User login
Use of complementary therapies to treat the pain of osteoarthritis
At last count in 2008, 27 million Americans were suffering with osteoarthritis (OA), by far the most common form of arthritis.1 That number has undoubtedly risen and will continue to do so as Baby Boomers age. Despite the benefits of conventional nonpharmacologic measures and available pharmacologic agents, many patients with OA achieve less than satisfactory pain relief and have impaired joint mobility, which can significantly limit their daily activities.2 Numerous studies have found that a host of complementary and alternative (CAM) therapies can provide safe and effective pain relief and enhanced joint mobility. The National Center for Complementary and Alternative Therapies found that musculoskeletal problems such as back pain, neck pain, joint pain, and arthritis were the top conditions for which adults used CAM therapies in 2007 [Figure 1].3
| FIGURE 1: Diseases/conditions for which CAM is most frequently used among US adults – 2007 |

| Source: Barnes PM, Bloom B, Nahin R. CDC National Health Statistics Report #12. Complementary and alternative medicine use amongadults and children. United States, 2007. December 2008. |
A survey to gather detailed information about CAM use by patients with arthritis (n 5 2140) found that most patients followed by specialists (90.5%) and a slightly smaller percentage followed by primary care physicians (82.8%) had tried at least one CAM therapy for relief of OA symptoms. The authors suggested that the higher percentage for specialist care may be because these patients have more severe disease and therefore experience more pain.4 An understanding of these therapies will allow primary care and specialist physicians to better communicate with and advise patients who seek options outside the usual spectrum of care.
Definition and therapeutic goals
OA is a progressive deterioration of joint tissues.5 A decrease in protective proteoglycans and collagen compromises joint cartilage.5-8 Deterioration of cartilage in turn leads to bone erosions, osteophyte formation, and bone restructuring. Inflammation, too, may result in reaction to cartilage degradation byproducts entering synovial spaces.6,7 Joint pain aggravated by physical activity and alleviated by rest is typical of OA. Also common are joint instability and stiffness upon rising in the morning or after extended inactivity.9 A patient’s history may additionally reveal that the level of pain experienced with activity has steadily increased with time. Physical examination may reveal bony enlargement or deformity of involved joints, crepitus, and restricted range of motion.9 The value of laboratory and radiology studies lies mainly in ruling out alternative diagnoses.
The goals of OA treatment are pain relief and preservation of joint function. Because the experience of pain is influenced by physical, psychological, and emotional factors, individuals vary in how they respond to specific therapies and in how they wish to achieve pain relief. Some patients may experience side effects from anti-inflammatory pain medications.10 Others may be hesitant to resort to surgery.9 All major guidelines agree that, for most patients, therapy combining nonpharmacologic measures and pharmacologic agents is required to achieve optimal relief of pain and preservation of joint mobility.11-14
Conventional treatment options for OA
Selecting appropriate treatment begins with consideration of the patient’s report of chronic pain and limitations in ambulation or other activities. Also important is assessment of the patient’s level of pain on manipulation, as well as muscle strength and ligament stability.7,8 Depending on physical examination results, a reasonable approach may be to start with nonpharmacologic measures and add pharmacologic agents in a stepwise manner to control pain.8 Self-management programs have been shown to improve symptoms as well as quality of life, and should be incorporated into the treatment plan.15
Nonpharmacologic measures, prescribed as needed for each individual, include weight loss for those who are overweight or obese.11-14 Weight loss has been shown to improve mobility and reduce pain. For every one pound of weight lost, there is a 4-pound reduction in the load exerted on the knee for each step taken during daily activities.16 A weight loss of only 15 pounds can cut knee pain in half for overweight individuals with arthritis.17 A low-carbohydrate diet has been shown to reduce weight in obese patients by ≥10% and lead to improvements in self-reported scores for overall progress and functional ability.18 A diet of fruits and vegetables (including alliums and cruciferous vegetables) that is high in phytonutrients has been shown to have a protective effect in patients with hip OA.19
Other measures are physical and occupational therapies, assistive devices for walking or accomplishing other daily tasks, and joint taping.11-14 Patient (and family) education regarding the progressive nature of OA is crucial to bolstering patient resolve in following through with self-management activities.15 Healthcare professionals can provide factual, disease-specific information on some effective self-management strategies for use between office visits that yield short- and long-term benefits. Self-management strategies can incorporate pain management education; joint-sparing exercise advice including daily walking, balance tips, and falls prevention; and emotional and cognitive skills to improve quality of life.
Pharmacologic intervention is best begun with the least risky agent, at the lowest effective dose, for the shortest possible duration.12 For mild-to-moderate pain, the Osteoarthritis Research Society International recommends acetaminophen as initial therapy, given up to 4 g/d. If pain relief is insufficient, consider adding or switching to an oral or topical non-steroidal anti-inflammatory drug (NSAID), keeping in mind the possibility for gastrointestinal (GI) adverse effects with oral agents. When using an oral NSAID in patients at risk for GI effects, give a proton-pump inhibitor (PPI) as well. A topical NSAID or capsaicin may provide additional local pain relief.20
If pain remains inadequately controlled, or if a patient is intolerant to NSAIDs, a selective COX-2 inhibitor given alone or in
combination with acetaminophen may be an appropriate choice—provided the patient undergoes thorough evaluation for cardiovascular (CV) and GI risks.12,21 For those with GI risks taking a COX-2 inhibitor, give either misoprostol or a PPI concomitantly. Monitor those with CV risks closely.
When OA pain is severe or unresponsive to the previous medications, alternative agents include tramadol with or without acetaminophen, or opioid analgesics. In either instance, start at the lowest possible dosage (adjusted, as needed, for renal or hepatic impairment) and then titrate upward slowly to minimize adverse effects.9,12
Evidence-based complementary treatment options for OA
In its executive guidelines for US hospitals, the Joint Commission clearly states that institutions seeking accreditation, or wishing to maintain it, must include in its options for treating chronic pain such complementary measures as massage, acupuncture, and mind-body therapy. The Joint Commission’s basis for mandating an integrative approach to pain control is the growing body of clinical evidence supporting the efficacy of complementary treatments. In addition, many patients prefer nonpharmacologic options for personal reasons.
Hyaluronic acid. In a recently published meta-analysis of studies looking at intra-articular hyaluronic acid (IAHA) for knee OA pain, the authors reported therapeutic effectiveness beginning at 4 weeks, reaching a peak effect at 8 weeks, and exerting a residual effect still detectable at 24 weeks.22 Moreover, the peak effect of IAHA is greater than published effects for acetaminophen, NSAIDs, and COX-2 inhibitors.22 IAHA could be useful in combination with other therapies, and in certain clinical situations such as when patients are undecided about surgery. Other studies and meta-analyses of IAHA have yielded different results regarding length of efficacy.23,24
Acupuncture. While studies of acupuncture for OA have varied in quality and duration of effect, many show benefits. One meta-analysis found that patients who received acupuncture reported clinically relevant short- and long-term pain reduction and improved function compared with control patients who received usual care.25 In a study of
294 patients with OA of the knee, patients received 8 weeksof acupuncture, minimal acupuncture (superficial needling at nonacupuncture points), or no treatment. Based on the Western Ontario and McMasters Universities Osteoarthritis (WOMAC) questionnaire assessing pain, function, and stiffness due to OA, those who received acupuncture had significantly better scores than the other 2 groups. However, after 26 and 52 weeks, the differences between acupuncture and minimal acupuncture were no longer significant.26
In the largest study ever conducted on acupuncture for OA of the knee, 570 patients were randomly assigned to receive acupuncture, sham acupuncture, or education.27 At
26 weeks postintervention, based on the WOMAC function score those who underwent true acupuncture experienced significantly greater pain relief and functional improvement than those who received sham acupuncture or education only.
Mindfulness-Based Stress Reduction (MBSR), pioneered by Jon Kabat-Zinn, PhD, uses mindfulness meditation to alleviate chronic pain. First reported in a 1982 study, 88% of enrolled patients with chronic pain of 6 months to 48 years’ duration who were trained in stress reduction using mindfulness meditation perceived a decrease in pain of at least 50%.28 This method of focusing on the mind and body does not simply “distract” patients from their pain but achieves measurable changes in how pain is perceived. With MBSR, pain reduction reported by patients was significantly greater than that achieved with morphine and other drugs.29 Brain scans show that, after MBSR, regions normally associated with processing painful stimuli become quiescent.29 MBSR has also been shown in imaging studies to increase gray matter concentrations in regions of the brain associated with learning and memory, regulation of emotion, and self-referential processing [Figure 2].30
| FIGURE 2: Gray matter is more concentrated for persons who meditate compared with nonmeditators. |
| Source: Holzel BK, et al. Soc Cogn Affect Neurosci. 2008;3:55-61 |
A 2004 meta-analysis of 20 reports on mindfulness meditation that met study criteria included patients with anxiety, cancer diagnoses, chronic pain, coronary artery disease, depression, fibromyalgia, obesity and binge eating disorders, and psychiatric disorders. The analysis found a relatively consistent and strong effect, suggesting that mindfulness training may enhance the ability to cope with distress and disability and the sense of physical well-being.31
Chiropractic. One small randomized controlled trial (RCT) matched participants with knee OA pain measured by visual analog scale (VAS) and assigned them to receive either treatment according to the Macquarie Injury Management Group Knee Protocol or nonforceful contact.32 The treatment group reported significantly decreased knee symptoms such as crepitus and improvement in mobility and ability to perform general activities. A systematic review found fair evidence for efficacy of manipulative therapy of the knee combined with multimodal or exercise therapy for knee OA.33
Transcutaneous electrical nerve stimulation (TENS). Evidence for the effectiveness of TENS for OA is mixed. A 2009 Cochrane systematic review of TENS for knee OA concluded that available studies were too small and poorly designed to judge its effectiveness.34 However, its use in tibiofemoral OA has yielded good results.35
Tai Chi. This ancient form of meditative exercise has long been acknowledged as a means for enhancing balance and dexterity, but studies of chronic pain reduction and improved mobility with Tai Chi have been few and usually underpowered to yield reliable results. One systematic review of 9 RCTs, 23 non-RCTs, and 15 observational studies included studies that showed benefits for osteoarthritic symptoms with improved functional mobility and quality of life.36 Another systematic review of 5 RCTs and 7 nonrandomized clinical trials found that Tai Chi was effective for pain control of OA of the knee, but that evidence was inconclusive for pain reduction or mobility improvement.37 In a long-term study comparing Tai Chi with regular exercise in patients with symptomatic OA of the knee, researchers found that those in the Tai Chi group showed greater improvement in pain, physical function, self-efficacy, depression, and health-related quality of life, with some improvements lasting as long as 48 weeks.38
Massage. A longitudinal analysis of the use of a variety of alternative therapy offerings by older adults with OA found that the most commonly utilized treatment during a 20-week intervention period was massage (57%) followed by chiropractic (20.7%).39 In the first prospective randomized trial to evaluate the efficacy of massage for adults with OA of the knee, participants who received 8 weeks of Swedish massage therapy showed significant improvements in pain, stiffness, and physical function. These improvements persisted at the 16-week evaluation.40
Dietary supplements. Omega-3 fatty acids have shown promise in reducing chronic neuropathic pain when taken at levels that exceed 2 g/d of eicosapetaenoic acid and docosahexaenoic acid.41 This effect is probably enhanced when combined with a dietary reduction of omega-6 fatty acids and saturated fats. This dietary measure may also have some effect on joint pain.42
Glucosamine/chondroitin has been shown in a meta-analysis to reduce OA pain,43 although another meta-analysis showed no effect on either joint pain or narrowing of joint space.44 More recently, data from the long-term Glucosamine/Chondroitin Arthritis Intervention (GAIT) trial found that patients who took glucosamine or glucosamine/chondroitin in combination had similar results to those who took celecoxib or placebo. Over the 2-year study period, all groups showed improvement in painand function.45
It is worth noting that not all supplements are made the same, and there may be varying potencies. For example, some glucosamine products work better than others.46 When discussing supplements, to get the most potential benefit it is advisable to suggest those with a strong body of evidence or refer patients to providers who are familiar with CAM therapies.
Other promising dietary aids for reducing OA pain include soy protein,47 avocado-soybean unsaponifiables,48 Cat’s claw,49 white willow,49 green tea,49,50 turmeric,51 ginger,52 and propolis.53
Widening your integrative approach to OA treatment
There is emerging evidence that integrating multiple conventional and CAM therapies such as glucosamine and walking may provide the best results for OA patients.54 The realization that many patients with pain and diminished mobility are already exploring CAM therapies presents an opportunity for you to discuss their decisions and direct their attention to options that are supported by strong evidence. By evaluating the increasing body of evidence in support of specific CAM therapies, you can feel confident in offering your patients a wider range of choices than standard pharmacologic and nonpharmacologic OA options, and integrate these options to improve care.
Acknowledgement—The author wishes to thank Charles F. Williams for his assistance in researching and writing this article.
REFERENCES
1. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35.
2. Centers for Disease Control and Prevention. Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation—United States, 2007–2009. MMWR Morb Mortal Wkly Rep. 2010;59:1261-1265.
3. Barnes PM, Bloom B, Nahin R. CDC National Health Statistics Report #12. Complementary and alternative medicine use among adults and children: United States, 2007. December 2008. Available at: http://nccam.nih.gov/news/camstats/2007/camsurvey_fs1.htm#use.
4. Callahan LF, Wiley-Exley EK, Mielenz TJ, et al. Use of complementary and alternative medicine among patients with arthritis. Prev Chronic Dis. 2009;6:1-23.
5. Felson DT. An update on the pathogenesis and epidemiology of osteoarthritis. Radiol Clin North Am. 2004;42:1-9.
6. Breedveld FC. Osteoarthritis—the impact of a serious disease. Rheumatology. 2004;43(suppl 1):i4-i8.
7. Urquhart N, Stanish WD. Drugs for knee osteoarthritis: what primary care physicians need to know—a clear understanding of the symptoms and treatment options is required. J Musculoskel Med. 2007;24:178-187.
8. Hunter DJ. In the clinic: osteoarthritis. Arch Intern Med. 2007;147:ITC8-1–ITC8-16.
9. American College of Rheumatology. Recommendations for the medical management of osteoarthritisof the hip and knee. 2000. Available at: http://www.rheumatology.org/practice/clinical/guidelines/oa-mgmt.asp.
10. Tramer MR, Moore RA, Reynolds DJ, et al. Quantitative estimation of rare adverse events which follow a biological progression: a new model applied to chronic NSAID use. Pain. 2000;85:169-182.
11. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part I. Critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage. 2007;15:981-1000.
12. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part II. OARSI evidence-based, expert consensus and guidelines. Osteoarthritis Cartilage. 2008;16:137-162.
13. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part III. Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthritis Cartilage. 2010;18:476-499.
14. Richmond J, Hunter D, Irrgang J, et al. Treatment of osteoarthritis of the knee (nonarthroscopy). J Am Acad Orthop Surg. 2009;17:591-600.
15. Coleman S, Briffa K, Conroy H, et al. Short and medium-term effects of an education self-management program for individuals with osteoarthritis of the knee, designed and delivered by health professionals: a quality assurance study. BMC Musculoskelet Disord. 2008;9:117.
16. Gutekunst DJ, Davis C, DeVita P. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005; 52:2026-2032.
17. Bartlett SJ, Haaz S, Wrobleski P, et al. Small weight losses can yield significant improvements in knee OA symptoms. Arthritis Rheum. 2004;50(suppl):S658.
18. Christensen R, Astrup A, Bliddal H. Weight loss: the treatment of choice for osteoarthritis? A randomized trial. Osteoarthritis Cartilage. 2005;13:20-27.
19. Williams FMK, Skinner J, Spector TD, et al. Dietary garlic and hip osteoarthritis: evidence of a protective effect and putative mechanism of action. BMC Musculoskelet Disord. 2010;11:280.
20. Lin J, Zhang W, Jones A, et al. Efficacy of topical non-steroidal anti-inflammatory drugs in the treatment of osteoarthritis: Meta-analysis of randomized controlled trials. BMJ. 2004;329:324.
21. Stitik TP, Altschuler E, Foye PM. Pharmacotherapy of osteoarthritis. Am J Phys Med Rehabil. 2008; 85(suppl):S15-S28.
22. Bannuru RR, Natov NS, Dasi UR, et al. Therapeutic trajectory following intra-articular hyaluronic acid injection in knee osteoarthritis—meta-analysis.Osteoarthritis Cartilage. 2011;19:611-619.
23. Karlsson J, Sjogren LS, Lohmander LS. Comparison of two hyaluronan drugs and placebo in patients with knee osteoarthritis. A controlled, randomized, double-blind, parallel-design multicentre study. Rheumatology. 2002;41:1240-1248.
24. Modawal A, Ferrer M, Choi HK, et al. Hyaluronic acid injections relieve knee pain. J Fam Pract. 2005;54:758-677.
25. Manheimer E, Lind K, Lao L, et al. Meta-analysis: Acupuncture for osteoarthritis of the knee. Ann Intern Med. 2007;146:868-877.
26. Witt C, Brinkhaus B, Jena S, et al. Acupuncture in patients with osteoarthritis of the knee: a randomised trial. Lancet. 2005;366:136-143.
27. Berman BM, Lao L, Langenberg P, et al. Effectiveness of acupuncture as adjunctive therapy in osteoarthritis of the knee: a randomized, controlled trial. Ann Intern Med. 2004;141:901-910.
28. Kabat-Zinn J. An outpatient program in behavioral medicine for chronic pain patients based on the practice of mindfulness meditation: theoretical considerations and preliminary results. Gen Hosp Psychiatry. 1982;4:33-47.
29. Zeidan F, Martucci KT, Kraft RA, et al. Brain mechanisms supporting the modulation of pain by mindfulness meditation. J Neurosci. 2011;31:5540-5548.
30. Holzel BK, Ott U, Gard T, et al. Investigation of mindfulness meditation practitioners with voxel-based morphometry: Soc Cogn Affect Neurosci. 2008;3:55-61
31. Grossman P, Niemann L, Schmidt S, et al. Mindfulness-based stress reduction and health benefits: a meta-analysis. Psychiatry Res. 2004;57:35-43.
32. Pollard H, Ward G, Hoskins W, et al. The effect of a manual knee protocol on osteoarthritic knee pain: a randomised controlled trial. J Can Chiropr Assoc. 2008;52:229-242.
33. Brantingham JW, Globe G, Pollard H, et al. Manipulative therapy for lower extremity conditions: expansion of literature review. J Manipulative Physiol Ther. 2009;32:53-71.
34. Rutjes AW, Nuesch E, Sterchi R, et al. Transcutaneous electrostimulation for osteoarthritis of the knee. Cochrane Database Syst Rev. 2009;(4):CD002823.
35. Pietrosimone BG, Saliba SA, Hart JM, et al. Effects of transcutaneous electrical nerve stimulation and therapeutic exercise on quadriceps activation in people with tibiofemoral osteoarthritis. J Orthop Sports Phys Ther. 2011;41:4-12.
36. Wang C, Collet JP, Lau J. The effect of Tai Chi on health outcomes in patients with chronic conditions: a systematic review. Arch Intern Med. 2004;164:493-501.
37. Lee MS, Pittler MH, Ernst E. Tai Chi for osteoarthritis: a systematic review. Clin Rheumatol. 2008;27:211-218.
38. Wang C, Schmid CH, Hibberd PL, et al. Tai chi is effective in treating knee osteoarthritis: a randomized controlled trial. Arthritis Rheum. 2009;61:1545-1553.
39. Ramsey SD, Spenser AC, Topolski TD, et al. Use of alternative therapies by older adults with osteoarthritis. Arthritis Care Res. 2001;45:222-227.
40. Perlman AI, Sabina A, Williams AL, et al. Massage therapy for osteoarthritis of the knee: a randomized controlled trial. Arch Intern Med. 2006;166:2533-2538.
41. Ko GD, Nowacki NB, Arseneau L, et al. Omega-3 fatty acids for neuropathic pain: case series. Clin J Pain. 2010;26:168-172.
42. Goldberg RJ, Katz J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain. 2007;129:210-223.
43. McAlindon TE, LaValley MP, Gulin JP, et al. Glucosamine and chondroitin for treatment of osteoarthritis: a systematic quality assessment and meta-analysis. JAMA.2000;283:1469-1475.
44. Wandel S, Jüni P, Tendal B, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: Network meta-analysis. BMJ. 2010;341:c4675.
45. Sawitzke AD, Shi H, Finco MF, et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann Rheum Dis. 2010;69:1459-1464.
46. Berman B. A 60-year-old woman considering acupuncture for knee pain. JAMA. 2007;297:1697-1707.
47. Arjmandi BH, Khalil DA, Lucas EA, et al. Soy protein may alleviate osteoarthritis symptoms. Phytomedicine. 2004;11:567-575.
48. Maheu E, Mazieres B, Valat JP, et al. Symptomatic efficacy of avocado/soybean unsaponifiables in the treatment of osteoarthritis of the knee and hip: a prospective, randomized, double-blind, placebo-controlled, multicenter clinical trial with a six-month treatment period and a two-month follow-up demonstrating a persistent effect. Arthritis Rheum. 1998;41:81-91.
49. Maroon JC, Bost JW, Maroon A. Natural anti-inflammatory agents for pain relief. Surg Neurol Int. 2010;1:80.
50. Ahmed S, Wang N, Lalonde M, et al. Green tea polyphenol epigallocatechin-3-gallate (EGCG) differentially inhibits interleukin-1 beta-induced expression of matrix metalloproteinase-1 and -13 in human chondrocytes. J Pharmacol Exp Ther. 2004;308:767-773.
51. Funk JL, Oyarzo JN, Frye JB, et al. Turmeric extracts containing curcuminoids prevent experimental rheumatoid arthritis. J Nat Prod. 2006;69:351-355.
52. Altman RD, Marcussen KC. Effects of a ginger extract on knee pain in patients with osteoarthritis. Arthritis Rheum. 2001;44:2531-2538.
53. Cardile V, Panico A, Gentile B, et al. Effect of propolis on human cartilage and chondrocytes. Life Sci. 2003;73:1027-1035.
54. Ng NT, Heesch KC, Brown WJ. Efficacy of a progressive walking program and glucosamine sulphate supplementation on osteoarthritic symptoms of the hip and knee: a feasibility trial. Arthritis Res Ther. 2010;12:R25.
At last count in 2008, 27 million Americans were suffering with osteoarthritis (OA), by far the most common form of arthritis.1 That number has undoubtedly risen and will continue to do so as Baby Boomers age. Despite the benefits of conventional nonpharmacologic measures and available pharmacologic agents, many patients with OA achieve less than satisfactory pain relief and have impaired joint mobility, which can significantly limit their daily activities.2 Numerous studies have found that a host of complementary and alternative (CAM) therapies can provide safe and effective pain relief and enhanced joint mobility. The National Center for Complementary and Alternative Therapies found that musculoskeletal problems such as back pain, neck pain, joint pain, and arthritis were the top conditions for which adults used CAM therapies in 2007 [Figure 1].3
| FIGURE 1: Diseases/conditions for which CAM is most frequently used among US adults – 2007 |

| Source: Barnes PM, Bloom B, Nahin R. CDC National Health Statistics Report #12. Complementary and alternative medicine use amongadults and children. United States, 2007. December 2008. |
A survey to gather detailed information about CAM use by patients with arthritis (n 5 2140) found that most patients followed by specialists (90.5%) and a slightly smaller percentage followed by primary care physicians (82.8%) had tried at least one CAM therapy for relief of OA symptoms. The authors suggested that the higher percentage for specialist care may be because these patients have more severe disease and therefore experience more pain.4 An understanding of these therapies will allow primary care and specialist physicians to better communicate with and advise patients who seek options outside the usual spectrum of care.
Definition and therapeutic goals
OA is a progressive deterioration of joint tissues.5 A decrease in protective proteoglycans and collagen compromises joint cartilage.5-8 Deterioration of cartilage in turn leads to bone erosions, osteophyte formation, and bone restructuring. Inflammation, too, may result in reaction to cartilage degradation byproducts entering synovial spaces.6,7 Joint pain aggravated by physical activity and alleviated by rest is typical of OA. Also common are joint instability and stiffness upon rising in the morning or after extended inactivity.9 A patient’s history may additionally reveal that the level of pain experienced with activity has steadily increased with time. Physical examination may reveal bony enlargement or deformity of involved joints, crepitus, and restricted range of motion.9 The value of laboratory and radiology studies lies mainly in ruling out alternative diagnoses.
The goals of OA treatment are pain relief and preservation of joint function. Because the experience of pain is influenced by physical, psychological, and emotional factors, individuals vary in how they respond to specific therapies and in how they wish to achieve pain relief. Some patients may experience side effects from anti-inflammatory pain medications.10 Others may be hesitant to resort to surgery.9 All major guidelines agree that, for most patients, therapy combining nonpharmacologic measures and pharmacologic agents is required to achieve optimal relief of pain and preservation of joint mobility.11-14
Conventional treatment options for OA
Selecting appropriate treatment begins with consideration of the patient’s report of chronic pain and limitations in ambulation or other activities. Also important is assessment of the patient’s level of pain on manipulation, as well as muscle strength and ligament stability.7,8 Depending on physical examination results, a reasonable approach may be to start with nonpharmacologic measures and add pharmacologic agents in a stepwise manner to control pain.8 Self-management programs have been shown to improve symptoms as well as quality of life, and should be incorporated into the treatment plan.15
Nonpharmacologic measures, prescribed as needed for each individual, include weight loss for those who are overweight or obese.11-14 Weight loss has been shown to improve mobility and reduce pain. For every one pound of weight lost, there is a 4-pound reduction in the load exerted on the knee for each step taken during daily activities.16 A weight loss of only 15 pounds can cut knee pain in half for overweight individuals with arthritis.17 A low-carbohydrate diet has been shown to reduce weight in obese patients by ≥10% and lead to improvements in self-reported scores for overall progress and functional ability.18 A diet of fruits and vegetables (including alliums and cruciferous vegetables) that is high in phytonutrients has been shown to have a protective effect in patients with hip OA.19
Other measures are physical and occupational therapies, assistive devices for walking or accomplishing other daily tasks, and joint taping.11-14 Patient (and family) education regarding the progressive nature of OA is crucial to bolstering patient resolve in following through with self-management activities.15 Healthcare professionals can provide factual, disease-specific information on some effective self-management strategies for use between office visits that yield short- and long-term benefits. Self-management strategies can incorporate pain management education; joint-sparing exercise advice including daily walking, balance tips, and falls prevention; and emotional and cognitive skills to improve quality of life.
Pharmacologic intervention is best begun with the least risky agent, at the lowest effective dose, for the shortest possible duration.12 For mild-to-moderate pain, the Osteoarthritis Research Society International recommends acetaminophen as initial therapy, given up to 4 g/d. If pain relief is insufficient, consider adding or switching to an oral or topical non-steroidal anti-inflammatory drug (NSAID), keeping in mind the possibility for gastrointestinal (GI) adverse effects with oral agents. When using an oral NSAID in patients at risk for GI effects, give a proton-pump inhibitor (PPI) as well. A topical NSAID or capsaicin may provide additional local pain relief.20
If pain remains inadequately controlled, or if a patient is intolerant to NSAIDs, a selective COX-2 inhibitor given alone or in
combination with acetaminophen may be an appropriate choice—provided the patient undergoes thorough evaluation for cardiovascular (CV) and GI risks.12,21 For those with GI risks taking a COX-2 inhibitor, give either misoprostol or a PPI concomitantly. Monitor those with CV risks closely.
When OA pain is severe or unresponsive to the previous medications, alternative agents include tramadol with or without acetaminophen, or opioid analgesics. In either instance, start at the lowest possible dosage (adjusted, as needed, for renal or hepatic impairment) and then titrate upward slowly to minimize adverse effects.9,12
Evidence-based complementary treatment options for OA
In its executive guidelines for US hospitals, the Joint Commission clearly states that institutions seeking accreditation, or wishing to maintain it, must include in its options for treating chronic pain such complementary measures as massage, acupuncture, and mind-body therapy. The Joint Commission’s basis for mandating an integrative approach to pain control is the growing body of clinical evidence supporting the efficacy of complementary treatments. In addition, many patients prefer nonpharmacologic options for personal reasons.
Hyaluronic acid. In a recently published meta-analysis of studies looking at intra-articular hyaluronic acid (IAHA) for knee OA pain, the authors reported therapeutic effectiveness beginning at 4 weeks, reaching a peak effect at 8 weeks, and exerting a residual effect still detectable at 24 weeks.22 Moreover, the peak effect of IAHA is greater than published effects for acetaminophen, NSAIDs, and COX-2 inhibitors.22 IAHA could be useful in combination with other therapies, and in certain clinical situations such as when patients are undecided about surgery. Other studies and meta-analyses of IAHA have yielded different results regarding length of efficacy.23,24
Acupuncture. While studies of acupuncture for OA have varied in quality and duration of effect, many show benefits. One meta-analysis found that patients who received acupuncture reported clinically relevant short- and long-term pain reduction and improved function compared with control patients who received usual care.25 In a study of
294 patients with OA of the knee, patients received 8 weeksof acupuncture, minimal acupuncture (superficial needling at nonacupuncture points), or no treatment. Based on the Western Ontario and McMasters Universities Osteoarthritis (WOMAC) questionnaire assessing pain, function, and stiffness due to OA, those who received acupuncture had significantly better scores than the other 2 groups. However, after 26 and 52 weeks, the differences between acupuncture and minimal acupuncture were no longer significant.26
In the largest study ever conducted on acupuncture for OA of the knee, 570 patients were randomly assigned to receive acupuncture, sham acupuncture, or education.27 At
26 weeks postintervention, based on the WOMAC function score those who underwent true acupuncture experienced significantly greater pain relief and functional improvement than those who received sham acupuncture or education only.
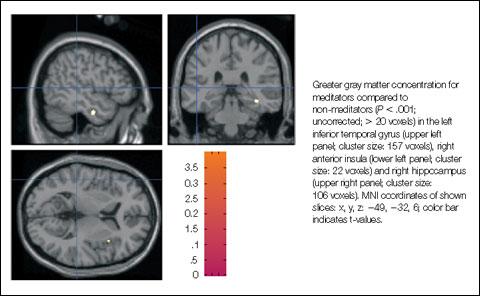
Mindfulness-Based Stress Reduction (MBSR), pioneered by Jon Kabat-Zinn, PhD, uses mindfulness meditation to alleviate chronic pain. First reported in a 1982 study, 88% of enrolled patients with chronic pain of 6 months to 48 years’ duration who were trained in stress reduction using mindfulness meditation perceived a decrease in pain of at least 50%.28 This method of focusing on the mind and body does not simply “distract” patients from their pain but achieves measurable changes in how pain is perceived. With MBSR, pain reduction reported by patients was significantly greater than that achieved with morphine and other drugs.29 Brain scans show that, after MBSR, regions normally associated with processing painful stimuli become quiescent.29 MBSR has also been shown in imaging studies to increase gray matter concentrations in regions of the brain associated with learning and memory, regulation of emotion, and self-referential processing [Figure 2].30
| FIGURE 2: Gray matter is more concentrated for persons who meditate compared with nonmeditators. |

| Source: Holzel BK, et al. Soc Cogn Affect Neurosci. 2008;3:55-61 |
A 2004 meta-analysis of 20 reports on mindfulness meditation that met study criteria included patients with anxiety, cancer diagnoses, chronic pain, coronary artery disease, depression, fibromyalgia, obesity and binge eating disorders, and psychiatric disorders. The analysis found a relatively consistent and strong effect, suggesting that mindfulness training may enhance the ability to cope with distress and disability and the sense of physical well-being.31
Chiropractic. One small randomized controlled trial (RCT) matched participants with knee OA pain measured by visual analog scale (VAS) and assigned them to receive either treatment according to the Macquarie Injury Management Group Knee Protocol or nonforceful contact.32 The treatment group reported significantly decreased knee symptoms such as crepitus and improvement in mobility and ability to perform general activities. A systematic review found fair evidence for efficacy of manipulative therapy of the knee combined with multimodal or exercise therapy for knee OA.33
Transcutaneous electrical nerve stimulation (TENS). Evidence for the effectiveness of TENS for OA is mixed. A 2009 Cochrane systematic review of TENS for knee OA concluded that available studies were too small and poorly designed to judge its effectiveness.34 However, its use in tibiofemoral OA has yielded good results.35
Tai Chi. This ancient form of meditative exercise has long been acknowledged as a means for enhancing balance and dexterity, but studies of chronic pain reduction and improved mobility with Tai Chi have been few and usually underpowered to yield reliable results. One systematic review of 9 RCTs, 23 non-RCTs, and 15 observational studies included studies that showed benefits for osteoarthritic symptoms with improved functional mobility and quality of life.36 Another systematic review of 5 RCTs and 7 nonrandomized clinical trials found that Tai Chi was effective for pain control of OA of the knee, but that evidence was inconclusive for pain reduction or mobility improvement.37 In a long-term study comparing Tai Chi with regular exercise in patients with symptomatic OA of the knee, researchers found that those in the Tai Chi group showed greater improvement in pain, physical function, self-efficacy, depression, and health-related quality of life, with some improvements lasting as long as 48 weeks.38
Massage. A longitudinal analysis of the use of a variety of alternative therapy offerings by older adults with OA found that the most commonly utilized treatment during a 20-week intervention period was massage (57%) followed by chiropractic (20.7%).39 In the first prospective randomized trial to evaluate the efficacy of massage for adults with OA of the knee, participants who received 8 weeks of Swedish massage therapy showed significant improvements in pain, stiffness, and physical function. These improvements persisted at the 16-week evaluation.40
Dietary supplements. Omega-3 fatty acids have shown promise in reducing chronic neuropathic pain when taken at levels that exceed 2 g/d of eicosapetaenoic acid and docosahexaenoic acid.41 This effect is probably enhanced when combined with a dietary reduction of omega-6 fatty acids and saturated fats. This dietary measure may also have some effect on joint pain.42
Glucosamine/chondroitin has been shown in a meta-analysis to reduce OA pain,43 although another meta-analysis showed no effect on either joint pain or narrowing of joint space.44 More recently, data from the long-term Glucosamine/Chondroitin Arthritis Intervention (GAIT) trial found that patients who took glucosamine or glucosamine/chondroitin in combination had similar results to those who took celecoxib or placebo. Over the 2-year study period, all groups showed improvement in painand function.45
It is worth noting that not all supplements are made the same, and there may be varying potencies. For example, some glucosamine products work better than others.46 When discussing supplements, to get the most potential benefit it is advisable to suggest those with a strong body of evidence or refer patients to providers who are familiar with CAM therapies.
Other promising dietary aids for reducing OA pain include soy protein,47 avocado-soybean unsaponifiables,48 Cat’s claw,49 white willow,49 green tea,49,50 turmeric,51 ginger,52 and propolis.53
Widening your integrative approach to OA treatment
There is emerging evidence that integrating multiple conventional and CAM therapies such as glucosamine and walking may provide the best results for OA patients.54 The realization that many patients with pain and diminished mobility are already exploring CAM therapies presents an opportunity for you to discuss their decisions and direct their attention to options that are supported by strong evidence. By evaluating the increasing body of evidence in support of specific CAM therapies, you can feel confident in offering your patients a wider range of choices than standard pharmacologic and nonpharmacologic OA options, and integrate these options to improve care.
Acknowledgement—The author wishes to thank Charles F. Williams for his assistance in researching and writing this article.
REFERENCES
1. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35.
2. Centers for Disease Control and Prevention. Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation—United States, 2007–2009. MMWR Morb Mortal Wkly Rep. 2010;59:1261-1265.
3. Barnes PM, Bloom B, Nahin R. CDC National Health Statistics Report #12. Complementary and alternative medicine use among adults and children: United States, 2007. December 2008. Available at: http://nccam.nih.gov/news/camstats/2007/camsurvey_fs1.htm#use.
4. Callahan LF, Wiley-Exley EK, Mielenz TJ, et al. Use of complementary and alternative medicine among patients with arthritis. Prev Chronic Dis. 2009;6:1-23.
5. Felson DT. An update on the pathogenesis and epidemiology of osteoarthritis. Radiol Clin North Am. 2004;42:1-9.
6. Breedveld FC. Osteoarthritis—the impact of a serious disease. Rheumatology. 2004;43(suppl 1):i4-i8.
7. Urquhart N, Stanish WD. Drugs for knee osteoarthritis: what primary care physicians need to know—a clear understanding of the symptoms and treatment options is required. J Musculoskel Med. 2007;24:178-187.
8. Hunter DJ. In the clinic: osteoarthritis. Arch Intern Med. 2007;147:ITC8-1–ITC8-16.
9. American College of Rheumatology. Recommendations for the medical management of osteoarthritisof the hip and knee. 2000. Available at: http://www.rheumatology.org/practice/clinical/guidelines/oa-mgmt.asp.
10. Tramer MR, Moore RA, Reynolds DJ, et al. Quantitative estimation of rare adverse events which follow a biological progression: a new model applied to chronic NSAID use. Pain. 2000;85:169-182.
11. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part I. Critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage. 2007;15:981-1000.
12. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part II. OARSI evidence-based, expert consensus and guidelines. Osteoarthritis Cartilage. 2008;16:137-162.
13. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part III. Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthritis Cartilage. 2010;18:476-499.
14. Richmond J, Hunter D, Irrgang J, et al. Treatment of osteoarthritis of the knee (nonarthroscopy). J Am Acad Orthop Surg. 2009;17:591-600.
15. Coleman S, Briffa K, Conroy H, et al. Short and medium-term effects of an education self-management program for individuals with osteoarthritis of the knee, designed and delivered by health professionals: a quality assurance study. BMC Musculoskelet Disord. 2008;9:117.
16. Gutekunst DJ, Davis C, DeVita P. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005; 52:2026-2032.
17. Bartlett SJ, Haaz S, Wrobleski P, et al. Small weight losses can yield significant improvements in knee OA symptoms. Arthritis Rheum. 2004;50(suppl):S658.
18. Christensen R, Astrup A, Bliddal H. Weight loss: the treatment of choice for osteoarthritis? A randomized trial. Osteoarthritis Cartilage. 2005;13:20-27.
19. Williams FMK, Skinner J, Spector TD, et al. Dietary garlic and hip osteoarthritis: evidence of a protective effect and putative mechanism of action. BMC Musculoskelet Disord. 2010;11:280.
20. Lin J, Zhang W, Jones A, et al. Efficacy of topical non-steroidal anti-inflammatory drugs in the treatment of osteoarthritis: Meta-analysis of randomized controlled trials. BMJ. 2004;329:324.
21. Stitik TP, Altschuler E, Foye PM. Pharmacotherapy of osteoarthritis. Am J Phys Med Rehabil. 2008; 85(suppl):S15-S28.
22. Bannuru RR, Natov NS, Dasi UR, et al. Therapeutic trajectory following intra-articular hyaluronic acid injection in knee osteoarthritis—meta-analysis.Osteoarthritis Cartilage. 2011;19:611-619.
23. Karlsson J, Sjogren LS, Lohmander LS. Comparison of two hyaluronan drugs and placebo in patients with knee osteoarthritis. A controlled, randomized, double-blind, parallel-design multicentre study. Rheumatology. 2002;41:1240-1248.
24. Modawal A, Ferrer M, Choi HK, et al. Hyaluronic acid injections relieve knee pain. J Fam Pract. 2005;54:758-677.
25. Manheimer E, Lind K, Lao L, et al. Meta-analysis: Acupuncture for osteoarthritis of the knee. Ann Intern Med. 2007;146:868-877.
26. Witt C, Brinkhaus B, Jena S, et al. Acupuncture in patients with osteoarthritis of the knee: a randomised trial. Lancet. 2005;366:136-143.
27. Berman BM, Lao L, Langenberg P, et al. Effectiveness of acupuncture as adjunctive therapy in osteoarthritis of the knee: a randomized, controlled trial. Ann Intern Med. 2004;141:901-910.
28. Kabat-Zinn J. An outpatient program in behavioral medicine for chronic pain patients based on the practice of mindfulness meditation: theoretical considerations and preliminary results. Gen Hosp Psychiatry. 1982;4:33-47.
29. Zeidan F, Martucci KT, Kraft RA, et al. Brain mechanisms supporting the modulation of pain by mindfulness meditation. J Neurosci. 2011;31:5540-5548.
30. Holzel BK, Ott U, Gard T, et al. Investigation of mindfulness meditation practitioners with voxel-based morphometry: Soc Cogn Affect Neurosci. 2008;3:55-61
31. Grossman P, Niemann L, Schmidt S, et al. Mindfulness-based stress reduction and health benefits: a meta-analysis. Psychiatry Res. 2004;57:35-43.
32. Pollard H, Ward G, Hoskins W, et al. The effect of a manual knee protocol on osteoarthritic knee pain: a randomised controlled trial. J Can Chiropr Assoc. 2008;52:229-242.
33. Brantingham JW, Globe G, Pollard H, et al. Manipulative therapy for lower extremity conditions: expansion of literature review. J Manipulative Physiol Ther. 2009;32:53-71.
34. Rutjes AW, Nuesch E, Sterchi R, et al. Transcutaneous electrostimulation for osteoarthritis of the knee. Cochrane Database Syst Rev. 2009;(4):CD002823.
35. Pietrosimone BG, Saliba SA, Hart JM, et al. Effects of transcutaneous electrical nerve stimulation and therapeutic exercise on quadriceps activation in people with tibiofemoral osteoarthritis. J Orthop Sports Phys Ther. 2011;41:4-12.
36. Wang C, Collet JP, Lau J. The effect of Tai Chi on health outcomes in patients with chronic conditions: a systematic review. Arch Intern Med. 2004;164:493-501.
37. Lee MS, Pittler MH, Ernst E. Tai Chi for osteoarthritis: a systematic review. Clin Rheumatol. 2008;27:211-218.
38. Wang C, Schmid CH, Hibberd PL, et al. Tai chi is effective in treating knee osteoarthritis: a randomized controlled trial. Arthritis Rheum. 2009;61:1545-1553.
39. Ramsey SD, Spenser AC, Topolski TD, et al. Use of alternative therapies by older adults with osteoarthritis. Arthritis Care Res. 2001;45:222-227.
40. Perlman AI, Sabina A, Williams AL, et al. Massage therapy for osteoarthritis of the knee: a randomized controlled trial. Arch Intern Med. 2006;166:2533-2538.
41. Ko GD, Nowacki NB, Arseneau L, et al. Omega-3 fatty acids for neuropathic pain: case series. Clin J Pain. 2010;26:168-172.
42. Goldberg RJ, Katz J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain. 2007;129:210-223.
43. McAlindon TE, LaValley MP, Gulin JP, et al. Glucosamine and chondroitin for treatment of osteoarthritis: a systematic quality assessment and meta-analysis. JAMA.2000;283:1469-1475.
44. Wandel S, Jüni P, Tendal B, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: Network meta-analysis. BMJ. 2010;341:c4675.
45. Sawitzke AD, Shi H, Finco MF, et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann Rheum Dis. 2010;69:1459-1464.
46. Berman B. A 60-year-old woman considering acupuncture for knee pain. JAMA. 2007;297:1697-1707.
47. Arjmandi BH, Khalil DA, Lucas EA, et al. Soy protein may alleviate osteoarthritis symptoms. Phytomedicine. 2004;11:567-575.
48. Maheu E, Mazieres B, Valat JP, et al. Symptomatic efficacy of avocado/soybean unsaponifiables in the treatment of osteoarthritis of the knee and hip: a prospective, randomized, double-blind, placebo-controlled, multicenter clinical trial with a six-month treatment period and a two-month follow-up demonstrating a persistent effect. Arthritis Rheum. 1998;41:81-91.
49. Maroon JC, Bost JW, Maroon A. Natural anti-inflammatory agents for pain relief. Surg Neurol Int. 2010;1:80.
50. Ahmed S, Wang N, Lalonde M, et al. Green tea polyphenol epigallocatechin-3-gallate (EGCG) differentially inhibits interleukin-1 beta-induced expression of matrix metalloproteinase-1 and -13 in human chondrocytes. J Pharmacol Exp Ther. 2004;308:767-773.
51. Funk JL, Oyarzo JN, Frye JB, et al. Turmeric extracts containing curcuminoids prevent experimental rheumatoid arthritis. J Nat Prod. 2006;69:351-355.
52. Altman RD, Marcussen KC. Effects of a ginger extract on knee pain in patients with osteoarthritis. Arthritis Rheum. 2001;44:2531-2538.
53. Cardile V, Panico A, Gentile B, et al. Effect of propolis on human cartilage and chondrocytes. Life Sci. 2003;73:1027-1035.
54. Ng NT, Heesch KC, Brown WJ. Efficacy of a progressive walking program and glucosamine sulphate supplementation on osteoarthritic symptoms of the hip and knee: a feasibility trial. Arthritis Res Ther. 2010;12:R25.
At last count in 2008, 27 million Americans were suffering with osteoarthritis (OA), by far the most common form of arthritis.1 That number has undoubtedly risen and will continue to do so as Baby Boomers age. Despite the benefits of conventional nonpharmacologic measures and available pharmacologic agents, many patients with OA achieve less than satisfactory pain relief and have impaired joint mobility, which can significantly limit their daily activities.2 Numerous studies have found that a host of complementary and alternative (CAM) therapies can provide safe and effective pain relief and enhanced joint mobility. The National Center for Complementary and Alternative Therapies found that musculoskeletal problems such as back pain, neck pain, joint pain, and arthritis were the top conditions for which adults used CAM therapies in 2007 [Figure 1].3
| FIGURE 1: Diseases/conditions for which CAM is most frequently used among US adults – 2007 |

| Source: Barnes PM, Bloom B, Nahin R. CDC National Health Statistics Report #12. Complementary and alternative medicine use amongadults and children. United States, 2007. December 2008. |
A survey to gather detailed information about CAM use by patients with arthritis (n 5 2140) found that most patients followed by specialists (90.5%) and a slightly smaller percentage followed by primary care physicians (82.8%) had tried at least one CAM therapy for relief of OA symptoms. The authors suggested that the higher percentage for specialist care may be because these patients have more severe disease and therefore experience more pain.4 An understanding of these therapies will allow primary care and specialist physicians to better communicate with and advise patients who seek options outside the usual spectrum of care.
Definition and therapeutic goals
OA is a progressive deterioration of joint tissues.5 A decrease in protective proteoglycans and collagen compromises joint cartilage.5-8 Deterioration of cartilage in turn leads to bone erosions, osteophyte formation, and bone restructuring. Inflammation, too, may result in reaction to cartilage degradation byproducts entering synovial spaces.6,7 Joint pain aggravated by physical activity and alleviated by rest is typical of OA. Also common are joint instability and stiffness upon rising in the morning or after extended inactivity.9 A patient’s history may additionally reveal that the level of pain experienced with activity has steadily increased with time. Physical examination may reveal bony enlargement or deformity of involved joints, crepitus, and restricted range of motion.9 The value of laboratory and radiology studies lies mainly in ruling out alternative diagnoses.
The goals of OA treatment are pain relief and preservation of joint function. Because the experience of pain is influenced by physical, psychological, and emotional factors, individuals vary in how they respond to specific therapies and in how they wish to achieve pain relief. Some patients may experience side effects from anti-inflammatory pain medications.10 Others may be hesitant to resort to surgery.9 All major guidelines agree that, for most patients, therapy combining nonpharmacologic measures and pharmacologic agents is required to achieve optimal relief of pain and preservation of joint mobility.11-14
Conventional treatment options for OA
Selecting appropriate treatment begins with consideration of the patient’s report of chronic pain and limitations in ambulation or other activities. Also important is assessment of the patient’s level of pain on manipulation, as well as muscle strength and ligament stability.7,8 Depending on physical examination results, a reasonable approach may be to start with nonpharmacologic measures and add pharmacologic agents in a stepwise manner to control pain.8 Self-management programs have been shown to improve symptoms as well as quality of life, and should be incorporated into the treatment plan.15
Nonpharmacologic measures, prescribed as needed for each individual, include weight loss for those who are overweight or obese.11-14 Weight loss has been shown to improve mobility and reduce pain. For every one pound of weight lost, there is a 4-pound reduction in the load exerted on the knee for each step taken during daily activities.16 A weight loss of only 15 pounds can cut knee pain in half for overweight individuals with arthritis.17 A low-carbohydrate diet has been shown to reduce weight in obese patients by ≥10% and lead to improvements in self-reported scores for overall progress and functional ability.18 A diet of fruits and vegetables (including alliums and cruciferous vegetables) that is high in phytonutrients has been shown to have a protective effect in patients with hip OA.19
Other measures are physical and occupational therapies, assistive devices for walking or accomplishing other daily tasks, and joint taping.11-14 Patient (and family) education regarding the progressive nature of OA is crucial to bolstering patient resolve in following through with self-management activities.15 Healthcare professionals can provide factual, disease-specific information on some effective self-management strategies for use between office visits that yield short- and long-term benefits. Self-management strategies can incorporate pain management education; joint-sparing exercise advice including daily walking, balance tips, and falls prevention; and emotional and cognitive skills to improve quality of life.
Pharmacologic intervention is best begun with the least risky agent, at the lowest effective dose, for the shortest possible duration.12 For mild-to-moderate pain, the Osteoarthritis Research Society International recommends acetaminophen as initial therapy, given up to 4 g/d. If pain relief is insufficient, consider adding or switching to an oral or topical non-steroidal anti-inflammatory drug (NSAID), keeping in mind the possibility for gastrointestinal (GI) adverse effects with oral agents. When using an oral NSAID in patients at risk for GI effects, give a proton-pump inhibitor (PPI) as well. A topical NSAID or capsaicin may provide additional local pain relief.20
If pain remains inadequately controlled, or if a patient is intolerant to NSAIDs, a selective COX-2 inhibitor given alone or in
combination with acetaminophen may be an appropriate choice—provided the patient undergoes thorough evaluation for cardiovascular (CV) and GI risks.12,21 For those with GI risks taking a COX-2 inhibitor, give either misoprostol or a PPI concomitantly. Monitor those with CV risks closely.
When OA pain is severe or unresponsive to the previous medications, alternative agents include tramadol with or without acetaminophen, or opioid analgesics. In either instance, start at the lowest possible dosage (adjusted, as needed, for renal or hepatic impairment) and then titrate upward slowly to minimize adverse effects.9,12
Evidence-based complementary treatment options for OA
In its executive guidelines for US hospitals, the Joint Commission clearly states that institutions seeking accreditation, or wishing to maintain it, must include in its options for treating chronic pain such complementary measures as massage, acupuncture, and mind-body therapy. The Joint Commission’s basis for mandating an integrative approach to pain control is the growing body of clinical evidence supporting the efficacy of complementary treatments. In addition, many patients prefer nonpharmacologic options for personal reasons.
Hyaluronic acid. In a recently published meta-analysis of studies looking at intra-articular hyaluronic acid (IAHA) for knee OA pain, the authors reported therapeutic effectiveness beginning at 4 weeks, reaching a peak effect at 8 weeks, and exerting a residual effect still detectable at 24 weeks.22 Moreover, the peak effect of IAHA is greater than published effects for acetaminophen, NSAIDs, and COX-2 inhibitors.22 IAHA could be useful in combination with other therapies, and in certain clinical situations such as when patients are undecided about surgery. Other studies and meta-analyses of IAHA have yielded different results regarding length of efficacy.23,24
Acupuncture. While studies of acupuncture for OA have varied in quality and duration of effect, many show benefits. One meta-analysis found that patients who received acupuncture reported clinically relevant short- and long-term pain reduction and improved function compared with control patients who received usual care.25 In a study of
294 patients with OA of the knee, patients received 8 weeksof acupuncture, minimal acupuncture (superficial needling at nonacupuncture points), or no treatment. Based on the Western Ontario and McMasters Universities Osteoarthritis (WOMAC) questionnaire assessing pain, function, and stiffness due to OA, those who received acupuncture had significantly better scores than the other 2 groups. However, after 26 and 52 weeks, the differences between acupuncture and minimal acupuncture were no longer significant.26
In the largest study ever conducted on acupuncture for OA of the knee, 570 patients were randomly assigned to receive acupuncture, sham acupuncture, or education.27 At
26 weeks postintervention, based on the WOMAC function score those who underwent true acupuncture experienced significantly greater pain relief and functional improvement than those who received sham acupuncture or education only.
Mindfulness-Based Stress Reduction (MBSR), pioneered by Jon Kabat-Zinn, PhD, uses mindfulness meditation to alleviate chronic pain. First reported in a 1982 study, 88% of enrolled patients with chronic pain of 6 months to 48 years’ duration who were trained in stress reduction using mindfulness meditation perceived a decrease in pain of at least 50%.28 This method of focusing on the mind and body does not simply “distract” patients from their pain but achieves measurable changes in how pain is perceived. With MBSR, pain reduction reported by patients was significantly greater than that achieved with morphine and other drugs.29 Brain scans show that, after MBSR, regions normally associated with processing painful stimuli become quiescent.29 MBSR has also been shown in imaging studies to increase gray matter concentrations in regions of the brain associated with learning and memory, regulation of emotion, and self-referential processing [Figure 2].30
| FIGURE 2: Gray matter is more concentrated for persons who meditate compared with nonmeditators. |

| Source: Holzel BK, et al. Soc Cogn Affect Neurosci. 2008;3:55-61 |
A 2004 meta-analysis of 20 reports on mindfulness meditation that met study criteria included patients with anxiety, cancer diagnoses, chronic pain, coronary artery disease, depression, fibromyalgia, obesity and binge eating disorders, and psychiatric disorders. The analysis found a relatively consistent and strong effect, suggesting that mindfulness training may enhance the ability to cope with distress and disability and the sense of physical well-being.31
Chiropractic. One small randomized controlled trial (RCT) matched participants with knee OA pain measured by visual analog scale (VAS) and assigned them to receive either treatment according to the Macquarie Injury Management Group Knee Protocol or nonforceful contact.32 The treatment group reported significantly decreased knee symptoms such as crepitus and improvement in mobility and ability to perform general activities. A systematic review found fair evidence for efficacy of manipulative therapy of the knee combined with multimodal or exercise therapy for knee OA.33
Transcutaneous electrical nerve stimulation (TENS). Evidence for the effectiveness of TENS for OA is mixed. A 2009 Cochrane systematic review of TENS for knee OA concluded that available studies were too small and poorly designed to judge its effectiveness.34 However, its use in tibiofemoral OA has yielded good results.35
Tai Chi. This ancient form of meditative exercise has long been acknowledged as a means for enhancing balance and dexterity, but studies of chronic pain reduction and improved mobility with Tai Chi have been few and usually underpowered to yield reliable results. One systematic review of 9 RCTs, 23 non-RCTs, and 15 observational studies included studies that showed benefits for osteoarthritic symptoms with improved functional mobility and quality of life.36 Another systematic review of 5 RCTs and 7 nonrandomized clinical trials found that Tai Chi was effective for pain control of OA of the knee, but that evidence was inconclusive for pain reduction or mobility improvement.37 In a long-term study comparing Tai Chi with regular exercise in patients with symptomatic OA of the knee, researchers found that those in the Tai Chi group showed greater improvement in pain, physical function, self-efficacy, depression, and health-related quality of life, with some improvements lasting as long as 48 weeks.38
Massage. A longitudinal analysis of the use of a variety of alternative therapy offerings by older adults with OA found that the most commonly utilized treatment during a 20-week intervention period was massage (57%) followed by chiropractic (20.7%).39 In the first prospective randomized trial to evaluate the efficacy of massage for adults with OA of the knee, participants who received 8 weeks of Swedish massage therapy showed significant improvements in pain, stiffness, and physical function. These improvements persisted at the 16-week evaluation.40
Dietary supplements. Omega-3 fatty acids have shown promise in reducing chronic neuropathic pain when taken at levels that exceed 2 g/d of eicosapetaenoic acid and docosahexaenoic acid.41 This effect is probably enhanced when combined with a dietary reduction of omega-6 fatty acids and saturated fats. This dietary measure may also have some effect on joint pain.42
Glucosamine/chondroitin has been shown in a meta-analysis to reduce OA pain,43 although another meta-analysis showed no effect on either joint pain or narrowing of joint space.44 More recently, data from the long-term Glucosamine/Chondroitin Arthritis Intervention (GAIT) trial found that patients who took glucosamine or glucosamine/chondroitin in combination had similar results to those who took celecoxib or placebo. Over the 2-year study period, all groups showed improvement in painand function.45
It is worth noting that not all supplements are made the same, and there may be varying potencies. For example, some glucosamine products work better than others.46 When discussing supplements, to get the most potential benefit it is advisable to suggest those with a strong body of evidence or refer patients to providers who are familiar with CAM therapies.
Other promising dietary aids for reducing OA pain include soy protein,47 avocado-soybean unsaponifiables,48 Cat’s claw,49 white willow,49 green tea,49,50 turmeric,51 ginger,52 and propolis.53
Widening your integrative approach to OA treatment
There is emerging evidence that integrating multiple conventional and CAM therapies such as glucosamine and walking may provide the best results for OA patients.54 The realization that many patients with pain and diminished mobility are already exploring CAM therapies presents an opportunity for you to discuss their decisions and direct their attention to options that are supported by strong evidence. By evaluating the increasing body of evidence in support of specific CAM therapies, you can feel confident in offering your patients a wider range of choices than standard pharmacologic and nonpharmacologic OA options, and integrate these options to improve care.
Acknowledgement—The author wishes to thank Charles F. Williams for his assistance in researching and writing this article.
REFERENCES
1. Lawrence RC, Felson DT, Helmick CG, et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part II. Arthritis Rheum. 2008;58:26-35.
2. Centers for Disease Control and Prevention. Prevalence of doctor-diagnosed arthritis and arthritis-attributable activity limitation—United States, 2007–2009. MMWR Morb Mortal Wkly Rep. 2010;59:1261-1265.
3. Barnes PM, Bloom B, Nahin R. CDC National Health Statistics Report #12. Complementary and alternative medicine use among adults and children: United States, 2007. December 2008. Available at: http://nccam.nih.gov/news/camstats/2007/camsurvey_fs1.htm#use.
4. Callahan LF, Wiley-Exley EK, Mielenz TJ, et al. Use of complementary and alternative medicine among patients with arthritis. Prev Chronic Dis. 2009;6:1-23.
5. Felson DT. An update on the pathogenesis and epidemiology of osteoarthritis. Radiol Clin North Am. 2004;42:1-9.
6. Breedveld FC. Osteoarthritis—the impact of a serious disease. Rheumatology. 2004;43(suppl 1):i4-i8.
7. Urquhart N, Stanish WD. Drugs for knee osteoarthritis: what primary care physicians need to know—a clear understanding of the symptoms and treatment options is required. J Musculoskel Med. 2007;24:178-187.
8. Hunter DJ. In the clinic: osteoarthritis. Arch Intern Med. 2007;147:ITC8-1–ITC8-16.
9. American College of Rheumatology. Recommendations for the medical management of osteoarthritisof the hip and knee. 2000. Available at: http://www.rheumatology.org/practice/clinical/guidelines/oa-mgmt.asp.
10. Tramer MR, Moore RA, Reynolds DJ, et al. Quantitative estimation of rare adverse events which follow a biological progression: a new model applied to chronic NSAID use. Pain. 2000;85:169-182.
11. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part I. Critical appraisal of existing treatment guidelines and systematic review of current research evidence. Osteoarthritis Cartilage. 2007;15:981-1000.
12. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part II. OARSI evidence-based, expert consensus and guidelines. Osteoarthritis Cartilage. 2008;16:137-162.
13. Zhang W, Moskowitz RW, Nuki G, et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part III. Changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthritis Cartilage. 2010;18:476-499.
14. Richmond J, Hunter D, Irrgang J, et al. Treatment of osteoarthritis of the knee (nonarthroscopy). J Am Acad Orthop Surg. 2009;17:591-600.
15. Coleman S, Briffa K, Conroy H, et al. Short and medium-term effects of an education self-management program for individuals with osteoarthritis of the knee, designed and delivered by health professionals: a quality assurance study. BMC Musculoskelet Disord. 2008;9:117.
16. Gutekunst DJ, Davis C, DeVita P. Weight loss reduces knee-joint loads in overweight and obese older adults with knee osteoarthritis. Arthritis Rheum. 2005; 52:2026-2032.
17. Bartlett SJ, Haaz S, Wrobleski P, et al. Small weight losses can yield significant improvements in knee OA symptoms. Arthritis Rheum. 2004;50(suppl):S658.
18. Christensen R, Astrup A, Bliddal H. Weight loss: the treatment of choice for osteoarthritis? A randomized trial. Osteoarthritis Cartilage. 2005;13:20-27.
19. Williams FMK, Skinner J, Spector TD, et al. Dietary garlic and hip osteoarthritis: evidence of a protective effect and putative mechanism of action. BMC Musculoskelet Disord. 2010;11:280.
20. Lin J, Zhang W, Jones A, et al. Efficacy of topical non-steroidal anti-inflammatory drugs in the treatment of osteoarthritis: Meta-analysis of randomized controlled trials. BMJ. 2004;329:324.
21. Stitik TP, Altschuler E, Foye PM. Pharmacotherapy of osteoarthritis. Am J Phys Med Rehabil. 2008; 85(suppl):S15-S28.
22. Bannuru RR, Natov NS, Dasi UR, et al. Therapeutic trajectory following intra-articular hyaluronic acid injection in knee osteoarthritis—meta-analysis.Osteoarthritis Cartilage. 2011;19:611-619.
23. Karlsson J, Sjogren LS, Lohmander LS. Comparison of two hyaluronan drugs and placebo in patients with knee osteoarthritis. A controlled, randomized, double-blind, parallel-design multicentre study. Rheumatology. 2002;41:1240-1248.
24. Modawal A, Ferrer M, Choi HK, et al. Hyaluronic acid injections relieve knee pain. J Fam Pract. 2005;54:758-677.
25. Manheimer E, Lind K, Lao L, et al. Meta-analysis: Acupuncture for osteoarthritis of the knee. Ann Intern Med. 2007;146:868-877.
26. Witt C, Brinkhaus B, Jena S, et al. Acupuncture in patients with osteoarthritis of the knee: a randomised trial. Lancet. 2005;366:136-143.
27. Berman BM, Lao L, Langenberg P, et al. Effectiveness of acupuncture as adjunctive therapy in osteoarthritis of the knee: a randomized, controlled trial. Ann Intern Med. 2004;141:901-910.
28. Kabat-Zinn J. An outpatient program in behavioral medicine for chronic pain patients based on the practice of mindfulness meditation: theoretical considerations and preliminary results. Gen Hosp Psychiatry. 1982;4:33-47.
29. Zeidan F, Martucci KT, Kraft RA, et al. Brain mechanisms supporting the modulation of pain by mindfulness meditation. J Neurosci. 2011;31:5540-5548.
30. Holzel BK, Ott U, Gard T, et al. Investigation of mindfulness meditation practitioners with voxel-based morphometry: Soc Cogn Affect Neurosci. 2008;3:55-61
31. Grossman P, Niemann L, Schmidt S, et al. Mindfulness-based stress reduction and health benefits: a meta-analysis. Psychiatry Res. 2004;57:35-43.
32. Pollard H, Ward G, Hoskins W, et al. The effect of a manual knee protocol on osteoarthritic knee pain: a randomised controlled trial. J Can Chiropr Assoc. 2008;52:229-242.
33. Brantingham JW, Globe G, Pollard H, et al. Manipulative therapy for lower extremity conditions: expansion of literature review. J Manipulative Physiol Ther. 2009;32:53-71.
34. Rutjes AW, Nuesch E, Sterchi R, et al. Transcutaneous electrostimulation for osteoarthritis of the knee. Cochrane Database Syst Rev. 2009;(4):CD002823.
35. Pietrosimone BG, Saliba SA, Hart JM, et al. Effects of transcutaneous electrical nerve stimulation and therapeutic exercise on quadriceps activation in people with tibiofemoral osteoarthritis. J Orthop Sports Phys Ther. 2011;41:4-12.
36. Wang C, Collet JP, Lau J. The effect of Tai Chi on health outcomes in patients with chronic conditions: a systematic review. Arch Intern Med. 2004;164:493-501.
37. Lee MS, Pittler MH, Ernst E. Tai Chi for osteoarthritis: a systematic review. Clin Rheumatol. 2008;27:211-218.
38. Wang C, Schmid CH, Hibberd PL, et al. Tai chi is effective in treating knee osteoarthritis: a randomized controlled trial. Arthritis Rheum. 2009;61:1545-1553.
39. Ramsey SD, Spenser AC, Topolski TD, et al. Use of alternative therapies by older adults with osteoarthritis. Arthritis Care Res. 2001;45:222-227.
40. Perlman AI, Sabina A, Williams AL, et al. Massage therapy for osteoarthritis of the knee: a randomized controlled trial. Arch Intern Med. 2006;166:2533-2538.
41. Ko GD, Nowacki NB, Arseneau L, et al. Omega-3 fatty acids for neuropathic pain: case series. Clin J Pain. 2010;26:168-172.
42. Goldberg RJ, Katz J. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain. 2007;129:210-223.
43. McAlindon TE, LaValley MP, Gulin JP, et al. Glucosamine and chondroitin for treatment of osteoarthritis: a systematic quality assessment and meta-analysis. JAMA.2000;283:1469-1475.
44. Wandel S, Jüni P, Tendal B, et al. Effects of glucosamine, chondroitin, or placebo in patients with osteoarthritis of hip or knee: Network meta-analysis. BMJ. 2010;341:c4675.
45. Sawitzke AD, Shi H, Finco MF, et al. Clinical efficacy and safety of glucosamine, chondroitin sulphate, their combination, celecoxib or placebo taken to treat osteoarthritis of the knee: 2-year results from GAIT. Ann Rheum Dis. 2010;69:1459-1464.
46. Berman B. A 60-year-old woman considering acupuncture for knee pain. JAMA. 2007;297:1697-1707.
47. Arjmandi BH, Khalil DA, Lucas EA, et al. Soy protein may alleviate osteoarthritis symptoms. Phytomedicine. 2004;11:567-575.
48. Maheu E, Mazieres B, Valat JP, et al. Symptomatic efficacy of avocado/soybean unsaponifiables in the treatment of osteoarthritis of the knee and hip: a prospective, randomized, double-blind, placebo-controlled, multicenter clinical trial with a six-month treatment period and a two-month follow-up demonstrating a persistent effect. Arthritis Rheum. 1998;41:81-91.
49. Maroon JC, Bost JW, Maroon A. Natural anti-inflammatory agents for pain relief. Surg Neurol Int. 2010;1:80.
50. Ahmed S, Wang N, Lalonde M, et al. Green tea polyphenol epigallocatechin-3-gallate (EGCG) differentially inhibits interleukin-1 beta-induced expression of matrix metalloproteinase-1 and -13 in human chondrocytes. J Pharmacol Exp Ther. 2004;308:767-773.
51. Funk JL, Oyarzo JN, Frye JB, et al. Turmeric extracts containing curcuminoids prevent experimental rheumatoid arthritis. J Nat Prod. 2006;69:351-355.
52. Altman RD, Marcussen KC. Effects of a ginger extract on knee pain in patients with osteoarthritis. Arthritis Rheum. 2001;44:2531-2538.
53. Cardile V, Panico A, Gentile B, et al. Effect of propolis on human cartilage and chondrocytes. Life Sci. 2003;73:1027-1035.
54. Ng NT, Heesch KC, Brown WJ. Efficacy of a progressive walking program and glucosamine sulphate supplementation on osteoarthritic symptoms of the hip and knee: a feasibility trial. Arthritis Res Ther. 2010;12:R25.