User login
New Culprit in Allergic Dermatitis
ISTANBUL, TURKEY – Oxidized R-limonene, found in more than 60% of personal care and hygiene products, has been found to be a major cause of fragrance allergy, according to the findings of a large, international, patch test study.
Overall, 5.2% of almost 3,000 patients had a positive patch test result for oxidized limonene, and 37% of those reactions were deemed to be clinically relevant. Shampoos, soaps, perfumes, domestic cleaners, sunscreens, and massage creams are among the household products likely to contain limonene, Dr. Johanna Bråred Christensson said at the annual congress of the European Academy of Dermatology and Venereology. R-limonene is also used in high concentrations in industry as a solvent and degreaser.
R-limonene is structurally a fragrance terpene, with a citrus odor. It is found in nature and produced industrially in mass quantities. In its pure form, R-limonene is nonallergenic or at most a weak allergen. However, when R-limonene is oxidized, it includes limonene hydroperoxides, which Dr. Christensson found to be a contact allergen. This oxidation can occur during storage or handling of R-limonene. The hydroperoxides are often already present in containers of R-limonene when shipped from chemical plants to manufacturers of consumer products, according to Dr. Christensson, a dermatologist at the University of Gothenburg (Sweden).
A stable oxidized R-limonene 3.0% in petrolatum with a controlled 0.33% concentration of limonene hydroperoxides, which is commercially available as a patch test material, was used in the patch test study of 2,900 consecutive dermatitis patients who presented to contact dermatitis clinics in Sweden, Denmark, the United Kingdom, Spain, Australia, and Singapore. Participants completed a questionnaire assessing the relevance of a positive patch test result as indicated by exposure to limonene-containing products on the area of their dermatitis.
The study was funded by grants from national allergy research centers. Dr. Christensson reported having no relevant financial conflicts.
ISTANBUL, TURKEY – Oxidized R-limonene, found in more than 60% of personal care and hygiene products, has been found to be a major cause of fragrance allergy, according to the findings of a large, international, patch test study.
Overall, 5.2% of almost 3,000 patients had a positive patch test result for oxidized limonene, and 37% of those reactions were deemed to be clinically relevant. Shampoos, soaps, perfumes, domestic cleaners, sunscreens, and massage creams are among the household products likely to contain limonene, Dr. Johanna Bråred Christensson said at the annual congress of the European Academy of Dermatology and Venereology. R-limonene is also used in high concentrations in industry as a solvent and degreaser.
R-limonene is structurally a fragrance terpene, with a citrus odor. It is found in nature and produced industrially in mass quantities. In its pure form, R-limonene is nonallergenic or at most a weak allergen. However, when R-limonene is oxidized, it includes limonene hydroperoxides, which Dr. Christensson found to be a contact allergen. This oxidation can occur during storage or handling of R-limonene. The hydroperoxides are often already present in containers of R-limonene when shipped from chemical plants to manufacturers of consumer products, according to Dr. Christensson, a dermatologist at the University of Gothenburg (Sweden).
A stable oxidized R-limonene 3.0% in petrolatum with a controlled 0.33% concentration of limonene hydroperoxides, which is commercially available as a patch test material, was used in the patch test study of 2,900 consecutive dermatitis patients who presented to contact dermatitis clinics in Sweden, Denmark, the United Kingdom, Spain, Australia, and Singapore. Participants completed a questionnaire assessing the relevance of a positive patch test result as indicated by exposure to limonene-containing products on the area of their dermatitis.
The study was funded by grants from national allergy research centers. Dr. Christensson reported having no relevant financial conflicts.
ISTANBUL, TURKEY – Oxidized R-limonene, found in more than 60% of personal care and hygiene products, has been found to be a major cause of fragrance allergy, according to the findings of a large, international, patch test study.
Overall, 5.2% of almost 3,000 patients had a positive patch test result for oxidized limonene, and 37% of those reactions were deemed to be clinically relevant. Shampoos, soaps, perfumes, domestic cleaners, sunscreens, and massage creams are among the household products likely to contain limonene, Dr. Johanna Bråred Christensson said at the annual congress of the European Academy of Dermatology and Venereology. R-limonene is also used in high concentrations in industry as a solvent and degreaser.
R-limonene is structurally a fragrance terpene, with a citrus odor. It is found in nature and produced industrially in mass quantities. In its pure form, R-limonene is nonallergenic or at most a weak allergen. However, when R-limonene is oxidized, it includes limonene hydroperoxides, which Dr. Christensson found to be a contact allergen. This oxidation can occur during storage or handling of R-limonene. The hydroperoxides are often already present in containers of R-limonene when shipped from chemical plants to manufacturers of consumer products, according to Dr. Christensson, a dermatologist at the University of Gothenburg (Sweden).
A stable oxidized R-limonene 3.0% in petrolatum with a controlled 0.33% concentration of limonene hydroperoxides, which is commercially available as a patch test material, was used in the patch test study of 2,900 consecutive dermatitis patients who presented to contact dermatitis clinics in Sweden, Denmark, the United Kingdom, Spain, Australia, and Singapore. Participants completed a questionnaire assessing the relevance of a positive patch test result as indicated by exposure to limonene-containing products on the area of their dermatitis.
The study was funded by grants from national allergy research centers. Dr. Christensson reported having no relevant financial conflicts.
AT THE EADV CONGRESS
Oxidized limonene: New culprit in allergic dermatitis
ISTANBUL, TURKEY – Oxidized R-limonene, found in more than 60% of personal care and hygiene products, has been found to be a major cause of fragrance allergy, according to the findings of a large, international, patch test study.
Overall, 5.2% of almost 3,000 patients had a positive patch test result for oxidized limonene, and 37% of those reactions were deemed to be clinically relevant. Shampoos, soaps, perfumes, domestic cleaners, sunscreens, and massage creams are among the household products likely to contain limonene, Dr. Johanna Bråred Christensson said at the annual congress of the European Academy of Dermatology and Venereology. R-limonene is also used in high concentrations in industry as a solvent and degreaser.
R-limonene is structurally a fragrance terpene, with a citrus odor. It is found in nature and produced industrially in mass quantities. In its pure form, R-limonene is nonallergenic or at most a weak allergen. However, when R-limonene is oxidized, it includes limonene hydroperoxides, which Dr. Christensson found to be a contact allergen. This oxidation can occur during storage or handling of R-limonene. The hydroperoxides are often already present in containers of R-limonene when shipped from chemical plants to manufacturers of consumer products, according to Dr. Christensson, a dermatologist at the University of Gothenburg (Sweden).
A stable oxidized R-limonene 3.0% in petrolatum with a controlled 0.33% concentration of limonene hydroperoxides, which is commercially available as a patch test material, was used in the patch test study of 2,900 consecutive dermatitis patients who presented to contact dermatitis clinics in Sweden, Denmark, the United Kingdom, Spain, Australia, and Singapore. Participants completed a questionnaire assessing the relevance of a positive patch test result as indicated by exposure to limonene-containing products on the area of their dermatitis.
The study was funded by grants from national allergy research centers. Dr. Christensson reported having no relevant financial conflicts.
ISTANBUL, TURKEY – Oxidized R-limonene, found in more than 60% of personal care and hygiene products, has been found to be a major cause of fragrance allergy, according to the findings of a large, international, patch test study.
Overall, 5.2% of almost 3,000 patients had a positive patch test result for oxidized limonene, and 37% of those reactions were deemed to be clinically relevant. Shampoos, soaps, perfumes, domestic cleaners, sunscreens, and massage creams are among the household products likely to contain limonene, Dr. Johanna Bråred Christensson said at the annual congress of the European Academy of Dermatology and Venereology. R-limonene is also used in high concentrations in industry as a solvent and degreaser.
R-limonene is structurally a fragrance terpene, with a citrus odor. It is found in nature and produced industrially in mass quantities. In its pure form, R-limonene is nonallergenic or at most a weak allergen. However, when R-limonene is oxidized, it includes limonene hydroperoxides, which Dr. Christensson found to be a contact allergen. This oxidation can occur during storage or handling of R-limonene. The hydroperoxides are often already present in containers of R-limonene when shipped from chemical plants to manufacturers of consumer products, according to Dr. Christensson, a dermatologist at the University of Gothenburg (Sweden).
A stable oxidized R-limonene 3.0% in petrolatum with a controlled 0.33% concentration of limonene hydroperoxides, which is commercially available as a patch test material, was used in the patch test study of 2,900 consecutive dermatitis patients who presented to contact dermatitis clinics in Sweden, Denmark, the United Kingdom, Spain, Australia, and Singapore. Participants completed a questionnaire assessing the relevance of a positive patch test result as indicated by exposure to limonene-containing products on the area of their dermatitis.
The study was funded by grants from national allergy research centers. Dr. Christensson reported having no relevant financial conflicts.
ISTANBUL, TURKEY – Oxidized R-limonene, found in more than 60% of personal care and hygiene products, has been found to be a major cause of fragrance allergy, according to the findings of a large, international, patch test study.
Overall, 5.2% of almost 3,000 patients had a positive patch test result for oxidized limonene, and 37% of those reactions were deemed to be clinically relevant. Shampoos, soaps, perfumes, domestic cleaners, sunscreens, and massage creams are among the household products likely to contain limonene, Dr. Johanna Bråred Christensson said at the annual congress of the European Academy of Dermatology and Venereology. R-limonene is also used in high concentrations in industry as a solvent and degreaser.
R-limonene is structurally a fragrance terpene, with a citrus odor. It is found in nature and produced industrially in mass quantities. In its pure form, R-limonene is nonallergenic or at most a weak allergen. However, when R-limonene is oxidized, it includes limonene hydroperoxides, which Dr. Christensson found to be a contact allergen. This oxidation can occur during storage or handling of R-limonene. The hydroperoxides are often already present in containers of R-limonene when shipped from chemical plants to manufacturers of consumer products, according to Dr. Christensson, a dermatologist at the University of Gothenburg (Sweden).
A stable oxidized R-limonene 3.0% in petrolatum with a controlled 0.33% concentration of limonene hydroperoxides, which is commercially available as a patch test material, was used in the patch test study of 2,900 consecutive dermatitis patients who presented to contact dermatitis clinics in Sweden, Denmark, the United Kingdom, Spain, Australia, and Singapore. Participants completed a questionnaire assessing the relevance of a positive patch test result as indicated by exposure to limonene-containing products on the area of their dermatitis.
The study was funded by grants from national allergy research centers. Dr. Christensson reported having no relevant financial conflicts.
AT THE EADV CONGRESS
Major finding: More than 5% of 2,900 consecutive patch test patients had a positive result for oxidized limonene. More than one-third of the positive results were deemed clinically relevant.
Data source: A prospective study involving 2,900 consecutive dermatitis patients who underwent patch testing at contact dermatitis clinics in six countries.
Disclosures: The study was funded by grants from national allergy research centers. Dr. Christensson reported having no relevant financial conflicts.
Delayed-release prednisone lessened rheumatoid arthritis symptoms
SAN DIEGO – Rheumatoid arthritis patients who switched from conventional immediate-release prednisone to delayed-release prednisone taken at bedtime experienced a mean reduction of more than 60 minutes in daily morning stiffness that was maintained throughout a 9-month open-label study.
This reduction from a mean baseline of 143.5 minutes of daily morning stiffness during 3 months on a mean 6.8 mg/day of immediate-release prednisone to a similar dose of proprietary delayed-release prednisone (Rayos) constitutes a clinically meaningful benefit. It was accompanied by improvements in other important patient-reported outcomes, Dr. Allan Gibofsky observed at the annual meeting of the American College of Rheumatology.
He presented an analysis of the 9-month open-label phase of the CAPRA-1 (Circadian Administration of Prednisone in Rheumatoid Arthritis-1) study, which also featured a previously reported double-blind phase. The open-label phase involved 207 patients with baseline moderate-to-severe active RA on a stable regimen of disease-modifying antirheumatic drug (DMARD) therapy. After 3 months on immediate-release prednisone during the double-blind phase of CAPRI-1, 110 study participants were switched to delayed-release prednisone; the other 97 had been on delayed-release prednisone during the double-blind phase and continued taking it at about 10 p.m. during the open-label phase.
Levels of the inflammatory cytokine interleukin-6 (IL-6) were measured at the start and conclusion of the 9-month open-label study. The 107 patients who switched from immediate- to delayed-release prednisone showed a median 53% drop in IL-6 levels from a baseline of 1,055 IU/L, reported Dr. Gibofsky, professor of medicine and public health at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Self-reported pain scores on a 0-100 visual analog scale dropped from a baseline of 44 by a mean 6.1 points after the switch to delayed-release prednisone and remained stable thereafter. In addition, patient-reported global assessment improved significantly at 3 and 6 months despite 3 months of previous double-blind therapy with immediate-release prednisone.
Delayed-release prednisone is approved as an anti-inflammatory or immunosuppressive agent for a wide array of dermatologic, respiratory, GI, renal, and rheumatologic disorders, including RA. The underlying rationale for this form of chronotherapy is that it permits timed delivery of prednisone during the early morning hours in accord with the circadian pattern of inflammatory cytokine levels.
CAPRI-1 was funded by Horizon Pharma, which markets Rayos. Dr. Gibofsky is a consultant to the company.
SAN DIEGO – Rheumatoid arthritis patients who switched from conventional immediate-release prednisone to delayed-release prednisone taken at bedtime experienced a mean reduction of more than 60 minutes in daily morning stiffness that was maintained throughout a 9-month open-label study.
This reduction from a mean baseline of 143.5 minutes of daily morning stiffness during 3 months on a mean 6.8 mg/day of immediate-release prednisone to a similar dose of proprietary delayed-release prednisone (Rayos) constitutes a clinically meaningful benefit. It was accompanied by improvements in other important patient-reported outcomes, Dr. Allan Gibofsky observed at the annual meeting of the American College of Rheumatology.
He presented an analysis of the 9-month open-label phase of the CAPRA-1 (Circadian Administration of Prednisone in Rheumatoid Arthritis-1) study, which also featured a previously reported double-blind phase. The open-label phase involved 207 patients with baseline moderate-to-severe active RA on a stable regimen of disease-modifying antirheumatic drug (DMARD) therapy. After 3 months on immediate-release prednisone during the double-blind phase of CAPRI-1, 110 study participants were switched to delayed-release prednisone; the other 97 had been on delayed-release prednisone during the double-blind phase and continued taking it at about 10 p.m. during the open-label phase.
Levels of the inflammatory cytokine interleukin-6 (IL-6) were measured at the start and conclusion of the 9-month open-label study. The 107 patients who switched from immediate- to delayed-release prednisone showed a median 53% drop in IL-6 levels from a baseline of 1,055 IU/L, reported Dr. Gibofsky, professor of medicine and public health at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Self-reported pain scores on a 0-100 visual analog scale dropped from a baseline of 44 by a mean 6.1 points after the switch to delayed-release prednisone and remained stable thereafter. In addition, patient-reported global assessment improved significantly at 3 and 6 months despite 3 months of previous double-blind therapy with immediate-release prednisone.
Delayed-release prednisone is approved as an anti-inflammatory or immunosuppressive agent for a wide array of dermatologic, respiratory, GI, renal, and rheumatologic disorders, including RA. The underlying rationale for this form of chronotherapy is that it permits timed delivery of prednisone during the early morning hours in accord with the circadian pattern of inflammatory cytokine levels.
CAPRI-1 was funded by Horizon Pharma, which markets Rayos. Dr. Gibofsky is a consultant to the company.
SAN DIEGO – Rheumatoid arthritis patients who switched from conventional immediate-release prednisone to delayed-release prednisone taken at bedtime experienced a mean reduction of more than 60 minutes in daily morning stiffness that was maintained throughout a 9-month open-label study.
This reduction from a mean baseline of 143.5 minutes of daily morning stiffness during 3 months on a mean 6.8 mg/day of immediate-release prednisone to a similar dose of proprietary delayed-release prednisone (Rayos) constitutes a clinically meaningful benefit. It was accompanied by improvements in other important patient-reported outcomes, Dr. Allan Gibofsky observed at the annual meeting of the American College of Rheumatology.
He presented an analysis of the 9-month open-label phase of the CAPRA-1 (Circadian Administration of Prednisone in Rheumatoid Arthritis-1) study, which also featured a previously reported double-blind phase. The open-label phase involved 207 patients with baseline moderate-to-severe active RA on a stable regimen of disease-modifying antirheumatic drug (DMARD) therapy. After 3 months on immediate-release prednisone during the double-blind phase of CAPRI-1, 110 study participants were switched to delayed-release prednisone; the other 97 had been on delayed-release prednisone during the double-blind phase and continued taking it at about 10 p.m. during the open-label phase.
Levels of the inflammatory cytokine interleukin-6 (IL-6) were measured at the start and conclusion of the 9-month open-label study. The 107 patients who switched from immediate- to delayed-release prednisone showed a median 53% drop in IL-6 levels from a baseline of 1,055 IU/L, reported Dr. Gibofsky, professor of medicine and public health at Cornell University and a rheumatologist at the Hospital for Special Surgery, both in New York.
Self-reported pain scores on a 0-100 visual analog scale dropped from a baseline of 44 by a mean 6.1 points after the switch to delayed-release prednisone and remained stable thereafter. In addition, patient-reported global assessment improved significantly at 3 and 6 months despite 3 months of previous double-blind therapy with immediate-release prednisone.
Delayed-release prednisone is approved as an anti-inflammatory or immunosuppressive agent for a wide array of dermatologic, respiratory, GI, renal, and rheumatologic disorders, including RA. The underlying rationale for this form of chronotherapy is that it permits timed delivery of prednisone during the early morning hours in accord with the circadian pattern of inflammatory cytokine levels.
CAPRI-1 was funded by Horizon Pharma, which markets Rayos. Dr. Gibofsky is a consultant to the company.
AT THE ACR ANNUAL MEETING
Major finding: Patients with moderate to severe active rheumatoid arthritis on stable disease-modifying antirheumatic drug therapy experienced a greater than 60-minute reduction in daily morning stiffness from a baseline of 143.5 minutes when they were switched from immediate- to delayed-release prednisone.
Data source: This was an analysis of the 207 RA patients who participated in the 9-month open-label phase of the prospective CAPRA-1 study.
Disclosures: The study was funded by Horizon Pharma, which markets immediate-release prednisone. Dr. Gibofsky is a consultant to the company.
Novel topical agent reduces chronic itch
ISTANBUL, TURKEY – A first-in-class topical tyrosine kinase inhibitor markedly reduced chronic pruritus in psoriasis patients in a phase-IIb study.
Moreover, the investigational agent, known for now as CT327, produced no application site reactions or indeed any other adverse events. Nor was it absorbed systemically, Dr. David Roblin reported at the annual congress of the European Academy of Dermatology and Venereology.
A significant unmet need exists for safe and effective therapies for chronic itching, not only in psoriasis but in other diseases for which chronic pruritus figures prominently and has a debilitating effect on quality of life. At present, there is no medication with an indication for treatment of chronic pruritus, noted Dr. Roblin, chief medical officer at Creabilis in Canterbury, England.
Tyrosine kinase is a high-affinity receptor of nerve growth factor. Thus, CT327 targets the sensory neurons implicated in the pathogenesis of chronic pruritis, he explained.
The multicenter, randomized, double-blind clinical trial included 160 patients with mild-to-moderate psoriasis affecting up to 10% of their body surface area. They were assigned to twice-daily application of CT327 at 0.05%, 0.1%, or 0.5% or to the vehicle for 8 weeks. Participants treated all their plaques except those on the face or scalp.
The safety assessment included all 160 patients; however, efficacy endpoints were assessed only in the 108 with at least moderate baseline pruritus as defined by a score greater than 40 mm on a 0-100 mm visual analog scale (VAS).
From a baseline mean pruritus VAS of 65.2, scores decreased at week 8 by 37.1 mm in the CT327 0.05% group, 31.5 mm in patients using CT327 0.1%, 36.4 mm in the 0.5% group, and 16.1 mm in vehicle-treated controls. A 20-mm reduction on the VAS is considered clinically meaningful. Four weeks after treatment stopped, pruritus scores had returned to baseline.
The 108 patients with at least moderate baseline pruritus had a baseline mean Psoriasis Area and Severity Index score, modified to exclude the untreated face and scalp (mPASI), of 9.3. At 8 weeks, patients in the CT327 0.05%, 0.1%, and 0.5% groups averaged mPASI score reductions of 46%, 36%, and 37%, respectively, compared with a 17% drop in the control group.
Of note, there was no correlation between baseline mPASI and pruritus severity scores, which suggests that trying to treat pruritus with agents directed at the inflammatory aspect of psoriasis may be a suboptimal strategy. It’s better to target the sensory neurons involved in the itch, according to Dr. Roblin.
The study was sponsored by Creabilis, which is developing CT327. Dr. Roblin is the company’s medical director.
ISTANBUL, TURKEY – A first-in-class topical tyrosine kinase inhibitor markedly reduced chronic pruritus in psoriasis patients in a phase-IIb study.
Moreover, the investigational agent, known for now as CT327, produced no application site reactions or indeed any other adverse events. Nor was it absorbed systemically, Dr. David Roblin reported at the annual congress of the European Academy of Dermatology and Venereology.
A significant unmet need exists for safe and effective therapies for chronic itching, not only in psoriasis but in other diseases for which chronic pruritus figures prominently and has a debilitating effect on quality of life. At present, there is no medication with an indication for treatment of chronic pruritus, noted Dr. Roblin, chief medical officer at Creabilis in Canterbury, England.
Tyrosine kinase is a high-affinity receptor of nerve growth factor. Thus, CT327 targets the sensory neurons implicated in the pathogenesis of chronic pruritis, he explained.
The multicenter, randomized, double-blind clinical trial included 160 patients with mild-to-moderate psoriasis affecting up to 10% of their body surface area. They were assigned to twice-daily application of CT327 at 0.05%, 0.1%, or 0.5% or to the vehicle for 8 weeks. Participants treated all their plaques except those on the face or scalp.
The safety assessment included all 160 patients; however, efficacy endpoints were assessed only in the 108 with at least moderate baseline pruritus as defined by a score greater than 40 mm on a 0-100 mm visual analog scale (VAS).
From a baseline mean pruritus VAS of 65.2, scores decreased at week 8 by 37.1 mm in the CT327 0.05% group, 31.5 mm in patients using CT327 0.1%, 36.4 mm in the 0.5% group, and 16.1 mm in vehicle-treated controls. A 20-mm reduction on the VAS is considered clinically meaningful. Four weeks after treatment stopped, pruritus scores had returned to baseline.
The 108 patients with at least moderate baseline pruritus had a baseline mean Psoriasis Area and Severity Index score, modified to exclude the untreated face and scalp (mPASI), of 9.3. At 8 weeks, patients in the CT327 0.05%, 0.1%, and 0.5% groups averaged mPASI score reductions of 46%, 36%, and 37%, respectively, compared with a 17% drop in the control group.
Of note, there was no correlation between baseline mPASI and pruritus severity scores, which suggests that trying to treat pruritus with agents directed at the inflammatory aspect of psoriasis may be a suboptimal strategy. It’s better to target the sensory neurons involved in the itch, according to Dr. Roblin.
The study was sponsored by Creabilis, which is developing CT327. Dr. Roblin is the company’s medical director.
ISTANBUL, TURKEY – A first-in-class topical tyrosine kinase inhibitor markedly reduced chronic pruritus in psoriasis patients in a phase-IIb study.
Moreover, the investigational agent, known for now as CT327, produced no application site reactions or indeed any other adverse events. Nor was it absorbed systemically, Dr. David Roblin reported at the annual congress of the European Academy of Dermatology and Venereology.
A significant unmet need exists for safe and effective therapies for chronic itching, not only in psoriasis but in other diseases for which chronic pruritus figures prominently and has a debilitating effect on quality of life. At present, there is no medication with an indication for treatment of chronic pruritus, noted Dr. Roblin, chief medical officer at Creabilis in Canterbury, England.
Tyrosine kinase is a high-affinity receptor of nerve growth factor. Thus, CT327 targets the sensory neurons implicated in the pathogenesis of chronic pruritis, he explained.
The multicenter, randomized, double-blind clinical trial included 160 patients with mild-to-moderate psoriasis affecting up to 10% of their body surface area. They were assigned to twice-daily application of CT327 at 0.05%, 0.1%, or 0.5% or to the vehicle for 8 weeks. Participants treated all their plaques except those on the face or scalp.
The safety assessment included all 160 patients; however, efficacy endpoints were assessed only in the 108 with at least moderate baseline pruritus as defined by a score greater than 40 mm on a 0-100 mm visual analog scale (VAS).
From a baseline mean pruritus VAS of 65.2, scores decreased at week 8 by 37.1 mm in the CT327 0.05% group, 31.5 mm in patients using CT327 0.1%, 36.4 mm in the 0.5% group, and 16.1 mm in vehicle-treated controls. A 20-mm reduction on the VAS is considered clinically meaningful. Four weeks after treatment stopped, pruritus scores had returned to baseline.
The 108 patients with at least moderate baseline pruritus had a baseline mean Psoriasis Area and Severity Index score, modified to exclude the untreated face and scalp (mPASI), of 9.3. At 8 weeks, patients in the CT327 0.05%, 0.1%, and 0.5% groups averaged mPASI score reductions of 46%, 36%, and 37%, respectively, compared with a 17% drop in the control group.
Of note, there was no correlation between baseline mPASI and pruritus severity scores, which suggests that trying to treat pruritus with agents directed at the inflammatory aspect of psoriasis may be a suboptimal strategy. It’s better to target the sensory neurons involved in the itch, according to Dr. Roblin.
The study was sponsored by Creabilis, which is developing CT327. Dr. Roblin is the company’s medical director.
AT THE EADV CONGRESS
Major finding: Treatment of chronic pruritus using various doses of CT327, a novel topical tyrosine kinase inhibitor, resulted in pruritus score reductions of 32-37 points from a mean baseline of 65 on a 0-100 scale.
Data source: This was a randomized, double-blind, multicenter, phase-IIb study including 160 patients with mild-to-moderate psoriasis who applied the investigational agent or its vehicle to their plaques twice daily for 8 weeks.
Disclosures: The study was sponsored by Creabilis. Dr. Roblin is the company’s chief medical officer.
Psoriasis drug research aimed at severe disease
ISTANBUL, TURKEY – The current intensive period of new drug development in psoriasis is cause for enthusiastic celebration by patients and physicians alike. That is, except for one glaring research gap.
Almost all of the promising drugs now in phase-II or -III clinical trials are systemic agents aimed at the minority of psoriasis patients with more severe disease. There’s not nearly as much in the pipeline for the many patients with mild psoriasis who are most appropriately treated with topical therapy, Dr. Hervé Bachelez observed at the annual congress of the European Academy of Dermatology and Venereology.
Indeed, he identified only two promising topical agents advancing through the developmental pipeline: Pfizer’s tofacitinib, a small-molecule Janus kinase inhibitor under study in both topical and oral formulations, and Anacor’s AN2728, a boron-containing molecule that inhibits phosphodiesterase-4 (PDE-4).
Industry’s lack of enthusiasm for developing new topical agents is difficult to understand, given that 84% of all U.S. prescriptions written for psoriasis in 2009 were for topical agents, according to IMS. Moreover, patients clearly believe there is a major unmet need for new and better topicals: a recent survey of 2,151 European psoriasis patients and their dermatologists found that only 45% of patients on topical therapy were satisfied with their treatment. And a mere 35% of dermatologists rated current topical treatments as satisfactory (J. Dermatolog. Treat. 2013;24:193-8).
The great bulk of the action in psoriasis drug development now is focused on injectable biologics and oral small molecules. Among them are three interleukin-17 inhibitors: Novartis’ secukinumab, Amgen’s brodalumab, and Eli Lilly’s ixekizumab. Celgene is developing apremilast as an oral PDE-4 inhibitor. Merck and Janssen Biotech are developing MK-3222 and CNTO 1959, respectively, as interleukin-23 inhibitors.
Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris, reported serving on the advisory boards of 10 pharmaceutical companies.
ISTANBUL, TURKEY – The current intensive period of new drug development in psoriasis is cause for enthusiastic celebration by patients and physicians alike. That is, except for one glaring research gap.
Almost all of the promising drugs now in phase-II or -III clinical trials are systemic agents aimed at the minority of psoriasis patients with more severe disease. There’s not nearly as much in the pipeline for the many patients with mild psoriasis who are most appropriately treated with topical therapy, Dr. Hervé Bachelez observed at the annual congress of the European Academy of Dermatology and Venereology.
Indeed, he identified only two promising topical agents advancing through the developmental pipeline: Pfizer’s tofacitinib, a small-molecule Janus kinase inhibitor under study in both topical and oral formulations, and Anacor’s AN2728, a boron-containing molecule that inhibits phosphodiesterase-4 (PDE-4).
Industry’s lack of enthusiasm for developing new topical agents is difficult to understand, given that 84% of all U.S. prescriptions written for psoriasis in 2009 were for topical agents, according to IMS. Moreover, patients clearly believe there is a major unmet need for new and better topicals: a recent survey of 2,151 European psoriasis patients and their dermatologists found that only 45% of patients on topical therapy were satisfied with their treatment. And a mere 35% of dermatologists rated current topical treatments as satisfactory (J. Dermatolog. Treat. 2013;24:193-8).
The great bulk of the action in psoriasis drug development now is focused on injectable biologics and oral small molecules. Among them are three interleukin-17 inhibitors: Novartis’ secukinumab, Amgen’s brodalumab, and Eli Lilly’s ixekizumab. Celgene is developing apremilast as an oral PDE-4 inhibitor. Merck and Janssen Biotech are developing MK-3222 and CNTO 1959, respectively, as interleukin-23 inhibitors.
Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris, reported serving on the advisory boards of 10 pharmaceutical companies.
ISTANBUL, TURKEY – The current intensive period of new drug development in psoriasis is cause for enthusiastic celebration by patients and physicians alike. That is, except for one glaring research gap.
Almost all of the promising drugs now in phase-II or -III clinical trials are systemic agents aimed at the minority of psoriasis patients with more severe disease. There’s not nearly as much in the pipeline for the many patients with mild psoriasis who are most appropriately treated with topical therapy, Dr. Hervé Bachelez observed at the annual congress of the European Academy of Dermatology and Venereology.
Indeed, he identified only two promising topical agents advancing through the developmental pipeline: Pfizer’s tofacitinib, a small-molecule Janus kinase inhibitor under study in both topical and oral formulations, and Anacor’s AN2728, a boron-containing molecule that inhibits phosphodiesterase-4 (PDE-4).
Industry’s lack of enthusiasm for developing new topical agents is difficult to understand, given that 84% of all U.S. prescriptions written for psoriasis in 2009 were for topical agents, according to IMS. Moreover, patients clearly believe there is a major unmet need for new and better topicals: a recent survey of 2,151 European psoriasis patients and their dermatologists found that only 45% of patients on topical therapy were satisfied with their treatment. And a mere 35% of dermatologists rated current topical treatments as satisfactory (J. Dermatolog. Treat. 2013;24:193-8).
The great bulk of the action in psoriasis drug development now is focused on injectable biologics and oral small molecules. Among them are three interleukin-17 inhibitors: Novartis’ secukinumab, Amgen’s brodalumab, and Eli Lilly’s ixekizumab. Celgene is developing apremilast as an oral PDE-4 inhibitor. Merck and Janssen Biotech are developing MK-3222 and CNTO 1959, respectively, as interleukin-23 inhibitors.
Dr. Bachelez, professor of dermatology and head of the inflammatory skin diseases unit at Saint Louis University Hospital, Paris, reported serving on the advisory boards of 10 pharmaceutical companies.
EXPERT ANALYSIS FROM THE EADV CONGRESS
Losartan effective in Marfan syndrome
AMSTERDAM – Daily losartan significantly slowed the aortic root dilatation rate in adults with Marfan syndrome in a 3-year randomized clinical trial.
"I think we can be positive about this treatment. We can now recommend losartan in clinical practice," Dr. Maarten Groenink said at the annual congress of the European Society of Cardiology.
The COMPARE (Cozaar in Marfan Patients Reduces Aortic Enlargement) trial included 218 patients at all four university Marfan centers in the Netherlands. Patients were randomized to oral losartan at a target dose of 100 mg/day or no losartan in addition to standard-of-care treatment with beta-blockers. Roughly half of the patients in the losartan group were unable to tolerate the full dose of losartan in addition to a beta-blocker; those patients were maintained on losartan at 50 mg/day. Aortic root diameter was measured by MRI at enrollment and after 3 years of prospective follow-up. The aortic dilatation rate was significantly lower in the losartan group than in controls both in the patients with a native root and in those who had undergone aortic root replacement surgery, reported Dr. Groenink, a cardiologist at the Academic Medical Center, Amsterdam.
There were no aortic dissections in the losartan group and two in the control arm. Elective aortic replacement surgery was performed in a similar number of patients in both groups.
Blood pressure was lower in the losartan group, yet blood pressure didn’t correlate with the aortic dilatation rate. Dr. Groenink speculated that losartan’s chief mechanism of benefit in Marfan syndrome is its ability to curb overexpression of transforming growth factor-beta, which weakens the structure of the media layer of the aortic wall. He said it’s unknown whether losartan’s benefits are specific to that drug or are a class effect.
Ongoing clinical trials are evaluating losartan in children and adolescents with Marfan syndrome, he said, adding that there is a solid rationale for beginning treatment as early in life as possible. "I believe the adverse effects on the aortic wall in Marfan syndrome are caused by the fibrillin defect but also by wear and tear due to cyclic stress by the beating heart. So you can hypothesize that the earlier you start treatment, the better," he explained.
The prevalence of Marfan syndrome has been estimated at 1 in 5,000, but Dr. Groenink suspects the syndrome may actually be more common than that.
Simultaneous with Dr. Groenink’s presentation at the ESC, the COMPARE results were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht334]).
Dr. Groenink reported having no disclosures.

|
| Dr. John Gordon Harold |
COMPARE is a very important study whose results are going to mean a paradigm shift for the management of Marfan syndrome.
It is intriguing to consider that the benefits of losartan might possibly also extend to patients with thoracic aortic disease in general, a worthy topic for future investigation.
Dr. John Gordon Harold is with Cedars-Sinai Heart Institute, Los Angeles, and president of the American College of Cardiology. He had no relevant financial disclosures.

|
| Dr. John Gordon Harold |
COMPARE is a very important study whose results are going to mean a paradigm shift for the management of Marfan syndrome.
It is intriguing to consider that the benefits of losartan might possibly also extend to patients with thoracic aortic disease in general, a worthy topic for future investigation.
Dr. John Gordon Harold is with Cedars-Sinai Heart Institute, Los Angeles, and president of the American College of Cardiology. He had no relevant financial disclosures.

|
| Dr. John Gordon Harold |
COMPARE is a very important study whose results are going to mean a paradigm shift for the management of Marfan syndrome.
It is intriguing to consider that the benefits of losartan might possibly also extend to patients with thoracic aortic disease in general, a worthy topic for future investigation.
Dr. John Gordon Harold is with Cedars-Sinai Heart Institute, Los Angeles, and president of the American College of Cardiology. He had no relevant financial disclosures.
AMSTERDAM – Daily losartan significantly slowed the aortic root dilatation rate in adults with Marfan syndrome in a 3-year randomized clinical trial.
"I think we can be positive about this treatment. We can now recommend losartan in clinical practice," Dr. Maarten Groenink said at the annual congress of the European Society of Cardiology.
The COMPARE (Cozaar in Marfan Patients Reduces Aortic Enlargement) trial included 218 patients at all four university Marfan centers in the Netherlands. Patients were randomized to oral losartan at a target dose of 100 mg/day or no losartan in addition to standard-of-care treatment with beta-blockers. Roughly half of the patients in the losartan group were unable to tolerate the full dose of losartan in addition to a beta-blocker; those patients were maintained on losartan at 50 mg/day. Aortic root diameter was measured by MRI at enrollment and after 3 years of prospective follow-up. The aortic dilatation rate was significantly lower in the losartan group than in controls both in the patients with a native root and in those who had undergone aortic root replacement surgery, reported Dr. Groenink, a cardiologist at the Academic Medical Center, Amsterdam.
There were no aortic dissections in the losartan group and two in the control arm. Elective aortic replacement surgery was performed in a similar number of patients in both groups.
Blood pressure was lower in the losartan group, yet blood pressure didn’t correlate with the aortic dilatation rate. Dr. Groenink speculated that losartan’s chief mechanism of benefit in Marfan syndrome is its ability to curb overexpression of transforming growth factor-beta, which weakens the structure of the media layer of the aortic wall. He said it’s unknown whether losartan’s benefits are specific to that drug or are a class effect.
Ongoing clinical trials are evaluating losartan in children and adolescents with Marfan syndrome, he said, adding that there is a solid rationale for beginning treatment as early in life as possible. "I believe the adverse effects on the aortic wall in Marfan syndrome are caused by the fibrillin defect but also by wear and tear due to cyclic stress by the beating heart. So you can hypothesize that the earlier you start treatment, the better," he explained.
The prevalence of Marfan syndrome has been estimated at 1 in 5,000, but Dr. Groenink suspects the syndrome may actually be more common than that.
Simultaneous with Dr. Groenink’s presentation at the ESC, the COMPARE results were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht334]).
Dr. Groenink reported having no disclosures.
AMSTERDAM – Daily losartan significantly slowed the aortic root dilatation rate in adults with Marfan syndrome in a 3-year randomized clinical trial.
"I think we can be positive about this treatment. We can now recommend losartan in clinical practice," Dr. Maarten Groenink said at the annual congress of the European Society of Cardiology.
The COMPARE (Cozaar in Marfan Patients Reduces Aortic Enlargement) trial included 218 patients at all four university Marfan centers in the Netherlands. Patients were randomized to oral losartan at a target dose of 100 mg/day or no losartan in addition to standard-of-care treatment with beta-blockers. Roughly half of the patients in the losartan group were unable to tolerate the full dose of losartan in addition to a beta-blocker; those patients were maintained on losartan at 50 mg/day. Aortic root diameter was measured by MRI at enrollment and after 3 years of prospective follow-up. The aortic dilatation rate was significantly lower in the losartan group than in controls both in the patients with a native root and in those who had undergone aortic root replacement surgery, reported Dr. Groenink, a cardiologist at the Academic Medical Center, Amsterdam.
There were no aortic dissections in the losartan group and two in the control arm. Elective aortic replacement surgery was performed in a similar number of patients in both groups.
Blood pressure was lower in the losartan group, yet blood pressure didn’t correlate with the aortic dilatation rate. Dr. Groenink speculated that losartan’s chief mechanism of benefit in Marfan syndrome is its ability to curb overexpression of transforming growth factor-beta, which weakens the structure of the media layer of the aortic wall. He said it’s unknown whether losartan’s benefits are specific to that drug or are a class effect.
Ongoing clinical trials are evaluating losartan in children and adolescents with Marfan syndrome, he said, adding that there is a solid rationale for beginning treatment as early in life as possible. "I believe the adverse effects on the aortic wall in Marfan syndrome are caused by the fibrillin defect but also by wear and tear due to cyclic stress by the beating heart. So you can hypothesize that the earlier you start treatment, the better," he explained.
The prevalence of Marfan syndrome has been estimated at 1 in 5,000, but Dr. Groenink suspects the syndrome may actually be more common than that.
Simultaneous with Dr. Groenink’s presentation at the ESC, the COMPARE results were published online (Eur. Heart J. 2013 [doi:10.1093/eurheartj/eht334]).
Dr. Groenink reported having no disclosures.
Major finding: The rate of aortic root enlargement during 3 years of prospective follow-up was 0.77 mm in losartan-treated patients with Marfan syndrome, significantly less than the 1.35 mm in patients on standard-of-care treatment with no losartan.
Data source: The COMPARE trial was a randomized, prospective, open-label multicenter study in which 218 patients with Marfan syndrome were randomized to losartan at a target dose of 100 mg or to no losartan and followed for 3 years with the aortic root dilatation rate as measured by MRI the primary endpoint.
Disclosures: The COMPARE trial was supported by the Dutch Heart Association. Dr. Groenink reported having no financial conflicts.
Unsuspected Primary Hyperparathyroidism Common in Fibromyalgia Patients
SAN DIEGO – The prevalence of unsuspected primary hyperparathyroidism among patients diagnosed with fibromyalgia was 11% in a small exploratory study, Dr. Michael Tsoukas reported at the annual meeting of the American College of Rheumatology.
That’s roughly 100-fold greater than the prevalence of primary hyperparathyroidism in the general population, noted Dr. Tsoukas of McGill University, Montreal.
The classic symptoms of primary hyperparathyroidism include musculoskeletal pain, fatigue, mood disorders, and sleep disturbances, a clinical picture closely mimicking that of fibromyalgia, he observed.
Dr. Tsoukas presented a retrospective study of a convenience sample of 38 consecutive patients with a primary diagnosis of fibromyalgia attending a multidisciplinary tertiary care pain center where physicians obtained routine blood tests. Four of the 38, or 11%, met criteria for the biochemical diagnosis of hyperthyroidism. Two had what’s known as normohormonal hyperparathyroidism, marked by a serum ionized calcium elevated above 1.32 mmol/L or 5.3 mg/dL, with inappropriately high parathyroid hormone levels. The other two had a parathyroid hormone level in excess of 9.3 pmol/L or 93 pg/mL, with inappropriately nonsuppressed calcium.
This was a pilot study. Four cases of unsuspected hyperparathyroidism hardly make for a definitive study. But this was the first study to look at hyperparathyroidism in fibromyalgia patients, and given how common fibromyalgia is and the difficulties and frustrations often encountered in its treatment, these intriguing preliminary findings warrant large, prospective, multicenter studies to further clarify the relationship between these two disorders with closely similar symptoms, Dr. Tsoukas concluded.
He reported having no financial conflicts of interest in this unfunded study.
SAN DIEGO – The prevalence of unsuspected primary hyperparathyroidism among patients diagnosed with fibromyalgia was 11% in a small exploratory study, Dr. Michael Tsoukas reported at the annual meeting of the American College of Rheumatology.
That’s roughly 100-fold greater than the prevalence of primary hyperparathyroidism in the general population, noted Dr. Tsoukas of McGill University, Montreal.
The classic symptoms of primary hyperparathyroidism include musculoskeletal pain, fatigue, mood disorders, and sleep disturbances, a clinical picture closely mimicking that of fibromyalgia, he observed.
Dr. Tsoukas presented a retrospective study of a convenience sample of 38 consecutive patients with a primary diagnosis of fibromyalgia attending a multidisciplinary tertiary care pain center where physicians obtained routine blood tests. Four of the 38, or 11%, met criteria for the biochemical diagnosis of hyperthyroidism. Two had what’s known as normohormonal hyperparathyroidism, marked by a serum ionized calcium elevated above 1.32 mmol/L or 5.3 mg/dL, with inappropriately high parathyroid hormone levels. The other two had a parathyroid hormone level in excess of 9.3 pmol/L or 93 pg/mL, with inappropriately nonsuppressed calcium.
This was a pilot study. Four cases of unsuspected hyperparathyroidism hardly make for a definitive study. But this was the first study to look at hyperparathyroidism in fibromyalgia patients, and given how common fibromyalgia is and the difficulties and frustrations often encountered in its treatment, these intriguing preliminary findings warrant large, prospective, multicenter studies to further clarify the relationship between these two disorders with closely similar symptoms, Dr. Tsoukas concluded.
He reported having no financial conflicts of interest in this unfunded study.
SAN DIEGO – The prevalence of unsuspected primary hyperparathyroidism among patients diagnosed with fibromyalgia was 11% in a small exploratory study, Dr. Michael Tsoukas reported at the annual meeting of the American College of Rheumatology.
That’s roughly 100-fold greater than the prevalence of primary hyperparathyroidism in the general population, noted Dr. Tsoukas of McGill University, Montreal.
The classic symptoms of primary hyperparathyroidism include musculoskeletal pain, fatigue, mood disorders, and sleep disturbances, a clinical picture closely mimicking that of fibromyalgia, he observed.
Dr. Tsoukas presented a retrospective study of a convenience sample of 38 consecutive patients with a primary diagnosis of fibromyalgia attending a multidisciplinary tertiary care pain center where physicians obtained routine blood tests. Four of the 38, or 11%, met criteria for the biochemical diagnosis of hyperthyroidism. Two had what’s known as normohormonal hyperparathyroidism, marked by a serum ionized calcium elevated above 1.32 mmol/L or 5.3 mg/dL, with inappropriately high parathyroid hormone levels. The other two had a parathyroid hormone level in excess of 9.3 pmol/L or 93 pg/mL, with inappropriately nonsuppressed calcium.
This was a pilot study. Four cases of unsuspected hyperparathyroidism hardly make for a definitive study. But this was the first study to look at hyperparathyroidism in fibromyalgia patients, and given how common fibromyalgia is and the difficulties and frustrations often encountered in its treatment, these intriguing preliminary findings warrant large, prospective, multicenter studies to further clarify the relationship between these two disorders with closely similar symptoms, Dr. Tsoukas concluded.
He reported having no financial conflicts of interest in this unfunded study.
AT THE ACR ANNUAL MEETING
Unsuspected primary hyperparathyroidism common in fibromyalgia patients
SAN DIEGO – The prevalence of unsuspected primary hyperparathyroidism among patients diagnosed with fibromyalgia was 11% in a small exploratory study, Dr. Michael Tsoukas reported at the annual meeting of the American College of Rheumatology.
That’s roughly 100-fold greater than the prevalence of primary hyperparathyroidism in the general population, noted Dr. Tsoukas of McGill University, Montreal.
The classic symptoms of primary hyperparathyroidism include musculoskeletal pain, fatigue, mood disorders, and sleep disturbances, a clinical picture closely mimicking that of fibromyalgia, he observed.
Dr. Tsoukas presented a retrospective study of a convenience sample of 38 consecutive patients with a primary diagnosis of fibromyalgia attending a multidisciplinary tertiary care pain center where physicians obtained routine blood tests. Four of the 38, or 11%, met criteria for the biochemical diagnosis of hyperthyroidism. Two had what’s known as normohormonal hyperparathyroidism, marked by a serum ionized calcium elevated above 1.32 mmol/L or 5.3 mg/dL, with inappropriately high parathyroid hormone levels. The other two had a parathyroid hormone level in excess of 9.3 pmol/L or 93 pg/mL, with inappropriately nonsuppressed calcium.
This was a pilot study. Four cases of unsuspected hyperparathyroidism hardly make for a definitive study. But this was the first study to look at hyperparathyroidism in fibromyalgia patients, and given how common fibromyalgia is and the difficulties and frustrations often encountered in its treatment, these intriguing preliminary findings warrant large, prospective, multicenter studies to further clarify the relationship between these two disorders with closely similar symptoms, Dr. Tsoukas concluded.
He reported having no financial conflicts of interest in this unfunded study.
SAN DIEGO – The prevalence of unsuspected primary hyperparathyroidism among patients diagnosed with fibromyalgia was 11% in a small exploratory study, Dr. Michael Tsoukas reported at the annual meeting of the American College of Rheumatology.
That’s roughly 100-fold greater than the prevalence of primary hyperparathyroidism in the general population, noted Dr. Tsoukas of McGill University, Montreal.
The classic symptoms of primary hyperparathyroidism include musculoskeletal pain, fatigue, mood disorders, and sleep disturbances, a clinical picture closely mimicking that of fibromyalgia, he observed.
Dr. Tsoukas presented a retrospective study of a convenience sample of 38 consecutive patients with a primary diagnosis of fibromyalgia attending a multidisciplinary tertiary care pain center where physicians obtained routine blood tests. Four of the 38, or 11%, met criteria for the biochemical diagnosis of hyperthyroidism. Two had what’s known as normohormonal hyperparathyroidism, marked by a serum ionized calcium elevated above 1.32 mmol/L or 5.3 mg/dL, with inappropriately high parathyroid hormone levels. The other two had a parathyroid hormone level in excess of 9.3 pmol/L or 93 pg/mL, with inappropriately nonsuppressed calcium.
This was a pilot study. Four cases of unsuspected hyperparathyroidism hardly make for a definitive study. But this was the first study to look at hyperparathyroidism in fibromyalgia patients, and given how common fibromyalgia is and the difficulties and frustrations often encountered in its treatment, these intriguing preliminary findings warrant large, prospective, multicenter studies to further clarify the relationship between these two disorders with closely similar symptoms, Dr. Tsoukas concluded.
He reported having no financial conflicts of interest in this unfunded study.
SAN DIEGO – The prevalence of unsuspected primary hyperparathyroidism among patients diagnosed with fibromyalgia was 11% in a small exploratory study, Dr. Michael Tsoukas reported at the annual meeting of the American College of Rheumatology.
That’s roughly 100-fold greater than the prevalence of primary hyperparathyroidism in the general population, noted Dr. Tsoukas of McGill University, Montreal.
The classic symptoms of primary hyperparathyroidism include musculoskeletal pain, fatigue, mood disorders, and sleep disturbances, a clinical picture closely mimicking that of fibromyalgia, he observed.
Dr. Tsoukas presented a retrospective study of a convenience sample of 38 consecutive patients with a primary diagnosis of fibromyalgia attending a multidisciplinary tertiary care pain center where physicians obtained routine blood tests. Four of the 38, or 11%, met criteria for the biochemical diagnosis of hyperthyroidism. Two had what’s known as normohormonal hyperparathyroidism, marked by a serum ionized calcium elevated above 1.32 mmol/L or 5.3 mg/dL, with inappropriately high parathyroid hormone levels. The other two had a parathyroid hormone level in excess of 9.3 pmol/L or 93 pg/mL, with inappropriately nonsuppressed calcium.
This was a pilot study. Four cases of unsuspected hyperparathyroidism hardly make for a definitive study. But this was the first study to look at hyperparathyroidism in fibromyalgia patients, and given how common fibromyalgia is and the difficulties and frustrations often encountered in its treatment, these intriguing preliminary findings warrant large, prospective, multicenter studies to further clarify the relationship between these two disorders with closely similar symptoms, Dr. Tsoukas concluded.
He reported having no financial conflicts of interest in this unfunded study.
AT THE ACR ANNUAL MEETING
Major finding: Four of 38 patients with a primary diagnosis of fibromyalgia proved to have primary hyperparathyroidism. That 11% prevalence is roughly 100 times greater than in the general population.
Data source: This was a chart review involving a convenience sample of 38 consecutive patients with a primary diagnosis of fibromyalgia attending a multidisciplinary tertiary care pain clinic.
Disclosures: Dr. Tsoukas reported having no financial conflicts of interest in this unfunded study.
Encouraging results for oral JAK 3 inhibitor in rheumatoid arthritis
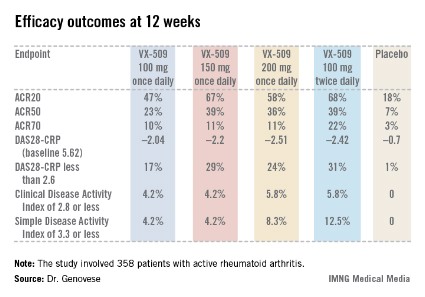
SAN DIEGO – A novel oral Janus kinase 3 inhibitor known for now as VX-509 readily hit both of its coprimary endpoints in a phase IIb study in rheumatoid arthritis patients presented at the annual meeting of the American College of Rheumatology.
The 24-week, double-blind, international study included 358 patients with active rheumatoid arthritis on stable doses of background methotrexate who were randomized to one of four VX-509 dosing regimens or placebo.
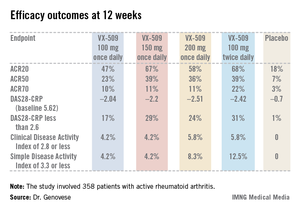
An ACR20 response at 12 weeks occurred in 47%-68% of subjects on various doses of the oral JAK 3 inhibitor, compared with 18% of controls. Robust improvements in the Disease Activity Score 28 C-reactive protein (DAS28-CRP) were also noted in the VX-509-treated patients. The improvement in these two primary endpoints was rapid, with the oral JAK 3 inhibitor’s advantage over placebo becoming significant during the first week, reported Dr. Mark C. Genovese, professor of medicine at Stanford (Calif.) University.
Infections, the most common type of adverse events, occurred in 22% of patients on VX-509 and 15.5% on placebo. Serious infections occurred in 2.8% of patients on the JAK 3 inhibitor, twice the rate observed in controls. Bronchitis, pneumonia, cellulitis, and one severe case of herpes zoster accounted for most of the serious infections in the VX-509 group.
Modest elevations in transaminase levels and reductions in median neutrophil and lymphocyte counts occurred in the VX-509 groups. In addition, dose-dependent increases were observed in low-density lipoprotein (LDL) cholesterol and fasting triglycerides. The LDL increases ranged from 3.1 to 11.6 mg/dL, while triglyceride levels in VX-509–treated patients climbed by 25.7-39 mg/dL.
JAK 3 is exclusively involved in immune function and prevents signaling by inflammatory cytokines, including interleukin-2, -4, -7, -9, -15, and -21. VX-509’s high degree of selectivity for JAK 3 is desirable, Dr. Genovese explained, because the drug doesn’t target JAK 2, which is involved growth factor and hematopoietic signaling and whose inhibition could thereby lead to unwanted effects.
Vertex Pharmaceuticals, which sponsored the phase IIb study, subsequently announced its strong interest in further developing VX-509 for the marketplace. Dr. Genovese disclosed having received research grants from and serving as a consultant to Vertex.
SAN DIEGO – A novel oral Janus kinase 3 inhibitor known for now as VX-509 readily hit both of its coprimary endpoints in a phase IIb study in rheumatoid arthritis patients presented at the annual meeting of the American College of Rheumatology.
The 24-week, double-blind, international study included 358 patients with active rheumatoid arthritis on stable doses of background methotrexate who were randomized to one of four VX-509 dosing regimens or placebo.

An ACR20 response at 12 weeks occurred in 47%-68% of subjects on various doses of the oral JAK 3 inhibitor, compared with 18% of controls. Robust improvements in the Disease Activity Score 28 C-reactive protein (DAS28-CRP) were also noted in the VX-509-treated patients. The improvement in these two primary endpoints was rapid, with the oral JAK 3 inhibitor’s advantage over placebo becoming significant during the first week, reported Dr. Mark C. Genovese, professor of medicine at Stanford (Calif.) University.
Infections, the most common type of adverse events, occurred in 22% of patients on VX-509 and 15.5% on placebo. Serious infections occurred in 2.8% of patients on the JAK 3 inhibitor, twice the rate observed in controls. Bronchitis, pneumonia, cellulitis, and one severe case of herpes zoster accounted for most of the serious infections in the VX-509 group.
Modest elevations in transaminase levels and reductions in median neutrophil and lymphocyte counts occurred in the VX-509 groups. In addition, dose-dependent increases were observed in low-density lipoprotein (LDL) cholesterol and fasting triglycerides. The LDL increases ranged from 3.1 to 11.6 mg/dL, while triglyceride levels in VX-509–treated patients climbed by 25.7-39 mg/dL.
JAK 3 is exclusively involved in immune function and prevents signaling by inflammatory cytokines, including interleukin-2, -4, -7, -9, -15, and -21. VX-509’s high degree of selectivity for JAK 3 is desirable, Dr. Genovese explained, because the drug doesn’t target JAK 2, which is involved growth factor and hematopoietic signaling and whose inhibition could thereby lead to unwanted effects.
Vertex Pharmaceuticals, which sponsored the phase IIb study, subsequently announced its strong interest in further developing VX-509 for the marketplace. Dr. Genovese disclosed having received research grants from and serving as a consultant to Vertex.
SAN DIEGO – A novel oral Janus kinase 3 inhibitor known for now as VX-509 readily hit both of its coprimary endpoints in a phase IIb study in rheumatoid arthritis patients presented at the annual meeting of the American College of Rheumatology.
The 24-week, double-blind, international study included 358 patients with active rheumatoid arthritis on stable doses of background methotrexate who were randomized to one of four VX-509 dosing regimens or placebo.

An ACR20 response at 12 weeks occurred in 47%-68% of subjects on various doses of the oral JAK 3 inhibitor, compared with 18% of controls. Robust improvements in the Disease Activity Score 28 C-reactive protein (DAS28-CRP) were also noted in the VX-509-treated patients. The improvement in these two primary endpoints was rapid, with the oral JAK 3 inhibitor’s advantage over placebo becoming significant during the first week, reported Dr. Mark C. Genovese, professor of medicine at Stanford (Calif.) University.
Infections, the most common type of adverse events, occurred in 22% of patients on VX-509 and 15.5% on placebo. Serious infections occurred in 2.8% of patients on the JAK 3 inhibitor, twice the rate observed in controls. Bronchitis, pneumonia, cellulitis, and one severe case of herpes zoster accounted for most of the serious infections in the VX-509 group.
Modest elevations in transaminase levels and reductions in median neutrophil and lymphocyte counts occurred in the VX-509 groups. In addition, dose-dependent increases were observed in low-density lipoprotein (LDL) cholesterol and fasting triglycerides. The LDL increases ranged from 3.1 to 11.6 mg/dL, while triglyceride levels in VX-509–treated patients climbed by 25.7-39 mg/dL.
JAK 3 is exclusively involved in immune function and prevents signaling by inflammatory cytokines, including interleukin-2, -4, -7, -9, -15, and -21. VX-509’s high degree of selectivity for JAK 3 is desirable, Dr. Genovese explained, because the drug doesn’t target JAK 2, which is involved growth factor and hematopoietic signaling and whose inhibition could thereby lead to unwanted effects.
Vertex Pharmaceuticals, which sponsored the phase IIb study, subsequently announced its strong interest in further developing VX-509 for the marketplace. Dr. Genovese disclosed having received research grants from and serving as a consultant to Vertex.
AT THE ACR ANNUAL MEETING
Major finding: Rheumatoid arthritis patients had ACR20 responses of 47%-68% to various dosing regimens of the novel oral Janus kinase 3 inhibitor VX-509 at 12 weeks, compared with 18% in placebo-treated controls.
Data source: This was a phase IIb, double-blind, placebo-controlled, international study involving 358 patients with rheumatoid arthritis on background stable doses of methotrexate, continued during the trial.
Disclosures: The study was sponsored by Vertex Pharmaceuticals. The presenter disclosed receiving research grants from and serving as a consultant to Vertex.
Two new trials bolster omalizumab for urticaria
ISTANBUL, TURKEY – The case for omalizumab as a novel, safe, and highly effective therapy for H1-antihistamine-refractory chronic idiopathic/spontaneous urticaria received a boost in the form of two new phase III clinical trials presented at the annual congress of the European Academy of Dermatology and Venereology.
The two new multicenter, randomized, double-blind, placebo-controlled studies join another trial published earlier this year (N. Engl. J. Med. 2013;368:924-35). Together, the three trials total nearly 1,000 participants aged 12-75 years who remained severely affected by chronic idiopathic/spontaneous urticaria despite treatment with nonsedating H1-antihistamines, the sole approved form of treatment.
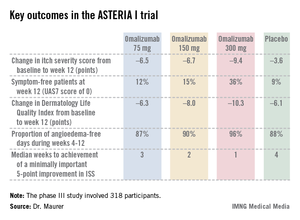
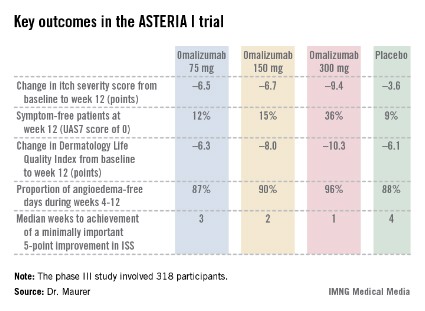
The GLACIAL trial, one of the new studies, set the bar even higher: Participants not only had to be refractory to H1-antihistamines at up to four times the approved dose, but also to off-label H2-antihistamines and/or leukotriene receptor antagonists.
The results were highly consistent across all three clinical trials – 80%-90% of patients demonstrated a dramatic clinical response to omalizumab in dose-dependent fashion. The top dose studied, 300 mg per subcutaneous injection once every 4 weeks, brought the greatest reductions in itching, wheals, and days with angioedema, Dr. Marcus Maurer at the meeting, where he presented the phase III data from the GLACIAL and ASTERIA I trials.
"Omalizumab is the new hope in urticaria treatment. The response is super-fast. We saw a massive and rapid decline in urticaria activity as early as in the first days after the first injection," he said. "In fact, the vast majority of responders respond within the first week of treatment. The disease activity then stays down as long as treatment continues," noted Dr. Maurer, professor of dermatology and allergy at Charité University Hospital, Berlin.
"The other important thing is, as soon as you stop the drug, you stop the benefit. Patients go back to baseline," he added. "So omalizumab is a very effective way to control disease activity, but it’s no cure. It doesn’t make the disease go away, it makes the symptoms go away. There is spontaneous remission in chronic idiopathic/spontaneous urticaria, but until that happens, patients need treatment that makes them free of symptoms. I’ve had patients on omalizumab for 5-6 years now."
As for the safety profile, Dr. Maurer called it "boring."
"This is a safe drug. This is a drug that doesn’t have any differences in its safety profile compared to placebo. The number and types of adverse events are virtually identical," he said.
Less than a week after Dr. Maurer’s presentation of the GLACIAL and ASTERIA I phase III trial results in Istanbul, Genentech filed an application with the Food and Drug Administration requesting approval of a supplemental indication for omalizumab in the treatment of chronic idiopathic/spontaneous urticaria in patients who remain symptomatic despite H1-antihistamine therapy at approved doses. A decision by the agency is expected in the spring of 2014.
Omalizumab is a humanized anti-IgE monoclonal antibody already approved as Xolair for treatment of moderate to severe allergic asthma. Unlike in allergic asthma, however, the response of chronic urticaria to omalizumab is independent of serum IgE level. Indeed, IgE levels are only modestly elevated in patients with chronic idiopathic/spontaneous urticaria – nothing like the high levels that can occur in allergic asthma. It’s not useful to measure IgE as a guide to the likelihood that chronic idiopathic/spontaneous urticaria will respond to omalizumab, Dr. Maurer advised.
The ASTERIA I trial was arguably the most clinically relevant of the three phase III studies because it employed omalizumab as add-on therapy to H1-antihistamines at approved doses. The 318 participants had a baseline mean weekly Itch Severity Scale (ISS) score of 14.3 out of a possible 21 despite being on H1-antihistamines. Their baseline urticaria activity score over 7 days (UAS7) was 31.1 on a scale of 0-42.
The study population was typical for patients with moderate to severe chronic idiopathic/spontaneous urticaria refractory to first-line therapy with H1-antihistamines. The average age was 41 years, three-quarters were female, and the mean body mass index was 29 kg/m2. The patients were diagnosed with chronic idiopathic/spontaneous urticaria an average of 4 years earlier and had been on an average of five previous unsuccessful medications for the condition. Half of the patients had angioedema.
Study participants received six subcutaneous doses of omalizumab at either 75-, 150-, or 300-mg doses; or placebo at 4-week intervals. They were then observed for 16 weeks. The primary study endpoint was the change in weekly ISS from baseline to week 12. The mean reduction was 3.6 points in the control group, significantly less than the 6.5-point fall in the low-dose omalizumab arm, the 6.7-point reduction with omalizumab 150 mg, or the 9.4-point decrease with omalizumab at 300 mg.
Omalizumab-treated patients also fared significantly better than did the placebo group in all secondary endpoints.
The anti-IgE biologic was well tolerated, with no new safety signals arising in ASTERIA I or the other two phase III trials beyond the agent’s well-established safety profile in treating allergic asthma.
The diagnosis of chronic idiopathic/spontaneous urticaria is based upon the finding of itchy hives and/or angioedema for 6 weeks or more with no specific external trigger. The estimated prevalence in the general population is 0.5%-1%. More than half of affected patients continue to experience symptoms despite treatment with H1-antihistamines.
The phase III trials were sponsored by Genentech and Novartis, which are jointly developing omalizumab for the treatment of chronic idiopathic/spontaneous urticaria. Dr. Maurer has received research grants from, and served on the advisory boards of, those pharmaceutical companies, as well as more than a dozen others.
ISTANBUL, TURKEY – The case for omalizumab as a novel, safe, and highly effective therapy for H1-antihistamine-refractory chronic idiopathic/spontaneous urticaria received a boost in the form of two new phase III clinical trials presented at the annual congress of the European Academy of Dermatology and Venereology.
The two new multicenter, randomized, double-blind, placebo-controlled studies join another trial published earlier this year (N. Engl. J. Med. 2013;368:924-35). Together, the three trials total nearly 1,000 participants aged 12-75 years who remained severely affected by chronic idiopathic/spontaneous urticaria despite treatment with nonsedating H1-antihistamines, the sole approved form of treatment.

The GLACIAL trial, one of the new studies, set the bar even higher: Participants not only had to be refractory to H1-antihistamines at up to four times the approved dose, but also to off-label H2-antihistamines and/or leukotriene receptor antagonists.
The results were highly consistent across all three clinical trials – 80%-90% of patients demonstrated a dramatic clinical response to omalizumab in dose-dependent fashion. The top dose studied, 300 mg per subcutaneous injection once every 4 weeks, brought the greatest reductions in itching, wheals, and days with angioedema, Dr. Marcus Maurer at the meeting, where he presented the phase III data from the GLACIAL and ASTERIA I trials.
"Omalizumab is the new hope in urticaria treatment. The response is super-fast. We saw a massive and rapid decline in urticaria activity as early as in the first days after the first injection," he said. "In fact, the vast majority of responders respond within the first week of treatment. The disease activity then stays down as long as treatment continues," noted Dr. Maurer, professor of dermatology and allergy at Charité University Hospital, Berlin.
"The other important thing is, as soon as you stop the drug, you stop the benefit. Patients go back to baseline," he added. "So omalizumab is a very effective way to control disease activity, but it’s no cure. It doesn’t make the disease go away, it makes the symptoms go away. There is spontaneous remission in chronic idiopathic/spontaneous urticaria, but until that happens, patients need treatment that makes them free of symptoms. I’ve had patients on omalizumab for 5-6 years now."
As for the safety profile, Dr. Maurer called it "boring."
"This is a safe drug. This is a drug that doesn’t have any differences in its safety profile compared to placebo. The number and types of adverse events are virtually identical," he said.
Less than a week after Dr. Maurer’s presentation of the GLACIAL and ASTERIA I phase III trial results in Istanbul, Genentech filed an application with the Food and Drug Administration requesting approval of a supplemental indication for omalizumab in the treatment of chronic idiopathic/spontaneous urticaria in patients who remain symptomatic despite H1-antihistamine therapy at approved doses. A decision by the agency is expected in the spring of 2014.
Omalizumab is a humanized anti-IgE monoclonal antibody already approved as Xolair for treatment of moderate to severe allergic asthma. Unlike in allergic asthma, however, the response of chronic urticaria to omalizumab is independent of serum IgE level. Indeed, IgE levels are only modestly elevated in patients with chronic idiopathic/spontaneous urticaria – nothing like the high levels that can occur in allergic asthma. It’s not useful to measure IgE as a guide to the likelihood that chronic idiopathic/spontaneous urticaria will respond to omalizumab, Dr. Maurer advised.
The ASTERIA I trial was arguably the most clinically relevant of the three phase III studies because it employed omalizumab as add-on therapy to H1-antihistamines at approved doses. The 318 participants had a baseline mean weekly Itch Severity Scale (ISS) score of 14.3 out of a possible 21 despite being on H1-antihistamines. Their baseline urticaria activity score over 7 days (UAS7) was 31.1 on a scale of 0-42.
The study population was typical for patients with moderate to severe chronic idiopathic/spontaneous urticaria refractory to first-line therapy with H1-antihistamines. The average age was 41 years, three-quarters were female, and the mean body mass index was 29 kg/m2. The patients were diagnosed with chronic idiopathic/spontaneous urticaria an average of 4 years earlier and had been on an average of five previous unsuccessful medications for the condition. Half of the patients had angioedema.
Study participants received six subcutaneous doses of omalizumab at either 75-, 150-, or 300-mg doses; or placebo at 4-week intervals. They were then observed for 16 weeks. The primary study endpoint was the change in weekly ISS from baseline to week 12. The mean reduction was 3.6 points in the control group, significantly less than the 6.5-point fall in the low-dose omalizumab arm, the 6.7-point reduction with omalizumab 150 mg, or the 9.4-point decrease with omalizumab at 300 mg.
Omalizumab-treated patients also fared significantly better than did the placebo group in all secondary endpoints.
The anti-IgE biologic was well tolerated, with no new safety signals arising in ASTERIA I or the other two phase III trials beyond the agent’s well-established safety profile in treating allergic asthma.
The diagnosis of chronic idiopathic/spontaneous urticaria is based upon the finding of itchy hives and/or angioedema for 6 weeks or more with no specific external trigger. The estimated prevalence in the general population is 0.5%-1%. More than half of affected patients continue to experience symptoms despite treatment with H1-antihistamines.
The phase III trials were sponsored by Genentech and Novartis, which are jointly developing omalizumab for the treatment of chronic idiopathic/spontaneous urticaria. Dr. Maurer has received research grants from, and served on the advisory boards of, those pharmaceutical companies, as well as more than a dozen others.
ISTANBUL, TURKEY – The case for omalizumab as a novel, safe, and highly effective therapy for H1-antihistamine-refractory chronic idiopathic/spontaneous urticaria received a boost in the form of two new phase III clinical trials presented at the annual congress of the European Academy of Dermatology and Venereology.
The two new multicenter, randomized, double-blind, placebo-controlled studies join another trial published earlier this year (N. Engl. J. Med. 2013;368:924-35). Together, the three trials total nearly 1,000 participants aged 12-75 years who remained severely affected by chronic idiopathic/spontaneous urticaria despite treatment with nonsedating H1-antihistamines, the sole approved form of treatment.

The GLACIAL trial, one of the new studies, set the bar even higher: Participants not only had to be refractory to H1-antihistamines at up to four times the approved dose, but also to off-label H2-antihistamines and/or leukotriene receptor antagonists.
The results were highly consistent across all three clinical trials – 80%-90% of patients demonstrated a dramatic clinical response to omalizumab in dose-dependent fashion. The top dose studied, 300 mg per subcutaneous injection once every 4 weeks, brought the greatest reductions in itching, wheals, and days with angioedema, Dr. Marcus Maurer at the meeting, where he presented the phase III data from the GLACIAL and ASTERIA I trials.
"Omalizumab is the new hope in urticaria treatment. The response is super-fast. We saw a massive and rapid decline in urticaria activity as early as in the first days after the first injection," he said. "In fact, the vast majority of responders respond within the first week of treatment. The disease activity then stays down as long as treatment continues," noted Dr. Maurer, professor of dermatology and allergy at Charité University Hospital, Berlin.
"The other important thing is, as soon as you stop the drug, you stop the benefit. Patients go back to baseline," he added. "So omalizumab is a very effective way to control disease activity, but it’s no cure. It doesn’t make the disease go away, it makes the symptoms go away. There is spontaneous remission in chronic idiopathic/spontaneous urticaria, but until that happens, patients need treatment that makes them free of symptoms. I’ve had patients on omalizumab for 5-6 years now."
As for the safety profile, Dr. Maurer called it "boring."
"This is a safe drug. This is a drug that doesn’t have any differences in its safety profile compared to placebo. The number and types of adverse events are virtually identical," he said.
Less than a week after Dr. Maurer’s presentation of the GLACIAL and ASTERIA I phase III trial results in Istanbul, Genentech filed an application with the Food and Drug Administration requesting approval of a supplemental indication for omalizumab in the treatment of chronic idiopathic/spontaneous urticaria in patients who remain symptomatic despite H1-antihistamine therapy at approved doses. A decision by the agency is expected in the spring of 2014.
Omalizumab is a humanized anti-IgE monoclonal antibody already approved as Xolair for treatment of moderate to severe allergic asthma. Unlike in allergic asthma, however, the response of chronic urticaria to omalizumab is independent of serum IgE level. Indeed, IgE levels are only modestly elevated in patients with chronic idiopathic/spontaneous urticaria – nothing like the high levels that can occur in allergic asthma. It’s not useful to measure IgE as a guide to the likelihood that chronic idiopathic/spontaneous urticaria will respond to omalizumab, Dr. Maurer advised.
The ASTERIA I trial was arguably the most clinically relevant of the three phase III studies because it employed omalizumab as add-on therapy to H1-antihistamines at approved doses. The 318 participants had a baseline mean weekly Itch Severity Scale (ISS) score of 14.3 out of a possible 21 despite being on H1-antihistamines. Their baseline urticaria activity score over 7 days (UAS7) was 31.1 on a scale of 0-42.
The study population was typical for patients with moderate to severe chronic idiopathic/spontaneous urticaria refractory to first-line therapy with H1-antihistamines. The average age was 41 years, three-quarters were female, and the mean body mass index was 29 kg/m2. The patients were diagnosed with chronic idiopathic/spontaneous urticaria an average of 4 years earlier and had been on an average of five previous unsuccessful medications for the condition. Half of the patients had angioedema.
Study participants received six subcutaneous doses of omalizumab at either 75-, 150-, or 300-mg doses; or placebo at 4-week intervals. They were then observed for 16 weeks. The primary study endpoint was the change in weekly ISS from baseline to week 12. The mean reduction was 3.6 points in the control group, significantly less than the 6.5-point fall in the low-dose omalizumab arm, the 6.7-point reduction with omalizumab 150 mg, or the 9.4-point decrease with omalizumab at 300 mg.
Omalizumab-treated patients also fared significantly better than did the placebo group in all secondary endpoints.
The anti-IgE biologic was well tolerated, with no new safety signals arising in ASTERIA I or the other two phase III trials beyond the agent’s well-established safety profile in treating allergic asthma.
The diagnosis of chronic idiopathic/spontaneous urticaria is based upon the finding of itchy hives and/or angioedema for 6 weeks or more with no specific external trigger. The estimated prevalence in the general population is 0.5%-1%. More than half of affected patients continue to experience symptoms despite treatment with H1-antihistamines.
The phase III trials were sponsored by Genentech and Novartis, which are jointly developing omalizumab for the treatment of chronic idiopathic/spontaneous urticaria. Dr. Maurer has received research grants from, and served on the advisory boards of, those pharmaceutical companies, as well as more than a dozen others.
AT THE EADV CONGRESS
Major finding: Among 318 patients with chronic idiopathic/spontaneous urticaria refractory to H1-antihistamines at standard doses who participated in the phase III ASTERIA I trial, add-on omalizumab at 300 mg resulted in a mean 9.4-point reduction in itch severity score at week 12 compared to a 3.6-point drop with placebo from a baseline score of 14.3.
Data source: ASTERIA I, one of three recent phase III randomized, double-blind, multicenter, placebo-controlled clinical trials of omalizumab for chronic idiopathic/spontaneous urticaria totaling nearly 1,000 participants.
Disclosures: The phase III clinical trials were jointly sponsored by Genentech and Novartis. The presenter reported having received research funds from the two companies as well as serving on their advisory boards.