User login
Sharon Worcester is an award-winning medical journalist for MDedge News. She has been with the company since 1996, first as the Southeast Bureau Chief (1996-2009) when the company was known as International Medical News Group, then as a freelance writer (2010-2015) before returning as a reporter in 2015. She previously worked as a daily newspaper reporter covering health and local government. Sharon currently reports primarily on oncology and hematology. She has a BA from Eckerd College and an MA in Mass Communication/Print Journalism from the University of Florida. Connect with her via LinkedIn and follow her on twitter @SW_MedReporter.
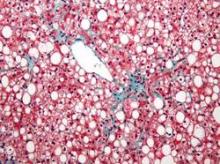
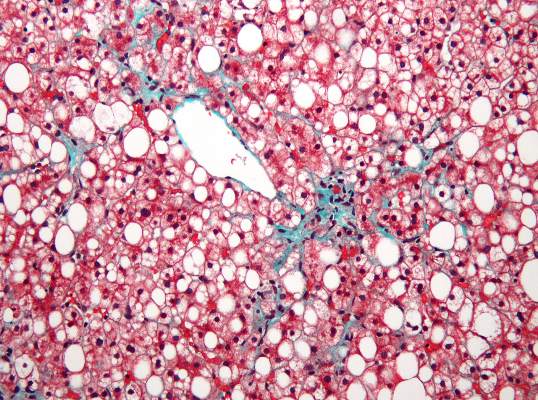
Aramchol shows promise for NAFLD; resveratrol disappoints
Administration of a novel fatty acid–bile acid conjugate safely and significantly reduced liver fat content in patients with nonalcoholic fatty liver disease in a phase II trial.
Administration of resveratrol for 8 weeks, however, failed to improve any features of the disease in a separate placebo-controlled study.
In 60 patients aged 18-75 years in a randomized, double-blind, placebo-controlled study of the fatty acid–bile acid conjugate aramchol (Trima Israel Pharmaceutical Products Ltd. Maabarot), 3 months of treatment was associated with a decrease in liver fat content by a mean of 12.57% as measured by magnetic resonance spectroscopy, compared with an increase in liver fat by a mean of 6.39% in patients who received placebo, Dr. Rifaat Safadi of Hadassah University Medical Center, Jerusalem and colleagues reported in the DEcember issue of Clinical Gastroenterology and Hepatology.
The difference between the treatment and placebo groups was significant, even after age, sex, and body mass index were adjusted for.
Liver fat content also decreased in patients who received treatment with a 100-mg dose of aramchol, but the difference compared with placebo was not statistically significant, indicating a dose-response relationship in treated patients, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.1016/j.cgh.2014.04.038]).
A trend was seen for improvements over time in endothelial function and levels of alanine aminotransferase and adiponectin.
Patients in the study were from 10 centers in Israel, and all had biopsy-confirmed NAFLD (54 patients) or nonalcoholic steatohepatitis (NASH, 6 patients). They were randomized to receive 100 or 300 mg of aramchol or placebo once daily for 3 months.
No serious or drug-related adverse events occurred in the treated patients, the investigators said.
The findings suggest that aramchol is a candidate for the treatment of fatty liver–related diseases, which are “an increasingly relevant public health issue because of their association with the worldwide epidemics of diabetes and obesity,” and for which treatments are lacking, they said, concluding that longer trials in patients with NASH and metabolic complications are warranted to evaluate metabolic and histologic benefits of treatment.
Findings from the resveratrol study were less encouraging.
Eight weeks of treatment at 3,000 mg daily in overweight or obese men with NAFLD who were recruited from clinics in Brisbane from 2011 through 2012 was no better than placebo for improving insulin resistance, hepatic steatosis, or abdominal fat distribution, Dr. Veronique S. Chachay of the University of Queensland, Brisbane, Australia and her colleagues also reported in the December issue of Clinical Gastroenterology and Hepatology.
No changes were observed in plasma lipids or antioxidant activity, and levels of alanine and aspartate aminotransferases increased significantly (from 45 to 63 U/L and from 35 to 45 U/L, respectively) until week 6 in the 10 treated patients, compared with the 10 who received placebo (40 to 48 U/L and 36 to 38 U/L, respectively). Resveratrol did not significantly alter transcription of NQ01, PTP1B, IL6, or H01, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.106/j.cgh.2014.02.024]).
NAFLD is associated with abdominal obesity, insulin resistance, and inflammation, and while weight loss via calorie restriction is known to reduce features of the disease, there is no pharmacologic therapy available to achieve this end.
The investigators sought to determine if resveratrol – a polyphenol that has been shown to prevent high-energy diet-induced steatosis and insulin resistance in animals – might be beneficial in patients with NAFLD.
“The present study demonstrates that the preventive role of resveratrol observed in diet-induced preclinical models of NAFLD does not translate into a therapeutic role in clinically established NAFLD,” they said, adding that clinical dose-finding studies are “paramount to elucidate the dose-response relationship” and that “the purported calorie-restriction mimicking of resveratrol may require investigation in combination with dietary prescription, standard care, and lifestyle modifications to target adequately the complexity of dysregulation in obesity-related chronic disease.”
The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
Administration of a novel fatty acid–bile acid conjugate safely and significantly reduced liver fat content in patients with nonalcoholic fatty liver disease in a phase II trial.
Administration of resveratrol for 8 weeks, however, failed to improve any features of the disease in a separate placebo-controlled study.
In 60 patients aged 18-75 years in a randomized, double-blind, placebo-controlled study of the fatty acid–bile acid conjugate aramchol (Trima Israel Pharmaceutical Products Ltd. Maabarot), 3 months of treatment was associated with a decrease in liver fat content by a mean of 12.57% as measured by magnetic resonance spectroscopy, compared with an increase in liver fat by a mean of 6.39% in patients who received placebo, Dr. Rifaat Safadi of Hadassah University Medical Center, Jerusalem and colleagues reported in the DEcember issue of Clinical Gastroenterology and Hepatology.
The difference between the treatment and placebo groups was significant, even after age, sex, and body mass index were adjusted for.
Liver fat content also decreased in patients who received treatment with a 100-mg dose of aramchol, but the difference compared with placebo was not statistically significant, indicating a dose-response relationship in treated patients, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.1016/j.cgh.2014.04.038]).
A trend was seen for improvements over time in endothelial function and levels of alanine aminotransferase and adiponectin.
Patients in the study were from 10 centers in Israel, and all had biopsy-confirmed NAFLD (54 patients) or nonalcoholic steatohepatitis (NASH, 6 patients). They were randomized to receive 100 or 300 mg of aramchol or placebo once daily for 3 months.
No serious or drug-related adverse events occurred in the treated patients, the investigators said.
The findings suggest that aramchol is a candidate for the treatment of fatty liver–related diseases, which are “an increasingly relevant public health issue because of their association with the worldwide epidemics of diabetes and obesity,” and for which treatments are lacking, they said, concluding that longer trials in patients with NASH and metabolic complications are warranted to evaluate metabolic and histologic benefits of treatment.
Findings from the resveratrol study were less encouraging.
Eight weeks of treatment at 3,000 mg daily in overweight or obese men with NAFLD who were recruited from clinics in Brisbane from 2011 through 2012 was no better than placebo for improving insulin resistance, hepatic steatosis, or abdominal fat distribution, Dr. Veronique S. Chachay of the University of Queensland, Brisbane, Australia and her colleagues also reported in the December issue of Clinical Gastroenterology and Hepatology.
No changes were observed in plasma lipids or antioxidant activity, and levels of alanine and aspartate aminotransferases increased significantly (from 45 to 63 U/L and from 35 to 45 U/L, respectively) until week 6 in the 10 treated patients, compared with the 10 who received placebo (40 to 48 U/L and 36 to 38 U/L, respectively). Resveratrol did not significantly alter transcription of NQ01, PTP1B, IL6, or H01, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.106/j.cgh.2014.02.024]).
NAFLD is associated with abdominal obesity, insulin resistance, and inflammation, and while weight loss via calorie restriction is known to reduce features of the disease, there is no pharmacologic therapy available to achieve this end.
The investigators sought to determine if resveratrol – a polyphenol that has been shown to prevent high-energy diet-induced steatosis and insulin resistance in animals – might be beneficial in patients with NAFLD.
“The present study demonstrates that the preventive role of resveratrol observed in diet-induced preclinical models of NAFLD does not translate into a therapeutic role in clinically established NAFLD,” they said, adding that clinical dose-finding studies are “paramount to elucidate the dose-response relationship” and that “the purported calorie-restriction mimicking of resveratrol may require investigation in combination with dietary prescription, standard care, and lifestyle modifications to target adequately the complexity of dysregulation in obesity-related chronic disease.”
The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
Administration of a novel fatty acid–bile acid conjugate safely and significantly reduced liver fat content in patients with nonalcoholic fatty liver disease in a phase II trial.
Administration of resveratrol for 8 weeks, however, failed to improve any features of the disease in a separate placebo-controlled study.
In 60 patients aged 18-75 years in a randomized, double-blind, placebo-controlled study of the fatty acid–bile acid conjugate aramchol (Trima Israel Pharmaceutical Products Ltd. Maabarot), 3 months of treatment was associated with a decrease in liver fat content by a mean of 12.57% as measured by magnetic resonance spectroscopy, compared with an increase in liver fat by a mean of 6.39% in patients who received placebo, Dr. Rifaat Safadi of Hadassah University Medical Center, Jerusalem and colleagues reported in the DEcember issue of Clinical Gastroenterology and Hepatology.
The difference between the treatment and placebo groups was significant, even after age, sex, and body mass index were adjusted for.
Liver fat content also decreased in patients who received treatment with a 100-mg dose of aramchol, but the difference compared with placebo was not statistically significant, indicating a dose-response relationship in treated patients, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.1016/j.cgh.2014.04.038]).
A trend was seen for improvements over time in endothelial function and levels of alanine aminotransferase and adiponectin.
Patients in the study were from 10 centers in Israel, and all had biopsy-confirmed NAFLD (54 patients) or nonalcoholic steatohepatitis (NASH, 6 patients). They were randomized to receive 100 or 300 mg of aramchol or placebo once daily for 3 months.
No serious or drug-related adverse events occurred in the treated patients, the investigators said.
The findings suggest that aramchol is a candidate for the treatment of fatty liver–related diseases, which are “an increasingly relevant public health issue because of their association with the worldwide epidemics of diabetes and obesity,” and for which treatments are lacking, they said, concluding that longer trials in patients with NASH and metabolic complications are warranted to evaluate metabolic and histologic benefits of treatment.
Findings from the resveratrol study were less encouraging.
Eight weeks of treatment at 3,000 mg daily in overweight or obese men with NAFLD who were recruited from clinics in Brisbane from 2011 through 2012 was no better than placebo for improving insulin resistance, hepatic steatosis, or abdominal fat distribution, Dr. Veronique S. Chachay of the University of Queensland, Brisbane, Australia and her colleagues also reported in the December issue of Clinical Gastroenterology and Hepatology.
No changes were observed in plasma lipids or antioxidant activity, and levels of alanine and aspartate aminotransferases increased significantly (from 45 to 63 U/L and from 35 to 45 U/L, respectively) until week 6 in the 10 treated patients, compared with the 10 who received placebo (40 to 48 U/L and 36 to 38 U/L, respectively). Resveratrol did not significantly alter transcription of NQ01, PTP1B, IL6, or H01, the investigators said (Clin Gastroenterol Hepatol 2014[doi:10.106/j.cgh.2014.02.024]).
NAFLD is associated with abdominal obesity, insulin resistance, and inflammation, and while weight loss via calorie restriction is known to reduce features of the disease, there is no pharmacologic therapy available to achieve this end.
The investigators sought to determine if resveratrol – a polyphenol that has been shown to prevent high-energy diet-induced steatosis and insulin resistance in animals – might be beneficial in patients with NAFLD.
“The present study demonstrates that the preventive role of resveratrol observed in diet-induced preclinical models of NAFLD does not translate into a therapeutic role in clinically established NAFLD,” they said, adding that clinical dose-finding studies are “paramount to elucidate the dose-response relationship” and that “the purported calorie-restriction mimicking of resveratrol may require investigation in combination with dietary prescription, standard care, and lifestyle modifications to target adequately the complexity of dysregulation in obesity-related chronic disease.”
The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
Key clinical point: Aramchol is a candidate treatment for fatty liver–related diseases; resveratrol requires dose-finding and other studies.
Major finding: The change in liver fat content was a mean of -12.57% with aramchol vs. +6.39% with placebo. Resveratol had no therapeutic benefit in NAFLD.
Data source: A phase II study of 60 patients, and a randomized, controlled study of 20 patients.
Disclosures: The aramchol study was supported by Galmed Medical Research, Ltd. Dr. Safadi reported having no disclosures. The resveratrol study was supported by the Princess Alexandra Research Foundation, the Lions Medical Research Foundation, and the National Health and Medical Research Council of Australia. The authors reported having no disclosures.
Infliximab serum concentrations, efficacy linked in ulcerative colitis
Higher serum concentrations of infliximab are associated with clinical response, mucosal healing, and clinical remission in adults with moderate to severe ulcerative colitis, according to post hoc analyses of data from the Active Ulcerative Colitis Trials, ACT-1 and ACT-2.
In the 728 patients from the two randomized, controlled phase III pivotal trials, median serum concentrations of infliximab at week 8 were higher among patients with clinical response or mucosal healing during induction than in those who did not achieve these endpoints. For example, the median concentration among those who received 5-mg/kg doses of infliximab was 35.0 mcg/mL in responders, compared with 25.8 mcg/mL in nonresponders, Omoniyi J. Adedokun of Janssen Research and Development, Spring House, Pa., and colleagues reported in the December issue of Gastroenterology [doi:10.1053/j.gastro.2014.08.035].
“Similar results were observed for clinical response and mucosal healing during maintenance at week 30 and week 54,” the investigators wrote, noting that in the 5-mg/kg group, the median trough serum infliximab concentration was several-fold higher in responders than in nonresponders (3.9 vs. 1.2 mcg/mL at week 30 and 5.0 vs. 0.7 mcg/mL at week 54).
Concentrations did not differ significantly at 8 weeks in remitters and nonremitters in the 5-mg/kg group, but they did in patients who received 10 mg/kg and in patients in both dose groups at weeks 30 and 54, the investigators reported.
When assessed by infliximab concentration quartiles, treatment efficacy – defined by clinical response, mucosal healing, and/or clinical remission – generally improved with increasing concentrations in patients in both the 5- and 10-mg/kg groups; those with concentrations in the lowest quartile consistently were less likely to show clinical response, clinical remission, or mucosal healing, and had rates of success approaching those observed in the placebo groups.
Optimal infliximab concentration target thresholds associated with clinical improvement in ulcerative colitis patients in these analyses were 41 mcg/mL at week 8 (sensitivity, specificity, and positive predictive values of 63%, 62%, and 80%, respectively) and 3.7 mcg/mL at week 30 for maintenance of clinical response (sensitivity, specificity, and positive predictive values of 65%, 71%, and 82%, respectively). The data at week 54 suggested a range for serum infliximab concentrations of similar sensitivity, specificity, and positive predictive value, but those data represented only a subset of patients assessed, the investigators said.
Patients who achieved an efficacy outcome, but who failed to maintain that outcome, had lower serum infliximab concentrations earlier in the course of therapy than did those who maintained the outcome, the investigators said.
“In general, the lower the infliximab concentration at a given time point, the more likely the patients were to fail to maintain remission,” they wrote.
The findings demonstrate a strong association between serum infliximab concentration and efficacy outcomes in patients with ulcerative colitis, and highlight the possibility of infliximab dose optimization – particularly in patients who are likely to lose efficacy while receiving a standard dose of infliximab, the investigators said.
Target threshold concentrations identified by this analysis could help clinicians understand why an individual patient fails to achieve the expected efficacy, but a prospective study designed to confirm the importance of optimizing infliximab concentrations is needed before it can be determined whether these results can be exploited to achieve better outcomes for patients with ulcerative colitis, Mr. Adedokun and coinvestigators said.
The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Higher serum concentrations of infliximab are associated with clinical response, mucosal healing, and clinical remission in adults with moderate to severe ulcerative colitis, according to post hoc analyses of data from the Active Ulcerative Colitis Trials, ACT-1 and ACT-2.
In the 728 patients from the two randomized, controlled phase III pivotal trials, median serum concentrations of infliximab at week 8 were higher among patients with clinical response or mucosal healing during induction than in those who did not achieve these endpoints. For example, the median concentration among those who received 5-mg/kg doses of infliximab was 35.0 mcg/mL in responders, compared with 25.8 mcg/mL in nonresponders, Omoniyi J. Adedokun of Janssen Research and Development, Spring House, Pa., and colleagues reported in the December issue of Gastroenterology [doi:10.1053/j.gastro.2014.08.035].
“Similar results were observed for clinical response and mucosal healing during maintenance at week 30 and week 54,” the investigators wrote, noting that in the 5-mg/kg group, the median trough serum infliximab concentration was several-fold higher in responders than in nonresponders (3.9 vs. 1.2 mcg/mL at week 30 and 5.0 vs. 0.7 mcg/mL at week 54).
Concentrations did not differ significantly at 8 weeks in remitters and nonremitters in the 5-mg/kg group, but they did in patients who received 10 mg/kg and in patients in both dose groups at weeks 30 and 54, the investigators reported.
When assessed by infliximab concentration quartiles, treatment efficacy – defined by clinical response, mucosal healing, and/or clinical remission – generally improved with increasing concentrations in patients in both the 5- and 10-mg/kg groups; those with concentrations in the lowest quartile consistently were less likely to show clinical response, clinical remission, or mucosal healing, and had rates of success approaching those observed in the placebo groups.
Optimal infliximab concentration target thresholds associated with clinical improvement in ulcerative colitis patients in these analyses were 41 mcg/mL at week 8 (sensitivity, specificity, and positive predictive values of 63%, 62%, and 80%, respectively) and 3.7 mcg/mL at week 30 for maintenance of clinical response (sensitivity, specificity, and positive predictive values of 65%, 71%, and 82%, respectively). The data at week 54 suggested a range for serum infliximab concentrations of similar sensitivity, specificity, and positive predictive value, but those data represented only a subset of patients assessed, the investigators said.
Patients who achieved an efficacy outcome, but who failed to maintain that outcome, had lower serum infliximab concentrations earlier in the course of therapy than did those who maintained the outcome, the investigators said.
“In general, the lower the infliximab concentration at a given time point, the more likely the patients were to fail to maintain remission,” they wrote.
The findings demonstrate a strong association between serum infliximab concentration and efficacy outcomes in patients with ulcerative colitis, and highlight the possibility of infliximab dose optimization – particularly in patients who are likely to lose efficacy while receiving a standard dose of infliximab, the investigators said.
Target threshold concentrations identified by this analysis could help clinicians understand why an individual patient fails to achieve the expected efficacy, but a prospective study designed to confirm the importance of optimizing infliximab concentrations is needed before it can be determined whether these results can be exploited to achieve better outcomes for patients with ulcerative colitis, Mr. Adedokun and coinvestigators said.
The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Higher serum concentrations of infliximab are associated with clinical response, mucosal healing, and clinical remission in adults with moderate to severe ulcerative colitis, according to post hoc analyses of data from the Active Ulcerative Colitis Trials, ACT-1 and ACT-2.
In the 728 patients from the two randomized, controlled phase III pivotal trials, median serum concentrations of infliximab at week 8 were higher among patients with clinical response or mucosal healing during induction than in those who did not achieve these endpoints. For example, the median concentration among those who received 5-mg/kg doses of infliximab was 35.0 mcg/mL in responders, compared with 25.8 mcg/mL in nonresponders, Omoniyi J. Adedokun of Janssen Research and Development, Spring House, Pa., and colleagues reported in the December issue of Gastroenterology [doi:10.1053/j.gastro.2014.08.035].
“Similar results were observed for clinical response and mucosal healing during maintenance at week 30 and week 54,” the investigators wrote, noting that in the 5-mg/kg group, the median trough serum infliximab concentration was several-fold higher in responders than in nonresponders (3.9 vs. 1.2 mcg/mL at week 30 and 5.0 vs. 0.7 mcg/mL at week 54).
Concentrations did not differ significantly at 8 weeks in remitters and nonremitters in the 5-mg/kg group, but they did in patients who received 10 mg/kg and in patients in both dose groups at weeks 30 and 54, the investigators reported.
When assessed by infliximab concentration quartiles, treatment efficacy – defined by clinical response, mucosal healing, and/or clinical remission – generally improved with increasing concentrations in patients in both the 5- and 10-mg/kg groups; those with concentrations in the lowest quartile consistently were less likely to show clinical response, clinical remission, or mucosal healing, and had rates of success approaching those observed in the placebo groups.
Optimal infliximab concentration target thresholds associated with clinical improvement in ulcerative colitis patients in these analyses were 41 mcg/mL at week 8 (sensitivity, specificity, and positive predictive values of 63%, 62%, and 80%, respectively) and 3.7 mcg/mL at week 30 for maintenance of clinical response (sensitivity, specificity, and positive predictive values of 65%, 71%, and 82%, respectively). The data at week 54 suggested a range for serum infliximab concentrations of similar sensitivity, specificity, and positive predictive value, but those data represented only a subset of patients assessed, the investigators said.
Patients who achieved an efficacy outcome, but who failed to maintain that outcome, had lower serum infliximab concentrations earlier in the course of therapy than did those who maintained the outcome, the investigators said.
“In general, the lower the infliximab concentration at a given time point, the more likely the patients were to fail to maintain remission,” they wrote.
The findings demonstrate a strong association between serum infliximab concentration and efficacy outcomes in patients with ulcerative colitis, and highlight the possibility of infliximab dose optimization – particularly in patients who are likely to lose efficacy while receiving a standard dose of infliximab, the investigators said.
Target threshold concentrations identified by this analysis could help clinicians understand why an individual patient fails to achieve the expected efficacy, but a prospective study designed to confirm the importance of optimizing infliximab concentrations is needed before it can be determined whether these results can be exploited to achieve better outcomes for patients with ulcerative colitis, Mr. Adedokun and coinvestigators said.
The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Key clinical point: Serum concentrations of infliximab are associated with response, and could allow for dose optimization.
Major finding: Median concentrations of infliximab at 8 weeks in those receiving 5 mg/kg were 35.0 mcg/mL in responders vs. 25.8 mcg/mL in nonresponders.
Data source: Post hoc analyses of the ACT trials involving 728 patients.
Disclosures: The ACT trials were funded by Janssen Research and Development, which also employs Mr. Adedokun.
Endoscopic mucosal resection proves effective, durable for Barrett’s-associated neoplasia
Complete endoscopic mucosal resection is an effective, durable, and relatively safe treatment for Barrett’s esophagus with high-grade dysplasia, findings from a series of 107 patients suggest.
A particular benefit of endoscopic mucosal resection is that it provides large and intact tissue specimens that allow for accurate staging, according to a report by Dr. Vani J.A. Konda of the University of Chicago and her colleagues in the December issue of Clinical Gastroenterology and Hepatology (doi:10.1016/j.cgh.2014.04.010).
Based on an intention-to-treat analysis, the approach resulted in complete eradication of Barrett’s esophagus in 86 of the 107 patients (80%) who were referred to the Center for Endoscopic Research and Therapeutics at the University of Chicago between August 2003 and December 2012 for Barrett’s esophagus with suspected high-grade dysplasia or intramucosal carcinoma. Based on a per-protocol analysis, the approach resulted in complete eradication in 79 of 80 patients (98.8%), the investigators reported.
Endoscopic mucosal resection resulted in a change in the diagnosis in 27 cases (25.2%), which were upstaged based on assessment of the resection specimens. Four of the cases in which the diagnosis was changed were initially diagnosed as high-grade dysplasia on biopsy and were found on endoscopic mucosal resection to have evidence of submucosal invasion, they said.
Patients included in the series had a mean lesion length of 3.6 cm. All patients underwent complete endoscopic mucosal resection performed on an outpatient basis by a single endoscopist and were followed through January 2014 for a median of 33 months. Most (78.5%) were treated using the cap-assisted technique, while 8.4% were treated using band ligation, 2.8% were treated using a mixed band ligation and cap technique, and 11.2% were treated using an injection-assisted, free-hand technique. Two patients with suspected submucosal invasion underwent a combination of endoscopic submucosal dissection and endoscopic mucosal resection.
Esophageal strictures occurred in 44 patients (41.1%) and 40 were symptomatic; strictures required an average of 2.3 dilations. Symptomatic dysphagia developed in 37.3% of patients, and perforations occurred in two patients, suggesting a need for surgical back-up for patients undergoing endoscopic mucosal resection, the investigators said.High-grade dysplasia and intramucosal carcinoma each recurred in one patient, and both were treated successfully with endoscopic mucosal resection, they reported.
At the most recent analysis, 53 of 74 patients (71.6%) in a durability cohort were in complete remission from intestinal metaplasia, and all were in complete remission from high-grade dysplasia or intramucosal carcinoma, they noted.
Endoscopic therapy is now preferred over esophagectomy in most cases of Barrett’s esophagus with high-grade dysplasia or intramucosal carcinoma, and the current findings show that complete endoscopic mucosal resection is an effective and durable method to treat Barrett’s-associated neoplasia, the investigators concluded, adding that the results support the replacement of esophagectomy by endoscopic therapy for the condition.
“The most commonly encountered complication of esophageal stricture formation can be addressed with endoscopic dilation,” the investigators said.
However, long-term outcomes, particularly in those with advanced pathology who undergo endoscopic management, are still needed, as is an improved understanding of the behavior of the disease following treatment with specific modalities in order to optimize endoscopic therapy, they added.
This study was supported by the Francis L. Lederer Foundation. The authors reported having no disclosures.
Localized treatments aimed at the eradication of dysplasia, early cancer, and surrounding mucosa in Barrett’s esophagus have been a major advancement in gastroenterology. Although radiofrequency ablation (RFA) has become the procedure of choice, debate exists regarding the role of other mucosal extirpative procedures. The work by Dr. Konda and her colleagues gives life to another proposed procedure, complete endoscopic mucosal resection (EMR) of dysplastic and Barrett’s mucosa. In this study, complete EMR led to results comparable with previous reports on RFA (Gastroenterology 2013;145:96-104). EMR also has theoretical advantages over RFA, including avoidance of promoting protumorigenic mutations and clonal expansion in remaining mucosa (Am. J. Gastroenterol. 2013;108:1584-92).

|
| Dr. David A. Katzka |
EMR, like RFA, however, is not risk free; perforations occurred in three patients (one during dilation) and 38% developed symptomatic strictures. Furthermore, the endoscopic skill required to complete this protocol also necessitated expertise in cap technique, band ligation, injection-assisted free-hand technique, focal RFA, and endoscopic submucosal dissection. In other words, performing complete EMR is for a select group of endoscopic intensivists only. Unfortunately, endoscopic eradication of Barrett’s mucosa remains a procedure that is betwixt and between. It is between the mucosal and submucosal layer but is betwixt because of limitations on the depth of penetration of RFA (around 0.7 mm) and EMR (Dig. Dis. 2009;26:280-4) needed to eradicate the elusive Barrett’s stem cell, coupled with the increasing chance of esophageal stricture formation with these deeper levels of injury. Nevertheless, work such as this from Dr. Konda and her colleagues allows us to continue to claim at least a partial victory over Barrett’s dysplasia. It is hoped that with future techniques, we may come up with a “kinder” and easier method of removing all Barrett’s mucosa without the provocation of a substantial fibrotic reaction of the remaining esophageal wall.
Dr. David A. Katzka is in the department of gastroenterology and hepatology, Mayo Clinic, Rochester, Minn. He has no conflicts of interest.
Localized treatments aimed at the eradication of dysplasia, early cancer, and surrounding mucosa in Barrett’s esophagus have been a major advancement in gastroenterology. Although radiofrequency ablation (RFA) has become the procedure of choice, debate exists regarding the role of other mucosal extirpative procedures. The work by Dr. Konda and her colleagues gives life to another proposed procedure, complete endoscopic mucosal resection (EMR) of dysplastic and Barrett’s mucosa. In this study, complete EMR led to results comparable with previous reports on RFA (Gastroenterology 2013;145:96-104). EMR also has theoretical advantages over RFA, including avoidance of promoting protumorigenic mutations and clonal expansion in remaining mucosa (Am. J. Gastroenterol. 2013;108:1584-92).

|
| Dr. David A. Katzka |
EMR, like RFA, however, is not risk free; perforations occurred in three patients (one during dilation) and 38% developed symptomatic strictures. Furthermore, the endoscopic skill required to complete this protocol also necessitated expertise in cap technique, band ligation, injection-assisted free-hand technique, focal RFA, and endoscopic submucosal dissection. In other words, performing complete EMR is for a select group of endoscopic intensivists only. Unfortunately, endoscopic eradication of Barrett’s mucosa remains a procedure that is betwixt and between. It is between the mucosal and submucosal layer but is betwixt because of limitations on the depth of penetration of RFA (around 0.7 mm) and EMR (Dig. Dis. 2009;26:280-4) needed to eradicate the elusive Barrett’s stem cell, coupled with the increasing chance of esophageal stricture formation with these deeper levels of injury. Nevertheless, work such as this from Dr. Konda and her colleagues allows us to continue to claim at least a partial victory over Barrett’s dysplasia. It is hoped that with future techniques, we may come up with a “kinder” and easier method of removing all Barrett’s mucosa without the provocation of a substantial fibrotic reaction of the remaining esophageal wall.
Dr. David A. Katzka is in the department of gastroenterology and hepatology, Mayo Clinic, Rochester, Minn. He has no conflicts of interest.
Localized treatments aimed at the eradication of dysplasia, early cancer, and surrounding mucosa in Barrett’s esophagus have been a major advancement in gastroenterology. Although radiofrequency ablation (RFA) has become the procedure of choice, debate exists regarding the role of other mucosal extirpative procedures. The work by Dr. Konda and her colleagues gives life to another proposed procedure, complete endoscopic mucosal resection (EMR) of dysplastic and Barrett’s mucosa. In this study, complete EMR led to results comparable with previous reports on RFA (Gastroenterology 2013;145:96-104). EMR also has theoretical advantages over RFA, including avoidance of promoting protumorigenic mutations and clonal expansion in remaining mucosa (Am. J. Gastroenterol. 2013;108:1584-92).

|
| Dr. David A. Katzka |
EMR, like RFA, however, is not risk free; perforations occurred in three patients (one during dilation) and 38% developed symptomatic strictures. Furthermore, the endoscopic skill required to complete this protocol also necessitated expertise in cap technique, band ligation, injection-assisted free-hand technique, focal RFA, and endoscopic submucosal dissection. In other words, performing complete EMR is for a select group of endoscopic intensivists only. Unfortunately, endoscopic eradication of Barrett’s mucosa remains a procedure that is betwixt and between. It is between the mucosal and submucosal layer but is betwixt because of limitations on the depth of penetration of RFA (around 0.7 mm) and EMR (Dig. Dis. 2009;26:280-4) needed to eradicate the elusive Barrett’s stem cell, coupled with the increasing chance of esophageal stricture formation with these deeper levels of injury. Nevertheless, work such as this from Dr. Konda and her colleagues allows us to continue to claim at least a partial victory over Barrett’s dysplasia. It is hoped that with future techniques, we may come up with a “kinder” and easier method of removing all Barrett’s mucosa without the provocation of a substantial fibrotic reaction of the remaining esophageal wall.
Dr. David A. Katzka is in the department of gastroenterology and hepatology, Mayo Clinic, Rochester, Minn. He has no conflicts of interest.
Complete endoscopic mucosal resection is an effective, durable, and relatively safe treatment for Barrett’s esophagus with high-grade dysplasia, findings from a series of 107 patients suggest.
A particular benefit of endoscopic mucosal resection is that it provides large and intact tissue specimens that allow for accurate staging, according to a report by Dr. Vani J.A. Konda of the University of Chicago and her colleagues in the December issue of Clinical Gastroenterology and Hepatology (doi:10.1016/j.cgh.2014.04.010).
Based on an intention-to-treat analysis, the approach resulted in complete eradication of Barrett’s esophagus in 86 of the 107 patients (80%) who were referred to the Center for Endoscopic Research and Therapeutics at the University of Chicago between August 2003 and December 2012 for Barrett’s esophagus with suspected high-grade dysplasia or intramucosal carcinoma. Based on a per-protocol analysis, the approach resulted in complete eradication in 79 of 80 patients (98.8%), the investigators reported.
Endoscopic mucosal resection resulted in a change in the diagnosis in 27 cases (25.2%), which were upstaged based on assessment of the resection specimens. Four of the cases in which the diagnosis was changed were initially diagnosed as high-grade dysplasia on biopsy and were found on endoscopic mucosal resection to have evidence of submucosal invasion, they said.
Patients included in the series had a mean lesion length of 3.6 cm. All patients underwent complete endoscopic mucosal resection performed on an outpatient basis by a single endoscopist and were followed through January 2014 for a median of 33 months. Most (78.5%) were treated using the cap-assisted technique, while 8.4% were treated using band ligation, 2.8% were treated using a mixed band ligation and cap technique, and 11.2% were treated using an injection-assisted, free-hand technique. Two patients with suspected submucosal invasion underwent a combination of endoscopic submucosal dissection and endoscopic mucosal resection.
Esophageal strictures occurred in 44 patients (41.1%) and 40 were symptomatic; strictures required an average of 2.3 dilations. Symptomatic dysphagia developed in 37.3% of patients, and perforations occurred in two patients, suggesting a need for surgical back-up for patients undergoing endoscopic mucosal resection, the investigators said.High-grade dysplasia and intramucosal carcinoma each recurred in one patient, and both were treated successfully with endoscopic mucosal resection, they reported.
At the most recent analysis, 53 of 74 patients (71.6%) in a durability cohort were in complete remission from intestinal metaplasia, and all were in complete remission from high-grade dysplasia or intramucosal carcinoma, they noted.
Endoscopic therapy is now preferred over esophagectomy in most cases of Barrett’s esophagus with high-grade dysplasia or intramucosal carcinoma, and the current findings show that complete endoscopic mucosal resection is an effective and durable method to treat Barrett’s-associated neoplasia, the investigators concluded, adding that the results support the replacement of esophagectomy by endoscopic therapy for the condition.
“The most commonly encountered complication of esophageal stricture formation can be addressed with endoscopic dilation,” the investigators said.
However, long-term outcomes, particularly in those with advanced pathology who undergo endoscopic management, are still needed, as is an improved understanding of the behavior of the disease following treatment with specific modalities in order to optimize endoscopic therapy, they added.
This study was supported by the Francis L. Lederer Foundation. The authors reported having no disclosures.
Complete endoscopic mucosal resection is an effective, durable, and relatively safe treatment for Barrett’s esophagus with high-grade dysplasia, findings from a series of 107 patients suggest.
A particular benefit of endoscopic mucosal resection is that it provides large and intact tissue specimens that allow for accurate staging, according to a report by Dr. Vani J.A. Konda of the University of Chicago and her colleagues in the December issue of Clinical Gastroenterology and Hepatology (doi:10.1016/j.cgh.2014.04.010).
Based on an intention-to-treat analysis, the approach resulted in complete eradication of Barrett’s esophagus in 86 of the 107 patients (80%) who were referred to the Center for Endoscopic Research and Therapeutics at the University of Chicago between August 2003 and December 2012 for Barrett’s esophagus with suspected high-grade dysplasia or intramucosal carcinoma. Based on a per-protocol analysis, the approach resulted in complete eradication in 79 of 80 patients (98.8%), the investigators reported.
Endoscopic mucosal resection resulted in a change in the diagnosis in 27 cases (25.2%), which were upstaged based on assessment of the resection specimens. Four of the cases in which the diagnosis was changed were initially diagnosed as high-grade dysplasia on biopsy and were found on endoscopic mucosal resection to have evidence of submucosal invasion, they said.
Patients included in the series had a mean lesion length of 3.6 cm. All patients underwent complete endoscopic mucosal resection performed on an outpatient basis by a single endoscopist and were followed through January 2014 for a median of 33 months. Most (78.5%) were treated using the cap-assisted technique, while 8.4% were treated using band ligation, 2.8% were treated using a mixed band ligation and cap technique, and 11.2% were treated using an injection-assisted, free-hand technique. Two patients with suspected submucosal invasion underwent a combination of endoscopic submucosal dissection and endoscopic mucosal resection.
Esophageal strictures occurred in 44 patients (41.1%) and 40 were symptomatic; strictures required an average of 2.3 dilations. Symptomatic dysphagia developed in 37.3% of patients, and perforations occurred in two patients, suggesting a need for surgical back-up for patients undergoing endoscopic mucosal resection, the investigators said.High-grade dysplasia and intramucosal carcinoma each recurred in one patient, and both were treated successfully with endoscopic mucosal resection, they reported.
At the most recent analysis, 53 of 74 patients (71.6%) in a durability cohort were in complete remission from intestinal metaplasia, and all were in complete remission from high-grade dysplasia or intramucosal carcinoma, they noted.
Endoscopic therapy is now preferred over esophagectomy in most cases of Barrett’s esophagus with high-grade dysplasia or intramucosal carcinoma, and the current findings show that complete endoscopic mucosal resection is an effective and durable method to treat Barrett’s-associated neoplasia, the investigators concluded, adding that the results support the replacement of esophagectomy by endoscopic therapy for the condition.
“The most commonly encountered complication of esophageal stricture formation can be addressed with endoscopic dilation,” the investigators said.
However, long-term outcomes, particularly in those with advanced pathology who undergo endoscopic management, are still needed, as is an improved understanding of the behavior of the disease following treatment with specific modalities in order to optimize endoscopic therapy, they added.
This study was supported by the Francis L. Lederer Foundation. The authors reported having no disclosures.
Key clinical point: Endoscopic therapy should replace esophagectomy for Barrett’s-associated neoplasia.
Major finding: Barrett’s esophagus was completely eradicated in 86 of 107 patients (80%) on intention-to-treat analysis.
Data source: A series of 107 cases.
Disclosures: This study was supported by the Francis L. Lederer Foundation. The authors reported having no disclosures.
Large study shows no link between celiac disease and fertility problems
With the exception of those diagnosed between the age of 25 and 29 years, women with celiac disease are no more likely than are women without celiac disease to have fertility problems, according to findings from a large population-based study in the United Kingdom.
Of more than 2.4 million women with prospective primary care records available during their childbearing years (ages 15-49 years) between 1990 and 2013, 6,506 were diagnosed with celiac disease. The women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%), Nafeesa N. Dhalwani of the University of Nottingham and City Hospital Nottingham, U.K., and colleagues reported in the December issue of Gastroenterology (doi:10.1053/j.gastro.2014.08.025).
Source: American Gastroenterological Association
Further, the rates of infertility in those with celiac disease were similar to those without celiac disease both before and after diagnosis except in those aged 25-29 years at the time of diagnosis; the rates in those women were 41% higher, compared with those without celiac disease who were the same age (incidence rate ratio, 1.41), the investigators said.
“However, the absolute excess risk [for those diagnosed at age 25-29 years] was only 0.5% (5.2/1,000 person-years), they said.
Women included in the analysis were identified from the Health Improvement Network database. Rates of new clinically recorded fertility problems among those with and without diagnosed celiac disease were stratified by timing of celiac disease diagnosis after adjustment for sociodemographics, comorbidities, and calendar year.
The findings contrast with those from a number of smaller studies that demonstrated an association between infertility and celiac disease, but those studies included small numbers of women, including many who were receiving infertility specialist services, the investigators said, explaining that the women may not have been representative of the general population, and that other small studies found no link between celiac disease and fertility problems.
Celiac disease affects about 1% of the population in North America and Western Europe, and between 60% and 70% of those who are diagnosed are women. Several mechanisms through which celiac disease might affect a woman’s fertility have been described in the literature, but no conclusive evidence exists to support them, they noted.
Despite this lack of evidence and the inconsistent findings from small studies, a number of reviews include infertility as a key nongastrointestinal manifestation of celiac disease. The current findings suggest that most women with celiac disease – whether diagnosed or undiagnosed – do not have a substantially greater likelihood of having clinically recorded fertility problems than do those without celiac disease.
“Therefore, screening when women initially present with fertility problems may not identify a significant number of women with celiac disease, beyond the general population prevalence. This may not always apply to subgroups of women with severe celiac disease. However, in terms of the clinical burden of fertility problems at a population level, these findings should be reassuring for women with celiac disease and all stakeholders involved in their care,” the investigators concluded.
This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
With the exception of those diagnosed between the age of 25 and 29 years, women with celiac disease are no more likely than are women without celiac disease to have fertility problems, according to findings from a large population-based study in the United Kingdom.
Of more than 2.4 million women with prospective primary care records available during their childbearing years (ages 15-49 years) between 1990 and 2013, 6,506 were diagnosed with celiac disease. The women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%), Nafeesa N. Dhalwani of the University of Nottingham and City Hospital Nottingham, U.K., and colleagues reported in the December issue of Gastroenterology (doi:10.1053/j.gastro.2014.08.025).
Source: American Gastroenterological Association
Further, the rates of infertility in those with celiac disease were similar to those without celiac disease both before and after diagnosis except in those aged 25-29 years at the time of diagnosis; the rates in those women were 41% higher, compared with those without celiac disease who were the same age (incidence rate ratio, 1.41), the investigators said.
“However, the absolute excess risk [for those diagnosed at age 25-29 years] was only 0.5% (5.2/1,000 person-years), they said.
Women included in the analysis were identified from the Health Improvement Network database. Rates of new clinically recorded fertility problems among those with and without diagnosed celiac disease were stratified by timing of celiac disease diagnosis after adjustment for sociodemographics, comorbidities, and calendar year.
The findings contrast with those from a number of smaller studies that demonstrated an association between infertility and celiac disease, but those studies included small numbers of women, including many who were receiving infertility specialist services, the investigators said, explaining that the women may not have been representative of the general population, and that other small studies found no link between celiac disease and fertility problems.
Celiac disease affects about 1% of the population in North America and Western Europe, and between 60% and 70% of those who are diagnosed are women. Several mechanisms through which celiac disease might affect a woman’s fertility have been described in the literature, but no conclusive evidence exists to support them, they noted.
Despite this lack of evidence and the inconsistent findings from small studies, a number of reviews include infertility as a key nongastrointestinal manifestation of celiac disease. The current findings suggest that most women with celiac disease – whether diagnosed or undiagnosed – do not have a substantially greater likelihood of having clinically recorded fertility problems than do those without celiac disease.
“Therefore, screening when women initially present with fertility problems may not identify a significant number of women with celiac disease, beyond the general population prevalence. This may not always apply to subgroups of women with severe celiac disease. However, in terms of the clinical burden of fertility problems at a population level, these findings should be reassuring for women with celiac disease and all stakeholders involved in their care,” the investigators concluded.
This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
With the exception of those diagnosed between the age of 25 and 29 years, women with celiac disease are no more likely than are women without celiac disease to have fertility problems, according to findings from a large population-based study in the United Kingdom.
Of more than 2.4 million women with prospective primary care records available during their childbearing years (ages 15-49 years) between 1990 and 2013, 6,506 were diagnosed with celiac disease. The women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%), Nafeesa N. Dhalwani of the University of Nottingham and City Hospital Nottingham, U.K., and colleagues reported in the December issue of Gastroenterology (doi:10.1053/j.gastro.2014.08.025).
Source: American Gastroenterological Association
Further, the rates of infertility in those with celiac disease were similar to those without celiac disease both before and after diagnosis except in those aged 25-29 years at the time of diagnosis; the rates in those women were 41% higher, compared with those without celiac disease who were the same age (incidence rate ratio, 1.41), the investigators said.
“However, the absolute excess risk [for those diagnosed at age 25-29 years] was only 0.5% (5.2/1,000 person-years), they said.
Women included in the analysis were identified from the Health Improvement Network database. Rates of new clinically recorded fertility problems among those with and without diagnosed celiac disease were stratified by timing of celiac disease diagnosis after adjustment for sociodemographics, comorbidities, and calendar year.
The findings contrast with those from a number of smaller studies that demonstrated an association between infertility and celiac disease, but those studies included small numbers of women, including many who were receiving infertility specialist services, the investigators said, explaining that the women may not have been representative of the general population, and that other small studies found no link between celiac disease and fertility problems.
Celiac disease affects about 1% of the population in North America and Western Europe, and between 60% and 70% of those who are diagnosed are women. Several mechanisms through which celiac disease might affect a woman’s fertility have been described in the literature, but no conclusive evidence exists to support them, they noted.
Despite this lack of evidence and the inconsistent findings from small studies, a number of reviews include infertility as a key nongastrointestinal manifestation of celiac disease. The current findings suggest that most women with celiac disease – whether diagnosed or undiagnosed – do not have a substantially greater likelihood of having clinically recorded fertility problems than do those without celiac disease.
“Therefore, screening when women initially present with fertility problems may not identify a significant number of women with celiac disease, beyond the general population prevalence. This may not always apply to subgroups of women with severe celiac disease. However, in terms of the clinical burden of fertility problems at a population level, these findings should be reassuring for women with celiac disease and all stakeholders involved in their care,” the investigators concluded.
This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
Key clinical point: Women with celiac disease are not at increased risk of fertility problems.
Major finding: Women with celiac disease had a similar rate of recorded fertility problems as did those without celiac disease (4.4% vs. 4.1%).
Data source: A population-based study of more than 2.4 million women.
Disclosures: This study was supported by CORE/Coeliac UK, and by a University of Nottingham/Nottingham University Hospitals National Health Service Trust Senior Clinical Research Fellowship. The authors reported having no disclosures.
Liraglutide lowers HbA1c in diabetic CKD patients
PHILADELPHIA – Once-daily add-on liraglutide reduces HbA1c in patients with type 2 diabetes and moderate renal impairment when compared with placebo, according to findings from the phase III LIRA-RENAL trial.
The effects on blood glucose occurred across glomerular filtration rate (GFR) subgroups, and no worsening of kidney function occurred during the randomized, double-blind, 26-week study, Dr. David Scott reported at Kidney Week 2014.
In 277 patients with stage 3 chronic kidney disease (CKD) who were randomized to receive either an oral daily dose of 1.8 mg of liraglutide or placebo in addition to existing oral antidiabetic agents and/or insulin therapy, active treatment was associated with a significantly greater overall reduction in mean HbA1c from baseline to week 26 (–1.05% vs. –0.38%; estimated treatment difference, –0.66%). Similar reductions were seen in those with stage 3a disease (–1.10% vs. –0.35%; estimated treatment difference, –0.72%), and stage 3b disease (–0.97% vs. –0.40%; estimated treatment difference, –0.57%), Dr. Scott of Clinical Research Development Associates, Springfield Gardens, N.Y., reported at the meeting, which was sponsored by the American Society of Nephrology.
No significant treatment effect was observed on urinary albumin/creatinine ratio in any of the albuminuria subgroups. The estimated treatment ratios (liraglutide/placebo) were 1.09, 0.73, and 0.66 in those with normal (less than 30 mg/g), moderately increased (30-300 mg/g) or severely increased (greater than 300 mg/g) albuminuria levels.
Also, no difference was seen in estimated GFR change from baseline between the liraglutide and placebo groups (–1% vs. +1%, respectively), Dr. Scott said.
Adverse events and serious adverse events occurred in similar numbers of patients in the treatment and placebo groups (76% and 69%, and 10% and 11% of liraglutide and placebo patients, respectively), although those in the treatment group experienced more nausea and vomiting than did those in the placebo group (35.7% vs. 17.5%), he noted.
Confirmed hypoglycemia occurred in 11% and 17% of the liraglutide and placebo patients, respectively, but no real difference was seen between the groups with respect to severe hypoglycemia, he said.
Subjects were adults with body mass index of 20-45 kg/m2 and HbA1c of 7.0%-10.0%. All were on stable diabetes medication and had moderate renal impairment (estimated GFR of 30-59 mL/min/1.73 m2; modification of diet in renal disease).
“I think this is a very important study. It’s important, because as a clinical nephrologist, I’m faced day-to-day with the challenges of how to treat my diabetic patients with CKD. There are therapies that are contraindicated, there are therapies that are not well tolerated,” Dr. Scott said, explaining that treatment options remain limited for patients with diabetes and impaired renal function.
Further, while the incidence of diabetic nephropathy and diabetes has been declining, diabetes remains one of the most prevalent causes of CKD, and the incidence in some populations, including the elderly – who are at greatest risk for complications from therapy – has increased.
Although prior studies demonstrated that liraglutide was safe and well tolerated in patients with mild renal insufficiency, guidelines continue to include specific language suggesting that it is not indicated in the setting of CKD, Dr. Scott said.
The current findings provide additional evidence of the safety and efficacy of liraglutide in patients with type 2 diabetes and moderate renal impairment, he concluded.
The LIRA-RENAL trial was funded by Novo Nordisk. Dr. Scott reported having no disclosures.
PHILADELPHIA – Once-daily add-on liraglutide reduces HbA1c in patients with type 2 diabetes and moderate renal impairment when compared with placebo, according to findings from the phase III LIRA-RENAL trial.
The effects on blood glucose occurred across glomerular filtration rate (GFR) subgroups, and no worsening of kidney function occurred during the randomized, double-blind, 26-week study, Dr. David Scott reported at Kidney Week 2014.
In 277 patients with stage 3 chronic kidney disease (CKD) who were randomized to receive either an oral daily dose of 1.8 mg of liraglutide or placebo in addition to existing oral antidiabetic agents and/or insulin therapy, active treatment was associated with a significantly greater overall reduction in mean HbA1c from baseline to week 26 (–1.05% vs. –0.38%; estimated treatment difference, –0.66%). Similar reductions were seen in those with stage 3a disease (–1.10% vs. –0.35%; estimated treatment difference, –0.72%), and stage 3b disease (–0.97% vs. –0.40%; estimated treatment difference, –0.57%), Dr. Scott of Clinical Research Development Associates, Springfield Gardens, N.Y., reported at the meeting, which was sponsored by the American Society of Nephrology.
No significant treatment effect was observed on urinary albumin/creatinine ratio in any of the albuminuria subgroups. The estimated treatment ratios (liraglutide/placebo) were 1.09, 0.73, and 0.66 in those with normal (less than 30 mg/g), moderately increased (30-300 mg/g) or severely increased (greater than 300 mg/g) albuminuria levels.
Also, no difference was seen in estimated GFR change from baseline between the liraglutide and placebo groups (–1% vs. +1%, respectively), Dr. Scott said.
Adverse events and serious adverse events occurred in similar numbers of patients in the treatment and placebo groups (76% and 69%, and 10% and 11% of liraglutide and placebo patients, respectively), although those in the treatment group experienced more nausea and vomiting than did those in the placebo group (35.7% vs. 17.5%), he noted.
Confirmed hypoglycemia occurred in 11% and 17% of the liraglutide and placebo patients, respectively, but no real difference was seen between the groups with respect to severe hypoglycemia, he said.
Subjects were adults with body mass index of 20-45 kg/m2 and HbA1c of 7.0%-10.0%. All were on stable diabetes medication and had moderate renal impairment (estimated GFR of 30-59 mL/min/1.73 m2; modification of diet in renal disease).
“I think this is a very important study. It’s important, because as a clinical nephrologist, I’m faced day-to-day with the challenges of how to treat my diabetic patients with CKD. There are therapies that are contraindicated, there are therapies that are not well tolerated,” Dr. Scott said, explaining that treatment options remain limited for patients with diabetes and impaired renal function.
Further, while the incidence of diabetic nephropathy and diabetes has been declining, diabetes remains one of the most prevalent causes of CKD, and the incidence in some populations, including the elderly – who are at greatest risk for complications from therapy – has increased.
Although prior studies demonstrated that liraglutide was safe and well tolerated in patients with mild renal insufficiency, guidelines continue to include specific language suggesting that it is not indicated in the setting of CKD, Dr. Scott said.
The current findings provide additional evidence of the safety and efficacy of liraglutide in patients with type 2 diabetes and moderate renal impairment, he concluded.
The LIRA-RENAL trial was funded by Novo Nordisk. Dr. Scott reported having no disclosures.
PHILADELPHIA – Once-daily add-on liraglutide reduces HbA1c in patients with type 2 diabetes and moderate renal impairment when compared with placebo, according to findings from the phase III LIRA-RENAL trial.
The effects on blood glucose occurred across glomerular filtration rate (GFR) subgroups, and no worsening of kidney function occurred during the randomized, double-blind, 26-week study, Dr. David Scott reported at Kidney Week 2014.
In 277 patients with stage 3 chronic kidney disease (CKD) who were randomized to receive either an oral daily dose of 1.8 mg of liraglutide or placebo in addition to existing oral antidiabetic agents and/or insulin therapy, active treatment was associated with a significantly greater overall reduction in mean HbA1c from baseline to week 26 (–1.05% vs. –0.38%; estimated treatment difference, –0.66%). Similar reductions were seen in those with stage 3a disease (–1.10% vs. –0.35%; estimated treatment difference, –0.72%), and stage 3b disease (–0.97% vs. –0.40%; estimated treatment difference, –0.57%), Dr. Scott of Clinical Research Development Associates, Springfield Gardens, N.Y., reported at the meeting, which was sponsored by the American Society of Nephrology.
No significant treatment effect was observed on urinary albumin/creatinine ratio in any of the albuminuria subgroups. The estimated treatment ratios (liraglutide/placebo) were 1.09, 0.73, and 0.66 in those with normal (less than 30 mg/g), moderately increased (30-300 mg/g) or severely increased (greater than 300 mg/g) albuminuria levels.
Also, no difference was seen in estimated GFR change from baseline between the liraglutide and placebo groups (–1% vs. +1%, respectively), Dr. Scott said.
Adverse events and serious adverse events occurred in similar numbers of patients in the treatment and placebo groups (76% and 69%, and 10% and 11% of liraglutide and placebo patients, respectively), although those in the treatment group experienced more nausea and vomiting than did those in the placebo group (35.7% vs. 17.5%), he noted.
Confirmed hypoglycemia occurred in 11% and 17% of the liraglutide and placebo patients, respectively, but no real difference was seen between the groups with respect to severe hypoglycemia, he said.
Subjects were adults with body mass index of 20-45 kg/m2 and HbA1c of 7.0%-10.0%. All were on stable diabetes medication and had moderate renal impairment (estimated GFR of 30-59 mL/min/1.73 m2; modification of diet in renal disease).
“I think this is a very important study. It’s important, because as a clinical nephrologist, I’m faced day-to-day with the challenges of how to treat my diabetic patients with CKD. There are therapies that are contraindicated, there are therapies that are not well tolerated,” Dr. Scott said, explaining that treatment options remain limited for patients with diabetes and impaired renal function.
Further, while the incidence of diabetic nephropathy and diabetes has been declining, diabetes remains one of the most prevalent causes of CKD, and the incidence in some populations, including the elderly – who are at greatest risk for complications from therapy – has increased.
Although prior studies demonstrated that liraglutide was safe and well tolerated in patients with mild renal insufficiency, guidelines continue to include specific language suggesting that it is not indicated in the setting of CKD, Dr. Scott said.
The current findings provide additional evidence of the safety and efficacy of liraglutide in patients with type 2 diabetes and moderate renal impairment, he concluded.
The LIRA-RENAL trial was funded by Novo Nordisk. Dr. Scott reported having no disclosures.
Key clinical point: Liraglutide is safe and effective as add-on therapy for lowering blood glucose in patients with type 2 diabetes and CKD.
Major finding: Mean reductions in HbA1c with liraglutide and placebo were –1.05% and –0.38%, respectively.
Data source: The phase III LIRA-RENAL trial in 277 patients.
Disclosures: The LIRA-RENAL trial was funded by Novo Nordisk. Dr. Scott reported having no disclosures.
ZS-9 promotes normokalemia in patients with diabetes and CKD
PHILADELPHIA – ZS-9, an investigational cation exchanger designed to trap potassium in the gut, effectively and rapidly restores and maintains normokalemia in hyperkalemic patients with diabetes and significant renal impairment, according to a subgroup analysis of data from a randomized, placebo-controlled phase III trial.
Treatment with the first-in-class insoluble, nonabsorbed zirconium silicate, which was designed and engineered to have a three-dimensional crystalline lattice structure that preferentially traps potassium ions, was shown to have clinically and statistically significant benefit at daily oral doses of 5 mg and 10 mg, compared with placebo in the overall study population of 753 patients with hyperkalemia and underlying diabetes, heart failure, or chronic kidney disease.
The current analysis included 366 patients who had diabetes and an estimated glomerular filtration rate less than 60 mL/min/1.73m2. The patients, who had baseline potassium levels of 5.0-6.5 mEq/L (mean of 5.3 mEq/L) were randomized to oral ZS-9 at doses of 1.25, 2.5, 5, or 10 g or placebo given three times daily for the first 48 hours. Following this acute phase, those with a potassium level of 3.5-5.0 mEq/L were rerandomized to either the same ZS-9 acute-phase dose once daily for days 3-15 or placebo.
In the subgroup, ZS-9 treatment was associated with a significant decrease in potassium at 48 hours (decreases of -0.45, -0.59, and -0.81 mEq/L in the 2.5-, 5-, and 10-g groups, respectively, compared with +0.24 mEq/L in the placebo group, Dr. Bhupinder Singh reported at the meeting sponsored by the American Society of Nephrology.
During the maintenance phase, patients receiving a 10-g dose of ZS-9 maintained normokalemia (4.5 mEq/L at day 3 vs. 4.6 mEq/L at day 15) vs. placebo (4.4 mEq/L at day 3, vs. 5.1 mEq/L at day 15). Similar results were seen in the 5-mg treatment group, Dr. Singh of Apex Research, Riverside, Calif. said.
“Hyperkalemia is a common and serious complication in patients with diabetes who have chronic kidney disease, particularly those who are treated with renin-angiotensin-aldosterone system (RAAS) inhibitors. Even mild to moderate hyperkalemia has been recently shown to be associated with increased mortality,” Dr. Singh said, noting that concerns about the seriousness of hyperkalemia have likely led to under-prescription of RAAS inhibitors, which have been proven to have cardio- and renoprotective action, but which are associated with hyperkalemia.
In head-to-head in vitro studies, ZS-9 has been shown to be more than 125 times more selective than sodium polystyrene sulfonate (SPS) – the current mainstay of treatment for hyperkalemia), and to have more than 9 times the potassium-binding capacity.
In the current analysis, ZS-9 was associated with a “fairly acute” effect, reducing potassium levels significantly within 1 hour, and to less than 5 mEq/L within 4 hours. The study also was published Nov. 21 in the New England Journal of Medicine (doi:10.1056/NEJMoa1411487).
In both the overall study population and the subgroup with diabetes and chronic kidney disease, ZS-9 discontinuation at the end of the study was associated with recurrence of hyperkalemia, indicating a need for continued use. The adverse event profile was similar in the treatment and placebo groups in both studies.
The findings suggest that long-term treatment with ZS-9 may allow continuation of chronic RAAS inhibition in patients with diabetes and chronic kidney disease, Dr. Sing said, adding that long-term treatment studies are underway.
This study was funded by ZS Pharma Inc., which is developing ZS-9. Dr. Singh is a consultant to ZS Pharma Inc.
PHILADELPHIA – ZS-9, an investigational cation exchanger designed to trap potassium in the gut, effectively and rapidly restores and maintains normokalemia in hyperkalemic patients with diabetes and significant renal impairment, according to a subgroup analysis of data from a randomized, placebo-controlled phase III trial.
Treatment with the first-in-class insoluble, nonabsorbed zirconium silicate, which was designed and engineered to have a three-dimensional crystalline lattice structure that preferentially traps potassium ions, was shown to have clinically and statistically significant benefit at daily oral doses of 5 mg and 10 mg, compared with placebo in the overall study population of 753 patients with hyperkalemia and underlying diabetes, heart failure, or chronic kidney disease.
The current analysis included 366 patients who had diabetes and an estimated glomerular filtration rate less than 60 mL/min/1.73m2. The patients, who had baseline potassium levels of 5.0-6.5 mEq/L (mean of 5.3 mEq/L) were randomized to oral ZS-9 at doses of 1.25, 2.5, 5, or 10 g or placebo given three times daily for the first 48 hours. Following this acute phase, those with a potassium level of 3.5-5.0 mEq/L were rerandomized to either the same ZS-9 acute-phase dose once daily for days 3-15 or placebo.
In the subgroup, ZS-9 treatment was associated with a significant decrease in potassium at 48 hours (decreases of -0.45, -0.59, and -0.81 mEq/L in the 2.5-, 5-, and 10-g groups, respectively, compared with +0.24 mEq/L in the placebo group, Dr. Bhupinder Singh reported at the meeting sponsored by the American Society of Nephrology.
During the maintenance phase, patients receiving a 10-g dose of ZS-9 maintained normokalemia (4.5 mEq/L at day 3 vs. 4.6 mEq/L at day 15) vs. placebo (4.4 mEq/L at day 3, vs. 5.1 mEq/L at day 15). Similar results were seen in the 5-mg treatment group, Dr. Singh of Apex Research, Riverside, Calif. said.
“Hyperkalemia is a common and serious complication in patients with diabetes who have chronic kidney disease, particularly those who are treated with renin-angiotensin-aldosterone system (RAAS) inhibitors. Even mild to moderate hyperkalemia has been recently shown to be associated with increased mortality,” Dr. Singh said, noting that concerns about the seriousness of hyperkalemia have likely led to under-prescription of RAAS inhibitors, which have been proven to have cardio- and renoprotective action, but which are associated with hyperkalemia.
In head-to-head in vitro studies, ZS-9 has been shown to be more than 125 times more selective than sodium polystyrene sulfonate (SPS) – the current mainstay of treatment for hyperkalemia), and to have more than 9 times the potassium-binding capacity.
In the current analysis, ZS-9 was associated with a “fairly acute” effect, reducing potassium levels significantly within 1 hour, and to less than 5 mEq/L within 4 hours. The study also was published Nov. 21 in the New England Journal of Medicine (doi:10.1056/NEJMoa1411487).
In both the overall study population and the subgroup with diabetes and chronic kidney disease, ZS-9 discontinuation at the end of the study was associated with recurrence of hyperkalemia, indicating a need for continued use. The adverse event profile was similar in the treatment and placebo groups in both studies.
The findings suggest that long-term treatment with ZS-9 may allow continuation of chronic RAAS inhibition in patients with diabetes and chronic kidney disease, Dr. Sing said, adding that long-term treatment studies are underway.
This study was funded by ZS Pharma Inc., which is developing ZS-9. Dr. Singh is a consultant to ZS Pharma Inc.
PHILADELPHIA – ZS-9, an investigational cation exchanger designed to trap potassium in the gut, effectively and rapidly restores and maintains normokalemia in hyperkalemic patients with diabetes and significant renal impairment, according to a subgroup analysis of data from a randomized, placebo-controlled phase III trial.
Treatment with the first-in-class insoluble, nonabsorbed zirconium silicate, which was designed and engineered to have a three-dimensional crystalline lattice structure that preferentially traps potassium ions, was shown to have clinically and statistically significant benefit at daily oral doses of 5 mg and 10 mg, compared with placebo in the overall study population of 753 patients with hyperkalemia and underlying diabetes, heart failure, or chronic kidney disease.
The current analysis included 366 patients who had diabetes and an estimated glomerular filtration rate less than 60 mL/min/1.73m2. The patients, who had baseline potassium levels of 5.0-6.5 mEq/L (mean of 5.3 mEq/L) were randomized to oral ZS-9 at doses of 1.25, 2.5, 5, or 10 g or placebo given three times daily for the first 48 hours. Following this acute phase, those with a potassium level of 3.5-5.0 mEq/L were rerandomized to either the same ZS-9 acute-phase dose once daily for days 3-15 or placebo.
In the subgroup, ZS-9 treatment was associated with a significant decrease in potassium at 48 hours (decreases of -0.45, -0.59, and -0.81 mEq/L in the 2.5-, 5-, and 10-g groups, respectively, compared with +0.24 mEq/L in the placebo group, Dr. Bhupinder Singh reported at the meeting sponsored by the American Society of Nephrology.
During the maintenance phase, patients receiving a 10-g dose of ZS-9 maintained normokalemia (4.5 mEq/L at day 3 vs. 4.6 mEq/L at day 15) vs. placebo (4.4 mEq/L at day 3, vs. 5.1 mEq/L at day 15). Similar results were seen in the 5-mg treatment group, Dr. Singh of Apex Research, Riverside, Calif. said.
“Hyperkalemia is a common and serious complication in patients with diabetes who have chronic kidney disease, particularly those who are treated with renin-angiotensin-aldosterone system (RAAS) inhibitors. Even mild to moderate hyperkalemia has been recently shown to be associated with increased mortality,” Dr. Singh said, noting that concerns about the seriousness of hyperkalemia have likely led to under-prescription of RAAS inhibitors, which have been proven to have cardio- and renoprotective action, but which are associated with hyperkalemia.
In head-to-head in vitro studies, ZS-9 has been shown to be more than 125 times more selective than sodium polystyrene sulfonate (SPS) – the current mainstay of treatment for hyperkalemia), and to have more than 9 times the potassium-binding capacity.
In the current analysis, ZS-9 was associated with a “fairly acute” effect, reducing potassium levels significantly within 1 hour, and to less than 5 mEq/L within 4 hours. The study also was published Nov. 21 in the New England Journal of Medicine (doi:10.1056/NEJMoa1411487).
In both the overall study population and the subgroup with diabetes and chronic kidney disease, ZS-9 discontinuation at the end of the study was associated with recurrence of hyperkalemia, indicating a need for continued use. The adverse event profile was similar in the treatment and placebo groups in both studies.
The findings suggest that long-term treatment with ZS-9 may allow continuation of chronic RAAS inhibition in patients with diabetes and chronic kidney disease, Dr. Sing said, adding that long-term treatment studies are underway.
This study was funded by ZS Pharma Inc., which is developing ZS-9. Dr. Singh is a consultant to ZS Pharma Inc.
Key clinical point: ZS-9 may facilitate safe administration of RAAS inhibition in patients with diabetes and significant renal impairment.
Major finding: A daily 10-g dose of ZS-9 maintained normokalemia (4.5 mEq/L at day 3 vs. 4.6 mEq/L at day 15) vs. placebo (4.4 mEq/L at day 3, vs. 5.1 mEq/L at day 15).
Data source: A subgroup analysis of data from 366 patients in a randomized, placebo-controlled phase III trial.
Disclosures: This study was funded by ZS Pharma Inc., which is developing ZS-9. Dr. Singh is a consultant to ZS Pharma Inc.
Dialysis device removed Ebola from patient’s blood
PHILADELPHIA – An experimental dialysis device safely and dramatically reduced the viral load in a critically ill Ebola patient treated in Frankfurt, Germany.
Dialysis performed using the device resulted in a reduction in viral load from 400,000 copies/mL to 1,000 copies/mL in the 36-year-old patient, said Dr. Helmut Geiger, chief of nephrology at Frankfurt University Hospital, at Kidney Week 2014.
The patient, a pediatrician who had treated Ebola patients in Sierra Leone, was diagnosed in September. He was transferred in October to Frankfurt University Hospital, where he presented with fever, chills, and weakness, and rapidly deteriorated. He developed multiorgan failure – including kidney failure – despite treatment with various experimental therapies.
The patient was placed on dialysis. The experimental device – the Hemopurifier, made by Aethlon Medical of San Diego – was incorporated into the treatment, with special approval from Germany’s Federal Institute for Drugs and Medical Devices, Dr. Geiger said at the meeting, which was sponsored by the American Society of Nephrology.
The Hemopurifier is a first-in-class biofiltration device designed to eliminate viruses and immunosuppressive proteins from the circulatory system of infected individuals, according to Aethlon. The company has been testing the device in patients with hepatitis C and in HIV patients in India. Aethlon plans to initiate U.S. clinical studies under an investigational device exemption approved by the U.S. Food and Drug Administration.
Successful in vitro validation studies have been conducted by researchers at the U.S. Army Medical Research Institute of Infectious Diseases and the Centers for Disease Control and Prevention.
The Hemopurifier is a cylindrical cartridge that attaches to an existing dialysis machine. Inside, a gluelike protein selectively binds to viral particles and fragments, removing them from blood circulation.
The patient treated in Frankfurt by Dr. Geiger and his team underwent a 6.5-hour dialysis procedure using the device, and an analysis performed at a laboratory equipped to handle Ebola virus showed that the device had trapped 242 million copies of the virus, Dr. Geiger said.
The patient is now out of isolation, off dialysis, and in very good condition, he said, noting that the patient would be discharged within days.
Although additional study is needed, it appears that the Hemopurifier device may have contributed to the patient’s recovery, Dr. Geiger said, and no adverse events occurred during the treatment. The device ultimately could play a role in treating multiple types of viral infection, he added.
Use of the Hemopurifier is safe and feasible, the device can be used with intermittent hemodialysis or in the setting of continuous renal replacement therapy, and it represents a promising new supportive tool for severe Ebola infection, he concluded.
Dr. Geiger reported having consultancy agreements with AbbVie, Amgen, and Otsuka, and receiving research funding from Fresenius Medical Care.
PHILADELPHIA – An experimental dialysis device safely and dramatically reduced the viral load in a critically ill Ebola patient treated in Frankfurt, Germany.
Dialysis performed using the device resulted in a reduction in viral load from 400,000 copies/mL to 1,000 copies/mL in the 36-year-old patient, said Dr. Helmut Geiger, chief of nephrology at Frankfurt University Hospital, at Kidney Week 2014.
The patient, a pediatrician who had treated Ebola patients in Sierra Leone, was diagnosed in September. He was transferred in October to Frankfurt University Hospital, where he presented with fever, chills, and weakness, and rapidly deteriorated. He developed multiorgan failure – including kidney failure – despite treatment with various experimental therapies.
The patient was placed on dialysis. The experimental device – the Hemopurifier, made by Aethlon Medical of San Diego – was incorporated into the treatment, with special approval from Germany’s Federal Institute for Drugs and Medical Devices, Dr. Geiger said at the meeting, which was sponsored by the American Society of Nephrology.
The Hemopurifier is a first-in-class biofiltration device designed to eliminate viruses and immunosuppressive proteins from the circulatory system of infected individuals, according to Aethlon. The company has been testing the device in patients with hepatitis C and in HIV patients in India. Aethlon plans to initiate U.S. clinical studies under an investigational device exemption approved by the U.S. Food and Drug Administration.
Successful in vitro validation studies have been conducted by researchers at the U.S. Army Medical Research Institute of Infectious Diseases and the Centers for Disease Control and Prevention.
The Hemopurifier is a cylindrical cartridge that attaches to an existing dialysis machine. Inside, a gluelike protein selectively binds to viral particles and fragments, removing them from blood circulation.
The patient treated in Frankfurt by Dr. Geiger and his team underwent a 6.5-hour dialysis procedure using the device, and an analysis performed at a laboratory equipped to handle Ebola virus showed that the device had trapped 242 million copies of the virus, Dr. Geiger said.
The patient is now out of isolation, off dialysis, and in very good condition, he said, noting that the patient would be discharged within days.
Although additional study is needed, it appears that the Hemopurifier device may have contributed to the patient’s recovery, Dr. Geiger said, and no adverse events occurred during the treatment. The device ultimately could play a role in treating multiple types of viral infection, he added.
Use of the Hemopurifier is safe and feasible, the device can be used with intermittent hemodialysis or in the setting of continuous renal replacement therapy, and it represents a promising new supportive tool for severe Ebola infection, he concluded.
Dr. Geiger reported having consultancy agreements with AbbVie, Amgen, and Otsuka, and receiving research funding from Fresenius Medical Care.
PHILADELPHIA – An experimental dialysis device safely and dramatically reduced the viral load in a critically ill Ebola patient treated in Frankfurt, Germany.
Dialysis performed using the device resulted in a reduction in viral load from 400,000 copies/mL to 1,000 copies/mL in the 36-year-old patient, said Dr. Helmut Geiger, chief of nephrology at Frankfurt University Hospital, at Kidney Week 2014.
The patient, a pediatrician who had treated Ebola patients in Sierra Leone, was diagnosed in September. He was transferred in October to Frankfurt University Hospital, where he presented with fever, chills, and weakness, and rapidly deteriorated. He developed multiorgan failure – including kidney failure – despite treatment with various experimental therapies.
The patient was placed on dialysis. The experimental device – the Hemopurifier, made by Aethlon Medical of San Diego – was incorporated into the treatment, with special approval from Germany’s Federal Institute for Drugs and Medical Devices, Dr. Geiger said at the meeting, which was sponsored by the American Society of Nephrology.
The Hemopurifier is a first-in-class biofiltration device designed to eliminate viruses and immunosuppressive proteins from the circulatory system of infected individuals, according to Aethlon. The company has been testing the device in patients with hepatitis C and in HIV patients in India. Aethlon plans to initiate U.S. clinical studies under an investigational device exemption approved by the U.S. Food and Drug Administration.
Successful in vitro validation studies have been conducted by researchers at the U.S. Army Medical Research Institute of Infectious Diseases and the Centers for Disease Control and Prevention.
The Hemopurifier is a cylindrical cartridge that attaches to an existing dialysis machine. Inside, a gluelike protein selectively binds to viral particles and fragments, removing them from blood circulation.
The patient treated in Frankfurt by Dr. Geiger and his team underwent a 6.5-hour dialysis procedure using the device, and an analysis performed at a laboratory equipped to handle Ebola virus showed that the device had trapped 242 million copies of the virus, Dr. Geiger said.
The patient is now out of isolation, off dialysis, and in very good condition, he said, noting that the patient would be discharged within days.
Although additional study is needed, it appears that the Hemopurifier device may have contributed to the patient’s recovery, Dr. Geiger said, and no adverse events occurred during the treatment. The device ultimately could play a role in treating multiple types of viral infection, he added.
Use of the Hemopurifier is safe and feasible, the device can be used with intermittent hemodialysis or in the setting of continuous renal replacement therapy, and it represents a promising new supportive tool for severe Ebola infection, he concluded.
Dr. Geiger reported having consultancy agreements with AbbVie, Amgen, and Otsuka, and receiving research funding from Fresenius Medical Care.
FROM KIDNEY WEEK 2014
Key clinical point: An investigational dialysis device shows promise for supportive treatment of Ebola and other viral infections.
Major finding: The Hemopurifier dialysis device removed 242 million Ebola virus particles over 6.5 hours.
Data source: A single case report.
Disclosures: Dr. Geiger reported having consultancy agreements with AbbVie, Amgen, and Otsuka, and receiving research funding from Fresenius Medical Care.
Acid suppression results in more severe CAP, poorer outcomes
AUSTIN, TEX. – Use of acid-suppressing drugs was associated with more severe presentation, longer hospital stay, and increased mortality among patients with community-acquired pneumonia presenting to an inner city community hospital, according to a retrospective analysis.
Of 866 patients admitted with community-acquired pneumonia (CAP) between 2010 and 2013, 468 (54%) were on acid suppression, and those patients were significantly more likely than those not on acid suppression to present with positive blood cultures (12% vs. 5.5%) and thrombocytopenia (22% vs. 17%). The difference between the groups in length of stay (10.51 days vs. 8.96 days, respectively), and mortality (15.1% vs. 11.5%, respectively) trended toward significance, Dr. Bikash Bhattarai reported at the annual meeting of the American College of Chest Physicians.
Study subjects included 419 men and 447 women, most of whom (86%) were African American. About half (53%) were smokers. The study excluded patients with suspected aspiration pneumonia and those who were immunosuppressed.
The groups were similar with respect to demographic characteristics, but patients with comorbidities, including chronic obstructive pulmonary disease, diabetes, previous stroke, or neoplastic diseases, were more likely to be on acid suppression, said Dr. Bhattarai of the Interfaith Medical Center in Brooklyn, N.Y.
Gastrointestinal symptoms are very common in the general population – as is the use of either prescribed or over-the-counter medication for acid suppression to treat them, he said, adding that it has been shown that reduction of gastric acid secretion permits colonization of the upper gastrointestinal tract with oral bacteria and is therefore considered a risk factor for CAP.
The findings of this analysis are limited by the retrospective, observational nature of the study, and additional subgroup analyses – including an analysis based on whether patients in the acid suppression group were being treated with proton pump inhibitors or histamine2 blockers could be useful, he said, acknowledging that some data suggest that proton pump inhibitor use is associated with greater risk.
“Acid-suppressing drugs should be used with caution, and we strongly encourage using those medications only when indicated,” he said.
Dr. Bhattarai reported having no disclosures.
AUSTIN, TEX. – Use of acid-suppressing drugs was associated with more severe presentation, longer hospital stay, and increased mortality among patients with community-acquired pneumonia presenting to an inner city community hospital, according to a retrospective analysis.
Of 866 patients admitted with community-acquired pneumonia (CAP) between 2010 and 2013, 468 (54%) were on acid suppression, and those patients were significantly more likely than those not on acid suppression to present with positive blood cultures (12% vs. 5.5%) and thrombocytopenia (22% vs. 17%). The difference between the groups in length of stay (10.51 days vs. 8.96 days, respectively), and mortality (15.1% vs. 11.5%, respectively) trended toward significance, Dr. Bikash Bhattarai reported at the annual meeting of the American College of Chest Physicians.
Study subjects included 419 men and 447 women, most of whom (86%) were African American. About half (53%) were smokers. The study excluded patients with suspected aspiration pneumonia and those who were immunosuppressed.
The groups were similar with respect to demographic characteristics, but patients with comorbidities, including chronic obstructive pulmonary disease, diabetes, previous stroke, or neoplastic diseases, were more likely to be on acid suppression, said Dr. Bhattarai of the Interfaith Medical Center in Brooklyn, N.Y.
Gastrointestinal symptoms are very common in the general population – as is the use of either prescribed or over-the-counter medication for acid suppression to treat them, he said, adding that it has been shown that reduction of gastric acid secretion permits colonization of the upper gastrointestinal tract with oral bacteria and is therefore considered a risk factor for CAP.
The findings of this analysis are limited by the retrospective, observational nature of the study, and additional subgroup analyses – including an analysis based on whether patients in the acid suppression group were being treated with proton pump inhibitors or histamine2 blockers could be useful, he said, acknowledging that some data suggest that proton pump inhibitor use is associated with greater risk.
“Acid-suppressing drugs should be used with caution, and we strongly encourage using those medications only when indicated,” he said.
Dr. Bhattarai reported having no disclosures.
AUSTIN, TEX. – Use of acid-suppressing drugs was associated with more severe presentation, longer hospital stay, and increased mortality among patients with community-acquired pneumonia presenting to an inner city community hospital, according to a retrospective analysis.
Of 866 patients admitted with community-acquired pneumonia (CAP) between 2010 and 2013, 468 (54%) were on acid suppression, and those patients were significantly more likely than those not on acid suppression to present with positive blood cultures (12% vs. 5.5%) and thrombocytopenia (22% vs. 17%). The difference between the groups in length of stay (10.51 days vs. 8.96 days, respectively), and mortality (15.1% vs. 11.5%, respectively) trended toward significance, Dr. Bikash Bhattarai reported at the annual meeting of the American College of Chest Physicians.
Study subjects included 419 men and 447 women, most of whom (86%) were African American. About half (53%) were smokers. The study excluded patients with suspected aspiration pneumonia and those who were immunosuppressed.
The groups were similar with respect to demographic characteristics, but patients with comorbidities, including chronic obstructive pulmonary disease, diabetes, previous stroke, or neoplastic diseases, were more likely to be on acid suppression, said Dr. Bhattarai of the Interfaith Medical Center in Brooklyn, N.Y.
Gastrointestinal symptoms are very common in the general population – as is the use of either prescribed or over-the-counter medication for acid suppression to treat them, he said, adding that it has been shown that reduction of gastric acid secretion permits colonization of the upper gastrointestinal tract with oral bacteria and is therefore considered a risk factor for CAP.
The findings of this analysis are limited by the retrospective, observational nature of the study, and additional subgroup analyses – including an analysis based on whether patients in the acid suppression group were being treated with proton pump inhibitors or histamine2 blockers could be useful, he said, acknowledging that some data suggest that proton pump inhibitor use is associated with greater risk.
“Acid-suppressing drugs should be used with caution, and we strongly encourage using those medications only when indicated,” he said.
Dr. Bhattarai reported having no disclosures.
Key clinical point: Acid suppression worsens CAP outcomes and should be used only when indicated.
Major finding: Patients on acid suppression were more likely to have positive blood cultures (12% vs. 5.5%) and thrombocytopenia (22% vs. 17%).
Data source: A retrospective cohort study of 866 patients.
Disclosures: Dr. Bhattarai reported having no disclosures.
PPI, steroid use raises risk of CDAD recurrence
AUSTIN, TEX. – Use of proton pump inhibitors and steroids was independently associated with recurrences of Clostridium difficile–associated diarrhea among patients in an intensive care unit, based on a retrospective chart review reported at the annual meeting of the American College of Chest Physicians.
Recurrences were noted in 268 of 2,019 patients who were admitted to a single intensive care unit during a 6-year period and were initially treated successfully for C. difficile–associated diarrhea (CDAD). In a univariate analysis, recurrence was correlated with use of proton pump inhibitors (PPIs) and steroids, but not with age, male gender, or length of hospital stay. After adjusting for age, sex, length of stay, and treatment used, the relationships between recurrence and PPI and steroid use remained statistically significant (P = .0331 and P = .0305, respectively), Dr. Ala Nijim of Akron (Ohio) General Medical Center.
The study subjects comprised 798 men and 1,221 women with an average age of 68 years and an average hospital length of stay of 10 days. Severe disease was present in 233 patients, and 51 had cancer.
CDAD in this study was defined as at least three episodes of loose stools in less than 24 hours with a positive C. difficile toxin assay. Recurrence was defined as a second positive stool test within 90 days following complete resolution of a previous episode of diarrhea episode and cessation of treatment comprising a 10-day period.
Data suggest that the rate of CDAD recurrence is between 10% and 25% at a cost of between $3.2 and $4.8 billion, Dr. Nijim said.
Glucocorticoids are known risk factors for acquiring CDAD, likely due to their immunosuppressive effects, and PPIs have also been suggested as risk factors for acquiring CDAD, although the findings have been mixed. Likewise, treatment with metronidazole for an initial episode has been linked with treatment failure and recurrence risk. In the current study, one of the largest to date to evaluate factors associated with CDAD recurrence, both PPIs and glucocorticoids were shown to be associated with recurrence risk, but no link was found between metronidazole or any CDAD treatment modality and recurrence, Dr. Nijim said.
Though limited by the single-center, retrospective study design and its inherent information and selection biases, the study includes a large ICU patient sample, and the findings suggest that intensivists should watch carefully for recurrence in patients using PPIs and/or steroids.
Also, clinicians should think carefully before starting patients on PPIs – and perhaps use histamine2 blockers instead – in patients with a history of CDAD, she said.
Dr. Nijim reported having no disclosures.
AUSTIN, TEX. – Use of proton pump inhibitors and steroids was independently associated with recurrences of Clostridium difficile–associated diarrhea among patients in an intensive care unit, based on a retrospective chart review reported at the annual meeting of the American College of Chest Physicians.
Recurrences were noted in 268 of 2,019 patients who were admitted to a single intensive care unit during a 6-year period and were initially treated successfully for C. difficile–associated diarrhea (CDAD). In a univariate analysis, recurrence was correlated with use of proton pump inhibitors (PPIs) and steroids, but not with age, male gender, or length of hospital stay. After adjusting for age, sex, length of stay, and treatment used, the relationships between recurrence and PPI and steroid use remained statistically significant (P = .0331 and P = .0305, respectively), Dr. Ala Nijim of Akron (Ohio) General Medical Center.
The study subjects comprised 798 men and 1,221 women with an average age of 68 years and an average hospital length of stay of 10 days. Severe disease was present in 233 patients, and 51 had cancer.
CDAD in this study was defined as at least three episodes of loose stools in less than 24 hours with a positive C. difficile toxin assay. Recurrence was defined as a second positive stool test within 90 days following complete resolution of a previous episode of diarrhea episode and cessation of treatment comprising a 10-day period.
Data suggest that the rate of CDAD recurrence is between 10% and 25% at a cost of between $3.2 and $4.8 billion, Dr. Nijim said.
Glucocorticoids are known risk factors for acquiring CDAD, likely due to their immunosuppressive effects, and PPIs have also been suggested as risk factors for acquiring CDAD, although the findings have been mixed. Likewise, treatment with metronidazole for an initial episode has been linked with treatment failure and recurrence risk. In the current study, one of the largest to date to evaluate factors associated with CDAD recurrence, both PPIs and glucocorticoids were shown to be associated with recurrence risk, but no link was found between metronidazole or any CDAD treatment modality and recurrence, Dr. Nijim said.
Though limited by the single-center, retrospective study design and its inherent information and selection biases, the study includes a large ICU patient sample, and the findings suggest that intensivists should watch carefully for recurrence in patients using PPIs and/or steroids.
Also, clinicians should think carefully before starting patients on PPIs – and perhaps use histamine2 blockers instead – in patients with a history of CDAD, she said.
Dr. Nijim reported having no disclosures.
AUSTIN, TEX. – Use of proton pump inhibitors and steroids was independently associated with recurrences of Clostridium difficile–associated diarrhea among patients in an intensive care unit, based on a retrospective chart review reported at the annual meeting of the American College of Chest Physicians.
Recurrences were noted in 268 of 2,019 patients who were admitted to a single intensive care unit during a 6-year period and were initially treated successfully for C. difficile–associated diarrhea (CDAD). In a univariate analysis, recurrence was correlated with use of proton pump inhibitors (PPIs) and steroids, but not with age, male gender, or length of hospital stay. After adjusting for age, sex, length of stay, and treatment used, the relationships between recurrence and PPI and steroid use remained statistically significant (P = .0331 and P = .0305, respectively), Dr. Ala Nijim of Akron (Ohio) General Medical Center.
The study subjects comprised 798 men and 1,221 women with an average age of 68 years and an average hospital length of stay of 10 days. Severe disease was present in 233 patients, and 51 had cancer.
CDAD in this study was defined as at least three episodes of loose stools in less than 24 hours with a positive C. difficile toxin assay. Recurrence was defined as a second positive stool test within 90 days following complete resolution of a previous episode of diarrhea episode and cessation of treatment comprising a 10-day period.
Data suggest that the rate of CDAD recurrence is between 10% and 25% at a cost of between $3.2 and $4.8 billion, Dr. Nijim said.
Glucocorticoids are known risk factors for acquiring CDAD, likely due to their immunosuppressive effects, and PPIs have also been suggested as risk factors for acquiring CDAD, although the findings have been mixed. Likewise, treatment with metronidazole for an initial episode has been linked with treatment failure and recurrence risk. In the current study, one of the largest to date to evaluate factors associated with CDAD recurrence, both PPIs and glucocorticoids were shown to be associated with recurrence risk, but no link was found between metronidazole or any CDAD treatment modality and recurrence, Dr. Nijim said.
Though limited by the single-center, retrospective study design and its inherent information and selection biases, the study includes a large ICU patient sample, and the findings suggest that intensivists should watch carefully for recurrence in patients using PPIs and/or steroids.
Also, clinicians should think carefully before starting patients on PPIs – and perhaps use histamine2 blockers instead – in patients with a history of CDAD, she said.
Dr. Nijim reported having no disclosures.
Key clinical point: PPIs may be inadvisable in patients with a history of CDAD.
Major finding: CDAD recurrence was significantly associated with PPI and steroid use (P = .0331 and P = .0305, respectively).
Data source: A retrospective cohort study of 2,019 ICU patients.
Disclosures: Dr. Nijim reported having no disclosures.
Lisinopril monotherapy controls blood pressure as well as combo therapy in ADPKD
PHILADELPHIA – Dual blockade of the renin-angiotensin-aldosterone system with combined angiotensin-converting enzyme inhibitor and angiotensin receptor blocker therapy was no more effective for maintaining blood pressure control than was ACE inihibitor monotherapy in patients with moderately advanced autosomal dominant polycystic kidney disease and stage 3 chronic kidney disease.
That finding emerged from a study of the randomized placebo-controlled Halt Progression of Polycystic Kidney Disease (HALD-PKD) trial reported at Kidney Week 2014.
The composite primary outcome of time to death, end-stage renal disease, or 50% reduction from baseline in estimated glomerular filtration rate (eGFR) was similar in 242 patients with stages 1-3 autosomal dominant polcystic kidney disease (ADPKD) randomized to receive lisinopril plus placebo, and in 243 such patients randomized to receive lisinopril plus telmisartan (number of events in the groups, respectively, 116 and 115; hazard ratio, 1.08), said Dr. Vicente E. Torres.
Also, no significant differences were seen between the groups with respect to the individual components of the composite outcome; the hazard ratios for death, end-stage renal disease, and 50% reduction in eGFR were 0.93, 0.78, and 1.05, respectively, Dr. Torres of the Mayo Clinic College of Medicine, Rochester, Minn. said at the meeting, which was sponsored by the American Society of Nephrology.
The findings were simultaneously published online Nov. 15 in the New England Journal of Medicine (N Engl J Med 2014 Nov. 15[doi: 10.1056/NEJMoa1402686]).
The rates of change in eGFR urinary albumin excretion, and other secondary outcomes and adverse events, including hyperkalemia and acute kidney injury, also were similar in the two groups, Dr. Torres said.
Participants in the double-blind study were enrolled from February 2006 through June 2009. They were aged 18 to 64 years and had eGFR of 25 to 60 ml per minute per 1.73 m2 of body-surface area. Lisinopril doses in both groups were adjusted to achieve a blood pressure of 110/70 to 130/80 mm Hg, and patients were followed for a mean of 5 years. Systolic blood pressures and mean arterial pressures remained within the target range throughout the trial in 73% to 86% and in 70% to 83% of participants, respectively. Diastolic pressures were in the target range in 56% to 65% of participants.
Hypertension develops early and contributes to the progression of disease in patients with ADPKD, and the renin-angiotensin-aldosterone system (RAAS) is implicated in the pathogenesis of hypertension in ADPKD patients. Angiotensin-converting enzyme inhibitors like lisinopril are known to slow the progression of renal dysfunction in non-diabetic kidney disease, and thus have become standard first-line agents for the treatment of hypertension in patients with ADPKD, Dr. Torres said.
“Although ACE inhibitors have become the first-line therapy for hypertension in patients with chronic kidney disease, including ADPKD, their renoprotective effect may be limited by compensatory-feedback increases in renin release and the generation of angiotensin,” he and his colleagues wrote, noting that dual RAAS blockade has been proposed as a strategy to circumvent this compensatory feedback.
The current study showed that adding telmisartan did result in slightly lower blood pressures, but did not reduce the incidence of primary or secondary outcomes.
Both combination therapy and lisinopril monotherapy were safe, with a similar rate of adverse events in the two groups, but the addition of an ARB did not confer additional benefit, Dr. Torres said, concluding that ACE inhibitor monotherapy achieved excellent standard blood pressure control in more than 70% of patients with stages 1-3 ADPKD.The HALT-PKD trial was funded by grants from the National Institute of Diabetes and Digestive and Kidney Diseases (including a grant to Dr. Torres), the National Center for Research Resources’ General Clinical Research Centers, the National Center for Advancing Translational Sciences’ Clinical and Translational Science Awards, the Zell Family Foundation, and the PKD Foundation.
PHILADELPHIA – Dual blockade of the renin-angiotensin-aldosterone system with combined angiotensin-converting enzyme inhibitor and angiotensin receptor blocker therapy was no more effective for maintaining blood pressure control than was ACE inihibitor monotherapy in patients with moderately advanced autosomal dominant polycystic kidney disease and stage 3 chronic kidney disease.
That finding emerged from a study of the randomized placebo-controlled Halt Progression of Polycystic Kidney Disease (HALD-PKD) trial reported at Kidney Week 2014.
The composite primary outcome of time to death, end-stage renal disease, or 50% reduction from baseline in estimated glomerular filtration rate (eGFR) was similar in 242 patients with stages 1-3 autosomal dominant polcystic kidney disease (ADPKD) randomized to receive lisinopril plus placebo, and in 243 such patients randomized to receive lisinopril plus telmisartan (number of events in the groups, respectively, 116 and 115; hazard ratio, 1.08), said Dr. Vicente E. Torres.
Also, no significant differences were seen between the groups with respect to the individual components of the composite outcome; the hazard ratios for death, end-stage renal disease, and 50% reduction in eGFR were 0.93, 0.78, and 1.05, respectively, Dr. Torres of the Mayo Clinic College of Medicine, Rochester, Minn. said at the meeting, which was sponsored by the American Society of Nephrology.
The findings were simultaneously published online Nov. 15 in the New England Journal of Medicine (N Engl J Med 2014 Nov. 15[doi: 10.1056/NEJMoa1402686]).
The rates of change in eGFR urinary albumin excretion, and other secondary outcomes and adverse events, including hyperkalemia and acute kidney injury, also were similar in the two groups, Dr. Torres said.
Participants in the double-blind study were enrolled from February 2006 through June 2009. They were aged 18 to 64 years and had eGFR of 25 to 60 ml per minute per 1.73 m2 of body-surface area. Lisinopril doses in both groups were adjusted to achieve a blood pressure of 110/70 to 130/80 mm Hg, and patients were followed for a mean of 5 years. Systolic blood pressures and mean arterial pressures remained within the target range throughout the trial in 73% to 86% and in 70% to 83% of participants, respectively. Diastolic pressures were in the target range in 56% to 65% of participants.
Hypertension develops early and contributes to the progression of disease in patients with ADPKD, and the renin-angiotensin-aldosterone system (RAAS) is implicated in the pathogenesis of hypertension in ADPKD patients. Angiotensin-converting enzyme inhibitors like lisinopril are known to slow the progression of renal dysfunction in non-diabetic kidney disease, and thus have become standard first-line agents for the treatment of hypertension in patients with ADPKD, Dr. Torres said.
“Although ACE inhibitors have become the first-line therapy for hypertension in patients with chronic kidney disease, including ADPKD, their renoprotective effect may be limited by compensatory-feedback increases in renin release and the generation of angiotensin,” he and his colleagues wrote, noting that dual RAAS blockade has been proposed as a strategy to circumvent this compensatory feedback.
The current study showed that adding telmisartan did result in slightly lower blood pressures, but did not reduce the incidence of primary or secondary outcomes.
Both combination therapy and lisinopril monotherapy were safe, with a similar rate of adverse events in the two groups, but the addition of an ARB did not confer additional benefit, Dr. Torres said, concluding that ACE inhibitor monotherapy achieved excellent standard blood pressure control in more than 70% of patients with stages 1-3 ADPKD.The HALT-PKD trial was funded by grants from the National Institute of Diabetes and Digestive and Kidney Diseases (including a grant to Dr. Torres), the National Center for Research Resources’ General Clinical Research Centers, the National Center for Advancing Translational Sciences’ Clinical and Translational Science Awards, the Zell Family Foundation, and the PKD Foundation.
PHILADELPHIA – Dual blockade of the renin-angiotensin-aldosterone system with combined angiotensin-converting enzyme inhibitor and angiotensin receptor blocker therapy was no more effective for maintaining blood pressure control than was ACE inihibitor monotherapy in patients with moderately advanced autosomal dominant polycystic kidney disease and stage 3 chronic kidney disease.
That finding emerged from a study of the randomized placebo-controlled Halt Progression of Polycystic Kidney Disease (HALD-PKD) trial reported at Kidney Week 2014.
The composite primary outcome of time to death, end-stage renal disease, or 50% reduction from baseline in estimated glomerular filtration rate (eGFR) was similar in 242 patients with stages 1-3 autosomal dominant polcystic kidney disease (ADPKD) randomized to receive lisinopril plus placebo, and in 243 such patients randomized to receive lisinopril plus telmisartan (number of events in the groups, respectively, 116 and 115; hazard ratio, 1.08), said Dr. Vicente E. Torres.
Also, no significant differences were seen between the groups with respect to the individual components of the composite outcome; the hazard ratios for death, end-stage renal disease, and 50% reduction in eGFR were 0.93, 0.78, and 1.05, respectively, Dr. Torres of the Mayo Clinic College of Medicine, Rochester, Minn. said at the meeting, which was sponsored by the American Society of Nephrology.
The findings were simultaneously published online Nov. 15 in the New England Journal of Medicine (N Engl J Med 2014 Nov. 15[doi: 10.1056/NEJMoa1402686]).
The rates of change in eGFR urinary albumin excretion, and other secondary outcomes and adverse events, including hyperkalemia and acute kidney injury, also were similar in the two groups, Dr. Torres said.
Participants in the double-blind study were enrolled from February 2006 through June 2009. They were aged 18 to 64 years and had eGFR of 25 to 60 ml per minute per 1.73 m2 of body-surface area. Lisinopril doses in both groups were adjusted to achieve a blood pressure of 110/70 to 130/80 mm Hg, and patients were followed for a mean of 5 years. Systolic blood pressures and mean arterial pressures remained within the target range throughout the trial in 73% to 86% and in 70% to 83% of participants, respectively. Diastolic pressures were in the target range in 56% to 65% of participants.
Hypertension develops early and contributes to the progression of disease in patients with ADPKD, and the renin-angiotensin-aldosterone system (RAAS) is implicated in the pathogenesis of hypertension in ADPKD patients. Angiotensin-converting enzyme inhibitors like lisinopril are known to slow the progression of renal dysfunction in non-diabetic kidney disease, and thus have become standard first-line agents for the treatment of hypertension in patients with ADPKD, Dr. Torres said.
“Although ACE inhibitors have become the first-line therapy for hypertension in patients with chronic kidney disease, including ADPKD, their renoprotective effect may be limited by compensatory-feedback increases in renin release and the generation of angiotensin,” he and his colleagues wrote, noting that dual RAAS blockade has been proposed as a strategy to circumvent this compensatory feedback.
The current study showed that adding telmisartan did result in slightly lower blood pressures, but did not reduce the incidence of primary or secondary outcomes.
Both combination therapy and lisinopril monotherapy were safe, with a similar rate of adverse events in the two groups, but the addition of an ARB did not confer additional benefit, Dr. Torres said, concluding that ACE inhibitor monotherapy achieved excellent standard blood pressure control in more than 70% of patients with stages 1-3 ADPKD.The HALT-PKD trial was funded by grants from the National Institute of Diabetes and Digestive and Kidney Diseases (including a grant to Dr. Torres), the National Center for Research Resources’ General Clinical Research Centers, the National Center for Advancing Translational Sciences’ Clinical and Translational Science Awards, the Zell Family Foundation, and the PKD Foundation.
Key clinical point: Dual RAAS blockade is no better than lisinopril monotherapy in patients with moderately advanced ADPKD.
Major finding: The composite primary endpoint was similar in the combination and monotherapy groups (hazard ratio, 1.08).
Data source: The randomized, double-blind, placebo-controlled HALT-PKD trial, study involving 485 patients.
Disclosures: The HALT-PKD trial was funded by grants from the National Institute of Diabetes and Digestive and Kidney Diseases (including a grant to Dr. Torres), the National Center for Research Resources’ General Clinical Research Centers, the National Center for Advancing Translational Sciences’ Clinical and Translational Science Awards, the Zell Family Foundation, and the PKD Foundation.