User login
Mitchel is a reporter for MDedge based in the Philadelphia area. He started with the company in 1992, when it was International Medical News Group (IMNG), and has since covered a range of medical specialties. Mitchel trained as a virologist at Roswell Park Memorial Institute in Buffalo, and then worked briefly as a researcher at Boston Children's Hospital before pivoting to journalism as a AAAS Mass Media Fellow in 1980. His first reporting job was with Science Digest magazine, and from the mid-1980s to early-1990s he was a reporter with Medical World News. @mitchelzoler
VIDEO: Childhood second-hand smoke boosts RA incidence
MADRID – Second-hand smoke exposure to children was about as potent a trigger for future rheumatoid arthritis as active smoking by an adult, based on an analysis of data collected from more than 70,000 French women followed for an average of more than 20 years
“This is the first demonstration of a rheumatoid arthritis risk associated with passive smoking,” Raphaèle Seror, MD, said at the European Congress of Rheumatology.
“This is an important finding because we can avoid passive smoke exposure,” Dr. Seror added in a video interview . The imperative to eliminate second-hand smoke exposure to children is particularly acute for those with a genetic risk for developing rheumatoid arthritis (RA), specifically children with a parent diagnosed with RA, suggested Dr. Seror, a professor of rheumatology at the University of Paris–South.
She and her associates used data collected in the E3N, a longitudinal French epidemiological study that enrolled nearly 100,000 women in 1990 when they were 40-65 years old and collected health data by questionnaire every 2-3 years for an average of 21 years. They identified from this cohort women with “confirmed” RA based on a self report of having incident RA during follow-up plus a coincident record of reimbursement for a prescription for an RA-specific treatment, such as methotrexate or a biological disease-modifying drug.
This identified 389 women with confirmed incident RA, including 350 with a complete smoking history that made the current analysis possible. The study also included 70,248 women who did not develop RA and who had provided a complete smoking history.
The analysis showed that women who reported a history of second-hand smoke exposure estimated at more than an hour daily as children but without a history of active smoking had a 43% higher rate of incident RA compared with never smoker women without a history of passive smoke exposure, Dr. Seror reported. This association just missed reaching statistical significance, a limitation that Dr. Seror attributed to a power issue as the analysis included only 30 women who had incident RA and a history of second-hand smoke exposure without adult smoke exposure. By comparison, women in the study with a history of active smoking without childhood exposure linked had a 37% increased incidence of RA, a finding that confirmed the well-known link between smoking and RA incidence.
The study also found that women with both second-hand smoke exposure as children and adult smoking linked with a 73% higher RA incidence, an indication that the contributions from second-hand smoke in children and active smoking by adults were not only similar in magnitude but also worked additively to promote RA development.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MADRID – Second-hand smoke exposure to children was about as potent a trigger for future rheumatoid arthritis as active smoking by an adult, based on an analysis of data collected from more than 70,000 French women followed for an average of more than 20 years
“This is the first demonstration of a rheumatoid arthritis risk associated with passive smoking,” Raphaèle Seror, MD, said at the European Congress of Rheumatology.
“This is an important finding because we can avoid passive smoke exposure,” Dr. Seror added in a video interview . The imperative to eliminate second-hand smoke exposure to children is particularly acute for those with a genetic risk for developing rheumatoid arthritis (RA), specifically children with a parent diagnosed with RA, suggested Dr. Seror, a professor of rheumatology at the University of Paris–South.
She and her associates used data collected in the E3N, a longitudinal French epidemiological study that enrolled nearly 100,000 women in 1990 when they were 40-65 years old and collected health data by questionnaire every 2-3 years for an average of 21 years. They identified from this cohort women with “confirmed” RA based on a self report of having incident RA during follow-up plus a coincident record of reimbursement for a prescription for an RA-specific treatment, such as methotrexate or a biological disease-modifying drug.
This identified 389 women with confirmed incident RA, including 350 with a complete smoking history that made the current analysis possible. The study also included 70,248 women who did not develop RA and who had provided a complete smoking history.
The analysis showed that women who reported a history of second-hand smoke exposure estimated at more than an hour daily as children but without a history of active smoking had a 43% higher rate of incident RA compared with never smoker women without a history of passive smoke exposure, Dr. Seror reported. This association just missed reaching statistical significance, a limitation that Dr. Seror attributed to a power issue as the analysis included only 30 women who had incident RA and a history of second-hand smoke exposure without adult smoke exposure. By comparison, women in the study with a history of active smoking without childhood exposure linked had a 37% increased incidence of RA, a finding that confirmed the well-known link between smoking and RA incidence.
The study also found that women with both second-hand smoke exposure as children and adult smoking linked with a 73% higher RA incidence, an indication that the contributions from second-hand smoke in children and active smoking by adults were not only similar in magnitude but also worked additively to promote RA development.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MADRID – Second-hand smoke exposure to children was about as potent a trigger for future rheumatoid arthritis as active smoking by an adult, based on an analysis of data collected from more than 70,000 French women followed for an average of more than 20 years
“This is the first demonstration of a rheumatoid arthritis risk associated with passive smoking,” Raphaèle Seror, MD, said at the European Congress of Rheumatology.
“This is an important finding because we can avoid passive smoke exposure,” Dr. Seror added in a video interview . The imperative to eliminate second-hand smoke exposure to children is particularly acute for those with a genetic risk for developing rheumatoid arthritis (RA), specifically children with a parent diagnosed with RA, suggested Dr. Seror, a professor of rheumatology at the University of Paris–South.
She and her associates used data collected in the E3N, a longitudinal French epidemiological study that enrolled nearly 100,000 women in 1990 when they were 40-65 years old and collected health data by questionnaire every 2-3 years for an average of 21 years. They identified from this cohort women with “confirmed” RA based on a self report of having incident RA during follow-up plus a coincident record of reimbursement for a prescription for an RA-specific treatment, such as methotrexate or a biological disease-modifying drug.
This identified 389 women with confirmed incident RA, including 350 with a complete smoking history that made the current analysis possible. The study also included 70,248 women who did not develop RA and who had provided a complete smoking history.
The analysis showed that women who reported a history of second-hand smoke exposure estimated at more than an hour daily as children but without a history of active smoking had a 43% higher rate of incident RA compared with never smoker women without a history of passive smoke exposure, Dr. Seror reported. This association just missed reaching statistical significance, a limitation that Dr. Seror attributed to a power issue as the analysis included only 30 women who had incident RA and a history of second-hand smoke exposure without adult smoke exposure. By comparison, women in the study with a history of active smoking without childhood exposure linked had a 37% increased incidence of RA, a finding that confirmed the well-known link between smoking and RA incidence.
The study also found that women with both second-hand smoke exposure as children and adult smoking linked with a 73% higher RA incidence, an indication that the contributions from second-hand smoke in children and active smoking by adults were not only similar in magnitude but also worked additively to promote RA development.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE EULAR 2017 CONGRESS
Key clinical point:
Major finding: Women with significant second-hand smoke exposure as children had a 43% increased rate of incident rheumatoid arthritis.
Data source: E3N, a prospective, longitudinal, observational study of nearly 100,000 French women begun in 1990.
Disclosures: Dr. Seror had no relevant disclosures.
VIDEO: Rheumatology biosimilars gain U.S. momentum
MADRID – With biosimilar infliximab on the U.S. market since November 2016 and producing an immediate, albeit modest, price drop for this tumor necrosis factor inhibitor (TNFi) and a second biosimilar infliximab now approved by the Food and Drug Administration and awaiting market entry, biosimilars are in a new phase of integration into U.S. practice.
“Physicians are willing to prescribe Inflectra,” the first biosimilar infliximab and the first TNFi to be sold in the United States last November, Jonathan Kay, MD, said in a video interview during the European Congress of Rheumatology. “Rheumatologists who were initially skeptical are now on the bandwagon and willing to prescribe biosimilars,” said Dr. Kay, a rheumatologist who has often consulted on biosimilar issues and has recently spoken to rheumatologists at various state society meetings to explain the U.S. biosimilar regulatory concepts and spread the message of the societal value of these agents.
“This is not a quick and casual drug evaluation” that produces “knockoff drugs,” but a “careful and extensive” FDA review that results in drugs that are equivalent in efficacy, safety, and immunogenicity to the reference drug and only compete on price, he explained.
When Pfizer began marketing Inflectra last Fall, it set the drug’s list price 15% lower than the list price at the time for Remicade, the reference-product infliximab. However, complex pricing and rebate strategies actually led to Remicade selling for a lower price than Inflectra, at least for some U.S. hospitals, including the University of Massachusetts in Worcester, where Dr. Kay is a professor of medicine.
“The effect of biosimilars is to reduce the cost to patients of an effective treatment. Whether that cost is for the reference drug or for the biosimilar drug doesn’t matter [from society’s perspective] as long as patients are able to receive an effective therapy at a [more] affordable cost, making the effective therapy available to more patients,” he said.
While Inflectra’s price impact my have been modest so far, the biosimilar effect on infliximab’s cost may soon intensify now that a second biosimilar of this TNFi, Renflexis – made by Samsung Bioepis and with U.S. marketing by Merck, received FDA approval on April 21, 2017. Until recently, U.S. pharmaceutical regulations had been understood to require a 180-day hiatus between FDA marketing approval for a biosimilar and the start of U.S. sales. But, on June 12, 2017, the U.S. Supreme Court, in a 9-0 decision, ruled that this 180-day wait was not required, making it possible for U.S. marketing of Renflexis to begin soon. (In mid-June, a statement on the Merck U.S. website for Renflexis says that the product is not currently available.)
Availability of a second biosimilar infliximab “is likely to drive the price down rapidly,” predicted Dr. Kay, citing what happened when multiple biosimilars for a reference drug came onto the European market.
Two other biosimilar TNFi have also received FDA marketing approvals but remain on hold as patent issues and litigation barriers play out. Erelzi – biosimilar etanercept – received FDA approval in August 2016, and Amjevita, biosimilar adalimumab, received FDA approval last September.
The efficacy and safety of Inflectra specifically, and by extension all biosimilars, received a recent boost with publication of findings from a randomized study with 482 patients that provided a real-world test of the core principle of biosimilar equivalence. After Inflectra came onto the Norwegian market, during July 2014 to August 2015, Norwegian researchers ran the NOR-SWTICH trial, which randomized patients who were on stable treatment with Remicade for a variety of indications (including 41% with a rheumatologic disease) to either stay on Remicade or to abruptly switch to treatment with Inflectra. During 1-year follow-up, the incidence of adverse effects and of episodes of disease worsening were virtually identical in the two treatment arms (Lancet. 2017 June 10;389[10086]:2304-16).
Dr. Kay has been a consultant to several companies that develop or market biosimilars, including Samsung Bioepis, Amgen, Pfizer, and Sandoz (Novartis), and to AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Genentech, Janssen, Roche, and UCB.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MADRID – With biosimilar infliximab on the U.S. market since November 2016 and producing an immediate, albeit modest, price drop for this tumor necrosis factor inhibitor (TNFi) and a second biosimilar infliximab now approved by the Food and Drug Administration and awaiting market entry, biosimilars are in a new phase of integration into U.S. practice.
“Physicians are willing to prescribe Inflectra,” the first biosimilar infliximab and the first TNFi to be sold in the United States last November, Jonathan Kay, MD, said in a video interview during the European Congress of Rheumatology. “Rheumatologists who were initially skeptical are now on the bandwagon and willing to prescribe biosimilars,” said Dr. Kay, a rheumatologist who has often consulted on biosimilar issues and has recently spoken to rheumatologists at various state society meetings to explain the U.S. biosimilar regulatory concepts and spread the message of the societal value of these agents.
“This is not a quick and casual drug evaluation” that produces “knockoff drugs,” but a “careful and extensive” FDA review that results in drugs that are equivalent in efficacy, safety, and immunogenicity to the reference drug and only compete on price, he explained.
When Pfizer began marketing Inflectra last Fall, it set the drug’s list price 15% lower than the list price at the time for Remicade, the reference-product infliximab. However, complex pricing and rebate strategies actually led to Remicade selling for a lower price than Inflectra, at least for some U.S. hospitals, including the University of Massachusetts in Worcester, where Dr. Kay is a professor of medicine.
“The effect of biosimilars is to reduce the cost to patients of an effective treatment. Whether that cost is for the reference drug or for the biosimilar drug doesn’t matter [from society’s perspective] as long as patients are able to receive an effective therapy at a [more] affordable cost, making the effective therapy available to more patients,” he said.
While Inflectra’s price impact my have been modest so far, the biosimilar effect on infliximab’s cost may soon intensify now that a second biosimilar of this TNFi, Renflexis – made by Samsung Bioepis and with U.S. marketing by Merck, received FDA approval on April 21, 2017. Until recently, U.S. pharmaceutical regulations had been understood to require a 180-day hiatus between FDA marketing approval for a biosimilar and the start of U.S. sales. But, on June 12, 2017, the U.S. Supreme Court, in a 9-0 decision, ruled that this 180-day wait was not required, making it possible for U.S. marketing of Renflexis to begin soon. (In mid-June, a statement on the Merck U.S. website for Renflexis says that the product is not currently available.)
Availability of a second biosimilar infliximab “is likely to drive the price down rapidly,” predicted Dr. Kay, citing what happened when multiple biosimilars for a reference drug came onto the European market.
Two other biosimilar TNFi have also received FDA marketing approvals but remain on hold as patent issues and litigation barriers play out. Erelzi – biosimilar etanercept – received FDA approval in August 2016, and Amjevita, biosimilar adalimumab, received FDA approval last September.
The efficacy and safety of Inflectra specifically, and by extension all biosimilars, received a recent boost with publication of findings from a randomized study with 482 patients that provided a real-world test of the core principle of biosimilar equivalence. After Inflectra came onto the Norwegian market, during July 2014 to August 2015, Norwegian researchers ran the NOR-SWTICH trial, which randomized patients who were on stable treatment with Remicade for a variety of indications (including 41% with a rheumatologic disease) to either stay on Remicade or to abruptly switch to treatment with Inflectra. During 1-year follow-up, the incidence of adverse effects and of episodes of disease worsening were virtually identical in the two treatment arms (Lancet. 2017 June 10;389[10086]:2304-16).
Dr. Kay has been a consultant to several companies that develop or market biosimilars, including Samsung Bioepis, Amgen, Pfizer, and Sandoz (Novartis), and to AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Genentech, Janssen, Roche, and UCB.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MADRID – With biosimilar infliximab on the U.S. market since November 2016 and producing an immediate, albeit modest, price drop for this tumor necrosis factor inhibitor (TNFi) and a second biosimilar infliximab now approved by the Food and Drug Administration and awaiting market entry, biosimilars are in a new phase of integration into U.S. practice.
“Physicians are willing to prescribe Inflectra,” the first biosimilar infliximab and the first TNFi to be sold in the United States last November, Jonathan Kay, MD, said in a video interview during the European Congress of Rheumatology. “Rheumatologists who were initially skeptical are now on the bandwagon and willing to prescribe biosimilars,” said Dr. Kay, a rheumatologist who has often consulted on biosimilar issues and has recently spoken to rheumatologists at various state society meetings to explain the U.S. biosimilar regulatory concepts and spread the message of the societal value of these agents.
“This is not a quick and casual drug evaluation” that produces “knockoff drugs,” but a “careful and extensive” FDA review that results in drugs that are equivalent in efficacy, safety, and immunogenicity to the reference drug and only compete on price, he explained.
When Pfizer began marketing Inflectra last Fall, it set the drug’s list price 15% lower than the list price at the time for Remicade, the reference-product infliximab. However, complex pricing and rebate strategies actually led to Remicade selling for a lower price than Inflectra, at least for some U.S. hospitals, including the University of Massachusetts in Worcester, where Dr. Kay is a professor of medicine.
“The effect of biosimilars is to reduce the cost to patients of an effective treatment. Whether that cost is for the reference drug or for the biosimilar drug doesn’t matter [from society’s perspective] as long as patients are able to receive an effective therapy at a [more] affordable cost, making the effective therapy available to more patients,” he said.
While Inflectra’s price impact my have been modest so far, the biosimilar effect on infliximab’s cost may soon intensify now that a second biosimilar of this TNFi, Renflexis – made by Samsung Bioepis and with U.S. marketing by Merck, received FDA approval on April 21, 2017. Until recently, U.S. pharmaceutical regulations had been understood to require a 180-day hiatus between FDA marketing approval for a biosimilar and the start of U.S. sales. But, on June 12, 2017, the U.S. Supreme Court, in a 9-0 decision, ruled that this 180-day wait was not required, making it possible for U.S. marketing of Renflexis to begin soon. (In mid-June, a statement on the Merck U.S. website for Renflexis says that the product is not currently available.)
Availability of a second biosimilar infliximab “is likely to drive the price down rapidly,” predicted Dr. Kay, citing what happened when multiple biosimilars for a reference drug came onto the European market.
Two other biosimilar TNFi have also received FDA marketing approvals but remain on hold as patent issues and litigation barriers play out. Erelzi – biosimilar etanercept – received FDA approval in August 2016, and Amjevita, biosimilar adalimumab, received FDA approval last September.
The efficacy and safety of Inflectra specifically, and by extension all biosimilars, received a recent boost with publication of findings from a randomized study with 482 patients that provided a real-world test of the core principle of biosimilar equivalence. After Inflectra came onto the Norwegian market, during July 2014 to August 2015, Norwegian researchers ran the NOR-SWTICH trial, which randomized patients who were on stable treatment with Remicade for a variety of indications (including 41% with a rheumatologic disease) to either stay on Remicade or to abruptly switch to treatment with Inflectra. During 1-year follow-up, the incidence of adverse effects and of episodes of disease worsening were virtually identical in the two treatment arms (Lancet. 2017 June 10;389[10086]:2304-16).
Dr. Kay has been a consultant to several companies that develop or market biosimilars, including Samsung Bioepis, Amgen, Pfizer, and Sandoz (Novartis), and to AbbVie, Boehringer Ingelheim, Bristol-Myers Squibb, Genentech, Janssen, Roche, and UCB.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM THE EULAR 2017 CONGRESS
Studies provide insight into link between cancer immunotherapy and autoimmune disease
MADRID – Rheumatologists all over the world are beginning to find that the new class of anticancer immune checkpoint inhibitor therapies have the potential to elicit symptoms of rheumatoid arthritis (RA) and other rheumatic diseases in patients with no previous history of them, and two reports from the European Congress of Rheumatology provide typical examples.
These immune checkpoint inhibitor (ICI) agents, which include ipilimumab (Yervoy), nivolumab (Opdivo), and pembrolizumab (Keytruda), target regulatory pathways in T cells to boost antitumor immune responses, leading to improved survival for many cancer patients, but the induction of rheumatic disease can sometimes lead to the suspension of the agents, according to investigators.
“This phenomenon was unknown to me and my group before [February 2016], when we started noting referrals of patients from oncology,” Dr. Calabrese said. “We were seeing symptoms of everything from Sjögren’s syndrome to inflammatory arthritis and myositis in patients being treated with these drugs for their cancer.” The same year, Dr. Calabrese and her team began coordinating an ongoing study to assess these patients.
Dr. Calabrese said that the cohort has shown so far that patients who develop autoimmune disease after immune checkpoint inhibitors “require much higher doses – of steroids in particular – to treat their symptoms,” and this can all too often result in being taken out of a clinical trial or having to stop cancer treatment.
Most of the patients in the cohort were treated with steroids only, while three patients received biologic agents, and four received methotrexate or antimalarials.
Dr. Calabrese said that the serology results were available for all the patients in the cohort and “were largely unremarkable.”
She noted that the rheumatic symptoms did not always resolve after pausing or stopping the cancer treatment. “We have some patients that have been off their checkpoint inhibitors for over a year and still have symptoms, so it’s looking like it might be a more long-term effect,” she said.
“In my unit, we also manage patients with myeloma, and I developed a weekly consultation with a cancer center,” Dr. Belkhir said. In 2015, she saw her first patient with RA and no previous history who had been treated with checkpoint inhibitors. That patient’s symptoms resolved after treatment with nonsteroidal anti-inflammatory drugs alone.
Dr. Belkhir is sharing results from this and five other patients presenting with symptoms of RA after their cancer treatment with immune checkpoint inhibitors, taken from a larger cohort of patients (n = 13) with a spectrum of rheumatic disease–like adverse effects. None of the six patients in this study had a previous clinical history of RA. They manifested their RA symptoms after a median of 1 month on cancer immunotherapy.
Some were able to continue their checkpoint inhibitors and be treated simultaneously for RA with steroids, antimalarials, methotrexate, and NSAIDs, Dr. Belkhir said. None received biologic agents, and each medication strategy, she said, was arrived at in consultation with the treating oncologist.
Dr. Belkhir’s team also looked closely at serology and found all six patients to be at least weakly, and mostly strongly, seropositive for RA. Three patients underwent testing for anticyclic citrullinated protein antibodies prior to starting cancer immunotherapy and two of these three were anti-CCP positive. Now, she said, the oncologists she’s working with are testing for anticyclic citrullinated peptides and rheumatoid factor prior to initiating cancer immunotherapy, so that this relationship is better understood.
“It is possible that antibodies were already present and that the anti-PD1 immunotherapy,” one type of immune checkpoint inhibitor, “acted as a trigger for the disease.” Animal studies have suggested a role for PD1 in the development of autoimmune disease, “but it’s not well investigated,” Dr. Belkhir said.
Dr. Belkhir and Dr. Calabrese both acknowledged that the understanding of checkpoint inhibitor–induced autoimmune disease is in its infancy. Clinical trials largely missed the phenomenon, the researchers said, because the trials were not designed to capture musculoskeletal adverse effects with the same granularity as other serious adverse events.
“This will be a long discussion in the months and the years ahead with oncologists,” Dr. Belkhir said.
Neither Dr. Calabrese nor Dr. Belkhir reported having any relevant conflicts of interest.
MADRID – Rheumatologists all over the world are beginning to find that the new class of anticancer immune checkpoint inhibitor therapies have the potential to elicit symptoms of rheumatoid arthritis (RA) and other rheumatic diseases in patients with no previous history of them, and two reports from the European Congress of Rheumatology provide typical examples.
These immune checkpoint inhibitor (ICI) agents, which include ipilimumab (Yervoy), nivolumab (Opdivo), and pembrolizumab (Keytruda), target regulatory pathways in T cells to boost antitumor immune responses, leading to improved survival for many cancer patients, but the induction of rheumatic disease can sometimes lead to the suspension of the agents, according to investigators.
“This phenomenon was unknown to me and my group before [February 2016], when we started noting referrals of patients from oncology,” Dr. Calabrese said. “We were seeing symptoms of everything from Sjögren’s syndrome to inflammatory arthritis and myositis in patients being treated with these drugs for their cancer.” The same year, Dr. Calabrese and her team began coordinating an ongoing study to assess these patients.
Dr. Calabrese said that the cohort has shown so far that patients who develop autoimmune disease after immune checkpoint inhibitors “require much higher doses – of steroids in particular – to treat their symptoms,” and this can all too often result in being taken out of a clinical trial or having to stop cancer treatment.
Most of the patients in the cohort were treated with steroids only, while three patients received biologic agents, and four received methotrexate or antimalarials.
Dr. Calabrese said that the serology results were available for all the patients in the cohort and “were largely unremarkable.”
She noted that the rheumatic symptoms did not always resolve after pausing or stopping the cancer treatment. “We have some patients that have been off their checkpoint inhibitors for over a year and still have symptoms, so it’s looking like it might be a more long-term effect,” she said.
“In my unit, we also manage patients with myeloma, and I developed a weekly consultation with a cancer center,” Dr. Belkhir said. In 2015, she saw her first patient with RA and no previous history who had been treated with checkpoint inhibitors. That patient’s symptoms resolved after treatment with nonsteroidal anti-inflammatory drugs alone.
Dr. Belkhir is sharing results from this and five other patients presenting with symptoms of RA after their cancer treatment with immune checkpoint inhibitors, taken from a larger cohort of patients (n = 13) with a spectrum of rheumatic disease–like adverse effects. None of the six patients in this study had a previous clinical history of RA. They manifested their RA symptoms after a median of 1 month on cancer immunotherapy.
Some were able to continue their checkpoint inhibitors and be treated simultaneously for RA with steroids, antimalarials, methotrexate, and NSAIDs, Dr. Belkhir said. None received biologic agents, and each medication strategy, she said, was arrived at in consultation with the treating oncologist.
Dr. Belkhir’s team also looked closely at serology and found all six patients to be at least weakly, and mostly strongly, seropositive for RA. Three patients underwent testing for anticyclic citrullinated protein antibodies prior to starting cancer immunotherapy and two of these three were anti-CCP positive. Now, she said, the oncologists she’s working with are testing for anticyclic citrullinated peptides and rheumatoid factor prior to initiating cancer immunotherapy, so that this relationship is better understood.
“It is possible that antibodies were already present and that the anti-PD1 immunotherapy,” one type of immune checkpoint inhibitor, “acted as a trigger for the disease.” Animal studies have suggested a role for PD1 in the development of autoimmune disease, “but it’s not well investigated,” Dr. Belkhir said.
Dr. Belkhir and Dr. Calabrese both acknowledged that the understanding of checkpoint inhibitor–induced autoimmune disease is in its infancy. Clinical trials largely missed the phenomenon, the researchers said, because the trials were not designed to capture musculoskeletal adverse effects with the same granularity as other serious adverse events.
“This will be a long discussion in the months and the years ahead with oncologists,” Dr. Belkhir said.
Neither Dr. Calabrese nor Dr. Belkhir reported having any relevant conflicts of interest.
MADRID – Rheumatologists all over the world are beginning to find that the new class of anticancer immune checkpoint inhibitor therapies have the potential to elicit symptoms of rheumatoid arthritis (RA) and other rheumatic diseases in patients with no previous history of them, and two reports from the European Congress of Rheumatology provide typical examples.
These immune checkpoint inhibitor (ICI) agents, which include ipilimumab (Yervoy), nivolumab (Opdivo), and pembrolizumab (Keytruda), target regulatory pathways in T cells to boost antitumor immune responses, leading to improved survival for many cancer patients, but the induction of rheumatic disease can sometimes lead to the suspension of the agents, according to investigators.
“This phenomenon was unknown to me and my group before [February 2016], when we started noting referrals of patients from oncology,” Dr. Calabrese said. “We were seeing symptoms of everything from Sjögren’s syndrome to inflammatory arthritis and myositis in patients being treated with these drugs for their cancer.” The same year, Dr. Calabrese and her team began coordinating an ongoing study to assess these patients.
Dr. Calabrese said that the cohort has shown so far that patients who develop autoimmune disease after immune checkpoint inhibitors “require much higher doses – of steroids in particular – to treat their symptoms,” and this can all too often result in being taken out of a clinical trial or having to stop cancer treatment.
Most of the patients in the cohort were treated with steroids only, while three patients received biologic agents, and four received methotrexate or antimalarials.
Dr. Calabrese said that the serology results were available for all the patients in the cohort and “were largely unremarkable.”
She noted that the rheumatic symptoms did not always resolve after pausing or stopping the cancer treatment. “We have some patients that have been off their checkpoint inhibitors for over a year and still have symptoms, so it’s looking like it might be a more long-term effect,” she said.
“In my unit, we also manage patients with myeloma, and I developed a weekly consultation with a cancer center,” Dr. Belkhir said. In 2015, she saw her first patient with RA and no previous history who had been treated with checkpoint inhibitors. That patient’s symptoms resolved after treatment with nonsteroidal anti-inflammatory drugs alone.
Dr. Belkhir is sharing results from this and five other patients presenting with symptoms of RA after their cancer treatment with immune checkpoint inhibitors, taken from a larger cohort of patients (n = 13) with a spectrum of rheumatic disease–like adverse effects. None of the six patients in this study had a previous clinical history of RA. They manifested their RA symptoms after a median of 1 month on cancer immunotherapy.
Some were able to continue their checkpoint inhibitors and be treated simultaneously for RA with steroids, antimalarials, methotrexate, and NSAIDs, Dr. Belkhir said. None received biologic agents, and each medication strategy, she said, was arrived at in consultation with the treating oncologist.
Dr. Belkhir’s team also looked closely at serology and found all six patients to be at least weakly, and mostly strongly, seropositive for RA. Three patients underwent testing for anticyclic citrullinated protein antibodies prior to starting cancer immunotherapy and two of these three were anti-CCP positive. Now, she said, the oncologists she’s working with are testing for anticyclic citrullinated peptides and rheumatoid factor prior to initiating cancer immunotherapy, so that this relationship is better understood.
“It is possible that antibodies were already present and that the anti-PD1 immunotherapy,” one type of immune checkpoint inhibitor, “acted as a trigger for the disease.” Animal studies have suggested a role for PD1 in the development of autoimmune disease, “but it’s not well investigated,” Dr. Belkhir said.
Dr. Belkhir and Dr. Calabrese both acknowledged that the understanding of checkpoint inhibitor–induced autoimmune disease is in its infancy. Clinical trials largely missed the phenomenon, the researchers said, because the trials were not designed to capture musculoskeletal adverse effects with the same granularity as other serious adverse events.
“This will be a long discussion in the months and the years ahead with oncologists,” Dr. Belkhir said.
Neither Dr. Calabrese nor Dr. Belkhir reported having any relevant conflicts of interest.
AT THE EULAR 2017 CONGRESS
Key clinical point:
Major finding: Rheumatic symptoms did not always resolve after pausing or stopping the cancer treatment, and some were able to continue their checkpoint inhibitors and be treated simultaneously for RA.
Data source: Two retrospective cohort reviews of patients on immune checkpoint inhibitors.
Disclosures: Neither Dr. Calabrese nor Dr. Belkhir reported having any relevant conflicts of interest.
VIDEO: No cancer risk found from biological DMARDs
MADRID – Additional real-world evidence confirmed that biological disease modifying drugs used to treat rheumatoid arthritis produced no spikes in new cancers or in cancer recurrences in registry data from tens of thousands of Swedish patients.
Among rheumatoid arthritis patients with a history of cancer, patients treated with a tumor necrosis factor inhibitor (TNFi) were not at an increased risk for cancer recurrence, Johan Askling, MD, said at the European Congress of Rheumatology. In a second study, patients with rheumatoid arthritis (RA) treated with a non-TNFi, biological, disease-modifying drug, specifically abatacept, rituximab, or tocilizumab, had no significantly different rate of new cancer onset when compared with RA patients who never received a biological disease modifying drug nor when compared with the general Swedish adult population, said Dr. Askling, a professor of clinical epidemiology at the Karolinska Institute in Stockholm.
“Five-year data are a good start, but we need data on 30-year risk,” Dr. Askling said in an interview.
The cancer-recurrence risk study with TNFi treatment used data collected by the Swedish national outpatient care registry on nearly 62,000 people, the Swedish cancer registry, and a rheumatology treatment registry called ARTIS. It also included patients treated during 2001-2014. From these sources, the researchers identified 446 RA patients with a history of at least one cancer who then began treatment with any type of TNFi and matched these cases with 1,278 similar RA patients with a cancer history who had never received a biologic drug. On average, the patients were nearly 10 years removed from their initial cancer diagnoses, and the average duration on TNFi treatment was nearly 5 years.
The adjusted hazard ratio for cancer recurrence among the TNFi recipients was reduced by a nominal 30%, compared with that of the controls, a difference that was not statistically significant, Dr. Askling reported.
The second study used data from similar sources for patients treated during 2006-2014 and included nearly 100,000 Swedes from the general population, more than 42,000 RA patients who did not receive a biological drug, more than 14,000 treated with either a first or second TNFi drug, and 1,693 patients treated with tocilizumab (Actemra), 1,894 on abatacept (Orencia), and 3,119 on rituximab (Rituxan).
The rates of new onset cancer in any of these treatment groups, including the patients on tocilizumab, abatacept, or rituximab, was not significantly different from the rate among RA patients who never received a biologic drug, nor from the general Swedish population rate, Dr. Askling said.
This is “one of the first large-scale assessments” of the cancer risk posed by non-TNFi biological drugs, aside from what was reported from the pivotal trials for these drugs, Dr. Askling said.
Dr. Askling has received research support from AbbVie, Lilly, MSD, Pfizer, Roche, and UCB.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Rheumatologists began having concerns about the possible impact of biological drugs on cancer when these types of drugs first became available 20 or more years ago. Registries have allowed us to follow these patients, and, so far, we have consistently seen that the risk for cancer is very low. The major adverse effect from treatment with biological drugs is infection.
The most confirmed finding has been that biologic drugs do not cause new cancers. We have known less about the risk patients with a history of cancer face for recurrence by taking a biological drug. The data on this have so far been scarce. Most guidelines advise that, when patients have had cancer, the possible use of a biologic drug should be the subject of a shared-decision discussion with the patient. The new data reported by Dr. Askling add to the risk information we have available to discuss with patients.
The risk that biologic drugs poses for infections is more complex. The infection risk also depends on a patient’s use of glucocorticoids, their age, and their comorbidities. The infection risk faced by a patient from treatment with a biological drug requires an individualized discussion that takes into account the severity of all the relevant risk factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
João E. Fonseca, MD is a professor of rheumatology at the University of Lisbon. He has been a speaker for or has received research funding from Abbvie, MSD, Pfizer, Roche, and UCB. He made these comments in a video interview.
Rheumatologists began having concerns about the possible impact of biological drugs on cancer when these types of drugs first became available 20 or more years ago. Registries have allowed us to follow these patients, and, so far, we have consistently seen that the risk for cancer is very low. The major adverse effect from treatment with biological drugs is infection.
The most confirmed finding has been that biologic drugs do not cause new cancers. We have known less about the risk patients with a history of cancer face for recurrence by taking a biological drug. The data on this have so far been scarce. Most guidelines advise that, when patients have had cancer, the possible use of a biologic drug should be the subject of a shared-decision discussion with the patient. The new data reported by Dr. Askling add to the risk information we have available to discuss with patients.
The risk that biologic drugs poses for infections is more complex. The infection risk also depends on a patient’s use of glucocorticoids, their age, and their comorbidities. The infection risk faced by a patient from treatment with a biological drug requires an individualized discussion that takes into account the severity of all the relevant risk factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
João E. Fonseca, MD is a professor of rheumatology at the University of Lisbon. He has been a speaker for or has received research funding from Abbvie, MSD, Pfizer, Roche, and UCB. He made these comments in a video interview.
Rheumatologists began having concerns about the possible impact of biological drugs on cancer when these types of drugs first became available 20 or more years ago. Registries have allowed us to follow these patients, and, so far, we have consistently seen that the risk for cancer is very low. The major adverse effect from treatment with biological drugs is infection.
The most confirmed finding has been that biologic drugs do not cause new cancers. We have known less about the risk patients with a history of cancer face for recurrence by taking a biological drug. The data on this have so far been scarce. Most guidelines advise that, when patients have had cancer, the possible use of a biologic drug should be the subject of a shared-decision discussion with the patient. The new data reported by Dr. Askling add to the risk information we have available to discuss with patients.
The risk that biologic drugs poses for infections is more complex. The infection risk also depends on a patient’s use of glucocorticoids, their age, and their comorbidities. The infection risk faced by a patient from treatment with a biological drug requires an individualized discussion that takes into account the severity of all the relevant risk factors.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
João E. Fonseca, MD is a professor of rheumatology at the University of Lisbon. He has been a speaker for or has received research funding from Abbvie, MSD, Pfizer, Roche, and UCB. He made these comments in a video interview.
MADRID – Additional real-world evidence confirmed that biological disease modifying drugs used to treat rheumatoid arthritis produced no spikes in new cancers or in cancer recurrences in registry data from tens of thousands of Swedish patients.
Among rheumatoid arthritis patients with a history of cancer, patients treated with a tumor necrosis factor inhibitor (TNFi) were not at an increased risk for cancer recurrence, Johan Askling, MD, said at the European Congress of Rheumatology. In a second study, patients with rheumatoid arthritis (RA) treated with a non-TNFi, biological, disease-modifying drug, specifically abatacept, rituximab, or tocilizumab, had no significantly different rate of new cancer onset when compared with RA patients who never received a biological disease modifying drug nor when compared with the general Swedish adult population, said Dr. Askling, a professor of clinical epidemiology at the Karolinska Institute in Stockholm.
“Five-year data are a good start, but we need data on 30-year risk,” Dr. Askling said in an interview.
The cancer-recurrence risk study with TNFi treatment used data collected by the Swedish national outpatient care registry on nearly 62,000 people, the Swedish cancer registry, and a rheumatology treatment registry called ARTIS. It also included patients treated during 2001-2014. From these sources, the researchers identified 446 RA patients with a history of at least one cancer who then began treatment with any type of TNFi and matched these cases with 1,278 similar RA patients with a cancer history who had never received a biologic drug. On average, the patients were nearly 10 years removed from their initial cancer diagnoses, and the average duration on TNFi treatment was nearly 5 years.
The adjusted hazard ratio for cancer recurrence among the TNFi recipients was reduced by a nominal 30%, compared with that of the controls, a difference that was not statistically significant, Dr. Askling reported.
The second study used data from similar sources for patients treated during 2006-2014 and included nearly 100,000 Swedes from the general population, more than 42,000 RA patients who did not receive a biological drug, more than 14,000 treated with either a first or second TNFi drug, and 1,693 patients treated with tocilizumab (Actemra), 1,894 on abatacept (Orencia), and 3,119 on rituximab (Rituxan).
The rates of new onset cancer in any of these treatment groups, including the patients on tocilizumab, abatacept, or rituximab, was not significantly different from the rate among RA patients who never received a biologic drug, nor from the general Swedish population rate, Dr. Askling said.
This is “one of the first large-scale assessments” of the cancer risk posed by non-TNFi biological drugs, aside from what was reported from the pivotal trials for these drugs, Dr. Askling said.
Dr. Askling has received research support from AbbVie, Lilly, MSD, Pfizer, Roche, and UCB.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
MADRID – Additional real-world evidence confirmed that biological disease modifying drugs used to treat rheumatoid arthritis produced no spikes in new cancers or in cancer recurrences in registry data from tens of thousands of Swedish patients.
Among rheumatoid arthritis patients with a history of cancer, patients treated with a tumor necrosis factor inhibitor (TNFi) were not at an increased risk for cancer recurrence, Johan Askling, MD, said at the European Congress of Rheumatology. In a second study, patients with rheumatoid arthritis (RA) treated with a non-TNFi, biological, disease-modifying drug, specifically abatacept, rituximab, or tocilizumab, had no significantly different rate of new cancer onset when compared with RA patients who never received a biological disease modifying drug nor when compared with the general Swedish adult population, said Dr. Askling, a professor of clinical epidemiology at the Karolinska Institute in Stockholm.
“Five-year data are a good start, but we need data on 30-year risk,” Dr. Askling said in an interview.
The cancer-recurrence risk study with TNFi treatment used data collected by the Swedish national outpatient care registry on nearly 62,000 people, the Swedish cancer registry, and a rheumatology treatment registry called ARTIS. It also included patients treated during 2001-2014. From these sources, the researchers identified 446 RA patients with a history of at least one cancer who then began treatment with any type of TNFi and matched these cases with 1,278 similar RA patients with a cancer history who had never received a biologic drug. On average, the patients were nearly 10 years removed from their initial cancer diagnoses, and the average duration on TNFi treatment was nearly 5 years.
The adjusted hazard ratio for cancer recurrence among the TNFi recipients was reduced by a nominal 30%, compared with that of the controls, a difference that was not statistically significant, Dr. Askling reported.
The second study used data from similar sources for patients treated during 2006-2014 and included nearly 100,000 Swedes from the general population, more than 42,000 RA patients who did not receive a biological drug, more than 14,000 treated with either a first or second TNFi drug, and 1,693 patients treated with tocilizumab (Actemra), 1,894 on abatacept (Orencia), and 3,119 on rituximab (Rituxan).
The rates of new onset cancer in any of these treatment groups, including the patients on tocilizumab, abatacept, or rituximab, was not significantly different from the rate among RA patients who never received a biologic drug, nor from the general Swedish population rate, Dr. Askling said.
This is “one of the first large-scale assessments” of the cancer risk posed by non-TNFi biological drugs, aside from what was reported from the pivotal trials for these drugs, Dr. Askling said.
Dr. Askling has received research support from AbbVie, Lilly, MSD, Pfizer, Roche, and UCB.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT THE EULAR 2017 CONGRESS
Key clinical point:
Major finding: Former cancer patients on a TNFi had no increased cancer recurrences, compared with patients on other rheumatic treatments.
Data source: Data from the Swedish national registries.
Disclosures: Dr. Askling has received research support from AbbVie, Lilly, MSD, Pfizer, Roche, and UCB.
HFrEF mortality halved when treatment matches guidelines
PARIS – Heart failure patients who received guideline-directed pharmacotherapy, at dosages that approached guideline-directed levels, had roughly half the 6-month mortality as did similar patients who did not receive this level of treatment in a real-world, observational study with more than 6,000 patients.
Adherence to pharmacologic treatment guidelines for patients with heart failure with reduced ejection fraction (HFrEF) “was strongly associated with clinical outcomes during 6-month follow-up,” Michel Komajda, MD, said at a meeting held by the Heart Failure Association of the European Society of Cardiology. The findings highlight the importance of closely following guideline recommendations in routine practice.
[polldaddy:9772629]
The analysis used data collected in the QUALIFY (Quality of Adherence to Guideline Recommendations for Life-Saving Treatment in Heart Failure: an International Survey) registry, which enrolled 7,127 HFrEF patients during September 2013–December 2014 at 547 centers in 36 countries, mostly in Europe, Asia, and Africa but also in Canada, Ecuador, and Australia. All enrolled patients had to have been hospitalized for worsening heart failure at least once during the 1-15 months before they entered QUALIFY.
Dr. Komajda and his associates assessed each enrolled patient at baseline by their treatment with each of four guideline-recommended drug classes: an ACE inhibitor or angiotensin receptor blocker; a beta-blocker; an aldosterone receptor antagonist (ARA) if the patient’s functional status was rated as New York Heart Association class II, III, or IV; and ivabradine (Corlanor) if the patient was in NYHA class II, III, or IV, in sinus rhythm, had a heart rate of at least 70 beats per minute, and if the patient was in a country where ivabradine was available. Because patient enrollment occurred in 2013 and 2014, the study couldn’t include the new formulation of sacubitril plus valsartan (Entresto) in its analysis.
For each eligible drug class a patient received 1 point if their daily prescribed dosage was at least 50% of the recommended dosage (or 100% for an ARA), 0.5 points if the patient received the recommended drug but at a lower dosage, and no points if the drug wasn’t given. A patient also received 1 point if they were appropriately not given a drug because of a documented contraindication or intolerance. The researchers then calculated each patient’s “adherence score” by dividing their point total by the potentially maximum number of points that each patient could have received (a number that ranged from 2 to 4). They defined a score of 1 (which meant the patients received at least half the recommended dosage of all recommended drugs) as good adherence, a score of 0.51-0.99 as moderate adherence, and a score of 0.5 or less as poor adherence.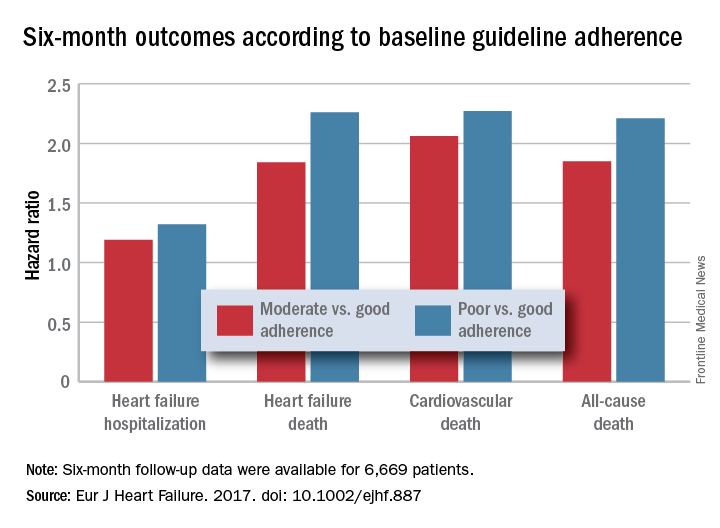
Because patient enrollment occurred during 2013 and 2014, the benchmark heart failure treatment guidelines were those issued by the European Society of Cardiology in 2012 (Eur Heart J. 2012 July;33[14]:1787-1847).
Concurrently with Dr. Komajda’s report at the meeting the results appeared in an article online (Eur J Heart Failure. 2017. doi: 10.1002/ejhf.887).
QUANTIFY was sponsored by Servier. Dr. Komajda has received honoraria from Servier and from Amgen, AstraZeneca, Bristol-Myers Squibb, Menarini, MSD, Novartis, Novo Nordisk, and Sanofi.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
This was a wonderful and useful study. It was large, prospective, covered a wide geographic area, and showed that drug dosage matters when treating heart failure with reduced ejection fraction.
The geographic diversity was a strength, but also a potential weakness because of the resulting differences among the enrolled patients in financial resources, ethnic and genetic makeup, and their tolerance to various drugs. I hope that further research can dissect the role that each of these factors played in the results.
Another limitation is the relatively simplistic approach used for assessing drug dosages to calculate the adherence scores. While it is a very useful step forward to classify patients by the drug dosages they received, there could be some very legitimate variations in dosages based on parameters such as blood pressure. An important question to address in the future is whether it is better to use all the recommended drugs at reduced dosages if necessary, or to use fewer agents at higher dosages.
But these are just quibbles about what is a very important study.
Andrew J.S. Coats, MD , is a cardiologist, professor of medicine, and academic vice-president for the Monash Warwick Alliance of Warwick University in Coventry, England, and Monash University in Melbourne. He has received honoraria from Impulse Dynamics, Menarini, PsiOxus, ResMed, Respicardia, and Servier. He made these comments as designated discussant for Dr. Komajda’s report.
This was a wonderful and useful study. It was large, prospective, covered a wide geographic area, and showed that drug dosage matters when treating heart failure with reduced ejection fraction.
The geographic diversity was a strength, but also a potential weakness because of the resulting differences among the enrolled patients in financial resources, ethnic and genetic makeup, and their tolerance to various drugs. I hope that further research can dissect the role that each of these factors played in the results.
Another limitation is the relatively simplistic approach used for assessing drug dosages to calculate the adherence scores. While it is a very useful step forward to classify patients by the drug dosages they received, there could be some very legitimate variations in dosages based on parameters such as blood pressure. An important question to address in the future is whether it is better to use all the recommended drugs at reduced dosages if necessary, or to use fewer agents at higher dosages.
But these are just quibbles about what is a very important study.
Andrew J.S. Coats, MD , is a cardiologist, professor of medicine, and academic vice-president for the Monash Warwick Alliance of Warwick University in Coventry, England, and Monash University in Melbourne. He has received honoraria from Impulse Dynamics, Menarini, PsiOxus, ResMed, Respicardia, and Servier. He made these comments as designated discussant for Dr. Komajda’s report.
This was a wonderful and useful study. It was large, prospective, covered a wide geographic area, and showed that drug dosage matters when treating heart failure with reduced ejection fraction.
The geographic diversity was a strength, but also a potential weakness because of the resulting differences among the enrolled patients in financial resources, ethnic and genetic makeup, and their tolerance to various drugs. I hope that further research can dissect the role that each of these factors played in the results.
Another limitation is the relatively simplistic approach used for assessing drug dosages to calculate the adherence scores. While it is a very useful step forward to classify patients by the drug dosages they received, there could be some very legitimate variations in dosages based on parameters such as blood pressure. An important question to address in the future is whether it is better to use all the recommended drugs at reduced dosages if necessary, or to use fewer agents at higher dosages.
But these are just quibbles about what is a very important study.
Andrew J.S. Coats, MD , is a cardiologist, professor of medicine, and academic vice-president for the Monash Warwick Alliance of Warwick University in Coventry, England, and Monash University in Melbourne. He has received honoraria from Impulse Dynamics, Menarini, PsiOxus, ResMed, Respicardia, and Servier. He made these comments as designated discussant for Dr. Komajda’s report.
PARIS – Heart failure patients who received guideline-directed pharmacotherapy, at dosages that approached guideline-directed levels, had roughly half the 6-month mortality as did similar patients who did not receive this level of treatment in a real-world, observational study with more than 6,000 patients.
Adherence to pharmacologic treatment guidelines for patients with heart failure with reduced ejection fraction (HFrEF) “was strongly associated with clinical outcomes during 6-month follow-up,” Michel Komajda, MD, said at a meeting held by the Heart Failure Association of the European Society of Cardiology. The findings highlight the importance of closely following guideline recommendations in routine practice.
[polldaddy:9772629]
The analysis used data collected in the QUALIFY (Quality of Adherence to Guideline Recommendations for Life-Saving Treatment in Heart Failure: an International Survey) registry, which enrolled 7,127 HFrEF patients during September 2013–December 2014 at 547 centers in 36 countries, mostly in Europe, Asia, and Africa but also in Canada, Ecuador, and Australia. All enrolled patients had to have been hospitalized for worsening heart failure at least once during the 1-15 months before they entered QUALIFY.
Dr. Komajda and his associates assessed each enrolled patient at baseline by their treatment with each of four guideline-recommended drug classes: an ACE inhibitor or angiotensin receptor blocker; a beta-blocker; an aldosterone receptor antagonist (ARA) if the patient’s functional status was rated as New York Heart Association class II, III, or IV; and ivabradine (Corlanor) if the patient was in NYHA class II, III, or IV, in sinus rhythm, had a heart rate of at least 70 beats per minute, and if the patient was in a country where ivabradine was available. Because patient enrollment occurred in 2013 and 2014, the study couldn’t include the new formulation of sacubitril plus valsartan (Entresto) in its analysis.
For each eligible drug class a patient received 1 point if their daily prescribed dosage was at least 50% of the recommended dosage (or 100% for an ARA), 0.5 points if the patient received the recommended drug but at a lower dosage, and no points if the drug wasn’t given. A patient also received 1 point if they were appropriately not given a drug because of a documented contraindication or intolerance. The researchers then calculated each patient’s “adherence score” by dividing their point total by the potentially maximum number of points that each patient could have received (a number that ranged from 2 to 4). They defined a score of 1 (which meant the patients received at least half the recommended dosage of all recommended drugs) as good adherence, a score of 0.51-0.99 as moderate adherence, and a score of 0.5 or less as poor adherence.
Because patient enrollment occurred during 2013 and 2014, the benchmark heart failure treatment guidelines were those issued by the European Society of Cardiology in 2012 (Eur Heart J. 2012 July;33[14]:1787-1847).
Concurrently with Dr. Komajda’s report at the meeting the results appeared in an article online (Eur J Heart Failure. 2017. doi: 10.1002/ejhf.887).
QUANTIFY was sponsored by Servier. Dr. Komajda has received honoraria from Servier and from Amgen, AstraZeneca, Bristol-Myers Squibb, Menarini, MSD, Novartis, Novo Nordisk, and Sanofi.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
PARIS – Heart failure patients who received guideline-directed pharmacotherapy, at dosages that approached guideline-directed levels, had roughly half the 6-month mortality as did similar patients who did not receive this level of treatment in a real-world, observational study with more than 6,000 patients.
Adherence to pharmacologic treatment guidelines for patients with heart failure with reduced ejection fraction (HFrEF) “was strongly associated with clinical outcomes during 6-month follow-up,” Michel Komajda, MD, said at a meeting held by the Heart Failure Association of the European Society of Cardiology. The findings highlight the importance of closely following guideline recommendations in routine practice.
[polldaddy:9772629]
The analysis used data collected in the QUALIFY (Quality of Adherence to Guideline Recommendations for Life-Saving Treatment in Heart Failure: an International Survey) registry, which enrolled 7,127 HFrEF patients during September 2013–December 2014 at 547 centers in 36 countries, mostly in Europe, Asia, and Africa but also in Canada, Ecuador, and Australia. All enrolled patients had to have been hospitalized for worsening heart failure at least once during the 1-15 months before they entered QUALIFY.
Dr. Komajda and his associates assessed each enrolled patient at baseline by their treatment with each of four guideline-recommended drug classes: an ACE inhibitor or angiotensin receptor blocker; a beta-blocker; an aldosterone receptor antagonist (ARA) if the patient’s functional status was rated as New York Heart Association class II, III, or IV; and ivabradine (Corlanor) if the patient was in NYHA class II, III, or IV, in sinus rhythm, had a heart rate of at least 70 beats per minute, and if the patient was in a country where ivabradine was available. Because patient enrollment occurred in 2013 and 2014, the study couldn’t include the new formulation of sacubitril plus valsartan (Entresto) in its analysis.
For each eligible drug class a patient received 1 point if their daily prescribed dosage was at least 50% of the recommended dosage (or 100% for an ARA), 0.5 points if the patient received the recommended drug but at a lower dosage, and no points if the drug wasn’t given. A patient also received 1 point if they were appropriately not given a drug because of a documented contraindication or intolerance. The researchers then calculated each patient’s “adherence score” by dividing their point total by the potentially maximum number of points that each patient could have received (a number that ranged from 2 to 4). They defined a score of 1 (which meant the patients received at least half the recommended dosage of all recommended drugs) as good adherence, a score of 0.51-0.99 as moderate adherence, and a score of 0.5 or less as poor adherence.
Because patient enrollment occurred during 2013 and 2014, the benchmark heart failure treatment guidelines were those issued by the European Society of Cardiology in 2012 (Eur Heart J. 2012 July;33[14]:1787-1847).
Concurrently with Dr. Komajda’s report at the meeting the results appeared in an article online (Eur J Heart Failure. 2017. doi: 10.1002/ejhf.887).
QUANTIFY was sponsored by Servier. Dr. Komajda has received honoraria from Servier and from Amgen, AstraZeneca, Bristol-Myers Squibb, Menarini, MSD, Novartis, Novo Nordisk, and Sanofi.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT HEART FAILURE 2017
Key clinical point:
Major finding: Six-month all-cause, cardiovascular, and heart failure mortalities were doubled in patients not on guideline-adherent therapy and dosages.
Data source: QUANTIFY, an international registry with 6,669 HFrEF patients followed for 6 months.
Disclosures: QUANTIFY was sponsored by Servier. Dr. Komajda has received honoraria from Servier and from Amgen, AstraZeneca, Bristol-Myers Squibb, Menarini, MSD, Novartis, Novo Nordisk, and Sanofi.
Atrial fibrillation blunts beta-blockers for HFrEF
PARIS – Maximal beta-blocker treatment and lower heart rates are effective at cutting all-cause mortality in patients with heart failure with reduced ejection fraction (HFrEF) who are also in sinus rhythm, but it’s a totally different story for patients with similar heart failure plus atrial fibrillation. In the atrial fibrillation subgroup, treatment with a beta-blocker linked with no mortality benefit, and lower heart rates – below 70 beats per minute – appeared to actually link with worse patient survival, based on a meta-analysis of data from 11 beta-blocker trials with a total of more than 17,000 patients.
“Beta blockers may be doing good in heart failure patients with atrial fibrillation, but they also are doing harm that neutralizes any good they do.” In patients with HFrEF and atrial fibrillation, “I don’t like to see the heart rate below 80 beats per minute,” John G.F. Cleland, MD, said at a meeting held by the Heart Failure Association of the ESC.

“We’ve perhaps been too aggressive with heart-rate control in HFrEF patients with atrial fibrillation,” he added in an interview. In these patients “in the range of 60-100 bpm it doesn’t seem to make a lot of difference what the heart rate is, and, if it is less than 70 bpm, patients seem to do a little worse. When we treat these patients with a beta-blocker we don’t see benefit in any way that we’ve looked at the data.”
In contrast, among HFrEF patients in sinus rhythm “beta-blocker treatment is similarly effective regardless of what the baseline heart rate was. The benefit was as great when the baseline rate was 70 bpm or 90 bpm, so heart rate is not a great predictor of beta-blocker benefit in these patients. Patients who tolerated the full beta-blocker dosage had the greatest benefit, and patients who achieved the slowest heart rates also had the greatest benefit.”
In the multivariate models that Dr. Cleland and his associates tested in their meta-analysis, in HFrEF patients in sinus rhythm, the relationship between reduced heart rate and mortality benefit was stronger statistically than between beta-blocker dosage and reduced mortality, he said. “This suggests to me that, while we should use the targeted beta-blocker dosages when we can, it’s more important to achieve a target heart rate in these patients of 55-65 bpm.”
Dr. Cleland hypothesized, based on a report presented at the same meeting by a different research group, that reduced heart rate is not beneficial in HFrEF patients with atrial fibrillation because in this subgroup slower heart rates linked with an increased number of brief pauses in left ventricular pumping. These pauses may result in ventricular arrhythmias, he speculated. “It may be that beta-blockers are equally effective at slowing heart rate in patients with or without atrial fibrillation, but there is also harm from beta-blockers because they’re causing pauses in patients with atrial fibrillation,” he said.
These days, if he has a HFrEF patient with atrial fibrillation whose heart rate slows to 60 bpm, he will stop digoxin treatment if the patient is on that drug, and he will also reduce the beta-blocker dosage but not discontinue it.
The findings came from the Collaborative Systematic Overview of Randomized Controlled Trials of Beta-Blockers in the Treatment of Heart Failure (BB-META-HF), which included data from 11 large beta-blocker randomized trials in heart failure that had been published during 1993-2005. The analysis included data from 17,378 HFrEF patients, with 14,313 (82%) in sinus rhythm and 3,065 (18%) with atrial fibrillation. Follow-up data of patients on treatment was available for 15,007 of these patients.
Dr. Cleland and his associates showed in multivariate analyses that, when they controlled for several baseline demographic and clinical variables among patients in sinus rhythm who received a beta-blocker, the follow-up all-cause mortality fell by 36%, compared with placebo, in patients with a resting baseline heart rate of less than 70 bpm; by 21%, compared with placebo, in patients with a baseline heart rate of 70-90 bpm; and by 38%, compared with placebo, in patients with a baseline heart rate of more than 90 bpm. All three reductions were statistically significant. In contrast, among patients who also had atrial fibrillation beta-blocker treatment linked with no significant mortality reduction, compared with placebo, for patients with any baseline heart rate. Concurrently with Dr. Cleland’s report at the meeting the results appeared online (J Amer Coll Cardiol. 2017 Apr 30. doi: 10.1016/j.jacc.2017.04.001).
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
The findings from this analysis have several implications. First, the association of reduced mortality with reduced heart rate occurred only in patients in sinus rhythm. The irregular heart rhythms in patients with atrial fibrillation may counterbalance any reverse remodeling effects that come from reducing heart rate.
Also, the beneficial effect of beta-blocker treatment was roughly similar regardless of whether baseline heart rate was high or low. This distinguishes beta-blockers from ivabradine, a drug that only reduces heart rate. The magnitude of benefit from ivabradine treatment depends on a patient’s baseline heart rate. The observation that beta-blockers do not have the same limitation suggests that the mechanism of action of beta-blockers may go beyond their heart rate effect. It may also result from the effect of beta-blockers on antagonizing toxic effects from beta-adrenergic stimulation.
The pooled analysis also showed that many patients with HFrEF in sinus rhythm continued to have a high heart rate despite beta-blocker treatment. These patients may get additional benefit from further treatment to reduce their heart rate, with an agent like ivabradine.
But we must be cautious in interpreting the findings because they represent a secondary analysis, and the endpoint studied does not take into account quality of life, exercise tolerance, heart rate control, and tachyarrhythmias. We need prospective, randomized trials of HFrEF patients in sinus rhythm and with atrial fibrillation to better understand how to optimally treat these different types of patients.
The findings highlight that beta-blockers remain a mainstay of treatment for patients with HFrEF in sinus rhythm, and that we have more limited treatment options for HFrEF patients with atrial fibrillation.
Michael Böhm, MD, is professor and director of the cardiology clinic at Saarland University Hospital in Homburg, Germany. He has received honoraria from Bayer, Medtronic, Servier, and Pfizer, and he was a coauthor on the report presented by Dr. Cleland. He made these comments as designated discussant for the study.
The findings from this analysis have several implications. First, the association of reduced mortality with reduced heart rate occurred only in patients in sinus rhythm. The irregular heart rhythms in patients with atrial fibrillation may counterbalance any reverse remodeling effects that come from reducing heart rate.
Also, the beneficial effect of beta-blocker treatment was roughly similar regardless of whether baseline heart rate was high or low. This distinguishes beta-blockers from ivabradine, a drug that only reduces heart rate. The magnitude of benefit from ivabradine treatment depends on a patient’s baseline heart rate. The observation that beta-blockers do not have the same limitation suggests that the mechanism of action of beta-blockers may go beyond their heart rate effect. It may also result from the effect of beta-blockers on antagonizing toxic effects from beta-adrenergic stimulation.
The pooled analysis also showed that many patients with HFrEF in sinus rhythm continued to have a high heart rate despite beta-blocker treatment. These patients may get additional benefit from further treatment to reduce their heart rate, with an agent like ivabradine.
But we must be cautious in interpreting the findings because they represent a secondary analysis, and the endpoint studied does not take into account quality of life, exercise tolerance, heart rate control, and tachyarrhythmias. We need prospective, randomized trials of HFrEF patients in sinus rhythm and with atrial fibrillation to better understand how to optimally treat these different types of patients.
The findings highlight that beta-blockers remain a mainstay of treatment for patients with HFrEF in sinus rhythm, and that we have more limited treatment options for HFrEF patients with atrial fibrillation.
Michael Böhm, MD, is professor and director of the cardiology clinic at Saarland University Hospital in Homburg, Germany. He has received honoraria from Bayer, Medtronic, Servier, and Pfizer, and he was a coauthor on the report presented by Dr. Cleland. He made these comments as designated discussant for the study.
The findings from this analysis have several implications. First, the association of reduced mortality with reduced heart rate occurred only in patients in sinus rhythm. The irregular heart rhythms in patients with atrial fibrillation may counterbalance any reverse remodeling effects that come from reducing heart rate.
Also, the beneficial effect of beta-blocker treatment was roughly similar regardless of whether baseline heart rate was high or low. This distinguishes beta-blockers from ivabradine, a drug that only reduces heart rate. The magnitude of benefit from ivabradine treatment depends on a patient’s baseline heart rate. The observation that beta-blockers do not have the same limitation suggests that the mechanism of action of beta-blockers may go beyond their heart rate effect. It may also result from the effect of beta-blockers on antagonizing toxic effects from beta-adrenergic stimulation.
The pooled analysis also showed that many patients with HFrEF in sinus rhythm continued to have a high heart rate despite beta-blocker treatment. These patients may get additional benefit from further treatment to reduce their heart rate, with an agent like ivabradine.
But we must be cautious in interpreting the findings because they represent a secondary analysis, and the endpoint studied does not take into account quality of life, exercise tolerance, heart rate control, and tachyarrhythmias. We need prospective, randomized trials of HFrEF patients in sinus rhythm and with atrial fibrillation to better understand how to optimally treat these different types of patients.
The findings highlight that beta-blockers remain a mainstay of treatment for patients with HFrEF in sinus rhythm, and that we have more limited treatment options for HFrEF patients with atrial fibrillation.
Michael Böhm, MD, is professor and director of the cardiology clinic at Saarland University Hospital in Homburg, Germany. He has received honoraria from Bayer, Medtronic, Servier, and Pfizer, and he was a coauthor on the report presented by Dr. Cleland. He made these comments as designated discussant for the study.
PARIS – Maximal beta-blocker treatment and lower heart rates are effective at cutting all-cause mortality in patients with heart failure with reduced ejection fraction (HFrEF) who are also in sinus rhythm, but it’s a totally different story for patients with similar heart failure plus atrial fibrillation. In the atrial fibrillation subgroup, treatment with a beta-blocker linked with no mortality benefit, and lower heart rates – below 70 beats per minute – appeared to actually link with worse patient survival, based on a meta-analysis of data from 11 beta-blocker trials with a total of more than 17,000 patients.
“Beta blockers may be doing good in heart failure patients with atrial fibrillation, but they also are doing harm that neutralizes any good they do.” In patients with HFrEF and atrial fibrillation, “I don’t like to see the heart rate below 80 beats per minute,” John G.F. Cleland, MD, said at a meeting held by the Heart Failure Association of the ESC.

“We’ve perhaps been too aggressive with heart-rate control in HFrEF patients with atrial fibrillation,” he added in an interview. In these patients “in the range of 60-100 bpm it doesn’t seem to make a lot of difference what the heart rate is, and, if it is less than 70 bpm, patients seem to do a little worse. When we treat these patients with a beta-blocker we don’t see benefit in any way that we’ve looked at the data.”
In contrast, among HFrEF patients in sinus rhythm “beta-blocker treatment is similarly effective regardless of what the baseline heart rate was. The benefit was as great when the baseline rate was 70 bpm or 90 bpm, so heart rate is not a great predictor of beta-blocker benefit in these patients. Patients who tolerated the full beta-blocker dosage had the greatest benefit, and patients who achieved the slowest heart rates also had the greatest benefit.”
In the multivariate models that Dr. Cleland and his associates tested in their meta-analysis, in HFrEF patients in sinus rhythm, the relationship between reduced heart rate and mortality benefit was stronger statistically than between beta-blocker dosage and reduced mortality, he said. “This suggests to me that, while we should use the targeted beta-blocker dosages when we can, it’s more important to achieve a target heart rate in these patients of 55-65 bpm.”
Dr. Cleland hypothesized, based on a report presented at the same meeting by a different research group, that reduced heart rate is not beneficial in HFrEF patients with atrial fibrillation because in this subgroup slower heart rates linked with an increased number of brief pauses in left ventricular pumping. These pauses may result in ventricular arrhythmias, he speculated. “It may be that beta-blockers are equally effective at slowing heart rate in patients with or without atrial fibrillation, but there is also harm from beta-blockers because they’re causing pauses in patients with atrial fibrillation,” he said.
These days, if he has a HFrEF patient with atrial fibrillation whose heart rate slows to 60 bpm, he will stop digoxin treatment if the patient is on that drug, and he will also reduce the beta-blocker dosage but not discontinue it.
The findings came from the Collaborative Systematic Overview of Randomized Controlled Trials of Beta-Blockers in the Treatment of Heart Failure (BB-META-HF), which included data from 11 large beta-blocker randomized trials in heart failure that had been published during 1993-2005. The analysis included data from 17,378 HFrEF patients, with 14,313 (82%) in sinus rhythm and 3,065 (18%) with atrial fibrillation. Follow-up data of patients on treatment was available for 15,007 of these patients.
Dr. Cleland and his associates showed in multivariate analyses that, when they controlled for several baseline demographic and clinical variables among patients in sinus rhythm who received a beta-blocker, the follow-up all-cause mortality fell by 36%, compared with placebo, in patients with a resting baseline heart rate of less than 70 bpm; by 21%, compared with placebo, in patients with a baseline heart rate of 70-90 bpm; and by 38%, compared with placebo, in patients with a baseline heart rate of more than 90 bpm. All three reductions were statistically significant. In contrast, among patients who also had atrial fibrillation beta-blocker treatment linked with no significant mortality reduction, compared with placebo, for patients with any baseline heart rate. Concurrently with Dr. Cleland’s report at the meeting the results appeared online (J Amer Coll Cardiol. 2017 Apr 30. doi: 10.1016/j.jacc.2017.04.001).
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
PARIS – Maximal beta-blocker treatment and lower heart rates are effective at cutting all-cause mortality in patients with heart failure with reduced ejection fraction (HFrEF) who are also in sinus rhythm, but it’s a totally different story for patients with similar heart failure plus atrial fibrillation. In the atrial fibrillation subgroup, treatment with a beta-blocker linked with no mortality benefit, and lower heart rates – below 70 beats per minute – appeared to actually link with worse patient survival, based on a meta-analysis of data from 11 beta-blocker trials with a total of more than 17,000 patients.
“Beta blockers may be doing good in heart failure patients with atrial fibrillation, but they also are doing harm that neutralizes any good they do.” In patients with HFrEF and atrial fibrillation, “I don’t like to see the heart rate below 80 beats per minute,” John G.F. Cleland, MD, said at a meeting held by the Heart Failure Association of the ESC.

“We’ve perhaps been too aggressive with heart-rate control in HFrEF patients with atrial fibrillation,” he added in an interview. In these patients “in the range of 60-100 bpm it doesn’t seem to make a lot of difference what the heart rate is, and, if it is less than 70 bpm, patients seem to do a little worse. When we treat these patients with a beta-blocker we don’t see benefit in any way that we’ve looked at the data.”
In contrast, among HFrEF patients in sinus rhythm “beta-blocker treatment is similarly effective regardless of what the baseline heart rate was. The benefit was as great when the baseline rate was 70 bpm or 90 bpm, so heart rate is not a great predictor of beta-blocker benefit in these patients. Patients who tolerated the full beta-blocker dosage had the greatest benefit, and patients who achieved the slowest heart rates also had the greatest benefit.”
In the multivariate models that Dr. Cleland and his associates tested in their meta-analysis, in HFrEF patients in sinus rhythm, the relationship between reduced heart rate and mortality benefit was stronger statistically than between beta-blocker dosage and reduced mortality, he said. “This suggests to me that, while we should use the targeted beta-blocker dosages when we can, it’s more important to achieve a target heart rate in these patients of 55-65 bpm.”
Dr. Cleland hypothesized, based on a report presented at the same meeting by a different research group, that reduced heart rate is not beneficial in HFrEF patients with atrial fibrillation because in this subgroup slower heart rates linked with an increased number of brief pauses in left ventricular pumping. These pauses may result in ventricular arrhythmias, he speculated. “It may be that beta-blockers are equally effective at slowing heart rate in patients with or without atrial fibrillation, but there is also harm from beta-blockers because they’re causing pauses in patients with atrial fibrillation,” he said.
These days, if he has a HFrEF patient with atrial fibrillation whose heart rate slows to 60 bpm, he will stop digoxin treatment if the patient is on that drug, and he will also reduce the beta-blocker dosage but not discontinue it.
The findings came from the Collaborative Systematic Overview of Randomized Controlled Trials of Beta-Blockers in the Treatment of Heart Failure (BB-META-HF), which included data from 11 large beta-blocker randomized trials in heart failure that had been published during 1993-2005. The analysis included data from 17,378 HFrEF patients, with 14,313 (82%) in sinus rhythm and 3,065 (18%) with atrial fibrillation. Follow-up data of patients on treatment was available for 15,007 of these patients.
Dr. Cleland and his associates showed in multivariate analyses that, when they controlled for several baseline demographic and clinical variables among patients in sinus rhythm who received a beta-blocker, the follow-up all-cause mortality fell by 36%, compared with placebo, in patients with a resting baseline heart rate of less than 70 bpm; by 21%, compared with placebo, in patients with a baseline heart rate of 70-90 bpm; and by 38%, compared with placebo, in patients with a baseline heart rate of more than 90 bpm. All three reductions were statistically significant. In contrast, among patients who also had atrial fibrillation beta-blocker treatment linked with no significant mortality reduction, compared with placebo, for patients with any baseline heart rate. Concurrently with Dr. Cleland’s report at the meeting the results appeared online (J Amer Coll Cardiol. 2017 Apr 30. doi: 10.1016/j.jacc.2017.04.001).
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT HEART FAILURE 2017
Key clinical point:
Major finding: All-cause mortality was similar in patients with HFrEF and atrial fibrillation regardless of whether they received a beta-blocker or placebo.
Data source: BB-META-HF, a meta-analysis of 11 beta-blocker treatment trials with 17,378 HFrEF patients.
Disclosures: BB-META-HF received funding from Menarini and GlaxoSmithKline. Dr. Cleland has received research funding and honoraria from GlaxoSmithKline.
Lung cancer linked to suicide
WASHINGTON – U.S. patients diagnosed with lung cancer have had the highest suicide rates among patients diagnosed with any of the other most common, non-skin cancers, and they also had a substantially higher suicide risk, compared with the general U.S. adult population, based on U.S. national data collected during 1973-2013.
Although U.S. lung cancer patients showed a “steep” decline in suicide rates starting in about 1985 that then accelerated beginning in the mid-1990s, as recently as 2010-2013 the rate was roughly twice as high in lung cancer patients when compared with the general U.S. adult population. The rate of lung cancer patients taking their lives was also significantly above the suicide rates among patients with breast, colorectal, or prostate cancer, Mohamed Rahouma, MD, reported at an international conference of the American Thoracic Society.
However, he also stressed that identification of lung cancer patients at especially high suicide risk was important to allow “proper psychological assessment, support, and counseling to reduce [suicide] rates.”
Lung cancer patients with the highest rates included men, widowed individuals, septuagenarians, and Asians, his analysis showed. Standardized mortality ratios (SMRs) for suicide of these highest-risk subgroups were near or exceeding 10 times fold higher than the suicide rates of comparable demographic groups among the general U.S. adult population, according to Dr. Rahouma and his associates.
The overall SMR for all lung cancer patients during the entire four decades studied, compared with the overall U.S. adult population, was 4. Even during the period 2005-2013, when suicide among lung cancer patients had fallen to its lowest level, the SMR for this group was still more than 2.
The investigators used data collected by the U.S. Surveillance Epidemiology and End Results (SEER) Program cancer database maintained by the National Cancer Institute. For suicide rates among the general U.S. population they used data from the National Vital Statistics Reports produced by the Centers for Disease Control and Prevention. The SEER database included entries for more than 3.6 million U.S. cancer patients during 1973-2013, of whom 6,661 patents had committed suicide, an overall SMR of 1.6.
When the researchers drilled down the SMRs for individual cancer types they found that while the SMR for lung cancer patients throughout the period studied was just above 4, the SMRs for breast and colorectal cancer patients were both 1.4, and 1.2 for patients with prostate cancer. This analysis adjusted for patients’ age, sex, race, and year of diagnosis, Dr. Rahouma reported.
The time from diagnosis to suicide was also strikingly quicker among lung cancer patients, at an average of 8 months, compared with average delays from diagnosis to suicide of 40-60 months for patients with breast, colorectal, or prostate cancer. Dr. Rahouma’s time-trend analysis showed that the SMRs for these three other cancer types held more or less steady within the range of 1-2 throughout the 4 decades examined, and by 2010-2013 the three SMRs all were at or just above 1. Lung cancer was the only malignancy in this group that showed a wide range in SMR over time, with the peak some 30-40 years ago.
Among the lung cancer patient subgroups that showed the highest SMRs for suicide during the entire period studied, men had a SMR of 9, Asians had a SMR of nearly 14, those with a deceased spouse had a SMR for suicide of almost 12, and septuagenarians had a SMR of 12, said Dr. Rahouma. The impact of these risk factors was greatest during the first 8 months following lung cancer diagnosis. After 8 months, the strength of the risk factors diminished, with the SMRs within each risk category dropping by roughly half.
The highest-risk subgroups that the analysis identified should especially be referred for psychiatric support, Dr. Rahouma concluded. “These data will change our practice” at Cornell, he predicted.
Dr. Rahouma had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
WASHINGTON – U.S. patients diagnosed with lung cancer have had the highest suicide rates among patients diagnosed with any of the other most common, non-skin cancers, and they also had a substantially higher suicide risk, compared with the general U.S. adult population, based on U.S. national data collected during 1973-2013.
Although U.S. lung cancer patients showed a “steep” decline in suicide rates starting in about 1985 that then accelerated beginning in the mid-1990s, as recently as 2010-2013 the rate was roughly twice as high in lung cancer patients when compared with the general U.S. adult population. The rate of lung cancer patients taking their lives was also significantly above the suicide rates among patients with breast, colorectal, or prostate cancer, Mohamed Rahouma, MD, reported at an international conference of the American Thoracic Society.
However, he also stressed that identification of lung cancer patients at especially high suicide risk was important to allow “proper psychological assessment, support, and counseling to reduce [suicide] rates.”
Lung cancer patients with the highest rates included men, widowed individuals, septuagenarians, and Asians, his analysis showed. Standardized mortality ratios (SMRs) for suicide of these highest-risk subgroups were near or exceeding 10 times fold higher than the suicide rates of comparable demographic groups among the general U.S. adult population, according to Dr. Rahouma and his associates.
The overall SMR for all lung cancer patients during the entire four decades studied, compared with the overall U.S. adult population, was 4. Even during the period 2005-2013, when suicide among lung cancer patients had fallen to its lowest level, the SMR for this group was still more than 2.
The investigators used data collected by the U.S. Surveillance Epidemiology and End Results (SEER) Program cancer database maintained by the National Cancer Institute. For suicide rates among the general U.S. population they used data from the National Vital Statistics Reports produced by the Centers for Disease Control and Prevention. The SEER database included entries for more than 3.6 million U.S. cancer patients during 1973-2013, of whom 6,661 patents had committed suicide, an overall SMR of 1.6.
When the researchers drilled down the SMRs for individual cancer types they found that while the SMR for lung cancer patients throughout the period studied was just above 4, the SMRs for breast and colorectal cancer patients were both 1.4, and 1.2 for patients with prostate cancer. This analysis adjusted for patients’ age, sex, race, and year of diagnosis, Dr. Rahouma reported.
The time from diagnosis to suicide was also strikingly quicker among lung cancer patients, at an average of 8 months, compared with average delays from diagnosis to suicide of 40-60 months for patients with breast, colorectal, or prostate cancer. Dr. Rahouma’s time-trend analysis showed that the SMRs for these three other cancer types held more or less steady within the range of 1-2 throughout the 4 decades examined, and by 2010-2013 the three SMRs all were at or just above 1. Lung cancer was the only malignancy in this group that showed a wide range in SMR over time, with the peak some 30-40 years ago.
Among the lung cancer patient subgroups that showed the highest SMRs for suicide during the entire period studied, men had a SMR of 9, Asians had a SMR of nearly 14, those with a deceased spouse had a SMR for suicide of almost 12, and septuagenarians had a SMR of 12, said Dr. Rahouma. The impact of these risk factors was greatest during the first 8 months following lung cancer diagnosis. After 8 months, the strength of the risk factors diminished, with the SMRs within each risk category dropping by roughly half.
The highest-risk subgroups that the analysis identified should especially be referred for psychiatric support, Dr. Rahouma concluded. “These data will change our practice” at Cornell, he predicted.
Dr. Rahouma had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
WASHINGTON – U.S. patients diagnosed with lung cancer have had the highest suicide rates among patients diagnosed with any of the other most common, non-skin cancers, and they also had a substantially higher suicide risk, compared with the general U.S. adult population, based on U.S. national data collected during 1973-2013.
Although U.S. lung cancer patients showed a “steep” decline in suicide rates starting in about 1985 that then accelerated beginning in the mid-1990s, as recently as 2010-2013 the rate was roughly twice as high in lung cancer patients when compared with the general U.S. adult population. The rate of lung cancer patients taking their lives was also significantly above the suicide rates among patients with breast, colorectal, or prostate cancer, Mohamed Rahouma, MD, reported at an international conference of the American Thoracic Society.
However, he also stressed that identification of lung cancer patients at especially high suicide risk was important to allow “proper psychological assessment, support, and counseling to reduce [suicide] rates.”
Lung cancer patients with the highest rates included men, widowed individuals, septuagenarians, and Asians, his analysis showed. Standardized mortality ratios (SMRs) for suicide of these highest-risk subgroups were near or exceeding 10 times fold higher than the suicide rates of comparable demographic groups among the general U.S. adult population, according to Dr. Rahouma and his associates.
The overall SMR for all lung cancer patients during the entire four decades studied, compared with the overall U.S. adult population, was 4. Even during the period 2005-2013, when suicide among lung cancer patients had fallen to its lowest level, the SMR for this group was still more than 2.
The investigators used data collected by the U.S. Surveillance Epidemiology and End Results (SEER) Program cancer database maintained by the National Cancer Institute. For suicide rates among the general U.S. population they used data from the National Vital Statistics Reports produced by the Centers for Disease Control and Prevention. The SEER database included entries for more than 3.6 million U.S. cancer patients during 1973-2013, of whom 6,661 patents had committed suicide, an overall SMR of 1.6.
When the researchers drilled down the SMRs for individual cancer types they found that while the SMR for lung cancer patients throughout the period studied was just above 4, the SMRs for breast and colorectal cancer patients were both 1.4, and 1.2 for patients with prostate cancer. This analysis adjusted for patients’ age, sex, race, and year of diagnosis, Dr. Rahouma reported.
The time from diagnosis to suicide was also strikingly quicker among lung cancer patients, at an average of 8 months, compared with average delays from diagnosis to suicide of 40-60 months for patients with breast, colorectal, or prostate cancer. Dr. Rahouma’s time-trend analysis showed that the SMRs for these three other cancer types held more or less steady within the range of 1-2 throughout the 4 decades examined, and by 2010-2013 the three SMRs all were at or just above 1. Lung cancer was the only malignancy in this group that showed a wide range in SMR over time, with the peak some 30-40 years ago.
Among the lung cancer patient subgroups that showed the highest SMRs for suicide during the entire period studied, men had a SMR of 9, Asians had a SMR of nearly 14, those with a deceased spouse had a SMR for suicide of almost 12, and septuagenarians had a SMR of 12, said Dr. Rahouma. The impact of these risk factors was greatest during the first 8 months following lung cancer diagnosis. After 8 months, the strength of the risk factors diminished, with the SMRs within each risk category dropping by roughly half.
The highest-risk subgroups that the analysis identified should especially be referred for psychiatric support, Dr. Rahouma concluded. “These data will change our practice” at Cornell, he predicted.
Dr. Rahouma had no disclosures.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT ATS 2017
Key clinical point:
Major finding: During 1973-2013, suicide among U.S. lung cancer patients was four times higher than the general adult U.S. population.
Data source: Statistics on more than 3.6 million U.S. cancer patients in the SEER Program.
Disclosures: Dr. Rahouma had no disclosures.
New chest x-ray assessment reflects ARDS severity
WASHINGTON – A new way to semiquantitatively score chest x-rays that takes into account lung density and consolidation may be a useful adjunct to current methods for assessing severity of acute respiratory distress syndrome.
The score, know as the Radiographic Assessment of Lung Edema (RALE) score, showed good correlations with lung edema, the severity of acute respiratory distress syndrome (ARDS), and response to fluid management resulting in reduced pulmonary edema, Melissa A. Warren, MD, said at an international conference of the American Thoracic Society.
The RALE score that Dr. Warren and her associates devised rates a patient’s chest x-ray for two parameters: consolidation, which is based on the extent of alveolar opacity in each of the four lung quadrants (left upper, left lower, right upper, and right lower), and a density score that is based on the density of alveolar opacity in each quadrant.
The consolidation score for each quadrant is rated on a 0-4 scale with 0 corresponding to no opacity, 1 for 1%-24% opacity, 2 for 25%-49% opacity, 3 for 50%-75% opacity, and 4 for more than 75%. The density score is rated on a scale of 1-3 with 1 for hazy opacity, 2 for moderate opacity, and 3 for dense opacity. The score for each quadrant is obtained by multiplying the extent score by the density score. A patient’s total RALE score sums the scores from all four quadrants.
The researchers ran three tests of the clinical relevance of this scoring system. First, they used it to score chest x-rays of 72 preprocurement lungs donated for transplant but unable to be used for that purpose, and compared the scores with the extent of lung edema measured by the actual weight of each explanted lung. This showed high correlation between the scores and the amount of edema, Dr. Warren reported.
Next they assessed the RALE score as a marker of ARDS by retrospectively calculating the scores of 174 patients with baseline chest x-rays enrolled in the Fluids and Catheters Treatment Trial (FACTT) (N Engl J Med. 2006;354[24]:2564-75). This analysis showed that patients with the highest RALE scores had significantly worse survival during 90-day follow-up, compared with the patients with the lowest scores.
Finally, the researchers assessed how the RALE score changed in response to either the liberal or conservative fluid management approaches tested in FACTT. This showed that at baseline the average RALE scores were similar among 92 patients randomized to the liberal fluid management treatment arm and 82 patients assigned to the conservative fluid management arm. But after 3 days of treatment, patients in the conservative arm showed a roughly one third reduction in their average RALE score, while patients in the liberal fluid arm showed virtually no change in their score.
“A conservative fluid management strategy favorably impacted the RALE score, reflecting a decrease in pulmonary edema,” Dr. Warren concluded.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
Eric Gartman, MD, FCCP, comments: The results obtained from the use of this scoring system could be important in the prognostication of patients with ARDS, although it is unclear how the score would be used to alter clinical decision making. Further, issues may arise in its implementation given the somewhat subjective nature of the scoring (e.g., hazy vs. moderate vs. dense opacity), changing factors in the ICU that may affect the lung density on x-ray (e.g., different levels of positive end-expiratory pressure), and the variable quality of chest x-rays in the ICU.
Eric Gartman, MD, FCCP, comments: The results obtained from the use of this scoring system could be important in the prognostication of patients with ARDS, although it is unclear how the score would be used to alter clinical decision making. Further, issues may arise in its implementation given the somewhat subjective nature of the scoring (e.g., hazy vs. moderate vs. dense opacity), changing factors in the ICU that may affect the lung density on x-ray (e.g., different levels of positive end-expiratory pressure), and the variable quality of chest x-rays in the ICU.
Eric Gartman, MD, FCCP, comments: The results obtained from the use of this scoring system could be important in the prognostication of patients with ARDS, although it is unclear how the score would be used to alter clinical decision making. Further, issues may arise in its implementation given the somewhat subjective nature of the scoring (e.g., hazy vs. moderate vs. dense opacity), changing factors in the ICU that may affect the lung density on x-ray (e.g., different levels of positive end-expiratory pressure), and the variable quality of chest x-rays in the ICU.
WASHINGTON – A new way to semiquantitatively score chest x-rays that takes into account lung density and consolidation may be a useful adjunct to current methods for assessing severity of acute respiratory distress syndrome.
The score, know as the Radiographic Assessment of Lung Edema (RALE) score, showed good correlations with lung edema, the severity of acute respiratory distress syndrome (ARDS), and response to fluid management resulting in reduced pulmonary edema, Melissa A. Warren, MD, said at an international conference of the American Thoracic Society.
The RALE score that Dr. Warren and her associates devised rates a patient’s chest x-ray for two parameters: consolidation, which is based on the extent of alveolar opacity in each of the four lung quadrants (left upper, left lower, right upper, and right lower), and a density score that is based on the density of alveolar opacity in each quadrant.
The consolidation score for each quadrant is rated on a 0-4 scale with 0 corresponding to no opacity, 1 for 1%-24% opacity, 2 for 25%-49% opacity, 3 for 50%-75% opacity, and 4 for more than 75%. The density score is rated on a scale of 1-3 with 1 for hazy opacity, 2 for moderate opacity, and 3 for dense opacity. The score for each quadrant is obtained by multiplying the extent score by the density score. A patient’s total RALE score sums the scores from all four quadrants.
The researchers ran three tests of the clinical relevance of this scoring system. First, they used it to score chest x-rays of 72 preprocurement lungs donated for transplant but unable to be used for that purpose, and compared the scores with the extent of lung edema measured by the actual weight of each explanted lung. This showed high correlation between the scores and the amount of edema, Dr. Warren reported.
Next they assessed the RALE score as a marker of ARDS by retrospectively calculating the scores of 174 patients with baseline chest x-rays enrolled in the Fluids and Catheters Treatment Trial (FACTT) (N Engl J Med. 2006;354[24]:2564-75). This analysis showed that patients with the highest RALE scores had significantly worse survival during 90-day follow-up, compared with the patients with the lowest scores.
Finally, the researchers assessed how the RALE score changed in response to either the liberal or conservative fluid management approaches tested in FACTT. This showed that at baseline the average RALE scores were similar among 92 patients randomized to the liberal fluid management treatment arm and 82 patients assigned to the conservative fluid management arm. But after 3 days of treatment, patients in the conservative arm showed a roughly one third reduction in their average RALE score, while patients in the liberal fluid arm showed virtually no change in their score.
“A conservative fluid management strategy favorably impacted the RALE score, reflecting a decrease in pulmonary edema,” Dr. Warren concluded.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
WASHINGTON – A new way to semiquantitatively score chest x-rays that takes into account lung density and consolidation may be a useful adjunct to current methods for assessing severity of acute respiratory distress syndrome.
The score, know as the Radiographic Assessment of Lung Edema (RALE) score, showed good correlations with lung edema, the severity of acute respiratory distress syndrome (ARDS), and response to fluid management resulting in reduced pulmonary edema, Melissa A. Warren, MD, said at an international conference of the American Thoracic Society.
The RALE score that Dr. Warren and her associates devised rates a patient’s chest x-ray for two parameters: consolidation, which is based on the extent of alveolar opacity in each of the four lung quadrants (left upper, left lower, right upper, and right lower), and a density score that is based on the density of alveolar opacity in each quadrant.
The consolidation score for each quadrant is rated on a 0-4 scale with 0 corresponding to no opacity, 1 for 1%-24% opacity, 2 for 25%-49% opacity, 3 for 50%-75% opacity, and 4 for more than 75%. The density score is rated on a scale of 1-3 with 1 for hazy opacity, 2 for moderate opacity, and 3 for dense opacity. The score for each quadrant is obtained by multiplying the extent score by the density score. A patient’s total RALE score sums the scores from all four quadrants.
The researchers ran three tests of the clinical relevance of this scoring system. First, they used it to score chest x-rays of 72 preprocurement lungs donated for transplant but unable to be used for that purpose, and compared the scores with the extent of lung edema measured by the actual weight of each explanted lung. This showed high correlation between the scores and the amount of edema, Dr. Warren reported.
Next they assessed the RALE score as a marker of ARDS by retrospectively calculating the scores of 174 patients with baseline chest x-rays enrolled in the Fluids and Catheters Treatment Trial (FACTT) (N Engl J Med. 2006;354[24]:2564-75). This analysis showed that patients with the highest RALE scores had significantly worse survival during 90-day follow-up, compared with the patients with the lowest scores.
Finally, the researchers assessed how the RALE score changed in response to either the liberal or conservative fluid management approaches tested in FACTT. This showed that at baseline the average RALE scores were similar among 92 patients randomized to the liberal fluid management treatment arm and 82 patients assigned to the conservative fluid management arm. But after 3 days of treatment, patients in the conservative arm showed a roughly one third reduction in their average RALE score, while patients in the liberal fluid arm showed virtually no change in their score.
“A conservative fluid management strategy favorably impacted the RALE score, reflecting a decrease in pulmonary edema,” Dr. Warren concluded.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
AT ATS 2017
Key clinical point:
Major finding: The RALE score correlated with lung edema and ARDS severity, and changed in response to conservative fluid management.
Data source: A subgroup of 174 patients with ARDS enrolled in the FACTT trial, and also 72 donor lungs not available for transplantation.
Disclosures: Dr. Warren had no disclosures.
A prescription for heart failure success: Change the name
PARIS – Does heart failure’s name doom any progress against the disease?
That was the provocative premise advanced by Lynne Warner Stevenson, MD, who suggested that efforts to prevent, diagnose, and treat the disease would go better if it could only jettison that unfortunate word “failure,” its hard-wired albatross.
Dr. Stevenson offered several potentially superior alternatives, including cardiac insufficiency, heart dysfunction, and her favorite, cardiomyopathy.
“Is heart failure still the best diagnosis” for the entire spectrum of disease that most patients progress through ,including the many patients in earlier stages of the disease who do not have a truly failing heart? “Perhaps cardiomyopathy is the condition and heart failure is the transition,” she proposed.
To Dr. Stevenson, it’s more than just semantics.
“Words are hugely powerful,” she explained in an interview following her talk. “I think patients do not want to be seen as having heart failure. They don’t want to think of themselves as having heart failure. I think it can make them delay getting care, and it makes them ignore the disease. I worry about that a lot. I also worry that patients don’t provide support to each other that they could. Patients tend to hide that they have heart failure. We need to come up with a term that does not make patients ashamed of their disease.”
Part of the problem, Dr. Stevenson said, is that the name heart failure can be very misleading depending on the stage of the disease that patients have. Patients with stage B (presymptomatic) disease and those with mild stage C disease “don’t see themselves as having heart failure,” as having a heart that has failed them. “We need to be able to convince these patients that they have a disease that we need to treat carefully and aggressively.”
Additionally, labeling tens of millions of people as having stage A heart failure, which is presymptomatic and occurs before the heart shows any sign of damage or dysfunction, is also counterproductive, maintained Dr. Stevenson, professor of medicine at Harvard Medical School and director of the Cardiomyopathy and Heart Failure Program at Brigham and Women’s Hospital in Boston.
“So many people are at risk of developing heart failure,” she noted, including patients with hypertension, diabetes, or coronary artery disease. To label them all as already also having heart failure at that stage “tends to make them ignore the disease that we are trying to get them to pay attention to. Telling patients they have the disease that we are trying to prevent doesn’t help.”
Calling the whole range of the disease heart failure also confuses patients and others. “Patients ask me, ‘How can I have heart failure without any symptoms?’ ‘My ejection fraction improved to almost normal; do I still have heart failure?’ and ‘I don’t understand how my heart muscle is strong but my heart is failing,’ ” she said
For Dr. Stevenson, perhaps the biggest problem is the stigma of failure and the way that word ties a huge weight to the disease that prompts patients and caregivers alike to relegate it to a hidden and neglected place.
“It’s failure. Who is proud to have heart failure? Where are the marches for heart failure? Where are the celebrity champions for heart failure? We have celebrities who are happy to admit that they have Parkinson’s disease, ALS [amyotrophic lateral sclerosis], drug addiction, and even erectile dysfunction, but no one wants to say they have heart failure. We can’t get any traction behind heart failure. It doesn’t sound very inspiring,” an issue that even percolates down to dissuading clinicians from pursuing a career in heart failure care. Young people do not aspire to go into failure, she said.
“We need to call it something else.”
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
PARIS – Does heart failure’s name doom any progress against the disease?
That was the provocative premise advanced by Lynne Warner Stevenson, MD, who suggested that efforts to prevent, diagnose, and treat the disease would go better if it could only jettison that unfortunate word “failure,” its hard-wired albatross.
Dr. Stevenson offered several potentially superior alternatives, including cardiac insufficiency, heart dysfunction, and her favorite, cardiomyopathy.
“Is heart failure still the best diagnosis” for the entire spectrum of disease that most patients progress through ,including the many patients in earlier stages of the disease who do not have a truly failing heart? “Perhaps cardiomyopathy is the condition and heart failure is the transition,” she proposed.
To Dr. Stevenson, it’s more than just semantics.
“Words are hugely powerful,” she explained in an interview following her talk. “I think patients do not want to be seen as having heart failure. They don’t want to think of themselves as having heart failure. I think it can make them delay getting care, and it makes them ignore the disease. I worry about that a lot. I also worry that patients don’t provide support to each other that they could. Patients tend to hide that they have heart failure. We need to come up with a term that does not make patients ashamed of their disease.”
Part of the problem, Dr. Stevenson said, is that the name heart failure can be very misleading depending on the stage of the disease that patients have. Patients with stage B (presymptomatic) disease and those with mild stage C disease “don’t see themselves as having heart failure,” as having a heart that has failed them. “We need to be able to convince these patients that they have a disease that we need to treat carefully and aggressively.”
Additionally, labeling tens of millions of people as having stage A heart failure, which is presymptomatic and occurs before the heart shows any sign of damage or dysfunction, is also counterproductive, maintained Dr. Stevenson, professor of medicine at Harvard Medical School and director of the Cardiomyopathy and Heart Failure Program at Brigham and Women’s Hospital in Boston.
“So many people are at risk of developing heart failure,” she noted, including patients with hypertension, diabetes, or coronary artery disease. To label them all as already also having heart failure at that stage “tends to make them ignore the disease that we are trying to get them to pay attention to. Telling patients they have the disease that we are trying to prevent doesn’t help.”
Calling the whole range of the disease heart failure also confuses patients and others. “Patients ask me, ‘How can I have heart failure without any symptoms?’ ‘My ejection fraction improved to almost normal; do I still have heart failure?’ and ‘I don’t understand how my heart muscle is strong but my heart is failing,’ ” she said
For Dr. Stevenson, perhaps the biggest problem is the stigma of failure and the way that word ties a huge weight to the disease that prompts patients and caregivers alike to relegate it to a hidden and neglected place.
“It’s failure. Who is proud to have heart failure? Where are the marches for heart failure? Where are the celebrity champions for heart failure? We have celebrities who are happy to admit that they have Parkinson’s disease, ALS [amyotrophic lateral sclerosis], drug addiction, and even erectile dysfunction, but no one wants to say they have heart failure. We can’t get any traction behind heart failure. It doesn’t sound very inspiring,” an issue that even percolates down to dissuading clinicians from pursuing a career in heart failure care. Young people do not aspire to go into failure, she said.
“We need to call it something else.”
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
PARIS – Does heart failure’s name doom any progress against the disease?
That was the provocative premise advanced by Lynne Warner Stevenson, MD, who suggested that efforts to prevent, diagnose, and treat the disease would go better if it could only jettison that unfortunate word “failure,” its hard-wired albatross.
Dr. Stevenson offered several potentially superior alternatives, including cardiac insufficiency, heart dysfunction, and her favorite, cardiomyopathy.
“Is heart failure still the best diagnosis” for the entire spectrum of disease that most patients progress through ,including the many patients in earlier stages of the disease who do not have a truly failing heart? “Perhaps cardiomyopathy is the condition and heart failure is the transition,” she proposed.
To Dr. Stevenson, it’s more than just semantics.
“Words are hugely powerful,” she explained in an interview following her talk. “I think patients do not want to be seen as having heart failure. They don’t want to think of themselves as having heart failure. I think it can make them delay getting care, and it makes them ignore the disease. I worry about that a lot. I also worry that patients don’t provide support to each other that they could. Patients tend to hide that they have heart failure. We need to come up with a term that does not make patients ashamed of their disease.”
Part of the problem, Dr. Stevenson said, is that the name heart failure can be very misleading depending on the stage of the disease that patients have. Patients with stage B (presymptomatic) disease and those with mild stage C disease “don’t see themselves as having heart failure,” as having a heart that has failed them. “We need to be able to convince these patients that they have a disease that we need to treat carefully and aggressively.”
Additionally, labeling tens of millions of people as having stage A heart failure, which is presymptomatic and occurs before the heart shows any sign of damage or dysfunction, is also counterproductive, maintained Dr. Stevenson, professor of medicine at Harvard Medical School and director of the Cardiomyopathy and Heart Failure Program at Brigham and Women’s Hospital in Boston.
“So many people are at risk of developing heart failure,” she noted, including patients with hypertension, diabetes, or coronary artery disease. To label them all as already also having heart failure at that stage “tends to make them ignore the disease that we are trying to get them to pay attention to. Telling patients they have the disease that we are trying to prevent doesn’t help.”
Calling the whole range of the disease heart failure also confuses patients and others. “Patients ask me, ‘How can I have heart failure without any symptoms?’ ‘My ejection fraction improved to almost normal; do I still have heart failure?’ and ‘I don’t understand how my heart muscle is strong but my heart is failing,’ ” she said
For Dr. Stevenson, perhaps the biggest problem is the stigma of failure and the way that word ties a huge weight to the disease that prompts patients and caregivers alike to relegate it to a hidden and neglected place.
“It’s failure. Who is proud to have heart failure? Where are the marches for heart failure? Where are the celebrity champions for heart failure? We have celebrities who are happy to admit that they have Parkinson’s disease, ALS [amyotrophic lateral sclerosis], drug addiction, and even erectile dysfunction, but no one wants to say they have heart failure. We can’t get any traction behind heart failure. It doesn’t sound very inspiring,” an issue that even percolates down to dissuading clinicians from pursuing a career in heart failure care. Young people do not aspire to go into failure, she said.
“We need to call it something else.”
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM HEART FAILURE 2017
VIDEO: Hyperinflammatory ARDS responds to simvastatin
WASHINGTON – Acute respiratory distress syndrome (ARDS) appears to exist in at least two major forms, and one of these, the hyperinflammatory form, seemed responsive to simvastatin in a post-hoc analysis of trial data.
The other version of ARDs is a hypoinflammatory form, which occurred in 70% of ARDS patients in most of the analyses that have been done.
Researchers classified the 540 ARDS patients enrolled in a 2014 study of simvastatin as either hyperinflammatory or hypoinflammatory. Separating out the hyperinflammatory patients created a subclass that responded to simvastatin, with a 13% absolute reduction in mortality during follow-up, compared with no response among patients in the hypoinflammatory group, Carolyn S. Calfee, MD, said at an international conference of the American Thoracic Society.
“Hyperinflammatory patients treated with simvastatin may have improved outcomes, compared with hypoinflammatory* patients treated with placebo,” said Dr. Calfee, a pulmonologist at the University of California, San Francisco.
The finding raises the possibility that simvastatin, as well as other statins, may be an effective treatment for selected patients with ARDS, but proving this requires new prospective, randomized trials in hyperinflammatory patients, Dr. Calfee said in a video interview.
Currently, the tests Dr. Calfee uses to distinguish hyperinflammatory and hypoinflammatory ARDS patients take about 6-8 hours to complete. A “point of care test to stratify patients in real time,” is needed to further study the various forms of ARDs, Dr. Calfee noted. A critical next step would be the development of a “practical, rapid, bedside assay” to ease identification of hyperinflammatory ARDS patients. “The work we’ve done prior seems to indicate that we are going to definitely need to measure biomarkers in order to identity these subgroups,” she noted.
Hypoinflammatory patients also merit study, she added. Although hyperinflammatory patients have significant worse mortality rates, the hypoinflammatory subclass includes about 70% of ARDS patients, “so we need to better understand how to potentially treat this group.”
Dr. Calfee and her associates first reported finding the two ARDS subclasses, what they also call subphenotypes or endotypes, in two separate cohorts of ARDS patients in a 2014 report (Lancet Resp Med. 2014 Aug;2[8]:611-20). Then, they confirmed the finding in a third ARDS cohort in a 2017 report (Amer J Resp Crit Care Med. 2017 Feb 1;195[3]:331-8). These reports have documented other characteristics of the hyperinflammatory ARDS subclass: hypotension, metabolic acidosis, more frequent treatment with vasopressors, and a higher prevalence of sepsis and shock. Concurrent with the 2017 report, an editorial hailed the finding as “the dawn of personalized medicine for ARDS” (Amer J resp Crit Care Med. 2017 Feb 1;195[3]: 280-1).
To build on this, Dr. Calfee and her associates applied their method for identifying ARDS subclasses to a different cohort of 540 patients enrolled in the The HARP (Hydroxymethylglutaryl-CoA Reductase Inhibition with Simvastatin in Acute Lung Injury to Reduce Pulmonary Dysfunction)–2 study, a multicenter UK and Irish study designed to test the efficacy of daily simvastatin treatment in a heterogeneous group of ARDS patients. A 2014 report of the study’s primary results showed no significant effect from simvastatin for increasing the number of ventilator-free days nor did the drug improve any other measured efficacy endpoints (New Engl J Med. 2014 Oct 30;371[18]:1695-703).
Applying a statistical analysis called “latent class analysis,” which is designed to recognize subclass groupings that might not be readily apparent, Dr. Calfee and her team first confirmed that, in this fourth cohort, the ARDS patients again split into a hyperinflammatory subclass, in this case including 188 (35%) of the cohort, and a hypoinflammatory subclass with 352 (65%) patients. The next step was to see what impact simvastatin treatment had in each of the two patient subclasses. They focused the analysis on a secondary outcome in HARP-2, 28-day survival.
They found that simvastatin produced no significant difference in 28-day survival, compared with placebo among the hypoinflammatory patients, but, in the hyperinflammatory subclass, 28-day survival was 68% for patients on simvastatin and 55% for those on placebo, a statistically significant difference, Dr. Calfee reported (Am J Resp Crit Care Med. 2017;195:A6749).
“I’m excited that we are seeing, for the first time, a different response to pharmacotherapy” after dividing ARDS patients into these two subclasses, she said. But, the work remains in an early stage, she cautioned. “We need to test treatments [like statins] prospectively.” The new finding for simvastatin “is not the same as showing benefit in a prospective, randomized trial.”
In the meantime, Dr. Calfee plans to apply the same analytic approach to data collected in another failed statin trial in ARDS patients, the SAILS trial. That study failed to show benefit from rosuvastatin treatment in an unselected population of patients with sepsis-associated ARDS (New Engl J Med. 2014 June 5;370[23]:2191-200).
Dr. Calfee is a consultant to Bayer, Boehringer Ingelheim, and GlaxoSmithKline. She received research funding from GlaxoSmithKline.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
*An earlier version of this article misquoted Dr. Calfee.
WASHINGTON – Acute respiratory distress syndrome (ARDS) appears to exist in at least two major forms, and one of these, the hyperinflammatory form, seemed responsive to simvastatin in a post-hoc analysis of trial data.
The other version of ARDs is a hypoinflammatory form, which occurred in 70% of ARDS patients in most of the analyses that have been done.
Researchers classified the 540 ARDS patients enrolled in a 2014 study of simvastatin as either hyperinflammatory or hypoinflammatory. Separating out the hyperinflammatory patients created a subclass that responded to simvastatin, with a 13% absolute reduction in mortality during follow-up, compared with no response among patients in the hypoinflammatory group, Carolyn S. Calfee, MD, said at an international conference of the American Thoracic Society.
“Hyperinflammatory patients treated with simvastatin may have improved outcomes, compared with hypoinflammatory* patients treated with placebo,” said Dr. Calfee, a pulmonologist at the University of California, San Francisco.
The finding raises the possibility that simvastatin, as well as other statins, may be an effective treatment for selected patients with ARDS, but proving this requires new prospective, randomized trials in hyperinflammatory patients, Dr. Calfee said in a video interview.
Currently, the tests Dr. Calfee uses to distinguish hyperinflammatory and hypoinflammatory ARDS patients take about 6-8 hours to complete. A “point of care test to stratify patients in real time,” is needed to further study the various forms of ARDs, Dr. Calfee noted. A critical next step would be the development of a “practical, rapid, bedside assay” to ease identification of hyperinflammatory ARDS patients. “The work we’ve done prior seems to indicate that we are going to definitely need to measure biomarkers in order to identity these subgroups,” she noted.
Hypoinflammatory patients also merit study, she added. Although hyperinflammatory patients have significant worse mortality rates, the hypoinflammatory subclass includes about 70% of ARDS patients, “so we need to better understand how to potentially treat this group.”
Dr. Calfee and her associates first reported finding the two ARDS subclasses, what they also call subphenotypes or endotypes, in two separate cohorts of ARDS patients in a 2014 report (Lancet Resp Med. 2014 Aug;2[8]:611-20). Then, they confirmed the finding in a third ARDS cohort in a 2017 report (Amer J Resp Crit Care Med. 2017 Feb 1;195[3]:331-8). These reports have documented other characteristics of the hyperinflammatory ARDS subclass: hypotension, metabolic acidosis, more frequent treatment with vasopressors, and a higher prevalence of sepsis and shock. Concurrent with the 2017 report, an editorial hailed the finding as “the dawn of personalized medicine for ARDS” (Amer J resp Crit Care Med. 2017 Feb 1;195[3]: 280-1).
To build on this, Dr. Calfee and her associates applied their method for identifying ARDS subclasses to a different cohort of 540 patients enrolled in the The HARP (Hydroxymethylglutaryl-CoA Reductase Inhibition with Simvastatin in Acute Lung Injury to Reduce Pulmonary Dysfunction)–2 study, a multicenter UK and Irish study designed to test the efficacy of daily simvastatin treatment in a heterogeneous group of ARDS patients. A 2014 report of the study’s primary results showed no significant effect from simvastatin for increasing the number of ventilator-free days nor did the drug improve any other measured efficacy endpoints (New Engl J Med. 2014 Oct 30;371[18]:1695-703).
Applying a statistical analysis called “latent class analysis,” which is designed to recognize subclass groupings that might not be readily apparent, Dr. Calfee and her team first confirmed that, in this fourth cohort, the ARDS patients again split into a hyperinflammatory subclass, in this case including 188 (35%) of the cohort, and a hypoinflammatory subclass with 352 (65%) patients. The next step was to see what impact simvastatin treatment had in each of the two patient subclasses. They focused the analysis on a secondary outcome in HARP-2, 28-day survival.
They found that simvastatin produced no significant difference in 28-day survival, compared with placebo among the hypoinflammatory patients, but, in the hyperinflammatory subclass, 28-day survival was 68% for patients on simvastatin and 55% for those on placebo, a statistically significant difference, Dr. Calfee reported (Am J Resp Crit Care Med. 2017;195:A6749).
“I’m excited that we are seeing, for the first time, a different response to pharmacotherapy” after dividing ARDS patients into these two subclasses, she said. But, the work remains in an early stage, she cautioned. “We need to test treatments [like statins] prospectively.” The new finding for simvastatin “is not the same as showing benefit in a prospective, randomized trial.”
In the meantime, Dr. Calfee plans to apply the same analytic approach to data collected in another failed statin trial in ARDS patients, the SAILS trial. That study failed to show benefit from rosuvastatin treatment in an unselected population of patients with sepsis-associated ARDS (New Engl J Med. 2014 June 5;370[23]:2191-200).
Dr. Calfee is a consultant to Bayer, Boehringer Ingelheim, and GlaxoSmithKline. She received research funding from GlaxoSmithKline.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
*An earlier version of this article misquoted Dr. Calfee.
WASHINGTON – Acute respiratory distress syndrome (ARDS) appears to exist in at least two major forms, and one of these, the hyperinflammatory form, seemed responsive to simvastatin in a post-hoc analysis of trial data.
The other version of ARDs is a hypoinflammatory form, which occurred in 70% of ARDS patients in most of the analyses that have been done.
Researchers classified the 540 ARDS patients enrolled in a 2014 study of simvastatin as either hyperinflammatory or hypoinflammatory. Separating out the hyperinflammatory patients created a subclass that responded to simvastatin, with a 13% absolute reduction in mortality during follow-up, compared with no response among patients in the hypoinflammatory group, Carolyn S. Calfee, MD, said at an international conference of the American Thoracic Society.
“Hyperinflammatory patients treated with simvastatin may have improved outcomes, compared with hypoinflammatory* patients treated with placebo,” said Dr. Calfee, a pulmonologist at the University of California, San Francisco.
The finding raises the possibility that simvastatin, as well as other statins, may be an effective treatment for selected patients with ARDS, but proving this requires new prospective, randomized trials in hyperinflammatory patients, Dr. Calfee said in a video interview.
Currently, the tests Dr. Calfee uses to distinguish hyperinflammatory and hypoinflammatory ARDS patients take about 6-8 hours to complete. A “point of care test to stratify patients in real time,” is needed to further study the various forms of ARDs, Dr. Calfee noted. A critical next step would be the development of a “practical, rapid, bedside assay” to ease identification of hyperinflammatory ARDS patients. “The work we’ve done prior seems to indicate that we are going to definitely need to measure biomarkers in order to identity these subgroups,” she noted.
Hypoinflammatory patients also merit study, she added. Although hyperinflammatory patients have significant worse mortality rates, the hypoinflammatory subclass includes about 70% of ARDS patients, “so we need to better understand how to potentially treat this group.”
Dr. Calfee and her associates first reported finding the two ARDS subclasses, what they also call subphenotypes or endotypes, in two separate cohorts of ARDS patients in a 2014 report (Lancet Resp Med. 2014 Aug;2[8]:611-20). Then, they confirmed the finding in a third ARDS cohort in a 2017 report (Amer J Resp Crit Care Med. 2017 Feb 1;195[3]:331-8). These reports have documented other characteristics of the hyperinflammatory ARDS subclass: hypotension, metabolic acidosis, more frequent treatment with vasopressors, and a higher prevalence of sepsis and shock. Concurrent with the 2017 report, an editorial hailed the finding as “the dawn of personalized medicine for ARDS” (Amer J resp Crit Care Med. 2017 Feb 1;195[3]: 280-1).
To build on this, Dr. Calfee and her associates applied their method for identifying ARDS subclasses to a different cohort of 540 patients enrolled in the The HARP (Hydroxymethylglutaryl-CoA Reductase Inhibition with Simvastatin in Acute Lung Injury to Reduce Pulmonary Dysfunction)–2 study, a multicenter UK and Irish study designed to test the efficacy of daily simvastatin treatment in a heterogeneous group of ARDS patients. A 2014 report of the study’s primary results showed no significant effect from simvastatin for increasing the number of ventilator-free days nor did the drug improve any other measured efficacy endpoints (New Engl J Med. 2014 Oct 30;371[18]:1695-703).
Applying a statistical analysis called “latent class analysis,” which is designed to recognize subclass groupings that might not be readily apparent, Dr. Calfee and her team first confirmed that, in this fourth cohort, the ARDS patients again split into a hyperinflammatory subclass, in this case including 188 (35%) of the cohort, and a hypoinflammatory subclass with 352 (65%) patients. The next step was to see what impact simvastatin treatment had in each of the two patient subclasses. They focused the analysis on a secondary outcome in HARP-2, 28-day survival.
They found that simvastatin produced no significant difference in 28-day survival, compared with placebo among the hypoinflammatory patients, but, in the hyperinflammatory subclass, 28-day survival was 68% for patients on simvastatin and 55% for those on placebo, a statistically significant difference, Dr. Calfee reported (Am J Resp Crit Care Med. 2017;195:A6749).
“I’m excited that we are seeing, for the first time, a different response to pharmacotherapy” after dividing ARDS patients into these two subclasses, she said. But, the work remains in an early stage, she cautioned. “We need to test treatments [like statins] prospectively.” The new finding for simvastatin “is not the same as showing benefit in a prospective, randomized trial.”
In the meantime, Dr. Calfee plans to apply the same analytic approach to data collected in another failed statin trial in ARDS patients, the SAILS trial. That study failed to show benefit from rosuvastatin treatment in an unselected population of patients with sepsis-associated ARDS (New Engl J Med. 2014 June 5;370[23]:2191-200).
Dr. Calfee is a consultant to Bayer, Boehringer Ingelheim, and GlaxoSmithKline. She received research funding from GlaxoSmithKline.
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
*An earlier version of this article misquoted Dr. Calfee.
AT ATS 2017
Key clinical point:
Major finding: Among hyperinflammatory ARDS patients, 28-day survival was 68% with simvastatin and 55% with placebo, a statistically significant difference.
Data source: A post-hoc analysis of a multicenter randomized trial of 540 patients.
Disclosures: Dr. Calfee is a consultant to Bayer, Boehringer Ingelheim, and GlaxoSmithKline. She received research funding from GlaxoSmithKline.



















