User login
VIDEO: Awardee's work targets mucosal stem cells in Crohn’s, ulcerative colitis
BOSTON – A series of important technological advances in cloning stem cells of the gastrointestinal tract is rewriting the basic concepts surrounding these cells and has the potential to advance understanding of GI conditions such as Barrett’s esophagus and Crohn’s disease, according to the winner of the 2016 AGA-Medtronic Research and Development Pilot Award in Technology.
The award is sponsored by Medtronic and administered through the AGA Research Foundation. “Medtronic is pleased to partner with the AGA Research Foundation in supporting research to improve patient outcomes,” said Vafa Jamali, president of Early Technologies’ business with Medtronic. “We are dedicated to support patient-friendly innovations enabling the early detection and treatment of chronic GI diseases and cancers.”
“The enabling technology we developed is the means of cloning and propagating single mucosal stem cells from discrete regions of the gastrointestinal tract in their most immature form. What we have shown is that these stem cells from normal individuals are essentially immortal, are genetically stable, and, critically, are rigorously regiospecific,” Dr. Xian of the Institute of Molecular Medicine for the Prevention of Human Diseases, University of Texas Health Science Center, Houston, said in an interview.
“This regiospecificity – duodenum stem cells always yield duodenum epithelia, ascending colon stem cells produce ascending colon epithelia – has important implications for both regenerative medicine and drug discovery,” she added.
With regard to the dominant stem cell technologies in use today, including induced pluripotent stem cells and organoid cultures of the gastrointestinal tract, Dr. Xian’s technology represents a paradigm shift.
Dr. Xian’s system generates pure “ground state” stem cells that differentiate in 3-D culture to an epithelium indistinguishable from textbook images of in situ epithelia, at rates 250 times faster than intestinal organoid systems.
This system is being applied in major investigations into Barrett’s esophagus and inflammatory bowel disease in collaboration with investigators at multiple centers in the United States and Singapore.
For Barrett’s, Dr. Xian has developed patient-matched stem cells from endoscopic biopsies of esophageal, Barrett’s, and gastric cardia to show that each arises from distinct lineages, and that the Barrett’s stem cells accumulate genetic alterations not found in those of the matched normal epithelia. These stem cells have been adapted to a drug-screening platform to identify leads that specifically eliminate Barrett’s with an eye on preemptive therapies.
Her work on mucosal stem cells in patients with Crohn’s and ulcerative colitis is revealing a coexistence of normal and pathogenic stem cells in these patients that may alter concepts of both the etiology and treatment of these conditions.
Dr. Xian suggests that treatment of Crohn’s may become analogous to treatment of cancer – emphasizing preservation of healthy cells while eliminating diseased cells – and move therapy from the current effort to induce a quiescent inflammatory state to one in which the goal is cure.
“These findings appear to indicate that some of the chronic inflammatory conditions so prevalent in gastroenterology, as well as the cancers arising from them, may have as their basis profound alterations in mucosal stem cell biology,” she added.
Though it’s still early days with this technology, its single-cell resolution naturally melds with other emerging genomic and genome editing technologies into a powerful means of dissecting the molecular basis of disease and realizing the potential of autologous regenerative medicine, according to Dr. Xian.
Following Dr. Xian’s presentation, Michael L. Kochman, MD, who is executive committee chair of the AGA Center for GI Innovation and Technology, announced the winner of the 2017 AGA-Medtronic Research and Development Pilot Award in Technology. The award was presented to Bani Chander Roland, MD, of Lenox Hill Hospital & Northwell Health System, New York. Her research will involve assessment of the ileocecal valve.
“She has several aims in this proposal to really understand better the causes of ileosphincter dysfunction using a novel method of measuring ileocecal junction pressures,” reported Dr. Kochman. He explained that other parts of the proposal included ileocecal evaluation with wireless capsule endoscopy as well as an evaluation of flora across the GI tract.
A new award to foster innovation in GI medtech is also planned. In partnership with Boston Scientific, the AGA has established the AGA-Boston Scientific Technology and Innovation Pilot Award. This $30,000 research grant will support investigators at any career stage who are developing and testing new medical devices and technologies with applications in gastroenterology and hepatology. The details of the award will be available on the AGA’s website in May. Applications for the award will be accepted beginning in the fall.
BOSTON – A series of important technological advances in cloning stem cells of the gastrointestinal tract is rewriting the basic concepts surrounding these cells and has the potential to advance understanding of GI conditions such as Barrett’s esophagus and Crohn’s disease, according to the winner of the 2016 AGA-Medtronic Research and Development Pilot Award in Technology.
The award is sponsored by Medtronic and administered through the AGA Research Foundation. “Medtronic is pleased to partner with the AGA Research Foundation in supporting research to improve patient outcomes,” said Vafa Jamali, president of Early Technologies’ business with Medtronic. “We are dedicated to support patient-friendly innovations enabling the early detection and treatment of chronic GI diseases and cancers.”
“The enabling technology we developed is the means of cloning and propagating single mucosal stem cells from discrete regions of the gastrointestinal tract in their most immature form. What we have shown is that these stem cells from normal individuals are essentially immortal, are genetically stable, and, critically, are rigorously regiospecific,” Dr. Xian of the Institute of Molecular Medicine for the Prevention of Human Diseases, University of Texas Health Science Center, Houston, said in an interview.
“This regiospecificity – duodenum stem cells always yield duodenum epithelia, ascending colon stem cells produce ascending colon epithelia – has important implications for both regenerative medicine and drug discovery,” she added.
With regard to the dominant stem cell technologies in use today, including induced pluripotent stem cells and organoid cultures of the gastrointestinal tract, Dr. Xian’s technology represents a paradigm shift.
Dr. Xian’s system generates pure “ground state” stem cells that differentiate in 3-D culture to an epithelium indistinguishable from textbook images of in situ epithelia, at rates 250 times faster than intestinal organoid systems.
This system is being applied in major investigations into Barrett’s esophagus and inflammatory bowel disease in collaboration with investigators at multiple centers in the United States and Singapore.
For Barrett’s, Dr. Xian has developed patient-matched stem cells from endoscopic biopsies of esophageal, Barrett’s, and gastric cardia to show that each arises from distinct lineages, and that the Barrett’s stem cells accumulate genetic alterations not found in those of the matched normal epithelia. These stem cells have been adapted to a drug-screening platform to identify leads that specifically eliminate Barrett’s with an eye on preemptive therapies.
Her work on mucosal stem cells in patients with Crohn’s and ulcerative colitis is revealing a coexistence of normal and pathogenic stem cells in these patients that may alter concepts of both the etiology and treatment of these conditions.
Dr. Xian suggests that treatment of Crohn’s may become analogous to treatment of cancer – emphasizing preservation of healthy cells while eliminating diseased cells – and move therapy from the current effort to induce a quiescent inflammatory state to one in which the goal is cure.
“These findings appear to indicate that some of the chronic inflammatory conditions so prevalent in gastroenterology, as well as the cancers arising from them, may have as their basis profound alterations in mucosal stem cell biology,” she added.
Though it’s still early days with this technology, its single-cell resolution naturally melds with other emerging genomic and genome editing technologies into a powerful means of dissecting the molecular basis of disease and realizing the potential of autologous regenerative medicine, according to Dr. Xian.
Following Dr. Xian’s presentation, Michael L. Kochman, MD, who is executive committee chair of the AGA Center for GI Innovation and Technology, announced the winner of the 2017 AGA-Medtronic Research and Development Pilot Award in Technology. The award was presented to Bani Chander Roland, MD, of Lenox Hill Hospital & Northwell Health System, New York. Her research will involve assessment of the ileocecal valve.
“She has several aims in this proposal to really understand better the causes of ileosphincter dysfunction using a novel method of measuring ileocecal junction pressures,” reported Dr. Kochman. He explained that other parts of the proposal included ileocecal evaluation with wireless capsule endoscopy as well as an evaluation of flora across the GI tract.
A new award to foster innovation in GI medtech is also planned. In partnership with Boston Scientific, the AGA has established the AGA-Boston Scientific Technology and Innovation Pilot Award. This $30,000 research grant will support investigators at any career stage who are developing and testing new medical devices and technologies with applications in gastroenterology and hepatology. The details of the award will be available on the AGA’s website in May. Applications for the award will be accepted beginning in the fall.
BOSTON – A series of important technological advances in cloning stem cells of the gastrointestinal tract is rewriting the basic concepts surrounding these cells and has the potential to advance understanding of GI conditions such as Barrett’s esophagus and Crohn’s disease, according to the winner of the 2016 AGA-Medtronic Research and Development Pilot Award in Technology.
The award is sponsored by Medtronic and administered through the AGA Research Foundation. “Medtronic is pleased to partner with the AGA Research Foundation in supporting research to improve patient outcomes,” said Vafa Jamali, president of Early Technologies’ business with Medtronic. “We are dedicated to support patient-friendly innovations enabling the early detection and treatment of chronic GI diseases and cancers.”
“The enabling technology we developed is the means of cloning and propagating single mucosal stem cells from discrete regions of the gastrointestinal tract in their most immature form. What we have shown is that these stem cells from normal individuals are essentially immortal, are genetically stable, and, critically, are rigorously regiospecific,” Dr. Xian of the Institute of Molecular Medicine for the Prevention of Human Diseases, University of Texas Health Science Center, Houston, said in an interview.
“This regiospecificity – duodenum stem cells always yield duodenum epithelia, ascending colon stem cells produce ascending colon epithelia – has important implications for both regenerative medicine and drug discovery,” she added.
With regard to the dominant stem cell technologies in use today, including induced pluripotent stem cells and organoid cultures of the gastrointestinal tract, Dr. Xian’s technology represents a paradigm shift.
Dr. Xian’s system generates pure “ground state” stem cells that differentiate in 3-D culture to an epithelium indistinguishable from textbook images of in situ epithelia, at rates 250 times faster than intestinal organoid systems.
This system is being applied in major investigations into Barrett’s esophagus and inflammatory bowel disease in collaboration with investigators at multiple centers in the United States and Singapore.
For Barrett’s, Dr. Xian has developed patient-matched stem cells from endoscopic biopsies of esophageal, Barrett’s, and gastric cardia to show that each arises from distinct lineages, and that the Barrett’s stem cells accumulate genetic alterations not found in those of the matched normal epithelia. These stem cells have been adapted to a drug-screening platform to identify leads that specifically eliminate Barrett’s with an eye on preemptive therapies.
Her work on mucosal stem cells in patients with Crohn’s and ulcerative colitis is revealing a coexistence of normal and pathogenic stem cells in these patients that may alter concepts of both the etiology and treatment of these conditions.
Dr. Xian suggests that treatment of Crohn’s may become analogous to treatment of cancer – emphasizing preservation of healthy cells while eliminating diseased cells – and move therapy from the current effort to induce a quiescent inflammatory state to one in which the goal is cure.
“These findings appear to indicate that some of the chronic inflammatory conditions so prevalent in gastroenterology, as well as the cancers arising from them, may have as their basis profound alterations in mucosal stem cell biology,” she added.
Though it’s still early days with this technology, its single-cell resolution naturally melds with other emerging genomic and genome editing technologies into a powerful means of dissecting the molecular basis of disease and realizing the potential of autologous regenerative medicine, according to Dr. Xian.
Following Dr. Xian’s presentation, Michael L. Kochman, MD, who is executive committee chair of the AGA Center for GI Innovation and Technology, announced the winner of the 2017 AGA-Medtronic Research and Development Pilot Award in Technology. The award was presented to Bani Chander Roland, MD, of Lenox Hill Hospital & Northwell Health System, New York. Her research will involve assessment of the ileocecal valve.
“She has several aims in this proposal to really understand better the causes of ileosphincter dysfunction using a novel method of measuring ileocecal junction pressures,” reported Dr. Kochman. He explained that other parts of the proposal included ileocecal evaluation with wireless capsule endoscopy as well as an evaluation of flora across the GI tract.
A new award to foster innovation in GI medtech is also planned. In partnership with Boston Scientific, the AGA has established the AGA-Boston Scientific Technology and Innovation Pilot Award. This $30,000 research grant will support investigators at any career stage who are developing and testing new medical devices and technologies with applications in gastroenterology and hepatology. The details of the award will be available on the AGA’s website in May. Applications for the award will be accepted beginning in the fall.
FROM THE AGA 2017 TECH SUMMIT
VIDEO: Making the most of MACRA reimbursement methods
BOSTON – “There’s a hunger out there” among physicians to participate in novel practice models made possible by value-based care, according to Lawrence R. Kosinski, MD, MBA, AGAF, clinical private practice councilor on the American Gastroenterological Association governing board.
In this video interview, conducted during the 2017 AGA Tech Summit, Dr. Kosinski explained how physicians can leverage the reimbursement methods of the recent MACRA legislation in their practices. He discusses how, in his own practice, he and his colleagues created an alternative payment model that uses a proprietary patient portal, SonarMD, to help physicians improve the management of chronic diseases such as inflammatory bowel disease at a lower cost and with markedly improved patient satisfaction.
“We look forward very much to the success of this project going forward, and the AGA will continue to be an integral part of the development of Project Sonar,” he said.
Dr. Kosinski is president of SonarMD.
BOSTON – “There’s a hunger out there” among physicians to participate in novel practice models made possible by value-based care, according to Lawrence R. Kosinski, MD, MBA, AGAF, clinical private practice councilor on the American Gastroenterological Association governing board.
In this video interview, conducted during the 2017 AGA Tech Summit, Dr. Kosinski explained how physicians can leverage the reimbursement methods of the recent MACRA legislation in their practices. He discusses how, in his own practice, he and his colleagues created an alternative payment model that uses a proprietary patient portal, SonarMD, to help physicians improve the management of chronic diseases such as inflammatory bowel disease at a lower cost and with markedly improved patient satisfaction.
“We look forward very much to the success of this project going forward, and the AGA will continue to be an integral part of the development of Project Sonar,” he said.
Dr. Kosinski is president of SonarMD.
BOSTON – “There’s a hunger out there” among physicians to participate in novel practice models made possible by value-based care, according to Lawrence R. Kosinski, MD, MBA, AGAF, clinical private practice councilor on the American Gastroenterological Association governing board.
In this video interview, conducted during the 2017 AGA Tech Summit, Dr. Kosinski explained how physicians can leverage the reimbursement methods of the recent MACRA legislation in their practices. He discusses how, in his own practice, he and his colleagues created an alternative payment model that uses a proprietary patient portal, SonarMD, to help physicians improve the management of chronic diseases such as inflammatory bowel disease at a lower cost and with markedly improved patient satisfaction.
“We look forward very much to the success of this project going forward, and the AGA will continue to be an integral part of the development of Project Sonar,” he said.
Dr. Kosinski is president of SonarMD.
FROM THE 2017 AGA TECH SUMMIT
VIDEO: AGA Center chair seeks to streamline innovation
BOSTON – A lot has changed in GI technology since V. Raman Muthusamy, MD, the incoming chair of the AGA Center for GI Innovation and Technology(CGIT), completed his GI fellowship training 15 years ago.
“I would say that between half and two-thirds of what I do today for actual patient care delivery is from technologies that have been completed since my fellowship or [from] applications of those technologies that are new,” he said in a video interview conducted during the 2017 AGA Technology Summit, which is sponsored by the center. So if you think you’re not impacted by innovation, think again.
“I think like many practicing physicians we have great technology, but we can think of ways to do things better,” continued Dr. Muthusamy, professor of medicine at the David Geffen School of Medicine at the University of California, Los Angeles.
One of the goals of the center is to streamline the process of taking a good idea and making it into a product and a company. Dr. Muthusamy said such streamlining is important because the current “daunting” development process has led to many good ideas “dying on the vine” before they could be implemented. He says the CGIT, by bringing together key stakeholders from the medical, business, and regulatory arenas, can help bring forth the latest technologies to directly benefit GI patients.
BOSTON – A lot has changed in GI technology since V. Raman Muthusamy, MD, the incoming chair of the AGA Center for GI Innovation and Technology(CGIT), completed his GI fellowship training 15 years ago.
“I would say that between half and two-thirds of what I do today for actual patient care delivery is from technologies that have been completed since my fellowship or [from] applications of those technologies that are new,” he said in a video interview conducted during the 2017 AGA Technology Summit, which is sponsored by the center. So if you think you’re not impacted by innovation, think again.
“I think like many practicing physicians we have great technology, but we can think of ways to do things better,” continued Dr. Muthusamy, professor of medicine at the David Geffen School of Medicine at the University of California, Los Angeles.
One of the goals of the center is to streamline the process of taking a good idea and making it into a product and a company. Dr. Muthusamy said such streamlining is important because the current “daunting” development process has led to many good ideas “dying on the vine” before they could be implemented. He says the CGIT, by bringing together key stakeholders from the medical, business, and regulatory arenas, can help bring forth the latest technologies to directly benefit GI patients.
BOSTON – A lot has changed in GI technology since V. Raman Muthusamy, MD, the incoming chair of the AGA Center for GI Innovation and Technology(CGIT), completed his GI fellowship training 15 years ago.
“I would say that between half and two-thirds of what I do today for actual patient care delivery is from technologies that have been completed since my fellowship or [from] applications of those technologies that are new,” he said in a video interview conducted during the 2017 AGA Technology Summit, which is sponsored by the center. So if you think you’re not impacted by innovation, think again.
“I think like many practicing physicians we have great technology, but we can think of ways to do things better,” continued Dr. Muthusamy, professor of medicine at the David Geffen School of Medicine at the University of California, Los Angeles.
One of the goals of the center is to streamline the process of taking a good idea and making it into a product and a company. Dr. Muthusamy said such streamlining is important because the current “daunting” development process has led to many good ideas “dying on the vine” before they could be implemented. He says the CGIT, by bringing together key stakeholders from the medical, business, and regulatory arenas, can help bring forth the latest technologies to directly benefit GI patients.
VIDEO: Innovation fuels push toward therapeutic, safety advances in gastroenterology
BOSTON – In the next 5 years there is going to be a “huge swing” in gastroenterology to minimally invasive therapies, improved diagnostics, and additional therapeutics and disease management strategies that can be applied on an ambulatory basis and, perhaps, even in the office.
That’s the prediction of Michael L. Kochman, MD, AGAF, of the University of Pennsylvania, Philadelphia, who is the executive committee chair of the American Gastroenterological Association Center for GI Innovation and Technology. In a video interview conducted at the 2017 AGA Tech Summit, sponsored by the center, Dr. Kochman discussed five emerging trends in gastroenterology.
The trends include advances in endoscopic technologies and an expansion of the role of personalized medicine that would allow gastroenterologists to better target medications to individual patients. Office-based physicians will be able to evaluate patient risk for various diseases more easily through the use of stool or blood samples or cheek swabs and, then, steer toward additional tests for those found to be at high risk. Improvements in patient safety also are on the horizon, he said, as new technologies will provide earlier warning signs of failed therapies and potential complications.
Finally, as innovation continues in the area of minimally invasive surgery and transorifice surgery, “we need to look more and more to what we will end up calling organ-preserving surgeries, where we are able to treat just the area of pathology,” he noted. “We will be moving to leaving the patient without lasting scars and remembrances of having to have an organ removed.”
Dr. Kochman disclosed that he serves as a consultant to Boston Scientific and Dark Canyon Labs.
BOSTON – In the next 5 years there is going to be a “huge swing” in gastroenterology to minimally invasive therapies, improved diagnostics, and additional therapeutics and disease management strategies that can be applied on an ambulatory basis and, perhaps, even in the office.
That’s the prediction of Michael L. Kochman, MD, AGAF, of the University of Pennsylvania, Philadelphia, who is the executive committee chair of the American Gastroenterological Association Center for GI Innovation and Technology. In a video interview conducted at the 2017 AGA Tech Summit, sponsored by the center, Dr. Kochman discussed five emerging trends in gastroenterology.
The trends include advances in endoscopic technologies and an expansion of the role of personalized medicine that would allow gastroenterologists to better target medications to individual patients. Office-based physicians will be able to evaluate patient risk for various diseases more easily through the use of stool or blood samples or cheek swabs and, then, steer toward additional tests for those found to be at high risk. Improvements in patient safety also are on the horizon, he said, as new technologies will provide earlier warning signs of failed therapies and potential complications.
Finally, as innovation continues in the area of minimally invasive surgery and transorifice surgery, “we need to look more and more to what we will end up calling organ-preserving surgeries, where we are able to treat just the area of pathology,” he noted. “We will be moving to leaving the patient without lasting scars and remembrances of having to have an organ removed.”
Dr. Kochman disclosed that he serves as a consultant to Boston Scientific and Dark Canyon Labs.
BOSTON – In the next 5 years there is going to be a “huge swing” in gastroenterology to minimally invasive therapies, improved diagnostics, and additional therapeutics and disease management strategies that can be applied on an ambulatory basis and, perhaps, even in the office.
That’s the prediction of Michael L. Kochman, MD, AGAF, of the University of Pennsylvania, Philadelphia, who is the executive committee chair of the American Gastroenterological Association Center for GI Innovation and Technology. In a video interview conducted at the 2017 AGA Tech Summit, sponsored by the center, Dr. Kochman discussed five emerging trends in gastroenterology.
The trends include advances in endoscopic technologies and an expansion of the role of personalized medicine that would allow gastroenterologists to better target medications to individual patients. Office-based physicians will be able to evaluate patient risk for various diseases more easily through the use of stool or blood samples or cheek swabs and, then, steer toward additional tests for those found to be at high risk. Improvements in patient safety also are on the horizon, he said, as new technologies will provide earlier warning signs of failed therapies and potential complications.
Finally, as innovation continues in the area of minimally invasive surgery and transorifice surgery, “we need to look more and more to what we will end up calling organ-preserving surgeries, where we are able to treat just the area of pathology,” he noted. “We will be moving to leaving the patient without lasting scars and remembrances of having to have an organ removed.”
Dr. Kochman disclosed that he serves as a consultant to Boston Scientific and Dark Canyon Labs.
FROM THE 2017 AGA TECH SUMMIT
VIDEO: Five ‘revolutionary’ market forces poised to impact medical practice
BOSTON – Five “revolutionary” forces in medicine are going to impact patients and physicians, two health care consultants say, and the federal government will act either as a brake or an accelerator on those forces.
In a video interview conducted during the 2017 AGA Tech Summit, Patrick D. Pilch, managing director and national healthcare advisory leader at the BDO Center for Healthcare Excellence and Innovation, New York, and his colleague David B. Friend, MD, MBA, managing director and chief medical officer at the center, discussed these market forces and their potential effects on the practice of medicine.
One such force is the convergence of molecular biology and computer science, which will lead to the creation of new medical devices that use software and will require Food and Drug Administration approval, including devices that can remotely monitor patient compliance with taking medication. Some health care apps also would need FDA approval.
“Increasingly, health care will be mobile,” Dr. Friend continued. “The concept of the right care at the right place at the right time at the right cost is going to increase, and the ability to deliver this in the mobile setting is going to be very powerful.”
A change in who bears financial risk is another important driver. Insurance companies are decreasing their risk, Dr. Friend said, while health care providers and health systems are bearing more financial risk as they increasingly accept bundled payments and value-based payments. Physicians will need to understand and be prepared for this shift in risk, he warned.
Dr. Friend also noted a move toward an integrated supply chain model that will have “profound implications” throughout the delivery process and a movement of care away from the hospital to a nonhospital setting such as home care or skilled nursing facilities.
The summit was sponsored by the AGA Center for GI Innovation and Technology.
BOSTON – Five “revolutionary” forces in medicine are going to impact patients and physicians, two health care consultants say, and the federal government will act either as a brake or an accelerator on those forces.
In a video interview conducted during the 2017 AGA Tech Summit, Patrick D. Pilch, managing director and national healthcare advisory leader at the BDO Center for Healthcare Excellence and Innovation, New York, and his colleague David B. Friend, MD, MBA, managing director and chief medical officer at the center, discussed these market forces and their potential effects on the practice of medicine.
One such force is the convergence of molecular biology and computer science, which will lead to the creation of new medical devices that use software and will require Food and Drug Administration approval, including devices that can remotely monitor patient compliance with taking medication. Some health care apps also would need FDA approval.
“Increasingly, health care will be mobile,” Dr. Friend continued. “The concept of the right care at the right place at the right time at the right cost is going to increase, and the ability to deliver this in the mobile setting is going to be very powerful.”
A change in who bears financial risk is another important driver. Insurance companies are decreasing their risk, Dr. Friend said, while health care providers and health systems are bearing more financial risk as they increasingly accept bundled payments and value-based payments. Physicians will need to understand and be prepared for this shift in risk, he warned.
Dr. Friend also noted a move toward an integrated supply chain model that will have “profound implications” throughout the delivery process and a movement of care away from the hospital to a nonhospital setting such as home care or skilled nursing facilities.
The summit was sponsored by the AGA Center for GI Innovation and Technology.
BOSTON – Five “revolutionary” forces in medicine are going to impact patients and physicians, two health care consultants say, and the federal government will act either as a brake or an accelerator on those forces.
In a video interview conducted during the 2017 AGA Tech Summit, Patrick D. Pilch, managing director and national healthcare advisory leader at the BDO Center for Healthcare Excellence and Innovation, New York, and his colleague David B. Friend, MD, MBA, managing director and chief medical officer at the center, discussed these market forces and their potential effects on the practice of medicine.
One such force is the convergence of molecular biology and computer science, which will lead to the creation of new medical devices that use software and will require Food and Drug Administration approval, including devices that can remotely monitor patient compliance with taking medication. Some health care apps also would need FDA approval.
“Increasingly, health care will be mobile,” Dr. Friend continued. “The concept of the right care at the right place at the right time at the right cost is going to increase, and the ability to deliver this in the mobile setting is going to be very powerful.”
A change in who bears financial risk is another important driver. Insurance companies are decreasing their risk, Dr. Friend said, while health care providers and health systems are bearing more financial risk as they increasingly accept bundled payments and value-based payments. Physicians will need to understand and be prepared for this shift in risk, he warned.
Dr. Friend also noted a move toward an integrated supply chain model that will have “profound implications” throughout the delivery process and a movement of care away from the hospital to a nonhospital setting such as home care or skilled nursing facilities.
The summit was sponsored by the AGA Center for GI Innovation and Technology.
EXPERT ANALYSIS FROM THE AGA 2017 TECH SUMMIT
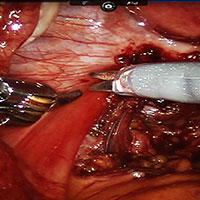
Robot-assisted laparoscopic excision of a rectovaginal endometriotic nodule
A rectovaginal endometriosis (RVE) is the most severe form of endometriosis. The gold standard for diagnosis is laparoscopy with histologic confirmation. A review of the literature suggests that surgery improves up to 70% of symptoms with generally favorable outcomes.
In this video, we provide a general introduction to endometriosis and a discussion of disease treatment options, ranging from hormonal suppression to radical bowel resections. We also illustrate the steps in robot-assisted laparoscopic excision of an RVE nodule:
- identify the borders of the rectosigmoid
- dissect the pararectal spaces
- release the rectosigmoid from its attachment to the RVE nodule
- identify and isolate the ureter(s)
- determine the margins of the nodule
- ensure complete resection.
Excision of an RVE nodule is a technically challenging surgical procedure. Use of the robot for resection is safe and feasible when performed by a trained and experienced surgeon.
I am pleased to bring you this video, and I hope that it is helpful to your practice.
>> Arnold P. Advincula, MD

Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
A rectovaginal endometriosis (RVE) is the most severe form of endometriosis. The gold standard for diagnosis is laparoscopy with histologic confirmation. A review of the literature suggests that surgery improves up to 70% of symptoms with generally favorable outcomes.
In this video, we provide a general introduction to endometriosis and a discussion of disease treatment options, ranging from hormonal suppression to radical bowel resections. We also illustrate the steps in robot-assisted laparoscopic excision of an RVE nodule:
- identify the borders of the rectosigmoid
- dissect the pararectal spaces
- release the rectosigmoid from its attachment to the RVE nodule
- identify and isolate the ureter(s)
- determine the margins of the nodule
- ensure complete resection.
Excision of an RVE nodule is a technically challenging surgical procedure. Use of the robot for resection is safe and feasible when performed by a trained and experienced surgeon.
I am pleased to bring you this video, and I hope that it is helpful to your practice.
>> Arnold P. Advincula, MD

Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
A rectovaginal endometriosis (RVE) is the most severe form of endometriosis. The gold standard for diagnosis is laparoscopy with histologic confirmation. A review of the literature suggests that surgery improves up to 70% of symptoms with generally favorable outcomes.
In this video, we provide a general introduction to endometriosis and a discussion of disease treatment options, ranging from hormonal suppression to radical bowel resections. We also illustrate the steps in robot-assisted laparoscopic excision of an RVE nodule:
- identify the borders of the rectosigmoid
- dissect the pararectal spaces
- release the rectosigmoid from its attachment to the RVE nodule
- identify and isolate the ureter(s)
- determine the margins of the nodule
- ensure complete resection.
Excision of an RVE nodule is a technically challenging surgical procedure. Use of the robot for resection is safe and feasible when performed by a trained and experienced surgeon.
I am pleased to bring you this video, and I hope that it is helpful to your practice.
>> Arnold P. Advincula, MD

Share your thoughts! Send your Letter to the Editor to rbarbieri@frontlinemedcom.com. Please include your name and the city and state in which you practice.
VIDEO: Blinatumomab, inotuzumab reshape relapsed ALL treatment
NEW YORK – A pair of new monoclonal antibodies have dramatically changed treatment for patients with acute lymphoblastic leukemia to prepare them for a stem cell transplant, Daniel J. DeAngelo, MD, said at a conference held by Imedex.
“We don’t use standard chemotherapy for reinduction anymore; we use blinatumomab or inotuzumab,” said Dr. DeAngelo, a hematologist oncologist at Dana-Farber Cancer Institute in Boston.
Blinatumomab (Blincyto), approved by the Food and Drug Administration in 2014, has produced “exceptional” response rates, becoming “standard of care” for patients with relapsed acute lymphoblastic leukemia (ALL) that does not have a Philadelphia chromosome, Dr. DeAngelo said in a video interview.
Approved based on results from a phase II study, blinatumomab’s efficacy and safety were recently further delineated in results from the first phase III trial (N Engl J Med. 2017 Mar 2;376[9]:836-74), with 376 treated patients. In that trial, blinatumomab more than doubled the complete remission rate, compared with control patients (34% vs. 16%), and nearly doubled median overall survival – 7.7 months with blinatumomab, compared with 4.0 months for control patients treated with standard chemotherapy.
These findings “further substantiated” blinatumomab’s role, he said.
Blinatumomab’s big limitations are certain adverse effects and the logistics of its dosing. The major adverse effect is “cytokine release syndrome,” which manifests as fever, low blood pressure, and neurologic toxicities that can range from tremors to encephalopathy and seizure. These are manageable by close observation of patients by experienced nurses, Dr. DeAngelo said.
Dosing involves 4 weeks of continuous infusion, starting with 10 days done entirely in the hospital, with the remaining 18 days with patients going home but needing to return every 48 hours to have their infusion bag changed. “Depending on how far the patient lives from the clinic, it can be a logistical challenge,” he said.
A second new antibody he has used on many patients is inotuzumab, which was accepted for review for approval by the FDA in February 2017, with action expected by August.
Dr. DeAngelo served as a coinvestigator in a phase III trial reported in 2016 with 218 evaluable patients. In that trial, investigators reported an 81% complete remission rate with inotuzumab treatment, compared with a 29% among control patients on chemotherapy (N Engl J Med. 2016 Aug 25;375[8]:740-53).
Inotuzumab was effective against patients with Philadelphia chromosome positive ALL, but it will not work for the roughly 5%-10% of ALL patients who lack CD-22 expression in their B-cell ALL.
Inotuzumab is easier to administer than blinatumomab, requiring a once a week infusion, and causes little immediate toxicity – although thrombocytopenia and liver-function abnormalities can occur with continued use, and the risk of veno-occlusive disease is increased when patients later receive a stem cell transplant, Dr. DeAngelo said.
“It’s nice to have options” when choosing antibody-based treatment, he said. Blinatumomab is a good choice for patients with a lower tumor burden – either patients with early relapse or with minimal residual disease – while inotuzumab works better for patients with more bulky disease, as well as those who are not able to accommodate the logistic demands of blinatumomab infusions.
Dr. DeAngelo also highlighted several trials now underway that are testing the efficacy of both antibodies when used as part of first-line treatment.
Dr. DeAngelo has been a consultant to Amgen, the company that markets blinatumomab (Blincyto); to Pfizer, the company developing inotuzumab; and to Ariad, InCyte, and Novartis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
NEW YORK – A pair of new monoclonal antibodies have dramatically changed treatment for patients with acute lymphoblastic leukemia to prepare them for a stem cell transplant, Daniel J. DeAngelo, MD, said at a conference held by Imedex.
“We don’t use standard chemotherapy for reinduction anymore; we use blinatumomab or inotuzumab,” said Dr. DeAngelo, a hematologist oncologist at Dana-Farber Cancer Institute in Boston.
Blinatumomab (Blincyto), approved by the Food and Drug Administration in 2014, has produced “exceptional” response rates, becoming “standard of care” for patients with relapsed acute lymphoblastic leukemia (ALL) that does not have a Philadelphia chromosome, Dr. DeAngelo said in a video interview.
Approved based on results from a phase II study, blinatumomab’s efficacy and safety were recently further delineated in results from the first phase III trial (N Engl J Med. 2017 Mar 2;376[9]:836-74), with 376 treated patients. In that trial, blinatumomab more than doubled the complete remission rate, compared with control patients (34% vs. 16%), and nearly doubled median overall survival – 7.7 months with blinatumomab, compared with 4.0 months for control patients treated with standard chemotherapy.
These findings “further substantiated” blinatumomab’s role, he said.
Blinatumomab’s big limitations are certain adverse effects and the logistics of its dosing. The major adverse effect is “cytokine release syndrome,” which manifests as fever, low blood pressure, and neurologic toxicities that can range from tremors to encephalopathy and seizure. These are manageable by close observation of patients by experienced nurses, Dr. DeAngelo said.
Dosing involves 4 weeks of continuous infusion, starting with 10 days done entirely in the hospital, with the remaining 18 days with patients going home but needing to return every 48 hours to have their infusion bag changed. “Depending on how far the patient lives from the clinic, it can be a logistical challenge,” he said.
A second new antibody he has used on many patients is inotuzumab, which was accepted for review for approval by the FDA in February 2017, with action expected by August.
Dr. DeAngelo served as a coinvestigator in a phase III trial reported in 2016 with 218 evaluable patients. In that trial, investigators reported an 81% complete remission rate with inotuzumab treatment, compared with a 29% among control patients on chemotherapy (N Engl J Med. 2016 Aug 25;375[8]:740-53).
Inotuzumab was effective against patients with Philadelphia chromosome positive ALL, but it will not work for the roughly 5%-10% of ALL patients who lack CD-22 expression in their B-cell ALL.
Inotuzumab is easier to administer than blinatumomab, requiring a once a week infusion, and causes little immediate toxicity – although thrombocytopenia and liver-function abnormalities can occur with continued use, and the risk of veno-occlusive disease is increased when patients later receive a stem cell transplant, Dr. DeAngelo said.
“It’s nice to have options” when choosing antibody-based treatment, he said. Blinatumomab is a good choice for patients with a lower tumor burden – either patients with early relapse or with minimal residual disease – while inotuzumab works better for patients with more bulky disease, as well as those who are not able to accommodate the logistic demands of blinatumomab infusions.
Dr. DeAngelo also highlighted several trials now underway that are testing the efficacy of both antibodies when used as part of first-line treatment.
Dr. DeAngelo has been a consultant to Amgen, the company that markets blinatumomab (Blincyto); to Pfizer, the company developing inotuzumab; and to Ariad, InCyte, and Novartis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
NEW YORK – A pair of new monoclonal antibodies have dramatically changed treatment for patients with acute lymphoblastic leukemia to prepare them for a stem cell transplant, Daniel J. DeAngelo, MD, said at a conference held by Imedex.
“We don’t use standard chemotherapy for reinduction anymore; we use blinatumomab or inotuzumab,” said Dr. DeAngelo, a hematologist oncologist at Dana-Farber Cancer Institute in Boston.
Blinatumomab (Blincyto), approved by the Food and Drug Administration in 2014, has produced “exceptional” response rates, becoming “standard of care” for patients with relapsed acute lymphoblastic leukemia (ALL) that does not have a Philadelphia chromosome, Dr. DeAngelo said in a video interview.
Approved based on results from a phase II study, blinatumomab’s efficacy and safety were recently further delineated in results from the first phase III trial (N Engl J Med. 2017 Mar 2;376[9]:836-74), with 376 treated patients. In that trial, blinatumomab more than doubled the complete remission rate, compared with control patients (34% vs. 16%), and nearly doubled median overall survival – 7.7 months with blinatumomab, compared with 4.0 months for control patients treated with standard chemotherapy.
These findings “further substantiated” blinatumomab’s role, he said.
Blinatumomab’s big limitations are certain adverse effects and the logistics of its dosing. The major adverse effect is “cytokine release syndrome,” which manifests as fever, low blood pressure, and neurologic toxicities that can range from tremors to encephalopathy and seizure. These are manageable by close observation of patients by experienced nurses, Dr. DeAngelo said.
Dosing involves 4 weeks of continuous infusion, starting with 10 days done entirely in the hospital, with the remaining 18 days with patients going home but needing to return every 48 hours to have their infusion bag changed. “Depending on how far the patient lives from the clinic, it can be a logistical challenge,” he said.
A second new antibody he has used on many patients is inotuzumab, which was accepted for review for approval by the FDA in February 2017, with action expected by August.
Dr. DeAngelo served as a coinvestigator in a phase III trial reported in 2016 with 218 evaluable patients. In that trial, investigators reported an 81% complete remission rate with inotuzumab treatment, compared with a 29% among control patients on chemotherapy (N Engl J Med. 2016 Aug 25;375[8]:740-53).
Inotuzumab was effective against patients with Philadelphia chromosome positive ALL, but it will not work for the roughly 5%-10% of ALL patients who lack CD-22 expression in their B-cell ALL.
Inotuzumab is easier to administer than blinatumomab, requiring a once a week infusion, and causes little immediate toxicity – although thrombocytopenia and liver-function abnormalities can occur with continued use, and the risk of veno-occlusive disease is increased when patients later receive a stem cell transplant, Dr. DeAngelo said.
“It’s nice to have options” when choosing antibody-based treatment, he said. Blinatumomab is a good choice for patients with a lower tumor burden – either patients with early relapse or with minimal residual disease – while inotuzumab works better for patients with more bulky disease, as well as those who are not able to accommodate the logistic demands of blinatumomab infusions.
Dr. DeAngelo also highlighted several trials now underway that are testing the efficacy of both antibodies when used as part of first-line treatment.
Dr. DeAngelo has been a consultant to Amgen, the company that markets blinatumomab (Blincyto); to Pfizer, the company developing inotuzumab; and to Ariad, InCyte, and Novartis.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM A MEETING ON HEMATOLOGIC MALIGNANCIES
VIDEO: Careful TKI hiatus makes CML pregnancy possible
NEW YORK – The success that tyrosine kinase inhibitors have had in prolonging life and producing deep hematologic and molecular remissions in patients with chronic myeloid leukemia has led to an unexpected bonus for young women living with the disease: an opportunity to safely become pregnant and mother a child.
The approach is not yet routine and poses a level of risk to both the mother and fetus, especially because tyrosine kinase inhibitors (TKIs) are teratogenic. But with careful planning, close gestational monitoring, and with support from skilled obstetricians, the scenario of a successful pregnancy in women with chronic myeloid leukemia (CML) has now played out several dozen times at a handful of U.S. centers, Mrinal S. Patnaik, MD, said in a talk at the conference held by Imedex.
“We make it clear that this is experimental and is associated with risk, and we share the data [from case reports]; but if the woman wants to go forward,” a protocol now exists “to successfully get them to pregnancy,” said Dr. Patnaik, a hematologist oncologist at the Mayo Clinic in Rochester, Minn.
At Mayo alone, upwards of 20 women with CML have been successfully shepherded through pregnancy, he said in a video interview.
The prospect for a planned pregnancy is reserved for women with their CML well controlled for at least 2 years using a TKI, most often imatinib (Gleevec). In addition to being under complete hematologic control, the candidate patient must also show a deep molecular response, which means a blood level of the BRC-ABL tyrosine kinase that drives CML at least 4 or 4.5 logs (10,000-50,000-fold) below pretreatment levels or molecularly undetectable.
The patient then monitors her ovulatory cycle and stops her medication at the time of ovulation, attempts conception, and then monitors whether pregnancy has actually started. If it has, she needs to stay off her TKI regimen through at least the first 18 weeks of gestation, although an even longer drug holiday is preferred. If not, she resumes the medication and repeats the process later if she wants.
Once the women is pregnant and remains off her TKI regimen Dr. Patnaik and his associates closely follow the woman for signs of a molecular or hematologic relapse, although the latter are unusual. If a resurgence of CML stem cells occurs, the woman receives treatment with pegylated interferon-alpha, which is safe during pregnancy. When possible, TKI treatment remains on hold into the breast-feeding period.
During pregnancy and delivery, the patient requires careful and regular follow-up by a maternal-fetal medicine specialist and has an ongoing risk for high platelet counts causing placental blood clots, fetuses that are small for gestational age, preterm labor, premature rupture of membranes, and other complications.
“These are manageable with good obstetrical care,” Dr. Patnaik said. “We have developed a good system to work out the obstetrical complications.
“By and large we can be successful, but it requires a lot of monitoring and a lot of patient compliance with regular follow-ups,” he stressed.
In a video interview at the meeting, Dr. Patnaik discussed the approach he takes with his patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
NEW YORK – The success that tyrosine kinase inhibitors have had in prolonging life and producing deep hematologic and molecular remissions in patients with chronic myeloid leukemia has led to an unexpected bonus for young women living with the disease: an opportunity to safely become pregnant and mother a child.
The approach is not yet routine and poses a level of risk to both the mother and fetus, especially because tyrosine kinase inhibitors (TKIs) are teratogenic. But with careful planning, close gestational monitoring, and with support from skilled obstetricians, the scenario of a successful pregnancy in women with chronic myeloid leukemia (CML) has now played out several dozen times at a handful of U.S. centers, Mrinal S. Patnaik, MD, said in a talk at the conference held by Imedex.
“We make it clear that this is experimental and is associated with risk, and we share the data [from case reports]; but if the woman wants to go forward,” a protocol now exists “to successfully get them to pregnancy,” said Dr. Patnaik, a hematologist oncologist at the Mayo Clinic in Rochester, Minn.
At Mayo alone, upwards of 20 women with CML have been successfully shepherded through pregnancy, he said in a video interview.
The prospect for a planned pregnancy is reserved for women with their CML well controlled for at least 2 years using a TKI, most often imatinib (Gleevec). In addition to being under complete hematologic control, the candidate patient must also show a deep molecular response, which means a blood level of the BRC-ABL tyrosine kinase that drives CML at least 4 or 4.5 logs (10,000-50,000-fold) below pretreatment levels or molecularly undetectable.
The patient then monitors her ovulatory cycle and stops her medication at the time of ovulation, attempts conception, and then monitors whether pregnancy has actually started. If it has, she needs to stay off her TKI regimen through at least the first 18 weeks of gestation, although an even longer drug holiday is preferred. If not, she resumes the medication and repeats the process later if she wants.
Once the women is pregnant and remains off her TKI regimen Dr. Patnaik and his associates closely follow the woman for signs of a molecular or hematologic relapse, although the latter are unusual. If a resurgence of CML stem cells occurs, the woman receives treatment with pegylated interferon-alpha, which is safe during pregnancy. When possible, TKI treatment remains on hold into the breast-feeding period.
During pregnancy and delivery, the patient requires careful and regular follow-up by a maternal-fetal medicine specialist and has an ongoing risk for high platelet counts causing placental blood clots, fetuses that are small for gestational age, preterm labor, premature rupture of membranes, and other complications.
“These are manageable with good obstetrical care,” Dr. Patnaik said. “We have developed a good system to work out the obstetrical complications.
“By and large we can be successful, but it requires a lot of monitoring and a lot of patient compliance with regular follow-ups,” he stressed.
In a video interview at the meeting, Dr. Patnaik discussed the approach he takes with his patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
NEW YORK – The success that tyrosine kinase inhibitors have had in prolonging life and producing deep hematologic and molecular remissions in patients with chronic myeloid leukemia has led to an unexpected bonus for young women living with the disease: an opportunity to safely become pregnant and mother a child.
The approach is not yet routine and poses a level of risk to both the mother and fetus, especially because tyrosine kinase inhibitors (TKIs) are teratogenic. But with careful planning, close gestational monitoring, and with support from skilled obstetricians, the scenario of a successful pregnancy in women with chronic myeloid leukemia (CML) has now played out several dozen times at a handful of U.S. centers, Mrinal S. Patnaik, MD, said in a talk at the conference held by Imedex.
“We make it clear that this is experimental and is associated with risk, and we share the data [from case reports]; but if the woman wants to go forward,” a protocol now exists “to successfully get them to pregnancy,” said Dr. Patnaik, a hematologist oncologist at the Mayo Clinic in Rochester, Minn.
At Mayo alone, upwards of 20 women with CML have been successfully shepherded through pregnancy, he said in a video interview.
The prospect for a planned pregnancy is reserved for women with their CML well controlled for at least 2 years using a TKI, most often imatinib (Gleevec). In addition to being under complete hematologic control, the candidate patient must also show a deep molecular response, which means a blood level of the BRC-ABL tyrosine kinase that drives CML at least 4 or 4.5 logs (10,000-50,000-fold) below pretreatment levels or molecularly undetectable.
The patient then monitors her ovulatory cycle and stops her medication at the time of ovulation, attempts conception, and then monitors whether pregnancy has actually started. If it has, she needs to stay off her TKI regimen through at least the first 18 weeks of gestation, although an even longer drug holiday is preferred. If not, she resumes the medication and repeats the process later if she wants.
Once the women is pregnant and remains off her TKI regimen Dr. Patnaik and his associates closely follow the woman for signs of a molecular or hematologic relapse, although the latter are unusual. If a resurgence of CML stem cells occurs, the woman receives treatment with pegylated interferon-alpha, which is safe during pregnancy. When possible, TKI treatment remains on hold into the breast-feeding period.
During pregnancy and delivery, the patient requires careful and regular follow-up by a maternal-fetal medicine specialist and has an ongoing risk for high platelet counts causing placental blood clots, fetuses that are small for gestational age, preterm labor, premature rupture of membranes, and other complications.
“These are manageable with good obstetrical care,” Dr. Patnaik said. “We have developed a good system to work out the obstetrical complications.
“By and large we can be successful, but it requires a lot of monitoring and a lot of patient compliance with regular follow-ups,” he stressed.
In a video interview at the meeting, Dr. Patnaik discussed the approach he takes with his patients.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
mzoler@frontlinemedcom.com
On Twitter @mitchelzoler
EXPERT ANALYSIS FROM A MEETING ON HEMATOLOGIC MALIGNANCIES
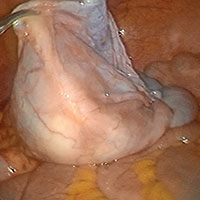
Advanced techniques in cystectomy for mature cystic teratomas

Visit the Society of Gynecologic Surgeons online: sgsonline.org
More videos from SGS:
- Novel classification of labial anatomy and evaluation in the treatment of labial agglutination
- Strategies for prophylactic oophoropexy
- Tips and tricks for open laparoscopy
- Complete colpectomy & colpocleisis: Model for simulation
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist

Visit the Society of Gynecologic Surgeons online: sgsonline.org
More videos from SGS:
- Novel classification of labial anatomy and evaluation in the treatment of labial agglutination
- Strategies for prophylactic oophoropexy
- Tips and tricks for open laparoscopy
- Complete colpectomy & colpocleisis: Model for simulation
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist

Visit the Society of Gynecologic Surgeons online: sgsonline.org
More videos from SGS:
- Novel classification of labial anatomy and evaluation in the treatment of labial agglutination
- Strategies for prophylactic oophoropexy
- Tips and tricks for open laparoscopy
- Complete colpectomy & colpocleisis: Model for simulation
- Natural orifice sacral colpopexy
- Alternative options for visualizing ureteral patency during intraoperative cystoscopy
- Use of suprapubic Carter-Thomason needle to assist in cystoscopic excision of an intravesical foreign object
- Uterine artery ligation: Advanced techniques and considerations for the difficult laparoscopic hysterectomy
- Cervical injection of methylene blue for identification of sentinel lymph nodes in cervical cancer
- Misplaced hysteroscopic sterilization micro-insert in the peritoneal cavity: A corpus alienum
- Laparoscopic cystectomy for large, bilateral ovarian dermoids
- Small bowel surgery for the benign gynecologist
This video is brought to you by
VIDEO: Compassionate care, decriminalization crucial to mitigating addiction epidemic
SAN DIEGO – Health care providers need to practice compassionate care to achieve the best results when managing patients who are dealing with opioid addiction, according to a panel of experts who spoke at the annual meeting of the American College of Physicians.
Caring “compassionately is not enabling, [it’s] doing the right thing by the patient,” explained Chwen-Yuen Angie Chen, MD, of Stanford (Calif.) University. “You can practice compassionate care if you have a knowledge base. Knowledge is extremely powerful and enables you to follow evidence-based medicine, which is truly compassionate care.”
Dr. Chen spoke at length about addiction medicine during a press conference outlining the ACP’s new position paper on preventing and treating substance abuse, where she was joined by ACP President Nitin S. Damle, MD, and ACP Board of Regents Chair Thomas G. Tape, MD. All three emphasized the need for decriminalization and destigmatization of opioid abuse, and they called on physicians to guide patients through resources and compassionate care to help them overcome the affliction.
“We know that we need to either taper, detoxify, or reduce opioid dosing. We know that we ought not to coprescribe with sedatives. We know that, if you need addiction treatment, you get referred, and you don’t just get cut off,” explained Dr. Chen.
In a video interview, Dr. Chen talked about the key take-home messages of the position paper, and she explained other aspects of substance abuse that requires provider’s awareness.
Dr. Chen did not report any relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN DIEGO – Health care providers need to practice compassionate care to achieve the best results when managing patients who are dealing with opioid addiction, according to a panel of experts who spoke at the annual meeting of the American College of Physicians.
Caring “compassionately is not enabling, [it’s] doing the right thing by the patient,” explained Chwen-Yuen Angie Chen, MD, of Stanford (Calif.) University. “You can practice compassionate care if you have a knowledge base. Knowledge is extremely powerful and enables you to follow evidence-based medicine, which is truly compassionate care.”
Dr. Chen spoke at length about addiction medicine during a press conference outlining the ACP’s new position paper on preventing and treating substance abuse, where she was joined by ACP President Nitin S. Damle, MD, and ACP Board of Regents Chair Thomas G. Tape, MD. All three emphasized the need for decriminalization and destigmatization of opioid abuse, and they called on physicians to guide patients through resources and compassionate care to help them overcome the affliction.
“We know that we need to either taper, detoxify, or reduce opioid dosing. We know that we ought not to coprescribe with sedatives. We know that, if you need addiction treatment, you get referred, and you don’t just get cut off,” explained Dr. Chen.
In a video interview, Dr. Chen talked about the key take-home messages of the position paper, and she explained other aspects of substance abuse that requires provider’s awareness.
Dr. Chen did not report any relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
SAN DIEGO – Health care providers need to practice compassionate care to achieve the best results when managing patients who are dealing with opioid addiction, according to a panel of experts who spoke at the annual meeting of the American College of Physicians.
Caring “compassionately is not enabling, [it’s] doing the right thing by the patient,” explained Chwen-Yuen Angie Chen, MD, of Stanford (Calif.) University. “You can practice compassionate care if you have a knowledge base. Knowledge is extremely powerful and enables you to follow evidence-based medicine, which is truly compassionate care.”
Dr. Chen spoke at length about addiction medicine during a press conference outlining the ACP’s new position paper on preventing and treating substance abuse, where she was joined by ACP President Nitin S. Damle, MD, and ACP Board of Regents Chair Thomas G. Tape, MD. All three emphasized the need for decriminalization and destigmatization of opioid abuse, and they called on physicians to guide patients through resources and compassionate care to help them overcome the affliction.
“We know that we need to either taper, detoxify, or reduce opioid dosing. We know that we ought not to coprescribe with sedatives. We know that, if you need addiction treatment, you get referred, and you don’t just get cut off,” explained Dr. Chen.
In a video interview, Dr. Chen talked about the key take-home messages of the position paper, and she explained other aspects of substance abuse that requires provider’s awareness.
Dr. Chen did not report any relevant financial disclosures.
The video associated with this article is no longer available on this site. Please view all of our videos on the MDedge YouTube channel
AT ACP INTERNAL MEDICINE