User login
NAFLD increases risk for severe infections
People with nonalcoholic fatty liver disease (NAFLD) are more likely to develop severe infections requiring hospitalization, according to findings from a large Swedish cohort study.
The increased risk was equal to one extra severe infection in every six patients with NAFLD by 20 years after diagnosis, wrote Fahim Ebrahimi, MD, of the Karolinska Institute in Stockholm, and coauthors.
“Accumulating evidence suggests that NAFLD can affect multiple organ systems, which is not surprising, as the liver has multiple functions – regulating metabolism and being a central organ of the immune system,” Dr. Ebrahimi said in an interview.
The study was published online in Clinical Gastroenterology and Hepatology.
“Up to a fifth of cells in the liver are immune cells that process numerous antigens and pathogens from the gastrointestinal tract,” Dr. Ebrahimi noted. “We were intrigued by experimental studies showing that, in NAFLD, many of these key immune cells become dysfunctional at various levels, which may affect disease progression, but at the same time also increase the susceptibility to viral, bacterial, and fungal infections.”
Patients with NAFLD have metabolic risk factors known to increase infection risk, but a smaller study by a different research group had found that NAFLD could independently predispose patients to bacterial infections.
To further explore a connection between NAFLD and infection risk, the researchers looked at data for 12,133 Swedish adults with simple steatosis, nonfibrotic steatohepatitis, noncirrhotic fibrosis, or cirrhosis caused by NAFLD confirmed by liver biopsies performed between 1969 and 2017.
Each patient was matched to five or more contemporary controls from the general population by age, sex, and region of residence. The authors conducted an additional analysis that also adjusted for education, country of birth, and baseline clinical comorbidities, including diabetes, obesity, dyslipidemia, and hypertension, as well as hospitalization preceding the biopsy and chronic obstructive pulmonary disease.
The primary endpoint was severe infections requiring hospital admission. Secondary endpoints included seven prespecified infection subgroups: sepsis; respiratory tract; most gastrointestinal infections; bacterial peritonitis; urogenital; muscle, skin, and soft tissue; and other infections.
Elevated risk at all NAFLD stages
Dr. Ebrahimi and colleagues found that over a median follow-up of 14 years, patients with NAFLD had a higher incidence of severe infections – most often respiratory or urinary tract infections – compared with those without NAFLD (32% vs. 17%, respectively).
Biopsy-confirmed NAFLD was also associated with a 71% higher hazard and a 20-year absolute excess risk of 17.3% for severe infections requiring hospital admission versus comparators. The elevated risk showed up in patients with steatosis and increased with the severity of NAFLD. Simple steatosis saw a 64% higher risk (adjusted hazard ratio, 1.64; 95% confidence interval, 1.55-1.73), whereas patients with cirrhosis saw a more than twofold higher risk, compared with controls (aHR, 2.32; 95% CI, 1.92-2.82).
When Dr. Ebrahimi and colleagues adjusted for parameters of the metabolic syndrome, they found an independent increased risk for severe infection. For patients with NAFLD, the increased risk may come from greater susceptibility to infections in general or to a more severe course of infections.
“Our study clearly demonstrates the complexity and high disease burden associated with NAFLD,” Dr. Ebrahimi said. “We are beginning to understand the different layers involved and will eventually move away from a liver-centric view to a more holistic view of the disease.”
Clinicians caring for patients with NAFLD need to be aware of the increased risk for infection, Dr. Ebrahimi said. They also should assess their patients’ vaccination status, and seek to control modifiable risk factors, such as diabetes.
Nancy Reau, MD, of Rush University, Chicago, described the study’s message as important.
“Patients with NAFLD and advancing liver disease are at risk for severe infections,” Dr. Reau said. “When we consider the fact that patients with advanced liver disease tend to die from infectious complications, awareness leading to early recognition and efficient treatment is imperative.”
The authors acknowledged the following limitations: only severe infections requiring hospitalization could be captured; whether infection led to decompensation or vice versa among patients with cirrhosis could not be determined; and detailed data on smoking, alcohol, vaccinations, body mass, and other potentially relevant measures were not available.
The Swiss National Science Foundation, Syskonen Svensson Foundation, and Bengt Ihre Foundation provided grants to Dr. Ebrahimi or coauthors. One coauthor disclosed previous research funding from Janssen and MSD. Dr. Reau disclosed receiving research support and consulting fees from AbbVie and Gilead, as well as consulting fees from Arbutus, Intercept, and Salix.
A version of this article first appeared on Medscape.com.
People with nonalcoholic fatty liver disease (NAFLD) are more likely to develop severe infections requiring hospitalization, according to findings from a large Swedish cohort study.
The increased risk was equal to one extra severe infection in every six patients with NAFLD by 20 years after diagnosis, wrote Fahim Ebrahimi, MD, of the Karolinska Institute in Stockholm, and coauthors.
“Accumulating evidence suggests that NAFLD can affect multiple organ systems, which is not surprising, as the liver has multiple functions – regulating metabolism and being a central organ of the immune system,” Dr. Ebrahimi said in an interview.
The study was published online in Clinical Gastroenterology and Hepatology.
“Up to a fifth of cells in the liver are immune cells that process numerous antigens and pathogens from the gastrointestinal tract,” Dr. Ebrahimi noted. “We were intrigued by experimental studies showing that, in NAFLD, many of these key immune cells become dysfunctional at various levels, which may affect disease progression, but at the same time also increase the susceptibility to viral, bacterial, and fungal infections.”
Patients with NAFLD have metabolic risk factors known to increase infection risk, but a smaller study by a different research group had found that NAFLD could independently predispose patients to bacterial infections.
To further explore a connection between NAFLD and infection risk, the researchers looked at data for 12,133 Swedish adults with simple steatosis, nonfibrotic steatohepatitis, noncirrhotic fibrosis, or cirrhosis caused by NAFLD confirmed by liver biopsies performed between 1969 and 2017.
Each patient was matched to five or more contemporary controls from the general population by age, sex, and region of residence. The authors conducted an additional analysis that also adjusted for education, country of birth, and baseline clinical comorbidities, including diabetes, obesity, dyslipidemia, and hypertension, as well as hospitalization preceding the biopsy and chronic obstructive pulmonary disease.
The primary endpoint was severe infections requiring hospital admission. Secondary endpoints included seven prespecified infection subgroups: sepsis; respiratory tract; most gastrointestinal infections; bacterial peritonitis; urogenital; muscle, skin, and soft tissue; and other infections.
Elevated risk at all NAFLD stages
Dr. Ebrahimi and colleagues found that over a median follow-up of 14 years, patients with NAFLD had a higher incidence of severe infections – most often respiratory or urinary tract infections – compared with those without NAFLD (32% vs. 17%, respectively).
Biopsy-confirmed NAFLD was also associated with a 71% higher hazard and a 20-year absolute excess risk of 17.3% for severe infections requiring hospital admission versus comparators. The elevated risk showed up in patients with steatosis and increased with the severity of NAFLD. Simple steatosis saw a 64% higher risk (adjusted hazard ratio, 1.64; 95% confidence interval, 1.55-1.73), whereas patients with cirrhosis saw a more than twofold higher risk, compared with controls (aHR, 2.32; 95% CI, 1.92-2.82).
When Dr. Ebrahimi and colleagues adjusted for parameters of the metabolic syndrome, they found an independent increased risk for severe infection. For patients with NAFLD, the increased risk may come from greater susceptibility to infections in general or to a more severe course of infections.
“Our study clearly demonstrates the complexity and high disease burden associated with NAFLD,” Dr. Ebrahimi said. “We are beginning to understand the different layers involved and will eventually move away from a liver-centric view to a more holistic view of the disease.”
Clinicians caring for patients with NAFLD need to be aware of the increased risk for infection, Dr. Ebrahimi said. They also should assess their patients’ vaccination status, and seek to control modifiable risk factors, such as diabetes.
Nancy Reau, MD, of Rush University, Chicago, described the study’s message as important.
“Patients with NAFLD and advancing liver disease are at risk for severe infections,” Dr. Reau said. “When we consider the fact that patients with advanced liver disease tend to die from infectious complications, awareness leading to early recognition and efficient treatment is imperative.”
The authors acknowledged the following limitations: only severe infections requiring hospitalization could be captured; whether infection led to decompensation or vice versa among patients with cirrhosis could not be determined; and detailed data on smoking, alcohol, vaccinations, body mass, and other potentially relevant measures were not available.
The Swiss National Science Foundation, Syskonen Svensson Foundation, and Bengt Ihre Foundation provided grants to Dr. Ebrahimi or coauthors. One coauthor disclosed previous research funding from Janssen and MSD. Dr. Reau disclosed receiving research support and consulting fees from AbbVie and Gilead, as well as consulting fees from Arbutus, Intercept, and Salix.
A version of this article first appeared on Medscape.com.
People with nonalcoholic fatty liver disease (NAFLD) are more likely to develop severe infections requiring hospitalization, according to findings from a large Swedish cohort study.
The increased risk was equal to one extra severe infection in every six patients with NAFLD by 20 years after diagnosis, wrote Fahim Ebrahimi, MD, of the Karolinska Institute in Stockholm, and coauthors.
“Accumulating evidence suggests that NAFLD can affect multiple organ systems, which is not surprising, as the liver has multiple functions – regulating metabolism and being a central organ of the immune system,” Dr. Ebrahimi said in an interview.
The study was published online in Clinical Gastroenterology and Hepatology.
“Up to a fifth of cells in the liver are immune cells that process numerous antigens and pathogens from the gastrointestinal tract,” Dr. Ebrahimi noted. “We were intrigued by experimental studies showing that, in NAFLD, many of these key immune cells become dysfunctional at various levels, which may affect disease progression, but at the same time also increase the susceptibility to viral, bacterial, and fungal infections.”
Patients with NAFLD have metabolic risk factors known to increase infection risk, but a smaller study by a different research group had found that NAFLD could independently predispose patients to bacterial infections.
To further explore a connection between NAFLD and infection risk, the researchers looked at data for 12,133 Swedish adults with simple steatosis, nonfibrotic steatohepatitis, noncirrhotic fibrosis, or cirrhosis caused by NAFLD confirmed by liver biopsies performed between 1969 and 2017.
Each patient was matched to five or more contemporary controls from the general population by age, sex, and region of residence. The authors conducted an additional analysis that also adjusted for education, country of birth, and baseline clinical comorbidities, including diabetes, obesity, dyslipidemia, and hypertension, as well as hospitalization preceding the biopsy and chronic obstructive pulmonary disease.
The primary endpoint was severe infections requiring hospital admission. Secondary endpoints included seven prespecified infection subgroups: sepsis; respiratory tract; most gastrointestinal infections; bacterial peritonitis; urogenital; muscle, skin, and soft tissue; and other infections.
Elevated risk at all NAFLD stages
Dr. Ebrahimi and colleagues found that over a median follow-up of 14 years, patients with NAFLD had a higher incidence of severe infections – most often respiratory or urinary tract infections – compared with those without NAFLD (32% vs. 17%, respectively).
Biopsy-confirmed NAFLD was also associated with a 71% higher hazard and a 20-year absolute excess risk of 17.3% for severe infections requiring hospital admission versus comparators. The elevated risk showed up in patients with steatosis and increased with the severity of NAFLD. Simple steatosis saw a 64% higher risk (adjusted hazard ratio, 1.64; 95% confidence interval, 1.55-1.73), whereas patients with cirrhosis saw a more than twofold higher risk, compared with controls (aHR, 2.32; 95% CI, 1.92-2.82).
When Dr. Ebrahimi and colleagues adjusted for parameters of the metabolic syndrome, they found an independent increased risk for severe infection. For patients with NAFLD, the increased risk may come from greater susceptibility to infections in general or to a more severe course of infections.
“Our study clearly demonstrates the complexity and high disease burden associated with NAFLD,” Dr. Ebrahimi said. “We are beginning to understand the different layers involved and will eventually move away from a liver-centric view to a more holistic view of the disease.”
Clinicians caring for patients with NAFLD need to be aware of the increased risk for infection, Dr. Ebrahimi said. They also should assess their patients’ vaccination status, and seek to control modifiable risk factors, such as diabetes.
Nancy Reau, MD, of Rush University, Chicago, described the study’s message as important.
“Patients with NAFLD and advancing liver disease are at risk for severe infections,” Dr. Reau said. “When we consider the fact that patients with advanced liver disease tend to die from infectious complications, awareness leading to early recognition and efficient treatment is imperative.”
The authors acknowledged the following limitations: only severe infections requiring hospitalization could be captured; whether infection led to decompensation or vice versa among patients with cirrhosis could not be determined; and detailed data on smoking, alcohol, vaccinations, body mass, and other potentially relevant measures were not available.
The Swiss National Science Foundation, Syskonen Svensson Foundation, and Bengt Ihre Foundation provided grants to Dr. Ebrahimi or coauthors. One coauthor disclosed previous research funding from Janssen and MSD. Dr. Reau disclosed receiving research support and consulting fees from AbbVie and Gilead, as well as consulting fees from Arbutus, Intercept, and Salix.
A version of this article first appeared on Medscape.com.
FROM CLINICAL GASTROENTEROLOGY AND HEPATOLOGY
Study explains link between fatty liver and CRC liver metastasis
according to the authors of new research.
These findings support the previously reported link between fatty liver and colorectal cancer (CRC) liver metastasis, and suggest that CRC patients with nonalcoholic fatty liver disease (NAFLD) may respond differently to treatment than CRC patients without NAFLD, wrote lead author Zhijun Wang, MD, PhD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues, in their paper.
“Obesity and NAFLD are the significant risk factors for CRC,” the investigators explained, in Cell Metabolism. “A growing body of epidemiological evidence indicates that fatty liver increases the occurrence of CRC liver metastasis and the local recurrence after resection of CRC liver metastases, thereby worsening prognosis ... There is an urgent need to understand the molecular mechanisms of metastasis in patients with fatty liver to manage those patients effectively.”
To this end, Dr. Wang and colleagues conducted a series of experiments involving mice, cell cultures, and human sera. They found that fatty liver increases risk of CRC liver metastasis via extracellular vesicles (EVs) that contain procarcinogenic miRNAs. As these EVs transfer microRNAs from fatty liver hepatocytes to metastatic cancer cells, YAP activity increases, which, in turn, suppresses immune activity within the tumor microenvironment, promoting growth of CRC metastasis.
Beyond the increased risk of liver metastasis presented by fatty liver, the investigators suggested that NAFLD may cause “more complex” metastatic tumor microenvironments, potentially explaining “diverse responses” to cancer therapies among patients with CRC and liver metastases.
“In summary, our study demonstrates that the pre- and prometastatic liver environment of fatty liver is induced by procarcinogenic EVs and results in an immunosuppressive tumor microenvironemnt, in which YAP plays an important role,” the investigators concluded. “Our study provides new insight into [the] distinct liver tumor microenvironment in patients with fatty liver and without fatty liver, which may contribute to the aggressiveness of metastatic tumors and weak responses to anticancer therapy in patients with fatty liver. Additional studies are warranted to develop precision medicine for treating patients with CRC and liver metastasis.”
One of the study authors disclosed relationships with Altimmune, Cytodyn, Novo Nordisk, and others. The other investigators had no relevant financial disclosures.
according to the authors of new research.
These findings support the previously reported link between fatty liver and colorectal cancer (CRC) liver metastasis, and suggest that CRC patients with nonalcoholic fatty liver disease (NAFLD) may respond differently to treatment than CRC patients without NAFLD, wrote lead author Zhijun Wang, MD, PhD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues, in their paper.
“Obesity and NAFLD are the significant risk factors for CRC,” the investigators explained, in Cell Metabolism. “A growing body of epidemiological evidence indicates that fatty liver increases the occurrence of CRC liver metastasis and the local recurrence after resection of CRC liver metastases, thereby worsening prognosis ... There is an urgent need to understand the molecular mechanisms of metastasis in patients with fatty liver to manage those patients effectively.”
To this end, Dr. Wang and colleagues conducted a series of experiments involving mice, cell cultures, and human sera. They found that fatty liver increases risk of CRC liver metastasis via extracellular vesicles (EVs) that contain procarcinogenic miRNAs. As these EVs transfer microRNAs from fatty liver hepatocytes to metastatic cancer cells, YAP activity increases, which, in turn, suppresses immune activity within the tumor microenvironment, promoting growth of CRC metastasis.
Beyond the increased risk of liver metastasis presented by fatty liver, the investigators suggested that NAFLD may cause “more complex” metastatic tumor microenvironments, potentially explaining “diverse responses” to cancer therapies among patients with CRC and liver metastases.
“In summary, our study demonstrates that the pre- and prometastatic liver environment of fatty liver is induced by procarcinogenic EVs and results in an immunosuppressive tumor microenvironemnt, in which YAP plays an important role,” the investigators concluded. “Our study provides new insight into [the] distinct liver tumor microenvironment in patients with fatty liver and without fatty liver, which may contribute to the aggressiveness of metastatic tumors and weak responses to anticancer therapy in patients with fatty liver. Additional studies are warranted to develop precision medicine for treating patients with CRC and liver metastasis.”
One of the study authors disclosed relationships with Altimmune, Cytodyn, Novo Nordisk, and others. The other investigators had no relevant financial disclosures.
according to the authors of new research.
These findings support the previously reported link between fatty liver and colorectal cancer (CRC) liver metastasis, and suggest that CRC patients with nonalcoholic fatty liver disease (NAFLD) may respond differently to treatment than CRC patients without NAFLD, wrote lead author Zhijun Wang, MD, PhD, of Cedars-Sinai Medical Center, Los Angeles, and colleagues, in their paper.
“Obesity and NAFLD are the significant risk factors for CRC,” the investigators explained, in Cell Metabolism. “A growing body of epidemiological evidence indicates that fatty liver increases the occurrence of CRC liver metastasis and the local recurrence after resection of CRC liver metastases, thereby worsening prognosis ... There is an urgent need to understand the molecular mechanisms of metastasis in patients with fatty liver to manage those patients effectively.”
To this end, Dr. Wang and colleagues conducted a series of experiments involving mice, cell cultures, and human sera. They found that fatty liver increases risk of CRC liver metastasis via extracellular vesicles (EVs) that contain procarcinogenic miRNAs. As these EVs transfer microRNAs from fatty liver hepatocytes to metastatic cancer cells, YAP activity increases, which, in turn, suppresses immune activity within the tumor microenvironment, promoting growth of CRC metastasis.
Beyond the increased risk of liver metastasis presented by fatty liver, the investigators suggested that NAFLD may cause “more complex” metastatic tumor microenvironments, potentially explaining “diverse responses” to cancer therapies among patients with CRC and liver metastases.
“In summary, our study demonstrates that the pre- and prometastatic liver environment of fatty liver is induced by procarcinogenic EVs and results in an immunosuppressive tumor microenvironemnt, in which YAP plays an important role,” the investigators concluded. “Our study provides new insight into [the] distinct liver tumor microenvironment in patients with fatty liver and without fatty liver, which may contribute to the aggressiveness of metastatic tumors and weak responses to anticancer therapy in patients with fatty liver. Additional studies are warranted to develop precision medicine for treating patients with CRC and liver metastasis.”
One of the study authors disclosed relationships with Altimmune, Cytodyn, Novo Nordisk, and others. The other investigators had no relevant financial disclosures.
FROM CELL METABOLISM
Liver disease does not worsen IVF outcomes
Having chronic liver disease does not affect outcomes for women who undergo in vitro fertilization (IVF), new research suggests.
The study, published online in the American Journal of Gastroenterology, compared women with and those without chronic liver disease who had normal ovarian reserve and who underwent assisted reproductive technology (ART) treatment in a high-volume fertility practice from 2002 to 2021.
“IVF treatment and pregnancy outcomes were not significantly different compared to controls,” the researchers wrote.
Women with liver disease may experience impaired fertility. For example, women with chronic liver disease, such as hepatitis C virus infection, may have premature ovarian insufficiency, while women with advanced liver disease, cirrhosis, and hepatic decompensation are known to have abnormally low gonadotropin levels.
leading to “an immediate need for the clinical assessment of reproductive potential in women with chronic liver disease,” the authors wrote.
The literature about ART treatment outcomes for women with liver disease was limited and may not reflect current therapy protocols, researchers found.
“To the best of our knowledge, this study is the largest to date to evaluate IVF efficacy in women with liver disease,” they wrote.
Similar outcomes
Researchers identified 295 women with liver disease (mean age, 37.8 ± 5.2 years) who underwent 1,033 contemporary, standard ART treatment cycles. Six patients (2%) had cirrhosis, eight (2.7%) had undergone liver transplantation, and 281 (95.3%) had chronic liver disease, of which viral hepatitis B and C infections were the most prevalent. The final study population consisted of 115 women who underwent 186 IVF cycles, as well as embryo biopsy for genetic testing.
The control group consisted of all the women at the treatment center without liver disease who received contemporary, standard ART treatment because of male factor infertility, which served as an indication that the women had normal ovarian reserve and were considered fertile. These 624 patients underwent 868 IVF cycles with embryo biopsy.
The mean age of the patients with liver disease was significantly higher than that of the control participants. Mean body mass index was also significantly higher for the patients with liver disease, and there were differences in baseline levels of selected hormones, compared with control participants. In addition, among those with liver disease, the number of oocytes retrieved was significantly lower (12.3 ± 7.6 vs. 16.5 ± 8.2; P < .05), as were the number of mature oocytes (9.1 ± 6.2 vs. 12.6 ± 6.7; P < .05), the number of fertilized embryos (7.0 ± 5.2 vs. 9.9 ± 5.9; P < .05), the number of embryos for which biopsy was performed (3.4 ± 2.2 vs. 5.1 ± 3.5; P < .05), and the number of euploid embryos (1.6 ± 1.4 vs. 2.7 ± 2.4; P < .05), compared with control participants.
Among the two groups, there were no statistically significant differences in mature oocyte rate (an indicator of response to controlled ovarian stimulation), fertilization per mature oocyte rate (an indicator of oocyte quality and ability to be fertilized), or embryo ploidy rate (an indicator of genetically normal embryos), as determined by embryo biopsy, the researchers write.
A subanalysis of women who went on to have a single thawed euploid (chromosomally normal) embryo transfer to achieve pregnancy found no statistically significant differences in rates of clinical pregnancy, clinical pregnancy loss, or live births between the liver disease group and the control group.
“Overall, women with chronic liver disease can be counseled that IVF treatment will not significantly differ in response to controlled ovarian stimulation, embryo fertilization rate, or ploidy outcome compared to women without liver disease,” the researchers wrote.
Data for patient counseling
The results could change the current common thinking among clinicians that IVF should not be conducted until liver disease is under optimal control, first author Jessica Lee, BS, a student at Icahn School of Medicine at Mount Sinai, New York, said.
“There was a knowledge gap for studies in the United States, and we hope this study will not only help patients with liver disease but also providers with counseling,” she said.
The findings suggest that “even if you have chronic liver disease and it’s not fully optimized, that should not interfere with pursuing IVF,” said principal investigator Tatyana Kushner, MD, an associate professor of medicine in liver diseases at the Icahn School.
Women with liver disease whose fertility is impaired should receive counseling about fertility preservation options earlier to help access fertility care, the researchers write.
The study’s findings are “encouraging,” said Monika Sarkar, MD, associate professor of medicine in gastroenterology at the University of California, San Francisco.
“With rising numbers of young adults with liver disease, it is encouraging to see dedicated studies that address a topic of importance to our patients – namely, their reproductive health,” she said. “The current study nicely expands beyond previous data to include a control population without liver disease.”
Differences in oocyte numbers
Although there were no differences in the success rate of embryo transfer, the researchers did see differences in the number of oocytes. Only 37 mature oocytes made it to transfer in the liver disease group, compared with 609 in the control group, noted Dr. Sarkar, who was not involved with the study.
“The challenge of ART is less at the level of embryo transfer, which is very successful once a euploid embryo is achieved, but rather at the earlier step of retrieval of mature oocytes,” Dr. Sarkar said. “Here, the authors found that patients with liver disease had a significantly lower number of oocytes retrieved, number of mature oocytes, and lower number of fertilized embryos.”
The data suggest that fewer eggs are retrieved per cycle from patients with liver disease, “which ultimately will lower the success per cycle,” Dr. Sarkar said.
“This suggests that referring women with chronic liver disease to ART sooner may help to optimize outcomes,” she added. “Larger data evaluating ability to achieve mature oocytes and subsequent fertilization will also be key for determining whether ART success differs by presence, severity, and type of liver disease.”
As more research on ART outcomes in women with liver disease is conducted, subspecialists in gastrointestinal and liver disease may gain confidence in counseling patients, Dr. Sarkar said.
Ms. Lee, Dr. Kushner, and Dr. Sarkar report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Having chronic liver disease does not affect outcomes for women who undergo in vitro fertilization (IVF), new research suggests.
The study, published online in the American Journal of Gastroenterology, compared women with and those without chronic liver disease who had normal ovarian reserve and who underwent assisted reproductive technology (ART) treatment in a high-volume fertility practice from 2002 to 2021.
“IVF treatment and pregnancy outcomes were not significantly different compared to controls,” the researchers wrote.
Women with liver disease may experience impaired fertility. For example, women with chronic liver disease, such as hepatitis C virus infection, may have premature ovarian insufficiency, while women with advanced liver disease, cirrhosis, and hepatic decompensation are known to have abnormally low gonadotropin levels.
leading to “an immediate need for the clinical assessment of reproductive potential in women with chronic liver disease,” the authors wrote.
The literature about ART treatment outcomes for women with liver disease was limited and may not reflect current therapy protocols, researchers found.
“To the best of our knowledge, this study is the largest to date to evaluate IVF efficacy in women with liver disease,” they wrote.
Similar outcomes
Researchers identified 295 women with liver disease (mean age, 37.8 ± 5.2 years) who underwent 1,033 contemporary, standard ART treatment cycles. Six patients (2%) had cirrhosis, eight (2.7%) had undergone liver transplantation, and 281 (95.3%) had chronic liver disease, of which viral hepatitis B and C infections were the most prevalent. The final study population consisted of 115 women who underwent 186 IVF cycles, as well as embryo biopsy for genetic testing.
The control group consisted of all the women at the treatment center without liver disease who received contemporary, standard ART treatment because of male factor infertility, which served as an indication that the women had normal ovarian reserve and were considered fertile. These 624 patients underwent 868 IVF cycles with embryo biopsy.
The mean age of the patients with liver disease was significantly higher than that of the control participants. Mean body mass index was also significantly higher for the patients with liver disease, and there were differences in baseline levels of selected hormones, compared with control participants. In addition, among those with liver disease, the number of oocytes retrieved was significantly lower (12.3 ± 7.6 vs. 16.5 ± 8.2; P < .05), as were the number of mature oocytes (9.1 ± 6.2 vs. 12.6 ± 6.7; P < .05), the number of fertilized embryos (7.0 ± 5.2 vs. 9.9 ± 5.9; P < .05), the number of embryos for which biopsy was performed (3.4 ± 2.2 vs. 5.1 ± 3.5; P < .05), and the number of euploid embryos (1.6 ± 1.4 vs. 2.7 ± 2.4; P < .05), compared with control participants.
Among the two groups, there were no statistically significant differences in mature oocyte rate (an indicator of response to controlled ovarian stimulation), fertilization per mature oocyte rate (an indicator of oocyte quality and ability to be fertilized), or embryo ploidy rate (an indicator of genetically normal embryos), as determined by embryo biopsy, the researchers write.
A subanalysis of women who went on to have a single thawed euploid (chromosomally normal) embryo transfer to achieve pregnancy found no statistically significant differences in rates of clinical pregnancy, clinical pregnancy loss, or live births between the liver disease group and the control group.
“Overall, women with chronic liver disease can be counseled that IVF treatment will not significantly differ in response to controlled ovarian stimulation, embryo fertilization rate, or ploidy outcome compared to women without liver disease,” the researchers wrote.
Data for patient counseling
The results could change the current common thinking among clinicians that IVF should not be conducted until liver disease is under optimal control, first author Jessica Lee, BS, a student at Icahn School of Medicine at Mount Sinai, New York, said.
“There was a knowledge gap for studies in the United States, and we hope this study will not only help patients with liver disease but also providers with counseling,” she said.
The findings suggest that “even if you have chronic liver disease and it’s not fully optimized, that should not interfere with pursuing IVF,” said principal investigator Tatyana Kushner, MD, an associate professor of medicine in liver diseases at the Icahn School.
Women with liver disease whose fertility is impaired should receive counseling about fertility preservation options earlier to help access fertility care, the researchers write.
The study’s findings are “encouraging,” said Monika Sarkar, MD, associate professor of medicine in gastroenterology at the University of California, San Francisco.
“With rising numbers of young adults with liver disease, it is encouraging to see dedicated studies that address a topic of importance to our patients – namely, their reproductive health,” she said. “The current study nicely expands beyond previous data to include a control population without liver disease.”
Differences in oocyte numbers
Although there were no differences in the success rate of embryo transfer, the researchers did see differences in the number of oocytes. Only 37 mature oocytes made it to transfer in the liver disease group, compared with 609 in the control group, noted Dr. Sarkar, who was not involved with the study.
“The challenge of ART is less at the level of embryo transfer, which is very successful once a euploid embryo is achieved, but rather at the earlier step of retrieval of mature oocytes,” Dr. Sarkar said. “Here, the authors found that patients with liver disease had a significantly lower number of oocytes retrieved, number of mature oocytes, and lower number of fertilized embryos.”
The data suggest that fewer eggs are retrieved per cycle from patients with liver disease, “which ultimately will lower the success per cycle,” Dr. Sarkar said.
“This suggests that referring women with chronic liver disease to ART sooner may help to optimize outcomes,” she added. “Larger data evaluating ability to achieve mature oocytes and subsequent fertilization will also be key for determining whether ART success differs by presence, severity, and type of liver disease.”
As more research on ART outcomes in women with liver disease is conducted, subspecialists in gastrointestinal and liver disease may gain confidence in counseling patients, Dr. Sarkar said.
Ms. Lee, Dr. Kushner, and Dr. Sarkar report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
Having chronic liver disease does not affect outcomes for women who undergo in vitro fertilization (IVF), new research suggests.
The study, published online in the American Journal of Gastroenterology, compared women with and those without chronic liver disease who had normal ovarian reserve and who underwent assisted reproductive technology (ART) treatment in a high-volume fertility practice from 2002 to 2021.
“IVF treatment and pregnancy outcomes were not significantly different compared to controls,” the researchers wrote.
Women with liver disease may experience impaired fertility. For example, women with chronic liver disease, such as hepatitis C virus infection, may have premature ovarian insufficiency, while women with advanced liver disease, cirrhosis, and hepatic decompensation are known to have abnormally low gonadotropin levels.
leading to “an immediate need for the clinical assessment of reproductive potential in women with chronic liver disease,” the authors wrote.
The literature about ART treatment outcomes for women with liver disease was limited and may not reflect current therapy protocols, researchers found.
“To the best of our knowledge, this study is the largest to date to evaluate IVF efficacy in women with liver disease,” they wrote.
Similar outcomes
Researchers identified 295 women with liver disease (mean age, 37.8 ± 5.2 years) who underwent 1,033 contemporary, standard ART treatment cycles. Six patients (2%) had cirrhosis, eight (2.7%) had undergone liver transplantation, and 281 (95.3%) had chronic liver disease, of which viral hepatitis B and C infections were the most prevalent. The final study population consisted of 115 women who underwent 186 IVF cycles, as well as embryo biopsy for genetic testing.
The control group consisted of all the women at the treatment center without liver disease who received contemporary, standard ART treatment because of male factor infertility, which served as an indication that the women had normal ovarian reserve and were considered fertile. These 624 patients underwent 868 IVF cycles with embryo biopsy.
The mean age of the patients with liver disease was significantly higher than that of the control participants. Mean body mass index was also significantly higher for the patients with liver disease, and there were differences in baseline levels of selected hormones, compared with control participants. In addition, among those with liver disease, the number of oocytes retrieved was significantly lower (12.3 ± 7.6 vs. 16.5 ± 8.2; P < .05), as were the number of mature oocytes (9.1 ± 6.2 vs. 12.6 ± 6.7; P < .05), the number of fertilized embryos (7.0 ± 5.2 vs. 9.9 ± 5.9; P < .05), the number of embryos for which biopsy was performed (3.4 ± 2.2 vs. 5.1 ± 3.5; P < .05), and the number of euploid embryos (1.6 ± 1.4 vs. 2.7 ± 2.4; P < .05), compared with control participants.
Among the two groups, there were no statistically significant differences in mature oocyte rate (an indicator of response to controlled ovarian stimulation), fertilization per mature oocyte rate (an indicator of oocyte quality and ability to be fertilized), or embryo ploidy rate (an indicator of genetically normal embryos), as determined by embryo biopsy, the researchers write.
A subanalysis of women who went on to have a single thawed euploid (chromosomally normal) embryo transfer to achieve pregnancy found no statistically significant differences in rates of clinical pregnancy, clinical pregnancy loss, or live births between the liver disease group and the control group.
“Overall, women with chronic liver disease can be counseled that IVF treatment will not significantly differ in response to controlled ovarian stimulation, embryo fertilization rate, or ploidy outcome compared to women without liver disease,” the researchers wrote.
Data for patient counseling
The results could change the current common thinking among clinicians that IVF should not be conducted until liver disease is under optimal control, first author Jessica Lee, BS, a student at Icahn School of Medicine at Mount Sinai, New York, said.
“There was a knowledge gap for studies in the United States, and we hope this study will not only help patients with liver disease but also providers with counseling,” she said.
The findings suggest that “even if you have chronic liver disease and it’s not fully optimized, that should not interfere with pursuing IVF,” said principal investigator Tatyana Kushner, MD, an associate professor of medicine in liver diseases at the Icahn School.
Women with liver disease whose fertility is impaired should receive counseling about fertility preservation options earlier to help access fertility care, the researchers write.
The study’s findings are “encouraging,” said Monika Sarkar, MD, associate professor of medicine in gastroenterology at the University of California, San Francisco.
“With rising numbers of young adults with liver disease, it is encouraging to see dedicated studies that address a topic of importance to our patients – namely, their reproductive health,” she said. “The current study nicely expands beyond previous data to include a control population without liver disease.”
Differences in oocyte numbers
Although there were no differences in the success rate of embryo transfer, the researchers did see differences in the number of oocytes. Only 37 mature oocytes made it to transfer in the liver disease group, compared with 609 in the control group, noted Dr. Sarkar, who was not involved with the study.
“The challenge of ART is less at the level of embryo transfer, which is very successful once a euploid embryo is achieved, but rather at the earlier step of retrieval of mature oocytes,” Dr. Sarkar said. “Here, the authors found that patients with liver disease had a significantly lower number of oocytes retrieved, number of mature oocytes, and lower number of fertilized embryos.”
The data suggest that fewer eggs are retrieved per cycle from patients with liver disease, “which ultimately will lower the success per cycle,” Dr. Sarkar said.
“This suggests that referring women with chronic liver disease to ART sooner may help to optimize outcomes,” she added. “Larger data evaluating ability to achieve mature oocytes and subsequent fertilization will also be key for determining whether ART success differs by presence, severity, and type of liver disease.”
As more research on ART outcomes in women with liver disease is conducted, subspecialists in gastrointestinal and liver disease may gain confidence in counseling patients, Dr. Sarkar said.
Ms. Lee, Dr. Kushner, and Dr. Sarkar report no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM THE AMERICAN JOURNAL OF GASTROENTEROLOGY
Dried blood spot test validated for HIV, hep B, and hep C
A test that uses a single drop of dried blood to detect HIV, hepatitis B virus, and HCV has been validated and is now in use in some high-risk settings in Denmark, according to research presented at the annual European Congress of Clinical Microbiology & Infectious Diseases.
Molecular biologist Stephen Nilsson-Møller, MSc, and colleagues at the department of clinical microbiology, Copenhagen University Hospital, developed and validated the test, known as the Dried Blood Spot (DBS), for HIV, HBV, and HCV.
The “test that can detect low viral loads for all three viruses from a single drop of blood, and can be done using existing hospital equipment,” Mr. Nilsson-Møller said in an interview. “Importantly, it does not require venipuncture, but can be done from a drop of dried blood from the finger.”
He highlighted the utility of the new test in more challenging settings. “This method is particularly useful in high-risk settings such as homeless shelters, drug rehabilitation centers, and prisons, where needles might be misused, and it can be difficult to convince people to have the more invasive test.”
“Also, in some places – such as in low- and middle-income settings – there is a distinct risk of ruining blood samples before analysis due to limited refrigeration for transit and storage,” he added. “[Standard] blood samples need to be analyzed within 6 hours when kept at room temperature, while dried blood spots can last for 9 months at room temperature and can be mailed to a laboratory with the right equipment to analyze it.”
Tiny amounts of virus detected
Mr. Nilsson-Møller was tasked with developing a test for use by the university’s department of infectious diseases to screen people in high-risk settings in the capital region of Copenhagen. The work forms part of a PhD project by Jonas Demant at the University of Copenhagen, for which he is screening for HIV, HBV, and HCV in drug rehabilitation centers, prisons, and homeless shelters.
The study is the first to use the Hologic Panther system (a nucleic acid amplification test) combining all three viruses, Mr. Nilsson-Møller pointed out. “A tiny amount of virus can be detected because it is a very sensitive platform using transcription-mediated amplification.”
“If it detects low amounts of virus, it will create many copies very quickly, creating a signal that tells us that the sample is positive,” he explained.
The researchers collected whole blood from a finger prick, dried it out on a protein saver card (filter paper), and cut out a 1.2-cm diameter dry blood spot which was then prepared for analysis.
Twenty blood samples with known amounts of HIV, HBV, and HCV were analyzed via the DBS method (60 in total) and the viruses were detected in all of the samples.
To validate the method, the researchers used plasma with a known viral load, and a series of dilutions were performed to determine the lower limit for positive detection of all three viruses.
“Untreated patients typically have above 1 million IU/mL of viral loads in their plasma, and we found that we can detect much lower levels,” said Mr. Nilsson-Møller. “Ideally, 40 mcL of blood is good, but less should be sufficient if the test is on untreated patients.”
Early testing and treatment reduces morbidity and mortality
Elimination of HBV, HCV, and HIV by 2030 is a global health strategy set by the World Health Organization, but to meet this goal, new approaches for diagnostic testing are required. The DBS test for HIV, HBV, and HCV promises to make a significant contribution toward this goal.
“One in two people currently living with HIV is diagnosed late in the course of their infection, and an even larger proportion of the estimated 6 million Europeans living with chronic hepatitis B or C are not aware that they are infected,” said Anastasia Pharris, PhD, from the European Center for Disease Prevention and Control Principal Expert Infectious Diseases.
“Increasing testing coverage and uptake, especially for those most at risk, is an essential element of any strategy to eliminate HBV, HCV, and HIV in the European Union and European Economic Area,” she pointed out.
Dr. Pharris also highlighted that, while HIV, and often HBV infection, require lifelong treatment, HCV infection is now curable within a few weeks. “To maximize the benefits of individual treatment for all three infections, it is critical to test and diagnose people as soon as possible – in itself a challenge given that these infections can typically be asymptomatic for years.
“Early diagnosis of HBV, HCV, or HIV is vital as it allows people to access treatment, which significantly reduces associated long-term morbidity and mortality.
“In many cases, those most at risk of one of these infections are also more vulnerable to infection with one or both of the other viruses, making the argument for integrated testing even stronger,” she said in an interview.
Mr. Nilsson-Møller and Dr. Pharris reported no relevant financial relationships. Aptima kits for validation were provided by Hologic.
A version of this article first appeared on Medscape.com.
A test that uses a single drop of dried blood to detect HIV, hepatitis B virus, and HCV has been validated and is now in use in some high-risk settings in Denmark, according to research presented at the annual European Congress of Clinical Microbiology & Infectious Diseases.
Molecular biologist Stephen Nilsson-Møller, MSc, and colleagues at the department of clinical microbiology, Copenhagen University Hospital, developed and validated the test, known as the Dried Blood Spot (DBS), for HIV, HBV, and HCV.
The “test that can detect low viral loads for all three viruses from a single drop of blood, and can be done using existing hospital equipment,” Mr. Nilsson-Møller said in an interview. “Importantly, it does not require venipuncture, but can be done from a drop of dried blood from the finger.”
He highlighted the utility of the new test in more challenging settings. “This method is particularly useful in high-risk settings such as homeless shelters, drug rehabilitation centers, and prisons, where needles might be misused, and it can be difficult to convince people to have the more invasive test.”
“Also, in some places – such as in low- and middle-income settings – there is a distinct risk of ruining blood samples before analysis due to limited refrigeration for transit and storage,” he added. “[Standard] blood samples need to be analyzed within 6 hours when kept at room temperature, while dried blood spots can last for 9 months at room temperature and can be mailed to a laboratory with the right equipment to analyze it.”
Tiny amounts of virus detected
Mr. Nilsson-Møller was tasked with developing a test for use by the university’s department of infectious diseases to screen people in high-risk settings in the capital region of Copenhagen. The work forms part of a PhD project by Jonas Demant at the University of Copenhagen, for which he is screening for HIV, HBV, and HCV in drug rehabilitation centers, prisons, and homeless shelters.
The study is the first to use the Hologic Panther system (a nucleic acid amplification test) combining all three viruses, Mr. Nilsson-Møller pointed out. “A tiny amount of virus can be detected because it is a very sensitive platform using transcription-mediated amplification.”
“If it detects low amounts of virus, it will create many copies very quickly, creating a signal that tells us that the sample is positive,” he explained.
The researchers collected whole blood from a finger prick, dried it out on a protein saver card (filter paper), and cut out a 1.2-cm diameter dry blood spot which was then prepared for analysis.
Twenty blood samples with known amounts of HIV, HBV, and HCV were analyzed via the DBS method (60 in total) and the viruses were detected in all of the samples.
To validate the method, the researchers used plasma with a known viral load, and a series of dilutions were performed to determine the lower limit for positive detection of all three viruses.
“Untreated patients typically have above 1 million IU/mL of viral loads in their plasma, and we found that we can detect much lower levels,” said Mr. Nilsson-Møller. “Ideally, 40 mcL of blood is good, but less should be sufficient if the test is on untreated patients.”
Early testing and treatment reduces morbidity and mortality
Elimination of HBV, HCV, and HIV by 2030 is a global health strategy set by the World Health Organization, but to meet this goal, new approaches for diagnostic testing are required. The DBS test for HIV, HBV, and HCV promises to make a significant contribution toward this goal.
“One in two people currently living with HIV is diagnosed late in the course of their infection, and an even larger proportion of the estimated 6 million Europeans living with chronic hepatitis B or C are not aware that they are infected,” said Anastasia Pharris, PhD, from the European Center for Disease Prevention and Control Principal Expert Infectious Diseases.
“Increasing testing coverage and uptake, especially for those most at risk, is an essential element of any strategy to eliminate HBV, HCV, and HIV in the European Union and European Economic Area,” she pointed out.
Dr. Pharris also highlighted that, while HIV, and often HBV infection, require lifelong treatment, HCV infection is now curable within a few weeks. “To maximize the benefits of individual treatment for all three infections, it is critical to test and diagnose people as soon as possible – in itself a challenge given that these infections can typically be asymptomatic for years.
“Early diagnosis of HBV, HCV, or HIV is vital as it allows people to access treatment, which significantly reduces associated long-term morbidity and mortality.
“In many cases, those most at risk of one of these infections are also more vulnerable to infection with one or both of the other viruses, making the argument for integrated testing even stronger,” she said in an interview.
Mr. Nilsson-Møller and Dr. Pharris reported no relevant financial relationships. Aptima kits for validation were provided by Hologic.
A version of this article first appeared on Medscape.com.
A test that uses a single drop of dried blood to detect HIV, hepatitis B virus, and HCV has been validated and is now in use in some high-risk settings in Denmark, according to research presented at the annual European Congress of Clinical Microbiology & Infectious Diseases.
Molecular biologist Stephen Nilsson-Møller, MSc, and colleagues at the department of clinical microbiology, Copenhagen University Hospital, developed and validated the test, known as the Dried Blood Spot (DBS), for HIV, HBV, and HCV.
The “test that can detect low viral loads for all three viruses from a single drop of blood, and can be done using existing hospital equipment,” Mr. Nilsson-Møller said in an interview. “Importantly, it does not require venipuncture, but can be done from a drop of dried blood from the finger.”
He highlighted the utility of the new test in more challenging settings. “This method is particularly useful in high-risk settings such as homeless shelters, drug rehabilitation centers, and prisons, where needles might be misused, and it can be difficult to convince people to have the more invasive test.”
“Also, in some places – such as in low- and middle-income settings – there is a distinct risk of ruining blood samples before analysis due to limited refrigeration for transit and storage,” he added. “[Standard] blood samples need to be analyzed within 6 hours when kept at room temperature, while dried blood spots can last for 9 months at room temperature and can be mailed to a laboratory with the right equipment to analyze it.”
Tiny amounts of virus detected
Mr. Nilsson-Møller was tasked with developing a test for use by the university’s department of infectious diseases to screen people in high-risk settings in the capital region of Copenhagen. The work forms part of a PhD project by Jonas Demant at the University of Copenhagen, for which he is screening for HIV, HBV, and HCV in drug rehabilitation centers, prisons, and homeless shelters.
The study is the first to use the Hologic Panther system (a nucleic acid amplification test) combining all three viruses, Mr. Nilsson-Møller pointed out. “A tiny amount of virus can be detected because it is a very sensitive platform using transcription-mediated amplification.”
“If it detects low amounts of virus, it will create many copies very quickly, creating a signal that tells us that the sample is positive,” he explained.
The researchers collected whole blood from a finger prick, dried it out on a protein saver card (filter paper), and cut out a 1.2-cm diameter dry blood spot which was then prepared for analysis.
Twenty blood samples with known amounts of HIV, HBV, and HCV were analyzed via the DBS method (60 in total) and the viruses were detected in all of the samples.
To validate the method, the researchers used plasma with a known viral load, and a series of dilutions were performed to determine the lower limit for positive detection of all three viruses.
“Untreated patients typically have above 1 million IU/mL of viral loads in their plasma, and we found that we can detect much lower levels,” said Mr. Nilsson-Møller. “Ideally, 40 mcL of blood is good, but less should be sufficient if the test is on untreated patients.”
Early testing and treatment reduces morbidity and mortality
Elimination of HBV, HCV, and HIV by 2030 is a global health strategy set by the World Health Organization, but to meet this goal, new approaches for diagnostic testing are required. The DBS test for HIV, HBV, and HCV promises to make a significant contribution toward this goal.
“One in two people currently living with HIV is diagnosed late in the course of their infection, and an even larger proportion of the estimated 6 million Europeans living with chronic hepatitis B or C are not aware that they are infected,” said Anastasia Pharris, PhD, from the European Center for Disease Prevention and Control Principal Expert Infectious Diseases.
“Increasing testing coverage and uptake, especially for those most at risk, is an essential element of any strategy to eliminate HBV, HCV, and HIV in the European Union and European Economic Area,” she pointed out.
Dr. Pharris also highlighted that, while HIV, and often HBV infection, require lifelong treatment, HCV infection is now curable within a few weeks. “To maximize the benefits of individual treatment for all three infections, it is critical to test and diagnose people as soon as possible – in itself a challenge given that these infections can typically be asymptomatic for years.
“Early diagnosis of HBV, HCV, or HIV is vital as it allows people to access treatment, which significantly reduces associated long-term morbidity and mortality.
“In many cases, those most at risk of one of these infections are also more vulnerable to infection with one or both of the other viruses, making the argument for integrated testing even stronger,” she said in an interview.
Mr. Nilsson-Møller and Dr. Pharris reported no relevant financial relationships. Aptima kits for validation were provided by Hologic.
A version of this article first appeared on Medscape.com.
FROM ECCMID 2023
Racial disparities not found in chronic hepatitis B treatment initiation
Researchers studying differences in treatment initiation for chronic hepatitis B (CHB) among a large, multiracial cohort in North America did not find evidence of disparities by race or socioeconomic status.
.
That gap suggests that treatment guidelines need to be simplified and that efforts to increase hepatitis B virus (HBV) awareness and train more clinicians are needed to achieve the World Health Organization’s goal of eliminating HBV by 2030, the researchers write.
The Hepatitis B Research Network study was published online in JAMA Network Open.
The prevalence of CHB in the United States is estimated at 2.4 million. It disproportionately affects persons of Asian or African descent, the investigators note. Their study examined whether treatment initiation and outcomes differ between African American and Black, Asian, and White participants, as well as between African American and Black participants born in North America and East or West Africa.
The research involved 1,550 adult patients: 1,157 Asian American, 193 African American or Black (39 born in the United States, 90 in East Africa, 53 in West Africa, and 11 elsewhere), 157 White, and 43 who identified as being of “other races.” All had CHB but were not receiving antiviral treatment at enrollment.
Participants came from 20 centers in the United States and one in Canada. They underwent clinical and laboratory assessments and could receive anti-HBV treatment after they enrolled. Enrollment was from Jan. 14, 2011, to Jan. 28, 2018. Participants were followed at 12 and 24 weeks and every 24 weeks thereafter in the longitudinal cohort study by Mandana Khalili, MD, division of gastroenterology and hepatology, University of California, San Francisco, and colleagues.
Information on patients’ country of birth, duration of U.S. or Canadian residency, educational level, employment, insurance, prior antiviral treatment, family history of HBV or hepatocellular carcinoma (HCC), and mode of transmission were collected by research coordinators.
Treatment initiation
During the study period, slightly fewer than one-third (32.5%) of the participants initiated treatment. The incidences were 4.8 per 100 person-years in African American or Black participants, 9.9 per 100 person-years in Asian participants, 6.6 per 100 person-years in White participants, and 7.9 per 100 person-years in those of other races (P < .001).
A lower percentage of African American and Black participants (14%) met the American Association for the Study of Liver Diseases treatment criteria, compared with Asian (22%) and White (27%) participants (P = .01).
When the researchers compared cumulative probability of initiating treatment by race for those who met criteria for treatment, they found no significant differences by race.
At 72 weeks, initiation probability was 0.45 for African American and Black patients and 0.51 for Asian and White patients (P = .68). Similarly, among African American and Black participants who met treatment criteria, there were no significant differences in cumulative probability of treatment by region of birth.
The cumulative percentage of treatment initiation for those who met guideline-based criteria was 62%.
“Among participants with a treatment indication, treatment rates did not differ significantly by race, despite marked differences in educational level, income, and type of health care insurance across the racial groups,” the researchers write. “Moreover, race was not an independent estimator of treatment initiation when adjusting for known factors associated with a higher risk of adverse clinical outcomes, namely, HBV DNA, disease severity, sex, and age.”
Adverse liver outcomes (hepatic decompensation, HCC, liver transplant, and death) were rare and did not vary significantly by race, the researchers write.
One study limitation is that participants were linked to specialty liver clinics, so the findings may not be generalizable to patients who receive care in other settings, the authors note.
The results are “reassuring,” said senior author Anna S. Lok, MD, division of gastroenterology and hepatology at University of Michigan in Ann Arbor. However, she noted, study participants had already overcome barriers to receiving care at major academic centers.
“Once you get into the big academic liver centers, then maybe everything is equal, but in the real world, a lot of people don’t ever get to the big liver centers,” she said. The question becomes: “Are we serving only a portion of the patient population?”
Many factors drive the decision to undergo treatment, including the doctor’s opinion as to need and the patient’s desire to receive treatment, she said.
The study participants who were more likely to get treated were those with higher-level disease who had a stronger indication for treatment, Dr. Lok said.
Finding the disparities
Centers for Disease Control and Prevention statistics show that Black people are 3.9 times more likely to have CHB and 2.5 times more likely to die from it than White people, notes H. Nina Kim, MD, with the department of medicine, University of Washington, Seattle, in an accompanying invited commentary.
“The fact that we have not observed racial disparities in treatment initiation does not mean none exist; it means we have to look harder to find them,” she writes.
“We need to examine whether our guidelines for HBV treatment are so complex that it becomes the purview of specialists, thereby restricting access and deepening inequities,” Dr. Kim adds. “We should look closely at retention in care, the step that precedes treatment, and stratify this outcome by race and ethnicity.”
Primary care physicians in some regions might find it difficult to manage patients who have hepatitis B because they see so few of them, Dr. Lok noted.
Dr. Khalili has received grants and consulting fees from Gilead Sciences Inc and grants from Intercept Pharmaceuticals outside the submitted work. Dr. Lok has received grants from Target and consultant fees from Abbott, Ambys, Arbutus, Chroma, Clear B, Enanta, Enochian, GNI, GlaxoSmithKline, Eli Lilly, and Virion outside the submitted work. Coauthors have received grants, consulting fees, or personal fees from Bayer, Boston Scientific, Exact Sciences, Fujifilm Medical Sciences, Gilead Sciences, Glycotest, Redhill Biopharma, Target RWE, MedEd Design, Pontifax, Global Life, the Lynx Group, AstraZeneca, Eisai, Novartis Venture Fund, Grail, QED Therapeutics, Genentech, Hepion Pharmaceuticals, Roche, Abbott, AbbVie, and Pfizer. Dr. Kim has received grants from Gilead Sciences (paid to her institution) outside the submitted work.
A version of this article first appeared on Medscape.com.
Researchers studying differences in treatment initiation for chronic hepatitis B (CHB) among a large, multiracial cohort in North America did not find evidence of disparities by race or socioeconomic status.
.
That gap suggests that treatment guidelines need to be simplified and that efforts to increase hepatitis B virus (HBV) awareness and train more clinicians are needed to achieve the World Health Organization’s goal of eliminating HBV by 2030, the researchers write.
The Hepatitis B Research Network study was published online in JAMA Network Open.
The prevalence of CHB in the United States is estimated at 2.4 million. It disproportionately affects persons of Asian or African descent, the investigators note. Their study examined whether treatment initiation and outcomes differ between African American and Black, Asian, and White participants, as well as between African American and Black participants born in North America and East or West Africa.
The research involved 1,550 adult patients: 1,157 Asian American, 193 African American or Black (39 born in the United States, 90 in East Africa, 53 in West Africa, and 11 elsewhere), 157 White, and 43 who identified as being of “other races.” All had CHB but were not receiving antiviral treatment at enrollment.
Participants came from 20 centers in the United States and one in Canada. They underwent clinical and laboratory assessments and could receive anti-HBV treatment after they enrolled. Enrollment was from Jan. 14, 2011, to Jan. 28, 2018. Participants were followed at 12 and 24 weeks and every 24 weeks thereafter in the longitudinal cohort study by Mandana Khalili, MD, division of gastroenterology and hepatology, University of California, San Francisco, and colleagues.
Information on patients’ country of birth, duration of U.S. or Canadian residency, educational level, employment, insurance, prior antiviral treatment, family history of HBV or hepatocellular carcinoma (HCC), and mode of transmission were collected by research coordinators.
Treatment initiation
During the study period, slightly fewer than one-third (32.5%) of the participants initiated treatment. The incidences were 4.8 per 100 person-years in African American or Black participants, 9.9 per 100 person-years in Asian participants, 6.6 per 100 person-years in White participants, and 7.9 per 100 person-years in those of other races (P < .001).
A lower percentage of African American and Black participants (14%) met the American Association for the Study of Liver Diseases treatment criteria, compared with Asian (22%) and White (27%) participants (P = .01).
When the researchers compared cumulative probability of initiating treatment by race for those who met criteria for treatment, they found no significant differences by race.
At 72 weeks, initiation probability was 0.45 for African American and Black patients and 0.51 for Asian and White patients (P = .68). Similarly, among African American and Black participants who met treatment criteria, there were no significant differences in cumulative probability of treatment by region of birth.
The cumulative percentage of treatment initiation for those who met guideline-based criteria was 62%.
“Among participants with a treatment indication, treatment rates did not differ significantly by race, despite marked differences in educational level, income, and type of health care insurance across the racial groups,” the researchers write. “Moreover, race was not an independent estimator of treatment initiation when adjusting for known factors associated with a higher risk of adverse clinical outcomes, namely, HBV DNA, disease severity, sex, and age.”
Adverse liver outcomes (hepatic decompensation, HCC, liver transplant, and death) were rare and did not vary significantly by race, the researchers write.
One study limitation is that participants were linked to specialty liver clinics, so the findings may not be generalizable to patients who receive care in other settings, the authors note.
The results are “reassuring,” said senior author Anna S. Lok, MD, division of gastroenterology and hepatology at University of Michigan in Ann Arbor. However, she noted, study participants had already overcome barriers to receiving care at major academic centers.
“Once you get into the big academic liver centers, then maybe everything is equal, but in the real world, a lot of people don’t ever get to the big liver centers,” she said. The question becomes: “Are we serving only a portion of the patient population?”
Many factors drive the decision to undergo treatment, including the doctor’s opinion as to need and the patient’s desire to receive treatment, she said.
The study participants who were more likely to get treated were those with higher-level disease who had a stronger indication for treatment, Dr. Lok said.
Finding the disparities
Centers for Disease Control and Prevention statistics show that Black people are 3.9 times more likely to have CHB and 2.5 times more likely to die from it than White people, notes H. Nina Kim, MD, with the department of medicine, University of Washington, Seattle, in an accompanying invited commentary.
“The fact that we have not observed racial disparities in treatment initiation does not mean none exist; it means we have to look harder to find them,” she writes.
“We need to examine whether our guidelines for HBV treatment are so complex that it becomes the purview of specialists, thereby restricting access and deepening inequities,” Dr. Kim adds. “We should look closely at retention in care, the step that precedes treatment, and stratify this outcome by race and ethnicity.”
Primary care physicians in some regions might find it difficult to manage patients who have hepatitis B because they see so few of them, Dr. Lok noted.
Dr. Khalili has received grants and consulting fees from Gilead Sciences Inc and grants from Intercept Pharmaceuticals outside the submitted work. Dr. Lok has received grants from Target and consultant fees from Abbott, Ambys, Arbutus, Chroma, Clear B, Enanta, Enochian, GNI, GlaxoSmithKline, Eli Lilly, and Virion outside the submitted work. Coauthors have received grants, consulting fees, or personal fees from Bayer, Boston Scientific, Exact Sciences, Fujifilm Medical Sciences, Gilead Sciences, Glycotest, Redhill Biopharma, Target RWE, MedEd Design, Pontifax, Global Life, the Lynx Group, AstraZeneca, Eisai, Novartis Venture Fund, Grail, QED Therapeutics, Genentech, Hepion Pharmaceuticals, Roche, Abbott, AbbVie, and Pfizer. Dr. Kim has received grants from Gilead Sciences (paid to her institution) outside the submitted work.
A version of this article first appeared on Medscape.com.
Researchers studying differences in treatment initiation for chronic hepatitis B (CHB) among a large, multiracial cohort in North America did not find evidence of disparities by race or socioeconomic status.
.
That gap suggests that treatment guidelines need to be simplified and that efforts to increase hepatitis B virus (HBV) awareness and train more clinicians are needed to achieve the World Health Organization’s goal of eliminating HBV by 2030, the researchers write.
The Hepatitis B Research Network study was published online in JAMA Network Open.
The prevalence of CHB in the United States is estimated at 2.4 million. It disproportionately affects persons of Asian or African descent, the investigators note. Their study examined whether treatment initiation and outcomes differ between African American and Black, Asian, and White participants, as well as between African American and Black participants born in North America and East or West Africa.
The research involved 1,550 adult patients: 1,157 Asian American, 193 African American or Black (39 born in the United States, 90 in East Africa, 53 in West Africa, and 11 elsewhere), 157 White, and 43 who identified as being of “other races.” All had CHB but were not receiving antiviral treatment at enrollment.
Participants came from 20 centers in the United States and one in Canada. They underwent clinical and laboratory assessments and could receive anti-HBV treatment after they enrolled. Enrollment was from Jan. 14, 2011, to Jan. 28, 2018. Participants were followed at 12 and 24 weeks and every 24 weeks thereafter in the longitudinal cohort study by Mandana Khalili, MD, division of gastroenterology and hepatology, University of California, San Francisco, and colleagues.
Information on patients’ country of birth, duration of U.S. or Canadian residency, educational level, employment, insurance, prior antiviral treatment, family history of HBV or hepatocellular carcinoma (HCC), and mode of transmission were collected by research coordinators.
Treatment initiation
During the study period, slightly fewer than one-third (32.5%) of the participants initiated treatment. The incidences were 4.8 per 100 person-years in African American or Black participants, 9.9 per 100 person-years in Asian participants, 6.6 per 100 person-years in White participants, and 7.9 per 100 person-years in those of other races (P < .001).
A lower percentage of African American and Black participants (14%) met the American Association for the Study of Liver Diseases treatment criteria, compared with Asian (22%) and White (27%) participants (P = .01).
When the researchers compared cumulative probability of initiating treatment by race for those who met criteria for treatment, they found no significant differences by race.
At 72 weeks, initiation probability was 0.45 for African American and Black patients and 0.51 for Asian and White patients (P = .68). Similarly, among African American and Black participants who met treatment criteria, there were no significant differences in cumulative probability of treatment by region of birth.
The cumulative percentage of treatment initiation for those who met guideline-based criteria was 62%.
“Among participants with a treatment indication, treatment rates did not differ significantly by race, despite marked differences in educational level, income, and type of health care insurance across the racial groups,” the researchers write. “Moreover, race was not an independent estimator of treatment initiation when adjusting for known factors associated with a higher risk of adverse clinical outcomes, namely, HBV DNA, disease severity, sex, and age.”
Adverse liver outcomes (hepatic decompensation, HCC, liver transplant, and death) were rare and did not vary significantly by race, the researchers write.
One study limitation is that participants were linked to specialty liver clinics, so the findings may not be generalizable to patients who receive care in other settings, the authors note.
The results are “reassuring,” said senior author Anna S. Lok, MD, division of gastroenterology and hepatology at University of Michigan in Ann Arbor. However, she noted, study participants had already overcome barriers to receiving care at major academic centers.
“Once you get into the big academic liver centers, then maybe everything is equal, but in the real world, a lot of people don’t ever get to the big liver centers,” she said. The question becomes: “Are we serving only a portion of the patient population?”
Many factors drive the decision to undergo treatment, including the doctor’s opinion as to need and the patient’s desire to receive treatment, she said.
The study participants who were more likely to get treated were those with higher-level disease who had a stronger indication for treatment, Dr. Lok said.
Finding the disparities
Centers for Disease Control and Prevention statistics show that Black people are 3.9 times more likely to have CHB and 2.5 times more likely to die from it than White people, notes H. Nina Kim, MD, with the department of medicine, University of Washington, Seattle, in an accompanying invited commentary.
“The fact that we have not observed racial disparities in treatment initiation does not mean none exist; it means we have to look harder to find them,” she writes.
“We need to examine whether our guidelines for HBV treatment are so complex that it becomes the purview of specialists, thereby restricting access and deepening inequities,” Dr. Kim adds. “We should look closely at retention in care, the step that precedes treatment, and stratify this outcome by race and ethnicity.”
Primary care physicians in some regions might find it difficult to manage patients who have hepatitis B because they see so few of them, Dr. Lok noted.
Dr. Khalili has received grants and consulting fees from Gilead Sciences Inc and grants from Intercept Pharmaceuticals outside the submitted work. Dr. Lok has received grants from Target and consultant fees from Abbott, Ambys, Arbutus, Chroma, Clear B, Enanta, Enochian, GNI, GlaxoSmithKline, Eli Lilly, and Virion outside the submitted work. Coauthors have received grants, consulting fees, or personal fees from Bayer, Boston Scientific, Exact Sciences, Fujifilm Medical Sciences, Gilead Sciences, Glycotest, Redhill Biopharma, Target RWE, MedEd Design, Pontifax, Global Life, the Lynx Group, AstraZeneca, Eisai, Novartis Venture Fund, Grail, QED Therapeutics, Genentech, Hepion Pharmaceuticals, Roche, Abbott, AbbVie, and Pfizer. Dr. Kim has received grants from Gilead Sciences (paid to her institution) outside the submitted work.
A version of this article first appeared on Medscape.com.
FROM JAMA NETWORK OPEN
Likely cause of mysterious hepatitis outbreak in children identified
Coinfection with AAV2 and a human adenovirus (HAdV), in particular, appears to leave some children more vulnerable to this acute hepatitis of unknown origin, researchers reported in three studies published online in Nature. Coinfection with Epstein-Barr virus (EBV), herpes, and enterovirus also were found. Adeno-associated viruses are not considered pathogenic on their own and require a “helper” virus for productive infection.
“I am quite confident that we have identified the key viruses involved because we used a comprehensive metagenomic sequencing approach to look for potential infections from any virus or non-viral pathogen,” Charles Chiu, MD, PhD, senior author and professor of laboratory medicine and medicine/infectious diseases at the University of California, San Francisco, said in an interview.
Dr. Chiu and colleagues propose that lockdowns and social isolation during the COVID-19 pandemic left more children susceptible. A major aspect of immunity in childhood is the adaptive immune response – both cell-mediated and humoral – shaped in part by exposure to viruses and other pathogens early in life, Dr. Chiu said.
“Due to COVID-19, a large population of children did not experience this, so it is possible once restrictions were lifted, they were suddenly exposed over a short period of time to multiple viruses that, in a poorly trained immune system, would have increased their risk of developing severe disease,” he said.
This theory has been popular, especially because cases of unexplained acute hepatitis peaked during the height of the COVID-19 pandemic when isolation was common, William F. Balistreri, MD, who was not affiliated with the study, told this news organization. Dr. Balistreri is professor of pediatrics and director emeritus of the Pediatric Liver Care Center at Cincinnati Children’s Hospital Medical Center.
Identifying the culprits
Determining what factors might be involved was the main aim of the etiology study by Dr. Chiu and colleagues published online in Nature.
The journal simultaneously published a genomic study confirming the presence of AAV2 and other suspected viruses and a genomic and laboratory study further corroborating the results.
More than 1,000 children worldwide had been diagnosed with unexplained acute pediatric hepatitis as of August 2022. In the United States, there have been 358 cases, including 22 in which the child required a liver transplant and 13 in which the child died.
This new form of hepatitis, first detected in October 2021, does not fit into existing classifications of types A through E, so some researchers refer to the condition as acute non–A-E hepatitis of unknown etiology.
The investigators started with an important clue based on previous research: the role adenovirus might play. Dr. Chiu and colleagues assessed 27 blood, stool, and other samples from 16 affected children who each previously tested positive for adenoviruses. The researchers included cases of the condition identified up until May 22, 2022. The median age was 3 years, and approximately half were boys.
They compared viruses present in these children with those in 113 controls without the mysterious hepatitis. The control group consisted of 15 children who were hospitalized with a nonhepatitis inflammatory condition, 27 with a noninflammatory condition, 30 with acute hepatitis of known origin, 12 with acute gastroenteritis and an HAdV-positive stool sample, and 11 with acute gastroenteritis and an HAdV-negative stool sample, as well as 18 blood donors. The median age was 7 years.
The researchers assessed samples using multiple technologies, including metagenomic sequencing, tiling multiplex polymerase chain reaction (PCR) amplicon sequencing, metagenomic sequencing with probe capture viral enrichment, and virus-specific PCR. Many of these advanced techniques were not even available 5-10 years ago, Dr. Chiu said.
Key findings
Blood samples were available for 14 of the 16 children with acute hepatitis of unknown origin. Among this study group, AAV2 was found in 13 (93%). No other adeno-associated viruses were found. HAdV was detected in all 14 children: HAdV-41 in 11 children and HAdV-40, HAdV-2, and an untypeable strain in one child each. This finding was not intuitive because HAdVs are not commonly associated with hepatitis, according to the study.
AAV2 was much less common in the control group. For example, it was found in none of the children with hepatitis of known origin and in only four children (3.5%) with acute gastroenteritis and HAdV-positive stool. Of note, neither AAV2 nor HAdV-41 was detected among the 30 pediatric controls with acute hepatitis of defined etiology nor 42 of the hospitalized children without hepatitis, the researchers wrote.
In the search for other viruses in the study group, metagenomic sequencing detected EBV, also known as human herpesvirus (HHV)–4, in two children, cytomegalovirus (CMV) in one child, and HAdV type C in one child.
Analysis of whole blood revealed enterovirus A71 in one patient. HAdV type C also was detected in one child on the basis of a nasopharyngeal swab, and picobirnavirus was found in a stool sample from another patient.
Researchers conducted virus-specific PCR tests on both patient groups to identify additional viruses that may be associated with the unexplained acute hepatitis. EBV/HHV-4 was detected in 11 children (79%) in the study group vs. in 1 child (0.88%) in the control group. HHV-6 was detected in seven children (50%) in the study group, compared with one case in the control group. CMV was not detected in any of the children in the study group versus vs. two children (1.8%) in the control group.
“Although we found significant differences in the relative proportions of EBV and HHV-6 in cases compared to controls, we do not believe that these viruses are the primary cause of acute severe hepatitis,” the researchers wrote. The viral load of the two herpes viruses were very low, so the positive results could represent integrated proviral DNA rather than bona fide low-level herpesvirus. In addition, herpesvirus can be reactivated by an inflammatory condition.
“Nevertheless, it is striking that among the 16 cases (in the study group), dual, triple, or quadruple infections with AAV2, adenovirus, and one or both herpesviruses were detected in whole blood from at least 12 cases (75%),” the researchers wrote.
Management of suspected hepatitis
The study’s key messages for parents and health care providers “are awareness and reassurance,” Dr. Balistreri said in an interview.
Vigilance also is warranted if a child develops prodromal symptoms including respiratory and/or gastrointestinal signs such as nausea, vomiting, diarrhea, and abdomen pain, he said. If jaundice or scleral icterus is noted, then hepatitis should be suspected.
Some patients need hospitalization and quickly recover. In very rare instances, the inflammation may progress to liver failure and transplantation, Dr. Balistreri said.
“Reassurance is based on the good news that most children with acute hepatitis get better. If a case arises, it is good practice to keep the child well hydrated, offer a normal diet, and avoid medications that may be cleared by the liver,” Dr. Balistreri added.
“Of course, COVID-19 vaccination is strongly suggested,” he said.
Some existing treatments could help against unexplained acute hepatitis, Dr. Chiu said. “The findings suggest that antiviral therapy might be effective in these cases.”
Cidofovir can be effective against adenovirus, according to a report in The Lancet . Similarly, ganciclovir or valganciclovir may have activity against EBV/HHV-4 or HHV-6, Dr. Chiu said. “However, antiviral therapy is not available for AAV2.”
The three studies published in Nature “offer compelling evidence, from disparate centers, of a linkage of outbreak cases to infection by AAV2,” Dr. Balistreri said. The studies also suggest that liver injury was related to abnormal immune responses. This is an important clinical distinction, indicating a potential therapeutic approach to future cases – immunosuppression rather than anti-adenoviral agents, he said.
“We await further studies of this important concept,” Dr. Balistreri said.
Many unanswered questions remain about the condition’s etiology, he added. Is there a synergy or shared susceptibility related to SARS-CoV-2? Is the COVID-19 virus helping to trigger these infections, or does it increase the risk once infected? Also, are other epigenetic factors or viruses involved?
Moving forward
The next steps in the research could go beyond identifying presence of these different viruses and determining which one(s) are contributing the most to the acute pediatric hepatitis, Dr. Chiu said.
The researchers also would like to test early results from the United Kingdom that identified a potential association of acute severe hepatitis with the presence of human leukocyte antigen genotype DRB1*04:01, he added.
They also might investigate other unintended potential clinical consequences of the COVID-19 pandemic, including long COVID and resurgence of infections from other viruses, such as respiratory syncytial virus, influenza, and enterovirus D68.
The study was supported by the Centers for Disease Control and Prevention, the National Institutes of Health, the Department of Homeland Security, and other grants. Dr. Chiu is a founder of Delve Bio and on the scientific advisory board for Delve Bio, Mammoth Biosciences, BiomeSense, and Poppy Health. Dr. Balistreri had no relevant disclosures.
A version of this article first appeared on Medscape.com.
Coinfection with AAV2 and a human adenovirus (HAdV), in particular, appears to leave some children more vulnerable to this acute hepatitis of unknown origin, researchers reported in three studies published online in Nature. Coinfection with Epstein-Barr virus (EBV), herpes, and enterovirus also were found. Adeno-associated viruses are not considered pathogenic on their own and require a “helper” virus for productive infection.
“I am quite confident that we have identified the key viruses involved because we used a comprehensive metagenomic sequencing approach to look for potential infections from any virus or non-viral pathogen,” Charles Chiu, MD, PhD, senior author and professor of laboratory medicine and medicine/infectious diseases at the University of California, San Francisco, said in an interview.
Dr. Chiu and colleagues propose that lockdowns and social isolation during the COVID-19 pandemic left more children susceptible. A major aspect of immunity in childhood is the adaptive immune response – both cell-mediated and humoral – shaped in part by exposure to viruses and other pathogens early in life, Dr. Chiu said.
“Due to COVID-19, a large population of children did not experience this, so it is possible once restrictions were lifted, they were suddenly exposed over a short period of time to multiple viruses that, in a poorly trained immune system, would have increased their risk of developing severe disease,” he said.
This theory has been popular, especially because cases of unexplained acute hepatitis peaked during the height of the COVID-19 pandemic when isolation was common, William F. Balistreri, MD, who was not affiliated with the study, told this news organization. Dr. Balistreri is professor of pediatrics and director emeritus of the Pediatric Liver Care Center at Cincinnati Children’s Hospital Medical Center.
Identifying the culprits
Determining what factors might be involved was the main aim of the etiology study by Dr. Chiu and colleagues published online in Nature.
The journal simultaneously published a genomic study confirming the presence of AAV2 and other suspected viruses and a genomic and laboratory study further corroborating the results.
More than 1,000 children worldwide had been diagnosed with unexplained acute pediatric hepatitis as of August 2022. In the United States, there have been 358 cases, including 22 in which the child required a liver transplant and 13 in which the child died.
This new form of hepatitis, first detected in October 2021, does not fit into existing classifications of types A through E, so some researchers refer to the condition as acute non–A-E hepatitis of unknown etiology.
The investigators started with an important clue based on previous research: the role adenovirus might play. Dr. Chiu and colleagues assessed 27 blood, stool, and other samples from 16 affected children who each previously tested positive for adenoviruses. The researchers included cases of the condition identified up until May 22, 2022. The median age was 3 years, and approximately half were boys.
They compared viruses present in these children with those in 113 controls without the mysterious hepatitis. The control group consisted of 15 children who were hospitalized with a nonhepatitis inflammatory condition, 27 with a noninflammatory condition, 30 with acute hepatitis of known origin, 12 with acute gastroenteritis and an HAdV-positive stool sample, and 11 with acute gastroenteritis and an HAdV-negative stool sample, as well as 18 blood donors. The median age was 7 years.
The researchers assessed samples using multiple technologies, including metagenomic sequencing, tiling multiplex polymerase chain reaction (PCR) amplicon sequencing, metagenomic sequencing with probe capture viral enrichment, and virus-specific PCR. Many of these advanced techniques were not even available 5-10 years ago, Dr. Chiu said.
Key findings
Blood samples were available for 14 of the 16 children with acute hepatitis of unknown origin. Among this study group, AAV2 was found in 13 (93%). No other adeno-associated viruses were found. HAdV was detected in all 14 children: HAdV-41 in 11 children and HAdV-40, HAdV-2, and an untypeable strain in one child each. This finding was not intuitive because HAdVs are not commonly associated with hepatitis, according to the study.
AAV2 was much less common in the control group. For example, it was found in none of the children with hepatitis of known origin and in only four children (3.5%) with acute gastroenteritis and HAdV-positive stool. Of note, neither AAV2 nor HAdV-41 was detected among the 30 pediatric controls with acute hepatitis of defined etiology nor 42 of the hospitalized children without hepatitis, the researchers wrote.
In the search for other viruses in the study group, metagenomic sequencing detected EBV, also known as human herpesvirus (HHV)–4, in two children, cytomegalovirus (CMV) in one child, and HAdV type C in one child.
Analysis of whole blood revealed enterovirus A71 in one patient. HAdV type C also was detected in one child on the basis of a nasopharyngeal swab, and picobirnavirus was found in a stool sample from another patient.
Researchers conducted virus-specific PCR tests on both patient groups to identify additional viruses that may be associated with the unexplained acute hepatitis. EBV/HHV-4 was detected in 11 children (79%) in the study group vs. in 1 child (0.88%) in the control group. HHV-6 was detected in seven children (50%) in the study group, compared with one case in the control group. CMV was not detected in any of the children in the study group versus vs. two children (1.8%) in the control group.
“Although we found significant differences in the relative proportions of EBV and HHV-6 in cases compared to controls, we do not believe that these viruses are the primary cause of acute severe hepatitis,” the researchers wrote. The viral load of the two herpes viruses were very low, so the positive results could represent integrated proviral DNA rather than bona fide low-level herpesvirus. In addition, herpesvirus can be reactivated by an inflammatory condition.
“Nevertheless, it is striking that among the 16 cases (in the study group), dual, triple, or quadruple infections with AAV2, adenovirus, and one or both herpesviruses were detected in whole blood from at least 12 cases (75%),” the researchers wrote.
Management of suspected hepatitis
The study’s key messages for parents and health care providers “are awareness and reassurance,” Dr. Balistreri said in an interview.
Vigilance also is warranted if a child develops prodromal symptoms including respiratory and/or gastrointestinal signs such as nausea, vomiting, diarrhea, and abdomen pain, he said. If jaundice or scleral icterus is noted, then hepatitis should be suspected.
Some patients need hospitalization and quickly recover. In very rare instances, the inflammation may progress to liver failure and transplantation, Dr. Balistreri said.
“Reassurance is based on the good news that most children with acute hepatitis get better. If a case arises, it is good practice to keep the child well hydrated, offer a normal diet, and avoid medications that may be cleared by the liver,” Dr. Balistreri added.
“Of course, COVID-19 vaccination is strongly suggested,” he said.
Some existing treatments could help against unexplained acute hepatitis, Dr. Chiu said. “The findings suggest that antiviral therapy might be effective in these cases.”
Cidofovir can be effective against adenovirus, according to a report in The Lancet . Similarly, ganciclovir or valganciclovir may have activity against EBV/HHV-4 or HHV-6, Dr. Chiu said. “However, antiviral therapy is not available for AAV2.”
The three studies published in Nature “offer compelling evidence, from disparate centers, of a linkage of outbreak cases to infection by AAV2,” Dr. Balistreri said. The studies also suggest that liver injury was related to abnormal immune responses. This is an important clinical distinction, indicating a potential therapeutic approach to future cases – immunosuppression rather than anti-adenoviral agents, he said.
“We await further studies of this important concept,” Dr. Balistreri said.
Many unanswered questions remain about the condition’s etiology, he added. Is there a synergy or shared susceptibility related to SARS-CoV-2? Is the COVID-19 virus helping to trigger these infections, or does it increase the risk once infected? Also, are other epigenetic factors or viruses involved?
Moving forward
The next steps in the research could go beyond identifying presence of these different viruses and determining which one(s) are contributing the most to the acute pediatric hepatitis, Dr. Chiu said.
The researchers also would like to test early results from the United Kingdom that identified a potential association of acute severe hepatitis with the presence of human leukocyte antigen genotype DRB1*04:01, he added.
They also might investigate other unintended potential clinical consequences of the COVID-19 pandemic, including long COVID and resurgence of infections from other viruses, such as respiratory syncytial virus, influenza, and enterovirus D68.
The study was supported by the Centers for Disease Control and Prevention, the National Institutes of Health, the Department of Homeland Security, and other grants. Dr. Chiu is a founder of Delve Bio and on the scientific advisory board for Delve Bio, Mammoth Biosciences, BiomeSense, and Poppy Health. Dr. Balistreri had no relevant disclosures.
A version of this article first appeared on Medscape.com.
Coinfection with AAV2 and a human adenovirus (HAdV), in particular, appears to leave some children more vulnerable to this acute hepatitis of unknown origin, researchers reported in three studies published online in Nature. Coinfection with Epstein-Barr virus (EBV), herpes, and enterovirus also were found. Adeno-associated viruses are not considered pathogenic on their own and require a “helper” virus for productive infection.
“I am quite confident that we have identified the key viruses involved because we used a comprehensive metagenomic sequencing approach to look for potential infections from any virus or non-viral pathogen,” Charles Chiu, MD, PhD, senior author and professor of laboratory medicine and medicine/infectious diseases at the University of California, San Francisco, said in an interview.
Dr. Chiu and colleagues propose that lockdowns and social isolation during the COVID-19 pandemic left more children susceptible. A major aspect of immunity in childhood is the adaptive immune response – both cell-mediated and humoral – shaped in part by exposure to viruses and other pathogens early in life, Dr. Chiu said.
“Due to COVID-19, a large population of children did not experience this, so it is possible once restrictions were lifted, they were suddenly exposed over a short period of time to multiple viruses that, in a poorly trained immune system, would have increased their risk of developing severe disease,” he said.
This theory has been popular, especially because cases of unexplained acute hepatitis peaked during the height of the COVID-19 pandemic when isolation was common, William F. Balistreri, MD, who was not affiliated with the study, told this news organization. Dr. Balistreri is professor of pediatrics and director emeritus of the Pediatric Liver Care Center at Cincinnati Children’s Hospital Medical Center.
Identifying the culprits
Determining what factors might be involved was the main aim of the etiology study by Dr. Chiu and colleagues published online in Nature.
The journal simultaneously published a genomic study confirming the presence of AAV2 and other suspected viruses and a genomic and laboratory study further corroborating the results.
More than 1,000 children worldwide had been diagnosed with unexplained acute pediatric hepatitis as of August 2022. In the United States, there have been 358 cases, including 22 in which the child required a liver transplant and 13 in which the child died.
This new form of hepatitis, first detected in October 2021, does not fit into existing classifications of types A through E, so some researchers refer to the condition as acute non–A-E hepatitis of unknown etiology.
The investigators started with an important clue based on previous research: the role adenovirus might play. Dr. Chiu and colleagues assessed 27 blood, stool, and other samples from 16 affected children who each previously tested positive for adenoviruses. The researchers included cases of the condition identified up until May 22, 2022. The median age was 3 years, and approximately half were boys.
They compared viruses present in these children with those in 113 controls without the mysterious hepatitis. The control group consisted of 15 children who were hospitalized with a nonhepatitis inflammatory condition, 27 with a noninflammatory condition, 30 with acute hepatitis of known origin, 12 with acute gastroenteritis and an HAdV-positive stool sample, and 11 with acute gastroenteritis and an HAdV-negative stool sample, as well as 18 blood donors. The median age was 7 years.
The researchers assessed samples using multiple technologies, including metagenomic sequencing, tiling multiplex polymerase chain reaction (PCR) amplicon sequencing, metagenomic sequencing with probe capture viral enrichment, and virus-specific PCR. Many of these advanced techniques were not even available 5-10 years ago, Dr. Chiu said.
Key findings
Blood samples were available for 14 of the 16 children with acute hepatitis of unknown origin. Among this study group, AAV2 was found in 13 (93%). No other adeno-associated viruses were found. HAdV was detected in all 14 children: HAdV-41 in 11 children and HAdV-40, HAdV-2, and an untypeable strain in one child each. This finding was not intuitive because HAdVs are not commonly associated with hepatitis, according to the study.
AAV2 was much less common in the control group. For example, it was found in none of the children with hepatitis of known origin and in only four children (3.5%) with acute gastroenteritis and HAdV-positive stool. Of note, neither AAV2 nor HAdV-41 was detected among the 30 pediatric controls with acute hepatitis of defined etiology nor 42 of the hospitalized children without hepatitis, the researchers wrote.
In the search for other viruses in the study group, metagenomic sequencing detected EBV, also known as human herpesvirus (HHV)–4, in two children, cytomegalovirus (CMV) in one child, and HAdV type C in one child.
Analysis of whole blood revealed enterovirus A71 in one patient. HAdV type C also was detected in one child on the basis of a nasopharyngeal swab, and picobirnavirus was found in a stool sample from another patient.
Researchers conducted virus-specific PCR tests on both patient groups to identify additional viruses that may be associated with the unexplained acute hepatitis. EBV/HHV-4 was detected in 11 children (79%) in the study group vs. in 1 child (0.88%) in the control group. HHV-6 was detected in seven children (50%) in the study group, compared with one case in the control group. CMV was not detected in any of the children in the study group versus vs. two children (1.8%) in the control group.
“Although we found significant differences in the relative proportions of EBV and HHV-6 in cases compared to controls, we do not believe that these viruses are the primary cause of acute severe hepatitis,” the researchers wrote. The viral load of the two herpes viruses were very low, so the positive results could represent integrated proviral DNA rather than bona fide low-level herpesvirus. In addition, herpesvirus can be reactivated by an inflammatory condition.
“Nevertheless, it is striking that among the 16 cases (in the study group), dual, triple, or quadruple infections with AAV2, adenovirus, and one or both herpesviruses were detected in whole blood from at least 12 cases (75%),” the researchers wrote.
Management of suspected hepatitis
The study’s key messages for parents and health care providers “are awareness and reassurance,” Dr. Balistreri said in an interview.
Vigilance also is warranted if a child develops prodromal symptoms including respiratory and/or gastrointestinal signs such as nausea, vomiting, diarrhea, and abdomen pain, he said. If jaundice or scleral icterus is noted, then hepatitis should be suspected.
Some patients need hospitalization and quickly recover. In very rare instances, the inflammation may progress to liver failure and transplantation, Dr. Balistreri said.
“Reassurance is based on the good news that most children with acute hepatitis get better. If a case arises, it is good practice to keep the child well hydrated, offer a normal diet, and avoid medications that may be cleared by the liver,” Dr. Balistreri added.
“Of course, COVID-19 vaccination is strongly suggested,” he said.
Some existing treatments could help against unexplained acute hepatitis, Dr. Chiu said. “The findings suggest that antiviral therapy might be effective in these cases.”
Cidofovir can be effective against adenovirus, according to a report in The Lancet . Similarly, ganciclovir or valganciclovir may have activity against EBV/HHV-4 or HHV-6, Dr. Chiu said. “However, antiviral therapy is not available for AAV2.”
The three studies published in Nature “offer compelling evidence, from disparate centers, of a linkage of outbreak cases to infection by AAV2,” Dr. Balistreri said. The studies also suggest that liver injury was related to abnormal immune responses. This is an important clinical distinction, indicating a potential therapeutic approach to future cases – immunosuppression rather than anti-adenoviral agents, he said.
“We await further studies of this important concept,” Dr. Balistreri said.
Many unanswered questions remain about the condition’s etiology, he added. Is there a synergy or shared susceptibility related to SARS-CoV-2? Is the COVID-19 virus helping to trigger these infections, or does it increase the risk once infected? Also, are other epigenetic factors or viruses involved?
Moving forward
The next steps in the research could go beyond identifying presence of these different viruses and determining which one(s) are contributing the most to the acute pediatric hepatitis, Dr. Chiu said.
The researchers also would like to test early results from the United Kingdom that identified a potential association of acute severe hepatitis with the presence of human leukocyte antigen genotype DRB1*04:01, he added.
They also might investigate other unintended potential clinical consequences of the COVID-19 pandemic, including long COVID and resurgence of infections from other viruses, such as respiratory syncytial virus, influenza, and enterovirus D68.
The study was supported by the Centers for Disease Control and Prevention, the National Institutes of Health, the Department of Homeland Security, and other grants. Dr. Chiu is a founder of Delve Bio and on the scientific advisory board for Delve Bio, Mammoth Biosciences, BiomeSense, and Poppy Health. Dr. Balistreri had no relevant disclosures.
A version of this article first appeared on Medscape.com.
FROM NATURE
Celebrity death finally solved – with locks of hair
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.
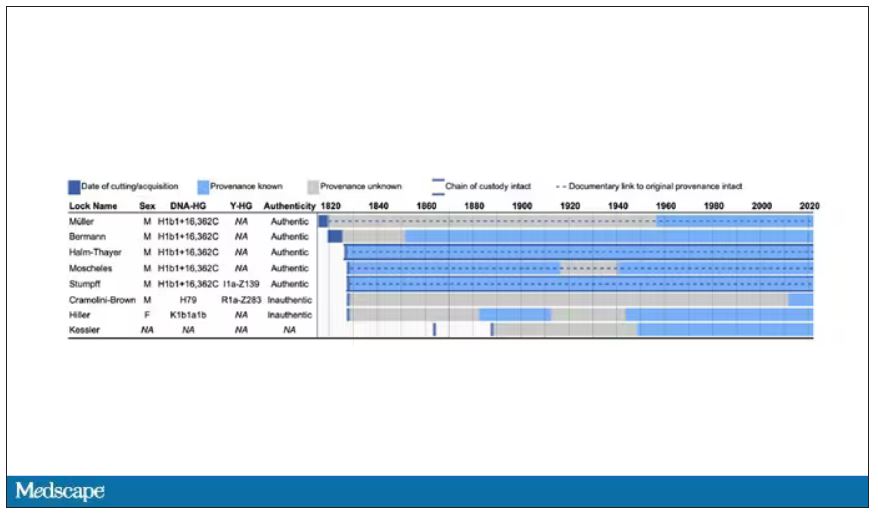
The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.

DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.
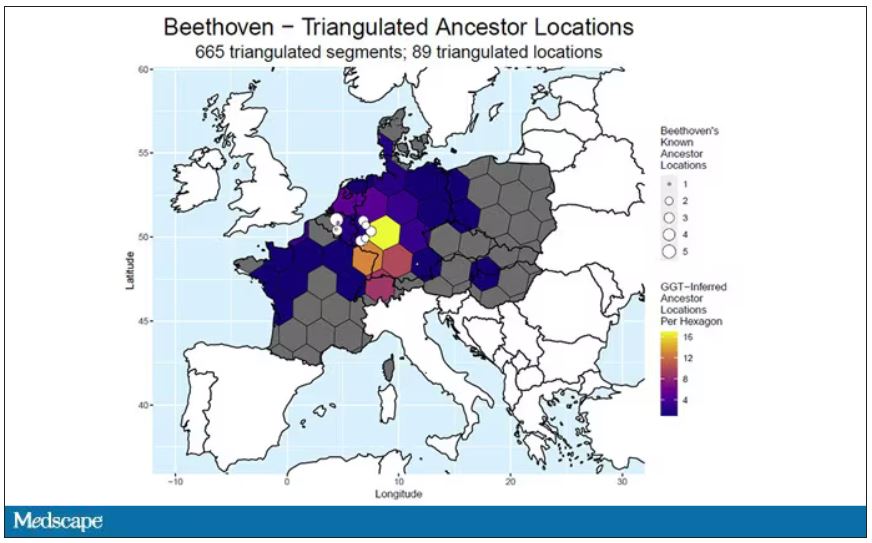
In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.
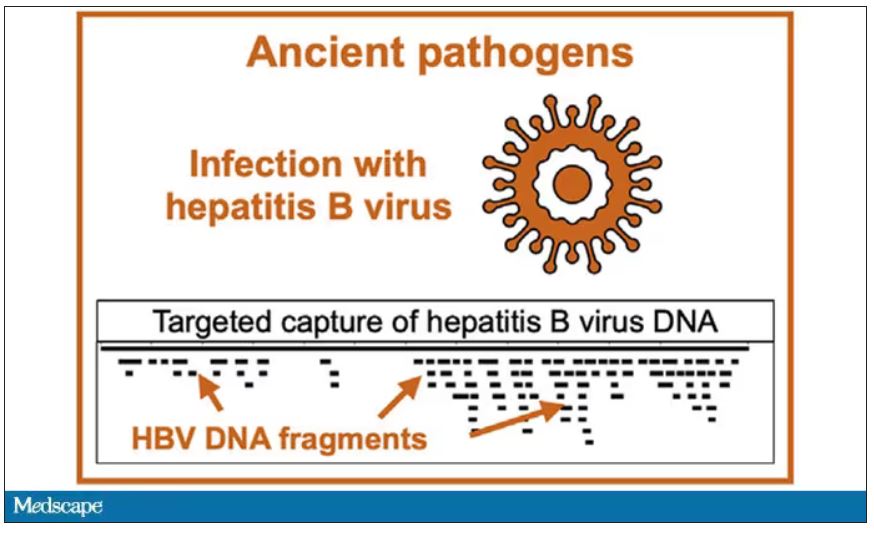
It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.

The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.

DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.

In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.

It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
This transcript has been edited for clarity.
I’m going to open this week with a case.
A 56-year-old musician presents with diffuse abdominal pain, cramping, and jaundice. His medical history is notable for years of diffuse abdominal complaints, characterized by disabling bouts of diarrhea.
In addition to the jaundice, this acute illness was accompanied by fever as well as diffuse edema and ascites. The patient underwent several abdominal paracenteses to drain excess fluid. One consulting physician administered alcohol to relieve pain, to little avail.
The patient succumbed to his illness. An autopsy showed diffuse liver injury, as well as papillary necrosis of the kidneys. Notably, the nerves of his auditory canal were noted to be thickened, along with the bony part of the skull, consistent with Paget disease of the bone and explaining, potentially, why the talented musician had gone deaf at such a young age.
An interesting note on social history: The patient had apparently developed some feelings for the niece of that doctor who prescribed alcohol. Her name was Therese, perhaps mistranscribed as Elise, and it seems that he may have written this song for her.
We’re talking about this paper in Current Biology, by Tristan Begg and colleagues, which gives us a look into the very genome of what some would argue is the world’s greatest composer.
The ability to extract DNA from older specimens has transformed the fields of anthropology, archaeology, and history, and now, perhaps, musicology as well.
The researchers identified eight locks of hair in private and public collections, all attributed to the maestro.
Four of the samples had an intact chain of custody from the time the hair was cut. DNA sequencing on these four and an additional one of the eight locks came from the same individual, a male of European heritage.

The three locks with less documentation came from three other unrelated individuals. Interestingly, analysis of one of those hair samples – the so-called Hiller Lock – had shown high levels of lead, leading historians to speculate that lead poisoning could account for some of Beethoven’s symptoms.

DNA analysis of that hair reveals it to have come from a woman likely of North African, Middle Eastern, or Jewish ancestry. We can no longer presume that plumbism was involved in Beethoven’s death. Beethoven’s ancestry turns out to be less exotic and maps quite well to ethnic German populations today.

In fact, there are van Beethovens alive as we speak, primarily in Belgium. Genealogic records suggest that these van Beethovens share a common ancestor with the virtuoso composer, a man by the name of Aert van Beethoven.
But the DNA reveals a scandal.
The Y-chromosome that Beethoven inherited was not Aert van Beethoven’s. Questions of Beethoven’s paternity have been raised before, but this evidence strongly suggests an extramarital paternity event, at least in the generations preceding his birth. That’s right – Beethoven may not have been a Beethoven.
With five locks now essentially certain to have come from Beethoven himself, the authors could use DNA analysis to try to explain three significant health problems he experienced throughout his life and death: his hearing loss, his terrible gastrointestinal issues, and his liver failure.
Let’s start with the most disappointing results, explanations for his hearing loss. No genetic cause was forthcoming, though the authors note that they have little to go on in regard to the genetic risk for otosclerosis, to which his hearing loss has often been attributed. Lead poisoning is, of course, possible here, though this report focuses only on genetics – there was no testing for lead – and as I mentioned, the lock that was strongly lead-positive in prior studies is almost certainly inauthentic.
What about his lifelong GI complaints? Some have suggested celiac disease or lactose intolerance as explanations. These can essentially be ruled out by the genetic analysis, which shows no risk alleles for celiac disease and the presence of the lactase-persistence gene which confers the ability to metabolize lactose throughout one’s life. IBS is harder to assess genetically, but for what it’s worth, he scored quite low on a polygenic risk score for the condition, in just the 9th percentile of risk. We should probably be looking elsewhere to explain the GI distress.
The genetic information bore much more fruit in regard to his liver disease. Remember that Beethoven’s autopsy showed cirrhosis. His polygenic risk score for liver cirrhosis puts him in the 96th percentile of risk. He was also heterozygous for two variants that can cause hereditary hemochromatosis. The risk for cirrhosis among those with these variants is increased by the use of alcohol. And historical accounts are quite clear that Beethoven consumed more than his share.
But it wasn’t just Beethoven’s DNA in these hair follicles. Analysis of a follicle from later in his life revealed the unmistakable presence of hepatitis B virus. Endemic in Europe at the time, this was a common cause of liver failure and is likely to have contributed to, if not directly caused, Beethoven’s demise.

It’s hard to read these results and not marvel at the fact that, two centuries after his death, our fascination with Beethoven has led us to probe every corner of his life – his letters, his writings, his medical records, and now his very DNA. What are we actually looking for? Is it relevant to us today what caused his hearing loss? His stomach troubles? Even his death? Will it help any patients in the future? I propose that what we are actually trying to understand is something ineffable: Genius of magnitude that is rarely seen in one or many lifetimes. And our scientific tools, as sharp as they may have become, are still far too blunt to probe the depths of that transcendence.
In any case, friends, no more of these sounds. Let us sing more cheerful songs, more full of joy.
For Medscape, I’m Perry Wilson.
Dr. Wilson is associate professor, department of medicine, and director, Clinical and Translational Research Accelerator, at Yale University, New Haven, Conn. He reported no conflicts of interest.
A version of this article first appeared on Medscape.com.
Increased cancer in military pilots and ground crew: Pentagon
“Military aircrew and ground crew were overall more likely to be diagnosed with cancer, but less likely to die from cancer compared to the U.S. population,” the report concludes.
The study involved 156,050 aircrew and 737,891 ground crew. Participants were followed between 1992 and 2017. Both groups were predominantly male and non-Hispanic.
Data on cancer incidence and mortality for these two groups were compared with data from groups of similar age in the general population through use of the Surveillance, Epidemiology, and End Results (SEER) Database of the National Cancer Institute.
For aircrew, the study found an 87% higher rate of melanoma, a 39% higher rate of thyroid cancer, a 16% higher rate of prostate cancer, and a 24% higher rate of cancer for all sites combined.
A higher rate of melanoma and prostate cancer among aircrew has been reported previously, but the increased rate of thyroid cancer is a new finding, the authors note.
The uptick in melanoma has also been reported in studies of civilian pilots and cabin crew. It has been attributed to exposure to hazardous ultraviolet and cosmic radiation.
For ground crew members, the analysis found a 19% higher rate of cancers of the brain and nervous system, a 15% higher rate of thyroid cancer, a 9% higher rate of melanoma and of kidney and renal pelvis cancers, and a 3% higher rate of cancer for all sites combined.
There is little to compare these findings with: This is the first time that cancer risk has been evaluated in such a large population of military ground crew.
Lower rates of cancer mortality
In contrast to the increase in cancer incidence, the report found a decrease in cancer mortality.
When compared with a demographically similar U.S. population, the mortality rate among aircrew was 56% lower for all cancer sites; for ground crew, the mortality rate was 35% lower.
However, the report authors emphasize that “it is important to note that the military study population was relatively young.”
The median age at the end of follow-up for the cancer incidence analysis was 41 years for aircrew and 26 years for ground crew. The median age at the end of follow-up for the cancer mortality analysis was 48 years for aircrew and 41 years for ground crew.
“Results may have differed if additional older former Service members had been included in the study, since cancer risk and mortality rates increase with age,” the authors comment.
Other studies have found an increase in deaths from melanoma as well as an increase in the incidence of melanoma. A meta-analysis published in 2019 in the British Journal of Dermatology found that airline pilots and cabin crew have about twice the risk of melanoma and other skin cancers than the general population. Pilots are also more likely to die from melanoma.
Further study underway
The findings on military air and ground crew come from phase 1 of a study that was required by Congress in the 2021 defense bill. Because the investigators found an increase in the incidence of cancer, phase 2 of the study is now necessary.
The report authors explain that phase 2 will consist of identifying the carcinogenic toxicants or hazardous materials associated with military flight operations; identifying operating environments that could be associated with increased amounts of ionizing and nonionizing radiation; identifying specific duties, dates of service, and types of aircraft flown that could have increased the risk for cancer; identifying duty locations associated with a higher incidence of cancers; identifying potential exposures through military service that are not related to aviation; and determining the appropriate age to begin screening military aircrew and ground crew for cancers.
A version of this article first appeared on Medscape.com.
“Military aircrew and ground crew were overall more likely to be diagnosed with cancer, but less likely to die from cancer compared to the U.S. population,” the report concludes.
The study involved 156,050 aircrew and 737,891 ground crew. Participants were followed between 1992 and 2017. Both groups were predominantly male and non-Hispanic.
Data on cancer incidence and mortality for these two groups were compared with data from groups of similar age in the general population through use of the Surveillance, Epidemiology, and End Results (SEER) Database of the National Cancer Institute.
For aircrew, the study found an 87% higher rate of melanoma, a 39% higher rate of thyroid cancer, a 16% higher rate of prostate cancer, and a 24% higher rate of cancer for all sites combined.
A higher rate of melanoma and prostate cancer among aircrew has been reported previously, but the increased rate of thyroid cancer is a new finding, the authors note.
The uptick in melanoma has also been reported in studies of civilian pilots and cabin crew. It has been attributed to exposure to hazardous ultraviolet and cosmic radiation.
For ground crew members, the analysis found a 19% higher rate of cancers of the brain and nervous system, a 15% higher rate of thyroid cancer, a 9% higher rate of melanoma and of kidney and renal pelvis cancers, and a 3% higher rate of cancer for all sites combined.
There is little to compare these findings with: This is the first time that cancer risk has been evaluated in such a large population of military ground crew.
Lower rates of cancer mortality
In contrast to the increase in cancer incidence, the report found a decrease in cancer mortality.
When compared with a demographically similar U.S. population, the mortality rate among aircrew was 56% lower for all cancer sites; for ground crew, the mortality rate was 35% lower.
However, the report authors emphasize that “it is important to note that the military study population was relatively young.”
The median age at the end of follow-up for the cancer incidence analysis was 41 years for aircrew and 26 years for ground crew. The median age at the end of follow-up for the cancer mortality analysis was 48 years for aircrew and 41 years for ground crew.
“Results may have differed if additional older former Service members had been included in the study, since cancer risk and mortality rates increase with age,” the authors comment.
Other studies have found an increase in deaths from melanoma as well as an increase in the incidence of melanoma. A meta-analysis published in 2019 in the British Journal of Dermatology found that airline pilots and cabin crew have about twice the risk of melanoma and other skin cancers than the general population. Pilots are also more likely to die from melanoma.
Further study underway
The findings on military air and ground crew come from phase 1 of a study that was required by Congress in the 2021 defense bill. Because the investigators found an increase in the incidence of cancer, phase 2 of the study is now necessary.
The report authors explain that phase 2 will consist of identifying the carcinogenic toxicants or hazardous materials associated with military flight operations; identifying operating environments that could be associated with increased amounts of ionizing and nonionizing radiation; identifying specific duties, dates of service, and types of aircraft flown that could have increased the risk for cancer; identifying duty locations associated with a higher incidence of cancers; identifying potential exposures through military service that are not related to aviation; and determining the appropriate age to begin screening military aircrew and ground crew for cancers.
A version of this article first appeared on Medscape.com.
“Military aircrew and ground crew were overall more likely to be diagnosed with cancer, but less likely to die from cancer compared to the U.S. population,” the report concludes.
The study involved 156,050 aircrew and 737,891 ground crew. Participants were followed between 1992 and 2017. Both groups were predominantly male and non-Hispanic.
Data on cancer incidence and mortality for these two groups were compared with data from groups of similar age in the general population through use of the Surveillance, Epidemiology, and End Results (SEER) Database of the National Cancer Institute.
For aircrew, the study found an 87% higher rate of melanoma, a 39% higher rate of thyroid cancer, a 16% higher rate of prostate cancer, and a 24% higher rate of cancer for all sites combined.
A higher rate of melanoma and prostate cancer among aircrew has been reported previously, but the increased rate of thyroid cancer is a new finding, the authors note.
The uptick in melanoma has also been reported in studies of civilian pilots and cabin crew. It has been attributed to exposure to hazardous ultraviolet and cosmic radiation.
For ground crew members, the analysis found a 19% higher rate of cancers of the brain and nervous system, a 15% higher rate of thyroid cancer, a 9% higher rate of melanoma and of kidney and renal pelvis cancers, and a 3% higher rate of cancer for all sites combined.
There is little to compare these findings with: This is the first time that cancer risk has been evaluated in such a large population of military ground crew.
Lower rates of cancer mortality
In contrast to the increase in cancer incidence, the report found a decrease in cancer mortality.
When compared with a demographically similar U.S. population, the mortality rate among aircrew was 56% lower for all cancer sites; for ground crew, the mortality rate was 35% lower.
However, the report authors emphasize that “it is important to note that the military study population was relatively young.”
The median age at the end of follow-up for the cancer incidence analysis was 41 years for aircrew and 26 years for ground crew. The median age at the end of follow-up for the cancer mortality analysis was 48 years for aircrew and 41 years for ground crew.
“Results may have differed if additional older former Service members had been included in the study, since cancer risk and mortality rates increase with age,” the authors comment.
Other studies have found an increase in deaths from melanoma as well as an increase in the incidence of melanoma. A meta-analysis published in 2019 in the British Journal of Dermatology found that airline pilots and cabin crew have about twice the risk of melanoma and other skin cancers than the general population. Pilots are also more likely to die from melanoma.
Further study underway
The findings on military air and ground crew come from phase 1 of a study that was required by Congress in the 2021 defense bill. Because the investigators found an increase in the incidence of cancer, phase 2 of the study is now necessary.
The report authors explain that phase 2 will consist of identifying the carcinogenic toxicants or hazardous materials associated with military flight operations; identifying operating environments that could be associated with increased amounts of ionizing and nonionizing radiation; identifying specific duties, dates of service, and types of aircraft flown that could have increased the risk for cancer; identifying duty locations associated with a higher incidence of cancers; identifying potential exposures through military service that are not related to aviation; and determining the appropriate age to begin screening military aircrew and ground crew for cancers.
A version of this article first appeared on Medscape.com.
CDC recommends screening all adults for hepatitis B
This is the first update to HBV screening guidelines since 2008, the agency said.
“Risk-based testing alone has not identified most persons living with chronic HBV infection and is considered inefficient for providers to implement,” the authors wrote in the new guidance, published in the CDC’s Morbidity and Mortality Weekly Report. “Universal screening of adults for HBV infection is cost-effective, compared with risk-based screening and averts liver disease and death. Although a curative treatment is not yet available, early diagnosis and treatment of chronic HBV infections reduces the risk for cirrhosis, liver cancer, and death.”
Howard Lee, MD, an assistant professor in the section of gastroenterology and hepatology at Baylor College of Medicine in Houston, agreed that risk-based screening has not been effective. A universal screening approach “is the way to go,” he said. With this new screening approach, patients can get tested without having to admit that they may be at risk for a chronic disease like HIV and HBV, which can be stigmatizing, said Dr. Lee, who was not involved with making these recommendations.
An estimated 580,000 to 2.4 million individuals are living with HBV infection in the United States, and two-thirds may be unaware they are infected, according to the CDC. The virus spreads through contact with blood, semen, and other body fluids of an infected person.
The guidance now recommends using the triple panel (HBsAg, anti-HBs, total anti-HBc) for initial screening.
“It can help identify persons who have an active HBV infection and could be linked to care; have resolved infection and might be susceptible to reactivation (for example, immunosuppressed persons); are susceptible and need vaccination; or are vaccinated,” the authors wrote.
Patients with previous HBV infection can have the infection reactivated with immunosuppressive treatments, Dr. Lee said, which is why detecting prior infection via the triple panel screening is important.
Women who are pregnant should be screened, ideally, in the first trimester of each pregnancy, regardless of vaccination status or testing history. If they have already received timely triple panel screening for hepatitis B and have no new HBV exposures, pregnant women only need HBsAg screening, the guidelines state.
The guidelines also specify that higher risk groups, specifically those incarcerated or formerly incarcerated, adults with current or past hepatitis C virus infection, and those with current or past sexually transmitted infections and multiple sex partners.
People who are susceptible for infection, refuse vaccination and are at higher risk for HBV should be screened periodically, but how often they should be screened should be based on shared decision-making between the provider and patient as well as individual risk and immune status.
Additional research into the optimal frequency of periodic testing is necessary, the authors say.
“Along with vaccination strategies, universal screening of adults and appropriate testing of persons at increased risk for HBV infection will improve health outcomes, reduce the prevalence of HBV infection in the United States, and advance viral hepatitis elimination goals,” the authors wrote.
The new recommendations now contrast with the 2020 screening guidelines issued by the U.S. Preventive Services Task Force (USPSTF) that recommend risk-based screening for hepatitis B.
“When that recommendation was published, the Task Force was aligned with several other organizations, including the CDC, in supporting screening for hepatitis B in high-risk populations — and importantly, we’re all still aligned in making sure that people get the care that they need,” said Michael Barry, MD, chair of the USPSTF, in an emailed statement. “The evidence on clinical preventive services is always changing, and the Task Force aims to keep all recommendations current, updating each recommendation approximately every 5 years.”
“In the meantime, we always encourage clinicians to use their judgment as they provide care for their patients — including those who may benefit from screening for hepatitis B — and to decide together with each patient which preventive services can best help them live a long and healthy life,” Dr. Barry said.
The American Association for the Study of Liver Diseases is currently updating their HBV screening recommendations, Dr. Lee said, and he expects other professional societies to follow the CDC recommendations.
“It’s not uncommon that we see the CDC or societies making recommendations and the USPSTF following along, so hopefully that’s the case for hepatitis B as well,” he said.
The authors reported no potential conflicts of interest.
A version of this article originally appeared on Medscape.com.
This is the first update to HBV screening guidelines since 2008, the agency said.
“Risk-based testing alone has not identified most persons living with chronic HBV infection and is considered inefficient for providers to implement,” the authors wrote in the new guidance, published in the CDC’s Morbidity and Mortality Weekly Report. “Universal screening of adults for HBV infection is cost-effective, compared with risk-based screening and averts liver disease and death. Although a curative treatment is not yet available, early diagnosis and treatment of chronic HBV infections reduces the risk for cirrhosis, liver cancer, and death.”
Howard Lee, MD, an assistant professor in the section of gastroenterology and hepatology at Baylor College of Medicine in Houston, agreed that risk-based screening has not been effective. A universal screening approach “is the way to go,” he said. With this new screening approach, patients can get tested without having to admit that they may be at risk for a chronic disease like HIV and HBV, which can be stigmatizing, said Dr. Lee, who was not involved with making these recommendations.
An estimated 580,000 to 2.4 million individuals are living with HBV infection in the United States, and two-thirds may be unaware they are infected, according to the CDC. The virus spreads through contact with blood, semen, and other body fluids of an infected person.
The guidance now recommends using the triple panel (HBsAg, anti-HBs, total anti-HBc) for initial screening.
“It can help identify persons who have an active HBV infection and could be linked to care; have resolved infection and might be susceptible to reactivation (for example, immunosuppressed persons); are susceptible and need vaccination; or are vaccinated,” the authors wrote.
Patients with previous HBV infection can have the infection reactivated with immunosuppressive treatments, Dr. Lee said, which is why detecting prior infection via the triple panel screening is important.
Women who are pregnant should be screened, ideally, in the first trimester of each pregnancy, regardless of vaccination status or testing history. If they have already received timely triple panel screening for hepatitis B and have no new HBV exposures, pregnant women only need HBsAg screening, the guidelines state.
The guidelines also specify that higher risk groups, specifically those incarcerated or formerly incarcerated, adults with current or past hepatitis C virus infection, and those with current or past sexually transmitted infections and multiple sex partners.
People who are susceptible for infection, refuse vaccination and are at higher risk for HBV should be screened periodically, but how often they should be screened should be based on shared decision-making between the provider and patient as well as individual risk and immune status.
Additional research into the optimal frequency of periodic testing is necessary, the authors say.
“Along with vaccination strategies, universal screening of adults and appropriate testing of persons at increased risk for HBV infection will improve health outcomes, reduce the prevalence of HBV infection in the United States, and advance viral hepatitis elimination goals,” the authors wrote.
The new recommendations now contrast with the 2020 screening guidelines issued by the U.S. Preventive Services Task Force (USPSTF) that recommend risk-based screening for hepatitis B.
“When that recommendation was published, the Task Force was aligned with several other organizations, including the CDC, in supporting screening for hepatitis B in high-risk populations — and importantly, we’re all still aligned in making sure that people get the care that they need,” said Michael Barry, MD, chair of the USPSTF, in an emailed statement. “The evidence on clinical preventive services is always changing, and the Task Force aims to keep all recommendations current, updating each recommendation approximately every 5 years.”
“In the meantime, we always encourage clinicians to use their judgment as they provide care for their patients — including those who may benefit from screening for hepatitis B — and to decide together with each patient which preventive services can best help them live a long and healthy life,” Dr. Barry said.
The American Association for the Study of Liver Diseases is currently updating their HBV screening recommendations, Dr. Lee said, and he expects other professional societies to follow the CDC recommendations.
“It’s not uncommon that we see the CDC or societies making recommendations and the USPSTF following along, so hopefully that’s the case for hepatitis B as well,” he said.
The authors reported no potential conflicts of interest.
A version of this article originally appeared on Medscape.com.
This is the first update to HBV screening guidelines since 2008, the agency said.
“Risk-based testing alone has not identified most persons living with chronic HBV infection and is considered inefficient for providers to implement,” the authors wrote in the new guidance, published in the CDC’s Morbidity and Mortality Weekly Report. “Universal screening of adults for HBV infection is cost-effective, compared with risk-based screening and averts liver disease and death. Although a curative treatment is not yet available, early diagnosis and treatment of chronic HBV infections reduces the risk for cirrhosis, liver cancer, and death.”
Howard Lee, MD, an assistant professor in the section of gastroenterology and hepatology at Baylor College of Medicine in Houston, agreed that risk-based screening has not been effective. A universal screening approach “is the way to go,” he said. With this new screening approach, patients can get tested without having to admit that they may be at risk for a chronic disease like HIV and HBV, which can be stigmatizing, said Dr. Lee, who was not involved with making these recommendations.
An estimated 580,000 to 2.4 million individuals are living with HBV infection in the United States, and two-thirds may be unaware they are infected, according to the CDC. The virus spreads through contact with blood, semen, and other body fluids of an infected person.
The guidance now recommends using the triple panel (HBsAg, anti-HBs, total anti-HBc) for initial screening.
“It can help identify persons who have an active HBV infection and could be linked to care; have resolved infection and might be susceptible to reactivation (for example, immunosuppressed persons); are susceptible and need vaccination; or are vaccinated,” the authors wrote.
Patients with previous HBV infection can have the infection reactivated with immunosuppressive treatments, Dr. Lee said, which is why detecting prior infection via the triple panel screening is important.
Women who are pregnant should be screened, ideally, in the first trimester of each pregnancy, regardless of vaccination status or testing history. If they have already received timely triple panel screening for hepatitis B and have no new HBV exposures, pregnant women only need HBsAg screening, the guidelines state.
The guidelines also specify that higher risk groups, specifically those incarcerated or formerly incarcerated, adults with current or past hepatitis C virus infection, and those with current or past sexually transmitted infections and multiple sex partners.
People who are susceptible for infection, refuse vaccination and are at higher risk for HBV should be screened periodically, but how often they should be screened should be based on shared decision-making between the provider and patient as well as individual risk and immune status.
Additional research into the optimal frequency of periodic testing is necessary, the authors say.
“Along with vaccination strategies, universal screening of adults and appropriate testing of persons at increased risk for HBV infection will improve health outcomes, reduce the prevalence of HBV infection in the United States, and advance viral hepatitis elimination goals,” the authors wrote.
The new recommendations now contrast with the 2020 screening guidelines issued by the U.S. Preventive Services Task Force (USPSTF) that recommend risk-based screening for hepatitis B.
“When that recommendation was published, the Task Force was aligned with several other organizations, including the CDC, in supporting screening for hepatitis B in high-risk populations — and importantly, we’re all still aligned in making sure that people get the care that they need,” said Michael Barry, MD, chair of the USPSTF, in an emailed statement. “The evidence on clinical preventive services is always changing, and the Task Force aims to keep all recommendations current, updating each recommendation approximately every 5 years.”
“In the meantime, we always encourage clinicians to use their judgment as they provide care for their patients — including those who may benefit from screening for hepatitis B — and to decide together with each patient which preventive services can best help them live a long and healthy life,” Dr. Barry said.
The American Association for the Study of Liver Diseases is currently updating their HBV screening recommendations, Dr. Lee said, and he expects other professional societies to follow the CDC recommendations.
“It’s not uncommon that we see the CDC or societies making recommendations and the USPSTF following along, so hopefully that’s the case for hepatitis B as well,” he said.
The authors reported no potential conflicts of interest.
A version of this article originally appeared on Medscape.com.
Posttransplant NASH patients fare worse with older donor livers
All-cause mortality was twice as high and death from an infectious cause was more than three times as high for patients with NASH who received liver grafts from octogenarian donors than for those who received a liver from someone younger than 50.
“The findings from this study implicate a critical need to investigate donor age as an important risk factor for poorer host and graft survival,” wrote the authors, led by David Lee, MD, of the University of Maryland, Baltimore.
“Given the possibility of infectious graft complications, post–liver transplant follow-ups may need to be more comprehensive and frequent in these individuals who receive grafts from older donors,” they add.
The study was published online in Digestive and Liver Disease.
Donor age trends
Dr. Lee and colleagues pulled data from the United Network for Organ Sharing (UNOS) Standard Transplant Analysis and Research database, a national database of deidentified donor and recipient transplant data. The analysis excluded recipients younger than 18, those with a living donor, those who had hepatocellular carcinoma prior to transplant, and those who had been diagnosed with additional liver disorders apart from NASH.
The team identified 8,88 recipients with NASH who received a liver transplant from 2005–2019. They stratified recipients by donor age. The 5,187 patients who received livers from donors who were younger than 50 served as the reference group. The remainder were placed into four cohorts – 1,842 whose donors were in their 50s, 1,290 whose donors were in their 60s, 504 whose donors were in their 70s, and 65 whose donors were in their 80s.
The researchers found that in comparison with the reference group, the average age of recipients in each donor-age cohort was progressively older. Two donor-age cohorts had significantly higher proportions of recipients with diabetes than the 46.5% in the reference group – the sexagenarian cohort (51.7%) and the octogenarian group (66.2%).
The median follow-up time ranged from 2.35–3.61 years across all age groups.
The researchers found that for all donor-age groups excluding donors in their 60s, recipients had higher risk of all-cause mortality after transplant than the reference group. Recipients with donors in their 50s had a 16% greater risk for death (P = .01), and recipients with donors in their 70s had a 20% greater risk (P = .05). For recipients with octogenarian donors, the adjusted hazard ratio for all-cause mortality was 2.01 (P < .001).
Only recipients in the octogenarian donor cohort were at increased risk of graft failure, compared with the reference group (aHR, 3.72; P = .002).
As donor age increased, the recipient’s risk of dying from sepsis and infectious causes rose, compared with the reference group. Recipients’ likelihood of sepsis death increased by 71% (P = .001) with donors in their 50s, 73% (P = .003) with donors in their 60s, and 76% (P = .03) with donors in their 70s. For recipients with octogenarian donors, the risk more than tripled (aHR, 3.58; P = .007). Likewise, recipients with donors in their 70s were 73% more likely to die from infectious causes. That risk nearly quadrupled among those with donors in their 80s.
Recipient factors at play?
While the study found a relationship between liver donor age and recipient outcomes, it is not clear whether any other recipient factors may have contributed to the higher risk of all-cause mortality, sad Nancy Reau, MD, chief of the hepatology section at Rush University Medical Center, Chicago. The researchers did not parse out whether younger recipients did better with older organs than older recipients or whether older recipients fared worse with younger organs, she said in an interview.
“I wasn’t convinced that they had demonstrated that the recipient may not have played a role in that,” said Dr. Reau, who wasn’t involved with the study.
The analysis only a found an increased risk of graft failure among recipients who received organs from octogenarian donors, so factors other than liver transplant may have contributed to all-cause mortality, she noted.
The UNOS database has some limitations, noted Timothy Pruett, MD, who directs the liver transplant program at the University of Minnesota, Minneapolis. Because the database pulls information from transplantation centers across the country, it can be difficult to standardize specific patient variables in the data.
While it’s clear that a patient died, it’s less certain whether an infection was the cause of death and whether that infection was somehow associated with the liver, noted Dr. Pruett, who wasn’t involved in the research. For example, a patient could have had broken a hip, gone to the hospital, and contracted pneumonia, which led to their death.
“There’s just not much granularity in the database, and we can’t overextrapolate what we see,” Dr. Pruett said in an interview.
Knowledge gaps
Dr. Lee agreed that more research is needed to understand what may be driving higher mortality rates among patients who receive older organs. “There are still a lot of gaps in knowledge with respect to why.”
Dr. Reau said she is curious as to whether certain comorbidities, such as previous infection, diabetes, or obesity, could predict worse outcomes for recipients with older organs.
“We would love to give all of our patients younger organs, but if that leads to even more disparity in need [compared] to availability and the alternative is not surviving, I think you have to place [this work] into context,” she said.
The study findings shouldn’t be used to deter patients with NASH from considering older organs, Dr. Reau said. More insight as to which populations might want to be choosier owing to an elevated risk would be beneficial, she added.
Dr. Lee, Dr. Pruett, and Dr. Reau reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
All-cause mortality was twice as high and death from an infectious cause was more than three times as high for patients with NASH who received liver grafts from octogenarian donors than for those who received a liver from someone younger than 50.
“The findings from this study implicate a critical need to investigate donor age as an important risk factor for poorer host and graft survival,” wrote the authors, led by David Lee, MD, of the University of Maryland, Baltimore.
“Given the possibility of infectious graft complications, post–liver transplant follow-ups may need to be more comprehensive and frequent in these individuals who receive grafts from older donors,” they add.
The study was published online in Digestive and Liver Disease.
Donor age trends
Dr. Lee and colleagues pulled data from the United Network for Organ Sharing (UNOS) Standard Transplant Analysis and Research database, a national database of deidentified donor and recipient transplant data. The analysis excluded recipients younger than 18, those with a living donor, those who had hepatocellular carcinoma prior to transplant, and those who had been diagnosed with additional liver disorders apart from NASH.
The team identified 8,88 recipients with NASH who received a liver transplant from 2005–2019. They stratified recipients by donor age. The 5,187 patients who received livers from donors who were younger than 50 served as the reference group. The remainder were placed into four cohorts – 1,842 whose donors were in their 50s, 1,290 whose donors were in their 60s, 504 whose donors were in their 70s, and 65 whose donors were in their 80s.
The researchers found that in comparison with the reference group, the average age of recipients in each donor-age cohort was progressively older. Two donor-age cohorts had significantly higher proportions of recipients with diabetes than the 46.5% in the reference group – the sexagenarian cohort (51.7%) and the octogenarian group (66.2%).
The median follow-up time ranged from 2.35–3.61 years across all age groups.
The researchers found that for all donor-age groups excluding donors in their 60s, recipients had higher risk of all-cause mortality after transplant than the reference group. Recipients with donors in their 50s had a 16% greater risk for death (P = .01), and recipients with donors in their 70s had a 20% greater risk (P = .05). For recipients with octogenarian donors, the adjusted hazard ratio for all-cause mortality was 2.01 (P < .001).
Only recipients in the octogenarian donor cohort were at increased risk of graft failure, compared with the reference group (aHR, 3.72; P = .002).
As donor age increased, the recipient’s risk of dying from sepsis and infectious causes rose, compared with the reference group. Recipients’ likelihood of sepsis death increased by 71% (P = .001) with donors in their 50s, 73% (P = .003) with donors in their 60s, and 76% (P = .03) with donors in their 70s. For recipients with octogenarian donors, the risk more than tripled (aHR, 3.58; P = .007). Likewise, recipients with donors in their 70s were 73% more likely to die from infectious causes. That risk nearly quadrupled among those with donors in their 80s.
Recipient factors at play?
While the study found a relationship between liver donor age and recipient outcomes, it is not clear whether any other recipient factors may have contributed to the higher risk of all-cause mortality, sad Nancy Reau, MD, chief of the hepatology section at Rush University Medical Center, Chicago. The researchers did not parse out whether younger recipients did better with older organs than older recipients or whether older recipients fared worse with younger organs, she said in an interview.
“I wasn’t convinced that they had demonstrated that the recipient may not have played a role in that,” said Dr. Reau, who wasn’t involved with the study.
The analysis only a found an increased risk of graft failure among recipients who received organs from octogenarian donors, so factors other than liver transplant may have contributed to all-cause mortality, she noted.
The UNOS database has some limitations, noted Timothy Pruett, MD, who directs the liver transplant program at the University of Minnesota, Minneapolis. Because the database pulls information from transplantation centers across the country, it can be difficult to standardize specific patient variables in the data.
While it’s clear that a patient died, it’s less certain whether an infection was the cause of death and whether that infection was somehow associated with the liver, noted Dr. Pruett, who wasn’t involved in the research. For example, a patient could have had broken a hip, gone to the hospital, and contracted pneumonia, which led to their death.
“There’s just not much granularity in the database, and we can’t overextrapolate what we see,” Dr. Pruett said in an interview.
Knowledge gaps
Dr. Lee agreed that more research is needed to understand what may be driving higher mortality rates among patients who receive older organs. “There are still a lot of gaps in knowledge with respect to why.”
Dr. Reau said she is curious as to whether certain comorbidities, such as previous infection, diabetes, or obesity, could predict worse outcomes for recipients with older organs.
“We would love to give all of our patients younger organs, but if that leads to even more disparity in need [compared] to availability and the alternative is not surviving, I think you have to place [this work] into context,” she said.
The study findings shouldn’t be used to deter patients with NASH from considering older organs, Dr. Reau said. More insight as to which populations might want to be choosier owing to an elevated risk would be beneficial, she added.
Dr. Lee, Dr. Pruett, and Dr. Reau reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
All-cause mortality was twice as high and death from an infectious cause was more than three times as high for patients with NASH who received liver grafts from octogenarian donors than for those who received a liver from someone younger than 50.
“The findings from this study implicate a critical need to investigate donor age as an important risk factor for poorer host and graft survival,” wrote the authors, led by David Lee, MD, of the University of Maryland, Baltimore.
“Given the possibility of infectious graft complications, post–liver transplant follow-ups may need to be more comprehensive and frequent in these individuals who receive grafts from older donors,” they add.
The study was published online in Digestive and Liver Disease.
Donor age trends
Dr. Lee and colleagues pulled data from the United Network for Organ Sharing (UNOS) Standard Transplant Analysis and Research database, a national database of deidentified donor and recipient transplant data. The analysis excluded recipients younger than 18, those with a living donor, those who had hepatocellular carcinoma prior to transplant, and those who had been diagnosed with additional liver disorders apart from NASH.
The team identified 8,88 recipients with NASH who received a liver transplant from 2005–2019. They stratified recipients by donor age. The 5,187 patients who received livers from donors who were younger than 50 served as the reference group. The remainder were placed into four cohorts – 1,842 whose donors were in their 50s, 1,290 whose donors were in their 60s, 504 whose donors were in their 70s, and 65 whose donors were in their 80s.
The researchers found that in comparison with the reference group, the average age of recipients in each donor-age cohort was progressively older. Two donor-age cohorts had significantly higher proportions of recipients with diabetes than the 46.5% in the reference group – the sexagenarian cohort (51.7%) and the octogenarian group (66.2%).
The median follow-up time ranged from 2.35–3.61 years across all age groups.
The researchers found that for all donor-age groups excluding donors in their 60s, recipients had higher risk of all-cause mortality after transplant than the reference group. Recipients with donors in their 50s had a 16% greater risk for death (P = .01), and recipients with donors in their 70s had a 20% greater risk (P = .05). For recipients with octogenarian donors, the adjusted hazard ratio for all-cause mortality was 2.01 (P < .001).
Only recipients in the octogenarian donor cohort were at increased risk of graft failure, compared with the reference group (aHR, 3.72; P = .002).
As donor age increased, the recipient’s risk of dying from sepsis and infectious causes rose, compared with the reference group. Recipients’ likelihood of sepsis death increased by 71% (P = .001) with donors in their 50s, 73% (P = .003) with donors in their 60s, and 76% (P = .03) with donors in their 70s. For recipients with octogenarian donors, the risk more than tripled (aHR, 3.58; P = .007). Likewise, recipients with donors in their 70s were 73% more likely to die from infectious causes. That risk nearly quadrupled among those with donors in their 80s.
Recipient factors at play?
While the study found a relationship between liver donor age and recipient outcomes, it is not clear whether any other recipient factors may have contributed to the higher risk of all-cause mortality, sad Nancy Reau, MD, chief of the hepatology section at Rush University Medical Center, Chicago. The researchers did not parse out whether younger recipients did better with older organs than older recipients or whether older recipients fared worse with younger organs, she said in an interview.
“I wasn’t convinced that they had demonstrated that the recipient may not have played a role in that,” said Dr. Reau, who wasn’t involved with the study.
The analysis only a found an increased risk of graft failure among recipients who received organs from octogenarian donors, so factors other than liver transplant may have contributed to all-cause mortality, she noted.
The UNOS database has some limitations, noted Timothy Pruett, MD, who directs the liver transplant program at the University of Minnesota, Minneapolis. Because the database pulls information from transplantation centers across the country, it can be difficult to standardize specific patient variables in the data.
While it’s clear that a patient died, it’s less certain whether an infection was the cause of death and whether that infection was somehow associated with the liver, noted Dr. Pruett, who wasn’t involved in the research. For example, a patient could have had broken a hip, gone to the hospital, and contracted pneumonia, which led to their death.
“There’s just not much granularity in the database, and we can’t overextrapolate what we see,” Dr. Pruett said in an interview.
Knowledge gaps
Dr. Lee agreed that more research is needed to understand what may be driving higher mortality rates among patients who receive older organs. “There are still a lot of gaps in knowledge with respect to why.”
Dr. Reau said she is curious as to whether certain comorbidities, such as previous infection, diabetes, or obesity, could predict worse outcomes for recipients with older organs.
“We would love to give all of our patients younger organs, but if that leads to even more disparity in need [compared] to availability and the alternative is not surviving, I think you have to place [this work] into context,” she said.
The study findings shouldn’t be used to deter patients with NASH from considering older organs, Dr. Reau said. More insight as to which populations might want to be choosier owing to an elevated risk would be beneficial, she added.
Dr. Lee, Dr. Pruett, and Dr. Reau reported no relevant financial relationships.
A version of this article first appeared on Medscape.com.
FROM DIGESTIVE AND LIVER DISEASE


