User login
Rapid Implementation of a Clinical Workflow Support Tool to Engage Rural Veterans about a Smoking Cessation Trial
Background
Offering participation in clinical trials is a standard of care practice in oncology. It is also considered a quality indicator by various professional cancer societies, including the American Societies of Hematology (ASH) and Clinical Oncology (ASCO). In 2023, VA launched Clinical Cancer Research Services (CCRS) to ensure that all Veterans with cancer can participate in a clinical trial should they choose to do so. Research teams struggle to identify and engage potentially eligible patients. This is a complex process involving eligibility screening, outreach, and personalized support, which frequently involves a manual workflow with inefficiencies, delays, and missed opportunities for patients. To support CCRS’s mission, we used VA Enterprise Cloud (VAEC) to rapidly develop a clinical workflow support application for CCRS team members.
Methods
We used an internally developed framework to rapidly define program aims, provider workflows, opportunities to augment with data products, and lean principles applied to health information technology to design a clinical workflow supporting application. Data products leveraged VAEC’s Summit Data Platform (SDP), an open, multi-cloud platform for ingesting, curating, and managing multi-source VHA data into usable products. User interface was developed in a low code/no code power platform environment, which integrates with SDP and is also available in VAEC.
Results
An initial aim was identified as supporting engagement for the ‘Reaching Rural Cancer Survivors Who Smoke Using Text-based Cessation Interventions’ study. Augmented workflow was identified by meeting principal stakeholders and staff. Data product development involved retrieval of cancer diagnoses from the VA cancer registry system and smoking status from CDW HealthFactors. Rural residence was identified using 2023 Rural-Urban Continuum Codes. Application design, testing and refinement followed. Design to implementation was accomplished over the span of two months: from Aug 5, 2024 to Oct 3, 2024. Over the next seven months, the application identified 2,603 potentially eligible Veterans, and a single navigator using the tool was able to review 456 cases, send 189 study letters, and enroll 5 Veterans.
Conclusions
Clinical workflow support tools that leverage cloud infrastructure such as VAEC and Summit Data Platform can improve system efficiencies and increase access to clinical trials.
Background
Offering participation in clinical trials is a standard of care practice in oncology. It is also considered a quality indicator by various professional cancer societies, including the American Societies of Hematology (ASH) and Clinical Oncology (ASCO). In 2023, VA launched Clinical Cancer Research Services (CCRS) to ensure that all Veterans with cancer can participate in a clinical trial should they choose to do so. Research teams struggle to identify and engage potentially eligible patients. This is a complex process involving eligibility screening, outreach, and personalized support, which frequently involves a manual workflow with inefficiencies, delays, and missed opportunities for patients. To support CCRS’s mission, we used VA Enterprise Cloud (VAEC) to rapidly develop a clinical workflow support application for CCRS team members.
Methods
We used an internally developed framework to rapidly define program aims, provider workflows, opportunities to augment with data products, and lean principles applied to health information technology to design a clinical workflow supporting application. Data products leveraged VAEC’s Summit Data Platform (SDP), an open, multi-cloud platform for ingesting, curating, and managing multi-source VHA data into usable products. User interface was developed in a low code/no code power platform environment, which integrates with SDP and is also available in VAEC.
Results
An initial aim was identified as supporting engagement for the ‘Reaching Rural Cancer Survivors Who Smoke Using Text-based Cessation Interventions’ study. Augmented workflow was identified by meeting principal stakeholders and staff. Data product development involved retrieval of cancer diagnoses from the VA cancer registry system and smoking status from CDW HealthFactors. Rural residence was identified using 2023 Rural-Urban Continuum Codes. Application design, testing and refinement followed. Design to implementation was accomplished over the span of two months: from Aug 5, 2024 to Oct 3, 2024. Over the next seven months, the application identified 2,603 potentially eligible Veterans, and a single navigator using the tool was able to review 456 cases, send 189 study letters, and enroll 5 Veterans.
Conclusions
Clinical workflow support tools that leverage cloud infrastructure such as VAEC and Summit Data Platform can improve system efficiencies and increase access to clinical trials.
Background
Offering participation in clinical trials is a standard of care practice in oncology. It is also considered a quality indicator by various professional cancer societies, including the American Societies of Hematology (ASH) and Clinical Oncology (ASCO). In 2023, VA launched Clinical Cancer Research Services (CCRS) to ensure that all Veterans with cancer can participate in a clinical trial should they choose to do so. Research teams struggle to identify and engage potentially eligible patients. This is a complex process involving eligibility screening, outreach, and personalized support, which frequently involves a manual workflow with inefficiencies, delays, and missed opportunities for patients. To support CCRS’s mission, we used VA Enterprise Cloud (VAEC) to rapidly develop a clinical workflow support application for CCRS team members.
Methods
We used an internally developed framework to rapidly define program aims, provider workflows, opportunities to augment with data products, and lean principles applied to health information technology to design a clinical workflow supporting application. Data products leveraged VAEC’s Summit Data Platform (SDP), an open, multi-cloud platform for ingesting, curating, and managing multi-source VHA data into usable products. User interface was developed in a low code/no code power platform environment, which integrates with SDP and is also available in VAEC.
Results
An initial aim was identified as supporting engagement for the ‘Reaching Rural Cancer Survivors Who Smoke Using Text-based Cessation Interventions’ study. Augmented workflow was identified by meeting principal stakeholders and staff. Data product development involved retrieval of cancer diagnoses from the VA cancer registry system and smoking status from CDW HealthFactors. Rural residence was identified using 2023 Rural-Urban Continuum Codes. Application design, testing and refinement followed. Design to implementation was accomplished over the span of two months: from Aug 5, 2024 to Oct 3, 2024. Over the next seven months, the application identified 2,603 potentially eligible Veterans, and a single navigator using the tool was able to review 456 cases, send 189 study letters, and enroll 5 Veterans.
Conclusions
Clinical workflow support tools that leverage cloud infrastructure such as VAEC and Summit Data Platform can improve system efficiencies and increase access to clinical trials.
Hematology and Oncology Staffing Levels for Fiscal Years 19–24
Background
Department of Veterans Affairs (VA) faces a landscape of increasingly complex practice, especially in Hematology/Oncology (H/O), and a nationwide shortage of healthcare providers, while serving more Veterans than ever before. To understand current and future staffing needs, the VA National Oncology Program performed an assessment of H/O staffing, including attending physicians, residents/ fellows, licensed independent practitioners (LIPs) (nurse practitioners/physician assistants), and nurses for fiscal years (FY) 19–24.
Methods
Using VA Corporate Data Warehouse, we identified H/O visits in VA from 10/01/2018 through 09/30/2024 using stop codes. No-show (< 0.00001%) and National TeleOncology appointments (1%) were removed. We retrieved all notes associated with resulting visits and used area-ofspecialization and provider-type data to identify all attending physicians, trainees, LIPs, and nurses who authored or cosigned these notes. We identified H/O staff as 1. those associated with H/O clinic locations, 2. physicians who consistently cosigned H/O notes authored by fellows and LIPs associated with H/O locations, 3. fellows and LIPs authoring notes that were then cosigned by H/O physicians, and 4. nurses authoring notes associated with H/O visits.
Analysis
For each FY, we obtained total numbers of visits, unique patients, and care-providing staff by type. For validation, collaborating providers at several sites reviewed visit information, and a colleague also performed an independent, parallel data extraction. We adjusted FY totals to account for the growing patient population by dividing unique staff count by number of unique patients and multiplying by 200,000 (the approximate number of unique patients in FY19).
Results
From FY19 through FY24, VA Hematology/ Oncology saw a 14.6% rise in unique patients (from 232,084 to 265,926) and a 15.4% rise in visits (from 923,175 to 1,065,186). The absolute number of attendings rose by 4 (0.6%); of LIPs, by 138 (14.4%); and of nurses, by 142 (4.9%); trainees fell by 102 (4.3%). Adjusted to 200,000 patients, the number of attendings fell by 76 (12.3%); LIPs, by 1 (0.1%); trainees, by 335 (16.5%); and nurses, by 211 (8.4%).
Conclusions
Adjusted to number of Veterans, there are 10.4% fewer staff in Hematology/Oncology in FY24 compared to FY19.
Background
Department of Veterans Affairs (VA) faces a landscape of increasingly complex practice, especially in Hematology/Oncology (H/O), and a nationwide shortage of healthcare providers, while serving more Veterans than ever before. To understand current and future staffing needs, the VA National Oncology Program performed an assessment of H/O staffing, including attending physicians, residents/ fellows, licensed independent practitioners (LIPs) (nurse practitioners/physician assistants), and nurses for fiscal years (FY) 19–24.
Methods
Using VA Corporate Data Warehouse, we identified H/O visits in VA from 10/01/2018 through 09/30/2024 using stop codes. No-show (< 0.00001%) and National TeleOncology appointments (1%) were removed. We retrieved all notes associated with resulting visits and used area-ofspecialization and provider-type data to identify all attending physicians, trainees, LIPs, and nurses who authored or cosigned these notes. We identified H/O staff as 1. those associated with H/O clinic locations, 2. physicians who consistently cosigned H/O notes authored by fellows and LIPs associated with H/O locations, 3. fellows and LIPs authoring notes that were then cosigned by H/O physicians, and 4. nurses authoring notes associated with H/O visits.
Analysis
For each FY, we obtained total numbers of visits, unique patients, and care-providing staff by type. For validation, collaborating providers at several sites reviewed visit information, and a colleague also performed an independent, parallel data extraction. We adjusted FY totals to account for the growing patient population by dividing unique staff count by number of unique patients and multiplying by 200,000 (the approximate number of unique patients in FY19).
Results
From FY19 through FY24, VA Hematology/ Oncology saw a 14.6% rise in unique patients (from 232,084 to 265,926) and a 15.4% rise in visits (from 923,175 to 1,065,186). The absolute number of attendings rose by 4 (0.6%); of LIPs, by 138 (14.4%); and of nurses, by 142 (4.9%); trainees fell by 102 (4.3%). Adjusted to 200,000 patients, the number of attendings fell by 76 (12.3%); LIPs, by 1 (0.1%); trainees, by 335 (16.5%); and nurses, by 211 (8.4%).
Conclusions
Adjusted to number of Veterans, there are 10.4% fewer staff in Hematology/Oncology in FY24 compared to FY19.
Background
Department of Veterans Affairs (VA) faces a landscape of increasingly complex practice, especially in Hematology/Oncology (H/O), and a nationwide shortage of healthcare providers, while serving more Veterans than ever before. To understand current and future staffing needs, the VA National Oncology Program performed an assessment of H/O staffing, including attending physicians, residents/ fellows, licensed independent practitioners (LIPs) (nurse practitioners/physician assistants), and nurses for fiscal years (FY) 19–24.
Methods
Using VA Corporate Data Warehouse, we identified H/O visits in VA from 10/01/2018 through 09/30/2024 using stop codes. No-show (< 0.00001%) and National TeleOncology appointments (1%) were removed. We retrieved all notes associated with resulting visits and used area-ofspecialization and provider-type data to identify all attending physicians, trainees, LIPs, and nurses who authored or cosigned these notes. We identified H/O staff as 1. those associated with H/O clinic locations, 2. physicians who consistently cosigned H/O notes authored by fellows and LIPs associated with H/O locations, 3. fellows and LIPs authoring notes that were then cosigned by H/O physicians, and 4. nurses authoring notes associated with H/O visits.
Analysis
For each FY, we obtained total numbers of visits, unique patients, and care-providing staff by type. For validation, collaborating providers at several sites reviewed visit information, and a colleague also performed an independent, parallel data extraction. We adjusted FY totals to account for the growing patient population by dividing unique staff count by number of unique patients and multiplying by 200,000 (the approximate number of unique patients in FY19).
Results
From FY19 through FY24, VA Hematology/ Oncology saw a 14.6% rise in unique patients (from 232,084 to 265,926) and a 15.4% rise in visits (from 923,175 to 1,065,186). The absolute number of attendings rose by 4 (0.6%); of LIPs, by 138 (14.4%); and of nurses, by 142 (4.9%); trainees fell by 102 (4.3%). Adjusted to 200,000 patients, the number of attendings fell by 76 (12.3%); LIPs, by 1 (0.1%); trainees, by 335 (16.5%); and nurses, by 211 (8.4%).
Conclusions
Adjusted to number of Veterans, there are 10.4% fewer staff in Hematology/Oncology in FY24 compared to FY19.
Anchors Aweigh, Clinical Trial Navigation at the VA!
Background
Despite the benefit of cancer clinical trials (CTs) in increasing medical knowledge and broadening treatment options, VA oncologists face challenges referring or enrolling Veterans in CTs including identifying appropriate CTs and navigating the referral process especially for non-VA CTs. To address these challenges, the VA National Oncology Program (NOP) provided guidance regarding community care referral for CT participation and established the Cancer Clinical Trial Nurse Navigation (CTN) service.
Methods
Referrals to CTN occur via Precision Oncology consult or email to CancerClinicalTrialsNavigation@va.gov. The CT nurse navigator educates Veterans about CTs, identifies CTs for Veterans based on disease and geographic area, provides written summaries to Veterans and VA oncologists, and facilitates communication between clinical and research teams. Descriptive statistics were used to summarize characteristics of Veterans referred to CTN and results of the CTN searches. A semi-structured survey was used to assess satisfaction from 50 VA oncologists who had used the CTN service.
Results
Between June 2023 and May 2024, 72 Veterans were referred to CTN. Patient characteristics include male (94%), non-rural (65%), median age 66.5 (range 27-80), self-reported race as White (74%) and Black (22%), cancer type as solid tumor (73%) and blood cancer (27%). The median number of CTs found for each Veteran was two (range 0 - 12). No referred Veterans enrolled in CTs, with the most common causes being CT ineligibility and desire to receive standard therapy in the VA. Twenty oncologists were educated about NOP CT guidance. The response rate to the feedback survey was modest (34%) but 94% of survey respondents rated their overall satisfaction as highly satisfied or satisfied.
Conclusions
The CTN assists Veterans and VA oncologists in connecting with CTs. The high satisfaction rate and ability to reach a racially and geographically diverse Veteran population are measures of early program success. By lowering the barriers for VA oncologists to consider CTs for their patients, the CTN expects increased and earlier referrals of Veterans, which may improve CT eligibility and participation. Future efforts to provide disease-directed education about CTs to Veterans and VA oncologists is intended to encourage early consideration of CTs.
Background
Despite the benefit of cancer clinical trials (CTs) in increasing medical knowledge and broadening treatment options, VA oncologists face challenges referring or enrolling Veterans in CTs including identifying appropriate CTs and navigating the referral process especially for non-VA CTs. To address these challenges, the VA National Oncology Program (NOP) provided guidance regarding community care referral for CT participation and established the Cancer Clinical Trial Nurse Navigation (CTN) service.
Methods
Referrals to CTN occur via Precision Oncology consult or email to CancerClinicalTrialsNavigation@va.gov. The CT nurse navigator educates Veterans about CTs, identifies CTs for Veterans based on disease and geographic area, provides written summaries to Veterans and VA oncologists, and facilitates communication between clinical and research teams. Descriptive statistics were used to summarize characteristics of Veterans referred to CTN and results of the CTN searches. A semi-structured survey was used to assess satisfaction from 50 VA oncologists who had used the CTN service.
Results
Between June 2023 and May 2024, 72 Veterans were referred to CTN. Patient characteristics include male (94%), non-rural (65%), median age 66.5 (range 27-80), self-reported race as White (74%) and Black (22%), cancer type as solid tumor (73%) and blood cancer (27%). The median number of CTs found for each Veteran was two (range 0 - 12). No referred Veterans enrolled in CTs, with the most common causes being CT ineligibility and desire to receive standard therapy in the VA. Twenty oncologists were educated about NOP CT guidance. The response rate to the feedback survey was modest (34%) but 94% of survey respondents rated their overall satisfaction as highly satisfied or satisfied.
Conclusions
The CTN assists Veterans and VA oncologists in connecting with CTs. The high satisfaction rate and ability to reach a racially and geographically diverse Veteran population are measures of early program success. By lowering the barriers for VA oncologists to consider CTs for their patients, the CTN expects increased and earlier referrals of Veterans, which may improve CT eligibility and participation. Future efforts to provide disease-directed education about CTs to Veterans and VA oncologists is intended to encourage early consideration of CTs.
Background
Despite the benefit of cancer clinical trials (CTs) in increasing medical knowledge and broadening treatment options, VA oncologists face challenges referring or enrolling Veterans in CTs including identifying appropriate CTs and navigating the referral process especially for non-VA CTs. To address these challenges, the VA National Oncology Program (NOP) provided guidance regarding community care referral for CT participation and established the Cancer Clinical Trial Nurse Navigation (CTN) service.
Methods
Referrals to CTN occur via Precision Oncology consult or email to CancerClinicalTrialsNavigation@va.gov. The CT nurse navigator educates Veterans about CTs, identifies CTs for Veterans based on disease and geographic area, provides written summaries to Veterans and VA oncologists, and facilitates communication between clinical and research teams. Descriptive statistics were used to summarize characteristics of Veterans referred to CTN and results of the CTN searches. A semi-structured survey was used to assess satisfaction from 50 VA oncologists who had used the CTN service.
Results
Between June 2023 and May 2024, 72 Veterans were referred to CTN. Patient characteristics include male (94%), non-rural (65%), median age 66.5 (range 27-80), self-reported race as White (74%) and Black (22%), cancer type as solid tumor (73%) and blood cancer (27%). The median number of CTs found for each Veteran was two (range 0 - 12). No referred Veterans enrolled in CTs, with the most common causes being CT ineligibility and desire to receive standard therapy in the VA. Twenty oncologists were educated about NOP CT guidance. The response rate to the feedback survey was modest (34%) but 94% of survey respondents rated their overall satisfaction as highly satisfied or satisfied.
Conclusions
The CTN assists Veterans and VA oncologists in connecting with CTs. The high satisfaction rate and ability to reach a racially and geographically diverse Veteran population are measures of early program success. By lowering the barriers for VA oncologists to consider CTs for their patients, the CTN expects increased and earlier referrals of Veterans, which may improve CT eligibility and participation. Future efforts to provide disease-directed education about CTs to Veterans and VA oncologists is intended to encourage early consideration of CTs.
RVD With Weekly Bortezomib Has a Favorable Toxicity and Effectiveness Profile in a Large Cohort of US Veterans With Multiple Myeloma
Background
Lenalidomide, bortezomib, and dexamethasone (RVD) is standard triplet induction for fit newly-diagnosed myeloma (NDMM) patients, with response rate (RR)>90%. A 21-day cycle with bortezomib given days 1, 4, 8, and 11 (2x/w) is standard. However, up to 80% of patients develop neuropathy. Weekly bortezomib dosing (1x/w), subcutaneous route, and 28- to 35-day cycle length may optimize tolerance. We present an effectiveness and toxicity analysis of Veterans who received RVD with 1x/w and 2x/w bortezomib for NDMM.
Methods
The VA Corporate Data Warehouse identified 1499 Veterans with NDMM given RVD ≤42 days of treatment start. 840 Veterans were grouped for initial analysis based on criteria: 1) lenalidomide and ≥ 3 bortezomib doses during cycle 1; 2) ≥6 mean days between bortezomib treatments=1x/w); and 3) number of lenalidomide days informed cycle length (21d, 28d, or 35d; default 7-day rest). Investigators reviewed algorithm results to finalize group assignments. Endpoints included depth of response, time to next treatment (TTNT), overall survival (OS), and neuropathy. Neuropathy was defined as use of neuropathy medications and neuropathy ICD-10 codes.
Results
Our algorithm correctly assigned 82% of 840 cycle 1 RVD schedules. The largest groups were 21d 1x/w (n=291), 21d 2x/w (n=193), 28d 1x/w (n=188), and 28d 2x/w (n=82). Median age was 68.3; 53% were non-Hispanic White. Demographics and ISS stage of groups were similar. 30% underwent autologous transplant. Tolerability. Median number of bortezomib doses ranged from 22.5-25.5 (p=0.57). Neuropathy favored 1x/w, 17.7 vs 30.2% (p=0.0001) and was highest (34.7%) in 21d 2x/w. Effectiveness. Response was assessable for 28% of patients. RR (72%, p=0.68) and median TTNT (19.3 months, p=0.20) were similar, including 1x/w vs 2x/w comparison (p=0.79). 21d regimens optimized TTNT (21.4 vs 13.9 months, p=0.045) and trended to better OS (73 vs 65 months, p=0.06).
Conclusions
1x/w RVD preserved effectiveness compared to “standard” RVD in a large Veteran cohort. 1x/w reduced neuropathy incidence. 28d regimens demonstrated inferior longer-term outcomes. Certain endpoints, such as RR and neuropathy, appear underestimated due to data source limitations. 21d 1x/w RVD optimizes effectiveness, tolerability, and administration and should be considered for broader utilization in Veterans with NDMM.
Background
Lenalidomide, bortezomib, and dexamethasone (RVD) is standard triplet induction for fit newly-diagnosed myeloma (NDMM) patients, with response rate (RR)>90%. A 21-day cycle with bortezomib given days 1, 4, 8, and 11 (2x/w) is standard. However, up to 80% of patients develop neuropathy. Weekly bortezomib dosing (1x/w), subcutaneous route, and 28- to 35-day cycle length may optimize tolerance. We present an effectiveness and toxicity analysis of Veterans who received RVD with 1x/w and 2x/w bortezomib for NDMM.
Methods
The VA Corporate Data Warehouse identified 1499 Veterans with NDMM given RVD ≤42 days of treatment start. 840 Veterans were grouped for initial analysis based on criteria: 1) lenalidomide and ≥ 3 bortezomib doses during cycle 1; 2) ≥6 mean days between bortezomib treatments=1x/w); and 3) number of lenalidomide days informed cycle length (21d, 28d, or 35d; default 7-day rest). Investigators reviewed algorithm results to finalize group assignments. Endpoints included depth of response, time to next treatment (TTNT), overall survival (OS), and neuropathy. Neuropathy was defined as use of neuropathy medications and neuropathy ICD-10 codes.
Results
Our algorithm correctly assigned 82% of 840 cycle 1 RVD schedules. The largest groups were 21d 1x/w (n=291), 21d 2x/w (n=193), 28d 1x/w (n=188), and 28d 2x/w (n=82). Median age was 68.3; 53% were non-Hispanic White. Demographics and ISS stage of groups were similar. 30% underwent autologous transplant. Tolerability. Median number of bortezomib doses ranged from 22.5-25.5 (p=0.57). Neuropathy favored 1x/w, 17.7 vs 30.2% (p=0.0001) and was highest (34.7%) in 21d 2x/w. Effectiveness. Response was assessable for 28% of patients. RR (72%, p=0.68) and median TTNT (19.3 months, p=0.20) were similar, including 1x/w vs 2x/w comparison (p=0.79). 21d regimens optimized TTNT (21.4 vs 13.9 months, p=0.045) and trended to better OS (73 vs 65 months, p=0.06).
Conclusions
1x/w RVD preserved effectiveness compared to “standard” RVD in a large Veteran cohort. 1x/w reduced neuropathy incidence. 28d regimens demonstrated inferior longer-term outcomes. Certain endpoints, such as RR and neuropathy, appear underestimated due to data source limitations. 21d 1x/w RVD optimizes effectiveness, tolerability, and administration and should be considered for broader utilization in Veterans with NDMM.
Background
Lenalidomide, bortezomib, and dexamethasone (RVD) is standard triplet induction for fit newly-diagnosed myeloma (NDMM) patients, with response rate (RR)>90%. A 21-day cycle with bortezomib given days 1, 4, 8, and 11 (2x/w) is standard. However, up to 80% of patients develop neuropathy. Weekly bortezomib dosing (1x/w), subcutaneous route, and 28- to 35-day cycle length may optimize tolerance. We present an effectiveness and toxicity analysis of Veterans who received RVD with 1x/w and 2x/w bortezomib for NDMM.
Methods
The VA Corporate Data Warehouse identified 1499 Veterans with NDMM given RVD ≤42 days of treatment start. 840 Veterans were grouped for initial analysis based on criteria: 1) lenalidomide and ≥ 3 bortezomib doses during cycle 1; 2) ≥6 mean days between bortezomib treatments=1x/w); and 3) number of lenalidomide days informed cycle length (21d, 28d, or 35d; default 7-day rest). Investigators reviewed algorithm results to finalize group assignments. Endpoints included depth of response, time to next treatment (TTNT), overall survival (OS), and neuropathy. Neuropathy was defined as use of neuropathy medications and neuropathy ICD-10 codes.
Results
Our algorithm correctly assigned 82% of 840 cycle 1 RVD schedules. The largest groups were 21d 1x/w (n=291), 21d 2x/w (n=193), 28d 1x/w (n=188), and 28d 2x/w (n=82). Median age was 68.3; 53% were non-Hispanic White. Demographics and ISS stage of groups were similar. 30% underwent autologous transplant. Tolerability. Median number of bortezomib doses ranged from 22.5-25.5 (p=0.57). Neuropathy favored 1x/w, 17.7 vs 30.2% (p=0.0001) and was highest (34.7%) in 21d 2x/w. Effectiveness. Response was assessable for 28% of patients. RR (72%, p=0.68) and median TTNT (19.3 months, p=0.20) were similar, including 1x/w vs 2x/w comparison (p=0.79). 21d regimens optimized TTNT (21.4 vs 13.9 months, p=0.045) and trended to better OS (73 vs 65 months, p=0.06).
Conclusions
1x/w RVD preserved effectiveness compared to “standard” RVD in a large Veteran cohort. 1x/w reduced neuropathy incidence. 28d regimens demonstrated inferior longer-term outcomes. Certain endpoints, such as RR and neuropathy, appear underestimated due to data source limitations. 21d 1x/w RVD optimizes effectiveness, tolerability, and administration and should be considered for broader utilization in Veterans with NDMM.
Sequential Targeted Treatment for a Geriatric Patient with Acute Myeloid Leukemia with Concurrent FLT3-TKD and IDH1 Mutations
Nearly 20,000 patients are diagnosed with acute myeloid leukemia (AML) in the US annually.1 Despite the use of aggressive chemotherapeutic agents, the prognosis remains poor, with a mean 5-year survival of 28.3%.2 Fortunately, with the refinement of next-generation sequencing (NGS) hematology panels and development of systemic targeted therapies, the treatment landscape for eligible patients has improved, both in frontline and relapsed or refractory (R/R) patients.
Specifically, investigations into alterations within the FMS-like tyrosine kinase (FLT3) and isocitrate dehydrogenase (IDH) genes have led to the discovery of a number of targeted treatments. Midostaurin is US Food and Drug Administration (FDA)-approved for use in combination with induction chemotherapy for patients with internal tandem duplication of the FLT3 (FLT3-ITD) gene or mutations within the tyrosine kinase domain (FLT3-TKD).3 Ivosidenib is indicated for frontline treatment for those who are poor candidates for induction chemotherapy, and R/R patients who have an R132H mutation in IDH1.4,5 Enasidenib is FDA-approved for R/R patients with R140Q, R172S, and R172K mutations in IDH2.6
The optimal treatment for patients with AML with ≥ 2 clinically actionable mutations has not been established. In this article we describe a geriatric patient who initially was diagnosed with AML with concurrent FLT3-TKD and IDH1 mutations and received targeted, sequential management. We detail changes in disease phenotype and mutational status by repeating an NGS hematology panel and cytogenetic studies after each stage of therapy. Lastly, we discuss the clonal evolution apparent within leukemic cells with use of ≥ 1 or more targeted agents.
Case Presentation
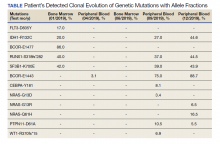
A 68-year-old man presented to the Emergency Department at The Durham Veterans Affairs Medical Center in North Carolina with fatigue and light-headedness. Because of his symptoms and pancytopenia, a bone marrow aspiration and trephine biopsy were performed, which showed 57% myeloblasts, 12% promyelocytes/myelocytes, and 2% metamyelocytes in 20 to 30% cellular bone marrow. Flow cytometry confirmed a blast population consistent with AML. A LeukoVantage (Quest Diagnostics) hematologic NGS panel revealed the presence of FLT3-TKD, IDH1, RUNX1, BCOR-E1477, and SF3B1 mutations (Table). Initial fluorescence in situ hybridization (FISH) results showed a normal pattern of hybridization with no translocations. His disease was deemed to be intermediate-high risk because of the presence of FLT3-TKD and RUNX1 mutations, despite the normal cytogenetic profile and absence of additional clinical features.
Induction chemotherapy was started with idarubicin, 12 mg/m2, on days 1 to 3 and cytarabine, 200 mg/m2, on days 1 to 7. Because of the presence of a FLT3-TKD mutation, midostaurin was planned for days 8 to 21. After induction chemotherapy, a bone marrow biopsy on day 14 revealed an acellular marrow with no observed myeloblasts. A bone marrow biopsy conducted before initiating consolidation therapy, revealed 30% cellularity with morphologic remission. However, flow cytometry found 5% myeloblasts expressing CD34, CD117, CD13, CD38, and HLA-DR, consistent with measurable residual disease. He received 2 cycles of consolidation therapy with high-dose cytarabine combined with midostaurin. After the patient's second cycle of consolidation, he continued to experience transfusion-dependent cytopenias. Another bone marrow evaluation demonstrated 10% cellularity with nearly all cells appearing to be myeloblasts. A repeat LeukoVantage NGS panel demonstrated undetectable FLT3-TKD mutation and persistent IDH1-R123C mutation. FISH studies revealed a complex karyotype with monosomy of chromosomes 5 and 7 and trisomy of chromosome 8.
We discussed with the patient and his family the options available, which included initiating targeted therapy for his IDH1 mutation, administering hypomethylation therapy with or without venetoclax, or pursuing palliative measures. We collectively decided to pursue therapy with single-agent oral ivosidenib, 500 mg daily. After 1 month of treatment, our patient developed worsening fatigue. His white blood cell count had increased to > 43 k/cm2, raising concern for differentiation syndrome.
A review of the peripheral smear showed a wide-spectrum of maturing granulocytes, with a large percentage of blasts. Peripheral flow cytometry confirmed a blast population of 15%. After a short period of symptom improvement with steroids, the patient developed worsening confusion. Brain imaging identified 2 subdural hemorrhages. Because of a significant peripheral blast population and the development of these hemorrhages, palliative measures were pursued, and the patient was discharged to an inpatient hospice facility. A final NGS panel performed from peripheral blood detected mutations in IDH1, RUNX1, PTPN11, NRAS, BCOR-E1443, and SF3B1 genes.
Discussion
To our knowledge, this is the first reported case of a patient who sequentially received targeted treatments directed against both FLT3 and IDH1 mutations. Initial management with midostaurin and cytarabine resulted in sustained remission of his FLT3-TKD mutation. However, despite receiving prompt standard of care with combination induction chemotherapy and targeted therapy, the patient experienced unfavorable clonal evolution based upon his molecular and cytogenetic testing. Addition of ivosidenib as a second targeting agent for his IDH1 mutation did not achieve a second remission.
Clonal evolution is a well-described phenomenon in hematology. Indolent conditions, such as clonal hematopoiesis of intermediate potential, or malignancies, such as myelodysplastic syndromes and myeloproliferative neoplasms, could transform into acute leukemia through the accumulation of driver mutations and/or cytogenetic abnormalities. Clonal evolution often is viewed as the culprit in patients with AML whose disease relapses after remission with initial chemotherapy.7-10 With the increasing availability of commercial NGS panels designed to assess mutations among patients experiencing hematologic malignancies, patterns of relapse, and, models of clonal evolution could be observed closely in patients with AML.
We were able to monitor molecular changes within our patient’s predominant clonal populations by repeating peripheral comprehensive NGS panels after lines of targeted therapies. The repeated sequencing revealed that clones with FLT3-TKD mutations responded to midostaurin with first-line chemotherapy whereas it was unclear whether clones with IDH1 mutation responded to ivosidenib. Development of complex cytogenetic findings along with the clonal expansion of BCOR mutation-harboring cells likely contributed to our patient’s acutely worsening condition. Several studies have found that the presence of a BCOR mutation in adults with AML leads to lower overall survival and relapse-free survival.11,12 As of now, there are no treatments specifically targeting BCOR mutations.
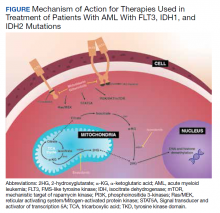
Although there are novel targeting agents with proven efficacy for both FLT3 and IDH1 mutations (Figure), it is difficult to determine which pathogenic mutation drives disease onset. No evidence suggests that these drugs could be administered in tandem. At the present time, interest is directed towards targeting all AML subclones simultaneously, which could reduce the likelihood of evolution among founder clones.7,10,13 In their comparison between molecular profiles and outcomes of patients with AML, Papaemmanuil and colleagues observed that > 80% of patients with AML harbor ≥ 2 driver mutations concurrently.14 Moreover, FLT3-ITD and IDH1 mutations tend to co-occur in approximately 9 to 27% of AML cases.15-18 Available targeted agents for AML are relatively new and hematologists’ familiarity with these drugs is continuing to grow. As the number of novel agents increases, investigations directed toward assessing the safety profile and efficacy of combining targeted agents will be beneficial for patients with AML with ≥ 1 driver mutation.
Conclusions
For our patient with AML, sequential targeted management of FLT3-TKD and IDH1 mutations was not beneficial. Higher-risk disease features, such as the development of a complex karyotype, likely contributed to our patient’s poor response to second-line ivosidenib. The sequential NGS malignant hematology panels allowed us to closely monitor changes to the molecular structure of our patient’s AML after each line of targeted therapy. Future investigations of combining targeted agents for patients with AML with concurrent actionable mutations would provide insight into outcomes of treating multiple clonal populations simultaneously.
1. De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: a comprehensive review and 2016 update. Blood Cancer J. 2016;6(7):e441. doi:10.1038/bcj.2016.50.
2. National Cancer Institute. Cancer Stat Facts: Leukemia — acute myeloid leukemia (AML). Accessed November 4, 2020. https://seer.cancer.gov/statfacts/html/amyl.html
3. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464. doi:10.1056/NEJMoa1614359.
4. DiNardo CD, Stein EM, de Botton S, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378(25):2386-2398. doi:10.1056/NEJMoa1716984.
5. Roboz, GJ, DiNardo, CD, Stein, EM, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2019;135(7), 463-471. doi: 10.1182/blood.2019002140
6. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731. doi:10.1182/blood-2017-04-779405.
7. Jan M, Majeti R. Clonal evolution of acute leukemia genomes. Oncogene. 2013;32(2):135-140. doi:10.1038/onc.2012.48.
8. Grove CS, Vassiliou GS. Acute myeloid leukaemia: a paradigm for the clonal evolution of cancer? Dis Model Mech. 2014;7(8):941-951. doi:10.1242/dmm.015974.
9. Anderson K, Lutz C, van Delft FW, et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature. 2011;469(7330):356-561. doi: 10.1038/nature09650.
10. Ding L, Ley TJ, Larson DE, et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature. 2012;481(7382):506-510. doi:10.1038/nature10738.
11. Terada K, Yamaguchi H, Ueki T, et al. Usefulness of BCOR gene mutation as a prognostic factor in acute myeloid leukemia with intermediate cytogenetic prognosis. Genes Chromosomes Cancer. 2018;57(8):401-408. doi:10.1002/gcc.22542.
12. Grossmann V, Tiacci E, Holmes AB, et al. Whole-exome sequencing identifies somatic mutations of BCOR in acute myeloid leukemia with normal karyotype. Blood. 2011;118(23):6153-6163. doi:10.1182/blood-2011-07-365320.
13. Parkin B, Ouillette P, Li Y, et al. Clonal evolution and devolution after chemotherapy in adult acute myelogenous leukemia. Blood. 2013;121(2):369-377. doi:10.1182/blood-2012-04-427039.
14. Papaemmanuil E, Gerstung M, Bullinger L, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374(23):2209-2221. doi:10.1056/NEJMoa1516192.
15. DiNardo CD, Ravandi F, Agresta S, et al. Characteristics, clinical outcome, and prognostic significance of IDH mutations in AML. Am J Hematol. 2015;90(8):732-736. doi:10.1002/ajh.24072.
16. Rakheja D, Konoplev S, Medeiros LJ, Chen W. IDH mutations in acute myeloid leukemia. Hum Pathol. 2012;43 (10):1541-1551. doi:10.1016/j.humpath.2012.05.003.
17. Lai C, Doucette K, Norsworthy K. Recent drug approvals for acute myeloid leukemia. J H Oncol. 2019;12(1):100. doi:10.1186/s13045-019-0774-x.
18. Boddu P, Takahashi K, Pemmaraju N, et al. Influence of IDH on FLT3-ITD status in newly diagnosed AML. Leukemia. 2017;31(11):2526-2529. doi:10.1038/leu.2017.244.
Nearly 20,000 patients are diagnosed with acute myeloid leukemia (AML) in the US annually.1 Despite the use of aggressive chemotherapeutic agents, the prognosis remains poor, with a mean 5-year survival of 28.3%.2 Fortunately, with the refinement of next-generation sequencing (NGS) hematology panels and development of systemic targeted therapies, the treatment landscape for eligible patients has improved, both in frontline and relapsed or refractory (R/R) patients.
Specifically, investigations into alterations within the FMS-like tyrosine kinase (FLT3) and isocitrate dehydrogenase (IDH) genes have led to the discovery of a number of targeted treatments. Midostaurin is US Food and Drug Administration (FDA)-approved for use in combination with induction chemotherapy for patients with internal tandem duplication of the FLT3 (FLT3-ITD) gene or mutations within the tyrosine kinase domain (FLT3-TKD).3 Ivosidenib is indicated for frontline treatment for those who are poor candidates for induction chemotherapy, and R/R patients who have an R132H mutation in IDH1.4,5 Enasidenib is FDA-approved for R/R patients with R140Q, R172S, and R172K mutations in IDH2.6
The optimal treatment for patients with AML with ≥ 2 clinically actionable mutations has not been established. In this article we describe a geriatric patient who initially was diagnosed with AML with concurrent FLT3-TKD and IDH1 mutations and received targeted, sequential management. We detail changes in disease phenotype and mutational status by repeating an NGS hematology panel and cytogenetic studies after each stage of therapy. Lastly, we discuss the clonal evolution apparent within leukemic cells with use of ≥ 1 or more targeted agents.
Case Presentation
A 68-year-old man presented to the Emergency Department at The Durham Veterans Affairs Medical Center in North Carolina with fatigue and light-headedness. Because of his symptoms and pancytopenia, a bone marrow aspiration and trephine biopsy were performed, which showed 57% myeloblasts, 12% promyelocytes/myelocytes, and 2% metamyelocytes in 20 to 30% cellular bone marrow. Flow cytometry confirmed a blast population consistent with AML. A LeukoVantage (Quest Diagnostics) hematologic NGS panel revealed the presence of FLT3-TKD, IDH1, RUNX1, BCOR-E1477, and SF3B1 mutations (Table). Initial fluorescence in situ hybridization (FISH) results showed a normal pattern of hybridization with no translocations. His disease was deemed to be intermediate-high risk because of the presence of FLT3-TKD and RUNX1 mutations, despite the normal cytogenetic profile and absence of additional clinical features.
Induction chemotherapy was started with idarubicin, 12 mg/m2, on days 1 to 3 and cytarabine, 200 mg/m2, on days 1 to 7. Because of the presence of a FLT3-TKD mutation, midostaurin was planned for days 8 to 21. After induction chemotherapy, a bone marrow biopsy on day 14 revealed an acellular marrow with no observed myeloblasts. A bone marrow biopsy conducted before initiating consolidation therapy, revealed 30% cellularity with morphologic remission. However, flow cytometry found 5% myeloblasts expressing CD34, CD117, CD13, CD38, and HLA-DR, consistent with measurable residual disease. He received 2 cycles of consolidation therapy with high-dose cytarabine combined with midostaurin. After the patient's second cycle of consolidation, he continued to experience transfusion-dependent cytopenias. Another bone marrow evaluation demonstrated 10% cellularity with nearly all cells appearing to be myeloblasts. A repeat LeukoVantage NGS panel demonstrated undetectable FLT3-TKD mutation and persistent IDH1-R123C mutation. FISH studies revealed a complex karyotype with monosomy of chromosomes 5 and 7 and trisomy of chromosome 8.
We discussed with the patient and his family the options available, which included initiating targeted therapy for his IDH1 mutation, administering hypomethylation therapy with or without venetoclax, or pursuing palliative measures. We collectively decided to pursue therapy with single-agent oral ivosidenib, 500 mg daily. After 1 month of treatment, our patient developed worsening fatigue. His white blood cell count had increased to > 43 k/cm2, raising concern for differentiation syndrome.
A review of the peripheral smear showed a wide-spectrum of maturing granulocytes, with a large percentage of blasts. Peripheral flow cytometry confirmed a blast population of 15%. After a short period of symptom improvement with steroids, the patient developed worsening confusion. Brain imaging identified 2 subdural hemorrhages. Because of a significant peripheral blast population and the development of these hemorrhages, palliative measures were pursued, and the patient was discharged to an inpatient hospice facility. A final NGS panel performed from peripheral blood detected mutations in IDH1, RUNX1, PTPN11, NRAS, BCOR-E1443, and SF3B1 genes.
Discussion
To our knowledge, this is the first reported case of a patient who sequentially received targeted treatments directed against both FLT3 and IDH1 mutations. Initial management with midostaurin and cytarabine resulted in sustained remission of his FLT3-TKD mutation. However, despite receiving prompt standard of care with combination induction chemotherapy and targeted therapy, the patient experienced unfavorable clonal evolution based upon his molecular and cytogenetic testing. Addition of ivosidenib as a second targeting agent for his IDH1 mutation did not achieve a second remission.
Clonal evolution is a well-described phenomenon in hematology. Indolent conditions, such as clonal hematopoiesis of intermediate potential, or malignancies, such as myelodysplastic syndromes and myeloproliferative neoplasms, could transform into acute leukemia through the accumulation of driver mutations and/or cytogenetic abnormalities. Clonal evolution often is viewed as the culprit in patients with AML whose disease relapses after remission with initial chemotherapy.7-10 With the increasing availability of commercial NGS panels designed to assess mutations among patients experiencing hematologic malignancies, patterns of relapse, and, models of clonal evolution could be observed closely in patients with AML.
We were able to monitor molecular changes within our patient’s predominant clonal populations by repeating peripheral comprehensive NGS panels after lines of targeted therapies. The repeated sequencing revealed that clones with FLT3-TKD mutations responded to midostaurin with first-line chemotherapy whereas it was unclear whether clones with IDH1 mutation responded to ivosidenib. Development of complex cytogenetic findings along with the clonal expansion of BCOR mutation-harboring cells likely contributed to our patient’s acutely worsening condition. Several studies have found that the presence of a BCOR mutation in adults with AML leads to lower overall survival and relapse-free survival.11,12 As of now, there are no treatments specifically targeting BCOR mutations.
Although there are novel targeting agents with proven efficacy for both FLT3 and IDH1 mutations (Figure), it is difficult to determine which pathogenic mutation drives disease onset. No evidence suggests that these drugs could be administered in tandem. At the present time, interest is directed towards targeting all AML subclones simultaneously, which could reduce the likelihood of evolution among founder clones.7,10,13 In their comparison between molecular profiles and outcomes of patients with AML, Papaemmanuil and colleagues observed that > 80% of patients with AML harbor ≥ 2 driver mutations concurrently.14 Moreover, FLT3-ITD and IDH1 mutations tend to co-occur in approximately 9 to 27% of AML cases.15-18 Available targeted agents for AML are relatively new and hematologists’ familiarity with these drugs is continuing to grow. As the number of novel agents increases, investigations directed toward assessing the safety profile and efficacy of combining targeted agents will be beneficial for patients with AML with ≥ 1 driver mutation.
Conclusions
For our patient with AML, sequential targeted management of FLT3-TKD and IDH1 mutations was not beneficial. Higher-risk disease features, such as the development of a complex karyotype, likely contributed to our patient’s poor response to second-line ivosidenib. The sequential NGS malignant hematology panels allowed us to closely monitor changes to the molecular structure of our patient’s AML after each line of targeted therapy. Future investigations of combining targeted agents for patients with AML with concurrent actionable mutations would provide insight into outcomes of treating multiple clonal populations simultaneously.
Nearly 20,000 patients are diagnosed with acute myeloid leukemia (AML) in the US annually.1 Despite the use of aggressive chemotherapeutic agents, the prognosis remains poor, with a mean 5-year survival of 28.3%.2 Fortunately, with the refinement of next-generation sequencing (NGS) hematology panels and development of systemic targeted therapies, the treatment landscape for eligible patients has improved, both in frontline and relapsed or refractory (R/R) patients.
Specifically, investigations into alterations within the FMS-like tyrosine kinase (FLT3) and isocitrate dehydrogenase (IDH) genes have led to the discovery of a number of targeted treatments. Midostaurin is US Food and Drug Administration (FDA)-approved for use in combination with induction chemotherapy for patients with internal tandem duplication of the FLT3 (FLT3-ITD) gene or mutations within the tyrosine kinase domain (FLT3-TKD).3 Ivosidenib is indicated for frontline treatment for those who are poor candidates for induction chemotherapy, and R/R patients who have an R132H mutation in IDH1.4,5 Enasidenib is FDA-approved for R/R patients with R140Q, R172S, and R172K mutations in IDH2.6
The optimal treatment for patients with AML with ≥ 2 clinically actionable mutations has not been established. In this article we describe a geriatric patient who initially was diagnosed with AML with concurrent FLT3-TKD and IDH1 mutations and received targeted, sequential management. We detail changes in disease phenotype and mutational status by repeating an NGS hematology panel and cytogenetic studies after each stage of therapy. Lastly, we discuss the clonal evolution apparent within leukemic cells with use of ≥ 1 or more targeted agents.
Case Presentation
A 68-year-old man presented to the Emergency Department at The Durham Veterans Affairs Medical Center in North Carolina with fatigue and light-headedness. Because of his symptoms and pancytopenia, a bone marrow aspiration and trephine biopsy were performed, which showed 57% myeloblasts, 12% promyelocytes/myelocytes, and 2% metamyelocytes in 20 to 30% cellular bone marrow. Flow cytometry confirmed a blast population consistent with AML. A LeukoVantage (Quest Diagnostics) hematologic NGS panel revealed the presence of FLT3-TKD, IDH1, RUNX1, BCOR-E1477, and SF3B1 mutations (Table). Initial fluorescence in situ hybridization (FISH) results showed a normal pattern of hybridization with no translocations. His disease was deemed to be intermediate-high risk because of the presence of FLT3-TKD and RUNX1 mutations, despite the normal cytogenetic profile and absence of additional clinical features.
Induction chemotherapy was started with idarubicin, 12 mg/m2, on days 1 to 3 and cytarabine, 200 mg/m2, on days 1 to 7. Because of the presence of a FLT3-TKD mutation, midostaurin was planned for days 8 to 21. After induction chemotherapy, a bone marrow biopsy on day 14 revealed an acellular marrow with no observed myeloblasts. A bone marrow biopsy conducted before initiating consolidation therapy, revealed 30% cellularity with morphologic remission. However, flow cytometry found 5% myeloblasts expressing CD34, CD117, CD13, CD38, and HLA-DR, consistent with measurable residual disease. He received 2 cycles of consolidation therapy with high-dose cytarabine combined with midostaurin. After the patient's second cycle of consolidation, he continued to experience transfusion-dependent cytopenias. Another bone marrow evaluation demonstrated 10% cellularity with nearly all cells appearing to be myeloblasts. A repeat LeukoVantage NGS panel demonstrated undetectable FLT3-TKD mutation and persistent IDH1-R123C mutation. FISH studies revealed a complex karyotype with monosomy of chromosomes 5 and 7 and trisomy of chromosome 8.
We discussed with the patient and his family the options available, which included initiating targeted therapy for his IDH1 mutation, administering hypomethylation therapy with or without venetoclax, or pursuing palliative measures. We collectively decided to pursue therapy with single-agent oral ivosidenib, 500 mg daily. After 1 month of treatment, our patient developed worsening fatigue. His white blood cell count had increased to > 43 k/cm2, raising concern for differentiation syndrome.
A review of the peripheral smear showed a wide-spectrum of maturing granulocytes, with a large percentage of blasts. Peripheral flow cytometry confirmed a blast population of 15%. After a short period of symptom improvement with steroids, the patient developed worsening confusion. Brain imaging identified 2 subdural hemorrhages. Because of a significant peripheral blast population and the development of these hemorrhages, palliative measures were pursued, and the patient was discharged to an inpatient hospice facility. A final NGS panel performed from peripheral blood detected mutations in IDH1, RUNX1, PTPN11, NRAS, BCOR-E1443, and SF3B1 genes.
Discussion
To our knowledge, this is the first reported case of a patient who sequentially received targeted treatments directed against both FLT3 and IDH1 mutations. Initial management with midostaurin and cytarabine resulted in sustained remission of his FLT3-TKD mutation. However, despite receiving prompt standard of care with combination induction chemotherapy and targeted therapy, the patient experienced unfavorable clonal evolution based upon his molecular and cytogenetic testing. Addition of ivosidenib as a second targeting agent for his IDH1 mutation did not achieve a second remission.
Clonal evolution is a well-described phenomenon in hematology. Indolent conditions, such as clonal hematopoiesis of intermediate potential, or malignancies, such as myelodysplastic syndromes and myeloproliferative neoplasms, could transform into acute leukemia through the accumulation of driver mutations and/or cytogenetic abnormalities. Clonal evolution often is viewed as the culprit in patients with AML whose disease relapses after remission with initial chemotherapy.7-10 With the increasing availability of commercial NGS panels designed to assess mutations among patients experiencing hematologic malignancies, patterns of relapse, and, models of clonal evolution could be observed closely in patients with AML.
We were able to monitor molecular changes within our patient’s predominant clonal populations by repeating peripheral comprehensive NGS panels after lines of targeted therapies. The repeated sequencing revealed that clones with FLT3-TKD mutations responded to midostaurin with first-line chemotherapy whereas it was unclear whether clones with IDH1 mutation responded to ivosidenib. Development of complex cytogenetic findings along with the clonal expansion of BCOR mutation-harboring cells likely contributed to our patient’s acutely worsening condition. Several studies have found that the presence of a BCOR mutation in adults with AML leads to lower overall survival and relapse-free survival.11,12 As of now, there are no treatments specifically targeting BCOR mutations.
Although there are novel targeting agents with proven efficacy for both FLT3 and IDH1 mutations (Figure), it is difficult to determine which pathogenic mutation drives disease onset. No evidence suggests that these drugs could be administered in tandem. At the present time, interest is directed towards targeting all AML subclones simultaneously, which could reduce the likelihood of evolution among founder clones.7,10,13 In their comparison between molecular profiles and outcomes of patients with AML, Papaemmanuil and colleagues observed that > 80% of patients with AML harbor ≥ 2 driver mutations concurrently.14 Moreover, FLT3-ITD and IDH1 mutations tend to co-occur in approximately 9 to 27% of AML cases.15-18 Available targeted agents for AML are relatively new and hematologists’ familiarity with these drugs is continuing to grow. As the number of novel agents increases, investigations directed toward assessing the safety profile and efficacy of combining targeted agents will be beneficial for patients with AML with ≥ 1 driver mutation.
Conclusions
For our patient with AML, sequential targeted management of FLT3-TKD and IDH1 mutations was not beneficial. Higher-risk disease features, such as the development of a complex karyotype, likely contributed to our patient’s poor response to second-line ivosidenib. The sequential NGS malignant hematology panels allowed us to closely monitor changes to the molecular structure of our patient’s AML after each line of targeted therapy. Future investigations of combining targeted agents for patients with AML with concurrent actionable mutations would provide insight into outcomes of treating multiple clonal populations simultaneously.
1. De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: a comprehensive review and 2016 update. Blood Cancer J. 2016;6(7):e441. doi:10.1038/bcj.2016.50.
2. National Cancer Institute. Cancer Stat Facts: Leukemia — acute myeloid leukemia (AML). Accessed November 4, 2020. https://seer.cancer.gov/statfacts/html/amyl.html
3. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464. doi:10.1056/NEJMoa1614359.
4. DiNardo CD, Stein EM, de Botton S, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378(25):2386-2398. doi:10.1056/NEJMoa1716984.
5. Roboz, GJ, DiNardo, CD, Stein, EM, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2019;135(7), 463-471. doi: 10.1182/blood.2019002140
6. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731. doi:10.1182/blood-2017-04-779405.
7. Jan M, Majeti R. Clonal evolution of acute leukemia genomes. Oncogene. 2013;32(2):135-140. doi:10.1038/onc.2012.48.
8. Grove CS, Vassiliou GS. Acute myeloid leukaemia: a paradigm for the clonal evolution of cancer? Dis Model Mech. 2014;7(8):941-951. doi:10.1242/dmm.015974.
9. Anderson K, Lutz C, van Delft FW, et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature. 2011;469(7330):356-561. doi: 10.1038/nature09650.
10. Ding L, Ley TJ, Larson DE, et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature. 2012;481(7382):506-510. doi:10.1038/nature10738.
11. Terada K, Yamaguchi H, Ueki T, et al. Usefulness of BCOR gene mutation as a prognostic factor in acute myeloid leukemia with intermediate cytogenetic prognosis. Genes Chromosomes Cancer. 2018;57(8):401-408. doi:10.1002/gcc.22542.
12. Grossmann V, Tiacci E, Holmes AB, et al. Whole-exome sequencing identifies somatic mutations of BCOR in acute myeloid leukemia with normal karyotype. Blood. 2011;118(23):6153-6163. doi:10.1182/blood-2011-07-365320.
13. Parkin B, Ouillette P, Li Y, et al. Clonal evolution and devolution after chemotherapy in adult acute myelogenous leukemia. Blood. 2013;121(2):369-377. doi:10.1182/blood-2012-04-427039.
14. Papaemmanuil E, Gerstung M, Bullinger L, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374(23):2209-2221. doi:10.1056/NEJMoa1516192.
15. DiNardo CD, Ravandi F, Agresta S, et al. Characteristics, clinical outcome, and prognostic significance of IDH mutations in AML. Am J Hematol. 2015;90(8):732-736. doi:10.1002/ajh.24072.
16. Rakheja D, Konoplev S, Medeiros LJ, Chen W. IDH mutations in acute myeloid leukemia. Hum Pathol. 2012;43 (10):1541-1551. doi:10.1016/j.humpath.2012.05.003.
17. Lai C, Doucette K, Norsworthy K. Recent drug approvals for acute myeloid leukemia. J H Oncol. 2019;12(1):100. doi:10.1186/s13045-019-0774-x.
18. Boddu P, Takahashi K, Pemmaraju N, et al. Influence of IDH on FLT3-ITD status in newly diagnosed AML. Leukemia. 2017;31(11):2526-2529. doi:10.1038/leu.2017.244.
1. De Kouchkovsky I, Abdul-Hay M. Acute myeloid leukemia: a comprehensive review and 2016 update. Blood Cancer J. 2016;6(7):e441. doi:10.1038/bcj.2016.50.
2. National Cancer Institute. Cancer Stat Facts: Leukemia — acute myeloid leukemia (AML). Accessed November 4, 2020. https://seer.cancer.gov/statfacts/html/amyl.html
3. Stone RM, Mandrekar SJ, Sanford BL, et al. Midostaurin plus chemotherapy for acute myeloid leukemia with a FLT3 mutation. N Engl J Med. 2017;377(5):454-464. doi:10.1056/NEJMoa1614359.
4. DiNardo CD, Stein EM, de Botton S, et al. Durable remissions with ivosidenib in IDH1-mutated relapsed or refractory AML. N Engl J Med. 2018;378(25):2386-2398. doi:10.1056/NEJMoa1716984.
5. Roboz, GJ, DiNardo, CD, Stein, EM, et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood. 2019;135(7), 463-471. doi: 10.1182/blood.2019002140
6. Stein EM, DiNardo CD, Pollyea DA, et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood. 2017;130(6):722-731. doi:10.1182/blood-2017-04-779405.
7. Jan M, Majeti R. Clonal evolution of acute leukemia genomes. Oncogene. 2013;32(2):135-140. doi:10.1038/onc.2012.48.
8. Grove CS, Vassiliou GS. Acute myeloid leukaemia: a paradigm for the clonal evolution of cancer? Dis Model Mech. 2014;7(8):941-951. doi:10.1242/dmm.015974.
9. Anderson K, Lutz C, van Delft FW, et al. Genetic variegation of clonal architecture and propagating cells in leukaemia. Nature. 2011;469(7330):356-561. doi: 10.1038/nature09650.
10. Ding L, Ley TJ, Larson DE, et al. Clonal evolution in relapsed acute myeloid leukaemia revealed by whole-genome sequencing. Nature. 2012;481(7382):506-510. doi:10.1038/nature10738.
11. Terada K, Yamaguchi H, Ueki T, et al. Usefulness of BCOR gene mutation as a prognostic factor in acute myeloid leukemia with intermediate cytogenetic prognosis. Genes Chromosomes Cancer. 2018;57(8):401-408. doi:10.1002/gcc.22542.
12. Grossmann V, Tiacci E, Holmes AB, et al. Whole-exome sequencing identifies somatic mutations of BCOR in acute myeloid leukemia with normal karyotype. Blood. 2011;118(23):6153-6163. doi:10.1182/blood-2011-07-365320.
13. Parkin B, Ouillette P, Li Y, et al. Clonal evolution and devolution after chemotherapy in adult acute myelogenous leukemia. Blood. 2013;121(2):369-377. doi:10.1182/blood-2012-04-427039.
14. Papaemmanuil E, Gerstung M, Bullinger L, et al. Genomic classification and prognosis in acute myeloid leukemia. N Engl J Med. 2016;374(23):2209-2221. doi:10.1056/NEJMoa1516192.
15. DiNardo CD, Ravandi F, Agresta S, et al. Characteristics, clinical outcome, and prognostic significance of IDH mutations in AML. Am J Hematol. 2015;90(8):732-736. doi:10.1002/ajh.24072.
16. Rakheja D, Konoplev S, Medeiros LJ, Chen W. IDH mutations in acute myeloid leukemia. Hum Pathol. 2012;43 (10):1541-1551. doi:10.1016/j.humpath.2012.05.003.
17. Lai C, Doucette K, Norsworthy K. Recent drug approvals for acute myeloid leukemia. J H Oncol. 2019;12(1):100. doi:10.1186/s13045-019-0774-x.
18. Boddu P, Takahashi K, Pemmaraju N, et al. Influence of IDH on FLT3-ITD status in newly diagnosed AML. Leukemia. 2017;31(11):2526-2529. doi:10.1038/leu.2017.244.
Sequential Targeted Treatment of an Elderly Patient With Acute Myeloid Leukemia Harboring Concurrent FLT3-TKD and IDH1 Mutations: A Case Report
INTRODUCTION: With the increasing availability of novel targeted therapies and next-generation sequencing (NGS) hematology panels, the treatment paradigm for patients with acute myeloid leukemia (AML) has recently been altered. Specifically, patients who bear mutations within the FMS-like tyrosine kinase (FLT3) gene or the isocitrate dehydrogenase (IDH) 1 or IDH2 genes may now be candidates for targeted treatments either in the frontline or relapsed or refractory (R/R) settings. The sequential targeted approach to AML patients who harbor mutations within both FLT3 and IDH genes has yet to be elucidated.
CASE PRESENTATION: Herein, we report a case of an elderly patient with FLT3 and IDH1 mutations who underwent induction chemotherapy in combination with midostaurin, and subsequently, ivosidenib in the R/R setting. Clonal evaluation was demonstrated with repeated cytogenetic analysis and NGS of blood and bone marrow specimens. At diagnosis, the patient’s AML harbored several pathogenic gene variants, including FLT3 and IDH1 mutations. Following induction chemotherapy with midostaurin, the patient’s FLT3 mutation was no longer detected. Upon relapse, the FLT3 mutation was still undetectable, however the IDH1 mutation remained. Unfortunately, the patient’s AML did not respond to ivosidenib, and expansion of a leukemic clone with a BCOR mutation was observed.
CONCLUSION: This case conveys the use of multiple targeted therapies in a sequential fashion for an AML patient with frequent completion of NGS panels to monitor clonal evolution. Given that a considerable minority of patients harbor both FLT3 and IDH mutations, further investigations evaluating optimal sequencing or combinations of targeted therapies are required.
INTRODUCTION: With the increasing availability of novel targeted therapies and next-generation sequencing (NGS) hematology panels, the treatment paradigm for patients with acute myeloid leukemia (AML) has recently been altered. Specifically, patients who bear mutations within the FMS-like tyrosine kinase (FLT3) gene or the isocitrate dehydrogenase (IDH) 1 or IDH2 genes may now be candidates for targeted treatments either in the frontline or relapsed or refractory (R/R) settings. The sequential targeted approach to AML patients who harbor mutations within both FLT3 and IDH genes has yet to be elucidated.
CASE PRESENTATION: Herein, we report a case of an elderly patient with FLT3 and IDH1 mutations who underwent induction chemotherapy in combination with midostaurin, and subsequently, ivosidenib in the R/R setting. Clonal evaluation was demonstrated with repeated cytogenetic analysis and NGS of blood and bone marrow specimens. At diagnosis, the patient’s AML harbored several pathogenic gene variants, including FLT3 and IDH1 mutations. Following induction chemotherapy with midostaurin, the patient’s FLT3 mutation was no longer detected. Upon relapse, the FLT3 mutation was still undetectable, however the IDH1 mutation remained. Unfortunately, the patient’s AML did not respond to ivosidenib, and expansion of a leukemic clone with a BCOR mutation was observed.
CONCLUSION: This case conveys the use of multiple targeted therapies in a sequential fashion for an AML patient with frequent completion of NGS panels to monitor clonal evolution. Given that a considerable minority of patients harbor both FLT3 and IDH mutations, further investigations evaluating optimal sequencing or combinations of targeted therapies are required.
INTRODUCTION: With the increasing availability of novel targeted therapies and next-generation sequencing (NGS) hematology panels, the treatment paradigm for patients with acute myeloid leukemia (AML) has recently been altered. Specifically, patients who bear mutations within the FMS-like tyrosine kinase (FLT3) gene or the isocitrate dehydrogenase (IDH) 1 or IDH2 genes may now be candidates for targeted treatments either in the frontline or relapsed or refractory (R/R) settings. The sequential targeted approach to AML patients who harbor mutations within both FLT3 and IDH genes has yet to be elucidated.
CASE PRESENTATION: Herein, we report a case of an elderly patient with FLT3 and IDH1 mutations who underwent induction chemotherapy in combination with midostaurin, and subsequently, ivosidenib in the R/R setting. Clonal evaluation was demonstrated with repeated cytogenetic analysis and NGS of blood and bone marrow specimens. At diagnosis, the patient’s AML harbored several pathogenic gene variants, including FLT3 and IDH1 mutations. Following induction chemotherapy with midostaurin, the patient’s FLT3 mutation was no longer detected. Upon relapse, the FLT3 mutation was still undetectable, however the IDH1 mutation remained. Unfortunately, the patient’s AML did not respond to ivosidenib, and expansion of a leukemic clone with a BCOR mutation was observed.
CONCLUSION: This case conveys the use of multiple targeted therapies in a sequential fashion for an AML patient with frequent completion of NGS panels to monitor clonal evolution. Given that a considerable minority of patients harbor both FLT3 and IDH mutations, further investigations evaluating optimal sequencing or combinations of targeted therapies are required.