User login
HIV drug may enhance efficacy of chemoradiation in locally advanced lung cancer
Administering an HIV drug concurrently with chemoradiotherapy resulted in promising local control and overall survival in patients with unresectable, locally advanced non–small cell lung cancer, researchers reported.
There was no overt exacerbation of the toxic effects of chemoradiotherapy with the addition of nelfinavir, a protease inhibitor, in the prospective, open-label, phase 1/2 study, the researchers wrote.
Nelfinavir plus chemoradiotherapy yielded a median progression-free survival of 11.7 months and median survival of 41.1 months, while the cumulative local failure incidence was 39% according to their report.
Those outcomes compare favorably with historical data, the investigators wrote in JAMA Oncology.
In benchmark results of the RTOG 0617 study of chemoradiotherapy in locally advanced non–small cell lung cancer, median overall survival was 28.7 months receiving radiotherapy at a standard dose of 60 Gy, and 20.3 months for those receiving high-dose (74 Gy) radiotherapy.
However, a randomized, phase 3 trial is needed to confirm these latest results with a protease inhibitor added to chemotherapy, according to Ramesh Rengan, MD, PhD, of the University of Washington, Seattle, and coinvestigators.
“As nelfinavir is a U.S. Food and Drug Administration–approved oral drug, this treatment approach is feasible and is potentially a readily exportable platform for daily clinical use,” Dr. Rengan and coauthors wrote.
In vitro and in vivo studies have shown that nelfinavir inhibited PI3K and Akt signaling, sensitized tumor cells to ionizing radiation, and improved tumor perfusion in animal models. “We hypothesize that it is these properties that drive the clinical results observed in this study,” Dr. Rengan and coauthors wrote.
They reported on a total of 35 patients with stage IIIA/IIIB non–small cell lung cancer who received nelfinavir at either 625 mg or 1,250 mg twice daily, starting 7-14 days before starting radiotherapy to 66.6 Gy at 1.8 Gy per fraction, and throughout the full course of radiotherapy.
There were no dose-limiting toxic effects observed in the study, and toxic effects were “acceptable,” with no grade 4 nonhematologic toxic effects seen, according to investigators. Leukopenia was the primary grade 3-4 hematologic toxic effect, observed in 2 of 5 patients receiving the lower nelfinavir dose and 18 of 30 at the higher dose.
Beyond non–small cell lung cancer, the efficacy and safety nelfinavir given concurrently with radiotherapy has been looked at in other disease settings. Data from those trials suggest that this protease inhibitor could “augment tumor response” not only in non–small cell lung cancer, but in locally advanced pancreatic cancer and glioblastoma, all of which are relatively radioresistant, according to Dr. Rengan and colleagues.
Study support came from grants from the National Institutes of Health and Abramson Cancer Center, and an American Society for Radiation Oncology training award to Dr. Rengan. Study authors reported disclosures related to Pfizer, 511 Pharma, Progenics Pharmaceuticals, Siemens, Actinium, AstraZeneca, Merck, Bristol-Myers Squibb, and others.
SOURCE: Rengan R et al. JAMA Oncol. 2019 Aug 22. doi: 10.1001/jamaoncol.2019.2095.
Administering an HIV drug concurrently with chemoradiotherapy resulted in promising local control and overall survival in patients with unresectable, locally advanced non–small cell lung cancer, researchers reported.
There was no overt exacerbation of the toxic effects of chemoradiotherapy with the addition of nelfinavir, a protease inhibitor, in the prospective, open-label, phase 1/2 study, the researchers wrote.
Nelfinavir plus chemoradiotherapy yielded a median progression-free survival of 11.7 months and median survival of 41.1 months, while the cumulative local failure incidence was 39% according to their report.
Those outcomes compare favorably with historical data, the investigators wrote in JAMA Oncology.
In benchmark results of the RTOG 0617 study of chemoradiotherapy in locally advanced non–small cell lung cancer, median overall survival was 28.7 months receiving radiotherapy at a standard dose of 60 Gy, and 20.3 months for those receiving high-dose (74 Gy) radiotherapy.
However, a randomized, phase 3 trial is needed to confirm these latest results with a protease inhibitor added to chemotherapy, according to Ramesh Rengan, MD, PhD, of the University of Washington, Seattle, and coinvestigators.
“As nelfinavir is a U.S. Food and Drug Administration–approved oral drug, this treatment approach is feasible and is potentially a readily exportable platform for daily clinical use,” Dr. Rengan and coauthors wrote.
In vitro and in vivo studies have shown that nelfinavir inhibited PI3K and Akt signaling, sensitized tumor cells to ionizing radiation, and improved tumor perfusion in animal models. “We hypothesize that it is these properties that drive the clinical results observed in this study,” Dr. Rengan and coauthors wrote.
They reported on a total of 35 patients with stage IIIA/IIIB non–small cell lung cancer who received nelfinavir at either 625 mg or 1,250 mg twice daily, starting 7-14 days before starting radiotherapy to 66.6 Gy at 1.8 Gy per fraction, and throughout the full course of radiotherapy.
There were no dose-limiting toxic effects observed in the study, and toxic effects were “acceptable,” with no grade 4 nonhematologic toxic effects seen, according to investigators. Leukopenia was the primary grade 3-4 hematologic toxic effect, observed in 2 of 5 patients receiving the lower nelfinavir dose and 18 of 30 at the higher dose.
Beyond non–small cell lung cancer, the efficacy and safety nelfinavir given concurrently with radiotherapy has been looked at in other disease settings. Data from those trials suggest that this protease inhibitor could “augment tumor response” not only in non–small cell lung cancer, but in locally advanced pancreatic cancer and glioblastoma, all of which are relatively radioresistant, according to Dr. Rengan and colleagues.
Study support came from grants from the National Institutes of Health and Abramson Cancer Center, and an American Society for Radiation Oncology training award to Dr. Rengan. Study authors reported disclosures related to Pfizer, 511 Pharma, Progenics Pharmaceuticals, Siemens, Actinium, AstraZeneca, Merck, Bristol-Myers Squibb, and others.
SOURCE: Rengan R et al. JAMA Oncol. 2019 Aug 22. doi: 10.1001/jamaoncol.2019.2095.
Administering an HIV drug concurrently with chemoradiotherapy resulted in promising local control and overall survival in patients with unresectable, locally advanced non–small cell lung cancer, researchers reported.
There was no overt exacerbation of the toxic effects of chemoradiotherapy with the addition of nelfinavir, a protease inhibitor, in the prospective, open-label, phase 1/2 study, the researchers wrote.
Nelfinavir plus chemoradiotherapy yielded a median progression-free survival of 11.7 months and median survival of 41.1 months, while the cumulative local failure incidence was 39% according to their report.
Those outcomes compare favorably with historical data, the investigators wrote in JAMA Oncology.
In benchmark results of the RTOG 0617 study of chemoradiotherapy in locally advanced non–small cell lung cancer, median overall survival was 28.7 months receiving radiotherapy at a standard dose of 60 Gy, and 20.3 months for those receiving high-dose (74 Gy) radiotherapy.
However, a randomized, phase 3 trial is needed to confirm these latest results with a protease inhibitor added to chemotherapy, according to Ramesh Rengan, MD, PhD, of the University of Washington, Seattle, and coinvestigators.
“As nelfinavir is a U.S. Food and Drug Administration–approved oral drug, this treatment approach is feasible and is potentially a readily exportable platform for daily clinical use,” Dr. Rengan and coauthors wrote.
In vitro and in vivo studies have shown that nelfinavir inhibited PI3K and Akt signaling, sensitized tumor cells to ionizing radiation, and improved tumor perfusion in animal models. “We hypothesize that it is these properties that drive the clinical results observed in this study,” Dr. Rengan and coauthors wrote.
They reported on a total of 35 patients with stage IIIA/IIIB non–small cell lung cancer who received nelfinavir at either 625 mg or 1,250 mg twice daily, starting 7-14 days before starting radiotherapy to 66.6 Gy at 1.8 Gy per fraction, and throughout the full course of radiotherapy.
There were no dose-limiting toxic effects observed in the study, and toxic effects were “acceptable,” with no grade 4 nonhematologic toxic effects seen, according to investigators. Leukopenia was the primary grade 3-4 hematologic toxic effect, observed in 2 of 5 patients receiving the lower nelfinavir dose and 18 of 30 at the higher dose.
Beyond non–small cell lung cancer, the efficacy and safety nelfinavir given concurrently with radiotherapy has been looked at in other disease settings. Data from those trials suggest that this protease inhibitor could “augment tumor response” not only in non–small cell lung cancer, but in locally advanced pancreatic cancer and glioblastoma, all of which are relatively radioresistant, according to Dr. Rengan and colleagues.
Study support came from grants from the National Institutes of Health and Abramson Cancer Center, and an American Society for Radiation Oncology training award to Dr. Rengan. Study authors reported disclosures related to Pfizer, 511 Pharma, Progenics Pharmaceuticals, Siemens, Actinium, AstraZeneca, Merck, Bristol-Myers Squibb, and others.
SOURCE: Rengan R et al. JAMA Oncol. 2019 Aug 22. doi: 10.1001/jamaoncol.2019.2095.
FROM JAMA ONCOLOGY
Molecular profiling a must in advanced NSCLC
All patients with locally advanced or metastatic non–small cell lung cancer (NSCLC) should undergo molecular testing for targetable mutations and for tumor expression of the programmed death–ligand 1 (PD-L1) protein, authors of a review of systemic therapies for NSCLC recommend.
Their opinion is based on evidence showing that 5-year overall survival rate for patients whose tumors have high levels of PD-L1 expression now exceeds 25%, and that patients with ALK-positive tumors have 5-year overall survival rates over 40%. In contrast, 5-year survival rates for patients with metastatic NSCLC prior to the 21st century were less than 5%, according to Kathryn C. Arbour, MD, and Gregory J. Riely, MD, PhD, from Memorial Sloan Kettering Cancer Center in New York.
“Improved understanding of the biology and molecular subtypes of non–small cell lung cancer have led to more biomarker-directed therapies for patients with metastatic disease. These biomarker-directed therapies and newer empirical treatment regimens have improved overall survival for patients with metastatic non–small cell lung cancer,” they wrote in JAMA.
The authors reviewed published studies of clinical trials of medical therapies for NSCLC, including articles on randomized trials, nonrandomized trials leading to practice changes or regulatory approval of new therapies for patients with locally advanced or metastatic NSCLC, and clinical practice guidelines.
Their review showed that approximately 30% of patients with NSCLC have molecular alterations predictive of response to treatment, such as mutations in EGFR, the gene encoding for epidermal growth factor receptor; rearrangements in the ALK (anaplastic lymphoma kinase) and ROS1 genes; and mutations in BRAF V600E.
Patients with somatic activating mutations in EGFR, which occur in approximately 20% of those with advanced NSCLC, have better progression-free survival when treated with an EGFR-target tyrosine kinase inhibitor such as gefitinib (Iressa), erlotinib (Tarceva), or afatinib (Gilotrif), compared with cytotoxic chemotherapy.
Similarly, they noted, patients with ALK rearrangements leading to overexpression of the ALK protein had better overall response rates and progression-free survival when treated with the ALK inhibitor crizotinib (Xalkori), compared with patients with ALK rearrangements treated with pemetrexed and a platinum agent.
For some patients without targetable mutations, immune checkpoint inhibitors either alone or in combination with chemotherapy have resulted in improvements in overall survival.
“These advances are substantial, but long-term durable responses remain uncommon for most patients. These insights into treating metastatic disease have informed the design of trials for new treatment strategies among patients with early-stage disease. The goal of NSCLC research is to understand and address mechanisms of resistant and refractory disease in patients with advanced disease and, ultimately, to increase cure rates,” the reviewers wrote.
The review was supported in part by a grant from the National Cancer Institute to Memorial Sloan Kettering. Dr. Arbour reported serving as a consultant to AstraZeneca and nonfinancial research support from Novartis and Takeda. Dr. Riely reported grants and nonfinancial support from Pfizer, Roche/Genentech/Chugai, Novartis, Merck, and Takeda; a patent pending for an alternate dosing of erlotinib for which he has no right to royalties; and payments from the National Comprehensive Cancer Network to participate in a committee overseeing solicitation and selection of grants to be awarded by AstraZeneca.
SOURCE: Arbour KC and Riely GJ. JAMA. 2019;322(8):764-74.
All patients with locally advanced or metastatic non–small cell lung cancer (NSCLC) should undergo molecular testing for targetable mutations and for tumor expression of the programmed death–ligand 1 (PD-L1) protein, authors of a review of systemic therapies for NSCLC recommend.
Their opinion is based on evidence showing that 5-year overall survival rate for patients whose tumors have high levels of PD-L1 expression now exceeds 25%, and that patients with ALK-positive tumors have 5-year overall survival rates over 40%. In contrast, 5-year survival rates for patients with metastatic NSCLC prior to the 21st century were less than 5%, according to Kathryn C. Arbour, MD, and Gregory J. Riely, MD, PhD, from Memorial Sloan Kettering Cancer Center in New York.
“Improved understanding of the biology and molecular subtypes of non–small cell lung cancer have led to more biomarker-directed therapies for patients with metastatic disease. These biomarker-directed therapies and newer empirical treatment regimens have improved overall survival for patients with metastatic non–small cell lung cancer,” they wrote in JAMA.
The authors reviewed published studies of clinical trials of medical therapies for NSCLC, including articles on randomized trials, nonrandomized trials leading to practice changes or regulatory approval of new therapies for patients with locally advanced or metastatic NSCLC, and clinical practice guidelines.
Their review showed that approximately 30% of patients with NSCLC have molecular alterations predictive of response to treatment, such as mutations in EGFR, the gene encoding for epidermal growth factor receptor; rearrangements in the ALK (anaplastic lymphoma kinase) and ROS1 genes; and mutations in BRAF V600E.
Patients with somatic activating mutations in EGFR, which occur in approximately 20% of those with advanced NSCLC, have better progression-free survival when treated with an EGFR-target tyrosine kinase inhibitor such as gefitinib (Iressa), erlotinib (Tarceva), or afatinib (Gilotrif), compared with cytotoxic chemotherapy.
Similarly, they noted, patients with ALK rearrangements leading to overexpression of the ALK protein had better overall response rates and progression-free survival when treated with the ALK inhibitor crizotinib (Xalkori), compared with patients with ALK rearrangements treated with pemetrexed and a platinum agent.
For some patients without targetable mutations, immune checkpoint inhibitors either alone or in combination with chemotherapy have resulted in improvements in overall survival.
“These advances are substantial, but long-term durable responses remain uncommon for most patients. These insights into treating metastatic disease have informed the design of trials for new treatment strategies among patients with early-stage disease. The goal of NSCLC research is to understand and address mechanisms of resistant and refractory disease in patients with advanced disease and, ultimately, to increase cure rates,” the reviewers wrote.
The review was supported in part by a grant from the National Cancer Institute to Memorial Sloan Kettering. Dr. Arbour reported serving as a consultant to AstraZeneca and nonfinancial research support from Novartis and Takeda. Dr. Riely reported grants and nonfinancial support from Pfizer, Roche/Genentech/Chugai, Novartis, Merck, and Takeda; a patent pending for an alternate dosing of erlotinib for which he has no right to royalties; and payments from the National Comprehensive Cancer Network to participate in a committee overseeing solicitation and selection of grants to be awarded by AstraZeneca.
SOURCE: Arbour KC and Riely GJ. JAMA. 2019;322(8):764-74.
All patients with locally advanced or metastatic non–small cell lung cancer (NSCLC) should undergo molecular testing for targetable mutations and for tumor expression of the programmed death–ligand 1 (PD-L1) protein, authors of a review of systemic therapies for NSCLC recommend.
Their opinion is based on evidence showing that 5-year overall survival rate for patients whose tumors have high levels of PD-L1 expression now exceeds 25%, and that patients with ALK-positive tumors have 5-year overall survival rates over 40%. In contrast, 5-year survival rates for patients with metastatic NSCLC prior to the 21st century were less than 5%, according to Kathryn C. Arbour, MD, and Gregory J. Riely, MD, PhD, from Memorial Sloan Kettering Cancer Center in New York.
“Improved understanding of the biology and molecular subtypes of non–small cell lung cancer have led to more biomarker-directed therapies for patients with metastatic disease. These biomarker-directed therapies and newer empirical treatment regimens have improved overall survival for patients with metastatic non–small cell lung cancer,” they wrote in JAMA.
The authors reviewed published studies of clinical trials of medical therapies for NSCLC, including articles on randomized trials, nonrandomized trials leading to practice changes or regulatory approval of new therapies for patients with locally advanced or metastatic NSCLC, and clinical practice guidelines.
Their review showed that approximately 30% of patients with NSCLC have molecular alterations predictive of response to treatment, such as mutations in EGFR, the gene encoding for epidermal growth factor receptor; rearrangements in the ALK (anaplastic lymphoma kinase) and ROS1 genes; and mutations in BRAF V600E.
Patients with somatic activating mutations in EGFR, which occur in approximately 20% of those with advanced NSCLC, have better progression-free survival when treated with an EGFR-target tyrosine kinase inhibitor such as gefitinib (Iressa), erlotinib (Tarceva), or afatinib (Gilotrif), compared with cytotoxic chemotherapy.
Similarly, they noted, patients with ALK rearrangements leading to overexpression of the ALK protein had better overall response rates and progression-free survival when treated with the ALK inhibitor crizotinib (Xalkori), compared with patients with ALK rearrangements treated with pemetrexed and a platinum agent.
For some patients without targetable mutations, immune checkpoint inhibitors either alone or in combination with chemotherapy have resulted in improvements in overall survival.
“These advances are substantial, but long-term durable responses remain uncommon for most patients. These insights into treating metastatic disease have informed the design of trials for new treatment strategies among patients with early-stage disease. The goal of NSCLC research is to understand and address mechanisms of resistant and refractory disease in patients with advanced disease and, ultimately, to increase cure rates,” the reviewers wrote.
The review was supported in part by a grant from the National Cancer Institute to Memorial Sloan Kettering. Dr. Arbour reported serving as a consultant to AstraZeneca and nonfinancial research support from Novartis and Takeda. Dr. Riely reported grants and nonfinancial support from Pfizer, Roche/Genentech/Chugai, Novartis, Merck, and Takeda; a patent pending for an alternate dosing of erlotinib for which he has no right to royalties; and payments from the National Comprehensive Cancer Network to participate in a committee overseeing solicitation and selection of grants to be awarded by AstraZeneca.
SOURCE: Arbour KC and Riely GJ. JAMA. 2019;322(8):764-74.
FROM JAMA
Nivolumab yields long-term survival benefit in advanced NSCLC
The use of nivolumab is associated with a long-term survival benefit, compared with docetaxel in patients with previously treated advanced non–small cell lung cancer (NSCLC), according to a pooled analysis of four trials.
The survival outcomes comparing the two therapies demonstrates an extended survival advantage for nivolumab up to and past a duration of 4 years.
“We aimed to evaluate the long-term benefit of nivolumab and the effect of response and disease control on subsequent survival,” wrote Scott J. Antonia, MD, PhD, formally of the H. Lee Moffitt Cancer Center in Tampa and now at Duke Cancer Center, Durham, N.C., and colleagues. The findings were published in the Lancet Oncology.
The researchers combined data from four clinical studies (CheckMate 003, 017, 057, and 063) that assessed survival outcomes in patients receiving second-line or later nivolumab therapy. Across the four trials, a total of 664 patients were administered nivolumab.
The CheckMate 057 and 017 phase 3, randomized clinical trials compared docetaxel versus nivolumab in patients with previously treated nonsquamous and squamous NSCLC, respectively.
With respect to safety analyses, Dr. Antonia and colleagues included patients who were administered a minimum of one dose of nivolumab.
Across the four trials, the 4-year overall survival with nivolumab was 14% (11%-17%), including 11% (7%-16%) for patients with under 1% programmed death–ligand 1 expression, and 19% (15%-24%) for patients with a minimum of 1% programmed death–ligand 1 expression.
In CheckMate 057 and 017, the 4-year overall survival with nivolumab was 14% (11%-18%) versus 5% (3%-7%) in patients who received docetaxel.
With respect to safety, analysis of the long-term data did not reveal any novel safety signals.
The researchers acknowledged that a key limitation of the study was the exclusion of patients who were maintained in stable disease or in response at the point of data lock.
As a result, the findings likely minimize the survival advantage seen post–disease progression for nivolumab, compared with docetaxel.
“Additional analyses assessing the effect of various factors on long-term survival with immunotherapy versus chemotherapy are planned,” they wrote.
The study was funded by Bristol-Myers Squibb. The authors reported financial affiliations with AstraZeneca, Boehringer Ingelheim, Cellular Biomedicine Group, FLX Bio, Genentech, Novartis, Regeneron, and several others.
SOURCE: Antonia SJ et al. Lancet Oncol. 2019 Aug 14. doi: 10.1016/S1470-2045(19)30407-3.
Recent advances in the treatment of metastatic non–small cell lung cancer are largely attributable to the arrival of checkpoint inhibitor therapy. As the body of evidence continues to grow, the long-term survival advantages seen with these agents are becoming increasingly apparent.
The findings of a combined analysis of four trials evaluating second-line nivolumab in patients with non–small cell lung cancer were recently reported. In comparison with historical data, which estimated 5-year overall survival rates to be less than 5%, the 4-year overall survival of 14% found by Dr. Antonia and colleagues is significant. The results suggest a plateau in overall survival of approximately 15% for patients in this setting.
With respect to safety, recent findings confirm that patients maintained on checkpoint inhibitor therapy should be closely monitored for the complete duration of exposure. In contrast to chemotherapy, there has been no evidence suggesting cumulative toxicity is related to immunotherapy treatment.
One question that remains from the current study is the ideal duration of checkpoint inhibitor therapy for patients who achieve an objective response. In addition, whether therapy should be maintained until adverse events or disease progression are seen also remains unclear. Various trials are presently ongoing in an attempt to help answer these remaining questions.
Pierre-Jean Souquet, MD, is affiliated with the University Hospital of Lyon (France). Sébastien Couraud, MD, PhD, is affiliated with the Université de Lyon (France). The authors reported financial affiliations with AstraZeneca, Bristol-Myers Squibb, Dohme, Merck, and Roche. These comments are adapted from their editorial (Lancet Oncol. 2019 Aug 14. doi: 10.1016/S1470-2045(19)30508-X ).
Recent advances in the treatment of metastatic non–small cell lung cancer are largely attributable to the arrival of checkpoint inhibitor therapy. As the body of evidence continues to grow, the long-term survival advantages seen with these agents are becoming increasingly apparent.
The findings of a combined analysis of four trials evaluating second-line nivolumab in patients with non–small cell lung cancer were recently reported. In comparison with historical data, which estimated 5-year overall survival rates to be less than 5%, the 4-year overall survival of 14% found by Dr. Antonia and colleagues is significant. The results suggest a plateau in overall survival of approximately 15% for patients in this setting.
With respect to safety, recent findings confirm that patients maintained on checkpoint inhibitor therapy should be closely monitored for the complete duration of exposure. In contrast to chemotherapy, there has been no evidence suggesting cumulative toxicity is related to immunotherapy treatment.
One question that remains from the current study is the ideal duration of checkpoint inhibitor therapy for patients who achieve an objective response. In addition, whether therapy should be maintained until adverse events or disease progression are seen also remains unclear. Various trials are presently ongoing in an attempt to help answer these remaining questions.
Pierre-Jean Souquet, MD, is affiliated with the University Hospital of Lyon (France). Sébastien Couraud, MD, PhD, is affiliated with the Université de Lyon (France). The authors reported financial affiliations with AstraZeneca, Bristol-Myers Squibb, Dohme, Merck, and Roche. These comments are adapted from their editorial (Lancet Oncol. 2019 Aug 14. doi: 10.1016/S1470-2045(19)30508-X ).
Recent advances in the treatment of metastatic non–small cell lung cancer are largely attributable to the arrival of checkpoint inhibitor therapy. As the body of evidence continues to grow, the long-term survival advantages seen with these agents are becoming increasingly apparent.
The findings of a combined analysis of four trials evaluating second-line nivolumab in patients with non–small cell lung cancer were recently reported. In comparison with historical data, which estimated 5-year overall survival rates to be less than 5%, the 4-year overall survival of 14% found by Dr. Antonia and colleagues is significant. The results suggest a plateau in overall survival of approximately 15% for patients in this setting.
With respect to safety, recent findings confirm that patients maintained on checkpoint inhibitor therapy should be closely monitored for the complete duration of exposure. In contrast to chemotherapy, there has been no evidence suggesting cumulative toxicity is related to immunotherapy treatment.
One question that remains from the current study is the ideal duration of checkpoint inhibitor therapy for patients who achieve an objective response. In addition, whether therapy should be maintained until adverse events or disease progression are seen also remains unclear. Various trials are presently ongoing in an attempt to help answer these remaining questions.
Pierre-Jean Souquet, MD, is affiliated with the University Hospital of Lyon (France). Sébastien Couraud, MD, PhD, is affiliated with the Université de Lyon (France). The authors reported financial affiliations with AstraZeneca, Bristol-Myers Squibb, Dohme, Merck, and Roche. These comments are adapted from their editorial (Lancet Oncol. 2019 Aug 14. doi: 10.1016/S1470-2045(19)30508-X ).
The use of nivolumab is associated with a long-term survival benefit, compared with docetaxel in patients with previously treated advanced non–small cell lung cancer (NSCLC), according to a pooled analysis of four trials.
The survival outcomes comparing the two therapies demonstrates an extended survival advantage for nivolumab up to and past a duration of 4 years.
“We aimed to evaluate the long-term benefit of nivolumab and the effect of response and disease control on subsequent survival,” wrote Scott J. Antonia, MD, PhD, formally of the H. Lee Moffitt Cancer Center in Tampa and now at Duke Cancer Center, Durham, N.C., and colleagues. The findings were published in the Lancet Oncology.
The researchers combined data from four clinical studies (CheckMate 003, 017, 057, and 063) that assessed survival outcomes in patients receiving second-line or later nivolumab therapy. Across the four trials, a total of 664 patients were administered nivolumab.
The CheckMate 057 and 017 phase 3, randomized clinical trials compared docetaxel versus nivolumab in patients with previously treated nonsquamous and squamous NSCLC, respectively.
With respect to safety analyses, Dr. Antonia and colleagues included patients who were administered a minimum of one dose of nivolumab.
Across the four trials, the 4-year overall survival with nivolumab was 14% (11%-17%), including 11% (7%-16%) for patients with under 1% programmed death–ligand 1 expression, and 19% (15%-24%) for patients with a minimum of 1% programmed death–ligand 1 expression.
In CheckMate 057 and 017, the 4-year overall survival with nivolumab was 14% (11%-18%) versus 5% (3%-7%) in patients who received docetaxel.
With respect to safety, analysis of the long-term data did not reveal any novel safety signals.
The researchers acknowledged that a key limitation of the study was the exclusion of patients who were maintained in stable disease or in response at the point of data lock.
As a result, the findings likely minimize the survival advantage seen post–disease progression for nivolumab, compared with docetaxel.
“Additional analyses assessing the effect of various factors on long-term survival with immunotherapy versus chemotherapy are planned,” they wrote.
The study was funded by Bristol-Myers Squibb. The authors reported financial affiliations with AstraZeneca, Boehringer Ingelheim, Cellular Biomedicine Group, FLX Bio, Genentech, Novartis, Regeneron, and several others.
SOURCE: Antonia SJ et al. Lancet Oncol. 2019 Aug 14. doi: 10.1016/S1470-2045(19)30407-3.
The use of nivolumab is associated with a long-term survival benefit, compared with docetaxel in patients with previously treated advanced non–small cell lung cancer (NSCLC), according to a pooled analysis of four trials.
The survival outcomes comparing the two therapies demonstrates an extended survival advantage for nivolumab up to and past a duration of 4 years.
“We aimed to evaluate the long-term benefit of nivolumab and the effect of response and disease control on subsequent survival,” wrote Scott J. Antonia, MD, PhD, formally of the H. Lee Moffitt Cancer Center in Tampa and now at Duke Cancer Center, Durham, N.C., and colleagues. The findings were published in the Lancet Oncology.
The researchers combined data from four clinical studies (CheckMate 003, 017, 057, and 063) that assessed survival outcomes in patients receiving second-line or later nivolumab therapy. Across the four trials, a total of 664 patients were administered nivolumab.
The CheckMate 057 and 017 phase 3, randomized clinical trials compared docetaxel versus nivolumab in patients with previously treated nonsquamous and squamous NSCLC, respectively.
With respect to safety analyses, Dr. Antonia and colleagues included patients who were administered a minimum of one dose of nivolumab.
Across the four trials, the 4-year overall survival with nivolumab was 14% (11%-17%), including 11% (7%-16%) for patients with under 1% programmed death–ligand 1 expression, and 19% (15%-24%) for patients with a minimum of 1% programmed death–ligand 1 expression.
In CheckMate 057 and 017, the 4-year overall survival with nivolumab was 14% (11%-18%) versus 5% (3%-7%) in patients who received docetaxel.
With respect to safety, analysis of the long-term data did not reveal any novel safety signals.
The researchers acknowledged that a key limitation of the study was the exclusion of patients who were maintained in stable disease or in response at the point of data lock.
As a result, the findings likely minimize the survival advantage seen post–disease progression for nivolumab, compared with docetaxel.
“Additional analyses assessing the effect of various factors on long-term survival with immunotherapy versus chemotherapy are planned,” they wrote.
The study was funded by Bristol-Myers Squibb. The authors reported financial affiliations with AstraZeneca, Boehringer Ingelheim, Cellular Biomedicine Group, FLX Bio, Genentech, Novartis, Regeneron, and several others.
SOURCE: Antonia SJ et al. Lancet Oncol. 2019 Aug 14. doi: 10.1016/S1470-2045(19)30407-3.
FROM THE LANCET ONCOLOGY
Local treatment boosts survival for some with oligometastatic lung cancer
Adding local treatment to systemic therapy may extend survival among certain patients with oligometastatic non–small cell lung cancer (NSCLC), according to a retrospective look at more than 34,000 patients.
Surgical resection provided the greatest survival benefit, followed by external beam radiotherapy or thermal ablation (EBRT/TA), reported lead author Johannes Uhlig, MD, of University Medical Center Göttingen (Germany) and colleagues.
NSCLC patients with five or fewer metastatic sites (oligometastatic disease) are thought to achieve better outcomes than patients with more widely disseminated disease, the investigators noted in JAMA Network Open, but the benefit of local therapy for this population is unclear.
“A recent randomized, prospective study of 74 patients with oligometastatic NSCLC identified superior progression-free survival with local control after hypofractionated radiotherapy or surgical resection and radiotherapy compared with systemic therapy alone, suggesting an important application of local treatment options for patients with metastatic disease,” the investigators wrote.
To build on these findings, the investigators retrospectively evaluated 34,887 patients with stage IV NSCLC who had up to one distant metastatic lesion in the liver, lung, brain, or bone, as documented in the National Cancer Database. Treatment groups were divided into patients who received systemic therapy alone, surgical resection plus systemic therapy, or EBRT/TA plus systemic therapy. Multivariable Cox proportional hazards models were used to compare overall survival among the three groups.
Including a median follow-up of 39.4 months, data analysis showed that patients who underwent surgery and systemic therapy fared the best. Adding surgery reduced mortality risk by 38% and 41%, compared with EBRT/TA plus systemic therapy and systemic therapy alone, respectively (P less than .001 for both). Compared with systemic therapy alone, adding EBRT/TA reduced mortality risk by 5% (P = .002).
The impact of EBRT/TA varied among subgroups. For those with squamous cell carcinoma who had limited nodal disease, adding EBRT/TA resulted in a clear benefit, reducing mortality risk by 32% (P less than .001). Compared with systemic therapy alone, this benefit translated to higher survival rates for up to 3 years. Conversely, adding EBRT/TA increased risk of death by 39% among patients with extended local and distant adenocarcinoma (P less than .001). In this subgroup, survival rates over the next 3 years were higher among patients treated with systemic therapy alone.
“The present study supports a combined approach of local therapy in addition to systemic treatment for select patients with oligometastatic NSCLC,” the investigators concluded.
The study was funded by the U.S. Department of Defense. The investigators disclosed additional relationships with Bayer, AstraZeneca, Bristol-Myers Squibb, and others.
SOURCE: Uhlig et al. JAMA Netw Open. 2019 Aug 21. doi: 10.1001/jamanetworkopen.2019.9702.
Adding local treatment to systemic therapy may extend survival among certain patients with oligometastatic non–small cell lung cancer (NSCLC), according to a retrospective look at more than 34,000 patients.
Surgical resection provided the greatest survival benefit, followed by external beam radiotherapy or thermal ablation (EBRT/TA), reported lead author Johannes Uhlig, MD, of University Medical Center Göttingen (Germany) and colleagues.
NSCLC patients with five or fewer metastatic sites (oligometastatic disease) are thought to achieve better outcomes than patients with more widely disseminated disease, the investigators noted in JAMA Network Open, but the benefit of local therapy for this population is unclear.
“A recent randomized, prospective study of 74 patients with oligometastatic NSCLC identified superior progression-free survival with local control after hypofractionated radiotherapy or surgical resection and radiotherapy compared with systemic therapy alone, suggesting an important application of local treatment options for patients with metastatic disease,” the investigators wrote.
To build on these findings, the investigators retrospectively evaluated 34,887 patients with stage IV NSCLC who had up to one distant metastatic lesion in the liver, lung, brain, or bone, as documented in the National Cancer Database. Treatment groups were divided into patients who received systemic therapy alone, surgical resection plus systemic therapy, or EBRT/TA plus systemic therapy. Multivariable Cox proportional hazards models were used to compare overall survival among the three groups.
Including a median follow-up of 39.4 months, data analysis showed that patients who underwent surgery and systemic therapy fared the best. Adding surgery reduced mortality risk by 38% and 41%, compared with EBRT/TA plus systemic therapy and systemic therapy alone, respectively (P less than .001 for both). Compared with systemic therapy alone, adding EBRT/TA reduced mortality risk by 5% (P = .002).
The impact of EBRT/TA varied among subgroups. For those with squamous cell carcinoma who had limited nodal disease, adding EBRT/TA resulted in a clear benefit, reducing mortality risk by 32% (P less than .001). Compared with systemic therapy alone, this benefit translated to higher survival rates for up to 3 years. Conversely, adding EBRT/TA increased risk of death by 39% among patients with extended local and distant adenocarcinoma (P less than .001). In this subgroup, survival rates over the next 3 years were higher among patients treated with systemic therapy alone.
“The present study supports a combined approach of local therapy in addition to systemic treatment for select patients with oligometastatic NSCLC,” the investigators concluded.
The study was funded by the U.S. Department of Defense. The investigators disclosed additional relationships with Bayer, AstraZeneca, Bristol-Myers Squibb, and others.
SOURCE: Uhlig et al. JAMA Netw Open. 2019 Aug 21. doi: 10.1001/jamanetworkopen.2019.9702.
Adding local treatment to systemic therapy may extend survival among certain patients with oligometastatic non–small cell lung cancer (NSCLC), according to a retrospective look at more than 34,000 patients.
Surgical resection provided the greatest survival benefit, followed by external beam radiotherapy or thermal ablation (EBRT/TA), reported lead author Johannes Uhlig, MD, of University Medical Center Göttingen (Germany) and colleagues.
NSCLC patients with five or fewer metastatic sites (oligometastatic disease) are thought to achieve better outcomes than patients with more widely disseminated disease, the investigators noted in JAMA Network Open, but the benefit of local therapy for this population is unclear.
“A recent randomized, prospective study of 74 patients with oligometastatic NSCLC identified superior progression-free survival with local control after hypofractionated radiotherapy or surgical resection and radiotherapy compared with systemic therapy alone, suggesting an important application of local treatment options for patients with metastatic disease,” the investigators wrote.
To build on these findings, the investigators retrospectively evaluated 34,887 patients with stage IV NSCLC who had up to one distant metastatic lesion in the liver, lung, brain, or bone, as documented in the National Cancer Database. Treatment groups were divided into patients who received systemic therapy alone, surgical resection plus systemic therapy, or EBRT/TA plus systemic therapy. Multivariable Cox proportional hazards models were used to compare overall survival among the three groups.
Including a median follow-up of 39.4 months, data analysis showed that patients who underwent surgery and systemic therapy fared the best. Adding surgery reduced mortality risk by 38% and 41%, compared with EBRT/TA plus systemic therapy and systemic therapy alone, respectively (P less than .001 for both). Compared with systemic therapy alone, adding EBRT/TA reduced mortality risk by 5% (P = .002).
The impact of EBRT/TA varied among subgroups. For those with squamous cell carcinoma who had limited nodal disease, adding EBRT/TA resulted in a clear benefit, reducing mortality risk by 32% (P less than .001). Compared with systemic therapy alone, this benefit translated to higher survival rates for up to 3 years. Conversely, adding EBRT/TA increased risk of death by 39% among patients with extended local and distant adenocarcinoma (P less than .001). In this subgroup, survival rates over the next 3 years were higher among patients treated with systemic therapy alone.
“The present study supports a combined approach of local therapy in addition to systemic treatment for select patients with oligometastatic NSCLC,” the investigators concluded.
The study was funded by the U.S. Department of Defense. The investigators disclosed additional relationships with Bayer, AstraZeneca, Bristol-Myers Squibb, and others.
SOURCE: Uhlig et al. JAMA Netw Open. 2019 Aug 21. doi: 10.1001/jamanetworkopen.2019.9702.
FROM JAMA NETWORK OPEN
Key clinical point: Adding local treatment to systemic therapy may extend survival among certain patients with oligometastatic non–small cell lung cancer (NSCLC).
Major finding: Patients treated with a combination of surgical resection and systemic therapy had better overall survival than patients treated with systemic therapy alone (hazard ratio, 0.59).
Study details: A retrospective analysis of 34,887 patients with stage IV NSCLC.
Disclosures: The study was funded by the U.S. Department of Defense. The investigators disclosed additional relationships with Bayer, AstraZeneca, Bristol-Myers Squibb, and others.
Source: Uhlig J et al. JAMA Netw Open. 2019 Aug 21. doi: 10.1001/jamanetworkopen.2019.9702.
Adding chemo beats standard gefitinib for EGFR-mutated lung cancer
For patients with EGFR-mutated, advanced non–small cell lung cancer (NSCLC), adding pemetrexed and carboplatin to standard gefitinib therapy markedly extends progression free survival, but at the cost of twice as many serious toxicities, in a recent phase 3 trial.
Two previous phase 2 trials (J Clin Oncol. 2016 Sep 20;34[27]:3258-66 and Ann Oncol. 2015 Feb 10;26[5]:888-94) suggested that adding chemotherapy could improve outcomes over gefitinib alone, but this is the first study to clearly demonstrate better overall survival, reported lead author Vanita Noronha, MD, of Tata Memorial Hospital in Mumbai, India, and colleagues. They noted that this is the second regimen to demonstrate better overall survival than standard gefitinib for EGFR-mutated lung cancer, with dacomitinib being the first, as shown by the ARCHER 1050 trial.
The present study involved 350 patients with advanced, EGFR-mutated NSCLC who had an Eastern Cooperative Oncology (ECOG) performance status of 0-2 and were candidates for first-line palliative therapy. Approximately one-fifth of patients (21%) had a performance status of 2, and almost as many (18%) had brain metastases. After stratification for performance status and mutation type, patients were randomized in a 1:1 ratio to receive either gefitinib monotherapy (250 mg once daily) or gefitinib plus a chemotherapy combination of pemetrexed (500 mg/m2) and carboplatin (area under the curve of 5 with Calvert formula) on day 1 of four 21-day cycles. Subsequently, nonprogressing patients in the chemotherapy group received maintenance therapy with pemetrexed at the same dose and frequency. Treatment was continued until progression, toxicity, or withdrawal of consent. The primary endpoint was progression-free survival (PFS). Secondary outcomes included overall survival (OS), response rate, quality of life, and toxicity.
After a median follow-up of 17 months, the investigators found that adding chemotherapy to gefitinib resulted in a clear benefit, with estimated median PFS increasing from 8 months to 16 months (P less than .001). Estimated median overall survival also increased, with a figure not reached in the chemotherapy/gefitinib group, compared with 17 months among those who received gefitinib alone. Response rates echoed these findings, with more patients in the chemotherapy/gefitinib group achieving complete (2.9% vs. 0.6%) and partial remission (72.4% vs. 61.9%).
“[T]he PFS attained in our study is noteworthy, considering that 21% of our study patients had a [performance status] of 2, whereas the FLAURA study, [which demonstrated a PFS of 18.9 months with osimertinib], only included patients with a [performance status] of 1 or lower,” the investigators wrote. Their report is in Journal of Clinical Oncology.
Still, introducing chemotherapy was not without negative consequences. Compared with the gefitinib monotherapy group, patients who also received chemotherapy more often had grade 3 or higher adverse events (75% vs. 49.4%), and twice as many had clinically significant, serious toxicities (50.6% vs. 25.3%). The additional toxicities were predominantly due to myelosuppression and nephrotoxicity.
Despite these drawbacks, the investigators concluded that combination therapy was superior to gefitinib alone. “The combination of gefitinib, pemetrexed, and carboplatin represents a new standard first-line therapy for EGFR-mutant NSCLC,” the investigators concluded.
The study was funded by Tata Memorial Center Research Administration Council, Fresenius Kabi India, Lung Cancer Consortium India, and others. The investigators reported relationships with Roche, Biocon, Amgen, and others.
SOURCE: Noronha et al. Journal of Clinical Oncology. 2019 Aug 14. doi: 10.1200/JCO.19.01154.
For patients with EGFR-mutated, advanced non–small cell lung cancer (NSCLC), adding pemetrexed and carboplatin to standard gefitinib therapy markedly extends progression free survival, but at the cost of twice as many serious toxicities, in a recent phase 3 trial.
Two previous phase 2 trials (J Clin Oncol. 2016 Sep 20;34[27]:3258-66 and Ann Oncol. 2015 Feb 10;26[5]:888-94) suggested that adding chemotherapy could improve outcomes over gefitinib alone, but this is the first study to clearly demonstrate better overall survival, reported lead author Vanita Noronha, MD, of Tata Memorial Hospital in Mumbai, India, and colleagues. They noted that this is the second regimen to demonstrate better overall survival than standard gefitinib for EGFR-mutated lung cancer, with dacomitinib being the first, as shown by the ARCHER 1050 trial.
The present study involved 350 patients with advanced, EGFR-mutated NSCLC who had an Eastern Cooperative Oncology (ECOG) performance status of 0-2 and were candidates for first-line palliative therapy. Approximately one-fifth of patients (21%) had a performance status of 2, and almost as many (18%) had brain metastases. After stratification for performance status and mutation type, patients were randomized in a 1:1 ratio to receive either gefitinib monotherapy (250 mg once daily) or gefitinib plus a chemotherapy combination of pemetrexed (500 mg/m2) and carboplatin (area under the curve of 5 with Calvert formula) on day 1 of four 21-day cycles. Subsequently, nonprogressing patients in the chemotherapy group received maintenance therapy with pemetrexed at the same dose and frequency. Treatment was continued until progression, toxicity, or withdrawal of consent. The primary endpoint was progression-free survival (PFS). Secondary outcomes included overall survival (OS), response rate, quality of life, and toxicity.
After a median follow-up of 17 months, the investigators found that adding chemotherapy to gefitinib resulted in a clear benefit, with estimated median PFS increasing from 8 months to 16 months (P less than .001). Estimated median overall survival also increased, with a figure not reached in the chemotherapy/gefitinib group, compared with 17 months among those who received gefitinib alone. Response rates echoed these findings, with more patients in the chemotherapy/gefitinib group achieving complete (2.9% vs. 0.6%) and partial remission (72.4% vs. 61.9%).
“[T]he PFS attained in our study is noteworthy, considering that 21% of our study patients had a [performance status] of 2, whereas the FLAURA study, [which demonstrated a PFS of 18.9 months with osimertinib], only included patients with a [performance status] of 1 or lower,” the investigators wrote. Their report is in Journal of Clinical Oncology.
Still, introducing chemotherapy was not without negative consequences. Compared with the gefitinib monotherapy group, patients who also received chemotherapy more often had grade 3 or higher adverse events (75% vs. 49.4%), and twice as many had clinically significant, serious toxicities (50.6% vs. 25.3%). The additional toxicities were predominantly due to myelosuppression and nephrotoxicity.
Despite these drawbacks, the investigators concluded that combination therapy was superior to gefitinib alone. “The combination of gefitinib, pemetrexed, and carboplatin represents a new standard first-line therapy for EGFR-mutant NSCLC,” the investigators concluded.
The study was funded by Tata Memorial Center Research Administration Council, Fresenius Kabi India, Lung Cancer Consortium India, and others. The investigators reported relationships with Roche, Biocon, Amgen, and others.
SOURCE: Noronha et al. Journal of Clinical Oncology. 2019 Aug 14. doi: 10.1200/JCO.19.01154.
For patients with EGFR-mutated, advanced non–small cell lung cancer (NSCLC), adding pemetrexed and carboplatin to standard gefitinib therapy markedly extends progression free survival, but at the cost of twice as many serious toxicities, in a recent phase 3 trial.
Two previous phase 2 trials (J Clin Oncol. 2016 Sep 20;34[27]:3258-66 and Ann Oncol. 2015 Feb 10;26[5]:888-94) suggested that adding chemotherapy could improve outcomes over gefitinib alone, but this is the first study to clearly demonstrate better overall survival, reported lead author Vanita Noronha, MD, of Tata Memorial Hospital in Mumbai, India, and colleagues. They noted that this is the second regimen to demonstrate better overall survival than standard gefitinib for EGFR-mutated lung cancer, with dacomitinib being the first, as shown by the ARCHER 1050 trial.
The present study involved 350 patients with advanced, EGFR-mutated NSCLC who had an Eastern Cooperative Oncology (ECOG) performance status of 0-2 and were candidates for first-line palliative therapy. Approximately one-fifth of patients (21%) had a performance status of 2, and almost as many (18%) had brain metastases. After stratification for performance status and mutation type, patients were randomized in a 1:1 ratio to receive either gefitinib monotherapy (250 mg once daily) or gefitinib plus a chemotherapy combination of pemetrexed (500 mg/m2) and carboplatin (area under the curve of 5 with Calvert formula) on day 1 of four 21-day cycles. Subsequently, nonprogressing patients in the chemotherapy group received maintenance therapy with pemetrexed at the same dose and frequency. Treatment was continued until progression, toxicity, or withdrawal of consent. The primary endpoint was progression-free survival (PFS). Secondary outcomes included overall survival (OS), response rate, quality of life, and toxicity.
After a median follow-up of 17 months, the investigators found that adding chemotherapy to gefitinib resulted in a clear benefit, with estimated median PFS increasing from 8 months to 16 months (P less than .001). Estimated median overall survival also increased, with a figure not reached in the chemotherapy/gefitinib group, compared with 17 months among those who received gefitinib alone. Response rates echoed these findings, with more patients in the chemotherapy/gefitinib group achieving complete (2.9% vs. 0.6%) and partial remission (72.4% vs. 61.9%).
“[T]he PFS attained in our study is noteworthy, considering that 21% of our study patients had a [performance status] of 2, whereas the FLAURA study, [which demonstrated a PFS of 18.9 months with osimertinib], only included patients with a [performance status] of 1 or lower,” the investigators wrote. Their report is in Journal of Clinical Oncology.
Still, introducing chemotherapy was not without negative consequences. Compared with the gefitinib monotherapy group, patients who also received chemotherapy more often had grade 3 or higher adverse events (75% vs. 49.4%), and twice as many had clinically significant, serious toxicities (50.6% vs. 25.3%). The additional toxicities were predominantly due to myelosuppression and nephrotoxicity.
Despite these drawbacks, the investigators concluded that combination therapy was superior to gefitinib alone. “The combination of gefitinib, pemetrexed, and carboplatin represents a new standard first-line therapy for EGFR-mutant NSCLC,” the investigators concluded.
The study was funded by Tata Memorial Center Research Administration Council, Fresenius Kabi India, Lung Cancer Consortium India, and others. The investigators reported relationships with Roche, Biocon, Amgen, and others.
SOURCE: Noronha et al. Journal of Clinical Oncology. 2019 Aug 14. doi: 10.1200/JCO.19.01154.
FROM THE JOURNAL OF CLINICAL ONCOLOGY
Timely Diagnosis of Lung Cancer in a Dedicated VA Referral Unit with Endobronchial Ultrasound Capability (FULL)
Lung cancer is the leading cause of cancer death in the US, with 154 050 deaths in 2018.1 There have been many attempts to reduce mortality of the disease through early diagnosis with use of computed tomography (CT). The National Lung Cancer Screening trial showed that screening high-risk populations with low-dose CT (LDCT) can reduce mortality.2 However, implementing LDCT screening in the clinical setting has proven challenging, as illustrated by the VA Lung Cancer Screening Demonstration Project (LCSDP).3 A lung cancer diagnosis typically comprises several steps that require different medical specialties; this can lead to delays. In the LCSDP, the mean time to diagnosis was 137 days.3 There are no federal standards for timeliness of lung cancer diagnosis.
The nonprofit RAND Corporation is the only American research organization that has published guidelines specifying acceptable intervals for the diagnosis and treatment of lung cancer. In Quality of Care for Oncologic Conditions and HIV, RAND Corporation researchers propose management quality indicators: lung cancer diagnosis within 2 months of an abnormal radiologic study and treatment within 6 weeks of diagnosis.4 The Swedish Lung Cancer Study5 and the Canadian Strategy for Cancer Control6 both recommended a standard of about 30 days—half the time recommended by the RAND Corporation.
Bukhari and colleagues at the Dayton US Department of Veterans Affairs (VA) Medical Center (VAMC) conducted a quality improvement study that examined lung cancer diagnosis and management.7 They found the time (SD) from abnormal chest imaging to diagnosis was 35.5 (31.6) days. Of those veterans who received a lung cancer diagnosis, 89.2% had the diagnosis made within the 60 days recommended by the RAND Corporation. Although these results surpass those of the LCSDP, they can be exceeded.
Beyond the potential emotional distress of awaiting the final diagnosis of a lung lesion, a delay in diagnosis and treatment may adversely affect outcomes. LDCT screening has been shown to reduce mortality, which implies a link between survival and time to intervention. There is no published evidence that time to diagnosis in advanced stage lung cancer affects outcome. The National Cancer Database (NCDB) contains informtion on about 70% of the cancers diagnosed each year in the US.8 An analysis of 4984 patients with stage IA squamous cell lung cancer undergoing lobectomy from NCDB showed that earlier surgery was associated with an absolute decrease in 5-year mortality of 5% to 8%. 9 Hence, at least in early-stage disease, reduced time from initial suspect imaging to definitive treatment may improve survival.
A system that coordinates the requisite diagnostic steps and avoids delays should provide a significant improvement in patient care. The results of such an approach that utilized nurse navigators has been previously published. 10 Here, we present the results of a dedicated VA referral clinic with priority access to pulmonary consultation and procedures in place that are designed to expedite the diagnosis of potential lung cancer.
Methods
The John L. McClellan Memorial Veterans Hospital (JLMMVH) in Little Rock, Arkansas institutional review board approved this study, which was performed in accordance with the Declaration of Helsinki. Requirement for informed consent was waived, and patient confidentiality was maintained throughout.
We have developed a plan of care specifically to facilitate diagnosis and treatment of the large number of veterans referred to the JLMMVH Diagnostic Clinic for abnormal results of chest imaging. The clinic has priority access to same-day imaging and subspecialty consultation services. In the clinic, medical students and residents perform evaluations and a registered nurse (RN) manager coordinates care.
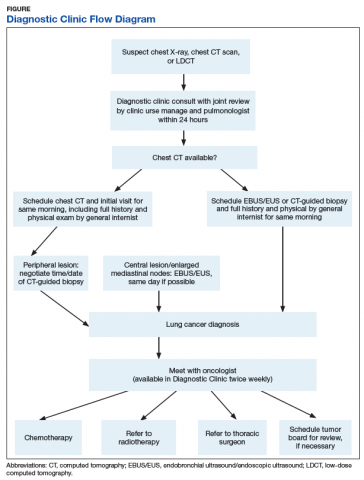
A Diagnostic Clinic consult for abnormal thoracic imaging immediately triggers an e-consult to an interventional pulmonologist (Figure). The RN manager and pulmonologist perform a joint review of records/imaging prior to scheduling, and the pulmonologist triages the patient. Triage options include follow-up imaging, bronchoscopy with endobronchial ultrasound (EBUS), endoscopic ultrasound (EUS), and CT-guided biopsy.
The RN manager then schedules a clinic visit that includes a medical evaluation by clinic staff and any indicated procedures on the same day. The interventional pulmonologist performs EBUS, EUS with the convex curvilinear bronchoscope, or both combined as indicated for diagnosis and staging. All procedures are performed in the JLMMVH bronchoscopy suite with standard conscious sedation using midazolam and fentanyl. Any other relevant procedures, such as pleural tap, also are performed at time of procedure. The pulmonologist and an attending pathologist interpret biopsies obtained in the bronchoscopy suite.
We performed a retrospective chart review of patients diagnosed with primary lung cancer through referral to the JLMMVH Diagnostic Clinic. The primary outcome was time from initial suspect chest imaging to cancer diagnosis. The study population consisted of patients referred for abnormal thoracic imaging between January 1, 2013 and December 31, 2016 and subsequently diagnosed with a primary lung cancer.
Subjects were excluded if (1) the patient was referred from outside our care network and a delay of > 10 days occurred between initial lesion imaging and referral; (2) the patient did not show up for appointments or chose to delay evaluation following referral; (3) biopsy demonstrated a nonlung primary cancer; and (4) serious intercurrent illness interrupted the diagnostic plan. In some cases, the radiologist or consulting pulmonologist had judged the lung lesion too small for immediate biopsy and recommended repeat imaging at a later date.
Patients were included in the study if the follow- up imaging led to a lung cancer diagnosis. However, because the interval between the initial imaging and the follow-up imaging in these patients did not represent a systems delay problem, the date of the scheduled follow-up abnormal imaging, which resulted in initiation of a potential cancer evaluation, served as the index suspect imaging date for this study.
Patient electronic medical records were reviewed and the following data were abstracted: date of the abnormal imaging that led to referral and time from abnormal chest X-ray to chest CT scan if applicable; date of referral and date of clinic visit; date of biopsy; date of lung cancer diagnosis; method of obtaining diagnostic specimen; lung cancer type and stage; type and date of treatment initiation or decision for supportive care only; and decision to seek further evaluation or care outside of our system.
All patients diagnosed with lung cancer during the study period were reviewed for inclusion, hence no required sample-size estimate was calculated. All outcomes were assessed as calendar days. The primary outcome was the time from the index suspect chest imaging study to the date of diagnosis of lung cancer. Prior to the initiation of our study, we chose this more stringent 30-day recommendation of the Canadian6 and Swedish5 studies as the comparator for our primary outcome, although data with respect to the 60-day Rand Corporation guidelines also are reported.4
Statistical Methods
The mean time to lung cancer diagnosis in our cohort was compared with this 30-day standard using a 2-sided Mann–Whitney U test. Normality of data distribution was determined using the Kolmogorov–Smirnov test. For statistical significance testing a P value of .05 was used. Statistical calculations were performed using R statistical software version 3.2.4. Secondary outcomes consisted of time from diagnosis to treatment; proportion of subjects diagnosed within 60 days; time from initial clinic visit to biopsy; and time from biopsy to diagnosis.
Results
Overall, 222 patients were diagnosed with a malignant lung lesion, of which 63 were excluded from analysis: 22 cancelled or did not appear for appointments, declined further evaluation, or completed evaluation outside of our network; 13 had the diagnosis made prior to Diagnostic Clinic visit; 13 proved to have a nonlung primary tumor presenting in the lung or mediastinal nodes; 12 were delayed > 10 days in referral from an outside network; and 3 had an intervening serious acute medical problem forcing delay in the diagnostic process.
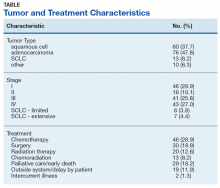
Of the 159 included subjects, 154 (96.9%) were male, and the mean (SD) age was 67.6 (8.1) years. For 76 subjects, the abnormal chest X-ray and subsequent chest CT scan were performed the same day or the lung lesion had initially been noted on a CT scan. For 54 subjects, there was a delay of ≥ 1 week in obtaining a chest CT scan. The mean (SD) time from placement of the Diagnostic Clinic consultation by the primary care provider (PCP) or other provider and the initial Diagnostic Clinic visit was 6.3 (4.4) days. The mean (SD) time from suspect imaging to diagnosis (primary outcome) was 22.6(16.6) days.
The distribution of this outcome was nonnormal (Kolmogorov-Smirnov test P < .01). When compared with the standard of 30 days, the primary outcome of 22.6 days was significantly shorter (2-sided Mann–Whitney U test P < .01). Three-quarters (76.1%) of subjects were diagnosed within 30 days and 95.0% of subjects were diagnosed within 60 days of the initial imaging. For the 8 subjects diagnosed after 60 days, contributing factors included PCP delay in Diagnostic Clinic consultation, initial negative biopsy, delay in performance of chest CT scan prior to consultation, and outsourcing of positron emission tomography (PET) scans.
Overall, 57 (35.8%) of the subjects underwent biopsy on the day of their Diagnostic Clinic visit: 14 underwent CT-guided biopsy and 43 underwent EBUS/EUS. Within 2 days of the initial visit 106 subjects (66.7%) had undergone biopsy. The mean (SD) time from initial Diagnostic Clinic visit to biopsy was 6.3 (9.5) days. The mean (SD) interval was 1.8 (3.0) days for EBUS/ EUS and 11.3 (11.7) days for CT-guided biopsy. The mean (SD) interval from biopsy to diagnosis was 3.2 (6.2) days with 64 cases (40.3%) diagnosed the day of biopsy.
Excluding subjects whose treatment was delayed by patient choice or intercurrent illness, and those who left the VA system to seek treatment elsewhere (n = 21), 24 opted for palliative care, 5 died before treatment could be initiated, and 109 underwent treatment for their tumors (Table). The mean times (SD) from diagnosis to treatment were: chemotherapy alone 34.7 (25.3) days; chemoradiation 37.0 (22.8) days; surgery 44.3 (24.4) days; radiation therapy alone 47.9 (26.0) days. With respect to the RAND Corporation recommended diagnosis to treatment time, 60.9% of chemotherapy alone, 61.5% of chemoradiation, 66.7% of surgery, and 45.0% of radiation therapy alone treatments were initiated within the 6-week window.
Discussion
This retrospective case study demonstrates the effectiveness of a dedicated diagnostic clinic with priority EBUS/EUS access in diagnosing lung cancer within the VA system. Although there is no universally accepted quality standard for comparison, the RAND Corporation recommendation of 60 days from abnormal imaging to diagnosis and the Dayton VAMC published mean of 35.5 days are guideposts; however, the results from the Dayton VAMC may have been affected negatively by some subjects undergoing serial imaging for asymptomatic nodules. We chose a more stringent standard of 30 days as recommended by Swedish and Canadian task forces.
When diagnosing lung cancer, the overriding purpose of the Diagnostic Clinic is to minimize system delays. The method is to have as simple a task as possible for the PCP or other provider who identifies a lung nodule or mass and submits a single consultation request to the Diagnostic Clinic. Once this consultation is placed, the clinic RN manager oversees all further steps required for diagnosis and referral for treatment. The key factor in achieving a mean diagnosis time of 22.6 days is the cooperation between the RN manager and the interventional pulmonologist. When a consultation is received, the RN manager and pulmonologist review the data together and schedule the initial clinic visit; the goal is same-day biopsy, which is achieved in more than one-third of cases. Not all patients with a chest image suspected for lung cancer had it ordered by their PCP. For this reason, a Diagnostic Clinic consultation is available to all health care providers in our system. Many patients reach the clinic after the discovery of a suspect chest X-ray during an emergency department visit, a regularly scheduled subspecialty appointment, or during a preoperative evaluation.
The mean time from initial visit to biopsy was 1.8 days for EBUS/EUS compared with an interval of 11.3 days for CT-guided biopsy. This difference reflects the pulmonologist’s involvement in initial scheduling of Diagnostic Clinic patients. The ability of the pulmonologist to provide an accurate assessment of sample adequacy and a preliminary diagnosis at bedside, with concurrent confirmation by a staff pathologist, permitted the Diagnostic Clinic to inform 40.3% of patients of the finding of malignancy on the day of biopsy. A published comparison of the onsite review of biopsy material showed our pulmonologist and staff pathologists to be equally accurate in their interpretations.11
Sources of Delays
While this study documents the shortest intervals from suspect imaging to diagnosis reported to date, it also identifies sources of system delay in diagnosing lung cancer that JLMMVH could further optimize. The first is the time from initial abnormal chest X-ray imaging to performance of the chest CT scan. On occasion, the index lung lesion is identified unexpectedly on an outpatient or emergency department chest CT scan. With greater use of LDCT lung cancer screening, the initial detection of suspect lesions by CT scanning will increase in the future. However, the PCP most often investigates a patient complaint with a standard chest X-ray that reveals a suspect nodule or mass. When ordered by the PCP as an outpatient test, scheduling of the follow-up chest CT scan is not given priority. More than a third of subjects experienced a delay ≥ 1 week in obtaining a chest CT scan ordered by the PCP; for 29 subjects the delay was ≥ 3weeks. At JLMMVH, the Diagnostic Clinic is given priority in scheduling CT scans. Hence, for suspect lung lesions, the chest CT scan, if not already obtained, is generally performed on the morning of the clinic visit. Educating the PCP to refer the patient immediately to the Diagnostic Clinic rather than waiting to obtain an outpatient chest CT scan may remove this source of unnecessary delay.
Scheduling a CT-guided fine needle aspiration of a lung lesion is another source of system delay. When the chest CT scan is available at the time of the Diagnostic Clinic referral, the clinic visit is scheduled for the earliest day a required CT-guided biopsy can be performed. However, the mean time of 11.3 days from initial Diagnostic Clinic visit to CT-guided biopsy is indicative of the backlog faced by the interventional radiologists.
Although infrequent, PET scans that are required before biopsy can lead to substantial delays. PET scans are performed at our university affiliate, and the joint VA-university lung tumor board sometimes generates requests for such scans prior to tissue diagnosis, yet another source of delay.
The time from referral receipt to the Diagnostic Clinic visit averaged 6.3 days. This delay usually was determined by the availability of the CT-guided biopsy or the dedicated interventional pulmonologist. Although other interventional pulmonologists at JLMMVH may perform the requisite diagnostic procedures, they are not always available for immediate review of imaging studies of referred patients nor can their schedules flexibly accommodate the number of patients seen in our clinic for evaluation.
Lung Cancer Diagnosis
Prompt diagnosis in the setting of a worrisome chest X-ray may help decrease patient anxiety, but does the clinic improve lung cancer treatment outcomes? Such improvement has been demonstrated only in stage IA squamous cell lung cancer.9 Of our study population, 37.7% had squamous cell carcinoma, and 85.5% had non-small cell lung cancer. Of those with non-small cell lung cancer, 28.9% had a clinical stage I tumor. Stage I squamous cell carcinoma, the type of tumor most likely to benefit from early diagnosis and treatment, was diagnosed in 11.3% of patients. With the increased application of LDCT screening, the proportion of veterans identified with early stage lung cancer may rise. The Providence VAMC in Rhode Island reported its results from instituting LDCT screening.12 Prior to screening, 28% of patients diagnosed with lung cancer had a stage I tumor. Following the introduction of LDCT screening, 49% diagnosed by LDCT screening had a stage I tumor. Nearly a third of their patients diagnosed with lung cancer through LDCT screening had squamous cell tumor histology. Thus, we can anticipate an increasing number of veterans with early stage lung cancer who would benefit from timely diagnosis.
The JLMMVH is a referral center for the entire state of Arkansas. Quite a few of its referred patients come from a long distance, which may require overnight housing and other related travel expenses. Apart from any potential outcome benefit, the efficiencies of the system described herein include the minimization of extra trips, an inconvenience and cost to both patient and JLMMVH.
Although the primary task of the clinic is diagnosis, we also seek to facilitate timely treatment. Our lack of an on-site PET scanner and radiation therapy, resources present on-site at the Dayton VAMC, contribute to longer therapy wait times. The shortest mean wait time at JLMMVH is for chemotherapy alone (34.7 days), in part because the JLMMVH oncologists, performing initial consultations 2 to 3 times weekly in the Diagnostic Clinic, are more readily available than are our thoracic surgeons or radiation therapists. Yet overall, JLMMVH patients often face delay from the time of lung cancer diagnosis to initiation of treatment.
The Connecticut Veterans Affairs Healthcare System has published the results of changes in lung cancer management associated with a nurse navigator system.10 Prior to creating the position of cancer care coordinator, filled by an advanced practice RNs, the mean time from clinical suspicion of lung cancer to treatment was 117 days. After 4 years of such care navigation, this waiting time had decreased to 52.4 days. Associated with this dramatic improvement in overall waiting time were decreases in the turnaround time required for performance of CT and PET scans. With respect to this big picture view of lung cancer care, our Diagnostic Clinic serves as a model for the initial step of diagnosis. Coordination and streamlining of the various steps from diagnosis to definitive therapy shall require a more system-wide effort involving all the key players in cancer care.
Conclusion
We have developed a care pathway based in a dedicated diagnostic clinic and have been able to document the shortest interval from abnormality to diagnosis of lung cancer reported in the literature to date. Efficient functioning of this clinic is dependent upon the close cooperation between a full-time RN clinic manager and an interventional pulmonologist experienced in lung cancer management and able to interpret cytologic samples at the time of biopsy. Shortening the delay between diagnosis and definitive therapy remains a challenge and may benefit from the oncology nurse navigator model previously described within the VA system. 10
1. American Cancer Society. Cancer Facts & Figures. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2018/cancer-facts-and-figures-2018.pdf. Accessed July 13, 2019.
2. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Eng J Med. 2011;365(5):395-409.
3. Kinsinger LS, Anderson C, Kim J, et al. Implementation of lung cancer screening in the Veterans Health Administration. JAMA Intern Med. 2017;177(3):399-406.
4. Asch SM, Kerr EA, Hamilton EG, Reifel JL, McGlynn EA, eds. Quality of Care for Oncologic Conditions and HIV: A Review of the Literature and Quality Indicators. Santa Monica, CA: RAND Corporation; 2000.
5. Hillerdal G. [Recommendations from the Swedish Lung Cancer Study Group: Shorter waiting times are demanded for quality in diagnostic work-ups for lung care.] Swedish Med J 1999; 96: 4691.
6. Simunovic M, Gagliardi A, McCready D, Coates A, Levine M, DePetrillo D. A snapshot of waiting times for cancer surgery provided by surgeons affiliated with regional cancer centres in Ontario. CMAJ. 2001;165(4):421-425. [Canadian Strategy for Cancer Control]
7. Bukhari A, Kumar G, Rajsheker R, Markert R. Timeliness of lung cancer diagnosis and treatment. Fed Pract. 2017;34(suppl 1):24S-29S.
8. Bilimoria KY, Ko CY, Tomlinson JS, et al. Wait times for cancer surgery in the United States: trends and predictors of delays. Ann Surg. 2011;253(4):779-785.
9. Yang CJ, Wang H, Kumar A, et al. Impact of timing of lobectomy on survival for clinical stage IA lung squamous cell carcinoma. Chest. 2017;152(6):1239-1250.
10. Hunnibell LS, Rose MG, Connery DM, et al. Using nurse navigation to improve timeliness of lung cancer care at a veterans hospital. Clin J Oncol Nurs. 2012;16(1):29-36.
11. Meena N, Jeffus S, Massoll N, et al. Rapid onsite evaluation: a comparison of cytopathologist and pulmonologist performance. Cancer Cytopatho. 2016;124(4):279-84.
12. Okereke IC, Bates MF, Jankowich MD, et al. Effects of implementation of lung cancer screening at one Veterans Affairs Medical Center. Chest 2016;150(5):1023-1029.
Lung cancer is the leading cause of cancer death in the US, with 154 050 deaths in 2018.1 There have been many attempts to reduce mortality of the disease through early diagnosis with use of computed tomography (CT). The National Lung Cancer Screening trial showed that screening high-risk populations with low-dose CT (LDCT) can reduce mortality.2 However, implementing LDCT screening in the clinical setting has proven challenging, as illustrated by the VA Lung Cancer Screening Demonstration Project (LCSDP).3 A lung cancer diagnosis typically comprises several steps that require different medical specialties; this can lead to delays. In the LCSDP, the mean time to diagnosis was 137 days.3 There are no federal standards for timeliness of lung cancer diagnosis.
The nonprofit RAND Corporation is the only American research organization that has published guidelines specifying acceptable intervals for the diagnosis and treatment of lung cancer. In Quality of Care for Oncologic Conditions and HIV, RAND Corporation researchers propose management quality indicators: lung cancer diagnosis within 2 months of an abnormal radiologic study and treatment within 6 weeks of diagnosis.4 The Swedish Lung Cancer Study5 and the Canadian Strategy for Cancer Control6 both recommended a standard of about 30 days—half the time recommended by the RAND Corporation.
Bukhari and colleagues at the Dayton US Department of Veterans Affairs (VA) Medical Center (VAMC) conducted a quality improvement study that examined lung cancer diagnosis and management.7 They found the time (SD) from abnormal chest imaging to diagnosis was 35.5 (31.6) days. Of those veterans who received a lung cancer diagnosis, 89.2% had the diagnosis made within the 60 days recommended by the RAND Corporation. Although these results surpass those of the LCSDP, they can be exceeded.
Beyond the potential emotional distress of awaiting the final diagnosis of a lung lesion, a delay in diagnosis and treatment may adversely affect outcomes. LDCT screening has been shown to reduce mortality, which implies a link between survival and time to intervention. There is no published evidence that time to diagnosis in advanced stage lung cancer affects outcome. The National Cancer Database (NCDB) contains informtion on about 70% of the cancers diagnosed each year in the US.8 An analysis of 4984 patients with stage IA squamous cell lung cancer undergoing lobectomy from NCDB showed that earlier surgery was associated with an absolute decrease in 5-year mortality of 5% to 8%. 9 Hence, at least in early-stage disease, reduced time from initial suspect imaging to definitive treatment may improve survival.
A system that coordinates the requisite diagnostic steps and avoids delays should provide a significant improvement in patient care. The results of such an approach that utilized nurse navigators has been previously published. 10 Here, we present the results of a dedicated VA referral clinic with priority access to pulmonary consultation and procedures in place that are designed to expedite the diagnosis of potential lung cancer.
Methods
The John L. McClellan Memorial Veterans Hospital (JLMMVH) in Little Rock, Arkansas institutional review board approved this study, which was performed in accordance with the Declaration of Helsinki. Requirement for informed consent was waived, and patient confidentiality was maintained throughout.
We have developed a plan of care specifically to facilitate diagnosis and treatment of the large number of veterans referred to the JLMMVH Diagnostic Clinic for abnormal results of chest imaging. The clinic has priority access to same-day imaging and subspecialty consultation services. In the clinic, medical students and residents perform evaluations and a registered nurse (RN) manager coordinates care.
A Diagnostic Clinic consult for abnormal thoracic imaging immediately triggers an e-consult to an interventional pulmonologist (Figure). The RN manager and pulmonologist perform a joint review of records/imaging prior to scheduling, and the pulmonologist triages the patient. Triage options include follow-up imaging, bronchoscopy with endobronchial ultrasound (EBUS), endoscopic ultrasound (EUS), and CT-guided biopsy.
The RN manager then schedules a clinic visit that includes a medical evaluation by clinic staff and any indicated procedures on the same day. The interventional pulmonologist performs EBUS, EUS with the convex curvilinear bronchoscope, or both combined as indicated for diagnosis and staging. All procedures are performed in the JLMMVH bronchoscopy suite with standard conscious sedation using midazolam and fentanyl. Any other relevant procedures, such as pleural tap, also are performed at time of procedure. The pulmonologist and an attending pathologist interpret biopsies obtained in the bronchoscopy suite.
We performed a retrospective chart review of patients diagnosed with primary lung cancer through referral to the JLMMVH Diagnostic Clinic. The primary outcome was time from initial suspect chest imaging to cancer diagnosis. The study population consisted of patients referred for abnormal thoracic imaging between January 1, 2013 and December 31, 2016 and subsequently diagnosed with a primary lung cancer.
Subjects were excluded if (1) the patient was referred from outside our care network and a delay of > 10 days occurred between initial lesion imaging and referral; (2) the patient did not show up for appointments or chose to delay evaluation following referral; (3) biopsy demonstrated a nonlung primary cancer; and (4) serious intercurrent illness interrupted the diagnostic plan. In some cases, the radiologist or consulting pulmonologist had judged the lung lesion too small for immediate biopsy and recommended repeat imaging at a later date.
Patients were included in the study if the follow- up imaging led to a lung cancer diagnosis. However, because the interval between the initial imaging and the follow-up imaging in these patients did not represent a systems delay problem, the date of the scheduled follow-up abnormal imaging, which resulted in initiation of a potential cancer evaluation, served as the index suspect imaging date for this study.
Patient electronic medical records were reviewed and the following data were abstracted: date of the abnormal imaging that led to referral and time from abnormal chest X-ray to chest CT scan if applicable; date of referral and date of clinic visit; date of biopsy; date of lung cancer diagnosis; method of obtaining diagnostic specimen; lung cancer type and stage; type and date of treatment initiation or decision for supportive care only; and decision to seek further evaluation or care outside of our system.
All patients diagnosed with lung cancer during the study period were reviewed for inclusion, hence no required sample-size estimate was calculated. All outcomes were assessed as calendar days. The primary outcome was the time from the index suspect chest imaging study to the date of diagnosis of lung cancer. Prior to the initiation of our study, we chose this more stringent 30-day recommendation of the Canadian6 and Swedish5 studies as the comparator for our primary outcome, although data with respect to the 60-day Rand Corporation guidelines also are reported.4
Statistical Methods
The mean time to lung cancer diagnosis in our cohort was compared with this 30-day standard using a 2-sided Mann–Whitney U test. Normality of data distribution was determined using the Kolmogorov–Smirnov test. For statistical significance testing a P value of .05 was used. Statistical calculations were performed using R statistical software version 3.2.4. Secondary outcomes consisted of time from diagnosis to treatment; proportion of subjects diagnosed within 60 days; time from initial clinic visit to biopsy; and time from biopsy to diagnosis.
Results
Overall, 222 patients were diagnosed with a malignant lung lesion, of which 63 were excluded from analysis: 22 cancelled or did not appear for appointments, declined further evaluation, or completed evaluation outside of our network; 13 had the diagnosis made prior to Diagnostic Clinic visit; 13 proved to have a nonlung primary tumor presenting in the lung or mediastinal nodes; 12 were delayed > 10 days in referral from an outside network; and 3 had an intervening serious acute medical problem forcing delay in the diagnostic process.
Of the 159 included subjects, 154 (96.9%) were male, and the mean (SD) age was 67.6 (8.1) years. For 76 subjects, the abnormal chest X-ray and subsequent chest CT scan were performed the same day or the lung lesion had initially been noted on a CT scan. For 54 subjects, there was a delay of ≥ 1 week in obtaining a chest CT scan. The mean (SD) time from placement of the Diagnostic Clinic consultation by the primary care provider (PCP) or other provider and the initial Diagnostic Clinic visit was 6.3 (4.4) days. The mean (SD) time from suspect imaging to diagnosis (primary outcome) was 22.6(16.6) days.
The distribution of this outcome was nonnormal (Kolmogorov-Smirnov test P < .01). When compared with the standard of 30 days, the primary outcome of 22.6 days was significantly shorter (2-sided Mann–Whitney U test P < .01). Three-quarters (76.1%) of subjects were diagnosed within 30 days and 95.0% of subjects were diagnosed within 60 days of the initial imaging. For the 8 subjects diagnosed after 60 days, contributing factors included PCP delay in Diagnostic Clinic consultation, initial negative biopsy, delay in performance of chest CT scan prior to consultation, and outsourcing of positron emission tomography (PET) scans.
Overall, 57 (35.8%) of the subjects underwent biopsy on the day of their Diagnostic Clinic visit: 14 underwent CT-guided biopsy and 43 underwent EBUS/EUS. Within 2 days of the initial visit 106 subjects (66.7%) had undergone biopsy. The mean (SD) time from initial Diagnostic Clinic visit to biopsy was 6.3 (9.5) days. The mean (SD) interval was 1.8 (3.0) days for EBUS/ EUS and 11.3 (11.7) days for CT-guided biopsy. The mean (SD) interval from biopsy to diagnosis was 3.2 (6.2) days with 64 cases (40.3%) diagnosed the day of biopsy.
Excluding subjects whose treatment was delayed by patient choice or intercurrent illness, and those who left the VA system to seek treatment elsewhere (n = 21), 24 opted for palliative care, 5 died before treatment could be initiated, and 109 underwent treatment for their tumors (Table). The mean times (SD) from diagnosis to treatment were: chemotherapy alone 34.7 (25.3) days; chemoradiation 37.0 (22.8) days; surgery 44.3 (24.4) days; radiation therapy alone 47.9 (26.0) days. With respect to the RAND Corporation recommended diagnosis to treatment time, 60.9% of chemotherapy alone, 61.5% of chemoradiation, 66.7% of surgery, and 45.0% of radiation therapy alone treatments were initiated within the 6-week window.
Discussion
This retrospective case study demonstrates the effectiveness of a dedicated diagnostic clinic with priority EBUS/EUS access in diagnosing lung cancer within the VA system. Although there is no universally accepted quality standard for comparison, the RAND Corporation recommendation of 60 days from abnormal imaging to diagnosis and the Dayton VAMC published mean of 35.5 days are guideposts; however, the results from the Dayton VAMC may have been affected negatively by some subjects undergoing serial imaging for asymptomatic nodules. We chose a more stringent standard of 30 days as recommended by Swedish and Canadian task forces.
When diagnosing lung cancer, the overriding purpose of the Diagnostic Clinic is to minimize system delays. The method is to have as simple a task as possible for the PCP or other provider who identifies a lung nodule or mass and submits a single consultation request to the Diagnostic Clinic. Once this consultation is placed, the clinic RN manager oversees all further steps required for diagnosis and referral for treatment. The key factor in achieving a mean diagnosis time of 22.6 days is the cooperation between the RN manager and the interventional pulmonologist. When a consultation is received, the RN manager and pulmonologist review the data together and schedule the initial clinic visit; the goal is same-day biopsy, which is achieved in more than one-third of cases. Not all patients with a chest image suspected for lung cancer had it ordered by their PCP. For this reason, a Diagnostic Clinic consultation is available to all health care providers in our system. Many patients reach the clinic after the discovery of a suspect chest X-ray during an emergency department visit, a regularly scheduled subspecialty appointment, or during a preoperative evaluation.
The mean time from initial visit to biopsy was 1.8 days for EBUS/EUS compared with an interval of 11.3 days for CT-guided biopsy. This difference reflects the pulmonologist’s involvement in initial scheduling of Diagnostic Clinic patients. The ability of the pulmonologist to provide an accurate assessment of sample adequacy and a preliminary diagnosis at bedside, with concurrent confirmation by a staff pathologist, permitted the Diagnostic Clinic to inform 40.3% of patients of the finding of malignancy on the day of biopsy. A published comparison of the onsite review of biopsy material showed our pulmonologist and staff pathologists to be equally accurate in their interpretations.11
Sources of Delays
While this study documents the shortest intervals from suspect imaging to diagnosis reported to date, it also identifies sources of system delay in diagnosing lung cancer that JLMMVH could further optimize. The first is the time from initial abnormal chest X-ray imaging to performance of the chest CT scan. On occasion, the index lung lesion is identified unexpectedly on an outpatient or emergency department chest CT scan. With greater use of LDCT lung cancer screening, the initial detection of suspect lesions by CT scanning will increase in the future. However, the PCP most often investigates a patient complaint with a standard chest X-ray that reveals a suspect nodule or mass. When ordered by the PCP as an outpatient test, scheduling of the follow-up chest CT scan is not given priority. More than a third of subjects experienced a delay ≥ 1 week in obtaining a chest CT scan ordered by the PCP; for 29 subjects the delay was ≥ 3weeks. At JLMMVH, the Diagnostic Clinic is given priority in scheduling CT scans. Hence, for suspect lung lesions, the chest CT scan, if not already obtained, is generally performed on the morning of the clinic visit. Educating the PCP to refer the patient immediately to the Diagnostic Clinic rather than waiting to obtain an outpatient chest CT scan may remove this source of unnecessary delay.
Scheduling a CT-guided fine needle aspiration of a lung lesion is another source of system delay. When the chest CT scan is available at the time of the Diagnostic Clinic referral, the clinic visit is scheduled for the earliest day a required CT-guided biopsy can be performed. However, the mean time of 11.3 days from initial Diagnostic Clinic visit to CT-guided biopsy is indicative of the backlog faced by the interventional radiologists.
Although infrequent, PET scans that are required before biopsy can lead to substantial delays. PET scans are performed at our university affiliate, and the joint VA-university lung tumor board sometimes generates requests for such scans prior to tissue diagnosis, yet another source of delay.
The time from referral receipt to the Diagnostic Clinic visit averaged 6.3 days. This delay usually was determined by the availability of the CT-guided biopsy or the dedicated interventional pulmonologist. Although other interventional pulmonologists at JLMMVH may perform the requisite diagnostic procedures, they are not always available for immediate review of imaging studies of referred patients nor can their schedules flexibly accommodate the number of patients seen in our clinic for evaluation.
Lung Cancer Diagnosis
Prompt diagnosis in the setting of a worrisome chest X-ray may help decrease patient anxiety, but does the clinic improve lung cancer treatment outcomes? Such improvement has been demonstrated only in stage IA squamous cell lung cancer.9 Of our study population, 37.7% had squamous cell carcinoma, and 85.5% had non-small cell lung cancer. Of those with non-small cell lung cancer, 28.9% had a clinical stage I tumor. Stage I squamous cell carcinoma, the type of tumor most likely to benefit from early diagnosis and treatment, was diagnosed in 11.3% of patients. With the increased application of LDCT screening, the proportion of veterans identified with early stage lung cancer may rise. The Providence VAMC in Rhode Island reported its results from instituting LDCT screening.12 Prior to screening, 28% of patients diagnosed with lung cancer had a stage I tumor. Following the introduction of LDCT screening, 49% diagnosed by LDCT screening had a stage I tumor. Nearly a third of their patients diagnosed with lung cancer through LDCT screening had squamous cell tumor histology. Thus, we can anticipate an increasing number of veterans with early stage lung cancer who would benefit from timely diagnosis.
The JLMMVH is a referral center for the entire state of Arkansas. Quite a few of its referred patients come from a long distance, which may require overnight housing and other related travel expenses. Apart from any potential outcome benefit, the efficiencies of the system described herein include the minimization of extra trips, an inconvenience and cost to both patient and JLMMVH.
Although the primary task of the clinic is diagnosis, we also seek to facilitate timely treatment. Our lack of an on-site PET scanner and radiation therapy, resources present on-site at the Dayton VAMC, contribute to longer therapy wait times. The shortest mean wait time at JLMMVH is for chemotherapy alone (34.7 days), in part because the JLMMVH oncologists, performing initial consultations 2 to 3 times weekly in the Diagnostic Clinic, are more readily available than are our thoracic surgeons or radiation therapists. Yet overall, JLMMVH patients often face delay from the time of lung cancer diagnosis to initiation of treatment.
The Connecticut Veterans Affairs Healthcare System has published the results of changes in lung cancer management associated with a nurse navigator system.10 Prior to creating the position of cancer care coordinator, filled by an advanced practice RNs, the mean time from clinical suspicion of lung cancer to treatment was 117 days. After 4 years of such care navigation, this waiting time had decreased to 52.4 days. Associated with this dramatic improvement in overall waiting time were decreases in the turnaround time required for performance of CT and PET scans. With respect to this big picture view of lung cancer care, our Diagnostic Clinic serves as a model for the initial step of diagnosis. Coordination and streamlining of the various steps from diagnosis to definitive therapy shall require a more system-wide effort involving all the key players in cancer care.
Conclusion
We have developed a care pathway based in a dedicated diagnostic clinic and have been able to document the shortest interval from abnormality to diagnosis of lung cancer reported in the literature to date. Efficient functioning of this clinic is dependent upon the close cooperation between a full-time RN clinic manager and an interventional pulmonologist experienced in lung cancer management and able to interpret cytologic samples at the time of biopsy. Shortening the delay between diagnosis and definitive therapy remains a challenge and may benefit from the oncology nurse navigator model previously described within the VA system. 10
Lung cancer is the leading cause of cancer death in the US, with 154 050 deaths in 2018.1 There have been many attempts to reduce mortality of the disease through early diagnosis with use of computed tomography (CT). The National Lung Cancer Screening trial showed that screening high-risk populations with low-dose CT (LDCT) can reduce mortality.2 However, implementing LDCT screening in the clinical setting has proven challenging, as illustrated by the VA Lung Cancer Screening Demonstration Project (LCSDP).3 A lung cancer diagnosis typically comprises several steps that require different medical specialties; this can lead to delays. In the LCSDP, the mean time to diagnosis was 137 days.3 There are no federal standards for timeliness of lung cancer diagnosis.
The nonprofit RAND Corporation is the only American research organization that has published guidelines specifying acceptable intervals for the diagnosis and treatment of lung cancer. In Quality of Care for Oncologic Conditions and HIV, RAND Corporation researchers propose management quality indicators: lung cancer diagnosis within 2 months of an abnormal radiologic study and treatment within 6 weeks of diagnosis.4 The Swedish Lung Cancer Study5 and the Canadian Strategy for Cancer Control6 both recommended a standard of about 30 days—half the time recommended by the RAND Corporation.
Bukhari and colleagues at the Dayton US Department of Veterans Affairs (VA) Medical Center (VAMC) conducted a quality improvement study that examined lung cancer diagnosis and management.7 They found the time (SD) from abnormal chest imaging to diagnosis was 35.5 (31.6) days. Of those veterans who received a lung cancer diagnosis, 89.2% had the diagnosis made within the 60 days recommended by the RAND Corporation. Although these results surpass those of the LCSDP, they can be exceeded.
Beyond the potential emotional distress of awaiting the final diagnosis of a lung lesion, a delay in diagnosis and treatment may adversely affect outcomes. LDCT screening has been shown to reduce mortality, which implies a link between survival and time to intervention. There is no published evidence that time to diagnosis in advanced stage lung cancer affects outcome. The National Cancer Database (NCDB) contains informtion on about 70% of the cancers diagnosed each year in the US.8 An analysis of 4984 patients with stage IA squamous cell lung cancer undergoing lobectomy from NCDB showed that earlier surgery was associated with an absolute decrease in 5-year mortality of 5% to 8%. 9 Hence, at least in early-stage disease, reduced time from initial suspect imaging to definitive treatment may improve survival.
A system that coordinates the requisite diagnostic steps and avoids delays should provide a significant improvement in patient care. The results of such an approach that utilized nurse navigators has been previously published. 10 Here, we present the results of a dedicated VA referral clinic with priority access to pulmonary consultation and procedures in place that are designed to expedite the diagnosis of potential lung cancer.
Methods
The John L. McClellan Memorial Veterans Hospital (JLMMVH) in Little Rock, Arkansas institutional review board approved this study, which was performed in accordance with the Declaration of Helsinki. Requirement for informed consent was waived, and patient confidentiality was maintained throughout.
We have developed a plan of care specifically to facilitate diagnosis and treatment of the large number of veterans referred to the JLMMVH Diagnostic Clinic for abnormal results of chest imaging. The clinic has priority access to same-day imaging and subspecialty consultation services. In the clinic, medical students and residents perform evaluations and a registered nurse (RN) manager coordinates care.
A Diagnostic Clinic consult for abnormal thoracic imaging immediately triggers an e-consult to an interventional pulmonologist (Figure). The RN manager and pulmonologist perform a joint review of records/imaging prior to scheduling, and the pulmonologist triages the patient. Triage options include follow-up imaging, bronchoscopy with endobronchial ultrasound (EBUS), endoscopic ultrasound (EUS), and CT-guided biopsy.
The RN manager then schedules a clinic visit that includes a medical evaluation by clinic staff and any indicated procedures on the same day. The interventional pulmonologist performs EBUS, EUS with the convex curvilinear bronchoscope, or both combined as indicated for diagnosis and staging. All procedures are performed in the JLMMVH bronchoscopy suite with standard conscious sedation using midazolam and fentanyl. Any other relevant procedures, such as pleural tap, also are performed at time of procedure. The pulmonologist and an attending pathologist interpret biopsies obtained in the bronchoscopy suite.
We performed a retrospective chart review of patients diagnosed with primary lung cancer through referral to the JLMMVH Diagnostic Clinic. The primary outcome was time from initial suspect chest imaging to cancer diagnosis. The study population consisted of patients referred for abnormal thoracic imaging between January 1, 2013 and December 31, 2016 and subsequently diagnosed with a primary lung cancer.
Subjects were excluded if (1) the patient was referred from outside our care network and a delay of > 10 days occurred between initial lesion imaging and referral; (2) the patient did not show up for appointments or chose to delay evaluation following referral; (3) biopsy demonstrated a nonlung primary cancer; and (4) serious intercurrent illness interrupted the diagnostic plan. In some cases, the radiologist or consulting pulmonologist had judged the lung lesion too small for immediate biopsy and recommended repeat imaging at a later date.
Patients were included in the study if the follow- up imaging led to a lung cancer diagnosis. However, because the interval between the initial imaging and the follow-up imaging in these patients did not represent a systems delay problem, the date of the scheduled follow-up abnormal imaging, which resulted in initiation of a potential cancer evaluation, served as the index suspect imaging date for this study.
Patient electronic medical records were reviewed and the following data were abstracted: date of the abnormal imaging that led to referral and time from abnormal chest X-ray to chest CT scan if applicable; date of referral and date of clinic visit; date of biopsy; date of lung cancer diagnosis; method of obtaining diagnostic specimen; lung cancer type and stage; type and date of treatment initiation or decision for supportive care only; and decision to seek further evaluation or care outside of our system.
All patients diagnosed with lung cancer during the study period were reviewed for inclusion, hence no required sample-size estimate was calculated. All outcomes were assessed as calendar days. The primary outcome was the time from the index suspect chest imaging study to the date of diagnosis of lung cancer. Prior to the initiation of our study, we chose this more stringent 30-day recommendation of the Canadian6 and Swedish5 studies as the comparator for our primary outcome, although data with respect to the 60-day Rand Corporation guidelines also are reported.4
Statistical Methods
The mean time to lung cancer diagnosis in our cohort was compared with this 30-day standard using a 2-sided Mann–Whitney U test. Normality of data distribution was determined using the Kolmogorov–Smirnov test. For statistical significance testing a P value of .05 was used. Statistical calculations were performed using R statistical software version 3.2.4. Secondary outcomes consisted of time from diagnosis to treatment; proportion of subjects diagnosed within 60 days; time from initial clinic visit to biopsy; and time from biopsy to diagnosis.
Results
Overall, 222 patients were diagnosed with a malignant lung lesion, of which 63 were excluded from analysis: 22 cancelled or did not appear for appointments, declined further evaluation, or completed evaluation outside of our network; 13 had the diagnosis made prior to Diagnostic Clinic visit; 13 proved to have a nonlung primary tumor presenting in the lung or mediastinal nodes; 12 were delayed > 10 days in referral from an outside network; and 3 had an intervening serious acute medical problem forcing delay in the diagnostic process.
Of the 159 included subjects, 154 (96.9%) were male, and the mean (SD) age was 67.6 (8.1) years. For 76 subjects, the abnormal chest X-ray and subsequent chest CT scan were performed the same day or the lung lesion had initially been noted on a CT scan. For 54 subjects, there was a delay of ≥ 1 week in obtaining a chest CT scan. The mean (SD) time from placement of the Diagnostic Clinic consultation by the primary care provider (PCP) or other provider and the initial Diagnostic Clinic visit was 6.3 (4.4) days. The mean (SD) time from suspect imaging to diagnosis (primary outcome) was 22.6(16.6) days.
The distribution of this outcome was nonnormal (Kolmogorov-Smirnov test P < .01). When compared with the standard of 30 days, the primary outcome of 22.6 days was significantly shorter (2-sided Mann–Whitney U test P < .01). Three-quarters (76.1%) of subjects were diagnosed within 30 days and 95.0% of subjects were diagnosed within 60 days of the initial imaging. For the 8 subjects diagnosed after 60 days, contributing factors included PCP delay in Diagnostic Clinic consultation, initial negative biopsy, delay in performance of chest CT scan prior to consultation, and outsourcing of positron emission tomography (PET) scans.
Overall, 57 (35.8%) of the subjects underwent biopsy on the day of their Diagnostic Clinic visit: 14 underwent CT-guided biopsy and 43 underwent EBUS/EUS. Within 2 days of the initial visit 106 subjects (66.7%) had undergone biopsy. The mean (SD) time from initial Diagnostic Clinic visit to biopsy was 6.3 (9.5) days. The mean (SD) interval was 1.8 (3.0) days for EBUS/ EUS and 11.3 (11.7) days for CT-guided biopsy. The mean (SD) interval from biopsy to diagnosis was 3.2 (6.2) days with 64 cases (40.3%) diagnosed the day of biopsy.
Excluding subjects whose treatment was delayed by patient choice or intercurrent illness, and those who left the VA system to seek treatment elsewhere (n = 21), 24 opted for palliative care, 5 died before treatment could be initiated, and 109 underwent treatment for their tumors (Table). The mean times (SD) from diagnosis to treatment were: chemotherapy alone 34.7 (25.3) days; chemoradiation 37.0 (22.8) days; surgery 44.3 (24.4) days; radiation therapy alone 47.9 (26.0) days. With respect to the RAND Corporation recommended diagnosis to treatment time, 60.9% of chemotherapy alone, 61.5% of chemoradiation, 66.7% of surgery, and 45.0% of radiation therapy alone treatments were initiated within the 6-week window.
Discussion
This retrospective case study demonstrates the effectiveness of a dedicated diagnostic clinic with priority EBUS/EUS access in diagnosing lung cancer within the VA system. Although there is no universally accepted quality standard for comparison, the RAND Corporation recommendation of 60 days from abnormal imaging to diagnosis and the Dayton VAMC published mean of 35.5 days are guideposts; however, the results from the Dayton VAMC may have been affected negatively by some subjects undergoing serial imaging for asymptomatic nodules. We chose a more stringent standard of 30 days as recommended by Swedish and Canadian task forces.
When diagnosing lung cancer, the overriding purpose of the Diagnostic Clinic is to minimize system delays. The method is to have as simple a task as possible for the PCP or other provider who identifies a lung nodule or mass and submits a single consultation request to the Diagnostic Clinic. Once this consultation is placed, the clinic RN manager oversees all further steps required for diagnosis and referral for treatment. The key factor in achieving a mean diagnosis time of 22.6 days is the cooperation between the RN manager and the interventional pulmonologist. When a consultation is received, the RN manager and pulmonologist review the data together and schedule the initial clinic visit; the goal is same-day biopsy, which is achieved in more than one-third of cases. Not all patients with a chest image suspected for lung cancer had it ordered by their PCP. For this reason, a Diagnostic Clinic consultation is available to all health care providers in our system. Many patients reach the clinic after the discovery of a suspect chest X-ray during an emergency department visit, a regularly scheduled subspecialty appointment, or during a preoperative evaluation.
The mean time from initial visit to biopsy was 1.8 days for EBUS/EUS compared with an interval of 11.3 days for CT-guided biopsy. This difference reflects the pulmonologist’s involvement in initial scheduling of Diagnostic Clinic patients. The ability of the pulmonologist to provide an accurate assessment of sample adequacy and a preliminary diagnosis at bedside, with concurrent confirmation by a staff pathologist, permitted the Diagnostic Clinic to inform 40.3% of patients of the finding of malignancy on the day of biopsy. A published comparison of the onsite review of biopsy material showed our pulmonologist and staff pathologists to be equally accurate in their interpretations.11
Sources of Delays
While this study documents the shortest intervals from suspect imaging to diagnosis reported to date, it also identifies sources of system delay in diagnosing lung cancer that JLMMVH could further optimize. The first is the time from initial abnormal chest X-ray imaging to performance of the chest CT scan. On occasion, the index lung lesion is identified unexpectedly on an outpatient or emergency department chest CT scan. With greater use of LDCT lung cancer screening, the initial detection of suspect lesions by CT scanning will increase in the future. However, the PCP most often investigates a patient complaint with a standard chest X-ray that reveals a suspect nodule or mass. When ordered by the PCP as an outpatient test, scheduling of the follow-up chest CT scan is not given priority. More than a third of subjects experienced a delay ≥ 1 week in obtaining a chest CT scan ordered by the PCP; for 29 subjects the delay was ≥ 3weeks. At JLMMVH, the Diagnostic Clinic is given priority in scheduling CT scans. Hence, for suspect lung lesions, the chest CT scan, if not already obtained, is generally performed on the morning of the clinic visit. Educating the PCP to refer the patient immediately to the Diagnostic Clinic rather than waiting to obtain an outpatient chest CT scan may remove this source of unnecessary delay.
Scheduling a CT-guided fine needle aspiration of a lung lesion is another source of system delay. When the chest CT scan is available at the time of the Diagnostic Clinic referral, the clinic visit is scheduled for the earliest day a required CT-guided biopsy can be performed. However, the mean time of 11.3 days from initial Diagnostic Clinic visit to CT-guided biopsy is indicative of the backlog faced by the interventional radiologists.
Although infrequent, PET scans that are required before biopsy can lead to substantial delays. PET scans are performed at our university affiliate, and the joint VA-university lung tumor board sometimes generates requests for such scans prior to tissue diagnosis, yet another source of delay.
The time from referral receipt to the Diagnostic Clinic visit averaged 6.3 days. This delay usually was determined by the availability of the CT-guided biopsy or the dedicated interventional pulmonologist. Although other interventional pulmonologists at JLMMVH may perform the requisite diagnostic procedures, they are not always available for immediate review of imaging studies of referred patients nor can their schedules flexibly accommodate the number of patients seen in our clinic for evaluation.
Lung Cancer Diagnosis
Prompt diagnosis in the setting of a worrisome chest X-ray may help decrease patient anxiety, but does the clinic improve lung cancer treatment outcomes? Such improvement has been demonstrated only in stage IA squamous cell lung cancer.9 Of our study population, 37.7% had squamous cell carcinoma, and 85.5% had non-small cell lung cancer. Of those with non-small cell lung cancer, 28.9% had a clinical stage I tumor. Stage I squamous cell carcinoma, the type of tumor most likely to benefit from early diagnosis and treatment, was diagnosed in 11.3% of patients. With the increased application of LDCT screening, the proportion of veterans identified with early stage lung cancer may rise. The Providence VAMC in Rhode Island reported its results from instituting LDCT screening.12 Prior to screening, 28% of patients diagnosed with lung cancer had a stage I tumor. Following the introduction of LDCT screening, 49% diagnosed by LDCT screening had a stage I tumor. Nearly a third of their patients diagnosed with lung cancer through LDCT screening had squamous cell tumor histology. Thus, we can anticipate an increasing number of veterans with early stage lung cancer who would benefit from timely diagnosis.
The JLMMVH is a referral center for the entire state of Arkansas. Quite a few of its referred patients come from a long distance, which may require overnight housing and other related travel expenses. Apart from any potential outcome benefit, the efficiencies of the system described herein include the minimization of extra trips, an inconvenience and cost to both patient and JLMMVH.
Although the primary task of the clinic is diagnosis, we also seek to facilitate timely treatment. Our lack of an on-site PET scanner and radiation therapy, resources present on-site at the Dayton VAMC, contribute to longer therapy wait times. The shortest mean wait time at JLMMVH is for chemotherapy alone (34.7 days), in part because the JLMMVH oncologists, performing initial consultations 2 to 3 times weekly in the Diagnostic Clinic, are more readily available than are our thoracic surgeons or radiation therapists. Yet overall, JLMMVH patients often face delay from the time of lung cancer diagnosis to initiation of treatment.
The Connecticut Veterans Affairs Healthcare System has published the results of changes in lung cancer management associated with a nurse navigator system.10 Prior to creating the position of cancer care coordinator, filled by an advanced practice RNs, the mean time from clinical suspicion of lung cancer to treatment was 117 days. After 4 years of such care navigation, this waiting time had decreased to 52.4 days. Associated with this dramatic improvement in overall waiting time were decreases in the turnaround time required for performance of CT and PET scans. With respect to this big picture view of lung cancer care, our Diagnostic Clinic serves as a model for the initial step of diagnosis. Coordination and streamlining of the various steps from diagnosis to definitive therapy shall require a more system-wide effort involving all the key players in cancer care.
Conclusion
We have developed a care pathway based in a dedicated diagnostic clinic and have been able to document the shortest interval from abnormality to diagnosis of lung cancer reported in the literature to date. Efficient functioning of this clinic is dependent upon the close cooperation between a full-time RN clinic manager and an interventional pulmonologist experienced in lung cancer management and able to interpret cytologic samples at the time of biopsy. Shortening the delay between diagnosis and definitive therapy remains a challenge and may benefit from the oncology nurse navigator model previously described within the VA system. 10
1. American Cancer Society. Cancer Facts & Figures. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2018/cancer-facts-and-figures-2018.pdf. Accessed July 13, 2019.
2. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Eng J Med. 2011;365(5):395-409.
3. Kinsinger LS, Anderson C, Kim J, et al. Implementation of lung cancer screening in the Veterans Health Administration. JAMA Intern Med. 2017;177(3):399-406.
4. Asch SM, Kerr EA, Hamilton EG, Reifel JL, McGlynn EA, eds. Quality of Care for Oncologic Conditions and HIV: A Review of the Literature and Quality Indicators. Santa Monica, CA: RAND Corporation; 2000.
5. Hillerdal G. [Recommendations from the Swedish Lung Cancer Study Group: Shorter waiting times are demanded for quality in diagnostic work-ups for lung care.] Swedish Med J 1999; 96: 4691.
6. Simunovic M, Gagliardi A, McCready D, Coates A, Levine M, DePetrillo D. A snapshot of waiting times for cancer surgery provided by surgeons affiliated with regional cancer centres in Ontario. CMAJ. 2001;165(4):421-425. [Canadian Strategy for Cancer Control]
7. Bukhari A, Kumar G, Rajsheker R, Markert R. Timeliness of lung cancer diagnosis and treatment. Fed Pract. 2017;34(suppl 1):24S-29S.
8. Bilimoria KY, Ko CY, Tomlinson JS, et al. Wait times for cancer surgery in the United States: trends and predictors of delays. Ann Surg. 2011;253(4):779-785.
9. Yang CJ, Wang H, Kumar A, et al. Impact of timing of lobectomy on survival for clinical stage IA lung squamous cell carcinoma. Chest. 2017;152(6):1239-1250.
10. Hunnibell LS, Rose MG, Connery DM, et al. Using nurse navigation to improve timeliness of lung cancer care at a veterans hospital. Clin J Oncol Nurs. 2012;16(1):29-36.
11. Meena N, Jeffus S, Massoll N, et al. Rapid onsite evaluation: a comparison of cytopathologist and pulmonologist performance. Cancer Cytopatho. 2016;124(4):279-84.
12. Okereke IC, Bates MF, Jankowich MD, et al. Effects of implementation of lung cancer screening at one Veterans Affairs Medical Center. Chest 2016;150(5):1023-1029.
1. American Cancer Society. Cancer Facts & Figures. https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/annual-cancer-facts-and-figures/2018/cancer-facts-and-figures-2018.pdf. Accessed July 13, 2019.
2. National Lung Screening Trial Research Team, Aberle DR, Adams AM, et al. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Eng J Med. 2011;365(5):395-409.
3. Kinsinger LS, Anderson C, Kim J, et al. Implementation of lung cancer screening in the Veterans Health Administration. JAMA Intern Med. 2017;177(3):399-406.
4. Asch SM, Kerr EA, Hamilton EG, Reifel JL, McGlynn EA, eds. Quality of Care for Oncologic Conditions and HIV: A Review of the Literature and Quality Indicators. Santa Monica, CA: RAND Corporation; 2000.
5. Hillerdal G. [Recommendations from the Swedish Lung Cancer Study Group: Shorter waiting times are demanded for quality in diagnostic work-ups for lung care.] Swedish Med J 1999; 96: 4691.
6. Simunovic M, Gagliardi A, McCready D, Coates A, Levine M, DePetrillo D. A snapshot of waiting times for cancer surgery provided by surgeons affiliated with regional cancer centres in Ontario. CMAJ. 2001;165(4):421-425. [Canadian Strategy for Cancer Control]
7. Bukhari A, Kumar G, Rajsheker R, Markert R. Timeliness of lung cancer diagnosis and treatment. Fed Pract. 2017;34(suppl 1):24S-29S.
8. Bilimoria KY, Ko CY, Tomlinson JS, et al. Wait times for cancer surgery in the United States: trends and predictors of delays. Ann Surg. 2011;253(4):779-785.
9. Yang CJ, Wang H, Kumar A, et al. Impact of timing of lobectomy on survival for clinical stage IA lung squamous cell carcinoma. Chest. 2017;152(6):1239-1250.
10. Hunnibell LS, Rose MG, Connery DM, et al. Using nurse navigation to improve timeliness of lung cancer care at a veterans hospital. Clin J Oncol Nurs. 2012;16(1):29-36.
11. Meena N, Jeffus S, Massoll N, et al. Rapid onsite evaluation: a comparison of cytopathologist and pulmonologist performance. Cancer Cytopatho. 2016;124(4):279-84.
12. Okereke IC, Bates MF, Jankowich MD, et al. Effects of implementation of lung cancer screening at one Veterans Affairs Medical Center. Chest 2016;150(5):1023-1029.
EHR-derived data on tumor progression may predict survival
Real-world electronic health record data on tumor progression appears to be a good surrogate for overall survival, a finding that could help to improve estimates of benefit for complex cancer therapy regimens for use in both clinical trials and treatment planning, investigators say.
A study of electronic health record (EHR) data on more than 30,000 patients with non–small cell lung cancer (NSCLC) showed that real-world data on progression-free survival (PFS) showed a high degree of correlation with overall survival (OS), reported Sandra D. Griffith, PhD, of Flatiron Health, New York, and colleagues.
“As survival rates have improved and more than one therapy line has become typical, OS has become an inadequate metric for ascertaining the benefit of some cancer treatment interventions. Intermediate endpoints are essential for evaluating treatment benefits in real-world contexts. This study presents a scalable, feasible, and replicable approach to yield an EHR-generated progression variable ready for incorporation into large, contemporary real-world analyses,” they wrote in JCO Clinical Cancer Informatics.
The investigators studied retrospectively whether it would be feasible on a large scale to extract clinically relevant endpoints from an EHR-derived database that relies on technology-assisted abstraction of data from patient charts.
Their cohort included 30,276 patients diagnosed with advanced NSCLC from January 2011 through February 2018 who had two or more visits documented on EHR and who had been started on one or more lines of systemic therapy. Of this group, 16,606 had one or more documented tumor progression events, and 11,366 either died, discontinued therapy, or started on a new therapy.
The investigators found that among 20,000 evaluable patients, correlation of real-word PFS with OS was moderately high (Spearman’s P = .076), although for 11,902 patients with a progression event who died the correlation of EHR data with actual OS was somewhat lower (Spearman’s P = .069).
The strength of the correlations was verified in an duplicate random sample reviewed by independent abstractors.
The median time to abstract data on disease progression from individual electronic health records was 18 minutes.
“More work is needed to document how real-world progression under different treatments relates to treatment effects observed in clinical trials; understand the impact of different imaging and assessment cadences—for example, informative censoring; and clarify the relationship between real-world progression and other endpoints, such as real-world tumor response. Similar work is also needed in other tumor types,” Dr. Griffiths and colleagues wrote.
The study was supported by Flatiron Health, a subsidiary of Roche. Dr. Griffith and multiple co-authors are employees and stockholders of Flatiron Health and/or Roche.
SOURCE: Griffith SD et al. JCO Clinical Informatics. 2019 Aug 12. doi: 10.1200/CCI.19.00013.
Real-world electronic health record data on tumor progression appears to be a good surrogate for overall survival, a finding that could help to improve estimates of benefit for complex cancer therapy regimens for use in both clinical trials and treatment planning, investigators say.
A study of electronic health record (EHR) data on more than 30,000 patients with non–small cell lung cancer (NSCLC) showed that real-world data on progression-free survival (PFS) showed a high degree of correlation with overall survival (OS), reported Sandra D. Griffith, PhD, of Flatiron Health, New York, and colleagues.
“As survival rates have improved and more than one therapy line has become typical, OS has become an inadequate metric for ascertaining the benefit of some cancer treatment interventions. Intermediate endpoints are essential for evaluating treatment benefits in real-world contexts. This study presents a scalable, feasible, and replicable approach to yield an EHR-generated progression variable ready for incorporation into large, contemporary real-world analyses,” they wrote in JCO Clinical Cancer Informatics.
The investigators studied retrospectively whether it would be feasible on a large scale to extract clinically relevant endpoints from an EHR-derived database that relies on technology-assisted abstraction of data from patient charts.
Their cohort included 30,276 patients diagnosed with advanced NSCLC from January 2011 through February 2018 who had two or more visits documented on EHR and who had been started on one or more lines of systemic therapy. Of this group, 16,606 had one or more documented tumor progression events, and 11,366 either died, discontinued therapy, or started on a new therapy.
The investigators found that among 20,000 evaluable patients, correlation of real-word PFS with OS was moderately high (Spearman’s P = .076), although for 11,902 patients with a progression event who died the correlation of EHR data with actual OS was somewhat lower (Spearman’s P = .069).
The strength of the correlations was verified in an duplicate random sample reviewed by independent abstractors.
The median time to abstract data on disease progression from individual electronic health records was 18 minutes.
“More work is needed to document how real-world progression under different treatments relates to treatment effects observed in clinical trials; understand the impact of different imaging and assessment cadences—for example, informative censoring; and clarify the relationship between real-world progression and other endpoints, such as real-world tumor response. Similar work is also needed in other tumor types,” Dr. Griffiths and colleagues wrote.
The study was supported by Flatiron Health, a subsidiary of Roche. Dr. Griffith and multiple co-authors are employees and stockholders of Flatiron Health and/or Roche.
SOURCE: Griffith SD et al. JCO Clinical Informatics. 2019 Aug 12. doi: 10.1200/CCI.19.00013.
Real-world electronic health record data on tumor progression appears to be a good surrogate for overall survival, a finding that could help to improve estimates of benefit for complex cancer therapy regimens for use in both clinical trials and treatment planning, investigators say.
A study of electronic health record (EHR) data on more than 30,000 patients with non–small cell lung cancer (NSCLC) showed that real-world data on progression-free survival (PFS) showed a high degree of correlation with overall survival (OS), reported Sandra D. Griffith, PhD, of Flatiron Health, New York, and colleagues.
“As survival rates have improved and more than one therapy line has become typical, OS has become an inadequate metric for ascertaining the benefit of some cancer treatment interventions. Intermediate endpoints are essential for evaluating treatment benefits in real-world contexts. This study presents a scalable, feasible, and replicable approach to yield an EHR-generated progression variable ready for incorporation into large, contemporary real-world analyses,” they wrote in JCO Clinical Cancer Informatics.
The investigators studied retrospectively whether it would be feasible on a large scale to extract clinically relevant endpoints from an EHR-derived database that relies on technology-assisted abstraction of data from patient charts.
Their cohort included 30,276 patients diagnosed with advanced NSCLC from January 2011 through February 2018 who had two or more visits documented on EHR and who had been started on one or more lines of systemic therapy. Of this group, 16,606 had one or more documented tumor progression events, and 11,366 either died, discontinued therapy, or started on a new therapy.
The investigators found that among 20,000 evaluable patients, correlation of real-word PFS with OS was moderately high (Spearman’s P = .076), although for 11,902 patients with a progression event who died the correlation of EHR data with actual OS was somewhat lower (Spearman’s P = .069).
The strength of the correlations was verified in an duplicate random sample reviewed by independent abstractors.
The median time to abstract data on disease progression from individual electronic health records was 18 minutes.
“More work is needed to document how real-world progression under different treatments relates to treatment effects observed in clinical trials; understand the impact of different imaging and assessment cadences—for example, informative censoring; and clarify the relationship between real-world progression and other endpoints, such as real-world tumor response. Similar work is also needed in other tumor types,” Dr. Griffiths and colleagues wrote.
The study was supported by Flatiron Health, a subsidiary of Roche. Dr. Griffith and multiple co-authors are employees and stockholders of Flatiron Health and/or Roche.
SOURCE: Griffith SD et al. JCO Clinical Informatics. 2019 Aug 12. doi: 10.1200/CCI.19.00013.
FROM JCO CLINICAL INFORMATICS
Screening for pancreatic and lung cancers
In this edition of “How I will treat my next patient,” I examine the U.S. Preventive Services Task Force Reaffirmation Recommendation Statement regarding screening for pancreatic cancer in normal-risk populations. I also review newly published information regarding low-dose CT screening (LDCT) for lung cancer in a commonly screened population – individuals with chronic obstructive pulmonary disease (COPD). Both publications highlight the complexity of implementing shared decision making in clinicians’ efforts to find these highly lethal cancers in their earliest, most curable stages.
Pancreatic cancer screening
In their recommendation, the USPSTF considered data relevant to the benefits and harms (exclusive of costs) of screening for pancreatic cancer in the 85%-90% of individuals who are at normal risk because they lack a known familial or genetic syndrome and do not have at least two affected relatives or one first-degree affected relative (JAMA. 2019;322[5]:438-44).
After reviewing 13 cohort studies employing image-based technologies (CT, MRI, endoscopic ultrasound) and biomarkers for screening, the USPSTF reaffirmed its 2004 recommendation against pancreatic cancer screening. They found no new evidence of sufficient strength and quality to alter their previous “D grade” for screening (i.e., “Don’t do it.”). There were at least moderate harms of screening and subsequent treatment in normal-risk populations. These recommendations apply to asymptomatic individuals with new-onset diabetes mellitus, smokers, older adults, obese patients, and patients with a history of chronic pancreatitis.
What this means in practice
In the nicely written and comprehensive recommendation statement and in the two accompanying editorials (JAMA Surg. 2019 Aug 6. doi: 10.1001/jamasurg.2019.2832; JAMA. 2019;322[5]:407-8), the authors were explicit that the “D grade” for pancreatic cancer screening did not apply to individuals from familial pancreatic cancer kindreds and those with germline mutations and Lynch syndrome mismatch repair genes. For them, the relative risk of pancreatic cancer (greater than 5%) may justify the morbidity of available surveillance technologies, especially since U.S. and International screening studies in these high-risk individuals have generated data suggesting a benefit for treatment of screen-detected cancers.
The USPSTF and the editorial authors were strongly supportive of, and enthusiastic about, ongoing research efforts. Recently, a joint effort at the National Institutes of Health began recruiting centers for a study to assess the sensitivity of novel biomarkers in detecting pancreatic cancer among adults with new-onset diabetes (A211701).
To me, it is clear that the pathway to identifying effective screening for pancreatic cancer – which is forecast to become the second leading cause of cancer death in the United States by 2020 – will focus on high-risk populations first, enabling accurate determination of sensitivity and specificity before being applied to the general population. This is as it should be.
Lung cancer screening
Currently, guidelines from the National Comprehensive Cancer Network recommend LDCT screening annually for high-risk smokers, former smokers, and individuals with additional risk factors aged 55-77 years. The National Lung Screening Trial indicated that LDCT screening achieved a 20% relative reduction in lung cancer mortality and 6.7% relative reduction in overall mortality in a similar population. NCCN guidelines stress the importance of shared decision making and include a table of risks and benefits that should be considered.
Jonathan M. Iaccarino, MD, and colleagues quantified the risks of screening among COPD patients in a secondary analysis of the more than 75,000 LDCT scans that were performed among the more than 26,000 participants in the National Lung Screening Trial (Chest 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016). In comparison with participants who did not self-report a diagnosis of COPD, the 4,632 participants with self-reported COPD were significantly more likely to require further diagnostic studies, have an invasive procedure, have a complication of any type from the invasive procedure, and suffer a serious complication. The establishment of a lung cancer diagnosis from the invasive procedure, however, occurred in just 6.1% of COPD patients versus 3.6% of patients without COPD.
What this means in practice
At a consensus conference convened by the National Quality Forum, shared decision making was defined as a process of communication in which clinicians and patients work together to make decisions that align with what matters most to patients. Ideally, shared decision making requires clear, accurate, unbiased medical evidence about reasonable alternatives; tailored evidence for individual patients; and the incorporation of patient values, goals, informed preferences and concerns, including a discussion of treatment burdens. All of us wrestle with the challenge of conducting these conversations in a comprehensive and unbiased manner. I am not sure that I have ever achieved an ideal shared decision-making conversation in my practice.
Despite the limitations acknowledged by the authors – self-reported diagnosis of COPD, outcomes that were not the primary focus of the trial, failure to incorporate other important comorbid conditions – the study by Dr. Iaccarino and colleagues helps to quantify risks and benefits for a commonly screened population, specifically COPD patients. Most importantly, it focuses our attention on the key goal of all cancer-screening efforts – applying our personal and technological resources to patients who benefit the most and will suffer the least harm from our efforts.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I will treat my next patient,” I examine the U.S. Preventive Services Task Force Reaffirmation Recommendation Statement regarding screening for pancreatic cancer in normal-risk populations. I also review newly published information regarding low-dose CT screening (LDCT) for lung cancer in a commonly screened population – individuals with chronic obstructive pulmonary disease (COPD). Both publications highlight the complexity of implementing shared decision making in clinicians’ efforts to find these highly lethal cancers in their earliest, most curable stages.
Pancreatic cancer screening
In their recommendation, the USPSTF considered data relevant to the benefits and harms (exclusive of costs) of screening for pancreatic cancer in the 85%-90% of individuals who are at normal risk because they lack a known familial or genetic syndrome and do not have at least two affected relatives or one first-degree affected relative (JAMA. 2019;322[5]:438-44).
After reviewing 13 cohort studies employing image-based technologies (CT, MRI, endoscopic ultrasound) and biomarkers for screening, the USPSTF reaffirmed its 2004 recommendation against pancreatic cancer screening. They found no new evidence of sufficient strength and quality to alter their previous “D grade” for screening (i.e., “Don’t do it.”). There were at least moderate harms of screening and subsequent treatment in normal-risk populations. These recommendations apply to asymptomatic individuals with new-onset diabetes mellitus, smokers, older adults, obese patients, and patients with a history of chronic pancreatitis.
What this means in practice
In the nicely written and comprehensive recommendation statement and in the two accompanying editorials (JAMA Surg. 2019 Aug 6. doi: 10.1001/jamasurg.2019.2832; JAMA. 2019;322[5]:407-8), the authors were explicit that the “D grade” for pancreatic cancer screening did not apply to individuals from familial pancreatic cancer kindreds and those with germline mutations and Lynch syndrome mismatch repair genes. For them, the relative risk of pancreatic cancer (greater than 5%) may justify the morbidity of available surveillance technologies, especially since U.S. and International screening studies in these high-risk individuals have generated data suggesting a benefit for treatment of screen-detected cancers.
The USPSTF and the editorial authors were strongly supportive of, and enthusiastic about, ongoing research efforts. Recently, a joint effort at the National Institutes of Health began recruiting centers for a study to assess the sensitivity of novel biomarkers in detecting pancreatic cancer among adults with new-onset diabetes (A211701).
To me, it is clear that the pathway to identifying effective screening for pancreatic cancer – which is forecast to become the second leading cause of cancer death in the United States by 2020 – will focus on high-risk populations first, enabling accurate determination of sensitivity and specificity before being applied to the general population. This is as it should be.
Lung cancer screening
Currently, guidelines from the National Comprehensive Cancer Network recommend LDCT screening annually for high-risk smokers, former smokers, and individuals with additional risk factors aged 55-77 years. The National Lung Screening Trial indicated that LDCT screening achieved a 20% relative reduction in lung cancer mortality and 6.7% relative reduction in overall mortality in a similar population. NCCN guidelines stress the importance of shared decision making and include a table of risks and benefits that should be considered.
Jonathan M. Iaccarino, MD, and colleagues quantified the risks of screening among COPD patients in a secondary analysis of the more than 75,000 LDCT scans that were performed among the more than 26,000 participants in the National Lung Screening Trial (Chest 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016). In comparison with participants who did not self-report a diagnosis of COPD, the 4,632 participants with self-reported COPD were significantly more likely to require further diagnostic studies, have an invasive procedure, have a complication of any type from the invasive procedure, and suffer a serious complication. The establishment of a lung cancer diagnosis from the invasive procedure, however, occurred in just 6.1% of COPD patients versus 3.6% of patients without COPD.
What this means in practice
At a consensus conference convened by the National Quality Forum, shared decision making was defined as a process of communication in which clinicians and patients work together to make decisions that align with what matters most to patients. Ideally, shared decision making requires clear, accurate, unbiased medical evidence about reasonable alternatives; tailored evidence for individual patients; and the incorporation of patient values, goals, informed preferences and concerns, including a discussion of treatment burdens. All of us wrestle with the challenge of conducting these conversations in a comprehensive and unbiased manner. I am not sure that I have ever achieved an ideal shared decision-making conversation in my practice.
Despite the limitations acknowledged by the authors – self-reported diagnosis of COPD, outcomes that were not the primary focus of the trial, failure to incorporate other important comorbid conditions – the study by Dr. Iaccarino and colleagues helps to quantify risks and benefits for a commonly screened population, specifically COPD patients. Most importantly, it focuses our attention on the key goal of all cancer-screening efforts – applying our personal and technological resources to patients who benefit the most and will suffer the least harm from our efforts.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
In this edition of “How I will treat my next patient,” I examine the U.S. Preventive Services Task Force Reaffirmation Recommendation Statement regarding screening for pancreatic cancer in normal-risk populations. I also review newly published information regarding low-dose CT screening (LDCT) for lung cancer in a commonly screened population – individuals with chronic obstructive pulmonary disease (COPD). Both publications highlight the complexity of implementing shared decision making in clinicians’ efforts to find these highly lethal cancers in their earliest, most curable stages.
Pancreatic cancer screening
In their recommendation, the USPSTF considered data relevant to the benefits and harms (exclusive of costs) of screening for pancreatic cancer in the 85%-90% of individuals who are at normal risk because they lack a known familial or genetic syndrome and do not have at least two affected relatives or one first-degree affected relative (JAMA. 2019;322[5]:438-44).
After reviewing 13 cohort studies employing image-based technologies (CT, MRI, endoscopic ultrasound) and biomarkers for screening, the USPSTF reaffirmed its 2004 recommendation against pancreatic cancer screening. They found no new evidence of sufficient strength and quality to alter their previous “D grade” for screening (i.e., “Don’t do it.”). There were at least moderate harms of screening and subsequent treatment in normal-risk populations. These recommendations apply to asymptomatic individuals with new-onset diabetes mellitus, smokers, older adults, obese patients, and patients with a history of chronic pancreatitis.
What this means in practice
In the nicely written and comprehensive recommendation statement and in the two accompanying editorials (JAMA Surg. 2019 Aug 6. doi: 10.1001/jamasurg.2019.2832; JAMA. 2019;322[5]:407-8), the authors were explicit that the “D grade” for pancreatic cancer screening did not apply to individuals from familial pancreatic cancer kindreds and those with germline mutations and Lynch syndrome mismatch repair genes. For them, the relative risk of pancreatic cancer (greater than 5%) may justify the morbidity of available surveillance technologies, especially since U.S. and International screening studies in these high-risk individuals have generated data suggesting a benefit for treatment of screen-detected cancers.
The USPSTF and the editorial authors were strongly supportive of, and enthusiastic about, ongoing research efforts. Recently, a joint effort at the National Institutes of Health began recruiting centers for a study to assess the sensitivity of novel biomarkers in detecting pancreatic cancer among adults with new-onset diabetes (A211701).
To me, it is clear that the pathway to identifying effective screening for pancreatic cancer – which is forecast to become the second leading cause of cancer death in the United States by 2020 – will focus on high-risk populations first, enabling accurate determination of sensitivity and specificity before being applied to the general population. This is as it should be.
Lung cancer screening
Currently, guidelines from the National Comprehensive Cancer Network recommend LDCT screening annually for high-risk smokers, former smokers, and individuals with additional risk factors aged 55-77 years. The National Lung Screening Trial indicated that LDCT screening achieved a 20% relative reduction in lung cancer mortality and 6.7% relative reduction in overall mortality in a similar population. NCCN guidelines stress the importance of shared decision making and include a table of risks and benefits that should be considered.
Jonathan M. Iaccarino, MD, and colleagues quantified the risks of screening among COPD patients in a secondary analysis of the more than 75,000 LDCT scans that were performed among the more than 26,000 participants in the National Lung Screening Trial (Chest 2019 Jul 5. doi: 10.1016/j.chest.2019.06.016). In comparison with participants who did not self-report a diagnosis of COPD, the 4,632 participants with self-reported COPD were significantly more likely to require further diagnostic studies, have an invasive procedure, have a complication of any type from the invasive procedure, and suffer a serious complication. The establishment of a lung cancer diagnosis from the invasive procedure, however, occurred in just 6.1% of COPD patients versus 3.6% of patients without COPD.
What this means in practice
At a consensus conference convened by the National Quality Forum, shared decision making was defined as a process of communication in which clinicians and patients work together to make decisions that align with what matters most to patients. Ideally, shared decision making requires clear, accurate, unbiased medical evidence about reasonable alternatives; tailored evidence for individual patients; and the incorporation of patient values, goals, informed preferences and concerns, including a discussion of treatment burdens. All of us wrestle with the challenge of conducting these conversations in a comprehensive and unbiased manner. I am not sure that I have ever achieved an ideal shared decision-making conversation in my practice.
Despite the limitations acknowledged by the authors – self-reported diagnosis of COPD, outcomes that were not the primary focus of the trial, failure to incorporate other important comorbid conditions – the study by Dr. Iaccarino and colleagues helps to quantify risks and benefits for a commonly screened population, specifically COPD patients. Most importantly, it focuses our attention on the key goal of all cancer-screening efforts – applying our personal and technological resources to patients who benefit the most and will suffer the least harm from our efforts.
Dr. Lyss has been a community-based medical oncologist and clinical researcher for more than 35 years, practicing in St. Louis. His clinical and research interests are in the prevention, diagnosis, and treatment of breast and lung cancers and in expanding access to clinical trials to medically underserved populations.
Rozlytrek approved for ROS1-positive metastatic NSCLC, cancers with NTRK gene fusion defects
Rozlytrek (entrectinib) has been approved to treat cancers with neurotrophic tyrosine receptor kinase (NTRK) gene fusion defects in adults and adolescents for whom there are no effective treatments, the Food and Drug Administration announced in a press release.
Entrectinib was also approved for the treatment of adults with metastatic non–small cell lung cancers that are ROS1-positive.
“We are in an exciting era of innovation in cancer treatment as we continue to see development in tissue-agnostic therapies, which have the potential to transform cancer treatment. We’re seeing continued advances in the use of biomarkers to guide drug development and the more targeted delivery of medicine,” FDA Acting Commissioner Ned Sharpless, MD, said in the release.
This is the third time the agency has approved a cancer treatment based on a common biomarker across different types of tumors rather than on the original tumor’s location. The previous tissue-agnostic indications approved by the FDA were pembrolizumab (Keytruda) for tumors with microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) tumors in 2017 and larotrectinib (Vitrakvi) for NTRK gene fusion tumors in 2018.
The approval of entrectinib was granted to Genentech. “Rozlytrek is the first FDA-approved treatment that selectively targets both ROS1 and NTRK fusions, and, importantly, has also shown responses in these rare cancer types that have spread to the brain,” Sandra Horning, MD, chief medical officer and head of global product development for Genentech, said in a separate press release.
Foundation Medicine will submit Foundation One CDx to the FDA for approval as a companion diagnostic for entrectinib, according to the Genentech release; an FDA-approved companion diagnostic for entrectinib is not available at this time.
“Today’s approval includes an indication for pediatric patients, 12 years of age and older, who have NTRK fusion–positive tumors by relying on efficacy information obtained primarily in adults,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Efficacy in adolescents was derived from adult data and safety was demonstrated in 30 pediatric patients.”
Entrectinib was evaluated in four clinical trials that included 54 adults with NTRK fusion–positive tumors. The overall response rate was 57%, with 7.4% of patients having complete disappearance of the tumor. Among the 31 patients with tumor shrinkage, 61% had tumor shrinkage persist for 9 months or longer. The most common cancer locations were the lung, salivary gland, breast, thyroid, and colon/rectum.
Clinical studies evaluated 51 adults with ROS1-positive lung cancer. The overall response rate was 78%, with 5.9% of patients having complete disappearance of their cancer. Among the 40 patients with tumor shrinkage, 55% had tumor shrinkage persist for 12 months or longer.
The most serious side effects of entrectinib are heart failure, central nervous system effects, changes in sleep pattern, skeletal fractures, hepatotoxicity, hyperuricemia, QT prolongation, and vision disorders. Females of reproductive age and males with a female partner of reproductive potential are advised to use effective contraception during treatment; the drug may cause harm to a developing fetus or newborn baby.
Genentech must provide additional clinical trial data to the FDA as a condition of the approval.
Rozlytrek (entrectinib) has been approved to treat cancers with neurotrophic tyrosine receptor kinase (NTRK) gene fusion defects in adults and adolescents for whom there are no effective treatments, the Food and Drug Administration announced in a press release.
Entrectinib was also approved for the treatment of adults with metastatic non–small cell lung cancers that are ROS1-positive.
“We are in an exciting era of innovation in cancer treatment as we continue to see development in tissue-agnostic therapies, which have the potential to transform cancer treatment. We’re seeing continued advances in the use of biomarkers to guide drug development and the more targeted delivery of medicine,” FDA Acting Commissioner Ned Sharpless, MD, said in the release.
This is the third time the agency has approved a cancer treatment based on a common biomarker across different types of tumors rather than on the original tumor’s location. The previous tissue-agnostic indications approved by the FDA were pembrolizumab (Keytruda) for tumors with microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) tumors in 2017 and larotrectinib (Vitrakvi) for NTRK gene fusion tumors in 2018.
The approval of entrectinib was granted to Genentech. “Rozlytrek is the first FDA-approved treatment that selectively targets both ROS1 and NTRK fusions, and, importantly, has also shown responses in these rare cancer types that have spread to the brain,” Sandra Horning, MD, chief medical officer and head of global product development for Genentech, said in a separate press release.
Foundation Medicine will submit Foundation One CDx to the FDA for approval as a companion diagnostic for entrectinib, according to the Genentech release; an FDA-approved companion diagnostic for entrectinib is not available at this time.
“Today’s approval includes an indication for pediatric patients, 12 years of age and older, who have NTRK fusion–positive tumors by relying on efficacy information obtained primarily in adults,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Efficacy in adolescents was derived from adult data and safety was demonstrated in 30 pediatric patients.”
Entrectinib was evaluated in four clinical trials that included 54 adults with NTRK fusion–positive tumors. The overall response rate was 57%, with 7.4% of patients having complete disappearance of the tumor. Among the 31 patients with tumor shrinkage, 61% had tumor shrinkage persist for 9 months or longer. The most common cancer locations were the lung, salivary gland, breast, thyroid, and colon/rectum.
Clinical studies evaluated 51 adults with ROS1-positive lung cancer. The overall response rate was 78%, with 5.9% of patients having complete disappearance of their cancer. Among the 40 patients with tumor shrinkage, 55% had tumor shrinkage persist for 12 months or longer.
The most serious side effects of entrectinib are heart failure, central nervous system effects, changes in sleep pattern, skeletal fractures, hepatotoxicity, hyperuricemia, QT prolongation, and vision disorders. Females of reproductive age and males with a female partner of reproductive potential are advised to use effective contraception during treatment; the drug may cause harm to a developing fetus or newborn baby.
Genentech must provide additional clinical trial data to the FDA as a condition of the approval.
Rozlytrek (entrectinib) has been approved to treat cancers with neurotrophic tyrosine receptor kinase (NTRK) gene fusion defects in adults and adolescents for whom there are no effective treatments, the Food and Drug Administration announced in a press release.
Entrectinib was also approved for the treatment of adults with metastatic non–small cell lung cancers that are ROS1-positive.
“We are in an exciting era of innovation in cancer treatment as we continue to see development in tissue-agnostic therapies, which have the potential to transform cancer treatment. We’re seeing continued advances in the use of biomarkers to guide drug development and the more targeted delivery of medicine,” FDA Acting Commissioner Ned Sharpless, MD, said in the release.
This is the third time the agency has approved a cancer treatment based on a common biomarker across different types of tumors rather than on the original tumor’s location. The previous tissue-agnostic indications approved by the FDA were pembrolizumab (Keytruda) for tumors with microsatellite instability-high (MSI-H) or mismatch repair deficient (dMMR) tumors in 2017 and larotrectinib (Vitrakvi) for NTRK gene fusion tumors in 2018.
The approval of entrectinib was granted to Genentech. “Rozlytrek is the first FDA-approved treatment that selectively targets both ROS1 and NTRK fusions, and, importantly, has also shown responses in these rare cancer types that have spread to the brain,” Sandra Horning, MD, chief medical officer and head of global product development for Genentech, said in a separate press release.
Foundation Medicine will submit Foundation One CDx to the FDA for approval as a companion diagnostic for entrectinib, according to the Genentech release; an FDA-approved companion diagnostic for entrectinib is not available at this time.
“Today’s approval includes an indication for pediatric patients, 12 years of age and older, who have NTRK fusion–positive tumors by relying on efficacy information obtained primarily in adults,” said Richard Pazdur, MD, director of the FDA’s Oncology Center of Excellence and acting director of the Office of Hematology and Oncology Products in the FDA’s Center for Drug Evaluation and Research. “Efficacy in adolescents was derived from adult data and safety was demonstrated in 30 pediatric patients.”
Entrectinib was evaluated in four clinical trials that included 54 adults with NTRK fusion–positive tumors. The overall response rate was 57%, with 7.4% of patients having complete disappearance of the tumor. Among the 31 patients with tumor shrinkage, 61% had tumor shrinkage persist for 9 months or longer. The most common cancer locations were the lung, salivary gland, breast, thyroid, and colon/rectum.
Clinical studies evaluated 51 adults with ROS1-positive lung cancer. The overall response rate was 78%, with 5.9% of patients having complete disappearance of their cancer. Among the 40 patients with tumor shrinkage, 55% had tumor shrinkage persist for 12 months or longer.
The most serious side effects of entrectinib are heart failure, central nervous system effects, changes in sleep pattern, skeletal fractures, hepatotoxicity, hyperuricemia, QT prolongation, and vision disorders. Females of reproductive age and males with a female partner of reproductive potential are advised to use effective contraception during treatment; the drug may cause harm to a developing fetus or newborn baby.
Genentech must provide additional clinical trial data to the FDA as a condition of the approval.
Survey: PCI use has declined in ES-SCLC
A nationwide survey of radiation oncologists showed a decline in the use of prophylactic cranial irradiation (PCI) for extensive-stage small cell lung cancer (ES-SCLC) after the release of phase 3 study results by Takahashi et al.
The recent study by Toshiaki Takahashi, MD, and colleagues (Lancet Oncol. 2017;18[5]:663-71) demonstrated no overall survival benefit when comparing active magnetic resonance imaging surveillance with PCI in patients with ES-SCLC. The results challenged the formerly accepted belief of the benefit of PCI for patients with ES-SCLC.
“We conducted a nationwide survey study of radiation oncologists to assess changes in the use of PCI for patients with ES-SCLC following publication of the trial by Takahashi et al.,” wrote Olsi Gjyshi, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston, and colleagues. The findings were reported in a research letter published in JAMA Network Open.
Dr. Gjyshi and colleagues distributed the anonymous survey to 3,851 United States–based radiation oncologists registered with the American Society for Radiation Oncology (ASTRO). In total, 487 members (12.6%) completed the survey.
After analysis, the researchers found that 72% of respondents regularly offered PCI to patients before the publication of the study by Takahashi et al., while only 44% do so currently (difference, 28%; 95% confidence interval, 25%-31%; P less than .001).
With respect to radiation oncologists in private versus academic settings, no significant difference in practice patterns was observed (P = .71).
“Regression analysis showed no difference in likelihood of offering PCI based on practice setting, location, or size; volume of patients treated for lung cancer; or years in practice,” the researchers wrote.
In respondents unaware of the Takahashi et al. study, 85% continued to propose PCI, which was greater than those aware of the trial (odds ratio, 0.11; 95% CI, 0.04-0.32; P less than .001).
The researchers acknowledged a key limitation of the study was that radiation oncologists familiar with the study by Dr. Takahashi and colleagues may have been more apt to respond to the survey. As a result, participation bias could have influenced the results.
“These results highlight the continued lack of consensus for PCI in SCLC and support ongoing investigations,” they concluded.
No funding sources were reported. One co-author reported financial affiliations with Novocure, Inc. The other authors reported no conflicts of interest.
SOURCE: Gjyshi O et al. JAMA Netw Open. 2019 Aug 14. doi: 10.1001/jamanetworkopen.2019.9135.
A nationwide survey of radiation oncologists showed a decline in the use of prophylactic cranial irradiation (PCI) for extensive-stage small cell lung cancer (ES-SCLC) after the release of phase 3 study results by Takahashi et al.
The recent study by Toshiaki Takahashi, MD, and colleagues (Lancet Oncol. 2017;18[5]:663-71) demonstrated no overall survival benefit when comparing active magnetic resonance imaging surveillance with PCI in patients with ES-SCLC. The results challenged the formerly accepted belief of the benefit of PCI for patients with ES-SCLC.
“We conducted a nationwide survey study of radiation oncologists to assess changes in the use of PCI for patients with ES-SCLC following publication of the trial by Takahashi et al.,” wrote Olsi Gjyshi, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston, and colleagues. The findings were reported in a research letter published in JAMA Network Open.
Dr. Gjyshi and colleagues distributed the anonymous survey to 3,851 United States–based radiation oncologists registered with the American Society for Radiation Oncology (ASTRO). In total, 487 members (12.6%) completed the survey.
After analysis, the researchers found that 72% of respondents regularly offered PCI to patients before the publication of the study by Takahashi et al., while only 44% do so currently (difference, 28%; 95% confidence interval, 25%-31%; P less than .001).
With respect to radiation oncologists in private versus academic settings, no significant difference in practice patterns was observed (P = .71).
“Regression analysis showed no difference in likelihood of offering PCI based on practice setting, location, or size; volume of patients treated for lung cancer; or years in practice,” the researchers wrote.
In respondents unaware of the Takahashi et al. study, 85% continued to propose PCI, which was greater than those aware of the trial (odds ratio, 0.11; 95% CI, 0.04-0.32; P less than .001).
The researchers acknowledged a key limitation of the study was that radiation oncologists familiar with the study by Dr. Takahashi and colleagues may have been more apt to respond to the survey. As a result, participation bias could have influenced the results.
“These results highlight the continued lack of consensus for PCI in SCLC and support ongoing investigations,” they concluded.
No funding sources were reported. One co-author reported financial affiliations with Novocure, Inc. The other authors reported no conflicts of interest.
SOURCE: Gjyshi O et al. JAMA Netw Open. 2019 Aug 14. doi: 10.1001/jamanetworkopen.2019.9135.
A nationwide survey of radiation oncologists showed a decline in the use of prophylactic cranial irradiation (PCI) for extensive-stage small cell lung cancer (ES-SCLC) after the release of phase 3 study results by Takahashi et al.
The recent study by Toshiaki Takahashi, MD, and colleagues (Lancet Oncol. 2017;18[5]:663-71) demonstrated no overall survival benefit when comparing active magnetic resonance imaging surveillance with PCI in patients with ES-SCLC. The results challenged the formerly accepted belief of the benefit of PCI for patients with ES-SCLC.
“We conducted a nationwide survey study of radiation oncologists to assess changes in the use of PCI for patients with ES-SCLC following publication of the trial by Takahashi et al.,” wrote Olsi Gjyshi, MD, PhD, of the University of Texas MD Anderson Cancer Center, Houston, and colleagues. The findings were reported in a research letter published in JAMA Network Open.
Dr. Gjyshi and colleagues distributed the anonymous survey to 3,851 United States–based radiation oncologists registered with the American Society for Radiation Oncology (ASTRO). In total, 487 members (12.6%) completed the survey.
After analysis, the researchers found that 72% of respondents regularly offered PCI to patients before the publication of the study by Takahashi et al., while only 44% do so currently (difference, 28%; 95% confidence interval, 25%-31%; P less than .001).
With respect to radiation oncologists in private versus academic settings, no significant difference in practice patterns was observed (P = .71).
“Regression analysis showed no difference in likelihood of offering PCI based on practice setting, location, or size; volume of patients treated for lung cancer; or years in practice,” the researchers wrote.
In respondents unaware of the Takahashi et al. study, 85% continued to propose PCI, which was greater than those aware of the trial (odds ratio, 0.11; 95% CI, 0.04-0.32; P less than .001).
The researchers acknowledged a key limitation of the study was that radiation oncologists familiar with the study by Dr. Takahashi and colleagues may have been more apt to respond to the survey. As a result, participation bias could have influenced the results.
“These results highlight the continued lack of consensus for PCI in SCLC and support ongoing investigations,” they concluded.
No funding sources were reported. One co-author reported financial affiliations with Novocure, Inc. The other authors reported no conflicts of interest.
SOURCE: Gjyshi O et al. JAMA Netw Open. 2019 Aug 14. doi: 10.1001/jamanetworkopen.2019.9135.
FROM JAMA NETWORK OPEN