User login
An extremely indolent T-cell leukemia: an 18-year follow-up
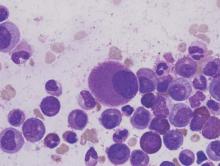
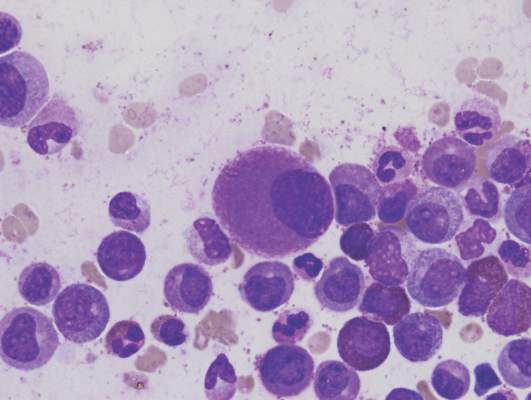
T-cell prolymphocytic leukemia (T-PLL) is a rare malignancy that comprises about 2% of all mature lymphoid neoplasms. Patients usually present with prominent peripheral blood lymphocytosis, splenomegaly, hepatomegaly, lymphadenopathy, B symptoms, and occasionally with skin lesions.1 The disease follows an aggressive clinical course with rapid progression and typically has a median survival of less than 1 year. In some cases, the disease is indolent for a period of time before becoming aggressive.2 In 2002, 7 years after initial diagnosis in 1995, the case discussed herein was reported as a rare, indolent form of T-PLL.3 We now present 11 additional years of follow-up of this case, during which time the patient remained asymptomatic with respect to his lymphoid neoplasm.
Click on the PDF icon at the top of this introduction to read the full article.
T-cell prolymphocytic leukemia (T-PLL) is a rare malignancy that comprises about 2% of all mature lymphoid neoplasms. Patients usually present with prominent peripheral blood lymphocytosis, splenomegaly, hepatomegaly, lymphadenopathy, B symptoms, and occasionally with skin lesions.1 The disease follows an aggressive clinical course with rapid progression and typically has a median survival of less than 1 year. In some cases, the disease is indolent for a period of time before becoming aggressive.2 In 2002, 7 years after initial diagnosis in 1995, the case discussed herein was reported as a rare, indolent form of T-PLL.3 We now present 11 additional years of follow-up of this case, during which time the patient remained asymptomatic with respect to his lymphoid neoplasm.
Click on the PDF icon at the top of this introduction to read the full article.
T-cell prolymphocytic leukemia (T-PLL) is a rare malignancy that comprises about 2% of all mature lymphoid neoplasms. Patients usually present with prominent peripheral blood lymphocytosis, splenomegaly, hepatomegaly, lymphadenopathy, B symptoms, and occasionally with skin lesions.1 The disease follows an aggressive clinical course with rapid progression and typically has a median survival of less than 1 year. In some cases, the disease is indolent for a period of time before becoming aggressive.2 In 2002, 7 years after initial diagnosis in 1995, the case discussed herein was reported as a rare, indolent form of T-PLL.3 We now present 11 additional years of follow-up of this case, during which time the patient remained asymptomatic with respect to his lymphoid neoplasm.
Click on the PDF icon at the top of this introduction to read the full article.
Prognostic value of complete remission with superior platelet counts in acute myeloid leukemia
Background Complete remission (CR) in acute myeloid leukemia (AML) is defined as having ≤5% leukemic blast cells in the bone marrow and return of normal hematopoiesis after the first induction cycle. There is a subset of patients, however, who achieve reduction of leukemic blast cells with a subnormal platelet count, designated as CR with incomplete platelet recovery (platelet count, ≤100,000/mcL; normal, 150,000-450,000/mcL), which is associated with inferior outcomes when compared with CR. Furthermore, there is another subset of patients with CR but superior platelet counts (≥400,000/mcL) whose prognostic significance is unclear.
Objective To establish whether CR with superior platelet counts is associated with better outcomes and can be used as a separate entity for prognostication.
Methods A retrospective chart review of 104 cases of AML was conducted. The highest platelet count during days 25-35 from initiation of induction chemotherapy (designated as day 30 platelet count) was documented. A multivariate analysis for other factors such as age, sex, risk categories, day 14+ plasma cell count (average plasma cell percentage at days 14-21), infections, allogeneic bone marrow transplant, and remission status was done.
Results Day 30 platelet count was found to be an independent predictor of survival in AML. On the multivariate analysis, the subgroup with superior platelet counts (≥400,000/mcL) was found to be associated with better outcomes.
Limitations Results need to be validated in a larger cohort.
Conclusions CR with superior platelet recovery (≥400,000/mcL) is a unique subcategory in itself and has prognostic significance. This may help better assess response to chemotherapeutic agents and aid in further decision-making regarding treatment.
Click on the PDF icon at the top of this introduction to read the full article.
Background Complete remission (CR) in acute myeloid leukemia (AML) is defined as having ≤5% leukemic blast cells in the bone marrow and return of normal hematopoiesis after the first induction cycle. There is a subset of patients, however, who achieve reduction of leukemic blast cells with a subnormal platelet count, designated as CR with incomplete platelet recovery (platelet count, ≤100,000/mcL; normal, 150,000-450,000/mcL), which is associated with inferior outcomes when compared with CR. Furthermore, there is another subset of patients with CR but superior platelet counts (≥400,000/mcL) whose prognostic significance is unclear.
Objective To establish whether CR with superior platelet counts is associated with better outcomes and can be used as a separate entity for prognostication.
Methods A retrospective chart review of 104 cases of AML was conducted. The highest platelet count during days 25-35 from initiation of induction chemotherapy (designated as day 30 platelet count) was documented. A multivariate analysis for other factors such as age, sex, risk categories, day 14+ plasma cell count (average plasma cell percentage at days 14-21), infections, allogeneic bone marrow transplant, and remission status was done.
Results Day 30 platelet count was found to be an independent predictor of survival in AML. On the multivariate analysis, the subgroup with superior platelet counts (≥400,000/mcL) was found to be associated with better outcomes.
Limitations Results need to be validated in a larger cohort.
Conclusions CR with superior platelet recovery (≥400,000/mcL) is a unique subcategory in itself and has prognostic significance. This may help better assess response to chemotherapeutic agents and aid in further decision-making regarding treatment.
Click on the PDF icon at the top of this introduction to read the full article.
Background Complete remission (CR) in acute myeloid leukemia (AML) is defined as having ≤5% leukemic blast cells in the bone marrow and return of normal hematopoiesis after the first induction cycle. There is a subset of patients, however, who achieve reduction of leukemic blast cells with a subnormal platelet count, designated as CR with incomplete platelet recovery (platelet count, ≤100,000/mcL; normal, 150,000-450,000/mcL), which is associated with inferior outcomes when compared with CR. Furthermore, there is another subset of patients with CR but superior platelet counts (≥400,000/mcL) whose prognostic significance is unclear.
Objective To establish whether CR with superior platelet counts is associated with better outcomes and can be used as a separate entity for prognostication.
Methods A retrospective chart review of 104 cases of AML was conducted. The highest platelet count during days 25-35 from initiation of induction chemotherapy (designated as day 30 platelet count) was documented. A multivariate analysis for other factors such as age, sex, risk categories, day 14+ plasma cell count (average plasma cell percentage at days 14-21), infections, allogeneic bone marrow transplant, and remission status was done.
Results Day 30 platelet count was found to be an independent predictor of survival in AML. On the multivariate analysis, the subgroup with superior platelet counts (≥400,000/mcL) was found to be associated with better outcomes.
Limitations Results need to be validated in a larger cohort.
Conclusions CR with superior platelet recovery (≥400,000/mcL) is a unique subcategory in itself and has prognostic significance. This may help better assess response to chemotherapeutic agents and aid in further decision-making regarding treatment.
Click on the PDF icon at the top of this introduction to read the full article.
Ponatinib effective in chronic phase CML regardless of baseline mutation status
In heavily pretreated patients with chronic phase chronic myeloid leukemia (CP-CML), response to the tyrosine kinase inhibitor ponatinib did not depend on baseline mutation status, and no single or compound mutation was a major driver of primary or secondary resistance to ponatinib, according to researchers.
Irrespective of baseline mutation status, responses to ponatinib were durable. As determined by next-generation sequencing (NGS), patients with zero, one, or two or more BCR-ABL1 mutations had rates of 50%-61% for major cytogenetic response (MCyR) by 1 year and 29%-45% for major molecular response (MMR) at any time. The rates were similar to those observed with mutation status determined by Sanger sequencing. Rates of sustained response at 2 years for MCyR and MMR were 87% and 65%, respectively (Blood. 2016 Feb 11. doi: 10.1182/blood-2015-08-660977).
Sanger sequencing typically is used to identify BCR-ABL1 mutations associated with tyrosine kinase inhibitor (TKI) resistance, but the method fails to detect low-level mutations that occur in less than 10%-20% of cells. Researchers used NGS to determine the impact of low-level mutations, as well as compound mutations, on the efficacy of the third generation TKI ponatinib.
Ponatinib is the most potent BCR-ABL1 TKI but is associated with considerable cardiovascular toxicity.
“The role of NGS in this setting may be to identify patients with (low level) T315I who are unlikely to derive lasting benefit from second-generation TKIs, but have a high likelihood of achieving durable cytogenetic and molecular responses to ponatinib, an important factor for balancing risks and benefits of salvage therapy selection,” wrote Dr. Michael W. Deininger, Chief of Hematology at the Huntsman Cancer Institute at the University of Utah, Salt Lake City, and his colleagues.
Patients with low-level mutations had similar response rates to those with no mutations: MCyR by one year and MMR at any time were 43% and 31%, respectively, compared with 50% and 29%. Response rates were higher in patients with compound mutations (64% and 52%) or one or more mutation (57% and 64%). The researchers speculated that the lower response rates in patients with low level or no mutations may reflect resistance mechanisms independent of BCR-ABL1.
Analysis of postbaseline samples from 127 patients (24 of whom had discontinued ponatinib for at least 1 month) determined the impact of acquired resistance. At a median follow-up of 30.1 months, emergence of previously undetected single and compound mutants during ponatinib therapy was observed in 8 patients.
The study analyzed patients from the PACE trial who had CP-CML with resistance or intolerance to dasatinib or nilotinib, or with a T315I mutation, and who were treated with ponatinib. All 267 patients had baseline mutation status determined by Sanger sequencing, with 161 mutations detected in 131 patients. NGS identified these and 105 additional mutations. Consistent with greater sensitivity of NGS, the proportion of patients with no baseline mutations by 39% by NGS vs. 51% by Sanger sequencing, and the proportion with multiple mutations was 23% vs. 10%.
The study was funded by ARIAD Pharmaceuticals. Dr. Deininger reported financial ties to ARIAD, Bristol Myers-Squibb, Novartis, Celgene, Genzyme, Gilead, Incyte, and Pfizer. Several of his coauthors reported ties to industry.
In heavily pretreated patients with chronic phase chronic myeloid leukemia (CP-CML), response to the tyrosine kinase inhibitor ponatinib did not depend on baseline mutation status, and no single or compound mutation was a major driver of primary or secondary resistance to ponatinib, according to researchers.
Irrespective of baseline mutation status, responses to ponatinib were durable. As determined by next-generation sequencing (NGS), patients with zero, one, or two or more BCR-ABL1 mutations had rates of 50%-61% for major cytogenetic response (MCyR) by 1 year and 29%-45% for major molecular response (MMR) at any time. The rates were similar to those observed with mutation status determined by Sanger sequencing. Rates of sustained response at 2 years for MCyR and MMR were 87% and 65%, respectively (Blood. 2016 Feb 11. doi: 10.1182/blood-2015-08-660977).
Sanger sequencing typically is used to identify BCR-ABL1 mutations associated with tyrosine kinase inhibitor (TKI) resistance, but the method fails to detect low-level mutations that occur in less than 10%-20% of cells. Researchers used NGS to determine the impact of low-level mutations, as well as compound mutations, on the efficacy of the third generation TKI ponatinib.
Ponatinib is the most potent BCR-ABL1 TKI but is associated with considerable cardiovascular toxicity.
“The role of NGS in this setting may be to identify patients with (low level) T315I who are unlikely to derive lasting benefit from second-generation TKIs, but have a high likelihood of achieving durable cytogenetic and molecular responses to ponatinib, an important factor for balancing risks and benefits of salvage therapy selection,” wrote Dr. Michael W. Deininger, Chief of Hematology at the Huntsman Cancer Institute at the University of Utah, Salt Lake City, and his colleagues.
Patients with low-level mutations had similar response rates to those with no mutations: MCyR by one year and MMR at any time were 43% and 31%, respectively, compared with 50% and 29%. Response rates were higher in patients with compound mutations (64% and 52%) or one or more mutation (57% and 64%). The researchers speculated that the lower response rates in patients with low level or no mutations may reflect resistance mechanisms independent of BCR-ABL1.
Analysis of postbaseline samples from 127 patients (24 of whom had discontinued ponatinib for at least 1 month) determined the impact of acquired resistance. At a median follow-up of 30.1 months, emergence of previously undetected single and compound mutants during ponatinib therapy was observed in 8 patients.
The study analyzed patients from the PACE trial who had CP-CML with resistance or intolerance to dasatinib or nilotinib, or with a T315I mutation, and who were treated with ponatinib. All 267 patients had baseline mutation status determined by Sanger sequencing, with 161 mutations detected in 131 patients. NGS identified these and 105 additional mutations. Consistent with greater sensitivity of NGS, the proportion of patients with no baseline mutations by 39% by NGS vs. 51% by Sanger sequencing, and the proportion with multiple mutations was 23% vs. 10%.
The study was funded by ARIAD Pharmaceuticals. Dr. Deininger reported financial ties to ARIAD, Bristol Myers-Squibb, Novartis, Celgene, Genzyme, Gilead, Incyte, and Pfizer. Several of his coauthors reported ties to industry.
In heavily pretreated patients with chronic phase chronic myeloid leukemia (CP-CML), response to the tyrosine kinase inhibitor ponatinib did not depend on baseline mutation status, and no single or compound mutation was a major driver of primary or secondary resistance to ponatinib, according to researchers.
Irrespective of baseline mutation status, responses to ponatinib were durable. As determined by next-generation sequencing (NGS), patients with zero, one, or two or more BCR-ABL1 mutations had rates of 50%-61% for major cytogenetic response (MCyR) by 1 year and 29%-45% for major molecular response (MMR) at any time. The rates were similar to those observed with mutation status determined by Sanger sequencing. Rates of sustained response at 2 years for MCyR and MMR were 87% and 65%, respectively (Blood. 2016 Feb 11. doi: 10.1182/blood-2015-08-660977).
Sanger sequencing typically is used to identify BCR-ABL1 mutations associated with tyrosine kinase inhibitor (TKI) resistance, but the method fails to detect low-level mutations that occur in less than 10%-20% of cells. Researchers used NGS to determine the impact of low-level mutations, as well as compound mutations, on the efficacy of the third generation TKI ponatinib.
Ponatinib is the most potent BCR-ABL1 TKI but is associated with considerable cardiovascular toxicity.
“The role of NGS in this setting may be to identify patients with (low level) T315I who are unlikely to derive lasting benefit from second-generation TKIs, but have a high likelihood of achieving durable cytogenetic and molecular responses to ponatinib, an important factor for balancing risks and benefits of salvage therapy selection,” wrote Dr. Michael W. Deininger, Chief of Hematology at the Huntsman Cancer Institute at the University of Utah, Salt Lake City, and his colleagues.
Patients with low-level mutations had similar response rates to those with no mutations: MCyR by one year and MMR at any time were 43% and 31%, respectively, compared with 50% and 29%. Response rates were higher in patients with compound mutations (64% and 52%) or one or more mutation (57% and 64%). The researchers speculated that the lower response rates in patients with low level or no mutations may reflect resistance mechanisms independent of BCR-ABL1.
Analysis of postbaseline samples from 127 patients (24 of whom had discontinued ponatinib for at least 1 month) determined the impact of acquired resistance. At a median follow-up of 30.1 months, emergence of previously undetected single and compound mutants during ponatinib therapy was observed in 8 patients.
The study analyzed patients from the PACE trial who had CP-CML with resistance or intolerance to dasatinib or nilotinib, or with a T315I mutation, and who were treated with ponatinib. All 267 patients had baseline mutation status determined by Sanger sequencing, with 161 mutations detected in 131 patients. NGS identified these and 105 additional mutations. Consistent with greater sensitivity of NGS, the proportion of patients with no baseline mutations by 39% by NGS vs. 51% by Sanger sequencing, and the proportion with multiple mutations was 23% vs. 10%.
The study was funded by ARIAD Pharmaceuticals. Dr. Deininger reported financial ties to ARIAD, Bristol Myers-Squibb, Novartis, Celgene, Genzyme, Gilead, Incyte, and Pfizer. Several of his coauthors reported ties to industry.
FROM BLOOD
Key clinical point: Baseline mutation status had little impact on ponatinib response, and no single or compound mutation was a major driver of primary or secondary resistance to ponatinib in patients with chronic phase chronic myeloid leukemia (CP-CML).
Major finding: In patients with zero, one, or two or more BCR-ABL1 mutations at baseline by next-generation sequencing were 50%-61% for major cytogenetic response (MCyR) by 1 year and 29%-45% for major molecular response (MMR) at any time; rates of sustained response at 2 years for MCyR and MMR were 87% and 65%, respectively.
Data source: From the PACE trial, 267 patients with CP-CML with resistance or intolerance to dasatinib or nilotinib, or with a T315I mutation, were treated with ponatinib.
Disclosures: The study was funded by ARIAD Pharmaceuticals. Dr. Deininger reported financial ties to ARIAD, Bristol Myers-Squibb, Novartis, Celgene, Genzyme, Gilead, Incyte, and Pfizer. Several of his coauthors reported ties to industry.
Drug granted breakthrough designation for AML
The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia. ![]()

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia. ![]()

The US Food and Drug Administration (FDA) has granted breakthrough therapy designation for midostaurin (PKC412) to treat acute myeloid leukemia (AML).
Midostaurin is a multi-targeted kinase inhibitor being developed for adults with newly diagnosed AML who are FLT3-positive, as detected by an FDA-approved test, and who are eligible to receive standard induction and consolidation chemotherapy.
Breakthrough therapy designation is intended to expedite the development and review of new medicines intended to treat serious or life-threatening conditions. The therapy must demonstrate substantial improvement over an available therapy on at least one clinically significant endpoint.
The designation includes all of the fast track program features, as well as more intensive FDA guidance on an efficient drug development program.
Phase 3 trial
The breakthrough designation for midostaurin is primarily based on the results of the phase 3 RATIFY trial, which were presented at the 2015 ASH Annual Meeting.
The trial included 717 patients with newly diagnosed, FLT3-positive AML who were younger than 60 at enrollment. All of the patients received standard induction and consolidation therapy. Roughly half also received midostaurin (n=360), while the other half received placebo (n=357).
Patients who received midostaurin experienced a significant improvement in overall survival (hazard ratio=0.77, P=0.0074). The median overall survival was 74.4 months in the midostaurin arm and 25.6 months in the placebo arm.
The median event-free survival was 8 months in the midostaurin arm and 3.6 months in the placebo arm (P=0.0032). The 5-year event-free survival was 27.5% for midostaurin and 19.3% for placebo.
There was no significant difference between the treatment arms with regard to most non-hematologic grade 3/4 adverse events. The exception was rash/desquamation, which occurred in 13% of patients in the midostaurin arm and 8% of patients in the placebo arm (P=0.02).
Other grade 3/4 non-hematologic events occurring in 10% of patients or more included, in the midostaurin and placebo arms, respectively: febrile neutropenia (81%, 82%), infection (40%, 38%), diarrhea (15%, 16%), hypokalemia (13%, 17%), pain (13%, 13%), other infection (12%, 12%), ALT/SGPT (12%, 9%), and fatigue (9%, 11%).
There were 18 deaths (5%) in the midostaurin arm and 19 (5.3%) in the placebo arm during induction and consolidation.
Midostaurin development
Novartis has opened a Global Individual Patient Program (compassionate use program) and a US Expanded Treatment Protocol (ETP) to enable midostaurin access. Patients 18 years of age and older with newly diagnosed FLT3-mutated AML who are able to receive standard induction and consolidation therapy will be considered.
To help identify patients who may have a FLT3 mutation and potentially benefit from treatment with midostaurin, Novartis is collaborating with Invivoscribe Technologies, Inc. which is leading regulatory submissions for a companion diagnostic.
Midostaurin is also being investigated for the treatment of aggressive systemic mastocytosis/mast cell leukemia. ![]()
FDA gives breakthrough status to midostaurin for AML
An experimental treatment targeting a form of acute myeloid leukemia has been designated a breakthrough therapy by the Food and Drug Administration, according to the drug’s manufacturer.
Midostaurin (Novartis) is an oral drug used alongside standard chemotherapy for adults with newly-diagnosed AML who are positive for the FMS-like tyrosine 3 (FLT-3) mutation and can undergo chemotherapy. AML has the lowest survival rate of all leukemias, and about one-third of AML patients have the FLT-3 mutation.
The FDA’s breakthrough therapy designation, in place since 2012, is an intensive form of fast-tracking in which the agency offers the manufacturer more guidance on an efficient drug development program and a higher level of organizational support, though future approval is not guaranteed. To qualify, a therapy must come with preliminary clinical evidence demonstrating substantial improvement on at least one clinically significant endpoint over available therapy, according to the agency.
Results from a phase III clinical trial, presented in December 2015 at the 57th annual meeting of the American Society of Hematology, showed that among 717 patients randomized to receive midostaurin alongside standard induction and consolidation chemotherapy or the same chemotherapy protocol alone, the midostaurin group saw significant improvement in overall survival (hazard ratio, 0.77, P = .0074).
Mean OS for patients in the midostaurin arm was 74.7 months (95% CI: 31.7, not attained), compared with 25.6 months for the placebo arm (18.6, 42.9). Median follow-up in the study was 57 months for surviving patients.
In a news release Feb. 19, Novartis said that midostaurin would be submitted for FDA approval for FLT-3-positive AML and that the company had launched compassionate use and expanded access programs allowing newly diagnosed patients aged 18 and older to receive midostaurin alongside standard induction and consolidation therapy. No targeted AML treatments are currently approved by FDA.
FLT3 is a receptor tyrosine kinase that plays a role in the proliferation in the number of certain blood cells. Midostaurin is a multi-targeted kinase inhibitor that is also being investigated for the treatment of aggressive systemic mast cell leukemia, according to Novartis.
An experimental treatment targeting a form of acute myeloid leukemia has been designated a breakthrough therapy by the Food and Drug Administration, according to the drug’s manufacturer.
Midostaurin (Novartis) is an oral drug used alongside standard chemotherapy for adults with newly-diagnosed AML who are positive for the FMS-like tyrosine 3 (FLT-3) mutation and can undergo chemotherapy. AML has the lowest survival rate of all leukemias, and about one-third of AML patients have the FLT-3 mutation.
The FDA’s breakthrough therapy designation, in place since 2012, is an intensive form of fast-tracking in which the agency offers the manufacturer more guidance on an efficient drug development program and a higher level of organizational support, though future approval is not guaranteed. To qualify, a therapy must come with preliminary clinical evidence demonstrating substantial improvement on at least one clinically significant endpoint over available therapy, according to the agency.
Results from a phase III clinical trial, presented in December 2015 at the 57th annual meeting of the American Society of Hematology, showed that among 717 patients randomized to receive midostaurin alongside standard induction and consolidation chemotherapy or the same chemotherapy protocol alone, the midostaurin group saw significant improvement in overall survival (hazard ratio, 0.77, P = .0074).
Mean OS for patients in the midostaurin arm was 74.7 months (95% CI: 31.7, not attained), compared with 25.6 months for the placebo arm (18.6, 42.9). Median follow-up in the study was 57 months for surviving patients.
In a news release Feb. 19, Novartis said that midostaurin would be submitted for FDA approval for FLT-3-positive AML and that the company had launched compassionate use and expanded access programs allowing newly diagnosed patients aged 18 and older to receive midostaurin alongside standard induction and consolidation therapy. No targeted AML treatments are currently approved by FDA.
FLT3 is a receptor tyrosine kinase that plays a role in the proliferation in the number of certain blood cells. Midostaurin is a multi-targeted kinase inhibitor that is also being investigated for the treatment of aggressive systemic mast cell leukemia, according to Novartis.
An experimental treatment targeting a form of acute myeloid leukemia has been designated a breakthrough therapy by the Food and Drug Administration, according to the drug’s manufacturer.
Midostaurin (Novartis) is an oral drug used alongside standard chemotherapy for adults with newly-diagnosed AML who are positive for the FMS-like tyrosine 3 (FLT-3) mutation and can undergo chemotherapy. AML has the lowest survival rate of all leukemias, and about one-third of AML patients have the FLT-3 mutation.
The FDA’s breakthrough therapy designation, in place since 2012, is an intensive form of fast-tracking in which the agency offers the manufacturer more guidance on an efficient drug development program and a higher level of organizational support, though future approval is not guaranteed. To qualify, a therapy must come with preliminary clinical evidence demonstrating substantial improvement on at least one clinically significant endpoint over available therapy, according to the agency.
Results from a phase III clinical trial, presented in December 2015 at the 57th annual meeting of the American Society of Hematology, showed that among 717 patients randomized to receive midostaurin alongside standard induction and consolidation chemotherapy or the same chemotherapy protocol alone, the midostaurin group saw significant improvement in overall survival (hazard ratio, 0.77, P = .0074).
Mean OS for patients in the midostaurin arm was 74.7 months (95% CI: 31.7, not attained), compared with 25.6 months for the placebo arm (18.6, 42.9). Median follow-up in the study was 57 months for surviving patients.
In a news release Feb. 19, Novartis said that midostaurin would be submitted for FDA approval for FLT-3-positive AML and that the company had launched compassionate use and expanded access programs allowing newly diagnosed patients aged 18 and older to receive midostaurin alongside standard induction and consolidation therapy. No targeted AML treatments are currently approved by FDA.
FLT3 is a receptor tyrosine kinase that plays a role in the proliferation in the number of certain blood cells. Midostaurin is a multi-targeted kinase inhibitor that is also being investigated for the treatment of aggressive systemic mast cell leukemia, according to Novartis.
Targeting EZH2 to treat ETP-ALL

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()

The gene EZH2 is a driver of, and potential therapeutic target for, early T-cell precursor acute lymphoblastic leukemia (ETP-ALL), according to a new study.
A previous study, published in Nature in 2012, suggested that nearly half of ETP-ALLs have inactivating alterations in EZH2.
Loss of EZH2 function can inactivate Polycomb repressive complex 2 (PRC2), but it was not clear how PRC2 loss-of-function mutations would aid leukemia growth.
The new study, published in Cell Reports, provides some insight.
Tobias Neff, MD, of the University of Colorado Denver in Aurora, and his colleagues developed a mouse model of NRASQ61K-driven leukemia that recapitulates phenotypic and transcriptional features of ETP-ALL.
Experiments with this model revealed that inactivation of EZH2 helps accelerate leukemia development and enhances a stem-cell-related transcriptional program.
“We have 2 major features of [ETP-ALL]—stem-like cells and increased growth—and, now, we show an actor implicated in both—namely, EZH2/PRC2,” Dr Neff said.
“How exactly the stem-cell-like gene expression profile contributes to the aggressiveness of ETP-ALL is unknown, but we’ve known that these stem-like cells are associated with poor prognosis in acute leukemia.”
The researchers also found that EZH2 inactivation resulted in increased activation of STAT3 by tyrosine 705 phosphorylation. This led them to wonder whether the JAK/STAT pathway might be important in their ETP-ALL model.
The team tested the JAK1/2 inhibitor ruxolitinib in NRASQ61K cells with EZH2 deletion and observed inhibition of cell growth.
“Ruxolitinib is unlikely to treat the disease by itself, but this model will help us test possible drug combinations that could eventually benefit ETP-ALL patients,” Dr Neff said.
He and his colleagues also plan to test the activity of different drugs against other cell types with inactivated EZH2.
“In addition to our specific finding in this disease, we are excited to now have a model that allows us to explore consequences of EZH2 inactivation that may enrich our understanding of a number of other conditions with a similar set of genetic changes,” Dr Neff said.
He and his colleagues noted that EZH2 is known to be inactivated in myelodysplastic syndromes, myeloproliferative neoplasms, and other hematologic malignancies. ![]()
Risk of reproductive problems in male cancer survivors

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()

A study of Norwegian men has revealed several factors that may help predict reproductive problems among males diagnosed with cancer before age 25.
Cancer type, age at diagnosis, and time period of diagnosis were all associated with the likelihood of paternity.
And although cancer survivors were less likely to reproduce and more likely to use assisted reproductive technology, their first offspring were no less healthy than the offspring of control subjects.
This research was published in the British Journal of Cancer.
The study began with all Norwegian men born between 1965 and 1985 (n=626,495). The researchers excluded men who emigrated or died before reaching fertile age, which left 2687 men who were diagnosed with cancer before age 25 and 607,668 cancer-free controls.
The most common cancers were testicular cancer (27%), CNS tumors (18%), lymphoma (15%), and leukemia (13%). Thirty percent of the cancer cases were diagnosed in childhood (0–14 years of age), 26% in adolescence (15–19 years), and 43% in young adulthood (20–24 years).
Nine percent (n=247) of cancer cases were diagnosed from 1965 through 1979, 50% (n=1346) from 1980 through 1994, and 41% (n=1094) from 1995 through 2007.
The cancer survivors were less likely to have children than controls, with a hazard ratio (HR) of 0.72.
The reduction in paternity was significant for survivors of leukemia (HR=0.78), lymphoma (HR=0.78), testicular cancer (HR=0.77), CNS tumors (HR=0.45), bone tumors (HR=0.69), sympathetic nervous system tumors (HR=0.50), and retinoblastoma (HR=0.52).
The reduction in paternity was also more pronounced for cancer patients diagnosed before 1995. The HR was 0.61 for those diagnosed from 1965 through 1979 and 0.66 for those diagnosed from 1980 through 1994.
Patients who were diagnosed before age 15 were less likely to reproduce as well, with an HR of 0.59.
“These finds are important for male cancer survivors, seeing as we can identify groups at risk of having reproduction problems,” said study author Maria Winther Gunnes, a PhD candidate at the University of Bergen in Norway.
Another finding was that male cancer survivors were more likely than controls to have pregnancies resulting from assisted reproductive technology. The relative risk was 3.32.
When assessed by cancer type, the relative risk was 2.29 for leukemia, 3.79 for lymphoma, 2.41 for CNS tumors, 5.71 for sympathetic nervous system tumors, 2.20 for renal tumors, 4.77 for bone tumors, 1.32 for soft tissue sarcomas, 3.70 for testicular cancer, 4.36 for thyroid carcinoma, and 0.45 for malignant melanoma.
There was no increased risk among the first offspring of cancer survivors for perinatal death, congenital malformations, being small for gestational age, low birth weight, or preterm birth.
“It is important to be able to assure young male cancer survivors that their illness and treatment will not have a negative impact on their own children,” Gunnes said. ![]()
Pinpointing the cells that cause CML relapse

Image by Difu Wu
Preclinical research suggests chronic myeloid leukemia (CML) patients have a heterogeneous population of long-term hematopoietic stem cells (LTHSCs)—some that can initiate leukemia and some that cannot.
Researchers found they could identify the leukemia-initiating cells by measuring expression of the thrombopoietin receptor MPL. Cells with high MPL expression could initiate CML in mice.
The team said these results suggest the leukemic LTHSCs are the cells responsible for relapse in CML.
“This shows that not all leukemia stem cells are equal,” said study author Ravi Bhatia, MD, of the University of Alabama Birmingham.
“Some are more prone to causing leukemia and relapses, while some others may just hang around without potential for contributing to relapse.”
Dr Bhatia and his colleagues reported these findings in The Journal of Clinical Investigation.
In their experiments, the team used an inducible transgenic mouse model of CML, where the BCR-ABL gene fusion is under the control of a tetracycline-regulated enhancer. This model creates a chronic myeloproliferative disorder that resembles chronic phase CML.
Previous work had shown that only cells with an LTHSC phenotype were capable of long-term repopulation and leukemia-initiating capacity after transplantation to another mouse.
When the researchers transplanted LTHSCs from CML-model mice to other mice, 11 of 20 recipients developed CML, and 9 of 20 showed engraftment with CML cells but did not develop the leukocytosis characteristic of leukemia.
When the LTHSCs from the primary-recipient mice were transferred to secondary recipients, 7 of 17 mice receiving cells from leukemic mice developed CML, and none of the secondary-recipient mice receiving cells from the non-leukemic mice developed CML.
The researchers tested these 2 groups of LTHSCs for differences in gene expression. They found significant differences between the leukemic and non-leukemic LTHSCs for the genes Mpl, c-Myc, CD47, Pten, Sirt1, Ptch1, and Tie2.
The team then decided to focus on Mpl. They used flow cytometry to select LTHSCs with either high or low Mpl expression from CML-model BCR-ABL mice.
Seven of 16 mice receiving Mpl-Hi LTHSCs developed leukemia after transplantation, compared with 1 of 17 receiving Mpl-Lo LTHSCs. This suggested an increased leukemogenic capacity for the Mpl-Hi LTHSCs.
The researchers also investigated the impact of cell-cycle status. They found that CML Mpl-Hi LTHSCs that were in a resting stage of the cell cycle had enhanced long-term engraftment and leukemogenic capacity compared with cycling Mpl-Hi LTHSCs.
The team used virus vectors and shRNA to create Mpl knockdown BCR-ABL LTHSCs and showed that the knockdown cells had a greatly reduced ability to produce leukemia in recipient mice.
The Mpl knockdown cells, after stimulation by the Mpl ligand thrombopoietin, also had reduced expression of the activated transcription factors p-STAT3 and p-STAT5, compared with controls.
Finally, the researchers examined human CML cells for differences between MPL-Hi LTHSCs and MPL-Lo LTHSCs. The results were similar to those observed in mice.
The human MPL-Hi LTHSCs had a higher rate of engraftment than the human MPL-Lo LTHSCs, as tested in a xenograft model using immunodeficient mice.
Additionally, the human MPL-Hi LTHSCs had reduced sensitivity to nilotinib compared with MPL-Lo LTHSCs. However, a Jak/STAT inhibitor significantly reduced cell growth and increased apoptosis in human MPL-Hi LTHSCs.
The researchers concluded that MPL expression is a marker and key regulator of leukemogenic potential and drug sensitivity of CML LTHSCs. They said their findings support further investigation of approaches to antagonize MPL signaling as a potential therapeutic strategy to eliminate leukemia-initiating LTHSCs. ![]()

Image by Difu Wu
Preclinical research suggests chronic myeloid leukemia (CML) patients have a heterogeneous population of long-term hematopoietic stem cells (LTHSCs)—some that can initiate leukemia and some that cannot.
Researchers found they could identify the leukemia-initiating cells by measuring expression of the thrombopoietin receptor MPL. Cells with high MPL expression could initiate CML in mice.
The team said these results suggest the leukemic LTHSCs are the cells responsible for relapse in CML.
“This shows that not all leukemia stem cells are equal,” said study author Ravi Bhatia, MD, of the University of Alabama Birmingham.
“Some are more prone to causing leukemia and relapses, while some others may just hang around without potential for contributing to relapse.”
Dr Bhatia and his colleagues reported these findings in The Journal of Clinical Investigation.
In their experiments, the team used an inducible transgenic mouse model of CML, where the BCR-ABL gene fusion is under the control of a tetracycline-regulated enhancer. This model creates a chronic myeloproliferative disorder that resembles chronic phase CML.
Previous work had shown that only cells with an LTHSC phenotype were capable of long-term repopulation and leukemia-initiating capacity after transplantation to another mouse.
When the researchers transplanted LTHSCs from CML-model mice to other mice, 11 of 20 recipients developed CML, and 9 of 20 showed engraftment with CML cells but did not develop the leukocytosis characteristic of leukemia.
When the LTHSCs from the primary-recipient mice were transferred to secondary recipients, 7 of 17 mice receiving cells from leukemic mice developed CML, and none of the secondary-recipient mice receiving cells from the non-leukemic mice developed CML.
The researchers tested these 2 groups of LTHSCs for differences in gene expression. They found significant differences between the leukemic and non-leukemic LTHSCs for the genes Mpl, c-Myc, CD47, Pten, Sirt1, Ptch1, and Tie2.
The team then decided to focus on Mpl. They used flow cytometry to select LTHSCs with either high or low Mpl expression from CML-model BCR-ABL mice.
Seven of 16 mice receiving Mpl-Hi LTHSCs developed leukemia after transplantation, compared with 1 of 17 receiving Mpl-Lo LTHSCs. This suggested an increased leukemogenic capacity for the Mpl-Hi LTHSCs.
The researchers also investigated the impact of cell-cycle status. They found that CML Mpl-Hi LTHSCs that were in a resting stage of the cell cycle had enhanced long-term engraftment and leukemogenic capacity compared with cycling Mpl-Hi LTHSCs.
The team used virus vectors and shRNA to create Mpl knockdown BCR-ABL LTHSCs and showed that the knockdown cells had a greatly reduced ability to produce leukemia in recipient mice.
The Mpl knockdown cells, after stimulation by the Mpl ligand thrombopoietin, also had reduced expression of the activated transcription factors p-STAT3 and p-STAT5, compared with controls.
Finally, the researchers examined human CML cells for differences between MPL-Hi LTHSCs and MPL-Lo LTHSCs. The results were similar to those observed in mice.
The human MPL-Hi LTHSCs had a higher rate of engraftment than the human MPL-Lo LTHSCs, as tested in a xenograft model using immunodeficient mice.
Additionally, the human MPL-Hi LTHSCs had reduced sensitivity to nilotinib compared with MPL-Lo LTHSCs. However, a Jak/STAT inhibitor significantly reduced cell growth and increased apoptosis in human MPL-Hi LTHSCs.
The researchers concluded that MPL expression is a marker and key regulator of leukemogenic potential and drug sensitivity of CML LTHSCs. They said their findings support further investigation of approaches to antagonize MPL signaling as a potential therapeutic strategy to eliminate leukemia-initiating LTHSCs. ![]()

Image by Difu Wu
Preclinical research suggests chronic myeloid leukemia (CML) patients have a heterogeneous population of long-term hematopoietic stem cells (LTHSCs)—some that can initiate leukemia and some that cannot.
Researchers found they could identify the leukemia-initiating cells by measuring expression of the thrombopoietin receptor MPL. Cells with high MPL expression could initiate CML in mice.
The team said these results suggest the leukemic LTHSCs are the cells responsible for relapse in CML.
“This shows that not all leukemia stem cells are equal,” said study author Ravi Bhatia, MD, of the University of Alabama Birmingham.
“Some are more prone to causing leukemia and relapses, while some others may just hang around without potential for contributing to relapse.”
Dr Bhatia and his colleagues reported these findings in The Journal of Clinical Investigation.
In their experiments, the team used an inducible transgenic mouse model of CML, where the BCR-ABL gene fusion is under the control of a tetracycline-regulated enhancer. This model creates a chronic myeloproliferative disorder that resembles chronic phase CML.
Previous work had shown that only cells with an LTHSC phenotype were capable of long-term repopulation and leukemia-initiating capacity after transplantation to another mouse.
When the researchers transplanted LTHSCs from CML-model mice to other mice, 11 of 20 recipients developed CML, and 9 of 20 showed engraftment with CML cells but did not develop the leukocytosis characteristic of leukemia.
When the LTHSCs from the primary-recipient mice were transferred to secondary recipients, 7 of 17 mice receiving cells from leukemic mice developed CML, and none of the secondary-recipient mice receiving cells from the non-leukemic mice developed CML.
The researchers tested these 2 groups of LTHSCs for differences in gene expression. They found significant differences between the leukemic and non-leukemic LTHSCs for the genes Mpl, c-Myc, CD47, Pten, Sirt1, Ptch1, and Tie2.
The team then decided to focus on Mpl. They used flow cytometry to select LTHSCs with either high or low Mpl expression from CML-model BCR-ABL mice.
Seven of 16 mice receiving Mpl-Hi LTHSCs developed leukemia after transplantation, compared with 1 of 17 receiving Mpl-Lo LTHSCs. This suggested an increased leukemogenic capacity for the Mpl-Hi LTHSCs.
The researchers also investigated the impact of cell-cycle status. They found that CML Mpl-Hi LTHSCs that were in a resting stage of the cell cycle had enhanced long-term engraftment and leukemogenic capacity compared with cycling Mpl-Hi LTHSCs.
The team used virus vectors and shRNA to create Mpl knockdown BCR-ABL LTHSCs and showed that the knockdown cells had a greatly reduced ability to produce leukemia in recipient mice.
The Mpl knockdown cells, after stimulation by the Mpl ligand thrombopoietin, also had reduced expression of the activated transcription factors p-STAT3 and p-STAT5, compared with controls.
Finally, the researchers examined human CML cells for differences between MPL-Hi LTHSCs and MPL-Lo LTHSCs. The results were similar to those observed in mice.
The human MPL-Hi LTHSCs had a higher rate of engraftment than the human MPL-Lo LTHSCs, as tested in a xenograft model using immunodeficient mice.
Additionally, the human MPL-Hi LTHSCs had reduced sensitivity to nilotinib compared with MPL-Lo LTHSCs. However, a Jak/STAT inhibitor significantly reduced cell growth and increased apoptosis in human MPL-Hi LTHSCs.
The researchers concluded that MPL expression is a marker and key regulator of leukemogenic potential and drug sensitivity of CML LTHSCs. They said their findings support further investigation of approaches to antagonize MPL signaling as a potential therapeutic strategy to eliminate leukemia-initiating LTHSCs. ![]()
Drug shows promise for treating resistant AML, MCL

Preclinical research suggests the investigational anticancer drug ONC201 can be effective against mantle cell lymphoma (MCL) and acute myeloid leukemia (AML).
ONC201 induced p53-independent apoptosis in AML and MCL cell lines and in samples from patients with either disease.
Investigators noted that p53 dysfunction occurs in more than half of malignancies and can promote resistance to standard chemotherapy.
“The clinical challenge posed by p53 abnormalities in blood malignancies is that therapeutic strategies other than standard chemotherapies are required,” said Michael Andreeff, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“We found that ONC201 caused p53-independent cell death and cell cycle arrest in cell lines and in lymphoma and acute leukemia patient samples.”
Dr Andreeff and his colleagues reported these findings in Science Signaling. Some of the investigators involved in this research are affiliated with Oncoceutics Inc., the company developing ONC201.
Dr Andreeff and his colleagues assessed the effects of ONC201 against AML and MCL, in both cultured cell lines and primary cells bearing either wild-type or mutant p53.
The patient samples included those that demonstrated genetic abnormalities linked to poor prognosis (FLT3 mutations, TP53 mutations) or resistance to ibrutinib. The team also tested ONC201 in a bortezomib-resistant myeloma cell line.
The experiments showed that ONC201 exerted anticancer activity regardless of p53 status, FLT3 mutations, or drug resistance. ONC201 proved active in the bortezomib-resistant myeloma cell line and in ibrutinib-resistant samples from MCL patients.
Experiments in mice showed that ONC201 caused cell death in AML and leukemia stem cells while sparing normal bone marrow cells.
And the investigators found that combining ONC201 with the BCL-2 antagonist venetoclax (ABT-199) synergistically increased apoptosis.
Further investigation revealed that ONC201 increased translation of the stress-induced protein ATF4 through stress signals similar to those caused by unfolded protein response (UPR) and integrated stress response (ISR).
“This increase in ATF4 in ONC201-treated hematopoietic cells promoted cell death,” Dr Andreeff explained. “However, unlike with UPR and ISR, the increase in ATF4 in ONC201-treated cells was not regulated by standard molecular signaling, indicating a novel mechanism of stressing cancer cells to death regardless of p53 status.”
The investigators noted that the mechanisms of ONC201 identified in solid tumors—namely, induction of TRAIL and DR5—were not operational in leukemia and lymphoma.
A study of ONC201 in solid tumors and multiple myeloma was published alongside this study in Science Signaling.
“There is clear evidence that ONC201 has clinical potential in hematological malignancies,” Dr Andreeff noted. “Clinical trials in leukemia and lymphoma patients have recently been initiated at MD Anderson.” ![]()

Preclinical research suggests the investigational anticancer drug ONC201 can be effective against mantle cell lymphoma (MCL) and acute myeloid leukemia (AML).
ONC201 induced p53-independent apoptosis in AML and MCL cell lines and in samples from patients with either disease.
Investigators noted that p53 dysfunction occurs in more than half of malignancies and can promote resistance to standard chemotherapy.
“The clinical challenge posed by p53 abnormalities in blood malignancies is that therapeutic strategies other than standard chemotherapies are required,” said Michael Andreeff, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“We found that ONC201 caused p53-independent cell death and cell cycle arrest in cell lines and in lymphoma and acute leukemia patient samples.”
Dr Andreeff and his colleagues reported these findings in Science Signaling. Some of the investigators involved in this research are affiliated with Oncoceutics Inc., the company developing ONC201.
Dr Andreeff and his colleagues assessed the effects of ONC201 against AML and MCL, in both cultured cell lines and primary cells bearing either wild-type or mutant p53.
The patient samples included those that demonstrated genetic abnormalities linked to poor prognosis (FLT3 mutations, TP53 mutations) or resistance to ibrutinib. The team also tested ONC201 in a bortezomib-resistant myeloma cell line.
The experiments showed that ONC201 exerted anticancer activity regardless of p53 status, FLT3 mutations, or drug resistance. ONC201 proved active in the bortezomib-resistant myeloma cell line and in ibrutinib-resistant samples from MCL patients.
Experiments in mice showed that ONC201 caused cell death in AML and leukemia stem cells while sparing normal bone marrow cells.
And the investigators found that combining ONC201 with the BCL-2 antagonist venetoclax (ABT-199) synergistically increased apoptosis.
Further investigation revealed that ONC201 increased translation of the stress-induced protein ATF4 through stress signals similar to those caused by unfolded protein response (UPR) and integrated stress response (ISR).
“This increase in ATF4 in ONC201-treated hematopoietic cells promoted cell death,” Dr Andreeff explained. “However, unlike with UPR and ISR, the increase in ATF4 in ONC201-treated cells was not regulated by standard molecular signaling, indicating a novel mechanism of stressing cancer cells to death regardless of p53 status.”
The investigators noted that the mechanisms of ONC201 identified in solid tumors—namely, induction of TRAIL and DR5—were not operational in leukemia and lymphoma.
A study of ONC201 in solid tumors and multiple myeloma was published alongside this study in Science Signaling.
“There is clear evidence that ONC201 has clinical potential in hematological malignancies,” Dr Andreeff noted. “Clinical trials in leukemia and lymphoma patients have recently been initiated at MD Anderson.” ![]()

Preclinical research suggests the investigational anticancer drug ONC201 can be effective against mantle cell lymphoma (MCL) and acute myeloid leukemia (AML).
ONC201 induced p53-independent apoptosis in AML and MCL cell lines and in samples from patients with either disease.
Investigators noted that p53 dysfunction occurs in more than half of malignancies and can promote resistance to standard chemotherapy.
“The clinical challenge posed by p53 abnormalities in blood malignancies is that therapeutic strategies other than standard chemotherapies are required,” said Michael Andreeff, MD, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“We found that ONC201 caused p53-independent cell death and cell cycle arrest in cell lines and in lymphoma and acute leukemia patient samples.”
Dr Andreeff and his colleagues reported these findings in Science Signaling. Some of the investigators involved in this research are affiliated with Oncoceutics Inc., the company developing ONC201.
Dr Andreeff and his colleagues assessed the effects of ONC201 against AML and MCL, in both cultured cell lines and primary cells bearing either wild-type or mutant p53.
The patient samples included those that demonstrated genetic abnormalities linked to poor prognosis (FLT3 mutations, TP53 mutations) or resistance to ibrutinib. The team also tested ONC201 in a bortezomib-resistant myeloma cell line.
The experiments showed that ONC201 exerted anticancer activity regardless of p53 status, FLT3 mutations, or drug resistance. ONC201 proved active in the bortezomib-resistant myeloma cell line and in ibrutinib-resistant samples from MCL patients.
Experiments in mice showed that ONC201 caused cell death in AML and leukemia stem cells while sparing normal bone marrow cells.
And the investigators found that combining ONC201 with the BCL-2 antagonist venetoclax (ABT-199) synergistically increased apoptosis.
Further investigation revealed that ONC201 increased translation of the stress-induced protein ATF4 through stress signals similar to those caused by unfolded protein response (UPR) and integrated stress response (ISR).
“This increase in ATF4 in ONC201-treated hematopoietic cells promoted cell death,” Dr Andreeff explained. “However, unlike with UPR and ISR, the increase in ATF4 in ONC201-treated cells was not regulated by standard molecular signaling, indicating a novel mechanism of stressing cancer cells to death regardless of p53 status.”
The investigators noted that the mechanisms of ONC201 identified in solid tumors—namely, induction of TRAIL and DR5—were not operational in leukemia and lymphoma.
A study of ONC201 in solid tumors and multiple myeloma was published alongside this study in Science Signaling.
“There is clear evidence that ONC201 has clinical potential in hematological malignancies,” Dr Andreeff noted. “Clinical trials in leukemia and lymphoma patients have recently been initiated at MD Anderson.” ![]()
Gene variants linked to drug intolerance

Photo courtesy of the CDC
New research has revealed inherited genetic variations that may predispose patients to severe toxicity from thiopurines, a class of medications used as anticancer and immunosuppressive drugs.
Investigators identified 4 variations in the NUDT15 gene that alter thiopurine metabolism, leaving patients particularly sensitive to the drugs and at risk for toxicity.
One in 3 Japanese patients in this study carried the variations.
And evidence suggests the variations are common in other populations across Asia and in individuals of Hispanic ethnicity.
Jun J. Yang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these findings in Nature Genetics.
In 2015, Dr Yang and his colleagues published evidence linking a NUDT15 variant to reduced tolerance of mercaptopurine and reported the variant was more common in patients of East Asian ancestry.
With the current study, the investigators identified 3 additional NUDT15 variants and found that all 4 variants—p.Arg139Cys, p.Arg139His, p.Val18Ile, and p.Val18_Val19insGlyVal—were associated with lower levels of enzymatic activity and imbalance of thiopurine metabolism.
In a group of 270 children with acute lymphoblastic leukemia (ALL), the variants caused a 74.4% to 100% loss of NUDT15 function. They also predicted enzyme activity and mercaptopurine tolerance. In Singapore and Japan, for example, patients with the 2 highest risk variants had the lowest level of enzyme activity.
“These patients had excessive levels of the active drug metabolites per mercaptopurine dose, which suggests we may reduce the drug dose to achieve the level necessary to kill leukemia cells without causing toxicity,” Dr Yang said, adding that the NUDT15 variants have no other known health consequences.
The investigators also checked leukemic cells from 285 children newly diagnosed with ALL and found that patients with NUDT15 variants were more sensitive to thiopurines.
“That suggests we can screen for NUDT15 variants and potentially plan mercaptopurine doses according to each patient’s genotype before the therapy starts,” Dr Yang said. “This way, we hope to avoid toxicity without compromising treatment effectiveness.”
The investigators noted that future studies are needed to determine optimal thiopurine doses for patients with different NUDT15 variants.
Meanwhile, the search continues for variants in NUDT15 or other genes that influence chemotherapy effectiveness and safety. The NUDT15 variants and previously identified TPMT variants could not fully explain why Guatemalan patients in this study tolerated the lowest doses of mercaptopurine. ![]()

Photo courtesy of the CDC
New research has revealed inherited genetic variations that may predispose patients to severe toxicity from thiopurines, a class of medications used as anticancer and immunosuppressive drugs.
Investigators identified 4 variations in the NUDT15 gene that alter thiopurine metabolism, leaving patients particularly sensitive to the drugs and at risk for toxicity.
One in 3 Japanese patients in this study carried the variations.
And evidence suggests the variations are common in other populations across Asia and in individuals of Hispanic ethnicity.
Jun J. Yang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these findings in Nature Genetics.
In 2015, Dr Yang and his colleagues published evidence linking a NUDT15 variant to reduced tolerance of mercaptopurine and reported the variant was more common in patients of East Asian ancestry.
With the current study, the investigators identified 3 additional NUDT15 variants and found that all 4 variants—p.Arg139Cys, p.Arg139His, p.Val18Ile, and p.Val18_Val19insGlyVal—were associated with lower levels of enzymatic activity and imbalance of thiopurine metabolism.
In a group of 270 children with acute lymphoblastic leukemia (ALL), the variants caused a 74.4% to 100% loss of NUDT15 function. They also predicted enzyme activity and mercaptopurine tolerance. In Singapore and Japan, for example, patients with the 2 highest risk variants had the lowest level of enzyme activity.
“These patients had excessive levels of the active drug metabolites per mercaptopurine dose, which suggests we may reduce the drug dose to achieve the level necessary to kill leukemia cells without causing toxicity,” Dr Yang said, adding that the NUDT15 variants have no other known health consequences.
The investigators also checked leukemic cells from 285 children newly diagnosed with ALL and found that patients with NUDT15 variants were more sensitive to thiopurines.
“That suggests we can screen for NUDT15 variants and potentially plan mercaptopurine doses according to each patient’s genotype before the therapy starts,” Dr Yang said. “This way, we hope to avoid toxicity without compromising treatment effectiveness.”
The investigators noted that future studies are needed to determine optimal thiopurine doses for patients with different NUDT15 variants.
Meanwhile, the search continues for variants in NUDT15 or other genes that influence chemotherapy effectiveness and safety. The NUDT15 variants and previously identified TPMT variants could not fully explain why Guatemalan patients in this study tolerated the lowest doses of mercaptopurine. ![]()

Photo courtesy of the CDC
New research has revealed inherited genetic variations that may predispose patients to severe toxicity from thiopurines, a class of medications used as anticancer and immunosuppressive drugs.
Investigators identified 4 variations in the NUDT15 gene that alter thiopurine metabolism, leaving patients particularly sensitive to the drugs and at risk for toxicity.
One in 3 Japanese patients in this study carried the variations.
And evidence suggests the variations are common in other populations across Asia and in individuals of Hispanic ethnicity.
Jun J. Yang, PhD, of St. Jude Children’s Research Hospital in Memphis, Tennessee, and his colleagues reported these findings in Nature Genetics.
In 2015, Dr Yang and his colleagues published evidence linking a NUDT15 variant to reduced tolerance of mercaptopurine and reported the variant was more common in patients of East Asian ancestry.
With the current study, the investigators identified 3 additional NUDT15 variants and found that all 4 variants—p.Arg139Cys, p.Arg139His, p.Val18Ile, and p.Val18_Val19insGlyVal—were associated with lower levels of enzymatic activity and imbalance of thiopurine metabolism.
In a group of 270 children with acute lymphoblastic leukemia (ALL), the variants caused a 74.4% to 100% loss of NUDT15 function. They also predicted enzyme activity and mercaptopurine tolerance. In Singapore and Japan, for example, patients with the 2 highest risk variants had the lowest level of enzyme activity.
“These patients had excessive levels of the active drug metabolites per mercaptopurine dose, which suggests we may reduce the drug dose to achieve the level necessary to kill leukemia cells without causing toxicity,” Dr Yang said, adding that the NUDT15 variants have no other known health consequences.
The investigators also checked leukemic cells from 285 children newly diagnosed with ALL and found that patients with NUDT15 variants were more sensitive to thiopurines.
“That suggests we can screen for NUDT15 variants and potentially plan mercaptopurine doses according to each patient’s genotype before the therapy starts,” Dr Yang said. “This way, we hope to avoid toxicity without compromising treatment effectiveness.”
The investigators noted that future studies are needed to determine optimal thiopurine doses for patients with different NUDT15 variants.
Meanwhile, the search continues for variants in NUDT15 or other genes that influence chemotherapy effectiveness and safety. The NUDT15 variants and previously identified TPMT variants could not fully explain why Guatemalan patients in this study tolerated the lowest doses of mercaptopurine. ![]()