User login
Golimumab earns new FDA approvals
The U.S. Food and Drug Administration has approved golimumab (Simponi Aria) for use in adults with active psoriatic arthritis (PsA) or active ankylosing spondylitis (AS).
Simponi Aria is an intravenous formulation of golimumab that is already approved for moderate to severe rheumatoid arthritis. The subcutaneous injection formulation of golimumab, Simponi, is already approved for RA, PsA, AS, and ulcerative colitis. Golimumab is a fully human anti–tumor necrosis factor-alpha therapy, and the intravenous formulation is designed for use as a 30-minute infusion.
“In the study for the treatment of active PsA, patients experienced improvement in joint symptoms and inhibition of structural damage. In the study for treatment of active AS, results showed improvement in measures of disease activity,” according to an Oct. 20 announcement from the manufacturer of golimumab, Janssen Biotech.
Read the revised prescribing information for Simponi Aria here.
The U.S. Food and Drug Administration has approved golimumab (Simponi Aria) for use in adults with active psoriatic arthritis (PsA) or active ankylosing spondylitis (AS).
Simponi Aria is an intravenous formulation of golimumab that is already approved for moderate to severe rheumatoid arthritis. The subcutaneous injection formulation of golimumab, Simponi, is already approved for RA, PsA, AS, and ulcerative colitis. Golimumab is a fully human anti–tumor necrosis factor-alpha therapy, and the intravenous formulation is designed for use as a 30-minute infusion.
“In the study for the treatment of active PsA, patients experienced improvement in joint symptoms and inhibition of structural damage. In the study for treatment of active AS, results showed improvement in measures of disease activity,” according to an Oct. 20 announcement from the manufacturer of golimumab, Janssen Biotech.
Read the revised prescribing information for Simponi Aria here.
The U.S. Food and Drug Administration has approved golimumab (Simponi Aria) for use in adults with active psoriatic arthritis (PsA) or active ankylosing spondylitis (AS).
Simponi Aria is an intravenous formulation of golimumab that is already approved for moderate to severe rheumatoid arthritis. The subcutaneous injection formulation of golimumab, Simponi, is already approved for RA, PsA, AS, and ulcerative colitis. Golimumab is a fully human anti–tumor necrosis factor-alpha therapy, and the intravenous formulation is designed for use as a 30-minute infusion.
“In the study for the treatment of active PsA, patients experienced improvement in joint symptoms and inhibition of structural damage. In the study for treatment of active AS, results showed improvement in measures of disease activity,” according to an Oct. 20 announcement from the manufacturer of golimumab, Janssen Biotech.
Read the revised prescribing information for Simponi Aria here.
Genetic analysis indicates ovarian cancer may originate in fallopian tubes
Many of the most severe ovarian cancer cases may originate in the fallopian tube (FT), based on data from an analysis of nine patients published online in Nature Communications.
“Our data suggest that FT neoplasia is the origin of ovarian serous carcinogenesis, and can directly lead to cancer of the ovaries and of other sites,” wrote Sana Intidhar Labidi-Galy, MD, of Dana-Farber Cancer Institute, Boston, and her colleagues (Nature Commun. 2017 Oct 23. doi: 10.1038/s41467-017-00962-1).
Preliminary evidence suggests that fallopian tube cancers may develop into high-grade serous ovarian carcinoma (HGSOC), but evolutionary evidence is limited, the researchers said.
They conducted genetic sequencing on 37 tumor samples from five adult patients with HGSOC. They identified changes in the TP53 tumor suppressor gene in all cases of HGSOC. They also studied serous tubal intraepithelial carcinomas from four patients.
“As expected, we identified sequence changes in the TP53 tumor suppressor gene, a well-known driver gene in HGSOC, in all cases,” the researchers wrote.
“The TP53 alterations were identical in all samples analyzed for each patient including in the p53 signatures, the [serous tubal intraepithelial carcinoma] lesions, and other carcinomas,” Dr. Labidi-Galy and her associates said. Although TP53 was the only gene analyzed in this study, the researchers also noted changes in areas of several known ovarian cancer genes, including BRCA1 and BRCA2.
The study findings were limited by the small size of the tumor samples and small number of cells, the researchers noted.
The results, however, suggest an avenue for further research to help guide early detection and treatment of ovarian cancer, such as the potential removal of fallopian tubes rather than the ovaries in some cases, they concluded.
The research was supported by multiple foundations and organizations, including the National Institutes of Health. One of the investigators is a founder of Personal Genome Diagnostics and a member of its scientific advisory board and board of directors. The other researchers had no financial conflicts to disclose.
Many of the most severe ovarian cancer cases may originate in the fallopian tube (FT), based on data from an analysis of nine patients published online in Nature Communications.
“Our data suggest that FT neoplasia is the origin of ovarian serous carcinogenesis, and can directly lead to cancer of the ovaries and of other sites,” wrote Sana Intidhar Labidi-Galy, MD, of Dana-Farber Cancer Institute, Boston, and her colleagues (Nature Commun. 2017 Oct 23. doi: 10.1038/s41467-017-00962-1).
Preliminary evidence suggests that fallopian tube cancers may develop into high-grade serous ovarian carcinoma (HGSOC), but evolutionary evidence is limited, the researchers said.
They conducted genetic sequencing on 37 tumor samples from five adult patients with HGSOC. They identified changes in the TP53 tumor suppressor gene in all cases of HGSOC. They also studied serous tubal intraepithelial carcinomas from four patients.
“As expected, we identified sequence changes in the TP53 tumor suppressor gene, a well-known driver gene in HGSOC, in all cases,” the researchers wrote.
“The TP53 alterations were identical in all samples analyzed for each patient including in the p53 signatures, the [serous tubal intraepithelial carcinoma] lesions, and other carcinomas,” Dr. Labidi-Galy and her associates said. Although TP53 was the only gene analyzed in this study, the researchers also noted changes in areas of several known ovarian cancer genes, including BRCA1 and BRCA2.
The study findings were limited by the small size of the tumor samples and small number of cells, the researchers noted.
The results, however, suggest an avenue for further research to help guide early detection and treatment of ovarian cancer, such as the potential removal of fallopian tubes rather than the ovaries in some cases, they concluded.
The research was supported by multiple foundations and organizations, including the National Institutes of Health. One of the investigators is a founder of Personal Genome Diagnostics and a member of its scientific advisory board and board of directors. The other researchers had no financial conflicts to disclose.
Many of the most severe ovarian cancer cases may originate in the fallopian tube (FT), based on data from an analysis of nine patients published online in Nature Communications.
“Our data suggest that FT neoplasia is the origin of ovarian serous carcinogenesis, and can directly lead to cancer of the ovaries and of other sites,” wrote Sana Intidhar Labidi-Galy, MD, of Dana-Farber Cancer Institute, Boston, and her colleagues (Nature Commun. 2017 Oct 23. doi: 10.1038/s41467-017-00962-1).
Preliminary evidence suggests that fallopian tube cancers may develop into high-grade serous ovarian carcinoma (HGSOC), but evolutionary evidence is limited, the researchers said.
They conducted genetic sequencing on 37 tumor samples from five adult patients with HGSOC. They identified changes in the TP53 tumor suppressor gene in all cases of HGSOC. They also studied serous tubal intraepithelial carcinomas from four patients.
“As expected, we identified sequence changes in the TP53 tumor suppressor gene, a well-known driver gene in HGSOC, in all cases,” the researchers wrote.
“The TP53 alterations were identical in all samples analyzed for each patient including in the p53 signatures, the [serous tubal intraepithelial carcinoma] lesions, and other carcinomas,” Dr. Labidi-Galy and her associates said. Although TP53 was the only gene analyzed in this study, the researchers also noted changes in areas of several known ovarian cancer genes, including BRCA1 and BRCA2.
The study findings were limited by the small size of the tumor samples and small number of cells, the researchers noted.
The results, however, suggest an avenue for further research to help guide early detection and treatment of ovarian cancer, such as the potential removal of fallopian tubes rather than the ovaries in some cases, they concluded.
The research was supported by multiple foundations and organizations, including the National Institutes of Health. One of the investigators is a founder of Personal Genome Diagnostics and a member of its scientific advisory board and board of directors. The other researchers had no financial conflicts to disclose.
FROM NATURE COMMUNICATIONS
FDA panel advises approval of semaglutide to lower HbA1c in patients with type 2 diabetes
The Food and drug Administration’s Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC) recommended the approval of a once-weekly semaglutide injection for adults with type 2 diabetes mellitus. The vote was 16-0 in favor of approval, with one committee member abstaining.
The committee met on Oct. 18 and discussed the safety and efficacy of new drug application (NDA) 209637 for semaglutide injection when used to help glycemic control in addition to diet and exercise. Semaglutide, manufactured and submitted for approval by Novo Nordisk, is described by the company as “an investigational analog of native human glucagonlike peptide–1,” with a half-life of approximately 1 week, making the agent appropriate for weekly dosing.
The researchers found some increase in diabetic retinopathy in the SUSTAIN trials, but a post-hoc analysis found that “To the extent that the data suggest a signal that there was progression of diabetic retinopathy in patients with significant decreases in HbA1c, these events should be expected because they are consistent with treatments that decrease HbA1c. While this decrease may result in an initial increase in retinopathy, ocular health is ultimately benefited by decreasing HbA1c.”
Novo Nordisk submitted the application for semaglutide in December 2016; the drug also is being reviewed in Europe and Japan.
The FDA is not obligated to follow the committee’s recommendation but considers it as part of the review process of new drug applications.
The Food and drug Administration’s Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC) recommended the approval of a once-weekly semaglutide injection for adults with type 2 diabetes mellitus. The vote was 16-0 in favor of approval, with one committee member abstaining.
The committee met on Oct. 18 and discussed the safety and efficacy of new drug application (NDA) 209637 for semaglutide injection when used to help glycemic control in addition to diet and exercise. Semaglutide, manufactured and submitted for approval by Novo Nordisk, is described by the company as “an investigational analog of native human glucagonlike peptide–1,” with a half-life of approximately 1 week, making the agent appropriate for weekly dosing.
The researchers found some increase in diabetic retinopathy in the SUSTAIN trials, but a post-hoc analysis found that “To the extent that the data suggest a signal that there was progression of diabetic retinopathy in patients with significant decreases in HbA1c, these events should be expected because they are consistent with treatments that decrease HbA1c. While this decrease may result in an initial increase in retinopathy, ocular health is ultimately benefited by decreasing HbA1c.”
Novo Nordisk submitted the application for semaglutide in December 2016; the drug also is being reviewed in Europe and Japan.
The FDA is not obligated to follow the committee’s recommendation but considers it as part of the review process of new drug applications.
The Food and drug Administration’s Endocrinologic and Metabolic Drugs Advisory Committee (EMDAC) recommended the approval of a once-weekly semaglutide injection for adults with type 2 diabetes mellitus. The vote was 16-0 in favor of approval, with one committee member abstaining.
The committee met on Oct. 18 and discussed the safety and efficacy of new drug application (NDA) 209637 for semaglutide injection when used to help glycemic control in addition to diet and exercise. Semaglutide, manufactured and submitted for approval by Novo Nordisk, is described by the company as “an investigational analog of native human glucagonlike peptide–1,” with a half-life of approximately 1 week, making the agent appropriate for weekly dosing.
The researchers found some increase in diabetic retinopathy in the SUSTAIN trials, but a post-hoc analysis found that “To the extent that the data suggest a signal that there was progression of diabetic retinopathy in patients with significant decreases in HbA1c, these events should be expected because they are consistent with treatments that decrease HbA1c. While this decrease may result in an initial increase in retinopathy, ocular health is ultimately benefited by decreasing HbA1c.”
Novo Nordisk submitted the application for semaglutide in December 2016; the drug also is being reviewed in Europe and Japan.
The FDA is not obligated to follow the committee’s recommendation but considers it as part of the review process of new drug applications.
AT AN FDA ADVISORY COMMITTEE MEETING
Guidelines cut acute chest syndrome hospital returns in pediatric sickle cell
Children with sickle cell disease who experience acute chest syndrome benefit from the current guideline-recommended antibiotic regimen, based on data from more than 7,000 patients.
Although acute chest syndrome (ACS) is among the most common complications of sickle cell disease (SCD), data on the effectiveness of the recommended antibiotic therapies (macrolides and cephalosporins) are lacking, wrote David G. Bundy, MD, of the Medical University of South Carolina, Charleston, and colleagues. ACS often leads to intensive hospital care and 1%-2% morbidity, they noted.
The most recent guidelines from the National Heart, Lung, and Blood Institute call for “an intravenous cephalosporin and an oral macrolide antibiotic,” the researchers said.
To determine the impact of antibiotic use as directed on reducing hospital readmissions in young SCD patients, the researchers reviewed data from 14,480 hospitalizations for ACS involving 7,178 children and young adults aged 0-22 years seen at 41 hospitals in the United States (JAMA Pediatr. 2017 Sep 11. doi: 10.1001/jamapediatrics.2017.2526).
“This high level of interhospital variation also suggests possible clinician disagreement regarding the ideal antibiotic treatment for children with ACS,” the researchers wrote.
Rates of all-cause readmission and 30-day ACS-related readmission were significantly lower among patients who received the recommended antibiotics (odds ratio, 0.50 and 0.71, respectively). Children aged 5-9 years were most likely to receive the recommended antibiotics (80%), while young adults aged 19-22 years were the least likely (64%).
The findings were limited by several factors, including coding errors and incomplete clinical information, the researchers noted. But the results suggest that the guideline-recommended antibiotics are effective, “so more robust dissemination and implementation of existing treatment guidelines may reduce readmissions in this high-risk population,” they said.
The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.
Children with sickle cell disease who experience acute chest syndrome benefit from the current guideline-recommended antibiotic regimen, based on data from more than 7,000 patients.
Although acute chest syndrome (ACS) is among the most common complications of sickle cell disease (SCD), data on the effectiveness of the recommended antibiotic therapies (macrolides and cephalosporins) are lacking, wrote David G. Bundy, MD, of the Medical University of South Carolina, Charleston, and colleagues. ACS often leads to intensive hospital care and 1%-2% morbidity, they noted.
The most recent guidelines from the National Heart, Lung, and Blood Institute call for “an intravenous cephalosporin and an oral macrolide antibiotic,” the researchers said.
To determine the impact of antibiotic use as directed on reducing hospital readmissions in young SCD patients, the researchers reviewed data from 14,480 hospitalizations for ACS involving 7,178 children and young adults aged 0-22 years seen at 41 hospitals in the United States (JAMA Pediatr. 2017 Sep 11. doi: 10.1001/jamapediatrics.2017.2526).
“This high level of interhospital variation also suggests possible clinician disagreement regarding the ideal antibiotic treatment for children with ACS,” the researchers wrote.
Rates of all-cause readmission and 30-day ACS-related readmission were significantly lower among patients who received the recommended antibiotics (odds ratio, 0.50 and 0.71, respectively). Children aged 5-9 years were most likely to receive the recommended antibiotics (80%), while young adults aged 19-22 years were the least likely (64%).
The findings were limited by several factors, including coding errors and incomplete clinical information, the researchers noted. But the results suggest that the guideline-recommended antibiotics are effective, “so more robust dissemination and implementation of existing treatment guidelines may reduce readmissions in this high-risk population,” they said.
The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.
Children with sickle cell disease who experience acute chest syndrome benefit from the current guideline-recommended antibiotic regimen, based on data from more than 7,000 patients.
Although acute chest syndrome (ACS) is among the most common complications of sickle cell disease (SCD), data on the effectiveness of the recommended antibiotic therapies (macrolides and cephalosporins) are lacking, wrote David G. Bundy, MD, of the Medical University of South Carolina, Charleston, and colleagues. ACS often leads to intensive hospital care and 1%-2% morbidity, they noted.
The most recent guidelines from the National Heart, Lung, and Blood Institute call for “an intravenous cephalosporin and an oral macrolide antibiotic,” the researchers said.
To determine the impact of antibiotic use as directed on reducing hospital readmissions in young SCD patients, the researchers reviewed data from 14,480 hospitalizations for ACS involving 7,178 children and young adults aged 0-22 years seen at 41 hospitals in the United States (JAMA Pediatr. 2017 Sep 11. doi: 10.1001/jamapediatrics.2017.2526).
“This high level of interhospital variation also suggests possible clinician disagreement regarding the ideal antibiotic treatment for children with ACS,” the researchers wrote.
Rates of all-cause readmission and 30-day ACS-related readmission were significantly lower among patients who received the recommended antibiotics (odds ratio, 0.50 and 0.71, respectively). Children aged 5-9 years were most likely to receive the recommended antibiotics (80%), while young adults aged 19-22 years were the least likely (64%).
The findings were limited by several factors, including coding errors and incomplete clinical information, the researchers noted. But the results suggest that the guideline-recommended antibiotics are effective, “so more robust dissemination and implementation of existing treatment guidelines may reduce readmissions in this high-risk population,” they said.
The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.
FROM JAMA PEDIATRICS
Key clinical point: Treatment with the recommended antibiotics was effective in reducing hospital readmissions for acute chest syndrome in children and young adults up to age 22 years with sickle cell disease.
Major finding: Hospital readmission for 30-day acute chest syndrome and all-cause readmission were significantly lower (odds ratio, 0.71 and 0.50, respectively) among children with sickle cell disease who received antibiotics (macrolides and cephalosporins) according to current guidelines, compared with those who did not.
Data source: A retrospective, multicenter study of 14,480 hospitalizations at 41 locations involving 7,178 children and young adults aged 0-22 years.
Disclosures: The researchers had no financial conflicts to disclose. Study coauthor Staci Arnold, MD, was supported in part by the Robert Wood Johnson Foundation Harold Amos Medical Faculty Development Program.
Metals may surprise as sources of contact dermatitis
, according to Jennifer H. Perryman, MD, of the Greeley Skin Clinic in Fort Collins, Colo.
For example, metal from orthopedic implants can cause contact dermatitis, Dr. Perryman said at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
The cutaneous complications of metal implants generally are eczematous, but they can be urticarial and vasculitic as well, with symptoms either generalized or localized. Dr. Perryman explained. Noncutaneous complications from contact dermatitis associated with the metal include chronic joint pain, and a loosening and dysfunction of the device.
It is a case of “chicken or the egg: Metal allergy causes device failure, or device failure causes metal allergy,” Dr. Perryman said.
Dental implants also can be unforeseen causes of contact dermatitis, she noted. The bone cement used in some implants may contain a variety of potential irritants such as methyl methacrylate, N,N-dimethyl-p-toluidine (DPT), benzoyl peroxide, gentamicin, and hydroquinone.
Metal allergy in the mouth most often presents as a reaction resembling oral lichen planus, with lesions that are reticular, atrophic, erosive, or plaque-like. These lesions usually erupt next to the implant, she said. Some patients also experience burning mouth syndrome from amalgam tattoos. However, some patients who test positive for metal allergies in general have developed a tolerance for dental implants as a result of having worn braces in the past.
Metal eyelid weights implanted to treat lagophthalmos are another rare, but potential allergen to consider, said Dr. Perryman. These weights often are made of gold, and Dr. Perryman cited a study in which four patients with gold eyelid weights experienced inflammatory reactions. Patch testing revealed gold sodium thiosulfate as the cause of their allergic contact dermatitis (Dermatitis. 2008 May-Jun;19[3]:148-53). Other options for these patients include platinum weights, hyaluronic acid, ointment, and taping, she said.
Dr. Perryman had no financial conflicts to disclose. SDEF and this news organization are owned by Frontline Medical Communications.
, according to Jennifer H. Perryman, MD, of the Greeley Skin Clinic in Fort Collins, Colo.
For example, metal from orthopedic implants can cause contact dermatitis, Dr. Perryman said at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
The cutaneous complications of metal implants generally are eczematous, but they can be urticarial and vasculitic as well, with symptoms either generalized or localized. Dr. Perryman explained. Noncutaneous complications from contact dermatitis associated with the metal include chronic joint pain, and a loosening and dysfunction of the device.
It is a case of “chicken or the egg: Metal allergy causes device failure, or device failure causes metal allergy,” Dr. Perryman said.
Dental implants also can be unforeseen causes of contact dermatitis, she noted. The bone cement used in some implants may contain a variety of potential irritants such as methyl methacrylate, N,N-dimethyl-p-toluidine (DPT), benzoyl peroxide, gentamicin, and hydroquinone.
Metal allergy in the mouth most often presents as a reaction resembling oral lichen planus, with lesions that are reticular, atrophic, erosive, or plaque-like. These lesions usually erupt next to the implant, she said. Some patients also experience burning mouth syndrome from amalgam tattoos. However, some patients who test positive for metal allergies in general have developed a tolerance for dental implants as a result of having worn braces in the past.
Metal eyelid weights implanted to treat lagophthalmos are another rare, but potential allergen to consider, said Dr. Perryman. These weights often are made of gold, and Dr. Perryman cited a study in which four patients with gold eyelid weights experienced inflammatory reactions. Patch testing revealed gold sodium thiosulfate as the cause of their allergic contact dermatitis (Dermatitis. 2008 May-Jun;19[3]:148-53). Other options for these patients include platinum weights, hyaluronic acid, ointment, and taping, she said.
Dr. Perryman had no financial conflicts to disclose. SDEF and this news organization are owned by Frontline Medical Communications.
, according to Jennifer H. Perryman, MD, of the Greeley Skin Clinic in Fort Collins, Colo.
For example, metal from orthopedic implants can cause contact dermatitis, Dr. Perryman said at Skin Disease Education Foundation’s Women’s & Pediatric Dermatology Seminar.
The cutaneous complications of metal implants generally are eczematous, but they can be urticarial and vasculitic as well, with symptoms either generalized or localized. Dr. Perryman explained. Noncutaneous complications from contact dermatitis associated with the metal include chronic joint pain, and a loosening and dysfunction of the device.
It is a case of “chicken or the egg: Metal allergy causes device failure, or device failure causes metal allergy,” Dr. Perryman said.
Dental implants also can be unforeseen causes of contact dermatitis, she noted. The bone cement used in some implants may contain a variety of potential irritants such as methyl methacrylate, N,N-dimethyl-p-toluidine (DPT), benzoyl peroxide, gentamicin, and hydroquinone.
Metal allergy in the mouth most often presents as a reaction resembling oral lichen planus, with lesions that are reticular, atrophic, erosive, or plaque-like. These lesions usually erupt next to the implant, she said. Some patients also experience burning mouth syndrome from amalgam tattoos. However, some patients who test positive for metal allergies in general have developed a tolerance for dental implants as a result of having worn braces in the past.
Metal eyelid weights implanted to treat lagophthalmos are another rare, but potential allergen to consider, said Dr. Perryman. These weights often are made of gold, and Dr. Perryman cited a study in which four patients with gold eyelid weights experienced inflammatory reactions. Patch testing revealed gold sodium thiosulfate as the cause of their allergic contact dermatitis (Dermatitis. 2008 May-Jun;19[3]:148-53). Other options for these patients include platinum weights, hyaluronic acid, ointment, and taping, she said.
Dr. Perryman had no financial conflicts to disclose. SDEF and this news organization are owned by Frontline Medical Communications.
FROM SDEF WOMEN’S & PEDIATRIC DERMATOLOGY SEMINAR
Counsel fair-skinned patients on cancer prevention, says task force
The U.S. Preventive Services Task Force has recommended that clinicians counsel fair-skinned young adults, adolescents, children, and parents of young children about minimizing exposure to ultraviolet radiation to reduce their risk of skin cancer, in a draft recommendation statement that is available online.
The grade B recommendation applies to asymptomatic individuals who have fair skin, are aged 6 months to 24 years, and have no history of skin cancer; the recommendation is being issued because members of this population are at increased risk and are the subject of most of the existing research on skin cancer counseling, according to the USPSTF. The task force found a moderate net benefit when clinicians in a primary care setting offered behavioral counseling on skin cancer prevention to members of this population.
The draft recommendation is open for public comment until 8:00 p.m. Eastern Standard Time on Nov. 6, 2017.
The draft recommendation can be viewed and comments can be submitted online at the USPSTF site.
The U.S. Preventive Services Task Force has recommended that clinicians counsel fair-skinned young adults, adolescents, children, and parents of young children about minimizing exposure to ultraviolet radiation to reduce their risk of skin cancer, in a draft recommendation statement that is available online.
The grade B recommendation applies to asymptomatic individuals who have fair skin, are aged 6 months to 24 years, and have no history of skin cancer; the recommendation is being issued because members of this population are at increased risk and are the subject of most of the existing research on skin cancer counseling, according to the USPSTF. The task force found a moderate net benefit when clinicians in a primary care setting offered behavioral counseling on skin cancer prevention to members of this population.
The draft recommendation is open for public comment until 8:00 p.m. Eastern Standard Time on Nov. 6, 2017.
The draft recommendation can be viewed and comments can be submitted online at the USPSTF site.
The U.S. Preventive Services Task Force has recommended that clinicians counsel fair-skinned young adults, adolescents, children, and parents of young children about minimizing exposure to ultraviolet radiation to reduce their risk of skin cancer, in a draft recommendation statement that is available online.
The grade B recommendation applies to asymptomatic individuals who have fair skin, are aged 6 months to 24 years, and have no history of skin cancer; the recommendation is being issued because members of this population are at increased risk and are the subject of most of the existing research on skin cancer counseling, according to the USPSTF. The task force found a moderate net benefit when clinicians in a primary care setting offered behavioral counseling on skin cancer prevention to members of this population.
The draft recommendation is open for public comment until 8:00 p.m. Eastern Standard Time on Nov. 6, 2017.
The draft recommendation can be viewed and comments can be submitted online at the USPSTF site.
FROM USPSTF
Nearly 80% of health care personnel stepped up for flu shots
Nearly four out of five health care personnel in the United States received a flu vaccination during the 2016-2017 flu season, but a majority of those working in long-term care settings were not vaccinated, based on data from an Internet survey of more than 2,000 individuals that was conducted by the Centers for Disease Control and Prevention.
A total of 78.6% of the survey’s respondents said they’d been vaccinated during the 2016-2017 season. Vaccination coverage for health care personnel overall has remained in the 77%-79% range in recent years, but that represents an increase from 64% in 2010-2011.
“As in previous seasons, the highest coverage was among HCP whose workplace had vaccination requirements,” noted Carla L. Black, PhD, of the CDC, and colleagues (MMWR Morb Mortal Wkly Rep. 2017 Sep 29;66[38]:1009-15). The researchers reviewed data collected from an Internet panel survey of 2,438 health care personnel between March 28, 2017, and April 19, 2017.
Physicians boasted the highest vaccination coverage in 2016-2017 (96%), followed by pharmacists (94%), nurses (93%), nurse practitioners and physician assistants (92%), other clinical providers (80%), nonclinical health care providers (74%), and aides and assistants (69%).
Flu vaccination rates were highest among HCPs working in a hospital setting (92%); 94% of survey respondents in hospitals reported either having a vaccination requirement at work or being provided at least 1 day of on-site vaccination.
Vaccination rates were lowest among health care personnel in long-term care settings (68%), where only 26% reported a workplace vaccination requirement. However, vaccination rates in long-term care rose to 90% when employers required vaccination.
The report’s findings were limited by several factors, including the use of a volunteer sample, the reliance on self-reports, and the potential differences between Internet survey results and population-based estimates of flu vaccination.
However, “in the absence of vaccination requirements, the findings in this study support the recommendations found in the Guide to Community Preventive Services, which include active promotion of on-site vaccination at no cost or low cost to increase influenza vaccination coverage among HCPs,” the researchers said.
The researchers had no financial conflicts to disclose.
Nearly four out of five health care personnel in the United States received a flu vaccination during the 2016-2017 flu season, but a majority of those working in long-term care settings were not vaccinated, based on data from an Internet survey of more than 2,000 individuals that was conducted by the Centers for Disease Control and Prevention.
A total of 78.6% of the survey’s respondents said they’d been vaccinated during the 2016-2017 season. Vaccination coverage for health care personnel overall has remained in the 77%-79% range in recent years, but that represents an increase from 64% in 2010-2011.
“As in previous seasons, the highest coverage was among HCP whose workplace had vaccination requirements,” noted Carla L. Black, PhD, of the CDC, and colleagues (MMWR Morb Mortal Wkly Rep. 2017 Sep 29;66[38]:1009-15). The researchers reviewed data collected from an Internet panel survey of 2,438 health care personnel between March 28, 2017, and April 19, 2017.
Physicians boasted the highest vaccination coverage in 2016-2017 (96%), followed by pharmacists (94%), nurses (93%), nurse practitioners and physician assistants (92%), other clinical providers (80%), nonclinical health care providers (74%), and aides and assistants (69%).
Flu vaccination rates were highest among HCPs working in a hospital setting (92%); 94% of survey respondents in hospitals reported either having a vaccination requirement at work or being provided at least 1 day of on-site vaccination.
Vaccination rates were lowest among health care personnel in long-term care settings (68%), where only 26% reported a workplace vaccination requirement. However, vaccination rates in long-term care rose to 90% when employers required vaccination.
The report’s findings were limited by several factors, including the use of a volunteer sample, the reliance on self-reports, and the potential differences between Internet survey results and population-based estimates of flu vaccination.
However, “in the absence of vaccination requirements, the findings in this study support the recommendations found in the Guide to Community Preventive Services, which include active promotion of on-site vaccination at no cost or low cost to increase influenza vaccination coverage among HCPs,” the researchers said.
The researchers had no financial conflicts to disclose.
Nearly four out of five health care personnel in the United States received a flu vaccination during the 2016-2017 flu season, but a majority of those working in long-term care settings were not vaccinated, based on data from an Internet survey of more than 2,000 individuals that was conducted by the Centers for Disease Control and Prevention.
A total of 78.6% of the survey’s respondents said they’d been vaccinated during the 2016-2017 season. Vaccination coverage for health care personnel overall has remained in the 77%-79% range in recent years, but that represents an increase from 64% in 2010-2011.
“As in previous seasons, the highest coverage was among HCP whose workplace had vaccination requirements,” noted Carla L. Black, PhD, of the CDC, and colleagues (MMWR Morb Mortal Wkly Rep. 2017 Sep 29;66[38]:1009-15). The researchers reviewed data collected from an Internet panel survey of 2,438 health care personnel between March 28, 2017, and April 19, 2017.
Physicians boasted the highest vaccination coverage in 2016-2017 (96%), followed by pharmacists (94%), nurses (93%), nurse practitioners and physician assistants (92%), other clinical providers (80%), nonclinical health care providers (74%), and aides and assistants (69%).
Flu vaccination rates were highest among HCPs working in a hospital setting (92%); 94% of survey respondents in hospitals reported either having a vaccination requirement at work or being provided at least 1 day of on-site vaccination.
Vaccination rates were lowest among health care personnel in long-term care settings (68%), where only 26% reported a workplace vaccination requirement. However, vaccination rates in long-term care rose to 90% when employers required vaccination.
The report’s findings were limited by several factors, including the use of a volunteer sample, the reliance on self-reports, and the potential differences between Internet survey results and population-based estimates of flu vaccination.
However, “in the absence of vaccination requirements, the findings in this study support the recommendations found in the Guide to Community Preventive Services, which include active promotion of on-site vaccination at no cost or low cost to increase influenza vaccination coverage among HCPs,” the researchers said.
The researchers had no financial conflicts to disclose.
FROM MMWR
Key clinical point:
Major finding: Overall flu vaccination coverage among U.S. health care personnel was 78.6% in the 2016-2017 season
Data source: The data come from an Internet survey of 2,438 health care personnel.
Disclosures: The researchers had no financial conflicts to disclose.
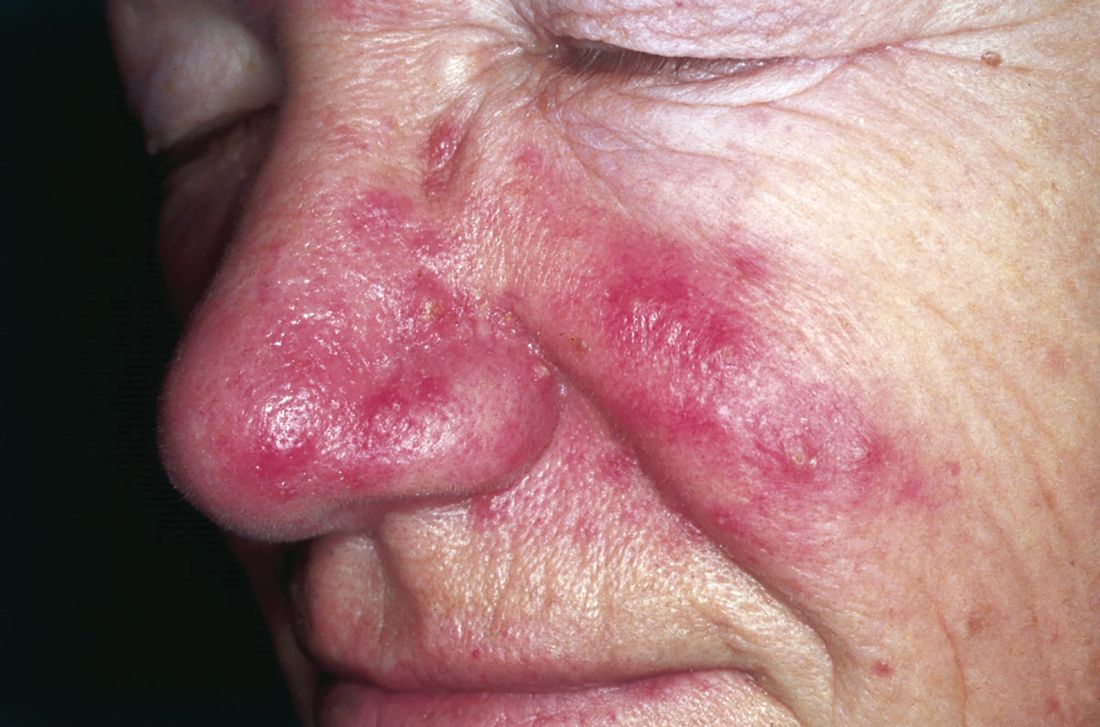
Rosacea patients host the most mites
Infestation with Demodex mites was significantly more common in patients with rosacea compared with healthy controls, based on data from a meta-analysis of 1,513 adults with rosacea. The findings were published in the September issue of the Journal of the American Academy of Dermatology.
The cause of rosacea remains unclear and differs within subgroups, but previous studies have suggested an association between rosacea and the presence of Demodex mites, wrote Yin-Shuo Chang, MD, and Yu-Chen Huang, MD, both of Taipei Medical University, Taiwan (J Am Acad Dermatol. 2017; 77[3]:441-7).
The researchers had no financial conflicts to disclose.
Find the full study online here: http://www.jaad.org/article/S0190-9622(17)30429-2/fulltext.
Infestation with Demodex mites was significantly more common in patients with rosacea compared with healthy controls, based on data from a meta-analysis of 1,513 adults with rosacea. The findings were published in the September issue of the Journal of the American Academy of Dermatology.
The cause of rosacea remains unclear and differs within subgroups, but previous studies have suggested an association between rosacea and the presence of Demodex mites, wrote Yin-Shuo Chang, MD, and Yu-Chen Huang, MD, both of Taipei Medical University, Taiwan (J Am Acad Dermatol. 2017; 77[3]:441-7).

The researchers had no financial conflicts to disclose.
Find the full study online here: http://www.jaad.org/article/S0190-9622(17)30429-2/fulltext.
Infestation with Demodex mites was significantly more common in patients with rosacea compared with healthy controls, based on data from a meta-analysis of 1,513 adults with rosacea. The findings were published in the September issue of the Journal of the American Academy of Dermatology.
The cause of rosacea remains unclear and differs within subgroups, but previous studies have suggested an association between rosacea and the presence of Demodex mites, wrote Yin-Shuo Chang, MD, and Yu-Chen Huang, MD, both of Taipei Medical University, Taiwan (J Am Acad Dermatol. 2017; 77[3]:441-7).

The researchers had no financial conflicts to disclose.
Find the full study online here: http://www.jaad.org/article/S0190-9622(17)30429-2/fulltext.
FROM JAAD
Key clinical point: Demodex mite infestations are significantly associated with rosacea.
Major finding: Rosacea patients were 9 times more likely to experience Demodex mite infestations compared with healthy controls.
Data source: The data come from a meta-analysis of 1,513 adults with rosacea.
Disclosures: The researchers had no financial conflicts to disclose.
New data dissect global burden of scabies
Scabies hits hardest in tropical parts of the world, and children and teens are most affected by the condition, based on data from a global epidemiological assessment.
“,” wrote Chante Karimkhani, MD, of the University of Colorado, Aurora, and her colleagues. To estimate the global impact of scabies, the researchers used the Global Burden of Disease (GBD) Study 2015, which included literature searches between 1980 and 2014 in both English and Spanish.
Overall, the prevalence of scabies worldwide was 204,151,715, and the age-standardized DALYs was 71/100,000 people. The highest DALYs in terms of world regions were reported in East Asia (136/100,000), Southeast Asia (135/100,000) the region described as Oceania (120/100,000) (which included American Samoa, Fiji, Guam, Marshall Islands, Northern Mariana Islands, Papua New Guinea, Samoa, Solomon Islands, and others), tropical Latin America (100/100,000), and South Asia (69/100,000). Scabies caused 0.21% of DALYs from all the conditions that are studied by GBD 2015 globally. The mean percent change in age-standardized DALY from 1990 to 2015 increased 22% in North America, but the age-standardized DALY still remained less than 5/100,000.
In addition, global DALYs from scabies skin infection were highest in children aged 1-4 years (116/100,000 individuals) and adolescents aged 15-19 years (102/100,000). DALYs were not significantly different between men and women across all age groups, Dr. Karimkhani and her associates reported.
“As a worldwide epidemiological assessment, GBD 2015 provides broad and frequently updated measures of scabies burden in terms of skin effects. These global data might help guide research protocols and prioritization efforts and focus scabies treatment and control measures,” the researchers said.
Of note is that these analyses do not include information about complications of scabies such as impetigo, local and systemic bacterial infections, glomerulonephritis, and rheumatic fever.
The study was funded by the Bill and Melinda Gates Foundation. The researchers had no financial conflicts to disclose.
Read the full study here: (Lancet Infect. Dis. 2017. doi: 10.1016/S1473-3099[17]30483-8).
Scabies hits hardest in tropical parts of the world, and children and teens are most affected by the condition, based on data from a global epidemiological assessment.
“,” wrote Chante Karimkhani, MD, of the University of Colorado, Aurora, and her colleagues. To estimate the global impact of scabies, the researchers used the Global Burden of Disease (GBD) Study 2015, which included literature searches between 1980 and 2014 in both English and Spanish.
Overall, the prevalence of scabies worldwide was 204,151,715, and the age-standardized DALYs was 71/100,000 people. The highest DALYs in terms of world regions were reported in East Asia (136/100,000), Southeast Asia (135/100,000) the region described as Oceania (120/100,000) (which included American Samoa, Fiji, Guam, Marshall Islands, Northern Mariana Islands, Papua New Guinea, Samoa, Solomon Islands, and others), tropical Latin America (100/100,000), and South Asia (69/100,000). Scabies caused 0.21% of DALYs from all the conditions that are studied by GBD 2015 globally. The mean percent change in age-standardized DALY from 1990 to 2015 increased 22% in North America, but the age-standardized DALY still remained less than 5/100,000.
In addition, global DALYs from scabies skin infection were highest in children aged 1-4 years (116/100,000 individuals) and adolescents aged 15-19 years (102/100,000). DALYs were not significantly different between men and women across all age groups, Dr. Karimkhani and her associates reported.
“As a worldwide epidemiological assessment, GBD 2015 provides broad and frequently updated measures of scabies burden in terms of skin effects. These global data might help guide research protocols and prioritization efforts and focus scabies treatment and control measures,” the researchers said.
Of note is that these analyses do not include information about complications of scabies such as impetigo, local and systemic bacterial infections, glomerulonephritis, and rheumatic fever.
The study was funded by the Bill and Melinda Gates Foundation. The researchers had no financial conflicts to disclose.
Read the full study here: (Lancet Infect. Dis. 2017. doi: 10.1016/S1473-3099[17]30483-8).
Scabies hits hardest in tropical parts of the world, and children and teens are most affected by the condition, based on data from a global epidemiological assessment.
“,” wrote Chante Karimkhani, MD, of the University of Colorado, Aurora, and her colleagues. To estimate the global impact of scabies, the researchers used the Global Burden of Disease (GBD) Study 2015, which included literature searches between 1980 and 2014 in both English and Spanish.
Overall, the prevalence of scabies worldwide was 204,151,715, and the age-standardized DALYs was 71/100,000 people. The highest DALYs in terms of world regions were reported in East Asia (136/100,000), Southeast Asia (135/100,000) the region described as Oceania (120/100,000) (which included American Samoa, Fiji, Guam, Marshall Islands, Northern Mariana Islands, Papua New Guinea, Samoa, Solomon Islands, and others), tropical Latin America (100/100,000), and South Asia (69/100,000). Scabies caused 0.21% of DALYs from all the conditions that are studied by GBD 2015 globally. The mean percent change in age-standardized DALY from 1990 to 2015 increased 22% in North America, but the age-standardized DALY still remained less than 5/100,000.
In addition, global DALYs from scabies skin infection were highest in children aged 1-4 years (116/100,000 individuals) and adolescents aged 15-19 years (102/100,000). DALYs were not significantly different between men and women across all age groups, Dr. Karimkhani and her associates reported.
“As a worldwide epidemiological assessment, GBD 2015 provides broad and frequently updated measures of scabies burden in terms of skin effects. These global data might help guide research protocols and prioritization efforts and focus scabies treatment and control measures,” the researchers said.
Of note is that these analyses do not include information about complications of scabies such as impetigo, local and systemic bacterial infections, glomerulonephritis, and rheumatic fever.
The study was funded by the Bill and Melinda Gates Foundation. The researchers had no financial conflicts to disclose.
Read the full study here: (Lancet Infect. Dis. 2017. doi: 10.1016/S1473-3099[17]30483-8).
FROM THE LANCET INFECTIOUS DISEASES
Baseline lab data is class specific for biologics to treat psoriasis patients
said April Armstrong, MD, of the University of Southern California, Los Angeles.
Keep class-specific considerations in mind when collecting baseline lab data to help support the success of biologics in treating psoriasis, Dr. Armstrong said at the annual Coastal Dermatology Symposium.
When clinicians consider biologics, they must balance efficacy, safety, convenience, and costs of treatment, Dr. Armstrong said.
She addressed general considerations when selecting biologics for psoriasis and stressed the importance of assessing patients for tuberculosis and reviewing underlying cancer risk. Confirm that a patient has no active infections and consider whether a patient has completed all age-appropriate immunizations. Consider a complete blood count and metabolic panel for the following biologics:
- Ustekinumab: Baseline HIV or pregnancy test, and a TB evaluation at baseline as well as annual monitoring.
- Etanercept, adalimumab, infliximab: Baseline TB evaluation and screening hepatitis panel, liver function tests, and blood count, with option to add pregnancy test or HIV test. A liver function test/hepatitis panel is indicated annually, and TB should be monitored annually. Be cautious about using this class of drugs in patients with heart failure, and verify the absence of demyelinating disease in patients prior to prescribing this class of drugs.
- Guselkumab: Baseline TB evaluation, possible pregnancy or HIV tests, followed by annual TB evaluation.
- Secukinumab, ixekizumab, and brodalumab: Baseline TB evaluation, consider HIV or pregnancy tests, followed by annual TB evaluation. Be cautious about using this class of drugs in patients with ulcerative colitis or Crohn’s disease; assess and counsel for increased risk of suicidality when considering brodalumab.
Beyond the general considerations, several other factors can help maximize success with particular biologics, Dr. Armstrong said at the meeting, which is jointly presented by the University of Louisville and Global Academy for Medical Education.
The number of injections given in the first year, which range from 5 (ustekinumab) to 64 (etanercept) is an important consideration for some patients, Dr. Armstrong noted; the number of injections for the remaining biologics are guselkumab, 8; ixekizumab, 17; brodalumab and adalimumab, both 27, and secukinumab, 32. In addition, the IL-17 inhibitors carry some risk of oral candidiasis and inflammatory bowel disease.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News.
Dr. Armstrong disclosed relationships with multiple companies including AbbVie, Janssen, Novartis, Lilly, Regeneron, Sanofi, Modernizing Medicine, and Valeant.
said April Armstrong, MD, of the University of Southern California, Los Angeles.
Keep class-specific considerations in mind when collecting baseline lab data to help support the success of biologics in treating psoriasis, Dr. Armstrong said at the annual Coastal Dermatology Symposium.
When clinicians consider biologics, they must balance efficacy, safety, convenience, and costs of treatment, Dr. Armstrong said.
She addressed general considerations when selecting biologics for psoriasis and stressed the importance of assessing patients for tuberculosis and reviewing underlying cancer risk. Confirm that a patient has no active infections and consider whether a patient has completed all age-appropriate immunizations. Consider a complete blood count and metabolic panel for the following biologics:
- Ustekinumab: Baseline HIV or pregnancy test, and a TB evaluation at baseline as well as annual monitoring.
- Etanercept, adalimumab, infliximab: Baseline TB evaluation and screening hepatitis panel, liver function tests, and blood count, with option to add pregnancy test or HIV test. A liver function test/hepatitis panel is indicated annually, and TB should be monitored annually. Be cautious about using this class of drugs in patients with heart failure, and verify the absence of demyelinating disease in patients prior to prescribing this class of drugs.
- Guselkumab: Baseline TB evaluation, possible pregnancy or HIV tests, followed by annual TB evaluation.
- Secukinumab, ixekizumab, and brodalumab: Baseline TB evaluation, consider HIV or pregnancy tests, followed by annual TB evaluation. Be cautious about using this class of drugs in patients with ulcerative colitis or Crohn’s disease; assess and counsel for increased risk of suicidality when considering brodalumab.
Beyond the general considerations, several other factors can help maximize success with particular biologics, Dr. Armstrong said at the meeting, which is jointly presented by the University of Louisville and Global Academy for Medical Education.
The number of injections given in the first year, which range from 5 (ustekinumab) to 64 (etanercept) is an important consideration for some patients, Dr. Armstrong noted; the number of injections for the remaining biologics are guselkumab, 8; ixekizumab, 17; brodalumab and adalimumab, both 27, and secukinumab, 32. In addition, the IL-17 inhibitors carry some risk of oral candidiasis and inflammatory bowel disease.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News.
Dr. Armstrong disclosed relationships with multiple companies including AbbVie, Janssen, Novartis, Lilly, Regeneron, Sanofi, Modernizing Medicine, and Valeant.
said April Armstrong, MD, of the University of Southern California, Los Angeles.
Keep class-specific considerations in mind when collecting baseline lab data to help support the success of biologics in treating psoriasis, Dr. Armstrong said at the annual Coastal Dermatology Symposium.
When clinicians consider biologics, they must balance efficacy, safety, convenience, and costs of treatment, Dr. Armstrong said.
She addressed general considerations when selecting biologics for psoriasis and stressed the importance of assessing patients for tuberculosis and reviewing underlying cancer risk. Confirm that a patient has no active infections and consider whether a patient has completed all age-appropriate immunizations. Consider a complete blood count and metabolic panel for the following biologics:
- Ustekinumab: Baseline HIV or pregnancy test, and a TB evaluation at baseline as well as annual monitoring.
- Etanercept, adalimumab, infliximab: Baseline TB evaluation and screening hepatitis panel, liver function tests, and blood count, with option to add pregnancy test or HIV test. A liver function test/hepatitis panel is indicated annually, and TB should be monitored annually. Be cautious about using this class of drugs in patients with heart failure, and verify the absence of demyelinating disease in patients prior to prescribing this class of drugs.
- Guselkumab: Baseline TB evaluation, possible pregnancy or HIV tests, followed by annual TB evaluation.
- Secukinumab, ixekizumab, and brodalumab: Baseline TB evaluation, consider HIV or pregnancy tests, followed by annual TB evaluation. Be cautious about using this class of drugs in patients with ulcerative colitis or Crohn’s disease; assess and counsel for increased risk of suicidality when considering brodalumab.
Beyond the general considerations, several other factors can help maximize success with particular biologics, Dr. Armstrong said at the meeting, which is jointly presented by the University of Louisville and Global Academy for Medical Education.
The number of injections given in the first year, which range from 5 (ustekinumab) to 64 (etanercept) is an important consideration for some patients, Dr. Armstrong noted; the number of injections for the remaining biologics are guselkumab, 8; ixekizumab, 17; brodalumab and adalimumab, both 27, and secukinumab, 32. In addition, the IL-17 inhibitors carry some risk of oral candidiasis and inflammatory bowel disease.
This publication and the Global Academy for Medical Education are owned by Frontline Medical News.
Dr. Armstrong disclosed relationships with multiple companies including AbbVie, Janssen, Novartis, Lilly, Regeneron, Sanofi, Modernizing Medicine, and Valeant.
EXPERT ANALYSIS FROM THE COASTAL DERMATOLOGY SYMPOSIUM