User login
Mucinous Adenocarcinoma of the Rectum: Clinical Outcomes and Characteristics of 14,044 Patients From the National Cancer Database
PURPOSE
Study clinical characteristics of Rectal Mucinous Adenocarcinoma (RMA).
BACKGROUND
RMA is a rare histological subtype with an impaired response to chemoradiotherapy and an overall poor prognosis. High-grade tumors are associated with older age. Previous studies have shown conflicting results on prognosis.
METHODS
Retrospective analysis of National Cancer Database was conducted from 2004-2020 for subjects with histology code 8480 in primary sites C19 and C20 (rectosigmoid-junction and rectum, n = 14,044), using multivariate analysis with Cox regression.
RESULTS
Median age of diagnosis was 65 years with 69.5% were in the 45-75 years age range. 59.2% were male while 40.8% were female. 84.7% were White, 9.7% were Black, 0.4% were American Indian and 3.4% were Asian. 6.9% were Hispanic. 33.9% were in the upper-income quartile. 40.6% were seen at community cancer programs while 33% went to academic programs. 36.5% had stage III RMA. Out of the 14,044 patients with RMA, 10,546 received surgery, 5,179 received chemotherapy, 233 received immunotherapy and 55 received hormone therapy. Patients >75 years had significantly lower overall survival (OS) compared to those <45 years (HR 0.67). Female patients had significantly higher OS than male (HR - 0.07). Black patients had significantly lower OS than White (HR 0.08). Hispanic patients had significantly higher OS than non- Hispanic (HR - 0.14). Patients with private and government insurance had significantly higher OS than noninsured patients (HR - 0.35 and - 0.26 respectively). Patients with median higher-income quartiles had significantly higher OS than lower quartiles (HR - 0.13). Academic facilities had significantly higher OS than community programs (HR - 0.13). Patients who received surgery had significantly higher OS than those that did not (HR - 0.67); median survival for patients who received surgery was 71 months vs 28 months for non-surgical candidates.
CONCLUSIONS
Surgery is the most important treatment modality in RMA. Uninsured, older Black male patients from lower-income quartiles had significantly lower OS. Access to academic centers also contributed to differences in OS outcomes which throws light on healthcare disparities.
IMPLICATIONS
Additional studies need to be conducted for viable solutions to assist with social determinants of healthcare in RMA.
PURPOSE
Study clinical characteristics of Rectal Mucinous Adenocarcinoma (RMA).
BACKGROUND
RMA is a rare histological subtype with an impaired response to chemoradiotherapy and an overall poor prognosis. High-grade tumors are associated with older age. Previous studies have shown conflicting results on prognosis.
METHODS
Retrospective analysis of National Cancer Database was conducted from 2004-2020 for subjects with histology code 8480 in primary sites C19 and C20 (rectosigmoid-junction and rectum, n = 14,044), using multivariate analysis with Cox regression.
RESULTS
Median age of diagnosis was 65 years with 69.5% were in the 45-75 years age range. 59.2% were male while 40.8% were female. 84.7% were White, 9.7% were Black, 0.4% were American Indian and 3.4% were Asian. 6.9% were Hispanic. 33.9% were in the upper-income quartile. 40.6% were seen at community cancer programs while 33% went to academic programs. 36.5% had stage III RMA. Out of the 14,044 patients with RMA, 10,546 received surgery, 5,179 received chemotherapy, 233 received immunotherapy and 55 received hormone therapy. Patients >75 years had significantly lower overall survival (OS) compared to those <45 years (HR 0.67). Female patients had significantly higher OS than male (HR - 0.07). Black patients had significantly lower OS than White (HR 0.08). Hispanic patients had significantly higher OS than non- Hispanic (HR - 0.14). Patients with private and government insurance had significantly higher OS than noninsured patients (HR - 0.35 and - 0.26 respectively). Patients with median higher-income quartiles had significantly higher OS than lower quartiles (HR - 0.13). Academic facilities had significantly higher OS than community programs (HR - 0.13). Patients who received surgery had significantly higher OS than those that did not (HR - 0.67); median survival for patients who received surgery was 71 months vs 28 months for non-surgical candidates.
CONCLUSIONS
Surgery is the most important treatment modality in RMA. Uninsured, older Black male patients from lower-income quartiles had significantly lower OS. Access to academic centers also contributed to differences in OS outcomes which throws light on healthcare disparities.
IMPLICATIONS
Additional studies need to be conducted for viable solutions to assist with social determinants of healthcare in RMA.
PURPOSE
Study clinical characteristics of Rectal Mucinous Adenocarcinoma (RMA).
BACKGROUND
RMA is a rare histological subtype with an impaired response to chemoradiotherapy and an overall poor prognosis. High-grade tumors are associated with older age. Previous studies have shown conflicting results on prognosis.
METHODS
Retrospective analysis of National Cancer Database was conducted from 2004-2020 for subjects with histology code 8480 in primary sites C19 and C20 (rectosigmoid-junction and rectum, n = 14,044), using multivariate analysis with Cox regression.
RESULTS
Median age of diagnosis was 65 years with 69.5% were in the 45-75 years age range. 59.2% were male while 40.8% were female. 84.7% were White, 9.7% were Black, 0.4% were American Indian and 3.4% were Asian. 6.9% were Hispanic. 33.9% were in the upper-income quartile. 40.6% were seen at community cancer programs while 33% went to academic programs. 36.5% had stage III RMA. Out of the 14,044 patients with RMA, 10,546 received surgery, 5,179 received chemotherapy, 233 received immunotherapy and 55 received hormone therapy. Patients >75 years had significantly lower overall survival (OS) compared to those <45 years (HR 0.67). Female patients had significantly higher OS than male (HR - 0.07). Black patients had significantly lower OS than White (HR 0.08). Hispanic patients had significantly higher OS than non- Hispanic (HR - 0.14). Patients with private and government insurance had significantly higher OS than noninsured patients (HR - 0.35 and - 0.26 respectively). Patients with median higher-income quartiles had significantly higher OS than lower quartiles (HR - 0.13). Academic facilities had significantly higher OS than community programs (HR - 0.13). Patients who received surgery had significantly higher OS than those that did not (HR - 0.67); median survival for patients who received surgery was 71 months vs 28 months for non-surgical candidates.
CONCLUSIONS
Surgery is the most important treatment modality in RMA. Uninsured, older Black male patients from lower-income quartiles had significantly lower OS. Access to academic centers also contributed to differences in OS outcomes which throws light on healthcare disparities.
IMPLICATIONS
Additional studies need to be conducted for viable solutions to assist with social determinants of healthcare in RMA.
Comparison of Precision Oncology Annotation Services in the National Precision Oncology Program
BACKGROUND
The National Precision Oncology Program (NPOP) provides comprehensive genomic profiling (CGP) through external vendors to patients within the Veterans Affairs Healthcare System who meet testing guidelines. We sought to assess the concordance of cancer therapy recommendations between Foundation Medicine (FM), one of the NPOP vendors, and OncoKB, an FDA-recognized public precision oncology knowledge database, which annotates human genetic variants associated with therapies guidance at varying levels of evidence.
METHODS
We selected FM CGP test reports with at least one therapy recommendation regardless of FDA approval or level of evidence were selected to compare FM and OncoKB therapy annotations of different mutation types, including short variants (SVs), rearrangements, and copy number alterations (CNAs) between 02/01/2019-03/13/2023. Therapy recommendations of annotations for unique combinations of gene, variant, and cancer type from FM and OncoKB were compared. Comparisons were scored as an Exact Match (EM) if FM and OncoKB therapy annotation was the same or a Partial Match (PM) if the FM therapy annotation was a subset of OncoKB’s or vice versa.
RESULTS
For annotations involving FDA-approved therapies, a total of 10,435 cases were compared for SVs, 546 for rearrangements, and 732 for CNAs. Among SVs annotations, 7,029 (67.4%) were EM and 787 (7.5%) were PM. Of rearrangement annotations, 328 (60.1%) were EM and 95 (17.4%) were PM. Of CNA annotations, 469 (64.1%) were EM and 28 (3.8%) were PM. For off-label therapies, agreement between annotation sources was much lower in all above scenarios. Examples included 3022 (29%) cases were identified as EM plus PM for SVs, 324 (59.3%) for rearrangements, and 42 (5.7%) for CNAs.
CONCLUSIONS
Therapy recommendations were inconsistent between FM and OncoKB annotation services, with a substantial disagreement among both FDA-approved and off-label therapy annotations. The limitation of time difference of annotations performed between FM and OncoKB therapy annotations accounted for some disagreement. Establishing accuracy and improving concordance between different annotation services is needed to better match treatments to patients and improve provider trust and reliability of annotation service.
BACKGROUND
The National Precision Oncology Program (NPOP) provides comprehensive genomic profiling (CGP) through external vendors to patients within the Veterans Affairs Healthcare System who meet testing guidelines. We sought to assess the concordance of cancer therapy recommendations between Foundation Medicine (FM), one of the NPOP vendors, and OncoKB, an FDA-recognized public precision oncology knowledge database, which annotates human genetic variants associated with therapies guidance at varying levels of evidence.
METHODS
We selected FM CGP test reports with at least one therapy recommendation regardless of FDA approval or level of evidence were selected to compare FM and OncoKB therapy annotations of different mutation types, including short variants (SVs), rearrangements, and copy number alterations (CNAs) between 02/01/2019-03/13/2023. Therapy recommendations of annotations for unique combinations of gene, variant, and cancer type from FM and OncoKB were compared. Comparisons were scored as an Exact Match (EM) if FM and OncoKB therapy annotation was the same or a Partial Match (PM) if the FM therapy annotation was a subset of OncoKB’s or vice versa.
RESULTS
For annotations involving FDA-approved therapies, a total of 10,435 cases were compared for SVs, 546 for rearrangements, and 732 for CNAs. Among SVs annotations, 7,029 (67.4%) were EM and 787 (7.5%) were PM. Of rearrangement annotations, 328 (60.1%) were EM and 95 (17.4%) were PM. Of CNA annotations, 469 (64.1%) were EM and 28 (3.8%) were PM. For off-label therapies, agreement between annotation sources was much lower in all above scenarios. Examples included 3022 (29%) cases were identified as EM plus PM for SVs, 324 (59.3%) for rearrangements, and 42 (5.7%) for CNAs.
CONCLUSIONS
Therapy recommendations were inconsistent between FM and OncoKB annotation services, with a substantial disagreement among both FDA-approved and off-label therapy annotations. The limitation of time difference of annotations performed between FM and OncoKB therapy annotations accounted for some disagreement. Establishing accuracy and improving concordance between different annotation services is needed to better match treatments to patients and improve provider trust and reliability of annotation service.
BACKGROUND
The National Precision Oncology Program (NPOP) provides comprehensive genomic profiling (CGP) through external vendors to patients within the Veterans Affairs Healthcare System who meet testing guidelines. We sought to assess the concordance of cancer therapy recommendations between Foundation Medicine (FM), one of the NPOP vendors, and OncoKB, an FDA-recognized public precision oncology knowledge database, which annotates human genetic variants associated with therapies guidance at varying levels of evidence.
METHODS
We selected FM CGP test reports with at least one therapy recommendation regardless of FDA approval or level of evidence were selected to compare FM and OncoKB therapy annotations of different mutation types, including short variants (SVs), rearrangements, and copy number alterations (CNAs) between 02/01/2019-03/13/2023. Therapy recommendations of annotations for unique combinations of gene, variant, and cancer type from FM and OncoKB were compared. Comparisons were scored as an Exact Match (EM) if FM and OncoKB therapy annotation was the same or a Partial Match (PM) if the FM therapy annotation was a subset of OncoKB’s or vice versa.
RESULTS
For annotations involving FDA-approved therapies, a total of 10,435 cases were compared for SVs, 546 for rearrangements, and 732 for CNAs. Among SVs annotations, 7,029 (67.4%) were EM and 787 (7.5%) were PM. Of rearrangement annotations, 328 (60.1%) were EM and 95 (17.4%) were PM. Of CNA annotations, 469 (64.1%) were EM and 28 (3.8%) were PM. For off-label therapies, agreement between annotation sources was much lower in all above scenarios. Examples included 3022 (29%) cases were identified as EM plus PM for SVs, 324 (59.3%) for rearrangements, and 42 (5.7%) for CNAs.
CONCLUSIONS
Therapy recommendations were inconsistent between FM and OncoKB annotation services, with a substantial disagreement among both FDA-approved and off-label therapy annotations. The limitation of time difference of annotations performed between FM and OncoKB therapy annotations accounted for some disagreement. Establishing accuracy and improving concordance between different annotation services is needed to better match treatments to patients and improve provider trust and reliability of annotation service.
Comparing Outcomes and Toxicities With Standard and Reduced Dose Melphalan in Autologous Stem Cell Transplant Patients With Multiple Myeloma
BACKGROUND
Multiple myeloma, an incurable plasma cell malignancy, has an average age of diagnosis over 65 years. For transplant-eligible patients, high-dose melphalan 200 mg/m2 (MEL200), followed by autologous stem cell rescue (ASCR) is the standard in consolidation therapy. Most clinical trials evaluating MEL200 with ASCR excluded patients over 65 due to concerns for toxicity and treatment-related mortality, leading to use of reduced dose melphalan 140 mg/m2 (MEL140) in clinical practice for older patients. As this dose has limited studies surrounding its reduction, the purpose of this study was to compare outcomes and toxicities of MEL140 in patients over the age of 65 to MEL200 in patients 65 and under.
METHODS
This single-center institutional review board approved retrospective study was conducted at VA Tennessee Valley Healthcare System. All multiple myeloma patients greater than 18 years of age who received melphalan with ASCR from January 1, 2018, to December 31, 2021, were included. Patients were divided into two arms: age < 65 treated with MEL200 and age >65 treated with MEL140. The primary endpoint was oneyear progression-free survival (PFS). The secondary endpoints were one-year overall survival (OS), treatment related mortality, time to neutrophil engraftment, and toxicities including febrile neutropenia, diarrhea, mucositis, infection, and intensive care unit transfers.
RESULTS
A total of 222 patients were included, 114 patients in the MEL200 arm and 108 patients in the MEL140 arm. The primary endpoint of one-year PFS had no significant difference, with 103 (90.4%) patients in the MEL200 group compared to 99 (91.7%) patients in the MEL140 group (p=0.732). Similarly, there was no statistically significant difference in the secondary endpoint of one-year OS with 112 (98.3%) patients in the MEL200 group compared to 106 (98.2%) in the MEL140 group (p=0.956). Toxicities were similar; however, grade 3 mucositis was higher in the MEL200 arm.
CONCLUSIONS
Our study found no difference in oneyear PFS or one-year OS when comparing MEL140 to MEL200 with minimal differences in regimen-related toxicities. Although not powered to detect statistical difference, results suggests that dose reduction with MEL140 in patients >65 years does not impact one-year PFS when compared to patients <65 receiving standard MEL200.
BACKGROUND
Multiple myeloma, an incurable plasma cell malignancy, has an average age of diagnosis over 65 years. For transplant-eligible patients, high-dose melphalan 200 mg/m2 (MEL200), followed by autologous stem cell rescue (ASCR) is the standard in consolidation therapy. Most clinical trials evaluating MEL200 with ASCR excluded patients over 65 due to concerns for toxicity and treatment-related mortality, leading to use of reduced dose melphalan 140 mg/m2 (MEL140) in clinical practice for older patients. As this dose has limited studies surrounding its reduction, the purpose of this study was to compare outcomes and toxicities of MEL140 in patients over the age of 65 to MEL200 in patients 65 and under.
METHODS
This single-center institutional review board approved retrospective study was conducted at VA Tennessee Valley Healthcare System. All multiple myeloma patients greater than 18 years of age who received melphalan with ASCR from January 1, 2018, to December 31, 2021, were included. Patients were divided into two arms: age < 65 treated with MEL200 and age >65 treated with MEL140. The primary endpoint was oneyear progression-free survival (PFS). The secondary endpoints were one-year overall survival (OS), treatment related mortality, time to neutrophil engraftment, and toxicities including febrile neutropenia, diarrhea, mucositis, infection, and intensive care unit transfers.
RESULTS
A total of 222 patients were included, 114 patients in the MEL200 arm and 108 patients in the MEL140 arm. The primary endpoint of one-year PFS had no significant difference, with 103 (90.4%) patients in the MEL200 group compared to 99 (91.7%) patients in the MEL140 group (p=0.732). Similarly, there was no statistically significant difference in the secondary endpoint of one-year OS with 112 (98.3%) patients in the MEL200 group compared to 106 (98.2%) in the MEL140 group (p=0.956). Toxicities were similar; however, grade 3 mucositis was higher in the MEL200 arm.
CONCLUSIONS
Our study found no difference in oneyear PFS or one-year OS when comparing MEL140 to MEL200 with minimal differences in regimen-related toxicities. Although not powered to detect statistical difference, results suggests that dose reduction with MEL140 in patients >65 years does not impact one-year PFS when compared to patients <65 receiving standard MEL200.
BACKGROUND
Multiple myeloma, an incurable plasma cell malignancy, has an average age of diagnosis over 65 years. For transplant-eligible patients, high-dose melphalan 200 mg/m2 (MEL200), followed by autologous stem cell rescue (ASCR) is the standard in consolidation therapy. Most clinical trials evaluating MEL200 with ASCR excluded patients over 65 due to concerns for toxicity and treatment-related mortality, leading to use of reduced dose melphalan 140 mg/m2 (MEL140) in clinical practice for older patients. As this dose has limited studies surrounding its reduction, the purpose of this study was to compare outcomes and toxicities of MEL140 in patients over the age of 65 to MEL200 in patients 65 and under.
METHODS
This single-center institutional review board approved retrospective study was conducted at VA Tennessee Valley Healthcare System. All multiple myeloma patients greater than 18 years of age who received melphalan with ASCR from January 1, 2018, to December 31, 2021, were included. Patients were divided into two arms: age < 65 treated with MEL200 and age >65 treated with MEL140. The primary endpoint was oneyear progression-free survival (PFS). The secondary endpoints were one-year overall survival (OS), treatment related mortality, time to neutrophil engraftment, and toxicities including febrile neutropenia, diarrhea, mucositis, infection, and intensive care unit transfers.
RESULTS
A total of 222 patients were included, 114 patients in the MEL200 arm and 108 patients in the MEL140 arm. The primary endpoint of one-year PFS had no significant difference, with 103 (90.4%) patients in the MEL200 group compared to 99 (91.7%) patients in the MEL140 group (p=0.732). Similarly, there was no statistically significant difference in the secondary endpoint of one-year OS with 112 (98.3%) patients in the MEL200 group compared to 106 (98.2%) in the MEL140 group (p=0.956). Toxicities were similar; however, grade 3 mucositis was higher in the MEL200 arm.
CONCLUSIONS
Our study found no difference in oneyear PFS or one-year OS when comparing MEL140 to MEL200 with minimal differences in regimen-related toxicities. Although not powered to detect statistical difference, results suggests that dose reduction with MEL140 in patients >65 years does not impact one-year PFS when compared to patients <65 receiving standard MEL200.
Quality Improvement Project of All Resected Lung Specimens for Pathologic Findings and Synoptic Surgical Reports for Accuracy in Staging: A Critical Review of 91 Specimens
BACKGROUND
In 2017, the Thoracic Tumor Board realized that there were patients whose lung resections had critical review of the slides and reports prior to presentation. Errors were found which resulted in a change of the pathology Tumor Nodal Metastases (pTNM) staging for the patient. The impacts were important for determining appropriate therapy. It was decided to systematically review all lung cancer resections for accuracy before determining definitive therapy recommendations.
METHODS
All lung resections for malignancy were examined prior and up to 2 days of completion for accuracy of tumor type, tumor size, tumor grade, lymph node metastases and pathologic stage (pTNM). Any errors found were given to the original pathologist for a change in the report before release or for a modified report to be issued.
RESULTS
From June 2017 to December 2020, there were 91 lung resections with 28 (30.77%) errors. Errors included: 16 incorrect pathologic staging, 5 missed tumors in lung and lymph nodes, 2 unexamined stapled surgical margins, 1 wrong site, 1 incorrect lymph node number and 2 missed tumor vascular invasion.
IMPLICATIONS
Quality improvement (QI) review of lung resections by a second pathologist is important and may clearly improve pathologic staging for lung cancer patients. It can be added to QI programs currently used in Surgical Pathology. It is important in directing appropriate postsurgical therapies.
BACKGROUND
In 2017, the Thoracic Tumor Board realized that there were patients whose lung resections had critical review of the slides and reports prior to presentation. Errors were found which resulted in a change of the pathology Tumor Nodal Metastases (pTNM) staging for the patient. The impacts were important for determining appropriate therapy. It was decided to systematically review all lung cancer resections for accuracy before determining definitive therapy recommendations.
METHODS
All lung resections for malignancy were examined prior and up to 2 days of completion for accuracy of tumor type, tumor size, tumor grade, lymph node metastases and pathologic stage (pTNM). Any errors found were given to the original pathologist for a change in the report before release or for a modified report to be issued.
RESULTS
From June 2017 to December 2020, there were 91 lung resections with 28 (30.77%) errors. Errors included: 16 incorrect pathologic staging, 5 missed tumors in lung and lymph nodes, 2 unexamined stapled surgical margins, 1 wrong site, 1 incorrect lymph node number and 2 missed tumor vascular invasion.
IMPLICATIONS
Quality improvement (QI) review of lung resections by a second pathologist is important and may clearly improve pathologic staging for lung cancer patients. It can be added to QI programs currently used in Surgical Pathology. It is important in directing appropriate postsurgical therapies.
BACKGROUND
In 2017, the Thoracic Tumor Board realized that there were patients whose lung resections had critical review of the slides and reports prior to presentation. Errors were found which resulted in a change of the pathology Tumor Nodal Metastases (pTNM) staging for the patient. The impacts were important for determining appropriate therapy. It was decided to systematically review all lung cancer resections for accuracy before determining definitive therapy recommendations.
METHODS
All lung resections for malignancy were examined prior and up to 2 days of completion for accuracy of tumor type, tumor size, tumor grade, lymph node metastases and pathologic stage (pTNM). Any errors found were given to the original pathologist for a change in the report before release or for a modified report to be issued.
RESULTS
From June 2017 to December 2020, there were 91 lung resections with 28 (30.77%) errors. Errors included: 16 incorrect pathologic staging, 5 missed tumors in lung and lymph nodes, 2 unexamined stapled surgical margins, 1 wrong site, 1 incorrect lymph node number and 2 missed tumor vascular invasion.
IMPLICATIONS
Quality improvement (QI) review of lung resections by a second pathologist is important and may clearly improve pathologic staging for lung cancer patients. It can be added to QI programs currently used in Surgical Pathology. It is important in directing appropriate postsurgical therapies.
A Multi-Disciplinary Approach to Increasing Germline Genetic Testing for Prostate Cancer
PURPOSE
This quality improvement project aims to enhance the rate of germline genetic testing for prostate cancer at the Stratton VA Medical Center, improving risk reduction strategies and therapeutic options for patients.
BACKGROUND
Prostate cancer is prevalent at the Stratton VA Medical Center, yet the rate of genetic evaluation for prostate cancer remains suboptimal. National guidelines recommend genetic counseling and testing in specific patient populations. To address this gap, an interdisciplinary working group conducted gap analysis and root cause analysis, identifying four significant barriers.
METHODS
The working group comprised medical oncologists, urologists, primary care physicians, genetics counselors, data experts, and a LEAN coach. Interventions included implementing a prostate cancer pathway to educate staff on genetic testing indications and integrating genetic testing screening into clinic visits. After the interventions were implemented in January 2022, patient charts were reviewed for all genetic referrals and new prostate cancer diagnoses from January to December 2022.
DATA ANALYSIS
Descriptive analysis was conducted on referral rates, evaluation visit completion rates, and genetic testing outcomes among prostate cancer patients.
RESULTS
During the study period, 59 prostate cancer patients were referred for genetic evaluation. Notably, this was a large increase from no genetic referrals for prostate cancer in the previous year. Among them, 43 completed the evaluation visit, and 34 underwent genetic testing. Noteworthy findings were observed in 5 patients, including 3 variants of unknown significance and 2 pathogenic germline variants: HOXB13 and BRCA2 mutations.
IMPLICATIONS
This project highlights the power of a collaborative, multidisciplinary approach to overcome barriers and enhance the quality of care for prostate cancer patients. The team’s use of gap analysis and root cause analysis successfully identified barriers and proposed solutions, leading to increased referrals and the identification of significant genetic findings. Continued efforts to improve access to germline genetic testing are crucial for enhanced patient care and improved outcomes.
PURPOSE
This quality improvement project aims to enhance the rate of germline genetic testing for prostate cancer at the Stratton VA Medical Center, improving risk reduction strategies and therapeutic options for patients.
BACKGROUND
Prostate cancer is prevalent at the Stratton VA Medical Center, yet the rate of genetic evaluation for prostate cancer remains suboptimal. National guidelines recommend genetic counseling and testing in specific patient populations. To address this gap, an interdisciplinary working group conducted gap analysis and root cause analysis, identifying four significant barriers.
METHODS
The working group comprised medical oncologists, urologists, primary care physicians, genetics counselors, data experts, and a LEAN coach. Interventions included implementing a prostate cancer pathway to educate staff on genetic testing indications and integrating genetic testing screening into clinic visits. After the interventions were implemented in January 2022, patient charts were reviewed for all genetic referrals and new prostate cancer diagnoses from January to December 2022.
DATA ANALYSIS
Descriptive analysis was conducted on referral rates, evaluation visit completion rates, and genetic testing outcomes among prostate cancer patients.
RESULTS
During the study period, 59 prostate cancer patients were referred for genetic evaluation. Notably, this was a large increase from no genetic referrals for prostate cancer in the previous year. Among them, 43 completed the evaluation visit, and 34 underwent genetic testing. Noteworthy findings were observed in 5 patients, including 3 variants of unknown significance and 2 pathogenic germline variants: HOXB13 and BRCA2 mutations.
IMPLICATIONS
This project highlights the power of a collaborative, multidisciplinary approach to overcome barriers and enhance the quality of care for prostate cancer patients. The team’s use of gap analysis and root cause analysis successfully identified barriers and proposed solutions, leading to increased referrals and the identification of significant genetic findings. Continued efforts to improve access to germline genetic testing are crucial for enhanced patient care and improved outcomes.
PURPOSE
This quality improvement project aims to enhance the rate of germline genetic testing for prostate cancer at the Stratton VA Medical Center, improving risk reduction strategies and therapeutic options for patients.
BACKGROUND
Prostate cancer is prevalent at the Stratton VA Medical Center, yet the rate of genetic evaluation for prostate cancer remains suboptimal. National guidelines recommend genetic counseling and testing in specific patient populations. To address this gap, an interdisciplinary working group conducted gap analysis and root cause analysis, identifying four significant barriers.
METHODS
The working group comprised medical oncologists, urologists, primary care physicians, genetics counselors, data experts, and a LEAN coach. Interventions included implementing a prostate cancer pathway to educate staff on genetic testing indications and integrating genetic testing screening into clinic visits. After the interventions were implemented in January 2022, patient charts were reviewed for all genetic referrals and new prostate cancer diagnoses from January to December 2022.
DATA ANALYSIS
Descriptive analysis was conducted on referral rates, evaluation visit completion rates, and genetic testing outcomes among prostate cancer patients.
RESULTS
During the study period, 59 prostate cancer patients were referred for genetic evaluation. Notably, this was a large increase from no genetic referrals for prostate cancer in the previous year. Among them, 43 completed the evaluation visit, and 34 underwent genetic testing. Noteworthy findings were observed in 5 patients, including 3 variants of unknown significance and 2 pathogenic germline variants: HOXB13 and BRCA2 mutations.
IMPLICATIONS
This project highlights the power of a collaborative, multidisciplinary approach to overcome barriers and enhance the quality of care for prostate cancer patients. The team’s use of gap analysis and root cause analysis successfully identified barriers and proposed solutions, leading to increased referrals and the identification of significant genetic findings. Continued efforts to improve access to germline genetic testing are crucial for enhanced patient care and improved outcomes.
Results From the First Annual Association of Professors of Dermatology Program Directors Survey
Educational organizations across several specialties, including internal medicine and obstetrics and gynecology, have formal surveys1; however, the field of dermatology has been without one. This study aimed to establish a formal survey for dermatology program directors (PDs) and clinician-educators. Because the Accreditation Council for Graduate Medical Education (ACGME) and American Board of Dermatology surveys do not capture all metrics relevant to dermatology residency educators, an annual survey for our specialty may be helpful to compare dermatology-specific data among programs. Responses could provide context and perspective to faculty and residents who respond to the ACGME annual survey, as our Association of Professors of Dermatology (APD) survey asks more in-depth questions, such as how often didactics occur and who leads them. Resident commute time and faculty demographics and training also are covered. Current ad hoc surveys disseminated through listserves of various medical associations contain overlapping questions and reflect relatively low response rates; dermatology PDs may benefit from a survey with a high response rate to which they can contribute future questions and topics that reflect recent trends and current needs in graduate medical education. As future surveys are administered, the results can be captured in a centralized database accessible by dermatology PDs.
Methods
A survey of PDs from 141 ACGME-accredited dermatology residency programs was conducted by the Residency Program Director Steering Committee of the APD from November 2022 to January 2023 using a prevalidated questionnaire. Personalized survey links were created and sent individually to each PD’s email listed in the ACGME accreditation data system. All survey responses were captured anonymously, with a number assigned to keep de-identified responses separate and organized. The survey consisted of 137 survey questions addressing topics that included program characteristics, PD demographics, the impact of the COVID-19 pandemic on clinical rotation and educational conferences, available resident resources, quality improvement, clinical and didactic instruction, research content, diversity and inclusion, wellness, professionalism, evaluation systems, and graduate outcomes.
Data were collected using Qualtrics survey tools. After removing duplicate and incomplete surveys, data were analyzed using Qualtrics reports and Microsoft Excel for data plotting, averages, and range calculations.
Results
One hundred forty-one personalized survey links were created and sent individually to each program’s filed email obtained from the APD listserv. Fifty-three responses were recorded after removing duplicate or incomplete surveys (38% [53/141] response rate). As of May 2023, there were 144 ACGME-accredited dermatology residency programs due to 3 newly accredited programs in 2022-2023 academic year, which were not included in our survey population.
Program Characteristics—Forty-four respondents (83%) were from a university-based program. Fifty respondents (94%) were from programs that were ACGME accredited prior to 2020, while 3 programs (6%) were American Osteopathic Association accredited prior to singular accreditation. Seventy-one percent (38/53) of respondents had 1 or more associate PDs.
PD Demographics—Eighty-seven percent (45/52) of PDs who responded to the survey graduated from a US allopathic medical school (MD), 10% (5/52) graduated from a US osteopathic medical school (DO), and 4% (2/52) graduated from an international medical school. Seventy-four percent (35/47) of respondents were White, 17% (8/47) were Asian, and 2% (1/47) were Black or African American; this data was not provided for 4 respondents. Forty-eight percent (23/48) of PDs identified as cisgender man, 48% (23/48) identified as cisgender woman, and 4% (2/48) preferred not to answer. Eighty-one percent (38/47) of PDs identified as heterosexual or straight, 15% (7/47) identified as gay or lesbian, and 4% (2/47) preferred not to answer.
Impact of COVID-19 Pandemic on Residency Training—Due to the COVID-19 pandemic, 88% (45/51) of respondents incorporated telemedicine into the resident clinical rotation schedule. Moving forward, 75% (38/51) of respondents indicated that their programs plan to continue to incorporate telemedicine into the rotation schedule. Based on 50 responses, the average of educational conferences that became virtual at the start of the COVID-19 pandemic was 87%; based on 46 responses, the percentage of educational conferences that will remain virtual moving forward is 46%, while 90% (46/51) of respondents indicated that their programs plan to use virtual conferences in some capacity moving forward. Seventy-three percent (37/51) of respondents indicated that they plan to use virtual interviews as part of residency recruitment moving forward.
Available Resources—Twenty-four percent (11/46) of respondents indicated that residents in their program do not get protected time or time off for CORE examinations. Seventy-five percent (33/44) of PDs said their program provides funding for residents to participate in board review courses. The chief residents at 63% (31/49) of programs receive additional compensation, and 69% (34/49) provide additional administrative time to chief residents. Seventy-one percent (24/34) of PDs reported their programs have scribes for attendings, and 12% (4/34) have scribes for residents. Support staff help residents with callbacks and in-basket messages according to 76% (35/46) of respondents. The majority (98% [45/46]) of PDs indicated that residents follow-up on results and messages from patients seen in resident clinics, and 43% (20/46) of programs have residents follow-up with patients seen in faculty clinics. Only 15% (7/46) of PDs responded they have schedules with residents dedicated to handle these tasks. According to respondents, 33% (17/52) have residents who are required to travel more than 25 miles to distant clinical sites. Of them, 35% (6/17) provide accommodations.
Quality Improvement—Seventy-one percent (35/49) of respondents indicated their department has a quality improvement/patient safety team or committee, and 94% (33/35) of these teams include residents. A lecture series on quality improvement and patient safety is offered at 67% (33/49) of the respondents’ programs, while morbidity and mortality conferences are offered in 73% (36/49).
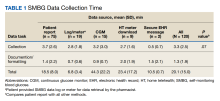
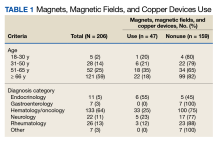
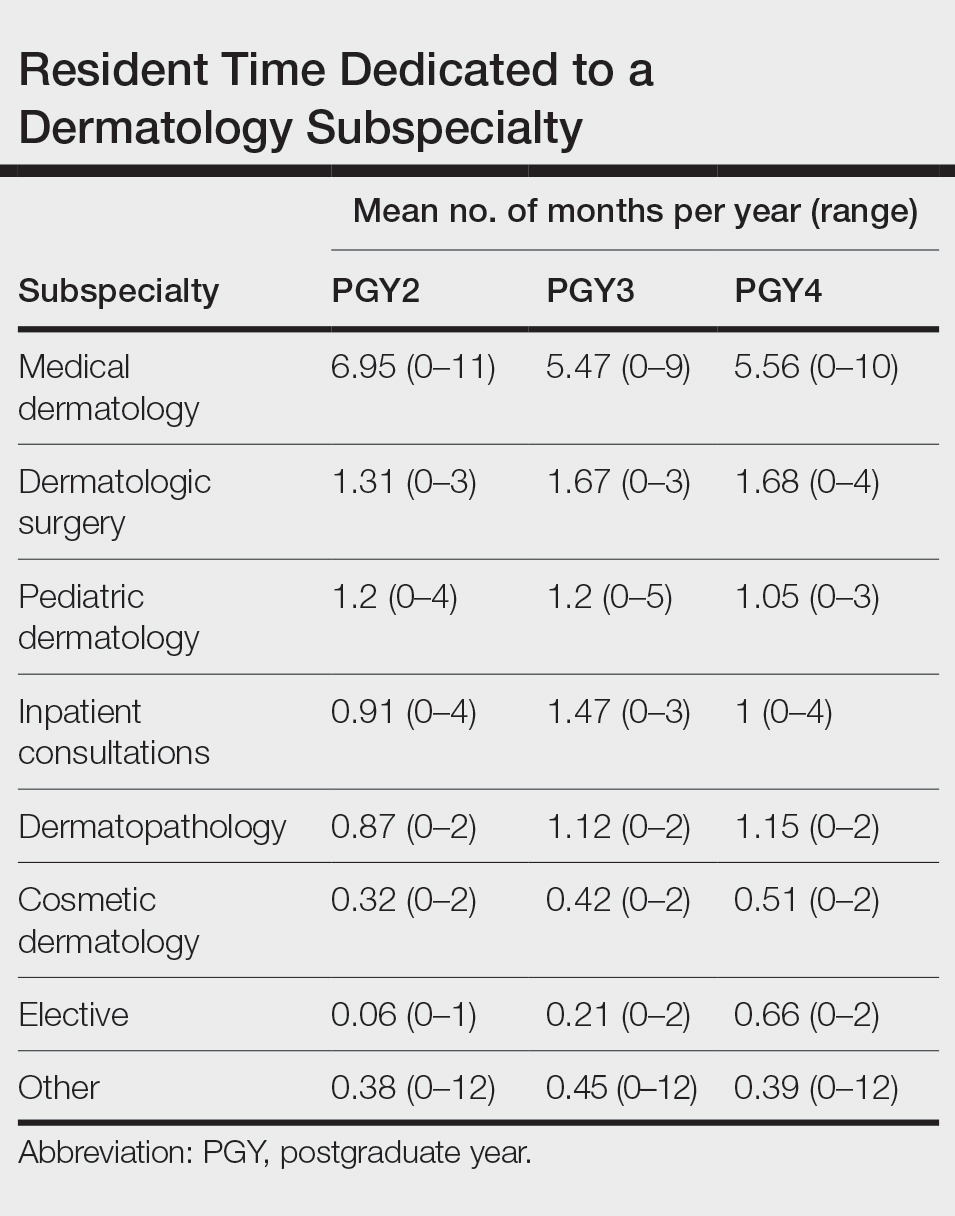
Clinical Instruction—Our survey asked PDs how many months each residency year spends on a certain rotational service. Based on 46 respondents, the average number of months dedicated to medical dermatology is 7, 5, and 6 months for postgraduate year (PGY) 2, PGY3, and PGY4, respectively. The average number of months spent in other subspecialties is provided in the Table. On average, PGY2 residents spend 8 half-days per week seeing patients in clinic, while PGY3 and PGY4 residents see patients for 7 half-days. The median and mean number of patients staffed by a single attending per hour in teaching clinics are 6 and 5.88, respectively. Respondents indicated that residents participate in the following specialty clinics: pediatric dermatology (96% [44/46]), laser/cosmetic (87% [40/44]), high-risk skin cancer (ie, immunosuppressed/transplant patient)(65% [30/44]), pigmented lesion/melanoma (52% [24/44]), connective tissue disease (52% [24/44]), teledermatology (50% [23/44]), free clinic for homeless and/or indigent populations (48% [22/44]), contact dermatitis (43% [20/44]), skin of color (43% [20/44]), oncodermatology (41% [19/44]), and bullous disease (33% [15/44]).
Additionally, in 87% (40/46) of programs, residents participate in a dedicated inpatient consultation service. Most respondents (98% [45/46]) responded that they utilize in-person consultations with a teledermatology supplement. Fifteen percent (7/46) utilize virtual teledermatology (live video-based consultations), and 57% (26/46) utilize asynchronous teledermatology (picture-based consultations). All respondents (n=46) indicated that 0% to 25% of patient encounters involving residents are teledermatology visits. Thirty-three percent (6/18) of programs have a global health special training track, 56% (10/18) have a Specialty Training and Advanced Research/Physician-Scientist Research Training track, 28% (5/18) have a diversity training track, and 50% (9/18) have a clinician educator training track.
Didactic Instruction—Five programs have a full day per week dedicated to didactics, while 36 programs have at least one half-day per week for didactics. On average, didactics in 57% (26/46) of programs are led by faculty alone, while 43% (20/46) are led at least in part by residents or fellows.
Research Content—Fifty percent (23/46) of programs have a specific research requirement for residents beyond general ACGME requirements, and 35% (16/46) require residents to participate in a longitudinal research project over the course of residency. There is a dedicated research coordinator for resident support at 63% (29/46) of programs. Dedicated biostatistics research support is available for resident projects at 42% (19/45) of programs. Additionally, at 42% (19/45) of programs, there is a dedicated faculty member for oversight of resident research.

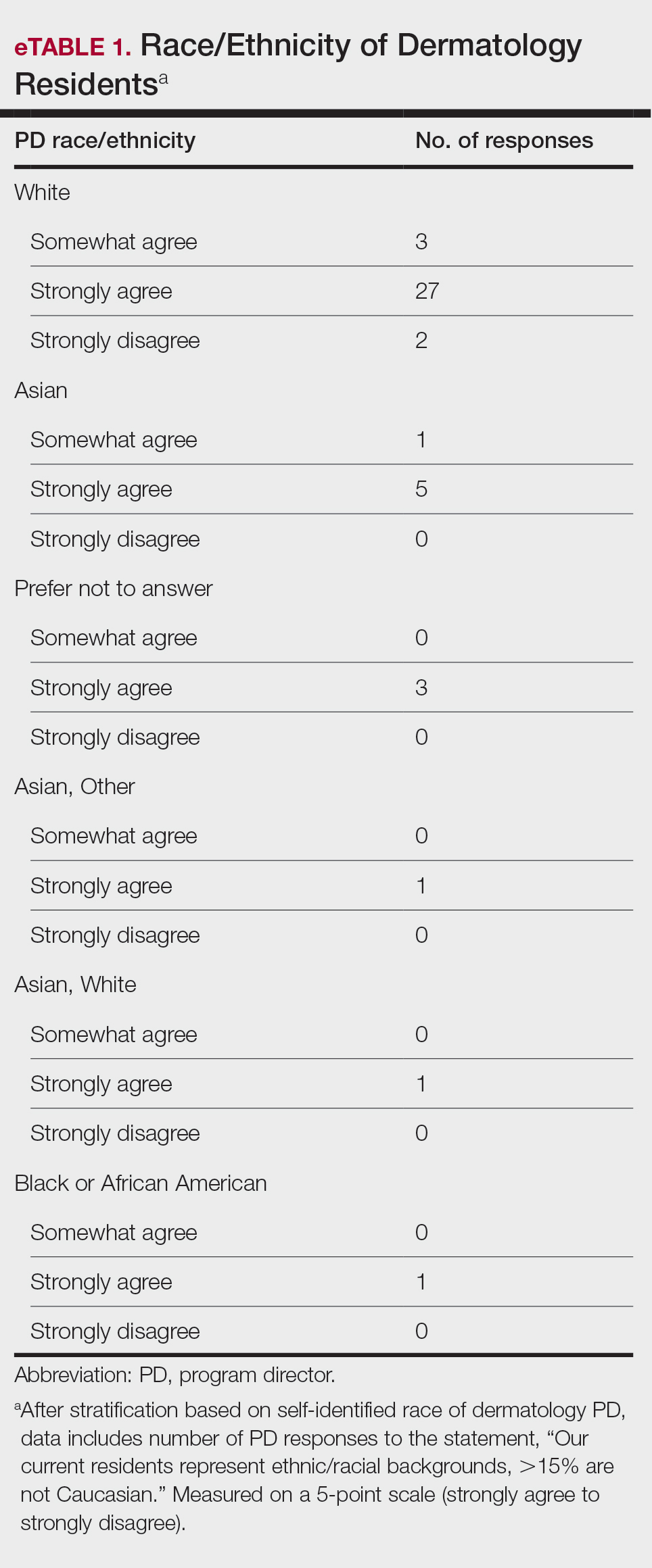
Diversity, Equity, and Inclusion—Seventy-three percent (29/40) of programs have special diversity, equity, and inclusion programs or meetings specific to residency, 60% (24/40) have residency initiatives, and 55% (22/40) have a residency diversity committee. Eighty-six percent (42/49) of respondents strongly agreed that their current residents represent diverse ethnic and racial backgrounds (ie, >15% are not White). eTable 1 shows PD responses to this statement, which were stratified based on self-identified race. eTable 2 shows PD responses to the statement, “Our current residents represent an inclusion of gender/sexual orientation,” which were stratified based on self-identified gender identity/sexual orientation. Lastly, eTable 3 highlights the percentage of residents with an MD and DO degree, stratified based on PD degree.

Wellness—Forty-eight percent (20/42) of respondents indicated they are under stress and do not always have as much energy as before becoming a PD but do not feel burned out. Thirty-one percent (13/42) indicated they have 1 or more symptoms of burnout, such as emotional exhaustion. Eighty-six percent (36/42) are satisfied with their jobs overall (43% agree and 43% strongly agree [18/42 each]).
Evaluation System—Seventy-five percent (33/44) of programs deliver evaluations of residents by faculty online, 86% (38/44) of programs have PDs discuss evaluations in-person, and 20% (9/44) of programs have faculty evaluators discuss evaluations in-person. Seventy-seven percent (34/44) of programs have formal faculty-resident mentor-mentee programs. Clinical competency committee chair positions are filled by PDs, assistant PDs, or core faculty members 47%, 38%, and 16% of the time, respectively.
Graduation Outcomes of PGY4 Residents—About 28% (55/199) of graduating residents applied to a fellowship position, with the majority (15% [29/55]) matching into Mohs micrographic surgery and dermatologic oncology (MSDO) fellowships. Approximately 5% (9/199) and 4% (7/199) of graduates matched into dermatopathology and pediatric dermatology, respectively. The remaining 5% (10/199) of graduating residents applied to a fellowship but did not match. The majority (45% [91/199]) of residency graduates entered private practice after graduation. Approximately 21% (42/199) of graduating residents chose an academic practice with 17% (33/199), 2% (4/199), and 2% (3/199) of those positions being full-time, part-time, and adjunct, respectively.
Comment
The first annual APD survey is a novel data source and provides opportunities for areas of discussion and investigation. Evaluating the similarities and differences among dermatology residency programs across the United States can strengthen individual programs through collaboration and provide areas of cohesion among programs.
Diversity of PDs—An important area of discussion is diversity and PD demographics. Although DO students make up 1 in 4 US graduating medical students, they are not interviewed or ranked as often as MD students.2 Diversity in PD race and ethnicity may be worthy of investigation in future studies, as match rates and recruitment of diverse medical school applicants may be impacted by these demographics.
Continued Use of Telemedicine in Training—Since 2020, the benefits of virtual residency recruitment have been debated among PDs across all medical specialties. Points in favor of virtual interviews include cost savings for programs and especially for applicants, as well as time efficiency, reduced burden of travel, and reduced carbon footprint. A problem posed by virtual interviews is that candidates are unable to fully learn institutional cultures and social environments of the programs.3 Likewise, telehealth was an important means of clinical teaching for residents during the height of the COVID-19 pandemic, with benefits that included cost-effectiveness and reduction of disparities in access to dermatologic care.4 Seventy-five percent (38/51) of PDs indicated that their program plans to include telemedicine in resident clinical rotation moving forward.
Resources Available—Our survey showed that resources available for residents, delivery of lectures and program time allocated to didactics, protected academic or study time for residents, and allocation of program time for CORE examinations are highly variable across programs. This could inspire future studies to be done to determine the differences in success of the resident on CORE examinations and in digesting material.
Postgraduate Career Plans and Fellowship Matches—Residents of programs that have a home MSDO fellowship are more likely to successfully match into a MSDO fellowship.5 Based on this survey, approximately 28% of graduating residents applied to a fellowship position, with 15%, 5%, and 3% matching into desired MSDO, dermatopathology, and pediatric dermatology fellowships, respectively. Additional studies are needed to determine advantages and disadvantages that lead to residents reaching their career goals.
Limitations—Limitations of this study include a small sample size that may not adequately represent all ACGME-accredited dermatology residency programs and selection bias toward respondents who are more likely to participate in survey-based research.
Conclusion
The APD plans to continue to administer this survey on an annual basis, with updates to the content and questions based on input from PDs. This survey will continue to provide valuable information to drive collaboration among residency programs and optimize the learning experience for residents. Our hope is that the response rate will increase in coming years, allowing us to draw more generalizable conclusions. Nonetheless, the survey data allow individual dermatology residency programs to compare their specific characteristics to other programs.
- Maciejko L, Cope A, Mara K, et al. A national survey of obstetrics and gynecology emergency training and deficits in office emergency preparation [A53]. Obstet Gynecol. 2022;139:16S. doi:10.1097/01.AOG.0000826548.05758.26
- Lavertue SM, Terry R. A comparison of surgical subspecialty match rates in 2022 in the United States. Cureus. 2023;15:E37178. doi:10.7759/cureus.37178
- Domingo A, Rdesinski RE, Stenson A, et al. Virtual residency interviews: applicant perceptions regarding virtual interview effectiveness, advantages, and barriers. J Grad Med Educ. 2022;14:224-228. doi:10.4300/JGME-D-21-00675.1
- Rustad AM, Lio PA. Pandemic pressure: teledermatology and health care disparities. J Patient Exp. 2021;8:2374373521996982. doi:10.1177/2374373521996982
- Rickstrew J, Rajpara A, Hocker TLH. Dermatology residency program influences chance of successful surgery fellowship match. Dermatol Surg. 2021;47:1040-1042. doi:10.1097/DSS.0000000000002859
Educational organizations across several specialties, including internal medicine and obstetrics and gynecology, have formal surveys1; however, the field of dermatology has been without one. This study aimed to establish a formal survey for dermatology program directors (PDs) and clinician-educators. Because the Accreditation Council for Graduate Medical Education (ACGME) and American Board of Dermatology surveys do not capture all metrics relevant to dermatology residency educators, an annual survey for our specialty may be helpful to compare dermatology-specific data among programs. Responses could provide context and perspective to faculty and residents who respond to the ACGME annual survey, as our Association of Professors of Dermatology (APD) survey asks more in-depth questions, such as how often didactics occur and who leads them. Resident commute time and faculty demographics and training also are covered. Current ad hoc surveys disseminated through listserves of various medical associations contain overlapping questions and reflect relatively low response rates; dermatology PDs may benefit from a survey with a high response rate to which they can contribute future questions and topics that reflect recent trends and current needs in graduate medical education. As future surveys are administered, the results can be captured in a centralized database accessible by dermatology PDs.
Methods
A survey of PDs from 141 ACGME-accredited dermatology residency programs was conducted by the Residency Program Director Steering Committee of the APD from November 2022 to January 2023 using a prevalidated questionnaire. Personalized survey links were created and sent individually to each PD’s email listed in the ACGME accreditation data system. All survey responses were captured anonymously, with a number assigned to keep de-identified responses separate and organized. The survey consisted of 137 survey questions addressing topics that included program characteristics, PD demographics, the impact of the COVID-19 pandemic on clinical rotation and educational conferences, available resident resources, quality improvement, clinical and didactic instruction, research content, diversity and inclusion, wellness, professionalism, evaluation systems, and graduate outcomes.
Data were collected using Qualtrics survey tools. After removing duplicate and incomplete surveys, data were analyzed using Qualtrics reports and Microsoft Excel for data plotting, averages, and range calculations.
Results
One hundred forty-one personalized survey links were created and sent individually to each program’s filed email obtained from the APD listserv. Fifty-three responses were recorded after removing duplicate or incomplete surveys (38% [53/141] response rate). As of May 2023, there were 144 ACGME-accredited dermatology residency programs due to 3 newly accredited programs in 2022-2023 academic year, which were not included in our survey population.
Program Characteristics—Forty-four respondents (83%) were from a university-based program. Fifty respondents (94%) were from programs that were ACGME accredited prior to 2020, while 3 programs (6%) were American Osteopathic Association accredited prior to singular accreditation. Seventy-one percent (38/53) of respondents had 1 or more associate PDs.
PD Demographics—Eighty-seven percent (45/52) of PDs who responded to the survey graduated from a US allopathic medical school (MD), 10% (5/52) graduated from a US osteopathic medical school (DO), and 4% (2/52) graduated from an international medical school. Seventy-four percent (35/47) of respondents were White, 17% (8/47) were Asian, and 2% (1/47) were Black or African American; this data was not provided for 4 respondents. Forty-eight percent (23/48) of PDs identified as cisgender man, 48% (23/48) identified as cisgender woman, and 4% (2/48) preferred not to answer. Eighty-one percent (38/47) of PDs identified as heterosexual or straight, 15% (7/47) identified as gay or lesbian, and 4% (2/47) preferred not to answer.
Impact of COVID-19 Pandemic on Residency Training—Due to the COVID-19 pandemic, 88% (45/51) of respondents incorporated telemedicine into the resident clinical rotation schedule. Moving forward, 75% (38/51) of respondents indicated that their programs plan to continue to incorporate telemedicine into the rotation schedule. Based on 50 responses, the average of educational conferences that became virtual at the start of the COVID-19 pandemic was 87%; based on 46 responses, the percentage of educational conferences that will remain virtual moving forward is 46%, while 90% (46/51) of respondents indicated that their programs plan to use virtual conferences in some capacity moving forward. Seventy-three percent (37/51) of respondents indicated that they plan to use virtual interviews as part of residency recruitment moving forward.
Available Resources—Twenty-four percent (11/46) of respondents indicated that residents in their program do not get protected time or time off for CORE examinations. Seventy-five percent (33/44) of PDs said their program provides funding for residents to participate in board review courses. The chief residents at 63% (31/49) of programs receive additional compensation, and 69% (34/49) provide additional administrative time to chief residents. Seventy-one percent (24/34) of PDs reported their programs have scribes for attendings, and 12% (4/34) have scribes for residents. Support staff help residents with callbacks and in-basket messages according to 76% (35/46) of respondents. The majority (98% [45/46]) of PDs indicated that residents follow-up on results and messages from patients seen in resident clinics, and 43% (20/46) of programs have residents follow-up with patients seen in faculty clinics. Only 15% (7/46) of PDs responded they have schedules with residents dedicated to handle these tasks. According to respondents, 33% (17/52) have residents who are required to travel more than 25 miles to distant clinical sites. Of them, 35% (6/17) provide accommodations.
Quality Improvement—Seventy-one percent (35/49) of respondents indicated their department has a quality improvement/patient safety team or committee, and 94% (33/35) of these teams include residents. A lecture series on quality improvement and patient safety is offered at 67% (33/49) of the respondents’ programs, while morbidity and mortality conferences are offered in 73% (36/49).
Clinical Instruction—Our survey asked PDs how many months each residency year spends on a certain rotational service. Based on 46 respondents, the average number of months dedicated to medical dermatology is 7, 5, and 6 months for postgraduate year (PGY) 2, PGY3, and PGY4, respectively. The average number of months spent in other subspecialties is provided in the Table. On average, PGY2 residents spend 8 half-days per week seeing patients in clinic, while PGY3 and PGY4 residents see patients for 7 half-days. The median and mean number of patients staffed by a single attending per hour in teaching clinics are 6 and 5.88, respectively. Respondents indicated that residents participate in the following specialty clinics: pediatric dermatology (96% [44/46]), laser/cosmetic (87% [40/44]), high-risk skin cancer (ie, immunosuppressed/transplant patient)(65% [30/44]), pigmented lesion/melanoma (52% [24/44]), connective tissue disease (52% [24/44]), teledermatology (50% [23/44]), free clinic for homeless and/or indigent populations (48% [22/44]), contact dermatitis (43% [20/44]), skin of color (43% [20/44]), oncodermatology (41% [19/44]), and bullous disease (33% [15/44]).

Additionally, in 87% (40/46) of programs, residents participate in a dedicated inpatient consultation service. Most respondents (98% [45/46]) responded that they utilize in-person consultations with a teledermatology supplement. Fifteen percent (7/46) utilize virtual teledermatology (live video-based consultations), and 57% (26/46) utilize asynchronous teledermatology (picture-based consultations). All respondents (n=46) indicated that 0% to 25% of patient encounters involving residents are teledermatology visits. Thirty-three percent (6/18) of programs have a global health special training track, 56% (10/18) have a Specialty Training and Advanced Research/Physician-Scientist Research Training track, 28% (5/18) have a diversity training track, and 50% (9/18) have a clinician educator training track.
Didactic Instruction—Five programs have a full day per week dedicated to didactics, while 36 programs have at least one half-day per week for didactics. On average, didactics in 57% (26/46) of programs are led by faculty alone, while 43% (20/46) are led at least in part by residents or fellows.

Research Content—Fifty percent (23/46) of programs have a specific research requirement for residents beyond general ACGME requirements, and 35% (16/46) require residents to participate in a longitudinal research project over the course of residency. There is a dedicated research coordinator for resident support at 63% (29/46) of programs. Dedicated biostatistics research support is available for resident projects at 42% (19/45) of programs. Additionally, at 42% (19/45) of programs, there is a dedicated faculty member for oversight of resident research.

Diversity, Equity, and Inclusion—Seventy-three percent (29/40) of programs have special diversity, equity, and inclusion programs or meetings specific to residency, 60% (24/40) have residency initiatives, and 55% (22/40) have a residency diversity committee. Eighty-six percent (42/49) of respondents strongly agreed that their current residents represent diverse ethnic and racial backgrounds (ie, >15% are not White). eTable 1 shows PD responses to this statement, which were stratified based on self-identified race. eTable 2 shows PD responses to the statement, “Our current residents represent an inclusion of gender/sexual orientation,” which were stratified based on self-identified gender identity/sexual orientation. Lastly, eTable 3 highlights the percentage of residents with an MD and DO degree, stratified based on PD degree.

Wellness—Forty-eight percent (20/42) of respondents indicated they are under stress and do not always have as much energy as before becoming a PD but do not feel burned out. Thirty-one percent (13/42) indicated they have 1 or more symptoms of burnout, such as emotional exhaustion. Eighty-six percent (36/42) are satisfied with their jobs overall (43% agree and 43% strongly agree [18/42 each]).
Evaluation System—Seventy-five percent (33/44) of programs deliver evaluations of residents by faculty online, 86% (38/44) of programs have PDs discuss evaluations in-person, and 20% (9/44) of programs have faculty evaluators discuss evaluations in-person. Seventy-seven percent (34/44) of programs have formal faculty-resident mentor-mentee programs. Clinical competency committee chair positions are filled by PDs, assistant PDs, or core faculty members 47%, 38%, and 16% of the time, respectively.
Graduation Outcomes of PGY4 Residents—About 28% (55/199) of graduating residents applied to a fellowship position, with the majority (15% [29/55]) matching into Mohs micrographic surgery and dermatologic oncology (MSDO) fellowships. Approximately 5% (9/199) and 4% (7/199) of graduates matched into dermatopathology and pediatric dermatology, respectively. The remaining 5% (10/199) of graduating residents applied to a fellowship but did not match. The majority (45% [91/199]) of residency graduates entered private practice after graduation. Approximately 21% (42/199) of graduating residents chose an academic practice with 17% (33/199), 2% (4/199), and 2% (3/199) of those positions being full-time, part-time, and adjunct, respectively.
Comment
The first annual APD survey is a novel data source and provides opportunities for areas of discussion and investigation. Evaluating the similarities and differences among dermatology residency programs across the United States can strengthen individual programs through collaboration and provide areas of cohesion among programs.
Diversity of PDs—An important area of discussion is diversity and PD demographics. Although DO students make up 1 in 4 US graduating medical students, they are not interviewed or ranked as often as MD students.2 Diversity in PD race and ethnicity may be worthy of investigation in future studies, as match rates and recruitment of diverse medical school applicants may be impacted by these demographics.
Continued Use of Telemedicine in Training—Since 2020, the benefits of virtual residency recruitment have been debated among PDs across all medical specialties. Points in favor of virtual interviews include cost savings for programs and especially for applicants, as well as time efficiency, reduced burden of travel, and reduced carbon footprint. A problem posed by virtual interviews is that candidates are unable to fully learn institutional cultures and social environments of the programs.3 Likewise, telehealth was an important means of clinical teaching for residents during the height of the COVID-19 pandemic, with benefits that included cost-effectiveness and reduction of disparities in access to dermatologic care.4 Seventy-five percent (38/51) of PDs indicated that their program plans to include telemedicine in resident clinical rotation moving forward.
Resources Available—Our survey showed that resources available for residents, delivery of lectures and program time allocated to didactics, protected academic or study time for residents, and allocation of program time for CORE examinations are highly variable across programs. This could inspire future studies to be done to determine the differences in success of the resident on CORE examinations and in digesting material.
Postgraduate Career Plans and Fellowship Matches—Residents of programs that have a home MSDO fellowship are more likely to successfully match into a MSDO fellowship.5 Based on this survey, approximately 28% of graduating residents applied to a fellowship position, with 15%, 5%, and 3% matching into desired MSDO, dermatopathology, and pediatric dermatology fellowships, respectively. Additional studies are needed to determine advantages and disadvantages that lead to residents reaching their career goals.
Limitations—Limitations of this study include a small sample size that may not adequately represent all ACGME-accredited dermatology residency programs and selection bias toward respondents who are more likely to participate in survey-based research.
Conclusion
The APD plans to continue to administer this survey on an annual basis, with updates to the content and questions based on input from PDs. This survey will continue to provide valuable information to drive collaboration among residency programs and optimize the learning experience for residents. Our hope is that the response rate will increase in coming years, allowing us to draw more generalizable conclusions. Nonetheless, the survey data allow individual dermatology residency programs to compare their specific characteristics to other programs.
Educational organizations across several specialties, including internal medicine and obstetrics and gynecology, have formal surveys1; however, the field of dermatology has been without one. This study aimed to establish a formal survey for dermatology program directors (PDs) and clinician-educators. Because the Accreditation Council for Graduate Medical Education (ACGME) and American Board of Dermatology surveys do not capture all metrics relevant to dermatology residency educators, an annual survey for our specialty may be helpful to compare dermatology-specific data among programs. Responses could provide context and perspective to faculty and residents who respond to the ACGME annual survey, as our Association of Professors of Dermatology (APD) survey asks more in-depth questions, such as how often didactics occur and who leads them. Resident commute time and faculty demographics and training also are covered. Current ad hoc surveys disseminated through listserves of various medical associations contain overlapping questions and reflect relatively low response rates; dermatology PDs may benefit from a survey with a high response rate to which they can contribute future questions and topics that reflect recent trends and current needs in graduate medical education. As future surveys are administered, the results can be captured in a centralized database accessible by dermatology PDs.
Methods
A survey of PDs from 141 ACGME-accredited dermatology residency programs was conducted by the Residency Program Director Steering Committee of the APD from November 2022 to January 2023 using a prevalidated questionnaire. Personalized survey links were created and sent individually to each PD’s email listed in the ACGME accreditation data system. All survey responses were captured anonymously, with a number assigned to keep de-identified responses separate and organized. The survey consisted of 137 survey questions addressing topics that included program characteristics, PD demographics, the impact of the COVID-19 pandemic on clinical rotation and educational conferences, available resident resources, quality improvement, clinical and didactic instruction, research content, diversity and inclusion, wellness, professionalism, evaluation systems, and graduate outcomes.
Data were collected using Qualtrics survey tools. After removing duplicate and incomplete surveys, data were analyzed using Qualtrics reports and Microsoft Excel for data plotting, averages, and range calculations.
Results
One hundred forty-one personalized survey links were created and sent individually to each program’s filed email obtained from the APD listserv. Fifty-three responses were recorded after removing duplicate or incomplete surveys (38% [53/141] response rate). As of May 2023, there were 144 ACGME-accredited dermatology residency programs due to 3 newly accredited programs in 2022-2023 academic year, which were not included in our survey population.
Program Characteristics—Forty-four respondents (83%) were from a university-based program. Fifty respondents (94%) were from programs that were ACGME accredited prior to 2020, while 3 programs (6%) were American Osteopathic Association accredited prior to singular accreditation. Seventy-one percent (38/53) of respondents had 1 or more associate PDs.
PD Demographics—Eighty-seven percent (45/52) of PDs who responded to the survey graduated from a US allopathic medical school (MD), 10% (5/52) graduated from a US osteopathic medical school (DO), and 4% (2/52) graduated from an international medical school. Seventy-four percent (35/47) of respondents were White, 17% (8/47) were Asian, and 2% (1/47) were Black or African American; this data was not provided for 4 respondents. Forty-eight percent (23/48) of PDs identified as cisgender man, 48% (23/48) identified as cisgender woman, and 4% (2/48) preferred not to answer. Eighty-one percent (38/47) of PDs identified as heterosexual or straight, 15% (7/47) identified as gay or lesbian, and 4% (2/47) preferred not to answer.
Impact of COVID-19 Pandemic on Residency Training—Due to the COVID-19 pandemic, 88% (45/51) of respondents incorporated telemedicine into the resident clinical rotation schedule. Moving forward, 75% (38/51) of respondents indicated that their programs plan to continue to incorporate telemedicine into the rotation schedule. Based on 50 responses, the average of educational conferences that became virtual at the start of the COVID-19 pandemic was 87%; based on 46 responses, the percentage of educational conferences that will remain virtual moving forward is 46%, while 90% (46/51) of respondents indicated that their programs plan to use virtual conferences in some capacity moving forward. Seventy-three percent (37/51) of respondents indicated that they plan to use virtual interviews as part of residency recruitment moving forward.
Available Resources—Twenty-four percent (11/46) of respondents indicated that residents in their program do not get protected time or time off for CORE examinations. Seventy-five percent (33/44) of PDs said their program provides funding for residents to participate in board review courses. The chief residents at 63% (31/49) of programs receive additional compensation, and 69% (34/49) provide additional administrative time to chief residents. Seventy-one percent (24/34) of PDs reported their programs have scribes for attendings, and 12% (4/34) have scribes for residents. Support staff help residents with callbacks and in-basket messages according to 76% (35/46) of respondents. The majority (98% [45/46]) of PDs indicated that residents follow-up on results and messages from patients seen in resident clinics, and 43% (20/46) of programs have residents follow-up with patients seen in faculty clinics. Only 15% (7/46) of PDs responded they have schedules with residents dedicated to handle these tasks. According to respondents, 33% (17/52) have residents who are required to travel more than 25 miles to distant clinical sites. Of them, 35% (6/17) provide accommodations.
Quality Improvement—Seventy-one percent (35/49) of respondents indicated their department has a quality improvement/patient safety team or committee, and 94% (33/35) of these teams include residents. A lecture series on quality improvement and patient safety is offered at 67% (33/49) of the respondents’ programs, while morbidity and mortality conferences are offered in 73% (36/49).
Clinical Instruction—Our survey asked PDs how many months each residency year spends on a certain rotational service. Based on 46 respondents, the average number of months dedicated to medical dermatology is 7, 5, and 6 months for postgraduate year (PGY) 2, PGY3, and PGY4, respectively. The average number of months spent in other subspecialties is provided in the Table. On average, PGY2 residents spend 8 half-days per week seeing patients in clinic, while PGY3 and PGY4 residents see patients for 7 half-days. The median and mean number of patients staffed by a single attending per hour in teaching clinics are 6 and 5.88, respectively. Respondents indicated that residents participate in the following specialty clinics: pediatric dermatology (96% [44/46]), laser/cosmetic (87% [40/44]), high-risk skin cancer (ie, immunosuppressed/transplant patient)(65% [30/44]), pigmented lesion/melanoma (52% [24/44]), connective tissue disease (52% [24/44]), teledermatology (50% [23/44]), free clinic for homeless and/or indigent populations (48% [22/44]), contact dermatitis (43% [20/44]), skin of color (43% [20/44]), oncodermatology (41% [19/44]), and bullous disease (33% [15/44]).

Additionally, in 87% (40/46) of programs, residents participate in a dedicated inpatient consultation service. Most respondents (98% [45/46]) responded that they utilize in-person consultations with a teledermatology supplement. Fifteen percent (7/46) utilize virtual teledermatology (live video-based consultations), and 57% (26/46) utilize asynchronous teledermatology (picture-based consultations). All respondents (n=46) indicated that 0% to 25% of patient encounters involving residents are teledermatology visits. Thirty-three percent (6/18) of programs have a global health special training track, 56% (10/18) have a Specialty Training and Advanced Research/Physician-Scientist Research Training track, 28% (5/18) have a diversity training track, and 50% (9/18) have a clinician educator training track.
Didactic Instruction—Five programs have a full day per week dedicated to didactics, while 36 programs have at least one half-day per week for didactics. On average, didactics in 57% (26/46) of programs are led by faculty alone, while 43% (20/46) are led at least in part by residents or fellows.

Research Content—Fifty percent (23/46) of programs have a specific research requirement for residents beyond general ACGME requirements, and 35% (16/46) require residents to participate in a longitudinal research project over the course of residency. There is a dedicated research coordinator for resident support at 63% (29/46) of programs. Dedicated biostatistics research support is available for resident projects at 42% (19/45) of programs. Additionally, at 42% (19/45) of programs, there is a dedicated faculty member for oversight of resident research.

Diversity, Equity, and Inclusion—Seventy-three percent (29/40) of programs have special diversity, equity, and inclusion programs or meetings specific to residency, 60% (24/40) have residency initiatives, and 55% (22/40) have a residency diversity committee. Eighty-six percent (42/49) of respondents strongly agreed that their current residents represent diverse ethnic and racial backgrounds (ie, >15% are not White). eTable 1 shows PD responses to this statement, which were stratified based on self-identified race. eTable 2 shows PD responses to the statement, “Our current residents represent an inclusion of gender/sexual orientation,” which were stratified based on self-identified gender identity/sexual orientation. Lastly, eTable 3 highlights the percentage of residents with an MD and DO degree, stratified based on PD degree.

Wellness—Forty-eight percent (20/42) of respondents indicated they are under stress and do not always have as much energy as before becoming a PD but do not feel burned out. Thirty-one percent (13/42) indicated they have 1 or more symptoms of burnout, such as emotional exhaustion. Eighty-six percent (36/42) are satisfied with their jobs overall (43% agree and 43% strongly agree [18/42 each]).
Evaluation System—Seventy-five percent (33/44) of programs deliver evaluations of residents by faculty online, 86% (38/44) of programs have PDs discuss evaluations in-person, and 20% (9/44) of programs have faculty evaluators discuss evaluations in-person. Seventy-seven percent (34/44) of programs have formal faculty-resident mentor-mentee programs. Clinical competency committee chair positions are filled by PDs, assistant PDs, or core faculty members 47%, 38%, and 16% of the time, respectively.
Graduation Outcomes of PGY4 Residents—About 28% (55/199) of graduating residents applied to a fellowship position, with the majority (15% [29/55]) matching into Mohs micrographic surgery and dermatologic oncology (MSDO) fellowships. Approximately 5% (9/199) and 4% (7/199) of graduates matched into dermatopathology and pediatric dermatology, respectively. The remaining 5% (10/199) of graduating residents applied to a fellowship but did not match. The majority (45% [91/199]) of residency graduates entered private practice after graduation. Approximately 21% (42/199) of graduating residents chose an academic practice with 17% (33/199), 2% (4/199), and 2% (3/199) of those positions being full-time, part-time, and adjunct, respectively.
Comment
The first annual APD survey is a novel data source and provides opportunities for areas of discussion and investigation. Evaluating the similarities and differences among dermatology residency programs across the United States can strengthen individual programs through collaboration and provide areas of cohesion among programs.
Diversity of PDs—An important area of discussion is diversity and PD demographics. Although DO students make up 1 in 4 US graduating medical students, they are not interviewed or ranked as often as MD students.2 Diversity in PD race and ethnicity may be worthy of investigation in future studies, as match rates and recruitment of diverse medical school applicants may be impacted by these demographics.
Continued Use of Telemedicine in Training—Since 2020, the benefits of virtual residency recruitment have been debated among PDs across all medical specialties. Points in favor of virtual interviews include cost savings for programs and especially for applicants, as well as time efficiency, reduced burden of travel, and reduced carbon footprint. A problem posed by virtual interviews is that candidates are unable to fully learn institutional cultures and social environments of the programs.3 Likewise, telehealth was an important means of clinical teaching for residents during the height of the COVID-19 pandemic, with benefits that included cost-effectiveness and reduction of disparities in access to dermatologic care.4 Seventy-five percent (38/51) of PDs indicated that their program plans to include telemedicine in resident clinical rotation moving forward.
Resources Available—Our survey showed that resources available for residents, delivery of lectures and program time allocated to didactics, protected academic or study time for residents, and allocation of program time for CORE examinations are highly variable across programs. This could inspire future studies to be done to determine the differences in success of the resident on CORE examinations and in digesting material.
Postgraduate Career Plans and Fellowship Matches—Residents of programs that have a home MSDO fellowship are more likely to successfully match into a MSDO fellowship.5 Based on this survey, approximately 28% of graduating residents applied to a fellowship position, with 15%, 5%, and 3% matching into desired MSDO, dermatopathology, and pediatric dermatology fellowships, respectively. Additional studies are needed to determine advantages and disadvantages that lead to residents reaching their career goals.
Limitations—Limitations of this study include a small sample size that may not adequately represent all ACGME-accredited dermatology residency programs and selection bias toward respondents who are more likely to participate in survey-based research.
Conclusion
The APD plans to continue to administer this survey on an annual basis, with updates to the content and questions based on input from PDs. This survey will continue to provide valuable information to drive collaboration among residency programs and optimize the learning experience for residents. Our hope is that the response rate will increase in coming years, allowing us to draw more generalizable conclusions. Nonetheless, the survey data allow individual dermatology residency programs to compare their specific characteristics to other programs.
- Maciejko L, Cope A, Mara K, et al. A national survey of obstetrics and gynecology emergency training and deficits in office emergency preparation [A53]. Obstet Gynecol. 2022;139:16S. doi:10.1097/01.AOG.0000826548.05758.26
- Lavertue SM, Terry R. A comparison of surgical subspecialty match rates in 2022 in the United States. Cureus. 2023;15:E37178. doi:10.7759/cureus.37178
- Domingo A, Rdesinski RE, Stenson A, et al. Virtual residency interviews: applicant perceptions regarding virtual interview effectiveness, advantages, and barriers. J Grad Med Educ. 2022;14:224-228. doi:10.4300/JGME-D-21-00675.1
- Rustad AM, Lio PA. Pandemic pressure: teledermatology and health care disparities. J Patient Exp. 2021;8:2374373521996982. doi:10.1177/2374373521996982
- Rickstrew J, Rajpara A, Hocker TLH. Dermatology residency program influences chance of successful surgery fellowship match. Dermatol Surg. 2021;47:1040-1042. doi:10.1097/DSS.0000000000002859
- Maciejko L, Cope A, Mara K, et al. A national survey of obstetrics and gynecology emergency training and deficits in office emergency preparation [A53]. Obstet Gynecol. 2022;139:16S. doi:10.1097/01.AOG.0000826548.05758.26
- Lavertue SM, Terry R. A comparison of surgical subspecialty match rates in 2022 in the United States. Cureus. 2023;15:E37178. doi:10.7759/cureus.37178
- Domingo A, Rdesinski RE, Stenson A, et al. Virtual residency interviews: applicant perceptions regarding virtual interview effectiveness, advantages, and barriers. J Grad Med Educ. 2022;14:224-228. doi:10.4300/JGME-D-21-00675.1
- Rustad AM, Lio PA. Pandemic pressure: teledermatology and health care disparities. J Patient Exp. 2021;8:2374373521996982. doi:10.1177/2374373521996982
- Rickstrew J, Rajpara A, Hocker TLH. Dermatology residency program influences chance of successful surgery fellowship match. Dermatol Surg. 2021;47:1040-1042. doi:10.1097/DSS.0000000000002859
Practice Points
- The first annual Association of Professors of Dermatology program directors survey allows faculty to compare their programs to other dermatology residency programs across the United States.
- The results should inspire opportunities for growth, improvement, and collaboration among dermatology residency programs.
Effect of COVID-19 Vaccination on Disease Severity in Patients With Stable Plaque Psoriasis: A Cross-sectional Study
To the Editor:
COVID-19 infection has resulted in 6.9 million deaths worldwide. India has the third highest mortality from COVID-19 infection after the United States and Brazil.1 Vaccination plays a crucial role in containing COVID-19 infection and reducing its severity. At present, 11 vaccines have been approved by the World Health Organization. India started its vaccination program on January 16, 2021, with approval for use of Covaxin (Bharat Biotech) and Covishield (Oxford/AstraZeneca formulation)(Serum Institute of India). More than 2 billion doses have been administered since then.2,3
Patients with psoriasis are prone to develop a severe form of COVID-19 due to comorbidities and the intake of immunosuppressive drugs.4 These patients often are hesitant to receive the vaccine without an expert opinion. COVID-19 vaccines are considered to increase tumor necrosis factor α (TNF-α) and IFN-γ production by CD4+ T cells. Tumor necrosis factor α is a key proinflammatory cytokine implicated in the pathogenesis of psoriasis. COVID-19 messenger RNA vaccines induce elevation of IL-6 and helper T cells (TH17), which can induce a flare of psoriasis in a subset of patients.5The International Psoriasis Council recommends that patients with psoriasis receive one of the vaccines approved to prevent COVID-19 infection as soon as possible.6 Reports of new-onset psoriasis and flare of psoriasis after the use of COVID-19 vaccines, such as those manufactured by Pfizer-BioNTech, Moderna, and AstraZeneca, have been published from different parts of the world.7 India used locally developed whole virion inactivated BBV152 (Covaxin) and nonreplicating viral vaccine ChAdOx1 nCoV-19 (Covishield) in its vaccination program and exported them to other developing countries. There is a dearth of data on the safety of these vaccines in patients with psoriasis, which needs to be assessed. Later, Covaxin, ZyCoV-D (DNA plasmid vaccine; Cadila Healthcare), and CorbeVax (protein subunit vaccine; Biological E) were approved for usage in children.8 We conducted a cross-sectional study using the direct interview method.
Patients with psoriasis who attended the outpatient department of the Postgraduate Institute of Medical Education and Research (Chandigarh, India) from April 2022 to June 2022 were invited to participate in the study after written informed consent was received. Patients 18 years and older with chronic plaque psoriasis who had received a COVID-19 vaccine dose in the last 90 days were enrolled. Data on demographics, comorbidities, treatment received for psoriasis, vaccination concerns, history of COVID-19 infection, type of vaccine received with doses, adverse effects, and psoriasis flare after receiving the vaccine (considered up to 2 weeks from the date of vaccination) were collected. Ordinal logistic regression was used to identify factors associated with a psoriasis flare following vaccination. P<.05 was considered statistically significant.
A total of 202 patients with chronic plaque psoriasis who received either Covaxin or Covishield were enrolled during the study period. The mean age (SD) was 40.3 (13.1) years, and 149 (73.8%) patients were male. One hundred thirty-five (66.8%) patients completed 2 doses of the vaccine. eTable 1 provides the clinicodemographic details of the patients. Eighty-three (41.1%) patients had a fear of psoriasis flare after vaccination. Seventy-two (35.6%) patients received the vaccine after clearance from their treating physician/dermatologist. One hundred sixty-four (81.2%) patients received the Covishield vaccine, and 38 (18.8%) patients received Covaxin. Eighty-three (41.1%) patients reported flulike symptoms, such as fever, myalgia, or body pain, within the first week of vaccination. Sixty-one (30.2%) patients reported a psoriasis flare after vaccination in the form of new lesions or worsening of pre-existing lesions. Of these patients, 51 reported a flare after receiving the first dose of vaccine, 8 patients reported a flare after receiving the second dose of vaccine, and 2 patients reported a flare after receiving both doses of vaccine. The mean (SD) flare onset was 8.1 (3.4) days after the vaccination. Eighteen patients considered the flare to be severe. Seventeen (8.4%) patients reported a positive history of COVID-19 infection before vaccination. None of the patients reported breakthrough COVID-19 infection or pustular aggravation of psoriasis following the vaccination.
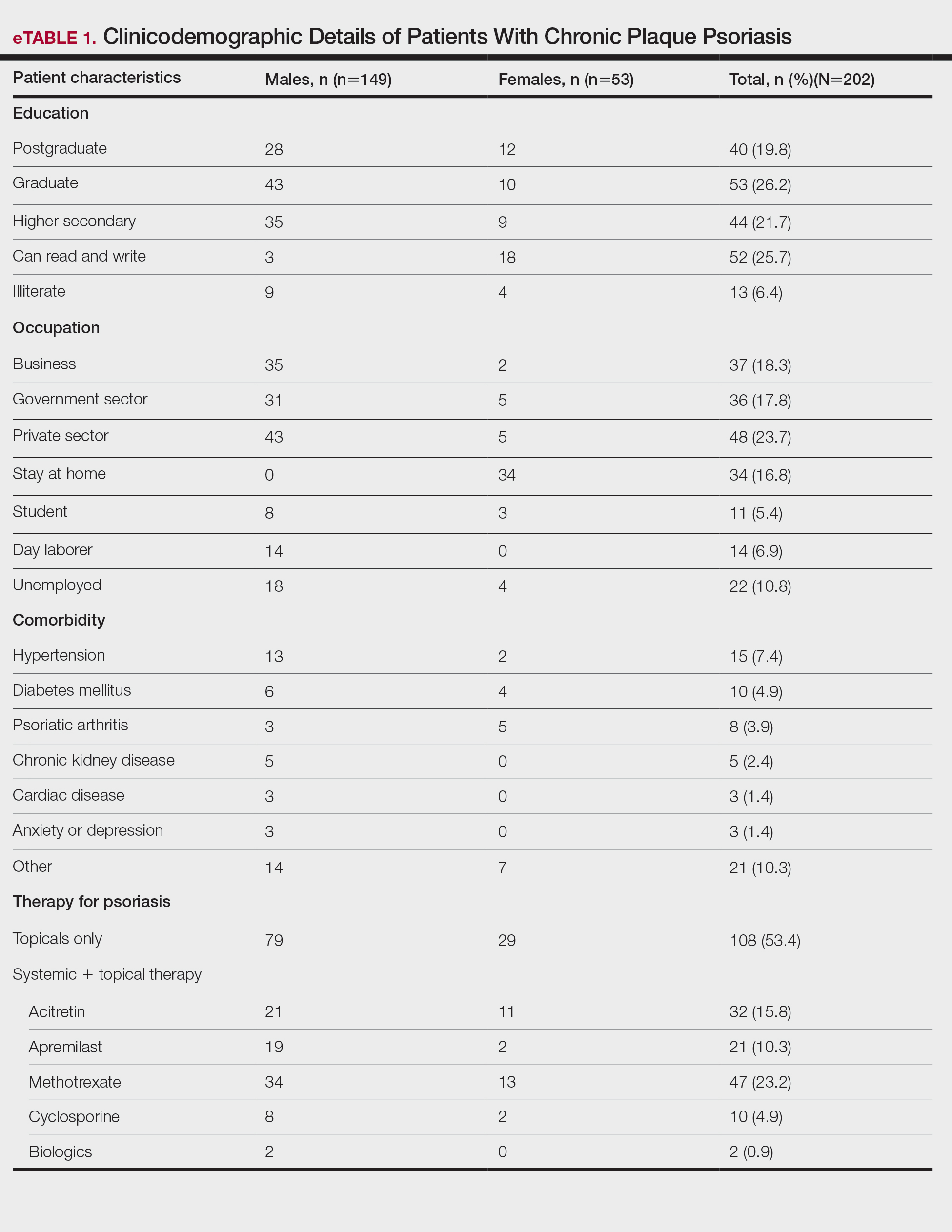
The self-reported psoriasis flare after receiving the COVID-19 vaccine was significantly higher in patients who experienced immediate adverse effects (P=.005), which included fever, myalgia, joint pain, and injection-site reaction. The reported postvaccination psoriasis flare was not significantly associated with patient sex, history of COVID-19 infection, type of vaccine received, comorbidities, or therapy for psoriasis (eTable 2).

Nearly 30% of our patients reported a postvaccination psoriasis flare, which was more common after the first vaccine dose. Sotiriou et al7 reported 14 cases of psoriasis flare in patients after receiving Pfizer-BioNTech, Moderna, and AstraZeneca COVID-19 vaccines. These patients experienced an exacerbation of disease soon after the second dose of vaccine (mean [SD], 10.36 [7.71] days), and 21% of the 713 enrolled patients wanted to forego the immunization due to concern of a postvaccination psoriasis flare.7 In another report, 14 (27%) patients developed a psoriasis flare after COVID-19 vaccination; the mean (SD) flare onset was 9.3 (4.3) days after vaccination.9
Data on the safety of the COVID-19 vaccine in patients using immunosuppressive drugs are limited. We did not find a significant association between the psoriasis flare and use of immunosuppressive drugs or type of vaccine received. Huang and Tsai9 observed similar results, with no association between psoriasis flare and use of immunosuppressive drugs or biologics, while Damiani et al10 demonstrated a protective role of biologics in preventing vaccine-induced psoriasis flare.
Similar to another study from India,11 the immediate adverse effects due to immunization with Covaxin and Covishield were mild in our study and resolved within a week. The incidence of psoriasis flare was significantly higher in patients who reported adverse effects (P=.005). Activation of immune response after vaccination leads to the release of proinflammatory and pyrogenic cytokines (ie, IL-1, IL-6, TNF-α), which may explain the higher incidence of psoriasis flare in patients experiencing adverse effects to vaccination.12
Our study showed approximately 30% of patients developed a psoriasis flare after COVID-19 vaccination, with no patients experiencing any vaccine-related serious adverse events, which suggests that Covaxin and Covishield are safe for patients with psoriasis in India. Limitations of our study include potential inaccuracy of the patient’s self-assessment of symptoms and disease flare, recall bias that may lead to errors in estimating patient-reported outcomes, the flare of psoriasis potentially being a part of disease fluctuation, and flare being enhanced by the psychological stress of vaccination.
Considering a high risk for severe COVID-19 infection in patients with psoriasis with comorbidities and those using immunosuppressive drugs, Covaxin and Covishield can be safely recommended in India. However, caution needs to be exercised when vaccinating patients with an unstable disease or severe psoriasis.
- COVID-19 coronavirus pandemic: weekly trends. Worldometer. Accessed August 21, 2023. https://www.worldometers.info/coronavirus/
- National COVID-19 vaccination programme meets its goals by overcoming R&D and logistical challenges, says economic survey 2022-23. Government of India Press Information Bureau website. Published January 31, 2023. Accessed August 24, 2023. https://pib.gov.in/PressReleasePage.aspx?PRID=1894907
- Ministry of Health and Family Welfare. CoWIN. Accessed August 21, 2023. https://www.cowin.gov.in/
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315.
- Wu D, Yang XO. TH17 responses in cytokine storm of COVID-19: anemerging target of JAK2 inhibitor fedratinib. J Microbiol Immunol Infect. 2020;53:368-370.
- International Psoriasis Council. Revised IPC statement on COVID-19. Published December 19, 2022. Accessed August 24, 2023. https://psoriasiscouncil.org/covid-19/revised-statement-covid-19/
- Sotiriou E, Tsentemeidou A, Bakirtzi K, et al. Psoriasis exacerbation after COVID-19 vaccination: a report of 14 cases from a single centre. J Eur Acad Dermatol Venereol. 2021;35:E857-E859.
- Kaul R. India clears 2 vaccines for kids under 12 years. Hindustan Times. Published April 27, 2022. Accessed August 24, 2023. https://www.hindustantimes.com/india-news/india-clears-2-vaccines-for-kids-under-12-years-101650998027336.html
- Huang YW, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010.
- Damiani G, Allocco F, Young Dermatologists Italian Network, et al. COVID-19 vaccination and patients with psoriasis under biologics: real-life evidence on safety and effectiveness from Italian vaccinated healthcare workers. Clin Exp Dermatol. 2021;460:1106-1108.
- Joshi RK, Muralidharan CG, Gulati DS, et al. Higher incidence of reported adverse events following immunisation (AEFI) after first dose of COVID-19 vaccine among previously infected health care workers. Med J Armed Forces India. 2021;77(suppl 2):S505-S507.
- Hervé C, Laupèze B, Del Giudice G, et al. The how’s and what’s of vaccine reactogenicity. NPJ Vaccines. 2019;4:39.
To the Editor:
COVID-19 infection has resulted in 6.9 million deaths worldwide. India has the third highest mortality from COVID-19 infection after the United States and Brazil.1 Vaccination plays a crucial role in containing COVID-19 infection and reducing its severity. At present, 11 vaccines have been approved by the World Health Organization. India started its vaccination program on January 16, 2021, with approval for use of Covaxin (Bharat Biotech) and Covishield (Oxford/AstraZeneca formulation)(Serum Institute of India). More than 2 billion doses have been administered since then.2,3
Patients with psoriasis are prone to develop a severe form of COVID-19 due to comorbidities and the intake of immunosuppressive drugs.4 These patients often are hesitant to receive the vaccine without an expert opinion. COVID-19 vaccines are considered to increase tumor necrosis factor α (TNF-α) and IFN-γ production by CD4+ T cells. Tumor necrosis factor α is a key proinflammatory cytokine implicated in the pathogenesis of psoriasis. COVID-19 messenger RNA vaccines induce elevation of IL-6 and helper T cells (TH17), which can induce a flare of psoriasis in a subset of patients.5The International Psoriasis Council recommends that patients with psoriasis receive one of the vaccines approved to prevent COVID-19 infection as soon as possible.6 Reports of new-onset psoriasis and flare of psoriasis after the use of COVID-19 vaccines, such as those manufactured by Pfizer-BioNTech, Moderna, and AstraZeneca, have been published from different parts of the world.7 India used locally developed whole virion inactivated BBV152 (Covaxin) and nonreplicating viral vaccine ChAdOx1 nCoV-19 (Covishield) in its vaccination program and exported them to other developing countries. There is a dearth of data on the safety of these vaccines in patients with psoriasis, which needs to be assessed. Later, Covaxin, ZyCoV-D (DNA plasmid vaccine; Cadila Healthcare), and CorbeVax (protein subunit vaccine; Biological E) were approved for usage in children.8 We conducted a cross-sectional study using the direct interview method.
Patients with psoriasis who attended the outpatient department of the Postgraduate Institute of Medical Education and Research (Chandigarh, India) from April 2022 to June 2022 were invited to participate in the study after written informed consent was received. Patients 18 years and older with chronic plaque psoriasis who had received a COVID-19 vaccine dose in the last 90 days were enrolled. Data on demographics, comorbidities, treatment received for psoriasis, vaccination concerns, history of COVID-19 infection, type of vaccine received with doses, adverse effects, and psoriasis flare after receiving the vaccine (considered up to 2 weeks from the date of vaccination) were collected. Ordinal logistic regression was used to identify factors associated with a psoriasis flare following vaccination. P<.05 was considered statistically significant.
A total of 202 patients with chronic plaque psoriasis who received either Covaxin or Covishield were enrolled during the study period. The mean age (SD) was 40.3 (13.1) years, and 149 (73.8%) patients were male. One hundred thirty-five (66.8%) patients completed 2 doses of the vaccine. eTable 1 provides the clinicodemographic details of the patients. Eighty-three (41.1%) patients had a fear of psoriasis flare after vaccination. Seventy-two (35.6%) patients received the vaccine after clearance from their treating physician/dermatologist. One hundred sixty-four (81.2%) patients received the Covishield vaccine, and 38 (18.8%) patients received Covaxin. Eighty-three (41.1%) patients reported flulike symptoms, such as fever, myalgia, or body pain, within the first week of vaccination. Sixty-one (30.2%) patients reported a psoriasis flare after vaccination in the form of new lesions or worsening of pre-existing lesions. Of these patients, 51 reported a flare after receiving the first dose of vaccine, 8 patients reported a flare after receiving the second dose of vaccine, and 2 patients reported a flare after receiving both doses of vaccine. The mean (SD) flare onset was 8.1 (3.4) days after the vaccination. Eighteen patients considered the flare to be severe. Seventeen (8.4%) patients reported a positive history of COVID-19 infection before vaccination. None of the patients reported breakthrough COVID-19 infection or pustular aggravation of psoriasis following the vaccination.

The self-reported psoriasis flare after receiving the COVID-19 vaccine was significantly higher in patients who experienced immediate adverse effects (P=.005), which included fever, myalgia, joint pain, and injection-site reaction. The reported postvaccination psoriasis flare was not significantly associated with patient sex, history of COVID-19 infection, type of vaccine received, comorbidities, or therapy for psoriasis (eTable 2).

Nearly 30% of our patients reported a postvaccination psoriasis flare, which was more common after the first vaccine dose. Sotiriou et al7 reported 14 cases of psoriasis flare in patients after receiving Pfizer-BioNTech, Moderna, and AstraZeneca COVID-19 vaccines. These patients experienced an exacerbation of disease soon after the second dose of vaccine (mean [SD], 10.36 [7.71] days), and 21% of the 713 enrolled patients wanted to forego the immunization due to concern of a postvaccination psoriasis flare.7 In another report, 14 (27%) patients developed a psoriasis flare after COVID-19 vaccination; the mean (SD) flare onset was 9.3 (4.3) days after vaccination.9
Data on the safety of the COVID-19 vaccine in patients using immunosuppressive drugs are limited. We did not find a significant association between the psoriasis flare and use of immunosuppressive drugs or type of vaccine received. Huang and Tsai9 observed similar results, with no association between psoriasis flare and use of immunosuppressive drugs or biologics, while Damiani et al10 demonstrated a protective role of biologics in preventing vaccine-induced psoriasis flare.
Similar to another study from India,11 the immediate adverse effects due to immunization with Covaxin and Covishield were mild in our study and resolved within a week. The incidence of psoriasis flare was significantly higher in patients who reported adverse effects (P=.005). Activation of immune response after vaccination leads to the release of proinflammatory and pyrogenic cytokines (ie, IL-1, IL-6, TNF-α), which may explain the higher incidence of psoriasis flare in patients experiencing adverse effects to vaccination.12
Our study showed approximately 30% of patients developed a psoriasis flare after COVID-19 vaccination, with no patients experiencing any vaccine-related serious adverse events, which suggests that Covaxin and Covishield are safe for patients with psoriasis in India. Limitations of our study include potential inaccuracy of the patient’s self-assessment of symptoms and disease flare, recall bias that may lead to errors in estimating patient-reported outcomes, the flare of psoriasis potentially being a part of disease fluctuation, and flare being enhanced by the psychological stress of vaccination.
Considering a high risk for severe COVID-19 infection in patients with psoriasis with comorbidities and those using immunosuppressive drugs, Covaxin and Covishield can be safely recommended in India. However, caution needs to be exercised when vaccinating patients with an unstable disease or severe psoriasis.
To the Editor:
COVID-19 infection has resulted in 6.9 million deaths worldwide. India has the third highest mortality from COVID-19 infection after the United States and Brazil.1 Vaccination plays a crucial role in containing COVID-19 infection and reducing its severity. At present, 11 vaccines have been approved by the World Health Organization. India started its vaccination program on January 16, 2021, with approval for use of Covaxin (Bharat Biotech) and Covishield (Oxford/AstraZeneca formulation)(Serum Institute of India). More than 2 billion doses have been administered since then.2,3
Patients with psoriasis are prone to develop a severe form of COVID-19 due to comorbidities and the intake of immunosuppressive drugs.4 These patients often are hesitant to receive the vaccine without an expert opinion. COVID-19 vaccines are considered to increase tumor necrosis factor α (TNF-α) and IFN-γ production by CD4+ T cells. Tumor necrosis factor α is a key proinflammatory cytokine implicated in the pathogenesis of psoriasis. COVID-19 messenger RNA vaccines induce elevation of IL-6 and helper T cells (TH17), which can induce a flare of psoriasis in a subset of patients.5The International Psoriasis Council recommends that patients with psoriasis receive one of the vaccines approved to prevent COVID-19 infection as soon as possible.6 Reports of new-onset psoriasis and flare of psoriasis after the use of COVID-19 vaccines, such as those manufactured by Pfizer-BioNTech, Moderna, and AstraZeneca, have been published from different parts of the world.7 India used locally developed whole virion inactivated BBV152 (Covaxin) and nonreplicating viral vaccine ChAdOx1 nCoV-19 (Covishield) in its vaccination program and exported them to other developing countries. There is a dearth of data on the safety of these vaccines in patients with psoriasis, which needs to be assessed. Later, Covaxin, ZyCoV-D (DNA plasmid vaccine; Cadila Healthcare), and CorbeVax (protein subunit vaccine; Biological E) were approved for usage in children.8 We conducted a cross-sectional study using the direct interview method.
Patients with psoriasis who attended the outpatient department of the Postgraduate Institute of Medical Education and Research (Chandigarh, India) from April 2022 to June 2022 were invited to participate in the study after written informed consent was received. Patients 18 years and older with chronic plaque psoriasis who had received a COVID-19 vaccine dose in the last 90 days were enrolled. Data on demographics, comorbidities, treatment received for psoriasis, vaccination concerns, history of COVID-19 infection, type of vaccine received with doses, adverse effects, and psoriasis flare after receiving the vaccine (considered up to 2 weeks from the date of vaccination) were collected. Ordinal logistic regression was used to identify factors associated with a psoriasis flare following vaccination. P<.05 was considered statistically significant.
A total of 202 patients with chronic plaque psoriasis who received either Covaxin or Covishield were enrolled during the study period. The mean age (SD) was 40.3 (13.1) years, and 149 (73.8%) patients were male. One hundred thirty-five (66.8%) patients completed 2 doses of the vaccine. eTable 1 provides the clinicodemographic details of the patients. Eighty-three (41.1%) patients had a fear of psoriasis flare after vaccination. Seventy-two (35.6%) patients received the vaccine after clearance from their treating physician/dermatologist. One hundred sixty-four (81.2%) patients received the Covishield vaccine, and 38 (18.8%) patients received Covaxin. Eighty-three (41.1%) patients reported flulike symptoms, such as fever, myalgia, or body pain, within the first week of vaccination. Sixty-one (30.2%) patients reported a psoriasis flare after vaccination in the form of new lesions or worsening of pre-existing lesions. Of these patients, 51 reported a flare after receiving the first dose of vaccine, 8 patients reported a flare after receiving the second dose of vaccine, and 2 patients reported a flare after receiving both doses of vaccine. The mean (SD) flare onset was 8.1 (3.4) days after the vaccination. Eighteen patients considered the flare to be severe. Seventeen (8.4%) patients reported a positive history of COVID-19 infection before vaccination. None of the patients reported breakthrough COVID-19 infection or pustular aggravation of psoriasis following the vaccination.

The self-reported psoriasis flare after receiving the COVID-19 vaccine was significantly higher in patients who experienced immediate adverse effects (P=.005), which included fever, myalgia, joint pain, and injection-site reaction. The reported postvaccination psoriasis flare was not significantly associated with patient sex, history of COVID-19 infection, type of vaccine received, comorbidities, or therapy for psoriasis (eTable 2).

Nearly 30% of our patients reported a postvaccination psoriasis flare, which was more common after the first vaccine dose. Sotiriou et al7 reported 14 cases of psoriasis flare in patients after receiving Pfizer-BioNTech, Moderna, and AstraZeneca COVID-19 vaccines. These patients experienced an exacerbation of disease soon after the second dose of vaccine (mean [SD], 10.36 [7.71] days), and 21% of the 713 enrolled patients wanted to forego the immunization due to concern of a postvaccination psoriasis flare.7 In another report, 14 (27%) patients developed a psoriasis flare after COVID-19 vaccination; the mean (SD) flare onset was 9.3 (4.3) days after vaccination.9
Data on the safety of the COVID-19 vaccine in patients using immunosuppressive drugs are limited. We did not find a significant association between the psoriasis flare and use of immunosuppressive drugs or type of vaccine received. Huang and Tsai9 observed similar results, with no association between psoriasis flare and use of immunosuppressive drugs or biologics, while Damiani et al10 demonstrated a protective role of biologics in preventing vaccine-induced psoriasis flare.
Similar to another study from India,11 the immediate adverse effects due to immunization with Covaxin and Covishield were mild in our study and resolved within a week. The incidence of psoriasis flare was significantly higher in patients who reported adverse effects (P=.005). Activation of immune response after vaccination leads to the release of proinflammatory and pyrogenic cytokines (ie, IL-1, IL-6, TNF-α), which may explain the higher incidence of psoriasis flare in patients experiencing adverse effects to vaccination.12
Our study showed approximately 30% of patients developed a psoriasis flare after COVID-19 vaccination, with no patients experiencing any vaccine-related serious adverse events, which suggests that Covaxin and Covishield are safe for patients with psoriasis in India. Limitations of our study include potential inaccuracy of the patient’s self-assessment of symptoms and disease flare, recall bias that may lead to errors in estimating patient-reported outcomes, the flare of psoriasis potentially being a part of disease fluctuation, and flare being enhanced by the psychological stress of vaccination.
Considering a high risk for severe COVID-19 infection in patients with psoriasis with comorbidities and those using immunosuppressive drugs, Covaxin and Covishield can be safely recommended in India. However, caution needs to be exercised when vaccinating patients with an unstable disease or severe psoriasis.
- COVID-19 coronavirus pandemic: weekly trends. Worldometer. Accessed August 21, 2023. https://www.worldometers.info/coronavirus/
- National COVID-19 vaccination programme meets its goals by overcoming R&D and logistical challenges, says economic survey 2022-23. Government of India Press Information Bureau website. Published January 31, 2023. Accessed August 24, 2023. https://pib.gov.in/PressReleasePage.aspx?PRID=1894907
- Ministry of Health and Family Welfare. CoWIN. Accessed August 21, 2023. https://www.cowin.gov.in/
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315.
- Wu D, Yang XO. TH17 responses in cytokine storm of COVID-19: anemerging target of JAK2 inhibitor fedratinib. J Microbiol Immunol Infect. 2020;53:368-370.
- International Psoriasis Council. Revised IPC statement on COVID-19. Published December 19, 2022. Accessed August 24, 2023. https://psoriasiscouncil.org/covid-19/revised-statement-covid-19/
- Sotiriou E, Tsentemeidou A, Bakirtzi K, et al. Psoriasis exacerbation after COVID-19 vaccination: a report of 14 cases from a single centre. J Eur Acad Dermatol Venereol. 2021;35:E857-E859.
- Kaul R. India clears 2 vaccines for kids under 12 years. Hindustan Times. Published April 27, 2022. Accessed August 24, 2023. https://www.hindustantimes.com/india-news/india-clears-2-vaccines-for-kids-under-12-years-101650998027336.html
- Huang YW, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010.
- Damiani G, Allocco F, Young Dermatologists Italian Network, et al. COVID-19 vaccination and patients with psoriasis under biologics: real-life evidence on safety and effectiveness from Italian vaccinated healthcare workers. Clin Exp Dermatol. 2021;460:1106-1108.
- Joshi RK, Muralidharan CG, Gulati DS, et al. Higher incidence of reported adverse events following immunisation (AEFI) after first dose of COVID-19 vaccine among previously infected health care workers. Med J Armed Forces India. 2021;77(suppl 2):S505-S507.
- Hervé C, Laupèze B, Del Giudice G, et al. The how’s and what’s of vaccine reactogenicity. NPJ Vaccines. 2019;4:39.
- COVID-19 coronavirus pandemic: weekly trends. Worldometer. Accessed August 21, 2023. https://www.worldometers.info/coronavirus/
- National COVID-19 vaccination programme meets its goals by overcoming R&D and logistical challenges, says economic survey 2022-23. Government of India Press Information Bureau website. Published January 31, 2023. Accessed August 24, 2023. https://pib.gov.in/PressReleasePage.aspx?PRID=1894907
- Ministry of Health and Family Welfare. CoWIN. Accessed August 21, 2023. https://www.cowin.gov.in/
- Griffiths CEM, Armstrong AW, Gudjonsson JE, et al. Psoriasis. Lancet. 2021;397:1301-1315.
- Wu D, Yang XO. TH17 responses in cytokine storm of COVID-19: anemerging target of JAK2 inhibitor fedratinib. J Microbiol Immunol Infect. 2020;53:368-370.
- International Psoriasis Council. Revised IPC statement on COVID-19. Published December 19, 2022. Accessed August 24, 2023. https://psoriasiscouncil.org/covid-19/revised-statement-covid-19/
- Sotiriou E, Tsentemeidou A, Bakirtzi K, et al. Psoriasis exacerbation after COVID-19 vaccination: a report of 14 cases from a single centre. J Eur Acad Dermatol Venereol. 2021;35:E857-E859.
- Kaul R. India clears 2 vaccines for kids under 12 years. Hindustan Times. Published April 27, 2022. Accessed August 24, 2023. https://www.hindustantimes.com/india-news/india-clears-2-vaccines-for-kids-under-12-years-101650998027336.html
- Huang YW, Tsai TF. Exacerbation of psoriasis following COVID-19 vaccination: report from a single center. Front Med (Lausanne). 2021;8:812010.
- Damiani G, Allocco F, Young Dermatologists Italian Network, et al. COVID-19 vaccination and patients with psoriasis under biologics: real-life evidence on safety and effectiveness from Italian vaccinated healthcare workers. Clin Exp Dermatol. 2021;460:1106-1108.
- Joshi RK, Muralidharan CG, Gulati DS, et al. Higher incidence of reported adverse events following immunisation (AEFI) after first dose of COVID-19 vaccine among previously infected health care workers. Med J Armed Forces India. 2021;77(suppl 2):S505-S507.
- Hervé C, Laupèze B, Del Giudice G, et al. The how’s and what’s of vaccine reactogenicity. NPJ Vaccines. 2019;4:39.
Practice Points
- Vaccines are known to induce a psoriasis flare.
- Given the high risk for severe COVID infection in individuals with psoriasis who have comorbidities, vaccination with Covaxin and Covishield can be safely recommended in India for this population.
Financial Insecurity Among US Adults With Psoriasis
To the Editor:
Approximately 3% of the US population, or 6.9 million adults, is affected by psoriasis.1 Psoriasis has a substantial impact on quality of life and is associated with increased health care expenses and medication costs. In 2013, it was reported that the estimated US annual cost—direct, indirect, intangible, and comorbidity costs—of psoriasis for adults was $112 billion.2 We investigated the prevalence and sociodemographic characteristics of adult psoriasis patients (aged ≥20 years) with financial insecurity utilizing the 2009–2014 National Health and Nutrition Examination Survey (NHANES) data.3
We conducted a population-based, cross-sectional study focused on patients 20 years and older with psoriasis from the 2009-2014 NHANES database to evaluate financial insecurity. Financial insecurity was evaluated by 2 outcome variables. The primary outcome variable was assessed by the question “Are you covered by health insurance or some other kind of health care plan (including health insurance obtained through employment or purchased directly as well as government programs like Medicare and Medicaid that provide medical care or help pay medical bills)?”3 Our secondary outcome variable was evaluated by a reported annual household income of less than $20,000. P values in Table 1 were calculated using Pearson χ2 tests. In Table 2, multivariate logistic regressions were performed using Stata/MP 17 (StataCorp LLC) to analyze associations between outcome variables and sociodemographic characteristics. Additionally, we controlled for age, race/ethnicity, sex, education, marital status, US citizenship status, and tobacco use. Subsequently, relationships with P<.05 were considered statistically significant.
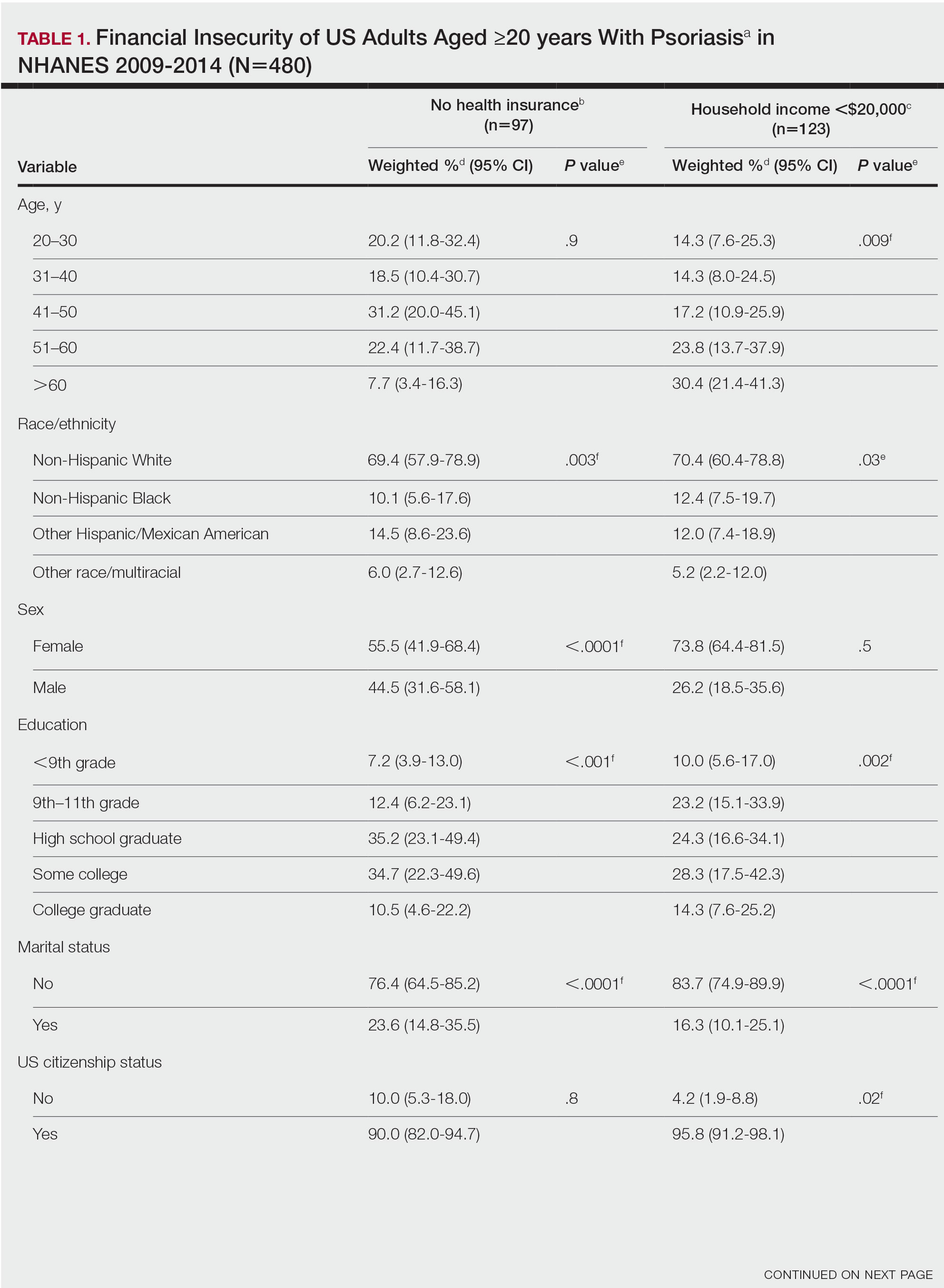

Our analysis comprised 480 individuals with psoriasis; 40 individuals were excluded from our analysis because they did not report annual household income and health insurance status (Table 1). Among the 480 individuals with psoriasis, approximately 16% (weighted) reported a lack of health insurance, and approximately 17% (weighted) reported an annual household income of less than $20,000. Among those who reported an annual household income of less than $20,000, approximately 38% (weighted) of them reported that they did not have health insurance.
Multivariate logistic regression analyses revealed that elderly individuals (aged >60 years), college graduates, married individuals, and US citizens had decreased odds of lacking health insurance (Table 2). Additionally, those with a history of tobacco use (adjusted odds ratio [AOR] 2.02; 95% CI, 1.00-4.05) were associated with lacking health insurance. Non-Hispanic Black individuals (AOR 2.26; 95% CI, 1.09-4.71) and US citizens (AOR 5.01; 95% CI, 1.28-19.63) had a significant association with an annual household income of less than $20,000 (P<.05). Lastly, males, those with education beyond ninth grade, and married individuals had a significantly decreased odds of having an annual household income of less than $20,000 (P<.05)(Table 2).

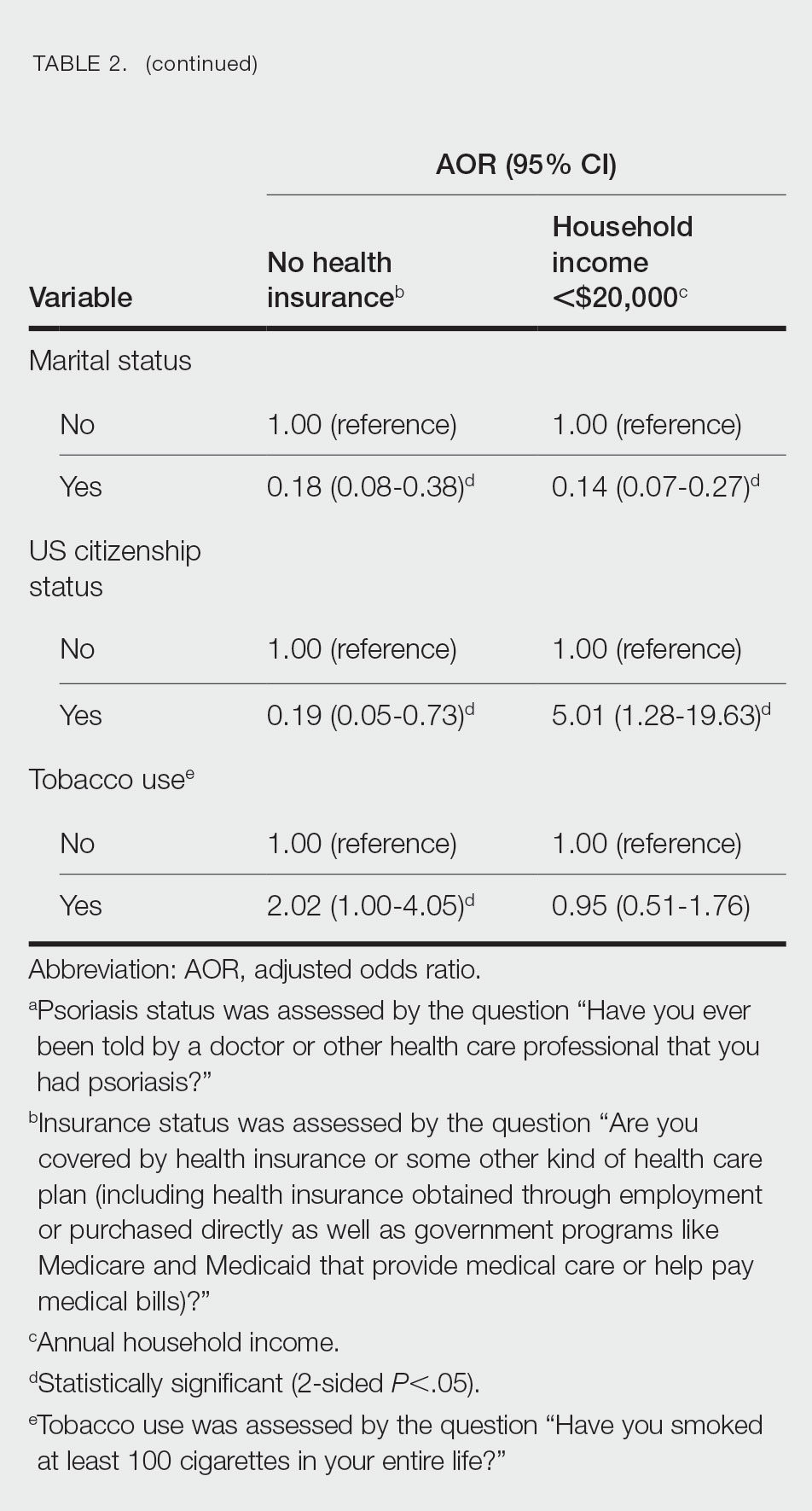
Our findings indicate that certain sociodemographic groups of psoriasis patients have an increased risk for being financially insecure. It is important to evaluate the cost of treatment, number of necessary visits to the office, and cost of transportation, as these factors can serve as a major economic burden to patients being managed for psoriasis.4 Additionally, the cost of biologics has been increasing over time.5 Taking all of this into account when caring for psoriasis patients is crucial, as understanding the financial status of patients can assist with determining appropriate individualized treatment regimens.
- Liu J, Thatiparthi A, Martin A, et al. Prevalence of psoriasis among adults in the US 2009-2010 and 2013-2014 National Health and Nutrition Examination Surveys. J Am Acad Dermatol. 2021;84:767-769. doi:10.1016/j.jaad.2020.10.035
- Brezinski EA, Dhillon JS, Armstrong AW. Economic burden of psoriasis in the United States: a systematic review. JAMA Dermatol. 2015;151:651-658. doi:10.1001/jamadermatol.2014.3593
- National Center for Health Statistics. NHANES questionnaires, datasets, and related documentation. Centers for Disease Control and Prevention website. Accessed June 22, 2023. https://wwwn.cdc.govnchs/nhanes/Default.aspx
- Maya-Rico AM, Londoño-García Á, Palacios-Barahona AU, et al. Out-of-pocket costs for patients with psoriasis in an outpatient dermatology referral service. An Bras Dermatol. 2021;96:295-300. doi:10.1016/j.abd.2020.09.004
- Cheng J, Feldman SR. The cost of biologics for psoriasis is increasing. Drugs Context. 2014;3:212266. doi:10.7573/dic.212266
To the Editor:
Approximately 3% of the US population, or 6.9 million adults, is affected by psoriasis.1 Psoriasis has a substantial impact on quality of life and is associated with increased health care expenses and medication costs. In 2013, it was reported that the estimated US annual cost—direct, indirect, intangible, and comorbidity costs—of psoriasis for adults was $112 billion.2 We investigated the prevalence and sociodemographic characteristics of adult psoriasis patients (aged ≥20 years) with financial insecurity utilizing the 2009–2014 National Health and Nutrition Examination Survey (NHANES) data.3
We conducted a population-based, cross-sectional study focused on patients 20 years and older with psoriasis from the 2009-2014 NHANES database to evaluate financial insecurity. Financial insecurity was evaluated by 2 outcome variables. The primary outcome variable was assessed by the question “Are you covered by health insurance or some other kind of health care plan (including health insurance obtained through employment or purchased directly as well as government programs like Medicare and Medicaid that provide medical care or help pay medical bills)?”3 Our secondary outcome variable was evaluated by a reported annual household income of less than $20,000. P values in Table 1 were calculated using Pearson χ2 tests. In Table 2, multivariate logistic regressions were performed using Stata/MP 17 (StataCorp LLC) to analyze associations between outcome variables and sociodemographic characteristics. Additionally, we controlled for age, race/ethnicity, sex, education, marital status, US citizenship status, and tobacco use. Subsequently, relationships with P<.05 were considered statistically significant.


Our analysis comprised 480 individuals with psoriasis; 40 individuals were excluded from our analysis because they did not report annual household income and health insurance status (Table 1). Among the 480 individuals with psoriasis, approximately 16% (weighted) reported a lack of health insurance, and approximately 17% (weighted) reported an annual household income of less than $20,000. Among those who reported an annual household income of less than $20,000, approximately 38% (weighted) of them reported that they did not have health insurance.
Multivariate logistic regression analyses revealed that elderly individuals (aged >60 years), college graduates, married individuals, and US citizens had decreased odds of lacking health insurance (Table 2). Additionally, those with a history of tobacco use (adjusted odds ratio [AOR] 2.02; 95% CI, 1.00-4.05) were associated with lacking health insurance. Non-Hispanic Black individuals (AOR 2.26; 95% CI, 1.09-4.71) and US citizens (AOR 5.01; 95% CI, 1.28-19.63) had a significant association with an annual household income of less than $20,000 (P<.05). Lastly, males, those with education beyond ninth grade, and married individuals had a significantly decreased odds of having an annual household income of less than $20,000 (P<.05)(Table 2).


Our findings indicate that certain sociodemographic groups of psoriasis patients have an increased risk for being financially insecure. It is important to evaluate the cost of treatment, number of necessary visits to the office, and cost of transportation, as these factors can serve as a major economic burden to patients being managed for psoriasis.4 Additionally, the cost of biologics has been increasing over time.5 Taking all of this into account when caring for psoriasis patients is crucial, as understanding the financial status of patients can assist with determining appropriate individualized treatment regimens.
To the Editor:
Approximately 3% of the US population, or 6.9 million adults, is affected by psoriasis.1 Psoriasis has a substantial impact on quality of life and is associated with increased health care expenses and medication costs. In 2013, it was reported that the estimated US annual cost—direct, indirect, intangible, and comorbidity costs—of psoriasis for adults was $112 billion.2 We investigated the prevalence and sociodemographic characteristics of adult psoriasis patients (aged ≥20 years) with financial insecurity utilizing the 2009–2014 National Health and Nutrition Examination Survey (NHANES) data.3
We conducted a population-based, cross-sectional study focused on patients 20 years and older with psoriasis from the 2009-2014 NHANES database to evaluate financial insecurity. Financial insecurity was evaluated by 2 outcome variables. The primary outcome variable was assessed by the question “Are you covered by health insurance or some other kind of health care plan (including health insurance obtained through employment or purchased directly as well as government programs like Medicare and Medicaid that provide medical care or help pay medical bills)?”3 Our secondary outcome variable was evaluated by a reported annual household income of less than $20,000. P values in Table 1 were calculated using Pearson χ2 tests. In Table 2, multivariate logistic regressions were performed using Stata/MP 17 (StataCorp LLC) to analyze associations between outcome variables and sociodemographic characteristics. Additionally, we controlled for age, race/ethnicity, sex, education, marital status, US citizenship status, and tobacco use. Subsequently, relationships with P<.05 were considered statistically significant.


Our analysis comprised 480 individuals with psoriasis; 40 individuals were excluded from our analysis because they did not report annual household income and health insurance status (Table 1). Among the 480 individuals with psoriasis, approximately 16% (weighted) reported a lack of health insurance, and approximately 17% (weighted) reported an annual household income of less than $20,000. Among those who reported an annual household income of less than $20,000, approximately 38% (weighted) of them reported that they did not have health insurance.
Multivariate logistic regression analyses revealed that elderly individuals (aged >60 years), college graduates, married individuals, and US citizens had decreased odds of lacking health insurance (Table 2). Additionally, those with a history of tobacco use (adjusted odds ratio [AOR] 2.02; 95% CI, 1.00-4.05) were associated with lacking health insurance. Non-Hispanic Black individuals (AOR 2.26; 95% CI, 1.09-4.71) and US citizens (AOR 5.01; 95% CI, 1.28-19.63) had a significant association with an annual household income of less than $20,000 (P<.05). Lastly, males, those with education beyond ninth grade, and married individuals had a significantly decreased odds of having an annual household income of less than $20,000 (P<.05)(Table 2).


Our findings indicate that certain sociodemographic groups of psoriasis patients have an increased risk for being financially insecure. It is important to evaluate the cost of treatment, number of necessary visits to the office, and cost of transportation, as these factors can serve as a major economic burden to patients being managed for psoriasis.4 Additionally, the cost of biologics has been increasing over time.5 Taking all of this into account when caring for psoriasis patients is crucial, as understanding the financial status of patients can assist with determining appropriate individualized treatment regimens.
- Liu J, Thatiparthi A, Martin A, et al. Prevalence of psoriasis among adults in the US 2009-2010 and 2013-2014 National Health and Nutrition Examination Surveys. J Am Acad Dermatol. 2021;84:767-769. doi:10.1016/j.jaad.2020.10.035
- Brezinski EA, Dhillon JS, Armstrong AW. Economic burden of psoriasis in the United States: a systematic review. JAMA Dermatol. 2015;151:651-658. doi:10.1001/jamadermatol.2014.3593
- National Center for Health Statistics. NHANES questionnaires, datasets, and related documentation. Centers for Disease Control and Prevention website. Accessed June 22, 2023. https://wwwn.cdc.govnchs/nhanes/Default.aspx
- Maya-Rico AM, Londoño-García Á, Palacios-Barahona AU, et al. Out-of-pocket costs for patients with psoriasis in an outpatient dermatology referral service. An Bras Dermatol. 2021;96:295-300. doi:10.1016/j.abd.2020.09.004
- Cheng J, Feldman SR. The cost of biologics for psoriasis is increasing. Drugs Context. 2014;3:212266. doi:10.7573/dic.212266
- Liu J, Thatiparthi A, Martin A, et al. Prevalence of psoriasis among adults in the US 2009-2010 and 2013-2014 National Health and Nutrition Examination Surveys. J Am Acad Dermatol. 2021;84:767-769. doi:10.1016/j.jaad.2020.10.035
- Brezinski EA, Dhillon JS, Armstrong AW. Economic burden of psoriasis in the United States: a systematic review. JAMA Dermatol. 2015;151:651-658. doi:10.1001/jamadermatol.2014.3593
- National Center for Health Statistics. NHANES questionnaires, datasets, and related documentation. Centers for Disease Control and Prevention website. Accessed June 22, 2023. https://wwwn.cdc.govnchs/nhanes/Default.aspx
- Maya-Rico AM, Londoño-García Á, Palacios-Barahona AU, et al. Out-of-pocket costs for patients with psoriasis in an outpatient dermatology referral service. An Bras Dermatol. 2021;96:295-300. doi:10.1016/j.abd.2020.09.004
- Cheng J, Feldman SR. The cost of biologics for psoriasis is increasing. Drugs Context. 2014;3:212266. doi:10.7573/dic.212266
Practice Points
- The economic burden on patients with psoriasis has been rising over time, as the disease impacts many aspects of patients’ lives.
- Various sociodemographic groups among patients with psoriasis are financially insecure. Knowing which groups are at higher risk for poor outcomes due to financial insecurity can assist with appropriate treatment regimens.
Evaluating Pharmacists’ Time Collecting Self-Monitoring Blood Glucose Data
The American Diabetes Association recommends that patients on intensive insulin regimens self-monitor blood glucose (SMBG) to assist in therapy optimization.1 To be useful, SMBG data must be captured by patients, shared with care teams, and used and interpreted by patients and practitioners.2,3 Communication of SMBG data from the patient to practitioner can be challenging. Although technology can help in this process, limitations exist, such as manual data entry into systems, patient and/or practitioner technological challenges (eg, accessing interface), and compatibility and integration between SMBG devices and electronic health record (EHR) systems.4
The Boise Veterans Affairs Medical Center (BVAMC) in Idaho serves more than 100,000 veterans. It includes a main site, community-based outpatient clinics, and a clinical resource hub that provides telehealth services to veterans residing in rural neighboring states. The BVAMC pharmacy department provides both inpatient and outpatient services. At the BVAMC, clinical pharmacist practitioners (CPPs) are independent practitioners who support their care teams in comprehensive medication management and have the ability to initiate, modify, and discontinue drug therapy for referred patients.5 A prominent role of CPPs in primary care teams is to manage patients with uncontrolled diabetes and intensive insulin regimens, in which SMBG data are vital to therapy optimization. As collecting SMBG data from patients is seen anecdotally as time intensive, we determined the mean time spent by CPPs collecting patient SMBG data and its potential implications.
Methods
Pharmacists at BVAMC were asked to estimate and record the following: SMBG data collection method, time spent collecting data, extra time spent documenting or formatting SMBG readings, total patient visit time, and visit type. Time was collected in minutes. Extra time spent documenting or formatting SMBG readings included any additional time formatting or entering data in the clinical note after talking to the patient; if this was done while multitasking and talking to the patient, it was not considered extra time. For total patient visit time, pharmacists were asked to estimate only time spent discussing diabetes care and collecting SMBG data. Visit types were categorized as in-person/face-to-face, telephone, and telehealth using clinical video telehealth (CVT)/VA Video Connect (VVC). Data were collected using a standardized spreadsheet. The spreadsheet was pilot tested by a CPP before distribution to all pharmacists.
CPPs were educated about the project in March 2021 and were asked to record data for a 1-week period between April 5, 2021, and April 30, 2021. One CPP also provided delayed data collected from May 17 to 21, 2021, and these data were included in our analysis.
Descriptive statistics were used to determine the mean time spent by CPPs collecting SMBG data. Unpaired t tests were used to compare time spent collecting SMBG data by different collection methods and patient visit types. A P value of ≤ .05 was considered statistically significant. Data were organized in Microsoft Excel, and statistics were completed with JMP Pro v15.
Results
Eight CPPs provided data from 120 patient encounters. For all pa
When compared by the SMBG collection method, the longest time spent collecting SMBG data was with patient report (3.7 minutes), and the longest time spent documenting/formatting time was with meter download/home telehealth (2 minutes). There was no statistically significant difference in the time to collect SMBG data between patient report and other methods (3.7 minutes vs 2.8 minutes; P = .07).
When compared by visit type, there was not a statistically significant difference between time spent collecting SMBG data (3.8 minutes vs 3.2 minutes; P = .39) (Table 2).
Discussion
We found that the mean amount of time spent collecting and documenting/formatting SMBG data was only 4.6 minutes; however, this still represented a substantial portion of visit time. For telephone and CVT/VVC appointments, this represented > 25% of total visit time. While CPPs make important contributions to interprofessional team management of patients with diabetes, their cost is not trivial.6-8 It is worth exploring the most effective and efficient ways to use CPPs. Our results indicate that streamlining SMBG data collection may be beneficial.
Pharmacy technicians, licensed practical nurses/clinical associates, registered nurses/nurse care managers, or other team members could help improve SMBG data collection. Using other team members is also an opportunity for comanagement, for team collaboration, and for more patients to be seen. For example, if a CPP currently has 12 patient encounters that last 20 minutes each, this results in about 240 minutes of direct patient care. If patient encounters were 16 minutes, CPPS could have 15 patient encounters in 240 minutes. Saved time could be used for other clinical tasks involved in disease management or clinical reminder reviews. While there are benefits to CPPs collecting SMBG data, such as further inquiry about patient-reported values, other team members could also be trained to ask appropriate follow-up questions for abnormal blood glucose readings. In addition, leveraging current team members and optimizing their roles could prevent the need to acquire additional full-time equivalent employees.
Another opportunity to increase efficiency in SMBG data collection is with SMBG devices and EHR integration.4,9 However, integration can be difficult with different types of SMBG devices and EHR platforms. Education for patients and practitioners could help to ensure accurate and reliable data uploads; patient internet availability; data protection, privacy, and sharing; workflow management; and clear patient-practitioner expectations.10 For example, if patient SMBG data are automatically uploaded to practitioners, patients’ expectations for practitioner review of data and follow-up need to be determined.
We found a subset of patient encounters (n = 23) where data collection and documenting/formatting represented more than half of the total visit time. In this subset, 13 SMBG reports were pulled from a log or meter, 8 were patient reported, and 3 were meter download or home telehealth.
Limitations
A potential reason for the lack of statistically significant differences in SMBG collection method or visit type in this study includes the small sample size. Participation in this work was voluntary, and all participating CPPs had ≥ 3 years of practice in their current setting, which includes a heavy workload of diabetes management. These pharmacists noted self-established procedures/systems for SMBG data collection, including the use of Excel spreadsheets with pregenerated formulas. For less experienced CPPs, SMBG data collection time may be even longer. Pharmacists also noted that they may limit time spent collecting SMBG data depending on the patient encounter and whether they have gathered sufficient data to guide clinical care. Other limitations of this work include data collection from a single institution and that the time documented represented estimates; there was no external monitor.
Conclusions
In this analysis, we found that CPPs spend about 3 minutes collecting SMBG data from patients, and about an additional 1 minute documenting and formatting data. While 4 to 5 minutes may not represent a substantial amount of time for one patient, it can be when multiplied by several patient encounters. The time spent collecting SMBG data did not significantly differ by collection method or visit type. Opportunities to increase efficiency in SMBG data collection, such as the use of nonpharmacist team members are worth exploring.
Acknowledgments
Thank you to the pharmacists at the Boise Veterans Affairs Medical Center for their time and support of this work: Danielle Ahlstrom, Paul Black, Robyn Cruz, Sarah Naidoo, Anthony Nelson, Laura Spoutz, Eileen Twomey, Donovan Victorine, and Michelle Wilkin.
1. American Diabetes Association. 7. Diabetes Technology: Standards of Medical Care in Diabetes-2021. Diabetes Care. 2021;44(suppl 1):S85-S99. doi:10.2337/dc21-S007
2. Austin MM. The two skill sets of self-monitoring of blood glucose education: the operational and the interpretive. Diabetes Spectr. 2013;26(2):83-90. doi:10.2337/diaspect.26.2.83
3. Gallichan M. Self monitoring of glucose by people with diabetes: evidence based practice. BMJ. 1997;314(7085):964-967. doi:10.1136/bmj.314.7085.964
4. Lewinski AA, Drake C, Shaw RJ, et al. Bridging the integration gap between patient-generated blood glucose data and electronic health records. J Am Med Inform Assoc. 2019;26(7):667-672. doi:10.1093/jamia/ocz039
5. McFarland MS, Groppi J, Jorgenson T, et al. Role of the US Veterans Health Administration clinical pharmacy specialist provider: shaping the future of comprehensive medication management. Can J Hosp Pharm. 2020;73(2):152-158. doi:10.4212/cjhp.v73i2.2982
6. Schmidt K, Caudill J. Hamilton T. Impact of clinical pharmacy specialists on glycemic control in veterans with type 2 diabetes. Am J Health Syst Pharm. 2019;76(suppl 1):S9-S14. doi:10.1093/ajhp/zxy015
7. Sullivan J, Jett BP, Cradick M, Zuber J. Effect of clinical pharmacist intervention on hemoglobin A1c reduction in veteran patients with type 2 diabetes in a rural setting. Ann Pharmacother. 2016;50(12):1023-1027. doi:10.1177/1060028016663564
8. Bloom CI, Ku M, Williams M. Clinical pharmacy specialists’ impact in patient aligned care teams for type 2 diabetes management. J Am Pharm Assoc (2003). 2019;59(5):717-721. doi:10.1016/j.japh.2019.05.002
9. Kumar RB, Goren ND, Stark DE, Wall DP, Longhurst CA. Automated integration of continuous glucose monitor data in the electronic health record using consumer technology. J Am Med Inform Assoc. 2016;23(3):532-537. doi:10.1093/jamia/ocv206
10. Reading MJ, Merrill JA. Converging and diverging needs between patients and providers who are collecting and using patient-generated health data: an integrative review. J Am Med Inform Assoc. 2018;25(6):759-771. doi:10.1093/jamia/ocy006
The American Diabetes Association recommends that patients on intensive insulin regimens self-monitor blood glucose (SMBG) to assist in therapy optimization.1 To be useful, SMBG data must be captured by patients, shared with care teams, and used and interpreted by patients and practitioners.2,3 Communication of SMBG data from the patient to practitioner can be challenging. Although technology can help in this process, limitations exist, such as manual data entry into systems, patient and/or practitioner technological challenges (eg, accessing interface), and compatibility and integration between SMBG devices and electronic health record (EHR) systems.4
The Boise Veterans Affairs Medical Center (BVAMC) in Idaho serves more than 100,000 veterans. It includes a main site, community-based outpatient clinics, and a clinical resource hub that provides telehealth services to veterans residing in rural neighboring states. The BVAMC pharmacy department provides both inpatient and outpatient services. At the BVAMC, clinical pharmacist practitioners (CPPs) are independent practitioners who support their care teams in comprehensive medication management and have the ability to initiate, modify, and discontinue drug therapy for referred patients.5 A prominent role of CPPs in primary care teams is to manage patients with uncontrolled diabetes and intensive insulin regimens, in which SMBG data are vital to therapy optimization. As collecting SMBG data from patients is seen anecdotally as time intensive, we determined the mean time spent by CPPs collecting patient SMBG data and its potential implications.
Methods
Pharmacists at BVAMC were asked to estimate and record the following: SMBG data collection method, time spent collecting data, extra time spent documenting or formatting SMBG readings, total patient visit time, and visit type. Time was collected in minutes. Extra time spent documenting or formatting SMBG readings included any additional time formatting or entering data in the clinical note after talking to the patient; if this was done while multitasking and talking to the patient, it was not considered extra time. For total patient visit time, pharmacists were asked to estimate only time spent discussing diabetes care and collecting SMBG data. Visit types were categorized as in-person/face-to-face, telephone, and telehealth using clinical video telehealth (CVT)/VA Video Connect (VVC). Data were collected using a standardized spreadsheet. The spreadsheet was pilot tested by a CPP before distribution to all pharmacists.
CPPs were educated about the project in March 2021 and were asked to record data for a 1-week period between April 5, 2021, and April 30, 2021. One CPP also provided delayed data collected from May 17 to 21, 2021, and these data were included in our analysis.
Descriptive statistics were used to determine the mean time spent by CPPs collecting SMBG data. Unpaired t tests were used to compare time spent collecting SMBG data by different collection methods and patient visit types. A P value of ≤ .05 was considered statistically significant. Data were organized in Microsoft Excel, and statistics were completed with JMP Pro v15.
Results
Eight CPPs provided data from 120 patient encounters. For all pa
When compared by the SMBG collection method, the longest time spent collecting SMBG data was with patient report (3.7 minutes), and the longest time spent documenting/formatting time was with meter download/home telehealth (2 minutes). There was no statistically significant difference in the time to collect SMBG data between patient report and other methods (3.7 minutes vs 2.8 minutes; P = .07).
When compared by visit type, there was not a statistically significant difference between time spent collecting SMBG data (3.8 minutes vs 3.2 minutes; P = .39) (Table 2).
Discussion
We found that the mean amount of time spent collecting and documenting/formatting SMBG data was only 4.6 minutes; however, this still represented a substantial portion of visit time. For telephone and CVT/VVC appointments, this represented > 25% of total visit time. While CPPs make important contributions to interprofessional team management of patients with diabetes, their cost is not trivial.6-8 It is worth exploring the most effective and efficient ways to use CPPs. Our results indicate that streamlining SMBG data collection may be beneficial.
Pharmacy technicians, licensed practical nurses/clinical associates, registered nurses/nurse care managers, or other team members could help improve SMBG data collection. Using other team members is also an opportunity for comanagement, for team collaboration, and for more patients to be seen. For example, if a CPP currently has 12 patient encounters that last 20 minutes each, this results in about 240 minutes of direct patient care. If patient encounters were 16 minutes, CPPS could have 15 patient encounters in 240 minutes. Saved time could be used for other clinical tasks involved in disease management or clinical reminder reviews. While there are benefits to CPPs collecting SMBG data, such as further inquiry about patient-reported values, other team members could also be trained to ask appropriate follow-up questions for abnormal blood glucose readings. In addition, leveraging current team members and optimizing their roles could prevent the need to acquire additional full-time equivalent employees.
Another opportunity to increase efficiency in SMBG data collection is with SMBG devices and EHR integration.4,9 However, integration can be difficult with different types of SMBG devices and EHR platforms. Education for patients and practitioners could help to ensure accurate and reliable data uploads; patient internet availability; data protection, privacy, and sharing; workflow management; and clear patient-practitioner expectations.10 For example, if patient SMBG data are automatically uploaded to practitioners, patients’ expectations for practitioner review of data and follow-up need to be determined.
We found a subset of patient encounters (n = 23) where data collection and documenting/formatting represented more than half of the total visit time. In this subset, 13 SMBG reports were pulled from a log or meter, 8 were patient reported, and 3 were meter download or home telehealth.
Limitations
A potential reason for the lack of statistically significant differences in SMBG collection method or visit type in this study includes the small sample size. Participation in this work was voluntary, and all participating CPPs had ≥ 3 years of practice in their current setting, which includes a heavy workload of diabetes management. These pharmacists noted self-established procedures/systems for SMBG data collection, including the use of Excel spreadsheets with pregenerated formulas. For less experienced CPPs, SMBG data collection time may be even longer. Pharmacists also noted that they may limit time spent collecting SMBG data depending on the patient encounter and whether they have gathered sufficient data to guide clinical care. Other limitations of this work include data collection from a single institution and that the time documented represented estimates; there was no external monitor.
Conclusions
In this analysis, we found that CPPs spend about 3 minutes collecting SMBG data from patients, and about an additional 1 minute documenting and formatting data. While 4 to 5 minutes may not represent a substantial amount of time for one patient, it can be when multiplied by several patient encounters. The time spent collecting SMBG data did not significantly differ by collection method or visit type. Opportunities to increase efficiency in SMBG data collection, such as the use of nonpharmacist team members are worth exploring.
Acknowledgments
Thank you to the pharmacists at the Boise Veterans Affairs Medical Center for their time and support of this work: Danielle Ahlstrom, Paul Black, Robyn Cruz, Sarah Naidoo, Anthony Nelson, Laura Spoutz, Eileen Twomey, Donovan Victorine, and Michelle Wilkin.
The American Diabetes Association recommends that patients on intensive insulin regimens self-monitor blood glucose (SMBG) to assist in therapy optimization.1 To be useful, SMBG data must be captured by patients, shared with care teams, and used and interpreted by patients and practitioners.2,3 Communication of SMBG data from the patient to practitioner can be challenging. Although technology can help in this process, limitations exist, such as manual data entry into systems, patient and/or practitioner technological challenges (eg, accessing interface), and compatibility and integration between SMBG devices and electronic health record (EHR) systems.4
The Boise Veterans Affairs Medical Center (BVAMC) in Idaho serves more than 100,000 veterans. It includes a main site, community-based outpatient clinics, and a clinical resource hub that provides telehealth services to veterans residing in rural neighboring states. The BVAMC pharmacy department provides both inpatient and outpatient services. At the BVAMC, clinical pharmacist practitioners (CPPs) are independent practitioners who support their care teams in comprehensive medication management and have the ability to initiate, modify, and discontinue drug therapy for referred patients.5 A prominent role of CPPs in primary care teams is to manage patients with uncontrolled diabetes and intensive insulin regimens, in which SMBG data are vital to therapy optimization. As collecting SMBG data from patients is seen anecdotally as time intensive, we determined the mean time spent by CPPs collecting patient SMBG data and its potential implications.
Methods
Pharmacists at BVAMC were asked to estimate and record the following: SMBG data collection method, time spent collecting data, extra time spent documenting or formatting SMBG readings, total patient visit time, and visit type. Time was collected in minutes. Extra time spent documenting or formatting SMBG readings included any additional time formatting or entering data in the clinical note after talking to the patient; if this was done while multitasking and talking to the patient, it was not considered extra time. For total patient visit time, pharmacists were asked to estimate only time spent discussing diabetes care and collecting SMBG data. Visit types were categorized as in-person/face-to-face, telephone, and telehealth using clinical video telehealth (CVT)/VA Video Connect (VVC). Data were collected using a standardized spreadsheet. The spreadsheet was pilot tested by a CPP before distribution to all pharmacists.
CPPs were educated about the project in March 2021 and were asked to record data for a 1-week period between April 5, 2021, and April 30, 2021. One CPP also provided delayed data collected from May 17 to 21, 2021, and these data were included in our analysis.
Descriptive statistics were used to determine the mean time spent by CPPs collecting SMBG data. Unpaired t tests were used to compare time spent collecting SMBG data by different collection methods and patient visit types. A P value of ≤ .05 was considered statistically significant. Data were organized in Microsoft Excel, and statistics were completed with JMP Pro v15.
Results
Eight CPPs provided data from 120 patient encounters. For all pa
When compared by the SMBG collection method, the longest time spent collecting SMBG data was with patient report (3.7 minutes), and the longest time spent documenting/formatting time was with meter download/home telehealth (2 minutes). There was no statistically significant difference in the time to collect SMBG data between patient report and other methods (3.7 minutes vs 2.8 minutes; P = .07).
When compared by visit type, there was not a statistically significant difference between time spent collecting SMBG data (3.8 minutes vs 3.2 minutes; P = .39) (Table 2).
Discussion
We found that the mean amount of time spent collecting and documenting/formatting SMBG data was only 4.6 minutes; however, this still represented a substantial portion of visit time. For telephone and CVT/VVC appointments, this represented > 25% of total visit time. While CPPs make important contributions to interprofessional team management of patients with diabetes, their cost is not trivial.6-8 It is worth exploring the most effective and efficient ways to use CPPs. Our results indicate that streamlining SMBG data collection may be beneficial.
Pharmacy technicians, licensed practical nurses/clinical associates, registered nurses/nurse care managers, or other team members could help improve SMBG data collection. Using other team members is also an opportunity for comanagement, for team collaboration, and for more patients to be seen. For example, if a CPP currently has 12 patient encounters that last 20 minutes each, this results in about 240 minutes of direct patient care. If patient encounters were 16 minutes, CPPS could have 15 patient encounters in 240 minutes. Saved time could be used for other clinical tasks involved in disease management or clinical reminder reviews. While there are benefits to CPPs collecting SMBG data, such as further inquiry about patient-reported values, other team members could also be trained to ask appropriate follow-up questions for abnormal blood glucose readings. In addition, leveraging current team members and optimizing their roles could prevent the need to acquire additional full-time equivalent employees.
Another opportunity to increase efficiency in SMBG data collection is with SMBG devices and EHR integration.4,9 However, integration can be difficult with different types of SMBG devices and EHR platforms. Education for patients and practitioners could help to ensure accurate and reliable data uploads; patient internet availability; data protection, privacy, and sharing; workflow management; and clear patient-practitioner expectations.10 For example, if patient SMBG data are automatically uploaded to practitioners, patients’ expectations for practitioner review of data and follow-up need to be determined.
We found a subset of patient encounters (n = 23) where data collection and documenting/formatting represented more than half of the total visit time. In this subset, 13 SMBG reports were pulled from a log or meter, 8 were patient reported, and 3 were meter download or home telehealth.
Limitations
A potential reason for the lack of statistically significant differences in SMBG collection method or visit type in this study includes the small sample size. Participation in this work was voluntary, and all participating CPPs had ≥ 3 years of practice in their current setting, which includes a heavy workload of diabetes management. These pharmacists noted self-established procedures/systems for SMBG data collection, including the use of Excel spreadsheets with pregenerated formulas. For less experienced CPPs, SMBG data collection time may be even longer. Pharmacists also noted that they may limit time spent collecting SMBG data depending on the patient encounter and whether they have gathered sufficient data to guide clinical care. Other limitations of this work include data collection from a single institution and that the time documented represented estimates; there was no external monitor.
Conclusions
In this analysis, we found that CPPs spend about 3 minutes collecting SMBG data from patients, and about an additional 1 minute documenting and formatting data. While 4 to 5 minutes may not represent a substantial amount of time for one patient, it can be when multiplied by several patient encounters. The time spent collecting SMBG data did not significantly differ by collection method or visit type. Opportunities to increase efficiency in SMBG data collection, such as the use of nonpharmacist team members are worth exploring.
Acknowledgments
Thank you to the pharmacists at the Boise Veterans Affairs Medical Center for their time and support of this work: Danielle Ahlstrom, Paul Black, Robyn Cruz, Sarah Naidoo, Anthony Nelson, Laura Spoutz, Eileen Twomey, Donovan Victorine, and Michelle Wilkin.
1. American Diabetes Association. 7. Diabetes Technology: Standards of Medical Care in Diabetes-2021. Diabetes Care. 2021;44(suppl 1):S85-S99. doi:10.2337/dc21-S007
2. Austin MM. The two skill sets of self-monitoring of blood glucose education: the operational and the interpretive. Diabetes Spectr. 2013;26(2):83-90. doi:10.2337/diaspect.26.2.83
3. Gallichan M. Self monitoring of glucose by people with diabetes: evidence based practice. BMJ. 1997;314(7085):964-967. doi:10.1136/bmj.314.7085.964
4. Lewinski AA, Drake C, Shaw RJ, et al. Bridging the integration gap between patient-generated blood glucose data and electronic health records. J Am Med Inform Assoc. 2019;26(7):667-672. doi:10.1093/jamia/ocz039
5. McFarland MS, Groppi J, Jorgenson T, et al. Role of the US Veterans Health Administration clinical pharmacy specialist provider: shaping the future of comprehensive medication management. Can J Hosp Pharm. 2020;73(2):152-158. doi:10.4212/cjhp.v73i2.2982
6. Schmidt K, Caudill J. Hamilton T. Impact of clinical pharmacy specialists on glycemic control in veterans with type 2 diabetes. Am J Health Syst Pharm. 2019;76(suppl 1):S9-S14. doi:10.1093/ajhp/zxy015
7. Sullivan J, Jett BP, Cradick M, Zuber J. Effect of clinical pharmacist intervention on hemoglobin A1c reduction in veteran patients with type 2 diabetes in a rural setting. Ann Pharmacother. 2016;50(12):1023-1027. doi:10.1177/1060028016663564
8. Bloom CI, Ku M, Williams M. Clinical pharmacy specialists’ impact in patient aligned care teams for type 2 diabetes management. J Am Pharm Assoc (2003). 2019;59(5):717-721. doi:10.1016/j.japh.2019.05.002
9. Kumar RB, Goren ND, Stark DE, Wall DP, Longhurst CA. Automated integration of continuous glucose monitor data in the electronic health record using consumer technology. J Am Med Inform Assoc. 2016;23(3):532-537. doi:10.1093/jamia/ocv206
10. Reading MJ, Merrill JA. Converging and diverging needs between patients and providers who are collecting and using patient-generated health data: an integrative review. J Am Med Inform Assoc. 2018;25(6):759-771. doi:10.1093/jamia/ocy006
1. American Diabetes Association. 7. Diabetes Technology: Standards of Medical Care in Diabetes-2021. Diabetes Care. 2021;44(suppl 1):S85-S99. doi:10.2337/dc21-S007
2. Austin MM. The two skill sets of self-monitoring of blood glucose education: the operational and the interpretive. Diabetes Spectr. 2013;26(2):83-90. doi:10.2337/diaspect.26.2.83
3. Gallichan M. Self monitoring of glucose by people with diabetes: evidence based practice. BMJ. 1997;314(7085):964-967. doi:10.1136/bmj.314.7085.964
4. Lewinski AA, Drake C, Shaw RJ, et al. Bridging the integration gap between patient-generated blood glucose data and electronic health records. J Am Med Inform Assoc. 2019;26(7):667-672. doi:10.1093/jamia/ocz039
5. McFarland MS, Groppi J, Jorgenson T, et al. Role of the US Veterans Health Administration clinical pharmacy specialist provider: shaping the future of comprehensive medication management. Can J Hosp Pharm. 2020;73(2):152-158. doi:10.4212/cjhp.v73i2.2982
6. Schmidt K, Caudill J. Hamilton T. Impact of clinical pharmacy specialists on glycemic control in veterans with type 2 diabetes. Am J Health Syst Pharm. 2019;76(suppl 1):S9-S14. doi:10.1093/ajhp/zxy015
7. Sullivan J, Jett BP, Cradick M, Zuber J. Effect of clinical pharmacist intervention on hemoglobin A1c reduction in veteran patients with type 2 diabetes in a rural setting. Ann Pharmacother. 2016;50(12):1023-1027. doi:10.1177/1060028016663564
8. Bloom CI, Ku M, Williams M. Clinical pharmacy specialists’ impact in patient aligned care teams for type 2 diabetes management. J Am Pharm Assoc (2003). 2019;59(5):717-721. doi:10.1016/j.japh.2019.05.002
9. Kumar RB, Goren ND, Stark DE, Wall DP, Longhurst CA. Automated integration of continuous glucose monitor data in the electronic health record using consumer technology. J Am Med Inform Assoc. 2016;23(3):532-537. doi:10.1093/jamia/ocv206
10. Reading MJ, Merrill JA. Converging and diverging needs between patients and providers who are collecting and using patient-generated health data: an integrative review. J Am Med Inform Assoc. 2018;25(6):759-771. doi:10.1093/jamia/ocy006
The Use of Magnets, Magnetic Fields, and Copper Devices in a Veteran Population
Complementary and alternative medicine (CAM) is a therapeutic approach to health care used in association with or in place of standard medical therapeutic approaches. When describing CAM, the terms complementary and alternative are often used interchangeably, but the terms refer to different concepts. A nonmainstream approach used together with conventional medicine is considered complementary, whereas an approach used in place of conventional medicine is considered alternative. Most people who use nonmainstream approaches also use conventional health care.1
Integrative medicine represents therapeutic interventions that bring conventional and complementary approaches together in a coordinated way. Integrative health also emphasizes multimodal interventions, which are ≥ 2 interventions such as conventional (eg, medication, physical rehabilitation, psychotherapy) and complementary health approaches (eg, acupuncture, yoga, and probiotics) in various combinations, with an emphasis on treating the whole person rather than 1 organ system. Integrative health aims for well-coordinated care among different practitioners and institutions.1
Functional medicine requires an individualized assessment and therapeutic plan for each patient, including optimizing the function of each organ system. It uses research to understand a patient’s unique needs and formulates a plan that often uses diet, exercise, and stress reduction methods. Functional medicine may use combinations of naturopathic, osteopathic, and chiropractic medicine, among other therapies. Functional medicine has been called a systems biology model, and patients and practitioners work together to achieve the highest expression of health by addressing the underlying causes of disease.2,3
According to a 2012 national survey, more than 30% of adults and about 12% of children use health care approaches that are not part of conventional medical care or that may have unconventional origins. A National Center for Health Statistics study found that the most common complementary medical interventions from 2002 to 2012 included natural products, deep breathing, yoga and other movement programs, and chiropractic, among others. Magnets, magnetic fields, and copper devices (MMFC), which are the focus of this study, were not among the top listed interventions.4 Recent data showed that individuals in the United States are high users of CAM, including many patients who have neoplastic disease.5,6
MMFCs are a part of CAM and are reported to be a billion-dollar industry worldwide, although it is not well studied.7,8 In our study, magnet refers to the use of a magnet in contact with the body, magnetic field refers to exposure to a magnetic field administered without direct contact with the body, and copper devices refer to devices that are in contact with the body, such as bracelets, necklaces, wraps, and joint braces. These devices are often constructed using copper mesh, or weaved copper wires. Advertising has helped to increase interest in the use of these devices for musculoskeletal pain and restricted joint movement therapies. However, it is less clear whether MMFCs are being used to provide therapy for other medical conditions, such as neoplastic disease.
It is unclear how widespread MMFC use is or how it is accessed. A 2016 study of veterans and CAM use did not specifically address MMFCs.9 A Japanese study of the use of CAM provided or prescribed by a physician found that just 12 of 1575 respondents (0.7%) described using magnetic therapy.10 A Korean internet study that assessed the use of CAM found that of 1668 respondents who received CAM therapy by practice or advice of a physician, 1.2% used magnet therapy.11,12 An online study of CAM use in patients with multiple sclerosis found that 9 of 1286 respondents (0.7%) had used magnetic field therapy in the previous 3 months.13
In this study, we aimed to assess MMFC use and perspectives in a veteran population at the Carl T. Hayden Veterans Affairs Medical Center (CTHVAMC) in Phoenix, Arizona.
METHODS
We created a brief questionnaire regarding MMFC use and perspectives and distributed it to veteran patients at the infusion center at the CTHVAMC. The study was approved by the CTHVAMC department of research, and the institutional review board determined that informed consent was not required. The questionnaire did not collect any specific personal identifying data but included the participant’s sex, age, and diagnosis. Although there are standardized questionnaires concerning the use of CAM, we designed a new survey for MMFCs. The participants in the study were consecutive patients referred to the CTHVAMC infusion center for IV or other nonoral therapies. Referrals came from endocrinology, gastroenterology, hematology/oncology, neurology, rheumatology, and other specialties (eg, allergy/immunology).
The questionnaire was 1 page (front and back) and was completed anonymously without involvement by the study investigators or infusion center staff. Dated and consecutively numbered questionnaires were given to patients receiving therapy regardless of their diagnosis. Ages were categorized into groups: 18 to 30 years; 31 to 50 years; 51 to 65 years; and ≥ 66 years. Diagnoses were categorized by specialty: endocrinology, gastroenterology, hematology/oncology, neurology, rheumatology, and other. We noted in a previous similar study that the exact diagnosis was often left blank, but the specialty was more often completed.9 Since some patients required multiple visits to the infusion center, respondents were asked whether they had previously answered the questionnaire; there were no duplications.
The population we studied was under stress while receiving therapy for underlying illnesses. To improve the response rate and accuracy of the responses, we limited the number of survey questions. Since many of the respondents in the infusion center for therapy received medications that could alter their ability to respond, all questionnaires were administered prior to therapeutic intervention. In addition to the background data, respondents were asked: Do you apply magnets to your body, use magnetic field therapy, or copper devices? If you use any of these therapies, is it for pain, your diagnosis, or other? Would you consider participating in a clinical trial using magnets applied to the body or magnetic therapy?
RESULTS
We collected 210 surveys. Four surveys were missing data and were excluded. The majority of respondents (n = 133, 64%) were in the hematology/oncology diagnostic group and 121 (59%) were aged ≥ 66 years (Table 1).
Respondents were asked whether they were using MMFC therapies. The results from all age groups showed an 18% overall use and in the diagnosis groups an overall use of 23%. Eighteen respondents (35%) aged 51 to 65 years reported using MMFC, followed by 6 respondents (21%) aged 31 to 50 years. Patients with an endocrinology diagnosis had the highest rate of MMFC use (6 of 11 patients; 55%) but more patients (33 of 133 [25%]) with a hematology/oncology diagnosis used MMFCs.
Copper was the most widely used MMFC therapy among individuals who used a single MMFC therapy. Twenty respondents reported copper use, 6 used magnets, and no respondents used magnetic field therapy (Table 2).
Although we were interested in understanding veterans’ use of these therapies, we were also interested in whether the respondent group would see MMFC as a potential therapy. The highest level of interest in participation in magnet clinical trials was reported by patients aged 31 to 50 years (64%) age group, followed by those aged 51 to 65 (62%). All of the respondents in hematology/oncology, rheumatology, neurology, endocrinology, and gastroenterology groups indicated that they would consider participating in clinical studies using magnets.
DISCUSSION
We surveyed a population of veterans at the CTHVAMC infusion center who were receiving antineoplastic chemotherapy, biologic therapy, immunomodulatory therapy, transfusion, and other therapies to evaluate their use of MMFC. We chose this group to sample because of how accessible this group was and the belief that there would be an adequate survey response. We hypothesized that by asking about a specific group of CAM therapies and not, as in many surveys, multiple CAM therapies, there would be an improved response rate. We expected that very few respondents would indicate MMFC use because in a similar study conducted in 2003 to 2004 at CTHVAMC, none of the 380 survey respondents (all with a hematology/oncology diagnosis) indicated magnet or magnetic field use (JR Salvatore, unpublished data). Although copper devices were available at that time, they were not included in that study. The current survey added copper devices and showed a greater use of MMFC, including copper devices. We identified veterans who used either 1 MMFC or multiple therapies. In both groups, copper devices were the most common. This may be due to the ubiquity and availability of copper devices. These devices are highly visible and promoted by professional athletes and other well-known personalities.
Our findings showed 2 unexpected results. First, there was greater than expected use of magnets and copper devices. Second, an even less expected result that there was considerable interest in participating in clinical research that used magnets or magnetic fields.
Respondents indicated a high interest in participating in clinical trials using magnets or magnetic fields regardless of their history of MMFC use. We did not ask about a trial using copper devices because there is less scientific/medical research to justify studying those devices as opposed to data that support the use of magnets or magnetic fields. The data presented in this study suggest interest in participating in clinical trials using magnets or magnetic field therapy. One clinical trial combined static magnets as an adjuvant to antineoplastic chemotherapy.14 We believe this is the first publication to specifically quantify both MMFC use in a veteran (or any) population, and to identify the desire to participate in clinical studies that would utilize magnets or magnetic fields, whether or not they currently use magnets or magnetic fields. Based on current knowledge, it is not clear whether use of MMFC by patients represents a risk or a benefit to the population studied, and seeking that information is part of the continuation of our work. We also believe that the data in this study will help practitioners to consider asking patients specifically whether they are using these therapies, and if so why and with what result. We are extending our work to a more generalized patient population.
The use of copper devices relates to beliefs (dating to the mid-1800s) that there was a relationship between copper deficiency and rheumatologic disorders. Copper devices are used as therapies because of the belief that small amounts of copper are absorbed through the skin, decreasing inflammation, particularly around joint spaces.15 Recent data suggest a mechanism for copper-induced cell death.16 Although this recent research suggests a mechanism for how copper might induce cell death, it is unclear how this would be applied to establishing a mechanism for the health effects of wearing copper devices. Since copper devices are thought to decrease inflammation, they may have a theoretical function by decreasing the number of inflammatory cells in an affected space.
CAM magnetics are typically of lower strength. The field generated by magnets is measured and reported in Tesla. Magnetic resonance imaging typically generates from 1.5 to 3 Tesla. A refrigerator magnet is about 1 milliTesla.17 In a study conducted at the CTHVAMC, the strength of the magnets used was measured at distances from the magnet. For example, at 2 cm from the magnet, the measured strength was 18 milliTesla.14 Many MMFC devices approved by the US Food and Drug Administration are pulsed electromagnetic fields (PEMF) devices for healing of nonunion fractures (approved in 1979); cervical and lumbar fusion therapies (approved in 2004); and therapy for anxiety and depression (approved in 2006).18
Limitations
Patients with endocrinology diagnoses were the most likely to use MMFCs but were a very small percentage of the infusion center population, which could skew the data. The surveyed individuals may not have been representative of the overall patient population. Similarly, the patient population at CTHVAMC, which is primarily male and aged ≥ 66 years, may not be representative of other veteran and nonveteran patient populations.
Conclusions
MMFC devices are being used regularly by patients as a form of CAM therapy, but few studies researching the use of CAM therapy have generated data that are as specific as this study is about the use of these MMFC devices. Although there is considerable general public awareness of MMFC therapies and devices, we believe that there is a need to quantify the use of these devices. We further believe that our study is one of the first to look specifically at the use of MMFCs in a veteran population. We have found a considerable use of MMFCs in the veteran population studied, and we also showed that whether or not veterans are using these devices, they are willing to be part of research that uses the devices. Further studies would look at a more general veteran population, look more in depth at the way and for what purpose these devices are being used, and consider the development of clinical research studies that use MMFCs.
1. National Institute of Health. National Center for Complementary and Integrative Health. Updated April 2021. Accessed June 26, 2023. https://www.nccih.nih.gov/health/complementary-alternative-or-integrative-health-whats-in-a-name
2. Hanaway P. Form follows function: a functional medicine overview. Perm J. 2016;20(4):16-109. doi:10.7812/TPP/16-109
3. Bland JS. Functional medicine past, present, and future. Integr Med (Encinitas). 2022;21(2):22-26.
4. Clarke TC, Black LI, Stussman BJ, Barnes PM, Nahin RL. Trends in the use of complementary health approaches among adults: United States, 2002-2012. Natl Health Stat Report. 2015;(79):1-16.
5. Horneber M, Bueschel G, Dennert G, Less D, Ritter E, Zwahlen M. How many cancer patients use complementary and alternative medicine: a systematic review and metaanalysis. Integr Cancer Ther. 2012;11(3):187-203. doi:10.1177/1534735411423920
6. Buckner CA, Lafrenie RM, Dénommée JA, Caswell JM, Want DA. Complementary and alternative medicine use in patients before and after a cancer diagnosis. Curr Oncol. 2018;25(4):e275-e281. doi:10.3747/co.25.3884
7. Weintraub MI. Magnetic bio-stimulation in painful diabetic peripheral neuropathy: a novel intervention–a randomized, double-placebo crossover study. Am J Pain Manage. 1999; 9(1):8-17.
8. Colbert AP, Wahbeh H, Harling N, et al. Static magnetic field therapy: a critical review of treatment parameters. Evid Based Complement Alternat Med. 2009;6(2):133-139. doi:10.1093/ecam/nem131
9. Held RF, Santos S, Marki M, Helmer D. Veteran perceptions, interest, and use of complementary and alternative medicine. Fed Pract. 2016;33(9):41-47.
10. Motoo Y, Yukawa K, Arai I, Hisamura K, Tsutani K. Use of complementary and alternative medicine in Japan: a cross-sectional internet survey using the Japanese version of the International Complementary and Alternative Medicine Questionnaire. JMAJ. 2019;2(1):35-46. doi:10.31662/jmaj.2018-0044
11. Quandt SA, Verhoef MJ, Arcury TA, et al. Development of an international questionnaire to measure use of complementary and alternative medicine (I-CAM-Q). J Altern Complement Med. 2009;15(4):331-339. doi:10.1089/acm.2008.0521
12. Lee JA, Sasaki Y, Arai I, et al. An assessment of the use of complementary and alternative medicine by Korean people using an adapted version of the standardized international questionnaire (I-CAM-QK): a cross-sectional study of an internet survey. BMC Complement Altern Med. 2018;18(1):238. Published 2018 Aug 13. doi:10.1186/s12906-018-2294-6
13. Campbell E, Coulter E, Mattison P, McFadyen A, Miller L, Paul L. Access, delivery and perceived efficacy of physiotherapy and use of complementary and alternative therapies by people with progressive multiple sclerosis in the United Kingdom: An online survey. Mult Scler Relat Disord. 2017;12:64-69. doi:10.1016/j.msard.2017.01.002
14. Salvatore JR, Harrington J, Kummet T. Phase I clinical study of a static magnetic field combined with anti-neoplastic chemotherapy in the treatment of human malignancy: initial safety and toxicity data. Bioelectromagnetics. 2003;24(7):524-527. doi:10.1002/bem.10149
15. Richmond SJ, Gunadasa S, Bland M, Macpherson H. Copper bracelets and magnetic wrist straps for rheumatoid arthritis--analgesic and anti-inflammatory effects: a randomised double-blind placebo controlled crossover trial. PLoS One. 2013;8(9):e71529. Published 2013 Sep 16. doi:10.1371/journal.pone.0071529
16. Tsvetkov P, Coy S, Petrova B, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375(6586):1254-1261. doi:10.1126/science.abf0529
17. Simon NJ. Biological Effects of Static Magnetic Fields: A Review. International Cryogenic Materials Commission; 1992:179.
18. Waldorff EI, Zhang N, Ryaby JT. Pulsed electromagnetic field applications: a corporate perspective. J Orthop Translat. 2017;9:60-68. Published 2017 Mar 31. doi:10.1016/j.jot.2017.02.006
Complementary and alternative medicine (CAM) is a therapeutic approach to health care used in association with or in place of standard medical therapeutic approaches. When describing CAM, the terms complementary and alternative are often used interchangeably, but the terms refer to different concepts. A nonmainstream approach used together with conventional medicine is considered complementary, whereas an approach used in place of conventional medicine is considered alternative. Most people who use nonmainstream approaches also use conventional health care.1
Integrative medicine represents therapeutic interventions that bring conventional and complementary approaches together in a coordinated way. Integrative health also emphasizes multimodal interventions, which are ≥ 2 interventions such as conventional (eg, medication, physical rehabilitation, psychotherapy) and complementary health approaches (eg, acupuncture, yoga, and probiotics) in various combinations, with an emphasis on treating the whole person rather than 1 organ system. Integrative health aims for well-coordinated care among different practitioners and institutions.1
Functional medicine requires an individualized assessment and therapeutic plan for each patient, including optimizing the function of each organ system. It uses research to understand a patient’s unique needs and formulates a plan that often uses diet, exercise, and stress reduction methods. Functional medicine may use combinations of naturopathic, osteopathic, and chiropractic medicine, among other therapies. Functional medicine has been called a systems biology model, and patients and practitioners work together to achieve the highest expression of health by addressing the underlying causes of disease.2,3
According to a 2012 national survey, more than 30% of adults and about 12% of children use health care approaches that are not part of conventional medical care or that may have unconventional origins. A National Center for Health Statistics study found that the most common complementary medical interventions from 2002 to 2012 included natural products, deep breathing, yoga and other movement programs, and chiropractic, among others. Magnets, magnetic fields, and copper devices (MMFC), which are the focus of this study, were not among the top listed interventions.4 Recent data showed that individuals in the United States are high users of CAM, including many patients who have neoplastic disease.5,6
MMFCs are a part of CAM and are reported to be a billion-dollar industry worldwide, although it is not well studied.7,8 In our study, magnet refers to the use of a magnet in contact with the body, magnetic field refers to exposure to a magnetic field administered without direct contact with the body, and copper devices refer to devices that are in contact with the body, such as bracelets, necklaces, wraps, and joint braces. These devices are often constructed using copper mesh, or weaved copper wires. Advertising has helped to increase interest in the use of these devices for musculoskeletal pain and restricted joint movement therapies. However, it is less clear whether MMFCs are being used to provide therapy for other medical conditions, such as neoplastic disease.
It is unclear how widespread MMFC use is or how it is accessed. A 2016 study of veterans and CAM use did not specifically address MMFCs.9 A Japanese study of the use of CAM provided or prescribed by a physician found that just 12 of 1575 respondents (0.7%) described using magnetic therapy.10 A Korean internet study that assessed the use of CAM found that of 1668 respondents who received CAM therapy by practice or advice of a physician, 1.2% used magnet therapy.11,12 An online study of CAM use in patients with multiple sclerosis found that 9 of 1286 respondents (0.7%) had used magnetic field therapy in the previous 3 months.13
In this study, we aimed to assess MMFC use and perspectives in a veteran population at the Carl T. Hayden Veterans Affairs Medical Center (CTHVAMC) in Phoenix, Arizona.
METHODS
We created a brief questionnaire regarding MMFC use and perspectives and distributed it to veteran patients at the infusion center at the CTHVAMC. The study was approved by the CTHVAMC department of research, and the institutional review board determined that informed consent was not required. The questionnaire did not collect any specific personal identifying data but included the participant’s sex, age, and diagnosis. Although there are standardized questionnaires concerning the use of CAM, we designed a new survey for MMFCs. The participants in the study were consecutive patients referred to the CTHVAMC infusion center for IV or other nonoral therapies. Referrals came from endocrinology, gastroenterology, hematology/oncology, neurology, rheumatology, and other specialties (eg, allergy/immunology).
The questionnaire was 1 page (front and back) and was completed anonymously without involvement by the study investigators or infusion center staff. Dated and consecutively numbered questionnaires were given to patients receiving therapy regardless of their diagnosis. Ages were categorized into groups: 18 to 30 years; 31 to 50 years; 51 to 65 years; and ≥ 66 years. Diagnoses were categorized by specialty: endocrinology, gastroenterology, hematology/oncology, neurology, rheumatology, and other. We noted in a previous similar study that the exact diagnosis was often left blank, but the specialty was more often completed.9 Since some patients required multiple visits to the infusion center, respondents were asked whether they had previously answered the questionnaire; there were no duplications.
The population we studied was under stress while receiving therapy for underlying illnesses. To improve the response rate and accuracy of the responses, we limited the number of survey questions. Since many of the respondents in the infusion center for therapy received medications that could alter their ability to respond, all questionnaires were administered prior to therapeutic intervention. In addition to the background data, respondents were asked: Do you apply magnets to your body, use magnetic field therapy, or copper devices? If you use any of these therapies, is it for pain, your diagnosis, or other? Would you consider participating in a clinical trial using magnets applied to the body or magnetic therapy?
RESULTS
We collected 210 surveys. Four surveys were missing data and were excluded. The majority of respondents (n = 133, 64%) were in the hematology/oncology diagnostic group and 121 (59%) were aged ≥ 66 years (Table 1).
Respondents were asked whether they were using MMFC therapies. The results from all age groups showed an 18% overall use and in the diagnosis groups an overall use of 23%. Eighteen respondents (35%) aged 51 to 65 years reported using MMFC, followed by 6 respondents (21%) aged 31 to 50 years. Patients with an endocrinology diagnosis had the highest rate of MMFC use (6 of 11 patients; 55%) but more patients (33 of 133 [25%]) with a hematology/oncology diagnosis used MMFCs.
Copper was the most widely used MMFC therapy among individuals who used a single MMFC therapy. Twenty respondents reported copper use, 6 used magnets, and no respondents used magnetic field therapy (Table 2).
Although we were interested in understanding veterans’ use of these therapies, we were also interested in whether the respondent group would see MMFC as a potential therapy. The highest level of interest in participation in magnet clinical trials was reported by patients aged 31 to 50 years (64%) age group, followed by those aged 51 to 65 (62%). All of the respondents in hematology/oncology, rheumatology, neurology, endocrinology, and gastroenterology groups indicated that they would consider participating in clinical studies using magnets.
DISCUSSION
We surveyed a population of veterans at the CTHVAMC infusion center who were receiving antineoplastic chemotherapy, biologic therapy, immunomodulatory therapy, transfusion, and other therapies to evaluate their use of MMFC. We chose this group to sample because of how accessible this group was and the belief that there would be an adequate survey response. We hypothesized that by asking about a specific group of CAM therapies and not, as in many surveys, multiple CAM therapies, there would be an improved response rate. We expected that very few respondents would indicate MMFC use because in a similar study conducted in 2003 to 2004 at CTHVAMC, none of the 380 survey respondents (all with a hematology/oncology diagnosis) indicated magnet or magnetic field use (JR Salvatore, unpublished data). Although copper devices were available at that time, they were not included in that study. The current survey added copper devices and showed a greater use of MMFC, including copper devices. We identified veterans who used either 1 MMFC or multiple therapies. In both groups, copper devices were the most common. This may be due to the ubiquity and availability of copper devices. These devices are highly visible and promoted by professional athletes and other well-known personalities.
Our findings showed 2 unexpected results. First, there was greater than expected use of magnets and copper devices. Second, an even less expected result that there was considerable interest in participating in clinical research that used magnets or magnetic fields.
Respondents indicated a high interest in participating in clinical trials using magnets or magnetic fields regardless of their history of MMFC use. We did not ask about a trial using copper devices because there is less scientific/medical research to justify studying those devices as opposed to data that support the use of magnets or magnetic fields. The data presented in this study suggest interest in participating in clinical trials using magnets or magnetic field therapy. One clinical trial combined static magnets as an adjuvant to antineoplastic chemotherapy.14 We believe this is the first publication to specifically quantify both MMFC use in a veteran (or any) population, and to identify the desire to participate in clinical studies that would utilize magnets or magnetic fields, whether or not they currently use magnets or magnetic fields. Based on current knowledge, it is not clear whether use of MMFC by patients represents a risk or a benefit to the population studied, and seeking that information is part of the continuation of our work. We also believe that the data in this study will help practitioners to consider asking patients specifically whether they are using these therapies, and if so why and with what result. We are extending our work to a more generalized patient population.
The use of copper devices relates to beliefs (dating to the mid-1800s) that there was a relationship between copper deficiency and rheumatologic disorders. Copper devices are used as therapies because of the belief that small amounts of copper are absorbed through the skin, decreasing inflammation, particularly around joint spaces.15 Recent data suggest a mechanism for copper-induced cell death.16 Although this recent research suggests a mechanism for how copper might induce cell death, it is unclear how this would be applied to establishing a mechanism for the health effects of wearing copper devices. Since copper devices are thought to decrease inflammation, they may have a theoretical function by decreasing the number of inflammatory cells in an affected space.
CAM magnetics are typically of lower strength. The field generated by magnets is measured and reported in Tesla. Magnetic resonance imaging typically generates from 1.5 to 3 Tesla. A refrigerator magnet is about 1 milliTesla.17 In a study conducted at the CTHVAMC, the strength of the magnets used was measured at distances from the magnet. For example, at 2 cm from the magnet, the measured strength was 18 milliTesla.14 Many MMFC devices approved by the US Food and Drug Administration are pulsed electromagnetic fields (PEMF) devices for healing of nonunion fractures (approved in 1979); cervical and lumbar fusion therapies (approved in 2004); and therapy for anxiety and depression (approved in 2006).18
Limitations
Patients with endocrinology diagnoses were the most likely to use MMFCs but were a very small percentage of the infusion center population, which could skew the data. The surveyed individuals may not have been representative of the overall patient population. Similarly, the patient population at CTHVAMC, which is primarily male and aged ≥ 66 years, may not be representative of other veteran and nonveteran patient populations.
Conclusions
MMFC devices are being used regularly by patients as a form of CAM therapy, but few studies researching the use of CAM therapy have generated data that are as specific as this study is about the use of these MMFC devices. Although there is considerable general public awareness of MMFC therapies and devices, we believe that there is a need to quantify the use of these devices. We further believe that our study is one of the first to look specifically at the use of MMFCs in a veteran population. We have found a considerable use of MMFCs in the veteran population studied, and we also showed that whether or not veterans are using these devices, they are willing to be part of research that uses the devices. Further studies would look at a more general veteran population, look more in depth at the way and for what purpose these devices are being used, and consider the development of clinical research studies that use MMFCs.
Complementary and alternative medicine (CAM) is a therapeutic approach to health care used in association with or in place of standard medical therapeutic approaches. When describing CAM, the terms complementary and alternative are often used interchangeably, but the terms refer to different concepts. A nonmainstream approach used together with conventional medicine is considered complementary, whereas an approach used in place of conventional medicine is considered alternative. Most people who use nonmainstream approaches also use conventional health care.1
Integrative medicine represents therapeutic interventions that bring conventional and complementary approaches together in a coordinated way. Integrative health also emphasizes multimodal interventions, which are ≥ 2 interventions such as conventional (eg, medication, physical rehabilitation, psychotherapy) and complementary health approaches (eg, acupuncture, yoga, and probiotics) in various combinations, with an emphasis on treating the whole person rather than 1 organ system. Integrative health aims for well-coordinated care among different practitioners and institutions.1
Functional medicine requires an individualized assessment and therapeutic plan for each patient, including optimizing the function of each organ system. It uses research to understand a patient’s unique needs and formulates a plan that often uses diet, exercise, and stress reduction methods. Functional medicine may use combinations of naturopathic, osteopathic, and chiropractic medicine, among other therapies. Functional medicine has been called a systems biology model, and patients and practitioners work together to achieve the highest expression of health by addressing the underlying causes of disease.2,3
According to a 2012 national survey, more than 30% of adults and about 12% of children use health care approaches that are not part of conventional medical care or that may have unconventional origins. A National Center for Health Statistics study found that the most common complementary medical interventions from 2002 to 2012 included natural products, deep breathing, yoga and other movement programs, and chiropractic, among others. Magnets, magnetic fields, and copper devices (MMFC), which are the focus of this study, were not among the top listed interventions.4 Recent data showed that individuals in the United States are high users of CAM, including many patients who have neoplastic disease.5,6
MMFCs are a part of CAM and are reported to be a billion-dollar industry worldwide, although it is not well studied.7,8 In our study, magnet refers to the use of a magnet in contact with the body, magnetic field refers to exposure to a magnetic field administered without direct contact with the body, and copper devices refer to devices that are in contact with the body, such as bracelets, necklaces, wraps, and joint braces. These devices are often constructed using copper mesh, or weaved copper wires. Advertising has helped to increase interest in the use of these devices for musculoskeletal pain and restricted joint movement therapies. However, it is less clear whether MMFCs are being used to provide therapy for other medical conditions, such as neoplastic disease.
It is unclear how widespread MMFC use is or how it is accessed. A 2016 study of veterans and CAM use did not specifically address MMFCs.9 A Japanese study of the use of CAM provided or prescribed by a physician found that just 12 of 1575 respondents (0.7%) described using magnetic therapy.10 A Korean internet study that assessed the use of CAM found that of 1668 respondents who received CAM therapy by practice or advice of a physician, 1.2% used magnet therapy.11,12 An online study of CAM use in patients with multiple sclerosis found that 9 of 1286 respondents (0.7%) had used magnetic field therapy in the previous 3 months.13
In this study, we aimed to assess MMFC use and perspectives in a veteran population at the Carl T. Hayden Veterans Affairs Medical Center (CTHVAMC) in Phoenix, Arizona.
METHODS
We created a brief questionnaire regarding MMFC use and perspectives and distributed it to veteran patients at the infusion center at the CTHVAMC. The study was approved by the CTHVAMC department of research, and the institutional review board determined that informed consent was not required. The questionnaire did not collect any specific personal identifying data but included the participant’s sex, age, and diagnosis. Although there are standardized questionnaires concerning the use of CAM, we designed a new survey for MMFCs. The participants in the study were consecutive patients referred to the CTHVAMC infusion center for IV or other nonoral therapies. Referrals came from endocrinology, gastroenterology, hematology/oncology, neurology, rheumatology, and other specialties (eg, allergy/immunology).
The questionnaire was 1 page (front and back) and was completed anonymously without involvement by the study investigators or infusion center staff. Dated and consecutively numbered questionnaires were given to patients receiving therapy regardless of their diagnosis. Ages were categorized into groups: 18 to 30 years; 31 to 50 years; 51 to 65 years; and ≥ 66 years. Diagnoses were categorized by specialty: endocrinology, gastroenterology, hematology/oncology, neurology, rheumatology, and other. We noted in a previous similar study that the exact diagnosis was often left blank, but the specialty was more often completed.9 Since some patients required multiple visits to the infusion center, respondents were asked whether they had previously answered the questionnaire; there were no duplications.
The population we studied was under stress while receiving therapy for underlying illnesses. To improve the response rate and accuracy of the responses, we limited the number of survey questions. Since many of the respondents in the infusion center for therapy received medications that could alter their ability to respond, all questionnaires were administered prior to therapeutic intervention. In addition to the background data, respondents were asked: Do you apply magnets to your body, use magnetic field therapy, or copper devices? If you use any of these therapies, is it for pain, your diagnosis, or other? Would you consider participating in a clinical trial using magnets applied to the body or magnetic therapy?
RESULTS
We collected 210 surveys. Four surveys were missing data and were excluded. The majority of respondents (n = 133, 64%) were in the hematology/oncology diagnostic group and 121 (59%) were aged ≥ 66 years (Table 1).
Respondents were asked whether they were using MMFC therapies. The results from all age groups showed an 18% overall use and in the diagnosis groups an overall use of 23%. Eighteen respondents (35%) aged 51 to 65 years reported using MMFC, followed by 6 respondents (21%) aged 31 to 50 years. Patients with an endocrinology diagnosis had the highest rate of MMFC use (6 of 11 patients; 55%) but more patients (33 of 133 [25%]) with a hematology/oncology diagnosis used MMFCs.
Copper was the most widely used MMFC therapy among individuals who used a single MMFC therapy. Twenty respondents reported copper use, 6 used magnets, and no respondents used magnetic field therapy (Table 2).
Although we were interested in understanding veterans’ use of these therapies, we were also interested in whether the respondent group would see MMFC as a potential therapy. The highest level of interest in participation in magnet clinical trials was reported by patients aged 31 to 50 years (64%) age group, followed by those aged 51 to 65 (62%). All of the respondents in hematology/oncology, rheumatology, neurology, endocrinology, and gastroenterology groups indicated that they would consider participating in clinical studies using magnets.
DISCUSSION
We surveyed a population of veterans at the CTHVAMC infusion center who were receiving antineoplastic chemotherapy, biologic therapy, immunomodulatory therapy, transfusion, and other therapies to evaluate their use of MMFC. We chose this group to sample because of how accessible this group was and the belief that there would be an adequate survey response. We hypothesized that by asking about a specific group of CAM therapies and not, as in many surveys, multiple CAM therapies, there would be an improved response rate. We expected that very few respondents would indicate MMFC use because in a similar study conducted in 2003 to 2004 at CTHVAMC, none of the 380 survey respondents (all with a hematology/oncology diagnosis) indicated magnet or magnetic field use (JR Salvatore, unpublished data). Although copper devices were available at that time, they were not included in that study. The current survey added copper devices and showed a greater use of MMFC, including copper devices. We identified veterans who used either 1 MMFC or multiple therapies. In both groups, copper devices were the most common. This may be due to the ubiquity and availability of copper devices. These devices are highly visible and promoted by professional athletes and other well-known personalities.
Our findings showed 2 unexpected results. First, there was greater than expected use of magnets and copper devices. Second, an even less expected result that there was considerable interest in participating in clinical research that used magnets or magnetic fields.
Respondents indicated a high interest in participating in clinical trials using magnets or magnetic fields regardless of their history of MMFC use. We did not ask about a trial using copper devices because there is less scientific/medical research to justify studying those devices as opposed to data that support the use of magnets or magnetic fields. The data presented in this study suggest interest in participating in clinical trials using magnets or magnetic field therapy. One clinical trial combined static magnets as an adjuvant to antineoplastic chemotherapy.14 We believe this is the first publication to specifically quantify both MMFC use in a veteran (or any) population, and to identify the desire to participate in clinical studies that would utilize magnets or magnetic fields, whether or not they currently use magnets or magnetic fields. Based on current knowledge, it is not clear whether use of MMFC by patients represents a risk or a benefit to the population studied, and seeking that information is part of the continuation of our work. We also believe that the data in this study will help practitioners to consider asking patients specifically whether they are using these therapies, and if so why and with what result. We are extending our work to a more generalized patient population.
The use of copper devices relates to beliefs (dating to the mid-1800s) that there was a relationship between copper deficiency and rheumatologic disorders. Copper devices are used as therapies because of the belief that small amounts of copper are absorbed through the skin, decreasing inflammation, particularly around joint spaces.15 Recent data suggest a mechanism for copper-induced cell death.16 Although this recent research suggests a mechanism for how copper might induce cell death, it is unclear how this would be applied to establishing a mechanism for the health effects of wearing copper devices. Since copper devices are thought to decrease inflammation, they may have a theoretical function by decreasing the number of inflammatory cells in an affected space.
CAM magnetics are typically of lower strength. The field generated by magnets is measured and reported in Tesla. Magnetic resonance imaging typically generates from 1.5 to 3 Tesla. A refrigerator magnet is about 1 milliTesla.17 In a study conducted at the CTHVAMC, the strength of the magnets used was measured at distances from the magnet. For example, at 2 cm from the magnet, the measured strength was 18 milliTesla.14 Many MMFC devices approved by the US Food and Drug Administration are pulsed electromagnetic fields (PEMF) devices for healing of nonunion fractures (approved in 1979); cervical and lumbar fusion therapies (approved in 2004); and therapy for anxiety and depression (approved in 2006).18
Limitations
Patients with endocrinology diagnoses were the most likely to use MMFCs but were a very small percentage of the infusion center population, which could skew the data. The surveyed individuals may not have been representative of the overall patient population. Similarly, the patient population at CTHVAMC, which is primarily male and aged ≥ 66 years, may not be representative of other veteran and nonveteran patient populations.
Conclusions
MMFC devices are being used regularly by patients as a form of CAM therapy, but few studies researching the use of CAM therapy have generated data that are as specific as this study is about the use of these MMFC devices. Although there is considerable general public awareness of MMFC therapies and devices, we believe that there is a need to quantify the use of these devices. We further believe that our study is one of the first to look specifically at the use of MMFCs in a veteran population. We have found a considerable use of MMFCs in the veteran population studied, and we also showed that whether or not veterans are using these devices, they are willing to be part of research that uses the devices. Further studies would look at a more general veteran population, look more in depth at the way and for what purpose these devices are being used, and consider the development of clinical research studies that use MMFCs.
1. National Institute of Health. National Center for Complementary and Integrative Health. Updated April 2021. Accessed June 26, 2023. https://www.nccih.nih.gov/health/complementary-alternative-or-integrative-health-whats-in-a-name
2. Hanaway P. Form follows function: a functional medicine overview. Perm J. 2016;20(4):16-109. doi:10.7812/TPP/16-109
3. Bland JS. Functional medicine past, present, and future. Integr Med (Encinitas). 2022;21(2):22-26.
4. Clarke TC, Black LI, Stussman BJ, Barnes PM, Nahin RL. Trends in the use of complementary health approaches among adults: United States, 2002-2012. Natl Health Stat Report. 2015;(79):1-16.
5. Horneber M, Bueschel G, Dennert G, Less D, Ritter E, Zwahlen M. How many cancer patients use complementary and alternative medicine: a systematic review and metaanalysis. Integr Cancer Ther. 2012;11(3):187-203. doi:10.1177/1534735411423920
6. Buckner CA, Lafrenie RM, Dénommée JA, Caswell JM, Want DA. Complementary and alternative medicine use in patients before and after a cancer diagnosis. Curr Oncol. 2018;25(4):e275-e281. doi:10.3747/co.25.3884
7. Weintraub MI. Magnetic bio-stimulation in painful diabetic peripheral neuropathy: a novel intervention–a randomized, double-placebo crossover study. Am J Pain Manage. 1999; 9(1):8-17.
8. Colbert AP, Wahbeh H, Harling N, et al. Static magnetic field therapy: a critical review of treatment parameters. Evid Based Complement Alternat Med. 2009;6(2):133-139. doi:10.1093/ecam/nem131
9. Held RF, Santos S, Marki M, Helmer D. Veteran perceptions, interest, and use of complementary and alternative medicine. Fed Pract. 2016;33(9):41-47.
10. Motoo Y, Yukawa K, Arai I, Hisamura K, Tsutani K. Use of complementary and alternative medicine in Japan: a cross-sectional internet survey using the Japanese version of the International Complementary and Alternative Medicine Questionnaire. JMAJ. 2019;2(1):35-46. doi:10.31662/jmaj.2018-0044
11. Quandt SA, Verhoef MJ, Arcury TA, et al. Development of an international questionnaire to measure use of complementary and alternative medicine (I-CAM-Q). J Altern Complement Med. 2009;15(4):331-339. doi:10.1089/acm.2008.0521
12. Lee JA, Sasaki Y, Arai I, et al. An assessment of the use of complementary and alternative medicine by Korean people using an adapted version of the standardized international questionnaire (I-CAM-QK): a cross-sectional study of an internet survey. BMC Complement Altern Med. 2018;18(1):238. Published 2018 Aug 13. doi:10.1186/s12906-018-2294-6
13. Campbell E, Coulter E, Mattison P, McFadyen A, Miller L, Paul L. Access, delivery and perceived efficacy of physiotherapy and use of complementary and alternative therapies by people with progressive multiple sclerosis in the United Kingdom: An online survey. Mult Scler Relat Disord. 2017;12:64-69. doi:10.1016/j.msard.2017.01.002
14. Salvatore JR, Harrington J, Kummet T. Phase I clinical study of a static magnetic field combined with anti-neoplastic chemotherapy in the treatment of human malignancy: initial safety and toxicity data. Bioelectromagnetics. 2003;24(7):524-527. doi:10.1002/bem.10149
15. Richmond SJ, Gunadasa S, Bland M, Macpherson H. Copper bracelets and magnetic wrist straps for rheumatoid arthritis--analgesic and anti-inflammatory effects: a randomised double-blind placebo controlled crossover trial. PLoS One. 2013;8(9):e71529. Published 2013 Sep 16. doi:10.1371/journal.pone.0071529
16. Tsvetkov P, Coy S, Petrova B, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375(6586):1254-1261. doi:10.1126/science.abf0529
17. Simon NJ. Biological Effects of Static Magnetic Fields: A Review. International Cryogenic Materials Commission; 1992:179.
18. Waldorff EI, Zhang N, Ryaby JT. Pulsed electromagnetic field applications: a corporate perspective. J Orthop Translat. 2017;9:60-68. Published 2017 Mar 31. doi:10.1016/j.jot.2017.02.006
1. National Institute of Health. National Center for Complementary and Integrative Health. Updated April 2021. Accessed June 26, 2023. https://www.nccih.nih.gov/health/complementary-alternative-or-integrative-health-whats-in-a-name
2. Hanaway P. Form follows function: a functional medicine overview. Perm J. 2016;20(4):16-109. doi:10.7812/TPP/16-109
3. Bland JS. Functional medicine past, present, and future. Integr Med (Encinitas). 2022;21(2):22-26.
4. Clarke TC, Black LI, Stussman BJ, Barnes PM, Nahin RL. Trends in the use of complementary health approaches among adults: United States, 2002-2012. Natl Health Stat Report. 2015;(79):1-16.
5. Horneber M, Bueschel G, Dennert G, Less D, Ritter E, Zwahlen M. How many cancer patients use complementary and alternative medicine: a systematic review and metaanalysis. Integr Cancer Ther. 2012;11(3):187-203. doi:10.1177/1534735411423920
6. Buckner CA, Lafrenie RM, Dénommée JA, Caswell JM, Want DA. Complementary and alternative medicine use in patients before and after a cancer diagnosis. Curr Oncol. 2018;25(4):e275-e281. doi:10.3747/co.25.3884
7. Weintraub MI. Magnetic bio-stimulation in painful diabetic peripheral neuropathy: a novel intervention–a randomized, double-placebo crossover study. Am J Pain Manage. 1999; 9(1):8-17.
8. Colbert AP, Wahbeh H, Harling N, et al. Static magnetic field therapy: a critical review of treatment parameters. Evid Based Complement Alternat Med. 2009;6(2):133-139. doi:10.1093/ecam/nem131
9. Held RF, Santos S, Marki M, Helmer D. Veteran perceptions, interest, and use of complementary and alternative medicine. Fed Pract. 2016;33(9):41-47.
10. Motoo Y, Yukawa K, Arai I, Hisamura K, Tsutani K. Use of complementary and alternative medicine in Japan: a cross-sectional internet survey using the Japanese version of the International Complementary and Alternative Medicine Questionnaire. JMAJ. 2019;2(1):35-46. doi:10.31662/jmaj.2018-0044
11. Quandt SA, Verhoef MJ, Arcury TA, et al. Development of an international questionnaire to measure use of complementary and alternative medicine (I-CAM-Q). J Altern Complement Med. 2009;15(4):331-339. doi:10.1089/acm.2008.0521
12. Lee JA, Sasaki Y, Arai I, et al. An assessment of the use of complementary and alternative medicine by Korean people using an adapted version of the standardized international questionnaire (I-CAM-QK): a cross-sectional study of an internet survey. BMC Complement Altern Med. 2018;18(1):238. Published 2018 Aug 13. doi:10.1186/s12906-018-2294-6
13. Campbell E, Coulter E, Mattison P, McFadyen A, Miller L, Paul L. Access, delivery and perceived efficacy of physiotherapy and use of complementary and alternative therapies by people with progressive multiple sclerosis in the United Kingdom: An online survey. Mult Scler Relat Disord. 2017;12:64-69. doi:10.1016/j.msard.2017.01.002
14. Salvatore JR, Harrington J, Kummet T. Phase I clinical study of a static magnetic field combined with anti-neoplastic chemotherapy in the treatment of human malignancy: initial safety and toxicity data. Bioelectromagnetics. 2003;24(7):524-527. doi:10.1002/bem.10149
15. Richmond SJ, Gunadasa S, Bland M, Macpherson H. Copper bracelets and magnetic wrist straps for rheumatoid arthritis--analgesic and anti-inflammatory effects: a randomised double-blind placebo controlled crossover trial. PLoS One. 2013;8(9):e71529. Published 2013 Sep 16. doi:10.1371/journal.pone.0071529
16. Tsvetkov P, Coy S, Petrova B, et al. Copper induces cell death by targeting lipoylated TCA cycle proteins. Science. 2022;375(6586):1254-1261. doi:10.1126/science.abf0529
17. Simon NJ. Biological Effects of Static Magnetic Fields: A Review. International Cryogenic Materials Commission; 1992:179.
18. Waldorff EI, Zhang N, Ryaby JT. Pulsed electromagnetic field applications: a corporate perspective. J Orthop Translat. 2017;9:60-68. Published 2017 Mar 31. doi:10.1016/j.jot.2017.02.006