User login
Endofibrinolysis a 'game changer' in acute PE
WASHINGTON – Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive or submassive pulmonary embolism significantly improves right ventricular function, reduces pulmonary hypertension and angiographic evidence of obstruction, and lessens the risk of fibrinolysis-associated intracranial hemorrhage, according to a prospective multicenter clinical trial.
"By minimizing the risk of intracranial bleeding, ultrasound-facilitated, catheter-directed, low-dose fibrinolysis represents a potential game changer in the treatment of high-risk pulmonary embolism patients," Dr. Gregory Piazza said in presenting the results of the SEATTLE II study at the annual meeting of the American College of Cardiology.
Full-dose systemic fibrinolysis has long been the go-to advanced therapy for high-risk pulmonary embolism (PE), but physicians are leery of the associated 2%-3% risk of catastrophic intracranial hemorrhage, noted Dr. Piazza of Brigham and Women’s Hospital and Harvard University, Boston.
SEATTLE II was a single-arm, 21-site, prospective study in which 150 patients with high-risk PE underwent treatment using the commercially available EKOS EkoSonic Endovascular System.
Twenty-one percent of patients had massive PE, defined as presentation with syncope, cardiogenic shock, resuscitated cardiac arrest, or persistent hypotension. The remaining 79% had submassive PE, with normal blood pressure but evidence of right ventricular dysfunction. All participants had to have a right ventricular/left ventricular ratio (RV/LV) of 0.9 or greater on the same chest CT scan used in diagnosing the PE. This CT documentation of RV dysfunction has been associated in a meta-analysis of patients with submassive PE with a 7.4-fold increased risk of death from PE, compared with normotensive PE patients with normal RV function (J. Thromb. Haemost. 2013;11:1823-32).
The primary endpoint was change in RV/LV on chest CT from baseline to 48 hours after initiation of fibrinolysis. This ratio improved from 1.55 to 1.13, for a statistically and clinically significant 27% reduction. A similar-size improvement was seen in pulmonary artery systolic pressure – a secondary efficacy endpoint – which decreased from 51.4 mm Hg before treatment to 37.5 mm Hg post procedure and 36.9 mm Hg at 48 hours. Both efficacy endpoints improved to a similar extent regardless of whether patients had massive or submassive PE.
The mean Modified Miller Pulmonary Artery Angiographic Obstruction Score improved by 30%, from 22.5 pretreatment to 15.8 at 48 hours.
Three in-hospital deaths occurred. One was due to massive PE that occurred before the fibrinolysis procedure could be completed. The others were not directly attributable to PE: One involved overwhelming sepsis and the other was due to progressive respiratory failure. Major bleeding occurred in 11% of patients; however, 16 of the 17 events were classified as GUSTO moderate bleeds, with only a single GUSTO severe hemorrhage.
There were no intracranial hemorrhages.
The fibrinolytic agent used in SEATTLE II was tissue plasminogen activator, delivered at 1 mg/hr for a total dose of 24 mg. Patients with unilateral PE received a single device and 24 hours of infusion time. The 86% of patients who had bilateral disease got two devices and 12 hours of therapy.
The proprietary EKOS system consists of two catheters: an outer infusion catheter with side holes that elute the fibrinolytic agent, and an inner-core catheter with ultrasound transducers placed at regular intervals. These transducers produce low-intensity ultrasound that serves two purposes. Through a process called acoustic streaming, the low-intensity ultrasound helps push the fibrinolytic agent closer to the thrombus. Plus, the ultrasound energy causes the clot fibrin to reconfigure from a tight lattice to a more porous structure that promotes deeper penetration of the fibrinolytic, Dr. Piazza explained.
Dr. Piazza said the next step in defining the EKOS system’s role in clinical practice will be to study briefer infusion times as a means of achieving faster patient improvement with reduced use of hospital resources.
The EKOS system has been approved in the United States since 2005 for treatment of blood clots in the arms and legs. In Europe it gained an additional indication for treatment of massive and submassive PE in 2011.
The SEATTLE II study was sponsored by EKOS Corp. Dr. Piazza reported receiving a research grant from the company.
WASHINGTON – Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive or submassive pulmonary embolism significantly improves right ventricular function, reduces pulmonary hypertension and angiographic evidence of obstruction, and lessens the risk of fibrinolysis-associated intracranial hemorrhage, according to a prospective multicenter clinical trial.
"By minimizing the risk of intracranial bleeding, ultrasound-facilitated, catheter-directed, low-dose fibrinolysis represents a potential game changer in the treatment of high-risk pulmonary embolism patients," Dr. Gregory Piazza said in presenting the results of the SEATTLE II study at the annual meeting of the American College of Cardiology.
Full-dose systemic fibrinolysis has long been the go-to advanced therapy for high-risk pulmonary embolism (PE), but physicians are leery of the associated 2%-3% risk of catastrophic intracranial hemorrhage, noted Dr. Piazza of Brigham and Women’s Hospital and Harvard University, Boston.
SEATTLE II was a single-arm, 21-site, prospective study in which 150 patients with high-risk PE underwent treatment using the commercially available EKOS EkoSonic Endovascular System.
Twenty-one percent of patients had massive PE, defined as presentation with syncope, cardiogenic shock, resuscitated cardiac arrest, or persistent hypotension. The remaining 79% had submassive PE, with normal blood pressure but evidence of right ventricular dysfunction. All participants had to have a right ventricular/left ventricular ratio (RV/LV) of 0.9 or greater on the same chest CT scan used in diagnosing the PE. This CT documentation of RV dysfunction has been associated in a meta-analysis of patients with submassive PE with a 7.4-fold increased risk of death from PE, compared with normotensive PE patients with normal RV function (J. Thromb. Haemost. 2013;11:1823-32).
The primary endpoint was change in RV/LV on chest CT from baseline to 48 hours after initiation of fibrinolysis. This ratio improved from 1.55 to 1.13, for a statistically and clinically significant 27% reduction. A similar-size improvement was seen in pulmonary artery systolic pressure – a secondary efficacy endpoint – which decreased from 51.4 mm Hg before treatment to 37.5 mm Hg post procedure and 36.9 mm Hg at 48 hours. Both efficacy endpoints improved to a similar extent regardless of whether patients had massive or submassive PE.
The mean Modified Miller Pulmonary Artery Angiographic Obstruction Score improved by 30%, from 22.5 pretreatment to 15.8 at 48 hours.
Three in-hospital deaths occurred. One was due to massive PE that occurred before the fibrinolysis procedure could be completed. The others were not directly attributable to PE: One involved overwhelming sepsis and the other was due to progressive respiratory failure. Major bleeding occurred in 11% of patients; however, 16 of the 17 events were classified as GUSTO moderate bleeds, with only a single GUSTO severe hemorrhage.
There were no intracranial hemorrhages.
The fibrinolytic agent used in SEATTLE II was tissue plasminogen activator, delivered at 1 mg/hr for a total dose of 24 mg. Patients with unilateral PE received a single device and 24 hours of infusion time. The 86% of patients who had bilateral disease got two devices and 12 hours of therapy.
The proprietary EKOS system consists of two catheters: an outer infusion catheter with side holes that elute the fibrinolytic agent, and an inner-core catheter with ultrasound transducers placed at regular intervals. These transducers produce low-intensity ultrasound that serves two purposes. Through a process called acoustic streaming, the low-intensity ultrasound helps push the fibrinolytic agent closer to the thrombus. Plus, the ultrasound energy causes the clot fibrin to reconfigure from a tight lattice to a more porous structure that promotes deeper penetration of the fibrinolytic, Dr. Piazza explained.
Dr. Piazza said the next step in defining the EKOS system’s role in clinical practice will be to study briefer infusion times as a means of achieving faster patient improvement with reduced use of hospital resources.
The EKOS system has been approved in the United States since 2005 for treatment of blood clots in the arms and legs. In Europe it gained an additional indication for treatment of massive and submassive PE in 2011.
The SEATTLE II study was sponsored by EKOS Corp. Dr. Piazza reported receiving a research grant from the company.
WASHINGTON – Ultrasound-facilitated, catheter-directed, low-dose fibrinolysis for acute massive or submassive pulmonary embolism significantly improves right ventricular function, reduces pulmonary hypertension and angiographic evidence of obstruction, and lessens the risk of fibrinolysis-associated intracranial hemorrhage, according to a prospective multicenter clinical trial.
"By minimizing the risk of intracranial bleeding, ultrasound-facilitated, catheter-directed, low-dose fibrinolysis represents a potential game changer in the treatment of high-risk pulmonary embolism patients," Dr. Gregory Piazza said in presenting the results of the SEATTLE II study at the annual meeting of the American College of Cardiology.
Full-dose systemic fibrinolysis has long been the go-to advanced therapy for high-risk pulmonary embolism (PE), but physicians are leery of the associated 2%-3% risk of catastrophic intracranial hemorrhage, noted Dr. Piazza of Brigham and Women’s Hospital and Harvard University, Boston.
SEATTLE II was a single-arm, 21-site, prospective study in which 150 patients with high-risk PE underwent treatment using the commercially available EKOS EkoSonic Endovascular System.
Twenty-one percent of patients had massive PE, defined as presentation with syncope, cardiogenic shock, resuscitated cardiac arrest, or persistent hypotension. The remaining 79% had submassive PE, with normal blood pressure but evidence of right ventricular dysfunction. All participants had to have a right ventricular/left ventricular ratio (RV/LV) of 0.9 or greater on the same chest CT scan used in diagnosing the PE. This CT documentation of RV dysfunction has been associated in a meta-analysis of patients with submassive PE with a 7.4-fold increased risk of death from PE, compared with normotensive PE patients with normal RV function (J. Thromb. Haemost. 2013;11:1823-32).
The primary endpoint was change in RV/LV on chest CT from baseline to 48 hours after initiation of fibrinolysis. This ratio improved from 1.55 to 1.13, for a statistically and clinically significant 27% reduction. A similar-size improvement was seen in pulmonary artery systolic pressure – a secondary efficacy endpoint – which decreased from 51.4 mm Hg before treatment to 37.5 mm Hg post procedure and 36.9 mm Hg at 48 hours. Both efficacy endpoints improved to a similar extent regardless of whether patients had massive or submassive PE.
The mean Modified Miller Pulmonary Artery Angiographic Obstruction Score improved by 30%, from 22.5 pretreatment to 15.8 at 48 hours.
Three in-hospital deaths occurred. One was due to massive PE that occurred before the fibrinolysis procedure could be completed. The others were not directly attributable to PE: One involved overwhelming sepsis and the other was due to progressive respiratory failure. Major bleeding occurred in 11% of patients; however, 16 of the 17 events were classified as GUSTO moderate bleeds, with only a single GUSTO severe hemorrhage.
There were no intracranial hemorrhages.
The fibrinolytic agent used in SEATTLE II was tissue plasminogen activator, delivered at 1 mg/hr for a total dose of 24 mg. Patients with unilateral PE received a single device and 24 hours of infusion time. The 86% of patients who had bilateral disease got two devices and 12 hours of therapy.
The proprietary EKOS system consists of two catheters: an outer infusion catheter with side holes that elute the fibrinolytic agent, and an inner-core catheter with ultrasound transducers placed at regular intervals. These transducers produce low-intensity ultrasound that serves two purposes. Through a process called acoustic streaming, the low-intensity ultrasound helps push the fibrinolytic agent closer to the thrombus. Plus, the ultrasound energy causes the clot fibrin to reconfigure from a tight lattice to a more porous structure that promotes deeper penetration of the fibrinolytic, Dr. Piazza explained.
Dr. Piazza said the next step in defining the EKOS system’s role in clinical practice will be to study briefer infusion times as a means of achieving faster patient improvement with reduced use of hospital resources.
The EKOS system has been approved in the United States since 2005 for treatment of blood clots in the arms and legs. In Europe it gained an additional indication for treatment of massive and submassive PE in 2011.
The SEATTLE II study was sponsored by EKOS Corp. Dr. Piazza reported receiving a research grant from the company.
Screen for Barrett’s in All With Central Obesity?
CHICAGO – The prevalence of erosive esophagitis and Barrett’s esophagus is comparable in individuals regardless of whether they have gastroesophageal reflux symptoms, according to a population-based study.
"These results directly challenge the established GERD-based Barrett’s esophagus screening paradigm and provide strong rationale for using central obesity in Caucasian males with or without symptomatic GERD as criteria for Barrett’s esophagus screening," Dr. Nicholas R. Crews said at the annual Digestive Disease Week.
"In this study, waist-hip ratio was our surrogate marker for central obesity. It’s easily obtainable and usable in clinical practice," noted Dr. Crews of the Mayo Clinic in Rochester, Minn.
Barrett’s esophagus is the precursor lesion and principal risk factor for esophageal adenocarcinoma, a malignancy whose incidence in the United States and other developed nations is increasing at an alarming rate. Improved methods of screening for esophageal adenocarcinoma are sorely needed, he added.
Dr. Crews presented a study in which a representative sample of Olmsted County, Minn., residents over age 50 with no history of endoscopy were randomized to screening for Barrett’s esophagus by one of three methods: sedated endoscopy in the GI suite, unsedated transnasal endoscopy in the clinic, or unsedated transnasal endoscopy in a Mayo mobile research van.
Participants’ mean age was 70 years, 46% were men, 206 of the 209 were white, and only one-third of subjects had GERD symptoms.
The prevalence of esophagitis grades A-C proved to be 32% in the symptomatic GERD group and similar at 29% in those without GERD symptoms. Similarly, Barrett’s esophagus was identified in 8.7% of the symptomatic GERD group and 7.9% of subjects without GERD symptoms. Dysplasia was present in 1.4% of each group. The mean length of the esophageal segment with Barrett’s esophagus was 2.4 cm in patients with GERD symptoms and not significantly different in those who were asymptomatic.
Three risk factors proved significant as predictors of esophageal injury as defined by esophagitis or Barrett’s esophagus: male sex, central obesity as defined by a waist-hip ratio greater than 0.9, and consumption of more than two alcoholic drinks per day. Age, smoking status, and body mass index were not predictive.
The mean waist-to-hip ratio was 0.89 in screened subjects with no esophagitis or Barrett’s esophagus, 0.91 in those with positive endoscopic findings and symptomatic gastroesophageal reflux, and 0.95 in those with positive findings who were asymptomatic.
Audience members expressed skepticism about the notion of routinely screening for Barrett’s esophagus in individuals with central obesity in an era of an unprecedented obesity epidemic.
For example, Dr. Joel E. Richter, who described himself as "an anti-Barrett’s person," commented that he believes gastroenterologists are already overdiagnosing and overtreating the condition, needlessly alarming many patients.
In women, particularly, it’s increasingly clear that Barrett’s esophagus only rarely develops into esophageal adenocarcinoma, he said.
"Others have said that women with Barrett’s esophagus are as likely to get esophageal cancer as men are to get breast cancer," commented Dr. Richter, professor of internal medicine and director of the center for swallowing disorders at the University of South Florida, Tampa.
Another audience member told Dr. Crews, "I totally agree with you that we miss most people with Barrett’s by our current screening process. The problem is, it’s unclear whether it’s important or not to find them. To extrapolate from your study and say that anyone with central obesity ought to be screened for [Barrett’s esophagus] is a little strong, I think."
"It’s very controversial," Dr. Crews agreed. "It’s something we continue to struggle with."
He reported having no relevant financial conflicts.
CHICAGO – The prevalence of erosive esophagitis and Barrett’s esophagus is comparable in individuals regardless of whether they have gastroesophageal reflux symptoms, according to a population-based study.
"These results directly challenge the established GERD-based Barrett’s esophagus screening paradigm and provide strong rationale for using central obesity in Caucasian males with or without symptomatic GERD as criteria for Barrett’s esophagus screening," Dr. Nicholas R. Crews said at the annual Digestive Disease Week.
"In this study, waist-hip ratio was our surrogate marker for central obesity. It’s easily obtainable and usable in clinical practice," noted Dr. Crews of the Mayo Clinic in Rochester, Minn.
Barrett’s esophagus is the precursor lesion and principal risk factor for esophageal adenocarcinoma, a malignancy whose incidence in the United States and other developed nations is increasing at an alarming rate. Improved methods of screening for esophageal adenocarcinoma are sorely needed, he added.
Dr. Crews presented a study in which a representative sample of Olmsted County, Minn., residents over age 50 with no history of endoscopy were randomized to screening for Barrett’s esophagus by one of three methods: sedated endoscopy in the GI suite, unsedated transnasal endoscopy in the clinic, or unsedated transnasal endoscopy in a Mayo mobile research van.
Participants’ mean age was 70 years, 46% were men, 206 of the 209 were white, and only one-third of subjects had GERD symptoms.
The prevalence of esophagitis grades A-C proved to be 32% in the symptomatic GERD group and similar at 29% in those without GERD symptoms. Similarly, Barrett’s esophagus was identified in 8.7% of the symptomatic GERD group and 7.9% of subjects without GERD symptoms. Dysplasia was present in 1.4% of each group. The mean length of the esophageal segment with Barrett’s esophagus was 2.4 cm in patients with GERD symptoms and not significantly different in those who were asymptomatic.
Three risk factors proved significant as predictors of esophageal injury as defined by esophagitis or Barrett’s esophagus: male sex, central obesity as defined by a waist-hip ratio greater than 0.9, and consumption of more than two alcoholic drinks per day. Age, smoking status, and body mass index were not predictive.
The mean waist-to-hip ratio was 0.89 in screened subjects with no esophagitis or Barrett’s esophagus, 0.91 in those with positive endoscopic findings and symptomatic gastroesophageal reflux, and 0.95 in those with positive findings who were asymptomatic.
Audience members expressed skepticism about the notion of routinely screening for Barrett’s esophagus in individuals with central obesity in an era of an unprecedented obesity epidemic.
For example, Dr. Joel E. Richter, who described himself as "an anti-Barrett’s person," commented that he believes gastroenterologists are already overdiagnosing and overtreating the condition, needlessly alarming many patients.
In women, particularly, it’s increasingly clear that Barrett’s esophagus only rarely develops into esophageal adenocarcinoma, he said.
"Others have said that women with Barrett’s esophagus are as likely to get esophageal cancer as men are to get breast cancer," commented Dr. Richter, professor of internal medicine and director of the center for swallowing disorders at the University of South Florida, Tampa.
Another audience member told Dr. Crews, "I totally agree with you that we miss most people with Barrett’s by our current screening process. The problem is, it’s unclear whether it’s important or not to find them. To extrapolate from your study and say that anyone with central obesity ought to be screened for [Barrett’s esophagus] is a little strong, I think."
"It’s very controversial," Dr. Crews agreed. "It’s something we continue to struggle with."
He reported having no relevant financial conflicts.
CHICAGO – The prevalence of erosive esophagitis and Barrett’s esophagus is comparable in individuals regardless of whether they have gastroesophageal reflux symptoms, according to a population-based study.
"These results directly challenge the established GERD-based Barrett’s esophagus screening paradigm and provide strong rationale for using central obesity in Caucasian males with or without symptomatic GERD as criteria for Barrett’s esophagus screening," Dr. Nicholas R. Crews said at the annual Digestive Disease Week.
"In this study, waist-hip ratio was our surrogate marker for central obesity. It’s easily obtainable and usable in clinical practice," noted Dr. Crews of the Mayo Clinic in Rochester, Minn.
Barrett’s esophagus is the precursor lesion and principal risk factor for esophageal adenocarcinoma, a malignancy whose incidence in the United States and other developed nations is increasing at an alarming rate. Improved methods of screening for esophageal adenocarcinoma are sorely needed, he added.
Dr. Crews presented a study in which a representative sample of Olmsted County, Minn., residents over age 50 with no history of endoscopy were randomized to screening for Barrett’s esophagus by one of three methods: sedated endoscopy in the GI suite, unsedated transnasal endoscopy in the clinic, or unsedated transnasal endoscopy in a Mayo mobile research van.
Participants’ mean age was 70 years, 46% were men, 206 of the 209 were white, and only one-third of subjects had GERD symptoms.
The prevalence of esophagitis grades A-C proved to be 32% in the symptomatic GERD group and similar at 29% in those without GERD symptoms. Similarly, Barrett’s esophagus was identified in 8.7% of the symptomatic GERD group and 7.9% of subjects without GERD symptoms. Dysplasia was present in 1.4% of each group. The mean length of the esophageal segment with Barrett’s esophagus was 2.4 cm in patients with GERD symptoms and not significantly different in those who were asymptomatic.
Three risk factors proved significant as predictors of esophageal injury as defined by esophagitis or Barrett’s esophagus: male sex, central obesity as defined by a waist-hip ratio greater than 0.9, and consumption of more than two alcoholic drinks per day. Age, smoking status, and body mass index were not predictive.
The mean waist-to-hip ratio was 0.89 in screened subjects with no esophagitis or Barrett’s esophagus, 0.91 in those with positive endoscopic findings and symptomatic gastroesophageal reflux, and 0.95 in those with positive findings who were asymptomatic.
Audience members expressed skepticism about the notion of routinely screening for Barrett’s esophagus in individuals with central obesity in an era of an unprecedented obesity epidemic.
For example, Dr. Joel E. Richter, who described himself as "an anti-Barrett’s person," commented that he believes gastroenterologists are already overdiagnosing and overtreating the condition, needlessly alarming many patients.
In women, particularly, it’s increasingly clear that Barrett’s esophagus only rarely develops into esophageal adenocarcinoma, he said.
"Others have said that women with Barrett’s esophagus are as likely to get esophageal cancer as men are to get breast cancer," commented Dr. Richter, professor of internal medicine and director of the center for swallowing disorders at the University of South Florida, Tampa.
Another audience member told Dr. Crews, "I totally agree with you that we miss most people with Barrett’s by our current screening process. The problem is, it’s unclear whether it’s important or not to find them. To extrapolate from your study and say that anyone with central obesity ought to be screened for [Barrett’s esophagus] is a little strong, I think."
"It’s very controversial," Dr. Crews agreed. "It’s something we continue to struggle with."
He reported having no relevant financial conflicts.
AT DDW 2014
Screen for Barrett’s in all with central obesity?
CHICAGO – The prevalence of erosive esophagitis and Barrett’s esophagus is comparable in individuals regardless of whether they have gastroesophageal reflux symptoms, according to a population-based study.
"These results directly challenge the established GERD-based Barrett’s esophagus screening paradigm and provide strong rationale for using central obesity in Caucasian males with or without symptomatic GERD as criteria for Barrett’s esophagus screening," Dr. Nicholas R. Crews said at the annual Digestive Disease Week.
"In this study, waist-hip ratio was our surrogate marker for central obesity. It’s easily obtainable and usable in clinical practice," noted Dr. Crews of the Mayo Clinic in Rochester, Minn.
Barrett’s esophagus is the precursor lesion and principal risk factor for esophageal adenocarcinoma, a malignancy whose incidence in the United States and other developed nations is increasing at an alarming rate. Improved methods of screening for esophageal adenocarcinoma are sorely needed, he added.
Dr. Crews presented a study in which a representative sample of Olmsted County, Minn., residents over age 50 with no history of endoscopy were randomized to screening for Barrett’s esophagus by one of three methods: sedated endoscopy in the GI suite, unsedated transnasal endoscopy in the clinic, or unsedated transnasal endoscopy in a Mayo mobile research van.
Participants’ mean age was 70 years, 46% were men, 206 of the 209 were white, and only one-third of subjects had GERD symptoms.
The prevalence of esophagitis grades A-C proved to be 32% in the symptomatic GERD group and similar at 29% in those without GERD symptoms. Similarly, Barrett’s esophagus was identified in 8.7% of the symptomatic GERD group and 7.9% of subjects without GERD symptoms. Dysplasia was present in 1.4% of each group. The mean length of the esophageal segment with Barrett’s esophagus was 2.4 cm in patients with GERD symptoms and not significantly different in those who were asymptomatic.
Three risk factors proved significant as predictors of esophageal injury as defined by esophagitis or Barrett’s esophagus: male sex, central obesity as defined by a waist-hip ratio greater than 0.9, and consumption of more than two alcoholic drinks per day. Age, smoking status, and body mass index were not predictive.
The mean waist-to-hip ratio was 0.89 in screened subjects with no esophagitis or Barrett’s esophagus, 0.91 in those with positive endoscopic findings and symptomatic gastroesophageal reflux, and 0.95 in those with positive findings who were asymptomatic.
Audience members expressed skepticism about the notion of routinely screening for Barrett’s esophagus in individuals with central obesity in an era of an unprecedented obesity epidemic.
For example, Dr. Joel E. Richter, who described himself as "an anti-Barrett’s person," commented that he believes gastroenterologists are already overdiagnosing and overtreating the condition, needlessly alarming many patients.
In women, particularly, it’s increasingly clear that Barrett’s esophagus only rarely develops into esophageal adenocarcinoma, he said.
"Others have said that women with Barrett’s esophagus are as likely to get esophageal cancer as men are to get breast cancer," commented Dr. Richter, professor of internal medicine and director of the center for swallowing disorders at the University of South Florida, Tampa.
Another audience member told Dr. Crews, "I totally agree with you that we miss most people with Barrett’s by our current screening process. The problem is, it’s unclear whether it’s important or not to find them. To extrapolate from your study and say that anyone with central obesity ought to be screened for [Barrett’s esophagus] is a little strong, I think."
"It’s very controversial," Dr. Crews agreed. "It’s something we continue to struggle with."
He reported having no relevant financial conflicts.
CHICAGO – The prevalence of erosive esophagitis and Barrett’s esophagus is comparable in individuals regardless of whether they have gastroesophageal reflux symptoms, according to a population-based study.
"These results directly challenge the established GERD-based Barrett’s esophagus screening paradigm and provide strong rationale for using central obesity in Caucasian males with or without symptomatic GERD as criteria for Barrett’s esophagus screening," Dr. Nicholas R. Crews said at the annual Digestive Disease Week.
"In this study, waist-hip ratio was our surrogate marker for central obesity. It’s easily obtainable and usable in clinical practice," noted Dr. Crews of the Mayo Clinic in Rochester, Minn.
Barrett’s esophagus is the precursor lesion and principal risk factor for esophageal adenocarcinoma, a malignancy whose incidence in the United States and other developed nations is increasing at an alarming rate. Improved methods of screening for esophageal adenocarcinoma are sorely needed, he added.
Dr. Crews presented a study in which a representative sample of Olmsted County, Minn., residents over age 50 with no history of endoscopy were randomized to screening for Barrett’s esophagus by one of three methods: sedated endoscopy in the GI suite, unsedated transnasal endoscopy in the clinic, or unsedated transnasal endoscopy in a Mayo mobile research van.
Participants’ mean age was 70 years, 46% were men, 206 of the 209 were white, and only one-third of subjects had GERD symptoms.
The prevalence of esophagitis grades A-C proved to be 32% in the symptomatic GERD group and similar at 29% in those without GERD symptoms. Similarly, Barrett’s esophagus was identified in 8.7% of the symptomatic GERD group and 7.9% of subjects without GERD symptoms. Dysplasia was present in 1.4% of each group. The mean length of the esophageal segment with Barrett’s esophagus was 2.4 cm in patients with GERD symptoms and not significantly different in those who were asymptomatic.
Three risk factors proved significant as predictors of esophageal injury as defined by esophagitis or Barrett’s esophagus: male sex, central obesity as defined by a waist-hip ratio greater than 0.9, and consumption of more than two alcoholic drinks per day. Age, smoking status, and body mass index were not predictive.
The mean waist-to-hip ratio was 0.89 in screened subjects with no esophagitis or Barrett’s esophagus, 0.91 in those with positive endoscopic findings and symptomatic gastroesophageal reflux, and 0.95 in those with positive findings who were asymptomatic.
Audience members expressed skepticism about the notion of routinely screening for Barrett’s esophagus in individuals with central obesity in an era of an unprecedented obesity epidemic.
For example, Dr. Joel E. Richter, who described himself as "an anti-Barrett’s person," commented that he believes gastroenterologists are already overdiagnosing and overtreating the condition, needlessly alarming many patients.
In women, particularly, it’s increasingly clear that Barrett’s esophagus only rarely develops into esophageal adenocarcinoma, he said.
"Others have said that women with Barrett’s esophagus are as likely to get esophageal cancer as men are to get breast cancer," commented Dr. Richter, professor of internal medicine and director of the center for swallowing disorders at the University of South Florida, Tampa.
Another audience member told Dr. Crews, "I totally agree with you that we miss most people with Barrett’s by our current screening process. The problem is, it’s unclear whether it’s important or not to find them. To extrapolate from your study and say that anyone with central obesity ought to be screened for [Barrett’s esophagus] is a little strong, I think."
"It’s very controversial," Dr. Crews agreed. "It’s something we continue to struggle with."
He reported having no relevant financial conflicts.
CHICAGO – The prevalence of erosive esophagitis and Barrett’s esophagus is comparable in individuals regardless of whether they have gastroesophageal reflux symptoms, according to a population-based study.
"These results directly challenge the established GERD-based Barrett’s esophagus screening paradigm and provide strong rationale for using central obesity in Caucasian males with or without symptomatic GERD as criteria for Barrett’s esophagus screening," Dr. Nicholas R. Crews said at the annual Digestive Disease Week.
"In this study, waist-hip ratio was our surrogate marker for central obesity. It’s easily obtainable and usable in clinical practice," noted Dr. Crews of the Mayo Clinic in Rochester, Minn.
Barrett’s esophagus is the precursor lesion and principal risk factor for esophageal adenocarcinoma, a malignancy whose incidence in the United States and other developed nations is increasing at an alarming rate. Improved methods of screening for esophageal adenocarcinoma are sorely needed, he added.
Dr. Crews presented a study in which a representative sample of Olmsted County, Minn., residents over age 50 with no history of endoscopy were randomized to screening for Barrett’s esophagus by one of three methods: sedated endoscopy in the GI suite, unsedated transnasal endoscopy in the clinic, or unsedated transnasal endoscopy in a Mayo mobile research van.
Participants’ mean age was 70 years, 46% were men, 206 of the 209 were white, and only one-third of subjects had GERD symptoms.
The prevalence of esophagitis grades A-C proved to be 32% in the symptomatic GERD group and similar at 29% in those without GERD symptoms. Similarly, Barrett’s esophagus was identified in 8.7% of the symptomatic GERD group and 7.9% of subjects without GERD symptoms. Dysplasia was present in 1.4% of each group. The mean length of the esophageal segment with Barrett’s esophagus was 2.4 cm in patients with GERD symptoms and not significantly different in those who were asymptomatic.
Three risk factors proved significant as predictors of esophageal injury as defined by esophagitis or Barrett’s esophagus: male sex, central obesity as defined by a waist-hip ratio greater than 0.9, and consumption of more than two alcoholic drinks per day. Age, smoking status, and body mass index were not predictive.
The mean waist-to-hip ratio was 0.89 in screened subjects with no esophagitis or Barrett’s esophagus, 0.91 in those with positive endoscopic findings and symptomatic gastroesophageal reflux, and 0.95 in those with positive findings who were asymptomatic.
Audience members expressed skepticism about the notion of routinely screening for Barrett’s esophagus in individuals with central obesity in an era of an unprecedented obesity epidemic.
For example, Dr. Joel E. Richter, who described himself as "an anti-Barrett’s person," commented that he believes gastroenterologists are already overdiagnosing and overtreating the condition, needlessly alarming many patients.
In women, particularly, it’s increasingly clear that Barrett’s esophagus only rarely develops into esophageal adenocarcinoma, he said.
"Others have said that women with Barrett’s esophagus are as likely to get esophageal cancer as men are to get breast cancer," commented Dr. Richter, professor of internal medicine and director of the center for swallowing disorders at the University of South Florida, Tampa.
Another audience member told Dr. Crews, "I totally agree with you that we miss most people with Barrett’s by our current screening process. The problem is, it’s unclear whether it’s important or not to find them. To extrapolate from your study and say that anyone with central obesity ought to be screened for [Barrett’s esophagus] is a little strong, I think."
"It’s very controversial," Dr. Crews agreed. "It’s something we continue to struggle with."
He reported having no relevant financial conflicts.
AT DDW 2014
Key clinical point: The current recommended strategy of screening for Barrett’s esophagus on the basis of symptoms of gastroesophageal reflux is called into question by a new study showing the esophageal cancer precursor lesion is just as common in screened asymptomatic individuals.
Major finding: The mean waist-to-hip ratio was 0.89 in screened subjects with no esophagitis or Barrett’s esophagus, 0.91 in those with positive endoscopic findings and symptomatic gastroesophageal reflux, and 0.95 in those with positive findings who were asymptomatic.
Data source: This was a prospective population-based study in which 209 individuals over age 50 with no history of endoscopy, two-thirds of whom had no gastroesophageal reflux symptoms, underwent screening endoscopy.
Disclosures: The presenter reported having no relevant financial conflicts.
Increased intestinal permeability is central to alcoholic hepatitis
CHICAGO – Why is it that only about 20% of individuals who consume 10 or more alcoholic drinks daily for years on end will develop alcoholic hepatitis?
New insight into the mechanism of this disease points to markedly increased intestinal permeability as playing a key role. Increased intestinal permeability is consistently present in patients with alcoholic hepatitis, but not in heavy drinkers without the disease or in normal healthy controls, Dr. George Holman reported at the annual Digestive Disease Week.
"Our study suggests that intestinal permeability is disrupted in alcoholic hepatitis. We speculate that increased intestinal permeability allows passage of lipopolysaccharides from gut bacteria into the serum. These endotoxins are carried to the liver and cause subsequent hepatic inflammation," according to Dr. Holman of the University of New Mexico, Albuquerque.
Alcoholic hepatitis is characterized by steatohepatitis and rapid clinical decompensation. It is typically seen only in patients who have consumed more than 100 g of alcohol – that’s roughly 10 drinks – daily for years.
Dr. Holman presented a prospective case-control study involving 22 patients hospitalized for severe alcoholic hepatitis and 33 healthy volunteers. The study hypothesis was that patients with alcoholic hepatitis have defective intestinal barrier function which allows gut-derived bacterial endotoxins, known as lipopolysaccharides, to enter the systemic circulation, leading to a resultant inflammatory response in the liver.
To test this hypothesis, the investigators utilized urinary excretion of lactulose and mannitol as intestinal permeability markers. They also measured serum lipopolysaccharide levels as well as circulating levels of the proinflammatory cytokines interleukin-6 and tumor necrosis factor–alpha.
The alcoholic hepatitis group showed on average an eightfold increase in intestinal permeability as measured by the lactulose/mannitol excretion ratio, compared with controls. Also, a near-perfect linear correlation was found between the degree of intestinal permeability and the magnitude of the elevation in serum lipopolysaccharide levels. Paralleling these increases in intestinal permeability and lipopolysaccharides, serum levels of interleukin-6 and tumor necrosis factor–alpha were also significantly higher in the alcoholic hepatitis patients than in controls. These data, taken as a whole, point to increased intestinal permeability as a pathogenic factor resulting in endotoxemia and immune activation, he said.
Also worthy of note, intestinal permeability tracked with MELD (model for end-stage liver disease) scores such that as intestinal permeability increased, MELD scores climbed nearly exponentially, Dr. Holman continued.
Audience members wondered whether treating alcoholic hepatitis patients, typically with prednisone or pentoxifylline, had a favorable impact upon their abnormal intestinal permeability. Dr. Holman replied that although he and his coinvestigators had planned to look at this issue, follow-up simply wasn’t possible. This was a very sick patient cohort – their mean alcohol consumption was 26 drinks daily for years – and despite treatment, nearly half of them were dead within several months. However, other investigators have previously shown that alcoholic hepatitis patients who respond favorably to treatment and are able to leave the hospital show a decrease in their previously high endotoxin levels, while more severely affected patients do not.
The cause of the defective intestinal barrier that figures prominently in alcoholic hepatitis remains a mystery. Other studies have shown that individuals who drink heavily without developing alcoholic liver injury don’t have increased intestinal permeability. Thus, increased intestinal permeability is not caused by heavy alcohol use. Some other as yet unidentified factor must be causing it, Dr. Holman concluded.
This study was supported by university research funds. Dr. Holman reported having no financial conflicts.
CHICAGO – Why is it that only about 20% of individuals who consume 10 or more alcoholic drinks daily for years on end will develop alcoholic hepatitis?
New insight into the mechanism of this disease points to markedly increased intestinal permeability as playing a key role. Increased intestinal permeability is consistently present in patients with alcoholic hepatitis, but not in heavy drinkers without the disease or in normal healthy controls, Dr. George Holman reported at the annual Digestive Disease Week.
"Our study suggests that intestinal permeability is disrupted in alcoholic hepatitis. We speculate that increased intestinal permeability allows passage of lipopolysaccharides from gut bacteria into the serum. These endotoxins are carried to the liver and cause subsequent hepatic inflammation," according to Dr. Holman of the University of New Mexico, Albuquerque.
Alcoholic hepatitis is characterized by steatohepatitis and rapid clinical decompensation. It is typically seen only in patients who have consumed more than 100 g of alcohol – that’s roughly 10 drinks – daily for years.
Dr. Holman presented a prospective case-control study involving 22 patients hospitalized for severe alcoholic hepatitis and 33 healthy volunteers. The study hypothesis was that patients with alcoholic hepatitis have defective intestinal barrier function which allows gut-derived bacterial endotoxins, known as lipopolysaccharides, to enter the systemic circulation, leading to a resultant inflammatory response in the liver.
To test this hypothesis, the investigators utilized urinary excretion of lactulose and mannitol as intestinal permeability markers. They also measured serum lipopolysaccharide levels as well as circulating levels of the proinflammatory cytokines interleukin-6 and tumor necrosis factor–alpha.
The alcoholic hepatitis group showed on average an eightfold increase in intestinal permeability as measured by the lactulose/mannitol excretion ratio, compared with controls. Also, a near-perfect linear correlation was found between the degree of intestinal permeability and the magnitude of the elevation in serum lipopolysaccharide levels. Paralleling these increases in intestinal permeability and lipopolysaccharides, serum levels of interleukin-6 and tumor necrosis factor–alpha were also significantly higher in the alcoholic hepatitis patients than in controls. These data, taken as a whole, point to increased intestinal permeability as a pathogenic factor resulting in endotoxemia and immune activation, he said.
Also worthy of note, intestinal permeability tracked with MELD (model for end-stage liver disease) scores such that as intestinal permeability increased, MELD scores climbed nearly exponentially, Dr. Holman continued.
Audience members wondered whether treating alcoholic hepatitis patients, typically with prednisone or pentoxifylline, had a favorable impact upon their abnormal intestinal permeability. Dr. Holman replied that although he and his coinvestigators had planned to look at this issue, follow-up simply wasn’t possible. This was a very sick patient cohort – their mean alcohol consumption was 26 drinks daily for years – and despite treatment, nearly half of them were dead within several months. However, other investigators have previously shown that alcoholic hepatitis patients who respond favorably to treatment and are able to leave the hospital show a decrease in their previously high endotoxin levels, while more severely affected patients do not.
The cause of the defective intestinal barrier that figures prominently in alcoholic hepatitis remains a mystery. Other studies have shown that individuals who drink heavily without developing alcoholic liver injury don’t have increased intestinal permeability. Thus, increased intestinal permeability is not caused by heavy alcohol use. Some other as yet unidentified factor must be causing it, Dr. Holman concluded.
This study was supported by university research funds. Dr. Holman reported having no financial conflicts.
CHICAGO – Why is it that only about 20% of individuals who consume 10 or more alcoholic drinks daily for years on end will develop alcoholic hepatitis?
New insight into the mechanism of this disease points to markedly increased intestinal permeability as playing a key role. Increased intestinal permeability is consistently present in patients with alcoholic hepatitis, but not in heavy drinkers without the disease or in normal healthy controls, Dr. George Holman reported at the annual Digestive Disease Week.
"Our study suggests that intestinal permeability is disrupted in alcoholic hepatitis. We speculate that increased intestinal permeability allows passage of lipopolysaccharides from gut bacteria into the serum. These endotoxins are carried to the liver and cause subsequent hepatic inflammation," according to Dr. Holman of the University of New Mexico, Albuquerque.
Alcoholic hepatitis is characterized by steatohepatitis and rapid clinical decompensation. It is typically seen only in patients who have consumed more than 100 g of alcohol – that’s roughly 10 drinks – daily for years.
Dr. Holman presented a prospective case-control study involving 22 patients hospitalized for severe alcoholic hepatitis and 33 healthy volunteers. The study hypothesis was that patients with alcoholic hepatitis have defective intestinal barrier function which allows gut-derived bacterial endotoxins, known as lipopolysaccharides, to enter the systemic circulation, leading to a resultant inflammatory response in the liver.
To test this hypothesis, the investigators utilized urinary excretion of lactulose and mannitol as intestinal permeability markers. They also measured serum lipopolysaccharide levels as well as circulating levels of the proinflammatory cytokines interleukin-6 and tumor necrosis factor–alpha.
The alcoholic hepatitis group showed on average an eightfold increase in intestinal permeability as measured by the lactulose/mannitol excretion ratio, compared with controls. Also, a near-perfect linear correlation was found between the degree of intestinal permeability and the magnitude of the elevation in serum lipopolysaccharide levels. Paralleling these increases in intestinal permeability and lipopolysaccharides, serum levels of interleukin-6 and tumor necrosis factor–alpha were also significantly higher in the alcoholic hepatitis patients than in controls. These data, taken as a whole, point to increased intestinal permeability as a pathogenic factor resulting in endotoxemia and immune activation, he said.
Also worthy of note, intestinal permeability tracked with MELD (model for end-stage liver disease) scores such that as intestinal permeability increased, MELD scores climbed nearly exponentially, Dr. Holman continued.
Audience members wondered whether treating alcoholic hepatitis patients, typically with prednisone or pentoxifylline, had a favorable impact upon their abnormal intestinal permeability. Dr. Holman replied that although he and his coinvestigators had planned to look at this issue, follow-up simply wasn’t possible. This was a very sick patient cohort – their mean alcohol consumption was 26 drinks daily for years – and despite treatment, nearly half of them were dead within several months. However, other investigators have previously shown that alcoholic hepatitis patients who respond favorably to treatment and are able to leave the hospital show a decrease in their previously high endotoxin levels, while more severely affected patients do not.
The cause of the defective intestinal barrier that figures prominently in alcoholic hepatitis remains a mystery. Other studies have shown that individuals who drink heavily without developing alcoholic liver injury don’t have increased intestinal permeability. Thus, increased intestinal permeability is not caused by heavy alcohol use. Some other as yet unidentified factor must be causing it, Dr. Holman concluded.
This study was supported by university research funds. Dr. Holman reported having no financial conflicts.
AT DDW 2014
Key clinical point: Endotoxins from gut bacteria may inflame the liver via increased intestinal permeability in alcoholic hepatitis.
Major finding: Alcoholic hepatitis is distinguished by a defective intestinal barrier as manifest in an eightfold increase in intestinal permeability in affected patients, compared with normal controls.
Data source: This was a prospective case-control study involving 22 patients hospitalized for alcoholic hepatitis and 33 healthy controls.
Disclosures: The study was supported by university research funds. The presenter reported having no financial conflicts of interest.
New definition of clinically meaningful CKD progression
LAS VEGAS – A landmark study has provided physicians with an improved definition of clinically significant progression of chronic kidney disease for use as a major endpoint in clinical trials as well as in daily medical practice.
Specifically, it’s now clear from the study conducted by the global CKD Prognosis Consortium that a 30% decline in estimated glomerular filtration rate (eGFR) over the course of 2 years portends a 5- to 6-fold increased risk of developing end-stage renal disease (ESRD) during the next 2-3 years, Dr. Josef Coresh reported at a meeting sponsored by the National Kidney Foundation.
The magnitude of increased risk of ESRD in patients with a 30% 2-year decline in eGFR was similar regardless of their baseline stage of CKD. That is, patients with a low baseline eGFR of less than 60 mL/min per 1.73 m2 had an adjusted 5.4-fold increased risk if they had a 30% drop in eGFR, compared with those whose eGFR remained unchanged over a 2-year period, while those with a 30% eGFR drop starting from a higher baseline of 60 mL/min per 1.73 m2 or more had a 6-fold increased risk, said Dr. Coresh, professor of epidemiology and director of the George W. Comstock Center for Public Health Research and Prevention at Johns Hopkins University, Baltimore.
He presented an individual-level meta-analysis of 1.5 million participants in 28 cohorts. Among roughly half a million patients in the 19 cohorts with an initial eGFR below 60 mL/min per 1.73 m2, there were 7,523 ESRD events during a mean 2.4 years of follow-up beginning at the close of the 2-year baseline period. An additional 1,009 ESRD events occurred during the follow-up period in participants with an initial eGFR of 60 mL/min per 1.73 m2 or more.
This study grew out of a collaboration between the National Kidney Foundation and the Food and Drug Administration. Officials at the regulatory agency now accept that the established endpoint used to document CKD progression in clinical trials – that is, a doubling of serum creatinine concentration from baseline – has held back therapeutic progress in the field. That’s because this FDA-mandated surrogate endpoint is a late event and thus requires studies with large sample sizes and long follow-up times. The agency is eager for evidence in support of a better surrogate endpoint, explained Dr. Coresh, who is also director of the cardiovascular epidemiology training program at Johns Hopkins.
The new meta-analysis confirmed that a doubling of serum creatinine concentration over the course of 2 years is indeed a valid endpoint for CKD progression. It was associated with a 32-fold increased risk of developing ESRD in patients who started with an eGFR below 60 mL/min per 1.73 m2 and a 57-fold increased risk in patients with a higher starting eGFR. However, a doubling of serum creatinine in a 2-year time frame was also a rare outcome, occurring in less than 1% of patients with a low initial eGFR and in 0.1% of those with a higher initial value. In contrast, a 30% drop in eGFR over a 2-year period was 10 times more common.
"A doubling of serum creatinine captures only 10% of the excess or population-attributable risk of ESRD during follow-up, whereas a 30% decline in eGFR captures 44% of the population with ESRD in the baseline low-eGFR group and 28% in those with a baseline eGFR of 60 mL/min per 1.73 m2 or greater," Dr. Coresh said.
An impressive level of consistency in the results was seen across the 28 studies included in the meta-analysis, which featured adjustment for confounders.
"I think this level of remarkable consistency makes it safer to think that in many settings, if you have a 30% decline in eGFR over 2 years, you will have a substantially increased risk of ESRD of approximately 5-fold," according to the physician.
He stressed that the meta-analysis demonstrated that it’s not just the change in eGFR over the course of 2 years that’s important in determining risk, but that the starting eGFR level and duration of follow-up are also key in determining absolute risk.
"For example, someone who starts at 50 mL/min per 1.73 m2 and is stable for 2 years has a subsequent risk of ESRD 10 years later of only 5%. If, on the other hand, that patient had a 30% decline in eGFR during 2 years, the 10-year risk becomes 21%. And if you start out at an eGFR of 35 mL/min per 1.73 m2, your 10-year risk of ESRD goes from 18% if your eGFR is unchanged for 2 years to 64% if your eGFR falls by 30%. So within the first 2 years, you have the ability to detect things that will happen a long time from now, with pretty good power," Dr. Coresh said.
The findings held true regardless of patient age, the presence or absence of diabetes, and albuminuria.
This meta-analysis, which was supported by the National Kidney Foundation and the National Institute of Diabetes and Digestive and Kidney Diseases, will result in six publications during the next year. The first was published online on June 3 (JAMA 2014 [doi:10.1001/jama.2014.6634]). The JAMA report goes beyond Dr. Coresh’s time-limited Las Vegas presentation in that it also includes data on all-cause mortality risk according to change in eGFR. He noted this is important because the majority of patients with CKD die of cardiovascular and other causes without ever reaching ESRD. In the meta-analysis, a 30% decline in eGFR over the course of 2 years was associated with an 80% increased risk in all-cause mortality.
Dr. Coresh reported having no relevant financial conflicts.
LAS VEGAS – A landmark study has provided physicians with an improved definition of clinically significant progression of chronic kidney disease for use as a major endpoint in clinical trials as well as in daily medical practice.
Specifically, it’s now clear from the study conducted by the global CKD Prognosis Consortium that a 30% decline in estimated glomerular filtration rate (eGFR) over the course of 2 years portends a 5- to 6-fold increased risk of developing end-stage renal disease (ESRD) during the next 2-3 years, Dr. Josef Coresh reported at a meeting sponsored by the National Kidney Foundation.
The magnitude of increased risk of ESRD in patients with a 30% 2-year decline in eGFR was similar regardless of their baseline stage of CKD. That is, patients with a low baseline eGFR of less than 60 mL/min per 1.73 m2 had an adjusted 5.4-fold increased risk if they had a 30% drop in eGFR, compared with those whose eGFR remained unchanged over a 2-year period, while those with a 30% eGFR drop starting from a higher baseline of 60 mL/min per 1.73 m2 or more had a 6-fold increased risk, said Dr. Coresh, professor of epidemiology and director of the George W. Comstock Center for Public Health Research and Prevention at Johns Hopkins University, Baltimore.
He presented an individual-level meta-analysis of 1.5 million participants in 28 cohorts. Among roughly half a million patients in the 19 cohorts with an initial eGFR below 60 mL/min per 1.73 m2, there were 7,523 ESRD events during a mean 2.4 years of follow-up beginning at the close of the 2-year baseline period. An additional 1,009 ESRD events occurred during the follow-up period in participants with an initial eGFR of 60 mL/min per 1.73 m2 or more.
This study grew out of a collaboration between the National Kidney Foundation and the Food and Drug Administration. Officials at the regulatory agency now accept that the established endpoint used to document CKD progression in clinical trials – that is, a doubling of serum creatinine concentration from baseline – has held back therapeutic progress in the field. That’s because this FDA-mandated surrogate endpoint is a late event and thus requires studies with large sample sizes and long follow-up times. The agency is eager for evidence in support of a better surrogate endpoint, explained Dr. Coresh, who is also director of the cardiovascular epidemiology training program at Johns Hopkins.
The new meta-analysis confirmed that a doubling of serum creatinine concentration over the course of 2 years is indeed a valid endpoint for CKD progression. It was associated with a 32-fold increased risk of developing ESRD in patients who started with an eGFR below 60 mL/min per 1.73 m2 and a 57-fold increased risk in patients with a higher starting eGFR. However, a doubling of serum creatinine in a 2-year time frame was also a rare outcome, occurring in less than 1% of patients with a low initial eGFR and in 0.1% of those with a higher initial value. In contrast, a 30% drop in eGFR over a 2-year period was 10 times more common.
"A doubling of serum creatinine captures only 10% of the excess or population-attributable risk of ESRD during follow-up, whereas a 30% decline in eGFR captures 44% of the population with ESRD in the baseline low-eGFR group and 28% in those with a baseline eGFR of 60 mL/min per 1.73 m2 or greater," Dr. Coresh said.
An impressive level of consistency in the results was seen across the 28 studies included in the meta-analysis, which featured adjustment for confounders.
"I think this level of remarkable consistency makes it safer to think that in many settings, if you have a 30% decline in eGFR over 2 years, you will have a substantially increased risk of ESRD of approximately 5-fold," according to the physician.
He stressed that the meta-analysis demonstrated that it’s not just the change in eGFR over the course of 2 years that’s important in determining risk, but that the starting eGFR level and duration of follow-up are also key in determining absolute risk.
"For example, someone who starts at 50 mL/min per 1.73 m2 and is stable for 2 years has a subsequent risk of ESRD 10 years later of only 5%. If, on the other hand, that patient had a 30% decline in eGFR during 2 years, the 10-year risk becomes 21%. And if you start out at an eGFR of 35 mL/min per 1.73 m2, your 10-year risk of ESRD goes from 18% if your eGFR is unchanged for 2 years to 64% if your eGFR falls by 30%. So within the first 2 years, you have the ability to detect things that will happen a long time from now, with pretty good power," Dr. Coresh said.
The findings held true regardless of patient age, the presence or absence of diabetes, and albuminuria.
This meta-analysis, which was supported by the National Kidney Foundation and the National Institute of Diabetes and Digestive and Kidney Diseases, will result in six publications during the next year. The first was published online on June 3 (JAMA 2014 [doi:10.1001/jama.2014.6634]). The JAMA report goes beyond Dr. Coresh’s time-limited Las Vegas presentation in that it also includes data on all-cause mortality risk according to change in eGFR. He noted this is important because the majority of patients with CKD die of cardiovascular and other causes without ever reaching ESRD. In the meta-analysis, a 30% decline in eGFR over the course of 2 years was associated with an 80% increased risk in all-cause mortality.
Dr. Coresh reported having no relevant financial conflicts.
LAS VEGAS – A landmark study has provided physicians with an improved definition of clinically significant progression of chronic kidney disease for use as a major endpoint in clinical trials as well as in daily medical practice.
Specifically, it’s now clear from the study conducted by the global CKD Prognosis Consortium that a 30% decline in estimated glomerular filtration rate (eGFR) over the course of 2 years portends a 5- to 6-fold increased risk of developing end-stage renal disease (ESRD) during the next 2-3 years, Dr. Josef Coresh reported at a meeting sponsored by the National Kidney Foundation.
The magnitude of increased risk of ESRD in patients with a 30% 2-year decline in eGFR was similar regardless of their baseline stage of CKD. That is, patients with a low baseline eGFR of less than 60 mL/min per 1.73 m2 had an adjusted 5.4-fold increased risk if they had a 30% drop in eGFR, compared with those whose eGFR remained unchanged over a 2-year period, while those with a 30% eGFR drop starting from a higher baseline of 60 mL/min per 1.73 m2 or more had a 6-fold increased risk, said Dr. Coresh, professor of epidemiology and director of the George W. Comstock Center for Public Health Research and Prevention at Johns Hopkins University, Baltimore.
He presented an individual-level meta-analysis of 1.5 million participants in 28 cohorts. Among roughly half a million patients in the 19 cohorts with an initial eGFR below 60 mL/min per 1.73 m2, there were 7,523 ESRD events during a mean 2.4 years of follow-up beginning at the close of the 2-year baseline period. An additional 1,009 ESRD events occurred during the follow-up period in participants with an initial eGFR of 60 mL/min per 1.73 m2 or more.
This study grew out of a collaboration between the National Kidney Foundation and the Food and Drug Administration. Officials at the regulatory agency now accept that the established endpoint used to document CKD progression in clinical trials – that is, a doubling of serum creatinine concentration from baseline – has held back therapeutic progress in the field. That’s because this FDA-mandated surrogate endpoint is a late event and thus requires studies with large sample sizes and long follow-up times. The agency is eager for evidence in support of a better surrogate endpoint, explained Dr. Coresh, who is also director of the cardiovascular epidemiology training program at Johns Hopkins.
The new meta-analysis confirmed that a doubling of serum creatinine concentration over the course of 2 years is indeed a valid endpoint for CKD progression. It was associated with a 32-fold increased risk of developing ESRD in patients who started with an eGFR below 60 mL/min per 1.73 m2 and a 57-fold increased risk in patients with a higher starting eGFR. However, a doubling of serum creatinine in a 2-year time frame was also a rare outcome, occurring in less than 1% of patients with a low initial eGFR and in 0.1% of those with a higher initial value. In contrast, a 30% drop in eGFR over a 2-year period was 10 times more common.
"A doubling of serum creatinine captures only 10% of the excess or population-attributable risk of ESRD during follow-up, whereas a 30% decline in eGFR captures 44% of the population with ESRD in the baseline low-eGFR group and 28% in those with a baseline eGFR of 60 mL/min per 1.73 m2 or greater," Dr. Coresh said.
An impressive level of consistency in the results was seen across the 28 studies included in the meta-analysis, which featured adjustment for confounders.
"I think this level of remarkable consistency makes it safer to think that in many settings, if you have a 30% decline in eGFR over 2 years, you will have a substantially increased risk of ESRD of approximately 5-fold," according to the physician.
He stressed that the meta-analysis demonstrated that it’s not just the change in eGFR over the course of 2 years that’s important in determining risk, but that the starting eGFR level and duration of follow-up are also key in determining absolute risk.
"For example, someone who starts at 50 mL/min per 1.73 m2 and is stable for 2 years has a subsequent risk of ESRD 10 years later of only 5%. If, on the other hand, that patient had a 30% decline in eGFR during 2 years, the 10-year risk becomes 21%. And if you start out at an eGFR of 35 mL/min per 1.73 m2, your 10-year risk of ESRD goes from 18% if your eGFR is unchanged for 2 years to 64% if your eGFR falls by 30%. So within the first 2 years, you have the ability to detect things that will happen a long time from now, with pretty good power," Dr. Coresh said.
The findings held true regardless of patient age, the presence or absence of diabetes, and albuminuria.
This meta-analysis, which was supported by the National Kidney Foundation and the National Institute of Diabetes and Digestive and Kidney Diseases, will result in six publications during the next year. The first was published online on June 3 (JAMA 2014 [doi:10.1001/jama.2014.6634]). The JAMA report goes beyond Dr. Coresh’s time-limited Las Vegas presentation in that it also includes data on all-cause mortality risk according to change in eGFR. He noted this is important because the majority of patients with CKD die of cardiovascular and other causes without ever reaching ESRD. In the meta-analysis, a 30% decline in eGFR over the course of 2 years was associated with an 80% increased risk in all-cause mortality.
Dr. Coresh reported having no relevant financial conflicts.
AT SCM 14
Key clinical point: A decline of 30% in eGFR over the course of 2 years helps identify those who will progress to end-stage renal disease in the next 2-3 years.
Major finding: Patients with a 30% drop in estimated glomerular filtration rate during a 2-year period had a 5- to 6-fold increased risk of developing end-stage renal disease during a subsequent mean 2.4 years of follow-up, compared with patients whose eGFR remained unchanged.
Data source: An individual-level meta-analysis of 28 studies involving roughly 1.5 million patients with chronic kidney disease.
Disclosures: The study was supported by the National Kidney Foundation and the National Institute of Diabetes and Digestive and Kidney Diseases. The presenter reported having no relevant financial conflicts.
STEMI: Prereperfusion IV metoprolol shows long-term benefits
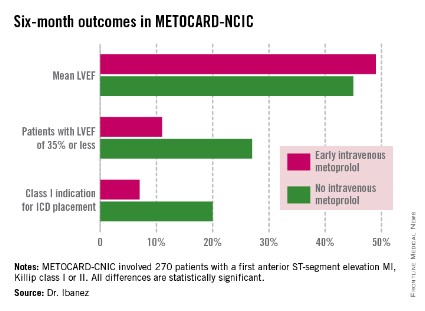
WASHINGTON – Early administration of intravenous metoprolol prior to primary percutaneous coronary intervention in patients with Killip class I or II anterior ST-elevation myocardial infarction reaped impressive long-term benefits in updated results from the Spanish METOCARD-CNIC trial.
At 6 months post infarct, the mean left ventricular ejection fraction (LVEF) was significantly higher in the early IV beta-blocker group than in controls. Also, the prevalence of a severely depressed LVEF of 35% or less was significantly lower, as was the proportion of patients having a class I indication for an implantable cardioverter-defibrillator, Dr. Borja Ibanez reported at the annual meeting of the American College of Cardiology.
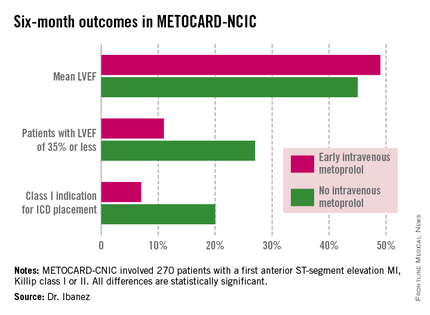
METOCARD-CNIC
The primary study endpoint was infarct size as measured by magnetic resonance imaging 7 days post STEMI. As previously reported, infarct size was 20% smaller in the IV metoprolol recipients (Circulation 2013;128:1495-1503). This finding was encouraging, Dr. Ibanez observed, because infarct size is a major determinant of long-term morbidity and mortality. And while primary PCI for STEMI results in very low acute mortality, there is a high residual risk of subsequent heart failure and death. So the search is on for treatments that reduce infarct size. And prior to METOCARD-CNIC there had been no randomized controlled trials of early IV beta-blocker therapy during the primary PCI era.
At ACC 14, Dr. Ibanez presented prespecified secondary endpoints based upon outcomes at 6 months (see chart) and 2 years post STEMI.
At a median follow-up of 2 years, the composite endpoint comprised of death, reinfarction, hospital admission for heart failure, or malignant arrhythmia had occurred in 10.8% of the IV metoprolol group, compared with 18.3% of controls. This translated to an adjusted 45% relative risk reduction which didn’t quite reach statistical significance, but then again the trial wasn’t of sufficient size to be powered to evaluate clinical endpoints.
Of note, the heart failure hospital admission component of the composite endpoint occurred in 2.2% of the IV beta-blocker group, compared with 6.9% of controls, for a 68% relative risk reduction that was statistically significant. The curves began to split at 12 months and continued to diverge through 24 months, according to Dr. Ibanez.
Based upon the encouraging findings of METOCARD-CNIC, planning is underway for a large randomized trial powered to evaluate hard clinical endpoints. It will be called the MOVE ON! trial, the cardiologist added.
METOCARD-CNIC was funded by the Carlos III National Center for Cardiovascular Investigations and the Spanish Ministry of Health and Social Policy. Dr. Ibanez reported having no financial conflicts of interest.
WASHINGTON – Early administration of intravenous metoprolol prior to primary percutaneous coronary intervention in patients with Killip class I or II anterior ST-elevation myocardial infarction reaped impressive long-term benefits in updated results from the Spanish METOCARD-CNIC trial.
At 6 months post infarct, the mean left ventricular ejection fraction (LVEF) was significantly higher in the early IV beta-blocker group than in controls. Also, the prevalence of a severely depressed LVEF of 35% or less was significantly lower, as was the proportion of patients having a class I indication for an implantable cardioverter-defibrillator, Dr. Borja Ibanez reported at the annual meeting of the American College of Cardiology.

METOCARD-CNIC
The primary study endpoint was infarct size as measured by magnetic resonance imaging 7 days post STEMI. As previously reported, infarct size was 20% smaller in the IV metoprolol recipients (Circulation 2013;128:1495-1503). This finding was encouraging, Dr. Ibanez observed, because infarct size is a major determinant of long-term morbidity and mortality. And while primary PCI for STEMI results in very low acute mortality, there is a high residual risk of subsequent heart failure and death. So the search is on for treatments that reduce infarct size. And prior to METOCARD-CNIC there had been no randomized controlled trials of early IV beta-blocker therapy during the primary PCI era.
At ACC 14, Dr. Ibanez presented prespecified secondary endpoints based upon outcomes at 6 months (see chart) and 2 years post STEMI.
At a median follow-up of 2 years, the composite endpoint comprised of death, reinfarction, hospital admission for heart failure, or malignant arrhythmia had occurred in 10.8% of the IV metoprolol group, compared with 18.3% of controls. This translated to an adjusted 45% relative risk reduction which didn’t quite reach statistical significance, but then again the trial wasn’t of sufficient size to be powered to evaluate clinical endpoints.
Of note, the heart failure hospital admission component of the composite endpoint occurred in 2.2% of the IV beta-blocker group, compared with 6.9% of controls, for a 68% relative risk reduction that was statistically significant. The curves began to split at 12 months and continued to diverge through 24 months, according to Dr. Ibanez.
Based upon the encouraging findings of METOCARD-CNIC, planning is underway for a large randomized trial powered to evaluate hard clinical endpoints. It will be called the MOVE ON! trial, the cardiologist added.
METOCARD-CNIC was funded by the Carlos III National Center for Cardiovascular Investigations and the Spanish Ministry of Health and Social Policy. Dr. Ibanez reported having no financial conflicts of interest.
WASHINGTON – Early administration of intravenous metoprolol prior to primary percutaneous coronary intervention in patients with Killip class I or II anterior ST-elevation myocardial infarction reaped impressive long-term benefits in updated results from the Spanish METOCARD-CNIC trial.
At 6 months post infarct, the mean left ventricular ejection fraction (LVEF) was significantly higher in the early IV beta-blocker group than in controls. Also, the prevalence of a severely depressed LVEF of 35% or less was significantly lower, as was the proportion of patients having a class I indication for an implantable cardioverter-defibrillator, Dr. Borja Ibanez reported at the annual meeting of the American College of Cardiology.

METOCARD-CNIC
The primary study endpoint was infarct size as measured by magnetic resonance imaging 7 days post STEMI. As previously reported, infarct size was 20% smaller in the IV metoprolol recipients (Circulation 2013;128:1495-1503). This finding was encouraging, Dr. Ibanez observed, because infarct size is a major determinant of long-term morbidity and mortality. And while primary PCI for STEMI results in very low acute mortality, there is a high residual risk of subsequent heart failure and death. So the search is on for treatments that reduce infarct size. And prior to METOCARD-CNIC there had been no randomized controlled trials of early IV beta-blocker therapy during the primary PCI era.
At ACC 14, Dr. Ibanez presented prespecified secondary endpoints based upon outcomes at 6 months (see chart) and 2 years post STEMI.
At a median follow-up of 2 years, the composite endpoint comprised of death, reinfarction, hospital admission for heart failure, or malignant arrhythmia had occurred in 10.8% of the IV metoprolol group, compared with 18.3% of controls. This translated to an adjusted 45% relative risk reduction which didn’t quite reach statistical significance, but then again the trial wasn’t of sufficient size to be powered to evaluate clinical endpoints.
Of note, the heart failure hospital admission component of the composite endpoint occurred in 2.2% of the IV beta-blocker group, compared with 6.9% of controls, for a 68% relative risk reduction that was statistically significant. The curves began to split at 12 months and continued to diverge through 24 months, according to Dr. Ibanez.
Based upon the encouraging findings of METOCARD-CNIC, planning is underway for a large randomized trial powered to evaluate hard clinical endpoints. It will be called the MOVE ON! trial, the cardiologist added.
METOCARD-CNIC was funded by the Carlos III National Center for Cardiovascular Investigations and the Spanish Ministry of Health and Social Policy. Dr. Ibanez reported having no financial conflicts of interest.
AT ACC 14
Key clinical point: Prereperfusion IV metoprolol may reduce heart failure readmissions.
Major finding: Patients with anterior ST-elevation MI who received intravenous metoprolol prior to primary PCI had a significantly greater left ventricular ejection fraction at 6 months follow-up than those who didn’t. They also were 68% less likely to be hospitalized for heart failure during 2 years of follow-up.
Data source: A six-center prospective trial in which 270 Spanish patients with a first anterior STEMI were randomized to have administration of IV metoprolol or not while being transported for primary PCI.
Disclosures: The METOCARD-CNIC trial was sponsored by the Spanish Ministry of Health and Social Policy and the Carlos III National Center for Cardiovascular Investigations. The presenter reported having no financial conflicts.
EHR stroke order-set in ED improves outcomes
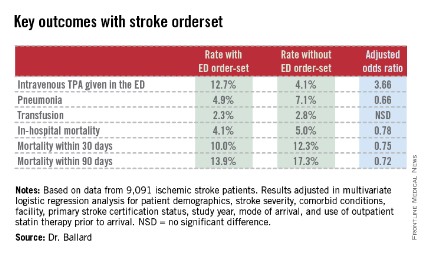
DALLAS – The use of an electronic health record order-set with built-in clinical decision support for ischemic stroke patients in the emergency department was associated with markedly improved patient outcomes in a large observational study.
Where the order-set was used, stroke patients were significantly more likely to receive tissue plasminogen activator (TPA) therapy in the ED. They were also significantly less likely to develop pneumonia, including aspiration pneumonia. And they had significantly reduced in-hospital, 30- and 90-day mortality, compared with patients in EDs where the stroke order-set wasn’t yet available, Dr. Dustin W. Ballard reported at the annual meeting of the Society for Academic Emergency Medicine.
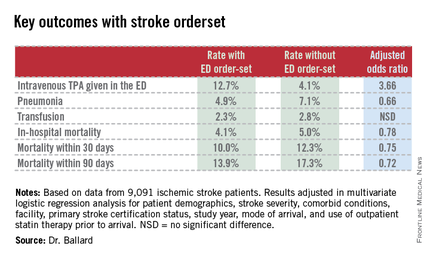
"The magnitude of the difference in outcomes was greater than we expected, for certain," commented Dr. Ballard, an emergency medicine physician at Kaiser Permanente San Rafael (Calif.) Medical Center.
This retrospective observational study took advantage of a natural experiment going on within the 21–medical center Kaiser Permanente of Northern California integrated health care delivery system. During 2007-2012, an electronic health record (EHR) with stroke order-set was introduced across the system in staggered fashion such that it was up and running in some of the 21 EDs, while in others, it wasn’t yet available. As a result, Dr. Ballard was able to report on 9,091 ischemic stroke patients who presented to Kaiser Permanente EDs during the study period, 53% of whom were managed with the assistance of the stroke order-set.
The order-set incorporates laboratory tests, neuroimaging orders, and other diagnostic options as well as inclusion and exclusion criteria for TPA therapy, TPA dosing recommendations, criteria for conducting a swallowing evaluation, and dysphagia orders.
"Where it was available for use, there was no mandate that ED physicians had to use it. It’s intended to be the first thing you open up when you see a patient who you think might have a stroke. I use the order-set in all patients I think might have stroke. It gives me back-up, regardless of whether I think I’m going to use TPA or not. But certainly if I were going to use TPA, I can scan through the inclusion/exclusion criteria really quickly, and the dosing recommendations are right there. There are certainly other ways to get that information, but the order-set just makes it much simpler and much easier. I think that’s the primary reason why we’re seeing much higher TPA utilization rates," Dr. Ballard explained.
Indeed, when the order-set was used, 12.7% of stroke patients received intravenous TPA in the ED. Where it wasn’t, the rate was 4.1%. And reassuringly, this nearly 3.7-fold greater use of TPA wasn’t accompanied by an increased rate of bleeding requiring transfusion (see chart).
Audience members who feel pressure to convince their hospital administrators that an ED provides good value to a medical center even if the ED isn’t a major profit center greeted the study results enthusiastically.
"This could be very exciting," one physician rose to comment. "Now we’ve not only got a checklist for stroke, but we’ve also got a mortality reduction in those patients it’s provided to."
Other audience members cautioned that confounding by indication is always a potential concern in a retrospective study such as this. That is, perhaps when ED physicians encountered a patient with a really severe stroke and low likelihood of successful recovery, they dispensed with the order-set. Dr. Ballard responded that patient stroke severity scores and overall comorbidity burden were similar in the groups with and without the order-set. And while secular trends in stroke diagnosis and treatment during 2007-2012 led to an overall reduction in stroke mortality at Kaiser Permanente during that period, "it was not anywhere near as dramatic as what we see with the order-set," he added.
Dr. Ballard noted that the Kaiser Permanente results add to a growing body of evidence that carefully designed order-sets for selected disorders leads to improved clinical outcomes. Most of those studies, however, involved paper order-sets rather than electronic aids.
The study was supported by the Kaiser Permanente Division of Research. Dr. Ballard is a Kaiser Permanente employee.

|
|
Physician apprehension of "cookbook" medicine should be relegated to a bygone era. The seamless use of standard work processes such as checklists or well constructed EMR order sets can support the delivery of consistent evidence-based care and let the clinician focus their time on communication and management decisions for the clinical outliers. The growing body of evidence supporting integration of standard work continues to expand–an ED based stroke order set in this study by Kaiser is just one more excellent example.
Dr. Robert Pendleton is codirector of University of Utah Hospitalist Program, director of University Healthcare Thrombosis Service, both in Salt Lake City, and an adviser to Hospitalist News.

|
|
Physician apprehension of "cookbook" medicine should be relegated to a bygone era. The seamless use of standard work processes such as checklists or well constructed EMR order sets can support the delivery of consistent evidence-based care and let the clinician focus their time on communication and management decisions for the clinical outliers. The growing body of evidence supporting integration of standard work continues to expand–an ED based stroke order set in this study by Kaiser is just one more excellent example.
Dr. Robert Pendleton is codirector of University of Utah Hospitalist Program, director of University Healthcare Thrombosis Service, both in Salt Lake City, and an adviser to Hospitalist News.

|
|
Physician apprehension of "cookbook" medicine should be relegated to a bygone era. The seamless use of standard work processes such as checklists or well constructed EMR order sets can support the delivery of consistent evidence-based care and let the clinician focus their time on communication and management decisions for the clinical outliers. The growing body of evidence supporting integration of standard work continues to expand–an ED based stroke order set in this study by Kaiser is just one more excellent example.
Dr. Robert Pendleton is codirector of University of Utah Hospitalist Program, director of University Healthcare Thrombosis Service, both in Salt Lake City, and an adviser to Hospitalist News.
DALLAS – The use of an electronic health record order-set with built-in clinical decision support for ischemic stroke patients in the emergency department was associated with markedly improved patient outcomes in a large observational study.
Where the order-set was used, stroke patients were significantly more likely to receive tissue plasminogen activator (TPA) therapy in the ED. They were also significantly less likely to develop pneumonia, including aspiration pneumonia. And they had significantly reduced in-hospital, 30- and 90-day mortality, compared with patients in EDs where the stroke order-set wasn’t yet available, Dr. Dustin W. Ballard reported at the annual meeting of the Society for Academic Emergency Medicine.

"The magnitude of the difference in outcomes was greater than we expected, for certain," commented Dr. Ballard, an emergency medicine physician at Kaiser Permanente San Rafael (Calif.) Medical Center.
This retrospective observational study took advantage of a natural experiment going on within the 21–medical center Kaiser Permanente of Northern California integrated health care delivery system. During 2007-2012, an electronic health record (EHR) with stroke order-set was introduced across the system in staggered fashion such that it was up and running in some of the 21 EDs, while in others, it wasn’t yet available. As a result, Dr. Ballard was able to report on 9,091 ischemic stroke patients who presented to Kaiser Permanente EDs during the study period, 53% of whom were managed with the assistance of the stroke order-set.
The order-set incorporates laboratory tests, neuroimaging orders, and other diagnostic options as well as inclusion and exclusion criteria for TPA therapy, TPA dosing recommendations, criteria for conducting a swallowing evaluation, and dysphagia orders.
"Where it was available for use, there was no mandate that ED physicians had to use it. It’s intended to be the first thing you open up when you see a patient who you think might have a stroke. I use the order-set in all patients I think might have stroke. It gives me back-up, regardless of whether I think I’m going to use TPA or not. But certainly if I were going to use TPA, I can scan through the inclusion/exclusion criteria really quickly, and the dosing recommendations are right there. There are certainly other ways to get that information, but the order-set just makes it much simpler and much easier. I think that’s the primary reason why we’re seeing much higher TPA utilization rates," Dr. Ballard explained.
Indeed, when the order-set was used, 12.7% of stroke patients received intravenous TPA in the ED. Where it wasn’t, the rate was 4.1%. And reassuringly, this nearly 3.7-fold greater use of TPA wasn’t accompanied by an increased rate of bleeding requiring transfusion (see chart).
Audience members who feel pressure to convince their hospital administrators that an ED provides good value to a medical center even if the ED isn’t a major profit center greeted the study results enthusiastically.
"This could be very exciting," one physician rose to comment. "Now we’ve not only got a checklist for stroke, but we’ve also got a mortality reduction in those patients it’s provided to."
Other audience members cautioned that confounding by indication is always a potential concern in a retrospective study such as this. That is, perhaps when ED physicians encountered a patient with a really severe stroke and low likelihood of successful recovery, they dispensed with the order-set. Dr. Ballard responded that patient stroke severity scores and overall comorbidity burden were similar in the groups with and without the order-set. And while secular trends in stroke diagnosis and treatment during 2007-2012 led to an overall reduction in stroke mortality at Kaiser Permanente during that period, "it was not anywhere near as dramatic as what we see with the order-set," he added.
Dr. Ballard noted that the Kaiser Permanente results add to a growing body of evidence that carefully designed order-sets for selected disorders leads to improved clinical outcomes. Most of those studies, however, involved paper order-sets rather than electronic aids.
The study was supported by the Kaiser Permanente Division of Research. Dr. Ballard is a Kaiser Permanente employee.
DALLAS – The use of an electronic health record order-set with built-in clinical decision support for ischemic stroke patients in the emergency department was associated with markedly improved patient outcomes in a large observational study.
Where the order-set was used, stroke patients were significantly more likely to receive tissue plasminogen activator (TPA) therapy in the ED. They were also significantly less likely to develop pneumonia, including aspiration pneumonia. And they had significantly reduced in-hospital, 30- and 90-day mortality, compared with patients in EDs where the stroke order-set wasn’t yet available, Dr. Dustin W. Ballard reported at the annual meeting of the Society for Academic Emergency Medicine.

"The magnitude of the difference in outcomes was greater than we expected, for certain," commented Dr. Ballard, an emergency medicine physician at Kaiser Permanente San Rafael (Calif.) Medical Center.
This retrospective observational study took advantage of a natural experiment going on within the 21–medical center Kaiser Permanente of Northern California integrated health care delivery system. During 2007-2012, an electronic health record (EHR) with stroke order-set was introduced across the system in staggered fashion such that it was up and running in some of the 21 EDs, while in others, it wasn’t yet available. As a result, Dr. Ballard was able to report on 9,091 ischemic stroke patients who presented to Kaiser Permanente EDs during the study period, 53% of whom were managed with the assistance of the stroke order-set.
The order-set incorporates laboratory tests, neuroimaging orders, and other diagnostic options as well as inclusion and exclusion criteria for TPA therapy, TPA dosing recommendations, criteria for conducting a swallowing evaluation, and dysphagia orders.
"Where it was available for use, there was no mandate that ED physicians had to use it. It’s intended to be the first thing you open up when you see a patient who you think might have a stroke. I use the order-set in all patients I think might have stroke. It gives me back-up, regardless of whether I think I’m going to use TPA or not. But certainly if I were going to use TPA, I can scan through the inclusion/exclusion criteria really quickly, and the dosing recommendations are right there. There are certainly other ways to get that information, but the order-set just makes it much simpler and much easier. I think that’s the primary reason why we’re seeing much higher TPA utilization rates," Dr. Ballard explained.
Indeed, when the order-set was used, 12.7% of stroke patients received intravenous TPA in the ED. Where it wasn’t, the rate was 4.1%. And reassuringly, this nearly 3.7-fold greater use of TPA wasn’t accompanied by an increased rate of bleeding requiring transfusion (see chart).
Audience members who feel pressure to convince their hospital administrators that an ED provides good value to a medical center even if the ED isn’t a major profit center greeted the study results enthusiastically.
"This could be very exciting," one physician rose to comment. "Now we’ve not only got a checklist for stroke, but we’ve also got a mortality reduction in those patients it’s provided to."
Other audience members cautioned that confounding by indication is always a potential concern in a retrospective study such as this. That is, perhaps when ED physicians encountered a patient with a really severe stroke and low likelihood of successful recovery, they dispensed with the order-set. Dr. Ballard responded that patient stroke severity scores and overall comorbidity burden were similar in the groups with and without the order-set. And while secular trends in stroke diagnosis and treatment during 2007-2012 led to an overall reduction in stroke mortality at Kaiser Permanente during that period, "it was not anywhere near as dramatic as what we see with the order-set," he added.
Dr. Ballard noted that the Kaiser Permanente results add to a growing body of evidence that carefully designed order-sets for selected disorders leads to improved clinical outcomes. Most of those studies, however, involved paper order-sets rather than electronic aids.
The study was supported by the Kaiser Permanente Division of Research. Dr. Ballard is a Kaiser Permanente employee.
AT SAEM 2014
Key clinical point: Embrace EHR stroke order-sets to improve clinical decision making.
Major finding: In-hospital mortality in patients who presented to the ED with ischemic stroke was 4.1% if ED physicians used an EHR stroke order-set and significantly worse at 5.0% if physicians didn’t yet have access to the electronic order-set.
Data source: This was a retrospective observational study of 9,091 patients who presented with ischemic stroke to 1 of 21 EDs in an integrated health system during 2007-2012. A total of 53% were managed with the assistance of an EHR stroke order-set for the ED.
Disclosures: The study was sponsored by Kaiser Permanente. The presenter is employed as an emergency medicine physician by the health system.
Novel Noninvasive Therapy for Extraesophageal Reflux
CHICAGO – A novel, externally worn device for the treatment of extraesophageal reflux proved effective, safe, and well tolerated in a multicenter clinical trial.
"Given the poor response to PPI [proton-pump inhibitors] therapy in many patients with extraesophageal reflux, this device may be an alternative form of treating this difficult disorder," Dr. Michael F. Vaezi said in a presentation of the study findings at the annual Digestive Disease Week.
Extraesophageal reflux (EER) affects more than 15 million Americans. The cost of care is estimated to be in excess of $50 billion annually, most of which goes for PPIs, which are overused and generally not effective in treating this common disorder, observed Dr. Vaezi, professor of medicine at Vanderbilt University, Nashville, Tenn.
EER is caused by reflux of gastroduodenal contents into the laryngopharynx, resulting in a range of symptoms including chronic cough, postnasal drip, throat clearing, hoarseness, and a sensation of a lump in the throat.
The investigational device, known as the Reza-Band, is an individually fitted upper esophageal sphincter assist device to be worn at bedtime. It is designed to apply slight pressure to the cricoid cartilage area sufficient to increase the intraluminal pressure of the upper esophageal sphincter to 20 mm Hg. In earlier studies, this modest increase in pressure eliminated pharyngeal reflux without affecting normal activities such as swallowing and belching.
Dr. Vaezi reported on 47 patients with EER treated with the upper esophageal assist device at four medical centers. Their mean age was 50 years, with an average body mass index of 26.1 kg/m2. Three-quarters of patients were on PPI therapy, which they felt to be ineffective.
The primary study endpoint was the change in Reflux Symptom Index scores from baseline through week 4 of nighttime use of the assist device. The median score improved from 27 at baseline to 14 at week 2 and 12 at week 4. That translated to a mean 54% reduction. A total of 86% of subjects were deemed to have experienced treatment success as defined by at least a 25% improvement in their symptom score; 30% of participants showed a greater than 75% improvement.
Treatment side effects were few, mild, and brief, consisting chiefly of skin irritation from the device’s Velcro strap. No one withdrew from the study.
The Reza-Band device was developed by gastroenterologists at the Medical College of Wisconsin, Milwaukee. Dr. Vaezi said he and his coinvestigators decided to formally investigate the device because "we were intrigued and impressed by the fact that it has helped so many patients."
"For me, it’s a welcome development," Dr. Vaezi added. "Just anecdotally, many patients in the study wanted to know how they could purchase this device. It’s really welcome in an area [in which] we don’t have many therapeutic options."
Still, he noted, it’s probably a good idea for device users to continue to keep a couple of bricks under the head of the bed, in accord with standard medical advice.
Future plans include a sham-controlled study designed to assess the device’s true benefit over placebo therapy; this will be an important study because EER is known to have a substantial placebo response rate, he continued. Also, plans are afoot for a safety study assessing whether the device has any clinically meaningful impact upon intraocular pressure.
The study was supported by Somna Therapeutics, which is developing the device. Dr. Vaezi reported having no financial relationship with the company.
CHICAGO – A novel, externally worn device for the treatment of extraesophageal reflux proved effective, safe, and well tolerated in a multicenter clinical trial.
"Given the poor response to PPI [proton-pump inhibitors] therapy in many patients with extraesophageal reflux, this device may be an alternative form of treating this difficult disorder," Dr. Michael F. Vaezi said in a presentation of the study findings at the annual Digestive Disease Week.
Extraesophageal reflux (EER) affects more than 15 million Americans. The cost of care is estimated to be in excess of $50 billion annually, most of which goes for PPIs, which are overused and generally not effective in treating this common disorder, observed Dr. Vaezi, professor of medicine at Vanderbilt University, Nashville, Tenn.
EER is caused by reflux of gastroduodenal contents into the laryngopharynx, resulting in a range of symptoms including chronic cough, postnasal drip, throat clearing, hoarseness, and a sensation of a lump in the throat.
The investigational device, known as the Reza-Band, is an individually fitted upper esophageal sphincter assist device to be worn at bedtime. It is designed to apply slight pressure to the cricoid cartilage area sufficient to increase the intraluminal pressure of the upper esophageal sphincter to 20 mm Hg. In earlier studies, this modest increase in pressure eliminated pharyngeal reflux without affecting normal activities such as swallowing and belching.
Dr. Vaezi reported on 47 patients with EER treated with the upper esophageal assist device at four medical centers. Their mean age was 50 years, with an average body mass index of 26.1 kg/m2. Three-quarters of patients were on PPI therapy, which they felt to be ineffective.
The primary study endpoint was the change in Reflux Symptom Index scores from baseline through week 4 of nighttime use of the assist device. The median score improved from 27 at baseline to 14 at week 2 and 12 at week 4. That translated to a mean 54% reduction. A total of 86% of subjects were deemed to have experienced treatment success as defined by at least a 25% improvement in their symptom score; 30% of participants showed a greater than 75% improvement.
Treatment side effects were few, mild, and brief, consisting chiefly of skin irritation from the device’s Velcro strap. No one withdrew from the study.
The Reza-Band device was developed by gastroenterologists at the Medical College of Wisconsin, Milwaukee. Dr. Vaezi said he and his coinvestigators decided to formally investigate the device because "we were intrigued and impressed by the fact that it has helped so many patients."
"For me, it’s a welcome development," Dr. Vaezi added. "Just anecdotally, many patients in the study wanted to know how they could purchase this device. It’s really welcome in an area [in which] we don’t have many therapeutic options."
Still, he noted, it’s probably a good idea for device users to continue to keep a couple of bricks under the head of the bed, in accord with standard medical advice.
Future plans include a sham-controlled study designed to assess the device’s true benefit over placebo therapy; this will be an important study because EER is known to have a substantial placebo response rate, he continued. Also, plans are afoot for a safety study assessing whether the device has any clinically meaningful impact upon intraocular pressure.
The study was supported by Somna Therapeutics, which is developing the device. Dr. Vaezi reported having no financial relationship with the company.
CHICAGO – A novel, externally worn device for the treatment of extraesophageal reflux proved effective, safe, and well tolerated in a multicenter clinical trial.
"Given the poor response to PPI [proton-pump inhibitors] therapy in many patients with extraesophageal reflux, this device may be an alternative form of treating this difficult disorder," Dr. Michael F. Vaezi said in a presentation of the study findings at the annual Digestive Disease Week.
Extraesophageal reflux (EER) affects more than 15 million Americans. The cost of care is estimated to be in excess of $50 billion annually, most of which goes for PPIs, which are overused and generally not effective in treating this common disorder, observed Dr. Vaezi, professor of medicine at Vanderbilt University, Nashville, Tenn.
EER is caused by reflux of gastroduodenal contents into the laryngopharynx, resulting in a range of symptoms including chronic cough, postnasal drip, throat clearing, hoarseness, and a sensation of a lump in the throat.
The investigational device, known as the Reza-Band, is an individually fitted upper esophageal sphincter assist device to be worn at bedtime. It is designed to apply slight pressure to the cricoid cartilage area sufficient to increase the intraluminal pressure of the upper esophageal sphincter to 20 mm Hg. In earlier studies, this modest increase in pressure eliminated pharyngeal reflux without affecting normal activities such as swallowing and belching.
Dr. Vaezi reported on 47 patients with EER treated with the upper esophageal assist device at four medical centers. Their mean age was 50 years, with an average body mass index of 26.1 kg/m2. Three-quarters of patients were on PPI therapy, which they felt to be ineffective.
The primary study endpoint was the change in Reflux Symptom Index scores from baseline through week 4 of nighttime use of the assist device. The median score improved from 27 at baseline to 14 at week 2 and 12 at week 4. That translated to a mean 54% reduction. A total of 86% of subjects were deemed to have experienced treatment success as defined by at least a 25% improvement in their symptom score; 30% of participants showed a greater than 75% improvement.
Treatment side effects were few, mild, and brief, consisting chiefly of skin irritation from the device’s Velcro strap. No one withdrew from the study.
The Reza-Band device was developed by gastroenterologists at the Medical College of Wisconsin, Milwaukee. Dr. Vaezi said he and his coinvestigators decided to formally investigate the device because "we were intrigued and impressed by the fact that it has helped so many patients."
"For me, it’s a welcome development," Dr. Vaezi added. "Just anecdotally, many patients in the study wanted to know how they could purchase this device. It’s really welcome in an area [in which] we don’t have many therapeutic options."
Still, he noted, it’s probably a good idea for device users to continue to keep a couple of bricks under the head of the bed, in accord with standard medical advice.
Future plans include a sham-controlled study designed to assess the device’s true benefit over placebo therapy; this will be an important study because EER is known to have a substantial placebo response rate, he continued. Also, plans are afoot for a safety study assessing whether the device has any clinically meaningful impact upon intraocular pressure.
The study was supported by Somna Therapeutics, which is developing the device. Dr. Vaezi reported having no financial relationship with the company.
AT DDW 2014
Novel noninvasive therapy for extraesophageal reflux
CHICAGO – A novel, externally worn device for the treatment of extraesophageal reflux proved effective, safe, and well tolerated in a multicenter clinical trial.
"Given the poor response to PPI [proton-pump inhibitors] therapy in many patients with extraesophageal reflux, this device may be an alternative form of treating this difficult disorder," Dr. Michael F. Vaezi said in a presentation of the study findings at the annual Digestive Disease Week.
Extraesophageal reflux (EER) affects more than 15 million Americans. The cost of care is estimated to be in excess of $50 billion annually, most of which goes for PPIs, which are overused and generally not effective in treating this common disorder, observed Dr. Vaezi, professor of medicine at Vanderbilt University, Nashville, Tenn.
EER is caused by reflux of gastroduodenal contents into the laryngopharynx, resulting in a range of symptoms including chronic cough, postnasal drip, throat clearing, hoarseness, and a sensation of a lump in the throat.
The investigational device, known as the Reza-Band, is an individually fitted upper esophageal sphincter assist device to be worn at bedtime. It is designed to apply slight pressure to the cricoid cartilage area sufficient to increase the intraluminal pressure of the upper esophageal sphincter to 20 mm Hg. In earlier studies, this modest increase in pressure eliminated pharyngeal reflux without affecting normal activities such as swallowing and belching.
Dr. Vaezi reported on 47 patients with EER treated with the upper esophageal assist device at four medical centers. Their mean age was 50 years, with an average body mass index of 26.1 kg/m2. Three-quarters of patients were on PPI therapy, which they felt to be ineffective.
The primary study endpoint was the change in Reflux Symptom Index scores from baseline through week 4 of nighttime use of the assist device. The median score improved from 27 at baseline to 14 at week 2 and 12 at week 4. That translated to a mean 54% reduction. A total of 86% of subjects were deemed to have experienced treatment success as defined by at least a 25% improvement in their symptom score; 30% of participants showed a greater than 75% improvement.
Treatment side effects were few, mild, and brief, consisting chiefly of skin irritation from the device’s Velcro strap. No one withdrew from the study.
The Reza-Band device was developed by gastroenterologists at the Medical College of Wisconsin, Milwaukee. Dr. Vaezi said he and his coinvestigators decided to formally investigate the device because "we were intrigued and impressed by the fact that it has helped so many patients."
"For me, it’s a welcome development," Dr. Vaezi added. "Just anecdotally, many patients in the study wanted to know how they could purchase this device. It’s really welcome in an area [in which] we don’t have many therapeutic options."
Still, he noted, it’s probably a good idea for device users to continue to keep a couple of bricks under the head of the bed, in accord with standard medical advice.
Future plans include a sham-controlled study designed to assess the device’s true benefit over placebo therapy; this will be an important study because EER is known to have a substantial placebo response rate, he continued. Also, plans are afoot for a safety study assessing whether the device has any clinically meaningful impact upon intraocular pressure.
The study was supported by Somna Therapeutics, which is developing the device. Dr. Vaezi reported having no financial relationship with the company.
CHICAGO – A novel, externally worn device for the treatment of extraesophageal reflux proved effective, safe, and well tolerated in a multicenter clinical trial.
"Given the poor response to PPI [proton-pump inhibitors] therapy in many patients with extraesophageal reflux, this device may be an alternative form of treating this difficult disorder," Dr. Michael F. Vaezi said in a presentation of the study findings at the annual Digestive Disease Week.
Extraesophageal reflux (EER) affects more than 15 million Americans. The cost of care is estimated to be in excess of $50 billion annually, most of which goes for PPIs, which are overused and generally not effective in treating this common disorder, observed Dr. Vaezi, professor of medicine at Vanderbilt University, Nashville, Tenn.
EER is caused by reflux of gastroduodenal contents into the laryngopharynx, resulting in a range of symptoms including chronic cough, postnasal drip, throat clearing, hoarseness, and a sensation of a lump in the throat.
The investigational device, known as the Reza-Band, is an individually fitted upper esophageal sphincter assist device to be worn at bedtime. It is designed to apply slight pressure to the cricoid cartilage area sufficient to increase the intraluminal pressure of the upper esophageal sphincter to 20 mm Hg. In earlier studies, this modest increase in pressure eliminated pharyngeal reflux without affecting normal activities such as swallowing and belching.
Dr. Vaezi reported on 47 patients with EER treated with the upper esophageal assist device at four medical centers. Their mean age was 50 years, with an average body mass index of 26.1 kg/m2. Three-quarters of patients were on PPI therapy, which they felt to be ineffective.
The primary study endpoint was the change in Reflux Symptom Index scores from baseline through week 4 of nighttime use of the assist device. The median score improved from 27 at baseline to 14 at week 2 and 12 at week 4. That translated to a mean 54% reduction. A total of 86% of subjects were deemed to have experienced treatment success as defined by at least a 25% improvement in their symptom score; 30% of participants showed a greater than 75% improvement.
Treatment side effects were few, mild, and brief, consisting chiefly of skin irritation from the device’s Velcro strap. No one withdrew from the study.
The Reza-Band device was developed by gastroenterologists at the Medical College of Wisconsin, Milwaukee. Dr. Vaezi said he and his coinvestigators decided to formally investigate the device because "we were intrigued and impressed by the fact that it has helped so many patients."
"For me, it’s a welcome development," Dr. Vaezi added. "Just anecdotally, many patients in the study wanted to know how they could purchase this device. It’s really welcome in an area [in which] we don’t have many therapeutic options."
Still, he noted, it’s probably a good idea for device users to continue to keep a couple of bricks under the head of the bed, in accord with standard medical advice.
Future plans include a sham-controlled study designed to assess the device’s true benefit over placebo therapy; this will be an important study because EER is known to have a substantial placebo response rate, he continued. Also, plans are afoot for a safety study assessing whether the device has any clinically meaningful impact upon intraocular pressure.
The study was supported by Somna Therapeutics, which is developing the device. Dr. Vaezi reported having no financial relationship with the company.
CHICAGO – A novel, externally worn device for the treatment of extraesophageal reflux proved effective, safe, and well tolerated in a multicenter clinical trial.
"Given the poor response to PPI [proton-pump inhibitors] therapy in many patients with extraesophageal reflux, this device may be an alternative form of treating this difficult disorder," Dr. Michael F. Vaezi said in a presentation of the study findings at the annual Digestive Disease Week.
Extraesophageal reflux (EER) affects more than 15 million Americans. The cost of care is estimated to be in excess of $50 billion annually, most of which goes for PPIs, which are overused and generally not effective in treating this common disorder, observed Dr. Vaezi, professor of medicine at Vanderbilt University, Nashville, Tenn.
EER is caused by reflux of gastroduodenal contents into the laryngopharynx, resulting in a range of symptoms including chronic cough, postnasal drip, throat clearing, hoarseness, and a sensation of a lump in the throat.
The investigational device, known as the Reza-Band, is an individually fitted upper esophageal sphincter assist device to be worn at bedtime. It is designed to apply slight pressure to the cricoid cartilage area sufficient to increase the intraluminal pressure of the upper esophageal sphincter to 20 mm Hg. In earlier studies, this modest increase in pressure eliminated pharyngeal reflux without affecting normal activities such as swallowing and belching.
Dr. Vaezi reported on 47 patients with EER treated with the upper esophageal assist device at four medical centers. Their mean age was 50 years, with an average body mass index of 26.1 kg/m2. Three-quarters of patients were on PPI therapy, which they felt to be ineffective.
The primary study endpoint was the change in Reflux Symptom Index scores from baseline through week 4 of nighttime use of the assist device. The median score improved from 27 at baseline to 14 at week 2 and 12 at week 4. That translated to a mean 54% reduction. A total of 86% of subjects were deemed to have experienced treatment success as defined by at least a 25% improvement in their symptom score; 30% of participants showed a greater than 75% improvement.
Treatment side effects were few, mild, and brief, consisting chiefly of skin irritation from the device’s Velcro strap. No one withdrew from the study.
The Reza-Band device was developed by gastroenterologists at the Medical College of Wisconsin, Milwaukee. Dr. Vaezi said he and his coinvestigators decided to formally investigate the device because "we were intrigued and impressed by the fact that it has helped so many patients."
"For me, it’s a welcome development," Dr. Vaezi added. "Just anecdotally, many patients in the study wanted to know how they could purchase this device. It’s really welcome in an area [in which] we don’t have many therapeutic options."
Still, he noted, it’s probably a good idea for device users to continue to keep a couple of bricks under the head of the bed, in accord with standard medical advice.
Future plans include a sham-controlled study designed to assess the device’s true benefit over placebo therapy; this will be an important study because EER is known to have a substantial placebo response rate, he continued. Also, plans are afoot for a safety study assessing whether the device has any clinically meaningful impact upon intraocular pressure.
The study was supported by Somna Therapeutics, which is developing the device. Dr. Vaezi reported having no financial relationship with the company.
AT DDW 2014
Key clinical point: An investigational device that modestly increases intraluminal pressure at the upper esophageal sphincter shows promise as a noninvasive therapy for extraesophageal reflux, a common disorder for which no effective pharmacotherapy exists.
Major finding: Patients with extraesophageal reflux experienced a mean 54% reduction in scores on the Reflux Symptom Index after 4 weeks of nighttime use of the upper esophageal sphincter assist device.
Data source: This was a 4-week multicenter, prospective, uncontrolled study involving 47 patients.
Disclosures: The study was supported by Somna Therapeutics. The presenter reported having no financial relationship with the company.
Atopic dermatitis update taps top therapy choices
WAIKOLOA, HAWAII – The 2014 update of the American Academy of Dermatology guidelines on atopic dermatitis take a strong stance in favor of the use of topical calcineurin inhibitors as topical corticosteroid-sparing agents, even in children less than 2 years old, where the use of the medications remains off-label.
The evidence-based AAD guidelines bestow a Class A, Level of Evidence I recommendation for the use of topical calcineurin inhibitors as topical steroid-sparing agents. The proactive use of topical calcineurin inhibitors as proactively scheduled, short-term, intermittent maintenance therapy to prevent disease flares also gets an A-I recommendation in the guidelines. The report states that there is no need to monitor blood levels of topical calcineurin inhibitors, Dr. Wynnis Tom noted at the Hawaii Dermatology Seminar, sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The guidelines emphasize the importance of having a proactive discussion with the patient and/or parents about the black box warning for topical calcineurin inhibitors, stressing the fact that interim analyses of long-term surveillance studies do not show an increase in malignancies, said Dr. Tom, a member of the working group that developed the AAD guidelines and a pediatric dermatologist at the University of California, San Diego.
The section of the guidelines devoted to topical therapies includes a detailed description of wet wrap therapy, which is useful for quickly reducing atopic dermatitis severity during flares. The use of topical antihistamines gets a strong thumbs-down in the guidelines.
The guidelines go into considerable detail about the importance of fixing the skin barrier. Bathing is recommended as an important component of therapy, with the caveat that there is no good evidence as to the optimal frequency or duration.
"You want to get the crusting off and hydrate the skin; but you do have to be careful of how long the bath lasts, because you don’t want the skin to dry out as the water evaporates," Dr. Tom said. "I find bathing even daily is good, so long as people are using moisturizers afterward. That’s the key part both for treatment and maintenance: liberal use of moisturizers."
The guideline panel determined that while moisturizers are a cornerstone of atopic dermatitis therapy, no one moisturizer product has been shown to be better than the others. And that includes the prescription devices containing ceramides or hydrolipids, which haven’t persuasively been shown to have clinical advantages over inexpensive over-the-counter moisturizers.
The 2014 atopic dermatitis guidelines are the first-ever AAD guidelines to include an entire section devoted to the diagnosis of a dermatologic disorder (J. Am. Acad. Dermatol. 2014;70:338-51). This was deemed necessary because misdiagnosis of atopic dermatitis is a problem, particularly in adults.
The guidelines stress that atopic dermatitis is a clinical diagnosis that requires ruling out conditions including contact dermatitis, cutaneous T-cell lymphoma, psoriasis, photosensitivity reactions, seborrheic dermatitis, and immune deficiency diseases. At this time, no specific biomarkers can be recommended for the diagnosis of atopic dermatitis or assessment of its severity. In particular, according to the guidelines, the popular practice of monitoring immunoglobulin E levels isn’t recommended.
The comprehensive guidelines also include a section on phototherapy and systemic agents. In addition, a section on preventing disease flares and the use of adjunctive therapies is slated for release in June.
Dr. Tom reported serving as a financially uncompensated investigator for studies sponsored by Amgen and Anacor. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The 2014 update of the American Academy of Dermatology guidelines on atopic dermatitis take a strong stance in favor of the use of topical calcineurin inhibitors as topical corticosteroid-sparing agents, even in children less than 2 years old, where the use of the medications remains off-label.
The evidence-based AAD guidelines bestow a Class A, Level of Evidence I recommendation for the use of topical calcineurin inhibitors as topical steroid-sparing agents. The proactive use of topical calcineurin inhibitors as proactively scheduled, short-term, intermittent maintenance therapy to prevent disease flares also gets an A-I recommendation in the guidelines. The report states that there is no need to monitor blood levels of topical calcineurin inhibitors, Dr. Wynnis Tom noted at the Hawaii Dermatology Seminar, sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The guidelines emphasize the importance of having a proactive discussion with the patient and/or parents about the black box warning for topical calcineurin inhibitors, stressing the fact that interim analyses of long-term surveillance studies do not show an increase in malignancies, said Dr. Tom, a member of the working group that developed the AAD guidelines and a pediatric dermatologist at the University of California, San Diego.
The section of the guidelines devoted to topical therapies includes a detailed description of wet wrap therapy, which is useful for quickly reducing atopic dermatitis severity during flares. The use of topical antihistamines gets a strong thumbs-down in the guidelines.
The guidelines go into considerable detail about the importance of fixing the skin barrier. Bathing is recommended as an important component of therapy, with the caveat that there is no good evidence as to the optimal frequency or duration.
"You want to get the crusting off and hydrate the skin; but you do have to be careful of how long the bath lasts, because you don’t want the skin to dry out as the water evaporates," Dr. Tom said. "I find bathing even daily is good, so long as people are using moisturizers afterward. That’s the key part both for treatment and maintenance: liberal use of moisturizers."
The guideline panel determined that while moisturizers are a cornerstone of atopic dermatitis therapy, no one moisturizer product has been shown to be better than the others. And that includes the prescription devices containing ceramides or hydrolipids, which haven’t persuasively been shown to have clinical advantages over inexpensive over-the-counter moisturizers.
The 2014 atopic dermatitis guidelines are the first-ever AAD guidelines to include an entire section devoted to the diagnosis of a dermatologic disorder (J. Am. Acad. Dermatol. 2014;70:338-51). This was deemed necessary because misdiagnosis of atopic dermatitis is a problem, particularly in adults.
The guidelines stress that atopic dermatitis is a clinical diagnosis that requires ruling out conditions including contact dermatitis, cutaneous T-cell lymphoma, psoriasis, photosensitivity reactions, seborrheic dermatitis, and immune deficiency diseases. At this time, no specific biomarkers can be recommended for the diagnosis of atopic dermatitis or assessment of its severity. In particular, according to the guidelines, the popular practice of monitoring immunoglobulin E levels isn’t recommended.
The comprehensive guidelines also include a section on phototherapy and systemic agents. In addition, a section on preventing disease flares and the use of adjunctive therapies is slated for release in June.
Dr. Tom reported serving as a financially uncompensated investigator for studies sponsored by Amgen and Anacor. SDEF and this news organization are owned by the same parent company.
WAIKOLOA, HAWAII – The 2014 update of the American Academy of Dermatology guidelines on atopic dermatitis take a strong stance in favor of the use of topical calcineurin inhibitors as topical corticosteroid-sparing agents, even in children less than 2 years old, where the use of the medications remains off-label.
The evidence-based AAD guidelines bestow a Class A, Level of Evidence I recommendation for the use of topical calcineurin inhibitors as topical steroid-sparing agents. The proactive use of topical calcineurin inhibitors as proactively scheduled, short-term, intermittent maintenance therapy to prevent disease flares also gets an A-I recommendation in the guidelines. The report states that there is no need to monitor blood levels of topical calcineurin inhibitors, Dr. Wynnis Tom noted at the Hawaii Dermatology Seminar, sponsored by the Global Academy for Medical Education/Skin Disease Education Foundation.
The guidelines emphasize the importance of having a proactive discussion with the patient and/or parents about the black box warning for topical calcineurin inhibitors, stressing the fact that interim analyses of long-term surveillance studies do not show an increase in malignancies, said Dr. Tom, a member of the working group that developed the AAD guidelines and a pediatric dermatologist at the University of California, San Diego.
The section of the guidelines devoted to topical therapies includes a detailed description of wet wrap therapy, which is useful for quickly reducing atopic dermatitis severity during flares. The use of topical antihistamines gets a strong thumbs-down in the guidelines.
The guidelines go into considerable detail about the importance of fixing the skin barrier. Bathing is recommended as an important component of therapy, with the caveat that there is no good evidence as to the optimal frequency or duration.
"You want to get the crusting off and hydrate the skin; but you do have to be careful of how long the bath lasts, because you don’t want the skin to dry out as the water evaporates," Dr. Tom said. "I find bathing even daily is good, so long as people are using moisturizers afterward. That’s the key part both for treatment and maintenance: liberal use of moisturizers."
The guideline panel determined that while moisturizers are a cornerstone of atopic dermatitis therapy, no one moisturizer product has been shown to be better than the others. And that includes the prescription devices containing ceramides or hydrolipids, which haven’t persuasively been shown to have clinical advantages over inexpensive over-the-counter moisturizers.
The 2014 atopic dermatitis guidelines are the first-ever AAD guidelines to include an entire section devoted to the diagnosis of a dermatologic disorder (J. Am. Acad. Dermatol. 2014;70:338-51). This was deemed necessary because misdiagnosis of atopic dermatitis is a problem, particularly in adults.
The guidelines stress that atopic dermatitis is a clinical diagnosis that requires ruling out conditions including contact dermatitis, cutaneous T-cell lymphoma, psoriasis, photosensitivity reactions, seborrheic dermatitis, and immune deficiency diseases. At this time, no specific biomarkers can be recommended for the diagnosis of atopic dermatitis or assessment of its severity. In particular, according to the guidelines, the popular practice of monitoring immunoglobulin E levels isn’t recommended.
The comprehensive guidelines also include a section on phototherapy and systemic agents. In addition, a section on preventing disease flares and the use of adjunctive therapies is slated for release in June.
Dr. Tom reported serving as a financially uncompensated investigator for studies sponsored by Amgen and Anacor. SDEF and this news organization are owned by the same parent company.
EXPERT ANALYSIS FROM SDEF HAWAII DERMATOLOGY SEMINAR