User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
Scoring formula consolidates stroke, bleeding risk in atrial fib patients
ROME – A new risk-stratification formula for atrial fibrillation patients starting oral anticoagulant therapy helps sort out their potential net benefit on edoxaban, compared with warfarin.
This risk score “could help guide selection of treatment” with a vitamin K antagonist such as warfarin or a new oral anticoagulant (NOAC) such as edoxaban, Christina L. Fanola, MD, said at the annual congress of the European Society of Cardiology.
“It’s a great time to think about this type of score, because so many more patients are being diagnosed with atrial fibrillation and there is a lot of clinical equipoise” over which anticoagulant to start patients on, said Dr. Fanola, a cardiologist at Brigham and Women’s Hospital in Boston. She said she and her associates hope to externally validate the score and test it in cohorts that received other NOACs, such as apixaban (Eliquis), dabigatran (Pradaxa), or rivaroxaban (Xarelto), but it is very possible that scoring might differ from one NOAC to the next. “Each NOAC may need its own scoring formula,” Dr. Fanola said in an interview.
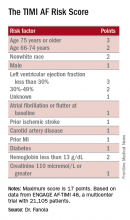
A Cox proportional hazards model identified 10 demographic, clinical, and laboratory features that had significant, independent correlations to a primary outcome of disabling stroke, life-threatening bleeding, or death. After weighing the point allocation for each item by the strength of its association, the researchers developed a scoring formula in a model that could account for about 69% of the three combined adverse outcomes.
An analysis that applied the scoring formula back to the ENGAGE AF-TIMI 48 database showed that a low-risk score of 0-6 correlated with a 4% per year rate of disabling stroke, life-threatening bleed, or death; an intermediate-risk score of 7-9 correlated with a 10% per year incidence of this combined outcome, and a high-risk score of 10 or greater linked with a 21% annual event rate.
Dr. Fanola and her associates ran a further analysis that evaluated the efficacy of edoxaban, compared with warfarin, among the patients in each of these risk strata. The high-risk patients received a major benefit from edoxaban, with a 30% overall incidence of the combined endpoint during 3 years of follow-up, compared with a 51% rate among patients on warfarin, a 21-percentage-point reduction in adverse events. Intermediate-risk patients also received a significant benefit, with a 26% event rate on warfarin and an 18% rate on edoxaban. But low-risk patients had identical 10% event rates with either treatment.
These findings suggest that atrial fibrillation patients with a TIMI AF score that is high or intermediate would have a better chance for a good outcome on edoxaban, or perhaps a different NOAC, than on warfarin. Low-risk patients seem to have similar outcomes on edoxaban or warfarin, so other considerations can come into play for choosing between these drug options, such as the cost of treatment and the inconvenience of regular warfarin monitoring, Dr. Fanola said.
ENGAGE AF-TIMI 48 was sponsored by Daiichi Sankyo, the company that markets edoxaban. Dr. Fanola had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ROME – A new risk-stratification formula for atrial fibrillation patients starting oral anticoagulant therapy helps sort out their potential net benefit on edoxaban, compared with warfarin.
This risk score “could help guide selection of treatment” with a vitamin K antagonist such as warfarin or a new oral anticoagulant (NOAC) such as edoxaban, Christina L. Fanola, MD, said at the annual congress of the European Society of Cardiology.
“It’s a great time to think about this type of score, because so many more patients are being diagnosed with atrial fibrillation and there is a lot of clinical equipoise” over which anticoagulant to start patients on, said Dr. Fanola, a cardiologist at Brigham and Women’s Hospital in Boston. She said she and her associates hope to externally validate the score and test it in cohorts that received other NOACs, such as apixaban (Eliquis), dabigatran (Pradaxa), or rivaroxaban (Xarelto), but it is very possible that scoring might differ from one NOAC to the next. “Each NOAC may need its own scoring formula,” Dr. Fanola said in an interview.
A Cox proportional hazards model identified 10 demographic, clinical, and laboratory features that had significant, independent correlations to a primary outcome of disabling stroke, life-threatening bleeding, or death. After weighing the point allocation for each item by the strength of its association, the researchers developed a scoring formula in a model that could account for about 69% of the three combined adverse outcomes.
An analysis that applied the scoring formula back to the ENGAGE AF-TIMI 48 database showed that a low-risk score of 0-6 correlated with a 4% per year rate of disabling stroke, life-threatening bleed, or death; an intermediate-risk score of 7-9 correlated with a 10% per year incidence of this combined outcome, and a high-risk score of 10 or greater linked with a 21% annual event rate.
Dr. Fanola and her associates ran a further analysis that evaluated the efficacy of edoxaban, compared with warfarin, among the patients in each of these risk strata. The high-risk patients received a major benefit from edoxaban, with a 30% overall incidence of the combined endpoint during 3 years of follow-up, compared with a 51% rate among patients on warfarin, a 21-percentage-point reduction in adverse events. Intermediate-risk patients also received a significant benefit, with a 26% event rate on warfarin and an 18% rate on edoxaban. But low-risk patients had identical 10% event rates with either treatment.
These findings suggest that atrial fibrillation patients with a TIMI AF score that is high or intermediate would have a better chance for a good outcome on edoxaban, or perhaps a different NOAC, than on warfarin. Low-risk patients seem to have similar outcomes on edoxaban or warfarin, so other considerations can come into play for choosing between these drug options, such as the cost of treatment and the inconvenience of regular warfarin monitoring, Dr. Fanola said.
ENGAGE AF-TIMI 48 was sponsored by Daiichi Sankyo, the company that markets edoxaban. Dr. Fanola had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ROME – A new risk-stratification formula for atrial fibrillation patients starting oral anticoagulant therapy helps sort out their potential net benefit on edoxaban, compared with warfarin.
This risk score “could help guide selection of treatment” with a vitamin K antagonist such as warfarin or a new oral anticoagulant (NOAC) such as edoxaban, Christina L. Fanola, MD, said at the annual congress of the European Society of Cardiology.
“It’s a great time to think about this type of score, because so many more patients are being diagnosed with atrial fibrillation and there is a lot of clinical equipoise” over which anticoagulant to start patients on, said Dr. Fanola, a cardiologist at Brigham and Women’s Hospital in Boston. She said she and her associates hope to externally validate the score and test it in cohorts that received other NOACs, such as apixaban (Eliquis), dabigatran (Pradaxa), or rivaroxaban (Xarelto), but it is very possible that scoring might differ from one NOAC to the next. “Each NOAC may need its own scoring formula,” Dr. Fanola said in an interview.
A Cox proportional hazards model identified 10 demographic, clinical, and laboratory features that had significant, independent correlations to a primary outcome of disabling stroke, life-threatening bleeding, or death. After weighing the point allocation for each item by the strength of its association, the researchers developed a scoring formula in a model that could account for about 69% of the three combined adverse outcomes.
An analysis that applied the scoring formula back to the ENGAGE AF-TIMI 48 database showed that a low-risk score of 0-6 correlated with a 4% per year rate of disabling stroke, life-threatening bleed, or death; an intermediate-risk score of 7-9 correlated with a 10% per year incidence of this combined outcome, and a high-risk score of 10 or greater linked with a 21% annual event rate.
Dr. Fanola and her associates ran a further analysis that evaluated the efficacy of edoxaban, compared with warfarin, among the patients in each of these risk strata. The high-risk patients received a major benefit from edoxaban, with a 30% overall incidence of the combined endpoint during 3 years of follow-up, compared with a 51% rate among patients on warfarin, a 21-percentage-point reduction in adverse events. Intermediate-risk patients also received a significant benefit, with a 26% event rate on warfarin and an 18% rate on edoxaban. But low-risk patients had identical 10% event rates with either treatment.
These findings suggest that atrial fibrillation patients with a TIMI AF score that is high or intermediate would have a better chance for a good outcome on edoxaban, or perhaps a different NOAC, than on warfarin. Low-risk patients seem to have similar outcomes on edoxaban or warfarin, so other considerations can come into play for choosing between these drug options, such as the cost of treatment and the inconvenience of regular warfarin monitoring, Dr. Fanola said.
ENGAGE AF-TIMI 48 was sponsored by Daiichi Sankyo, the company that markets edoxaban. Dr. Fanola had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: Among high-risk patients, edoxaban cut adverse events by 21 percentage points, compared with warfarin.
Data source: ENGAGE AF-TIMI 48, a multicenter trial with 21,105 patients.
Disclosures: ENGAGE AF-TIMI 48 was sponsored by Daiichi Sankyo, the company that markets edoxaban (Savaysa). Dr. Fanola had no relevant financial disclosures.
Heart failure targets African Americans
ORLANDO – The disparity in U.S. heart failure incidence continued undiminished during 2002-2013, with African Americans maintaining a steady 2.3-fold increased rate of heart failure, compared with whites, based on national levels of heart failure hospitalizations, a reasonable surrogate for incidence rates, Boback Ziaeian, MD, reported at the annual scientific meeting of the Heart Failure Society of America.
The same period also showed a substantial relative improvement in the heart failure hospitalization rates among U.S. Hispanics, compared with whites, so that, by 2013, the ethnic disparity seen in 2002 between Hispanics and whites largely disappeared, reported Dr. Ziaeian, a cardiologist at the University of California, Los Angeles. The data he analyzed also showed that Asian Americans had the lowest heart failure hospitalization rates of any racial or ethnic group throughout the 11-year period, and that the incidence of heart failure fell more sharply in women than in men during the period, based on the hospitalization numbers.
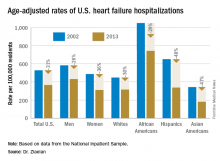
Age-adjusted heart failure hospitalizations among whites dropped by 30%, and among African Americans by a nearly identical 29%. But this maintained a greater than twofold disparity in rates between the two groups. Among whites, the rate per 100,000 fell from 448 to 315; among African Americans, it dropped from 1,048 to 741. In 2013, the rate of heart failure hospitalizations was 2.4-fold higher in African Americans, compared with whites.
Heart failure hospitalizations fell among Hispanics from 650 per 100,000 to 337 per 100,000 in 2013, a 48% drop that brought the rate among Hispanics to nearly the same as among whites. Asian Americans remained the group with the least heart failure throughout the period, falling from 343 hospitalizations per 100,000 in 2002 to 181 per 100,000 in 2013, a 47% drop.
Among women, the age-adjusted rate per 100,000 fell from 486 to 311, a 36% drop, compared with a decrease from 582 to 431 per 100,000 in men, a 26% reduction. Lower incidence in women may reflect better risk factor control during the study period, compared with men, such as a higher rate of quiting smoking and better treatment compliance, Dr. Ziaeian suggested.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – The disparity in U.S. heart failure incidence continued undiminished during 2002-2013, with African Americans maintaining a steady 2.3-fold increased rate of heart failure, compared with whites, based on national levels of heart failure hospitalizations, a reasonable surrogate for incidence rates, Boback Ziaeian, MD, reported at the annual scientific meeting of the Heart Failure Society of America.
The same period also showed a substantial relative improvement in the heart failure hospitalization rates among U.S. Hispanics, compared with whites, so that, by 2013, the ethnic disparity seen in 2002 between Hispanics and whites largely disappeared, reported Dr. Ziaeian, a cardiologist at the University of California, Los Angeles. The data he analyzed also showed that Asian Americans had the lowest heart failure hospitalization rates of any racial or ethnic group throughout the 11-year period, and that the incidence of heart failure fell more sharply in women than in men during the period, based on the hospitalization numbers.
Age-adjusted heart failure hospitalizations among whites dropped by 30%, and among African Americans by a nearly identical 29%. But this maintained a greater than twofold disparity in rates between the two groups. Among whites, the rate per 100,000 fell from 448 to 315; among African Americans, it dropped from 1,048 to 741. In 2013, the rate of heart failure hospitalizations was 2.4-fold higher in African Americans, compared with whites.
Heart failure hospitalizations fell among Hispanics from 650 per 100,000 to 337 per 100,000 in 2013, a 48% drop that brought the rate among Hispanics to nearly the same as among whites. Asian Americans remained the group with the least heart failure throughout the period, falling from 343 hospitalizations per 100,000 in 2002 to 181 per 100,000 in 2013, a 47% drop.
Among women, the age-adjusted rate per 100,000 fell from 486 to 311, a 36% drop, compared with a decrease from 582 to 431 per 100,000 in men, a 26% reduction. Lower incidence in women may reflect better risk factor control during the study period, compared with men, such as a higher rate of quiting smoking and better treatment compliance, Dr. Ziaeian suggested.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – The disparity in U.S. heart failure incidence continued undiminished during 2002-2013, with African Americans maintaining a steady 2.3-fold increased rate of heart failure, compared with whites, based on national levels of heart failure hospitalizations, a reasonable surrogate for incidence rates, Boback Ziaeian, MD, reported at the annual scientific meeting of the Heart Failure Society of America.
The same period also showed a substantial relative improvement in the heart failure hospitalization rates among U.S. Hispanics, compared with whites, so that, by 2013, the ethnic disparity seen in 2002 between Hispanics and whites largely disappeared, reported Dr. Ziaeian, a cardiologist at the University of California, Los Angeles. The data he analyzed also showed that Asian Americans had the lowest heart failure hospitalization rates of any racial or ethnic group throughout the 11-year period, and that the incidence of heart failure fell more sharply in women than in men during the period, based on the hospitalization numbers.
Age-adjusted heart failure hospitalizations among whites dropped by 30%, and among African Americans by a nearly identical 29%. But this maintained a greater than twofold disparity in rates between the two groups. Among whites, the rate per 100,000 fell from 448 to 315; among African Americans, it dropped from 1,048 to 741. In 2013, the rate of heart failure hospitalizations was 2.4-fold higher in African Americans, compared with whites.
Heart failure hospitalizations fell among Hispanics from 650 per 100,000 to 337 per 100,000 in 2013, a 48% drop that brought the rate among Hispanics to nearly the same as among whites. Asian Americans remained the group with the least heart failure throughout the period, falling from 343 hospitalizations per 100,000 in 2002 to 181 per 100,000 in 2013, a 47% drop.
Among women, the age-adjusted rate per 100,000 fell from 486 to 311, a 36% drop, compared with a decrease from 582 to 431 per 100,000 in men, a 26% reduction. Lower incidence in women may reflect better risk factor control during the study period, compared with men, such as a higher rate of quiting smoking and better treatment compliance, Dr. Ziaeian suggested.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Key clinical point:
Major finding: In 2013, age-adjusted heart failure hospitalization was 741/100,000 in African Americans and 315/100,000 in whites.
Data source: The National Inpatient Sample and U.S. Census data.
Disclosures: Dr. Ziaeian had no disclosures.
VIDEO: Two PCI vs. CABG trials produce conflicting results
WASHINGTON – Results from two large, multicenter comparisons of coronary stenting and coronary bypass surgery for treating patients with unprotected left main coronary disease may have superficially shown sharp differences, but the bottom line will likely be greater empowerment of percutaneous coronary intervention as an option for selected patients with less complex coronary disease.
Prior to the results from the EXCEL and NOBLE trials, reported at the Transcatheter Cardiovascular Therapeutics annual meeting, “guidelines put PCI [percutaneous coronary intervention] into a class 1, 2 or 3 status for treating left main coronary disease depending on disease complexity, but in the United States, PCI for patients eligible for CABG [coronary artery bypass grafting] has not been frequently done. I think these results, in a very circumscribed subset of patients and using a state-of-the-art stent, will affect the guidelines,” predicted Gregg W. Stone, MD, lead investigator for EXCEL and professor of medicine at Columbia University in New York.
“What the guidelines have not addressed are the patients with low- or intermediate-complexity disease who have an acceptable risk for undergoing either PCI or CABG. I think the trial results answer this question,” said David Kandzari, MD, director of interventional cardiology and chief scientific officer at the Piedmont Heart Institute in Atlanta and an EXCEL investigator.
While the EXCEL and NOBLE results don’t provide a simple answer on the relative merits of PCI and CABG, many of their outcome differences seem explicable, several experts said at the meeting.
The Nordic-Baltic-British Left Main Revascularisation (NOBLE) trial randomized 1,201 patients who had unprotected left main coronary disease and were judged by a heart team to be reasonable candidates for both PCI or CABG at 36 centers in nine European countries during 2008-2015. The primary endpoint was death from any cause, nonprocedural MIs, stroke, or repeat revascularization.
The researchers followed patients for a median of just over 3 years, but they calculated the primary endpoint based on a Kaplan-Meier estimate for 5-year outcomes, which showed the primary endpoint in 29% of the PCI patients and in 19% of the CABG patients, a statistically significant benefit in favor of CABG, Evald H. Christiansen, MD, reported at the meeting, which was sponsored by the Cardiovascular Research Foundation. Concurrently with his report, the results were published online (Lancet. 2016 Oct. 31. doi: 10.1016/S0140-6736[16]32052-9).
This difference between PCI and CABG was largely driven by an excess of postprocedural MIs and repeat revascularizations among the PCI patients, said Dr. Christiansen, an interventional cardiologist at Aarhus University Hospital in Denmark. Another notable finding was that the superior outcomes with CABG primarily occurred among patients with a SYNTAX score – a measure of coronary disease complexity – of 22 or less, which identifies patients with low complexity disease. The outcomes of patients with SYNTAX scores of 23-32, which identifies intermediate complexity disease, and of patients with scores of 33 or higher, with very complex disease, were similar in the PCI and CABG arms, he reported. This finding was “very surprising,” Dr. Christiansen said, because it reversed the finding originally made in the SYNTAX trial that PCI performed best compared with CABG in patients with the lowest scores and least-complex coronary disease.
The superiority of CABG over PCI seen in the NOBLE results, especially in patients with lower SYNTAX scores, seemed at odds with the EXCEL results, reported at the meeting by Dr. Stone and simultaneously online (N Engl J Med. 2016 Oct. 31. doi: 10.1056/NEJMoa1610227). In EXCEL, which enrolled only patients with a SYNTAX score of 32 or less (low- or intermediate-complexity coronary disease), patients had a 3-year incidence of death, stroke or MI of 15% in both the PCI and CABG arms.
But the EXCEL and NOBLE trials had several important differences, and it seemed like cumulatively these differences account for their differing results.
“One of the biggest differences” was the exclusion of procedural MIs in the NOBLE tally of adverse events, noted Dr. Stone. These were diagnosed in EXCEL using the MI definition published in 2013 by a panel of the Society for Cardiovascular Angiography and Interventions (SCAI). NOBLE disregarded procedural MIs because many of its participating hospitals did not have the laboratory resources to make these diagnoses and because the trial’s design predated the SCAI definition by several years, Dr. Christiansen explained.
Other important differences included the shorter follow-up in EXCEL, the inclusion of revascularization as an endpoint component in NOBLE but not in EXCEL, and differences in the stents used. In EXCEL, all patients undergoing PCI received Xience everolimus-eluting stents. In NOBLE, the first 11% of the enrolled patients received first-generation, sirolimus-eluting Cypher stents; the next 89% of enrollees received a biolimus-eluting Biomatrix Flex stent. Dr. Christiansen acknowledged the confounding caused by having many patients in the NOBLE PCI arm who received outmoded Cypher stents, especially because their relatively longer follow-up made them overrepresented in the primary outcome results. Plus, the Biomatrix Flex stent was disparaged by Martin B. Leon, MD, an EXCEL investigator and professor of medicine at Columbia University, who called the device “not currently widely used for PCI and more of historic interest.”
Dr. Leon added that the EXCEL and NOBLE patients also had substantially different prevalence rates of diabetes and acute coronary syndrome.
“The huge difference [between EXCEL and NOBLE] is the endpoint,” declared Marc Ruel, MD, another EXCEL investigator and head of cardiac surgery at the Ottawa Heart Institute. “The EXCEL endpoint was driven by the high rate of periprocedural MIs in the CABG arm. Once you get past 30 days, the noninferiority is not met by PCI.”
Another big endpoint difference was leaving revascularizations out of the EXCEL composite. “Once you take revascularization out of the primary endpoint, the outcome [of EXCEL] was more or less preordained,” noted Craig R. Smith, MD, chairman of surgery at Columbia University and an EXCEL investigator. “It’s the slope of events [in the PCI arm] after 3 months that’s the story. I think the CABG and PCI slopes in EXCEL will continue to diverge with time” beyond the current 3-year follow-up, Dr. Smith said.
“I agree that after 30 days surgery was the more durable procedure,” said Dr. Stone. “There is a big upfront hit for patients to take with surgery compared with PCI. If patients get through that, then they have a more durable procedure. That’s the trade-off.”
Dr. Stone hinted that future reports of EXCEL data will highlight other hits that patients must endure upfront if they choose CABG over PCI. “The early difference was quite profound not only for the primary endpoint but also for renal failure, infections, arrhythmias, and blood transfusions,” he said. Choosing between PCI and CABG for patients with left main disease and a lower SYNTAX score “is a decision that should be made by the heart team and patients. Some patients will prefer surgery, and some will prefer PCI.”
The NOBLE trial received partial funding from Biosensors, the company that markets the Biomatrix Flex stent used in the trial. The EXCEL trial was sponsored by Abbott Vascular, the company that markets the Xience stent used in the trial. Dr. Stone, Dr. Kandzari, Dr. Christiansen, Dr. Ruel, and Dr. Smith had no disclosures. Dr. Leon has been a consultant to and received research support from Abbott Vascular and Boston Scientific and has also received research support from Edwards, Medtronic and St. Jude.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
The results from EXCEL and NOBLE were not that different, but what was different was how the two trials were designed and how their endpoints were defined. The biggest difference between percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG) seemed to be in the rate of late MIs, with a little higher rate following PCI, and more repeat revascularizations with PCI, but with similar mortality rates with both treatments. There was a lot of similarity in the results despite the differences in the trials.
The evidence in both studies gives more support to the concept that, for patients with simpler left main lesions, PCI is a competitive alternative to CABG. Until now, in U.S. practice patients with left main coronary disease have been preferentially referred for CABG. These results will open us up to giving selected patients a more balanced view of the two options. After we explain differences in recovery and late events patients can decide which treatment to receive.
Despite these new findings, PCI is still not for every patient. A substantial fraction of patients with left main coronary disease were excluded from these studies because they had more complex coronary anatomy, and for patients like that, CABG remains the clear standard of care.
David J. Cohen, MD, is director of cardiovascular research and an interventional cardiologist at Saint Luke’s Health System in Kansas City, Mo. He made these comments in a video interview. He had received research support from Abbott Vascular, and is an investigator in the EXCEL trial.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The results from EXCEL and NOBLE were not that different, but what was different was how the two trials were designed and how their endpoints were defined. The biggest difference between percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG) seemed to be in the rate of late MIs, with a little higher rate following PCI, and more repeat revascularizations with PCI, but with similar mortality rates with both treatments. There was a lot of similarity in the results despite the differences in the trials.
The evidence in both studies gives more support to the concept that, for patients with simpler left main lesions, PCI is a competitive alternative to CABG. Until now, in U.S. practice patients with left main coronary disease have been preferentially referred for CABG. These results will open us up to giving selected patients a more balanced view of the two options. After we explain differences in recovery and late events patients can decide which treatment to receive.
Despite these new findings, PCI is still not for every patient. A substantial fraction of patients with left main coronary disease were excluded from these studies because they had more complex coronary anatomy, and for patients like that, CABG remains the clear standard of care.
David J. Cohen, MD, is director of cardiovascular research and an interventional cardiologist at Saint Luke’s Health System in Kansas City, Mo. He made these comments in a video interview. He had received research support from Abbott Vascular, and is an investigator in the EXCEL trial.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
The results from EXCEL and NOBLE were not that different, but what was different was how the two trials were designed and how their endpoints were defined. The biggest difference between percutaneous coronary intervention (PCI) and coronary artery bypass grafting (CABG) seemed to be in the rate of late MIs, with a little higher rate following PCI, and more repeat revascularizations with PCI, but with similar mortality rates with both treatments. There was a lot of similarity in the results despite the differences in the trials.
The evidence in both studies gives more support to the concept that, for patients with simpler left main lesions, PCI is a competitive alternative to CABG. Until now, in U.S. practice patients with left main coronary disease have been preferentially referred for CABG. These results will open us up to giving selected patients a more balanced view of the two options. After we explain differences in recovery and late events patients can decide which treatment to receive.
Despite these new findings, PCI is still not for every patient. A substantial fraction of patients with left main coronary disease were excluded from these studies because they had more complex coronary anatomy, and for patients like that, CABG remains the clear standard of care.
David J. Cohen, MD, is director of cardiovascular research and an interventional cardiologist at Saint Luke’s Health System in Kansas City, Mo. He made these comments in a video interview. He had received research support from Abbott Vascular, and is an investigator in the EXCEL trial.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON – Results from two large, multicenter comparisons of coronary stenting and coronary bypass surgery for treating patients with unprotected left main coronary disease may have superficially shown sharp differences, but the bottom line will likely be greater empowerment of percutaneous coronary intervention as an option for selected patients with less complex coronary disease.
Prior to the results from the EXCEL and NOBLE trials, reported at the Transcatheter Cardiovascular Therapeutics annual meeting, “guidelines put PCI [percutaneous coronary intervention] into a class 1, 2 or 3 status for treating left main coronary disease depending on disease complexity, but in the United States, PCI for patients eligible for CABG [coronary artery bypass grafting] has not been frequently done. I think these results, in a very circumscribed subset of patients and using a state-of-the-art stent, will affect the guidelines,” predicted Gregg W. Stone, MD, lead investigator for EXCEL and professor of medicine at Columbia University in New York.
“What the guidelines have not addressed are the patients with low- or intermediate-complexity disease who have an acceptable risk for undergoing either PCI or CABG. I think the trial results answer this question,” said David Kandzari, MD, director of interventional cardiology and chief scientific officer at the Piedmont Heart Institute in Atlanta and an EXCEL investigator.
While the EXCEL and NOBLE results don’t provide a simple answer on the relative merits of PCI and CABG, many of their outcome differences seem explicable, several experts said at the meeting.
The Nordic-Baltic-British Left Main Revascularisation (NOBLE) trial randomized 1,201 patients who had unprotected left main coronary disease and were judged by a heart team to be reasonable candidates for both PCI or CABG at 36 centers in nine European countries during 2008-2015. The primary endpoint was death from any cause, nonprocedural MIs, stroke, or repeat revascularization.
The researchers followed patients for a median of just over 3 years, but they calculated the primary endpoint based on a Kaplan-Meier estimate for 5-year outcomes, which showed the primary endpoint in 29% of the PCI patients and in 19% of the CABG patients, a statistically significant benefit in favor of CABG, Evald H. Christiansen, MD, reported at the meeting, which was sponsored by the Cardiovascular Research Foundation. Concurrently with his report, the results were published online (Lancet. 2016 Oct. 31. doi: 10.1016/S0140-6736[16]32052-9).
This difference between PCI and CABG was largely driven by an excess of postprocedural MIs and repeat revascularizations among the PCI patients, said Dr. Christiansen, an interventional cardiologist at Aarhus University Hospital in Denmark. Another notable finding was that the superior outcomes with CABG primarily occurred among patients with a SYNTAX score – a measure of coronary disease complexity – of 22 or less, which identifies patients with low complexity disease. The outcomes of patients with SYNTAX scores of 23-32, which identifies intermediate complexity disease, and of patients with scores of 33 or higher, with very complex disease, were similar in the PCI and CABG arms, he reported. This finding was “very surprising,” Dr. Christiansen said, because it reversed the finding originally made in the SYNTAX trial that PCI performed best compared with CABG in patients with the lowest scores and least-complex coronary disease.
The superiority of CABG over PCI seen in the NOBLE results, especially in patients with lower SYNTAX scores, seemed at odds with the EXCEL results, reported at the meeting by Dr. Stone and simultaneously online (N Engl J Med. 2016 Oct. 31. doi: 10.1056/NEJMoa1610227). In EXCEL, which enrolled only patients with a SYNTAX score of 32 or less (low- or intermediate-complexity coronary disease), patients had a 3-year incidence of death, stroke or MI of 15% in both the PCI and CABG arms.
But the EXCEL and NOBLE trials had several important differences, and it seemed like cumulatively these differences account for their differing results.
“One of the biggest differences” was the exclusion of procedural MIs in the NOBLE tally of adverse events, noted Dr. Stone. These were diagnosed in EXCEL using the MI definition published in 2013 by a panel of the Society for Cardiovascular Angiography and Interventions (SCAI). NOBLE disregarded procedural MIs because many of its participating hospitals did not have the laboratory resources to make these diagnoses and because the trial’s design predated the SCAI definition by several years, Dr. Christiansen explained.
Other important differences included the shorter follow-up in EXCEL, the inclusion of revascularization as an endpoint component in NOBLE but not in EXCEL, and differences in the stents used. In EXCEL, all patients undergoing PCI received Xience everolimus-eluting stents. In NOBLE, the first 11% of the enrolled patients received first-generation, sirolimus-eluting Cypher stents; the next 89% of enrollees received a biolimus-eluting Biomatrix Flex stent. Dr. Christiansen acknowledged the confounding caused by having many patients in the NOBLE PCI arm who received outmoded Cypher stents, especially because their relatively longer follow-up made them overrepresented in the primary outcome results. Plus, the Biomatrix Flex stent was disparaged by Martin B. Leon, MD, an EXCEL investigator and professor of medicine at Columbia University, who called the device “not currently widely used for PCI and more of historic interest.”
Dr. Leon added that the EXCEL and NOBLE patients also had substantially different prevalence rates of diabetes and acute coronary syndrome.
“The huge difference [between EXCEL and NOBLE] is the endpoint,” declared Marc Ruel, MD, another EXCEL investigator and head of cardiac surgery at the Ottawa Heart Institute. “The EXCEL endpoint was driven by the high rate of periprocedural MIs in the CABG arm. Once you get past 30 days, the noninferiority is not met by PCI.”
Another big endpoint difference was leaving revascularizations out of the EXCEL composite. “Once you take revascularization out of the primary endpoint, the outcome [of EXCEL] was more or less preordained,” noted Craig R. Smith, MD, chairman of surgery at Columbia University and an EXCEL investigator. “It’s the slope of events [in the PCI arm] after 3 months that’s the story. I think the CABG and PCI slopes in EXCEL will continue to diverge with time” beyond the current 3-year follow-up, Dr. Smith said.
“I agree that after 30 days surgery was the more durable procedure,” said Dr. Stone. “There is a big upfront hit for patients to take with surgery compared with PCI. If patients get through that, then they have a more durable procedure. That’s the trade-off.”
Dr. Stone hinted that future reports of EXCEL data will highlight other hits that patients must endure upfront if they choose CABG over PCI. “The early difference was quite profound not only for the primary endpoint but also for renal failure, infections, arrhythmias, and blood transfusions,” he said. Choosing between PCI and CABG for patients with left main disease and a lower SYNTAX score “is a decision that should be made by the heart team and patients. Some patients will prefer surgery, and some will prefer PCI.”
The NOBLE trial received partial funding from Biosensors, the company that markets the Biomatrix Flex stent used in the trial. The EXCEL trial was sponsored by Abbott Vascular, the company that markets the Xience stent used in the trial. Dr. Stone, Dr. Kandzari, Dr. Christiansen, Dr. Ruel, and Dr. Smith had no disclosures. Dr. Leon has been a consultant to and received research support from Abbott Vascular and Boston Scientific and has also received research support from Edwards, Medtronic and St. Jude.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
WASHINGTON – Results from two large, multicenter comparisons of coronary stenting and coronary bypass surgery for treating patients with unprotected left main coronary disease may have superficially shown sharp differences, but the bottom line will likely be greater empowerment of percutaneous coronary intervention as an option for selected patients with less complex coronary disease.
Prior to the results from the EXCEL and NOBLE trials, reported at the Transcatheter Cardiovascular Therapeutics annual meeting, “guidelines put PCI [percutaneous coronary intervention] into a class 1, 2 or 3 status for treating left main coronary disease depending on disease complexity, but in the United States, PCI for patients eligible for CABG [coronary artery bypass grafting] has not been frequently done. I think these results, in a very circumscribed subset of patients and using a state-of-the-art stent, will affect the guidelines,” predicted Gregg W. Stone, MD, lead investigator for EXCEL and professor of medicine at Columbia University in New York.
“What the guidelines have not addressed are the patients with low- or intermediate-complexity disease who have an acceptable risk for undergoing either PCI or CABG. I think the trial results answer this question,” said David Kandzari, MD, director of interventional cardiology and chief scientific officer at the Piedmont Heart Institute in Atlanta and an EXCEL investigator.
While the EXCEL and NOBLE results don’t provide a simple answer on the relative merits of PCI and CABG, many of their outcome differences seem explicable, several experts said at the meeting.
The Nordic-Baltic-British Left Main Revascularisation (NOBLE) trial randomized 1,201 patients who had unprotected left main coronary disease and were judged by a heart team to be reasonable candidates for both PCI or CABG at 36 centers in nine European countries during 2008-2015. The primary endpoint was death from any cause, nonprocedural MIs, stroke, or repeat revascularization.
The researchers followed patients for a median of just over 3 years, but they calculated the primary endpoint based on a Kaplan-Meier estimate for 5-year outcomes, which showed the primary endpoint in 29% of the PCI patients and in 19% of the CABG patients, a statistically significant benefit in favor of CABG, Evald H. Christiansen, MD, reported at the meeting, which was sponsored by the Cardiovascular Research Foundation. Concurrently with his report, the results were published online (Lancet. 2016 Oct. 31. doi: 10.1016/S0140-6736[16]32052-9).
This difference between PCI and CABG was largely driven by an excess of postprocedural MIs and repeat revascularizations among the PCI patients, said Dr. Christiansen, an interventional cardiologist at Aarhus University Hospital in Denmark. Another notable finding was that the superior outcomes with CABG primarily occurred among patients with a SYNTAX score – a measure of coronary disease complexity – of 22 or less, which identifies patients with low complexity disease. The outcomes of patients with SYNTAX scores of 23-32, which identifies intermediate complexity disease, and of patients with scores of 33 or higher, with very complex disease, were similar in the PCI and CABG arms, he reported. This finding was “very surprising,” Dr. Christiansen said, because it reversed the finding originally made in the SYNTAX trial that PCI performed best compared with CABG in patients with the lowest scores and least-complex coronary disease.
The superiority of CABG over PCI seen in the NOBLE results, especially in patients with lower SYNTAX scores, seemed at odds with the EXCEL results, reported at the meeting by Dr. Stone and simultaneously online (N Engl J Med. 2016 Oct. 31. doi: 10.1056/NEJMoa1610227). In EXCEL, which enrolled only patients with a SYNTAX score of 32 or less (low- or intermediate-complexity coronary disease), patients had a 3-year incidence of death, stroke or MI of 15% in both the PCI and CABG arms.
But the EXCEL and NOBLE trials had several important differences, and it seemed like cumulatively these differences account for their differing results.
“One of the biggest differences” was the exclusion of procedural MIs in the NOBLE tally of adverse events, noted Dr. Stone. These were diagnosed in EXCEL using the MI definition published in 2013 by a panel of the Society for Cardiovascular Angiography and Interventions (SCAI). NOBLE disregarded procedural MIs because many of its participating hospitals did not have the laboratory resources to make these diagnoses and because the trial’s design predated the SCAI definition by several years, Dr. Christiansen explained.
Other important differences included the shorter follow-up in EXCEL, the inclusion of revascularization as an endpoint component in NOBLE but not in EXCEL, and differences in the stents used. In EXCEL, all patients undergoing PCI received Xience everolimus-eluting stents. In NOBLE, the first 11% of the enrolled patients received first-generation, sirolimus-eluting Cypher stents; the next 89% of enrollees received a biolimus-eluting Biomatrix Flex stent. Dr. Christiansen acknowledged the confounding caused by having many patients in the NOBLE PCI arm who received outmoded Cypher stents, especially because their relatively longer follow-up made them overrepresented in the primary outcome results. Plus, the Biomatrix Flex stent was disparaged by Martin B. Leon, MD, an EXCEL investigator and professor of medicine at Columbia University, who called the device “not currently widely used for PCI and more of historic interest.”
Dr. Leon added that the EXCEL and NOBLE patients also had substantially different prevalence rates of diabetes and acute coronary syndrome.
“The huge difference [between EXCEL and NOBLE] is the endpoint,” declared Marc Ruel, MD, another EXCEL investigator and head of cardiac surgery at the Ottawa Heart Institute. “The EXCEL endpoint was driven by the high rate of periprocedural MIs in the CABG arm. Once you get past 30 days, the noninferiority is not met by PCI.”
Another big endpoint difference was leaving revascularizations out of the EXCEL composite. “Once you take revascularization out of the primary endpoint, the outcome [of EXCEL] was more or less preordained,” noted Craig R. Smith, MD, chairman of surgery at Columbia University and an EXCEL investigator. “It’s the slope of events [in the PCI arm] after 3 months that’s the story. I think the CABG and PCI slopes in EXCEL will continue to diverge with time” beyond the current 3-year follow-up, Dr. Smith said.
“I agree that after 30 days surgery was the more durable procedure,” said Dr. Stone. “There is a big upfront hit for patients to take with surgery compared with PCI. If patients get through that, then they have a more durable procedure. That’s the trade-off.”
Dr. Stone hinted that future reports of EXCEL data will highlight other hits that patients must endure upfront if they choose CABG over PCI. “The early difference was quite profound not only for the primary endpoint but also for renal failure, infections, arrhythmias, and blood transfusions,” he said. Choosing between PCI and CABG for patients with left main disease and a lower SYNTAX score “is a decision that should be made by the heart team and patients. Some patients will prefer surgery, and some will prefer PCI.”
The NOBLE trial received partial funding from Biosensors, the company that markets the Biomatrix Flex stent used in the trial. The EXCEL trial was sponsored by Abbott Vascular, the company that markets the Xience stent used in the trial. Dr. Stone, Dr. Kandzari, Dr. Christiansen, Dr. Ruel, and Dr. Smith had no disclosures. Dr. Leon has been a consultant to and received research support from Abbott Vascular and Boston Scientific and has also received research support from Edwards, Medtronic and St. Jude.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM TCT 2016
Selected liver-transplant patients thrive off immunosuppression
MONTREAL – Three-fifths of pediatric liver-transplant recipients who were doing well enough to attempt weaning from their immunosuppression regimen succeeded in getting off immunosuppression and staying off for more than a year. In the process, they also significantly improved their health-related quality of life.
“Health-related quality of life domains associated with social interactions, worry, and medications improved” in pediatric liver recipients who had undergone immunosuppression withdrawal, Saeed Mohammad, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology and Nutrition.
Patients who succeeded in staying off immunosuppressant drugs for at least 2 years after they first began ratcheting down their regimen showed better quality of life scores compared with their scores at baseline, and also compared with the scores of other pediatric liver transplant patients who unsuccessfully tried coming off immunosuppression.
Not every pediatric liver transplant patient should attempt withdrawing immunosuppression, cautioned Dr. Mohammad, a pediatric gastroenterologist at Northwestern University in Chicago. “To be successful withdrawal of immunosuppression needs to be in selected patients; not every patient is a good candidate.”
The Immunosuppression Withdrawal for Stable Pediatric Liver Transplant Recipients (iWITH) study ran at 11 U.S. center and one center in Toronto during October 2012 through June 2014. Pediatric liver transplant recipients were eligible to start a 9-10 month graduated withdrawal from their immunosuppression regimen if they met several criteria of stability including no rejection episode over at least the prior 12 months, normal laboratory-test results, no autoimmune disease and no problems detected in a liver biopsy. The prospective study enrolled 88 patients who averaged 10 years old. Patients underwent comprehensive examinations and laboratory testing at baseline and again several times during the subsequent 2 years including assessment of several quality of life measures.
During follow-up, 35 of the 88 patients (40%) developed symptoms of rejection and had to go back on immunosuppression. Most of these patients developed their rejection symptoms early during immunosuppression weaning, but a few patients failed later including one patient who failed 22 months after starting immunosuppression withdrawal, Dr. Mohammad said. Researchers from the iWITH study first reported these results at the American Transplant Congress in June 2016.
The quality of life findings reported by Dr. Mohammad came from assessments at baseline, after 12 months, and after 24 months, and included 30 of the patients who resumed immunosuppression and 48 patients who remained off immunosuppression for 2 years. All of these 78 patients had relatively robust quality of life profiles at baseline. Their scores for both physical and social subscales as well as for total score were significantly superior to the average scores for a large number of primarily U.S. pediatric liver transplant patients in the SPLIT database. Dr. Mohammad called the patients who attempted immunosuppression discontinuation as the “creme de la creme” of pediatric liver transplant patients in terms of their clinical status.
Analysis of scores after 2 years compared with baseline showed statistically significant improvements among patients who stayed off immunosuppression for the domains of social function, treatment attitudes and compliance, communication, and worry. A comparison of changes in quality of life scores from baseline to 2 years showed that patients who stayed off immunosuppression had improvements in several of their scores while patients who went back onto immunosuppression had on average a small deterioration of their scores.
Dr. Mohammad had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – Three-fifths of pediatric liver-transplant recipients who were doing well enough to attempt weaning from their immunosuppression regimen succeeded in getting off immunosuppression and staying off for more than a year. In the process, they also significantly improved their health-related quality of life.
“Health-related quality of life domains associated with social interactions, worry, and medications improved” in pediatric liver recipients who had undergone immunosuppression withdrawal, Saeed Mohammad, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology and Nutrition.
Patients who succeeded in staying off immunosuppressant drugs for at least 2 years after they first began ratcheting down their regimen showed better quality of life scores compared with their scores at baseline, and also compared with the scores of other pediatric liver transplant patients who unsuccessfully tried coming off immunosuppression.
Not every pediatric liver transplant patient should attempt withdrawing immunosuppression, cautioned Dr. Mohammad, a pediatric gastroenterologist at Northwestern University in Chicago. “To be successful withdrawal of immunosuppression needs to be in selected patients; not every patient is a good candidate.”
The Immunosuppression Withdrawal for Stable Pediatric Liver Transplant Recipients (iWITH) study ran at 11 U.S. center and one center in Toronto during October 2012 through June 2014. Pediatric liver transplant recipients were eligible to start a 9-10 month graduated withdrawal from their immunosuppression regimen if they met several criteria of stability including no rejection episode over at least the prior 12 months, normal laboratory-test results, no autoimmune disease and no problems detected in a liver biopsy. The prospective study enrolled 88 patients who averaged 10 years old. Patients underwent comprehensive examinations and laboratory testing at baseline and again several times during the subsequent 2 years including assessment of several quality of life measures.
During follow-up, 35 of the 88 patients (40%) developed symptoms of rejection and had to go back on immunosuppression. Most of these patients developed their rejection symptoms early during immunosuppression weaning, but a few patients failed later including one patient who failed 22 months after starting immunosuppression withdrawal, Dr. Mohammad said. Researchers from the iWITH study first reported these results at the American Transplant Congress in June 2016.
The quality of life findings reported by Dr. Mohammad came from assessments at baseline, after 12 months, and after 24 months, and included 30 of the patients who resumed immunosuppression and 48 patients who remained off immunosuppression for 2 years. All of these 78 patients had relatively robust quality of life profiles at baseline. Their scores for both physical and social subscales as well as for total score were significantly superior to the average scores for a large number of primarily U.S. pediatric liver transplant patients in the SPLIT database. Dr. Mohammad called the patients who attempted immunosuppression discontinuation as the “creme de la creme” of pediatric liver transplant patients in terms of their clinical status.
Analysis of scores after 2 years compared with baseline showed statistically significant improvements among patients who stayed off immunosuppression for the domains of social function, treatment attitudes and compliance, communication, and worry. A comparison of changes in quality of life scores from baseline to 2 years showed that patients who stayed off immunosuppression had improvements in several of their scores while patients who went back onto immunosuppression had on average a small deterioration of their scores.
Dr. Mohammad had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – Three-fifths of pediatric liver-transplant recipients who were doing well enough to attempt weaning from their immunosuppression regimen succeeded in getting off immunosuppression and staying off for more than a year. In the process, they also significantly improved their health-related quality of life.
“Health-related quality of life domains associated with social interactions, worry, and medications improved” in pediatric liver recipients who had undergone immunosuppression withdrawal, Saeed Mohammad, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology and Nutrition.
Patients who succeeded in staying off immunosuppressant drugs for at least 2 years after they first began ratcheting down their regimen showed better quality of life scores compared with their scores at baseline, and also compared with the scores of other pediatric liver transplant patients who unsuccessfully tried coming off immunosuppression.
Not every pediatric liver transplant patient should attempt withdrawing immunosuppression, cautioned Dr. Mohammad, a pediatric gastroenterologist at Northwestern University in Chicago. “To be successful withdrawal of immunosuppression needs to be in selected patients; not every patient is a good candidate.”
The Immunosuppression Withdrawal for Stable Pediatric Liver Transplant Recipients (iWITH) study ran at 11 U.S. center and one center in Toronto during October 2012 through June 2014. Pediatric liver transplant recipients were eligible to start a 9-10 month graduated withdrawal from their immunosuppression regimen if they met several criteria of stability including no rejection episode over at least the prior 12 months, normal laboratory-test results, no autoimmune disease and no problems detected in a liver biopsy. The prospective study enrolled 88 patients who averaged 10 years old. Patients underwent comprehensive examinations and laboratory testing at baseline and again several times during the subsequent 2 years including assessment of several quality of life measures.
During follow-up, 35 of the 88 patients (40%) developed symptoms of rejection and had to go back on immunosuppression. Most of these patients developed their rejection symptoms early during immunosuppression weaning, but a few patients failed later including one patient who failed 22 months after starting immunosuppression withdrawal, Dr. Mohammad said. Researchers from the iWITH study first reported these results at the American Transplant Congress in June 2016.
The quality of life findings reported by Dr. Mohammad came from assessments at baseline, after 12 months, and after 24 months, and included 30 of the patients who resumed immunosuppression and 48 patients who remained off immunosuppression for 2 years. All of these 78 patients had relatively robust quality of life profiles at baseline. Their scores for both physical and social subscales as well as for total score were significantly superior to the average scores for a large number of primarily U.S. pediatric liver transplant patients in the SPLIT database. Dr. Mohammad called the patients who attempted immunosuppression discontinuation as the “creme de la creme” of pediatric liver transplant patients in terms of their clinical status.
Analysis of scores after 2 years compared with baseline showed statistically significant improvements among patients who stayed off immunosuppression for the domains of social function, treatment attitudes and compliance, communication, and worry. A comparison of changes in quality of life scores from baseline to 2 years showed that patients who stayed off immunosuppression had improvements in several of their scores while patients who went back onto immunosuppression had on average a small deterioration of their scores.
Dr. Mohammad had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT WCPGHAN 2016
Key clinical point: Selected pediatric liver-transplant patients who successfully weaned off immunosuppression responded with significantly improved quality of life scores.
Major finding: Patient and parent treatment satisfaction improved by 6-7 points when patients stopped immunosuppression and fell by 2-3 points when they did not.
Data source: iWISH, a multicenter study with 88 enrolled patients.
Disclosures: Dr. Mohammad had no disclosures.
Causes of recurrent pediatric pancreatitis start to emerge
MONTREAL – Once children have a first bout of acute pancreatitis, a second, separate episode of acute pancreatitis most often occurs in patients with genetically triggered pancreatitis, those who are taller or weigh more than average, and patients with pancreatic necrosis, based on multicenter, prospective data collected from 83 patients.
This is the first reported study to prospectively follow pediatric cases of acute pancreatitis, and additional studies with more patients are needed to better identify the factors predisposing patients to recurrent episodes of acute pancreatitis and to quantify the amount of risk these factors pose, Katherine F. Sweeny, MD, said at the annual meeting of the Federation of the International Societies of Pediatric Gastroenterology, Hepatology, and Nutrition.
The analysis focused on the 83 patients with at least 3 months of follow-up. During observation, 17 (20%) of the patients developed a second episode of acute pancreatitis that was distinguished from the initial episode by either at least 1 pain-free month or by complete normalization of amylase and lipase levels between the two episodes. Thirteen of the 17 recurrences occurred within 5 months of the first episode, with 11 of these occurring within the first 3 months after the first attack, a subgroup Dr. Sweeny called the “rapid progressors.”
Comparison of the 11 rapid progressors with the other 72 patients showed that the rapid progressors were significantly taller and weighed more. In addition, two of the 11 rapid progressors had pancreatic necrosis while none of the other patients had this complication.
The pancreatitis etiologies of the 11 rapid progressors also highlighted the potent influence a mutation can have on producing recurrent acute pancreatitis. Four of the 11 rapid progressors had a genetic mutation linked to pancreatitis susceptibility, and five of the six patients with a genetic cause for their index episode of pancreatitis developed a second acute episode during follow-up, said Dr. Sweeny, a pediatrician at Cincinnati Children’s Hospital Medical Center. In contrast, the next most effective cause of recurrent pancreatitis was a toxin or drug, which resulted in about a 25% incidence rate of a second episode. All of the other pancreatitis etiologies had recurrence rates of 10% or less.
Collecting better information on the causes of recurrent pancreatitis and chronic pancreatitis is especially important because of the rising incidence of acute pediatric pancreatitis, currently about one case in every 10,000 children and adolescents. Prior to formation of the INSPPIRE consortium, studies of pediatric pancreatitis had largely been limited to single-center retrospective reviews. The limitations of these data have made it hard to predict which patients with a first episode of acute pancreatitis will progress to a second episode or beyond, Dr. Sweeny said.
Dr. Sweeny had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – Once children have a first bout of acute pancreatitis, a second, separate episode of acute pancreatitis most often occurs in patients with genetically triggered pancreatitis, those who are taller or weigh more than average, and patients with pancreatic necrosis, based on multicenter, prospective data collected from 83 patients.
This is the first reported study to prospectively follow pediatric cases of acute pancreatitis, and additional studies with more patients are needed to better identify the factors predisposing patients to recurrent episodes of acute pancreatitis and to quantify the amount of risk these factors pose, Katherine F. Sweeny, MD, said at the annual meeting of the Federation of the International Societies of Pediatric Gastroenterology, Hepatology, and Nutrition.
The analysis focused on the 83 patients with at least 3 months of follow-up. During observation, 17 (20%) of the patients developed a second episode of acute pancreatitis that was distinguished from the initial episode by either at least 1 pain-free month or by complete normalization of amylase and lipase levels between the two episodes. Thirteen of the 17 recurrences occurred within 5 months of the first episode, with 11 of these occurring within the first 3 months after the first attack, a subgroup Dr. Sweeny called the “rapid progressors.”
Comparison of the 11 rapid progressors with the other 72 patients showed that the rapid progressors were significantly taller and weighed more. In addition, two of the 11 rapid progressors had pancreatic necrosis while none of the other patients had this complication.
The pancreatitis etiologies of the 11 rapid progressors also highlighted the potent influence a mutation can have on producing recurrent acute pancreatitis. Four of the 11 rapid progressors had a genetic mutation linked to pancreatitis susceptibility, and five of the six patients with a genetic cause for their index episode of pancreatitis developed a second acute episode during follow-up, said Dr. Sweeny, a pediatrician at Cincinnati Children’s Hospital Medical Center. In contrast, the next most effective cause of recurrent pancreatitis was a toxin or drug, which resulted in about a 25% incidence rate of a second episode. All of the other pancreatitis etiologies had recurrence rates of 10% or less.
Collecting better information on the causes of recurrent pancreatitis and chronic pancreatitis is especially important because of the rising incidence of acute pediatric pancreatitis, currently about one case in every 10,000 children and adolescents. Prior to formation of the INSPPIRE consortium, studies of pediatric pancreatitis had largely been limited to single-center retrospective reviews. The limitations of these data have made it hard to predict which patients with a first episode of acute pancreatitis will progress to a second episode or beyond, Dr. Sweeny said.
Dr. Sweeny had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – Once children have a first bout of acute pancreatitis, a second, separate episode of acute pancreatitis most often occurs in patients with genetically triggered pancreatitis, those who are taller or weigh more than average, and patients with pancreatic necrosis, based on multicenter, prospective data collected from 83 patients.
This is the first reported study to prospectively follow pediatric cases of acute pancreatitis, and additional studies with more patients are needed to better identify the factors predisposing patients to recurrent episodes of acute pancreatitis and to quantify the amount of risk these factors pose, Katherine F. Sweeny, MD, said at the annual meeting of the Federation of the International Societies of Pediatric Gastroenterology, Hepatology, and Nutrition.
The analysis focused on the 83 patients with at least 3 months of follow-up. During observation, 17 (20%) of the patients developed a second episode of acute pancreatitis that was distinguished from the initial episode by either at least 1 pain-free month or by complete normalization of amylase and lipase levels between the two episodes. Thirteen of the 17 recurrences occurred within 5 months of the first episode, with 11 of these occurring within the first 3 months after the first attack, a subgroup Dr. Sweeny called the “rapid progressors.”
Comparison of the 11 rapid progressors with the other 72 patients showed that the rapid progressors were significantly taller and weighed more. In addition, two of the 11 rapid progressors had pancreatic necrosis while none of the other patients had this complication.
The pancreatitis etiologies of the 11 rapid progressors also highlighted the potent influence a mutation can have on producing recurrent acute pancreatitis. Four of the 11 rapid progressors had a genetic mutation linked to pancreatitis susceptibility, and five of the six patients with a genetic cause for their index episode of pancreatitis developed a second acute episode during follow-up, said Dr. Sweeny, a pediatrician at Cincinnati Children’s Hospital Medical Center. In contrast, the next most effective cause of recurrent pancreatitis was a toxin or drug, which resulted in about a 25% incidence rate of a second episode. All of the other pancreatitis etiologies had recurrence rates of 10% or less.
Collecting better information on the causes of recurrent pancreatitis and chronic pancreatitis is especially important because of the rising incidence of acute pediatric pancreatitis, currently about one case in every 10,000 children and adolescents. Prior to formation of the INSPPIRE consortium, studies of pediatric pancreatitis had largely been limited to single-center retrospective reviews. The limitations of these data have made it hard to predict which patients with a first episode of acute pancreatitis will progress to a second episode or beyond, Dr. Sweeny said.
Dr. Sweeny had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT WCPGHAN 2016
Key clinical point:
Major finding: Overall, 17 of 83 patients (20%) had recurrent acute pancreatitis, but among six patients with a genetic cause, five had recurrences.
Data source: Eighty-three patients enrolled in INSPPIRE, an international consortium formed to prospectively study pediatric pancreatitis.
Disclosures: Dr. Sweeny had no disclosures.
VIDEO: Bioresorbable Absorb unexpectedly humbled by metallic DES
WASHINGTON – The bioabsorbable vascular scaffold bubble suddenly burst with the first 3-year follow-up data from a randomized trial that unexpectedly showed that the Absorb device significantly underperformed compared with Xience, a widely-used, second-generation metallic drug-eluting stent.
“As a pioneer of BVS [bioresorbable vascular scaffold] I’m disappointed,” Patrick W. Serruys, MD, said at the Transcatheter Cardiovascular Therapeutics annual meeting. “The performance of the comparator stent was spectacular.”
Xience surpassed Absorb in several other secondary endpoints. For example, the in-device binary restenosis rate was 7.0% with Absorb and 0.7% with Xience; the in-segment binary restenosis rate was 8% with Absorb and 3% with Xience. Target-vessel MIs occurred in 7% of the Absorb patients and 1% of the Xience patients, while clinically indicated target-lesion revascularization occurred in 6% of the Absorb patients and 1% of the Xience patients.
Another notable finding was that definite or probable in-device thrombosis occurred in nine Absorb patents and in none of the Xience patients, a statistically significant difference. Six of the Absorb thrombotic events occurred more than 1 year after the device was placed, and in several instances these thromboses occurred more than 900 days after placement, when the BVS had largely resorbed.
“These thromboses are occurring at the late stages of BVS degradation,” Dr. Serruys noted. “The Absorb polymer is basically gone after 3 years, but it’s replaced by a proteoglycan, and some proteoglycans are quite thrombogenic,” a possible explanation for the “mysterious” very late thromboses, he said.
These “disappointing” results my be linked to inadequate lesion preparation, appropriate sizing of the BVS for the lesion, and inconsistent postdilatation of the BVS, three steps that became the guiding mantra for BVS use starting a couple of years ago, said Giulio G. Stefanini, MD, an interventional cardiologist at Humanitas Research Hospital in Milan and a discussant for the report at the meeting, which was sponsored by the Cardiovascular Research Foundation.
Dr. Stefanini said that even though the Absorb stent became available for routine use in Europe starting in 2012, the device gained little traction since then in his own practice and throughout Italy. Currently it’s used for fewer than 5% of coronary interventions in Italy, he estimated. That’s largely because “we have failed to identify a population that benefits.” Other issues include the extra time needed to place a BVS, and the need for longer treatment with dual antiplatelet therapy for patients who receive a BVS, compared with when they receive a modern metallic drug-eluting stent. The Absorb BVS received Food and Drug Administration approval for routine U.S. use in July 2016.
“It would be beautiful to have a fully bioresorbable stent. It’s a lovely concept, but we’re not there yet,” Dr. Stefanini observed.
ABSORB II was sponsored by Abbott Vascular, which markets the Absorb device. Dr. Serruys has received research support from Abbott Vascular and has been a consultant to several other device and drug companies. Dr. Stefanini has been a consultant to Boston Scientific, B.Braun, and Edwards.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
We were all disappointed by the ABSORB II 3-year results. It was really surprising that even the vasomotion endpoint was, if anything, a little better with Xience, which performed amazingly well in this trial. Both arms of the study did well out to 3 years, but the Xience patients did better.
The Absorb bioresorbable vascular scaffold (BVS) is an early-stage device, and based on these results I wouldn’t give up on the BVS concept. But we need to be very careful with Absorb and which patients we implant with it. We need to make sure we carefully use thorough lesion preparation, correct sizing, and postdilatation in every patient, and we need to carefully select the right patients.
The main issue with the Absorb BVS in this trial was scaffold thrombosis, so we need to use a BVS only in patients with the lowest thrombosis risk, which means younger patients without renal failure, calcified vessels, and a larger-diameter target coronary artery. Younger patients have the most to gain from receiving a BVS. Younger patients who need a coronary intervention often collect several stents over the balance of their life, and it’s in these patients where you’d prefer that the stents eventually disappear.
Paul S. Teirstein, MD , is chief of cardiology and director of interventional cardiology at the Scripps Clinic in La Jolla, Calif. He has received research support from and has been a consultant to Abbott Vascular, Boston Scientific, and Medtronic. He made these comments in an interview .
We were all disappointed by the ABSORB II 3-year results. It was really surprising that even the vasomotion endpoint was, if anything, a little better with Xience, which performed amazingly well in this trial. Both arms of the study did well out to 3 years, but the Xience patients did better.
The Absorb bioresorbable vascular scaffold (BVS) is an early-stage device, and based on these results I wouldn’t give up on the BVS concept. But we need to be very careful with Absorb and which patients we implant with it. We need to make sure we carefully use thorough lesion preparation, correct sizing, and postdilatation in every patient, and we need to carefully select the right patients.
The main issue with the Absorb BVS in this trial was scaffold thrombosis, so we need to use a BVS only in patients with the lowest thrombosis risk, which means younger patients without renal failure, calcified vessels, and a larger-diameter target coronary artery. Younger patients have the most to gain from receiving a BVS. Younger patients who need a coronary intervention often collect several stents over the balance of their life, and it’s in these patients where you’d prefer that the stents eventually disappear.
Paul S. Teirstein, MD , is chief of cardiology and director of interventional cardiology at the Scripps Clinic in La Jolla, Calif. He has received research support from and has been a consultant to Abbott Vascular, Boston Scientific, and Medtronic. He made these comments in an interview .
We were all disappointed by the ABSORB II 3-year results. It was really surprising that even the vasomotion endpoint was, if anything, a little better with Xience, which performed amazingly well in this trial. Both arms of the study did well out to 3 years, but the Xience patients did better.
The Absorb bioresorbable vascular scaffold (BVS) is an early-stage device, and based on these results I wouldn’t give up on the BVS concept. But we need to be very careful with Absorb and which patients we implant with it. We need to make sure we carefully use thorough lesion preparation, correct sizing, and postdilatation in every patient, and we need to carefully select the right patients.
The main issue with the Absorb BVS in this trial was scaffold thrombosis, so we need to use a BVS only in patients with the lowest thrombosis risk, which means younger patients without renal failure, calcified vessels, and a larger-diameter target coronary artery. Younger patients have the most to gain from receiving a BVS. Younger patients who need a coronary intervention often collect several stents over the balance of their life, and it’s in these patients where you’d prefer that the stents eventually disappear.
Paul S. Teirstein, MD , is chief of cardiology and director of interventional cardiology at the Scripps Clinic in La Jolla, Calif. He has received research support from and has been a consultant to Abbott Vascular, Boston Scientific, and Medtronic. He made these comments in an interview .
WASHINGTON – The bioabsorbable vascular scaffold bubble suddenly burst with the first 3-year follow-up data from a randomized trial that unexpectedly showed that the Absorb device significantly underperformed compared with Xience, a widely-used, second-generation metallic drug-eluting stent.
“As a pioneer of BVS [bioresorbable vascular scaffold] I’m disappointed,” Patrick W. Serruys, MD, said at the Transcatheter Cardiovascular Therapeutics annual meeting. “The performance of the comparator stent was spectacular.”
Xience surpassed Absorb in several other secondary endpoints. For example, the in-device binary restenosis rate was 7.0% with Absorb and 0.7% with Xience; the in-segment binary restenosis rate was 8% with Absorb and 3% with Xience. Target-vessel MIs occurred in 7% of the Absorb patients and 1% of the Xience patients, while clinically indicated target-lesion revascularization occurred in 6% of the Absorb patients and 1% of the Xience patients.
Another notable finding was that definite or probable in-device thrombosis occurred in nine Absorb patents and in none of the Xience patients, a statistically significant difference. Six of the Absorb thrombotic events occurred more than 1 year after the device was placed, and in several instances these thromboses occurred more than 900 days after placement, when the BVS had largely resorbed.
“These thromboses are occurring at the late stages of BVS degradation,” Dr. Serruys noted. “The Absorb polymer is basically gone after 3 years, but it’s replaced by a proteoglycan, and some proteoglycans are quite thrombogenic,” a possible explanation for the “mysterious” very late thromboses, he said.
These “disappointing” results my be linked to inadequate lesion preparation, appropriate sizing of the BVS for the lesion, and inconsistent postdilatation of the BVS, three steps that became the guiding mantra for BVS use starting a couple of years ago, said Giulio G. Stefanini, MD, an interventional cardiologist at Humanitas Research Hospital in Milan and a discussant for the report at the meeting, which was sponsored by the Cardiovascular Research Foundation.
Dr. Stefanini said that even though the Absorb stent became available for routine use in Europe starting in 2012, the device gained little traction since then in his own practice and throughout Italy. Currently it’s used for fewer than 5% of coronary interventions in Italy, he estimated. That’s largely because “we have failed to identify a population that benefits.” Other issues include the extra time needed to place a BVS, and the need for longer treatment with dual antiplatelet therapy for patients who receive a BVS, compared with when they receive a modern metallic drug-eluting stent. The Absorb BVS received Food and Drug Administration approval for routine U.S. use in July 2016.
“It would be beautiful to have a fully bioresorbable stent. It’s a lovely concept, but we’re not there yet,” Dr. Stefanini observed.
ABSORB II was sponsored by Abbott Vascular, which markets the Absorb device. Dr. Serruys has received research support from Abbott Vascular and has been a consultant to several other device and drug companies. Dr. Stefanini has been a consultant to Boston Scientific, B.Braun, and Edwards.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
WASHINGTON – The bioabsorbable vascular scaffold bubble suddenly burst with the first 3-year follow-up data from a randomized trial that unexpectedly showed that the Absorb device significantly underperformed compared with Xience, a widely-used, second-generation metallic drug-eluting stent.
“As a pioneer of BVS [bioresorbable vascular scaffold] I’m disappointed,” Patrick W. Serruys, MD, said at the Transcatheter Cardiovascular Therapeutics annual meeting. “The performance of the comparator stent was spectacular.”
Xience surpassed Absorb in several other secondary endpoints. For example, the in-device binary restenosis rate was 7.0% with Absorb and 0.7% with Xience; the in-segment binary restenosis rate was 8% with Absorb and 3% with Xience. Target-vessel MIs occurred in 7% of the Absorb patients and 1% of the Xience patients, while clinically indicated target-lesion revascularization occurred in 6% of the Absorb patients and 1% of the Xience patients.
Another notable finding was that definite or probable in-device thrombosis occurred in nine Absorb patents and in none of the Xience patients, a statistically significant difference. Six of the Absorb thrombotic events occurred more than 1 year after the device was placed, and in several instances these thromboses occurred more than 900 days after placement, when the BVS had largely resorbed.
“These thromboses are occurring at the late stages of BVS degradation,” Dr. Serruys noted. “The Absorb polymer is basically gone after 3 years, but it’s replaced by a proteoglycan, and some proteoglycans are quite thrombogenic,” a possible explanation for the “mysterious” very late thromboses, he said.
These “disappointing” results my be linked to inadequate lesion preparation, appropriate sizing of the BVS for the lesion, and inconsistent postdilatation of the BVS, three steps that became the guiding mantra for BVS use starting a couple of years ago, said Giulio G. Stefanini, MD, an interventional cardiologist at Humanitas Research Hospital in Milan and a discussant for the report at the meeting, which was sponsored by the Cardiovascular Research Foundation.
Dr. Stefanini said that even though the Absorb stent became available for routine use in Europe starting in 2012, the device gained little traction since then in his own practice and throughout Italy. Currently it’s used for fewer than 5% of coronary interventions in Italy, he estimated. That’s largely because “we have failed to identify a population that benefits.” Other issues include the extra time needed to place a BVS, and the need for longer treatment with dual antiplatelet therapy for patients who receive a BVS, compared with when they receive a modern metallic drug-eluting stent. The Absorb BVS received Food and Drug Administration approval for routine U.S. use in July 2016.
“It would be beautiful to have a fully bioresorbable stent. It’s a lovely concept, but we’re not there yet,” Dr. Stefanini observed.
ABSORB II was sponsored by Abbott Vascular, which markets the Absorb device. Dr. Serruys has received research support from Abbott Vascular and has been a consultant to several other device and drug companies. Dr. Stefanini has been a consultant to Boston Scientific, B.Braun, and Edwards.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Key clinical point:
Major finding: In-stent or in-scaffold late luminal loss averaged 0.25 mm with Xience and 0.37 mm with Absorb, a statistically significant difference.
Data source: ABSORB II, a multicenter, randomized trial that enrolled 501 patients.
Disclosures: ABSORB II was sponsored by Abbott Vascular, which markets the Absorb device. Dr. Serruys has received research support from Abbott Vascular and has been a consultant to several other device and drug companies. Dr. Stefanini has been a consultant to Boston Scientific, B.Braun and Edwards.
Adaptive servo ventilation cuts atrial fib burden
ORLANDO – Adaptive servo ventilation produced a significant and clinically meaningful reduction in atrial fibrillation burden in patients with heart failure and sleep apnea in results from an exploratory, prospective, randomized study with 35 patients.
Adaptive servo ventilation (ASV) “may be an effective antiarrhythmic treatment producing a significant reduction in atrial fibrillation without clear evidence of being proarrhythmogenic,” Jonathan P. Piccini, MD, said at the annual scientific meeting of the Heart Failure Society of America. “Given the potential importance of this finding further studies should validate and quantify the efficacy of ASV for reducing atrial fibrillation in patients with or without heart failure.”
“A mound of data has shown that treating sleep apnea reduced arrhythmias, but until now it’s all been observational and retrospective,” Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C., said in an interview. The study he reported is “the first time” the arrhythmia effects of a sleep apnea intervention, in this case ASV, was studied in a prospective, randomized way while using implanted devices to measure the antiarrhythmic effect of the treatment.
The new finding means that additional, larger studies are now needed, he said. “If patients have sleep apnea, treating the apnea may be an incredibly important way to prevent AF or reduce its burden”
The CAT-HF (Cardiovascular Improvements With Minute Ventilation-Targeted ASV Therapy in Heart Failure) trial was originally designed to randomize 215 heart failure patients with sleep disordered breathing – and who were hospitalized for heart failure – to optimal medical therapy with or without ASV at any of 15 centers in the United States and Germany. But in August 2015, results from the SERVE-HF (Treatment of Sleep-Disordered Breathing with Predominant Central Sleep Apnea by Adaptive Servo Ventilation in Patients with Heart Failure) trial, which generally had a similar design to CAT-HF, showed an unexpected danger from ASV in patients with central sleep apnea and heart failure with reduced ejection fraction (N Engl J Med. 2015 Sept 17;373[12]:1095-105). In SERVE-HF, ASV was associated with significant increases in all-cause and cardiovascular mortality. As a result, enrollment into CAT-HF stopped prematurely with just 126 patients entered, and ASV treatment of patients already enrolled came to a halt.
The primary endpoint in the underpowered and shortened CAT-HF study, survival without cardiovascular hospitalization and with improved functional capacity measured on a 6-minute walk test, showed similar outcomes in both the ASV and control arms. But in a prespecified subgroup analysis by baseline ejection fraction, the 24 patients with heart failure with preserved ejection fraction (19% of the CAT-HF enrollment) showed a statistically significant, 62% relative improvement in the primary endpoint linked with ASV treatment compared with similar patients who did not receive ASV, Christopher M. O’Connor, MD, professor of medicine at Duke University, reported in May 2016 at the European Heart Failure meeting in Florence.
Dr. Piccini’s report focused on a prespecified subgroup analysis of CAT-HF designed to examine the impact of ASV on arrhythmias. Assessment of the impact of ASV on atrial fibrillation was possible in 35 of the 126 patients in CAT-HF who had an implanted cardiac device (pacemaker, defibrillator, or cardiac resynchronization device) with an atrial lead, and assessment of ventricular arrhythmias occurred in 46 of the CAT-HF patients with an implanted high-voltage device (a defibrillator or resynchronization device) that allowed monitoring of ventricular arrhythmias.
For the atrial fibrillation analysis, the 35 patients averaged 60 years of age, and about 90% had a reduced ejection fraction. About two-thirds had an apnea-hypopnea index greater than 30.
The results showed that the 19 patients randomized to receive ASV had an average atrial fibrillation burden of 30% at baseline that dropped to 14% after 6 months of treatment. In contrast, the 16 patients in the control arm had a AF burden of 6% at baseline and 8% after 6 months. The between-group difference for change in AF burden was statistically significant, Dr. Piccini reported, with a burden that decreased by a relative 21% with ASV treatment and increased by a relative 31% in the control arm.
Analysis of the ventricular arrhythmia subgroup showed that ASV had no statistically significant impact for either lowering or raising ventricular tachyarrhythmias or fibrillations.
Trying to reconcile this AF benefit and lack of ventricular arrhythmia harm from ASV in CAT-HF with the excess in cardiovascular deaths seen with ASV in SERVE-HF, Dr. Piccini speculated that some of the SERVE-HF deaths may not have been related to arrhythmia.
“Sudden cardiac death adjudication is profoundly difficult, and does not always equal ventricular arrhythmia,” he said. “We need to consider that some of the adverse events in patients with severe central sleep apnea and low left ventricular ejection fraction [enrolled in SERVE-HF] may have been due to causes other than arrhythmias. The CAT-HF results should motivate investigations of alternative mechanisms of death in SERVE-HF.”
The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support from ResMed and from Janssen, Gilead, St. Jude, Spectranetics, and he has been a consultant to Janssen, Spectranetics, Medtronic, GSK and BMS-Pfizer. Dr. O’Connor has been a consultant to ResMed and to several other drug and device companies.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
A small prespecified sub-group of patients in the CAT-HF (Cardiovascuar improvements with minute ventilation-targeted ASV therapy in heart failure) trial randomized to adaptive servo ventilation (ASV) showed a 21% relative reduction in atrial fibrillation burden as compared to the control arm which had only 31% relative reduction. While the CAT-HF study was discontinued following results of SERVE-HF trial, this subgroup analysis included 35 patients (19 ASV arm; 16 control arm), the majority of whom had a reduced ejection fraction. This report poses interesting questions about effects of ASV on atrial fibrillation burden in those with reduced EF given the finding that central sleep apnea and Cheyne-Stokes respiration are shown to be associated with incident atrial fibrillation in older men (May et al. Am J Respir Crit Care Med 2016).
A small prespecified sub-group of patients in the CAT-HF (Cardiovascuar improvements with minute ventilation-targeted ASV therapy in heart failure) trial randomized to adaptive servo ventilation (ASV) showed a 21% relative reduction in atrial fibrillation burden as compared to the control arm which had only 31% relative reduction. While the CAT-HF study was discontinued following results of SERVE-HF trial, this subgroup analysis included 35 patients (19 ASV arm; 16 control arm), the majority of whom had a reduced ejection fraction. This report poses interesting questions about effects of ASV on atrial fibrillation burden in those with reduced EF given the finding that central sleep apnea and Cheyne-Stokes respiration are shown to be associated with incident atrial fibrillation in older men (May et al. Am J Respir Crit Care Med 2016).
A small prespecified sub-group of patients in the CAT-HF (Cardiovascuar improvements with minute ventilation-targeted ASV therapy in heart failure) trial randomized to adaptive servo ventilation (ASV) showed a 21% relative reduction in atrial fibrillation burden as compared to the control arm which had only 31% relative reduction. While the CAT-HF study was discontinued following results of SERVE-HF trial, this subgroup analysis included 35 patients (19 ASV arm; 16 control arm), the majority of whom had a reduced ejection fraction. This report poses interesting questions about effects of ASV on atrial fibrillation burden in those with reduced EF given the finding that central sleep apnea and Cheyne-Stokes respiration are shown to be associated with incident atrial fibrillation in older men (May et al. Am J Respir Crit Care Med 2016).
ORLANDO – Adaptive servo ventilation produced a significant and clinically meaningful reduction in atrial fibrillation burden in patients with heart failure and sleep apnea in results from an exploratory, prospective, randomized study with 35 patients.
Adaptive servo ventilation (ASV) “may be an effective antiarrhythmic treatment producing a significant reduction in atrial fibrillation without clear evidence of being proarrhythmogenic,” Jonathan P. Piccini, MD, said at the annual scientific meeting of the Heart Failure Society of America. “Given the potential importance of this finding further studies should validate and quantify the efficacy of ASV for reducing atrial fibrillation in patients with or without heart failure.”
“A mound of data has shown that treating sleep apnea reduced arrhythmias, but until now it’s all been observational and retrospective,” Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C., said in an interview. The study he reported is “the first time” the arrhythmia effects of a sleep apnea intervention, in this case ASV, was studied in a prospective, randomized way while using implanted devices to measure the antiarrhythmic effect of the treatment.
The new finding means that additional, larger studies are now needed, he said. “If patients have sleep apnea, treating the apnea may be an incredibly important way to prevent AF or reduce its burden”
The CAT-HF (Cardiovascular Improvements With Minute Ventilation-Targeted ASV Therapy in Heart Failure) trial was originally designed to randomize 215 heart failure patients with sleep disordered breathing – and who were hospitalized for heart failure – to optimal medical therapy with or without ASV at any of 15 centers in the United States and Germany. But in August 2015, results from the SERVE-HF (Treatment of Sleep-Disordered Breathing with Predominant Central Sleep Apnea by Adaptive Servo Ventilation in Patients with Heart Failure) trial, which generally had a similar design to CAT-HF, showed an unexpected danger from ASV in patients with central sleep apnea and heart failure with reduced ejection fraction (N Engl J Med. 2015 Sept 17;373[12]:1095-105). In SERVE-HF, ASV was associated with significant increases in all-cause and cardiovascular mortality. As a result, enrollment into CAT-HF stopped prematurely with just 126 patients entered, and ASV treatment of patients already enrolled came to a halt.
The primary endpoint in the underpowered and shortened CAT-HF study, survival without cardiovascular hospitalization and with improved functional capacity measured on a 6-minute walk test, showed similar outcomes in both the ASV and control arms. But in a prespecified subgroup analysis by baseline ejection fraction, the 24 patients with heart failure with preserved ejection fraction (19% of the CAT-HF enrollment) showed a statistically significant, 62% relative improvement in the primary endpoint linked with ASV treatment compared with similar patients who did not receive ASV, Christopher M. O’Connor, MD, professor of medicine at Duke University, reported in May 2016 at the European Heart Failure meeting in Florence.
Dr. Piccini’s report focused on a prespecified subgroup analysis of CAT-HF designed to examine the impact of ASV on arrhythmias. Assessment of the impact of ASV on atrial fibrillation was possible in 35 of the 126 patients in CAT-HF who had an implanted cardiac device (pacemaker, defibrillator, or cardiac resynchronization device) with an atrial lead, and assessment of ventricular arrhythmias occurred in 46 of the CAT-HF patients with an implanted high-voltage device (a defibrillator or resynchronization device) that allowed monitoring of ventricular arrhythmias.
For the atrial fibrillation analysis, the 35 patients averaged 60 years of age, and about 90% had a reduced ejection fraction. About two-thirds had an apnea-hypopnea index greater than 30.
The results showed that the 19 patients randomized to receive ASV had an average atrial fibrillation burden of 30% at baseline that dropped to 14% after 6 months of treatment. In contrast, the 16 patients in the control arm had a AF burden of 6% at baseline and 8% after 6 months. The between-group difference for change in AF burden was statistically significant, Dr. Piccini reported, with a burden that decreased by a relative 21% with ASV treatment and increased by a relative 31% in the control arm.
Analysis of the ventricular arrhythmia subgroup showed that ASV had no statistically significant impact for either lowering or raising ventricular tachyarrhythmias or fibrillations.
Trying to reconcile this AF benefit and lack of ventricular arrhythmia harm from ASV in CAT-HF with the excess in cardiovascular deaths seen with ASV in SERVE-HF, Dr. Piccini speculated that some of the SERVE-HF deaths may not have been related to arrhythmia.
“Sudden cardiac death adjudication is profoundly difficult, and does not always equal ventricular arrhythmia,” he said. “We need to consider that some of the adverse events in patients with severe central sleep apnea and low left ventricular ejection fraction [enrolled in SERVE-HF] may have been due to causes other than arrhythmias. The CAT-HF results should motivate investigations of alternative mechanisms of death in SERVE-HF.”
The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support from ResMed and from Janssen, Gilead, St. Jude, Spectranetics, and he has been a consultant to Janssen, Spectranetics, Medtronic, GSK and BMS-Pfizer. Dr. O’Connor has been a consultant to ResMed and to several other drug and device companies.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – Adaptive servo ventilation produced a significant and clinically meaningful reduction in atrial fibrillation burden in patients with heart failure and sleep apnea in results from an exploratory, prospective, randomized study with 35 patients.
Adaptive servo ventilation (ASV) “may be an effective antiarrhythmic treatment producing a significant reduction in atrial fibrillation without clear evidence of being proarrhythmogenic,” Jonathan P. Piccini, MD, said at the annual scientific meeting of the Heart Failure Society of America. “Given the potential importance of this finding further studies should validate and quantify the efficacy of ASV for reducing atrial fibrillation in patients with or without heart failure.”
“A mound of data has shown that treating sleep apnea reduced arrhythmias, but until now it’s all been observational and retrospective,” Dr. Piccini, an electrophysiologist at Duke University in Durham, N.C., said in an interview. The study he reported is “the first time” the arrhythmia effects of a sleep apnea intervention, in this case ASV, was studied in a prospective, randomized way while using implanted devices to measure the antiarrhythmic effect of the treatment.
The new finding means that additional, larger studies are now needed, he said. “If patients have sleep apnea, treating the apnea may be an incredibly important way to prevent AF or reduce its burden”
The CAT-HF (Cardiovascular Improvements With Minute Ventilation-Targeted ASV Therapy in Heart Failure) trial was originally designed to randomize 215 heart failure patients with sleep disordered breathing – and who were hospitalized for heart failure – to optimal medical therapy with or without ASV at any of 15 centers in the United States and Germany. But in August 2015, results from the SERVE-HF (Treatment of Sleep-Disordered Breathing with Predominant Central Sleep Apnea by Adaptive Servo Ventilation in Patients with Heart Failure) trial, which generally had a similar design to CAT-HF, showed an unexpected danger from ASV in patients with central sleep apnea and heart failure with reduced ejection fraction (N Engl J Med. 2015 Sept 17;373[12]:1095-105). In SERVE-HF, ASV was associated with significant increases in all-cause and cardiovascular mortality. As a result, enrollment into CAT-HF stopped prematurely with just 126 patients entered, and ASV treatment of patients already enrolled came to a halt.
The primary endpoint in the underpowered and shortened CAT-HF study, survival without cardiovascular hospitalization and with improved functional capacity measured on a 6-minute walk test, showed similar outcomes in both the ASV and control arms. But in a prespecified subgroup analysis by baseline ejection fraction, the 24 patients with heart failure with preserved ejection fraction (19% of the CAT-HF enrollment) showed a statistically significant, 62% relative improvement in the primary endpoint linked with ASV treatment compared with similar patients who did not receive ASV, Christopher M. O’Connor, MD, professor of medicine at Duke University, reported in May 2016 at the European Heart Failure meeting in Florence.
Dr. Piccini’s report focused on a prespecified subgroup analysis of CAT-HF designed to examine the impact of ASV on arrhythmias. Assessment of the impact of ASV on atrial fibrillation was possible in 35 of the 126 patients in CAT-HF who had an implanted cardiac device (pacemaker, defibrillator, or cardiac resynchronization device) with an atrial lead, and assessment of ventricular arrhythmias occurred in 46 of the CAT-HF patients with an implanted high-voltage device (a defibrillator or resynchronization device) that allowed monitoring of ventricular arrhythmias.
For the atrial fibrillation analysis, the 35 patients averaged 60 years of age, and about 90% had a reduced ejection fraction. About two-thirds had an apnea-hypopnea index greater than 30.
The results showed that the 19 patients randomized to receive ASV had an average atrial fibrillation burden of 30% at baseline that dropped to 14% after 6 months of treatment. In contrast, the 16 patients in the control arm had a AF burden of 6% at baseline and 8% after 6 months. The between-group difference for change in AF burden was statistically significant, Dr. Piccini reported, with a burden that decreased by a relative 21% with ASV treatment and increased by a relative 31% in the control arm.
Analysis of the ventricular arrhythmia subgroup showed that ASV had no statistically significant impact for either lowering or raising ventricular tachyarrhythmias or fibrillations.
Trying to reconcile this AF benefit and lack of ventricular arrhythmia harm from ASV in CAT-HF with the excess in cardiovascular deaths seen with ASV in SERVE-HF, Dr. Piccini speculated that some of the SERVE-HF deaths may not have been related to arrhythmia.
“Sudden cardiac death adjudication is profoundly difficult, and does not always equal ventricular arrhythmia,” he said. “We need to consider that some of the adverse events in patients with severe central sleep apnea and low left ventricular ejection fraction [enrolled in SERVE-HF] may have been due to causes other than arrhythmias. The CAT-HF results should motivate investigations of alternative mechanisms of death in SERVE-HF.”
The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support from ResMed and from Janssen, Gilead, St. Jude, Spectranetics, and he has been a consultant to Janssen, Spectranetics, Medtronic, GSK and BMS-Pfizer. Dr. O’Connor has been a consultant to ResMed and to several other drug and device companies.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Key clinical point:
Major finding: After 6 months, ASV produced a relative 21% drop in atrial fibrillation burden, compared with increased burden in control patients.
Data source: CAT-HF, a multicenter randomized trial that enrolled 126 heart failure patients with sleep apnea.
Disclosures: The CAT-HF trial was funded by ResMed, a company that markets adaptive servo ventilation equipment. Dr. Piccini has received research support and/or consultant fees from ResMed, Janssen, Gilead, St. Jude, Spectranetics, Medtronic, GSK and BMS-Pfizer.
Advanced heart failure symptoms linked to mortality
ORLANDO – Advanced heart failure patients who are hospitalized for heart failure and have a higher symptom burden at discharge have a significantly increased rate of death or rehospitalization over the next 6 months, based on an analysis of 393 patients enrolled in a heart failure trial.
The strong link between severe symptom burden and poor near-term outcomes persisted despite adjustment for various markers of heart failure severity, suggesting that treatment aimed at reducing symptoms may be able to reduce mortality or heart failure hospitalization in advanced heart failure patients, Ellen K. Hummel, MD, said at the annual scientific meeting of the Heart Failure Society of America.
In her analysis, a severe symptom burden at the time of hospital discharge linked with an adjusted 2.9-fold increased mortality rate and a 2.5-fold increased rate of days dead or hospitalized during the next 6 months, said Dr. Hummel, a geriatric and palliative care specialist at the University of Michigan in Ann Arbor. These elevated rate ratios for patients with severe symptoms at hospital discharge were in comparison to the ratios for advanced heart failure patients in the study with no symptoms at discharge.
Three symptoms contributed to the symptom score she used in her analysis: fatigue, scored on a scale of 0-3; dyspnea, also scored 0-3; and gastrointestinal distress, scored as 0-2, creating a maximum score of 8. Her analysis categorized mild as a total score of 1-4 and severe as 5 or greater. In the study population she used for her analysis, patients enrolled in the multicenter ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial, 111 of the 393 evaluable patients (28%) had none of these symptoms, 239 (61%) had mild symptoms, and 43 (11%) had severe symptoms. Scoring was done by patients based on their subjective self-assessment at the time of hospital discharge.
The absolute, observed 6-month mortality rates were roughly 45% among patients with severe symptoms, about 17% in patients with mild symptoms, and about 12% in those with no symptoms.
The primary purpose of ESCAPE was to assess the impact that routine collection of data from a pulmonary artery catheter during hospitalization has on outcomes; the results showed no significant link between improved outcomes and getting these data (JAMA. 2005 Oct 5;294[13]:1625-33). The study ran during 2000-2003 at 26 centers in the United States and Canada. Of the 433 advanced heart failure patients enrolled in ESCAPE, 393 had complete records to allow the current analysis.
The adjustments that Dr. Hummel made in the proportional hazard analysis took into account New York Heart Association class, and severity of disease at the time of hospital discharge measured by the ESCAPE Discharge Risk Score. This score takes into account age, 6-minute walk distance, blood urea nitrogen, brain natriuretic peptide levels, blood pressure, selected drug treatments, sodium level, and history of cardiopulmonary resuscitation or mechanical ventilation.
Dr. Hummel had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – Advanced heart failure patients who are hospitalized for heart failure and have a higher symptom burden at discharge have a significantly increased rate of death or rehospitalization over the next 6 months, based on an analysis of 393 patients enrolled in a heart failure trial.
The strong link between severe symptom burden and poor near-term outcomes persisted despite adjustment for various markers of heart failure severity, suggesting that treatment aimed at reducing symptoms may be able to reduce mortality or heart failure hospitalization in advanced heart failure patients, Ellen K. Hummel, MD, said at the annual scientific meeting of the Heart Failure Society of America.
In her analysis, a severe symptom burden at the time of hospital discharge linked with an adjusted 2.9-fold increased mortality rate and a 2.5-fold increased rate of days dead or hospitalized during the next 6 months, said Dr. Hummel, a geriatric and palliative care specialist at the University of Michigan in Ann Arbor. These elevated rate ratios for patients with severe symptoms at hospital discharge were in comparison to the ratios for advanced heart failure patients in the study with no symptoms at discharge.
Three symptoms contributed to the symptom score she used in her analysis: fatigue, scored on a scale of 0-3; dyspnea, also scored 0-3; and gastrointestinal distress, scored as 0-2, creating a maximum score of 8. Her analysis categorized mild as a total score of 1-4 and severe as 5 or greater. In the study population she used for her analysis, patients enrolled in the multicenter ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial, 111 of the 393 evaluable patients (28%) had none of these symptoms, 239 (61%) had mild symptoms, and 43 (11%) had severe symptoms. Scoring was done by patients based on their subjective self-assessment at the time of hospital discharge.
The absolute, observed 6-month mortality rates were roughly 45% among patients with severe symptoms, about 17% in patients with mild symptoms, and about 12% in those with no symptoms.
The primary purpose of ESCAPE was to assess the impact that routine collection of data from a pulmonary artery catheter during hospitalization has on outcomes; the results showed no significant link between improved outcomes and getting these data (JAMA. 2005 Oct 5;294[13]:1625-33). The study ran during 2000-2003 at 26 centers in the United States and Canada. Of the 433 advanced heart failure patients enrolled in ESCAPE, 393 had complete records to allow the current analysis.
The adjustments that Dr. Hummel made in the proportional hazard analysis took into account New York Heart Association class, and severity of disease at the time of hospital discharge measured by the ESCAPE Discharge Risk Score. This score takes into account age, 6-minute walk distance, blood urea nitrogen, brain natriuretic peptide levels, blood pressure, selected drug treatments, sodium level, and history of cardiopulmonary resuscitation or mechanical ventilation.
Dr. Hummel had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ORLANDO – Advanced heart failure patients who are hospitalized for heart failure and have a higher symptom burden at discharge have a significantly increased rate of death or rehospitalization over the next 6 months, based on an analysis of 393 patients enrolled in a heart failure trial.
The strong link between severe symptom burden and poor near-term outcomes persisted despite adjustment for various markers of heart failure severity, suggesting that treatment aimed at reducing symptoms may be able to reduce mortality or heart failure hospitalization in advanced heart failure patients, Ellen K. Hummel, MD, said at the annual scientific meeting of the Heart Failure Society of America.
In her analysis, a severe symptom burden at the time of hospital discharge linked with an adjusted 2.9-fold increased mortality rate and a 2.5-fold increased rate of days dead or hospitalized during the next 6 months, said Dr. Hummel, a geriatric and palliative care specialist at the University of Michigan in Ann Arbor. These elevated rate ratios for patients with severe symptoms at hospital discharge were in comparison to the ratios for advanced heart failure patients in the study with no symptoms at discharge.
Three symptoms contributed to the symptom score she used in her analysis: fatigue, scored on a scale of 0-3; dyspnea, also scored 0-3; and gastrointestinal distress, scored as 0-2, creating a maximum score of 8. Her analysis categorized mild as a total score of 1-4 and severe as 5 or greater. In the study population she used for her analysis, patients enrolled in the multicenter ESCAPE (Evaluation Study of Congestive Heart Failure and Pulmonary Artery Catheterization Effectiveness) trial, 111 of the 393 evaluable patients (28%) had none of these symptoms, 239 (61%) had mild symptoms, and 43 (11%) had severe symptoms. Scoring was done by patients based on their subjective self-assessment at the time of hospital discharge.
The absolute, observed 6-month mortality rates were roughly 45% among patients with severe symptoms, about 17% in patients with mild symptoms, and about 12% in those with no symptoms.
The primary purpose of ESCAPE was to assess the impact that routine collection of data from a pulmonary artery catheter during hospitalization has on outcomes; the results showed no significant link between improved outcomes and getting these data (JAMA. 2005 Oct 5;294[13]:1625-33). The study ran during 2000-2003 at 26 centers in the United States and Canada. Of the 433 advanced heart failure patients enrolled in ESCAPE, 393 had complete records to allow the current analysis.
The adjustments that Dr. Hummel made in the proportional hazard analysis took into account New York Heart Association class, and severity of disease at the time of hospital discharge measured by the ESCAPE Discharge Risk Score. This score takes into account age, 6-minute walk distance, blood urea nitrogen, brain natriuretic peptide levels, blood pressure, selected drug treatments, sodium level, and history of cardiopulmonary resuscitation or mechanical ventilation.
Dr. Hummel had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE HFSA ANNUAL SCIENTIFIC MEETING
Key clinical point:
Major finding: Patients with severe symptoms at discharge had a 2.9-fold increased rate of death, compared with those with no symptoms.
Data source: A post hoc analysis of data collected from 393 patients enrolled in the ESCAPE trial.
Disclosures: Dr. Hummel had no relevant financial disclosures.
Ezetimibe’s ACS benefit centers on high-risk, post-CABG patients
ROME – Patients who have undergone coronary artery bypass surgery and who later have an acute coronary syndrome event gain the most from an aggressive lipid-lowering regimen, according to an exploratory analysis of data from more than 18,000 patients enrolled in the IMPROVE-IT trial that tested the incremental benefit from ezetimibe treatment when added to a statin.
Additional exploratory analyses further showed that high-risk acute coronary syndrome (ACS) patients without a history of coronary artery bypass grafting (CABG) also benefited from adding ezetimibe to a background regimen of simvastatin, but the benefit from adding ezetimibe completely disappeared in low-risk ACS patients, Alon Eisen, MD, said at the annual congress of the European Society of Cardiology.
‘The benefit of adding ezetimibe to a statin was enhanced in patients with prior CABG and in other high-risk patients with no prior CABG, supporting the use of more intensive lipid-lowering therapy in these high-risk patients,” said Dr. Eisen, a cardiologist at Brigham and Women’s Hospital in Boston. He also highlighted that ezetimibe is “a safe drug that is coming off patent.” Adding ezetimibe had a moderate effect on LDL cholesterol levels, cutting them from a median of 70 mg/dL in patients in the placebo arm to a median of 54 mg/dL in the group who received ezetimibe.
These results “show that if we pick the right patients, a very benign drug can have a great benefit,” said Eugene Braunwald, MD, a coinvestigator on the IMPROVE-IT trial and a collaborator with Dr. Eisen on the new analysis. The new findings “emphasize that the higher a patient’s risk, the more effect they get from cholesterol-lowering treatment,” said Dr. Braunwald, professor of medicine at Harvard University and a cardiologist at Brigham and Women’s Hospital, both in Boston.
The second exploratory analysis reported by Dr. Eisen looked at the more than 16,000 patients in IMPROVE-IT without history of CABG. The analysis applied a newly developed, nine-item formula for stratifying atherothrombotic risk (Circulation. 2016 July 26;134[4];304-13) to divide these patients into low-, intermediate- and high-risk subgroups. Patients in the high-risk subgroup (20% of the IMPROVE-IT subgroup) had a 6–percentage point reduction in their primary endpoint event rate with added ezetimibe treatment, while those at intermediate risk (31%) got a 2–percentage point decrease in endpoint events, and low-risk patients (49%) actually showed a small, less than 1–percentage point increase in endpoint events with added ezetimibe, Dr. Eisen reported.
IMPROVE-IT was funded by MERCK, the company that markets ezetimibe (Zetia). Dr. Eisen had no disclosures. Dr. Braunwald has been a consultant to Merck as well as to Bayer, Daiichi Sankyo, The Medicines Company, Novartis, and Sanofi.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
I suspect that the patients in IMPROVE-IT with a history of coronary artery bypass graft surgery were more likely than the other enrolled acute coronary syndrome patients to have more extensive and systemic atherosclerotic disease. Although coronary artery bypass addresses the most acute obstructions to coronary flow that exist at the time of surgery, the procedure does not cure the patient’s underlying vascular disease. We know that a substantial majority of coronary events occur in arteries that are not heavily stenosed.
Another important limitation to keep in mind about the IMPROVE-IT trial was that the background statin treatment all patients received was modest – 40 mg of simvastatin daily. In real-world practice, high-risk patients should go on the most potent statin regimen they can tolerate – ideally, 40 mg daily of rosuvastatin. The need for additional lipid-lowering interventions, with ezetimibe or other drugs, can then be considered as an add-on to aggressive statin therapy.
Richard A. Chazal, MD, is an invasive cardiologist and medical director of the Heart and Vascular Institute of Lee Memorial Health System in Fort Myers, Fla. He is also the current president of the American College of Cardiology. He had no disclosures. He made these comments in an interview.
I suspect that the patients in IMPROVE-IT with a history of coronary artery bypass graft surgery were more likely than the other enrolled acute coronary syndrome patients to have more extensive and systemic atherosclerotic disease. Although coronary artery bypass addresses the most acute obstructions to coronary flow that exist at the time of surgery, the procedure does not cure the patient’s underlying vascular disease. We know that a substantial majority of coronary events occur in arteries that are not heavily stenosed.
Another important limitation to keep in mind about the IMPROVE-IT trial was that the background statin treatment all patients received was modest – 40 mg of simvastatin daily. In real-world practice, high-risk patients should go on the most potent statin regimen they can tolerate – ideally, 40 mg daily of rosuvastatin. The need for additional lipid-lowering interventions, with ezetimibe or other drugs, can then be considered as an add-on to aggressive statin therapy.
Richard A. Chazal, MD, is an invasive cardiologist and medical director of the Heart and Vascular Institute of Lee Memorial Health System in Fort Myers, Fla. He is also the current president of the American College of Cardiology. He had no disclosures. He made these comments in an interview.
I suspect that the patients in IMPROVE-IT with a history of coronary artery bypass graft surgery were more likely than the other enrolled acute coronary syndrome patients to have more extensive and systemic atherosclerotic disease. Although coronary artery bypass addresses the most acute obstructions to coronary flow that exist at the time of surgery, the procedure does not cure the patient’s underlying vascular disease. We know that a substantial majority of coronary events occur in arteries that are not heavily stenosed.
Another important limitation to keep in mind about the IMPROVE-IT trial was that the background statin treatment all patients received was modest – 40 mg of simvastatin daily. In real-world practice, high-risk patients should go on the most potent statin regimen they can tolerate – ideally, 40 mg daily of rosuvastatin. The need for additional lipid-lowering interventions, with ezetimibe or other drugs, can then be considered as an add-on to aggressive statin therapy.
Richard A. Chazal, MD, is an invasive cardiologist and medical director of the Heart and Vascular Institute of Lee Memorial Health System in Fort Myers, Fla. He is also the current president of the American College of Cardiology. He had no disclosures. He made these comments in an interview.
ROME – Patients who have undergone coronary artery bypass surgery and who later have an acute coronary syndrome event gain the most from an aggressive lipid-lowering regimen, according to an exploratory analysis of data from more than 18,000 patients enrolled in the IMPROVE-IT trial that tested the incremental benefit from ezetimibe treatment when added to a statin.
Additional exploratory analyses further showed that high-risk acute coronary syndrome (ACS) patients without a history of coronary artery bypass grafting (CABG) also benefited from adding ezetimibe to a background regimen of simvastatin, but the benefit from adding ezetimibe completely disappeared in low-risk ACS patients, Alon Eisen, MD, said at the annual congress of the European Society of Cardiology.
‘The benefit of adding ezetimibe to a statin was enhanced in patients with prior CABG and in other high-risk patients with no prior CABG, supporting the use of more intensive lipid-lowering therapy in these high-risk patients,” said Dr. Eisen, a cardiologist at Brigham and Women’s Hospital in Boston. He also highlighted that ezetimibe is “a safe drug that is coming off patent.” Adding ezetimibe had a moderate effect on LDL cholesterol levels, cutting them from a median of 70 mg/dL in patients in the placebo arm to a median of 54 mg/dL in the group who received ezetimibe.
These results “show that if we pick the right patients, a very benign drug can have a great benefit,” said Eugene Braunwald, MD, a coinvestigator on the IMPROVE-IT trial and a collaborator with Dr. Eisen on the new analysis. The new findings “emphasize that the higher a patient’s risk, the more effect they get from cholesterol-lowering treatment,” said Dr. Braunwald, professor of medicine at Harvard University and a cardiologist at Brigham and Women’s Hospital, both in Boston.
The second exploratory analysis reported by Dr. Eisen looked at the more than 16,000 patients in IMPROVE-IT without history of CABG. The analysis applied a newly developed, nine-item formula for stratifying atherothrombotic risk (Circulation. 2016 July 26;134[4];304-13) to divide these patients into low-, intermediate- and high-risk subgroups. Patients in the high-risk subgroup (20% of the IMPROVE-IT subgroup) had a 6–percentage point reduction in their primary endpoint event rate with added ezetimibe treatment, while those at intermediate risk (31%) got a 2–percentage point decrease in endpoint events, and low-risk patients (49%) actually showed a small, less than 1–percentage point increase in endpoint events with added ezetimibe, Dr. Eisen reported.
IMPROVE-IT was funded by MERCK, the company that markets ezetimibe (Zetia). Dr. Eisen had no disclosures. Dr. Braunwald has been a consultant to Merck as well as to Bayer, Daiichi Sankyo, The Medicines Company, Novartis, and Sanofi.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
ROME – Patients who have undergone coronary artery bypass surgery and who later have an acute coronary syndrome event gain the most from an aggressive lipid-lowering regimen, according to an exploratory analysis of data from more than 18,000 patients enrolled in the IMPROVE-IT trial that tested the incremental benefit from ezetimibe treatment when added to a statin.
Additional exploratory analyses further showed that high-risk acute coronary syndrome (ACS) patients without a history of coronary artery bypass grafting (CABG) also benefited from adding ezetimibe to a background regimen of simvastatin, but the benefit from adding ezetimibe completely disappeared in low-risk ACS patients, Alon Eisen, MD, said at the annual congress of the European Society of Cardiology.
‘The benefit of adding ezetimibe to a statin was enhanced in patients with prior CABG and in other high-risk patients with no prior CABG, supporting the use of more intensive lipid-lowering therapy in these high-risk patients,” said Dr. Eisen, a cardiologist at Brigham and Women’s Hospital in Boston. He also highlighted that ezetimibe is “a safe drug that is coming off patent.” Adding ezetimibe had a moderate effect on LDL cholesterol levels, cutting them from a median of 70 mg/dL in patients in the placebo arm to a median of 54 mg/dL in the group who received ezetimibe.
These results “show that if we pick the right patients, a very benign drug can have a great benefit,” said Eugene Braunwald, MD, a coinvestigator on the IMPROVE-IT trial and a collaborator with Dr. Eisen on the new analysis. The new findings “emphasize that the higher a patient’s risk, the more effect they get from cholesterol-lowering treatment,” said Dr. Braunwald, professor of medicine at Harvard University and a cardiologist at Brigham and Women’s Hospital, both in Boston.
The second exploratory analysis reported by Dr. Eisen looked at the more than 16,000 patients in IMPROVE-IT without history of CABG. The analysis applied a newly developed, nine-item formula for stratifying atherothrombotic risk (Circulation. 2016 July 26;134[4];304-13) to divide these patients into low-, intermediate- and high-risk subgroups. Patients in the high-risk subgroup (20% of the IMPROVE-IT subgroup) had a 6–percentage point reduction in their primary endpoint event rate with added ezetimibe treatment, while those at intermediate risk (31%) got a 2–percentage point decrease in endpoint events, and low-risk patients (49%) actually showed a small, less than 1–percentage point increase in endpoint events with added ezetimibe, Dr. Eisen reported.
IMPROVE-IT was funded by MERCK, the company that markets ezetimibe (Zetia). Dr. Eisen had no disclosures. Dr. Braunwald has been a consultant to Merck as well as to Bayer, Daiichi Sankyo, The Medicines Company, Novartis, and Sanofi.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE ESC CONGRESS 2016
Key clinical point:
Major finding: The absolute primary-event risk reduction was 9% in post-CABG patients and 1% in all other patients.
Data source: An exploratory, post-hoc analysis of data collected in IMPROVE-IT, a multicenter trial with 18,144 patients.
Disclosures: IMPROVE-IT was funded by MERCK, the company that markets ezetimibe (Zetia). Dr. Eisen had no disclosures. Dr. Braunwald has been a consultant to Merck as well as to Bayer, Daiichi Sankyo, The Medicines Company, Novartis, and Sanofi.
Frailty stratifies pediatric liver disease severity
MONTREAL – A newly devised measurement of frailty in children effectively determined the severity of liver disease in pediatric patients and might serve as a useful, independent predictor of outcomes following liver transplantations in children and adolescents.
The adapted pediatric frailty assessment formula is a “very valid, feasible, and valuable tool” for assessing children with chronic liver disease, Eberhard Lurz, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology and Nutrition. “Frailty captures an additional marker of ill health that is independent of the MELD-Na [Model for End-Stage Liver Disease–Na] and PELD,” [Pediatric End-Stage Liver Disease] said Dr. Lurz, a pediatric gastroenterologist at the Hospital for Sick Children in Toronto.
The idea of frailty assessment of children with liver disease sprang from a 2014 report that showed a five-item frailty index could predict mortality in adults with liver disease who were listed for liver transplantation and that this predictive power was independent of the patients’ MELD scores (Am J Transplant. 2014 Aug;14[8]:1870-9). That study used a five-item frailty index developed for adults (J Gerontol A Biol Sci Med Sci. 2001;56[3]:M146-57).
Dr. Lurz came up with a pediatric version of this frailty score using pediatric-oriented measures for each of the five items. To measure exhaustion he used the PedsQL (Pediatric Quality of Life Inventory) Multidimensional Fatigue Scale; for slowness he used a 6-minute walk test; for weakness he measured grip strength; for shrinkage he measured triceps skinfold thickness; and for diminished activity he used an age-appropriate physical activity questionnaire. He prespecified that a patient’s scores for each of these five measures are calculated by comparing their test results against age-specific norms. A patient with a value that fell more than one standard deviation below the normal range scores one point for the item and those with values more than two standard deviations below the normal range score two points. Hence the maximum score for all five items is 10.
Researchers at the collaborating centers completed full assessments for 71 of 85 pediatric patients with chronic liver disease in their clinics, and each full assessment took a median of 60 minutes. The patients ranged from 8-16 years old, with an average age of 13. The cohort included 36 patients with compensated chronic liver disease (CCLD) and 35 with end-stage liver disease (ESLD) who were listed for liver transplantation.
The median frailty score of the CCLD patients was 3 and the median score for those with ESLD was 5, a statistically significant difference that was largely driven by between-group differences in fatigue scores and physical activity scores. A receiver operating characteristic curve analysis by area under the curve showed that the frailty score accounted for 83% of the difference between patients with CCLD and ESLD, comparable to the distinguishing power of the MELD-Na score. Using a cutoff on the score of 6 or greater identified patients with ESLD with 47% sensitivity and 98% specificity, and this diagnostic capability was independent of a patient’s MELD-Na or PELD score.
The five elements that contribute to this pediatric frailty score could be the focus for targeted interventions to improve the outcomes of patients scheduled to undergo liver transplantation, Dr. Lurz said.
Dr. Lurz had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – A newly devised measurement of frailty in children effectively determined the severity of liver disease in pediatric patients and might serve as a useful, independent predictor of outcomes following liver transplantations in children and adolescents.
The adapted pediatric frailty assessment formula is a “very valid, feasible, and valuable tool” for assessing children with chronic liver disease, Eberhard Lurz, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology and Nutrition. “Frailty captures an additional marker of ill health that is independent of the MELD-Na [Model for End-Stage Liver Disease–Na] and PELD,” [Pediatric End-Stage Liver Disease] said Dr. Lurz, a pediatric gastroenterologist at the Hospital for Sick Children in Toronto.
The idea of frailty assessment of children with liver disease sprang from a 2014 report that showed a five-item frailty index could predict mortality in adults with liver disease who were listed for liver transplantation and that this predictive power was independent of the patients’ MELD scores (Am J Transplant. 2014 Aug;14[8]:1870-9). That study used a five-item frailty index developed for adults (J Gerontol A Biol Sci Med Sci. 2001;56[3]:M146-57).
Dr. Lurz came up with a pediatric version of this frailty score using pediatric-oriented measures for each of the five items. To measure exhaustion he used the PedsQL (Pediatric Quality of Life Inventory) Multidimensional Fatigue Scale; for slowness he used a 6-minute walk test; for weakness he measured grip strength; for shrinkage he measured triceps skinfold thickness; and for diminished activity he used an age-appropriate physical activity questionnaire. He prespecified that a patient’s scores for each of these five measures are calculated by comparing their test results against age-specific norms. A patient with a value that fell more than one standard deviation below the normal range scores one point for the item and those with values more than two standard deviations below the normal range score two points. Hence the maximum score for all five items is 10.
Researchers at the collaborating centers completed full assessments for 71 of 85 pediatric patients with chronic liver disease in their clinics, and each full assessment took a median of 60 minutes. The patients ranged from 8-16 years old, with an average age of 13. The cohort included 36 patients with compensated chronic liver disease (CCLD) and 35 with end-stage liver disease (ESLD) who were listed for liver transplantation.
The median frailty score of the CCLD patients was 3 and the median score for those with ESLD was 5, a statistically significant difference that was largely driven by between-group differences in fatigue scores and physical activity scores. A receiver operating characteristic curve analysis by area under the curve showed that the frailty score accounted for 83% of the difference between patients with CCLD and ESLD, comparable to the distinguishing power of the MELD-Na score. Using a cutoff on the score of 6 or greater identified patients with ESLD with 47% sensitivity and 98% specificity, and this diagnostic capability was independent of a patient’s MELD-Na or PELD score.
The five elements that contribute to this pediatric frailty score could be the focus for targeted interventions to improve the outcomes of patients scheduled to undergo liver transplantation, Dr. Lurz said.
Dr. Lurz had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MONTREAL – A newly devised measurement of frailty in children effectively determined the severity of liver disease in pediatric patients and might serve as a useful, independent predictor of outcomes following liver transplantations in children and adolescents.
The adapted pediatric frailty assessment formula is a “very valid, feasible, and valuable tool” for assessing children with chronic liver disease, Eberhard Lurz, MD, said at the World Congress of Pediatric Gastroenterology, Hepatology and Nutrition. “Frailty captures an additional marker of ill health that is independent of the MELD-Na [Model for End-Stage Liver Disease–Na] and PELD,” [Pediatric End-Stage Liver Disease] said Dr. Lurz, a pediatric gastroenterologist at the Hospital for Sick Children in Toronto.
The idea of frailty assessment of children with liver disease sprang from a 2014 report that showed a five-item frailty index could predict mortality in adults with liver disease who were listed for liver transplantation and that this predictive power was independent of the patients’ MELD scores (Am J Transplant. 2014 Aug;14[8]:1870-9). That study used a five-item frailty index developed for adults (J Gerontol A Biol Sci Med Sci. 2001;56[3]:M146-57).
Dr. Lurz came up with a pediatric version of this frailty score using pediatric-oriented measures for each of the five items. To measure exhaustion he used the PedsQL (Pediatric Quality of Life Inventory) Multidimensional Fatigue Scale; for slowness he used a 6-minute walk test; for weakness he measured grip strength; for shrinkage he measured triceps skinfold thickness; and for diminished activity he used an age-appropriate physical activity questionnaire. He prespecified that a patient’s scores for each of these five measures are calculated by comparing their test results against age-specific norms. A patient with a value that fell more than one standard deviation below the normal range scores one point for the item and those with values more than two standard deviations below the normal range score two points. Hence the maximum score for all five items is 10.
Researchers at the collaborating centers completed full assessments for 71 of 85 pediatric patients with chronic liver disease in their clinics, and each full assessment took a median of 60 minutes. The patients ranged from 8-16 years old, with an average age of 13. The cohort included 36 patients with compensated chronic liver disease (CCLD) and 35 with end-stage liver disease (ESLD) who were listed for liver transplantation.
The median frailty score of the CCLD patients was 3 and the median score for those with ESLD was 5, a statistically significant difference that was largely driven by between-group differences in fatigue scores and physical activity scores. A receiver operating characteristic curve analysis by area under the curve showed that the frailty score accounted for 83% of the difference between patients with CCLD and ESLD, comparable to the distinguishing power of the MELD-Na score. Using a cutoff on the score of 6 or greater identified patients with ESLD with 47% sensitivity and 98% specificity, and this diagnostic capability was independent of a patient’s MELD-Na or PELD score.
The five elements that contribute to this pediatric frailty score could be the focus for targeted interventions to improve the outcomes of patients scheduled to undergo liver transplantation, Dr. Lurz said.
Dr. Lurz had no relevant financial disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT WCPGHAN 2016
Key clinical point:
Major finding: The pediatric frailty score identified patients with end-stage liver disease with sensitivity of 47% and specificity of 98%.
Data source: A series of 71 pediatric patients with liver disease compiled from 17 U.S. and Canadian centers.
Disclosures: Dr. Lurz had no relevant financial disclosures.