User login
RA Assessment Via Automated Ultrasound Scanner With AI Saves Time, Performs as Well as Rheumatologists
WASHINGTON — A fully automated ultrasound scanning system combined with artificial intelligence–based disease activity scoring performed as well as expert rheumatologists in hand joint assessment of patients with rheumatoid arthritis (RA), new research found.
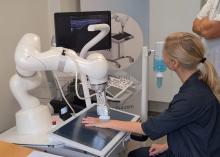
The system, made by a Danish company called ROPCA, comprises an ultrasound scanner called ARTHUR (RA Ultrasound Robot) that interacts directly with the patient and scans 11 joints per hand and a neural network–based software system, DIANA (Diagnosis Aid Network for RA), that evaluates the images and monitors RA activity.
The combined system classifies the degree of RA according to the joint European Alliance of Associations for Rheumatology (EULAR)–Outcome Measures in Rheumatology (OMERACT) standards for RA diagnosis. It received a CE Mark in Europe in 2022 and is currently in use in six rheumatology clinics in Denmark, Germany, Switzerland, and Austria, with more to come, ROPCA Co-founder and Chief Medical Officer Søren A. Just, MD, said in an interview.
“Automated systems could help rheumatologists in the early detection and monitoring of arthritis diseases. Systems can be placed or move in areas with insufficient rheumatological expertise,” Just said during a special late-breaker session presentation at the annual meeting of the American College of Rheumatology (ACR).
He said in an interview: “Currently, there are so many people referred and few and fewer rheumatologists. So we need to think differently. We need good automated assistants.” As a screening tool, the system can determine whether a person with hand pain has RA or just osteoarthritis “and also can give the patient an immediate answer, instead of waiting sometimes up to 6 months to get the information.”
Just, who is also a senior physician in the Department of Internal Medicine at Odense University Hospital in Denmark, said that his department is also using the system to assess flares in patients with established RA. “They can have a blood sample taken. They’re scanned by the robot, and you can see if there is any disease activity. But I think that screening of patients with joint pain is the beginning.”
Asked to comment, session moderator Gregory C. Gardner, MD, Emeritus Professor in the Division of Rheumatology at the University of Washington, Seattle, and a member of the ACR conference program committee, said in an interview “one of the reasons we chose to feature this abstract is because we’re interested in science at the convergence. We really thought this was a potential way to move the field forward for rheumatologists.”
Gardner said it’s an advantage that the patient could potentially have an ARTHUR scan with a DIANA report and get blood tests done prior to a visit with the rheumatologist. “It’s really time-consuming for a human to do these studies, so if you automate it, that’s a step forward in terms of having the data available for the rheumatologist to view and use sequentially to follow how patients are doing.”
When introducing Just’s presentation, Gardner called it “the coolest abstract of the meeting.”
Both DIANA and ARTHUR Performed At Least as Well as Human Rheumatologists
In the study, 30 patients with RA underwent two scans by ARTHUR, followed by a scan from a rheumatologist specialist in musculoskeletal ultrasound. The scans were sent to DIANA, who graded the images according to the Global OMERACT-EULAR Synovitis Score, as did the human rheumatologist.
A “ground truth” was established by another human expert who evaluated both ARTHUR’s and the other rheumatologist’s images, blinded to the scanning method. The image with the highest disease activity was deemed “ground truth,” and agreement with that was assessed for the two individual methods.
Just showed a video of a patient being scanned by ARTHUR. The machine verbally guided her through removing her jewelry, applying the gel, and placing her hand on the screen under the scanner. ARTHUR’s arm moved around on the patient’s hand, locating the best angles to take grayscale images and Doppler images and Doppler video. The scan takes 15-20 minutes, and the images are stored, Just said.
The study patients had a mean age of 65 years, and 23 of the 30 were men. Their average disease duration was 11 years, and mean Disease Activity Score in 28 joints using C-reactive protein was 3.86, indicating moderate disease. A majority (73%) of patients were taking disease-modifying antirheumatic drugs, and about one third were taking biologics. ARTHUR scanned a total of 660 joints, and 564 scans were successful.
For repeatability between the two ARTHUR scans, percent exact agreement was 63% for synovial hypertrophy, 75% for Doppler activity, and 60% combined. Percent close (within a point) agreements were 93%, 94%, and 92%, respectively. Binary agreements as to whether the joint was healthy vs diseased were 88%, 91%, and 85%, respectively.
At the joint level, ARTHUR and DIANA’s percent exact agreement with ground truth was 49% for synovial hypertrophy, 63% for Doppler activity, and 48% combined. Binary agreements with disease vs healthy were 80%, 88%, and 78%, respectively.
The human rheumatologists scored very similarly. Percent exact agreement with ground truth was 51% for synovial hypertrophy, 64% for Doppler activity, and 50% combined. Percent close agreements were 94%, 94%, and 92%, respectively. And binary agreements with diseased vs healthy were 83%, 91%, and 80%, respectively.
At the patient level (all joints combined), ARTHUR and DIANA’s binary disease assessment of healthy vs disease showed agreement with the ground truth of 87% for synovial hypertrophy, 83% for Doppler activity, and 87% combined. Here, the rheumatologists scored lower, at 53%, 67%, and 60%, respectively.
“In this study, we think the precision of ARTHUR and DIANA was comparable to that of an experienced rheumatologist, at both the joint and patient level,” Just said.
Gardner pointed out another advantage of the system. “DIANA doesn’t get fatigued. ... With human reading, the precision may change based on the time of day or stress level. ... But with DIANA, you’re going to get consistent information.”
Just said that the Arthritis Foundation in Germany recently put ARTHUR and DIANA on a bus and took it to cities that lacked a rheumatologist. Patients lined up, answered a questionnaire, had blood drawn, and received their scans. A rheumatologist on the bus then interpreted the data and consulted with the individuals about their RA risk. “In the last trip, we screened 800 patients in 6 days. So there are definitely possibilities here.”
Just is co-owner of ROPCA. Gardner had no disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — A fully automated ultrasound scanning system combined with artificial intelligence–based disease activity scoring performed as well as expert rheumatologists in hand joint assessment of patients with rheumatoid arthritis (RA), new research found.
The system, made by a Danish company called ROPCA, comprises an ultrasound scanner called ARTHUR (RA Ultrasound Robot) that interacts directly with the patient and scans 11 joints per hand and a neural network–based software system, DIANA (Diagnosis Aid Network for RA), that evaluates the images and monitors RA activity.
The combined system classifies the degree of RA according to the joint European Alliance of Associations for Rheumatology (EULAR)–Outcome Measures in Rheumatology (OMERACT) standards for RA diagnosis. It received a CE Mark in Europe in 2022 and is currently in use in six rheumatology clinics in Denmark, Germany, Switzerland, and Austria, with more to come, ROPCA Co-founder and Chief Medical Officer Søren A. Just, MD, said in an interview.
“Automated systems could help rheumatologists in the early detection and monitoring of arthritis diseases. Systems can be placed or move in areas with insufficient rheumatological expertise,” Just said during a special late-breaker session presentation at the annual meeting of the American College of Rheumatology (ACR).
He said in an interview: “Currently, there are so many people referred and few and fewer rheumatologists. So we need to think differently. We need good automated assistants.” As a screening tool, the system can determine whether a person with hand pain has RA or just osteoarthritis “and also can give the patient an immediate answer, instead of waiting sometimes up to 6 months to get the information.”
Just, who is also a senior physician in the Department of Internal Medicine at Odense University Hospital in Denmark, said that his department is also using the system to assess flares in patients with established RA. “They can have a blood sample taken. They’re scanned by the robot, and you can see if there is any disease activity. But I think that screening of patients with joint pain is the beginning.”
Asked to comment, session moderator Gregory C. Gardner, MD, Emeritus Professor in the Division of Rheumatology at the University of Washington, Seattle, and a member of the ACR conference program committee, said in an interview “one of the reasons we chose to feature this abstract is because we’re interested in science at the convergence. We really thought this was a potential way to move the field forward for rheumatologists.”
Gardner said it’s an advantage that the patient could potentially have an ARTHUR scan with a DIANA report and get blood tests done prior to a visit with the rheumatologist. “It’s really time-consuming for a human to do these studies, so if you automate it, that’s a step forward in terms of having the data available for the rheumatologist to view and use sequentially to follow how patients are doing.”
When introducing Just’s presentation, Gardner called it “the coolest abstract of the meeting.”
Both DIANA and ARTHUR Performed At Least as Well as Human Rheumatologists
In the study, 30 patients with RA underwent two scans by ARTHUR, followed by a scan from a rheumatologist specialist in musculoskeletal ultrasound. The scans were sent to DIANA, who graded the images according to the Global OMERACT-EULAR Synovitis Score, as did the human rheumatologist.
A “ground truth” was established by another human expert who evaluated both ARTHUR’s and the other rheumatologist’s images, blinded to the scanning method. The image with the highest disease activity was deemed “ground truth,” and agreement with that was assessed for the two individual methods.
Just showed a video of a patient being scanned by ARTHUR. The machine verbally guided her through removing her jewelry, applying the gel, and placing her hand on the screen under the scanner. ARTHUR’s arm moved around on the patient’s hand, locating the best angles to take grayscale images and Doppler images and Doppler video. The scan takes 15-20 minutes, and the images are stored, Just said.
The study patients had a mean age of 65 years, and 23 of the 30 were men. Their average disease duration was 11 years, and mean Disease Activity Score in 28 joints using C-reactive protein was 3.86, indicating moderate disease. A majority (73%) of patients were taking disease-modifying antirheumatic drugs, and about one third were taking biologics. ARTHUR scanned a total of 660 joints, and 564 scans were successful.
For repeatability between the two ARTHUR scans, percent exact agreement was 63% for synovial hypertrophy, 75% for Doppler activity, and 60% combined. Percent close (within a point) agreements were 93%, 94%, and 92%, respectively. Binary agreements as to whether the joint was healthy vs diseased were 88%, 91%, and 85%, respectively.
At the joint level, ARTHUR and DIANA’s percent exact agreement with ground truth was 49% for synovial hypertrophy, 63% for Doppler activity, and 48% combined. Binary agreements with disease vs healthy were 80%, 88%, and 78%, respectively.
The human rheumatologists scored very similarly. Percent exact agreement with ground truth was 51% for synovial hypertrophy, 64% for Doppler activity, and 50% combined. Percent close agreements were 94%, 94%, and 92%, respectively. And binary agreements with diseased vs healthy were 83%, 91%, and 80%, respectively.
At the patient level (all joints combined), ARTHUR and DIANA’s binary disease assessment of healthy vs disease showed agreement with the ground truth of 87% for synovial hypertrophy, 83% for Doppler activity, and 87% combined. Here, the rheumatologists scored lower, at 53%, 67%, and 60%, respectively.
“In this study, we think the precision of ARTHUR and DIANA was comparable to that of an experienced rheumatologist, at both the joint and patient level,” Just said.
Gardner pointed out another advantage of the system. “DIANA doesn’t get fatigued. ... With human reading, the precision may change based on the time of day or stress level. ... But with DIANA, you’re going to get consistent information.”
Just said that the Arthritis Foundation in Germany recently put ARTHUR and DIANA on a bus and took it to cities that lacked a rheumatologist. Patients lined up, answered a questionnaire, had blood drawn, and received their scans. A rheumatologist on the bus then interpreted the data and consulted with the individuals about their RA risk. “In the last trip, we screened 800 patients in 6 days. So there are definitely possibilities here.”
Just is co-owner of ROPCA. Gardner had no disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — A fully automated ultrasound scanning system combined with artificial intelligence–based disease activity scoring performed as well as expert rheumatologists in hand joint assessment of patients with rheumatoid arthritis (RA), new research found.
The system, made by a Danish company called ROPCA, comprises an ultrasound scanner called ARTHUR (RA Ultrasound Robot) that interacts directly with the patient and scans 11 joints per hand and a neural network–based software system, DIANA (Diagnosis Aid Network for RA), that evaluates the images and monitors RA activity.
The combined system classifies the degree of RA according to the joint European Alliance of Associations for Rheumatology (EULAR)–Outcome Measures in Rheumatology (OMERACT) standards for RA diagnosis. It received a CE Mark in Europe in 2022 and is currently in use in six rheumatology clinics in Denmark, Germany, Switzerland, and Austria, with more to come, ROPCA Co-founder and Chief Medical Officer Søren A. Just, MD, said in an interview.
“Automated systems could help rheumatologists in the early detection and monitoring of arthritis diseases. Systems can be placed or move in areas with insufficient rheumatological expertise,” Just said during a special late-breaker session presentation at the annual meeting of the American College of Rheumatology (ACR).
He said in an interview: “Currently, there are so many people referred and few and fewer rheumatologists. So we need to think differently. We need good automated assistants.” As a screening tool, the system can determine whether a person with hand pain has RA or just osteoarthritis “and also can give the patient an immediate answer, instead of waiting sometimes up to 6 months to get the information.”
Just, who is also a senior physician in the Department of Internal Medicine at Odense University Hospital in Denmark, said that his department is also using the system to assess flares in patients with established RA. “They can have a blood sample taken. They’re scanned by the robot, and you can see if there is any disease activity. But I think that screening of patients with joint pain is the beginning.”
Asked to comment, session moderator Gregory C. Gardner, MD, Emeritus Professor in the Division of Rheumatology at the University of Washington, Seattle, and a member of the ACR conference program committee, said in an interview “one of the reasons we chose to feature this abstract is because we’re interested in science at the convergence. We really thought this was a potential way to move the field forward for rheumatologists.”
Gardner said it’s an advantage that the patient could potentially have an ARTHUR scan with a DIANA report and get blood tests done prior to a visit with the rheumatologist. “It’s really time-consuming for a human to do these studies, so if you automate it, that’s a step forward in terms of having the data available for the rheumatologist to view and use sequentially to follow how patients are doing.”
When introducing Just’s presentation, Gardner called it “the coolest abstract of the meeting.”
Both DIANA and ARTHUR Performed At Least as Well as Human Rheumatologists
In the study, 30 patients with RA underwent two scans by ARTHUR, followed by a scan from a rheumatologist specialist in musculoskeletal ultrasound. The scans were sent to DIANA, who graded the images according to the Global OMERACT-EULAR Synovitis Score, as did the human rheumatologist.
A “ground truth” was established by another human expert who evaluated both ARTHUR’s and the other rheumatologist’s images, blinded to the scanning method. The image with the highest disease activity was deemed “ground truth,” and agreement with that was assessed for the two individual methods.
Just showed a video of a patient being scanned by ARTHUR. The machine verbally guided her through removing her jewelry, applying the gel, and placing her hand on the screen under the scanner. ARTHUR’s arm moved around on the patient’s hand, locating the best angles to take grayscale images and Doppler images and Doppler video. The scan takes 15-20 minutes, and the images are stored, Just said.
The study patients had a mean age of 65 years, and 23 of the 30 were men. Their average disease duration was 11 years, and mean Disease Activity Score in 28 joints using C-reactive protein was 3.86, indicating moderate disease. A majority (73%) of patients were taking disease-modifying antirheumatic drugs, and about one third were taking biologics. ARTHUR scanned a total of 660 joints, and 564 scans were successful.
For repeatability between the two ARTHUR scans, percent exact agreement was 63% for synovial hypertrophy, 75% for Doppler activity, and 60% combined. Percent close (within a point) agreements were 93%, 94%, and 92%, respectively. Binary agreements as to whether the joint was healthy vs diseased were 88%, 91%, and 85%, respectively.
At the joint level, ARTHUR and DIANA’s percent exact agreement with ground truth was 49% for synovial hypertrophy, 63% for Doppler activity, and 48% combined. Binary agreements with disease vs healthy were 80%, 88%, and 78%, respectively.
The human rheumatologists scored very similarly. Percent exact agreement with ground truth was 51% for synovial hypertrophy, 64% for Doppler activity, and 50% combined. Percent close agreements were 94%, 94%, and 92%, respectively. And binary agreements with diseased vs healthy were 83%, 91%, and 80%, respectively.
At the patient level (all joints combined), ARTHUR and DIANA’s binary disease assessment of healthy vs disease showed agreement with the ground truth of 87% for synovial hypertrophy, 83% for Doppler activity, and 87% combined. Here, the rheumatologists scored lower, at 53%, 67%, and 60%, respectively.
“In this study, we think the precision of ARTHUR and DIANA was comparable to that of an experienced rheumatologist, at both the joint and patient level,” Just said.
Gardner pointed out another advantage of the system. “DIANA doesn’t get fatigued. ... With human reading, the precision may change based on the time of day or stress level. ... But with DIANA, you’re going to get consistent information.”
Just said that the Arthritis Foundation in Germany recently put ARTHUR and DIANA on a bus and took it to cities that lacked a rheumatologist. Patients lined up, answered a questionnaire, had blood drawn, and received their scans. A rheumatologist on the bus then interpreted the data and consulted with the individuals about their RA risk. “In the last trip, we screened 800 patients in 6 days. So there are definitely possibilities here.”
Just is co-owner of ROPCA. Gardner had no disclosures.
A version of this article appeared on Medscape.com.
FROM ACR 2024
Cancer Mortality Not Higher for Patients With Autoimmune Disease on Checkpoint Inhibitors
WASHINGTON — Immune checkpoint inhibitor (ICI) therapy does not increase mortality in people with preexisting autoimmune diseases, new research has found.
Results from a large database analysis of patients with and without autoimmune diseases suggest it is safe to treat them with ICI if they develop a cancer for which it is indicated, Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said at the American College of Rheumatology 2024 Annual Meeting.
“One message is that, when rheumatologists are asked by oncologists about patients with rheumatoid arthritis or vasculitis or other autoimmune diseases and whether it’s safe to treat them with immune checkpoint inhibitors, this result provides some evidence that it probably is safe…. Checkpoint inhibitors are really incredible drugs, and they’ve improved mortality for a lot of cancers, particularly melanoma, and so I think there should be a pretty high threshold for us to say a patient shouldn’t receive them because of an autoimmune condition,” he told this news organization.
Another implication, Challener said, is that people with autoimmune diseases shouldn’t routinely be excluded from clinical trials of ICIs. Currently they are excluded because of concerns about exacerbation of underlying autoimmunity, possible interference between the ICI and the immunosuppressive drugs used to treat the autoimmune condition, and a theoretical risk for serious adverse events.
“Clinical trials are continuing to exclude these patients, and they paint with a very broad brush anyone with underlying autoimmunity ... I’m hoping that that changes. I don’t think there’s a great evidence base to support that practice, and it’s unfortunate that patients with underlying autoimmune diseases are excluded from important studies,” Challener said.
Asked to comment, session moderator Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, told this news organization that he agrees the data are generally reassuring. “If one of our patients gets cancer and their oncologist wants to use a checkpoint inhibitor, we’d obviously still monitor them for complications, but we wouldn’t automatically assume the combination of a checkpoint inhibitor and autoimmune disease would increase their mortality.”
No Difference in Mortality for Those With and Without Autoimmune Disease
Challener and colleagues used administrative health data from the TriNetX Diamond network of 92 US healthcare sites with 212 million patients. All patients included in the study were receiving anti-programmed death protein 1/programmed death ligand 1 to treat malignancies involving the skin, lung/bronchus, digestive organs, or urinary tract. The study population also had at least one rheumatologic, gastrointestinal, neurologic, dermatologic, or endocrine autoimmune disease.
Propensity score matching between those with and without autoimmune disease was performed for about 100 covariates. Prior to the matching, the autoimmune disease group had significantly higher rates of cardiovascular and other comorbidities. The matching yielded 23,714 individuals with autoimmune disease and the same number without who had similar demographics and comorbidity rates, as well as malignancy type, alcohol/tobacco use, and medication use.
At a median follow-up of 250 days, the risk for mortality prior to propensity matching was 40.0% in the autoimmune disease group and 38.1% for those without, a significant difference with hazard ratio 1.07 (95% CI, 1.05-1.10). But after the matching, the difference was no longer significant: 39.8% vs 40.2%, respectively (0.97, 0.94-1.00).
The Kaplan-Meier curves for survival probability for those with or without autoimmune disease were nearly superimposed, showing no difference up to 1600 days. An analysis of just the patients with rheumatic diseases yielded similar results, Challener said.
Some Caveats About the Data
Jeffries, who is also an associate professor of medicine at the University of Oklahoma Health Sciences Center, Oklahoma City, and the Oklahoma VA, said he would like to see additional data on outcomes, both for the autoimmune conditions and the cancers. Challener said there are plans to look at other hard endpoints such as myocardial infarction and end-stage renal disease, but that the database is limited.
Both Challener and Jeffries also cautioned that the reassurance may not apply to patients with active disease.
“One thing this research doesn’t address is whether active autoimmune disease might have a different outcome compared to more kind of quiet disease…. If you have a patient who has extremely active rheumatoid arthritis or extremely active giant cell arthritis, for instance, I think that could be more challenging. I would be frightened to put a patient with really active GCA on pembrolizumab or say that it’s safe without their disease being controlled. But for someone who has well-controlled disease or minimally active disease, this is very reassuring,” Challener told this news organization.
“I think this may also be important in that it’s a good argument to tell the drug companies to include autoimmune patients in these trials so we can get better data,” Jeffries said.
Challener and Jeffries had no relevant disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — Immune checkpoint inhibitor (ICI) therapy does not increase mortality in people with preexisting autoimmune diseases, new research has found.
Results from a large database analysis of patients with and without autoimmune diseases suggest it is safe to treat them with ICI if they develop a cancer for which it is indicated, Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said at the American College of Rheumatology 2024 Annual Meeting.
“One message is that, when rheumatologists are asked by oncologists about patients with rheumatoid arthritis or vasculitis or other autoimmune diseases and whether it’s safe to treat them with immune checkpoint inhibitors, this result provides some evidence that it probably is safe…. Checkpoint inhibitors are really incredible drugs, and they’ve improved mortality for a lot of cancers, particularly melanoma, and so I think there should be a pretty high threshold for us to say a patient shouldn’t receive them because of an autoimmune condition,” he told this news organization.
Another implication, Challener said, is that people with autoimmune diseases shouldn’t routinely be excluded from clinical trials of ICIs. Currently they are excluded because of concerns about exacerbation of underlying autoimmunity, possible interference between the ICI and the immunosuppressive drugs used to treat the autoimmune condition, and a theoretical risk for serious adverse events.
“Clinical trials are continuing to exclude these patients, and they paint with a very broad brush anyone with underlying autoimmunity ... I’m hoping that that changes. I don’t think there’s a great evidence base to support that practice, and it’s unfortunate that patients with underlying autoimmune diseases are excluded from important studies,” Challener said.
Asked to comment, session moderator Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, told this news organization that he agrees the data are generally reassuring. “If one of our patients gets cancer and their oncologist wants to use a checkpoint inhibitor, we’d obviously still monitor them for complications, but we wouldn’t automatically assume the combination of a checkpoint inhibitor and autoimmune disease would increase their mortality.”
No Difference in Mortality for Those With and Without Autoimmune Disease
Challener and colleagues used administrative health data from the TriNetX Diamond network of 92 US healthcare sites with 212 million patients. All patients included in the study were receiving anti-programmed death protein 1/programmed death ligand 1 to treat malignancies involving the skin, lung/bronchus, digestive organs, or urinary tract. The study population also had at least one rheumatologic, gastrointestinal, neurologic, dermatologic, or endocrine autoimmune disease.
Propensity score matching between those with and without autoimmune disease was performed for about 100 covariates. Prior to the matching, the autoimmune disease group had significantly higher rates of cardiovascular and other comorbidities. The matching yielded 23,714 individuals with autoimmune disease and the same number without who had similar demographics and comorbidity rates, as well as malignancy type, alcohol/tobacco use, and medication use.
At a median follow-up of 250 days, the risk for mortality prior to propensity matching was 40.0% in the autoimmune disease group and 38.1% for those without, a significant difference with hazard ratio 1.07 (95% CI, 1.05-1.10). But after the matching, the difference was no longer significant: 39.8% vs 40.2%, respectively (0.97, 0.94-1.00).
The Kaplan-Meier curves for survival probability for those with or without autoimmune disease were nearly superimposed, showing no difference up to 1600 days. An analysis of just the patients with rheumatic diseases yielded similar results, Challener said.
Some Caveats About the Data
Jeffries, who is also an associate professor of medicine at the University of Oklahoma Health Sciences Center, Oklahoma City, and the Oklahoma VA, said he would like to see additional data on outcomes, both for the autoimmune conditions and the cancers. Challener said there are plans to look at other hard endpoints such as myocardial infarction and end-stage renal disease, but that the database is limited.
Both Challener and Jeffries also cautioned that the reassurance may not apply to patients with active disease.
“One thing this research doesn’t address is whether active autoimmune disease might have a different outcome compared to more kind of quiet disease…. If you have a patient who has extremely active rheumatoid arthritis or extremely active giant cell arthritis, for instance, I think that could be more challenging. I would be frightened to put a patient with really active GCA on pembrolizumab or say that it’s safe without their disease being controlled. But for someone who has well-controlled disease or minimally active disease, this is very reassuring,” Challener told this news organization.
“I think this may also be important in that it’s a good argument to tell the drug companies to include autoimmune patients in these trials so we can get better data,” Jeffries said.
Challener and Jeffries had no relevant disclosures.
A version of this article appeared on Medscape.com.
WASHINGTON — Immune checkpoint inhibitor (ICI) therapy does not increase mortality in people with preexisting autoimmune diseases, new research has found.
Results from a large database analysis of patients with and without autoimmune diseases suggest it is safe to treat them with ICI if they develop a cancer for which it is indicated, Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said at the American College of Rheumatology 2024 Annual Meeting.
“One message is that, when rheumatologists are asked by oncologists about patients with rheumatoid arthritis or vasculitis or other autoimmune diseases and whether it’s safe to treat them with immune checkpoint inhibitors, this result provides some evidence that it probably is safe…. Checkpoint inhibitors are really incredible drugs, and they’ve improved mortality for a lot of cancers, particularly melanoma, and so I think there should be a pretty high threshold for us to say a patient shouldn’t receive them because of an autoimmune condition,” he told this news organization.
Another implication, Challener said, is that people with autoimmune diseases shouldn’t routinely be excluded from clinical trials of ICIs. Currently they are excluded because of concerns about exacerbation of underlying autoimmunity, possible interference between the ICI and the immunosuppressive drugs used to treat the autoimmune condition, and a theoretical risk for serious adverse events.
“Clinical trials are continuing to exclude these patients, and they paint with a very broad brush anyone with underlying autoimmunity ... I’m hoping that that changes. I don’t think there’s a great evidence base to support that practice, and it’s unfortunate that patients with underlying autoimmune diseases are excluded from important studies,” Challener said.
Asked to comment, session moderator Matlock Jeffries, MD, director of the Arthritis Research Unit at the Oklahoma Medical Research Foundation, Oklahoma City, told this news organization that he agrees the data are generally reassuring. “If one of our patients gets cancer and their oncologist wants to use a checkpoint inhibitor, we’d obviously still monitor them for complications, but we wouldn’t automatically assume the combination of a checkpoint inhibitor and autoimmune disease would increase their mortality.”
No Difference in Mortality for Those With and Without Autoimmune Disease
Challener and colleagues used administrative health data from the TriNetX Diamond network of 92 US healthcare sites with 212 million patients. All patients included in the study were receiving anti-programmed death protein 1/programmed death ligand 1 to treat malignancies involving the skin, lung/bronchus, digestive organs, or urinary tract. The study population also had at least one rheumatologic, gastrointestinal, neurologic, dermatologic, or endocrine autoimmune disease.
Propensity score matching between those with and without autoimmune disease was performed for about 100 covariates. Prior to the matching, the autoimmune disease group had significantly higher rates of cardiovascular and other comorbidities. The matching yielded 23,714 individuals with autoimmune disease and the same number without who had similar demographics and comorbidity rates, as well as malignancy type, alcohol/tobacco use, and medication use.
At a median follow-up of 250 days, the risk for mortality prior to propensity matching was 40.0% in the autoimmune disease group and 38.1% for those without, a significant difference with hazard ratio 1.07 (95% CI, 1.05-1.10). But after the matching, the difference was no longer significant: 39.8% vs 40.2%, respectively (0.97, 0.94-1.00).
The Kaplan-Meier curves for survival probability for those with or without autoimmune disease were nearly superimposed, showing no difference up to 1600 days. An analysis of just the patients with rheumatic diseases yielded similar results, Challener said.
Some Caveats About the Data
Jeffries, who is also an associate professor of medicine at the University of Oklahoma Health Sciences Center, Oklahoma City, and the Oklahoma VA, said he would like to see additional data on outcomes, both for the autoimmune conditions and the cancers. Challener said there are plans to look at other hard endpoints such as myocardial infarction and end-stage renal disease, but that the database is limited.
Both Challener and Jeffries also cautioned that the reassurance may not apply to patients with active disease.
“One thing this research doesn’t address is whether active autoimmune disease might have a different outcome compared to more kind of quiet disease…. If you have a patient who has extremely active rheumatoid arthritis or extremely active giant cell arthritis, for instance, I think that could be more challenging. I would be frightened to put a patient with really active GCA on pembrolizumab or say that it’s safe without their disease being controlled. But for someone who has well-controlled disease or minimally active disease, this is very reassuring,” Challener told this news organization.
“I think this may also be important in that it’s a good argument to tell the drug companies to include autoimmune patients in these trials so we can get better data,” Jeffries said.
Challener and Jeffries had no relevant disclosures.
A version of this article appeared on Medscape.com.
FROM ACR 2024
Oral Semaglutide 25-mg Dose May Provide Most Benefit
SAN ANTONIO — Oral semaglutide 25 mg appears to be just as effective in promoting weight loss and other beneficial outcomes as are the investigational 50-mg oral dose and the injectable 2.4-mg dose (Wegovy), in new research.
Data from Novo Nordisk’s OASIS 4 trial suggest that “oral semaglutide 25 mg may represent an efficacious option for the treatment of overweight and obesity, particularly in patients who prefer oral administration,” W. Timothy Garvey, MD, professor in the Department of Nutrition Sciences at the University of Alabama at Birmingham (UAB), said at the Obesity Society’s Obesity Week 2024 meeting.
In an interview, Garvey, who is also senior scientist at the UAB Nutrition Obesity Research Center, added: “There’s a principle in medicine that you always use the lowest dose that has highest efficacy, and for oral semaglutide for obesity, that appears to be the 25-mg dose. We need oral medicines to offer as an option for patients that could lead to a longer persistence in adherence to obesity medications, which is a big problem. Less than half the people maintain their adherence after a year.”
Asked to comment, session moderator and obesity researcher Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said that OASIS 4 was “extremely well done, especially given that we’re all concerned about the high prevalence of people stopping these medications at a year. ... I love the idea of trying to find these lower doses and increasing options for people.”
(The oral semaglutide approved for treating type 2 diabetes [Rybelsus] is sold in 7-mg and 14-mg doses.)
With Oral Semaglutide, Lower May Be Better
OASIS 4 was a double-blind, randomized, placebo-controlled 64-week multicenter trial involving a total of 307 participants with overweight/obesity randomized 2:1 to oral semaglutide 25 mg or placebo. Of those, 167 in the semaglutide 25 mg and 76 in the placebo groups completed the trial.
For the co–primary endpoint change in body weight at week 64 (including 52-week maintenance and 7-week follow-up periods), there was a drop of 13.6% with oral semaglutide vs just 2.2% with placebo (P < .0001), based on in-trial observation regardless of adherence. For the analysis including just the on-treatment period, those reductions were 16.6% and 2.7%, respectively (P < .0001).
For the other co–primary endpoint, 79.2% semaglutide vs 31.1% placebo lost at least 5% of their body weight, 63.0% vs 14.4% lost ≥ 10%, 50.0% vs 5.6% lost ≥ 15%, and 29.7% vs 3.3% lost ≥ 20%. All were statistically significant differences, at P < .0001.
On the Impact of Weight on Quality of Life-Lite Clinical Trials Version, change from baseline scores were 16.2 with oral semaglutide vs 8.4 for placebo (P = .0006) and the proportion of participants achieving clinically meaningful increases in those scores (≥ 14.6 points) were 55.3% vs 34.8% (P = .0022).
Waist circumference was also significantly reduced from baseline with oral semaglutide, by 12.2 cm vs 2.8 cm (P < .0001). Both systolic and diastolic blood pressure dropped more with semaglutide than placebo, but not significantly. However, the inflammatory marker C-reactive protein dropped from baseline by 46.4% vs just 4.2% with placebo, a significant difference (P < .0001).
Hemoglobin A1c dropped by 0.29 percentage points vs just 0.06 with placebo (P = .0012) and fasting plasma glucose by 6.6 mg/dL, while rising by 0.4 mg/dL in the placebo group (P = .0012). Lipid levels also improved more with oral semaglutide.
Overall adverse events occurred in 93.1% on oral semaglutide and 85.3% with placebo, and gastrointestinal adverse events in 74% and 42.2%, respectively. Other types of adverse events didn’t differ between the groups. The proportions experiencing severe adverse events were less in the semaglutide group (3.9% vs 8.8%), although adverse events leading to permanent treatment discontinuation were slightly higher with oral semaglutide (6.9% vs 5.9%). There were no deaths.
The estimated treatment difference from placebo in body weight change of −11.4% in OASIS 4 (P < .0001) was similar to the –12.7% (P < .0001) seen with the 50-mg oral semaglutide dose studied in OASIS 1 and the –12.4% (P < .0001) difference with subcutaneous semaglutide 2.4 mg in the STEP 1 trial. “All had pretty comparable efficacy,” Garvey noted.
The side-effect profiles, including frequency of gastrointestinal side effects, were also similar across the three trials. However, Garvey added, “the mean duration of nausea in those patients that experienced nausea was shorter in patients on a 25-mg dose, 13 days, whereas the mean duration of nausea was greater, 19 days, in those in OASIS 1 taking 50 mg a day.”
Garvey has consulted for Boehringer Ingelheim, Carmot Therapeutics/Roche, Eli Lilly, Fractyl Laboratories, Inogen, Lilly, Merck, Novo Nordisk, and Zealand Pharma; has ownership interest (stock, stock options in a publicly owned company) for Bristol-Myers Squibb, Isis, Lilly, and Novartis; serves as site principal investigator for Carmot Therapeutics/Roche, Eli Lilly, Epitomee Medical, Lilly, Neurovalens, Novo Nordisk, and Zealand Pharmaceuticals; and as a data monitoring committee member for Boehringer Ingelheim and Eli Lilly. Skelton is editor in chief of the journal Childhood Obesity.
A version of this article first appeared on Medscape.com.
SAN ANTONIO — Oral semaglutide 25 mg appears to be just as effective in promoting weight loss and other beneficial outcomes as are the investigational 50-mg oral dose and the injectable 2.4-mg dose (Wegovy), in new research.
Data from Novo Nordisk’s OASIS 4 trial suggest that “oral semaglutide 25 mg may represent an efficacious option for the treatment of overweight and obesity, particularly in patients who prefer oral administration,” W. Timothy Garvey, MD, professor in the Department of Nutrition Sciences at the University of Alabama at Birmingham (UAB), said at the Obesity Society’s Obesity Week 2024 meeting.
In an interview, Garvey, who is also senior scientist at the UAB Nutrition Obesity Research Center, added: “There’s a principle in medicine that you always use the lowest dose that has highest efficacy, and for oral semaglutide for obesity, that appears to be the 25-mg dose. We need oral medicines to offer as an option for patients that could lead to a longer persistence in adherence to obesity medications, which is a big problem. Less than half the people maintain their adherence after a year.”
Asked to comment, session moderator and obesity researcher Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said that OASIS 4 was “extremely well done, especially given that we’re all concerned about the high prevalence of people stopping these medications at a year. ... I love the idea of trying to find these lower doses and increasing options for people.”
(The oral semaglutide approved for treating type 2 diabetes [Rybelsus] is sold in 7-mg and 14-mg doses.)
With Oral Semaglutide, Lower May Be Better
OASIS 4 was a double-blind, randomized, placebo-controlled 64-week multicenter trial involving a total of 307 participants with overweight/obesity randomized 2:1 to oral semaglutide 25 mg or placebo. Of those, 167 in the semaglutide 25 mg and 76 in the placebo groups completed the trial.
For the co–primary endpoint change in body weight at week 64 (including 52-week maintenance and 7-week follow-up periods), there was a drop of 13.6% with oral semaglutide vs just 2.2% with placebo (P < .0001), based on in-trial observation regardless of adherence. For the analysis including just the on-treatment period, those reductions were 16.6% and 2.7%, respectively (P < .0001).
For the other co–primary endpoint, 79.2% semaglutide vs 31.1% placebo lost at least 5% of their body weight, 63.0% vs 14.4% lost ≥ 10%, 50.0% vs 5.6% lost ≥ 15%, and 29.7% vs 3.3% lost ≥ 20%. All were statistically significant differences, at P < .0001.
On the Impact of Weight on Quality of Life-Lite Clinical Trials Version, change from baseline scores were 16.2 with oral semaglutide vs 8.4 for placebo (P = .0006) and the proportion of participants achieving clinically meaningful increases in those scores (≥ 14.6 points) were 55.3% vs 34.8% (P = .0022).
Waist circumference was also significantly reduced from baseline with oral semaglutide, by 12.2 cm vs 2.8 cm (P < .0001). Both systolic and diastolic blood pressure dropped more with semaglutide than placebo, but not significantly. However, the inflammatory marker C-reactive protein dropped from baseline by 46.4% vs just 4.2% with placebo, a significant difference (P < .0001).
Hemoglobin A1c dropped by 0.29 percentage points vs just 0.06 with placebo (P = .0012) and fasting plasma glucose by 6.6 mg/dL, while rising by 0.4 mg/dL in the placebo group (P = .0012). Lipid levels also improved more with oral semaglutide.
Overall adverse events occurred in 93.1% on oral semaglutide and 85.3% with placebo, and gastrointestinal adverse events in 74% and 42.2%, respectively. Other types of adverse events didn’t differ between the groups. The proportions experiencing severe adverse events were less in the semaglutide group (3.9% vs 8.8%), although adverse events leading to permanent treatment discontinuation were slightly higher with oral semaglutide (6.9% vs 5.9%). There were no deaths.
The estimated treatment difference from placebo in body weight change of −11.4% in OASIS 4 (P < .0001) was similar to the –12.7% (P < .0001) seen with the 50-mg oral semaglutide dose studied in OASIS 1 and the –12.4% (P < .0001) difference with subcutaneous semaglutide 2.4 mg in the STEP 1 trial. “All had pretty comparable efficacy,” Garvey noted.
The side-effect profiles, including frequency of gastrointestinal side effects, were also similar across the three trials. However, Garvey added, “the mean duration of nausea in those patients that experienced nausea was shorter in patients on a 25-mg dose, 13 days, whereas the mean duration of nausea was greater, 19 days, in those in OASIS 1 taking 50 mg a day.”
Garvey has consulted for Boehringer Ingelheim, Carmot Therapeutics/Roche, Eli Lilly, Fractyl Laboratories, Inogen, Lilly, Merck, Novo Nordisk, and Zealand Pharma; has ownership interest (stock, stock options in a publicly owned company) for Bristol-Myers Squibb, Isis, Lilly, and Novartis; serves as site principal investigator for Carmot Therapeutics/Roche, Eli Lilly, Epitomee Medical, Lilly, Neurovalens, Novo Nordisk, and Zealand Pharmaceuticals; and as a data monitoring committee member for Boehringer Ingelheim and Eli Lilly. Skelton is editor in chief of the journal Childhood Obesity.
A version of this article first appeared on Medscape.com.
SAN ANTONIO — Oral semaglutide 25 mg appears to be just as effective in promoting weight loss and other beneficial outcomes as are the investigational 50-mg oral dose and the injectable 2.4-mg dose (Wegovy), in new research.
Data from Novo Nordisk’s OASIS 4 trial suggest that “oral semaglutide 25 mg may represent an efficacious option for the treatment of overweight and obesity, particularly in patients who prefer oral administration,” W. Timothy Garvey, MD, professor in the Department of Nutrition Sciences at the University of Alabama at Birmingham (UAB), said at the Obesity Society’s Obesity Week 2024 meeting.
In an interview, Garvey, who is also senior scientist at the UAB Nutrition Obesity Research Center, added: “There’s a principle in medicine that you always use the lowest dose that has highest efficacy, and for oral semaglutide for obesity, that appears to be the 25-mg dose. We need oral medicines to offer as an option for patients that could lead to a longer persistence in adherence to obesity medications, which is a big problem. Less than half the people maintain their adherence after a year.”
Asked to comment, session moderator and obesity researcher Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said that OASIS 4 was “extremely well done, especially given that we’re all concerned about the high prevalence of people stopping these medications at a year. ... I love the idea of trying to find these lower doses and increasing options for people.”
(The oral semaglutide approved for treating type 2 diabetes [Rybelsus] is sold in 7-mg and 14-mg doses.)
With Oral Semaglutide, Lower May Be Better
OASIS 4 was a double-blind, randomized, placebo-controlled 64-week multicenter trial involving a total of 307 participants with overweight/obesity randomized 2:1 to oral semaglutide 25 mg or placebo. Of those, 167 in the semaglutide 25 mg and 76 in the placebo groups completed the trial.
For the co–primary endpoint change in body weight at week 64 (including 52-week maintenance and 7-week follow-up periods), there was a drop of 13.6% with oral semaglutide vs just 2.2% with placebo (P < .0001), based on in-trial observation regardless of adherence. For the analysis including just the on-treatment period, those reductions were 16.6% and 2.7%, respectively (P < .0001).
For the other co–primary endpoint, 79.2% semaglutide vs 31.1% placebo lost at least 5% of their body weight, 63.0% vs 14.4% lost ≥ 10%, 50.0% vs 5.6% lost ≥ 15%, and 29.7% vs 3.3% lost ≥ 20%. All were statistically significant differences, at P < .0001.
On the Impact of Weight on Quality of Life-Lite Clinical Trials Version, change from baseline scores were 16.2 with oral semaglutide vs 8.4 for placebo (P = .0006) and the proportion of participants achieving clinically meaningful increases in those scores (≥ 14.6 points) were 55.3% vs 34.8% (P = .0022).
Waist circumference was also significantly reduced from baseline with oral semaglutide, by 12.2 cm vs 2.8 cm (P < .0001). Both systolic and diastolic blood pressure dropped more with semaglutide than placebo, but not significantly. However, the inflammatory marker C-reactive protein dropped from baseline by 46.4% vs just 4.2% with placebo, a significant difference (P < .0001).
Hemoglobin A1c dropped by 0.29 percentage points vs just 0.06 with placebo (P = .0012) and fasting plasma glucose by 6.6 mg/dL, while rising by 0.4 mg/dL in the placebo group (P = .0012). Lipid levels also improved more with oral semaglutide.
Overall adverse events occurred in 93.1% on oral semaglutide and 85.3% with placebo, and gastrointestinal adverse events in 74% and 42.2%, respectively. Other types of adverse events didn’t differ between the groups. The proportions experiencing severe adverse events were less in the semaglutide group (3.9% vs 8.8%), although adverse events leading to permanent treatment discontinuation were slightly higher with oral semaglutide (6.9% vs 5.9%). There were no deaths.
The estimated treatment difference from placebo in body weight change of −11.4% in OASIS 4 (P < .0001) was similar to the –12.7% (P < .0001) seen with the 50-mg oral semaglutide dose studied in OASIS 1 and the –12.4% (P < .0001) difference with subcutaneous semaglutide 2.4 mg in the STEP 1 trial. “All had pretty comparable efficacy,” Garvey noted.
The side-effect profiles, including frequency of gastrointestinal side effects, were also similar across the three trials. However, Garvey added, “the mean duration of nausea in those patients that experienced nausea was shorter in patients on a 25-mg dose, 13 days, whereas the mean duration of nausea was greater, 19 days, in those in OASIS 1 taking 50 mg a day.”
Garvey has consulted for Boehringer Ingelheim, Carmot Therapeutics/Roche, Eli Lilly, Fractyl Laboratories, Inogen, Lilly, Merck, Novo Nordisk, and Zealand Pharma; has ownership interest (stock, stock options in a publicly owned company) for Bristol-Myers Squibb, Isis, Lilly, and Novartis; serves as site principal investigator for Carmot Therapeutics/Roche, Eli Lilly, Epitomee Medical, Lilly, Neurovalens, Novo Nordisk, and Zealand Pharmaceuticals; and as a data monitoring committee member for Boehringer Ingelheim and Eli Lilly. Skelton is editor in chief of the journal Childhood Obesity.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2024
Holding RA, SpA Drugs Did Not Improve Antibody Response to COVID Vaccine
WASHINGTON — There is no benefit to interrupting treatment with many of the available targeted synthetic or biologic disease-modifying antirheumatic drugs for rheumatoid arthritis (RA) or spondyloarthritis (SpA) at the time of a repeat COVID-19 vaccine dose, new research found.
In the multicenter, randomized controlled COVID Vaccine Response (COVER) trial of 577 patients with RA or SpA taking either abatacept, Janus kinase (JAK) inhibitors, interleukin (IL)–17 inhibitors, or tumor necrosis factor (TNF) inhibitors, holding those drugs for 2 weeks at the time of COVID-19 vaccination supplemental doses didn’t improve antibody response to the vaccine but did lead to disease flares. Most participants had significant antibody responses to the vaccine, regardless of whether their medication had been held or continued, Jeffrey R. Curtis, MD, the Harbert-Ball Professor of Medicine, Epidemiology, and Computer Science at the University of Alabama at Birmingham, reported at the annual meeting of the American College of Rheumatology (ACR).
Guidelines issued by ACR in 2023 recommended holding abatacept for the COVID vaccine but said that “the task force failed to reach consensus” on whether or not to temporarily interrupt the other medications following primary vaccination or supplemental/booster dosing.
Curtis, who was an author on those guidelines, said in an interview, “to date, we haven’t known whether it might be a good idea to hold certain drugs at the time patients receive their next dose of the COVID vaccine. ... That’s because without direct evidence, you have people trading opinions based on extrapolated data.”
The inability to measure cell-mediated immunity and only humoral (ie, antibody-based) immunity is a limitation in COVER. “Nevertheless, based on what we know now, it isn’t advisable to hold any of the four drug classes that we studied at the time patients receive their next COVID vaccine dose. This finding is in contrast to data from a different trial showing that holding methotrexate for 2 weeks does appear to help in response to COVID-19 vaccination, as well as influenza vaccine,” Curtis said.
Asked to comment, session moderator Elena Myasoedova, MD, PhD, consultant rheumatologist and director of the Inflammatory Arthritis Clinic at the Mayo Clinic, Rochester, Minnesota, said in an interview: “This has been an area of clinical uncertainty. It raises a lot of questions from patients and from physicians alike as to whether or not to hold the medication because the implications are flares, and that’s impactful for patients. Patients care about their RA status and how it is controlled, and if there is no difference, then there is no reason to change the medication regimen.”
To Hold or Not to Hold: COVER Shows It Makes Little Difference to Vaccine Response
In COVER, 128 patients were taking abatacept, 96 IL-17 inhibitors, 237 JAK inhibitors, and 116 TNF inhibitors. The study was conducted within 30 sites of the Excellence Network in Rheumatology, a rheumatology practice–based research network launched in 2021. Participants were identified and enrolled at clinic visits immediately prior to receiving their COVID-19 boosters (in routine settings).
All had previously received two or more doses of the mRNA vaccines made by Pfizer or Moderna. Blood was drawn, and they were randomized 1:1 to either continue or hold their disease medication for 2 weeks following the booster. Blood was collected again at 6 weeks post vaccine.
Anti–receptor-binding domain (RBD) IgG antibody titers increased significantly in all drug categories across both study arms, with no differences between the hold vs continue medication groups, even after adjustments for age, sex, body mass index, methotrexate use, steroid use, and time from booster to measurement. All groups also showed increases in geometric mean fold rise of more than 3%.
Subgroup analyses showed no major differences between antibody responses in the hold vs continue groups. The anti-RBD IgG response was lower for abatacept and JAK inhibitors than for the other two drugs, but there was still no significant benefit to holding them for 2 weeks post vaccination.
Holding Drugs Leads to Disease Flares
On the flip side, there were significant differences between the two groups in their responses to the question: “Did you experience any flare or worsening of your autoimmune disease following your recent COVID-19 booster dose?” Overall, 27% of the hold group responded that they had, compared with just 13% of the continue group (P < .05). This difference was greatest in the JAK inhibitor group (33% vs 9%; P < .05).
Among those reporting flares or worsening disease, both the severity and the duration of the flares were about the same. “Interestingly, the duration is beyond a week for the majority of patients. The reason that’s important is, any symptoms that are so-called flare might simply be reactogenicity symptoms, and that might be confused for flare or disease worsening, but you see that a majority of patients actually have those symptoms extending beyond the week. Most of them are worsening in arthritis, as you might expect,” Curtis said in his presentation.
Asked what they did about the flare, only a minority of patients reported contacting a healthcare provider. In all, 68% of the hold group and 78% of the continue group took no action. That’s good in the sense that most of the flares weren’t severe, but it has implications for research, Curtis pointed out.
“A lot of times in the vaccine literature, people do retrospective chart review by looking to see what the doctor said as to whether the patient had a flare. And what this would tell you is patients may be reporting a lot of flares that their doctor doesn’t know anything about. So if you really want to know whether people are having a flare, even a mild flare, you really have to collect prospective data.”
COVID is Not the Last Pandemic
“These results are reassuring, although I think we need a bit more data on abatacept,” Myasoedova said, adding, “I was also interested in the outcomes, such as severe infections, that actually happened to these patients. What we see in the labs in their immune response is one thing, but then also important is what actually evolves in terms of the outcomes, especially with abatacept.”
Overall, she said, “I think it’s reassuring and definitely informs clinical practice going forward. But then probably we’ll learn more. What we’re hearing is COVID is not the last pandemic.”
The COVER trial receives support from AbbVie, BMS, Eli Lilly, Novartis, and Pfizer. Curtis has received research grants and consulting fees from AbbVie, Amgen, BMS, GSK, Eli Lilly, Novartis, Pfizer, Sanofi, and UCB. Myasoedova has no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — There is no benefit to interrupting treatment with many of the available targeted synthetic or biologic disease-modifying antirheumatic drugs for rheumatoid arthritis (RA) or spondyloarthritis (SpA) at the time of a repeat COVID-19 vaccine dose, new research found.
In the multicenter, randomized controlled COVID Vaccine Response (COVER) trial of 577 patients with RA or SpA taking either abatacept, Janus kinase (JAK) inhibitors, interleukin (IL)–17 inhibitors, or tumor necrosis factor (TNF) inhibitors, holding those drugs for 2 weeks at the time of COVID-19 vaccination supplemental doses didn’t improve antibody response to the vaccine but did lead to disease flares. Most participants had significant antibody responses to the vaccine, regardless of whether their medication had been held or continued, Jeffrey R. Curtis, MD, the Harbert-Ball Professor of Medicine, Epidemiology, and Computer Science at the University of Alabama at Birmingham, reported at the annual meeting of the American College of Rheumatology (ACR).
Guidelines issued by ACR in 2023 recommended holding abatacept for the COVID vaccine but said that “the task force failed to reach consensus” on whether or not to temporarily interrupt the other medications following primary vaccination or supplemental/booster dosing.
Curtis, who was an author on those guidelines, said in an interview, “to date, we haven’t known whether it might be a good idea to hold certain drugs at the time patients receive their next dose of the COVID vaccine. ... That’s because without direct evidence, you have people trading opinions based on extrapolated data.”
The inability to measure cell-mediated immunity and only humoral (ie, antibody-based) immunity is a limitation in COVER. “Nevertheless, based on what we know now, it isn’t advisable to hold any of the four drug classes that we studied at the time patients receive their next COVID vaccine dose. This finding is in contrast to data from a different trial showing that holding methotrexate for 2 weeks does appear to help in response to COVID-19 vaccination, as well as influenza vaccine,” Curtis said.
Asked to comment, session moderator Elena Myasoedova, MD, PhD, consultant rheumatologist and director of the Inflammatory Arthritis Clinic at the Mayo Clinic, Rochester, Minnesota, said in an interview: “This has been an area of clinical uncertainty. It raises a lot of questions from patients and from physicians alike as to whether or not to hold the medication because the implications are flares, and that’s impactful for patients. Patients care about their RA status and how it is controlled, and if there is no difference, then there is no reason to change the medication regimen.”
To Hold or Not to Hold: COVER Shows It Makes Little Difference to Vaccine Response
In COVER, 128 patients were taking abatacept, 96 IL-17 inhibitors, 237 JAK inhibitors, and 116 TNF inhibitors. The study was conducted within 30 sites of the Excellence Network in Rheumatology, a rheumatology practice–based research network launched in 2021. Participants were identified and enrolled at clinic visits immediately prior to receiving their COVID-19 boosters (in routine settings).
All had previously received two or more doses of the mRNA vaccines made by Pfizer or Moderna. Blood was drawn, and they were randomized 1:1 to either continue or hold their disease medication for 2 weeks following the booster. Blood was collected again at 6 weeks post vaccine.
Anti–receptor-binding domain (RBD) IgG antibody titers increased significantly in all drug categories across both study arms, with no differences between the hold vs continue medication groups, even after adjustments for age, sex, body mass index, methotrexate use, steroid use, and time from booster to measurement. All groups also showed increases in geometric mean fold rise of more than 3%.
Subgroup analyses showed no major differences between antibody responses in the hold vs continue groups. The anti-RBD IgG response was lower for abatacept and JAK inhibitors than for the other two drugs, but there was still no significant benefit to holding them for 2 weeks post vaccination.
Holding Drugs Leads to Disease Flares
On the flip side, there were significant differences between the two groups in their responses to the question: “Did you experience any flare or worsening of your autoimmune disease following your recent COVID-19 booster dose?” Overall, 27% of the hold group responded that they had, compared with just 13% of the continue group (P < .05). This difference was greatest in the JAK inhibitor group (33% vs 9%; P < .05).
Among those reporting flares or worsening disease, both the severity and the duration of the flares were about the same. “Interestingly, the duration is beyond a week for the majority of patients. The reason that’s important is, any symptoms that are so-called flare might simply be reactogenicity symptoms, and that might be confused for flare or disease worsening, but you see that a majority of patients actually have those symptoms extending beyond the week. Most of them are worsening in arthritis, as you might expect,” Curtis said in his presentation.
Asked what they did about the flare, only a minority of patients reported contacting a healthcare provider. In all, 68% of the hold group and 78% of the continue group took no action. That’s good in the sense that most of the flares weren’t severe, but it has implications for research, Curtis pointed out.
“A lot of times in the vaccine literature, people do retrospective chart review by looking to see what the doctor said as to whether the patient had a flare. And what this would tell you is patients may be reporting a lot of flares that their doctor doesn’t know anything about. So if you really want to know whether people are having a flare, even a mild flare, you really have to collect prospective data.”
COVID is Not the Last Pandemic
“These results are reassuring, although I think we need a bit more data on abatacept,” Myasoedova said, adding, “I was also interested in the outcomes, such as severe infections, that actually happened to these patients. What we see in the labs in their immune response is one thing, but then also important is what actually evolves in terms of the outcomes, especially with abatacept.”
Overall, she said, “I think it’s reassuring and definitely informs clinical practice going forward. But then probably we’ll learn more. What we’re hearing is COVID is not the last pandemic.”
The COVER trial receives support from AbbVie, BMS, Eli Lilly, Novartis, and Pfizer. Curtis has received research grants and consulting fees from AbbVie, Amgen, BMS, GSK, Eli Lilly, Novartis, Pfizer, Sanofi, and UCB. Myasoedova has no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — There is no benefit to interrupting treatment with many of the available targeted synthetic or biologic disease-modifying antirheumatic drugs for rheumatoid arthritis (RA) or spondyloarthritis (SpA) at the time of a repeat COVID-19 vaccine dose, new research found.
In the multicenter, randomized controlled COVID Vaccine Response (COVER) trial of 577 patients with RA or SpA taking either abatacept, Janus kinase (JAK) inhibitors, interleukin (IL)–17 inhibitors, or tumor necrosis factor (TNF) inhibitors, holding those drugs for 2 weeks at the time of COVID-19 vaccination supplemental doses didn’t improve antibody response to the vaccine but did lead to disease flares. Most participants had significant antibody responses to the vaccine, regardless of whether their medication had been held or continued, Jeffrey R. Curtis, MD, the Harbert-Ball Professor of Medicine, Epidemiology, and Computer Science at the University of Alabama at Birmingham, reported at the annual meeting of the American College of Rheumatology (ACR).
Guidelines issued by ACR in 2023 recommended holding abatacept for the COVID vaccine but said that “the task force failed to reach consensus” on whether or not to temporarily interrupt the other medications following primary vaccination or supplemental/booster dosing.
Curtis, who was an author on those guidelines, said in an interview, “to date, we haven’t known whether it might be a good idea to hold certain drugs at the time patients receive their next dose of the COVID vaccine. ... That’s because without direct evidence, you have people trading opinions based on extrapolated data.”
The inability to measure cell-mediated immunity and only humoral (ie, antibody-based) immunity is a limitation in COVER. “Nevertheless, based on what we know now, it isn’t advisable to hold any of the four drug classes that we studied at the time patients receive their next COVID vaccine dose. This finding is in contrast to data from a different trial showing that holding methotrexate for 2 weeks does appear to help in response to COVID-19 vaccination, as well as influenza vaccine,” Curtis said.
Asked to comment, session moderator Elena Myasoedova, MD, PhD, consultant rheumatologist and director of the Inflammatory Arthritis Clinic at the Mayo Clinic, Rochester, Minnesota, said in an interview: “This has been an area of clinical uncertainty. It raises a lot of questions from patients and from physicians alike as to whether or not to hold the medication because the implications are flares, and that’s impactful for patients. Patients care about their RA status and how it is controlled, and if there is no difference, then there is no reason to change the medication regimen.”
To Hold or Not to Hold: COVER Shows It Makes Little Difference to Vaccine Response
In COVER, 128 patients were taking abatacept, 96 IL-17 inhibitors, 237 JAK inhibitors, and 116 TNF inhibitors. The study was conducted within 30 sites of the Excellence Network in Rheumatology, a rheumatology practice–based research network launched in 2021. Participants were identified and enrolled at clinic visits immediately prior to receiving their COVID-19 boosters (in routine settings).
All had previously received two or more doses of the mRNA vaccines made by Pfizer or Moderna. Blood was drawn, and they were randomized 1:1 to either continue or hold their disease medication for 2 weeks following the booster. Blood was collected again at 6 weeks post vaccine.
Anti–receptor-binding domain (RBD) IgG antibody titers increased significantly in all drug categories across both study arms, with no differences between the hold vs continue medication groups, even after adjustments for age, sex, body mass index, methotrexate use, steroid use, and time from booster to measurement. All groups also showed increases in geometric mean fold rise of more than 3%.
Subgroup analyses showed no major differences between antibody responses in the hold vs continue groups. The anti-RBD IgG response was lower for abatacept and JAK inhibitors than for the other two drugs, but there was still no significant benefit to holding them for 2 weeks post vaccination.
Holding Drugs Leads to Disease Flares
On the flip side, there were significant differences between the two groups in their responses to the question: “Did you experience any flare or worsening of your autoimmune disease following your recent COVID-19 booster dose?” Overall, 27% of the hold group responded that they had, compared with just 13% of the continue group (P < .05). This difference was greatest in the JAK inhibitor group (33% vs 9%; P < .05).
Among those reporting flares or worsening disease, both the severity and the duration of the flares were about the same. “Interestingly, the duration is beyond a week for the majority of patients. The reason that’s important is, any symptoms that are so-called flare might simply be reactogenicity symptoms, and that might be confused for flare or disease worsening, but you see that a majority of patients actually have those symptoms extending beyond the week. Most of them are worsening in arthritis, as you might expect,” Curtis said in his presentation.
Asked what they did about the flare, only a minority of patients reported contacting a healthcare provider. In all, 68% of the hold group and 78% of the continue group took no action. That’s good in the sense that most of the flares weren’t severe, but it has implications for research, Curtis pointed out.
“A lot of times in the vaccine literature, people do retrospective chart review by looking to see what the doctor said as to whether the patient had a flare. And what this would tell you is patients may be reporting a lot of flares that their doctor doesn’t know anything about. So if you really want to know whether people are having a flare, even a mild flare, you really have to collect prospective data.”
COVID is Not the Last Pandemic
“These results are reassuring, although I think we need a bit more data on abatacept,” Myasoedova said, adding, “I was also interested in the outcomes, such as severe infections, that actually happened to these patients. What we see in the labs in their immune response is one thing, but then also important is what actually evolves in terms of the outcomes, especially with abatacept.”
Overall, she said, “I think it’s reassuring and definitely informs clinical practice going forward. But then probably we’ll learn more. What we’re hearing is COVID is not the last pandemic.”
The COVER trial receives support from AbbVie, BMS, Eli Lilly, Novartis, and Pfizer. Curtis has received research grants and consulting fees from AbbVie, Amgen, BMS, GSK, Eli Lilly, Novartis, Pfizer, Sanofi, and UCB. Myasoedova has no disclosures.
A version of this article first appeared on Medscape.com.
FROM ACR 2024
Gout and SGLT2 Inhibitors: Evidence Points to Reduced Need for ULT, Flare Drugs
WASHINGTON — Use of sodium-glucose cotransporter 2 inhibitors (SGLT2i) reduced the need for urate-lowering therapy (ULT) and gout flare therapies in people who had both type 2 diabetes (T2D) and gout, new research has found.
Data from a large US claims database showed that SGLT2i use was associated with a 31% lower rate of initiation of ULT. “This provides further support for the use of SLGT2i therapy in patients with gout, particularly those with high-risk multimorbidity and polypharmacy,” Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said in his presentation of the data at the annual meeting of the American College of Rheumatology.
The first agent of the SGLT2i class, dapagliflozin, was initially approved in the United States a decade ago for treating T2D. Since then, several other “flozins” have become available, and some have also received additional indications for heart failure and albuminuric chronic kidney disease. Several prior studies have linked SGLT2i use with lower rates of gout flares as well as lower likelihood of developing gout in the first place, although not all studies have found this benefit.
Asked about the clinical implications of the new data, Challener said in an interview that “I don’t think we’re quite at the point where this is changing gout management per se, but this just helps us understand that [SGT2is] may have a role at some point, maybe as a combination on top of another agent. Or, in some patients, it really may be enough if they’re already on an SGLT2i where we don’t need to jump to adding allopurinol. Maybe they have tophi, but they were just started on an SGLT2i and they’re not flaring. Typically, you would start those patients on allopurinol, but you could potentially just monitor them if they were just started on one of those [SGLT2i] agents.”
Asked to comment, session moderator J. Antonio Aviña-Zubieta, MD, PhD, head of the Division of Rheumatology at the University of British Columbia, Vancouver, Canada, and senior scientist at Arthritis Research Canada, said in an interview: “What I can see possibly happening when there’s more evidence is that SGLT2is may be used or even become standard of care as an adjuvant therapy to decrease flares, and by that, decrease the risk of complications.”
Reductions in ULT, Flares, and Healthcare Visits
The new study used administrative health data from the multicenter TriNetX Diamond network of electronic medical record and claims data from 92 healthcare sites with 212 million patients. Among those with both T2D and gout who were not taking ULT at baseline, a total of 16,104 initiated SGLT2is and 16,046 initiated glucagon-like peptide 1 receptor agonists (GLP-1 RA).
Propensity score matching was conducted for demographics including age, race, and sex; comorbidities; use of emergency, inpatient, and critical care services; medications; labs; and body mass index. That yielded 11,800 individuals each in the SGLT2i and GLP-1 RA groups.
Over 5 years, 9.9% of the SGLT2i group vs 13.4% of those using GLP-1 RA had initiated ULT, a significant difference with a hazard ratio (HR) of 0.69 (95% CI, 0.64-0.75). The risk for initiation of colchicine for gout flares was 4.7% with SGLT2i vs 6.0% for GLP-1 RA — also a significant difference with an HR of 0.74 (0.65-0.83).
Medical visits for gout occurred in 28.0% vs 28.4% of patients, which also reached statistical significance (HR, 0.94; 95% CI, 0.89-0.99).
Aviña-Zubieta, an author of one of the previous studies finding a reduction in gout flares with SGLT2i, said, “many patients do not want to start gout therapy until they start having more acute attacks. ... So, for a lot of people, it’s a burden taking another pill to prevent one attack. But, if you don’t treat it over time, the attacks come more often. So, can we still delay the initiation of therapy? If you’re not having that many flares, you’re decreasing the burden of the disease and polypharmacy, which I think is the potential benefit in the long run if you already have an indication for the therapy for diabetes. ... These data are supporting that.”
Indeed, Challener said these data can help in counseling patients. “Taking your SGLT2i for your heart failure and your diabetes is also providing some benefit for your gout, and we know that there is also cardiac benefit when gout is controlled.”
Challener and Aviña-Zubieta had no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — Use of sodium-glucose cotransporter 2 inhibitors (SGLT2i) reduced the need for urate-lowering therapy (ULT) and gout flare therapies in people who had both type 2 diabetes (T2D) and gout, new research has found.
Data from a large US claims database showed that SGLT2i use was associated with a 31% lower rate of initiation of ULT. “This provides further support for the use of SLGT2i therapy in patients with gout, particularly those with high-risk multimorbidity and polypharmacy,” Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said in his presentation of the data at the annual meeting of the American College of Rheumatology.
The first agent of the SGLT2i class, dapagliflozin, was initially approved in the United States a decade ago for treating T2D. Since then, several other “flozins” have become available, and some have also received additional indications for heart failure and albuminuric chronic kidney disease. Several prior studies have linked SGLT2i use with lower rates of gout flares as well as lower likelihood of developing gout in the first place, although not all studies have found this benefit.
Asked about the clinical implications of the new data, Challener said in an interview that “I don’t think we’re quite at the point where this is changing gout management per se, but this just helps us understand that [SGT2is] may have a role at some point, maybe as a combination on top of another agent. Or, in some patients, it really may be enough if they’re already on an SGLT2i where we don’t need to jump to adding allopurinol. Maybe they have tophi, but they were just started on an SGLT2i and they’re not flaring. Typically, you would start those patients on allopurinol, but you could potentially just monitor them if they were just started on one of those [SGLT2i] agents.”
Asked to comment, session moderator J. Antonio Aviña-Zubieta, MD, PhD, head of the Division of Rheumatology at the University of British Columbia, Vancouver, Canada, and senior scientist at Arthritis Research Canada, said in an interview: “What I can see possibly happening when there’s more evidence is that SGLT2is may be used or even become standard of care as an adjuvant therapy to decrease flares, and by that, decrease the risk of complications.”
Reductions in ULT, Flares, and Healthcare Visits
The new study used administrative health data from the multicenter TriNetX Diamond network of electronic medical record and claims data from 92 healthcare sites with 212 million patients. Among those with both T2D and gout who were not taking ULT at baseline, a total of 16,104 initiated SGLT2is and 16,046 initiated glucagon-like peptide 1 receptor agonists (GLP-1 RA).
Propensity score matching was conducted for demographics including age, race, and sex; comorbidities; use of emergency, inpatient, and critical care services; medications; labs; and body mass index. That yielded 11,800 individuals each in the SGLT2i and GLP-1 RA groups.
Over 5 years, 9.9% of the SGLT2i group vs 13.4% of those using GLP-1 RA had initiated ULT, a significant difference with a hazard ratio (HR) of 0.69 (95% CI, 0.64-0.75). The risk for initiation of colchicine for gout flares was 4.7% with SGLT2i vs 6.0% for GLP-1 RA — also a significant difference with an HR of 0.74 (0.65-0.83).
Medical visits for gout occurred in 28.0% vs 28.4% of patients, which also reached statistical significance (HR, 0.94; 95% CI, 0.89-0.99).
Aviña-Zubieta, an author of one of the previous studies finding a reduction in gout flares with SGLT2i, said, “many patients do not want to start gout therapy until they start having more acute attacks. ... So, for a lot of people, it’s a burden taking another pill to prevent one attack. But, if you don’t treat it over time, the attacks come more often. So, can we still delay the initiation of therapy? If you’re not having that many flares, you’re decreasing the burden of the disease and polypharmacy, which I think is the potential benefit in the long run if you already have an indication for the therapy for diabetes. ... These data are supporting that.”
Indeed, Challener said these data can help in counseling patients. “Taking your SGLT2i for your heart failure and your diabetes is also providing some benefit for your gout, and we know that there is also cardiac benefit when gout is controlled.”
Challener and Aviña-Zubieta had no disclosures.
A version of this article first appeared on Medscape.com.
WASHINGTON — Use of sodium-glucose cotransporter 2 inhibitors (SGLT2i) reduced the need for urate-lowering therapy (ULT) and gout flare therapies in people who had both type 2 diabetes (T2D) and gout, new research has found.
Data from a large US claims database showed that SGLT2i use was associated with a 31% lower rate of initiation of ULT. “This provides further support for the use of SLGT2i therapy in patients with gout, particularly those with high-risk multimorbidity and polypharmacy,” Greg Challener, MD, a postdoctoral fellow at the Rheumatology and Allergy Clinical Epidemiology Research Center, Massachusetts General Hospital, Boston, said in his presentation of the data at the annual meeting of the American College of Rheumatology.
The first agent of the SGLT2i class, dapagliflozin, was initially approved in the United States a decade ago for treating T2D. Since then, several other “flozins” have become available, and some have also received additional indications for heart failure and albuminuric chronic kidney disease. Several prior studies have linked SGLT2i use with lower rates of gout flares as well as lower likelihood of developing gout in the first place, although not all studies have found this benefit.
Asked about the clinical implications of the new data, Challener said in an interview that “I don’t think we’re quite at the point where this is changing gout management per se, but this just helps us understand that [SGT2is] may have a role at some point, maybe as a combination on top of another agent. Or, in some patients, it really may be enough if they’re already on an SGLT2i where we don’t need to jump to adding allopurinol. Maybe they have tophi, but they were just started on an SGLT2i and they’re not flaring. Typically, you would start those patients on allopurinol, but you could potentially just monitor them if they were just started on one of those [SGLT2i] agents.”
Asked to comment, session moderator J. Antonio Aviña-Zubieta, MD, PhD, head of the Division of Rheumatology at the University of British Columbia, Vancouver, Canada, and senior scientist at Arthritis Research Canada, said in an interview: “What I can see possibly happening when there’s more evidence is that SGLT2is may be used or even become standard of care as an adjuvant therapy to decrease flares, and by that, decrease the risk of complications.”
Reductions in ULT, Flares, and Healthcare Visits
The new study used administrative health data from the multicenter TriNetX Diamond network of electronic medical record and claims data from 92 healthcare sites with 212 million patients. Among those with both T2D and gout who were not taking ULT at baseline, a total of 16,104 initiated SGLT2is and 16,046 initiated glucagon-like peptide 1 receptor agonists (GLP-1 RA).
Propensity score matching was conducted for demographics including age, race, and sex; comorbidities; use of emergency, inpatient, and critical care services; medications; labs; and body mass index. That yielded 11,800 individuals each in the SGLT2i and GLP-1 RA groups.
Over 5 years, 9.9% of the SGLT2i group vs 13.4% of those using GLP-1 RA had initiated ULT, a significant difference with a hazard ratio (HR) of 0.69 (95% CI, 0.64-0.75). The risk for initiation of colchicine for gout flares was 4.7% with SGLT2i vs 6.0% for GLP-1 RA — also a significant difference with an HR of 0.74 (0.65-0.83).
Medical visits for gout occurred in 28.0% vs 28.4% of patients, which also reached statistical significance (HR, 0.94; 95% CI, 0.89-0.99).
Aviña-Zubieta, an author of one of the previous studies finding a reduction in gout flares with SGLT2i, said, “many patients do not want to start gout therapy until they start having more acute attacks. ... So, for a lot of people, it’s a burden taking another pill to prevent one attack. But, if you don’t treat it over time, the attacks come more often. So, can we still delay the initiation of therapy? If you’re not having that many flares, you’re decreasing the burden of the disease and polypharmacy, which I think is the potential benefit in the long run if you already have an indication for the therapy for diabetes. ... These data are supporting that.”
Indeed, Challener said these data can help in counseling patients. “Taking your SGLT2i for your heart failure and your diabetes is also providing some benefit for your gout, and we know that there is also cardiac benefit when gout is controlled.”
Challener and Aviña-Zubieta had no disclosures.
A version of this article first appeared on Medscape.com.
FROM ACR 2024
First Phase 3 Drug Trial in IgG4-Related Disease Has Success
WASHINGTON — The B cell–depleting agent inebilizumab (Uplizna) dramatically reduced the risk of flares and increased year-long remission of IgG4-related disease (RD), new research has found.
In a phase 3, multicenter, double-blind, randomized, placebo-controlled trial of 135 adults with active IgG4-RD, treatment with inebilizumab resulted in a significant 87% reduction in flare risk and nearly fivefold greater likelihood of flare-free remission at 1 year. The results were published online November 14 in The New England Journal of Medicine and were presented at the annual meeting of the American College of Rheumatology (ACR).
The drug’s manufacturer, Amgen, released top-line results of the trial, called MITIGATE, in June 2024.
Until now, the mainstay of management for the chronic multiorgan disease IgG4-RD has been glucocorticoids, which can cause numerous adverse effects. “It is hoped that inebilizumab can be used as an important steroid-sparing medication in this disease to reduce steroid toxicity,” lead author John H. Stone, MD, professor of medicine at Harvard Medical School, Boston, Massachusetts, said in an interview, noting that it may not entirely eliminate the need for steroid treatment, but for many, it appears to work after the remission induction period as a monotherapy without steroids.
Asked to comment, Leonard H. Calabrese, DO, head of the Section of Clinical Immunology and manager of the Clinical Immunology Clinic at the Cleveland Clinic, Ohio, said: “There has been anecdotal or observational evidence for some effect with other immunosuppressive agents, including rituximab, but no robust clinical trial until this study. This clearly has demonstrated efficacy by reducing the risk of flares. And most importantly, putting people into remission means no active disease in any given organ. ... This gives us another tool in the toolbox to attack B cell–directed diseases, and I think it really makes a lot of sense.”
Calabrese cautioned, though, that “this is a disease that extends over many years. This is just a 1-year study. Label extensions will be important.”
And several questions remain, Calabrese noted: “How long do patients need to remain on drug? What will happen when the drug is stopped? Can they be retreated? These are the natural questions that arise in any sentinel study like this. But this is extremely encouraging. And I think it’s great for patients. I also think it’s a clarion call to increase awareness about this disease since there’s now strong evidence of effective treatment.”
Underrecognized, Often Misdiagnosed as Cancer
Indeed, IgG4-RD, a chronic, relapsing, autoimmune, fibro-inflammatory multiorgan disease, was only first described in Japan in 2003. Since then, it has been reported all over the world yet remains vastly underrecognized. It is often misdiagnosed as cancer because it produces lesions in multiple organs. It received an ICD-10 code only about a year ago. A previous study estimated a prevalence of about 5.3 persons per 100,000 but that is likely to be a three- to fourfold underestimate, said Stone, who is also executive chairman of the IgG4ward! Foundation.
“Nobody had heard of the disease until about 20 years ago. ... And there are many people in the world who have still not heard of it despite the fact that it is a multiorgan autoimmune disease and is probably as common, or more common, than many other diseases that rheumatologists spend a lot of time thinking about, such as scleroderma.”
While knowledge about the disease is increasing in rheumatology circles, it’s less well-recognized among many of the specialties where patients present, depending on the location of their lesions. These include gastroenterology, ophthalmology, pulmonary medicine, neurology, and nephrology. “All would be likely to see this disease,” Stone said.
The disease can be mistaken for tumors in many of those locations and even as metastatic cancer, he noted, adding that “any time a patient has a mass lesion in a typical organ, the pancreas, the major salivary glands, the lungs, or the kidneys, this should be on the differential diagnosis.”
The diagnosis of IgG4-RD is a clinical one, involving “quadrangulation between clinical features, serological findings, IgG4 levels in the blood, radiology studies, and then pathology biopsies when those are available,” Stone said.
Calabrese characterized the current situation as “we’re all blind men on the elephant. To the neurologist or the neurosurgeon, it’s a mass in the brain. It could present to the ophthalmologist as an [eye] tumor. It can be thyroid gland failure, pulmonary disease, retroperitoneal fibrosis, hepatobiliary disease, and beyond. So, whoever sees that patient, there’s often a long lag time in recognizing it.”
And interestingly, Stone noted that unlike other autoimmune diseases, IgG4-RD primarily affects middle-aged men rather than younger-to-middle-aged women. And when IgG4-RD is diagnosed, glucocorticoid treatment can be particularly toxic when the pancreas is involved, heightening the risk for hyperglycemia and potentially causing diabetes.
Dramatic Improvement in Flares, Remission Achievement
MITIGATE is a phase 3, multicenter, double-blind, randomized, placebo-controlled trial in which 135 adults (mean age 58.2 years, 88 men) with active IgG4-RD were randomized 1:1 to receive 300-mg intravenous infusions of inebilizumab or placebo on days 1 and 15, and again at week 26. At baseline, 62 (45.9%) participants had newly diagnosed IgG4-RD and 73 (54.1%) had recurrent disease.
Both groups received identical glucocorticoid tapers. Overall, 127 (94.1%) completed the 52 weeks of treatment.
By 52 weeks, only seven patients in the inebilizumab group (10%) had experienced disease flares vs 40 (60%) in the placebo group, a significant difference with a hazard ratio of 0.13 (P < .001).
The percentage of participants achieving flare-free, treatment-free complete remission was 59 with inebilizumab (57%), compared with just 15 (22%) in the placebo group (odds ratio [OR], 4.68; P < .001). And for flare-free, glucocorticoid-free complete remission, those proportions were 40 (59%) vs 15 (22%), respectively (OR, 4.96; P < .001).
Excluding the 8-week glucocorticoid taper period, mean total glucocorticoid use was 1264.2 mg less in the inebilizumab than the placebo group, a significant reduction. Overall, 61 participants (90%) were able to entirely discontinue glucocorticoids during the trial, compared with just 25 (37%) in the placebo group.
Adverse events of grade 3 or higher occurred in 12 participants (18%) in the inebilizumab group and 8 (12%) in the placebo group; serious adverse events occurred in 12 (18%) and 6 (9%), respectively. However, no serious adverse event occurred in more than one participant, and there were no deaths. Adverse events led to withdrawal from the trial in six patients (9%) in the inebilizumab group and three patients (4%) in the placebo group.
Adverse events that occurred in more than 10% of participants in the inebilizumab group were COVID-19 in 16 participants (24%), lymphopenia in 11 (16%), and urinary tract infection in 8 (12%).
Importantly, Stone noted, B-cell depletion can reduce responses to vaccines, so patients should receive all recommended vaccinations, including COVID-19, influenza, respiratory syncytial virus, and others, prior to initiating therapy.
Uplizna (inebilizumab-cdon) was approved by the Food and Drug Administration (FDA) for the treatment of neuromyelitis optica spectrum disorder in 2020. In October 2024, the FDA granted Amgen breakthrough therapy designation for use in IgG4-RD. The company is also developing the drug for use in myasthenia gravis.
The study was funded by Amgen. Stone has reported being a consultant for Amgen, Zenas, Argenx, Bristol Myers Squibb, Novartis, Sanofi, and Horizon Pharma. Calabrese has reported being a consultant and/or speaker for Amgen, AstraZeneca, Jansen, Sanofi, and UCB.
A version of this article first appeared on Medscape.com.
WASHINGTON — The B cell–depleting agent inebilizumab (Uplizna) dramatically reduced the risk of flares and increased year-long remission of IgG4-related disease (RD), new research has found.
In a phase 3, multicenter, double-blind, randomized, placebo-controlled trial of 135 adults with active IgG4-RD, treatment with inebilizumab resulted in a significant 87% reduction in flare risk and nearly fivefold greater likelihood of flare-free remission at 1 year. The results were published online November 14 in The New England Journal of Medicine and were presented at the annual meeting of the American College of Rheumatology (ACR).
The drug’s manufacturer, Amgen, released top-line results of the trial, called MITIGATE, in June 2024.
Until now, the mainstay of management for the chronic multiorgan disease IgG4-RD has been glucocorticoids, which can cause numerous adverse effects. “It is hoped that inebilizumab can be used as an important steroid-sparing medication in this disease to reduce steroid toxicity,” lead author John H. Stone, MD, professor of medicine at Harvard Medical School, Boston, Massachusetts, said in an interview, noting that it may not entirely eliminate the need for steroid treatment, but for many, it appears to work after the remission induction period as a monotherapy without steroids.
Asked to comment, Leonard H. Calabrese, DO, head of the Section of Clinical Immunology and manager of the Clinical Immunology Clinic at the Cleveland Clinic, Ohio, said: “There has been anecdotal or observational evidence for some effect with other immunosuppressive agents, including rituximab, but no robust clinical trial until this study. This clearly has demonstrated efficacy by reducing the risk of flares. And most importantly, putting people into remission means no active disease in any given organ. ... This gives us another tool in the toolbox to attack B cell–directed diseases, and I think it really makes a lot of sense.”
Calabrese cautioned, though, that “this is a disease that extends over many years. This is just a 1-year study. Label extensions will be important.”
And several questions remain, Calabrese noted: “How long do patients need to remain on drug? What will happen when the drug is stopped? Can they be retreated? These are the natural questions that arise in any sentinel study like this. But this is extremely encouraging. And I think it’s great for patients. I also think it’s a clarion call to increase awareness about this disease since there’s now strong evidence of effective treatment.”
Underrecognized, Often Misdiagnosed as Cancer
Indeed, IgG4-RD, a chronic, relapsing, autoimmune, fibro-inflammatory multiorgan disease, was only first described in Japan in 2003. Since then, it has been reported all over the world yet remains vastly underrecognized. It is often misdiagnosed as cancer because it produces lesions in multiple organs. It received an ICD-10 code only about a year ago. A previous study estimated a prevalence of about 5.3 persons per 100,000 but that is likely to be a three- to fourfold underestimate, said Stone, who is also executive chairman of the IgG4ward! Foundation.
“Nobody had heard of the disease until about 20 years ago. ... And there are many people in the world who have still not heard of it despite the fact that it is a multiorgan autoimmune disease and is probably as common, or more common, than many other diseases that rheumatologists spend a lot of time thinking about, such as scleroderma.”
While knowledge about the disease is increasing in rheumatology circles, it’s less well-recognized among many of the specialties where patients present, depending on the location of their lesions. These include gastroenterology, ophthalmology, pulmonary medicine, neurology, and nephrology. “All would be likely to see this disease,” Stone said.
The disease can be mistaken for tumors in many of those locations and even as metastatic cancer, he noted, adding that “any time a patient has a mass lesion in a typical organ, the pancreas, the major salivary glands, the lungs, or the kidneys, this should be on the differential diagnosis.”
The diagnosis of IgG4-RD is a clinical one, involving “quadrangulation between clinical features, serological findings, IgG4 levels in the blood, radiology studies, and then pathology biopsies when those are available,” Stone said.
Calabrese characterized the current situation as “we’re all blind men on the elephant. To the neurologist or the neurosurgeon, it’s a mass in the brain. It could present to the ophthalmologist as an [eye] tumor. It can be thyroid gland failure, pulmonary disease, retroperitoneal fibrosis, hepatobiliary disease, and beyond. So, whoever sees that patient, there’s often a long lag time in recognizing it.”
And interestingly, Stone noted that unlike other autoimmune diseases, IgG4-RD primarily affects middle-aged men rather than younger-to-middle-aged women. And when IgG4-RD is diagnosed, glucocorticoid treatment can be particularly toxic when the pancreas is involved, heightening the risk for hyperglycemia and potentially causing diabetes.
Dramatic Improvement in Flares, Remission Achievement
MITIGATE is a phase 3, multicenter, double-blind, randomized, placebo-controlled trial in which 135 adults (mean age 58.2 years, 88 men) with active IgG4-RD were randomized 1:1 to receive 300-mg intravenous infusions of inebilizumab or placebo on days 1 and 15, and again at week 26. At baseline, 62 (45.9%) participants had newly diagnosed IgG4-RD and 73 (54.1%) had recurrent disease.
Both groups received identical glucocorticoid tapers. Overall, 127 (94.1%) completed the 52 weeks of treatment.
By 52 weeks, only seven patients in the inebilizumab group (10%) had experienced disease flares vs 40 (60%) in the placebo group, a significant difference with a hazard ratio of 0.13 (P < .001).
The percentage of participants achieving flare-free, treatment-free complete remission was 59 with inebilizumab (57%), compared with just 15 (22%) in the placebo group (odds ratio [OR], 4.68; P < .001). And for flare-free, glucocorticoid-free complete remission, those proportions were 40 (59%) vs 15 (22%), respectively (OR, 4.96; P < .001).
Excluding the 8-week glucocorticoid taper period, mean total glucocorticoid use was 1264.2 mg less in the inebilizumab than the placebo group, a significant reduction. Overall, 61 participants (90%) were able to entirely discontinue glucocorticoids during the trial, compared with just 25 (37%) in the placebo group.
Adverse events of grade 3 or higher occurred in 12 participants (18%) in the inebilizumab group and 8 (12%) in the placebo group; serious adverse events occurred in 12 (18%) and 6 (9%), respectively. However, no serious adverse event occurred in more than one participant, and there were no deaths. Adverse events led to withdrawal from the trial in six patients (9%) in the inebilizumab group and three patients (4%) in the placebo group.
Adverse events that occurred in more than 10% of participants in the inebilizumab group were COVID-19 in 16 participants (24%), lymphopenia in 11 (16%), and urinary tract infection in 8 (12%).
Importantly, Stone noted, B-cell depletion can reduce responses to vaccines, so patients should receive all recommended vaccinations, including COVID-19, influenza, respiratory syncytial virus, and others, prior to initiating therapy.
Uplizna (inebilizumab-cdon) was approved by the Food and Drug Administration (FDA) for the treatment of neuromyelitis optica spectrum disorder in 2020. In October 2024, the FDA granted Amgen breakthrough therapy designation for use in IgG4-RD. The company is also developing the drug for use in myasthenia gravis.
The study was funded by Amgen. Stone has reported being a consultant for Amgen, Zenas, Argenx, Bristol Myers Squibb, Novartis, Sanofi, and Horizon Pharma. Calabrese has reported being a consultant and/or speaker for Amgen, AstraZeneca, Jansen, Sanofi, and UCB.
A version of this article first appeared on Medscape.com.
WASHINGTON — The B cell–depleting agent inebilizumab (Uplizna) dramatically reduced the risk of flares and increased year-long remission of IgG4-related disease (RD), new research has found.
In a phase 3, multicenter, double-blind, randomized, placebo-controlled trial of 135 adults with active IgG4-RD, treatment with inebilizumab resulted in a significant 87% reduction in flare risk and nearly fivefold greater likelihood of flare-free remission at 1 year. The results were published online November 14 in The New England Journal of Medicine and were presented at the annual meeting of the American College of Rheumatology (ACR).
The drug’s manufacturer, Amgen, released top-line results of the trial, called MITIGATE, in June 2024.
Until now, the mainstay of management for the chronic multiorgan disease IgG4-RD has been glucocorticoids, which can cause numerous adverse effects. “It is hoped that inebilizumab can be used as an important steroid-sparing medication in this disease to reduce steroid toxicity,” lead author John H. Stone, MD, professor of medicine at Harvard Medical School, Boston, Massachusetts, said in an interview, noting that it may not entirely eliminate the need for steroid treatment, but for many, it appears to work after the remission induction period as a monotherapy without steroids.
Asked to comment, Leonard H. Calabrese, DO, head of the Section of Clinical Immunology and manager of the Clinical Immunology Clinic at the Cleveland Clinic, Ohio, said: “There has been anecdotal or observational evidence for some effect with other immunosuppressive agents, including rituximab, but no robust clinical trial until this study. This clearly has demonstrated efficacy by reducing the risk of flares. And most importantly, putting people into remission means no active disease in any given organ. ... This gives us another tool in the toolbox to attack B cell–directed diseases, and I think it really makes a lot of sense.”
Calabrese cautioned, though, that “this is a disease that extends over many years. This is just a 1-year study. Label extensions will be important.”
And several questions remain, Calabrese noted: “How long do patients need to remain on drug? What will happen when the drug is stopped? Can they be retreated? These are the natural questions that arise in any sentinel study like this. But this is extremely encouraging. And I think it’s great for patients. I also think it’s a clarion call to increase awareness about this disease since there’s now strong evidence of effective treatment.”
Underrecognized, Often Misdiagnosed as Cancer
Indeed, IgG4-RD, a chronic, relapsing, autoimmune, fibro-inflammatory multiorgan disease, was only first described in Japan in 2003. Since then, it has been reported all over the world yet remains vastly underrecognized. It is often misdiagnosed as cancer because it produces lesions in multiple organs. It received an ICD-10 code only about a year ago. A previous study estimated a prevalence of about 5.3 persons per 100,000 but that is likely to be a three- to fourfold underestimate, said Stone, who is also executive chairman of the IgG4ward! Foundation.
“Nobody had heard of the disease until about 20 years ago. ... And there are many people in the world who have still not heard of it despite the fact that it is a multiorgan autoimmune disease and is probably as common, or more common, than many other diseases that rheumatologists spend a lot of time thinking about, such as scleroderma.”
While knowledge about the disease is increasing in rheumatology circles, it’s less well-recognized among many of the specialties where patients present, depending on the location of their lesions. These include gastroenterology, ophthalmology, pulmonary medicine, neurology, and nephrology. “All would be likely to see this disease,” Stone said.
The disease can be mistaken for tumors in many of those locations and even as metastatic cancer, he noted, adding that “any time a patient has a mass lesion in a typical organ, the pancreas, the major salivary glands, the lungs, or the kidneys, this should be on the differential diagnosis.”
The diagnosis of IgG4-RD is a clinical one, involving “quadrangulation between clinical features, serological findings, IgG4 levels in the blood, radiology studies, and then pathology biopsies when those are available,” Stone said.
Calabrese characterized the current situation as “we’re all blind men on the elephant. To the neurologist or the neurosurgeon, it’s a mass in the brain. It could present to the ophthalmologist as an [eye] tumor. It can be thyroid gland failure, pulmonary disease, retroperitoneal fibrosis, hepatobiliary disease, and beyond. So, whoever sees that patient, there’s often a long lag time in recognizing it.”
And interestingly, Stone noted that unlike other autoimmune diseases, IgG4-RD primarily affects middle-aged men rather than younger-to-middle-aged women. And when IgG4-RD is diagnosed, glucocorticoid treatment can be particularly toxic when the pancreas is involved, heightening the risk for hyperglycemia and potentially causing diabetes.
Dramatic Improvement in Flares, Remission Achievement
MITIGATE is a phase 3, multicenter, double-blind, randomized, placebo-controlled trial in which 135 adults (mean age 58.2 years, 88 men) with active IgG4-RD were randomized 1:1 to receive 300-mg intravenous infusions of inebilizumab or placebo on days 1 and 15, and again at week 26. At baseline, 62 (45.9%) participants had newly diagnosed IgG4-RD and 73 (54.1%) had recurrent disease.
Both groups received identical glucocorticoid tapers. Overall, 127 (94.1%) completed the 52 weeks of treatment.
By 52 weeks, only seven patients in the inebilizumab group (10%) had experienced disease flares vs 40 (60%) in the placebo group, a significant difference with a hazard ratio of 0.13 (P < .001).
The percentage of participants achieving flare-free, treatment-free complete remission was 59 with inebilizumab (57%), compared with just 15 (22%) in the placebo group (odds ratio [OR], 4.68; P < .001). And for flare-free, glucocorticoid-free complete remission, those proportions were 40 (59%) vs 15 (22%), respectively (OR, 4.96; P < .001).
Excluding the 8-week glucocorticoid taper period, mean total glucocorticoid use was 1264.2 mg less in the inebilizumab than the placebo group, a significant reduction. Overall, 61 participants (90%) were able to entirely discontinue glucocorticoids during the trial, compared with just 25 (37%) in the placebo group.
Adverse events of grade 3 or higher occurred in 12 participants (18%) in the inebilizumab group and 8 (12%) in the placebo group; serious adverse events occurred in 12 (18%) and 6 (9%), respectively. However, no serious adverse event occurred in more than one participant, and there were no deaths. Adverse events led to withdrawal from the trial in six patients (9%) in the inebilizumab group and three patients (4%) in the placebo group.
Adverse events that occurred in more than 10% of participants in the inebilizumab group were COVID-19 in 16 participants (24%), lymphopenia in 11 (16%), and urinary tract infection in 8 (12%).
Importantly, Stone noted, B-cell depletion can reduce responses to vaccines, so patients should receive all recommended vaccinations, including COVID-19, influenza, respiratory syncytial virus, and others, prior to initiating therapy.
Uplizna (inebilizumab-cdon) was approved by the Food and Drug Administration (FDA) for the treatment of neuromyelitis optica spectrum disorder in 2020. In October 2024, the FDA granted Amgen breakthrough therapy designation for use in IgG4-RD. The company is also developing the drug for use in myasthenia gravis.
The study was funded by Amgen. Stone has reported being a consultant for Amgen, Zenas, Argenx, Bristol Myers Squibb, Novartis, Sanofi, and Horizon Pharma. Calabrese has reported being a consultant and/or speaker for Amgen, AstraZeneca, Jansen, Sanofi, and UCB.
A version of this article first appeared on Medscape.com.
FROM ACR 2024
Weight Loss Treatments Beyond the Reach of Many in Need
SAN ANTONIO — Weight loss treatments aren’t reaching many of the people who need them most because of coverage barriers, new data suggested.
Findings from three studies presented at The Obesity Society’s Obesity Week 2024 meeting illustrate different aspects of the problem.
One, presented by Alissa S. Chen, MD, MPH, a postdoctoral fellow at Yale University, New Haven, Connecticut, found that people with obesity, particularly those with cardiovascular disease (CVD) and those who are Black and Hispanic, have high rates of cost-related prescription drug rationing. Those findings were simultaneously published as a research letter in JAMA Network Open.
“The implications are that structural barriers impede access to medications for Black and Hispanic adults with obesity, which might worsen if there’s not expanding coverage for GLP-1 RAs [glucagon-like peptide 1 receptor agonists], and it’s possible that broader insurance coverage could ameliorate some of these issues,” Chen said.
She noted that patients don’t always volunteer that information. “In my clinical practice, I always start by saying something like, ‘I have a lot of patients who can’t afford their medications. In the last week, was there a time [you didn’t take your medications due to cost]?’ ”
State Medicaid programs vary widely in the degree to which they cover weight loss treatments. But not a single one covers all modalities — nutrition counseling (NC), intensive behavioral therapy (IBT), obesity medications (OMs), and metabolic and bariatric surgery (MBS) — without restrictions or limitations, and only seven states cover them all with restrictions, according to a dual presentation by Christine Gallagher, MPAff, associate director for research and policy with the STOP Obesity Alliance at George Washington University, Washington, DC, and Tracy Zvenyach, PhD, MS, RN, director of policy strategy and alliances at the Obesity Action Coalition, also in Washington, DC.
Detailed Medicaid coverage data for each state are posted on the STOP Obesity Alliance website. (As of now, Medicare doesn’t cover medications specifically for obesity at all.)
A third presentation, by Treah Haggerty, MD, of the Department of Family Medicine and director of the Pediatric Medical Weight Management program at West Virginia University, Morgantown, was of a qualitative descriptive study exploring the impact on 22 individuals enrolled in a medical weight loss management program whose state employee insurance carrier made a policy decision to stop covering all anti-OMs in March 2024. All had been prescribed GLP-1 agonists for weight loss, and the decision forced most to stop using them.
Those findings were published in the September 2024 issue of Obesity Pillars.
“Patients perceive the discontinuation of anti-obesity medication coverage as stigmatizing and unjust, leading to feelings of hopelessness and fear. With more insurance companies denying coverage for these costly medications, more information is needed to identify best ways to address the loss of coverage with patients. Clinical management of these patients should incorporate evidence-based obesity treatments while navigating insurance constraints,” Haggerty said.
Create a Safe Space to Discuss the Barriers
Asked to comment, Session Moderator John D. Clark, MD, PhD, chief population health officer at Sharp Rees-Stealy Medical Group, San Diego, California, told Medscape Medical News, “Health systems and payers are determining what can and can’t be covered, and at the end of the day, it frequently comes down to finances…I think the big challenge is really identifying patients who may have the greatest need. ... If we have limited resources, how and where should we be directing those resources? I would say the current system hasn’t really answered that question or identified patients for whom we would say that the cost truly is less than either the financial or long-term health benefits.”
But Clark said, “When some of these newer anti-obesity medications are able to go generic and be less expensive, which will happen eventually, I think things will change ... and in the future, there will be more options on the market as well.”
In the meantime, he advised that clinicians “try to have conversations with patients about these barriers, acknowledge that these barriers exist, and create a safe space to discuss those barriers. ... Let’s see where we are right now, and let’s come up with a plan.”
People With Obesity More Susceptible to Drug Rationing
Chen reported on a sample of 51,720 adults who participated in the 2020-2022 National Health Interview Survey who did not have diabetes and who used at least one prescription medication of any type. Of those, 80% were White, 9.7% were Hispanic, and 9.7% were Black, and 33.9% overall had obesity.
Cost-related prescription rationing, defined as any self-reported skipping, taking less, or delaying filling a prescription to save money, was reported by 8.3% of those with obesity vs 5.9% without. After adjustment, rationing among those with obesity was significantly associated with younger age (aged 18-44 years), female sex, lower incomes, lack of health insurance coverage, and CVD.
The adjusted estimated probability of cost-related prescription drug rationing was 7.4% for those with CVD vs 4.4% for those without. By race/ethnicity, the proportions reporting rationing were 7.7%, 9.8%, and 10.7% for White, Black, and Hispanics, respectively.
“Given that few insurance providers cover GLP-1 RAs for obesity, cost-related prescription drug rationing could be exacerbated if patients were prescribed GLP-1 RAs at their current price of more than $1000 a month,” Chen noted, adding that the high prices could worsen health disparities among Black and Hispanic individuals with obesity.
Medicaid Coverage Lacking for All Obesity Treatments
For their project, Gallagher and Zvenyach delved into a database that aggregates Medicaid manuals, fee schedules, statutes, regulations, and preferred drug lists for each US state, both for Medicaid fee for service and top Medicaid managed care plans, in order to determine 2024 levels of coverage for NC, IBT, OMs, and MBS for adults with obesity.
No state provides coverage for all those treatments without either limitations — such as body mass index (BMI) cutoffs, age, or “comorbidity regardless of body mass index (BMI)” for OM and MBS — or outright restrictions, such as “proof of failed attempts.” And only seven states provide coverage for the four modalities “with limitations”: California, Arizona, New Mexico, South Carolina, Delaware, Rhode Island, and Massachusetts.
Twenty-two states don’t cover NC, although just one state doesn’t cover IBT. Overall, 37 states don’t cover OMs, and other states ranged considerably in various restrictions and limitations for OMs and MBS. Only four states fully covered the surgery without limitations or restrictions.
“The vast majority of states have significant barriers and conditions of coverage for obesity care,” Gallagher said.
Zvenyach added, “Most of the applied exclusions, limitations, or restrictions do not align with evidence-based practice standards or guidelines.”
When Coverage Stops, Hopelessness and Anger Emerge
Haggerty and colleagues’ research involved semi-structured interviews of the 22 participants — all of them women — who had lost their obesity medication coverage due to their insurers’ decision. Four themes emerged:
- 1. Feelings of hope replaced by hopelessness upon loss of medication coverage: One person said, “I’m afraid for my mental health. It’s tough to be in a situation where you’re never right. And it doesn’t matter what you do; it’s not going to work, and then to have just a glimmer of hope, a little spark of hey, look, this might help. And for someone else to take that away from you for no reason. I don’t know what am I supposed to do.”
- 2. Anger regarding the perceived injustice of anti-obesity medication coverage termination: For example, “They can pay for heart attacks, they could pay for me to have a stroke, they could pay for me to have diabetes, but they won’t let me have this one medicine that could take all of that away. Makes no sense.”
- 3. Perceptions of past and present stigma within the healthcare system and insurance company: “I’m not trying for vanity. I’m way too old to be a Victoria’s Secret model. I’m not trying to do it to be cute and skinny and hot. I just want to make it through a day of work and not be exhausted.”
- 4. Generational influences on obesity treatment: “I’m married, and my husband, since I’ve started this medicine, he’s been eating better. He’s been eating what I eat, and he’s been losing weight as well.”
- Some participants said they planned to cope in different ways, including trying to obtain compounded versions of the drugs from “spas” or online pharmacies, as well as skipping doses, reducing doses, or sharing medications — in other words, rationing.
Asked by this news organization what clinicians should keep in mind, Haggerty said, “that there are big barriers and that we need to take care of the patients within this system that has their arm tied behind their back.
Chen was funded by a grant from the National Institute on Aging outside the submitted work, and she was funded as a Yale National Clinician Scholar. A coauthor received grants from the Food and Drug Administration, Johnson & Johnson, the National Institutes of Health, the Agency for Healthcare Research and Quality, and Arnold Ventures. Another coauthor reported receiving personal fees from UpToDate and the Centers for Medicare & Medicaid Services. Gallagher has received research funding from Altimmune, Amgen, Boehringer Ingelheim, Currax, Eli Lilly and Company, Found, Novo Nordisk, Pfizer, Structure Therapeutics, and WeightWatchers. Haggerty reported article publishing charge was provided by West Virginia Alliance for Creative Health Solutions. Zvenyach and Clark had no disclosures.
A version of this article first appeared on Medscape.com.
SAN ANTONIO — Weight loss treatments aren’t reaching many of the people who need them most because of coverage barriers, new data suggested.
Findings from three studies presented at The Obesity Society’s Obesity Week 2024 meeting illustrate different aspects of the problem.
One, presented by Alissa S. Chen, MD, MPH, a postdoctoral fellow at Yale University, New Haven, Connecticut, found that people with obesity, particularly those with cardiovascular disease (CVD) and those who are Black and Hispanic, have high rates of cost-related prescription drug rationing. Those findings were simultaneously published as a research letter in JAMA Network Open.
“The implications are that structural barriers impede access to medications for Black and Hispanic adults with obesity, which might worsen if there’s not expanding coverage for GLP-1 RAs [glucagon-like peptide 1 receptor agonists], and it’s possible that broader insurance coverage could ameliorate some of these issues,” Chen said.
She noted that patients don’t always volunteer that information. “In my clinical practice, I always start by saying something like, ‘I have a lot of patients who can’t afford their medications. In the last week, was there a time [you didn’t take your medications due to cost]?’ ”
State Medicaid programs vary widely in the degree to which they cover weight loss treatments. But not a single one covers all modalities — nutrition counseling (NC), intensive behavioral therapy (IBT), obesity medications (OMs), and metabolic and bariatric surgery (MBS) — without restrictions or limitations, and only seven states cover them all with restrictions, according to a dual presentation by Christine Gallagher, MPAff, associate director for research and policy with the STOP Obesity Alliance at George Washington University, Washington, DC, and Tracy Zvenyach, PhD, MS, RN, director of policy strategy and alliances at the Obesity Action Coalition, also in Washington, DC.
Detailed Medicaid coverage data for each state are posted on the STOP Obesity Alliance website. (As of now, Medicare doesn’t cover medications specifically for obesity at all.)
A third presentation, by Treah Haggerty, MD, of the Department of Family Medicine and director of the Pediatric Medical Weight Management program at West Virginia University, Morgantown, was of a qualitative descriptive study exploring the impact on 22 individuals enrolled in a medical weight loss management program whose state employee insurance carrier made a policy decision to stop covering all anti-OMs in March 2024. All had been prescribed GLP-1 agonists for weight loss, and the decision forced most to stop using them.
Those findings were published in the September 2024 issue of Obesity Pillars.
“Patients perceive the discontinuation of anti-obesity medication coverage as stigmatizing and unjust, leading to feelings of hopelessness and fear. With more insurance companies denying coverage for these costly medications, more information is needed to identify best ways to address the loss of coverage with patients. Clinical management of these patients should incorporate evidence-based obesity treatments while navigating insurance constraints,” Haggerty said.
Create a Safe Space to Discuss the Barriers
Asked to comment, Session Moderator John D. Clark, MD, PhD, chief population health officer at Sharp Rees-Stealy Medical Group, San Diego, California, told Medscape Medical News, “Health systems and payers are determining what can and can’t be covered, and at the end of the day, it frequently comes down to finances…I think the big challenge is really identifying patients who may have the greatest need. ... If we have limited resources, how and where should we be directing those resources? I would say the current system hasn’t really answered that question or identified patients for whom we would say that the cost truly is less than either the financial or long-term health benefits.”
But Clark said, “When some of these newer anti-obesity medications are able to go generic and be less expensive, which will happen eventually, I think things will change ... and in the future, there will be more options on the market as well.”
In the meantime, he advised that clinicians “try to have conversations with patients about these barriers, acknowledge that these barriers exist, and create a safe space to discuss those barriers. ... Let’s see where we are right now, and let’s come up with a plan.”
People With Obesity More Susceptible to Drug Rationing
Chen reported on a sample of 51,720 adults who participated in the 2020-2022 National Health Interview Survey who did not have diabetes and who used at least one prescription medication of any type. Of those, 80% were White, 9.7% were Hispanic, and 9.7% were Black, and 33.9% overall had obesity.
Cost-related prescription rationing, defined as any self-reported skipping, taking less, or delaying filling a prescription to save money, was reported by 8.3% of those with obesity vs 5.9% without. After adjustment, rationing among those with obesity was significantly associated with younger age (aged 18-44 years), female sex, lower incomes, lack of health insurance coverage, and CVD.
The adjusted estimated probability of cost-related prescription drug rationing was 7.4% for those with CVD vs 4.4% for those without. By race/ethnicity, the proportions reporting rationing were 7.7%, 9.8%, and 10.7% for White, Black, and Hispanics, respectively.
“Given that few insurance providers cover GLP-1 RAs for obesity, cost-related prescription drug rationing could be exacerbated if patients were prescribed GLP-1 RAs at their current price of more than $1000 a month,” Chen noted, adding that the high prices could worsen health disparities among Black and Hispanic individuals with obesity.
Medicaid Coverage Lacking for All Obesity Treatments
For their project, Gallagher and Zvenyach delved into a database that aggregates Medicaid manuals, fee schedules, statutes, regulations, and preferred drug lists for each US state, both for Medicaid fee for service and top Medicaid managed care plans, in order to determine 2024 levels of coverage for NC, IBT, OMs, and MBS for adults with obesity.
No state provides coverage for all those treatments without either limitations — such as body mass index (BMI) cutoffs, age, or “comorbidity regardless of body mass index (BMI)” for OM and MBS — or outright restrictions, such as “proof of failed attempts.” And only seven states provide coverage for the four modalities “with limitations”: California, Arizona, New Mexico, South Carolina, Delaware, Rhode Island, and Massachusetts.
Twenty-two states don’t cover NC, although just one state doesn’t cover IBT. Overall, 37 states don’t cover OMs, and other states ranged considerably in various restrictions and limitations for OMs and MBS. Only four states fully covered the surgery without limitations or restrictions.
“The vast majority of states have significant barriers and conditions of coverage for obesity care,” Gallagher said.
Zvenyach added, “Most of the applied exclusions, limitations, or restrictions do not align with evidence-based practice standards or guidelines.”
When Coverage Stops, Hopelessness and Anger Emerge
Haggerty and colleagues’ research involved semi-structured interviews of the 22 participants — all of them women — who had lost their obesity medication coverage due to their insurers’ decision. Four themes emerged:
- 1. Feelings of hope replaced by hopelessness upon loss of medication coverage: One person said, “I’m afraid for my mental health. It’s tough to be in a situation where you’re never right. And it doesn’t matter what you do; it’s not going to work, and then to have just a glimmer of hope, a little spark of hey, look, this might help. And for someone else to take that away from you for no reason. I don’t know what am I supposed to do.”
- 2. Anger regarding the perceived injustice of anti-obesity medication coverage termination: For example, “They can pay for heart attacks, they could pay for me to have a stroke, they could pay for me to have diabetes, but they won’t let me have this one medicine that could take all of that away. Makes no sense.”
- 3. Perceptions of past and present stigma within the healthcare system and insurance company: “I’m not trying for vanity. I’m way too old to be a Victoria’s Secret model. I’m not trying to do it to be cute and skinny and hot. I just want to make it through a day of work and not be exhausted.”
- 4. Generational influences on obesity treatment: “I’m married, and my husband, since I’ve started this medicine, he’s been eating better. He’s been eating what I eat, and he’s been losing weight as well.”
- Some participants said they planned to cope in different ways, including trying to obtain compounded versions of the drugs from “spas” or online pharmacies, as well as skipping doses, reducing doses, or sharing medications — in other words, rationing.
Asked by this news organization what clinicians should keep in mind, Haggerty said, “that there are big barriers and that we need to take care of the patients within this system that has their arm tied behind their back.
Chen was funded by a grant from the National Institute on Aging outside the submitted work, and she was funded as a Yale National Clinician Scholar. A coauthor received grants from the Food and Drug Administration, Johnson & Johnson, the National Institutes of Health, the Agency for Healthcare Research and Quality, and Arnold Ventures. Another coauthor reported receiving personal fees from UpToDate and the Centers for Medicare & Medicaid Services. Gallagher has received research funding from Altimmune, Amgen, Boehringer Ingelheim, Currax, Eli Lilly and Company, Found, Novo Nordisk, Pfizer, Structure Therapeutics, and WeightWatchers. Haggerty reported article publishing charge was provided by West Virginia Alliance for Creative Health Solutions. Zvenyach and Clark had no disclosures.
A version of this article first appeared on Medscape.com.
SAN ANTONIO — Weight loss treatments aren’t reaching many of the people who need them most because of coverage barriers, new data suggested.
Findings from three studies presented at The Obesity Society’s Obesity Week 2024 meeting illustrate different aspects of the problem.
One, presented by Alissa S. Chen, MD, MPH, a postdoctoral fellow at Yale University, New Haven, Connecticut, found that people with obesity, particularly those with cardiovascular disease (CVD) and those who are Black and Hispanic, have high rates of cost-related prescription drug rationing. Those findings were simultaneously published as a research letter in JAMA Network Open.
“The implications are that structural barriers impede access to medications for Black and Hispanic adults with obesity, which might worsen if there’s not expanding coverage for GLP-1 RAs [glucagon-like peptide 1 receptor agonists], and it’s possible that broader insurance coverage could ameliorate some of these issues,” Chen said.
She noted that patients don’t always volunteer that information. “In my clinical practice, I always start by saying something like, ‘I have a lot of patients who can’t afford their medications. In the last week, was there a time [you didn’t take your medications due to cost]?’ ”
State Medicaid programs vary widely in the degree to which they cover weight loss treatments. But not a single one covers all modalities — nutrition counseling (NC), intensive behavioral therapy (IBT), obesity medications (OMs), and metabolic and bariatric surgery (MBS) — without restrictions or limitations, and only seven states cover them all with restrictions, according to a dual presentation by Christine Gallagher, MPAff, associate director for research and policy with the STOP Obesity Alliance at George Washington University, Washington, DC, and Tracy Zvenyach, PhD, MS, RN, director of policy strategy and alliances at the Obesity Action Coalition, also in Washington, DC.
Detailed Medicaid coverage data for each state are posted on the STOP Obesity Alliance website. (As of now, Medicare doesn’t cover medications specifically for obesity at all.)
A third presentation, by Treah Haggerty, MD, of the Department of Family Medicine and director of the Pediatric Medical Weight Management program at West Virginia University, Morgantown, was of a qualitative descriptive study exploring the impact on 22 individuals enrolled in a medical weight loss management program whose state employee insurance carrier made a policy decision to stop covering all anti-OMs in March 2024. All had been prescribed GLP-1 agonists for weight loss, and the decision forced most to stop using them.
Those findings were published in the September 2024 issue of Obesity Pillars.
“Patients perceive the discontinuation of anti-obesity medication coverage as stigmatizing and unjust, leading to feelings of hopelessness and fear. With more insurance companies denying coverage for these costly medications, more information is needed to identify best ways to address the loss of coverage with patients. Clinical management of these patients should incorporate evidence-based obesity treatments while navigating insurance constraints,” Haggerty said.
Create a Safe Space to Discuss the Barriers
Asked to comment, Session Moderator John D. Clark, MD, PhD, chief population health officer at Sharp Rees-Stealy Medical Group, San Diego, California, told Medscape Medical News, “Health systems and payers are determining what can and can’t be covered, and at the end of the day, it frequently comes down to finances…I think the big challenge is really identifying patients who may have the greatest need. ... If we have limited resources, how and where should we be directing those resources? I would say the current system hasn’t really answered that question or identified patients for whom we would say that the cost truly is less than either the financial or long-term health benefits.”
But Clark said, “When some of these newer anti-obesity medications are able to go generic and be less expensive, which will happen eventually, I think things will change ... and in the future, there will be more options on the market as well.”
In the meantime, he advised that clinicians “try to have conversations with patients about these barriers, acknowledge that these barriers exist, and create a safe space to discuss those barriers. ... Let’s see where we are right now, and let’s come up with a plan.”
People With Obesity More Susceptible to Drug Rationing
Chen reported on a sample of 51,720 adults who participated in the 2020-2022 National Health Interview Survey who did not have diabetes and who used at least one prescription medication of any type. Of those, 80% were White, 9.7% were Hispanic, and 9.7% were Black, and 33.9% overall had obesity.
Cost-related prescription rationing, defined as any self-reported skipping, taking less, or delaying filling a prescription to save money, was reported by 8.3% of those with obesity vs 5.9% without. After adjustment, rationing among those with obesity was significantly associated with younger age (aged 18-44 years), female sex, lower incomes, lack of health insurance coverage, and CVD.
The adjusted estimated probability of cost-related prescription drug rationing was 7.4% for those with CVD vs 4.4% for those without. By race/ethnicity, the proportions reporting rationing were 7.7%, 9.8%, and 10.7% for White, Black, and Hispanics, respectively.
“Given that few insurance providers cover GLP-1 RAs for obesity, cost-related prescription drug rationing could be exacerbated if patients were prescribed GLP-1 RAs at their current price of more than $1000 a month,” Chen noted, adding that the high prices could worsen health disparities among Black and Hispanic individuals with obesity.
Medicaid Coverage Lacking for All Obesity Treatments
For their project, Gallagher and Zvenyach delved into a database that aggregates Medicaid manuals, fee schedules, statutes, regulations, and preferred drug lists for each US state, both for Medicaid fee for service and top Medicaid managed care plans, in order to determine 2024 levels of coverage for NC, IBT, OMs, and MBS for adults with obesity.
No state provides coverage for all those treatments without either limitations — such as body mass index (BMI) cutoffs, age, or “comorbidity regardless of body mass index (BMI)” for OM and MBS — or outright restrictions, such as “proof of failed attempts.” And only seven states provide coverage for the four modalities “with limitations”: California, Arizona, New Mexico, South Carolina, Delaware, Rhode Island, and Massachusetts.
Twenty-two states don’t cover NC, although just one state doesn’t cover IBT. Overall, 37 states don’t cover OMs, and other states ranged considerably in various restrictions and limitations for OMs and MBS. Only four states fully covered the surgery without limitations or restrictions.
“The vast majority of states have significant barriers and conditions of coverage for obesity care,” Gallagher said.
Zvenyach added, “Most of the applied exclusions, limitations, or restrictions do not align with evidence-based practice standards or guidelines.”
When Coverage Stops, Hopelessness and Anger Emerge
Haggerty and colleagues’ research involved semi-structured interviews of the 22 participants — all of them women — who had lost their obesity medication coverage due to their insurers’ decision. Four themes emerged:
- 1. Feelings of hope replaced by hopelessness upon loss of medication coverage: One person said, “I’m afraid for my mental health. It’s tough to be in a situation where you’re never right. And it doesn’t matter what you do; it’s not going to work, and then to have just a glimmer of hope, a little spark of hey, look, this might help. And for someone else to take that away from you for no reason. I don’t know what am I supposed to do.”
- 2. Anger regarding the perceived injustice of anti-obesity medication coverage termination: For example, “They can pay for heart attacks, they could pay for me to have a stroke, they could pay for me to have diabetes, but they won’t let me have this one medicine that could take all of that away. Makes no sense.”
- 3. Perceptions of past and present stigma within the healthcare system and insurance company: “I’m not trying for vanity. I’m way too old to be a Victoria’s Secret model. I’m not trying to do it to be cute and skinny and hot. I just want to make it through a day of work and not be exhausted.”
- 4. Generational influences on obesity treatment: “I’m married, and my husband, since I’ve started this medicine, he’s been eating better. He’s been eating what I eat, and he’s been losing weight as well.”
- Some participants said they planned to cope in different ways, including trying to obtain compounded versions of the drugs from “spas” or online pharmacies, as well as skipping doses, reducing doses, or sharing medications — in other words, rationing.
Asked by this news organization what clinicians should keep in mind, Haggerty said, “that there are big barriers and that we need to take care of the patients within this system that has their arm tied behind their back.
Chen was funded by a grant from the National Institute on Aging outside the submitted work, and she was funded as a Yale National Clinician Scholar. A coauthor received grants from the Food and Drug Administration, Johnson & Johnson, the National Institutes of Health, the Agency for Healthcare Research and Quality, and Arnold Ventures. Another coauthor reported receiving personal fees from UpToDate and the Centers for Medicare & Medicaid Services. Gallagher has received research funding from Altimmune, Amgen, Boehringer Ingelheim, Currax, Eli Lilly and Company, Found, Novo Nordisk, Pfizer, Structure Therapeutics, and WeightWatchers. Haggerty reported article publishing charge was provided by West Virginia Alliance for Creative Health Solutions. Zvenyach and Clark had no disclosures.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2024
Do Patients on Anti-Obesity Drugs Decrease Alcohol Use?
SAN ANTONIO —
The findings, from surveys of more than 14,000 participants in WeightWatchers’ telehealth weight management program, were presented on November 6 at the Obesity Society’s Obesity Week 2024 meeting by the company’s Chief Nutrition Officer, Michelle I. Cardel, PhD, RD, based in Gainesville, Florida.
Similar reductions in alcohol consumption were seen in people taking different classes of AOMs, suggesting “an additional mechanism by which AOMs reduce energy intake, and also signal a potential role for these medications to reduce alcohol use,” Cardel said, adding “Clinicians treating individuals for obesity may consider anti-obesity medications particularly among those who report higher alcohol intake.”
Asked to comment, session moderator and obesity researcher Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said, “I think there are some overlapping pathways there, possibly a reward system or something like that in the brain. I don’t think we know exactly what the end result will be as a potential use of the medications. But there’s a signal that needs to be investigated more.”
Cardel noted that there was one previous large cohort study finding that semaglutide was associated with a lower risk for alcohol use disorder, and another study that analyzed social media threads of people saying they’d quit drinking after starting a GLP-1 drug. But this new study is the first to examine the relationship with different classes of AOMs and to quantify the amount of alcohol consumed.
About Half Reported Reduced Alcohol Consumption, Regardless the AOM Class
The study included 14,053 WeightWatchers’ telehealth program participants who initiated an AOM between January 2022 and August 2023 and refilled the same AOM between October and November 2023. Those who had previously used AOMs before coming to the program or who had undergone bariatric surgery were excluded.
Participants had a mean age of 43 years, were 86% women, were 60% White, and had a mean body mass index of 36. They were surveyed about their weekly alcohol use prior to AOM initiation and again at the time of AOM refill.
At baseline, they were divided into categories of 0 (no alcohol use; n = 6562), category 1 (one to three drinks for women and one to six for men; n = 5948), category 2 (4-6 for women and 7-14 for men; n = 1216), and category 3 (≥ 7 for women and ≥ 15 for men; n = 327).
At the second survey, 24% reported decreased drinking after starting an AOM, 71% reported no change, and 4% reported increased drinking (P < .0001). But when just the 7491 individuals who reported any alcohol use at baseline were included, 45% reported decreased drinking after starting an AOM, 52% reported no change, and only 2% reported increased drinking.
The decrease in drinking with AOM use rose with greater alcohol use at baseline, from 37% for category 1, 76% for category 2, and 91% for category 3. The proportions reporting increased drinking were just 3%, 1%, and 0%, respectively. The adjusted odds ratios (ORs) for decreasing drinking were 5.97 for category 2 (P < .0001) and 19.18 for category 3 (P < .0001) vs category 1.
The proportions reporting reduced drinking were similar across AOM classes: 51% for metformin, 46% for bupropion/naltrexone, 46% for first-generation GLP-1s (Saxenda, Trulicity, and Victoza), and 45% for the second-generation GLP-1 drugs (Mounjaro, Ozempic, Rybelsus, Wegovy, and Zepbound). All were statistically significant at P < .0001.
The highest proportion reporting increased drinking was 4% for bupropion/naltrexone. Compared with women, men were significantly more likely to report decreased drinking with AOM use (adjusted OR, 0.74; P < .001), but there were no differences by race/ethnicity or age.
Compared with those who had overweight, those in obesity classes I, II, and III were all more likely to decrease drinking with AOM use, with adjusted ORs of 1.26 (P = .0045), 1.49 (P < .001), and 1.63 (P < .001), respectively.
Mechanisms Appear Both Biological and Behavioral
During the discussion, Cardel said that qualitative assessments with participants suggest that there are at least two mechanisms behind this phenomenon: One biological and the other intentional.
“What we hear from them is twofold, one, particularly amongst those folks on GLP-1 medications, we’re hearing that physiologically, they feel different with the medications, that their cravings for alcohol are decreased, and that when they do choose to drink that there’s often a very much a negative reinforcement ... I’ve had a patient tell me, ‘I used to be able to have two or three margaritas, and maybe I didn’t feel like the best I’d ever felt in the morning, but I was okay. And now if I have two or three drinks, I will be throwing up for 5 hours, and it’s the worst hangover I’ve ever had in my life.’ And so it very much creates that negative reinforcement loop.”
But at the same time, “folks who are coming to us and seeking these medications are very much on a on a health-based journey. That’s what they tell us. The majority of our patients are there to improve their health. We rarely hear about the vanity or aesthetic part of it. So perhaps it’s that, in terms of trying to improve their health, they’re also trying to reduce their alcohol consumption, either just for their overall health or also as a means of trying to decrease their overall calorie consumption.”
In future research, Cardel said, “we want to examine whether the anti-obesity medications are more successful at reducing alcohol use compared to non-pharmacological weight management interventions, as we know that people often reduce their alcohol consumption on a weight management journey as a means of prioritizing their calories for food and decreasing the calories from alcohol.”
Cardel and all the study coauthors were employees and shareholders at WeightWatchers at the time the research was conducted. Skelton is editor in chief of the journal Childhood Obesity.
A version of this article appeared on Medscape.com.
SAN ANTONIO —
The findings, from surveys of more than 14,000 participants in WeightWatchers’ telehealth weight management program, were presented on November 6 at the Obesity Society’s Obesity Week 2024 meeting by the company’s Chief Nutrition Officer, Michelle I. Cardel, PhD, RD, based in Gainesville, Florida.
Similar reductions in alcohol consumption were seen in people taking different classes of AOMs, suggesting “an additional mechanism by which AOMs reduce energy intake, and also signal a potential role for these medications to reduce alcohol use,” Cardel said, adding “Clinicians treating individuals for obesity may consider anti-obesity medications particularly among those who report higher alcohol intake.”
Asked to comment, session moderator and obesity researcher Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said, “I think there are some overlapping pathways there, possibly a reward system or something like that in the brain. I don’t think we know exactly what the end result will be as a potential use of the medications. But there’s a signal that needs to be investigated more.”
Cardel noted that there was one previous large cohort study finding that semaglutide was associated with a lower risk for alcohol use disorder, and another study that analyzed social media threads of people saying they’d quit drinking after starting a GLP-1 drug. But this new study is the first to examine the relationship with different classes of AOMs and to quantify the amount of alcohol consumed.
About Half Reported Reduced Alcohol Consumption, Regardless the AOM Class
The study included 14,053 WeightWatchers’ telehealth program participants who initiated an AOM between January 2022 and August 2023 and refilled the same AOM between October and November 2023. Those who had previously used AOMs before coming to the program or who had undergone bariatric surgery were excluded.
Participants had a mean age of 43 years, were 86% women, were 60% White, and had a mean body mass index of 36. They were surveyed about their weekly alcohol use prior to AOM initiation and again at the time of AOM refill.
At baseline, they were divided into categories of 0 (no alcohol use; n = 6562), category 1 (one to three drinks for women and one to six for men; n = 5948), category 2 (4-6 for women and 7-14 for men; n = 1216), and category 3 (≥ 7 for women and ≥ 15 for men; n = 327).
At the second survey, 24% reported decreased drinking after starting an AOM, 71% reported no change, and 4% reported increased drinking (P < .0001). But when just the 7491 individuals who reported any alcohol use at baseline were included, 45% reported decreased drinking after starting an AOM, 52% reported no change, and only 2% reported increased drinking.
The decrease in drinking with AOM use rose with greater alcohol use at baseline, from 37% for category 1, 76% for category 2, and 91% for category 3. The proportions reporting increased drinking were just 3%, 1%, and 0%, respectively. The adjusted odds ratios (ORs) for decreasing drinking were 5.97 for category 2 (P < .0001) and 19.18 for category 3 (P < .0001) vs category 1.
The proportions reporting reduced drinking were similar across AOM classes: 51% for metformin, 46% for bupropion/naltrexone, 46% for first-generation GLP-1s (Saxenda, Trulicity, and Victoza), and 45% for the second-generation GLP-1 drugs (Mounjaro, Ozempic, Rybelsus, Wegovy, and Zepbound). All were statistically significant at P < .0001.
The highest proportion reporting increased drinking was 4% for bupropion/naltrexone. Compared with women, men were significantly more likely to report decreased drinking with AOM use (adjusted OR, 0.74; P < .001), but there were no differences by race/ethnicity or age.
Compared with those who had overweight, those in obesity classes I, II, and III were all more likely to decrease drinking with AOM use, with adjusted ORs of 1.26 (P = .0045), 1.49 (P < .001), and 1.63 (P < .001), respectively.
Mechanisms Appear Both Biological and Behavioral
During the discussion, Cardel said that qualitative assessments with participants suggest that there are at least two mechanisms behind this phenomenon: One biological and the other intentional.
“What we hear from them is twofold, one, particularly amongst those folks on GLP-1 medications, we’re hearing that physiologically, they feel different with the medications, that their cravings for alcohol are decreased, and that when they do choose to drink that there’s often a very much a negative reinforcement ... I’ve had a patient tell me, ‘I used to be able to have two or three margaritas, and maybe I didn’t feel like the best I’d ever felt in the morning, but I was okay. And now if I have two or three drinks, I will be throwing up for 5 hours, and it’s the worst hangover I’ve ever had in my life.’ And so it very much creates that negative reinforcement loop.”
But at the same time, “folks who are coming to us and seeking these medications are very much on a on a health-based journey. That’s what they tell us. The majority of our patients are there to improve their health. We rarely hear about the vanity or aesthetic part of it. So perhaps it’s that, in terms of trying to improve their health, they’re also trying to reduce their alcohol consumption, either just for their overall health or also as a means of trying to decrease their overall calorie consumption.”
In future research, Cardel said, “we want to examine whether the anti-obesity medications are more successful at reducing alcohol use compared to non-pharmacological weight management interventions, as we know that people often reduce their alcohol consumption on a weight management journey as a means of prioritizing their calories for food and decreasing the calories from alcohol.”
Cardel and all the study coauthors were employees and shareholders at WeightWatchers at the time the research was conducted. Skelton is editor in chief of the journal Childhood Obesity.
A version of this article appeared on Medscape.com.
SAN ANTONIO —
The findings, from surveys of more than 14,000 participants in WeightWatchers’ telehealth weight management program, were presented on November 6 at the Obesity Society’s Obesity Week 2024 meeting by the company’s Chief Nutrition Officer, Michelle I. Cardel, PhD, RD, based in Gainesville, Florida.
Similar reductions in alcohol consumption were seen in people taking different classes of AOMs, suggesting “an additional mechanism by which AOMs reduce energy intake, and also signal a potential role for these medications to reduce alcohol use,” Cardel said, adding “Clinicians treating individuals for obesity may consider anti-obesity medications particularly among those who report higher alcohol intake.”
Asked to comment, session moderator and obesity researcher Joseph A. Skelton, MD, professor of pediatrics at Wake Forest University School of Medicine, Winston-Salem, North Carolina, said, “I think there are some overlapping pathways there, possibly a reward system or something like that in the brain. I don’t think we know exactly what the end result will be as a potential use of the medications. But there’s a signal that needs to be investigated more.”
Cardel noted that there was one previous large cohort study finding that semaglutide was associated with a lower risk for alcohol use disorder, and another study that analyzed social media threads of people saying they’d quit drinking after starting a GLP-1 drug. But this new study is the first to examine the relationship with different classes of AOMs and to quantify the amount of alcohol consumed.
About Half Reported Reduced Alcohol Consumption, Regardless the AOM Class
The study included 14,053 WeightWatchers’ telehealth program participants who initiated an AOM between January 2022 and August 2023 and refilled the same AOM between October and November 2023. Those who had previously used AOMs before coming to the program or who had undergone bariatric surgery were excluded.
Participants had a mean age of 43 years, were 86% women, were 60% White, and had a mean body mass index of 36. They were surveyed about their weekly alcohol use prior to AOM initiation and again at the time of AOM refill.
At baseline, they were divided into categories of 0 (no alcohol use; n = 6562), category 1 (one to three drinks for women and one to six for men; n = 5948), category 2 (4-6 for women and 7-14 for men; n = 1216), and category 3 (≥ 7 for women and ≥ 15 for men; n = 327).
At the second survey, 24% reported decreased drinking after starting an AOM, 71% reported no change, and 4% reported increased drinking (P < .0001). But when just the 7491 individuals who reported any alcohol use at baseline were included, 45% reported decreased drinking after starting an AOM, 52% reported no change, and only 2% reported increased drinking.
The decrease in drinking with AOM use rose with greater alcohol use at baseline, from 37% for category 1, 76% for category 2, and 91% for category 3. The proportions reporting increased drinking were just 3%, 1%, and 0%, respectively. The adjusted odds ratios (ORs) for decreasing drinking were 5.97 for category 2 (P < .0001) and 19.18 for category 3 (P < .0001) vs category 1.
The proportions reporting reduced drinking were similar across AOM classes: 51% for metformin, 46% for bupropion/naltrexone, 46% for first-generation GLP-1s (Saxenda, Trulicity, and Victoza), and 45% for the second-generation GLP-1 drugs (Mounjaro, Ozempic, Rybelsus, Wegovy, and Zepbound). All were statistically significant at P < .0001.
The highest proportion reporting increased drinking was 4% for bupropion/naltrexone. Compared with women, men were significantly more likely to report decreased drinking with AOM use (adjusted OR, 0.74; P < .001), but there were no differences by race/ethnicity or age.
Compared with those who had overweight, those in obesity classes I, II, and III were all more likely to decrease drinking with AOM use, with adjusted ORs of 1.26 (P = .0045), 1.49 (P < .001), and 1.63 (P < .001), respectively.
Mechanisms Appear Both Biological and Behavioral
During the discussion, Cardel said that qualitative assessments with participants suggest that there are at least two mechanisms behind this phenomenon: One biological and the other intentional.
“What we hear from them is twofold, one, particularly amongst those folks on GLP-1 medications, we’re hearing that physiologically, they feel different with the medications, that their cravings for alcohol are decreased, and that when they do choose to drink that there’s often a very much a negative reinforcement ... I’ve had a patient tell me, ‘I used to be able to have two or three margaritas, and maybe I didn’t feel like the best I’d ever felt in the morning, but I was okay. And now if I have two or three drinks, I will be throwing up for 5 hours, and it’s the worst hangover I’ve ever had in my life.’ And so it very much creates that negative reinforcement loop.”
But at the same time, “folks who are coming to us and seeking these medications are very much on a on a health-based journey. That’s what they tell us. The majority of our patients are there to improve their health. We rarely hear about the vanity or aesthetic part of it. So perhaps it’s that, in terms of trying to improve their health, they’re also trying to reduce their alcohol consumption, either just for their overall health or also as a means of trying to decrease their overall calorie consumption.”
In future research, Cardel said, “we want to examine whether the anti-obesity medications are more successful at reducing alcohol use compared to non-pharmacological weight management interventions, as we know that people often reduce their alcohol consumption on a weight management journey as a means of prioritizing their calories for food and decreasing the calories from alcohol.”
Cardel and all the study coauthors were employees and shareholders at WeightWatchers at the time the research was conducted. Skelton is editor in chief of the journal Childhood Obesity.
A version of this article appeared on Medscape.com.
FROM OBESITY WEEK 2024
Knowledge Gaps About Obesity Medicine Seen in Primary Care
SAN ANTONIO — Despite the prevalence of obesity in primary care, there appear to be major knowledge gaps among providers regarding obesity management, new research suggests.
Anonymous surveys of 96 primary care providers at a Boston, Massachusetts, safety-net hospital revealed that participants had limited understanding of criteria for prescribing antiobesity medications (AOM), and expressed discomfort in prescribing AOMs because of knowledge concerns, especially for non–glucagon-like peptide 1 (GLP-1) receptor agonists. One third reported that they didn’t prescribe AOMs, and rates of referral for bariatric surgery were also low.
The findings were presented at the Obesity Society’s annual Obesity Week meeting by Alejandro Campos, MD, a third-year resident in the section of internal medicine, Boston Medical Center, and the Department of Medicine, Boston University.
“I think it comes down to education. ... Not only training primary care physicians or residents about criteria and pathophysiology, but also stigma. Perceptions need to be addressed from the start of training in the healthcare field,” Campos told this news organization in an interview.
During his presentation, Campos noted this is the first such study in the setting of a safety-net hospital, which cares for lower-income people who experience disproportionate rates of obesity. But, “these findings are similar to ones observed from non–safety-net settings, which can indicate some potential transferability.”
Asked to comment, session moderator John D. Clark, MD, PhD, Chief Population Health Officer at Sharp Rees-Stealy Medical Group, San Diego, California, told this news organization that the findings didn’t surprise him. “I’d say that medical education around obesity has definitely improved, and training is improving but it’s not uniform. The treatment of obesity as a disease, especially with some of the newer medical treatments, is not standard of care and practiced widely.”
The study involved a standard-model Knowledge, Attitudes, and Practices questionnaire, distributed electronically for anonymous responses among both trained and in-training primary care providers. It contained a total of 43 items, 7 of them demographic, 11 on knowledge, 9 regarding attitudes, and 16 asking about practices.
The hospital is the largest safety-net hospital in New England, with a patient population that includes 58% enrolled in Medicaid, 32% Black/African American individuals, 24% identifying as Hispanic/Latino individuals, and 37% living below the poverty line.
The 96 responding providers (from a total 350 invited) all worked in either family medicine or internal medicine. The trained providers included both attending MDs and nurse practitioners, while those in-training were residents in one of those two specialties. Two thirds were women. The majority were aged 20-30 years (49.45%) or 31-40 years (27.47%).
Overall, 73.63% reported having received some type of obesity training. Just over half (52.08%) reported receiving that training during medical or nursing school, while 43.75% reported receiving it during residency.
When asked to choose from a list of conditions to pick which are considered weight-related comorbidities, between 80% and 90% choose type 2 diabetes, obstructive sleep apnea (OSA), hypertension, hyperlipidemia, nonalcoholic fatty liver disease, and coronary artery disease. Fewer, but still a majority, also listed osteoarthritis and gastroesophageal reflux disease. However, respondents were less likely to cite cancer, mood disorders, or chronic kidney disease as being related to obesity.
Asked to list benefits of a 10% body weight loss, most recognized reductions in OSA, glycemia, cardiovascular disease risk, osteoarthritis, and hepatic steatosis. But, only about half knew weight loss could also improve urinary incontinence.
Only 25% could correctly name both indications for AOMs. Just 27.1% knew that one was a body mass index (BMI) ≥ 27 with comorbidities, while 46.9% knew BMI ≥ 30 without comorbidities was an AOM indication. Only 9.4% were correct on both of those indications for bariatric surgery.
“Reassuringly,” Campos said, the majority either “disagreed” or “strongly disagreed” that “lack of will power” contributes to obesity. However, more than 20% agreed that “lack of exercise or physical activity” contributed.
Overall, 73% of the trained providers and 59% of those in training reported that they prescribe AOMs. Asked about their comfort level in prescribing specific types of AOMs, many more endorsed semaglutide and liraglutide than older medications such as bupropion/naltrexone and phentermine/topiramate.
Asked about factors that influence their comfort with prescribing AOMs, the top five factors selected, in order, were side-effect knowledge, insurance coverage, safety issues, and dosing knowledge. Fewer respondents endorsed “patient’s ideas, concerns, and expectations,” cost, or efficacy.
Referrals to nutrition services were endorsed more often than to obesity medicine specialists or bariatric surgery.
Asked about barriers to obesity treatment in their practices, “time constraints” was the most frequently endorsed, followed by “lack of training or knowledge,” “patient adherence and motivation,” and “limited resources.”
“What are the future directives? We feel we have the need to provide ongoing obesity management, education and assistance to primary care providers, including support for securing coverage for treatments,” Campos said.
He added that Boston Medical Center is now developing and implementing an embedded weight management program within primary care “to assist the front line of obesity care.”
Asked by this news organization whether he believes the rise of GLP-1 drugs will make a difference, Campos said “Definitely, I think with that momentum obesity medicine as a whole will gain more attention and hopefully more implementation in the curricula for medical and nursing schools, because in the end it requires a multidisciplinary approach.”
Campos and Clark had no disclosures.
A version of this article first appeared on Medscape.com.
SAN ANTONIO — Despite the prevalence of obesity in primary care, there appear to be major knowledge gaps among providers regarding obesity management, new research suggests.
Anonymous surveys of 96 primary care providers at a Boston, Massachusetts, safety-net hospital revealed that participants had limited understanding of criteria for prescribing antiobesity medications (AOM), and expressed discomfort in prescribing AOMs because of knowledge concerns, especially for non–glucagon-like peptide 1 (GLP-1) receptor agonists. One third reported that they didn’t prescribe AOMs, and rates of referral for bariatric surgery were also low.
The findings were presented at the Obesity Society’s annual Obesity Week meeting by Alejandro Campos, MD, a third-year resident in the section of internal medicine, Boston Medical Center, and the Department of Medicine, Boston University.
“I think it comes down to education. ... Not only training primary care physicians or residents about criteria and pathophysiology, but also stigma. Perceptions need to be addressed from the start of training in the healthcare field,” Campos told this news organization in an interview.
During his presentation, Campos noted this is the first such study in the setting of a safety-net hospital, which cares for lower-income people who experience disproportionate rates of obesity. But, “these findings are similar to ones observed from non–safety-net settings, which can indicate some potential transferability.”
Asked to comment, session moderator John D. Clark, MD, PhD, Chief Population Health Officer at Sharp Rees-Stealy Medical Group, San Diego, California, told this news organization that the findings didn’t surprise him. “I’d say that medical education around obesity has definitely improved, and training is improving but it’s not uniform. The treatment of obesity as a disease, especially with some of the newer medical treatments, is not standard of care and practiced widely.”
The study involved a standard-model Knowledge, Attitudes, and Practices questionnaire, distributed electronically for anonymous responses among both trained and in-training primary care providers. It contained a total of 43 items, 7 of them demographic, 11 on knowledge, 9 regarding attitudes, and 16 asking about practices.
The hospital is the largest safety-net hospital in New England, with a patient population that includes 58% enrolled in Medicaid, 32% Black/African American individuals, 24% identifying as Hispanic/Latino individuals, and 37% living below the poverty line.
The 96 responding providers (from a total 350 invited) all worked in either family medicine or internal medicine. The trained providers included both attending MDs and nurse practitioners, while those in-training were residents in one of those two specialties. Two thirds were women. The majority were aged 20-30 years (49.45%) or 31-40 years (27.47%).
Overall, 73.63% reported having received some type of obesity training. Just over half (52.08%) reported receiving that training during medical or nursing school, while 43.75% reported receiving it during residency.
When asked to choose from a list of conditions to pick which are considered weight-related comorbidities, between 80% and 90% choose type 2 diabetes, obstructive sleep apnea (OSA), hypertension, hyperlipidemia, nonalcoholic fatty liver disease, and coronary artery disease. Fewer, but still a majority, also listed osteoarthritis and gastroesophageal reflux disease. However, respondents were less likely to cite cancer, mood disorders, or chronic kidney disease as being related to obesity.
Asked to list benefits of a 10% body weight loss, most recognized reductions in OSA, glycemia, cardiovascular disease risk, osteoarthritis, and hepatic steatosis. But, only about half knew weight loss could also improve urinary incontinence.
Only 25% could correctly name both indications for AOMs. Just 27.1% knew that one was a body mass index (BMI) ≥ 27 with comorbidities, while 46.9% knew BMI ≥ 30 without comorbidities was an AOM indication. Only 9.4% were correct on both of those indications for bariatric surgery.
“Reassuringly,” Campos said, the majority either “disagreed” or “strongly disagreed” that “lack of will power” contributes to obesity. However, more than 20% agreed that “lack of exercise or physical activity” contributed.
Overall, 73% of the trained providers and 59% of those in training reported that they prescribe AOMs. Asked about their comfort level in prescribing specific types of AOMs, many more endorsed semaglutide and liraglutide than older medications such as bupropion/naltrexone and phentermine/topiramate.
Asked about factors that influence their comfort with prescribing AOMs, the top five factors selected, in order, were side-effect knowledge, insurance coverage, safety issues, and dosing knowledge. Fewer respondents endorsed “patient’s ideas, concerns, and expectations,” cost, or efficacy.
Referrals to nutrition services were endorsed more often than to obesity medicine specialists or bariatric surgery.
Asked about barriers to obesity treatment in their practices, “time constraints” was the most frequently endorsed, followed by “lack of training or knowledge,” “patient adherence and motivation,” and “limited resources.”
“What are the future directives? We feel we have the need to provide ongoing obesity management, education and assistance to primary care providers, including support for securing coverage for treatments,” Campos said.
He added that Boston Medical Center is now developing and implementing an embedded weight management program within primary care “to assist the front line of obesity care.”
Asked by this news organization whether he believes the rise of GLP-1 drugs will make a difference, Campos said “Definitely, I think with that momentum obesity medicine as a whole will gain more attention and hopefully more implementation in the curricula for medical and nursing schools, because in the end it requires a multidisciplinary approach.”
Campos and Clark had no disclosures.
A version of this article first appeared on Medscape.com.
SAN ANTONIO — Despite the prevalence of obesity in primary care, there appear to be major knowledge gaps among providers regarding obesity management, new research suggests.
Anonymous surveys of 96 primary care providers at a Boston, Massachusetts, safety-net hospital revealed that participants had limited understanding of criteria for prescribing antiobesity medications (AOM), and expressed discomfort in prescribing AOMs because of knowledge concerns, especially for non–glucagon-like peptide 1 (GLP-1) receptor agonists. One third reported that they didn’t prescribe AOMs, and rates of referral for bariatric surgery were also low.
The findings were presented at the Obesity Society’s annual Obesity Week meeting by Alejandro Campos, MD, a third-year resident in the section of internal medicine, Boston Medical Center, and the Department of Medicine, Boston University.
“I think it comes down to education. ... Not only training primary care physicians or residents about criteria and pathophysiology, but also stigma. Perceptions need to be addressed from the start of training in the healthcare field,” Campos told this news organization in an interview.
During his presentation, Campos noted this is the first such study in the setting of a safety-net hospital, which cares for lower-income people who experience disproportionate rates of obesity. But, “these findings are similar to ones observed from non–safety-net settings, which can indicate some potential transferability.”
Asked to comment, session moderator John D. Clark, MD, PhD, Chief Population Health Officer at Sharp Rees-Stealy Medical Group, San Diego, California, told this news organization that the findings didn’t surprise him. “I’d say that medical education around obesity has definitely improved, and training is improving but it’s not uniform. The treatment of obesity as a disease, especially with some of the newer medical treatments, is not standard of care and practiced widely.”
The study involved a standard-model Knowledge, Attitudes, and Practices questionnaire, distributed electronically for anonymous responses among both trained and in-training primary care providers. It contained a total of 43 items, 7 of them demographic, 11 on knowledge, 9 regarding attitudes, and 16 asking about practices.
The hospital is the largest safety-net hospital in New England, with a patient population that includes 58% enrolled in Medicaid, 32% Black/African American individuals, 24% identifying as Hispanic/Latino individuals, and 37% living below the poverty line.
The 96 responding providers (from a total 350 invited) all worked in either family medicine or internal medicine. The trained providers included both attending MDs and nurse practitioners, while those in-training were residents in one of those two specialties. Two thirds were women. The majority were aged 20-30 years (49.45%) or 31-40 years (27.47%).
Overall, 73.63% reported having received some type of obesity training. Just over half (52.08%) reported receiving that training during medical or nursing school, while 43.75% reported receiving it during residency.
When asked to choose from a list of conditions to pick which are considered weight-related comorbidities, between 80% and 90% choose type 2 diabetes, obstructive sleep apnea (OSA), hypertension, hyperlipidemia, nonalcoholic fatty liver disease, and coronary artery disease. Fewer, but still a majority, also listed osteoarthritis and gastroesophageal reflux disease. However, respondents were less likely to cite cancer, mood disorders, or chronic kidney disease as being related to obesity.
Asked to list benefits of a 10% body weight loss, most recognized reductions in OSA, glycemia, cardiovascular disease risk, osteoarthritis, and hepatic steatosis. But, only about half knew weight loss could also improve urinary incontinence.
Only 25% could correctly name both indications for AOMs. Just 27.1% knew that one was a body mass index (BMI) ≥ 27 with comorbidities, while 46.9% knew BMI ≥ 30 without comorbidities was an AOM indication. Only 9.4% were correct on both of those indications for bariatric surgery.
“Reassuringly,” Campos said, the majority either “disagreed” or “strongly disagreed” that “lack of will power” contributes to obesity. However, more than 20% agreed that “lack of exercise or physical activity” contributed.
Overall, 73% of the trained providers and 59% of those in training reported that they prescribe AOMs. Asked about their comfort level in prescribing specific types of AOMs, many more endorsed semaglutide and liraglutide than older medications such as bupropion/naltrexone and phentermine/topiramate.
Asked about factors that influence their comfort with prescribing AOMs, the top five factors selected, in order, were side-effect knowledge, insurance coverage, safety issues, and dosing knowledge. Fewer respondents endorsed “patient’s ideas, concerns, and expectations,” cost, or efficacy.
Referrals to nutrition services were endorsed more often than to obesity medicine specialists or bariatric surgery.
Asked about barriers to obesity treatment in their practices, “time constraints” was the most frequently endorsed, followed by “lack of training or knowledge,” “patient adherence and motivation,” and “limited resources.”
“What are the future directives? We feel we have the need to provide ongoing obesity management, education and assistance to primary care providers, including support for securing coverage for treatments,” Campos said.
He added that Boston Medical Center is now developing and implementing an embedded weight management program within primary care “to assist the front line of obesity care.”
Asked by this news organization whether he believes the rise of GLP-1 drugs will make a difference, Campos said “Definitely, I think with that momentum obesity medicine as a whole will gain more attention and hopefully more implementation in the curricula for medical and nursing schools, because in the end it requires a multidisciplinary approach.”
Campos and Clark had no disclosures.
A version of this article first appeared on Medscape.com.
FROM OBESITY WEEK 2024
Coming Soon: A New Disease Definition, ‘Clinical Obesity’
SAN ANTONIO, TEXAS —
The authors of the new framework are a Lancet Commission of 56 of the world’s leading obesity experts, including academic clinicians, scientists, public health experts, patient representatives, and officers from the World Health Organization. Following peer review, it will be launched via livestream and published in Lancet Diabetes & Endocrinology in mid-January 2025, with formal endorsement from more than 75 medical societies and other relevant stakeholder organizations.
On November 4, 2024, at the Obesity Society’s Obesity Week meeting, the publication’s lead author, Francesco Rubino, MD, Chair of Bariatric and Metabolic Surgery at King’s College London in England, gave a preview. He began by noting that, despite the declaration of obesity as a chronic disease over a decade ago, the concept is still debated and not widely accepted by the public or even by all in the medical community.
“The idea of obesity as a disease remains highly controversial,” Rubino noted, adding that the current body mass index (BMI)–based definition contributes to this because it doesn’t distinguish between people whose excess adiposity place them at excess risk for disease but they’re currently healthy vs those who already have undergone bodily harm from that adiposity.
“Having a framework that distinguishes at an individual level when you are in a condition of risk and when you have a condition of disease is fundamentally important. You don’t want to blur the picture in either direction, because obviously the consequence would be quite significant. ... So, the commission focused exactly on that point,” he said.
The new paper will propose a two-part clinical approach: First, assess whether the patient has excess adiposity, with methods that will be outlined. Next, assess on an organ-by-organ basis for the presence of abnormalities related to excess adiposity, or “clinical obesity.” The document will also provide those specific criteria, Rubino said, noting that those details are under embargo until January.
However, he did say that “We are going to propose a pragmatic approach to say that BMI alone is not enough in the clinic. It’s okay as a screening tool, but when somebody potentially has obesity, then you have to add additional measures of adiposity that makes sure you decrease the level of risk… Once you have obesity, then you need to establish if it’s clinical or nonclinical.”
Asked to comment, session moderator John D. Clark, MD, PhD, Chief Population Health Officer at Sharp Rees-Stealy Medical Group, San Diego, California, said in an interview, “I think it’ll help explain and move medicine as a whole in a direction to a greater understanding of obesity actually being a disease, how to define it, and how to identify it. And will, I think, lead to a greater understanding of the underlying disease.”
And, Clark said, it should also help target individuals with preventive vs therapeutic approaches. “I would describe it as matching the right tool to the right patient. If a person has clinical obesity, they likely can and would benefit from either different or additional tools, as opposed to otherwise healthy obesity.”
Rubino said he hopes the new framework will prompt improvements in reimbursement and public policy. “Policymakers scratch their heads when they have limited resources and you need to prioritize things. Having an obesity definition that is blurry doesn’t allow you to have a fair, human, and meaningful prioritization. ... Now that we have drugs that cannot be given to 100% of people, how do you decide who gets them first? I hope this will make it easier for people to access treatment. At the moment, it is not only difficult, but it’s also unfair. It’s random. Somebody gets access, while somebody else who is very, very sick has no access. I don’t think that’s what we want.”
A version of this article appeared on Medscape.com.
SAN ANTONIO, TEXAS —
The authors of the new framework are a Lancet Commission of 56 of the world’s leading obesity experts, including academic clinicians, scientists, public health experts, patient representatives, and officers from the World Health Organization. Following peer review, it will be launched via livestream and published in Lancet Diabetes & Endocrinology in mid-January 2025, with formal endorsement from more than 75 medical societies and other relevant stakeholder organizations.
On November 4, 2024, at the Obesity Society’s Obesity Week meeting, the publication’s lead author, Francesco Rubino, MD, Chair of Bariatric and Metabolic Surgery at King’s College London in England, gave a preview. He began by noting that, despite the declaration of obesity as a chronic disease over a decade ago, the concept is still debated and not widely accepted by the public or even by all in the medical community.
“The idea of obesity as a disease remains highly controversial,” Rubino noted, adding that the current body mass index (BMI)–based definition contributes to this because it doesn’t distinguish between people whose excess adiposity place them at excess risk for disease but they’re currently healthy vs those who already have undergone bodily harm from that adiposity.
“Having a framework that distinguishes at an individual level when you are in a condition of risk and when you have a condition of disease is fundamentally important. You don’t want to blur the picture in either direction, because obviously the consequence would be quite significant. ... So, the commission focused exactly on that point,” he said.
The new paper will propose a two-part clinical approach: First, assess whether the patient has excess adiposity, with methods that will be outlined. Next, assess on an organ-by-organ basis for the presence of abnormalities related to excess adiposity, or “clinical obesity.” The document will also provide those specific criteria, Rubino said, noting that those details are under embargo until January.
However, he did say that “We are going to propose a pragmatic approach to say that BMI alone is not enough in the clinic. It’s okay as a screening tool, but when somebody potentially has obesity, then you have to add additional measures of adiposity that makes sure you decrease the level of risk… Once you have obesity, then you need to establish if it’s clinical or nonclinical.”
Asked to comment, session moderator John D. Clark, MD, PhD, Chief Population Health Officer at Sharp Rees-Stealy Medical Group, San Diego, California, said in an interview, “I think it’ll help explain and move medicine as a whole in a direction to a greater understanding of obesity actually being a disease, how to define it, and how to identify it. And will, I think, lead to a greater understanding of the underlying disease.”
And, Clark said, it should also help target individuals with preventive vs therapeutic approaches. “I would describe it as matching the right tool to the right patient. If a person has clinical obesity, they likely can and would benefit from either different or additional tools, as opposed to otherwise healthy obesity.”
Rubino said he hopes the new framework will prompt improvements in reimbursement and public policy. “Policymakers scratch their heads when they have limited resources and you need to prioritize things. Having an obesity definition that is blurry doesn’t allow you to have a fair, human, and meaningful prioritization. ... Now that we have drugs that cannot be given to 100% of people, how do you decide who gets them first? I hope this will make it easier for people to access treatment. At the moment, it is not only difficult, but it’s also unfair. It’s random. Somebody gets access, while somebody else who is very, very sick has no access. I don’t think that’s what we want.”
A version of this article appeared on Medscape.com.
SAN ANTONIO, TEXAS —
The authors of the new framework are a Lancet Commission of 56 of the world’s leading obesity experts, including academic clinicians, scientists, public health experts, patient representatives, and officers from the World Health Organization. Following peer review, it will be launched via livestream and published in Lancet Diabetes & Endocrinology in mid-January 2025, with formal endorsement from more than 75 medical societies and other relevant stakeholder organizations.
On November 4, 2024, at the Obesity Society’s Obesity Week meeting, the publication’s lead author, Francesco Rubino, MD, Chair of Bariatric and Metabolic Surgery at King’s College London in England, gave a preview. He began by noting that, despite the declaration of obesity as a chronic disease over a decade ago, the concept is still debated and not widely accepted by the public or even by all in the medical community.
“The idea of obesity as a disease remains highly controversial,” Rubino noted, adding that the current body mass index (BMI)–based definition contributes to this because it doesn’t distinguish between people whose excess adiposity place them at excess risk for disease but they’re currently healthy vs those who already have undergone bodily harm from that adiposity.
“Having a framework that distinguishes at an individual level when you are in a condition of risk and when you have a condition of disease is fundamentally important. You don’t want to blur the picture in either direction, because obviously the consequence would be quite significant. ... So, the commission focused exactly on that point,” he said.
The new paper will propose a two-part clinical approach: First, assess whether the patient has excess adiposity, with methods that will be outlined. Next, assess on an organ-by-organ basis for the presence of abnormalities related to excess adiposity, or “clinical obesity.” The document will also provide those specific criteria, Rubino said, noting that those details are under embargo until January.
However, he did say that “We are going to propose a pragmatic approach to say that BMI alone is not enough in the clinic. It’s okay as a screening tool, but when somebody potentially has obesity, then you have to add additional measures of adiposity that makes sure you decrease the level of risk… Once you have obesity, then you need to establish if it’s clinical or nonclinical.”
Asked to comment, session moderator John D. Clark, MD, PhD, Chief Population Health Officer at Sharp Rees-Stealy Medical Group, San Diego, California, said in an interview, “I think it’ll help explain and move medicine as a whole in a direction to a greater understanding of obesity actually being a disease, how to define it, and how to identify it. And will, I think, lead to a greater understanding of the underlying disease.”
And, Clark said, it should also help target individuals with preventive vs therapeutic approaches. “I would describe it as matching the right tool to the right patient. If a person has clinical obesity, they likely can and would benefit from either different or additional tools, as opposed to otherwise healthy obesity.”
Rubino said he hopes the new framework will prompt improvements in reimbursement and public policy. “Policymakers scratch their heads when they have limited resources and you need to prioritize things. Having an obesity definition that is blurry doesn’t allow you to have a fair, human, and meaningful prioritization. ... Now that we have drugs that cannot be given to 100% of people, how do you decide who gets them first? I hope this will make it easier for people to access treatment. At the moment, it is not only difficult, but it’s also unfair. It’s random. Somebody gets access, while somebody else who is very, very sick has no access. I don’t think that’s what we want.”
A version of this article appeared on Medscape.com.
FROM OBESITY WEEK