User login
House votes to speed sunscreen approvals
The U.S. House of Representatives passed legislation to speed up Food and Drug Administration approvals of new sunscreen ingredients and to eliminate a backlog.
The Sunscreen Innovation Act (H.R. 4250) was passed by a voice vote and now awaits action by the Senate. The companion bill in the Senate, S. 2141, was introduced by Jack Reed (D-R.I.) and has been referred to the Committee on Health, Education, Labor, and Pensions but has not received a hearing yet.
Backers of the legislation are encouraging the Senate to move quickly.
"Even though the Food and Drug Administration has listed action on sunscreen ingredient applications as a priority since 2008, no new sunscreen ingredients have been approved by the FDA in 15 years," Rep. Ed Whitfield (R-Ky.), one of the lead sponsors of the House bill, said in a statement after the approval.
"The framework outlined in this legislation strikes an appropriate balance between consumer safety and access to new sunscreen products," he said, adding, "I am pleased to see my legislation pass the House of Representatives, and call on the Senate to act."
The bill requires the FDA to make final decisions within a year on the backlog of ingredients under review, and within a year and a half on new applications. It also sets up more Congressional oversight of the process and makes "sunscreens that have been marketed for five continuous years in the United States or other countries and in sufficient quantity eligible for review under this Act."
Currently, according to the American Cancer Society Cancer Action Network (ACS CAN), only three of the seven UVA-blocking ingredients sold in Europe are approved in the United States.
The Sunscreen Innovation Act has many supporters in the health care field and among consumer advocates and manufacturers, many of whom belong to the PASS (Public Access to SunScreens) Coalition. In a statement issued by the coalition, the American College of Mohs Surgery said, "This important legislation will go a long way in helping prevent skin cancers by improving the availability of more effective sunscreen products, and we are committed to its passage."
The Environmental Working Group, a frequent critic of the FDA’s regulation of sunscreens and of manufacturers, also applauded House passage of the bill.
The American Cancer Society was pleased as well. "The House took a critical step today to fix a broken process at FDA for the review of new sunscreen ingredients that could potentially help more Americans prevent skin cancer," Chris Hansen, president of the ACS CAN, said in a statement.
"American consumers should have access to the broadest choice of sunscreens – including those in use for years in other countries – once they are shown to be safe and effective," he noted.
On Twitter @aliciaault
The U.S. House of Representatives passed legislation to speed up Food and Drug Administration approvals of new sunscreen ingredients and to eliminate a backlog.
The Sunscreen Innovation Act (H.R. 4250) was passed by a voice vote and now awaits action by the Senate. The companion bill in the Senate, S. 2141, was introduced by Jack Reed (D-R.I.) and has been referred to the Committee on Health, Education, Labor, and Pensions but has not received a hearing yet.
Backers of the legislation are encouraging the Senate to move quickly.
"Even though the Food and Drug Administration has listed action on sunscreen ingredient applications as a priority since 2008, no new sunscreen ingredients have been approved by the FDA in 15 years," Rep. Ed Whitfield (R-Ky.), one of the lead sponsors of the House bill, said in a statement after the approval.
"The framework outlined in this legislation strikes an appropriate balance between consumer safety and access to new sunscreen products," he said, adding, "I am pleased to see my legislation pass the House of Representatives, and call on the Senate to act."
The bill requires the FDA to make final decisions within a year on the backlog of ingredients under review, and within a year and a half on new applications. It also sets up more Congressional oversight of the process and makes "sunscreens that have been marketed for five continuous years in the United States or other countries and in sufficient quantity eligible for review under this Act."
Currently, according to the American Cancer Society Cancer Action Network (ACS CAN), only three of the seven UVA-blocking ingredients sold in Europe are approved in the United States.
The Sunscreen Innovation Act has many supporters in the health care field and among consumer advocates and manufacturers, many of whom belong to the PASS (Public Access to SunScreens) Coalition. In a statement issued by the coalition, the American College of Mohs Surgery said, "This important legislation will go a long way in helping prevent skin cancers by improving the availability of more effective sunscreen products, and we are committed to its passage."
The Environmental Working Group, a frequent critic of the FDA’s regulation of sunscreens and of manufacturers, also applauded House passage of the bill.
The American Cancer Society was pleased as well. "The House took a critical step today to fix a broken process at FDA for the review of new sunscreen ingredients that could potentially help more Americans prevent skin cancer," Chris Hansen, president of the ACS CAN, said in a statement.
"American consumers should have access to the broadest choice of sunscreens – including those in use for years in other countries – once they are shown to be safe and effective," he noted.
On Twitter @aliciaault
The U.S. House of Representatives passed legislation to speed up Food and Drug Administration approvals of new sunscreen ingredients and to eliminate a backlog.
The Sunscreen Innovation Act (H.R. 4250) was passed by a voice vote and now awaits action by the Senate. The companion bill in the Senate, S. 2141, was introduced by Jack Reed (D-R.I.) and has been referred to the Committee on Health, Education, Labor, and Pensions but has not received a hearing yet.
Backers of the legislation are encouraging the Senate to move quickly.
"Even though the Food and Drug Administration has listed action on sunscreen ingredient applications as a priority since 2008, no new sunscreen ingredients have been approved by the FDA in 15 years," Rep. Ed Whitfield (R-Ky.), one of the lead sponsors of the House bill, said in a statement after the approval.
"The framework outlined in this legislation strikes an appropriate balance between consumer safety and access to new sunscreen products," he said, adding, "I am pleased to see my legislation pass the House of Representatives, and call on the Senate to act."
The bill requires the FDA to make final decisions within a year on the backlog of ingredients under review, and within a year and a half on new applications. It also sets up more Congressional oversight of the process and makes "sunscreens that have been marketed for five continuous years in the United States or other countries and in sufficient quantity eligible for review under this Act."
Currently, according to the American Cancer Society Cancer Action Network (ACS CAN), only three of the seven UVA-blocking ingredients sold in Europe are approved in the United States.
The Sunscreen Innovation Act has many supporters in the health care field and among consumer advocates and manufacturers, many of whom belong to the PASS (Public Access to SunScreens) Coalition. In a statement issued by the coalition, the American College of Mohs Surgery said, "This important legislation will go a long way in helping prevent skin cancers by improving the availability of more effective sunscreen products, and we are committed to its passage."
The Environmental Working Group, a frequent critic of the FDA’s regulation of sunscreens and of manufacturers, also applauded House passage of the bill.
The American Cancer Society was pleased as well. "The House took a critical step today to fix a broken process at FDA for the review of new sunscreen ingredients that could potentially help more Americans prevent skin cancer," Chris Hansen, president of the ACS CAN, said in a statement.
"American consumers should have access to the broadest choice of sunscreens – including those in use for years in other countries – once they are shown to be safe and effective," he noted.
On Twitter @aliciaault
Surgeon General: Tanning, indoor tanning must stop
The U.S. Surgeon General’s office is calling on Americans to do more to help prevent skin cancer, saying that it is a growing public health problem.
The five-point call to action singled out ultraviolet light exposure – from indoor and outdoor tanning – as a major culprit in the growing incidence of all cancers, and melanoma, in particular.
"Tanned skin is damaged skin," Acting Surgeon General, Boris D. Lushniak, a dermatologist, said at a briefing to release the report.
"When people tan or get sunburned, they increase their risk of getting skin cancer later in life," he said, adding that the nation needs to change its cultural acceptance of tanning as a sign of health.
The report noted that about 5 million Americans are treated for skin cancer each year, at a cost of $8 billion. Melanoma is of the greatest concern; its incidence has tripled over the past 3 decades, with 63,000 new cases per year. Some 9,000 Americans die from melanoma each year, and death rates are on the rise in men, Dr. Howard Koh, assistant secretary for health at the U.S. Department of Health & Human Services, said at the briefing.
Both Dr. Koh and Dr. Lushniak singled out indoor tanning as particularly dangerous. The Surgeon General’s report cited research showing that indoor tanning may be associated with more than 400,000 cases of skin cancer, including 6,000 melanomas, per year.
Forty-four states and the District of Columbia have some type of indoor tanning laws or regulations. Eleven states – California, Delaware, Hawaii, Illinois, Louisiana, Minnesota, Nevada, Oregon, Texas, Vermont and Washington – prohibit indoor tanning by individuals under age 18. But indoor tanning remains popular: One in three white women aged 16-25 years engages in indoor tanning each year, according to the report.
In May, the Food and Drug Administration issued stricter rules for sunlamps used with indoor tanning, requiring a boxed warning that they should not be used on anyone younger than 18 years. The agency also required that more information on risks and contraindications be provided to sunlamp users, FDA Commissioner Margaret Hamburg noted in a statement on the Surgeon General’s report.
The five main points in the Surgeon General’s call to action are:
• Increase opportunities for sun protection in outdoor settings, such as increasing shade in recreational areas, and providing more protection for school children and outdoor workers.
• Give individuals information to make informed, healthy choices about UV exposure, including providing materials in workplaces and schools, and partnering with physicians and health systems.
• Promote policies that advance the goal of preventing skin cancer, such as encouraging electronic reporting of reportable skin cancers.
• Reduce harms from indoor tanning by enforcing existing laws and considering additional restrictions.
• Strengthen research, surveillance, monitoring, and evaluation.
The American Academy of Dermatology Association applauded the report.
"The American public needs to be aware that the dangers of ultraviolet radiation exposure are real," Dr. Brett M. Coldiron, president of the AADA, said in a statement. "The AADA is particularly pleased that the HHS and the Office of the Surgeon General have highlighted methods for the public to prevent skin cancer in this white paper, that include seeking shade, wearing protective clothing, applying sunscreen, and avoiding dangerous indoor tanning devices."
John Seffrin, Ph.D., chief executive officer of the American Cancer Society Cancer Action Network, noted that this is the first time there has been a national action plan regarding skin cancer.
"By bringing national attention to this growing public health crisis, the Surgeon General is calling on all of us to reinvigorate the fight against skin cancer," he said. "The Surgeon General’s Call to Action outlines achievable goals and strategies to support more Americans in making healthy choices about protecting their skin," Dr. Seffrin said.
On Twitter @aliciaault
The U.S. Surgeon General’s office is calling on Americans to do more to help prevent skin cancer, saying that it is a growing public health problem.
The five-point call to action singled out ultraviolet light exposure – from indoor and outdoor tanning – as a major culprit in the growing incidence of all cancers, and melanoma, in particular.
"Tanned skin is damaged skin," Acting Surgeon General, Boris D. Lushniak, a dermatologist, said at a briefing to release the report.
"When people tan or get sunburned, they increase their risk of getting skin cancer later in life," he said, adding that the nation needs to change its cultural acceptance of tanning as a sign of health.
The report noted that about 5 million Americans are treated for skin cancer each year, at a cost of $8 billion. Melanoma is of the greatest concern; its incidence has tripled over the past 3 decades, with 63,000 new cases per year. Some 9,000 Americans die from melanoma each year, and death rates are on the rise in men, Dr. Howard Koh, assistant secretary for health at the U.S. Department of Health & Human Services, said at the briefing.
Both Dr. Koh and Dr. Lushniak singled out indoor tanning as particularly dangerous. The Surgeon General’s report cited research showing that indoor tanning may be associated with more than 400,000 cases of skin cancer, including 6,000 melanomas, per year.
Forty-four states and the District of Columbia have some type of indoor tanning laws or regulations. Eleven states – California, Delaware, Hawaii, Illinois, Louisiana, Minnesota, Nevada, Oregon, Texas, Vermont and Washington – prohibit indoor tanning by individuals under age 18. But indoor tanning remains popular: One in three white women aged 16-25 years engages in indoor tanning each year, according to the report.
In May, the Food and Drug Administration issued stricter rules for sunlamps used with indoor tanning, requiring a boxed warning that they should not be used on anyone younger than 18 years. The agency also required that more information on risks and contraindications be provided to sunlamp users, FDA Commissioner Margaret Hamburg noted in a statement on the Surgeon General’s report.
The five main points in the Surgeon General’s call to action are:
• Increase opportunities for sun protection in outdoor settings, such as increasing shade in recreational areas, and providing more protection for school children and outdoor workers.
• Give individuals information to make informed, healthy choices about UV exposure, including providing materials in workplaces and schools, and partnering with physicians and health systems.
• Promote policies that advance the goal of preventing skin cancer, such as encouraging electronic reporting of reportable skin cancers.
• Reduce harms from indoor tanning by enforcing existing laws and considering additional restrictions.
• Strengthen research, surveillance, monitoring, and evaluation.
The American Academy of Dermatology Association applauded the report.
"The American public needs to be aware that the dangers of ultraviolet radiation exposure are real," Dr. Brett M. Coldiron, president of the AADA, said in a statement. "The AADA is particularly pleased that the HHS and the Office of the Surgeon General have highlighted methods for the public to prevent skin cancer in this white paper, that include seeking shade, wearing protective clothing, applying sunscreen, and avoiding dangerous indoor tanning devices."
John Seffrin, Ph.D., chief executive officer of the American Cancer Society Cancer Action Network, noted that this is the first time there has been a national action plan regarding skin cancer.
"By bringing national attention to this growing public health crisis, the Surgeon General is calling on all of us to reinvigorate the fight against skin cancer," he said. "The Surgeon General’s Call to Action outlines achievable goals and strategies to support more Americans in making healthy choices about protecting their skin," Dr. Seffrin said.
On Twitter @aliciaault
The U.S. Surgeon General’s office is calling on Americans to do more to help prevent skin cancer, saying that it is a growing public health problem.
The five-point call to action singled out ultraviolet light exposure – from indoor and outdoor tanning – as a major culprit in the growing incidence of all cancers, and melanoma, in particular.
"Tanned skin is damaged skin," Acting Surgeon General, Boris D. Lushniak, a dermatologist, said at a briefing to release the report.
"When people tan or get sunburned, they increase their risk of getting skin cancer later in life," he said, adding that the nation needs to change its cultural acceptance of tanning as a sign of health.
The report noted that about 5 million Americans are treated for skin cancer each year, at a cost of $8 billion. Melanoma is of the greatest concern; its incidence has tripled over the past 3 decades, with 63,000 new cases per year. Some 9,000 Americans die from melanoma each year, and death rates are on the rise in men, Dr. Howard Koh, assistant secretary for health at the U.S. Department of Health & Human Services, said at the briefing.
Both Dr. Koh and Dr. Lushniak singled out indoor tanning as particularly dangerous. The Surgeon General’s report cited research showing that indoor tanning may be associated with more than 400,000 cases of skin cancer, including 6,000 melanomas, per year.
Forty-four states and the District of Columbia have some type of indoor tanning laws or regulations. Eleven states – California, Delaware, Hawaii, Illinois, Louisiana, Minnesota, Nevada, Oregon, Texas, Vermont and Washington – prohibit indoor tanning by individuals under age 18. But indoor tanning remains popular: One in three white women aged 16-25 years engages in indoor tanning each year, according to the report.
In May, the Food and Drug Administration issued stricter rules for sunlamps used with indoor tanning, requiring a boxed warning that they should not be used on anyone younger than 18 years. The agency also required that more information on risks and contraindications be provided to sunlamp users, FDA Commissioner Margaret Hamburg noted in a statement on the Surgeon General’s report.
The five main points in the Surgeon General’s call to action are:
• Increase opportunities for sun protection in outdoor settings, such as increasing shade in recreational areas, and providing more protection for school children and outdoor workers.
• Give individuals information to make informed, healthy choices about UV exposure, including providing materials in workplaces and schools, and partnering with physicians and health systems.
• Promote policies that advance the goal of preventing skin cancer, such as encouraging electronic reporting of reportable skin cancers.
• Reduce harms from indoor tanning by enforcing existing laws and considering additional restrictions.
• Strengthen research, surveillance, monitoring, and evaluation.
The American Academy of Dermatology Association applauded the report.
"The American public needs to be aware that the dangers of ultraviolet radiation exposure are real," Dr. Brett M. Coldiron, president of the AADA, said in a statement. "The AADA is particularly pleased that the HHS and the Office of the Surgeon General have highlighted methods for the public to prevent skin cancer in this white paper, that include seeking shade, wearing protective clothing, applying sunscreen, and avoiding dangerous indoor tanning devices."
John Seffrin, Ph.D., chief executive officer of the American Cancer Society Cancer Action Network, noted that this is the first time there has been a national action plan regarding skin cancer.
"By bringing national attention to this growing public health crisis, the Surgeon General is calling on all of us to reinvigorate the fight against skin cancer," he said. "The Surgeon General’s Call to Action outlines achievable goals and strategies to support more Americans in making healthy choices about protecting their skin," Dr. Seffrin said.
On Twitter @aliciaault
ASCO at 50 … we’ve come a long way, we know a lot, but we’ve only just begun
I recently had both the pleasure and the challenge of attending ASCO 2014, the annual meeting of the American Society of Clinical Oncology. It was my first official ASCO meeting, and as an almost-third-year fellow in oncology, no amount of reading, research, scheduling, ASCO 2014 iPad app organizing, or even attending the day 1 early morning How to Navigate the Annual Meeting seminar could have prepared me for the experience…
Click on the PDF icon at the top of this introduction to read the full article.
I recently had both the pleasure and the challenge of attending ASCO 2014, the annual meeting of the American Society of Clinical Oncology. It was my first official ASCO meeting, and as an almost-third-year fellow in oncology, no amount of reading, research, scheduling, ASCO 2014 iPad app organizing, or even attending the day 1 early morning How to Navigate the Annual Meeting seminar could have prepared me for the experience…
Click on the PDF icon at the top of this introduction to read the full article.
I recently had both the pleasure and the challenge of attending ASCO 2014, the annual meeting of the American Society of Clinical Oncology. It was my first official ASCO meeting, and as an almost-third-year fellow in oncology, no amount of reading, research, scheduling, ASCO 2014 iPad app organizing, or even attending the day 1 early morning How to Navigate the Annual Meeting seminar could have prepared me for the experience…
Click on the PDF icon at the top of this introduction to read the full article.
Trametinib plus dabrafenib for unresectable or metastatic melanoma with BRAF V600E or V600K mutations
On January 9, 2014, the combination of trametinib and dabrafenib was granted accelerated approval by the US Food and Drug Administration for the treatment of patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations as detected by an FDA-approved test.1,2 Approval of the combination is based on durable response rate observed in an open-label study.2,3 Improvements in disease-related symptoms and overall survival have not yet been demonstrated for the combination. Both drugs were approved for use as single agents in this setting in May 2013.
Click on the PDF icon at the top of this introduction to read the full article.
On January 9, 2014, the combination of trametinib and dabrafenib was granted accelerated approval by the US Food and Drug Administration for the treatment of patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations as detected by an FDA-approved test.1,2 Approval of the combination is based on durable response rate observed in an open-label study.2,3 Improvements in disease-related symptoms and overall survival have not yet been demonstrated for the combination. Both drugs were approved for use as single agents in this setting in May 2013.
Click on the PDF icon at the top of this introduction to read the full article.
On January 9, 2014, the combination of trametinib and dabrafenib was granted accelerated approval by the US Food and Drug Administration for the treatment of patients with unresectable or metastatic melanoma with BRAF V600E or V600K mutations as detected by an FDA-approved test.1,2 Approval of the combination is based on durable response rate observed in an open-label study.2,3 Improvements in disease-related symptoms and overall survival have not yet been demonstrated for the combination. Both drugs were approved for use as single agents in this setting in May 2013.
Click on the PDF icon at the top of this introduction to read the full article.
Adding ingenol mebutate after cryosurgery enhances clearance of actinic keratoses
Complete clearance occurred at 12 months in 31% of patients who received field treatment with ingenol mebutate gel after cryotherapy for actinic keratoses, compared with 19% of placebo patients in a phase III trial.
The findings were published in the June issue of the Journal of Drugs in Dermatology.
Recurrence rates for AKs treated with cryotherapy alone are high, possibly because the treatment is lesion-directed and fails to address field cancerization, wrote Dr. Brian Berman of the University of Miami.
The FIELD study 1, a phase III, multicenter, randomized trial, showed that ingenol mebutate gel was safe and effective as a follow-up to cryosurgery for AKs. Short-term efficacy data (11 weeks) have been previously reported.
In this study, the patients were followed for 12 months (J. Drugs Dermatol. 2014;13:741-7).
"In phase III clinical trials, 2 or 3 days of field-directed therapy with ingenol mebutate demonstrated high and sustained clearance of AKs on the head and body, and was well tolerated," noted Dr. Berman and his coauthors.
The percentage reduction in the number of AKs was significantly higher at 12 months in the treatment group compared with the placebo group (68% vs. 54%), and significantly fewer patients in the treatment group developed new lesions in the treatment area compared with the placebo group (39% vs. 52%). In addition, the probability of sustained clearance of AKs at 12 months was 55% in the treatment group vs. 40% in the placebo group.
The study included 329 adults aged 18 years and older with 4-8 clinically typical, visible, and discrete AKs within a 25-cm2 treatment area on the face or scalp. The patients underwent cryosurgery, and 3 weeks later underwent field therapy with 0.015% ingenol mebutate gel or vehicle gel once a day for 3 consecutive days. Patients with suspected basal cell carcinoma or squamous cell carcinoma were excluded.
The findings were limited by the possible unblinding of treatment because of localized skin reactions, the researchers noted. However, few drug-related adverse events and no drug-related serious adverse events were reported, they said.
Dr. Berman has served as a consultant, received honoraria, and served on the speakers bureau for Leo Pharma, which funded the study.
Complete clearance occurred at 12 months in 31% of patients who received field treatment with ingenol mebutate gel after cryotherapy for actinic keratoses, compared with 19% of placebo patients in a phase III trial.
The findings were published in the June issue of the Journal of Drugs in Dermatology.
Recurrence rates for AKs treated with cryotherapy alone are high, possibly because the treatment is lesion-directed and fails to address field cancerization, wrote Dr. Brian Berman of the University of Miami.
The FIELD study 1, a phase III, multicenter, randomized trial, showed that ingenol mebutate gel was safe and effective as a follow-up to cryosurgery for AKs. Short-term efficacy data (11 weeks) have been previously reported.
In this study, the patients were followed for 12 months (J. Drugs Dermatol. 2014;13:741-7).
"In phase III clinical trials, 2 or 3 days of field-directed therapy with ingenol mebutate demonstrated high and sustained clearance of AKs on the head and body, and was well tolerated," noted Dr. Berman and his coauthors.
The percentage reduction in the number of AKs was significantly higher at 12 months in the treatment group compared with the placebo group (68% vs. 54%), and significantly fewer patients in the treatment group developed new lesions in the treatment area compared with the placebo group (39% vs. 52%). In addition, the probability of sustained clearance of AKs at 12 months was 55% in the treatment group vs. 40% in the placebo group.
The study included 329 adults aged 18 years and older with 4-8 clinically typical, visible, and discrete AKs within a 25-cm2 treatment area on the face or scalp. The patients underwent cryosurgery, and 3 weeks later underwent field therapy with 0.015% ingenol mebutate gel or vehicle gel once a day for 3 consecutive days. Patients with suspected basal cell carcinoma or squamous cell carcinoma were excluded.
The findings were limited by the possible unblinding of treatment because of localized skin reactions, the researchers noted. However, few drug-related adverse events and no drug-related serious adverse events were reported, they said.
Dr. Berman has served as a consultant, received honoraria, and served on the speakers bureau for Leo Pharma, which funded the study.
Complete clearance occurred at 12 months in 31% of patients who received field treatment with ingenol mebutate gel after cryotherapy for actinic keratoses, compared with 19% of placebo patients in a phase III trial.
The findings were published in the June issue of the Journal of Drugs in Dermatology.
Recurrence rates for AKs treated with cryotherapy alone are high, possibly because the treatment is lesion-directed and fails to address field cancerization, wrote Dr. Brian Berman of the University of Miami.
The FIELD study 1, a phase III, multicenter, randomized trial, showed that ingenol mebutate gel was safe and effective as a follow-up to cryosurgery for AKs. Short-term efficacy data (11 weeks) have been previously reported.
In this study, the patients were followed for 12 months (J. Drugs Dermatol. 2014;13:741-7).
"In phase III clinical trials, 2 or 3 days of field-directed therapy with ingenol mebutate demonstrated high and sustained clearance of AKs on the head and body, and was well tolerated," noted Dr. Berman and his coauthors.
The percentage reduction in the number of AKs was significantly higher at 12 months in the treatment group compared with the placebo group (68% vs. 54%), and significantly fewer patients in the treatment group developed new lesions in the treatment area compared with the placebo group (39% vs. 52%). In addition, the probability of sustained clearance of AKs at 12 months was 55% in the treatment group vs. 40% in the placebo group.
The study included 329 adults aged 18 years and older with 4-8 clinically typical, visible, and discrete AKs within a 25-cm2 treatment area on the face or scalp. The patients underwent cryosurgery, and 3 weeks later underwent field therapy with 0.015% ingenol mebutate gel or vehicle gel once a day for 3 consecutive days. Patients with suspected basal cell carcinoma or squamous cell carcinoma were excluded.
The findings were limited by the possible unblinding of treatment because of localized skin reactions, the researchers noted. However, few drug-related adverse events and no drug-related serious adverse events were reported, they said.
Dr. Berman has served as a consultant, received honoraria, and served on the speakers bureau for Leo Pharma, which funded the study.
FROM THE JOURNAL OF DRUGS IN DERMATOLOGY
Key clinical point: The findings support the safety and effectiveness of adding field therapy to cryosurgery when treating actinic keratosis lesions.
Major finding: Complete clearance rates at 12 months were significantly higher in patients treated with ingenol mebutate gel vs. a vehicle (31% vs. 19%).
Data source: A phase III, multicenter, randomized trial of adults with clinically typical AKs.
Disclosures: Dr. Berman has served as a consultant, received honoraria, and served on the speakers bureau for Leo Pharma, which funded the study.
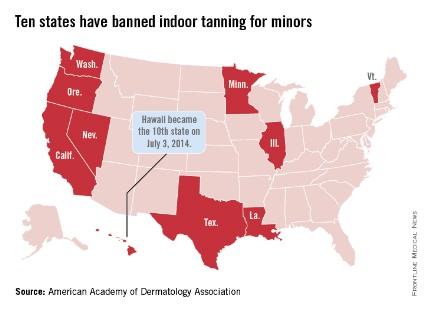
Hawaii bans indoor tanning for minors
Hawaii Gov. Neil Abercrombie signed a bill on July 3 that makes Hawaii the 10th state to prohibit anyone under age 18 years from using tanning beds, according to the American Academy of Dermatology Association.
The law goes into effect immediately. The other states that have banned indoor tanning for minors are California, Illinois, Louisiana, Minnesota, Nevada, Oregon, Texas, Vermont, and Washington.
"The science is clear. The risk for developing melanoma increases by 59% in individuals who have been exposed to UV radiation from indoor tanning devices, and the risks increase with each subsequent use," AADA President Brent M. Coldiron said in a statement.
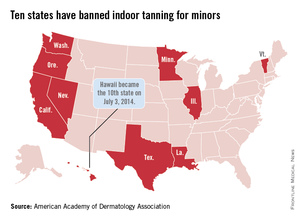
Hawaii Gov. Neil Abercrombie signed a bill on July 3 that makes Hawaii the 10th state to prohibit anyone under age 18 years from using tanning beds, according to the American Academy of Dermatology Association.
The law goes into effect immediately. The other states that have banned indoor tanning for minors are California, Illinois, Louisiana, Minnesota, Nevada, Oregon, Texas, Vermont, and Washington.
"The science is clear. The risk for developing melanoma increases by 59% in individuals who have been exposed to UV radiation from indoor tanning devices, and the risks increase with each subsequent use," AADA President Brent M. Coldiron said in a statement.

Hawaii Gov. Neil Abercrombie signed a bill on July 3 that makes Hawaii the 10th state to prohibit anyone under age 18 years from using tanning beds, according to the American Academy of Dermatology Association.
The law goes into effect immediately. The other states that have banned indoor tanning for minors are California, Illinois, Louisiana, Minnesota, Nevada, Oregon, Texas, Vermont, and Washington.
"The science is clear. The risk for developing melanoma increases by 59% in individuals who have been exposed to UV radiation from indoor tanning devices, and the risks increase with each subsequent use," AADA President Brent M. Coldiron said in a statement.

Cutaneous Melanoma
Series Editor: Arthur T. Skarin, MD, FACP, FCCP
Melanoma is the sixth most common cancer in the United States and the leading cause of deaths among all cutaneous malignancies. In 2012, it was estimated that approximately 75,000 individuals were diagnosed with melanoma and more than 9000 died. The incidence of melanoma is rising the fastest among all major malignancies, and the lifetime risk of melanoma among men and women now exceeds 1 in 68, as compared with 1:1500 in 1930.4 The incidence of melanoma is predicted to continue increasing, and there has been no corresponding decrease in mortality. This case-based review summarizes the etiology, risk factors, clinical presentation, and management of cutaneous melanomas, which comprise the majority of melanoma cases. The biology and management for other noncutaneous melanomas (such as mucosal or ocular melanomas) are beyond the scope of this review.
To read the full article in PDF:
Series Editor: Arthur T. Skarin, MD, FACP, FCCP
Melanoma is the sixth most common cancer in the United States and the leading cause of deaths among all cutaneous malignancies. In 2012, it was estimated that approximately 75,000 individuals were diagnosed with melanoma and more than 9000 died. The incidence of melanoma is rising the fastest among all major malignancies, and the lifetime risk of melanoma among men and women now exceeds 1 in 68, as compared with 1:1500 in 1930.4 The incidence of melanoma is predicted to continue increasing, and there has been no corresponding decrease in mortality. This case-based review summarizes the etiology, risk factors, clinical presentation, and management of cutaneous melanomas, which comprise the majority of melanoma cases. The biology and management for other noncutaneous melanomas (such as mucosal or ocular melanomas) are beyond the scope of this review.
To read the full article in PDF:
Series Editor: Arthur T. Skarin, MD, FACP, FCCP
Melanoma is the sixth most common cancer in the United States and the leading cause of deaths among all cutaneous malignancies. In 2012, it was estimated that approximately 75,000 individuals were diagnosed with melanoma and more than 9000 died. The incidence of melanoma is rising the fastest among all major malignancies, and the lifetime risk of melanoma among men and women now exceeds 1 in 68, as compared with 1:1500 in 1930.4 The incidence of melanoma is predicted to continue increasing, and there has been no corresponding decrease in mortality. This case-based review summarizes the etiology, risk factors, clinical presentation, and management of cutaneous melanomas, which comprise the majority of melanoma cases. The biology and management for other noncutaneous melanomas (such as mucosal or ocular melanomas) are beyond the scope of this review.
To read the full article in PDF:
House subcommittee OKs sunscreen ingredient proposal
The Health subcommittee of the House Energy & Commerce Committee unanimously advanced a proposal to make it easier for manufacturers to get new sunscreen ingredients approved by the Food and Drug Administration.
The subcommittee approved the legislation, the Sunscreen Innovation Act (H.R. 4250) on June 19. It has now been referred to the full Energy & Commerce Committee for approval.
One of the bill’s cosponsors, Rep. Ed Whitfield (R-Ky.) said that too many potentially innovative ingredients were languishing at the FDA, including at least one that had been there for a decade. "The American public is being denied effective sunscreen products that have been used safely overseas for years," said Rep. Whitfield, at a mark-up of the legislation.
He said that two outside advocacy groups – the Public Access To SunScreens (PASS) Coalition, a partnership of health organizations, sunscreen ingredient companies, and citizens, and the Environmental Working Group – had been instrumental in crafting the legislation and making sure there was an agreement that would satisfy manufacturers and also protect the public’s health.
Energy & Commerce Committee Chairman Fred Upton (R-Mich.) said in a statement submitted for the record that "it is unacceptable that the FDA has not approved a new sunscreen ingredient in nearly 2 decades." He said this was especially a problem given that there is now "a much better understanding of the deadly consequences of excessive sun exposure."
The Sunscreen Innovation Act would overhaul the reviews and approval process, said Rep. Upton.
Members of the committee are still hashing out some issues on the legislation, including timeframes for FDA review of pending and new applications. Rep. Henry Waxman (D-Calif.) said that the review process had led to "unacceptable backlogs" and unnecessary delays, but, he added, "I will not support a bill that sets FDA up for failure by imposing unrealistic timeframes for action."
The full Energy & Commerce Committee has not yet scheduled a date for taking up the bill.
On Twitter @aliciaault
The Health subcommittee of the House Energy & Commerce Committee unanimously advanced a proposal to make it easier for manufacturers to get new sunscreen ingredients approved by the Food and Drug Administration.
The subcommittee approved the legislation, the Sunscreen Innovation Act (H.R. 4250) on June 19. It has now been referred to the full Energy & Commerce Committee for approval.
One of the bill’s cosponsors, Rep. Ed Whitfield (R-Ky.) said that too many potentially innovative ingredients were languishing at the FDA, including at least one that had been there for a decade. "The American public is being denied effective sunscreen products that have been used safely overseas for years," said Rep. Whitfield, at a mark-up of the legislation.
He said that two outside advocacy groups – the Public Access To SunScreens (PASS) Coalition, a partnership of health organizations, sunscreen ingredient companies, and citizens, and the Environmental Working Group – had been instrumental in crafting the legislation and making sure there was an agreement that would satisfy manufacturers and also protect the public’s health.
Energy & Commerce Committee Chairman Fred Upton (R-Mich.) said in a statement submitted for the record that "it is unacceptable that the FDA has not approved a new sunscreen ingredient in nearly 2 decades." He said this was especially a problem given that there is now "a much better understanding of the deadly consequences of excessive sun exposure."
The Sunscreen Innovation Act would overhaul the reviews and approval process, said Rep. Upton.
Members of the committee are still hashing out some issues on the legislation, including timeframes for FDA review of pending and new applications. Rep. Henry Waxman (D-Calif.) said that the review process had led to "unacceptable backlogs" and unnecessary delays, but, he added, "I will not support a bill that sets FDA up for failure by imposing unrealistic timeframes for action."
The full Energy & Commerce Committee has not yet scheduled a date for taking up the bill.
On Twitter @aliciaault
The Health subcommittee of the House Energy & Commerce Committee unanimously advanced a proposal to make it easier for manufacturers to get new sunscreen ingredients approved by the Food and Drug Administration.
The subcommittee approved the legislation, the Sunscreen Innovation Act (H.R. 4250) on June 19. It has now been referred to the full Energy & Commerce Committee for approval.
One of the bill’s cosponsors, Rep. Ed Whitfield (R-Ky.) said that too many potentially innovative ingredients were languishing at the FDA, including at least one that had been there for a decade. "The American public is being denied effective sunscreen products that have been used safely overseas for years," said Rep. Whitfield, at a mark-up of the legislation.
He said that two outside advocacy groups – the Public Access To SunScreens (PASS) Coalition, a partnership of health organizations, sunscreen ingredient companies, and citizens, and the Environmental Working Group – had been instrumental in crafting the legislation and making sure there was an agreement that would satisfy manufacturers and also protect the public’s health.
Energy & Commerce Committee Chairman Fred Upton (R-Mich.) said in a statement submitted for the record that "it is unacceptable that the FDA has not approved a new sunscreen ingredient in nearly 2 decades." He said this was especially a problem given that there is now "a much better understanding of the deadly consequences of excessive sun exposure."
The Sunscreen Innovation Act would overhaul the reviews and approval process, said Rep. Upton.
Members of the committee are still hashing out some issues on the legislation, including timeframes for FDA review of pending and new applications. Rep. Henry Waxman (D-Calif.) said that the review process had led to "unacceptable backlogs" and unnecessary delays, but, he added, "I will not support a bill that sets FDA up for failure by imposing unrealistic timeframes for action."
The full Energy & Commerce Committee has not yet scheduled a date for taking up the bill.
On Twitter @aliciaault
Sticker Shock
A recent online study by Gerami et al in the Journal of the American Academy of Dermatology highlighted a new genomic method using messenger RNA to classify pigmented lesions as benign or malignant using a noninvasive adhesive patch developed by DermTech International. Patches were applied to the surface of pigmented lesions (42 melanomas; 22 nevi), vigorously rubbed, removed, frozen, and sent to the proprietary laboratory for RNA extraction and gene expression analysis. Then each lesion was excised for pathologic review. A 2-gene signature was discovered, including CMIP and LINC00518, differentiating melanoma from nevi with sensitivity of 97.6% and specificity of 72.7%.
What’s the issue?
Along with our evolving understanding and case-specific use of noninvasive modalities to diagnose difficult pigmented lesions, we add this test to the number of other tests and imaging approaches that seem perhaps too good to be true. A test that strips epithelial cells and involves no wound care but explores true gene differences likely sits better with us than surface microscopy, dermoscopy, and other imaging because, in this case, it provides a signature. A result. Similar to a pregnancy test, right? We wish. The diversity of pigmented lesions, especially the ones that stump us even on pathologic review, will likely prove too cryptic for 1 test to decode, but as these modalities evolve, their signatures will hopefully merge between researchers and industry to create a pigmented lesion map that we can all read. What noninvasive modalities do you use in your practices for pigmented lesions? How do you think this test will fit in?
A recent online study by Gerami et al in the Journal of the American Academy of Dermatology highlighted a new genomic method using messenger RNA to classify pigmented lesions as benign or malignant using a noninvasive adhesive patch developed by DermTech International. Patches were applied to the surface of pigmented lesions (42 melanomas; 22 nevi), vigorously rubbed, removed, frozen, and sent to the proprietary laboratory for RNA extraction and gene expression analysis. Then each lesion was excised for pathologic review. A 2-gene signature was discovered, including CMIP and LINC00518, differentiating melanoma from nevi with sensitivity of 97.6% and specificity of 72.7%.
What’s the issue?
Along with our evolving understanding and case-specific use of noninvasive modalities to diagnose difficult pigmented lesions, we add this test to the number of other tests and imaging approaches that seem perhaps too good to be true. A test that strips epithelial cells and involves no wound care but explores true gene differences likely sits better with us than surface microscopy, dermoscopy, and other imaging because, in this case, it provides a signature. A result. Similar to a pregnancy test, right? We wish. The diversity of pigmented lesions, especially the ones that stump us even on pathologic review, will likely prove too cryptic for 1 test to decode, but as these modalities evolve, their signatures will hopefully merge between researchers and industry to create a pigmented lesion map that we can all read. What noninvasive modalities do you use in your practices for pigmented lesions? How do you think this test will fit in?
A recent online study by Gerami et al in the Journal of the American Academy of Dermatology highlighted a new genomic method using messenger RNA to classify pigmented lesions as benign or malignant using a noninvasive adhesive patch developed by DermTech International. Patches were applied to the surface of pigmented lesions (42 melanomas; 22 nevi), vigorously rubbed, removed, frozen, and sent to the proprietary laboratory for RNA extraction and gene expression analysis. Then each lesion was excised for pathologic review. A 2-gene signature was discovered, including CMIP and LINC00518, differentiating melanoma from nevi with sensitivity of 97.6% and specificity of 72.7%.
What’s the issue?
Along with our evolving understanding and case-specific use of noninvasive modalities to diagnose difficult pigmented lesions, we add this test to the number of other tests and imaging approaches that seem perhaps too good to be true. A test that strips epithelial cells and involves no wound care but explores true gene differences likely sits better with us than surface microscopy, dermoscopy, and other imaging because, in this case, it provides a signature. A result. Similar to a pregnancy test, right? We wish. The diversity of pigmented lesions, especially the ones that stump us even on pathologic review, will likely prove too cryptic for 1 test to decode, but as these modalities evolve, their signatures will hopefully merge between researchers and industry to create a pigmented lesion map that we can all read. What noninvasive modalities do you use in your practices for pigmented lesions? How do you think this test will fit in?
VIDEO: Drug combo delivers ‘unprecedented’ metastatic melanoma survival
CHICAGO – Pairing the immune checkpoint inhibitors nivolumab and ipilimumab delivered deep and durable responses in patients with advanced metastatic melanoma, producing a 2-year overall survival rate of 79% among 53 patients.
"These are remarkable data even for a trial of this size in metastatic melanoma," noted Dr. Mario Sznol, a professor of medical oncology at Yale University, New Haven, Conn.
In a video interview at the annual meeting of the American Society for Clinical Oncology, Dr. Sznol discusses the study results, and he outlines how combinations of new agents with older treatments such as interferon and interleukin-2 may be used in the future.
CHICAGO – Pairing the immune checkpoint inhibitors nivolumab and ipilimumab delivered deep and durable responses in patients with advanced metastatic melanoma, producing a 2-year overall survival rate of 79% among 53 patients.
"These are remarkable data even for a trial of this size in metastatic melanoma," noted Dr. Mario Sznol, a professor of medical oncology at Yale University, New Haven, Conn.
In a video interview at the annual meeting of the American Society for Clinical Oncology, Dr. Sznol discusses the study results, and he outlines how combinations of new agents with older treatments such as interferon and interleukin-2 may be used in the future.
CHICAGO – Pairing the immune checkpoint inhibitors nivolumab and ipilimumab delivered deep and durable responses in patients with advanced metastatic melanoma, producing a 2-year overall survival rate of 79% among 53 patients.
"These are remarkable data even for a trial of this size in metastatic melanoma," noted Dr. Mario Sznol, a professor of medical oncology at Yale University, New Haven, Conn.
In a video interview at the annual meeting of the American Society for Clinical Oncology, Dr. Sznol discusses the study results, and he outlines how combinations of new agents with older treatments such as interferon and interleukin-2 may be used in the future.
AT THE ASCO ANNUAL MEETING 2014