User login
Increased Ordering of Diagnostic Tests Associated with Longer Lengths of Stay in Pediatric Pneumonia
Clinical question: What is the relationship between variation in resource utilization and outcomes in children with community-acquired pneumonia (CAP)?
Background: Variation in clinical care, particularly when driven by provider preferences, often highlights opportunities for improvement in the quality of our care. CAP is one of the most common reasons for hospitalization in children. The relationship between variation in care processes, utilization, and outcomes in pediatric CAP is not well defined.
Study design: Retrospective database review.
Setting: Twenty-nine freestanding children's hospitals.
Synopsis: The Pediatric Health Information System (PHIS) database was used to review utilization and outcomes data on 43,819 children admitted with nonsevere CAP during a five-year period. Substantial degrees of variation in test ordering, empiric antibiotic selection, length of stay (LOS), and 14-day readmissions were found. An association was noted between increased resource utilization—specifically, ordering of diagnostic tests—and longer LOS.
The association between increased resource utilization and LOS has been suggested in other work in respiratory illness in children. Although the retrospective nature of this work precludes detailed resolution of whether confounding by severity was an issue, this appears unlikely based on the relatively homogeneous patient populations and hospital types. Additional limitations of this work exist, and include an inability to further assess the appropriateness of the testing that was ordered—as well as relatively crude rankings of hospitals based on resource utilization. Nevertheless, in an era where a premium is placed on finding value in clinical medicine, these results should prompt further exploration of the link between testing and LOS in children hospitalized with CAP.
Bottom line: Unnecessary testing in children hospitalized with pneumonia may lead to longer LOS.
Citation: Brogan TV, Hall M, Williams DJ, et al. Variability in processes of care and outcomes among children hospitalized with community-acquired pneumonia. Ped Infect Dis J. 2012;31:1036-1041.
Reviewed by Pediatric Editor Mark Shen, MD, SFHM, medical director of hospital medicine at Dell Children's Medical Center, Austin, Texas.
Clinical question: What is the relationship between variation in resource utilization and outcomes in children with community-acquired pneumonia (CAP)?
Background: Variation in clinical care, particularly when driven by provider preferences, often highlights opportunities for improvement in the quality of our care. CAP is one of the most common reasons for hospitalization in children. The relationship between variation in care processes, utilization, and outcomes in pediatric CAP is not well defined.
Study design: Retrospective database review.
Setting: Twenty-nine freestanding children's hospitals.
Synopsis: The Pediatric Health Information System (PHIS) database was used to review utilization and outcomes data on 43,819 children admitted with nonsevere CAP during a five-year period. Substantial degrees of variation in test ordering, empiric antibiotic selection, length of stay (LOS), and 14-day readmissions were found. An association was noted between increased resource utilization—specifically, ordering of diagnostic tests—and longer LOS.
The association between increased resource utilization and LOS has been suggested in other work in respiratory illness in children. Although the retrospective nature of this work precludes detailed resolution of whether confounding by severity was an issue, this appears unlikely based on the relatively homogeneous patient populations and hospital types. Additional limitations of this work exist, and include an inability to further assess the appropriateness of the testing that was ordered—as well as relatively crude rankings of hospitals based on resource utilization. Nevertheless, in an era where a premium is placed on finding value in clinical medicine, these results should prompt further exploration of the link between testing and LOS in children hospitalized with CAP.
Bottom line: Unnecessary testing in children hospitalized with pneumonia may lead to longer LOS.
Citation: Brogan TV, Hall M, Williams DJ, et al. Variability in processes of care and outcomes among children hospitalized with community-acquired pneumonia. Ped Infect Dis J. 2012;31:1036-1041.
Reviewed by Pediatric Editor Mark Shen, MD, SFHM, medical director of hospital medicine at Dell Children's Medical Center, Austin, Texas.
Clinical question: What is the relationship between variation in resource utilization and outcomes in children with community-acquired pneumonia (CAP)?
Background: Variation in clinical care, particularly when driven by provider preferences, often highlights opportunities for improvement in the quality of our care. CAP is one of the most common reasons for hospitalization in children. The relationship between variation in care processes, utilization, and outcomes in pediatric CAP is not well defined.
Study design: Retrospective database review.
Setting: Twenty-nine freestanding children's hospitals.
Synopsis: The Pediatric Health Information System (PHIS) database was used to review utilization and outcomes data on 43,819 children admitted with nonsevere CAP during a five-year period. Substantial degrees of variation in test ordering, empiric antibiotic selection, length of stay (LOS), and 14-day readmissions were found. An association was noted between increased resource utilization—specifically, ordering of diagnostic tests—and longer LOS.
The association between increased resource utilization and LOS has been suggested in other work in respiratory illness in children. Although the retrospective nature of this work precludes detailed resolution of whether confounding by severity was an issue, this appears unlikely based on the relatively homogeneous patient populations and hospital types. Additional limitations of this work exist, and include an inability to further assess the appropriateness of the testing that was ordered—as well as relatively crude rankings of hospitals based on resource utilization. Nevertheless, in an era where a premium is placed on finding value in clinical medicine, these results should prompt further exploration of the link between testing and LOS in children hospitalized with CAP.
Bottom line: Unnecessary testing in children hospitalized with pneumonia may lead to longer LOS.
Citation: Brogan TV, Hall M, Williams DJ, et al. Variability in processes of care and outcomes among children hospitalized with community-acquired pneumonia. Ped Infect Dis J. 2012;31:1036-1041.
Reviewed by Pediatric Editor Mark Shen, MD, SFHM, medical director of hospital medicine at Dell Children's Medical Center, Austin, Texas.
What Is the Best Treatment for an Adult Patient with Staphylococcus aureus Bacteremia?
Case
An 82-year-old man with non-Hodgkin’s lymphoma in remission and a history of congestive heart failure and hypertension presents with one week of generalized malaise and intermittent fevers. Vitals show a temperature of 101oF, blood pressure of 130/60 mmHg, and heart rate of 100. His exam is notable for an erythematous and tender chest port site, with no murmurs. Blood cultures drawn upon presentation show gram-positive cocci speciated to Staphylococcus aureus. What are the next steps in management of this patient?
Overview
S. aureus bacteremia (SAB) is a common infectious cause of morbidity and mortality worldwide, causing both community-acquired and hospital-acquired bacteremia. In the U.S. alone, it accounts for 23% of all bloodstream infections and is the bacterial pathogen most strongly associated with death.1 Mortality rates are approximately 42% in those with methicillin-resistant S. aureus (MRSA) bacteremia and 28% in those with methicillin-sensitive S. aureus (MSSA) bacteremia.2
Recognizing the severity of SAB, the Infectious Disease Society of America (IDSA) published treatment guidelines in 2011 to help direct the clinical care of this disease process.3 However, the majority of the recommendations are based on observational studies and expert opinion, as less than 1,500 patients have been enrolled in randomized controlled trials specifically targeted to investigate the treatment of SAB.4
Review of the Data
A clinically significant SAB usually is defined as the isolation of S. aureus from a venous blood culture with associated symptoms and signs of systemic infection.5 As SAB contamination is rare and can be associated with multiple complications, including metastatic infections, embolic stroke, recurrent infection, and death, any finding of a positive blood culture must be taken seriously.4
SAB treatment is multifaceted and should focus on the removal of any nidus of infection, such as a catheter or a prosthetic device, the use of prolonged antimicrobial therapy, and the evaluation of potential complications. In a retrospective study, Johnson et al showed that failure to remove the source is one of the strongest independent predictors of relapse in patients with SAB.6 However, 10% to 40% of patients have no identifiable focus, which increases the impetus to evaluate for complications.7-8 Overall, approximately one-third of patients with SAB develop metastatic complications, either from hematogenous seeding or local extension of infection.9
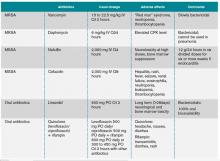
In addition to advanced age and such comorbid conditions as cirrhosis, the strongest predictor of complications is a positive blood culture at 48 to 96 hours after an initial positive blood culture, as shown in a large prospective cohort study by Fowler et al.7,10-11 Additional independent risk factors (see Table 1) include community acquisition (likely due to prolonged duration of bacteremia), skin examination suggesting the presence of acute systemic infection, and persistent fever at 72 hours after the first positive blood culture. Patients with even one of these risk factors are at high risk for a complicated course (which occurs in about 35%). In a case-control study, Chihara et al showed that S. aureus bacteruria in the absence of urinary tract pathology or recent urinary tract instrumentation might be associated with threefold increased mortality compared with those without bacteriuria, even after adjustment for comorbid conditions.12
Antimicrobial Treatment
The initial choice of antibiotic therapy for SAB must take into account the MRSA prevalence in the community and hospital. If suspicion is high enough for MRSA, the IDSA’s 2011 guidelines suggest treatment with vancomycin or daptomycin.3 Although there are no published RCTs to support a particular antibiotic regimen, there are trials to suggest that a delay in treatment could be harmful. One study, by Lordis et al, showed that a delay in treatment, as defined by treatment after 44.75 hours, was associated with a longer hospital stay, with the delayed treatment group being hospitalized for 20.2 days and the early treatment group being hospitalized for 14.3 days.13 A delay in treatment was also found to be an independent predictor of mortality.13
Once susceptibilities are known, it is important to appropriately tailor antibiotics, as studies have shown lower treatment failure rates with the use of beta-lactam antibiotics when compared with empiric MRSA coverage.14-15 In one prospective study of 123 hemodialysis patients with MSSA bacteremia, Stryjewski et al showed that those treated with vancomycin were at higher risk of experiencing treatment failure than those treated with cefazolin.15 In another prospective observational study of 505 patients with SAB, Chang et al found that treatment with nafcillin was superior to vancomycin in preventing persistent bacteremia or relapse for MSSA bacteremia.14 These studies highlight the benefits of adjusting the empirically selected antibiotics, as narrowing the spectrum can result in less treatment failure.
If susceptibilities confirm MRSA, the IDSA recommends continued treatment with vancomycin or daptomycin.3 Although vancomycin is most commonly used, partly because of low cost and familiarity, Fowler et al published a study of 246 patients with SAB with or without endocarditis, assigning them to treatment with daptomycin, initial low-dose gentamicin plus vancomycin or an antistaphylococcal penicillin.16 The study found that daptomycin was not inferior to the other therapies, confirming that daptomycin is a reasonable choice in the treatment of MRSA infections.
Oral antibiotics are an option to treat SAB when necessary. A RCT by Heldman et al of 85 intravenous drug users with SAB (and suspected right-sided endocarditis, 65% of which had HIV) showed similar efficacy of ciprofloxacin plus rifampin versus standard intravenous therapy.17 A subsequent randomized trial of 104 patients with SAB comparing oral fleroxacin plus rifampin against conventional intravenous therapy also showed similar cure rates, with the added benefit of earlier discharge.18 Furthermore, in a meta-analysis of five randomized studies by Shorr et al (see Table 2), linezolid was found to have outcomes that were not inferior to vancomycin (clinical cure/microbiological success of 56%/69% in the linezolid group and 46%/73% in the vancomycin group).19
Treatment Duration
Recommendations for the duration of antibiotic treatment for SAB are mainly based on observational studies, which show mixed results. In one study done in the 1950s, about two-thirds of cases of SAB were associated with endocarditis, and longer courses of intravenous therapy (greater than four weeks) were recommended.20
More recently, with the increasing rates of catheter-related SAB and its relatively high rate of expeditious blood culture clearance, a shorter duration has been evaluated in several studies. In 1992, an analysis of published data and a retrospective case series concluded that fewer than 10 days of intravenous antibiotics might be associated with an increased risk of recurrence, but 10 to 14 days of intravenous therapy was effective for most cases of catheter-associated SAB.5 In another prospective study, Fowler et al found that a seven-day course of intravenous antibiotic therapy may be sufficient for simple, catheter-related infections.21 A subsequent prospective study by Jensen et al reported that a course of antibiotic therapy of less than 14 days might be associated with higher mortality compared to a longer course.9 A prospective study of 276 patients by Thomas et al found there was no relationship between relapse and duration of treatment (seven to 15 days) in catheter-related SAB, concluding that more than 14 days of antibiotic therapy was unnecessary.22
Per IDSA guidelines, uncomplicated SAB (no implanted prosthesis, negative blood cultures within two to four days, defervescence within 72 hours of initiating therapy, and lack of metastatic complication) can be treated with a two-week course of antibiotics, while complicated bacteremia (any of above criteria) should be treated within four to six weeks.3
Monitoring for Complications: Echos
Based on the IDSA guidelines, echocardiography is recommended in all patients with bacteremia, with a preference of transesophageal echocardiography (TEE) over transthoracic echocardiography (TTE).3 More recently, Kaasch et al developed simple criteria to identify patients with nosocomial SAB at low risk for infective endocarditis based on two prospective cohort studies.23 Lack of any of these criteria, which include prolonged bacteremia of more than four days’ duration, presence of a permanent intracardiac device, hemodialysis dependency, spinal infection and nonvertebral osteomyelitis, along with a negative TTE indicates that a TEE is not necessary (see Table 3). However, these patients need close follow-up to ensure that bacteremia clears and no new signs or symptoms concerning for metastatic infection develop.
ID Consultation
Several studies have shown that ID consultation not only improves adherence to evidence-based management of SAB, but it also reduces mortality.24-27 In a recent prospective cohort study in a tertiary-care center, even after adjusting for pre-existing comorbidities and severity of disease, an ID consult was associated with a 56% reduction in 28-day mortality.24 The patients who were followed by an ID consult service were more likely to receive appropriate duration of antibiotics (81% vs. 29%, respectively) and undergo appropriate workup for the evaluation of metastatic infections (34% and 8%, respectively). This study concluded that routine ID consult should be considered in patients with SAB, especially those with severe illness and multiple comorbid conditions.
Back to the Case
The patient was started on empiric therapy with vancomycin and serial blood cultures were obtained. He remained hemodynamically stable but febrile, with persistently positive blood and urine cultures. Given concern for the port being the source of his infection, his chest port was removed. A high-quality TTE was performed and was unremarkable.
ID was consulted. Blood cultures subsequently grew MSSA and vancomycin was switched to cefazolin 2g every eight hours. On hospital Day 5, his fever resolved and blood cultures turned negative. There were no clinical signs or symptoms for metastatic infections. A PICC line was placed after blood cultures remained negative for 48 hours. The decision was made to treat him with four weeks of antibiotics from his last positive blood culture, with follow-up in ID clinic.
Bottom Line
SAB is a common worldwide cause of morbidity and mortality. Treatment should include removing the nidus if present, finding and administering the appropriate antimicrobial therapy, evaluating for possible complications, and consulting with ID.
Dr. Ward is an assistant professor, Dr. Kim a clinical instructor, and Dr. Stojan a clinical lecturer at the University of Michigan Health System in Ann Arbor.
Acknowledgement
The authors would like to thank Dr. Jeffrey Rohde for reviewing the manuscript.
References
- Shorr AF, Tabak YP, Killian AD, et al. Healthcare-associated bloodstream infection: a distinct entity? Insights from a large U.S. database. Crit Care Med. 2006;34:3588-3595.
- Mylotte JM, McDermott C, Spooner JA. Prospective study of 114 consecutive episodes of Staphylococcus aureus bacteremia. Rev Infect Dis. 1987;9:891-907.
- Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3).
- Naber CK, Baddour LM, Giamarellos-Bourboulis EJ, et al. Clinical consensus conference: survey on gram-positive bloodstream infections with a focus on Staphylococcus aureus. Clin Infect Dis. 2011;52(3):285-292.
- Thwaites GE, Edgeworth JD, Gkrania-Klotsas E, et al. Clinical management of Staphylococcus bacteremia. Lancet Infect Dis. 2011;11(3):208-222.
- Johnson LB, Almoujahed MO, Ilg K, et al. Staphylococcus aureus bacteremia: compliance with standard treatment, long-term outcome and predictors of relapse. Scand J Infec Dis. 2003;35(11-12):782-789.
- Fowler VG Jr., Olsen MK, Corey GR, et al. Clinical identifiers of complicated Staphylococcus aureus bacteremia. Arch Intern Med. 2003;163:2066-2072.
- Mylotte JM, Tayara A. Staphylococcus aureus bacteremia: predictors of 30-day mortality in a large cohort. Clin Infect Dis. 2000;31:1170-1174.
- Jensen AG, Wachmann CH, Espersen F, et al. Treatment and outcome of Staphylococcus aureus bacteremia: a prospective study of 278 cases. Arch Intern Med. 2002;162(1):25-32.
- Malani PN, Rana MM, Banerjee M, et al. Staphylococcus aureus bloodstream infections: the association between age and mortality and functional status. J Am Geriatr Soc. 2008;56(8):1485-1489.
- Kim SH, Park WB, Lee KD, et al. Outcome of Staphylococcus aureus bacteremia in patients with eradicable foci versus noneradicable foci. Clin Infect Dis. 2003;37(6):794-799.
- Chihara S. Popovich KJ, Weinstein RA, et al. Staphylococcus aureus bacteriuria as a prognosticator for outcome of Staphylococcus aureus bacteremia: a case control study. BMC Inf Dis. 2010;10:225.
- Lodise TP, McKinnon PS, Swiderski L, Rybak MJ. Outcomes analysis of delayed antibiotic treatment for hospital-acquired Staphylococcus aureus bacteremia. Clin Infect Dis. 2003;36:1418-1423.
- Chang FY, Peacock JE Jr., Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore). 2003;82:333.
- Stryjewski ME, Szczech LA, Benjamin DK Jr., et al. Use of vancomycin or first-generation cephalosporins for the treatment of hemodialysisdependent patients with methicillin-susceptible Staphylococcus aureus bacteremia. Clin Infect Dis. 2007;44:190-196.
- Fowler VG Jr., Boucher HW, Corey GR, et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med. 2006;355:653.
- Heldman AW, Hartert TV, Ray SC, et al. Oral antibiotic treatment of right-sided staphylococcal endocarditis in injection drug users: prospective randomized comparison with parenteral therapy. Am J Med. 1996;101:68-76.
- Schrenzel J, Harbarth S, Schockmel G, et al. A randomized clinical trial to compare fleroxacin-rifampicin with flucloxacillin or vancomycin for the treatment of Staphylococcal infection. Clin Infect Dis. 2004;39:1285-1292.
- Shorr AF, Kunkel MJ, Kollef M. Linezolid versus vancomycin for Staphylococcus aureus bacteraemia: pooled analysis of randomized studies. J Antimicrob Chemother. 2005;56:923-929.
- Wilson R, Hamburger M. Fifteen years’ experience with Staphylococcus septicemia in a large city hospital; analysis of fifty-five cases in the Cincinnati General Hospital 1940 to 1954. Am J Med. 1957;22:437-457.
- Fowler VG Jr., Sanders LL, Sexton DJ, et al. Outcome of Staphylococcus aureus bacteremia according to compliance with recommendation of infectious disease specialists: experience with 244 patients. Clin Infect Dis. 1998;27:478-486.
- Thomas MG, Morris AJ. Cannula-associated Staphylococcus aureus bacteremia: outcome in relation to treatment. Intern Med J. 2005;35:319-330.
- Kaasch AJ, Fowler VG Jr, Rieg S, et al. Use of a simple criteria set for guiding echocardiography in nosocomidal Staphylococcus aureus bacteremia. Clin Infect Dis. 2011;53:1-9.
- Honda H, Krauss MJ, Jones JC, et al. The value of infectious disease consultation in Staphylococcus aureus bacteremia. Am J Med. 2010;123:631-637.
- Nagao M, Iinuma Y, Saito T, et al. Close cooperation between infectious disease physicians and attending physicians can result in better management and outcome for patients with Staphylococcus aureus bacteremia. Euro Soc Clin Microbiology Infect Dis. 2010;16:1783-1788.
- Fowler VG Jr., Sanders LL, Sexton DJ, et al. Outcome of Staphylococcus aureus bacteremia according to compliance with recommendations of infectious diseases specialist: experience with 244 patients. Clin Infect Dis. 1998;27(3):478-486.
- Jenkins TC, Price CS, Sabel AL et al. Impact of routine infectious diseases service consultation on the evaluation, management, and outcome of Staphylococcus aureus bacteremia. Clin Infect Dis. 2008;46(7):1000-1008.
Case
An 82-year-old man with non-Hodgkin’s lymphoma in remission and a history of congestive heart failure and hypertension presents with one week of generalized malaise and intermittent fevers. Vitals show a temperature of 101oF, blood pressure of 130/60 mmHg, and heart rate of 100. His exam is notable for an erythematous and tender chest port site, with no murmurs. Blood cultures drawn upon presentation show gram-positive cocci speciated to Staphylococcus aureus. What are the next steps in management of this patient?
Overview
S. aureus bacteremia (SAB) is a common infectious cause of morbidity and mortality worldwide, causing both community-acquired and hospital-acquired bacteremia. In the U.S. alone, it accounts for 23% of all bloodstream infections and is the bacterial pathogen most strongly associated with death.1 Mortality rates are approximately 42% in those with methicillin-resistant S. aureus (MRSA) bacteremia and 28% in those with methicillin-sensitive S. aureus (MSSA) bacteremia.2
Recognizing the severity of SAB, the Infectious Disease Society of America (IDSA) published treatment guidelines in 2011 to help direct the clinical care of this disease process.3 However, the majority of the recommendations are based on observational studies and expert opinion, as less than 1,500 patients have been enrolled in randomized controlled trials specifically targeted to investigate the treatment of SAB.4
Review of the Data
A clinically significant SAB usually is defined as the isolation of S. aureus from a venous blood culture with associated symptoms and signs of systemic infection.5 As SAB contamination is rare and can be associated with multiple complications, including metastatic infections, embolic stroke, recurrent infection, and death, any finding of a positive blood culture must be taken seriously.4
SAB treatment is multifaceted and should focus on the removal of any nidus of infection, such as a catheter or a prosthetic device, the use of prolonged antimicrobial therapy, and the evaluation of potential complications. In a retrospective study, Johnson et al showed that failure to remove the source is one of the strongest independent predictors of relapse in patients with SAB.6 However, 10% to 40% of patients have no identifiable focus, which increases the impetus to evaluate for complications.7-8 Overall, approximately one-third of patients with SAB develop metastatic complications, either from hematogenous seeding or local extension of infection.9
In addition to advanced age and such comorbid conditions as cirrhosis, the strongest predictor of complications is a positive blood culture at 48 to 96 hours after an initial positive blood culture, as shown in a large prospective cohort study by Fowler et al.7,10-11 Additional independent risk factors (see Table 1) include community acquisition (likely due to prolonged duration of bacteremia), skin examination suggesting the presence of acute systemic infection, and persistent fever at 72 hours after the first positive blood culture. Patients with even one of these risk factors are at high risk for a complicated course (which occurs in about 35%). In a case-control study, Chihara et al showed that S. aureus bacteruria in the absence of urinary tract pathology or recent urinary tract instrumentation might be associated with threefold increased mortality compared with those without bacteriuria, even after adjustment for comorbid conditions.12
Antimicrobial Treatment
The initial choice of antibiotic therapy for SAB must take into account the MRSA prevalence in the community and hospital. If suspicion is high enough for MRSA, the IDSA’s 2011 guidelines suggest treatment with vancomycin or daptomycin.3 Although there are no published RCTs to support a particular antibiotic regimen, there are trials to suggest that a delay in treatment could be harmful. One study, by Lordis et al, showed that a delay in treatment, as defined by treatment after 44.75 hours, was associated with a longer hospital stay, with the delayed treatment group being hospitalized for 20.2 days and the early treatment group being hospitalized for 14.3 days.13 A delay in treatment was also found to be an independent predictor of mortality.13
Once susceptibilities are known, it is important to appropriately tailor antibiotics, as studies have shown lower treatment failure rates with the use of beta-lactam antibiotics when compared with empiric MRSA coverage.14-15 In one prospective study of 123 hemodialysis patients with MSSA bacteremia, Stryjewski et al showed that those treated with vancomycin were at higher risk of experiencing treatment failure than those treated with cefazolin.15 In another prospective observational study of 505 patients with SAB, Chang et al found that treatment with nafcillin was superior to vancomycin in preventing persistent bacteremia or relapse for MSSA bacteremia.14 These studies highlight the benefits of adjusting the empirically selected antibiotics, as narrowing the spectrum can result in less treatment failure.
If susceptibilities confirm MRSA, the IDSA recommends continued treatment with vancomycin or daptomycin.3 Although vancomycin is most commonly used, partly because of low cost and familiarity, Fowler et al published a study of 246 patients with SAB with or without endocarditis, assigning them to treatment with daptomycin, initial low-dose gentamicin plus vancomycin or an antistaphylococcal penicillin.16 The study found that daptomycin was not inferior to the other therapies, confirming that daptomycin is a reasonable choice in the treatment of MRSA infections.
Oral antibiotics are an option to treat SAB when necessary. A RCT by Heldman et al of 85 intravenous drug users with SAB (and suspected right-sided endocarditis, 65% of which had HIV) showed similar efficacy of ciprofloxacin plus rifampin versus standard intravenous therapy.17 A subsequent randomized trial of 104 patients with SAB comparing oral fleroxacin plus rifampin against conventional intravenous therapy also showed similar cure rates, with the added benefit of earlier discharge.18 Furthermore, in a meta-analysis of five randomized studies by Shorr et al (see Table 2), linezolid was found to have outcomes that were not inferior to vancomycin (clinical cure/microbiological success of 56%/69% in the linezolid group and 46%/73% in the vancomycin group).19
Treatment Duration
Recommendations for the duration of antibiotic treatment for SAB are mainly based on observational studies, which show mixed results. In one study done in the 1950s, about two-thirds of cases of SAB were associated with endocarditis, and longer courses of intravenous therapy (greater than four weeks) were recommended.20
More recently, with the increasing rates of catheter-related SAB and its relatively high rate of expeditious blood culture clearance, a shorter duration has been evaluated in several studies. In 1992, an analysis of published data and a retrospective case series concluded that fewer than 10 days of intravenous antibiotics might be associated with an increased risk of recurrence, but 10 to 14 days of intravenous therapy was effective for most cases of catheter-associated SAB.5 In another prospective study, Fowler et al found that a seven-day course of intravenous antibiotic therapy may be sufficient for simple, catheter-related infections.21 A subsequent prospective study by Jensen et al reported that a course of antibiotic therapy of less than 14 days might be associated with higher mortality compared to a longer course.9 A prospective study of 276 patients by Thomas et al found there was no relationship between relapse and duration of treatment (seven to 15 days) in catheter-related SAB, concluding that more than 14 days of antibiotic therapy was unnecessary.22
Per IDSA guidelines, uncomplicated SAB (no implanted prosthesis, negative blood cultures within two to four days, defervescence within 72 hours of initiating therapy, and lack of metastatic complication) can be treated with a two-week course of antibiotics, while complicated bacteremia (any of above criteria) should be treated within four to six weeks.3
Monitoring for Complications: Echos
Based on the IDSA guidelines, echocardiography is recommended in all patients with bacteremia, with a preference of transesophageal echocardiography (TEE) over transthoracic echocardiography (TTE).3 More recently, Kaasch et al developed simple criteria to identify patients with nosocomial SAB at low risk for infective endocarditis based on two prospective cohort studies.23 Lack of any of these criteria, which include prolonged bacteremia of more than four days’ duration, presence of a permanent intracardiac device, hemodialysis dependency, spinal infection and nonvertebral osteomyelitis, along with a negative TTE indicates that a TEE is not necessary (see Table 3). However, these patients need close follow-up to ensure that bacteremia clears and no new signs or symptoms concerning for metastatic infection develop.
ID Consultation
Several studies have shown that ID consultation not only improves adherence to evidence-based management of SAB, but it also reduces mortality.24-27 In a recent prospective cohort study in a tertiary-care center, even after adjusting for pre-existing comorbidities and severity of disease, an ID consult was associated with a 56% reduction in 28-day mortality.24 The patients who were followed by an ID consult service were more likely to receive appropriate duration of antibiotics (81% vs. 29%, respectively) and undergo appropriate workup for the evaluation of metastatic infections (34% and 8%, respectively). This study concluded that routine ID consult should be considered in patients with SAB, especially those with severe illness and multiple comorbid conditions.
Back to the Case
The patient was started on empiric therapy with vancomycin and serial blood cultures were obtained. He remained hemodynamically stable but febrile, with persistently positive blood and urine cultures. Given concern for the port being the source of his infection, his chest port was removed. A high-quality TTE was performed and was unremarkable.
ID was consulted. Blood cultures subsequently grew MSSA and vancomycin was switched to cefazolin 2g every eight hours. On hospital Day 5, his fever resolved and blood cultures turned negative. There were no clinical signs or symptoms for metastatic infections. A PICC line was placed after blood cultures remained negative for 48 hours. The decision was made to treat him with four weeks of antibiotics from his last positive blood culture, with follow-up in ID clinic.
Bottom Line
SAB is a common worldwide cause of morbidity and mortality. Treatment should include removing the nidus if present, finding and administering the appropriate antimicrobial therapy, evaluating for possible complications, and consulting with ID.
Dr. Ward is an assistant professor, Dr. Kim a clinical instructor, and Dr. Stojan a clinical lecturer at the University of Michigan Health System in Ann Arbor.
Acknowledgement
The authors would like to thank Dr. Jeffrey Rohde for reviewing the manuscript.
References
- Shorr AF, Tabak YP, Killian AD, et al. Healthcare-associated bloodstream infection: a distinct entity? Insights from a large U.S. database. Crit Care Med. 2006;34:3588-3595.
- Mylotte JM, McDermott C, Spooner JA. Prospective study of 114 consecutive episodes of Staphylococcus aureus bacteremia. Rev Infect Dis. 1987;9:891-907.
- Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3).
- Naber CK, Baddour LM, Giamarellos-Bourboulis EJ, et al. Clinical consensus conference: survey on gram-positive bloodstream infections with a focus on Staphylococcus aureus. Clin Infect Dis. 2011;52(3):285-292.
- Thwaites GE, Edgeworth JD, Gkrania-Klotsas E, et al. Clinical management of Staphylococcus bacteremia. Lancet Infect Dis. 2011;11(3):208-222.
- Johnson LB, Almoujahed MO, Ilg K, et al. Staphylococcus aureus bacteremia: compliance with standard treatment, long-term outcome and predictors of relapse. Scand J Infec Dis. 2003;35(11-12):782-789.
- Fowler VG Jr., Olsen MK, Corey GR, et al. Clinical identifiers of complicated Staphylococcus aureus bacteremia. Arch Intern Med. 2003;163:2066-2072.
- Mylotte JM, Tayara A. Staphylococcus aureus bacteremia: predictors of 30-day mortality in a large cohort. Clin Infect Dis. 2000;31:1170-1174.
- Jensen AG, Wachmann CH, Espersen F, et al. Treatment and outcome of Staphylococcus aureus bacteremia: a prospective study of 278 cases. Arch Intern Med. 2002;162(1):25-32.
- Malani PN, Rana MM, Banerjee M, et al. Staphylococcus aureus bloodstream infections: the association between age and mortality and functional status. J Am Geriatr Soc. 2008;56(8):1485-1489.
- Kim SH, Park WB, Lee KD, et al. Outcome of Staphylococcus aureus bacteremia in patients with eradicable foci versus noneradicable foci. Clin Infect Dis. 2003;37(6):794-799.
- Chihara S. Popovich KJ, Weinstein RA, et al. Staphylococcus aureus bacteriuria as a prognosticator for outcome of Staphylococcus aureus bacteremia: a case control study. BMC Inf Dis. 2010;10:225.
- Lodise TP, McKinnon PS, Swiderski L, Rybak MJ. Outcomes analysis of delayed antibiotic treatment for hospital-acquired Staphylococcus aureus bacteremia. Clin Infect Dis. 2003;36:1418-1423.
- Chang FY, Peacock JE Jr., Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore). 2003;82:333.
- Stryjewski ME, Szczech LA, Benjamin DK Jr., et al. Use of vancomycin or first-generation cephalosporins for the treatment of hemodialysisdependent patients with methicillin-susceptible Staphylococcus aureus bacteremia. Clin Infect Dis. 2007;44:190-196.
- Fowler VG Jr., Boucher HW, Corey GR, et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med. 2006;355:653.
- Heldman AW, Hartert TV, Ray SC, et al. Oral antibiotic treatment of right-sided staphylococcal endocarditis in injection drug users: prospective randomized comparison with parenteral therapy. Am J Med. 1996;101:68-76.
- Schrenzel J, Harbarth S, Schockmel G, et al. A randomized clinical trial to compare fleroxacin-rifampicin with flucloxacillin or vancomycin for the treatment of Staphylococcal infection. Clin Infect Dis. 2004;39:1285-1292.
- Shorr AF, Kunkel MJ, Kollef M. Linezolid versus vancomycin for Staphylococcus aureus bacteraemia: pooled analysis of randomized studies. J Antimicrob Chemother. 2005;56:923-929.
- Wilson R, Hamburger M. Fifteen years’ experience with Staphylococcus septicemia in a large city hospital; analysis of fifty-five cases in the Cincinnati General Hospital 1940 to 1954. Am J Med. 1957;22:437-457.
- Fowler VG Jr., Sanders LL, Sexton DJ, et al. Outcome of Staphylococcus aureus bacteremia according to compliance with recommendation of infectious disease specialists: experience with 244 patients. Clin Infect Dis. 1998;27:478-486.
- Thomas MG, Morris AJ. Cannula-associated Staphylococcus aureus bacteremia: outcome in relation to treatment. Intern Med J. 2005;35:319-330.
- Kaasch AJ, Fowler VG Jr, Rieg S, et al. Use of a simple criteria set for guiding echocardiography in nosocomidal Staphylococcus aureus bacteremia. Clin Infect Dis. 2011;53:1-9.
- Honda H, Krauss MJ, Jones JC, et al. The value of infectious disease consultation in Staphylococcus aureus bacteremia. Am J Med. 2010;123:631-637.
- Nagao M, Iinuma Y, Saito T, et al. Close cooperation between infectious disease physicians and attending physicians can result in better management and outcome for patients with Staphylococcus aureus bacteremia. Euro Soc Clin Microbiology Infect Dis. 2010;16:1783-1788.
- Fowler VG Jr., Sanders LL, Sexton DJ, et al. Outcome of Staphylococcus aureus bacteremia according to compliance with recommendations of infectious diseases specialist: experience with 244 patients. Clin Infect Dis. 1998;27(3):478-486.
- Jenkins TC, Price CS, Sabel AL et al. Impact of routine infectious diseases service consultation on the evaluation, management, and outcome of Staphylococcus aureus bacteremia. Clin Infect Dis. 2008;46(7):1000-1008.
Case
An 82-year-old man with non-Hodgkin’s lymphoma in remission and a history of congestive heart failure and hypertension presents with one week of generalized malaise and intermittent fevers. Vitals show a temperature of 101oF, blood pressure of 130/60 mmHg, and heart rate of 100. His exam is notable for an erythematous and tender chest port site, with no murmurs. Blood cultures drawn upon presentation show gram-positive cocci speciated to Staphylococcus aureus. What are the next steps in management of this patient?
Overview
S. aureus bacteremia (SAB) is a common infectious cause of morbidity and mortality worldwide, causing both community-acquired and hospital-acquired bacteremia. In the U.S. alone, it accounts for 23% of all bloodstream infections and is the bacterial pathogen most strongly associated with death.1 Mortality rates are approximately 42% in those with methicillin-resistant S. aureus (MRSA) bacteremia and 28% in those with methicillin-sensitive S. aureus (MSSA) bacteremia.2
Recognizing the severity of SAB, the Infectious Disease Society of America (IDSA) published treatment guidelines in 2011 to help direct the clinical care of this disease process.3 However, the majority of the recommendations are based on observational studies and expert opinion, as less than 1,500 patients have been enrolled in randomized controlled trials specifically targeted to investigate the treatment of SAB.4
Review of the Data
A clinically significant SAB usually is defined as the isolation of S. aureus from a venous blood culture with associated symptoms and signs of systemic infection.5 As SAB contamination is rare and can be associated with multiple complications, including metastatic infections, embolic stroke, recurrent infection, and death, any finding of a positive blood culture must be taken seriously.4
SAB treatment is multifaceted and should focus on the removal of any nidus of infection, such as a catheter or a prosthetic device, the use of prolonged antimicrobial therapy, and the evaluation of potential complications. In a retrospective study, Johnson et al showed that failure to remove the source is one of the strongest independent predictors of relapse in patients with SAB.6 However, 10% to 40% of patients have no identifiable focus, which increases the impetus to evaluate for complications.7-8 Overall, approximately one-third of patients with SAB develop metastatic complications, either from hematogenous seeding or local extension of infection.9
In addition to advanced age and such comorbid conditions as cirrhosis, the strongest predictor of complications is a positive blood culture at 48 to 96 hours after an initial positive blood culture, as shown in a large prospective cohort study by Fowler et al.7,10-11 Additional independent risk factors (see Table 1) include community acquisition (likely due to prolonged duration of bacteremia), skin examination suggesting the presence of acute systemic infection, and persistent fever at 72 hours after the first positive blood culture. Patients with even one of these risk factors are at high risk for a complicated course (which occurs in about 35%). In a case-control study, Chihara et al showed that S. aureus bacteruria in the absence of urinary tract pathology or recent urinary tract instrumentation might be associated with threefold increased mortality compared with those without bacteriuria, even after adjustment for comorbid conditions.12
Antimicrobial Treatment
The initial choice of antibiotic therapy for SAB must take into account the MRSA prevalence in the community and hospital. If suspicion is high enough for MRSA, the IDSA’s 2011 guidelines suggest treatment with vancomycin or daptomycin.3 Although there are no published RCTs to support a particular antibiotic regimen, there are trials to suggest that a delay in treatment could be harmful. One study, by Lordis et al, showed that a delay in treatment, as defined by treatment after 44.75 hours, was associated with a longer hospital stay, with the delayed treatment group being hospitalized for 20.2 days and the early treatment group being hospitalized for 14.3 days.13 A delay in treatment was also found to be an independent predictor of mortality.13
Once susceptibilities are known, it is important to appropriately tailor antibiotics, as studies have shown lower treatment failure rates with the use of beta-lactam antibiotics when compared with empiric MRSA coverage.14-15 In one prospective study of 123 hemodialysis patients with MSSA bacteremia, Stryjewski et al showed that those treated with vancomycin were at higher risk of experiencing treatment failure than those treated with cefazolin.15 In another prospective observational study of 505 patients with SAB, Chang et al found that treatment with nafcillin was superior to vancomycin in preventing persistent bacteremia or relapse for MSSA bacteremia.14 These studies highlight the benefits of adjusting the empirically selected antibiotics, as narrowing the spectrum can result in less treatment failure.
If susceptibilities confirm MRSA, the IDSA recommends continued treatment with vancomycin or daptomycin.3 Although vancomycin is most commonly used, partly because of low cost and familiarity, Fowler et al published a study of 246 patients with SAB with or without endocarditis, assigning them to treatment with daptomycin, initial low-dose gentamicin plus vancomycin or an antistaphylococcal penicillin.16 The study found that daptomycin was not inferior to the other therapies, confirming that daptomycin is a reasonable choice in the treatment of MRSA infections.
Oral antibiotics are an option to treat SAB when necessary. A RCT by Heldman et al of 85 intravenous drug users with SAB (and suspected right-sided endocarditis, 65% of which had HIV) showed similar efficacy of ciprofloxacin plus rifampin versus standard intravenous therapy.17 A subsequent randomized trial of 104 patients with SAB comparing oral fleroxacin plus rifampin against conventional intravenous therapy also showed similar cure rates, with the added benefit of earlier discharge.18 Furthermore, in a meta-analysis of five randomized studies by Shorr et al (see Table 2), linezolid was found to have outcomes that were not inferior to vancomycin (clinical cure/microbiological success of 56%/69% in the linezolid group and 46%/73% in the vancomycin group).19
Treatment Duration
Recommendations for the duration of antibiotic treatment for SAB are mainly based on observational studies, which show mixed results. In one study done in the 1950s, about two-thirds of cases of SAB were associated with endocarditis, and longer courses of intravenous therapy (greater than four weeks) were recommended.20
More recently, with the increasing rates of catheter-related SAB and its relatively high rate of expeditious blood culture clearance, a shorter duration has been evaluated in several studies. In 1992, an analysis of published data and a retrospective case series concluded that fewer than 10 days of intravenous antibiotics might be associated with an increased risk of recurrence, but 10 to 14 days of intravenous therapy was effective for most cases of catheter-associated SAB.5 In another prospective study, Fowler et al found that a seven-day course of intravenous antibiotic therapy may be sufficient for simple, catheter-related infections.21 A subsequent prospective study by Jensen et al reported that a course of antibiotic therapy of less than 14 days might be associated with higher mortality compared to a longer course.9 A prospective study of 276 patients by Thomas et al found there was no relationship between relapse and duration of treatment (seven to 15 days) in catheter-related SAB, concluding that more than 14 days of antibiotic therapy was unnecessary.22
Per IDSA guidelines, uncomplicated SAB (no implanted prosthesis, negative blood cultures within two to four days, defervescence within 72 hours of initiating therapy, and lack of metastatic complication) can be treated with a two-week course of antibiotics, while complicated bacteremia (any of above criteria) should be treated within four to six weeks.3
Monitoring for Complications: Echos
Based on the IDSA guidelines, echocardiography is recommended in all patients with bacteremia, with a preference of transesophageal echocardiography (TEE) over transthoracic echocardiography (TTE).3 More recently, Kaasch et al developed simple criteria to identify patients with nosocomial SAB at low risk for infective endocarditis based on two prospective cohort studies.23 Lack of any of these criteria, which include prolonged bacteremia of more than four days’ duration, presence of a permanent intracardiac device, hemodialysis dependency, spinal infection and nonvertebral osteomyelitis, along with a negative TTE indicates that a TEE is not necessary (see Table 3). However, these patients need close follow-up to ensure that bacteremia clears and no new signs or symptoms concerning for metastatic infection develop.
ID Consultation
Several studies have shown that ID consultation not only improves adherence to evidence-based management of SAB, but it also reduces mortality.24-27 In a recent prospective cohort study in a tertiary-care center, even after adjusting for pre-existing comorbidities and severity of disease, an ID consult was associated with a 56% reduction in 28-day mortality.24 The patients who were followed by an ID consult service were more likely to receive appropriate duration of antibiotics (81% vs. 29%, respectively) and undergo appropriate workup for the evaluation of metastatic infections (34% and 8%, respectively). This study concluded that routine ID consult should be considered in patients with SAB, especially those with severe illness and multiple comorbid conditions.
Back to the Case
The patient was started on empiric therapy with vancomycin and serial blood cultures were obtained. He remained hemodynamically stable but febrile, with persistently positive blood and urine cultures. Given concern for the port being the source of his infection, his chest port was removed. A high-quality TTE was performed and was unremarkable.
ID was consulted. Blood cultures subsequently grew MSSA and vancomycin was switched to cefazolin 2g every eight hours. On hospital Day 5, his fever resolved and blood cultures turned negative. There were no clinical signs or symptoms for metastatic infections. A PICC line was placed after blood cultures remained negative for 48 hours. The decision was made to treat him with four weeks of antibiotics from his last positive blood culture, with follow-up in ID clinic.
Bottom Line
SAB is a common worldwide cause of morbidity and mortality. Treatment should include removing the nidus if present, finding and administering the appropriate antimicrobial therapy, evaluating for possible complications, and consulting with ID.
Dr. Ward is an assistant professor, Dr. Kim a clinical instructor, and Dr. Stojan a clinical lecturer at the University of Michigan Health System in Ann Arbor.
Acknowledgement
The authors would like to thank Dr. Jeffrey Rohde for reviewing the manuscript.
References
- Shorr AF, Tabak YP, Killian AD, et al. Healthcare-associated bloodstream infection: a distinct entity? Insights from a large U.S. database. Crit Care Med. 2006;34:3588-3595.
- Mylotte JM, McDermott C, Spooner JA. Prospective study of 114 consecutive episodes of Staphylococcus aureus bacteremia. Rev Infect Dis. 1987;9:891-907.
- Liu C, Bayer A, Cosgrove SE, et al. Clinical practice guidelines by the Infectious Diseases Society of America for the treatment of methicillin-resistant Staphylococcus aureus infections in adults and children. Clin Infect Dis. 2011;52(3).
- Naber CK, Baddour LM, Giamarellos-Bourboulis EJ, et al. Clinical consensus conference: survey on gram-positive bloodstream infections with a focus on Staphylococcus aureus. Clin Infect Dis. 2011;52(3):285-292.
- Thwaites GE, Edgeworth JD, Gkrania-Klotsas E, et al. Clinical management of Staphylococcus bacteremia. Lancet Infect Dis. 2011;11(3):208-222.
- Johnson LB, Almoujahed MO, Ilg K, et al. Staphylococcus aureus bacteremia: compliance with standard treatment, long-term outcome and predictors of relapse. Scand J Infec Dis. 2003;35(11-12):782-789.
- Fowler VG Jr., Olsen MK, Corey GR, et al. Clinical identifiers of complicated Staphylococcus aureus bacteremia. Arch Intern Med. 2003;163:2066-2072.
- Mylotte JM, Tayara A. Staphylococcus aureus bacteremia: predictors of 30-day mortality in a large cohort. Clin Infect Dis. 2000;31:1170-1174.
- Jensen AG, Wachmann CH, Espersen F, et al. Treatment and outcome of Staphylococcus aureus bacteremia: a prospective study of 278 cases. Arch Intern Med. 2002;162(1):25-32.
- Malani PN, Rana MM, Banerjee M, et al. Staphylococcus aureus bloodstream infections: the association between age and mortality and functional status. J Am Geriatr Soc. 2008;56(8):1485-1489.
- Kim SH, Park WB, Lee KD, et al. Outcome of Staphylococcus aureus bacteremia in patients with eradicable foci versus noneradicable foci. Clin Infect Dis. 2003;37(6):794-799.
- Chihara S. Popovich KJ, Weinstein RA, et al. Staphylococcus aureus bacteriuria as a prognosticator for outcome of Staphylococcus aureus bacteremia: a case control study. BMC Inf Dis. 2010;10:225.
- Lodise TP, McKinnon PS, Swiderski L, Rybak MJ. Outcomes analysis of delayed antibiotic treatment for hospital-acquired Staphylococcus aureus bacteremia. Clin Infect Dis. 2003;36:1418-1423.
- Chang FY, Peacock JE Jr., Musher DM, et al. Staphylococcus aureus bacteremia: recurrence and the impact of antibiotic treatment in a prospective multicenter study. Medicine (Baltimore). 2003;82:333.
- Stryjewski ME, Szczech LA, Benjamin DK Jr., et al. Use of vancomycin or first-generation cephalosporins for the treatment of hemodialysisdependent patients with methicillin-susceptible Staphylococcus aureus bacteremia. Clin Infect Dis. 2007;44:190-196.
- Fowler VG Jr., Boucher HW, Corey GR, et al. Daptomycin versus standard therapy for bacteremia and endocarditis caused by Staphylococcus aureus. N Engl J Med. 2006;355:653.
- Heldman AW, Hartert TV, Ray SC, et al. Oral antibiotic treatment of right-sided staphylococcal endocarditis in injection drug users: prospective randomized comparison with parenteral therapy. Am J Med. 1996;101:68-76.
- Schrenzel J, Harbarth S, Schockmel G, et al. A randomized clinical trial to compare fleroxacin-rifampicin with flucloxacillin or vancomycin for the treatment of Staphylococcal infection. Clin Infect Dis. 2004;39:1285-1292.
- Shorr AF, Kunkel MJ, Kollef M. Linezolid versus vancomycin for Staphylococcus aureus bacteraemia: pooled analysis of randomized studies. J Antimicrob Chemother. 2005;56:923-929.
- Wilson R, Hamburger M. Fifteen years’ experience with Staphylococcus septicemia in a large city hospital; analysis of fifty-five cases in the Cincinnati General Hospital 1940 to 1954. Am J Med. 1957;22:437-457.
- Fowler VG Jr., Sanders LL, Sexton DJ, et al. Outcome of Staphylococcus aureus bacteremia according to compliance with recommendation of infectious disease specialists: experience with 244 patients. Clin Infect Dis. 1998;27:478-486.
- Thomas MG, Morris AJ. Cannula-associated Staphylococcus aureus bacteremia: outcome in relation to treatment. Intern Med J. 2005;35:319-330.
- Kaasch AJ, Fowler VG Jr, Rieg S, et al. Use of a simple criteria set for guiding echocardiography in nosocomidal Staphylococcus aureus bacteremia. Clin Infect Dis. 2011;53:1-9.
- Honda H, Krauss MJ, Jones JC, et al. The value of infectious disease consultation in Staphylococcus aureus bacteremia. Am J Med. 2010;123:631-637.
- Nagao M, Iinuma Y, Saito T, et al. Close cooperation between infectious disease physicians and attending physicians can result in better management and outcome for patients with Staphylococcus aureus bacteremia. Euro Soc Clin Microbiology Infect Dis. 2010;16:1783-1788.
- Fowler VG Jr., Sanders LL, Sexton DJ, et al. Outcome of Staphylococcus aureus bacteremia according to compliance with recommendations of infectious diseases specialist: experience with 244 patients. Clin Infect Dis. 1998;27(3):478-486.
- Jenkins TC, Price CS, Sabel AL et al. Impact of routine infectious diseases service consultation on the evaluation, management, and outcome of Staphylococcus aureus bacteremia. Clin Infect Dis. 2008;46(7):1000-1008.
Local Factors Play Major Role in Determining Compensation Rates for Pediatric Hospitalists
Although pediatricians make up less than 6% of the hospitalists surveyed by the Medical Group Management Association (MGMA), they represent a very different data profile from other specialties reported in SHM’s 2012 State of Hospital Medicine report.
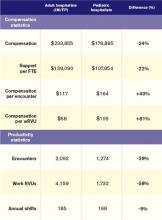
The nonpediatric HM specialties (internal medicine, family medicine, and med/peds) have similar data profiles with regard to productivity and compensation statistics. They are all within 2% of the $233,855 “all adult hospitalists” median compensation. Although there is a bit more variability in the productivity data, all three groups are clustered within 10% of each other. The key to understanding their similarity is that they all serve mostly adult inpatients. While some of these physicians may also care for hospitalized children, I suspect this population is a small proportion of their daily workload.
Pediatric hospitalists only treat pediatric patients and differ significantly from adult hospitalists, as summarized in Table 1.
Pediatricians remain among the lowest-earning specialties nationally, whether in the office or on children’s wards. The key to understanding the differences between adult and pediatric hospitalists is that they derive their compensation and productivity expectations from two separate and distinct physician marketplaces. Adult hospitalists benefit from more than a decade of rapidly growing demand for their services, as well as higher compensation for their office-based counterparts. Meanwhile, the market for pediatric hospitalists remains smaller and more segmented, allowing local factors to drive compensation more than a national demand for their services would.
Pediatric hospitalists appear to earn about a quarter less than their adult counterparts while receiving a similarly lower amount of hospital financial support per provider. Pediatric hospitalists also appear to work less than adult hospitalists, reflected in fewer shifts annually and fewer hours per shift; 75% of adult hospitalist groups report shift lengths of 12 hours or more, compared with 48% of pediatric hospitalist groups. This may stem from the frequent lulls in census common to a community hospital pediatrics service, in contrast to more consistent demand posed by geriatric populations. Although pediatric hospitalists receive more compensation per encounter or wRVU, they cannot generate those encounters or work RVUs at the same clip as adult hospitalists. Pediatricians must hold a family meeting for every single patient, and even something as seemingly simple as obtaining intravenous access might consume 45 minutes of a hospitalist’s time.
Thus, pediatric hospitalists find themselves caught in the same market as other pediatric specialists. These providers remain undervalued compared to virtually all other physicians. Those who seek to improve their financial prospects likely need to work more shifts or generate more workload relative to the expectations of their pediatrician peers.
Personally, I can’t help but wonder what attention pediatric care might enjoy if kids had a vote, a pension, an entitlement program, and a lobby on K Street like their grandparents do.
Dr. Ahlstrom is clinical director of Hospitalists of Northern Michigan and a member of SHM’s Practice Analysis Committee.
Although pediatricians make up less than 6% of the hospitalists surveyed by the Medical Group Management Association (MGMA), they represent a very different data profile from other specialties reported in SHM’s 2012 State of Hospital Medicine report.
The nonpediatric HM specialties (internal medicine, family medicine, and med/peds) have similar data profiles with regard to productivity and compensation statistics. They are all within 2% of the $233,855 “all adult hospitalists” median compensation. Although there is a bit more variability in the productivity data, all three groups are clustered within 10% of each other. The key to understanding their similarity is that they all serve mostly adult inpatients. While some of these physicians may also care for hospitalized children, I suspect this population is a small proportion of their daily workload.
Pediatric hospitalists only treat pediatric patients and differ significantly from adult hospitalists, as summarized in Table 1.
Pediatricians remain among the lowest-earning specialties nationally, whether in the office or on children’s wards. The key to understanding the differences between adult and pediatric hospitalists is that they derive their compensation and productivity expectations from two separate and distinct physician marketplaces. Adult hospitalists benefit from more than a decade of rapidly growing demand for their services, as well as higher compensation for their office-based counterparts. Meanwhile, the market for pediatric hospitalists remains smaller and more segmented, allowing local factors to drive compensation more than a national demand for their services would.
Pediatric hospitalists appear to earn about a quarter less than their adult counterparts while receiving a similarly lower amount of hospital financial support per provider. Pediatric hospitalists also appear to work less than adult hospitalists, reflected in fewer shifts annually and fewer hours per shift; 75% of adult hospitalist groups report shift lengths of 12 hours or more, compared with 48% of pediatric hospitalist groups. This may stem from the frequent lulls in census common to a community hospital pediatrics service, in contrast to more consistent demand posed by geriatric populations. Although pediatric hospitalists receive more compensation per encounter or wRVU, they cannot generate those encounters or work RVUs at the same clip as adult hospitalists. Pediatricians must hold a family meeting for every single patient, and even something as seemingly simple as obtaining intravenous access might consume 45 minutes of a hospitalist’s time.
Thus, pediatric hospitalists find themselves caught in the same market as other pediatric specialists. These providers remain undervalued compared to virtually all other physicians. Those who seek to improve their financial prospects likely need to work more shifts or generate more workload relative to the expectations of their pediatrician peers.
Personally, I can’t help but wonder what attention pediatric care might enjoy if kids had a vote, a pension, an entitlement program, and a lobby on K Street like their grandparents do.
Dr. Ahlstrom is clinical director of Hospitalists of Northern Michigan and a member of SHM’s Practice Analysis Committee.
Although pediatricians make up less than 6% of the hospitalists surveyed by the Medical Group Management Association (MGMA), they represent a very different data profile from other specialties reported in SHM’s 2012 State of Hospital Medicine report.
The nonpediatric HM specialties (internal medicine, family medicine, and med/peds) have similar data profiles with regard to productivity and compensation statistics. They are all within 2% of the $233,855 “all adult hospitalists” median compensation. Although there is a bit more variability in the productivity data, all three groups are clustered within 10% of each other. The key to understanding their similarity is that they all serve mostly adult inpatients. While some of these physicians may also care for hospitalized children, I suspect this population is a small proportion of their daily workload.
Pediatric hospitalists only treat pediatric patients and differ significantly from adult hospitalists, as summarized in Table 1.
Pediatricians remain among the lowest-earning specialties nationally, whether in the office or on children’s wards. The key to understanding the differences between adult and pediatric hospitalists is that they derive their compensation and productivity expectations from two separate and distinct physician marketplaces. Adult hospitalists benefit from more than a decade of rapidly growing demand for their services, as well as higher compensation for their office-based counterparts. Meanwhile, the market for pediatric hospitalists remains smaller and more segmented, allowing local factors to drive compensation more than a national demand for their services would.
Pediatric hospitalists appear to earn about a quarter less than their adult counterparts while receiving a similarly lower amount of hospital financial support per provider. Pediatric hospitalists also appear to work less than adult hospitalists, reflected in fewer shifts annually and fewer hours per shift; 75% of adult hospitalist groups report shift lengths of 12 hours or more, compared with 48% of pediatric hospitalist groups. This may stem from the frequent lulls in census common to a community hospital pediatrics service, in contrast to more consistent demand posed by geriatric populations. Although pediatric hospitalists receive more compensation per encounter or wRVU, they cannot generate those encounters or work RVUs at the same clip as adult hospitalists. Pediatricians must hold a family meeting for every single patient, and even something as seemingly simple as obtaining intravenous access might consume 45 minutes of a hospitalist’s time.
Thus, pediatric hospitalists find themselves caught in the same market as other pediatric specialists. These providers remain undervalued compared to virtually all other physicians. Those who seek to improve their financial prospects likely need to work more shifts or generate more workload relative to the expectations of their pediatrician peers.
Personally, I can’t help but wonder what attention pediatric care might enjoy if kids had a vote, a pension, an entitlement program, and a lobby on K Street like their grandparents do.
Dr. Ahlstrom is clinical director of Hospitalists of Northern Michigan and a member of SHM’s Practice Analysis Committee.
Hospitalists to Unveil Patient Care Recommendations As Part of Choosing Wisely Campaign
This month, hospitalists will be a vital part of Choosing Wisely, an important public initiative from the American Board of Internal Medicine (ABIM) Foundation that identifies treatments and procedures that might be overused by caregivers.
On Feb. 21 in Washington, D.C., the ABIM Foundation, SHM, and more than a dozen other medical specialties will announce recommendations that, in the ABIM Foundation’s words, “represent specific, evidence-based recommendations physicians and patients should discuss to help make wise decisions about the most appropriate care based on their individual situation.” Hospitalists who helped SHM develop its recommendations will be in attendance to help field questions about SHM’s work with Choosing Wisely and its lists.
SHM has developed two lists of recommendations: one for adult HM and another for pediatric HM. SHM will make a special announcement Feb. 21 in The Hospitalist eWire with both lists and commentary for how hospitalists can have informed conversations with their patients about the lists. The Hospitalist will follow up with a feature story and other information about Choosing Wisely in its March issue.
As part of the campaign, the ABIM Foundation, SHM, and consumer magazine Consumer Reports have teamed up to develop material specifically designed to educate patients about the Choosing Wisely recommendations. Materials will be available on the ABIM Foundation and SHM websites.
SHM will continue the conversation about high-value care and working with patients to make wise decisions well beyond February and March. At HM13, SHM’s annual meeting in Washington, D.C., SHM will offer a pre-course on Choosing Wisely and its philosophy. The pre-course is May 16, the day before the official start of HM13.
For more information about Choosing Wisely, visit www.choosingwisely.org. To register for the Choosing Wisely pre-course at HM13, visit www.hospitalmedicine2013.org.
This month, hospitalists will be a vital part of Choosing Wisely, an important public initiative from the American Board of Internal Medicine (ABIM) Foundation that identifies treatments and procedures that might be overused by caregivers.
On Feb. 21 in Washington, D.C., the ABIM Foundation, SHM, and more than a dozen other medical specialties will announce recommendations that, in the ABIM Foundation’s words, “represent specific, evidence-based recommendations physicians and patients should discuss to help make wise decisions about the most appropriate care based on their individual situation.” Hospitalists who helped SHM develop its recommendations will be in attendance to help field questions about SHM’s work with Choosing Wisely and its lists.
SHM has developed two lists of recommendations: one for adult HM and another for pediatric HM. SHM will make a special announcement Feb. 21 in The Hospitalist eWire with both lists and commentary for how hospitalists can have informed conversations with their patients about the lists. The Hospitalist will follow up with a feature story and other information about Choosing Wisely in its March issue.
As part of the campaign, the ABIM Foundation, SHM, and consumer magazine Consumer Reports have teamed up to develop material specifically designed to educate patients about the Choosing Wisely recommendations. Materials will be available on the ABIM Foundation and SHM websites.
SHM will continue the conversation about high-value care and working with patients to make wise decisions well beyond February and March. At HM13, SHM’s annual meeting in Washington, D.C., SHM will offer a pre-course on Choosing Wisely and its philosophy. The pre-course is May 16, the day before the official start of HM13.
For more information about Choosing Wisely, visit www.choosingwisely.org. To register for the Choosing Wisely pre-course at HM13, visit www.hospitalmedicine2013.org.
This month, hospitalists will be a vital part of Choosing Wisely, an important public initiative from the American Board of Internal Medicine (ABIM) Foundation that identifies treatments and procedures that might be overused by caregivers.
On Feb. 21 in Washington, D.C., the ABIM Foundation, SHM, and more than a dozen other medical specialties will announce recommendations that, in the ABIM Foundation’s words, “represent specific, evidence-based recommendations physicians and patients should discuss to help make wise decisions about the most appropriate care based on their individual situation.” Hospitalists who helped SHM develop its recommendations will be in attendance to help field questions about SHM’s work with Choosing Wisely and its lists.
SHM has developed two lists of recommendations: one for adult HM and another for pediatric HM. SHM will make a special announcement Feb. 21 in The Hospitalist eWire with both lists and commentary for how hospitalists can have informed conversations with their patients about the lists. The Hospitalist will follow up with a feature story and other information about Choosing Wisely in its March issue.
As part of the campaign, the ABIM Foundation, SHM, and consumer magazine Consumer Reports have teamed up to develop material specifically designed to educate patients about the Choosing Wisely recommendations. Materials will be available on the ABIM Foundation and SHM websites.
SHM will continue the conversation about high-value care and working with patients to make wise decisions well beyond February and March. At HM13, SHM’s annual meeting in Washington, D.C., SHM will offer a pre-course on Choosing Wisely and its philosophy. The pre-course is May 16, the day before the official start of HM13.
For more information about Choosing Wisely, visit www.choosingwisely.org. To register for the Choosing Wisely pre-course at HM13, visit www.hospitalmedicine2013.org.
Pharmacist-Hospitalist Collaboration Can Improve Care, Save Money
A healthy collaboration between hospitalists and pharmacists can generate cost savings and promote positive outcomes, such as preventing adverse drug events and improving care transitions, says Jonathan Edwards, PharmD, BCPS, a clinical pharmacy specialist at Huntsville Hospital in Alabama.
At the 2012 national conference of the American College of Clinical Pharmacy in Hollywood, Fla., Edwards presented a poster that detailed the effectiveness of such interdisciplinary collaboration at Huntsville Hospital, where pharmacists and physicians developed six order sets, a collaborative practice, and a patient interaction program from November 2011 to February 2012. During the study period, researchers documented a total cost savings of $9,825 resulting from 156 patient interventions.
Edwards’ collaborative study at Huntsville started with two physicians who had launched a service teaching hospitalists what pharmacists do, and how they could help in their efforts.
“We got together and developed an order set for treating acute alcohol withdrawal. That went well, so we did five more order sets,” Edwards says. “Then we thought: What if pharmacists got more involved by meeting directly with patients in the hospital to optimize their medication management and help them reach their goals for treatment? We now evaluate patients on the hospitalist service in three units.”
For Edwards, key factors that make the hospitalist-pharmacist relationship work include communicating the pharmacist’s availability to help with the hospitalist’s patients, identifying the physician’s openness to help, and clarifying how the physician prefers to be contacted.
Last October, the American Society of Health-System Pharmacists (ASHP) and the American Pharmacists Association (APhA) recognized eight care-transitions programs for best practices that improved patient outcomes and reduced hospital readmissions as part of the Medication Management in Care Transitions (MMCT) Project.
“The MMCT project highlights the valuable role pharmacists can play in addressing medication-related problems that can lead to hospital readmissions,” APhA chief executive officer Thomas E. Menighan, BSPharm, MBA, ScD (Hon), FAPhA, said in a news release. “By putting together these best practices, our goal is to provide a model for better coordination of care and better connectivity between pharmacists and healthcare providers in different practice settings that leads to improved patient health.”
Visit our website for more information about maximizing patient care through pharmacist-hospitalist collaboration.
Larry Beresford is a freelance writer in Oakland, Calif.
Articles first published in the Jan. 16, 2013, edition of The Hospitalist eWire.
A healthy collaboration between hospitalists and pharmacists can generate cost savings and promote positive outcomes, such as preventing adverse drug events and improving care transitions, says Jonathan Edwards, PharmD, BCPS, a clinical pharmacy specialist at Huntsville Hospital in Alabama.
At the 2012 national conference of the American College of Clinical Pharmacy in Hollywood, Fla., Edwards presented a poster that detailed the effectiveness of such interdisciplinary collaboration at Huntsville Hospital, where pharmacists and physicians developed six order sets, a collaborative practice, and a patient interaction program from November 2011 to February 2012. During the study period, researchers documented a total cost savings of $9,825 resulting from 156 patient interventions.
Edwards’ collaborative study at Huntsville started with two physicians who had launched a service teaching hospitalists what pharmacists do, and how they could help in their efforts.
“We got together and developed an order set for treating acute alcohol withdrawal. That went well, so we did five more order sets,” Edwards says. “Then we thought: What if pharmacists got more involved by meeting directly with patients in the hospital to optimize their medication management and help them reach their goals for treatment? We now evaluate patients on the hospitalist service in three units.”
For Edwards, key factors that make the hospitalist-pharmacist relationship work include communicating the pharmacist’s availability to help with the hospitalist’s patients, identifying the physician’s openness to help, and clarifying how the physician prefers to be contacted.
Last October, the American Society of Health-System Pharmacists (ASHP) and the American Pharmacists Association (APhA) recognized eight care-transitions programs for best practices that improved patient outcomes and reduced hospital readmissions as part of the Medication Management in Care Transitions (MMCT) Project.
“The MMCT project highlights the valuable role pharmacists can play in addressing medication-related problems that can lead to hospital readmissions,” APhA chief executive officer Thomas E. Menighan, BSPharm, MBA, ScD (Hon), FAPhA, said in a news release. “By putting together these best practices, our goal is to provide a model for better coordination of care and better connectivity between pharmacists and healthcare providers in different practice settings that leads to improved patient health.”
Visit our website for more information about maximizing patient care through pharmacist-hospitalist collaboration.
Larry Beresford is a freelance writer in Oakland, Calif.
Articles first published in the Jan. 16, 2013, edition of The Hospitalist eWire.
A healthy collaboration between hospitalists and pharmacists can generate cost savings and promote positive outcomes, such as preventing adverse drug events and improving care transitions, says Jonathan Edwards, PharmD, BCPS, a clinical pharmacy specialist at Huntsville Hospital in Alabama.
At the 2012 national conference of the American College of Clinical Pharmacy in Hollywood, Fla., Edwards presented a poster that detailed the effectiveness of such interdisciplinary collaboration at Huntsville Hospital, where pharmacists and physicians developed six order sets, a collaborative practice, and a patient interaction program from November 2011 to February 2012. During the study period, researchers documented a total cost savings of $9,825 resulting from 156 patient interventions.
Edwards’ collaborative study at Huntsville started with two physicians who had launched a service teaching hospitalists what pharmacists do, and how they could help in their efforts.
“We got together and developed an order set for treating acute alcohol withdrawal. That went well, so we did five more order sets,” Edwards says. “Then we thought: What if pharmacists got more involved by meeting directly with patients in the hospital to optimize their medication management and help them reach their goals for treatment? We now evaluate patients on the hospitalist service in three units.”
For Edwards, key factors that make the hospitalist-pharmacist relationship work include communicating the pharmacist’s availability to help with the hospitalist’s patients, identifying the physician’s openness to help, and clarifying how the physician prefers to be contacted.
Last October, the American Society of Health-System Pharmacists (ASHP) and the American Pharmacists Association (APhA) recognized eight care-transitions programs for best practices that improved patient outcomes and reduced hospital readmissions as part of the Medication Management in Care Transitions (MMCT) Project.
“The MMCT project highlights the valuable role pharmacists can play in addressing medication-related problems that can lead to hospital readmissions,” APhA chief executive officer Thomas E. Menighan, BSPharm, MBA, ScD (Hon), FAPhA, said in a news release. “By putting together these best practices, our goal is to provide a model for better coordination of care and better connectivity between pharmacists and healthcare providers in different practice settings that leads to improved patient health.”
Visit our website for more information about maximizing patient care through pharmacist-hospitalist collaboration.
Larry Beresford is a freelance writer in Oakland, Calif.
Articles first published in the Jan. 16, 2013, edition of The Hospitalist eWire.
The Patient-Doctor Relationship Gap
Physicians who rank poorly in their communication skills with patients were associated with reduced rates of medication adherence in a new report.
A cross-sectional study of nearly 9,4000 patients in the Diabetes Study of Northern California (DISTANCE) found roughly 30% of patients who gave their physicians poor ratings when it came to involving them in decisions, understanding their problems with medications, and eliciting their trust were less likely to refill their cardiometabolic medications than those whose doctors were deemed to be good communicators, researchers found. For each 10-point decrease in the Consumer Assessment of Healthcare Providers and Systems Survey (CAHPS), the prevalence of poor medication adherence increased by 0.9% (P +0.1), the researchers added.
“One of the tricks is that medication adherence is an inherently physician-centric concept,” says lead author Neda Ratanawongsa, MD, MPH, assistant professor in the department of medicine at the University of California at San Francisco (UCSF). “We’re asking you to take medicine that we think will be best for you. That’s been the way that physicians operate for years, often appropriately so. But part of this is figuring out how to encourage the patients to disclose their decision that ‘Yes, I do want to take that medicine’ or ‘No, here’s why I don’t want to take that medicine.’”
Dr. Ratanawongsa adds that hospitalists and other physicians have to develop a sense of trust with patients to build relationships. Future studies could then track patient satisfaction and adherence over time to see if a corollary exists. Also, she says, hospitalists shouldn’t be discouraged that most of their relationships aren’t long-term ones like those found in other specialties.
“I wouldn’t underestimate the impact a hospitalist could have, whether one-time interaction or not, to change an existing therapy program,” Dr. Ratanawongsa says. “It’s important for hospitalists to understand the power of their words.”
Richard Quinn is a freelance writer in New Jersey.
Physicians who rank poorly in their communication skills with patients were associated with reduced rates of medication adherence in a new report.
A cross-sectional study of nearly 9,4000 patients in the Diabetes Study of Northern California (DISTANCE) found roughly 30% of patients who gave their physicians poor ratings when it came to involving them in decisions, understanding their problems with medications, and eliciting their trust were less likely to refill their cardiometabolic medications than those whose doctors were deemed to be good communicators, researchers found. For each 10-point decrease in the Consumer Assessment of Healthcare Providers and Systems Survey (CAHPS), the prevalence of poor medication adherence increased by 0.9% (P +0.1), the researchers added.
“One of the tricks is that medication adherence is an inherently physician-centric concept,” says lead author Neda Ratanawongsa, MD, MPH, assistant professor in the department of medicine at the University of California at San Francisco (UCSF). “We’re asking you to take medicine that we think will be best for you. That’s been the way that physicians operate for years, often appropriately so. But part of this is figuring out how to encourage the patients to disclose their decision that ‘Yes, I do want to take that medicine’ or ‘No, here’s why I don’t want to take that medicine.’”
Dr. Ratanawongsa adds that hospitalists and other physicians have to develop a sense of trust with patients to build relationships. Future studies could then track patient satisfaction and adherence over time to see if a corollary exists. Also, she says, hospitalists shouldn’t be discouraged that most of their relationships aren’t long-term ones like those found in other specialties.
“I wouldn’t underestimate the impact a hospitalist could have, whether one-time interaction or not, to change an existing therapy program,” Dr. Ratanawongsa says. “It’s important for hospitalists to understand the power of their words.”
Richard Quinn is a freelance writer in New Jersey.
Physicians who rank poorly in their communication skills with patients were associated with reduced rates of medication adherence in a new report.
A cross-sectional study of nearly 9,4000 patients in the Diabetes Study of Northern California (DISTANCE) found roughly 30% of patients who gave their physicians poor ratings when it came to involving them in decisions, understanding their problems with medications, and eliciting their trust were less likely to refill their cardiometabolic medications than those whose doctors were deemed to be good communicators, researchers found. For each 10-point decrease in the Consumer Assessment of Healthcare Providers and Systems Survey (CAHPS), the prevalence of poor medication adherence increased by 0.9% (P +0.1), the researchers added.
“One of the tricks is that medication adherence is an inherently physician-centric concept,” says lead author Neda Ratanawongsa, MD, MPH, assistant professor in the department of medicine at the University of California at San Francisco (UCSF). “We’re asking you to take medicine that we think will be best for you. That’s been the way that physicians operate for years, often appropriately so. But part of this is figuring out how to encourage the patients to disclose their decision that ‘Yes, I do want to take that medicine’ or ‘No, here’s why I don’t want to take that medicine.’”
Dr. Ratanawongsa adds that hospitalists and other physicians have to develop a sense of trust with patients to build relationships. Future studies could then track patient satisfaction and adherence over time to see if a corollary exists. Also, she says, hospitalists shouldn’t be discouraged that most of their relationships aren’t long-term ones like those found in other specialties.
“I wouldn’t underestimate the impact a hospitalist could have, whether one-time interaction or not, to change an existing therapy program,” Dr. Ratanawongsa says. “It’s important for hospitalists to understand the power of their words.”
Richard Quinn is a freelance writer in New Jersey.
New Oral Anticoagulants Advance Treatment Options, Create Complications for Hospitalists
New oral anticoagulants promise to impact hospitalists and their patients—but the question is how much
When the FDA gave the nod to factor Xa-inhibitor rivaroxaban in November for use in treating acute DVT and pulmonary embolism (PE), it was just the latest development in the swiftly evolving world of oral anticoagulants—a world that hospitalists had better get used to living in, and quick.
As many as 80% of the patients that hospitalists encounter are on some kind of anticoagulant, experts say. But the extent to which the emergence of the new drugs— particularly rivaroxaban, which also is approved for stroke prevention in nonvalvular atrial fibrillation (afib) and for DVT and PE prevention in knee and hip replacement patients—will affect the daily routines of hospitalists remains to be seen.
Hospitalists specializing in VTE prevention and vascular experts say that the new drugs will make life simpler for hospitalists in some ways, mainly because for some patients, a pill will replace the injectable enoxaparin that has been used to bridge patients to warfarin. But with more options available, things will become more complicated as well, they say.
Approvals for the new agents, which aim to replace warfarin and its need for constant monitoring and concern over drug and food interactions, have been coming rapid-fire. Along with rivaroxaban in the new oral anticoagulant group are dabigatran, approved in late 2010 for stroke prevention in nonvalvular afib, and apixaban, which is expected to be approved for the same indication this year.
“Of all three drugs, [rivaroxaban] has the broadest indications for use,” said Hiren Shah, MD, assistant professor of medicine at Northwestern University’s Feinberg School of Medicine and medical director of hospital medicine at Northwestern Memorial Hospital in Chicago. “Because of that, it’s likely that it’s going to be the agent that will be adopted much more broadly and more easily than dabigatran.”
But apixaban might come on strong in the U.S. when it’s approved because it shows better promise for patients with renal impairment and has a lower risk of intracranial hemorrhage, says Geno Merli, MD, director of the Jefferson Center for Vascular Disease and chief medical officer at Thomas Jefferson University Hospital in Philadelphia.
“I think the two other [manufacturers] are afraid of apixaban because apixaban’s safety profile was much better,” he says.
Steven Deitelzweig, MD, FACP, SFHM, secretary of the Gulf State chapter of SHM and a DVT prevention specialist at Ochsner Health System New Orleans, says hurdles to adoption of the new agents will include whether a system is integrated and can assure appropriate follow-up and concerns over proper patient selection and cost, as the cost-benefit analyses haven’t been done yet.
“The learning curve, or the adoption curve, is really going to be very variable around the country,” he says.
Experts agree changes are on the way as the new anticoagulants gain more traction. Here are some things they say hospitalists should watch out for.
Care and Discharge
Dr. Shah says the availability of the oral agents will streamline care and discharge of patients.
“The care and coordination process that needs to occur with the use of parenteral agents and warfarin is significantly more complex than the patient education and care coordination that will be required with the new oral anticoagulants,” he says. “That’s where there’s a significant time savings.”
Ian Jenkins, MD, assistant professor in the division of hospital medicine at the University of California at San Diego, says the windfall of time saved might not hit hospitalists directly, at least at some centers.
“The education is being done by pharmacy here for warfarin, and nurses handle enoxaparin injection teaching,” he says. “So the workload that benefits may be that of our colleagues.”
Dr. Shah notes that the responsibility in patient counseling ultimately falls within hospitalists’ purview, so he predicts that any greater simplicity in that regard would help hospitalists.
Who Ends up Hospitalized?
The option of oral agents might help diminish the number of patients who have to stay in the hospital for enoxaparin injections that bridge them to warfarin, a topic at a recent roundtable discussion Dr. Shah attended.
“It was shocking to me that I have many colleagues throughout the country who have patients who are in the hospital simply to get parenteral injections because they can’t take them themselves at home and have no loved ones or friends to help them,” he says.
Dr. Merli agrees that the new agents might affect hospitalists’ patient census. Many patients, he says, will be discharged straight from the ED.
“The DVT patients probably won’t get [admitted]. You’re going to put them on rivaroxaban and send them home,” he says. “You’re not going to get [admitted] with a DVT anymore, unless it’s extensive. And if it’s an extensive DVT, you’re not going to get rivaroxaban. You’re going to get enoxaparin or you’re going to get thrombolytics therapy followed by IV heparin followed by enoxaparin. So I don’t see rivaroxaban jumping into the marketplace and being a boon to hospitalists immediately.”
If patients skip hospitalization and are discharged straight from the ED, Dr. Deitelzweig says, “there will be patients who will backfill those spots.”
“I think most of the people will come in as observation status, if not inpatient,” he says, although simpler DVT patients will be likelier candidates for discharge from the ER. He predicts that stays might be shorter, though.
But Dr. Merli says hospitalists shouldn’t expect a big effect on length of stay.
“I don’t think you’re going to reduce dramatically length of stay because you have an oral pill,” he says.
QI Initiatives
What might be a boon, though, are opportunities for quality-improvement (QI) initiatives related to the new therapies, Dr. Jenkins says. “Many of these projects that are being done with anticoagulants … do focus on warfarin safety; it’s a frequent part of readmission and patient harm,” he says. “Having it much simpler to treat and educate these patients is actually going to be a boon, I think, for hospitalists working on quality-improvement projects, and for people who do that education, whether that is a hospitalist or a pharmacist or some other member of the staff.”
There are downsides, though, he notes. One is cost. Another is reversibility. Warfarin can be easily reversed in the event of a bleed, but that’s not the case with rivaroxaban and dabigatran. And none of the new therapies are suitable for patients with renal failure.
“Right now, we’re stuck with IV heparin and Coumadin in the hospital, and rivaroxaban won’t change this,” he says. “Rivaroxaban patients one might help with PCC; but with dabigatran, I don’t think much will help besides time and dialysis, which is dangerous in unstable anticoagulated patients.”
Dr. Shah, though, says he’s aware of only two times that bleeding reversal protocols—based on anecdotal evidence, because no method has been scientifically proven—had to be invoked at Northwestern in the past year.
It might be “more of a theoretical problem than one in reality,” he explains, “simply because we have not found the need to reverse oral anticoagulants very often given their short half-lives.”
The new agents, all the experts agree, will require hospitalists to stay on their toes.
“There are so many different facets in each case, whether it’s the age or the renal function or whether there’s a fall risk and what their compliance is, what their funding is, what the exact indication is,” Dr. Jenkins says. “Keeping up with those things is actually quite challenging.”
His main resources are the American College of Chest Physicians’ guidelines on anticoagulants, his center’s own protocols, and the primary literature for the main trials (see “Additional Reading,” right). He also looks to the inpatient pharmacist for guidance.
Dr. Shah says it is important to be aware of the patient-inclusion criteria, study design, and outcomes measured for each agent through their trials.
“There is a lot of information out there, and there are very subtle aspects of some of these trials and you’ve got to really understand: Does it apply to the patient that is front of me?” he said. “There’s a lot to know, there’s no doubt about it.”
Thomas R. Collins is a freelance writer in South Florida.
New oral anticoagulants promise to impact hospitalists and their patients—but the question is how much
When the FDA gave the nod to factor Xa-inhibitor rivaroxaban in November for use in treating acute DVT and pulmonary embolism (PE), it was just the latest development in the swiftly evolving world of oral anticoagulants—a world that hospitalists had better get used to living in, and quick.
As many as 80% of the patients that hospitalists encounter are on some kind of anticoagulant, experts say. But the extent to which the emergence of the new drugs— particularly rivaroxaban, which also is approved for stroke prevention in nonvalvular atrial fibrillation (afib) and for DVT and PE prevention in knee and hip replacement patients—will affect the daily routines of hospitalists remains to be seen.
Hospitalists specializing in VTE prevention and vascular experts say that the new drugs will make life simpler for hospitalists in some ways, mainly because for some patients, a pill will replace the injectable enoxaparin that has been used to bridge patients to warfarin. But with more options available, things will become more complicated as well, they say.
Approvals for the new agents, which aim to replace warfarin and its need for constant monitoring and concern over drug and food interactions, have been coming rapid-fire. Along with rivaroxaban in the new oral anticoagulant group are dabigatran, approved in late 2010 for stroke prevention in nonvalvular afib, and apixaban, which is expected to be approved for the same indication this year.
“Of all three drugs, [rivaroxaban] has the broadest indications for use,” said Hiren Shah, MD, assistant professor of medicine at Northwestern University’s Feinberg School of Medicine and medical director of hospital medicine at Northwestern Memorial Hospital in Chicago. “Because of that, it’s likely that it’s going to be the agent that will be adopted much more broadly and more easily than dabigatran.”
But apixaban might come on strong in the U.S. when it’s approved because it shows better promise for patients with renal impairment and has a lower risk of intracranial hemorrhage, says Geno Merli, MD, director of the Jefferson Center for Vascular Disease and chief medical officer at Thomas Jefferson University Hospital in Philadelphia.
“I think the two other [manufacturers] are afraid of apixaban because apixaban’s safety profile was much better,” he says.
Steven Deitelzweig, MD, FACP, SFHM, secretary of the Gulf State chapter of SHM and a DVT prevention specialist at Ochsner Health System New Orleans, says hurdles to adoption of the new agents will include whether a system is integrated and can assure appropriate follow-up and concerns over proper patient selection and cost, as the cost-benefit analyses haven’t been done yet.
“The learning curve, or the adoption curve, is really going to be very variable around the country,” he says.
Experts agree changes are on the way as the new anticoagulants gain more traction. Here are some things they say hospitalists should watch out for.
Care and Discharge
Dr. Shah says the availability of the oral agents will streamline care and discharge of patients.
“The care and coordination process that needs to occur with the use of parenteral agents and warfarin is significantly more complex than the patient education and care coordination that will be required with the new oral anticoagulants,” he says. “That’s where there’s a significant time savings.”
Ian Jenkins, MD, assistant professor in the division of hospital medicine at the University of California at San Diego, says the windfall of time saved might not hit hospitalists directly, at least at some centers.
“The education is being done by pharmacy here for warfarin, and nurses handle enoxaparin injection teaching,” he says. “So the workload that benefits may be that of our colleagues.”
Dr. Shah notes that the responsibility in patient counseling ultimately falls within hospitalists’ purview, so he predicts that any greater simplicity in that regard would help hospitalists.
Who Ends up Hospitalized?
The option of oral agents might help diminish the number of patients who have to stay in the hospital for enoxaparin injections that bridge them to warfarin, a topic at a recent roundtable discussion Dr. Shah attended.
“It was shocking to me that I have many colleagues throughout the country who have patients who are in the hospital simply to get parenteral injections because they can’t take them themselves at home and have no loved ones or friends to help them,” he says.
Dr. Merli agrees that the new agents might affect hospitalists’ patient census. Many patients, he says, will be discharged straight from the ED.
“The DVT patients probably won’t get [admitted]. You’re going to put them on rivaroxaban and send them home,” he says. “You’re not going to get [admitted] with a DVT anymore, unless it’s extensive. And if it’s an extensive DVT, you’re not going to get rivaroxaban. You’re going to get enoxaparin or you’re going to get thrombolytics therapy followed by IV heparin followed by enoxaparin. So I don’t see rivaroxaban jumping into the marketplace and being a boon to hospitalists immediately.”
If patients skip hospitalization and are discharged straight from the ED, Dr. Deitelzweig says, “there will be patients who will backfill those spots.”
“I think most of the people will come in as observation status, if not inpatient,” he says, although simpler DVT patients will be likelier candidates for discharge from the ER. He predicts that stays might be shorter, though.
But Dr. Merli says hospitalists shouldn’t expect a big effect on length of stay.
“I don’t think you’re going to reduce dramatically length of stay because you have an oral pill,” he says.
QI Initiatives
What might be a boon, though, are opportunities for quality-improvement (QI) initiatives related to the new therapies, Dr. Jenkins says. “Many of these projects that are being done with anticoagulants … do focus on warfarin safety; it’s a frequent part of readmission and patient harm,” he says. “Having it much simpler to treat and educate these patients is actually going to be a boon, I think, for hospitalists working on quality-improvement projects, and for people who do that education, whether that is a hospitalist or a pharmacist or some other member of the staff.”
There are downsides, though, he notes. One is cost. Another is reversibility. Warfarin can be easily reversed in the event of a bleed, but that’s not the case with rivaroxaban and dabigatran. And none of the new therapies are suitable for patients with renal failure.
“Right now, we’re stuck with IV heparin and Coumadin in the hospital, and rivaroxaban won’t change this,” he says. “Rivaroxaban patients one might help with PCC; but with dabigatran, I don’t think much will help besides time and dialysis, which is dangerous in unstable anticoagulated patients.”
Dr. Shah, though, says he’s aware of only two times that bleeding reversal protocols—based on anecdotal evidence, because no method has been scientifically proven—had to be invoked at Northwestern in the past year.
It might be “more of a theoretical problem than one in reality,” he explains, “simply because we have not found the need to reverse oral anticoagulants very often given their short half-lives.”
The new agents, all the experts agree, will require hospitalists to stay on their toes.
“There are so many different facets in each case, whether it’s the age or the renal function or whether there’s a fall risk and what their compliance is, what their funding is, what the exact indication is,” Dr. Jenkins says. “Keeping up with those things is actually quite challenging.”
His main resources are the American College of Chest Physicians’ guidelines on anticoagulants, his center’s own protocols, and the primary literature for the main trials (see “Additional Reading,” right). He also looks to the inpatient pharmacist for guidance.
Dr. Shah says it is important to be aware of the patient-inclusion criteria, study design, and outcomes measured for each agent through their trials.
“There is a lot of information out there, and there are very subtle aspects of some of these trials and you’ve got to really understand: Does it apply to the patient that is front of me?” he said. “There’s a lot to know, there’s no doubt about it.”
Thomas R. Collins is a freelance writer in South Florida.
New oral anticoagulants promise to impact hospitalists and their patients—but the question is how much
When the FDA gave the nod to factor Xa-inhibitor rivaroxaban in November for use in treating acute DVT and pulmonary embolism (PE), it was just the latest development in the swiftly evolving world of oral anticoagulants—a world that hospitalists had better get used to living in, and quick.
As many as 80% of the patients that hospitalists encounter are on some kind of anticoagulant, experts say. But the extent to which the emergence of the new drugs— particularly rivaroxaban, which also is approved for stroke prevention in nonvalvular atrial fibrillation (afib) and for DVT and PE prevention in knee and hip replacement patients—will affect the daily routines of hospitalists remains to be seen.
Hospitalists specializing in VTE prevention and vascular experts say that the new drugs will make life simpler for hospitalists in some ways, mainly because for some patients, a pill will replace the injectable enoxaparin that has been used to bridge patients to warfarin. But with more options available, things will become more complicated as well, they say.
Approvals for the new agents, which aim to replace warfarin and its need for constant monitoring and concern over drug and food interactions, have been coming rapid-fire. Along with rivaroxaban in the new oral anticoagulant group are dabigatran, approved in late 2010 for stroke prevention in nonvalvular afib, and apixaban, which is expected to be approved for the same indication this year.
“Of all three drugs, [rivaroxaban] has the broadest indications for use,” said Hiren Shah, MD, assistant professor of medicine at Northwestern University’s Feinberg School of Medicine and medical director of hospital medicine at Northwestern Memorial Hospital in Chicago. “Because of that, it’s likely that it’s going to be the agent that will be adopted much more broadly and more easily than dabigatran.”
But apixaban might come on strong in the U.S. when it’s approved because it shows better promise for patients with renal impairment and has a lower risk of intracranial hemorrhage, says Geno Merli, MD, director of the Jefferson Center for Vascular Disease and chief medical officer at Thomas Jefferson University Hospital in Philadelphia.
“I think the two other [manufacturers] are afraid of apixaban because apixaban’s safety profile was much better,” he says.
Steven Deitelzweig, MD, FACP, SFHM, secretary of the Gulf State chapter of SHM and a DVT prevention specialist at Ochsner Health System New Orleans, says hurdles to adoption of the new agents will include whether a system is integrated and can assure appropriate follow-up and concerns over proper patient selection and cost, as the cost-benefit analyses haven’t been done yet.
“The learning curve, or the adoption curve, is really going to be very variable around the country,” he says.
Experts agree changes are on the way as the new anticoagulants gain more traction. Here are some things they say hospitalists should watch out for.
Care and Discharge
Dr. Shah says the availability of the oral agents will streamline care and discharge of patients.
“The care and coordination process that needs to occur with the use of parenteral agents and warfarin is significantly more complex than the patient education and care coordination that will be required with the new oral anticoagulants,” he says. “That’s where there’s a significant time savings.”
Ian Jenkins, MD, assistant professor in the division of hospital medicine at the University of California at San Diego, says the windfall of time saved might not hit hospitalists directly, at least at some centers.
“The education is being done by pharmacy here for warfarin, and nurses handle enoxaparin injection teaching,” he says. “So the workload that benefits may be that of our colleagues.”
Dr. Shah notes that the responsibility in patient counseling ultimately falls within hospitalists’ purview, so he predicts that any greater simplicity in that regard would help hospitalists.
Who Ends up Hospitalized?
The option of oral agents might help diminish the number of patients who have to stay in the hospital for enoxaparin injections that bridge them to warfarin, a topic at a recent roundtable discussion Dr. Shah attended.
“It was shocking to me that I have many colleagues throughout the country who have patients who are in the hospital simply to get parenteral injections because they can’t take them themselves at home and have no loved ones or friends to help them,” he says.
Dr. Merli agrees that the new agents might affect hospitalists’ patient census. Many patients, he says, will be discharged straight from the ED.
“The DVT patients probably won’t get [admitted]. You’re going to put them on rivaroxaban and send them home,” he says. “You’re not going to get [admitted] with a DVT anymore, unless it’s extensive. And if it’s an extensive DVT, you’re not going to get rivaroxaban. You’re going to get enoxaparin or you’re going to get thrombolytics therapy followed by IV heparin followed by enoxaparin. So I don’t see rivaroxaban jumping into the marketplace and being a boon to hospitalists immediately.”
If patients skip hospitalization and are discharged straight from the ED, Dr. Deitelzweig says, “there will be patients who will backfill those spots.”
“I think most of the people will come in as observation status, if not inpatient,” he says, although simpler DVT patients will be likelier candidates for discharge from the ER. He predicts that stays might be shorter, though.
But Dr. Merli says hospitalists shouldn’t expect a big effect on length of stay.
“I don’t think you’re going to reduce dramatically length of stay because you have an oral pill,” he says.
QI Initiatives
What might be a boon, though, are opportunities for quality-improvement (QI) initiatives related to the new therapies, Dr. Jenkins says. “Many of these projects that are being done with anticoagulants … do focus on warfarin safety; it’s a frequent part of readmission and patient harm,” he says. “Having it much simpler to treat and educate these patients is actually going to be a boon, I think, for hospitalists working on quality-improvement projects, and for people who do that education, whether that is a hospitalist or a pharmacist or some other member of the staff.”
There are downsides, though, he notes. One is cost. Another is reversibility. Warfarin can be easily reversed in the event of a bleed, but that’s not the case with rivaroxaban and dabigatran. And none of the new therapies are suitable for patients with renal failure.
“Right now, we’re stuck with IV heparin and Coumadin in the hospital, and rivaroxaban won’t change this,” he says. “Rivaroxaban patients one might help with PCC; but with dabigatran, I don’t think much will help besides time and dialysis, which is dangerous in unstable anticoagulated patients.”
Dr. Shah, though, says he’s aware of only two times that bleeding reversal protocols—based on anecdotal evidence, because no method has been scientifically proven—had to be invoked at Northwestern in the past year.
It might be “more of a theoretical problem than one in reality,” he explains, “simply because we have not found the need to reverse oral anticoagulants very often given their short half-lives.”
The new agents, all the experts agree, will require hospitalists to stay on their toes.
“There are so many different facets in each case, whether it’s the age or the renal function or whether there’s a fall risk and what their compliance is, what their funding is, what the exact indication is,” Dr. Jenkins says. “Keeping up with those things is actually quite challenging.”
His main resources are the American College of Chest Physicians’ guidelines on anticoagulants, his center’s own protocols, and the primary literature for the main trials (see “Additional Reading,” right). He also looks to the inpatient pharmacist for guidance.
Dr. Shah says it is important to be aware of the patient-inclusion criteria, study design, and outcomes measured for each agent through their trials.
“There is a lot of information out there, and there are very subtle aspects of some of these trials and you’ve got to really understand: Does it apply to the patient that is front of me?” he said. “There’s a lot to know, there’s no doubt about it.”
Thomas R. Collins is a freelance writer in South Florida.
Quality Improvement Project Helps Hospital Patients Get Needed Prescriptions
A quality-improvement (QI) project to give high-risk patients ready access to prescribed medications at the time of hospital discharge achieved an 86% success rate, according to an abstract poster presented at HM12 in San Diego last April.1
Lead author Elizabeth Le, MD, then a resident at the University of California at San Francisco Medical Center (UCSF) and now a practicing hospitalist at the Veterans Administration Medical Center in Palo Alto, Calif., says the multidisciplinary “brown bag medications” project involved training house staff to recognize patients at risk. Staff meetings and rounds were used to identify appropriate candidates—those with limited mobility or cognitive issues, lacking insurance coverage or financial resources, a history of medication noncompliance, or leaving the hospital against medical advice—as well as those prescribed medications with a greater urgency for administration on schedule, such as anticoagulants or antibiotics.
About one-quarter of patients on the unit where this approach was first tested were found to need the service, which involved faxing prescriptions to an outpatient pharmacy across the street from the hospital for either pick-up by the family or delivery to the patient’s hospital room. For those with financial impediments, hospital social workers and case managers explored other options, including the social work department’s discretionary use fund, to pay for the drugs.
Dr. Le believes the project could be replicated in other facilities that lack access to in-house pharmacy services at discharge. She recommends involving social workers and case managers in the planning.
At UCSF, recent EHR implementation has automated the ordering of medications, but the challenge of recognizing who could benefit from extra help in obtaining their discharge medications remains a critical issue for hospitals trying to bring readmissions under control.
For more information about the brown bag medications program, contact Dr. Le at Elizabeth.Le@va.gov.
References
A quality-improvement (QI) project to give high-risk patients ready access to prescribed medications at the time of hospital discharge achieved an 86% success rate, according to an abstract poster presented at HM12 in San Diego last April.1
Lead author Elizabeth Le, MD, then a resident at the University of California at San Francisco Medical Center (UCSF) and now a practicing hospitalist at the Veterans Administration Medical Center in Palo Alto, Calif., says the multidisciplinary “brown bag medications” project involved training house staff to recognize patients at risk. Staff meetings and rounds were used to identify appropriate candidates—those with limited mobility or cognitive issues, lacking insurance coverage or financial resources, a history of medication noncompliance, or leaving the hospital against medical advice—as well as those prescribed medications with a greater urgency for administration on schedule, such as anticoagulants or antibiotics.
About one-quarter of patients on the unit where this approach was first tested were found to need the service, which involved faxing prescriptions to an outpatient pharmacy across the street from the hospital for either pick-up by the family or delivery to the patient’s hospital room. For those with financial impediments, hospital social workers and case managers explored other options, including the social work department’s discretionary use fund, to pay for the drugs.
Dr. Le believes the project could be replicated in other facilities that lack access to in-house pharmacy services at discharge. She recommends involving social workers and case managers in the planning.
At UCSF, recent EHR implementation has automated the ordering of medications, but the challenge of recognizing who could benefit from extra help in obtaining their discharge medications remains a critical issue for hospitals trying to bring readmissions under control.
For more information about the brown bag medications program, contact Dr. Le at Elizabeth.Le@va.gov.
References
A quality-improvement (QI) project to give high-risk patients ready access to prescribed medications at the time of hospital discharge achieved an 86% success rate, according to an abstract poster presented at HM12 in San Diego last April.1
Lead author Elizabeth Le, MD, then a resident at the University of California at San Francisco Medical Center (UCSF) and now a practicing hospitalist at the Veterans Administration Medical Center in Palo Alto, Calif., says the multidisciplinary “brown bag medications” project involved training house staff to recognize patients at risk. Staff meetings and rounds were used to identify appropriate candidates—those with limited mobility or cognitive issues, lacking insurance coverage or financial resources, a history of medication noncompliance, or leaving the hospital against medical advice—as well as those prescribed medications with a greater urgency for administration on schedule, such as anticoagulants or antibiotics.
About one-quarter of patients on the unit where this approach was first tested were found to need the service, which involved faxing prescriptions to an outpatient pharmacy across the street from the hospital for either pick-up by the family or delivery to the patient’s hospital room. For those with financial impediments, hospital social workers and case managers explored other options, including the social work department’s discretionary use fund, to pay for the drugs.
Dr. Le believes the project could be replicated in other facilities that lack access to in-house pharmacy services at discharge. She recommends involving social workers and case managers in the planning.
At UCSF, recent EHR implementation has automated the ordering of medications, but the challenge of recognizing who could benefit from extra help in obtaining their discharge medications remains a critical issue for hospitals trying to bring readmissions under control.
For more information about the brown bag medications program, contact Dr. Le at Elizabeth.Le@va.gov.
References
Bloodsteam Infections in ICU Patients Plummet
Reduction in bloodstream infection rates resulting from a simple intervention: bathing all ICU patients daily with antimicrobial chlorhexidine soap rather than the widely mandated practice of screening ICU patients to determine which ones harbor methicillin-resistant Staphylococcus aureus (MRSA) and then implementing an MRSA treatment protocol for them.
According to data on 75,000 patients at hospitals in 16 states presented in October at the Infectious Diseases Society of America annual meeting, there also was a 37% reduction in patients with MRSA.
Lead researcher Susan Huang, MD, an infectious-disease specialist at the University of California at Irvine, says the results show the benefits of this preventive approach, which included applying an antibiotic ointment to the patient’s nasal passage, and could make ICU screening for drug-resistant organisms, such as MRSA, unnecessary.
Reduction in bloodstream infection rates resulting from a simple intervention: bathing all ICU patients daily with antimicrobial chlorhexidine soap rather than the widely mandated practice of screening ICU patients to determine which ones harbor methicillin-resistant Staphylococcus aureus (MRSA) and then implementing an MRSA treatment protocol for them.
According to data on 75,000 patients at hospitals in 16 states presented in October at the Infectious Diseases Society of America annual meeting, there also was a 37% reduction in patients with MRSA.
Lead researcher Susan Huang, MD, an infectious-disease specialist at the University of California at Irvine, says the results show the benefits of this preventive approach, which included applying an antibiotic ointment to the patient’s nasal passage, and could make ICU screening for drug-resistant organisms, such as MRSA, unnecessary.
Reduction in bloodstream infection rates resulting from a simple intervention: bathing all ICU patients daily with antimicrobial chlorhexidine soap rather than the widely mandated practice of screening ICU patients to determine which ones harbor methicillin-resistant Staphylococcus aureus (MRSA) and then implementing an MRSA treatment protocol for them.
According to data on 75,000 patients at hospitals in 16 states presented in October at the Infectious Diseases Society of America annual meeting, there also was a 37% reduction in patients with MRSA.
Lead researcher Susan Huang, MD, an infectious-disease specialist at the University of California at Irvine, says the results show the benefits of this preventive approach, which included applying an antibiotic ointment to the patient’s nasal passage, and could make ICU screening for drug-resistant organisms, such as MRSA, unnecessary.
Hospitalists Should Consider Fall Risks with Sleep Agent
An author of a new study associating the hypnotic zolpidem (Ambien) with higher rates of patient falls says hospitalists should keep the popular drug’s risks front of mind.
The retrospective cohort study in the Journal of Hospital Medicine, “Zolpidem is Independently Associated with Increased Risk of Inpatient Falls,” found that the rate of falls increased nearly six times among patients taking the sleep agent.1 The research team at the Center for Sleep Medicine at the Mayo Clinic in Rochester, N.Y., calculated one additional fall for every 55 admitted patients who were administered the treatment.
“What this says to me is if one is going to use zolpidem, you have to be aware you’re increasing the risk of fall,” says sleep specialist Timothy Morgenthaler, MD, the Mayo Clinic’s chief patient officer. “Knowledgeable of that, one ought to consider whether there are alternatives or whether the risks outweigh the goal in that setting.”
Dr. Morgenthaler says zolpidem is the most commonly prescribed hypnotic at his hospital, and believes it to be the most common treatment in the U.S. He began studying the issue after nurses reported that it appeared patients were falling after taking the agent. In response to the study, Mayo Clinic removed zolpidem from many of its admission order sets and attempted to help improve patient sleep via other methods, including noise reduction.
“We haven’t removed it from our formulary, and I’m not saying it doesn’t have a role in some points,” he says, “but rather than encouraging it as an option in patients being admitted into the patient, we’re choosing instead now to encourage nonpharmacologic sleep enhancements.”
Visit the-hospitalist.org for more information about HM’s approach to patient falls.
Reference
An author of a new study associating the hypnotic zolpidem (Ambien) with higher rates of patient falls says hospitalists should keep the popular drug’s risks front of mind.
The retrospective cohort study in the Journal of Hospital Medicine, “Zolpidem is Independently Associated with Increased Risk of Inpatient Falls,” found that the rate of falls increased nearly six times among patients taking the sleep agent.1 The research team at the Center for Sleep Medicine at the Mayo Clinic in Rochester, N.Y., calculated one additional fall for every 55 admitted patients who were administered the treatment.
“What this says to me is if one is going to use zolpidem, you have to be aware you’re increasing the risk of fall,” says sleep specialist Timothy Morgenthaler, MD, the Mayo Clinic’s chief patient officer. “Knowledgeable of that, one ought to consider whether there are alternatives or whether the risks outweigh the goal in that setting.”
Dr. Morgenthaler says zolpidem is the most commonly prescribed hypnotic at his hospital, and believes it to be the most common treatment in the U.S. He began studying the issue after nurses reported that it appeared patients were falling after taking the agent. In response to the study, Mayo Clinic removed zolpidem from many of its admission order sets and attempted to help improve patient sleep via other methods, including noise reduction.
“We haven’t removed it from our formulary, and I’m not saying it doesn’t have a role in some points,” he says, “but rather than encouraging it as an option in patients being admitted into the patient, we’re choosing instead now to encourage nonpharmacologic sleep enhancements.”
Visit the-hospitalist.org for more information about HM’s approach to patient falls.
Reference
An author of a new study associating the hypnotic zolpidem (Ambien) with higher rates of patient falls says hospitalists should keep the popular drug’s risks front of mind.
The retrospective cohort study in the Journal of Hospital Medicine, “Zolpidem is Independently Associated with Increased Risk of Inpatient Falls,” found that the rate of falls increased nearly six times among patients taking the sleep agent.1 The research team at the Center for Sleep Medicine at the Mayo Clinic in Rochester, N.Y., calculated one additional fall for every 55 admitted patients who were administered the treatment.
“What this says to me is if one is going to use zolpidem, you have to be aware you’re increasing the risk of fall,” says sleep specialist Timothy Morgenthaler, MD, the Mayo Clinic’s chief patient officer. “Knowledgeable of that, one ought to consider whether there are alternatives or whether the risks outweigh the goal in that setting.”
Dr. Morgenthaler says zolpidem is the most commonly prescribed hypnotic at his hospital, and believes it to be the most common treatment in the U.S. He began studying the issue after nurses reported that it appeared patients were falling after taking the agent. In response to the study, Mayo Clinic removed zolpidem from many of its admission order sets and attempted to help improve patient sleep via other methods, including noise reduction.
“We haven’t removed it from our formulary, and I’m not saying it doesn’t have a role in some points,” he says, “but rather than encouraging it as an option in patients being admitted into the patient, we’re choosing instead now to encourage nonpharmacologic sleep enhancements.”
Visit the-hospitalist.org for more information about HM’s approach to patient falls.