User login
Higher plasma cell-free DNA tracks with worse PAH survival
NEW ORLEANS – Cell-free (cf) DNA looked like an informative biomarker for both the severity of pulmonary artery hypertension and the survival prognosis for patients with this disease, based on results from two preliminary studies involving a total of 173 people.
“Plasma levels of cell-free DNA are elevated in patients with pulmonary artery hypertension, compared with healthy controls, and may predict disease severity and mortality,” Samuel B. Brusca, MD, said at the at the annual meeting of the American College of Cardiology.
A growing biomedical literature has documented a role for cfDNA in tracking the course of cancer, septic shock, and transplanted organs (Transplantation. 2019 Feb;103[2]:273-83) (Cell-Free DNA: Applications in Different Diseases, in “Cell-free DNA as Diagnostic Markers.” [New York: Humana Press, 2018, pp. 3-12]). Based on this background Dr. Brusca and his associates decided to examine whether plasma levels of cfDNA linked with pulmonary artery hypertension (PAH) severity and survival.
Their first study included seven patients with mild PAH (defined as patients with a tricuspid annular plane systolic excursion [TAPSE] of more than 18 mm and a maximum oxygen uptake [VO2] of at least 75% of predicted), eight with severe PAH (a TAPSE of 18 mm or less and a VO2 of less than 75%), and seven healthy adult controls. Measurement of plasma cfDNA showed an average level of 19.4 ng/mL among the healthy controls (prior reports had indicated that 10-20 ng/mL were normal levels), 22.0 ng/mL among patients with mild PAH, and 36.2 ng/mL in those with severe PAH. The level among the severe PAH patients was significantly higher than the level in controls by two different statistical tests, said Dr. Brusca, a critical care medicine physician at the National Institutes of Health Clinical Center in Bethesda, Md.
The second analysis by Dr. Brusca and his associates included 151 PAH patients followed by physicians at the Clinical Center for an average of 40 months. Their analysis tracked survival of these patients relative to their baseline levels of cfDNA and divided into tertiles. Patients in the lowest tertile had a starting cfDNA level of up to 39 ng/mL, those in the middle tertile had levels of 39.1-64.0 ng/mL, and those in the top tertile had levels of at least 64.1 ng/mL. A Kaplan-Meier analysis showed statistically significant differences in survival rates between each of the tertiles. Patients in the lowest tertile had a 5-year actuarial survival rate of about 65%, those in the middle tertile had a survival rate of about 48%, and those in the tertile with the highest level of cfDNA had a survival rate of about 28%.
Additional studies of cfDNA are needed in larger numbers of PAH patients, and cfDNA levels should be compared with levels of other, more established biomarkers, such as inflammatory cytokines, Dr. Brusca said in an interview.
Dr. Brusca had no disclosures. The study received no commercial funding.
SOURCE: Brusca SB et al. J Am Coll Cardiol. 2019 March 12;73(9 Suppl 1):1897.
I was very excited to hear Dr. Brusca’s report on using cell-free (cf) DNA to track the severity of pulmonary artery hypertension and survival of these patients. I’m now using cfDNA frequently to monitor heart transplant patients, and the information it provides has been very valuable. But cfDNA may be even better suited to assessing patients with pulmonary artery hypertension (PAH) because it’s a vascular disease, and increases in cfDNA appears to reflect damage to the vascular endothelium. It’s a brilliant application of this technology. Brain natriuretic peptide and troponin are markers of right heart damage, but cfDNA appears to be able to track the progression of the vascular component of PAH. It appears to be the first disease-specific biomarker we have for PAH. It’s time to start routinely measuring levels of cfDNA in trials so we can gather more data on the clinical correlates of changing levels of this biomarker.
I was very excited to hear Dr. Brusca’s report on using cell-free (cf) DNA to track the severity of pulmonary artery hypertension and survival of these patients. I’m now using cfDNA frequently to monitor heart transplant patients, and the information it provides has been very valuable. But cfDNA may be even better suited to assessing patients with pulmonary artery hypertension (PAH) because it’s a vascular disease, and increases in cfDNA appears to reflect damage to the vascular endothelium. It’s a brilliant application of this technology. Brain natriuretic peptide and troponin are markers of right heart damage, but cfDNA appears to be able to track the progression of the vascular component of PAH. It appears to be the first disease-specific biomarker we have for PAH. It’s time to start routinely measuring levels of cfDNA in trials so we can gather more data on the clinical correlates of changing levels of this biomarker.
I was very excited to hear Dr. Brusca’s report on using cell-free (cf) DNA to track the severity of pulmonary artery hypertension and survival of these patients. I’m now using cfDNA frequently to monitor heart transplant patients, and the information it provides has been very valuable. But cfDNA may be even better suited to assessing patients with pulmonary artery hypertension (PAH) because it’s a vascular disease, and increases in cfDNA appears to reflect damage to the vascular endothelium. It’s a brilliant application of this technology. Brain natriuretic peptide and troponin are markers of right heart damage, but cfDNA appears to be able to track the progression of the vascular component of PAH. It appears to be the first disease-specific biomarker we have for PAH. It’s time to start routinely measuring levels of cfDNA in trials so we can gather more data on the clinical correlates of changing levels of this biomarker.
NEW ORLEANS – Cell-free (cf) DNA looked like an informative biomarker for both the severity of pulmonary artery hypertension and the survival prognosis for patients with this disease, based on results from two preliminary studies involving a total of 173 people.
“Plasma levels of cell-free DNA are elevated in patients with pulmonary artery hypertension, compared with healthy controls, and may predict disease severity and mortality,” Samuel B. Brusca, MD, said at the at the annual meeting of the American College of Cardiology.
A growing biomedical literature has documented a role for cfDNA in tracking the course of cancer, septic shock, and transplanted organs (Transplantation. 2019 Feb;103[2]:273-83) (Cell-Free DNA: Applications in Different Diseases, in “Cell-free DNA as Diagnostic Markers.” [New York: Humana Press, 2018, pp. 3-12]). Based on this background Dr. Brusca and his associates decided to examine whether plasma levels of cfDNA linked with pulmonary artery hypertension (PAH) severity and survival.
Their first study included seven patients with mild PAH (defined as patients with a tricuspid annular plane systolic excursion [TAPSE] of more than 18 mm and a maximum oxygen uptake [VO2] of at least 75% of predicted), eight with severe PAH (a TAPSE of 18 mm or less and a VO2 of less than 75%), and seven healthy adult controls. Measurement of plasma cfDNA showed an average level of 19.4 ng/mL among the healthy controls (prior reports had indicated that 10-20 ng/mL were normal levels), 22.0 ng/mL among patients with mild PAH, and 36.2 ng/mL in those with severe PAH. The level among the severe PAH patients was significantly higher than the level in controls by two different statistical tests, said Dr. Brusca, a critical care medicine physician at the National Institutes of Health Clinical Center in Bethesda, Md.
The second analysis by Dr. Brusca and his associates included 151 PAH patients followed by physicians at the Clinical Center for an average of 40 months. Their analysis tracked survival of these patients relative to their baseline levels of cfDNA and divided into tertiles. Patients in the lowest tertile had a starting cfDNA level of up to 39 ng/mL, those in the middle tertile had levels of 39.1-64.0 ng/mL, and those in the top tertile had levels of at least 64.1 ng/mL. A Kaplan-Meier analysis showed statistically significant differences in survival rates between each of the tertiles. Patients in the lowest tertile had a 5-year actuarial survival rate of about 65%, those in the middle tertile had a survival rate of about 48%, and those in the tertile with the highest level of cfDNA had a survival rate of about 28%.
Additional studies of cfDNA are needed in larger numbers of PAH patients, and cfDNA levels should be compared with levels of other, more established biomarkers, such as inflammatory cytokines, Dr. Brusca said in an interview.
Dr. Brusca had no disclosures. The study received no commercial funding.
SOURCE: Brusca SB et al. J Am Coll Cardiol. 2019 March 12;73(9 Suppl 1):1897.
NEW ORLEANS – Cell-free (cf) DNA looked like an informative biomarker for both the severity of pulmonary artery hypertension and the survival prognosis for patients with this disease, based on results from two preliminary studies involving a total of 173 people.
“Plasma levels of cell-free DNA are elevated in patients with pulmonary artery hypertension, compared with healthy controls, and may predict disease severity and mortality,” Samuel B. Brusca, MD, said at the at the annual meeting of the American College of Cardiology.
A growing biomedical literature has documented a role for cfDNA in tracking the course of cancer, septic shock, and transplanted organs (Transplantation. 2019 Feb;103[2]:273-83) (Cell-Free DNA: Applications in Different Diseases, in “Cell-free DNA as Diagnostic Markers.” [New York: Humana Press, 2018, pp. 3-12]). Based on this background Dr. Brusca and his associates decided to examine whether plasma levels of cfDNA linked with pulmonary artery hypertension (PAH) severity and survival.
Their first study included seven patients with mild PAH (defined as patients with a tricuspid annular plane systolic excursion [TAPSE] of more than 18 mm and a maximum oxygen uptake [VO2] of at least 75% of predicted), eight with severe PAH (a TAPSE of 18 mm or less and a VO2 of less than 75%), and seven healthy adult controls. Measurement of plasma cfDNA showed an average level of 19.4 ng/mL among the healthy controls (prior reports had indicated that 10-20 ng/mL were normal levels), 22.0 ng/mL among patients with mild PAH, and 36.2 ng/mL in those with severe PAH. The level among the severe PAH patients was significantly higher than the level in controls by two different statistical tests, said Dr. Brusca, a critical care medicine physician at the National Institutes of Health Clinical Center in Bethesda, Md.
The second analysis by Dr. Brusca and his associates included 151 PAH patients followed by physicians at the Clinical Center for an average of 40 months. Their analysis tracked survival of these patients relative to their baseline levels of cfDNA and divided into tertiles. Patients in the lowest tertile had a starting cfDNA level of up to 39 ng/mL, those in the middle tertile had levels of 39.1-64.0 ng/mL, and those in the top tertile had levels of at least 64.1 ng/mL. A Kaplan-Meier analysis showed statistically significant differences in survival rates between each of the tertiles. Patients in the lowest tertile had a 5-year actuarial survival rate of about 65%, those in the middle tertile had a survival rate of about 48%, and those in the tertile with the highest level of cfDNA had a survival rate of about 28%.
Additional studies of cfDNA are needed in larger numbers of PAH patients, and cfDNA levels should be compared with levels of other, more established biomarkers, such as inflammatory cytokines, Dr. Brusca said in an interview.
Dr. Brusca had no disclosures. The study received no commercial funding.
SOURCE: Brusca SB et al. J Am Coll Cardiol. 2019 March 12;73(9 Suppl 1):1897.
REPORTING FROM ACC 2019
CardioMEMS cuts heart failure hospitalizations in post-approval study
NEW ORLEANS – Frequent, noninvasive measurement of pulmonary artery pressure in patients with advanced heart failure and an implanted CardioMEMS device that allows this measurement led to management that produced a substantial reduction in heart failure hospitalizations, compared with each patient’s history, in a real-world study.
The Food and Drug Administration–mandated CardioMEMS Post-Approval Study included 1,200 patients who received CardioMEMS implants after it received U.S. marketing approval. The study showed that when clinicians and patients used the device in routine practice, presumably as part of a structured management system designed to take advantage of the pulmonary artery (PA) pressures the device provides, the result safely produced a 58% cut in heart failure hospitalizations during the year following device placement when compared to each patient’s own hospitalization history during the year before they got the CardioMEMS device, David M. Shavelle, MD, said at the at the annual meeting of the American College of Cardiology. This statistically significant result for the study’s primary endpoint showed an absolute reduction in the average rate of heart failure hospitalizations from 1.24 per patient during the year before the CardioMEMS placement to 0.52 hospitalizations per patient during the 12 months after placement, an average reduction of 0.72 hospitalizations/patient, said Dr. Shavelle, an interventional cardiologist at the University of Southern California in Los Angeles.
Another notable finding was that this benefit from CardioMEMS placement and use occurred at roughly similar rates in patients with New York Heart Association class III heart failure regardless of whether they had a reduced ejection fraction (40% or less), a mid-range ejection fraction (41%-50%), or preserved ejection fraction (greater than 50%), making CardioMEMS use one of the few treatments to produce any proven benefit in patients with heart failure with preserved ejection fraction. In that subgroup, 30% of the 1,200 enrolled patients had an average cut of 0.68 hospitalizations in the year after CardioMEMS implantation, a 61% drop, relative to the year before they received the device.
The results also fulfilled the study’s two prespecified safety measures. Among the 1,214 patients in the study assessed for safety, which included the 1,200 patients who received the device and 4 patients in whom placement failed, 4 patients had a device or system related complication during the study, a 0.3% rate, compared with a prespecified objective performance criteria of less than 20%. Among the 1,200 patients with a functioning CardioMEMS sensor, one patient (0.1%) had a device failure, compared with the study’s objective performance criteria of less than 10%.
The performance of the CardioMEMS device and the benefit it provided to patients in the post-approval study closely tracked its performance during the published pivotal trial (Lancet. 2011 Feb 19;377[9766]:658-66). On the basis of the pivotal trial results, the FDA approved CardioMEMS for U.S. marketing in 2014. Since then, the company has reported that about 10,000 U.S. heart failure patients have received these devices, Dr. Shavelle said.
“The benefit was seen across the range of ejection fractions; that’s very important,” commented Gurusher Panjrath, MD, director of advanced heart failure at George Washington University in Washington and a designated discussant for Dr. Shavelle’s report. “The safety seemed very good, and the efficacy was consistent” with prior reports. “There also was high compliance. The key to success is the structure” of patient management, Dr. Pangroth said. “The data are limited by who is monitoring patients and their data and how much of that contact influences patient outcomes.”
That final comment by Dr. Panjrath highlighted the biggest caveat that heart failure clinicians have raised about judging the efficacy of CardioMEMS. To achieve clinical efficacy, the implanted device requires diligent, virtually daily interrogation and data transmission by the patient, assessment of a large amount of data for each patient by the patient’s clinical team, and responsiveness by the patient to medication adjustments directed by the clinical team to deal with episodes of rising PA pressure.
“The device itself has no benefit. It’s the actions prompted by the device that have benefit,” noted Clyde W. Yancy, MD, professor of medicine and chief of cardiology at Northwestern University in Chicago and a second designated discussant for the report.
Dr. Shavelle agreed that for the CardioMEMS device to have an impact, one basic requirement is to identify patients who will cooperate with data collection and transmission and also with changes in their medications that are sent to them in response to PA pressure changes. This means selecting patients who appear to have problems with volume overload, including prior hospitalizations for decompensation, and patients who are comfortable interacting with their clinical-care providers. It also means excluding patients who are too sick to benefit from this intervention. He estimated that at his center more than 95% of class III heart failure patients who qualified for inclusion in the post-approval study by clinical criteria were also judged reasonable recipients of the device based on their willingness to cooperate with this system. He also estimated that at the University of Southern California the heart failure clinical team is now caring for about 150 patients with a CardioMEMS device implanted.
Another concern is teasing apart the specific benefit of collecting and using PA pressure data from the contact that the clinical team maintains with CardioMEMS patients.
“If nurses are contacting patients more often, is it the device or the communication? We need to look at that very carefully in a study that had no control group,” Dr. Yancy said in an interview. Contact with a nurse “is the best thing you can do for heart failure patients.”
Dr. Shavelle countered that several reports from past studies that assessed case management and regular monitoring of and contact with heart failure patients but without PA pressure data failed to showed any consistent benefit to patients.
“If you pick the right patients, CardioMEMS works. There is no question in my mind that the device works,” Dr. Shavelle said in an interview. “If you pick the wrong patient, who will not send the data or follow dose changes, then it won’t work.”
The study was sponsored by Abbott, the company that markets the CardioMEMS HF System. Dr. Shavelle has been a consultant to and speaker on behalf of Abbott Vascular and he has received research funding from Abbott Vascular, Abiomed, Biocardia, and V-Wave. Dr. Yancy had an unspecified financial relationship with Abbott Laboratories. Dr. Panjrath had no disclosures.
mzoler@mdedge.com
On Twitter @mitchelzoler
SOURCE: Shavelle DM et al. American College of Cardiology annual meeting, abstract 405-16.
NEW ORLEANS – Frequent, noninvasive measurement of pulmonary artery pressure in patients with advanced heart failure and an implanted CardioMEMS device that allows this measurement led to management that produced a substantial reduction in heart failure hospitalizations, compared with each patient’s history, in a real-world study.
The Food and Drug Administration–mandated CardioMEMS Post-Approval Study included 1,200 patients who received CardioMEMS implants after it received U.S. marketing approval. The study showed that when clinicians and patients used the device in routine practice, presumably as part of a structured management system designed to take advantage of the pulmonary artery (PA) pressures the device provides, the result safely produced a 58% cut in heart failure hospitalizations during the year following device placement when compared to each patient’s own hospitalization history during the year before they got the CardioMEMS device, David M. Shavelle, MD, said at the at the annual meeting of the American College of Cardiology. This statistically significant result for the study’s primary endpoint showed an absolute reduction in the average rate of heart failure hospitalizations from 1.24 per patient during the year before the CardioMEMS placement to 0.52 hospitalizations per patient during the 12 months after placement, an average reduction of 0.72 hospitalizations/patient, said Dr. Shavelle, an interventional cardiologist at the University of Southern California in Los Angeles.
Another notable finding was that this benefit from CardioMEMS placement and use occurred at roughly similar rates in patients with New York Heart Association class III heart failure regardless of whether they had a reduced ejection fraction (40% or less), a mid-range ejection fraction (41%-50%), or preserved ejection fraction (greater than 50%), making CardioMEMS use one of the few treatments to produce any proven benefit in patients with heart failure with preserved ejection fraction. In that subgroup, 30% of the 1,200 enrolled patients had an average cut of 0.68 hospitalizations in the year after CardioMEMS implantation, a 61% drop, relative to the year before they received the device.
The results also fulfilled the study’s two prespecified safety measures. Among the 1,214 patients in the study assessed for safety, which included the 1,200 patients who received the device and 4 patients in whom placement failed, 4 patients had a device or system related complication during the study, a 0.3% rate, compared with a prespecified objective performance criteria of less than 20%. Among the 1,200 patients with a functioning CardioMEMS sensor, one patient (0.1%) had a device failure, compared with the study’s objective performance criteria of less than 10%.
The performance of the CardioMEMS device and the benefit it provided to patients in the post-approval study closely tracked its performance during the published pivotal trial (Lancet. 2011 Feb 19;377[9766]:658-66). On the basis of the pivotal trial results, the FDA approved CardioMEMS for U.S. marketing in 2014. Since then, the company has reported that about 10,000 U.S. heart failure patients have received these devices, Dr. Shavelle said.
“The benefit was seen across the range of ejection fractions; that’s very important,” commented Gurusher Panjrath, MD, director of advanced heart failure at George Washington University in Washington and a designated discussant for Dr. Shavelle’s report. “The safety seemed very good, and the efficacy was consistent” with prior reports. “There also was high compliance. The key to success is the structure” of patient management, Dr. Pangroth said. “The data are limited by who is monitoring patients and their data and how much of that contact influences patient outcomes.”
That final comment by Dr. Panjrath highlighted the biggest caveat that heart failure clinicians have raised about judging the efficacy of CardioMEMS. To achieve clinical efficacy, the implanted device requires diligent, virtually daily interrogation and data transmission by the patient, assessment of a large amount of data for each patient by the patient’s clinical team, and responsiveness by the patient to medication adjustments directed by the clinical team to deal with episodes of rising PA pressure.
“The device itself has no benefit. It’s the actions prompted by the device that have benefit,” noted Clyde W. Yancy, MD, professor of medicine and chief of cardiology at Northwestern University in Chicago and a second designated discussant for the report.
Dr. Shavelle agreed that for the CardioMEMS device to have an impact, one basic requirement is to identify patients who will cooperate with data collection and transmission and also with changes in their medications that are sent to them in response to PA pressure changes. This means selecting patients who appear to have problems with volume overload, including prior hospitalizations for decompensation, and patients who are comfortable interacting with their clinical-care providers. It also means excluding patients who are too sick to benefit from this intervention. He estimated that at his center more than 95% of class III heart failure patients who qualified for inclusion in the post-approval study by clinical criteria were also judged reasonable recipients of the device based on their willingness to cooperate with this system. He also estimated that at the University of Southern California the heart failure clinical team is now caring for about 150 patients with a CardioMEMS device implanted.
Another concern is teasing apart the specific benefit of collecting and using PA pressure data from the contact that the clinical team maintains with CardioMEMS patients.
“If nurses are contacting patients more often, is it the device or the communication? We need to look at that very carefully in a study that had no control group,” Dr. Yancy said in an interview. Contact with a nurse “is the best thing you can do for heart failure patients.”
Dr. Shavelle countered that several reports from past studies that assessed case management and regular monitoring of and contact with heart failure patients but without PA pressure data failed to showed any consistent benefit to patients.
“If you pick the right patients, CardioMEMS works. There is no question in my mind that the device works,” Dr. Shavelle said in an interview. “If you pick the wrong patient, who will not send the data or follow dose changes, then it won’t work.”
The study was sponsored by Abbott, the company that markets the CardioMEMS HF System. Dr. Shavelle has been a consultant to and speaker on behalf of Abbott Vascular and he has received research funding from Abbott Vascular, Abiomed, Biocardia, and V-Wave. Dr. Yancy had an unspecified financial relationship with Abbott Laboratories. Dr. Panjrath had no disclosures.
mzoler@mdedge.com
On Twitter @mitchelzoler
SOURCE: Shavelle DM et al. American College of Cardiology annual meeting, abstract 405-16.
NEW ORLEANS – Frequent, noninvasive measurement of pulmonary artery pressure in patients with advanced heart failure and an implanted CardioMEMS device that allows this measurement led to management that produced a substantial reduction in heart failure hospitalizations, compared with each patient’s history, in a real-world study.
The Food and Drug Administration–mandated CardioMEMS Post-Approval Study included 1,200 patients who received CardioMEMS implants after it received U.S. marketing approval. The study showed that when clinicians and patients used the device in routine practice, presumably as part of a structured management system designed to take advantage of the pulmonary artery (PA) pressures the device provides, the result safely produced a 58% cut in heart failure hospitalizations during the year following device placement when compared to each patient’s own hospitalization history during the year before they got the CardioMEMS device, David M. Shavelle, MD, said at the at the annual meeting of the American College of Cardiology. This statistically significant result for the study’s primary endpoint showed an absolute reduction in the average rate of heart failure hospitalizations from 1.24 per patient during the year before the CardioMEMS placement to 0.52 hospitalizations per patient during the 12 months after placement, an average reduction of 0.72 hospitalizations/patient, said Dr. Shavelle, an interventional cardiologist at the University of Southern California in Los Angeles.
Another notable finding was that this benefit from CardioMEMS placement and use occurred at roughly similar rates in patients with New York Heart Association class III heart failure regardless of whether they had a reduced ejection fraction (40% or less), a mid-range ejection fraction (41%-50%), or preserved ejection fraction (greater than 50%), making CardioMEMS use one of the few treatments to produce any proven benefit in patients with heart failure with preserved ejection fraction. In that subgroup, 30% of the 1,200 enrolled patients had an average cut of 0.68 hospitalizations in the year after CardioMEMS implantation, a 61% drop, relative to the year before they received the device.
The results also fulfilled the study’s two prespecified safety measures. Among the 1,214 patients in the study assessed for safety, which included the 1,200 patients who received the device and 4 patients in whom placement failed, 4 patients had a device or system related complication during the study, a 0.3% rate, compared with a prespecified objective performance criteria of less than 20%. Among the 1,200 patients with a functioning CardioMEMS sensor, one patient (0.1%) had a device failure, compared with the study’s objective performance criteria of less than 10%.
The performance of the CardioMEMS device and the benefit it provided to patients in the post-approval study closely tracked its performance during the published pivotal trial (Lancet. 2011 Feb 19;377[9766]:658-66). On the basis of the pivotal trial results, the FDA approved CardioMEMS for U.S. marketing in 2014. Since then, the company has reported that about 10,000 U.S. heart failure patients have received these devices, Dr. Shavelle said.
“The benefit was seen across the range of ejection fractions; that’s very important,” commented Gurusher Panjrath, MD, director of advanced heart failure at George Washington University in Washington and a designated discussant for Dr. Shavelle’s report. “The safety seemed very good, and the efficacy was consistent” with prior reports. “There also was high compliance. The key to success is the structure” of patient management, Dr. Pangroth said. “The data are limited by who is monitoring patients and their data and how much of that contact influences patient outcomes.”
That final comment by Dr. Panjrath highlighted the biggest caveat that heart failure clinicians have raised about judging the efficacy of CardioMEMS. To achieve clinical efficacy, the implanted device requires diligent, virtually daily interrogation and data transmission by the patient, assessment of a large amount of data for each patient by the patient’s clinical team, and responsiveness by the patient to medication adjustments directed by the clinical team to deal with episodes of rising PA pressure.
“The device itself has no benefit. It’s the actions prompted by the device that have benefit,” noted Clyde W. Yancy, MD, professor of medicine and chief of cardiology at Northwestern University in Chicago and a second designated discussant for the report.
Dr. Shavelle agreed that for the CardioMEMS device to have an impact, one basic requirement is to identify patients who will cooperate with data collection and transmission and also with changes in their medications that are sent to them in response to PA pressure changes. This means selecting patients who appear to have problems with volume overload, including prior hospitalizations for decompensation, and patients who are comfortable interacting with their clinical-care providers. It also means excluding patients who are too sick to benefit from this intervention. He estimated that at his center more than 95% of class III heart failure patients who qualified for inclusion in the post-approval study by clinical criteria were also judged reasonable recipients of the device based on their willingness to cooperate with this system. He also estimated that at the University of Southern California the heart failure clinical team is now caring for about 150 patients with a CardioMEMS device implanted.
Another concern is teasing apart the specific benefit of collecting and using PA pressure data from the contact that the clinical team maintains with CardioMEMS patients.
“If nurses are contacting patients more often, is it the device or the communication? We need to look at that very carefully in a study that had no control group,” Dr. Yancy said in an interview. Contact with a nurse “is the best thing you can do for heart failure patients.”
Dr. Shavelle countered that several reports from past studies that assessed case management and regular monitoring of and contact with heart failure patients but without PA pressure data failed to showed any consistent benefit to patients.
“If you pick the right patients, CardioMEMS works. There is no question in my mind that the device works,” Dr. Shavelle said in an interview. “If you pick the wrong patient, who will not send the data or follow dose changes, then it won’t work.”
The study was sponsored by Abbott, the company that markets the CardioMEMS HF System. Dr. Shavelle has been a consultant to and speaker on behalf of Abbott Vascular and he has received research funding from Abbott Vascular, Abiomed, Biocardia, and V-Wave. Dr. Yancy had an unspecified financial relationship with Abbott Laboratories. Dr. Panjrath had no disclosures.
mzoler@mdedge.com
On Twitter @mitchelzoler
SOURCE: Shavelle DM et al. American College of Cardiology annual meeting, abstract 405-16.
REPORTING FROM ACC 2019
SGLT2 inhibitors prevent HF hospitalization regardless of baseline LVEF
NEW ORLEANS – based on data from a large real-world patient registry.
“The observed beneficial effects of SGLT2 inhibitors on heart failure may extend across the range of baseline ejection fractions,” Mikhail Kosiborod, MD, observed at the annual meeting of the American College of Cardiology.
This is an important new insight. The major randomized cardiovascular outcome trials that showed lower risks of heart failure hospitalization and all-cause mortality in type 2 diabetic patients on an SGLT2 inhibitor, such as EMPA-REG OUTCOME for empagliflozin (Jardiance) and CANVAS for canagliflozin (Invokana), didn’t include information on baseline LVEF. So until now it has been unclear whether the beneficial effects of the SGLT2 inhibitors preventing heart failure hospitalization vary depending upon LVEF, explained Dr. Kosiborod, a cardiologist at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
He presented an analysis drawn from the patient database kept by Maccabi Healthcare Services in Israel. The study included 5,307 patients with type 2 diabetes and an LVEF measurement recorded in their chart at the time they started on either empagliflozin or dapagliflozin (Farxiga) and an equal number of propensity-matched type 2 diabetic controls who started on other glucose-lowering drugs, most commonly an oral dipeptidyl peptidase-4 inhibitor.
During roughly 16,000 person-years of follow-up, 239 deaths occurred. Compared with patients on another glucose-lowering drug, the risk of death from all causes was reduced by 47% among patients who were on an SGLT2 inhibitor and had a baseline LVEF of 50% or greater and by 62% among the 9% of subjects who had a baseline LVEF less than 50%.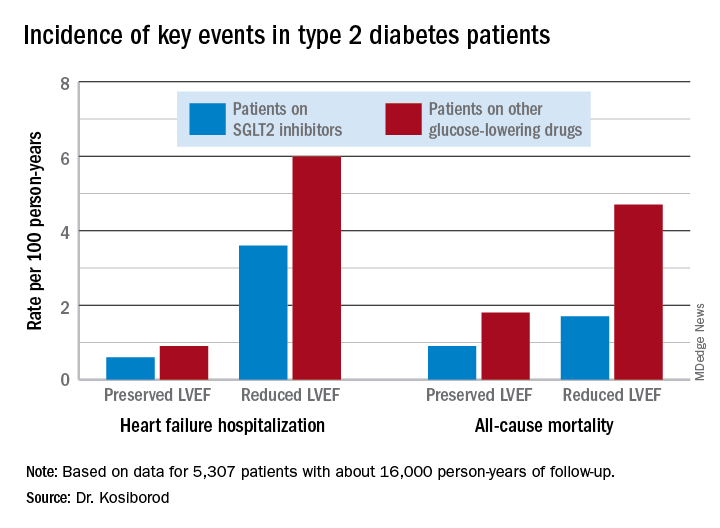
Similarly, the risk of heart failure hospitalization was reduced by 29% in SGLT2 inhibitor users with a preserved LVEF and by 27% if they had a reduced LVEF.
For the composite endpoint of heart failure hospitalization or all-cause mortality, the risk reductions associated with SGLT2 inhibitor therapy were 45% with preserved and 39% with reduced LVEF.
Session comoderator Prakash C. Deedwania, MD, noted that there are ongoing major randomized trials of various SGLT2 inhibitors in patients with known heart failure, with cardiovascular death and heart failure hospitalization as primary endpoints. He asked Dr. Kosiborod whether, given that the results of these studies aren’t in yet, he thinks clinicians should be prescribing SGLT2 inhibitors to diabetic or prediabetic patients who don’t have clinical symptoms of heart failure but may have a marker of increased risk, such as an elevated B-type natriuretic peptide.
“At least in my mind, we have more than enough evidence at this point to say that SGLT2 inhibitors are effective in preventing heart failure,” Dr. Kosiborod replied.
“Obviously, if your risk for developing a condition is higher at baseline, then the absolute benefit that you’re going to get from using an agent that’s effective in preventing that event is going to be higher and the number needed to treat is going to be lower. So if you have a patient at high risk for heart failure by whatever risk predictor you’re using and the patient doesn’t yet have heart failure but does have diabetes, which is already a risk factor for heart failure, I think we have pretty solid data now that SGLT2 inhibitors will likely be effective in preventing heart failure in that kind of patient population. But I don’t think we have definitive data at this point to say that the drugs are effective in treating heart failure in people who already have a manifest clinical syndrome of heart failure, which is why we’re doing all these clinical trials now,” he continued.
Dr. Deedwania urged audience members to make the effort to become comfortable in prescribing SGLT2 inhibitors for their patients with type 2 diabetes.
“Many different surveys show that these drugs are not being utilized effectively by cardiologists,” noted Dr. Deedwania, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the university’s Fresno campus.
“As cardiologists, we may not want to own diabetes, but we at least have to feel that we have the ownership of treating the diabetic patient with cardiovascular disease with appropriate drugs. We don’t need to depend on endocrinologists because if we do these patients may become lost,” he said.
Dr. Kosiborod concurred, citing evidence that diabetic patients with cardiovascular disease are much more likely to see a cardiologist than an endocrinologist in the course of usual care.
“There’s definitely a golden opportunity here to intervene to reduce risk,” he said.
Dr. Kosiborod reported serving as a consultant to roughly a dozen pharmaceutical companies.
SOURCE: Kosiborod M. ACC 19, Abstract #1024-07.
NEW ORLEANS – based on data from a large real-world patient registry.
“The observed beneficial effects of SGLT2 inhibitors on heart failure may extend across the range of baseline ejection fractions,” Mikhail Kosiborod, MD, observed at the annual meeting of the American College of Cardiology.
This is an important new insight. The major randomized cardiovascular outcome trials that showed lower risks of heart failure hospitalization and all-cause mortality in type 2 diabetic patients on an SGLT2 inhibitor, such as EMPA-REG OUTCOME for empagliflozin (Jardiance) and CANVAS for canagliflozin (Invokana), didn’t include information on baseline LVEF. So until now it has been unclear whether the beneficial effects of the SGLT2 inhibitors preventing heart failure hospitalization vary depending upon LVEF, explained Dr. Kosiborod, a cardiologist at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
He presented an analysis drawn from the patient database kept by Maccabi Healthcare Services in Israel. The study included 5,307 patients with type 2 diabetes and an LVEF measurement recorded in their chart at the time they started on either empagliflozin or dapagliflozin (Farxiga) and an equal number of propensity-matched type 2 diabetic controls who started on other glucose-lowering drugs, most commonly an oral dipeptidyl peptidase-4 inhibitor.
During roughly 16,000 person-years of follow-up, 239 deaths occurred. Compared with patients on another glucose-lowering drug, the risk of death from all causes was reduced by 47% among patients who were on an SGLT2 inhibitor and had a baseline LVEF of 50% or greater and by 62% among the 9% of subjects who had a baseline LVEF less than 50%.
Similarly, the risk of heart failure hospitalization was reduced by 29% in SGLT2 inhibitor users with a preserved LVEF and by 27% if they had a reduced LVEF.
For the composite endpoint of heart failure hospitalization or all-cause mortality, the risk reductions associated with SGLT2 inhibitor therapy were 45% with preserved and 39% with reduced LVEF.
Session comoderator Prakash C. Deedwania, MD, noted that there are ongoing major randomized trials of various SGLT2 inhibitors in patients with known heart failure, with cardiovascular death and heart failure hospitalization as primary endpoints. He asked Dr. Kosiborod whether, given that the results of these studies aren’t in yet, he thinks clinicians should be prescribing SGLT2 inhibitors to diabetic or prediabetic patients who don’t have clinical symptoms of heart failure but may have a marker of increased risk, such as an elevated B-type natriuretic peptide.
“At least in my mind, we have more than enough evidence at this point to say that SGLT2 inhibitors are effective in preventing heart failure,” Dr. Kosiborod replied.
“Obviously, if your risk for developing a condition is higher at baseline, then the absolute benefit that you’re going to get from using an agent that’s effective in preventing that event is going to be higher and the number needed to treat is going to be lower. So if you have a patient at high risk for heart failure by whatever risk predictor you’re using and the patient doesn’t yet have heart failure but does have diabetes, which is already a risk factor for heart failure, I think we have pretty solid data now that SGLT2 inhibitors will likely be effective in preventing heart failure in that kind of patient population. But I don’t think we have definitive data at this point to say that the drugs are effective in treating heart failure in people who already have a manifest clinical syndrome of heart failure, which is why we’re doing all these clinical trials now,” he continued.
Dr. Deedwania urged audience members to make the effort to become comfortable in prescribing SGLT2 inhibitors for their patients with type 2 diabetes.
“Many different surveys show that these drugs are not being utilized effectively by cardiologists,” noted Dr. Deedwania, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the university’s Fresno campus.
“As cardiologists, we may not want to own diabetes, but we at least have to feel that we have the ownership of treating the diabetic patient with cardiovascular disease with appropriate drugs. We don’t need to depend on endocrinologists because if we do these patients may become lost,” he said.
Dr. Kosiborod concurred, citing evidence that diabetic patients with cardiovascular disease are much more likely to see a cardiologist than an endocrinologist in the course of usual care.
“There’s definitely a golden opportunity here to intervene to reduce risk,” he said.
Dr. Kosiborod reported serving as a consultant to roughly a dozen pharmaceutical companies.
SOURCE: Kosiborod M. ACC 19, Abstract #1024-07.
NEW ORLEANS – based on data from a large real-world patient registry.
“The observed beneficial effects of SGLT2 inhibitors on heart failure may extend across the range of baseline ejection fractions,” Mikhail Kosiborod, MD, observed at the annual meeting of the American College of Cardiology.
This is an important new insight. The major randomized cardiovascular outcome trials that showed lower risks of heart failure hospitalization and all-cause mortality in type 2 diabetic patients on an SGLT2 inhibitor, such as EMPA-REG OUTCOME for empagliflozin (Jardiance) and CANVAS for canagliflozin (Invokana), didn’t include information on baseline LVEF. So until now it has been unclear whether the beneficial effects of the SGLT2 inhibitors preventing heart failure hospitalization vary depending upon LVEF, explained Dr. Kosiborod, a cardiologist at Saint Luke’s Mid America Heart Institute in Kansas City, Mo.
He presented an analysis drawn from the patient database kept by Maccabi Healthcare Services in Israel. The study included 5,307 patients with type 2 diabetes and an LVEF measurement recorded in their chart at the time they started on either empagliflozin or dapagliflozin (Farxiga) and an equal number of propensity-matched type 2 diabetic controls who started on other glucose-lowering drugs, most commonly an oral dipeptidyl peptidase-4 inhibitor.
During roughly 16,000 person-years of follow-up, 239 deaths occurred. Compared with patients on another glucose-lowering drug, the risk of death from all causes was reduced by 47% among patients who were on an SGLT2 inhibitor and had a baseline LVEF of 50% or greater and by 62% among the 9% of subjects who had a baseline LVEF less than 50%.
Similarly, the risk of heart failure hospitalization was reduced by 29% in SGLT2 inhibitor users with a preserved LVEF and by 27% if they had a reduced LVEF.
For the composite endpoint of heart failure hospitalization or all-cause mortality, the risk reductions associated with SGLT2 inhibitor therapy were 45% with preserved and 39% with reduced LVEF.
Session comoderator Prakash C. Deedwania, MD, noted that there are ongoing major randomized trials of various SGLT2 inhibitors in patients with known heart failure, with cardiovascular death and heart failure hospitalization as primary endpoints. He asked Dr. Kosiborod whether, given that the results of these studies aren’t in yet, he thinks clinicians should be prescribing SGLT2 inhibitors to diabetic or prediabetic patients who don’t have clinical symptoms of heart failure but may have a marker of increased risk, such as an elevated B-type natriuretic peptide.
“At least in my mind, we have more than enough evidence at this point to say that SGLT2 inhibitors are effective in preventing heart failure,” Dr. Kosiborod replied.
“Obviously, if your risk for developing a condition is higher at baseline, then the absolute benefit that you’re going to get from using an agent that’s effective in preventing that event is going to be higher and the number needed to treat is going to be lower. So if you have a patient at high risk for heart failure by whatever risk predictor you’re using and the patient doesn’t yet have heart failure but does have diabetes, which is already a risk factor for heart failure, I think we have pretty solid data now that SGLT2 inhibitors will likely be effective in preventing heart failure in that kind of patient population. But I don’t think we have definitive data at this point to say that the drugs are effective in treating heart failure in people who already have a manifest clinical syndrome of heart failure, which is why we’re doing all these clinical trials now,” he continued.
Dr. Deedwania urged audience members to make the effort to become comfortable in prescribing SGLT2 inhibitors for their patients with type 2 diabetes.
“Many different surveys show that these drugs are not being utilized effectively by cardiologists,” noted Dr. Deedwania, professor of medicine at the University of California, San Francisco, and director of the heart failure program at the university’s Fresno campus.
“As cardiologists, we may not want to own diabetes, but we at least have to feel that we have the ownership of treating the diabetic patient with cardiovascular disease with appropriate drugs. We don’t need to depend on endocrinologists because if we do these patients may become lost,” he said.
Dr. Kosiborod concurred, citing evidence that diabetic patients with cardiovascular disease are much more likely to see a cardiologist than an endocrinologist in the course of usual care.
“There’s definitely a golden opportunity here to intervene to reduce risk,” he said.
Dr. Kosiborod reported serving as a consultant to roughly a dozen pharmaceutical companies.
SOURCE: Kosiborod M. ACC 19, Abstract #1024-07.
REPORTING FROM ACC 19
FDA approves Vyndaqel, Vyndamax for amyloidosis-based heart disease
The disease is caused by the buildup of abnormal deposits of amyloid in the body’s organs and tissues, interfering with normal function, and most often occurs in the heart and nervous system. Symptoms associated with amyloid buildup in the heart include shortness of breath, fatigue, heart failure, loss of consciousness, abnormal heart rhythms, and death.
FDA approval of both drugs was based on results of a clinical trial in which 441 patients with transthyretin-mediated amyloidosis received either tafamidis meglumine or placebo. After a mean of 30 months, patients who received tafamidis meglumine had a higher survival rate and a lower number of cardiovascular-related hospitalizations than did patients in the placebo group.
No drug-associated side effects have yet been identified; however, tafamidis can cause fetal harm when administered to a pregnant woman.
“Transthyretin-mediated amyloidosis is a rare, debilitating, and often fatal disease. The treatments we’re approving today are an important advancement in the treatment of the cardiomyopathy caused by transthyretin-mediated amyloidosis,” said Norman Stockbridge, MD, PhD, director of the Division of Cardiovascular and Renal Drugs in the FDA’s Center for Drug Evaluation and Research.
Find the full press release on the FDA website.
The disease is caused by the buildup of abnormal deposits of amyloid in the body’s organs and tissues, interfering with normal function, and most often occurs in the heart and nervous system. Symptoms associated with amyloid buildup in the heart include shortness of breath, fatigue, heart failure, loss of consciousness, abnormal heart rhythms, and death.
FDA approval of both drugs was based on results of a clinical trial in which 441 patients with transthyretin-mediated amyloidosis received either tafamidis meglumine or placebo. After a mean of 30 months, patients who received tafamidis meglumine had a higher survival rate and a lower number of cardiovascular-related hospitalizations than did patients in the placebo group.
No drug-associated side effects have yet been identified; however, tafamidis can cause fetal harm when administered to a pregnant woman.
“Transthyretin-mediated amyloidosis is a rare, debilitating, and often fatal disease. The treatments we’re approving today are an important advancement in the treatment of the cardiomyopathy caused by transthyretin-mediated amyloidosis,” said Norman Stockbridge, MD, PhD, director of the Division of Cardiovascular and Renal Drugs in the FDA’s Center for Drug Evaluation and Research.
Find the full press release on the FDA website.
The disease is caused by the buildup of abnormal deposits of amyloid in the body’s organs and tissues, interfering with normal function, and most often occurs in the heart and nervous system. Symptoms associated with amyloid buildup in the heart include shortness of breath, fatigue, heart failure, loss of consciousness, abnormal heart rhythms, and death.
FDA approval of both drugs was based on results of a clinical trial in which 441 patients with transthyretin-mediated amyloidosis received either tafamidis meglumine or placebo. After a mean of 30 months, patients who received tafamidis meglumine had a higher survival rate and a lower number of cardiovascular-related hospitalizations than did patients in the placebo group.
No drug-associated side effects have yet been identified; however, tafamidis can cause fetal harm when administered to a pregnant woman.
“Transthyretin-mediated amyloidosis is a rare, debilitating, and often fatal disease. The treatments we’re approving today are an important advancement in the treatment of the cardiomyopathy caused by transthyretin-mediated amyloidosis,” said Norman Stockbridge, MD, PhD, director of the Division of Cardiovascular and Renal Drugs in the FDA’s Center for Drug Evaluation and Research.
Find the full press release on the FDA website.
When to transition heart failure patient to alternative loop diuretic
PHILADELPHIA – While many internists might think a switch to spironolactone would be warranted for a heart failure patient with inadequate response to oral furosemide (Lasix), transitioning to an alternative loop diuretic may be the preferable approach, a cardiologist said at the annual meeting of the American College of Physicians.
“Lasix is associated with very high variability in terms of absorption, so torsemide and bumetanide should be considered in patients who have a poor response,” said Paul McKie, MD, MPH, a cardiologist and internist with Mayo Clinic, Rochester, Minn., in a session at the meeting.
When polled, only 22% of attendees at the session picked “transition to torsemide” as the best approach for restoring fluid balance with the lowest adverse potential in a 74-year-old woman with nonischemic cardiomyopathy on furosemide 80 mg twice daily who has been hospitalized for fluid overload three times in the year.
The majority of attendees (41%) said they would have added spironolactone. Dr. McKie disagreed with this approach. Instead, Dr. McKie said he would have transitioned this person to an alternative loop diuretic.
he added.
The rationale for considering an alternative loop diuretic in this patient hinges on bioavailability, which is “highly variable” for oral furosemide, at 10%-100%, while by contrast, torsemide and bumetanide have a very consistent bioavailability of 80%-100%, according to Dr. McKie.
“For this reason, I think about using torsemide or bumetanide in patients who are not responding to oral Lasix,” he said.
Dr. McKie described an algorithm that he and his colleagues use in clinic to intensify outpatient therapy for patients not achieving diuresis.
The first step is to ensure adherence and ask patients whether they are following sodium and fluid restriction: “I always ask about that first,” he said. “I tell patients, ‘You can out-eat and out-drink any diuretic regimen.’ ”
The next step is to double the dose of the loop diuretic and, sometimes, triple the dose if the double dose is not effective.
“If they’re diuresing but it’s just not adequate, then I’ll move to twice-daily dosing,” he said. “A practical tip is I tell patients to take their first dose as soon as they wake up and the second dose around 1:00 PM so that they’re not urinating all night.”
If twice-daily dosing doesn’t help, then that’s the point where an alternative loop diuretic would be warranted, according to Dr. McKie’s algorithm.
“Then I add a thiazide like metolazone, but I only do that after I’ve increased the dose of the loop diuretic,” he added.
If all else fails, then outpatient IV diuretics can be considered, according to the algorithmic approach.
Dr. McKie reported no relevant disclosures.
PHILADELPHIA – While many internists might think a switch to spironolactone would be warranted for a heart failure patient with inadequate response to oral furosemide (Lasix), transitioning to an alternative loop diuretic may be the preferable approach, a cardiologist said at the annual meeting of the American College of Physicians.
“Lasix is associated with very high variability in terms of absorption, so torsemide and bumetanide should be considered in patients who have a poor response,” said Paul McKie, MD, MPH, a cardiologist and internist with Mayo Clinic, Rochester, Minn., in a session at the meeting.
When polled, only 22% of attendees at the session picked “transition to torsemide” as the best approach for restoring fluid balance with the lowest adverse potential in a 74-year-old woman with nonischemic cardiomyopathy on furosemide 80 mg twice daily who has been hospitalized for fluid overload three times in the year.
The majority of attendees (41%) said they would have added spironolactone. Dr. McKie disagreed with this approach. Instead, Dr. McKie said he would have transitioned this person to an alternative loop diuretic.
he added.
The rationale for considering an alternative loop diuretic in this patient hinges on bioavailability, which is “highly variable” for oral furosemide, at 10%-100%, while by contrast, torsemide and bumetanide have a very consistent bioavailability of 80%-100%, according to Dr. McKie.
“For this reason, I think about using torsemide or bumetanide in patients who are not responding to oral Lasix,” he said.
Dr. McKie described an algorithm that he and his colleagues use in clinic to intensify outpatient therapy for patients not achieving diuresis.
The first step is to ensure adherence and ask patients whether they are following sodium and fluid restriction: “I always ask about that first,” he said. “I tell patients, ‘You can out-eat and out-drink any diuretic regimen.’ ”
The next step is to double the dose of the loop diuretic and, sometimes, triple the dose if the double dose is not effective.
“If they’re diuresing but it’s just not adequate, then I’ll move to twice-daily dosing,” he said. “A practical tip is I tell patients to take their first dose as soon as they wake up and the second dose around 1:00 PM so that they’re not urinating all night.”
If twice-daily dosing doesn’t help, then that’s the point where an alternative loop diuretic would be warranted, according to Dr. McKie’s algorithm.
“Then I add a thiazide like metolazone, but I only do that after I’ve increased the dose of the loop diuretic,” he added.
If all else fails, then outpatient IV diuretics can be considered, according to the algorithmic approach.
Dr. McKie reported no relevant disclosures.
PHILADELPHIA – While many internists might think a switch to spironolactone would be warranted for a heart failure patient with inadequate response to oral furosemide (Lasix), transitioning to an alternative loop diuretic may be the preferable approach, a cardiologist said at the annual meeting of the American College of Physicians.
“Lasix is associated with very high variability in terms of absorption, so torsemide and bumetanide should be considered in patients who have a poor response,” said Paul McKie, MD, MPH, a cardiologist and internist with Mayo Clinic, Rochester, Minn., in a session at the meeting.
When polled, only 22% of attendees at the session picked “transition to torsemide” as the best approach for restoring fluid balance with the lowest adverse potential in a 74-year-old woman with nonischemic cardiomyopathy on furosemide 80 mg twice daily who has been hospitalized for fluid overload three times in the year.
The majority of attendees (41%) said they would have added spironolactone. Dr. McKie disagreed with this approach. Instead, Dr. McKie said he would have transitioned this person to an alternative loop diuretic.
he added.
The rationale for considering an alternative loop diuretic in this patient hinges on bioavailability, which is “highly variable” for oral furosemide, at 10%-100%, while by contrast, torsemide and bumetanide have a very consistent bioavailability of 80%-100%, according to Dr. McKie.
“For this reason, I think about using torsemide or bumetanide in patients who are not responding to oral Lasix,” he said.
Dr. McKie described an algorithm that he and his colleagues use in clinic to intensify outpatient therapy for patients not achieving diuresis.
The first step is to ensure adherence and ask patients whether they are following sodium and fluid restriction: “I always ask about that first,” he said. “I tell patients, ‘You can out-eat and out-drink any diuretic regimen.’ ”
The next step is to double the dose of the loop diuretic and, sometimes, triple the dose if the double dose is not effective.
“If they’re diuresing but it’s just not adequate, then I’ll move to twice-daily dosing,” he said. “A practical tip is I tell patients to take their first dose as soon as they wake up and the second dose around 1:00 PM so that they’re not urinating all night.”
If twice-daily dosing doesn’t help, then that’s the point where an alternative loop diuretic would be warranted, according to Dr. McKie’s algorithm.
“Then I add a thiazide like metolazone, but I only do that after I’ve increased the dose of the loop diuretic,” he added.
If all else fails, then outpatient IV diuretics can be considered, according to the algorithmic approach.
Dr. McKie reported no relevant disclosures.
REPORTING FROM INTERNAL MEDICINE 2019
Plant-based diet lowers risk of heart failure
according to an analysis published online in the Journal of the American College of Cardiology.
Conversely, a Southern diet, defined as favoring fried and processed foods, is associated with an increased risk of heart failure. The results support a population-based dietary strategy for decreasing the risk of incident heart failure, according to the investigators.
Campaigns to prevent heart failure often emphasize the maintenance of a healthy diet and weight; however, little research has examined the relationship between dietary patterns and incident heart failure in patients without coronary heart disease.
Kyla M. Lara, MD, postgraduate fellow of cardiology and general internal medicine at the Icahn School of Medicine at Mount Sinai, New York, and colleagues sought to analyze the associations between five dietary patterns and incident hospitalizations for heart failure among adults in the United States. They examined data from the REGARDS (Reasons for Geographic and Racial Differences in Stroke) trial, a prospective study of black and white adults who were followed from 2003-2007 to 2014. Eligible participants completed a food frequency questionnaire and had no coronary heart disease or heart failure at baseline.
The REGARDS researchers’ principal component analysis identified the following five dietary patterns: convenience (for example, Mexican and Chinese dishes and fast food), plant based (for example, vegetables, fruit, and fish), sweets (for example, desserts, breads, and candy), Southern (for example, fried food, processed meats, and sugary beverages), and alcohol/salads. Dr. Lara and colleagues chose incident heart failure hospitalization as their primary endpoint.
The investigators included 16,068 participants in their analysis. Mean age was 64 years, roughly 59% of the sample were women, and 34% were black.
After a median 8.7 years of follow-up, 363 participants had incident heart failure hospitalizations. The highest quartile of adherence to the plant-based dietary pattern was associated with a 41% lower risk of heart failure in multivariate models, compared with the lowest quartile. The highest adherence to the Southern dietary pattern was linked with a 72% higher risk of heart failure after adjustments for age, sex, race, and other potential confounders such as education, income, smoking, and physical activity.
After further adjustments for body mass index, waist circumference, hypertension, dyslipidemia, diabetes mellitus, atrial fibrillation, and chronic kidney disease, the association was attenuated and no longer statistically significant. Dr. Lara and colleagues found no statistically significant associations between incident heart failure with reduced or preserved ejection fraction hospitalizations and the dietary patterns. They also found no associations with the other three dietary patterns.
One researcher reported receiving research funding from Amgen and has consulted for Novartis. The other researchers reported no relevant conflicts.
SOURCE: Lara KM et al. J Am Coll Cardiol. 2019 Apr 30;73(16):2036-45.
This analysis of the REGARDS study contributes toward creating a strong evidence base for the prevention of heart failure through dietary measures, wrote Dong D. Wang, SCD, MD, a visiting scientist at Harvard School of Public Health, Boston, in an accompanying editorial. Empirically derived dietary patterns, such as those described in this study, can form the basis for recommendations easily, he added. “We usually have greater confidence when interpreting the associations with dietary patterns as causal than we have for the associations with specific nutrients or foods. Furthermore, the findings are particularly useful for making recommendations to a general population because of their use of a baseline coronary heart disease–free study population and the inclusion of black participants with greater susceptibility to heart failure. Thus, this study possesses a great potential of informing the population-level strategies for the prevention of heart failure.”
Nutritional epidemiologic studies examining subtypes of heart failure are valuable in light of the disease’s phenotypic and pathophysiological heterogeneity, Dr. Wang wrote. “These findings, if confirmed in future studies, will not only contribute to in-depth biological understanding and phenotypic refinement of heart failure, but also inform dietary prevention approaches customized for specific heart failure phenotypes. In addition, they perfectly fit into key missions of precision medicine [i.e., understanding large variability between individuals in both the development and the clinical manifestations of the specific disease, as well as variability in individual’s response to dietary, lifestyle, and pharmacological interventions].”
Dr. Wang reported no relationships relevant to the contents of this paper.
This analysis of the REGARDS study contributes toward creating a strong evidence base for the prevention of heart failure through dietary measures, wrote Dong D. Wang, SCD, MD, a visiting scientist at Harvard School of Public Health, Boston, in an accompanying editorial. Empirically derived dietary patterns, such as those described in this study, can form the basis for recommendations easily, he added. “We usually have greater confidence when interpreting the associations with dietary patterns as causal than we have for the associations with specific nutrients or foods. Furthermore, the findings are particularly useful for making recommendations to a general population because of their use of a baseline coronary heart disease–free study population and the inclusion of black participants with greater susceptibility to heart failure. Thus, this study possesses a great potential of informing the population-level strategies for the prevention of heart failure.”
Nutritional epidemiologic studies examining subtypes of heart failure are valuable in light of the disease’s phenotypic and pathophysiological heterogeneity, Dr. Wang wrote. “These findings, if confirmed in future studies, will not only contribute to in-depth biological understanding and phenotypic refinement of heart failure, but also inform dietary prevention approaches customized for specific heart failure phenotypes. In addition, they perfectly fit into key missions of precision medicine [i.e., understanding large variability between individuals in both the development and the clinical manifestations of the specific disease, as well as variability in individual’s response to dietary, lifestyle, and pharmacological interventions].”
Dr. Wang reported no relationships relevant to the contents of this paper.
This analysis of the REGARDS study contributes toward creating a strong evidence base for the prevention of heart failure through dietary measures, wrote Dong D. Wang, SCD, MD, a visiting scientist at Harvard School of Public Health, Boston, in an accompanying editorial. Empirically derived dietary patterns, such as those described in this study, can form the basis for recommendations easily, he added. “We usually have greater confidence when interpreting the associations with dietary patterns as causal than we have for the associations with specific nutrients or foods. Furthermore, the findings are particularly useful for making recommendations to a general population because of their use of a baseline coronary heart disease–free study population and the inclusion of black participants with greater susceptibility to heart failure. Thus, this study possesses a great potential of informing the population-level strategies for the prevention of heart failure.”
Nutritional epidemiologic studies examining subtypes of heart failure are valuable in light of the disease’s phenotypic and pathophysiological heterogeneity, Dr. Wang wrote. “These findings, if confirmed in future studies, will not only contribute to in-depth biological understanding and phenotypic refinement of heart failure, but also inform dietary prevention approaches customized for specific heart failure phenotypes. In addition, they perfectly fit into key missions of precision medicine [i.e., understanding large variability between individuals in both the development and the clinical manifestations of the specific disease, as well as variability in individual’s response to dietary, lifestyle, and pharmacological interventions].”
Dr. Wang reported no relationships relevant to the contents of this paper.
according to an analysis published online in the Journal of the American College of Cardiology.
Conversely, a Southern diet, defined as favoring fried and processed foods, is associated with an increased risk of heart failure. The results support a population-based dietary strategy for decreasing the risk of incident heart failure, according to the investigators.
Campaigns to prevent heart failure often emphasize the maintenance of a healthy diet and weight; however, little research has examined the relationship between dietary patterns and incident heart failure in patients without coronary heart disease.
Kyla M. Lara, MD, postgraduate fellow of cardiology and general internal medicine at the Icahn School of Medicine at Mount Sinai, New York, and colleagues sought to analyze the associations between five dietary patterns and incident hospitalizations for heart failure among adults in the United States. They examined data from the REGARDS (Reasons for Geographic and Racial Differences in Stroke) trial, a prospective study of black and white adults who were followed from 2003-2007 to 2014. Eligible participants completed a food frequency questionnaire and had no coronary heart disease or heart failure at baseline.
The REGARDS researchers’ principal component analysis identified the following five dietary patterns: convenience (for example, Mexican and Chinese dishes and fast food), plant based (for example, vegetables, fruit, and fish), sweets (for example, desserts, breads, and candy), Southern (for example, fried food, processed meats, and sugary beverages), and alcohol/salads. Dr. Lara and colleagues chose incident heart failure hospitalization as their primary endpoint.
The investigators included 16,068 participants in their analysis. Mean age was 64 years, roughly 59% of the sample were women, and 34% were black.
After a median 8.7 years of follow-up, 363 participants had incident heart failure hospitalizations. The highest quartile of adherence to the plant-based dietary pattern was associated with a 41% lower risk of heart failure in multivariate models, compared with the lowest quartile. The highest adherence to the Southern dietary pattern was linked with a 72% higher risk of heart failure after adjustments for age, sex, race, and other potential confounders such as education, income, smoking, and physical activity.
After further adjustments for body mass index, waist circumference, hypertension, dyslipidemia, diabetes mellitus, atrial fibrillation, and chronic kidney disease, the association was attenuated and no longer statistically significant. Dr. Lara and colleagues found no statistically significant associations between incident heart failure with reduced or preserved ejection fraction hospitalizations and the dietary patterns. They also found no associations with the other three dietary patterns.
One researcher reported receiving research funding from Amgen and has consulted for Novartis. The other researchers reported no relevant conflicts.
SOURCE: Lara KM et al. J Am Coll Cardiol. 2019 Apr 30;73(16):2036-45.
according to an analysis published online in the Journal of the American College of Cardiology.
Conversely, a Southern diet, defined as favoring fried and processed foods, is associated with an increased risk of heart failure. The results support a population-based dietary strategy for decreasing the risk of incident heart failure, according to the investigators.
Campaigns to prevent heart failure often emphasize the maintenance of a healthy diet and weight; however, little research has examined the relationship between dietary patterns and incident heart failure in patients without coronary heart disease.
Kyla M. Lara, MD, postgraduate fellow of cardiology and general internal medicine at the Icahn School of Medicine at Mount Sinai, New York, and colleagues sought to analyze the associations between five dietary patterns and incident hospitalizations for heart failure among adults in the United States. They examined data from the REGARDS (Reasons for Geographic and Racial Differences in Stroke) trial, a prospective study of black and white adults who were followed from 2003-2007 to 2014. Eligible participants completed a food frequency questionnaire and had no coronary heart disease or heart failure at baseline.
The REGARDS researchers’ principal component analysis identified the following five dietary patterns: convenience (for example, Mexican and Chinese dishes and fast food), plant based (for example, vegetables, fruit, and fish), sweets (for example, desserts, breads, and candy), Southern (for example, fried food, processed meats, and sugary beverages), and alcohol/salads. Dr. Lara and colleagues chose incident heart failure hospitalization as their primary endpoint.
The investigators included 16,068 participants in their analysis. Mean age was 64 years, roughly 59% of the sample were women, and 34% were black.
After a median 8.7 years of follow-up, 363 participants had incident heart failure hospitalizations. The highest quartile of adherence to the plant-based dietary pattern was associated with a 41% lower risk of heart failure in multivariate models, compared with the lowest quartile. The highest adherence to the Southern dietary pattern was linked with a 72% higher risk of heart failure after adjustments for age, sex, race, and other potential confounders such as education, income, smoking, and physical activity.
After further adjustments for body mass index, waist circumference, hypertension, dyslipidemia, diabetes mellitus, atrial fibrillation, and chronic kidney disease, the association was attenuated and no longer statistically significant. Dr. Lara and colleagues found no statistically significant associations between incident heart failure with reduced or preserved ejection fraction hospitalizations and the dietary patterns. They also found no associations with the other three dietary patterns.
One researcher reported receiving research funding from Amgen and has consulted for Novartis. The other researchers reported no relevant conflicts.
SOURCE: Lara KM et al. J Am Coll Cardiol. 2019 Apr 30;73(16):2036-45.
FROM THE JOURNAL OF THE AMERICAN COLLEGE OF CARDIOLOGY
Key clinical point: Plant-based and Southern diets influence the risk of heart failure.
Major finding: Adherence to a plant-based diet reduces the risk of heart failure by 41%.
Study details: An analysis of data for 16,068 participants in the REGARDS study.
Disclosures: One coauthor reported receiving research funding from Amgen and has consulted for Novartis.
Source: Lara KM et al. J Am Coll Cardiol. 2019 Apr 30;73(16):2036-45.
PIONEER-HF Extension: Don’t stall starting sacubitril/valsartan
NEW ORLEANS – Waiting a few months after a patient has been hospitalized for acute decompensated heart failure before launching a switch from enalapril to sacubitril/valsartan imposes a steep price in terms of extra major cardiovascular events, compared with starting the angiotensin-neprilysin inhibitor during the initial hospitalization, according to the open-label extension of the PIONEER-HF trial.
“We think these data have important clinical implications: While sacubitril/valsartan decreases NT-proBNP compared with enalapril regardless of when it is initiated, the early improvement in postdischarge outcomes supports the in-hospital initiation of sacubitril/valsartan in stabilized patients with acute decompensated heart failure,” Adam D. DeVore, MD, declared in presenting the PIONEER-HF Extension results at the annual meeting of the American College of Cardiology.
PIONEER-HF was a landmark, practice-changing, double-blind clinical trial in which 881 patients were randomized to initiation of sacubitril/valsartan (Entresto) or enalapril during hospitalization for acute decompensated heart failure. In the previously reported main outcome, 8 weeks after discharge the sacubitril/valsartan group had a 29% greater reduction in NT-proBNP (the N-terminal prohormone of brain natriuretic peptide) and a 42% lower rate of the composite clinical endpoint of cardiovascular death or heart failure rehospitalization than the enalapril group (N Engl J Med. 2019 Feb 7;380[6]:539-48).
The 4-week open-label extension of PIONEER-HF began at week 8, when participants initially randomized to enalapril during the double-blind phase were switched to sacubitril/valsartan, while those assigned to in-hospital initiation of the angiotensin-neprilysin inhibitor (ARNI) stayed the course.
At week 12, after 4 weeks of open-label treatment, patients on sacubitril/valsartan from the start experienced an additional 18.5% drop in NT-proBNP from their week-8 baseline of 1,218 pg/mL. Meanwhile, the NT-proBNP level in the switch group plunged by 35.8% from a week-8 baseline of 1,630 pg/mL. As a result, both groups ended up at the same much-improved biomarker level at week 12, observed Dr. DeVore, a cardiologist at Duke University in Durham, N.C.
Clinical event rates, however, were another story altogether. The clinical event gap between the two study arms documented at week 8 in the double-blind phase of the trial didn’t close significantly in the 4 weeks after the enalapril group crossed over to open-label sacubitril/valsartan. Indeed, the relative risk of the composite endpoint of cardiovascular death, heart failure rehospitalization, or left ventricular assist device implantation during the 4-week extension phase was 33% lower in the continuous sacubitril/valsartan group than in the switchers. The absolute risk reduction was 5.6%, with a favorable number needed to treat of 18.
This difference was driven mainly by less rehospitalization for heart failure. Few cardiovascular deaths or LVAD implantations occurred during the relatively brief 4-week extension phase of the trial.
“But this is an important thing as we think about what we’re trying to accomplish in heart failure: trying to find tools that improve rehospitalization rates after people leave the hospital is extremely important,” Dr. DeVore said. “We do know that the really vulnerable period for rehospitalization is early on, so my sus```picion – though I can’t prove it – is that’s the important part. That’s when we need to have patients on the best therapy.”
He was asked how practical it is to initiate sacubitril/valsartan during hospitalization for acute decompensated heart failure in real-world clinical practice, given that it can be done only after patients achieve hemodynamic stability.
“I think the definition of hemodynamic stability we used in the trial was a fairly straightforward one, very clinical, and one we can translate to the bedside,” Dr. DeVore replied. “Patients had to have a systolic blood pressure of 100 mm Hg or greater for 6 hours, which is easily documented in the hospital, no changes in IV diuretics or IV vasodilators for 6 hours, and no IV inotropes for the last 24 hours. That’s how we defined hemodynamic stability. I think we should be able to find these patients.”
Average length of stay in the index hospitalization in PIONEER-HF was just over 5 days, but the study protocol actually resulted in longer-than-needed hospitalization because it required that patients had to receive three double-blind doses of their study medication before discharge. In routine practice, it’s unlikely that in-hospital initiation of sacubitril/valsartan will result in a length of stay greater than the national average of about 4.5 days, according the cardiologist.
Current ACC/American Heart Association/Heart Failure Society of American guidelines on management of heart failure include a Class Ia recommendation to switch patients from an ACE inhibitor or angiotensin inhibitor to sacubitril/valsartan (Circulation. 2017 Aug 8;136[6]:e137-61).
However, heart failure specialists are concerned by national data showing that sacubitril/valsartan remains widely underprescribed.
Dr. DeVore reported serving as a consultant to Novartis and receiving research grants from a half dozen pharmaceutical companies as well as the American Heart Association, National Heart, Lung, and Blood Institute, and the Patient-Centered Outcomes Research Institute .
NEW ORLEANS – Waiting a few months after a patient has been hospitalized for acute decompensated heart failure before launching a switch from enalapril to sacubitril/valsartan imposes a steep price in terms of extra major cardiovascular events, compared with starting the angiotensin-neprilysin inhibitor during the initial hospitalization, according to the open-label extension of the PIONEER-HF trial.
“We think these data have important clinical implications: While sacubitril/valsartan decreases NT-proBNP compared with enalapril regardless of when it is initiated, the early improvement in postdischarge outcomes supports the in-hospital initiation of sacubitril/valsartan in stabilized patients with acute decompensated heart failure,” Adam D. DeVore, MD, declared in presenting the PIONEER-HF Extension results at the annual meeting of the American College of Cardiology.
PIONEER-HF was a landmark, practice-changing, double-blind clinical trial in which 881 patients were randomized to initiation of sacubitril/valsartan (Entresto) or enalapril during hospitalization for acute decompensated heart failure. In the previously reported main outcome, 8 weeks after discharge the sacubitril/valsartan group had a 29% greater reduction in NT-proBNP (the N-terminal prohormone of brain natriuretic peptide) and a 42% lower rate of the composite clinical endpoint of cardiovascular death or heart failure rehospitalization than the enalapril group (N Engl J Med. 2019 Feb 7;380[6]:539-48).
The 4-week open-label extension of PIONEER-HF began at week 8, when participants initially randomized to enalapril during the double-blind phase were switched to sacubitril/valsartan, while those assigned to in-hospital initiation of the angiotensin-neprilysin inhibitor (ARNI) stayed the course.
At week 12, after 4 weeks of open-label treatment, patients on sacubitril/valsartan from the start experienced an additional 18.5% drop in NT-proBNP from their week-8 baseline of 1,218 pg/mL. Meanwhile, the NT-proBNP level in the switch group plunged by 35.8% from a week-8 baseline of 1,630 pg/mL. As a result, both groups ended up at the same much-improved biomarker level at week 12, observed Dr. DeVore, a cardiologist at Duke University in Durham, N.C.
Clinical event rates, however, were another story altogether. The clinical event gap between the two study arms documented at week 8 in the double-blind phase of the trial didn’t close significantly in the 4 weeks after the enalapril group crossed over to open-label sacubitril/valsartan. Indeed, the relative risk of the composite endpoint of cardiovascular death, heart failure rehospitalization, or left ventricular assist device implantation during the 4-week extension phase was 33% lower in the continuous sacubitril/valsartan group than in the switchers. The absolute risk reduction was 5.6%, with a favorable number needed to treat of 18.
This difference was driven mainly by less rehospitalization for heart failure. Few cardiovascular deaths or LVAD implantations occurred during the relatively brief 4-week extension phase of the trial.
“But this is an important thing as we think about what we’re trying to accomplish in heart failure: trying to find tools that improve rehospitalization rates after people leave the hospital is extremely important,” Dr. DeVore said. “We do know that the really vulnerable period for rehospitalization is early on, so my sus```picion – though I can’t prove it – is that’s the important part. That’s when we need to have patients on the best therapy.”
He was asked how practical it is to initiate sacubitril/valsartan during hospitalization for acute decompensated heart failure in real-world clinical practice, given that it can be done only after patients achieve hemodynamic stability.
“I think the definition of hemodynamic stability we used in the trial was a fairly straightforward one, very clinical, and one we can translate to the bedside,” Dr. DeVore replied. “Patients had to have a systolic blood pressure of 100 mm Hg or greater for 6 hours, which is easily documented in the hospital, no changes in IV diuretics or IV vasodilators for 6 hours, and no IV inotropes for the last 24 hours. That’s how we defined hemodynamic stability. I think we should be able to find these patients.”
Average length of stay in the index hospitalization in PIONEER-HF was just over 5 days, but the study protocol actually resulted in longer-than-needed hospitalization because it required that patients had to receive three double-blind doses of their study medication before discharge. In routine practice, it’s unlikely that in-hospital initiation of sacubitril/valsartan will result in a length of stay greater than the national average of about 4.5 days, according the cardiologist.
Current ACC/American Heart Association/Heart Failure Society of American guidelines on management of heart failure include a Class Ia recommendation to switch patients from an ACE inhibitor or angiotensin inhibitor to sacubitril/valsartan (Circulation. 2017 Aug 8;136[6]:e137-61).
However, heart failure specialists are concerned by national data showing that sacubitril/valsartan remains widely underprescribed.
Dr. DeVore reported serving as a consultant to Novartis and receiving research grants from a half dozen pharmaceutical companies as well as the American Heart Association, National Heart, Lung, and Blood Institute, and the Patient-Centered Outcomes Research Institute .
NEW ORLEANS – Waiting a few months after a patient has been hospitalized for acute decompensated heart failure before launching a switch from enalapril to sacubitril/valsartan imposes a steep price in terms of extra major cardiovascular events, compared with starting the angiotensin-neprilysin inhibitor during the initial hospitalization, according to the open-label extension of the PIONEER-HF trial.
“We think these data have important clinical implications: While sacubitril/valsartan decreases NT-proBNP compared with enalapril regardless of when it is initiated, the early improvement in postdischarge outcomes supports the in-hospital initiation of sacubitril/valsartan in stabilized patients with acute decompensated heart failure,” Adam D. DeVore, MD, declared in presenting the PIONEER-HF Extension results at the annual meeting of the American College of Cardiology.
PIONEER-HF was a landmark, practice-changing, double-blind clinical trial in which 881 patients were randomized to initiation of sacubitril/valsartan (Entresto) or enalapril during hospitalization for acute decompensated heart failure. In the previously reported main outcome, 8 weeks after discharge the sacubitril/valsartan group had a 29% greater reduction in NT-proBNP (the N-terminal prohormone of brain natriuretic peptide) and a 42% lower rate of the composite clinical endpoint of cardiovascular death or heart failure rehospitalization than the enalapril group (N Engl J Med. 2019 Feb 7;380[6]:539-48).
The 4-week open-label extension of PIONEER-HF began at week 8, when participants initially randomized to enalapril during the double-blind phase were switched to sacubitril/valsartan, while those assigned to in-hospital initiation of the angiotensin-neprilysin inhibitor (ARNI) stayed the course.
At week 12, after 4 weeks of open-label treatment, patients on sacubitril/valsartan from the start experienced an additional 18.5% drop in NT-proBNP from their week-8 baseline of 1,218 pg/mL. Meanwhile, the NT-proBNP level in the switch group plunged by 35.8% from a week-8 baseline of 1,630 pg/mL. As a result, both groups ended up at the same much-improved biomarker level at week 12, observed Dr. DeVore, a cardiologist at Duke University in Durham, N.C.
Clinical event rates, however, were another story altogether. The clinical event gap between the two study arms documented at week 8 in the double-blind phase of the trial didn’t close significantly in the 4 weeks after the enalapril group crossed over to open-label sacubitril/valsartan. Indeed, the relative risk of the composite endpoint of cardiovascular death, heart failure rehospitalization, or left ventricular assist device implantation during the 4-week extension phase was 33% lower in the continuous sacubitril/valsartan group than in the switchers. The absolute risk reduction was 5.6%, with a favorable number needed to treat of 18.
This difference was driven mainly by less rehospitalization for heart failure. Few cardiovascular deaths or LVAD implantations occurred during the relatively brief 4-week extension phase of the trial.
“But this is an important thing as we think about what we’re trying to accomplish in heart failure: trying to find tools that improve rehospitalization rates after people leave the hospital is extremely important,” Dr. DeVore said. “We do know that the really vulnerable period for rehospitalization is early on, so my sus```picion – though I can’t prove it – is that’s the important part. That’s when we need to have patients on the best therapy.”
He was asked how practical it is to initiate sacubitril/valsartan during hospitalization for acute decompensated heart failure in real-world clinical practice, given that it can be done only after patients achieve hemodynamic stability.
“I think the definition of hemodynamic stability we used in the trial was a fairly straightforward one, very clinical, and one we can translate to the bedside,” Dr. DeVore replied. “Patients had to have a systolic blood pressure of 100 mm Hg or greater for 6 hours, which is easily documented in the hospital, no changes in IV diuretics or IV vasodilators for 6 hours, and no IV inotropes for the last 24 hours. That’s how we defined hemodynamic stability. I think we should be able to find these patients.”
Average length of stay in the index hospitalization in PIONEER-HF was just over 5 days, but the study protocol actually resulted in longer-than-needed hospitalization because it required that patients had to receive three double-blind doses of their study medication before discharge. In routine practice, it’s unlikely that in-hospital initiation of sacubitril/valsartan will result in a length of stay greater than the national average of about 4.5 days, according the cardiologist.
Current ACC/American Heart Association/Heart Failure Society of American guidelines on management of heart failure include a Class Ia recommendation to switch patients from an ACE inhibitor or angiotensin inhibitor to sacubitril/valsartan (Circulation. 2017 Aug 8;136[6]:e137-61).
However, heart failure specialists are concerned by national data showing that sacubitril/valsartan remains widely underprescribed.
Dr. DeVore reported serving as a consultant to Novartis and receiving research grants from a half dozen pharmaceutical companies as well as the American Heart Association, National Heart, Lung, and Blood Institute, and the Patient-Centered Outcomes Research Institute .
REPORTING FROM ACC 19
Key clinical point: over the next 12 weeks, compared with initiation of enalapril followed by a delayed switch to sacubitril/valsartan at 8 weeks.
Major finding: The number needed to treat with in-hospital initiation of sacubitril/valsartan instead of enalapril to avoid one cardiovascular death, heart failure rehospitalization, or implantation of a left ventricular assist device was 18.
Study details: The PIONEER-HF Extension study included 881 heart failure patients, all on open-label sacubitril/valsartan during the 4-week extension phase.
Disclosures: The study was sponsored by AstraZeneca. The presenter reported receiving research grants from and serving as a consultant to the company.
Clinicians, CMS confer over heart failure–readmission penalty
NEW ORLEANS – Mounting evidence shows that heart failure patient mortality increased as an unintended consequence of a Medicare program that penalizes hospitals with too many 30-day readmissions of heart failure patients. This has prompted discussions among cardiologists, Medicare officials, and other stakeholders in an attempt to modify the penalty program so it no longer considers just readmissions but instead bases penalties on broader and more nuanced measures of patient outcomes.
Staffers at the Centers for Medicare & Medicaid Services, the federal agency that manages Medicare, “said that they take this seriously and will look into it, and they are interested in next-generation measures that are more patient centered” than simply the 30-day readmission rate, Gregg C. Fonarow, MD, said in an interview at the annual meeting of the American College of Cardiology. “This is a case where there is credible evidence of increased mortality that is consistent, reproducible, and strongly associated with the penalty and cannot be otherwise explained,” said Dr. Fonarow, professor of medicine and cochief of cardiology at the University of California, Los Angeles.
He is among the most active researchers to document that, while CMS’s Hospital Readmissions Reduction Program (HRRP) led to significantly reduced readmission rates in patients with heart failure, this came at a cost of a significant increase in mortality among the same patients. For example, an article he published in 2018 that analyzed more than 115,000 Medicare beneficiaries during 2006-2014 showed that during the penalty phase, which began in 2012, readmissions fell after adjustment by a relative 8%, but adjusted mortality rose by a relative 10%, compared with how patients had fared prior to launching the HRRP (JAMA Cardiol. 2018 Jan;3[1]:44-53). Recent reports from other research groups have had similar findings, such as a study of more than 3 million Medicare beneficiaries with heart failure during 2005-2015 that also showed significantly increased mortality after the penalty phase for readmissions began (JAMA. 2018 Dec 25;320[24]:2542-52). In a commentary that accompanied this report, Dr. Fonarow cited the multiple analyses that show consistent findings and the need for CMS to “initiate an expeditious reconsideration and revision” of their current approach to penalizing hospitals for heart failure readmissions (JAMA. 2018 Dec 25;320[24]:2539-41).
The groups recently in discussion with CMS about this issue include the American College of Cardiology, the American Heart Association, the Heart Failure Society of America, the American College of Physicians, the American Hospital Association, and several other medical professional groups, said Biykem Bozkurt, MD, who has worked with Dr. Fonarow and representatives from these organizations in talks with CMS.
“We are trying to find a harmonized approach with patient-centric outcomes that reflect true improvements in quality of care,” she said in an interview. One possibility up for consideration is a combined measure of heart failure readmissions, mortality, and a patient-reported outcome. The measure would go to CMS directly from each patient’s electronic medical record, making data collection less burdensome to clinicians, said Dr. Bozkurt, professor of medicine at Baylor College of Medicine and cardiology section chief at the VA Medical Center in Houston. She expressed hope that a change in the CMS metric might happen later this year.
“CMS can’t simply stop the HRRP, so the discussion is on how to get a meaningful change. I’m increasingly optimistic, because the findings of harm [from current policies] are impossible to ignore,” Dr. Fonarow said. “There will be increasing pressure on CMS to develop a pathway to make modifications. It’s egregious to continue a policy that’s been associated with harm” to heart failure patients.
Dr. Fonarow and Dr. Bozkurt had no relevant commercial disclosures.
NEW ORLEANS – Mounting evidence shows that heart failure patient mortality increased as an unintended consequence of a Medicare program that penalizes hospitals with too many 30-day readmissions of heart failure patients. This has prompted discussions among cardiologists, Medicare officials, and other stakeholders in an attempt to modify the penalty program so it no longer considers just readmissions but instead bases penalties on broader and more nuanced measures of patient outcomes.
Staffers at the Centers for Medicare & Medicaid Services, the federal agency that manages Medicare, “said that they take this seriously and will look into it, and they are interested in next-generation measures that are more patient centered” than simply the 30-day readmission rate, Gregg C. Fonarow, MD, said in an interview at the annual meeting of the American College of Cardiology. “This is a case where there is credible evidence of increased mortality that is consistent, reproducible, and strongly associated with the penalty and cannot be otherwise explained,” said Dr. Fonarow, professor of medicine and cochief of cardiology at the University of California, Los Angeles.
He is among the most active researchers to document that, while CMS’s Hospital Readmissions Reduction Program (HRRP) led to significantly reduced readmission rates in patients with heart failure, this came at a cost of a significant increase in mortality among the same patients. For example, an article he published in 2018 that analyzed more than 115,000 Medicare beneficiaries during 2006-2014 showed that during the penalty phase, which began in 2012, readmissions fell after adjustment by a relative 8%, but adjusted mortality rose by a relative 10%, compared with how patients had fared prior to launching the HRRP (JAMA Cardiol. 2018 Jan;3[1]:44-53). Recent reports from other research groups have had similar findings, such as a study of more than 3 million Medicare beneficiaries with heart failure during 2005-2015 that also showed significantly increased mortality after the penalty phase for readmissions began (JAMA. 2018 Dec 25;320[24]:2542-52). In a commentary that accompanied this report, Dr. Fonarow cited the multiple analyses that show consistent findings and the need for CMS to “initiate an expeditious reconsideration and revision” of their current approach to penalizing hospitals for heart failure readmissions (JAMA. 2018 Dec 25;320[24]:2539-41).
The groups recently in discussion with CMS about this issue include the American College of Cardiology, the American Heart Association, the Heart Failure Society of America, the American College of Physicians, the American Hospital Association, and several other medical professional groups, said Biykem Bozkurt, MD, who has worked with Dr. Fonarow and representatives from these organizations in talks with CMS.
“We are trying to find a harmonized approach with patient-centric outcomes that reflect true improvements in quality of care,” she said in an interview. One possibility up for consideration is a combined measure of heart failure readmissions, mortality, and a patient-reported outcome. The measure would go to CMS directly from each patient’s electronic medical record, making data collection less burdensome to clinicians, said Dr. Bozkurt, professor of medicine at Baylor College of Medicine and cardiology section chief at the VA Medical Center in Houston. She expressed hope that a change in the CMS metric might happen later this year.
“CMS can’t simply stop the HRRP, so the discussion is on how to get a meaningful change. I’m increasingly optimistic, because the findings of harm [from current policies] are impossible to ignore,” Dr. Fonarow said. “There will be increasing pressure on CMS to develop a pathway to make modifications. It’s egregious to continue a policy that’s been associated with harm” to heart failure patients.
Dr. Fonarow and Dr. Bozkurt had no relevant commercial disclosures.
NEW ORLEANS – Mounting evidence shows that heart failure patient mortality increased as an unintended consequence of a Medicare program that penalizes hospitals with too many 30-day readmissions of heart failure patients. This has prompted discussions among cardiologists, Medicare officials, and other stakeholders in an attempt to modify the penalty program so it no longer considers just readmissions but instead bases penalties on broader and more nuanced measures of patient outcomes.
Staffers at the Centers for Medicare & Medicaid Services, the federal agency that manages Medicare, “said that they take this seriously and will look into it, and they are interested in next-generation measures that are more patient centered” than simply the 30-day readmission rate, Gregg C. Fonarow, MD, said in an interview at the annual meeting of the American College of Cardiology. “This is a case where there is credible evidence of increased mortality that is consistent, reproducible, and strongly associated with the penalty and cannot be otherwise explained,” said Dr. Fonarow, professor of medicine and cochief of cardiology at the University of California, Los Angeles.
He is among the most active researchers to document that, while CMS’s Hospital Readmissions Reduction Program (HRRP) led to significantly reduced readmission rates in patients with heart failure, this came at a cost of a significant increase in mortality among the same patients. For example, an article he published in 2018 that analyzed more than 115,000 Medicare beneficiaries during 2006-2014 showed that during the penalty phase, which began in 2012, readmissions fell after adjustment by a relative 8%, but adjusted mortality rose by a relative 10%, compared with how patients had fared prior to launching the HRRP (JAMA Cardiol. 2018 Jan;3[1]:44-53). Recent reports from other research groups have had similar findings, such as a study of more than 3 million Medicare beneficiaries with heart failure during 2005-2015 that also showed significantly increased mortality after the penalty phase for readmissions began (JAMA. 2018 Dec 25;320[24]:2542-52). In a commentary that accompanied this report, Dr. Fonarow cited the multiple analyses that show consistent findings and the need for CMS to “initiate an expeditious reconsideration and revision” of their current approach to penalizing hospitals for heart failure readmissions (JAMA. 2018 Dec 25;320[24]:2539-41).
The groups recently in discussion with CMS about this issue include the American College of Cardiology, the American Heart Association, the Heart Failure Society of America, the American College of Physicians, the American Hospital Association, and several other medical professional groups, said Biykem Bozkurt, MD, who has worked with Dr. Fonarow and representatives from these organizations in talks with CMS.
“We are trying to find a harmonized approach with patient-centric outcomes that reflect true improvements in quality of care,” she said in an interview. One possibility up for consideration is a combined measure of heart failure readmissions, mortality, and a patient-reported outcome. The measure would go to CMS directly from each patient’s electronic medical record, making data collection less burdensome to clinicians, said Dr. Bozkurt, professor of medicine at Baylor College of Medicine and cardiology section chief at the VA Medical Center in Houston. She expressed hope that a change in the CMS metric might happen later this year.
“CMS can’t simply stop the HRRP, so the discussion is on how to get a meaningful change. I’m increasingly optimistic, because the findings of harm [from current policies] are impossible to ignore,” Dr. Fonarow said. “There will be increasing pressure on CMS to develop a pathway to make modifications. It’s egregious to continue a policy that’s been associated with harm” to heart failure patients.
Dr. Fonarow and Dr. Bozkurt had no relevant commercial disclosures.
REPORTING FROM ACC 19
Novel percutaneous therapy for HOCM in development
SNOWMASS, COLO. – Percutaneous mitral valve plication shows early promise as a primary therapy for severely symptomatic, drug-refractory hypertrophic obstructive cardiomyopathy (HOCM), Paul Sorajja, MD, said at the Annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
He and his coworkers at the Minneapolis Heart Institute developed the novel procedure and published the first experience in the world with it. But further study in a much larger patient population is needed before percutaneous mitral plication is ready for prime time as a treatment for symptomatic HOCM, according to Dr. Sorajja, director of the Center of Valve and Structural Heart Disease at the institute.
Surgical myectomy is the guideline-recommended standard for treatment of patients with disabling symptoms caused by left ventricular outflow tract obstruction in the setting of hypertrophic cardiomyopathy. Alcohol septal ablation is a widely utilized alternative in patients who are frail and elderly or otherwise high-risk surgical candidates, or who lack ready access to an experienced surgical myectomy center, where the best outcomes are achieved. But roughly 20% of patients evaluated for alcohol septal ablation don’t have a septal artery anatomy amenable to the procedure. Other patients are put off by the 10%-50% risk that they will require a permanent pacemaker after alcohol septal ablation, depending upon their baseline ECG. And a small percentage of well-chosen patients – less than 10% – will experience inadequate gradient relief following alcohol septal ablation. So there is room for a novel therapeutic approach.
A percutaneous mitral clip–based solution, while technically challenging, offers several advantages over surgical myectomy and alcohol septal ablation. It’s less invasive, doesn’t create a potentially arrhythmogenic ablation scar requiring a permanent pacemaker, and it targets the mitral valve directly, addressing the mitral regurgitation that causes the left ventricular outflow tract obstruction. In contrast, both surgical myectomy and alcohol septal ablation target the ventricular septum.
Dr. Sorajja summarized his experience to date with percutaneous mitral valve plication for symptomatic HOCM, which encompasses the six patients in the published study (J Am Coll Cardiol. 2016 Jun 21;67[24]:2811-8) and the four others treated since then. The procedure was completed with placement of a single MitraClip in 9 of 10 patients. One patient experienced cardiac tamponade, resulting in a halt of the procedure; in the other nine, the intervention eliminated systolic anterior motion and markedly decreased the intraoperative left ventricular outflow tract gradient and mitral regurgitation.
At 2-5 years of follow-up, all patients have improved from New York Heart Association functional class III preprocedurally to class I or II. One patient required alcohol septal ablation. High systolic left ventricular outflow tract velocities in excess of 4 cm/s have been documented via follow-up echocardiography in some patients, the significance of which is under study.
Dr. Sorajja reported receiving research funding from Abbott Structural, Boston Scientific, Edwards Lifesciences, and Medtronic, and serving as a consultant to those companies and several others.
SNOWMASS, COLO. – Percutaneous mitral valve plication shows early promise as a primary therapy for severely symptomatic, drug-refractory hypertrophic obstructive cardiomyopathy (HOCM), Paul Sorajja, MD, said at the Annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
He and his coworkers at the Minneapolis Heart Institute developed the novel procedure and published the first experience in the world with it. But further study in a much larger patient population is needed before percutaneous mitral plication is ready for prime time as a treatment for symptomatic HOCM, according to Dr. Sorajja, director of the Center of Valve and Structural Heart Disease at the institute.
Surgical myectomy is the guideline-recommended standard for treatment of patients with disabling symptoms caused by left ventricular outflow tract obstruction in the setting of hypertrophic cardiomyopathy. Alcohol septal ablation is a widely utilized alternative in patients who are frail and elderly or otherwise high-risk surgical candidates, or who lack ready access to an experienced surgical myectomy center, where the best outcomes are achieved. But roughly 20% of patients evaluated for alcohol septal ablation don’t have a septal artery anatomy amenable to the procedure. Other patients are put off by the 10%-50% risk that they will require a permanent pacemaker after alcohol septal ablation, depending upon their baseline ECG. And a small percentage of well-chosen patients – less than 10% – will experience inadequate gradient relief following alcohol septal ablation. So there is room for a novel therapeutic approach.
A percutaneous mitral clip–based solution, while technically challenging, offers several advantages over surgical myectomy and alcohol septal ablation. It’s less invasive, doesn’t create a potentially arrhythmogenic ablation scar requiring a permanent pacemaker, and it targets the mitral valve directly, addressing the mitral regurgitation that causes the left ventricular outflow tract obstruction. In contrast, both surgical myectomy and alcohol septal ablation target the ventricular septum.
Dr. Sorajja summarized his experience to date with percutaneous mitral valve plication for symptomatic HOCM, which encompasses the six patients in the published study (J Am Coll Cardiol. 2016 Jun 21;67[24]:2811-8) and the four others treated since then. The procedure was completed with placement of a single MitraClip in 9 of 10 patients. One patient experienced cardiac tamponade, resulting in a halt of the procedure; in the other nine, the intervention eliminated systolic anterior motion and markedly decreased the intraoperative left ventricular outflow tract gradient and mitral regurgitation.
At 2-5 years of follow-up, all patients have improved from New York Heart Association functional class III preprocedurally to class I or II. One patient required alcohol septal ablation. High systolic left ventricular outflow tract velocities in excess of 4 cm/s have been documented via follow-up echocardiography in some patients, the significance of which is under study.
Dr. Sorajja reported receiving research funding from Abbott Structural, Boston Scientific, Edwards Lifesciences, and Medtronic, and serving as a consultant to those companies and several others.
SNOWMASS, COLO. – Percutaneous mitral valve plication shows early promise as a primary therapy for severely symptomatic, drug-refractory hypertrophic obstructive cardiomyopathy (HOCM), Paul Sorajja, MD, said at the Annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
He and his coworkers at the Minneapolis Heart Institute developed the novel procedure and published the first experience in the world with it. But further study in a much larger patient population is needed before percutaneous mitral plication is ready for prime time as a treatment for symptomatic HOCM, according to Dr. Sorajja, director of the Center of Valve and Structural Heart Disease at the institute.
Surgical myectomy is the guideline-recommended standard for treatment of patients with disabling symptoms caused by left ventricular outflow tract obstruction in the setting of hypertrophic cardiomyopathy. Alcohol septal ablation is a widely utilized alternative in patients who are frail and elderly or otherwise high-risk surgical candidates, or who lack ready access to an experienced surgical myectomy center, where the best outcomes are achieved. But roughly 20% of patients evaluated for alcohol septal ablation don’t have a septal artery anatomy amenable to the procedure. Other patients are put off by the 10%-50% risk that they will require a permanent pacemaker after alcohol septal ablation, depending upon their baseline ECG. And a small percentage of well-chosen patients – less than 10% – will experience inadequate gradient relief following alcohol septal ablation. So there is room for a novel therapeutic approach.
A percutaneous mitral clip–based solution, while technically challenging, offers several advantages over surgical myectomy and alcohol septal ablation. It’s less invasive, doesn’t create a potentially arrhythmogenic ablation scar requiring a permanent pacemaker, and it targets the mitral valve directly, addressing the mitral regurgitation that causes the left ventricular outflow tract obstruction. In contrast, both surgical myectomy and alcohol septal ablation target the ventricular septum.
Dr. Sorajja summarized his experience to date with percutaneous mitral valve plication for symptomatic HOCM, which encompasses the six patients in the published study (J Am Coll Cardiol. 2016 Jun 21;67[24]:2811-8) and the four others treated since then. The procedure was completed with placement of a single MitraClip in 9 of 10 patients. One patient experienced cardiac tamponade, resulting in a halt of the procedure; in the other nine, the intervention eliminated systolic anterior motion and markedly decreased the intraoperative left ventricular outflow tract gradient and mitral regurgitation.
At 2-5 years of follow-up, all patients have improved from New York Heart Association functional class III preprocedurally to class I or II. One patient required alcohol septal ablation. High systolic left ventricular outflow tract velocities in excess of 4 cm/s have been documented via follow-up echocardiography in some patients, the significance of which is under study.
Dr. Sorajja reported receiving research funding from Abbott Structural, Boston Scientific, Edwards Lifesciences, and Medtronic, and serving as a consultant to those companies and several others.
REPORTING FROM ACC SNOWMASS 2019
Look for alcohol septal ablation in the next HOCM guideline
SNOWMASS, COLO. – Recent data on long-term outcomes of alcohol septal ablation for hypertrophic obstructive cardiomyopathy are “quite favorable” and will be considered in the deliberations of the task force charged with revising the 2011 American College of Cardiology/American Heart Association guidelines.
Paul Sorajja, MD, a member of the task force and director of the Center of Valve and Structural Heart Disease at the Minneapolis Heart Institute, explained that the 2011 ACC/AHA guidelines on hypertrophic cardiomyopathy took an appropriately cautious stance regarding alcohol septal ablation (ASA) in light of a 2010 Dutch report warning of an increased risk of sudden cardiac death following the procedure (Circ Heart Fail. 2010 May;3[3]:362-9) and a dearth of evidence to the contrary.
The 2011 guidelines recommend surgical myectomy performed in an experienced center as the class I treatment of choice for patients with severely symptomatic, drug-refractory hypertrophic obstructive cardiomyopathy (HOCM). ASA gets a class IIa recommendation for patients at high surgical risk, and is class III – meaning don’t do it – for patients under age 40 years if myectomy is a viable option (J Am Coll Cardiol. 2011 Dec 13;58[25]:e212-60), Dr. Sorajja noted at the Annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
However, the cautionary Dutch study that influenced the 2011 guidelines is considered controversial, he explained. It was small – just 91 patients – and the operators used twice the normal volume of alcohol, with a resultant much larger, potentially arrhythmogenic myocardial ablation scar. So, many experts have been eagerly awaiting additional long-term studies. And that long-sought data has recently been piling up. Since the 2011 guidelines, six long-term studies have been published, including one led by Dr. Sorajja (Circulation. 2012 Nov 13;126[20]:2374-80). The results have been consistently favorable, with 5-year survival rates of 87%-96%, in line with rates in the general population.
The largest of these studies included 1,197 patients who underwent ASA at seven centers in four European countries. The 30-day mortality and pacemaker implantation rates were significantly lower in patients aged up to 50 years, compared with those aged 65 and up. The annual mortality rate during a mean follow-up of 5.4 years was 1% in patients age 50 years and younger, 2.1% in those aged 51-64, and 5.1% in the oldest group. Arrhythmic events occurred at a rate of about 1% per year in all three age groups. And 95% of patients in the youngest group were in New York Heart Association class I or II at last follow-up (JACC Cardiovasc Interv. 2017 Jun 12;10[11]:1134-43).
In an accompanying editorial, Michael A. Fifer, MD, of Massachusetts General Hospital, Boston, commented that “high-volume surgical myectomy centers are few and far between” and there is “a clear inverse relation between [surgical] procedure volume and outcomes.”
The study “provides the most robust data to date regarding the outcomes of ASA in younger patients, precisely the type of data that were missing at the time of writing of the ACCF/AHA and European Society of Cardiology guidelines. Given the favorable outcomes of ASA in this age group, and the unavailability of high-volume myectomy programs in many geographic regions, the time has come to liberalize the indication for ASA in younger patients,” declared Dr. Fifer (JACC Cardiovasc Interv. 2017 Jun 12;10[11]:1144-6).
The second-largest long-term study of ASA was a recent report on 952 German patients with a minimum 6-year follow-up. The estimated 5-, 10-, and 15-year survival rates were 95.8%, 88.3%, and 79.7%, respectively. Estimated survival free of cardiac events was 98.9% at 5 years, 97.0% at 10 years, and 96.5% at 15 years. About 5% of patients received an implantable cardioverter defibrillator.
The investigators concluded, “In this study, PTSMA [percutaneous transluminal septal myocardial ablation] could be proofed as a safe procedure with ongoing symptomatic improvement and excellent long-term survival. Therefore, PTSMA is a reasonable alternative to surgical myectomy in HOCM.” (J Am Coll Cardiol. 2018 Dec 18;72[24]:3087-94) It’s way too early in the ACC/AHA guideline revision process to say what the new recommendations will be, according to Dr. Sorajja.
One unsettled issue, in his view, is whether ASA outcomes are significantly better in high-volume centers. A study of all 11,248 patients who underwent surgical myectomy of ASA during 2003-2011 in a large U.S. inpatient database concluded that undergoing surgical myectomy in a bottom-tertile-volume hospital was independently associated with an adjusted 210% increased risk of inpatient all-cause mortality and a 280% increased risk of bleeding, but that being in the lowest tertile of ASA hospital volume wasn’t independently associated with increased risk after adjustment for potential confounders (JAMA Cardiol. 2016 Jun 1;1:[3]:324-32).
However, Dr. Sorajja indicated he didn’t find the statistically adjusted results in the ASA cohort persuasive.
“I will tell you that the favorable results in the long-term studies came from hospitals in the highest-volume tertile,” the cardiologist said.
At present, he considers surgical myectomy the gold standard therapy. With well-selected patients for ASA – that is, those for whom imaging has identified an appropriate septal artery for delivery of the alcohol, along with no more than 24 mm of septal hypertrophy so the alcohol dose can be limited to a maximum of 20-25 cc – it’s reasonable to expect gradient relief in more than 90% of patients, surgical-like results with optimal relief of left ventricular outflow tract obstruction and a residual gradient of less than 10 mm Hg in about 75%, and a procedural mortality of about 1%, he said.
Dr. Sorajja reported receiving research funding from Abbott Structural, Boston Scientific, Edwards Lifesciences, and Medtronic, and serving as a consultant to those companies and several others.
SNOWMASS, COLO. – Recent data on long-term outcomes of alcohol septal ablation for hypertrophic obstructive cardiomyopathy are “quite favorable” and will be considered in the deliberations of the task force charged with revising the 2011 American College of Cardiology/American Heart Association guidelines.
Paul Sorajja, MD, a member of the task force and director of the Center of Valve and Structural Heart Disease at the Minneapolis Heart Institute, explained that the 2011 ACC/AHA guidelines on hypertrophic cardiomyopathy took an appropriately cautious stance regarding alcohol septal ablation (ASA) in light of a 2010 Dutch report warning of an increased risk of sudden cardiac death following the procedure (Circ Heart Fail. 2010 May;3[3]:362-9) and a dearth of evidence to the contrary.
The 2011 guidelines recommend surgical myectomy performed in an experienced center as the class I treatment of choice for patients with severely symptomatic, drug-refractory hypertrophic obstructive cardiomyopathy (HOCM). ASA gets a class IIa recommendation for patients at high surgical risk, and is class III – meaning don’t do it – for patients under age 40 years if myectomy is a viable option (J Am Coll Cardiol. 2011 Dec 13;58[25]:e212-60), Dr. Sorajja noted at the Annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
However, the cautionary Dutch study that influenced the 2011 guidelines is considered controversial, he explained. It was small – just 91 patients – and the operators used twice the normal volume of alcohol, with a resultant much larger, potentially arrhythmogenic myocardial ablation scar. So, many experts have been eagerly awaiting additional long-term studies. And that long-sought data has recently been piling up. Since the 2011 guidelines, six long-term studies have been published, including one led by Dr. Sorajja (Circulation. 2012 Nov 13;126[20]:2374-80). The results have been consistently favorable, with 5-year survival rates of 87%-96%, in line with rates in the general population.
The largest of these studies included 1,197 patients who underwent ASA at seven centers in four European countries. The 30-day mortality and pacemaker implantation rates were significantly lower in patients aged up to 50 years, compared with those aged 65 and up. The annual mortality rate during a mean follow-up of 5.4 years was 1% in patients age 50 years and younger, 2.1% in those aged 51-64, and 5.1% in the oldest group. Arrhythmic events occurred at a rate of about 1% per year in all three age groups. And 95% of patients in the youngest group were in New York Heart Association class I or II at last follow-up (JACC Cardiovasc Interv. 2017 Jun 12;10[11]:1134-43).
In an accompanying editorial, Michael A. Fifer, MD, of Massachusetts General Hospital, Boston, commented that “high-volume surgical myectomy centers are few and far between” and there is “a clear inverse relation between [surgical] procedure volume and outcomes.”
The study “provides the most robust data to date regarding the outcomes of ASA in younger patients, precisely the type of data that were missing at the time of writing of the ACCF/AHA and European Society of Cardiology guidelines. Given the favorable outcomes of ASA in this age group, and the unavailability of high-volume myectomy programs in many geographic regions, the time has come to liberalize the indication for ASA in younger patients,” declared Dr. Fifer (JACC Cardiovasc Interv. 2017 Jun 12;10[11]:1144-6).
The second-largest long-term study of ASA was a recent report on 952 German patients with a minimum 6-year follow-up. The estimated 5-, 10-, and 15-year survival rates were 95.8%, 88.3%, and 79.7%, respectively. Estimated survival free of cardiac events was 98.9% at 5 years, 97.0% at 10 years, and 96.5% at 15 years. About 5% of patients received an implantable cardioverter defibrillator.
The investigators concluded, “In this study, PTSMA [percutaneous transluminal septal myocardial ablation] could be proofed as a safe procedure with ongoing symptomatic improvement and excellent long-term survival. Therefore, PTSMA is a reasonable alternative to surgical myectomy in HOCM.” (J Am Coll Cardiol. 2018 Dec 18;72[24]:3087-94) It’s way too early in the ACC/AHA guideline revision process to say what the new recommendations will be, according to Dr. Sorajja.
One unsettled issue, in his view, is whether ASA outcomes are significantly better in high-volume centers. A study of all 11,248 patients who underwent surgical myectomy of ASA during 2003-2011 in a large U.S. inpatient database concluded that undergoing surgical myectomy in a bottom-tertile-volume hospital was independently associated with an adjusted 210% increased risk of inpatient all-cause mortality and a 280% increased risk of bleeding, but that being in the lowest tertile of ASA hospital volume wasn’t independently associated with increased risk after adjustment for potential confounders (JAMA Cardiol. 2016 Jun 1;1:[3]:324-32).
However, Dr. Sorajja indicated he didn’t find the statistically adjusted results in the ASA cohort persuasive.
“I will tell you that the favorable results in the long-term studies came from hospitals in the highest-volume tertile,” the cardiologist said.
At present, he considers surgical myectomy the gold standard therapy. With well-selected patients for ASA – that is, those for whom imaging has identified an appropriate septal artery for delivery of the alcohol, along with no more than 24 mm of septal hypertrophy so the alcohol dose can be limited to a maximum of 20-25 cc – it’s reasonable to expect gradient relief in more than 90% of patients, surgical-like results with optimal relief of left ventricular outflow tract obstruction and a residual gradient of less than 10 mm Hg in about 75%, and a procedural mortality of about 1%, he said.
Dr. Sorajja reported receiving research funding from Abbott Structural, Boston Scientific, Edwards Lifesciences, and Medtronic, and serving as a consultant to those companies and several others.
SNOWMASS, COLO. – Recent data on long-term outcomes of alcohol septal ablation for hypertrophic obstructive cardiomyopathy are “quite favorable” and will be considered in the deliberations of the task force charged with revising the 2011 American College of Cardiology/American Heart Association guidelines.
Paul Sorajja, MD, a member of the task force and director of the Center of Valve and Structural Heart Disease at the Minneapolis Heart Institute, explained that the 2011 ACC/AHA guidelines on hypertrophic cardiomyopathy took an appropriately cautious stance regarding alcohol septal ablation (ASA) in light of a 2010 Dutch report warning of an increased risk of sudden cardiac death following the procedure (Circ Heart Fail. 2010 May;3[3]:362-9) and a dearth of evidence to the contrary.
The 2011 guidelines recommend surgical myectomy performed in an experienced center as the class I treatment of choice for patients with severely symptomatic, drug-refractory hypertrophic obstructive cardiomyopathy (HOCM). ASA gets a class IIa recommendation for patients at high surgical risk, and is class III – meaning don’t do it – for patients under age 40 years if myectomy is a viable option (J Am Coll Cardiol. 2011 Dec 13;58[25]:e212-60), Dr. Sorajja noted at the Annual Cardiovascular Conference at Snowmass sponsored by the American College of Cardiology.
However, the cautionary Dutch study that influenced the 2011 guidelines is considered controversial, he explained. It was small – just 91 patients – and the operators used twice the normal volume of alcohol, with a resultant much larger, potentially arrhythmogenic myocardial ablation scar. So, many experts have been eagerly awaiting additional long-term studies. And that long-sought data has recently been piling up. Since the 2011 guidelines, six long-term studies have been published, including one led by Dr. Sorajja (Circulation. 2012 Nov 13;126[20]:2374-80). The results have been consistently favorable, with 5-year survival rates of 87%-96%, in line with rates in the general population.
The largest of these studies included 1,197 patients who underwent ASA at seven centers in four European countries. The 30-day mortality and pacemaker implantation rates were significantly lower in patients aged up to 50 years, compared with those aged 65 and up. The annual mortality rate during a mean follow-up of 5.4 years was 1% in patients age 50 years and younger, 2.1% in those aged 51-64, and 5.1% in the oldest group. Arrhythmic events occurred at a rate of about 1% per year in all three age groups. And 95% of patients in the youngest group were in New York Heart Association class I or II at last follow-up (JACC Cardiovasc Interv. 2017 Jun 12;10[11]:1134-43).
In an accompanying editorial, Michael A. Fifer, MD, of Massachusetts General Hospital, Boston, commented that “high-volume surgical myectomy centers are few and far between” and there is “a clear inverse relation between [surgical] procedure volume and outcomes.”
The study “provides the most robust data to date regarding the outcomes of ASA in younger patients, precisely the type of data that were missing at the time of writing of the ACCF/AHA and European Society of Cardiology guidelines. Given the favorable outcomes of ASA in this age group, and the unavailability of high-volume myectomy programs in many geographic regions, the time has come to liberalize the indication for ASA in younger patients,” declared Dr. Fifer (JACC Cardiovasc Interv. 2017 Jun 12;10[11]:1144-6).
The second-largest long-term study of ASA was a recent report on 952 German patients with a minimum 6-year follow-up. The estimated 5-, 10-, and 15-year survival rates were 95.8%, 88.3%, and 79.7%, respectively. Estimated survival free of cardiac events was 98.9% at 5 years, 97.0% at 10 years, and 96.5% at 15 years. About 5% of patients received an implantable cardioverter defibrillator.
The investigators concluded, “In this study, PTSMA [percutaneous transluminal septal myocardial ablation] could be proofed as a safe procedure with ongoing symptomatic improvement and excellent long-term survival. Therefore, PTSMA is a reasonable alternative to surgical myectomy in HOCM.” (J Am Coll Cardiol. 2018 Dec 18;72[24]:3087-94) It’s way too early in the ACC/AHA guideline revision process to say what the new recommendations will be, according to Dr. Sorajja.
One unsettled issue, in his view, is whether ASA outcomes are significantly better in high-volume centers. A study of all 11,248 patients who underwent surgical myectomy of ASA during 2003-2011 in a large U.S. inpatient database concluded that undergoing surgical myectomy in a bottom-tertile-volume hospital was independently associated with an adjusted 210% increased risk of inpatient all-cause mortality and a 280% increased risk of bleeding, but that being in the lowest tertile of ASA hospital volume wasn’t independently associated with increased risk after adjustment for potential confounders (JAMA Cardiol. 2016 Jun 1;1:[3]:324-32).
However, Dr. Sorajja indicated he didn’t find the statistically adjusted results in the ASA cohort persuasive.
“I will tell you that the favorable results in the long-term studies came from hospitals in the highest-volume tertile,” the cardiologist said.
At present, he considers surgical myectomy the gold standard therapy. With well-selected patients for ASA – that is, those for whom imaging has identified an appropriate septal artery for delivery of the alcohol, along with no more than 24 mm of septal hypertrophy so the alcohol dose can be limited to a maximum of 20-25 cc – it’s reasonable to expect gradient relief in more than 90% of patients, surgical-like results with optimal relief of left ventricular outflow tract obstruction and a residual gradient of less than 10 mm Hg in about 75%, and a procedural mortality of about 1%, he said.
Dr. Sorajja reported receiving research funding from Abbott Structural, Boston Scientific, Edwards Lifesciences, and Medtronic, and serving as a consultant to those companies and several others.
EXPERT ANALYSIS FROM ACC SNOWMASS 2019



















