User login
Post-Hospital Syndrome Contributes to Readmission Risk for Elderly
Post-hospital syndrome, as labeled in a recent, widely publicized opinion piece in the New England Journal of Medicine, is not a new concept, according to one hospitalist pioneer.1
Harlan Krumholz, MD, of the Yale School of Medicine in New Haven, Conn., writes in NEJM what others previously have described as “hospitalization-associated disability,” says Mark Williams, MD, MHM, chief of hospital medicine at Northwestern University School of Medicine and principal investigator of SHM’s Project BOOST (www.hospitalmedicine.org/boost).2
Dr. Krumholz found that the majority of 30-day readmissions for elderly patients with heart failure, pneumonia, or chronic obstructive pulmonary disease are for conditions other than the diagnosis named at discharge. He attributes this phenomenon to hospitalization-related sleep deprivation, malnourishment, pain and discomfort, cognition- and physical function-altering medications, deconditioning from bed rest or inactivity, and the experience of confronting stressful, mentally challenging situations in the hospital.1 Such stressors leave elderly patients with post-hospitalization disabilities comparable to a bad case of jet lag.
For Dr. Williams, the physical deterioration leading to rehospitalizations is better attributed to the underlying serious illness and comorbidities experienced by elderly patients—a kind of high-risk, post-illness syndrome. Prior research also has demonstrated the effects of bed rest for hospitalized elderly patients.
Regardless of the origins, is there anything hospitalists can do about this syndrome? “Absolutely,” Dr. Williams says. “Get elderly, hospitalized patients out of bed as quickly as possible, and be mindful of medications and their effects on elderly patients. But most hospitalists already think about these things when managing elderly patients.”
References
Post-hospital syndrome, as labeled in a recent, widely publicized opinion piece in the New England Journal of Medicine, is not a new concept, according to one hospitalist pioneer.1
Harlan Krumholz, MD, of the Yale School of Medicine in New Haven, Conn., writes in NEJM what others previously have described as “hospitalization-associated disability,” says Mark Williams, MD, MHM, chief of hospital medicine at Northwestern University School of Medicine and principal investigator of SHM’s Project BOOST (www.hospitalmedicine.org/boost).2
Dr. Krumholz found that the majority of 30-day readmissions for elderly patients with heart failure, pneumonia, or chronic obstructive pulmonary disease are for conditions other than the diagnosis named at discharge. He attributes this phenomenon to hospitalization-related sleep deprivation, malnourishment, pain and discomfort, cognition- and physical function-altering medications, deconditioning from bed rest or inactivity, and the experience of confronting stressful, mentally challenging situations in the hospital.1 Such stressors leave elderly patients with post-hospitalization disabilities comparable to a bad case of jet lag.
For Dr. Williams, the physical deterioration leading to rehospitalizations is better attributed to the underlying serious illness and comorbidities experienced by elderly patients—a kind of high-risk, post-illness syndrome. Prior research also has demonstrated the effects of bed rest for hospitalized elderly patients.
Regardless of the origins, is there anything hospitalists can do about this syndrome? “Absolutely,” Dr. Williams says. “Get elderly, hospitalized patients out of bed as quickly as possible, and be mindful of medications and their effects on elderly patients. But most hospitalists already think about these things when managing elderly patients.”
References
Post-hospital syndrome, as labeled in a recent, widely publicized opinion piece in the New England Journal of Medicine, is not a new concept, according to one hospitalist pioneer.1
Harlan Krumholz, MD, of the Yale School of Medicine in New Haven, Conn., writes in NEJM what others previously have described as “hospitalization-associated disability,” says Mark Williams, MD, MHM, chief of hospital medicine at Northwestern University School of Medicine and principal investigator of SHM’s Project BOOST (www.hospitalmedicine.org/boost).2
Dr. Krumholz found that the majority of 30-day readmissions for elderly patients with heart failure, pneumonia, or chronic obstructive pulmonary disease are for conditions other than the diagnosis named at discharge. He attributes this phenomenon to hospitalization-related sleep deprivation, malnourishment, pain and discomfort, cognition- and physical function-altering medications, deconditioning from bed rest or inactivity, and the experience of confronting stressful, mentally challenging situations in the hospital.1 Such stressors leave elderly patients with post-hospitalization disabilities comparable to a bad case of jet lag.
For Dr. Williams, the physical deterioration leading to rehospitalizations is better attributed to the underlying serious illness and comorbidities experienced by elderly patients—a kind of high-risk, post-illness syndrome. Prior research also has demonstrated the effects of bed rest for hospitalized elderly patients.
Regardless of the origins, is there anything hospitalists can do about this syndrome? “Absolutely,” Dr. Williams says. “Get elderly, hospitalized patients out of bed as quickly as possible, and be mindful of medications and their effects on elderly patients. But most hospitalists already think about these things when managing elderly patients.”
References
How Should Physicians Assess and Manage Pressure Ulcers in the Hospitalized Patient?
The Case
An 85-year-old woman with stroke, functional quadriplegia, and diabetes mellitus presents with altered mental status. She is febrile (38.5°C) with leukocytosis (14,400 cells/mm3) and has a 5 cm x 4 cm x 2 cm Stage III malodorous sacral ulcer without surrounding erythema, tunneling, or pain. The ulcer base is partially covered by green slough. How should this pressure ulcer be evaluated and treated?
Overview
Pressure ulcers in vulnerable populations, such as the elderly and those with limited mobility, are exceedingly common. In the acute-care setting, the incidence of pressure ulcers ranges from 0.4% to 38%, with 2.5 million cases treated annually at an estimated cost of $11 billion per year.1,2 Moreover, as of Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) guideline states that hospitals will no longer receive additional payment when a hospitalized patient develops Stage III or IV pressure ulcers that are not present on admission.
A pressure ulcer is a localized injury to skin and underlying soft tissue over a bony prominence due to sustained external pressure.3 Prolonged pressure on these weight-bearing areas leads to reduced blood flow, ischemia, cell death, and necrosis of local tissues.4 Risk factors for developing pressure ulcers include increased external pressure, shear, friction, moisture, poor perfusion, immobility, incontinence, malnutrition, and impaired mental status.4 Inadequately treated pressure ulcers can lead to pain, tunneling, fistula formation, disfigurement, infection, prolonged hospitalization, lower quality of life, and increased mortality.4
Because of the significant morbidities and high costs associated with the care of pressure ulcers in acute care, hospitalists must be familiar with the assessment and treatment of pressure ulcers in vulnerable patients.
Review of the Data
The management of pressure ulcers in the hospitalized patient starts with a comprehensive assessment of the patient’s medical comorbidities, risk factors, and wound-staging. Considerations must be given to differentiate an infected pressure ulcer from a noninfected ulcer. These evaluations then guide the appropriate treatments of pressure ulcers, including the prevention of progression or formation of new ulcers, debridement, application of wound dressing, and antibiotic use.
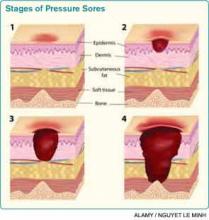
Assessing pressure ulcer stage. The National Pressure Ulcer Advisory Panel (NPUAP) Classification System is the most commonly used staging tool. It describes four stages of pressure ulcers (see Table 1).3 A Stage 1 pressure ulcer is characterized by intact skin with nonblanchable erythema and may be discolored, painful, soft, firm, and warmer or cooler compared to adjacent area. A Stage II pressure ulcer presents with partial thickness skin loss with a shallow red-pink wound bed without slough, or as an intact or ruptured serum-filled blister. Stage II pressure ulcers do not include skin tears, tape burns, macerations, or excoriations. A Stage III pressure ulcer has full thickness skin loss with or without visible subcutaneous fat. Bone, tendon, or muscle are not exposed or directly palpable. Slough may be present but it does not obscure the depth of ulcer. Deep ulcers can develop in anatomical regions with high adiposity, such as the pelvic girdle. A Stage IV pressure ulcer has full thickness tissue loss with exposed and palpable bone, tendon, or muscle. Slough, eschar, undermining, and tunneling may be present. The depth of a Stage IV ulcer varies depending on anatomical location and adiposity. Stage IV ulcers also create a nidus for osteomyelitis.
NPUAP describes two additional categories of pressure ulcers: unstageable and deep tissue injury.3 An unstageable ulcer has full thickness skin or tissue loss of unknown depth because the wound base is completely obscured by slough or eschar. The ulcer can only be accurately categorized as Stage III or IV after sufficient slough or eschar is removed to identify wound depth. Lastly, suspected deep tissue injury describes a localized area of discolored intact skin (purple or maroon) or blood-filled blister due to damage of underlying tissue from pressure or shear.
Diagnosing infected pressure ulcers. Pressure ulcer infection delays wound healing and increases risks for sepsis, cellulitis, osteomyelitis, and death.5,6 Clinical evidence of soft tissue involvement, such as erythema, warmth, tenderness, foul odor, or purulent discharge, and systemic inflammatory response (fever, tachycardia, or leukocytosis) are suggestive of a wound infection.3,5 However, these clinical signs may be absent and thus make the distinction between chronic wound and infected pressure ulcer difficult.7 Delayed healing with friable granulation tissue and increased pain in a treated wound may be the only signs of a pressure ulcer infection.3,5,7
Routine laboratory tests (i.e. white blood cell count, C-reactive protein, and erythrocyte sedimentation rate) are neither sensitive nor specific in diagnosing wound infection. Moreover, because pressure ulcers are typically colonized with ≥105 organisms/mL of normal skin flora and bacteria from adjacent gastrointestinal or urogenital environments, swab cultures identify colonizing organisms and are not recommended as a diagnostic test for pressure ulcer microbiologic evaluation.5,6 If microbiological data are needed to guide antibiotic use, cultures of blood, bone, or deep tissue biopsied from a surgically debrided wound should be used.5 Importantly, a higher index of suspicion should be maintained for infection of Stage III or IV pressure ulcers because they are more commonly infected than Stage I or II ulcers.3
Prevention. The prevention of wound progression is essential in treating acute, chronic, or infected pressure ulcers. Although management guidelines are limited by few high-quality, randomized controlled trials, NPUAP recommends a number of prevention strategies targeting risk factors that contribute to pressure ulcer development.2,3,8
For all bed-bound and chair-bound persons with impaired ability to self-reposition, risk assessment for pressure ulcer should be done on admission and repeated every 24 hours. The presence of such risk factors as immobility, shear, friction, moisture, incontinence, and malnutrition should be used to guide preventive treatments. Pressure relief on an ulcer can be achieved by repositioning the immobile patient at one- to two-hour intervals. Pressure-redistributing support surfaces (static, overlays, or dynamic) reduce tissue pressure and decrease overall incidence of pressure ulcers. Due to a lack of relative efficacy data, the selection of a support surface should be determined by the patient’s individual needs in order to reduce pressure and shear.3 For instance, dynamic support is an appropriate surface for an immobile patient with multiple or nonhealing ulcers. Shearing force and friction can be reduced by limiting head-of-bed elevation to <30° and using such transfer aids as bed linens while repositioning patients. The use of pillows, foam wedges, or other devices should be used to eliminate direct contact of bony prominences or reduce pressure on heels.8
Skin care should be optimized to limit excessive dryness or moisture. This includes using moisturizers for dry skin, particularly for the sacrum, and implementing bowel and bladder programs and absorbent underpads in patients with bowel or bladder incontinence.2 Given that patients with pressure ulcers are in a catabolic state, those who are nutritionally compromised may benefit from nutritional supplementation.3 Lastly, appropriate use of local and systemic pain regimen for painful pressure ulcers can improve patient cooperation in repositioning, dressing change, and quality of life.
Debridement. Wound debridement removes necrotic tissue often present in infected or chronic pressure ulcers, reduces risk for further infection, and promotes granulation tissue formation and wound healing. Debridement, however, is not indicated for ulcers of an ischemic limb or dry eschar of the heel, due to propensity for complications.3,4 The five common debridement methods are sharp, mechanical, autolytic, enzymatic, and biosurgical. The debridement method of choice is determined by clinician preference and availability.4
Sharp debridement results in rapid removal of large amounts of nonviable necrotic tissues and eschar using sharp instruments and, therefore, is indicated if wound infection or sepsis is present. Mechanical debridement by wet-to-dry dressing or whirlpool nonselectively removes granulation tissue and, thus, should be used cautiously. Autolytic debridement uses occlusive dressings (i.e. hydrocolloid or hydrogel) to maintain a moist wound environment in order to optimize the body’s inherent ability to selectively self-digest necrotic tissues. Enzymatic debridement with concentrated topical proteolytic enzymes (i.e. collagenase) digests necrotic tissues and achieves faster debridement than autolysis while being less invasive than surgical intervention. Biosurgery utilizes maggots (i.e. Lucilia sericata) that produce enzymes to effectively debride necrotic tissues.
Wound care and dressing. Pressure ulcers should be cleansed with each dressing change using such physiologic solutions as normal saline. Cleansing with antimicrobial solutions for ulcers with large necrotic debris or infection needs to be thoughtfully administered due to the potential impairment on wound healing.4 Wound dressing should maintain a moist wound environment to allow epithelialization and limit excessive exudates in order to prevent maceration. Although there are many categories of moisture retentive dressings, their comparative effectiveness remain unclear.4 Table 2 summarizes characteristics of common wound dressings and their applications.
Antibiotic use. Topical antibiotics are appropriate for Stage III or IV ulcers with signs of local infection, including periwound erythema and friable granulation tissue.4 The Agency for Health Care Policy and Research recommends a two-week trial of a topical antibiotic, such as silver sulfadiazine, for pressure ulcers that fail to heal after two to four weeks of optimal care.6 Systemic antibiotics should be used for patients who demonstrate evidence of systemic infection including sepsis, osteomyelitis, or cellulitis with associated fever and leukocytosis. The choice of systemic antibiotics should be based on cultures from blood, bone, or deep tissue biopsied from a surgically debrided wound.4,6
Back to the Case
The patient was hospitalized for altered mental status. She was at high risk for the progression of her sacral ulcer and the development of new pressure ulcers due to immobility, incontinence, malnutrition, and impaired mental status. The sacral wound was a chronic, Stage III pressure ulcer without evidence of local tissue infection. However, the presence of leukocytosis and fever were suggestive of an underlying infection. Her urine analysis was consistent with a urinary tract infection.
Trimethoprim/sulfamethoxazole was administered with subsequent resolution of leukocytosis, fever, and delirium. The sacral ulcer was cleansed with normal saline and covered with hydrocolloid dressing every 72 hours in order to maintain a moist wound environment and facilitate autolysis. Preventive interventions guided by her risk factors for pressure ulcer were implemented. Interventions included:
- Daily skin and wound assessment;
- Pressure relief with repositioning every two hours;
- Use of a dynamic support surface;
- Head-of-bed elevation of no more than <30° to reduce shear and friction;
- Use of transfer aids;
- Use of devices to eliminate direct contact of bony prominences;
- Optimizing skin care with moisturizers for dry skin and frequent changing of absorbent under pads for incontinence; and
- Consulting nutrition service to optimize nutritional intake.
Bottom Line
Assessments of pressure ulcer stage, wound infection, and risk factors guide targeted therapeutic interventions that facilitate wound healing and prevent new pressure ulcer formation.
Dr. Prager is a fellow in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai School of Medicine in New York City. Dr. Ko is a hospitalist and an assistant professor in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai.
References
- Pressure ulcers in America: prevalence, incidence, and implications for the future. An executive summary of the National Pressure Ulcer Advisory Panel monograph. Adv Skin Wound Care. 2001;14(4):208-215.
- Reddy M, Gill SS, Rochon PA. Preventing pressure ulcers: a systematic review. JAMA. 2006;296(8):974-984.
- European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Treatment of Pressure Ulcers: Quick Reference Guide. Washington, D.C.: National Pressure Ulcer Advisory Panel; 2009.
- Bates-Jensen BM. Chapter 58. Pressure Ulcers. In: Halter JB, Ouslander JG, Tinetti ME, Studenski S, High KP, Asthana S, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York: McGraw-Hill; 2009.
- Livesley NJ, Chow AW. Infected pressure ulcers in elderly individuals. Clin Infect Dis. 2002;35(11):1390-1396.
- Agency for Health Care Policy and Research (AHCPR). Treatment of Pressure Ulcers. Clinical Practice Guideline Number 15. U.S. Department of Health and Human Services. 1994.
- Reddy M, Gill SS, Wu W, Kalkar SR, Rochon PA. Does this patient have an infection of a chronic wound? JAMA. 2012;307(6):605-611.
- National Pressure Ulcer Advisory Panel. Pressure Ulcer Prevention Points. National Pressure Ulcer Advisory Panel website. Available at: http://www.npuap.org/resources/educational-and-clinical-resources/pressure-ulcer-prevention-points/. Accessed Aug. 1, 2012.
- Reuben DB, Herr KA, Pacala JT, et al. Skin Ulcers. In: Geriatrics At Your Fingertips. 12th ed. New York: The American Geriatrics Society; 2010.
The Case
An 85-year-old woman with stroke, functional quadriplegia, and diabetes mellitus presents with altered mental status. She is febrile (38.5°C) with leukocytosis (14,400 cells/mm3) and has a 5 cm x 4 cm x 2 cm Stage III malodorous sacral ulcer without surrounding erythema, tunneling, or pain. The ulcer base is partially covered by green slough. How should this pressure ulcer be evaluated and treated?
Overview
Pressure ulcers in vulnerable populations, such as the elderly and those with limited mobility, are exceedingly common. In the acute-care setting, the incidence of pressure ulcers ranges from 0.4% to 38%, with 2.5 million cases treated annually at an estimated cost of $11 billion per year.1,2 Moreover, as of Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) guideline states that hospitals will no longer receive additional payment when a hospitalized patient develops Stage III or IV pressure ulcers that are not present on admission.
A pressure ulcer is a localized injury to skin and underlying soft tissue over a bony prominence due to sustained external pressure.3 Prolonged pressure on these weight-bearing areas leads to reduced blood flow, ischemia, cell death, and necrosis of local tissues.4 Risk factors for developing pressure ulcers include increased external pressure, shear, friction, moisture, poor perfusion, immobility, incontinence, malnutrition, and impaired mental status.4 Inadequately treated pressure ulcers can lead to pain, tunneling, fistula formation, disfigurement, infection, prolonged hospitalization, lower quality of life, and increased mortality.4
Because of the significant morbidities and high costs associated with the care of pressure ulcers in acute care, hospitalists must be familiar with the assessment and treatment of pressure ulcers in vulnerable patients.
Review of the Data
The management of pressure ulcers in the hospitalized patient starts with a comprehensive assessment of the patient’s medical comorbidities, risk factors, and wound-staging. Considerations must be given to differentiate an infected pressure ulcer from a noninfected ulcer. These evaluations then guide the appropriate treatments of pressure ulcers, including the prevention of progression or formation of new ulcers, debridement, application of wound dressing, and antibiotic use.
Assessing pressure ulcer stage. The National Pressure Ulcer Advisory Panel (NPUAP) Classification System is the most commonly used staging tool. It describes four stages of pressure ulcers (see Table 1).3 A Stage 1 pressure ulcer is characterized by intact skin with nonblanchable erythema and may be discolored, painful, soft, firm, and warmer or cooler compared to adjacent area. A Stage II pressure ulcer presents with partial thickness skin loss with a shallow red-pink wound bed without slough, or as an intact or ruptured serum-filled blister. Stage II pressure ulcers do not include skin tears, tape burns, macerations, or excoriations. A Stage III pressure ulcer has full thickness skin loss with or without visible subcutaneous fat. Bone, tendon, or muscle are not exposed or directly palpable. Slough may be present but it does not obscure the depth of ulcer. Deep ulcers can develop in anatomical regions with high adiposity, such as the pelvic girdle. A Stage IV pressure ulcer has full thickness tissue loss with exposed and palpable bone, tendon, or muscle. Slough, eschar, undermining, and tunneling may be present. The depth of a Stage IV ulcer varies depending on anatomical location and adiposity. Stage IV ulcers also create a nidus for osteomyelitis.
NPUAP describes two additional categories of pressure ulcers: unstageable and deep tissue injury.3 An unstageable ulcer has full thickness skin or tissue loss of unknown depth because the wound base is completely obscured by slough or eschar. The ulcer can only be accurately categorized as Stage III or IV after sufficient slough or eschar is removed to identify wound depth. Lastly, suspected deep tissue injury describes a localized area of discolored intact skin (purple or maroon) or blood-filled blister due to damage of underlying tissue from pressure or shear.
Diagnosing infected pressure ulcers. Pressure ulcer infection delays wound healing and increases risks for sepsis, cellulitis, osteomyelitis, and death.5,6 Clinical evidence of soft tissue involvement, such as erythema, warmth, tenderness, foul odor, or purulent discharge, and systemic inflammatory response (fever, tachycardia, or leukocytosis) are suggestive of a wound infection.3,5 However, these clinical signs may be absent and thus make the distinction between chronic wound and infected pressure ulcer difficult.7 Delayed healing with friable granulation tissue and increased pain in a treated wound may be the only signs of a pressure ulcer infection.3,5,7
Routine laboratory tests (i.e. white blood cell count, C-reactive protein, and erythrocyte sedimentation rate) are neither sensitive nor specific in diagnosing wound infection. Moreover, because pressure ulcers are typically colonized with ≥105 organisms/mL of normal skin flora and bacteria from adjacent gastrointestinal or urogenital environments, swab cultures identify colonizing organisms and are not recommended as a diagnostic test for pressure ulcer microbiologic evaluation.5,6 If microbiological data are needed to guide antibiotic use, cultures of blood, bone, or deep tissue biopsied from a surgically debrided wound should be used.5 Importantly, a higher index of suspicion should be maintained for infection of Stage III or IV pressure ulcers because they are more commonly infected than Stage I or II ulcers.3
Prevention. The prevention of wound progression is essential in treating acute, chronic, or infected pressure ulcers. Although management guidelines are limited by few high-quality, randomized controlled trials, NPUAP recommends a number of prevention strategies targeting risk factors that contribute to pressure ulcer development.2,3,8
For all bed-bound and chair-bound persons with impaired ability to self-reposition, risk assessment for pressure ulcer should be done on admission and repeated every 24 hours. The presence of such risk factors as immobility, shear, friction, moisture, incontinence, and malnutrition should be used to guide preventive treatments. Pressure relief on an ulcer can be achieved by repositioning the immobile patient at one- to two-hour intervals. Pressure-redistributing support surfaces (static, overlays, or dynamic) reduce tissue pressure and decrease overall incidence of pressure ulcers. Due to a lack of relative efficacy data, the selection of a support surface should be determined by the patient’s individual needs in order to reduce pressure and shear.3 For instance, dynamic support is an appropriate surface for an immobile patient with multiple or nonhealing ulcers. Shearing force and friction can be reduced by limiting head-of-bed elevation to <30° and using such transfer aids as bed linens while repositioning patients. The use of pillows, foam wedges, or other devices should be used to eliminate direct contact of bony prominences or reduce pressure on heels.8
Skin care should be optimized to limit excessive dryness or moisture. This includes using moisturizers for dry skin, particularly for the sacrum, and implementing bowel and bladder programs and absorbent underpads in patients with bowel or bladder incontinence.2 Given that patients with pressure ulcers are in a catabolic state, those who are nutritionally compromised may benefit from nutritional supplementation.3 Lastly, appropriate use of local and systemic pain regimen for painful pressure ulcers can improve patient cooperation in repositioning, dressing change, and quality of life.
Debridement. Wound debridement removes necrotic tissue often present in infected or chronic pressure ulcers, reduces risk for further infection, and promotes granulation tissue formation and wound healing. Debridement, however, is not indicated for ulcers of an ischemic limb or dry eschar of the heel, due to propensity for complications.3,4 The five common debridement methods are sharp, mechanical, autolytic, enzymatic, and biosurgical. The debridement method of choice is determined by clinician preference and availability.4
Sharp debridement results in rapid removal of large amounts of nonviable necrotic tissues and eschar using sharp instruments and, therefore, is indicated if wound infection or sepsis is present. Mechanical debridement by wet-to-dry dressing or whirlpool nonselectively removes granulation tissue and, thus, should be used cautiously. Autolytic debridement uses occlusive dressings (i.e. hydrocolloid or hydrogel) to maintain a moist wound environment in order to optimize the body’s inherent ability to selectively self-digest necrotic tissues. Enzymatic debridement with concentrated topical proteolytic enzymes (i.e. collagenase) digests necrotic tissues and achieves faster debridement than autolysis while being less invasive than surgical intervention. Biosurgery utilizes maggots (i.e. Lucilia sericata) that produce enzymes to effectively debride necrotic tissues.
Wound care and dressing. Pressure ulcers should be cleansed with each dressing change using such physiologic solutions as normal saline. Cleansing with antimicrobial solutions for ulcers with large necrotic debris or infection needs to be thoughtfully administered due to the potential impairment on wound healing.4 Wound dressing should maintain a moist wound environment to allow epithelialization and limit excessive exudates in order to prevent maceration. Although there are many categories of moisture retentive dressings, their comparative effectiveness remain unclear.4 Table 2 summarizes characteristics of common wound dressings and their applications.
Antibiotic use. Topical antibiotics are appropriate for Stage III or IV ulcers with signs of local infection, including periwound erythema and friable granulation tissue.4 The Agency for Health Care Policy and Research recommends a two-week trial of a topical antibiotic, such as silver sulfadiazine, for pressure ulcers that fail to heal after two to four weeks of optimal care.6 Systemic antibiotics should be used for patients who demonstrate evidence of systemic infection including sepsis, osteomyelitis, or cellulitis with associated fever and leukocytosis. The choice of systemic antibiotics should be based on cultures from blood, bone, or deep tissue biopsied from a surgically debrided wound.4,6
Back to the Case
The patient was hospitalized for altered mental status. She was at high risk for the progression of her sacral ulcer and the development of new pressure ulcers due to immobility, incontinence, malnutrition, and impaired mental status. The sacral wound was a chronic, Stage III pressure ulcer without evidence of local tissue infection. However, the presence of leukocytosis and fever were suggestive of an underlying infection. Her urine analysis was consistent with a urinary tract infection.
Trimethoprim/sulfamethoxazole was administered with subsequent resolution of leukocytosis, fever, and delirium. The sacral ulcer was cleansed with normal saline and covered with hydrocolloid dressing every 72 hours in order to maintain a moist wound environment and facilitate autolysis. Preventive interventions guided by her risk factors for pressure ulcer were implemented. Interventions included:
- Daily skin and wound assessment;
- Pressure relief with repositioning every two hours;
- Use of a dynamic support surface;
- Head-of-bed elevation of no more than <30° to reduce shear and friction;
- Use of transfer aids;
- Use of devices to eliminate direct contact of bony prominences;
- Optimizing skin care with moisturizers for dry skin and frequent changing of absorbent under pads for incontinence; and
- Consulting nutrition service to optimize nutritional intake.
Bottom Line
Assessments of pressure ulcer stage, wound infection, and risk factors guide targeted therapeutic interventions that facilitate wound healing and prevent new pressure ulcer formation.
Dr. Prager is a fellow in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai School of Medicine in New York City. Dr. Ko is a hospitalist and an assistant professor in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai.
References
- Pressure ulcers in America: prevalence, incidence, and implications for the future. An executive summary of the National Pressure Ulcer Advisory Panel monograph. Adv Skin Wound Care. 2001;14(4):208-215.
- Reddy M, Gill SS, Rochon PA. Preventing pressure ulcers: a systematic review. JAMA. 2006;296(8):974-984.
- European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Treatment of Pressure Ulcers: Quick Reference Guide. Washington, D.C.: National Pressure Ulcer Advisory Panel; 2009.
- Bates-Jensen BM. Chapter 58. Pressure Ulcers. In: Halter JB, Ouslander JG, Tinetti ME, Studenski S, High KP, Asthana S, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York: McGraw-Hill; 2009.
- Livesley NJ, Chow AW. Infected pressure ulcers in elderly individuals. Clin Infect Dis. 2002;35(11):1390-1396.
- Agency for Health Care Policy and Research (AHCPR). Treatment of Pressure Ulcers. Clinical Practice Guideline Number 15. U.S. Department of Health and Human Services. 1994.
- Reddy M, Gill SS, Wu W, Kalkar SR, Rochon PA. Does this patient have an infection of a chronic wound? JAMA. 2012;307(6):605-611.
- National Pressure Ulcer Advisory Panel. Pressure Ulcer Prevention Points. National Pressure Ulcer Advisory Panel website. Available at: http://www.npuap.org/resources/educational-and-clinical-resources/pressure-ulcer-prevention-points/. Accessed Aug. 1, 2012.
- Reuben DB, Herr KA, Pacala JT, et al. Skin Ulcers. In: Geriatrics At Your Fingertips. 12th ed. New York: The American Geriatrics Society; 2010.
The Case
An 85-year-old woman with stroke, functional quadriplegia, and diabetes mellitus presents with altered mental status. She is febrile (38.5°C) with leukocytosis (14,400 cells/mm3) and has a 5 cm x 4 cm x 2 cm Stage III malodorous sacral ulcer without surrounding erythema, tunneling, or pain. The ulcer base is partially covered by green slough. How should this pressure ulcer be evaluated and treated?
Overview
Pressure ulcers in vulnerable populations, such as the elderly and those with limited mobility, are exceedingly common. In the acute-care setting, the incidence of pressure ulcers ranges from 0.4% to 38%, with 2.5 million cases treated annually at an estimated cost of $11 billion per year.1,2 Moreover, as of Oct. 1, 2008, the Centers for Medicare & Medicaid Services (CMS) guideline states that hospitals will no longer receive additional payment when a hospitalized patient develops Stage III or IV pressure ulcers that are not present on admission.
A pressure ulcer is a localized injury to skin and underlying soft tissue over a bony prominence due to sustained external pressure.3 Prolonged pressure on these weight-bearing areas leads to reduced blood flow, ischemia, cell death, and necrosis of local tissues.4 Risk factors for developing pressure ulcers include increased external pressure, shear, friction, moisture, poor perfusion, immobility, incontinence, malnutrition, and impaired mental status.4 Inadequately treated pressure ulcers can lead to pain, tunneling, fistula formation, disfigurement, infection, prolonged hospitalization, lower quality of life, and increased mortality.4
Because of the significant morbidities and high costs associated with the care of pressure ulcers in acute care, hospitalists must be familiar with the assessment and treatment of pressure ulcers in vulnerable patients.
Review of the Data
The management of pressure ulcers in the hospitalized patient starts with a comprehensive assessment of the patient’s medical comorbidities, risk factors, and wound-staging. Considerations must be given to differentiate an infected pressure ulcer from a noninfected ulcer. These evaluations then guide the appropriate treatments of pressure ulcers, including the prevention of progression or formation of new ulcers, debridement, application of wound dressing, and antibiotic use.
Assessing pressure ulcer stage. The National Pressure Ulcer Advisory Panel (NPUAP) Classification System is the most commonly used staging tool. It describes four stages of pressure ulcers (see Table 1).3 A Stage 1 pressure ulcer is characterized by intact skin with nonblanchable erythema and may be discolored, painful, soft, firm, and warmer or cooler compared to adjacent area. A Stage II pressure ulcer presents with partial thickness skin loss with a shallow red-pink wound bed without slough, or as an intact or ruptured serum-filled blister. Stage II pressure ulcers do not include skin tears, tape burns, macerations, or excoriations. A Stage III pressure ulcer has full thickness skin loss with or without visible subcutaneous fat. Bone, tendon, or muscle are not exposed or directly palpable. Slough may be present but it does not obscure the depth of ulcer. Deep ulcers can develop in anatomical regions with high adiposity, such as the pelvic girdle. A Stage IV pressure ulcer has full thickness tissue loss with exposed and palpable bone, tendon, or muscle. Slough, eschar, undermining, and tunneling may be present. The depth of a Stage IV ulcer varies depending on anatomical location and adiposity. Stage IV ulcers also create a nidus for osteomyelitis.
NPUAP describes two additional categories of pressure ulcers: unstageable and deep tissue injury.3 An unstageable ulcer has full thickness skin or tissue loss of unknown depth because the wound base is completely obscured by slough or eschar. The ulcer can only be accurately categorized as Stage III or IV after sufficient slough or eschar is removed to identify wound depth. Lastly, suspected deep tissue injury describes a localized area of discolored intact skin (purple or maroon) or blood-filled blister due to damage of underlying tissue from pressure or shear.
Diagnosing infected pressure ulcers. Pressure ulcer infection delays wound healing and increases risks for sepsis, cellulitis, osteomyelitis, and death.5,6 Clinical evidence of soft tissue involvement, such as erythema, warmth, tenderness, foul odor, or purulent discharge, and systemic inflammatory response (fever, tachycardia, or leukocytosis) are suggestive of a wound infection.3,5 However, these clinical signs may be absent and thus make the distinction between chronic wound and infected pressure ulcer difficult.7 Delayed healing with friable granulation tissue and increased pain in a treated wound may be the only signs of a pressure ulcer infection.3,5,7
Routine laboratory tests (i.e. white blood cell count, C-reactive protein, and erythrocyte sedimentation rate) are neither sensitive nor specific in diagnosing wound infection. Moreover, because pressure ulcers are typically colonized with ≥105 organisms/mL of normal skin flora and bacteria from adjacent gastrointestinal or urogenital environments, swab cultures identify colonizing organisms and are not recommended as a diagnostic test for pressure ulcer microbiologic evaluation.5,6 If microbiological data are needed to guide antibiotic use, cultures of blood, bone, or deep tissue biopsied from a surgically debrided wound should be used.5 Importantly, a higher index of suspicion should be maintained for infection of Stage III or IV pressure ulcers because they are more commonly infected than Stage I or II ulcers.3
Prevention. The prevention of wound progression is essential in treating acute, chronic, or infected pressure ulcers. Although management guidelines are limited by few high-quality, randomized controlled trials, NPUAP recommends a number of prevention strategies targeting risk factors that contribute to pressure ulcer development.2,3,8
For all bed-bound and chair-bound persons with impaired ability to self-reposition, risk assessment for pressure ulcer should be done on admission and repeated every 24 hours. The presence of such risk factors as immobility, shear, friction, moisture, incontinence, and malnutrition should be used to guide preventive treatments. Pressure relief on an ulcer can be achieved by repositioning the immobile patient at one- to two-hour intervals. Pressure-redistributing support surfaces (static, overlays, or dynamic) reduce tissue pressure and decrease overall incidence of pressure ulcers. Due to a lack of relative efficacy data, the selection of a support surface should be determined by the patient’s individual needs in order to reduce pressure and shear.3 For instance, dynamic support is an appropriate surface for an immobile patient with multiple or nonhealing ulcers. Shearing force and friction can be reduced by limiting head-of-bed elevation to <30° and using such transfer aids as bed linens while repositioning patients. The use of pillows, foam wedges, or other devices should be used to eliminate direct contact of bony prominences or reduce pressure on heels.8
Skin care should be optimized to limit excessive dryness or moisture. This includes using moisturizers for dry skin, particularly for the sacrum, and implementing bowel and bladder programs and absorbent underpads in patients with bowel or bladder incontinence.2 Given that patients with pressure ulcers are in a catabolic state, those who are nutritionally compromised may benefit from nutritional supplementation.3 Lastly, appropriate use of local and systemic pain regimen for painful pressure ulcers can improve patient cooperation in repositioning, dressing change, and quality of life.
Debridement. Wound debridement removes necrotic tissue often present in infected or chronic pressure ulcers, reduces risk for further infection, and promotes granulation tissue formation and wound healing. Debridement, however, is not indicated for ulcers of an ischemic limb or dry eschar of the heel, due to propensity for complications.3,4 The five common debridement methods are sharp, mechanical, autolytic, enzymatic, and biosurgical. The debridement method of choice is determined by clinician preference and availability.4
Sharp debridement results in rapid removal of large amounts of nonviable necrotic tissues and eschar using sharp instruments and, therefore, is indicated if wound infection or sepsis is present. Mechanical debridement by wet-to-dry dressing or whirlpool nonselectively removes granulation tissue and, thus, should be used cautiously. Autolytic debridement uses occlusive dressings (i.e. hydrocolloid or hydrogel) to maintain a moist wound environment in order to optimize the body’s inherent ability to selectively self-digest necrotic tissues. Enzymatic debridement with concentrated topical proteolytic enzymes (i.e. collagenase) digests necrotic tissues and achieves faster debridement than autolysis while being less invasive than surgical intervention. Biosurgery utilizes maggots (i.e. Lucilia sericata) that produce enzymes to effectively debride necrotic tissues.
Wound care and dressing. Pressure ulcers should be cleansed with each dressing change using such physiologic solutions as normal saline. Cleansing with antimicrobial solutions for ulcers with large necrotic debris or infection needs to be thoughtfully administered due to the potential impairment on wound healing.4 Wound dressing should maintain a moist wound environment to allow epithelialization and limit excessive exudates in order to prevent maceration. Although there are many categories of moisture retentive dressings, their comparative effectiveness remain unclear.4 Table 2 summarizes characteristics of common wound dressings and their applications.
Antibiotic use. Topical antibiotics are appropriate for Stage III or IV ulcers with signs of local infection, including periwound erythema and friable granulation tissue.4 The Agency for Health Care Policy and Research recommends a two-week trial of a topical antibiotic, such as silver sulfadiazine, for pressure ulcers that fail to heal after two to four weeks of optimal care.6 Systemic antibiotics should be used for patients who demonstrate evidence of systemic infection including sepsis, osteomyelitis, or cellulitis with associated fever and leukocytosis. The choice of systemic antibiotics should be based on cultures from blood, bone, or deep tissue biopsied from a surgically debrided wound.4,6
Back to the Case
The patient was hospitalized for altered mental status. She was at high risk for the progression of her sacral ulcer and the development of new pressure ulcers due to immobility, incontinence, malnutrition, and impaired mental status. The sacral wound was a chronic, Stage III pressure ulcer without evidence of local tissue infection. However, the presence of leukocytosis and fever were suggestive of an underlying infection. Her urine analysis was consistent with a urinary tract infection.
Trimethoprim/sulfamethoxazole was administered with subsequent resolution of leukocytosis, fever, and delirium. The sacral ulcer was cleansed with normal saline and covered with hydrocolloid dressing every 72 hours in order to maintain a moist wound environment and facilitate autolysis. Preventive interventions guided by her risk factors for pressure ulcer were implemented. Interventions included:
- Daily skin and wound assessment;
- Pressure relief with repositioning every two hours;
- Use of a dynamic support surface;
- Head-of-bed elevation of no more than <30° to reduce shear and friction;
- Use of transfer aids;
- Use of devices to eliminate direct contact of bony prominences;
- Optimizing skin care with moisturizers for dry skin and frequent changing of absorbent under pads for incontinence; and
- Consulting nutrition service to optimize nutritional intake.
Bottom Line
Assessments of pressure ulcer stage, wound infection, and risk factors guide targeted therapeutic interventions that facilitate wound healing and prevent new pressure ulcer formation.
Dr. Prager is a fellow in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai School of Medicine in New York City. Dr. Ko is a hospitalist and an assistant professor in the Brookdale Department of Geriatrics and Palliative Medicine at Mount Sinai.
References
- Pressure ulcers in America: prevalence, incidence, and implications for the future. An executive summary of the National Pressure Ulcer Advisory Panel monograph. Adv Skin Wound Care. 2001;14(4):208-215.
- Reddy M, Gill SS, Rochon PA. Preventing pressure ulcers: a systematic review. JAMA. 2006;296(8):974-984.
- European Pressure Ulcer Advisory Panel and National Pressure Ulcer Advisory Panel. Treatment of Pressure Ulcers: Quick Reference Guide. Washington, D.C.: National Pressure Ulcer Advisory Panel; 2009.
- Bates-Jensen BM. Chapter 58. Pressure Ulcers. In: Halter JB, Ouslander JG, Tinetti ME, Studenski S, High KP, Asthana S, eds. Hazzard’s Geriatric Medicine and Gerontology. 6th ed. New York: McGraw-Hill; 2009.
- Livesley NJ, Chow AW. Infected pressure ulcers in elderly individuals. Clin Infect Dis. 2002;35(11):1390-1396.
- Agency for Health Care Policy and Research (AHCPR). Treatment of Pressure Ulcers. Clinical Practice Guideline Number 15. U.S. Department of Health and Human Services. 1994.
- Reddy M, Gill SS, Wu W, Kalkar SR, Rochon PA. Does this patient have an infection of a chronic wound? JAMA. 2012;307(6):605-611.
- National Pressure Ulcer Advisory Panel. Pressure Ulcer Prevention Points. National Pressure Ulcer Advisory Panel website. Available at: http://www.npuap.org/resources/educational-and-clinical-resources/pressure-ulcer-prevention-points/. Accessed Aug. 1, 2012.
- Reuben DB, Herr KA, Pacala JT, et al. Skin Ulcers. In: Geriatrics At Your Fingertips. 12th ed. New York: The American Geriatrics Society; 2010.
John Nelson: Why Spinal Epidural Abcess Poses A Particular Liability Risk for Hospitalists
Delayed diagnosis of, or treatment for, a spinal epidural abscess (SEA): that will be the case over which you are sued.
Over the last 15 years, I’ve served as an expert witness for six or seven malpractice cases. Most were related to spinal cord injuries, and in all but one of those, the etiology was epidural abscess. I’ve been asked to review about 40 or 50 additional cases, and while I’ve turned them down (I just don’t have time to do reviews), I nearly always ask about the clinical picture in every case. A significant number have been SEA-related. This experience has convinced me that SEA poses a particular liability risk for hospitalists.
Of course, it is patients who bear the real risk and unfortunate consequences of SEA. Being a defendant physician in a lawsuit is stressful, but it’s nothing compared to the distress of permanent loss of neurologic function. To prevent permanent sequelae, we need to maintain a very high index of suspicion to try to make a prompt diagnosis, and ensure immediate intervention once the diagnosis is made.
Data from Malpractice Insurers
I had the pleasure of getting to know a number of leaders at The Doctor’s Company, a large malpractice insurer that provides malpractice policies for all specialties, including a lot of hospitalists. From 2007 to 2011, they closed 28 SEA-related claims, for which they spent an average of $212,000 defending each one. Eleven of the 28 resulted in indemnity payments averaging $754,000 each (median was $455,000). These dollar amounts are roughly double what might be seen for all other claims and reflect only the payments made on behalf of the company’s insured doctors. The total award to each patient was likely much higher, because in most cases, several defendants (other doctors and a hospital) probably paid money to the patient.
The Physician Insurers Association of America (PIAA) “is the insurance industry trade association representing domestic and international medical professional liability insurance companies.” Their member malpractice insurance companies have the opportunity to report claims data that PIAA aggregates and makes available. Data from 2002 to 2011 showed 312 closed claims related to any diagnosis (not just SEA) for hospitalists, with an average indemnity payment of $272,553 (the highest hospitalist-related payment was $1.4 million). The most common allegations related to paid claims were 1) “errors in diagnosis,” 2) “failure/delay in referral or consultation,” and 3) “failure to supervise/monitor case.” Although only three of the 312 claims were related to “diseases of the spinal cord,” that was exceeded in frequency only by “diabetes.”
I think these numbers from the malpractice insurance industry support my concern that SEA is a high-risk area, but it doesn’t really support my anecdotal experience that SEA is clearly hospitalists’ highest-risk area. Maybe SEA is only one of several high-risk areas. Nevertheless, I’m going to stick to my sensationalist guns to get your attention.
Why Is Epidural Abscess a High Risk?
There likely are several reasons SEA is a treacherous liability problem. It can lead to devastating permanent disabling neurologic deficits in people who were previously healthy, and if the medical care was substandard, then significant financial compensation seems appropriate.
Delays in diagnosis of SEA are common. It can be a very sneaky illness that in the early stages is very easy to confuse with less-serious causes of back pain or fever. Even though I think about this particular diagnosis all the time, just last year I had a patient who reported an increase in his usual back pain. I felt reassured that he had no neurologic deficit or fever, and took the time to explain why there was no reason to repeat the spine MRI that had been done about two weeks prior to admission. But he was insistent that he have another MRI, and after a day or two I finally agreed to order it, assuring him it would not explain the cause of his pain. But it did. He had a significant SEA and went to emergency surgery. I was stunned, and profoundly relieved that he had no neurologic sequelae.
One of the remarkable things I’ve seen in the cases I’ve reviewed is that even when there is clear cause for concern, there is too often no action taken. In a number of cases, the nurses’ note indicates increasing back pain, loss of ability to stand, urinary retention, and other alarming signs. Yet the doctors either never learn of these issues, or they choose to attribute them to other causes.
Even when the diagnosis of SEA is clearly established, it is all too common for doctors caring for the patient not to act on this information. In several cases I reviewed, a radiologist had documented reporting the diagnosis to the hospitalist (and in one case the neurosurgeon as well), yet nothing was done for 12 hours or more. It is hard to imagine that establishing this diagnosis doesn’t reliably lead to an emergent response, but it doesn’t. (In some cases, nonsurgical management may be an option, but in these malpractices cases, there was just a failure to act on the diagnosis with any sort of plan.)
Practice Management Perspective
I usually discuss hospitalist practice operations in this space—things like work schedules and compensation. But attending to risk management is one component of effective practice operations, so I thought I’d raise the topic here. Obviously, there is a lot more to hospitalist risk management than one diagnosis, but a column on the whole universe of risk management would probably serve no purpose other than as a sleep aid. I hope that by focusing solely on SEA, there is some chance that you’ll remember it, and you’ll make sure that you disprove my first two sentences.
Lowering your risk of a malpractice lawsuit is valuable and worth spending time on. But far more important is that by keeping the diagnosis in mind, and ensuring that you act emergently when there is cause for concern, you might save someone from the devastating consequences of this disease.
Dr. Nelson has been a practicing hospitalist since 1988. He is co-founder and past president of SHM, and principal in Nelson Flores Hospital Medicine Consultants. He is co-director for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. Write to him at john.nelson@nelsonflores.com.
Delayed diagnosis of, or treatment for, a spinal epidural abscess (SEA): that will be the case over which you are sued.
Over the last 15 years, I’ve served as an expert witness for six or seven malpractice cases. Most were related to spinal cord injuries, and in all but one of those, the etiology was epidural abscess. I’ve been asked to review about 40 or 50 additional cases, and while I’ve turned them down (I just don’t have time to do reviews), I nearly always ask about the clinical picture in every case. A significant number have been SEA-related. This experience has convinced me that SEA poses a particular liability risk for hospitalists.
Of course, it is patients who bear the real risk and unfortunate consequences of SEA. Being a defendant physician in a lawsuit is stressful, but it’s nothing compared to the distress of permanent loss of neurologic function. To prevent permanent sequelae, we need to maintain a very high index of suspicion to try to make a prompt diagnosis, and ensure immediate intervention once the diagnosis is made.
Data from Malpractice Insurers
I had the pleasure of getting to know a number of leaders at The Doctor’s Company, a large malpractice insurer that provides malpractice policies for all specialties, including a lot of hospitalists. From 2007 to 2011, they closed 28 SEA-related claims, for which they spent an average of $212,000 defending each one. Eleven of the 28 resulted in indemnity payments averaging $754,000 each (median was $455,000). These dollar amounts are roughly double what might be seen for all other claims and reflect only the payments made on behalf of the company’s insured doctors. The total award to each patient was likely much higher, because in most cases, several defendants (other doctors and a hospital) probably paid money to the patient.
The Physician Insurers Association of America (PIAA) “is the insurance industry trade association representing domestic and international medical professional liability insurance companies.” Their member malpractice insurance companies have the opportunity to report claims data that PIAA aggregates and makes available. Data from 2002 to 2011 showed 312 closed claims related to any diagnosis (not just SEA) for hospitalists, with an average indemnity payment of $272,553 (the highest hospitalist-related payment was $1.4 million). The most common allegations related to paid claims were 1) “errors in diagnosis,” 2) “failure/delay in referral or consultation,” and 3) “failure to supervise/monitor case.” Although only three of the 312 claims were related to “diseases of the spinal cord,” that was exceeded in frequency only by “diabetes.”
I think these numbers from the malpractice insurance industry support my concern that SEA is a high-risk area, but it doesn’t really support my anecdotal experience that SEA is clearly hospitalists’ highest-risk area. Maybe SEA is only one of several high-risk areas. Nevertheless, I’m going to stick to my sensationalist guns to get your attention.
Why Is Epidural Abscess a High Risk?
There likely are several reasons SEA is a treacherous liability problem. It can lead to devastating permanent disabling neurologic deficits in people who were previously healthy, and if the medical care was substandard, then significant financial compensation seems appropriate.
Delays in diagnosis of SEA are common. It can be a very sneaky illness that in the early stages is very easy to confuse with less-serious causes of back pain or fever. Even though I think about this particular diagnosis all the time, just last year I had a patient who reported an increase in his usual back pain. I felt reassured that he had no neurologic deficit or fever, and took the time to explain why there was no reason to repeat the spine MRI that had been done about two weeks prior to admission. But he was insistent that he have another MRI, and after a day or two I finally agreed to order it, assuring him it would not explain the cause of his pain. But it did. He had a significant SEA and went to emergency surgery. I was stunned, and profoundly relieved that he had no neurologic sequelae.
One of the remarkable things I’ve seen in the cases I’ve reviewed is that even when there is clear cause for concern, there is too often no action taken. In a number of cases, the nurses’ note indicates increasing back pain, loss of ability to stand, urinary retention, and other alarming signs. Yet the doctors either never learn of these issues, or they choose to attribute them to other causes.
Even when the diagnosis of SEA is clearly established, it is all too common for doctors caring for the patient not to act on this information. In several cases I reviewed, a radiologist had documented reporting the diagnosis to the hospitalist (and in one case the neurosurgeon as well), yet nothing was done for 12 hours or more. It is hard to imagine that establishing this diagnosis doesn’t reliably lead to an emergent response, but it doesn’t. (In some cases, nonsurgical management may be an option, but in these malpractices cases, there was just a failure to act on the diagnosis with any sort of plan.)
Practice Management Perspective
I usually discuss hospitalist practice operations in this space—things like work schedules and compensation. But attending to risk management is one component of effective practice operations, so I thought I’d raise the topic here. Obviously, there is a lot more to hospitalist risk management than one diagnosis, but a column on the whole universe of risk management would probably serve no purpose other than as a sleep aid. I hope that by focusing solely on SEA, there is some chance that you’ll remember it, and you’ll make sure that you disprove my first two sentences.
Lowering your risk of a malpractice lawsuit is valuable and worth spending time on. But far more important is that by keeping the diagnosis in mind, and ensuring that you act emergently when there is cause for concern, you might save someone from the devastating consequences of this disease.
Dr. Nelson has been a practicing hospitalist since 1988. He is co-founder and past president of SHM, and principal in Nelson Flores Hospital Medicine Consultants. He is co-director for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. Write to him at john.nelson@nelsonflores.com.
Delayed diagnosis of, or treatment for, a spinal epidural abscess (SEA): that will be the case over which you are sued.
Over the last 15 years, I’ve served as an expert witness for six or seven malpractice cases. Most were related to spinal cord injuries, and in all but one of those, the etiology was epidural abscess. I’ve been asked to review about 40 or 50 additional cases, and while I’ve turned them down (I just don’t have time to do reviews), I nearly always ask about the clinical picture in every case. A significant number have been SEA-related. This experience has convinced me that SEA poses a particular liability risk for hospitalists.
Of course, it is patients who bear the real risk and unfortunate consequences of SEA. Being a defendant physician in a lawsuit is stressful, but it’s nothing compared to the distress of permanent loss of neurologic function. To prevent permanent sequelae, we need to maintain a very high index of suspicion to try to make a prompt diagnosis, and ensure immediate intervention once the diagnosis is made.
Data from Malpractice Insurers
I had the pleasure of getting to know a number of leaders at The Doctor’s Company, a large malpractice insurer that provides malpractice policies for all specialties, including a lot of hospitalists. From 2007 to 2011, they closed 28 SEA-related claims, for which they spent an average of $212,000 defending each one. Eleven of the 28 resulted in indemnity payments averaging $754,000 each (median was $455,000). These dollar amounts are roughly double what might be seen for all other claims and reflect only the payments made on behalf of the company’s insured doctors. The total award to each patient was likely much higher, because in most cases, several defendants (other doctors and a hospital) probably paid money to the patient.
The Physician Insurers Association of America (PIAA) “is the insurance industry trade association representing domestic and international medical professional liability insurance companies.” Their member malpractice insurance companies have the opportunity to report claims data that PIAA aggregates and makes available. Data from 2002 to 2011 showed 312 closed claims related to any diagnosis (not just SEA) for hospitalists, with an average indemnity payment of $272,553 (the highest hospitalist-related payment was $1.4 million). The most common allegations related to paid claims were 1) “errors in diagnosis,” 2) “failure/delay in referral or consultation,” and 3) “failure to supervise/monitor case.” Although only three of the 312 claims were related to “diseases of the spinal cord,” that was exceeded in frequency only by “diabetes.”
I think these numbers from the malpractice insurance industry support my concern that SEA is a high-risk area, but it doesn’t really support my anecdotal experience that SEA is clearly hospitalists’ highest-risk area. Maybe SEA is only one of several high-risk areas. Nevertheless, I’m going to stick to my sensationalist guns to get your attention.
Why Is Epidural Abscess a High Risk?
There likely are several reasons SEA is a treacherous liability problem. It can lead to devastating permanent disabling neurologic deficits in people who were previously healthy, and if the medical care was substandard, then significant financial compensation seems appropriate.
Delays in diagnosis of SEA are common. It can be a very sneaky illness that in the early stages is very easy to confuse with less-serious causes of back pain or fever. Even though I think about this particular diagnosis all the time, just last year I had a patient who reported an increase in his usual back pain. I felt reassured that he had no neurologic deficit or fever, and took the time to explain why there was no reason to repeat the spine MRI that had been done about two weeks prior to admission. But he was insistent that he have another MRI, and after a day or two I finally agreed to order it, assuring him it would not explain the cause of his pain. But it did. He had a significant SEA and went to emergency surgery. I was stunned, and profoundly relieved that he had no neurologic sequelae.
One of the remarkable things I’ve seen in the cases I’ve reviewed is that even when there is clear cause for concern, there is too often no action taken. In a number of cases, the nurses’ note indicates increasing back pain, loss of ability to stand, urinary retention, and other alarming signs. Yet the doctors either never learn of these issues, or they choose to attribute them to other causes.
Even when the diagnosis of SEA is clearly established, it is all too common for doctors caring for the patient not to act on this information. In several cases I reviewed, a radiologist had documented reporting the diagnosis to the hospitalist (and in one case the neurosurgeon as well), yet nothing was done for 12 hours or more. It is hard to imagine that establishing this diagnosis doesn’t reliably lead to an emergent response, but it doesn’t. (In some cases, nonsurgical management may be an option, but in these malpractices cases, there was just a failure to act on the diagnosis with any sort of plan.)
Practice Management Perspective
I usually discuss hospitalist practice operations in this space—things like work schedules and compensation. But attending to risk management is one component of effective practice operations, so I thought I’d raise the topic here. Obviously, there is a lot more to hospitalist risk management than one diagnosis, but a column on the whole universe of risk management would probably serve no purpose other than as a sleep aid. I hope that by focusing solely on SEA, there is some chance that you’ll remember it, and you’ll make sure that you disprove my first two sentences.
Lowering your risk of a malpractice lawsuit is valuable and worth spending time on. But far more important is that by keeping the diagnosis in mind, and ensuring that you act emergently when there is cause for concern, you might save someone from the devastating consequences of this disease.
Dr. Nelson has been a practicing hospitalist since 1988. He is co-founder and past president of SHM, and principal in Nelson Flores Hospital Medicine Consultants. He is co-director for SHM’s “Best Practices in Managing a Hospital Medicine Program” course. Write to him at john.nelson@nelsonflores.com.
New Anticoagulants Offer Promise, but Obstacles Remain
I see more and more people taking one of the newer anticoagulants. I’ve also seen a few disasters with these drugs. What’s the story?
Stacy M. Harper, Green Bay, Wis.
Dr. Hospitalist responds:
Although warfarin (Coumadin) has been a mainstay anticoagulant for decades, it can often be a frustrating medicine to manage due to its myriad drug interactions and the constant need for therapeutic testing. Recently, we have seen new medications hit the market (with one more likely to be approved soon), each with its pros and cons. Here’s an overview:
- Dabigatran (Pradaxa): It’s a direct thrombin inhibitor, taken twice daily. It has been approved for use in stroke prevention for atrial fibrillation (afib) (RELY trial) at 150 mg bid. It’s also been extensively studied for VTE prevention after orthopedic surgery, but it has not yet been approved in the U.S. for this indication.
As with all of these drugs, there is no reversal agent and there are no levels to measure. A recent report noted an increased risk of bleeding in patients who are older, have a low BMI, or have renal dysfunction. The manufacturer recommends a dose of 75 mg bid for patients with renal dysfunction, defined as a GFR of 15 to 30 mL/min; however, that dosing regimen was never explicitly studied.
Overall, it’s become quite a popular drug with the cardiologists in my neck of the woods. GERD can be a bothersome side effect. I avoid using it in patients older than 80, or in a patient with any renal dysfunction. Also, remember that it is not approved for VTE prevention or treatment.
- Rivaroxaban (Xarelto): An oral factor Xa inhibitor. Usually taken once daily at 10 mg for VTE prevention (RECORD trials). It is dosed at 20 mg/day for stroke prevention in afib (ROCKET-AF trial). Just recently, it was approved by the FDA for use in the acute treatment of DVT and PE (EINSTEIN trial), dosed at 15 mg BID for the first 21 days, and then continued at 20 mg daily after the initial period (see “Game-Changer,” p. 41). It is more hepatically metabolized than dabigatran, but it still has a significant renal clearance component. When compared to lovenox in orthopedic patients, it’s as effective but with a slightly higher risk of bleeding. I would avoid using it in any patients with significant renal or hepatic dysfunction.
- Apixaban (Eliquis): Another oral factor Xa inhibitor. Studied at 2.5 mg BID for VTE prevention in orthopedic patients (ADVANCE trials). Studied at 5 mg BID for stroke prevention in afib (ARISTOTLE trial). It is not yet approved in the U.S for any indication, but a final decision is expected from the FDA by March. Overall, the data are fairly compelling, and it looks like a strong candidate. The data show a drug that is potentially more effective than lovenox, with less risk of bleeding for orthopedic patients. It is mainly hepatically metabolized.
So, with no drug company relationships to disclose, here are my general observations: For starters, I think dabigatran is being overused in older patients with renal dysfunction. I seem to stop it more than I recommend it, and it is far from my favorite drug. With rivaroxaban, it looks appropriate for VTE prevention, and now having the option of being able to transition patients who develop a clot onto a treatment dose of the drug is appealing. Apixaban’s data look the best out of all three agents in terms of both efficacy and bleeding, and although it is yet to be approved here, I imagine that will change in the near future. For all of these drugs, remember that we have no long-term safety data, and no reversal agents. It will be interesting to see how this plays out and which of these drugs have staying power. For all of warfarin’s faults, at least we know how to measure it and how to stop it.
I see more and more people taking one of the newer anticoagulants. I’ve also seen a few disasters with these drugs. What’s the story?
Stacy M. Harper, Green Bay, Wis.
Dr. Hospitalist responds:
Although warfarin (Coumadin) has been a mainstay anticoagulant for decades, it can often be a frustrating medicine to manage due to its myriad drug interactions and the constant need for therapeutic testing. Recently, we have seen new medications hit the market (with one more likely to be approved soon), each with its pros and cons. Here’s an overview:
- Dabigatran (Pradaxa): It’s a direct thrombin inhibitor, taken twice daily. It has been approved for use in stroke prevention for atrial fibrillation (afib) (RELY trial) at 150 mg bid. It’s also been extensively studied for VTE prevention after orthopedic surgery, but it has not yet been approved in the U.S. for this indication.
As with all of these drugs, there is no reversal agent and there are no levels to measure. A recent report noted an increased risk of bleeding in patients who are older, have a low BMI, or have renal dysfunction. The manufacturer recommends a dose of 75 mg bid for patients with renal dysfunction, defined as a GFR of 15 to 30 mL/min; however, that dosing regimen was never explicitly studied.
Overall, it’s become quite a popular drug with the cardiologists in my neck of the woods. GERD can be a bothersome side effect. I avoid using it in patients older than 80, or in a patient with any renal dysfunction. Also, remember that it is not approved for VTE prevention or treatment.
- Rivaroxaban (Xarelto): An oral factor Xa inhibitor. Usually taken once daily at 10 mg for VTE prevention (RECORD trials). It is dosed at 20 mg/day for stroke prevention in afib (ROCKET-AF trial). Just recently, it was approved by the FDA for use in the acute treatment of DVT and PE (EINSTEIN trial), dosed at 15 mg BID for the first 21 days, and then continued at 20 mg daily after the initial period (see “Game-Changer,” p. 41). It is more hepatically metabolized than dabigatran, but it still has a significant renal clearance component. When compared to lovenox in orthopedic patients, it’s as effective but with a slightly higher risk of bleeding. I would avoid using it in any patients with significant renal or hepatic dysfunction.
- Apixaban (Eliquis): Another oral factor Xa inhibitor. Studied at 2.5 mg BID for VTE prevention in orthopedic patients (ADVANCE trials). Studied at 5 mg BID for stroke prevention in afib (ARISTOTLE trial). It is not yet approved in the U.S for any indication, but a final decision is expected from the FDA by March. Overall, the data are fairly compelling, and it looks like a strong candidate. The data show a drug that is potentially more effective than lovenox, with less risk of bleeding for orthopedic patients. It is mainly hepatically metabolized.
So, with no drug company relationships to disclose, here are my general observations: For starters, I think dabigatran is being overused in older patients with renal dysfunction. I seem to stop it more than I recommend it, and it is far from my favorite drug. With rivaroxaban, it looks appropriate for VTE prevention, and now having the option of being able to transition patients who develop a clot onto a treatment dose of the drug is appealing. Apixaban’s data look the best out of all three agents in terms of both efficacy and bleeding, and although it is yet to be approved here, I imagine that will change in the near future. For all of these drugs, remember that we have no long-term safety data, and no reversal agents. It will be interesting to see how this plays out and which of these drugs have staying power. For all of warfarin’s faults, at least we know how to measure it and how to stop it.
I see more and more people taking one of the newer anticoagulants. I’ve also seen a few disasters with these drugs. What’s the story?
Stacy M. Harper, Green Bay, Wis.
Dr. Hospitalist responds:
Although warfarin (Coumadin) has been a mainstay anticoagulant for decades, it can often be a frustrating medicine to manage due to its myriad drug interactions and the constant need for therapeutic testing. Recently, we have seen new medications hit the market (with one more likely to be approved soon), each with its pros and cons. Here’s an overview:
- Dabigatran (Pradaxa): It’s a direct thrombin inhibitor, taken twice daily. It has been approved for use in stroke prevention for atrial fibrillation (afib) (RELY trial) at 150 mg bid. It’s also been extensively studied for VTE prevention after orthopedic surgery, but it has not yet been approved in the U.S. for this indication.
As with all of these drugs, there is no reversal agent and there are no levels to measure. A recent report noted an increased risk of bleeding in patients who are older, have a low BMI, or have renal dysfunction. The manufacturer recommends a dose of 75 mg bid for patients with renal dysfunction, defined as a GFR of 15 to 30 mL/min; however, that dosing regimen was never explicitly studied.
Overall, it’s become quite a popular drug with the cardiologists in my neck of the woods. GERD can be a bothersome side effect. I avoid using it in patients older than 80, or in a patient with any renal dysfunction. Also, remember that it is not approved for VTE prevention or treatment.
- Rivaroxaban (Xarelto): An oral factor Xa inhibitor. Usually taken once daily at 10 mg for VTE prevention (RECORD trials). It is dosed at 20 mg/day for stroke prevention in afib (ROCKET-AF trial). Just recently, it was approved by the FDA for use in the acute treatment of DVT and PE (EINSTEIN trial), dosed at 15 mg BID for the first 21 days, and then continued at 20 mg daily after the initial period (see “Game-Changer,” p. 41). It is more hepatically metabolized than dabigatran, but it still has a significant renal clearance component. When compared to lovenox in orthopedic patients, it’s as effective but with a slightly higher risk of bleeding. I would avoid using it in any patients with significant renal or hepatic dysfunction.
- Apixaban (Eliquis): Another oral factor Xa inhibitor. Studied at 2.5 mg BID for VTE prevention in orthopedic patients (ADVANCE trials). Studied at 5 mg BID for stroke prevention in afib (ARISTOTLE trial). It is not yet approved in the U.S for any indication, but a final decision is expected from the FDA by March. Overall, the data are fairly compelling, and it looks like a strong candidate. The data show a drug that is potentially more effective than lovenox, with less risk of bleeding for orthopedic patients. It is mainly hepatically metabolized.
So, with no drug company relationships to disclose, here are my general observations: For starters, I think dabigatran is being overused in older patients with renal dysfunction. I seem to stop it more than I recommend it, and it is far from my favorite drug. With rivaroxaban, it looks appropriate for VTE prevention, and now having the option of being able to transition patients who develop a clot onto a treatment dose of the drug is appealing. Apixaban’s data look the best out of all three agents in terms of both efficacy and bleeding, and although it is yet to be approved here, I imagine that will change in the near future. For all of these drugs, remember that we have no long-term safety data, and no reversal agents. It will be interesting to see how this plays out and which of these drugs have staying power. For all of warfarin’s faults, at least we know how to measure it and how to stop it.
Clinical Shorts
INFLUENZA VACCINE EFFECTIVENESS VARIES BY AGE
Case-control study of the 2010-2011 influenza vaccine found overall vaccine effectiveness to be 60%, ranging from 69% in those ages 3 to 8 to just 36% in those 65 or older.
Citation: Treanor JJ, Talbot HK, Ohmit SE, et al. Effectiveness of seasonal influenza vaccines in the United States during a season with circulation of all three vaccine strains. Clin Infect Dis. 2012;55:951-959.
NSAIDS INCREASE CV RISK AFTER MI, REGARDLESS OF LENGTH OF TIME
A nationwide cohort study in Denmark shows increased coronary risk with NSAID use for at least five years after first-time myocardial infarction.
Citation: Olsen AM, Fosbøl EL, Lindhardsen J, et al. Long-term cardiovascular risk of nonsteroidal anti-inflammatory drug use according to time passed after first-time myocardial infarction: a nationwide cohort study. Circulation. 2012;126:1955-1963.
PATIENTS WITH METASTATIC CANCER OFTEN OVERESTIMATE CHEMOTHERAPEUTIC EFFICACY
Survey of patients with metastatic solid tumors reveals significant misunderstanding regarding the curative potential of chemotherapy, and an inverse relationship between level of understanding and patients’ satisfaction with physician communication.
Citation: Weeks JC, Catalano PJ, Cronin A, et al. Patients’ expectations about effects of chemotherapy for advanced cancer. N Engl J Med. 2012;367:1616-1625.
RISKS ASSOCIATED WITH SYNTHETIC CANNABINOID ABUSE
This case series from the National Poison Data System indicates that adverse effects of synthetic cannabinoids are generally mild and self-limited, though rare reports of life-threatening seizures were identified.
Citation: Hoyte CO, Jacob J, Monte AA, Al-Jumaan M, Bronstein AC, Heard KJ. A characterization of synthetic cannabinoid exposures reported to the National Poison Data System in 2010. Ann Emerg Med. 2012;60:435-438.
FDA APPROVES FIRST SUBCUTANEOUS HEART DEFIBRILLATOR
Based on a multicenter study of 321 patients, the FDA approved the first subcutaneous heart defibrillator, which might be useful for patients in whom intravascular lead placement is problematic.
Citation: Bolek M. FDA approves first subcutaneous heart defibrillator. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm321755.htm. Accessed Jan. 2, 2013.
ADD PCV13 TO THE LIST OF ADULT IMMUNIZATIONS
Thirteen-valent pneumococcal conjugate vaccine (PCV13) is now recommended in addition to 23-valent pneumococcal polysaccharide vaccine in adults 19 and older with immunocompromising conditions to decrease the risk of invasive pneumococcal disease.
Citation: Bennett NM, Whitney CG, Moore M, et al. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for adults with immunocompromising conditions: recommendations of the advisory committee on immunization practices (ACIP). MMWR Morb Mortal Wkly Rep. 2012;61:816-819.
INFLUENZA VACCINE EFFECTIVENESS VARIES BY AGE
Case-control study of the 2010-2011 influenza vaccine found overall vaccine effectiveness to be 60%, ranging from 69% in those ages 3 to 8 to just 36% in those 65 or older.
Citation: Treanor JJ, Talbot HK, Ohmit SE, et al. Effectiveness of seasonal influenza vaccines in the United States during a season with circulation of all three vaccine strains. Clin Infect Dis. 2012;55:951-959.
NSAIDS INCREASE CV RISK AFTER MI, REGARDLESS OF LENGTH OF TIME
A nationwide cohort study in Denmark shows increased coronary risk with NSAID use for at least five years after first-time myocardial infarction.
Citation: Olsen AM, Fosbøl EL, Lindhardsen J, et al. Long-term cardiovascular risk of nonsteroidal anti-inflammatory drug use according to time passed after first-time myocardial infarction: a nationwide cohort study. Circulation. 2012;126:1955-1963.
PATIENTS WITH METASTATIC CANCER OFTEN OVERESTIMATE CHEMOTHERAPEUTIC EFFICACY
Survey of patients with metastatic solid tumors reveals significant misunderstanding regarding the curative potential of chemotherapy, and an inverse relationship between level of understanding and patients’ satisfaction with physician communication.
Citation: Weeks JC, Catalano PJ, Cronin A, et al. Patients’ expectations about effects of chemotherapy for advanced cancer. N Engl J Med. 2012;367:1616-1625.
RISKS ASSOCIATED WITH SYNTHETIC CANNABINOID ABUSE
This case series from the National Poison Data System indicates that adverse effects of synthetic cannabinoids are generally mild and self-limited, though rare reports of life-threatening seizures were identified.
Citation: Hoyte CO, Jacob J, Monte AA, Al-Jumaan M, Bronstein AC, Heard KJ. A characterization of synthetic cannabinoid exposures reported to the National Poison Data System in 2010. Ann Emerg Med. 2012;60:435-438.
FDA APPROVES FIRST SUBCUTANEOUS HEART DEFIBRILLATOR
Based on a multicenter study of 321 patients, the FDA approved the first subcutaneous heart defibrillator, which might be useful for patients in whom intravascular lead placement is problematic.
Citation: Bolek M. FDA approves first subcutaneous heart defibrillator. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm321755.htm. Accessed Jan. 2, 2013.
ADD PCV13 TO THE LIST OF ADULT IMMUNIZATIONS
Thirteen-valent pneumococcal conjugate vaccine (PCV13) is now recommended in addition to 23-valent pneumococcal polysaccharide vaccine in adults 19 and older with immunocompromising conditions to decrease the risk of invasive pneumococcal disease.
Citation: Bennett NM, Whitney CG, Moore M, et al. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for adults with immunocompromising conditions: recommendations of the advisory committee on immunization practices (ACIP). MMWR Morb Mortal Wkly Rep. 2012;61:816-819.
INFLUENZA VACCINE EFFECTIVENESS VARIES BY AGE
Case-control study of the 2010-2011 influenza vaccine found overall vaccine effectiveness to be 60%, ranging from 69% in those ages 3 to 8 to just 36% in those 65 or older.
Citation: Treanor JJ, Talbot HK, Ohmit SE, et al. Effectiveness of seasonal influenza vaccines in the United States during a season with circulation of all three vaccine strains. Clin Infect Dis. 2012;55:951-959.
NSAIDS INCREASE CV RISK AFTER MI, REGARDLESS OF LENGTH OF TIME
A nationwide cohort study in Denmark shows increased coronary risk with NSAID use for at least five years after first-time myocardial infarction.
Citation: Olsen AM, Fosbøl EL, Lindhardsen J, et al. Long-term cardiovascular risk of nonsteroidal anti-inflammatory drug use according to time passed after first-time myocardial infarction: a nationwide cohort study. Circulation. 2012;126:1955-1963.
PATIENTS WITH METASTATIC CANCER OFTEN OVERESTIMATE CHEMOTHERAPEUTIC EFFICACY
Survey of patients with metastatic solid tumors reveals significant misunderstanding regarding the curative potential of chemotherapy, and an inverse relationship between level of understanding and patients’ satisfaction with physician communication.
Citation: Weeks JC, Catalano PJ, Cronin A, et al. Patients’ expectations about effects of chemotherapy for advanced cancer. N Engl J Med. 2012;367:1616-1625.
RISKS ASSOCIATED WITH SYNTHETIC CANNABINOID ABUSE
This case series from the National Poison Data System indicates that adverse effects of synthetic cannabinoids are generally mild and self-limited, though rare reports of life-threatening seizures were identified.
Citation: Hoyte CO, Jacob J, Monte AA, Al-Jumaan M, Bronstein AC, Heard KJ. A characterization of synthetic cannabinoid exposures reported to the National Poison Data System in 2010. Ann Emerg Med. 2012;60:435-438.
FDA APPROVES FIRST SUBCUTANEOUS HEART DEFIBRILLATOR
Based on a multicenter study of 321 patients, the FDA approved the first subcutaneous heart defibrillator, which might be useful for patients in whom intravascular lead placement is problematic.
Citation: Bolek M. FDA approves first subcutaneous heart defibrillator. Food and Drug Administration website. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm321755.htm. Accessed Jan. 2, 2013.
ADD PCV13 TO THE LIST OF ADULT IMMUNIZATIONS
Thirteen-valent pneumococcal conjugate vaccine (PCV13) is now recommended in addition to 23-valent pneumococcal polysaccharide vaccine in adults 19 and older with immunocompromising conditions to decrease the risk of invasive pneumococcal disease.
Citation: Bennett NM, Whitney CG, Moore M, et al. Use of 13-valent pneumococcal conjugate vaccine and 23-valent pneumococcal polysaccharide vaccine for adults with immunocompromising conditions: recommendations of the advisory committee on immunization practices (ACIP). MMWR Morb Mortal Wkly Rep. 2012;61:816-819.
Procalcitonin Nears Prime Time
Clinical question: What is the relationship between procalcitonin (ProCT) levels and radiographic abnormalities in patients with suspected pneumonia?
Background: Pneumonia is a common clinical diagnosis and reason for admission to the hospital. In a number of cases, however, chest X-rays might not conclusively show an infiltrate. The correlation between ProCT levels and X-ray findings has not been well studied.
Study design: Prospective cohort study.
Setting: Tertiary-care U.S. hospital.
Synopsis: In all, 528 patients with acute respiratory illness were enrolled in the study. Both a blinded radiologist and a pulmonologist involved in the care of each patient reviewed the chest films independently. The pulmonologist was more likely to judge that an infiltrate was present. ProCT levels correlated well with the diagnosis of pneumonia and the presence of an infiltrate by chest X-ray. For patients with an indeterminant film, as determined by the radiologist, ProCT levels were higher in those patients in whom the pulmonologist judged an infiltrate was indeed present. Nearly every patient with an indeterminant film received antibiotics.
One limitation of the study was the lack of a clear gold standard for the determination of a pneumonia diagnosis. And as an observational study, it is uncertain what the effect might be of withholding antibiotics from patients with an indeterminant film and low ProCT levels.
Bottom line: Serum procalcitonin levels correlate well with the presence of an infiltrate by chest X-ray and might have a future role for determining whether patients with indeterminant films can be safely treated without antimicrobials.
Citation: Walsh EE, Swinburne AJ, Becker KL, et al. Can serum procalcitonin levels help interpret indeterminate chest radiographs in patients hospitalized with acute respiratory illness? J Hosp Med. 2012. doi:10.1002/jhm.1984 [Epub ahead of print].
Clinical question: What is the relationship between procalcitonin (ProCT) levels and radiographic abnormalities in patients with suspected pneumonia?
Background: Pneumonia is a common clinical diagnosis and reason for admission to the hospital. In a number of cases, however, chest X-rays might not conclusively show an infiltrate. The correlation between ProCT levels and X-ray findings has not been well studied.
Study design: Prospective cohort study.
Setting: Tertiary-care U.S. hospital.
Synopsis: In all, 528 patients with acute respiratory illness were enrolled in the study. Both a blinded radiologist and a pulmonologist involved in the care of each patient reviewed the chest films independently. The pulmonologist was more likely to judge that an infiltrate was present. ProCT levels correlated well with the diagnosis of pneumonia and the presence of an infiltrate by chest X-ray. For patients with an indeterminant film, as determined by the radiologist, ProCT levels were higher in those patients in whom the pulmonologist judged an infiltrate was indeed present. Nearly every patient with an indeterminant film received antibiotics.
One limitation of the study was the lack of a clear gold standard for the determination of a pneumonia diagnosis. And as an observational study, it is uncertain what the effect might be of withholding antibiotics from patients with an indeterminant film and low ProCT levels.
Bottom line: Serum procalcitonin levels correlate well with the presence of an infiltrate by chest X-ray and might have a future role for determining whether patients with indeterminant films can be safely treated without antimicrobials.
Citation: Walsh EE, Swinburne AJ, Becker KL, et al. Can serum procalcitonin levels help interpret indeterminate chest radiographs in patients hospitalized with acute respiratory illness? J Hosp Med. 2012. doi:10.1002/jhm.1984 [Epub ahead of print].
Clinical question: What is the relationship between procalcitonin (ProCT) levels and radiographic abnormalities in patients with suspected pneumonia?
Background: Pneumonia is a common clinical diagnosis and reason for admission to the hospital. In a number of cases, however, chest X-rays might not conclusively show an infiltrate. The correlation between ProCT levels and X-ray findings has not been well studied.
Study design: Prospective cohort study.
Setting: Tertiary-care U.S. hospital.
Synopsis: In all, 528 patients with acute respiratory illness were enrolled in the study. Both a blinded radiologist and a pulmonologist involved in the care of each patient reviewed the chest films independently. The pulmonologist was more likely to judge that an infiltrate was present. ProCT levels correlated well with the diagnosis of pneumonia and the presence of an infiltrate by chest X-ray. For patients with an indeterminant film, as determined by the radiologist, ProCT levels were higher in those patients in whom the pulmonologist judged an infiltrate was indeed present. Nearly every patient with an indeterminant film received antibiotics.
One limitation of the study was the lack of a clear gold standard for the determination of a pneumonia diagnosis. And as an observational study, it is uncertain what the effect might be of withholding antibiotics from patients with an indeterminant film and low ProCT levels.
Bottom line: Serum procalcitonin levels correlate well with the presence of an infiltrate by chest X-ray and might have a future role for determining whether patients with indeterminant films can be safely treated without antimicrobials.
Citation: Walsh EE, Swinburne AJ, Becker KL, et al. Can serum procalcitonin levels help interpret indeterminate chest radiographs in patients hospitalized with acute respiratory illness? J Hosp Med. 2012. doi:10.1002/jhm.1984 [Epub ahead of print].
Readmission after Initial Injury Is Common
Clinical question: How frequently are patients readmitted after an initial inpatient stay for an injury, and what factors might predict readmission?
Background: Readmission to the hospital is a vexing healthcare problem prompting substantial investigation into factors that predict readmission after a medical or surgical illness. Data regarding readmission rates following injuries are lacking, as is our understanding of the factors that predict these readmissions.
Study design: Retrospective cohort study.
Setting: Hospitals in 11 U.S. states participating in the 2006 Healthcare Cost and Utilization Project State Inpatient Databases and State Emergency Department Databases.
Synopsis: The authors studied more than 200,000 patients aged 65 and older admitted to the hospital with an injury. Fracture was the most common injury (75%) and falls were the most common mechanism of injury (75%). The overall 30-day readmission rate was 13.7%, or about 1 in 7, which is below the rate commonly seen with medical illnesses.
The most common reasons for readmission were surgery (7.4%) and pneumonia (7.2%). Factors that predicted readmission included an initially “moderate” or “severe” injury, as defined by the validated New Injury Severity Score, the need for blood transfusion during admission, the presence of an infection, and the occurrence of a patient safety event, such as a fall. Discharge to a nursing home was associated with increased risk for readmission.
Bottom line: Readmission after an acute injury is less common than after a medical illness but still occurred in 1 in 7 patients.
Citation: Spector WD, Mutter R, Owens P, Limcangco R. Thirty-day, all-cause readmissions for elderly patients who have an injury-related inpatient stay. Med Care. 2012;50:863-869.
Clinical question: How frequently are patients readmitted after an initial inpatient stay for an injury, and what factors might predict readmission?
Background: Readmission to the hospital is a vexing healthcare problem prompting substantial investigation into factors that predict readmission after a medical or surgical illness. Data regarding readmission rates following injuries are lacking, as is our understanding of the factors that predict these readmissions.
Study design: Retrospective cohort study.
Setting: Hospitals in 11 U.S. states participating in the 2006 Healthcare Cost and Utilization Project State Inpatient Databases and State Emergency Department Databases.
Synopsis: The authors studied more than 200,000 patients aged 65 and older admitted to the hospital with an injury. Fracture was the most common injury (75%) and falls were the most common mechanism of injury (75%). The overall 30-day readmission rate was 13.7%, or about 1 in 7, which is below the rate commonly seen with medical illnesses.
The most common reasons for readmission were surgery (7.4%) and pneumonia (7.2%). Factors that predicted readmission included an initially “moderate” or “severe” injury, as defined by the validated New Injury Severity Score, the need for blood transfusion during admission, the presence of an infection, and the occurrence of a patient safety event, such as a fall. Discharge to a nursing home was associated with increased risk for readmission.
Bottom line: Readmission after an acute injury is less common than after a medical illness but still occurred in 1 in 7 patients.
Citation: Spector WD, Mutter R, Owens P, Limcangco R. Thirty-day, all-cause readmissions for elderly patients who have an injury-related inpatient stay. Med Care. 2012;50:863-869.
Clinical question: How frequently are patients readmitted after an initial inpatient stay for an injury, and what factors might predict readmission?
Background: Readmission to the hospital is a vexing healthcare problem prompting substantial investigation into factors that predict readmission after a medical or surgical illness. Data regarding readmission rates following injuries are lacking, as is our understanding of the factors that predict these readmissions.
Study design: Retrospective cohort study.
Setting: Hospitals in 11 U.S. states participating in the 2006 Healthcare Cost and Utilization Project State Inpatient Databases and State Emergency Department Databases.
Synopsis: The authors studied more than 200,000 patients aged 65 and older admitted to the hospital with an injury. Fracture was the most common injury (75%) and falls were the most common mechanism of injury (75%). The overall 30-day readmission rate was 13.7%, or about 1 in 7, which is below the rate commonly seen with medical illnesses.
The most common reasons for readmission were surgery (7.4%) and pneumonia (7.2%). Factors that predicted readmission included an initially “moderate” or “severe” injury, as defined by the validated New Injury Severity Score, the need for blood transfusion during admission, the presence of an infection, and the occurrence of a patient safety event, such as a fall. Discharge to a nursing home was associated with increased risk for readmission.
Bottom line: Readmission after an acute injury is less common than after a medical illness but still occurred in 1 in 7 patients.
Citation: Spector WD, Mutter R, Owens P, Limcangco R. Thirty-day, all-cause readmissions for elderly patients who have an injury-related inpatient stay. Med Care. 2012;50:863-869.
Daily Sedation Interruption among Intubated Not Helpful
Clinical question: Does sedation by protocol, in combination with daily sedative interruption, reduce the duration of mechanical ventilation and ICU stay?
Background: Limiting excessive sedation in mechanically ventilated adults is associated with shorter duration of mechanical ventilation and lower risk of delirium. Two strategies to minimize sedation are daily sedation interruptions and protocolized sedation. These strategies have not been evaluated in combination.
Study design: Randomized controlled trial.
Setting: Tertiary-care medical and surgical ICUs in Canada and the U.S.
Synopsis: This randomized controlled trial conducted in 430 critically ill, mechanically ventilated adults at 16 tertiary-care ICUs showed no difference in time to extubation (mean seven days in each group) or ICU length of stay (mean 10 days in each group) in protocolized sedation and sedation interruption compared with protocolized sedation alone. The daily interruption group received higher mean daily doses of midazolam and fentanyl, increased number of boluses of benzodiazepines and opiates, and required increased nurse workload. There was no difference in unintentional endotracheal tube removal or rate of delirium.
A limitation of this study was the use of continuous opioid and/or benzodiazepine infusions instead of bolus dosing. Hospitalists involved in critical care should be careful about changing their practice based on this study alone.
Bottom line: The addition of daily sedation interruptions in mechanically ventilated patients treated with protocolized sedation does not reduce duration of ventilation or ICU stay.
Citation: Mehta S, Burry L, Cook D, et al. Daily sedation interruption in mechanically ventilated critically ill patients cared for with a sedation protocol. JAMA. 2012;308:1985-1992.
Clinical question: Does sedation by protocol, in combination with daily sedative interruption, reduce the duration of mechanical ventilation and ICU stay?
Background: Limiting excessive sedation in mechanically ventilated adults is associated with shorter duration of mechanical ventilation and lower risk of delirium. Two strategies to minimize sedation are daily sedation interruptions and protocolized sedation. These strategies have not been evaluated in combination.
Study design: Randomized controlled trial.
Setting: Tertiary-care medical and surgical ICUs in Canada and the U.S.
Synopsis: This randomized controlled trial conducted in 430 critically ill, mechanically ventilated adults at 16 tertiary-care ICUs showed no difference in time to extubation (mean seven days in each group) or ICU length of stay (mean 10 days in each group) in protocolized sedation and sedation interruption compared with protocolized sedation alone. The daily interruption group received higher mean daily doses of midazolam and fentanyl, increased number of boluses of benzodiazepines and opiates, and required increased nurse workload. There was no difference in unintentional endotracheal tube removal or rate of delirium.
A limitation of this study was the use of continuous opioid and/or benzodiazepine infusions instead of bolus dosing. Hospitalists involved in critical care should be careful about changing their practice based on this study alone.
Bottom line: The addition of daily sedation interruptions in mechanically ventilated patients treated with protocolized sedation does not reduce duration of ventilation or ICU stay.
Citation: Mehta S, Burry L, Cook D, et al. Daily sedation interruption in mechanically ventilated critically ill patients cared for with a sedation protocol. JAMA. 2012;308:1985-1992.
Clinical question: Does sedation by protocol, in combination with daily sedative interruption, reduce the duration of mechanical ventilation and ICU stay?
Background: Limiting excessive sedation in mechanically ventilated adults is associated with shorter duration of mechanical ventilation and lower risk of delirium. Two strategies to minimize sedation are daily sedation interruptions and protocolized sedation. These strategies have not been evaluated in combination.
Study design: Randomized controlled trial.
Setting: Tertiary-care medical and surgical ICUs in Canada and the U.S.
Synopsis: This randomized controlled trial conducted in 430 critically ill, mechanically ventilated adults at 16 tertiary-care ICUs showed no difference in time to extubation (mean seven days in each group) or ICU length of stay (mean 10 days in each group) in protocolized sedation and sedation interruption compared with protocolized sedation alone. The daily interruption group received higher mean daily doses of midazolam and fentanyl, increased number of boluses of benzodiazepines and opiates, and required increased nurse workload. There was no difference in unintentional endotracheal tube removal or rate of delirium.
A limitation of this study was the use of continuous opioid and/or benzodiazepine infusions instead of bolus dosing. Hospitalists involved in critical care should be careful about changing their practice based on this study alone.
Bottom line: The addition of daily sedation interruptions in mechanically ventilated patients treated with protocolized sedation does not reduce duration of ventilation or ICU stay.
Citation: Mehta S, Burry L, Cook D, et al. Daily sedation interruption in mechanically ventilated critically ill patients cared for with a sedation protocol. JAMA. 2012;308:1985-1992.
Risk for Falls Might Not Affect Anticoagulation Decision
Clinical question: Do patients on oral anticoagulation with high fall risk have an increased incidence of major bleeding?
Background: Despite proven efficacy, oral anticoagulation remains underprescribed. The most commonly cited reasons for not providing oral anticoagulation when clinically indicated are risk of falls and concern for major bleeding.
Study design: Prospective cohort study.
Setting: Internal-medicine inpatient and outpatient services of a university hospital in Switzerland.
Synopsis: This study followed 515 patients on oral anticoagulation for 12 months. Patients at high risk for falls were identified using validated questions known to predict fall risk. Overall, 35 patients had a first major bleed. In multivariate analysis, high fall risk was not associated with an increased incidence of major bleeding (hazard ratio 1.09; 95% confidence interval, 0.54-2.21). Only 1 in 3 fall-related bleeds occurred in the high-fall-risk group.
This study was limited significantly by selection bias. The majority of patients studied already were on anticoagulation therapy for at least three months prior to enrolling in the study, presumably without major bleeding. It is probable that some higher-risk patients were not offered anticoagulation at all and would have been ineligible for the study. This study cohort might have had a lower bleeding risk than members of the general population being started on anticoagulation.
Bottom line: This prospective cohort study shows that patients on oral anticoagulation at high risk of falls did not have significantly increased rates of major bleeding; however, selection bias might have led to an underestimation of bleeding risk. Hospitalists should continue to individualize anticoagulation decisions.
Citation: Donzé J, Clair C, Hug B, et al. Risk of falls and major bleeds in patients on oral anticoagulation therapy. Am J Med. 2012;125:773-778.
Clinical question: Do patients on oral anticoagulation with high fall risk have an increased incidence of major bleeding?
Background: Despite proven efficacy, oral anticoagulation remains underprescribed. The most commonly cited reasons for not providing oral anticoagulation when clinically indicated are risk of falls and concern for major bleeding.
Study design: Prospective cohort study.
Setting: Internal-medicine inpatient and outpatient services of a university hospital in Switzerland.
Synopsis: This study followed 515 patients on oral anticoagulation for 12 months. Patients at high risk for falls were identified using validated questions known to predict fall risk. Overall, 35 patients had a first major bleed. In multivariate analysis, high fall risk was not associated with an increased incidence of major bleeding (hazard ratio 1.09; 95% confidence interval, 0.54-2.21). Only 1 in 3 fall-related bleeds occurred in the high-fall-risk group.
This study was limited significantly by selection bias. The majority of patients studied already were on anticoagulation therapy for at least three months prior to enrolling in the study, presumably without major bleeding. It is probable that some higher-risk patients were not offered anticoagulation at all and would have been ineligible for the study. This study cohort might have had a lower bleeding risk than members of the general population being started on anticoagulation.
Bottom line: This prospective cohort study shows that patients on oral anticoagulation at high risk of falls did not have significantly increased rates of major bleeding; however, selection bias might have led to an underestimation of bleeding risk. Hospitalists should continue to individualize anticoagulation decisions.
Citation: Donzé J, Clair C, Hug B, et al. Risk of falls and major bleeds in patients on oral anticoagulation therapy. Am J Med. 2012;125:773-778.
Clinical question: Do patients on oral anticoagulation with high fall risk have an increased incidence of major bleeding?
Background: Despite proven efficacy, oral anticoagulation remains underprescribed. The most commonly cited reasons for not providing oral anticoagulation when clinically indicated are risk of falls and concern for major bleeding.
Study design: Prospective cohort study.
Setting: Internal-medicine inpatient and outpatient services of a university hospital in Switzerland.
Synopsis: This study followed 515 patients on oral anticoagulation for 12 months. Patients at high risk for falls were identified using validated questions known to predict fall risk. Overall, 35 patients had a first major bleed. In multivariate analysis, high fall risk was not associated with an increased incidence of major bleeding (hazard ratio 1.09; 95% confidence interval, 0.54-2.21). Only 1 in 3 fall-related bleeds occurred in the high-fall-risk group.
This study was limited significantly by selection bias. The majority of patients studied already were on anticoagulation therapy for at least three months prior to enrolling in the study, presumably without major bleeding. It is probable that some higher-risk patients were not offered anticoagulation at all and would have been ineligible for the study. This study cohort might have had a lower bleeding risk than members of the general population being started on anticoagulation.
Bottom line: This prospective cohort study shows that patients on oral anticoagulation at high risk of falls did not have significantly increased rates of major bleeding; however, selection bias might have led to an underestimation of bleeding risk. Hospitalists should continue to individualize anticoagulation decisions.
Citation: Donzé J, Clair C, Hug B, et al. Risk of falls and major bleeds in patients on oral anticoagulation therapy. Am J Med. 2012;125:773-778.
Improving Transitions from ED to Inpatient Care
Clinical question: Can a multidisciplinary focus group identify “best practices” for ensuring efficient and effective transitions of care between the ED and the inpatient setting?
Background: In the admission process, communication failures can lead to preventable adverse effects. Little has been done to evaluate or improve the interservice handoff between the ED physician and the HM physician.
Study design: Concept article.
Synopsis: Handoffs between ED physicians and HM physicians are complex due to differing pressures, cultures, and expectations. The authors recommend an interactive handoff conversation that is organized, focuses on key principles, and is accompanied by a mutual understanding of the differences between specialties. ED physicians and hospitalists should work together to develop joint expectations on content, delivery, and timing of patient handoffs.
One proposed method includes the current clinical condition of the patient, a working problem statement with degree of certainty and rationale, essential aspects of the history and physical, a brief summary of the ED course, analysis of key tests, pending data with unambiguous assignment for follow-up, and any unusual circumstances. Further research is required to determine if these suggestions improve patient outcomes.
Bottom line: Joint expectations and standardized handoff methods between emergency physicians and hospitalists are likely to foster improved communication and patient care.
Citation: Beach C, Cheung DS, Apker J, et al. Improving inter-unit transitions of care between emergency physicians and hospital medicine physicians: a conceptual approach. Acad Emerg Med. 2012;19:1188-1195.
Clinical question: Can a multidisciplinary focus group identify “best practices” for ensuring efficient and effective transitions of care between the ED and the inpatient setting?
Background: In the admission process, communication failures can lead to preventable adverse effects. Little has been done to evaluate or improve the interservice handoff between the ED physician and the HM physician.
Study design: Concept article.
Synopsis: Handoffs between ED physicians and HM physicians are complex due to differing pressures, cultures, and expectations. The authors recommend an interactive handoff conversation that is organized, focuses on key principles, and is accompanied by a mutual understanding of the differences between specialties. ED physicians and hospitalists should work together to develop joint expectations on content, delivery, and timing of patient handoffs.
One proposed method includes the current clinical condition of the patient, a working problem statement with degree of certainty and rationale, essential aspects of the history and physical, a brief summary of the ED course, analysis of key tests, pending data with unambiguous assignment for follow-up, and any unusual circumstances. Further research is required to determine if these suggestions improve patient outcomes.
Bottom line: Joint expectations and standardized handoff methods between emergency physicians and hospitalists are likely to foster improved communication and patient care.
Citation: Beach C, Cheung DS, Apker J, et al. Improving inter-unit transitions of care between emergency physicians and hospital medicine physicians: a conceptual approach. Acad Emerg Med. 2012;19:1188-1195.
Clinical question: Can a multidisciplinary focus group identify “best practices” for ensuring efficient and effective transitions of care between the ED and the inpatient setting?
Background: In the admission process, communication failures can lead to preventable adverse effects. Little has been done to evaluate or improve the interservice handoff between the ED physician and the HM physician.
Study design: Concept article.
Synopsis: Handoffs between ED physicians and HM physicians are complex due to differing pressures, cultures, and expectations. The authors recommend an interactive handoff conversation that is organized, focuses on key principles, and is accompanied by a mutual understanding of the differences between specialties. ED physicians and hospitalists should work together to develop joint expectations on content, delivery, and timing of patient handoffs.
One proposed method includes the current clinical condition of the patient, a working problem statement with degree of certainty and rationale, essential aspects of the history and physical, a brief summary of the ED course, analysis of key tests, pending data with unambiguous assignment for follow-up, and any unusual circumstances. Further research is required to determine if these suggestions improve patient outcomes.
Bottom line: Joint expectations and standardized handoff methods between emergency physicians and hospitalists are likely to foster improved communication and patient care.
Citation: Beach C, Cheung DS, Apker J, et al. Improving inter-unit transitions of care between emergency physicians and hospital medicine physicians: a conceptual approach. Acad Emerg Med. 2012;19:1188-1195.