User login
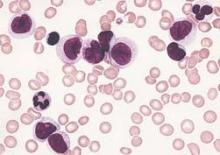
Targeting a protein to improve AML treatment

Photo courtesy of VCU
Massey Cancer Center
Preclinical experiments have shown that blocking production of the protein CHD4 may help increase the effectiveness of first-line treatments for acute myeloid leukemia (AML).
Researchers found that depleting AML cells of CHD4 makes them more susceptible to standard chemotherapeutic agents by reducing the cells’ ability to repair DNA damage.
Depleting CHD4 also decreased AML cells’ ability to form colonies in vitro and tumors in vivo.
On the other hand, CHD4 depletion did not have detrimental effects on healthy bone marrow cells. The cells were no more sensitive to chemotherapy, and their growth was not affected.
Researchers reported these results in Blood.
“We are very encouraged by these findings,” said study author Gordon Ginder, MD, of Virginia Commonwealth University in Richmond.
“Targeting the CHD4 protein could allow us to reduce chemotherapy doses, which could potentially mean more effective first- and second-line treatments with fewer serious side effects.”
CHD4 is involved in silencing tumor suppressor genes in cancer cells. Recently, it has been shown to play a role in repairing DNA damage.
With their experiments, Dr Ginder and his colleagues found that CHD4 depletion severely restricted the ability of AML cells to develop colonies in soft agar models and establish tumors in mouse models.
In addition, blocking the production of CHD4 rendered AML cells more sensitive to daunorubicin and cytarabine, both in vitro and in vivo.
“This study builds on our team’s efforts to understand the molecular processes through which epigenetic regulators impact gene expression,” Dr Ginder said.
“Future studies will attempt to uncover the detailed mechanism through which CHD4 decreases the ability of AML cells to initiate leukemia and will look for potential ways to target this important protein. The fact that it functions as an enzyme suggests it may be druggable.” ![]()

Photo courtesy of VCU
Massey Cancer Center
Preclinical experiments have shown that blocking production of the protein CHD4 may help increase the effectiveness of first-line treatments for acute myeloid leukemia (AML).
Researchers found that depleting AML cells of CHD4 makes them more susceptible to standard chemotherapeutic agents by reducing the cells’ ability to repair DNA damage.
Depleting CHD4 also decreased AML cells’ ability to form colonies in vitro and tumors in vivo.
On the other hand, CHD4 depletion did not have detrimental effects on healthy bone marrow cells. The cells were no more sensitive to chemotherapy, and their growth was not affected.
Researchers reported these results in Blood.
“We are very encouraged by these findings,” said study author Gordon Ginder, MD, of Virginia Commonwealth University in Richmond.
“Targeting the CHD4 protein could allow us to reduce chemotherapy doses, which could potentially mean more effective first- and second-line treatments with fewer serious side effects.”
CHD4 is involved in silencing tumor suppressor genes in cancer cells. Recently, it has been shown to play a role in repairing DNA damage.
With their experiments, Dr Ginder and his colleagues found that CHD4 depletion severely restricted the ability of AML cells to develop colonies in soft agar models and establish tumors in mouse models.
In addition, blocking the production of CHD4 rendered AML cells more sensitive to daunorubicin and cytarabine, both in vitro and in vivo.
“This study builds on our team’s efforts to understand the molecular processes through which epigenetic regulators impact gene expression,” Dr Ginder said.
“Future studies will attempt to uncover the detailed mechanism through which CHD4 decreases the ability of AML cells to initiate leukemia and will look for potential ways to target this important protein. The fact that it functions as an enzyme suggests it may be druggable.” ![]()

Photo courtesy of VCU
Massey Cancer Center
Preclinical experiments have shown that blocking production of the protein CHD4 may help increase the effectiveness of first-line treatments for acute myeloid leukemia (AML).
Researchers found that depleting AML cells of CHD4 makes them more susceptible to standard chemotherapeutic agents by reducing the cells’ ability to repair DNA damage.
Depleting CHD4 also decreased AML cells’ ability to form colonies in vitro and tumors in vivo.
On the other hand, CHD4 depletion did not have detrimental effects on healthy bone marrow cells. The cells were no more sensitive to chemotherapy, and their growth was not affected.
Researchers reported these results in Blood.
“We are very encouraged by these findings,” said study author Gordon Ginder, MD, of Virginia Commonwealth University in Richmond.
“Targeting the CHD4 protein could allow us to reduce chemotherapy doses, which could potentially mean more effective first- and second-line treatments with fewer serious side effects.”
CHD4 is involved in silencing tumor suppressor genes in cancer cells. Recently, it has been shown to play a role in repairing DNA damage.
With their experiments, Dr Ginder and his colleagues found that CHD4 depletion severely restricted the ability of AML cells to develop colonies in soft agar models and establish tumors in mouse models.
In addition, blocking the production of CHD4 rendered AML cells more sensitive to daunorubicin and cytarabine, both in vitro and in vivo.
“This study builds on our team’s efforts to understand the molecular processes through which epigenetic regulators impact gene expression,” Dr Ginder said.
“Future studies will attempt to uncover the detailed mechanism through which CHD4 decreases the ability of AML cells to initiate leukemia and will look for potential ways to target this important protein. The fact that it functions as an enzyme suggests it may be druggable.” ![]()
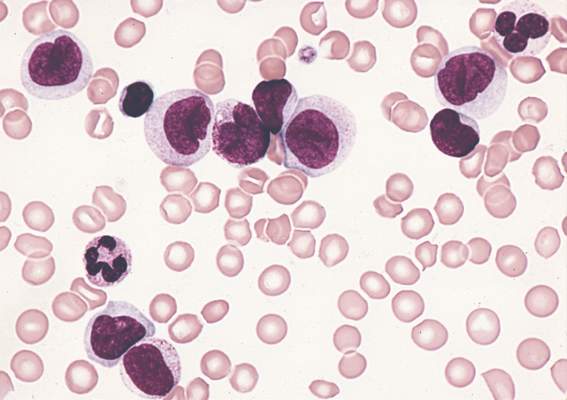
Study reveals tumor suppressor in AML
The protein-coding gene hnRNP K acts as a tumor suppressor in acute myeloid leukemia (AML), according to research published in Cancer Cell.
Investigators found that AML patients who carried a partial deletion of chromosome 9 also experienced a significant decrease in hnRNP K expression.
This deletion, 9q21.32, along with the decreased levels of hnRNP K, led to reduced survival and increased tumor formation.
“Our data implicates hnRNP K in the development of blood disorders and suggests it acts as a tumor suppressor,” said Sean Post, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“Both in vivo and in vitro results indicate that hnRNP K achieves this through regulation of key genetic pathways. Our study found that hnRNP K expression must be maintained for proper cellular regulation and to prevent tumor formation.”
Dr Post and his colleagues examined hnRNP K’s role in tumorigenesis by generating a mouse model harboring an Hnrnpk knockout allele.
They found that Hnrnpk haploinsufficiency resulted in reduced survival, increased tumor formation, genomic instability, and the development of transplantable hematopoietic neoplasms with myeloproliferation.
“Our findings showed that Hnrnpk haploinsufficiency led to tumor development by deregulating cell proliferation and differentiation programs through control of key cellular pathways, which suggests these pathways may be exploited by targeted therapies,” Dr Post said.
Specifically, he and his colleagues found that reduced hnRNP K expression attenuated p21 activation, downregulated C/EBP levels, and activated STAT3 signaling.
The investigators also analyzed samples from AML patients who harbored 9q21.32 and found a significant decrease in hnRNP K expression.
“It was clear that these changes in AML patients with the 9q21.32 deletion resulted in a tumor suppressor gene involved in blood cancer development,” Dr Post said. ![]()

The protein-coding gene hnRNP K acts as a tumor suppressor in acute myeloid leukemia (AML), according to research published in Cancer Cell.
Investigators found that AML patients who carried a partial deletion of chromosome 9 also experienced a significant decrease in hnRNP K expression.
This deletion, 9q21.32, along with the decreased levels of hnRNP K, led to reduced survival and increased tumor formation.
“Our data implicates hnRNP K in the development of blood disorders and suggests it acts as a tumor suppressor,” said Sean Post, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“Both in vivo and in vitro results indicate that hnRNP K achieves this through regulation of key genetic pathways. Our study found that hnRNP K expression must be maintained for proper cellular regulation and to prevent tumor formation.”
Dr Post and his colleagues examined hnRNP K’s role in tumorigenesis by generating a mouse model harboring an Hnrnpk knockout allele.
They found that Hnrnpk haploinsufficiency resulted in reduced survival, increased tumor formation, genomic instability, and the development of transplantable hematopoietic neoplasms with myeloproliferation.
“Our findings showed that Hnrnpk haploinsufficiency led to tumor development by deregulating cell proliferation and differentiation programs through control of key cellular pathways, which suggests these pathways may be exploited by targeted therapies,” Dr Post said.
Specifically, he and his colleagues found that reduced hnRNP K expression attenuated p21 activation, downregulated C/EBP levels, and activated STAT3 signaling.
The investigators also analyzed samples from AML patients who harbored 9q21.32 and found a significant decrease in hnRNP K expression.
“It was clear that these changes in AML patients with the 9q21.32 deletion resulted in a tumor suppressor gene involved in blood cancer development,” Dr Post said. ![]()

The protein-coding gene hnRNP K acts as a tumor suppressor in acute myeloid leukemia (AML), according to research published in Cancer Cell.
Investigators found that AML patients who carried a partial deletion of chromosome 9 also experienced a significant decrease in hnRNP K expression.
This deletion, 9q21.32, along with the decreased levels of hnRNP K, led to reduced survival and increased tumor formation.
“Our data implicates hnRNP K in the development of blood disorders and suggests it acts as a tumor suppressor,” said Sean Post, PhD, of The University of Texas MD Anderson Cancer Center in Houston.
“Both in vivo and in vitro results indicate that hnRNP K achieves this through regulation of key genetic pathways. Our study found that hnRNP K expression must be maintained for proper cellular regulation and to prevent tumor formation.”
Dr Post and his colleagues examined hnRNP K’s role in tumorigenesis by generating a mouse model harboring an Hnrnpk knockout allele.
They found that Hnrnpk haploinsufficiency resulted in reduced survival, increased tumor formation, genomic instability, and the development of transplantable hematopoietic neoplasms with myeloproliferation.
“Our findings showed that Hnrnpk haploinsufficiency led to tumor development by deregulating cell proliferation and differentiation programs through control of key cellular pathways, which suggests these pathways may be exploited by targeted therapies,” Dr Post said.
Specifically, he and his colleagues found that reduced hnRNP K expression attenuated p21 activation, downregulated C/EBP levels, and activated STAT3 signaling.
The investigators also analyzed samples from AML patients who harbored 9q21.32 and found a significant decrease in hnRNP K expression.
“It was clear that these changes in AML patients with the 9q21.32 deletion resulted in a tumor suppressor gene involved in blood cancer development,” Dr Post said. ![]()
CHMP endorses expanded indication for azacitidine

Image by Lance Liotta
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended expanding the marketing authorization for azacitidine for injection (Vidaza).
The CHMP is recommending that azacitidine be approved to treat adults age 65 and older with acute myeloid leukemia (AML) who are not eligible for hematopoietic stem cell transplant (HSCT) and have more than 30% blasts according to the WHO classification.
The CHMP’s recommendation will be reviewed by the European Commission (EC). The EC usually follows the CHMP’s recommendations and is expected to deliver its final decision in 2 months.
The CHMP said this new indication for azacitidine would bring significant clinical benefit in comparison with existing therapies. If the EC follows the CHMP’s recommendation, azacitidine will receive extended market protection in all its indications for an additional year throughout the European Economic Area.
Azacitidine is already approved in the European Economic Area for the treatment of HSCT-ineligible adults diagnosed with intermediate-2- and high-risk myelodysplastic syndromes; chronic myelomonocytic leukemia with 10%-29% marrow blasts without myeloproliferative disorder; or AML with 20%-30% blasts and multi-lineage dysplasia.
AML-001 trial
The CHMP’s recommendation to expand the indication of azacitidine in AML was based on data from the AML-001 trial. This randomized study included patients age 65 and older with newly diagnosed or secondary AML with greater than 30% blasts.
Patients were pre-selected to receive 1 of 3 regimens per investigator’s choice. This included intensive chemotherapy (standard 7+3 regimen), low-dose cytarabine (20 mg subcutaneously twice a day for 10 days of each 28-day cycle) or best supportive care only.
Patients were then randomized to receive either azacitidine (75 mg/m2/day subcutaneously for 7 days of each 28-day cycle, n=241) or their predetermined conventional care regimen (CCR, n=247).
Median overall survival, the study’s primary endpoint, was 10.4 months for patients receiving azacitidine and 6.5 months for patients receiving CCR (hazard ratio=0.85, P=0.1009).
One-year survival rates with azacitidine and CCR were 46.5% and 34.2%, respectively.
Grade 3/4 anemia occurred in 16% of patients who received azacitidine, 5% who received best supportive care, 23% who received low-dose cytarabine, and 14% who received intensive chemotherapy.
Grade 3/4 neutropenia occurred in 26%, 5%, 25%, and 33%, respectively. Grade 3/4 febrile neutropenia occurred in 28%, 28%, 30%, and 31%, respectively. And grade 3/4 thrombocytopenia occurred in 24%, 5%, 28%, and 21%, respectively. ![]()

Image by Lance Liotta
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended expanding the marketing authorization for azacitidine for injection (Vidaza).
The CHMP is recommending that azacitidine be approved to treat adults age 65 and older with acute myeloid leukemia (AML) who are not eligible for hematopoietic stem cell transplant (HSCT) and have more than 30% blasts according to the WHO classification.
The CHMP’s recommendation will be reviewed by the European Commission (EC). The EC usually follows the CHMP’s recommendations and is expected to deliver its final decision in 2 months.
The CHMP said this new indication for azacitidine would bring significant clinical benefit in comparison with existing therapies. If the EC follows the CHMP’s recommendation, azacitidine will receive extended market protection in all its indications for an additional year throughout the European Economic Area.
Azacitidine is already approved in the European Economic Area for the treatment of HSCT-ineligible adults diagnosed with intermediate-2- and high-risk myelodysplastic syndromes; chronic myelomonocytic leukemia with 10%-29% marrow blasts without myeloproliferative disorder; or AML with 20%-30% blasts and multi-lineage dysplasia.
AML-001 trial
The CHMP’s recommendation to expand the indication of azacitidine in AML was based on data from the AML-001 trial. This randomized study included patients age 65 and older with newly diagnosed or secondary AML with greater than 30% blasts.
Patients were pre-selected to receive 1 of 3 regimens per investigator’s choice. This included intensive chemotherapy (standard 7+3 regimen), low-dose cytarabine (20 mg subcutaneously twice a day for 10 days of each 28-day cycle) or best supportive care only.
Patients were then randomized to receive either azacitidine (75 mg/m2/day subcutaneously for 7 days of each 28-day cycle, n=241) or their predetermined conventional care regimen (CCR, n=247).
Median overall survival, the study’s primary endpoint, was 10.4 months for patients receiving azacitidine and 6.5 months for patients receiving CCR (hazard ratio=0.85, P=0.1009).
One-year survival rates with azacitidine and CCR were 46.5% and 34.2%, respectively.
Grade 3/4 anemia occurred in 16% of patients who received azacitidine, 5% who received best supportive care, 23% who received low-dose cytarabine, and 14% who received intensive chemotherapy.
Grade 3/4 neutropenia occurred in 26%, 5%, 25%, and 33%, respectively. Grade 3/4 febrile neutropenia occurred in 28%, 28%, 30%, and 31%, respectively. And grade 3/4 thrombocytopenia occurred in 24%, 5%, 28%, and 21%, respectively. ![]()

Image by Lance Liotta
The European Medicines Agency’s Committee for Medicinal Products for Human Use (CHMP) has recommended expanding the marketing authorization for azacitidine for injection (Vidaza).
The CHMP is recommending that azacitidine be approved to treat adults age 65 and older with acute myeloid leukemia (AML) who are not eligible for hematopoietic stem cell transplant (HSCT) and have more than 30% blasts according to the WHO classification.
The CHMP’s recommendation will be reviewed by the European Commission (EC). The EC usually follows the CHMP’s recommendations and is expected to deliver its final decision in 2 months.
The CHMP said this new indication for azacitidine would bring significant clinical benefit in comparison with existing therapies. If the EC follows the CHMP’s recommendation, azacitidine will receive extended market protection in all its indications for an additional year throughout the European Economic Area.
Azacitidine is already approved in the European Economic Area for the treatment of HSCT-ineligible adults diagnosed with intermediate-2- and high-risk myelodysplastic syndromes; chronic myelomonocytic leukemia with 10%-29% marrow blasts without myeloproliferative disorder; or AML with 20%-30% blasts and multi-lineage dysplasia.
AML-001 trial
The CHMP’s recommendation to expand the indication of azacitidine in AML was based on data from the AML-001 trial. This randomized study included patients age 65 and older with newly diagnosed or secondary AML with greater than 30% blasts.
Patients were pre-selected to receive 1 of 3 regimens per investigator’s choice. This included intensive chemotherapy (standard 7+3 regimen), low-dose cytarabine (20 mg subcutaneously twice a day for 10 days of each 28-day cycle) or best supportive care only.
Patients were then randomized to receive either azacitidine (75 mg/m2/day subcutaneously for 7 days of each 28-day cycle, n=241) or their predetermined conventional care regimen (CCR, n=247).
Median overall survival, the study’s primary endpoint, was 10.4 months for patients receiving azacitidine and 6.5 months for patients receiving CCR (hazard ratio=0.85, P=0.1009).
One-year survival rates with azacitidine and CCR were 46.5% and 34.2%, respectively.
Grade 3/4 anemia occurred in 16% of patients who received azacitidine, 5% who received best supportive care, 23% who received low-dose cytarabine, and 14% who received intensive chemotherapy.
Grade 3/4 neutropenia occurred in 26%, 5%, 25%, and 33%, respectively. Grade 3/4 febrile neutropenia occurred in 28%, 28%, 30%, and 31%, respectively. And grade 3/4 thrombocytopenia occurred in 24%, 5%, 28%, and 21%, respectively. ![]()
Variations in blood cancer survival across Europe

chemotherapy
Photo by Rhoda Baer
VIENNA—Results of the EUROCARE-5 study have revealed regional differences in survival for European patients with hematologic malignancies.
The data showed regional variations in 5-year relative survival rates for a number of cancers.
But the differences were particularly pronounced for leukemias, non-Hodgkin lymphomas (NHLs), and plasma cell neoplasms (PCNs).
Milena Sant, MD, of the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan, Italy, presented these results at the 2015 European Cancer Congress (LBA 1).
Data from this study have also been published in several articles in the October 2015 issue of the European Journal of Cancer.
EUROCARE-5 includes records from 22 million cancer patients diagnosed between 1978 and 2007. The latest data encompass more than 10 million patients (ages 15 and older) diagnosed from 1995 to 2007 and followed up to 2008.
The data came from 107 cancer registries in 29 countries. The researchers estimated 5-year relative survival and trends from 1999 to 2007 according to region—Ireland/UK, Northern Europe, Central Europe, Southern Europe, and Eastern Europe.
“In general, 5-year relative survival—survival that is adjusted for causes of death other than cancer—increased steadily over time in Europe, particularly in Eastern Europe, for most cancers,” Dr Sant said.
“However, the most dramatic geographical variations were observed for cancers of the blood where there have been recent advances in treatment, such as chronic myeloid and lymphocytic leukemias, non-Hodgkin lymphoma and 2 of its subtypes (follicular and diffuse large B-cell lymphoma), and multiple myeloma. Hodgkin lymphoma was the exception, with smaller regional variations and a fairly good prognosis in most countries.”
Hodgkin lymphoma and NHL
Of all the hematologic malignancies, 5-year relative survival was highest for Hodgkin lymphoma, at 80.8% (40,625 cases). Five-year survival was 79.4% in Ireland and the UK, 85% in Northern countries, and 74.3% in Eastern Europe, which was significantly below the European average (P<0.0001).
For NHL, the 5-year relative survival was 59.4% (329,204 cases). Survival rates for NHL patients ranged from 49.7% in Eastern Europe to 63.3% in Northern Europe.
CLL/SLL
For chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), the 5-year relative survival was 70.4% (81,914 cases). CLL/SLL survival ranged from 58% in Eastern Europe to about 74% in Central and Northern Europe.
The researchers noted that between-country variations in CLL/SLL survival were high in all regions. Outliers that were significantly below the regional average were Austria (67%), Croatia (52%), and Bulgaria (45.5%).
PCNs
PCNs included multiple myeloma, plasmacytoma, and plasma cell leukemias. The 5-year relative survival for all PCNs was 39.2% (94,024 cases).
PCN survival rates were lowest in Eastern Europe (31.7%), slightly higher in the UK/Ireland (35.9%), and between 39.1% and 42% in the rest of Europe.
Myeloid leukemias
Of all the hematologic malignancies, 5-year relative survival was poorest for patients with acute myeloid leukemia (AML), at 17.1% (57,026 cases).
AML survival rates in Ireland/UK (15.0%) and Eastern Europe (13.0%) were significantly below the European average. But AML survival in Sweden, Belgium, France, and Germany was significantly higher than the average (P<0.005).
Five-year relative survival for chronic myeloid leukemia (CML) was 52.9% (17,713 cases).
Of all the hematologic malignancies, the survival gap between Eastern Europe and the rest of Europe was highest for CML. Five-year survival for CML patients was 33% in Eastern Europe and ranged from 51% to 58% in the rest of Europe.
The researchers also said there were striking survival variations by country in all areas. They found significant deviations from the regional average in Sweden (69.7%), Scotland (64.6%), France (71.7%), Austria (48.2%), Croatia (37.8%), Estonia (48.9%), Czech Republic (45.2%), and Latvia (22.1%).
“Results from EUROCARE can help to identify regions of low survival where action is needed to improve patients’ outcomes,” Dr Sant noted.
“Population-based survival information is essential for physicians, policy-makers, administrators, researchers, and patient organizations who deal with the needs of cancer patients, as well as with the issue of the growing expenditure on healthcare.” ![]()

chemotherapy
Photo by Rhoda Baer
VIENNA—Results of the EUROCARE-5 study have revealed regional differences in survival for European patients with hematologic malignancies.
The data showed regional variations in 5-year relative survival rates for a number of cancers.
But the differences were particularly pronounced for leukemias, non-Hodgkin lymphomas (NHLs), and plasma cell neoplasms (PCNs).
Milena Sant, MD, of the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan, Italy, presented these results at the 2015 European Cancer Congress (LBA 1).
Data from this study have also been published in several articles in the October 2015 issue of the European Journal of Cancer.
EUROCARE-5 includes records from 22 million cancer patients diagnosed between 1978 and 2007. The latest data encompass more than 10 million patients (ages 15 and older) diagnosed from 1995 to 2007 and followed up to 2008.
The data came from 107 cancer registries in 29 countries. The researchers estimated 5-year relative survival and trends from 1999 to 2007 according to region—Ireland/UK, Northern Europe, Central Europe, Southern Europe, and Eastern Europe.
“In general, 5-year relative survival—survival that is adjusted for causes of death other than cancer—increased steadily over time in Europe, particularly in Eastern Europe, for most cancers,” Dr Sant said.
“However, the most dramatic geographical variations were observed for cancers of the blood where there have been recent advances in treatment, such as chronic myeloid and lymphocytic leukemias, non-Hodgkin lymphoma and 2 of its subtypes (follicular and diffuse large B-cell lymphoma), and multiple myeloma. Hodgkin lymphoma was the exception, with smaller regional variations and a fairly good prognosis in most countries.”
Hodgkin lymphoma and NHL
Of all the hematologic malignancies, 5-year relative survival was highest for Hodgkin lymphoma, at 80.8% (40,625 cases). Five-year survival was 79.4% in Ireland and the UK, 85% in Northern countries, and 74.3% in Eastern Europe, which was significantly below the European average (P<0.0001).
For NHL, the 5-year relative survival was 59.4% (329,204 cases). Survival rates for NHL patients ranged from 49.7% in Eastern Europe to 63.3% in Northern Europe.
CLL/SLL
For chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), the 5-year relative survival was 70.4% (81,914 cases). CLL/SLL survival ranged from 58% in Eastern Europe to about 74% in Central and Northern Europe.
The researchers noted that between-country variations in CLL/SLL survival were high in all regions. Outliers that were significantly below the regional average were Austria (67%), Croatia (52%), and Bulgaria (45.5%).
PCNs
PCNs included multiple myeloma, plasmacytoma, and plasma cell leukemias. The 5-year relative survival for all PCNs was 39.2% (94,024 cases).
PCN survival rates were lowest in Eastern Europe (31.7%), slightly higher in the UK/Ireland (35.9%), and between 39.1% and 42% in the rest of Europe.
Myeloid leukemias
Of all the hematologic malignancies, 5-year relative survival was poorest for patients with acute myeloid leukemia (AML), at 17.1% (57,026 cases).
AML survival rates in Ireland/UK (15.0%) and Eastern Europe (13.0%) were significantly below the European average. But AML survival in Sweden, Belgium, France, and Germany was significantly higher than the average (P<0.005).
Five-year relative survival for chronic myeloid leukemia (CML) was 52.9% (17,713 cases).
Of all the hematologic malignancies, the survival gap between Eastern Europe and the rest of Europe was highest for CML. Five-year survival for CML patients was 33% in Eastern Europe and ranged from 51% to 58% in the rest of Europe.
The researchers also said there were striking survival variations by country in all areas. They found significant deviations from the regional average in Sweden (69.7%), Scotland (64.6%), France (71.7%), Austria (48.2%), Croatia (37.8%), Estonia (48.9%), Czech Republic (45.2%), and Latvia (22.1%).
“Results from EUROCARE can help to identify regions of low survival where action is needed to improve patients’ outcomes,” Dr Sant noted.
“Population-based survival information is essential for physicians, policy-makers, administrators, researchers, and patient organizations who deal with the needs of cancer patients, as well as with the issue of the growing expenditure on healthcare.” ![]()

chemotherapy
Photo by Rhoda Baer
VIENNA—Results of the EUROCARE-5 study have revealed regional differences in survival for European patients with hematologic malignancies.
The data showed regional variations in 5-year relative survival rates for a number of cancers.
But the differences were particularly pronounced for leukemias, non-Hodgkin lymphomas (NHLs), and plasma cell neoplasms (PCNs).
Milena Sant, MD, of the Fondazione IRCCS Istituto Nazionale dei Tumori in Milan, Italy, presented these results at the 2015 European Cancer Congress (LBA 1).
Data from this study have also been published in several articles in the October 2015 issue of the European Journal of Cancer.
EUROCARE-5 includes records from 22 million cancer patients diagnosed between 1978 and 2007. The latest data encompass more than 10 million patients (ages 15 and older) diagnosed from 1995 to 2007 and followed up to 2008.
The data came from 107 cancer registries in 29 countries. The researchers estimated 5-year relative survival and trends from 1999 to 2007 according to region—Ireland/UK, Northern Europe, Central Europe, Southern Europe, and Eastern Europe.
“In general, 5-year relative survival—survival that is adjusted for causes of death other than cancer—increased steadily over time in Europe, particularly in Eastern Europe, for most cancers,” Dr Sant said.
“However, the most dramatic geographical variations were observed for cancers of the blood where there have been recent advances in treatment, such as chronic myeloid and lymphocytic leukemias, non-Hodgkin lymphoma and 2 of its subtypes (follicular and diffuse large B-cell lymphoma), and multiple myeloma. Hodgkin lymphoma was the exception, with smaller regional variations and a fairly good prognosis in most countries.”
Hodgkin lymphoma and NHL
Of all the hematologic malignancies, 5-year relative survival was highest for Hodgkin lymphoma, at 80.8% (40,625 cases). Five-year survival was 79.4% in Ireland and the UK, 85% in Northern countries, and 74.3% in Eastern Europe, which was significantly below the European average (P<0.0001).
For NHL, the 5-year relative survival was 59.4% (329,204 cases). Survival rates for NHL patients ranged from 49.7% in Eastern Europe to 63.3% in Northern Europe.
CLL/SLL
For chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), the 5-year relative survival was 70.4% (81,914 cases). CLL/SLL survival ranged from 58% in Eastern Europe to about 74% in Central and Northern Europe.
The researchers noted that between-country variations in CLL/SLL survival were high in all regions. Outliers that were significantly below the regional average were Austria (67%), Croatia (52%), and Bulgaria (45.5%).
PCNs
PCNs included multiple myeloma, plasmacytoma, and plasma cell leukemias. The 5-year relative survival for all PCNs was 39.2% (94,024 cases).
PCN survival rates were lowest in Eastern Europe (31.7%), slightly higher in the UK/Ireland (35.9%), and between 39.1% and 42% in the rest of Europe.
Myeloid leukemias
Of all the hematologic malignancies, 5-year relative survival was poorest for patients with acute myeloid leukemia (AML), at 17.1% (57,026 cases).
AML survival rates in Ireland/UK (15.0%) and Eastern Europe (13.0%) were significantly below the European average. But AML survival in Sweden, Belgium, France, and Germany was significantly higher than the average (P<0.005).
Five-year relative survival for chronic myeloid leukemia (CML) was 52.9% (17,713 cases).
Of all the hematologic malignancies, the survival gap between Eastern Europe and the rest of Europe was highest for CML. Five-year survival for CML patients was 33% in Eastern Europe and ranged from 51% to 58% in the rest of Europe.
The researchers also said there were striking survival variations by country in all areas. They found significant deviations from the regional average in Sweden (69.7%), Scotland (64.6%), France (71.7%), Austria (48.2%), Croatia (37.8%), Estonia (48.9%), Czech Republic (45.2%), and Latvia (22.1%).
“Results from EUROCARE can help to identify regions of low survival where action is needed to improve patients’ outcomes,” Dr Sant noted.
“Population-based survival information is essential for physicians, policy-makers, administrators, researchers, and patient organizations who deal with the needs of cancer patients, as well as with the issue of the growing expenditure on healthcare.” ![]()
Abs from transplanted AML patients enhance GvL effect in vitro

NEW YORK—Investigators have found that B cells may play a role in stimulating graft-versus-leukemia (GvL) responses in patients with acute myeloid leukemia (AML) who have undergone allogeneic hematopoietic stem cell transplant (HSCT).
The team created B cell lines from these patients, isolated AML-specific antibodies, and found that these antibodies can induce the death of AML cells through oncosis.
Oncosis is a non-apoptotic type of cell death that involves swelling and coagulation of the cytoplasm.
Mette Hazenberg, MD, PhD, from the Academic Medical Center in Amsterdam, The Netherlands, reported these findings at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference as poster B052.
The investigators cloned B cells from 3 high-risk AML patients who had a strong GvL response after HSCT.
The team transduced memory B cells from the patients’ peripheral blood using BCL-6 and BCL-xL. They then screened the B cells for those that produced antibodies that bound specifically to surface antigens on AML cell lines and blasts.
Six of the 15 AML antibodies retrieved from the patients bound specifically to snRNp200. In normal cells, snRNp200 is in the nucleus, but, in AML, it is exposed on the cell membrane.
The investigators then confirmed this by ELISA.
They found 7 of the 15 AML antibodies directly lysed AML blasts without the addition of effector cells or complement. Time-lapse images showed that cell death by the AML antibodies occurred rapidly, within minutes after incubation.
“The leukemia blasts popped like balloons,” Dr Hazenberg said.
The investigators confirmed that the antibodies induced cell death by oncosis and that oncosis occurred independently of temperature. The antibodies were cytotoxic at 4°C and 37°C.
Cytotoxicity of the antibodies could be blocked by the membrane stabilizer cytochalasin D but not by apoptosis inhibitors.
The team concluded that a potent GvL response could be mediated by these antibodies against tumor-associated antigens on AML cells.
Dr Hazenberg’s hope is that, at some point, these antibodies can be combined with chemotherapy—as is rituximab—so patients won’t need to undergo transplant. ![]()

NEW YORK—Investigators have found that B cells may play a role in stimulating graft-versus-leukemia (GvL) responses in patients with acute myeloid leukemia (AML) who have undergone allogeneic hematopoietic stem cell transplant (HSCT).
The team created B cell lines from these patients, isolated AML-specific antibodies, and found that these antibodies can induce the death of AML cells through oncosis.
Oncosis is a non-apoptotic type of cell death that involves swelling and coagulation of the cytoplasm.
Mette Hazenberg, MD, PhD, from the Academic Medical Center in Amsterdam, The Netherlands, reported these findings at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference as poster B052.
The investigators cloned B cells from 3 high-risk AML patients who had a strong GvL response after HSCT.
The team transduced memory B cells from the patients’ peripheral blood using BCL-6 and BCL-xL. They then screened the B cells for those that produced antibodies that bound specifically to surface antigens on AML cell lines and blasts.
Six of the 15 AML antibodies retrieved from the patients bound specifically to snRNp200. In normal cells, snRNp200 is in the nucleus, but, in AML, it is exposed on the cell membrane.
The investigators then confirmed this by ELISA.
They found 7 of the 15 AML antibodies directly lysed AML blasts without the addition of effector cells or complement. Time-lapse images showed that cell death by the AML antibodies occurred rapidly, within minutes after incubation.
“The leukemia blasts popped like balloons,” Dr Hazenberg said.
The investigators confirmed that the antibodies induced cell death by oncosis and that oncosis occurred independently of temperature. The antibodies were cytotoxic at 4°C and 37°C.
Cytotoxicity of the antibodies could be blocked by the membrane stabilizer cytochalasin D but not by apoptosis inhibitors.
The team concluded that a potent GvL response could be mediated by these antibodies against tumor-associated antigens on AML cells.
Dr Hazenberg’s hope is that, at some point, these antibodies can be combined with chemotherapy—as is rituximab—so patients won’t need to undergo transplant. ![]()

NEW YORK—Investigators have found that B cells may play a role in stimulating graft-versus-leukemia (GvL) responses in patients with acute myeloid leukemia (AML) who have undergone allogeneic hematopoietic stem cell transplant (HSCT).
The team created B cell lines from these patients, isolated AML-specific antibodies, and found that these antibodies can induce the death of AML cells through oncosis.
Oncosis is a non-apoptotic type of cell death that involves swelling and coagulation of the cytoplasm.
Mette Hazenberg, MD, PhD, from the Academic Medical Center in Amsterdam, The Netherlands, reported these findings at the inaugural CRI-CIMT-EATI-AACR International Cancer Immunotherapy Conference as poster B052.
The investigators cloned B cells from 3 high-risk AML patients who had a strong GvL response after HSCT.
The team transduced memory B cells from the patients’ peripheral blood using BCL-6 and BCL-xL. They then screened the B cells for those that produced antibodies that bound specifically to surface antigens on AML cell lines and blasts.
Six of the 15 AML antibodies retrieved from the patients bound specifically to snRNp200. In normal cells, snRNp200 is in the nucleus, but, in AML, it is exposed on the cell membrane.
The investigators then confirmed this by ELISA.
They found 7 of the 15 AML antibodies directly lysed AML blasts without the addition of effector cells or complement. Time-lapse images showed that cell death by the AML antibodies occurred rapidly, within minutes after incubation.
“The leukemia blasts popped like balloons,” Dr Hazenberg said.
The investigators confirmed that the antibodies induced cell death by oncosis and that oncosis occurred independently of temperature. The antibodies were cytotoxic at 4°C and 37°C.
Cytotoxicity of the antibodies could be blocked by the membrane stabilizer cytochalasin D but not by apoptosis inhibitors.
The team concluded that a potent GvL response could be mediated by these antibodies against tumor-associated antigens on AML cells.
Dr Hazenberg’s hope is that, at some point, these antibodies can be combined with chemotherapy—as is rituximab—so patients won’t need to undergo transplant. ![]()
Monitoring microbiome may help reduce infection in AML

Photo by Rhoda Baer
SAN DIEGO—Monitoring the microbiome during chemotherapy might help reduce infections in leukemia patients, according to research presented at ICAAC/ICC 2015.
The researchers studied buccal and fecal samples from patients with acute myeloid leukemia (AML) who were undergoing induction chemotherapy.
This revealed that decreased microbial diversity was associated with an increased risk of infection.
Jessica Galloway-Peña, PhD, of The University of Texas MD Anderson Cancer Center in Houston, and her colleagues described this work in a poster presentation at the meeting (poster B-993).
The team analyzed samples from 34 AML patients. All of the patients received prophylactic antimicrobials, and 91% received systemic antibiotics. The patients received an average of 5.4 different antibiotics for an average duration of 6.5 days.
The researchers collected buccal and fecal specimens from the patients every 96 hours over the course of induction chemotherapy. This yielded 276 buccal and 202 fecal samples—an average of 8 oral and 6 stool samples per patient.
The team used 16S rRNA V4 region sequencing to assign bacterial taxa and calculate α- and β-diversities. They had a total of 16,082,550 high-quality reads.
Analyzing these data, the researchers found that decreased microbial diversity, both at baseline and throughout induction, was associated with an increased risk of infection.
“We found the baseline microbial diversities from stool samples were significantly lower in patients that developed infections during chemotherapy compared to those that did not [P=0.006],” Dr Galloway-Peña said.
She and her colleagues also found that, overall, there was a significant decrease in oral (P=0.006) and intestinal (P<0.001) microbial diversity over the course of chemotherapy, although not all patients experienced decreases. There was a linear correlation between oral and stool microbiome changes (P=0.004).
In addition, over the course of induction, there was a significant increase (P=0.02) in the rates of bacterial domination (>30% of the microbiome dominated by 1 organism) by common causes of bacteremia, such as Streptococcus, Bacteriodes, Rothia, and Staphylococcus.
However, if patients were able to maintain a healthy microbiome overall or if they experienced an increase in microbial diversity over the induction course, they remained infection-free in the 90 days after induction.
Dr Galloway-Peña and her colleagues also assessed the role common antibiotics play in microbial diversity. And they found that carbapenems significantly decreased diversity.
There was a significant difference in oral and stool diversity when patients received carbapenems for at least 72 hours and when they did not (P=0.03). But there was no significant difference for piperacillin-tazobactam (P=1.0) or cefepime (P=0.48).
“This study shows that, in the future, doctors could use microbiome sampling in order to predict the chance of infectious complications during chemotherapy and that monitoring of a patient’s microbiome during induction chemotherapy could also predict their risk for microbial-related illness during subsequent treatments,” Dr Galloway-Peña said.
In addition, monitoring the microbiome could potentially mitigate the overuse of antimicrobials by allowing physicians to stratify patients according to their risk of developing an infection. ![]()

Photo by Rhoda Baer
SAN DIEGO—Monitoring the microbiome during chemotherapy might help reduce infections in leukemia patients, according to research presented at ICAAC/ICC 2015.
The researchers studied buccal and fecal samples from patients with acute myeloid leukemia (AML) who were undergoing induction chemotherapy.
This revealed that decreased microbial diversity was associated with an increased risk of infection.
Jessica Galloway-Peña, PhD, of The University of Texas MD Anderson Cancer Center in Houston, and her colleagues described this work in a poster presentation at the meeting (poster B-993).
The team analyzed samples from 34 AML patients. All of the patients received prophylactic antimicrobials, and 91% received systemic antibiotics. The patients received an average of 5.4 different antibiotics for an average duration of 6.5 days.
The researchers collected buccal and fecal specimens from the patients every 96 hours over the course of induction chemotherapy. This yielded 276 buccal and 202 fecal samples—an average of 8 oral and 6 stool samples per patient.
The team used 16S rRNA V4 region sequencing to assign bacterial taxa and calculate α- and β-diversities. They had a total of 16,082,550 high-quality reads.
Analyzing these data, the researchers found that decreased microbial diversity, both at baseline and throughout induction, was associated with an increased risk of infection.
“We found the baseline microbial diversities from stool samples were significantly lower in patients that developed infections during chemotherapy compared to those that did not [P=0.006],” Dr Galloway-Peña said.
She and her colleagues also found that, overall, there was a significant decrease in oral (P=0.006) and intestinal (P<0.001) microbial diversity over the course of chemotherapy, although not all patients experienced decreases. There was a linear correlation between oral and stool microbiome changes (P=0.004).
In addition, over the course of induction, there was a significant increase (P=0.02) in the rates of bacterial domination (>30% of the microbiome dominated by 1 organism) by common causes of bacteremia, such as Streptococcus, Bacteriodes, Rothia, and Staphylococcus.
However, if patients were able to maintain a healthy microbiome overall or if they experienced an increase in microbial diversity over the induction course, they remained infection-free in the 90 days after induction.
Dr Galloway-Peña and her colleagues also assessed the role common antibiotics play in microbial diversity. And they found that carbapenems significantly decreased diversity.
There was a significant difference in oral and stool diversity when patients received carbapenems for at least 72 hours and when they did not (P=0.03). But there was no significant difference for piperacillin-tazobactam (P=1.0) or cefepime (P=0.48).
“This study shows that, in the future, doctors could use microbiome sampling in order to predict the chance of infectious complications during chemotherapy and that monitoring of a patient’s microbiome during induction chemotherapy could also predict their risk for microbial-related illness during subsequent treatments,” Dr Galloway-Peña said.
In addition, monitoring the microbiome could potentially mitigate the overuse of antimicrobials by allowing physicians to stratify patients according to their risk of developing an infection. ![]()

Photo by Rhoda Baer
SAN DIEGO—Monitoring the microbiome during chemotherapy might help reduce infections in leukemia patients, according to research presented at ICAAC/ICC 2015.
The researchers studied buccal and fecal samples from patients with acute myeloid leukemia (AML) who were undergoing induction chemotherapy.
This revealed that decreased microbial diversity was associated with an increased risk of infection.
Jessica Galloway-Peña, PhD, of The University of Texas MD Anderson Cancer Center in Houston, and her colleagues described this work in a poster presentation at the meeting (poster B-993).
The team analyzed samples from 34 AML patients. All of the patients received prophylactic antimicrobials, and 91% received systemic antibiotics. The patients received an average of 5.4 different antibiotics for an average duration of 6.5 days.
The researchers collected buccal and fecal specimens from the patients every 96 hours over the course of induction chemotherapy. This yielded 276 buccal and 202 fecal samples—an average of 8 oral and 6 stool samples per patient.
The team used 16S rRNA V4 region sequencing to assign bacterial taxa and calculate α- and β-diversities. They had a total of 16,082,550 high-quality reads.
Analyzing these data, the researchers found that decreased microbial diversity, both at baseline and throughout induction, was associated with an increased risk of infection.
“We found the baseline microbial diversities from stool samples were significantly lower in patients that developed infections during chemotherapy compared to those that did not [P=0.006],” Dr Galloway-Peña said.
She and her colleagues also found that, overall, there was a significant decrease in oral (P=0.006) and intestinal (P<0.001) microbial diversity over the course of chemotherapy, although not all patients experienced decreases. There was a linear correlation between oral and stool microbiome changes (P=0.004).
In addition, over the course of induction, there was a significant increase (P=0.02) in the rates of bacterial domination (>30% of the microbiome dominated by 1 organism) by common causes of bacteremia, such as Streptococcus, Bacteriodes, Rothia, and Staphylococcus.
However, if patients were able to maintain a healthy microbiome overall or if they experienced an increase in microbial diversity over the induction course, they remained infection-free in the 90 days after induction.
Dr Galloway-Peña and her colleagues also assessed the role common antibiotics play in microbial diversity. And they found that carbapenems significantly decreased diversity.
There was a significant difference in oral and stool diversity when patients received carbapenems for at least 72 hours and when they did not (P=0.03). But there was no significant difference for piperacillin-tazobactam (P=1.0) or cefepime (P=0.48).
“This study shows that, in the future, doctors could use microbiome sampling in order to predict the chance of infectious complications during chemotherapy and that monitoring of a patient’s microbiome during induction chemotherapy could also predict their risk for microbial-related illness during subsequent treatments,” Dr Galloway-Peña said.
In addition, monitoring the microbiome could potentially mitigate the overuse of antimicrobials by allowing physicians to stratify patients according to their risk of developing an infection. ![]()
CDK inhibitor active against lymphoma too

HOUSTON—Preclinical data suggest the second-generation cyclin-dependent kinase (CDK) inhibitor CYC065 is active against lymphomas as well as leukemias.
Previous research showed that CYC065 can fight acute myeloid leukemia (AML) in vitro and in vivo.
New research shows that CYC065 can decrease cell viability in a range of B-cell lymphoma cell lines, and the drug synergizes with both venetoclax and cytarabine.
Sheelagh Frame, PhD, and her colleagues presented these results at the SOHO 2015 Annual Meeting (poster 213). All of the investigators involved in the research are employees of Cyclacel Ltd., the company developing CYC065.
The investigators analyzed the anticancer activity of CYC065 in a range of cell lines and found that CYC065 induced apoptosis by inhibiting the expression of CDK9-dependent oncogenic transcripts, including Mcl-1, c-Myc, Hoxa9, and Meis1.
Results in AML
Experiments in the AML-MLL cell line MOLM-13 showed that short pulses of CYC065 (6 hours), rather than continuous treatment, were sufficient to achieve maximal cytotoxicity.
CYC065 induced apoptosis, in a dose-dependent manner, in other AML cell lines as well, including EOL-1 (MLL-PTD), MV4-11 (MLL-AF4, FLT3-ITD, and trisomy chr 8), HL60 (Myc amplified), and Kasumi-1.
The investigators noted that AML cell lines with MLL rearrangements were especially sensitive to CYC065, and the reliance of AML on Mcl-1 confers sensitivity to CYC065.
They also found evidence to suggest that Bak and Bcl-xL levels may be predictive of CYC065 response in AML. Cell lines that were less sensitive to treatment had high levels of Bcl-xL and negligible levels of Bak.
B-cell lymphomas
In B-cell lymphoma cell lines, the investigators observed a dose-dependent reduction in cell viability after CYC065 treatment (8-hour pulses).
CYC065 proved most effective in cell lines without genomic alterations associated with poor prognosis (HT and U-698-M) and in cell lines with Myc rearrangements (SU-DHL-8 and WILL-1).
The drug also decreased cell viability—but to a lesser degree—in cell lines with Bcl-2 rearrangements (SU-DHL-4 and U2932) and in double-hit lymphoma cell lines (MAVER-1, RI-1, SC-1, and SU-DHL-10).
The investigators therefore theorized that combining CYC065 with a Bcl-2 inhibitor might prove more effective in these cell lines.
CYC065 in combination
CYC065 synergized with the Bcl-2 inhibitor venetoclax in all B-cell lymphoma cell lines tested. The drugs were “strongly synergistic” in U2932 and RI-1 cell lines but simply “synergistic” in SU-CHL-4 and MAVER-1 cell lines.
CYC065 also synergized with cytarabine to fight AML. The combination proved synergistic at a range of doses in the HL60 and MV4-11 cell lines.
Considering these results together, the investigators concluded that CYC065 has shown potential for treating a range of leukemias and lymphomas with unmet clinical need, including MLL-rearranged leukemia and Myc-driven lymphoma.
And the drug might prove effective in combination with standard cytotoxic agents or agents targeting apoptotic regulators. ![]()

HOUSTON—Preclinical data suggest the second-generation cyclin-dependent kinase (CDK) inhibitor CYC065 is active against lymphomas as well as leukemias.
Previous research showed that CYC065 can fight acute myeloid leukemia (AML) in vitro and in vivo.
New research shows that CYC065 can decrease cell viability in a range of B-cell lymphoma cell lines, and the drug synergizes with both venetoclax and cytarabine.
Sheelagh Frame, PhD, and her colleagues presented these results at the SOHO 2015 Annual Meeting (poster 213). All of the investigators involved in the research are employees of Cyclacel Ltd., the company developing CYC065.
The investigators analyzed the anticancer activity of CYC065 in a range of cell lines and found that CYC065 induced apoptosis by inhibiting the expression of CDK9-dependent oncogenic transcripts, including Mcl-1, c-Myc, Hoxa9, and Meis1.
Results in AML
Experiments in the AML-MLL cell line MOLM-13 showed that short pulses of CYC065 (6 hours), rather than continuous treatment, were sufficient to achieve maximal cytotoxicity.
CYC065 induced apoptosis, in a dose-dependent manner, in other AML cell lines as well, including EOL-1 (MLL-PTD), MV4-11 (MLL-AF4, FLT3-ITD, and trisomy chr 8), HL60 (Myc amplified), and Kasumi-1.
The investigators noted that AML cell lines with MLL rearrangements were especially sensitive to CYC065, and the reliance of AML on Mcl-1 confers sensitivity to CYC065.
They also found evidence to suggest that Bak and Bcl-xL levels may be predictive of CYC065 response in AML. Cell lines that were less sensitive to treatment had high levels of Bcl-xL and negligible levels of Bak.
B-cell lymphomas
In B-cell lymphoma cell lines, the investigators observed a dose-dependent reduction in cell viability after CYC065 treatment (8-hour pulses).
CYC065 proved most effective in cell lines without genomic alterations associated with poor prognosis (HT and U-698-M) and in cell lines with Myc rearrangements (SU-DHL-8 and WILL-1).
The drug also decreased cell viability—but to a lesser degree—in cell lines with Bcl-2 rearrangements (SU-DHL-4 and U2932) and in double-hit lymphoma cell lines (MAVER-1, RI-1, SC-1, and SU-DHL-10).
The investigators therefore theorized that combining CYC065 with a Bcl-2 inhibitor might prove more effective in these cell lines.
CYC065 in combination
CYC065 synergized with the Bcl-2 inhibitor venetoclax in all B-cell lymphoma cell lines tested. The drugs were “strongly synergistic” in U2932 and RI-1 cell lines but simply “synergistic” in SU-CHL-4 and MAVER-1 cell lines.
CYC065 also synergized with cytarabine to fight AML. The combination proved synergistic at a range of doses in the HL60 and MV4-11 cell lines.
Considering these results together, the investigators concluded that CYC065 has shown potential for treating a range of leukemias and lymphomas with unmet clinical need, including MLL-rearranged leukemia and Myc-driven lymphoma.
And the drug might prove effective in combination with standard cytotoxic agents or agents targeting apoptotic regulators. ![]()

HOUSTON—Preclinical data suggest the second-generation cyclin-dependent kinase (CDK) inhibitor CYC065 is active against lymphomas as well as leukemias.
Previous research showed that CYC065 can fight acute myeloid leukemia (AML) in vitro and in vivo.
New research shows that CYC065 can decrease cell viability in a range of B-cell lymphoma cell lines, and the drug synergizes with both venetoclax and cytarabine.
Sheelagh Frame, PhD, and her colleagues presented these results at the SOHO 2015 Annual Meeting (poster 213). All of the investigators involved in the research are employees of Cyclacel Ltd., the company developing CYC065.
The investigators analyzed the anticancer activity of CYC065 in a range of cell lines and found that CYC065 induced apoptosis by inhibiting the expression of CDK9-dependent oncogenic transcripts, including Mcl-1, c-Myc, Hoxa9, and Meis1.
Results in AML
Experiments in the AML-MLL cell line MOLM-13 showed that short pulses of CYC065 (6 hours), rather than continuous treatment, were sufficient to achieve maximal cytotoxicity.
CYC065 induced apoptosis, in a dose-dependent manner, in other AML cell lines as well, including EOL-1 (MLL-PTD), MV4-11 (MLL-AF4, FLT3-ITD, and trisomy chr 8), HL60 (Myc amplified), and Kasumi-1.
The investigators noted that AML cell lines with MLL rearrangements were especially sensitive to CYC065, and the reliance of AML on Mcl-1 confers sensitivity to CYC065.
They also found evidence to suggest that Bak and Bcl-xL levels may be predictive of CYC065 response in AML. Cell lines that were less sensitive to treatment had high levels of Bcl-xL and negligible levels of Bak.
B-cell lymphomas
In B-cell lymphoma cell lines, the investigators observed a dose-dependent reduction in cell viability after CYC065 treatment (8-hour pulses).
CYC065 proved most effective in cell lines without genomic alterations associated with poor prognosis (HT and U-698-M) and in cell lines with Myc rearrangements (SU-DHL-8 and WILL-1).
The drug also decreased cell viability—but to a lesser degree—in cell lines with Bcl-2 rearrangements (SU-DHL-4 and U2932) and in double-hit lymphoma cell lines (MAVER-1, RI-1, SC-1, and SU-DHL-10).
The investigators therefore theorized that combining CYC065 with a Bcl-2 inhibitor might prove more effective in these cell lines.
CYC065 in combination
CYC065 synergized with the Bcl-2 inhibitor venetoclax in all B-cell lymphoma cell lines tested. The drugs were “strongly synergistic” in U2932 and RI-1 cell lines but simply “synergistic” in SU-CHL-4 and MAVER-1 cell lines.
CYC065 also synergized with cytarabine to fight AML. The combination proved synergistic at a range of doses in the HL60 and MV4-11 cell lines.
Considering these results together, the investigators concluded that CYC065 has shown potential for treating a range of leukemias and lymphomas with unmet clinical need, including MLL-rearranged leukemia and Myc-driven lymphoma.
And the drug might prove effective in combination with standard cytotoxic agents or agents targeting apoptotic regulators.
Advances in AML understanding could translate to improved therapy
An understanding of the various molecular differences between acute myeloid leukemia (AML) particles is guiding the search for new drug combinations targeting a variety of AML cellular processes, according to research published in the New England Journal of Medicine.
After a brief overview of disease classification and prognostic factors for AML, the authors, led by Dr. Hartmut Döhner of University Hospital Ulm in Germany, described a number of new treatment strategies. These approaches include directly addressing mutant proteins by targeting mutation-specific dependencies, developing inhibitors that could cut down on the occurrence of mutations in receptor tyrosine kinase genes, and developing new epigenetic therapies based on targeting specific mutant metabolic enzymes such as IDH1 and IDH2.
Other strategies, such as antibody therapy that focuses on the development of new monoclonal antibodies targeting CD33, and new formulations of older cytotoxic agents, also are being developed or are in clinical trial stages; however, many of these developments have not yet been translated into clinical practice, the researchers noted.
“New compounds hold promise to improve treatment outcomes; however, it is unlikely that any of these compounds, when used as single agents, will cure the disease. A major challenge will be to identify predictors for a response to specific agents, which will allow for the rational design of combinatorial therapies,” they wrote.
Dr. Döhner and a coauthor disclosed ties with several pharmaceutical companies.
Read the article here.
An understanding of the various molecular differences between acute myeloid leukemia (AML) particles is guiding the search for new drug combinations targeting a variety of AML cellular processes, according to research published in the New England Journal of Medicine.
After a brief overview of disease classification and prognostic factors for AML, the authors, led by Dr. Hartmut Döhner of University Hospital Ulm in Germany, described a number of new treatment strategies. These approaches include directly addressing mutant proteins by targeting mutation-specific dependencies, developing inhibitors that could cut down on the occurrence of mutations in receptor tyrosine kinase genes, and developing new epigenetic therapies based on targeting specific mutant metabolic enzymes such as IDH1 and IDH2.
Other strategies, such as antibody therapy that focuses on the development of new monoclonal antibodies targeting CD33, and new formulations of older cytotoxic agents, also are being developed or are in clinical trial stages; however, many of these developments have not yet been translated into clinical practice, the researchers noted.
“New compounds hold promise to improve treatment outcomes; however, it is unlikely that any of these compounds, when used as single agents, will cure the disease. A major challenge will be to identify predictors for a response to specific agents, which will allow for the rational design of combinatorial therapies,” they wrote.
Dr. Döhner and a coauthor disclosed ties with several pharmaceutical companies.
Read the article here.
An understanding of the various molecular differences between acute myeloid leukemia (AML) particles is guiding the search for new drug combinations targeting a variety of AML cellular processes, according to research published in the New England Journal of Medicine.
After a brief overview of disease classification and prognostic factors for AML, the authors, led by Dr. Hartmut Döhner of University Hospital Ulm in Germany, described a number of new treatment strategies. These approaches include directly addressing mutant proteins by targeting mutation-specific dependencies, developing inhibitors that could cut down on the occurrence of mutations in receptor tyrosine kinase genes, and developing new epigenetic therapies based on targeting specific mutant metabolic enzymes such as IDH1 and IDH2.
Other strategies, such as antibody therapy that focuses on the development of new monoclonal antibodies targeting CD33, and new formulations of older cytotoxic agents, also are being developed or are in clinical trial stages; however, many of these developments have not yet been translated into clinical practice, the researchers noted.
“New compounds hold promise to improve treatment outcomes; however, it is unlikely that any of these compounds, when used as single agents, will cure the disease. A major challenge will be to identify predictors for a response to specific agents, which will allow for the rational design of combinatorial therapies,” they wrote.
Dr. Döhner and a coauthor disclosed ties with several pharmaceutical companies.
Read the article here.
FROM THE NEW ENGLAND JOURNAL OF MEDICINE
Traffic-related pollution linked to AML, not ALL, in kids

A French study has revealed an increased incidence of acute myeloid leukemia (AML) among children living close to heavily used roads.
The incidence of AML was 30% higher among children who lived within 150 m of heavily used roads and where the combined length of road sections
within this radius exceeded 260 m.
The researchers believe the association between AML and road proximity may be driven by traffic-related benzene exposure.
Previous research has shown an increased risk of leukemia among adults with a history of occupational exposure to benzene.
The current study did not suggest an increased risk of acute lymphoblastic leukemia (ALL) among children living closed to heavily used roads.
Jacqueline Clavel, MD, PhD, of INSERM in Paris, France, and her colleagues reported these findings in the American Journal of Epidemiology.
The team analyzed 2760 cases of leukemia diagnosed in children younger than 15 years of age in metropolitan France between 2002 and 2007, including 418 cases of AML and 2275 cases of ALL.
The researchers compared these cases to a contemporary sample of 30,000 control children representative of the metropolitan population.
The data showed that neither distance from the nearest major road(s) nor the length of major roads within 150 m of a child’s residence was associated with ALL.
However, there was an association for AML. For children whose home was less than 150 m from the nearest major road(s), the odds ratio (OR) was 1.2.
When the total length of major road(s) within 150 m from the child’s residence was 257-308 m (second tertile) or 309 m or greater (third tertile), the OR was 1.3. When the total length of major roads was 1-256 m (first tertile), the OR was 0.90.
The researchers noted that traffic-related nitrogen dioxide concentration was not associated with ALL or AML. But their data indicated that benzene concentration was associated with AML.
To assess this potential association, the team studied the Île-de-France region of Paris, the most urbanized region, for which the mean annual concentration of benzene, mainly from road traffic, was estimated in the vicinity of each residence.
The median estimated benzene concentration for controls living in the Île-de-France region was 1.3 μg/m3 (range, 0.3 to 8.5 μg/m3). And the length of major roads within 150 m of a child’s residence was positively and significantly correlated with log benzene concentration (r=0.3, P<0.001).
So it followed that exposure to an estimated benzene concentration greater than the median was associated with AML (OR=1.6).
The researchers also used a composite variable based on the estimated benzene concentration and the length of major roads around a child’s residence.
The association with AML was largest among children with at least 309 m of major roads within 150 m of their residence and estimated benzene concentrations of 1.3 μg/m3 or greater (OR=2.2).
The researchers said these results support a role for traffic-related benzene exposure in the etiology of childhood AML.

A French study has revealed an increased incidence of acute myeloid leukemia (AML) among children living close to heavily used roads.
The incidence of AML was 30% higher among children who lived within 150 m of heavily used roads and where the combined length of road sections
within this radius exceeded 260 m.
The researchers believe the association between AML and road proximity may be driven by traffic-related benzene exposure.
Previous research has shown an increased risk of leukemia among adults with a history of occupational exposure to benzene.
The current study did not suggest an increased risk of acute lymphoblastic leukemia (ALL) among children living closed to heavily used roads.
Jacqueline Clavel, MD, PhD, of INSERM in Paris, France, and her colleagues reported these findings in the American Journal of Epidemiology.
The team analyzed 2760 cases of leukemia diagnosed in children younger than 15 years of age in metropolitan France between 2002 and 2007, including 418 cases of AML and 2275 cases of ALL.
The researchers compared these cases to a contemporary sample of 30,000 control children representative of the metropolitan population.
The data showed that neither distance from the nearest major road(s) nor the length of major roads within 150 m of a child’s residence was associated with ALL.
However, there was an association for AML. For children whose home was less than 150 m from the nearest major road(s), the odds ratio (OR) was 1.2.
When the total length of major road(s) within 150 m from the child’s residence was 257-308 m (second tertile) or 309 m or greater (third tertile), the OR was 1.3. When the total length of major roads was 1-256 m (first tertile), the OR was 0.90.
The researchers noted that traffic-related nitrogen dioxide concentration was not associated with ALL or AML. But their data indicated that benzene concentration was associated with AML.
To assess this potential association, the team studied the Île-de-France region of Paris, the most urbanized region, for which the mean annual concentration of benzene, mainly from road traffic, was estimated in the vicinity of each residence.
The median estimated benzene concentration for controls living in the Île-de-France region was 1.3 μg/m3 (range, 0.3 to 8.5 μg/m3). And the length of major roads within 150 m of a child’s residence was positively and significantly correlated with log benzene concentration (r=0.3, P<0.001).
So it followed that exposure to an estimated benzene concentration greater than the median was associated with AML (OR=1.6).
The researchers also used a composite variable based on the estimated benzene concentration and the length of major roads around a child’s residence.
The association with AML was largest among children with at least 309 m of major roads within 150 m of their residence and estimated benzene concentrations of 1.3 μg/m3 or greater (OR=2.2).
The researchers said these results support a role for traffic-related benzene exposure in the etiology of childhood AML.

A French study has revealed an increased incidence of acute myeloid leukemia (AML) among children living close to heavily used roads.
The incidence of AML was 30% higher among children who lived within 150 m of heavily used roads and where the combined length of road sections
within this radius exceeded 260 m.
The researchers believe the association between AML and road proximity may be driven by traffic-related benzene exposure.
Previous research has shown an increased risk of leukemia among adults with a history of occupational exposure to benzene.
The current study did not suggest an increased risk of acute lymphoblastic leukemia (ALL) among children living closed to heavily used roads.
Jacqueline Clavel, MD, PhD, of INSERM in Paris, France, and her colleagues reported these findings in the American Journal of Epidemiology.
The team analyzed 2760 cases of leukemia diagnosed in children younger than 15 years of age in metropolitan France between 2002 and 2007, including 418 cases of AML and 2275 cases of ALL.
The researchers compared these cases to a contemporary sample of 30,000 control children representative of the metropolitan population.
The data showed that neither distance from the nearest major road(s) nor the length of major roads within 150 m of a child’s residence was associated with ALL.
However, there was an association for AML. For children whose home was less than 150 m from the nearest major road(s), the odds ratio (OR) was 1.2.
When the total length of major road(s) within 150 m from the child’s residence was 257-308 m (second tertile) or 309 m or greater (third tertile), the OR was 1.3. When the total length of major roads was 1-256 m (first tertile), the OR was 0.90.
The researchers noted that traffic-related nitrogen dioxide concentration was not associated with ALL or AML. But their data indicated that benzene concentration was associated with AML.
To assess this potential association, the team studied the Île-de-France region of Paris, the most urbanized region, for which the mean annual concentration of benzene, mainly from road traffic, was estimated in the vicinity of each residence.
The median estimated benzene concentration for controls living in the Île-de-France region was 1.3 μg/m3 (range, 0.3 to 8.5 μg/m3). And the length of major roads within 150 m of a child’s residence was positively and significantly correlated with log benzene concentration (r=0.3, P<0.001).
So it followed that exposure to an estimated benzene concentration greater than the median was associated with AML (OR=1.6).
The researchers also used a composite variable based on the estimated benzene concentration and the length of major roads around a child’s residence.
The association with AML was largest among children with at least 309 m of major roads within 150 m of their residence and estimated benzene concentrations of 1.3 μg/m3 or greater (OR=2.2).
The researchers said these results support a role for traffic-related benzene exposure in the etiology of childhood AML.
Gene linked to aggressive AML

Photo courtesy of NIH
The gene FOXC1 is associated with aggressive acute myeloid leukemia (AML), according to research published in Cancer Cell.
Researchers said tissue-inappropriate derepression of FOXC1 has functional consequences and prognostic significance in AML.
They found evidence suggesting that FOXC1 enhances clonogenic potential, helps block monocyte/macrophage differentiation, accelerates leukemia onset in mice, and leads to inferior survival in AML patients.
“This is an important finding which helps us understand how acute myeloid leukemia develops and why some cases of AML are more aggressive than others,” said study author Tim Somervaille, MBBS, PhD, of The University of Manchester in the UK.
“Here, instead of being faulty or mutated, this normal gene is turned on in the wrong place at the wrong time, which makes the cancer grow more rapidly. There are certain situations where this gene is necessary, as in the development of the eye and skeleton before birth, but when it’s switched on in the wrong tissue, it causes more aggressive forms of leukemia.”
Dr Somervaille and his colleagues said FOXC1 is expressed in at least 20% of human AML cases but not in normal hematopoietic populations.
The researchers analyzed levels of transcription factor genes in data from published studies to identify transcription regulators expressed in human AML hematopoietic stem and progenitor cells (HSPCs) but not normal HSPCs. In these studies, FOXC1 was among the genes that were most highly upregulated in AML HSPCs.
Further investigation revealed that FOXC1 expression is associated with mutations in NPM1 and t(6;9) but no other recurring mutations in AML.
When Dr Somervaille and his colleagues conducted experiments with human AML cells, they found that FOXC1 “contributes to oncogenic potential by maintaining differentiation block and clonogenic activity.”
In vitro experiments with normal HSPCs showed that FOXC1 expression temporarily impairs myeloid differentiation. In mice, expression of FOXC1 in normal HSPCs reduced donor:recipient chimerism in the blood and skewed differentiation toward the myeloid lineage and away from the B-cell lineage.
By comparing samples from AML patients, the researchers found that FOXC1 expression is associated with high HOX gene expression.
Subsequent experiments showed that FOXC1 collaborates with HOXA9 to enhance clonogenic potential and cell-cycle progression, help block monocyte/macrophage and B-lineage differentiation, and accelerate the onset of symptomatic leukemia in mice.
To determine if the same effects occur in humans, the researchers again analyzed data from AML patients. The results indicated that FOXC1 expression helps block monocyte/macrophage differentiation and leads to inferior survival.
Dr Somervaille and his colleagues said these findings may have therapeutic implications, as previous research has shown that, in basal-like breast cancer, high FOXC1 expression renders cells more susceptible to pharmacological inhibition of NF-kB. But additional research is needed to investigate therapeutic implications for AML.

Photo courtesy of NIH
The gene FOXC1 is associated with aggressive acute myeloid leukemia (AML), according to research published in Cancer Cell.
Researchers said tissue-inappropriate derepression of FOXC1 has functional consequences and prognostic significance in AML.
They found evidence suggesting that FOXC1 enhances clonogenic potential, helps block monocyte/macrophage differentiation, accelerates leukemia onset in mice, and leads to inferior survival in AML patients.
“This is an important finding which helps us understand how acute myeloid leukemia develops and why some cases of AML are more aggressive than others,” said study author Tim Somervaille, MBBS, PhD, of The University of Manchester in the UK.
“Here, instead of being faulty or mutated, this normal gene is turned on in the wrong place at the wrong time, which makes the cancer grow more rapidly. There are certain situations where this gene is necessary, as in the development of the eye and skeleton before birth, but when it’s switched on in the wrong tissue, it causes more aggressive forms of leukemia.”
Dr Somervaille and his colleagues said FOXC1 is expressed in at least 20% of human AML cases but not in normal hematopoietic populations.
The researchers analyzed levels of transcription factor genes in data from published studies to identify transcription regulators expressed in human AML hematopoietic stem and progenitor cells (HSPCs) but not normal HSPCs. In these studies, FOXC1 was among the genes that were most highly upregulated in AML HSPCs.
Further investigation revealed that FOXC1 expression is associated with mutations in NPM1 and t(6;9) but no other recurring mutations in AML.
When Dr Somervaille and his colleagues conducted experiments with human AML cells, they found that FOXC1 “contributes to oncogenic potential by maintaining differentiation block and clonogenic activity.”
In vitro experiments with normal HSPCs showed that FOXC1 expression temporarily impairs myeloid differentiation. In mice, expression of FOXC1 in normal HSPCs reduced donor:recipient chimerism in the blood and skewed differentiation toward the myeloid lineage and away from the B-cell lineage.
By comparing samples from AML patients, the researchers found that FOXC1 expression is associated with high HOX gene expression.
Subsequent experiments showed that FOXC1 collaborates with HOXA9 to enhance clonogenic potential and cell-cycle progression, help block monocyte/macrophage and B-lineage differentiation, and accelerate the onset of symptomatic leukemia in mice.
To determine if the same effects occur in humans, the researchers again analyzed data from AML patients. The results indicated that FOXC1 expression helps block monocyte/macrophage differentiation and leads to inferior survival.
Dr Somervaille and his colleagues said these findings may have therapeutic implications, as previous research has shown that, in basal-like breast cancer, high FOXC1 expression renders cells more susceptible to pharmacological inhibition of NF-kB. But additional research is needed to investigate therapeutic implications for AML.

Photo courtesy of NIH
The gene FOXC1 is associated with aggressive acute myeloid leukemia (AML), according to research published in Cancer Cell.
Researchers said tissue-inappropriate derepression of FOXC1 has functional consequences and prognostic significance in AML.
They found evidence suggesting that FOXC1 enhances clonogenic potential, helps block monocyte/macrophage differentiation, accelerates leukemia onset in mice, and leads to inferior survival in AML patients.
“This is an important finding which helps us understand how acute myeloid leukemia develops and why some cases of AML are more aggressive than others,” said study author Tim Somervaille, MBBS, PhD, of The University of Manchester in the UK.
“Here, instead of being faulty or mutated, this normal gene is turned on in the wrong place at the wrong time, which makes the cancer grow more rapidly. There are certain situations where this gene is necessary, as in the development of the eye and skeleton before birth, but when it’s switched on in the wrong tissue, it causes more aggressive forms of leukemia.”
Dr Somervaille and his colleagues said FOXC1 is expressed in at least 20% of human AML cases but not in normal hematopoietic populations.
The researchers analyzed levels of transcription factor genes in data from published studies to identify transcription regulators expressed in human AML hematopoietic stem and progenitor cells (HSPCs) but not normal HSPCs. In these studies, FOXC1 was among the genes that were most highly upregulated in AML HSPCs.
Further investigation revealed that FOXC1 expression is associated with mutations in NPM1 and t(6;9) but no other recurring mutations in AML.
When Dr Somervaille and his colleagues conducted experiments with human AML cells, they found that FOXC1 “contributes to oncogenic potential by maintaining differentiation block and clonogenic activity.”
In vitro experiments with normal HSPCs showed that FOXC1 expression temporarily impairs myeloid differentiation. In mice, expression of FOXC1 in normal HSPCs reduced donor:recipient chimerism in the blood and skewed differentiation toward the myeloid lineage and away from the B-cell lineage.
By comparing samples from AML patients, the researchers found that FOXC1 expression is associated with high HOX gene expression.
Subsequent experiments showed that FOXC1 collaborates with HOXA9 to enhance clonogenic potential and cell-cycle progression, help block monocyte/macrophage and B-lineage differentiation, and accelerate the onset of symptomatic leukemia in mice.
To determine if the same effects occur in humans, the researchers again analyzed data from AML patients. The results indicated that FOXC1 expression helps block monocyte/macrophage differentiation and leads to inferior survival.
Dr Somervaille and his colleagues said these findings may have therapeutic implications, as previous research has shown that, in basal-like breast cancer, high FOXC1 expression renders cells more susceptible to pharmacological inhibition of NF-kB. But additional research is needed to investigate therapeutic implications for AML.