User login
A Pharmacist-Led Process to Monitor Discrepant Urine Drug Screen Results
Urine drug screen (UDS) monitoring is a common risk-mitigation strategy tool for prescribing controlled substances.1-3 Not only is UDS monitoring highlighted by clinical practice guidelines for opioid prescribing for chronic pain,1,2 it has also been suggested as best practice for benzodiazepines3 and a consideration for other controlled substances. Monitoring UDSs helps confirm adherence to the prescribed treatment regimen while also screening for substance use that may increase patient risk.
UDS results can be complex and have profound implications for the patient’s treatment plan. Drug metabolites for opioids are particularly complicated; for example, synthetic and semisynthetic opioids are not detected on routine opiate immunoassays.4 This may lead a clinician to falsely assume the patient is not taking their fentanyl or tramadol medication as directed—or potentially even diverting—in the face of a negative opiate result.5 Routine UDSs are also subject to the pitfall of false-positive results due to coprescribed medications; for example, bupropion can lead to a false-positive amphetamine result, whereas sertraline can lead to a false-positive benzodiazepine result.6 Retrospective reviews of clinician behavior surrounding UDS interpretation have demonstrated knowledge gaps and inconsistent communication practices with patients.7,8
Given the complexity of UDS interpretation and its close relationship with medications, pharmacists are positioned to play an important role in the process. Pharmacists are embedded in pain-management teams and involved in prescription drug monitoring programs (PDMPs) for many health systems. The Veterans Health Administration (VHA) has supported the hiring of pain management, opioid safety, and PDMP coordinators (PMOP) at its facilities to provide clinical pain-management guidance, support national initiatives, and uphold legislative requirements.9 In many facilities, a pharmacist is hired specifically for these positions.
Clinical dashboards have been used by pharmacists in a variety of settings.10-13 They allow clinicians at a broad level to target interventions needed across a patient population, then produce a list of actionable patients to facilitate delivery of that intervention on an individual level.13 Between 2021 and 2022, a clinical dashboard to review potentially discrepant UDS results was made available for use at US Department of Veterans Affairs (VA) medical centers. Evidence exists in primary and specialty care settings that implementation of an opioid-prescribing clinical dashboard improves completion rates of risk-mitigation strategies such as UDS and opioid treatment agreements.14,15 To our knowledge there is no published research on the use and outcomes of a clinical dashboard that allows users to efficiently review discrepant UDS results when compared to a list of currently prescribed medications.
Given the availability of the UDS dashboard at the VA Black Hills Health Care System (VABHHCS) in South Dakota and the hiring of a PMOP coordinator pharmacist, the aim of this quality improvement project was 2-fold: to implement a pharmacist-led process to monitor the UDS dashboard for potentially discrepant results and to describe the quantity and types of interventions made by the clinical pharmacist leading this process.
Quality Improvement Project
A clinical UDS dashboard was created by the VA Northwest Health Network and made available for use at VHA sites between 2021 and 2022. The UDS dashboard is housed on a secure, Power BI Report Server (Microsoft), with access restricted to only those with patient health data privileges. The dashboard identifies all local patients with a UDS that returned with a potential discrepancy, defined as an unexpected positive result (eg, a detected substance not recently prescribed or documented on the patient’s medication list) and/or an unexpected negative result (eg, a prescribed substance not detected). The UDS dashboard identifies these discrepancies by comparing the patient’s current medication list (both VHA and non-VHA) to their UDS results.
The UDS dashboard displays a summary of UDSs performed, unexpected negative results, unexpected positive results, and potential discrepancies. The user may also specify the laboratory type and time frame of interest to limit displayed results. The user can then view patient-specific data for any category. Among the data are the patient’s UDS results and the completion date, detected (or nondetected) substance(s), ordering clinician, associated medication(s) with last fill date and days’ supply, and whether a confirmatory test has been performed in the past year.
VABHHCS uses an extended UDS immunoassay (PROFILE-V, MEDTOX Diagnostics) that reports on 11 substances: opiates, oxycodone, buprenorphine, methadone, amphetamines, methamphetamine, barbiturates, benzodiazepines, cocaine metabolites, cannabinoids (tetrahydrocannabinol [THC]), and phencyclidine. These substances appear on the UDS dashboard. The project protocol initially included monitoring for tramadol but that was later removed because it was not available with this UDS immunoassay.
Pharmacist Process
Either the PMOP coordinator or pharmacy resident monitored the UDS dashboard weekly. Any patients identified as having a potential discrepancy were reviewed. If the discrepancy was determined to be significant, the PMOP coordinator or pharmacy resident would review the patient electronic health record. If warranted, the patient was contacted and asked about newly prescribed medications, missed and recent medication doses, and illicit substance use. Potential interventions during in-depth review included: (1) discussing future actions with the primary care clinician and/or prescriber of the controlled substance; (2) ordering a confirmatory test on the original urine sample; (3) evaluating for sources of potential false-positive results; (4) completing an updated PDMP if not performed within the past year; (5) referring patients for substance use disorder treatment or counseling; or (6) consulting the local narcotics review committee. A progress note was entered into the electronic health record with the findings and any actions taken, and an alert for the primary care clinician and/or prescriber of the controlled substance.
Implementation and Analysis
This quality improvement project spanned 16 weeks from June 2022 through September 2022. Any patient with a UDS that returned with a significant discrepancy was reviewed. The primary outcome was interventions made by the PMOP coordinator or pharmacy resident, as well as time taken to perform the in-depth review of each patient. Patient demographics were also collected. The protocol for this project was approved by the VABHHCS pharmacy and therapeutics committee and was determined to meet guidelines for a nonresearch quality improvement project.
Results
From June 2022 through September 2022, 700 UDSs were performed at VABHHCS with 278 (39.7%) patients identified as having a potential discrepancy based on UDS results. Sixty patients (8.6%) had significant discrepancies that warranted in-depth review. The most common reasons for determining whether a potential discrepancy was not significant included unexpected negatives due to documented non-VA medications no longer being prescribed, unexpected positives due to recent expiration of a controlled substance prescription the patient was still taking, or unexpected positives due to the detection of a substance for which the clinician was already aware. During the 16-week study period, the mean number of patients warranting in-depth review was 4 per week.
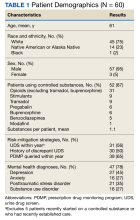
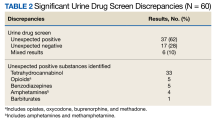
The patients were predominantly male with a mean age of 61 years, and most (87%) were prescribed at least 1 controlled substance (mean, 1.1), primarily opioids for pain management (Table 1). Most patients had recent substance risk mitigation with UDS (56%) and PDMP (65%) checks within the past year. Of the 60 patients reviewed with significant UDS discrepancies, 50% had a history of discrepant UDS results. Of the 60 UDS discrepancies, there were 37 unexpected positive results (62%), 17 unexpected negative results (28%), and 10 patients with both positive and negative results (17%). THC was the most frequently detected substance, followed by opiates, benzodiazepines, and amphetamines (Table 2).
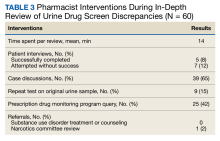
Each in-depth review with interventions by the PMOP coordinator or pharmacy resident lasted a mean of 14 minutes (Table 3). Five patients were successfully contacted for an interview and 7 patients could not be contacted. The ordering clinician of the UDS sometimes had contacted these patients prior to the PMOP coordinator or pharmacy resident reviewing the UDS dashboard, eliminating the need for additional follow-up.
The most common pharmacist intervention was discussing future actions with the primary care clinician and/or prescriber of the controlled substance (n = 39; 65%). These conversations resulted in actions such as ordering a repeat UDS with confirmatory testing at a future date or agreeing that the clinician would discuss the results and subsequent actions with the patient at an upcoming visit. Pharmacist interventions also included 25 PDMP queries (42%) and 9 orders of confirmatory UDS on the original urine sample (15%). Only 1 patient was evaluated by the narcotics review committee, which resulted in a controlled substance flag being placed on their profile. No patients were referred to substance use disorder treatment or counseling. It was offered to and declined by 1 patient, and 3 patients were already engaged in these services.
Medication therapies that could contribute to false-positive results were also evaluated. Fourteen patients who tested positive for THC had a prescription for a nonsteroidal anti-inflammatory drug or proton-pump inhibitor, which could have created a false-positive result.6 One patient who tested positive for amphetamines had a prescription for phentermine.16 No other potential false-positive results were identified.
Discussion
Findings of this project illustrate that the use of a clinical pharmacist to monitor a dashboard of discrepant UDS results created opportunities for collaboration with clinicians and impacted confirmatory testing and PDMP monitoring practices.
At the local level, the process had numerous benefits. First, it was a reasonable amount of workload to generate pharmacist interventions: the PMOP coordinator conducted an average of 4 in-depth reviews weekly, each lasting about 14 minutes. Thus, the UDS dashboard allowed the PMOP coordinator to actively surveil all incoming UDS results for potential discrepancies in about 1 hour each week. Pairing the automation of the UDS dashboard with the clinical judgment of the PMOP coordinator seemed to maximize efficiency. VABHHCS provides primary and secondary medical and surgical care to a rural population of approximately 20,000 patients across 5 states; the time required at facilities that serve a higher volume of patients may be greater.
Second, the project served as an opportunity for the PMOP coordinator to provide case-specific clinician education on UDS monitoring. As medication experts, pharmacists can apply their medication-related knowledge to UDS interpretation. This includes understanding drug metabolism and classification and how they apply to UDS results, as well as recognizing medication therapies that could contribute to false-positive UDS results. Research suggests that clinicians may have gaps in their knowledge and may welcome pharmacist assistance in interpreting UDS results.7,8
Third, the project helped improve rates of confirmatory testing for those with unexpected positive UDS results. Confirmatory testing should be strongly considered if positive results would have significant implications on the future course of treatment.4 The PMOP coordinator ordered a confirmatory test on 9 patients using the same urine sample used to conduct the initial UDS, minimizing the burden on the patient and laboratory staff. Confirmatory testing was limited by the laboratory’s sample retention period; if the need for confirmatory testing was not recognized soon enough, the sample would no longer be available for retesting. Health systems may consider the use of reflexive confirmatory testing with UDS as an alternative approach, although this may come at an additional cost and may not be warranted in many cases (eg, only 39.7% of all potential discrepancies were deemed as significant within our project).
There were notable incidental findings in our quality improvement project. Among patients with a significant discrepancy on UDS, 50% had a history of ≥ 1 discrepant UDS result. This further emphasizes the importance of appropriate use and interpretation of UDS monitoring for all clinicians, as this may prevent prolonged and potentially inappropriate treatment regimens. Secondly, rates of mental health diagnoses among those with a significant UDS discrepancy seemed relatively high compared to population-level data. For example, among veterans, the overall lifetime prevalence of posttraumatic stress disorder is estimated to be 8.0%; in our project, 35% of patients with a significant UDS discrepancy had a posttraumatic stress disorder diagnosis.17 This relationship may be an area of further study.
Lastly, it was surprising that the overall rates of UDS and PDMP checks within the past year were 56% and 65%, respectively. VABHHCS requires veterans on controlled substances to have these risk-mitigation strategies performed annually, so our suspicion is that many were falling out due to having been most recently evaluated 12 to 16 months prior. This may represent a limitation of our data-collection method, which reviewed only the previous 12 months.
Limitations
This project was carried out over a period of only 4 months. As a result, only 60 patients received an in-depth review from the PMOP coordinator. Second, the timeliness of the intervention seemed crucial, as delayed in-depth reviews resulted in fewer opportunities to order confirmatory tests or collaborate with clinicians prior to devising an updated plan. Additionally, our process called for UDS dashboard monitoring once a week. Given that the laboratory held samples for only 48 hours, twice- or thrice-weekly review of the UDS dashboard would have allowed for more confirmatory testing, along with more immediate clinician collaboration. Most importantly, the outcomes of this project are only presented via descriptive statistics and without the results of any comparison group, making it impossible to draw firm conclusions about this approach compared to standard-care processes.
Conclusions
This quality improvement project has proven to be valuable at VABHHCS and we intend to continue this pharmacist-led process to monitor the UDS dashboard. VABHHCS leadership are also discussing UDS practices more broadly to further enhance patient management. Within the VA, the PMOP coordinator—charged with being the local coordinator of appropriate pain management and opioid safety practices—is well positioned to assume these responsibilities. Outside of the VA, a pain-management clinical pharmacist or any pharmacist embedded within primary care could similarly perform these duties. Previous literature regarding the implementation of clinical dashboards suggests that with the appropriate software engineering teams and infrastructure, this tool could also be feasibly developed and implemented at other health systems relatively quickly.14
Overall, a pharmacist-led process to efficiently monitor a dashboard of discrepant UDS results led to opportunities for collaboration with clinicians and positively impacted confirmatory testing and PDMP monitoring at a rural VA health system.
Acknowledgments
The authors express their gratitude to Patrick Spoutz, PharmD, BCPS, VISN 20 Pharmacist Executive, for introducing and sharing the UDS dashboard with our team.
1. Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain - United States, 2022. MMWR Recomm Rep. 2022;71(3):1-95. doi:10.15585/mmwr.rr7103a1
2. US Department of Defense, US Department of Veterans Affairs. VA/DoD clinical practice guidelines for the use of opioids in the management of chronic pain. Version 4.0. Published 2002. Accessed January 22, 2024. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
3. Champion C, Kameg BN. Best practices in benzodiazepine prescribing and management in primary care. Nurse Pract. 2021;46(3):30-36.doi:10.1097/01.NPR.0000733684.24949.19
4. Kale N. Urine drug tests: ordering and interpretation. Am Fam Physician. 2019;99(1):33-39.
5. Gillespie E, Cunningham JM, Indovina KA. Interpretation of the urine drug screen. The Hospitalist. May 2, 2022. Accessed January 19, 2024. https://www.the-hospitalist.org/hospitalist/article/32085/interpreting-diagnostic-tests/interpretation-of-the-urine-drug-screen/
6. Schwebach A, Ball J. Urine drug screening: minimizing false-positives and false-negatives to optimize patient care. US Pharm. 2016;41(8):26-30.
7. Starrels JL, Fox AD, Kunins HV, Cunningham CO. They don’t know what they don’t know: internal medicine residents’ knowledge and confidence in urine drug test interpretation for patients with chronic pain. J Gen Intern Med. 2012;27(11):1521-1527. doi:10.1007/s11606-012-2165-7
8. Chua I, Petrides AK, Schiff GD, et al. Provider misinterpretation, documentation, and follow-up of definitive urine drug testing results. J Gen Intern Med. 2020;35(1):283-290. doi:10.1007/s11606-019-05514-5
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program (PMOP) National Program Field Roles and Responsibilities Manual. October 2021 (V1).[Source not verified]
10. Dorsch MP, Chen CS, Allen AL, et al. Nationwide implementation of a population management dashboard for monitoring direct oral anticoagulants: insights from the Veterans Affairs Health System. Circ Cardiovasc Qual Outcomes. 2023;16(2):e009256. doi:10.1161/CIRCOUTCOMES.122.009256
11. Hu AM, Pepin MJ, Hashem MG, et al. Development of a specialty medication clinical dashboard to improve tumor necrosis factor-α inhibitor safety and adherence monitoring. Am J Health Syst Pharm. 2022;79(8):683-688. doi:10.1093/ajhp/zxab454
12. Homsted FAE, Magee CE, Nesin N. Population health management in a small health system: impact of controlled substance stewardship in a patient-centered medical home. Am J Health Syst Pharm. 2017;74(18):1468-1475. doi:10.2146/ajhp161032
13. US Department of Veterans Affairs, Veterans Health Administration, Pharmacy Benefits (PBM) Services, Clinical Pharmacy Practice Office. Fact Sheet: CPS Role in Population Health Management. 2019. [Source not verified]
14. Anderson D, Zlateva I, Khatri K, Ciaburri N. Using health information technology to improve adherence to opioid prescribing guidelines in primary care. Clin J Pain. 2015;31(6):573-579. doi:10.1097/AJP.0000000000000177
15. Wang EJ, Helgesen R, Johr CR, Lacko HS, Ashburn MA, Merkel PA. Targeted program in an academic rheumatology practice to improve compliance with opioid prescribing guidelines for the treatment of chronic pain. Arthritis Care Res (Hoboken). 2021;73(10):1425-1429. doi:10.1002/acr.24354
16. Moeller KE, Kissack JC, Atayee RS, Lee KC. Clinical interpretation of urine drug tests: what clinicians need to know about urine drug screens. Mayo Clin Proc. 2017;92(5):774-796. doi:10.1016/j.mayocp.2016.12.007
17. Wisco BE, Marx BP, Wolf EJ, Miller MW, Southwick SM, Pietrzak RH. Posttraumatic stress disorder in the US veteran population: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2014;75(12):1338-46. doi:10.4088/JCP.14m09328
Urine drug screen (UDS) monitoring is a common risk-mitigation strategy tool for prescribing controlled substances.1-3 Not only is UDS monitoring highlighted by clinical practice guidelines for opioid prescribing for chronic pain,1,2 it has also been suggested as best practice for benzodiazepines3 and a consideration for other controlled substances. Monitoring UDSs helps confirm adherence to the prescribed treatment regimen while also screening for substance use that may increase patient risk.
UDS results can be complex and have profound implications for the patient’s treatment plan. Drug metabolites for opioids are particularly complicated; for example, synthetic and semisynthetic opioids are not detected on routine opiate immunoassays.4 This may lead a clinician to falsely assume the patient is not taking their fentanyl or tramadol medication as directed—or potentially even diverting—in the face of a negative opiate result.5 Routine UDSs are also subject to the pitfall of false-positive results due to coprescribed medications; for example, bupropion can lead to a false-positive amphetamine result, whereas sertraline can lead to a false-positive benzodiazepine result.6 Retrospective reviews of clinician behavior surrounding UDS interpretation have demonstrated knowledge gaps and inconsistent communication practices with patients.7,8
Given the complexity of UDS interpretation and its close relationship with medications, pharmacists are positioned to play an important role in the process. Pharmacists are embedded in pain-management teams and involved in prescription drug monitoring programs (PDMPs) for many health systems. The Veterans Health Administration (VHA) has supported the hiring of pain management, opioid safety, and PDMP coordinators (PMOP) at its facilities to provide clinical pain-management guidance, support national initiatives, and uphold legislative requirements.9 In many facilities, a pharmacist is hired specifically for these positions.
Clinical dashboards have been used by pharmacists in a variety of settings.10-13 They allow clinicians at a broad level to target interventions needed across a patient population, then produce a list of actionable patients to facilitate delivery of that intervention on an individual level.13 Between 2021 and 2022, a clinical dashboard to review potentially discrepant UDS results was made available for use at US Department of Veterans Affairs (VA) medical centers. Evidence exists in primary and specialty care settings that implementation of an opioid-prescribing clinical dashboard improves completion rates of risk-mitigation strategies such as UDS and opioid treatment agreements.14,15 To our knowledge there is no published research on the use and outcomes of a clinical dashboard that allows users to efficiently review discrepant UDS results when compared to a list of currently prescribed medications.
Given the availability of the UDS dashboard at the VA Black Hills Health Care System (VABHHCS) in South Dakota and the hiring of a PMOP coordinator pharmacist, the aim of this quality improvement project was 2-fold: to implement a pharmacist-led process to monitor the UDS dashboard for potentially discrepant results and to describe the quantity and types of interventions made by the clinical pharmacist leading this process.
Quality Improvement Project
A clinical UDS dashboard was created by the VA Northwest Health Network and made available for use at VHA sites between 2021 and 2022. The UDS dashboard is housed on a secure, Power BI Report Server (Microsoft), with access restricted to only those with patient health data privileges. The dashboard identifies all local patients with a UDS that returned with a potential discrepancy, defined as an unexpected positive result (eg, a detected substance not recently prescribed or documented on the patient’s medication list) and/or an unexpected negative result (eg, a prescribed substance not detected). The UDS dashboard identifies these discrepancies by comparing the patient’s current medication list (both VHA and non-VHA) to their UDS results.
The UDS dashboard displays a summary of UDSs performed, unexpected negative results, unexpected positive results, and potential discrepancies. The user may also specify the laboratory type and time frame of interest to limit displayed results. The user can then view patient-specific data for any category. Among the data are the patient’s UDS results and the completion date, detected (or nondetected) substance(s), ordering clinician, associated medication(s) with last fill date and days’ supply, and whether a confirmatory test has been performed in the past year.
VABHHCS uses an extended UDS immunoassay (PROFILE-V, MEDTOX Diagnostics) that reports on 11 substances: opiates, oxycodone, buprenorphine, methadone, amphetamines, methamphetamine, barbiturates, benzodiazepines, cocaine metabolites, cannabinoids (tetrahydrocannabinol [THC]), and phencyclidine. These substances appear on the UDS dashboard. The project protocol initially included monitoring for tramadol but that was later removed because it was not available with this UDS immunoassay.
Pharmacist Process
Either the PMOP coordinator or pharmacy resident monitored the UDS dashboard weekly. Any patients identified as having a potential discrepancy were reviewed. If the discrepancy was determined to be significant, the PMOP coordinator or pharmacy resident would review the patient electronic health record. If warranted, the patient was contacted and asked about newly prescribed medications, missed and recent medication doses, and illicit substance use. Potential interventions during in-depth review included: (1) discussing future actions with the primary care clinician and/or prescriber of the controlled substance; (2) ordering a confirmatory test on the original urine sample; (3) evaluating for sources of potential false-positive results; (4) completing an updated PDMP if not performed within the past year; (5) referring patients for substance use disorder treatment or counseling; or (6) consulting the local narcotics review committee. A progress note was entered into the electronic health record with the findings and any actions taken, and an alert for the primary care clinician and/or prescriber of the controlled substance.
Implementation and Analysis
This quality improvement project spanned 16 weeks from June 2022 through September 2022. Any patient with a UDS that returned with a significant discrepancy was reviewed. The primary outcome was interventions made by the PMOP coordinator or pharmacy resident, as well as time taken to perform the in-depth review of each patient. Patient demographics were also collected. The protocol for this project was approved by the VABHHCS pharmacy and therapeutics committee and was determined to meet guidelines for a nonresearch quality improvement project.
Results
From June 2022 through September 2022, 700 UDSs were performed at VABHHCS with 278 (39.7%) patients identified as having a potential discrepancy based on UDS results. Sixty patients (8.6%) had significant discrepancies that warranted in-depth review. The most common reasons for determining whether a potential discrepancy was not significant included unexpected negatives due to documented non-VA medications no longer being prescribed, unexpected positives due to recent expiration of a controlled substance prescription the patient was still taking, or unexpected positives due to the detection of a substance for which the clinician was already aware. During the 16-week study period, the mean number of patients warranting in-depth review was 4 per week.
The patients were predominantly male with a mean age of 61 years, and most (87%) were prescribed at least 1 controlled substance (mean, 1.1), primarily opioids for pain management (Table 1). Most patients had recent substance risk mitigation with UDS (56%) and PDMP (65%) checks within the past year. Of the 60 patients reviewed with significant UDS discrepancies, 50% had a history of discrepant UDS results. Of the 60 UDS discrepancies, there were 37 unexpected positive results (62%), 17 unexpected negative results (28%), and 10 patients with both positive and negative results (17%). THC was the most frequently detected substance, followed by opiates, benzodiazepines, and amphetamines (Table 2).
Each in-depth review with interventions by the PMOP coordinator or pharmacy resident lasted a mean of 14 minutes (Table 3). Five patients were successfully contacted for an interview and 7 patients could not be contacted. The ordering clinician of the UDS sometimes had contacted these patients prior to the PMOP coordinator or pharmacy resident reviewing the UDS dashboard, eliminating the need for additional follow-up.
The most common pharmacist intervention was discussing future actions with the primary care clinician and/or prescriber of the controlled substance (n = 39; 65%). These conversations resulted in actions such as ordering a repeat UDS with confirmatory testing at a future date or agreeing that the clinician would discuss the results and subsequent actions with the patient at an upcoming visit. Pharmacist interventions also included 25 PDMP queries (42%) and 9 orders of confirmatory UDS on the original urine sample (15%). Only 1 patient was evaluated by the narcotics review committee, which resulted in a controlled substance flag being placed on their profile. No patients were referred to substance use disorder treatment or counseling. It was offered to and declined by 1 patient, and 3 patients were already engaged in these services.
Medication therapies that could contribute to false-positive results were also evaluated. Fourteen patients who tested positive for THC had a prescription for a nonsteroidal anti-inflammatory drug or proton-pump inhibitor, which could have created a false-positive result.6 One patient who tested positive for amphetamines had a prescription for phentermine.16 No other potential false-positive results were identified.
Discussion
Findings of this project illustrate that the use of a clinical pharmacist to monitor a dashboard of discrepant UDS results created opportunities for collaboration with clinicians and impacted confirmatory testing and PDMP monitoring practices.
At the local level, the process had numerous benefits. First, it was a reasonable amount of workload to generate pharmacist interventions: the PMOP coordinator conducted an average of 4 in-depth reviews weekly, each lasting about 14 minutes. Thus, the UDS dashboard allowed the PMOP coordinator to actively surveil all incoming UDS results for potential discrepancies in about 1 hour each week. Pairing the automation of the UDS dashboard with the clinical judgment of the PMOP coordinator seemed to maximize efficiency. VABHHCS provides primary and secondary medical and surgical care to a rural population of approximately 20,000 patients across 5 states; the time required at facilities that serve a higher volume of patients may be greater.
Second, the project served as an opportunity for the PMOP coordinator to provide case-specific clinician education on UDS monitoring. As medication experts, pharmacists can apply their medication-related knowledge to UDS interpretation. This includes understanding drug metabolism and classification and how they apply to UDS results, as well as recognizing medication therapies that could contribute to false-positive UDS results. Research suggests that clinicians may have gaps in their knowledge and may welcome pharmacist assistance in interpreting UDS results.7,8
Third, the project helped improve rates of confirmatory testing for those with unexpected positive UDS results. Confirmatory testing should be strongly considered if positive results would have significant implications on the future course of treatment.4 The PMOP coordinator ordered a confirmatory test on 9 patients using the same urine sample used to conduct the initial UDS, minimizing the burden on the patient and laboratory staff. Confirmatory testing was limited by the laboratory’s sample retention period; if the need for confirmatory testing was not recognized soon enough, the sample would no longer be available for retesting. Health systems may consider the use of reflexive confirmatory testing with UDS as an alternative approach, although this may come at an additional cost and may not be warranted in many cases (eg, only 39.7% of all potential discrepancies were deemed as significant within our project).
There were notable incidental findings in our quality improvement project. Among patients with a significant discrepancy on UDS, 50% had a history of ≥ 1 discrepant UDS result. This further emphasizes the importance of appropriate use and interpretation of UDS monitoring for all clinicians, as this may prevent prolonged and potentially inappropriate treatment regimens. Secondly, rates of mental health diagnoses among those with a significant UDS discrepancy seemed relatively high compared to population-level data. For example, among veterans, the overall lifetime prevalence of posttraumatic stress disorder is estimated to be 8.0%; in our project, 35% of patients with a significant UDS discrepancy had a posttraumatic stress disorder diagnosis.17 This relationship may be an area of further study.
Lastly, it was surprising that the overall rates of UDS and PDMP checks within the past year were 56% and 65%, respectively. VABHHCS requires veterans on controlled substances to have these risk-mitigation strategies performed annually, so our suspicion is that many were falling out due to having been most recently evaluated 12 to 16 months prior. This may represent a limitation of our data-collection method, which reviewed only the previous 12 months.
Limitations
This project was carried out over a period of only 4 months. As a result, only 60 patients received an in-depth review from the PMOP coordinator. Second, the timeliness of the intervention seemed crucial, as delayed in-depth reviews resulted in fewer opportunities to order confirmatory tests or collaborate with clinicians prior to devising an updated plan. Additionally, our process called for UDS dashboard monitoring once a week. Given that the laboratory held samples for only 48 hours, twice- or thrice-weekly review of the UDS dashboard would have allowed for more confirmatory testing, along with more immediate clinician collaboration. Most importantly, the outcomes of this project are only presented via descriptive statistics and without the results of any comparison group, making it impossible to draw firm conclusions about this approach compared to standard-care processes.
Conclusions
This quality improvement project has proven to be valuable at VABHHCS and we intend to continue this pharmacist-led process to monitor the UDS dashboard. VABHHCS leadership are also discussing UDS practices more broadly to further enhance patient management. Within the VA, the PMOP coordinator—charged with being the local coordinator of appropriate pain management and opioid safety practices—is well positioned to assume these responsibilities. Outside of the VA, a pain-management clinical pharmacist or any pharmacist embedded within primary care could similarly perform these duties. Previous literature regarding the implementation of clinical dashboards suggests that with the appropriate software engineering teams and infrastructure, this tool could also be feasibly developed and implemented at other health systems relatively quickly.14
Overall, a pharmacist-led process to efficiently monitor a dashboard of discrepant UDS results led to opportunities for collaboration with clinicians and positively impacted confirmatory testing and PDMP monitoring at a rural VA health system.
Acknowledgments
The authors express their gratitude to Patrick Spoutz, PharmD, BCPS, VISN 20 Pharmacist Executive, for introducing and sharing the UDS dashboard with our team.
Urine drug screen (UDS) monitoring is a common risk-mitigation strategy tool for prescribing controlled substances.1-3 Not only is UDS monitoring highlighted by clinical practice guidelines for opioid prescribing for chronic pain,1,2 it has also been suggested as best practice for benzodiazepines3 and a consideration for other controlled substances. Monitoring UDSs helps confirm adherence to the prescribed treatment regimen while also screening for substance use that may increase patient risk.
UDS results can be complex and have profound implications for the patient’s treatment plan. Drug metabolites for opioids are particularly complicated; for example, synthetic and semisynthetic opioids are not detected on routine opiate immunoassays.4 This may lead a clinician to falsely assume the patient is not taking their fentanyl or tramadol medication as directed—or potentially even diverting—in the face of a negative opiate result.5 Routine UDSs are also subject to the pitfall of false-positive results due to coprescribed medications; for example, bupropion can lead to a false-positive amphetamine result, whereas sertraline can lead to a false-positive benzodiazepine result.6 Retrospective reviews of clinician behavior surrounding UDS interpretation have demonstrated knowledge gaps and inconsistent communication practices with patients.7,8
Given the complexity of UDS interpretation and its close relationship with medications, pharmacists are positioned to play an important role in the process. Pharmacists are embedded in pain-management teams and involved in prescription drug monitoring programs (PDMPs) for many health systems. The Veterans Health Administration (VHA) has supported the hiring of pain management, opioid safety, and PDMP coordinators (PMOP) at its facilities to provide clinical pain-management guidance, support national initiatives, and uphold legislative requirements.9 In many facilities, a pharmacist is hired specifically for these positions.
Clinical dashboards have been used by pharmacists in a variety of settings.10-13 They allow clinicians at a broad level to target interventions needed across a patient population, then produce a list of actionable patients to facilitate delivery of that intervention on an individual level.13 Between 2021 and 2022, a clinical dashboard to review potentially discrepant UDS results was made available for use at US Department of Veterans Affairs (VA) medical centers. Evidence exists in primary and specialty care settings that implementation of an opioid-prescribing clinical dashboard improves completion rates of risk-mitigation strategies such as UDS and opioid treatment agreements.14,15 To our knowledge there is no published research on the use and outcomes of a clinical dashboard that allows users to efficiently review discrepant UDS results when compared to a list of currently prescribed medications.
Given the availability of the UDS dashboard at the VA Black Hills Health Care System (VABHHCS) in South Dakota and the hiring of a PMOP coordinator pharmacist, the aim of this quality improvement project was 2-fold: to implement a pharmacist-led process to monitor the UDS dashboard for potentially discrepant results and to describe the quantity and types of interventions made by the clinical pharmacist leading this process.
Quality Improvement Project
A clinical UDS dashboard was created by the VA Northwest Health Network and made available for use at VHA sites between 2021 and 2022. The UDS dashboard is housed on a secure, Power BI Report Server (Microsoft), with access restricted to only those with patient health data privileges. The dashboard identifies all local patients with a UDS that returned with a potential discrepancy, defined as an unexpected positive result (eg, a detected substance not recently prescribed or documented on the patient’s medication list) and/or an unexpected negative result (eg, a prescribed substance not detected). The UDS dashboard identifies these discrepancies by comparing the patient’s current medication list (both VHA and non-VHA) to their UDS results.
The UDS dashboard displays a summary of UDSs performed, unexpected negative results, unexpected positive results, and potential discrepancies. The user may also specify the laboratory type and time frame of interest to limit displayed results. The user can then view patient-specific data for any category. Among the data are the patient’s UDS results and the completion date, detected (or nondetected) substance(s), ordering clinician, associated medication(s) with last fill date and days’ supply, and whether a confirmatory test has been performed in the past year.
VABHHCS uses an extended UDS immunoassay (PROFILE-V, MEDTOX Diagnostics) that reports on 11 substances: opiates, oxycodone, buprenorphine, methadone, amphetamines, methamphetamine, barbiturates, benzodiazepines, cocaine metabolites, cannabinoids (tetrahydrocannabinol [THC]), and phencyclidine. These substances appear on the UDS dashboard. The project protocol initially included monitoring for tramadol but that was later removed because it was not available with this UDS immunoassay.
Pharmacist Process
Either the PMOP coordinator or pharmacy resident monitored the UDS dashboard weekly. Any patients identified as having a potential discrepancy were reviewed. If the discrepancy was determined to be significant, the PMOP coordinator or pharmacy resident would review the patient electronic health record. If warranted, the patient was contacted and asked about newly prescribed medications, missed and recent medication doses, and illicit substance use. Potential interventions during in-depth review included: (1) discussing future actions with the primary care clinician and/or prescriber of the controlled substance; (2) ordering a confirmatory test on the original urine sample; (3) evaluating for sources of potential false-positive results; (4) completing an updated PDMP if not performed within the past year; (5) referring patients for substance use disorder treatment or counseling; or (6) consulting the local narcotics review committee. A progress note was entered into the electronic health record with the findings and any actions taken, and an alert for the primary care clinician and/or prescriber of the controlled substance.
Implementation and Analysis
This quality improvement project spanned 16 weeks from June 2022 through September 2022. Any patient with a UDS that returned with a significant discrepancy was reviewed. The primary outcome was interventions made by the PMOP coordinator or pharmacy resident, as well as time taken to perform the in-depth review of each patient. Patient demographics were also collected. The protocol for this project was approved by the VABHHCS pharmacy and therapeutics committee and was determined to meet guidelines for a nonresearch quality improvement project.
Results
From June 2022 through September 2022, 700 UDSs were performed at VABHHCS with 278 (39.7%) patients identified as having a potential discrepancy based on UDS results. Sixty patients (8.6%) had significant discrepancies that warranted in-depth review. The most common reasons for determining whether a potential discrepancy was not significant included unexpected negatives due to documented non-VA medications no longer being prescribed, unexpected positives due to recent expiration of a controlled substance prescription the patient was still taking, or unexpected positives due to the detection of a substance for which the clinician was already aware. During the 16-week study period, the mean number of patients warranting in-depth review was 4 per week.
The patients were predominantly male with a mean age of 61 years, and most (87%) were prescribed at least 1 controlled substance (mean, 1.1), primarily opioids for pain management (Table 1). Most patients had recent substance risk mitigation with UDS (56%) and PDMP (65%) checks within the past year. Of the 60 patients reviewed with significant UDS discrepancies, 50% had a history of discrepant UDS results. Of the 60 UDS discrepancies, there were 37 unexpected positive results (62%), 17 unexpected negative results (28%), and 10 patients with both positive and negative results (17%). THC was the most frequently detected substance, followed by opiates, benzodiazepines, and amphetamines (Table 2).
Each in-depth review with interventions by the PMOP coordinator or pharmacy resident lasted a mean of 14 minutes (Table 3). Five patients were successfully contacted for an interview and 7 patients could not be contacted. The ordering clinician of the UDS sometimes had contacted these patients prior to the PMOP coordinator or pharmacy resident reviewing the UDS dashboard, eliminating the need for additional follow-up.
The most common pharmacist intervention was discussing future actions with the primary care clinician and/or prescriber of the controlled substance (n = 39; 65%). These conversations resulted in actions such as ordering a repeat UDS with confirmatory testing at a future date or agreeing that the clinician would discuss the results and subsequent actions with the patient at an upcoming visit. Pharmacist interventions also included 25 PDMP queries (42%) and 9 orders of confirmatory UDS on the original urine sample (15%). Only 1 patient was evaluated by the narcotics review committee, which resulted in a controlled substance flag being placed on their profile. No patients were referred to substance use disorder treatment or counseling. It was offered to and declined by 1 patient, and 3 patients were already engaged in these services.
Medication therapies that could contribute to false-positive results were also evaluated. Fourteen patients who tested positive for THC had a prescription for a nonsteroidal anti-inflammatory drug or proton-pump inhibitor, which could have created a false-positive result.6 One patient who tested positive for amphetamines had a prescription for phentermine.16 No other potential false-positive results were identified.
Discussion
Findings of this project illustrate that the use of a clinical pharmacist to monitor a dashboard of discrepant UDS results created opportunities for collaboration with clinicians and impacted confirmatory testing and PDMP monitoring practices.
At the local level, the process had numerous benefits. First, it was a reasonable amount of workload to generate pharmacist interventions: the PMOP coordinator conducted an average of 4 in-depth reviews weekly, each lasting about 14 minutes. Thus, the UDS dashboard allowed the PMOP coordinator to actively surveil all incoming UDS results for potential discrepancies in about 1 hour each week. Pairing the automation of the UDS dashboard with the clinical judgment of the PMOP coordinator seemed to maximize efficiency. VABHHCS provides primary and secondary medical and surgical care to a rural population of approximately 20,000 patients across 5 states; the time required at facilities that serve a higher volume of patients may be greater.
Second, the project served as an opportunity for the PMOP coordinator to provide case-specific clinician education on UDS monitoring. As medication experts, pharmacists can apply their medication-related knowledge to UDS interpretation. This includes understanding drug metabolism and classification and how they apply to UDS results, as well as recognizing medication therapies that could contribute to false-positive UDS results. Research suggests that clinicians may have gaps in their knowledge and may welcome pharmacist assistance in interpreting UDS results.7,8
Third, the project helped improve rates of confirmatory testing for those with unexpected positive UDS results. Confirmatory testing should be strongly considered if positive results would have significant implications on the future course of treatment.4 The PMOP coordinator ordered a confirmatory test on 9 patients using the same urine sample used to conduct the initial UDS, minimizing the burden on the patient and laboratory staff. Confirmatory testing was limited by the laboratory’s sample retention period; if the need for confirmatory testing was not recognized soon enough, the sample would no longer be available for retesting. Health systems may consider the use of reflexive confirmatory testing with UDS as an alternative approach, although this may come at an additional cost and may not be warranted in many cases (eg, only 39.7% of all potential discrepancies were deemed as significant within our project).
There were notable incidental findings in our quality improvement project. Among patients with a significant discrepancy on UDS, 50% had a history of ≥ 1 discrepant UDS result. This further emphasizes the importance of appropriate use and interpretation of UDS monitoring for all clinicians, as this may prevent prolonged and potentially inappropriate treatment regimens. Secondly, rates of mental health diagnoses among those with a significant UDS discrepancy seemed relatively high compared to population-level data. For example, among veterans, the overall lifetime prevalence of posttraumatic stress disorder is estimated to be 8.0%; in our project, 35% of patients with a significant UDS discrepancy had a posttraumatic stress disorder diagnosis.17 This relationship may be an area of further study.
Lastly, it was surprising that the overall rates of UDS and PDMP checks within the past year were 56% and 65%, respectively. VABHHCS requires veterans on controlled substances to have these risk-mitigation strategies performed annually, so our suspicion is that many were falling out due to having been most recently evaluated 12 to 16 months prior. This may represent a limitation of our data-collection method, which reviewed only the previous 12 months.
Limitations
This project was carried out over a period of only 4 months. As a result, only 60 patients received an in-depth review from the PMOP coordinator. Second, the timeliness of the intervention seemed crucial, as delayed in-depth reviews resulted in fewer opportunities to order confirmatory tests or collaborate with clinicians prior to devising an updated plan. Additionally, our process called for UDS dashboard monitoring once a week. Given that the laboratory held samples for only 48 hours, twice- or thrice-weekly review of the UDS dashboard would have allowed for more confirmatory testing, along with more immediate clinician collaboration. Most importantly, the outcomes of this project are only presented via descriptive statistics and without the results of any comparison group, making it impossible to draw firm conclusions about this approach compared to standard-care processes.
Conclusions
This quality improvement project has proven to be valuable at VABHHCS and we intend to continue this pharmacist-led process to monitor the UDS dashboard. VABHHCS leadership are also discussing UDS practices more broadly to further enhance patient management. Within the VA, the PMOP coordinator—charged with being the local coordinator of appropriate pain management and opioid safety practices—is well positioned to assume these responsibilities. Outside of the VA, a pain-management clinical pharmacist or any pharmacist embedded within primary care could similarly perform these duties. Previous literature regarding the implementation of clinical dashboards suggests that with the appropriate software engineering teams and infrastructure, this tool could also be feasibly developed and implemented at other health systems relatively quickly.14
Overall, a pharmacist-led process to efficiently monitor a dashboard of discrepant UDS results led to opportunities for collaboration with clinicians and positively impacted confirmatory testing and PDMP monitoring at a rural VA health system.
Acknowledgments
The authors express their gratitude to Patrick Spoutz, PharmD, BCPS, VISN 20 Pharmacist Executive, for introducing and sharing the UDS dashboard with our team.
1. Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain - United States, 2022. MMWR Recomm Rep. 2022;71(3):1-95. doi:10.15585/mmwr.rr7103a1
2. US Department of Defense, US Department of Veterans Affairs. VA/DoD clinical practice guidelines for the use of opioids in the management of chronic pain. Version 4.0. Published 2002. Accessed January 22, 2024. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
3. Champion C, Kameg BN. Best practices in benzodiazepine prescribing and management in primary care. Nurse Pract. 2021;46(3):30-36.doi:10.1097/01.NPR.0000733684.24949.19
4. Kale N. Urine drug tests: ordering and interpretation. Am Fam Physician. 2019;99(1):33-39.
5. Gillespie E, Cunningham JM, Indovina KA. Interpretation of the urine drug screen. The Hospitalist. May 2, 2022. Accessed January 19, 2024. https://www.the-hospitalist.org/hospitalist/article/32085/interpreting-diagnostic-tests/interpretation-of-the-urine-drug-screen/
6. Schwebach A, Ball J. Urine drug screening: minimizing false-positives and false-negatives to optimize patient care. US Pharm. 2016;41(8):26-30.
7. Starrels JL, Fox AD, Kunins HV, Cunningham CO. They don’t know what they don’t know: internal medicine residents’ knowledge and confidence in urine drug test interpretation for patients with chronic pain. J Gen Intern Med. 2012;27(11):1521-1527. doi:10.1007/s11606-012-2165-7
8. Chua I, Petrides AK, Schiff GD, et al. Provider misinterpretation, documentation, and follow-up of definitive urine drug testing results. J Gen Intern Med. 2020;35(1):283-290. doi:10.1007/s11606-019-05514-5
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program (PMOP) National Program Field Roles and Responsibilities Manual. October 2021 (V1).[Source not verified]
10. Dorsch MP, Chen CS, Allen AL, et al. Nationwide implementation of a population management dashboard for monitoring direct oral anticoagulants: insights from the Veterans Affairs Health System. Circ Cardiovasc Qual Outcomes. 2023;16(2):e009256. doi:10.1161/CIRCOUTCOMES.122.009256
11. Hu AM, Pepin MJ, Hashem MG, et al. Development of a specialty medication clinical dashboard to improve tumor necrosis factor-α inhibitor safety and adherence monitoring. Am J Health Syst Pharm. 2022;79(8):683-688. doi:10.1093/ajhp/zxab454
12. Homsted FAE, Magee CE, Nesin N. Population health management in a small health system: impact of controlled substance stewardship in a patient-centered medical home. Am J Health Syst Pharm. 2017;74(18):1468-1475. doi:10.2146/ajhp161032
13. US Department of Veterans Affairs, Veterans Health Administration, Pharmacy Benefits (PBM) Services, Clinical Pharmacy Practice Office. Fact Sheet: CPS Role in Population Health Management. 2019. [Source not verified]
14. Anderson D, Zlateva I, Khatri K, Ciaburri N. Using health information technology to improve adherence to opioid prescribing guidelines in primary care. Clin J Pain. 2015;31(6):573-579. doi:10.1097/AJP.0000000000000177
15. Wang EJ, Helgesen R, Johr CR, Lacko HS, Ashburn MA, Merkel PA. Targeted program in an academic rheumatology practice to improve compliance with opioid prescribing guidelines for the treatment of chronic pain. Arthritis Care Res (Hoboken). 2021;73(10):1425-1429. doi:10.1002/acr.24354
16. Moeller KE, Kissack JC, Atayee RS, Lee KC. Clinical interpretation of urine drug tests: what clinicians need to know about urine drug screens. Mayo Clin Proc. 2017;92(5):774-796. doi:10.1016/j.mayocp.2016.12.007
17. Wisco BE, Marx BP, Wolf EJ, Miller MW, Southwick SM, Pietrzak RH. Posttraumatic stress disorder in the US veteran population: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2014;75(12):1338-46. doi:10.4088/JCP.14m09328
1. Dowell D, Ragan KR, Jones CM, Baldwin GT, Chou R. CDC Clinical Practice Guideline for Prescribing Opioids for Pain - United States, 2022. MMWR Recomm Rep. 2022;71(3):1-95. doi:10.15585/mmwr.rr7103a1
2. US Department of Defense, US Department of Veterans Affairs. VA/DoD clinical practice guidelines for the use of opioids in the management of chronic pain. Version 4.0. Published 2002. Accessed January 22, 2024. https://www.healthquality.va.gov/guidelines/Pain/cot/VADoDOpioidsCPG.pdf
3. Champion C, Kameg BN. Best practices in benzodiazepine prescribing and management in primary care. Nurse Pract. 2021;46(3):30-36.doi:10.1097/01.NPR.0000733684.24949.19
4. Kale N. Urine drug tests: ordering and interpretation. Am Fam Physician. 2019;99(1):33-39.
5. Gillespie E, Cunningham JM, Indovina KA. Interpretation of the urine drug screen. The Hospitalist. May 2, 2022. Accessed January 19, 2024. https://www.the-hospitalist.org/hospitalist/article/32085/interpreting-diagnostic-tests/interpretation-of-the-urine-drug-screen/
6. Schwebach A, Ball J. Urine drug screening: minimizing false-positives and false-negatives to optimize patient care. US Pharm. 2016;41(8):26-30.
7. Starrels JL, Fox AD, Kunins HV, Cunningham CO. They don’t know what they don’t know: internal medicine residents’ knowledge and confidence in urine drug test interpretation for patients with chronic pain. J Gen Intern Med. 2012;27(11):1521-1527. doi:10.1007/s11606-012-2165-7
8. Chua I, Petrides AK, Schiff GD, et al. Provider misinterpretation, documentation, and follow-up of definitive urine drug testing results. J Gen Intern Med. 2020;35(1):283-290. doi:10.1007/s11606-019-05514-5
9. US Department of Veterans Affairs, Veterans Health Administration. VHA Pain Management, Opioid Safety, and Prescription Drug Monitoring Program (PMOP) National Program Field Roles and Responsibilities Manual. October 2021 (V1).[Source not verified]
10. Dorsch MP, Chen CS, Allen AL, et al. Nationwide implementation of a population management dashboard for monitoring direct oral anticoagulants: insights from the Veterans Affairs Health System. Circ Cardiovasc Qual Outcomes. 2023;16(2):e009256. doi:10.1161/CIRCOUTCOMES.122.009256
11. Hu AM, Pepin MJ, Hashem MG, et al. Development of a specialty medication clinical dashboard to improve tumor necrosis factor-α inhibitor safety and adherence monitoring. Am J Health Syst Pharm. 2022;79(8):683-688. doi:10.1093/ajhp/zxab454
12. Homsted FAE, Magee CE, Nesin N. Population health management in a small health system: impact of controlled substance stewardship in a patient-centered medical home. Am J Health Syst Pharm. 2017;74(18):1468-1475. doi:10.2146/ajhp161032
13. US Department of Veterans Affairs, Veterans Health Administration, Pharmacy Benefits (PBM) Services, Clinical Pharmacy Practice Office. Fact Sheet: CPS Role in Population Health Management. 2019. [Source not verified]
14. Anderson D, Zlateva I, Khatri K, Ciaburri N. Using health information technology to improve adherence to opioid prescribing guidelines in primary care. Clin J Pain. 2015;31(6):573-579. doi:10.1097/AJP.0000000000000177
15. Wang EJ, Helgesen R, Johr CR, Lacko HS, Ashburn MA, Merkel PA. Targeted program in an academic rheumatology practice to improve compliance with opioid prescribing guidelines for the treatment of chronic pain. Arthritis Care Res (Hoboken). 2021;73(10):1425-1429. doi:10.1002/acr.24354
16. Moeller KE, Kissack JC, Atayee RS, Lee KC. Clinical interpretation of urine drug tests: what clinicians need to know about urine drug screens. Mayo Clin Proc. 2017;92(5):774-796. doi:10.1016/j.mayocp.2016.12.007
17. Wisco BE, Marx BP, Wolf EJ, Miller MW, Southwick SM, Pietrzak RH. Posttraumatic stress disorder in the US veteran population: results from the National Health and Resilience in Veterans Study. J Clin Psychiatry. 2014;75(12):1338-46. doi:10.4088/JCP.14m09328