User login
Inhibitor exhibits activity against resistant AML

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()

Preclinical research indicates that a novel inhibitor can overcome resistance-conferring FLT3 mutations in acute myeloid leukemia (AML).
The MERTK/FLT3 inhibitor MRX-2843 induced apoptosis and inhibited colony formation in AML cell lines and primary patient samples expressing MERTK and/or FLT3-ITD.
MRX-2843 also improved survival in mouse models of AML, including cases where tumors were resistant to the FLT3 inhibitor quizartinib.
Douglas Graham, MD, PhD, of the University of Colorado in Aurora, and his colleagues conducted this research and reported the results in JCI Insight.
The researchers previously showed that the receptor tyrosine kinase MERTK is overexpressed in 80% to 90% of AMLs and contributes to leukemogenesis.
With the current study, they showed that MRX-2843 abrogates activation of MERTK, FLT3, and their downstream effectors. And this translates to antileukemic activity in vitro and in vivo.
MRX-2843 inhibited MERTK signaling, induced cell death, and abolished oncogenic phenotypes in AML cells. The drug also exhibited therapeutic activity in a MERTK-dependent xenograft model.
MRX-2843 was able to stop the activation of FLT3 and its signaling pathways almost completely. The researchers said this suggests the drug has somewhat higher cellular potency against FLT3 relative to MERTK.
In mouse models of FLT3-ITD AML, MRX-2843 significantly prolonged survival when compared to vehicle control.
The researchers also said MRX-2843 selectively inhibited colony formation in primary AML patient samples. Primary human MERTK-expressing leukemic blasts, with or without FLT3-ITD mutations, proved sensitive to treatment with MRX-2843.
In addition, MRX-2843 increased survival and decreased peripheral disease burden in patient-derived xenograft models of AML—both MERTK+FLT3-WT and MERTK+FLT3-ITD models.
Finally, the researchers found that MRX-2843 was active against quizartinib-resistant FLT3-mutant proteins, induced apoptosis and inhibited colony formation in quizartinib-resistant FLT3-ITD cell lines, and prolonged survival in quizartinib-resistant FLT3-ITD xenograft models. ![]()
Financial burdens reduce QOL for cancer survivors

receiving treatment
Photo by Rhoda Baer
An analysis of nearly 20 million cancer survivors showed that almost 29% had financial burdens as a result of their cancer diagnosis and/or treatment.
In other words, they borrowed money, declared bankruptcy, worried about paying large medical bills, were unable to cover the cost of medical visits, or made other financial sacrifices.
Furthermore, such hardships could have lasting effects on a cancer survivor’s quality of life (QOL).
Hrishikesh Kale and Norman Carroll, PhD, both of Virginia Commonwealth University School of Pharmacy in Richmond, reported these findings in Cancer.
The pair analyzed 2011 Medical Expenditure Panel Survey data on 19.6 million cancer survivors, assessing financial burden and QOL.
Subjects were considered to have financial burden if they reported 1 of the following problems: borrowed money/declared bankruptcy, worried about paying large medical bills, unable to cover the cost of medical care visits, or other financial sacrifices.
Nearly 29% of the cancer survivors reported at least 1 financial problem resulting from cancer diagnosis, treatment, or lasting effects of that treatment.
Of all the cancer survivors in the analysis, 20.9% worried about paying large medical bills, 11.5% were unable to cover the cost of medical care visits, 7.6% reported borrowing money or going into debt, 1.4% declared bankruptcy, and 8.6% reported other financial sacrifices.
Cancer survivors who faced such financial difficulties had lower physical and mental health-related QOL, higher risk for depressed mood and psychological distress, and were more likely to worry about cancer recurrence, when compared with cancer survivors who did not face financial problems.
In addition, as the number of financial problems reported by cancer survivors increased, their QOL continued to decrease. And their risk for depressed mood, psychological distress, and worries about cancer recurrence continued to increase.
“Our results suggest that policies and practices that minimize cancer patients’ out-of-pocket costs can improve survivors’ health-related quality of life and psychological health,” Dr Carroll said.
“Reducing the financial burden of cancer care requires integrated efforts, and the study findings are useful for survivorship care programs, oncologists, payers, pharmaceutical companies, and patients and their family members.” ![]()

receiving treatment
Photo by Rhoda Baer
An analysis of nearly 20 million cancer survivors showed that almost 29% had financial burdens as a result of their cancer diagnosis and/or treatment.
In other words, they borrowed money, declared bankruptcy, worried about paying large medical bills, were unable to cover the cost of medical visits, or made other financial sacrifices.
Furthermore, such hardships could have lasting effects on a cancer survivor’s quality of life (QOL).
Hrishikesh Kale and Norman Carroll, PhD, both of Virginia Commonwealth University School of Pharmacy in Richmond, reported these findings in Cancer.
The pair analyzed 2011 Medical Expenditure Panel Survey data on 19.6 million cancer survivors, assessing financial burden and QOL.
Subjects were considered to have financial burden if they reported 1 of the following problems: borrowed money/declared bankruptcy, worried about paying large medical bills, unable to cover the cost of medical care visits, or other financial sacrifices.
Nearly 29% of the cancer survivors reported at least 1 financial problem resulting from cancer diagnosis, treatment, or lasting effects of that treatment.
Of all the cancer survivors in the analysis, 20.9% worried about paying large medical bills, 11.5% were unable to cover the cost of medical care visits, 7.6% reported borrowing money or going into debt, 1.4% declared bankruptcy, and 8.6% reported other financial sacrifices.
Cancer survivors who faced such financial difficulties had lower physical and mental health-related QOL, higher risk for depressed mood and psychological distress, and were more likely to worry about cancer recurrence, when compared with cancer survivors who did not face financial problems.
In addition, as the number of financial problems reported by cancer survivors increased, their QOL continued to decrease. And their risk for depressed mood, psychological distress, and worries about cancer recurrence continued to increase.
“Our results suggest that policies and practices that minimize cancer patients’ out-of-pocket costs can improve survivors’ health-related quality of life and psychological health,” Dr Carroll said.
“Reducing the financial burden of cancer care requires integrated efforts, and the study findings are useful for survivorship care programs, oncologists, payers, pharmaceutical companies, and patients and their family members.” ![]()

receiving treatment
Photo by Rhoda Baer
An analysis of nearly 20 million cancer survivors showed that almost 29% had financial burdens as a result of their cancer diagnosis and/or treatment.
In other words, they borrowed money, declared bankruptcy, worried about paying large medical bills, were unable to cover the cost of medical visits, or made other financial sacrifices.
Furthermore, such hardships could have lasting effects on a cancer survivor’s quality of life (QOL).
Hrishikesh Kale and Norman Carroll, PhD, both of Virginia Commonwealth University School of Pharmacy in Richmond, reported these findings in Cancer.
The pair analyzed 2011 Medical Expenditure Panel Survey data on 19.6 million cancer survivors, assessing financial burden and QOL.
Subjects were considered to have financial burden if they reported 1 of the following problems: borrowed money/declared bankruptcy, worried about paying large medical bills, unable to cover the cost of medical care visits, or other financial sacrifices.
Nearly 29% of the cancer survivors reported at least 1 financial problem resulting from cancer diagnosis, treatment, or lasting effects of that treatment.
Of all the cancer survivors in the analysis, 20.9% worried about paying large medical bills, 11.5% were unable to cover the cost of medical care visits, 7.6% reported borrowing money or going into debt, 1.4% declared bankruptcy, and 8.6% reported other financial sacrifices.
Cancer survivors who faced such financial difficulties had lower physical and mental health-related QOL, higher risk for depressed mood and psychological distress, and were more likely to worry about cancer recurrence, when compared with cancer survivors who did not face financial problems.
In addition, as the number of financial problems reported by cancer survivors increased, their QOL continued to decrease. And their risk for depressed mood, psychological distress, and worries about cancer recurrence continued to increase.
“Our results suggest that policies and practices that minimize cancer patients’ out-of-pocket costs can improve survivors’ health-related quality of life and psychological health,” Dr Carroll said.
“Reducing the financial burden of cancer care requires integrated efforts, and the study findings are useful for survivorship care programs, oncologists, payers, pharmaceutical companies, and patients and their family members.” ![]()
RPS15 mutations prevalent in aggressive chronic lymphocytic leukemia
Mutations in the RPS15 gene occurred in 8 of 41 patients with relapsing chronic lymphocytic leukemia (CLL), and the mutations were present before treatment in 7 of the 8, a possible indication that the aberrations are early genetic events in aggressive CLL pathobiology.
RPS15 mutations may lead to defective p53 stability and increased degradation, representing a potential novel mechanism in CLL pathobiology. The findings suggest “RPS15-mutant cases should be treated with alternative regimens that act independently of the p53 pathway,” wrote Dr. Viktor Ljungström of the department of immunology, genetics, and pathology, Uppsala (Sweden) University, and colleagues (Blood 2016 Feb 25. doi: 10.1182/blood-2015-10-674572).
In their study, the researchers performed whole exome sequencing of 110 samples collected before and after treatment from 41 patients with aggressive CLL that relapsed after a median of 2 years; 7 patients had mutations in RPS15 before treatment, and 8 had RPS15 mutations after treatment. The findings suggest that standard therapy with fludarabine, cyclophosphamide, and rituximab was not intrinsically mutagenic.
High frequencies of mutations were linked to poor outcome in both pretreated and relapse samples. These mutations included NOTCH1, TP53, ATM, SF3B1, MGA, and BIRC3. At least one mutation was seen before treatment in 26 of the 41 patients, and that rate rose to 33 of 41 patients at relapse. Two or more mutations were noted before treatment in 12 of 41 patients, and that rose to 15 of 41 at relapse.
In response to their findings, the researchers next performed targeted resequencing of the RPS15 hot spot (exon 4) in an extended series of 790 patients with CLL, intentionally enriched with 605 cases with adverse prognostic profiles. They found an additional 36 mutations in RPS15 (36/605, 6%). In contrast, none of the 185 patients with more favorable prognostic, IGHV-mutated CLL carried RPS15 mutations. RPS15-mutant patients without concomitant TP53 aberrations had an overall survival similar to other aggressive CLL subgroups, but none of the patients with both mutations survived at 10 years, compared with 59% of patients with wild-type RPS15 and wild-type TP53, “pointing to a dismal prognosis for RPS15-mutated CLL,” they wrote.
They also analyzed 30 cases with Richter syndrome (CLL transformed into diffuse large B-cell lymphoma), and only a single case was found to carry an RPS15 mutation, and the mutation was also observed in the preceding CLL phase. This finding indicates that RPS15 mutation probably does not underlie the transformation of CLL to Richter syndrome, according to the researchers.
Dr. Ljungström and coauthors reported having no relevant financial disclosures.
In support of the authors’ hypothesis that RPS15 mutations may be an early-acquired driver in high-risk disease, the variant allele frequency in eight serially analyzed cases remained static, with only one case gaining a mutation in RPS15, whereas the variable allele frequency increased at relapse for other well-characterized mutations in ATM, BIRC3, NFKBIE, and TP53.
Pilot experiments demonstrated specific interactions between TP53 and RPS15, and p53 stability was reduced in the presence of mutant RPS15.
The findings should prompt further investigation to determine if the consequences of RPS15 mutations depend on its interaction with TP53, or if the mutations found in other ribosomal proteins indicate a different mechanism related to the 40S subunit.
Given that RPS15 is not included in common academic or commercial sequencing panels, the presence of RPS15 mutations in other diseases may be underestimated as well.
More generally, are there other cancers with subgroups enriched for other benign-appearing genes?
Dr. James Blachly is with Wexner Medical Center, the Ohio State University, Columbus. These remarks were part of an editorial accompanying a report in Blood (2016 Feb 25. doi: 10.1182/blood-2015-10-674572).
In support of the authors’ hypothesis that RPS15 mutations may be an early-acquired driver in high-risk disease, the variant allele frequency in eight serially analyzed cases remained static, with only one case gaining a mutation in RPS15, whereas the variable allele frequency increased at relapse for other well-characterized mutations in ATM, BIRC3, NFKBIE, and TP53.
Pilot experiments demonstrated specific interactions between TP53 and RPS15, and p53 stability was reduced in the presence of mutant RPS15.
The findings should prompt further investigation to determine if the consequences of RPS15 mutations depend on its interaction with TP53, or if the mutations found in other ribosomal proteins indicate a different mechanism related to the 40S subunit.
Given that RPS15 is not included in common academic or commercial sequencing panels, the presence of RPS15 mutations in other diseases may be underestimated as well.
More generally, are there other cancers with subgroups enriched for other benign-appearing genes?
Dr. James Blachly is with Wexner Medical Center, the Ohio State University, Columbus. These remarks were part of an editorial accompanying a report in Blood (2016 Feb 25. doi: 10.1182/blood-2015-10-674572).
In support of the authors’ hypothesis that RPS15 mutations may be an early-acquired driver in high-risk disease, the variant allele frequency in eight serially analyzed cases remained static, with only one case gaining a mutation in RPS15, whereas the variable allele frequency increased at relapse for other well-characterized mutations in ATM, BIRC3, NFKBIE, and TP53.
Pilot experiments demonstrated specific interactions between TP53 and RPS15, and p53 stability was reduced in the presence of mutant RPS15.
The findings should prompt further investigation to determine if the consequences of RPS15 mutations depend on its interaction with TP53, or if the mutations found in other ribosomal proteins indicate a different mechanism related to the 40S subunit.
Given that RPS15 is not included in common academic or commercial sequencing panels, the presence of RPS15 mutations in other diseases may be underestimated as well.
More generally, are there other cancers with subgroups enriched for other benign-appearing genes?
Dr. James Blachly is with Wexner Medical Center, the Ohio State University, Columbus. These remarks were part of an editorial accompanying a report in Blood (2016 Feb 25. doi: 10.1182/blood-2015-10-674572).
Mutations in the RPS15 gene occurred in 8 of 41 patients with relapsing chronic lymphocytic leukemia (CLL), and the mutations were present before treatment in 7 of the 8, a possible indication that the aberrations are early genetic events in aggressive CLL pathobiology.
RPS15 mutations may lead to defective p53 stability and increased degradation, representing a potential novel mechanism in CLL pathobiology. The findings suggest “RPS15-mutant cases should be treated with alternative regimens that act independently of the p53 pathway,” wrote Dr. Viktor Ljungström of the department of immunology, genetics, and pathology, Uppsala (Sweden) University, and colleagues (Blood 2016 Feb 25. doi: 10.1182/blood-2015-10-674572).
In their study, the researchers performed whole exome sequencing of 110 samples collected before and after treatment from 41 patients with aggressive CLL that relapsed after a median of 2 years; 7 patients had mutations in RPS15 before treatment, and 8 had RPS15 mutations after treatment. The findings suggest that standard therapy with fludarabine, cyclophosphamide, and rituximab was not intrinsically mutagenic.
High frequencies of mutations were linked to poor outcome in both pretreated and relapse samples. These mutations included NOTCH1, TP53, ATM, SF3B1, MGA, and BIRC3. At least one mutation was seen before treatment in 26 of the 41 patients, and that rate rose to 33 of 41 patients at relapse. Two or more mutations were noted before treatment in 12 of 41 patients, and that rose to 15 of 41 at relapse.
In response to their findings, the researchers next performed targeted resequencing of the RPS15 hot spot (exon 4) in an extended series of 790 patients with CLL, intentionally enriched with 605 cases with adverse prognostic profiles. They found an additional 36 mutations in RPS15 (36/605, 6%). In contrast, none of the 185 patients with more favorable prognostic, IGHV-mutated CLL carried RPS15 mutations. RPS15-mutant patients without concomitant TP53 aberrations had an overall survival similar to other aggressive CLL subgroups, but none of the patients with both mutations survived at 10 years, compared with 59% of patients with wild-type RPS15 and wild-type TP53, “pointing to a dismal prognosis for RPS15-mutated CLL,” they wrote.
They also analyzed 30 cases with Richter syndrome (CLL transformed into diffuse large B-cell lymphoma), and only a single case was found to carry an RPS15 mutation, and the mutation was also observed in the preceding CLL phase. This finding indicates that RPS15 mutation probably does not underlie the transformation of CLL to Richter syndrome, according to the researchers.
Dr. Ljungström and coauthors reported having no relevant financial disclosures.
Mutations in the RPS15 gene occurred in 8 of 41 patients with relapsing chronic lymphocytic leukemia (CLL), and the mutations were present before treatment in 7 of the 8, a possible indication that the aberrations are early genetic events in aggressive CLL pathobiology.
RPS15 mutations may lead to defective p53 stability and increased degradation, representing a potential novel mechanism in CLL pathobiology. The findings suggest “RPS15-mutant cases should be treated with alternative regimens that act independently of the p53 pathway,” wrote Dr. Viktor Ljungström of the department of immunology, genetics, and pathology, Uppsala (Sweden) University, and colleagues (Blood 2016 Feb 25. doi: 10.1182/blood-2015-10-674572).
In their study, the researchers performed whole exome sequencing of 110 samples collected before and after treatment from 41 patients with aggressive CLL that relapsed after a median of 2 years; 7 patients had mutations in RPS15 before treatment, and 8 had RPS15 mutations after treatment. The findings suggest that standard therapy with fludarabine, cyclophosphamide, and rituximab was not intrinsically mutagenic.
High frequencies of mutations were linked to poor outcome in both pretreated and relapse samples. These mutations included NOTCH1, TP53, ATM, SF3B1, MGA, and BIRC3. At least one mutation was seen before treatment in 26 of the 41 patients, and that rate rose to 33 of 41 patients at relapse. Two or more mutations were noted before treatment in 12 of 41 patients, and that rose to 15 of 41 at relapse.
In response to their findings, the researchers next performed targeted resequencing of the RPS15 hot spot (exon 4) in an extended series of 790 patients with CLL, intentionally enriched with 605 cases with adverse prognostic profiles. They found an additional 36 mutations in RPS15 (36/605, 6%). In contrast, none of the 185 patients with more favorable prognostic, IGHV-mutated CLL carried RPS15 mutations. RPS15-mutant patients without concomitant TP53 aberrations had an overall survival similar to other aggressive CLL subgroups, but none of the patients with both mutations survived at 10 years, compared with 59% of patients with wild-type RPS15 and wild-type TP53, “pointing to a dismal prognosis for RPS15-mutated CLL,” they wrote.
They also analyzed 30 cases with Richter syndrome (CLL transformed into diffuse large B-cell lymphoma), and only a single case was found to carry an RPS15 mutation, and the mutation was also observed in the preceding CLL phase. This finding indicates that RPS15 mutation probably does not underlie the transformation of CLL to Richter syndrome, according to the researchers.
Dr. Ljungström and coauthors reported having no relevant financial disclosures.
FROM BLOOD
Key clinical point: Aberrations in the RPS15 gene before therapy may be an indicator of aggressive pathobiology in chronic lymphocytic leukemia.
Major finding: Mutations in the RPS15 gene occurred in 8 of 41 patients with relapsing CLL, and the mutations were present before treatment in 7 of the 8.
Data sources: Whole exome sequencing of 110 samples collected before and after fludarabine, cyclophosphamide, and rituximab therapy from 41 patients with relapsed CLL.
Disclosures: Dr. Ljungström and coauthors reported having no relevant financial disclosures.
Starting all CML patients on imatinib could cut costs, team says

Photo courtesy of the CDC
New research suggests that starting all patients with chronic myeloid leukemia (CML) on the generic form of Gleevec, imatinib, could result in substantial savings for patients and insurance companies in the US.
Researchers found that starting treatment with generic imatinib and then switching patients to the second-generation tyrosine kinase inhibitors (TKIs) dasatinib (Sprycel) and nilotinib (Tasigna) if necessary would be more cost-effective than the current standard of care, which allows physicians to start patients on any of the aforementioned TKIs.
In fact, the data suggest that starting all CML patients on generic imatinib could reduce the cost of treatment per patient over 5 years by nearly $90,000.
“If we start all patients on the generic form of Gleevec, and it works, then they are on a generic for the rest of their lives,” said study author William V. Padula, PhD, of Johns Hopkins University in Baltimore, Maryland.
“This amounts to a huge cost savings for them and their insurers.”
Dr Padula and his colleagues described these potential savings in the Journal of the National Cancer Institute.
The researchers compared the cost-effectiveness of the different TKIs by analyzing Truven Health Analytics MarketScan data from newly diagnosed CML patients between January 1, 2011, and December 31, 2012.
The team constructed Markov models to compare the 5-year cost-effectiveness of imatinib-first vs physician’s choice. The main outcome of the models was cost per quality-adjusted life-year (QALY).
The researchers interpreted outcomes based on a willingness-to-pay threshold of $100,000/QALY. They found that both imatinib-first and physician’s choice met that threshold.
However, imatinib-first cost less over 5 years and conferred only slightly fewer QALYs than physician’s choice—$277,401 and 3.87 QALYs for imatinib-first and $365,744 and 3.97 QALYs for physician’s choice.
So that’s a 0.10 decrement in QALYs and a savings of $88,343 over 5 years with imatinib-first. The imatinib-first incremental cost-effectiveness ratio was about $883,730/QALY.
“There is minimal risk to starting all patients on imatinib first,” Dr Padula said. “If the patient can’t tolerate the medication or it seems to be ineffective in that patient, then we can switch the patient to a more expensive drug.”
“Insurance companies have the ability to dictate which drugs physicians prescribe first, and they regularly do. Doing so here would mean very little risk to health and a lot of cost savings.” ![]()

Photo courtesy of the CDC
New research suggests that starting all patients with chronic myeloid leukemia (CML) on the generic form of Gleevec, imatinib, could result in substantial savings for patients and insurance companies in the US.
Researchers found that starting treatment with generic imatinib and then switching patients to the second-generation tyrosine kinase inhibitors (TKIs) dasatinib (Sprycel) and nilotinib (Tasigna) if necessary would be more cost-effective than the current standard of care, which allows physicians to start patients on any of the aforementioned TKIs.
In fact, the data suggest that starting all CML patients on generic imatinib could reduce the cost of treatment per patient over 5 years by nearly $90,000.
“If we start all patients on the generic form of Gleevec, and it works, then they are on a generic for the rest of their lives,” said study author William V. Padula, PhD, of Johns Hopkins University in Baltimore, Maryland.
“This amounts to a huge cost savings for them and their insurers.”
Dr Padula and his colleagues described these potential savings in the Journal of the National Cancer Institute.
The researchers compared the cost-effectiveness of the different TKIs by analyzing Truven Health Analytics MarketScan data from newly diagnosed CML patients between January 1, 2011, and December 31, 2012.
The team constructed Markov models to compare the 5-year cost-effectiveness of imatinib-first vs physician’s choice. The main outcome of the models was cost per quality-adjusted life-year (QALY).
The researchers interpreted outcomes based on a willingness-to-pay threshold of $100,000/QALY. They found that both imatinib-first and physician’s choice met that threshold.
However, imatinib-first cost less over 5 years and conferred only slightly fewer QALYs than physician’s choice—$277,401 and 3.87 QALYs for imatinib-first and $365,744 and 3.97 QALYs for physician’s choice.
So that’s a 0.10 decrement in QALYs and a savings of $88,343 over 5 years with imatinib-first. The imatinib-first incremental cost-effectiveness ratio was about $883,730/QALY.
“There is minimal risk to starting all patients on imatinib first,” Dr Padula said. “If the patient can’t tolerate the medication or it seems to be ineffective in that patient, then we can switch the patient to a more expensive drug.”
“Insurance companies have the ability to dictate which drugs physicians prescribe first, and they regularly do. Doing so here would mean very little risk to health and a lot of cost savings.” ![]()

Photo courtesy of the CDC
New research suggests that starting all patients with chronic myeloid leukemia (CML) on the generic form of Gleevec, imatinib, could result in substantial savings for patients and insurance companies in the US.
Researchers found that starting treatment with generic imatinib and then switching patients to the second-generation tyrosine kinase inhibitors (TKIs) dasatinib (Sprycel) and nilotinib (Tasigna) if necessary would be more cost-effective than the current standard of care, which allows physicians to start patients on any of the aforementioned TKIs.
In fact, the data suggest that starting all CML patients on generic imatinib could reduce the cost of treatment per patient over 5 years by nearly $90,000.
“If we start all patients on the generic form of Gleevec, and it works, then they are on a generic for the rest of their lives,” said study author William V. Padula, PhD, of Johns Hopkins University in Baltimore, Maryland.
“This amounts to a huge cost savings for them and their insurers.”
Dr Padula and his colleagues described these potential savings in the Journal of the National Cancer Institute.
The researchers compared the cost-effectiveness of the different TKIs by analyzing Truven Health Analytics MarketScan data from newly diagnosed CML patients between January 1, 2011, and December 31, 2012.
The team constructed Markov models to compare the 5-year cost-effectiveness of imatinib-first vs physician’s choice. The main outcome of the models was cost per quality-adjusted life-year (QALY).
The researchers interpreted outcomes based on a willingness-to-pay threshold of $100,000/QALY. They found that both imatinib-first and physician’s choice met that threshold.
However, imatinib-first cost less over 5 years and conferred only slightly fewer QALYs than physician’s choice—$277,401 and 3.87 QALYs for imatinib-first and $365,744 and 3.97 QALYs for physician’s choice.
So that’s a 0.10 decrement in QALYs and a savings of $88,343 over 5 years with imatinib-first. The imatinib-first incremental cost-effectiveness ratio was about $883,730/QALY.
“There is minimal risk to starting all patients on imatinib first,” Dr Padula said. “If the patient can’t tolerate the medication or it seems to be ineffective in that patient, then we can switch the patient to a more expensive drug.”
“Insurance companies have the ability to dictate which drugs physicians prescribe first, and they regularly do. Doing so here would mean very little risk to health and a lot of cost savings.” ![]()
Idelalisib use halted in six combo therapy trials, FDA announces
An increased rate of adverse events, including deaths, have been reported in clinical trials with idelalisib (Zydelig) in combination with other cancer medicines, the U.S. Food and Drug Administration announced.
Gilead Sciences, Inc. has confirmed that they are stopping six clinical trials in patients with chronic lymphocytic leukemia, small lymphocytic lymphoma and indolent non-Hodgkin lymphomas. The FDA is reviewing the findings of the clinical trials and will communicate new information as necessary, according to the FDA press release.
Idelalisib is not approved for previously untreated chronic lymphocytic leukemia. It is approved by the FDA for the treatment of:
• Relapsed chronic lymphocytic leukemia, in combination with rituximab, in patients for whom rituximab alone would be considered appropriate therapy due to other co-morbidities.
• Relapsed follicular B-cell non-Hodgkin lymphoma in patients who have received at least two prior systemic therapies.
• Relapsed small lymphocytic lymphoma in patients who have received at least two prior systemic therapies.
Adverse events involving idelalisib should be reported to the FDA MedWatch program, the release advised.
On Twitter @maryjodales
An increased rate of adverse events, including deaths, have been reported in clinical trials with idelalisib (Zydelig) in combination with other cancer medicines, the U.S. Food and Drug Administration announced.
Gilead Sciences, Inc. has confirmed that they are stopping six clinical trials in patients with chronic lymphocytic leukemia, small lymphocytic lymphoma and indolent non-Hodgkin lymphomas. The FDA is reviewing the findings of the clinical trials and will communicate new information as necessary, according to the FDA press release.
Idelalisib is not approved for previously untreated chronic lymphocytic leukemia. It is approved by the FDA for the treatment of:
• Relapsed chronic lymphocytic leukemia, in combination with rituximab, in patients for whom rituximab alone would be considered appropriate therapy due to other co-morbidities.
• Relapsed follicular B-cell non-Hodgkin lymphoma in patients who have received at least two prior systemic therapies.
• Relapsed small lymphocytic lymphoma in patients who have received at least two prior systemic therapies.
Adverse events involving idelalisib should be reported to the FDA MedWatch program, the release advised.
On Twitter @maryjodales
An increased rate of adverse events, including deaths, have been reported in clinical trials with idelalisib (Zydelig) in combination with other cancer medicines, the U.S. Food and Drug Administration announced.
Gilead Sciences, Inc. has confirmed that they are stopping six clinical trials in patients with chronic lymphocytic leukemia, small lymphocytic lymphoma and indolent non-Hodgkin lymphomas. The FDA is reviewing the findings of the clinical trials and will communicate new information as necessary, according to the FDA press release.
Idelalisib is not approved for previously untreated chronic lymphocytic leukemia. It is approved by the FDA for the treatment of:
• Relapsed chronic lymphocytic leukemia, in combination with rituximab, in patients for whom rituximab alone would be considered appropriate therapy due to other co-morbidities.
• Relapsed follicular B-cell non-Hodgkin lymphoma in patients who have received at least two prior systemic therapies.
• Relapsed small lymphocytic lymphoma in patients who have received at least two prior systemic therapies.
Adverse events involving idelalisib should be reported to the FDA MedWatch program, the release advised.
On Twitter @maryjodales
Protein plays key role in B-ALL subtype

An RNA binding protein promotes the development of MLL-rearranged B-cell acute lymphoblastic leukemia (B-ALL), according to research published in The Journal of Clinical Investigation.
Researchers found an overabundance of the protein, IGF2BP3, in MLL-rearranged B-ALL.
They also identified genes that are directly regulated by IFG2BP3, and many of them turned out to be oncogenes that have already been implicated in cancers.
The overall effect of IFG2BP3 in MLL-rearranged B-ALL is to promote the proliferation of B cells by shifting the expression of a large number of genes, explained study author Jeremy Sanford, PhD, of the University of California Santa Cruz.
“This protein, IFG2BP3, has been correlated with many types of malignancies and with the worst prognoses,” he noted. “What is exciting about this study is that it goes beyond correlation and shows causation, because we demonstrated, for the first time, that aberrant expression of this protein is sufficient to induce pathology.”
This research began in the lab of Dinesh Rao, PhD, an assistant professor at the University of California Los Angeles who was studying MLL-rearranged B-ALL.
After researchers in Dr Rao’s lab identified IGF2BP3 as one of the top dysregulated genes in this malignancy, they began working with Dr Sanford’s lab to figure out which genes were being directly regulated by IGF2BP3.
Dr Sanford’s lab was among the few using individual nucleotide resolution crosslinking immunoprecipitation (iCLIP), a technique that can capture RNA molecules bound to a particular protein.
iCLIP enabled the researchers to identify IGF2BP3 binding sites in several hundred RNA transcripts in 2 B-ALL cell lines.
The work also revealed that IGF2BP3 enhanced the expression of MYC and other oncogenes in hematopoietic stem cells.
In experiments with mice, the researchers found that overexpression of IGF2BP3 in the bone marrow leads to proliferation of hematopoietic stem cells and B-cell progenitors, reproducing some features of MLL-rearranged B-ALL.
“Understanding its mechanism of action is important for thinking about therapeutics that might interfere with the action of this protein in disease,” Dr Sanford said.
“One possibility is an RNA-based therapeutic that could sequester the protein and keep it from binding to RNA transcripts. That would be a way to influence the expression of many genes involved in the proliferation of cancer cells.” ![]()

An RNA binding protein promotes the development of MLL-rearranged B-cell acute lymphoblastic leukemia (B-ALL), according to research published in The Journal of Clinical Investigation.
Researchers found an overabundance of the protein, IGF2BP3, in MLL-rearranged B-ALL.
They also identified genes that are directly regulated by IFG2BP3, and many of them turned out to be oncogenes that have already been implicated in cancers.
The overall effect of IFG2BP3 in MLL-rearranged B-ALL is to promote the proliferation of B cells by shifting the expression of a large number of genes, explained study author Jeremy Sanford, PhD, of the University of California Santa Cruz.
“This protein, IFG2BP3, has been correlated with many types of malignancies and with the worst prognoses,” he noted. “What is exciting about this study is that it goes beyond correlation and shows causation, because we demonstrated, for the first time, that aberrant expression of this protein is sufficient to induce pathology.”
This research began in the lab of Dinesh Rao, PhD, an assistant professor at the University of California Los Angeles who was studying MLL-rearranged B-ALL.
After researchers in Dr Rao’s lab identified IGF2BP3 as one of the top dysregulated genes in this malignancy, they began working with Dr Sanford’s lab to figure out which genes were being directly regulated by IGF2BP3.
Dr Sanford’s lab was among the few using individual nucleotide resolution crosslinking immunoprecipitation (iCLIP), a technique that can capture RNA molecules bound to a particular protein.
iCLIP enabled the researchers to identify IGF2BP3 binding sites in several hundred RNA transcripts in 2 B-ALL cell lines.
The work also revealed that IGF2BP3 enhanced the expression of MYC and other oncogenes in hematopoietic stem cells.
In experiments with mice, the researchers found that overexpression of IGF2BP3 in the bone marrow leads to proliferation of hematopoietic stem cells and B-cell progenitors, reproducing some features of MLL-rearranged B-ALL.
“Understanding its mechanism of action is important for thinking about therapeutics that might interfere with the action of this protein in disease,” Dr Sanford said.
“One possibility is an RNA-based therapeutic that could sequester the protein and keep it from binding to RNA transcripts. That would be a way to influence the expression of many genes involved in the proliferation of cancer cells.” ![]()

An RNA binding protein promotes the development of MLL-rearranged B-cell acute lymphoblastic leukemia (B-ALL), according to research published in The Journal of Clinical Investigation.
Researchers found an overabundance of the protein, IGF2BP3, in MLL-rearranged B-ALL.
They also identified genes that are directly regulated by IFG2BP3, and many of them turned out to be oncogenes that have already been implicated in cancers.
The overall effect of IFG2BP3 in MLL-rearranged B-ALL is to promote the proliferation of B cells by shifting the expression of a large number of genes, explained study author Jeremy Sanford, PhD, of the University of California Santa Cruz.
“This protein, IFG2BP3, has been correlated with many types of malignancies and with the worst prognoses,” he noted. “What is exciting about this study is that it goes beyond correlation and shows causation, because we demonstrated, for the first time, that aberrant expression of this protein is sufficient to induce pathology.”
This research began in the lab of Dinesh Rao, PhD, an assistant professor at the University of California Los Angeles who was studying MLL-rearranged B-ALL.
After researchers in Dr Rao’s lab identified IGF2BP3 as one of the top dysregulated genes in this malignancy, they began working with Dr Sanford’s lab to figure out which genes were being directly regulated by IGF2BP3.
Dr Sanford’s lab was among the few using individual nucleotide resolution crosslinking immunoprecipitation (iCLIP), a technique that can capture RNA molecules bound to a particular protein.
iCLIP enabled the researchers to identify IGF2BP3 binding sites in several hundred RNA transcripts in 2 B-ALL cell lines.
The work also revealed that IGF2BP3 enhanced the expression of MYC and other oncogenes in hematopoietic stem cells.
In experiments with mice, the researchers found that overexpression of IGF2BP3 in the bone marrow leads to proliferation of hematopoietic stem cells and B-cell progenitors, reproducing some features of MLL-rearranged B-ALL.
“Understanding its mechanism of action is important for thinking about therapeutics that might interfere with the action of this protein in disease,” Dr Sanford said.
“One possibility is an RNA-based therapeutic that could sequester the protein and keep it from binding to RNA transcripts. That would be a way to influence the expression of many genes involved in the proliferation of cancer cells.” ![]()
Idelalisib trials stopped due to AEs

Photo courtesy of
Gilead Sciences
The US Food and Drug Administration (FDA) has reported that Gilead Sciences, Inc., is stopping 6 clinical trials of idelalisib (Zydelig) due to adverse events (AEs) observed in patients receiving idelalisib in combination with other drugs.
The AEs, which include deaths, were mostly related to infections.
The trials include patients with chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma, and indolent non-Hodgkin lymphomas.
The FDA said it is reviewing the findings of these trials and will communicate new information as necessary.
A few days ago, the European Medicines Agency (EMA) announced its decision to review the safety of idelalisib due to the aforementioned AEs. The EMA said it is reviewing data from 3 idelalisib trials.
While this review is underway, the EMA advised that patients starting or already on treatment with idelalisib be carefully monitored for signs of infection. If the drug is well tolerated, treatment should not be stopped.
The FDA has not made any recommendations about treatment with idelalisib.
About idelalisib
Idelalisib is currently approved by the FDA for use in combination with rituximab to treat patients with relapsed CLL who cannot receive rituximab alone.
Idelalisib also has accelerated approval from the FDA to treat patients with relapsed follicular lymphoma who have received at least 2 prior systemic therapies and patients with relapsed small lymphocytic lymphoma who have received at least 2 prior systemic therapies.
In the European Union, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients deemed unsuitable for chemo-immunotherapy.
Idelalisib is also approved in the European Union as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment. ![]()

Photo courtesy of
Gilead Sciences
The US Food and Drug Administration (FDA) has reported that Gilead Sciences, Inc., is stopping 6 clinical trials of idelalisib (Zydelig) due to adverse events (AEs) observed in patients receiving idelalisib in combination with other drugs.
The AEs, which include deaths, were mostly related to infections.
The trials include patients with chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma, and indolent non-Hodgkin lymphomas.
The FDA said it is reviewing the findings of these trials and will communicate new information as necessary.
A few days ago, the European Medicines Agency (EMA) announced its decision to review the safety of idelalisib due to the aforementioned AEs. The EMA said it is reviewing data from 3 idelalisib trials.
While this review is underway, the EMA advised that patients starting or already on treatment with idelalisib be carefully monitored for signs of infection. If the drug is well tolerated, treatment should not be stopped.
The FDA has not made any recommendations about treatment with idelalisib.
About idelalisib
Idelalisib is currently approved by the FDA for use in combination with rituximab to treat patients with relapsed CLL who cannot receive rituximab alone.
Idelalisib also has accelerated approval from the FDA to treat patients with relapsed follicular lymphoma who have received at least 2 prior systemic therapies and patients with relapsed small lymphocytic lymphoma who have received at least 2 prior systemic therapies.
In the European Union, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients deemed unsuitable for chemo-immunotherapy.
Idelalisib is also approved in the European Union as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment. ![]()

Photo courtesy of
Gilead Sciences
The US Food and Drug Administration (FDA) has reported that Gilead Sciences, Inc., is stopping 6 clinical trials of idelalisib (Zydelig) due to adverse events (AEs) observed in patients receiving idelalisib in combination with other drugs.
The AEs, which include deaths, were mostly related to infections.
The trials include patients with chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma, and indolent non-Hodgkin lymphomas.
The FDA said it is reviewing the findings of these trials and will communicate new information as necessary.
A few days ago, the European Medicines Agency (EMA) announced its decision to review the safety of idelalisib due to the aforementioned AEs. The EMA said it is reviewing data from 3 idelalisib trials.
While this review is underway, the EMA advised that patients starting or already on treatment with idelalisib be carefully monitored for signs of infection. If the drug is well tolerated, treatment should not be stopped.
The FDA has not made any recommendations about treatment with idelalisib.
About idelalisib
Idelalisib is currently approved by the FDA for use in combination with rituximab to treat patients with relapsed CLL who cannot receive rituximab alone.
Idelalisib also has accelerated approval from the FDA to treat patients with relapsed follicular lymphoma who have received at least 2 prior systemic therapies and patients with relapsed small lymphocytic lymphoma who have received at least 2 prior systemic therapies.
In the European Union, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients deemed unsuitable for chemo-immunotherapy.
Idelalisib is also approved in the European Union as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment. ![]()
AEs prompt EMA review of idelalisib

Photo courtesy of
Gilead Sciences, Inc.
The European Medicines Agency (EMA) is reviewing the safety of idelalisib (Zydelig), a drug approved to treat chronic lymphocytic leukemia (CLL) and follicular lymphoma in the European Union (EU).
The European Commission (EC) requested the review because of serious adverse events (AEs), including deaths, reported in 3 clinical trials investigating idelalisib in combination with other drugs.
The AEs were mostly infection-related.
The EMA is reviewing data from these studies to assess whether the findings have any consequences for the authorized uses of idelalisib.
In the meantime, the EMA advises that patients starting or already on treatment with idelalisib be carefully monitored for signs of infections. If the drug is well tolerated, treatment should not be stopped.
The EMA is considering whether any other immediate measures are necessary during the review period. The agency said it will communicate further and keep doctors and patients informed as appropriate.
About idelalisib
In the EU, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients unsuitable for chemo-immunotherapy.
Idelalisib is also approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
About the trials
The trials in which patients have experienced serious AEs involve patients with CLL and indolent non-Hodgkin lymphoma (NHL).
In one trial (NCT01732926), researchers are evaluating idelalisib in combination with bendamustine and rituximab for previously treated indolent NHL.
In another (NCT01732913), researchers are testing idelalisib in combination with rituximab for previously treated indolent NHL.
And in the third (NCT01980888), researchers are evaluating idelalisib in combination with bendamustine and rituximab in patients with previously untreated CLL.
The EMA noted that these studies are investigating combinations of drugs that are currently not approved in the EU and include patients with disease characteristics different from those covered by the approved indications for idelalisib.
About the review
The EMA has begun the review of idelalisib at the request of the EC, under Article 20 of Directive 2001/83/EC.
The review is being carried out by the EMA’s Pharmacovigilance Risk Assessment Committee, the committee responsible for the evaluation of safety issues for human medicines, which will make a set of recommendations.
Those recommendations will then be forwarded to the Committee for Medicinal Products for Human Use, which is responsible for questions concerning medicines for human use and will adopt a final opinion on the safety of idelalisib.
The final stage of the review procedure is the EC’s adoption of a legally binding decision that is applicable in all EU member states. ![]()

Photo courtesy of
Gilead Sciences, Inc.
The European Medicines Agency (EMA) is reviewing the safety of idelalisib (Zydelig), a drug approved to treat chronic lymphocytic leukemia (CLL) and follicular lymphoma in the European Union (EU).
The European Commission (EC) requested the review because of serious adverse events (AEs), including deaths, reported in 3 clinical trials investigating idelalisib in combination with other drugs.
The AEs were mostly infection-related.
The EMA is reviewing data from these studies to assess whether the findings have any consequences for the authorized uses of idelalisib.
In the meantime, the EMA advises that patients starting or already on treatment with idelalisib be carefully monitored for signs of infections. If the drug is well tolerated, treatment should not be stopped.
The EMA is considering whether any other immediate measures are necessary during the review period. The agency said it will communicate further and keep doctors and patients informed as appropriate.
About idelalisib
In the EU, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients unsuitable for chemo-immunotherapy.
Idelalisib is also approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
About the trials
The trials in which patients have experienced serious AEs involve patients with CLL and indolent non-Hodgkin lymphoma (NHL).
In one trial (NCT01732926), researchers are evaluating idelalisib in combination with bendamustine and rituximab for previously treated indolent NHL.
In another (NCT01732913), researchers are testing idelalisib in combination with rituximab for previously treated indolent NHL.
And in the third (NCT01980888), researchers are evaluating idelalisib in combination with bendamustine and rituximab in patients with previously untreated CLL.
The EMA noted that these studies are investigating combinations of drugs that are currently not approved in the EU and include patients with disease characteristics different from those covered by the approved indications for idelalisib.
About the review
The EMA has begun the review of idelalisib at the request of the EC, under Article 20 of Directive 2001/83/EC.
The review is being carried out by the EMA’s Pharmacovigilance Risk Assessment Committee, the committee responsible for the evaluation of safety issues for human medicines, which will make a set of recommendations.
Those recommendations will then be forwarded to the Committee for Medicinal Products for Human Use, which is responsible for questions concerning medicines for human use and will adopt a final opinion on the safety of idelalisib.
The final stage of the review procedure is the EC’s adoption of a legally binding decision that is applicable in all EU member states. ![]()

Photo courtesy of
Gilead Sciences, Inc.
The European Medicines Agency (EMA) is reviewing the safety of idelalisib (Zydelig), a drug approved to treat chronic lymphocytic leukemia (CLL) and follicular lymphoma in the European Union (EU).
The European Commission (EC) requested the review because of serious adverse events (AEs), including deaths, reported in 3 clinical trials investigating idelalisib in combination with other drugs.
The AEs were mostly infection-related.
The EMA is reviewing data from these studies to assess whether the findings have any consequences for the authorized uses of idelalisib.
In the meantime, the EMA advises that patients starting or already on treatment with idelalisib be carefully monitored for signs of infections. If the drug is well tolerated, treatment should not be stopped.
The EMA is considering whether any other immediate measures are necessary during the review period. The agency said it will communicate further and keep doctors and patients informed as appropriate.
About idelalisib
In the EU, idelalisib is approved for use in combination with rituximab to treat adults with CLL who have received at least 1 prior therapy or as first-line treatment in the presence of 17p deletion or TP53 mutation in CLL patients unsuitable for chemo-immunotherapy.
Idelalisib is also approved as monotherapy for adults with follicular lymphoma that is refractory to 2 prior lines of treatment.
About the trials
The trials in which patients have experienced serious AEs involve patients with CLL and indolent non-Hodgkin lymphoma (NHL).
In one trial (NCT01732926), researchers are evaluating idelalisib in combination with bendamustine and rituximab for previously treated indolent NHL.
In another (NCT01732913), researchers are testing idelalisib in combination with rituximab for previously treated indolent NHL.
And in the third (NCT01980888), researchers are evaluating idelalisib in combination with bendamustine and rituximab in patients with previously untreated CLL.
The EMA noted that these studies are investigating combinations of drugs that are currently not approved in the EU and include patients with disease characteristics different from those covered by the approved indications for idelalisib.
About the review
The EMA has begun the review of idelalisib at the request of the EC, under Article 20 of Directive 2001/83/EC.
The review is being carried out by the EMA’s Pharmacovigilance Risk Assessment Committee, the committee responsible for the evaluation of safety issues for human medicines, which will make a set of recommendations.
Those recommendations will then be forwarded to the Committee for Medicinal Products for Human Use, which is responsible for questions concerning medicines for human use and will adopt a final opinion on the safety of idelalisib.
The final stage of the review procedure is the EC’s adoption of a legally binding decision that is applicable in all EU member states. ![]()
Class of drugs could treat B-cell malignancies
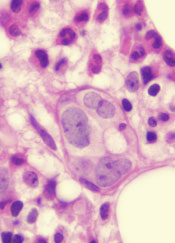
A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()

A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies. ![]()

A class of drugs targeting a protein found in the endoplasmic reticulum could be effective against B-cell malignancies, according to a study published in Cancer Research.
The protein, STING, plays a critical role in producing type I interferons that help regulate the immune system.
Previous research suggested that STING agonists can improve immune responses when used in cancer immunotherapy or as vaccine adjuvants.
However, the way B cells respond to STING agonists was not well understood.
Chih-Chi Andrew Hu, PhD, of The Wistar Institute in Philadelphia, Pennsylvania, and his colleagues conducted a study to gain some insight.
The researchers found that normal B cells respond to STING agonists by undergoing mitochondria-mediated apoptosis, and STING agonists induce apoptosis in
malignant B cells through binding to STING.
STING agonists proved cytotoxic to B-cell leukemia, lymphoma, and multiple myeloma in vitro. But the drugs did not induce apoptosis in solid tumor malignancies or normal T cells.
The research also revealed that the IRE-1/XBP-1 stress response pathway is required for normal STING function. And B-cell leukemia, lymphoma, and myeloma require the IRE-1/XBP-1 pathway to be activated for survival.
Stimulation by STING agonists suppressed the IRE-1/XBP-1 pathway, which increased the level of apoptosis in malignant B cells.
The researchers confirmed these results in animal models, as treatment with STING agonists led to regression of chronic lymphocytic leukemia and multiple myeloma in mice.
“This specific cytotoxicity toward B cells strongly supports the use of STING agonists in the treatment of B-cell hematologic malignancies,” said Chih-Hang Anthony Tang, MD, PhD, of The Wistar Institute.
“We also believe that cytotoxicity in normal B cells can be managed with the administration of intravenous immunoglobulin that can help maintain normal levels of antibodies while treatment is being administered. This is something we plan on studying further.”
The Wistar Institute’s business development team is looking for a development partner for the advancement of novel STING agonists in treating B-cell hematologic malignancies.
Guidelines on asparaginase hypersensitivity and silent inactivation in ALL
Recommendations for identification and management of asparaginase hypersensitivity and silent inactivation have been issued in consensus expert guidelines written by Dr. Inge M. van der Sluis of Sophia Children’s Hospital, Rotterdam, The Netherlands, and her colleagues.
The guidelines were published in Haematologica, the Journal of the European Hematology Association (Haematologica 2016 March;101:279-85).
Hypersensitivity reactions to asparaginase therapy can occur in 30% of children with acute lymphoblastic leukemia (ALL), and “silent inactivation” with the formation of neutralizing antibodies and reduced asparaginase activity can occur in the absence of a clinically evident allergic reaction. The guidelines seek to reduce the risk that patients will receive an inadequate course of asparaginase therapy due to either intolerable side effects or silent inactivation. The recommendations address therapeutic drug monitoring through serum asparaginase level assessment, indications for switching asparaginase preparations, and recommendations for monitoring after changes in asparaginase preparation.
The guidelines conclude that measures of serum asparaginase activity levels are the best and most reliable indicators of asparaginase efficacy. Trough asparaginase activity levels of at least 0.1 IU/mL appear to be a safe target level to ensure therapeutic benefit. Anti-asparaginase antibodies and asparagine measurements are not indicated for clinical decision making outside the context of a clinical trial.
Should clinical hypersensitivity occur, the patient has likely developed anti-asparaginase antibodies. Continued use of asparaginase of the same formulation will be ineffective in the treatment of leukemia and should be discouraged, even when it is clinically possible to administer the same preparation by using premedication such as steroids and antihistamines or by decreasing the infusion rate. Such measures reduce the symptoms of the allergy, but do not prevent asparaginase inactivation.
In patients with grade 1 allergic reactions (transient flushing or rash without need for intervention), the guideline writers recommend monitoring serum asparaginase level in real-time within 7 days to identify inactivation. In those with grade 2-4 reactions, the guideline writers recommend switching the asparaginase preparation, and there is no definite need to check asparaginase levels.
Screening for silent inactivation should be considered in all patients undergoing therapy for ALL with asparaginase, especially when there have been gaps in asparaginase therapy or in the setting of the treatment of relapsed leukemia.
“We recommend the testing of serum asparaginase activity level after the first dose of E. coli–derived asparaginase. With the use of pegaspargase, this should be done within 7 days of the dose. If the level is detectable but less than 0.1 IU/mL, activity should be rechecked at day 14,” according to the authors.
In patients who receive pegylated asparaginase every 14 days without a gap in dosing, it would be reasonable to confirm a low or undetectable level after a subsequent dose, and to change to an Erwinia preparation, for example, if two consecutive levels are undetectable.
“When there is a gap between asparaginase doses, we recommend checking a level after the first dose of asparaginase administered after the gap, with a gap defined as a period in which asparaginase activity level will have decreased to less than the [lower limit of quantification] between doses. In practice, this is usually the case when there is an interval of at least 4 weeks between pegylated asparaginase doses. With native E. coli asparaginase, we recommend measuring a trough level after the first dose and after every reintroduction of asparaginase,” the authors wrote.
When a limited number of asparaginase doses is planned or there are prolonged gaps between doses, the guidelines advise screening for silent inactivation after every asparaginase dose, and considering a switching in preparation based upon a single undetectable or low level.
The guidelines also recommend more insight into the costs of treatment with the various asparaginase preparations. In addition to the detection of silent inactivation, therapeutic drug monitoring can be used to individualize dosing based on activity levels and has the potential to reduce medication costs.
Several of the consensus writers have participated in advisory boards of companies involved in asparaginase, including Sigma-Tau, Medac, and Jazz Pharmaceuticals. Jazz Pharmaceuticals provided financial support for the investigator-initiated consensus meeting.
On Twitter @maryjodales
Recommendations for identification and management of asparaginase hypersensitivity and silent inactivation have been issued in consensus expert guidelines written by Dr. Inge M. van der Sluis of Sophia Children’s Hospital, Rotterdam, The Netherlands, and her colleagues.
The guidelines were published in Haematologica, the Journal of the European Hematology Association (Haematologica 2016 March;101:279-85).
Hypersensitivity reactions to asparaginase therapy can occur in 30% of children with acute lymphoblastic leukemia (ALL), and “silent inactivation” with the formation of neutralizing antibodies and reduced asparaginase activity can occur in the absence of a clinically evident allergic reaction. The guidelines seek to reduce the risk that patients will receive an inadequate course of asparaginase therapy due to either intolerable side effects or silent inactivation. The recommendations address therapeutic drug monitoring through serum asparaginase level assessment, indications for switching asparaginase preparations, and recommendations for monitoring after changes in asparaginase preparation.
The guidelines conclude that measures of serum asparaginase activity levels are the best and most reliable indicators of asparaginase efficacy. Trough asparaginase activity levels of at least 0.1 IU/mL appear to be a safe target level to ensure therapeutic benefit. Anti-asparaginase antibodies and asparagine measurements are not indicated for clinical decision making outside the context of a clinical trial.
Should clinical hypersensitivity occur, the patient has likely developed anti-asparaginase antibodies. Continued use of asparaginase of the same formulation will be ineffective in the treatment of leukemia and should be discouraged, even when it is clinically possible to administer the same preparation by using premedication such as steroids and antihistamines or by decreasing the infusion rate. Such measures reduce the symptoms of the allergy, but do not prevent asparaginase inactivation.
In patients with grade 1 allergic reactions (transient flushing or rash without need for intervention), the guideline writers recommend monitoring serum asparaginase level in real-time within 7 days to identify inactivation. In those with grade 2-4 reactions, the guideline writers recommend switching the asparaginase preparation, and there is no definite need to check asparaginase levels.
Screening for silent inactivation should be considered in all patients undergoing therapy for ALL with asparaginase, especially when there have been gaps in asparaginase therapy or in the setting of the treatment of relapsed leukemia.
“We recommend the testing of serum asparaginase activity level after the first dose of E. coli–derived asparaginase. With the use of pegaspargase, this should be done within 7 days of the dose. If the level is detectable but less than 0.1 IU/mL, activity should be rechecked at day 14,” according to the authors.
In patients who receive pegylated asparaginase every 14 days without a gap in dosing, it would be reasonable to confirm a low or undetectable level after a subsequent dose, and to change to an Erwinia preparation, for example, if two consecutive levels are undetectable.
“When there is a gap between asparaginase doses, we recommend checking a level after the first dose of asparaginase administered after the gap, with a gap defined as a period in which asparaginase activity level will have decreased to less than the [lower limit of quantification] between doses. In practice, this is usually the case when there is an interval of at least 4 weeks between pegylated asparaginase doses. With native E. coli asparaginase, we recommend measuring a trough level after the first dose and after every reintroduction of asparaginase,” the authors wrote.
When a limited number of asparaginase doses is planned or there are prolonged gaps between doses, the guidelines advise screening for silent inactivation after every asparaginase dose, and considering a switching in preparation based upon a single undetectable or low level.
The guidelines also recommend more insight into the costs of treatment with the various asparaginase preparations. In addition to the detection of silent inactivation, therapeutic drug monitoring can be used to individualize dosing based on activity levels and has the potential to reduce medication costs.
Several of the consensus writers have participated in advisory boards of companies involved in asparaginase, including Sigma-Tau, Medac, and Jazz Pharmaceuticals. Jazz Pharmaceuticals provided financial support for the investigator-initiated consensus meeting.
On Twitter @maryjodales
Recommendations for identification and management of asparaginase hypersensitivity and silent inactivation have been issued in consensus expert guidelines written by Dr. Inge M. van der Sluis of Sophia Children’s Hospital, Rotterdam, The Netherlands, and her colleagues.
The guidelines were published in Haematologica, the Journal of the European Hematology Association (Haematologica 2016 March;101:279-85).
Hypersensitivity reactions to asparaginase therapy can occur in 30% of children with acute lymphoblastic leukemia (ALL), and “silent inactivation” with the formation of neutralizing antibodies and reduced asparaginase activity can occur in the absence of a clinically evident allergic reaction. The guidelines seek to reduce the risk that patients will receive an inadequate course of asparaginase therapy due to either intolerable side effects or silent inactivation. The recommendations address therapeutic drug monitoring through serum asparaginase level assessment, indications for switching asparaginase preparations, and recommendations for monitoring after changes in asparaginase preparation.
The guidelines conclude that measures of serum asparaginase activity levels are the best and most reliable indicators of asparaginase efficacy. Trough asparaginase activity levels of at least 0.1 IU/mL appear to be a safe target level to ensure therapeutic benefit. Anti-asparaginase antibodies and asparagine measurements are not indicated for clinical decision making outside the context of a clinical trial.
Should clinical hypersensitivity occur, the patient has likely developed anti-asparaginase antibodies. Continued use of asparaginase of the same formulation will be ineffective in the treatment of leukemia and should be discouraged, even when it is clinically possible to administer the same preparation by using premedication such as steroids and antihistamines or by decreasing the infusion rate. Such measures reduce the symptoms of the allergy, but do not prevent asparaginase inactivation.
In patients with grade 1 allergic reactions (transient flushing or rash without need for intervention), the guideline writers recommend monitoring serum asparaginase level in real-time within 7 days to identify inactivation. In those with grade 2-4 reactions, the guideline writers recommend switching the asparaginase preparation, and there is no definite need to check asparaginase levels.
Screening for silent inactivation should be considered in all patients undergoing therapy for ALL with asparaginase, especially when there have been gaps in asparaginase therapy or in the setting of the treatment of relapsed leukemia.
“We recommend the testing of serum asparaginase activity level after the first dose of E. coli–derived asparaginase. With the use of pegaspargase, this should be done within 7 days of the dose. If the level is detectable but less than 0.1 IU/mL, activity should be rechecked at day 14,” according to the authors.
In patients who receive pegylated asparaginase every 14 days without a gap in dosing, it would be reasonable to confirm a low or undetectable level after a subsequent dose, and to change to an Erwinia preparation, for example, if two consecutive levels are undetectable.
“When there is a gap between asparaginase doses, we recommend checking a level after the first dose of asparaginase administered after the gap, with a gap defined as a period in which asparaginase activity level will have decreased to less than the [lower limit of quantification] between doses. In practice, this is usually the case when there is an interval of at least 4 weeks between pegylated asparaginase doses. With native E. coli asparaginase, we recommend measuring a trough level after the first dose and after every reintroduction of asparaginase,” the authors wrote.
When a limited number of asparaginase doses is planned or there are prolonged gaps between doses, the guidelines advise screening for silent inactivation after every asparaginase dose, and considering a switching in preparation based upon a single undetectable or low level.
The guidelines also recommend more insight into the costs of treatment with the various asparaginase preparations. In addition to the detection of silent inactivation, therapeutic drug monitoring can be used to individualize dosing based on activity levels and has the potential to reduce medication costs.
Several of the consensus writers have participated in advisory boards of companies involved in asparaginase, including Sigma-Tau, Medac, and Jazz Pharmaceuticals. Jazz Pharmaceuticals provided financial support for the investigator-initiated consensus meeting.
On Twitter @maryjodales
FROM HAEMATOLOGICA