User login
Enzyme tablet eases pain of gluten consumption
CHICAGO – Gluten-sensitive patients were able to tolerate small amounts of gluten after consuming an enzyme supplement derived from Aspergillus niger as part of a meal in a randomized, placebo-controlled trial of 18 adults. The data were presented at the annual Digestive Disease Week.
The enzyme, A. niger-derived prolyl endoprotease (AN-PEP), has demonstrated the ability to degrade gluten into nonimmunogenic compounds in vivo in healthy subjects, according to Julia König, PhD, of the School of Medical Sciences, Örebro (Sweden) University and her colleagues. The researchers tested the enzyme at two separate doses in 18 adults with self-reported gluten sensitivity.
The participants’ gastric and duodenal content was sampled several times over 180 minutes and analyzed for gluten epitopes using an enzyme-linked immunosorbent assay test. Participants also completed questionnaires after each day of testing.
After taking the enzyme in conjunction with the gluten, stomach gluten content averaged 31 microg x min/mL in the high-dose and low-dose enzyme patients (P = 0.001 for both doses), compared with 281 microg x min/mL in the placebo patients.
By the time the gluten reached the duodenum, the average levels had dropped to 12 microg x min/mL in the high-dose patients (P = 0.019) and 8 microg x min/mL in the low-dose patients (P = 0.015), compared with an average of 65 microg x min/mL in the placebo patients.
Overall, the enzyme was well tolerated by the patients, the researchers said. However, Dr. König emphasized that the enzyme tablet is meant to help avoid symptoms when gluten-sensitive patients encounter small amounts of gluten, and these patients should still follow a gluten-free diet.
AN-PEP is available in the United States in supplement form under several names and is manufactured by the Dutch company DSM. The AN-PEP enzyme used in the study was provided by DSM, but the company provided no other support. Dr. König had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
CHICAGO – Gluten-sensitive patients were able to tolerate small amounts of gluten after consuming an enzyme supplement derived from Aspergillus niger as part of a meal in a randomized, placebo-controlled trial of 18 adults. The data were presented at the annual Digestive Disease Week.
The enzyme, A. niger-derived prolyl endoprotease (AN-PEP), has demonstrated the ability to degrade gluten into nonimmunogenic compounds in vivo in healthy subjects, according to Julia König, PhD, of the School of Medical Sciences, Örebro (Sweden) University and her colleagues. The researchers tested the enzyme at two separate doses in 18 adults with self-reported gluten sensitivity.
The participants’ gastric and duodenal content was sampled several times over 180 minutes and analyzed for gluten epitopes using an enzyme-linked immunosorbent assay test. Participants also completed questionnaires after each day of testing.
After taking the enzyme in conjunction with the gluten, stomach gluten content averaged 31 microg x min/mL in the high-dose and low-dose enzyme patients (P = 0.001 for both doses), compared with 281 microg x min/mL in the placebo patients.
By the time the gluten reached the duodenum, the average levels had dropped to 12 microg x min/mL in the high-dose patients (P = 0.019) and 8 microg x min/mL in the low-dose patients (P = 0.015), compared with an average of 65 microg x min/mL in the placebo patients.
Overall, the enzyme was well tolerated by the patients, the researchers said. However, Dr. König emphasized that the enzyme tablet is meant to help avoid symptoms when gluten-sensitive patients encounter small amounts of gluten, and these patients should still follow a gluten-free diet.
AN-PEP is available in the United States in supplement form under several names and is manufactured by the Dutch company DSM. The AN-PEP enzyme used in the study was provided by DSM, but the company provided no other support. Dr. König had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
CHICAGO – Gluten-sensitive patients were able to tolerate small amounts of gluten after consuming an enzyme supplement derived from Aspergillus niger as part of a meal in a randomized, placebo-controlled trial of 18 adults. The data were presented at the annual Digestive Disease Week.
The enzyme, A. niger-derived prolyl endoprotease (AN-PEP), has demonstrated the ability to degrade gluten into nonimmunogenic compounds in vivo in healthy subjects, according to Julia König, PhD, of the School of Medical Sciences, Örebro (Sweden) University and her colleagues. The researchers tested the enzyme at two separate doses in 18 adults with self-reported gluten sensitivity.
The participants’ gastric and duodenal content was sampled several times over 180 minutes and analyzed for gluten epitopes using an enzyme-linked immunosorbent assay test. Participants also completed questionnaires after each day of testing.
After taking the enzyme in conjunction with the gluten, stomach gluten content averaged 31 microg x min/mL in the high-dose and low-dose enzyme patients (P = 0.001 for both doses), compared with 281 microg x min/mL in the placebo patients.
By the time the gluten reached the duodenum, the average levels had dropped to 12 microg x min/mL in the high-dose patients (P = 0.019) and 8 microg x min/mL in the low-dose patients (P = 0.015), compared with an average of 65 microg x min/mL in the placebo patients.
Overall, the enzyme was well tolerated by the patients, the researchers said. However, Dr. König emphasized that the enzyme tablet is meant to help avoid symptoms when gluten-sensitive patients encounter small amounts of gluten, and these patients should still follow a gluten-free diet.
AN-PEP is available in the United States in supplement form under several names and is manufactured by the Dutch company DSM. The AN-PEP enzyme used in the study was provided by DSM, but the company provided no other support. Dr. König had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
FROM DDW
Key clinical point: Consuming an enzyme tablet simultaneously with small amounts of gluten may reduce discomfort in gluten-sensitive individuals.
Major finding: On average, gluten levels in the stomach after enzyme consumption were 31 microg x min/mL in both high- and low-dose groups, vs. 281 microg x min/mL in a placebo group.
Data source: A randomized, placebo-controlled trial of 18 gluten-sensitive adults.
Disclosures: The enzyme used in the study, AN-PEP, was provided by the Dutch company DSM, but the company provided no other support. Dr. König had no relevant financial conflicts to disclose.
Genetic test predicts cirrhosis outcomes
CHICAGO – Cirrhosis patients with the rs738409 CG/GG genotype experienced worse outcomes, including a slower recovery of encephalopathy, ascites, and bilirubin, compared with those without this CG/GG genotype, based on data from a prospective study. The findings were presented at the annual Digestive Disease Week.
Most patients with hepatitis C virus–associated cirrhosis do well after treatment with direct-acting antiviral agents to achieve a sustained virologic response, according to Winston Dunn, MD, of Kansas University Medical Center, Kansas City, and his colleagues.
However, patients with decompensated cirrhosis may have a range of outcomes, and, to help predict treatment success, Dr. Dunn and his colleagues examined the possible genetic role of the rs738409 Single Nucleotide Polymorphism of Patatin-like Phospholipase Domain Containing 3 gene.
The researchers assessed 30 adults with Child-Pugh (CPT) Class B or C cirrhosis caused by HCV infection who underwent interferon-free, direct-acting antiviral therapy and achieved sustained virologic response. They collected DNA from each patient using a cheek swab. The study population included 16 patients with a CC genotype, 11 with CG, and 3 with GG.
They measured changes in CPT scores and Model for End-Stage Liver Disease (MELD) scores from before DAA treatment to 12 weeks after treatment. Baseline scores were similar among all patients, as were demographic characteristics, although patients with the rs738409 CC genotype averaged a higher body-mass index 35 kg/m2, vs. 29 kg/m2 (P = 0.03).
After 12 weeks, 13 of 16 patients with the CC genotype (81 %) had improved CPT scores, and 8 patients (50%) had improved MELD scores by at least 1 point. None had worsened CPT or MELD scores. By contrast, 5 of 14 patients with CG/GG genotype (36%) had improved CPT scores, and 4 (29%) had improved MELD scores by at least 1 point; 3 patients (21%) had worsened CPT scores and 4 (29%) had worsened MELD scores by at least 1 point.
Overall, patients in the CG/GG groups showed a 1.7-point higher delta CPT score and a 2.3-point higher delta MELD score after adjusting for confounding variables, compared with patients with CC after adjusting for confounding variables.
The study findings were limited by small numbers and prospective design, and the genetic test is not yet widely available, Dr. Dunn said. However, “Our results will help target patients for liver transplant evaluation based on individual genetic risk factors,” the researchers said.
The study was funded by the Frontiers Pilot and Collaborative Studies Funding Program. Dr. Dunn had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
CHICAGO – Cirrhosis patients with the rs738409 CG/GG genotype experienced worse outcomes, including a slower recovery of encephalopathy, ascites, and bilirubin, compared with those without this CG/GG genotype, based on data from a prospective study. The findings were presented at the annual Digestive Disease Week.
Most patients with hepatitis C virus–associated cirrhosis do well after treatment with direct-acting antiviral agents to achieve a sustained virologic response, according to Winston Dunn, MD, of Kansas University Medical Center, Kansas City, and his colleagues.
However, patients with decompensated cirrhosis may have a range of outcomes, and, to help predict treatment success, Dr. Dunn and his colleagues examined the possible genetic role of the rs738409 Single Nucleotide Polymorphism of Patatin-like Phospholipase Domain Containing 3 gene.
The researchers assessed 30 adults with Child-Pugh (CPT) Class B or C cirrhosis caused by HCV infection who underwent interferon-free, direct-acting antiviral therapy and achieved sustained virologic response. They collected DNA from each patient using a cheek swab. The study population included 16 patients with a CC genotype, 11 with CG, and 3 with GG.
They measured changes in CPT scores and Model for End-Stage Liver Disease (MELD) scores from before DAA treatment to 12 weeks after treatment. Baseline scores were similar among all patients, as were demographic characteristics, although patients with the rs738409 CC genotype averaged a higher body-mass index 35 kg/m2, vs. 29 kg/m2 (P = 0.03).
After 12 weeks, 13 of 16 patients with the CC genotype (81 %) had improved CPT scores, and 8 patients (50%) had improved MELD scores by at least 1 point. None had worsened CPT or MELD scores. By contrast, 5 of 14 patients with CG/GG genotype (36%) had improved CPT scores, and 4 (29%) had improved MELD scores by at least 1 point; 3 patients (21%) had worsened CPT scores and 4 (29%) had worsened MELD scores by at least 1 point.
Overall, patients in the CG/GG groups showed a 1.7-point higher delta CPT score and a 2.3-point higher delta MELD score after adjusting for confounding variables, compared with patients with CC after adjusting for confounding variables.
The study findings were limited by small numbers and prospective design, and the genetic test is not yet widely available, Dr. Dunn said. However, “Our results will help target patients for liver transplant evaluation based on individual genetic risk factors,” the researchers said.
The study was funded by the Frontiers Pilot and Collaborative Studies Funding Program. Dr. Dunn had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
CHICAGO – Cirrhosis patients with the rs738409 CG/GG genotype experienced worse outcomes, including a slower recovery of encephalopathy, ascites, and bilirubin, compared with those without this CG/GG genotype, based on data from a prospective study. The findings were presented at the annual Digestive Disease Week.
Most patients with hepatitis C virus–associated cirrhosis do well after treatment with direct-acting antiviral agents to achieve a sustained virologic response, according to Winston Dunn, MD, of Kansas University Medical Center, Kansas City, and his colleagues.
However, patients with decompensated cirrhosis may have a range of outcomes, and, to help predict treatment success, Dr. Dunn and his colleagues examined the possible genetic role of the rs738409 Single Nucleotide Polymorphism of Patatin-like Phospholipase Domain Containing 3 gene.
The researchers assessed 30 adults with Child-Pugh (CPT) Class B or C cirrhosis caused by HCV infection who underwent interferon-free, direct-acting antiviral therapy and achieved sustained virologic response. They collected DNA from each patient using a cheek swab. The study population included 16 patients with a CC genotype, 11 with CG, and 3 with GG.
They measured changes in CPT scores and Model for End-Stage Liver Disease (MELD) scores from before DAA treatment to 12 weeks after treatment. Baseline scores were similar among all patients, as were demographic characteristics, although patients with the rs738409 CC genotype averaged a higher body-mass index 35 kg/m2, vs. 29 kg/m2 (P = 0.03).
After 12 weeks, 13 of 16 patients with the CC genotype (81 %) had improved CPT scores, and 8 patients (50%) had improved MELD scores by at least 1 point. None had worsened CPT or MELD scores. By contrast, 5 of 14 patients with CG/GG genotype (36%) had improved CPT scores, and 4 (29%) had improved MELD scores by at least 1 point; 3 patients (21%) had worsened CPT scores and 4 (29%) had worsened MELD scores by at least 1 point.
Overall, patients in the CG/GG groups showed a 1.7-point higher delta CPT score and a 2.3-point higher delta MELD score after adjusting for confounding variables, compared with patients with CC after adjusting for confounding variables.
The study findings were limited by small numbers and prospective design, and the genetic test is not yet widely available, Dr. Dunn said. However, “Our results will help target patients for liver transplant evaluation based on individual genetic risk factors,” the researchers said.
The study was funded by the Frontiers Pilot and Collaborative Studies Funding Program. Dr. Dunn had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
FROM DDW
Key clinical point: Genotyping patients with advanced cirrhosis from HCV could help predict improvement and determine fitness for liver transplants.
Major finding: The rs738409 CG/GG genotype was associated with a 1.7-point higher delta CPT score, a 2.3 -point higher delta MELD score, and slower recovery of encepholpathy, ascites, and bilirubin, compared with those without this CG/GG genotype.
Data source: A prospective study of 35 adults with cirrhosis caused by HCV infection.
Disclosures: The study was funded by the Frontiers Pilot and Collaborative Studies Funding Program.
Endoscopic weight loss surgery cuts costs, side effects
Obese patients who underwent endoscopic sleeve gastroplasty had significantly fewer complications and shorter hospital stays than did those who had laparoscopic sleeve gastrectomy or laparoscopic band placement, according to results from a study of 278 adults. The data were presented at the annual Digestive Disease Week®.
Overall, 1% of patients who underwent endoscopic sleeve gastroplasty (ESG) experienced adverse events, compared with 8% of those who underwent laparoscopic sleeve gastrectomy (LSG) and 9% of those who underwent laparoscopic band placement (LAGB).
ESG, which reduces gastric volume by use of an endoscopic suturing system of full-thickness sutures through the greater curvature of the stomach, is becoming a popular weight-loss procedure for patients with a body mass index greater than 30 kg/m2 who are poor candidates for laparoscopic surgery or who would prefer a less invasive procedure, according to Reem Z. Sharaiha, MD, of Cornell University, New York.
Dr. Sharaiha and her colleagues randomized 91 patients to ESG, 120 to LSG, and 67 to LAGB. Patient demographic characteristics, including age, gender, and diabetes, were similar among the three groups. However, patients in the LSG group had a higher average BMI than did the LAGB and ESG groups (47.3 kg/m2, 45.7 kg/m2, and 38.8 kg/m2, respectively). In addition, the incidence of hypertension, and hyperlipidemia was significantly higher in each of the surgical groups compared to the ESG group (P less than .01).
The average postprocedure hospital stay was 0.13 days for ESG patients compared with 3.09 days for LSG patients and 1.68 days for LAGB patients. ESG also had the lowest cost of the three procedures, averaging $12,000 for the procedure compared to $22,000 for LSG and $15,000 for LAGB.
After 1 year, patients in the LSG group had the greatest percentage of total body weight loss (29.3%), followed by ESG patients (17.6%), and LAGB patients (14.5%). Rates of leaks, pulmonary embolism events, and 90-day readmission were not significantly different among the groups.
The study results do not imply that ESG will replace either LAGB or LSG for weight loss, Dr. Sharaiha noted, but the results suggest that ESG is a viable option for some patients.
Dr. Sharaiha had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
Obese patients who underwent endoscopic sleeve gastroplasty had significantly fewer complications and shorter hospital stays than did those who had laparoscopic sleeve gastrectomy or laparoscopic band placement, according to results from a study of 278 adults. The data were presented at the annual Digestive Disease Week®.
Overall, 1% of patients who underwent endoscopic sleeve gastroplasty (ESG) experienced adverse events, compared with 8% of those who underwent laparoscopic sleeve gastrectomy (LSG) and 9% of those who underwent laparoscopic band placement (LAGB).
ESG, which reduces gastric volume by use of an endoscopic suturing system of full-thickness sutures through the greater curvature of the stomach, is becoming a popular weight-loss procedure for patients with a body mass index greater than 30 kg/m2 who are poor candidates for laparoscopic surgery or who would prefer a less invasive procedure, according to Reem Z. Sharaiha, MD, of Cornell University, New York.
Dr. Sharaiha and her colleagues randomized 91 patients to ESG, 120 to LSG, and 67 to LAGB. Patient demographic characteristics, including age, gender, and diabetes, were similar among the three groups. However, patients in the LSG group had a higher average BMI than did the LAGB and ESG groups (47.3 kg/m2, 45.7 kg/m2, and 38.8 kg/m2, respectively). In addition, the incidence of hypertension, and hyperlipidemia was significantly higher in each of the surgical groups compared to the ESG group (P less than .01).
The average postprocedure hospital stay was 0.13 days for ESG patients compared with 3.09 days for LSG patients and 1.68 days for LAGB patients. ESG also had the lowest cost of the three procedures, averaging $12,000 for the procedure compared to $22,000 for LSG and $15,000 for LAGB.
After 1 year, patients in the LSG group had the greatest percentage of total body weight loss (29.3%), followed by ESG patients (17.6%), and LAGB patients (14.5%). Rates of leaks, pulmonary embolism events, and 90-day readmission were not significantly different among the groups.
The study results do not imply that ESG will replace either LAGB or LSG for weight loss, Dr. Sharaiha noted, but the results suggest that ESG is a viable option for some patients.
Dr. Sharaiha had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
Obese patients who underwent endoscopic sleeve gastroplasty had significantly fewer complications and shorter hospital stays than did those who had laparoscopic sleeve gastrectomy or laparoscopic band placement, according to results from a study of 278 adults. The data were presented at the annual Digestive Disease Week®.
Overall, 1% of patients who underwent endoscopic sleeve gastroplasty (ESG) experienced adverse events, compared with 8% of those who underwent laparoscopic sleeve gastrectomy (LSG) and 9% of those who underwent laparoscopic band placement (LAGB).
ESG, which reduces gastric volume by use of an endoscopic suturing system of full-thickness sutures through the greater curvature of the stomach, is becoming a popular weight-loss procedure for patients with a body mass index greater than 30 kg/m2 who are poor candidates for laparoscopic surgery or who would prefer a less invasive procedure, according to Reem Z. Sharaiha, MD, of Cornell University, New York.
Dr. Sharaiha and her colleagues randomized 91 patients to ESG, 120 to LSG, and 67 to LAGB. Patient demographic characteristics, including age, gender, and diabetes, were similar among the three groups. However, patients in the LSG group had a higher average BMI than did the LAGB and ESG groups (47.3 kg/m2, 45.7 kg/m2, and 38.8 kg/m2, respectively). In addition, the incidence of hypertension, and hyperlipidemia was significantly higher in each of the surgical groups compared to the ESG group (P less than .01).
The average postprocedure hospital stay was 0.13 days for ESG patients compared with 3.09 days for LSG patients and 1.68 days for LAGB patients. ESG also had the lowest cost of the three procedures, averaging $12,000 for the procedure compared to $22,000 for LSG and $15,000 for LAGB.
After 1 year, patients in the LSG group had the greatest percentage of total body weight loss (29.3%), followed by ESG patients (17.6%), and LAGB patients (14.5%). Rates of leaks, pulmonary embolism events, and 90-day readmission were not significantly different among the groups.
The study results do not imply that ESG will replace either LAGB or LSG for weight loss, Dr. Sharaiha noted, but the results suggest that ESG is a viable option for some patients.
Dr. Sharaiha had no relevant financial conflicts to disclose.
Digestive Disease Week® is jointly sponsored by the American Association for the Study of Liver Diseases (AASLD), the American Gastroenterological Association (AGA) Institute, the American Society for Gastrointestinal Endoscopy (ASGE), and the Society for Surgery of the Alimentary Tract (SSAT).
FROM DDW
Key clinical point: Endoscopic sleeve gastroplasty is a viable option for patients seeking weight loss but wishing to avoid major surgery.
Major finding: After 1 year, 1% of patients who underwent endoscopic sleeve gastroplasty experienced adverse events, compared with 8% of laparoscopic sleeve gastrectomy patients, and 9% of laparoscopic band placement patients.
Data source: A randomized trial of 278 obese adults who underwent one of three weight loss procedures.
Disclosures: Dr. Sharaiha had no relevant financial conflicts to disclose.
Twice-daily tofacitinib induces ulcerative colitis remission
A 10-mg dose of tofacitinib twice daily was significantly more effective than placebo for inducing remission in ulcerative colitis patients, based on data from a group of three randomized trials totaling approximately 1,500 adults. The findings were published online May 3 in the New England Journal of Medicine (2017;376:1723-36).
The series of OCTAVE trials (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) included adults with moderately to severely active ulcerative colitis (UC). Patients were randomized to 10 mg of tofacitinib, 5 mg tofacitinib, or placebo. The studies were conducted over a 4-year period, at 144 sites for OCTAVE 1, 169 sites for OCTAVE 2, and 297 sites for OCTAVE Sustain.
In both OCTAVE 1 and OCTAVE 2, the remission rates at 8 weeks were significantly higher in the 10-mg tofacitinib groups, compared with the placebo groups (18.5% vs. 8.2%, respectively; 16.6% vs. 3.6%, respectively). The rate of remission at 52 weeks was significantly higher in the 5-mg and 10-mg tofacitinib groups (34.3% and 40.6%, respectively) than in the placebo group (11.1%) in the OCTAVE Sustain trial.
“Pharmacokinetic results in the OCTAVE trials did not indicate a decrease in plasma tofacitinib concentrations during the course of treatment at any given dose in individual patients. These results are consistent with the previously established physicochemical characteristics and clearance mechanisms of tofacitinib,” noted William J. Sandborn, MD, of the University of California, San Diego, and his colleagues.
In the OCTAVE 1 trial, serious adverse events occurred in 4.2% and 8.0% of patients in the 10-mg and placebo groups, respectively. In the OCTAVE 2 trial, they occured in 3.4% and 4.1% of the 10-mg and placebo groups, respectively. The rate of serious adverse events in the OCTAVE Sustain trial was 5.1%, 5.6%, and 6.6% in the 10-mg, 5-mg, and placebo groups, respectively. Tofacitinib was associated with increased lipid levels, as well as higher rates of overall infection and herpes zoster infection, compared with placebo.
The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies including Pfizer.
AGA Resource
AGA offers materials to help you manage patients with Crohn’s disease and ulcerative colitis, and integrate patient education into your practice, including multimedia publications that are accessible and informative. Learn more at http://www.gastro.org/ibd
“This report is the culmination of an international effort,” over a 4-year period, wrote Sonia Friedman, MD, of Harvard University in an accompanying editorial.
“This study has all the elements of a high-quality trial: a large, international cohort of patients and investigators; reasonable enrollment criteria that would apply to many of my own patients with moderate-to-severe ulcerative colitis; rigorous and unbiased scoring for response, remission, and mucosal healing; fair adjudication of adverse events; and meticulous reporting of all meaningful outcomes and laboratory test results,” she said. Tofacitinib has proven its efficacy – its exact role will be determined by additional research, she noted.
“Only a continued combination of human ingenuity, worldwide cooperation, and enthusiastic funding will allow investigators to further explore the mechanisms by which JAK inhibition ameliorates inflammation in patients with ulcerative colitis and ... identify the specific subsets of patients who will most likely benefit from this new therapy,” Dr. Friedman emphasized (N. Engl. J. Med. 2017;376:1792-3).
Dr. Friedman is affiliated with Harvard University, Boston, Mass., and Brigham and Women’s Hospital Center for Crohn’s and Colitis, Chestnut Hill, Mass. She disclosed receiving personal fees from Boston University.
“This report is the culmination of an international effort,” over a 4-year period, wrote Sonia Friedman, MD, of Harvard University in an accompanying editorial.
“This study has all the elements of a high-quality trial: a large, international cohort of patients and investigators; reasonable enrollment criteria that would apply to many of my own patients with moderate-to-severe ulcerative colitis; rigorous and unbiased scoring for response, remission, and mucosal healing; fair adjudication of adverse events; and meticulous reporting of all meaningful outcomes and laboratory test results,” she said. Tofacitinib has proven its efficacy – its exact role will be determined by additional research, she noted.
“Only a continued combination of human ingenuity, worldwide cooperation, and enthusiastic funding will allow investigators to further explore the mechanisms by which JAK inhibition ameliorates inflammation in patients with ulcerative colitis and ... identify the specific subsets of patients who will most likely benefit from this new therapy,” Dr. Friedman emphasized (N. Engl. J. Med. 2017;376:1792-3).
Dr. Friedman is affiliated with Harvard University, Boston, Mass., and Brigham and Women’s Hospital Center for Crohn’s and Colitis, Chestnut Hill, Mass. She disclosed receiving personal fees from Boston University.
“This report is the culmination of an international effort,” over a 4-year period, wrote Sonia Friedman, MD, of Harvard University in an accompanying editorial.
“This study has all the elements of a high-quality trial: a large, international cohort of patients and investigators; reasonable enrollment criteria that would apply to many of my own patients with moderate-to-severe ulcerative colitis; rigorous and unbiased scoring for response, remission, and mucosal healing; fair adjudication of adverse events; and meticulous reporting of all meaningful outcomes and laboratory test results,” she said. Tofacitinib has proven its efficacy – its exact role will be determined by additional research, she noted.
“Only a continued combination of human ingenuity, worldwide cooperation, and enthusiastic funding will allow investigators to further explore the mechanisms by which JAK inhibition ameliorates inflammation in patients with ulcerative colitis and ... identify the specific subsets of patients who will most likely benefit from this new therapy,” Dr. Friedman emphasized (N. Engl. J. Med. 2017;376:1792-3).
Dr. Friedman is affiliated with Harvard University, Boston, Mass., and Brigham and Women’s Hospital Center for Crohn’s and Colitis, Chestnut Hill, Mass. She disclosed receiving personal fees from Boston University.
A 10-mg dose of tofacitinib twice daily was significantly more effective than placebo for inducing remission in ulcerative colitis patients, based on data from a group of three randomized trials totaling approximately 1,500 adults. The findings were published online May 3 in the New England Journal of Medicine (2017;376:1723-36).
The series of OCTAVE trials (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) included adults with moderately to severely active ulcerative colitis (UC). Patients were randomized to 10 mg of tofacitinib, 5 mg tofacitinib, or placebo. The studies were conducted over a 4-year period, at 144 sites for OCTAVE 1, 169 sites for OCTAVE 2, and 297 sites for OCTAVE Sustain.
In both OCTAVE 1 and OCTAVE 2, the remission rates at 8 weeks were significantly higher in the 10-mg tofacitinib groups, compared with the placebo groups (18.5% vs. 8.2%, respectively; 16.6% vs. 3.6%, respectively). The rate of remission at 52 weeks was significantly higher in the 5-mg and 10-mg tofacitinib groups (34.3% and 40.6%, respectively) than in the placebo group (11.1%) in the OCTAVE Sustain trial.
“Pharmacokinetic results in the OCTAVE trials did not indicate a decrease in plasma tofacitinib concentrations during the course of treatment at any given dose in individual patients. These results are consistent with the previously established physicochemical characteristics and clearance mechanisms of tofacitinib,” noted William J. Sandborn, MD, of the University of California, San Diego, and his colleagues.
In the OCTAVE 1 trial, serious adverse events occurred in 4.2% and 8.0% of patients in the 10-mg and placebo groups, respectively. In the OCTAVE 2 trial, they occured in 3.4% and 4.1% of the 10-mg and placebo groups, respectively. The rate of serious adverse events in the OCTAVE Sustain trial was 5.1%, 5.6%, and 6.6% in the 10-mg, 5-mg, and placebo groups, respectively. Tofacitinib was associated with increased lipid levels, as well as higher rates of overall infection and herpes zoster infection, compared with placebo.
The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies including Pfizer.
AGA Resource
AGA offers materials to help you manage patients with Crohn’s disease and ulcerative colitis, and integrate patient education into your practice, including multimedia publications that are accessible and informative. Learn more at http://www.gastro.org/ibd
A 10-mg dose of tofacitinib twice daily was significantly more effective than placebo for inducing remission in ulcerative colitis patients, based on data from a group of three randomized trials totaling approximately 1,500 adults. The findings were published online May 3 in the New England Journal of Medicine (2017;376:1723-36).
The series of OCTAVE trials (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) included adults with moderately to severely active ulcerative colitis (UC). Patients were randomized to 10 mg of tofacitinib, 5 mg tofacitinib, or placebo. The studies were conducted over a 4-year period, at 144 sites for OCTAVE 1, 169 sites for OCTAVE 2, and 297 sites for OCTAVE Sustain.
In both OCTAVE 1 and OCTAVE 2, the remission rates at 8 weeks were significantly higher in the 10-mg tofacitinib groups, compared with the placebo groups (18.5% vs. 8.2%, respectively; 16.6% vs. 3.6%, respectively). The rate of remission at 52 weeks was significantly higher in the 5-mg and 10-mg tofacitinib groups (34.3% and 40.6%, respectively) than in the placebo group (11.1%) in the OCTAVE Sustain trial.
“Pharmacokinetic results in the OCTAVE trials did not indicate a decrease in plasma tofacitinib concentrations during the course of treatment at any given dose in individual patients. These results are consistent with the previously established physicochemical characteristics and clearance mechanisms of tofacitinib,” noted William J. Sandborn, MD, of the University of California, San Diego, and his colleagues.
In the OCTAVE 1 trial, serious adverse events occurred in 4.2% and 8.0% of patients in the 10-mg and placebo groups, respectively. In the OCTAVE 2 trial, they occured in 3.4% and 4.1% of the 10-mg and placebo groups, respectively. The rate of serious adverse events in the OCTAVE Sustain trial was 5.1%, 5.6%, and 6.6% in the 10-mg, 5-mg, and placebo groups, respectively. Tofacitinib was associated with increased lipid levels, as well as higher rates of overall infection and herpes zoster infection, compared with placebo.
The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies including Pfizer.
AGA Resource
AGA offers materials to help you manage patients with Crohn’s disease and ulcerative colitis, and integrate patient education into your practice, including multimedia publications that are accessible and informative. Learn more at http://www.gastro.org/ibd
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Tofacitinib, a JAK inhibitor, was a significantly more effective induction and maintenance therapy for patients with moderate to severe ulcerative colitis compared with placebo.
Major finding: Tofacitinib dosed at 10 mg twice daily yielded a remission rate of 41% at 52 weeks, compared with 11% in a placebo group.
Data source: The OCTAVE series of three randomized trials totaled approximately 1,500 adults with moderate to severely active ulcerative colitis.
Disclosures: The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies, including Pfizer.
Twice-daily tofacitinib induces ulcerative colitis remission
A 10-mg dose of tofacitinib twice daily was significantly more effective than placebo for inducing remission in ulcerative colitis patients, based on data from a group of three randomized trials totaling approximately 1,500 adults. The findings were published online May 3 in the New England Journal of Medicine (2017;376:1723-36).
The series of OCTAVE trials (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) included adults with moderately to severely active ulcerative colitis (UC). Patients were randomized to 10 mg of tofacitinib, 5 mg tofacitinib, or placebo. The studies were conducted over a 4-year period, at 144 sites for OCTAVE 1, 169 sites for OCTAVE 2, and 297 sites for OCTAVE Sustain.
In both OCTAVE 1 and OCTAVE 2, the remission rates at 8 weeks were significantly higher in the 10-mg tofacitinib groups, compared with the placebo groups (18.5% vs. 8.2%, respectively; 16.6% vs. 3.6%, respectively). The rate of remission at 52 weeks was significantly higher in the 5-mg and 10-mg tofacitinib groups (34.3% and 40.6%, respectively) than in the placebo group (11.1%) in the OCTAVE Sustain trial.
In addition, rates of mucosal healing were greater in the tofacitinib group than in the placebo group at 8 weeks and 52 weeks.
In the OCTAVE 1 trial, serious adverse events occurred in 4.2% and 8.0% of patients in the 10-mg and placebo groups, respectively. In the OCTAVE 2 trial, they occured in 3.4% and 4.1% of the 10-mg and placebo groups, respectively. The rate of serious adverse events in the OCTAVE Sustain trial was 5.1%, 5.6%, and 6.6% in the 10-mg, 5-mg, and placebo groups, respectively. Tofacitinib was associated with increased lipid levels, as well as higher rates of overall infection and herpes zoster infection, compared with placebo.
The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies including Pfizer.
“This report is the culmination of an international effort,” over a 4-year period, wrote Sonia Friedman, MD, of Harvard University in an accompanying editorial.
“This study has all the elements of a high-quality trial: a large, international cohort of patients and investigators; reasonable enrollment criteria that would apply to many of my own patients with moderate-to-severe ulcerative colitis; rigorous and unbiased scoring for response, remission, and mucosal healing; fair adjudication of adverse events; and meticulous reporting of all meaningful outcomes and laboratory test results,” she said. Tofacitinib has proven its efficacy – its exact role will be determined by additional research, she noted.
“Only a continued combination of human ingenuity, worldwide cooperation, and enthusiastic funding will allow investigators to further explore the mechanisms by which JAK inhibition ameliorates inflammation in patients with ulcerative colitis and ... identify the specific subsets of patients who will most likely benefit from this new therapy,” Dr. Friedman emphasized (N. Engl. J. Med. 2017;376:1792-3).
Dr. Friedman is affiliated with Harvard University, Boston, Mass., and Brigham and Women’s Hospital Center for Crohn’s and Colitis, Chestnut Hill, Mass. She disclosed receiving personal fees from Boston University.
“This report is the culmination of an international effort,” over a 4-year period, wrote Sonia Friedman, MD, of Harvard University in an accompanying editorial.
“This study has all the elements of a high-quality trial: a large, international cohort of patients and investigators; reasonable enrollment criteria that would apply to many of my own patients with moderate-to-severe ulcerative colitis; rigorous and unbiased scoring for response, remission, and mucosal healing; fair adjudication of adverse events; and meticulous reporting of all meaningful outcomes and laboratory test results,” she said. Tofacitinib has proven its efficacy – its exact role will be determined by additional research, she noted.
“Only a continued combination of human ingenuity, worldwide cooperation, and enthusiastic funding will allow investigators to further explore the mechanisms by which JAK inhibition ameliorates inflammation in patients with ulcerative colitis and ... identify the specific subsets of patients who will most likely benefit from this new therapy,” Dr. Friedman emphasized (N. Engl. J. Med. 2017;376:1792-3).
Dr. Friedman is affiliated with Harvard University, Boston, Mass., and Brigham and Women’s Hospital Center for Crohn’s and Colitis, Chestnut Hill, Mass. She disclosed receiving personal fees from Boston University.
“This report is the culmination of an international effort,” over a 4-year period, wrote Sonia Friedman, MD, of Harvard University in an accompanying editorial.
“This study has all the elements of a high-quality trial: a large, international cohort of patients and investigators; reasonable enrollment criteria that would apply to many of my own patients with moderate-to-severe ulcerative colitis; rigorous and unbiased scoring for response, remission, and mucosal healing; fair adjudication of adverse events; and meticulous reporting of all meaningful outcomes and laboratory test results,” she said. Tofacitinib has proven its efficacy – its exact role will be determined by additional research, she noted.
“Only a continued combination of human ingenuity, worldwide cooperation, and enthusiastic funding will allow investigators to further explore the mechanisms by which JAK inhibition ameliorates inflammation in patients with ulcerative colitis and ... identify the specific subsets of patients who will most likely benefit from this new therapy,” Dr. Friedman emphasized (N. Engl. J. Med. 2017;376:1792-3).
Dr. Friedman is affiliated with Harvard University, Boston, Mass., and Brigham and Women’s Hospital Center for Crohn’s and Colitis, Chestnut Hill, Mass. She disclosed receiving personal fees from Boston University.
A 10-mg dose of tofacitinib twice daily was significantly more effective than placebo for inducing remission in ulcerative colitis patients, based on data from a group of three randomized trials totaling approximately 1,500 adults. The findings were published online May 3 in the New England Journal of Medicine (2017;376:1723-36).
The series of OCTAVE trials (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) included adults with moderately to severely active ulcerative colitis (UC). Patients were randomized to 10 mg of tofacitinib, 5 mg tofacitinib, or placebo. The studies were conducted over a 4-year period, at 144 sites for OCTAVE 1, 169 sites for OCTAVE 2, and 297 sites for OCTAVE Sustain.
In both OCTAVE 1 and OCTAVE 2, the remission rates at 8 weeks were significantly higher in the 10-mg tofacitinib groups, compared with the placebo groups (18.5% vs. 8.2%, respectively; 16.6% vs. 3.6%, respectively). The rate of remission at 52 weeks was significantly higher in the 5-mg and 10-mg tofacitinib groups (34.3% and 40.6%, respectively) than in the placebo group (11.1%) in the OCTAVE Sustain trial.
In addition, rates of mucosal healing were greater in the tofacitinib group than in the placebo group at 8 weeks and 52 weeks.
In the OCTAVE 1 trial, serious adverse events occurred in 4.2% and 8.0% of patients in the 10-mg and placebo groups, respectively. In the OCTAVE 2 trial, they occured in 3.4% and 4.1% of the 10-mg and placebo groups, respectively. The rate of serious adverse events in the OCTAVE Sustain trial was 5.1%, 5.6%, and 6.6% in the 10-mg, 5-mg, and placebo groups, respectively. Tofacitinib was associated with increased lipid levels, as well as higher rates of overall infection and herpes zoster infection, compared with placebo.
The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies including Pfizer.
A 10-mg dose of tofacitinib twice daily was significantly more effective than placebo for inducing remission in ulcerative colitis patients, based on data from a group of three randomized trials totaling approximately 1,500 adults. The findings were published online May 3 in the New England Journal of Medicine (2017;376:1723-36).
The series of OCTAVE trials (Oral Clinical Trials for Tofacitinib in Ulcerative Colitis) included adults with moderately to severely active ulcerative colitis (UC). Patients were randomized to 10 mg of tofacitinib, 5 mg tofacitinib, or placebo. The studies were conducted over a 4-year period, at 144 sites for OCTAVE 1, 169 sites for OCTAVE 2, and 297 sites for OCTAVE Sustain.
In both OCTAVE 1 and OCTAVE 2, the remission rates at 8 weeks were significantly higher in the 10-mg tofacitinib groups, compared with the placebo groups (18.5% vs. 8.2%, respectively; 16.6% vs. 3.6%, respectively). The rate of remission at 52 weeks was significantly higher in the 5-mg and 10-mg tofacitinib groups (34.3% and 40.6%, respectively) than in the placebo group (11.1%) in the OCTAVE Sustain trial.
In addition, rates of mucosal healing were greater in the tofacitinib group than in the placebo group at 8 weeks and 52 weeks.
In the OCTAVE 1 trial, serious adverse events occurred in 4.2% and 8.0% of patients in the 10-mg and placebo groups, respectively. In the OCTAVE 2 trial, they occured in 3.4% and 4.1% of the 10-mg and placebo groups, respectively. The rate of serious adverse events in the OCTAVE Sustain trial was 5.1%, 5.6%, and 6.6% in the 10-mg, 5-mg, and placebo groups, respectively. Tofacitinib was associated with increased lipid levels, as well as higher rates of overall infection and herpes zoster infection, compared with placebo.
The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies including Pfizer.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Key clinical point: Tofacitinib, a JAK inhibitor, was a significantly more effective induction and maintenance therapy for patients with moderate to severe ulcerative colitis compared with placebo.
Major finding: Tofacitinib dosed at 10 mg twice daily yielded a remission rate of 41% at 52 weeks, compared with 11% in a placebo group.
Data source: The OCTAVE series of three randomized trials totaled approximately 1,500 adults with moderate to severely active ulcerative colitis.
Disclosures: The study was supported by Pfizer. Lead author Dr. Sandborn and several coauthors disclosed financial relationships with multiple companies, including Pfizer.
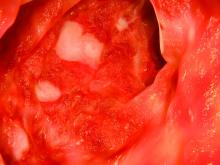
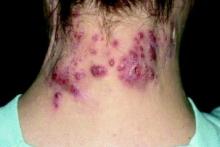
Hidradenitis suppurativa diagnosis typically delayed in children
WASHINGTON – Children with hidradenitis suppurativa (HS) may suffer with symptoms for an average of 7 years before they are diagnosed, according to pediatric dermatologist Anna Yasmine Kirkorian, MD.
Data from a 2015 study showed that 73% of pediatric patients with HS were diagnosed more than 2 years after the onset of symptoms, said Dr. Kirkorian of the department of dermatology at Children’s National Health System and George Washington University, Washington. (Br J Dermatol. 2015 Dec;173[6]:1546-9).
Genetics can play a role in HS, likely via mutations in the gamma-secretase protein that leads to epidermal differentiation and immune regulation, Dr. Kirkorian said. Most of her patients with HS are black, and a recent study described a gamma-secretase mutation in a black family of a proband and four family members, she noted (JAMA Dermatol. 2015 Jun;151[6]:668-70). Gamma-secretase mutations also have been identified in Han Chinese populations, she said.
HS also is associated with precocious puberty. However, defining the age of onset of puberty can be difficulty because pubertal onset may vary between different ethnicities, noted Dr. Kirkorian. “Prepubertal children presenting with HS warrant an endocrinologic evaluation,” she said.
Dr. Kirkorian added that more research is needed to pinpoint the possible genetic component of HS and to identify genetic susceptibility that could lead to targeted treatment strategies.
The optimal treatment plan for pediatric HS is multimodal and addresses the comorbidities common with the condition, she said, and she predicted that specialized clinic or treatment centers that bring together areas, including psychiatry, wound care, pain management, surgery, endocrinology, and genetics, will evolve to serve these patients. To support these collaborative efforts, Dr. Kirkorian is a member of the Pediatric Dermatology Research Alliance (PeDRA), an organization formed to accelerate research on skin diseases in children.
The symposium was sponsored by AbbVie. Dr. Kirkorian had no financial conflicts to disclose. She is on the editorial board of Dermatology News.
WASHINGTON – Children with hidradenitis suppurativa (HS) may suffer with symptoms for an average of 7 years before they are diagnosed, according to pediatric dermatologist Anna Yasmine Kirkorian, MD.
Data from a 2015 study showed that 73% of pediatric patients with HS were diagnosed more than 2 years after the onset of symptoms, said Dr. Kirkorian of the department of dermatology at Children’s National Health System and George Washington University, Washington. (Br J Dermatol. 2015 Dec;173[6]:1546-9).
Genetics can play a role in HS, likely via mutations in the gamma-secretase protein that leads to epidermal differentiation and immune regulation, Dr. Kirkorian said. Most of her patients with HS are black, and a recent study described a gamma-secretase mutation in a black family of a proband and four family members, she noted (JAMA Dermatol. 2015 Jun;151[6]:668-70). Gamma-secretase mutations also have been identified in Han Chinese populations, she said.
HS also is associated with precocious puberty. However, defining the age of onset of puberty can be difficulty because pubertal onset may vary between different ethnicities, noted Dr. Kirkorian. “Prepubertal children presenting with HS warrant an endocrinologic evaluation,” she said.
Dr. Kirkorian added that more research is needed to pinpoint the possible genetic component of HS and to identify genetic susceptibility that could lead to targeted treatment strategies.
The optimal treatment plan for pediatric HS is multimodal and addresses the comorbidities common with the condition, she said, and she predicted that specialized clinic or treatment centers that bring together areas, including psychiatry, wound care, pain management, surgery, endocrinology, and genetics, will evolve to serve these patients. To support these collaborative efforts, Dr. Kirkorian is a member of the Pediatric Dermatology Research Alliance (PeDRA), an organization formed to accelerate research on skin diseases in children.
The symposium was sponsored by AbbVie. Dr. Kirkorian had no financial conflicts to disclose. She is on the editorial board of Dermatology News.
WASHINGTON – Children with hidradenitis suppurativa (HS) may suffer with symptoms for an average of 7 years before they are diagnosed, according to pediatric dermatologist Anna Yasmine Kirkorian, MD.
Data from a 2015 study showed that 73% of pediatric patients with HS were diagnosed more than 2 years after the onset of symptoms, said Dr. Kirkorian of the department of dermatology at Children’s National Health System and George Washington University, Washington. (Br J Dermatol. 2015 Dec;173[6]:1546-9).
Genetics can play a role in HS, likely via mutations in the gamma-secretase protein that leads to epidermal differentiation and immune regulation, Dr. Kirkorian said. Most of her patients with HS are black, and a recent study described a gamma-secretase mutation in a black family of a proband and four family members, she noted (JAMA Dermatol. 2015 Jun;151[6]:668-70). Gamma-secretase mutations also have been identified in Han Chinese populations, she said.
HS also is associated with precocious puberty. However, defining the age of onset of puberty can be difficulty because pubertal onset may vary between different ethnicities, noted Dr. Kirkorian. “Prepubertal children presenting with HS warrant an endocrinologic evaluation,” she said.
Dr. Kirkorian added that more research is needed to pinpoint the possible genetic component of HS and to identify genetic susceptibility that could lead to targeted treatment strategies.
The optimal treatment plan for pediatric HS is multimodal and addresses the comorbidities common with the condition, she said, and she predicted that specialized clinic or treatment centers that bring together areas, including psychiatry, wound care, pain management, surgery, endocrinology, and genetics, will evolve to serve these patients. To support these collaborative efforts, Dr. Kirkorian is a member of the Pediatric Dermatology Research Alliance (PeDRA), an organization formed to accelerate research on skin diseases in children.
The symposium was sponsored by AbbVie. Dr. Kirkorian had no financial conflicts to disclose. She is on the editorial board of Dermatology News.
VIDEO: Hidradenitis suppurativa in pediatrics similar to that in adults
WASHINGTON– Hidradenitis suppurativa (HS) can present early in adolescence, and the approach to treatment in pediatric patients is similar to treatment in adults, “with a few caveats,” A. Yasmine Kirkorian, MD, said in a video interview at an educational session held by George Washington University.
While the literature often states that HS starts in a person’s 20s, it primarily starts in adolescence, sometimes earlier, with a presentation that is similar to that seen in adults and appearing at the same sites, such as the armpits and groin, said Dr. Kirkorian a pediatric dermatologist at Children’s National Health System and George Washington University, Washington.
“As dermatologists, we know this. Kids are getting this disease, but we need to characterize that better in the literature so that we can start to apply the adult style therapeutics in clinical trials ... to children,” she added.
In the interview, Dr. Kirkorian discussed treatment strategies, as well as familial cases of HS and the links between HS and Down syndrome and inflammatory bowel disease in children.
The meeting was sponsored by AbbVie. Dr. Kirkorian had no financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON– Hidradenitis suppurativa (HS) can present early in adolescence, and the approach to treatment in pediatric patients is similar to treatment in adults, “with a few caveats,” A. Yasmine Kirkorian, MD, said in a video interview at an educational session held by George Washington University.
While the literature often states that HS starts in a person’s 20s, it primarily starts in adolescence, sometimes earlier, with a presentation that is similar to that seen in adults and appearing at the same sites, such as the armpits and groin, said Dr. Kirkorian a pediatric dermatologist at Children’s National Health System and George Washington University, Washington.
“As dermatologists, we know this. Kids are getting this disease, but we need to characterize that better in the literature so that we can start to apply the adult style therapeutics in clinical trials ... to children,” she added.
In the interview, Dr. Kirkorian discussed treatment strategies, as well as familial cases of HS and the links between HS and Down syndrome and inflammatory bowel disease in children.
The meeting was sponsored by AbbVie. Dr. Kirkorian had no financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
WASHINGTON– Hidradenitis suppurativa (HS) can present early in adolescence, and the approach to treatment in pediatric patients is similar to treatment in adults, “with a few caveats,” A. Yasmine Kirkorian, MD, said in a video interview at an educational session held by George Washington University.
While the literature often states that HS starts in a person’s 20s, it primarily starts in adolescence, sometimes earlier, with a presentation that is similar to that seen in adults and appearing at the same sites, such as the armpits and groin, said Dr. Kirkorian a pediatric dermatologist at Children’s National Health System and George Washington University, Washington.
“As dermatologists, we know this. Kids are getting this disease, but we need to characterize that better in the literature so that we can start to apply the adult style therapeutics in clinical trials ... to children,” she added.
In the interview, Dr. Kirkorian discussed treatment strategies, as well as familial cases of HS and the links between HS and Down syndrome and inflammatory bowel disease in children.
The meeting was sponsored by AbbVie. Dr. Kirkorian had no financial conflicts to disclose.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
VIDEO: Surgery succeeds with select hidradenitis suppurativa patients
WASHINGTON – Medication has its limits for some patients with more severe hidradenitis suppurativa, and these patients can often benefit from surgical treatment, Chris Sayed, MD, said at an educational session held by George Washington University.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
“Especially if patients have relatively limited areas of sinus, being able to do some local procedures [is] what will get the patient a lot better,” Dr. Sayed of the department of dermatology at the University of North Carolina, Chapel Hill, said in a video interview. “Whereas the medicines would never have made that sinus go away.”
The meeting was supported by AbbVie. Dr. Sayed disclosed financial relationships with the company.
WASHINGTON – Medication has its limits for some patients with more severe hidradenitis suppurativa, and these patients can often benefit from surgical treatment, Chris Sayed, MD, said at an educational session held by George Washington University.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
“Especially if patients have relatively limited areas of sinus, being able to do some local procedures [is] what will get the patient a lot better,” Dr. Sayed of the department of dermatology at the University of North Carolina, Chapel Hill, said in a video interview. “Whereas the medicines would never have made that sinus go away.”
The meeting was supported by AbbVie. Dr. Sayed disclosed financial relationships with the company.
WASHINGTON – Medication has its limits for some patients with more severe hidradenitis suppurativa, and these patients can often benefit from surgical treatment, Chris Sayed, MD, said at an educational session held by George Washington University.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
“Especially if patients have relatively limited areas of sinus, being able to do some local procedures [is] what will get the patient a lot better,” Dr. Sayed of the department of dermatology at the University of North Carolina, Chapel Hill, said in a video interview. “Whereas the medicines would never have made that sinus go away.”
The meeting was supported by AbbVie. Dr. Sayed disclosed financial relationships with the company.
Risankizumab tops ustekinumab in phase II psoriasis trial
At 12 weeks, 77% of psoriasis patients treated with risankizumab showed a 90% or greater reduction in Psoriasis Area and Severity Index (PASI) score, compared with 40% of ustekinumab patients, in a phase II randomized trial.
The results were published April 19 in the New England Journal of Medicine.
Ustekinumab (Stelara), approved by the Food and Drug Administration in 2009, blocks interleukin-12 and interleukin-23 and has demonstrated effectiveness in psoriasis patients. However, the humanized IgG1 monoclonal antibody risankizumab goes farther and “selectively inhibits interleukin-23 by specifically targeting p19,” wrote Kim A. Papp, MD, PhD, of K. Papp Clinical Research and Probity Medical Research, Waterloo, Ont., and associates (N. Engl. J. Med. 2017;376:1551-60. doi: 10.1056/NEJMoa1607017).
To compare clinical response and safety, the researchers enrolled 166 adults aged 18-75 years with moderate to severe plaque psoriasis, in the phase II study. Patients were randomized to subcutaneous injections of risankizumab at one of three doses, or ustekinumab at one of two doses. Risankizumab patients received a single 18-mg dose at week 0, or 90-mg or 180-mg doses at weeks 0, 4, and 16. Ustekinumab patients weighing 100 kg or less received 45 mg at weeks 0, 4, and 16; those weighing more than 100 kg received 90 mg at weeks 0, 4, and 16. Demographics were similar among the treatment groups.
The primary end point was a 90% or greater reduction in the PASI score at week 12, compared with baseline.
In pooled results of the risankizumab 90-mg and 180-mg groups, 77% of patients achieved a PASI 90 at 12 weeks (73% of the 90-mg group and 81% of the 180-mg group), vs. 40% of ustekinumab patients (P less than .001). Complete clearance of lesions (PASI 100) occurred among risankizumab patients in 14% of the 18-mg group, 41% of the 90-mg group, and 48% of the 180-mg group, compared with 18% of the ustekinumab group.
Among risankizumab patients, the rates of adverse events through 48 weeks were 81% in the 180-mg group, 80% in the 90-mg group, and 69% in the 180-mg group, compared with 72% in those on ustekinumab, with nasopharyngitis the most commonly reported adverse event in all the treatment groups. The rates of serious adverse events were 12% and 15% among those in the 18-mg and 90-mg risankizumab groups, respectively; 0% among those on the 180-mg dose, and 8% among those on ustekinumab.
The study was not large or long enough to provide conclusive safety data on risankizumab, and additional studies are needed to review psoriasis lesions over a longer time period and include both placebo and active comparators, researchers noted. However, the results suggest that “the selective blockade of interleukin-23 through the inhibition of the p19 subunit rather than p40 provides a more complete inhibition of interleukin-23 activity, potentially resulting in a greater efficacy in the treatment of plaque psoriasis at the doses used,” they said.
The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies. Several authors are Boehringer Ingelheim employees.
At 12 weeks, 77% of psoriasis patients treated with risankizumab showed a 90% or greater reduction in Psoriasis Area and Severity Index (PASI) score, compared with 40% of ustekinumab patients, in a phase II randomized trial.
The results were published April 19 in the New England Journal of Medicine.
Ustekinumab (Stelara), approved by the Food and Drug Administration in 2009, blocks interleukin-12 and interleukin-23 and has demonstrated effectiveness in psoriasis patients. However, the humanized IgG1 monoclonal antibody risankizumab goes farther and “selectively inhibits interleukin-23 by specifically targeting p19,” wrote Kim A. Papp, MD, PhD, of K. Papp Clinical Research and Probity Medical Research, Waterloo, Ont., and associates (N. Engl. J. Med. 2017;376:1551-60. doi: 10.1056/NEJMoa1607017).
To compare clinical response and safety, the researchers enrolled 166 adults aged 18-75 years with moderate to severe plaque psoriasis, in the phase II study. Patients were randomized to subcutaneous injections of risankizumab at one of three doses, or ustekinumab at one of two doses. Risankizumab patients received a single 18-mg dose at week 0, or 90-mg or 180-mg doses at weeks 0, 4, and 16. Ustekinumab patients weighing 100 kg or less received 45 mg at weeks 0, 4, and 16; those weighing more than 100 kg received 90 mg at weeks 0, 4, and 16. Demographics were similar among the treatment groups.
The primary end point was a 90% or greater reduction in the PASI score at week 12, compared with baseline.
In pooled results of the risankizumab 90-mg and 180-mg groups, 77% of patients achieved a PASI 90 at 12 weeks (73% of the 90-mg group and 81% of the 180-mg group), vs. 40% of ustekinumab patients (P less than .001). Complete clearance of lesions (PASI 100) occurred among risankizumab patients in 14% of the 18-mg group, 41% of the 90-mg group, and 48% of the 180-mg group, compared with 18% of the ustekinumab group.
Among risankizumab patients, the rates of adverse events through 48 weeks were 81% in the 180-mg group, 80% in the 90-mg group, and 69% in the 180-mg group, compared with 72% in those on ustekinumab, with nasopharyngitis the most commonly reported adverse event in all the treatment groups. The rates of serious adverse events were 12% and 15% among those in the 18-mg and 90-mg risankizumab groups, respectively; 0% among those on the 180-mg dose, and 8% among those on ustekinumab.
The study was not large or long enough to provide conclusive safety data on risankizumab, and additional studies are needed to review psoriasis lesions over a longer time period and include both placebo and active comparators, researchers noted. However, the results suggest that “the selective blockade of interleukin-23 through the inhibition of the p19 subunit rather than p40 provides a more complete inhibition of interleukin-23 activity, potentially resulting in a greater efficacy in the treatment of plaque psoriasis at the doses used,” they said.
The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies. Several authors are Boehringer Ingelheim employees.
At 12 weeks, 77% of psoriasis patients treated with risankizumab showed a 90% or greater reduction in Psoriasis Area and Severity Index (PASI) score, compared with 40% of ustekinumab patients, in a phase II randomized trial.
The results were published April 19 in the New England Journal of Medicine.
Ustekinumab (Stelara), approved by the Food and Drug Administration in 2009, blocks interleukin-12 and interleukin-23 and has demonstrated effectiveness in psoriasis patients. However, the humanized IgG1 monoclonal antibody risankizumab goes farther and “selectively inhibits interleukin-23 by specifically targeting p19,” wrote Kim A. Papp, MD, PhD, of K. Papp Clinical Research and Probity Medical Research, Waterloo, Ont., and associates (N. Engl. J. Med. 2017;376:1551-60. doi: 10.1056/NEJMoa1607017).
To compare clinical response and safety, the researchers enrolled 166 adults aged 18-75 years with moderate to severe plaque psoriasis, in the phase II study. Patients were randomized to subcutaneous injections of risankizumab at one of three doses, or ustekinumab at one of two doses. Risankizumab patients received a single 18-mg dose at week 0, or 90-mg or 180-mg doses at weeks 0, 4, and 16. Ustekinumab patients weighing 100 kg or less received 45 mg at weeks 0, 4, and 16; those weighing more than 100 kg received 90 mg at weeks 0, 4, and 16. Demographics were similar among the treatment groups.
The primary end point was a 90% or greater reduction in the PASI score at week 12, compared with baseline.
In pooled results of the risankizumab 90-mg and 180-mg groups, 77% of patients achieved a PASI 90 at 12 weeks (73% of the 90-mg group and 81% of the 180-mg group), vs. 40% of ustekinumab patients (P less than .001). Complete clearance of lesions (PASI 100) occurred among risankizumab patients in 14% of the 18-mg group, 41% of the 90-mg group, and 48% of the 180-mg group, compared with 18% of the ustekinumab group.
Among risankizumab patients, the rates of adverse events through 48 weeks were 81% in the 180-mg group, 80% in the 90-mg group, and 69% in the 180-mg group, compared with 72% in those on ustekinumab, with nasopharyngitis the most commonly reported adverse event in all the treatment groups. The rates of serious adverse events were 12% and 15% among those in the 18-mg and 90-mg risankizumab groups, respectively; 0% among those on the 180-mg dose, and 8% among those on ustekinumab.
The study was not large or long enough to provide conclusive safety data on risankizumab, and additional studies are needed to review psoriasis lesions over a longer time period and include both placebo and active comparators, researchers noted. However, the results suggest that “the selective blockade of interleukin-23 through the inhibition of the p19 subunit rather than p40 provides a more complete inhibition of interleukin-23 activity, potentially resulting in a greater efficacy in the treatment of plaque psoriasis at the doses used,” they said.
The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies. Several authors are Boehringer Ingelheim employees.
Key clinical point: Clinical responses in psoriasis patients treated with risankizumab were superior to responses in patients treated with ustekinumab.
Major finding: At 12 weeks, 77% of risankizumab patients showed a 90% or greater reduction in PASI score, compared with 40% of ustekinumab patients.
Data source: A phase II randomized trial of 166 adults with moderate to severe plaque psoriasis.
Disclosures: The study was supported by Boehringer Ingelheim. Several study coauthors, including lead author Dr. Papp, disclosed relationships with Boehringer Ingelheim and other companies; several were employees of Boehringer.
Septic shock patients suffer most from delayed antibiotics
Hospital mortality for sepsis patients was 9% more likely with each hour of delayed administration of antibiotics, and the mortality rates increased with the severity of sepsis, based on data from 35,000 randomly selected sepsis patients.
Early administration of antibiotics in sepsis cases has become accepted as a way to improve outcomes, but the benefits have not been well studied, wrote Vincent X Liu, MD, MS, of Kaiser Permanente Division of Research, Oakland, Calif., and his colleagues.
To quantify the impact of antibiotic timing on mortality rates in different types of sepsis patients, the researchers reviewed data from 35,000 adults treated for sepsis at 21 emergency departments in northern California between 2010 and 2013. The time from registration at the emergency department to administration of the first antibiotics was less than 6 hours (Am J Respir Crit Care Med. 2017 March 27. doi: 10.1164/rccm.201609-1848OC).
The overall mortality rates were 3.9%, 8.8%, and 26.0% for sepsis, severe sepsis, and septic shock, respectively. Absolute mortality increased by 0.3% for sepsis, 0.4% for severe sepsis, and 1.8% for septic shock patients after an hour’s delay in the administration of antibiotics, and the adjusted odds ratio for hospital mortality was 1.09 for each hour between patient registration and antibiotic administration. The median time to the first administration of antibiotics was 2.1 hours, ranging from 1.7 hours for septic shock patients to 2.3 hours for sepsis patients, with ceftriaxone having been the most commonly used antibiotic across all groups.
Approximately 42% of patients received one antibiotic and 43% received two antibiotics. The odds of receiving two or more antibiotics were significantly higher for septic shock patients compared with sepsis patients (72% vs. 52%, respectively).
The findings were limited by several factors, including the inability to adjust for concomitant sepsis treatments and preexisting antibiotic treatments, the researchers said.
The study results do not resolve all questions about the timing of antibiotic administration for sepsis patients, such as whether there is additional benefit to giving the medications at 2 hours rather than 3 hours or 4 hours after ED admission, the researchers noted. However, “our findings support currently held beliefs that administering early antibiotics to infected patients with systemic inflammation is beneficial for reducing mortality,” they said.
The study was supported in part by the Permanente Medical Group and Kaiser Foundation Hospitals, the National Institute of General Medical Sciences, and the Veterans Affairs Health Services Research and Development Service.
Hospital mortality for sepsis patients was 9% more likely with each hour of delayed administration of antibiotics, and the mortality rates increased with the severity of sepsis, based on data from 35,000 randomly selected sepsis patients.
Early administration of antibiotics in sepsis cases has become accepted as a way to improve outcomes, but the benefits have not been well studied, wrote Vincent X Liu, MD, MS, of Kaiser Permanente Division of Research, Oakland, Calif., and his colleagues.
To quantify the impact of antibiotic timing on mortality rates in different types of sepsis patients, the researchers reviewed data from 35,000 adults treated for sepsis at 21 emergency departments in northern California between 2010 and 2013. The time from registration at the emergency department to administration of the first antibiotics was less than 6 hours (Am J Respir Crit Care Med. 2017 March 27. doi: 10.1164/rccm.201609-1848OC).
The overall mortality rates were 3.9%, 8.8%, and 26.0% for sepsis, severe sepsis, and septic shock, respectively. Absolute mortality increased by 0.3% for sepsis, 0.4% for severe sepsis, and 1.8% for septic shock patients after an hour’s delay in the administration of antibiotics, and the adjusted odds ratio for hospital mortality was 1.09 for each hour between patient registration and antibiotic administration. The median time to the first administration of antibiotics was 2.1 hours, ranging from 1.7 hours for septic shock patients to 2.3 hours for sepsis patients, with ceftriaxone having been the most commonly used antibiotic across all groups.
Approximately 42% of patients received one antibiotic and 43% received two antibiotics. The odds of receiving two or more antibiotics were significantly higher for septic shock patients compared with sepsis patients (72% vs. 52%, respectively).
The findings were limited by several factors, including the inability to adjust for concomitant sepsis treatments and preexisting antibiotic treatments, the researchers said.
The study results do not resolve all questions about the timing of antibiotic administration for sepsis patients, such as whether there is additional benefit to giving the medications at 2 hours rather than 3 hours or 4 hours after ED admission, the researchers noted. However, “our findings support currently held beliefs that administering early antibiotics to infected patients with systemic inflammation is beneficial for reducing mortality,” they said.
The study was supported in part by the Permanente Medical Group and Kaiser Foundation Hospitals, the National Institute of General Medical Sciences, and the Veterans Affairs Health Services Research and Development Service.
Hospital mortality for sepsis patients was 9% more likely with each hour of delayed administration of antibiotics, and the mortality rates increased with the severity of sepsis, based on data from 35,000 randomly selected sepsis patients.
Early administration of antibiotics in sepsis cases has become accepted as a way to improve outcomes, but the benefits have not been well studied, wrote Vincent X Liu, MD, MS, of Kaiser Permanente Division of Research, Oakland, Calif., and his colleagues.
To quantify the impact of antibiotic timing on mortality rates in different types of sepsis patients, the researchers reviewed data from 35,000 adults treated for sepsis at 21 emergency departments in northern California between 2010 and 2013. The time from registration at the emergency department to administration of the first antibiotics was less than 6 hours (Am J Respir Crit Care Med. 2017 March 27. doi: 10.1164/rccm.201609-1848OC).
The overall mortality rates were 3.9%, 8.8%, and 26.0% for sepsis, severe sepsis, and septic shock, respectively. Absolute mortality increased by 0.3% for sepsis, 0.4% for severe sepsis, and 1.8% for septic shock patients after an hour’s delay in the administration of antibiotics, and the adjusted odds ratio for hospital mortality was 1.09 for each hour between patient registration and antibiotic administration. The median time to the first administration of antibiotics was 2.1 hours, ranging from 1.7 hours for septic shock patients to 2.3 hours for sepsis patients, with ceftriaxone having been the most commonly used antibiotic across all groups.
Approximately 42% of patients received one antibiotic and 43% received two antibiotics. The odds of receiving two or more antibiotics were significantly higher for septic shock patients compared with sepsis patients (72% vs. 52%, respectively).
The findings were limited by several factors, including the inability to adjust for concomitant sepsis treatments and preexisting antibiotic treatments, the researchers said.
The study results do not resolve all questions about the timing of antibiotic administration for sepsis patients, such as whether there is additional benefit to giving the medications at 2 hours rather than 3 hours or 4 hours after ED admission, the researchers noted. However, “our findings support currently held beliefs that administering early antibiotics to infected patients with systemic inflammation is beneficial for reducing mortality,” they said.
The study was supported in part by the Permanente Medical Group and Kaiser Foundation Hospitals, the National Institute of General Medical Sciences, and the Veterans Affairs Health Services Research and Development Service.
FROM THE AMERICAN JOURNAL OF RESPIRATORY AND CRITICAL CARE MEDICINE
Key clinical point: Each hour of delay in antibiotic administration increased the odds of hospital mortality in sepsis patients.
Major finding: An hour delay in antibiotic administration increased the absolute mortality by 0.3% for cases of sepsis, 0.4% for severe sepsis, and 1.8% for septic shock.
Data source: A retrospective study of 35,000 adult patients treated with a sepsis quality improvement program at 21 emergency departments in California.
Disclosures: The study was supported in part by the Permanente Medical Group and Kaiser Foundation Hospitals, the National Institute of General Medical Sciences, and the Veterans Affairs Health Services Research and Development Service.