VUR is not supported by currently available data, and therefore voiding cystourethrography to detect VUR should no longer be used routinely after a first UTI in this population, according to a newly updated American Academy of Pediatrics practice guideline.
The update – the first since 1999 to the guideline regarding the diagnosis and management of the initial UTI in febrile infants and young children – is published in the Aug. 29 issue of Pediatrics (2011;128:595-610 [doi:10.1542/peds.2011-1330]).
The previous guideline included strong encouragement of imaging studies despite insufficient evidence supporting a routine voiding cystourethrogram (VCUG), but the "position of the current subcommittee reflects the new evidence demonstrating antimicrobial prophylaxis not to be effective as presumed previously. Moreover, prompt diagnosis and effective treatment of a febrile UTI recurrence may be of greater importance regardless of whether VUR is present or the child is receiving antimicrobial prophylaxis," according to the AAP Subcommittee on Urinary Tract Infection, which authored the updated guideline.
The update, which is intended for use in various clinical settings including offices, hospitals, and emergency departments for the diagnosis and management of the approximately 5% of children in this age group who are affected by UTIs, is based in large part on new data regarding imaging and diagnostic criteria in this population.
It also includes changes as to which children should undergo urine testing, how the sample should be obtained, and how treatment and follow-up should be conducted in those found to have a UTI, according to lead author Dr. Kenneth B. Roberts of Greensboro, N.C., and his subcommittee colleagues.
The "long-awaited update" provides an "exceptionally evidence-based guideline that differs in important ways from the 1999 guidelines and sets a high standard for transparency and scholarship," Dr. Thomas B. Newman of the University of California, San Francisco, wrote in an accompanying editorial (Pediatrics 2011;128:572-75 [doi:10.1542/peds.2011-1818]).
Dr. Newman also said that the guideline and a technical report containing the data on which the recommendations are based represent "a significant advance."
The guideline includes seven recommendations or action statements:
• Action Statement 1. It calls for obtaining a urine specimen for both culture and urinalysis prior to administration of an antimicrobial agent in infants with no apparent source for their fever, and in whom the clinician decides antimicrobial therapy should be administered due to ill appearance or another pressing reason. Since they assert that the diagnosis of UTI cannot be reliably established through culture of urine collected in a bag, the subcommittee says the specimen should be obtained through catheterization or suprapubic aspirate (SPA). The evidence quality for this "strong recommendation" was level A, meaning it is based on well-designed randomized controlled trials or diagnostic studies of relevant populations.
Although catheterization is invasive, the subcommittee determined that the risks of this approach are outweighed by the potential benefit of diagnosing a UTI and treating it appropriately before renal scarring occurs, and, conversely, by avoiding overtreatment and unnecessary and expensive imaging.
• Action Statements 2a and 2b. These also represent "strong recommendations" based on level A evidence, but address those febrile infants who the clinician does not feel require immediate antimicrobial therapy. In these infants, the clinician should assess whether the likelihood of UTI is low or high. Those with a low likelihood of UTI (such as those with another obvious infection, which reduces the likelihood of UTI) can undergo clinical follow-up monitoring without testing; those not in a low-risk group (such as girls and uncircumcised boys, who have a higher risk for UTI) can be managed either by obtaining a urine specimen via catheterization or SPA for culture and urinalysis, or by the most convenient means for urinalysis. If the urinalysis results suggest a UTI, a specimen should then be obtained by catheterization or SPA for culture.
A negative urinalysis, however, suggests it is reasonable to monitor the clinical course without initiating antimicrobial therapy.
• Action Statement 3. This also addresses UTI diagnosis, and states that clinicians should require both urinalysis results that suggest infection (pyuria and/or bacteriuria) and the presence of at least 50,000 colony-forming units per milliliter of a uropathogen cultured from a specimen obtained via catheterization or SPA. Unlike the first two action statements, this one is based on observational studies, and thus is considered level C evidence and represents a "recommendation" rather than a "strong recommendation."
"These criteria reduce the likelihood of overdiagnosis of UTI in infants with asymptomatic bacteriuria or contaminated specimens," the subcommittee wrote.
• Action Statement 4a. This recommendation states that a clinician initiating treatment should base the choice of route of administration on practical considerations; oral and parenteral treatment are equally efficacious, so the choice depends on local antimicrobial sensitivity patterns and sensitivity testing of the isolated uropathogen. This "strong recommendation" is based on level A evidence.
• Action Statement 4b. This "recommendation," based on level B evidence, states that treatment duration should be 7-14 days.
Most children can be treated orally, and although no data comparing 7-, 10-, and 14-day regimens were available, evidence does exist showing that courses of 1-3 days are inferior. Therefore, the minimal duration of treatment should be 7 days, the subcommittee said.
• Action Statement 5. This recommendation addresses the need for renal and bladder ultrasonography (RBUS) in all febrile infants with UTIs. Level C evidence resulted in this "recommendation."
"The purpose of RBUS is to detect anatomic abnormalities that require further evaluation, such as additional imaging or urologic consultation. RBUS also provides an evaluation of the renal parenchyma and an assessment of renal size that can be used to monitor renal growth" they wrote, adding that RBUS is recommended during the first 2 days of treatment unless substantial clinical improvement is demonstrated before that. In those cases, imaging should not occur early during acute infection, as the findings may be misleading.
• Action Statement 6a. This recommendation notes that VCUG should not be used routinely after a first febrile UTI, but should be reserved for use if RBUS reveals hydronephrosis, scarring, or other findings suggestive of high-grade VUR or obstructive uropathy. It may also be used in "other atypical or complex clinical circumstances." Level B evidence supports this "recommendation."
• Action Statement 6b states that further evaluation should be conducted if there is a recurrence of febrile UTI. This statement is supported by level X evidence, indicating that the "recommendation" is based on "exceptional situations where validating studies cannot be performed and there is a clear preponderance of benefit or harm."
Specifically, this statement reflects the fact that studies in recent years have brought into question the value of prophylaxis for preventing recurrent UTI, as well as the rationale for performing VCUG routinely after an initial febrile UTI.
The previous guideline included strong encouragement of imaging studies despite insufficient evidence supporting routine VCUG, but the "position of the current subcommittee reflects the new evidence demonstrating antimicrobial prophylaxis not to be effective as presumed previously. Moreover, prompt diagnosis and effective treatment of a febrile UTI recurrence may be of greater importance regardless of whether VUR is present or the child is receiving antimicrobial prophylaxis," they wrote.
• Action Statement 7. This recommendation states that after confirmation of a UTI, the clinician should instruct parents or guardians to seek prompt medical evaluations – ideally within 48 hours – if febrile illness recurs.
This is to ensure that recent infections are detected and treated promptly, they noted. Level C evidence supports this "recommendation."
"Early treatment limits renal damage better than late treatment, and the risk of renal scarring increases as the number of recurrences increases. For these reasons, all infants who have sustained a febrile UTI should have a urine specimen obtained at the onset of subsequent febrile illnesses so that UTI can be diagnosed and treated promptly," they wrote.
This updated guideline focuses on diagnosis and management of an initial UTI in children aged 2-24 months with no obvious neurologic or anatomic abnormalities known to be associated with recurrent UTI or renal damage. Fever for the purpose of the guideline was defined as a temperature of at least 100.4° F.
In developing the update, the subcommittee performed literature searches, including surveillance of Medline-listed literature over the past 10 years, and – in light of the increasing evidence questioning the value of antimicrobial prophylaxis to prevent recurrent febrile UTI in children with VUR – they also performed a systematic review of literature on the effectiveness of prophylactic antimicrobial therapy to prevent recurrent febrile UTI in this population. Using data from six randomized controlled trials for which raw data on infants aged 2-24 months were available, they also performed a meta-analysis addressing the effectiveness of prophylactic antimicrobial therapy. The findings are published in the companion technical report (Pediatrics 2011;128:e749–e770 [doi:10.1542/peds.2011-1332).
The authors of the guideline stressed that their recommendations do not represent standard of care or indicate an exclusive course of treatment, but rather are intended to assist clinicians in decision-making.
In his accompanying editorial, Dr. Newman praised many aspects of the updated guideline, but also highlighted some instances where he believes "alternative recommendations are supported by available evidence."
For example, while he agrees that the recommendation on selective urine testing based on the prior probability of UTI is "an important improvement," and he lauded the subcommittee’s job summarizing the factors that determine that probability, he questioned the assertion that the threshold probability for urine testing is below 3%, and proposed that until this is proven, the decision to test should be individualized.
Additionally, while he said he is "glad the new guideline continues to offer the option of obtaining urine for urinalyses noninvasively," he also said he is not convinced that the bag urine can never be used for culture. "If the urinalysis is used to select urine for culture, the prior probability may sometimes be in a range where the bag culture will be useful," he explained.
Despite these and other concerns addressed by Dr. Newman, he saluted the authors and noted that the updated guideline should prove helpful to clinicians and families dealing with this common problem.
Members of the subcommittee that developed this guideline included individuals with expertise in the fields of academic general pediatrics, epidemiology and informatics, pediatric infectious disease, pediatric nephrology, pediatric practice, pediatric radiology, and pediatric urology. The AAP funded the development of the guidelines; none of the subcommittee members had financial conflicts of interest. Dr. Newman also had no financial conflicts to report.
FROM PEDIATRICS