User login
Seek safe strategies to diagnose gestational diabetes during pandemic
Clinicians and pregnant women are less likely to prescribe and undergo the oral glucose tolerance test (OGTT) to diagnose gestational diabetes in the context of the COVID-19 pandemic, according to a review by H. David McIntyre, MD, of the University of Queensland, Brisbane, Australia, and Robert G. Moses, MD, of Wollongong (Australia) Hospital.
National and international discussions of whether a one- or two-step test for gestational diabetes mellitus (GDM) is optimal, and which women should be tested are ongoing, but the potential for exposure risks to COVID-19 are impacting the test process, they wrote in a commentary published in Diabetes Care.
“Any national or local guidelines should be developed with the primary aim of being protective for pregnant women and workable in the current health crisis,” they wrote.
Key concerns expressed by women and health care providers include the need for travel to be tested, the possible need for two visits, and the several hours spent in a potentially high-risk specimen collection center.
“Further, a GDM diagnosis generally involves additional health service visits for diabetes education, glucose monitoring review, and fetal ultrasonography, all of which carry exposure risks during a pandemic,” Dr. McIntyre and Dr. Moses noted.
Professional societies in the United Kingdom, Canada, and Australia have issued guidance to clinicians for modifying GDM diagnoses criteria during the pandemic that aim to reduce the need for the oral glucose tolerance test both during and after pregnancy.
Pandemic guidelines for all three of these countries support the identification of GDM using early pregnancy hemoglobin A1c (HbA1c) of at least 41 mmol/mol (5.9%).
Then, professionals in the United Kingdom recommend testing based on risk factors and diagnosing GDM based on any of these criteria: HbA1c of at least 39 mmol/mol (5.7%), fasting venous plasma glucose of at least 5.6 mmol/L (preferred), or random VPG of at least 9.0 mmol/L.
The revised testing pathway for Canada accepts an HbA1c of at least 39 mmol/mol (5.7%) and/or random VPG of at least 11.1 mmol/L.
“The revised Australian pathway does not include HbA1c but recommends a fasting VPG with progression to OGTT only if this result is 4.7-5.0 mmol/L,” Dr. McIntyre and Dr. Moses explained.
Overall, the revised guidelines for GDM testing will likely miss some women and only identify those with higher levels of hyperglycemia, the authors wrote. In addition, “the evidence base for these revised pathways is limited and that each alternative strategy should be evaluated over the course of the current pandemic.”
Validation of new testing strategies are needed, and the pandemic may provide and opportunity to adopt an alternative to the OGTT. The World Health Organization has not issued revised guidance for other methods of testing, but fasting VPG alone may be the simplest and most cost effective, at least for the short term, they noted.
“In this ‘new COVID world,’ GDM should not be ignored but pragmatically merits a lower priority than the avoidance of exposure to the COVID-19 virus,” although no single alternative strategy applies in all countries and situations, the authors concluded. Pragmatic measures and documentation of outcomes at the local level will offer the “least worst” solution while the pandemic continues.
The authors had no relevant financial disclosures.
SOURCE: McIntyre HD, Moses RG. Diabetes Care. 2020 May. doi: 10.2337/dci20-0026.
A major concern against the backdrop of COVID-19 is ensuring long-term health while urgent care is – understandably so – being prioritized over preventive care. We can already see the impact that the decrease in primary care has had: Rates of childhood vaccination appear to have dropped; the cancellation or indefinite delay of elective medical procedures has meant a reduction in preventive cancer screenings, such as colonoscopies and mammograms; and concerns about COVID-19 may be keeping those experiencing cardiac events from seeking emergency care.
However, an outcropping of the coronavirus pandemic is an ingenuity to adapt to our new “normal.” Medical licenses have been recognized across state lines to allow much-needed professionals to practice in the hardest-hit areas. Doctors retrofitted a sleep apnea machine to be used as a makeshift ventilator. Those in the wearable device market now have a greater onus to deliver on quality, utility, security, and accuracy.
Obstetricians have had to dramatically change delivery of ante-, intra- and postpartum care. The recent commentary by Dr. McIntyre and Dr. Moses focuses on one particular area of concern: screening, diagnosis, and management of gestational diabetes mellitus (GDM).
Screening and diagnosis are mainstays to reduce the adverse maternal and neonatal outcomes of diabetes in pregnancy. Although there is no universally accepted approach to evaluating GDM, all current methods utilize an oral glucose tolerance test (OGTT), which requires significant time spent in a clinical office setting, thus increasing risk for COVID-19 exposure.
Several countries have adopted modified GDM criteria within the last months. At the time of this writing, the United States has not. Although not testing women for GDM, which is what Dr. McIntyre and Dr. Moses point out may be happening in countries with modified guidelines, seems questionable, perhaps we should think differently about our approach.
More than 20 years ago, it was reported that jelly beans could be used as an alternative to the 50-g GDM screening test (Am J Obstet Gynecol. 1999 Nov;181[5 Pt 1]:1154‐7; Am J Obstet Gynecol. 1995 Dec;173[6]:1889‐92); more recently, candy twists were used with similar results (Am J Obstet Gynecol. 2015 Apr;212[4]:522.e1-5). In addition, a number of articles have reported on the utility of capillary whole blood glucose measurements to screen for GDM in developing and resource-limited countries (Diabetes Technol Ther. 2011;13[5]:586‐91; Acta Diabetol. 2016 Feb;53[1]:91‐7; Diabetes Technol Ther. 2012 Feb;14[2]:131-4). Therefore, rather than forgo GDM screening, women could self-administer a jelly bean test at home, measure blood sugar with a glucometer, and depending on the results, have an OGTT. Importantly, this would allow ob.gyns. to maintain medical standards while managing patients via telemedicine.
We have evidence that GDM can establish poor health for generations. We know that people with underlying conditions have greater morbidity and mortality from infectious diseases. We recognize that accurate screening and diagnosis is the key to prevention and management. Rather than accept a “least worst” scenario, as Dr. McIntyre and Dr. Moses state, we must find ways to provide the best possible care under the current circumstances.
E. Albert Reece, MD, PhD, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the University of Maryland School of Medicine. He said he had no relevant financial disclosures. He is a member of the Ob.Gyn. News editorial advisory board.
A major concern against the backdrop of COVID-19 is ensuring long-term health while urgent care is – understandably so – being prioritized over preventive care. We can already see the impact that the decrease in primary care has had: Rates of childhood vaccination appear to have dropped; the cancellation or indefinite delay of elective medical procedures has meant a reduction in preventive cancer screenings, such as colonoscopies and mammograms; and concerns about COVID-19 may be keeping those experiencing cardiac events from seeking emergency care.
However, an outcropping of the coronavirus pandemic is an ingenuity to adapt to our new “normal.” Medical licenses have been recognized across state lines to allow much-needed professionals to practice in the hardest-hit areas. Doctors retrofitted a sleep apnea machine to be used as a makeshift ventilator. Those in the wearable device market now have a greater onus to deliver on quality, utility, security, and accuracy.
Obstetricians have had to dramatically change delivery of ante-, intra- and postpartum care. The recent commentary by Dr. McIntyre and Dr. Moses focuses on one particular area of concern: screening, diagnosis, and management of gestational diabetes mellitus (GDM).
Screening and diagnosis are mainstays to reduce the adverse maternal and neonatal outcomes of diabetes in pregnancy. Although there is no universally accepted approach to evaluating GDM, all current methods utilize an oral glucose tolerance test (OGTT), which requires significant time spent in a clinical office setting, thus increasing risk for COVID-19 exposure.
Several countries have adopted modified GDM criteria within the last months. At the time of this writing, the United States has not. Although not testing women for GDM, which is what Dr. McIntyre and Dr. Moses point out may be happening in countries with modified guidelines, seems questionable, perhaps we should think differently about our approach.
More than 20 years ago, it was reported that jelly beans could be used as an alternative to the 50-g GDM screening test (Am J Obstet Gynecol. 1999 Nov;181[5 Pt 1]:1154‐7; Am J Obstet Gynecol. 1995 Dec;173[6]:1889‐92); more recently, candy twists were used with similar results (Am J Obstet Gynecol. 2015 Apr;212[4]:522.e1-5). In addition, a number of articles have reported on the utility of capillary whole blood glucose measurements to screen for GDM in developing and resource-limited countries (Diabetes Technol Ther. 2011;13[5]:586‐91; Acta Diabetol. 2016 Feb;53[1]:91‐7; Diabetes Technol Ther. 2012 Feb;14[2]:131-4). Therefore, rather than forgo GDM screening, women could self-administer a jelly bean test at home, measure blood sugar with a glucometer, and depending on the results, have an OGTT. Importantly, this would allow ob.gyns. to maintain medical standards while managing patients via telemedicine.
We have evidence that GDM can establish poor health for generations. We know that people with underlying conditions have greater morbidity and mortality from infectious diseases. We recognize that accurate screening and diagnosis is the key to prevention and management. Rather than accept a “least worst” scenario, as Dr. McIntyre and Dr. Moses state, we must find ways to provide the best possible care under the current circumstances.
E. Albert Reece, MD, PhD, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the University of Maryland School of Medicine. He said he had no relevant financial disclosures. He is a member of the Ob.Gyn. News editorial advisory board.
A major concern against the backdrop of COVID-19 is ensuring long-term health while urgent care is – understandably so – being prioritized over preventive care. We can already see the impact that the decrease in primary care has had: Rates of childhood vaccination appear to have dropped; the cancellation or indefinite delay of elective medical procedures has meant a reduction in preventive cancer screenings, such as colonoscopies and mammograms; and concerns about COVID-19 may be keeping those experiencing cardiac events from seeking emergency care.
However, an outcropping of the coronavirus pandemic is an ingenuity to adapt to our new “normal.” Medical licenses have been recognized across state lines to allow much-needed professionals to practice in the hardest-hit areas. Doctors retrofitted a sleep apnea machine to be used as a makeshift ventilator. Those in the wearable device market now have a greater onus to deliver on quality, utility, security, and accuracy.
Obstetricians have had to dramatically change delivery of ante-, intra- and postpartum care. The recent commentary by Dr. McIntyre and Dr. Moses focuses on one particular area of concern: screening, diagnosis, and management of gestational diabetes mellitus (GDM).
Screening and diagnosis are mainstays to reduce the adverse maternal and neonatal outcomes of diabetes in pregnancy. Although there is no universally accepted approach to evaluating GDM, all current methods utilize an oral glucose tolerance test (OGTT), which requires significant time spent in a clinical office setting, thus increasing risk for COVID-19 exposure.
Several countries have adopted modified GDM criteria within the last months. At the time of this writing, the United States has not. Although not testing women for GDM, which is what Dr. McIntyre and Dr. Moses point out may be happening in countries with modified guidelines, seems questionable, perhaps we should think differently about our approach.
More than 20 years ago, it was reported that jelly beans could be used as an alternative to the 50-g GDM screening test (Am J Obstet Gynecol. 1999 Nov;181[5 Pt 1]:1154‐7; Am J Obstet Gynecol. 1995 Dec;173[6]:1889‐92); more recently, candy twists were used with similar results (Am J Obstet Gynecol. 2015 Apr;212[4]:522.e1-5). In addition, a number of articles have reported on the utility of capillary whole blood glucose measurements to screen for GDM in developing and resource-limited countries (Diabetes Technol Ther. 2011;13[5]:586‐91; Acta Diabetol. 2016 Feb;53[1]:91‐7; Diabetes Technol Ther. 2012 Feb;14[2]:131-4). Therefore, rather than forgo GDM screening, women could self-administer a jelly bean test at home, measure blood sugar with a glucometer, and depending on the results, have an OGTT. Importantly, this would allow ob.gyns. to maintain medical standards while managing patients via telemedicine.
We have evidence that GDM can establish poor health for generations. We know that people with underlying conditions have greater morbidity and mortality from infectious diseases. We recognize that accurate screening and diagnosis is the key to prevention and management. Rather than accept a “least worst” scenario, as Dr. McIntyre and Dr. Moses state, we must find ways to provide the best possible care under the current circumstances.
E. Albert Reece, MD, PhD, who specializes in maternal-fetal medicine, is executive vice president for medical affairs at the University of Maryland, as well as the John Z. and Akiko K. Bowers Distinguished Professor and dean of the University of Maryland School of Medicine. He said he had no relevant financial disclosures. He is a member of the Ob.Gyn. News editorial advisory board.
Clinicians and pregnant women are less likely to prescribe and undergo the oral glucose tolerance test (OGTT) to diagnose gestational diabetes in the context of the COVID-19 pandemic, according to a review by H. David McIntyre, MD, of the University of Queensland, Brisbane, Australia, and Robert G. Moses, MD, of Wollongong (Australia) Hospital.
National and international discussions of whether a one- or two-step test for gestational diabetes mellitus (GDM) is optimal, and which women should be tested are ongoing, but the potential for exposure risks to COVID-19 are impacting the test process, they wrote in a commentary published in Diabetes Care.
“Any national or local guidelines should be developed with the primary aim of being protective for pregnant women and workable in the current health crisis,” they wrote.
Key concerns expressed by women and health care providers include the need for travel to be tested, the possible need for two visits, and the several hours spent in a potentially high-risk specimen collection center.
“Further, a GDM diagnosis generally involves additional health service visits for diabetes education, glucose monitoring review, and fetal ultrasonography, all of which carry exposure risks during a pandemic,” Dr. McIntyre and Dr. Moses noted.
Professional societies in the United Kingdom, Canada, and Australia have issued guidance to clinicians for modifying GDM diagnoses criteria during the pandemic that aim to reduce the need for the oral glucose tolerance test both during and after pregnancy.
Pandemic guidelines for all three of these countries support the identification of GDM using early pregnancy hemoglobin A1c (HbA1c) of at least 41 mmol/mol (5.9%).
Then, professionals in the United Kingdom recommend testing based on risk factors and diagnosing GDM based on any of these criteria: HbA1c of at least 39 mmol/mol (5.7%), fasting venous plasma glucose of at least 5.6 mmol/L (preferred), or random VPG of at least 9.0 mmol/L.
The revised testing pathway for Canada accepts an HbA1c of at least 39 mmol/mol (5.7%) and/or random VPG of at least 11.1 mmol/L.
“The revised Australian pathway does not include HbA1c but recommends a fasting VPG with progression to OGTT only if this result is 4.7-5.0 mmol/L,” Dr. McIntyre and Dr. Moses explained.
Overall, the revised guidelines for GDM testing will likely miss some women and only identify those with higher levels of hyperglycemia, the authors wrote. In addition, “the evidence base for these revised pathways is limited and that each alternative strategy should be evaluated over the course of the current pandemic.”
Validation of new testing strategies are needed, and the pandemic may provide and opportunity to adopt an alternative to the OGTT. The World Health Organization has not issued revised guidance for other methods of testing, but fasting VPG alone may be the simplest and most cost effective, at least for the short term, they noted.
“In this ‘new COVID world,’ GDM should not be ignored but pragmatically merits a lower priority than the avoidance of exposure to the COVID-19 virus,” although no single alternative strategy applies in all countries and situations, the authors concluded. Pragmatic measures and documentation of outcomes at the local level will offer the “least worst” solution while the pandemic continues.
The authors had no relevant financial disclosures.
SOURCE: McIntyre HD, Moses RG. Diabetes Care. 2020 May. doi: 10.2337/dci20-0026.
Clinicians and pregnant women are less likely to prescribe and undergo the oral glucose tolerance test (OGTT) to diagnose gestational diabetes in the context of the COVID-19 pandemic, according to a review by H. David McIntyre, MD, of the University of Queensland, Brisbane, Australia, and Robert G. Moses, MD, of Wollongong (Australia) Hospital.
National and international discussions of whether a one- or two-step test for gestational diabetes mellitus (GDM) is optimal, and which women should be tested are ongoing, but the potential for exposure risks to COVID-19 are impacting the test process, they wrote in a commentary published in Diabetes Care.
“Any national or local guidelines should be developed with the primary aim of being protective for pregnant women and workable in the current health crisis,” they wrote.
Key concerns expressed by women and health care providers include the need for travel to be tested, the possible need for two visits, and the several hours spent in a potentially high-risk specimen collection center.
“Further, a GDM diagnosis generally involves additional health service visits for diabetes education, glucose monitoring review, and fetal ultrasonography, all of which carry exposure risks during a pandemic,” Dr. McIntyre and Dr. Moses noted.
Professional societies in the United Kingdom, Canada, and Australia have issued guidance to clinicians for modifying GDM diagnoses criteria during the pandemic that aim to reduce the need for the oral glucose tolerance test both during and after pregnancy.
Pandemic guidelines for all three of these countries support the identification of GDM using early pregnancy hemoglobin A1c (HbA1c) of at least 41 mmol/mol (5.9%).
Then, professionals in the United Kingdom recommend testing based on risk factors and diagnosing GDM based on any of these criteria: HbA1c of at least 39 mmol/mol (5.7%), fasting venous plasma glucose of at least 5.6 mmol/L (preferred), or random VPG of at least 9.0 mmol/L.
The revised testing pathway for Canada accepts an HbA1c of at least 39 mmol/mol (5.7%) and/or random VPG of at least 11.1 mmol/L.
“The revised Australian pathway does not include HbA1c but recommends a fasting VPG with progression to OGTT only if this result is 4.7-5.0 mmol/L,” Dr. McIntyre and Dr. Moses explained.
Overall, the revised guidelines for GDM testing will likely miss some women and only identify those with higher levels of hyperglycemia, the authors wrote. In addition, “the evidence base for these revised pathways is limited and that each alternative strategy should be evaluated over the course of the current pandemic.”
Validation of new testing strategies are needed, and the pandemic may provide and opportunity to adopt an alternative to the OGTT. The World Health Organization has not issued revised guidance for other methods of testing, but fasting VPG alone may be the simplest and most cost effective, at least for the short term, they noted.
“In this ‘new COVID world,’ GDM should not be ignored but pragmatically merits a lower priority than the avoidance of exposure to the COVID-19 virus,” although no single alternative strategy applies in all countries and situations, the authors concluded. Pragmatic measures and documentation of outcomes at the local level will offer the “least worst” solution while the pandemic continues.
The authors had no relevant financial disclosures.
SOURCE: McIntyre HD, Moses RG. Diabetes Care. 2020 May. doi: 10.2337/dci20-0026.
FDA approves Phexxi for use as an on-demand contraceptive
Evofem Biosciences expects to release Phexxi – the first nonhormonal, on-demand, vaginal pH regulator contraceptive designed to maintain vaginal pH within the range of 3.5-4.5 – in September 2020 alongside the Phexxi Concierge Experience, a comprehensive patient and health care provider telemedicine support system, according to the company’s press release. The service is designed to provide physicians with on-demand educational support, and to speed and simplify women’s access to Phexxi.
In an open-label multicenter trial, women aged 18-35 with regular menstrual cycles intravaginally administered a 5-gram dose of Phexxi vaginal gel up to 1 hour prior to intercourse; they did so for up to seven cycles. There were 101 pregnancies in 1,183 subjects during 4,769 cycles. The 7-cycle cumulative pregnancy rate was 14% (95% confidence interval: 10.0%, 17.5%).
The most common adverse events associated with Phexxi were vulvovaginal burning sensation, vulvovaginal pruritus, vulvovaginal mycotic infection, urinary tract infection, bacterial vaginosis, vaginal discharge, dysuria, and vulvovaginal pain.
Evofem Biosciences expects to release Phexxi – the first nonhormonal, on-demand, vaginal pH regulator contraceptive designed to maintain vaginal pH within the range of 3.5-4.5 – in September 2020 alongside the Phexxi Concierge Experience, a comprehensive patient and health care provider telemedicine support system, according to the company’s press release. The service is designed to provide physicians with on-demand educational support, and to speed and simplify women’s access to Phexxi.
In an open-label multicenter trial, women aged 18-35 with regular menstrual cycles intravaginally administered a 5-gram dose of Phexxi vaginal gel up to 1 hour prior to intercourse; they did so for up to seven cycles. There were 101 pregnancies in 1,183 subjects during 4,769 cycles. The 7-cycle cumulative pregnancy rate was 14% (95% confidence interval: 10.0%, 17.5%).
The most common adverse events associated with Phexxi were vulvovaginal burning sensation, vulvovaginal pruritus, vulvovaginal mycotic infection, urinary tract infection, bacterial vaginosis, vaginal discharge, dysuria, and vulvovaginal pain.
Evofem Biosciences expects to release Phexxi – the first nonhormonal, on-demand, vaginal pH regulator contraceptive designed to maintain vaginal pH within the range of 3.5-4.5 – in September 2020 alongside the Phexxi Concierge Experience, a comprehensive patient and health care provider telemedicine support system, according to the company’s press release. The service is designed to provide physicians with on-demand educational support, and to speed and simplify women’s access to Phexxi.
In an open-label multicenter trial, women aged 18-35 with regular menstrual cycles intravaginally administered a 5-gram dose of Phexxi vaginal gel up to 1 hour prior to intercourse; they did so for up to seven cycles. There were 101 pregnancies in 1,183 subjects during 4,769 cycles. The 7-cycle cumulative pregnancy rate was 14% (95% confidence interval: 10.0%, 17.5%).
The most common adverse events associated with Phexxi were vulvovaginal burning sensation, vulvovaginal pruritus, vulvovaginal mycotic infection, urinary tract infection, bacterial vaginosis, vaginal discharge, dysuria, and vulvovaginal pain.
Most patients with lichen sclerosus receive appropriate treatment
The claims-based prevalence of 0.05% found in the study is lower than previously reported, and only 16% of the diagnoses were in women aged 18-44 years, Laura E. Melnick, MD, and associates wrote after identifying 10,004 females aged 0-65 years with lichen sclerosus in the IBM MarketScan Commercial Claims and Encounters Databases from 2015 to 2017. The majority (79%) of those diagnosed were aged 45-65 years (average, 50.8 years).
In pediatric patients (up to age 17 years), the low prevalence (0.01%) “may be attributable to several factors including relative rarity, as well as variability in pediatric clinicians’ familiarity with [lichen sclerosus] and in patients’ clinical symptoms,” said Dr. Melnick and associates in the department of dermatology at New York University.
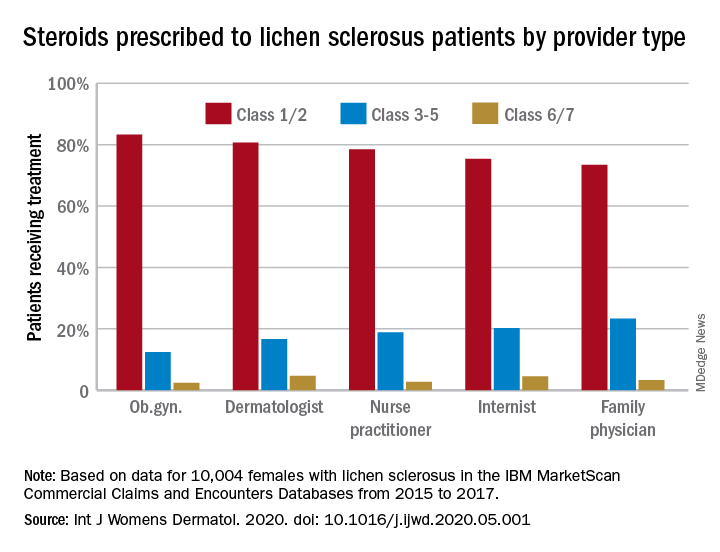
Just over half of all diagnoses (52.4%) were made by ob.gyns., with dermatologists next at 14.5%, followed by family physicians (6.5%), nurse practitioners (2.5%), and internists (0.4%), they reported in the International Journal of Women’s Dermatology.
Treatment for lichen sclerosus, in the form of high-potency topical corticosteroids, was mostly appropriate. Ob.gyns. prescribed class 1/2 steroids to 83% of their patients, tops among all clinicians. Dermatologists were just over 80%, and the other clinician categories were all over 70%, the investigators said.
“Understanding the current management of [lichen sclerosus] is important given that un- or undertreated disease can significantly impact patients’ quality of life, lead to increased lower urinary tract symptoms and irreversible architectural changes, and predispose women to squamous cell carcinoma,” they wrote.
SOURCE: Melnick LE et al. Int J Womens Dermatol. 2020. doi: 10.1016/j.ijwd.2020.05.001.

The claims-based prevalence of 0.05% found in the study is lower than previously reported, and only 16% of the diagnoses were in women aged 18-44 years, Laura E. Melnick, MD, and associates wrote after identifying 10,004 females aged 0-65 years with lichen sclerosus in the IBM MarketScan Commercial Claims and Encounters Databases from 2015 to 2017. The majority (79%) of those diagnosed were aged 45-65 years (average, 50.8 years).
In pediatric patients (up to age 17 years), the low prevalence (0.01%) “may be attributable to several factors including relative rarity, as well as variability in pediatric clinicians’ familiarity with [lichen sclerosus] and in patients’ clinical symptoms,” said Dr. Melnick and associates in the department of dermatology at New York University.
Just over half of all diagnoses (52.4%) were made by ob.gyns., with dermatologists next at 14.5%, followed by family physicians (6.5%), nurse practitioners (2.5%), and internists (0.4%), they reported in the International Journal of Women’s Dermatology.
Treatment for lichen sclerosus, in the form of high-potency topical corticosteroids, was mostly appropriate. Ob.gyns. prescribed class 1/2 steroids to 83% of their patients, tops among all clinicians. Dermatologists were just over 80%, and the other clinician categories were all over 70%, the investigators said.
“Understanding the current management of [lichen sclerosus] is important given that un- or undertreated disease can significantly impact patients’ quality of life, lead to increased lower urinary tract symptoms and irreversible architectural changes, and predispose women to squamous cell carcinoma,” they wrote.
SOURCE: Melnick LE et al. Int J Womens Dermatol. 2020. doi: 10.1016/j.ijwd.2020.05.001.

The claims-based prevalence of 0.05% found in the study is lower than previously reported, and only 16% of the diagnoses were in women aged 18-44 years, Laura E. Melnick, MD, and associates wrote after identifying 10,004 females aged 0-65 years with lichen sclerosus in the IBM MarketScan Commercial Claims and Encounters Databases from 2015 to 2017. The majority (79%) of those diagnosed were aged 45-65 years (average, 50.8 years).
In pediatric patients (up to age 17 years), the low prevalence (0.01%) “may be attributable to several factors including relative rarity, as well as variability in pediatric clinicians’ familiarity with [lichen sclerosus] and in patients’ clinical symptoms,” said Dr. Melnick and associates in the department of dermatology at New York University.
Just over half of all diagnoses (52.4%) were made by ob.gyns., with dermatologists next at 14.5%, followed by family physicians (6.5%), nurse practitioners (2.5%), and internists (0.4%), they reported in the International Journal of Women’s Dermatology.
Treatment for lichen sclerosus, in the form of high-potency topical corticosteroids, was mostly appropriate. Ob.gyns. prescribed class 1/2 steroids to 83% of their patients, tops among all clinicians. Dermatologists were just over 80%, and the other clinician categories were all over 70%, the investigators said.
“Understanding the current management of [lichen sclerosus] is important given that un- or undertreated disease can significantly impact patients’ quality of life, lead to increased lower urinary tract symptoms and irreversible architectural changes, and predispose women to squamous cell carcinoma,” they wrote.
SOURCE: Melnick LE et al. Int J Womens Dermatol. 2020. doi: 10.1016/j.ijwd.2020.05.001.
FROM THE INTERNATIONAL JOURNAL OF WOMEN’S DERMATOLOGY
Today’s top news highlights: ACE inhibitors in COVID patients, fewer AMI admissions, and more
Here are the stories our MDedge editors across specialties think you need to know about today:
Are ACE inhibitors protective in COVID-19?
Older patients with COVID-19 had a lower risk of developing severe illness if they were taking ACE inhibitors, according to a large observational U.S. study. ACE inhibitor use was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. Senior investigator Harlan M. Krumholz, MD, said that while he and his associates think this finding is worthy of further study, “We don’t believe this is enough info to change practice.” The study was published on the MedRxiv preprint server and has not yet been peer reviewed.
READ MORE.
AMI: Admissions drop, deaths rise
In Italy, sharp nationwide decreases in hospitalizations for acute myocardial infarctions (AMIs) during the height of COVID-19 were offset by higher mortality for patients who did present. The study counted AMIs at 54 hospitals nationwide for the week of March 12-19, 2020, and compared that with an equivalent week in 2019 – 319 vs. 618 AMIs, respectively, representing a 48% reduction in hospitalizations. Mortality for ST-segment elevation MI cases more than tripled to 14% during the outbreak, compared with 4% in 2019. “The concern is fewer MIs most likely means people are dying at home or presenting later as this study suggests,” commented Martha Gulati, MD, chief of cardiology at the University of Arizona, Phoenix, who was not involved with the study.
READ MORE.
Prenatal, postpartum screening for depression falls short
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Researchers analyzed self-reported data on postpartum depressive symptoms collected in 2018 by the Pregnancy Risk Assessment Monitoring System. Mental health conditions play a role in approximately 9% of pregnancy-related deaths and not asking about depression represents “missed opportunities to potentially identify and treat women with depression,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Are ACE inhibitors protective in COVID-19?
Older patients with COVID-19 had a lower risk of developing severe illness if they were taking ACE inhibitors, according to a large observational U.S. study. ACE inhibitor use was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. Senior investigator Harlan M. Krumholz, MD, said that while he and his associates think this finding is worthy of further study, “We don’t believe this is enough info to change practice.” The study was published on the MedRxiv preprint server and has not yet been peer reviewed.
READ MORE.
AMI: Admissions drop, deaths rise
In Italy, sharp nationwide decreases in hospitalizations for acute myocardial infarctions (AMIs) during the height of COVID-19 were offset by higher mortality for patients who did present. The study counted AMIs at 54 hospitals nationwide for the week of March 12-19, 2020, and compared that with an equivalent week in 2019 – 319 vs. 618 AMIs, respectively, representing a 48% reduction in hospitalizations. Mortality for ST-segment elevation MI cases more than tripled to 14% during the outbreak, compared with 4% in 2019. “The concern is fewer MIs most likely means people are dying at home or presenting later as this study suggests,” commented Martha Gulati, MD, chief of cardiology at the University of Arizona, Phoenix, who was not involved with the study.
READ MORE.
Prenatal, postpartum screening for depression falls short
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Researchers analyzed self-reported data on postpartum depressive symptoms collected in 2018 by the Pregnancy Risk Assessment Monitoring System. Mental health conditions play a role in approximately 9% of pregnancy-related deaths and not asking about depression represents “missed opportunities to potentially identify and treat women with depression,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Here are the stories our MDedge editors across specialties think you need to know about today:
Are ACE inhibitors protective in COVID-19?
Older patients with COVID-19 had a lower risk of developing severe illness if they were taking ACE inhibitors, according to a large observational U.S. study. ACE inhibitor use was associated with an almost 40% lower risk for COVID-19 hospitalization for older people enrolled in Medicare Advantage plans. Senior investigator Harlan M. Krumholz, MD, said that while he and his associates think this finding is worthy of further study, “We don’t believe this is enough info to change practice.” The study was published on the MedRxiv preprint server and has not yet been peer reviewed.
READ MORE.
AMI: Admissions drop, deaths rise
In Italy, sharp nationwide decreases in hospitalizations for acute myocardial infarctions (AMIs) during the height of COVID-19 were offset by higher mortality for patients who did present. The study counted AMIs at 54 hospitals nationwide for the week of March 12-19, 2020, and compared that with an equivalent week in 2019 – 319 vs. 618 AMIs, respectively, representing a 48% reduction in hospitalizations. Mortality for ST-segment elevation MI cases more than tripled to 14% during the outbreak, compared with 4% in 2019. “The concern is fewer MIs most likely means people are dying at home or presenting later as this study suggests,” commented Martha Gulati, MD, chief of cardiology at the University of Arizona, Phoenix, who was not involved with the study.
READ MORE.
Prenatal, postpartum screening for depression falls short
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Researchers analyzed self-reported data on postpartum depressive symptoms collected in 2018 by the Pregnancy Risk Assessment Monitoring System. Mental health conditions play a role in approximately 9% of pregnancy-related deaths and not asking about depression represents “missed opportunities to potentially identify and treat women with depression,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
For more on COVID-19, visit our Resource Center. All of our latest news is available on MDedge.com.
Many clinicians still not asking about postpartum depression
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Although the prevalence of screening has risen in recent years, many women could be suffering in silence.
“[U]ndetected and untreated perinatal depression can have negative health consequences for the mothers and their babies,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
Dr. Ko and colleagues reported their findings in an article published in Morbidity and Mortality Weekly Report.
The researchers analyzed self-reported data on postpartum depressive symptoms (PDS) collected in 2018 by the Pregnancy Risk Assessment Monitoring System (PRAMS). Participants were stratified on the basis of location and maternal and infant characteristics, including age, race/ethnicity, and education level. Women who had recently given birth to one or more live infants answered questions about whether they had been screened by health care providers for depression during perinatal visits.
The prevalence of PDS among women from 31 PRAMS sites was 13.2%. States with lower prevalences included Illinois (9.7%), Massachusetts (10.3%), and Wisconsin (10.5%); states with higher prevalences included Mississippi (23.5%), West Virginia (19.4%), and Michigan (16.4%).
Some groups were at higher risk for PDS than others. The prevalence was greater than 20% among women who were aged 19 years or younger, were of American Indian or Alaska Native ethnicity, smoked during the perinatal period, experienced perinatal depression, or whose infant died after birth.
Depressive symptoms were also more common among women who received assistance from the Women, Infants, and Children program; were Medicaid beneficiaries at the time of delivery; smoked cigarettes during the last trimester of pregnancy; breastfed their infants for fewer than 8 weeks; or had experienced intimate partner violence while pregnant or before.
Small rise in screening
Overall, 79.1% of women said a health care provider had inquired about depression during the prenatal period. Prenatal screening for depression was lowest in Puerto Rico (50.7%), Mississippi (69.4%), Utah (69.5%), and Kentucky (69.5%) and was highest in Alaska (90.7%), Minnesota (90.6%), and Maine (90.5%).
Among 22 continuously reporting sites, the prevalence of prenatal depression screening rose significantly from 76.2% in 2016 to 79.3% in 2018 (P < .05) .
“It is unclear what might account for this small increase,” Dr. Ko said. “There may be additional factors, such as women may be becoming more comfortable reporting symptoms of depression. With continued awareness about the need to screen every pregnant and postpartum woman for depression, we can expect things to continue to improve.”
Overall, 90.1% of respondents reported a postpartum visit; of those, 87.4% said a health care provider had asked about depression during that visit.
Screening during the postpartum period was highest in Vermont (96.2%), Minnesota (95.9%), and Maine (95.5%) and was lowest in Puerto Rico (50.7%), New York City (73.1%), and Louisiana (75.0%).
Among the 22 sites that reported continuously, the prevalence of screening for postpartum depression rose significantly from 84.1% to 88.0% (P < .05), “with an average annual percentage point increase of 1.8%,” the authors wrote.
‘Missed opportunities’
“PRAMS responses are reported an average of 4 months postpartum, which suggests persistence of [depressive] symptoms,” the authors wrote.
Dr. Ko said that mental health conditions play a role in approximately 9% of pregnancy-related deaths and that not asking about depression represents “missed opportunities to potentially identify and treat women with depression.” The United States Preventive Services Task Force recommends screening all adults for depression, including women during pregnancy and the postpartum period, she added.
When asked what can be done to improve screening that has not already been tried, Dr. Ko said the CDC is currently evaluating a study called the Program in Support of Moms (PRISM), which “is designed to help obstetrics and gynecology practices address the significant public health issue of depression during and after pregnancy. PRISM aims to close gaps in health care delivery to ensure that women with depression during and after pregnancy receive the best treatment, which can result in improvement in their symptoms.”
Dr. Ko added that the Health Resources and Services Administration has funded seven states to begin “programs to support providers to screen, assess, refer, and treat pregnant and postpartum women for depression and other behavioral health conditions. States can use initiatives like Healthy Start, home visiting, and Title V Maternal and Child Health Services Block Grant programs as levers to improve screening and address maternal depression.
“Screening is just one part of addressing perinatal depression. Health care providers need to refer women to appropriate resources in order to get the proper diagnosis, treatment, and follow-up care for management of depression,” Dr. Ko concluded.
The authors disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Although the prevalence of screening has risen in recent years, many women could be suffering in silence.
“[U]ndetected and untreated perinatal depression can have negative health consequences for the mothers and their babies,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
Dr. Ko and colleagues reported their findings in an article published in Morbidity and Mortality Weekly Report.
The researchers analyzed self-reported data on postpartum depressive symptoms (PDS) collected in 2018 by the Pregnancy Risk Assessment Monitoring System (PRAMS). Participants were stratified on the basis of location and maternal and infant characteristics, including age, race/ethnicity, and education level. Women who had recently given birth to one or more live infants answered questions about whether they had been screened by health care providers for depression during perinatal visits.
The prevalence of PDS among women from 31 PRAMS sites was 13.2%. States with lower prevalences included Illinois (9.7%), Massachusetts (10.3%), and Wisconsin (10.5%); states with higher prevalences included Mississippi (23.5%), West Virginia (19.4%), and Michigan (16.4%).
Some groups were at higher risk for PDS than others. The prevalence was greater than 20% among women who were aged 19 years or younger, were of American Indian or Alaska Native ethnicity, smoked during the perinatal period, experienced perinatal depression, or whose infant died after birth.
Depressive symptoms were also more common among women who received assistance from the Women, Infants, and Children program; were Medicaid beneficiaries at the time of delivery; smoked cigarettes during the last trimester of pregnancy; breastfed their infants for fewer than 8 weeks; or had experienced intimate partner violence while pregnant or before.
Small rise in screening
Overall, 79.1% of women said a health care provider had inquired about depression during the prenatal period. Prenatal screening for depression was lowest in Puerto Rico (50.7%), Mississippi (69.4%), Utah (69.5%), and Kentucky (69.5%) and was highest in Alaska (90.7%), Minnesota (90.6%), and Maine (90.5%).
Among 22 continuously reporting sites, the prevalence of prenatal depression screening rose significantly from 76.2% in 2016 to 79.3% in 2018 (P < .05) .
“It is unclear what might account for this small increase,” Dr. Ko said. “There may be additional factors, such as women may be becoming more comfortable reporting symptoms of depression. With continued awareness about the need to screen every pregnant and postpartum woman for depression, we can expect things to continue to improve.”
Overall, 90.1% of respondents reported a postpartum visit; of those, 87.4% said a health care provider had asked about depression during that visit.
Screening during the postpartum period was highest in Vermont (96.2%), Minnesota (95.9%), and Maine (95.5%) and was lowest in Puerto Rico (50.7%), New York City (73.1%), and Louisiana (75.0%).
Among the 22 sites that reported continuously, the prevalence of screening for postpartum depression rose significantly from 84.1% to 88.0% (P < .05), “with an average annual percentage point increase of 1.8%,” the authors wrote.
‘Missed opportunities’
“PRAMS responses are reported an average of 4 months postpartum, which suggests persistence of [depressive] symptoms,” the authors wrote.
Dr. Ko said that mental health conditions play a role in approximately 9% of pregnancy-related deaths and that not asking about depression represents “missed opportunities to potentially identify and treat women with depression.” The United States Preventive Services Task Force recommends screening all adults for depression, including women during pregnancy and the postpartum period, she added.
When asked what can be done to improve screening that has not already been tried, Dr. Ko said the CDC is currently evaluating a study called the Program in Support of Moms (PRISM), which “is designed to help obstetrics and gynecology practices address the significant public health issue of depression during and after pregnancy. PRISM aims to close gaps in health care delivery to ensure that women with depression during and after pregnancy receive the best treatment, which can result in improvement in their symptoms.”
Dr. Ko added that the Health Resources and Services Administration has funded seven states to begin “programs to support providers to screen, assess, refer, and treat pregnant and postpartum women for depression and other behavioral health conditions. States can use initiatives like Healthy Start, home visiting, and Title V Maternal and Child Health Services Block Grant programs as levers to improve screening and address maternal depression.
“Screening is just one part of addressing perinatal depression. Health care providers need to refer women to appropriate resources in order to get the proper diagnosis, treatment, and follow-up care for management of depression,” Dr. Ko concluded.
The authors disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
Health care providers fail to ask one in five prenatal patients and one in eight postpartum patients about depression, according to the Centers for Disease Control and Prevention. Although the prevalence of screening has risen in recent years, many women could be suffering in silence.
“[U]ndetected and untreated perinatal depression can have negative health consequences for the mothers and their babies,” said coauthor Jean Y. Ko, PhD, from the division of reproductive health at the National Center for Chronic Disease Prevention and Health Promotion.
Dr. Ko and colleagues reported their findings in an article published in Morbidity and Mortality Weekly Report.
The researchers analyzed self-reported data on postpartum depressive symptoms (PDS) collected in 2018 by the Pregnancy Risk Assessment Monitoring System (PRAMS). Participants were stratified on the basis of location and maternal and infant characteristics, including age, race/ethnicity, and education level. Women who had recently given birth to one or more live infants answered questions about whether they had been screened by health care providers for depression during perinatal visits.
The prevalence of PDS among women from 31 PRAMS sites was 13.2%. States with lower prevalences included Illinois (9.7%), Massachusetts (10.3%), and Wisconsin (10.5%); states with higher prevalences included Mississippi (23.5%), West Virginia (19.4%), and Michigan (16.4%).
Some groups were at higher risk for PDS than others. The prevalence was greater than 20% among women who were aged 19 years or younger, were of American Indian or Alaska Native ethnicity, smoked during the perinatal period, experienced perinatal depression, or whose infant died after birth.
Depressive symptoms were also more common among women who received assistance from the Women, Infants, and Children program; were Medicaid beneficiaries at the time of delivery; smoked cigarettes during the last trimester of pregnancy; breastfed their infants for fewer than 8 weeks; or had experienced intimate partner violence while pregnant or before.
Small rise in screening
Overall, 79.1% of women said a health care provider had inquired about depression during the prenatal period. Prenatal screening for depression was lowest in Puerto Rico (50.7%), Mississippi (69.4%), Utah (69.5%), and Kentucky (69.5%) and was highest in Alaska (90.7%), Minnesota (90.6%), and Maine (90.5%).
Among 22 continuously reporting sites, the prevalence of prenatal depression screening rose significantly from 76.2% in 2016 to 79.3% in 2018 (P < .05) .
“It is unclear what might account for this small increase,” Dr. Ko said. “There may be additional factors, such as women may be becoming more comfortable reporting symptoms of depression. With continued awareness about the need to screen every pregnant and postpartum woman for depression, we can expect things to continue to improve.”
Overall, 90.1% of respondents reported a postpartum visit; of those, 87.4% said a health care provider had asked about depression during that visit.
Screening during the postpartum period was highest in Vermont (96.2%), Minnesota (95.9%), and Maine (95.5%) and was lowest in Puerto Rico (50.7%), New York City (73.1%), and Louisiana (75.0%).
Among the 22 sites that reported continuously, the prevalence of screening for postpartum depression rose significantly from 84.1% to 88.0% (P < .05), “with an average annual percentage point increase of 1.8%,” the authors wrote.
‘Missed opportunities’
“PRAMS responses are reported an average of 4 months postpartum, which suggests persistence of [depressive] symptoms,” the authors wrote.
Dr. Ko said that mental health conditions play a role in approximately 9% of pregnancy-related deaths and that not asking about depression represents “missed opportunities to potentially identify and treat women with depression.” The United States Preventive Services Task Force recommends screening all adults for depression, including women during pregnancy and the postpartum period, she added.
When asked what can be done to improve screening that has not already been tried, Dr. Ko said the CDC is currently evaluating a study called the Program in Support of Moms (PRISM), which “is designed to help obstetrics and gynecology practices address the significant public health issue of depression during and after pregnancy. PRISM aims to close gaps in health care delivery to ensure that women with depression during and after pregnancy receive the best treatment, which can result in improvement in their symptoms.”
Dr. Ko added that the Health Resources and Services Administration has funded seven states to begin “programs to support providers to screen, assess, refer, and treat pregnant and postpartum women for depression and other behavioral health conditions. States can use initiatives like Healthy Start, home visiting, and Title V Maternal and Child Health Services Block Grant programs as levers to improve screening and address maternal depression.
“Screening is just one part of addressing perinatal depression. Health care providers need to refer women to appropriate resources in order to get the proper diagnosis, treatment, and follow-up care for management of depression,” Dr. Ko concluded.
The authors disclosed no relevant financial relationships.
A version of this article originally appeared on Medscape.com.
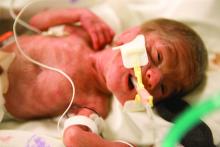
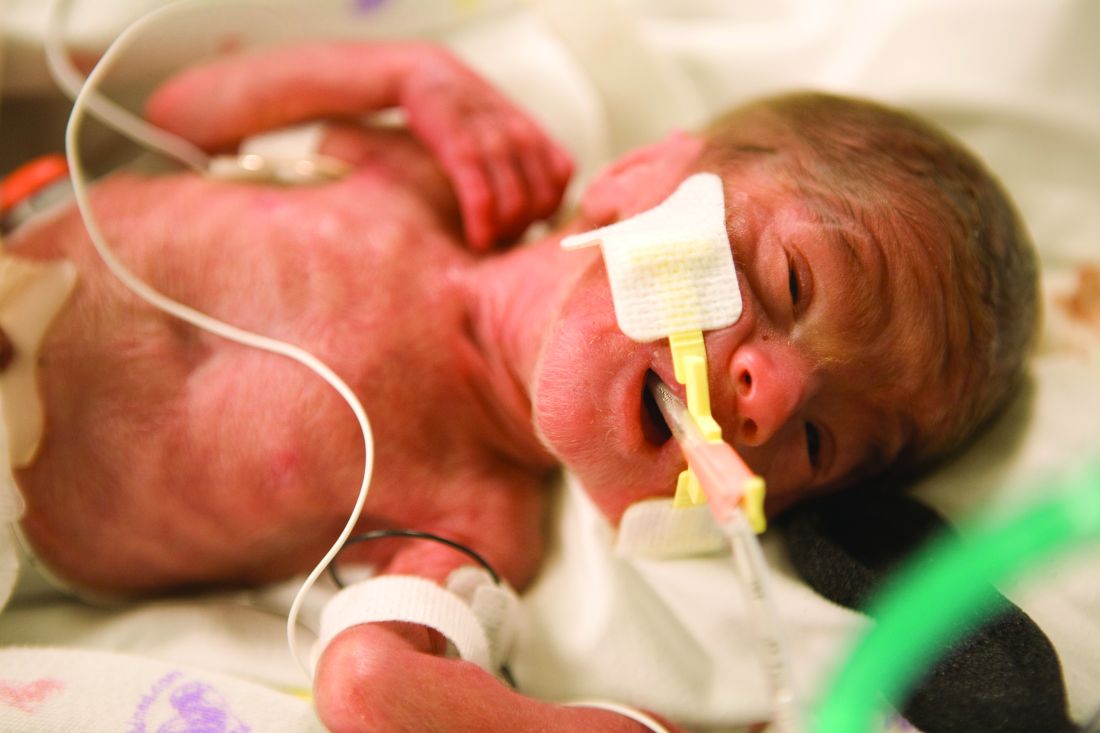
Extremely preterm infants fare better with corticosteroid and magnesium combo
Children born before 27 weeks’ gestation had lower combined risk of death or severe neurodevelopmental impairment when exposed to antenatal corticosteroids and magnesium sulfate together, compared with exposure of either or neither therapy, according to a prospective observational study published in Obstetrics & Gynecology.
“If there is sufficient time to administer antenatal corticosteroids, there should similarly be sufficient time to administer magnesium sulfate,” wrote Samuel J. Gentle, MD, of the University of Alabama at Birmingham, and colleagues. “Given the lower rate of severe neurodevelopmental impairment or death in children exposed to both antenatal corticosteroids and magnesium sulfate in the present study, compared with those exposed to antenatal corticosteroids alone, increasing the rates of magnesium sulfate exposure through quality improvement or other interventions may improve infant outcomes.”
Although previous randomized controlled trials had shown neurologic benefits of each therapy independently in preterm children, few data exist on extremely preterm children, the authors noted. They also pointed out differences in the findings when they analyzed neurodevelopmental outcomes and death separately.
“Whereas exposure to both therapies was associated with a lower rate of death, exposure to magnesium sulfate in addition to antenatal corticosteroids was not associated with a lower rate of severe neurodevelopmental impairment or components of severe neurodevelopmental impairment including Bayley scores, bilateral hearing impairment, and cerebral palsy,” Dr Gentle and his coauthors wrote.
The researchers used prospectively collected data from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network Generic Database to track 3,093 children born extremely preterm – from 22 weeks 0 days to 26 weeks 6 days – during 2011-2014.
The researchers compared outcomes of death or severe neurodevelopmental impairment when the children were 18-26 months of corrected age based on whether they had been exposed to antenatal corticosteroids alone (betamethasone or dexamethasone) or antenatal corticosteroids in addition to magnesium sulfate. Severe neurodevelopmental impairment included “severe cerebral palsy, motor or cognitive composite score less than 70 on the Bayley-III exam, bilateral blindness, or bilateral severe functional hearing impairment with or without amplification.”
The researchers also looked at severe neurodevelopmental impairment and death among children with only magnesium sulfate exposure or with no exposure to steroids or magnesium.
In the study population, 73% of infants had been exposed to both therapies, 16% had been exposed to only corticosteroids, 3% to only magnesium sulfate, and 8% to neither therapy.
“Importantly, a larger proportion of mothers unexposed to either therapy, compared with both therapies, received high school or less education or had no maternal private health insurance which may suggest health inequity as a driver for antenatal therapy exposure rates,” Dr. Gentle and associates noted.
Children whose mothers received corticosteroids and magnesium had a 27% lower risk of severe neurodevelopmental impairment or death, compared with those whose mothers only received corticosteroids (adjusted odds ratio, 0.73). Just over a third of children exposed to both interventions (36%) had severe neurodevelopmental impairment or died, compared with 44% of those exposed only to steroids.
Similarly, corticosteroids and magnesium together were associated with approximately half the risk of death or severe neurodevelopmental impairment, compared with magnesium alone (aOR, 0.49) and 34% lower risk, compared with neither therapy (aOR 0.66).
When the researchers uncoupled the outcomes, severe neurodevelopmental impairment rates were similar among all exposure groups, but rates of death were lower among those who received both therapies than among those who received just one or neither therapy.
“The therapeutic mechanism for neuroprotection in children exposed to magnesium sulfate is unclear but may result from neuronal stabilization or anti-inflammatory properties,” Dr. Gentle and colleagues said.
They also compared rates in the exposure groups of grade 3-4 intracranial hemorrhage, which has been linked to poor neurodevelopmental outcomes in extremely preterm children.
“The rate of grade 3-4 intracranial hemorrhage did not differ between children exposed to both antenatal corticosteroids and magnesium sulfate and those exposed to antenatal corticosteroids alone,” they said. “These findings further support data from randomized controlled trials showing benefit for antenatal corticosteroids but not for magnesium sulfate.”
They further noted a Cochrane Review that found significantly reduced risk of severe or any intracranial hemorrhage among children exposed to antenatal corticosteroids. No similar reduction in intracranial hemorrhage occurred in a separate Cochrane Review of antenatal magnesium sulfate trials.
The research was funded by the National Institutes of Health, Eunice Kennedy Shriver National Institute of Child Health and Human Development, and the National Center for Advancing Translational Sciences. One author is a consultant for Mednax who has received travel funds. Another author disclosed Catholic Health Professionals of Houston paid honorarium for an ethics talk he gave.
SOURCE: Gentle SJ et al. Obstet. Gynecol. 2020. doi: 10.1097/AOG.0000000000003882.
Children born before 27 weeks’ gestation had lower combined risk of death or severe neurodevelopmental impairment when exposed to antenatal corticosteroids and magnesium sulfate together, compared with exposure of either or neither therapy, according to a prospective observational study published in Obstetrics & Gynecology.
“If there is sufficient time to administer antenatal corticosteroids, there should similarly be sufficient time to administer magnesium sulfate,” wrote Samuel J. Gentle, MD, of the University of Alabama at Birmingham, and colleagues. “Given the lower rate of severe neurodevelopmental impairment or death in children exposed to both antenatal corticosteroids and magnesium sulfate in the present study, compared with those exposed to antenatal corticosteroids alone, increasing the rates of magnesium sulfate exposure through quality improvement or other interventions may improve infant outcomes.”
Although previous randomized controlled trials had shown neurologic benefits of each therapy independently in preterm children, few data exist on extremely preterm children, the authors noted. They also pointed out differences in the findings when they analyzed neurodevelopmental outcomes and death separately.
“Whereas exposure to both therapies was associated with a lower rate of death, exposure to magnesium sulfate in addition to antenatal corticosteroids was not associated with a lower rate of severe neurodevelopmental impairment or components of severe neurodevelopmental impairment including Bayley scores, bilateral hearing impairment, and cerebral palsy,” Dr Gentle and his coauthors wrote.
The researchers used prospectively collected data from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network Generic Database to track 3,093 children born extremely preterm – from 22 weeks 0 days to 26 weeks 6 days – during 2011-2014.
The researchers compared outcomes of death or severe neurodevelopmental impairment when the children were 18-26 months of corrected age based on whether they had been exposed to antenatal corticosteroids alone (betamethasone or dexamethasone) or antenatal corticosteroids in addition to magnesium sulfate. Severe neurodevelopmental impairment included “severe cerebral palsy, motor or cognitive composite score less than 70 on the Bayley-III exam, bilateral blindness, or bilateral severe functional hearing impairment with or without amplification.”
The researchers also looked at severe neurodevelopmental impairment and death among children with only magnesium sulfate exposure or with no exposure to steroids or magnesium.
In the study population, 73% of infants had been exposed to both therapies, 16% had been exposed to only corticosteroids, 3% to only magnesium sulfate, and 8% to neither therapy.
“Importantly, a larger proportion of mothers unexposed to either therapy, compared with both therapies, received high school or less education or had no maternal private health insurance which may suggest health inequity as a driver for antenatal therapy exposure rates,” Dr. Gentle and associates noted.
Children whose mothers received corticosteroids and magnesium had a 27% lower risk of severe neurodevelopmental impairment or death, compared with those whose mothers only received corticosteroids (adjusted odds ratio, 0.73). Just over a third of children exposed to both interventions (36%) had severe neurodevelopmental impairment or died, compared with 44% of those exposed only to steroids.
Similarly, corticosteroids and magnesium together were associated with approximately half the risk of death or severe neurodevelopmental impairment, compared with magnesium alone (aOR, 0.49) and 34% lower risk, compared with neither therapy (aOR 0.66).
When the researchers uncoupled the outcomes, severe neurodevelopmental impairment rates were similar among all exposure groups, but rates of death were lower among those who received both therapies than among those who received just one or neither therapy.
“The therapeutic mechanism for neuroprotection in children exposed to magnesium sulfate is unclear but may result from neuronal stabilization or anti-inflammatory properties,” Dr. Gentle and colleagues said.
They also compared rates in the exposure groups of grade 3-4 intracranial hemorrhage, which has been linked to poor neurodevelopmental outcomes in extremely preterm children.
“The rate of grade 3-4 intracranial hemorrhage did not differ between children exposed to both antenatal corticosteroids and magnesium sulfate and those exposed to antenatal corticosteroids alone,” they said. “These findings further support data from randomized controlled trials showing benefit for antenatal corticosteroids but not for magnesium sulfate.”
They further noted a Cochrane Review that found significantly reduced risk of severe or any intracranial hemorrhage among children exposed to antenatal corticosteroids. No similar reduction in intracranial hemorrhage occurred in a separate Cochrane Review of antenatal magnesium sulfate trials.
The research was funded by the National Institutes of Health, Eunice Kennedy Shriver National Institute of Child Health and Human Development, and the National Center for Advancing Translational Sciences. One author is a consultant for Mednax who has received travel funds. Another author disclosed Catholic Health Professionals of Houston paid honorarium for an ethics talk he gave.
SOURCE: Gentle SJ et al. Obstet. Gynecol. 2020. doi: 10.1097/AOG.0000000000003882.
Children born before 27 weeks’ gestation had lower combined risk of death or severe neurodevelopmental impairment when exposed to antenatal corticosteroids and magnesium sulfate together, compared with exposure of either or neither therapy, according to a prospective observational study published in Obstetrics & Gynecology.
“If there is sufficient time to administer antenatal corticosteroids, there should similarly be sufficient time to administer magnesium sulfate,” wrote Samuel J. Gentle, MD, of the University of Alabama at Birmingham, and colleagues. “Given the lower rate of severe neurodevelopmental impairment or death in children exposed to both antenatal corticosteroids and magnesium sulfate in the present study, compared with those exposed to antenatal corticosteroids alone, increasing the rates of magnesium sulfate exposure through quality improvement or other interventions may improve infant outcomes.”
Although previous randomized controlled trials had shown neurologic benefits of each therapy independently in preterm children, few data exist on extremely preterm children, the authors noted. They also pointed out differences in the findings when they analyzed neurodevelopmental outcomes and death separately.
“Whereas exposure to both therapies was associated with a lower rate of death, exposure to magnesium sulfate in addition to antenatal corticosteroids was not associated with a lower rate of severe neurodevelopmental impairment or components of severe neurodevelopmental impairment including Bayley scores, bilateral hearing impairment, and cerebral palsy,” Dr Gentle and his coauthors wrote.
The researchers used prospectively collected data from the Eunice Kennedy Shriver National Institute of Child Health and Human Development Neonatal Research Network Generic Database to track 3,093 children born extremely preterm – from 22 weeks 0 days to 26 weeks 6 days – during 2011-2014.
The researchers compared outcomes of death or severe neurodevelopmental impairment when the children were 18-26 months of corrected age based on whether they had been exposed to antenatal corticosteroids alone (betamethasone or dexamethasone) or antenatal corticosteroids in addition to magnesium sulfate. Severe neurodevelopmental impairment included “severe cerebral palsy, motor or cognitive composite score less than 70 on the Bayley-III exam, bilateral blindness, or bilateral severe functional hearing impairment with or without amplification.”
The researchers also looked at severe neurodevelopmental impairment and death among children with only magnesium sulfate exposure or with no exposure to steroids or magnesium.
In the study population, 73% of infants had been exposed to both therapies, 16% had been exposed to only corticosteroids, 3% to only magnesium sulfate, and 8% to neither therapy.
“Importantly, a larger proportion of mothers unexposed to either therapy, compared with both therapies, received high school or less education or had no maternal private health insurance which may suggest health inequity as a driver for antenatal therapy exposure rates,” Dr. Gentle and associates noted.
Children whose mothers received corticosteroids and magnesium had a 27% lower risk of severe neurodevelopmental impairment or death, compared with those whose mothers only received corticosteroids (adjusted odds ratio, 0.73). Just over a third of children exposed to both interventions (36%) had severe neurodevelopmental impairment or died, compared with 44% of those exposed only to steroids.
Similarly, corticosteroids and magnesium together were associated with approximately half the risk of death or severe neurodevelopmental impairment, compared with magnesium alone (aOR, 0.49) and 34% lower risk, compared with neither therapy (aOR 0.66).
When the researchers uncoupled the outcomes, severe neurodevelopmental impairment rates were similar among all exposure groups, but rates of death were lower among those who received both therapies than among those who received just one or neither therapy.
“The therapeutic mechanism for neuroprotection in children exposed to magnesium sulfate is unclear but may result from neuronal stabilization or anti-inflammatory properties,” Dr. Gentle and colleagues said.
They also compared rates in the exposure groups of grade 3-4 intracranial hemorrhage, which has been linked to poor neurodevelopmental outcomes in extremely preterm children.
“The rate of grade 3-4 intracranial hemorrhage did not differ between children exposed to both antenatal corticosteroids and magnesium sulfate and those exposed to antenatal corticosteroids alone,” they said. “These findings further support data from randomized controlled trials showing benefit for antenatal corticosteroids but not for magnesium sulfate.”
They further noted a Cochrane Review that found significantly reduced risk of severe or any intracranial hemorrhage among children exposed to antenatal corticosteroids. No similar reduction in intracranial hemorrhage occurred in a separate Cochrane Review of antenatal magnesium sulfate trials.
The research was funded by the National Institutes of Health, Eunice Kennedy Shriver National Institute of Child Health and Human Development, and the National Center for Advancing Translational Sciences. One author is a consultant for Mednax who has received travel funds. Another author disclosed Catholic Health Professionals of Houston paid honorarium for an ethics talk he gave.
SOURCE: Gentle SJ et al. Obstet. Gynecol. 2020. doi: 10.1097/AOG.0000000000003882.
FROM OBSTETRICS & GYNECOLOGY
U.S. fertility rates fall to record lows
and birth rates for women under age 30 fell to record lows, according to the National Center for Health Statistics.
To be exact – at least as exact as is possible from these provisional data – there were 3,745,540 births in the United States last year. That’s down about 1% from 2018 and is the lowest number of births since 1985, Brady E. Hamilton, PhD, and associates at the NCHS said in a rapid release report.
As births go, so goes the general fertility rate. A 2% decrease from 2018 to 2019 left the fertility rate at its lowest point ever: 58.2 births per 1,000 women aged 15-44 years, compared with 59.1 per 1,000 in 2018, the investigators said, based on data from the National Vital Statistics System.
The total fertility rate – defined as “the number of births that a hypothetical group of 1,000 women would have over their lifetimes, based on the age-specific birth rate in a given year” – also reached a record low of 1,705 births per 1,000 women last year after falling 1% from 2018, they reported.
The falling birth rates did not include women over age 35. The birth rate among women aged 40-44 increased by 2% from 2018, as it reached 12.0 births per 1,000 in 2019. “The rate for this age group has risen almost continuously since 1985 by an average of 3% per year,” Dr. Hamilton and associates wrote.
The birth rate for women aged 30-34 years, 98.3 per 1,000, was down 1% from 2018 but was still the highest for any age category. Among younger women, rates all dropped to record lows: 16.6 (ages 15-19), 66.6 (ages 20-24), and 93.7 (ages 25-29), they said.
Preterm birth rates, on the other hand, rose for the fifth year in a row. The rate for 2019, 10.23% of all births, represents an increase of 2% over 2018 and is “the highest level reported in more than a decade,” the investigators noted.
and birth rates for women under age 30 fell to record lows, according to the National Center for Health Statistics.
To be exact – at least as exact as is possible from these provisional data – there were 3,745,540 births in the United States last year. That’s down about 1% from 2018 and is the lowest number of births since 1985, Brady E. Hamilton, PhD, and associates at the NCHS said in a rapid release report.
As births go, so goes the general fertility rate. A 2% decrease from 2018 to 2019 left the fertility rate at its lowest point ever: 58.2 births per 1,000 women aged 15-44 years, compared with 59.1 per 1,000 in 2018, the investigators said, based on data from the National Vital Statistics System.
The total fertility rate – defined as “the number of births that a hypothetical group of 1,000 women would have over their lifetimes, based on the age-specific birth rate in a given year” – also reached a record low of 1,705 births per 1,000 women last year after falling 1% from 2018, they reported.
The falling birth rates did not include women over age 35. The birth rate among women aged 40-44 increased by 2% from 2018, as it reached 12.0 births per 1,000 in 2019. “The rate for this age group has risen almost continuously since 1985 by an average of 3% per year,” Dr. Hamilton and associates wrote.
The birth rate for women aged 30-34 years, 98.3 per 1,000, was down 1% from 2018 but was still the highest for any age category. Among younger women, rates all dropped to record lows: 16.6 (ages 15-19), 66.6 (ages 20-24), and 93.7 (ages 25-29), they said.
Preterm birth rates, on the other hand, rose for the fifth year in a row. The rate for 2019, 10.23% of all births, represents an increase of 2% over 2018 and is “the highest level reported in more than a decade,” the investigators noted.
and birth rates for women under age 30 fell to record lows, according to the National Center for Health Statistics.
To be exact – at least as exact as is possible from these provisional data – there were 3,745,540 births in the United States last year. That’s down about 1% from 2018 and is the lowest number of births since 1985, Brady E. Hamilton, PhD, and associates at the NCHS said in a rapid release report.
As births go, so goes the general fertility rate. A 2% decrease from 2018 to 2019 left the fertility rate at its lowest point ever: 58.2 births per 1,000 women aged 15-44 years, compared with 59.1 per 1,000 in 2018, the investigators said, based on data from the National Vital Statistics System.
The total fertility rate – defined as “the number of births that a hypothetical group of 1,000 women would have over their lifetimes, based on the age-specific birth rate in a given year” – also reached a record low of 1,705 births per 1,000 women last year after falling 1% from 2018, they reported.
The falling birth rates did not include women over age 35. The birth rate among women aged 40-44 increased by 2% from 2018, as it reached 12.0 births per 1,000 in 2019. “The rate for this age group has risen almost continuously since 1985 by an average of 3% per year,” Dr. Hamilton and associates wrote.
The birth rate for women aged 30-34 years, 98.3 per 1,000, was down 1% from 2018 but was still the highest for any age category. Among younger women, rates all dropped to record lows: 16.6 (ages 15-19), 66.6 (ages 20-24), and 93.7 (ages 25-29), they said.
Preterm birth rates, on the other hand, rose for the fifth year in a row. The rate for 2019, 10.23% of all births, represents an increase of 2% over 2018 and is “the highest level reported in more than a decade,” the investigators noted.
Pedometer use improves postcesarean mobility for high-risk patients
based on data from a randomized trial of 215 patients.
“Patient immobility after surgery is associated with an increased risk of VTE [venous thromboembolism], whereas adequate mobility offers the benefits of enhanced bowel movement resumption and decreasing hospitalization length,” wrote Hadas Ganer Herman, MD, of Tel Aviv University, and colleagues.
In a study published in Obstetrics & Gynecology, the researchers randomized 108 women to a personalized feedback program using pedometers to promote mobility after cesarean delivery; 107 served as controls. Patient demographics and intrapartum experiences, including age, body mass index, and gestation week at delivery, were similar between the groups, as were postpartum complications and the use of analgesics.
Patients who used the pedometers took significantly more steps, compared with controls (5,918 vs. 4,161, P < .001). In addition, women in the pedometer group reported improved physical and mental postpartum recovery and higher levels of satisfaction with their delivery experience, the researchers noted.
The study findings were limited by several factors including potential selection bias among patients who completed the full follow-up, as well as the effect of preset visits from the research team during the study and lack of blinding of the participants. In addition, data on thromboembolic events after hospital discharge were available only through patient phone calls, the researchers noted.
“Our trial is notable for its novelty in exploring an intervention to improve postcesarean delivery mobility, using an objective means of digital step counters,” and for focusing on high-risk patients of clinical interest, Dr. Herman and associates wrote.
Larger studies are needed to explore interventions to improve mobility after cesarean deliveries, they emphasized. However, “because the integration between technology and medicine has continued to evolve and has successfully been proven for additional patient care issues in obstetrics, the current trial offers a basis for interpretation, with the possible use of low-cost interventions such as smart phone applications in maternity wards and simple digital feedback.”
“VTEs are still among the leading causes of maternal morbidity and mortality with peak incidence in the immediate postpartum period,” Martina L. Badell, MD, of Emory University, Atlanta, said in an interview. “As the age and body mass index of our pregnant patients continues to increase, focused attention to prevent VTEs in high-risk populations is very important.”
Dr. Badell said that pedometers are a feasible strategy “provided there is funding available to pay for and provide them.” Pedometers “don’t cause pain/discomfort and can be easily worn and reused. If the hospital isn’t able to provide them, however, then cost could be a barrier to high-risk women using pedometers in the immediate postpartum period.”
“The take-home message is that wearing a pedometer is a simple, low-risk strategy to encourage increased ambulation in a high-risk postpartum population with good patient satisfaction,” Dr. Badell said. The next step for research in this area “is to determine how many steps during the immediate postpartum period is optimal to reduce not only VTE risk, but potentially other postoperative markers such as pain and infection,” she added. Another research question is whether “focused feedback-based pedometers during the prolonged postpartum period result in improved weight loss.”
The researchers had no relevant financial disclosures. Dr. Badell said she had no relevant financial disclosures.
SOURCE: Herman HG et al. Obstet Gynecol. 2020 May 7. doi: 10.1097/AOG.0000000000003879.
based on data from a randomized trial of 215 patients.
“Patient immobility after surgery is associated with an increased risk of VTE [venous thromboembolism], whereas adequate mobility offers the benefits of enhanced bowel movement resumption and decreasing hospitalization length,” wrote Hadas Ganer Herman, MD, of Tel Aviv University, and colleagues.
In a study published in Obstetrics & Gynecology, the researchers randomized 108 women to a personalized feedback program using pedometers to promote mobility after cesarean delivery; 107 served as controls. Patient demographics and intrapartum experiences, including age, body mass index, and gestation week at delivery, were similar between the groups, as were postpartum complications and the use of analgesics.
Patients who used the pedometers took significantly more steps, compared with controls (5,918 vs. 4,161, P < .001). In addition, women in the pedometer group reported improved physical and mental postpartum recovery and higher levels of satisfaction with their delivery experience, the researchers noted.
The study findings were limited by several factors including potential selection bias among patients who completed the full follow-up, as well as the effect of preset visits from the research team during the study and lack of blinding of the participants. In addition, data on thromboembolic events after hospital discharge were available only through patient phone calls, the researchers noted.
“Our trial is notable for its novelty in exploring an intervention to improve postcesarean delivery mobility, using an objective means of digital step counters,” and for focusing on high-risk patients of clinical interest, Dr. Herman and associates wrote.
Larger studies are needed to explore interventions to improve mobility after cesarean deliveries, they emphasized. However, “because the integration between technology and medicine has continued to evolve and has successfully been proven for additional patient care issues in obstetrics, the current trial offers a basis for interpretation, with the possible use of low-cost interventions such as smart phone applications in maternity wards and simple digital feedback.”
“VTEs are still among the leading causes of maternal morbidity and mortality with peak incidence in the immediate postpartum period,” Martina L. Badell, MD, of Emory University, Atlanta, said in an interview. “As the age and body mass index of our pregnant patients continues to increase, focused attention to prevent VTEs in high-risk populations is very important.”
Dr. Badell said that pedometers are a feasible strategy “provided there is funding available to pay for and provide them.” Pedometers “don’t cause pain/discomfort and can be easily worn and reused. If the hospital isn’t able to provide them, however, then cost could be a barrier to high-risk women using pedometers in the immediate postpartum period.”
“The take-home message is that wearing a pedometer is a simple, low-risk strategy to encourage increased ambulation in a high-risk postpartum population with good patient satisfaction,” Dr. Badell said. The next step for research in this area “is to determine how many steps during the immediate postpartum period is optimal to reduce not only VTE risk, but potentially other postoperative markers such as pain and infection,” she added. Another research question is whether “focused feedback-based pedometers during the prolonged postpartum period result in improved weight loss.”
The researchers had no relevant financial disclosures. Dr. Badell said she had no relevant financial disclosures.
SOURCE: Herman HG et al. Obstet Gynecol. 2020 May 7. doi: 10.1097/AOG.0000000000003879.
based on data from a randomized trial of 215 patients.
“Patient immobility after surgery is associated with an increased risk of VTE [venous thromboembolism], whereas adequate mobility offers the benefits of enhanced bowel movement resumption and decreasing hospitalization length,” wrote Hadas Ganer Herman, MD, of Tel Aviv University, and colleagues.
In a study published in Obstetrics & Gynecology, the researchers randomized 108 women to a personalized feedback program using pedometers to promote mobility after cesarean delivery; 107 served as controls. Patient demographics and intrapartum experiences, including age, body mass index, and gestation week at delivery, were similar between the groups, as were postpartum complications and the use of analgesics.
Patients who used the pedometers took significantly more steps, compared with controls (5,918 vs. 4,161, P < .001). In addition, women in the pedometer group reported improved physical and mental postpartum recovery and higher levels of satisfaction with their delivery experience, the researchers noted.
The study findings were limited by several factors including potential selection bias among patients who completed the full follow-up, as well as the effect of preset visits from the research team during the study and lack of blinding of the participants. In addition, data on thromboembolic events after hospital discharge were available only through patient phone calls, the researchers noted.
“Our trial is notable for its novelty in exploring an intervention to improve postcesarean delivery mobility, using an objective means of digital step counters,” and for focusing on high-risk patients of clinical interest, Dr. Herman and associates wrote.
Larger studies are needed to explore interventions to improve mobility after cesarean deliveries, they emphasized. However, “because the integration between technology and medicine has continued to evolve and has successfully been proven for additional patient care issues in obstetrics, the current trial offers a basis for interpretation, with the possible use of low-cost interventions such as smart phone applications in maternity wards and simple digital feedback.”
“VTEs are still among the leading causes of maternal morbidity and mortality with peak incidence in the immediate postpartum period,” Martina L. Badell, MD, of Emory University, Atlanta, said in an interview. “As the age and body mass index of our pregnant patients continues to increase, focused attention to prevent VTEs in high-risk populations is very important.”
Dr. Badell said that pedometers are a feasible strategy “provided there is funding available to pay for and provide them.” Pedometers “don’t cause pain/discomfort and can be easily worn and reused. If the hospital isn’t able to provide them, however, then cost could be a barrier to high-risk women using pedometers in the immediate postpartum period.”
“The take-home message is that wearing a pedometer is a simple, low-risk strategy to encourage increased ambulation in a high-risk postpartum population with good patient satisfaction,” Dr. Badell said. The next step for research in this area “is to determine how many steps during the immediate postpartum period is optimal to reduce not only VTE risk, but potentially other postoperative markers such as pain and infection,” she added. Another research question is whether “focused feedback-based pedometers during the prolonged postpartum period result in improved weight loss.”
The researchers had no relevant financial disclosures. Dr. Badell said she had no relevant financial disclosures.
SOURCE: Herman HG et al. Obstet Gynecol. 2020 May 7. doi: 10.1097/AOG.0000000000003879.
FROM OBSTETRICS & GYNECOLOGY
BMD preserved with investigational drug for uterine fibroid bleeding
Combination therapy with relugolix, an investigational oral gonadotropin-releasing hormone antagonist, estradiol, and norethindrone acetate effectively preserved bone mineral density (BMD) in two replicate phase 3 studies enrolling women with heavy menstrual bleeding associated with uterine fibroids.
The BMD findings, released ahead of the study’s scheduled presentation at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists, build upon previously reported positive primary endpoint data from the LIBERTY 1 and LIBERTY 2 studies. ACOG canceled the meeting and released abstracts for press coverage.
The developer of the drug, Myovant Sciences, plans to submit a new drug application to the Food and Drug Administration for approval of the single-tablet combination therapy for women with uterine fibroids, according to Albert Liao, the company’s director of corporate communications.
The two multinational LIBERTY studies randomized women who had a monthly menstrual blood loss volume of at least 80 mL in two consecutive cycles (or 160 mL in one cycle) in a 1:1:1 ratio to one of three groups: relugolix combination therapy for 24 weeks (once-daily relugolix 40 mg plus estradiol 1.0 mg plus norethindrone acetate 0.5 mg); relugolix alone (40 mg once daily) for 12 weeks followed by relugolix combination therapy for 12 weeks; or placebo for 24 weeks.
In October 2019 at the American Society for Reproductive Medicine Scientific Congress, investigators reported that 73% of women receiving combination therapy in the LIBERTY 1 trial achieved a menstrual blood loss of less than 80 mL and a 50% or greater reduction from baseline over the last 35 days of treatment, compared with 19% in the placebo group. The mean percent reduction in menstrual blood loss from baseline at week 24 was 84% for combination therapy and 23% for placebo.
Earlier in 2019, Myovant Sciences announced that, in the LIBERTY 2 study, 71% of women receiving combination therapy met the primary endpoints, compared with 15% in the placebo group. The reduction in menstrual blood loss in this study’s combination therapy arm was also 84%, according to a company press release from June 2019.
Each of the two clinical trials enrolled upwards of 380 women.
The new abstract released for press coverage by ACOG and published in Obstetrics & Gynecology reports that women receiving relugolix combination therapy in the LIBERTY 1 and LIBERTY 2 studies had a mean change in lumbar spine BMD of –0.36% and –0.13%, respectively, from baseline to 24 weeks. Percent change in lumbar spine BMD in the delayed combination therapy groups (12 initial weeks of relugolix monotherapy) was –1.82% and –2.12%. In the placebo groups, the change was 0.05% and 0.32%.
Michael R. McClung, MD, who is the lead author of the abstract and was scheduled to present the findings at the ACOG meeting, said in an interview that the slight decreases in lumbar spine BMD with combination therapy were noted largely at week 12 and are “clinically insignificant in my opinion.” BMD by dual-energy x-ray absorptiometry was assessed at weeks 12 and 24.
“There was no further increase [after week 12] and [in some patients] there was even a return to baseline,” said Dr. McClung, of the Oregon Osteoporosis Center in Portland.
The safety and efficacy of longer-term treatment with relugolix combination therapy has been investigated thus far through an open-label extension study that brought the treatment period to 52 weeks. The 1-year data has been positive and will be presented or published soon, said Mr. Liao. In addition, a “second, 52-week randomized withdrawal study has been designed to provide 2-year safety and efficacy data … and to evaluate the need for maintenance therapy.”
It’s important, Dr. McClung said, “for clinicians to be confident that BMD loss is prevented or minimized with longer-term relugolix combination therapy since treatment for uterine fibroids is not a short-term proposition. Given the stability of BMD values between weeks 12 and 24 in the LIBERTY studies, I’d anticipate that we will see stable values with longer-term treatment.”
Dr. McClung disclosed that he has served as a consultant/advisory board member and speaker for Amgen and a consultant/advisory board member for Myovant. Several of his coauthors disclosed employment and ownerships interests in Myovant.
SOURCE: McClung MR et al. Obstet Gynecol. 2020 May. doi: 10.1097/01.AOG.0000662944.34860.b4.
Combination therapy with relugolix, an investigational oral gonadotropin-releasing hormone antagonist, estradiol, and norethindrone acetate effectively preserved bone mineral density (BMD) in two replicate phase 3 studies enrolling women with heavy menstrual bleeding associated with uterine fibroids.
The BMD findings, released ahead of the study’s scheduled presentation at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists, build upon previously reported positive primary endpoint data from the LIBERTY 1 and LIBERTY 2 studies. ACOG canceled the meeting and released abstracts for press coverage.
The developer of the drug, Myovant Sciences, plans to submit a new drug application to the Food and Drug Administration for approval of the single-tablet combination therapy for women with uterine fibroids, according to Albert Liao, the company’s director of corporate communications.
The two multinational LIBERTY studies randomized women who had a monthly menstrual blood loss volume of at least 80 mL in two consecutive cycles (or 160 mL in one cycle) in a 1:1:1 ratio to one of three groups: relugolix combination therapy for 24 weeks (once-daily relugolix 40 mg plus estradiol 1.0 mg plus norethindrone acetate 0.5 mg); relugolix alone (40 mg once daily) for 12 weeks followed by relugolix combination therapy for 12 weeks; or placebo for 24 weeks.
In October 2019 at the American Society for Reproductive Medicine Scientific Congress, investigators reported that 73% of women receiving combination therapy in the LIBERTY 1 trial achieved a menstrual blood loss of less than 80 mL and a 50% or greater reduction from baseline over the last 35 days of treatment, compared with 19% in the placebo group. The mean percent reduction in menstrual blood loss from baseline at week 24 was 84% for combination therapy and 23% for placebo.
Earlier in 2019, Myovant Sciences announced that, in the LIBERTY 2 study, 71% of women receiving combination therapy met the primary endpoints, compared with 15% in the placebo group. The reduction in menstrual blood loss in this study’s combination therapy arm was also 84%, according to a company press release from June 2019.
Each of the two clinical trials enrolled upwards of 380 women.
The new abstract released for press coverage by ACOG and published in Obstetrics & Gynecology reports that women receiving relugolix combination therapy in the LIBERTY 1 and LIBERTY 2 studies had a mean change in lumbar spine BMD of –0.36% and –0.13%, respectively, from baseline to 24 weeks. Percent change in lumbar spine BMD in the delayed combination therapy groups (12 initial weeks of relugolix monotherapy) was –1.82% and –2.12%. In the placebo groups, the change was 0.05% and 0.32%.
Michael R. McClung, MD, who is the lead author of the abstract and was scheduled to present the findings at the ACOG meeting, said in an interview that the slight decreases in lumbar spine BMD with combination therapy were noted largely at week 12 and are “clinically insignificant in my opinion.” BMD by dual-energy x-ray absorptiometry was assessed at weeks 12 and 24.
“There was no further increase [after week 12] and [in some patients] there was even a return to baseline,” said Dr. McClung, of the Oregon Osteoporosis Center in Portland.
The safety and efficacy of longer-term treatment with relugolix combination therapy has been investigated thus far through an open-label extension study that brought the treatment period to 52 weeks. The 1-year data has been positive and will be presented or published soon, said Mr. Liao. In addition, a “second, 52-week randomized withdrawal study has been designed to provide 2-year safety and efficacy data … and to evaluate the need for maintenance therapy.”
It’s important, Dr. McClung said, “for clinicians to be confident that BMD loss is prevented or minimized with longer-term relugolix combination therapy since treatment for uterine fibroids is not a short-term proposition. Given the stability of BMD values between weeks 12 and 24 in the LIBERTY studies, I’d anticipate that we will see stable values with longer-term treatment.”
Dr. McClung disclosed that he has served as a consultant/advisory board member and speaker for Amgen and a consultant/advisory board member for Myovant. Several of his coauthors disclosed employment and ownerships interests in Myovant.
SOURCE: McClung MR et al. Obstet Gynecol. 2020 May. doi: 10.1097/01.AOG.0000662944.34860.b4.
Combination therapy with relugolix, an investigational oral gonadotropin-releasing hormone antagonist, estradiol, and norethindrone acetate effectively preserved bone mineral density (BMD) in two replicate phase 3 studies enrolling women with heavy menstrual bleeding associated with uterine fibroids.
The BMD findings, released ahead of the study’s scheduled presentation at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists, build upon previously reported positive primary endpoint data from the LIBERTY 1 and LIBERTY 2 studies. ACOG canceled the meeting and released abstracts for press coverage.
The developer of the drug, Myovant Sciences, plans to submit a new drug application to the Food and Drug Administration for approval of the single-tablet combination therapy for women with uterine fibroids, according to Albert Liao, the company’s director of corporate communications.
The two multinational LIBERTY studies randomized women who had a monthly menstrual blood loss volume of at least 80 mL in two consecutive cycles (or 160 mL in one cycle) in a 1:1:1 ratio to one of three groups: relugolix combination therapy for 24 weeks (once-daily relugolix 40 mg plus estradiol 1.0 mg plus norethindrone acetate 0.5 mg); relugolix alone (40 mg once daily) for 12 weeks followed by relugolix combination therapy for 12 weeks; or placebo for 24 weeks.
In October 2019 at the American Society for Reproductive Medicine Scientific Congress, investigators reported that 73% of women receiving combination therapy in the LIBERTY 1 trial achieved a menstrual blood loss of less than 80 mL and a 50% or greater reduction from baseline over the last 35 days of treatment, compared with 19% in the placebo group. The mean percent reduction in menstrual blood loss from baseline at week 24 was 84% for combination therapy and 23% for placebo.
Earlier in 2019, Myovant Sciences announced that, in the LIBERTY 2 study, 71% of women receiving combination therapy met the primary endpoints, compared with 15% in the placebo group. The reduction in menstrual blood loss in this study’s combination therapy arm was also 84%, according to a company press release from June 2019.
Each of the two clinical trials enrolled upwards of 380 women.
The new abstract released for press coverage by ACOG and published in Obstetrics & Gynecology reports that women receiving relugolix combination therapy in the LIBERTY 1 and LIBERTY 2 studies had a mean change in lumbar spine BMD of –0.36% and –0.13%, respectively, from baseline to 24 weeks. Percent change in lumbar spine BMD in the delayed combination therapy groups (12 initial weeks of relugolix monotherapy) was –1.82% and –2.12%. In the placebo groups, the change was 0.05% and 0.32%.
Michael R. McClung, MD, who is the lead author of the abstract and was scheduled to present the findings at the ACOG meeting, said in an interview that the slight decreases in lumbar spine BMD with combination therapy were noted largely at week 12 and are “clinically insignificant in my opinion.” BMD by dual-energy x-ray absorptiometry was assessed at weeks 12 and 24.
“There was no further increase [after week 12] and [in some patients] there was even a return to baseline,” said Dr. McClung, of the Oregon Osteoporosis Center in Portland.
The safety and efficacy of longer-term treatment with relugolix combination therapy has been investigated thus far through an open-label extension study that brought the treatment period to 52 weeks. The 1-year data has been positive and will be presented or published soon, said Mr. Liao. In addition, a “second, 52-week randomized withdrawal study has been designed to provide 2-year safety and efficacy data … and to evaluate the need for maintenance therapy.”
It’s important, Dr. McClung said, “for clinicians to be confident that BMD loss is prevented or minimized with longer-term relugolix combination therapy since treatment for uterine fibroids is not a short-term proposition. Given the stability of BMD values between weeks 12 and 24 in the LIBERTY studies, I’d anticipate that we will see stable values with longer-term treatment.”
Dr. McClung disclosed that he has served as a consultant/advisory board member and speaker for Amgen and a consultant/advisory board member for Myovant. Several of his coauthors disclosed employment and ownerships interests in Myovant.
SOURCE: McClung MR et al. Obstet Gynecol. 2020 May. doi: 10.1097/01.AOG.0000662944.34860.b4.
FROM ACOG 2020
Testicular sperm may improve IVF outcomes in some cases
and rates of clinical pregnancy and live birth, a retrospective observational study has found.
The findings were released ahead of the study’s scheduled presentation at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. ACOG canceled the meeting and released abstracts for press coverage.
The study, which won the college’s Donald F. Richardson Memorial Prize Research Paper award, evaluated 112 nonazoospermic couples with an average of 2.3 failed in vitro fertilization (IVF) cycles (range of 1-8). The couples, patients at Shade Grove Fertility in Washington, underwent 157 total intracytoplasmic sperm injection (ICSI) cycles (133 using fresh testicular sperm and 24 using frozen/thawed sperm) and had a total of 101 embryo transfers.
Use of ICSI with testicular sperm compared with prior cycles using ejaculated sperm significantly improved blastocyst development (65% vs. 33%, P < .001), blastocyst conversion rates (67% vs. 35%, P < .001) and the number of embryos available for vitrification (1.6 vs. 0.7, P < .001). Fertilization rates were similar (70% vs. 58%). The clinical pregnancy and live birth rates in couples who used testicular sperm were 44% and 32%, respectively.
The findings suggest improved embryo development and pregnancy rates, and offer more evidence “that this might be something we can offer patients who’ve had multiple failures and no other reason as to why,” M. Blake Evans, DO, clinical fellow in reproductive endocrinology and infertility at the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md., said in a interview. “It looks like there is promise, and we need more research to be conducted.”
The integration of the use of testicular sperm at Shady Grove, a private practice fertility center, and the newly completed analysis of outcomes, were driven by studies “showing that testicular sperm has a low DNA fragmentation index and suggesting that it [offers a] better chance of successful IVF outcomes in patients who have had prior failures,” he said.
Almost all of the men who had ICSC using testicular sperm – 105 of the 112 – had a sperm DNA fragmentation (SDF) assessment of their ejaculate sperm. The mean SDF was 32% and of these 105 men, 66 had an SDF greater than 25% (mean of 49%), a value considered abnormal. The outcomes for patients with elevated SDF did not differ significantly from the overall cohort, Dr. Evans and coinvestigators reported in their abstract.
Dr. Evans said that it’s too early to draw any conclusions about the utility of SDF testing, and that the investigators plan to start prospectively evaluating whether levels of sperm DNA damage as reflected in SDF testing correlate with IVF outcomes.
“Right now the evidence is so conflicting as to whether [SDF testing offers] information that all IVF patients or infertility patients should be receiving,” he said. “Is the reason that testicular sperm works better because there’s lower DNA fragmentation? We think so. … But now that we see [that it] appears the outcomes are better [using testicular sperm], we need to take it a step further and look prospectively at the impact of DNA fragmentation, comparing all the outcomes with normal and abnormal DNA [levels].”
Mark P. Trolice, MD, director of Fertility CARE: The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando, said in an interview that while “there is increasing evidence – and rather clear evidence – that testicular sperm has less DNA damage,” there has been controversy over available outcomes data, most of which have come from small, retrospective studies. Dr. Trolice was not involved in this study presented at ACOG.
In the case of “very poor outcomes with use of ejaculated sperm and a high SDF index, there seems to be support for the use of testicular sperm on the next IVF cycle,” he said. “But there’s also evidence to support that there’s no significant difference in the outcomes of IUI [intrauterine insemination] or IVF based on the SDF index. So this [study] really took a tremendous leap of faith.”
Dr. Trolice said he looks forward to more research – ideally prospective, randomized studies of men with high SDF levels who proceed with assisted reproductive technologies using ejaculated or testicular sperm.
The research was supported by the division of intramural research at the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Evans did not report any relevant financial disclosures. One of his coinvestigators. Micah J. Hill, DO, disclosed having served on the advisory board of Ohana Biosciences. Dr. Trolice reported that he has no relevant financial disclosures. He is a member of the Ob.Gyn. News editorial advisory board.
The abstract was first presented by coauthor Lt. Allison A. Eubanks, MD, of Walter Reed National Military Medical Center, at the ACOG Armed Forces District Annual District Meeting in September 2019.
and rates of clinical pregnancy and live birth, a retrospective observational study has found.
The findings were released ahead of the study’s scheduled presentation at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. ACOG canceled the meeting and released abstracts for press coverage.
The study, which won the college’s Donald F. Richardson Memorial Prize Research Paper award, evaluated 112 nonazoospermic couples with an average of 2.3 failed in vitro fertilization (IVF) cycles (range of 1-8). The couples, patients at Shade Grove Fertility in Washington, underwent 157 total intracytoplasmic sperm injection (ICSI) cycles (133 using fresh testicular sperm and 24 using frozen/thawed sperm) and had a total of 101 embryo transfers.
Use of ICSI with testicular sperm compared with prior cycles using ejaculated sperm significantly improved blastocyst development (65% vs. 33%, P < .001), blastocyst conversion rates (67% vs. 35%, P < .001) and the number of embryos available for vitrification (1.6 vs. 0.7, P < .001). Fertilization rates were similar (70% vs. 58%). The clinical pregnancy and live birth rates in couples who used testicular sperm were 44% and 32%, respectively.
The findings suggest improved embryo development and pregnancy rates, and offer more evidence “that this might be something we can offer patients who’ve had multiple failures and no other reason as to why,” M. Blake Evans, DO, clinical fellow in reproductive endocrinology and infertility at the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md., said in a interview. “It looks like there is promise, and we need more research to be conducted.”
The integration of the use of testicular sperm at Shady Grove, a private practice fertility center, and the newly completed analysis of outcomes, were driven by studies “showing that testicular sperm has a low DNA fragmentation index and suggesting that it [offers a] better chance of successful IVF outcomes in patients who have had prior failures,” he said.
Almost all of the men who had ICSC using testicular sperm – 105 of the 112 – had a sperm DNA fragmentation (SDF) assessment of their ejaculate sperm. The mean SDF was 32% and of these 105 men, 66 had an SDF greater than 25% (mean of 49%), a value considered abnormal. The outcomes for patients with elevated SDF did not differ significantly from the overall cohort, Dr. Evans and coinvestigators reported in their abstract.
Dr. Evans said that it’s too early to draw any conclusions about the utility of SDF testing, and that the investigators plan to start prospectively evaluating whether levels of sperm DNA damage as reflected in SDF testing correlate with IVF outcomes.
“Right now the evidence is so conflicting as to whether [SDF testing offers] information that all IVF patients or infertility patients should be receiving,” he said. “Is the reason that testicular sperm works better because there’s lower DNA fragmentation? We think so. … But now that we see [that it] appears the outcomes are better [using testicular sperm], we need to take it a step further and look prospectively at the impact of DNA fragmentation, comparing all the outcomes with normal and abnormal DNA [levels].”
Mark P. Trolice, MD, director of Fertility CARE: The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando, said in an interview that while “there is increasing evidence – and rather clear evidence – that testicular sperm has less DNA damage,” there has been controversy over available outcomes data, most of which have come from small, retrospective studies. Dr. Trolice was not involved in this study presented at ACOG.
In the case of “very poor outcomes with use of ejaculated sperm and a high SDF index, there seems to be support for the use of testicular sperm on the next IVF cycle,” he said. “But there’s also evidence to support that there’s no significant difference in the outcomes of IUI [intrauterine insemination] or IVF based on the SDF index. So this [study] really took a tremendous leap of faith.”
Dr. Trolice said he looks forward to more research – ideally prospective, randomized studies of men with high SDF levels who proceed with assisted reproductive technologies using ejaculated or testicular sperm.
The research was supported by the division of intramural research at the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Evans did not report any relevant financial disclosures. One of his coinvestigators. Micah J. Hill, DO, disclosed having served on the advisory board of Ohana Biosciences. Dr. Trolice reported that he has no relevant financial disclosures. He is a member of the Ob.Gyn. News editorial advisory board.
The abstract was first presented by coauthor Lt. Allison A. Eubanks, MD, of Walter Reed National Military Medical Center, at the ACOG Armed Forces District Annual District Meeting in September 2019.
and rates of clinical pregnancy and live birth, a retrospective observational study has found.
The findings were released ahead of the study’s scheduled presentation at the annual clinical and scientific meeting of the American College of Obstetricians and Gynecologists. ACOG canceled the meeting and released abstracts for press coverage.
The study, which won the college’s Donald F. Richardson Memorial Prize Research Paper award, evaluated 112 nonazoospermic couples with an average of 2.3 failed in vitro fertilization (IVF) cycles (range of 1-8). The couples, patients at Shade Grove Fertility in Washington, underwent 157 total intracytoplasmic sperm injection (ICSI) cycles (133 using fresh testicular sperm and 24 using frozen/thawed sperm) and had a total of 101 embryo transfers.
Use of ICSI with testicular sperm compared with prior cycles using ejaculated sperm significantly improved blastocyst development (65% vs. 33%, P < .001), blastocyst conversion rates (67% vs. 35%, P < .001) and the number of embryos available for vitrification (1.6 vs. 0.7, P < .001). Fertilization rates were similar (70% vs. 58%). The clinical pregnancy and live birth rates in couples who used testicular sperm were 44% and 32%, respectively.
The findings suggest improved embryo development and pregnancy rates, and offer more evidence “that this might be something we can offer patients who’ve had multiple failures and no other reason as to why,” M. Blake Evans, DO, clinical fellow in reproductive endocrinology and infertility at the Eunice Kennedy Shriver National Institute of Child Health and Human Development, Rockville, Md., said in a interview. “It looks like there is promise, and we need more research to be conducted.”
The integration of the use of testicular sperm at Shady Grove, a private practice fertility center, and the newly completed analysis of outcomes, were driven by studies “showing that testicular sperm has a low DNA fragmentation index and suggesting that it [offers a] better chance of successful IVF outcomes in patients who have had prior failures,” he said.
Almost all of the men who had ICSC using testicular sperm – 105 of the 112 – had a sperm DNA fragmentation (SDF) assessment of their ejaculate sperm. The mean SDF was 32% and of these 105 men, 66 had an SDF greater than 25% (mean of 49%), a value considered abnormal. The outcomes for patients with elevated SDF did not differ significantly from the overall cohort, Dr. Evans and coinvestigators reported in their abstract.
Dr. Evans said that it’s too early to draw any conclusions about the utility of SDF testing, and that the investigators plan to start prospectively evaluating whether levels of sperm DNA damage as reflected in SDF testing correlate with IVF outcomes.
“Right now the evidence is so conflicting as to whether [SDF testing offers] information that all IVF patients or infertility patients should be receiving,” he said. “Is the reason that testicular sperm works better because there’s lower DNA fragmentation? We think so. … But now that we see [that it] appears the outcomes are better [using testicular sperm], we need to take it a step further and look prospectively at the impact of DNA fragmentation, comparing all the outcomes with normal and abnormal DNA [levels].”
Mark P. Trolice, MD, director of Fertility CARE: The IVF Center in Winter Park, Fla., and professor of obstetrics and gynecology at the University of Central Florida, Orlando, said in an interview that while “there is increasing evidence – and rather clear evidence – that testicular sperm has less DNA damage,” there has been controversy over available outcomes data, most of which have come from small, retrospective studies. Dr. Trolice was not involved in this study presented at ACOG.
In the case of “very poor outcomes with use of ejaculated sperm and a high SDF index, there seems to be support for the use of testicular sperm on the next IVF cycle,” he said. “But there’s also evidence to support that there’s no significant difference in the outcomes of IUI [intrauterine insemination] or IVF based on the SDF index. So this [study] really took a tremendous leap of faith.”
Dr. Trolice said he looks forward to more research – ideally prospective, randomized studies of men with high SDF levels who proceed with assisted reproductive technologies using ejaculated or testicular sperm.
The research was supported by the division of intramural research at the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Dr. Evans did not report any relevant financial disclosures. One of his coinvestigators. Micah J. Hill, DO, disclosed having served on the advisory board of Ohana Biosciences. Dr. Trolice reported that he has no relevant financial disclosures. He is a member of the Ob.Gyn. News editorial advisory board.
The abstract was first presented by coauthor Lt. Allison A. Eubanks, MD, of Walter Reed National Military Medical Center, at the ACOG Armed Forces District Annual District Meeting in September 2019.
FROM ACOG 2020