User login
Abdominal Pain and Fever 48 Hours After Hysterosalpingography
A 37-year-old woman presented to the emergency department (ED) with 12 hours of fever and lower abdominal cramp pain. She had a history significant for hypothyroidism, infertility, and dysmenorrhea and had a hysterosalpingography (HSG) 48 hours prior for a comprehensive infertility workup.
On examination, the patient’s vital signs were a 94 bpm heart rate; 109/64 mm Hg blood pressure; 14 breaths per minute respiratory rate; 99% oxygen saturation on room air; and 101.2 °F temperature. The patient reported pain in the bilateral lower abdominal quadrants and no history of sexually transmitted infection, pelvic inflammatory disease, vaginal discharge or bleeding, dysuria, hematuria, melena, or bright red blood per rectum. A human chorionic gonadotropin urine test was negative on intake. The HSG 48 hours prior showed no concerning findings with normal uterine cavity and normal caliber fallopian tubes bilaterally.
On physical examination, the patient’s abdomen was nondistended, nonperitonitic, and without evidence of acute trauma or surgical scars. On palpation, the patient was tender in her suprapubic region and lower abdominal quadrants without evidence of guarding or rebound
The patient’s initial laboratory tests were as follows: white blood cells, 20.1 × 103/μL (reference range, 4-11 × 103/μL); hemoglobin, 12.1 g/dL (reference range, 12.1-15.1 g/dL); hematocrit, 37.1%, (reference range, 36%-48%); alanine aminotransferase, 84 U/L (reference range, 7-56 U/L); aspartate aminotransferase, 66 U/L (reference range, 8-33 U/L); and lipase, 25 U/L (5-60 U/L). Urinalysis was notable for only 5 red blood cells and negative for white blood cells, leukocyte esterase, and nitrites. The patient’s pain and fever were controlled with 1 g IV acetaminophen.
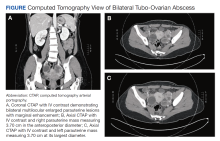
The patient’s fever, leukocytosis, and physical examination were concerning for possible intra-abdominal processes, so a
Discussion
The patient was diagnosed with bilateral tubo-ovarian abscess (TOA) likely secondary to her HSG procedure 48 hours before. TOA is a severe infectious, inflammatory condition involving a mass of the ovaries, fallopian tubes, or adjacent tissues of the upper female genital tract.1 Traditionally, TOAs are sequelae of undiagnosed or subclinical acute or chronic pelvic inflammatory disease (PID). This is known to occur via pathogen ascension from the lower to the upper female genital tract resulting in cervicitis, endometritis, salpingitis, oophoritis, and if left untreated, peritonitis.1 About 70,000 women are diagnosed with TOAs in the US every year. These patients require hospitalization as well as IV antibiotics for gold-standard treatment; however, some cases may require percutaneous drainage based on size, severity, and location.2
Diagnostic Considerations
Clinically, patients with TOAs present with fever, chills, lower abdominal pain, vaginal discharge with cervical motion tenderness, and an adnexal mass on examination.3 When a TOA is suspected, a urine human chorionic gonadotropin test and testing for C trachomatis and N gonorrhoeae are warranted. An ED workup often reveals leukocytosis, elevated C-reactive protein, and elevated erythrocyte sedimentation rate. Imaging is recommended once a TOA is suspected. Ultrasound is the gold-standard imaging modality and boasts a sensitivity of 93% and specificity of 98% for the detection of TOAs; however, CT has also been shown to be an effective diagnostic modality.4
Despite being common, TOAs are difficult to predict, detect, and diagnose; thus the clinician must often rely on thorough history taking and physical examination to raise suspicion.5 Although most frequently associated with sexual transmission, TOAs occur in not sexually active women in adolescence and adulthood. Specifically, TOAs also can present secondary to other intra-abdominal pathologies, such as appendicitis, diverticulitis, and pyelonephritis, as well as a complication of intrauterine procedures, such as an HSG, or less commonly, following intrauterine device (IUD) insertion.5-7
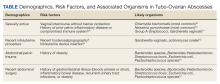
Given that sexually transmitted infections are the most common etiology of TOAs, C trachomatis and N gonorrhoeae are the most likely microorganisms to be isolated (Table).8-12 In not sexually active populations, Escherichia coli and Gardnerella vaginalis should be considered instead. Although rare, women with IUDs have been shown to have an increased incidence of PID/TOA secondary to Actinomyces israleii relative to women without IUDs.12 In patients with TOAs secondary to intraabdominal surgery, anaerobic bacteria, such as Bacterioides and Peptostreptococcus species in addition to Escherichia coli, are likely culprits.11
In patients after HSG, infectious complications are uncommon enough that the American College of Obstetricians and Gynecologists recommends against antibiotic prophylaxis unless there are risk factors of dilated fallopian tubes or a history of PID.13 Identifying a precise percentage of TOA as a complication of HSG is rather elusive in the literature, though, it is frequently noted that infection in general is uncommon, and the risk of developing PID is about 1.4% to 3.4%.14 However, in the retrospective study most often cited, all women who developed PID following HSG had evidence of dilated fallopian tubes. Given our patient had no history of PID or dilated fallopian tubes, her risk of developing infection (PID or postprocedural abscess) would be considered very low; therefore, the index of suspicion also was low.15
Following diagnosis, the patient was promptly admitted and treated with a course of IV ceftriaxone 1 g every 24 hours, IV doxycycline 100 mg every 12 hours, and a single dose of IV metronidazole 500 mg. Her leukocytosis, fever, and pain improved within 48 hours without the need for percutaneous drainage and the patient made a complete recovery.
Complications
TOAs can carry significant morbidity and mortality, and there are both acute and chronic complications associated. Even if properly treated, TOAs can rupture leading to severe illness, such as peritonitis and septic shock. This often requires surgical intervention and hemodynamic pressure support in the intensive care unit setting.16 One of the most feared long-term complications of TOAs is infertility secondary to structural abnormalities of the female reproductive tract.10 Adhesions, strictures, and scarring are associated with TOAs irrespective of medical or surgical management, and thus any women with a history of PID or TOA require advanced fertility workup if they are having difficulties with conception or implantation.17
Minimizing infectious transmission also is essential in the treatment of TOA/PID. All women who receive a diagnosis of PID or TOA should be evaluated for gonorrhea, chlamydia, HIV, and syphilis. Women should be instructed to abstain from sexual intercourse until therapy is complete, symptoms have resolved, and sex partners have been treated for potential chlamydial or gonococcal infections. All contraceptive methods can be continued during treatment.
Conclusions
This case presented several challenges as the bilateral TOAs developed postprocedure in a patient without risk factors. Furthermore, this case did not follow the classic presentation of ascending bacterial translocation to the ovaries over days to weeks. The diagnosis was complicated by a largely benign physical examination and a pelvic examination without evidence of abnormal vaginal discharge or cervicitis. The only indicators were fever, leukocytosis, and abdominal pain in the setting of a recent, uncomplicated HSG procedure. Vigilance is required to obtain the necessary history, and the differential of TOA must be broadened to include women without a history or symptoms of a sexually transmitted infection (contrary to the classic association). We aim to encourage heightened clinical suspicion for TOAs in patients who present with fever, leukocytosis, and abdominal pain after recent HSG or other intrauterine instrumentation procedures and therefore improve patient outcomes.
1. Gkrozou F, Tsonis O, Daniilidis A, Navrozoglou I, Paschopoulos M. Tubo-ovarian abscess: exploring optimal treatment options based on current evidence. J Endometr Pelvic Pain Disord. 2020;13(1):10-19. doi:10.1177/2284026520960649
2. Taylor KJ, Wasson JF, De Graaff C, Rosenfield AT, Andriole VT. Accuracy of grey-scale ultrasound diagnosis of abdominal and pelvic abscesses in 220 patients. Lancet. 1978;1(8055):83-84. doi:10.1016/s0140-6736(78)90016-8
3. Bridwell RE, Koyfman A, Long B. High risk and low prevalence diseases: tubo-ovarian abscess. Am J Emerg Med. 2022;57:70-75. doi:10.1016/j.ajem.2022.04.026
4. Lambert MJ, Villa M. Gynecologic ultrasound in emergency medicine. Emerg Med Clin North Am. 2004;22(3):683-696. doi:10.1016/j.emc.2004.04.016
5. Munro K, Gharaibeh A, Nagabushanam S, Martin C. Diagnosis and management of tubo-ovarian abscesses. Obstet Gynaecol. 2018;20(1):11-19. doi:10.1111/tog.12447
6. Fink D, Lim PPC, Desai A, Stephans AB, Wien MA. Recurrent tubo-ovarian abscess in a nonsexually active adolescent. Consultant. 2022;62(1):e26-e28. doi:10.25270/con.2021.04.00010
7. Hiller N, Fux T, Finkelstein A, Mezeh H, Simanovsky N. CT differentiation between tubo-ovarian and appendiceal origin of right lower quadrant abscess: CT, clinical, and laboratory correlation. Emerg Radiol. 2016;23:133-139. doi:10.1007/s10140-015-1372-z
8. Kairys N, Roepke C. Tubo-ovarian abscess. In: StatPearls. Treasure Island (FL): StatPearls Publishing; June 12, 2023.
9. Gao Y, Qu P, Zhou Y, Ding W. Risk factors for the development of tubo-ovarian abscesses in women with ovarian endometriosis: a retrospective matched case-control study. BMC Womens Health. 2021:21:43. doi:10.1186/s12905-021-01188-6
10. Curry A, Williams T, Penny ML. Pelvic inflammatory disease: diagnosis, management, and prevention. Am Fam Physician. 2019;100(6):357-364.
11. Landers DV, Sweet RL. Tubo-ovarian abscess: contemporary approach to management. Rev Infect Dis. 1983;5(5):876-884. doi:10.1093/clinids/5.5.876
12. Burkman R, Schlesselman S, McCaffrey L, Gupta PK, Spence M. The relationship of genital tract actinomycetes and the development of pelvic inflammatory disease. Am J Obstet Gynecol. 1982;143(5):585-589. doi:10.1016/0002-9378(82)90552-x
13. Armstrong C. ACOG releases guidelines on antibiotic prophylaxis for gynecologic procedures. Am Fam Physician. 2007;75(7):1094-1096.
14. Pittaway DE, Winfield AC, Maxson W, Daniell J, Herbert C, Wentz AC. Prevention of acute pelvic inflammatory disease after hysterosalpingography: efficacy of doxycycline prophylaxis. Am J Obstet Gynecol. 1983;147(6):623-626. doi:10.1016/0002-9378(83)90438-6
15. Committee on Practice Bulletins—Gynecology. Prevention of Infection After Gynecologic Procedures: ACOG Practice Bulletin, Number 195. Obstet Gynecol. 2018;131(6):e172-e189. doi:10.1097/AOG.0000000000002670
16. Tao X, Ge SQ, Chen L, Cai LS, Hwang MF, Wang CL. Relationships between female infertility and female genital infections and pelvic inflammatory disease: a population-based nested controlled study. Clinics (Sao Paulo). 2018;73:e364. Published 2018 Aug 9. doi:10.6061/clinics/2018/e364
17. Fouks Y, Azem F, Many A, Cohen Y, Levin I, Cohen A. Fertility outcomes in patients with tubo-ovarian abscesses after an oocyte retrieval: a longitudinal cohort analysis. Arch Gynecol Obstet. 2019;300(3):763-769. doi:10.1007/s00404-019-05230-9
A 37-year-old woman presented to the emergency department (ED) with 12 hours of fever and lower abdominal cramp pain. She had a history significant for hypothyroidism, infertility, and dysmenorrhea and had a hysterosalpingography (HSG) 48 hours prior for a comprehensive infertility workup.
On examination, the patient’s vital signs were a 94 bpm heart rate; 109/64 mm Hg blood pressure; 14 breaths per minute respiratory rate; 99% oxygen saturation on room air; and 101.2 °F temperature. The patient reported pain in the bilateral lower abdominal quadrants and no history of sexually transmitted infection, pelvic inflammatory disease, vaginal discharge or bleeding, dysuria, hematuria, melena, or bright red blood per rectum. A human chorionic gonadotropin urine test was negative on intake. The HSG 48 hours prior showed no concerning findings with normal uterine cavity and normal caliber fallopian tubes bilaterally.
On physical examination, the patient’s abdomen was nondistended, nonperitonitic, and without evidence of acute trauma or surgical scars. On palpation, the patient was tender in her suprapubic region and lower abdominal quadrants without evidence of guarding or rebound
The patient’s initial laboratory tests were as follows: white blood cells, 20.1 × 103/μL (reference range, 4-11 × 103/μL); hemoglobin, 12.1 g/dL (reference range, 12.1-15.1 g/dL); hematocrit, 37.1%, (reference range, 36%-48%); alanine aminotransferase, 84 U/L (reference range, 7-56 U/L); aspartate aminotransferase, 66 U/L (reference range, 8-33 U/L); and lipase, 25 U/L (5-60 U/L). Urinalysis was notable for only 5 red blood cells and negative for white blood cells, leukocyte esterase, and nitrites. The patient’s pain and fever were controlled with 1 g IV acetaminophen.
The patient’s fever, leukocytosis, and physical examination were concerning for possible intra-abdominal processes, so a
Discussion
The patient was diagnosed with bilateral tubo-ovarian abscess (TOA) likely secondary to her HSG procedure 48 hours before. TOA is a severe infectious, inflammatory condition involving a mass of the ovaries, fallopian tubes, or adjacent tissues of the upper female genital tract.1 Traditionally, TOAs are sequelae of undiagnosed or subclinical acute or chronic pelvic inflammatory disease (PID). This is known to occur via pathogen ascension from the lower to the upper female genital tract resulting in cervicitis, endometritis, salpingitis, oophoritis, and if left untreated, peritonitis.1 About 70,000 women are diagnosed with TOAs in the US every year. These patients require hospitalization as well as IV antibiotics for gold-standard treatment; however, some cases may require percutaneous drainage based on size, severity, and location.2
Diagnostic Considerations
Clinically, patients with TOAs present with fever, chills, lower abdominal pain, vaginal discharge with cervical motion tenderness, and an adnexal mass on examination.3 When a TOA is suspected, a urine human chorionic gonadotropin test and testing for C trachomatis and N gonorrhoeae are warranted. An ED workup often reveals leukocytosis, elevated C-reactive protein, and elevated erythrocyte sedimentation rate. Imaging is recommended once a TOA is suspected. Ultrasound is the gold-standard imaging modality and boasts a sensitivity of 93% and specificity of 98% for the detection of TOAs; however, CT has also been shown to be an effective diagnostic modality.4
Despite being common, TOAs are difficult to predict, detect, and diagnose; thus the clinician must often rely on thorough history taking and physical examination to raise suspicion.5 Although most frequently associated with sexual transmission, TOAs occur in not sexually active women in adolescence and adulthood. Specifically, TOAs also can present secondary to other intra-abdominal pathologies, such as appendicitis, diverticulitis, and pyelonephritis, as well as a complication of intrauterine procedures, such as an HSG, or less commonly, following intrauterine device (IUD) insertion.5-7
Given that sexually transmitted infections are the most common etiology of TOAs, C trachomatis and N gonorrhoeae are the most likely microorganisms to be isolated (Table).8-12 In not sexually active populations, Escherichia coli and Gardnerella vaginalis should be considered instead. Although rare, women with IUDs have been shown to have an increased incidence of PID/TOA secondary to Actinomyces israleii relative to women without IUDs.12 In patients with TOAs secondary to intraabdominal surgery, anaerobic bacteria, such as Bacterioides and Peptostreptococcus species in addition to Escherichia coli, are likely culprits.11
In patients after HSG, infectious complications are uncommon enough that the American College of Obstetricians and Gynecologists recommends against antibiotic prophylaxis unless there are risk factors of dilated fallopian tubes or a history of PID.13 Identifying a precise percentage of TOA as a complication of HSG is rather elusive in the literature, though, it is frequently noted that infection in general is uncommon, and the risk of developing PID is about 1.4% to 3.4%.14 However, in the retrospective study most often cited, all women who developed PID following HSG had evidence of dilated fallopian tubes. Given our patient had no history of PID or dilated fallopian tubes, her risk of developing infection (PID or postprocedural abscess) would be considered very low; therefore, the index of suspicion also was low.15
Following diagnosis, the patient was promptly admitted and treated with a course of IV ceftriaxone 1 g every 24 hours, IV doxycycline 100 mg every 12 hours, and a single dose of IV metronidazole 500 mg. Her leukocytosis, fever, and pain improved within 48 hours without the need for percutaneous drainage and the patient made a complete recovery.
Complications
TOAs can carry significant morbidity and mortality, and there are both acute and chronic complications associated. Even if properly treated, TOAs can rupture leading to severe illness, such as peritonitis and septic shock. This often requires surgical intervention and hemodynamic pressure support in the intensive care unit setting.16 One of the most feared long-term complications of TOAs is infertility secondary to structural abnormalities of the female reproductive tract.10 Adhesions, strictures, and scarring are associated with TOAs irrespective of medical or surgical management, and thus any women with a history of PID or TOA require advanced fertility workup if they are having difficulties with conception or implantation.17
Minimizing infectious transmission also is essential in the treatment of TOA/PID. All women who receive a diagnosis of PID or TOA should be evaluated for gonorrhea, chlamydia, HIV, and syphilis. Women should be instructed to abstain from sexual intercourse until therapy is complete, symptoms have resolved, and sex partners have been treated for potential chlamydial or gonococcal infections. All contraceptive methods can be continued during treatment.
Conclusions
This case presented several challenges as the bilateral TOAs developed postprocedure in a patient without risk factors. Furthermore, this case did not follow the classic presentation of ascending bacterial translocation to the ovaries over days to weeks. The diagnosis was complicated by a largely benign physical examination and a pelvic examination without evidence of abnormal vaginal discharge or cervicitis. The only indicators were fever, leukocytosis, and abdominal pain in the setting of a recent, uncomplicated HSG procedure. Vigilance is required to obtain the necessary history, and the differential of TOA must be broadened to include women without a history or symptoms of a sexually transmitted infection (contrary to the classic association). We aim to encourage heightened clinical suspicion for TOAs in patients who present with fever, leukocytosis, and abdominal pain after recent HSG or other intrauterine instrumentation procedures and therefore improve patient outcomes.
A 37-year-old woman presented to the emergency department (ED) with 12 hours of fever and lower abdominal cramp pain. She had a history significant for hypothyroidism, infertility, and dysmenorrhea and had a hysterosalpingography (HSG) 48 hours prior for a comprehensive infertility workup.
On examination, the patient’s vital signs were a 94 bpm heart rate; 109/64 mm Hg blood pressure; 14 breaths per minute respiratory rate; 99% oxygen saturation on room air; and 101.2 °F temperature. The patient reported pain in the bilateral lower abdominal quadrants and no history of sexually transmitted infection, pelvic inflammatory disease, vaginal discharge or bleeding, dysuria, hematuria, melena, or bright red blood per rectum. A human chorionic gonadotropin urine test was negative on intake. The HSG 48 hours prior showed no concerning findings with normal uterine cavity and normal caliber fallopian tubes bilaterally.
On physical examination, the patient’s abdomen was nondistended, nonperitonitic, and without evidence of acute trauma or surgical scars. On palpation, the patient was tender in her suprapubic region and lower abdominal quadrants without evidence of guarding or rebound
The patient’s initial laboratory tests were as follows: white blood cells, 20.1 × 103/μL (reference range, 4-11 × 103/μL); hemoglobin, 12.1 g/dL (reference range, 12.1-15.1 g/dL); hematocrit, 37.1%, (reference range, 36%-48%); alanine aminotransferase, 84 U/L (reference range, 7-56 U/L); aspartate aminotransferase, 66 U/L (reference range, 8-33 U/L); and lipase, 25 U/L (5-60 U/L). Urinalysis was notable for only 5 red blood cells and negative for white blood cells, leukocyte esterase, and nitrites. The patient’s pain and fever were controlled with 1 g IV acetaminophen.
The patient’s fever, leukocytosis, and physical examination were concerning for possible intra-abdominal processes, so a
Discussion
The patient was diagnosed with bilateral tubo-ovarian abscess (TOA) likely secondary to her HSG procedure 48 hours before. TOA is a severe infectious, inflammatory condition involving a mass of the ovaries, fallopian tubes, or adjacent tissues of the upper female genital tract.1 Traditionally, TOAs are sequelae of undiagnosed or subclinical acute or chronic pelvic inflammatory disease (PID). This is known to occur via pathogen ascension from the lower to the upper female genital tract resulting in cervicitis, endometritis, salpingitis, oophoritis, and if left untreated, peritonitis.1 About 70,000 women are diagnosed with TOAs in the US every year. These patients require hospitalization as well as IV antibiotics for gold-standard treatment; however, some cases may require percutaneous drainage based on size, severity, and location.2
Diagnostic Considerations
Clinically, patients with TOAs present with fever, chills, lower abdominal pain, vaginal discharge with cervical motion tenderness, and an adnexal mass on examination.3 When a TOA is suspected, a urine human chorionic gonadotropin test and testing for C trachomatis and N gonorrhoeae are warranted. An ED workup often reveals leukocytosis, elevated C-reactive protein, and elevated erythrocyte sedimentation rate. Imaging is recommended once a TOA is suspected. Ultrasound is the gold-standard imaging modality and boasts a sensitivity of 93% and specificity of 98% for the detection of TOAs; however, CT has also been shown to be an effective diagnostic modality.4
Despite being common, TOAs are difficult to predict, detect, and diagnose; thus the clinician must often rely on thorough history taking and physical examination to raise suspicion.5 Although most frequently associated with sexual transmission, TOAs occur in not sexually active women in adolescence and adulthood. Specifically, TOAs also can present secondary to other intra-abdominal pathologies, such as appendicitis, diverticulitis, and pyelonephritis, as well as a complication of intrauterine procedures, such as an HSG, or less commonly, following intrauterine device (IUD) insertion.5-7
Given that sexually transmitted infections are the most common etiology of TOAs, C trachomatis and N gonorrhoeae are the most likely microorganisms to be isolated (Table).8-12 In not sexually active populations, Escherichia coli and Gardnerella vaginalis should be considered instead. Although rare, women with IUDs have been shown to have an increased incidence of PID/TOA secondary to Actinomyces israleii relative to women without IUDs.12 In patients with TOAs secondary to intraabdominal surgery, anaerobic bacteria, such as Bacterioides and Peptostreptococcus species in addition to Escherichia coli, are likely culprits.11
In patients after HSG, infectious complications are uncommon enough that the American College of Obstetricians and Gynecologists recommends against antibiotic prophylaxis unless there are risk factors of dilated fallopian tubes or a history of PID.13 Identifying a precise percentage of TOA as a complication of HSG is rather elusive in the literature, though, it is frequently noted that infection in general is uncommon, and the risk of developing PID is about 1.4% to 3.4%.14 However, in the retrospective study most often cited, all women who developed PID following HSG had evidence of dilated fallopian tubes. Given our patient had no history of PID or dilated fallopian tubes, her risk of developing infection (PID or postprocedural abscess) would be considered very low; therefore, the index of suspicion also was low.15
Following diagnosis, the patient was promptly admitted and treated with a course of IV ceftriaxone 1 g every 24 hours, IV doxycycline 100 mg every 12 hours, and a single dose of IV metronidazole 500 mg. Her leukocytosis, fever, and pain improved within 48 hours without the need for percutaneous drainage and the patient made a complete recovery.
Complications
TOAs can carry significant morbidity and mortality, and there are both acute and chronic complications associated. Even if properly treated, TOAs can rupture leading to severe illness, such as peritonitis and septic shock. This often requires surgical intervention and hemodynamic pressure support in the intensive care unit setting.16 One of the most feared long-term complications of TOAs is infertility secondary to structural abnormalities of the female reproductive tract.10 Adhesions, strictures, and scarring are associated with TOAs irrespective of medical or surgical management, and thus any women with a history of PID or TOA require advanced fertility workup if they are having difficulties with conception or implantation.17
Minimizing infectious transmission also is essential in the treatment of TOA/PID. All women who receive a diagnosis of PID or TOA should be evaluated for gonorrhea, chlamydia, HIV, and syphilis. Women should be instructed to abstain from sexual intercourse until therapy is complete, symptoms have resolved, and sex partners have been treated for potential chlamydial or gonococcal infections. All contraceptive methods can be continued during treatment.
Conclusions
This case presented several challenges as the bilateral TOAs developed postprocedure in a patient without risk factors. Furthermore, this case did not follow the classic presentation of ascending bacterial translocation to the ovaries over days to weeks. The diagnosis was complicated by a largely benign physical examination and a pelvic examination without evidence of abnormal vaginal discharge or cervicitis. The only indicators were fever, leukocytosis, and abdominal pain in the setting of a recent, uncomplicated HSG procedure. Vigilance is required to obtain the necessary history, and the differential of TOA must be broadened to include women without a history or symptoms of a sexually transmitted infection (contrary to the classic association). We aim to encourage heightened clinical suspicion for TOAs in patients who present with fever, leukocytosis, and abdominal pain after recent HSG or other intrauterine instrumentation procedures and therefore improve patient outcomes.
1. Gkrozou F, Tsonis O, Daniilidis A, Navrozoglou I, Paschopoulos M. Tubo-ovarian abscess: exploring optimal treatment options based on current evidence. J Endometr Pelvic Pain Disord. 2020;13(1):10-19. doi:10.1177/2284026520960649
2. Taylor KJ, Wasson JF, De Graaff C, Rosenfield AT, Andriole VT. Accuracy of grey-scale ultrasound diagnosis of abdominal and pelvic abscesses in 220 patients. Lancet. 1978;1(8055):83-84. doi:10.1016/s0140-6736(78)90016-8
3. Bridwell RE, Koyfman A, Long B. High risk and low prevalence diseases: tubo-ovarian abscess. Am J Emerg Med. 2022;57:70-75. doi:10.1016/j.ajem.2022.04.026
4. Lambert MJ, Villa M. Gynecologic ultrasound in emergency medicine. Emerg Med Clin North Am. 2004;22(3):683-696. doi:10.1016/j.emc.2004.04.016
5. Munro K, Gharaibeh A, Nagabushanam S, Martin C. Diagnosis and management of tubo-ovarian abscesses. Obstet Gynaecol. 2018;20(1):11-19. doi:10.1111/tog.12447
6. Fink D, Lim PPC, Desai A, Stephans AB, Wien MA. Recurrent tubo-ovarian abscess in a nonsexually active adolescent. Consultant. 2022;62(1):e26-e28. doi:10.25270/con.2021.04.00010
7. Hiller N, Fux T, Finkelstein A, Mezeh H, Simanovsky N. CT differentiation between tubo-ovarian and appendiceal origin of right lower quadrant abscess: CT, clinical, and laboratory correlation. Emerg Radiol. 2016;23:133-139. doi:10.1007/s10140-015-1372-z
8. Kairys N, Roepke C. Tubo-ovarian abscess. In: StatPearls. Treasure Island (FL): StatPearls Publishing; June 12, 2023.
9. Gao Y, Qu P, Zhou Y, Ding W. Risk factors for the development of tubo-ovarian abscesses in women with ovarian endometriosis: a retrospective matched case-control study. BMC Womens Health. 2021:21:43. doi:10.1186/s12905-021-01188-6
10. Curry A, Williams T, Penny ML. Pelvic inflammatory disease: diagnosis, management, and prevention. Am Fam Physician. 2019;100(6):357-364.
11. Landers DV, Sweet RL. Tubo-ovarian abscess: contemporary approach to management. Rev Infect Dis. 1983;5(5):876-884. doi:10.1093/clinids/5.5.876
12. Burkman R, Schlesselman S, McCaffrey L, Gupta PK, Spence M. The relationship of genital tract actinomycetes and the development of pelvic inflammatory disease. Am J Obstet Gynecol. 1982;143(5):585-589. doi:10.1016/0002-9378(82)90552-x
13. Armstrong C. ACOG releases guidelines on antibiotic prophylaxis for gynecologic procedures. Am Fam Physician. 2007;75(7):1094-1096.
14. Pittaway DE, Winfield AC, Maxson W, Daniell J, Herbert C, Wentz AC. Prevention of acute pelvic inflammatory disease after hysterosalpingography: efficacy of doxycycline prophylaxis. Am J Obstet Gynecol. 1983;147(6):623-626. doi:10.1016/0002-9378(83)90438-6
15. Committee on Practice Bulletins—Gynecology. Prevention of Infection After Gynecologic Procedures: ACOG Practice Bulletin, Number 195. Obstet Gynecol. 2018;131(6):e172-e189. doi:10.1097/AOG.0000000000002670
16. Tao X, Ge SQ, Chen L, Cai LS, Hwang MF, Wang CL. Relationships between female infertility and female genital infections and pelvic inflammatory disease: a population-based nested controlled study. Clinics (Sao Paulo). 2018;73:e364. Published 2018 Aug 9. doi:10.6061/clinics/2018/e364
17. Fouks Y, Azem F, Many A, Cohen Y, Levin I, Cohen A. Fertility outcomes in patients with tubo-ovarian abscesses after an oocyte retrieval: a longitudinal cohort analysis. Arch Gynecol Obstet. 2019;300(3):763-769. doi:10.1007/s00404-019-05230-9
1. Gkrozou F, Tsonis O, Daniilidis A, Navrozoglou I, Paschopoulos M. Tubo-ovarian abscess: exploring optimal treatment options based on current evidence. J Endometr Pelvic Pain Disord. 2020;13(1):10-19. doi:10.1177/2284026520960649
2. Taylor KJ, Wasson JF, De Graaff C, Rosenfield AT, Andriole VT. Accuracy of grey-scale ultrasound diagnosis of abdominal and pelvic abscesses in 220 patients. Lancet. 1978;1(8055):83-84. doi:10.1016/s0140-6736(78)90016-8
3. Bridwell RE, Koyfman A, Long B. High risk and low prevalence diseases: tubo-ovarian abscess. Am J Emerg Med. 2022;57:70-75. doi:10.1016/j.ajem.2022.04.026
4. Lambert MJ, Villa M. Gynecologic ultrasound in emergency medicine. Emerg Med Clin North Am. 2004;22(3):683-696. doi:10.1016/j.emc.2004.04.016
5. Munro K, Gharaibeh A, Nagabushanam S, Martin C. Diagnosis and management of tubo-ovarian abscesses. Obstet Gynaecol. 2018;20(1):11-19. doi:10.1111/tog.12447
6. Fink D, Lim PPC, Desai A, Stephans AB, Wien MA. Recurrent tubo-ovarian abscess in a nonsexually active adolescent. Consultant. 2022;62(1):e26-e28. doi:10.25270/con.2021.04.00010
7. Hiller N, Fux T, Finkelstein A, Mezeh H, Simanovsky N. CT differentiation between tubo-ovarian and appendiceal origin of right lower quadrant abscess: CT, clinical, and laboratory correlation. Emerg Radiol. 2016;23:133-139. doi:10.1007/s10140-015-1372-z
8. Kairys N, Roepke C. Tubo-ovarian abscess. In: StatPearls. Treasure Island (FL): StatPearls Publishing; June 12, 2023.
9. Gao Y, Qu P, Zhou Y, Ding W. Risk factors for the development of tubo-ovarian abscesses in women with ovarian endometriosis: a retrospective matched case-control study. BMC Womens Health. 2021:21:43. doi:10.1186/s12905-021-01188-6
10. Curry A, Williams T, Penny ML. Pelvic inflammatory disease: diagnosis, management, and prevention. Am Fam Physician. 2019;100(6):357-364.
11. Landers DV, Sweet RL. Tubo-ovarian abscess: contemporary approach to management. Rev Infect Dis. 1983;5(5):876-884. doi:10.1093/clinids/5.5.876
12. Burkman R, Schlesselman S, McCaffrey L, Gupta PK, Spence M. The relationship of genital tract actinomycetes and the development of pelvic inflammatory disease. Am J Obstet Gynecol. 1982;143(5):585-589. doi:10.1016/0002-9378(82)90552-x
13. Armstrong C. ACOG releases guidelines on antibiotic prophylaxis for gynecologic procedures. Am Fam Physician. 2007;75(7):1094-1096.
14. Pittaway DE, Winfield AC, Maxson W, Daniell J, Herbert C, Wentz AC. Prevention of acute pelvic inflammatory disease after hysterosalpingography: efficacy of doxycycline prophylaxis. Am J Obstet Gynecol. 1983;147(6):623-626. doi:10.1016/0002-9378(83)90438-6
15. Committee on Practice Bulletins—Gynecology. Prevention of Infection After Gynecologic Procedures: ACOG Practice Bulletin, Number 195. Obstet Gynecol. 2018;131(6):e172-e189. doi:10.1097/AOG.0000000000002670
16. Tao X, Ge SQ, Chen L, Cai LS, Hwang MF, Wang CL. Relationships between female infertility and female genital infections and pelvic inflammatory disease: a population-based nested controlled study. Clinics (Sao Paulo). 2018;73:e364. Published 2018 Aug 9. doi:10.6061/clinics/2018/e364
17. Fouks Y, Azem F, Many A, Cohen Y, Levin I, Cohen A. Fertility outcomes in patients with tubo-ovarian abscesses after an oocyte retrieval: a longitudinal cohort analysis. Arch Gynecol Obstet. 2019;300(3):763-769. doi:10.1007/s00404-019-05230-9