User login
Reduce the use of perioperative opioids with a multimodal pain management strategy
Opioid-related deaths are a major cause of mortality in the United States. The Centers for Disease Control and Prevention (CDC) reported 72,151 and 93,331 drug overdose deaths in 2019 and 2020, respectively, and drug overdose deaths have continued to increase in 2021.1 The majority of drug overdose deaths are due to opioids. There are many factors contributing to this rise, including an incredibly high rate of opioid prescriptions in this country.2 The CDC reported that in 3.6% of US counties, there are more opioid prescriptions filled each year than number of residents in the county.3 The consumption of opioids per person in the US is approximately four times greater than countries with excellent health outcomes, including Sweden, Netherlands, Norway, and the United Kingdom.4 Some US physicians have opioid prescribing practices that are inconsistent with good medical practice in other countries, prescribing powerful opioids and an excessive number of pills per opioid prescription.2 We must continue to evolve our clinical practices to reduce opioid use while continually improving patient outcomes.
Cesarean birth is one of the most common major surgical procedures performed in the United States. The National Center for Health Statistics reported that in 2020 there were approximately 1,150,000 US cesarean births.5 Following cesarean birth, patients who were previously naïve to opioid medications were reported to have a 0.33% to 2.2% probability of transitioning to the persistent use of opioid prescriptions.6-8 Predictors of persistent opioid use after cesarean birth included a history of tobacco use, back pain, migraine headaches, and antidepressant or benzodiazepine use.6 The use of cesarean birth pain management protocols that prioritize multimodal analgesia and opioid sparing is warranted.
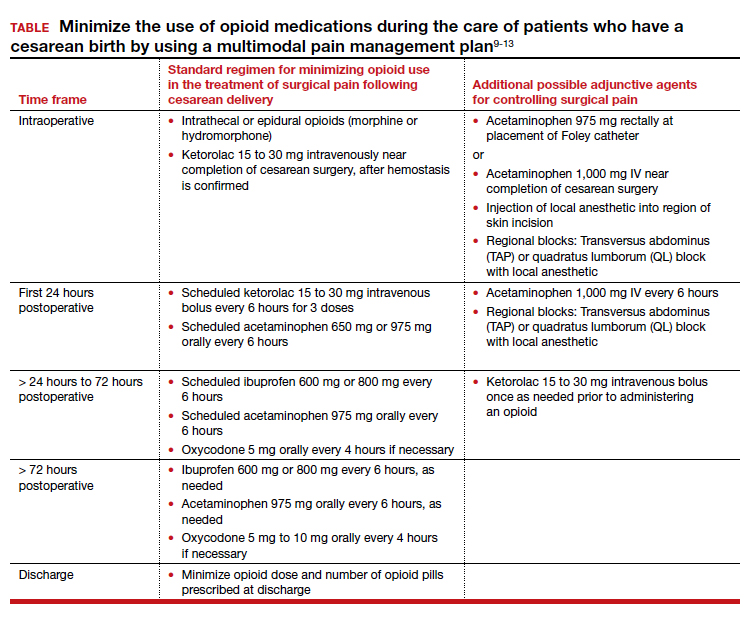
Multimodal pain management protocols for cesarean birth have been shown to reduce the use of opioid medications in the hospital and at discharge without a clinically significant increase in pain scores or a reduction in patient satisfaction (TABLE).9-13 For example, Holland and colleagues9 reported that the implementation of a multimodal pain management protocol reduced the percent of patients using oral opioids during hospitalization for cesarean birth from 68% to 45%, pre- and post-intervention, respectively. Mehraban and colleagues12 reported that the percent of patients using opioids during hospitalization for cesarean birth was reduced from 45% preintervention to 18% postintervention. In addition, these studies showed that multimodal pain management protocols for cesarean birth also reduced opioid prescribing at discharge. Holland and colleagues9 reported that the percent of patients provided an opioid prescription at discharge was reduced from 91% to 40%, pre- and post-intervention, respectively. Mehraban and colleagues12 reported that the percent of patients who took opioids after discharge was reduced from 24% preintervention to 9% postintervention. These studies were not randomized controlled clinical trials, but they do provide strong evidence that a focused intervention to reduce opioid medications in the management of pain after cesarean surgery can be successful without decreasing patient satisfaction or increasing reported pain scores. In these studies, it is likely that the influence, enthusiasm, and commitment of the study leaders to the change process contributed to the success of these opioid-sparing pain management programs.
Continue to: Key features of a multimodal analgesia intervention for cesarean surgery...
Key features of a multimodal analgesia intervention for cesarean surgery
Fundamental inclusions of multimodal analgesia for cesarean surgery include:
- exquisite attention to pain control during the surgical procedure by both the anesthesiologist and surgeon, with prioritization of spinal anesthesia that includes morphine and fentanyl
- regularly scheduled administration of intravenous ketorolac during the first 24 hours postcesarean
- regularly scheduled administration of both acetaminophen and ibuprofen, rather than “as needed” dosing
- using analgesics that work through different molecular pathways (ibuprofen and acetaminophen) (See Table.).
The significance of neuraxial and truncal nerve blockade for post-cesarean delivery pain control
Administration of a long-acting intrathecal opioid such as morphine lengthens time to first analgesic request after surgery and lowers 24-hour post‒cesarean delivery opioid requirement.14 If a patient requires general anesthesia and receives no spinal opioid, a transversus abdominis plane (TAP) block or quadratus lumborum (QL) block for postpartum pain control can lower associated postpartum opioid consumption. However, TAP or QL blocks confer no additional benefit to patients who receive spinal morphine,15 nor do they confer added benefit when combined with a multimodal pain management regimen postdelivery vs the multimodal regimen alone.16). TAP blocks administered to patients with severe breakthrough pain after spinal anesthesia help to lower opioid consumption.17 Further research is warranted on the use of TAP, QL, or other truncal blocks to spare opioid requirement after cesarean delivery in women with chronic pain, opioid use disorder, or those undergoing higher-complexity surgery such as cesarean hysterectomy for placenta accreta spectrum.
NSAIDs: Potential adverse effects
As we decrease the use of opioid medications and increase the use of nonsteroidal anti-inflammatory drugs (NSAIDs), we should reflect on the potential adverse effects of NSAID treatment in some patients. Specifically, the impact of ketorolac on hypertension, platelet function, and breastfeeding warrant consideration.
In the past, some studies reported that NSAID treatment is associated with a modest increase in blood pressure (BP), with a mean increase of 5 mm Hg.18 However, multiple recent studies report that in women with preeclampsia with and without severe features, postpartum administration of ibuprofen and ketorolac did not increase BP or delay resolution of hypertension.19-22 In a meta-analysis of randomized controlled studies comparing the effects of ibuprofen and acetaminophen on BP, neither medication was associated with an increase in BP.19 The American College of Obstetricians and Gynecologists supports the use of NSAIDs as one component of multimodal analgesia to help reduce the use of opioids.23
NSAIDs can inhibit platelet function and this effect is of clinical concern for people with platelet defects. However, a meta-analysis of clinical trials reported no difference in bleeding between surgical patients administered ketorolac or control participants.24 Alternative opioid-sparing adjuncts (TAP or QL blocks) may be considered for patients who cannot receive ketorolac based on a history of platelet deficiency. Furthermore, patients with ongoing coagulation defects after surgery from severe postpartum hemorrhage, hyperfibrinolysis, disseminated intravascular coagulation, or dilutional coagulopathy may have both limited platelet reserves and acute kidney injury. The need to postpone the initiation of NSAIDs in such patients should prompt alternate options such as TAP or QL blocks or dosing of an indwelling epidural when possible, in conjunction with acetaminophen. Patients who have a contraindication to ketorolac due to peptic ulcer disease or renal insufficiency may also benefit from TAP and QL blocks after cesarean delivery, although more studies are needed in these patients.
Both ketorolac and ibuprofen transfer to breast milk. The relative infant dose for ketorolac and ibuprofen is very low—0.2% and 0.9%, respectively.25,26 The World Health Organization advises that ibuprofen is compatible with breastfeeding.27 Of interest, in an enhanced recovery after cesarean clinical trial, scheduled ketorolac administration resulted in more mothers exclusively breastfeeding at discharge compared with “as needed” ketorolac treatment, 67% versus 48%, respectively; P = .046.28
Conclusion
Many factors influence a person’s experience of their surgery, including their pain symptoms. Factors that modulate a person’s perception of pain following surgery include their personality, social supports, and genetic factors. The technical skill of the anesthesiologist, surgeon, and nurses, and the confidence of the patient in the surgical care team are important factors influencing a person’s global experience of their surgery, including their experience of pain. Patients’ expectations regarding postoperative pain and psychological distress surrounding surgery may also influence their pain experience. Assuring patients that their pain will be addressed adequately, and helping them manage peripartum anxiety, also may favorably impact their pain experience.
Following a surgical procedure, a surgeon’s top goal is the full recovery of the patient to normal activity as soon as possible with as few complications as possible. Persistent opioid dependence is a serious long-term complication of surgery. Decades ago, most heroin users reported that heroin was the first opioid they used. However, the gateway drug to heroin use has evolved. In a recent study, 75% of heroin users reported that the first opioid they used was a prescription opioid.29 In managing surgical pain we want to minimize the use of opioids and reduce the risk of persistent opioid use following discharge. We believe that implementing a multimodal approach to the management of pain with additional targeted therapy for patients at risk for higher opioid requirement will reduce the perioperative and postdischarge use of opioid analgesics. ●
- Drug overdose deaths in the U.S. up 30% in 2020. Centers for Disease Control and Prevention web- site. July 14, 2020. https://www.cdc.gov/nchs /pressroom/nchs_press_releases/2021/20210714 .htm. Last reviewed July 14, 2021
- Jani M, Girard N, Bates DW, et al. Opioid prescribing among new users for non-cancer pain in the USA, Canada, UK, and Taiwan: a population-based cohort study. PLoS Med. 2021;18:e1003829.
- U.S. opioid dispensing rate maps. Centers for Disease Control and Prevention website. https://www. cdc.gov/drugoverdose/rxrate-maps/index.html. Last reviewed November 10, 2021.
- Richards GC, Aronson JK, Mahtani KR, et al. Global, regional, and national consumption of controlled opioids: a cross-sectional study of 214 countries and non-metropolitan areas. British J Pain. 2021. https://doi .org/10.1177/20494637211013052.
- Hamilton BE, Martin JA, Osterman MJK. Births: Provisional data for 2020. Vital Statistics Rapid Release; no 12. Hyattsville MD: National Center for Health Statistics. May 2021.
- Bateman BT, Franklin JM, Bykov K, et al. Persistent opioid use following cesarean delivery: patterns and predictors among opioid-naïve women. Am J Obstet Gynecol. 2016;215:353.e1-e8. doi: 10.1016/j.ajog.2016.03.016.
- Osmundson SS, Wiese AD, Min JY, et al. Delivery type, opioid prescribing and the risk of persistent opioid use after delivery. Am J Obstet Gynecol. 2019;220:405-407. doi: 10.1016/j.ajog.2018.10.026.
- Peahl AF, Dalton VK, Montgomery JR, et al. Rates of new persistent opioid use after vaginal or cesarean birth among U.S. women. JAMA Netw Open. 2019;e197863. doi: 10.1001/jamanetworkopen.2019.7863.
- Holland E, Bateman BT, Cole N, et al. Evaluation of a quality improvement intervention that eliminated routine use of opioids after cesarean delivery. Obstet Gynecol. 2019;133:91-97. doi: 10.1097/AOG.0000000000003010.
- Smith AM, Young P, Blosser CC, et al. Multimodal stepwise approach to reducing in-hospital opioid use after cesarean delivery. Obstet Gynecol. 2019;133:700-706. doi: 10.1097/AOG.0000000000003156.
- Herbert KA, Yuraschevich M, Fuller M, et al. Impact of multimodeal analgesic protocol modification on opioid consumption after cesarean delivery: a retrospective cohort study. J Matern Fetal Neonatal Med. 2021;3:1-7. doi: 10.1080/14767058.2020.1863364.
- Mehraban SS, Suddle R, Mehraban S, et al. Opioid-free multimodal analgesia pathway to decrease opioid utilization after cesarean delivery. J Obstet Gynaecol Res. 2021;47:873-881. doi: 10.1111/jog.14582.
- Meyer MF, Broman AT, Gnadt SE, et al. A standardized post-cesarean analgesia regimen reduces postpartum opioid use. J Matern Fetal Neonatal Med. 2021;26:1-8. doi: 10.1080/14767058.2021.1970132.
- Seki H, Shiga T, Mihara T, et al. Effects of intrathecal opioids on cesarean section: a systematic review and Bayesian network meta-analysis of randomized controlled trials. J Anesth. 2021;35:911-927. doi: 10.1007/s00540-021-02980-2.
- Yang TR, He XM, Li XH, et al. Intrathecal morphine versus transversus abdominis plane block for cesarean delivery: a systematic review and meta-analysis. BMC Anesthesiol. 2021;21:174. doi: 10.1186/s12871-021-01392-9.
- Yu Y, Gao S, Yuen VMY, et al. The analgesic efficacy of ultrasound-guided transversus abdominis plane (TAP) block combined with oral multimodal analgesia in comparison with oral multimodal analgesia after cesarean delivery: a randomized controlled trial. BMC Anesthesiol. 2021;21:7. doi: 10.1186/s12871-020-01223-3.
- Mirza F, Carvalho B. Transversus abdominis plane blocks for rescue analgesia following cesarean delivery: a case series. Can J Anesth. 2013;60:299-303.
- Johnson AG, Nguyen TV, Day RO. Do nonsteroidal anti-inflammatory drugs affect blood pressure? A meta-analysis. Ann Int Med. 1994;121:289-300.
- Wang B, Yang X, Yu H, et al. The comparison of ibuprofen versus acetaminophen for blood pressure in preeclampsia: a meta-analysis of randomized controlled studies. J Matern Fetal Neonatal Med. 2020:1-6. doi: 10.1080/14767058.2020.1720641.
- Viteri OA, England JA, Alrais MA, et al. Association of nonsteroidal anti-inflammatory drugs and postpartum hypertension in women with preeclampsia with severe features. Obstet Gynecol. 2017;130:830. doi: 10.1097/AOG.0000000000002247.
- Blue NR, Murray-Krezan C, Drake-Lavelle S, et al. Effect of ibuprofen vs acetaminophen on postpartum hypertension in preeclampsia with severe features: a double-masked, randomized controlled trial. Am J Obstet Gynecol. 2018;218:616.e1. doi: 10.1016/j.ajog.2018.02.016.
- Penfield CA, McNulty JA, Oakes MC, et al. Ibuprofen and postpartum blood pressure in women with hypertensive disorders of pregnancy: a randomized controlled trial. Obstet Gynecol. 2019;134:1219. doi: 10.1097/AOG.0000000000003553.
- American College of Obstetricians and Gynecologists. Pharmacologic stepwise multimodal approach for postpartum pain management. Obstet Gynecol. 2021;138:507-517. doi: 10.1097/AOG.0000000000004517.
- Gobble RM, Hoang HLT, Kachniarz B, et al. Ketorolac does not increase perioperative bleeding: a meta-analysis of randomized controlled trials. Plast Reconstr Surg. 2014;133:741. doi: 10.1097/01.prs.0000438459.60474.b5.
- Wischik A, Manth SM, Lloyd J, et al. The excretion of ketorolac tromethamine into breast milk after multiple oral dosing. Eur J Clin Pharmacol. 1989;36:521-524. doi: 10.1007/BF00558080.
- Rigourd V, de Villepin B, Amirouche A, et al. Ibuprofen concentrations in human mature milk-first data about pharmacokinetics study in breast milk with AOR-10127 “Antalait” study. The Drug Monit. 2014;36:590-596. doi: 10.1097/FTD.0000000000000058.
- World Health Organization. Breastfeeding and maternal medication, recommendations for drugs in the eleventh WHO model list of essential drugs. 2002. http://www.who.int/maternal _child_adolescent/documents/55732/en/.
- Teigen NC, Sahasrabudhe N, Doulaveris G. Enhanced recovery after surgery at cesarean delivery to reduce postoperative length of stay: a randomized controlled trial. Am J Obstet Gynecol. 2020;222:372.e1-e10. doi: 10.1016/j.ajog.2019.10.009.
- Cicero T, Ellis MS, Surratt HL, et al. The changing face of heroin use in the United States: a retrospective analysis of the past 50 years. JAMA Psychiatry. 2014;71:821-826. doi: 10.1001 /jamapsychiatry.2014.366.
Opioid-related deaths are a major cause of mortality in the United States. The Centers for Disease Control and Prevention (CDC) reported 72,151 and 93,331 drug overdose deaths in 2019 and 2020, respectively, and drug overdose deaths have continued to increase in 2021.1 The majority of drug overdose deaths are due to opioids. There are many factors contributing to this rise, including an incredibly high rate of opioid prescriptions in this country.2 The CDC reported that in 3.6% of US counties, there are more opioid prescriptions filled each year than number of residents in the county.3 The consumption of opioids per person in the US is approximately four times greater than countries with excellent health outcomes, including Sweden, Netherlands, Norway, and the United Kingdom.4 Some US physicians have opioid prescribing practices that are inconsistent with good medical practice in other countries, prescribing powerful opioids and an excessive number of pills per opioid prescription.2 We must continue to evolve our clinical practices to reduce opioid use while continually improving patient outcomes.
Cesarean birth is one of the most common major surgical procedures performed in the United States. The National Center for Health Statistics reported that in 2020 there were approximately 1,150,000 US cesarean births.5 Following cesarean birth, patients who were previously naïve to opioid medications were reported to have a 0.33% to 2.2% probability of transitioning to the persistent use of opioid prescriptions.6-8 Predictors of persistent opioid use after cesarean birth included a history of tobacco use, back pain, migraine headaches, and antidepressant or benzodiazepine use.6 The use of cesarean birth pain management protocols that prioritize multimodal analgesia and opioid sparing is warranted.
Multimodal pain management protocols for cesarean birth have been shown to reduce the use of opioid medications in the hospital and at discharge without a clinically significant increase in pain scores or a reduction in patient satisfaction (TABLE).9-13 For example, Holland and colleagues9 reported that the implementation of a multimodal pain management protocol reduced the percent of patients using oral opioids during hospitalization for cesarean birth from 68% to 45%, pre- and post-intervention, respectively. Mehraban and colleagues12 reported that the percent of patients using opioids during hospitalization for cesarean birth was reduced from 45% preintervention to 18% postintervention. In addition, these studies showed that multimodal pain management protocols for cesarean birth also reduced opioid prescribing at discharge. Holland and colleagues9 reported that the percent of patients provided an opioid prescription at discharge was reduced from 91% to 40%, pre- and post-intervention, respectively. Mehraban and colleagues12 reported that the percent of patients who took opioids after discharge was reduced from 24% preintervention to 9% postintervention. These studies were not randomized controlled clinical trials, but they do provide strong evidence that a focused intervention to reduce opioid medications in the management of pain after cesarean surgery can be successful without decreasing patient satisfaction or increasing reported pain scores. In these studies, it is likely that the influence, enthusiasm, and commitment of the study leaders to the change process contributed to the success of these opioid-sparing pain management programs.

Continue to: Key features of a multimodal analgesia intervention for cesarean surgery...
Key features of a multimodal analgesia intervention for cesarean surgery
Fundamental inclusions of multimodal analgesia for cesarean surgery include:
- exquisite attention to pain control during the surgical procedure by both the anesthesiologist and surgeon, with prioritization of spinal anesthesia that includes morphine and fentanyl
- regularly scheduled administration of intravenous ketorolac during the first 24 hours postcesarean
- regularly scheduled administration of both acetaminophen and ibuprofen, rather than “as needed” dosing
- using analgesics that work through different molecular pathways (ibuprofen and acetaminophen) (See Table.).
The significance of neuraxial and truncal nerve blockade for post-cesarean delivery pain control
Administration of a long-acting intrathecal opioid such as morphine lengthens time to first analgesic request after surgery and lowers 24-hour post‒cesarean delivery opioid requirement.14 If a patient requires general anesthesia and receives no spinal opioid, a transversus abdominis plane (TAP) block or quadratus lumborum (QL) block for postpartum pain control can lower associated postpartum opioid consumption. However, TAP or QL blocks confer no additional benefit to patients who receive spinal morphine,15 nor do they confer added benefit when combined with a multimodal pain management regimen postdelivery vs the multimodal regimen alone.16). TAP blocks administered to patients with severe breakthrough pain after spinal anesthesia help to lower opioid consumption.17 Further research is warranted on the use of TAP, QL, or other truncal blocks to spare opioid requirement after cesarean delivery in women with chronic pain, opioid use disorder, or those undergoing higher-complexity surgery such as cesarean hysterectomy for placenta accreta spectrum.
NSAIDs: Potential adverse effects
As we decrease the use of opioid medications and increase the use of nonsteroidal anti-inflammatory drugs (NSAIDs), we should reflect on the potential adverse effects of NSAID treatment in some patients. Specifically, the impact of ketorolac on hypertension, platelet function, and breastfeeding warrant consideration.
In the past, some studies reported that NSAID treatment is associated with a modest increase in blood pressure (BP), with a mean increase of 5 mm Hg.18 However, multiple recent studies report that in women with preeclampsia with and without severe features, postpartum administration of ibuprofen and ketorolac did not increase BP or delay resolution of hypertension.19-22 In a meta-analysis of randomized controlled studies comparing the effects of ibuprofen and acetaminophen on BP, neither medication was associated with an increase in BP.19 The American College of Obstetricians and Gynecologists supports the use of NSAIDs as one component of multimodal analgesia to help reduce the use of opioids.23
NSAIDs can inhibit platelet function and this effect is of clinical concern for people with platelet defects. However, a meta-analysis of clinical trials reported no difference in bleeding between surgical patients administered ketorolac or control participants.24 Alternative opioid-sparing adjuncts (TAP or QL blocks) may be considered for patients who cannot receive ketorolac based on a history of platelet deficiency. Furthermore, patients with ongoing coagulation defects after surgery from severe postpartum hemorrhage, hyperfibrinolysis, disseminated intravascular coagulation, or dilutional coagulopathy may have both limited platelet reserves and acute kidney injury. The need to postpone the initiation of NSAIDs in such patients should prompt alternate options such as TAP or QL blocks or dosing of an indwelling epidural when possible, in conjunction with acetaminophen. Patients who have a contraindication to ketorolac due to peptic ulcer disease or renal insufficiency may also benefit from TAP and QL blocks after cesarean delivery, although more studies are needed in these patients.
Both ketorolac and ibuprofen transfer to breast milk. The relative infant dose for ketorolac and ibuprofen is very low—0.2% and 0.9%, respectively.25,26 The World Health Organization advises that ibuprofen is compatible with breastfeeding.27 Of interest, in an enhanced recovery after cesarean clinical trial, scheduled ketorolac administration resulted in more mothers exclusively breastfeeding at discharge compared with “as needed” ketorolac treatment, 67% versus 48%, respectively; P = .046.28
Conclusion
Many factors influence a person’s experience of their surgery, including their pain symptoms. Factors that modulate a person’s perception of pain following surgery include their personality, social supports, and genetic factors. The technical skill of the anesthesiologist, surgeon, and nurses, and the confidence of the patient in the surgical care team are important factors influencing a person’s global experience of their surgery, including their experience of pain. Patients’ expectations regarding postoperative pain and psychological distress surrounding surgery may also influence their pain experience. Assuring patients that their pain will be addressed adequately, and helping them manage peripartum anxiety, also may favorably impact their pain experience.
Following a surgical procedure, a surgeon’s top goal is the full recovery of the patient to normal activity as soon as possible with as few complications as possible. Persistent opioid dependence is a serious long-term complication of surgery. Decades ago, most heroin users reported that heroin was the first opioid they used. However, the gateway drug to heroin use has evolved. In a recent study, 75% of heroin users reported that the first opioid they used was a prescription opioid.29 In managing surgical pain we want to minimize the use of opioids and reduce the risk of persistent opioid use following discharge. We believe that implementing a multimodal approach to the management of pain with additional targeted therapy for patients at risk for higher opioid requirement will reduce the perioperative and postdischarge use of opioid analgesics. ●
Opioid-related deaths are a major cause of mortality in the United States. The Centers for Disease Control and Prevention (CDC) reported 72,151 and 93,331 drug overdose deaths in 2019 and 2020, respectively, and drug overdose deaths have continued to increase in 2021.1 The majority of drug overdose deaths are due to opioids. There are many factors contributing to this rise, including an incredibly high rate of opioid prescriptions in this country.2 The CDC reported that in 3.6% of US counties, there are more opioid prescriptions filled each year than number of residents in the county.3 The consumption of opioids per person in the US is approximately four times greater than countries with excellent health outcomes, including Sweden, Netherlands, Norway, and the United Kingdom.4 Some US physicians have opioid prescribing practices that are inconsistent with good medical practice in other countries, prescribing powerful opioids and an excessive number of pills per opioid prescription.2 We must continue to evolve our clinical practices to reduce opioid use while continually improving patient outcomes.
Cesarean birth is one of the most common major surgical procedures performed in the United States. The National Center for Health Statistics reported that in 2020 there were approximately 1,150,000 US cesarean births.5 Following cesarean birth, patients who were previously naïve to opioid medications were reported to have a 0.33% to 2.2% probability of transitioning to the persistent use of opioid prescriptions.6-8 Predictors of persistent opioid use after cesarean birth included a history of tobacco use, back pain, migraine headaches, and antidepressant or benzodiazepine use.6 The use of cesarean birth pain management protocols that prioritize multimodal analgesia and opioid sparing is warranted.
Multimodal pain management protocols for cesarean birth have been shown to reduce the use of opioid medications in the hospital and at discharge without a clinically significant increase in pain scores or a reduction in patient satisfaction (TABLE).9-13 For example, Holland and colleagues9 reported that the implementation of a multimodal pain management protocol reduced the percent of patients using oral opioids during hospitalization for cesarean birth from 68% to 45%, pre- and post-intervention, respectively. Mehraban and colleagues12 reported that the percent of patients using opioids during hospitalization for cesarean birth was reduced from 45% preintervention to 18% postintervention. In addition, these studies showed that multimodal pain management protocols for cesarean birth also reduced opioid prescribing at discharge. Holland and colleagues9 reported that the percent of patients provided an opioid prescription at discharge was reduced from 91% to 40%, pre- and post-intervention, respectively. Mehraban and colleagues12 reported that the percent of patients who took opioids after discharge was reduced from 24% preintervention to 9% postintervention. These studies were not randomized controlled clinical trials, but they do provide strong evidence that a focused intervention to reduce opioid medications in the management of pain after cesarean surgery can be successful without decreasing patient satisfaction or increasing reported pain scores. In these studies, it is likely that the influence, enthusiasm, and commitment of the study leaders to the change process contributed to the success of these opioid-sparing pain management programs.

Continue to: Key features of a multimodal analgesia intervention for cesarean surgery...
Key features of a multimodal analgesia intervention for cesarean surgery
Fundamental inclusions of multimodal analgesia for cesarean surgery include:
- exquisite attention to pain control during the surgical procedure by both the anesthesiologist and surgeon, with prioritization of spinal anesthesia that includes morphine and fentanyl
- regularly scheduled administration of intravenous ketorolac during the first 24 hours postcesarean
- regularly scheduled administration of both acetaminophen and ibuprofen, rather than “as needed” dosing
- using analgesics that work through different molecular pathways (ibuprofen and acetaminophen) (See Table.).
The significance of neuraxial and truncal nerve blockade for post-cesarean delivery pain control
Administration of a long-acting intrathecal opioid such as morphine lengthens time to first analgesic request after surgery and lowers 24-hour post‒cesarean delivery opioid requirement.14 If a patient requires general anesthesia and receives no spinal opioid, a transversus abdominis plane (TAP) block or quadratus lumborum (QL) block for postpartum pain control can lower associated postpartum opioid consumption. However, TAP or QL blocks confer no additional benefit to patients who receive spinal morphine,15 nor do they confer added benefit when combined with a multimodal pain management regimen postdelivery vs the multimodal regimen alone.16). TAP blocks administered to patients with severe breakthrough pain after spinal anesthesia help to lower opioid consumption.17 Further research is warranted on the use of TAP, QL, or other truncal blocks to spare opioid requirement after cesarean delivery in women with chronic pain, opioid use disorder, or those undergoing higher-complexity surgery such as cesarean hysterectomy for placenta accreta spectrum.
NSAIDs: Potential adverse effects
As we decrease the use of opioid medications and increase the use of nonsteroidal anti-inflammatory drugs (NSAIDs), we should reflect on the potential adverse effects of NSAID treatment in some patients. Specifically, the impact of ketorolac on hypertension, platelet function, and breastfeeding warrant consideration.
In the past, some studies reported that NSAID treatment is associated with a modest increase in blood pressure (BP), with a mean increase of 5 mm Hg.18 However, multiple recent studies report that in women with preeclampsia with and without severe features, postpartum administration of ibuprofen and ketorolac did not increase BP or delay resolution of hypertension.19-22 In a meta-analysis of randomized controlled studies comparing the effects of ibuprofen and acetaminophen on BP, neither medication was associated with an increase in BP.19 The American College of Obstetricians and Gynecologists supports the use of NSAIDs as one component of multimodal analgesia to help reduce the use of opioids.23
NSAIDs can inhibit platelet function and this effect is of clinical concern for people with platelet defects. However, a meta-analysis of clinical trials reported no difference in bleeding between surgical patients administered ketorolac or control participants.24 Alternative opioid-sparing adjuncts (TAP or QL blocks) may be considered for patients who cannot receive ketorolac based on a history of platelet deficiency. Furthermore, patients with ongoing coagulation defects after surgery from severe postpartum hemorrhage, hyperfibrinolysis, disseminated intravascular coagulation, or dilutional coagulopathy may have both limited platelet reserves and acute kidney injury. The need to postpone the initiation of NSAIDs in such patients should prompt alternate options such as TAP or QL blocks or dosing of an indwelling epidural when possible, in conjunction with acetaminophen. Patients who have a contraindication to ketorolac due to peptic ulcer disease or renal insufficiency may also benefit from TAP and QL blocks after cesarean delivery, although more studies are needed in these patients.
Both ketorolac and ibuprofen transfer to breast milk. The relative infant dose for ketorolac and ibuprofen is very low—0.2% and 0.9%, respectively.25,26 The World Health Organization advises that ibuprofen is compatible with breastfeeding.27 Of interest, in an enhanced recovery after cesarean clinical trial, scheduled ketorolac administration resulted in more mothers exclusively breastfeeding at discharge compared with “as needed” ketorolac treatment, 67% versus 48%, respectively; P = .046.28
Conclusion
Many factors influence a person’s experience of their surgery, including their pain symptoms. Factors that modulate a person’s perception of pain following surgery include their personality, social supports, and genetic factors. The technical skill of the anesthesiologist, surgeon, and nurses, and the confidence of the patient in the surgical care team are important factors influencing a person’s global experience of their surgery, including their experience of pain. Patients’ expectations regarding postoperative pain and psychological distress surrounding surgery may also influence their pain experience. Assuring patients that their pain will be addressed adequately, and helping them manage peripartum anxiety, also may favorably impact their pain experience.
Following a surgical procedure, a surgeon’s top goal is the full recovery of the patient to normal activity as soon as possible with as few complications as possible. Persistent opioid dependence is a serious long-term complication of surgery. Decades ago, most heroin users reported that heroin was the first opioid they used. However, the gateway drug to heroin use has evolved. In a recent study, 75% of heroin users reported that the first opioid they used was a prescription opioid.29 In managing surgical pain we want to minimize the use of opioids and reduce the risk of persistent opioid use following discharge. We believe that implementing a multimodal approach to the management of pain with additional targeted therapy for patients at risk for higher opioid requirement will reduce the perioperative and postdischarge use of opioid analgesics. ●
- Drug overdose deaths in the U.S. up 30% in 2020. Centers for Disease Control and Prevention web- site. July 14, 2020. https://www.cdc.gov/nchs /pressroom/nchs_press_releases/2021/20210714 .htm. Last reviewed July 14, 2021
- Jani M, Girard N, Bates DW, et al. Opioid prescribing among new users for non-cancer pain in the USA, Canada, UK, and Taiwan: a population-based cohort study. PLoS Med. 2021;18:e1003829.
- U.S. opioid dispensing rate maps. Centers for Disease Control and Prevention website. https://www. cdc.gov/drugoverdose/rxrate-maps/index.html. Last reviewed November 10, 2021.
- Richards GC, Aronson JK, Mahtani KR, et al. Global, regional, and national consumption of controlled opioids: a cross-sectional study of 214 countries and non-metropolitan areas. British J Pain. 2021. https://doi .org/10.1177/20494637211013052.
- Hamilton BE, Martin JA, Osterman MJK. Births: Provisional data for 2020. Vital Statistics Rapid Release; no 12. Hyattsville MD: National Center for Health Statistics. May 2021.
- Bateman BT, Franklin JM, Bykov K, et al. Persistent opioid use following cesarean delivery: patterns and predictors among opioid-naïve women. Am J Obstet Gynecol. 2016;215:353.e1-e8. doi: 10.1016/j.ajog.2016.03.016.
- Osmundson SS, Wiese AD, Min JY, et al. Delivery type, opioid prescribing and the risk of persistent opioid use after delivery. Am J Obstet Gynecol. 2019;220:405-407. doi: 10.1016/j.ajog.2018.10.026.
- Peahl AF, Dalton VK, Montgomery JR, et al. Rates of new persistent opioid use after vaginal or cesarean birth among U.S. women. JAMA Netw Open. 2019;e197863. doi: 10.1001/jamanetworkopen.2019.7863.
- Holland E, Bateman BT, Cole N, et al. Evaluation of a quality improvement intervention that eliminated routine use of opioids after cesarean delivery. Obstet Gynecol. 2019;133:91-97. doi: 10.1097/AOG.0000000000003010.
- Smith AM, Young P, Blosser CC, et al. Multimodal stepwise approach to reducing in-hospital opioid use after cesarean delivery. Obstet Gynecol. 2019;133:700-706. doi: 10.1097/AOG.0000000000003156.
- Herbert KA, Yuraschevich M, Fuller M, et al. Impact of multimodeal analgesic protocol modification on opioid consumption after cesarean delivery: a retrospective cohort study. J Matern Fetal Neonatal Med. 2021;3:1-7. doi: 10.1080/14767058.2020.1863364.
- Mehraban SS, Suddle R, Mehraban S, et al. Opioid-free multimodal analgesia pathway to decrease opioid utilization after cesarean delivery. J Obstet Gynaecol Res. 2021;47:873-881. doi: 10.1111/jog.14582.
- Meyer MF, Broman AT, Gnadt SE, et al. A standardized post-cesarean analgesia regimen reduces postpartum opioid use. J Matern Fetal Neonatal Med. 2021;26:1-8. doi: 10.1080/14767058.2021.1970132.
- Seki H, Shiga T, Mihara T, et al. Effects of intrathecal opioids on cesarean section: a systematic review and Bayesian network meta-analysis of randomized controlled trials. J Anesth. 2021;35:911-927. doi: 10.1007/s00540-021-02980-2.
- Yang TR, He XM, Li XH, et al. Intrathecal morphine versus transversus abdominis plane block for cesarean delivery: a systematic review and meta-analysis. BMC Anesthesiol. 2021;21:174. doi: 10.1186/s12871-021-01392-9.
- Yu Y, Gao S, Yuen VMY, et al. The analgesic efficacy of ultrasound-guided transversus abdominis plane (TAP) block combined with oral multimodal analgesia in comparison with oral multimodal analgesia after cesarean delivery: a randomized controlled trial. BMC Anesthesiol. 2021;21:7. doi: 10.1186/s12871-020-01223-3.
- Mirza F, Carvalho B. Transversus abdominis plane blocks for rescue analgesia following cesarean delivery: a case series. Can J Anesth. 2013;60:299-303.
- Johnson AG, Nguyen TV, Day RO. Do nonsteroidal anti-inflammatory drugs affect blood pressure? A meta-analysis. Ann Int Med. 1994;121:289-300.
- Wang B, Yang X, Yu H, et al. The comparison of ibuprofen versus acetaminophen for blood pressure in preeclampsia: a meta-analysis of randomized controlled studies. J Matern Fetal Neonatal Med. 2020:1-6. doi: 10.1080/14767058.2020.1720641.
- Viteri OA, England JA, Alrais MA, et al. Association of nonsteroidal anti-inflammatory drugs and postpartum hypertension in women with preeclampsia with severe features. Obstet Gynecol. 2017;130:830. doi: 10.1097/AOG.0000000000002247.
- Blue NR, Murray-Krezan C, Drake-Lavelle S, et al. Effect of ibuprofen vs acetaminophen on postpartum hypertension in preeclampsia with severe features: a double-masked, randomized controlled trial. Am J Obstet Gynecol. 2018;218:616.e1. doi: 10.1016/j.ajog.2018.02.016.
- Penfield CA, McNulty JA, Oakes MC, et al. Ibuprofen and postpartum blood pressure in women with hypertensive disorders of pregnancy: a randomized controlled trial. Obstet Gynecol. 2019;134:1219. doi: 10.1097/AOG.0000000000003553.
- American College of Obstetricians and Gynecologists. Pharmacologic stepwise multimodal approach for postpartum pain management. Obstet Gynecol. 2021;138:507-517. doi: 10.1097/AOG.0000000000004517.
- Gobble RM, Hoang HLT, Kachniarz B, et al. Ketorolac does not increase perioperative bleeding: a meta-analysis of randomized controlled trials. Plast Reconstr Surg. 2014;133:741. doi: 10.1097/01.prs.0000438459.60474.b5.
- Wischik A, Manth SM, Lloyd J, et al. The excretion of ketorolac tromethamine into breast milk after multiple oral dosing. Eur J Clin Pharmacol. 1989;36:521-524. doi: 10.1007/BF00558080.
- Rigourd V, de Villepin B, Amirouche A, et al. Ibuprofen concentrations in human mature milk-first data about pharmacokinetics study in breast milk with AOR-10127 “Antalait” study. The Drug Monit. 2014;36:590-596. doi: 10.1097/FTD.0000000000000058.
- World Health Organization. Breastfeeding and maternal medication, recommendations for drugs in the eleventh WHO model list of essential drugs. 2002. http://www.who.int/maternal _child_adolescent/documents/55732/en/.
- Teigen NC, Sahasrabudhe N, Doulaveris G. Enhanced recovery after surgery at cesarean delivery to reduce postoperative length of stay: a randomized controlled trial. Am J Obstet Gynecol. 2020;222:372.e1-e10. doi: 10.1016/j.ajog.2019.10.009.
- Cicero T, Ellis MS, Surratt HL, et al. The changing face of heroin use in the United States: a retrospective analysis of the past 50 years. JAMA Psychiatry. 2014;71:821-826. doi: 10.1001 /jamapsychiatry.2014.366.
- Drug overdose deaths in the U.S. up 30% in 2020. Centers for Disease Control and Prevention web- site. July 14, 2020. https://www.cdc.gov/nchs /pressroom/nchs_press_releases/2021/20210714 .htm. Last reviewed July 14, 2021
- Jani M, Girard N, Bates DW, et al. Opioid prescribing among new users for non-cancer pain in the USA, Canada, UK, and Taiwan: a population-based cohort study. PLoS Med. 2021;18:e1003829.
- U.S. opioid dispensing rate maps. Centers for Disease Control and Prevention website. https://www. cdc.gov/drugoverdose/rxrate-maps/index.html. Last reviewed November 10, 2021.
- Richards GC, Aronson JK, Mahtani KR, et al. Global, regional, and national consumption of controlled opioids: a cross-sectional study of 214 countries and non-metropolitan areas. British J Pain. 2021. https://doi .org/10.1177/20494637211013052.
- Hamilton BE, Martin JA, Osterman MJK. Births: Provisional data for 2020. Vital Statistics Rapid Release; no 12. Hyattsville MD: National Center for Health Statistics. May 2021.
- Bateman BT, Franklin JM, Bykov K, et al. Persistent opioid use following cesarean delivery: patterns and predictors among opioid-naïve women. Am J Obstet Gynecol. 2016;215:353.e1-e8. doi: 10.1016/j.ajog.2016.03.016.
- Osmundson SS, Wiese AD, Min JY, et al. Delivery type, opioid prescribing and the risk of persistent opioid use after delivery. Am J Obstet Gynecol. 2019;220:405-407. doi: 10.1016/j.ajog.2018.10.026.
- Peahl AF, Dalton VK, Montgomery JR, et al. Rates of new persistent opioid use after vaginal or cesarean birth among U.S. women. JAMA Netw Open. 2019;e197863. doi: 10.1001/jamanetworkopen.2019.7863.
- Holland E, Bateman BT, Cole N, et al. Evaluation of a quality improvement intervention that eliminated routine use of opioids after cesarean delivery. Obstet Gynecol. 2019;133:91-97. doi: 10.1097/AOG.0000000000003010.
- Smith AM, Young P, Blosser CC, et al. Multimodal stepwise approach to reducing in-hospital opioid use after cesarean delivery. Obstet Gynecol. 2019;133:700-706. doi: 10.1097/AOG.0000000000003156.
- Herbert KA, Yuraschevich M, Fuller M, et al. Impact of multimodeal analgesic protocol modification on opioid consumption after cesarean delivery: a retrospective cohort study. J Matern Fetal Neonatal Med. 2021;3:1-7. doi: 10.1080/14767058.2020.1863364.
- Mehraban SS, Suddle R, Mehraban S, et al. Opioid-free multimodal analgesia pathway to decrease opioid utilization after cesarean delivery. J Obstet Gynaecol Res. 2021;47:873-881. doi: 10.1111/jog.14582.
- Meyer MF, Broman AT, Gnadt SE, et al. A standardized post-cesarean analgesia regimen reduces postpartum opioid use. J Matern Fetal Neonatal Med. 2021;26:1-8. doi: 10.1080/14767058.2021.1970132.
- Seki H, Shiga T, Mihara T, et al. Effects of intrathecal opioids on cesarean section: a systematic review and Bayesian network meta-analysis of randomized controlled trials. J Anesth. 2021;35:911-927. doi: 10.1007/s00540-021-02980-2.
- Yang TR, He XM, Li XH, et al. Intrathecal morphine versus transversus abdominis plane block for cesarean delivery: a systematic review and meta-analysis. BMC Anesthesiol. 2021;21:174. doi: 10.1186/s12871-021-01392-9.
- Yu Y, Gao S, Yuen VMY, et al. The analgesic efficacy of ultrasound-guided transversus abdominis plane (TAP) block combined with oral multimodal analgesia in comparison with oral multimodal analgesia after cesarean delivery: a randomized controlled trial. BMC Anesthesiol. 2021;21:7. doi: 10.1186/s12871-020-01223-3.
- Mirza F, Carvalho B. Transversus abdominis plane blocks for rescue analgesia following cesarean delivery: a case series. Can J Anesth. 2013;60:299-303.
- Johnson AG, Nguyen TV, Day RO. Do nonsteroidal anti-inflammatory drugs affect blood pressure? A meta-analysis. Ann Int Med. 1994;121:289-300.
- Wang B, Yang X, Yu H, et al. The comparison of ibuprofen versus acetaminophen for blood pressure in preeclampsia: a meta-analysis of randomized controlled studies. J Matern Fetal Neonatal Med. 2020:1-6. doi: 10.1080/14767058.2020.1720641.
- Viteri OA, England JA, Alrais MA, et al. Association of nonsteroidal anti-inflammatory drugs and postpartum hypertension in women with preeclampsia with severe features. Obstet Gynecol. 2017;130:830. doi: 10.1097/AOG.0000000000002247.
- Blue NR, Murray-Krezan C, Drake-Lavelle S, et al. Effect of ibuprofen vs acetaminophen on postpartum hypertension in preeclampsia with severe features: a double-masked, randomized controlled trial. Am J Obstet Gynecol. 2018;218:616.e1. doi: 10.1016/j.ajog.2018.02.016.
- Penfield CA, McNulty JA, Oakes MC, et al. Ibuprofen and postpartum blood pressure in women with hypertensive disorders of pregnancy: a randomized controlled trial. Obstet Gynecol. 2019;134:1219. doi: 10.1097/AOG.0000000000003553.
- American College of Obstetricians and Gynecologists. Pharmacologic stepwise multimodal approach for postpartum pain management. Obstet Gynecol. 2021;138:507-517. doi: 10.1097/AOG.0000000000004517.
- Gobble RM, Hoang HLT, Kachniarz B, et al. Ketorolac does not increase perioperative bleeding: a meta-analysis of randomized controlled trials. Plast Reconstr Surg. 2014;133:741. doi: 10.1097/01.prs.0000438459.60474.b5.
- Wischik A, Manth SM, Lloyd J, et al. The excretion of ketorolac tromethamine into breast milk after multiple oral dosing. Eur J Clin Pharmacol. 1989;36:521-524. doi: 10.1007/BF00558080.
- Rigourd V, de Villepin B, Amirouche A, et al. Ibuprofen concentrations in human mature milk-first data about pharmacokinetics study in breast milk with AOR-10127 “Antalait” study. The Drug Monit. 2014;36:590-596. doi: 10.1097/FTD.0000000000000058.
- World Health Organization. Breastfeeding and maternal medication, recommendations for drugs in the eleventh WHO model list of essential drugs. 2002. http://www.who.int/maternal _child_adolescent/documents/55732/en/.
- Teigen NC, Sahasrabudhe N, Doulaveris G. Enhanced recovery after surgery at cesarean delivery to reduce postoperative length of stay: a randomized controlled trial. Am J Obstet Gynecol. 2020;222:372.e1-e10. doi: 10.1016/j.ajog.2019.10.009.
- Cicero T, Ellis MS, Surratt HL, et al. The changing face of heroin use in the United States: a retrospective analysis of the past 50 years. JAMA Psychiatry. 2014;71:821-826. doi: 10.1001 /jamapsychiatry.2014.366.