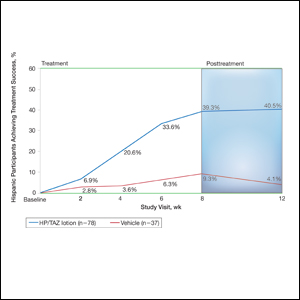
User login
Genome study examines heritability of psoriatic disease subtypes
Genetic variation plays a greater role in the phenotype of cutaneous psoriasis, compared with psoriatic arthritis or psoriasis vulgaris, according to a study published in Scientific Reports.
Psoriatic disease is known to have a strong genetic basis, but understanding variations in the heritability of different forms of psoriasis is important for research into new genes, risk factors, and potential treatments, wrote Quan Li, PhD, of the faculty of medicine at Memorial University, St. John’s, Nfld., and coauthors.
“Heritability essentially refers to how much variation in a trait is due to variation in genetic factors,” the authors wrote. “Better approximation of the heritability of PsC [cutaneous psoriasis], PsV [psoriasis vulgaris], and PsA [psoriatic arthritis] will culminate in more efficient genetic profiling of psoriatic disease and facilitate gene identification studies by providing more accurate estimates of sample sizes needed based on the heritability of different subsets of psoriatic disease.”
The analysis used data from a previous genome-wide association study that looked at single nucleotide polymorphisms – a variation in the DNA sequence of a particular gene – in 2,938 people with PsV, 1,155 with PsC, 715 with PsA, and 3,117 unaffected controls.
The authors used two different modeling approaches to estimate the contribution these genetic variations made to each condition. These both revealed that PsC had a greater heritability than both PsV and PsA.
“This is the first study to quantify the additive heritability of three subsets of psoriatic disease that is attributable to common susceptibility [single nucleotide polymorphisms] from large-scale genotyping arrays,” the authors wrote.
The authors wrote that this finding differed from other population-based genetic epidemiologic studies, which had pointed to much greater heritability for PsA than for PsV. However, these earlier results could be attributed to common environmental factors.
Given these heritability estimates previously made for PsA, the authors commented on the surprising absence of PsA-specific genes that reach significance in genome-wide association studies.
“This is partly explained by the much larger number of patients in the PsC or PsV [genome-wide association] studies to date, compared with PsA,” they wrote, also suggesting that this may be because of the greater disease heterogeneity seen with PsA, compared with psoriasis.
“Considerably increasing the number of PsA patients in [genome-wide association] studies will help clarify the heritability estimate question for PsA,” they wrote, but acknowledged that lower heritability or greater environmental influence could also be an explanation for this finding.
The study was supported by the Atlantic Innovation Fund. The authors reported having no conflicts of interest.
SOURCE: Li Q et al. Sci Rep. 2020 Mar 18. doi: 10.1038/s41598-020-61981-5
Genetic variation plays a greater role in the phenotype of cutaneous psoriasis, compared with psoriatic arthritis or psoriasis vulgaris, according to a study published in Scientific Reports.
Psoriatic disease is known to have a strong genetic basis, but understanding variations in the heritability of different forms of psoriasis is important for research into new genes, risk factors, and potential treatments, wrote Quan Li, PhD, of the faculty of medicine at Memorial University, St. John’s, Nfld., and coauthors.
“Heritability essentially refers to how much variation in a trait is due to variation in genetic factors,” the authors wrote. “Better approximation of the heritability of PsC [cutaneous psoriasis], PsV [psoriasis vulgaris], and PsA [psoriatic arthritis] will culminate in more efficient genetic profiling of psoriatic disease and facilitate gene identification studies by providing more accurate estimates of sample sizes needed based on the heritability of different subsets of psoriatic disease.”
The analysis used data from a previous genome-wide association study that looked at single nucleotide polymorphisms – a variation in the DNA sequence of a particular gene – in 2,938 people with PsV, 1,155 with PsC, 715 with PsA, and 3,117 unaffected controls.
The authors used two different modeling approaches to estimate the contribution these genetic variations made to each condition. These both revealed that PsC had a greater heritability than both PsV and PsA.
“This is the first study to quantify the additive heritability of three subsets of psoriatic disease that is attributable to common susceptibility [single nucleotide polymorphisms] from large-scale genotyping arrays,” the authors wrote.
The authors wrote that this finding differed from other population-based genetic epidemiologic studies, which had pointed to much greater heritability for PsA than for PsV. However, these earlier results could be attributed to common environmental factors.
Given these heritability estimates previously made for PsA, the authors commented on the surprising absence of PsA-specific genes that reach significance in genome-wide association studies.
“This is partly explained by the much larger number of patients in the PsC or PsV [genome-wide association] studies to date, compared with PsA,” they wrote, also suggesting that this may be because of the greater disease heterogeneity seen with PsA, compared with psoriasis.
“Considerably increasing the number of PsA patients in [genome-wide association] studies will help clarify the heritability estimate question for PsA,” they wrote, but acknowledged that lower heritability or greater environmental influence could also be an explanation for this finding.
The study was supported by the Atlantic Innovation Fund. The authors reported having no conflicts of interest.
SOURCE: Li Q et al. Sci Rep. 2020 Mar 18. doi: 10.1038/s41598-020-61981-5
Genetic variation plays a greater role in the phenotype of cutaneous psoriasis, compared with psoriatic arthritis or psoriasis vulgaris, according to a study published in Scientific Reports.
Psoriatic disease is known to have a strong genetic basis, but understanding variations in the heritability of different forms of psoriasis is important for research into new genes, risk factors, and potential treatments, wrote Quan Li, PhD, of the faculty of medicine at Memorial University, St. John’s, Nfld., and coauthors.
“Heritability essentially refers to how much variation in a trait is due to variation in genetic factors,” the authors wrote. “Better approximation of the heritability of PsC [cutaneous psoriasis], PsV [psoriasis vulgaris], and PsA [psoriatic arthritis] will culminate in more efficient genetic profiling of psoriatic disease and facilitate gene identification studies by providing more accurate estimates of sample sizes needed based on the heritability of different subsets of psoriatic disease.”
The analysis used data from a previous genome-wide association study that looked at single nucleotide polymorphisms – a variation in the DNA sequence of a particular gene – in 2,938 people with PsV, 1,155 with PsC, 715 with PsA, and 3,117 unaffected controls.
The authors used two different modeling approaches to estimate the contribution these genetic variations made to each condition. These both revealed that PsC had a greater heritability than both PsV and PsA.
“This is the first study to quantify the additive heritability of three subsets of psoriatic disease that is attributable to common susceptibility [single nucleotide polymorphisms] from large-scale genotyping arrays,” the authors wrote.
The authors wrote that this finding differed from other population-based genetic epidemiologic studies, which had pointed to much greater heritability for PsA than for PsV. However, these earlier results could be attributed to common environmental factors.
Given these heritability estimates previously made for PsA, the authors commented on the surprising absence of PsA-specific genes that reach significance in genome-wide association studies.
“This is partly explained by the much larger number of patients in the PsC or PsV [genome-wide association] studies to date, compared with PsA,” they wrote, also suggesting that this may be because of the greater disease heterogeneity seen with PsA, compared with psoriasis.
“Considerably increasing the number of PsA patients in [genome-wide association] studies will help clarify the heritability estimate question for PsA,” they wrote, but acknowledged that lower heritability or greater environmental influence could also be an explanation for this finding.
The study was supported by the Atlantic Innovation Fund. The authors reported having no conflicts of interest.
SOURCE: Li Q et al. Sci Rep. 2020 Mar 18. doi: 10.1038/s41598-020-61981-5
FROM SCIENTIFIC REPORTS
Rapid shift to adalimumab biosimilars in Denmark contrasts with U.S. experience
Adalimumab biosimilars are years away from entering the marketplace in the United States because of patent disputes, but they already have led to substantial discounts in Denmark, researchers wrote in JAMA Internal Medicine.
The Danish health care system switched almost entirely to adalimumab biosimilars after the patent on the original adalimumab product, Humira, expired there in October 2018. The switch to biosimilars led to an 82% decrease in costs for the medication, wrote Thomas Bo Jensen, MD, and colleagues in a research letter.
Denmark did not automatically substitute biosimilars, but the Danish Medicines Council recommended adalimumab biosimilars for all indications following Humira’s patent expiration. The recommendations “included switching patients to a biosimilar who were already well treated with the originator,” the researchers wrote.
To study the shift to adalimumab biosimilars across all indications in Denmark and calculate cost reductions, Dr. Jensen, of the department of clinical pharmacology at Copenhagen University Hospital Bispebjerg, and coinvestigators examined monthly data on drug sales from Amgros, which purchases all hospital drugs in the country.
“The proportion of adalimumab biosimilars increased from 71.6% (7,040 of 9,829 pens) in November 2018 to 95.1% (8,974 of 9,438 pens) in December 2018,” the researchers wrote. “Costs of adalimumab decreased by 82.8% from September 2018 to December 2018 (September: 8,197 pens at $5.13 million; December: 9,438 pens at $1.01 million).” The results were similar in rheumatology, dermatology, and gastroenterology.
The Food and Drug Administration has approved five adalimumab biosimilars in the United States, but “they will not enter the market until 2023 owing to patent disputes with AbbVie, the manufacturer of Humira,” wrote Jennifer D. Claytor, MD, of the department of internal medicine at University of California, San Francisco, and Walid Gellad, MD, of the division of general internal medicine at University of Pittsburgh, in an accompanying editorial.
The annual postrebate price of Humira doubled between 2013 and 2018, from $19,000 to $38,000, and these price increases may influence the price of biosimilars, “which will be priced using Humira’s price as an anchor,” Dr. Claytor and Dr. Gellad wrote.
A rapid shift to adalimumab biosimilars across the United States when they become available is “unlikely,” they wrote. Nonetheless, “some health care systems of comparable size to Denmark (e.g., the Veterans Affairs system) and others that are larger (e.g., Kaiser Permanente) ... have the ability to switch products quickly through use of formularies and a prescriber workforce. For example, Kaiser Permanente has successfully replaced Remicade (infliximab) with biosimilars in 80% of patients.”
Given the many biologics in development and increasing health care spending, “we need to take seriously the substantial savings offered by biosimilars and the feasibility, as evidenced by Denmark, of switching to biosimilars quickly once they are available on the market,” Dr. Claytor and Dr. Gellad concluded.
The research was supported by an unrestricted grant from Helsefonden. One author disclosed receiving grants from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb outside the current study. The editorial authors had no disclosures.
SOURCE: Jensen TB et al. JAMA Intern Med. 2020 Mar 30. doi: 10.1001/jamainternmed.2020.0338.
Adalimumab biosimilars are years away from entering the marketplace in the United States because of patent disputes, but they already have led to substantial discounts in Denmark, researchers wrote in JAMA Internal Medicine.
The Danish health care system switched almost entirely to adalimumab biosimilars after the patent on the original adalimumab product, Humira, expired there in October 2018. The switch to biosimilars led to an 82% decrease in costs for the medication, wrote Thomas Bo Jensen, MD, and colleagues in a research letter.
Denmark did not automatically substitute biosimilars, but the Danish Medicines Council recommended adalimumab biosimilars for all indications following Humira’s patent expiration. The recommendations “included switching patients to a biosimilar who were already well treated with the originator,” the researchers wrote.
To study the shift to adalimumab biosimilars across all indications in Denmark and calculate cost reductions, Dr. Jensen, of the department of clinical pharmacology at Copenhagen University Hospital Bispebjerg, and coinvestigators examined monthly data on drug sales from Amgros, which purchases all hospital drugs in the country.
“The proportion of adalimumab biosimilars increased from 71.6% (7,040 of 9,829 pens) in November 2018 to 95.1% (8,974 of 9,438 pens) in December 2018,” the researchers wrote. “Costs of adalimumab decreased by 82.8% from September 2018 to December 2018 (September: 8,197 pens at $5.13 million; December: 9,438 pens at $1.01 million).” The results were similar in rheumatology, dermatology, and gastroenterology.
The Food and Drug Administration has approved five adalimumab biosimilars in the United States, but “they will not enter the market until 2023 owing to patent disputes with AbbVie, the manufacturer of Humira,” wrote Jennifer D. Claytor, MD, of the department of internal medicine at University of California, San Francisco, and Walid Gellad, MD, of the division of general internal medicine at University of Pittsburgh, in an accompanying editorial.
The annual postrebate price of Humira doubled between 2013 and 2018, from $19,000 to $38,000, and these price increases may influence the price of biosimilars, “which will be priced using Humira’s price as an anchor,” Dr. Claytor and Dr. Gellad wrote.
A rapid shift to adalimumab biosimilars across the United States when they become available is “unlikely,” they wrote. Nonetheless, “some health care systems of comparable size to Denmark (e.g., the Veterans Affairs system) and others that are larger (e.g., Kaiser Permanente) ... have the ability to switch products quickly through use of formularies and a prescriber workforce. For example, Kaiser Permanente has successfully replaced Remicade (infliximab) with biosimilars in 80% of patients.”
Given the many biologics in development and increasing health care spending, “we need to take seriously the substantial savings offered by biosimilars and the feasibility, as evidenced by Denmark, of switching to biosimilars quickly once they are available on the market,” Dr. Claytor and Dr. Gellad concluded.
The research was supported by an unrestricted grant from Helsefonden. One author disclosed receiving grants from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb outside the current study. The editorial authors had no disclosures.
SOURCE: Jensen TB et al. JAMA Intern Med. 2020 Mar 30. doi: 10.1001/jamainternmed.2020.0338.
Adalimumab biosimilars are years away from entering the marketplace in the United States because of patent disputes, but they already have led to substantial discounts in Denmark, researchers wrote in JAMA Internal Medicine.
The Danish health care system switched almost entirely to adalimumab biosimilars after the patent on the original adalimumab product, Humira, expired there in October 2018. The switch to biosimilars led to an 82% decrease in costs for the medication, wrote Thomas Bo Jensen, MD, and colleagues in a research letter.
Denmark did not automatically substitute biosimilars, but the Danish Medicines Council recommended adalimumab biosimilars for all indications following Humira’s patent expiration. The recommendations “included switching patients to a biosimilar who were already well treated with the originator,” the researchers wrote.
To study the shift to adalimumab biosimilars across all indications in Denmark and calculate cost reductions, Dr. Jensen, of the department of clinical pharmacology at Copenhagen University Hospital Bispebjerg, and coinvestigators examined monthly data on drug sales from Amgros, which purchases all hospital drugs in the country.
“The proportion of adalimumab biosimilars increased from 71.6% (7,040 of 9,829 pens) in November 2018 to 95.1% (8,974 of 9,438 pens) in December 2018,” the researchers wrote. “Costs of adalimumab decreased by 82.8% from September 2018 to December 2018 (September: 8,197 pens at $5.13 million; December: 9,438 pens at $1.01 million).” The results were similar in rheumatology, dermatology, and gastroenterology.
The Food and Drug Administration has approved five adalimumab biosimilars in the United States, but “they will not enter the market until 2023 owing to patent disputes with AbbVie, the manufacturer of Humira,” wrote Jennifer D. Claytor, MD, of the department of internal medicine at University of California, San Francisco, and Walid Gellad, MD, of the division of general internal medicine at University of Pittsburgh, in an accompanying editorial.
The annual postrebate price of Humira doubled between 2013 and 2018, from $19,000 to $38,000, and these price increases may influence the price of biosimilars, “which will be priced using Humira’s price as an anchor,” Dr. Claytor and Dr. Gellad wrote.
A rapid shift to adalimumab biosimilars across the United States when they become available is “unlikely,” they wrote. Nonetheless, “some health care systems of comparable size to Denmark (e.g., the Veterans Affairs system) and others that are larger (e.g., Kaiser Permanente) ... have the ability to switch products quickly through use of formularies and a prescriber workforce. For example, Kaiser Permanente has successfully replaced Remicade (infliximab) with biosimilars in 80% of patients.”
Given the many biologics in development and increasing health care spending, “we need to take seriously the substantial savings offered by biosimilars and the feasibility, as evidenced by Denmark, of switching to biosimilars quickly once they are available on the market,” Dr. Claytor and Dr. Gellad concluded.
The research was supported by an unrestricted grant from Helsefonden. One author disclosed receiving grants from Pfizer, AbbVie, Roche, and Bristol-Myers Squibb outside the current study. The editorial authors had no disclosures.
SOURCE: Jensen TB et al. JAMA Intern Med. 2020 Mar 30. doi: 10.1001/jamainternmed.2020.0338.
FROM JAMA INTERNAL MEDICINE
FDA approves ixekizumab for pediatric plaque psoriasis
The according to an announcement from Lilly.
Patients need to be candidates for systemic therapy or phototherapy and have no known hypersensitivity to the biologic.
The safety, tolerability, and efficacy of the interleukin-17a antagonist were demonstrated in a phase 3 study that included 171 patients aged 6-17 years with moderate to severe plaque psoriasis. At 12 weeks, 89% those on ixekizumab achieved a 75% improvement on Psoriasis Area and Severity Index score, compared with 25% of those on placebo, and 81% achieved a static Physician’s Global Assessment of clear or almost clear, compared with 11% of those on placebo, according to the Lilly statement.
The safety profile seen with ixekizumab (Taltz) among the pediatric patients with plaque psoriasis is consistent with what has been observed among adult patients, although there were higher rates of conjunctivitis, influenza, and urticaria among the pediatric patients, the statement noted. The biologic may increase the risk of infection, and patients should be evaluated for tuberculosis, hypersensitivity, and inflammatory bowel disease. It is also recommended that routine immunizations be completed before initiating treatment.
Ixekizumab was initially approved for treating adults with moderate to severe plaque psoriasis in 2016, followed by approvals for treatment of adults with active psoriatic arthritis in 2017, and for adults with ankylosing spondylitis in August 2019.
The biologic therapies – etanercept, a tumor necrosis factor blocker, and ustekinumab (Stelara), an IL-12/23 antagonist – were previously approved by the FDA for pediatric psoriasis, in children ages 4 years and older and 12 years and older, respectively.
Updated prescribing information for ixekizumab can be found on the Lilly website.
cpalmer@mdedge.com
The according to an announcement from Lilly.
Patients need to be candidates for systemic therapy or phototherapy and have no known hypersensitivity to the biologic.
The safety, tolerability, and efficacy of the interleukin-17a antagonist were demonstrated in a phase 3 study that included 171 patients aged 6-17 years with moderate to severe plaque psoriasis. At 12 weeks, 89% those on ixekizumab achieved a 75% improvement on Psoriasis Area and Severity Index score, compared with 25% of those on placebo, and 81% achieved a static Physician’s Global Assessment of clear or almost clear, compared with 11% of those on placebo, according to the Lilly statement.
The safety profile seen with ixekizumab (Taltz) among the pediatric patients with plaque psoriasis is consistent with what has been observed among adult patients, although there were higher rates of conjunctivitis, influenza, and urticaria among the pediatric patients, the statement noted. The biologic may increase the risk of infection, and patients should be evaluated for tuberculosis, hypersensitivity, and inflammatory bowel disease. It is also recommended that routine immunizations be completed before initiating treatment.
Ixekizumab was initially approved for treating adults with moderate to severe plaque psoriasis in 2016, followed by approvals for treatment of adults with active psoriatic arthritis in 2017, and for adults with ankylosing spondylitis in August 2019.
The biologic therapies – etanercept, a tumor necrosis factor blocker, and ustekinumab (Stelara), an IL-12/23 antagonist – were previously approved by the FDA for pediatric psoriasis, in children ages 4 years and older and 12 years and older, respectively.
Updated prescribing information for ixekizumab can be found on the Lilly website.
cpalmer@mdedge.com
The according to an announcement from Lilly.
Patients need to be candidates for systemic therapy or phototherapy and have no known hypersensitivity to the biologic.
The safety, tolerability, and efficacy of the interleukin-17a antagonist were demonstrated in a phase 3 study that included 171 patients aged 6-17 years with moderate to severe plaque psoriasis. At 12 weeks, 89% those on ixekizumab achieved a 75% improvement on Psoriasis Area and Severity Index score, compared with 25% of those on placebo, and 81% achieved a static Physician’s Global Assessment of clear or almost clear, compared with 11% of those on placebo, according to the Lilly statement.
The safety profile seen with ixekizumab (Taltz) among the pediatric patients with plaque psoriasis is consistent with what has been observed among adult patients, although there were higher rates of conjunctivitis, influenza, and urticaria among the pediatric patients, the statement noted. The biologic may increase the risk of infection, and patients should be evaluated for tuberculosis, hypersensitivity, and inflammatory bowel disease. It is also recommended that routine immunizations be completed before initiating treatment.
Ixekizumab was initially approved for treating adults with moderate to severe plaque psoriasis in 2016, followed by approvals for treatment of adults with active psoriatic arthritis in 2017, and for adults with ankylosing spondylitis in August 2019.
The biologic therapies – etanercept, a tumor necrosis factor blocker, and ustekinumab (Stelara), an IL-12/23 antagonist – were previously approved by the FDA for pediatric psoriasis, in children ages 4 years and older and 12 years and older, respectively.
Updated prescribing information for ixekizumab can be found on the Lilly website.
cpalmer@mdedge.com
Psoriasis Therapy During the COVID-19 Pandemic: Should Patients Continue Biologics?


Dermatology therapies evolve as disease knowledge and investment grow
For much of the past 50 years, many of the drugs used in dermatology have been adopted – and often adapted – from other specialties and used for dermatologic conditions.
“Almost every drug was more or less a hand-me-down” developed first for cancer or other diseases and found later, often serendipitously, to be useful for the skin, said William Eaglstein, MD, thinking back to the 1970s and recalling steroids, tetracyclines, methotrexate, and 5-flourouracil. “The perception always was that skin diseases weren’t serious, that the market was small.”
Much has changed. by dermatologist investigators, and “more and more companies are recognizing the importance of our diseases and the ability to get a return on investment,” said Dr. Eaglstein, past professor and chair of the departments of dermatology at the University of Miami and the University of Pittsburgh, who worked in industry after his academic career.
Psoriasis was a game changer, he and other dermatologists said in interviews. The tumor necrosis factor (TNF)–alpha blockers were first used for other indications, but their marked follow-on success in psoriasis “offered proof of concept clinically – showing that by targeting immune pathways in the skin we could achieve a clinical effect – and proof of concept commercially” that dermatology drugs are worth pursuing by pharmaceutical companies, said William Ju, MD, a cofounder and president of Advancing Innovation in Dermatology, a nonprofit organization that brings together stakeholders to develop novel dermatologic drugs and products.
This resulted in the approval of subsequent biologics, such as ustekinumab (Stelara) which inhibits the signaling of interleukin (IL)–12/IL-23, for psoriasis as their initial indication. Then, biologics targeting IL-17 followed this dermatology-first approach. “Researchers have continued further dissecting out the immunopathological pathways, and antibody drugs targeting IL-23p19 have been approved for psoriasis as the lead indication,” said Dr. Ju, a dermatologist who has worked in industry.
Seth Orlow, MD, PhD, who chairs the department of dermatology at NYU Langone Health, remembers the 1970s through the 1990s as the “era of topicals” developed for dermatologic conditions – topical antifungals, topical corticosteroids, and topical retinoids. The next decade was characterized by formulation tweaks and few novel treatments for dermatology, said Dr. Orlow, who is also professor of pediatric dermatology and director of the program in cutaneous biology at New York University.
Now, given the succession of psoriasis discoveries in the last decade, “large companies are interested in dermatology,” he said in an interview. “There’s an explosion of interest in atopic dermatitis. … and companies are dipping their toes in the water for alopecia areata and vitiligo. That’s amazing.”
Rare diseases like epidermolysis bullosa, ichthyosis, and basal cell nevus syndrome are getting attention as well, boosted by the Orphan Drug Act of 1983, in addition to increased research on disease pathways and growing appreciation of skin diseases. “There’s a lot under development, from small molecules to biologics to gene-based therapies,” Dr. Orlow commented.
The new frontier of atopic dermatitis
The approval in 2017 of dupilumab (Dupixent), a monoclonal antibody that inhibits the signaling of both IL-4 and IL-13) for moderate-severe atopic dermatitis (AD) in adults illustrates the new standing of dermatologic diseases in the field of drug development and commercialization. “Atopic dermatitis had always been the forgotten chronic disease in dermatology. … We’ve had no good treatments,” said Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland. “Dupilumab coming to the forefront [as a dermatology-first indication] has changed the entire perspective of the field. … Everyone is now trying to find the next best drug.”
As with psoriasis, a targeted therapy for AD was made possible by the development in the 1990s of monoclonal antibody technology and the ensuing ability to create biologics that target specific molecules in the body – as well as bedside-to-bench research that homed in on the involvement of particular cytokines.
But there also is a “new understanding of the burden of the disease,” Dr. Simpson observed. In the last 5 years, he said, research funded by the National Eczema Association documented that AD “not only causes inflammation of the skin … but that it affects people at school and in the workplace, that people have multiple mental health comorbidities and skin infections, and that the disease profoundly affects the entire patient in ways that weren’t really recognized or appreciated.”
Having evolved in the footsteps of psoriasis, AD is at a higher starting point in terms of the safety and efficacy of its first biologic, sources said. On the other hand, AD is a much more complex and heterogeneous disease, and researchers are trying to determine which immune pathways and cytokines are most important – and in which populations.
“We’re at the beginning. We’re trying to figure out how to get 80% of patients clear or almost clear [as we can now with psoriasis biologics] rather than almost 40% [as in the dupilumab pivotal trials],” said Dr. Simpson, former cochair of the National Eczema Association’s scientific committee. Public data from ongoing phase 2 and 3 trials of other Th2 cytokine inhibitors suggest that 25%-45% of enrolled patients achieve high levels of clearance, he noted.
Emma Guttman-Yassky, MD, PhD, Sol and Clara Kest Professor and vice-chair for research in the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, said that AD’s heterogeneity involves “many factors, like ethnicity, age … and whether they have an atopic background such as asthma.”
Her research is showing, for instance, that AD in Asian and black patients is different than AD in European-American patients, and that the presence of comorbidities may well have treatment implications. She has also shown that children may have a different phenotype than adults, with greater activation of the Th17 axis that typifies psoriasis.
“For certain patients, we may need to target more than one pathway, or target a different pathway than the Th2 pathway. And treatment may be different in the setting of comorbidities,” said Dr. Guttman-Yassky, who is also director of the laboratory of inflammatory skin diseases at Mount Sinai. “We may think of one treatment – dupilumab, for example – for someone who has asthma and AD. But for patients who don’t have asthma and are Asian, for instance, or for children, we may need additional agents.”
Her research over the years on AD has taught her the importance of human studies over mouse model studies; it was in humans, she noted, that she and other investigators demonstrated “without doubt” that AD is an immune disease and not simply a barrier disease. The Th2 cytokine pathway appears to play the predominant role in AD, though “there still is a strong Th1 component,” she said.
“We’re in a better position to figure this out today [than in the past 20 or even 10 years],” said Dr. Guttman-Yassky, who recalls being told years ago that AD was a “dead end,” that it “would kill [her] career.” Given the evolution of science and the recognition of comorbidities and seriousness of dermatologic diseases, “the stars are aligned to get more [therapies] to these patients.”
Janus kinase (JAK) inhibitors are among these therapies. Three JAK inhibitors are in or have recently completed phase 3 studies for AD; two are currently approved for rheumatoid arthritis, and the other has been designed specifically for AD, Dr. Simpson pointed out. The drugs are oral small molecule drugs that block the JAK signaling pathways for certain proinflammatory cytokines.
“The JAK inhibitors are a real exciting story for dermatology,” he said. “Theoretically, by blocking more cytokines than biologics do, there could be some safety issues – that’s why we’re awaiting big phase 3 study results so we can figure out the risk-benefit balance and guide our patients as to which drug is best.”
Andrew Blauvelt, MD, MBA, president of Oregon Medical Research Center in Portland – a stand-alone dermatology clinical trial center founded in 1998 – likes to envision the evolution of drugs for dermatologic conditions as a funnel, with the most broad-acting drugs at the wide top of the funnel and the most targeted drugs at the bottom tip.
JAK inhibitors, he said, sit near the middle – more targeted and safer than cyclosporine and methotrexate, for instance, but not as targeted as the biologics now available for psoriasis and being developed for AD. “The oral medications that have been developed for psoriasis and those coming for AD are not quite as targeted to the disease,” he noted. “JAK inhibitors have great efficacy – it’s more a question of safety and being able to treat without causing collateral damage.”
Dr. Blauvelt expects the armamentarium of new drugs approved for AD to go from one (dupilumab) to seven within the next 2 years. This will include three new biologics and three new oral JAK inhibitors, he predicts. As the specialty sorts through and integrates these new drugs into practice, dermatologists will increasingly personalize treatment and will face the “nonscientific” challenge of the cost of new therapies and patient access to them, he noted.
In the meantime, said Dr. Simpson, recent drug discoveries have driven more non–pharmaceutical-funded translational research aimed at understanding the underlying biology of AD. The National Institutes of Health, for instance, “is interested in dupilumab and its impact on the skin barrier and skin defense mechanisms,” he said. “We’ll learn a lot more [in coming years].”
Spillover to other diseases
JAK inhibitors – some in oral and some in topical form – are showing efficacy in ongoing research for alopecia areata (AA) and vitiligo as well, Dr. Blauvelt said.
“We’re understanding more about the pathophysiology of these diseases, which historically have been tough diseases for dermatologists to treat,” he said. “The successes in alopecia areata and vitiligo are incredibly exciting actually – it’s very exciting to see hair and pigment coming back. And as we learn more, we should be able to develop [additional] drugs that are more disease targeted than the JAK inhibitors.”
Already, some of the biologics used to treat psoriasis have been studied in patients with hidradenitis suppurativa (HS), a disease in which painful lumps and sometimes tunnels form under the skin, with some success; adalimumab (Humira), a TNF-inhibitor, is now FDA approved for the treatment of moderate-severe HS, and studies are ongoing of IL-17 and IL-23 blockers for the disease.
“The pathophysiology [of HS] is very complex; it’s not nearly as straightforward as psoriasis, and there haven’t been any major breakthroughs yet,” Dr. Blauvelt said. “But the drugs seem to be working better than historical alternatives.”
Regarding AA, Dr. Guttman-Yassky, who is participating in a study of dupilumab for AA, recently found in a retrospective cross-sectional study that patients with the condition are more likely to have atopic comorbidities – asthma, allergic rhinitis, and AD, for instance. “The more comorbid conditions, the greater the risk of developing alopecia areata,” she said. “That could point to a potential pathogenic role of the Th2 axis in the disorder [challenging the traditional view of AA as a singularly Th1-centered disease.] The future will tell.”
Action on rare skin diseases
Both large and small companies have moved into the orphan drug space, investing in research and pursuing orphan drug indications for dermatologic conditions, because “it’s clear now in the marketplace that companies can develop effective drugs for rare disorders and be quite successful,” Dr. Orlow said.
According to a recent analysis, as a result of incentives for rare disease drug development contained in the Orphan Drug Act, 72 indications have been approved for rare skin disease, skin-related cancers, and hereditary disorders with prominent dermatologic manifestations since the law was passed in 1983 (J. Am. Acad. Dermatol. 2019;81[3]:867-77).
Epidermolysis bullosa (EB) is a good example, he and other sources said, of commercial interests merging with growing knowledge of disease pathogenesis as well as the tools needed to develop new treatments.
Research by dermatology scientists and others over the past 40 years, Dr. Ju explained, shed light on the molecular basis underlying the structure and function of the junction between the epidermis and dermis, including the pivotal role that type VII collagen plays in the normal adhesion of these two layers. Researchers then learned that, in EB, the family of genetic diseases characterized by skin fragility, “dystrophic types are caused by mutations in the gene encoding type VII collagen,” he said.
“Just as the advent of monoclonal antibodies allowed us to start attacking psoriasis and atopic dermatitis in unprecedented ways, the advent of gene therapy allows us to potentially address the fundamental molecular genetic defect of various types of EB,” Dr. Ju said.
While gene therapy is “still in its infancy,” companies have begun using the tools to address EB. One gene therapy in the pipeline – in phase 3 clinical trial testing – involves grafting back into patients with recessive dystrophic EB their skin cells that have been genetically modified to produce a correct (nonmutated) type VII collagen, he said.
Basal cell nevus syndrome, or Gorlin syndrome, a rare disease in which patients develop a multitude of basal cell carcinoma tumors, is another example of a “dermatology first” approach, Dr. Ju said. Research identified a genetic mutation that causes the hedgehog signaling pathway to be inappropriately activated in the disease, and a drug, vismodegib, was developed to inhibit this pathway. The drug was initially approved for patients with metastatic basal cell cancer and types of advanced basal cell cancer, and is now being tested in cancers affecting other organs, he said.
Basal cell cancer “is a huge market, but it was really unrecognized in the past,” Dr. Eaglstein said. “Seeing drugs come to market for basal cell cancer – this wouldn’t have happened [decades ago].”
Dr. Ju has worked in the pharmaceutical industry; all other sources in this story have worked with pharmaceutical manufacturers of treatments that are being developed or have been approved to treat dermatologic diseases mentioned in this story. In addition to Dr. Ju, Dr. Eaglstein and Dr. Orlow are cofounders of the Advancing Innovation in Dermatology group; Dr. Orlow is a member of the program committee for the organization’s dermatology summit conference.
For much of the past 50 years, many of the drugs used in dermatology have been adopted – and often adapted – from other specialties and used for dermatologic conditions.
“Almost every drug was more or less a hand-me-down” developed first for cancer or other diseases and found later, often serendipitously, to be useful for the skin, said William Eaglstein, MD, thinking back to the 1970s and recalling steroids, tetracyclines, methotrexate, and 5-flourouracil. “The perception always was that skin diseases weren’t serious, that the market was small.”
Much has changed. by dermatologist investigators, and “more and more companies are recognizing the importance of our diseases and the ability to get a return on investment,” said Dr. Eaglstein, past professor and chair of the departments of dermatology at the University of Miami and the University of Pittsburgh, who worked in industry after his academic career.
Psoriasis was a game changer, he and other dermatologists said in interviews. The tumor necrosis factor (TNF)–alpha blockers were first used for other indications, but their marked follow-on success in psoriasis “offered proof of concept clinically – showing that by targeting immune pathways in the skin we could achieve a clinical effect – and proof of concept commercially” that dermatology drugs are worth pursuing by pharmaceutical companies, said William Ju, MD, a cofounder and president of Advancing Innovation in Dermatology, a nonprofit organization that brings together stakeholders to develop novel dermatologic drugs and products.
This resulted in the approval of subsequent biologics, such as ustekinumab (Stelara) which inhibits the signaling of interleukin (IL)–12/IL-23, for psoriasis as their initial indication. Then, biologics targeting IL-17 followed this dermatology-first approach. “Researchers have continued further dissecting out the immunopathological pathways, and antibody drugs targeting IL-23p19 have been approved for psoriasis as the lead indication,” said Dr. Ju, a dermatologist who has worked in industry.
Seth Orlow, MD, PhD, who chairs the department of dermatology at NYU Langone Health, remembers the 1970s through the 1990s as the “era of topicals” developed for dermatologic conditions – topical antifungals, topical corticosteroids, and topical retinoids. The next decade was characterized by formulation tweaks and few novel treatments for dermatology, said Dr. Orlow, who is also professor of pediatric dermatology and director of the program in cutaneous biology at New York University.
Now, given the succession of psoriasis discoveries in the last decade, “large companies are interested in dermatology,” he said in an interview. “There’s an explosion of interest in atopic dermatitis. … and companies are dipping their toes in the water for alopecia areata and vitiligo. That’s amazing.”
Rare diseases like epidermolysis bullosa, ichthyosis, and basal cell nevus syndrome are getting attention as well, boosted by the Orphan Drug Act of 1983, in addition to increased research on disease pathways and growing appreciation of skin diseases. “There’s a lot under development, from small molecules to biologics to gene-based therapies,” Dr. Orlow commented.
The new frontier of atopic dermatitis
The approval in 2017 of dupilumab (Dupixent), a monoclonal antibody that inhibits the signaling of both IL-4 and IL-13) for moderate-severe atopic dermatitis (AD) in adults illustrates the new standing of dermatologic diseases in the field of drug development and commercialization. “Atopic dermatitis had always been the forgotten chronic disease in dermatology. … We’ve had no good treatments,” said Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland. “Dupilumab coming to the forefront [as a dermatology-first indication] has changed the entire perspective of the field. … Everyone is now trying to find the next best drug.”
As with psoriasis, a targeted therapy for AD was made possible by the development in the 1990s of monoclonal antibody technology and the ensuing ability to create biologics that target specific molecules in the body – as well as bedside-to-bench research that homed in on the involvement of particular cytokines.
But there also is a “new understanding of the burden of the disease,” Dr. Simpson observed. In the last 5 years, he said, research funded by the National Eczema Association documented that AD “not only causes inflammation of the skin … but that it affects people at school and in the workplace, that people have multiple mental health comorbidities and skin infections, and that the disease profoundly affects the entire patient in ways that weren’t really recognized or appreciated.”
Having evolved in the footsteps of psoriasis, AD is at a higher starting point in terms of the safety and efficacy of its first biologic, sources said. On the other hand, AD is a much more complex and heterogeneous disease, and researchers are trying to determine which immune pathways and cytokines are most important – and in which populations.
“We’re at the beginning. We’re trying to figure out how to get 80% of patients clear or almost clear [as we can now with psoriasis biologics] rather than almost 40% [as in the dupilumab pivotal trials],” said Dr. Simpson, former cochair of the National Eczema Association’s scientific committee. Public data from ongoing phase 2 and 3 trials of other Th2 cytokine inhibitors suggest that 25%-45% of enrolled patients achieve high levels of clearance, he noted.
Emma Guttman-Yassky, MD, PhD, Sol and Clara Kest Professor and vice-chair for research in the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, said that AD’s heterogeneity involves “many factors, like ethnicity, age … and whether they have an atopic background such as asthma.”
Her research is showing, for instance, that AD in Asian and black patients is different than AD in European-American patients, and that the presence of comorbidities may well have treatment implications. She has also shown that children may have a different phenotype than adults, with greater activation of the Th17 axis that typifies psoriasis.
“For certain patients, we may need to target more than one pathway, or target a different pathway than the Th2 pathway. And treatment may be different in the setting of comorbidities,” said Dr. Guttman-Yassky, who is also director of the laboratory of inflammatory skin diseases at Mount Sinai. “We may think of one treatment – dupilumab, for example – for someone who has asthma and AD. But for patients who don’t have asthma and are Asian, for instance, or for children, we may need additional agents.”
Her research over the years on AD has taught her the importance of human studies over mouse model studies; it was in humans, she noted, that she and other investigators demonstrated “without doubt” that AD is an immune disease and not simply a barrier disease. The Th2 cytokine pathway appears to play the predominant role in AD, though “there still is a strong Th1 component,” she said.
“We’re in a better position to figure this out today [than in the past 20 or even 10 years],” said Dr. Guttman-Yassky, who recalls being told years ago that AD was a “dead end,” that it “would kill [her] career.” Given the evolution of science and the recognition of comorbidities and seriousness of dermatologic diseases, “the stars are aligned to get more [therapies] to these patients.”
Janus kinase (JAK) inhibitors are among these therapies. Three JAK inhibitors are in or have recently completed phase 3 studies for AD; two are currently approved for rheumatoid arthritis, and the other has been designed specifically for AD, Dr. Simpson pointed out. The drugs are oral small molecule drugs that block the JAK signaling pathways for certain proinflammatory cytokines.
“The JAK inhibitors are a real exciting story for dermatology,” he said. “Theoretically, by blocking more cytokines than biologics do, there could be some safety issues – that’s why we’re awaiting big phase 3 study results so we can figure out the risk-benefit balance and guide our patients as to which drug is best.”
Andrew Blauvelt, MD, MBA, president of Oregon Medical Research Center in Portland – a stand-alone dermatology clinical trial center founded in 1998 – likes to envision the evolution of drugs for dermatologic conditions as a funnel, with the most broad-acting drugs at the wide top of the funnel and the most targeted drugs at the bottom tip.
JAK inhibitors, he said, sit near the middle – more targeted and safer than cyclosporine and methotrexate, for instance, but not as targeted as the biologics now available for psoriasis and being developed for AD. “The oral medications that have been developed for psoriasis and those coming for AD are not quite as targeted to the disease,” he noted. “JAK inhibitors have great efficacy – it’s more a question of safety and being able to treat without causing collateral damage.”
Dr. Blauvelt expects the armamentarium of new drugs approved for AD to go from one (dupilumab) to seven within the next 2 years. This will include three new biologics and three new oral JAK inhibitors, he predicts. As the specialty sorts through and integrates these new drugs into practice, dermatologists will increasingly personalize treatment and will face the “nonscientific” challenge of the cost of new therapies and patient access to them, he noted.
In the meantime, said Dr. Simpson, recent drug discoveries have driven more non–pharmaceutical-funded translational research aimed at understanding the underlying biology of AD. The National Institutes of Health, for instance, “is interested in dupilumab and its impact on the skin barrier and skin defense mechanisms,” he said. “We’ll learn a lot more [in coming years].”
Spillover to other diseases
JAK inhibitors – some in oral and some in topical form – are showing efficacy in ongoing research for alopecia areata (AA) and vitiligo as well, Dr. Blauvelt said.
“We’re understanding more about the pathophysiology of these diseases, which historically have been tough diseases for dermatologists to treat,” he said. “The successes in alopecia areata and vitiligo are incredibly exciting actually – it’s very exciting to see hair and pigment coming back. And as we learn more, we should be able to develop [additional] drugs that are more disease targeted than the JAK inhibitors.”
Already, some of the biologics used to treat psoriasis have been studied in patients with hidradenitis suppurativa (HS), a disease in which painful lumps and sometimes tunnels form under the skin, with some success; adalimumab (Humira), a TNF-inhibitor, is now FDA approved for the treatment of moderate-severe HS, and studies are ongoing of IL-17 and IL-23 blockers for the disease.
“The pathophysiology [of HS] is very complex; it’s not nearly as straightforward as psoriasis, and there haven’t been any major breakthroughs yet,” Dr. Blauvelt said. “But the drugs seem to be working better than historical alternatives.”
Regarding AA, Dr. Guttman-Yassky, who is participating in a study of dupilumab for AA, recently found in a retrospective cross-sectional study that patients with the condition are more likely to have atopic comorbidities – asthma, allergic rhinitis, and AD, for instance. “The more comorbid conditions, the greater the risk of developing alopecia areata,” she said. “That could point to a potential pathogenic role of the Th2 axis in the disorder [challenging the traditional view of AA as a singularly Th1-centered disease.] The future will tell.”
Action on rare skin diseases
Both large and small companies have moved into the orphan drug space, investing in research and pursuing orphan drug indications for dermatologic conditions, because “it’s clear now in the marketplace that companies can develop effective drugs for rare disorders and be quite successful,” Dr. Orlow said.
According to a recent analysis, as a result of incentives for rare disease drug development contained in the Orphan Drug Act, 72 indications have been approved for rare skin disease, skin-related cancers, and hereditary disorders with prominent dermatologic manifestations since the law was passed in 1983 (J. Am. Acad. Dermatol. 2019;81[3]:867-77).
Epidermolysis bullosa (EB) is a good example, he and other sources said, of commercial interests merging with growing knowledge of disease pathogenesis as well as the tools needed to develop new treatments.
Research by dermatology scientists and others over the past 40 years, Dr. Ju explained, shed light on the molecular basis underlying the structure and function of the junction between the epidermis and dermis, including the pivotal role that type VII collagen plays in the normal adhesion of these two layers. Researchers then learned that, in EB, the family of genetic diseases characterized by skin fragility, “dystrophic types are caused by mutations in the gene encoding type VII collagen,” he said.
“Just as the advent of monoclonal antibodies allowed us to start attacking psoriasis and atopic dermatitis in unprecedented ways, the advent of gene therapy allows us to potentially address the fundamental molecular genetic defect of various types of EB,” Dr. Ju said.
While gene therapy is “still in its infancy,” companies have begun using the tools to address EB. One gene therapy in the pipeline – in phase 3 clinical trial testing – involves grafting back into patients with recessive dystrophic EB their skin cells that have been genetically modified to produce a correct (nonmutated) type VII collagen, he said.
Basal cell nevus syndrome, or Gorlin syndrome, a rare disease in which patients develop a multitude of basal cell carcinoma tumors, is another example of a “dermatology first” approach, Dr. Ju said. Research identified a genetic mutation that causes the hedgehog signaling pathway to be inappropriately activated in the disease, and a drug, vismodegib, was developed to inhibit this pathway. The drug was initially approved for patients with metastatic basal cell cancer and types of advanced basal cell cancer, and is now being tested in cancers affecting other organs, he said.
Basal cell cancer “is a huge market, but it was really unrecognized in the past,” Dr. Eaglstein said. “Seeing drugs come to market for basal cell cancer – this wouldn’t have happened [decades ago].”
Dr. Ju has worked in the pharmaceutical industry; all other sources in this story have worked with pharmaceutical manufacturers of treatments that are being developed or have been approved to treat dermatologic diseases mentioned in this story. In addition to Dr. Ju, Dr. Eaglstein and Dr. Orlow are cofounders of the Advancing Innovation in Dermatology group; Dr. Orlow is a member of the program committee for the organization’s dermatology summit conference.
For much of the past 50 years, many of the drugs used in dermatology have been adopted – and often adapted – from other specialties and used for dermatologic conditions.
“Almost every drug was more or less a hand-me-down” developed first for cancer or other diseases and found later, often serendipitously, to be useful for the skin, said William Eaglstein, MD, thinking back to the 1970s and recalling steroids, tetracyclines, methotrexate, and 5-flourouracil. “The perception always was that skin diseases weren’t serious, that the market was small.”
Much has changed. by dermatologist investigators, and “more and more companies are recognizing the importance of our diseases and the ability to get a return on investment,” said Dr. Eaglstein, past professor and chair of the departments of dermatology at the University of Miami and the University of Pittsburgh, who worked in industry after his academic career.
Psoriasis was a game changer, he and other dermatologists said in interviews. The tumor necrosis factor (TNF)–alpha blockers were first used for other indications, but their marked follow-on success in psoriasis “offered proof of concept clinically – showing that by targeting immune pathways in the skin we could achieve a clinical effect – and proof of concept commercially” that dermatology drugs are worth pursuing by pharmaceutical companies, said William Ju, MD, a cofounder and president of Advancing Innovation in Dermatology, a nonprofit organization that brings together stakeholders to develop novel dermatologic drugs and products.
This resulted in the approval of subsequent biologics, such as ustekinumab (Stelara) which inhibits the signaling of interleukin (IL)–12/IL-23, for psoriasis as their initial indication. Then, biologics targeting IL-17 followed this dermatology-first approach. “Researchers have continued further dissecting out the immunopathological pathways, and antibody drugs targeting IL-23p19 have been approved for psoriasis as the lead indication,” said Dr. Ju, a dermatologist who has worked in industry.
Seth Orlow, MD, PhD, who chairs the department of dermatology at NYU Langone Health, remembers the 1970s through the 1990s as the “era of topicals” developed for dermatologic conditions – topical antifungals, topical corticosteroids, and topical retinoids. The next decade was characterized by formulation tweaks and few novel treatments for dermatology, said Dr. Orlow, who is also professor of pediatric dermatology and director of the program in cutaneous biology at New York University.
Now, given the succession of psoriasis discoveries in the last decade, “large companies are interested in dermatology,” he said in an interview. “There’s an explosion of interest in atopic dermatitis. … and companies are dipping their toes in the water for alopecia areata and vitiligo. That’s amazing.”
Rare diseases like epidermolysis bullosa, ichthyosis, and basal cell nevus syndrome are getting attention as well, boosted by the Orphan Drug Act of 1983, in addition to increased research on disease pathways and growing appreciation of skin diseases. “There’s a lot under development, from small molecules to biologics to gene-based therapies,” Dr. Orlow commented.
The new frontier of atopic dermatitis
The approval in 2017 of dupilumab (Dupixent), a monoclonal antibody that inhibits the signaling of both IL-4 and IL-13) for moderate-severe atopic dermatitis (AD) in adults illustrates the new standing of dermatologic diseases in the field of drug development and commercialization. “Atopic dermatitis had always been the forgotten chronic disease in dermatology. … We’ve had no good treatments,” said Eric Simpson, MD, professor of dermatology at Oregon Health & Science University, Portland. “Dupilumab coming to the forefront [as a dermatology-first indication] has changed the entire perspective of the field. … Everyone is now trying to find the next best drug.”
As with psoriasis, a targeted therapy for AD was made possible by the development in the 1990s of monoclonal antibody technology and the ensuing ability to create biologics that target specific molecules in the body – as well as bedside-to-bench research that homed in on the involvement of particular cytokines.
But there also is a “new understanding of the burden of the disease,” Dr. Simpson observed. In the last 5 years, he said, research funded by the National Eczema Association documented that AD “not only causes inflammation of the skin … but that it affects people at school and in the workplace, that people have multiple mental health comorbidities and skin infections, and that the disease profoundly affects the entire patient in ways that weren’t really recognized or appreciated.”
Having evolved in the footsteps of psoriasis, AD is at a higher starting point in terms of the safety and efficacy of its first biologic, sources said. On the other hand, AD is a much more complex and heterogeneous disease, and researchers are trying to determine which immune pathways and cytokines are most important – and in which populations.
“We’re at the beginning. We’re trying to figure out how to get 80% of patients clear or almost clear [as we can now with psoriasis biologics] rather than almost 40% [as in the dupilumab pivotal trials],” said Dr. Simpson, former cochair of the National Eczema Association’s scientific committee. Public data from ongoing phase 2 and 3 trials of other Th2 cytokine inhibitors suggest that 25%-45% of enrolled patients achieve high levels of clearance, he noted.
Emma Guttman-Yassky, MD, PhD, Sol and Clara Kest Professor and vice-chair for research in the department of dermatology at the Icahn School of Medicine at Mount Sinai, New York, said that AD’s heterogeneity involves “many factors, like ethnicity, age … and whether they have an atopic background such as asthma.”
Her research is showing, for instance, that AD in Asian and black patients is different than AD in European-American patients, and that the presence of comorbidities may well have treatment implications. She has also shown that children may have a different phenotype than adults, with greater activation of the Th17 axis that typifies psoriasis.
“For certain patients, we may need to target more than one pathway, or target a different pathway than the Th2 pathway. And treatment may be different in the setting of comorbidities,” said Dr. Guttman-Yassky, who is also director of the laboratory of inflammatory skin diseases at Mount Sinai. “We may think of one treatment – dupilumab, for example – for someone who has asthma and AD. But for patients who don’t have asthma and are Asian, for instance, or for children, we may need additional agents.”
Her research over the years on AD has taught her the importance of human studies over mouse model studies; it was in humans, she noted, that she and other investigators demonstrated “without doubt” that AD is an immune disease and not simply a barrier disease. The Th2 cytokine pathway appears to play the predominant role in AD, though “there still is a strong Th1 component,” she said.
“We’re in a better position to figure this out today [than in the past 20 or even 10 years],” said Dr. Guttman-Yassky, who recalls being told years ago that AD was a “dead end,” that it “would kill [her] career.” Given the evolution of science and the recognition of comorbidities and seriousness of dermatologic diseases, “the stars are aligned to get more [therapies] to these patients.”
Janus kinase (JAK) inhibitors are among these therapies. Three JAK inhibitors are in or have recently completed phase 3 studies for AD; two are currently approved for rheumatoid arthritis, and the other has been designed specifically for AD, Dr. Simpson pointed out. The drugs are oral small molecule drugs that block the JAK signaling pathways for certain proinflammatory cytokines.
“The JAK inhibitors are a real exciting story for dermatology,” he said. “Theoretically, by blocking more cytokines than biologics do, there could be some safety issues – that’s why we’re awaiting big phase 3 study results so we can figure out the risk-benefit balance and guide our patients as to which drug is best.”
Andrew Blauvelt, MD, MBA, president of Oregon Medical Research Center in Portland – a stand-alone dermatology clinical trial center founded in 1998 – likes to envision the evolution of drugs for dermatologic conditions as a funnel, with the most broad-acting drugs at the wide top of the funnel and the most targeted drugs at the bottom tip.
JAK inhibitors, he said, sit near the middle – more targeted and safer than cyclosporine and methotrexate, for instance, but not as targeted as the biologics now available for psoriasis and being developed for AD. “The oral medications that have been developed for psoriasis and those coming for AD are not quite as targeted to the disease,” he noted. “JAK inhibitors have great efficacy – it’s more a question of safety and being able to treat without causing collateral damage.”
Dr. Blauvelt expects the armamentarium of new drugs approved for AD to go from one (dupilumab) to seven within the next 2 years. This will include three new biologics and three new oral JAK inhibitors, he predicts. As the specialty sorts through and integrates these new drugs into practice, dermatologists will increasingly personalize treatment and will face the “nonscientific” challenge of the cost of new therapies and patient access to them, he noted.
In the meantime, said Dr. Simpson, recent drug discoveries have driven more non–pharmaceutical-funded translational research aimed at understanding the underlying biology of AD. The National Institutes of Health, for instance, “is interested in dupilumab and its impact on the skin barrier and skin defense mechanisms,” he said. “We’ll learn a lot more [in coming years].”
Spillover to other diseases
JAK inhibitors – some in oral and some in topical form – are showing efficacy in ongoing research for alopecia areata (AA) and vitiligo as well, Dr. Blauvelt said.
“We’re understanding more about the pathophysiology of these diseases, which historically have been tough diseases for dermatologists to treat,” he said. “The successes in alopecia areata and vitiligo are incredibly exciting actually – it’s very exciting to see hair and pigment coming back. And as we learn more, we should be able to develop [additional] drugs that are more disease targeted than the JAK inhibitors.”
Already, some of the biologics used to treat psoriasis have been studied in patients with hidradenitis suppurativa (HS), a disease in which painful lumps and sometimes tunnels form under the skin, with some success; adalimumab (Humira), a TNF-inhibitor, is now FDA approved for the treatment of moderate-severe HS, and studies are ongoing of IL-17 and IL-23 blockers for the disease.
“The pathophysiology [of HS] is very complex; it’s not nearly as straightforward as psoriasis, and there haven’t been any major breakthroughs yet,” Dr. Blauvelt said. “But the drugs seem to be working better than historical alternatives.”
Regarding AA, Dr. Guttman-Yassky, who is participating in a study of dupilumab for AA, recently found in a retrospective cross-sectional study that patients with the condition are more likely to have atopic comorbidities – asthma, allergic rhinitis, and AD, for instance. “The more comorbid conditions, the greater the risk of developing alopecia areata,” she said. “That could point to a potential pathogenic role of the Th2 axis in the disorder [challenging the traditional view of AA as a singularly Th1-centered disease.] The future will tell.”
Action on rare skin diseases
Both large and small companies have moved into the orphan drug space, investing in research and pursuing orphan drug indications for dermatologic conditions, because “it’s clear now in the marketplace that companies can develop effective drugs for rare disorders and be quite successful,” Dr. Orlow said.
According to a recent analysis, as a result of incentives for rare disease drug development contained in the Orphan Drug Act, 72 indications have been approved for rare skin disease, skin-related cancers, and hereditary disorders with prominent dermatologic manifestations since the law was passed in 1983 (J. Am. Acad. Dermatol. 2019;81[3]:867-77).
Epidermolysis bullosa (EB) is a good example, he and other sources said, of commercial interests merging with growing knowledge of disease pathogenesis as well as the tools needed to develop new treatments.
Research by dermatology scientists and others over the past 40 years, Dr. Ju explained, shed light on the molecular basis underlying the structure and function of the junction between the epidermis and dermis, including the pivotal role that type VII collagen plays in the normal adhesion of these two layers. Researchers then learned that, in EB, the family of genetic diseases characterized by skin fragility, “dystrophic types are caused by mutations in the gene encoding type VII collagen,” he said.
“Just as the advent of monoclonal antibodies allowed us to start attacking psoriasis and atopic dermatitis in unprecedented ways, the advent of gene therapy allows us to potentially address the fundamental molecular genetic defect of various types of EB,” Dr. Ju said.
While gene therapy is “still in its infancy,” companies have begun using the tools to address EB. One gene therapy in the pipeline – in phase 3 clinical trial testing – involves grafting back into patients with recessive dystrophic EB their skin cells that have been genetically modified to produce a correct (nonmutated) type VII collagen, he said.
Basal cell nevus syndrome, or Gorlin syndrome, a rare disease in which patients develop a multitude of basal cell carcinoma tumors, is another example of a “dermatology first” approach, Dr. Ju said. Research identified a genetic mutation that causes the hedgehog signaling pathway to be inappropriately activated in the disease, and a drug, vismodegib, was developed to inhibit this pathway. The drug was initially approved for patients with metastatic basal cell cancer and types of advanced basal cell cancer, and is now being tested in cancers affecting other organs, he said.
Basal cell cancer “is a huge market, but it was really unrecognized in the past,” Dr. Eaglstein said. “Seeing drugs come to market for basal cell cancer – this wouldn’t have happened [decades ago].”
Dr. Ju has worked in the pharmaceutical industry; all other sources in this story have worked with pharmaceutical manufacturers of treatments that are being developed or have been approved to treat dermatologic diseases mentioned in this story. In addition to Dr. Ju, Dr. Eaglstein and Dr. Orlow are cofounders of the Advancing Innovation in Dermatology group; Dr. Orlow is a member of the program committee for the organization’s dermatology summit conference.
AAD-NPF releases first guidelines for nonbiologic treatments of psoriasis
It’s been 11 years since monotherapy and suggest a framework for a number of off-label treatments.
The guidelines, issued jointly with the National Psoriasis Foundation (NPF), were published in the Journal of the American Academy of Dermatology.
“I think we are way behind,” Alan Menter, MD, chairman of the division of dermatology at Baylor University Medical Center, Dallas, and cochair of the guideline writing committee, said in an interview. “Most other countries update their guidelines every 1 or 2 years; we were 10 years behind.” The guidelines for systemic nonbiologic drugs follow up psoriasis guidelines issued by the AAD and the NPF on pediatric patients issued earlier this year, and on phototherapy, biologic treatments, and management of comorbidities issued last year.
“A lot has happened in the last 10 years,” said cochair Craig Elmets, MD, professor of dermatology at the University of Alabama at Birmingham. “While much of the interest is on biologic agents, nonbiologics are still used quite frequently, and the guidelines for their appropriate use have changed. Use of the guidelines provides people in the health profession with the most up to date evidence-based information so they can give their patients the best care.”
The guidelines acknowledge that the medications it covers are still widely used, either by themselves or in combination with biologic agents; readily available; easy to use; and, in the case of older therapies, relatively cheap.
Methotrexate has been available since the 1970s. Given as an injection or taken orally, the guidelines recommend supplementation with folic acid to counteract methotrexate’s side effects, particularly GI upset. The guidelines note that folic acid is less expensive than folinic acid. Combination therapy with methotrexate and tumor necrosis factor (TNF) inhibitors is more effective than methotrexate monotherapy, with a similar side effect profile, the guidelines state.
Methotrexate is more widely used outside the United States, “but it is a very good, quick fix and it’s much safer in children and young people than it is in people with cardiovascular disease,” Dr. Menter noted. “It’s still the most commonly used drug worldwide because it’s cheap, and you do have to worry about the long-term toxicity which is related the liver issues.”
The guidelines say that subcutaneous administration of methotrexate “may be particularly useful” for patients on higher doses, which when taken orally, are associated with a higher risk of GI effects.
Dr. Menter referred to a 2017 study, which reported 41% of patients treated with subcutaneous methotrexate once a week achieved a Psoriasis Area and Severity Index 75 score of 41% after a year of treatment, compared with 10% of those on placebo (Lancet. 2017 Feb 4;389[10068]:528-37).
The guidelines rate strength of recommendation as class A for methotrexate for moderate to severe psoriasis in adults, recommend supplementation with folic or folinic acid to counteract GI complications and liver problems, and note that adalimumab and infliximab are more effective than methotrexate for cutaneous psoriasis. Class B recommendations for methotrexate and psoriasis include statements that patients should begin with a test dose, especially if they have impaired kidney function; methotrexate is effective for peripheral, but not axial, psoriatic arthritis (PsA); and TNF inhibitors are more effective than methotrexate for PsA.
Approved by the FDA in 2014 for psoriasis, apremilast, which inhibits phosphodiesterase-4, is the newest drug in the recommendations. The guidelines recommend its use for moderate to severe psoriasis in adults, with a class A recommendation. Patients should start on a low dose and then build up to the 30-mg, twice-daily dose over 6 days and should be counseled about the risk of depression before starting treatment. Routine laboratory testing can be considered on an individual basis.
The guidelines also lay out three recommendations (and strength of recommendation) for cyclosporine, a drug that’s been around since the 1990s: for severe, recalcitrant cases (class A); for erythrodermic, general pustular, and palmoplantar psoriasis (class B); and as short-term therapy for psoriasis flare in patients already on another drug (class C).
Acitretin is another longstanding therapy used mostly for palmar-plantar psoriasis, but it can also be used as monotherapy for plaque psoriasis as well as erythrodermic and pustular disease. It can also be used in combination with psoralens with UVA for psoriasis and combined with broadband UVB phototherapy for plaque psoriasis. The acitretin recommendations are class B.
The oral Janus kinase (JAK) inhibitor tofacitinib isn’t specifically approved for psoriasis, but it is approved for RA, PsA, and ulcerative colitis. The drug targets the JAK-STAT signaling pathway that causes inflammation. The guidelines state that tofacitinib can be considered for moderate to severe psoriasis, but lists no strength of recommendation. The recommended dose is either 5 or 10 mg orally twice a day, with a caveat that the higher dose carries a higher risk of adverse events. Patients should be evaluated for getting a zoster vaccine before they begin therapy.
“We thought that, because there was probably a small chance that it might get approved for psoriasis, that we would discuss it briefly,” Dr. Menter said of tofacitinib.
Another off-label use the guidelines address is for fumaric and acid esters, also known as fumarates, which are used to in Europe to treat moderate to severe psoriasis. Dimethyl fumarate is approved for relapsing forms of multiple sclerosis in the United States. The guidelines state that fumarates can be used for psoriasis, but offer no strength of recommendation. Side effects include gastrointestinal disturbance and flushing.
Other treatments that are also addressed in the guidelines include a host of systemic immunosuppressants and antimetabolites: azathioprine, hydroxyurea, leflunomide, mycophenolate mofetil, thioguanine, and tacrolimus, none of which are FDA approved for psoriasis. They’re rarely used for psoriasis, but may have value in selected cases, the guidelines state.
Dr. Menter said that apremilast is the only oral drug in the guidelines, but they are the wave of the future for treating psoriasis. “I think there’s a tremendous potential for new oral drugs – TK2 [thymidine kinase], the JAK inhibitors, and other drugs coming down the pipelines. The majority of patients, if you ask them their preference, would like to take an oral drug rather than an injectable drug. And it would be much easier for dermatologists, they wouldn’t have to train patients on how to do the injections.”
Dr. Menter and Dr. Elmets disclosed financial relationships with numerous pharmaceutical companies. Other authors/work group members also had disclosures related to pharmaceutical manufacturers, and several had no disclosures.
SOURCE: Menter A et al. J Am Acad Dermatol. 2020 Feb 28. doi: 10.1016/j.jaad.2020.02.044.
It’s been 11 years since monotherapy and suggest a framework for a number of off-label treatments.
The guidelines, issued jointly with the National Psoriasis Foundation (NPF), were published in the Journal of the American Academy of Dermatology.
“I think we are way behind,” Alan Menter, MD, chairman of the division of dermatology at Baylor University Medical Center, Dallas, and cochair of the guideline writing committee, said in an interview. “Most other countries update their guidelines every 1 or 2 years; we were 10 years behind.” The guidelines for systemic nonbiologic drugs follow up psoriasis guidelines issued by the AAD and the NPF on pediatric patients issued earlier this year, and on phototherapy, biologic treatments, and management of comorbidities issued last year.
“A lot has happened in the last 10 years,” said cochair Craig Elmets, MD, professor of dermatology at the University of Alabama at Birmingham. “While much of the interest is on biologic agents, nonbiologics are still used quite frequently, and the guidelines for their appropriate use have changed. Use of the guidelines provides people in the health profession with the most up to date evidence-based information so they can give their patients the best care.”
The guidelines acknowledge that the medications it covers are still widely used, either by themselves or in combination with biologic agents; readily available; easy to use; and, in the case of older therapies, relatively cheap.
Methotrexate has been available since the 1970s. Given as an injection or taken orally, the guidelines recommend supplementation with folic acid to counteract methotrexate’s side effects, particularly GI upset. The guidelines note that folic acid is less expensive than folinic acid. Combination therapy with methotrexate and tumor necrosis factor (TNF) inhibitors is more effective than methotrexate monotherapy, with a similar side effect profile, the guidelines state.
Methotrexate is more widely used outside the United States, “but it is a very good, quick fix and it’s much safer in children and young people than it is in people with cardiovascular disease,” Dr. Menter noted. “It’s still the most commonly used drug worldwide because it’s cheap, and you do have to worry about the long-term toxicity which is related the liver issues.”
The guidelines say that subcutaneous administration of methotrexate “may be particularly useful” for patients on higher doses, which when taken orally, are associated with a higher risk of GI effects.
Dr. Menter referred to a 2017 study, which reported 41% of patients treated with subcutaneous methotrexate once a week achieved a Psoriasis Area and Severity Index 75 score of 41% after a year of treatment, compared with 10% of those on placebo (Lancet. 2017 Feb 4;389[10068]:528-37).
The guidelines rate strength of recommendation as class A for methotrexate for moderate to severe psoriasis in adults, recommend supplementation with folic or folinic acid to counteract GI complications and liver problems, and note that adalimumab and infliximab are more effective than methotrexate for cutaneous psoriasis. Class B recommendations for methotrexate and psoriasis include statements that patients should begin with a test dose, especially if they have impaired kidney function; methotrexate is effective for peripheral, but not axial, psoriatic arthritis (PsA); and TNF inhibitors are more effective than methotrexate for PsA.
Approved by the FDA in 2014 for psoriasis, apremilast, which inhibits phosphodiesterase-4, is the newest drug in the recommendations. The guidelines recommend its use for moderate to severe psoriasis in adults, with a class A recommendation. Patients should start on a low dose and then build up to the 30-mg, twice-daily dose over 6 days and should be counseled about the risk of depression before starting treatment. Routine laboratory testing can be considered on an individual basis.
The guidelines also lay out three recommendations (and strength of recommendation) for cyclosporine, a drug that’s been around since the 1990s: for severe, recalcitrant cases (class A); for erythrodermic, general pustular, and palmoplantar psoriasis (class B); and as short-term therapy for psoriasis flare in patients already on another drug (class C).
Acitretin is another longstanding therapy used mostly for palmar-plantar psoriasis, but it can also be used as monotherapy for plaque psoriasis as well as erythrodermic and pustular disease. It can also be used in combination with psoralens with UVA for psoriasis and combined with broadband UVB phototherapy for plaque psoriasis. The acitretin recommendations are class B.
The oral Janus kinase (JAK) inhibitor tofacitinib isn’t specifically approved for psoriasis, but it is approved for RA, PsA, and ulcerative colitis. The drug targets the JAK-STAT signaling pathway that causes inflammation. The guidelines state that tofacitinib can be considered for moderate to severe psoriasis, but lists no strength of recommendation. The recommended dose is either 5 or 10 mg orally twice a day, with a caveat that the higher dose carries a higher risk of adverse events. Patients should be evaluated for getting a zoster vaccine before they begin therapy.
“We thought that, because there was probably a small chance that it might get approved for psoriasis, that we would discuss it briefly,” Dr. Menter said of tofacitinib.
Another off-label use the guidelines address is for fumaric and acid esters, also known as fumarates, which are used to in Europe to treat moderate to severe psoriasis. Dimethyl fumarate is approved for relapsing forms of multiple sclerosis in the United States. The guidelines state that fumarates can be used for psoriasis, but offer no strength of recommendation. Side effects include gastrointestinal disturbance and flushing.
Other treatments that are also addressed in the guidelines include a host of systemic immunosuppressants and antimetabolites: azathioprine, hydroxyurea, leflunomide, mycophenolate mofetil, thioguanine, and tacrolimus, none of which are FDA approved for psoriasis. They’re rarely used for psoriasis, but may have value in selected cases, the guidelines state.
Dr. Menter said that apremilast is the only oral drug in the guidelines, but they are the wave of the future for treating psoriasis. “I think there’s a tremendous potential for new oral drugs – TK2 [thymidine kinase], the JAK inhibitors, and other drugs coming down the pipelines. The majority of patients, if you ask them their preference, would like to take an oral drug rather than an injectable drug. And it would be much easier for dermatologists, they wouldn’t have to train patients on how to do the injections.”
Dr. Menter and Dr. Elmets disclosed financial relationships with numerous pharmaceutical companies. Other authors/work group members also had disclosures related to pharmaceutical manufacturers, and several had no disclosures.
SOURCE: Menter A et al. J Am Acad Dermatol. 2020 Feb 28. doi: 10.1016/j.jaad.2020.02.044.
It’s been 11 years since monotherapy and suggest a framework for a number of off-label treatments.
The guidelines, issued jointly with the National Psoriasis Foundation (NPF), were published in the Journal of the American Academy of Dermatology.
“I think we are way behind,” Alan Menter, MD, chairman of the division of dermatology at Baylor University Medical Center, Dallas, and cochair of the guideline writing committee, said in an interview. “Most other countries update their guidelines every 1 or 2 years; we were 10 years behind.” The guidelines for systemic nonbiologic drugs follow up psoriasis guidelines issued by the AAD and the NPF on pediatric patients issued earlier this year, and on phototherapy, biologic treatments, and management of comorbidities issued last year.
“A lot has happened in the last 10 years,” said cochair Craig Elmets, MD, professor of dermatology at the University of Alabama at Birmingham. “While much of the interest is on biologic agents, nonbiologics are still used quite frequently, and the guidelines for their appropriate use have changed. Use of the guidelines provides people in the health profession with the most up to date evidence-based information so they can give their patients the best care.”
The guidelines acknowledge that the medications it covers are still widely used, either by themselves or in combination with biologic agents; readily available; easy to use; and, in the case of older therapies, relatively cheap.
Methotrexate has been available since the 1970s. Given as an injection or taken orally, the guidelines recommend supplementation with folic acid to counteract methotrexate’s side effects, particularly GI upset. The guidelines note that folic acid is less expensive than folinic acid. Combination therapy with methotrexate and tumor necrosis factor (TNF) inhibitors is more effective than methotrexate monotherapy, with a similar side effect profile, the guidelines state.
Methotrexate is more widely used outside the United States, “but it is a very good, quick fix and it’s much safer in children and young people than it is in people with cardiovascular disease,” Dr. Menter noted. “It’s still the most commonly used drug worldwide because it’s cheap, and you do have to worry about the long-term toxicity which is related the liver issues.”
The guidelines say that subcutaneous administration of methotrexate “may be particularly useful” for patients on higher doses, which when taken orally, are associated with a higher risk of GI effects.
Dr. Menter referred to a 2017 study, which reported 41% of patients treated with subcutaneous methotrexate once a week achieved a Psoriasis Area and Severity Index 75 score of 41% after a year of treatment, compared with 10% of those on placebo (Lancet. 2017 Feb 4;389[10068]:528-37).
The guidelines rate strength of recommendation as class A for methotrexate for moderate to severe psoriasis in adults, recommend supplementation with folic or folinic acid to counteract GI complications and liver problems, and note that adalimumab and infliximab are more effective than methotrexate for cutaneous psoriasis. Class B recommendations for methotrexate and psoriasis include statements that patients should begin with a test dose, especially if they have impaired kidney function; methotrexate is effective for peripheral, but not axial, psoriatic arthritis (PsA); and TNF inhibitors are more effective than methotrexate for PsA.
Approved by the FDA in 2014 for psoriasis, apremilast, which inhibits phosphodiesterase-4, is the newest drug in the recommendations. The guidelines recommend its use for moderate to severe psoriasis in adults, with a class A recommendation. Patients should start on a low dose and then build up to the 30-mg, twice-daily dose over 6 days and should be counseled about the risk of depression before starting treatment. Routine laboratory testing can be considered on an individual basis.
The guidelines also lay out three recommendations (and strength of recommendation) for cyclosporine, a drug that’s been around since the 1990s: for severe, recalcitrant cases (class A); for erythrodermic, general pustular, and palmoplantar psoriasis (class B); and as short-term therapy for psoriasis flare in patients already on another drug (class C).
Acitretin is another longstanding therapy used mostly for palmar-plantar psoriasis, but it can also be used as monotherapy for plaque psoriasis as well as erythrodermic and pustular disease. It can also be used in combination with psoralens with UVA for psoriasis and combined with broadband UVB phototherapy for plaque psoriasis. The acitretin recommendations are class B.
The oral Janus kinase (JAK) inhibitor tofacitinib isn’t specifically approved for psoriasis, but it is approved for RA, PsA, and ulcerative colitis. The drug targets the JAK-STAT signaling pathway that causes inflammation. The guidelines state that tofacitinib can be considered for moderate to severe psoriasis, but lists no strength of recommendation. The recommended dose is either 5 or 10 mg orally twice a day, with a caveat that the higher dose carries a higher risk of adverse events. Patients should be evaluated for getting a zoster vaccine before they begin therapy.
“We thought that, because there was probably a small chance that it might get approved for psoriasis, that we would discuss it briefly,” Dr. Menter said of tofacitinib.
Another off-label use the guidelines address is for fumaric and acid esters, also known as fumarates, which are used to in Europe to treat moderate to severe psoriasis. Dimethyl fumarate is approved for relapsing forms of multiple sclerosis in the United States. The guidelines state that fumarates can be used for psoriasis, but offer no strength of recommendation. Side effects include gastrointestinal disturbance and flushing.
Other treatments that are also addressed in the guidelines include a host of systemic immunosuppressants and antimetabolites: azathioprine, hydroxyurea, leflunomide, mycophenolate mofetil, thioguanine, and tacrolimus, none of which are FDA approved for psoriasis. They’re rarely used for psoriasis, but may have value in selected cases, the guidelines state.
Dr. Menter said that apremilast is the only oral drug in the guidelines, but they are the wave of the future for treating psoriasis. “I think there’s a tremendous potential for new oral drugs – TK2 [thymidine kinase], the JAK inhibitors, and other drugs coming down the pipelines. The majority of patients, if you ask them their preference, would like to take an oral drug rather than an injectable drug. And it would be much easier for dermatologists, they wouldn’t have to train patients on how to do the injections.”
Dr. Menter and Dr. Elmets disclosed financial relationships with numerous pharmaceutical companies. Other authors/work group members also had disclosures related to pharmaceutical manufacturers, and several had no disclosures.
SOURCE: Menter A et al. J Am Acad Dermatol. 2020 Feb 28. doi: 10.1016/j.jaad.2020.02.044.
FROM THE JOURNAL OF THE AMERICAN ACADEMY OF DERMATOLOGY
Human Immunodeficiency Virus Infection in a Hepatitis B Virus–Positive Psoriasis Patient Treated With Ustekinumab
To the Editor:
The incidence of psoriasis in human immunodeficiency virus (HIV)–infected patients is similar to the general population, but it usually becomes more severe as immunosuppression increases. Additionally, it tends to be more resistant to conventional therapies, and the incidence and severity of psoriatic arthropathy is increased. Psoriasis often worsens at the time of HIV primary infection.1 We describe a case of a patient with hepatitis B virus (HBV) whose severe plaque psoriasis was controlled on ustekinumab; he was later diagnosed with HIV infection.
A 42-year-old man with HBV treated with entecavir (HBV DNA viral load, <20 copies/mL [inactive carrier, <2000 copies/mL]) presented to our dermatology unit with severe plaque psoriasis (psoriasis area and severity index 23) that caused notable psychologic difficulties such as anxiety and depression. Treatment was attempted with cyclosporine; acitretin; psoralen plus UVA; infliximab; adalimumab; and eventually ustekinumab (45 mg every 3 months), which controlled the condition well (psoriasis area and severity index 0) in an almost completely sustained manner.
Serologic tests requested at one of his analytical control appointments 2 years after initiating treatment with ustekinumab revealed he was HIV positive. The patient reported unprotected sexual intercourse 4 months prior. He was referred to the infectious disease unit and was classified in subtype A1 of HIV infection (CD4 count, 583 cells/µL [reference range, 500-1200 cells/µL]; viral load, 159,268 copies/mL [rapid progression to AIDS, >100,000 copies/mL]). Treatment was initiated with raltegravir, ritonavir, darunavir, and abacavir; tolerance was suitable. Because of the patient’s history of severe psoriasis, treatment with ustekinumab was maintained. Normally, treatment with this drug would be contraindicated in patients with HIV, as it can lead to viral reactivation. Four years after his HIV diagnosis, neither the patient’s cutaneous nor HIV-associated condition had worsened.
For patients with HIV and mild or moderate psoriasis, topical therapies (eg, corticosteroids, vitamin D analogues, tazarotene) are recommended, similar to patients who are HIV negative. Human immunodeficiency virus–positive patients with severe psoriasis who do not respond to topical treatment should receive phototherapy (UVB or psoralen plus UVA) or acitretin along with their antiretroviral drugs.2 In refractory cases, immunosuppressants, including cyclosporine, methotrexate, or tumor necrosis factor α inhibitors, might be used, though experience with them is limited.3,4 Maintaining antiretroviral therapy and prophylaxis against opportunist disease is important in patients who receive such immunosuppressants, as is close monitoring of the viral load.
Ustekinumab is an IL-12/IL-23 monoclonal antibody indicated for the treatment of moderate to severe plaque psoriasis, active psoriatic arthritis, and inflammatory bowel disease. It is contraindicated in patients with clinically important active infections, such as HBV and hepatitis C virus infections.5 However, it was shown to be safe in a group of 18 patients with HBV who had received antiviral prophylaxis6; a degree of reactivation was observed in similar patients who received no such prophylaxis and in others with hepatitis C virus infection.7 The simultaneous use of methotrexate with ustekinumab in the treatment of psoriatic arthritis does not appear to affect the safety of the latter drug.8 Paparizos et al9 described a patient with HIV controlled with antiretroviral drugs who was treated with ustekinumab for psoriasis; no adverse effects were observed.
We report the case of a patient with HBV and psoriasis who was treated with ustekinumab and later became infected with HIV. His ustekinumab treatment was maintained without subsequent cutaneous or systemic complications.
- Menon K, Van Voorhees V, Bebo B, et al. Psoriasis in patients with HIV infection: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2010;62:291-299.
- Chiricozzi A, Saraceno R, Cannizzaro MV. Complete resolution of erythrodermic psoriasis in an HIV and HCV patient unresponsive to antipsoriatic treatments after highly active antiretroviral therapy. Dermatology. 2012;225:333-337.
- Barco D, Puig L, Alomar A. Treatment of moderate-severe psoriasis with etanercept in patients with chronic human immunodeficiency virus infection. Actas Dermosifiliogr. 2010;101(suppl 1):77-81.
- Lindsey SF, Weiss J, Lee ES, et al. Treatment of severe psoriasis and psoriatic arthritis with adalimumab in an HIV positive patient. J Drugs Dermatol. 2014;13:869-871.
- Rustin MH. Long-term safety of biologics in the treatment of moderate to severe plaque psoriasis: review of the current data. Br J Dermatol. 2012;167(suppl 3):3-11.
- Navarro R, Vilarrasa E, Herranz P, et al. Safety and effectiveness of ustekinumab and antitumour necrosis factor therapy in patients with psoriasis and chronic viral hepatitis B or C: a retrospective, multicentre study in a clinical setting. Br J Dermatol. 2013;168:609-616.
- Chiu HY, Chen CH, Wu MS, et al. The safety profile of ustekinumab in the treatment of patients with psoriasis and concurrent hepatitis B or C. Br J Dermatol. 2013;169:1295-1303.
- Weitz JE, Ritchlin CT. Ustekinumab: targeting the IL-17 pathway to improve outcomes in psoriatic arthritis. Expert Opin Biol Ther. 2014;14:515-526.
- Paparizos V, Rallis E, Kirsten L, et al. Ustekinumab for the treatment of HIV psoriasis. J Dermatol Treat. 2012;23:398-399.
To the Editor:
The incidence of psoriasis in human immunodeficiency virus (HIV)–infected patients is similar to the general population, but it usually becomes more severe as immunosuppression increases. Additionally, it tends to be more resistant to conventional therapies, and the incidence and severity of psoriatic arthropathy is increased. Psoriasis often worsens at the time of HIV primary infection.1 We describe a case of a patient with hepatitis B virus (HBV) whose severe plaque psoriasis was controlled on ustekinumab; he was later diagnosed with HIV infection.
A 42-year-old man with HBV treated with entecavir (HBV DNA viral load, <20 copies/mL [inactive carrier, <2000 copies/mL]) presented to our dermatology unit with severe plaque psoriasis (psoriasis area and severity index 23) that caused notable psychologic difficulties such as anxiety and depression. Treatment was attempted with cyclosporine; acitretin; psoralen plus UVA; infliximab; adalimumab; and eventually ustekinumab (45 mg every 3 months), which controlled the condition well (psoriasis area and severity index 0) in an almost completely sustained manner.
Serologic tests requested at one of his analytical control appointments 2 years after initiating treatment with ustekinumab revealed he was HIV positive. The patient reported unprotected sexual intercourse 4 months prior. He was referred to the infectious disease unit and was classified in subtype A1 of HIV infection (CD4 count, 583 cells/µL [reference range, 500-1200 cells/µL]; viral load, 159,268 copies/mL [rapid progression to AIDS, >100,000 copies/mL]). Treatment was initiated with raltegravir, ritonavir, darunavir, and abacavir; tolerance was suitable. Because of the patient’s history of severe psoriasis, treatment with ustekinumab was maintained. Normally, treatment with this drug would be contraindicated in patients with HIV, as it can lead to viral reactivation. Four years after his HIV diagnosis, neither the patient’s cutaneous nor HIV-associated condition had worsened.
For patients with HIV and mild or moderate psoriasis, topical therapies (eg, corticosteroids, vitamin D analogues, tazarotene) are recommended, similar to patients who are HIV negative. Human immunodeficiency virus–positive patients with severe psoriasis who do not respond to topical treatment should receive phototherapy (UVB or psoralen plus UVA) or acitretin along with their antiretroviral drugs.2 In refractory cases, immunosuppressants, including cyclosporine, methotrexate, or tumor necrosis factor α inhibitors, might be used, though experience with them is limited.3,4 Maintaining antiretroviral therapy and prophylaxis against opportunist disease is important in patients who receive such immunosuppressants, as is close monitoring of the viral load.
Ustekinumab is an IL-12/IL-23 monoclonal antibody indicated for the treatment of moderate to severe plaque psoriasis, active psoriatic arthritis, and inflammatory bowel disease. It is contraindicated in patients with clinically important active infections, such as HBV and hepatitis C virus infections.5 However, it was shown to be safe in a group of 18 patients with HBV who had received antiviral prophylaxis6; a degree of reactivation was observed in similar patients who received no such prophylaxis and in others with hepatitis C virus infection.7 The simultaneous use of methotrexate with ustekinumab in the treatment of psoriatic arthritis does not appear to affect the safety of the latter drug.8 Paparizos et al9 described a patient with HIV controlled with antiretroviral drugs who was treated with ustekinumab for psoriasis; no adverse effects were observed.
We report the case of a patient with HBV and psoriasis who was treated with ustekinumab and later became infected with HIV. His ustekinumab treatment was maintained without subsequent cutaneous or systemic complications.
To the Editor:
The incidence of psoriasis in human immunodeficiency virus (HIV)–infected patients is similar to the general population, but it usually becomes more severe as immunosuppression increases. Additionally, it tends to be more resistant to conventional therapies, and the incidence and severity of psoriatic arthropathy is increased. Psoriasis often worsens at the time of HIV primary infection.1 We describe a case of a patient with hepatitis B virus (HBV) whose severe plaque psoriasis was controlled on ustekinumab; he was later diagnosed with HIV infection.
A 42-year-old man with HBV treated with entecavir (HBV DNA viral load, <20 copies/mL [inactive carrier, <2000 copies/mL]) presented to our dermatology unit with severe plaque psoriasis (psoriasis area and severity index 23) that caused notable psychologic difficulties such as anxiety and depression. Treatment was attempted with cyclosporine; acitretin; psoralen plus UVA; infliximab; adalimumab; and eventually ustekinumab (45 mg every 3 months), which controlled the condition well (psoriasis area and severity index 0) in an almost completely sustained manner.
Serologic tests requested at one of his analytical control appointments 2 years after initiating treatment with ustekinumab revealed he was HIV positive. The patient reported unprotected sexual intercourse 4 months prior. He was referred to the infectious disease unit and was classified in subtype A1 of HIV infection (CD4 count, 583 cells/µL [reference range, 500-1200 cells/µL]; viral load, 159,268 copies/mL [rapid progression to AIDS, >100,000 copies/mL]). Treatment was initiated with raltegravir, ritonavir, darunavir, and abacavir; tolerance was suitable. Because of the patient’s history of severe psoriasis, treatment with ustekinumab was maintained. Normally, treatment with this drug would be contraindicated in patients with HIV, as it can lead to viral reactivation. Four years after his HIV diagnosis, neither the patient’s cutaneous nor HIV-associated condition had worsened.
For patients with HIV and mild or moderate psoriasis, topical therapies (eg, corticosteroids, vitamin D analogues, tazarotene) are recommended, similar to patients who are HIV negative. Human immunodeficiency virus–positive patients with severe psoriasis who do not respond to topical treatment should receive phototherapy (UVB or psoralen plus UVA) or acitretin along with their antiretroviral drugs.2 In refractory cases, immunosuppressants, including cyclosporine, methotrexate, or tumor necrosis factor α inhibitors, might be used, though experience with them is limited.3,4 Maintaining antiretroviral therapy and prophylaxis against opportunist disease is important in patients who receive such immunosuppressants, as is close monitoring of the viral load.
Ustekinumab is an IL-12/IL-23 monoclonal antibody indicated for the treatment of moderate to severe plaque psoriasis, active psoriatic arthritis, and inflammatory bowel disease. It is contraindicated in patients with clinically important active infections, such as HBV and hepatitis C virus infections.5 However, it was shown to be safe in a group of 18 patients with HBV who had received antiviral prophylaxis6; a degree of reactivation was observed in similar patients who received no such prophylaxis and in others with hepatitis C virus infection.7 The simultaneous use of methotrexate with ustekinumab in the treatment of psoriatic arthritis does not appear to affect the safety of the latter drug.8 Paparizos et al9 described a patient with HIV controlled with antiretroviral drugs who was treated with ustekinumab for psoriasis; no adverse effects were observed.
We report the case of a patient with HBV and psoriasis who was treated with ustekinumab and later became infected with HIV. His ustekinumab treatment was maintained without subsequent cutaneous or systemic complications.
- Menon K, Van Voorhees V, Bebo B, et al. Psoriasis in patients with HIV infection: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2010;62:291-299.
- Chiricozzi A, Saraceno R, Cannizzaro MV. Complete resolution of erythrodermic psoriasis in an HIV and HCV patient unresponsive to antipsoriatic treatments after highly active antiretroviral therapy. Dermatology. 2012;225:333-337.
- Barco D, Puig L, Alomar A. Treatment of moderate-severe psoriasis with etanercept in patients with chronic human immunodeficiency virus infection. Actas Dermosifiliogr. 2010;101(suppl 1):77-81.
- Lindsey SF, Weiss J, Lee ES, et al. Treatment of severe psoriasis and psoriatic arthritis with adalimumab in an HIV positive patient. J Drugs Dermatol. 2014;13:869-871.
- Rustin MH. Long-term safety of biologics in the treatment of moderate to severe plaque psoriasis: review of the current data. Br J Dermatol. 2012;167(suppl 3):3-11.
- Navarro R, Vilarrasa E, Herranz P, et al. Safety and effectiveness of ustekinumab and antitumour necrosis factor therapy in patients with psoriasis and chronic viral hepatitis B or C: a retrospective, multicentre study in a clinical setting. Br J Dermatol. 2013;168:609-616.
- Chiu HY, Chen CH, Wu MS, et al. The safety profile of ustekinumab in the treatment of patients with psoriasis and concurrent hepatitis B or C. Br J Dermatol. 2013;169:1295-1303.
- Weitz JE, Ritchlin CT. Ustekinumab: targeting the IL-17 pathway to improve outcomes in psoriatic arthritis. Expert Opin Biol Ther. 2014;14:515-526.
- Paparizos V, Rallis E, Kirsten L, et al. Ustekinumab for the treatment of HIV psoriasis. J Dermatol Treat. 2012;23:398-399.
- Menon K, Van Voorhees V, Bebo B, et al. Psoriasis in patients with HIV infection: from the Medical Board of the National Psoriasis Foundation. J Am Acad Dermatol. 2010;62:291-299.
- Chiricozzi A, Saraceno R, Cannizzaro MV. Complete resolution of erythrodermic psoriasis in an HIV and HCV patient unresponsive to antipsoriatic treatments after highly active antiretroviral therapy. Dermatology. 2012;225:333-337.
- Barco D, Puig L, Alomar A. Treatment of moderate-severe psoriasis with etanercept in patients with chronic human immunodeficiency virus infection. Actas Dermosifiliogr. 2010;101(suppl 1):77-81.
- Lindsey SF, Weiss J, Lee ES, et al. Treatment of severe psoriasis and psoriatic arthritis with adalimumab in an HIV positive patient. J Drugs Dermatol. 2014;13:869-871.
- Rustin MH. Long-term safety of biologics in the treatment of moderate to severe plaque psoriasis: review of the current data. Br J Dermatol. 2012;167(suppl 3):3-11.
- Navarro R, Vilarrasa E, Herranz P, et al. Safety and effectiveness of ustekinumab and antitumour necrosis factor therapy in patients with psoriasis and chronic viral hepatitis B or C: a retrospective, multicentre study in a clinical setting. Br J Dermatol. 2013;168:609-616.
- Chiu HY, Chen CH, Wu MS, et al. The safety profile of ustekinumab in the treatment of patients with psoriasis and concurrent hepatitis B or C. Br J Dermatol. 2013;169:1295-1303.
- Weitz JE, Ritchlin CT. Ustekinumab: targeting the IL-17 pathway to improve outcomes in psoriatic arthritis. Expert Opin Biol Ther. 2014;14:515-526.
- Paparizos V, Rallis E, Kirsten L, et al. Ustekinumab for the treatment of HIV psoriasis. J Dermatol Treat. 2012;23:398-399.
Practice Points
- Psoriasis in patients with human immunodeficiency virus (HIV) tends to be more resistant to conventional therapies.
- Experience is limited in the use of immunosuppressants and biologics to treat psoriasis in HIV patients.
- Maintaining antiretroviral therapy and prophylaxis against opportunist disease is important in HIV patients who receive biologics, as is close monitoring of the viral load.
Efficacy, Safety, and Tolerability of Halobetasol Propionate 0.01%–Tazarotene 0.045% Lotion for Moderate to Severe Plaque Psoriasis in the Hispanic Population: Post Hoc Analysis
Psoriasis is a common chronic inflammatory disease affecting a diverse patient population, yet epidemiological and clinical data related to psoriasis in patients with skin of color are sparse. The Hispanic ethnic group includes a broad range of skin types and cultures. Prevalence of psoriasis in a Hispanic population has been reported as lower than in a white population1; however, these data may be influenced by the finding that Hispanic patients are less likely to see a dermatologist when they have skin problems.2 In addition, socioeconomic disparities and cultural variations among racial/ethnic groups may contribute to differences in access to care and thresholds for seeking care,3 leading to a tendency for more severe disease in skin of color and Hispanic ethnic groups.4,5 Greater impairments in health-related quality of life have been reported in patients with skin of color and Hispanic racial/ethnic groups compared to white patients, independent of psoriasis severity.4,6 Postinflammatory pigment alteration at the sites of resolving lesions, a common clinical feature in skin of color, may contribute to the impact of psoriasis on quality of life in patients with skin of color. Psoriasis in darker skin types also can present diagnostic challenges due to overlapping features with other papulosquamous disorders and less conspicuous erythema.7
We present a post hoc analysis of the treatment of moderate to severe psoriasis with a novel fixed-combination halobetasol propionate (HP) 0.01%–tazarotene (TAZ) 0.045% lotion in a Hispanic patient population. Historically, clinical trials for psoriasis have enrolled low proportions of Hispanic patients and other patients with skin of color; in this analysis, the Hispanic population (115/418) represented 28% of the total study population and provided valuable insights.
Methods
Study Design
Two phase 3 randomized controlled trials were conducted to demonstrate the efficacy and safety of HP/TAZ lotion. Patients with a clinical diagnosis of moderate or severe localized psoriasis (N=418) were randomized to receive HP/TAZ lotion or vehicle (2:1 ratio) once daily for 8 weeks with a 4-week posttreatment follow-up.8,9 A post hoc analysis was conducted on data of the self-identified Hispanic population.
Assessments
Efficacy assessments included treatment success (at least a 2-grade improvement from baseline in the investigator global assessment [IGA] and a score of clear or almost clear) and impact on individual signs of psoriasis (at least a 2-grade improvement in erythema, plaque elevation, and scaling) at the target lesion. In addition, reduction in body surface area (BSA) was recorded, and an IGA×BSA score was calculated by multiplying IGA by BSA at each timepoint for each individual patient. A clinically meaningful improvement in disease severity (percentage of patients achieving a 75% reduction in IGA×BSA [IGA×BSA-75]) also was calculated.
Information on reported and observed adverse events (AEs) was obtained at each visit. The safety population included 112 participants (76 in the HP/TAZ group and 36 in the vehicle group).
Statistical Analysis
The statistical and analytical plan is detailed elsewhere9 and relevant to this post hoc analysis. No statistical analysis was carried out to compare data in the Hispanic population with either the overall study population or the non-Hispanic population.
Results
Overall, 115 Hispanic patients (27.5%) were enrolled (eFigure). Patients had a mean (standard deviation [SD]) age of 46.7 (13.12) years, and more than two-thirds were male (n=80, 69.6%).
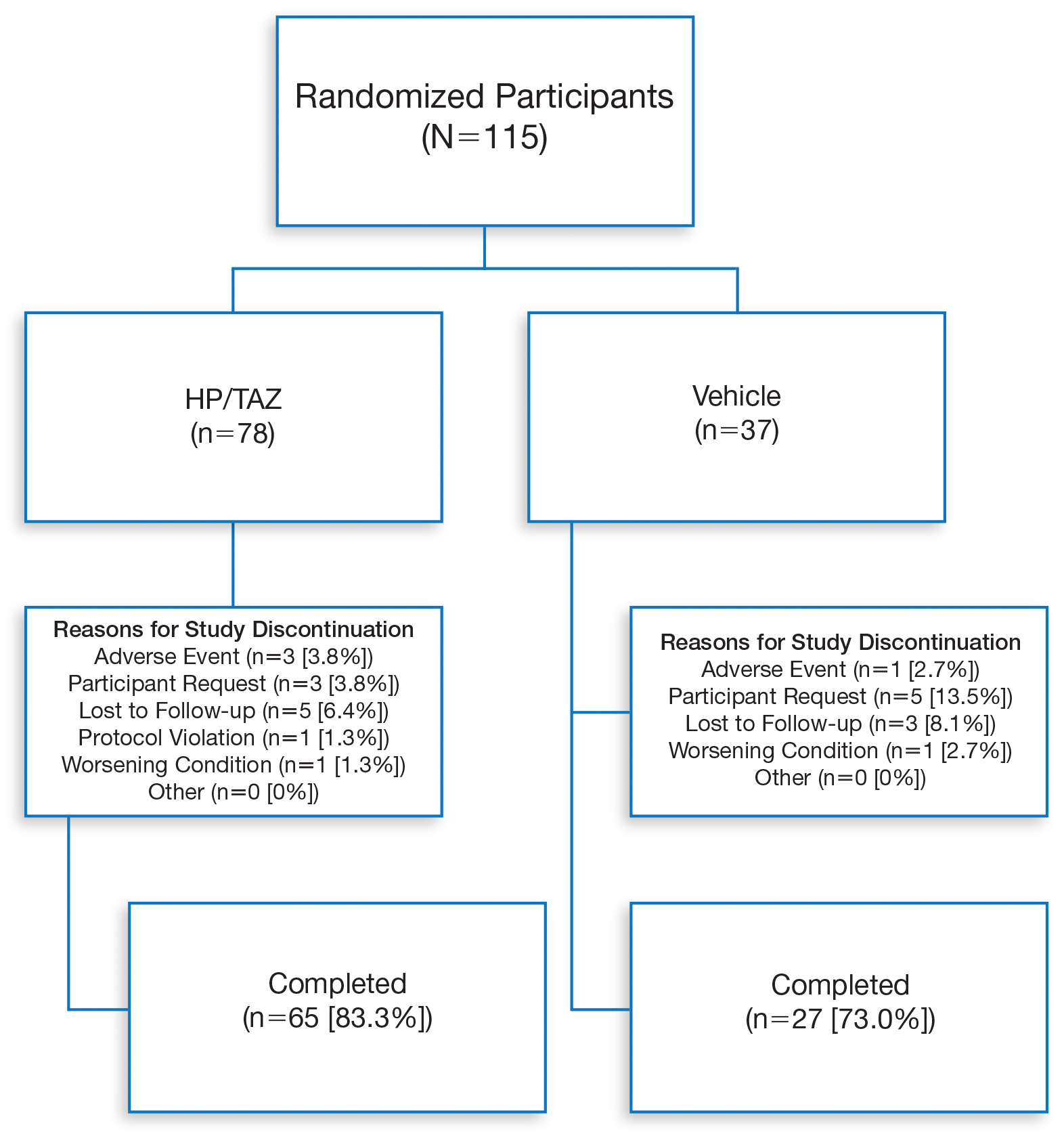
Overall completion rates (80.0%) for Hispanic patients were similar to those in the overall study population, though there were more discontinuations in the vehicle group. The main reasons for treatment discontinuation among Hispanic patients were participant request (n=8, 7.0%), lost to follow-up (n=8, 7.0%), and AEs (n=4, 3.5%). Hispanic patients in this study had more severe disease—18.3% (n=21) had an IGA score of 4 compared to 13.5% (n=41) of non-Hispanic patients—and more severe erythema (19.1% vs 9.6%), plaque elevation (20.0% vs 10.2%), and scaling (15.7% vs 12.9%) compared to the non-Hispanic populations (Table).
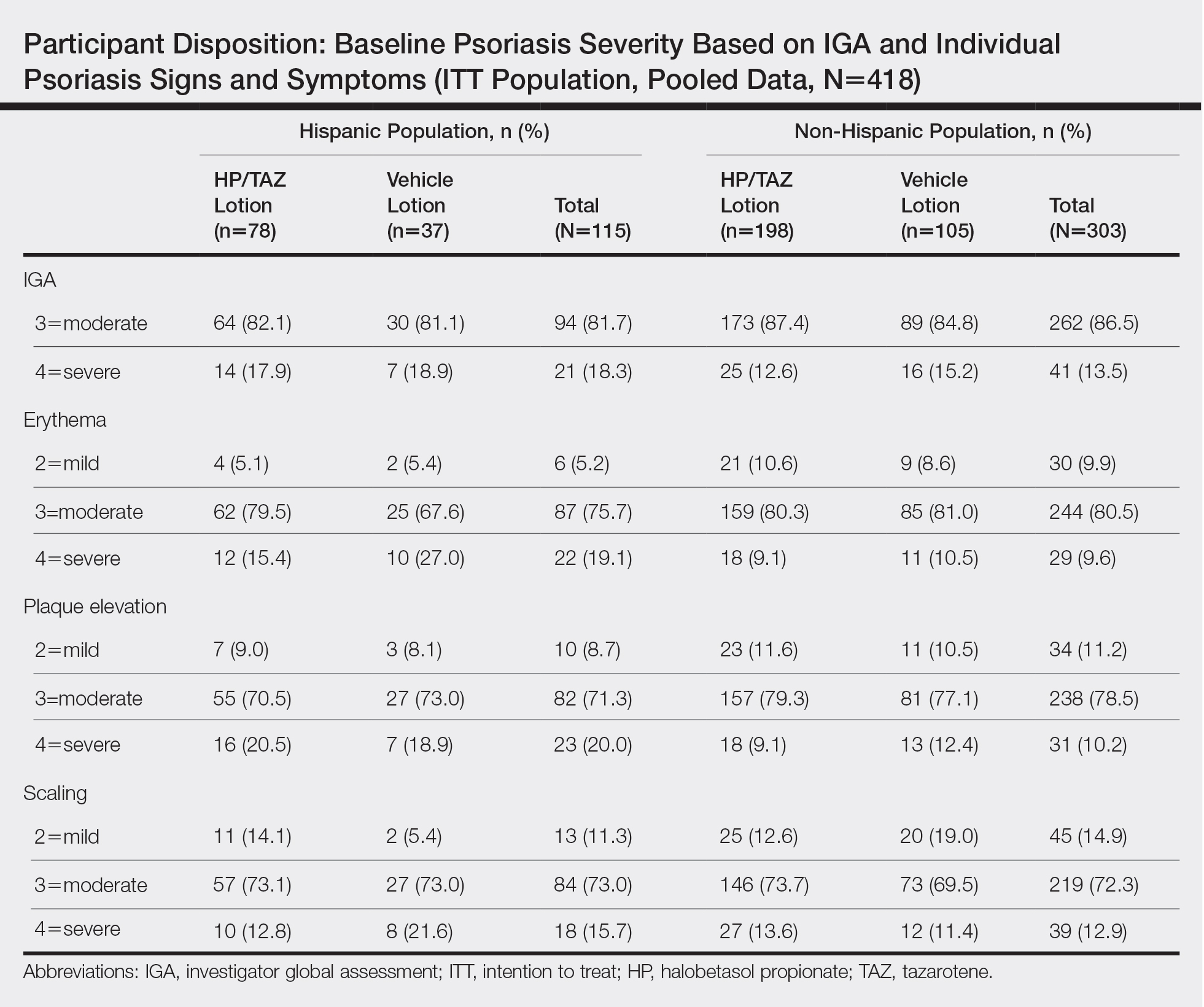
Efficacy of HP/TAZ lotion in Hispanic patients was similar to the overall study populations,9 though maintenance of effect posttreatment appeared to be better. The incidence of treatment-related AEs also was lower.
Halobetasol propionate 0.01%–TAZ 0.045% lotion demonstrated statistically significant superiority based on treatment success compared to vehicle as early as week 4 (P=.034). By week 8, 39.3% of participants treated with HP/TAZ lotion achieved treatment success compared to 9.3% of participants in the vehicle group (P=.002)(Figure 1). Treatment success was maintained over the 4-week posttreatment period, whereby 40.5% of the HP/TAZ-treated participants were treatment successes at week 12 compared to only 4.1% of participants in the vehicle group (P<.001).
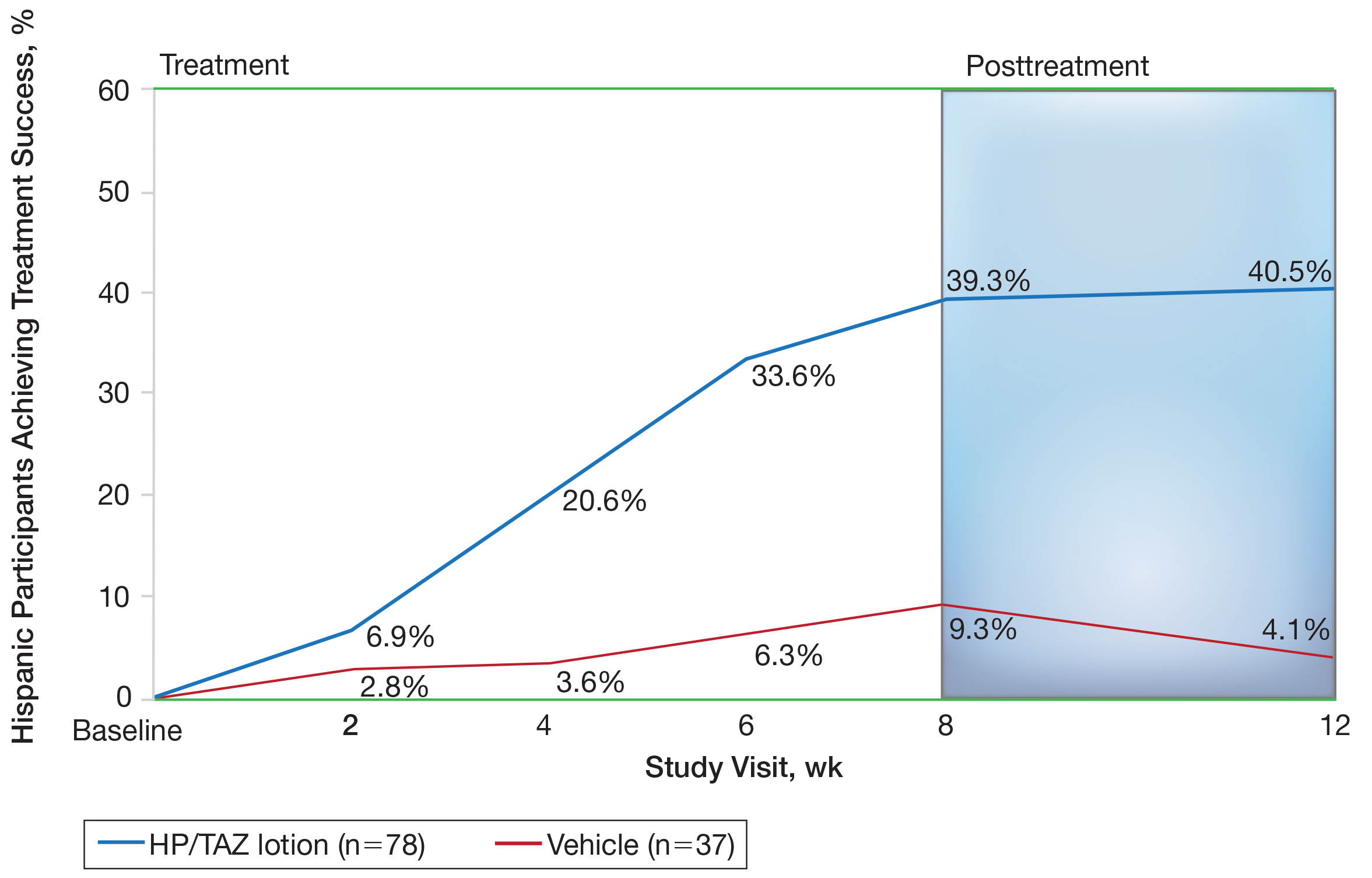
Improvements in psoriasis signs and symptoms at the target lesion were statistically significant compared to vehicle from week 2 (plaque elevation, P=.018) or week 4 (erythema, P=.004; scaling, P<.001)(Figure 2). By week 8, 46.8%, 58.1%, and 63.2% of participants showed at least a 2-grade improvement from baseline and were therefore treatment successes for erythema, plaque elevation, and scaling, respectively (all statistically significant [P<.001] compared to vehicle). The number of participants who achieved at least a 2-grade improvement in erythema with HP/TAZ lotion increased posttreatment from 46.8% to 53.0%.
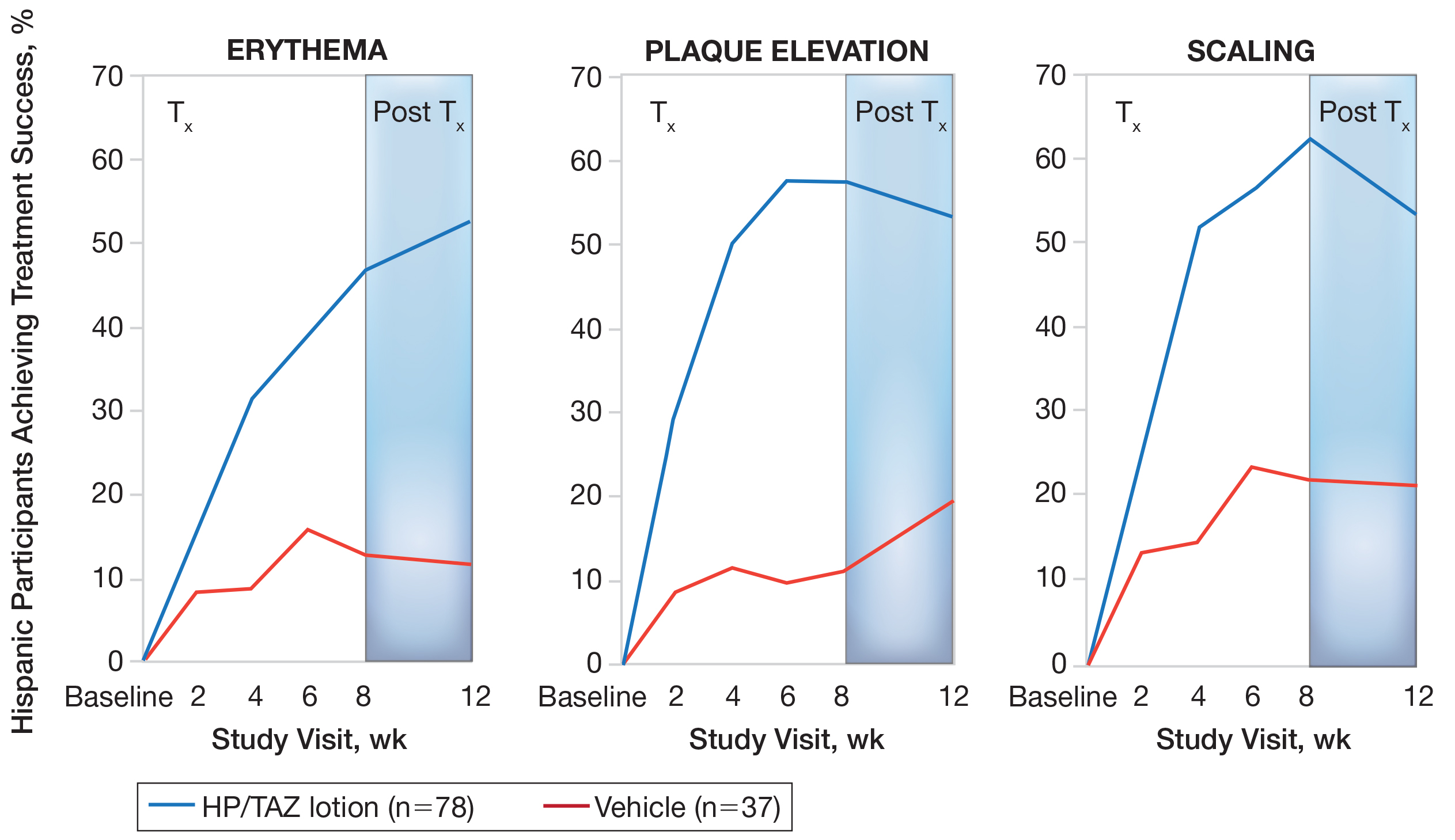
Mean (SD) baseline BSA was 6.2 (3.07), and the mean (SD) size of the target lesion was 36.3 (21.85) cm2. Overall, BSA also was significantly reduced in participants treated with HP/TAZ lotion compared to vehicle. At week 8, the mean percentage change from baseline was —40.7% compared to an increase (+10.1%) in the vehicle group (P=.002)(Figure 3). Improvements in BSA were maintained posttreatment, whereas in the vehicle group, mean (SD) BSA had increased to 6.1 (4.64).
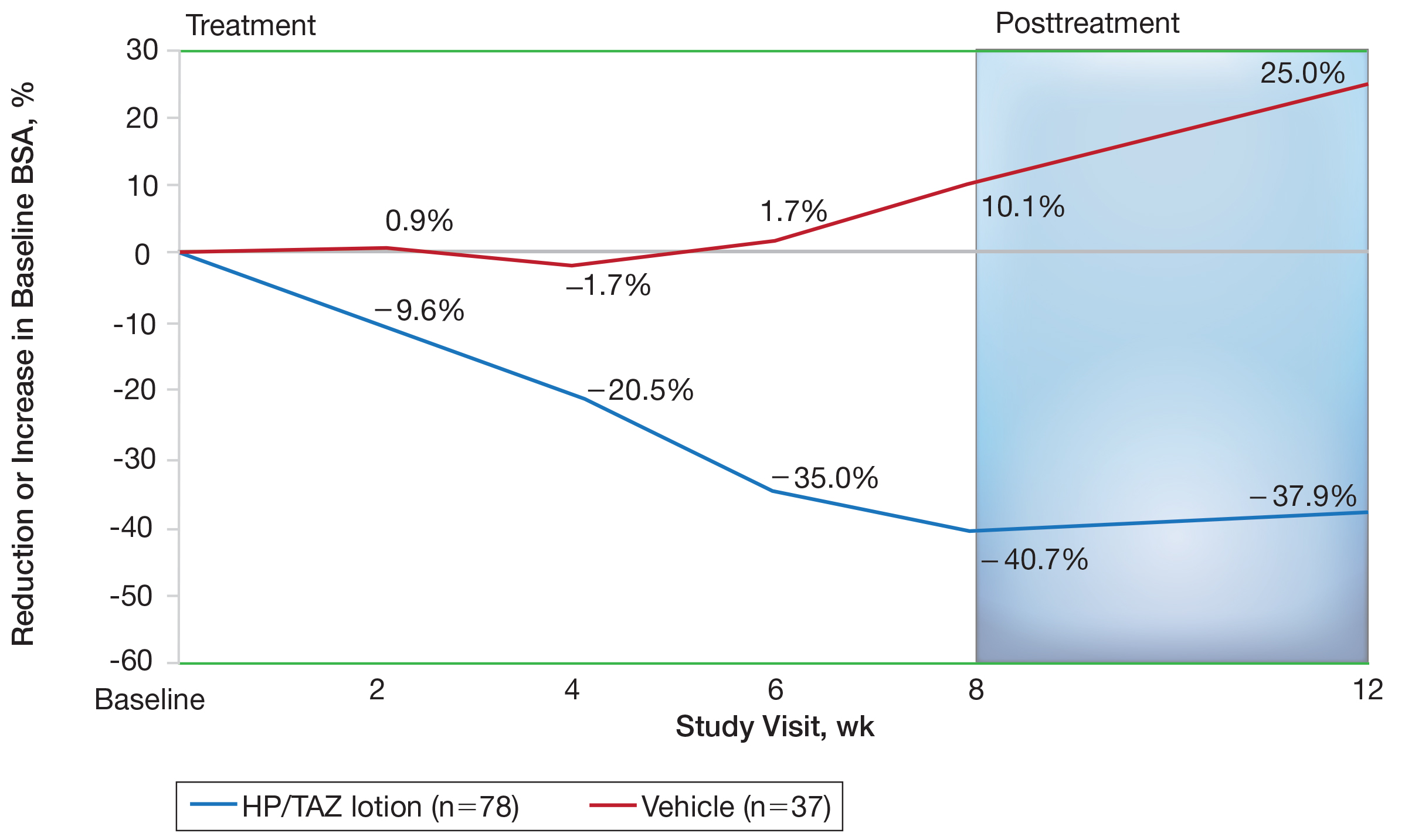
Halobetasol propionate 0.01%–TAZ 0.045% lotion achieved a 50.5% reduction from baseline IGA×BSA by week 8 compared to an 8.5% increase with vehicle (P<.001)(Figure 4). Differences in treatment groups were significant from week 2 (P=.016). Efficacy was maintained posttreatment, with a 50.6% reduction from baseline IGA×BSA at week 12 compared to an increase of 13.6% in the vehicle group (P<.001). Again, although results were similar to the overall study population at week 8 (50.5% vs 51.9%), maintenance of effect was better posttreatment (50.6% vs 46.6%).10
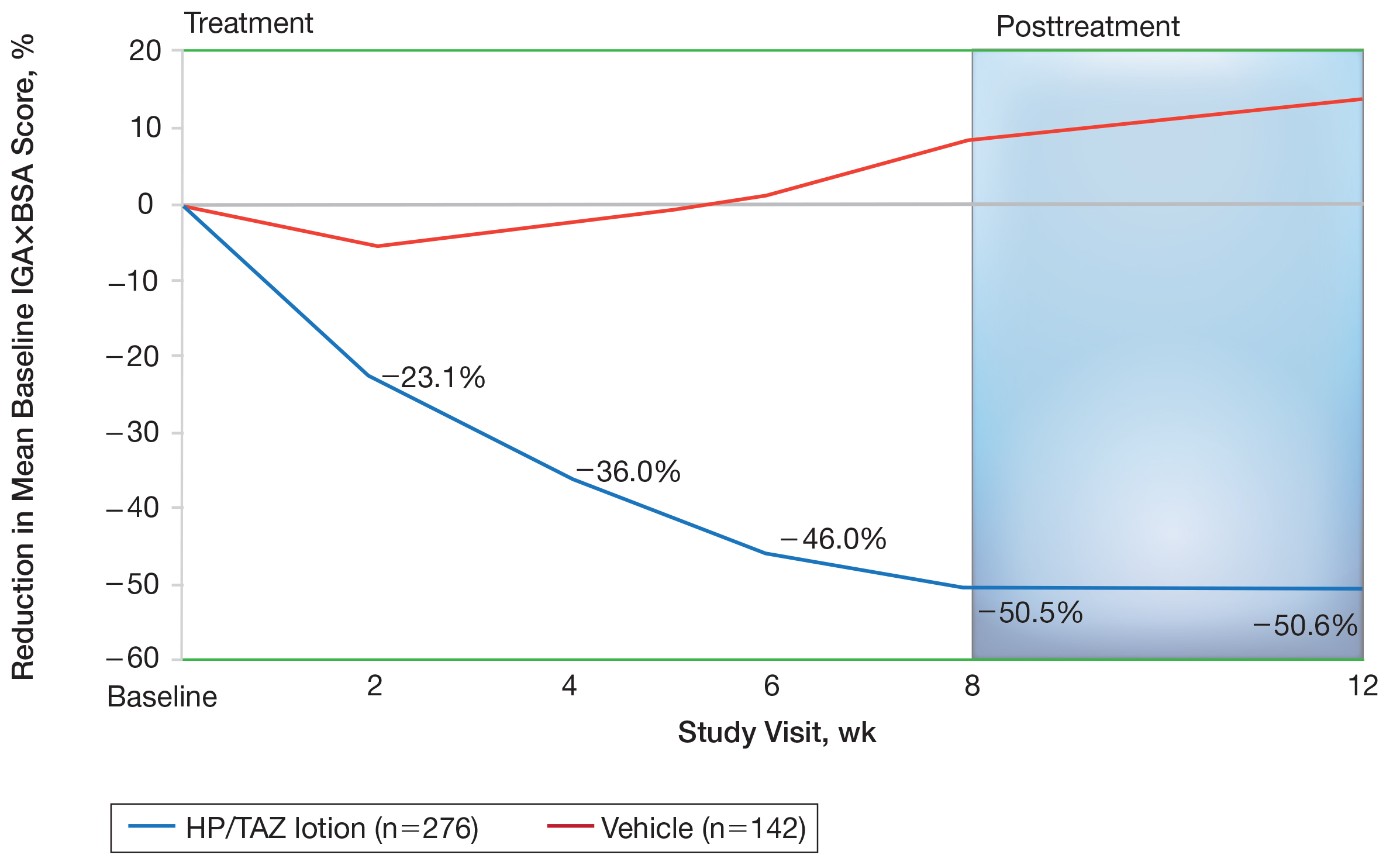
A clinically meaningful effect (IGA×BSA-75) was achieved in 39.7% of Hispanic participants treated with HP/TAZ lotion compared to 8.1% of participants treated with vehicle (P<.001) at week 8. The benefits were significantly different from week 4 and more participants maintained a clinically meaningful effect posttreatment (43.1% vs 7.1%, P<.001)(Figure 5).
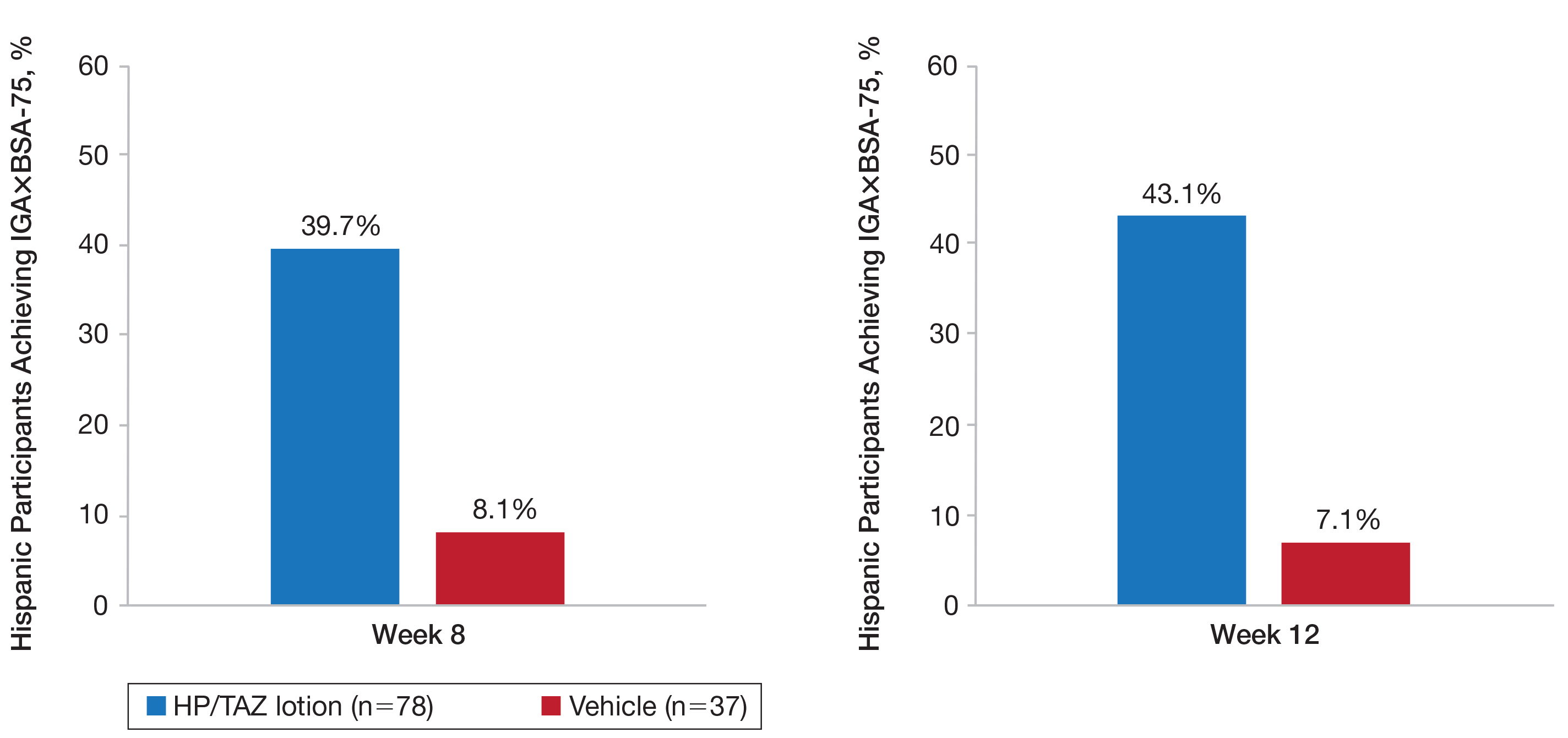
For Hispanic participants overall, 34 participants reported AEs: 26 (34.2%) treated with HP/TAZ lotion and 8 (22.2%) treated with vehicle (eTable). There was 1 (1.3%) serious AE in the HP/TAZ group. Most of the AEs were mild or moderate, with approximately half being related to study treatment. The most common treatment-related AEs in Hispanic participants treated with HP/TAZ lotion were contact dermatitis (n=3, 3.9%) and skin atrophy (n=3, 3.9%) compared to contact dermatitis (n=14, 7.2%) and application-site pain (n=7, 3.6%) in the non-Hispanic population. Pruritus was the most common AE in Hispanic participants treated with vehicle.
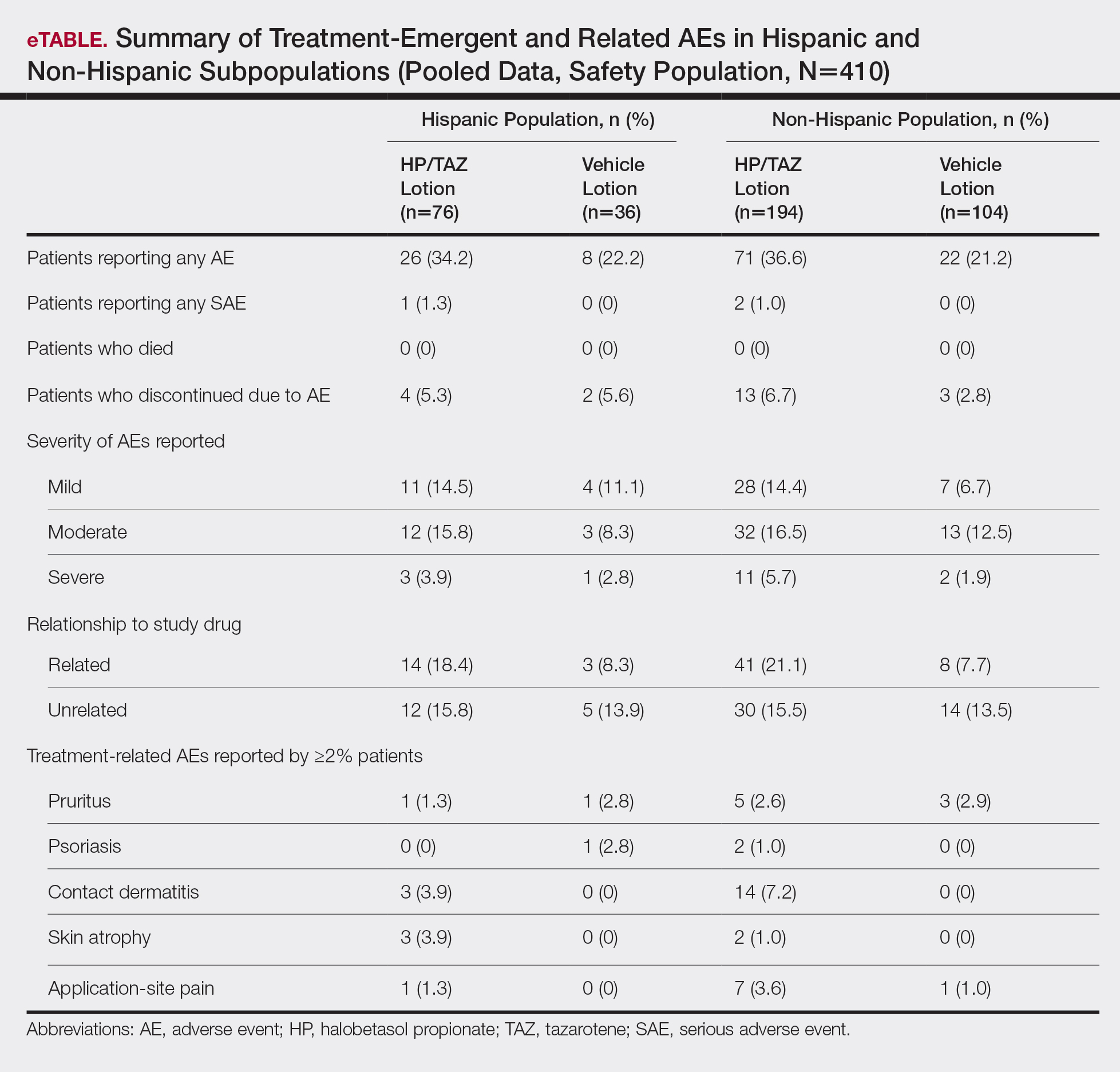
Comment
The large number of Hispanic patients in the 2 phase 3 trials8,9 allowed for this valuable subgroup analysis on the topical treatment of Hispanic patients with plaque psoriasis. Validation of observed differences in maintenance of effect and tolerability warrant further study. Prior clinical studies in psoriasis have tended to enroll a small proportion of Hispanic patients without any post hoc analysis. For example, in a pooled analysis of 4 phase 3 trials with secukinumab, Hispanic patients accounted for only 16% of the overall population.11 In our analysis, the Hispanic cohort represented 28% of the overall study population of 2 phase 3 studies investigating the efficacy, safety, and tolerability of HP/TAZ lotion in patients with moderate to severe psoriasis.8,9 In addition, proportionately more Hispanic patients had severe disease (IGA of 4) or severe signs and symptoms of psoriasis (erythema, plaque elevation, and scaling) than the non-Hispanic population. This finding supports other studies that have suggested Hispanic patients with psoriasis tend to have more severe disease but also may reflect thresholds for seeking care.3-5
Halobetasol propionate 0.01%–TAZ 0.045% lotion was significantly more effective than vehicle for all efficacy assessments. In general, efficacy results with HP/TAZ lotion were similar to those reported in the overall phase 3 study populations over the 8-week treatment period. The only noticeable difference was in the posttreatment period. In the overall study population, efficacy was maintained over the 4-week posttreatment period in the HP/TAZ group. In the Hispanic subpopulation, there appeared to be continued improvement in the number of participants achieving treatment success (IGA and erythema), clinically meaningful success, and further reductions in BSA. Although there is a paucity of studies evaluating psoriasis therapies in Hispanic populations, data on etanercept and secukinumab have been published.6,11
Onset of effect also is an important aspect of treatment. In patients with skin of color, including patients of Hispanic ethnicity and higher Fitzpatrick skin phototypes, early clearance of lesions may help limit the severity and duration of postinflammatory pigment alteration. Improvements in IGA×BSA scores were significant compared to vehicle from week 2 (P=.016), and a clinically meaningful improvement with HP/TAZ lotion (IGA×BSA-75) was seen by week 4 (P=.024).
Halobetasol propionate 0.01%–TAZ 0.045% lotion was well tolerated, both in the 2 phase 3 studies and in the post hoc analysis of the Hispanic subpopulation. The incidence of skin atrophy (n=3, 3.9%) was more common vs the non-Hispanic population (n=2, 1.0%). Other common AEs—contact dermatitis, pruritus, and application-site pain—were more common in the non-Hispanic population.
A limitation of our analysis was that it was a post hoc analysis of the Hispanic participants. The phase 3 studies were not designed to specifically study the impact of treatment on ethnicity/race, though the number of Hispanic participants enrolled in the 2 studies was relatively high. The absence of Fitzpatrick skin phototypes in this data set is another limitation of this study.
Conclusion
Halobetasol propionate 0.01%–TAZ 0.045% lotion was associated with significant, rapid, and sustained reductions in disease severity in a Hispanic population with moderate to severe psoriasis that continued to show improvement posttreatment with good tolerability and safety.
Acknowledgments
We thank Brian Bulley, MSc (Konic Limited, United Kingdom), for assistance with the preparation of the manuscript. Ortho Dermatologics funded Konic’s activities pertaining to this manuscript.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
- Setta-Kaffetzi N, Navarini AA, Patel VM, et al. Rare pathogenic variants in IL36RN underlie a spectrum of psoriasis-associated pustular phenotypes. J Invest Dermatol. 2013;133:1366-1369.
- Yan D, Afifi L, Jeon C, et al. A cross-sectional study of the distribution of psoriasis subtypes in different ethno-racial groups. Dermatol Online J. 2018;24. pii:13030/qt5z21q4k2.
- Abrouk M, Lee K, Brodsky M, et al. Ethnicity affects the presenting severity of psoriasis. J Am Acad Dermatol. 2017;77:180-182.
- Shah SK, Arthur A, Yang YC, et al. A retrospective study to investigate racial and ethnic variations in the treatment of psoriasis with etanercept. J Drugs Dermatol. 2011;10:866-872.
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Blauvelt A, Green LJ, Lebwohl MG, et al. Efficacy of a once-daily fixed combination halobetasol (0.01%) and tazarotene (0.045%) lotion in the treatment of localized moderate-to-severe plaque psoriasis. J Drugs Dermatol. 2019;18:297-299.
- Adsit S, Zaldivar ER, Sofen H, et al. Secukinumab is efficacious and safe in Hispanic patients with moderate-to-severe plaque psoriasis: pooled analysis of four phase 3 trials. Adv Ther. 2017;34:1327-1339.
Psoriasis is a common chronic inflammatory disease affecting a diverse patient population, yet epidemiological and clinical data related to psoriasis in patients with skin of color are sparse. The Hispanic ethnic group includes a broad range of skin types and cultures. Prevalence of psoriasis in a Hispanic population has been reported as lower than in a white population1; however, these data may be influenced by the finding that Hispanic patients are less likely to see a dermatologist when they have skin problems.2 In addition, socioeconomic disparities and cultural variations among racial/ethnic groups may contribute to differences in access to care and thresholds for seeking care,3 leading to a tendency for more severe disease in skin of color and Hispanic ethnic groups.4,5 Greater impairments in health-related quality of life have been reported in patients with skin of color and Hispanic racial/ethnic groups compared to white patients, independent of psoriasis severity.4,6 Postinflammatory pigment alteration at the sites of resolving lesions, a common clinical feature in skin of color, may contribute to the impact of psoriasis on quality of life in patients with skin of color. Psoriasis in darker skin types also can present diagnostic challenges due to overlapping features with other papulosquamous disorders and less conspicuous erythema.7
We present a post hoc analysis of the treatment of moderate to severe psoriasis with a novel fixed-combination halobetasol propionate (HP) 0.01%–tazarotene (TAZ) 0.045% lotion in a Hispanic patient population. Historically, clinical trials for psoriasis have enrolled low proportions of Hispanic patients and other patients with skin of color; in this analysis, the Hispanic population (115/418) represented 28% of the total study population and provided valuable insights.
Methods
Study Design
Two phase 3 randomized controlled trials were conducted to demonstrate the efficacy and safety of HP/TAZ lotion. Patients with a clinical diagnosis of moderate or severe localized psoriasis (N=418) were randomized to receive HP/TAZ lotion or vehicle (2:1 ratio) once daily for 8 weeks with a 4-week posttreatment follow-up.8,9 A post hoc analysis was conducted on data of the self-identified Hispanic population.
Assessments
Efficacy assessments included treatment success (at least a 2-grade improvement from baseline in the investigator global assessment [IGA] and a score of clear or almost clear) and impact on individual signs of psoriasis (at least a 2-grade improvement in erythema, plaque elevation, and scaling) at the target lesion. In addition, reduction in body surface area (BSA) was recorded, and an IGA×BSA score was calculated by multiplying IGA by BSA at each timepoint for each individual patient. A clinically meaningful improvement in disease severity (percentage of patients achieving a 75% reduction in IGA×BSA [IGA×BSA-75]) also was calculated.
Information on reported and observed adverse events (AEs) was obtained at each visit. The safety population included 112 participants (76 in the HP/TAZ group and 36 in the vehicle group).
Statistical Analysis
The statistical and analytical plan is detailed elsewhere9 and relevant to this post hoc analysis. No statistical analysis was carried out to compare data in the Hispanic population with either the overall study population or the non-Hispanic population.
Results
Overall, 115 Hispanic patients (27.5%) were enrolled (eFigure). Patients had a mean (standard deviation [SD]) age of 46.7 (13.12) years, and more than two-thirds were male (n=80, 69.6%).

Overall completion rates (80.0%) for Hispanic patients were similar to those in the overall study population, though there were more discontinuations in the vehicle group. The main reasons for treatment discontinuation among Hispanic patients were participant request (n=8, 7.0%), lost to follow-up (n=8, 7.0%), and AEs (n=4, 3.5%). Hispanic patients in this study had more severe disease—18.3% (n=21) had an IGA score of 4 compared to 13.5% (n=41) of non-Hispanic patients—and more severe erythema (19.1% vs 9.6%), plaque elevation (20.0% vs 10.2%), and scaling (15.7% vs 12.9%) compared to the non-Hispanic populations (Table).

Efficacy of HP/TAZ lotion in Hispanic patients was similar to the overall study populations,9 though maintenance of effect posttreatment appeared to be better. The incidence of treatment-related AEs also was lower.
Halobetasol propionate 0.01%–TAZ 0.045% lotion demonstrated statistically significant superiority based on treatment success compared to vehicle as early as week 4 (P=.034). By week 8, 39.3% of participants treated with HP/TAZ lotion achieved treatment success compared to 9.3% of participants in the vehicle group (P=.002)(Figure 1). Treatment success was maintained over the 4-week posttreatment period, whereby 40.5% of the HP/TAZ-treated participants were treatment successes at week 12 compared to only 4.1% of participants in the vehicle group (P<.001).

Improvements in psoriasis signs and symptoms at the target lesion were statistically significant compared to vehicle from week 2 (plaque elevation, P=.018) or week 4 (erythema, P=.004; scaling, P<.001)(Figure 2). By week 8, 46.8%, 58.1%, and 63.2% of participants showed at least a 2-grade improvement from baseline and were therefore treatment successes for erythema, plaque elevation, and scaling, respectively (all statistically significant [P<.001] compared to vehicle). The number of participants who achieved at least a 2-grade improvement in erythema with HP/TAZ lotion increased posttreatment from 46.8% to 53.0%.

Mean (SD) baseline BSA was 6.2 (3.07), and the mean (SD) size of the target lesion was 36.3 (21.85) cm2. Overall, BSA also was significantly reduced in participants treated with HP/TAZ lotion compared to vehicle. At week 8, the mean percentage change from baseline was —40.7% compared to an increase (+10.1%) in the vehicle group (P=.002)(Figure 3). Improvements in BSA were maintained posttreatment, whereas in the vehicle group, mean (SD) BSA had increased to 6.1 (4.64).

Halobetasol propionate 0.01%–TAZ 0.045% lotion achieved a 50.5% reduction from baseline IGA×BSA by week 8 compared to an 8.5% increase with vehicle (P<.001)(Figure 4). Differences in treatment groups were significant from week 2 (P=.016). Efficacy was maintained posttreatment, with a 50.6% reduction from baseline IGA×BSA at week 12 compared to an increase of 13.6% in the vehicle group (P<.001). Again, although results were similar to the overall study population at week 8 (50.5% vs 51.9%), maintenance of effect was better posttreatment (50.6% vs 46.6%).10

A clinically meaningful effect (IGA×BSA-75) was achieved in 39.7% of Hispanic participants treated with HP/TAZ lotion compared to 8.1% of participants treated with vehicle (P<.001) at week 8. The benefits were significantly different from week 4 and more participants maintained a clinically meaningful effect posttreatment (43.1% vs 7.1%, P<.001)(Figure 5).

For Hispanic participants overall, 34 participants reported AEs: 26 (34.2%) treated with HP/TAZ lotion and 8 (22.2%) treated with vehicle (eTable). There was 1 (1.3%) serious AE in the HP/TAZ group. Most of the AEs were mild or moderate, with approximately half being related to study treatment. The most common treatment-related AEs in Hispanic participants treated with HP/TAZ lotion were contact dermatitis (n=3, 3.9%) and skin atrophy (n=3, 3.9%) compared to contact dermatitis (n=14, 7.2%) and application-site pain (n=7, 3.6%) in the non-Hispanic population. Pruritus was the most common AE in Hispanic participants treated with vehicle.

Comment
The large number of Hispanic patients in the 2 phase 3 trials8,9 allowed for this valuable subgroup analysis on the topical treatment of Hispanic patients with plaque psoriasis. Validation of observed differences in maintenance of effect and tolerability warrant further study. Prior clinical studies in psoriasis have tended to enroll a small proportion of Hispanic patients without any post hoc analysis. For example, in a pooled analysis of 4 phase 3 trials with secukinumab, Hispanic patients accounted for only 16% of the overall population.11 In our analysis, the Hispanic cohort represented 28% of the overall study population of 2 phase 3 studies investigating the efficacy, safety, and tolerability of HP/TAZ lotion in patients with moderate to severe psoriasis.8,9 In addition, proportionately more Hispanic patients had severe disease (IGA of 4) or severe signs and symptoms of psoriasis (erythema, plaque elevation, and scaling) than the non-Hispanic population. This finding supports other studies that have suggested Hispanic patients with psoriasis tend to have more severe disease but also may reflect thresholds for seeking care.3-5
Halobetasol propionate 0.01%–TAZ 0.045% lotion was significantly more effective than vehicle for all efficacy assessments. In general, efficacy results with HP/TAZ lotion were similar to those reported in the overall phase 3 study populations over the 8-week treatment period. The only noticeable difference was in the posttreatment period. In the overall study population, efficacy was maintained over the 4-week posttreatment period in the HP/TAZ group. In the Hispanic subpopulation, there appeared to be continued improvement in the number of participants achieving treatment success (IGA and erythema), clinically meaningful success, and further reductions in BSA. Although there is a paucity of studies evaluating psoriasis therapies in Hispanic populations, data on etanercept and secukinumab have been published.6,11
Onset of effect also is an important aspect of treatment. In patients with skin of color, including patients of Hispanic ethnicity and higher Fitzpatrick skin phototypes, early clearance of lesions may help limit the severity and duration of postinflammatory pigment alteration. Improvements in IGA×BSA scores were significant compared to vehicle from week 2 (P=.016), and a clinically meaningful improvement with HP/TAZ lotion (IGA×BSA-75) was seen by week 4 (P=.024).
Halobetasol propionate 0.01%–TAZ 0.045% lotion was well tolerated, both in the 2 phase 3 studies and in the post hoc analysis of the Hispanic subpopulation. The incidence of skin atrophy (n=3, 3.9%) was more common vs the non-Hispanic population (n=2, 1.0%). Other common AEs—contact dermatitis, pruritus, and application-site pain—were more common in the non-Hispanic population.
A limitation of our analysis was that it was a post hoc analysis of the Hispanic participants. The phase 3 studies were not designed to specifically study the impact of treatment on ethnicity/race, though the number of Hispanic participants enrolled in the 2 studies was relatively high. The absence of Fitzpatrick skin phototypes in this data set is another limitation of this study.
Conclusion
Halobetasol propionate 0.01%–TAZ 0.045% lotion was associated with significant, rapid, and sustained reductions in disease severity in a Hispanic population with moderate to severe psoriasis that continued to show improvement posttreatment with good tolerability and safety.
Acknowledgments
We thank Brian Bulley, MSc (Konic Limited, United Kingdom), for assistance with the preparation of the manuscript. Ortho Dermatologics funded Konic’s activities pertaining to this manuscript.
Psoriasis is a common chronic inflammatory disease affecting a diverse patient population, yet epidemiological and clinical data related to psoriasis in patients with skin of color are sparse. The Hispanic ethnic group includes a broad range of skin types and cultures. Prevalence of psoriasis in a Hispanic population has been reported as lower than in a white population1; however, these data may be influenced by the finding that Hispanic patients are less likely to see a dermatologist when they have skin problems.2 In addition, socioeconomic disparities and cultural variations among racial/ethnic groups may contribute to differences in access to care and thresholds for seeking care,3 leading to a tendency for more severe disease in skin of color and Hispanic ethnic groups.4,5 Greater impairments in health-related quality of life have been reported in patients with skin of color and Hispanic racial/ethnic groups compared to white patients, independent of psoriasis severity.4,6 Postinflammatory pigment alteration at the sites of resolving lesions, a common clinical feature in skin of color, may contribute to the impact of psoriasis on quality of life in patients with skin of color. Psoriasis in darker skin types also can present diagnostic challenges due to overlapping features with other papulosquamous disorders and less conspicuous erythema.7
We present a post hoc analysis of the treatment of moderate to severe psoriasis with a novel fixed-combination halobetasol propionate (HP) 0.01%–tazarotene (TAZ) 0.045% lotion in a Hispanic patient population. Historically, clinical trials for psoriasis have enrolled low proportions of Hispanic patients and other patients with skin of color; in this analysis, the Hispanic population (115/418) represented 28% of the total study population and provided valuable insights.
Methods
Study Design
Two phase 3 randomized controlled trials were conducted to demonstrate the efficacy and safety of HP/TAZ lotion. Patients with a clinical diagnosis of moderate or severe localized psoriasis (N=418) were randomized to receive HP/TAZ lotion or vehicle (2:1 ratio) once daily for 8 weeks with a 4-week posttreatment follow-up.8,9 A post hoc analysis was conducted on data of the self-identified Hispanic population.
Assessments
Efficacy assessments included treatment success (at least a 2-grade improvement from baseline in the investigator global assessment [IGA] and a score of clear or almost clear) and impact on individual signs of psoriasis (at least a 2-grade improvement in erythema, plaque elevation, and scaling) at the target lesion. In addition, reduction in body surface area (BSA) was recorded, and an IGA×BSA score was calculated by multiplying IGA by BSA at each timepoint for each individual patient. A clinically meaningful improvement in disease severity (percentage of patients achieving a 75% reduction in IGA×BSA [IGA×BSA-75]) also was calculated.
Information on reported and observed adverse events (AEs) was obtained at each visit. The safety population included 112 participants (76 in the HP/TAZ group and 36 in the vehicle group).
Statistical Analysis
The statistical and analytical plan is detailed elsewhere9 and relevant to this post hoc analysis. No statistical analysis was carried out to compare data in the Hispanic population with either the overall study population or the non-Hispanic population.
Results
Overall, 115 Hispanic patients (27.5%) were enrolled (eFigure). Patients had a mean (standard deviation [SD]) age of 46.7 (13.12) years, and more than two-thirds were male (n=80, 69.6%).

Overall completion rates (80.0%) for Hispanic patients were similar to those in the overall study population, though there were more discontinuations in the vehicle group. The main reasons for treatment discontinuation among Hispanic patients were participant request (n=8, 7.0%), lost to follow-up (n=8, 7.0%), and AEs (n=4, 3.5%). Hispanic patients in this study had more severe disease—18.3% (n=21) had an IGA score of 4 compared to 13.5% (n=41) of non-Hispanic patients—and more severe erythema (19.1% vs 9.6%), plaque elevation (20.0% vs 10.2%), and scaling (15.7% vs 12.9%) compared to the non-Hispanic populations (Table).

Efficacy of HP/TAZ lotion in Hispanic patients was similar to the overall study populations,9 though maintenance of effect posttreatment appeared to be better. The incidence of treatment-related AEs also was lower.
Halobetasol propionate 0.01%–TAZ 0.045% lotion demonstrated statistically significant superiority based on treatment success compared to vehicle as early as week 4 (P=.034). By week 8, 39.3% of participants treated with HP/TAZ lotion achieved treatment success compared to 9.3% of participants in the vehicle group (P=.002)(Figure 1). Treatment success was maintained over the 4-week posttreatment period, whereby 40.5% of the HP/TAZ-treated participants were treatment successes at week 12 compared to only 4.1% of participants in the vehicle group (P<.001).

Improvements in psoriasis signs and symptoms at the target lesion were statistically significant compared to vehicle from week 2 (plaque elevation, P=.018) or week 4 (erythema, P=.004; scaling, P<.001)(Figure 2). By week 8, 46.8%, 58.1%, and 63.2% of participants showed at least a 2-grade improvement from baseline and were therefore treatment successes for erythema, plaque elevation, and scaling, respectively (all statistically significant [P<.001] compared to vehicle). The number of participants who achieved at least a 2-grade improvement in erythema with HP/TAZ lotion increased posttreatment from 46.8% to 53.0%.

Mean (SD) baseline BSA was 6.2 (3.07), and the mean (SD) size of the target lesion was 36.3 (21.85) cm2. Overall, BSA also was significantly reduced in participants treated with HP/TAZ lotion compared to vehicle. At week 8, the mean percentage change from baseline was —40.7% compared to an increase (+10.1%) in the vehicle group (P=.002)(Figure 3). Improvements in BSA were maintained posttreatment, whereas in the vehicle group, mean (SD) BSA had increased to 6.1 (4.64).

Halobetasol propionate 0.01%–TAZ 0.045% lotion achieved a 50.5% reduction from baseline IGA×BSA by week 8 compared to an 8.5% increase with vehicle (P<.001)(Figure 4). Differences in treatment groups were significant from week 2 (P=.016). Efficacy was maintained posttreatment, with a 50.6% reduction from baseline IGA×BSA at week 12 compared to an increase of 13.6% in the vehicle group (P<.001). Again, although results were similar to the overall study population at week 8 (50.5% vs 51.9%), maintenance of effect was better posttreatment (50.6% vs 46.6%).10

A clinically meaningful effect (IGA×BSA-75) was achieved in 39.7% of Hispanic participants treated with HP/TAZ lotion compared to 8.1% of participants treated with vehicle (P<.001) at week 8. The benefits were significantly different from week 4 and more participants maintained a clinically meaningful effect posttreatment (43.1% vs 7.1%, P<.001)(Figure 5).

For Hispanic participants overall, 34 participants reported AEs: 26 (34.2%) treated with HP/TAZ lotion and 8 (22.2%) treated with vehicle (eTable). There was 1 (1.3%) serious AE in the HP/TAZ group. Most of the AEs were mild or moderate, with approximately half being related to study treatment. The most common treatment-related AEs in Hispanic participants treated with HP/TAZ lotion were contact dermatitis (n=3, 3.9%) and skin atrophy (n=3, 3.9%) compared to contact dermatitis (n=14, 7.2%) and application-site pain (n=7, 3.6%) in the non-Hispanic population. Pruritus was the most common AE in Hispanic participants treated with vehicle.

Comment
The large number of Hispanic patients in the 2 phase 3 trials8,9 allowed for this valuable subgroup analysis on the topical treatment of Hispanic patients with plaque psoriasis. Validation of observed differences in maintenance of effect and tolerability warrant further study. Prior clinical studies in psoriasis have tended to enroll a small proportion of Hispanic patients without any post hoc analysis. For example, in a pooled analysis of 4 phase 3 trials with secukinumab, Hispanic patients accounted for only 16% of the overall population.11 In our analysis, the Hispanic cohort represented 28% of the overall study population of 2 phase 3 studies investigating the efficacy, safety, and tolerability of HP/TAZ lotion in patients with moderate to severe psoriasis.8,9 In addition, proportionately more Hispanic patients had severe disease (IGA of 4) or severe signs and symptoms of psoriasis (erythema, plaque elevation, and scaling) than the non-Hispanic population. This finding supports other studies that have suggested Hispanic patients with psoriasis tend to have more severe disease but also may reflect thresholds for seeking care.3-5
Halobetasol propionate 0.01%–TAZ 0.045% lotion was significantly more effective than vehicle for all efficacy assessments. In general, efficacy results with HP/TAZ lotion were similar to those reported in the overall phase 3 study populations over the 8-week treatment period. The only noticeable difference was in the posttreatment period. In the overall study population, efficacy was maintained over the 4-week posttreatment period in the HP/TAZ group. In the Hispanic subpopulation, there appeared to be continued improvement in the number of participants achieving treatment success (IGA and erythema), clinically meaningful success, and further reductions in BSA. Although there is a paucity of studies evaluating psoriasis therapies in Hispanic populations, data on etanercept and secukinumab have been published.6,11
Onset of effect also is an important aspect of treatment. In patients with skin of color, including patients of Hispanic ethnicity and higher Fitzpatrick skin phototypes, early clearance of lesions may help limit the severity and duration of postinflammatory pigment alteration. Improvements in IGA×BSA scores were significant compared to vehicle from week 2 (P=.016), and a clinically meaningful improvement with HP/TAZ lotion (IGA×BSA-75) was seen by week 4 (P=.024).
Halobetasol propionate 0.01%–TAZ 0.045% lotion was well tolerated, both in the 2 phase 3 studies and in the post hoc analysis of the Hispanic subpopulation. The incidence of skin atrophy (n=3, 3.9%) was more common vs the non-Hispanic population (n=2, 1.0%). Other common AEs—contact dermatitis, pruritus, and application-site pain—were more common in the non-Hispanic population.
A limitation of our analysis was that it was a post hoc analysis of the Hispanic participants. The phase 3 studies were not designed to specifically study the impact of treatment on ethnicity/race, though the number of Hispanic participants enrolled in the 2 studies was relatively high. The absence of Fitzpatrick skin phototypes in this data set is another limitation of this study.
Conclusion
Halobetasol propionate 0.01%–TAZ 0.045% lotion was associated with significant, rapid, and sustained reductions in disease severity in a Hispanic population with moderate to severe psoriasis that continued to show improvement posttreatment with good tolerability and safety.
Acknowledgments
We thank Brian Bulley, MSc (Konic Limited, United Kingdom), for assistance with the preparation of the manuscript. Ortho Dermatologics funded Konic’s activities pertaining to this manuscript.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
- Setta-Kaffetzi N, Navarini AA, Patel VM, et al. Rare pathogenic variants in IL36RN underlie a spectrum of psoriasis-associated pustular phenotypes. J Invest Dermatol. 2013;133:1366-1369.
- Yan D, Afifi L, Jeon C, et al. A cross-sectional study of the distribution of psoriasis subtypes in different ethno-racial groups. Dermatol Online J. 2018;24. pii:13030/qt5z21q4k2.
- Abrouk M, Lee K, Brodsky M, et al. Ethnicity affects the presenting severity of psoriasis. J Am Acad Dermatol. 2017;77:180-182.
- Shah SK, Arthur A, Yang YC, et al. A retrospective study to investigate racial and ethnic variations in the treatment of psoriasis with etanercept. J Drugs Dermatol. 2011;10:866-872.
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Blauvelt A, Green LJ, Lebwohl MG, et al. Efficacy of a once-daily fixed combination halobetasol (0.01%) and tazarotene (0.045%) lotion in the treatment of localized moderate-to-severe plaque psoriasis. J Drugs Dermatol. 2019;18:297-299.
- Adsit S, Zaldivar ER, Sofen H, et al. Secukinumab is efficacious and safe in Hispanic patients with moderate-to-severe plaque psoriasis: pooled analysis of four phase 3 trials. Adv Ther. 2017;34:1327-1339.
- Rachakonda TD, Schupp CW, Armstrong AW. Psoriasis prevalence among adults in the United States. J Am Acad Dermatol. 2014;70:512-516.
- Davis SA, Narahari S, Feldman SR, et al. Top dermatologic conditions in patients of color: an analysis of nationally representative data. J Drugs Dermatol. 2012;11:466-473.
- Setta-Kaffetzi N, Navarini AA, Patel VM, et al. Rare pathogenic variants in IL36RN underlie a spectrum of psoriasis-associated pustular phenotypes. J Invest Dermatol. 2013;133:1366-1369.
- Yan D, Afifi L, Jeon C, et al. A cross-sectional study of the distribution of psoriasis subtypes in different ethno-racial groups. Dermatol Online J. 2018;24. pii:13030/qt5z21q4k2.
- Abrouk M, Lee K, Brodsky M, et al. Ethnicity affects the presenting severity of psoriasis. J Am Acad Dermatol. 2017;77:180-182.
- Shah SK, Arthur A, Yang YC, et al. A retrospective study to investigate racial and ethnic variations in the treatment of psoriasis with etanercept. J Drugs Dermatol. 2011;10:866-872.
- Alexis AF, Blackcloud P. Psoriasis in skin of color: epidemiology, genetics, clinical presentation, and treatment nuances. J Clin Aesthet Dermatol. 2014;7:16-24.
- Gold LS, Lebwohl MG, Sugarman JL, et al. Safety and efficacy of a fixed combination of halobetasol and tazarotene in the treatment of moderate-to-severe plaque psoriasis: results of 2 phase 3 randomized controlled trials. J Am Acad Dermatol. 2018;79:287-293.
- Sugarman JL, Weiss J, Tanghetti EA, et al. Safety and efficacy of a fixed combination halobetasol and tazarotene lotion in the treatment of moderate-to-severe plaque psoriasis: a pooled analysis of two phase 3 studies. J Drugs Dermatol. 2018;17:855-861.
- Blauvelt A, Green LJ, Lebwohl MG, et al. Efficacy of a once-daily fixed combination halobetasol (0.01%) and tazarotene (0.045%) lotion in the treatment of localized moderate-to-severe plaque psoriasis. J Drugs Dermatol. 2019;18:297-299.
- Adsit S, Zaldivar ER, Sofen H, et al. Secukinumab is efficacious and safe in Hispanic patients with moderate-to-severe plaque psoriasis: pooled analysis of four phase 3 trials. Adv Ther. 2017;34:1327-1339.
Practice Points
- Although psoriasis is a common inflammatory disease, data in the Hispanic population are sparse and disease may be more severe.
- A recent clinical investigation with halobetasol propionate 0.01%–tazarotene 0.045% lotion included a number of Hispanic patients, affording an ideal opportunity to provide important data on this population.
- This fixed-combination therapy was associated with significant, rapid, and sustained reductions in disease severity in a Hispanic population with moderate to severe psoriasis that continued to show improvement posttreatment with good tolerability and safety.
Keep your eye on tapinarof, a topical antipsoriatic therapy
LAHAINA, HAWAII – Tapinarof is an investigational drug whose novel mechanism of action – and encouraging performance in phase 2 studies – are making waves for the topical treatment of both psoriasis and atopic dermatitis, Linda F. Stein Gold, MD, observed at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Tapinarof is a first-in-class agonist of the aryl hydrocarbon receptor.
“An aryl hydrocarbon receptor agonist – what in the world does that mean? It means that this drug actually acts at the receptor level inside the cell, and it does a lot of different things,” explained Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Health System in Detroit.
For one, tapinarof down-regulates Th17 cytokines, an attribute that positions the drug very well as a potential topical treatment for psoriasis. But in addition, the drug has a skin barrier repair element through up-regulation of the filaggrin and involucrin genes in keratinocytes, and it also down-regulates Th2 cytokines, actions desirable in a treatment for atopic dermatitis.
Dr. Stein Gold focused mainly on tapinarof’s potential as a novel treatment for psoriasis, a disease that hasn’t seen approval of a new nonsteroidal topical therapy in decades. There is a huge unmet need for safe and effective new topical therapies for this disease; despite all the attention devoted to biologics and other systemic therapies, the great majority of psoriasis patients are managed via topical therapy only.
The definitive trial was initiated based upon the results of a phase 2b, double-blind, six-arm study including 141 adults with body surface involvement of 1%-15% and a baseline Physician Global Assessment (PGA) score of 2 or more who were assigned to tapinarof at 0.5% or 1% once or twice daily or placebo. The phase 2b results, she commented, were very encouraging.
“When we look at the clinical efficacy, it looks like this drug has legs. It does work even as monotherapy to get patients clear,” she said.
The phase 2b, dose-finding study showed dose-dependent treatment efficacy. At week 12, the proportion of participants with a PGA of 0-1 and at least a 2-grade improvement – that is, clear or almost clear – was 36% with tapinarof monotherapy at 0.5% once daily, 46% with 0.5% twice daily, 56% with 1% once daily, and 65% with 1% twice daily, compared with 5% in controls on once-daily application of vehicle and 11% with twice-daily vehicle. Moreover, the improvement was maintained for 4 weeks post treatment. The drug was well tolerated other than some mild to moderate folliculitis and contact dermatitis (J Am Acad Dermatol. 2019 Mar;80[3]:714-21).
“With such small numbers in phase 2, we don’t necessarily need to see statistical significance, but we want to see a trend in the right direction. But every one of the active treatment arms was statistically significantly better than with vehicle. And at higher concentrations, greater efficacy,” noted Dr. Stein Gold.
A phase 2 study of tapinarof cream has also been completed in adults and adolescents with atopic dermatitis, again with positive results. A phase 3 study in atopic dermatitis is still in the planning stages.
Dr. Stein Gold wasn’t involved in the tapinarof psoriasis phase 2b study, sponsored by GlaxoSmithKline. She reported research funding from nine other pharmaceutical companies and serves as a consultant and/or scientific to more than a dozen companies.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Tapinarof is an investigational drug whose novel mechanism of action – and encouraging performance in phase 2 studies – are making waves for the topical treatment of both psoriasis and atopic dermatitis, Linda F. Stein Gold, MD, observed at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Tapinarof is a first-in-class agonist of the aryl hydrocarbon receptor.
“An aryl hydrocarbon receptor agonist – what in the world does that mean? It means that this drug actually acts at the receptor level inside the cell, and it does a lot of different things,” explained Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Health System in Detroit.
For one, tapinarof down-regulates Th17 cytokines, an attribute that positions the drug very well as a potential topical treatment for psoriasis. But in addition, the drug has a skin barrier repair element through up-regulation of the filaggrin and involucrin genes in keratinocytes, and it also down-regulates Th2 cytokines, actions desirable in a treatment for atopic dermatitis.
Dr. Stein Gold focused mainly on tapinarof’s potential as a novel treatment for psoriasis, a disease that hasn’t seen approval of a new nonsteroidal topical therapy in decades. There is a huge unmet need for safe and effective new topical therapies for this disease; despite all the attention devoted to biologics and other systemic therapies, the great majority of psoriasis patients are managed via topical therapy only.
The definitive trial was initiated based upon the results of a phase 2b, double-blind, six-arm study including 141 adults with body surface involvement of 1%-15% and a baseline Physician Global Assessment (PGA) score of 2 or more who were assigned to tapinarof at 0.5% or 1% once or twice daily or placebo. The phase 2b results, she commented, were very encouraging.
“When we look at the clinical efficacy, it looks like this drug has legs. It does work even as monotherapy to get patients clear,” she said.
The phase 2b, dose-finding study showed dose-dependent treatment efficacy. At week 12, the proportion of participants with a PGA of 0-1 and at least a 2-grade improvement – that is, clear or almost clear – was 36% with tapinarof monotherapy at 0.5% once daily, 46% with 0.5% twice daily, 56% with 1% once daily, and 65% with 1% twice daily, compared with 5% in controls on once-daily application of vehicle and 11% with twice-daily vehicle. Moreover, the improvement was maintained for 4 weeks post treatment. The drug was well tolerated other than some mild to moderate folliculitis and contact dermatitis (J Am Acad Dermatol. 2019 Mar;80[3]:714-21).
“With such small numbers in phase 2, we don’t necessarily need to see statistical significance, but we want to see a trend in the right direction. But every one of the active treatment arms was statistically significantly better than with vehicle. And at higher concentrations, greater efficacy,” noted Dr. Stein Gold.
A phase 2 study of tapinarof cream has also been completed in adults and adolescents with atopic dermatitis, again with positive results. A phase 3 study in atopic dermatitis is still in the planning stages.
Dr. Stein Gold wasn’t involved in the tapinarof psoriasis phase 2b study, sponsored by GlaxoSmithKline. She reported research funding from nine other pharmaceutical companies and serves as a consultant and/or scientific to more than a dozen companies.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
LAHAINA, HAWAII – Tapinarof is an investigational drug whose novel mechanism of action – and encouraging performance in phase 2 studies – are making waves for the topical treatment of both psoriasis and atopic dermatitis, Linda F. Stein Gold, MD, observed at the Hawaii Dermatology Seminar provided by the Global Academy for Medical Education/Skin Disease Education Foundation.
Tapinarof is a first-in-class agonist of the aryl hydrocarbon receptor.
“An aryl hydrocarbon receptor agonist – what in the world does that mean? It means that this drug actually acts at the receptor level inside the cell, and it does a lot of different things,” explained Dr. Stein Gold, director of dermatology clinical research at the Henry Ford Health System in Detroit.
For one, tapinarof down-regulates Th17 cytokines, an attribute that positions the drug very well as a potential topical treatment for psoriasis. But in addition, the drug has a skin barrier repair element through up-regulation of the filaggrin and involucrin genes in keratinocytes, and it also down-regulates Th2 cytokines, actions desirable in a treatment for atopic dermatitis.
Dr. Stein Gold focused mainly on tapinarof’s potential as a novel treatment for psoriasis, a disease that hasn’t seen approval of a new nonsteroidal topical therapy in decades. There is a huge unmet need for safe and effective new topical therapies for this disease; despite all the attention devoted to biologics and other systemic therapies, the great majority of psoriasis patients are managed via topical therapy only.
The definitive trial was initiated based upon the results of a phase 2b, double-blind, six-arm study including 141 adults with body surface involvement of 1%-15% and a baseline Physician Global Assessment (PGA) score of 2 or more who were assigned to tapinarof at 0.5% or 1% once or twice daily or placebo. The phase 2b results, she commented, were very encouraging.
“When we look at the clinical efficacy, it looks like this drug has legs. It does work even as monotherapy to get patients clear,” she said.
The phase 2b, dose-finding study showed dose-dependent treatment efficacy. At week 12, the proportion of participants with a PGA of 0-1 and at least a 2-grade improvement – that is, clear or almost clear – was 36% with tapinarof monotherapy at 0.5% once daily, 46% with 0.5% twice daily, 56% with 1% once daily, and 65% with 1% twice daily, compared with 5% in controls on once-daily application of vehicle and 11% with twice-daily vehicle. Moreover, the improvement was maintained for 4 weeks post treatment. The drug was well tolerated other than some mild to moderate folliculitis and contact dermatitis (J Am Acad Dermatol. 2019 Mar;80[3]:714-21).
“With such small numbers in phase 2, we don’t necessarily need to see statistical significance, but we want to see a trend in the right direction. But every one of the active treatment arms was statistically significantly better than with vehicle. And at higher concentrations, greater efficacy,” noted Dr. Stein Gold.
A phase 2 study of tapinarof cream has also been completed in adults and adolescents with atopic dermatitis, again with positive results. A phase 3 study in atopic dermatitis is still in the planning stages.
Dr. Stein Gold wasn’t involved in the tapinarof psoriasis phase 2b study, sponsored by GlaxoSmithKline. She reported research funding from nine other pharmaceutical companies and serves as a consultant and/or scientific to more than a dozen companies.
SDEF/Global Academy for Medical Education and this news organization are owned by the same parent company.
REPORTING FROM THE SDEF HAWAII DERMATOLOGY SEMINAR
Specific markers detect psoriatic disease inflammation without elevated CRP
according to a cross-sectional study of patients and healthy controls.
“Different clinical subsets of psoriatic disease based on skin, entheseal, and joint involvement are characterized by specific inflammation marker profiles,” Maria V. Sokolova, MD, of Friedrich-Alexander University Erlangen-Nuremberg and University Clinic Erlangen (Germany) and colleagues reported in Arthritis Research & Therapy. “Treatment of psoriatic disease with cytokine inhibitors reduces these elevated levels of systemic inflammation markers.”
Quantifying systemic inflammation in psoriatic disease has been a challenge, Dr. Sokolova and colleagues wrote. Levels of C-reactive protein (CRP), a commonly used measure of systemic inflammation, “are often low or absent.” To examine other potential markers of systemic inflammation in psoriatic disease, the investigators conducted cross-sectional and longitudinal studies that included healthy controls and patients with psoriatic disease. Patients had isolated or combined manifestations of psoriatic disease, including the skin, the entheses, and the joints. The researchers grouped patients by isolated psoriatic skin disease; isolated enthesitis; isolated arthritis; psoriatic skin disease with enthesitis; psoriatic skin disease with arthritis; arthritis and enthesitis; and combined psoriatic skin disease, arthritis, and enthesitis.
Data from more than 100 patients
The researchers first assessed 10 potential markers using enzyme-linked immunosorbent assay: calprotectin, interleukin-22, IL-8, lipocalin 2, beta-defensin 2, IL-17, IL-23, vascular endothelial growth factors, LL37 (cathelicidin), and pentraxin 3. They measured the markers in 10 healthy controls and 10 patients with active polymorphic psoriatic arthritis. Five parameters – beta-defensin 2, lipocalin 2, IL-22, IL-8, and calprotectin – significantly differed between healthy controls and patients with psoriatic disease. Lipocalin 2, beta-defensin 2, and IL-22 are associated with IL-17/IL-23 activation, and calprotectin and IL-8 are associated with innate immune cell activation. The other markers did not significantly differ or were not detectable in enough participants.
To validate the signals, the researchers measured the five parameters as well as CRP in 105 controls and 105 patients with psoriatic disease, including 15 patients in each of the seven disease pattern groups. “As expected, CRP levels were normal in the majority of individuals,” the authors wrote. The proportion of patients with CRP greater than 5 mg/L was 0% in isolated psoriatic skin disease, 0% in isolated enthesitis; 20% in isolated arthritis; 7% in psoriatic skin disease with enthesitis; 33% in psoriatic skin disease with arthritis; 27% in arthritis with enthesitis; and 33% in combined psoriatic skin disease, arthritis, and enthesitis.
“Only a subset of patients with arthritis, but not patients with skin or entheseal disease show elevated CRP,” the researchers wrote. “In sharp contrast,” beta-defensin 2 and lipocalin 2 were elevated in a majority of patients with monomorphic skin and entheseal disease, but not in joint disease. “Both proteins were significantly correlated to the extent of skin disease and to a lesser extent also entheseal disease,” they added. Calprotectin and IL-8 were elevated in a majority of patients with joint disease and correlated with the extent of arthritis. “IL-22 was elevated ... in all three manifestations of psoriatic disease,” and the vast majority of patients with polymorphic disease had “widespread marker elevation,” the researchers wrote.
Effects of treatment
In a study of 20 patients with psoriatic arthritis, treatment with secukinumab or adalimumab significantly lowered all five markers. Compared with tumor necrosis factor inhibition with adalimumab, “IL-17 inhibition [with secukinumab] showed a more pronounced lowering of lipocalin 2 and beta-defensin 2 levels,” the investigators noted.
“These results confirm earlier data showing elevated beta-defensin levels in psoriasis patients and its association with the extent of skin involvement,” Dr. Sokolova and colleagues wrote. “Overall, these results offer a new possibility to measure systemic inflammation in psoriatic disease.”
The study was supported by the German Research Foundation and other grant and fellowship funding. The authors had no competing interests.
SOURCE: Sokolova MV et al. Arthritis Res Ther. 2020;22:26.
according to a cross-sectional study of patients and healthy controls.
“Different clinical subsets of psoriatic disease based on skin, entheseal, and joint involvement are characterized by specific inflammation marker profiles,” Maria V. Sokolova, MD, of Friedrich-Alexander University Erlangen-Nuremberg and University Clinic Erlangen (Germany) and colleagues reported in Arthritis Research & Therapy. “Treatment of psoriatic disease with cytokine inhibitors reduces these elevated levels of systemic inflammation markers.”
Quantifying systemic inflammation in psoriatic disease has been a challenge, Dr. Sokolova and colleagues wrote. Levels of C-reactive protein (CRP), a commonly used measure of systemic inflammation, “are often low or absent.” To examine other potential markers of systemic inflammation in psoriatic disease, the investigators conducted cross-sectional and longitudinal studies that included healthy controls and patients with psoriatic disease. Patients had isolated or combined manifestations of psoriatic disease, including the skin, the entheses, and the joints. The researchers grouped patients by isolated psoriatic skin disease; isolated enthesitis; isolated arthritis; psoriatic skin disease with enthesitis; psoriatic skin disease with arthritis; arthritis and enthesitis; and combined psoriatic skin disease, arthritis, and enthesitis.
Data from more than 100 patients
The researchers first assessed 10 potential markers using enzyme-linked immunosorbent assay: calprotectin, interleukin-22, IL-8, lipocalin 2, beta-defensin 2, IL-17, IL-23, vascular endothelial growth factors, LL37 (cathelicidin), and pentraxin 3. They measured the markers in 10 healthy controls and 10 patients with active polymorphic psoriatic arthritis. Five parameters – beta-defensin 2, lipocalin 2, IL-22, IL-8, and calprotectin – significantly differed between healthy controls and patients with psoriatic disease. Lipocalin 2, beta-defensin 2, and IL-22 are associated with IL-17/IL-23 activation, and calprotectin and IL-8 are associated with innate immune cell activation. The other markers did not significantly differ or were not detectable in enough participants.
To validate the signals, the researchers measured the five parameters as well as CRP in 105 controls and 105 patients with psoriatic disease, including 15 patients in each of the seven disease pattern groups. “As expected, CRP levels were normal in the majority of individuals,” the authors wrote. The proportion of patients with CRP greater than 5 mg/L was 0% in isolated psoriatic skin disease, 0% in isolated enthesitis; 20% in isolated arthritis; 7% in psoriatic skin disease with enthesitis; 33% in psoriatic skin disease with arthritis; 27% in arthritis with enthesitis; and 33% in combined psoriatic skin disease, arthritis, and enthesitis.
“Only a subset of patients with arthritis, but not patients with skin or entheseal disease show elevated CRP,” the researchers wrote. “In sharp contrast,” beta-defensin 2 and lipocalin 2 were elevated in a majority of patients with monomorphic skin and entheseal disease, but not in joint disease. “Both proteins were significantly correlated to the extent of skin disease and to a lesser extent also entheseal disease,” they added. Calprotectin and IL-8 were elevated in a majority of patients with joint disease and correlated with the extent of arthritis. “IL-22 was elevated ... in all three manifestations of psoriatic disease,” and the vast majority of patients with polymorphic disease had “widespread marker elevation,” the researchers wrote.
Effects of treatment
In a study of 20 patients with psoriatic arthritis, treatment with secukinumab or adalimumab significantly lowered all five markers. Compared with tumor necrosis factor inhibition with adalimumab, “IL-17 inhibition [with secukinumab] showed a more pronounced lowering of lipocalin 2 and beta-defensin 2 levels,” the investigators noted.
“These results confirm earlier data showing elevated beta-defensin levels in psoriasis patients and its association with the extent of skin involvement,” Dr. Sokolova and colleagues wrote. “Overall, these results offer a new possibility to measure systemic inflammation in psoriatic disease.”
The study was supported by the German Research Foundation and other grant and fellowship funding. The authors had no competing interests.
SOURCE: Sokolova MV et al. Arthritis Res Ther. 2020;22:26.
according to a cross-sectional study of patients and healthy controls.
“Different clinical subsets of psoriatic disease based on skin, entheseal, and joint involvement are characterized by specific inflammation marker profiles,” Maria V. Sokolova, MD, of Friedrich-Alexander University Erlangen-Nuremberg and University Clinic Erlangen (Germany) and colleagues reported in Arthritis Research & Therapy. “Treatment of psoriatic disease with cytokine inhibitors reduces these elevated levels of systemic inflammation markers.”
Quantifying systemic inflammation in psoriatic disease has been a challenge, Dr. Sokolova and colleagues wrote. Levels of C-reactive protein (CRP), a commonly used measure of systemic inflammation, “are often low or absent.” To examine other potential markers of systemic inflammation in psoriatic disease, the investigators conducted cross-sectional and longitudinal studies that included healthy controls and patients with psoriatic disease. Patients had isolated or combined manifestations of psoriatic disease, including the skin, the entheses, and the joints. The researchers grouped patients by isolated psoriatic skin disease; isolated enthesitis; isolated arthritis; psoriatic skin disease with enthesitis; psoriatic skin disease with arthritis; arthritis and enthesitis; and combined psoriatic skin disease, arthritis, and enthesitis.
Data from more than 100 patients
The researchers first assessed 10 potential markers using enzyme-linked immunosorbent assay: calprotectin, interleukin-22, IL-8, lipocalin 2, beta-defensin 2, IL-17, IL-23, vascular endothelial growth factors, LL37 (cathelicidin), and pentraxin 3. They measured the markers in 10 healthy controls and 10 patients with active polymorphic psoriatic arthritis. Five parameters – beta-defensin 2, lipocalin 2, IL-22, IL-8, and calprotectin – significantly differed between healthy controls and patients with psoriatic disease. Lipocalin 2, beta-defensin 2, and IL-22 are associated with IL-17/IL-23 activation, and calprotectin and IL-8 are associated with innate immune cell activation. The other markers did not significantly differ or were not detectable in enough participants.
To validate the signals, the researchers measured the five parameters as well as CRP in 105 controls and 105 patients with psoriatic disease, including 15 patients in each of the seven disease pattern groups. “As expected, CRP levels were normal in the majority of individuals,” the authors wrote. The proportion of patients with CRP greater than 5 mg/L was 0% in isolated psoriatic skin disease, 0% in isolated enthesitis; 20% in isolated arthritis; 7% in psoriatic skin disease with enthesitis; 33% in psoriatic skin disease with arthritis; 27% in arthritis with enthesitis; and 33% in combined psoriatic skin disease, arthritis, and enthesitis.
“Only a subset of patients with arthritis, but not patients with skin or entheseal disease show elevated CRP,” the researchers wrote. “In sharp contrast,” beta-defensin 2 and lipocalin 2 were elevated in a majority of patients with monomorphic skin and entheseal disease, but not in joint disease. “Both proteins were significantly correlated to the extent of skin disease and to a lesser extent also entheseal disease,” they added. Calprotectin and IL-8 were elevated in a majority of patients with joint disease and correlated with the extent of arthritis. “IL-22 was elevated ... in all three manifestations of psoriatic disease,” and the vast majority of patients with polymorphic disease had “widespread marker elevation,” the researchers wrote.
Effects of treatment
In a study of 20 patients with psoriatic arthritis, treatment with secukinumab or adalimumab significantly lowered all five markers. Compared with tumor necrosis factor inhibition with adalimumab, “IL-17 inhibition [with secukinumab] showed a more pronounced lowering of lipocalin 2 and beta-defensin 2 levels,” the investigators noted.
“These results confirm earlier data showing elevated beta-defensin levels in psoriasis patients and its association with the extent of skin involvement,” Dr. Sokolova and colleagues wrote. “Overall, these results offer a new possibility to measure systemic inflammation in psoriatic disease.”
The study was supported by the German Research Foundation and other grant and fellowship funding. The authors had no competing interests.
SOURCE: Sokolova MV et al. Arthritis Res Ther. 2020;22:26.
FROM ARTHRITIS RESEARCH & THERAPY