User login
Ingenol mebutate improves outcomes in actinic keratosis
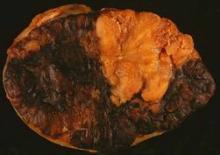
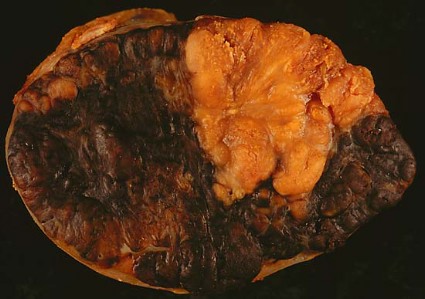
Sequential topical field-directed therapy with ingenol mebutate gel for 3 days following cryosurgery significantly improves the outcomes in patients with actinic keratosis, a phase III randomized study showed.
Enrolled patients had four to eight clinically typical lesions on the face or scalp and received cryosurgery to all visible lesions followed by once-daily treatment of ingenol mebutate 0.015% gel or vehicle gel for 3 consecutive days at home, after 3 weeks of healing.
At 11 weeks, patients who received treatment with ingenol mebutate gel (n = 167) postsurgery, saw higher rates of complete clearance of lesions, compared with patients receiving the vehicle alone after surgery (n = 162) (60.5% vs. 49.4%; P=.04), reported Dr. Stephen K. Tyring and his associates this month (J. Drugs Dermatol. 2014;13:154-60).
In longer-term follow-up at 12 months, ingenol mebutate gel continued to show significantly improved complete clearance rates, compared with vehicle gel (30.5% vs. 18.5%; P = .01), Dr. Tyring reported at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
The mean percentage reduction of lesions at 12 months was significantly higher with ingenol mebutate for all lesions (59.5% vs 44.4%, P=0.004) and 38.9% of patients receiving ingenol mebutate saw emergence of new lesions on the treatment area between baseline lesions, compared with 51.9% of patients in the vehicle group (P=0.01), he said.
The complete clearance at 11 weeks (60.5% vs 49.4%; P=0.04) with a relative complete actinic keratosis clearance ratio of 1.22 (1.01-1.49) increased to 1.67 (1.12-2.50) at 12 months in the treatment group vs vehicle, following cryosurgery, noted Dr. Tyring of University of Texas Health Science Center at Houston.
The most frequently reported adverse events in the treatment group were application site discomfort and pruritus. Over the 12-month follow-up, 16 patients in the ingenol mebutate group reported one or more adverse events, as did 6 in the vehicle group. These events, however, were minor and well tolerated, said Dr. Tyring.
Ingenol mebutate gel was approved by the Food and Drug Administration in 2012 to treat actinic keratosis. Its advantage over other topical field agents such as 5-fluorouracil, imiquimod, and diclofenac is its short treatment period of 2-3 days, depending on the treatment area. This reduces the likelihood of patients discontinuing because of inflammation, burning, or other adverse events, Dr. Tyring noted.
Ingenol mebutate significantly enhanced the efficacy of cryosurgery, a difference that comes "both from enhancing the effect of cryosurgery with ingenol mebutate on the visible baseline lesions, and from a field treatment effect of ingenol mebutate on subclinical lesions not visible at baseline," Dr. Tyring said.
Dr. Tyring disclosed financial relationships with Astellas, Epiphany, Catalyst, GlaxoSmithKline, Novartis, 3M, VaxGen, Merck, BMS, Amgen, Biogen, Genentech, Corixa, Abbott, Graceway, Leo, and Warner Chilcott. SDEF and this news organization are owned by the same parent company.
Sequential topical field-directed therapy with ingenol mebutate gel for 3 days following cryosurgery significantly improves the outcomes in patients with actinic keratosis, a phase III randomized study showed.
Enrolled patients had four to eight clinically typical lesions on the face or scalp and received cryosurgery to all visible lesions followed by once-daily treatment of ingenol mebutate 0.015% gel or vehicle gel for 3 consecutive days at home, after 3 weeks of healing.
At 11 weeks, patients who received treatment with ingenol mebutate gel (n = 167) postsurgery, saw higher rates of complete clearance of lesions, compared with patients receiving the vehicle alone after surgery (n = 162) (60.5% vs. 49.4%; P=.04), reported Dr. Stephen K. Tyring and his associates this month (J. Drugs Dermatol. 2014;13:154-60).
In longer-term follow-up at 12 months, ingenol mebutate gel continued to show significantly improved complete clearance rates, compared with vehicle gel (30.5% vs. 18.5%; P = .01), Dr. Tyring reported at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
The mean percentage reduction of lesions at 12 months was significantly higher with ingenol mebutate for all lesions (59.5% vs 44.4%, P=0.004) and 38.9% of patients receiving ingenol mebutate saw emergence of new lesions on the treatment area between baseline lesions, compared with 51.9% of patients in the vehicle group (P=0.01), he said.
The complete clearance at 11 weeks (60.5% vs 49.4%; P=0.04) with a relative complete actinic keratosis clearance ratio of 1.22 (1.01-1.49) increased to 1.67 (1.12-2.50) at 12 months in the treatment group vs vehicle, following cryosurgery, noted Dr. Tyring of University of Texas Health Science Center at Houston.
The most frequently reported adverse events in the treatment group were application site discomfort and pruritus. Over the 12-month follow-up, 16 patients in the ingenol mebutate group reported one or more adverse events, as did 6 in the vehicle group. These events, however, were minor and well tolerated, said Dr. Tyring.
Ingenol mebutate gel was approved by the Food and Drug Administration in 2012 to treat actinic keratosis. Its advantage over other topical field agents such as 5-fluorouracil, imiquimod, and diclofenac is its short treatment period of 2-3 days, depending on the treatment area. This reduces the likelihood of patients discontinuing because of inflammation, burning, or other adverse events, Dr. Tyring noted.
Ingenol mebutate significantly enhanced the efficacy of cryosurgery, a difference that comes "both from enhancing the effect of cryosurgery with ingenol mebutate on the visible baseline lesions, and from a field treatment effect of ingenol mebutate on subclinical lesions not visible at baseline," Dr. Tyring said.
Dr. Tyring disclosed financial relationships with Astellas, Epiphany, Catalyst, GlaxoSmithKline, Novartis, 3M, VaxGen, Merck, BMS, Amgen, Biogen, Genentech, Corixa, Abbott, Graceway, Leo, and Warner Chilcott. SDEF and this news organization are owned by the same parent company.
Sequential topical field-directed therapy with ingenol mebutate gel for 3 days following cryosurgery significantly improves the outcomes in patients with actinic keratosis, a phase III randomized study showed.
Enrolled patients had four to eight clinically typical lesions on the face or scalp and received cryosurgery to all visible lesions followed by once-daily treatment of ingenol mebutate 0.015% gel or vehicle gel for 3 consecutive days at home, after 3 weeks of healing.
At 11 weeks, patients who received treatment with ingenol mebutate gel (n = 167) postsurgery, saw higher rates of complete clearance of lesions, compared with patients receiving the vehicle alone after surgery (n = 162) (60.5% vs. 49.4%; P=.04), reported Dr. Stephen K. Tyring and his associates this month (J. Drugs Dermatol. 2014;13:154-60).
In longer-term follow-up at 12 months, ingenol mebutate gel continued to show significantly improved complete clearance rates, compared with vehicle gel (30.5% vs. 18.5%; P = .01), Dr. Tyring reported at the Hawaii Dermatology Seminar sponsored by Global Academy for Medical Education/Skin Disease Education Foundation.
The mean percentage reduction of lesions at 12 months was significantly higher with ingenol mebutate for all lesions (59.5% vs 44.4%, P=0.004) and 38.9% of patients receiving ingenol mebutate saw emergence of new lesions on the treatment area between baseline lesions, compared with 51.9% of patients in the vehicle group (P=0.01), he said.
The complete clearance at 11 weeks (60.5% vs 49.4%; P=0.04) with a relative complete actinic keratosis clearance ratio of 1.22 (1.01-1.49) increased to 1.67 (1.12-2.50) at 12 months in the treatment group vs vehicle, following cryosurgery, noted Dr. Tyring of University of Texas Health Science Center at Houston.
The most frequently reported adverse events in the treatment group were application site discomfort and pruritus. Over the 12-month follow-up, 16 patients in the ingenol mebutate group reported one or more adverse events, as did 6 in the vehicle group. These events, however, were minor and well tolerated, said Dr. Tyring.
Ingenol mebutate gel was approved by the Food and Drug Administration in 2012 to treat actinic keratosis. Its advantage over other topical field agents such as 5-fluorouracil, imiquimod, and diclofenac is its short treatment period of 2-3 days, depending on the treatment area. This reduces the likelihood of patients discontinuing because of inflammation, burning, or other adverse events, Dr. Tyring noted.
Ingenol mebutate significantly enhanced the efficacy of cryosurgery, a difference that comes "both from enhancing the effect of cryosurgery with ingenol mebutate on the visible baseline lesions, and from a field treatment effect of ingenol mebutate on subclinical lesions not visible at baseline," Dr. Tyring said.
Dr. Tyring disclosed financial relationships with Astellas, Epiphany, Catalyst, GlaxoSmithKline, Novartis, 3M, VaxGen, Merck, BMS, Amgen, Biogen, Genentech, Corixa, Abbott, Graceway, Leo, and Warner Chilcott. SDEF and this news organization are owned by the same parent company.
FROM SDEF HAWAII DERMATOLOGY SEMINAR
Major finding: Ingenol mebutate gel following cryosurgery significantly improved complete clearance rates, compared with vehicle gel at 12 months (30.5% vs. 18.5%; P =.01). Mean percentage reduction of AK lesions at 12 months was significantly higher with ingenol mebutate for all lesions than with vehicle (59.5% vs. 44.4%; P = .004)
Data source: Phase III multicenter, randomized, double-blind study of 329 patients with actinic keratosis on the face or scalp.
Disclosures: Dr. Tyring disclosed financial relationships with Astellas, Epiphany, Catalyst, GlaxoSmithKline, Novartis, 3M, VaxGen, Merck, BMS, Amgen, Biogen, Genentech, Corixa, Abbott, Graceway, Leo, and Warner Chilcott.
Youth tanning curbed in states with restrictions
States with stronger laws to restrict tanning bed access for minors reported significantly lower instances of tanning among teens than states without restrictions, according to a large-scale review published by the American Journal of Public Health.
Overall, female minors in states with tanning access laws used the beds 30% less than teens in states without any restrictions. The laws include warning signs on indoor tanning devices, limited advertising, and mandatory protective eyewear.
"State indoor tanning laws, especially age restrictions, may be effective in reducing indoor tanning among our nation’s youth," wrote Gery P. Guy Jr., Ph.D., of the Centers for Disease Control and Prevention and his colleagues.
Six states (California, Illinois, Nevada, Oregon, Texas, and Vermont), currently restrict indoor tanning among minors younger than 18 years, the researchers noted.
In addition, data from states with youth access laws – laws that require parental permission or place an outright ban on teen use of tanning beds – indicated that female students use tanning beds 42% less than those in states without the laws (Am. J. Public Health 2014 [doi: 10.2105/AJPH.2013.301850]).
The researchers used nationally representative data from 31,835 adolescents from 2009 and 2011 collected as part of the national Youth Risk Behavior Surveys of U.S. high school students in grades 9-12. They conducted a multivariable logistic regression analysis to examine the associations between the indoor tanning laws of different states and the reports of indoor tanning among high school students in those states. The survey found that 23.4 % of the young women surveyed said they used indoor tanning, and 6.5% of young men admitted to using the devices.
The researchers had no financial conflicts to disclose.
States with stronger laws to restrict tanning bed access for minors reported significantly lower instances of tanning among teens than states without restrictions, according to a large-scale review published by the American Journal of Public Health.
Overall, female minors in states with tanning access laws used the beds 30% less than teens in states without any restrictions. The laws include warning signs on indoor tanning devices, limited advertising, and mandatory protective eyewear.
"State indoor tanning laws, especially age restrictions, may be effective in reducing indoor tanning among our nation’s youth," wrote Gery P. Guy Jr., Ph.D., of the Centers for Disease Control and Prevention and his colleagues.
Six states (California, Illinois, Nevada, Oregon, Texas, and Vermont), currently restrict indoor tanning among minors younger than 18 years, the researchers noted.
In addition, data from states with youth access laws – laws that require parental permission or place an outright ban on teen use of tanning beds – indicated that female students use tanning beds 42% less than those in states without the laws (Am. J. Public Health 2014 [doi: 10.2105/AJPH.2013.301850]).
The researchers used nationally representative data from 31,835 adolescents from 2009 and 2011 collected as part of the national Youth Risk Behavior Surveys of U.S. high school students in grades 9-12. They conducted a multivariable logistic regression analysis to examine the associations between the indoor tanning laws of different states and the reports of indoor tanning among high school students in those states. The survey found that 23.4 % of the young women surveyed said they used indoor tanning, and 6.5% of young men admitted to using the devices.
The researchers had no financial conflicts to disclose.
States with stronger laws to restrict tanning bed access for minors reported significantly lower instances of tanning among teens than states without restrictions, according to a large-scale review published by the American Journal of Public Health.
Overall, female minors in states with tanning access laws used the beds 30% less than teens in states without any restrictions. The laws include warning signs on indoor tanning devices, limited advertising, and mandatory protective eyewear.
"State indoor tanning laws, especially age restrictions, may be effective in reducing indoor tanning among our nation’s youth," wrote Gery P. Guy Jr., Ph.D., of the Centers for Disease Control and Prevention and his colleagues.
Six states (California, Illinois, Nevada, Oregon, Texas, and Vermont), currently restrict indoor tanning among minors younger than 18 years, the researchers noted.
In addition, data from states with youth access laws – laws that require parental permission or place an outright ban on teen use of tanning beds – indicated that female students use tanning beds 42% less than those in states without the laws (Am. J. Public Health 2014 [doi: 10.2105/AJPH.2013.301850]).
The researchers used nationally representative data from 31,835 adolescents from 2009 and 2011 collected as part of the national Youth Risk Behavior Surveys of U.S. high school students in grades 9-12. They conducted a multivariable logistic regression analysis to examine the associations between the indoor tanning laws of different states and the reports of indoor tanning among high school students in those states. The survey found that 23.4 % of the young women surveyed said they used indoor tanning, and 6.5% of young men admitted to using the devices.
The researchers had no financial conflicts to disclose.
FROM THE AMERICAN JOURNAL OF PUBLIC HEALTH
Major finding: Youth access laws reduce indoor tanning use by 42% in teenage girls.
Data source: Data review performed by the CDC based on National Youth Risk Behavior Surveys from 2009 and 2011.
Final results validate sentinel-node biopsy for melanoma
Sentinel-node biopsy clearly benefits patients with intermediate-thickness or thick primary melanomas who are found to have nodal involvement and undergo immediate lymphadenectomy, tripling their disease-free survival and doubling their melanoma-specific survival and distant disease-free survival, according to a report published online Feb. 12 in the New England Journal of Medicine.
These long-term findings, the final results of 20 years of investigation in MSLT-I (Multicenter Selective Lymphadenectomy Trial), "clearly validate the use of sentinel-node biopsy" in this patient population. Among patients found to have no nodal involvement, "the procedure provides accurate and important staging information," while among those found to have nodal involvement, it enhances regional disease control and substantially improves survival, said Dr. Donald L. Morton of the John Wayne Cancer Institute at Saint John’s Health Center, Santa Monica, Calif., and his associates.
Dr. Morton passed away shortly before publication of this final MSLT-I report.
The MSLT-I, which began in 1994, was intended to determine whether sentinel-node biopsy – a new procedure at the time that was developed by Dr. Morton – could accurately identify clinically occult metastases. More important, since only about 20% of such patients are found to have metastases, the MSLT-I was to determine whether it was worthwhile to subject all patients to the procedure. In other words, did sentinel-node biopsy with immediate lymphadenectomy of involved nodes yield better outcomes than did watchful waiting with delayed lymphadenectomy, performed only when nodal recurrence became evident during observation?
This final report involved 1,560 patients with localized cutaneous primary melanomas: 1,270 with intermediate-thickness (1.20-3.50 mm) lesions and 290 with thick (greater than 3.50 mm) lesions. A total of 943 patients were randomly assigned to undergo sentinel-node biopsy plus immediate lymphadenectomy if metastases were detected in sentinel nodes, and 617 were randomly assigned to close observation with delayed lymphadenectomy if nodal metastases developed during observation.
All the study participants were monitored periodically throughout follow-up using clinical examination, blood testing for biomarkers of melanoma, chest radiography, PET scanning, CT scanning, and/or nodal ultrasonography.
The primary end point, melanoma-specific survival across the entire study cohort, was not significantly different between patients who underwent sentinel-node biopsy (81.4%) and those who had observation only (78.3%). This is not surprising as only the 20% of patients who actually had nodal metastases could potentially derive a survival benefit from the procedure.
Latent-subgroup statistical methods were used to assess only this 20% of patients with nodal metastases. In this, the most relevant, data analysis, disease-free survival was increased by a factor of 3.2, distant disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for patients who underwent sentinel-node biopsy compared with those who had observation only, Dr. Morton and his associates wrote (N. Engl. J. Med. 2014 Feb 12 [doi:10.1056/NEJMoa1310460]).
Among patients with intermediate-thickness melanomas, the estimated treatment effect on disease-free survival was 1.17 (P less than .001), and the estimated effect on distant disease-free survival was 0.73 (P = .04). For melanoma-specific survival, the estimated treatment effect was 0.68 (P = .05).
"Our long-term results confirm that sentinel-node biopsy correctly determines the pathologic status of the nodal basin in 96% of cases and is the most powerful prognostic indicator," they noted.
The data also indicate that "essentially all metastases detected by sentinel-node biopsy eventually would have become clinically evident if [they had] not [been] removed," the investigators added.
The MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
The final data from this landmark trial "corroborate the profound prognostic significance of micrometastasis identified by sentinel-node biopsy," said Dr. Charles M. Balch and Dr. Jeffrey E. Gershenwald.
The finding that early lymphadenectomy improved melanoma-specific survival by 44% implies that delaying lymphadenectomy until there was clinical evidence of metastasis allowed an opportunity for further dissemination of regional disease. And the fact that the primary end point (improved melanoma-specific survival across the entire cohort of patients with intermediate thickness melanomas) of MSLT-I was not achieved "doesn’t detract from the clinical importance of regional lymph-node staging," they said.
Dr. Balch is at the University of Texas Southwestern Medical Center, Dallas; he reported ties to Merck and Amgen. Dr. Gershenwald is in the department of surgical oncology at the melanoma and skin center at the University of Texas M.D. Anderson Cancer Center, Houston.; he reported ties to Navidea, GlaxoSmithKline, UpToDate, and Mercator Therapeutics. These remarks were taken from their editorial accompanying Dr. Morton’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056e1313690]).
The final data from this landmark trial "corroborate the profound prognostic significance of micrometastasis identified by sentinel-node biopsy," said Dr. Charles M. Balch and Dr. Jeffrey E. Gershenwald.
The finding that early lymphadenectomy improved melanoma-specific survival by 44% implies that delaying lymphadenectomy until there was clinical evidence of metastasis allowed an opportunity for further dissemination of regional disease. And the fact that the primary end point (improved melanoma-specific survival across the entire cohort of patients with intermediate thickness melanomas) of MSLT-I was not achieved "doesn’t detract from the clinical importance of regional lymph-node staging," they said.
Dr. Balch is at the University of Texas Southwestern Medical Center, Dallas; he reported ties to Merck and Amgen. Dr. Gershenwald is in the department of surgical oncology at the melanoma and skin center at the University of Texas M.D. Anderson Cancer Center, Houston.; he reported ties to Navidea, GlaxoSmithKline, UpToDate, and Mercator Therapeutics. These remarks were taken from their editorial accompanying Dr. Morton’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056e1313690]).
The final data from this landmark trial "corroborate the profound prognostic significance of micrometastasis identified by sentinel-node biopsy," said Dr. Charles M. Balch and Dr. Jeffrey E. Gershenwald.
The finding that early lymphadenectomy improved melanoma-specific survival by 44% implies that delaying lymphadenectomy until there was clinical evidence of metastasis allowed an opportunity for further dissemination of regional disease. And the fact that the primary end point (improved melanoma-specific survival across the entire cohort of patients with intermediate thickness melanomas) of MSLT-I was not achieved "doesn’t detract from the clinical importance of regional lymph-node staging," they said.
Dr. Balch is at the University of Texas Southwestern Medical Center, Dallas; he reported ties to Merck and Amgen. Dr. Gershenwald is in the department of surgical oncology at the melanoma and skin center at the University of Texas M.D. Anderson Cancer Center, Houston.; he reported ties to Navidea, GlaxoSmithKline, UpToDate, and Mercator Therapeutics. These remarks were taken from their editorial accompanying Dr. Morton’s report (N. Engl. J. Med. 2014 Feb. 12 [doi:10.1056e1313690]).
Sentinel-node biopsy clearly benefits patients with intermediate-thickness or thick primary melanomas who are found to have nodal involvement and undergo immediate lymphadenectomy, tripling their disease-free survival and doubling their melanoma-specific survival and distant disease-free survival, according to a report published online Feb. 12 in the New England Journal of Medicine.
These long-term findings, the final results of 20 years of investigation in MSLT-I (Multicenter Selective Lymphadenectomy Trial), "clearly validate the use of sentinel-node biopsy" in this patient population. Among patients found to have no nodal involvement, "the procedure provides accurate and important staging information," while among those found to have nodal involvement, it enhances regional disease control and substantially improves survival, said Dr. Donald L. Morton of the John Wayne Cancer Institute at Saint John’s Health Center, Santa Monica, Calif., and his associates.
Dr. Morton passed away shortly before publication of this final MSLT-I report.
The MSLT-I, which began in 1994, was intended to determine whether sentinel-node biopsy – a new procedure at the time that was developed by Dr. Morton – could accurately identify clinically occult metastases. More important, since only about 20% of such patients are found to have metastases, the MSLT-I was to determine whether it was worthwhile to subject all patients to the procedure. In other words, did sentinel-node biopsy with immediate lymphadenectomy of involved nodes yield better outcomes than did watchful waiting with delayed lymphadenectomy, performed only when nodal recurrence became evident during observation?
This final report involved 1,560 patients with localized cutaneous primary melanomas: 1,270 with intermediate-thickness (1.20-3.50 mm) lesions and 290 with thick (greater than 3.50 mm) lesions. A total of 943 patients were randomly assigned to undergo sentinel-node biopsy plus immediate lymphadenectomy if metastases were detected in sentinel nodes, and 617 were randomly assigned to close observation with delayed lymphadenectomy if nodal metastases developed during observation.
All the study participants were monitored periodically throughout follow-up using clinical examination, blood testing for biomarkers of melanoma, chest radiography, PET scanning, CT scanning, and/or nodal ultrasonography.
The primary end point, melanoma-specific survival across the entire study cohort, was not significantly different between patients who underwent sentinel-node biopsy (81.4%) and those who had observation only (78.3%). This is not surprising as only the 20% of patients who actually had nodal metastases could potentially derive a survival benefit from the procedure.
Latent-subgroup statistical methods were used to assess only this 20% of patients with nodal metastases. In this, the most relevant, data analysis, disease-free survival was increased by a factor of 3.2, distant disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for patients who underwent sentinel-node biopsy compared with those who had observation only, Dr. Morton and his associates wrote (N. Engl. J. Med. 2014 Feb 12 [doi:10.1056/NEJMoa1310460]).
Among patients with intermediate-thickness melanomas, the estimated treatment effect on disease-free survival was 1.17 (P less than .001), and the estimated effect on distant disease-free survival was 0.73 (P = .04). For melanoma-specific survival, the estimated treatment effect was 0.68 (P = .05).
"Our long-term results confirm that sentinel-node biopsy correctly determines the pathologic status of the nodal basin in 96% of cases and is the most powerful prognostic indicator," they noted.
The data also indicate that "essentially all metastases detected by sentinel-node biopsy eventually would have become clinically evident if [they had] not [been] removed," the investigators added.
The MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
Sentinel-node biopsy clearly benefits patients with intermediate-thickness or thick primary melanomas who are found to have nodal involvement and undergo immediate lymphadenectomy, tripling their disease-free survival and doubling their melanoma-specific survival and distant disease-free survival, according to a report published online Feb. 12 in the New England Journal of Medicine.
These long-term findings, the final results of 20 years of investigation in MSLT-I (Multicenter Selective Lymphadenectomy Trial), "clearly validate the use of sentinel-node biopsy" in this patient population. Among patients found to have no nodal involvement, "the procedure provides accurate and important staging information," while among those found to have nodal involvement, it enhances regional disease control and substantially improves survival, said Dr. Donald L. Morton of the John Wayne Cancer Institute at Saint John’s Health Center, Santa Monica, Calif., and his associates.
Dr. Morton passed away shortly before publication of this final MSLT-I report.
The MSLT-I, which began in 1994, was intended to determine whether sentinel-node biopsy – a new procedure at the time that was developed by Dr. Morton – could accurately identify clinically occult metastases. More important, since only about 20% of such patients are found to have metastases, the MSLT-I was to determine whether it was worthwhile to subject all patients to the procedure. In other words, did sentinel-node biopsy with immediate lymphadenectomy of involved nodes yield better outcomes than did watchful waiting with delayed lymphadenectomy, performed only when nodal recurrence became evident during observation?
This final report involved 1,560 patients with localized cutaneous primary melanomas: 1,270 with intermediate-thickness (1.20-3.50 mm) lesions and 290 with thick (greater than 3.50 mm) lesions. A total of 943 patients were randomly assigned to undergo sentinel-node biopsy plus immediate lymphadenectomy if metastases were detected in sentinel nodes, and 617 were randomly assigned to close observation with delayed lymphadenectomy if nodal metastases developed during observation.
All the study participants were monitored periodically throughout follow-up using clinical examination, blood testing for biomarkers of melanoma, chest radiography, PET scanning, CT scanning, and/or nodal ultrasonography.
The primary end point, melanoma-specific survival across the entire study cohort, was not significantly different between patients who underwent sentinel-node biopsy (81.4%) and those who had observation only (78.3%). This is not surprising as only the 20% of patients who actually had nodal metastases could potentially derive a survival benefit from the procedure.
Latent-subgroup statistical methods were used to assess only this 20% of patients with nodal metastases. In this, the most relevant, data analysis, disease-free survival was increased by a factor of 3.2, distant disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for patients who underwent sentinel-node biopsy compared with those who had observation only, Dr. Morton and his associates wrote (N. Engl. J. Med. 2014 Feb 12 [doi:10.1056/NEJMoa1310460]).
Among patients with intermediate-thickness melanomas, the estimated treatment effect on disease-free survival was 1.17 (P less than .001), and the estimated effect on distant disease-free survival was 0.73 (P = .04). For melanoma-specific survival, the estimated treatment effect was 0.68 (P = .05).
"Our long-term results confirm that sentinel-node biopsy correctly determines the pathologic status of the nodal basin in 96% of cases and is the most powerful prognostic indicator," they noted.
The data also indicate that "essentially all metastases detected by sentinel-node biopsy eventually would have become clinically evident if [they had] not [been] removed," the investigators added.
The MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Major finding: Among patients with nodal metastases, disease-free survival was increased by a factor of 3.2, distant-disease-free survival was increased by a factor of 2.1, and melanoma-specific survival was increased by a factor of 2.0 for those who underwent sentinel-node biopsy, compared with those who had observation only.
Data source: The 20-year international randomized MSLT-I study involving 1,560 patients with intermediate-thickness or thick localized cutaneous melanomas comparing SNB with nodal observation.
Disclosures: MSLT-I was supported by the National Cancer Institute and the Australian and New Zealand Melanoma Trials Group. Dr. Morton reported no financial conflicts of interest; his associates reported ties to GlaxoSmithKline, Roche, Provectus, Merck, Myriad Genetics, and Melanoma Diagnostics.
Cosmetic Corner: Dermatologists Weigh in on Sunless Tanners
To improve patient care and outcomes, leading dermatologists offered their recommendations on the top sunless tanners. Consideration must be given to:
La Roche-Posay Laboratoire Dermatologique
Recommended by Marta I. Rendon, MD, Boca Raton, Florida
Johnson & Johnson Consumer Companies, Inc
“For daily use Aveeno Continuous Radiance Moisturizing Lotion is excellent.”—Patricia K. Farris, Metairie, Lousiana
Clinique Laboratories, LLC
“Clinique Self Sun is my favorite as it provides a gradual tanning. Always explain to your patient that she needs to exfoliate the skin before applying.”—Antonella Tosti, MD, Miami, Florida
Kao USA Inc.
“I am a fan of Jergens sunless tanner. It is gradual, inexpensive, and easy to use. I always remind my patients that sunless tanners do not product from UV damage. You still need SPF on top of your sunless tan. But, a gradual tanner is a good and safe way to minimize minor imperfections like spider veins.”—Elizabeth K. Hale, MD, New York, New York
Clarins
“For a quick bronzing product, I like Clarins Self Tanning Instant Gel. It is easy to apply and gives you a great glow.”—Patricia K. Farris, MD, Metairie, Louisiana
PZ Cussons Beauty LLP
“There are many different products offered and shades. The tan produced is even and natural looking.”—Anthony M. Rossi, MD, New York, New York
Kiehl's
Recommended by Gary Goldenberg, MD, New York, New York
Iredale Mineral Cosmetics, Ltd
“Jane Iredale Tantasia is my favorite sunless tanner. It has a pleasant citrus scent, isn’t greasy or streaky, can be used on the face without causing acne, and gives a natural golden glow.”—Wm. Philip Werschler, MD, Seattle, Washington
Cutis invites readers to send us their recommendations. OTC antifungals, OTC hair restorations, and antiperspirants will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to msteiger@frontlinemedcom.com.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on the top sunless tanners. Consideration must be given to:
La Roche-Posay Laboratoire Dermatologique
Recommended by Marta I. Rendon, MD, Boca Raton, Florida
Johnson & Johnson Consumer Companies, Inc
“For daily use Aveeno Continuous Radiance Moisturizing Lotion is excellent.”—Patricia K. Farris, Metairie, Lousiana
Clinique Laboratories, LLC
“Clinique Self Sun is my favorite as it provides a gradual tanning. Always explain to your patient that she needs to exfoliate the skin before applying.”—Antonella Tosti, MD, Miami, Florida
Kao USA Inc.
“I am a fan of Jergens sunless tanner. It is gradual, inexpensive, and easy to use. I always remind my patients that sunless tanners do not product from UV damage. You still need SPF on top of your sunless tan. But, a gradual tanner is a good and safe way to minimize minor imperfections like spider veins.”—Elizabeth K. Hale, MD, New York, New York
Clarins
“For a quick bronzing product, I like Clarins Self Tanning Instant Gel. It is easy to apply and gives you a great glow.”—Patricia K. Farris, MD, Metairie, Louisiana
PZ Cussons Beauty LLP
“There are many different products offered and shades. The tan produced is even and natural looking.”—Anthony M. Rossi, MD, New York, New York
Kiehl's
Recommended by Gary Goldenberg, MD, New York, New York
Iredale Mineral Cosmetics, Ltd
“Jane Iredale Tantasia is my favorite sunless tanner. It has a pleasant citrus scent, isn’t greasy or streaky, can be used on the face without causing acne, and gives a natural golden glow.”—Wm. Philip Werschler, MD, Seattle, Washington
Cutis invites readers to send us their recommendations. OTC antifungals, OTC hair restorations, and antiperspirants will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to msteiger@frontlinemedcom.com.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
To improve patient care and outcomes, leading dermatologists offered their recommendations on the top sunless tanners. Consideration must be given to:
La Roche-Posay Laboratoire Dermatologique
Recommended by Marta I. Rendon, MD, Boca Raton, Florida
Johnson & Johnson Consumer Companies, Inc
“For daily use Aveeno Continuous Radiance Moisturizing Lotion is excellent.”—Patricia K. Farris, Metairie, Lousiana
Clinique Laboratories, LLC
“Clinique Self Sun is my favorite as it provides a gradual tanning. Always explain to your patient that she needs to exfoliate the skin before applying.”—Antonella Tosti, MD, Miami, Florida
Kao USA Inc.
“I am a fan of Jergens sunless tanner. It is gradual, inexpensive, and easy to use. I always remind my patients that sunless tanners do not product from UV damage. You still need SPF on top of your sunless tan. But, a gradual tanner is a good and safe way to minimize minor imperfections like spider veins.”—Elizabeth K. Hale, MD, New York, New York
Clarins
“For a quick bronzing product, I like Clarins Self Tanning Instant Gel. It is easy to apply and gives you a great glow.”—Patricia K. Farris, MD, Metairie, Louisiana
PZ Cussons Beauty LLP
“There are many different products offered and shades. The tan produced is even and natural looking.”—Anthony M. Rossi, MD, New York, New York
Kiehl's
Recommended by Gary Goldenberg, MD, New York, New York
Iredale Mineral Cosmetics, Ltd
“Jane Iredale Tantasia is my favorite sunless tanner. It has a pleasant citrus scent, isn’t greasy or streaky, can be used on the face without causing acne, and gives a natural golden glow.”—Wm. Philip Werschler, MD, Seattle, Washington
Cutis invites readers to send us their recommendations. OTC antifungals, OTC hair restorations, and antiperspirants will be featured in upcoming editions of Cosmetic Corner. Please e-mail your recommendation(s) to msteiger@frontlinemedcom.com.
Disclaimer: Opinions expressed herein do not necessarily reflect those of Cutis or Frontline Medical Communications Inc. and shall not be used for product endorsement purposes. Any reference made to a specific commercial product does not indicate or imply that Cutis or Frontline Medical Communications Inc. endorses, recommends, or favors the product mentioned. No guarantee is given to the effects of recommended products.
Product News: 02 2014
Current options and future directions in the systemic treatment of metastatic melanoma
Systemic treatment options for metastatic melanoma have historically been limited, with conventional cytotoxic chemotherapies demonstrating only modest benefit. Recent advances, however, have dramatically changed the treatment landscape and can be considered in 2 general categories: immunotherapeutic approaches that enhance antitumor immunity, and targeted therapeutic approaches that block oncogenic driver mutations. Immunotherapy with antibodies that block cytotoxic T-lymphocyte antigen 4 and programmed death-1 receptor can result in durable responses in a subset of patients. These treatments may be considered for patients irrespective of their mutational status, and ongoing research continues to investigate biomarkers associated with clinical outcomes. Side effects of these agents result from immune-mediated reactions involving various organ sites and can include: diarrhea, rash, hepatitis, and endocrinopathies.
Click on the PDF icon at the top of this introduction to read the full article.
Systemic treatment options for metastatic melanoma have historically been limited, with conventional cytotoxic chemotherapies demonstrating only modest benefit. Recent advances, however, have dramatically changed the treatment landscape and can be considered in 2 general categories: immunotherapeutic approaches that enhance antitumor immunity, and targeted therapeutic approaches that block oncogenic driver mutations. Immunotherapy with antibodies that block cytotoxic T-lymphocyte antigen 4 and programmed death-1 receptor can result in durable responses in a subset of patients. These treatments may be considered for patients irrespective of their mutational status, and ongoing research continues to investigate biomarkers associated with clinical outcomes. Side effects of these agents result from immune-mediated reactions involving various organ sites and can include: diarrhea, rash, hepatitis, and endocrinopathies.
Click on the PDF icon at the top of this introduction to read the full article.
Systemic treatment options for metastatic melanoma have historically been limited, with conventional cytotoxic chemotherapies demonstrating only modest benefit. Recent advances, however, have dramatically changed the treatment landscape and can be considered in 2 general categories: immunotherapeutic approaches that enhance antitumor immunity, and targeted therapeutic approaches that block oncogenic driver mutations. Immunotherapy with antibodies that block cytotoxic T-lymphocyte antigen 4 and programmed death-1 receptor can result in durable responses in a subset of patients. These treatments may be considered for patients irrespective of their mutational status, and ongoing research continues to investigate biomarkers associated with clinical outcomes. Side effects of these agents result from immune-mediated reactions involving various organ sites and can include: diarrhea, rash, hepatitis, and endocrinopathies.
Click on the PDF icon at the top of this introduction to read the full article.
Skin cancer risk stands out in anti-TNF biologics use
SNOWMASS, COLO. – The use of anti–tumor necrosis factor biologics in patients with rheumatoid arthritis, psoriasis, and other serious autoimmune diseases is not associated with increased risk of most forms of cancer, with two highly visible and important exceptions: nonmelanoma skin cancer and malignant melanoma.
That’s the largely reassuring conclusion to be drawn from two large studies conducted by researchers at Stockholm’s Karolinska Institute, according to Dr. Jeffrey R. Curtis, director of the arthritis clinical intervention program at the University of Alabama, Birmingham.
The Swedish group conducted a comprehensive meta-analysis of all 74 pharmaceutical company–sponsored randomized controlled trials of anti-TNF biologics lasting at least 4 weeks. The investigators used individual patient data for 15,418 anti-TNF recipients and 7,486 subjects randomized to methotrexate and other comparators. About half of the randomized trials focused on rheumatoid arthritis (RA) patients; the other half involved patients with the other approved indications for anti-TNF therapy.
The use of individual patient data was a nuance of the meta-analysis that wowed Dr. Curtis.
"This is a really great paper. One of the reasons I was very impressed with it is that getting drug companies to give up any raw person-level data to anybody is difficult. The European Medicines Agency requested it, and that’s probably the only reason it happened," Dr. Curtis observed at the Winter Rheumatology symposium sponsored by the American College of Rheumatology.
The overall relative risk for all forms of cancer, excluding nonmelanoma skin cancer, in patients on anti-TNF biologics was 0.99. Their cancer rate was 641 per 100,000 person-years (Pharmacoepidemiol. Drug Saf. 2011;20:119-30).
"You can tell your patients the risk for cancer is less than 1 in 100 – it’s about 6 per 1,000 – and it’s not increased compared to background therapy," the rheumatologist said.
The exception was nonmelanoma skin cancer. The relative risk for this malignancy among users of all anti-TNF biologics was increased twofold.
In a separate study, the same group used prospectively recorded data from Swedish national registries to investigate the association between anti-TNF therapy and melanoma. This analysis involved 10,878 RA patients treated with anti-TNF biologics, 42,198 others who were not, and 162,743 matched controls from the general population.
Compared with controls, RA patients not on anti-TNF biologics did not have a significantly increased risk of malignant melanoma. However, the 38 first invasive melanomas that occurred in RA patients taking a TNF antagonist signified a 1.5-fold increased risk of this malignancy compared to the general population. This 50% increase in relative risk was statistically significant. The absolute increase in risk was quite small: 20 additional cases per 100,000 person-years. The number needed to harm – that is, the number of patients who needed to be treated with an anti-TNF biologic to cause one additional case of melanoma – was 50,000 (BMJ 2013;346:f1939 [doi: 10.1136/bmj.f1939]).
Dr. Curtis noted that patients who have been urged to consider going on a TNF antagonist often return to the office waving a printout of the product labeling that warns of the possibility of malignancy.
"The bottom line: I tell patients that based on the data that there doesn’t appear to be an increased risk for all types of cancer, including hematologic, with the exceptions of nonmelanoma skin cancer and melanoma. Those are the two we need to advise patients about," he said.
Dr. Curtis reported receiving funding from the National Institutes of Health, the Agency for Healthcare Research and Quality, and 10 pharmaceutical companies.
SNOWMASS, COLO. – The use of anti–tumor necrosis factor biologics in patients with rheumatoid arthritis, psoriasis, and other serious autoimmune diseases is not associated with increased risk of most forms of cancer, with two highly visible and important exceptions: nonmelanoma skin cancer and malignant melanoma.
That’s the largely reassuring conclusion to be drawn from two large studies conducted by researchers at Stockholm’s Karolinska Institute, according to Dr. Jeffrey R. Curtis, director of the arthritis clinical intervention program at the University of Alabama, Birmingham.
The Swedish group conducted a comprehensive meta-analysis of all 74 pharmaceutical company–sponsored randomized controlled trials of anti-TNF biologics lasting at least 4 weeks. The investigators used individual patient data for 15,418 anti-TNF recipients and 7,486 subjects randomized to methotrexate and other comparators. About half of the randomized trials focused on rheumatoid arthritis (RA) patients; the other half involved patients with the other approved indications for anti-TNF therapy.
The use of individual patient data was a nuance of the meta-analysis that wowed Dr. Curtis.
"This is a really great paper. One of the reasons I was very impressed with it is that getting drug companies to give up any raw person-level data to anybody is difficult. The European Medicines Agency requested it, and that’s probably the only reason it happened," Dr. Curtis observed at the Winter Rheumatology symposium sponsored by the American College of Rheumatology.
The overall relative risk for all forms of cancer, excluding nonmelanoma skin cancer, in patients on anti-TNF biologics was 0.99. Their cancer rate was 641 per 100,000 person-years (Pharmacoepidemiol. Drug Saf. 2011;20:119-30).
"You can tell your patients the risk for cancer is less than 1 in 100 – it’s about 6 per 1,000 – and it’s not increased compared to background therapy," the rheumatologist said.
The exception was nonmelanoma skin cancer. The relative risk for this malignancy among users of all anti-TNF biologics was increased twofold.
In a separate study, the same group used prospectively recorded data from Swedish national registries to investigate the association between anti-TNF therapy and melanoma. This analysis involved 10,878 RA patients treated with anti-TNF biologics, 42,198 others who were not, and 162,743 matched controls from the general population.
Compared with controls, RA patients not on anti-TNF biologics did not have a significantly increased risk of malignant melanoma. However, the 38 first invasive melanomas that occurred in RA patients taking a TNF antagonist signified a 1.5-fold increased risk of this malignancy compared to the general population. This 50% increase in relative risk was statistically significant. The absolute increase in risk was quite small: 20 additional cases per 100,000 person-years. The number needed to harm – that is, the number of patients who needed to be treated with an anti-TNF biologic to cause one additional case of melanoma – was 50,000 (BMJ 2013;346:f1939 [doi: 10.1136/bmj.f1939]).
Dr. Curtis noted that patients who have been urged to consider going on a TNF antagonist often return to the office waving a printout of the product labeling that warns of the possibility of malignancy.
"The bottom line: I tell patients that based on the data that there doesn’t appear to be an increased risk for all types of cancer, including hematologic, with the exceptions of nonmelanoma skin cancer and melanoma. Those are the two we need to advise patients about," he said.
Dr. Curtis reported receiving funding from the National Institutes of Health, the Agency for Healthcare Research and Quality, and 10 pharmaceutical companies.
SNOWMASS, COLO. – The use of anti–tumor necrosis factor biologics in patients with rheumatoid arthritis, psoriasis, and other serious autoimmune diseases is not associated with increased risk of most forms of cancer, with two highly visible and important exceptions: nonmelanoma skin cancer and malignant melanoma.
That’s the largely reassuring conclusion to be drawn from two large studies conducted by researchers at Stockholm’s Karolinska Institute, according to Dr. Jeffrey R. Curtis, director of the arthritis clinical intervention program at the University of Alabama, Birmingham.
The Swedish group conducted a comprehensive meta-analysis of all 74 pharmaceutical company–sponsored randomized controlled trials of anti-TNF biologics lasting at least 4 weeks. The investigators used individual patient data for 15,418 anti-TNF recipients and 7,486 subjects randomized to methotrexate and other comparators. About half of the randomized trials focused on rheumatoid arthritis (RA) patients; the other half involved patients with the other approved indications for anti-TNF therapy.
The use of individual patient data was a nuance of the meta-analysis that wowed Dr. Curtis.
"This is a really great paper. One of the reasons I was very impressed with it is that getting drug companies to give up any raw person-level data to anybody is difficult. The European Medicines Agency requested it, and that’s probably the only reason it happened," Dr. Curtis observed at the Winter Rheumatology symposium sponsored by the American College of Rheumatology.
The overall relative risk for all forms of cancer, excluding nonmelanoma skin cancer, in patients on anti-TNF biologics was 0.99. Their cancer rate was 641 per 100,000 person-years (Pharmacoepidemiol. Drug Saf. 2011;20:119-30).
"You can tell your patients the risk for cancer is less than 1 in 100 – it’s about 6 per 1,000 – and it’s not increased compared to background therapy," the rheumatologist said.
The exception was nonmelanoma skin cancer. The relative risk for this malignancy among users of all anti-TNF biologics was increased twofold.
In a separate study, the same group used prospectively recorded data from Swedish national registries to investigate the association between anti-TNF therapy and melanoma. This analysis involved 10,878 RA patients treated with anti-TNF biologics, 42,198 others who were not, and 162,743 matched controls from the general population.
Compared with controls, RA patients not on anti-TNF biologics did not have a significantly increased risk of malignant melanoma. However, the 38 first invasive melanomas that occurred in RA patients taking a TNF antagonist signified a 1.5-fold increased risk of this malignancy compared to the general population. This 50% increase in relative risk was statistically significant. The absolute increase in risk was quite small: 20 additional cases per 100,000 person-years. The number needed to harm – that is, the number of patients who needed to be treated with an anti-TNF biologic to cause one additional case of melanoma – was 50,000 (BMJ 2013;346:f1939 [doi: 10.1136/bmj.f1939]).
Dr. Curtis noted that patients who have been urged to consider going on a TNF antagonist often return to the office waving a printout of the product labeling that warns of the possibility of malignancy.
"The bottom line: I tell patients that based on the data that there doesn’t appear to be an increased risk for all types of cancer, including hematologic, with the exceptions of nonmelanoma skin cancer and melanoma. Those are the two we need to advise patients about," he said.
Dr. Curtis reported receiving funding from the National Institutes of Health, the Agency for Healthcare Research and Quality, and 10 pharmaceutical companies.
EXPERT ANALYSIS FROM THE ACR WINTER RHEUMATOLOGY SYMPOSIUM
VEGF level may predict response to ipilimumab
Among patients with advanced melanoma who are being considered for ipilimumab therapy, serum vascular endothelial growth factor level may predict who will respond to the treatment and who will not, according to a report published online Feb. 4 in Cancer Immunology Research.
Only a subset of patients with advanced melanoma benefit from the CTLA-4 blocker ipilimumab, but those who do show marked and durable improvement, so there is "a critical need" to identify biomarkers that predict treatment response, said Dr. Jianda Yuan of the Ludwig Center for Cancer Immunotherapy, Sloan-Kettering Cancer Center, New York, and his associates.
The investigators assessed circulating vascular endothelial growth factor (VEGF) levels before and after ipilimumab therapy in 176 patients aged 16-91 years (mean age, 62) who were treated for advanced melanoma at Sloan-Kettering and at Dana-Farber/Harvard Cancer Center, Boston. A total of 78 patients received 10 mg/kg and 98 received 3 mg/kg every 3 weeks for 12 weeks. Those who showed clinical benefit and no dose-limiting toxicity at 24 weeks could continue receiving the drug until the disease progressed, they died, toxicity occurred, or they withdrew from the study.
One-third of the study participants (53 of the 159 patients evaluable at week 24) showed clinical benefit, including 3 complete responders, 13 partial responders, and 37 with stabilization of their disease, the investigators said (Cancer Immunol. Res 2014 Feb. 4 [doi:10.1158/2326-6066.CIR-13-0163]).
Patients with serum VEGF levels higher than 43 pg/mL were significantly less likely to respond to ipilimumab and had significantly shorter overall survival than those with lower levels, regardless of which dose of the drug they were given. Thirty-seven of the 90 patients with low VEGF (41%) showed clinical benefit with ipilimumab, compared with only 16 of the 69 patients with high VEGF (23%).
The findings of this preliminary research show that prospective studies are warranted to assess whether baseline serum VEGF can serve as a predictive biomarker, Dr. Yuan and his associates said.
This study was supported by the National Institutes of Health. Four of Dr. Yuan’s associates reported receiving support from and serving as consultants for Bristol-Myers Squibb, which developed the monoclonal antibody ipilimumab.
Among patients with advanced melanoma who are being considered for ipilimumab therapy, serum vascular endothelial growth factor level may predict who will respond to the treatment and who will not, according to a report published online Feb. 4 in Cancer Immunology Research.
Only a subset of patients with advanced melanoma benefit from the CTLA-4 blocker ipilimumab, but those who do show marked and durable improvement, so there is "a critical need" to identify biomarkers that predict treatment response, said Dr. Jianda Yuan of the Ludwig Center for Cancer Immunotherapy, Sloan-Kettering Cancer Center, New York, and his associates.
The investigators assessed circulating vascular endothelial growth factor (VEGF) levels before and after ipilimumab therapy in 176 patients aged 16-91 years (mean age, 62) who were treated for advanced melanoma at Sloan-Kettering and at Dana-Farber/Harvard Cancer Center, Boston. A total of 78 patients received 10 mg/kg and 98 received 3 mg/kg every 3 weeks for 12 weeks. Those who showed clinical benefit and no dose-limiting toxicity at 24 weeks could continue receiving the drug until the disease progressed, they died, toxicity occurred, or they withdrew from the study.
One-third of the study participants (53 of the 159 patients evaluable at week 24) showed clinical benefit, including 3 complete responders, 13 partial responders, and 37 with stabilization of their disease, the investigators said (Cancer Immunol. Res 2014 Feb. 4 [doi:10.1158/2326-6066.CIR-13-0163]).
Patients with serum VEGF levels higher than 43 pg/mL were significantly less likely to respond to ipilimumab and had significantly shorter overall survival than those with lower levels, regardless of which dose of the drug they were given. Thirty-seven of the 90 patients with low VEGF (41%) showed clinical benefit with ipilimumab, compared with only 16 of the 69 patients with high VEGF (23%).
The findings of this preliminary research show that prospective studies are warranted to assess whether baseline serum VEGF can serve as a predictive biomarker, Dr. Yuan and his associates said.
This study was supported by the National Institutes of Health. Four of Dr. Yuan’s associates reported receiving support from and serving as consultants for Bristol-Myers Squibb, which developed the monoclonal antibody ipilimumab.
Among patients with advanced melanoma who are being considered for ipilimumab therapy, serum vascular endothelial growth factor level may predict who will respond to the treatment and who will not, according to a report published online Feb. 4 in Cancer Immunology Research.
Only a subset of patients with advanced melanoma benefit from the CTLA-4 blocker ipilimumab, but those who do show marked and durable improvement, so there is "a critical need" to identify biomarkers that predict treatment response, said Dr. Jianda Yuan of the Ludwig Center for Cancer Immunotherapy, Sloan-Kettering Cancer Center, New York, and his associates.
The investigators assessed circulating vascular endothelial growth factor (VEGF) levels before and after ipilimumab therapy in 176 patients aged 16-91 years (mean age, 62) who were treated for advanced melanoma at Sloan-Kettering and at Dana-Farber/Harvard Cancer Center, Boston. A total of 78 patients received 10 mg/kg and 98 received 3 mg/kg every 3 weeks for 12 weeks. Those who showed clinical benefit and no dose-limiting toxicity at 24 weeks could continue receiving the drug until the disease progressed, they died, toxicity occurred, or they withdrew from the study.
One-third of the study participants (53 of the 159 patients evaluable at week 24) showed clinical benefit, including 3 complete responders, 13 partial responders, and 37 with stabilization of their disease, the investigators said (Cancer Immunol. Res 2014 Feb. 4 [doi:10.1158/2326-6066.CIR-13-0163]).
Patients with serum VEGF levels higher than 43 pg/mL were significantly less likely to respond to ipilimumab and had significantly shorter overall survival than those with lower levels, regardless of which dose of the drug they were given. Thirty-seven of the 90 patients with low VEGF (41%) showed clinical benefit with ipilimumab, compared with only 16 of the 69 patients with high VEGF (23%).
The findings of this preliminary research show that prospective studies are warranted to assess whether baseline serum VEGF can serve as a predictive biomarker, Dr. Yuan and his associates said.
This study was supported by the National Institutes of Health. Four of Dr. Yuan’s associates reported receiving support from and serving as consultants for Bristol-Myers Squibb, which developed the monoclonal antibody ipilimumab.
FROM CANCER IMMUNOLOGY RESEARCH
Major Finding: 41% of 90 patients with low serum VEGF showed clinical benefit with ipilimumab, compared with 23% of the 69 patients with a high VEGF level.
Data Source: A preliminary study involving 159 patients with advanced melanoma who received 12 weeks of ipilimumab therapy, of whom 33% showed clinical benefit ranging from disease stabilization to complete eradication of the tumor.
Disclosures: This study was supported by the National Institutes of Health. Four of Dr. Yuan’s associates reported receiving support from and serving as consultants for Bristol-Myers Squibb, which developed the monoclonal antibody ipilimumab.
IBD linked to increased risk of certain cancers
Immunosuppressive therapies for inflammatory bowel disease do not appear to be implicated in the increased risk for melanomas and hematologic malignancies seen in these patients, based on the results of a large database analysis and a separate meta-analysis published in the February edition of Clinical Gastroenterology and Hepatology.
The first study, based on a nationwide population-based cohort analysis conducted in Denmark, found that patients with inflammatory bowel disease (IBD) are at increased risk for cancer, in particular hematologic malignancies and melanoma at standardized incidence ratios of 1.9 and 1.4, respectively.
The second study, based on a systematic review and meta-analysis of the medical literature, found a diagnosis of Crohn’s disease or ulcerative colitis was associated with a 37% increase in the risk of developing melanoma. Melanoma risk was higher in studies performed before the introduction of biologic therapies in 1998, but not in those studies performed after 1998.
For the first study, researchers led by Dr. Michael D. Kappelman of the division of pediatric gastroenterology at the University of North Carolina at Chapel Hill used health care databases to identify patients in Denmark with a diagnosis of Crohn’s disease or ulcerative colitis from 1978 through 2010 (doi: 10.1016/j.cgh.2013.03.034). They followed the patients until the first occurrence of cancer, death, or emigration and used standardized incidence ratios (SIRs) to compare cancer incidence in patients with IBD with that expected in the general population.
"If immunosuppressive medications reduce the risk of gastrointestinal malignancy by suppressing intestinal inflammation but increase the risk of extraintestinal malignancies, IBD patients may be trading off one set of risks for another," the researchers wrote. "We therefore sought to evaluate both intestinal and extraintestinal malignancies comprehensively, as well as the occurrence of any invasive cancer, in a nationwide cohort of patients with IBD in Denmark."
The analysis included 13,756 patients with Crohn’s disease and 35,152 patients with ulcerative colitis who were followed up for a mean of nearly 8 years. After the exclusion of cancers diagnosed within 1 year of IBD diagnosis, 772 cases of invasive cancer occurred among patients with Crohn’s disease (SIR 1.3) and 2,331 occurred among patients with ulcerative colitis (SIR 1.1). Diagnosis of Crohn’s disease was weakly associated with gastrointestinal cancers (SIR 1.2) and extraintestinal cancers (SIR 1.3), with the strongest associations for hematologic malignancies (SIR 1.9), smoking-related cancers (SIR 1.5), and melanoma (SIR 1.4). Associations between ulcerative colitis and gastrointestinal and extraintestinal cancers were weaker (SIR of 1.1 for both).
The excess risk of cancer in patients with Crohn’s disease "largely is owing to extraintestinal cancers, particularly hematologic malignancies and melanoma, both of which may be related to immune suppression, and smoking-related cancers," the researchers concluded. They added that their study "provides reassuring data that the decreasing risk for gastrointestinal malignancy observed here ... has not been offset by a concomitant increase in the risk of extraintestinal or hematologic malignancies. These findings suggest that changes in the medical and surgical treatment of IBD and/or other secular trends have not substantially impacted cancer relative risk on a population basis."
The study was supported in part by grants from the National Institute for Diabetes and Digestive and Kidney Diseases, the Karen Elise Jensen Foundation, and the Clinical Epidemiological Research Foundation.
In the second study from the journal, researchers led by Dr. Suddharth Singh of the division of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn., conducted a systematic literature search through March 2013 for cohort studies showing incident melanoma after IBD diagnosis and an estimate of incident rate ratio or standardized incidence rate. Case reports or case series were excluded (doi: 10.1016/j.cgh.2013.04.033).
Of the 838 studies identified, 12 that comprised 172,837 patients with IBD were included in the final analysis. The pooled crude incidence rate of melanoma among patients in the 12 studies was 27.7/100,000 person-years. This translated into a 37% increase in the risk of melanoma (risk ratio of 1.37). The increase was observed independently in the patients with Crohn’s disease (RR 1.51) and in those with ulcerative colitis (RR 1.23). In addition, the risk of melanoma was higher in studies conducted prior to 1998, when biologic therapies were introduced (RR 1.52), but not in those conducted after 1998 (RR 1.08).
"The risk of melanoma has been shown to be increased in immunosuppressed patients, including patients with a prior solid organ transplant, lymphoma, and patients with human immunodeficiency virus infection and acquired immunodeficiency syndrome. It is possible that the increased risk of melanoma observed in patients with IBD may be related to underlying immune dysfunction in these patients, resulting in altered tumor surveillance" the researchers wrote.
They hypothesized that immunosuppressive medications may increase the risk of melanoma by "down-regulation of the tumor surveillance mechanisms, increased susceptibility to infection with oncogenic viruses such as melanoma-associated retroviruses, or through direct pharmacologic effects of medications on DNA metabolism. Although thiopurine analogs have been associated with an increased risk of nonmelanoma skin cancers, our pooled analysis did not suggest an increased risk of melanoma, albeit there were a limited number of studies."
Dr. Singh and his associates acknowledged certain limitations of the analysis, including the potential for misclassification bias in the included studies and the fact that studies "did not adjust for known risk factors for melanoma, including personal history of nonmelanoma skin cancers, family history of melanoma, or sun exposure."
For the meta-analysis, one author, Dr. Edward V. Loftus, disclosed that he has consulted for and has received research support from Janssen Biotech, Abbott Laboratories, and UCB Pharma. The others had no relevant conflicts to disclose.
Immunosuppressive therapies for inflammatory bowel disease do not appear to be implicated in the increased risk for melanomas and hematologic malignancies seen in these patients, based on the results of a large database analysis and a separate meta-analysis published in the February edition of Clinical Gastroenterology and Hepatology.
The first study, based on a nationwide population-based cohort analysis conducted in Denmark, found that patients with inflammatory bowel disease (IBD) are at increased risk for cancer, in particular hematologic malignancies and melanoma at standardized incidence ratios of 1.9 and 1.4, respectively.
The second study, based on a systematic review and meta-analysis of the medical literature, found a diagnosis of Crohn’s disease or ulcerative colitis was associated with a 37% increase in the risk of developing melanoma. Melanoma risk was higher in studies performed before the introduction of biologic therapies in 1998, but not in those studies performed after 1998.
For the first study, researchers led by Dr. Michael D. Kappelman of the division of pediatric gastroenterology at the University of North Carolina at Chapel Hill used health care databases to identify patients in Denmark with a diagnosis of Crohn’s disease or ulcerative colitis from 1978 through 2010 (doi: 10.1016/j.cgh.2013.03.034). They followed the patients until the first occurrence of cancer, death, or emigration and used standardized incidence ratios (SIRs) to compare cancer incidence in patients with IBD with that expected in the general population.
"If immunosuppressive medications reduce the risk of gastrointestinal malignancy by suppressing intestinal inflammation but increase the risk of extraintestinal malignancies, IBD patients may be trading off one set of risks for another," the researchers wrote. "We therefore sought to evaluate both intestinal and extraintestinal malignancies comprehensively, as well as the occurrence of any invasive cancer, in a nationwide cohort of patients with IBD in Denmark."
The analysis included 13,756 patients with Crohn’s disease and 35,152 patients with ulcerative colitis who were followed up for a mean of nearly 8 years. After the exclusion of cancers diagnosed within 1 year of IBD diagnosis, 772 cases of invasive cancer occurred among patients with Crohn’s disease (SIR 1.3) and 2,331 occurred among patients with ulcerative colitis (SIR 1.1). Diagnosis of Crohn’s disease was weakly associated with gastrointestinal cancers (SIR 1.2) and extraintestinal cancers (SIR 1.3), with the strongest associations for hematologic malignancies (SIR 1.9), smoking-related cancers (SIR 1.5), and melanoma (SIR 1.4). Associations between ulcerative colitis and gastrointestinal and extraintestinal cancers were weaker (SIR of 1.1 for both).
The excess risk of cancer in patients with Crohn’s disease "largely is owing to extraintestinal cancers, particularly hematologic malignancies and melanoma, both of which may be related to immune suppression, and smoking-related cancers," the researchers concluded. They added that their study "provides reassuring data that the decreasing risk for gastrointestinal malignancy observed here ... has not been offset by a concomitant increase in the risk of extraintestinal or hematologic malignancies. These findings suggest that changes in the medical and surgical treatment of IBD and/or other secular trends have not substantially impacted cancer relative risk on a population basis."
The study was supported in part by grants from the National Institute for Diabetes and Digestive and Kidney Diseases, the Karen Elise Jensen Foundation, and the Clinical Epidemiological Research Foundation.
In the second study from the journal, researchers led by Dr. Suddharth Singh of the division of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn., conducted a systematic literature search through March 2013 for cohort studies showing incident melanoma after IBD diagnosis and an estimate of incident rate ratio or standardized incidence rate. Case reports or case series were excluded (doi: 10.1016/j.cgh.2013.04.033).
Of the 838 studies identified, 12 that comprised 172,837 patients with IBD were included in the final analysis. The pooled crude incidence rate of melanoma among patients in the 12 studies was 27.7/100,000 person-years. This translated into a 37% increase in the risk of melanoma (risk ratio of 1.37). The increase was observed independently in the patients with Crohn’s disease (RR 1.51) and in those with ulcerative colitis (RR 1.23). In addition, the risk of melanoma was higher in studies conducted prior to 1998, when biologic therapies were introduced (RR 1.52), but not in those conducted after 1998 (RR 1.08).
"The risk of melanoma has been shown to be increased in immunosuppressed patients, including patients with a prior solid organ transplant, lymphoma, and patients with human immunodeficiency virus infection and acquired immunodeficiency syndrome. It is possible that the increased risk of melanoma observed in patients with IBD may be related to underlying immune dysfunction in these patients, resulting in altered tumor surveillance" the researchers wrote.
They hypothesized that immunosuppressive medications may increase the risk of melanoma by "down-regulation of the tumor surveillance mechanisms, increased susceptibility to infection with oncogenic viruses such as melanoma-associated retroviruses, or through direct pharmacologic effects of medications on DNA metabolism. Although thiopurine analogs have been associated with an increased risk of nonmelanoma skin cancers, our pooled analysis did not suggest an increased risk of melanoma, albeit there were a limited number of studies."
Dr. Singh and his associates acknowledged certain limitations of the analysis, including the potential for misclassification bias in the included studies and the fact that studies "did not adjust for known risk factors for melanoma, including personal history of nonmelanoma skin cancers, family history of melanoma, or sun exposure."
For the meta-analysis, one author, Dr. Edward V. Loftus, disclosed that he has consulted for and has received research support from Janssen Biotech, Abbott Laboratories, and UCB Pharma. The others had no relevant conflicts to disclose.
Immunosuppressive therapies for inflammatory bowel disease do not appear to be implicated in the increased risk for melanomas and hematologic malignancies seen in these patients, based on the results of a large database analysis and a separate meta-analysis published in the February edition of Clinical Gastroenterology and Hepatology.
The first study, based on a nationwide population-based cohort analysis conducted in Denmark, found that patients with inflammatory bowel disease (IBD) are at increased risk for cancer, in particular hematologic malignancies and melanoma at standardized incidence ratios of 1.9 and 1.4, respectively.
The second study, based on a systematic review and meta-analysis of the medical literature, found a diagnosis of Crohn’s disease or ulcerative colitis was associated with a 37% increase in the risk of developing melanoma. Melanoma risk was higher in studies performed before the introduction of biologic therapies in 1998, but not in those studies performed after 1998.
For the first study, researchers led by Dr. Michael D. Kappelman of the division of pediatric gastroenterology at the University of North Carolina at Chapel Hill used health care databases to identify patients in Denmark with a diagnosis of Crohn’s disease or ulcerative colitis from 1978 through 2010 (doi: 10.1016/j.cgh.2013.03.034). They followed the patients until the first occurrence of cancer, death, or emigration and used standardized incidence ratios (SIRs) to compare cancer incidence in patients with IBD with that expected in the general population.
"If immunosuppressive medications reduce the risk of gastrointestinal malignancy by suppressing intestinal inflammation but increase the risk of extraintestinal malignancies, IBD patients may be trading off one set of risks for another," the researchers wrote. "We therefore sought to evaluate both intestinal and extraintestinal malignancies comprehensively, as well as the occurrence of any invasive cancer, in a nationwide cohort of patients with IBD in Denmark."
The analysis included 13,756 patients with Crohn’s disease and 35,152 patients with ulcerative colitis who were followed up for a mean of nearly 8 years. After the exclusion of cancers diagnosed within 1 year of IBD diagnosis, 772 cases of invasive cancer occurred among patients with Crohn’s disease (SIR 1.3) and 2,331 occurred among patients with ulcerative colitis (SIR 1.1). Diagnosis of Crohn’s disease was weakly associated with gastrointestinal cancers (SIR 1.2) and extraintestinal cancers (SIR 1.3), with the strongest associations for hematologic malignancies (SIR 1.9), smoking-related cancers (SIR 1.5), and melanoma (SIR 1.4). Associations between ulcerative colitis and gastrointestinal and extraintestinal cancers were weaker (SIR of 1.1 for both).
The excess risk of cancer in patients with Crohn’s disease "largely is owing to extraintestinal cancers, particularly hematologic malignancies and melanoma, both of which may be related to immune suppression, and smoking-related cancers," the researchers concluded. They added that their study "provides reassuring data that the decreasing risk for gastrointestinal malignancy observed here ... has not been offset by a concomitant increase in the risk of extraintestinal or hematologic malignancies. These findings suggest that changes in the medical and surgical treatment of IBD and/or other secular trends have not substantially impacted cancer relative risk on a population basis."
The study was supported in part by grants from the National Institute for Diabetes and Digestive and Kidney Diseases, the Karen Elise Jensen Foundation, and the Clinical Epidemiological Research Foundation.
In the second study from the journal, researchers led by Dr. Suddharth Singh of the division of gastroenterology and hepatology at Mayo Clinic, Rochester, Minn., conducted a systematic literature search through March 2013 for cohort studies showing incident melanoma after IBD diagnosis and an estimate of incident rate ratio or standardized incidence rate. Case reports or case series were excluded (doi: 10.1016/j.cgh.2013.04.033).
Of the 838 studies identified, 12 that comprised 172,837 patients with IBD were included in the final analysis. The pooled crude incidence rate of melanoma among patients in the 12 studies was 27.7/100,000 person-years. This translated into a 37% increase in the risk of melanoma (risk ratio of 1.37). The increase was observed independently in the patients with Crohn’s disease (RR 1.51) and in those with ulcerative colitis (RR 1.23). In addition, the risk of melanoma was higher in studies conducted prior to 1998, when biologic therapies were introduced (RR 1.52), but not in those conducted after 1998 (RR 1.08).
"The risk of melanoma has been shown to be increased in immunosuppressed patients, including patients with a prior solid organ transplant, lymphoma, and patients with human immunodeficiency virus infection and acquired immunodeficiency syndrome. It is possible that the increased risk of melanoma observed in patients with IBD may be related to underlying immune dysfunction in these patients, resulting in altered tumor surveillance" the researchers wrote.
They hypothesized that immunosuppressive medications may increase the risk of melanoma by "down-regulation of the tumor surveillance mechanisms, increased susceptibility to infection with oncogenic viruses such as melanoma-associated retroviruses, or through direct pharmacologic effects of medications on DNA metabolism. Although thiopurine analogs have been associated with an increased risk of nonmelanoma skin cancers, our pooled analysis did not suggest an increased risk of melanoma, albeit there were a limited number of studies."
Dr. Singh and his associates acknowledged certain limitations of the analysis, including the potential for misclassification bias in the included studies and the fact that studies "did not adjust for known risk factors for melanoma, including personal history of nonmelanoma skin cancers, family history of melanoma, or sun exposure."
For the meta-analysis, one author, Dr. Edward V. Loftus, disclosed that he has consulted for and has received research support from Janssen Biotech, Abbott Laboratories, and UCB Pharma. The others had no relevant conflicts to disclose.
FROM CLINICAL GASTROENTEROLOGY AND hEPATOLOGY
Major finding: In a population-based cohort study, patients with inflammatory bowel disease (IBD) were found to be at increased risk for gastrointestinal and extraintestinal cancer, particularly hematologic malignancies and melanoma (standardized incidence ratios of 1.9 and 1.4, respectively). In a separate meta-analysis, patients with IBD were found to be at 37% increased risk of developing melanoma.
Data source: A population-based study from Denmark involving 48,908 patients with IBD and a meta-analysis involving 172,837 patients with IBD.
Disclosures: The population-based study was supported in part by grants from the National Institute for Diabetes and Digestive and Kidney Diseases, the Karen Elise Jensen Foundation, and the Clinical Epidemiological Research Foundation. For the meta-analysis, one author, Dr. Edward V. Loftus, disclosed that he has consulted for and has received research support from Janssen Biotech, Abbott Laboratories, and UCB Pharma. The others had no relevant conflicts to disclose.
Poor accrual halts one in five cancer clinical trials
Almost one-fifth of adult cancer clinical trials fail to reach completion for reasons unrelated to efficacy or adverse effects, according to data being reported at the 2014 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology. Poor accrual is the leading cause.
Trials were more likely to fail completion if they were phase II, single center, funded by industry, or conducted solely in the United States. Trials among patients with genitourinary cancers were no more or less likely to fail than trials among patients with other cancers.
"These findings really underscore the clinical trial accrual problem that we have in the United States. Not only does poor accrual lead to more expensive trials and trials that generate answers much more slowly, but it also prevents many trials from generating answers at all," senior author Dr. Matthew D. Galsky, director of the genitourinary medical oncology program, Icahn School of Medicine at Mount Sinai, New York, said in a press briefing at the 2014 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology.
The study is not meant to indict any specific stakeholders, Dr. Galsky stressed. "Rather, what we wanted to do was hold up a mirror to our activities as a cancer clinical trials community and really ask whether the system is optimized to bring better treatments to our patients as efficiently as possible. And clearly there is some work to do.
"Based on this analysis and others, it’s apparent that we need better collaboration and communication within the system and to use novel approaches to increase accrual to cancer clinical trials, which has really been quite steady at 3% to 5% of the adult cancer population for decades."
"This is a really interesting presentation which casts some light on one of the major frustrations that I think ... our whole community shares in designing and implementing clinical trials," said press briefing moderator Dr. Charles J. Ryan, leader of the genitourinary medical oncology program at the University of California, San Francisco. "Hopefully, this may be the beginning of a broader discussion that helps improve some of the efficiencies here."
Dr. Galsky speculated that the explanation for poor accrual is multifactorial. "Clearly, we need to engage patients more in the design of trials. We need to design trials that are more pragmatic; eligibility for trials is often so restrictive that only the patients who are the fittest and represent kind of an extreme of cancer patients can actually enroll," he said.
Other issues likely include financial barriers (such as whether insurers reimburse the costs of care for patients in trials); geographic accessibility; and the time and regulatory burdens of participating, especially as they affect community oncology practices. "Addressing the cancer clinical trials enterprise at large requires making the burden of participation lower because that does address accrual, that does address generalizability, that does address accessibility. It’s a problem that needs to be part of this conversation," Dr. Galsky maintained.
Dr. Ryan, the moderator, cited rapid treatment advances as yet another possible reason for poor accrual. "Some trials may take 2 or 3 years to accrue. During the first year, the standards of care may be one thing, and during the third year, the standards of care may have changed. We are really seeing very rapid evolution in our standards of care, so that could be one issue," he elaborated.
Introducing the study, Dr. Galsky noted that trials that fail to complete (that is, close without enrolling the intended number of patients) represent a major inefficiency of the cancer clinical trials enterprise. "Such trials contribute little knowledge, waste finite resources, and potentially divert patients from participating in other trials," he said.
The Institute of Medicine previously issued a report suggesting that about 40% of trials sponsored by the National Cancer Institute fail to achieve completion. But such trials account for only about 15% of all cancer trials.
The researchers analyzed 7,776 phase II or III interventional adult cancer clinical trials registered on ClinicalTrials.gov that had start dates between 2005 and 2011. They searched for those that had failed to reach completion, meaning that the trial had been stopped and had a "terminated" or "withdrawn" status.
The trials had a total of about 48,000 patients. Overall, 10% were trials in prostate, kidney, bladder, or testicular cancer.
The cumulative incidence of failure to reach completion for reasons unrelated to the efficacy or safety of the intervention was about 20%, according to Dr. Galsky.
Among all noncompleted trials, the largest share, 39%, failed to achieve completion because of poor accrual, topping other reasons such as logistics and efficacy/safety.
Trials were more likely to fail completion if they were funded by industry as compared with the federal government (hazard ratio, 1.97), were phase II as compared with phase III (HR, 1.29), or were single center as compared with multicenter (HR, 1.93).
On the other hand, trials were less likely to fail completion if they were conducted solely outside the United States (HR, 0.65) or both in and outside the United States (HR, 0.67), as compared with solely in the United States.
Dr. Galsky disclosed no relevant conflicts of interest.
Almost one-fifth of adult cancer clinical trials fail to reach completion for reasons unrelated to efficacy or adverse effects, according to data being reported at the 2014 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology. Poor accrual is the leading cause.
Trials were more likely to fail completion if they were phase II, single center, funded by industry, or conducted solely in the United States. Trials among patients with genitourinary cancers were no more or less likely to fail than trials among patients with other cancers.
"These findings really underscore the clinical trial accrual problem that we have in the United States. Not only does poor accrual lead to more expensive trials and trials that generate answers much more slowly, but it also prevents many trials from generating answers at all," senior author Dr. Matthew D. Galsky, director of the genitourinary medical oncology program, Icahn School of Medicine at Mount Sinai, New York, said in a press briefing at the 2014 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology.
The study is not meant to indict any specific stakeholders, Dr. Galsky stressed. "Rather, what we wanted to do was hold up a mirror to our activities as a cancer clinical trials community and really ask whether the system is optimized to bring better treatments to our patients as efficiently as possible. And clearly there is some work to do.
"Based on this analysis and others, it’s apparent that we need better collaboration and communication within the system and to use novel approaches to increase accrual to cancer clinical trials, which has really been quite steady at 3% to 5% of the adult cancer population for decades."
"This is a really interesting presentation which casts some light on one of the major frustrations that I think ... our whole community shares in designing and implementing clinical trials," said press briefing moderator Dr. Charles J. Ryan, leader of the genitourinary medical oncology program at the University of California, San Francisco. "Hopefully, this may be the beginning of a broader discussion that helps improve some of the efficiencies here."
Dr. Galsky speculated that the explanation for poor accrual is multifactorial. "Clearly, we need to engage patients more in the design of trials. We need to design trials that are more pragmatic; eligibility for trials is often so restrictive that only the patients who are the fittest and represent kind of an extreme of cancer patients can actually enroll," he said.
Other issues likely include financial barriers (such as whether insurers reimburse the costs of care for patients in trials); geographic accessibility; and the time and regulatory burdens of participating, especially as they affect community oncology practices. "Addressing the cancer clinical trials enterprise at large requires making the burden of participation lower because that does address accrual, that does address generalizability, that does address accessibility. It’s a problem that needs to be part of this conversation," Dr. Galsky maintained.
Dr. Ryan, the moderator, cited rapid treatment advances as yet another possible reason for poor accrual. "Some trials may take 2 or 3 years to accrue. During the first year, the standards of care may be one thing, and during the third year, the standards of care may have changed. We are really seeing very rapid evolution in our standards of care, so that could be one issue," he elaborated.
Introducing the study, Dr. Galsky noted that trials that fail to complete (that is, close without enrolling the intended number of patients) represent a major inefficiency of the cancer clinical trials enterprise. "Such trials contribute little knowledge, waste finite resources, and potentially divert patients from participating in other trials," he said.
The Institute of Medicine previously issued a report suggesting that about 40% of trials sponsored by the National Cancer Institute fail to achieve completion. But such trials account for only about 15% of all cancer trials.
The researchers analyzed 7,776 phase II or III interventional adult cancer clinical trials registered on ClinicalTrials.gov that had start dates between 2005 and 2011. They searched for those that had failed to reach completion, meaning that the trial had been stopped and had a "terminated" or "withdrawn" status.
The trials had a total of about 48,000 patients. Overall, 10% were trials in prostate, kidney, bladder, or testicular cancer.
The cumulative incidence of failure to reach completion for reasons unrelated to the efficacy or safety of the intervention was about 20%, according to Dr. Galsky.
Among all noncompleted trials, the largest share, 39%, failed to achieve completion because of poor accrual, topping other reasons such as logistics and efficacy/safety.
Trials were more likely to fail completion if they were funded by industry as compared with the federal government (hazard ratio, 1.97), were phase II as compared with phase III (HR, 1.29), or were single center as compared with multicenter (HR, 1.93).
On the other hand, trials were less likely to fail completion if they were conducted solely outside the United States (HR, 0.65) or both in and outside the United States (HR, 0.67), as compared with solely in the United States.
Dr. Galsky disclosed no relevant conflicts of interest.
Almost one-fifth of adult cancer clinical trials fail to reach completion for reasons unrelated to efficacy or adverse effects, according to data being reported at the 2014 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology. Poor accrual is the leading cause.
Trials were more likely to fail completion if they were phase II, single center, funded by industry, or conducted solely in the United States. Trials among patients with genitourinary cancers were no more or less likely to fail than trials among patients with other cancers.
"These findings really underscore the clinical trial accrual problem that we have in the United States. Not only does poor accrual lead to more expensive trials and trials that generate answers much more slowly, but it also prevents many trials from generating answers at all," senior author Dr. Matthew D. Galsky, director of the genitourinary medical oncology program, Icahn School of Medicine at Mount Sinai, New York, said in a press briefing at the 2014 Genitourinary Cancers Symposium sponsored by the American Society of Clinical Oncology.
The study is not meant to indict any specific stakeholders, Dr. Galsky stressed. "Rather, what we wanted to do was hold up a mirror to our activities as a cancer clinical trials community and really ask whether the system is optimized to bring better treatments to our patients as efficiently as possible. And clearly there is some work to do.
"Based on this analysis and others, it’s apparent that we need better collaboration and communication within the system and to use novel approaches to increase accrual to cancer clinical trials, which has really been quite steady at 3% to 5% of the adult cancer population for decades."
"This is a really interesting presentation which casts some light on one of the major frustrations that I think ... our whole community shares in designing and implementing clinical trials," said press briefing moderator Dr. Charles J. Ryan, leader of the genitourinary medical oncology program at the University of California, San Francisco. "Hopefully, this may be the beginning of a broader discussion that helps improve some of the efficiencies here."
Dr. Galsky speculated that the explanation for poor accrual is multifactorial. "Clearly, we need to engage patients more in the design of trials. We need to design trials that are more pragmatic; eligibility for trials is often so restrictive that only the patients who are the fittest and represent kind of an extreme of cancer patients can actually enroll," he said.
Other issues likely include financial barriers (such as whether insurers reimburse the costs of care for patients in trials); geographic accessibility; and the time and regulatory burdens of participating, especially as they affect community oncology practices. "Addressing the cancer clinical trials enterprise at large requires making the burden of participation lower because that does address accrual, that does address generalizability, that does address accessibility. It’s a problem that needs to be part of this conversation," Dr. Galsky maintained.
Dr. Ryan, the moderator, cited rapid treatment advances as yet another possible reason for poor accrual. "Some trials may take 2 or 3 years to accrue. During the first year, the standards of care may be one thing, and during the third year, the standards of care may have changed. We are really seeing very rapid evolution in our standards of care, so that could be one issue," he elaborated.
Introducing the study, Dr. Galsky noted that trials that fail to complete (that is, close without enrolling the intended number of patients) represent a major inefficiency of the cancer clinical trials enterprise. "Such trials contribute little knowledge, waste finite resources, and potentially divert patients from participating in other trials," he said.
The Institute of Medicine previously issued a report suggesting that about 40% of trials sponsored by the National Cancer Institute fail to achieve completion. But such trials account for only about 15% of all cancer trials.
The researchers analyzed 7,776 phase II or III interventional adult cancer clinical trials registered on ClinicalTrials.gov that had start dates between 2005 and 2011. They searched for those that had failed to reach completion, meaning that the trial had been stopped and had a "terminated" or "withdrawn" status.
The trials had a total of about 48,000 patients. Overall, 10% were trials in prostate, kidney, bladder, or testicular cancer.
The cumulative incidence of failure to reach completion for reasons unrelated to the efficacy or safety of the intervention was about 20%, according to Dr. Galsky.
Among all noncompleted trials, the largest share, 39%, failed to achieve completion because of poor accrual, topping other reasons such as logistics and efficacy/safety.
Trials were more likely to fail completion if they were funded by industry as compared with the federal government (hazard ratio, 1.97), were phase II as compared with phase III (HR, 1.29), or were single center as compared with multicenter (HR, 1.93).
On the other hand, trials were less likely to fail completion if they were conducted solely outside the United States (HR, 0.65) or both in and outside the United States (HR, 0.67), as compared with solely in the United States.
Dr. Galsky disclosed no relevant conflicts of interest.
AT THE GENITOURINARY CANCERS SYMPOSIUM
Major finding: About 20% of cancer clinical trials fail to reach completion for reasons unrelated to efficacy or toxicity, and the most common reason is poor accrual.
Data source: An analysis of 7,776 phase II and III interventional clinical trials involving adults with cancer.
Disclosures: Dr. Galsky disclosed no relevant conflicts of interest.