User login
Disorders of diminished motivation: What they are, and how to treat them
Disorders of diminished motivation (DDM)—including apathy, abulia, and akinetic mutism—are characterized by impairment in goal-directed behavior, thought, and emotion.1 These disorders can be observed clinically as a gross underproduction of speech, movement, and emotional response.
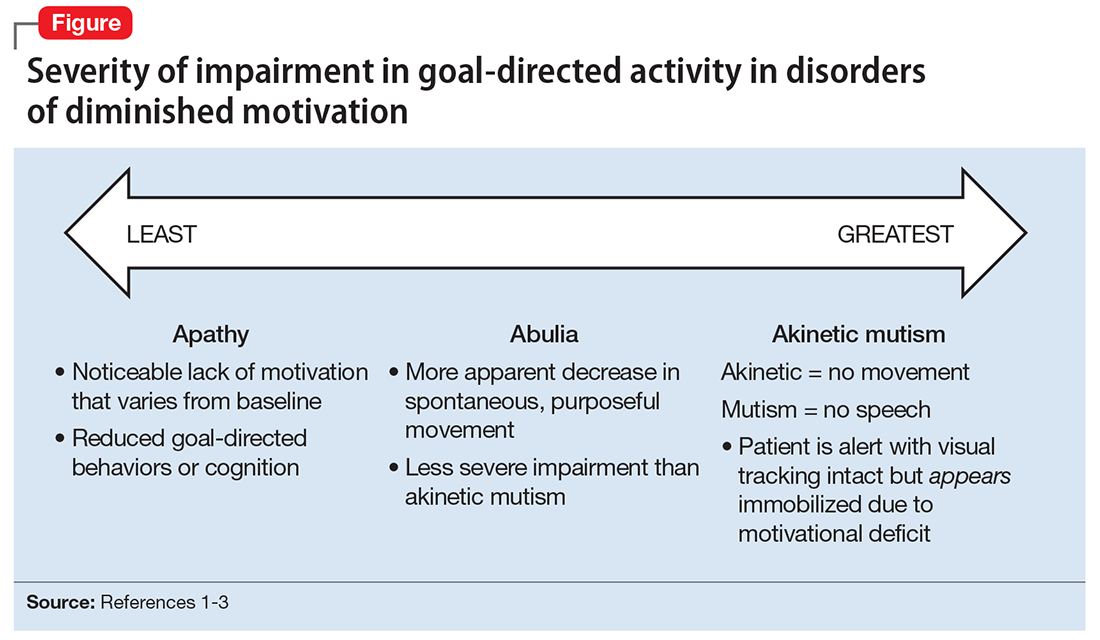
DDM are not classified as disorders within DSM-5, and it remains unclear if they are distinct disorders or symptoms that overlap in other conditions. Some sources support distinct diagnoses, while the traditional position is that DDM are variations along a spectrum, with apathy as the mildest form and akinetic mutism as the most severe form (Figure).1-3 DDM can result from various neurologic, medical, psychiatric, socioeconomic, and drug-induced pathologies, and may represent differing severity of the same underlying pathology.1,4 It is postulated that DDM arise from disruptions in the dopaminergic frontal-subcortical-mesolimbic networks.1,4
We present 2 cases of patients who developed distinct phenotypes within DDM. Despite differences in presentation and symptom severity, both patients showed clinical improvement on methylphenidate (not the only treatment option) as assessed by the Neuropsychiatric Inventory (NPI),5 a scale used to measure dementia-related behavioral symptoms that includes an Apathy/Indifference (A/I) subscale.
CASE 1
Apathy secondary to glioblastoma multiforme
Ms. E, age 59, presents with wound drainage 3 weeks after a repeat right craniotomy for recurrent glioblastoma multiforme (GBM) of the temporal lobe. Her medical history is not believed to have contributed to her current presentation.
On hospital day 2, Ms. E undergoes debridement and reclosure at the craniotomy site. Prior to the procedure, the patient was noted to have anhedonia and flat affect. Her family reports that she seems to get little enjoyment from life and “only slept and ate.” Psychiatry is consulted on hospital day 3 for evaluation and management of a perceived depressed mood.
On initial psychiatric evaluation, Ms. E continues to have a constricted affect with delayed psychomotor processing speed. However, she denies dysphoria or anhedonia. Richmond Agitation-Sedation Scale6 score is 0 (alert and calm) and test of sustained attention (‘Vigilant A’) is intact (ie, based on the Confusion Assessment Method for the Intensive Care Unit [CAM-ICU],7 Ms. E does not have delirium). The NPI A/I frequency score is 15, with a severity score of 3, for a total score of 45, indicating moderate behavioral disturbance on the NPI A/I subsection. A diagnosis of neuropsychiatric apathy due to recurrent GBM or craniotomy is made, although substance-induced mood disorder due to concurrent dexamethasone and opiate use is considered. Methylphenidate, 2.5 mg/d, is started, and Ms. E’s blood pressure remains stable with the initial dose.
Methylphenidate is titrated to 5 mg, twice daily, over a 1-week period. Ms. E’s NPI A/I subscale score improves to 3 (mild behavioral problem), with 3 points for frequency and a multiplier of 1 for mild severity, reflecting an improvement in neuropsychiatric apathy, and she is transferred to a long-term care rehabilitation center.
CASE 2
Akinetic mutism secondary to subarachnoid hemorrhage
Ms. G, age 47, is brought to an outside hospital with syncope and a severe headache radiating to her neck. Upon arrival, she is unconscious and requires intubation. A non-contrast head CT scan shows diffuse subarachnoid hemorrhage, 6 mm right midline shift, and a small left frontal subdural hematoma. A CT angiography of her head and neck reveals a 0.7 cm anterior paraclinoid left internal carotid artery aneurysm with ophthalmic involvement. Evidence of underlying left and right carotid fibromuscular dysplasia is also seen. Ms. G is transferred to our facility for neurosurgical intervention.
Neurosurgery proceeds with aneurysm coiling, followed by left craniotomy with subdural evacuation and ventriculostomy placement. Her postoperative course is complicated by prolonged nasogastric hyperalimentation, mild hypernatremia and hyperglycemia, tracheostomy, and recurrent central fever. She also develops persistent vasospasm, which requires balloon angioplasty of the left middle cerebral artery.
The psychiatry team is consulted on postoperative day 29 to assess for delirium. The CAM-ICU is positive for delirium, with nocturnal accentuation of agitation. Ms. G demonstrates paucity of speech and minimal verbal comprehension. She starts oral ziprasidone, 5 mg/d at bedtime. In addition to her original CNS insult, scopolamine patch, 1.5 mg, to decrease respiratory secretions, and IV metronidazole, 500 mg every 8 hours, for skin-site infection, may have been contributing to her delirium.
Ms. G’s delirium quickly resolves; however, on day 32 she continues to demonstrate behavioral and cognitive slowing; The NPI A/I frequency score is 28, with a severity score of 3, for a total score of 84, indicating severe behavioral disturbance on the NPI A/I subsection. Methylphenidate, 2.5 mg/d, is started and the next day is increased to 5 mg twice a day to treat severe akinetic mutism. Ms. G also is switched from ziprasidone to olanzapine, 2.5 mg/d at night.
By day 37, the tracheostomy is decannulated, and Ms. G demonstrates a full level of alertness, awareness, and attention. Her affect is full range and appropriate; however, she demonstrates residual language deficits, including dysnomia. On day 38, Ms. G is discharged with an NPI A/I subscale score of 5, indicating a mild behavioral problem.
What these cases demonstrate about DDM
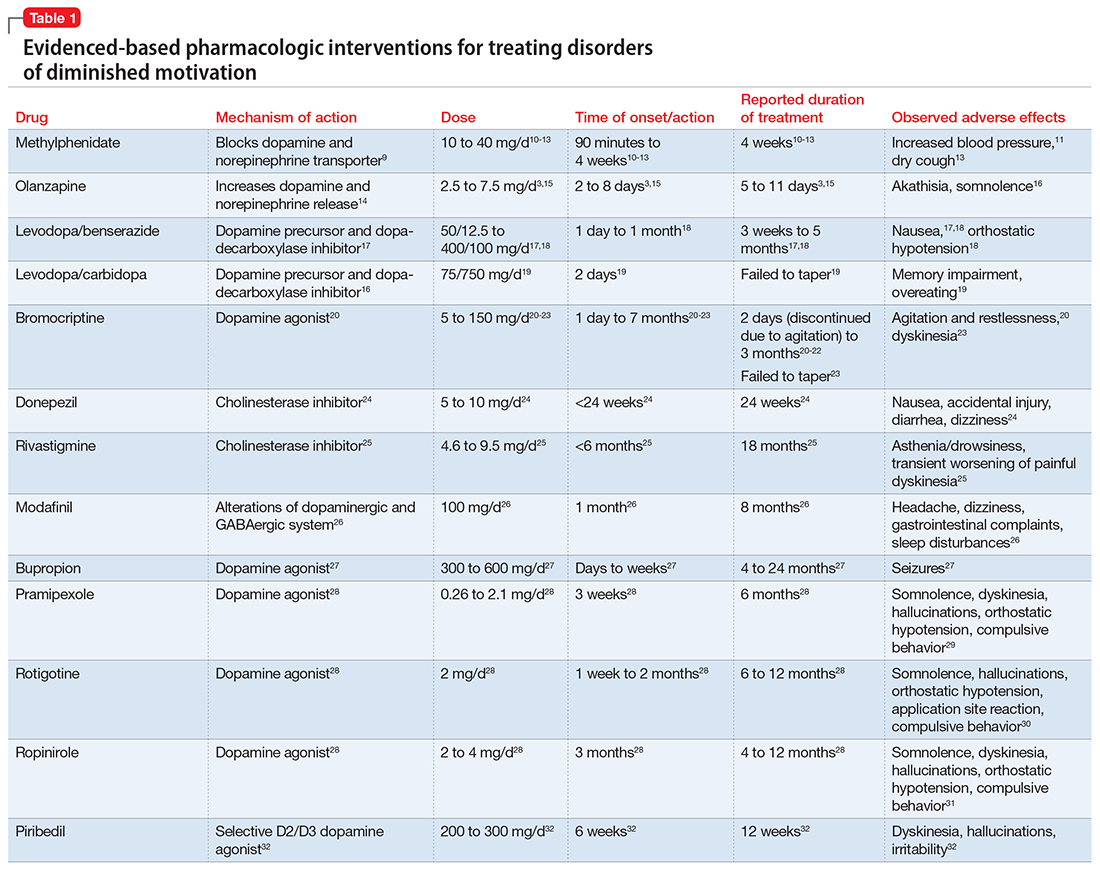
These 2 cases are part of a larger, emerging conversation about the role of dopamine in DDM. Although not fully elucidated, the pathophysiology of abulia, apathy, and akinetic mutism is thought to be related to multiple neurotransmitters—especially dopamine—involved in the cortico-striatal-pallidal-thalamic network.1,8 This position has been supported by reports of clinical improvement in patients with DDM who are given dopaminergic agonists (Table 1).3,9-32
The clinical improvement seen in both of our patients after initiating methylphenidate is consistent with previous reports.10-13 Methylphenidate was selected because of its favorable adverse effect profile and potentially rapid onset of action in DDM.10-13 In cases where oral medication cannot be administered, such as in patients with akinetic mutism, short-term adjunctive IM olanzapine may be helpful, although it is not a first-line treatment.3,15
Interestingly, both of our patients showed improvement with low doses of methylphenidate. Ms. E showed rapid improvement at 2.5 mg/d, but eventually was increased to 10 mg/d. For Ms. G, who demonstrated severe akinetic mutism, rapid improvement was noted after the initial 2.5 mg/d dose; however, because of reports of efficacy of olanzapine in treating akinetic mutism, it is possible that these medications worked synergistically. The proposed mechanism of action of olanzapine in akinetic mutism is through increased dopamine transmission in the medial prefrontal cortex.3,15 Ms. G’s methylphenidate dose was increased to 5 mg/d, which was still “subtherapeutic,” because most reports have used dosages ranging from 10 to 40 mg/d.10-13 Although there were favorable acute results in both patients, their long-term requirements are unknown because of a lack of follow-up. Our findings are also limited by the fact that both patients were recovering from neurosurgical procedures, which could lead to natural improvement in symptoms over time.
Prevalence of DDM in psychiatric disorders
The successful treatment of DDM with dopaminergic drugs is meaningful because of the coexistence of DDM in various neuropsychiatric conditions. In Alzheimer’s disease (AD), disturbances in the dopaminergic system may explain the high comorbidity of apathy, which ranges from 47% in mild AD to 80% in moderate AD.33 In the dopamine-reduced states of cocaine and amphetamine withdrawal, 67% of patients report apathy and lack of motivation.8,34 Additionally, the prevalence of apathy is reported at 27% in Parkinson’s disease, 43% in mild cognitive impairment, 70% in mixed dementia, 94% in a major depressive episode, and 53% in schizophrenia.35 In schizophrenia with predominately negative symptoms, in vivo and postmortem studies have found reduced dopamine receptors.8 Meanwhile, the high rate of akinetic mutism in Creutzfeldt-Jakob disease allows for its use as a reliable diagnostic criteria in this disorder.36
However, the prevalence of DDM is best documented as it relates to stroke and traumatic brain injury (TBI). For instance, after experiencing a stroke, 20% to 25% of patients suffer from apathy.37 Many case reports describe abulia and akinetic mutism after cerebral infarction or hemorrhage, although the incidence of these disorders is unknown.2,38-40 Apathy following TBI is common, especially in younger patients who have sustained a severe injury.41 One study evaluated the prevalence of apathy after TBI among 83 consecutive patients in a neuropsychiatric clinic. Of the 83 patients, 10.84% had apathy without depression, and an equal number were depressed without apathy; another 60% of patients exhibited both apathy and depression. Younger patients (mean age, 29 years) were more likely to be apathetic than older patients, who were more likely to be depressed or depressed and apathetic (mean age, 42 and 38 years, respectively).41 Interestingly, DDM often are associated with cerebral lesions in distinct and distant anatomical locations that are not clearly connected to the neural circuits of motivational pathways. This phenomenon may be explained by the concept of diaschisis, which states that injury to one part of an interconnected neural network can affect other, separate parts of that network.2 If this concept is accurate, it may broaden the impact of DDM, especially as it relates to stroke and TBI.
The differential diagnosis of DDM includes depression and hypokinetic delirium (Table 21,3,42-50). A potential overlapping but confounding condition is stuporous catatonia, with symptoms that include psychomotor slowing such as immobility, staring, and stupor.47 It is important to differentiate these disorders because the treatment for each differs. As previously discussed, there is a clear role for dopamine receptor agonists in the treatment of DDM.

Although major depressive disorder can be treated with medications that increase dopaminergic transmission, selective serotonin reuptake inhibitors (SSRIs) are more commonly used as first-line agents.44 However, an SSRI would theoretically be contraindicated in DDM, because increased serotonin transmission decreases dopamine release from the midbrain, and therefore an SSRI may not only result in a lack of improvement but may worsen DDM.48 Finally, although delirium is treated with atypical or conventional antipsychotics vis-a-vis dopamine type 2 receptor antagonism,45 stuporous catatonia is preferentially treated with gamma-aminobutyric acid-A receptor agonists such as lorazepam.50
What to do when your patient’s presentation suggests DDM
Assessment of DDM should be structured, with input from the patient and the caregiver, and should incorporate the physician’s perspective. A history should be obtained applying recent criteria of apathy. The 3 core domains of apathy—behavior, cognition, and emotion—need to be evaluated. The revised criteria are based on the premise that change in motivation can be measured by examining a patient’s responsiveness to internal or external stimuli. Therefore, each of the 3 domains includes 2 symptoms: (1) self-initiated or “internal” behaviors, cognitions, and emotions (initiation symptom), and (2) the patient’s responsiveness to “external” stimuli (responsiveness symptom).51
One of the main diagnostic dilemmas is how to separate DDM from depression. The differentiation is difficult because of substantial overlap in the manifestation of key symptoms, such as a lack of interest, anergia, psychomotor slowing, and fatigue. Caregivers often mistakenly describe DDM as a depressive state, even though a lack of sadness, desperation, crying, and a depressive mood distinguish DDM from depression. Usually, DDM patients lack negative thoughts, emotional distress, sadness, vegetative symptoms, and somatic concerns, which are frequently observed in mood disorders.51
Several instruments have been developed for assessing neuropsychiatric symptoms. Some were specifically designed to measure apathy, whereas others were designed to provide a broader neuropsychiatric assessment. The NPI is the most widely used multidimensional instrument for assessing neuropsychiatric functioning in patients with neurocognitive disorders (NCDs). It is a valid, reliable instrument that consists of an interview of the patient’s caregiver. It is designed to assess the presence and severity of 10 symptoms, including apathy. The NPI includes both apathy and depression items, which can help clinicians distinguish the 2 conditions. Although beyond the scope of this article, more recent standardized instruments that can assess DDM include the Apathy Inventory, the Dementia Apathy Interview and Rating, and the Structured Clinical Interview for Apathy.52
As previously mentioned, researchers have proposed that DDM are simply a continuum of severity of reduced behavior, and akinetic mutism may be the extreme form. The dilemma is how to formally diagnose states of abulia and akinetic mutism, given the lack of diagnostic criteria and paucity of standardized instruments. Thus, distinguishing between abulia and akinetic mutism (and apathy) is more of a quantitative than qualitative exercise. One could hypothesize that higher scores on a standardized scale to measure apathy (ie, NPI) could imply abulia or akinetic mutism, although to the best of our knowledge, no formal “cut-off scores” exist.53
Treatment of apathy. The duration of pharmacotherapy to treat apathy is unknown and their usage is off-label. Further studies, including double-blind, randomized controlled trials (RCTs), are needed. Nonetheless, the 2 classes of medications that have the most evidence for treating apathy/DDM are psychostimulants and acetylcholinesterase inhibitors (AChEIs).
AChEIs are primarily used for treating cognitive symptoms in NCDs, but recent findings indicate that they have beneficial effects on noncognitive symptoms such as apathy. Of all medications used to treat apathy in NCDs, AChEIs have been used to treat the largest number of patients. Of 26 studies, 24 demonstrated improvement in apathy, with 21 demonstrating statistical significance. These studies ranged in duration from 8 weeks to 1 year, and most were open-label.54
Five studies (3 RCTs and 2 open-label studies) assessed the efficacy of methylphenidate for treating apathy due to AD. All the studies demonstrated at least some benefit in apathy scores after treatment with methylphenidate. These studies ranged from 5 to 12 weeks in duration. Notably, some patients reported adverse effects, including delusions and irritability.54
Although available evidence suggests AChEIs may be the most effective medications for treating apathy in NCDs, methylphenidate has been demonstrated to work faster.55 Thus, in cases where apathy can significantly affect activities of daily living or instrumental activities of daily living, a quicker response may dictate treatment with methylphenidate. It is imperative to note that safety studies and more large-scale double-blind RCTs are needed to further demonstrate the effectiveness and safety of methylphenidate.
Published in 2007, the American Psychiatric Association (APA) guidelines56 state that psychostimulants are a possible treatment option for patients with severe apathy. At the same time, clinicians are reminded that these agents—especially at higher doses—can produce various problematic adverse effects, including tachycardia, hypertension, restlessness, dyskinesia, agitation, sleep disturbances, psychosis, confusion, and decreased appetite. The APA guidelines recommend using low initial doses, with slow and careful titration. For example, methylphenidate should be started at 2.5 to 5 mg once in the morning, with daily doses not to exceed 30 to 40 mg. In our clinical experience, doses >20 mg/d have not been necessary.57
Treatment of akinetic mutism and abulia. In patients with akinetic mutism and possible abulia, for whom oral medication administration is either impossible or contraindicated (ie, due to the potential risk of aspiration pneumonia), atypical antipsychotics, such as IM olanazapine, have produced a therapeutic response in apathetic patients with NCD. However, extensive use of antipsychotics in NCD is not recommended because this class of medications has been associated with serious adverse effects, including an increased risk of death.55
Bottom Line
Apathy, abulia, and akinetic mutism have been categorized as disorders of diminished motivation (DDM). They commonly present after a stroke or traumatic brain injury, and should be differentiated from depression, hypokinetic delirium, and stuporous catatonia. DDM can be successfully treated with dopamine agonists.
Related Resources
- Barnhart WJ, Makela EH, Latocha MJ. SSRI-induced apathy syndrome: a clinical review. J Psychiatr Pract. 2004;10(3):196-199.
- Dell’Osso B, Benatti B, Altamura AC, et al. Prevalence of selective serotonin reuptake inhibitor-related apathy in patients with obsessive compulsive disorder. J Clin Psychopharmacol. 2016;36(6):725-726.
- D’Souza G, Kakoullis A, Hegde N, et al. Recognition and management of abulia in the elderly. Prog Neurol Psychiatry. 2010;14(6):24-28.
Drug Brand Names
Bromocriptine • Parlodel
Bupropion • Wellbutrin XL, Zyban
Carbidopa • Lodosyn
Dexamethasone • DexPak, Ozurde
Donepezil • Aricept
Levodopa/benserazide • Prolopa
Levodopa/carbidopa • Pacopa Rytary Sinemet
Lorazepam • Ativan
Methylphenidate • Concerta, Methylin
Metronidazole • Flagyl, Metrogel
Modafinil • Provigil
Olanzapine • Zyprexa
Pramipexole • Mirapex
Rivastigmine • Exelon
Ropinirole • Requip
Rotigotine • Neurpro
Scopolamine • Transderm Scop
Ziprasidone • Geodon
1. Marin RS, Wilkosz PA. Disorders of diminished motivation. J Head Trauma Rehabil. 2005;20(4):377-388.
2. Ghoshal S, Gokhale S, Rebovich G, et al. The neurology of decreased activity: abulia. Rev Neurol Dis. 2011;8(3-4):e55-e67.
3. Spiegel DR, Chatterjee A. A case of abulia, status/post right middle cerebral artery territory infarct, treated successfully with olanzapine. Clin Neuropharmacol. 2014;37(6):186-189.
4. Marin RS. Differential diagnosis and classification of apathy. Am J Psychiatry. 1990;147(1):22-30.
5. Cummings JL, Mega M, Gray K, et al. The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology. 1994;44(12):2308-2314.
6. Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation-Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166(10):1338-1344.
7. Ely EW, Margolin R, Francis J, et al. Evaluation of delirium in critically ill patients: validation of the Confusion Assessment Method for the intensive care unit (CAM-ICU). Crit Care Med. 2001;29(7):1370-1379.
8. Al-Adawi S, Dawe GS, Al-Hussaini AA. Aboulia: neurobehavioural dysfunction of dopaminergic system? Med Hypotheses. 2000;54(4):523-530.
9. Volkow ND, Fowler JS, Wang G, et al. Mechanism of action of methylphenidate: insights from PET imaging studies. J Atten Disord. 2002;6(suppl 1):S31-S43.
10. Chatterjee A, Fahn S. Methylphenidate treats apathy in Parkinson’s disease. J Neuropsychiatry Clin Neurosci. 2002;14(4):461-462.
11. Keenan S, Mavaddat N, Iddon J, et al. Effects of methylphenidate on cognition and apathy in normal pressure hydrocephalus: a case study and review. Br J Neurosurg. 2005;19(1):46-50.
12. Padala PR, Petty F, Bhatia SC. Methylphenidate may treat apathy independent of depression. Ann Pharmacother. 2005;39(11):1947-1949.
13. Padala PR, Burke WJ, Bhatia SC, et al. Treatment of apathy with methylphenidate. J Neuropsychiatry Clin Neurosci. 2007;19(1):81-83.
14. Li XM, Perry KW, Wong DT, et al. Olanzapine increases in vivo dopamine and norepinephrine release in rat prefrontal cortex, nucleus accumbens and striatum. Psychopharmacology (Berl). 1998;136(2):153-161.
15. Spiegel DR, Casella DP, Callender DM, et al. Treatment of akinetic mutism with intramuscular olanzapine: a case series. J Neuropsychiatry Clin Neurosci. 2008;20(1):93-95.
16. Citrome L. Activating and sedating adverse effects of second-generation antipsychotics in the treatment of schizophrenia and major depressive disorder: absolute risk increase and number needed to harm. J Clin Psychopharmacol. 2017;37(2):138-147.
17. Bakheit AM, Fletcher K, Brennan A. Successful treatment of severe abulia with co-beneldopa. NeuroRehabilitation. 2011;29(4):347-351.
18. Debette S, Kozlowski O, Steinling M, et al. Levodopa and bromocriptine in hypoxic brain injury. J Neurol. 2002;249(12):1678-1682.
19. Combarros O, Infante J, Berciano J. Akinetic mutism from frontal lobe damage responding to levodopa. J Neurol. 2000;247(7):568-569.
20. Echiverri HC, Tatum WO, Merens TA, et al. Akinetic mutism: pharmacologic probe of the dopaminergic mesencephalofrontal activating system. Pediatr Neurol. 1988;4(4):228-230.
21. Psarros T, Zouros A, Coimbra C. Bromocriptine-responsive akinetic mutism following endoscopy for ventricular neurocysticercosis. Case report and review of the literature. J Neurosurg. 2003;99(2):397-401.
22. Naik VD. Abulia following an episode of cardiac arrest [published online July 1, 2015]. BMJ Case Rep. doi: 10.1136/bcr-2015-209357.
23. Kim MS, Rhee JJ, Lee SJ, et al. Akinetic mutism responsive to bromocriptine following subdural hematoma evacuation in a patient with hydrocephalus. Neurol Med Chir (Tokyo). 2007;47(9):419-423.
24. Rockwood K, Black S, Bedard MA; TOPS Study Investigators. Specific symptomatic changes following donepezil treatment of Alzheimer’s disease: a multi-centre, primary care, open-label study. Int J Geriatr Psychiatry. 2007;22(4):312-319.
25. Devos D, Moreau C, Maltête D, et al. Rivastigmine in apathetic but dementia and depression-free patients with Parkinson’s disease: a double-blind, placebo-controlled, randomised clinical trial. J Neurol Neurosurg Psychiatry. 2014;85(6):668-674.
26. Camargos EF, Quintas JL. Apathy syndrome treated successfully with modafinil [published online November 15, 2011]. BMJ Case Rep. doi: 10.1136/bcr.08.2011.4652.
27. Corcoran C, Wong ML, O’Keane V. Bupropion in the management of apathy. J Psychopharmacol. 2004;18(1):133-135.
28. Blundo C, Gerace C. Dopamine agonists can improve pure apathy associated with lesions of the prefrontal-basal ganglia functional system. Neurol Sci. 2015;36(7):1197-1201.
29. Mirapex [package insert]. Ridgefield, CT: Boehringer Ingelheim International GmbH; 2016.
30. Neupro [package insert]. Smyrna, GA: UBC, Inc.; 2012.
31. Requip [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2017.
32. Thobois S, Lhommée E, Klinger H, et al. Parkinsonian apathy responds to dopaminergic stimulation of D2/D3 receptors with piribedil. Brain. 2013;136(pt 5):1568-1577.
33. Mitchell RA, Herrmann N, Lanctôt KL. The role of dopamine in symptoms and treatment of apathy in Alzheimer’s disease. CNS Neurosci Ther. 2011;17(5):411-427.
34. Brower KJ, Maddahian E, Blow FC, et al. A comparison of self-reported symptoms and DSM-III-R criteria for cocaine withdrawal. Am J Drug Alcohol Abuse. 1988;14(3):347-356.
35. Mulin E, Leone E, Dujardin K, et al. Diagnostic criteria for apathy in clinical practice. Int J Geriatr Psychiatry. 2011;26(2):158-165.
36. Otto A, Zerr I, Lantsch M, et al. Akinetic mutism as a classification criterion for the diagnosis of Creutzfeldt-Jakob disease. J Neurol Neurosurg Psychiatry. 1998;64(4):524-528.
37. Jorge RE, Starkstein SE, Robinson RG. Apathy following stroke. Can J Psychiatry. 2010;55(6):350-354.
38. Hastak SM, Gorawara PS, Mishra NK. Abulia: no will, no way. J Assoc Physicians India. 2005;53:814-818.
39. Nagaratnam N, Nagaratnam K, Ng K, et al. Akinetic mutism following stroke. J Clin Neurosci. 2004;11(1):25-30.
40. Freemon FR. Akinetic mutism and bilateral anterior cerebral artery occlusion. J Neurol Neurosurg Psychiatry. 1971;34(6):693-698.
41. Schwarzbold M, Diaz A, Martins ET, et al. Psychiatric disorders and traumatic brain injury. Neuropsychiatr Dis Treat. 2008;4(4):797-816.
42. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
43. Levy ML, Cummings JL, Fairbanks LA, et al. Apathy is not depression. J Neuropsychiatry Clin Neurosci. 1998;10(3):314-319.
44. Snow V, Lascher S, Mottur-Pilson C. Pharmacologic treatment of acute major depression and dysthymia. American College of Physicians-American Society of Internal Medicine. Ann Intern Med. 2000;132(9):738-742.
45. Schwartz AC, Fisher TJ, Greenspan HN, et al. Pharmacologic and nonpharmacologic approaches to the prevention and management of delirium. Int J Psychiatry Med. 2016;51(2):160-170.
46. Kang H, Zhao F, You L, et al. Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 2014;17(2):147-154.
47. Fricchione GL, Beach SR, Huffman J, et al. Life-threatening conditions in psychiatry: catatonia, neuroleptic malignant syndrome, and serotonin syndrome. In: Stern TA, Fava M, Wilens TE, eds. Massachusetts General Hospital comprehensive clinical psychiatry. London, United Kingdom: Elsevier; 2016:608-617.
48. Rogers RD. The roles of dopamine and serotonin in decision making: evidence from pharmacological experiments in humans. Neuropsychopharmacology. 2011;36(1):114-132.
49. Stransky M, Schmidt C, Ganslmeier P, et al. Hypoactive delirium after cardiac surgery as an independent risk factor for prolonged mechanical ventilation. J Cardiothorac Vasc Anesth. 2011;25(6):968-974.
50. Wilcox JA, Reid Duffy P. The syndrome of catatonia. Behav Sci (Basel). 2015;5(4):576-588.
51. Robert PH, Mulin E, Malléa P, et al. REVIEW: apathy diagnosis, assessment, and treatment in Alzheimer’s disease. CNS Neurosci Ther. 2010;16(5):263-271.
52. Cipriani G, Lucetti C, Danti S, et al. Apathy and dementia. Nosology, assessment and management. J Nerv Ment Dis. 2014;202(10):718-724.
53. Starkstein SE, Leentjens AF. The nosological position of apathy in clinical practice. J Neurol Neurosurg Psychiatry. 2008;79(10):1088-1092.54. Berman K, Brodaty H, Withall A, et al. Pharmacologic treatment of apathy in dementia. Am J Geriatr Psychiatry. 2012;20(2):104-122.
55. Theleritis C, Siarkos K, Katirtzoglou E, et al. Pharmacological and nonpharmacological treatment for apathy in Alzheimer disease: a systematic review across modalities. J Geriatr Psychiatry Neurol. 2017;30(1):26-49.
56. APA Work Group on Alzheimer’s Disease and other Dementias; Rabins PV, Blacker D, Rovner BW, et al. American Psychiatric Association practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. Second edition. Am J Psychiatry. 2007;164(suppl 12):5-56.
57. Dolder CR, Davis LN, McKinsey J. Use of psychostimulants in patients with dementia. Ann Pharmacother. 2010;44(10):1624-1632.
Disorders of diminished motivation (DDM)—including apathy, abulia, and akinetic mutism—are characterized by impairment in goal-directed behavior, thought, and emotion.1 These disorders can be observed clinically as a gross underproduction of speech, movement, and emotional response.
DDM are not classified as disorders within DSM-5, and it remains unclear if they are distinct disorders or symptoms that overlap in other conditions. Some sources support distinct diagnoses, while the traditional position is that DDM are variations along a spectrum, with apathy as the mildest form and akinetic mutism as the most severe form (Figure).1-3 DDM can result from various neurologic, medical, psychiatric, socioeconomic, and drug-induced pathologies, and may represent differing severity of the same underlying pathology.1,4 It is postulated that DDM arise from disruptions in the dopaminergic frontal-subcortical-mesolimbic networks.1,4

We present 2 cases of patients who developed distinct phenotypes within DDM. Despite differences in presentation and symptom severity, both patients showed clinical improvement on methylphenidate (not the only treatment option) as assessed by the Neuropsychiatric Inventory (NPI),5 a scale used to measure dementia-related behavioral symptoms that includes an Apathy/Indifference (A/I) subscale.
CASE 1
Apathy secondary to glioblastoma multiforme
Ms. E, age 59, presents with wound drainage 3 weeks after a repeat right craniotomy for recurrent glioblastoma multiforme (GBM) of the temporal lobe. Her medical history is not believed to have contributed to her current presentation.
On hospital day 2, Ms. E undergoes debridement and reclosure at the craniotomy site. Prior to the procedure, the patient was noted to have anhedonia and flat affect. Her family reports that she seems to get little enjoyment from life and “only slept and ate.” Psychiatry is consulted on hospital day 3 for evaluation and management of a perceived depressed mood.
On initial psychiatric evaluation, Ms. E continues to have a constricted affect with delayed psychomotor processing speed. However, she denies dysphoria or anhedonia. Richmond Agitation-Sedation Scale6 score is 0 (alert and calm) and test of sustained attention (‘Vigilant A’) is intact (ie, based on the Confusion Assessment Method for the Intensive Care Unit [CAM-ICU],7 Ms. E does not have delirium). The NPI A/I frequency score is 15, with a severity score of 3, for a total score of 45, indicating moderate behavioral disturbance on the NPI A/I subsection. A diagnosis of neuropsychiatric apathy due to recurrent GBM or craniotomy is made, although substance-induced mood disorder due to concurrent dexamethasone and opiate use is considered. Methylphenidate, 2.5 mg/d, is started, and Ms. E’s blood pressure remains stable with the initial dose.
Methylphenidate is titrated to 5 mg, twice daily, over a 1-week period. Ms. E’s NPI A/I subscale score improves to 3 (mild behavioral problem), with 3 points for frequency and a multiplier of 1 for mild severity, reflecting an improvement in neuropsychiatric apathy, and she is transferred to a long-term care rehabilitation center.
CASE 2
Akinetic mutism secondary to subarachnoid hemorrhage
Ms. G, age 47, is brought to an outside hospital with syncope and a severe headache radiating to her neck. Upon arrival, she is unconscious and requires intubation. A non-contrast head CT scan shows diffuse subarachnoid hemorrhage, 6 mm right midline shift, and a small left frontal subdural hematoma. A CT angiography of her head and neck reveals a 0.7 cm anterior paraclinoid left internal carotid artery aneurysm with ophthalmic involvement. Evidence of underlying left and right carotid fibromuscular dysplasia is also seen. Ms. G is transferred to our facility for neurosurgical intervention.
Neurosurgery proceeds with aneurysm coiling, followed by left craniotomy with subdural evacuation and ventriculostomy placement. Her postoperative course is complicated by prolonged nasogastric hyperalimentation, mild hypernatremia and hyperglycemia, tracheostomy, and recurrent central fever. She also develops persistent vasospasm, which requires balloon angioplasty of the left middle cerebral artery.
The psychiatry team is consulted on postoperative day 29 to assess for delirium. The CAM-ICU is positive for delirium, with nocturnal accentuation of agitation. Ms. G demonstrates paucity of speech and minimal verbal comprehension. She starts oral ziprasidone, 5 mg/d at bedtime. In addition to her original CNS insult, scopolamine patch, 1.5 mg, to decrease respiratory secretions, and IV metronidazole, 500 mg every 8 hours, for skin-site infection, may have been contributing to her delirium.
Ms. G’s delirium quickly resolves; however, on day 32 she continues to demonstrate behavioral and cognitive slowing; The NPI A/I frequency score is 28, with a severity score of 3, for a total score of 84, indicating severe behavioral disturbance on the NPI A/I subsection. Methylphenidate, 2.5 mg/d, is started and the next day is increased to 5 mg twice a day to treat severe akinetic mutism. Ms. G also is switched from ziprasidone to olanzapine, 2.5 mg/d at night.
By day 37, the tracheostomy is decannulated, and Ms. G demonstrates a full level of alertness, awareness, and attention. Her affect is full range and appropriate; however, she demonstrates residual language deficits, including dysnomia. On day 38, Ms. G is discharged with an NPI A/I subscale score of 5, indicating a mild behavioral problem.
What these cases demonstrate about DDM
These 2 cases are part of a larger, emerging conversation about the role of dopamine in DDM. Although not fully elucidated, the pathophysiology of abulia, apathy, and akinetic mutism is thought to be related to multiple neurotransmitters—especially dopamine—involved in the cortico-striatal-pallidal-thalamic network.1,8 This position has been supported by reports of clinical improvement in patients with DDM who are given dopaminergic agonists (Table 1).3,9-32

The clinical improvement seen in both of our patients after initiating methylphenidate is consistent with previous reports.10-13 Methylphenidate was selected because of its favorable adverse effect profile and potentially rapid onset of action in DDM.10-13 In cases where oral medication cannot be administered, such as in patients with akinetic mutism, short-term adjunctive IM olanzapine may be helpful, although it is not a first-line treatment.3,15
Interestingly, both of our patients showed improvement with low doses of methylphenidate. Ms. E showed rapid improvement at 2.5 mg/d, but eventually was increased to 10 mg/d. For Ms. G, who demonstrated severe akinetic mutism, rapid improvement was noted after the initial 2.5 mg/d dose; however, because of reports of efficacy of olanzapine in treating akinetic mutism, it is possible that these medications worked synergistically. The proposed mechanism of action of olanzapine in akinetic mutism is through increased dopamine transmission in the medial prefrontal cortex.3,15 Ms. G’s methylphenidate dose was increased to 5 mg/d, which was still “subtherapeutic,” because most reports have used dosages ranging from 10 to 40 mg/d.10-13 Although there were favorable acute results in both patients, their long-term requirements are unknown because of a lack of follow-up. Our findings are also limited by the fact that both patients were recovering from neurosurgical procedures, which could lead to natural improvement in symptoms over time.
Prevalence of DDM in psychiatric disorders
The successful treatment of DDM with dopaminergic drugs is meaningful because of the coexistence of DDM in various neuropsychiatric conditions. In Alzheimer’s disease (AD), disturbances in the dopaminergic system may explain the high comorbidity of apathy, which ranges from 47% in mild AD to 80% in moderate AD.33 In the dopamine-reduced states of cocaine and amphetamine withdrawal, 67% of patients report apathy and lack of motivation.8,34 Additionally, the prevalence of apathy is reported at 27% in Parkinson’s disease, 43% in mild cognitive impairment, 70% in mixed dementia, 94% in a major depressive episode, and 53% in schizophrenia.35 In schizophrenia with predominately negative symptoms, in vivo and postmortem studies have found reduced dopamine receptors.8 Meanwhile, the high rate of akinetic mutism in Creutzfeldt-Jakob disease allows for its use as a reliable diagnostic criteria in this disorder.36
However, the prevalence of DDM is best documented as it relates to stroke and traumatic brain injury (TBI). For instance, after experiencing a stroke, 20% to 25% of patients suffer from apathy.37 Many case reports describe abulia and akinetic mutism after cerebral infarction or hemorrhage, although the incidence of these disorders is unknown.2,38-40 Apathy following TBI is common, especially in younger patients who have sustained a severe injury.41 One study evaluated the prevalence of apathy after TBI among 83 consecutive patients in a neuropsychiatric clinic. Of the 83 patients, 10.84% had apathy without depression, and an equal number were depressed without apathy; another 60% of patients exhibited both apathy and depression. Younger patients (mean age, 29 years) were more likely to be apathetic than older patients, who were more likely to be depressed or depressed and apathetic (mean age, 42 and 38 years, respectively).41 Interestingly, DDM often are associated with cerebral lesions in distinct and distant anatomical locations that are not clearly connected to the neural circuits of motivational pathways. This phenomenon may be explained by the concept of diaschisis, which states that injury to one part of an interconnected neural network can affect other, separate parts of that network.2 If this concept is accurate, it may broaden the impact of DDM, especially as it relates to stroke and TBI.
The differential diagnosis of DDM includes depression and hypokinetic delirium (Table 21,3,42-50). A potential overlapping but confounding condition is stuporous catatonia, with symptoms that include psychomotor slowing such as immobility, staring, and stupor.47 It is important to differentiate these disorders because the treatment for each differs. As previously discussed, there is a clear role for dopamine receptor agonists in the treatment of DDM.

Although major depressive disorder can be treated with medications that increase dopaminergic transmission, selective serotonin reuptake inhibitors (SSRIs) are more commonly used as first-line agents.44 However, an SSRI would theoretically be contraindicated in DDM, because increased serotonin transmission decreases dopamine release from the midbrain, and therefore an SSRI may not only result in a lack of improvement but may worsen DDM.48 Finally, although delirium is treated with atypical or conventional antipsychotics vis-a-vis dopamine type 2 receptor antagonism,45 stuporous catatonia is preferentially treated with gamma-aminobutyric acid-A receptor agonists such as lorazepam.50
What to do when your patient’s presentation suggests DDM
Assessment of DDM should be structured, with input from the patient and the caregiver, and should incorporate the physician’s perspective. A history should be obtained applying recent criteria of apathy. The 3 core domains of apathy—behavior, cognition, and emotion—need to be evaluated. The revised criteria are based on the premise that change in motivation can be measured by examining a patient’s responsiveness to internal or external stimuli. Therefore, each of the 3 domains includes 2 symptoms: (1) self-initiated or “internal” behaviors, cognitions, and emotions (initiation symptom), and (2) the patient’s responsiveness to “external” stimuli (responsiveness symptom).51
One of the main diagnostic dilemmas is how to separate DDM from depression. The differentiation is difficult because of substantial overlap in the manifestation of key symptoms, such as a lack of interest, anergia, psychomotor slowing, and fatigue. Caregivers often mistakenly describe DDM as a depressive state, even though a lack of sadness, desperation, crying, and a depressive mood distinguish DDM from depression. Usually, DDM patients lack negative thoughts, emotional distress, sadness, vegetative symptoms, and somatic concerns, which are frequently observed in mood disorders.51
Several instruments have been developed for assessing neuropsychiatric symptoms. Some were specifically designed to measure apathy, whereas others were designed to provide a broader neuropsychiatric assessment. The NPI is the most widely used multidimensional instrument for assessing neuropsychiatric functioning in patients with neurocognitive disorders (NCDs). It is a valid, reliable instrument that consists of an interview of the patient’s caregiver. It is designed to assess the presence and severity of 10 symptoms, including apathy. The NPI includes both apathy and depression items, which can help clinicians distinguish the 2 conditions. Although beyond the scope of this article, more recent standardized instruments that can assess DDM include the Apathy Inventory, the Dementia Apathy Interview and Rating, and the Structured Clinical Interview for Apathy.52
As previously mentioned, researchers have proposed that DDM are simply a continuum of severity of reduced behavior, and akinetic mutism may be the extreme form. The dilemma is how to formally diagnose states of abulia and akinetic mutism, given the lack of diagnostic criteria and paucity of standardized instruments. Thus, distinguishing between abulia and akinetic mutism (and apathy) is more of a quantitative than qualitative exercise. One could hypothesize that higher scores on a standardized scale to measure apathy (ie, NPI) could imply abulia or akinetic mutism, although to the best of our knowledge, no formal “cut-off scores” exist.53
Treatment of apathy. The duration of pharmacotherapy to treat apathy is unknown and their usage is off-label. Further studies, including double-blind, randomized controlled trials (RCTs), are needed. Nonetheless, the 2 classes of medications that have the most evidence for treating apathy/DDM are psychostimulants and acetylcholinesterase inhibitors (AChEIs).
AChEIs are primarily used for treating cognitive symptoms in NCDs, but recent findings indicate that they have beneficial effects on noncognitive symptoms such as apathy. Of all medications used to treat apathy in NCDs, AChEIs have been used to treat the largest number of patients. Of 26 studies, 24 demonstrated improvement in apathy, with 21 demonstrating statistical significance. These studies ranged in duration from 8 weeks to 1 year, and most were open-label.54
Five studies (3 RCTs and 2 open-label studies) assessed the efficacy of methylphenidate for treating apathy due to AD. All the studies demonstrated at least some benefit in apathy scores after treatment with methylphenidate. These studies ranged from 5 to 12 weeks in duration. Notably, some patients reported adverse effects, including delusions and irritability.54
Although available evidence suggests AChEIs may be the most effective medications for treating apathy in NCDs, methylphenidate has been demonstrated to work faster.55 Thus, in cases where apathy can significantly affect activities of daily living or instrumental activities of daily living, a quicker response may dictate treatment with methylphenidate. It is imperative to note that safety studies and more large-scale double-blind RCTs are needed to further demonstrate the effectiveness and safety of methylphenidate.
Published in 2007, the American Psychiatric Association (APA) guidelines56 state that psychostimulants are a possible treatment option for patients with severe apathy. At the same time, clinicians are reminded that these agents—especially at higher doses—can produce various problematic adverse effects, including tachycardia, hypertension, restlessness, dyskinesia, agitation, sleep disturbances, psychosis, confusion, and decreased appetite. The APA guidelines recommend using low initial doses, with slow and careful titration. For example, methylphenidate should be started at 2.5 to 5 mg once in the morning, with daily doses not to exceed 30 to 40 mg. In our clinical experience, doses >20 mg/d have not been necessary.57
Treatment of akinetic mutism and abulia. In patients with akinetic mutism and possible abulia, for whom oral medication administration is either impossible or contraindicated (ie, due to the potential risk of aspiration pneumonia), atypical antipsychotics, such as IM olanazapine, have produced a therapeutic response in apathetic patients with NCD. However, extensive use of antipsychotics in NCD is not recommended because this class of medications has been associated with serious adverse effects, including an increased risk of death.55
Bottom Line
Apathy, abulia, and akinetic mutism have been categorized as disorders of diminished motivation (DDM). They commonly present after a stroke or traumatic brain injury, and should be differentiated from depression, hypokinetic delirium, and stuporous catatonia. DDM can be successfully treated with dopamine agonists.
Related Resources
- Barnhart WJ, Makela EH, Latocha MJ. SSRI-induced apathy syndrome: a clinical review. J Psychiatr Pract. 2004;10(3):196-199.
- Dell’Osso B, Benatti B, Altamura AC, et al. Prevalence of selective serotonin reuptake inhibitor-related apathy in patients with obsessive compulsive disorder. J Clin Psychopharmacol. 2016;36(6):725-726.
- D’Souza G, Kakoullis A, Hegde N, et al. Recognition and management of abulia in the elderly. Prog Neurol Psychiatry. 2010;14(6):24-28.
Drug Brand Names
Bromocriptine • Parlodel
Bupropion • Wellbutrin XL, Zyban
Carbidopa • Lodosyn
Dexamethasone • DexPak, Ozurde
Donepezil • Aricept
Levodopa/benserazide • Prolopa
Levodopa/carbidopa • Pacopa Rytary Sinemet
Lorazepam • Ativan
Methylphenidate • Concerta, Methylin
Metronidazole • Flagyl, Metrogel
Modafinil • Provigil
Olanzapine • Zyprexa
Pramipexole • Mirapex
Rivastigmine • Exelon
Ropinirole • Requip
Rotigotine • Neurpro
Scopolamine • Transderm Scop
Ziprasidone • Geodon
Disorders of diminished motivation (DDM)—including apathy, abulia, and akinetic mutism—are characterized by impairment in goal-directed behavior, thought, and emotion.1 These disorders can be observed clinically as a gross underproduction of speech, movement, and emotional response.
DDM are not classified as disorders within DSM-5, and it remains unclear if they are distinct disorders or symptoms that overlap in other conditions. Some sources support distinct diagnoses, while the traditional position is that DDM are variations along a spectrum, with apathy as the mildest form and akinetic mutism as the most severe form (Figure).1-3 DDM can result from various neurologic, medical, psychiatric, socioeconomic, and drug-induced pathologies, and may represent differing severity of the same underlying pathology.1,4 It is postulated that DDM arise from disruptions in the dopaminergic frontal-subcortical-mesolimbic networks.1,4

We present 2 cases of patients who developed distinct phenotypes within DDM. Despite differences in presentation and symptom severity, both patients showed clinical improvement on methylphenidate (not the only treatment option) as assessed by the Neuropsychiatric Inventory (NPI),5 a scale used to measure dementia-related behavioral symptoms that includes an Apathy/Indifference (A/I) subscale.
CASE 1
Apathy secondary to glioblastoma multiforme
Ms. E, age 59, presents with wound drainage 3 weeks after a repeat right craniotomy for recurrent glioblastoma multiforme (GBM) of the temporal lobe. Her medical history is not believed to have contributed to her current presentation.
On hospital day 2, Ms. E undergoes debridement and reclosure at the craniotomy site. Prior to the procedure, the patient was noted to have anhedonia and flat affect. Her family reports that she seems to get little enjoyment from life and “only slept and ate.” Psychiatry is consulted on hospital day 3 for evaluation and management of a perceived depressed mood.
On initial psychiatric evaluation, Ms. E continues to have a constricted affect with delayed psychomotor processing speed. However, she denies dysphoria or anhedonia. Richmond Agitation-Sedation Scale6 score is 0 (alert and calm) and test of sustained attention (‘Vigilant A’) is intact (ie, based on the Confusion Assessment Method for the Intensive Care Unit [CAM-ICU],7 Ms. E does not have delirium). The NPI A/I frequency score is 15, with a severity score of 3, for a total score of 45, indicating moderate behavioral disturbance on the NPI A/I subsection. A diagnosis of neuropsychiatric apathy due to recurrent GBM or craniotomy is made, although substance-induced mood disorder due to concurrent dexamethasone and opiate use is considered. Methylphenidate, 2.5 mg/d, is started, and Ms. E’s blood pressure remains stable with the initial dose.
Methylphenidate is titrated to 5 mg, twice daily, over a 1-week period. Ms. E’s NPI A/I subscale score improves to 3 (mild behavioral problem), with 3 points for frequency and a multiplier of 1 for mild severity, reflecting an improvement in neuropsychiatric apathy, and she is transferred to a long-term care rehabilitation center.
CASE 2
Akinetic mutism secondary to subarachnoid hemorrhage
Ms. G, age 47, is brought to an outside hospital with syncope and a severe headache radiating to her neck. Upon arrival, she is unconscious and requires intubation. A non-contrast head CT scan shows diffuse subarachnoid hemorrhage, 6 mm right midline shift, and a small left frontal subdural hematoma. A CT angiography of her head and neck reveals a 0.7 cm anterior paraclinoid left internal carotid artery aneurysm with ophthalmic involvement. Evidence of underlying left and right carotid fibromuscular dysplasia is also seen. Ms. G is transferred to our facility for neurosurgical intervention.
Neurosurgery proceeds with aneurysm coiling, followed by left craniotomy with subdural evacuation and ventriculostomy placement. Her postoperative course is complicated by prolonged nasogastric hyperalimentation, mild hypernatremia and hyperglycemia, tracheostomy, and recurrent central fever. She also develops persistent vasospasm, which requires balloon angioplasty of the left middle cerebral artery.
The psychiatry team is consulted on postoperative day 29 to assess for delirium. The CAM-ICU is positive for delirium, with nocturnal accentuation of agitation. Ms. G demonstrates paucity of speech and minimal verbal comprehension. She starts oral ziprasidone, 5 mg/d at bedtime. In addition to her original CNS insult, scopolamine patch, 1.5 mg, to decrease respiratory secretions, and IV metronidazole, 500 mg every 8 hours, for skin-site infection, may have been contributing to her delirium.
Ms. G’s delirium quickly resolves; however, on day 32 she continues to demonstrate behavioral and cognitive slowing; The NPI A/I frequency score is 28, with a severity score of 3, for a total score of 84, indicating severe behavioral disturbance on the NPI A/I subsection. Methylphenidate, 2.5 mg/d, is started and the next day is increased to 5 mg twice a day to treat severe akinetic mutism. Ms. G also is switched from ziprasidone to olanzapine, 2.5 mg/d at night.
By day 37, the tracheostomy is decannulated, and Ms. G demonstrates a full level of alertness, awareness, and attention. Her affect is full range and appropriate; however, she demonstrates residual language deficits, including dysnomia. On day 38, Ms. G is discharged with an NPI A/I subscale score of 5, indicating a mild behavioral problem.
What these cases demonstrate about DDM
These 2 cases are part of a larger, emerging conversation about the role of dopamine in DDM. Although not fully elucidated, the pathophysiology of abulia, apathy, and akinetic mutism is thought to be related to multiple neurotransmitters—especially dopamine—involved in the cortico-striatal-pallidal-thalamic network.1,8 This position has been supported by reports of clinical improvement in patients with DDM who are given dopaminergic agonists (Table 1).3,9-32

The clinical improvement seen in both of our patients after initiating methylphenidate is consistent with previous reports.10-13 Methylphenidate was selected because of its favorable adverse effect profile and potentially rapid onset of action in DDM.10-13 In cases where oral medication cannot be administered, such as in patients with akinetic mutism, short-term adjunctive IM olanzapine may be helpful, although it is not a first-line treatment.3,15
Interestingly, both of our patients showed improvement with low doses of methylphenidate. Ms. E showed rapid improvement at 2.5 mg/d, but eventually was increased to 10 mg/d. For Ms. G, who demonstrated severe akinetic mutism, rapid improvement was noted after the initial 2.5 mg/d dose; however, because of reports of efficacy of olanzapine in treating akinetic mutism, it is possible that these medications worked synergistically. The proposed mechanism of action of olanzapine in akinetic mutism is through increased dopamine transmission in the medial prefrontal cortex.3,15 Ms. G’s methylphenidate dose was increased to 5 mg/d, which was still “subtherapeutic,” because most reports have used dosages ranging from 10 to 40 mg/d.10-13 Although there were favorable acute results in both patients, their long-term requirements are unknown because of a lack of follow-up. Our findings are also limited by the fact that both patients were recovering from neurosurgical procedures, which could lead to natural improvement in symptoms over time.
Prevalence of DDM in psychiatric disorders
The successful treatment of DDM with dopaminergic drugs is meaningful because of the coexistence of DDM in various neuropsychiatric conditions. In Alzheimer’s disease (AD), disturbances in the dopaminergic system may explain the high comorbidity of apathy, which ranges from 47% in mild AD to 80% in moderate AD.33 In the dopamine-reduced states of cocaine and amphetamine withdrawal, 67% of patients report apathy and lack of motivation.8,34 Additionally, the prevalence of apathy is reported at 27% in Parkinson’s disease, 43% in mild cognitive impairment, 70% in mixed dementia, 94% in a major depressive episode, and 53% in schizophrenia.35 In schizophrenia with predominately negative symptoms, in vivo and postmortem studies have found reduced dopamine receptors.8 Meanwhile, the high rate of akinetic mutism in Creutzfeldt-Jakob disease allows for its use as a reliable diagnostic criteria in this disorder.36
However, the prevalence of DDM is best documented as it relates to stroke and traumatic brain injury (TBI). For instance, after experiencing a stroke, 20% to 25% of patients suffer from apathy.37 Many case reports describe abulia and akinetic mutism after cerebral infarction or hemorrhage, although the incidence of these disorders is unknown.2,38-40 Apathy following TBI is common, especially in younger patients who have sustained a severe injury.41 One study evaluated the prevalence of apathy after TBI among 83 consecutive patients in a neuropsychiatric clinic. Of the 83 patients, 10.84% had apathy without depression, and an equal number were depressed without apathy; another 60% of patients exhibited both apathy and depression. Younger patients (mean age, 29 years) were more likely to be apathetic than older patients, who were more likely to be depressed or depressed and apathetic (mean age, 42 and 38 years, respectively).41 Interestingly, DDM often are associated with cerebral lesions in distinct and distant anatomical locations that are not clearly connected to the neural circuits of motivational pathways. This phenomenon may be explained by the concept of diaschisis, which states that injury to one part of an interconnected neural network can affect other, separate parts of that network.2 If this concept is accurate, it may broaden the impact of DDM, especially as it relates to stroke and TBI.
The differential diagnosis of DDM includes depression and hypokinetic delirium (Table 21,3,42-50). A potential overlapping but confounding condition is stuporous catatonia, with symptoms that include psychomotor slowing such as immobility, staring, and stupor.47 It is important to differentiate these disorders because the treatment for each differs. As previously discussed, there is a clear role for dopamine receptor agonists in the treatment of DDM.

Although major depressive disorder can be treated with medications that increase dopaminergic transmission, selective serotonin reuptake inhibitors (SSRIs) are more commonly used as first-line agents.44 However, an SSRI would theoretically be contraindicated in DDM, because increased serotonin transmission decreases dopamine release from the midbrain, and therefore an SSRI may not only result in a lack of improvement but may worsen DDM.48 Finally, although delirium is treated with atypical or conventional antipsychotics vis-a-vis dopamine type 2 receptor antagonism,45 stuporous catatonia is preferentially treated with gamma-aminobutyric acid-A receptor agonists such as lorazepam.50
What to do when your patient’s presentation suggests DDM
Assessment of DDM should be structured, with input from the patient and the caregiver, and should incorporate the physician’s perspective. A history should be obtained applying recent criteria of apathy. The 3 core domains of apathy—behavior, cognition, and emotion—need to be evaluated. The revised criteria are based on the premise that change in motivation can be measured by examining a patient’s responsiveness to internal or external stimuli. Therefore, each of the 3 domains includes 2 symptoms: (1) self-initiated or “internal” behaviors, cognitions, and emotions (initiation symptom), and (2) the patient’s responsiveness to “external” stimuli (responsiveness symptom).51
One of the main diagnostic dilemmas is how to separate DDM from depression. The differentiation is difficult because of substantial overlap in the manifestation of key symptoms, such as a lack of interest, anergia, psychomotor slowing, and fatigue. Caregivers often mistakenly describe DDM as a depressive state, even though a lack of sadness, desperation, crying, and a depressive mood distinguish DDM from depression. Usually, DDM patients lack negative thoughts, emotional distress, sadness, vegetative symptoms, and somatic concerns, which are frequently observed in mood disorders.51
Several instruments have been developed for assessing neuropsychiatric symptoms. Some were specifically designed to measure apathy, whereas others were designed to provide a broader neuropsychiatric assessment. The NPI is the most widely used multidimensional instrument for assessing neuropsychiatric functioning in patients with neurocognitive disorders (NCDs). It is a valid, reliable instrument that consists of an interview of the patient’s caregiver. It is designed to assess the presence and severity of 10 symptoms, including apathy. The NPI includes both apathy and depression items, which can help clinicians distinguish the 2 conditions. Although beyond the scope of this article, more recent standardized instruments that can assess DDM include the Apathy Inventory, the Dementia Apathy Interview and Rating, and the Structured Clinical Interview for Apathy.52
As previously mentioned, researchers have proposed that DDM are simply a continuum of severity of reduced behavior, and akinetic mutism may be the extreme form. The dilemma is how to formally diagnose states of abulia and akinetic mutism, given the lack of diagnostic criteria and paucity of standardized instruments. Thus, distinguishing between abulia and akinetic mutism (and apathy) is more of a quantitative than qualitative exercise. One could hypothesize that higher scores on a standardized scale to measure apathy (ie, NPI) could imply abulia or akinetic mutism, although to the best of our knowledge, no formal “cut-off scores” exist.53
Treatment of apathy. The duration of pharmacotherapy to treat apathy is unknown and their usage is off-label. Further studies, including double-blind, randomized controlled trials (RCTs), are needed. Nonetheless, the 2 classes of medications that have the most evidence for treating apathy/DDM are psychostimulants and acetylcholinesterase inhibitors (AChEIs).
AChEIs are primarily used for treating cognitive symptoms in NCDs, but recent findings indicate that they have beneficial effects on noncognitive symptoms such as apathy. Of all medications used to treat apathy in NCDs, AChEIs have been used to treat the largest number of patients. Of 26 studies, 24 demonstrated improvement in apathy, with 21 demonstrating statistical significance. These studies ranged in duration from 8 weeks to 1 year, and most were open-label.54
Five studies (3 RCTs and 2 open-label studies) assessed the efficacy of methylphenidate for treating apathy due to AD. All the studies demonstrated at least some benefit in apathy scores after treatment with methylphenidate. These studies ranged from 5 to 12 weeks in duration. Notably, some patients reported adverse effects, including delusions and irritability.54
Although available evidence suggests AChEIs may be the most effective medications for treating apathy in NCDs, methylphenidate has been demonstrated to work faster.55 Thus, in cases where apathy can significantly affect activities of daily living or instrumental activities of daily living, a quicker response may dictate treatment with methylphenidate. It is imperative to note that safety studies and more large-scale double-blind RCTs are needed to further demonstrate the effectiveness and safety of methylphenidate.
Published in 2007, the American Psychiatric Association (APA) guidelines56 state that psychostimulants are a possible treatment option for patients with severe apathy. At the same time, clinicians are reminded that these agents—especially at higher doses—can produce various problematic adverse effects, including tachycardia, hypertension, restlessness, dyskinesia, agitation, sleep disturbances, psychosis, confusion, and decreased appetite. The APA guidelines recommend using low initial doses, with slow and careful titration. For example, methylphenidate should be started at 2.5 to 5 mg once in the morning, with daily doses not to exceed 30 to 40 mg. In our clinical experience, doses >20 mg/d have not been necessary.57
Treatment of akinetic mutism and abulia. In patients with akinetic mutism and possible abulia, for whom oral medication administration is either impossible or contraindicated (ie, due to the potential risk of aspiration pneumonia), atypical antipsychotics, such as IM olanazapine, have produced a therapeutic response in apathetic patients with NCD. However, extensive use of antipsychotics in NCD is not recommended because this class of medications has been associated with serious adverse effects, including an increased risk of death.55
Bottom Line
Apathy, abulia, and akinetic mutism have been categorized as disorders of diminished motivation (DDM). They commonly present after a stroke or traumatic brain injury, and should be differentiated from depression, hypokinetic delirium, and stuporous catatonia. DDM can be successfully treated with dopamine agonists.
Related Resources
- Barnhart WJ, Makela EH, Latocha MJ. SSRI-induced apathy syndrome: a clinical review. J Psychiatr Pract. 2004;10(3):196-199.
- Dell’Osso B, Benatti B, Altamura AC, et al. Prevalence of selective serotonin reuptake inhibitor-related apathy in patients with obsessive compulsive disorder. J Clin Psychopharmacol. 2016;36(6):725-726.
- D’Souza G, Kakoullis A, Hegde N, et al. Recognition and management of abulia in the elderly. Prog Neurol Psychiatry. 2010;14(6):24-28.
Drug Brand Names
Bromocriptine • Parlodel
Bupropion • Wellbutrin XL, Zyban
Carbidopa • Lodosyn
Dexamethasone • DexPak, Ozurde
Donepezil • Aricept
Levodopa/benserazide • Prolopa
Levodopa/carbidopa • Pacopa Rytary Sinemet
Lorazepam • Ativan
Methylphenidate • Concerta, Methylin
Metronidazole • Flagyl, Metrogel
Modafinil • Provigil
Olanzapine • Zyprexa
Pramipexole • Mirapex
Rivastigmine • Exelon
Ropinirole • Requip
Rotigotine • Neurpro
Scopolamine • Transderm Scop
Ziprasidone • Geodon
1. Marin RS, Wilkosz PA. Disorders of diminished motivation. J Head Trauma Rehabil. 2005;20(4):377-388.
2. Ghoshal S, Gokhale S, Rebovich G, et al. The neurology of decreased activity: abulia. Rev Neurol Dis. 2011;8(3-4):e55-e67.
3. Spiegel DR, Chatterjee A. A case of abulia, status/post right middle cerebral artery territory infarct, treated successfully with olanzapine. Clin Neuropharmacol. 2014;37(6):186-189.
4. Marin RS. Differential diagnosis and classification of apathy. Am J Psychiatry. 1990;147(1):22-30.
5. Cummings JL, Mega M, Gray K, et al. The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology. 1994;44(12):2308-2314.
6. Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation-Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166(10):1338-1344.
7. Ely EW, Margolin R, Francis J, et al. Evaluation of delirium in critically ill patients: validation of the Confusion Assessment Method for the intensive care unit (CAM-ICU). Crit Care Med. 2001;29(7):1370-1379.
8. Al-Adawi S, Dawe GS, Al-Hussaini AA. Aboulia: neurobehavioural dysfunction of dopaminergic system? Med Hypotheses. 2000;54(4):523-530.
9. Volkow ND, Fowler JS, Wang G, et al. Mechanism of action of methylphenidate: insights from PET imaging studies. J Atten Disord. 2002;6(suppl 1):S31-S43.
10. Chatterjee A, Fahn S. Methylphenidate treats apathy in Parkinson’s disease. J Neuropsychiatry Clin Neurosci. 2002;14(4):461-462.
11. Keenan S, Mavaddat N, Iddon J, et al. Effects of methylphenidate on cognition and apathy in normal pressure hydrocephalus: a case study and review. Br J Neurosurg. 2005;19(1):46-50.
12. Padala PR, Petty F, Bhatia SC. Methylphenidate may treat apathy independent of depression. Ann Pharmacother. 2005;39(11):1947-1949.
13. Padala PR, Burke WJ, Bhatia SC, et al. Treatment of apathy with methylphenidate. J Neuropsychiatry Clin Neurosci. 2007;19(1):81-83.
14. Li XM, Perry KW, Wong DT, et al. Olanzapine increases in vivo dopamine and norepinephrine release in rat prefrontal cortex, nucleus accumbens and striatum. Psychopharmacology (Berl). 1998;136(2):153-161.
15. Spiegel DR, Casella DP, Callender DM, et al. Treatment of akinetic mutism with intramuscular olanzapine: a case series. J Neuropsychiatry Clin Neurosci. 2008;20(1):93-95.
16. Citrome L. Activating and sedating adverse effects of second-generation antipsychotics in the treatment of schizophrenia and major depressive disorder: absolute risk increase and number needed to harm. J Clin Psychopharmacol. 2017;37(2):138-147.
17. Bakheit AM, Fletcher K, Brennan A. Successful treatment of severe abulia with co-beneldopa. NeuroRehabilitation. 2011;29(4):347-351.
18. Debette S, Kozlowski O, Steinling M, et al. Levodopa and bromocriptine in hypoxic brain injury. J Neurol. 2002;249(12):1678-1682.
19. Combarros O, Infante J, Berciano J. Akinetic mutism from frontal lobe damage responding to levodopa. J Neurol. 2000;247(7):568-569.
20. Echiverri HC, Tatum WO, Merens TA, et al. Akinetic mutism: pharmacologic probe of the dopaminergic mesencephalofrontal activating system. Pediatr Neurol. 1988;4(4):228-230.
21. Psarros T, Zouros A, Coimbra C. Bromocriptine-responsive akinetic mutism following endoscopy for ventricular neurocysticercosis. Case report and review of the literature. J Neurosurg. 2003;99(2):397-401.
22. Naik VD. Abulia following an episode of cardiac arrest [published online July 1, 2015]. BMJ Case Rep. doi: 10.1136/bcr-2015-209357.
23. Kim MS, Rhee JJ, Lee SJ, et al. Akinetic mutism responsive to bromocriptine following subdural hematoma evacuation in a patient with hydrocephalus. Neurol Med Chir (Tokyo). 2007;47(9):419-423.
24. Rockwood K, Black S, Bedard MA; TOPS Study Investigators. Specific symptomatic changes following donepezil treatment of Alzheimer’s disease: a multi-centre, primary care, open-label study. Int J Geriatr Psychiatry. 2007;22(4):312-319.
25. Devos D, Moreau C, Maltête D, et al. Rivastigmine in apathetic but dementia and depression-free patients with Parkinson’s disease: a double-blind, placebo-controlled, randomised clinical trial. J Neurol Neurosurg Psychiatry. 2014;85(6):668-674.
26. Camargos EF, Quintas JL. Apathy syndrome treated successfully with modafinil [published online November 15, 2011]. BMJ Case Rep. doi: 10.1136/bcr.08.2011.4652.
27. Corcoran C, Wong ML, O’Keane V. Bupropion in the management of apathy. J Psychopharmacol. 2004;18(1):133-135.
28. Blundo C, Gerace C. Dopamine agonists can improve pure apathy associated with lesions of the prefrontal-basal ganglia functional system. Neurol Sci. 2015;36(7):1197-1201.
29. Mirapex [package insert]. Ridgefield, CT: Boehringer Ingelheim International GmbH; 2016.
30. Neupro [package insert]. Smyrna, GA: UBC, Inc.; 2012.
31. Requip [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2017.
32. Thobois S, Lhommée E, Klinger H, et al. Parkinsonian apathy responds to dopaminergic stimulation of D2/D3 receptors with piribedil. Brain. 2013;136(pt 5):1568-1577.
33. Mitchell RA, Herrmann N, Lanctôt KL. The role of dopamine in symptoms and treatment of apathy in Alzheimer’s disease. CNS Neurosci Ther. 2011;17(5):411-427.
34. Brower KJ, Maddahian E, Blow FC, et al. A comparison of self-reported symptoms and DSM-III-R criteria for cocaine withdrawal. Am J Drug Alcohol Abuse. 1988;14(3):347-356.
35. Mulin E, Leone E, Dujardin K, et al. Diagnostic criteria for apathy in clinical practice. Int J Geriatr Psychiatry. 2011;26(2):158-165.
36. Otto A, Zerr I, Lantsch M, et al. Akinetic mutism as a classification criterion for the diagnosis of Creutzfeldt-Jakob disease. J Neurol Neurosurg Psychiatry. 1998;64(4):524-528.
37. Jorge RE, Starkstein SE, Robinson RG. Apathy following stroke. Can J Psychiatry. 2010;55(6):350-354.
38. Hastak SM, Gorawara PS, Mishra NK. Abulia: no will, no way. J Assoc Physicians India. 2005;53:814-818.
39. Nagaratnam N, Nagaratnam K, Ng K, et al. Akinetic mutism following stroke. J Clin Neurosci. 2004;11(1):25-30.
40. Freemon FR. Akinetic mutism and bilateral anterior cerebral artery occlusion. J Neurol Neurosurg Psychiatry. 1971;34(6):693-698.
41. Schwarzbold M, Diaz A, Martins ET, et al. Psychiatric disorders and traumatic brain injury. Neuropsychiatr Dis Treat. 2008;4(4):797-816.
42. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
43. Levy ML, Cummings JL, Fairbanks LA, et al. Apathy is not depression. J Neuropsychiatry Clin Neurosci. 1998;10(3):314-319.
44. Snow V, Lascher S, Mottur-Pilson C. Pharmacologic treatment of acute major depression and dysthymia. American College of Physicians-American Society of Internal Medicine. Ann Intern Med. 2000;132(9):738-742.
45. Schwartz AC, Fisher TJ, Greenspan HN, et al. Pharmacologic and nonpharmacologic approaches to the prevention and management of delirium. Int J Psychiatry Med. 2016;51(2):160-170.
46. Kang H, Zhao F, You L, et al. Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 2014;17(2):147-154.
47. Fricchione GL, Beach SR, Huffman J, et al. Life-threatening conditions in psychiatry: catatonia, neuroleptic malignant syndrome, and serotonin syndrome. In: Stern TA, Fava M, Wilens TE, eds. Massachusetts General Hospital comprehensive clinical psychiatry. London, United Kingdom: Elsevier; 2016:608-617.
48. Rogers RD. The roles of dopamine and serotonin in decision making: evidence from pharmacological experiments in humans. Neuropsychopharmacology. 2011;36(1):114-132.
49. Stransky M, Schmidt C, Ganslmeier P, et al. Hypoactive delirium after cardiac surgery as an independent risk factor for prolonged mechanical ventilation. J Cardiothorac Vasc Anesth. 2011;25(6):968-974.
50. Wilcox JA, Reid Duffy P. The syndrome of catatonia. Behav Sci (Basel). 2015;5(4):576-588.
51. Robert PH, Mulin E, Malléa P, et al. REVIEW: apathy diagnosis, assessment, and treatment in Alzheimer’s disease. CNS Neurosci Ther. 2010;16(5):263-271.
52. Cipriani G, Lucetti C, Danti S, et al. Apathy and dementia. Nosology, assessment and management. J Nerv Ment Dis. 2014;202(10):718-724.
53. Starkstein SE, Leentjens AF. The nosological position of apathy in clinical practice. J Neurol Neurosurg Psychiatry. 2008;79(10):1088-1092.54. Berman K, Brodaty H, Withall A, et al. Pharmacologic treatment of apathy in dementia. Am J Geriatr Psychiatry. 2012;20(2):104-122.
55. Theleritis C, Siarkos K, Katirtzoglou E, et al. Pharmacological and nonpharmacological treatment for apathy in Alzheimer disease: a systematic review across modalities. J Geriatr Psychiatry Neurol. 2017;30(1):26-49.
56. APA Work Group on Alzheimer’s Disease and other Dementias; Rabins PV, Blacker D, Rovner BW, et al. American Psychiatric Association practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. Second edition. Am J Psychiatry. 2007;164(suppl 12):5-56.
57. Dolder CR, Davis LN, McKinsey J. Use of psychostimulants in patients with dementia. Ann Pharmacother. 2010;44(10):1624-1632.
1. Marin RS, Wilkosz PA. Disorders of diminished motivation. J Head Trauma Rehabil. 2005;20(4):377-388.
2. Ghoshal S, Gokhale S, Rebovich G, et al. The neurology of decreased activity: abulia. Rev Neurol Dis. 2011;8(3-4):e55-e67.
3. Spiegel DR, Chatterjee A. A case of abulia, status/post right middle cerebral artery territory infarct, treated successfully with olanzapine. Clin Neuropharmacol. 2014;37(6):186-189.
4. Marin RS. Differential diagnosis and classification of apathy. Am J Psychiatry. 1990;147(1):22-30.
5. Cummings JL, Mega M, Gray K, et al. The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology. 1994;44(12):2308-2314.
6. Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation-Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166(10):1338-1344.
7. Ely EW, Margolin R, Francis J, et al. Evaluation of delirium in critically ill patients: validation of the Confusion Assessment Method for the intensive care unit (CAM-ICU). Crit Care Med. 2001;29(7):1370-1379.
8. Al-Adawi S, Dawe GS, Al-Hussaini AA. Aboulia: neurobehavioural dysfunction of dopaminergic system? Med Hypotheses. 2000;54(4):523-530.
9. Volkow ND, Fowler JS, Wang G, et al. Mechanism of action of methylphenidate: insights from PET imaging studies. J Atten Disord. 2002;6(suppl 1):S31-S43.
10. Chatterjee A, Fahn S. Methylphenidate treats apathy in Parkinson’s disease. J Neuropsychiatry Clin Neurosci. 2002;14(4):461-462.
11. Keenan S, Mavaddat N, Iddon J, et al. Effects of methylphenidate on cognition and apathy in normal pressure hydrocephalus: a case study and review. Br J Neurosurg. 2005;19(1):46-50.
12. Padala PR, Petty F, Bhatia SC. Methylphenidate may treat apathy independent of depression. Ann Pharmacother. 2005;39(11):1947-1949.
13. Padala PR, Burke WJ, Bhatia SC, et al. Treatment of apathy with methylphenidate. J Neuropsychiatry Clin Neurosci. 2007;19(1):81-83.
14. Li XM, Perry KW, Wong DT, et al. Olanzapine increases in vivo dopamine and norepinephrine release in rat prefrontal cortex, nucleus accumbens and striatum. Psychopharmacology (Berl). 1998;136(2):153-161.
15. Spiegel DR, Casella DP, Callender DM, et al. Treatment of akinetic mutism with intramuscular olanzapine: a case series. J Neuropsychiatry Clin Neurosci. 2008;20(1):93-95.
16. Citrome L. Activating and sedating adverse effects of second-generation antipsychotics in the treatment of schizophrenia and major depressive disorder: absolute risk increase and number needed to harm. J Clin Psychopharmacol. 2017;37(2):138-147.
17. Bakheit AM, Fletcher K, Brennan A. Successful treatment of severe abulia with co-beneldopa. NeuroRehabilitation. 2011;29(4):347-351.
18. Debette S, Kozlowski O, Steinling M, et al. Levodopa and bromocriptine in hypoxic brain injury. J Neurol. 2002;249(12):1678-1682.
19. Combarros O, Infante J, Berciano J. Akinetic mutism from frontal lobe damage responding to levodopa. J Neurol. 2000;247(7):568-569.
20. Echiverri HC, Tatum WO, Merens TA, et al. Akinetic mutism: pharmacologic probe of the dopaminergic mesencephalofrontal activating system. Pediatr Neurol. 1988;4(4):228-230.
21. Psarros T, Zouros A, Coimbra C. Bromocriptine-responsive akinetic mutism following endoscopy for ventricular neurocysticercosis. Case report and review of the literature. J Neurosurg. 2003;99(2):397-401.
22. Naik VD. Abulia following an episode of cardiac arrest [published online July 1, 2015]. BMJ Case Rep. doi: 10.1136/bcr-2015-209357.
23. Kim MS, Rhee JJ, Lee SJ, et al. Akinetic mutism responsive to bromocriptine following subdural hematoma evacuation in a patient with hydrocephalus. Neurol Med Chir (Tokyo). 2007;47(9):419-423.
24. Rockwood K, Black S, Bedard MA; TOPS Study Investigators. Specific symptomatic changes following donepezil treatment of Alzheimer’s disease: a multi-centre, primary care, open-label study. Int J Geriatr Psychiatry. 2007;22(4):312-319.
25. Devos D, Moreau C, Maltête D, et al. Rivastigmine in apathetic but dementia and depression-free patients with Parkinson’s disease: a double-blind, placebo-controlled, randomised clinical trial. J Neurol Neurosurg Psychiatry. 2014;85(6):668-674.
26. Camargos EF, Quintas JL. Apathy syndrome treated successfully with modafinil [published online November 15, 2011]. BMJ Case Rep. doi: 10.1136/bcr.08.2011.4652.
27. Corcoran C, Wong ML, O’Keane V. Bupropion in the management of apathy. J Psychopharmacol. 2004;18(1):133-135.
28. Blundo C, Gerace C. Dopamine agonists can improve pure apathy associated with lesions of the prefrontal-basal ganglia functional system. Neurol Sci. 2015;36(7):1197-1201.
29. Mirapex [package insert]. Ridgefield, CT: Boehringer Ingelheim International GmbH; 2016.
30. Neupro [package insert]. Smyrna, GA: UBC, Inc.; 2012.
31. Requip [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2017.
32. Thobois S, Lhommée E, Klinger H, et al. Parkinsonian apathy responds to dopaminergic stimulation of D2/D3 receptors with piribedil. Brain. 2013;136(pt 5):1568-1577.
33. Mitchell RA, Herrmann N, Lanctôt KL. The role of dopamine in symptoms and treatment of apathy in Alzheimer’s disease. CNS Neurosci Ther. 2011;17(5):411-427.
34. Brower KJ, Maddahian E, Blow FC, et al. A comparison of self-reported symptoms and DSM-III-R criteria for cocaine withdrawal. Am J Drug Alcohol Abuse. 1988;14(3):347-356.
35. Mulin E, Leone E, Dujardin K, et al. Diagnostic criteria for apathy in clinical practice. Int J Geriatr Psychiatry. 2011;26(2):158-165.
36. Otto A, Zerr I, Lantsch M, et al. Akinetic mutism as a classification criterion for the diagnosis of Creutzfeldt-Jakob disease. J Neurol Neurosurg Psychiatry. 1998;64(4):524-528.
37. Jorge RE, Starkstein SE, Robinson RG. Apathy following stroke. Can J Psychiatry. 2010;55(6):350-354.
38. Hastak SM, Gorawara PS, Mishra NK. Abulia: no will, no way. J Assoc Physicians India. 2005;53:814-818.
39. Nagaratnam N, Nagaratnam K, Ng K, et al. Akinetic mutism following stroke. J Clin Neurosci. 2004;11(1):25-30.
40. Freemon FR. Akinetic mutism and bilateral anterior cerebral artery occlusion. J Neurol Neurosurg Psychiatry. 1971;34(6):693-698.
41. Schwarzbold M, Diaz A, Martins ET, et al. Psychiatric disorders and traumatic brain injury. Neuropsychiatr Dis Treat. 2008;4(4):797-816.
42. Diagnostic and statistical manual of mental disorders, 5th ed. Washington, DC: American Psychiatric Association; 2013.
43. Levy ML, Cummings JL, Fairbanks LA, et al. Apathy is not depression. J Neuropsychiatry Clin Neurosci. 1998;10(3):314-319.
44. Snow V, Lascher S, Mottur-Pilson C. Pharmacologic treatment of acute major depression and dysthymia. American College of Physicians-American Society of Internal Medicine. Ann Intern Med. 2000;132(9):738-742.
45. Schwartz AC, Fisher TJ, Greenspan HN, et al. Pharmacologic and nonpharmacologic approaches to the prevention and management of delirium. Int J Psychiatry Med. 2016;51(2):160-170.
46. Kang H, Zhao F, You L, et al. Pseudo-dementia: a neuropsychological review. Ann Indian Acad Neurol. 2014;17(2):147-154.
47. Fricchione GL, Beach SR, Huffman J, et al. Life-threatening conditions in psychiatry: catatonia, neuroleptic malignant syndrome, and serotonin syndrome. In: Stern TA, Fava M, Wilens TE, eds. Massachusetts General Hospital comprehensive clinical psychiatry. London, United Kingdom: Elsevier; 2016:608-617.
48. Rogers RD. The roles of dopamine and serotonin in decision making: evidence from pharmacological experiments in humans. Neuropsychopharmacology. 2011;36(1):114-132.
49. Stransky M, Schmidt C, Ganslmeier P, et al. Hypoactive delirium after cardiac surgery as an independent risk factor for prolonged mechanical ventilation. J Cardiothorac Vasc Anesth. 2011;25(6):968-974.
50. Wilcox JA, Reid Duffy P. The syndrome of catatonia. Behav Sci (Basel). 2015;5(4):576-588.
51. Robert PH, Mulin E, Malléa P, et al. REVIEW: apathy diagnosis, assessment, and treatment in Alzheimer’s disease. CNS Neurosci Ther. 2010;16(5):263-271.
52. Cipriani G, Lucetti C, Danti S, et al. Apathy and dementia. Nosology, assessment and management. J Nerv Ment Dis. 2014;202(10):718-724.
53. Starkstein SE, Leentjens AF. The nosological position of apathy in clinical practice. J Neurol Neurosurg Psychiatry. 2008;79(10):1088-1092.54. Berman K, Brodaty H, Withall A, et al. Pharmacologic treatment of apathy in dementia. Am J Geriatr Psychiatry. 2012;20(2):104-122.
55. Theleritis C, Siarkos K, Katirtzoglou E, et al. Pharmacological and nonpharmacological treatment for apathy in Alzheimer disease: a systematic review across modalities. J Geriatr Psychiatry Neurol. 2017;30(1):26-49.
56. APA Work Group on Alzheimer’s Disease and other Dementias; Rabins PV, Blacker D, Rovner BW, et al. American Psychiatric Association practice guideline for the treatment of patients with Alzheimer’s disease and other dementias. Second edition. Am J Psychiatry. 2007;164(suppl 12):5-56.
57. Dolder CR, Davis LN, McKinsey J. Use of psychostimulants in patients with dementia. Ann Pharmacother. 2010;44(10):1624-1632.
Etiologies of DDM
Alcohol withdrawal
Safer use of benzodiazepines for alcohol detoxification
Clinicians often use the symptom-triggered Clinical Institute Withdrawal Assessment for Alcohol Scale, Revised (CIWA-Ar)1 to assess patients’ risk for alcohol withdrawal because it has well-documented reliability, reproducibility, and validity based on comparison with ratings by expert clinicians.2,3 The CIWA-Ar commonly is used to determine when to administer lorazepam to limit or prevent morbidity and mortality in patients who are at risk of or are experiencing alcohol withdrawal. Refined to a list of 10 signs and symptoms, the CIWA-Ar is easy to administer and useful in a variety of clinical settings. The maximum score is 67, and patients with a score >15 are at increased risk for severe alcohol withdrawal.1 For a downloadable copy of the CIWA-Ar, click here.
Despite the benefits of using the CIWA-Ar, qualitative description of certain alcohol withdrawal symptoms is prone to subjective misinterpretation and can result in falsely elevated scores, excessive benzodiazepine administration, and associated sequelae.4 This article describes such a scenario, and examines factors that can contribute to a falsely elevated CIWA-Ar score.
CASE REPORT: Resistant alcohol withdrawal
Mr. J, age 24, is referred to the consultation-liaison service at our teaching hospital for “overall psychiatric assessment and help with alcohol withdrawal.” When brought to the hospital, Mr. J was experiencing diaphoresis and tachycardia. During the interview, he says he “experiences withdrawal symptoms all the time, so I am familiar with the signs.”
Mr. J is cooperative with the interview. Psychomotor agitation or retardation is not noted. His speech is goal-directed, his mood is “calm,” and his affect is within normal range. His thought content is devoid of psychoses or lethal ideations. On Mini-Mental State Examination, Mr. J scores 28 out of 30, which indicates normal cognitive functioning. He reports drinking eight 40-oz bottles of beer daily for the past 3 months. He started drinking alcohol at age 14 and has had only one 1-year period of sobriety. He denies using illicit drugs and his urine drug screen is unremarkable. Mr. J has a history of delirium tremens (DTs), no significant medical history, and was not taking any medications when admitted. His psychiatric history includes generalized anxiety disorder (GAD) and antisocial personality disorder and his family history is significant for alcohol dependence.
Laboratory workup is unremarkable except for a blood alcohol level of 0.23%. Review of systems is significant for mild tremor but no other symptoms of alcohol withdrawal. Physical examination is within normal limits.
Mr. J is started on a symptom-trigger alcohol detoxification protocol using the CIWA-Ar. Based on an elevated CIWA-Ar score of 33, he receives lorazepam IV, 11 mg on his first day of hospitalization and 8 mg on the second day. On the third day, Mr. J is agitated and pulls his IV lines in an attempt to leave. Over the next 24 hours, his blood pressure ranges from 136/90 mm Hg to 169/92 mm Hg and his pulse ranges from 94 to 115 beats per minute. He is given lorazepam, 30 mg, and is transferred to the intensive care unit (ICU).
At this time, Mr. J’s Delirium Rating Scale (DRS) score is 20 (maximum: 32). He remains in the ICU on lorazepam, 25 mg/hr. After 3 days in the ICU, lorazepam is titrated and stopped 2 days later. After lorazepam is stopped, Mr. J’s DRS score is 0, his vital signs are stable, and he no longer demonstrates signs or symptoms of DTs or alcohol withdrawal. He is discharged 1 day later.
Symptom-triggered treatment
Alcohol withdrawal symptoms mainly are caused by the effects of chronic alcohol exposure on brain γ–aminobutyric acid (GABA) and glutamate systems; benzodiazepines are the standard of care (Box).5,6 Mr. J had a history of DTs, which is a risk factor for more severe alcohol withdrawal symptoms and recurrence of DTs.7 Some authors report that fixed dosing intervals are the “gold standard therapy” for alcohol withdrawal, and may be preferable for patients with a history of DTs.8 However, Mr. J was placed on a symptom-triggered protocol, which is standard at our hospital. The decision to implement this protocol was based on concerns of oversedation and possible respiratory suppression. Clinical trials have demonstrated that compared with fixed scheduled therapy for alcohol withdrawal, symptom-triggered protocols result in a reduced need for benzodiazepines (Table).
This treatment strategy requires frequent patient reevaluations—particularly early on—with attention to signs and symptoms of alcohol withdrawal and excessive sedation from medications. Additionally, although most patients with alcohol withdrawal respond to standard treatment that includes benzodiazepines, optimal nutrition, and good supportive care, a subgroup may resist therapy (resistant alcohol withdrawal). Therefore, Mr. J—and others with resistant alcohol withdrawal—may require large doses of benzodiazepines and additional sedatives and undergo complicated hospitalizations.9 Nonetheless, as exemplified by Mr. J, symptom-triggered protocols for alcohol withdrawal can result in potential morbidity and mortality.
Common symptoms of alcohol withdrawal include autonomic hyperactivity, tremor, insomnia, nausea, vomiting, agitation, anxiety, grand mal seizures, and transient visual, tactile, or auditory hallucinations.5 These symptoms result, in part, from the effects of chronic alcohol exposure on brain γ–aminobutyric acid (GABA) and glutamate systems. Alcohol acutely enhances presynaptic GABA release through allosteric modulation at GABAA receptors and inhibits glutamate function through antagonism of N-methyl-d-aspartate (NMDA) receptors. Chronic alcohol exposure elicits compensatory downregulated GABAA and upregulated NMDA expression.
When alcohol intake abruptly stops and its acute effects dissipate, the sudden reduction in GABAergic tone and increase in glutamatergic tone cause alcohol withdrawal symptoms.6 Benzodiazepines, which bind at the benzodiazepine site on the GABAA receptor and, similar to alcohol, acutely enhance GABA and inhibit glutamate signaling, are the standard of care for alcohol withdrawal because they reduce anxiety and the risk of seizures and delirium tremens, which is a severe form of alcohol withdrawal characterized by disturbance in consciousness and cognition and hallucinations.5,6
Table
Benefits of symptom-triggered vs fixed scheduled therapy for alcohol withdrawal
| ST | FS | Benefits of ST | |
|---|---|---|---|
| Efficacy in alcohol withdrawal | Yes | Yes | |
| Flexibility in dosing with fluctuations in CIWA-Ar score | Yes | No | Less medication can be given overall if alcohol withdrawal signs resolve rapidly |
| Lower total benzodiazepine doses | + | – | Smaller chance of side effects such as oversedation, paradoxical agitation, delirium due to benzodiazepine intoxication, or respiratory depression |
| Fewer complications of higher benzodiazepine doses | + | – | Reduced risk of prolonged hospitalization, morbidity from aspiration pneumonia, or need to administer a reversal agent such as flumazenil |
| + = more likely; – = less likely CIWA-Ar: Clinical Institute Withdrawal Assessment of Alcohol Scale, Revised; FS: fixed scheduled; ST: symptom-triggered Bibliography Amato L, Minozzi S, Vecchi S, et al. Benzodiazepines for alcohol withdrawal. Cochrane Database Syst Rev. 2010;(3):CD005063. Cassidy EM, O’Sullivan I, Bradshaw P, et al. Symptom-triggered benzodiazepine therapy for alcohol withdrawal syndrome in the emergency department: a comparison with the standard fixed dose benzodiazepine regimen [published online ahead of print October 19, 2011]. Emerg Med J. doi: 10.1136/emermed-2011-200509. Daeppen JB, Gache P, Landry U, et al. Symptom-triggered vs fixed-schedule doses of benzodiazepine for alcohol withdrawal: a randomized treatment trial. Arch Intern Med. 2002;162(10):1117-1121. DeCarolis DD, Rice KL, Ho L, et al. Symptom-driven lorazepam protocol for treatment of severe alcohol withdrawal delirium in the intensive care unit. Pharmacotherapy. 2007;27(4):510-518. Jaeger TM, Lohr RH, Pankratz VS. Symptom-triggered therapy for alcohol withdrawal syndrome in medical inpatients. Mayo Clin Proc. 2001;76(7):695-701. Weaver MF, Hoffman HJ, Johnson RE, et al. Alcohol withdrawal pharmacotherapy for inpatients with medical comorbidity. J Addict Dis. 2006;25(2):17-24. | |||
Factors influencing CIWA-Ar score
Vital signs monitoring. One limitation of the CIWA-Ar is that vital signs—an objective measurement of alcohol withdrawal— are not used to determine the score. Indeed, Mr. J presented with vital sign dysregulation. However, research suggests that the best predictor of high withdrawal scores includes groups of symptoms rather than individual symptoms.10 In that study, pulse and blood pressure did not correlate with withdrawal severity. Pulse and blood pressure elevations occur in alcohol withdrawal, but other signs and symptoms are more reliable in assessing withdrawal severity. This is clinically important because physicians often prescribe medications for alcohol withdrawal treatment based on pulse and blood pressure measures.1 This needs to be balanced against research that found a systolic blood pressure >150 mm Hg and axillary temperature >38°C can predict development of DTs in patients experiencing alcohol withdrawal.7
Lorazepam-induced disinhibition. Benzodiazepines affect functions associated with processing within the orbital prefrontal cortex,11 including response inhibition and socially acceptable behavior, and impairment in this functioning can result in behavioral disinhibition.12 This effect could account for the apparent paradoxical clinical observation of aggression in benzodiazepine-sedated patients.13 Because agitation is scored on the CIWA-Ar,1,10 falsely elevated scores caused by interpreting benzodiazepine-induced aggression as agitation could result in patients (such as Mr. J) receiving more lorazepam, therefore perpetuating this cycle.
Comorbid anxiety disorders also could falsely accentuate CIWA-Ar scores. For example, the odds of an alcohol dependence diagnosis are 2 to 3 times greater among patients with an anxiety disorder.14 Additionally, the lifetime prevalence of comorbid alcohol dependence for patients with GAD—such as Mr. J— is 30% to 35%.14,15
Alcohol withdrawal can be more severe in patients with alcohol dependence and anxiety disorders because evidence suggests the neurochemical processes underlying both are similar and potentially additive. Studies have shown that these dual diagnosis patients experience more severe symptoms of alcohol withdrawal as assessed by total CIWA-Ar score than those without an anxiety disorder.15 Although such patients may require more aggressive pharmacologic treatment, the dangers of higher benzodiazepine dosages may be even greater.
Benzodiazepine-induced delirium. A recent meta-analysis suggested that benzodiazepines may be associated with an increased risk of delirium.16 Longer-acting benzodiazepines may be associated with increased risk of delirium compared with short-acting agents, and higher doses during a 24-hour period may be associated with increased risk of delirium compared with lower doses. However, wide confidence intervals imply significant uncertainty with these results, and not all patients in the studies reviewed were undergoing alcohol detoxification.16 Benzodiazepines have been reported to accentuate delirium when used to treat DTs.17
We postulate that although Mr. J received lorazepam—a short- to moderate-acting benzodiazepine with a half-life of 12 to 16 hours18—the cumulative dose was high enough to have accentuated—rather than attenuated—delirium.16
Personality disorders. Comorbid alcohol use disorders (AUDs) and personality disorders are well documented. One study found the prevalence of personality disorders in AUDs ranged from 22% to 78%.19 Psychologically, drinking to cope with negative subjective states and emotions (coping motives) and drinking to enhance positive emotions (enhancement motives) may explain the relation between Cluster B personality disorders and AUDs.20
Research on prefrontal functioning in alcoholics and individuals with antisocial personality disorder symptoms has suggested that both groups may be impaired on tasks sensitive to compromised orbitofrontal functioning.21 The orbitofrontal system is essential for maintaining normal inhibitory influences on behavior.22 Benzodiazepines can increase the likelihood of developing disinhibition or impulsivity, which are symptoms of antisocial personality disorder. Because Mr. J had antisocial personality disorder, treating his alcohol withdrawal with a benzodiazepine could have accentuated these symptoms, which were subsequently “treated” with additional lorazepam, therefore worsening the cycle.
Medical comorbidities. The CIWA-Ar relies on autonomic signs and subjective symptoms and was not designed for use in nonverbal patients in the ICU. It is possible that the presence of other acute illnesses may contribute to increased CIWA-Ar scores, but we are unaware of any studies that have evaluated such factors.23
However, tremor, which is scored on the CIWA-Ar, can falsely elevate scores if it is caused by something other than acute alcohol withdrawal. Although essential tremors attenuate with acute alcohol use, chronic alcohol use can result in parkinsonism with a resting tremor, and cerebellar degeneration, which can include an action tremor and cerebellar 3-Hz leg tremor.24 Finally, hepatic encephalopathy—a neuropsychiatric syndrome characterized by disturbances in consciousness, mood, behavior, and cognition—can occur in patients with advanced liver disease, which may be precipitated by alcohol use. The clinical presentation and symptom severity of hepatic encephalopathy varies from minor cognitive impairment to gross disorientation, confusion, and agitation,25 all of which can elevate CIWA-Ar scores.
The role of disinhibition
Disinhibition could serve as the “final common pathway” through which CIWA-Ar scores can be falsely elevated.11 For a Figure that illustrates this, see below. Mr. J presented with several variables that could have elevated his CIWA-Ar score; additional potential factors include other psychiatric diagnoses such as bipolar disorder, opiate withdrawal, dementia, drug-seeking behavior, or malingering.26,27
Treating disinhibition in patients with alcohol withdrawal. Continuing to administer escalating doses of benzodiazepines is counterintuitive for benzodiazepine-induced disinhibition. In a study of alcohol withdrawal in rats, antipsychotics evaluated had some beneficial effects on alcohol withdrawal signs.28 In this study, the comparative effectiveness of atypical antipsychotics was as follows: risperidone = quetiapine > ziprasidone > clozapine > olanzapine.
The American Society of Addiction Medicine’s practice guideline advises against using antipsychotics as the sole agent for DTs because these agents are associated with a longer duration of delirium, higher complication rates, and higher mortality.28 However, antipsychotics have a role as an adjunct to benzodiazepines when benzodiazepines don’t sufficiently control agitation, thought disorder, or perceptual disturbances. Although haloperidol use is well established in this scenario, chlorpromazine is contraindicated because it is epileptogenic, and little information is available on atypical antipsychotics.29 If Mr. J had not responded to tapering lorazepam, evidence would support using haloperidol.
Figure: Unifying concept for pathological BZ administration during alcohol withdrawal syndrome: Disinhibition
AWS: alcohol withdrawal syndrome; BZ: benzodiazepine; CIWA-Ar: Clinical Institute Withdrawal Assessment of Alcohol Scale, Revised; GABA: γ-aminobutyric acid
Source: Reference 11Related Resources
- Myrick H, Anton RF. Treatment of alcohol withdrawal. Alcohol Health & Research World. 1998;22(1):38-43. http://pubs.niaaa.nih.gov/publications/arh22-1/38-43.pdf.
- Amato L, Minozzi S, Davoli M. Efficacy and safety of pharmacological interventions for the treatment of the Alcohol Withdrawal Syndrome. Cochrane Database Syst Rev. 2011;(6):CD008537.
Drug Brand Names
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Flumazenil • Romazicon
- Haloperidol • Haldol
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Ziprasidone • Geodon
Disclosures
Dr. Spiegel is on the speaker’s bureau of Sunovion Pharmaceuticals.
Drs. Kumari and Petri report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgement
The authors thank Amy Herndon for her help in preparing this article.
1. Sullivan JT, Sykora K, Schneiderman J, et al. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict. 1989;84(11):1353-1357.
2. Knott DH, Lerner WD, Davis-Knott T, et al. Decision for alcohol detoxication: a method to standardize patient evaluation. Postgrad Med. 1981;69(5):65-69, 72-75, 78.
3. Wiehl WO, Hayner G, Galloway G. Haight Ashbury Free Clinics’ drug detoxification protocols—Part 4: alcohol. J Psychoactive Drugs. 1994;26(1):57-59.
4. Bostwick JM, Lapid MI. False positives on the clinical institute withdrawal assessment for alcohol-revised: is this scale appropriate for use in the medically ill? Psychosomatics. 2004;45(3):256-261.
5. Diagnostic and statistical manual of mental disorders, 4th ed text rev. Washington DC: American Psychiatric Association; 2000.
6. Schacht JP, Randall PK, Waid LR, et al. Neurocognitive performance, alcohol withdrawal, and effects of a combination of flumazenil and gabapentin in alcohol dependence. Alcohol Clin Exp Res. 2011;35(11):2030-2038.
7. Monte R, Rabuñal R, Casariego E, et al. Risk factors for delirium tremens in patients with alcohol withdrawal syndrome in a hospital setting. Eur J Intern Med. 2009;20(7):690-694.
8. Saitz R, O’Malley SS. Pharmacotherapies for alcohol abuse. Withdrawal and treatment. Med Clin North Am. 1997;81(4):881-907.
9. Hack JB, Hoffmann RS, Nelson LS. Resistant alcohol withdrawal: does an unexpectedly large sedative requirement identify these patients early? J Med Toxicol. 2006;2(2):55-60.
10. Pittman B, Gueorguieva R, Krupitsky E, et al. Multidimensionality of the Alcohol Withdrawal Symptom Checklist: a factor analysis of the Alcohol Withdrawal Symptom Checklist and CIWA-Ar. Alcohol Clin Exp Res. 2007;31(4):612-618.
11. Deakin JB, Aitken MR, Dowson JH, et al. Diazepam produces disinhibitory cognitive effects in male volunteers. Psychopharmacology (Berl). 2004;173(1-2):88-97.
12. Hornberger M, Geng J, Hodges JR. Convergent grey and white matter evidence of orbitofrontal cortex changes related to disinhibition in behavioural variant frontotemporal dementia. Brain. 2011;134(pt 9):2502-2512.
13. Jones KA, Nielsen S, Bruno R, et al. Benzodiazepines - their role in aggression and why GPs should prescribe with caution. Aust Fam Physician. 2011;40(11):862-865.
14. Scott EL, Hulvershorn L. Anxiety disorders with comorbid substance abuse. Psychiatric Times. 2011; 28(9).
15. Faingold CL, Knapp DJ, Chester JA, et al. Integrative neurobiology of the alcohol withdrawal syndrome—from anxiety to seizures. Alcohol Clin Exp Res. 2004;28(2):268-278.
16. Clegg A, Young JB. Which medications to avoid in people at risk of delirium: a systematic review. Age Ageing. 2011;40(1):23-29.
17. Hecksel KA, Bostwick JM, Jaeger TM, et al. Inappropriate use of symptom-triggered therapy for alcohol withdrawal in the general hospital. Mayo Clin Proc. 2008;83(3):274-279.
18. Lader M. Benzodiazepines revisited—will we ever learn? Addiction. 2011;106(12):2086-2109.
19. Mellos E, Liappas I, Paparrigopoulos T. Comorbidity of personality disorders with alcohol abuse. In Vivo. 2010;24(5):761-769.
20. Tragesser SL, Sher KJ, Trull TJ, et al. Personality disorder symptoms, drinking motives, and alcohol use and consequences: cross-sectional and prospective mediation. Exp Clin Psychopharmacol. 2007;15(3):282-292.
21. Oscar-Berman M, Valmas MM, Sawyer KS, et al. Frontal brain dysfunction in alcoholism with and without antisocial personality disorder. Neuropsychiatr Dis Treat. 2009;5:309-326.
22. Dom G, De Wilde B, Hulstijn W, et al. Behavioural aspects of impulsivity in alcoholics with and without a cluster-B personality disorder. Alcohol Alcohol. 2006;41(4):412-420.
23. de Wit M, Jones DG, Sessler CN, et al. Alcohol-use disorders in the critically ill patient. Chest. 2010;138(4):994-1003.
24. Mostile G, Jankovic J. Alcohol in essential tremor and other movement disorders. Mov Disord. 2010;25(14):2274-2284.
25. Crone CC, Gabriel GM, DiMartini A. An overview of psychiatric issues in liver disease for the consultation-liaison psychiatrist. Psychosomatics. 2006;47(3):188-205.
26. Reoux JP, Oreskovich MR. A comparison of two versions of the clinical institute withdrawal assessment for alcohol: the CIWA-Ar and CIWA-AD. Am J Addict. 2006;15(1):85-93.
27. Gray S, Borgundvaag B, Sirvastava A, et al. Feasibility and reliability of the SHOT: a short scale for measuring pretreatment severity of alcohol withdrawal in the emergency department. Acad Emerg Med. 2010;17(10):1048-1054.
28. Uzbay TI. Atypical antipsychotic drugs and ethanol withdrawal syndrome: a review. Alcohol Alcohol. 2012;47(1):33-41.
29. McKeon A, Frye MA, Delanty N. The alcohol withdrawal syndrome. J Neurol Neurosurg Psychiatry. 2008;79(8):854-862.
Clinicians often use the symptom-triggered Clinical Institute Withdrawal Assessment for Alcohol Scale, Revised (CIWA-Ar)1 to assess patients’ risk for alcohol withdrawal because it has well-documented reliability, reproducibility, and validity based on comparison with ratings by expert clinicians.2,3 The CIWA-Ar commonly is used to determine when to administer lorazepam to limit or prevent morbidity and mortality in patients who are at risk of or are experiencing alcohol withdrawal. Refined to a list of 10 signs and symptoms, the CIWA-Ar is easy to administer and useful in a variety of clinical settings. The maximum score is 67, and patients with a score >15 are at increased risk for severe alcohol withdrawal.1 For a downloadable copy of the CIWA-Ar, click here.
Despite the benefits of using the CIWA-Ar, qualitative description of certain alcohol withdrawal symptoms is prone to subjective misinterpretation and can result in falsely elevated scores, excessive benzodiazepine administration, and associated sequelae.4 This article describes such a scenario, and examines factors that can contribute to a falsely elevated CIWA-Ar score.
CASE REPORT: Resistant alcohol withdrawal
Mr. J, age 24, is referred to the consultation-liaison service at our teaching hospital for “overall psychiatric assessment and help with alcohol withdrawal.” When brought to the hospital, Mr. J was experiencing diaphoresis and tachycardia. During the interview, he says he “experiences withdrawal symptoms all the time, so I am familiar with the signs.”
Mr. J is cooperative with the interview. Psychomotor agitation or retardation is not noted. His speech is goal-directed, his mood is “calm,” and his affect is within normal range. His thought content is devoid of psychoses or lethal ideations. On Mini-Mental State Examination, Mr. J scores 28 out of 30, which indicates normal cognitive functioning. He reports drinking eight 40-oz bottles of beer daily for the past 3 months. He started drinking alcohol at age 14 and has had only one 1-year period of sobriety. He denies using illicit drugs and his urine drug screen is unremarkable. Mr. J has a history of delirium tremens (DTs), no significant medical history, and was not taking any medications when admitted. His psychiatric history includes generalized anxiety disorder (GAD) and antisocial personality disorder and his family history is significant for alcohol dependence.
Laboratory workup is unremarkable except for a blood alcohol level of 0.23%. Review of systems is significant for mild tremor but no other symptoms of alcohol withdrawal. Physical examination is within normal limits.
Mr. J is started on a symptom-trigger alcohol detoxification protocol using the CIWA-Ar. Based on an elevated CIWA-Ar score of 33, he receives lorazepam IV, 11 mg on his first day of hospitalization and 8 mg on the second day. On the third day, Mr. J is agitated and pulls his IV lines in an attempt to leave. Over the next 24 hours, his blood pressure ranges from 136/90 mm Hg to 169/92 mm Hg and his pulse ranges from 94 to 115 beats per minute. He is given lorazepam, 30 mg, and is transferred to the intensive care unit (ICU).
At this time, Mr. J’s Delirium Rating Scale (DRS) score is 20 (maximum: 32). He remains in the ICU on lorazepam, 25 mg/hr. After 3 days in the ICU, lorazepam is titrated and stopped 2 days later. After lorazepam is stopped, Mr. J’s DRS score is 0, his vital signs are stable, and he no longer demonstrates signs or symptoms of DTs or alcohol withdrawal. He is discharged 1 day later.
Symptom-triggered treatment
Alcohol withdrawal symptoms mainly are caused by the effects of chronic alcohol exposure on brain γ–aminobutyric acid (GABA) and glutamate systems; benzodiazepines are the standard of care (Box).5,6 Mr. J had a history of DTs, which is a risk factor for more severe alcohol withdrawal symptoms and recurrence of DTs.7 Some authors report that fixed dosing intervals are the “gold standard therapy” for alcohol withdrawal, and may be preferable for patients with a history of DTs.8 However, Mr. J was placed on a symptom-triggered protocol, which is standard at our hospital. The decision to implement this protocol was based on concerns of oversedation and possible respiratory suppression. Clinical trials have demonstrated that compared with fixed scheduled therapy for alcohol withdrawal, symptom-triggered protocols result in a reduced need for benzodiazepines (Table).
This treatment strategy requires frequent patient reevaluations—particularly early on—with attention to signs and symptoms of alcohol withdrawal and excessive sedation from medications. Additionally, although most patients with alcohol withdrawal respond to standard treatment that includes benzodiazepines, optimal nutrition, and good supportive care, a subgroup may resist therapy (resistant alcohol withdrawal). Therefore, Mr. J—and others with resistant alcohol withdrawal—may require large doses of benzodiazepines and additional sedatives and undergo complicated hospitalizations.9 Nonetheless, as exemplified by Mr. J, symptom-triggered protocols for alcohol withdrawal can result in potential morbidity and mortality.
Common symptoms of alcohol withdrawal include autonomic hyperactivity, tremor, insomnia, nausea, vomiting, agitation, anxiety, grand mal seizures, and transient visual, tactile, or auditory hallucinations.5 These symptoms result, in part, from the effects of chronic alcohol exposure on brain γ–aminobutyric acid (GABA) and glutamate systems. Alcohol acutely enhances presynaptic GABA release through allosteric modulation at GABAA receptors and inhibits glutamate function through antagonism of N-methyl-d-aspartate (NMDA) receptors. Chronic alcohol exposure elicits compensatory downregulated GABAA and upregulated NMDA expression.
When alcohol intake abruptly stops and its acute effects dissipate, the sudden reduction in GABAergic tone and increase in glutamatergic tone cause alcohol withdrawal symptoms.6 Benzodiazepines, which bind at the benzodiazepine site on the GABAA receptor and, similar to alcohol, acutely enhance GABA and inhibit glutamate signaling, are the standard of care for alcohol withdrawal because they reduce anxiety and the risk of seizures and delirium tremens, which is a severe form of alcohol withdrawal characterized by disturbance in consciousness and cognition and hallucinations.5,6
Table
Benefits of symptom-triggered vs fixed scheduled therapy for alcohol withdrawal
| ST | FS | Benefits of ST | |
|---|---|---|---|
| Efficacy in alcohol withdrawal | Yes | Yes | |
| Flexibility in dosing with fluctuations in CIWA-Ar score | Yes | No | Less medication can be given overall if alcohol withdrawal signs resolve rapidly |
| Lower total benzodiazepine doses | + | – | Smaller chance of side effects such as oversedation, paradoxical agitation, delirium due to benzodiazepine intoxication, or respiratory depression |
| Fewer complications of higher benzodiazepine doses | + | – | Reduced risk of prolonged hospitalization, morbidity from aspiration pneumonia, or need to administer a reversal agent such as flumazenil |
| + = more likely; – = less likely CIWA-Ar: Clinical Institute Withdrawal Assessment of Alcohol Scale, Revised; FS: fixed scheduled; ST: symptom-triggered Bibliography Amato L, Minozzi S, Vecchi S, et al. Benzodiazepines for alcohol withdrawal. Cochrane Database Syst Rev. 2010;(3):CD005063. Cassidy EM, O’Sullivan I, Bradshaw P, et al. Symptom-triggered benzodiazepine therapy for alcohol withdrawal syndrome in the emergency department: a comparison with the standard fixed dose benzodiazepine regimen [published online ahead of print October 19, 2011]. Emerg Med J. doi: 10.1136/emermed-2011-200509. Daeppen JB, Gache P, Landry U, et al. Symptom-triggered vs fixed-schedule doses of benzodiazepine for alcohol withdrawal: a randomized treatment trial. Arch Intern Med. 2002;162(10):1117-1121. DeCarolis DD, Rice KL, Ho L, et al. Symptom-driven lorazepam protocol for treatment of severe alcohol withdrawal delirium in the intensive care unit. Pharmacotherapy. 2007;27(4):510-518. Jaeger TM, Lohr RH, Pankratz VS. Symptom-triggered therapy for alcohol withdrawal syndrome in medical inpatients. Mayo Clin Proc. 2001;76(7):695-701. Weaver MF, Hoffman HJ, Johnson RE, et al. Alcohol withdrawal pharmacotherapy for inpatients with medical comorbidity. J Addict Dis. 2006;25(2):17-24. | |||
Factors influencing CIWA-Ar score
Vital signs monitoring. One limitation of the CIWA-Ar is that vital signs—an objective measurement of alcohol withdrawal— are not used to determine the score. Indeed, Mr. J presented with vital sign dysregulation. However, research suggests that the best predictor of high withdrawal scores includes groups of symptoms rather than individual symptoms.10 In that study, pulse and blood pressure did not correlate with withdrawal severity. Pulse and blood pressure elevations occur in alcohol withdrawal, but other signs and symptoms are more reliable in assessing withdrawal severity. This is clinically important because physicians often prescribe medications for alcohol withdrawal treatment based on pulse and blood pressure measures.1 This needs to be balanced against research that found a systolic blood pressure >150 mm Hg and axillary temperature >38°C can predict development of DTs in patients experiencing alcohol withdrawal.7
Lorazepam-induced disinhibition. Benzodiazepines affect functions associated with processing within the orbital prefrontal cortex,11 including response inhibition and socially acceptable behavior, and impairment in this functioning can result in behavioral disinhibition.12 This effect could account for the apparent paradoxical clinical observation of aggression in benzodiazepine-sedated patients.13 Because agitation is scored on the CIWA-Ar,1,10 falsely elevated scores caused by interpreting benzodiazepine-induced aggression as agitation could result in patients (such as Mr. J) receiving more lorazepam, therefore perpetuating this cycle.
Comorbid anxiety disorders also could falsely accentuate CIWA-Ar scores. For example, the odds of an alcohol dependence diagnosis are 2 to 3 times greater among patients with an anxiety disorder.14 Additionally, the lifetime prevalence of comorbid alcohol dependence for patients with GAD—such as Mr. J— is 30% to 35%.14,15
Alcohol withdrawal can be more severe in patients with alcohol dependence and anxiety disorders because evidence suggests the neurochemical processes underlying both are similar and potentially additive. Studies have shown that these dual diagnosis patients experience more severe symptoms of alcohol withdrawal as assessed by total CIWA-Ar score than those without an anxiety disorder.15 Although such patients may require more aggressive pharmacologic treatment, the dangers of higher benzodiazepine dosages may be even greater.
Benzodiazepine-induced delirium. A recent meta-analysis suggested that benzodiazepines may be associated with an increased risk of delirium.16 Longer-acting benzodiazepines may be associated with increased risk of delirium compared with short-acting agents, and higher doses during a 24-hour period may be associated with increased risk of delirium compared with lower doses. However, wide confidence intervals imply significant uncertainty with these results, and not all patients in the studies reviewed were undergoing alcohol detoxification.16 Benzodiazepines have been reported to accentuate delirium when used to treat DTs.17
We postulate that although Mr. J received lorazepam—a short- to moderate-acting benzodiazepine with a half-life of 12 to 16 hours18—the cumulative dose was high enough to have accentuated—rather than attenuated—delirium.16
Personality disorders. Comorbid alcohol use disorders (AUDs) and personality disorders are well documented. One study found the prevalence of personality disorders in AUDs ranged from 22% to 78%.19 Psychologically, drinking to cope with negative subjective states and emotions (coping motives) and drinking to enhance positive emotions (enhancement motives) may explain the relation between Cluster B personality disorders and AUDs.20
Research on prefrontal functioning in alcoholics and individuals with antisocial personality disorder symptoms has suggested that both groups may be impaired on tasks sensitive to compromised orbitofrontal functioning.21 The orbitofrontal system is essential for maintaining normal inhibitory influences on behavior.22 Benzodiazepines can increase the likelihood of developing disinhibition or impulsivity, which are symptoms of antisocial personality disorder. Because Mr. J had antisocial personality disorder, treating his alcohol withdrawal with a benzodiazepine could have accentuated these symptoms, which were subsequently “treated” with additional lorazepam, therefore worsening the cycle.
Medical comorbidities. The CIWA-Ar relies on autonomic signs and subjective symptoms and was not designed for use in nonverbal patients in the ICU. It is possible that the presence of other acute illnesses may contribute to increased CIWA-Ar scores, but we are unaware of any studies that have evaluated such factors.23
However, tremor, which is scored on the CIWA-Ar, can falsely elevate scores if it is caused by something other than acute alcohol withdrawal. Although essential tremors attenuate with acute alcohol use, chronic alcohol use can result in parkinsonism with a resting tremor, and cerebellar degeneration, which can include an action tremor and cerebellar 3-Hz leg tremor.24 Finally, hepatic encephalopathy—a neuropsychiatric syndrome characterized by disturbances in consciousness, mood, behavior, and cognition—can occur in patients with advanced liver disease, which may be precipitated by alcohol use. The clinical presentation and symptom severity of hepatic encephalopathy varies from minor cognitive impairment to gross disorientation, confusion, and agitation,25 all of which can elevate CIWA-Ar scores.
The role of disinhibition
Disinhibition could serve as the “final common pathway” through which CIWA-Ar scores can be falsely elevated.11 For a Figure that illustrates this, see below. Mr. J presented with several variables that could have elevated his CIWA-Ar score; additional potential factors include other psychiatric diagnoses such as bipolar disorder, opiate withdrawal, dementia, drug-seeking behavior, or malingering.26,27
Treating disinhibition in patients with alcohol withdrawal. Continuing to administer escalating doses of benzodiazepines is counterintuitive for benzodiazepine-induced disinhibition. In a study of alcohol withdrawal in rats, antipsychotics evaluated had some beneficial effects on alcohol withdrawal signs.28 In this study, the comparative effectiveness of atypical antipsychotics was as follows: risperidone = quetiapine > ziprasidone > clozapine > olanzapine.
The American Society of Addiction Medicine’s practice guideline advises against using antipsychotics as the sole agent for DTs because these agents are associated with a longer duration of delirium, higher complication rates, and higher mortality.28 However, antipsychotics have a role as an adjunct to benzodiazepines when benzodiazepines don’t sufficiently control agitation, thought disorder, or perceptual disturbances. Although haloperidol use is well established in this scenario, chlorpromazine is contraindicated because it is epileptogenic, and little information is available on atypical antipsychotics.29 If Mr. J had not responded to tapering lorazepam, evidence would support using haloperidol.

Figure: Unifying concept for pathological BZ administration during alcohol withdrawal syndrome: Disinhibition
AWS: alcohol withdrawal syndrome; BZ: benzodiazepine; CIWA-Ar: Clinical Institute Withdrawal Assessment of Alcohol Scale, Revised; GABA: γ-aminobutyric acid
Source: Reference 11Related Resources
- Myrick H, Anton RF. Treatment of alcohol withdrawal. Alcohol Health & Research World. 1998;22(1):38-43. http://pubs.niaaa.nih.gov/publications/arh22-1/38-43.pdf.
- Amato L, Minozzi S, Davoli M. Efficacy and safety of pharmacological interventions for the treatment of the Alcohol Withdrawal Syndrome. Cochrane Database Syst Rev. 2011;(6):CD008537.
Drug Brand Names
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Flumazenil • Romazicon
- Haloperidol • Haldol
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Ziprasidone • Geodon
Disclosures
Dr. Spiegel is on the speaker’s bureau of Sunovion Pharmaceuticals.
Drs. Kumari and Petri report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgement
The authors thank Amy Herndon for her help in preparing this article.
Clinicians often use the symptom-triggered Clinical Institute Withdrawal Assessment for Alcohol Scale, Revised (CIWA-Ar)1 to assess patients’ risk for alcohol withdrawal because it has well-documented reliability, reproducibility, and validity based on comparison with ratings by expert clinicians.2,3 The CIWA-Ar commonly is used to determine when to administer lorazepam to limit or prevent morbidity and mortality in patients who are at risk of or are experiencing alcohol withdrawal. Refined to a list of 10 signs and symptoms, the CIWA-Ar is easy to administer and useful in a variety of clinical settings. The maximum score is 67, and patients with a score >15 are at increased risk for severe alcohol withdrawal.1 For a downloadable copy of the CIWA-Ar, click here.
Despite the benefits of using the CIWA-Ar, qualitative description of certain alcohol withdrawal symptoms is prone to subjective misinterpretation and can result in falsely elevated scores, excessive benzodiazepine administration, and associated sequelae.4 This article describes such a scenario, and examines factors that can contribute to a falsely elevated CIWA-Ar score.
CASE REPORT: Resistant alcohol withdrawal
Mr. J, age 24, is referred to the consultation-liaison service at our teaching hospital for “overall psychiatric assessment and help with alcohol withdrawal.” When brought to the hospital, Mr. J was experiencing diaphoresis and tachycardia. During the interview, he says he “experiences withdrawal symptoms all the time, so I am familiar with the signs.”
Mr. J is cooperative with the interview. Psychomotor agitation or retardation is not noted. His speech is goal-directed, his mood is “calm,” and his affect is within normal range. His thought content is devoid of psychoses or lethal ideations. On Mini-Mental State Examination, Mr. J scores 28 out of 30, which indicates normal cognitive functioning. He reports drinking eight 40-oz bottles of beer daily for the past 3 months. He started drinking alcohol at age 14 and has had only one 1-year period of sobriety. He denies using illicit drugs and his urine drug screen is unremarkable. Mr. J has a history of delirium tremens (DTs), no significant medical history, and was not taking any medications when admitted. His psychiatric history includes generalized anxiety disorder (GAD) and antisocial personality disorder and his family history is significant for alcohol dependence.
Laboratory workup is unremarkable except for a blood alcohol level of 0.23%. Review of systems is significant for mild tremor but no other symptoms of alcohol withdrawal. Physical examination is within normal limits.
Mr. J is started on a symptom-trigger alcohol detoxification protocol using the CIWA-Ar. Based on an elevated CIWA-Ar score of 33, he receives lorazepam IV, 11 mg on his first day of hospitalization and 8 mg on the second day. On the third day, Mr. J is agitated and pulls his IV lines in an attempt to leave. Over the next 24 hours, his blood pressure ranges from 136/90 mm Hg to 169/92 mm Hg and his pulse ranges from 94 to 115 beats per minute. He is given lorazepam, 30 mg, and is transferred to the intensive care unit (ICU).
At this time, Mr. J’s Delirium Rating Scale (DRS) score is 20 (maximum: 32). He remains in the ICU on lorazepam, 25 mg/hr. After 3 days in the ICU, lorazepam is titrated and stopped 2 days later. After lorazepam is stopped, Mr. J’s DRS score is 0, his vital signs are stable, and he no longer demonstrates signs or symptoms of DTs or alcohol withdrawal. He is discharged 1 day later.
Symptom-triggered treatment
Alcohol withdrawal symptoms mainly are caused by the effects of chronic alcohol exposure on brain γ–aminobutyric acid (GABA) and glutamate systems; benzodiazepines are the standard of care (Box).5,6 Mr. J had a history of DTs, which is a risk factor for more severe alcohol withdrawal symptoms and recurrence of DTs.7 Some authors report that fixed dosing intervals are the “gold standard therapy” for alcohol withdrawal, and may be preferable for patients with a history of DTs.8 However, Mr. J was placed on a symptom-triggered protocol, which is standard at our hospital. The decision to implement this protocol was based on concerns of oversedation and possible respiratory suppression. Clinical trials have demonstrated that compared with fixed scheduled therapy for alcohol withdrawal, symptom-triggered protocols result in a reduced need for benzodiazepines (Table).
This treatment strategy requires frequent patient reevaluations—particularly early on—with attention to signs and symptoms of alcohol withdrawal and excessive sedation from medications. Additionally, although most patients with alcohol withdrawal respond to standard treatment that includes benzodiazepines, optimal nutrition, and good supportive care, a subgroup may resist therapy (resistant alcohol withdrawal). Therefore, Mr. J—and others with resistant alcohol withdrawal—may require large doses of benzodiazepines and additional sedatives and undergo complicated hospitalizations.9 Nonetheless, as exemplified by Mr. J, symptom-triggered protocols for alcohol withdrawal can result in potential morbidity and mortality.
Common symptoms of alcohol withdrawal include autonomic hyperactivity, tremor, insomnia, nausea, vomiting, agitation, anxiety, grand mal seizures, and transient visual, tactile, or auditory hallucinations.5 These symptoms result, in part, from the effects of chronic alcohol exposure on brain γ–aminobutyric acid (GABA) and glutamate systems. Alcohol acutely enhances presynaptic GABA release through allosteric modulation at GABAA receptors and inhibits glutamate function through antagonism of N-methyl-d-aspartate (NMDA) receptors. Chronic alcohol exposure elicits compensatory downregulated GABAA and upregulated NMDA expression.
When alcohol intake abruptly stops and its acute effects dissipate, the sudden reduction in GABAergic tone and increase in glutamatergic tone cause alcohol withdrawal symptoms.6 Benzodiazepines, which bind at the benzodiazepine site on the GABAA receptor and, similar to alcohol, acutely enhance GABA and inhibit glutamate signaling, are the standard of care for alcohol withdrawal because they reduce anxiety and the risk of seizures and delirium tremens, which is a severe form of alcohol withdrawal characterized by disturbance in consciousness and cognition and hallucinations.5,6
Table
Benefits of symptom-triggered vs fixed scheduled therapy for alcohol withdrawal
| ST | FS | Benefits of ST | |
|---|---|---|---|
| Efficacy in alcohol withdrawal | Yes | Yes | |
| Flexibility in dosing with fluctuations in CIWA-Ar score | Yes | No | Less medication can be given overall if alcohol withdrawal signs resolve rapidly |
| Lower total benzodiazepine doses | + | – | Smaller chance of side effects such as oversedation, paradoxical agitation, delirium due to benzodiazepine intoxication, or respiratory depression |
| Fewer complications of higher benzodiazepine doses | + | – | Reduced risk of prolonged hospitalization, morbidity from aspiration pneumonia, or need to administer a reversal agent such as flumazenil |
| + = more likely; – = less likely CIWA-Ar: Clinical Institute Withdrawal Assessment of Alcohol Scale, Revised; FS: fixed scheduled; ST: symptom-triggered Bibliography Amato L, Minozzi S, Vecchi S, et al. Benzodiazepines for alcohol withdrawal. Cochrane Database Syst Rev. 2010;(3):CD005063. Cassidy EM, O’Sullivan I, Bradshaw P, et al. Symptom-triggered benzodiazepine therapy for alcohol withdrawal syndrome in the emergency department: a comparison with the standard fixed dose benzodiazepine regimen [published online ahead of print October 19, 2011]. Emerg Med J. doi: 10.1136/emermed-2011-200509. Daeppen JB, Gache P, Landry U, et al. Symptom-triggered vs fixed-schedule doses of benzodiazepine for alcohol withdrawal: a randomized treatment trial. Arch Intern Med. 2002;162(10):1117-1121. DeCarolis DD, Rice KL, Ho L, et al. Symptom-driven lorazepam protocol for treatment of severe alcohol withdrawal delirium in the intensive care unit. Pharmacotherapy. 2007;27(4):510-518. Jaeger TM, Lohr RH, Pankratz VS. Symptom-triggered therapy for alcohol withdrawal syndrome in medical inpatients. Mayo Clin Proc. 2001;76(7):695-701. Weaver MF, Hoffman HJ, Johnson RE, et al. Alcohol withdrawal pharmacotherapy for inpatients with medical comorbidity. J Addict Dis. 2006;25(2):17-24. | |||
Factors influencing CIWA-Ar score
Vital signs monitoring. One limitation of the CIWA-Ar is that vital signs—an objective measurement of alcohol withdrawal— are not used to determine the score. Indeed, Mr. J presented with vital sign dysregulation. However, research suggests that the best predictor of high withdrawal scores includes groups of symptoms rather than individual symptoms.10 In that study, pulse and blood pressure did not correlate with withdrawal severity. Pulse and blood pressure elevations occur in alcohol withdrawal, but other signs and symptoms are more reliable in assessing withdrawal severity. This is clinically important because physicians often prescribe medications for alcohol withdrawal treatment based on pulse and blood pressure measures.1 This needs to be balanced against research that found a systolic blood pressure >150 mm Hg and axillary temperature >38°C can predict development of DTs in patients experiencing alcohol withdrawal.7
Lorazepam-induced disinhibition. Benzodiazepines affect functions associated with processing within the orbital prefrontal cortex,11 including response inhibition and socially acceptable behavior, and impairment in this functioning can result in behavioral disinhibition.12 This effect could account for the apparent paradoxical clinical observation of aggression in benzodiazepine-sedated patients.13 Because agitation is scored on the CIWA-Ar,1,10 falsely elevated scores caused by interpreting benzodiazepine-induced aggression as agitation could result in patients (such as Mr. J) receiving more lorazepam, therefore perpetuating this cycle.
Comorbid anxiety disorders also could falsely accentuate CIWA-Ar scores. For example, the odds of an alcohol dependence diagnosis are 2 to 3 times greater among patients with an anxiety disorder.14 Additionally, the lifetime prevalence of comorbid alcohol dependence for patients with GAD—such as Mr. J— is 30% to 35%.14,15
Alcohol withdrawal can be more severe in patients with alcohol dependence and anxiety disorders because evidence suggests the neurochemical processes underlying both are similar and potentially additive. Studies have shown that these dual diagnosis patients experience more severe symptoms of alcohol withdrawal as assessed by total CIWA-Ar score than those without an anxiety disorder.15 Although such patients may require more aggressive pharmacologic treatment, the dangers of higher benzodiazepine dosages may be even greater.
Benzodiazepine-induced delirium. A recent meta-analysis suggested that benzodiazepines may be associated with an increased risk of delirium.16 Longer-acting benzodiazepines may be associated with increased risk of delirium compared with short-acting agents, and higher doses during a 24-hour period may be associated with increased risk of delirium compared with lower doses. However, wide confidence intervals imply significant uncertainty with these results, and not all patients in the studies reviewed were undergoing alcohol detoxification.16 Benzodiazepines have been reported to accentuate delirium when used to treat DTs.17
We postulate that although Mr. J received lorazepam—a short- to moderate-acting benzodiazepine with a half-life of 12 to 16 hours18—the cumulative dose was high enough to have accentuated—rather than attenuated—delirium.16
Personality disorders. Comorbid alcohol use disorders (AUDs) and personality disorders are well documented. One study found the prevalence of personality disorders in AUDs ranged from 22% to 78%.19 Psychologically, drinking to cope with negative subjective states and emotions (coping motives) and drinking to enhance positive emotions (enhancement motives) may explain the relation between Cluster B personality disorders and AUDs.20
Research on prefrontal functioning in alcoholics and individuals with antisocial personality disorder symptoms has suggested that both groups may be impaired on tasks sensitive to compromised orbitofrontal functioning.21 The orbitofrontal system is essential for maintaining normal inhibitory influences on behavior.22 Benzodiazepines can increase the likelihood of developing disinhibition or impulsivity, which are symptoms of antisocial personality disorder. Because Mr. J had antisocial personality disorder, treating his alcohol withdrawal with a benzodiazepine could have accentuated these symptoms, which were subsequently “treated” with additional lorazepam, therefore worsening the cycle.
Medical comorbidities. The CIWA-Ar relies on autonomic signs and subjective symptoms and was not designed for use in nonverbal patients in the ICU. It is possible that the presence of other acute illnesses may contribute to increased CIWA-Ar scores, but we are unaware of any studies that have evaluated such factors.23
However, tremor, which is scored on the CIWA-Ar, can falsely elevate scores if it is caused by something other than acute alcohol withdrawal. Although essential tremors attenuate with acute alcohol use, chronic alcohol use can result in parkinsonism with a resting tremor, and cerebellar degeneration, which can include an action tremor and cerebellar 3-Hz leg tremor.24 Finally, hepatic encephalopathy—a neuropsychiatric syndrome characterized by disturbances in consciousness, mood, behavior, and cognition—can occur in patients with advanced liver disease, which may be precipitated by alcohol use. The clinical presentation and symptom severity of hepatic encephalopathy varies from minor cognitive impairment to gross disorientation, confusion, and agitation,25 all of which can elevate CIWA-Ar scores.
The role of disinhibition
Disinhibition could serve as the “final common pathway” through which CIWA-Ar scores can be falsely elevated.11 For a Figure that illustrates this, see below. Mr. J presented with several variables that could have elevated his CIWA-Ar score; additional potential factors include other psychiatric diagnoses such as bipolar disorder, opiate withdrawal, dementia, drug-seeking behavior, or malingering.26,27
Treating disinhibition in patients with alcohol withdrawal. Continuing to administer escalating doses of benzodiazepines is counterintuitive for benzodiazepine-induced disinhibition. In a study of alcohol withdrawal in rats, antipsychotics evaluated had some beneficial effects on alcohol withdrawal signs.28 In this study, the comparative effectiveness of atypical antipsychotics was as follows: risperidone = quetiapine > ziprasidone > clozapine > olanzapine.
The American Society of Addiction Medicine’s practice guideline advises against using antipsychotics as the sole agent for DTs because these agents are associated with a longer duration of delirium, higher complication rates, and higher mortality.28 However, antipsychotics have a role as an adjunct to benzodiazepines when benzodiazepines don’t sufficiently control agitation, thought disorder, or perceptual disturbances. Although haloperidol use is well established in this scenario, chlorpromazine is contraindicated because it is epileptogenic, and little information is available on atypical antipsychotics.29 If Mr. J had not responded to tapering lorazepam, evidence would support using haloperidol.

Figure: Unifying concept for pathological BZ administration during alcohol withdrawal syndrome: Disinhibition
AWS: alcohol withdrawal syndrome; BZ: benzodiazepine; CIWA-Ar: Clinical Institute Withdrawal Assessment of Alcohol Scale, Revised; GABA: γ-aminobutyric acid
Source: Reference 11Related Resources
- Myrick H, Anton RF. Treatment of alcohol withdrawal. Alcohol Health & Research World. 1998;22(1):38-43. http://pubs.niaaa.nih.gov/publications/arh22-1/38-43.pdf.
- Amato L, Minozzi S, Davoli M. Efficacy and safety of pharmacological interventions for the treatment of the Alcohol Withdrawal Syndrome. Cochrane Database Syst Rev. 2011;(6):CD008537.
Drug Brand Names
- Chlorpromazine • Thorazine
- Clozapine • Clozaril
- Flumazenil • Romazicon
- Haloperidol • Haldol
- Lorazepam • Ativan
- Olanzapine • Zyprexa
- Quetiapine • Seroquel
- Risperidone • Risperdal
- Ziprasidone • Geodon
Disclosures
Dr. Spiegel is on the speaker’s bureau of Sunovion Pharmaceuticals.
Drs. Kumari and Petri report no financial relationships with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgement
The authors thank Amy Herndon for her help in preparing this article.
1. Sullivan JT, Sykora K, Schneiderman J, et al. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict. 1989;84(11):1353-1357.
2. Knott DH, Lerner WD, Davis-Knott T, et al. Decision for alcohol detoxication: a method to standardize patient evaluation. Postgrad Med. 1981;69(5):65-69, 72-75, 78.
3. Wiehl WO, Hayner G, Galloway G. Haight Ashbury Free Clinics’ drug detoxification protocols—Part 4: alcohol. J Psychoactive Drugs. 1994;26(1):57-59.
4. Bostwick JM, Lapid MI. False positives on the clinical institute withdrawal assessment for alcohol-revised: is this scale appropriate for use in the medically ill? Psychosomatics. 2004;45(3):256-261.
5. Diagnostic and statistical manual of mental disorders, 4th ed text rev. Washington DC: American Psychiatric Association; 2000.
6. Schacht JP, Randall PK, Waid LR, et al. Neurocognitive performance, alcohol withdrawal, and effects of a combination of flumazenil and gabapentin in alcohol dependence. Alcohol Clin Exp Res. 2011;35(11):2030-2038.
7. Monte R, Rabuñal R, Casariego E, et al. Risk factors for delirium tremens in patients with alcohol withdrawal syndrome in a hospital setting. Eur J Intern Med. 2009;20(7):690-694.
8. Saitz R, O’Malley SS. Pharmacotherapies for alcohol abuse. Withdrawal and treatment. Med Clin North Am. 1997;81(4):881-907.
9. Hack JB, Hoffmann RS, Nelson LS. Resistant alcohol withdrawal: does an unexpectedly large sedative requirement identify these patients early? J Med Toxicol. 2006;2(2):55-60.
10. Pittman B, Gueorguieva R, Krupitsky E, et al. Multidimensionality of the Alcohol Withdrawal Symptom Checklist: a factor analysis of the Alcohol Withdrawal Symptom Checklist and CIWA-Ar. Alcohol Clin Exp Res. 2007;31(4):612-618.
11. Deakin JB, Aitken MR, Dowson JH, et al. Diazepam produces disinhibitory cognitive effects in male volunteers. Psychopharmacology (Berl). 2004;173(1-2):88-97.
12. Hornberger M, Geng J, Hodges JR. Convergent grey and white matter evidence of orbitofrontal cortex changes related to disinhibition in behavioural variant frontotemporal dementia. Brain. 2011;134(pt 9):2502-2512.
13. Jones KA, Nielsen S, Bruno R, et al. Benzodiazepines - their role in aggression and why GPs should prescribe with caution. Aust Fam Physician. 2011;40(11):862-865.
14. Scott EL, Hulvershorn L. Anxiety disorders with comorbid substance abuse. Psychiatric Times. 2011; 28(9).
15. Faingold CL, Knapp DJ, Chester JA, et al. Integrative neurobiology of the alcohol withdrawal syndrome—from anxiety to seizures. Alcohol Clin Exp Res. 2004;28(2):268-278.
16. Clegg A, Young JB. Which medications to avoid in people at risk of delirium: a systematic review. Age Ageing. 2011;40(1):23-29.
17. Hecksel KA, Bostwick JM, Jaeger TM, et al. Inappropriate use of symptom-triggered therapy for alcohol withdrawal in the general hospital. Mayo Clin Proc. 2008;83(3):274-279.
18. Lader M. Benzodiazepines revisited—will we ever learn? Addiction. 2011;106(12):2086-2109.
19. Mellos E, Liappas I, Paparrigopoulos T. Comorbidity of personality disorders with alcohol abuse. In Vivo. 2010;24(5):761-769.
20. Tragesser SL, Sher KJ, Trull TJ, et al. Personality disorder symptoms, drinking motives, and alcohol use and consequences: cross-sectional and prospective mediation. Exp Clin Psychopharmacol. 2007;15(3):282-292.
21. Oscar-Berman M, Valmas MM, Sawyer KS, et al. Frontal brain dysfunction in alcoholism with and without antisocial personality disorder. Neuropsychiatr Dis Treat. 2009;5:309-326.
22. Dom G, De Wilde B, Hulstijn W, et al. Behavioural aspects of impulsivity in alcoholics with and without a cluster-B personality disorder. Alcohol Alcohol. 2006;41(4):412-420.
23. de Wit M, Jones DG, Sessler CN, et al. Alcohol-use disorders in the critically ill patient. Chest. 2010;138(4):994-1003.
24. Mostile G, Jankovic J. Alcohol in essential tremor and other movement disorders. Mov Disord. 2010;25(14):2274-2284.
25. Crone CC, Gabriel GM, DiMartini A. An overview of psychiatric issues in liver disease for the consultation-liaison psychiatrist. Psychosomatics. 2006;47(3):188-205.
26. Reoux JP, Oreskovich MR. A comparison of two versions of the clinical institute withdrawal assessment for alcohol: the CIWA-Ar and CIWA-AD. Am J Addict. 2006;15(1):85-93.
27. Gray S, Borgundvaag B, Sirvastava A, et al. Feasibility and reliability of the SHOT: a short scale for measuring pretreatment severity of alcohol withdrawal in the emergency department. Acad Emerg Med. 2010;17(10):1048-1054.
28. Uzbay TI. Atypical antipsychotic drugs and ethanol withdrawal syndrome: a review. Alcohol Alcohol. 2012;47(1):33-41.
29. McKeon A, Frye MA, Delanty N. The alcohol withdrawal syndrome. J Neurol Neurosurg Psychiatry. 2008;79(8):854-862.
1. Sullivan JT, Sykora K, Schneiderman J, et al. Assessment of alcohol withdrawal: the revised clinical institute withdrawal assessment for alcohol scale (CIWA-Ar). Br J Addict. 1989;84(11):1353-1357.
2. Knott DH, Lerner WD, Davis-Knott T, et al. Decision for alcohol detoxication: a method to standardize patient evaluation. Postgrad Med. 1981;69(5):65-69, 72-75, 78.
3. Wiehl WO, Hayner G, Galloway G. Haight Ashbury Free Clinics’ drug detoxification protocols—Part 4: alcohol. J Psychoactive Drugs. 1994;26(1):57-59.
4. Bostwick JM, Lapid MI. False positives on the clinical institute withdrawal assessment for alcohol-revised: is this scale appropriate for use in the medically ill? Psychosomatics. 2004;45(3):256-261.
5. Diagnostic and statistical manual of mental disorders, 4th ed text rev. Washington DC: American Psychiatric Association; 2000.
6. Schacht JP, Randall PK, Waid LR, et al. Neurocognitive performance, alcohol withdrawal, and effects of a combination of flumazenil and gabapentin in alcohol dependence. Alcohol Clin Exp Res. 2011;35(11):2030-2038.
7. Monte R, Rabuñal R, Casariego E, et al. Risk factors for delirium tremens in patients with alcohol withdrawal syndrome in a hospital setting. Eur J Intern Med. 2009;20(7):690-694.
8. Saitz R, O’Malley SS. Pharmacotherapies for alcohol abuse. Withdrawal and treatment. Med Clin North Am. 1997;81(4):881-907.
9. Hack JB, Hoffmann RS, Nelson LS. Resistant alcohol withdrawal: does an unexpectedly large sedative requirement identify these patients early? J Med Toxicol. 2006;2(2):55-60.
10. Pittman B, Gueorguieva R, Krupitsky E, et al. Multidimensionality of the Alcohol Withdrawal Symptom Checklist: a factor analysis of the Alcohol Withdrawal Symptom Checklist and CIWA-Ar. Alcohol Clin Exp Res. 2007;31(4):612-618.
11. Deakin JB, Aitken MR, Dowson JH, et al. Diazepam produces disinhibitory cognitive effects in male volunteers. Psychopharmacology (Berl). 2004;173(1-2):88-97.
12. Hornberger M, Geng J, Hodges JR. Convergent grey and white matter evidence of orbitofrontal cortex changes related to disinhibition in behavioural variant frontotemporal dementia. Brain. 2011;134(pt 9):2502-2512.
13. Jones KA, Nielsen S, Bruno R, et al. Benzodiazepines - their role in aggression and why GPs should prescribe with caution. Aust Fam Physician. 2011;40(11):862-865.
14. Scott EL, Hulvershorn L. Anxiety disorders with comorbid substance abuse. Psychiatric Times. 2011; 28(9).
15. Faingold CL, Knapp DJ, Chester JA, et al. Integrative neurobiology of the alcohol withdrawal syndrome—from anxiety to seizures. Alcohol Clin Exp Res. 2004;28(2):268-278.
16. Clegg A, Young JB. Which medications to avoid in people at risk of delirium: a systematic review. Age Ageing. 2011;40(1):23-29.
17. Hecksel KA, Bostwick JM, Jaeger TM, et al. Inappropriate use of symptom-triggered therapy for alcohol withdrawal in the general hospital. Mayo Clin Proc. 2008;83(3):274-279.
18. Lader M. Benzodiazepines revisited—will we ever learn? Addiction. 2011;106(12):2086-2109.
19. Mellos E, Liappas I, Paparrigopoulos T. Comorbidity of personality disorders with alcohol abuse. In Vivo. 2010;24(5):761-769.
20. Tragesser SL, Sher KJ, Trull TJ, et al. Personality disorder symptoms, drinking motives, and alcohol use and consequences: cross-sectional and prospective mediation. Exp Clin Psychopharmacol. 2007;15(3):282-292.
21. Oscar-Berman M, Valmas MM, Sawyer KS, et al. Frontal brain dysfunction in alcoholism with and without antisocial personality disorder. Neuropsychiatr Dis Treat. 2009;5:309-326.
22. Dom G, De Wilde B, Hulstijn W, et al. Behavioural aspects of impulsivity in alcoholics with and without a cluster-B personality disorder. Alcohol Alcohol. 2006;41(4):412-420.
23. de Wit M, Jones DG, Sessler CN, et al. Alcohol-use disorders in the critically ill patient. Chest. 2010;138(4):994-1003.
24. Mostile G, Jankovic J. Alcohol in essential tremor and other movement disorders. Mov Disord. 2010;25(14):2274-2284.
25. Crone CC, Gabriel GM, DiMartini A. An overview of psychiatric issues in liver disease for the consultation-liaison psychiatrist. Psychosomatics. 2006;47(3):188-205.
26. Reoux JP, Oreskovich MR. A comparison of two versions of the clinical institute withdrawal assessment for alcohol: the CIWA-Ar and CIWA-AD. Am J Addict. 2006;15(1):85-93.
27. Gray S, Borgundvaag B, Sirvastava A, et al. Feasibility and reliability of the SHOT: a short scale for measuring pretreatment severity of alcohol withdrawal in the emergency department. Acad Emerg Med. 2010;17(10):1048-1054.
28. Uzbay TI. Atypical antipsychotic drugs and ethanol withdrawal syndrome: a review. Alcohol Alcohol. 2012;47(1):33-41.
29. McKeon A, Frye MA, Delanty N. The alcohol withdrawal syndrome. J Neurol Neurosurg Psychiatry. 2008;79(8):854-862.
When anticonvulsants might work best
Alcohol withdrawal: When to choose an adjunctive anticonvulsant
Benzodiazepines are the mainstay of alcohol detoxification treatment, with extensive evidence supporting their efficacy and relative safety.1 The risk of benzodiazepine-alcohol interaction, however, and psychomotor and cognitive impairments associated with benzodiazepine use may limit early rehabilitation efforts in hospitalized patients.2 Cross-tolerance with alcohol also limits benzodiazepines’ potential benefit in outpatients with substance use disorders.
Adding anticonvulsants to acute benzodiazepine therapy has been shown to decrease alcohol withdrawal symptom severity, reduce seizure risk, and support recovery, particularly in patients with multiple alcohol withdrawal episodes. After detoxification, long-term anticonvulsant use may reduce relapse risk by decreasing post-cessation craving, without abuse liability.3
Although not all studies endorse adding anticonvulsants to benzodiazepines for managing alcohol withdrawal syndrome (AWS),4 we present 3 cases in which anticonvulsants were used successfully as adjuncts to lorazepam. Valproic acid, levetiracetam, and gabapentin offer advantages in acute and long-term therapy of alcohol dependence with efficacy in AWS, low abuse potential, benign safety profile, and mood-stabilizing properties.
Neurobiologic rationale
AWS manifests as a cluster of clinical symptoms including delirium tremens (DTs) and seizures (Table 1). Its pathophysiology can be explained by alcohol’s agonist effect on the gamma-aminobutyric acid (GABA) system and antagonist effect on the glutamatergic system (Table 2).5
Chronic alcohol intake leads to neuroadaptation in the brain in the form of down-regulation of GABAA receptors and upregulation of N-methyl-D-aspartate receptors. During alcohol withdrawal, this neuroadaptation leads to a decrease in central GABA activity and an increase in glutamate activity, resulting in hyperexcitation, anxiety, and seizures.6
Little data exist regarding time to relapse after detoxification in alcohol-dependent patients. One theory—called “protracted withdrawal syndrome” (Table 1)—suggests that abstinent alcoholics return to drinking because of the same, but attenuated, neuroadaptations that trigger acute AWS.7
Advantages of adjunct therapy. Ntais et al8 evaluated benzodiazepines’ effectiveness and safety in treating AWS in a clinical review of 57 randomized, controlled trials totaling 4,051 patients. Benzodiazepines showed similar success rates as other drugs (relative risk [RR] 1.00) or anticonvulsants in particular (RR 0.88), as measured by changes in Clinical Institute Withdrawal Assessment for Alcohol (CIWA-Ar) scores at the end of treatment. Benzodiazepines also offered significant benefit for seizure control compared with nonanticonvulsants (RR 0.23), but less when compared with anti convulsants (RR 1.99).
Although the literature does not support anticonvulsant use for monotherapy in AWS, anticonvulsants show potential as adjunctive therapy. Valproic acid, levetiracetam, and gabapentin offer unique mechanisms of action (Table 3) and demonstrate advantages over benzodiazepine monotherapy for AWS. Adjunctive use of valproic acid,8,9 levetiracetam,10 and gabapentin11,12 in detoxification also has demonstrated efficacy in reducing risk of relapse and delaying relapse.
The neurobiologic rationale for using anticonvulsants in acute AWS is speculative, but these agents appear to:
- inhibit “kindling” (neuronal changes that may be associated with repeated intoxications)
- facilitate GABAergic mechanisms.9
Table 1
Alcohol withdrawal: Acute vs long-term symptoms
| Alcohol withdrawal syndrome | Protracted withdrawal syndrome | |
|---|---|---|
| Description | Cluster of symptoms in alcohol-dependent persons after heavy or prolonged alcohol use has lessened or ceased | Constellation of symptoms lasting weeks to months after alcohol use ends |
| Presentation | Develops during acute detoxification period and lasts 5 to 7 days | Develops after 5- to 7-day acute detoxification period and may persist for 1 year |
| Symptoms | Mild: insomnia, tremor, anxiety, GI upset, headache, diaphoresis, palpitations, anorexia Severe: alcoholic hallucinosis Seizures (generalized tonic-clonic) occur in up to 25% of withdrawal episodes, usually within 24 hours after alcohol cessation Delirium tremens (characterized by hallucinations, disorientation, tachycardia, hypertension, low-grade fever, agitation, and diaphoresis) occurs in up to 5% of patients undergoing withdrawal, may be delayed 4 to 5 days, and has mortality rates reaching 15% | Sleep disruption; anxiety; depressive symptoms; irritability; increased breathing rate, body temperature, blood pressure, and pulse |
| GI: gastrointestinal | ||
| Source: Click here for a bibliography | ||
Table 2
How alcohol affects GABA and glutamate neurotransmitters
| GABA | Glutamate | |
|---|---|---|
| GABA, the brain’s primary inhibitory neurotransmitter, renders nerve cells less sensitive to further signaling | Glutamate, the brain’s major excitatory neurotransmitter, renders nerve cells more sensitive to further signaling | |
| Alcohol facilitates the inhibitory function of the GABAA receptor, allowing more GABA to traverse the receptor, and leading to alcohol’s intoxicating effects | Alcohol seems to inhibit the excitatory function of the NMDA glutamate receptor, believed to play a role in memory, learning, and generation of seizures | |
| During alcohol withdrawal, brain GABA concentrations fall below normal and GABAA receptor sensitivity may be reduced | Long-term alcohol exposure produces an adaptive increase in the function of NMDA receptors and results in development of glutamate-NMDA supersensitivity | |
| In the absence of alcohol, the resulting decrease in inhibitory function may contribute to symptoms of CNS hyperactivity associated with acute and protracted alcohol withdrawal | Acute alcohol withdrawal activates glutamate systems, leading to autonomic nervous system hyperactivity; alcohol withdrawal seizures are associated with increased NMDA receptor function | |
| GABA: gamma-aminobutyric acid; NMDA: N-methyl-D-aspartate | ||
| Source: Click here for a bibliography | ||
Table 3
Mechanisms of action of benzodiazepines vs 3 anticonvulsants
| Agent | Mechanism of action |
|---|---|
| Benzodiazepines | Activate GABAA chloride ionophore, increasing affinity of GABAA receptor for GABA and augmenting frequency of chloride channel openinga |
| Valproic acid | GABA modulation and possibly second messenger systems; may inhibit Na1+ and/or Ca2+ channel, thereby boosting GABA and glutamate actionb |
| Levetiracetam | Decreases high voltage activated Ca2+ channels; unique binding site (synaptic vesicle protein SV2A) is thought to be involved in calcium-dependent regulation of neurotransmitter vesicle exocytosisc |
| Gabapentin | GABA analog; unique binding site (Ca2+ channel subunit in brain) decreases calcium influx and inhibits release of excitatory amino acids and monoaminesd |
| GABA: gamma-aminobutyric acid | |
| Source: Click here for a bibliography | |
CASE REPORT 1: Valproic acid for alcohol overdose
After attempting suicide with an alcohol overdose, Ms. J, age 45, is transferred from the emergency room (ER) to our psychiatry consult service 10 hours after admission. Her symptoms include nausea, tremor, headaches, agitation, disorientation, and auditory hallucinations.
Medical history reveals 25 years of alcohol dependence, multiple hospitalizations for withdrawal, and many failed attempts to quit. Ms. J reports consuming an average of 16 drink equivalents (eg, 12 oz beers) daily but denies illicit drug use.
Lab values on admission include blood alcohol concentration (BAC) 290 mg/dL (0.29%), mean corpuscular volume (MCV) 96 fL, gamma-glutamyltransferase (GGT) 164 U/L, aspartate aminotransferase (AST) 43 U/L, alanine aminotransferase (ALT) 31 U/L, and alkaline phosphatase (ALP) 151 U/L. Urine drug screen, acetaminophen, salicylate, vitamin B1 (thiamine), B12 (cyanocobalamin), B9 (folate), and electrolytes (including magnesium) are normal.
We assess alcohol withdrawal severity using the CIWA-Ar (Click here to view/download a copy of this scale). Ms. J’s initial score is 17, indicating a risk of moderate alcohol withdrawal if untreated.
In the ER, Ms. J is placed on a symptom-triggered benzodiazepine detoxification protocol with lorazepam. We add IV valproic acid, 1,250 mg (based on 20 mg/kg body weight)13 divided into 2 doses over the first 24 hours, then maintain IV valproic acid at 500 mg twice daily (Table 4). Within 12 hours of starting combination therapy, Ms. J scores 7 on the CIWA-Ar—indicating mild withdrawal—with subsequent scores <5. She scores 0 with no residual withdrawal symptoms within 36 hours.
Ms. J requires lorazepam, 7 mg, during the 10 hours before valproic acid is added. She requires only 2 mg lorazepam over the next 3 days and reports no side effects related to IV valproic acid. At discharge, Ms. J begins extended-release oral valproic acid, 1,250 mg (based on 25 mg/kg body weight)13 once daily for 2 weeks, until she can obtain outpatient follow-up.
Table 4
Benzodiazepines and anticonvulsants for alcohol detoxification
| Benzodiazepines | Valproic acid | Levetiracetam | Gabapentin | |
|---|---|---|---|---|
| Loading dose | None | 20 mg/kg of body weight, divided into 2 doses for first 24 hours | 1,500 mg IV once daily | 400 mg PO qid |
| Maintenance dose | Day 1: 2 mg tid Day 2: 2 mg morning, 1 mg afternoon, 2 mg evening Day 3: 1 mg tid Day 4: 1 mg bid Day 5: 1 mg Day 6: none | 500 mg IV bid | Either 500 mg IV tid or 1,000 mg PO bid after 2 to 3 days of treatment | 1,200 mg PO tid |
| Side effects | Impaired consciousness, respiratory depression, hypotension | Dizziness, drowsiness, hair loss/thinning, nausea, tremor, weight gain | Somnolence, asthenia, dizziness, coordination difficulties | Somnolence, dizziness, ataxia, fatigue |
| Drug interactions | ↑ BZ: cimetidine, oral contraceptives, ethanol (acute), disulfiram, isoniazid, propranolol ↓ BZ: rifampin, ethanol (chronic) | ↑ VPA: aspirin, felbamate, fluoxetine, isoniazid ↓ VPA: carbamazepine, lamotrigine, phenobarbital, phenytoin, ritonavir | None | ↓ GBP 20%: antacids |
| BZ: benzodiazepine; GBP: gabapentin; PO: per os (by mouth); VPA: valproic acid | ||||
| Source: Click here for a bibliography | ||||
Less lorazepam needed
Adjunctive anticonvulsants can reduce the amount of lorazepam required during detoxification.14,15 Compared with benzodiazepine monotherapy, the advantages of combination therapy—particularly in outpatient alcohol withdrawal treatment and relapse prevention—include:
- minimal interaction with alcohol (avoiding increased psychomotor deficits, cognitive impairment, and intoxication)15
- lower abuse potential
- possible efficacy in mood stabilization before, during, and after withdrawal (Table 5).16
Given the risk of seizures during AWS, anticonvulsants seem to make empirical sense. One study reported a 1% incidence of withdrawal-related seizures in 545 alcohol-dependent inpatients treated with valproic acid.17 Another case series of 37 patients found no acute sequelae when valproic acid was used for AWS.18
Anticonvulsants such as valproic acid may reduce the frequency and severity of alcohol relapse, whereas benzodiazepines may increase relapse risk.19 During a 6-week trial, patients receiving valproic acid maintenance therapy had greater abstinence rates and improved drinking outcomes compared with detoxification-only groups.9
One disadvantage of valproic acid is potential hepatotoxicity, an important consideration in patients with liver damage. Fortunately, Ms. J’s AST and ALT values remained within normal limits during valproic acid treatment.
Table 5
Pharmacologic profiles of benzodiazepines vs 3 anticonvulsants
| Benzodiazepines | Valproic acid | Levetiracetam | Gabapentin | |
|---|---|---|---|---|
| Metabolism | CYP 2C19: diazepam CYP 3A3/4: alprazolam, clonazepam, diazepam, triazolam Phase II only: lorazepam, temazepam, oxazepam | >95% hepatic, of which <20% occurs via CYP isoenzymes | Not extensively metabolized; renal clearance; not involved with hepatic CYP isoenzymes | Not metabolized; secreted via kidneys as unchanged drug |
| Sedation | Mild to moderate | Mild to moderate | Mild to moderate | Moderate to severe |
| Synergistic effects with alcohol | Yes | No | No | No |
| Paradoxical disinhibition | Yes | No | No | No |
| Risk of addiction in outpatient therapy | Yes | No | No | No |
| CYP: cytochrome P450 | ||||
| Source: Click here for a bibliography | ||||
CASE REPORT 2: Levetiracetam for withdrawal seizures
Mr. H, age 42, presents to the ER after suffering a seizure. His medical history includes hypertension, alcohol dependence, and seizures during alcohol withdrawal. He denies a history of psychiatric illness, and his family history is unknown. He is noncompliant with hypertension treatment, which includes clonidine. Mr. H reports his usual alcohol consumption as a 6-pack of beer nightly during the week and a 12-pack nightly on weekends. He says his last drink was 4 days before admission.
Mr. H scores 19 on the CIWA-Ar, placing him at risk for moderate withdrawal. Head CT shows diffuse atrophy, without evidence of an acute intracranial process. BAC is zero on admission, and urine drug screen is negative. Amylase, lipase, and lactate dehydrogenase (LDH) levels suggest acute pancreatitis. AST is elevated to 131 U/L, ALT is elevated to 42 U/L, but MCV is within normal limits.
The psychiatric service is consulted on day 2 of admission, and we prescribe levetiracetam, 500 mg IV every 8 hours.20 IV lorazepam also is available as needed: 1 mg every 8 hours for the first 2 days, then 1 mg every 12 hours for 2 days, then 1 mg every 24 hours. The patient’s CIWA-Ar score is 9 on days 2 and 3 of admission, followed by scores consistently between 2 and 3 after scheduled levetiracetam administration. Mr. H requires 3 mg of lorazepam the remainder of his hospitalization. He is discharged on day 7 with a CIWA-Ar score of 2, and reports no adverse effects related to levetiracetam. He leaves the hospital with a 2-week prescription for oral levetiracetam, 500 mg tid.
Advantages of levetiracetam
Levetiracetam is FDA-approved for adjunctive treatment of adults with partial-onset seizures.21 Successful AWS treatment with adjunctive levetiracetam has been supported by few but promising studies.10,20 Potential advantages of levetiracetam in detoxification include:
- a lack of GABAergic properties, which limits the risk of intoxication or respiratory insufficiency when combined with alcohol21
- low drug-drug interaction risk because of nonhepatic metabolism and primary renal excretion.22,23
We selected levetiracetam for Mr. H because of his history of alcohol withdrawal seizures and acute pancreatitis. Anticonvulsants may be more effective than lorazepam in reducing the risk of alcohol withdrawal seizures,24 and we felt valproic acid might not be safe for him because of its low but real risk of pancreatitis.13 We based our levetiracetam dosing on a small open-label trial20 and product information for treating adults with partial-onset seizures.25
Studies also demonstrate levetiracetam’s potential for relapse prevention during outpatient therapy. In a 10-week trial, levetiracetam decreased the number of standard drinks in alcohol-dependent patients from 5.3 to 1.7 per day.10 This was a small open trial, however, and large controlled trials support the usefulness of other, FDA-approved medications—including disulfiram, naltrexone, and acamprosate—for alcohol relapse prevention.
CASE REPORT 3: Gabapentin for acute withdrawal
Mr. B, age 38, presents to the ER after a 13-day alcohol binge. He has been drinking increasing amounts of alcohol over 6 weeks. Three months earlier, Mr. B was admitted for alcohol withdrawal treatment and received 49 mg of lorazepam over 3 days. This resulted in his transfer from the step-down unit to the intensive care unit for increased agitation, possibly caused by paradoxical disinhibition from excessive lorazepam use.26
Mr. B’s medical history is significant for alcohol-induced seizures, DTs, traumatic brain injury related to craniotomy, and right arm amputation. Mr. B drinks approximately 24 beers per day. He denies tobacco use but admits to past use of cocaine, marijuana, and heroin.
On admission, Mr. B’s BAC is 360 mg/dL (0.36%), AST is elevated at 72 U/L, ALT at 42 U/L, and LDH significantly elevated at 384 U/L. Urine drug screen is negative, and his CIWA-Ar score is 23. His score of –1 on the Richmond Agitation and Sedation Scale (RASS)27 correlates with very mild sedation.
Guided by Bonnet et al28 and clinical experience, we start Mr. B on gabapentin, 1,200 mg tid, and IV lorazepam, 2 mg every 8 hours as needed for breakthrough withdrawal. We decrease lorazepam by 50% every other day until Mr. B is discharged. On days 2, 3, and 4, Mr. B’s CIWA-Ar scores are 6, 9, and 2, respectively. His RASS score drops from –1 on days 1 and 2 to 0 until discharge, indicating an alert and calm state.
Mr. B requires a total of 2 mg of lorazepam throughout hospitalization. He finishes alcohol detoxification on day 4 and is discharged with a prescription for gabapentin, 1,200 mg tid. Two weeks later, when he is admitted to a 28-day inpatient alcohol rehabilitation unit, Mr. B has not relapsed.
More abstinent days
Gabapentin is FDA-approved as adjunctive therapy for partial seizures. Off-label, it has been generally efficacious as an adjunct in alcohol detoxification.29-32 We chose adjunctive anticonvulsant therapy for Mr. B because of his history of alcohol-induced seizures. We chose gabapentin instead of valproic acid because of Mr. B’s liver damage and gabapentin’s lack of hepatic metabolism.
Gabapentin may reduce alcohol consumption and craving in alcohol-dependent patients. By increasing the number of abstinent days, gabapentin may help patients maintain abstinence.33 Gabapentin does not appear to interact clinically with alcohol, causing neither sedation nor synergistic effects.34 Its relative lack of abuse potential may be valuable in outpatient alcohol withdrawal treatment and in maintaining alcohol abstinence after detoxification.
Related resource
- Asplund CA, Aaronson JW, Aaronson HE. 3 regimens for alcohol withdrawal and detoxification. J Fam Pract. 2004;53(7):545-554. www.jfponline.com/Pages.asp?AID=1730.
Drug brand names
- Acamprosate • Campral
- Alprazolam • Xanax
- Carbamazepine • Carbatrol
- Cimetidine • Tagamet
- Clonazepam • Klonopin
- Clonidine • Catapres
- Diazepam • Valium
- Disulfiram • Antabuse
- Felbamate • Felbatol
- Fluoxetine • Prozac
- Gabapentin • Neurontin
- Isoniazid • Nydrazid
- Lamotrigine • Lamictal
- Levetiracetam • Keppra
- Lorazepam • Ativan
- Naltrexone • ReVia, Vivitrol
- Oxazepam • Serax
- Phenobarbital • Luminal
- Phenytoin • Dilantin
- Propranolol • Inderal
- Rifampin • Rifadin
- Ritonavir • Norvir
- Temazepam • Restoril
- Triazolam • Halcion
- Valproic acid • Depakote, Depakene
Disclosures
Dr. Spiegel is a speaker for Pfizer Inc. and GlaxoSmithKline.
Dr. Radac reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The authors thank Rishi Laroia, MD, Robert Swanson, MD, and Adam W. Coe, MD for their contributions to this article.
1. Mayo-Smith MF, Cushman P, Hill AJ, et al. Pharmacological management of alcohol withdrawal. JAMA. 1997;278:144-151.
2. Myrick H, Brady KT, Malcom R. Divalproex in the treatment of alcohol withdrawal—statistical data included. Am J Drug Alcohol Abuse. 2000;26(1):155-160.
3. Johnson BA, Swift RM, Addolorato G, et al. Safety and efficacy of GABAergic medications for treating alcoholism. Alcohol Clin Exp Res. 2005;29(2):248-254.
4. Lum E, Gorman SK, Slavik RS. Valproic acid management of acute alcohol withdrawal. Ann Pharmacother. 2006;40(3):441-448.
5. Trevisan LA, Boutros N, Petrakis IL, et al. Complications of alcohol withdrawal: pathophysiological insights. Alcohol Health Res World. 1998;22(1):61-66.
6. Esel E. Neurobiology of alcohol withdrawal inhibitory and excitatory neurotransmitters. Turk Psikiyatri Derg. 2006;7(2):129-138.
7. Myrick H, Brady KT. The use of divalproex in the treatment of addictive disorders. Psychopharmacol Bull. 2003;37(suppl 2):89-97.
8. Brady KT, Myrick H, Henderson S, et al. The use of divalproex in alcohol relapse prevention: a pilot study. Drug Alcohol Depend. 2002;67(3):323-330.
9. Longo L, Campbell T, Hubatch S. Divalproex sodium (Depakote) for alcohol withdrawal and relapse prevention. J Addict Dis. 2002;21(2):55-64.
10. Sarid-Segal O, Piechniczek-Buczek J, Knapp C, et al. The effects of levetiracetam on alcohol consumption in alcohol-dependent subjects: an open label study. Am J Drug Alcohol Abuse. 2008;34(4):441-447.
11. American Psychiatric Association. Treatments of psychiatric disorders: a task force report of the American Psychiatric Association. Washington, DC: American Psychiatric Association Press; 1989:187.
12. Mason BJ, Light JM, Williams LD, et al. Proof-of-concept human laboratory study for protracted abstinence in alcohol dependence: effects of gabapentin. Addict Biol. 2009;14(1):73-83.
13. Physicians’ Desk Reference 2009. 63rd ed. Montvale, NJ: Physicians’ Desk Reference; 2008:423-431.
14. Malcolm R, Ballenger JC, Sturgis ET, et al. Double-blind controlled trial comparing carbamazepine to oxazepam treatment of alcohol withdrawal. Am J Psychiatry. 1989;146:617-621.
15. Myrick H, Anton R, Voronin K, et al. A double-blind evaluation of gabapentin on alcohol effects and drinking in a clinical laboratory paradigm. Alcohol Clin Exp Res. 2007;31(2):221-227.
16. Malcolm R, Myrick H, Brady KT, et al. Update on anticonvulsants for the treatment of alcohol withdrawal. Am J Addict. 2001;10(suppl):16-23.
17. Davis LL, Ryan W, Adinoff B, et al. Comprehensive review of the psychiatric uses of valproate. J Clin Psychopharm. 2000;20(1 suppl 1):1S-17S.
18. Rosenthal RN, Perkel C, Singh P, et al. A pilot open randomized trial of valproate and phenobarbital in the treatment of acute alcohol withdrawal. Am J Addict. 1998;7:189-197.
19. Book SW, Myrick H. Novel anticonvulsants in the treatment of alcoholism. Expert Opin Investig Drugs. 2005;14(4):371-376.
20. Krebs M, Leopold K, Richter C, et al. Levetiracetam for the treatment of alcohol withdrawal syndrome: an open-label pilot trial. J Clin Psychopharmacol. 2006;26(3):347-349.
21. LaRoche SM, Helmers SL. The new antiepileptic drugs: scientific review. JAMA. 2004;291:605-614.
22. Chabolla DR, Harnois DM, Meschia JF. Levetiracetam monotherapy for liver transplant patients with seizures. Transplant Proc. 2003;35:1480-1481.
23. Paul F, Meencke HJ. Levetiracetam in focal epilepsy and hepatic porphyria: a case report. Epilepsia. 2004;45:559-560.
24. Ntais C, Pakos E, Kyzas P, et al. Benzodiazepines for alcohol withdrawal. Cochrane Database Syst Rev. 2005;(3):CD005063.-
25. Physicians’ Desk Reference 2009. 63rd ed. Montvale, NJ: Physicians’ Desk Reference; 2008:3131-3143.
26. Saias T, Gallarda T. Paradoxical aggressive reactions to benzodiazepine use: a review. Encephale. 2008;34(4):330-336.
27. Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation–Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166:1338-1344.
28. Bonnet U, Banger M, Leweke FM, et al. Treatment of acute alcohol withdrawal with gabapentin: results from a controlled two-center trial. J Clin Psychopharmacol. 2003;23(5):514-519.
29. Myrick H, Malcolm R, Brady KT. Gabapentin treatment of alcohol withdrawal. Am J Psychiatry. 1998;155:1626.-
30. Bonnet U, Banger M, Leweke FM, et al. Treatment of alcohol withdrawal syndrome with gabapentin. Pharmacopsychiatry. 1999;32:107-109.
31. Mariani JJ, Rosenthal RN, Tross S, et al. A randomized, open-label, controlled trial of gabapentin and phenobarbital in the treatment of alcohol withdrawal. Am J Addict. 2006;15(1):76-84.
32. Voris J, Smith NL, Rao SM, et al. Gabapentin for the treatment of ethanol withdrawal. Subst Abus. 2003;24(2):129-132.
33. Furieri FA, Nakamura-Palacios EM. Gabapentin reduces alcohol consumption and craving: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2007;68(11):1691-1700.
34. Bisaga A, Evans SM. The acute effects of gabapentin in combination with alcohol in heavy drinkers. Drug Alcohol Depend. 2006;83(1):25-32.
Benzodiazepines are the mainstay of alcohol detoxification treatment, with extensive evidence supporting their efficacy and relative safety.1 The risk of benzodiazepine-alcohol interaction, however, and psychomotor and cognitive impairments associated with benzodiazepine use may limit early rehabilitation efforts in hospitalized patients.2 Cross-tolerance with alcohol also limits benzodiazepines’ potential benefit in outpatients with substance use disorders.
Adding anticonvulsants to acute benzodiazepine therapy has been shown to decrease alcohol withdrawal symptom severity, reduce seizure risk, and support recovery, particularly in patients with multiple alcohol withdrawal episodes. After detoxification, long-term anticonvulsant use may reduce relapse risk by decreasing post-cessation craving, without abuse liability.3
Although not all studies endorse adding anticonvulsants to benzodiazepines for managing alcohol withdrawal syndrome (AWS),4 we present 3 cases in which anticonvulsants were used successfully as adjuncts to lorazepam. Valproic acid, levetiracetam, and gabapentin offer advantages in acute and long-term therapy of alcohol dependence with efficacy in AWS, low abuse potential, benign safety profile, and mood-stabilizing properties.
Neurobiologic rationale
AWS manifests as a cluster of clinical symptoms including delirium tremens (DTs) and seizures (Table 1). Its pathophysiology can be explained by alcohol’s agonist effect on the gamma-aminobutyric acid (GABA) system and antagonist effect on the glutamatergic system (Table 2).5
Chronic alcohol intake leads to neuroadaptation in the brain in the form of down-regulation of GABAA receptors and upregulation of N-methyl-D-aspartate receptors. During alcohol withdrawal, this neuroadaptation leads to a decrease in central GABA activity and an increase in glutamate activity, resulting in hyperexcitation, anxiety, and seizures.6
Little data exist regarding time to relapse after detoxification in alcohol-dependent patients. One theory—called “protracted withdrawal syndrome” (Table 1)—suggests that abstinent alcoholics return to drinking because of the same, but attenuated, neuroadaptations that trigger acute AWS.7
Advantages of adjunct therapy. Ntais et al8 evaluated benzodiazepines’ effectiveness and safety in treating AWS in a clinical review of 57 randomized, controlled trials totaling 4,051 patients. Benzodiazepines showed similar success rates as other drugs (relative risk [RR] 1.00) or anticonvulsants in particular (RR 0.88), as measured by changes in Clinical Institute Withdrawal Assessment for Alcohol (CIWA-Ar) scores at the end of treatment. Benzodiazepines also offered significant benefit for seizure control compared with nonanticonvulsants (RR 0.23), but less when compared with anti convulsants (RR 1.99).
Although the literature does not support anticonvulsant use for monotherapy in AWS, anticonvulsants show potential as adjunctive therapy. Valproic acid, levetiracetam, and gabapentin offer unique mechanisms of action (Table 3) and demonstrate advantages over benzodiazepine monotherapy for AWS. Adjunctive use of valproic acid,8,9 levetiracetam,10 and gabapentin11,12 in detoxification also has demonstrated efficacy in reducing risk of relapse and delaying relapse.
The neurobiologic rationale for using anticonvulsants in acute AWS is speculative, but these agents appear to:
- inhibit “kindling” (neuronal changes that may be associated with repeated intoxications)
- facilitate GABAergic mechanisms.9
Table 1
Alcohol withdrawal: Acute vs long-term symptoms
| Alcohol withdrawal syndrome | Protracted withdrawal syndrome | |
|---|---|---|
| Description | Cluster of symptoms in alcohol-dependent persons after heavy or prolonged alcohol use has lessened or ceased | Constellation of symptoms lasting weeks to months after alcohol use ends |
| Presentation | Develops during acute detoxification period and lasts 5 to 7 days | Develops after 5- to 7-day acute detoxification period and may persist for 1 year |
| Symptoms | Mild: insomnia, tremor, anxiety, GI upset, headache, diaphoresis, palpitations, anorexia Severe: alcoholic hallucinosis Seizures (generalized tonic-clonic) occur in up to 25% of withdrawal episodes, usually within 24 hours after alcohol cessation Delirium tremens (characterized by hallucinations, disorientation, tachycardia, hypertension, low-grade fever, agitation, and diaphoresis) occurs in up to 5% of patients undergoing withdrawal, may be delayed 4 to 5 days, and has mortality rates reaching 15% | Sleep disruption; anxiety; depressive symptoms; irritability; increased breathing rate, body temperature, blood pressure, and pulse |
| GI: gastrointestinal | ||
| Source: Click here for a bibliography | ||
Table 2
How alcohol affects GABA and glutamate neurotransmitters
| GABA | Glutamate | |
|---|---|---|
| GABA, the brain’s primary inhibitory neurotransmitter, renders nerve cells less sensitive to further signaling | Glutamate, the brain’s major excitatory neurotransmitter, renders nerve cells more sensitive to further signaling | |
| Alcohol facilitates the inhibitory function of the GABAA receptor, allowing more GABA to traverse the receptor, and leading to alcohol’s intoxicating effects | Alcohol seems to inhibit the excitatory function of the NMDA glutamate receptor, believed to play a role in memory, learning, and generation of seizures | |
| During alcohol withdrawal, brain GABA concentrations fall below normal and GABAA receptor sensitivity may be reduced | Long-term alcohol exposure produces an adaptive increase in the function of NMDA receptors and results in development of glutamate-NMDA supersensitivity | |
| In the absence of alcohol, the resulting decrease in inhibitory function may contribute to symptoms of CNS hyperactivity associated with acute and protracted alcohol withdrawal | Acute alcohol withdrawal activates glutamate systems, leading to autonomic nervous system hyperactivity; alcohol withdrawal seizures are associated with increased NMDA receptor function | |
| GABA: gamma-aminobutyric acid; NMDA: N-methyl-D-aspartate | ||
| Source: Click here for a bibliography | ||
Table 3
Mechanisms of action of benzodiazepines vs 3 anticonvulsants
| Agent | Mechanism of action |
|---|---|
| Benzodiazepines | Activate GABAA chloride ionophore, increasing affinity of GABAA receptor for GABA and augmenting frequency of chloride channel openinga |
| Valproic acid | GABA modulation and possibly second messenger systems; may inhibit Na1+ and/or Ca2+ channel, thereby boosting GABA and glutamate actionb |
| Levetiracetam | Decreases high voltage activated Ca2+ channels; unique binding site (synaptic vesicle protein SV2A) is thought to be involved in calcium-dependent regulation of neurotransmitter vesicle exocytosisc |
| Gabapentin | GABA analog; unique binding site (Ca2+ channel subunit in brain) decreases calcium influx and inhibits release of excitatory amino acids and monoaminesd |
| GABA: gamma-aminobutyric acid | |
| Source: Click here for a bibliography | |
CASE REPORT 1: Valproic acid for alcohol overdose
After attempting suicide with an alcohol overdose, Ms. J, age 45, is transferred from the emergency room (ER) to our psychiatry consult service 10 hours after admission. Her symptoms include nausea, tremor, headaches, agitation, disorientation, and auditory hallucinations.
Medical history reveals 25 years of alcohol dependence, multiple hospitalizations for withdrawal, and many failed attempts to quit. Ms. J reports consuming an average of 16 drink equivalents (eg, 12 oz beers) daily but denies illicit drug use.
Lab values on admission include blood alcohol concentration (BAC) 290 mg/dL (0.29%), mean corpuscular volume (MCV) 96 fL, gamma-glutamyltransferase (GGT) 164 U/L, aspartate aminotransferase (AST) 43 U/L, alanine aminotransferase (ALT) 31 U/L, and alkaline phosphatase (ALP) 151 U/L. Urine drug screen, acetaminophen, salicylate, vitamin B1 (thiamine), B12 (cyanocobalamin), B9 (folate), and electrolytes (including magnesium) are normal.
We assess alcohol withdrawal severity using the CIWA-Ar (Click here to view/download a copy of this scale). Ms. J’s initial score is 17, indicating a risk of moderate alcohol withdrawal if untreated.
In the ER, Ms. J is placed on a symptom-triggered benzodiazepine detoxification protocol with lorazepam. We add IV valproic acid, 1,250 mg (based on 20 mg/kg body weight)13 divided into 2 doses over the first 24 hours, then maintain IV valproic acid at 500 mg twice daily (Table 4). Within 12 hours of starting combination therapy, Ms. J scores 7 on the CIWA-Ar—indicating mild withdrawal—with subsequent scores <5. She scores 0 with no residual withdrawal symptoms within 36 hours.
Ms. J requires lorazepam, 7 mg, during the 10 hours before valproic acid is added. She requires only 2 mg lorazepam over the next 3 days and reports no side effects related to IV valproic acid. At discharge, Ms. J begins extended-release oral valproic acid, 1,250 mg (based on 25 mg/kg body weight)13 once daily for 2 weeks, until she can obtain outpatient follow-up.
Table 4
Benzodiazepines and anticonvulsants for alcohol detoxification
| Benzodiazepines | Valproic acid | Levetiracetam | Gabapentin | |
|---|---|---|---|---|
| Loading dose | None | 20 mg/kg of body weight, divided into 2 doses for first 24 hours | 1,500 mg IV once daily | 400 mg PO qid |
| Maintenance dose | Day 1: 2 mg tid Day 2: 2 mg morning, 1 mg afternoon, 2 mg evening Day 3: 1 mg tid Day 4: 1 mg bid Day 5: 1 mg Day 6: none | 500 mg IV bid | Either 500 mg IV tid or 1,000 mg PO bid after 2 to 3 days of treatment | 1,200 mg PO tid |
| Side effects | Impaired consciousness, respiratory depression, hypotension | Dizziness, drowsiness, hair loss/thinning, nausea, tremor, weight gain | Somnolence, asthenia, dizziness, coordination difficulties | Somnolence, dizziness, ataxia, fatigue |
| Drug interactions | ↑ BZ: cimetidine, oral contraceptives, ethanol (acute), disulfiram, isoniazid, propranolol ↓ BZ: rifampin, ethanol (chronic) | ↑ VPA: aspirin, felbamate, fluoxetine, isoniazid ↓ VPA: carbamazepine, lamotrigine, phenobarbital, phenytoin, ritonavir | None | ↓ GBP 20%: antacids |
| BZ: benzodiazepine; GBP: gabapentin; PO: per os (by mouth); VPA: valproic acid | ||||
| Source: Click here for a bibliography | ||||
Less lorazepam needed
Adjunctive anticonvulsants can reduce the amount of lorazepam required during detoxification.14,15 Compared with benzodiazepine monotherapy, the advantages of combination therapy—particularly in outpatient alcohol withdrawal treatment and relapse prevention—include:
- minimal interaction with alcohol (avoiding increased psychomotor deficits, cognitive impairment, and intoxication)15
- lower abuse potential
- possible efficacy in mood stabilization before, during, and after withdrawal (Table 5).16
Given the risk of seizures during AWS, anticonvulsants seem to make empirical sense. One study reported a 1% incidence of withdrawal-related seizures in 545 alcohol-dependent inpatients treated with valproic acid.17 Another case series of 37 patients found no acute sequelae when valproic acid was used for AWS.18
Anticonvulsants such as valproic acid may reduce the frequency and severity of alcohol relapse, whereas benzodiazepines may increase relapse risk.19 During a 6-week trial, patients receiving valproic acid maintenance therapy had greater abstinence rates and improved drinking outcomes compared with detoxification-only groups.9
One disadvantage of valproic acid is potential hepatotoxicity, an important consideration in patients with liver damage. Fortunately, Ms. J’s AST and ALT values remained within normal limits during valproic acid treatment.
Table 5
Pharmacologic profiles of benzodiazepines vs 3 anticonvulsants
| Benzodiazepines | Valproic acid | Levetiracetam | Gabapentin | |
|---|---|---|---|---|
| Metabolism | CYP 2C19: diazepam CYP 3A3/4: alprazolam, clonazepam, diazepam, triazolam Phase II only: lorazepam, temazepam, oxazepam | >95% hepatic, of which <20% occurs via CYP isoenzymes | Not extensively metabolized; renal clearance; not involved with hepatic CYP isoenzymes | Not metabolized; secreted via kidneys as unchanged drug |
| Sedation | Mild to moderate | Mild to moderate | Mild to moderate | Moderate to severe |
| Synergistic effects with alcohol | Yes | No | No | No |
| Paradoxical disinhibition | Yes | No | No | No |
| Risk of addiction in outpatient therapy | Yes | No | No | No |
| CYP: cytochrome P450 | ||||
| Source: Click here for a bibliography | ||||
CASE REPORT 2: Levetiracetam for withdrawal seizures
Mr. H, age 42, presents to the ER after suffering a seizure. His medical history includes hypertension, alcohol dependence, and seizures during alcohol withdrawal. He denies a history of psychiatric illness, and his family history is unknown. He is noncompliant with hypertension treatment, which includes clonidine. Mr. H reports his usual alcohol consumption as a 6-pack of beer nightly during the week and a 12-pack nightly on weekends. He says his last drink was 4 days before admission.
Mr. H scores 19 on the CIWA-Ar, placing him at risk for moderate withdrawal. Head CT shows diffuse atrophy, without evidence of an acute intracranial process. BAC is zero on admission, and urine drug screen is negative. Amylase, lipase, and lactate dehydrogenase (LDH) levels suggest acute pancreatitis. AST is elevated to 131 U/L, ALT is elevated to 42 U/L, but MCV is within normal limits.
The psychiatric service is consulted on day 2 of admission, and we prescribe levetiracetam, 500 mg IV every 8 hours.20 IV lorazepam also is available as needed: 1 mg every 8 hours for the first 2 days, then 1 mg every 12 hours for 2 days, then 1 mg every 24 hours. The patient’s CIWA-Ar score is 9 on days 2 and 3 of admission, followed by scores consistently between 2 and 3 after scheduled levetiracetam administration. Mr. H requires 3 mg of lorazepam the remainder of his hospitalization. He is discharged on day 7 with a CIWA-Ar score of 2, and reports no adverse effects related to levetiracetam. He leaves the hospital with a 2-week prescription for oral levetiracetam, 500 mg tid.
Advantages of levetiracetam
Levetiracetam is FDA-approved for adjunctive treatment of adults with partial-onset seizures.21 Successful AWS treatment with adjunctive levetiracetam has been supported by few but promising studies.10,20 Potential advantages of levetiracetam in detoxification include:
- a lack of GABAergic properties, which limits the risk of intoxication or respiratory insufficiency when combined with alcohol21
- low drug-drug interaction risk because of nonhepatic metabolism and primary renal excretion.22,23
We selected levetiracetam for Mr. H because of his history of alcohol withdrawal seizures and acute pancreatitis. Anticonvulsants may be more effective than lorazepam in reducing the risk of alcohol withdrawal seizures,24 and we felt valproic acid might not be safe for him because of its low but real risk of pancreatitis.13 We based our levetiracetam dosing on a small open-label trial20 and product information for treating adults with partial-onset seizures.25
Studies also demonstrate levetiracetam’s potential for relapse prevention during outpatient therapy. In a 10-week trial, levetiracetam decreased the number of standard drinks in alcohol-dependent patients from 5.3 to 1.7 per day.10 This was a small open trial, however, and large controlled trials support the usefulness of other, FDA-approved medications—including disulfiram, naltrexone, and acamprosate—for alcohol relapse prevention.
CASE REPORT 3: Gabapentin for acute withdrawal
Mr. B, age 38, presents to the ER after a 13-day alcohol binge. He has been drinking increasing amounts of alcohol over 6 weeks. Three months earlier, Mr. B was admitted for alcohol withdrawal treatment and received 49 mg of lorazepam over 3 days. This resulted in his transfer from the step-down unit to the intensive care unit for increased agitation, possibly caused by paradoxical disinhibition from excessive lorazepam use.26
Mr. B’s medical history is significant for alcohol-induced seizures, DTs, traumatic brain injury related to craniotomy, and right arm amputation. Mr. B drinks approximately 24 beers per day. He denies tobacco use but admits to past use of cocaine, marijuana, and heroin.
On admission, Mr. B’s BAC is 360 mg/dL (0.36%), AST is elevated at 72 U/L, ALT at 42 U/L, and LDH significantly elevated at 384 U/L. Urine drug screen is negative, and his CIWA-Ar score is 23. His score of –1 on the Richmond Agitation and Sedation Scale (RASS)27 correlates with very mild sedation.
Guided by Bonnet et al28 and clinical experience, we start Mr. B on gabapentin, 1,200 mg tid, and IV lorazepam, 2 mg every 8 hours as needed for breakthrough withdrawal. We decrease lorazepam by 50% every other day until Mr. B is discharged. On days 2, 3, and 4, Mr. B’s CIWA-Ar scores are 6, 9, and 2, respectively. His RASS score drops from –1 on days 1 and 2 to 0 until discharge, indicating an alert and calm state.
Mr. B requires a total of 2 mg of lorazepam throughout hospitalization. He finishes alcohol detoxification on day 4 and is discharged with a prescription for gabapentin, 1,200 mg tid. Two weeks later, when he is admitted to a 28-day inpatient alcohol rehabilitation unit, Mr. B has not relapsed.
More abstinent days
Gabapentin is FDA-approved as adjunctive therapy for partial seizures. Off-label, it has been generally efficacious as an adjunct in alcohol detoxification.29-32 We chose adjunctive anticonvulsant therapy for Mr. B because of his history of alcohol-induced seizures. We chose gabapentin instead of valproic acid because of Mr. B’s liver damage and gabapentin’s lack of hepatic metabolism.
Gabapentin may reduce alcohol consumption and craving in alcohol-dependent patients. By increasing the number of abstinent days, gabapentin may help patients maintain abstinence.33 Gabapentin does not appear to interact clinically with alcohol, causing neither sedation nor synergistic effects.34 Its relative lack of abuse potential may be valuable in outpatient alcohol withdrawal treatment and in maintaining alcohol abstinence after detoxification.
Related resource
- Asplund CA, Aaronson JW, Aaronson HE. 3 regimens for alcohol withdrawal and detoxification. J Fam Pract. 2004;53(7):545-554. www.jfponline.com/Pages.asp?AID=1730.
Drug brand names
- Acamprosate • Campral
- Alprazolam • Xanax
- Carbamazepine • Carbatrol
- Cimetidine • Tagamet
- Clonazepam • Klonopin
- Clonidine • Catapres
- Diazepam • Valium
- Disulfiram • Antabuse
- Felbamate • Felbatol
- Fluoxetine • Prozac
- Gabapentin • Neurontin
- Isoniazid • Nydrazid
- Lamotrigine • Lamictal
- Levetiracetam • Keppra
- Lorazepam • Ativan
- Naltrexone • ReVia, Vivitrol
- Oxazepam • Serax
- Phenobarbital • Luminal
- Phenytoin • Dilantin
- Propranolol • Inderal
- Rifampin • Rifadin
- Ritonavir • Norvir
- Temazepam • Restoril
- Triazolam • Halcion
- Valproic acid • Depakote, Depakene
Disclosures
Dr. Spiegel is a speaker for Pfizer Inc. and GlaxoSmithKline.
Dr. Radac reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The authors thank Rishi Laroia, MD, Robert Swanson, MD, and Adam W. Coe, MD for their contributions to this article.
Benzodiazepines are the mainstay of alcohol detoxification treatment, with extensive evidence supporting their efficacy and relative safety.1 The risk of benzodiazepine-alcohol interaction, however, and psychomotor and cognitive impairments associated with benzodiazepine use may limit early rehabilitation efforts in hospitalized patients.2 Cross-tolerance with alcohol also limits benzodiazepines’ potential benefit in outpatients with substance use disorders.
Adding anticonvulsants to acute benzodiazepine therapy has been shown to decrease alcohol withdrawal symptom severity, reduce seizure risk, and support recovery, particularly in patients with multiple alcohol withdrawal episodes. After detoxification, long-term anticonvulsant use may reduce relapse risk by decreasing post-cessation craving, without abuse liability.3
Although not all studies endorse adding anticonvulsants to benzodiazepines for managing alcohol withdrawal syndrome (AWS),4 we present 3 cases in which anticonvulsants were used successfully as adjuncts to lorazepam. Valproic acid, levetiracetam, and gabapentin offer advantages in acute and long-term therapy of alcohol dependence with efficacy in AWS, low abuse potential, benign safety profile, and mood-stabilizing properties.
Neurobiologic rationale
AWS manifests as a cluster of clinical symptoms including delirium tremens (DTs) and seizures (Table 1). Its pathophysiology can be explained by alcohol’s agonist effect on the gamma-aminobutyric acid (GABA) system and antagonist effect on the glutamatergic system (Table 2).5
Chronic alcohol intake leads to neuroadaptation in the brain in the form of down-regulation of GABAA receptors and upregulation of N-methyl-D-aspartate receptors. During alcohol withdrawal, this neuroadaptation leads to a decrease in central GABA activity and an increase in glutamate activity, resulting in hyperexcitation, anxiety, and seizures.6
Little data exist regarding time to relapse after detoxification in alcohol-dependent patients. One theory—called “protracted withdrawal syndrome” (Table 1)—suggests that abstinent alcoholics return to drinking because of the same, but attenuated, neuroadaptations that trigger acute AWS.7
Advantages of adjunct therapy. Ntais et al8 evaluated benzodiazepines’ effectiveness and safety in treating AWS in a clinical review of 57 randomized, controlled trials totaling 4,051 patients. Benzodiazepines showed similar success rates as other drugs (relative risk [RR] 1.00) or anticonvulsants in particular (RR 0.88), as measured by changes in Clinical Institute Withdrawal Assessment for Alcohol (CIWA-Ar) scores at the end of treatment. Benzodiazepines also offered significant benefit for seizure control compared with nonanticonvulsants (RR 0.23), but less when compared with anti convulsants (RR 1.99).
Although the literature does not support anticonvulsant use for monotherapy in AWS, anticonvulsants show potential as adjunctive therapy. Valproic acid, levetiracetam, and gabapentin offer unique mechanisms of action (Table 3) and demonstrate advantages over benzodiazepine monotherapy for AWS. Adjunctive use of valproic acid,8,9 levetiracetam,10 and gabapentin11,12 in detoxification also has demonstrated efficacy in reducing risk of relapse and delaying relapse.
The neurobiologic rationale for using anticonvulsants in acute AWS is speculative, but these agents appear to:
- inhibit “kindling” (neuronal changes that may be associated with repeated intoxications)
- facilitate GABAergic mechanisms.9
Table 1
Alcohol withdrawal: Acute vs long-term symptoms
| Alcohol withdrawal syndrome | Protracted withdrawal syndrome | |
|---|---|---|
| Description | Cluster of symptoms in alcohol-dependent persons after heavy or prolonged alcohol use has lessened or ceased | Constellation of symptoms lasting weeks to months after alcohol use ends |
| Presentation | Develops during acute detoxification period and lasts 5 to 7 days | Develops after 5- to 7-day acute detoxification period and may persist for 1 year |
| Symptoms | Mild: insomnia, tremor, anxiety, GI upset, headache, diaphoresis, palpitations, anorexia Severe: alcoholic hallucinosis Seizures (generalized tonic-clonic) occur in up to 25% of withdrawal episodes, usually within 24 hours after alcohol cessation Delirium tremens (characterized by hallucinations, disorientation, tachycardia, hypertension, low-grade fever, agitation, and diaphoresis) occurs in up to 5% of patients undergoing withdrawal, may be delayed 4 to 5 days, and has mortality rates reaching 15% | Sleep disruption; anxiety; depressive symptoms; irritability; increased breathing rate, body temperature, blood pressure, and pulse |
| GI: gastrointestinal | ||
| Source: Click here for a bibliography | ||
Table 2
How alcohol affects GABA and glutamate neurotransmitters
| GABA | Glutamate | |
|---|---|---|
| GABA, the brain’s primary inhibitory neurotransmitter, renders nerve cells less sensitive to further signaling | Glutamate, the brain’s major excitatory neurotransmitter, renders nerve cells more sensitive to further signaling | |
| Alcohol facilitates the inhibitory function of the GABAA receptor, allowing more GABA to traverse the receptor, and leading to alcohol’s intoxicating effects | Alcohol seems to inhibit the excitatory function of the NMDA glutamate receptor, believed to play a role in memory, learning, and generation of seizures | |
| During alcohol withdrawal, brain GABA concentrations fall below normal and GABAA receptor sensitivity may be reduced | Long-term alcohol exposure produces an adaptive increase in the function of NMDA receptors and results in development of glutamate-NMDA supersensitivity | |
| In the absence of alcohol, the resulting decrease in inhibitory function may contribute to symptoms of CNS hyperactivity associated with acute and protracted alcohol withdrawal | Acute alcohol withdrawal activates glutamate systems, leading to autonomic nervous system hyperactivity; alcohol withdrawal seizures are associated with increased NMDA receptor function | |
| GABA: gamma-aminobutyric acid; NMDA: N-methyl-D-aspartate | ||
| Source: Click here for a bibliography | ||
Table 3
Mechanisms of action of benzodiazepines vs 3 anticonvulsants
| Agent | Mechanism of action |
|---|---|
| Benzodiazepines | Activate GABAA chloride ionophore, increasing affinity of GABAA receptor for GABA and augmenting frequency of chloride channel openinga |
| Valproic acid | GABA modulation and possibly second messenger systems; may inhibit Na1+ and/or Ca2+ channel, thereby boosting GABA and glutamate actionb |
| Levetiracetam | Decreases high voltage activated Ca2+ channels; unique binding site (synaptic vesicle protein SV2A) is thought to be involved in calcium-dependent regulation of neurotransmitter vesicle exocytosisc |
| Gabapentin | GABA analog; unique binding site (Ca2+ channel subunit in brain) decreases calcium influx and inhibits release of excitatory amino acids and monoaminesd |
| GABA: gamma-aminobutyric acid | |
| Source: Click here for a bibliography | |
CASE REPORT 1: Valproic acid for alcohol overdose
After attempting suicide with an alcohol overdose, Ms. J, age 45, is transferred from the emergency room (ER) to our psychiatry consult service 10 hours after admission. Her symptoms include nausea, tremor, headaches, agitation, disorientation, and auditory hallucinations.
Medical history reveals 25 years of alcohol dependence, multiple hospitalizations for withdrawal, and many failed attempts to quit. Ms. J reports consuming an average of 16 drink equivalents (eg, 12 oz beers) daily but denies illicit drug use.
Lab values on admission include blood alcohol concentration (BAC) 290 mg/dL (0.29%), mean corpuscular volume (MCV) 96 fL, gamma-glutamyltransferase (GGT) 164 U/L, aspartate aminotransferase (AST) 43 U/L, alanine aminotransferase (ALT) 31 U/L, and alkaline phosphatase (ALP) 151 U/L. Urine drug screen, acetaminophen, salicylate, vitamin B1 (thiamine), B12 (cyanocobalamin), B9 (folate), and electrolytes (including magnesium) are normal.
We assess alcohol withdrawal severity using the CIWA-Ar (Click here to view/download a copy of this scale). Ms. J’s initial score is 17, indicating a risk of moderate alcohol withdrawal if untreated.
In the ER, Ms. J is placed on a symptom-triggered benzodiazepine detoxification protocol with lorazepam. We add IV valproic acid, 1,250 mg (based on 20 mg/kg body weight)13 divided into 2 doses over the first 24 hours, then maintain IV valproic acid at 500 mg twice daily (Table 4). Within 12 hours of starting combination therapy, Ms. J scores 7 on the CIWA-Ar—indicating mild withdrawal—with subsequent scores <5. She scores 0 with no residual withdrawal symptoms within 36 hours.
Ms. J requires lorazepam, 7 mg, during the 10 hours before valproic acid is added. She requires only 2 mg lorazepam over the next 3 days and reports no side effects related to IV valproic acid. At discharge, Ms. J begins extended-release oral valproic acid, 1,250 mg (based on 25 mg/kg body weight)13 once daily for 2 weeks, until she can obtain outpatient follow-up.
Table 4
Benzodiazepines and anticonvulsants for alcohol detoxification
| Benzodiazepines | Valproic acid | Levetiracetam | Gabapentin | |
|---|---|---|---|---|
| Loading dose | None | 20 mg/kg of body weight, divided into 2 doses for first 24 hours | 1,500 mg IV once daily | 400 mg PO qid |
| Maintenance dose | Day 1: 2 mg tid Day 2: 2 mg morning, 1 mg afternoon, 2 mg evening Day 3: 1 mg tid Day 4: 1 mg bid Day 5: 1 mg Day 6: none | 500 mg IV bid | Either 500 mg IV tid or 1,000 mg PO bid after 2 to 3 days of treatment | 1,200 mg PO tid |
| Side effects | Impaired consciousness, respiratory depression, hypotension | Dizziness, drowsiness, hair loss/thinning, nausea, tremor, weight gain | Somnolence, asthenia, dizziness, coordination difficulties | Somnolence, dizziness, ataxia, fatigue |
| Drug interactions | ↑ BZ: cimetidine, oral contraceptives, ethanol (acute), disulfiram, isoniazid, propranolol ↓ BZ: rifampin, ethanol (chronic) | ↑ VPA: aspirin, felbamate, fluoxetine, isoniazid ↓ VPA: carbamazepine, lamotrigine, phenobarbital, phenytoin, ritonavir | None | ↓ GBP 20%: antacids |
| BZ: benzodiazepine; GBP: gabapentin; PO: per os (by mouth); VPA: valproic acid | ||||
| Source: Click here for a bibliography | ||||
Less lorazepam needed
Adjunctive anticonvulsants can reduce the amount of lorazepam required during detoxification.14,15 Compared with benzodiazepine monotherapy, the advantages of combination therapy—particularly in outpatient alcohol withdrawal treatment and relapse prevention—include:
- minimal interaction with alcohol (avoiding increased psychomotor deficits, cognitive impairment, and intoxication)15
- lower abuse potential
- possible efficacy in mood stabilization before, during, and after withdrawal (Table 5).16
Given the risk of seizures during AWS, anticonvulsants seem to make empirical sense. One study reported a 1% incidence of withdrawal-related seizures in 545 alcohol-dependent inpatients treated with valproic acid.17 Another case series of 37 patients found no acute sequelae when valproic acid was used for AWS.18
Anticonvulsants such as valproic acid may reduce the frequency and severity of alcohol relapse, whereas benzodiazepines may increase relapse risk.19 During a 6-week trial, patients receiving valproic acid maintenance therapy had greater abstinence rates and improved drinking outcomes compared with detoxification-only groups.9
One disadvantage of valproic acid is potential hepatotoxicity, an important consideration in patients with liver damage. Fortunately, Ms. J’s AST and ALT values remained within normal limits during valproic acid treatment.
Table 5
Pharmacologic profiles of benzodiazepines vs 3 anticonvulsants
| Benzodiazepines | Valproic acid | Levetiracetam | Gabapentin | |
|---|---|---|---|---|
| Metabolism | CYP 2C19: diazepam CYP 3A3/4: alprazolam, clonazepam, diazepam, triazolam Phase II only: lorazepam, temazepam, oxazepam | >95% hepatic, of which <20% occurs via CYP isoenzymes | Not extensively metabolized; renal clearance; not involved with hepatic CYP isoenzymes | Not metabolized; secreted via kidneys as unchanged drug |
| Sedation | Mild to moderate | Mild to moderate | Mild to moderate | Moderate to severe |
| Synergistic effects with alcohol | Yes | No | No | No |
| Paradoxical disinhibition | Yes | No | No | No |
| Risk of addiction in outpatient therapy | Yes | No | No | No |
| CYP: cytochrome P450 | ||||
| Source: Click here for a bibliography | ||||
CASE REPORT 2: Levetiracetam for withdrawal seizures
Mr. H, age 42, presents to the ER after suffering a seizure. His medical history includes hypertension, alcohol dependence, and seizures during alcohol withdrawal. He denies a history of psychiatric illness, and his family history is unknown. He is noncompliant with hypertension treatment, which includes clonidine. Mr. H reports his usual alcohol consumption as a 6-pack of beer nightly during the week and a 12-pack nightly on weekends. He says his last drink was 4 days before admission.
Mr. H scores 19 on the CIWA-Ar, placing him at risk for moderate withdrawal. Head CT shows diffuse atrophy, without evidence of an acute intracranial process. BAC is zero on admission, and urine drug screen is negative. Amylase, lipase, and lactate dehydrogenase (LDH) levels suggest acute pancreatitis. AST is elevated to 131 U/L, ALT is elevated to 42 U/L, but MCV is within normal limits.
The psychiatric service is consulted on day 2 of admission, and we prescribe levetiracetam, 500 mg IV every 8 hours.20 IV lorazepam also is available as needed: 1 mg every 8 hours for the first 2 days, then 1 mg every 12 hours for 2 days, then 1 mg every 24 hours. The patient’s CIWA-Ar score is 9 on days 2 and 3 of admission, followed by scores consistently between 2 and 3 after scheduled levetiracetam administration. Mr. H requires 3 mg of lorazepam the remainder of his hospitalization. He is discharged on day 7 with a CIWA-Ar score of 2, and reports no adverse effects related to levetiracetam. He leaves the hospital with a 2-week prescription for oral levetiracetam, 500 mg tid.
Advantages of levetiracetam
Levetiracetam is FDA-approved for adjunctive treatment of adults with partial-onset seizures.21 Successful AWS treatment with adjunctive levetiracetam has been supported by few but promising studies.10,20 Potential advantages of levetiracetam in detoxification include:
- a lack of GABAergic properties, which limits the risk of intoxication or respiratory insufficiency when combined with alcohol21
- low drug-drug interaction risk because of nonhepatic metabolism and primary renal excretion.22,23
We selected levetiracetam for Mr. H because of his history of alcohol withdrawal seizures and acute pancreatitis. Anticonvulsants may be more effective than lorazepam in reducing the risk of alcohol withdrawal seizures,24 and we felt valproic acid might not be safe for him because of its low but real risk of pancreatitis.13 We based our levetiracetam dosing on a small open-label trial20 and product information for treating adults with partial-onset seizures.25
Studies also demonstrate levetiracetam’s potential for relapse prevention during outpatient therapy. In a 10-week trial, levetiracetam decreased the number of standard drinks in alcohol-dependent patients from 5.3 to 1.7 per day.10 This was a small open trial, however, and large controlled trials support the usefulness of other, FDA-approved medications—including disulfiram, naltrexone, and acamprosate—for alcohol relapse prevention.
CASE REPORT 3: Gabapentin for acute withdrawal
Mr. B, age 38, presents to the ER after a 13-day alcohol binge. He has been drinking increasing amounts of alcohol over 6 weeks. Three months earlier, Mr. B was admitted for alcohol withdrawal treatment and received 49 mg of lorazepam over 3 days. This resulted in his transfer from the step-down unit to the intensive care unit for increased agitation, possibly caused by paradoxical disinhibition from excessive lorazepam use.26
Mr. B’s medical history is significant for alcohol-induced seizures, DTs, traumatic brain injury related to craniotomy, and right arm amputation. Mr. B drinks approximately 24 beers per day. He denies tobacco use but admits to past use of cocaine, marijuana, and heroin.
On admission, Mr. B’s BAC is 360 mg/dL (0.36%), AST is elevated at 72 U/L, ALT at 42 U/L, and LDH significantly elevated at 384 U/L. Urine drug screen is negative, and his CIWA-Ar score is 23. His score of –1 on the Richmond Agitation and Sedation Scale (RASS)27 correlates with very mild sedation.
Guided by Bonnet et al28 and clinical experience, we start Mr. B on gabapentin, 1,200 mg tid, and IV lorazepam, 2 mg every 8 hours as needed for breakthrough withdrawal. We decrease lorazepam by 50% every other day until Mr. B is discharged. On days 2, 3, and 4, Mr. B’s CIWA-Ar scores are 6, 9, and 2, respectively. His RASS score drops from –1 on days 1 and 2 to 0 until discharge, indicating an alert and calm state.
Mr. B requires a total of 2 mg of lorazepam throughout hospitalization. He finishes alcohol detoxification on day 4 and is discharged with a prescription for gabapentin, 1,200 mg tid. Two weeks later, when he is admitted to a 28-day inpatient alcohol rehabilitation unit, Mr. B has not relapsed.
More abstinent days
Gabapentin is FDA-approved as adjunctive therapy for partial seizures. Off-label, it has been generally efficacious as an adjunct in alcohol detoxification.29-32 We chose adjunctive anticonvulsant therapy for Mr. B because of his history of alcohol-induced seizures. We chose gabapentin instead of valproic acid because of Mr. B’s liver damage and gabapentin’s lack of hepatic metabolism.
Gabapentin may reduce alcohol consumption and craving in alcohol-dependent patients. By increasing the number of abstinent days, gabapentin may help patients maintain abstinence.33 Gabapentin does not appear to interact clinically with alcohol, causing neither sedation nor synergistic effects.34 Its relative lack of abuse potential may be valuable in outpatient alcohol withdrawal treatment and in maintaining alcohol abstinence after detoxification.
Related resource
- Asplund CA, Aaronson JW, Aaronson HE. 3 regimens for alcohol withdrawal and detoxification. J Fam Pract. 2004;53(7):545-554. www.jfponline.com/Pages.asp?AID=1730.
Drug brand names
- Acamprosate • Campral
- Alprazolam • Xanax
- Carbamazepine • Carbatrol
- Cimetidine • Tagamet
- Clonazepam • Klonopin
- Clonidine • Catapres
- Diazepam • Valium
- Disulfiram • Antabuse
- Felbamate • Felbatol
- Fluoxetine • Prozac
- Gabapentin • Neurontin
- Isoniazid • Nydrazid
- Lamotrigine • Lamictal
- Levetiracetam • Keppra
- Lorazepam • Ativan
- Naltrexone • ReVia, Vivitrol
- Oxazepam • Serax
- Phenobarbital • Luminal
- Phenytoin • Dilantin
- Propranolol • Inderal
- Rifampin • Rifadin
- Ritonavir • Norvir
- Temazepam • Restoril
- Triazolam • Halcion
- Valproic acid • Depakote, Depakene
Disclosures
Dr. Spiegel is a speaker for Pfizer Inc. and GlaxoSmithKline.
Dr. Radac reports no financial relationship with any company whose products are mentioned in this article or with manufacturers of competing products.
Acknowledgment
The authors thank Rishi Laroia, MD, Robert Swanson, MD, and Adam W. Coe, MD for their contributions to this article.
1. Mayo-Smith MF, Cushman P, Hill AJ, et al. Pharmacological management of alcohol withdrawal. JAMA. 1997;278:144-151.
2. Myrick H, Brady KT, Malcom R. Divalproex in the treatment of alcohol withdrawal—statistical data included. Am J Drug Alcohol Abuse. 2000;26(1):155-160.
3. Johnson BA, Swift RM, Addolorato G, et al. Safety and efficacy of GABAergic medications for treating alcoholism. Alcohol Clin Exp Res. 2005;29(2):248-254.
4. Lum E, Gorman SK, Slavik RS. Valproic acid management of acute alcohol withdrawal. Ann Pharmacother. 2006;40(3):441-448.
5. Trevisan LA, Boutros N, Petrakis IL, et al. Complications of alcohol withdrawal: pathophysiological insights. Alcohol Health Res World. 1998;22(1):61-66.
6. Esel E. Neurobiology of alcohol withdrawal inhibitory and excitatory neurotransmitters. Turk Psikiyatri Derg. 2006;7(2):129-138.
7. Myrick H, Brady KT. The use of divalproex in the treatment of addictive disorders. Psychopharmacol Bull. 2003;37(suppl 2):89-97.
8. Brady KT, Myrick H, Henderson S, et al. The use of divalproex in alcohol relapse prevention: a pilot study. Drug Alcohol Depend. 2002;67(3):323-330.
9. Longo L, Campbell T, Hubatch S. Divalproex sodium (Depakote) for alcohol withdrawal and relapse prevention. J Addict Dis. 2002;21(2):55-64.
10. Sarid-Segal O, Piechniczek-Buczek J, Knapp C, et al. The effects of levetiracetam on alcohol consumption in alcohol-dependent subjects: an open label study. Am J Drug Alcohol Abuse. 2008;34(4):441-447.
11. American Psychiatric Association. Treatments of psychiatric disorders: a task force report of the American Psychiatric Association. Washington, DC: American Psychiatric Association Press; 1989:187.
12. Mason BJ, Light JM, Williams LD, et al. Proof-of-concept human laboratory study for protracted abstinence in alcohol dependence: effects of gabapentin. Addict Biol. 2009;14(1):73-83.
13. Physicians’ Desk Reference 2009. 63rd ed. Montvale, NJ: Physicians’ Desk Reference; 2008:423-431.
14. Malcolm R, Ballenger JC, Sturgis ET, et al. Double-blind controlled trial comparing carbamazepine to oxazepam treatment of alcohol withdrawal. Am J Psychiatry. 1989;146:617-621.
15. Myrick H, Anton R, Voronin K, et al. A double-blind evaluation of gabapentin on alcohol effects and drinking in a clinical laboratory paradigm. Alcohol Clin Exp Res. 2007;31(2):221-227.
16. Malcolm R, Myrick H, Brady KT, et al. Update on anticonvulsants for the treatment of alcohol withdrawal. Am J Addict. 2001;10(suppl):16-23.
17. Davis LL, Ryan W, Adinoff B, et al. Comprehensive review of the psychiatric uses of valproate. J Clin Psychopharm. 2000;20(1 suppl 1):1S-17S.
18. Rosenthal RN, Perkel C, Singh P, et al. A pilot open randomized trial of valproate and phenobarbital in the treatment of acute alcohol withdrawal. Am J Addict. 1998;7:189-197.
19. Book SW, Myrick H. Novel anticonvulsants in the treatment of alcoholism. Expert Opin Investig Drugs. 2005;14(4):371-376.
20. Krebs M, Leopold K, Richter C, et al. Levetiracetam for the treatment of alcohol withdrawal syndrome: an open-label pilot trial. J Clin Psychopharmacol. 2006;26(3):347-349.
21. LaRoche SM, Helmers SL. The new antiepileptic drugs: scientific review. JAMA. 2004;291:605-614.
22. Chabolla DR, Harnois DM, Meschia JF. Levetiracetam monotherapy for liver transplant patients with seizures. Transplant Proc. 2003;35:1480-1481.
23. Paul F, Meencke HJ. Levetiracetam in focal epilepsy and hepatic porphyria: a case report. Epilepsia. 2004;45:559-560.
24. Ntais C, Pakos E, Kyzas P, et al. Benzodiazepines for alcohol withdrawal. Cochrane Database Syst Rev. 2005;(3):CD005063.-
25. Physicians’ Desk Reference 2009. 63rd ed. Montvale, NJ: Physicians’ Desk Reference; 2008:3131-3143.
26. Saias T, Gallarda T. Paradoxical aggressive reactions to benzodiazepine use: a review. Encephale. 2008;34(4):330-336.
27. Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation–Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166:1338-1344.
28. Bonnet U, Banger M, Leweke FM, et al. Treatment of acute alcohol withdrawal with gabapentin: results from a controlled two-center trial. J Clin Psychopharmacol. 2003;23(5):514-519.
29. Myrick H, Malcolm R, Brady KT. Gabapentin treatment of alcohol withdrawal. Am J Psychiatry. 1998;155:1626.-
30. Bonnet U, Banger M, Leweke FM, et al. Treatment of alcohol withdrawal syndrome with gabapentin. Pharmacopsychiatry. 1999;32:107-109.
31. Mariani JJ, Rosenthal RN, Tross S, et al. A randomized, open-label, controlled trial of gabapentin and phenobarbital in the treatment of alcohol withdrawal. Am J Addict. 2006;15(1):76-84.
32. Voris J, Smith NL, Rao SM, et al. Gabapentin for the treatment of ethanol withdrawal. Subst Abus. 2003;24(2):129-132.
33. Furieri FA, Nakamura-Palacios EM. Gabapentin reduces alcohol consumption and craving: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2007;68(11):1691-1700.
34. Bisaga A, Evans SM. The acute effects of gabapentin in combination with alcohol in heavy drinkers. Drug Alcohol Depend. 2006;83(1):25-32.
1. Mayo-Smith MF, Cushman P, Hill AJ, et al. Pharmacological management of alcohol withdrawal. JAMA. 1997;278:144-151.
2. Myrick H, Brady KT, Malcom R. Divalproex in the treatment of alcohol withdrawal—statistical data included. Am J Drug Alcohol Abuse. 2000;26(1):155-160.
3. Johnson BA, Swift RM, Addolorato G, et al. Safety and efficacy of GABAergic medications for treating alcoholism. Alcohol Clin Exp Res. 2005;29(2):248-254.
4. Lum E, Gorman SK, Slavik RS. Valproic acid management of acute alcohol withdrawal. Ann Pharmacother. 2006;40(3):441-448.
5. Trevisan LA, Boutros N, Petrakis IL, et al. Complications of alcohol withdrawal: pathophysiological insights. Alcohol Health Res World. 1998;22(1):61-66.
6. Esel E. Neurobiology of alcohol withdrawal inhibitory and excitatory neurotransmitters. Turk Psikiyatri Derg. 2006;7(2):129-138.
7. Myrick H, Brady KT. The use of divalproex in the treatment of addictive disorders. Psychopharmacol Bull. 2003;37(suppl 2):89-97.
8. Brady KT, Myrick H, Henderson S, et al. The use of divalproex in alcohol relapse prevention: a pilot study. Drug Alcohol Depend. 2002;67(3):323-330.
9. Longo L, Campbell T, Hubatch S. Divalproex sodium (Depakote) for alcohol withdrawal and relapse prevention. J Addict Dis. 2002;21(2):55-64.
10. Sarid-Segal O, Piechniczek-Buczek J, Knapp C, et al. The effects of levetiracetam on alcohol consumption in alcohol-dependent subjects: an open label study. Am J Drug Alcohol Abuse. 2008;34(4):441-447.
11. American Psychiatric Association. Treatments of psychiatric disorders: a task force report of the American Psychiatric Association. Washington, DC: American Psychiatric Association Press; 1989:187.
12. Mason BJ, Light JM, Williams LD, et al. Proof-of-concept human laboratory study for protracted abstinence in alcohol dependence: effects of gabapentin. Addict Biol. 2009;14(1):73-83.
13. Physicians’ Desk Reference 2009. 63rd ed. Montvale, NJ: Physicians’ Desk Reference; 2008:423-431.
14. Malcolm R, Ballenger JC, Sturgis ET, et al. Double-blind controlled trial comparing carbamazepine to oxazepam treatment of alcohol withdrawal. Am J Psychiatry. 1989;146:617-621.
15. Myrick H, Anton R, Voronin K, et al. A double-blind evaluation of gabapentin on alcohol effects and drinking in a clinical laboratory paradigm. Alcohol Clin Exp Res. 2007;31(2):221-227.
16. Malcolm R, Myrick H, Brady KT, et al. Update on anticonvulsants for the treatment of alcohol withdrawal. Am J Addict. 2001;10(suppl):16-23.
17. Davis LL, Ryan W, Adinoff B, et al. Comprehensive review of the psychiatric uses of valproate. J Clin Psychopharm. 2000;20(1 suppl 1):1S-17S.
18. Rosenthal RN, Perkel C, Singh P, et al. A pilot open randomized trial of valproate and phenobarbital in the treatment of acute alcohol withdrawal. Am J Addict. 1998;7:189-197.
19. Book SW, Myrick H. Novel anticonvulsants in the treatment of alcoholism. Expert Opin Investig Drugs. 2005;14(4):371-376.
20. Krebs M, Leopold K, Richter C, et al. Levetiracetam for the treatment of alcohol withdrawal syndrome: an open-label pilot trial. J Clin Psychopharmacol. 2006;26(3):347-349.
21. LaRoche SM, Helmers SL. The new antiepileptic drugs: scientific review. JAMA. 2004;291:605-614.
22. Chabolla DR, Harnois DM, Meschia JF. Levetiracetam monotherapy for liver transplant patients with seizures. Transplant Proc. 2003;35:1480-1481.
23. Paul F, Meencke HJ. Levetiracetam in focal epilepsy and hepatic porphyria: a case report. Epilepsia. 2004;45:559-560.
24. Ntais C, Pakos E, Kyzas P, et al. Benzodiazepines for alcohol withdrawal. Cochrane Database Syst Rev. 2005;(3):CD005063.-
25. Physicians’ Desk Reference 2009. 63rd ed. Montvale, NJ: Physicians’ Desk Reference; 2008:3131-3143.
26. Saias T, Gallarda T. Paradoxical aggressive reactions to benzodiazepine use: a review. Encephale. 2008;34(4):330-336.
27. Sessler CN, Gosnell MS, Grap MJ, et al. The Richmond Agitation–Sedation Scale: validity and reliability in adult intensive care unit patients. Am J Respir Crit Care Med. 2002;166:1338-1344.
28. Bonnet U, Banger M, Leweke FM, et al. Treatment of acute alcohol withdrawal with gabapentin: results from a controlled two-center trial. J Clin Psychopharmacol. 2003;23(5):514-519.
29. Myrick H, Malcolm R, Brady KT. Gabapentin treatment of alcohol withdrawal. Am J Psychiatry. 1998;155:1626.-
30. Bonnet U, Banger M, Leweke FM, et al. Treatment of alcohol withdrawal syndrome with gabapentin. Pharmacopsychiatry. 1999;32:107-109.
31. Mariani JJ, Rosenthal RN, Tross S, et al. A randomized, open-label, controlled trial of gabapentin and phenobarbital in the treatment of alcohol withdrawal. Am J Addict. 2006;15(1):76-84.
32. Voris J, Smith NL, Rao SM, et al. Gabapentin for the treatment of ethanol withdrawal. Subst Abus. 2003;24(2):129-132.
33. Furieri FA, Nakamura-Palacios EM. Gabapentin reduces alcohol consumption and craving: a randomized, double-blind, placebo-controlled trial. J Clin Psychiatry. 2007;68(11):1691-1700.
34. Bisaga A, Evans SM. The acute effects of gabapentin in combination with alcohol in heavy drinkers. Drug Alcohol Depend. 2006;83(1):25-32.
‘I’m sober, Doctor, really’: Best biomarkers for underreported alcohol use
Hospitalized patients who are not truthful about their alcohol consumption may be at risk for an unplanned withdrawal. Self-reports of alcohol use—such as CAGE and the Alcohol Use Disorders Identification Test (AUDIT)—are valid, inexpensive, and noninvasive, but patients easily can feign results.1 Biochemical measures are more objective, and combinations of markers are an effective tool to detect recent heavy drinking in the 10% to 25% of patients who underreport alcohol use.2
Biochemical measures can detect acute alcohol intoxication and recent prolonged drinking. Because marker levels return to normal after long-term abstinence, ongoing monitoring can help detect a relapse before a patient admits to it.3
This article presents 3 cases in which biochemical markers helped prevent alcohol withdrawal in patients who denied alcohol abuse. We discuss why we ordered biochemical tests and which combinations provided highly sensitive results.
CASE 1: Depression and substance abuse
Ms. C, age 39, presents with bleeding gums due to excessive warfarin, which she takes prophylactically for a history of deep vein thrombosis. She is seen by the psychiatric consultation service for depression—which she says she has experienced since “the day I was born”—and substance abuse that includes a history binge drinking. Ms. C says she has stopped drinking and remained abstinent for the past year because she is fearful of further damaging her kidneys. She also denies psychosis. She does not have a history or symptoms of hepatobiliary or hematologic disease.
Challenge. Despite Ms. C’s self-reported 1 year of sobriety, her history of binge drinking and depression calls for evaluating her alcohol withdrawal risk. Laboratory markers of alcohol abuse are the only means to assess her recent drinking behavior.
Discussion. Lab results include serum albumin of 3.4 g/dL, total bilirubin of 0.3 mg/dL, total protein of 6.3 g/dL, aspartate aminotransferase (AST) of 13 U/L, alanine aminotransferase (ALT) of 19 U/L, alkaline phosphatase of 136 U/L, and blood ammonia level of 37 μg/dL. Gamma-glutamyl transferase (GGT) is elevated at 104 U/L (normal range for women: 0 to 45 U/L). Mean corpuscular volume (MCV) is elevated at 101 fL (normal range 80 to 100 fL).
The combination of elevated MCV and GGT has a 95% sensitivity for alcohol abuse.4 GGT levels become elevated after 24 hours to 2 weeks of heavy alcohol consumption and return to normal within 2 to 6 weeks of abstinence, which allows them to detect binge drinking. MCV takes 6 to 8 weeks of heavy drinking—we which we define as consuming ≥40 grams of alcohol/day5—to become elevated and returns to normal within 3 months of abstinence.
These data provide evidence that Ms. C recently consumed substantial amounts of alcohol. As a result, we start her on alcohol withdrawal precautions (AWP).
Markers of alcohol abuse
Biochemical markers commonly used to detect alcohol abuse (Table 1) include:
- blood alcohol level (BAL)
- MCV
- liver function tests (LFTs) such as ALT, AST, and GGT
- carbohydrate deficient transferrin (CDT).
Table 1
By the numbers: Biomarkers of excessive alcohol consumption
| Biomarker | |||||
|---|---|---|---|---|---|
| CDT | GGT | AST | ALT | MCV | |
| Blood test normal range | Women: 0 to 45 U/L Men: 0 to 53 U/L | 10 to 34 U/L | 8 to 37 U/L | 80 to 100 fL | |
| Blood test abnormal range | >1.3% of total transferrin concentration | Women: >45 U/L Men: >53 U/L | Levels rarely exceed 500 U/L | Levels rarely exceed 300 U/L | >100 fL |
| Time to elevation | 2 to 3 weeks | 24 hours to 2 weeks | 3 to 7 days | 3 to 7 days | After 6 weeks |
| Time to descent to normal levels | 2 to 4 weeks of abstinence | 2 to 6 weeks of abstinence | Half-life 12 to 24 hours | Half-life 37 to 57 hours | 3 months |
| Dose-response of alcohol | 60 g/d | 80 to 200 g/d | ≥40 g/d | ≥40 g/d | ≥40 g/d |
| Sensitivity | 55% to 90%a-e | 37% to 85%b, f, g | AST:ALT ratio >2:1 has a 70% sensitivity and 92% to 100% specificity for alcoholic-induced liver diseaseh-j | 20% to 70%b,k | |
| Relapse sensitivity | 55% to 76%a,l,m | 50%a,e | 20%a,n | ||
| Specificity | 92% to 97%a,b | 18% to 93%a,b,e | 64% to 66%b,k,n | ||
| Positive predictive value | 46% to 75%c,g | 41%g | 36%g | ||
| Negative predictive value | 72% to 98%a,c,g | 69% to 92%a,e,g | 67%g | ||
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | |||||
| Source: Reference Citations: click here | |||||
BAL can document acute alcohol intoxication, but its use is limited because alcohol has a 4-hour half-life and an elimination rate of 7 grams/hour—equivalent to 1 drink/hour.6 (A “drink” typically is defined as a 12-ounce bottle of beer or wine cooler, a 5-ounce glass of wine, or 1.5 ounces of 80-proof distilled spirits.) Therefore, BAL will identify as false negatives alcohol-dependent patients who abstain from alcohol within 24 hours of testing.
MCV is an index of the average volume of erythrocytes. Macrocytosis occurs when the volume exceeds 100 fL. Elevated MCV is the most typical morphologic abnormality associated with excessive alcohol consumption7,8 and macrocytosis—sometimes without associated anemia—is often evident in persons with alcoholism. MCV elevates after 6 weeks of alcohol misuse and may remain elevated for up to 3 months after a person has stopped drinking.9
Because patients with disorders unrelated to alcohol use can have elevated MCV, alone it is not a useful screening marker for alcohol abuse.10 Additionally, because macrocytosis can persist under strictly controlled alcohol abstinence, MCV is not a reliable clinical indicator of relapse.11
LFTs measure enzymes and proteins. ALT, AST, and GGT are the most relevant for detecting heavy drinking. An AST:ALT ratio >2:1 supports a suspicion of alcohol abuse.12 More than 90% of patients with an AST:ALT ratio of 2:1 have alcoholic liver disease. This increases to more than 96% if the ratio is 3:1.13
GGT is an enzyme concentrated in the liver, bile ducts, and kidneys; normal range is 0 to 45 U/L (for females) or 53 U/L (for males).14 GGT levels >30 U/L correlate with alcohol consumption of >4 drinks per day.15 GGT has a half-life of 14 to 26 days and remains elevated for 4 to 6 weeks after drinking cessation, which make it useful for monitoring abstinence in treatment programs.16 Sensitivity ranges from 37% to 85% and specificity is as high as 93% in nonmedical populations.17 Although nonalcoholic liver disease can elevate GGT in persons who do not abuse alcohol, 50% to 72% of GGT elevations can be explained by excessive alcohol consumption.18
CDT is a newer biomarker used to monitor alcohol consumption. The most accurate way to express CDT level is as a percentage of total transferrin concentration. This method accounts for individual variations in transferrin levels, thus minimizing false positives.18 In persons who consume >4 or 5 drinks per day for 2 weeks or more, CDT is >1.3% of total transferrin.19 Unfortunately, because it is expensive and requires sophisticated test methodology, CDT testing is not available at most hospitals.20
Combinations improve detection
Each biochemical measure has strengths and weaknesses as a marker for determining patients’ alcohol consumption (Table 2). CDT and GGT show the highest sensitivity for heavy drinking, and CDT has a higher specificity than GGT (Table 3).21,22 Relapse to alcohol use after abstinence may be best identified by a simultaneous 30% increase in CDT and GGT.5
Because GGT has a longer half-life than CDT, its diagnostic efficiency in detecting alcohol relapse may not develop until 4 weeks after alcohol detoxification, whereas CDT may become clinically useful for detecting relapse as early as 1 week after detoxification.23
Table 2
Biomarkers of alcohol use: Strengths and weaknesses
| Biomarker | Strengths | Weaknesses |
|---|---|---|
| CDT | High specificity for alcohol use; few factors cause false positives High sensitivity in distinguishing alcoholics from social drinkers Marker of relapse and abstinence from drinking Confirmatory test for patients suspected of alcohol abuse | Low sensitivity; more valuable to confirm than exclude heavy drinking Cost (average $30/assay) and low availability of testing Likely less sensitive for women and younger patients compared with men Poor screening tool for alcohol use in general population |
| GGT | Elevations precede alcohol-induced liver damage High specificity in patients with suspected alcohol abuse Effective marker for patients suspected of binge drinking Inexpensive ( | Can be falsely elevated by liver and biliary disease, smoking, obesity, and medications that induce microsomal enzymes Low sensitivity makes it a poor screening tool in general population Poor marker of relapse |
| AST:ALT >2:1 | Highly sensitive and specific for alcohol-induced liver damage | Enzyme elevations can be detected only after periods of heavy drinking Elevations secondary to liver damage at the hepatocellular level (after fatty changes) |
| MCV | Accuracy similar in male and female patients Elevations in suspected cases of alcohol use indicate chronicity of drinking Routine laboratory test | Poor biomarker for relapse False positives caused by liver disease, hemolysis, bleeding disorders, anemia, folate deficiency, and medications that reduce folate Low sensitivity and specificity for alcohol use make it a poor screening tool for alcohol abuse |
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | ||
Table 3
Interpreting diagnostic test performance
| Term | Definition | Applicability |
|---|---|---|
| Sensitivity | Percent of persons with disease who test positive | High value is desirable for ruling out disease (low false-negative rate) |
| Specificity | Percent of persons without disease who test negative | High value is desirable for ruling in disease (low false-positive rate) |
| Positive predictive value | Percent of positive test results that are true positives | Probability that a person with a positive test result has the disease |
| Negative predictive value | Percent of negative test results that are true negatives | Probability that a person with a negative test result is disease-free |
| Source: References 21,22 | ||
There is evidence that combining tests can improve alcohol use detection.24 For example, Dolman et al25 found that the ability of the AUDIT questionnaire to correctly predict which patients would experience alcohol withdrawal increases when it is used in combination with biochemical markers. Specifically, the positive predictive value of an AUDIT score ≥8 increased from 17% to 47% when found in combination with ≥2 abnormal biochemical marker levels; the study looked at GGT, ALT, AST, and MCV. Sensitivity was 94% and specificity was 98%.
Similarly, combinations of biochemical markers—especially CDT and GGT—have improved detection of alcohol use and subsequent risk of withdrawal.26Table 4 provides a summary of studies that evaluated using combinations of biochemical markers.4,5,27-31
Table 4
Combining biomarker tests: An effective approach
| Combination | Study | Sensitivity* |
|---|---|---|
| GGT + MCV | Morgan et al4 | 95% |
| GGT + CDT | Hietala et al5 | 90% |
| Mundle et al29 | 90% | |
| Bell et al30 | 90% | |
| Sillanaukee et al31 | 95% | |
| GGT + AST:ALT >2:1 | Gluud et al27 | 92% |
| Morgan et al4 | 100% | |
| MCV + AST:ALT >2:1 | Kawachi et al28 | 97% |
| Morgan et al4 | 95% | |
| GGT + MCV + AST:ALT >2:1 | Morgan et al4 | 100% |
| GGT + MCV + CDT | Sillanaukee et al31 | 70% |
| * Sensitivity for detecting excessive alcohol consumption | ||
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | ||
Consider patients’ comorbidities
Patients at risk for underreporting alcohol use include those with unemployment histories, previous alcohol treatment, and higher scores on the Alcohol Dependence Scale (18.5, SD=8.1).2 Interpret biochemical testing results in the context of a patient’s overall clinical picture.
The following 2 case patients denied or underreported recent alcohol use but we determined they were at high risk for an alcohol disorder because of their medical and/or psychiatric histories. Analysis of biochemical markers helped assess the risk of alcohol withdrawal.
CASE 2: Altered mental status
Family members bring Mr. N, age 44, to the hospital because of his odd behavior. He presents with paranoid delusions and an inappropriate elated mood. His medical history includes acquired immune deficiency syndrome (AIDS). After cerebrospinal fluid analysis, computed tomography of the head, electroencephalogram, and metabolic workup are within normal limits, the patient is diagnosed with human immunodeficiency virus (HIV) mania and is admitted.
On admission, Mr. N denies alcohol use. A blood alcohol/urine toxicity screen is negative. One day after admission, Mr. M develops elevated blood pressure and tachycardia and reports headache and nausea.
Challenge. Gathering a valid history of Mr. N’s alcohol use is difficult because of his acutely altered mental status and manic-like state. We use laboratory data to assess his risk of alcohol withdrawal. His liver function tests include an AST of 33 U/L, ALT of 30 U/L, and an alkaline phosphatase of 94 U/L. MCV is normal at 90 fL. Interestingly, the GGT level is elevated almost 4 times normal at 164 U/L.
Discussion. Although Mr. N denied alcohol use and presented with a negative BAL, laboratory data support alcohol dependence. His GGT was elevated well beyond normal limits, without evidence of hepatobiliary disease. GGT has a sensitivity as high as 85%32 and limited specificity for alcohol abuse. Because of his high probability of recent alcohol consumption, we place Mr. N on AWP.
We postulate that our patient’s autonomic instability, headache, and nausea are related to alcohol withdrawal. We are aware that delirium occurs frequently in patients with HIV infection, and although Mr. N’s medical workup is negative, HIV infection can produce an acute encephalopathy that could resemble our patient’s clinical picture.33
Mr. N’s autonomic instability, headache, and nausea abated after treatment for alcohol withdrawal.
CASE 3: Suicide attempt?
Mr. S, age 28, presents to the trauma service with a self-inflicted gunshot wound to the face. He reports feeling depressed for the last year but denies a history of psychotic symptoms or heroin withdrawal symptoms. He also denies recent or past alcohol abuse and does not have a history of biliary tract disease or megaloblastic anemia. His mother tells us Mr. S has had a history of depression since childhood.
Challenge. Based on Mr. S’ apparent suicide attempt and history, we feel he is at high risk for alcohol abuse. We use laboratory markers to assess the likelihood of alcohol consumption and possibly decrease his risk of alcohol withdrawal.
Discussion. Mr. S’ lab data show an MCV of 91 fL, AST of 95 U/L, alanine ALT of 156 U/L, and alkaline phosphatase of 160 U/L. GGT was elevated at 122 U/L.
Although Mr. S’ MCV is within the normal range, his GGT is elevated, and the combination of an elevated GGT and MCV has a 95% sensitivity for the diagnosis of alcohol abuse. We place Mr. S on alcohol withdrawal precautions and discuss with him the potential life-threatening complications of alcohol withdrawal. Confronted with this information and the possible implication of his elevated LFTs, the patient admits his alcohol history—which consists of drinking 12 beers/day for at least the past 2 years. He admits this despite exhibiting no signs or symptoms of alcohol withdrawal.
Related Resources
- National Institute on Alcohol Abuse and Alcoholism Data/Statistical Tables. www.niaaa.nih.gov/Resources/DatabaseResources/QuickFacts.
- Maisto SA, Connors GJ, Allen JP. Contrasting self-report screens for alcohol problems: a review. Alcohol Clin Exp Res 1995;19(6):1510-6.
- Coulton S, Drummond C, James D, et al. Opportunistic screening for alcohol use disorders in primary care: comparative study. BMJ 2006;332:511-7.
Bottom line
Because CDT—the most accurate biomarker—is not available at most hospitals, we recommend using combinations of other measures to detect unreported recent alcohol consumption. If GGT and MCV are elevated, GGT is elevated and AST:ALT is >2:1, or MCV is elevated and AST:ALT is >2:1, consider initiating alcohol withdrawal precautions.
Acknowledgement
The authors acknowledge Daiana Radac, BA, a third-year medical student at Eastern Virginia Medical School, for her contributions to this article.
1. Allen JP, Anthenelli RM. Getting to the bottom of problem drinking: the case for routine screening. Current Psychiatry 2003;2(6):26-44.
2. Killeen TK, Brady KT, Gold PB, et al. Comparison of self-report versus agency records of service utilization in a community sample of individuals with alcohol use disorders. Drug Alcohol Depend 2004;73(2):141-7.
3. Alcohol withdrawal syndrome: how to predict, prevent, diagnose and treat it. Prescrire Int 2007;16(87):24-31.
4. Morgan MY, Colman JC, Sherlock S. The use of a combination of peripheral markers for diagnosing alcoholism and monitoring for continued abuse. Alcohol Alcohol 1981;16:167-77.
5. Hietala J, Koivisto H, Anttila P, et al. Comparison of the combined marker GGT-CDT and the conventional laboratory markers of alcohol abuse in heavy drinkers, moderate drinkers and abstainers. Alcohol Alcohol 2006;41(5):528-33.
6. Swift R. Direct measurement of alcohol and its metabolites. Addiction 2003;98:73-80.
7. Koivisto H, Hietala J, Anttila P, et al. Long-term ethanol consumption and macrocytosis: diagnostic and pathogenic implications. J Lab Clin Med 2005;147(4):191-6.
8. Savage DG, Ogundipe A, Allen RH, et al. Etiology and diagnostic evaluation of macrocytosis. Am J Med Sci 2000;319(6):343-52.
9. Gordon H. Detection of alcoholic liver disease. World J Gastroenterol 2001;7(3):297-302.
10. Bernadt M, Mumford J, Taylor C, et al. Comparison of questionnaire and laboratory tests in the detection of excessive drinking and alcoholism. Lancet 1982;1:325-8.
11. Hasselblatt M, Martin F, Maul O, et al. Persistent macrocytosis following abstinence from chronic alcohol use. JAMA 2001;286:2946.-
12. Prati D, Taioli E, Zanella A, et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med 2002;137:1-9.
13. Fancher T, Kamboj A, Onate J. Interpreting liver function tests. Current Psychiatry 2007;6(5):61-8.
14. Puukka K, Hietala J, Koivisto H, et al. Obesity and the clinical use of serum GGT activity as a marker of heavy drinking. Scand J Clin Lab Invest 2007;67(5):480-8.
15. Litten RZ, Allen JP, Fertig JB. y-glutamyl transpeptidase and carbohydrate deficient transferrin: alternative measures of excessive alcohol consumption. Alcohol Clin Exp Res 1995;19(6):1541-6.
16. National Institute on Alcohol Abuse and Alcoholism. Screening for alcohol problems—an update. Alcohol Alert No 56 Available at: http://pubs.niaaa.nih.gov/publications/aa56.htm. Accessed May 5, 2007.
17. DiMartini A. A clinical guide to assessing alcohol use and problems. Available at: http://www.alcoholmedicalscholars.org/clin-asmt.ppt. Accessed June 30, 2008.
18. Wolff K, Marshall E. Biological markers of alcohol use. Psychiatry 2006;5(12):437-8.
19. ARUP Laboratories. Carbohydrate-deficient transferrin (CDT) for alcohol use. 2006. Available at: http://www.aruplab.com/TestDirectory/resources/TechnicalBulletins/Carbohydrate-Deficient%20Transferrin%20(CDT)%20Mar%202006.pdf. Accessed July 30, 2008.
20. Allen JP, Litten RZ. The role of laboratory testing in alcoholism treatment. J Subst Abuse Treat 2001;20:81-5.
21. Bhushan V, Le T, Ozturk A, et al. Behavioral Science. In: Le T, Bhushan V, Rao DA, eds. First aid for the USMLE step 1: a student to student guide. New York, NY: McGraw Hill Medical Publishing Division; 2007.
22. Miller PM, Anton RF. Biochemical alcohol screening in primary care. Addict Behav 2004;29(7):1427-37.
23. Schmidt LG, Schmidt K, Dufeu P, et al. Superiority of carbohydrate-deficient transferrin to gamma-glutamyltransferase in detecting relapse in alcoholism. Am J Psychiatry 1997;154(1):75-80.
24. Salaspuro M. Carbohydrate-deficient transferrin as compared to other markers of alcoholism: a systematic review. Alcohol 1999;19(3):261-71.
25. Dolman JM, Hawkes ND. Combining the AUDIT questionnaire and biochemical markers to assess alcohol use and risk of alcohol withdrawal in medical patients. Alcohol Alcohol 2005;40(6):515-9.
26. Helander A, Carlsson AV, Borg S. Longitudinal comparison of carbohydrate-deficient transferrin and gamma-glutamyl transferase: complementary markers of excessive alcohol consumption. Alcohol Alcohol 1996;31(1):101-7.
27. Gluud C, Andersen I, Dietrichson O, et al. Gamma-glutamyltransferase, aspartate aminotransferase and alkaline phosphatase as markers of alcohol consumption in out-patient alcoholics. Eur J Clin Invest 1981;11(3):171-6.
28. Kawachi I, Robinson GM, Stace NH. A combination of raised serum AST: ALT ratio and erythrocyte mean cell volume level detects excessive alcohol consumption. N Z Med J 1990;103(887):145-8.
29. Mundle G, Ackerman K, Mann K. Biological markers as indicators for relapse in alcohol-dependent patients. Addict Biol 1999;4(2):209-14.
30. Bell H, Tallaksen C, Sjåheim T, et al. Serum carbohydrate-deficient transferrin as a marker of alcohol consumption in patients with chronic liver diseases. Alcohol Clin Exp Res 1993;17(2):246-52.
31. Sillanaukee P, Aalto M, Seppa K. Carbohydrate-deficient transferrin and conventional alcohol markers as indicators for brief intervention among heavy drinkers in primary health care. Alcohol Clin Exp Res 1998;22(4):892-6.
32. Salaspuro S. Conventional and coming laboratory markers of alcoholism and heavy drinking. Alcohol Clin Exp Res 1986;10(6 suppl):5-12.
33. Della Penna ND, Treisman GJ. HIV/AIDS. In: Levenson J, ed. Essentials of psychosomatic medicine. Washington, DC: American Psychiatric Publishing, Inc; 2007.
Hospitalized patients who are not truthful about their alcohol consumption may be at risk for an unplanned withdrawal. Self-reports of alcohol use—such as CAGE and the Alcohol Use Disorders Identification Test (AUDIT)—are valid, inexpensive, and noninvasive, but patients easily can feign results.1 Biochemical measures are more objective, and combinations of markers are an effective tool to detect recent heavy drinking in the 10% to 25% of patients who underreport alcohol use.2
Biochemical measures can detect acute alcohol intoxication and recent prolonged drinking. Because marker levels return to normal after long-term abstinence, ongoing monitoring can help detect a relapse before a patient admits to it.3
This article presents 3 cases in which biochemical markers helped prevent alcohol withdrawal in patients who denied alcohol abuse. We discuss why we ordered biochemical tests and which combinations provided highly sensitive results.
CASE 1: Depression and substance abuse
Ms. C, age 39, presents with bleeding gums due to excessive warfarin, which she takes prophylactically for a history of deep vein thrombosis. She is seen by the psychiatric consultation service for depression—which she says she has experienced since “the day I was born”—and substance abuse that includes a history binge drinking. Ms. C says she has stopped drinking and remained abstinent for the past year because she is fearful of further damaging her kidneys. She also denies psychosis. She does not have a history or symptoms of hepatobiliary or hematologic disease.
Challenge. Despite Ms. C’s self-reported 1 year of sobriety, her history of binge drinking and depression calls for evaluating her alcohol withdrawal risk. Laboratory markers of alcohol abuse are the only means to assess her recent drinking behavior.
Discussion. Lab results include serum albumin of 3.4 g/dL, total bilirubin of 0.3 mg/dL, total protein of 6.3 g/dL, aspartate aminotransferase (AST) of 13 U/L, alanine aminotransferase (ALT) of 19 U/L, alkaline phosphatase of 136 U/L, and blood ammonia level of 37 μg/dL. Gamma-glutamyl transferase (GGT) is elevated at 104 U/L (normal range for women: 0 to 45 U/L). Mean corpuscular volume (MCV) is elevated at 101 fL (normal range 80 to 100 fL).
The combination of elevated MCV and GGT has a 95% sensitivity for alcohol abuse.4 GGT levels become elevated after 24 hours to 2 weeks of heavy alcohol consumption and return to normal within 2 to 6 weeks of abstinence, which allows them to detect binge drinking. MCV takes 6 to 8 weeks of heavy drinking—we which we define as consuming ≥40 grams of alcohol/day5—to become elevated and returns to normal within 3 months of abstinence.
These data provide evidence that Ms. C recently consumed substantial amounts of alcohol. As a result, we start her on alcohol withdrawal precautions (AWP).
Markers of alcohol abuse
Biochemical markers commonly used to detect alcohol abuse (Table 1) include:
- blood alcohol level (BAL)
- MCV
- liver function tests (LFTs) such as ALT, AST, and GGT
- carbohydrate deficient transferrin (CDT).
Table 1
By the numbers: Biomarkers of excessive alcohol consumption
| Biomarker | |||||
|---|---|---|---|---|---|
| CDT | GGT | AST | ALT | MCV | |
| Blood test normal range | Women: 0 to 45 U/L Men: 0 to 53 U/L | 10 to 34 U/L | 8 to 37 U/L | 80 to 100 fL | |
| Blood test abnormal range | >1.3% of total transferrin concentration | Women: >45 U/L Men: >53 U/L | Levels rarely exceed 500 U/L | Levels rarely exceed 300 U/L | >100 fL |
| Time to elevation | 2 to 3 weeks | 24 hours to 2 weeks | 3 to 7 days | 3 to 7 days | After 6 weeks |
| Time to descent to normal levels | 2 to 4 weeks of abstinence | 2 to 6 weeks of abstinence | Half-life 12 to 24 hours | Half-life 37 to 57 hours | 3 months |
| Dose-response of alcohol | 60 g/d | 80 to 200 g/d | ≥40 g/d | ≥40 g/d | ≥40 g/d |
| Sensitivity | 55% to 90%a-e | 37% to 85%b, f, g | AST:ALT ratio >2:1 has a 70% sensitivity and 92% to 100% specificity for alcoholic-induced liver diseaseh-j | 20% to 70%b,k | |
| Relapse sensitivity | 55% to 76%a,l,m | 50%a,e | 20%a,n | ||
| Specificity | 92% to 97%a,b | 18% to 93%a,b,e | 64% to 66%b,k,n | ||
| Positive predictive value | 46% to 75%c,g | 41%g | 36%g | ||
| Negative predictive value | 72% to 98%a,c,g | 69% to 92%a,e,g | 67%g | ||
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | |||||
| Source: Reference Citations: click here | |||||
BAL can document acute alcohol intoxication, but its use is limited because alcohol has a 4-hour half-life and an elimination rate of 7 grams/hour—equivalent to 1 drink/hour.6 (A “drink” typically is defined as a 12-ounce bottle of beer or wine cooler, a 5-ounce glass of wine, or 1.5 ounces of 80-proof distilled spirits.) Therefore, BAL will identify as false negatives alcohol-dependent patients who abstain from alcohol within 24 hours of testing.
MCV is an index of the average volume of erythrocytes. Macrocytosis occurs when the volume exceeds 100 fL. Elevated MCV is the most typical morphologic abnormality associated with excessive alcohol consumption7,8 and macrocytosis—sometimes without associated anemia—is often evident in persons with alcoholism. MCV elevates after 6 weeks of alcohol misuse and may remain elevated for up to 3 months after a person has stopped drinking.9
Because patients with disorders unrelated to alcohol use can have elevated MCV, alone it is not a useful screening marker for alcohol abuse.10 Additionally, because macrocytosis can persist under strictly controlled alcohol abstinence, MCV is not a reliable clinical indicator of relapse.11
LFTs measure enzymes and proteins. ALT, AST, and GGT are the most relevant for detecting heavy drinking. An AST:ALT ratio >2:1 supports a suspicion of alcohol abuse.12 More than 90% of patients with an AST:ALT ratio of 2:1 have alcoholic liver disease. This increases to more than 96% if the ratio is 3:1.13
GGT is an enzyme concentrated in the liver, bile ducts, and kidneys; normal range is 0 to 45 U/L (for females) or 53 U/L (for males).14 GGT levels >30 U/L correlate with alcohol consumption of >4 drinks per day.15 GGT has a half-life of 14 to 26 days and remains elevated for 4 to 6 weeks after drinking cessation, which make it useful for monitoring abstinence in treatment programs.16 Sensitivity ranges from 37% to 85% and specificity is as high as 93% in nonmedical populations.17 Although nonalcoholic liver disease can elevate GGT in persons who do not abuse alcohol, 50% to 72% of GGT elevations can be explained by excessive alcohol consumption.18
CDT is a newer biomarker used to monitor alcohol consumption. The most accurate way to express CDT level is as a percentage of total transferrin concentration. This method accounts for individual variations in transferrin levels, thus minimizing false positives.18 In persons who consume >4 or 5 drinks per day for 2 weeks or more, CDT is >1.3% of total transferrin.19 Unfortunately, because it is expensive and requires sophisticated test methodology, CDT testing is not available at most hospitals.20
Combinations improve detection
Each biochemical measure has strengths and weaknesses as a marker for determining patients’ alcohol consumption (Table 2). CDT and GGT show the highest sensitivity for heavy drinking, and CDT has a higher specificity than GGT (Table 3).21,22 Relapse to alcohol use after abstinence may be best identified by a simultaneous 30% increase in CDT and GGT.5
Because GGT has a longer half-life than CDT, its diagnostic efficiency in detecting alcohol relapse may not develop until 4 weeks after alcohol detoxification, whereas CDT may become clinically useful for detecting relapse as early as 1 week after detoxification.23
Table 2
Biomarkers of alcohol use: Strengths and weaknesses
| Biomarker | Strengths | Weaknesses |
|---|---|---|
| CDT | High specificity for alcohol use; few factors cause false positives High sensitivity in distinguishing alcoholics from social drinkers Marker of relapse and abstinence from drinking Confirmatory test for patients suspected of alcohol abuse | Low sensitivity; more valuable to confirm than exclude heavy drinking Cost (average $30/assay) and low availability of testing Likely less sensitive for women and younger patients compared with men Poor screening tool for alcohol use in general population |
| GGT | Elevations precede alcohol-induced liver damage High specificity in patients with suspected alcohol abuse Effective marker for patients suspected of binge drinking Inexpensive ( | Can be falsely elevated by liver and biliary disease, smoking, obesity, and medications that induce microsomal enzymes Low sensitivity makes it a poor screening tool in general population Poor marker of relapse |
| AST:ALT >2:1 | Highly sensitive and specific for alcohol-induced liver damage | Enzyme elevations can be detected only after periods of heavy drinking Elevations secondary to liver damage at the hepatocellular level (after fatty changes) |
| MCV | Accuracy similar in male and female patients Elevations in suspected cases of alcohol use indicate chronicity of drinking Routine laboratory test | Poor biomarker for relapse False positives caused by liver disease, hemolysis, bleeding disorders, anemia, folate deficiency, and medications that reduce folate Low sensitivity and specificity for alcohol use make it a poor screening tool for alcohol abuse |
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | ||
Table 3
Interpreting diagnostic test performance
| Term | Definition | Applicability |
|---|---|---|
| Sensitivity | Percent of persons with disease who test positive | High value is desirable for ruling out disease (low false-negative rate) |
| Specificity | Percent of persons without disease who test negative | High value is desirable for ruling in disease (low false-positive rate) |
| Positive predictive value | Percent of positive test results that are true positives | Probability that a person with a positive test result has the disease |
| Negative predictive value | Percent of negative test results that are true negatives | Probability that a person with a negative test result is disease-free |
| Source: References 21,22 | ||
There is evidence that combining tests can improve alcohol use detection.24 For example, Dolman et al25 found that the ability of the AUDIT questionnaire to correctly predict which patients would experience alcohol withdrawal increases when it is used in combination with biochemical markers. Specifically, the positive predictive value of an AUDIT score ≥8 increased from 17% to 47% when found in combination with ≥2 abnormal biochemical marker levels; the study looked at GGT, ALT, AST, and MCV. Sensitivity was 94% and specificity was 98%.
Similarly, combinations of biochemical markers—especially CDT and GGT—have improved detection of alcohol use and subsequent risk of withdrawal.26Table 4 provides a summary of studies that evaluated using combinations of biochemical markers.4,5,27-31
Table 4
Combining biomarker tests: An effective approach
| Combination | Study | Sensitivity* |
|---|---|---|
| GGT + MCV | Morgan et al4 | 95% |
| GGT + CDT | Hietala et al5 | 90% |
| Mundle et al29 | 90% | |
| Bell et al30 | 90% | |
| Sillanaukee et al31 | 95% | |
| GGT + AST:ALT >2:1 | Gluud et al27 | 92% |
| Morgan et al4 | 100% | |
| MCV + AST:ALT >2:1 | Kawachi et al28 | 97% |
| Morgan et al4 | 95% | |
| GGT + MCV + AST:ALT >2:1 | Morgan et al4 | 100% |
| GGT + MCV + CDT | Sillanaukee et al31 | 70% |
| * Sensitivity for detecting excessive alcohol consumption | ||
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | ||
Consider patients’ comorbidities
Patients at risk for underreporting alcohol use include those with unemployment histories, previous alcohol treatment, and higher scores on the Alcohol Dependence Scale (18.5, SD=8.1).2 Interpret biochemical testing results in the context of a patient’s overall clinical picture.
The following 2 case patients denied or underreported recent alcohol use but we determined they were at high risk for an alcohol disorder because of their medical and/or psychiatric histories. Analysis of biochemical markers helped assess the risk of alcohol withdrawal.
CASE 2: Altered mental status
Family members bring Mr. N, age 44, to the hospital because of his odd behavior. He presents with paranoid delusions and an inappropriate elated mood. His medical history includes acquired immune deficiency syndrome (AIDS). After cerebrospinal fluid analysis, computed tomography of the head, electroencephalogram, and metabolic workup are within normal limits, the patient is diagnosed with human immunodeficiency virus (HIV) mania and is admitted.
On admission, Mr. N denies alcohol use. A blood alcohol/urine toxicity screen is negative. One day after admission, Mr. M develops elevated blood pressure and tachycardia and reports headache and nausea.
Challenge. Gathering a valid history of Mr. N’s alcohol use is difficult because of his acutely altered mental status and manic-like state. We use laboratory data to assess his risk of alcohol withdrawal. His liver function tests include an AST of 33 U/L, ALT of 30 U/L, and an alkaline phosphatase of 94 U/L. MCV is normal at 90 fL. Interestingly, the GGT level is elevated almost 4 times normal at 164 U/L.
Discussion. Although Mr. N denied alcohol use and presented with a negative BAL, laboratory data support alcohol dependence. His GGT was elevated well beyond normal limits, without evidence of hepatobiliary disease. GGT has a sensitivity as high as 85%32 and limited specificity for alcohol abuse. Because of his high probability of recent alcohol consumption, we place Mr. N on AWP.
We postulate that our patient’s autonomic instability, headache, and nausea are related to alcohol withdrawal. We are aware that delirium occurs frequently in patients with HIV infection, and although Mr. N’s medical workup is negative, HIV infection can produce an acute encephalopathy that could resemble our patient’s clinical picture.33
Mr. N’s autonomic instability, headache, and nausea abated after treatment for alcohol withdrawal.
CASE 3: Suicide attempt?
Mr. S, age 28, presents to the trauma service with a self-inflicted gunshot wound to the face. He reports feeling depressed for the last year but denies a history of psychotic symptoms or heroin withdrawal symptoms. He also denies recent or past alcohol abuse and does not have a history of biliary tract disease or megaloblastic anemia. His mother tells us Mr. S has had a history of depression since childhood.
Challenge. Based on Mr. S’ apparent suicide attempt and history, we feel he is at high risk for alcohol abuse. We use laboratory markers to assess the likelihood of alcohol consumption and possibly decrease his risk of alcohol withdrawal.
Discussion. Mr. S’ lab data show an MCV of 91 fL, AST of 95 U/L, alanine ALT of 156 U/L, and alkaline phosphatase of 160 U/L. GGT was elevated at 122 U/L.
Although Mr. S’ MCV is within the normal range, his GGT is elevated, and the combination of an elevated GGT and MCV has a 95% sensitivity for the diagnosis of alcohol abuse. We place Mr. S on alcohol withdrawal precautions and discuss with him the potential life-threatening complications of alcohol withdrawal. Confronted with this information and the possible implication of his elevated LFTs, the patient admits his alcohol history—which consists of drinking 12 beers/day for at least the past 2 years. He admits this despite exhibiting no signs or symptoms of alcohol withdrawal.
Related Resources
- National Institute on Alcohol Abuse and Alcoholism Data/Statistical Tables. www.niaaa.nih.gov/Resources/DatabaseResources/QuickFacts.
- Maisto SA, Connors GJ, Allen JP. Contrasting self-report screens for alcohol problems: a review. Alcohol Clin Exp Res 1995;19(6):1510-6.
- Coulton S, Drummond C, James D, et al. Opportunistic screening for alcohol use disorders in primary care: comparative study. BMJ 2006;332:511-7.
Bottom line
Because CDT—the most accurate biomarker—is not available at most hospitals, we recommend using combinations of other measures to detect unreported recent alcohol consumption. If GGT and MCV are elevated, GGT is elevated and AST:ALT is >2:1, or MCV is elevated and AST:ALT is >2:1, consider initiating alcohol withdrawal precautions.
Acknowledgement
The authors acknowledge Daiana Radac, BA, a third-year medical student at Eastern Virginia Medical School, for her contributions to this article.
Hospitalized patients who are not truthful about their alcohol consumption may be at risk for an unplanned withdrawal. Self-reports of alcohol use—such as CAGE and the Alcohol Use Disorders Identification Test (AUDIT)—are valid, inexpensive, and noninvasive, but patients easily can feign results.1 Biochemical measures are more objective, and combinations of markers are an effective tool to detect recent heavy drinking in the 10% to 25% of patients who underreport alcohol use.2
Biochemical measures can detect acute alcohol intoxication and recent prolonged drinking. Because marker levels return to normal after long-term abstinence, ongoing monitoring can help detect a relapse before a patient admits to it.3
This article presents 3 cases in which biochemical markers helped prevent alcohol withdrawal in patients who denied alcohol abuse. We discuss why we ordered biochemical tests and which combinations provided highly sensitive results.
CASE 1: Depression and substance abuse
Ms. C, age 39, presents with bleeding gums due to excessive warfarin, which she takes prophylactically for a history of deep vein thrombosis. She is seen by the psychiatric consultation service for depression—which she says she has experienced since “the day I was born”—and substance abuse that includes a history binge drinking. Ms. C says she has stopped drinking and remained abstinent for the past year because she is fearful of further damaging her kidneys. She also denies psychosis. She does not have a history or symptoms of hepatobiliary or hematologic disease.
Challenge. Despite Ms. C’s self-reported 1 year of sobriety, her history of binge drinking and depression calls for evaluating her alcohol withdrawal risk. Laboratory markers of alcohol abuse are the only means to assess her recent drinking behavior.
Discussion. Lab results include serum albumin of 3.4 g/dL, total bilirubin of 0.3 mg/dL, total protein of 6.3 g/dL, aspartate aminotransferase (AST) of 13 U/L, alanine aminotransferase (ALT) of 19 U/L, alkaline phosphatase of 136 U/L, and blood ammonia level of 37 μg/dL. Gamma-glutamyl transferase (GGT) is elevated at 104 U/L (normal range for women: 0 to 45 U/L). Mean corpuscular volume (MCV) is elevated at 101 fL (normal range 80 to 100 fL).
The combination of elevated MCV and GGT has a 95% sensitivity for alcohol abuse.4 GGT levels become elevated after 24 hours to 2 weeks of heavy alcohol consumption and return to normal within 2 to 6 weeks of abstinence, which allows them to detect binge drinking. MCV takes 6 to 8 weeks of heavy drinking—we which we define as consuming ≥40 grams of alcohol/day5—to become elevated and returns to normal within 3 months of abstinence.
These data provide evidence that Ms. C recently consumed substantial amounts of alcohol. As a result, we start her on alcohol withdrawal precautions (AWP).
Markers of alcohol abuse
Biochemical markers commonly used to detect alcohol abuse (Table 1) include:
- blood alcohol level (BAL)
- MCV
- liver function tests (LFTs) such as ALT, AST, and GGT
- carbohydrate deficient transferrin (CDT).
Table 1
By the numbers: Biomarkers of excessive alcohol consumption
| Biomarker | |||||
|---|---|---|---|---|---|
| CDT | GGT | AST | ALT | MCV | |
| Blood test normal range | Women: 0 to 45 U/L Men: 0 to 53 U/L | 10 to 34 U/L | 8 to 37 U/L | 80 to 100 fL | |
| Blood test abnormal range | >1.3% of total transferrin concentration | Women: >45 U/L Men: >53 U/L | Levels rarely exceed 500 U/L | Levels rarely exceed 300 U/L | >100 fL |
| Time to elevation | 2 to 3 weeks | 24 hours to 2 weeks | 3 to 7 days | 3 to 7 days | After 6 weeks |
| Time to descent to normal levels | 2 to 4 weeks of abstinence | 2 to 6 weeks of abstinence | Half-life 12 to 24 hours | Half-life 37 to 57 hours | 3 months |
| Dose-response of alcohol | 60 g/d | 80 to 200 g/d | ≥40 g/d | ≥40 g/d | ≥40 g/d |
| Sensitivity | 55% to 90%a-e | 37% to 85%b, f, g | AST:ALT ratio >2:1 has a 70% sensitivity and 92% to 100% specificity for alcoholic-induced liver diseaseh-j | 20% to 70%b,k | |
| Relapse sensitivity | 55% to 76%a,l,m | 50%a,e | 20%a,n | ||
| Specificity | 92% to 97%a,b | 18% to 93%a,b,e | 64% to 66%b,k,n | ||
| Positive predictive value | 46% to 75%c,g | 41%g | 36%g | ||
| Negative predictive value | 72% to 98%a,c,g | 69% to 92%a,e,g | 67%g | ||
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | |||||
| Source: Reference Citations: click here | |||||
BAL can document acute alcohol intoxication, but its use is limited because alcohol has a 4-hour half-life and an elimination rate of 7 grams/hour—equivalent to 1 drink/hour.6 (A “drink” typically is defined as a 12-ounce bottle of beer or wine cooler, a 5-ounce glass of wine, or 1.5 ounces of 80-proof distilled spirits.) Therefore, BAL will identify as false negatives alcohol-dependent patients who abstain from alcohol within 24 hours of testing.
MCV is an index of the average volume of erythrocytes. Macrocytosis occurs when the volume exceeds 100 fL. Elevated MCV is the most typical morphologic abnormality associated with excessive alcohol consumption7,8 and macrocytosis—sometimes without associated anemia—is often evident in persons with alcoholism. MCV elevates after 6 weeks of alcohol misuse and may remain elevated for up to 3 months after a person has stopped drinking.9
Because patients with disorders unrelated to alcohol use can have elevated MCV, alone it is not a useful screening marker for alcohol abuse.10 Additionally, because macrocytosis can persist under strictly controlled alcohol abstinence, MCV is not a reliable clinical indicator of relapse.11
LFTs measure enzymes and proteins. ALT, AST, and GGT are the most relevant for detecting heavy drinking. An AST:ALT ratio >2:1 supports a suspicion of alcohol abuse.12 More than 90% of patients with an AST:ALT ratio of 2:1 have alcoholic liver disease. This increases to more than 96% if the ratio is 3:1.13
GGT is an enzyme concentrated in the liver, bile ducts, and kidneys; normal range is 0 to 45 U/L (for females) or 53 U/L (for males).14 GGT levels >30 U/L correlate with alcohol consumption of >4 drinks per day.15 GGT has a half-life of 14 to 26 days and remains elevated for 4 to 6 weeks after drinking cessation, which make it useful for monitoring abstinence in treatment programs.16 Sensitivity ranges from 37% to 85% and specificity is as high as 93% in nonmedical populations.17 Although nonalcoholic liver disease can elevate GGT in persons who do not abuse alcohol, 50% to 72% of GGT elevations can be explained by excessive alcohol consumption.18
CDT is a newer biomarker used to monitor alcohol consumption. The most accurate way to express CDT level is as a percentage of total transferrin concentration. This method accounts for individual variations in transferrin levels, thus minimizing false positives.18 In persons who consume >4 or 5 drinks per day for 2 weeks or more, CDT is >1.3% of total transferrin.19 Unfortunately, because it is expensive and requires sophisticated test methodology, CDT testing is not available at most hospitals.20
Combinations improve detection
Each biochemical measure has strengths and weaknesses as a marker for determining patients’ alcohol consumption (Table 2). CDT and GGT show the highest sensitivity for heavy drinking, and CDT has a higher specificity than GGT (Table 3).21,22 Relapse to alcohol use after abstinence may be best identified by a simultaneous 30% increase in CDT and GGT.5
Because GGT has a longer half-life than CDT, its diagnostic efficiency in detecting alcohol relapse may not develop until 4 weeks after alcohol detoxification, whereas CDT may become clinically useful for detecting relapse as early as 1 week after detoxification.23
Table 2
Biomarkers of alcohol use: Strengths and weaknesses
| Biomarker | Strengths | Weaknesses |
|---|---|---|
| CDT | High specificity for alcohol use; few factors cause false positives High sensitivity in distinguishing alcoholics from social drinkers Marker of relapse and abstinence from drinking Confirmatory test for patients suspected of alcohol abuse | Low sensitivity; more valuable to confirm than exclude heavy drinking Cost (average $30/assay) and low availability of testing Likely less sensitive for women and younger patients compared with men Poor screening tool for alcohol use in general population |
| GGT | Elevations precede alcohol-induced liver damage High specificity in patients with suspected alcohol abuse Effective marker for patients suspected of binge drinking Inexpensive ( | Can be falsely elevated by liver and biliary disease, smoking, obesity, and medications that induce microsomal enzymes Low sensitivity makes it a poor screening tool in general population Poor marker of relapse |
| AST:ALT >2:1 | Highly sensitive and specific for alcohol-induced liver damage | Enzyme elevations can be detected only after periods of heavy drinking Elevations secondary to liver damage at the hepatocellular level (after fatty changes) |
| MCV | Accuracy similar in male and female patients Elevations in suspected cases of alcohol use indicate chronicity of drinking Routine laboratory test | Poor biomarker for relapse False positives caused by liver disease, hemolysis, bleeding disorders, anemia, folate deficiency, and medications that reduce folate Low sensitivity and specificity for alcohol use make it a poor screening tool for alcohol abuse |
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | ||
Table 3
Interpreting diagnostic test performance
| Term | Definition | Applicability |
|---|---|---|
| Sensitivity | Percent of persons with disease who test positive | High value is desirable for ruling out disease (low false-negative rate) |
| Specificity | Percent of persons without disease who test negative | High value is desirable for ruling in disease (low false-positive rate) |
| Positive predictive value | Percent of positive test results that are true positives | Probability that a person with a positive test result has the disease |
| Negative predictive value | Percent of negative test results that are true negatives | Probability that a person with a negative test result is disease-free |
| Source: References 21,22 | ||
There is evidence that combining tests can improve alcohol use detection.24 For example, Dolman et al25 found that the ability of the AUDIT questionnaire to correctly predict which patients would experience alcohol withdrawal increases when it is used in combination with biochemical markers. Specifically, the positive predictive value of an AUDIT score ≥8 increased from 17% to 47% when found in combination with ≥2 abnormal biochemical marker levels; the study looked at GGT, ALT, AST, and MCV. Sensitivity was 94% and specificity was 98%.
Similarly, combinations of biochemical markers—especially CDT and GGT—have improved detection of alcohol use and subsequent risk of withdrawal.26Table 4 provides a summary of studies that evaluated using combinations of biochemical markers.4,5,27-31
Table 4
Combining biomarker tests: An effective approach
| Combination | Study | Sensitivity* |
|---|---|---|
| GGT + MCV | Morgan et al4 | 95% |
| GGT + CDT | Hietala et al5 | 90% |
| Mundle et al29 | 90% | |
| Bell et al30 | 90% | |
| Sillanaukee et al31 | 95% | |
| GGT + AST:ALT >2:1 | Gluud et al27 | 92% |
| Morgan et al4 | 100% | |
| MCV + AST:ALT >2:1 | Kawachi et al28 | 97% |
| Morgan et al4 | 95% | |
| GGT + MCV + AST:ALT >2:1 | Morgan et al4 | 100% |
| GGT + MCV + CDT | Sillanaukee et al31 | 70% |
| * Sensitivity for detecting excessive alcohol consumption | ||
| AST: aspartate aminotransferase; ALT: alanine aminotransferase; CDT: carbohydrate deficient transferrin; GGT: gamma-glutamyl transferase; MCV: mean corpuscular volume | ||
Consider patients’ comorbidities
Patients at risk for underreporting alcohol use include those with unemployment histories, previous alcohol treatment, and higher scores on the Alcohol Dependence Scale (18.5, SD=8.1).2 Interpret biochemical testing results in the context of a patient’s overall clinical picture.
The following 2 case patients denied or underreported recent alcohol use but we determined they were at high risk for an alcohol disorder because of their medical and/or psychiatric histories. Analysis of biochemical markers helped assess the risk of alcohol withdrawal.
CASE 2: Altered mental status
Family members bring Mr. N, age 44, to the hospital because of his odd behavior. He presents with paranoid delusions and an inappropriate elated mood. His medical history includes acquired immune deficiency syndrome (AIDS). After cerebrospinal fluid analysis, computed tomography of the head, electroencephalogram, and metabolic workup are within normal limits, the patient is diagnosed with human immunodeficiency virus (HIV) mania and is admitted.
On admission, Mr. N denies alcohol use. A blood alcohol/urine toxicity screen is negative. One day after admission, Mr. M develops elevated blood pressure and tachycardia and reports headache and nausea.
Challenge. Gathering a valid history of Mr. N’s alcohol use is difficult because of his acutely altered mental status and manic-like state. We use laboratory data to assess his risk of alcohol withdrawal. His liver function tests include an AST of 33 U/L, ALT of 30 U/L, and an alkaline phosphatase of 94 U/L. MCV is normal at 90 fL. Interestingly, the GGT level is elevated almost 4 times normal at 164 U/L.
Discussion. Although Mr. N denied alcohol use and presented with a negative BAL, laboratory data support alcohol dependence. His GGT was elevated well beyond normal limits, without evidence of hepatobiliary disease. GGT has a sensitivity as high as 85%32 and limited specificity for alcohol abuse. Because of his high probability of recent alcohol consumption, we place Mr. N on AWP.
We postulate that our patient’s autonomic instability, headache, and nausea are related to alcohol withdrawal. We are aware that delirium occurs frequently in patients with HIV infection, and although Mr. N’s medical workup is negative, HIV infection can produce an acute encephalopathy that could resemble our patient’s clinical picture.33
Mr. N’s autonomic instability, headache, and nausea abated after treatment for alcohol withdrawal.
CASE 3: Suicide attempt?
Mr. S, age 28, presents to the trauma service with a self-inflicted gunshot wound to the face. He reports feeling depressed for the last year but denies a history of psychotic symptoms or heroin withdrawal symptoms. He also denies recent or past alcohol abuse and does not have a history of biliary tract disease or megaloblastic anemia. His mother tells us Mr. S has had a history of depression since childhood.
Challenge. Based on Mr. S’ apparent suicide attempt and history, we feel he is at high risk for alcohol abuse. We use laboratory markers to assess the likelihood of alcohol consumption and possibly decrease his risk of alcohol withdrawal.
Discussion. Mr. S’ lab data show an MCV of 91 fL, AST of 95 U/L, alanine ALT of 156 U/L, and alkaline phosphatase of 160 U/L. GGT was elevated at 122 U/L.
Although Mr. S’ MCV is within the normal range, his GGT is elevated, and the combination of an elevated GGT and MCV has a 95% sensitivity for the diagnosis of alcohol abuse. We place Mr. S on alcohol withdrawal precautions and discuss with him the potential life-threatening complications of alcohol withdrawal. Confronted with this information and the possible implication of his elevated LFTs, the patient admits his alcohol history—which consists of drinking 12 beers/day for at least the past 2 years. He admits this despite exhibiting no signs or symptoms of alcohol withdrawal.
Related Resources
- National Institute on Alcohol Abuse and Alcoholism Data/Statistical Tables. www.niaaa.nih.gov/Resources/DatabaseResources/QuickFacts.
- Maisto SA, Connors GJ, Allen JP. Contrasting self-report screens for alcohol problems: a review. Alcohol Clin Exp Res 1995;19(6):1510-6.
- Coulton S, Drummond C, James D, et al. Opportunistic screening for alcohol use disorders in primary care: comparative study. BMJ 2006;332:511-7.
Bottom line
Because CDT—the most accurate biomarker—is not available at most hospitals, we recommend using combinations of other measures to detect unreported recent alcohol consumption. If GGT and MCV are elevated, GGT is elevated and AST:ALT is >2:1, or MCV is elevated and AST:ALT is >2:1, consider initiating alcohol withdrawal precautions.
Acknowledgement
The authors acknowledge Daiana Radac, BA, a third-year medical student at Eastern Virginia Medical School, for her contributions to this article.
1. Allen JP, Anthenelli RM. Getting to the bottom of problem drinking: the case for routine screening. Current Psychiatry 2003;2(6):26-44.
2. Killeen TK, Brady KT, Gold PB, et al. Comparison of self-report versus agency records of service utilization in a community sample of individuals with alcohol use disorders. Drug Alcohol Depend 2004;73(2):141-7.
3. Alcohol withdrawal syndrome: how to predict, prevent, diagnose and treat it. Prescrire Int 2007;16(87):24-31.
4. Morgan MY, Colman JC, Sherlock S. The use of a combination of peripheral markers for diagnosing alcoholism and monitoring for continued abuse. Alcohol Alcohol 1981;16:167-77.
5. Hietala J, Koivisto H, Anttila P, et al. Comparison of the combined marker GGT-CDT and the conventional laboratory markers of alcohol abuse in heavy drinkers, moderate drinkers and abstainers. Alcohol Alcohol 2006;41(5):528-33.
6. Swift R. Direct measurement of alcohol and its metabolites. Addiction 2003;98:73-80.
7. Koivisto H, Hietala J, Anttila P, et al. Long-term ethanol consumption and macrocytosis: diagnostic and pathogenic implications. J Lab Clin Med 2005;147(4):191-6.
8. Savage DG, Ogundipe A, Allen RH, et al. Etiology and diagnostic evaluation of macrocytosis. Am J Med Sci 2000;319(6):343-52.
9. Gordon H. Detection of alcoholic liver disease. World J Gastroenterol 2001;7(3):297-302.
10. Bernadt M, Mumford J, Taylor C, et al. Comparison of questionnaire and laboratory tests in the detection of excessive drinking and alcoholism. Lancet 1982;1:325-8.
11. Hasselblatt M, Martin F, Maul O, et al. Persistent macrocytosis following abstinence from chronic alcohol use. JAMA 2001;286:2946.-
12. Prati D, Taioli E, Zanella A, et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med 2002;137:1-9.
13. Fancher T, Kamboj A, Onate J. Interpreting liver function tests. Current Psychiatry 2007;6(5):61-8.
14. Puukka K, Hietala J, Koivisto H, et al. Obesity and the clinical use of serum GGT activity as a marker of heavy drinking. Scand J Clin Lab Invest 2007;67(5):480-8.
15. Litten RZ, Allen JP, Fertig JB. y-glutamyl transpeptidase and carbohydrate deficient transferrin: alternative measures of excessive alcohol consumption. Alcohol Clin Exp Res 1995;19(6):1541-6.
16. National Institute on Alcohol Abuse and Alcoholism. Screening for alcohol problems—an update. Alcohol Alert No 56 Available at: http://pubs.niaaa.nih.gov/publications/aa56.htm. Accessed May 5, 2007.
17. DiMartini A. A clinical guide to assessing alcohol use and problems. Available at: http://www.alcoholmedicalscholars.org/clin-asmt.ppt. Accessed June 30, 2008.
18. Wolff K, Marshall E. Biological markers of alcohol use. Psychiatry 2006;5(12):437-8.
19. ARUP Laboratories. Carbohydrate-deficient transferrin (CDT) for alcohol use. 2006. Available at: http://www.aruplab.com/TestDirectory/resources/TechnicalBulletins/Carbohydrate-Deficient%20Transferrin%20(CDT)%20Mar%202006.pdf. Accessed July 30, 2008.
20. Allen JP, Litten RZ. The role of laboratory testing in alcoholism treatment. J Subst Abuse Treat 2001;20:81-5.
21. Bhushan V, Le T, Ozturk A, et al. Behavioral Science. In: Le T, Bhushan V, Rao DA, eds. First aid for the USMLE step 1: a student to student guide. New York, NY: McGraw Hill Medical Publishing Division; 2007.
22. Miller PM, Anton RF. Biochemical alcohol screening in primary care. Addict Behav 2004;29(7):1427-37.
23. Schmidt LG, Schmidt K, Dufeu P, et al. Superiority of carbohydrate-deficient transferrin to gamma-glutamyltransferase in detecting relapse in alcoholism. Am J Psychiatry 1997;154(1):75-80.
24. Salaspuro M. Carbohydrate-deficient transferrin as compared to other markers of alcoholism: a systematic review. Alcohol 1999;19(3):261-71.
25. Dolman JM, Hawkes ND. Combining the AUDIT questionnaire and biochemical markers to assess alcohol use and risk of alcohol withdrawal in medical patients. Alcohol Alcohol 2005;40(6):515-9.
26. Helander A, Carlsson AV, Borg S. Longitudinal comparison of carbohydrate-deficient transferrin and gamma-glutamyl transferase: complementary markers of excessive alcohol consumption. Alcohol Alcohol 1996;31(1):101-7.
27. Gluud C, Andersen I, Dietrichson O, et al. Gamma-glutamyltransferase, aspartate aminotransferase and alkaline phosphatase as markers of alcohol consumption in out-patient alcoholics. Eur J Clin Invest 1981;11(3):171-6.
28. Kawachi I, Robinson GM, Stace NH. A combination of raised serum AST: ALT ratio and erythrocyte mean cell volume level detects excessive alcohol consumption. N Z Med J 1990;103(887):145-8.
29. Mundle G, Ackerman K, Mann K. Biological markers as indicators for relapse in alcohol-dependent patients. Addict Biol 1999;4(2):209-14.
30. Bell H, Tallaksen C, Sjåheim T, et al. Serum carbohydrate-deficient transferrin as a marker of alcohol consumption in patients with chronic liver diseases. Alcohol Clin Exp Res 1993;17(2):246-52.
31. Sillanaukee P, Aalto M, Seppa K. Carbohydrate-deficient transferrin and conventional alcohol markers as indicators for brief intervention among heavy drinkers in primary health care. Alcohol Clin Exp Res 1998;22(4):892-6.
32. Salaspuro S. Conventional and coming laboratory markers of alcoholism and heavy drinking. Alcohol Clin Exp Res 1986;10(6 suppl):5-12.
33. Della Penna ND, Treisman GJ. HIV/AIDS. In: Levenson J, ed. Essentials of psychosomatic medicine. Washington, DC: American Psychiatric Publishing, Inc; 2007.
1. Allen JP, Anthenelli RM. Getting to the bottom of problem drinking: the case for routine screening. Current Psychiatry 2003;2(6):26-44.
2. Killeen TK, Brady KT, Gold PB, et al. Comparison of self-report versus agency records of service utilization in a community sample of individuals with alcohol use disorders. Drug Alcohol Depend 2004;73(2):141-7.
3. Alcohol withdrawal syndrome: how to predict, prevent, diagnose and treat it. Prescrire Int 2007;16(87):24-31.
4. Morgan MY, Colman JC, Sherlock S. The use of a combination of peripheral markers for diagnosing alcoholism and monitoring for continued abuse. Alcohol Alcohol 1981;16:167-77.
5. Hietala J, Koivisto H, Anttila P, et al. Comparison of the combined marker GGT-CDT and the conventional laboratory markers of alcohol abuse in heavy drinkers, moderate drinkers and abstainers. Alcohol Alcohol 2006;41(5):528-33.
6. Swift R. Direct measurement of alcohol and its metabolites. Addiction 2003;98:73-80.
7. Koivisto H, Hietala J, Anttila P, et al. Long-term ethanol consumption and macrocytosis: diagnostic and pathogenic implications. J Lab Clin Med 2005;147(4):191-6.
8. Savage DG, Ogundipe A, Allen RH, et al. Etiology and diagnostic evaluation of macrocytosis. Am J Med Sci 2000;319(6):343-52.
9. Gordon H. Detection of alcoholic liver disease. World J Gastroenterol 2001;7(3):297-302.
10. Bernadt M, Mumford J, Taylor C, et al. Comparison of questionnaire and laboratory tests in the detection of excessive drinking and alcoholism. Lancet 1982;1:325-8.
11. Hasselblatt M, Martin F, Maul O, et al. Persistent macrocytosis following abstinence from chronic alcohol use. JAMA 2001;286:2946.-
12. Prati D, Taioli E, Zanella A, et al. Updated definitions of healthy ranges for serum alanine aminotransferase levels. Ann Intern Med 2002;137:1-9.
13. Fancher T, Kamboj A, Onate J. Interpreting liver function tests. Current Psychiatry 2007;6(5):61-8.
14. Puukka K, Hietala J, Koivisto H, et al. Obesity and the clinical use of serum GGT activity as a marker of heavy drinking. Scand J Clin Lab Invest 2007;67(5):480-8.
15. Litten RZ, Allen JP, Fertig JB. y-glutamyl transpeptidase and carbohydrate deficient transferrin: alternative measures of excessive alcohol consumption. Alcohol Clin Exp Res 1995;19(6):1541-6.
16. National Institute on Alcohol Abuse and Alcoholism. Screening for alcohol problems—an update. Alcohol Alert No 56 Available at: http://pubs.niaaa.nih.gov/publications/aa56.htm. Accessed May 5, 2007.
17. DiMartini A. A clinical guide to assessing alcohol use and problems. Available at: http://www.alcoholmedicalscholars.org/clin-asmt.ppt. Accessed June 30, 2008.
18. Wolff K, Marshall E. Biological markers of alcohol use. Psychiatry 2006;5(12):437-8.
19. ARUP Laboratories. Carbohydrate-deficient transferrin (CDT) for alcohol use. 2006. Available at: http://www.aruplab.com/TestDirectory/resources/TechnicalBulletins/Carbohydrate-Deficient%20Transferrin%20(CDT)%20Mar%202006.pdf. Accessed July 30, 2008.
20. Allen JP, Litten RZ. The role of laboratory testing in alcoholism treatment. J Subst Abuse Treat 2001;20:81-5.
21. Bhushan V, Le T, Ozturk A, et al. Behavioral Science. In: Le T, Bhushan V, Rao DA, eds. First aid for the USMLE step 1: a student to student guide. New York, NY: McGraw Hill Medical Publishing Division; 2007.
22. Miller PM, Anton RF. Biochemical alcohol screening in primary care. Addict Behav 2004;29(7):1427-37.
23. Schmidt LG, Schmidt K, Dufeu P, et al. Superiority of carbohydrate-deficient transferrin to gamma-glutamyltransferase in detecting relapse in alcoholism. Am J Psychiatry 1997;154(1):75-80.
24. Salaspuro M. Carbohydrate-deficient transferrin as compared to other markers of alcoholism: a systematic review. Alcohol 1999;19(3):261-71.
25. Dolman JM, Hawkes ND. Combining the AUDIT questionnaire and biochemical markers to assess alcohol use and risk of alcohol withdrawal in medical patients. Alcohol Alcohol 2005;40(6):515-9.
26. Helander A, Carlsson AV, Borg S. Longitudinal comparison of carbohydrate-deficient transferrin and gamma-glutamyl transferase: complementary markers of excessive alcohol consumption. Alcohol Alcohol 1996;31(1):101-7.
27. Gluud C, Andersen I, Dietrichson O, et al. Gamma-glutamyltransferase, aspartate aminotransferase and alkaline phosphatase as markers of alcohol consumption in out-patient alcoholics. Eur J Clin Invest 1981;11(3):171-6.
28. Kawachi I, Robinson GM, Stace NH. A combination of raised serum AST: ALT ratio and erythrocyte mean cell volume level detects excessive alcohol consumption. N Z Med J 1990;103(887):145-8.
29. Mundle G, Ackerman K, Mann K. Biological markers as indicators for relapse in alcohol-dependent patients. Addict Biol 1999;4(2):209-14.
30. Bell H, Tallaksen C, Sjåheim T, et al. Serum carbohydrate-deficient transferrin as a marker of alcohol consumption in patients with chronic liver diseases. Alcohol Clin Exp Res 1993;17(2):246-52.
31. Sillanaukee P, Aalto M, Seppa K. Carbohydrate-deficient transferrin and conventional alcohol markers as indicators for brief intervention among heavy drinkers in primary health care. Alcohol Clin Exp Res 1998;22(4):892-6.
32. Salaspuro S. Conventional and coming laboratory markers of alcoholism and heavy drinking. Alcohol Clin Exp Res 1986;10(6 suppl):5-12.
33. Della Penna ND, Treisman GJ. HIV/AIDS. In: Levenson J, ed. Essentials of psychosomatic medicine. Washington, DC: American Psychiatric Publishing, Inc; 2007.


